Abstract
Radioimmunotherapy (RIT) has long been pursued to improve outcomes in acute leukemia and higher-risk myelodysplastic syndrome. Of increasing interest are alpha particle-emitting radionuclides such as astatine-211 (211At) as they deliver large amounts of radiation over just a few cell diameters, enabling efficient and selective target cell kill. Here, we developed 211At-based RIT targeting CD123, an antigen widely displayed on acute leukemia and MDS cells including underlying neoplastic stem cells. We generated and characterized new murine monoclonal antibodies (mAbs) specific for human CD123 and selected four, all of which were internalized by CD123+ target cells, for further characterization. All mAbs could be conjugated to a boron cage, isothiocyanatophenethyl-ureido-closo-decaborate(2-) (B10), and labeled with 211At. CD123+ cell targeting studies in immunodeficient mice demonstrated specific uptake of 211At-labeled anti-CD123 mAbs in human CD123+ MOLM-13 cell tumors in the flank. In mice injected intravenously with MOLM-13 cells or a CD123NULL MOLM-13 subline, a single dose of up to 40 μCi of 211At delivered via anti-CD123 mAb decreased tumor burdens and significantly prolonged survival dose-dependently in mice bearing CD123+ but not CD123− leukemia xenografts, demonstrating potent and target-specific in vivo anti-leukemia efficacy. These data support the further development of 211At-CD123 RIT toward clinical application.
INTRODUCTION
With current therapies, few adults with acute leukemia or higher-risk myelodysplastic syndrome (MDS) are alive 2-5 years after diagnosis.1-6 Radioimmunotherapy (RIT) has been long pursued to improve these outcomes.7-9 Of growing interest as therapeutic payloads are alpha particle emitting radionuclides such as actinium-225 (225Ac) or astatine-211 (211At) since they deliver substantially higher decay energies over a much shorter distance than beta emitters, rendering them more suitable for precise, potent, and efficient target cell killing while minimizing toxicity to surrounding bystander cells.10,11 For several alpha emitters, studies have documented that ≤10 hits kill a neoplastic hematopoietic cell,12-15 potentially reducing the antigen density threshold required for efficacy. With a half-life (t1/2) of 7.2 hours, 211At is particularly appealing for patient application.11
To date, work on alpha emitter-based RIT for acute leukemia and higher-risk MDS primarily involved CD33 (for acute myeloid leukemia [AML] and higher-risk MDS) and CD45 (for AML, higher-risk MDS, and acute lymphoblastic leukemia [ALL]) as targets. While anti-CD45 RIT is mostly explored as intensification of conditioning therapy before allogeneic hematopoietic cell transplantation (HCT) because of broad display of CD45 on normal blood cells, the more restricted expression of CD33 has prompted interest in alpha emitter based anti-CD33 RIT as a stand-alone anti-cancer therapy. Most advanced in clinical testing is the 225Ac-labeled anti-CD33 monoclonal antibody (mAb), 225Ac-lintuzumab (Actimab-A), which showed a response rate of 69% when used with low-dose cytarabine in 13 older adults with previously untreated AML at a dose of 2.0 μCi/kg. However, reflective of “on-target, off-leukemia cell” toxicity to normal blood cells, prolonged, fatal myelosuppression in some patients required reduction to 1.5 μCi/kg/dose, at which level objective responses were much less common (4/18 treated patients) and the study was closed early.16,17
Compared to CD33 or CD45, a more selective RIT approach for acute leukemia or higher-risk MDS may be to target the interleukin-3 (IL-3) receptor alpha chain (CD123), which is expressed on a much more discrete subset of normal hematopoietic cells and is virtually absent on non-blood cells.18-21 CD123 is not only expressed on blast cells in AML (55-near 100%), MDS (45-50%), B-ALL (50-100%), and T-ALL (up to 45%) but also widely overexpressed on leukemic stem/progenitor cells relative to normal hematopoietic stem cells.20,21 This expression pattern has raised great interest in CD123 as target for cancer (stem) cell-specific therapy for people with these malignancies, with several different therapeutic modalities currently under active clinical investigation.18,20,21 Here, we developed new anti-CD123 mAbs and conducted proof of concept in vivo studies of 211At-based anti-CD123 RIT in human acute leukemia xenografts in immunodeficient mice.
MATERIALS AND METHODS
Generation of lentivirus vector expressing human CD123.
A cDNA corresponding to full-length human CD123, with 100% nucleotide identity to human interleukin 3 receptor subunit alpha (IL3RA) transcript variant 1 (NM 002183.4) was generated by expression cloning from a human AML cell line using the forward primer 5’-ATGGTCCTCCTTTGGCTCACG-3’ and reverse primer 5’-TCAAGTTTTCTGCACGACCTG-3,’ via TOPO vector and standard PCR-based cloning procedures, verified by sequencing, and subsequently transferred into a pRRLsin.cPPT.MSCV lentivirus vector containing an internal ribosomal entry site (IRES)/Enhanced Green Fluorescent Protein (EGFP) cassette. Lentiviral particles were prepared as described previously.22
Parental and engineered human acute leukemia cell lines for in vitro studies.
A larger panel of human acute leukemia cell lines was screened for CD123 expression, and a subset of the cells either lacking CD123 or displaying CD123 on the cell surface at various were selected for use in our studies testing either anti-CD123 mAbs or CD123-directed RIT. Human myeloid MOLM-13, KG-1, and TF-1 cells were grown in RPMI-1640 medium with 10% fetal bovine serum (FBS) for MOLM-13, 20% FBS for KG-1, and 10% BCS plus 4 ng/mL GM-CSF (Peprotech; Cranbury, NJ, USA) for TF-1. Human lymphoid RS4;11 cells were grown in alpha-MEM with 10% FBS. MV4;11 cells were grown in IMDM, 10% FBS, 5 ng/mL GM-CSF with 1x Insulin-Transferrin-Selenium supplement (ThermoFisher Scientific; Waltham, MA, USA). All cell lines were maintained with penicillin/streptomycin. Lentivirally-transduced sublines overexpressing human CD123 were generated at multiplicities of infection (MOI) of 0.25-25; EGFP-positive cells were isolated by fluorescence-activated cell sorting (FACS) and re-cultured for further analysis/use. To generate cell sublines with genetic deletion of CD123, clustered regularly interspaced short palindromic repeat (CRISPR)/Cas9-editing was carried out by electroporating purified Cas9 protein (TrueCut Cas9 V2; ThermoFisher Scientific) complexed with synthetic guide RNA (sgRNA) targeting exon 2 of CD123 (sequence 5’-GUCUUUAACACACUCGAUAU-3’) using the ECM 380 Square Wave Electroporation system (Harvard Apparatus; Cambridge, MA, USA) as described. CD123− single cells were isolated via FACS, and loss of CD123 expression confirmed. All cell lines were routinely tested for mycoplasma contamination (MycoAlert™ Mycoplasma Detection Kit; Lonza, Basel, Switzerland) and were authenticated using standard STR CODIS typing.
Engineered MOLM-13 cells for use in immunodeficient mice.
To facilitate in vivo efficacy assessments of radiolabeled anti-CD123 mAbs, MOLM-13 cells were lentivirally transduced with luciferase using pHIV-iRFP720-E2A-Luc (Addgene, plasmid #104587). Following single cell sorting, a clonally derived MOLM-13LUC cell line was established. Using CRISPR/Cas9 followed by single cell cloning, we then derived a CD123-deficient subline of these cells (MOLM-13LUC/CD123KO).
Quantification of CD123 expression.
Expression of CD123 on human acute leukemia cell lines was qualitatively quantified by flow cytometry either using directly labeled commercial anti-CD123 mAbs (clones 6H6 [Invitrogen; Waltham, MA, USA], 9F5 [BD Biosciences; Franklin Lakes, NJ, USA], and 7G3 [BD Biosciences]) or either commercial unlabeled anti-CD123 mAbs (clones 6H6 [Biolegend; San Diego, CA, USA] and 7G3 [BD Biosciences]), or our own anti-CD123 mAbs (1H8, 5G4, 10C4, or 11F11) followed by APC-conjugated goat anti-mouse Ig (Multiple Adsorption, ThermoFisher Scientific). To identify non-viable cells, samples were stained with 4',6-diamidino-2-phenylindole (DAPI). 10,000 events were acquired on a BD FACSCelesta flow cytometer (BD Biosciences), and DAPI-negative cells analyzed using FlowJo version 10 (BD Biosciences).
Quantification of CD123 internalization.
CD123+ acute leukemia cells were incubated with 2 μg/mL unlabeled CD123 antibody at 37°C and aliquots were removed at multiple time points. Samples were then stained with APC-conjugated goat anti-mouse Ig to identify remaining antibody on the cell surface and fluorescence quantified by flow cytometry as described above. Secondary antibody and subsequent analysis by flow cytometry was performed as described above.
Generation of anti-CD123 antibodies.
A peptide immunogen consisting of the entire extracellular domain (ECD) of human CD123 (aa 1-303), fused to the mouse IgG1 Fc domain, was generated, expressed in Freestyle™ 293-F cells, and purified using size exclusion chromatography (SEC) with a Superdex 200 10/300 GL. This peptide was used to immunize BALB/c, CD1, and F1 mice. Hybridoma screening was done by flow cytometry using biotinylated beads coupled to a CD123 ECD His-Avi fusion protein (purified using nickel IMAC resin on an ÄKTA FPLC system, and then characterized by analytical SEC), or with parental human lymphoid or leukemic cells and sublines overexpressing CD123. Hybridomas with reactivity against human CD123 were subcloned, re-screened, and single hybridoma clones expanded for antibody production and purification using MabSelect Sure affinity chromatography. All peptides and antibodies were characterized under reducing and non-reducing conditions by SDS-PAGE to confirm high biochemical quality.
Antibody conjugation and astatination.
Following methodologies described previously, anti-CD123 mAbs were conjugated with isothiocyanatophenethyl-ureido-closo-decaborate(2-) (B10), a boron cage molecule for subsequent astatination.23 B10-conjugated mAbs were purified over size-exclusion (SE) PD-10 desalting columns (Cytivia; Marlborough, MA, USA), analyzed by SE-HPLC, isoelectric focusing, and SDS-PAGE, and assayed by mass spectral analysis to identify the average number of B10 moieties on them. 211At was produced on a Scanditronix MC-50 cyclotron with 29 MeV alpha-beam irradiation of a bismuth metal target.24 After irradiation, 211At was isolated in a “wet chemistry” procedure.23,25 All radioactive materials were handled according to approved Radiation Safety protocols at the Fred Hutchinson Cancer Research Center (Fred Hutch) and the University of Washington.
In vitro assessment of anti-leukemia efficacy of anti-CD123 mAbs.
CD123+ acute leukemia cells were incubated at 37°C (in 5% CO2 and air) in 96-well round bottom plates (Falcon™, Corning; Corning, NY, USA) at 5-10 x 103 cells/well in 225 μL culture medium containing various concentrations of 10C4 or 10C4-B10. After 3 days, cell numbers and drug-induced cytotoxicity, using DAPI to detect non-viable cells, were determined using a LSRII flow cytometer (BD Biosciences) and analyzed with FlowJo Software.
Immunodeficient mice.
Female and male NOD-Rag1null IL2rɣnull/J (NRG) mice (Fred Hutch colony) were maintained under standard protocols approved by the Fred Hutch Institutional Animal Care and Use Committee. This mouse strain was chosen because it tolerates higher levels of radiation and RIT compared to NOD.SCID/IL2rG−/− (NSG) mice and supports transplanted cells from human acute leukemia cell lines without anti-asialo injections to neutralize residual NK cell activity. In all studies, mice were randomized into groups with equivalent body weights, and tumor sizes, and, additionally for efficacy studies, sex ratios.
Assessment of CD123+ cell targeting of 211At-labeled anti-CD123 mAbs.
NRG mice were injected subcutaneously with 106 MOLM-13 cells in the flank. When tumors were palpable (~100 mm3), mice (5 mice/group/time point, randomly assigned) received 50-210 μg of B10-conjugated anti-CD123 mAb, anti-CD45 mAb, or negative control mAb labeled with 5 μCi 211At. Mice were euthanized and organs harvested at different time points within 2-24 hours. Tissues were analyzed on a gamma counter to calculate the percent of injected dose/gram of organ tissue (% ID/g), and radiation absorbed doses for harvested organs were calculated.
In vivo assessment of anti-leukemia efficacy of 211At-labeled anti-CD123 mAbs.
Disseminated leukemia was established by injecting NRG mice (8-11 mice per treatment condition) with 2x105 MOLM-13LUC or, in some experiments, MOLM-13LUC/CD123KO cells by tail vein. Two days later, mice were either left untreated or treated with 10C4-B10 or 10C4-B10 labeled with up to 40 μCi of 211At. All animals received 5x106 bone marrow cells from donor NRG mice 3 days later. Mice were monitored daily, and any animal exhibiting excessive toxicity, morbidity, or weight loss was euthanized per institutional protocol. Tumor burdens were visualized weekly via in vivo fluorescence imaging (IVIS Spectrum; PerkinElmer, Waltham, MA, USA).
Statistical considerations.
Treatment effects on mouse survival were determined by log-rank analyses of Kaplan-Meier survival functions using GraphPad Prism 9.2 (GraphPad Software; San Diego, CA, USA).
Data sharing statement.
For original data and reagents, please contact the corresponding author (rwalter@fredhutch.org).
RESULTS
Production and characterization of new anti-human CD123 mAbs
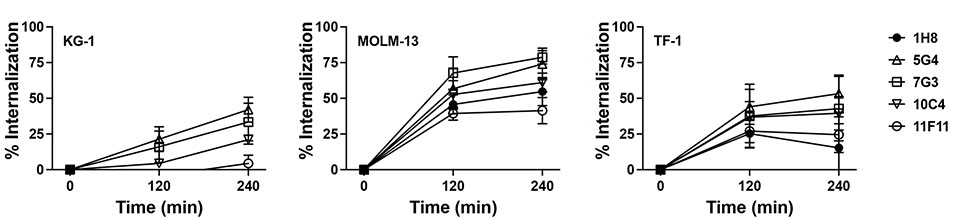
To raise new mAbs as the basis for CD123-directed RIT, we injected BALB/c, CD1, and F1 mice with an immunogen consisting of the murine IgG1 Fc domain linked to the entire ECD of human CD123. In screening assays in which we used CD123+ and CD123− human acute leukemia cell lines, several hybridomas were identified secreting mAbs with the desired specificity. Based on second-round screening following single cell subcloning of the hybridomas of interest, we focused on 4 hybridomas for further characterization (123-10C4 [“10C4”; IgG2b-kappa], 123-5G4 [“5G4”; IgG2a-kappa], 123-11F11 [“11F11”; IgG1-kappa], and 123-1H8 [“1H8”; IgG1-kappa]). Flow cytometric immunophenotyping studies with CD123-positive and CD123-negative human acute leukemia cell lines (including CD123-positive cell lines in which CD123 was deleted via CRISPR/Cas9) confirmed specific binding of all purified mAbs to human CD123 (binding intensity: 10C4>5G4=11F11=1H8), with 10C4 consistently yielding a higher median fluorescence intensity than the widely used commercial anti-CD123 mAb clone, 7G3 (Figure 1). To characterize our mAbs as carriers for cytotoxic cargo, we performed internalization assays, using several CD123-expressing human acute leukemia cell lines. These in vitro studies demonstrated time-dependent uptake of the different clones of anti-CD123 mAbs by all CD123-positive target cell lines although the kinetics of uptake varied across cell lines (Figure 2).
Figure 1. Binding characteristics of new anti-CD123 mAbs.
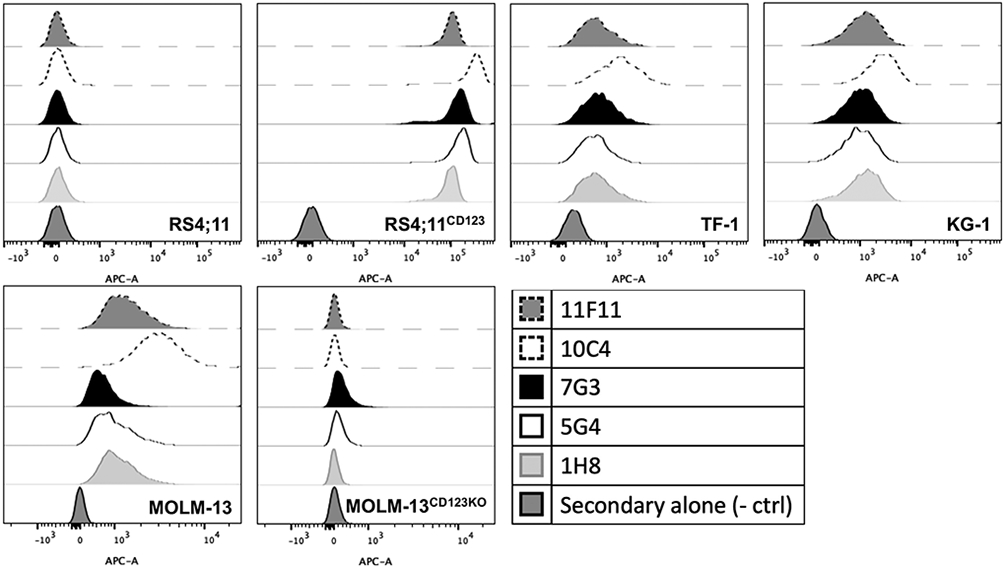
A panel of anti-CD123 mAbs (11F11, 10C4, 7G3, 5G4, 1H8; used at 2 μg/mL) was tested flow cytometrically against parental RS4;11 cells (endogenously lacking CD123), RS4;11 cells transduced with human CD123, CD123-expressing human AML cells lines (TF-1, KG-1, MOLM-13), and MOLM-13 cells with CRISPR/Cas9-mediated knockout of the CD123 locus (CD123KO). Secondary antibody only negative control is shown as well.
Figure 2. Internalization of anti-CD123 mAbs.
Flow cytometry-based in vitro internalization assays of anti-CD123 mAbs (1H8, 5G4, 10C4, 11F11) in CD123+ human acute leukemia cell lines (KG-1, MOLM-13, TF-1). Shown are mean±SEM values of 3-4 independent experiments.
B10 conjugation of anti-CD123 mAbs
All 4 anti-CD123 mAbs could be successfully conjugated with B10. Titration studies in which 5, 10, or 15 B10 molecules per 1 mAb molecule were reacted demonstrated that B10 loading had no impact on binding to CD123 (Supplementary Figure 1). Based on these studies, a conjugation ratio of 10 equivalents of B10 per mAb molecule was selected for subsequent studies. 10C4-B10 was prioritized for further characterization because this anti-CD123 mAb had the most favorable CD123 binding properties in both unconjugated and B10-conjugated form.
In vitro anti-leukemia efficacy of unconjugated and B10-conjugated anti-CD123 mAbs
To study whether anti-CD123 mAbs have any direct anti-leukemia efficacy without a toxic payload, we conducted 3-day in vitro cytotoxicity assays in which we treated several human CD123-expressing acute leukemia cell lines with either unconjugated 10C4 or B10-conjugated 10C4. As depicted in Figure 3, neither unconjugated nor B10-conjugated 10C4, in doses up to 1 μg/mL, inhibited cell growth or induced cell death to any measurable degree.
Figure 3. In vitro cytotoxic properties of 10C4 and 10C4-B10.
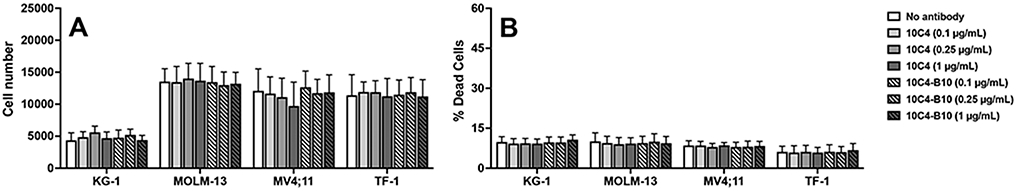
CD123+ human acute leukemia cell lines (KG-1, MOLM-13, MV4;11, and TF-1) were incubated either alone or with unconjugated or B10-conjugated 10C4 at various concentrations as indicated. 3 days later, cell numbers and the percentage of dead cells were quantified by flow cytometry. Shown are mean±SEM values of 3 independent experiments.
CD123+ cell targeting with 211At-labeled anti-CD123 mAbs in immunodeficient mice
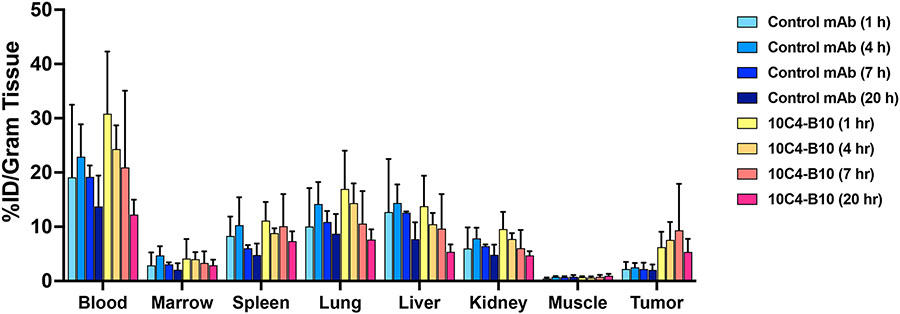
To begin testing anti-CD123 mAbs when labeled with 211At, we characterized the CD123-positive cell targeting properties of 211At-labeled 10C4-B10 in immunodeficient NRG mice bearing parental CD123-positive MOLM-13 AML cell flank tumors. In a first experiment, we tested 3 mAb doses (50 μg, 100 μg, 210 μg) of 10C4-B10 labeled with 5 μCi 211At. Tissues were harvested 7 hours after RIT administration for analysis on a gamma counter to calculate the percent of injected dose/gram of organ tissue (% ID/g), and radiation absorbed doses for harvested organs were calculated. The lowest mAb dose (50 μg) yielded good tumor cell accumulation of 211At and was used in subsequent studies (Supplementary Figure 2). In a time-course experiment, we found robust accumulation of 211At-labeled 10C4-B10 compared to a non-binding 211At-labeled control mAb in MOLM-13 flank tumors, but not other mouse tissues, 1, 4, 7, and 20 hours after RIT administration, indicating favorable CD123+ cell targeting properties of 211At-labeled anti-CD123 mAbs (Figure 4).
Figure 4. In vivo CD123+ cell targeting with 211At-CD123 RIT.
106 parental (CD123+) MOLM-13 cells were implanted into the flanks of NRG mice. One week later, animals (5/group) received 50 μg of either B10-conjugated anti-CD123 mAb (10C4-B10) or B10-conjugated murine IgG1 negative control mAb (BHV-1-B10) labeled with 5 μCi 211At. One hour, 4 hours, 7 hours, or 20 hours later, mice were euthanized, organs harvested, and tissues analyzed on a gamma counter to calculate the percent of injected dose/gram of organ tissue (% ID/g), and radiation absorbed doses for harvested organs calculated. Data are presented as mean±SD.
Assessment of anti-leukemia efficacy of 211At-labeled anti-CD123 mAbs in immunodeficient mice
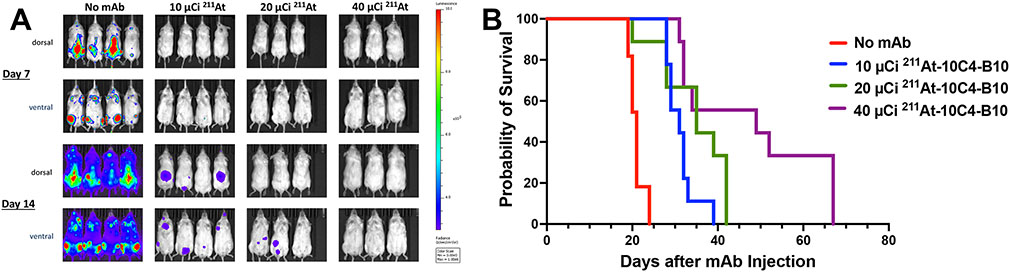
Following demonstration of efficient engraftment of MOLM-13LUC cells in NRG mice, with all animals dying from leukemia within 3-4 weeks of intravenous injection of 2x105 cells (data not shown), we used this disseminated leukemia model to assess the in vivo efficacy of 211At-labeled anti-CD123 mAbs against human acute leukemia. For this purpose, NRG mice were injected with 2x105 MOLM-13LUC cells into tail veins. Two days later, animals were either left untreated or given 50 μg of 10C4-B10 labeled with 10, 20, or 40 μCi of 211At (8-11 animals per group). Each animal then received 5x106 bone marrow cells from donor NRG mice as stem cell support 3 days later, a time when only ~1% of the 211At remained. As shown in Figure 5 and Supplementary Figure 3, a dose dependent decrease in tumor burden was observed in mice receiving 211At-10C4-B10 and survival was significantly prolonged relative to untreated animals (median survival: 49 days [40 μCi of 211At] vs. 31 days [10 μCi of 211At] vs. 21 days [Ctrl]; P<0.0001 for Ctrl vs. 10 μCi, P<0.004 for 10 μCi vs. 40 μCi), demonstrating potent in vivo anti-leukemia efficacy of a single dose of 211At-CD123 RIT. While formal necropsy studies were not performed, death due to progressive leukemia rather than late radiation toxicity was ascertained by serial (weekly) IVIS imaging demonstrating progressive tumor burdens in animals, visual inspection and palpation (demonstrating large tumor masses), rapid weight gain as tumors progressed, and signs/symptoms of disease (lethargy, hind limb paralysis, and labored breathing) being similar in disease-only groups and groups treated with radiation.
Figure 5. In vivo anti-AML efficacy of 211At-CD123 RIT.
(A) In vivo fluorescence imaging 7 and 14 days after administration of 10C4-B10 labeled with either 10 μCi, 20 μCi, or 40 μCi of 211At. Control group was left untreated. (B) Kaplan-Meier survival estimates. 8-11 animals per group. All deaths were attributed to progressive leukemia. P<0.0001 for Ctrl vs. 10 μCi, P<0.004 for 10 μCi vs. 40 μCi.
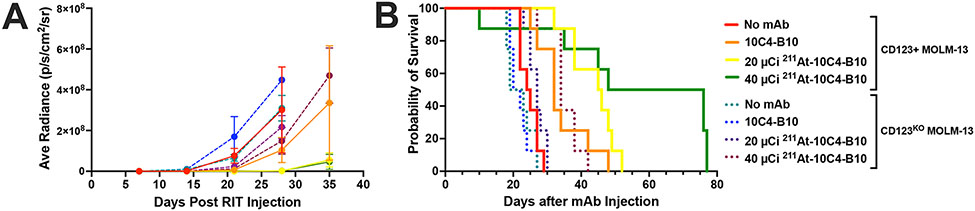
To assess for the target specificity of the anti-leukemia effects of 211At-10C4-B10, we then conducted a study in which parallel cohorts of NRG mice (n=8 per treatment condition) were injected with either MOLM-13LUC cells/mouse or MOLM-13LUC cells in which CD123 was deleted via CRISPR/Cas9 (MOLM-13LUC/CD123KO; 2x105 cells/mouse). Two days later, animals were either left untreated or given 50 μg of unlabeled 10C4-B10 or 50 μg of 10C4-B10 labeled with 20, or 40 μCi of 211At. Like in the initial experiment, each animal then received 5x106 bone marrow cells from donor NRG mice as stem cell support 3 days later. As shown in Figure 6 as well as Supplementary Figure 4 and Supplementary Figure 5, 211At-10C4-B10 led to a dose-dependent decrease in tumor burden and prolonged survival in animals engrafted with human CD123-positive acute leukemia cells, whereas 10C4-B10 alone (i.e. without 211At payload) had a very modest effect on survival (median survival: 62 days [40 μCi of 211At] vs. 45.5 days [20 μCi of 211At] vs. 32 days [10C4-B10 without 211At] vs. 24.5 days [Ctrl]; P=0.0014 for Ctrl vs. 10C4-B10, P=0.031 for 10C4-B10 vs. 20 μCi; P=0.0112 for 10C4-B10 vs. 40 μCi), demonstrating the need for the radioisotope delivered via anti-CD123 mAb for anti-leukemia efficacy of 211At-10C4-B10. In mice engrafted with human CD123-negative acute leukemia cells, 211At-10C4-B10 had a very modest effect of animal survival even at 40 μCi (median survival: 34 days [40 μCi of 211At] vs. 27 days [20 μCi of 211At] vs. 21 days [10C4-B10 without 211At] vs. 21 days [Ctrl]), demonstrating the requirement for CD123 expression for the anti-leukemia efficacy of 211At-CD123 RIT.
Figure 6. Target antigen specificity of 211At-CD123 RIT in vivo.
Two days after injecting NRG mice intravenously with either luciferase-transduced parental CD123+ MOLM-13 cells or a clonally derived subline in which CD123 was deleted via CRISPR/Cas9 (CD123KO MOLM-13), mice were left untreated or injected with 50 μg of 10C4-B10 (i.e. B10-conjugated 10C4 anti-CD123 mAb without radioisotope) or 50 μg of 10C4-B10 labeled with either 20 μCi or 40 μCi of 211At. (A) Average radiance values (mean±SEM) derived from in vivo fluorescence imaging. (B) Kaplan-Meier survival estimates. 8 animals per group. One early death was thought to be a sequelae of radiation exposure rather than leukemia (lack of weight returning to baseline after radiation exposure). All other deaths were attributed to progressive leukemia. For CD123+ cells: P=0.0014 for Ctrl vs. 10C4-B10, P=0.031 for 10C4-B10 vs. 20 μCi; P=0.0112 for 10C4-B10 vs. 40 μCi.
DISCUSSION
CD123 is an appealing drug target to treat acute leukemia and higher-grade MDS because it is expressed on neoplastic blasts in a majority of patients and because it is displayed on underlying malignant stem/progenitor cells.18-21 Contributing to its attractiveness as a target in AML are studies reporting a correlation between higher CD123-positive leukemic stem/progenitor cell burden and worse outcome with AML chemotherapy.26,27 Subsets of AML perhaps particularly suitable for CD123-targeted therapies are NPM1-mutated leukemias28 and cases featuring an expansion of plasmacytoid dendritic cells (pDCs), cells that are readily identified based on high expression of CD123.29,30 pDC-AML is a recently recognized disease entity encompassing approximately 5% of AML cases that is characterized by a high frequency of RUNX1 mutations. In addition to acute leukemias and higher-grade MDS, several other hematologic malignancies also typically express CD123, including classic hairy cell leukemia, Hodgkin lymphoma and some non-Hodgkin lymphomas, chronic myeloid leukemia (CML), eosinophilic leukemia, and systemic mastocytosis. In addition, blastic plasmacytoid dendritic cell neoplasms (BPDCN) is a neoplasm in which uniform, bright expression of CD123 is a hallmark of the disease.18,20,21 Efficacy of tagraxofusp (SL-401), an immunotoxin consisting of human IL-3 fused to truncated diphtheria toxin, in BPDCN validates CD123 as drug target.31,32 In patients with acute leukemia, ongoing efforts with investigational CD123-directed therapies focus on combination therapies with tagraxofusp,33 antibody-drug conjugates (e.g. IMGN632; explored as single agent and in combination with other agents in patients with AML34), T cell engaging bispecific molecules (e.g. flotetuzumab35 or APVO43636), and chimeric antigen receptor (CAR)-modified T cells after unconjugated anti-CD123 mAbs (e.g. talacotuzumab [JNJ-56022473, CSL362]) and single agent tagraxofusp were found to lack sufficient anti-tumor efficacy.37,38 Since some of these drugs have unique toxicities (e.g. capillary leak syndrome in the case of tagraxofusp) that are at least partly related to the mode of action rather than the target antigen, there remains need to explore alternative therapeutic approaches such as the one described herein with 211At.
Some prior studies with anti-CD123 mAbs labeled with gamma-ray emitting indium-111 (111In) suggested the possibility that CD123 could be therapeutically targeted with radionuclides.39-41 In our studies, we revisited this idea using a series of newly developed anti-CD123 mAbs and a highly potent alpha particle emitting radionuclide, 211At, as payload. Overall, the findings suggest the following main conclusions: first, mAbs with specific binding to human CD123 can be conjugated with B10, a boron cage molecule for subsequent astatination, and retain the full binding function of the unmodified parent antibody; second, anti-CD123-B10 mAbs can be labeled with 211At and show favorable CD123-positive cell targeting in immunodeficient mice bearing AML tumor xenografts; and third, 211At-CD123 RIT is effective in prolonging the survival of mice xenotransplanted with CD123-expressing acute leukemia cells in an 211At-dependent, CD123-dependent manner. Together, these data support the further development of 211At-CD123 RIT for patients with acute leukemia, higher-risk MDS, and other CD123-positive hematologic malignancies.
Previous studies have demonstrated that the stability of astatinated mAbs could be significantly improved with the use of the boron cage molecule, B10, for conjugation with lysine amines.23 Preclinical models have demonstrated the efficacy of 211At-B10 labeled anti-CD45, anti-CD38 and anti-CD20 mAbs,42-44 and optimized B10 conjugation followed by astatination is now used in institutional 211At-CD45 and 211At-CD38 RIT trials (e.g. NCT03128034, NCT03670966, NCT04083183, NCT04466475, NCT04579523). The same methodological approach was successfully applied in our studies with anti-CD123 mAbs, thus allowing seamless translation to the clinical setting.
While profound myelosuppression has not consistently been observed in clinical efforts with CD123-directed therapeutics, at least some45,46 (albeit not all47,48) preclinical studies with CD123 CAR T cells suggest the possibility that highly effective targeting of CD123 might lead to prolonged cytopenias. Thus, it may be prudent to explore 211At-CD123 RIT in patients first as a therapy before planned allogeneic HCT, or at least if an allogeneic donor source is available. However, the relatively limited expression of CD123 on normal hematopoietic and non-hematopoietic cells is expected to cause less on-target toxicities to normal tissues compared to CD45 or CD33. This expression pattern may suggest potential value of CD123-directed RIT not only before, or in conjunction with, allogeneic HCT but also as stand-alone therapy, particularly when alpha particle-emitting radionuclides such as 211At are employed. For our proof-of-concept studies, we took advantage of newly developed murine anti-CD123 mAbs. Particularly in the HCT setting, where the agent may be used only once, murine radiolabeled mAbs have established value, and their shorter half-life compared to human mAbs may provide a potential advantage as they could be given in closer relationship to the infused stem cells. Because measurable residual disease (MRD) before allogeneic HCT is well recognized as adverse prognostic factor for poor post-HCT outcome in acute leukemia and other hematologic malignancies,49,50 there is great interest in using MRD-directed therapies before transplantation to minimize relapses after transplantation.44 211At-labeled anti-CD123 mAbs may be ideal for this purpose considering the expression of CD123 on leukemic stem/progenitor cells. However, as a cancer (stem) cell-directed therapeutic, there may be interest in repeated administration in the non-HCT and HCT setting, when formation of neutralizing human anti-mouse antibody (HAMA) reactions might pose an important practical limitation with murine mAbs. Humanized or fully human anti-CD123 mAbs may provide a conceptual advantage in this situation, and efforts to develop such mAbs are currently ongoing.
Supplementary Material
ACKNOWLEDGEMENTS
We would like to thank Dr. Colin E. Correnti, Dr. Christopher Mehlin, Dr. James M. Olson, Jane Carter, and other members of the Molecular Design and Therapeutics (MDT) core facility as well as Benjamin G. Hoffstrom of the Antibody Technology Resource at Fred Hutchinson Cancer Research Center for help with the generation of anti-CD123 mAbs. Research reported in this publication was supported by the Leukemia & Lymphoma Society (Translational Research Program, grant 6489-16), the American Society of Hematology (Bridge Grant), the St. Baldrick Foundation (Emily Beazley Kures for Kids Research Grant), and the National Institutes of Health/National Cancer Institute (NIH/NCI; P30-CA015704). The Fred Hutchinson Cancer Research Center Antibody Technology Resource received support from the M.J. Murdock Charitable Trust.
Footnotes
CONFLICT OF INTEREST
G.S.L., J.J.O., and R.B.W. have filed a provisional patent application related to 211At-CD123 RIT. All other authors declare no competing conflict of interest.
REFERENCES
- 1.Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Büchner T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017; 129(4): 424–447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Malard F, Mohty M. Acute lymphoblastic leukaemia. Lancet 2020; 395(10230): 1146–1162. [DOI] [PubMed] [Google Scholar]
- 3.DeAngelo DJ, Jabbour E, Advani A. Recent advances in managing acute lymphoblastic leukemia. Am Soc Clin Oncol Educ Book 2020; 40: 330–342. [DOI] [PubMed] [Google Scholar]
- 4.Döhner H, Wei AH, Löwenberg B. Towards precision medicine for AML. Nat Rev Clin Oncol 2021; 18(9): 577–590. [DOI] [PubMed] [Google Scholar]
- 5.Platzbecker U Treatment of MDS. Blood 2019; 133(10): 1096–1107. [DOI] [PubMed] [Google Scholar]
- 6.Cazzola M Myelodysplastic syndromes. N Engl J Med 2020; 383(14): 1358–1374. [DOI] [PubMed] [Google Scholar]
- 7.Kotzerke J, Bunjes D, Scheinberg DA. Radioimmunoconjugates in acute leukemia treatment: the future is radiant. Bone Marrow Transplant 2005; 36(12): 1021–1026. [DOI] [PubMed] [Google Scholar]
- 8.Bodet-Milin C, Kraeber-Bodéré F, Eugène T, Guérard F, Gaschet J, Bailly C, et al. Radioimmunotherapy for treatment of acute leukemia. Semin Nucl Med 2016; 46(2): 135–146. [DOI] [PubMed] [Google Scholar]
- 9.Ali AM, Dehdashti F, DiPersio JF, Cashen AF. Radioimmunotherapy-based conditioning for hematopoietic stem cell transplantation: Another step forward. Blood Rev 2016; 30(5): 389–399. [DOI] [PubMed] [Google Scholar]
- 10.Larson SM, Carrasquillo JA, Cheung NK, Press OW. Radioimmunotherapy of human tumours. Nat Rev Cancer 2015; 15(6): 347–360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aghevlian S, Boyle AJ, Reilly RM. Radioimmunotherapy of cancer with high linear energy transfer (LET) radiation delivered by radionuclides emitting alpha-particles or Auger electrons. Adv Drug Deliv Rev 2017; 109: 102–118. [DOI] [PubMed] [Google Scholar]
- 12.Nikula TK, McDevitt MR, Finn RD, Wu C, Kozak RW, Garmestani K, et al. Alpha-emitting bismuth cyclohexylbenzyl DTPA constructs of recombinant humanized anti-CD33 antibodies: pharmacokinetics, bioactivity, toxicity and chemistry. J Nucl Med 1999; 40(1): 166–176. [PubMed] [Google Scholar]
- 13.Sawant SG, Randers-Pehrson G, Metting NF, Hall EJ. Adaptive response and the bystander effect induced by radiation in C3H 10T(1/2) cells in culture. Radiat Res 2001; 156(2): 177–180. [DOI] [PubMed] [Google Scholar]
- 14.Aurlien E, Kvinnsland Y, Larsen RH, Bruland ØS. Radiation doses to non-Hodgkin's lymphoma cells and normal bone marrow exposed in vitro. Comparison of an alpha-emitting radioimmunoconjugate and external gamma-irradiation. Int J Radiat Biol 2002; 78(2): 133–142. [DOI] [PubMed] [Google Scholar]
- 15.Dahle J, Borrebæk J, Jonasdottir TJ, Hjelmerud AK, Melhus KB, Bruland ØS, et al. Targeted cancer therapy with a novel low-dose rate alpha-emitting radioimmunoconjugate. Blood 2007; 110(6): 2049–2056. [DOI] [PubMed] [Google Scholar]
- 16.Finn LE, Levy M, Orozco JJ, Park JH, Atallah E, Craig M, et al. A phase 2 study of actinium-225 (225Ac)-lintuzumab in older patients with previously untreated acute myeloid leukemia (AML) unfit for intensive chemotherapy [abstract]. Blood 2017; 130(Suppl 1): 2638. [Google Scholar]
- 17.Atallah EL, Orozco JJ, Craig M, Levy MY, Finn LE, Khan SS, et al. A phase 2 study of actinium-225 (225Ac)-lintuzumab in older patients with untreated acute myeloid leukemia (AML) - interim analysis of 1.5 μCi/kg/dose [abstract]. Blood 2018; 132(Suppl 1): 1457. [Google Scholar]
- 18.Testa U, Pelosi E, Castelli G. CD123 as a therapeutic target in the treatment of hematological malignancies. Cancers 2019; 11(9). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sugita M, Guzman ML. CD123 as a therapeutic target against malignant stem cells. Hematol Oncol Clin North Am 2020; 34(3): 553–564. [DOI] [PubMed] [Google Scholar]
- 20.El Achi H, Dupont E, Paul S, Khoury JD. CD123 as a biomarker in hematolymphoid malignancies: principles of detection and targeted therapies. Cancers 2020; 12(11). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Patnaik MM, Mughal TI, Brooks C, Lindsay R, Pemmaraju N. Targeting CD123 in hematologic malignancies: identifying suitable patients for targeted therapy. Leuk Lymphoma 2021; 62(11): 2568–2586. [DOI] [PubMed] [Google Scholar]
- 22.Walter RB, Raden BW, Kamikura DM, Cooper JA, Bernstein ID. Influence of CD33 expression levels and ITIM-dependent internalization on gemtuzumab ozogamicin-induced cytotoxicity. Blood 2005; 105(3): 1295–1302. [DOI] [PubMed] [Google Scholar]
- 23.Wilbur DS, Chyan MK, Nakamae H, Chen Y, Hamlin DK, Santos EB, et al. Reagents for astatination of biomolecules. 6. An intact antibody conjugated with a maleimido-closo-decaborate(2-) reagent via sulfhydryl groups had considerably higher kidney concentrations than the same antibody conjugated with an isothiocyanato-closo-decaborate(2-) reagent via lysine amines. Bioconjug Chem 2012; 23(3): 409–420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gagnon K, Risler K, Pal S, Hamlin D, Orzechowski J, Pavan R, et al. Design and evaluation of an external high-current target for production of 211At. J Label Compd Radiopharm 2012; 55(12): 436–440. [Google Scholar]
- 25.Balkin ER, Hamlin DK, Gagnon K, Chyan MK, Pal S, Watanabe S, et al. Evaluation of a wet chemistry method for isolation of cyclotron produced [At-211] astatine. Appl Sci-Basel 2013; 3(3): 636–655. [Google Scholar]
- 26.Vergez F, Green AS, Tamburini J, Sarry JE, Gaillard B, Cornillet-Lefebvre P, et al. High levels of CD34+CD38low/-CD123+ blasts are predictive of an adverse outcome in acute myeloid leukemia: a Groupe Ouest-Est des Leucemies Aigues et Maladies du Sang (GOELAMS) study. Haematologica 2011; 96(12): 1792–1798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mishra P, Tyagi S, Sharma R, Halder R, Pati HP, Saxena R, et al. Proportion of CD34(+)CD38(−)CD123(+) leukemia stem cells at diagnosis varies in ELN risk groups and an emerging novel marker for prognosticating the intermediate risk patients of acute myeloid leukemia: a prospective study. Indian J Hematol Blood Transfus 2021; 37(3): 391–397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Perriello VM, Gionfriddo I, Rossi R, Milano F, Mezzasoma F, Marra A, et al. CD123 Is consistently expressed on NPM1-mutated AML cells. Cancers (Basel) 2021; 13(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Xiao W, Chan A, Waarts MR, Mishra T, Liu Y, Cai SF, et al. Plasmacytoid dendritic cell expansion defines a distinct subset of RUNX1-mutated acute myeloid leukemia. Blood 2021; 137(10): 1377–1391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pemmaraju N Targeting the p-D-C: easy as C-D-1-2-3? Blood 2021; 137(10): 1277–1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pemmaraju N, Lane AA, Sweet KL, Stein AS, Vasu S, Blum W, et al. Tagraxofusp in blastic plasmacytoid dendritic-cell neoplasm. N Engl J Med 2019; 380(17): 1628–1637. [DOI] [PubMed] [Google Scholar]
- 32.Wilson NR, Pemmaraju N. Evaluating tagraxofusp for the treatment of blastic plasmacytoid dendritic cell neoplasm (BPDCN). Expert Opin Pharmacother 2022; 23(4): 431–438. [DOI] [PubMed] [Google Scholar]
- 33.Lane AA, Stein AS, Garcia JS, Garzon JL, Galinsky I, Luskin MR, et al. Safety and efficacy of combining tagraxofusp (SL-401) with azacitidine or azacitidine and venetoclax in a phase 1b study for CD123 positive AML, MDS, or BPDCN [abstract]. Blood 2021; 138(Suppl 1): 2346. [Google Scholar]
- 34.Daver N, Aribi A, Montesinos P, Roboz GJ, Wang ES, Walter RB, et al. Safety and efficacy form a phase 1b/2 study of IMGN632 in combination with azacitidine and venetoclax for patients with CD123-positive acute myeloid leukemia [abstract]. Blood 2021; 138(Suppl 1): 372. [Google Scholar]
- 35.Uy GL, Aldoss I, Foster MC, Sayre PH, Wieduwilt MJ, Advani AS, et al. Flotetuzumab as salvage immunotherapy for refractory acute myeloid leukemia. Blood 2021; 137(6): 751–762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Uckun FM, Lin TL, Mims AS, Patel P, Lee C, Shahidzadeh A, et al. A clinical phase 1B study of the CD3xCD123 bispecific antibody APVO436 in patients with relapsed/refractory acute myeloid leukemia or myelodysplastic syndrome. Cancers (Basel) 2021; 13(16). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Montesinos P, Roboz GJ, Bulabois CE, Subklewe M, Platzbecker U, Ofran Y, et al. Safety and efficacy of talacotuzumab plus decitabine or decitabine alone in patients with acute myeloid leukemia not eligible for chemotherapy: results from a multicenter, randomized, phase 2/3 study. Leukemia 2021; 35(1): 62–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Aldoss I, Clark M, Song JY, Pullarkat V. Targeting the alpha subunit of IL-3 receptor (CD123) in patients with acute leukemia. Hum Vaccin Immunother 2020; 16(10): 2341–2348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Leyton JV, Gao C, Williams B, Keating A, Minden M, Reilly RM. A radiolabeled antibody targeting CD123(+) leukemia stem cells - initial radioimmunotherapy studies in NOD/SCID mice engrafted with primary human AML. Leuk Res Rep 2015; 4(2): 55–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gao C, Leyton JV, Schimmer AD, Minden M, Reilly RM. Auger electron-emitting (111)In-DTPA-NLS-CSL360 radioimmunoconjugates are cytotoxic to human acute myeloid leukemia (AML) cells displaying the CD123(+)/CD131(−) phenotype of leukemia stem cells. Appl Radiat Isot 2016; 110: 1–7. [DOI] [PubMed] [Google Scholar]
- 41.Bergstrom D, Leyton JV, Zereshkian A, Chan C, Cai Z, Reilly RM. Paradoxical effects of Auger electron-emitting (111)In-DTPA-NLS-CSL360 radioimmunoconjugates on hCD45(+) cells in the bone marrow and spleen of leukemia-engrafted NOD/SCID or NRG mice. Nucl Med Biol 2016; 43(10): 635–641. [DOI] [PubMed] [Google Scholar]
- 42.Orozco JJ, Bäck T, Kenoyer A, Balkin ER, Hamlin DK, Wilbur DS, et al. Anti-CD45 radioimmunotherapy using (211)At with bone marrow transplantation prolongs survival in a disseminated murine leukemia model. Blood 2013; 121(18): 3759–3767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Green DJ, Shadman M, Jones JC, Frayo SL, Kenoyer AL, Hylarides MD, et al. Astatine-211 conjugated to an anti-CD20 monoclonal antibody eradicates disseminated B-cell lymphoma in a mouse model. Blood 2015; 125(13): 2111–2119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.O'Steen S, Comstock ML, Orozco JJ, Hamlin DK, Wilbur DS, Jones JC, et al. The alpha-emitter astatine-211 targeted to CD38 can eradicate multiple myeloma in a disseminated disease model. Blood 2019; 134(15): 1247–1256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gill S, Tasian SK, Ruella M, Shestova O, Li Y, Porter DL, et al. Preclinical targeting of human acute myeloid leukemia and myeloablation using chimeric antigen receptor-modified T cells. Blood 2014; 123(15): 2343–2354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Baroni ML, Sanchez Martinez D, Gutierrez Aguera F, Roca Ho H, Castella M, Zanetti SR, et al. 41BB-based and CD28-based CD123-redirected T-cells ablate human normal hematopoiesis in vivo. J Immunother Cancer 2020; 8(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pizzitola I, Anjos-Afonso F, Rouault-Pierre K, Lassailly F, Tettamanti S, Spinelli O, et al. Chimeric antigen receptors against CD33/CD123 antigens efficiently target primary acute myeloid leukemia cells in vivo. Leukemia 2014; 28(8): 1596–1605. [DOI] [PubMed] [Google Scholar]
- 48.Stevens BM, Zhang W, Pollyea DA, Winters A, Gutman J, Smith C, et al. CD123 CAR T cells for the treatment of myelodysplastic syndrome. Exp Hematol 2019; 74: 52–63 e53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Berry DA, Zhou S, Higley H, Mukundan L, Fu S, Reaman GH, et al. Association of minimal residual disease with clinical outcome in pediatric and adult acute lymphoblastic leukemia: a meta-analysis. JAMA Oncol 2017; 3(7): e170580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Short NJ, Zhou S, Fu C, Berry DA, Walter RB, Freeman SD, et al. Impact of measurable residual disease on survival outcomes in patients with acute myeloid leukemia: a meta-analysis. JAMA Oncol 2020; 6(12): 1890–1899. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.