Abstract
Diabetic cardiomyopathy (DCM) is the main factor responsible for poor prognosis and survival in patients with diabetes. The highly complex pathogenesis of DCM involves multiple signaling pathways, including nuclear factor-κB (NF-κB) signaling pathway, adenosine monophosphate-activated protein kinase (AMPK) signaling pathway, phosphatidylinositol 3-kinase-protein kinase B (Akt) signaling pathway, mitogen-activated protein kinase (MAPK) signaling pathway, and transforming growth factor-β (TGF-β) signaling pathway. Nuclear factor erythroid-2-related factor 2 (Nrf2) seems essential to the amelioration of the progression of DCM, not only through counterbalancing oxidative stress, but also through interacting with other signaling pathways to combat inflammation, the disorder in energy homeostasis and insulin signaling, and fibrosis. It has been evidenced that Chinese herbal monomers could attenuate DCM through the crosstalk of Nrf2 with other signaling pathways. This article has summarized the pathogenesis of DCM (especially in oxidative stress), the beneficial effects of ameliorating DCM via the Nrf2 signaling pathway and its crosstalk, and examples of Chinese herbal monomers. It will facilitate pharmacological research and development to promote the utilization of traditional Chinese medicine in DCM.
1. Introduction
Diabetic cardiomyopathy (DCM) is a cardiac muscle-specific microvascular complication, which progresses in individuals with diabetes mellitus (DM) but without other cardiac risk factors including coronary artery disease, hypertension, and significant valvular disease [1]. The Framingham Heart Study showed that over the past 50 years, the proportion of diabetes-caused cardiovascular diseases has increased, which emphasizes the need to pay more attention to the cardiac condition in patients with DM [2]. DCM, which elevates mortality in type 1 and type 2 diabetes mellitus (T1DM and T2DM) patients, leads to a poor prognosis, and individuals with DM were 2.3 times more likely to develop heart failure than those without [3, 4]. Around 22% of patients with T2DM develop heart failure [5]. Furthermore, recent studies showed that even in mildly elevated blood glucose (prediabetes), the risk of heart failure was increased and associated with a poor prognosis [6, 7]. To date, there is no special effective medicine for DCM [8]. However, a plethora of scientific evidence revealed that Chinese herbal monomers might be potential drugs for the treatment of DCM.
Nuclear factor erythroid-2-related factor 2 (Nrf2) is a potent antioxidant gene, which can regulate cell signaling, transcription, anabolic metabolism, and extracellular matrix (ECM) remodeling through jointly acting on multiple proteins [9]. Nrf2 activates comprehensive cellular defense processes by affecting nearly 500 genes, thus augmenting the whole ability of cells to perform redox balancing factors, detoxifying enzymes, stress response proteins, and metabolic enzymes [10, 11]. Increasing investigations have suggested that Nrf2 could ameliorate DCM via crosstalk with different signaling pathways, and some Chinese herbal monomers have proved to have the capability of prompting that mechanism. This review provides a contemporary view of the pathogenesis of DCM (especially in oxidative stress), the beneficial effects of ameliorating DCM via the Nrf2 signaling pathway and its crosstalk, and the examples of Chinese herbal monomers.
2. Review
2.1. Oxidative Stress in the Pathogenesis of DCM
The progression from DM to DCM is extremely complicated. The heart is one of the important target organs of diabetes. Under physiological conditions, insulin stimulates the uptake of glucose into the cardiac muscle to maintain glucose homeostasis; however, insulin insistence (IR) and hyperinsulinemia are associated with the metabolic disorder in cardiovascular diseases [12]. In patients with DM, the main abnormalities of the inner environment are hyperglycemia, systemic IR, and hyperinsulinemia [1]. In those conditions, these abnormalities instigate disorders of systemic metabolism, activation of the sympathetic nervous system and renin-angiotensin-aldosterone system, response for maladaptive immune, inflammation, and accretion of advanced glycation end products (AGEs) that further prompt oxidative stress and lipid accumulation [13, 14]. Oxidative stress could trigger mitochondrial dysfunction, and endoplasmic reticulum stress (ERS), impair calcium handling, and increase Ca2+ sensitivity and Ca2+ influx [13]. The imbalance between mitophagy and mitochondrial biogenesis leads to damage to cardiomyocytes and fewer supply of energy to the myocardia [13, 15, 16]. Oxidative stress and ERS could induce abnormalities of calcium handling, which lead to diastolic dysfunction [13]. Besides, the interaction of reactive oxygen species (ROS), dysfunction mitochondrial, ERS, and abnormal calcium handling ultimately causes apoptosis [13]. Apoptosis is considered a major mechanism in maintaining cellular homeostasis in general, and it plays a crucial role in normal tissue turnover, immune development, and defense [17]. However, an increased level of apoptosis causes excess cell death in many diseases [17]. Regardless of the diabetes type, highly conserved intracellular pathways of apoptosis are triggered and lead to a point of no return in apoptosis to influence β-cells, which provokes more metabolic dysfunctions and thereby cause diabetic complications [18]. The results of the highly complex interaction of multiple distinct but overlapping mechanisms are some typical changes in the structure of the heart, including cardiac stiffness, hypertrophy, and fibrosis, leading to cardiac dysfunction, combined with cardiomyocyte death that will promote the progress of heart failure [1, 13].
Behind these pathological features, there are interactions among multiple signaling pathways. ROS could enhance nuclear factor-κB (NF-κB) signaling pathway as a maladaptive immune modulation that prompts cardiac remodeling and fibrosis [12, 19]. The increased ROS and impaired adenosine monophosphate-activated protein kinase (AMPK) signaling pathway further decrease fatty acid oxidation (FAO) and then lead to lipid accumulation and diastolic dysfunction [13]. Inappropriate activation of the renin-angiotensin-aldosterone system impairs the phosphatidylinositol 3-kinase (PI3K)-protein kinase B (Akt) signaling pathway, further increasing intracellular Ca2+ levels and Ca2+ sensitivity and then resulting in cardiac fibrosis/stiffness and diastolic dysfunction [12]. AGEs could increase fibrosis, cardiac stiffness, and impaired diastolic relaxation by increasing the production of ROS and activating the mitogen-activated protein kinase (MAPK) signaling pathway [13]. Meanwhile, AGEs could stimulate the expression of collagen, the crosslinks of collagen molecules, and the accumulation of collagen [13, 20, 21]. The receptor for AGEs could induce the expression of transforming growth factor-β (TGF-β) to elicit the forming of myofibroblasts [22, 23].
Oxidative stress, an imbalance status between prooxidants and antioxidants, may perform a central role in the pathogenesis of DCM via impacting β-cells and cardiac cells. A constant weakening in β-cells quantities and utility is one of the characteristics of the natural history of diabetes, which tightly relates to microvascular or macrovascular complications of DM, including DCM [24]. The pancreatic β-cells, with lower levels of free radical detoxifying and redox-regulating enzymes, contrasted to other cell types, may be at a higher hazard for oxidative injury with boosted sensitivity for apoptosis [25]. ROS and reactive nitrogen species (RNS) are the two chief cellular generation sites of redox-reactive species [19]. Under physiological conditions, ROS/RNS regulates insulin secretion and insulin action; conversely, under pathological conditions, ROS/RNS prompts the deactivation of metabolic enzymes, suppression of insulin secretion, and death of β-cells [26]. The activity of nicotinamide adenine dinucleotide phosphate [NAD(P)H] oxidase (NOX) elevated upon Ca2+ stimulation, then prompt ROS accumulation in β-cells increased rapidly, and attain harmful levels to influence the progression of DM and its complications [25]. NOXs, an important ROS-producing enzyme, not only trigger oxidative damage in β-cells but also regulate both adaptive and maladaptive changes in the cardiomyocytes [27]. The physiological amounts of nitric oxide (NO) are a vital coupling factor in insulin-secreting cells; however, the excessive NO production, which may relate to inflammation, can generate oxidative/nitrosative stress, which is one of the crucial procedures of β-cells death [19, 28].
ROS is regarded as a prototypical senescence inducer, and the considerable amount of ROS and RNS in adult cardiac muscle cells leads to cardiotoxicity [29]. ROS-induced aging and cardiotoxicity prompt cardiac stem cells senescence and then reduce cardiac muscle function, especially in patients with DM [29]. Besides broad oxidation inducing cell dysfunction, necrosis, or apoptosis, dysregulated ROS/RNS signaling also leads to specific posttranslational modifications which could alter the function of vital cellular proteins and signaling pathways in the heart [30]. For example, bromodomain-containing protein 4 is a critical protein in the modulation of various biological processes, and its expression has been detected upregulated in the DCM [31, 32]. ROS production could trigger the expression of bromodomain-containing protein 4 to prompt cardiac hypertrophy, and this progression also relates to signaling pathways for inflammation, fibrosis, and so on [31]. NO is an oxygen-derived free radical and is synthesized by three NO synthase isoforms including inducible NO synthase (iNOS), endothelial NOS (eNOS), and neuronal nitric oxide synthase [19, 33, 34]. Abnormalities in vascular NO production and transport accompany many disease states, including cardiovascular diseases and diabetes [34]. The pathological amounts of NO are related to vascular endothelial dysfunction, which is considered a major mediator in diabetic cardiomyopathy [35]. The reduction of bioavailability of NO in the vasculature is one of the characteristics of IR, and improving the bioavailability of NO could also help coronary vasodilation [36, 37].
The pathogenesis of DCM is extremely complex and involves multiple signaling pathways. Oxidative stress is the major factor responsible for poor prognosis and survival in patients with DCM, and it could impact both β-cells and cardiac cells. The mechanisms of oxidative stress in DCM have been summarized in Figure 1. Antioxidation is one of the promising therapeutic strategies for DCM and calls for more attention.
Figure 1.
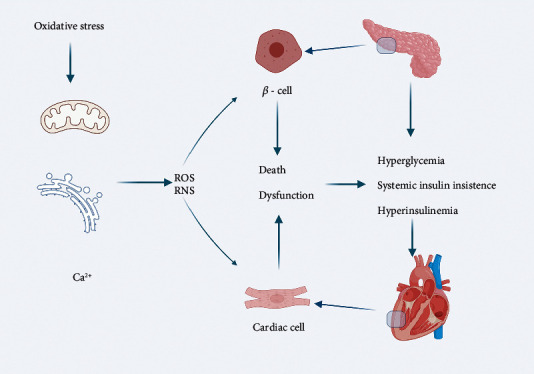
Oxidative stress in the pathogenesis of DCM (created with BioRender.com). In patients with diabetes, the main abnormalities of the inner environment are hyperglycemia, systemic insulin insistence, and hyperinsulinemia. Oxidative stress could trigger mitochondrial dysfunction and endoplasmic reticulum stress, and impair calcium handling and increase Ca2+ sensitivity and Ca2+ influx. ROS/RNS could impact both β-cells and cardiac cells leading to cell death and dysfunction. ROS: reactive oxygen species; RNS: reactive nitrogen species.
2.2. Relationship of Nrf2 and DCM
Nrf2 performs a critical character in counterbalancing oxidative stress and inflammation. Kelch-like ECH-associated protein 1 (Keap1) sequesters Nrf2 in cytoplasmic usually, but under conditions of oxidative stress, it resolves with Nrf2 in a dose-independent manner [38]. The resolved newly synthesized Nrf2 translocates into the nucleus and then binds to the small Maf proteins to form a new protein dimer [10]. Furthermore, the heterodimer can recognize the antioxidant response elements (AREs), which locate in the regulatory domains of multiple defense enzyme genes [11]. AREs then transcript heme oxygenase-1 (HO-1), NAD(P)H quinone dehydrogenase-1 (NQO1), superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPX), glutathione S-transferase (GST), and γ-glutamylcysteine synthetase (γ-GCS) to defend the cell against oxidative stress [39, 40].
Nrf2 is broadly accepted as having a remarkable role in combating oxidative stress, and investigations have claimed that the expression of Nrf2 in diabetic animals and patients is significantly diminished [41, 42]. The decreased expression of Nrf2 leads to cardiac damage and is correlated to IR, abnormal angiogenesis, and endothelial dysfunction [41]. However, increasing the expression of Nrf2 can guard the cardiac cells and heart against the hyperglycemia environment in vitro and in vivo [42]. Chinese herbal monomers could upregulate Nrf2 to reverse this condition. Abdelsamia et al. suggested the advantages of metformin/curcumin combination in counteracting DCM [43]. They treated diabetic rats with curcumin (100 mg/kg/d) for 6 weeks and then observed Nrf2; HO-1 upregulated; and the metformin/curcumin combination group is superior to the metformin and curcumin group [43]. Atta et al. noted that 12 weeks oral of thymoquinone (50 mg/kg/d) in diabetic rats could upregulate Nrf2 and SOD, as well as downregulate iNOS and NO [44].
Previous studies also suggested that activating Nrf2/HO-1 pathway could positively attenuate the death of cardiomyocytes [45]. The strategy of targeting Nrf2 could enhance the expression of Nrf2 in the cardiac to elevate the expression of HO-1 in the myocardial and then diminish cardiac hypertrophy and cardiac dysfunction [46]. Wang et al. fed T1DM mice with resveratrol (10 mg/kg per day) for 1 month and afterward observed for 6 months and showed that cardiac function improved and fibrosis reduced which is accompanied by upregulating Nrf2, HO-1, SOD, and NQO1 [47]. Dong et al. evidenced in vitro that gastrodin could defend against hyperglycemia-induced cardiomyocyte toxicity through upregulating Nrf2, SOD, and CAT [48]. Duan et al. found that Aralia taibaiensis could reduce intracellular ROS levels and cell oxidative injury accompanied by enhancing the expression of Nrf2, SOD, and GSH [49].
The Nrf2 system is also responsible for maintaining lipid metabolism and glucose metabolism by regulating glucose utilization and insulin secretion to convert the progression of DM [50]. Castillo et al. treated rats with quercetin (0.5% w/w) for 4 weeks and then observed quercetin counteracted hyperglycemia-induced bioenergetic deterioration, including avoiding cardiac cholesterol accumulation, accompanied by upregulating Nrf2, HO-1, SOD, and proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) [51]. PGC-1α is regarded as a master regulator of mitochondria, which is related to intracellular energy homeostasis [52]. In a word, Nrf2, as an antioxidant factor, could efficiently ameliorate DCM.
2.3. Main Signaling Pathways in the Pathogenesis of DCM and Crosstalk with Nrf2
The pathogenesis of DCM involves diverse signaling pathways to exercise different functions, and these mechanisms are summarized in Table 1. For ameliorating DCM, Nrf2 plays a pivotal role in crosstalk with these pathways and that would be discussed in detail in the following text.
Table 1.
Mechanisms of diverse signaling pathways in the pathogenesis of DCM.
| Signaling pathway | Mechanism | |
|---|---|---|
| NF-κB signaling pathway | Increase inflammation and apoptosis | |
| AMPK signaling pathway | Improve utilization of glucose Maintain energy homeostasis Decrease lipid accumulation, ROS production, and inflammation |
|
| Akt signaling pathway | Regulate insulin signaling Decrease apoptosis |
|
| MAPK signaling pathway | Regulate insulin signaling Lead growth and remodeling responses Increase inflammation |
|
| TGF-β signaling pathway | Increase fibrosis and apoptosis | |
2.3.1. Effects of NF-κB Signaling Pathway and Crosstalk with Nrf2
NF-κB is one of the major signaling pathways involved in the pathogenesis of DCM. Mainly transcription factors in mammals of the NF-κB family include p50, p52, p65, Rel, and RelB [53, 54]. NF-κB is expressed in nearly all cell sorts, and the family of inhibitors of NF-κB (IκB) could make it inactive in the cytoplasm [55, 56]. This signaling pathway could be activated in multiple ways, including ROS and RNS levels, toll-like receptors (TLRs), interleukin-1 (IL-1), IL-6, and tumor necrosis factor α (TNF-α) [19, 55, 57, 58, 59]. TLRs, inside the human body, it is named TLR4, are components of the innate immune, whose activation can produce inflammatory cytokines and systematically affects vascular function and remodeling [60]. Myeloid differentiation primary response protein 88 (MyD88), one of the adapters of TLR4, is a kinase that performs an essential role in triggering NF-κB signaling [57]. TLR4 can also bind to NOX4 and subsequently generate ROS, thus provoking oxidative damage [19, 61]. Proinflammatory cytokines, such as IL-1β, are produced by inflammasomes, which are a group of protein complexes built around several proteins, including nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) [62]. Besides that, NF-κB could transcript some cell adhesion molecules, which firmly adhesion to leukocytes to migrate into injured tissues, such as intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecular-1 (VCAM-1) [63]. Monocyte chemoattractant protein-1 (MCP-1), a member of chemotactic cytokines, could promote adhesion via the upregulated related receptor, such as the receptor of ICAM-1 [64].
NF-κB signaling pathway performs a vital part in the pathophysiology of DCM through involving in the transcription of different proinflammatory and inflammatory [65]. During inflammatory responses, cellular events are tightly associated with redox balance [66]. Between Nrf2 and NF-κB signaling pathways, there is an existing complex and dynamic interplay, and both modulate the physiological homeostasis of cellular redox status and responses to stress and inflammation [10]. Nrf2 signaling pathway could decrease the production of ROS in intracellular and then could inhibit proinflammatory signals in general [67]. Nrf2 plays a major role in anti-inflammatory and includes counteracting NF-κB-driven inflammatory, and this supposedly has been evidenced by multiple studies [10, 68]. Raish et al. stated that sinapic acid (20 and 40 mg/kg oral for 12 weeks) can upregulate GPX, SOD, CAT, IκB-α/β, Nrf2, and HO-1 while downregulating TNF-α, IL-6, and NF-κB [45]. Lian et al. observed the potential of chrysophanol for antioxidant and anti-inflammation is Nrf2-dependent [69]. They gave mice 25 and 50 mg/kg/d chrysophanol solution for 19 weeks and then noted Nrf2, HO-1 are upregulated and is accompanied with IL-6, IL-18, IL-1β, TNF-α, ICAM-1, and VCAM-1 are downregulated, but this result does not show in Nrf2 knockout mice [69]. Chen et al. also observed this dependency; they found that kaempferol can enhance Nrf2 activity in cells and upregulate HO-1, NQO1, SOD, and IκB-α, as well as downregulate TNF-α and IL-6, but when knockdown of Nrf2 in H9c2 cells, kaempferol has no attenuation effect on ROS production [70].
NF-κB signaling pathway also performs a crucial character in apoptosis. In β-cells, activated NF-κB could upregulate proapoptotic (e.g., Bax) and downregulate antiapoptotic (e.g., Bcl2) [19]. Besides, excessive production of ROS provokes the apoptosis of cells through combination with inflammatory [19]. Nrf2 signaling pathway could inhibit apoptosis in multiple ways, such as transcript antioxidant factors, suppressing NF-κB signaling pathway. Liang's study could approve this result. After 12 weeks of oral for 1, 10, and 20 mg/kg/d in diabetes mice, andrographolide could upregulate SOD, Nrf2, HO-1, and IκB-α, as well as downregulate p65, NF-κB, TNF-α, IL-1β, IL-6, and Bax/Bc12 [71]. Li et al. found that piceatannol in vitro could enhance the expression of Bc12, Nrf2, HO-1, SOD, and IκB-α and, meanwhile, reduce the expression of Bax, p65, and caspase3 [72]. The family of caspase is related to apoptosis β-cells [19].
The main crosstalk mechanisms in Nrf2 and NF-κB signaling pathway are as follows. Firstly, Nrf2-dependent antioxidant genes, such as HO-1 and NQO1, could limit the activation of NF-κB to attenuate inflammation via blocking TNF and TLR4-depending signaling pathways [68, 73]. Yan et al. observed that scutellarin could regulate both Keap1/Nrf2/ARE and TLR4/MyD88/NF-κB signaling pathways [74]. According to Yan's study, scutellarin could upregulate the expression of SOD, CAT, GPX, GST, Nrf2, NQO1, HO-1, and IκB-β while downregulating Keap1, TLR4, Myd88, p50, IL-6, and TNF-α after a 6 weeks oral (10 or 20 mg/kg/day) [74]. Xu et al. gave mice bixin solution 50, 100, and 200 mg/kg/d for 14 weeks and then found that Nrf2, SOD, HO-1, and CAT upregulated; meanwhile, TLR4, Myd88, IκB-α, and NF-κB downregulated [75]. Enhancing the expression of HO-1 not only inhibits the TNF-dependent activation of NF-κB but also reduces VCAM-1 expression in aortic endothelial cells; the behind mechanism may be that HO-1 can impede the transcriptional machinery of NF-κB in the nucleus [76]. Li et al. observed that 15 weeks oral of luteolin (20 mg/kg/d) could upregulate Nrf2, HO-1, and NQO1; meanwhile, it could downregulate IL-1β, IL-6, TNF-α, MCP-1, ICAM, and VCAM [77]. Secondly, Nrf2 could impede the activation of NLRP3 inflammasome. Nrf2 and NQO1 are involved in the progression, impeding the priming step to decrease the activity of NLRP3 inflammasome, and it also suppresses caspase-1 cleavage and subsequent IL-1β generation [78]. Thirdly, Keap1 could inhibit the activity of NF-κB via ubiquitinating IκB kinase [79]. Furthermore, Keap1 could be targeted by 15d-PGJ2 to initiate gene transcription with an overall anti-inflammatory result [80]. 15d-PGJ2 is a product of NF-κB-induced cyclooxygenase-2, where interesting is that NF-κB system could manage its termination by expression of other target genes [80]. Fourthly, NF-κB could compete with Nrf2 to combine with cAMP-response-element-binding protein-binding protein, a transcriptional co-activator [68]. The mechanisms have been summarized in the figure (see Figure 2). In conclusion, the NF-κB signaling pathway triggering inflammation and apoptosis to lead the poor prognosis of DCM and Nrf2 could improve this via crosstalk with it.
Figure 2.
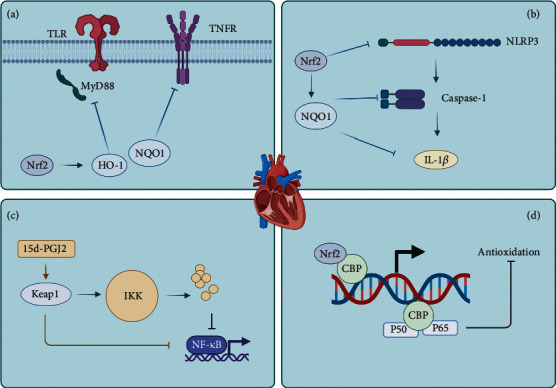
The crosstalk between Nrf2 and NF-κB signaling pathway (created with BioRender.com). (a) Nrf2-dependent antioxidant genes could block TLR and TNF-dependent signaling. (b) Nrf2 and NQO1 inhibit the priming step of NLRP3, and suppress caspase-1 cleavage and IL-1β generation. (c) Keap1 could ubiquitinate IKK and be targeted by 15d-PGJ2. (d) Nrf2 and NF-κB could compete to combine with CBP. Nrf2: nuclear factor erythroid-2 related factor 2; NF-κB: nuclear factor-κB; HO-1: heme oxygenase-1; NQO1: NAD(P)H quinone dehydrogenase-1; TLR: toll-like receptors; TNFR: tumor necrosis factor receptor; NLRP3: nucleotide-binding oligomerization domain-like receptor protein 3; IL-1β: interleukin-1β; Keap1: kelch-like ECH-associated protein 1; IKK: inhibitors of NF-κB kinase; CBP: cAMP-response-element-binding protein-binding protein.
2.3.2. Effects of AMPK Signaling Pathway and Crosstalk with Nrf2
AMPK has been regarded as an enzyme that performs a crucial part in maintaining energy homeostasis, reduction of ROS production in the cytosol, and utilization of glucose [81–83]. The activity of AMPK was considerably decreased in DCM, and increasing the activity of AMPK would significantly diminish lipid accumulation and revamp cardiac function [8]. In cardiac, AMPK is a major kinase to regulate myocardial metabolism through controlling numerous metabolic pathways, such as lipid metabolism and utilization [8, 83]. Under normal physiological conditions, the adult heart gains about 50-75% of its acetyl coenzyme A (CoA)-derived ATP from FAO, but also could rapidly adjust to alterations in substrate availability for the generation of ATP to incessantly maintain its energy requirements, which termed “metabolic flexibility” [83]. However, in pathological cardiac hypertrophy and dilated cardiomyopathy, there would be some changes in transcription that prompt the diminish of this metabolic flexibility, which contributes to the pathogenesis of heart failure [83]. Once AMPK is activated, it could increase fatty acids entering the mitochondria through carnitine palmitoyl CoA transferase 1 for FAO [84]. Acetyl-CoA carboxylase (ACC) is a protein that could catalyze the transformation of acetyl CoA to malonyl-CoA, and malonyl-CoA could negatively regulate carnitine palmitoyl CoA transferase 1 [85]. Activated AMPK could diminish malonyl-CoA levels and increase FAO through phosphorylating and inhibiting ACC [86]. Furthermore, both AMPK and silent information regulator 1 (SIRT1) are regarded as the gatekeepers of the activity of PGC-1α, and the activated AMPK/SIRT1/PGC1-α signaling pathway contributes to a regulatory network for metabolic homeostasis [52].
Both AMPK and Nrf1/2 are the crucial regulator of mitochondrial dynamics and synergistic to maintain cardiovascular energy homeostasis [15, 87]. Enhancing the expression of Nrf2 could help AMPK to improve cardiac function. Li et al. stated that bailcalin improves diabetes-induced cardiac dysfunction via AMPK/Nrf2 signaling [8]. After administering diabetic mice with bailcalin (100 mg/kg/d, 4 months), it was found that it can upregulate AMPKα, CPT-1, PGC1-α, glutathione (GSH), SOD, and Nrf2 while downregulating atrial natriuretic peptide (ANP), B-type natriuretic peptide (BNP), β-myosin heavy chain (β-MHC), ACC, and oxidized glutathione (GSSG) [8]. GSH is regarded as one of the most essential scavengers of ROS, and its ratio with GSSG may be considered a biomarker of oxidative stress [88]. BNP is a valuable indicator in the diagnosis of heart failure, and its elevation is correlated with disease severity, especially left ventricular systolic ejection fraction and left ventricular diastolic function [89]. The meaning of ANP is nearly the same as BNP, but it has a different mechanism [90]. In cardiomyocytes, isoforms of β-MHC have been evidenced to change cardiac muscle function both in healthy developing and diseased hearts [91]. Both BNP and β-MHC are cardiac hypertrophy marker proteins [92]. Du et al. found that in vitro notoginsenoside R1 could upregulate Nrf2, HO-1, and AMPK, as well as downregulate ANP and BNP [93].
Besides, AMPK could prompt the Nrf2-mediated antioxidative cascade while inhibiting inflammation via suppression of TLR-mediated proinflammatory cascades [94]. Kosuru et al. stated that pterostilbene, 8 weeks oral for 20 mg/kg/d in rats, could upregulate SOD, CAT, GSH, GPX, PGC-1α, Nrf2, HO-1, and AMPK; meanwhile, it could downregulate IL-1β, IL-6, TNF-α, NF-κB, TLR4, and NLRP3 [95]. Zhao et al. observed that fortunellin, 8 weeks oral for 10, 20, and 30 mg/kg in mice, could upregulate SOD, Nrf2, HO-1, and AMPK and downregulate TNF-α, IL-1β, IL-6, IL-18, NF-κB, and Keap1 [96]. Altamimi et al. revealed that ellagic acid, 8 weeks for 100 mg/kg/d in rats, could upregulate GSH, SOD, Nrf2, and SIRT1, as well as downregulate BNP, TNF-α, and IL-6 [97].
Although whether Nrf2 is a direct molecular target of AMPK is unclear, Nrf2 could be activated in an AMPK-dependent way and as a downstream factor [94]. The crosstalk mechanisms between Nrf2 and AMPK signaling pathway are concluded in the following aspects. Firstly, glycogen synthase kinase 3β (GSK3β), which is regarded as an activation switch of Nrf2 gene expression, is a key protein in the crosstalk between Nrf2 and AMPK. Nrf2 could be phosphorylated by AMPK at the Ser550 residue, and then combine with AMPK-mediated GSK3β inhibition, further enhancing the nuclear accumulation of Nrf2 for ARE-driven gene transactivation [98]. Phosphorylated GSK3β was also found that this could improve cardiac function [99]. Cao et al. found in vitro that Z-ligustilide could restore cardiomyocyte dysfunction via upregulating AMPK, Nrf2, and SOD while downregulating GSK3β [100]. Secondly, activated AMPK could promote p62-dependent autophagic degradation of Keap1, which leads Nrf2 to separate from Keap1 and translocate to the nucleus [101]. Thirdly, SIRT1, which could be activated by AMPK via increasing the substrate, could regulate Nrf2 to attenuate oxidative damage [102]. Besides, a family of secreted frizzled-related proteins (Sfrps) recently had been reported to be widely associated with the pathogenesis and prognosis of DCM, including apoptosis, inflammation, and oxidative stress, and then lead to the events of cardiac fibrosis and even heart failure [103–108]. Although Sfrps has been evidenced could reduce oxidative stress in an AMPK/PGC1-α-dependent manner, studies are deficient about the direct relationship between Nrf2 and Sfrps [108]. However, between Nrf2 and Sfrps, there are so many same proteins involved, such as GSK3β and PGC1-α, and the related studies should be expected [104, 105, 108]. In short, besides combating oxidative stress, AMPK and Nrf2 jointly work to revamp cardiac function via maintaining heart energy homeostasis, improving utilization of glucose, and decreasing lipid accumulation and inflammation based on the above mechanisms.
2.3.3. Effects of Akt Signaling Pathway, and Crosstalk with Nrf2
PI3K/Akt signaling pathway also significantly influences the prognosis of DCM. IR could independently predict the mortality of individuals with heart failure [109]. Akt signaling pathway is one typical pathway responsible for cellular insulin signaling, which is beneficial to the glucose uptake in the heart [13]. Impairment of the insulin-induced activation of PI3K/Akt is one of the characteristics of IR [109]. Activated Akt signaling pathway could ameliorate cardiac IR [110]. Recently, accumulating evidence has suggested that oxidative stress plays a causal role in the cardiac complications of IR too [109]. Akt signaling pathway evidenced that it could enhance the expression of Nrf2 and then jointly against IR to improve cardiac function [111, 112]. Xu et al. observed that cardiac function improved in mice after 4 weeks of intraperitoneal injection with scutellarin (5, 10, 20 mg/kg), accompanied by enhancing the expression of Nrf2, HO-1, and Akt [113].
Akt signaling pathway could be activated in multiple ways, including insulin receptor substrate (IRS), and estrogen receptors. IRS represented a classical insulin-induced way to activate Akt signaling pathway, which contributes to ameliorating prognosis, including improving cardiac function and apoptosis. Ma et al. evidenced that low expression of SIRT1 induces the decrease of IRS-2 and further does not activate Akt signaling pathway [110]. According to Ma's study, the level of ANP and BNP significantly increased in the SIRT1 knockout mice, and 5 consecutive days treated with resveratrol (25 mg/kg/d) in mice could reverse this condition, by enhancing the expression of Nrf2, SIRT1 [110]. Furthermore, IRS-1 could initiate eNOS through Akt activating way [37]. In diabetic rats, the blocked PI3K/Akt signaling pathway results in the reduction of protein expression of eNOS, which could also regulate the level of apoptosis [112]. Liu et al. showed that spiraeoside in vitro could upregulate Akt, Nrf2, HO-1, Bcl2, SOD, GPX, and CAT and downregulate caspase3, caspase7, and Bax [114]. Estrogen receptors-α36-G protein-coupled estrogen receptor signaling complex could rapidly induce the generating of ceramide, which is necessary for signaling of ceramide-protein kinase C ζ-casein kinase 2 (CK2) [115]. CK2 further supports the activation of diverse signaling kinases, including Akt signaling [115].
The crosstalk mechanisms between Akt and Nrf2 are the following aspects. Firstly, GSK3β is regarded as a crucial protein. GSK3β, a multifunctional serine/threonine kinase, could phosphorylate Fyn, and then phosphorylate Nrf2 tyrosine 568, finally provoking the degradation of Nrf2 [116]. Akt could phosphorylate GSK3β at Ser 9 to make it deactivate to facilitate the accumulation of Nrf2 [111, 116]. Zhang et al. found that in vitro myricitrin could increase Nrf2, HO-1, γ-GCS, NQO1, and Akt, as well as downregulate GSK3β [117]. Duan et al. showed that every other day for 15 days oral for 10, 20, and 40 mg/kg in diabetic mice, butin could upregulate SOD, Nrf2, HO-1, and Akt, as well as downregulate Keap1, GSK3β, and Fyn [116]. Besides, CK2 contributes to the accumulation of Nrf2 not only by activating the PI3K/Akt axis but also by directly phosphorylating Nrf2 to enhance its stability [115]. Briefly, Akt and Nrf2 signaling pathways could alleviate IR and apoptosis to improve the prognosis of DCM through regulating cellular insulin signaling.
2.3.4. Effects of MAPK Signaling Pathway and Crosstalk with Nrf2
MAPK is a vital target signaling pathway for treating DCM. MAPK is a key signal transduction pathway in regulating cellular insulin signaling, which mainly relates to the disturbance in the metabolic and growth effects of insulin signaling [13]. The activated MAPK signaling pathway is related to growth and remodeling responses, which leads to myocardial hypertrophy, cardiac fibrosis, impaired myocardial endothelial signaling, and death of myocardial and endothelial cells [13]. Besides, MAPK is regarded canonical intracellular signaling pathway related to inflammation and immune [118]. Downregulating the MAPK pathway could alleviate chronic inflammation in diabetic mice via Chinese herbal monomers, such as berberine [119].
Chinese herbal monomers have been observed that they can regulate MAPK and Nrf2 signaling pathways in cardiac at the same time. MAPK family includes c-Jun N-terminal kinase (JNK), extracellular signal-regulated kinase (ERK), and p38. Overactivated phosphorylated ERK, which tightly relates to IR in cardiac, is always accompanied by a depressed expression of cardiac Nrf2 [109]. Furthermore, insulin-induced ERK activity was significantly decreased by the forced activation of Nrf2, which indicated that activation of Nrf2 could diminish the activity of oxidative stress-induced ERK in adult cardiomyocytes [109]. Chinese herbal monomers have been evidenced that they could inhibit ERK1/2 and p38 MAPK phosphorylation in angiotensin II-treated neonatal rat ventricular myocytes, which contribute to alleviating cardiac hypertrophy [120, 121]. Activated JNK, a key marker of tissue injury, was previously shown to relate to IR, increased ROS generation, and ERS under hyperglycemic conditions [122, 123]. The Nrf2 inhibitor could increase the expression of JNK [123]. The ERK1/2 and JNK are downstream factors of the Nrf2 pathway, involved in DCM, and upregulated by NOX and Nrf2 deficiency-stimulated ROS production [123]. Gu et al. showed isoliquiritigenin in vitro could upregulate Nrf2 and HO-1 and meanwhile downregulate TNF-α, IL-6, IL-1β, VCAM-1, MCP-1, JNK, ERK, and p38 [118]. Lu et al. suggested that hinokinin could protect against cardiac injury; they treated diabetic mice with hinokinin (20 and 40 mg/kg) for 6 weeks and then observed that Nrf2, HO-1, and SOD upregulated, while Keap1, JNK1, ERK1/2, and p38 downregulated [124]. Ni et al. found that in vitro salidroside could upregulate Nrf2 and HO-1, as well as downregulate ERK, JNK, and p38 to protect against cardiomyocyte apoptosis and ventricular remodeling [125]. Nrf2 could inhibit MAPK signaling pathway to ameliorate the prognosis of DCM; however, the relationship between Nrf2 and MAPK calls for more research details.
2.3.5. Effects of TGF-β Signaling Pathway and Crosstalk with Nrf2
Fibrotic diseases are a result of an imbalance between profibrotic and antifibrotic cytokines and secreted proteins, whose character is excessive scarring caused by excessive production, deposition, and contraction of ECM [126]. The degradation of ECM is regulated by matrix metalloproteinases (MMPs), and the dysregulation of MMPs function, specifically MMP-2 and MMP-9, could provoke myocardial remodeling and the development of heart failure [127, 128]. Fibrosis is one of the most prevalent characteristics of diabetes, and recent evidence posted the term “redox fibrosis” [129]. The term “redox fibrosis” means that oxidative stress and the antioxidant system might be the essential mechanism behind fibrosis development and persistence, and the potential target of antifibrosis is the antioxidant system [129].
TGF-β and connective tissue growth factor (CTGF) are important profibrotic proteins [126]. TGF-β could induce fibroblasts to synthesize and contract ECM, and it has been regarded as a dominant regulator in the responding of fibrotic for a long time, which performs a central part of fibrogenesis in almost all organs [129, 130]. CTGF, whose regulation is mediated by TGF-β, could enhance the action of TGF-β on cells [131]. In DM, these factors prompt cardiomyopathy fibrosis and reduced compliance of the heart [13, 20, 21]. It has been evidenced that NOX4 is the most responsible factor for ROS-induced activation of fibroblast and mesangial cells and performs an essential part in the activation of TGF-β1 signaling and differentiation into a profibrotic myofibroblast phenotype and matrix production [132]. This implies that antioxidant stress can also be used to fight fibrosis. Several studies have evidenced that enhancing Nrf2 could antifibrosis via inhibiting TGF-β. Liao et al. demonstrated that those 6 months of myricetin treatment (200 mg/kg/d) could upregulate Nrf2, HO-1, NQO1, and SOD, as well as downregulate collagen I, collagen III, fibronectin, CTGF, Smad3, and TGF-β [133]. Ma et al. observed that bakuchiol in vitro could upregulate Nrf2, SOD, and GPX, as well as downregulate collagen I, collagen III, α-smooth muscle actin (α-SMA), TGF-β, and Smad3 [134]. α-SMA is the biomarker of mature myofibroblasts, which is also regarded as a cardiac fibrotic marker, and the mechanism behind it might be involved in the contraction and remodeling of the extracellular matrix [135]. Ying et al. treated mice with phloretin (10 mg/kg every 2 days) for 7weeks and showed that phloretin could upregulate Nrf2, HO-1, and NQO1 and, meanwhile, downregulate TGF-β, collagen I, and CTGF [136].
The increased activation of the TGF-β signaling pathway is one of the underlying mechanisms for increased rates of apoptosis [20]. Smad3 is the chief transcription factor of TGF-β, and the elevation of its phosphorylation levels in human T2DM islets indicates an autocrine role for TGF-β/Smad3 signaling in the apoptosis of β-cells; the same results are observed in diabetic mice [137]. NOX 4 regulates the activation of Smad2/3 to mediate the TGF-β1-induced transformation of fibroblasts to myofibroblasts [132]. Increasing the expression of Nrf2 could be antiapoptotic via inhibiting TGF-β. Alshehri et al. gave diabetic rats a daily oral dose of kaempferol solution (50 mg/kg) and then observed that Nrf2, GSH, and Bcl2 upregulated; meanwhile, TGF-β1 and Bax were downregulated [138].
The main crosstalk mechanisms between Nrf2 and TGF-β are as follows. Firstly, Nrf2 could reduce MMP-9 to decrease the levels of TGF-β [139, 140]. Secondly, Nrf2-mediated Smad inhibition could be tightly associated with enhanced Smad7 levels [141]. Smad7 could form a complex type I receptor, and it recruits Smad-mediated ubiquitination regulatory factor 1/2 to activate the type I receptor, thus negatively regulating the TGF-β signaling pathway [141]. Zhang et al. detected that 20-week oral of notoginsenoside R1 (7.5, 15, and 30 mg/kg/d) could upregulate Nrf2, HO-1, γ-GCS, NQO1, and Smurf2 as well as downregulate TGF-β, collagen I, Bax/Bc12, caspase-3, caspase-9, and Smad2/3 [40]. Li et al. showed that 8 weeks oral of syringaresinol (25 mg/kg every other day) in diabetic mice could upregulate Nrf2, NQO1, HO-1, and SOD, as well as downregulate TGF-β, fibronectin, α- SMA, Smad2/3, Bax/Bc12, and Keap1 [142]. There exists a tight relationship between fibrosis, apoptosis, and oxidative stress, and Nrf2-dependent combating oxidative stress would be a potential therapeutic strategy.
The mechanisms of crosstalk between Nrf2 and AMPK, Akt, MAPK, and TGF-β signaling pathways have been summarized in the figure (see Figure 3). The above-mentioned mechanisms of Chinese herbal monomers are summarized in Table 2.
Figure 3.
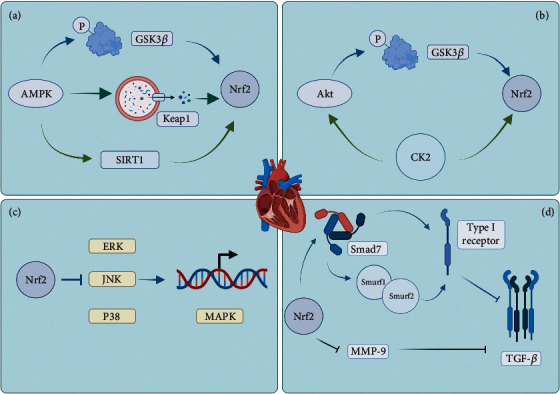
The crosstalk between Nrf2 and other signaling pathways (created with BioRender.com). (a) AMPK could phosphorylate both Nrf2 (active) and GSK3β (deactivate) to prevent Nrf2 ubiquitinated by GSK3β to improve the nuclear accumulation of Nrf2. AMPK could promote autophagic degradation of Keap1. Besides, AMPK could activate SIRT1 to regulate Nrf2. (b) Akt could phosphorylate GSK3β to prompt nuclear accumulation of Nrf2. Furthermore, CK2 could not only activate Akt signaling pathway but also could directly phosphorylate Nrf2 to enhance its stability. (c) ERK, JNK, and p38 are always observed accompany by suppression of Nrf2. But the underlying mechanism is unclear. (d) Nrf2 could reduce MMP-9 to decrease the levels of TGF-β. Besides, Smad7 could form a complex type I receptor, and it recruits Smurf 1/2 to activate the type I receptor, thus negatively regulating TGF-β signaling pathway. And Nrf2 could enhance the level of Smad7. AMPK: adenosine monophosphate-activated protein kinase; GSK3β: glycogen synthase kinase 3β; SIRT: silent information regulator 1; Akt: phosphatidylinositol 3-kinase-protein kinase B; CK2: ceramide-protein kinase C ζ-casein kinase 2; ERK: extracellular signal-regulated kinase; JNK: c-Jun N-terminal kinase; MMP: matrix metalloproteinases; TGF-β: transforming growth factor-β; Smurf: Smad-mediated ubiquitination regulatory factor.
Table 2.
Mechanisms behind Chinese herbal monomers ameliorate DCM based on Nrf2.
| Reference | Author | Component | Experiment | Mechanism |
|---|---|---|---|---|
| [43] | Eman M Abdelsamia | Curcumin | In vivo | Upregulate Nrf2, HO-1 |
| [44] | Mustafa S Atta | Thymoquinone | In vivo | Upregulate Nrf2, SOD Downregulate iNOS, NO |
| [47] | Guan Wang | Resveratrol | In vivo | Upregulate Nrf2, HO-1, SOD, NQO1 |
| [48] | Z Dong | Gastrodin | In vitro | Upregulate Nrf2, SOD, CAT |
| [49] | Jialin Duan | Aralia taibaiensis | In vitro | Upregulate Nrf2, SOD, GSH |
| [51] | Rodrigo L Castillo | Quercetin | In vivo | Upregulate Nrf2, HO-1, SOD, PGC-1α |
| [45] | Raish Mohammad | Sinapic acid | In vivo | Upregulate GPX, SOD, CAT, IκB-α/β, Nrf2, HO-1 Downregulate TNF-α, IL-6, NF-κB |
| [69] | Yonggang Lian | Chrysophanol | In vivo | Upregulate Nrf2, HO-1 Downregulate IL-6, IL-18, IL-1β, TNF-α, ICAM-1, VCAM-1 |
| [70] | Xuemei Chen | Kaempferol | In vitro | Upregulate Nrf2, HO-1, NQO1, SOD, IκB-α Downregulate TNF-α, IL-6 |
| [71] | Ershun Liang | Andrographolide | In vivo | Upregulate SOD, Nrf2, HO-1, IκB-α Downregulate p65, NF-κB, TNF-α, IL-1β, IL-6, Bax/Bc12 |
| [72] | Hao Li | Piceatannol | In vitro | Upregulate Bc12, Nrf2, HO-1, SOD, IκB-α Downregulate Bax, p65, caspase3 |
| [74] | Huo Yan | Scutellarin | In vivo | Upregulate SOD, CAT, GPX, GST, Nrf2, NQO1, HO-1, IκB-β Downregulate Keap1, TLR4, Myd88, p50, IL-6, TNF-α |
| [75] | Zhou Xu | Bixin | In vivo | Upregulate Nrf2, SOD, HO-1, CAT Downregulate TLR4, Myd88, IκB-α, NF-κB |
| [77] | Li Li | Luteolin | In vivo | Upregulate Nrf2, HO-1, NQO1 Downregulate IL-1β, IL-6, TNF-α, MCP-1, ICAM, VCAM |
| [8] | Li Ran | Bailcalin | In vivo | Upregulate AMPKα, SOD, CPT-1, PGC1-α, GSH, Nrf2 Downregulate ANP, BNP, β-MHC, ACC, GSSG |
| [93] | Fawang Du | Notoginsenoside R1 | In vitro | Upregulate Nrf2, HO-1, AMPK Downregulate ANP, BNP |
| [95] | Ramoji Kosuru | Pterostilbene | In vivo | Upregulate SOD, CAT, GSH, GPX, PGC-1α, Nrf2, HO-1, AMPK Downregulate IL-1β, IL-6, TNF-α, NF-κB, TLR4, NLRP3 |
| [96] | Cuihua Zhao | Fortunellin | In vivo | Upregulate SOD, Nrf2, HO-1, AMPK Downregulate TNF-α, IL-1β, IL-6, IL-18, NF-κB, Keap1 |
| [97] | J Z Altamimi | Ellagic acid | In vivo | Upregulate GSH, SOD, Nrf2, SIRT1 Downregulate BNP, TNF-α, IL-6 |
| [100] | Yiqiu Cao | Z-ligustilide | In vitro | Upregulate AMPK, Nrf2, SOD Downregulate GSK3β |
| [113] | Lijiao Xu | Scutellarin | In vivo | Upregulate Nrf2, HO-1, Akt |
| [110] | Sai Ma | Resveratrol | In vivo | Upregulate Nrf2, SIRT1 Downregulate ANP, BNP |
| [114] | Hongyang Liu | Spiraeoside | In vitro | Upregulate Akt, Nrf2, HO-1, Bcl2, SOD, GPX, CAT Downregulate caspase3, caspase7, Bax |
| [117] | Bin Zhang | Myricitrin | In vitro | Upregulate Nrf2, HO-1, γ-GCS, NQO1, Akt Downregulate GSK3β |
| [116] | Jialin Duan | Butin | In vivo | Upregulate SOD, Nrf2, HO-1, Akt Downregulate Keap1, GSK3β, Fyn |
| [118] | Xuemei Gu | Isoliquiritigenin | In vitro | Upregulate Nrf2, HO-1 Downregulate TNF-α, IL-6, IL-1β, VCAM-1, MCP-1, JNK, ERK, p38 |
| [124] | Qitong Lu | Hinokinin | In vivo | Upregulate Nrf2, HO-1, SOD Downregulate Keap1, JNK1, ERK1/2, p38 |
| [125] | Jing Ni | Salidroside | In vitro | Upregulate Nrf2, HO-1 Downregulate ERK, JNK, p38 |
| [133] | Hai-Han Liao | Myricetin | In vivo | Upregulate Nrf2, HO-1, NQO1, SOD Downregulate collagen I, collagen III, fibronectin, CTGF, Smad3, TGF-β |
| [134] | Wenshuai Ma | Bakuchiol | In vitro | Upregulate Nrf2, SOD, GPX Downregulate collagen I, collagen III, α-SMA, TGF-β, Smad3 |
| [136] | Yin Ying | Phloretin | In vivo | Upregulate Nrf2, HO-1, NQO1 Downregulate TGF-β, collagen I, CTGF |
| [138] | A S Alshehri | Kaempferol | In vivo | Upregulate Nrf2, GSH, Bcl2 Downregulate TGF-β1, Bax |
| [40] | Bin Zhang | Notoginsenoside R1 | In vivo | Upregulate Nrf2, HO-1, γ-GCS, NQO1, Smurf2 Downregulate TGF-β, collagen I, Bax/Bc12, caspase-3, caspase-9, Smad2/3 |
| [142] | Guangru Li | Syringaresinol | In vivo | Upregulate Nrf2, NQO1, HO-1, SOD Downregulate TGF-β, fibronectin, α-SMA, Smad2/3, Bax/Bc12, Keap1 |
3. Conclusions
The increasing morbidity and lethality of DCM related to poor prognosis and survival in patients with DM call for multiple measures to prevent it. The mechanisms behind the pathogenesis of DCM are highly complex, but the overlapping progression and many signaling pathways are involved in it. What is highlighted is that oxidative stress is one of the central mechanisms in the pathogenesis of DCM. Nrf2 signaling pathway is essential to counterbalance oxidative stress via crosstalk with other signaling pathways. Surprisingly, increasing studies indicate that Chinese herbal monomers attenuate DCM in different aspects at the same time via regulating Nrf2. However, the molecular mechanisms behind the crosstalk between Nrf2 and these pathways need to be explored furthermore deeply. The phenomenon of multiple targets regulation based on Chinese herbal monomers is looking forward to having more detailed and precise experiment results.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant nos. 81973801).
Conflicts of Interest
The author declares that there is no conflict of interest regarding the publication of this paper.
References
- 1.Jia G., Hill M. A., Sowers J. R. Diabetic cardiomyopathy: an update of mechanisms contributing to this clinical entity. Circulation Research . 2018;122(4):624–638. doi: 10.1161/CIRCRESAHA.117.311586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fox C. S., Coady S., Sorlie P. D., et al. Increasing cardiovascular disease burden due to diabetes mellitus: the Framingham heart study. Circulation . 2007;115(12):1544–1550. doi: 10.1161/CIRCULATIONAHA.106.658948. [DOI] [PubMed] [Google Scholar]
- 3.Aronow W. S., Chul A. Incidence of heart failure in 2,737 older persons with and without diabetes mellitus. Chest . 1999;115(3):867–868. doi: 10.1378/CHEST.115.3.867. [DOI] [PubMed] [Google Scholar]
- 4.Jankauskas S. S., et al. Heart failure in diabetes. Metabolism . 2021;125, article 154910 doi: 10.1016/J.METABOL.2021.154910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Roberts A. W., Clark A. L., Witte K. K. Review article: left ventricular dysfunction and heart failure in metabolic syndrome and diabetes without overt coronary artery disease—do we need to screen our patients? Diabetes & Vascular Disease Research . 2009;6(3):153–163. doi: 10.1177/1479164109338774. [DOI] [PubMed] [Google Scholar]
- 6.Cai X., Liu X., Sun L., et al. Prediabetes and the risk of heart failure: a meta-analysis. Diabetes, Obesity & Metabolism . 2021;23(8):1746–1753. doi: 10.1111/DOM.14388. [DOI] [PubMed] [Google Scholar]
- 7.Mai L., Wen W., Qiu M., et al. Association between prediabetes and adverse outcomes in heart failure. Diabetes, Obesity and Metabolism . 2021;23(11) doi: 10.1111/DOM.14490. [DOI] [PubMed] [Google Scholar]
- 8.Li R., Liu Y., Shan Y.-g., Gao L., Wang F., Qiu C.-g. Bailcalin protects against diabetic cardiomyopathy through Keap1/Nrf2/AMPK-mediated antioxidative and lipid-lowering effects. Oxidative Medicine and Cellular Longevity . 2019;2019 doi: 10.1155/2019/3206542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen Q. M., Maltagliati A. J. Nrf2 at the heart of oxidative stress and cardiac protection. Physiological Genomics . 2018;50(2):77–97. doi: 10.1152/PHYSIOLGENOMICS.00041.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bellezza I., Giambanco I., Minelli A., Donato R. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim Biophys Acta Mol Cell Res . 2018;1865(5):721–733. doi: 10.1016/J.BBAMCR.2018.02.010. [DOI] [PubMed] [Google Scholar]
- 11.Suzuki T., Yamamoto M. Molecular basis of the Keap1-Nrf2 system. Free Radical Biology & Medicine . 2015;88:93–100. doi: 10.1016/J.FREERADBIOMED.2015.06.006. [DOI] [PubMed] [Google Scholar]
- 12.Jia G., Whaley-Connell A., Sowers J. R. Diabetic cardiomyopathy: a hyperglycaemia- and insulin-resistance-induced heart disease. Diabetologia . 2018;61(1):21–28. doi: 10.1007/S00125-017-4390-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jia G., DeMarco V. G., Sowers J. R. Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nature Reviews. Endocrinology . 2016;12(3):144–153. doi: 10.1038/NRENDO.2015.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Giacco F., Brownlee M. Oxidative stress and diabetic complications. Circulation Research . 2010;107(9):1058–1070. doi: 10.1161/CIRCRESAHA.110.223545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zheng H., Zhu H., Liu X., Huang X., Huang A., Huang Y. Mitophagy in diabetic cardiomyopathy: roles and mechanisms. Frontiers in Cell and Development Biology . 2021;9 doi: 10.3389/FCELL.2021.750382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tao J., Chen H., Wang Y. J., et al. Ketogenic diet suppressed T-regulatory cells and promoted cardiac fibrosis via reducing mitochondria-associated membranes and inhibiting mitochondrial function. Oxidative Medicine and Cellular Longevity . 2021;2021:15. doi: 10.1155/2021/5512322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fleisher T. A. Apoptosis. Annals of Allergy, Asthma & Immunology . 1997;78(3):245–250. doi: 10.1016/S1081-1206(10)63176-6. [DOI] [PubMed] [Google Scholar]
- 18.Krijnen P. A. J., Simsek S., Niessen H. W. M. Apoptosis in diabetes. Apoptosis . 2009;14(12):1387–1388. doi: 10.1007/S10495-009-0419-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Newsholme P., Cruzat V. F., Keane K. N., Carlessi R., de Bittencourt P. I. H. Molecular mechanisms of ROS production and oxidative stress in diabetes. The Biochemical Journal . 2016;473(24):4527–4550. doi: 10.1042/BCJ20160503C. [DOI] [PubMed] [Google Scholar]
- 20.Bugger H., Abel E. D. Molecular mechanisms of diabetic cardiomyopathy. Diabetologia . 2014;57(4):660–671. doi: 10.1007/S00125-014-3171-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nielsen L. F., Moe D., Kirkeby S., Garbarsch C. Sirius red and acid fuchsin staining mechanisms. Biotechnic & Histochemistry . 1998;73(2):71–77. doi: 10.3109/10520299809140509. [DOI] [PubMed] [Google Scholar]
- 22.Russo I., Frangogiannis N. G. Diabetes-associated cardiac fibrosis: cellular effectors, molecular mechanisms and therapeutic opportunities. Journal of Molecular and Cellular Cardiology . 2016;90:84–93. doi: 10.1016/J.YJMCC.2015.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Oldfield M. D., Bach L. A., Forbes J. M., et al. Advanced glycation end products cause epithelial-myofibroblast transdifferentiation via the receptor for advanced glycation end products (RAGE) The Journal of Clinical Investigation . 2001;108(12):1853–1863. doi: 10.1172/JCI11951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nauck M., Smith U. Incretin-based therapy: how do incretin mimetics and DPP-4 inhibitors fit into treatment algorithms for type 2 diabetic patients? Best Practice & Research. Clinical Endocrinology & Metabolism . 2009;23(4):513–523. doi: 10.1016/J.BEEM.2009.03.002. [DOI] [PubMed] [Google Scholar]
- 25.Newsholme P., Haber E. P., Hirabara S. M., et al. Diabetes associated cell stress and dysfunction: role of mitochondrial and non-mitochondrial ROS production and activity. The Journal of Physiology . 2007;583:9–24. doi: 10.1113/JPHYSIOL.2007.135871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meares G. P., Fontanilla D., Broniowska K. A., Andreone T., Lancaster J. R., Corbett J. A. Differential responses of pancreatic β-cells to ROS and RNS. Am J Physiol Endocrinol Metab . 2013;304(6) doi: 10.1152/AJPENDO.00424.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hansen S. S., Aasum E., Hafstad A. D. The role of NADPH oxidases in diabetic cardiomyopathy. Biochim Biophys Acta Mol Basis Dis . 2018;1864(5):1908–1913. doi: 10.1016/J.BBADIS.2017.07.025. [DOI] [PubMed] [Google Scholar]
- 28.Krause M. S., Mcclenaghan N. H., Flatt P. R., de Bittencourt P. I. H., Murphy C., Newsholme P. L-arginine is essential for pancreatic β-cell functional integrity, metabolism and defense from inflammatory challenge. The Journal of Endocrinology . 2011;211(1):87–97. doi: 10.1530/JOE-11-0236. [DOI] [PubMed] [Google Scholar]
- 29.Cianflone E., Torella M., Biamonte F., et al. Targeting cardiac stem cell senescence to treat cardiac aging and disease. Cells . 2020;9(6) doi: 10.3390/CELLS9061558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ho E., Karimi Galougahi K., Liu C. C., Bhindi R., Figtree G. A. Biological markers of oxidative stress: applications to cardiovascular research and practice. Redox Biology . 2013;1(1):483–491. doi: 10.1016/J.REDOX.2013.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhu W., Wu R. D., Lv Y. G., Liu Y. M., Huang H., Xu J. Q. BRD4 blockage alleviates pathological cardiac hypertrophy through the suppression of fibrosis and inflammation via reducing ROS generation. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie . 2020;121 doi: 10.1016/J.BIOPHA.2019.109368. [DOI] [PubMed] [Google Scholar]
- 32.Mu J., Zhang D., Tian Y., Xie Z., Zou M. H. BRD4 inhibition by JQ1 prevents high-fat diet-induced diabetic cardiomyopathy by activating PINK1/Parkin-mediated mitophagy in vivo. Journal of Molecular and Cellular Cardiology . 2020;149:1–14. doi: 10.1016/J.YJMCC.2020.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Clementi E., Nisoli E. Nitric oxide and mitochondrial biogenesis: a key to long-term regulation of cellular metabolism. Comparative Biochemistry and Physiology. Part A, Molecular & Integrative Physiology . 2005;142(2):102–110. doi: 10.1016/J.CBPB.2005.04.022. [DOI] [PubMed] [Google Scholar]
- 34.Rochette L., Lorin J., Zeller M., et al. Nitric oxide synthase inhibition and oxidative stress in cardiovascular diseases: possible therapeutic targets? Pharmacology & Therapeutics . 2013;140(3):239–257. doi: 10.1016/J.PHARMTHERA.2013.07.004. [DOI] [PubMed] [Google Scholar]
- 35.Knapp M., Tu X., Wu R. Vascular endothelial dysfunction, a major mediator in diabetic cardiomyopathy. Acta Pharmacol Sin . 2019;40(1) doi: 10.1038/S41401-018-0042-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kim J. A., Montagnani M., Kwang K. K., Quon M. J. Reciprocal relationships between insulin resistance and endothelial dysfunction: molecular and pathophysiological mechanisms. Circulation . 2006;113(15):1888–1904. doi: 10.1161/CIRCULATIONAHA.105.563213. [DOI] [PubMed] [Google Scholar]
- 37.Kim J. A., Jang H. J., Martinez-Lemus L. A., Sowers J. R. Activation of mTOR/p70S6 kinase by ANG II inhibits insulin-stimulated endothelial nitric oxide synthase and vasodilation. Am J Physiol Endocrinol Metab . 2012;302(2) doi: 10.1152/AJPENDO.00497.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Itoh K., Ishii T., Wakabayashi N., Yamamoto M. Regulatory mechanisms of cellular response to oxidative stress. Free Radical Research . 1999;31(4):319–324. doi: 10.1080/10715769900300881. [DOI] [PubMed] [Google Scholar]
- 39.Sajadimajd S., Khazaei M. Oxidative stress and cancer: the role of Nrf2. Current Cancer Drug Targets . 2018;18(6):538–557. doi: 10.2174/1568009617666171002144228. [DOI] [PubMed] [Google Scholar]
- 40.Kaspar J. W., Niture S. K., Jaiswal A. K. Nrf2:INrf2 (Keap1) signaling in oxidative stress. Free Radical Biology & Medicine . 2009;47(9):1304–1309. doi: 10.1016/J.FREERADBIOMED.2009.07.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Parim B., Sathibabu Uddandrao V. V., Saravanan G. Diabetic cardiomyopathy: molecular mechanisms, detrimental effects of conventional treatment, and beneficial effects of natural therapy. Heart Failure Reviews . 2019;24(2):279–299. doi: 10.1007/S10741-018-9749-1. [DOI] [PubMed] [Google Scholar]
- 42.Chen J., Zhang Z., Cai L. Diabetic cardiomyopathy and its prevention by nrf2: current status. Diabetes and Metabolism Journal . 2014;38(5):337–345. doi: 10.4093/DMJ.2014.38.5.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Abdelsamia E. M., Khaleel S. A., Balah A., Abdel Baky N. A. Curcumin augments the cardioprotective effect of metformin in an experimental model of type I diabetes mellitus: impact of Nrf2/HO-1 and JAK/STAT pathways. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie . 2019;109:2136–2144. doi: 10.1016/J.BIOPHA.2018.11.064. [DOI] [PubMed] [Google Scholar]
- 44.Atta M. S., El-Far A. H., Farrag F. A., Abdel-Daim M. M., Al Jaouni S. K., Mousa S. A. Thymoquinone attenuates cardiomyopathy in Streptozotocin-treated diabetic rats. Oxidative Medicine and Cellular Longevity . 2018;2018:10. doi: 10.1155/2018/7845681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Raish M., Ahmad A., Jardan Y. A. B., et al. Sinapic acid ameliorates cardiac dysfunction and cardiomyopathy by modulating NF-κB and Nrf2/HO-1 signaling pathways in streptozocin induced diabetic rats. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie . 2021;145, article 112412 doi: 10.1016/J.BIOPHA.2021.112412. [DOI] [PubMed] [Google Scholar]
- 46.Luo J., Yan D., Li S., et al. Allopurinol reduces oxidative stress and activates Nrf2/p62 to attenuate diabetic cardiomyopathy in rats. Journal of Cellular and Molecular Medicine . 2020;24(2):1760–1773. doi: 10.1111/JCMM.14870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang G., Song X., Zhao L., Li Z., Liu B. Resveratrol prevents diabetic cardiomyopathy by increasing Nrf2 expression and transcriptional activity. BioMed Research International . 2018;2018:13. doi: 10.1155/2018/2150218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dong Z., Bian L., Wang Y. L., Sun L. M. Gastrodin protects against high glucose-induced cardiomyocyte toxicity via GSK-3β-mediated nuclear translocation of Nrf2. Human & Experimental Toxicology . 2021;40(9):1584–1597. doi: 10.1177/09603271211002885. [DOI] [PubMed] [Google Scholar]
- 49.Duan J., Wei G., Guo C., et al. Aralia taibaiensis protects cardiac myocytes against high glucose-induced oxidative stress and apoptosis. The American Journal of Chinese Medicine . 2015;43(6):1159–1175. doi: 10.1142/S0192415X15500664. [DOI] [PubMed] [Google Scholar]
- 50.Sathibabu Uddandrao V. V., Brahmanaidu P., Nivedha P. R., Vadivukkarasi S., Saravanan G. Beneficial role of some natural products to attenuate the diabetic cardiomyopathy through Nrf2 pathway in cell culture and animal models. Cardiovascular Toxicology . 2018;18(3):199–205. doi: 10.1007/S12012-017-9430-2. [DOI] [PubMed] [Google Scholar]
- 51.Castillo R. L., Herrera E. A., Gonzalez-Candia A., et al. Quercetin prevents diastolic dysfunction induced by a high-cholesterol diet: role of oxidative stress and bioenergetics in hyperglycemic rats. Oxidative Medicine and Cellular Longevity . 2018;2018:14. doi: 10.1155/2018/7239123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cantó C., Auwerx J. PGC-1alpha, SIRT1 and AMPK, an energy sensing network that controls energy expenditure. Current Opinion in Lipidology . 2009;20(2):98–105. doi: 10.1097/MOL.0B013E328328D0A4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hayden M. S., Ghosh S. NF-κB in immunobiology. Cell Research . 2011;21(2):223–244. doi: 10.1038/CR.2011.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ghosh S., Hayden M. S. New regulators of NF-kappaB in inflammation. Nature Reviews. Immunology . 2008;8(11):837–848. doi: 10.1038/NRI2423. [DOI] [PubMed] [Google Scholar]
- 55.Patel S., Santani D. Role of NF-kappa B in the pathogenesis of diabetes and its associated complications. Pharmacological Reports . 2009;61(4):595–603. doi: 10.1016/S1734-1140(09)70111-2. [DOI] [PubMed] [Google Scholar]
- 56.Gilmore T. D. Introduction to NF-kappaB: players, pathways, perspectives. Oncogene . 2006;25(51):6680–6684. doi: 10.1038/SJ.ONC.1209954. [DOI] [PubMed] [Google Scholar]
- 57.Kawagoe T., Sato S., Matsushita K., et al. Sequential control of toll-like receptor-dependent responses by IRAK1 and IRAK2. Nature Immunology . 2008;9(6):684–691. doi: 10.1038/NI.1606. [DOI] [PubMed] [Google Scholar]
- 58.Chiu J., Farhangkhoee H., Xu B. Y., Chen S., George B., Chakrabarti S. PARP mediates structural alterations in diabetic cardiomyopathy. Journal of Molecular and Cellular Cardiology . 2008;45(3):385–393. doi: 10.1016/J.YJMCC.2008.06.009. [DOI] [PubMed] [Google Scholar]
- 59.Hassa P. O., Haenni S. S., Buerki C., et al. Acetylation of poly(ADP-ribose) polymerase-1 by p300/CREB-binding protein regulates coactivation of NF-kappaB-dependent transcription. The Journal of Biological Chemistry . 2005;280(49):40450–40464. doi: 10.1074/JBC.M507553200. [DOI] [PubMed] [Google Scholar]
- 60.Goulopoulou S., McCarthy C. G., Clinton Webb R. Toll-like receptors in the vascular system: sensing the dangers within. Pharmacological Reviews . 2016;68(1):142–167. doi: 10.1124/PR.114.010090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Park H. S., Jung H. Y., Park E. Y., Kim J., Lee W. J., Bae Y. S. Cutting edge: direct interaction of TLR4 with NAD(P)H oxidase 4 isozyme is essential for lipopolysaccharide-induced production of reactive oxygen species and activation of NF-kappa B. Journal of Immunology . 2004;173(6):3589–3593. doi: 10.4049/JIMMUNOL.173.6.3589. [DOI] [PubMed] [Google Scholar]
- 62.Strowig T., Henao-Mejia J., Elinav E., Flavell R. Inflammasomes in health and disease. Nature . 2012;481(7381):278–286. doi: 10.1038/NATURE10759. [DOI] [PubMed] [Google Scholar]
- 63.Zhong L., Simard M. J., Huot J. Endothelial microRNAs regulating the NF-κB pathway and cell adhesion molecules during inflammation. The FASEB Journal . 2018;32(8):4070–4084. doi: 10.1096/FJ.201701536R. [DOI] [PubMed] [Google Scholar]
- 64.Deshmane S. L., Kremlev S., Amini S., Sawaya B. E. Monocyte chemoattractant protein-1 (MCP-1): an overview. Journal of Interferon & Cytokine Research . 2009;29(6):313–325. doi: 10.1089/JIR.2008.0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Suryavanshi S. V., Kulkarni Y. A. NF-κβ: a potential target in the management of vascular complications of diabetes. Front Pharmacol . 2017;8 doi: 10.3389/FPHAR.2017.00798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Dyson A., Bryan N. S., Fernandez B. O., et al. An integrated approach to assessing nitroso-redox balance in systemic inflammation. Free Radical Biology & Medicine . 2011;51(6):1137–1145. doi: 10.1016/J.FREERADBIOMED.2011.06.012. [DOI] [PubMed] [Google Scholar]
- 67.Wang H., Khor T. O., Saw C. L. L., et al. Role of Nrf2 in suppressing LPS-induced inflammation in mouse peritoneal macrophages by polyunsaturated fatty acids docosahexaenoic acid and eicosapentaenoic acid. Molecular Pharmaceutics . 2010;7(6):2185–2193. doi: 10.1021/MP100199M. [DOI] [PubMed] [Google Scholar]
- 68.Ahmed S. M. U., Luo L., Namani A., Wang X. J., Tang X. Nrf2 signaling pathway: pivotal roles in inflammation. Biochimica et Biophysica Acta - Molecular Basis of Disease . 2017;1863(2):585–597. doi: 10.1016/J.BBADIS.2016.11.005. [DOI] [PubMed] [Google Scholar]
- 69.Lian Y., Xia X., Zhao H., Zhu Y. The potential of chrysophanol in protecting against high fat-induced cardiac injury through Nrf2-regulated anti-inflammation, anti-oxidant and anti-fibrosis in Nrf2 knockout mice. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie . 2017;93:1175–1189. doi: 10.1016/J.BIOPHA.2017.05.148. [DOI] [PubMed] [Google Scholar]
- 70.Chen X., Qian J., Wang L., et al. Kaempferol attenuates hyperglycemia-induced cardiac injuries by inhibiting inflammatory responses and oxidative stress. Endocrine . 2018;60(1):83–94. doi: 10.1007/S12020-018-1525-4. [DOI] [PubMed] [Google Scholar]
- 71.Liang E., Liu X., Du Z., Yang R., Zhao Y. Andrographolide ameliorates diabetic cardiomyopathy in mice by blockage of oxidative damage and NF-κB-mediated inflammation. Oxidative Medicine and Cellular Longevity . 2018;2018 doi: 10.1155/2018/9086747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Li H., Shi Y., Wang X., et al. Piceatannol alleviates inflammation and oxidative stress via modulation of the Nrf2/HO-1 and NF-κB pathways in diabetic cardiomyopathy. Chemico-Biological Interactions . 2019;310 doi: 10.1016/J.CBI.2019.108754. [DOI] [PubMed] [Google Scholar]
- 73.Thimmulappa R. K., Scollick C., Traore K., et al. Nrf2-dependent protection from LPS induced inflammatory response and mortality by CDDO-Imidazolide. Biochemical and Biophysical Research Communications . 2006;351(4):883–889. doi: 10.1016/J.BBRC.2006.10.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Huo Y., Mijiti A., Cai R., et al. Scutellarin alleviates type 2 diabetes (HFD/low dose STZ)-induced cardiac injury through modulation of oxidative stress, inflammation, apoptosis and fibrosis in mice. Human & Experimental Toxicology . 2021;40 doi: 10.1177/09603271211045948. [DOI] [PubMed] [Google Scholar]
- 75.Xu Z., Kong X. Q. Bixin ameliorates high fat diet-induced cardiac injury in mice through inflammation and oxidative stress suppression. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie . 2017;89:991–1004. doi: 10.1016/J.BIOPHA.2017.02.052. [DOI] [PubMed] [Google Scholar]
- 76.Banning A., Brigelius-Flohé R. NF-kappaB, Nrf2, and HO-1 interplay in redox-regulated VCAM-1 expression. Antioxidants & Redox Signaling . 2005;7(7–8):889–899. doi: 10.1089/ARS.2005.7.889. [DOI] [PubMed] [Google Scholar]
- 77.Li L., Luo W., Qian Y., et al. Luteolin protects against diabetic cardiomyopathy by inhibiting NF-κB-mediated inflammation and activating the Nrf2-mediated antioxidant responses. Phytomedicine . 2019;59 doi: 10.1016/J.PHYMED.2018.11.034. [DOI] [PubMed] [Google Scholar]
- 78.Liu X., Zhang X., Ding Y., et al. Nuclear factor E2-related Factor-2 negatively regulates NLRP3 Inflammasome activity by inhibiting reactive oxygen species-induced NLRP3 priming. Antioxidants & Redox Signaling . 2017;26(1):28–43. doi: 10.1089/ARS.2015.6615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lee D. F., Kuo H. P., Liu M., et al. KEAP1 E3 ligase-mediated downregulation of NF-kappaB signaling by targeting IKKbeta. Molecular Cell . 2009;36(1):131–140. doi: 10.1016/J.MOLCEL.2009.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Brigelius-Flohé R., Flohé L. Basic principles and emerging concepts in the redox control of transcription factors. Antioxidants & Redox Signaling . 2011;15(8):2335–2381. doi: 10.1089/ARS.2010.3534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hardie D. G. The AMP-activated protein kinase pathway--new players upstream and downstream. Journal of Cell Science . 2004;117(23):5479–5487. doi: 10.1242/JCS.01540. [DOI] [PubMed] [Google Scholar]
- 82.Balteau M., Van Steenbergen A., Timmermans A. D., et al. AMPK activation by glucagon-like peptide-1 prevents NADPH oxidase activation induced by hyperglycemia in adult cardiomyocytes. American Journal of Physiology. Heart and Circulatory Physiology . 2014;307(8):H1120–H1133. doi: 10.1152/AJPHEART.00210.2014. [DOI] [PubMed] [Google Scholar]
- 83.Kim T. T., Dyck J. R. B. Is AMPK the savior of the failing heart? Trends in Endocrinology and Metabolism . 2015;26(1):40–48. doi: 10.1016/J.TEM.2014.11.001. [DOI] [PubMed] [Google Scholar]
- 84.Kudo N., Barr A. J., Barr R. L., Desai S., Lopaschuk G. D. High rates of fatty acid oxidation during reperfusion of ischemic hearts are associated with a decrease in malonyl-CoA levels due to an increase in 5’-AMP-activated protein kinase inhibition of acetyl-CoA carboxylase. The Journal of Biological Chemistry . 1995;270(29):17513–17520. doi: 10.1074/JBC.270.29.17513. [DOI] [PubMed] [Google Scholar]
- 85.Fillmore N., Mori J., Lopaschuk G. D. Mitochondrial fatty acid oxidation alterations in heart failure, ischaemic heart disease and diabetic cardiomyopathy. British Journal of Pharmacology . 2014;171(8):2080–2090. doi: 10.1111/BPH.12475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Park S. H., Gammon S. R., Knippers J. D., Paulsen S. R., Rubink D. S., Winder W. W. Phosphorylation-activity relationships of AMPK and acetyl-CoA carboxylase in muscle. Journal of Applied Physiology (Bethesda, MD: 1985) . 2002;92(6):2475–2482. doi: 10.1152/JAPPLPHYSIOL.00071.2002. [DOI] [PubMed] [Google Scholar]
- 87.Herzig S., Shaw R. J. AMPK: guardian of metabolism and mitochondrial homeostasis. Nature Reviews. Molecular Cell Biology . 2018;19(2):121–135. doi: 10.1038/NRM.2017.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zitka O., Skalickova S., Gumulec J., et al. Redox status expressed as GSH:GSSG ratio as a marker for oxidative stress in paediatric tumour patients. Oncology Letters . 2012;4(6):1247–1253. doi: 10.3892/OL.2012.931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Weber M., Hamm C. Role of B-type natriuretic peptide (BNP) and NT-proBNP in clinical routine. Heart . 2006;92(6):843–849. doi: 10.1136/HRT.2005.071233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Charloux A., Piquard F., Doutreleau S., Brandenberger G., Geny B. Mechanisms of renal hyporesponsiveness to ANP in heart failure. European Journal of Clinical Investigation . 2003;33(9):769–778. doi: 10.1046/J.1365-2362.2003.01222.X. [DOI] [PubMed] [Google Scholar]
- 91.Reda S. M., Gollapudi S. K., Chandra M. Developmental increase in β-MHC enhances sarcomere length-dependent activation in the myocardium. The Journal of General Physiology . 2019;151(5):635–644. doi: 10.1085/JGP.201812183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Ba L., Gao J., Chen Y., et al. Allicin attenuates pathological cardiac hypertrophy by inhibiting autophagy via activation of PI3K/Akt/mTOR and MAPK/ERK/mTOR signaling pathways. Phytomedicine . 2019;58 doi: 10.1016/J.PHYMED.2018.11.025. [DOI] [PubMed] [Google Scholar]
- 93.Du F., Huang H., Cao Y., Ran Y., Wu Q., Chen B. Notoginsenoside R1 protects against high glucose-induced cell injury through AMPK/Nrf2 and downstream HO-1 signaling. Frontiers in Cell and Development Biology . 2021;9 doi: 10.3389/FCELL.2021.791643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Mo C., Wang L., Zhang J., et al. The crosstalk between Nrf2 and AMPK signal pathways is important for the anti-inflammatory effect of berberine in LPS-stimulated macrophages and endotoxin-shocked mice. Antioxidants & Redox Signaling . 2014;20(4):574–588. doi: 10.1089/ARS.2012.5116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kosuru R., Kandula V., Rai U., Prakash S., Xia Z., Singh S. Pterostilbene decreases cardiac oxidative stress and inflammation via activation of AMPK/Nrf2/HO-1 pathway in fructose-fed diabetic rats. Cardiovascular Drugs and Therapy . 2018;32(2):147–163. doi: 10.1007/S10557-018-6780-3. [DOI] [PubMed] [Google Scholar]
- 96.Zhao C., Zhang Y., Liu H., Li P., Zhang H., Cheng G. Fortunellin protects against high fructose-induced diabetic heart injury in mice by suppressing inflammation and oxidative stress via AMPK/Nrf-2 pathway regulation. Biochemical and Biophysical Research Communications . 2017;490(2):552–559. doi: 10.1016/J.BBRC.2017.06.076. [DOI] [PubMed] [Google Scholar]
- 97.Altamimi J. Z., Alfaris N. A., Alshammari G. M., et al. Ellagic acid protects against diabetic cardiomyopathy in rats by stimulating cardiac silent information regulator 1 signaling. Journal of Physiology and Pharmacology . 2020;71(6):891–904. doi: 10.26402/JPP.2020.6.12. [DOI] [PubMed] [Google Scholar]
- 98.Joo M. S., Kim W. D., Lee K. Y., Kim J. H., Koo J. H., Kim S. G. AMPK facilitates nuclear accumulation of Nrf2 by phosphorylating at serine 550. Molecular and Cellular Biology . 2016;36(14):1931–1942. doi: 10.1128/MCB.00118-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhai P., Sciarretta S., Galeotti J., Volpe M., Sadoshima J. Differential roles of GSK-3β during myocardial ischemia and ischemia/reperfusion. Circulation Research . 2011;109(5):502–511. doi: 10.1161/CIRCRESAHA.111.249532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cao Y., Dong Z., Yang D., Ma X., Wang X. Alleviation of glucolipotoxicity-incurred cardiomyocyte dysfunction by Z-ligustilide involves in the suppression of oxidative insult, inflammation and fibrosis. Chemistry and Physics of Lipids . 2021;241 doi: 10.1016/J.CHEMPHYSLIP.2021.105138. [DOI] [PubMed] [Google Scholar]
- 101.Wang Z., Chen Z., Jiang Z., et al. Cordycepin prevents radiation ulcer by inhibiting cell senescence via NRF2 and AMPK in rodents. Nature Communications . 2019;10(1) doi: 10.1038/S41467-019-10386-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Nagappan A., Kim J. H., Jung D. Y., Jung M. H. Cryptotanshinone from the Salvia miltiorrhiza bunge attenuates ethanol-induced liver injury by activation of AMPK/SIRT1 and Nrf2 signaling pathways. Int J Mol Sci . 2019;21(1) doi: 10.3390/IJMS21010265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wu J., Zheng H., Liu X., et al. Prognostic value of secreted frizzled-related protein 5 in heart failure patients with and without type 2 diabetes mellitus. Circulation. Heart Failure . 2020;13(9):393–403. doi: 10.1161/CIRCHEARTFAILURE.120.007054. [DOI] [PubMed] [Google Scholar]
- 104.Wu Y. U., Liu X., Zheng H., et al. Multiple roles of sFRP2 in cardiac development and cardiovascular disease. International Journal of Biological Sciences . 2020;16(5):730–738. doi: 10.7150/IJBS.40923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Huang A., Huang Y. Role of Sfrps in cardiovascular disease. Ther Adv Chronic Dis . 2020;11 doi: 10.1177/2040622320901990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Yang S., Chen H., Tan K., et al. Secreted frizzled-related protein 2 and extracellular volume fraction in patients with heart failure. Oxidative Medicine and Cellular Longevity . 2020;2020:9. doi: 10.1155/2020/2563508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lin M., Liu X., Zheng H., et al. IGF-1 enhances BMSC viability, migration, and anti-apoptosis in myocardial infarction via secreted frizzled-related protein 2 pathway. Stem Cell Res Ther . 2020;11(1) doi: 10.1186/S13287-019-1544-Y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ma T., Huang X., Zheng H., et al. SFRP2 improves mitochondrial dynamics and mitochondrial biogenesis, oxidative stress, and apoptosis in diabetic cardiomyopathy. Oxidative Medicine and Cellular Longevity . 2021;2021:9. doi: 10.1155/2021/9265016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Tan Y., Ichikawa T., Li J., et al. Diabetic downregulation of Nrf2 activity via ERK contributes to oxidative stress-induced insulin resistance in cardiac cells in vitro and in vivo. Diabetes . 2011;60(2):625–633. doi: 10.2337/DB10-1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Ma S., Feng J., Zhang R., et al. SIRT1 activation by resveratrol alleviates cardiac dysfunction via mitochondrial regulation in diabetic cardiomyopathy mice. Oxidative Medicine and Cellular Longevity . 2017;2017:15. doi: 10.1155/2017/4602715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Lv Y., Jiang H., Li S., et al. Sulforaphane prevents chromium-induced lung injury in rats via activation of the Akt/GSK-3β/Fyn pathway. Environmental Pollution . 2020;259 doi: 10.1016/J.ENVPOL.2019.113812. [DOI] [PubMed] [Google Scholar]
- 112.Wang X., Pan J., Liu D., et al. Nicorandil alleviates apoptosis in diabetic cardiomyopathy through PI3K/Akt pathway. Journal of Cellular and Molecular Medicine . 2019;23(8):5349–5359. doi: 10.1111/JCMM.14413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Xu L., Chen R., Zhang X., et al. Scutellarin protects against diabetic cardiomyopathy via inhibiting oxidative stress and inflammatory response in mice. Ann Palliat Med . 2021;10(3):2481–2493. doi: 10.21037/APM-19-516. [DOI] [PubMed] [Google Scholar]
- 114.Liu H., Zhang Z., Zhang L., et al. Spiraeoside protects human cardiomyocytes against high glucose-induced injury, oxidative stress, and apoptosis by activation of PI3K/Akt/Nrf2 pathway. Journal of Biochemical and Molecular Toxicology . 2020;34(10) doi: 10.1002/JBT.22548. [DOI] [PubMed] [Google Scholar]
- 115.Ishii T., Warabi E. Mechanism of rapid nuclear factor-E2-related factor 2 (Nrf2) activation via membrane-associated estrogen receptors: roles of NADPH oxidase 1, neutral sphingomyelinase 2 and epidermal growth factor receptor (EGFR) Antioxidants (Basel) . 2019;8(3) doi: 10.3390/ANTIOX8030069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Duan J., Guan Y., Mu F., et al. Protective effect of butin against ischemia/reperfusion-induced myocardial injury in diabetic mice: involvement of the AMPK/GSK-3β/Nrf2 signaling pathway. Scientific Reports . 2017;7 doi: 10.1038/SREP41491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zhang B., Shen Q., Chen Y., et al. Myricitrin alleviates oxidative stress-induced inflammation and apoptosis and protects mice against diabetic cardiomyopathy. Scientific Reports . 2017;7 doi: 10.1038/SREP44239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Gu X., Shi Y., Chen X., et al. Isoliquiritigenin attenuates diabetic cardiomyopathy via inhibition of hyperglycemia-induced inflammatory response and oxidative stress. Phytomedicine . 2020;78 doi: 10.1016/J.PHYMED.2020.153319. [DOI] [PubMed] [Google Scholar]
- 119.Ma X., Chen Z., Wang L., et al. The pathogenesis of diabetes mellitus by oxidative stress and inflammation: its inhibition by berberine. Frontiers in Pharmacology . 2018;9 doi: 10.3389/FPHAR.2018.00782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Zhang Y., Cui Y., Dai S., et al. Isorhynchophylline enhances Nrf2 and inhibits MAPK pathway in cardiac hypertrophy. Naunyn-Schmiedeberg's Archives of Pharmacology . 2020;393(2):203–212. doi: 10.1007/S00210-019-01716-0. [DOI] [PubMed] [Google Scholar]
- 121.Liao H.-h., Zhang N., Meng Y.-y., et al. Myricetin alleviates pathological cardiac hypertrophy via TRAF6/TAK1/MAPK and Nrf2 signaling pathway. Oxidative Medicine and Cellular Longevity . 2019;2019:14. doi: 10.1155/2019/6304058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Eizirik D. L., Cardozo A. K., Cnop M. The role for endoplasmic reticulum stress in diabetes mellitus. Endocrine Reviews . 2008;29(1):42–61. doi: 10.1210/ER.2007-0015. [DOI] [PubMed] [Google Scholar]
- 123.Jiang X., Liu Y., Liu X., et al. Over-expression of a cardiac-specific human dopamine D5 receptor mutation in mice causes a dilated cardiomyopathy through ROS over-generation by NADPH oxidase activation and Nrf2 degradation. Redox Biology . 2018;19:134–146. doi: 10.1016/J.REDOX.2018.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Lu Q., Zheng R., Zhu P., Bian J., Liu Z., Du J. Hinokinin alleviates high fat diet/streptozotocin-induced cardiac injury in mice through modulation in oxidative stress, inflammation and apoptosis. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie . 2021;137 doi: 10.1016/J.BIOPHA.2021.111361. [DOI] [PubMed] [Google Scholar]
- 125.Ni J., Li Y., Xu Y., Guo R. Salidroside protects against cardiomyocyte apoptosis and ventricular remodeling by AKT/HO-1 signaling pathways in a diabetic cardiomyopathy mouse model. Phytomedicine . 2021;82 doi: 10.1016/J.PHYMED.2020.153406. [DOI] [PubMed] [Google Scholar]
- 126.Leask A., Abraham D. J. TGF-beta signaling and the fibrotic response. The FASEB Journal . 2004;18(7):816–827. doi: 10.1096/FJ.03-1273REV. [DOI] [PubMed] [Google Scholar]
- 127.Van Linthout S., Seeland U., Riad A., et al. Reduced MMP-2 activity contributes to cardiac fibrosis in experimental diabetic cardiomyopathy. Basic Research in Cardiology . 2008;103(4):319–327. doi: 10.1007/S00395-008-0715-2. [DOI] [PubMed] [Google Scholar]
- 128.Spinale F. G. Myocardial matrix remodeling and the matrix metalloproteinases: influence on cardiac form and function. Physiological Reviews . 2007;87(4):1285–1342. doi: 10.1152/PHYSREV.00012.2007. [DOI] [PubMed] [Google Scholar]
- 129.Richter K., Konzack A., Pihlajaniemi T., Heljasvaara R., Kietzmann T. Redox-fibrosis: impact of TGFβ1 on ROS generators, mediators and functional consequences. Redox Biology . 2015;6:344–352. doi: 10.1016/J.REDOX.2015.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.LeRoy E. C., Trojanowska M. I., Smith E. A. Cytokines and human fibrosis. Eur Cytokine Netw . 1990;1(4):215–219. https://pubmed.ncbi.nlm.nih.gov/1966552/ [PubMed] [Google Scholar]
- 131.Lang C., Sauter M., Szalay G., et al. Connective tissue growth factor: a crucial cytokine-mediating cardiac fibrosis in ongoing enterovirus myocarditis. Journal of Molecular Medicine (Berlin, Germany) . 2008;86(1):49–60. doi: 10.1007/S00109-007-0249-3. [DOI] [PubMed] [Google Scholar]
- 132.Cucoranu I., Clempus R., Dikalova A., et al. NAD(P)H oxidase 4 mediates transforming growth factor-β1-induced differentiation of cardiac fibroblasts into myofibroblasts. Circulation Research . 2005;97(9):900–907. doi: 10.1161/01.RES.0000187457.24338.3D. [DOI] [PubMed] [Google Scholar]
- 133.Liao H. H., Zhu J. X., Feng H., et al. Myricetin possesses potential protective effects on diabetic cardiomyopathy through inhibiting I κ B α/NF κ B and enhancing Nrf2/HO-1. Oxidative Medicine and Cellular Longevity . 2017;2017:14. doi: 10.1155/2017/8370593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Ma W., Guo W., Shang F., et al. Bakuchiol alleviates hyperglycemia-induced diabetic cardiomyopathy by reducing myocardial oxidative stress via activating the SIRT1/Nrf2 signaling pathway. Oxidative Medicine and Cellular Longevity . 2020;2020:15. doi: 10.1155/2020/3732718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Shinde A. V., Humeres C., Frangogiannis N. G. The role of α-smooth muscle actin in fibroblast-mediated matrix contraction and remodeling. Biochimica et Biophysica Acta - Molecular Basis of Disease . 2017;1863(1):298–309. doi: 10.1016/J.BBADIS.2016.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Ying Y., Jin J., Ye L., Sun P., Wang H., Wang X. Phloretin prevents diabetic cardiomyopathy by dissociating Keap1/Nrf2 complex and inhibiting oxidative stress. Front Endocrinol (Lausanne) . 2018;9 doi: 10.3389/FENDO.2018.00774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Lee J. H., Mellado-Gil J. M., Bahn Y. J., Pathy S. M., Zhang Y. E., Rane S. G. Protection from β-cell apoptosis by inhibition of TGF-β/Smad3 signaling. Cell Death Dis . 2020;11(3) doi: 10.1038/S41419-020-2365-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Alshehri A. S., El-Kott A. F., Eleawa S. M., et al. Kaempferol protects against streptozotocin-induced diabetic cardiomyopathy in rats by a hypoglycemic effect and upregulating SIRT1. J Physiol Pharmacol . 2021;72(3) doi: 10.26402/JPP.2021.3.04. [DOI] [PubMed] [Google Scholar]
- 139.Song M. K., Lee J. H., Ryoo I. G., Lee S. H., Ku S. K., Kwak M. K. Bardoxolone ameliorates TGF-β1-associated renal fibrosis through Nrf2/Smad7 elevation. Free Radical Biology & Medicine . 2019;138:33–42. doi: 10.1016/J.FREERADBIOMED.2019.04.033. [DOI] [PubMed] [Google Scholar]
- 140.Kim B.-C., Jeon W.-K., Hong H.-Y., et al. The anti-inflammatory activity of Phellinus linteus (Berk. & M.A. Curt.) is mediated through the PKCdelta/Nrf2/ARE signaling to up-regulation of heme oxygenase-1. Journal of Ethnopharmacology . 2007;113(2):240–247. doi: 10.1016/J.JEP.2007.05.032. [DOI] [PubMed] [Google Scholar]
- 141.Ryoo I. G., Ha H., Kwak M. K. Inhibitory role of the KEAP1-NRF2 pathway in TGFβ1-stimulated renal epithelial transition to fibroblastic cells: a modulatory effect on SMAD signaling. PLoS One . 2014;9(4) doi: 10.1371/JOURNAL.PONE.0093265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Li G., Yang L., Feng L., et al. Syringaresinol protects against type 1 diabetic cardiomyopathy by alleviating inflammation responses, cardiac fibrosis, and oxidative stress. Molecular Nutrition & Food Research . 2020;64(18) doi: 10.1002/MNFR.202000231. [DOI] [PubMed] [Google Scholar]


