Dear Editor,
Right ventricular failure (RVF) denotes the inability of the RV to meet the required blood flow without excessive use of the Frank–Starling mechanism [1]. RVF is prognostic in acute respiratory distress syndrome (ARDS) [2], but robust prospective study conducted in patients admitted in intensive care unit (ICU) affected by ARDS due to coronavirus disease 2019 (COVID-19) is lacking [3]. We sought to evaluate the association of RVF depicted by repeated echocardiography and mortality in ICU patients with COVID-19 ARDS, and its potential association with respiratory deterioration.
Between March 2020 and June 2021, all consecutive adult patients without chronic respiratory failure admitted to our ICU for COVID-19 moderate-to-severe ARDS were enrolled in this prospective, observational study (Supplementary materials). Initially, echocardiography was performed within 72 h following ICU admission (baseline), and secondarily on a weekly basis or if there was clinical deterioration. Since there is currently no consensual definition of RVF, we used that associating RV dilatation (RV/left venticular (LV) end-diastolic area > 0.6; severe if ≥ 1.0) and elevated central venous pressure (CVP ≥ 8 mmHg) reflecting systemic venous congestion [4]. Acute cor pulmonale (ACP) was conventionally defined [5].
Univariate Cox model analysis was used to determine variables to be included in the multivariate model (p < 0.10). RVF was considered as a time-dependent variable to identify parameters associated with 90 days mortality. In patients who developed RVF after baseline echocardiography, respiratory parameters and Sequential Organ Failure Assessment (SOFA) score were compared with those obtained at preceding assessment.
140 patients were studied (median age: 68[60–72] years; 93 men (66%); Simplified Acute Physiology Score (SAPS) II: 37[31–48]; SOFA: 4[4–6]; PaO2/FiO2: 95[75–143]) (Fig. E1). During ICU stay (15[10–25] days), 110 patients (78%) received invasive ventilation after a median delay of 1[0–2] day and 57 patients (41%) required vasopressors (Table E1). Overall, 401 echocardiography were performed: 105 at baseline (76% of patients; Table E2) and 296 during ICU stay (Fig.E2A). RVF was diagnosed in 75 patients (54%), at baseline (n = 35) or between Day 3 and Day 23 (n = 40) (Fig.E2B). The proportion of ACP and severe ACP remained stable over time (Fig. E2C). Forty-nine of RVF patients (64%) required vasopressors (Fig. E3A), compared to 8 patients without RVF. Reproducibility of RVF diagnosis was excellent (Kappa = 0.91).
ICU and 90 days mortality reached 31%, mainly secondary to shock (67%), and predominantly occurred > 7 days after RVF diagnosis (Fig.E3B). Thirty-eight patients who developed RVF died (50%), while only 5 (8%) of their counterparts. Multivariate analysis identified the presence of RVF at baseline or the development of new-onset RVF during ICU stay (adjusted hazard ratio [HR]:8.17; 95% confidence intervals [CI]:3.15–21.2; p < 0.001), ischemic cardiomyopathy (HR:2.26 [1.19–4.31]: p = 0.013), and age (HR/10 years:1.84[1.20–2.81]:p = 0.005) as factors independently associated with 90-day mortality. Sensitivity analysis provided similar results, including for RVF (HR:6.60[2.24–19.47]:p < 0.001) (Table E3).
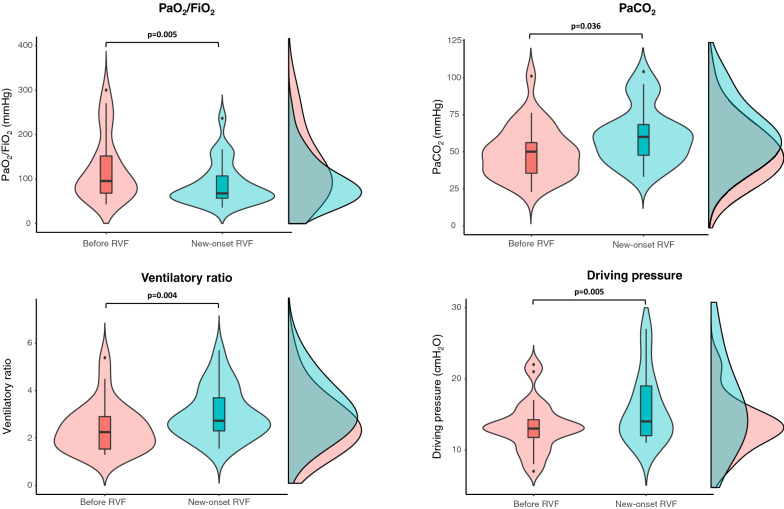
In patients with normal baseline echocardiography who subsequently developed RVF (n = 40), median PaO2/FiO2, PaCO2, ventilatory ratio and driving pressure significantly worsened between RVF diagnosis and preceding echocardiography assessment (Fig. 1), whereas SOFA score remained similar compared to ICU admission (5[3–14]vs.5[3–12]:p = 0.207).
Fig. 1.
Variation of respiratory parameters between the day of diagnosis of new-onset RVF and the previous echocardiography assessment. Violin plots, box plots and densities for each measured parameter are displayed. Within the box plots, the bold black line indicates the sample median, the hinges indicate 25th and 75th quantiles, and the whiskers point to 1.5 interquartile ranges beyond the hinges. Paired tests with Wilcoxon signed-rank test were used. Median PaO2/FiO2 (70 [57–126] vs. 96 [69–159] mmHg: p = 0.005), PaCO2 (60 [46–65] vs. 51 [35–61] mmHg: p = 0.036), ventilatory ratio (2.73 [2.29–3.57] vs. 2.25 [1.51–2.92]: p = 0.004) and driving pressure (14 [13–19] vs. 13 [10–14] cmH2O: p = 0.005) significantly worsened between RVF diagnosis and preceding echocardiography assessment. Abbreviations: RVF right ventricle failure
Development of RVF during ICU stay was a strong independent predictor of 90-day mortality. Significant worsening of PaO2/FiO2, PaCO2 and ventilatory ratio (increased pulmonary vasoconstriction) and augmented driving pressure could have contributed to RVF. This underlines the relevance of performing echocardiography to seek for RVF in COVID-19 ARDS patients, even when hemodynamically stable, especially in the presence of respiratory deterioration.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
We gratefully thank Charlotte Briat for her invaluable help in data collection.
Funding
None.
Declarations
Conflicts of interest
All the authors declared no conflicts of interest.
Ethical approval
The protocol was approved by local Ethics Committee (#492-2021-14).
Consent for participation
Non-opposition of the patients was obtained to participate in the study.
Consent for publication
NA.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Lahm T, Douglas IS, Archer SL, Bogaard HJ, Chesler NC, Haddad F, Hemnes AR, Kawut SM, Kline JA, Kolb TM, Mathai SC, Mercier O, Michelakis ED, Naeije R, Tuder RM, Ventetuolo CE, Vieillard-Baron A, Voelkel NF, Vonk-Noordegraaf A, Hassoun PM. Assessment of right ventricular function in the research setting: knowledge gaps and pathways forward. An official American Thoracic Society Research Statement. Am J Respir Crit Care Med. 2018;198:e15–e43. doi: 10.1164/rccm.201806-1160ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dong D, Zong Y, Li Z, Wang Y, Jing C. Mortality of right ventricular dysfunction in patients with acute respiratory distress syndrome subjected to lung protective ventilation: a systematic review and meta-analysis. Heart Lung J Crit Care. 2021;50:730–735. doi: 10.1016/j.hrtlng.2021.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Michard F, Vieillard-Baron A. Critically ill patients with COVID-19: are they hemodynamically unstable and do we know why? Intensive Care Med. 2021;47:254–255. doi: 10.1007/s00134-020-06238-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vieillard-Baron A, Prigent A, Repessé X, Goudelin M, Prat G, Evrard B, Charron C, Vignon P, Geri G. Right ventricular failure in septic shock: characterization, incidence and impact on fluid responsiveness. Crit Care. 2020;24:630. doi: 10.1186/s13054-020-03345-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jardin F, Dubourg O, Bourdarias JP. Echocardiographic pattern of acute cor pulmonale. Chest. 1997;111:209–217. doi: 10.1378/chest.111.1.209. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.