Abstract
Objectives
The epidemiological transition from infectious to chronic diseases leads to novel challenges in African health systems. The prevalence of diabetes mellitus (DM) is increasing dramatically. Undiagnosed and undertreated DM leads to numerous complications including end-organ damage and death. Our objectives were to collect the best locally generated evidence on DM interventions, identify knowledge gaps and determine underexplored research areas.
Design
A systematic review and meta-analysis of randomised controlled trials.
Participants and setting
African patients in primary, secondary and tertiary prevention, diagnosis and treatment DM type 1 (DM1), type 2 (DM2) and gestational DM (GDM).
Outcome
All-cause mortality, glycaemic control, complications, quality of life, hospital admission, treatment adherence and costs.
Data sources
Articles published in MEDLINE Ovid, CENTRAL, CINAHL, African Journals Online and African Index Medicus and the International Clinical Trials Registry Platform in English language without time restrictions. The systematic search was last updated in October 2020.
Results
Out of 3736 identified publications, we included 60 eligible studies conducted in 15 countries, 75% were conducted in urban healthcare settings, including 10 112 participants. We included 8 studies on DM1, 6 on GDM, 2 on pre-DM, 37 on mainly DM2 including 7 on DM-related complications. The design of the studied intervention was heterogeneous with a focus on educational strategies. The other studies investigated the efficacy of nutritional strategies including food supplementations, pharmacological strategies and strategies to enhance physical activity. Seven studies included interventions on DM-related complications.
Conclusions
Research activities increased in recent years, but available evidence is still not representative for all African countries. There is a big lack of evidence in primary healthcare and rural settings, implementation research, pharmacological interventions, especially in poorer countries. Nevertheless, the identified studies offer a variety of effective interventions that can inform medical care and future research.
PROSPERO registration number
CRD42019122785.
Keywords: diabetes in pregnancy, diabetes & endocrinology, general diabetes
Strengths and limitations of this study.
This systematic review includes studies at the highest level of evidence to provide an overview of the best available interventions to prevent, diagnose and treat diabetes mellitus (DM) in the African context.
Inclusion criteria are restricted to randomised controlled trials conducted in African countries published in English language with no restrictions on time of publication.
We performed a systematic search in four international databases and updated the search in October 2020.
The main aim of our systematic review is to provide an overview of interventions for DM. Meta-analyses are restricted to regularly reported results on haemoglobin A1c as strong clinical outcome indicator of an efficient DM management.
Limited external validity due to the origin from few countries and urban areas, results concentrate on glycaemic control due to short follow-up periods.
Introduction
Diabetes mellitus (DM) and other non-communicable diseases (NCDs) are responsible for a double burden in African countries due to the epidemiological transition from communicable to NCDs and resulting disabilities and deaths.1–3 In Africa, around 19.4 million adults are living with DM. Prevalence rates range from 4.7% in sub-Saharan Africa (SSA) to 12.2% in the Middle East and North Africa region.4 Due to the increasing prevalence of risk factors such as obesity and westernised lifestyle, the prevalence of DM is expected to increase by 96% in SSA until 2045.4 Currently, about 50%–60% of adults living with DM in African countries are undiagnosed.4 5 Low awareness as well as genetic differences and lifestyle habits result in very heterogeneous prevalence rates of DM between different countries in Africa as well as rural and urban regions.6 7 Undiagnosed and undertreated DM can result in organ damage, and lead to complications like cardiovascular diseases, peripheral neuropathy, retinopathy and diabetic foot.7 8 Moreover, these factors attribute to substantial financial costs for households and governments.9 Recently, almost one-fifth of COVID-19 deaths in African countries occurred among patients with DM.10
The United Nations 2030 Agenda aims to reduce the burden of premature mortality from NCD including DM through improvement in prevention and treatment.11 Proven and effective actions to prevent or delay the onset of DM base on the empowerment of the population, patients and healthcare providers.12 Measures on DM include early detection in primary healthcare settings, lifestyle modifications including diet, physical activity and, if necessary, medication. Primary prevention programmes include lifestyle measures to reduce consumption of sugar-sweetened beverages, mandatory detailed labels on food packaging as well as education and awareness campaigns to increase physical activity are crucial since onset of DM can be detained.13 Moreover, health systems must ensure technical and financial resources as well as training of healthcare staff to recognise the symptoms of DM, to perform and interpret diagnostic tests and provide adequate treatment and care.4 Since patients with DM need regular specialist assessment, a functioning referral system is necessary.14 Concerning pharmacotherapy, prioritisation of metformin, gliclazide and human insulin is recommended.15 Glucometers, needles and test strips should be provided for people with DM.4
Only a fraction of patients in African countries have access to the same treatment as recommended in high-income countries.16 17 At the moment, most guideline recommendations in low- and middle-income countries (LMIC) are based on studies conducted in high-income Western countries.18 These general management strategies have to be adjusted to local contexts in African countries including environmental, cultural and social aspects like the relatively young age of patients, coinfections, long distances to healthcare facilities, traditional beliefs, decision making in the families and socioeconomic status. Furthermore, there is a huge genetic diversity on the African continent.19 20
The purpose of this review was to collect the best locally generated evidence, regarding preventive, diagnostic and therapeutic intervention on DM, as the lack of evidence is one of the major challenges to prevent and control DM in African countries. Therefore, we aimed to address existing knowledge gaps and identify unexplored research areas in the African context. This may support the formulation of local evidence-based strategies to systematically strengthen clinical and preventive capacities of healthcare systems in African countries.
Methods
This systematic review follows the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses21 and the methods described in the Cochrane Handbook for Systematic Reviews of Interventions.22
Inclusion criteria and exclusion criteria
This systematic review includes studies conducted in African countries on the efficacy of interventions for prevention, diagnosis and treatment of patients with DM including pre-diabetes, type 1 (DM1), type 2 (DM2) and gestational DM (GDM). Primary outcome was defined to be all-cause mortality. Secondary outcomes included glycaemic control (haemoglobin A1c, HbA1c, fasting serum or plasma glucose, insulin resistance, oral glucose tolerance test), quality of life, treatment adherence, hospital admissions, complications of DM and resulting costs (see table 1 for detailed inclusion criteria).
Table 1.
Inclusion and exclusion criteria
| Design and setting | RCTs, mainly conducted in African countries (at least 50% African countries in international studies) |
| Population | African patients in primary, secondary or tertiary prevention with a clinical diagnosis of
As described by the authors |
| Interventions | All interventions to of prevent, diagnose and treat diabetes |
| Comparison | Placebo or standard care Another intervention or the same intervention with a different dose or timing |
| Outcome |
Primary: all-cause mortality Secondary:
Additional: costs at longest follow-up |
| Publications | Full-text publications according to CONSORT |
CONSORT, Consolidated Standards of Reporting Trials; DM2, Type 2 diabetes; DM, diabetes mellitus; DM1, type 1 diabetes; GDM, gestational diabetes; HbA1c, haemoglobin A1c; RCT, randomised controlled trial.
We included full-text publications on randomised controlled trials (RCTs) (eg, individual RCTs, cluster-RCTs and randomised cross-over trials) according to the Consolidated Standards of Reporting Trials23 published in English language. We excluded international multicentre studies with less than 50% of sites in African countries to ensure that the study location was in Africa.
Systematic search
We performed a systematic search in electronic bibliographic databases (MEDLINE Ovid, CENTRAL, International Clinical Trials Registry Platform of the WHO) as planned in the protocol and added a search in CINAHL and regional electronic databases (African Journals Online and African Index Medicus) (see online supplemental file 1). All searches were performed without time constrictions. The last search was conducted in October 2020. Search strings were based on Medical Subject Headings and terms on DM, Africa, a list of all 54 African countries and terms related to RCTs. All references retrieved from the literature search were exported into a reference manager software (EndNote).24 Duplicate references were identified in case of congruence of authors, title, year and journal and thusly deleted. The search strategy is available in online supplemental file.
bmjopen-2021-050021supp006.pdf (251.3KB, pdf)
Study selection and data extraction
Two authors independently checked titles and abstracts based on the inclusion criteria (table 1). The full texts of all potentially eligible papers were assessed for final inclusion. All disagreements were resolved by discussion until consensus was obtained.21 All reported information on the following were extracted and checked by another author:
Publications, registration and design.
Time and place (country, urban/rural setting and healthcare setting).
Study population (inclusion and exclusion criteria, sample size and baseline characteristics on age, gender, type and length of DM, body mass index (BMI) and gylcaemic control at baseline).
Intervention and control groups with the number of randomised participants per group and duration of the interventions.
Outcomes (classified into primary, secondary, non-specified).
Results on preplanned outcomes within the longest follow-up period with intervention effects with their 95% cCIs and level of significance.
The study names were defined by the surname of the first author and the year of the first full-text publication of the results. We compared study and patient characteristics across studies to ensure that each included study represents a unique publication of study data. In cross-over RCTs, only data from the first period were used.25
Quality assessment and risk of bias
Risk of bias was judged based on seven specific categories (sequence generation, allocation concealment, blinding of participants/personnel, blinding of outcome assessors, incomplete outcome data, selective outcome reporting and other sources of bias) of the Cochrane risk of bias tool as ‘low’, ‘high’ or ‘unclear’.22 Judgements were done by two of the authors and all discrepancies were resolved by discussion.
Judgements on blinding and incomplete outcome data were based on the primary outcome of included studies. Selective outcome reporting was defined as low when the study protocol with predefined primary and secondary outcomes was available and high when any result of preplanned outcomes was missing. Incomplete outcome data were judged as high when more than 10% of randomised participants dropped out from analyses. Other sources of bias were judged as high risk of bias including missing reporting of sample size calculation, no description of a primary endpoint, and relevant differences of main baseline characteristics between intervention and control groups.22
Data synthesis
The results of all predefined outcomes were described. Effect sizes on HbA1c for the longest follow-up period were visualised in forest plots using RevMan.26 Negative mean differences (MDs) describe lower HbA1c in the intervention compared with the control group. Statistically significant results on HbA1c with MDs over 0.25% for HbA1c were considered clinically relevant.27 Heterogeneity was interpreted based on the I2 statistics as not important (I2 <30 %), moderate (30%–60%) and substantial (I2 >60 %).22
Patient and public involvement
There is no patient involved.
Results
A total of 2865 references were identified from electronic databases and 871 additional trials from the Clinical Trials Registry Platform were checked. We evaluated 185 potentially eligible full-text publications and included 60 eligible studies in 68 publications in this review (figure 1 and online supplemental file).
Figure 1.
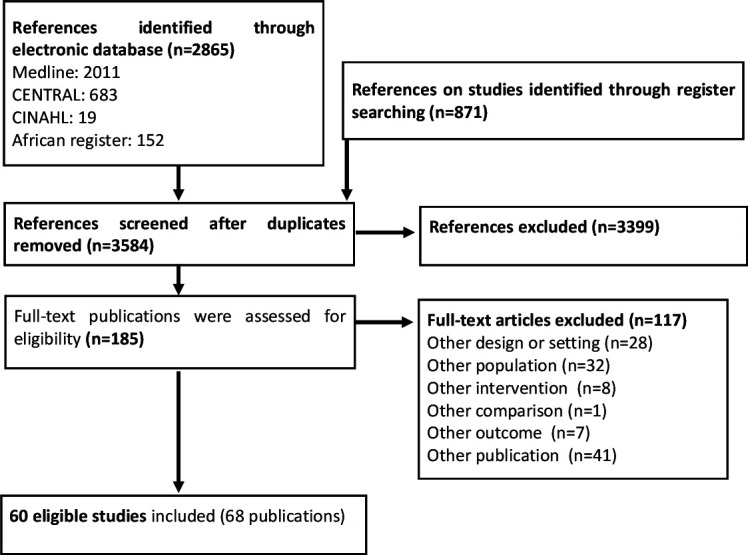
PRISMA flow chart describing the process of study selection. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Setting
In total 60 studies, which were conducted in 64 study centres of 15 African countries; North Africa (33 studies from four countries), West Africa (10 studies from three countries), East Africa (seven studies from 7 countries), Central Africa (3 studies from 2 countries) and Southern Africa (11 studies only from South Africa) were included. Two studies (Malek 2015 and Chraibi 2017) were conducted in more than one African country and partially conducted in non-African countries. Chraibi (2017) was conducted in Egypt, Morocco, South Arabia and Vietnam. Malek included four study centres in Algeria, Tunisia, Egypt and South Africa. Those additional study centres are presented in brackets behind the country names in figure 2. Egypt, South Africa and Nigeria are the three study centres included most often in this review (figure 2 and online supplemenal table 1).
Figure 2.
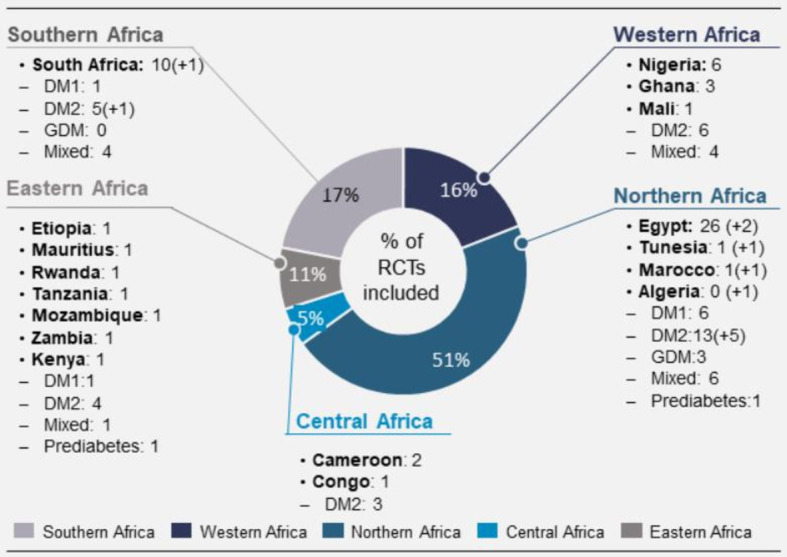
Geographical regions, countries and type of DM of the included studies. DM, diabetes mellitus; DM1, type 1 diabetes mellitus; DM2, type 2 diabetes mellitus; GDM, gestational diabetes mellitus.
bmjopen-2021-050021supp001.pdf (191.3KB, pdf)
Seventy-five per cent of the studies (45/60) were set in urban areas, 5% (3/60) were in rural areas only. The setting of the remaining 20% (12/60 studies) was mixed or remained unclear. The majority, 83% (50/60) of the studies, were conducted in secondary and tertiary healthcare centres, while 17% (10/60) took place in primary care settings.
Though the search had no time restrictions, the oldest eligible study (Anderson 2001) was published in 2001. More than 60% of the studies were published since 2015, and 22% of them had been published in 2019 or 2020 (see online supplemental table 1.)
Design
Fifty parallel-group studies randomised individual participants with DM. Six cluster randomised studies (Fairall 2016, Labhardt 2011, Mash 2014, Steyn 2013, Utz 2018, Webb 2015) randomly assigned healthcare facilities to intervention and control groups. In three randomised cross-over studies (Abdulrhman 2013, Krawinkel 2018, van der Hoogt 2017), each participant received different interventions in a random sequence, and in one study (Ghoneim 2013) each patient received two different treatment doses for each eye based on a random allocation of eyes and doses.
Interventions for patients with pre-DM
Two studies randomised a total of 112 overweight or obese patients (BMI 25–35 kg/m2) with pre-DM (HbA1c 5.7%–7.5%) and a mean age of 32.9 and 47.5 years (see online supplemental table 1: Characteristics and results of studies on patients with pre-DM available in online supplemental file 1). These studies stated the efficacy regarding glycaemic control of low and high volume, high-intensity interval training strategies (RezkAllah 2019), and the consumption of bitter gourd to improve glucose control (Krawinkel 2018).
Interventions for patients with DM1
A total of 8 studies were conducted including 595 patients diagnosed with DM1 (Abdulrhman 2013, Elbarbary 2016, Elbarbary 2018, Elbarbary 2020, Malipa 2013, Mohamad 2009, Salem 2010, van der Hoogt 2017) (see online supplemental table 2: Characteristics and results of studies on patients with DM1 available in online supplemental file). They mainly included children, adolescents, and young adults with a mean age between 10.4 and 19.9 years. The mean duration of DM ranged from 3.5 to 8.6 years and the mean baseline HbA1c from 7.21% to 9.52%. The studies investigated heterogeneous strategies. Malipa 2013 showed the efficacy of weekly meetings to improve treatment compliance, reduce impact and worries about DM and improve general life satisfaction in adolescents. Salem 2010 evaluated the efficacy of two exercise programmes to reduce cardiovascular risk with no relevant effect on glucose control. Three studies investigated different nutritional strategies and stated the beneficial effects of honey (Abdulrhman 2013) and camel milk (Mohamad 2009) on glucose control. Meals with low fat and protein (van der Hoogt 2017) caused less frequent hypoglycaemic events. Elbarbary 2016 showed the efficacy of a low-glucose suspension algorithm during Ramadan to reduce the number of hypoglycaemic and hyperglycaemic excursions. Two studies on food supplementation stated improved glycaemic control with carnosine (Elbarbary 2018), but no benefit from a vitamin B complex (Elbarbary 2020).
bmjopen-2021-050021supp002.pdf (229.7KB, pdf)
Interventions for patients with DM2
A total of 44 studies were conducted including 8881 patients suffering from DM2 or different diabetic illnesses (see online supplemental table 3: Characteristics and results of studies on patients with DM2 availble in online supplemental file 1). Most studies included patients with a mean age between 50 and 60 years, only four studies included younger patients (Adjei 2015, El Gayar 2019, Matter 2020, Maharaj 2016). Most studies included more females than males. These studies presented a wide variety of patients in different stages of DM2 and general conditions. They ranged from newly diagnosed DM (El Gayar 2019, Labhardt 2011, Mostafa 2019, Owolabi 2019, Somanah 2012), non-insulin dependency or oral insulin therapy (Adibe 2013, Ali 2019, Fayehun 2018, Maharaj 2016, Malek 2015, Ragheb 2020) to durations of over 10 years with severe DM-related complications (Abaza 2017, Nteleki 2015, Tsobigny-Tsague 2018, El-Shakawy 2016, Ghoneim 2013, Saeed 2013, Yakoot 2019). Thus, mean baseline HbA1c ranged from 6.75% to 11.1%. Most studies included high proportions of overweight and obese participants with mean BMIs ranging from 22.4 to 40.8 kg/m².
bmjopen-2021-050021supp003.pdf (474.5KB, pdf)
Educational strategies
A total of 19 studies with 6942 patients and follow-up periods between 2 and 14 months investigated the impact of educational strategies on diabetes treatment. These included providing information about lifestyle modification measures, dietary recommendations, drug-based therapy, DM-related complications and self-management. Training sessions were provided based on group-based educational sessions or individual treatment plans by nursing staff or pharmacists and complemented by lectures, discussion services, brochures, newsletters, computer programmes, electronic communication devices and telemonitoring systems. Three of these studies were led by nurses (Adibe 2013, Hailu 2018, Labhardt 2011) and two cluster randomised studies trained nurses to expand their role in the treatment of patients with NCDs (Fairall 2016) or aimed to improve guideline implementation in the treatment of patients with DM (Steyn 2013).
Three studies (Abaza 2017, Adjei 2015, Labhardt 2011) reported results on treatment adherence. All strategies lead to improved adherence, measured by improved perception of patients to treatment recommendations (Abaza 2017) or higher regularity of appointment schedules (Adjei 2015, Labhardt 2011). Two studies (Adibe 2013, Mash 2014) reported results on costs with lower costs for patients receiving educational strategies. Two studies reported fewer admissions to different healthcare facilities (hospital or emergency room and clinic visits) (Abaza 2017, Chraibi 2017).
Results on quality of life were reported in two studies with follow-up periods over 12 months and conflicting results. A structured self-care education programme by pharmacists and nurses (Adibe 2013) improved quality of life, but no benefit was shown after group education by trained professionals (Mash 2014).
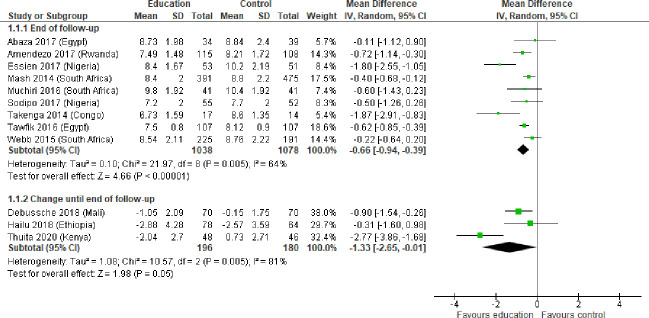
The majority of the educational strategies resulted in lower mean HbA1c levels in the intervention groups with a clinically relevant mean decrease of −0.66% (95% CI −0.94% to −0.39%) and substantial heterogeneity between results of different studies (I2=64 %) (figure 3).
Figure 3.
Results of educational strategies on HbA1c levels or changes of HbA1c levels of patients with DM2. DM2, type 2 diabetes mellitus; HbA1c, haemoglobin A1c.
Strategies to enhance physical activity
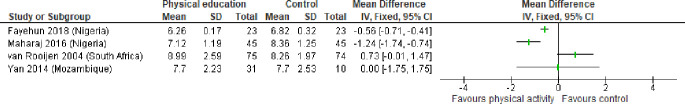
Five studies with 359 participants evaluated the efficacy of different strategies to enhance physical activity on glucose control. Strategies included counselling, setting goals and training sessions with different intensities or both over periods between 8 and 12 weeks.
Two studies were successful in lowering HbA1c where patients were given goals to accumulate 10 000 steps per day Fayehun 2018 or patients were allocated to rebound exercise (Maharaj 2016). A third study investigated the effects of aerobic exercise training and was able to decrease fasting plasma glucose.28
Two other exercise interventions failed to reduce HbA1c by incremental exercises compared with relaxation Van Rooijen 2004 or higher intensity of exercises (Yan 2014) (figure 4). Results were not pooled due to considerable heterogeneity with different directions of treatment effects.
Figure 4.
Results of strategies to enhance physical activity on HbA1c levels of patients with DM2. DM2, type 2 diabetes mellitus; HbA1c, haemoglobin A1c.
Pharmacological strategies
Three studies with 479 participants tested the efficacy of pharmacological treatment strategies on glucose control of patients with DM2. El-Haggar 2015 found ketotifen and glimepiride an effective dual therapy. Malek 2015 described the non-inferiority of once-daily basal-bolus insulin analogues and thrice daily insulin therapy. Distiller 2014 did not find an additional improvement with exenatide in addition to insulin and metformin therapy on glycaemic control.
Strategies on food supplementations
Several different food supplementations were tested in 10 studies including 762 participants. Vitamin D3 supplementation had a positive effect on glycaemic control in two studies (Ali 2019, Anyanwu 2016). Four studies tested the effect of plant-based substances. Ginger powder and balantines aegyptiaca (desert date) extract regimes supported glucose control (El Gayar 2019, Rashad 2017). Nigella sativa (black cumin) oil capsules slightly improved glucose control but were inferior to metformin (Moustafa 2019). A regimen based on fermented papaya did show beneficial results (Somanah 2012). Anderson 2001 and Matter 2020 showed positive effects of zinc/chromium in chronic DM and zinc supplementation in diabetic beta-thalassaemia major patients. The addition of rutin and vitamin C did not improve the results of oral antidiabetics (Ragheb 2020). The addition of l-carnitine improved diabetic control achieved by glimepiride treatment (El-Sheikh 2019).
Strategies on the treatment of DM-related complications
Seven studies with 351 participants and follow-up periods between 3 and 12 months evaluated different strategies to treat possibly DM-related complications including periodontitis (3 studies), foot ulcerations (2 studies) and macular oedema (2 studies).
El-Makaky 2020 and Tsobgny-Tsague 2018 described the benefit of immediate vs delayed non-surgical periodontal interventions on glucose control and El-Sharkawy 2016 stated the effectiveness of propolis as an additive in periodontitis treatment. Two studies stated a benefit of combined phototherapy and podiatric management (Nteleki 2015) and an additional local ointment application of royal jelly and panthenol (Yakoot 2019) on the healing of lower extremity ulcers. Ghoneim 2013 and Saeed 2013 tested different diabetic macular oedema treatment strategies. Both studies described generally positive treatment effects but also considerable adverse events including rise of intraocular pressure and glaucoma.
Interventions for patients with DM in a pregnant woman
Six studies included a total of 574 pregnant women at increased risk for gestational DM (GDM) (Embaby 2016), with newly diagnosed GDM (Utz 2018, El-Shamy 2018, Ashoush 2016) or with newly diagnosed GDM or pre-existing DM (Beyuo 2015, Ibrahim 2014) between the 20th and 34th week of pregnancy. The mean age ranged from 24.2 to 33.3 years (see online supplemental table 4: Characteristics and results of studies on pregnant women with DM available in online supplemental file 1).
bmjopen-2021-050021supp004.pdf (249.7KB, pdf)
Three studies (Ashoush 2016, Beyuo 2015, Ibrahim 2014) with 289 participants examined metformin as an additional medication to insulin in comparison to insulin therapy only. Effects on glycaemic control of metformin supported therapy ranged from a relevant decrease (Ashoush 2016) to no effect on fasting plasma glucose, but beneficial effect on 2-hour plasma glucose in a 75 g OGTT (Beyuo 2015) in women without insulin resistance. Adding metformin to insulin therapy of pregnant women with insulin resistant diabetes was associated with several benefits concerning the time of hospital stay, reduced occurrence of maternal or neonatal hyperglycaemic, less neonatal intensive care unit admissions and reduced cases of respiratory distress syndrome (Ibrahim 2014).
The other studies (285 participants) investigated non-pharmacological interventions. The tested interventions were aerobic exercise programme (treadmill walking) (Embaby 2016), acupressure (El-Shamy 2018) and screening for GDM, followed by nutritional and exercise counselling for positive tested women (Utz 2018). The aerobic exercise programme resulted in a relevant reduction of fasting plasma glucose until delivery (Embaby 2016). The acupressure intervention did not manage to show a benefit regarding glycaemic control (El-Shamy 2018). Screening, counselling and intensive follow-up were able to improve glycaemic control and reduce the number of newborns with macrosomia (Utz 2018).
Potential biases
None of the included studies was categorised as low risk of bias in all seven domains only (see online supplemental table 5: Judgement on risk of bias available in online supplemental file).
bmjopen-2021-050021supp005.pdf (189.1KB, pdf)
The most common restriction on study quality was found in the domain performance bias due to a lack of blinding of participants and personnel in 48 studies. Detection bias due to blinding of the outcome assessors was judged as high or unclear in 38 studies. Fourteen studies with high risk of bias due to no blinding of participants and personnel, reported adequate methods to ensure blinding of the outcome assessors.
Another frequent problem was an incomplete analyses of outcome data in 26 studies defined as a loss to follow-up over 10% of randomised participants or per-protocol analyses.
In 23 studies, a protocol was available. Risk of bias due to selective outcome reporting was judged as low in 15 studies. High risk of bias, meaning lack of reporting of results of some preplanned outcomes was judged in eight studies (Abdulrhman 2013, Beyuo 2015, Elbarbary 2020, Matter 2020, Owolabi 2019, Somanah 2012, Utz 2018 Yakoot 2019).
In the domain sequence generation, two studies were assessed as high risk. El- Nteleki 2015 randomised only seven patients into three different treatment groups. Shamy 2018 used a non-probability sampling method on the basis of the hospital admission code and was subsequently judged as high risk in domains sequence generation and allocation concealment.
In 31 studies, we identified further methodological limitations including missing reporting of information on sample-size calculation, definition of primary and secondary target criteria, relevant differences regarding baseline characteristics or reporting of intermediate results only.
Discussion
This systematic review describes interventions from 60 studies to summarise the available randomised trials on to prevention, diagnosis and treatment of DM with a total of 12 113 participants from 15 African countries. Several promising interventions were identified that can be used in settings with limited resources or involved locally available materials. Despite a trend of increasing research activity in recent years, many areas of diabetes research in African countries are still underexplored leaving knowledge gaps that should be tackled in the future.
Scarcity of randomised DM trials in African countries
While 60 included randomised trials are not nothing it also means an average only slightly higher than 1 randomised DM study per country for all types of diabetes that has ever been conducted and published. Only two studies on prediabetic interventions have been conducted, despite a clear need and aim to tackle early to avoid the future DM burden that is expected to arise.17 Implementation research, considered important in addressing know-do gaps in real-world settings, especially in primary care settings are still very rare.29 Implementing evidence-based care while observing, evaluating and publishing its result deems crucial in the massive challenge of creating diabetes care infrastructure for millions of diabetes patients. Nevertheless, 43 of the 60 studies have been conducted since 2015 demonstrating at positive trend of research activity.
Rural versus urban, primary versus secondary care and geographical disparities
Three out of four studies were set in urban areas and only 5% (3/60) were set in rural areas only. Despite decreasing population shares over the last decades, still almost 60% of people in SSA are living in rural areas with rising absolute numbers (currently about 667 million).30 Despite diabetes being considered to be associated with westernised lifestyle more prevalent in urban areas, prevalence rates in rural areas are still high, in some parts even higher.31 32
Moreover, the majority (83 %) of the studies were conducted in secondary and tertiary healthcare centres, leaving less than one-fifth in primary care settings were most routine and day-to-day diabetes care should be carried out to support people in their everyday life with this chronic long-term illness to prevent long-term consequences.
Another considerable aspect is the geographical distribution of the conducted studies. Almost half (46%) of the included trial were conducted in Egypt, the country ranking second on the African Infrastructure Development Index 2018 with the highest prevalence in Northern Africa.33 South Africa, ranking fourth on the index, contributed another share of 18% (11 studies) (7). Almost three-quarters of the studies were set in the top 10 ranking countries on that list, all Northern and Southern Africa leaving huge blank spaces in Central, Western and Eastern Africa including countries with high prevalences including Kenya and Zimbabwe and pointing to both the infrastructural necessities of research as well as the structural development that is still ahead before to increase research activity.34 The broad majority of included studies was conducted in urban settings, this is likely due to the better healthcare infrastructure and thusly the increased practicability of research. Healthcare workers, including doctors and nurses, seem to prefer providing services in urban areas leading to an even higher deficit of healthcare access in rural areas. The consequence is limited generalisability of the results on the needs of the rural population.
Screening strategies to diagnose DM and its complications
The rate of undiagnosed patients with DM is estimated to be between 3.9% in SSA35 and 12% in North Africa.36 This might be related to genetic disparities in the development level of the healthcare system and awareness in the general population.19 The high rates of undiagnosed DM highlight a high need for research on and implementation of DM screening strategies in the African context. We identified two studies37–39 investigating primary care strategies to detect and manage women with GDM37 and screen diabetic patients for complications.40 The observed GDM prevalence of 23.7% among pregnant Moroccan women underlines the importance of regular screening and management to enable early interventions at a primary care level (37). A diabetic population receiving primary care found a high rate of complications including retinopathy, maculopathy, neuropathy, nephropathy, possible infarction and severe erectile dysfunction.38–40
Intervention for patients with pre-DM for primary prevention of DM
We identified two studies patients41 42 with elevated blood glucose levels below diagnosis criteria of DM improving glucose levels via interval training bitter gourd, a plant with antidiabetic properties that is consumed in many Asian as well as some African countries. Both studies offer effective strategies, but further research is necessary, exemplarily on early educational strategies, as a measure of patient empowerment and early tackling of DM.43
Educational strategies for patients and healthcare providers
Education is essential for effective diabetes control. It must be accomplished at, personal (patient empowement), community (raise the awereness of the disease and its risk factors) and healthcare provider level (training of medical staff to diagnose, monitor and treat it correctely) to manage the rising burden of diabetes.44
Due to complex challenges for patients with DM and healthcare providers, educational campaigns are necessary to support healthcare providers and empower patients to manage their disease-associated decisions, lifestyle habits and medication use. Best benefits are proposed to be achieved by continuous individualised education, guided by patients’ concerns, preferences and needs.12 45
Several studies on DM2,46–58 DM159 and GDM37 investigated long-term interventions to support patient empowerment based on improved knowledge, motivation, and capacity to take control of their disease.12 Three studies trialled nurse led47 53 54 60 and two studies investigated strategies to train healthcare providers in the management of patients with DM.61 62 Improvement of patient empowerment improved adherence and glucose control, fewer admissions to healthcare facilities and lower costs. Only two studies reported on the quality of life with heterogeneous results.47 60 63
Currently, the COVID-19 pandemic has forced all nations to implement alternative, oftentimes digital strategies including telemonitoring and teleconsultation to continue care of NCDs.64 The application of telemedicine in DM management showed beneficial results.56 65 Lifestyle focused messages might be an effective low-cost option to keep patients motivated to adhere to healthy lifestyles and further research seems advisable.66
All included studies were adapted to local contexts and the trialled strategies hold the promise of adaptability to healthcare systems in other African and LMIC. Moreover, the tasks of nurses in NCD care could be reshaped and expanded in countries with comparably few physicians in order to improve DM diagnostics, treatment and education.
Strategies to increase physical activity
As in the literature (GDM,67 DM168 69 and DM270 71), exercise therapy generally showed positive effects (DM2,28 72–75 DM1,76 GDM77) on glycaemic control. Exemplarily, 4 weeks by setting the goal to accumulate 10 000 steps per day significantly reduced HbA1C levels.72 Due to limited follow-up periods, it is advisable to target long-term adherence to these strategies in future research.
Pharmacological strategies
Currently, the available research on pharmacological interventions for DM is sparse in Africa. We identified only six studies (three on DM2,78–80 three on GDM81–83) testing pharmacological interventions as a central part of DM care14 despite known differences between African and European Americans.19 This might be attributable to our criteria excluding international studies with less than 50% of the sites in African countries.84–89 Many major multicentric pharmacological studies only have few study centres in Africa. Nevertheless, in-depth research into differing effectiveness of diabetic medications is still lacking. reported the usability and safety of a basal-bolus insulin regime with stepwise intensification in an African setting The efficacy of basal-bolus insulin regimes, as an easy to handle, practical DM treatment option was successfully tested by80 80 and has been previously described in other settings.90 91 Further research should consider regional contexts like availability of medication, practicability of the medication (eg, insulin needs proper storage92 93) lifestyle habits and genetic aspects.94 95 Consideration of findings on African American cohorts seems advisable.96 97
Strategies on nutrition and food supplementations
Nutritional and food supplementation interventions can successfully be used supporting pharmacological care or in early and pre-DM stages improving glycaemic control, lipid profiles and management of DM-related complications.98–110 In this review, nutritional interventions,41 111–113 including long-term consumption of honey,111 camel milk112 and a low fat and protein content of meals113 with positive effects on metabolic control. Camel milk, traditionally used for treatment of DM in arid areas of Africa and Asia, improves glycaemic control, reduces insulin requirement and limits diabetic complications.114 Rashad115 stated the beneficial effects of balanites aegyptiaca (desert date) extract on glycaemic control. This evergreen tree is common in arid regions in Africa and was traditionally used in Egyptian traditional medicine.116
Several food supplementations (zinc-gluconate117 and zinc-chromium118 supplementations, ginger powder,119 Nigella sativa oil capsules,120 L-carnitine,121 L-carnosine122 as well as vitamin B, C or D supplementation63 123–125) had positive effects on glycaemic control.
Strategies on the treatment of DM-related complications
Three studies tested the role of periodontitis treatment in diabetic patients.126–128 Tsobgny-Tsague et al128 and El-Makaky and Shalaby126 described the importance of early treatment start, resulting in favourable patient outcomes in periodontal health and glucose control. El-Sharkawy et al127 found propolis to be a favourable addition to planing and scaling. In an Ethiopian cohort, only 21% of patients with DM received oral health screening.129 The WHO regards oral health as a crucial component of healthcare with 12%–14% of 35–44 years Africans suffering from periodontitis.130
Treatment options for diabetic wounds were tested in two studies.131 132 Phototherapy in addition to usual care was first trialled in an African cohort of patients suffering from diabetic foot ulcers, showing beneficial wound healing outcomes. Similar results were described in other settings.133 The addition of propolis to usual care regimes showed improved wound healing. These findings are supported by studies from other settings.134 135
Strength and limitations
The external validity of this systematic review is limited by the focus on a limited number of countries and urban healthcare setting. The included studies were set in 15 of the 54 African countries with a focus on the North African region, especially Egypt. Egypt is the country with the highest known prevalence of DM in the African continent.4 7 This might be related to economic expansion and urbanisation, but also due to specific dietary issues (eg, white bread, polished rice, transfats), reduced physical activity due to prohibition of exercise in public places, shortage of exercise facilities, poor physical education in schools. Poor diet and physical inactivity are causing a high rate of overweight and obesity among the Egyptian population.136
Our preplanned primary outcome was mortality which was not reported in any of the included studies. Since DM is a chronic disease with a slow progression and long-term development of organ damage, the survival time is higher than the follow-up time of most of the studies. The included studies looked at long-term treatment strategies rather than treatment for acute hypoglycaemic or hyperglycaemic events that can lead to acutely fatal events. Nevertheless, long-term glucose control, being represented by the HbA1c value is one of the strongest clinical-outcome indicators of efficient DM management and health outcomes.137 It is easy to measure and serves as a representation of the individual’s average blood glucose levels in the previous 3 months.137 Furthermore, it is up to discussion if improvement of glycaemic control based on blood glucose measures like HbA1C are necessary the best strategic in LMIC or if diabetes complications are more effectively prevented by targeting blood pressure or blood lipids.138
Next, this review does not include non-randomised study types including prospective cohort trials or qualitative research, probable not taking into account the evidence that has been accumulated. Nevertheless, our aim was to search for randomised trials, since these study types, if conducted well, have a high evidence quality, allowing to minimise biases. Moreover, many of the studies included had a high risk of bias.
This systematic review includes studies as the highest level of evidence to investigate the benefits and harms of interventions.139 We included studies published in the English language without time restrictions. Language bias was shown to be unlikely. Despite the high linguistic diversity on the African continent, the languages mostly spoken are English, Arabic and French.140 Eventually, we did not exclude any study due to the publication language, but we might have missed studies from journals that are not listened in searched databases.
Conclusion
This systematic review shows an increasing number of studies due to the rising prevalence and awareness of DM in African countries. However, the number of high-quality studies is still low and emphasises knowledge gaps and underexplored research areas. Available studies are not representative of all African regions and were mainly conducted in urban areas of higher developed countries. Especially primary care settings and implementation research are underrepresented.
An improvement of the prognosis of patients with DM in Africa requires adequate technical and financial resources, training of healthcare staff and the implementation of comprehensive strategies to improve early diagnostics, adherence to medical treatment and subsequent regular checks. The identified studies offer a variety of effective approaches as a basis for local guidelines in the different fields of action in DM care adjusted to regional circumstances.
Supplementary Material
Acknowledgments
Dawn Bielawski did the proofreading.
Footnotes
Contributors: ASS-Y and ESK are joint first authors. ASS-Y developed the concept of the review, performed the initial systematic search in the International Trials Registry, screened the references, extracted study data in 2019, wrote a draft of the manuscript and worked in the coauthors comments on the final version of the manuscript and finally submitted the manuscript. ESK updated the systematic search, added a search in two regional databases, screened the updated search results and extracted the updated data and wrote the final version of the manuscript. SU has expertise in systematic reviews and is the guarantor of the methodological quality of the systematic review, developed the review concept has registered the protocol, performed the systematic search in two databases, screened all references, checked the initial as well as the updated data extraction and wrote the final version of this manuscript. WA provided expertise on the needs of evidence in the African context, developed the review concept, discussed the protocol and critically read and commented on the manuscript. TF and provided expertise on primary care, developed the review concept, critically read and commented on the manuscript. EJK provided expertise on the needs of evidence in the African context, developed the review concept, critically read and commented on the manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
No data are available. search strategy ProtocolList of included and excluded studies.
Ethics statements
Patient consent for publication
Not applicable.
References
- 1.Kushitor MK, Boatemaa S. The double burden of disease and the challenge of health access: evidence from access, bottlenecks, cost and equity facility survey in Ghana. PLoS One 2018;13:e0194677. 10.1371/journal.pone.0194677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Misganaw A, Mariam DH, Araya T. The double mortality burden among adults in Addis Ababa, Ethiopia, 2006-2009. Prev Chronic Dis 2012;9:E84. 10.5888/pcd9.110142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Roth GA, Abate D, Abate KH, et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: a systematic analysis for the global burden of disease study 2017. The Lancet 2018;392:1736–88. 10.1016/S0140-6736(18)32203-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Federation ID . IDF diabetes atlas. 9th edition. International Diabetes Federation, 2019. https://www.diabetesatlas.org/en/ [Google Scholar]
- 5.Beagley J, Guariguata L, Weil C, et al. Global estimates of undiagnosed diabetes in adults. Diabetes Res Clin Pract 2014;103:150–60. 10.1016/j.diabres.2013.11.001 [DOI] [PubMed] [Google Scholar]
- 6.Asmelash D, Asmelash Y. The burden of undiagnosed diabetes mellitus in adult African population: a systematic review and meta-analysis. J Diabetes Res 2019;2019:4134937. 10.1155/2019/4134937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bos M, Agyemang C. Prevalence and complications of diabetes mellitus in northern Africa, a systematic review. BMC Public Health 2013;13:387. 10.1186/1471-2458-13-387 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Awadalla H, Noor SK, Elmadhoun WM, et al. Diabetes complications in Sudanese individuals with type 2 diabetes: overlooked problems in sub-Saharan Africa? Diabetes Metab Syndr 2017;11 Suppl 2:S1047–51. 10.1016/j.dsx.2017.07.039 [DOI] [PubMed] [Google Scholar]
- 9.Mutyambizi C, Pavlova M, Chola L, et al. Cost of diabetes mellitus in Africa: a systematic review of existing literature. Global Health 2018;14:3. 10.1186/s12992-017-0318-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Clark A, Jit M, Warren-Gash C, et al. Global, regional, and national estimates of the population at increased risk of severe COVID-19 due to underlying health conditions in 2020: a modelling study. Lancet Glob Health 2020;8:e1003–17. 10.1016/S2214-109X(20)30264-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nations U . Sustainable development goals. goal 3: ensure healthy living and promote well-being for all at all ages, 2019. Available: https://www.un.org/sustainabledevelopment/health/ [Accessed 29 Sep 2020].
- 12.Gómez-Velasco DV, Almeda-Valdes P, Martagón AJ, et al. Empowerment of patients with type 2 diabetes: current perspectives. Diabetes Metab Syndr Obes 2019;12:1311. 10.2147/DMSO.S174910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Audain K, Levy L, Ellahi B. Sugar-Sweetened beverage consumption in the early years and implications for type-2 diabetes: a sub-Saharan Africa context. Proc Nutr Soc 2019;78:547–53. 10.1017/S0029665118002860 [DOI] [PubMed] [Google Scholar]
- 14.Cosentino F, Grant PJ, Aboyans V, et al. 2019 ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J 2020;41:255–323. 10.1093/eurheartj/ehz486 [DOI] [PubMed] [Google Scholar]
- 15.Organization WH . World Health organization model list of essential medicines: 21st list 2019. World Health Organization, 2019. [Google Scholar]
- 16.American Diabetes Association . 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2020. Diabetes Care 2020;43:S14. 10.2337/dc20-S002 [DOI] [PubMed] [Google Scholar]
- 17.Roglic G. Who global report on diabetes: a summary. Int J Noncommun Dis 2016;1:3. 10.4103/2468-8827.184853 [DOI] [Google Scholar]
- 18.Checkley W, Ghannem H, Irazola V, et al. Management of ncd in low- and middle-income countries. Glob Heart 2014;9:431–43. 10.1016/j.gheart.2014.11.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gomez F, Hirbo J, Tishkoff SA. Genetic variation and adaptation in Africa: implications for human evolution and disease. Cold Spring Harb Perspect Biol 2014;6:a008524. 10.1101/cshperspect.a008524 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Owolabi MO, Yaria JO, Daivadanam M, et al. Gaps in guidelines for the management of diabetes in low- and middle-income versus high-income Countries-A systematic review. Diabetes Care 2018;41:1097–105. 10.2337/dc17-1795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097. 10.1371/journal.pmed.1000097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Higgins JPT GS. Cochrane Handbook for systematic reviews of interventions version 5.1. 0. Collaboration TC, 2011. www.handbook.cochrane.org [Google Scholar]
- 23.Moher D, Hopewell S, Schulz KF, et al. Consort 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. Int J Surg 2012;10:28–55. 10.1016/j.ijsu.2011.10.001 [DOI] [PubMed] [Google Scholar]
- 24.Team TE . Endnote X9. X9 edn. Philadelphia PA: Clarivate, 2013. [Google Scholar]
- 25.Higgins JPT, Eldridge S, Te L. Chapter 23: Including variants on randomized trials. In: Higgins JPT, Thomas J, Chandler J, et al., eds. Cochrane Handbook for systematic reviews of interventions version 61. Cochrane, 2020. www.training.cochrane.org/handbook [Google Scholar]
- 26.The Nordic Cochrane Centre, The Cochrane Collaboration . (RevMan) RM. version 5.3. Copenhagen, 2014. [Google Scholar]
- 27.Fortwaengler K, Parkin CG, Neeser K, et al. Description of a new predictive modeling approach that correlates the risk and associated cost of well-defined diabetes-related complications with changes in glycated hemoglobin (HbA1c). J Diabetes Sci Technol 2017;11:315–23. 10.1177/1932296816662048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Asuako B, Moses MO, Eghan BA, et al. Fasting plasma glucose and lipid profiles of diabetic patients improve with aerobic exercise training. Ghana Med J 2017;51:120–7. 10.4314/gmj.v51i3.5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Theobald S, Brandes N, Gyapong M, et al. Implementation research: new imperatives and opportunities in global health. Lancet 2018;392:2214–28. 10.1016/S0140-6736(18)32205-0 [DOI] [PubMed] [Google Scholar]
- 30.LLC M . Sub-Saharan Africa rural population 1960-2022, 2022. Available: https://www.macrotrends.net/countries/SSF/sub-saharan-africa-/rural-population
- 31.Chiwanga FS, Njelekela MA, Diamond MB, et al. Urban and rural prevalence of diabetes and pre-diabetes and risk factors associated with diabetes in Tanzania and Uganda. Glob Health Action 2016;9:31440. 10.3402/gha.v9.31440 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Price AJ, Crampin AC, Amberbir A, et al. Prevalence of obesity, hypertension, and diabetes, and cascade of care in sub-Saharan Africa: a cross-sectional, population-based study in rural and urban Malawi. Lancet Diabetes Endocrinol 2018;6:208–22. 10.1016/S2213-8587(17)30432-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bank AD. The Africa infrastructure development index (AIDI) 2018, 2018. Available: https://www.icafrica.org/en/knowledge-hub/article/the-africa-infrastructure-development-index-aidi-2018-358/
- 34.Hall V, Thomsen RW, Henriksen O, et al. Diabetes in sub Saharan Africa 1999-2011: epidemiology and public health implications. A systematic review. BMC Public Health 2011;11:1–12. 10.1186/1471-2458-11-564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Federation ID . IDF diabetes atlas. Africa. Available: https://diabetesatlas.org/upload/resources/material/20191218_144539_afr_factsheet_en.pdf2019
- 36.Federation ID . IDF diabetes Artlas. Muddle East and North Africa. Available: https://www.diabetesatlas.org/upload/resources/material/20191218_144557_mena_factsheet_en.pdf2019
- 37.Utz B, Assarag B, Smekens T, et al. Detection and initial management of gestational diabetes through primary health care services in Morocco: an effectiveness-implementation trial. PLoS One 2018;13:e0209322. 10.1371/journal.pone.0209322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Webb EM, Rheeder P. A cluster-randomized trial to estimate the effect of mobile screening and treatment feedback on HbA1c and diabetes-related complications in Tshwane primary health care clinics, South Africa. Prim Care Diabetes 2017;11:546–54. 10.1016/j.pcd.2017.05.010 [DOI] [PubMed] [Google Scholar]
- 39.Webb EM, Rheeder P, Roux P. Screening in primary care for diabetic retinopathy, maculopathy and visual loss in South Africa. Ophthalmologica 2016;235:141–9. 10.1159/000443972 [DOI] [PubMed] [Google Scholar]
- 40.Webb EM, Rheeder P, Van Zyl DG. Diabetes care and complications in primary care in the Tshwane district of South Africa. Prim Care Diabetes 2015;9:147–54. 10.1016/j.pcd.2014.05.002 [DOI] [PubMed] [Google Scholar]
- 41.Krawinkel MB, Ludwig C, Swai ME, et al. Bitter gourd reduces elevated fasting plasma glucose levels in an intervention study among prediabetics in Tanzania. J Ethnopharmacol 2018;216:1–7. 10.1016/j.jep.2018.01.016 [DOI] [PubMed] [Google Scholar]
- 42.RezkAllah SS, Takla MK. Effects of different dosages of interval training on glycemic control in people with prediabetes: a randomized controlled trial. Diabetes Spectr 2019;32:125–31. 10.2337/ds18-0024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mogueo A, Oga-Omenka C, Hatem M, et al. Effectiveness of interventions based on patient empowerment in the control of type 2 diabetes in sub-Saharan Africa: a review of randomized controlled trials. Endocrinol Diabetes Metab 2021;4:e00174. 10.1002/edm2.174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Atun R, Davies JI, Gale EAM, et al. Diabetes in sub-Saharan Africa: from clinical care to health policy. Lancet Diabetes Endocrinol 2017;5:622–67. 10.1016/S2213-8587(17)30181-X [DOI] [PubMed] [Google Scholar]
- 45.Beck J, Greenwood DA, Blanton L, et al. 2017 national standards for diabetes self-management education and support. Diabetes Educ 2018;44:35–50. 10.1177/0145721718754797 [DOI] [PubMed] [Google Scholar]
- 46.Abaza H, Marschollek M, Schulze M. SMS Education for the Promotion of Diabetes Self-Management in Low & Middle Income Countries: A Randomized Controlled Trial in Egypt. Stud Health Technol Inform 2017;245:1209. 10.1186/s12889-017-4973-5 [DOI] [PubMed] [Google Scholar]
- 47.Adibe MO, Ukwe CV, Aguwa CN. The impact of pharmaceutical care intervention on the quality of life of Nigerian patients receiving treatment for type 2 diabetes. Value Health Reg Issues 2013;2:240–7. 10.1016/j.vhri.2013.06.007 [DOI] [PubMed] [Google Scholar]
- 48.Adjei DN, Agyemang C, Dasah JB, et al. The effect of electronic reminders on risk management among diabetic patients in low resourced settings. J Diabetes Complications 2015;29:818–21. 10.1016/j.jdiacomp.2015.05.008 [DOI] [PubMed] [Google Scholar]
- 49.Amendezo E, Walker Timothy D, Karamuka V, et al. Effects of a lifestyle education program on glycemic control among patients with diabetes at Kigali university Hospital, Rwanda: a randomized controlled trial. Diabetes Res Clin Pract 2017;126:129–37. 10.1016/j.diabres.2017.02.001 [DOI] [PubMed] [Google Scholar]
- 50.Chraibi A, Al-Herz S, Nguyen BD, et al. An RCT investigating Patient-Driven versus Physician-Driven titration of BIAsp 30 in patients with type 2 diabetes uncontrolled using NPH insulin. Diabetes Ther 2017;8:767–80. 10.1007/s13300-017-0268-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Debussche X, Besançon S, Balcou-Debussche M, et al. Structured peer-led diabetes self-management and support in a low-income country: the ST2EP randomised controlled trial in Mali. PLoS One 2018;13:e0191262. 10.1371/journal.pone.0191262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Essien O, Otu A, Umoh V, et al. Intensive patient education improves glycaemic control in diabetes compared to conventional education: a randomised controlled trial in a Nigerian tertiary care hospital. PLoS One 2017;12:e0168835. 10.1371/journal.pone.0168835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hailu FB, Hjortdahl P, Moen A. Nurse-Led diabetes self-management education improves clinical parameters in Ethiopia. Front Public Health 2018;6:302. 10.3389/fpubh.2018.00302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Labhardt ND, Balo J-R, Ndam M, et al. Improved retention rates with low-cost interventions in hypertension and diabetes management in a rural African environment of nurse-led care: a cluster-randomised trial. Trop Med Int Health 2011;16:1276–84. 10.1111/j.1365-3156.2011.02827.x [DOI] [PubMed] [Google Scholar]
- 55.Mash RJ, Rhode H, Zwarenstein M, et al. Effectiveness of a group diabetes education programme in under-served communities in South Africa: a pragmatic cluster randomized controlled trial. Diabet Med 2014;31:987–93. 10.1111/dme.12475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Takenga C, Berndt R-D, Musongya O, et al. An ICT-Based diabetes management system tested for health care delivery in the African context. Int J Telemed Appl 2014;2014:437307. 10.1155/2014/437307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tawfik MY, Mohamed RA. The impact of communicating cardiovascular risk in type 2 diabetics on patient risk perception, diabetes self-care, glycosylated hemoglobin, and cardiovascular risk. J Public Health 2016;24:153–64. 10.1007/s10389-016-0710-2 [DOI] [Google Scholar]
- 58.Thuita AW, Kiage BN, Onyango AN, et al. Effect of a nutrition education programme on the metabolic syndrome in type 2 diabetes mellitus patients at a level 5 Hospital in Kenya: "a randomized controlled trial". BMC Nutr 2020;6:30. 10.1186/s40795-020-00355-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Malipa M, Menon J. The relationship between compliance and quality of life among adolescents with diabetes mellitus type1. Medical Journal of Zambia 2013;40:93–103 https://www.mjz.co.zm/index.php/mjz/article/view/463 [Google Scholar]
- 60.Adibe MO, Aguwa CN, Ukwe CV. Cost-Utility analysis of pharmaceutical care intervention versus usual care in management of Nigerian patients with type 2 diabetes. Value Health Reg Issues 2013;2:189–98. 10.1016/j.vhri.2013.06.009 [DOI] [PubMed] [Google Scholar]
- 61.Fairall LR, Folb N, Timmerman V, et al. Educational outreach with an integrated clinical tool for nurse-led non-communicable chronic disease management in primary care in South Africa: a pragmatic cluster randomised controlled trial. PLoS Med 2016;13:e1002178. 10.1371/journal.pmed.1002178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Steyn K, Lombard C, Gwebushe N, et al. Implementation of national guidelines, incorporated within structured diabetes and hypertension records at primary level care in Cape town, South Africa: a randomised controlled trial. Glob Health Action 2013;6:20796. 10.3402/gha.v6i0.20796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ragheb SR, El Wakeel LM, Nasr MS, et al. Impact of rutin and vitamin C combination on oxidative stress and glycemic control in patients with type 2 diabetes. Clin Nutr ESPEN 2020;35:128–35. 10.1016/j.clnesp.2019.10.015 [DOI] [PubMed] [Google Scholar]
- 64.Advisor E. Telemedicine for Diabetes Management During the COVID-19 Pandemic and Beyond Telemedicine for Diabetes Management During the COVID-19 Pandemic and Beyond - Endocrinology Advisor 2020.
- 65.Tchero H, Kangambega P, Briatte C, et al. Clinical effectiveness of telemedicine in diabetes mellitus: a meta-analysis of 42 randomized controlled trials. Telemed J E Health 2019;25:569–83. 10.1089/tmj.2018.0128 [DOI] [PubMed] [Google Scholar]
- 66.Haider R, Sudini L, Chow CK, et al. Mobile phone text messaging in improving glycaemic control for patients with type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetes Res Clin Pract 2019;150:27–37. 10.1016/j.diabres.2019.02.022 [DOI] [PubMed] [Google Scholar]
- 67.Russo LM, Nobles C, Ertel KA, et al. Physical activity interventions in pregnancy and risk of gestational diabetes mellitus: a systematic review and meta-analysis. Obstet Gynecol 2015;125:576–82. 10.1097/AOG.0000000000000691 [DOI] [PubMed] [Google Scholar]
- 68.Quirk H, Blake H, Tennyson R, et al. Physical activity interventions in children and young people with type 1 diabetes mellitus: a systematic review with meta-analysis. Diabet Med 2014;31:1163–73. 10.1111/dme.12531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Aljawarneh YM, Wardell DW, Wood GL, et al. A systematic review of physical activity and exercise on physiological and biochemical outcomes in children and adolescents with type 1 diabetes. J Nurs Scholarsh 2019;51:337–45. 10.1111/jnu.12472 [DOI] [PubMed] [Google Scholar]
- 70.Pan B, Ge L, Xun Y-Q, et al. Exercise training modalities in patients with type 2 diabetes mellitus: a systematic review and network meta-analysis. Int J Behav Nutr Phys Act 2018;15:72. 10.1186/s12966-018-0703-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Smith AD, Crippa A, Woodcock J. Physical activity and incident type 2 diabetes mellitus: a systematic review and dose–response meta-analysis of prospective cohort studies. Springer, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Fayehun AF, Olowookere OO, Ogunbode AM, et al. Walking prescription of 10 000 steps per day in patients with type 2 diabetes mellitus: a randomised trial in Nigerian general practice. British Journal of General Practice 2018;68:e139–45. 10.3399/bjgp18X694613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Maharaj SS, Nuhu JM. Rebound exercise: a beneficial adjuvant for sedentary non-insulin-dependent type 2 diabetic individuals in a rural environment. Aust J Rural Health 2016;24:123–9. 10.1111/ajr.12223 [DOI] [PubMed] [Google Scholar]
- 74.van Rooijen AJ, Rheeder P, Eales CJ, et al. Effect of exercise versus relaxation on haemoglobin A1c in black females with type 2 diabetes mellitus. QJM 2004;97:343–51. 10.1093/qjmed/hch061 [DOI] [PubMed] [Google Scholar]
- 75.Yan H, Prista A, Ranadive SM, et al. Effect of aerobic training on glucose control and blood pressure in T2DDM East African males. ISRN Endocrinol 2014;2014:864897. 10.1155/2014/864897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Salem MA, AboElAsrar MA, Elbarbary NS, et al. Is exercise a therapeutic tool for improvement of cardiovascular risk factors in adolescents with type 1 diabetes mellitus? a randomised controlled trial. Diabetol Metab Syndr 2010;2:47. 10.1186/1758-5996-2-47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Embaby H, Elsayed E, Fawzy M. Insulin sensitivity and plasma glucose response to aerobic exercise in pregnant women at risk for gestational diabetes mellitus. Ethiop J Health Sci 2016;26:409–14. ‐. 10.4314/ejhs.v26i5.2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Distiller LA, Nortje H, Wellmann H, et al. A 24-week, prospective, randomized, open-label, treat-to-target pilot study of obese type 2 diabetes patients with severe insulin resistance to assess the addition of exenatide on the efficacy of U-500 regular insulin plus metformin. Endocr Pract 2014;20:1143–50. 10.4158/EP14067.OR [DOI] [PubMed] [Google Scholar]
- 79.El-Haggar SM, Farrag WF, Kotkata FA. Effect of ketotifen in obese patients with type 2 diabetes mellitus. J Diabetes Complications 2015;29:427–32. 10.1016/j.jdiacomp.2015.01.013 [DOI] [PubMed] [Google Scholar]
- 80.Malek R, Ajili F, Assaad-Khalil SH, et al. Similar glucose control with basal-bolus regimen of insulin detemir plus insulin aspart and thrice-daily biphasic insulin aspart 30 in insulin-naive patients with type 2 diabetes: results of a 50-week randomized clinical trial of stepwise insulin intensification. Diabetes Metab 2015;41:223–30. 10.1016/j.diabet.2014.11.002 [DOI] [PubMed] [Google Scholar]
- 81.Ashoush S, El-Said M, Fathi H, et al. Identification of metformin poor responders, requiring supplemental insulin, during randomization of metformin versus insulin for the control of gestational diabetes mellitus. J Obstet Gynaecol Res 2016;42:640–7. 10.1111/jog.12950 [DOI] [PubMed] [Google Scholar]
- 82.Beyuo T, Obed SA, Adjepong-Yamoah KK, et al. Metformin versus insulin in the management of Pre-Gestational diabetes mellitus in pregnancy and gestational diabetes mellitus at the Korle BU teaching hospital: a randomized clinical trial. PLoS One 2015;10:e0125712. 10.1371/journal.pone.0125712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ibrahim MI, Hamdy A, Shafik A, et al. The role of adding metformin in insulin-resistant diabetic pregnant women: a randomized controlled trial. Arch Gynecol Obstet 2014;289:959–65. 10.1007/s00404-013-3090-7 [DOI] [PubMed] [Google Scholar]
- 84.Bailey CJ, Iqbal N, T'joen C, et al. Dapagliflozin monotherapy in drug-naïve patients with diabetes: a randomized-controlled trial of low-dose range. Diabetes Obes Metab 2012;14:951–9. 10.1111/j.1463-1326.2012.01659.x [DOI] [PubMed] [Google Scholar]
- 85.Chou HS, Truitt KE, Moberly JB, et al. A 26-week, placebo- and pioglitazone-controlled monotherapy study of rivoglitazone in subjects with type 2 diabetes mellitus. Diabetes Obes Metab 2012;14:1000–9. 10.1111/j.1463-1326.2012.01631.x [DOI] [PubMed] [Google Scholar]
- 86.De Caterina R, Andersson U, Alexander JH, et al. History of bleeding and outcomes with apixaban versus warfarin in patients with atrial fibrillation in the apixaban for reduction in stroke and other thromboembolic events in atrial fibrillation trial. Am Heart J 2016;175:175–83. 10.1016/j.ahj.2016.01.005 [DOI] [PubMed] [Google Scholar]
- 87.Kadiri A, Al-Nakhi A, El-Ghazali S, et al. Treatment of type 1 diabetes with insulin lispro during Ramadan. Diabetes Metab 2001;27:482–6. [PubMed] [Google Scholar]
- 88.Schumm-Draeger P-M, Burgess L, Korányi L, et al. Twice-Daily dapagliflozin co-administered with metformin in type 2 diabetes: a 16-week randomized, placebo-controlled clinical trial. Diabetes Obes Metab 2015;17:42–51. 10.1111/dom.12387 [DOI] [PubMed] [Google Scholar]
- 89.Van Olmen J, Van Pelt M, Malombo B, et al. Process evaluation of a mobile health intervention for people with diabetes in low income countries - the implementation of the TEXT4DSM study. J Telemed Telecare 2017;23:96–105. 10.1177/1357633X15617885 [DOI] [PubMed] [Google Scholar]
- 90.Wang C, Mamza J, Idris I. Biphasic vs basal bolus insulin regimen in Type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Diabet Med 2015;32:585–94. 10.1111/dme.12694 [DOI] [PubMed] [Google Scholar]
- 91.Meece J. Basal insulin intensification in patients with type 2 diabetes: a review. Diabetes Ther 2018;9:877–90. 10.1007/s13300-018-0395-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Heinemann L, Braune K, Carter A, et al. Insulin storage: a critical reappraisal. J Diabetes Sci Technol 2021;15:1932296819900258. 10.1177/1932296819900258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Bahendeka S, Kaushik R, Swai AB, et al. EADSG guidelines: insulin storage and optimisation of injection technique in diabetes management. Diabetes Ther 2019;10:341–66. 10.1007/s13300-019-0574-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Asamoah EA, Obirikorang C, Acheampong E, et al. Heritability and genetics of type 2 diabetes mellitus in sub-Saharan Africa: a systematic review and meta-analysis. J Diabetes Res 2020;2020:3198671. 10.1155/2020/3198671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Onengut-Gumuscu S, Chen W-M, Robertson CC, et al. Type 1 diabetes risk in African-Ancestry participants and utility of an ancestry-specific genetic risk score. Diabetes Care 2019;42:406–15. 10.2337/dc18-1727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Cheng C-Y, Reich D, Haiman CA, et al. African ancestry and its correlation to type 2 diabetes in African Americans: a genetic admixture analysis in three U.S. population cohorts. PLoS One 2012;7:e32840. 10.1371/journal.pone.0032840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ng MCY, Shriner D, Chen BH, et al. Meta-Analysis of genome-wide association studies in African Americans provides insights into the genetic architecture of type 2 diabetes. PLoS Genet 2014;10:e1004517. 10.1371/journal.pgen.1004517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Jayawardena R, Ranasinghe P, Galappatthy P, et al. Effects of zinc supplementation on diabetes mellitus: a systematic review and meta-analysis. Diabetol Metab Syndr 2012;4:13. 10.1186/1758-5996-4-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wang X, Wu W, Zheng W, et al. Zinc supplementation improves glycemic control for diabetes prevention and management: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr 2019;110:76–90. 10.1093/ajcn/nqz041 [DOI] [PubMed] [Google Scholar]
- 100.Daily JW, Yang M, Kim DS, et al. Efficacy of ginger for treating type 2 diabetes: a systematic review and meta-analysis of randomized clinical trials. Journal of Ethnic Foods 2015;2:36–43. 10.1016/j.jef.2015.02.007 [DOI] [Google Scholar]
- 101.Zhu J, Chen H, Song Z, et al. Effects of Ginger (Zingiber officinale Roscoe) on Type 2 Diabetes Mellitus and Components of the Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Evid Based Complement Alternat Med 2018;2018:1–11. 10.1155/2018/5692962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Daryabeygi-Khotbehsara R, Golzarand M, Ghaffari MP, et al. Nigella sativa improves glucose homeostasis and serum lipids in type 2 diabetes: a systematic review and meta-analysis. Complement Ther Med 2017;35:6–13. 10.1016/j.ctim.2017.08.016 [DOI] [PubMed] [Google Scholar]
- 103.Heshmati J, Namazi N. Effects of black seed (Nigella sativa) on metabolic parameters in diabetes mellitus: a systematic review. Complement Ther Med 2015;23:275–82. 10.1016/j.ctim.2015.01.013 [DOI] [PubMed] [Google Scholar]
- 104.Vidal-Casariego A, Burgos-Peláez R, Martínez-Faedo C, et al. Metabolic effects of L-carnitine on type 2 diabetes mellitus: systematic review and meta-analysis. Exp Clin Endocrinol Diabetes 2013;121:234–8. 10.1055/s-0033-1333688 [DOI] [PubMed] [Google Scholar]
- 105.Xu Y, Jiang W, Chen G, et al. L-Carnitine treatment of insulin resistance: a systematic review and meta-analysis. Adv Clin Exp Med 2017;26:333–8. 10.17219/acem/61609 [DOI] [PubMed] [Google Scholar]
- 106.Das UN. Vitamin C for type 2 diabetes mellitus and hypertension. Arch Med Res 2019;50:11–14. 10.1016/j.arcmed.2019.05.004 [DOI] [PubMed] [Google Scholar]
- 107.Afkhami-Ardekani M, Shojaoddiny-Ardekani A. Effect of vitamin C on blood glucose, serum lipids & serum insulin in type 2 diabetes patients. Indian Journal of medical research 2007;126:471 https://pubmed.ncbi.nlm.nih.gov/18160753/ [PubMed] [Google Scholar]
- 108.Maritim AC, Sanders RA, Watkins JB. Diabetes, oxidative stress, and antioxidants: a review. J Biochem Mol Toxicol 2003;17:24–38. 10.1002/jbt.10058 [DOI] [PubMed] [Google Scholar]
- 109.Zhou C, Na L, Shan R, et al. Dietary vitamin C intake reduces the risk of type 2 diabetes in Chinese adults: HOMA-IR and T-AOC as potential mediators. PLoS One 2016;11:e0163571. 10.1371/journal.pone.0163571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Hosseinzadeh H, Nassiri-Asl M. Review of the protective effects of rutin on the metabolic function as an important dietary flavonoid. J Endocrinol Invest 2014;37:783–8. 10.1007/s40618-014-0096-3 [DOI] [PubMed] [Google Scholar]
- 111.Abdulrhman MM, El-Hefnawy MH, Aly RH, et al. Metabolic effects of honey in type 1 diabetes mellitus: a randomized crossover pilot study. J Med Food 2013;16:66–72. 10.1089/jmf.2012.0108 [DOI] [PubMed] [Google Scholar]
- 112.Mohamad RH, Zekry ZK, Al-Mehdar HA, et al. Camel milk as an adjuvant therapy for the treatment of type 1 diabetes: verification of a traditional ethnomedical practice. J Med Food 2009;12:461–5. 10.1089/jmf.2008.0009 [DOI] [PubMed] [Google Scholar]
- 113.van der Hoogt M, van Dyk JC, Dolman RC, et al. Protein and fat meal content increase insulin requirement in children with type 1 diabetes - Role of duration of diabetes. J Clin Transl Endocrinol 2017;10:15–21. 10.1016/j.jcte.2017.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Shori AB. Camel milk as a potential therapy for controlling diabetes and its complications: A review of in vivo studies. J Food Drug Anal 2015;23:609–18. 10.1016/j.jfda.2015.02.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Rashad H, Metwally FM, Ezzat SM, et al. Randomized double-blinded pilot clinical study of the antidiabetic activity of Balanites aegyptiaca and UPLC-ESI-MS/MS identification of its metabolites. Pharm Biol 2017;55:1954–61. 10.1080/13880209.2017.1354388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Helal EGE, Abd El-Wahab SM, El Refaey H. Antidiabetic and Antihyperlipidemic Effect of Balanites Aegyptiaca Seeds (Aqueous Extract) on Diabetic Rats. Egypt J Hosp Med 2013;52:725–39. 10.12816/0000610 [DOI] [Google Scholar]
- 117.Matter RM, Elbarbary NS, Ismail EAR, et al. Zinc supplementation improves glucose homeostasis in patients with β-thalassemia major complicated with diabetes mellitus: a randomized controlled trial. Nutrition 2020;73:110702. 10.1016/j.nut.2019.110702 [DOI] [PubMed] [Google Scholar]
- 118.Anderson RA, Roussel AM, Zouari N, et al. Potential antioxidant effects of zinc and chromium supplementation in people with type 2 diabetes mellitus. J Am Coll Nutr 2001;20:212–8. 10.1080/07315724.2001.10719034 [DOI] [PubMed] [Google Scholar]
- 119.El Gayar MH, Aboromia MMM, Ibrahim NA, et al. Effects of ginger powder supplementation on glycemic status and lipid profile in newly diagnosed obese patients with type 2 diabetes mellitus. Obes Med 2019;14:100094. 10.1016/j.obmed.2019.100094 [DOI] [Google Scholar]
- 120.Moustafa HAM, El Wakeel LM, Halawa MR, et al. Effect of Nigella sativa oil versus metformin on glycemic control and biochemical parameters of newly diagnosed type 2 diabetes mellitus patients. Endocrine 2019;65:286–94. 10.1007/s12020-019-01963-4 [DOI] [PubMed] [Google Scholar]
- 121.El-Sheikh HM, El-Haggar SM, Elbedewy TA. Comparative study to evaluate the effect of L-carnitine plus glimepiride versus glimepiride alone on insulin resistance in type 2 diabetic patients. Diabetes Metab Syndr 2019;13:167–73. 10.1016/j.dsx.2018.08.035 [DOI] [PubMed] [Google Scholar]
- 122.Elbarbary NS, Ismail EAR, El-Naggar AR, et al. The effect of 12 weeks carnosine supplementation on renal functional integrity and oxidative stress in pediatric patients with diabetic nephropathy: a randomized placebo-controlled trial. Pediatr Diabetes 2018;19:470–7. 10.1111/pedi.12564 [DOI] [PubMed] [Google Scholar]
- 123.Ali S, Ghanem Y, et al. The impact of different regimens of vitamin D3 on glucose homeostasis in type 2 diabetic patients. Asian journal of pharmaceutical and clinical research 2019;12:21–6 https://innovareacademics.in/journals/index.php/ajpcr/article/view/35602 10.22159/ajpcr.2019.v12i12.35602 [DOI] [Google Scholar]
- 124.Elbarbary NS, Ismail EAR, Zaki MA, et al. Vitamin B complex supplementation as a homocysteine-lowering therapy for early stage diabetic nephropathy in pediatric patients with type 1 diabetes: a randomized controlled trial. Clin Nutr 2020;39:49–56. 10.1016/j.clnu.2019.01.006 [DOI] [PubMed] [Google Scholar]
- 125.Anyanwu AC, Fasanmade OA, Odeniyi IA, et al. Effect of vitamin D supplementation on glycemic control in type 2 diabetes subjects in Lagos, Nigeria. Indian J Endocrinol Metab 2016;20:189–94. 10.4103/2230-8210.176345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.El-Makaky Y, Shalaby HK. The effects of non-surgical periodontal therapy on glycemic control in diabetic patients: a randomized controlled trial. Oral Dis 2020;26:822–9. 10.1111/odi.13256 [DOI] [PubMed] [Google Scholar]
- 127.El-Sharkawy HM, Anees MM, Van Dyke TE. Propolis improves periodontal status and glycemic control in patients with type 2 diabetes mellitus and chronic periodontitis: a randomized clinical trial. J Periodontol 2016;87:1418–26. 10.1902/jop.2016.150694 [DOI] [PubMed] [Google Scholar]
- 128.Tsobgny-Tsague N-F, Lontchi-Yimagou E, Nana ARN, et al. Effects of nonsurgical periodontal treatment on glycated haemoglobin on type 2 diabetes patients (PARODIA 1 study): a randomized controlled trial in a sub-Saharan Africa population. BMC Oral Health 2018;18:28. 10.1186/s12903-018-0479-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Sahile AT, Mgutshini T, Ayehu SM. Oral health screening status of diabetes patients in selected hospitals of Addis Ababa, Ethiopia, 2018. Patient Relat Outcome Meas 2020;11:173–80. 10.2147/PROM.S269292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Organization WH . Promoting oral health in Africa: prevention and control of oral diseases and noma as part of essential noncommunicable disease interventions, 2016. [Google Scholar]
- 131.Nteleki B, Abrahamse H, Houreld NN. Conventional podiatric intervention and phototherapy in the treatment of diabetic ulcers. Semin Vasc Surg 2015;28:172–83. 10.1053/j.semvascsurg.2016.02.001 [DOI] [PubMed] [Google Scholar]
- 132.Yakoot M, Abdelatif M, Helmy S. Efficacy of a new local limb salvage treatment for limb-threatening diabetic foot wounds - a randomized controlled study. Diabetes Metab Syndr Obes 2019;12:1659–65. 10.2147/DMSO.S210680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Wang H-T, Yuan J-Q, Zhang B, et al. Phototherapy for treating foot ulcers in people with diabetes. Cochrane Database Syst Rev 2017;6:CD011979. 10.1002/14651858.CD011979.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Henshaw FR, Bolton T, Nube V, et al. Topical application of the bee hive protectant propolis is well tolerated and improves human diabetic foot ulcer healing in a prospective feasibility study. J Diabetes Complications 2014;28:850–7. 10.1016/j.jdiacomp.2014.07.012 [DOI] [PubMed] [Google Scholar]
- 135.Afkhamizadeh M, Aboutorabi R, Ravari H, et al. Topical propolis improves wound healing in patients with diabetic foot ulcer: a randomized controlled trial. Nat Prod Res 2018;32:2096–9. 10.1080/14786419.2017.1363755 [DOI] [PubMed] [Google Scholar]
- 136.Hegazi R, El-Gamal M, Abdel-Hady N, et al. Epidemiology of and risk factors for type 2 diabetes in Egypt. Ann Glob Health 2015;81:814–20. 10.1016/j.aogh.2015.12.011 [DOI] [PubMed] [Google Scholar]
- 137.Sherwani SI, Khan HA, Ekhzaimy A, et al. Significance of HbA1c test in diagnosis and prognosis of diabetic patients. Biomark Insights 2016;11:BMI. S38440. 10.4137/BMI.S38440 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Basu S, Flood D, Geldsetzer P, et al. Estimated effect of increased diagnosis, treatment, and control of diabetes and its associated cardiovascular risk factors among low-income and middle-income countries: a microsimulation model. Lancet Glob Health 2021;9:e1539–52. 10.1016/S2214-109X(21)00340-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Medicine OCfE-B . The Oxford 2011 levels of evidence, 2011. Available: https://www.cebm.net/wp-content/uploads/2014/06/CEBM-Levels-of-Evidence-2.1.pdf;
- 140.Maho JF. How many languages are there in Africa, really? Trends In Linguistics Studies And Monographs 2004;156:279–96. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2021-050021supp006.pdf (251.3KB, pdf)
bmjopen-2021-050021supp001.pdf (191.3KB, pdf)
bmjopen-2021-050021supp002.pdf (229.7KB, pdf)
bmjopen-2021-050021supp003.pdf (474.5KB, pdf)
bmjopen-2021-050021supp004.pdf (249.7KB, pdf)
bmjopen-2021-050021supp005.pdf (189.1KB, pdf)
Data Availability Statement
No data are available. search strategy ProtocolList of included and excluded studies.