Abstract
Background
The crystal-induced calcium pyrophosphate deposition disease (CPPD) clinically appearing as pseudogout differs from the mere radiographic finding of chondrocalcinosis (CC) but may cause symptoms resembling rheumatoid arthritis (RA).
Objective
To study the prevalence of CPPD and CC in rheumatic diseases focusing on differences between seropositive and seronegative RA.
Patients and methods
In a retrospective study design, we analysed records and radiographs of consecutive new patients presenting to our centre between January 2017 and May 2020. 503 patients were identified based on expert diagnoses: 181 with CPPD, 262 with RA, 142 seropositive (54.2%) and 120 seronegative RA, gout (n=30) and polymyalgia rheumatica (n=30), mean symptom duration <1 year in almost all patients.
Results
The majority of patients had only one rheumatological diagnosis (86.9%). Most patients with CPPD (92.6%) had radiographic CC, primarily in the wrists. The prevalence of CC was higher in seronegative (32.3%) than in seropositive RA (16.6%), respectively (p<0.001). Patients with CPPD were older (p<0.001) and had acute attacks more frequently than patients with RA (p<0.001), who had symmetric arthritis more often (p=0.007). The distribution pattern of osteoarthritic changes in radiographs of hands and wrists differs between patients with RA and CPPD. CC was present in more than one joint in 73.3% of patients with CPPD, 9.6% with seropositive and 18.7% with seronegative RA.
Discussion
CPPD and CC were more frequent in seronegative versus seropositive RA. Symmetry of arthritis and acuteness of attacks differentiated best between CPPD and RA but localisation of joint involvement did not. Co-occurrence of both diseases was frequently observed.
Keywords: Rheumatoid Arthritis, Chondrocalcinosis, Crystal arthropathies
WHAT IS ALREADY KNOWN ON THIS TOPIC?
Calcium pyrophosphate deposition disease (CPPD) is an important differential diagnosis to rheumatoid arthritis (RA) which is complicated due to similarity of clinical features.
WHAT THIS STUDY ADDS?
For the first time, in a large cohort, we show that the prevalence of CPPD occurs more often in seronegative than in patients with seropositive RA. Furthermore, patients with seronegative RA and CPPD differed in the acuteness of attacks and in the distribution pattern of osteoarthritic changes in hand and wrist radiographs.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE AND/OR POLICY?
In clinical practice, seronegative RA diagnoses should be regularly re-evaluated to ensure proper treatment. Why CPPD is more commonly found in patients with seronegative than in seropositive RA should be investigated.
Introduction
Calcium pyrophosphate crystal deposition disease (CPPD), a rather common but not well studied disease to date, may present as an acute arthritis or a chronic arthropathy with development of radiographic structural changes, or may present as an often incidental radiographic finding called chondrocalcinosis (CC).1 The clinical presentation of patients with CPPD is variable2 and may well resemble other rheumatic diseases such as rheumatoid arthritis (RA), polymyalgia rheumatica (PMR) and even ankylosing spondylitis. In the absence of a decisive test some uncertainty regarding the diagnosis often remains. Some years ago, a EULAR task force tried to standardise the terminology and classification of CPPD.3 The experts agreed that ‘CPPD’ should be used as the umbrella term that includes asymptomatic CPPD, acute CPP crystal arthritis, chronic inflammatory polyarthritis associated with CPPD and osteoarthritis (OA) with CPPD. CC defines cartilage calcification, most commonly due to CPPD and detected by imaging or histological examination. A total of 11 key recommendations were agreed on regarding the topics of clinical features, synovial fluid (SF) examination, imaging, comorbidities, and risk factors.3
The epidemiology of manifestations and phenotypes of CPPD has not been clarified to date.4 Although intra-articular CPPD occurs in all ages and both sexes, it is associated with older age, haemochromatosis, hyperparathyroidism, hypophosphatasia and hypomagnesaemia. Whether women are more frequently affected than men is unclear.5–7 Rarely, CPPD can be inherited as a monogenic autosomal dominant disease.8
The reported prevalence of CPPD depends on the joints studied. In an Italian survey CC was among the five most prevalent musculoskeletal conditions with a prevalence of 0.42%.9 In other epidemiological studies, CC was found in 7%–10% of knees in combination with hands, wrists, hips and the symphysis pubis, respectively.10–12 CPPD seems to predominantly affect knees, but it does also occur in the absence of knee CC.13 An association of CC with OA has been reported,14–18 but hip OA does not associate with CC at the hip or distant joints.11 18 CPPD may also manifest at the spine,19 not only in form of the crowned dens syndrome.20
No validated diagnostic or classification criteria for CPPD have been published to date.21 22 The diagnostic criteria proposed by McCarty and Ryan are the only criteria to define CPPD to date but they are not widely used.22 However, a multidisciplinary international working group is currently developing ACR/EULAR classification criteria.23 According to that proposal ‘A case is considered definite if CPPD crystals are demonstrated in tissues or synovial fluid or if crystals compatible with CPPD are demonstrated by compensated polarized light microscopy and typical calcifications are seen on radiographs. If only one of these criteria is found, a probable diagnosis is made’.24 The most direct but least sensitive method to diagnose CPPD is to detect calcium pyrophosphate crystals in the SF of patients under suspicion of CPPD by polarised light microscopy which has been considered as the gold standard for diagnosing CPPD3 but the reliability and feasibility of this approach has been challenged.21 Thus, conventional radiography is mostly used to diagnose CC and CPPD. However, the mere presence of CC is not sufficient to diagnose CPPD. Thus, it is critical to include clinical criteria such as the acuteness of attacks and flares, swollen, warm and often erythematous joints to increase the accuracy of diagnosis.21 However, such symptoms are also present and may mimic other inflammatory diseases such as gout, sarcoidosis, RA, PMR or septic arthritis. In the 2011 EULAR recommendations for CPPD, No.5 advises to consider chronic CPPD in the differential diagnosis of RA.3
Although conventional radiography and SF crystal analysis have remained reference standards for diagnosing CPPD, there are other imaging methods such as ultrasound, dual energy CT (DECT) and MRI which have been used for diagnosis and differential diagnosis of CPPD.25 26 However, these imaging procedures have not been established to diagnose CPPD and CC to date but are subject to more research.
The aim of this study was to compare the clinical picture and radiographs of affected joints in clinical diagnosed CPPD and CC with seropositive and seronegative RA, PMR and gout to determine the prevalence and the clinical differences of these rather common diseases in daily clinical practice.
Methods
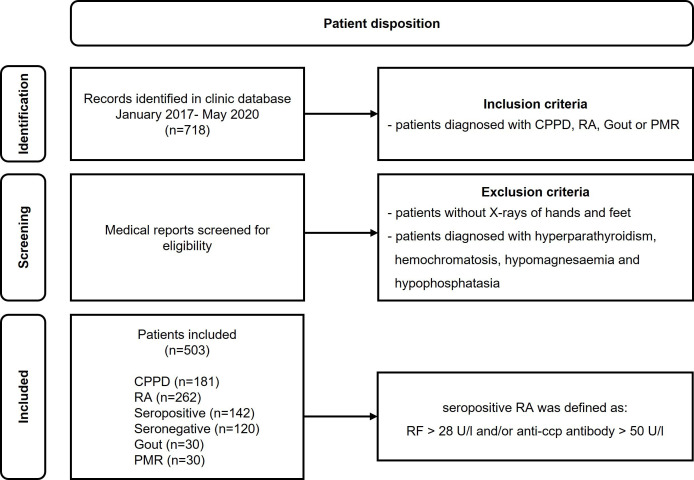
In this retrospective cross-sectional study, we analysed the records of consecutive patients presenting to our specialised centre between January 2017 and May 2020. A total of 718 patients were identified, 239 with CPPD, 279 with RA, 100 with PMR and 100 with gout. Only patients with available complete sets of X-rays of hands and feet were included. Patients with known hyperparathyroidism, haemochromatosis, hypomagnesaemia and hypophosphatasia were not included. Patients who presented more than once during this period were only counted as one patient (figure 1).
Figure 1.
Patient disposition. anti-CCP, anti-cyclic citrullinated peptide; CPPD, calcium pyrophosphate deposition disease; PMR, polymyalgia rheumatica; RA, rheumatoid arthritis; RF, rheumatoid factor.
Anonymised data were transferred to case report forms. Patients’ history and laboratory results were systematically analysed for sex, age, age at first diagnosis, height, weight, BMI, smoking history, morning stiffness, localisation of symptoms, symptom onset, erythrocyte sedimentation rate (ESR), C reactive protein (CRP), anticyclic citrullinated peptide (anti-CCP) antibodies, rheumatoid factor (RF) and uric acid. Patients diagnosed with RA and a rheumatic factor titre >28 U/L and or anti-CCP antibodies >50 U/L were defined as seropositive. All patients with RA including patients with CPPD plus RA fulfilled the 2010 EULAR criteria.27 Patients with PMR and gout were, after the clinical diagnosis, confirmed by fulfilling the last classification criteria for these diseases.28 29 CPPD was diagnosed by radiographs and/or ultrasound, in combination with the clinical picture including physical examination of the affected joints. CPPD was diagnosed in patients with typical cartilage or joint capsule calcification on radiological examinations and joint swelling, warmth, redness and/or pain on clinical examination of the affected joint. CC in cartilage or joint capsule was assumed if linear or punctate opacities in fibro or hyaline articular cartilage was detected. This is consistent with the established diagnostic criteria for CC on conventional radiography.22 These were recently confirmed in the new 2022 ACR/EULAR diagnostic criteria for CCPD.23 Symmetry of joint involvement was assumed if at least 50% of the symptomatic joints were affected on both sides. For the analysis of lab results, clinical symptoms and the radiological distribution of CC Chi-square-tests were used. To analyse the influence of age on the prevalence of a diagnosis the Mann-Whitney test was used. P values and 95% CIs are given where appropriate.
To assess possible correlations between individual risk factors and disease, patients with more than one established disease were excluded from the respective statistical analyses. Patients with only one disease are labelled as only CPPD and only RA, respectively. Radiographs were analysed by experienced musculoskeletal radiologist for the presence of erosions, CC and osteoarthritic changes.
Information about the medication at the time of hospitalisation and discharge included dosages of non-steroidal anti-inflammatory agents (NSAIDs), opioids, prednisolone, colchicine and disease modifying antirheumatic drugs (DMARDs), for example, conventional synthetic (cs)DMARDs, biologic (b)-DMARDs and targeted synthetic tsDMARD.
Results
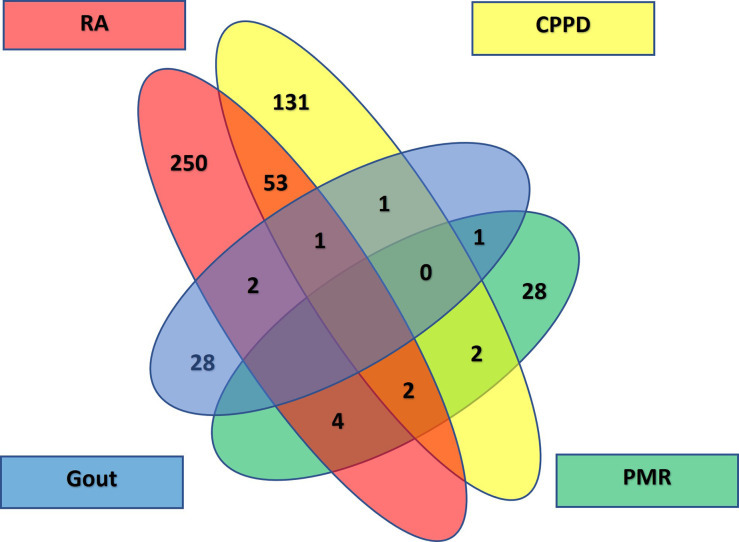
A total of 503 patients (70.1% of the initially identified patients) were included: 181 with CPPD (36%), 262 with RA (52.1%), 142 patients with seropositive (54.2% of all patients with RA) and 120 with seronegative RA (45.8%), 30 with gout and 30 with PMR, respectively. Of these 503 patients, 351 were female (69.8%) and 152 male (30.2%). An overview of patients’ disposition is shown in a Venn diagram (figure 2). A total of 437 patients had just one diagnosis (86.9%), 63 patients had two (12.5%) and 3 patients three diagnoses (0.6%). Among patients with one diagnosis, 131 had CPPD (30%), 103 seronegative RA (23.6%), 147 seropositive RA (33.6%), 28 PMR and 28 gout (6.4% each).
Figure 2.
Venn diagram patient overview. CPPD, calcium pyrophosphate deposition disease; PMR, polymyalgia rheumatica; RA, rheumatoid arthritis.
Patients with CPPD were older (74.3±9.7 years) than those with RA (63.8±14.0 years). The mean age at the time of diagnosis was 73.4±11.3 years in patients with CPPD, 53.2±14.8 years in those with seropositive and 59.4±14.4 years in those with seronegative RA (table 1).
Table 1.
Patient demographics (n=490)
| CPPD only | Seropositive RA only |
Seronegative RA only |
Gout only | PMR only | CPPD+RA | |
| N | 131 | 147 | 103 | 28 | 28 | 53 |
| Percentage of females (%) | 77.1 | 70.8 | 75.4 | 28.6 | 42.9 | 81.1 |
| Mean age (years) | 74.3±9.7 | 61.5±13.7 | 62.2±13.7 | 57.3±14.5 | 64.9±8.0 | 74.7±9.3 |
| Symptom duration (years) | 0.4±1.1 | 0.7±1.9 | 0.8±1.3 | 1.2±3.0 | 0.5±1.9 | 0.6±1.6 |
| Chondrocalcinosis (radiography, %)* | 90.1 | 10.9 | 4.9 | 7.1 | 3.6 | 100 |
| Erosions (radiography, %) | 25.2 | 44.2 | 27.2 | 53.6 | 3.6 | 37.7 |
| Osteoarthritis (radiography, %) | 97.8 | 89.8 | 92.2 | 96.4 | 100.0 | 98.1 |
| Clinical involvement of hands (%) | 64.2 | 74.1 | 76.7 | 21.4 | 42.9 | 83.0 |
| Clinical involvement of wrists (%) | 23.7 | 34.0 | 39.8 | 3.6 | 7.1 | 24.5 |
| Clinical involvement of feet excluding ankles (%) | 36.6 | 35.4 | 36.0 | 53.6 | 14.3 | 30.1 |
| Clinical involvement of knee (%) | 45.0 | 46.3 | 46.6 | 60.8 | 32.1 | 43.4 |
| Symmetric arthritis (%) ≥50% of the affected joints | 69.5 | 81.6 | 85.4 | 60.7 | 89.3 | 83.0 |
| Acute attacks, sudden onset (%) | 67.9 | 25.9 | 28.2 | 67.9 | 35.7 | 35.8 |
| Both diagnoses RA and CPPD (%) | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 100 |
| Therapy with MTX or another cDMARD (%) | 15.3 | 76.2 | 51.5 | 0.0 | 25.0 | 32.1 |
| Therapy with hydroxychloroquine (%) | 7.6 | 4.0 | 8.7 | 0.0 | 0.0 | 5.7 |
| Therapy with colchicine (%) | 48.1 | 1.0 | 0 | 82.1 | 0.0 | 34.0 |
*Patients with diagnoses other than CPPD who had chondrocalcinosis on radiographs but no clinical symptoms in these joints were not diagnosed and classified as having CPPD.
cDMARD, conventional disease modifying antirheumatic drug; CPPD, calcium pyrophosphate deposition disease; MTX, methotrexate; PMR, polymyalgia rheumatica; RA, rheumatoid arthritis.
Age was not different between males (mean age 72.6 years (95% CI 69.1 to 76.1)) and females (mean age 74.8 years (95% CI 73.8 to 75.8)) in patients with CPPD only, and neither age nor sex had an influence on the results of these patients.
The majority of patients with CPPD plus RA (83.0%) was first diagnosed with RA, and later with CPPD, while a minority was diagnosed simultaneously (9.4%), and for 7.6% no information was obtained. There was a time lag of 9.2±6.4 years between these two diagnoses.
There was no difference in age at diagnosis of CPPD between patients who had CPPD only and those who had CPPD plus RA (73.4±11.3 vs 73.6±9.7 years, respectively). Patients with both, CPPD and RA, were diagnosed with RA at a mean age of 65.9±9.5 years (95% CI 63.4 to 68.4), while patients with just RA were diagnosed at a mean age of 54.0±14.9 years (95% CI 52.2 to 55.8). Thus, patients with CPPD plus RA were older when diagnosed with RA than patients with RA only. More women than men were affected by CPPD (77.9%) and RA (72.0%), respectively (p<0.001). There was no difference in CRP levels and ESR between patients with CPPD and seronegative RA (table 2).
Table 2.
CRP, ESR, rheumatoid factor, anti-CCP and urid acid serum level
| Diagnosis | N | CRP >0.5 mg/dL |
ESR >20 mm/hour |
RF >28 U/L |
Anti-CCP abs >50 U/L |
Urid acid ♀>5.7 mg/dL ♂>7.0 mg/dL |
| CPPD only | 131 | 74 (56.5%) | 61 (46.4%) | 3 (2.3%) | 2 (1.5%) | 41 (31.3%) |
| Seronegative RA only | 103 | 59 (57.3%) | 38 (36.9%) | 0 (0.0%) | 0 (0.0%) | 23 (22.3%) |
| Seropositive RA only | 147 | 99 (67.3%) | 74 (50.5%) | 118 (80.3%) | 133 (90.5%) | 30 (20.4%) |
| Gout only | 28 | 20 (71.4%) | 14 (50.0%) | 1 (3.6%) | 0 (0.0%) | 17 (60.7%) |
| PMR only | 28 | 21 (75.0%) | 15 (54.0%) | 0 (0.0%) | 2 (7.1%) | 4 (14.3%) |
| CPPD+RA | 53 | 28 (52.8%) | 20 (37.7%) | 7 (13.2%) | 6 (11.3%) | 17 (32.1%) |
anti-CCP abs, anti-cyclic citrullinated peptide antibodies; CPPD, calcium pyrophosphate deposition disease; CRP, C reactive protein; ESR, erythrocyte sedimentation rate; PMR, polymyalgia rheumatica; RA, rheumatoid arthritis; RF, rheumatoid factor.
Among patients with more than one diagnosis, there were 53 patients with CPPD plus RA, 8 of whom were seropositive (15.1%) and 45 seronegative (84.9%). These only differ slightly when using the normal cut-offs for RF (28 U/L) and ACPA (<40 U/L) in our hospital: one more patient is seropositive and one less is seronegative. The mean disease duration was 0.5±1.9 years in patients with CPPD, 6.4±8.3 years in seronegative RA, 8.3±9.6 years in seropositive RA, 1.1±2.8 years in patients with gout and 0.6±1.1 years in those with PMR. The mean symptom duration at presentation was ≤1 year in all patients, except for gout (table 1).
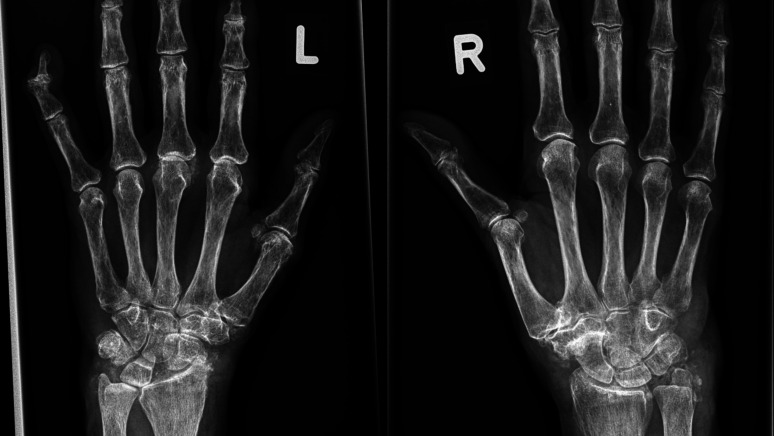
CC in at least one of the examined joints was detected in 90.1% of patients with CPPD only. A typical example is shown in figure 3. The prevalence of CC was different in seronegative (32.3%) versus seropositive (16.6%) RA (p<0.001). In patients with CPPD plus RA, CC was detected in all cases. CC was most frequently detected in wrists: in 74.0% of patients with CPPD only and in 81.1% of patients with both, CPPD and RA. The prevalence of CC in knees could not be exactly determined, because only about 60% of symptomatic patients had undergone conventional radiography of these joints at the time of presentation. Nevertheless, CC was found second most frequently in knees: in 42.7% of patients with CPPD and in 45.3% of those with CPPD and RA. A total of 75 patients with only CPPD and 33 patients with CPPD and RA had a full set of X-rays (hand, wrist, knee and feet). In these patients with CPPD only, 56 had CC in the knees (74.7%), and 54 had CC in the wrists (72.0%). In patients with CPPD and RA, 24 had CC in knees and wrists (72.0%).
Figure 3.
X-ray of the left hand of a 68 year old patient with CPPD which shows CC in the 3rd MCP joint and OA in the 2nd and 3rd MCP joint. X-ray of the right hand of a 75 year old patient which shows CC in ulnocarpal joint and severe STT OA.
Most patients with RA and CPPD had reported hand pain (table 1). There was no significant difference between CPPD and RA regarding involvement of hands (p=0.41), feet (p=0.95) and knees (p=0.77) but almost in wrists (p=0.06).
Erosions were nearly equally prevalent in seronegative RA (27.2%) and patients with CPPD (25.2%) but patients with seropositive RA had more erosions than patients with CPPD: 44.2% versus 25.2%, respectively (p0.001). Osteoarthritis was diagnosed in a least 90% of patients regardless of diagnosis but the distribution pattern of OA in hands and wrists differed in patients with CPPD in comparison to those without CPPD (table 3).
Table 3.
Distribution of osteoarthritis in hand and wrists on radiographs
| Joint | CPPD only (n=131) |
Seronegative RA only (n=103) | Seropositive RA only (n=147) | CPPD+RA (n=53) | PMR only (n=28) | Gout only (n=28) |
| DIP | 80 (61.0%) | 72 (69.9%) | 82 (55.8%) | 39 (73.6%) | 23 (82.1%) | 16 (57.1%) |
| PIP | 20 (15.3%) | 46 (44.7%) | 55 (37.4%) | 24 (45.3%) | 8 (28.6%) | 4 (14.3%) |
| MCP1 | 17 (12.9%) | 14 (13.6%) | 24 (16.3%) | 4 (7.5%) | 5 (17.9%) | 5 (17.9%) |
| MCP 2 | 49 (37.4%) | 20 (29.4%) | 30 (20.4%) | 27 (50.9%) | 0 | 1 (3.6%) |
| MCP 3 | 60 (45.8%) | 17 (16.5%) | 33 (22.4%) | 31 (58.5%) | 1 (3.6%) | 0 |
| MCP 4 | 20 (15.3%) | 6 (5.8%) | 6 (4.1%) | 6 (11.3%) | 1 (3.6%) | 0 |
| MCP 5 | 11 (8.4%) | 0 | 15 (10.2%) | 6 (11.3%) | 0 | 0 |
| Radiocarpal | 37 (28.2%) | 9 (8.7%) | 27 (18.4%) | 10 (18.9%) | 3 (10.7%) | 2 (7.1%) |
| Distal radioulnar | 17 (12.9%) | 6 (5.8%) | 18 (12.2%) | 4 (7.5%) | 0 | 0 |
| Scaphotrapeziotrapezoid joint (STT) | 66 (50.4%) | 29 (28.2%) | 51 (34.7%) | 33 (62.3%) | 7 (25.0%) | 5 (17.9%) |
| Mediocarpal | 11 (8.4%) | 3 (2.9%) | 7 (4.8%) | 2 (3.8%) | 2 (7.1%) | 0 |
| Carpometacarpal (CMC) | 94 (71.8%) | 77 (74.8%) | 94 (63.9%) | 43 (81.1%) | 20 (71.4%) | 17 (60.7%) |
| None | 8 (6.1%) | 14 (13.6%) | 29 (19.7%) | 2 (7.5%) | 0 | 9 (32.1%) |
CPPD, calcium pyrophosphate crystal deposition disease; PMR, polymymalgia rheumatica; RA, rheumatoid arthritis.
The frequency of involvement of the second and third metacarpophalangeal (MCP) joint and of the scaphotrapeziotrapezoid (STT) joint differed between patients with CPPD and without CPPD.
CC was present in more than one joint in 72.1% of patients with CPPD, 9.6% in seropositive RA and 18.7% in seronegative RA. The majority of patients with CPPD (91.6%) had pain in more than one joint: 48.9% presented with oligoarticular and 42.7% with polyarticular symptoms. An overview of the number of swollen and tender joints is given in table 4.
Table 4.
Disease activity: number of tender and swollen joints for each diagnosis
| Diagnosis | CPPD only (n=131) | Seronegative RA only (n=103) | Seropositive RA only (n=147) | CPPD+RA (n=53) | Gout only (n=28) | PMR only (n=28) | |
| Number of tender joints | 0 | 6 (4.6%) | 0 | 8 (5.4%) | 2 (3.4%) | 0 | 4 (14.3%) |
| 1 | 10 (7.6%) | 9 (8.7%) | 6 (4.1%) | 2 (3.4%) | 5 (17.9%) | 0 | |
| 2–4 | 32 (24.4%) | 21 (20.4%) | 34 (23.1%) | 12 (22.6%) | 10 (35.7%) | 6 (21.4%) | |
| ≥5 | 79 (60.3%) | 70 (68.0%) | 96 (65.3%) | 34 (64.2%) | 11 (39.2%) | 17 (60.7%) | |
| no data | 4 (3.1%) | 3 (2.9%) | 3 (2.0%) | 3 (5.7%) | 1 (3.6%) | 1 (3.6%) | |
| Number of swollen joints | 0 | 3 (2.3%) | 20 (19.4%) | 20 (13.6%) | 2 (3.4%) | 6 (21.4%) | 17 (60.7%) |
| 1 | 30 (22.9%) | 17 (16.6%) | 11 (7.5%) | 4 (7.5%) | 8 (28.6%) | 4 (14.3%) | |
| 2–4 | 56 (42.7%) | 31 (30.1%) | 50 (34.0%) | 29 (54.7%) | 8 (28.6%) | 5 (17.9%) | |
| ≥5 | 35 (26.7%) | 29 (28.2%) | 55 (37.4%) | 15 (28.3%) | 5 (17.9%) | 2 (7.1%) | |
| no data | 7 (5.3%) | 6 (5.5%) | 11 (7.5%) | 3 (5.7%) | 1 (3.6%) | 0 |
CPPD, calcium pyrophosphate crystal deposition disease; PMR, polymymalgia rheumatica; RA, rheumatoid arthritis.
Morning stiffness ≥30 min was observed in about half of the patients with CPPD, and in 63.3% of seropositive and 69.9% of seronegative RA patients, respectively. There were some missing data in seronegative (7.8%) and seropositive (8.2%) RA patients, while no data were missing in patients with CPPD.
The onset of symptoms was significantly different in CPPD versus patients with RA (p<0.001): while 67.9% of patients with CPPD reported acute attacks, this was only the case in 28.2% of patients with seronegative and in 25.9% with seropositive RA (table 5).
Table 5.
Onset of symptoms and symmetry of arthritis
| Diagnosis | N | Sudden onset | Slow onset | Onset unclear | Symmetry of arthritis clinically | Symmetry of CC radiologically |
| CPPD only | 131 | 89 (67.9%) | 31 (23.7%) | 11 (8.4%) | 91 (69.5 %) | 79 (60.3%) |
| Seronegative RA only | 103 | 29 (28.2%) | 33 (32.0%) | 41 (39.8%) | 88 (85.4%) | 1 (1.0%) |
| Seropositive RA only | 147 | 38 (25.9%) | 49 (33.3%) | 60 (40.9%) | 120 (81.6%) | 4 (2.7%) |
| CPPD+RA | 53 | 19 (35.8 %) | 25 (47.2%) | 9 (17.0%) | 44 (83.0%) | 35 (66.0%) |
| Gout only | 28 | 19 (67.9%) | 7 (25.0%) | 2 (7.1%) | 17 (60.7%) | 1 (3.6%) |
| PMR only | 28 | 10 (35.7%) | 12 (42.9%) | 6 (21.4%) | 25 (89.3%) | 0 (0.0%) |
Definition of symmetry: at least 50% of the symptomatic joints had to be affected on both sides.
CC, chondrocalcinosis; CPPD, calcium pyrophosphate deposition disease; PMR, polymyalgia rheumatica; RA, rheumatoid arthritis.
Symmetric arthritis was observed in all groups, but symmetric arthritis was more frequent in RA than in patients with CPPD (p=0.007), with no significant difference between seronegative and seropositive RA (p=0.86) patients (table 5). Symmetric CC was found in 60.3% of patients with CPPD only and in 66.0% of patients with CPPD plus RA (table 5).
After discharge patients with CPPD were treated significantly more often with colchicine than patients with RA (p<0.001) but not more often with hydroxychloroquine (p=0.306), while methotrexate was used significantly more often in RA than in CPPD (p<0.001, table 1). Data were missing in 12 seropositive (8.2%) and 8 seronegative (7.8%) RA patients.
Discussion
In this study based on a large real life data set using diagnoses made by senior rheumatologists in a tertiary care centre we found clinically relevant similarities but also some important differences in patients with CPPD and RA. However, there is no clinical feature that can precisely differentiate between these diseases and SF is very often not available to detect specific crystals. Nevertheless, and of clinical importance, co-occurrence of CPPD, CC and RA was frequently diagnosed. Indeed, more than every fourth patient primarily diagnosed with seronegative RA fulfilled the diagnostic criteria for CPPD and that was clearly different in patients with seropositive RA, gout and PMR. In addition, for the first time, we show a significant difference between seropositive and seronegative RA in the prevalence of CC on hand radiographs.
Overall CPPD was diagnosed about 9 years later than RA but the onset of CPPD did not differ between patients with and without RA suggesting an independent development of the two. However, patients with both, CPPD and RA, had developed RA about 10 years later as compared with patients with RA only. Before we discuss the issue of ascertainment which is of course critical here we review some clinical data of our patient groups for comparison with earlier studies.
Confirming previous data7 12 we also found that age is a major risk factor for CPPD. In addition, our study clearly shows that women are diagnosed with CPPD more often than men, and this was also true for the other diagnoses showing female preponderance if they had CPPD, too (table 6).
Table 6.
Diagnoses differentiated for patients’ sex
| Diagnosis | N | Men | Women |
| CPPD only | 131 | 30 (22.9%) | 101 (77.1%) |
| Seronegative RA only | 103 | 27 (26.2%) | 76 (73.8%) |
| Seropositive RA only | 147 | 43 (29.3%) | 104 (70.7%) |
| CPPD+RA | 53 | 10 (18.9%) | 43 (81.1%) |
| CPPD+seronegative RA | 44 | 9 (20.5%) | 35 (79.6%) |
| CPPD+seropositive RA | 9 | 1 (11.1%) | 8 (88.9%) |
| Gout only | 28 | 20 (71.4%) | 8 (28.6%) |
| PMR only | 28 | 16 (57.1%) | 12 (42.9%) |
CPPD, calcium pyrophosphate deposition disease; PMR, polymyalgia rheumatica; RA, rheumatoid arthritis.
Since females were on average only 2 years older than men we do not think that this can sufficiently explain the different prevalence of CPPD in both sexes as proposed by Abhishek.7 While patients with CPPD had a sudden onset more frequently than patients with RA (68% vs 27%, respectively), symmetry of arthritis and clinical involvement of hands and wrists was not a major differentiating factor. The rate of morning stiffness >30 min was also not so much different (50% vs 64% in RA). CPPD often affected more than one joint and patients frequently had elevated CRP levels suggesting that CPPD may be a systematic disease, as proposed by Abhishek.7 The radiological distribution of CC was similar but, expectedly, patients with seropositive RA had more often erosions. Elevated acute phase parameters did also not differentiate between diseases (table 2). Patients with CPPD only and patients with CPPD and RA had more often OA in the second and third MCP joint as well as in the STT joint than patients without CPPD (table 3). This is consistent with data from other studies and it is also a criterion in the new 2022 classification criteria for CPPD.17 23 30
Our results are largely consistent with earlier data showing that CC was most frequently found in wrists, second most in knees and third most in hands.6 Women had more often CPPD than men and 41.2% of patients had a polyarticular and 35.3% an oligoarticular involvement. In another study, the wrist was also most often affected by CC and that was often seen bilaterally.13 The age of onset of CPPD was also later than RA in three other studies with a range of 9–13 years.31–33
CPPD crystals were found in 25.8%31 and in 19.4%34 of patients with RA but no differences between seropositive and seronegative RA were searched for.31 34 The situation with CPPD and RA is actually similar for gout and RA—a comorbidity for which an increased prevalence has been reported35 and a considerable number of patients with RA were shown to display periarticular monosodium urate crystal deposits.36
The strength of this work, even though it is retrospective, is that it is a large study population with consecutive patients presenting to a tertiary hospital without selection bias. CPPD was diagnosed by experienced senior rheumatologists and radiologists based on clinical symptoms, laboratory data and radiological findings as diagnosed by experienced senior musculoskeletal radiologists. Since all patients had more or less acute symptoms leading to their presentation at the hospital a major influence of therapy is unlikely. However, a possible limitation is the ‘natural’ difference of disease duration between patients with RA and CPPD with the former having had the disease for already 7 years. Another limitation is that we did not have many patients with enough SF to detect crystals.
Furthermore, one can ask whether we have used the best imaging method to detect crystals, since promising studies about the use of ultrasound37 in the identification of CPPD have been published while the significance of DECT seems less clear.38 39 In this study we have not systematically used ultrasound to detect crystals in hand or feet due to lack of standardisation. However, we do routinely use ultrasound for larger joints to detect the double contour sign.37 Of interest, the joints radiographically examined in this study are consistent with recent recommendations which joints to examine in search of CPPD.40
Of course, one explanation for the co-occurrence of RA and CPPD is misdiagnosis implying that CPPD was previously underdiagnosed in favour of seronegative RA. The higher prevalence of CC in seronegative versus seropositive disease may be considered supportive of that. Thus, we cannot exclude that. In any case, our data confirm the clinical similarity of CPPD and RA. On the other hand, as recently shown in two early arthritis cohorts,41 seronegative RA may be underdiagnosed because the 2010 ACR/EULAR classification criteria for RA only detected 51% of patients with seronegative RA in an early disease stage. Another problem with the initial diagnosis of seronegative RA is that it often cannot be confirmed over time. Again, in an earlier Finnish study attention has been drawn to this issue by reporting that, after a 10-year follow-up, the diagnosis of seronegative RA had to be revised in the majority of patients.42 Thus, it is important to regularly review the diagnosis of seronegative RA in order to treat patients optimally. This is important since the management of RA and CPPD differs.
The 2010 ACR/EULAR classification criteria for RA also stress, next to the presence of an erosion which, in our study, was more often found in seropositive RA than in CPPD (48% vs 30%), that other inflammatory rheumatic diseases should have been excluded. In a clinical situation with a symmetric arthritis of the hands and or wrists and a radiograph showing CC this is clearly not so easy—especially if the onset was not sudden and did not have the character of an acute attack.
The differences in treatment found in our study are consistent with current strategies in the management of CPPD but there is no evidence for the treatment of this ‘common but neglected form of arthritis43 ’ by commonly administered agents such as NSAIDs, corticosteroids and colchicine. Nevertheless, there is limited evidence for an effect of the anti-interleukin (IL)-1 bDMARD anakinra.44 Although the effect of an anti-IL-1 agent may argue in favour of an autoinflammatory disease, it is stressed that anakinra is also approved for the treatment of RA.45
In summary, our data show for the first time that the comorbidity of CPPD and the prevalence of CC is more frequent in seronegative than in seropositive RA. The clinical similarity between these two diseases makes correct diagnoses difficult. More studies with innovative imaging techniques may change this. Prospective cohort studies and randomised clinical trials are urgently needed to provide an evidence basis for the management of this rather prevalent rheumatic disease in the elderly. The recent framework paper by the Outcome Measures in Rheumatology working group to develop core sets for short-term and long-term studies in CPPD46 may well be a good step forward in this direction.
Footnotes
Contributors: MK, XB, ST and JB contributed to the design, to the analysis of theresults and to the writing of the manuscript. JB is the guarantor of this work.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study involves human participants and was approved by Ethik-Kommission der Medizinischen Fakultät der Ruhr-Universität Bochum Germany (reference number: 20-6956-BR). It is a restrospective anonymised study which had no effect on the patients.
References
- 1.Kohn NN, Hughes RE, McCarty DJ. The significance of calcium phosphate crystals in the synovial fluid of arthritic patients: the "pseudogout syndrome". II. Identification of crystals. Ann Intern Med 1962;56:738–37. 10.7326/0003-4819-56-5-738 [DOI] [PubMed] [Google Scholar]
- 2.McCarty DJ. Calcium pyrophosphate dihydrate crystal deposition disease: Nomenclature and diagnostic criteria. Ann Intern Med 1977;87:240–2. 10.7326/0003-4819-87-2-240 [DOI] [PubMed] [Google Scholar]
- 3.Zhang W, Doherty M, Bardin T, et al. European League against rheumatism recommendations for calcium pyrophosphate deposition. Part I: terminology and diagnosis. Ann Rheum Dis 2011;70:563–70. 10.1136/ard.2010.139105 [DOI] [PubMed] [Google Scholar]
- 4.Abhishek A, Doherty M. Epidemiology of calcium pyrophosphate crystal arthritis and basic calcium phosphate crystal arthropathy. Rheum Dis Clin North Am 2014;40:177–91. 10.1016/j.rdc.2014.01.002 [DOI] [PubMed] [Google Scholar]
- 5.Stensby JD, Lawrence DA, Patrie JT, et al. Prevalence of asymptomatic chondrocalcinosis in the pelvis. Skeletal Radiol 2016;45:949–54. 10.1007/s00256-016-2376-9 [DOI] [PubMed] [Google Scholar]
- 6.Paalanen K, Rannio K, Rannio T, et al. Prevalence of calcium pyrophosphate deposition disease in a cohort of patients diagnosed with seronegative rheumatoid arthritis. Clin Exp Rheumatol 2020;38:99–106. [PubMed] [Google Scholar]
- 7.Abhishek A. Calcium pyrophosphate deposition disease: a review of epidemiologic findings. Curr Opin Rheumatol 2016;28:133–9. 10.1097/BOR.0000000000000246 [DOI] [PubMed] [Google Scholar]
- 8.Mitton-Fitzgerald E, Gohr CM, Bettendorf B, et al. The role of ANK in calcium pyrophosphate deposition disease. Curr Rheumatol Rep 2016;18:25. 10.1007/s11926-016-0574-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Salaffi F, De Angelis R, Grassi W, et al. Prevalence of musculoskeletal conditions in an Italian population sample: results of a regional community-based study. I. The mapping study. Clin Exp Rheumatol 2005;23:819–28. [PubMed] [Google Scholar]
- 10.Ramonda R, Musacchio E, Perissinotto E, et al. Prevalence of chondrocalcinosis in Italian subjects from northeastern Italy. The Pro.V.A. (PROgetto Veneto Anziani) study. Clin Exp Rheumatol 2009;27:981–4. [PubMed] [Google Scholar]
- 11.Felson DT, Anderson JJ, Naimark A, et al. The prevalence of chondrocalcinosis in the elderly and its association with knee osteoarthritis: the Framingham study. J Rheumatol 1989;16:1241–5. [PubMed] [Google Scholar]
- 12.Neame RL, Carr AJ, Muir K, et al. Uk community prevalence of knee chondrocalcinosis: evidence that correlation with osteoarthritis is through a shared association with osteophyte. Ann Rheum Dis 2003;62:513–8. 10.1136/ard.62.6.513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Abhishek A, Doherty S, Maciewicz R, et al. Chondrocalcinosis is common in the absence of knee involvement. Arthritis Res Ther 2012;14:R205. 10.1186/ar4043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sanmarti R, Kanterewicz E, Pladevall M, et al. Analysis of the association between chondrocalcinosis and osteoarthritis: a community based study. Ann Rheum Dis 1996;55:30–3. 10.1136/ard.55.1.30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Al-Arfaj AS. The relationship between chondrocalcinosis and osteoarthritis in Saudi Arabia. Clin Rheumatol 2002;21:493–6. 10.1007/s100670200121 [DOI] [PubMed] [Google Scholar]
- 16.Bourqui M, Vischer TL, Stasse P, et al. Pyrophosphate arthropathy in the carpal and metacarpophalangeal joints. Ann Rheum Dis 1983;42:626–30. 10.1136/ard.42.6.626 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Peter A, Simmen BR, Brühlmann P, et al. Osteoarthritis of the scaphoidtrapezium joint: an early sign of calcium pyrophosphate dihydrate disease. Clin Rheumatol 2001;20:20–4. 10.1007/PL00011183 [DOI] [PubMed] [Google Scholar]
- 18.Abhishek A, Doherty S, Maciewicz R, et al. Evidence of a systemic predisposition to chondrocalcinosis and association between chondrocalcinosis and osteoarthritis at distant joints: a cross-sectional study. Arthritis Care Res 2013;65:1052–8. 10.1002/acr.21952 [DOI] [PubMed] [Google Scholar]
- 19.Moshrif A, Laredo JD, Bassiouni H, et al. Spinal involvement with calcium pyrophosphate deposition disease in an academic rheumatology center: a series of 37 patients. Semin Arthritis Rheum 2019;48:1113–26. 10.1016/j.semarthrit.2018.10.009 [DOI] [PubMed] [Google Scholar]
- 20.Tedeschi SK. Issues in CPPD Nomenclature and classification. Curr Rheumatol Rep 2019;21:49. 10.1007/s11926-019-0847-4 [DOI] [PubMed] [Google Scholar]
- 21.Matsumura M, Hara S. Images in clinical medicine. Crowned dens syndrome. N Engl J Med 2012;367:e34. 10.1056/NEJMicm1100764 [DOI] [PubMed] [Google Scholar]
- 22.Miksanek J, Rosenthal AK. Imaging of calcium pyrophosphate deposition disease. Curr Rheumatol Rep 2015;17:20. 10.1007/s11926-015-0496-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tedeschi SK, Becce F, Pascart T, et al. Imaging features of calcium pyrophosphate deposition (CPPD) disease: consensus definitions from an international multidisciplinary Working group. Arthritis Care Res 2022. 10.1002/acr.24898. [Epub ahead of print: 19 Apr 2022]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rosenthal AK, Ryan LM. Chapter 117 Calcium Pyrophosphate Crystal Deposition Disease, Pseudogout, and Articular Chondrocalcinosis. In: Daniel JMcCarty, ed. Arthritis and allied conditions: a textbook of rheumatology. Philadelphia: Lea and Febiger, 1993: 1337–50. [Google Scholar]
- 25.Filippou G, Filippucci E, Mandl P, et al. A critical review of the available evidence on the diagnosis and clinical features of CPPD: do we really need imaging? Clin Rheumatol 2021;40:2581–92. 10.1007/s10067-020-05516-3 [DOI] [PubMed] [Google Scholar]
- 26.Wu Y, Chen K, Terkeltaub R. Systematic review and quality analysis of emerging diagnostic measures for calcium pyrophosphate crystal deposition disease. RMD Open 2016;2:e000339. 10.1136/rmdopen-2016-000339 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aletaha D, Neogi T, Silman AJ, et al. 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League against rheumatism collaborative initiative. Arthritis Rheum 2010;62:2569–81. 10.1002/art.27584 [DOI] [PubMed] [Google Scholar]
- 28.Neogi T, Jansen TLTA, Dalbeth N, et al. 2015 gout classification criteria: an American College of Rheumatology/European League against rheumatism collaborative initiative. Ann Rheum Dis 2015;74:1789–98. 10.1136/annrheumdis-2015-208237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dasgupta B, Cimmino MA, Maradit-Kremers H, et al. 2012 provisional classification criteria for polymyalgia rheumatica: a European League against Rheumatism/American College of rheumatology collaborative initiative. Ann Rheum Dis 2012;71:484–92. 10.1136/annrheumdis-2011-200329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sanmarti R, Kanterewicz E, Pladevall M, et al. Analysis of the association between chondrocalcinosis and osteoarthritis: a community based study. Ann Rheum Dis 1996;55:30–3. 10.1136/ard.55.1.30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gerster JC, Varisco PA, Kern J, et al. CPPD crystal deposition disease in patients with rheumatoid arthritis. Clin Rheumatol 2006;25:468–9. 10.1007/s10067-005-0082-4 [DOI] [PubMed] [Google Scholar]
- 32.Sabchyshyn V, Konon I, Ryan LM, et al. Concurrence of rheumatoid arthritis and calcium pyrophosphate deposition disease: a case collection and review of the literature. Semin Arthritis Rheum 2018;48:9–11. 10.1016/j.semarthrit.2017.11.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Theiler G, Quehenberger F, Rainer F, et al. The detection of calcium pyrophosphate crystals in the synovial fluid of patients with rheumatoid arthritis using the cytospin technique: prevalence and clinical correlation. Rheumatol Int 2014;34:137–9. 10.1007/s00296-012-2608-9 [DOI] [PubMed] [Google Scholar]
- 34.Galozzi P, Oliviero F, Frallonardo P, et al. The prevalence of monosodium urate and calcium pyrophosphate crystals in synovial fluid from wrist and finger joints. Rheumatol Int 2016;36:443–6. 10.1007/s00296-015-3376-0 [DOI] [PubMed] [Google Scholar]
- 35.Merdler-Rabinowicz R, Tiosano S, Comaneshter D, et al. Comorbidity of gout and rheumatoid arthritis in a large population database. Clin Rheumatol 2017;36:657–60. 10.1007/s10067-016-3477-5 [DOI] [PubMed] [Google Scholar]
- 36.Petsch C, Araujo EG, Englbrecht M, et al. Prevalence of monosodium urate deposits in a population of rheumatoid arthritis patients with hyperuricemia. Semin Arthritis Rheum 2016;45:663–8. 10.1016/j.semarthrit.2015.11.014 [DOI] [PubMed] [Google Scholar]
- 37.Filippou G, Scirè CA, Adinolfi A, et al. Identification of calcium pyrophosphate deposition disease (CPPD) by ultrasound: reliability of the OMERACT definitions in an extended set of joints-an international multiobserver study by the OMERACT calcium pyrophosphate deposition disease ultrasound Subtask force. Ann Rheum Dis 2018;77:annrheumdis-2017-212542–9. 10.1136/annrheumdis-2017-212542 [DOI] [PubMed] [Google Scholar]
- 38.Ziegeler K, Hermann S, Hermann KGA, et al. Dual-Energy CT in the differentiation of crystal depositions of the wrist: does it have added value? Skeletal Radiol 2020;49:707–13. 10.1007/s00256-019-03343-5 [DOI] [PubMed] [Google Scholar]
- 39.Budzik J-F, Marzin C, Legrand J, et al. Can dual-energy computed tomography be used to identify early calcium crystal deposition in the knees of patients with calcium pyrophosphate deposition? Arthritis Rheumatol 2021;73:687–92. 10.1002/art.41569 [DOI] [PubMed] [Google Scholar]
- 40.Tedeschi SK, Pascart T, Latourte A, et al. Identifying potential classification criteria for calcium pyrophosphate deposition disease (CPPD): item generation and item reduction. Arthritis Care Res. 10.1002/acr.24619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Boeters DM, Gaujoux-Viala C, Constantin A, et al. The 2010 ACR/EULAR criteria are not sufficiently accurate in the early identification of autoantibody-negative rheumatoid arthritis: results from the Leiden-EAC and ESPOIR cohorts. Semin Arthritis Rheum 2017;47:170–4. 10.1016/j.semarthrit.2017.04.009 [DOI] [PubMed] [Google Scholar]
- 42.Paalanen K, Rannio K, Rannio T, et al. Does early seronegative arthritis develop into rheumatoid arthritis? A 10-year observational study. Clin Exp Rheumatol 2019;37:37–43. [PubMed] [Google Scholar]
- 43.Parperis K, Papachristodoulou E, Kakoullis L, et al. Management of calcium pyrophosphate crystal deposition disease: a systematic review. Semin Arthritis Rheum 2021;51:84–94. 10.1016/j.semarthrit.2020.10.005 [DOI] [PubMed] [Google Scholar]
- 44.Cipolletta E, Di Matteo A, Scanu A, et al. Biologics in the treatment of calcium pyrophosphate deposition disease: a systematic literature review. Clin Exp Rheumatol 2020;38:447–8. 10.1136/annrheumdis-2020-eular.1080 [DOI] [PubMed] [Google Scholar]
- 45.Thaler K, Chandiramani DV, Hansen RA, et al. Efficacy and safety of anakinra for the treatment of rheumatoid arthritis: an update of the Oregon drug effectiveness review project. Biologics 2009;3:485–98. 10.2147/btt.2009.3755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cai K, Fuller A, Zhang Y, et al. Towards development of core domain sets for short term and long term studies of calcium pyrophosphate crystal deposition (CPPD) disease: a framework paper by the OMERACT CPPD Working group. Semin Arthritis Rheum 2021;51:946–50. 10.1016/j.semarthrit.2021.04.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplementary information.