Abstract
Purpose of Review
Masters athletes (MA) are generally considered healthier than their sedentary peers. However, the prevalence of chronic conditions in any population increases with age. Treatments involve pharmacological and non-pharmacological interventions. A substantial proportion of the general population also use dietary supplements (DS). This raises questions about the potential for drug-nutrient interactions which may lead to adverse effects. We sought to determine the potential for drug-nutrient interactions MA may be exposed to by examining the prevalence of chronic conditions treated with medications and their DS use.
Recent Findings
Common conditions in MA include hypertension, hyperlipidemia, asthma, osteoarthritis, depression and anxiety. Treatments may involve prescribed medications. Few recent studies were identified on DS use; however, indications are for around 60% prevalence of supplement usage.
Summary
The higher prevalence of DS use by MA may result in drug-nutrient interactions that impact the effectiveness and safety of prescribed medications for chronic conditions.
Keywords: Masters athletes, Dietary supplements, Drug-nutrient interactions, Chronic conditions, Sports nutrition, Medications
Introduction
Masters athletes (MA) are older individuals who exceed the recommended guidelines for physical activity or are involved in competition and/or systematic training [1]. In competition, the minimum age requirement is usually decided by each sport’s governing body [2], although they are generally over 35 years of age. Along with age, other characteristics such as level of activity and competitive goals (recreational through to internationally competitive), health and demographic characteristics vary widely. While MA are generally reported to be in better health than their sedentary peers [3, 4], physiological changes commonly associated with increasing age [5] and the increased incidence of chronic health conditions [6] may influence their overall health profile.
Masters athletes have been proposed by a number of investigators to be a model of successful ageing [3, 4, 7–9]. This is primarily based upon the well-documented benefits associated with physical activity and exercise on reduced all-cause cardiovascular mortality [10–12] and cause-specific mortality [13]. There is strong support in the literature for the effect of physical activity and exercise in reducing the risk for chronic disease and mortality, while also providing a means for primary chronic disease prevention, typically through mitigation of risk factors [11]. It is therefore presumable that life-long participation in physical activity and exercise is beneficial in preventing a number of chronic diseases and conditions, as well as premature death [14]. Specific to MA, health benefits have been documented with regard to a number of biomedical health determinants and risk factors [15] for chronic disease including obesity via lowering body mass index [4, 16–21], lipids [22], resting blood pressure [23] and improving glycemic control [24]. However, there has been no large-scale, longitudinal study which has investigated whether MA are immune from developing chronic diseases and conditions that are commonly associated with ageing and how they are managed.
The possibility of taking prescribed medications for a chronic condition along with the prevalent use of dietary supplements (DS) in the sporting environment may expose MA to adverse effects due to drug-nutrient interaction(s). Due to the ‘light touch’ regulatory frameworks in many countries, including the USA and Australia, access to DS products does not require consultation or interaction with a healthcare professional, and these products can be easily accessed through pharmacy, retail and online outlets [25, 26]. Concerns about potential drug-nutrient and drug-herb interactions have been reported [27]. Therefore, initiatives that encourage disclosure about prescribed medicine and DS use to healthcare professionals are needed to prevent adverse interactions from occurring [28–30].
This review sought to determine the prevalence of chronic conditions in MA that may involve treatment with prescribed medicines, and the types of DS used by MA as reported in the literature in the last decade. This is followed by a discussion of the possible nutrient-drug interactions to which MA may be exposed.
Chronic Conditions in Masters Athletes
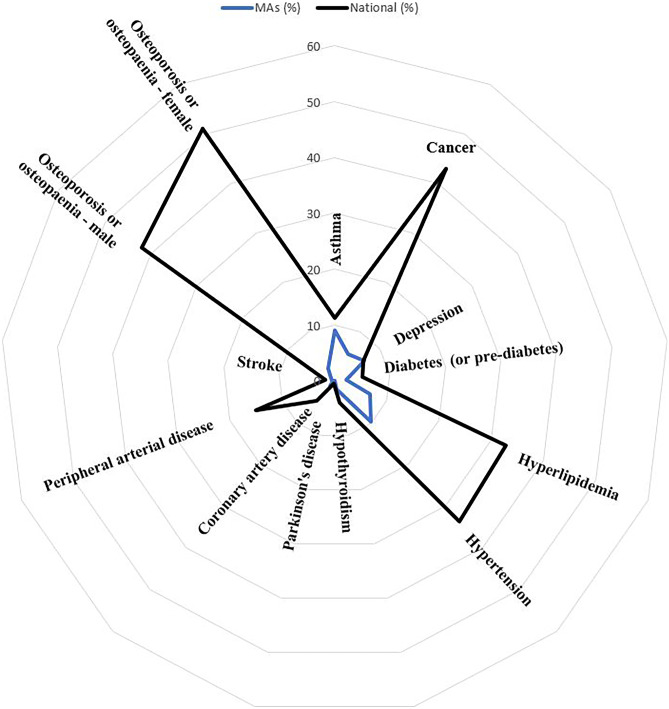
In the last decade, there have been limited studies on MA and their prevalence of chronic disease. Those available, primarily surveys, have investigated chronic disease and conditions in MA participating at sporting competitions. DeBeliso and colleagues [31] surveyed over 900 North American World Masters Games (WMG) participants and reported that MA had a lower prevalence of a number of chronic conditions as compared to the US general population including hypertension (9.1% vs 33%; p < 0.01), rheumatoid/osteoarthritis (10% vs 33%; p < 0.01), hypercholesterolemia (8.0% vs 6.7%; p < 0.01), asthma (6.5% vs 8.2%; p < 0.01) and depression (5.5% vs 8.0%; p < 0.01). Climstein et al. [32•] investigated the medical and health histories of over 8000 MA participating at the World Masters Games and compared their results to the Australian general population [33]. They reported a lower prevalence of asthma, coronary artery disease, stroke, cancer (all types combined), depression, diabetes (impaired fasting glucose, impaired glucose tolerance, type 1 and type 2, combined), hypercholesterolemia, hypertension, hypothyroidism, osteoporosis/osteopenia, Parkinson’s disease and peripheral arterial disease in MA as compared to the Australian general population (Fig. 1).
Fig. 1.
Radar plot of prevalence of chronic conditions in MA participating in the 2009 World Masters Games as compared to the Australian general population (modified with permission from reference [32•])
With regard to subsets of MA, Climstein and colleagues [34] recruited approximately 3200 veteran rugby players (aged 35 to 80 years) participating in an international masters rugby competition to investigate the prevalence of chronic conditions. Compared to the Australian general population [35], MA had a lower prevalence of anxiety (0.0% vs 3.0%; p < 0.05) and depression (3.2% vs 6.0%; p < 0.05). Halar and colleagues combined results from 817 MA surveyed at the Australian Masters Games (n = 130) and the Pan Pacific Masters Games (n = 687) and reported on the prevalence of chronic conditions compared to the Australian adult population [36•]. They found that MA had a lower age-gender-adjusted prevalence for cardiovascular conditions (11.7% vs 30.2%), hypertension (4.6% vs 18.7%), hyperlipidemia (1.6% vs 12.1%), type 2 diabetes mellitus (1.6% vs 5.5%), cancers (all types; 4.3% vs 11.9%), osteoporosis (1.9% vs 5.1%), asthma (9.9% vs 20.7%) and anxiety-related disorders (9.3% vs 18.1%). Kwon and colleagues [37] investigated cardiovascular related outcomes in MA (n = 50) as compared to age-matched healthy controls. The resting heart rate of MA was lower than controls (62.8 vs 74.0 bpm, p < 0.05) and MA had a lower incidence of pre-ventricular contractions during exercise (0.0% vs 24.0%; p < 0.001).
Despite limited research on chronic diseases and conditions in MA, it appears that MA may have a lower prevalence of several chronic conditions including hypertension, cardiovascular disease, hypercholesterolemia, cancer (all types combined), arthritis, osteoporosis, anxiety/depression, and asthma [32•, 36•, 38]. However, the potentially increased occurrence of coronary artery disease (CAD) in MA discussed below is unexpected in this context and not widely known. It is however recognised that the atherosclerotic process begins in early childhood to adolescence [39] and the long-term development of atherosclerosis may outweigh the beneficial effects of chronic physical activity and exercise. The atherosclerotic plaque development in middle-aged and older athletes has, however, been reported to be more stable (based upon plaque type) and hence associated with a lower cardiovascular morbidity and mortality [40].
Coronary Artery Conditions in Masters Athletes: a Special Case
Although the findings from limited studies suggest MA have a lower prevalence for several chronic conditions, some studies have identified risk for CAD in this cohort. Dores et al. [41] investigated 195 MA deemed low to intermediate cardiovascular risk by risk factors, via coronary artery calcium scoring (CAC) and CT angiography. They reported that over one-fourth (25.7%) of the participants were identified with a high (i.e. CAC scores ≥ 75th percentile) atherosclerotic burden (multivessel disease, including the left main coronary artery and left anterior descending artery). Approximately 6% of the participants were identified with obstructive lesions. Additional testing by these clinical researchers [42] identified abnormal graded exercise tests (12.4%), electrocardiographic criteria for myocardial ischemia (5.7%) and exercise-induced ventricular arrhythmias (considered pre-lethal, 6.7%) in the MA.
Merghani and colleagues [43] assessed CAD in 152 MA and compared to age- and gender-matched sedentary controls. They reported MA had a higher prevalence of atherosclerotic plaque (p = 0.009), predominantly calcified (72.7%) versus mixed (calcified and non-calcified) plaque in the sedentary controls (61.5%). Of interest, the number of years MA trained was the only independent variable associated with plaque risk and luminal stenosis ≥ 50%. These investigators also conducted 24-h ECG monitoring on all participants and reported male MA had a higher incidence of atrial fibrillation (sixfold), supraventricular tachycardia (a pre-lethal arrhythmia, threefold) and non-sustained supraventricular tachycardia (non-lethal, ninefold).
Dietary Supplement Use in Masters Athletes
There have been very few studies in the last decade reporting the prevalence of DS use in MA where data on MA can be separated out from other age groups. Guthrie et al. [44•] examined the prevalence of DS use in US Masters Swimming (USMS) members via an electronic survey and compared intake to the US population via data collected in the National Health and Nutrition Examination Survey (NHANES; 2007–2010). The authors hypothesised that MA would have a higher intake of DS than the general US population. Members were asked if they had taken DS in the previous 30 days. All supplements from both surveys were text entries with investigators manually extracting the data and grouping responses into nine categories with at least 10 respondents identifying usage. The USMS athletes (n = 1042) were older, more likely to be female and Caucasian, in better health, more educated and had a greater income than the NHANES sample. The prevalence of supplement use overall (62%) and all supplement categories (1.1–55.8%) was higher than the NHANES cohort in all instances except for ginkgo biloba. The most common supplements were DS used in the prevention or treatment of clinical conditions [45]: vitamins (55.8%), calcium supplements (23.1%), glucosamine and chondroitin (11.6%) and coenzyme Q10 (CoQ10; 5.4%). While the intake of the DS red rice yeast/extract by MA was lower than the general population, there are concerns about it interacting with statins as discussed below. Use of performance enhancing supplements was lower (DHEA/testosterone, 1.2%; creatine, 1.2%). While this study provides a useful insight into DS use by US masters swimmers, the authors acknowledge that the results cannot be generalised to all MA. Additionally, only data on those supplements that could be compared to the NHANES cohort was collected.
Evidence on DS use in athletes typically focusses on elite groups, however, is similarly estimated to be 40–100% [46]. Apart from the report from Guthrie et al. [44•], there are few recent studies specifically in MA that report the prevalence of supplement use. Graham-Paulson et al. reported that athletes over 41 years old with an impairment were most likely to take multivitamin and mineral supplements compared to younger groups. Specific data on different age categories could not be separated [47]. In contrast, Solheim and colleagues found that the use of non-ergogenic aids such as vitamins and minerals was lower in elite athletes and fitness customers aged 35–49 versus younger participants; however, there were only 8 participants reporting use in this age group compared to 215 younger participants [48]. Before 2010, Striegel et al. [49] reported an overall prevalence of DS use (60.5%) at the 2004 World Masters Athletic Championships (n = 598) similar to Guthrie et al. [44•]. The most common supplements reported were vitamins (35.4%) and minerals (29.9%), and creatine was used by 6.5% of those surveyed.
Importantly, reasons for use in MA appear to be different to elite athletes. Masters athletes may be more likely to use DS for health reasons (injury, 25.5%; other health reasons, 19.9%) rather than the performance-related reasons reported by elite groups [49]. This is consistent with findings in the fitness setting where older participants were more likely to use supplements for health and treating nutritional deficiencies, whereas younger participants were more likely to use ergogenic aids [50]. Data from the general population also indicates that the rate of consumption of special dietary foods such as protein powders was higher in young men than overall [51]. Recent evidence indicates that increased intake of protein [52, 53] (potentially via a supplement), creatine [54, 55], nitrates [56] (e.g. via beetroot juice) and other supplements [57] may be beneficial to the health of the ageing athlete and so in some cases, supplement intake may be encouraged by health professionals. Maters athletes’ concern for maintaining their health and increased risk of chronic conditions with age raises the possibility of concurrent use of prescribed medications and DS that may result in adverse side effects. Our observations from data collection at the 2017 Australian Masters Games (AMG) and the 2018 Pan Pacific Masters Games (PPMG) are that almost 50% of MA respondents reported taking up to 16 DS and sports foods, and again, almost 50% used two or more therapies inclusive of medications to treat chronic conditions [58]. Although there were no data collected on concurrent administration, use for treatment of conditions suggests this is the case.
While the focus of this review is not on aspects of the legality of medication and supplement taking in sport, it should be noted that the possibility of consuming a banned substance is also a consideration for MA. They are bound by the International Masters Games Association Anti-Doping rules [59] and may be tested for doping violations. We have observed that many athletes competing at masters games are relatively inexperienced (e.g. ≤ 5 years’ competitive experience) and compete for social or recreational reasons, while others participate for competitive reasons. Inexperienced MA may be unaware of adverse health effects of supplements as well anti-doping regulations in sport. Due to the social nature of many masters competitions, the use of alcohol in this context is an issue that is seldom raised but needs to be discussed. Additionally, we know anecdotally that more experienced competitors may risk adverse health effects including interactions between ingested substances (supplements or medications) that are potentially banned to enhance performance; to our knowledge, it is unreported in the literature. These athletes have often competed when younger and usually know that testing at masters level competitions occurs less frequently than in elite settings, effectively ‘gaming the system’. This activity should be brought to light and addressed at an administrative level. Education on these potentially fatal activities may assist in better understanding their consequences.
Potential Interactions Between Commonly Used Medicines for Chronic Conditions, and Dietary Supplements Used by Masters Athletes
The conditions experienced by MA including osteoarthritis, hypertension, hyperlipidemia depression, anxiety and asthma, albeit significantly lower than in the general population[36•] are typically managed with a combination of non-pharmacological (dietary and lifestyle) and pharmacological interventions [60]. Dietary supplements are also used for these conditions [44•]. A study exploring a representative sample of the USA population and involving 820 individuals reported one in every two people who were taking specific medications to treat infection and/or cardiovascular risk factors (tetracyclines, thiazides or angiotensin II receptor blockers) were at risk of an interaction with a DS [61•]. This section discusses terms around drug-herb-nutrient interactions followed by potential interactions between DS and medications likely to be taken by some MA to treat chronic conditions identified in this review.
Causes of a Drug-Herb-Nutrient Interaction
An interaction between a prescribed medication and a DS ingredient (herb or nutrient) can occur due to an alteration in the pharmacokinetic mechanisms and/or pharmacodynamic properties caused by concurrent intake [62]. A pharmacokinetic interaction is described as enhanced or reduced absorption, distribution, metabolism and excretion of the drug that occurs when two substances are combined [63]. A pharmacodynamic interaction refers to the alteration of a drug’s pharmacological effect and mechanism of action caused by the combination of two or more substances [27]. The clinical consequences of pharmacokinetic and pharmacodynamic interactions vary depending upon whether the effects of the drug are reduced or increased [64]. Increases in a drug’s activity can potentiate its pharmacological activity and increase unwanted side effects and toxicity profiles [64]. Reductions in a drug’s pharmacological activity can result in a sub-optimal therapeutic outcome [64]. Furthermore, interactions between drugs and nutrients may result in a compromised nutritional status [62, 63]. While not an extensive coverage of all potential drug-nutrient interactions, the following seeks to discuss some of the interactions that may be relevant to MA within the context of the conditions they have, medications used to treat those conditions and DS used.
Glucosamine and Chondroitin Interactions with Medicines Used in the Management of Cardiovascular Disease
The available evidence indicates that glucosamine and chondroitin are commonly used among MA for osteoarthritis pain and disability [44•]. However, the evidence regarding the effectiveness of glucosamine and chondroitin for osteoarthritis suggests any statistical trends towards a benefit are of questionable clinical significance [65]. Case report data suggests a pharmacodynamic interaction is possible when glucosamine and chondroitin are taken concurrently with warfarin [66, 67]. Warfarin is an anticoagulant medication commonly used in the prevention of blood clots in patients with atrial fibrillation, myocardial infarction and those with prosthetic heart valves. The potentiation of warfarin’s effect by glucosamine and chondroitin is evidenced by an increase in the International Normalisation Ratio (INR) and a resultant risk of bleeding [66, 67]. When considering the risk–benefit ratio, collectively the minimal clinical benefits of glucosamine and chondroitin in managing the symptoms of osteoarthritis and the potential for a moderately severe adverse interaction, it would be considered prudent to avoid this combination.
Ginkgo biloba and Interactions with Medicines Used in Cardiovascular Disease, Anxiety and Depression
Gingko biloba is a herb promoted for use in a range of chronic conditions including ‘improving circulation, and cognition and memory’ [68]. The use of gingko biloba supplements was also reported by Guthrie et al. to be higher among MA included in their study than the general population [44•]. Caution has been advised about concurrent use with gingko biloba in patients taking anticoagulant drugs such as warfarin, and drugs with anti-platelet effects such as aspirin and ticlopidine. This caution is based upon gingko biloba’s inhibitory effect on platelet aggregation as shown in both in vitro and animal studies and case reports of spontaneous and/or protracted bleeding [69, 70]. However, evidence obtained from studies evaluating the herb’s impact on coagulation status in healthy people have not identified any alterations to bleeding or pharmacokinetic and pharmacodynamic measurements of warfarin [71], or other anticoagulant or antiplatelet medications [64]. One study reported a potential interaction associated with the concurrent use of ginko biloba and the local anaesthetic agent bupivacaine [72]; the patient experienced a retrobulbar haemorrhage.
Increased risk of perioperative bleeding during hip and knee arthroplasty in people taking ginkgo biloba and selective serotonin reuptake inhibitor (SSRI) antidepressant medications has been reported [73]. Both gastrointestinal tract bleeding and intracranial bleeding have also been associated with SSRI use [74]. Therefore, the theoretical concerns and associations with increased risk of bleeding, and potentiation of SSRI pharmacological effects from ginkgo biloba warrant caution until further evidence is obtained.
Coenzyme Q10 and Interactions with Medicines Used to Treat Cardiovascular Risk Factors
Guthrie et al reported MA use of coenzyme Q10 (CoQ10) was substantially higher than the general population [44•]. Coenzyme Q10 is commonly used for its antioxidant and mitochondrial bioenergetic properties [75]. When considering drug interactions, theoretically, due to the structural similarity of CoQ10 and vitamin K, theoretically it is possible that concurrent use may reduce the effectiveness of warfarin’s anticoagulant effects [75]. Conversely and theoretically, the effect of CoQ10 effects on platelet size and receptors may increase the risk of bleeding in patients taking antiplatelet drugs including aspirin [76].
Regarding statin use, studies have reported significant reductions in serum CoQ10 associated with statin therapy for hyperlipidaemia [77–81]. Statins lower serum CoQ10 by inhibiting the enzyme 3-hydro-3-methylglutaryl Coenzyme-A (HMG-CoA) reductase, a rate-limiting step of cholesterol biosynthesis, which subsequently inhibits the conversion of mevalonate to CoQ10 [82]. Collectively these reductions in serum CoQ10 levels, coupled with an understanding the role of CoQ10 in mitochondrial energy production, have prompted the hypothesis that statin-induced CoQ10 deficiency is causal of statin-associated muscle symptoms [83]. Interestingly, to date, no studies have investigated a causal relationship between statin-associated muscle symptoms and CoQ10 depletion which may explain the conflicting results between reviews of clinical studies reporting on the efficacy of CoQ10 in treating this side effect [84, 85].Within the context of hypertension and diabetes, CoQ10 has been shown to have hypotensive [86] and antioxidant effects [86]. Therefore, monitoring blood pressure and blood glucose levels may be prudent during the early stage of concurrent use of CoQ10 and conventional antihypertensives and/or medications used to manage diabetes [86, 87].
Red Yeast Rice Extract and Interactions with Statins Used to Treat Hyperlipidaemia
Promotion of red yeast rice extract’s cholesterol lowering proprieties makes it one of the most commonly used DS in Asia and Europe [88], and may be used by MA [44•]. Red yeast rice contains chemicals that are similar to statin medications including monacolin K, which has the same chemical structure as the lipid lowering drug lovastatin [88]. Red yeast rice has shown to be effective in reducing cholesterol via its naturally occurring constituent ‘lovastatin’ [89]. Studies investigating the pharmacokinetic properties of pure lovastatin and red yeast rice extracts have been conducted to examine the potential for drug interactions of either forms [90]. They identified red rice extracts containing the lovastatin-like compounds inhibited several of the drug metabolising cytochrome P450 enzymes and drug transporter P-glycoprotein activity more than pure lovastatin [90, 91]. In addition, the concomitant administration of red yeast rice extract with medications that are strong inhibitors of the P450 enzyme CYP3A4 could theoretically increase plasma levels of monacolin K, and therefore reduce the safety of red yeast and increase its statin like side effects [92]. A recent review on the safety of red rice extract concluded that despite the overall lack of interaction data associated with red yeast rice extract, patients should be monitored when taking other medications, especially those that are considered to be at a higher risk of statin-associated muscle symptoms [92].
Calcium
While not a common condition reported by MA in this review, Halar et al reported that MA > 50 years old are more likely to experience poorer bone health (osteoporosis or osteopaenia) than younger MA [36•]. Osteoporosis is often treated with agents that affect bone metabolism including bisphosphonates [93]. Bisphosphonates have been shown to form insoluble complexes with multivalent cations such as calcium, aluminium magnesium and iron [93]. Therefore, co-administration of these minerals as DS and/or antacid medication may have a deleterious effect on the bioavailability of oral bisphosphonates [93, 94]. Co-administration of calcium is also known to interact with some antibiotics (quinolones and tetracyclines) by forming insoluble quinolone/tetracycline-calcium complexes, thereby reducing the absorption of the antibiotics [95]. Separation of calcium and these antibiotics by a minimum of 2 h may minimise this interaction and prevent sub-optimal therapeutic outcomes.
Creatine and Caffeine
The use of creatine supplements has been reported to be higher in MA than in the general population [44•]. Creatine is an amino acid derivative that plays a role in creatine kinase, cellular energy provision and intracellular energy shuttling and is used widely in sports recovery and performance [96]. More recently, it has been suggested to have a positive influence on brain function [55]. Little is known about potential interactions between creatine and drugs used to treat common conditions of MA. Interestingly, higher caffeine intake when used concurrently with creatine supplementation in people living and medicated for Parkinson disease has been associated with a significantly faster progression of the disease that is attributed to a negative synergistic effect [97]. Caffeine alone did not have this effect [97].
Conclusion
The literature is scarce regarding the prevalence of chronic conditions and DS use in the broader MA population. However, what is known is that MA do experience osteoarthritis, hypertension, hyperlipidemia, depression, anxiety and asthma, albeit lower than the general population. The available evidence to support pharmacodynamic and pharmacokinetic interactions between the medications used to treat these conditions and the DS used by MA suggests caution is warranted and monitoring required. In some cases, avoiding concurrent use is prudent to avoid harm. Importantly, further research is needed in many instances to establish stronger causal associations and build the evidence base. Furthermore, initiatives that promote awareness about potential drug-nutrient-herb interactions are encouraged and encouraging disclosure about DS to health professionals are encouraged. Masters athletes should be carefully counselled by sports physicians, sports dietitians, general practitioners and pharmacists about concurrent use of prescribed medications and DS. MA are encouraged to disclose their use. Health professionals advising MA on health and sporting performance should be proactive in seeking information from MA about medication and DS use as well as other co-occurring practices such as use of banned substances and alcohol which could potentially be dangerous.
Author Contribution
JG and JH developed the topic for this review. All authors contributed to developing the manuscript.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Compliance with Ethical Standards
Conflict of Interest
All authors have no conflicts of interest to disclose.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Ethics Approval
Where data is presented, ethics approval was obtained by the relevant academic institution (n2009 44, University of Sydney Human Ethics Committee (Protocol number: 2017/592)).
Footnotes
This article is part of the Topical Collection on Sports Nutrition
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Joanna Harnett, Email: Joanna.harnett@sydney.edu.au.
Mike Climstein, Email: michael.climstein@scu.edu.au.
Janelle Gifford, Email: janelle.gifford@sydney.edu.au.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
- 1.Brun SP. Clinical considerations for the ageing athlete. Aust Fam Physician. 2016;45(7):478–483. [PubMed] [Google Scholar]
- 2.International Masters Games Association. About the IMGA: FAQ 2020 [Available from: https://imga.ch/about-imga/faq/.
- 3.Hawkins SA, Wiswell RA, Marcell TJ. Exercise and the master athlete--a model of successful aging? J Gerontol A Biol Sci Med Sci. 2003;58(11):1009–11. 10.1093/gerona/58.11.m1009. [DOI] [PubMed]
- 4.Climstein M, Walsh J, Heazlewood I, DeBeliso M. Endurance masters athletes: a model of successful ageing with clinically superior BMI? The Sport Journal. 2019;21(April):1–11. [Google Scholar]
- 5.Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194–217. 10.1016/j.cell.2013.05.039. [DOI] [PMC free article] [PubMed]
- 6.Barzilai N, Huffman DM, Muzumdar RH, Bartke A. The critical role of metabolic pathways in aging. Diabetes. 2012;61(6):1315–22. 10.2337/db11-1300. [DOI] [PMC free article] [PubMed]
- 7.Geard D, Reaburn PRJ, Rebar AL, Dionigi RA. Masters athletes: exemplars of successful aging? Journal of aging and physical activity. 2017;25(3):490–500. 10.1123/japa.2016-0050. [DOI] [PubMed]
- 8.Tanaka H, Tarumi T, Rittweger J. Aging and physiological lessons from master athletes. Compr Physiol. 2019;10(1):261–96. 10.1002/cphy.c180041. [DOI] [PubMed]
- 9.DeVan AE, Seals DR. Vascular health in the ageing athlete. Exp Physiol. 2012;97(3):305–10. 10.1113/expphysiol.2011.058792. [DOI] [PMC free article] [PubMed]
- 10.Kraus WE, Powell KE, Haskell WL, Janz KF, Campbell WW, Jakicic JM, Troiano RP, Sprow K, Torres A, Piercy KL. Physical activity, all-cause and cardiovascular mortality, and cardiovascular disease. Med Sci Sports Exerc. 2019;51(6):1270–81. 10.1249/mss.0000000000001939. [DOI] [PMC free article] [PubMed]
- 11.Lear SA, Hu W, Rangarajan S, Gasevic D, Leong D, Iqbal R, Casanova A, Swaminathan S, Anjana RM, Kumar R, Rosengren A, Wei L, Yang W, Chuangshi W, Huaxing L, Nair S, Diaz R, Swidon H, Gupta R, Mohammadifard N, Lopez-Jaramillo P, Oguz A, Zatonska K, Seron P, Avezum A, Poirier P, Teo K, Yusuf S. The effect of physical activity on mortality and cardiovascular disease in 130 000 people from 17 high-income, middle-income, and low-income countries: the PURE study. Lancet. 2017;390(10113):2643–54. 10.1016/s0140-6736(17)31634-3. [DOI] [PubMed]
- 12.Lobelo F, Rohm Young D, Sallis R, Garber MD, Billinger SA, Duperly J, Hutber A, Pate RR, Thomas RJ, Widlansky ME, McConnell MV, Joy EA, American Heart Association Physical Activity Committee of the Council on Lifestyle and Cardiometabolic Health, Council on Epidemiology and Prevention, Council on Clinical Cardiology, Council on Genomic and Precision Medicine, Council on Cardiovascular Surgery and Anesthesia, Stroke Council. Routine assessment and promotion of physical activity in healthcare settings: a scientific statement from the American Heart Association. Circulation. 2018;137(18):e495-e522. 10.1161/cir.0000000000000559. [DOI] [PubMed]
- 13.Zhao M, Veeranki SP, Magnussen CG, Xi B. Recommended physical activity and all cause and cause specific mortality in US adults: prospective cohort study. BMJ. 2020;370:m2031. 10.1136/bmj.m2031. [DOI] [PMC free article] [PubMed]
- 14.Cunningham C, R OS, Caserotti P, Tully MA. Consequences of physical inactivity in older adults: a systematic review of reviews and meta-analyses. Scand J Med Sci Sports. 2020;30(5):816–27. 10.1111/sms.13616. [DOI] [PubMed]
- 15.Australian Institute of Health and Welfare. Risk factors contributing to chronic disease 2012. Available from: https://www.aihw.gov.au/reports/chronic-disease/risk-factors-contributing-to-chronic-disease/summary.
- 16.Walsh J, Climstein M, Heazelwood I,DeBeliso M, Adams K, Burke S. Body mass index of masters basketball players. Medicina Sportiva, J Romanian Sports Med Soc. 2013;7:1700–5. [PubMed]
- 17.Walsh J, Climstein M, Heazlewood IT, Burke S, Kettunen J, Adams K, et al. Body mass index for Australian athletes participating in rugby union, soccer and touch football at the World Masters Games. World Acad Sci Eng Technol. 2011;77:1119–1122. [Google Scholar]
- 18.Walsh J, Climstein M, Heazlewood IT, Burke S, Kettunen J, Adams K, DeBeliso M. Improved body mass index classification for football code masters athletes: a comparison to the Australian national population. Int J Biol Med Sci. 2011;1(1):37–40.
- 19.Walsh J, Climstein M, Heazlewood IT, Kettunen J, Burke S, Debeliso M, Adams KJ. Body mass index for athletes participating in swimming at the World Masters Games. J Sports Med Phys Fitness. 2013;53(2):162–8. [PubMed]
- 20.Walsh J, Heazlewood IT, DeBeliso M, Climstein M. Comparison of obesity prevalence across 28 world masters games sports. Sport Sci. 2018;11(1):30–36. [Google Scholar]
- 21.Walsh J, Heazlewood IT, Climstein M. Body mass index in master athletes: review of the literature. J Lifestyle Med. 2018;8(2):79–98. 10.15280/jlm.2018.8.2.79. [DOI] [PMC free article] [PubMed]
- 22.Climstein M, Walsh J, Debeliso M, Heazlewood T, Sevene T, Adams K. Cardiovascular risk profiles of world masters games participants. J Sports Med Phys Fitness. 2016;58(4):489–496. doi: 10.23736/S0022-4707.16.06673-1. [DOI] [PubMed] [Google Scholar]
- 23.Climstein M, Walsh J. Clinically superior blood pressure in world masters games participants? Unpublished Manuscript. 2020.
- 24.Walsh J, Heazlewood T, DeBeliso M, Climstein M. Investigation of fasting plasma glucose in masters athletes. Int J Sport Exerc Health Research. 2020;4(2):65–68. doi: 10.31254/sportmed.4207. [DOI] [Google Scholar]
- 25.Li J, Zhu J, Hu H, Harnett JE, Lei CI, Chau KY, Chan G, Ung COL. Internationalization of traditional/complementary medicine products: market entry as medicine. Chinese Med. 2018;13(1):50. 10.1186/s13020-018-0209-6. [DOI] [PMC free article] [PubMed]
- 26.Binns CW, Lee MK, Lee AH. Problems and prospects: public health regulation of dietary supplements. Annu Rev Public Health. 2018;39(1):403–20. 10.1146/annurev-publhealth-040617-013638. [DOI] [PubMed]
- 27.Gouws C, Hamman JH. What are the dangers of drug interactions with herbal medicines? Expert Opin Drug Metab Toxicol. 2020;16(3):165–167. doi: 10.1080/17425255.2020.1733969. [DOI] [PubMed] [Google Scholar]
- 28.McIntyre E, Foley H, Diezel H, Harnett J, Adams J, Sibritt D, Steel A. Development and preliminarily validation of the Complementary Medicine Disclosure Index. Patient Educ Couns. 2020;103(6):1237–44. [DOI] [PubMed]
- 29.McIntyre E, Foley H, Harnett J, Adams J, Steel A. Development and preliminary evaluation of the Conventional Medicine Disclosure Index. Res Social Adm Pharm. 2021. [DOI] [PubMed]
- 30.Foley H, Steel A, Cramer H, Wardle J, Adams J. Disclosure of complementary medicine use to medical providers: a systematic review and meta-analysis. Sci Rep. 2019;9(1):1–17. doi: 10.1038/s41598-018-38279-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.DeBeliso M, Walsh J, Climstein M, Heazlewood IT, Kettunen J, Sevene T, Adams K. World Masters Games: North American participant medical and health history survey. Sport J. 2014;55(17):1–9.
- 32.• Climstein M, Walsh J, Heazlewood I, Meir R. An overview of risk factors for disease in master athletes. In: Callary B, Young B, Rathwell S, editors. Coaching master athletes: advancing research and practice in adult sport. New York: Routledge; 2021. p. 1–196. In this cross-sectional survey study, the investigators conducted the largest health screening of chronic diseases and conditions in masters athletes. They reported that MA had a significantly lower prevalence of a number of conditions (asthma, coronary artery disease, stroke, cancer, depression, diabetes, hypercholesterolemia, hypertension, hypothyroidism, osteoporosis, Parkinson’s disease and peripheral arterial disease) compared to the Australian general population. It therefore appears that MA do reflect a model of successful ageing with regard to health in middle to older age individuals. However, MA are not completely immune from development of these health conditions.
- 33.Australian Bureau of Statistics. National Health Survey: First results Australia 2017–18 Canberra, Australia 2018 [Available from: https://www.abs.gov.au/statistics/health/health-conditions-and-risks/national-health-survey-first-results/latest-release.
- 34.Climstein M, Walsh J, Heazlewood T, Burke S, Kettunen J, Adams K, DeBeliso M. Incidence of chronic disease and lipid profile in veteran rugby athletes. Int J Biol Med Sci. 2011;1(1):41.
- 35.Australian Bureau of Statistics. 4364.0 - National Health Survey: summary of results, 2007–2008 (Reissue) 2009 [Available from: https://www.abs.gov.au/ausstats/abs@.nsf/mf/4364.0.
- 36.• Halar F, O’Connor H, Climstein M, Prvan T, Black D, Reaburn P, Stuart-Smith W, Gifford JA. Prevalence of chronic conditions in masters games athletes: predicators and comparison to the general population. Unpublished. 2021. This research is the most recent study in this space. The comprehensive analysis and large sample size make this an excellent resource.
- 37.Kwon O, Park S, Kim Y-J, Min S-Y, Kim YR, Nam G-B, Choi K-J, Kim Y-H. The exercise heart rate profile in master athletes compared to healthy controls. Clin Physiol Funct Imaging. 2016;36(4):286–92. 10.1111/cpf.12226. [DOI] [PubMed]
- 38.Gifford J, O’Connor H, Honey A, Caterson I. Nutrients, health and chronic disease in masters athletes. In: Reaburn P, editor. Nutrition and performance in masters athletes. Boca Raton, FL: CRC Press; 2015. pp. 213–41. [Google Scholar]
- 39.McGill HC, Jr., McMahan CA, Herderick EE, Malcom GT, Tracy RE, Strong JP. Origin of atherosclerosis in childhood and adolescence. Am J Clin Nutr. 2000;72(5 Suppl):1307S-15S. 10.1093/ajcn/72.5.1307s. [DOI] [PubMed]
- 40.Aengevaeren VL, Mosterd A, Sharma S, Prakken NHJ, Möhlenkamp S, Thompson PD, Velthuis BK, Eijsvogels TMH. Exercise and coronary atherosclerosis: observations, explanations, relevance, and clinical management. Circulation. 2020;141(16):1338–50. 10.1161/CIRCULATIONAHA.119.044467. [DOI] [PMC free article] [PubMed]
- 41.Dores H, de Araújo Gonçalves P, Monge J, Costa R, Tátá L, Cardim N, Neuparth N, Sharma S. Coronary atherosclerotic burden in veteran male recreational athletes with low to intermediate cardiovascular risk. Rev Port Cardiol (Engl Ed). 2020;39(10):587–94. 10.1016/j.repc.2019.10.009. [DOI] [PubMed]
- 42.Dores H, de Araújo Gonçalves P, Monge J, Costa R, Tátá L, Malhotra A, Sharma S, Cardim N, Neuparth N. Subclinical coronary artery disease in veteran athletes: is a new preparticipation methodology required? Br J Sports Med. 2020;54(6):349–53. 10.1136/bjsports-2018-099840. [DOI] [PubMed]
- 43.Merghani A, Maestrini V, Rosmini S, Cox AT, Dhutia H, Bastiaenan R, David S, Yeo TJ, Narain R, Malhotra A, Papadakis M, Wilson MG, Tome M, AlFakih K, Moon JC, Sharma S. Prevalence of subclinical coronary artery disease in masters endurance athletes with a low atherosclerotic risk profile. Circulation. 2017;136(2):126–37. 10.1161/circulationaha.116.026964. [DOI] [PubMed]
- 44.• Guthrie SK, Erickson SR. Masters swimmers use more dietary supplements than a large national comparison population in the United States. Int J Sport Nutr Exerc Metab. 2016;26(2):123–7. 10.1123/ijsnem.2015-0043. This research is the most recent published work in this space. Comparison to a large population dataset provides insight into differences in health and performance practices of MA. This is the only published work reporting prevalence of supplement taking in MA in the last decade. [DOI] [PubMed]
- 45.Australian Institute of Sport. Australian Institute of Sport Position Statement: supplements and sports foods in high performance sport. Canberra, Australia: Australian Institute of Sport; 2021.
- 46.Garthe I, Maughan RJ. Athletes and supplements: prevalence and perspectives. Int J Sport Nutr Exerc Metab. 2018;28(2):126–38. 10.1123/ijsnem.2017-0429. [DOI] [PubMed]
- 47.Graham-Paulson TS, Perret C, Smith B, Crosland J, Goosey-Tolfrey VL. Nutritional supplement habits of athletes with an impairment and their sources of information. Int J Sport Nutr Exerc Metab. 2015;25(4):387–95. 10.1123/ijsnem.2014-0155. [DOI] [PubMed]
- 48.Solheim SA, Nordsborg NB, Ritz C, Berget J, Kristensen AH, Morkeberg J. Use of nutritional supplements by Danish elite athletes and fitness customers. Scand J Med Sci Sports. 2017;27(8):801–8. 10.1111/sms.12704. [DOI] [PubMed]
- 49.Striegel H, Simon P, Wurster C, Niess AM, Ulrich R. The use of nutritional supplements among master athletes. Int J Sports Med. 2006;27(3):236–41. 10.1055/s-2005-865648. [DOI] [PubMed]
- 50.Saeedi P, Mohd Nasir MT, Hazizi AS, Vafa MR, Rahimi Foroushani A. Nutritional supplement use among fitness club participants in Tehran, Iran. Appetite. 2013;60(1):20–6. 10.1016/j.appet.2012.09.011. [DOI] [PubMed]
- 51.Australian Bureau of Statistics. Australian Health Survey: nutrition first results - foods and nutrients, 2011–12 Canberra: Australian Bureau of Statistics; 2014 [Available from: http://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/4364.0.55.007~2011-12~Main%20Features~Key%20Findings~1.
- 52.Bauer J, Biolo G, Cederholm T, Cesari M, Cruz-Jentoft AJ, Morley JE, Phillips S, Sieber C, Stehle P, Teta D, Visvanathan R, Volpi E, Boirie Y. Evidence-based recommendations for optimal dietary protein intake in older people: a position paper from the PROT-AGE Study Group. J Am Med Dir Assoc. 2013;14(8):542–59. 10.1016/j.jamda.2013.05.021. [DOI] [PubMed]
- 53.Doering TM, Reaburn PR, Phillips SM, Jenkins DG. Postexercise dietary protein strategies to maximize skeletal muscle repair and remodeling in masters endurance athletes: a review. Int J Sport Nutr Exerc Metab. 2016;26(2):168–78. 10.1123/ijsnem.2015-0102. [DOI] [PubMed]
- 54.Stares A, Bains M. The additive effects of creatine supplementation and exercise training in an aging population: a systematic review of randomized controlled trials. J Geriatr Phys Ther. 2020;43(2):99–112. 10.1519/JPT.0000000000000222. [DOI] [PubMed]
- 55.Dolan E, Gualano B, Rawson ES. Beyond muscle: the effects of creatine supplementation on brain creatine, cognitive processing, and traumatic brain injury. Eur J Sport Sci. 2019;19(1):1–14. 10.1080/17461391.2018.1500644. [DOI] [PubMed]
- 56.Coles LT, Clifton PM. Effect of beetroot juice on lowering blood pressure in free-living, disease-free adults: a randomized, placebo-controlled trial. Nutr J. 2012;11(1):106. 10.1186/1475-2891-11-106. [DOI] [PMC free article] [PubMed]
- 57.Brisswalter J, Louis J. Vitamin supplementation benefits in master athletes. Sports Med. 2014;44(3):311–8. 10.1007/s40279-013-0126-x. [DOI] [PubMed]
- 58.Chalmers J, Halar F, O'Connor H, Prvan T, Climstein M, Stuart-Smith W, Reaburn P, Gifford J. Medication and dietary supplement use in masters athletes; prevalence and safety. Sports Medicine New Zealand Conference 2019; 2019 31 October-2 November; Dunedin. Dunedin: Sports Medicine Inc; 2019.
- 59.International Masters Games Association. International Masters Games Assocation: anti-doping 2020 [Available from: https://imga.ch/masters-sports/anti-doping/.
- 60.Budreviciute A, Damiati S, Khdr Sabir D, Onder K, Schuller-Goetzburg P, Plakys G, Katileviciute A, Khoja S, Kodzius R. Management and prevention strategies for non-communicable diseases (NCDs) and their risk factors. Front Public Health. 2020;8:788. [DOI] [PMC free article] [PubMed]
- 61.• Aznar-Lou I, Carbonell-Duacastella C, Rodriguez A, Mera I, Rubio-Valera M. Prevalence of medication-dietary supplement combined use and associated factors. Nutrients. 2019;11(10):2466. This study provides contemporary data from a representative sample of the US population and identified one in every two people (of 820 people) who were taking medications to treat infection (tetracyclines) and/or cardiovascular risk factors such as hypertension (thiazides or angiotensin II receptor blockers) were at risk of an interaction with a dietary supplement that could result in a negative outcome for the patient. [DOI] [PMC free article] [PubMed]
- 62.Chan L-N. Drug-nutrient interactions. J Parenter Enteral Nutr. 2013;37(4):450–9. 10.1177/0148607113488799. [DOI] [PubMed]
- 63.Anadón A, Ares I, Martínez-Larrañaga M-R, Martínez M-A. Interactions between nutraceuticals/nutrients and nutrients and therapeutic drugs. Nutraceuticals: Elsevier; 2021. pp. 1175–1197. [Google Scholar]
- 64.Izzo AA. Interactions between herbs and conventional drugs: overview of the clinical data. Medical Principles and Practice. 2012;21(5):404–28. 10.1159/000334488. [DOI] [PubMed]
- 65.Yang W, Sun C, He SQ, Chen JY, Wang Y, Zhuo Q. The efficacy and safety of disease-modifying osteoarthritis drugs for knee and hip osteoarthritis—a systematic review and network meta-analysis. J Gen Intern Med. 2021:1–9. [DOI] [PMC free article] [PubMed]
- 66.Rozenfeld V, Crain JL, Kevin Callahan A. Possible augmentation of warfarin effect by glucosamine-chondroitin. Am J Health-Syst Pharm. 2004;61(3):306–7. 10.1093/ajhp/61.3.306. [DOI] [PubMed]
- 67.Knudsen JF, Sokol GH. Potential glucosamine-warfarin interaction resulting in increased international normalized ratio: case report and review of the literature and MedWatch database. Pharmacotherapy. 2008;28(4):540–8. 10.1592/phco.28.4.540. [DOI] [PubMed]
- 68.Diamond BJ, Mondragon A. Ginkgo biloba. Complementary and integrative treatments in psychiatric practice. 2017;149.
- 69.Hauser D, Gayowski T, Singh N. Bleeding complications precipitated by unrecognized Gingko biloba use after liver transplantation. Transpl Int. 2002;15(7):377–9. 10.1007/s00147-002-0432-y. [DOI] [PubMed]
- 70.Coxeter PD, Duke CC, Roufogalis B, McLachlan AJ. Ginkgo biloba interactions. Journal of Complementary Medicine: CM, The. 2003;2(3):62–64. [Google Scholar]
- 71.Köhler S, Funk P, Kieser M. Influence of a 7-day treatment with Ginkgo biloba special extract EGb 761 on bleeding time and coagulation: a randomized, placebo-controlled, double-blind study in healthy volunteers. Blood Coag Fibrinol. 2004;15(4):303–309. doi: 10.1097/00001721-200406000-00003. [DOI] [PubMed] [Google Scholar]
- 72.Fong K, Kinnear P. Retrobulbar haemorrhage associated with chronic Gingko biloba ingestion. Postgrad Med J. 2003;79(935):531–532. doi: 10.1136/pmj.79.935.531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Belay ES, Penrose CT, Ryan SP, Bergen MA, Bolognesi MP, Seyler TM. Perioperative selective serotonin reuptake inhibitor use is associated with an increased risk of transfusion in total hip and knee arthroplasty. J Arthroplasty. 2019;34(12):2898–2902. doi: 10.1016/j.arth.2019.04.057. [DOI] [PubMed] [Google Scholar]
- 74.Yuet WC, Derasari D, Sivoravong J, Mason D, Jann M. Selective serotonin reuptake inhibitor use and risk of gastrointestinal and intracranial bleeding. J Am Osteopath Assoc. 2019;119(2):102–111. doi: 10.7556/jaoa.2019.016. [DOI] [PubMed] [Google Scholar]
- 75.Arenas-Jal M, Suñé-Negre JM, García-Montoya E. Coenzyme Q10 supplementation: efficacy, safety, and formulation challenges. Compr Rev Food Sci Food Saf. 2020;19(2):574–94. 10.1111/1541-4337.12539. [DOI] [PubMed]
- 76.Serebruany VL, Ordonez JV, Herzog WR, Rohde M, Mortensen SA, Folkers K, Gurbel PA. Dietary coenzyme Q10 supplementation alters platelet size and inhibits human vitronectin (CD51/CD61) receptor expression. J Cardiovasc Pharmacol. 1997;29(1):16–22. [DOI] [PubMed]
- 77.Chitose T, Sugiyama S, Sakamoto K, Shimomura H, Yamashita T, Hokamaki J, Tsunoda R, Shiraishi S, Yamashita Y, Ogawa H. Effect of a hydrophilic and a hydrophobic statin on cardiac salvage after ST-elevated acute myocardial infarction - a pilot study. Atherosclerosis. 2014;237(1):251–8. 10.1016/j.atherosclerosis.2014.08.053. [DOI] [PubMed]
- 78.McMurray JJV, Dunselman P, Wedel H, Cleland JGF, Lindberg M, Hjalmarson A, Kjekshus J, Waagstein F, Apetrei E, Barrios V, Böhm M, Kamenský G, Komajda M, Mareev V, Wikstrand J, CORONA Study Group. Coenzyme Q10, rosuvastatin, and clinical outcomes in heart failure: a pre-specified substudy of CORONA (controlled rosuvastatin multinational study in heart failure). J Am Coll Cardiol. 2010;56(15):1196–204. 10.1016/j.jacc.2010.02.075. [DOI] [PubMed]
- 79.Mabuchi H, Higashikata T, Kawashiri M, Katsuda S, Mizuno M, Nohara A, Inazu A, Koizumi J, Kobayashi J. Reduction of serum ubiquinol-10 and ubiquinone-10 levels by atorvastatin in hypercholesterolemic patients. J Atheroscler Thromb. 2005;12(2):111–9. 10.5551/jat.12.111. [DOI] [PubMed]
- 80.Rundek T, Naini A, Sacco R, Coates K, DiMauro S. Atorvastatin decreases the coenzyme Q10 level in the blood of patients at risk for cardiovascular disease and stroke. Arch Neurol. 2004;61(6):889–92. 10.1001/archneur.61.6.889. [DOI] [PubMed]
- 81.Oranje WA, Sels JP, Rondas-Colbers GJ, Lemmens PJ, Wolffenbuttel BH. Effect of atorvastatin on LDL oxidation and antioxidants in normocholesterolemic type 2 diabetic patients. Clin Chim Acta. 2001;311(2):91–4. 10.1016/s0009-8981(01)00549-6. [DOI] [PubMed]
- 82.Bouitbir J, Sanvee GM, Panajatovic MV, Singh F, Krähenbühl S. Mechanisms of statin-associated skeletal muscle-associated symptoms. Pharmacol Rev. 2020;154:104201. [DOI] [PubMed]
- 83.Marcoff L, Thompson PD. The role of coenzyme Q10 in statin-associated myopathy: a systematic review. J Am Coll Cardiol. 2007;49(23):2231–7. 10.1016/j.jacc.2007.02.049. [DOI] [PubMed]
- 84.Kennedy C, Köller Y, Surkova E. Effect of Coenzyme Q10 on statin-associated myalgia and adherence to statin therapy: a systematic review and meta-analysis. Atherosclerosis. 2020;299:1–8. 10.1016/j.atherosclerosis.2020.03.006. [DOI] [PubMed]
- 85.Qu H, Guo M, Chai H, Wang WT, Gao ZY, Shi DZ. Effects of coenzyme Q10 on statin-induced myopathy: an updated meta-analysis of randomized controlled trials. J Am Heart Assoc. 2018;7(19):e009835. 10.1161/jaha.118.009835. [DOI] [PMC free article] [PubMed]
- 86.Zozina VI, Covantev S, Goroshko OA, Krasnykh LM, Kukes VG. Coenzyme Q10 in cardiovascular and metabolic diseases: current state of the problem. Curr Cardiol Rev. 2018;14(3):164–174. doi: 10.2174/1573403X14666180416115428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Bonakdar RA, Guarneri E. Coenzyme Q10. Am Fam Physician. 2005;72(6):1065–1070. [PubMed] [Google Scholar]
- 88.Sahebkar A, Serban M-C, Gluba-Brzózka A, Mikhailidis DP, Cicero AF, Rysz J, Banach M. Lipid-modifying effects of nutraceuticals: an evidence-based approach. Nutrition. 2016;32(11):1179–92. 10.1016/j.nut.2016.04.007. [DOI] [PubMed]
- 89.Gerards MC, Terlou RJ, Yu H, Koks C, Gerdes V. Traditional Chinese lipid-lowering agent red yeast rice results in significant LDL reduction but safety is uncertain–a systematic review and meta-analysis. Atherosclerosis. 2015;240(2):415–423. doi: 10.1016/j.atherosclerosis.2015.04.004. [DOI] [PubMed] [Google Scholar]
- 90.Chen C-H, Uang Y-S, Wang S-T, Yang J-C, Lin C-J. Interaction between red yeast rice and CYP450 enzymes/P-glycoprotein and its implication for the clinical pharmacokinetics of lovastatin. Evid Based Complementary Altern Med. 2012;2012. [DOI] [PMC free article] [PubMed]
- 91.Hong S-P, Yang J-S, Han J-Y, Ha S-I, Chung J-W, Koh Y-Y, Chang K-S, Choi D-H. Effects of lovastatin on the pharmacokinetics of diltiazem and its main metabolite, desacetyldiltiazem, in rats: possible role of cytochrome P450 3A4 and P-glycoprotein inhibition by lovastatin. J Pharm Pharmacol. 2011;63(1):129–35. 10.1111/j.2042-7158.2010.01160.x. [DOI] [PubMed]
- 92.Fogacci F, Banach M, Mikhailidis DP, Bruckert E, Toth PP, Watts GF, Reiner Z, Mancini J, Rizzo M, Mitchenko O, Pella D, Fras Z, Sahebkar A, Vrablik M, Cicero AFG, Lipid and Blood Pressure Meta-analysis Collaboration (LBPMC) Group, International Lipid Expert Panel (ILEP). Safety of red yeast rice supplementation: a systematic review and meta-analysis of randomized controlled trials. Pharmacol Res. 2019;143:1–16. 10.1016/j.phrs.2019.02.028.
- 93.Wiesner A, Szuta M, Galanty A, Paśko P. Optimal dosing regimen of osteoporosis drugs in relation to food intake as the key for the enhancement of the treatment effectiveness—a concise literature review. Foods. 2021;10(4):720. doi: 10.3390/foods10040720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Peters ML, Leonard M, Licata AA. Role of alendronate and risedronate in preventing and treating osteoporosis. Cleve Clin J Med. 2001;68(11):945–51. 10.3949/ccjm.68.11.945. [DOI] [PubMed]
- 95.Kowalska-Krochmal B, Dudek-Wicher R. The minimum inhibitory concentration of antibiotics: methods, interpretation, clinical relevance. Pathogens. 2021;10(2):165. doi: 10.3390/pathogens10020165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Clarke H, Kim D-H, Meza CA, Ormsbee MJ, Hickner RC. The evolving applications of creatine supplementation: could creatine improve vascular health? Nutrients. 2020;12(9):2834. doi: 10.3390/nu12092834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Simon DK, Wu C, Tilley BC, Wills A-M, Aminoff MJ, Bainbridge J, et al. Caffeine and progression of Parkinson’s disease: a deleterious interaction with creatine. Clin Neuropharmacol. 2015;38(5):163. doi: 10.1097/WNF.0000000000000102. [DOI] [PMC free article] [PubMed] [Google Scholar]