Abstract
Background
Sentinel lymph node biopsy (SLNB) is the standard axillary staging approach for early breast cancer with clinically negative axillary involvement. Adequate SLNB should include the removal of not only radioactive tracer-labeled lymph nodes (hot nodes or SLNs) but also suspicious unlabeled nodes (non-hot nodes or non-SLNs). However, the biopsy of non-hot nodes is highly dependent on the surgeons' experiences. This article aims to facilitate the surgeon's decision making by elucidating parameters that correlate with non-hot node metastasis.
Methods
From 2013 to 2016, clinically node-negative (cN0) breast cancer patients receiving axillary SLNB using single Tc-99m tracer method at our institute were recruited. Patients were excluded if they had received prior neoadjuvant chemotherapy. Among them, cases that have at least one non-isotope-hot node biopsied were retrospectively reviewed with a particular focus on patients with pathologically negative isotope-hot SLNs. The correlation of clinicopathological data with metastasis to axillary lymph nodes and sentinel lymph nodes was analyzed with the Chi-squared test, Fisher's exact test, and multivariate logistic regression. Receiver operating curve (ROC) was applied for continuous variables that predicted non-hot node metastasis; relapse-free survival (RFS) and locoregional relapse-free survival (LRRFS) were compared by Kaplan–Meier analysis.
Results
In 632 isotope-hot SLN negative patients, T stage showed a correlation with non-isotope-hot SLN metastasis (p = 0.035, odds ratio (OR) 9.65). Tumors larger than 2.5 cm best predict non-isotope-hot SLN metastasis (area under curve (AUC) = 0.71). With a median follow up of 41.80 months, locoregional relapse-free survival was significantly worse in cases with non-hot node metastasis (66.2% vs. 69.0%, p = 0.001).
Conclusion
In the setting of SLNB using single radioisotope tracer, non-hot node metastasis in cases with negative hot SLN still carries a higher locoregional recurrence rate (13.3%). For early breast cancer larger than 2.5 cm, removal of suspicious non-hot nodes should be included for a precision therapy.
Keywords: Breast cancer, Sentinel lymph node, Sentinel lymph node biopsy, Lymphovascular invasion, Tumor size
At a glance commentary
Scientific background on the subject
T2 tumor compared with T1mi/T1a has greater likelihood (OR 9.65, p=0.028) of axillary non-hot node metastasis in cN0 cases with cancer-free non-hot nodesretrieved by SLNB using single Tc-99m tracer. Tumor size > 2.5cm (AUC 0.71) indicates greater chance of metastasis in non-hot nodes, which correlates withhigher locoregional metastasis (13.3% vs 1.8%, p=0.035) in our cohort.
What this study adds to the field
Clinical parameters such as tumor size, or tumor location to a lesser extent, may indicate careful palpation of the axillary bed during SLNB, to include the sampling of suspicious non-hot nodes so as to avoid under-staging of nodal status by missing non-hot node metastasis.
Axillary staging in breast cancer plays a pivotal role in both early and late outcome assessment and determines the appropriate adjuvant treatment strategy. For estrogen receptor-positive T1 tumors, the risk of distant recurrence at 20 years after 5 years of endocrine therapy increased from 13% in N0 to above 20% with the presence of axillary lymph node metastasis. In the T2 stage, distant recurrence risk also escalated from 19% to above 26% [1]. In triple-negative breast cancer and HER2-enriched breast cancer, lymph node status is also a crucial factor influencing the choice of adjuvant chemotherapy and target therapies [2,3]. Preoperative clinical assessment, in combination with sentinel lymph node biopsy (SLNB), is the current standard of axillary staging for clinically node-negative (cN0) patients, as it provides an accurate determination of nodal status and, at the same time, avoids arm lymphedema caused by extensive axillary dissection. However, missed detection of nodal metastasis risks understaging and precludes adequate adjuvant treatment.
Frequently used tracer options for SLNB fall into two categories: blue dye and technetium-99m (Tc-99m)-labeled sulfur colloid. Isosulfan blue, patent blue, and methylene blue are the blue dyes usually used. Although it has rapid transmission through the lymphatic channel and is visible to surgeons, the risk of anaphylaxis (approximately 1–2%) [4,5], hives, skin necrosis, and discoloration makes blue dye a less favorable tracer for some surgeons. Radioisotope sulfur colloid tracer has low radioactivity, a short half-life (about 6 h), and requires a gamma-detecting probe to identify sentinel lymph nodes (SLN). With equally high accuracy of 97.5% and providing lymphoscintigraphy for lymphatic mapping [6], Tc-99m colloid has long been the preferred tracer in our institute. In a systemic review by He et al. [7], although the combination of blue dye and radioisotope show a higher SLN identification rate than radioisotope alone (OR = 2.03), the key parameter, false-negative rate, was not improved (OR = 0.76).
On the contrary, intraoperative palpation of the axillary bed for clinically suspicious SLNs has been shown to be an important technique to reduce the false-negative rate of SLNB [8]. Removal of suspicious non-radioactive or non-blue firm nodes during axillary SLNB is a mandatory part of a successful SLNB procedure. Carmen et al. stressed that systemic axillary palpation through a generous axillary incision reduces false-negative rate [8]. Ozkurt et al. [9] demonstrated in their prospective study that, when performed by dedicated breast surgeons, intraoperative pathological assessment of lymph nodes could be safely omitted if SLN detection was combined with palpation. In their study, the revised accuracy of intraoperative nodal palpation (INP) could reach 87%; meanwhile, in the INP negative group (n = 217), only 0.5% (n = 1) needed complete axillary lymph node dissection after final pathology analysis and multidisciplinary tumor board discussion. The role of intraoperative palpation during SLNB is appropriately emphasized in this study.
Earlier literature about SLNB reported that its false-negative rate correlates closely with the experience of the surgeon. Surgeons with a surgical volume of more than 6 SLNBs per month had a success rate of 97.810.44%, while surgeons performing three to six cases per month had a lower success rate of 88.73%6.36% [10]. The learning curve after 40 SLNBs allows breast surgeons to reach a satisfactory SLN localization rate and acceptable false-negative rate [11]. However, successful identification of at least 20 cases with positive axillary nodes should be documented to calculate a trustworthy false-negative rate [12].
Intraoperative palpation and meticulous evaluation of the preoperative cancer image and biology contribute to a surgeon's perspective of the SLNB procedure. The integration of preoperative staging with axillary ultrasound examination and ultrasound-guided aspiration cytology precludes SLNB in node-positive cases with insignificant axillary lymph node enlargement by physical examination [13]. These factors stand out from the method of tracer used or the location (intradermal, subdermal, subareolar, or peritumoral) of tracer injection [14] as a more essential element for a meaningful reduction of false-negative rate in SLNB.
In our institute, breast ultrasound is routinely performed before surgery. Clinical axillary nodal status is assessed by ultrasound and fine-needle aspiration cytology when indicated. Only patients determined “cN0” were candidates for SLNB in this adjuvant setting. When single radioisotope tracer method is used, “standard SLNB” includes the removal of both radioisotope containing nodes (hot nodes or SLNs) [15] and probably abnormal nodes (non-hot nodes, or non-SLNs) [16,17]. Biopsy of hot nodes alone is the standard procedure in conventional SLNB. In addition, we looked for the gritty suspicious nodes as the “probably abnormal” nodes encountered during careful palpation of the axilla. The proportion of non-hot nodes sampled reflects an experience-based judgement by the surgeon that may help to reduce the false-negative rate in SLNB.
In this study, we retrospectively reviewed the non-hot node status in cN0 patients with negative hot SLN. Factors that warrant more careful exploration of non-hot nodes were determined. Clinicopathological factors that correlate with non-hot node metastasis were analyzed to substantiate the subjective “surgeons’ experience” factor and facilitate experience building of new hands.
Methods
From 2013 to 2016, early breast cancer patients in Chang Gung Memorial Hospital (CGMH) receiving primary surgical treatment including SLNB were retrospectively recruited for qualified patients. Clinically negative nodal status (cN0) was defined as no palpable axillary lymph node by physical examination, no ultrasound detection of eccentrically thickened cortex or loss of hilum, no computed tomography finding of lymph nodes with reduced long to short axis ratio, irregular appearance or thickened cortex, and absence of cytological evidence of nodal metastasis. Patients who were cN0 and received SLNB with excision of at least one non-isotope-hot nodes (non-SLNs) were selected. Patients were excluded if they had bilateral breast cancer, received neoadjuvant chemotherapy, or had previous breast or axillary surgery. Breast surgeons personally performed the whole breast and axillary ultrasound preoperatively for each patient. The type of breast surgery was determined according to the tumor extent and location. Adjuvant radiotherapy, chemotherapy, endocrine therapy, or target therapy was given according to tumor stage, biology, and clinical risks. Molecular subtype determination was based on immunohistochemical (IHC) stain results: luminal A (HR (hormone receptor) +, HER2-, low proliferation), luminal B (HR+, HER2-, high proliferation), HER2+ and HR+, HER2+ and HR-, and triple-negative (HR-, and HER2-).
SLNB was performed with the single Tc-99m colloid tracer method by injecting 0.5 mCi radiocolloid peritumorally or subdermally in the affected breast the same day before surgery or injecting 1.0 mCi one day before surgery. Lymphoscintigraphy was routinely performed right after injection, and a gamma-detecting probe was applied to guide SLN exploration in the axilla during surgery. Hot nodes including the node with top counts and the nodes that count more than 10% of the highest signal were biopsied first. Then the axillary bed was carefully examined for non-hot nodes with intraoperative palpation. Any firm, gritty, or exceptionally enlarged lymph nodes were biopsied and labeled as non-hot nodes either within or outside the SLN basin of the axilla.
All SLNs biopsied, including hot nodes and non-hot nodes, were sent to a pathologist for intraoperative frozen section examination. If nodal metastasis was reported under frozen section, complete dissection of axillary lymphatics was done. A permanent pathological examination of all nodes was reported one week after surgery. The final pathological status of all nodes removed was recorded. Nodal metastasis was determined positive if the size of invasive tumor in the lymph node was larger than 1 mm. For cases with nodal metastasis detected by SLNB, subsequent axillary dissection, regional nodal radiotherapy or systemic treatment will be performed according to physicians' choice based on current evidence and patients' preference. The clinicopathological data of these patients were also obtained in our retrospective analysis. Tumor location was defined as “the central portion” if it was located within a 1 cm distance from the nipple. “Strictly upper outer quadrant” referred to tumors located at the upper outer quadrant, excluding tumors situated at 12 o'clock and 9′o' clock for the right breast and tumors at 12 o'clock and 3 o'clock for the left breast. All clinical data including age, tumor size, T stage, laterality, tumor foci, breast and axillary surgical method, tumor subtype, IHC markers, lymphovascular invasion status, and location were recorded. For cases with negative hot nodes, the above factors were compared between groups with negative and positive non-hot nodes. Univariate analysis was performed using the Chi-squared test or Fisher's exact test when appropriate. Multivariate analysis was conducted to determine factors significantly related to lymph node metastasis, with p < 0.05 in the univariate analysis. The receiver operating curve (ROC) analysis was used to determine the cut-off for continuous variables. All cases were followed up till December 2018, and recurrence status was also compared between these groups. Kaplan–Meier method was applied to estimate survival, and the survival outcomes of different lymph node status in axillary lymph nodes and non-hot nodes were compared using the log-rank test.
Results
Between the years 2013 and 2016, a total of 2893 breast cancer patients were operated in our hospital. The percentage of cN0 patients was 62.29% (n = 1802). Our study included only unilateral cN0 breast cancer patients treated primarily with surgery and successfully mapped with Tc-99m as the single SLN tracer. Cases without a clear record of non-hot node status were excluded. A total of 750 patients met all the above criteria. Table 1 detailed the characteristics of our cN0 breast cancer patients (N = 750). Final pathological nodal status was positive (pN+) in 135 (18%) cases and negative (pN-) in 615 (82.0%) cases. SLNB alone was performed in 626 (83.5%) patients, axillary lymph node sampling (ALNS) was done following SLNB in 36 cases (4.8%), and 88 cases (11.4%) received axillary lymph node dissection (ALND) after SLNB. Multifocal breast cancer was found in 37 (4.9%) patients. Type of breast surgery included mastectomy (359 cases, 47.9%) and breast conservation surgery (391 cases, 52.1%). The proportion of breast cancer subtype by IHC was luminal A (183, 24.4%), luminal B (365, 48.7%), HER2+/HR+ (58, 7.7%), HER2+/HR- (57, 7.6%), and triple-negative (87, 11.6%) (Table 1). The median number of harvested lymph nodes in SLNB was 2 (range 1–10) for hot nodes, while it was 3 (range 1–13) for non-hot nodes.
Table 1.
Characteristics of clinically node negative patients undergoing SLNB with Tc-99m tracer from 2013 to 2016.
| Characteristic | Total N = 750 (%) | Characteristic | Total N = 750 (%) | Characteristic | Total N = 750 (%) |
|---|---|---|---|---|---|
| Age | Final Pathological Nodal Status | Injection Interval | |||
| ≤50 years old | 302 (40.3) | Negative | 615 (82.0) | Same day | 594 (79.2) |
| >50 years old | 448 (59.7) | Positive | 135 (18.0) | Overnight | 156 (20.8) |
| Year of Operation | Lymphovascular Invasion | ||||
| 2013 | 154 (20.5) | Tumor Size | Yes | 613 (81.7) | |
| 2014 | 169 (22.5) | T1mic/T1a | 89 (11.9) | No | 119 (15.9) |
| 2015 | 195 (26.0) | T1b | 63 (8.4) | Unknown | 18 (2.4) |
| 2016 | 232 (30.9) | T1c | 277 (36.9) | IHC Marker | |
| Laterality | T2 | 311 (41.5) | ER (+) | 596 (79.5) | |
| Left | 371 (49.5) | T3 | 10 (1.3) | PR (+) | 547 (72.9) |
| Right | 379 (50.5) | HER2 (+) | 120 (16.0) | ||
| Quadrant | Stage | Molecular Subtype (IHC) | |||
| Upper Outer | 267 (35.6) | IA | 382 (50.9) | Luminal A | 183 (24.4) |
| Upper Inner | 190 (25.3) | IB | 13 (1.7) | Luminal B | 365 (48.7) |
| Lower Inner | 71 (9.5) | IIA | 257 (34.3) | HER2(+) HR(+) | 58 (7.7) |
| Lower Outer | 146 (19.5) | IIB | 98 (13.1) | HER2 (+) HR (−) | 57 (7.6) |
| Central | 76 (10.1) | Triple Negative | 87 (11.6) | ||
| Strictly Upper Outer Quadranta | Axillary Surgical Method | Recurrence | |||
| Strictly UOQ | 247 (32.9) | SLNB | 626 (83.5) | None | 716 (95.5) |
| Others | 503 (67.1) | SLNB + ALNS | 36 (4.8) | Locoregional | 18 (2.4) |
| SLNB + ALND | 88 (11.4) | Distant | 16 (2.1) | ||
| Tumor Foci | Operative Method | Mortality | |||
| Unifocal | 713 (95.1) | Mastectomy | 359 (47.9) | None | 742 (98.2) |
| Multifocal | 37 (4.9) | Breast Conserving Surgery | 391 (52.1) | Yes | 8 (1.1) |
Abbreviations: SLNB: Sentinel lymph node biopsy; ALNS: Axillary lymph node sampling: removing <10 nodes from the axilla; ALND: Axillary lymph node dissection: removing >10 nodes from the axilla; IHC: immunohistochemical staining; HR: hormoae receptor.
Strictly Upper Outer Quadrant: Tumors located at the upper outer quadrant, excluding tumors locating at 12 o'clock, 9′o' clock for the right breast and 3 o'clock for the left breast.
In our cohort, clinical factors that correlated with lymph node metastasis (pN+) include lymphovascular invasion (p < 0.001) and T-stage (p < 0.001) (Table 2). The odds ratio (OR) of lymph node metastasis for lymphovascular invasion was 3.11 [ 95% confidence interval (CI) 1.98–4.86]. The odds ratio of lymph node metastasis for T1c and T2 was 4.27 (95% CI 1.51–12.09, p = 0.006) and 7.10 (95% CI 2.55–19.78, p < 0.001) respectively compared with T1mi/T1a. Total mastectomy showed a trend but no significant correlation with lymph node metastasis compared with breast conservation surgery. Tumor foci number or tumor location did not correlate with final pathological node-positive (pN+) (Table 2).
Table 2.
Factors correlated with nodal metastasis in clinically node negative (cN0) patients.
| Parameters |
Total N = 750 (%) |
LN (+) N = 135 (%) |
LN (−) N = 615 (%) |
p value |
Multivariate analysis |
|||
|---|---|---|---|---|---|---|---|---|
| Odds ratio | 95% CI | p value | ||||||
| Age (years) | ≤50 >50 |
302 (40.3) 448 (59.7) |
59 (43.7) 76 (56.3) |
243 (39.5) 372 (60.5) |
0.369 | |||
| Quadrant | Upper outer Upper inner Lower inner Lower outer Central |
267 (35.6) 190 (25.3) 71 (9.5) 146 (19.5) 76 (10.1) |
54 (40.0) 24 (17.8) 9 (6.7) 32 (23.7) 16 (11.9) |
213 (34.6) 166 (27.0) 62 (10.1) 114 (18.5) 60 (9.8) |
0.093 | – | ||
| Quadrant | Strictly UOQ Others |
247 (32.9) 503 (67.1) |
52 (38.5) 83 (61.5) |
195 (31.7) 420 (68.3) |
0.127 | – | ||
| Tumor foci | Unifocal Multifocal |
713 (95.1) 37 (4.9) |
126 (93.3) 9 (6.7) |
587 (95.4) 28 (4.6) |
0.304 | – | ||
| Injection interval | Same day Overnight |
594 (79.2) 156 (20.8) |
110 (81.5) 25 (18.5) |
484 (78.7) 131 (21.3) |
0.471 | – | ||
| Breast surgical method | BCS Mastectomy |
391 (52.1) 359 (47.9) |
58 (43.0) 77 (57.0) |
333 (54.1) 282 (45.9) |
0.019 | 1 1.28 |
0.85–1.93 | 0.234 |
| IHC markers | ER (+) ER (−) |
596 (79.5) 154 (20.5) |
109 (80.7) 26 (19.3) |
487 (79.2) 128 (20.8) |
0.686 | – | ||
| PR (+) PR (−) |
547 (72.9) 203 (27.1) |
103 (76.3) 32 (23.7) |
444 (72.2) 171 (27.8) |
0.331 | – | |||
| HER2 (+) HER2(−) |
120 (16.0) 630 (84.0) |
17 (12.6) 118 (87.4) |
103 (16.7) 512 (83.3) |
0.233 | – | |||
| Subtype by IHC | Luminal A Luminal B HER2(+)HR (+) HER2(+)HR (−) Triple Negative |
183 (24.4) 365 (48.7) 58 (7.7) 87 (11.6) 57 (7.6) |
35 (25.9) 65 (48.1) 12 (8.9) 18 (13.3) 5 (3.7) |
148 (24.1) 300 (48.8) 46 (7.5) 69 (11.2) 52 (8.5) |
0.384 | – | ||
| T stage | T1mi/T1a T1b T1c T2 T3 |
89 (11.9) 63 (8.4) 277 (36.9) 311 (41.5) 10 (1.3) |
3 (2.2) 1 (0.7) 43 (31.9) 88 (65.2) 0 |
86 (14.0) 62 (10.1) 234 (38.0) 223 (36.3) 10 (1.6) |
<0.0001 | 1 0.48 4.27 7.10 0.43 |
0.08–2.95 1.51–12.09 2.55–19.78 0.02–10.68 |
0.424 0.006 <0.001 0.606 |
| Lymphovascular invasion | No Yes Unknown |
613 (81.7) 119 (15.9) 18 (2.4) |
83 (61.5) 50 (37.0) 2 (1.5) |
530 (86.2) 69 (11.2) 16 (2.6) |
<0.0001 | 1 3.11 0.88 |
1.98–4.86 0.22–3.52 |
<0.0001 0.853 |
Abbreviation: BCS: breast conserving surgery.
Non-hot node metastases were found in 30 (4%) of the 750 cN0 cases (Table 3). The cN0 patients were divided into SLN negative (absence of tumor cells in the “hot nodes”, or the node with top counts and nodes with more than 10% of the highest signal) group and SLN positive (presence of tumor cells in hot nodes) group. There was a significant difference in the axillary cancer metastasis (2.4% vs 12.7%) between the two groups after surgery (p < 0.001, by Fisher's exact test statistical analysis) (Table 3). This particular group constitutes 11.1% of all patients with final pathologically confirmed lymph node metastasis (135 cases) and 2% of all cN0 cases, which might have missed the detection or result in false-negative staging for positive axillary lymph node metastasis. A detailed list of the lymph node status in the 15 cases where metastasis was detected exclusively in non-hot node was presented in Supplementary Table 1. Among them, 11 cases received further ALND, and 1 received ALNS. None had a tumor located in the lower inner quadrant; one was located centrally, one in the upper inner quadrant, four in the outer lower quadrant, and 9 cases in the outer upper quadrant. Therefore, the upper outer quadrant seemed to be the predominant (60%) location of cN0 breast cancer with axillary lymph node metastasis that might have failed to be detected, if only the radio-isotope hot nodes had been biopsied.
Table 3.
Sentinel lymph node metastasis and its detection.
| Positive Non-Hot Node |
Negative Non-Hot Node |
Total | p value | |
|---|---|---|---|---|
| Positive Hot Node | 15 (12.7%) | 103 (87.3%) | 118 (100%) | <0.001 |
| Negative Hot Node | 15 (2.4%) | 617 (97.6%) | 632 (100%) | |
| Total | 30 (4%) | 720 (96%) | 750 (100%) |
Hot Node: sentinel lymph node (SLN) carrying radioisotope signal.
Non-Hot Node (non-SLN): nodes biopsied without radioisotope signal.
Positive: pathologically confirmed metastasis.
Negative: no metastasis.
In those with negative metastasis of the hot nodes in SLNB, positive metastasis of the non-hot nodes would potentially contribute to false-negative SLNB. Since SLNB is a well-developed clinical practice, ALND was omitted for the majority of patients following SLNB in this cohort; therefore, the true false-negative rate of SLNB cannot be calculated in our series. However, the additional biopsy of non-hot-nodes did result in 2.4% (15/632) reduction or improvement in the false-negative rate of SLNB compared to hot-node biopsy only. Clinical parameters that correlated with non-hot node metastasis include tumor quadrant, breast surgical method, and T-stage. However, multivariate analysis showed only T-stage, especially T2, to be related to an odds ratio as high as 9.65 (95% CI 1.18–78.67, p = 0.034). Strictly upper outer quadrant showed a good trend toward non-hot node metastasis (OR 2.75, 95% CI 1.00–7.59, p = 0.050) in the multivariate analysis despite a very limited number of non-hot node metastatic cases (Table 4). We further looked into the impact of tumor size by analyzing the area under the receiver operating curve (AUC by ROC). The optimal cut-off value predicting non-hot node metastasis in a hot node-negative patient was 2.5 cm. The AUC score was 0.71 or categorized as “fair” (Fig. 1).
Table 4.
Factors correlated with non-hot node (non-SLN) metastasis in hot node (SLN) -negative patients.
| Parameters | Total N = 632 (%) |
Non-Hot Node (+) N = 15 (%) |
Non-Hot Node (−) N = 617 (%) |
p value | Bayesian logistic regression |
|||
|---|---|---|---|---|---|---|---|---|
| Odds Ratio |
95% CI | p value | ||||||
| Age (years) | ≤50 >50 |
250 (39.6) 382 (60.4) |
7 (46.7) 8 (53.3) |
243 (39.4) 374 (60.6) |
0.569 | – | ||
| Quadrant | Upper outer Upper inner Lower inner Lower outer Central |
224 (35.4) 167 (26.4) 62 99.8) 118 (18.7) 61 (9.7) |
9 (60.0) 1 (6.7) 0 4 (26.7) 1 (6.7) |
215 (34.8) 166 (26.9) 62 (10.0) 114 (18.5) 60 (9.7) |
0.128 | – | ||
| Quadrant | Strictly UOQ Others |
206 (32.6) 426 (67.4) |
9 (60.0) 6 (40.0) |
197 (31.9) 420 (68.1) |
0.046 | 1 2.75 |
1.00–7.59 | 0.050 |
| Tumor foci | Unifocal Multifocal |
603 (95.4) 29 (4.6) |
14 (93.3) 1 (6.7) |
589 (95.5) 28 (4.5) |
0.510 | – | ||
| Injection interval | Same day Overnight |
498 (78.8) 134 (21.2) |
12 (80.0) 3 (20.0) |
486 (78.8) 131 (21.2) | >0.999 | – | ||
| Breast surgical method | BCS Mastectomy |
338 (53.5) 294 (46.5) |
4 (26.7) 11 (73.3) |
334 (54.1) 283 (45.9) |
0.035 | 1 2.51 |
0.82–7.66 | 0.106 |
| IHC markers | ER (+) ER (−) |
498 (78.8) 134 (21.2) |
10 (66.7) 5 (33.3) |
488 (79.1) 129 (20.9) |
0.332 | – | ||
| PR (+) PR (−) |
454 (71.8) 178 (28.2) |
9 (60.0) 6 (40.0) |
445 (72.1) 172 (27.9) |
0.382 | – | |||
| HER2(+) HER2(−) |
107 (83.1) 525 (83.1) |
3 (20.0) 12 (80.0) |
104 (16.9) 513 (83.1) |
0.728 | – | |||
| Molecular Subtype (IHC) | Luminal A Luminal B HER2(+)HR (+) HER2(+)HR (−) Triple negative |
152 (24.1) 305 (48.3) 48 (7.6) 73 (11.6) 54 (8.5) |
3 (20.0) 5 (33.3) 2 (13.3) 4 (26.7) 1 (6.7) |
149 (24.1) 300 (48.6) 46 (7.5) 69 (11.2) 53 (8.6) |
0.244 | – | ||
| T stage | T1mi/T1a T1b T1c T2 T3 |
86 (13.6) 62 (9.8) 238 (37.7) 236 (37.3) 10 (1.6) |
0 0 3 (20.0) 12 (80.0) 0 |
86 (13.9) 62 (10.0) 235 (38.1) 224 (36.3) 10 (1.6) |
0.028 | 1 0.51 2.91 9.65 0.77 |
0.02–15.74 0.32–26.54 1.18–78.67 0.01–42.80 |
0.701 0.344 0.034 0.898 |
| Lymphovascular invasion | No Yes Unknown |
541 (85.6) 75 (11.9) 16 (2.5) |
10 (66.7) 5 (33.3) 0 |
531 (86.1) 70 (11.3) 16 (2.6) |
0.059 | – | ||
Fig. 1.
Receiver operating curve showing cut-off of tumor size that warrants careful exploration of non-Isotope-hot nodes.
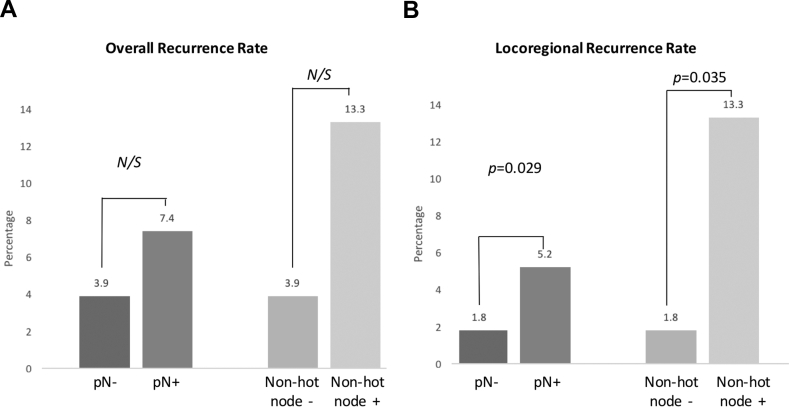
Next, we compared the outcome of early breast cancer patients in our study (Table 5). The breast cancer specific mortality rate in this cohort was 1.1% with a median follow up of 41.80 months, which was extremely low as expected. The recurrence pattern between these groups was then compared. Between the pN+ (n = 135) and final pathological node-negative (pN-) groups (n = 615), there were no statistically significant differences in the overall recurrence (7.4% versus 3.9%, p = 0.076, Table 5 and Fig. 2A) and also distant metastasis (2.2% versus 2.1%, p > 0.999: Table 5). However, locoregional relapse rates were significantly different between pN+ (n = 135) and pN- groups (n = 615) (5.2% vs 1.8%, p = 0.029; Fig. 2B) and between the non-hot node-positive group (n = 15) and non-hot node-negative group (n = 617) (13.3% vs 1.8%, p = 0.035; Fig. 2B). Similarly, there was also no significant difference in overall recurrence (13.3% vs 3.9%, p = 0.123; Table 5 and Fig. 2a) and distant metastasis (0% vs 2.1%, p > 0.999; Table 5), between the non-hot node-positive group (n = 15) and non-hot node-negative group (n = 617). Using the Kaplan–Meier analysis with log-rank test, we found that there was no significant difference in relapse-free survival (RFS) between cases with pathologically negative and positive nodes (Fig. 3A, 69% vs. 66.2%, p = 0.079); similar results were observed in the non-hot node-negative and positive groups (Fig. 4A, 69.0% vs. 61.8%, p = 0.056). Pathologically node-positive group had, for certain, significantly worse locoregional relapse-free survival (LRRFS) (Figs. 3B and 67.3% vs. 70.2%, p = 0.022). Notably, its significance was even more prominent between non-hot node-positive and negative patients (Fig. 4B, LRRFS 66.2% vs. 69.0%, p = 0.001).
Table 5.
Outcomes of clinically node negative patients receiving sentinel lymph node biopsy (upper panel) and of hot-node negative patients (lower panel) after median follow-up of 48.16 months.
| cN0 patients | Total N = 750 (%) | Lymph Node (+) pN+, N = 135 (%) | Lymph Node (−) pN-, N = 615 (%) | p-value | |
|---|---|---|---|---|---|
| Recurrence pattern | Overall | ||||
| No | 716 (95.5) | 125 (92.6) | 591 (96.1) | 0.076 | |
| Yes | 34 (4.5) | 10 (7.4) | 24 (3.9) | ||
| Locoregional | |||||
| No | 732 (97.6) | 128 (94.8) | 604 (98.2) | 0.029 | |
| Yes | 18 (2.4) | 7 (5.2) | 11 (1.8) | ||
| Distant metastasis | |||||
| No | 734 (97.9) | 132 (97.8) | 602 (97.9) | >0.999 | |
| Yes | 16 (2.1) | 3 (2.2) | 13 (2.1) | ||
| Breast cancer specific mortality | Yes | 8 (1.1) | 2 (1.5) | 6 (1.0) | 0.640 |
| No | 742 (98.9) | 133 (98.5) | 609 (99.0) | ||
| SLN (−) patients | Total N = 632 (%) |
Non-Hot Node (+) N = 15 (%) |
Non-Hot- Node (−) N = 617 (%) |
p-value | |
| Recurrence pattern | Overall | ||||
| No | 606 (95.9) | 13 (86.7) | 593 (96.1) | 0.123 | |
| Yes | 26 (4.1) | 2 (13.3) | 24 (3.9) | ||
| Locoregional | |||||
| No | 619 (97.9) | 13 (86.7) | 606 (98.2) | 0.035 | |
| Yes | 13 (2.1) | 2 (13.3) | 11 (1.8) | ||
| Distant metastasis | |||||
| No | 619 (97.9) | 15 (100.0) | 604 (97.9) | >0.999 | |
| Yes | 13 (2.1) | 0 | 13 (2.1) | ||
| Breast cancer specific mortality | Yes | 7 (1.1) | 1 (6.7) | 6 (1.0) | 0.155 |
| No | 625 (98.9) | 14 (93.3) | 611 (99.0) | ||
Abbreviations: cN0: clinically node negative; SLN (-): sentinel lymph node negative.
Fig. 2.
The rate of overall recurrence (A) and locoregional recurrence (B) of pathological node-negative, pathological node-positive, non-hot node-negative and non-hot node-positive groups after a median follow up of 41.80 months. Abbreviations used: pN-: pathological node-negative; pN+: pathological node-positive; non-hot node-: non-hot node-negative; non-hot node+: non-hot node-positive; N/S: non-significant.
Fig. 3.
Kaplan–Meier survival curve displaying relapse-free survival (RFS) (A) and locoregional relapse-free survival (LRRFS) (B) in pN- and pN + groups.
Fig. 4.
Kaplan–Meier survival curve displaying relapse-free survival (RFS) (A) and locoregional relapse-free survival (LRRFS) (B) in non-hot node-negative and non-hot node-positive groups.
Discussion
We have shown that tracer-free sentinel lymph nodes picked up based on surgeons' experiences and intraoperative evaluation impact locoregional recurrence at a median follow-up of 41.80 months. The observation that there is no impact on the overall survival suggested that the risk of this potentially false-negative group of patients may have been successfully rescued by the detection of occult lymph node metastasis and administering appropriate adjuvant systemic treatment. Surgeon's experience is indeed an important factor associated with the false-negative rate, as previously emphasized in literature [18] or technical guidelines [19]. The volume of a breast surgeon also affects the likelihood for a patient to receive high-quality SLNB [20]. Although some articles had claimed that nomogram is superior to clinical evaluation [21] in predicting non-SLN metastasis, our study is the first to demonstrate the usefulness of intraoperative palpation to improve the accuracy of axillary staging by aiding in the detection of non-hot node metastasis in cases with negative hot SLNs.
In our institute, specialized breast surgeons personally perform the ultrasound examination for each patient before breast cancer surgery. Both image and clinicopathological findings affect the surgeons’ decision and judgement on whether to perform SLNB, and how many and which SLNs should be biopsied [22]. Pilewskie et al. reported that more than one abnormal lymph node detected by ultrasound implied three or more pathologically involved lymph nodes [23]. Our surgeons can mark off clinically node-positive patients more precisely by performing preoperative axillary ultrasound evaluation or even lymph node biopsy under ultrasound guidance. Although only a single radioisotope tracer was used in this study, the precautions are the same for cases using a dual tracer method. At the same time, specialized breast surgeons should know the consequences of positive SLNB for their patients as to the subsequent surgical and systemic management. As SLNB is a well-established surgical procedure, it would be ethically inappropriate to dissect all axillary nodes from patients, whether SLN is metastatic or not, to obtain the valid false-negative rate of SLNB for our study. Our focus is to optimize the SLNB technique regarding “which SLN should be biopsied”, especially among the non-hot nodes. Indeed, this is the limitation of this study to determine the magnitude of the false-negative rate improved by additional non-hot node biopsy. However, we demonstrated the benefit in an alternative way by showing the degree of improvement in LRRFS.
Yu et al. [24] demonstrated their excellent result of SLN detection in a small series after neoadjuvant chemotherapy with the aid of intraoperative ultrasound. It is valuable since the accuracy of SLN detection with an injected tracer may be reduced due to fibrotic change or obscured lymphatic drainage after treatment. Our cohort is different as we only included patients receiving upfront surgery who had been pre-excluded with radiologically significant lymph node or by preoperative ultrasound with or without an ultrasound-guided biopsy. The visualization of the remaining equivocal lymph nodes by intraoperative ultrasound in our cohort can be tricky. Intraoperative palpation is a convenient and handy add-on method to SLN tracer in this setting to explore other possibly involved nodes in the axillary bed without causing the concern of over-sampling.
The result that tumor size larger than 2.5 cm predicts more lymph node metastasis in non-hot nodes can be supported by the observation that lymphatic drainage of a larger tumor may be directed to a node other than true sentinel nodes [25]. Larger tumors are more likely to present with node-positive disease. In node-positive breast cancer, 39% of patients had cancer cells in the nodal basin outside of the sentinel lymph node. Uptake of tracer could also be interfered with by gross tumor involvement, particularly in larger tumors [26].
In the 15 cases in which metastasis was detected in non-hot nodes but not in hot nodes, no tumor was located in the lower inner quadrant. Sixty percent was located in the upper outer quadrant. Due to the small case number, no statistical significance could be found regarding tumor location. However, this interesting numerical trend still provides important information for surgeons to examine the axillary bed carefully, particularly for tumors from the upper outer quadrant. The prognostic impact of tumor location has been reported in other literature [27]. In contrast to tumors located in the lower inner quadrant, which showed worse prognosis than other quadrants, a better prognosis was however reported in tumors in the upper outer quadrant [28,29]. These findings support our speculation that tumors in the upper outer quadrant may have parallel or independent direct lymphatic drainage to the axilla, in addition to the mainstream lymphatic routes that are much easily mapped with tracer. Therefore, even for smaller tumors, axillary lymph node metastasis may develop through these shorter and independent lymphatics, which does not necessarily reflect tumor aggressiveness or biology. This finding supported the importance of removing non-hot, suspicious axillary lymph nodes in addition to conventional SLNB especially for the upper outer quadrant breast cancers. After stratification by the TN stage, upper outer quadrant tumors eventually present with better outcomes.
A noteworthy finding in our study is that the overall relapse rate does not differ between the non-hot node-positive and non-hot node-negative patients, which was also observed between the pN+ and pN- groups. A possible explanation is that in this study, only clinically node-negative patients were enrolled, representing patients with earlier stage, lower tumor burden despite pathologically proven nodal metastasis, and heterogeneous subtypes; therefore, the impact of nodal metastasis was not prominent on the overall recurrence rate or 5-year relapse-free survival rate, and could only be observed through a higher locoregional recurrence.
In clinical practice, lymph node metastasis is a strong indication for adjuvant chemotherapy. Under-staging the nodal status may lead to inadequate adjuvant systemic treatment, resulting in a worse outcome. The role of sophisticated SLNB hence outweighs that of the type of mastectomy (total mastectomy vs. breast conservation) performed, which requires shared multifactorial decision-making [30] and contributes less to the indication of adjuvant chemotherapy and does not impact on survival or local recurrence [31].
Conclusions
For cN0 breast cancer patients receiving SLNB with a single radioisotope tracer method, non-hot node metastasis occurs more often in cases where tumor size is larger than 2.5 cm. The presence of non-hot node metastasis correlates with a higher locoregional recurrence rate. Surgeons should consider these parameters to improve their SLNB skills to increase the accuracy of axillary staging.
Conflicts of interest
The authors have no financial or ethical conflicts of interest to report.
Acknowledgments
Shu-Fang Huang, statistical assistance, Carol Hung, data collection.
Footnotes
Peer review under responsibility of Chang Gung University.
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bj.2021.04.009.
Funding sources
This work was supported by Chang Gung Memorial Research Project, Taiwan [CMRP CORPG1F0041].
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Pan H., Gray R., Braybrooke J., Davies C., Taylor C., McGale P., et al. 20-Year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N Engl J Med. 2017;377:1836–1846. doi: 10.1056/NEJMoa1701830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liao G.-S., Hsu H.-M., Chu C.-H., Hong Z.-J., Fu C.-Y., Chou Y.-C., et al. Prognostic role of lymphovascular invasion and lymph node status among breast cancer subtypes. J Med Sci. 2018;38:54–61. [Google Scholar]
- 3.von Minckwitz G., Procter M., de Azambuja E., Zardavas D., Benyunes M., Viale G., et al. Adjuvant Pertuzumab and Trastuzumab in early HER2-positive breast cancer. N Engl J Med. 2017;377:122–131. doi: 10.1056/NEJMoa1703643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Raut C.P., Hunt K.K., Akins J.S., Daley M.D., Ross M.I., Singletary S.E., et al. Incidence of anaphylactoid reactions to isosulfan blue dye during breast carcinoma lymphatic mapping in patients treated with preoperative prophylaxis: results of a surgical prospective clinical practice protocol. Cancer. 2005;104:692–699. doi: 10.1002/cncr.21226. [DOI] [PubMed] [Google Scholar]
- 5.Cimmino V.M., Brown A.C., Szocik J.F., Pass H.A., Moline S., De S.K., et al. Allergic reactions to isosulfan blue during sentinel node biopsy--a common event. Surgery. 2001;130:439–442. doi: 10.1067/msy.2001.116407. [DOI] [PubMed] [Google Scholar]
- 6.Veronesi U., Paganelli G., Galimberti V., Viale G., Zurrida S., Bedoni M., et al. Sentinel-node biopsy to avoid axillary dissection in breast cancer with clinically negative lymph-nodes. Lancet. 1997;349:1864–1867. doi: 10.1016/S0140-6736(97)01004-0. [DOI] [PubMed] [Google Scholar]
- 7.He P.S., Li F., Li G.H., Guo C., Chen T.J. The combination of blue dye and radioisotope versus radioisotope alone during sentinel lymph node biopsy for breast cancer: a systematic review. BMC Cancer. 2016;16:107. doi: 10.1186/s12885-016-2137-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Carmon M., Olsha O., Rivkin L., Spira R.M., Golomb E. Intraoperative palpation for clinically suspicious axillary sentinel lymph nodes reduces the false-negative rate of sentinel lymph node biopsy in breast cancer. Breast J. 2006;12:199–201. doi: 10.1111/j.1075-122X.2006.00241.x. [DOI] [PubMed] [Google Scholar]
- 9.Ozkurt E., Yardimci E., Tukenmez M., Ersoy Y.E., Yilmaz R., Cabioglu N., et al. Intraoperative palpation of sentinel lymph nodes can accurately predict axilla in early breast cancer. Breast J. 2019;25:96–102. doi: 10.1111/tbj.13149. [DOI] [PubMed] [Google Scholar]
- 10.Cox C.E., Salud C.J., Cantor A., Bass S.S., Peltz E.S., Ebert M.D., et al. Learning curves for breast cancer sentinel lymph node mapping based on surgical volume analysis. J Am Coll Surg. 2001;193:593–600. doi: 10.1016/s1072-7515(01)01086-9. [DOI] [PubMed] [Google Scholar]
- 11.Clarke D., Newcombe R.G., Mansel R.E. The learning curve in sentinel node biopsy: the ALMANAC experience. Ann Surg Oncol. 2004;11Suppl 3 doi: 10.1007/BF02523631. 211s-5s. [DOI] [PubMed] [Google Scholar]
- 12.Sanidas E.E., Daskalakis M., Sbyrakis N., Tsiftsis D.D. Modifications of the learning curve guidelines for breast cancer sentinel node biopsy. Eur J Surg Oncol. 2005;31:357–363. doi: 10.1016/j.ejso.2005.01.002. [DOI] [PubMed] [Google Scholar]
- 13.Testori A., Meroni S., Moscovici O.C., Magnoni P., Malerba P., Chiti A., et al. Surgical sentinel lymph node biopsy in early breast cancer. Could it be avoided by performing a preoperative staging procedure? A pilot study. Med Sci Mon Int Med J Exp Clin Res. 2012;18:Cr543–Cr549. doi: 10.12659/MSM.883349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pesek S., Ashikaga T., Krag L.E., Krag D. The false-negative rate of sentinel node biopsy in patients with breast cancer: a meta-analysis. World J Surg. 2012;36:2239–2251. doi: 10.1007/s00268-012-1623-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Martin R.C., 2nd, Edwards M.J., Wong S.L., Tuttle T.M., Carlson D.J., Brown C.M., et al. Practical guidelines for optimal gamma probe detection of sentinel lymph nodes in breast cancer: results of a multi-institutional study. For the University of Louisville Breast Cancer Study Group. Surgery. 2000;128:139–144. doi: 10.1067/msy.2000.108064. [DOI] [PubMed] [Google Scholar]
- 16.Krag D.N., Anderson S.J., Julian T.B., Brown A.M., Harlow S.P., Ashikaga T., et al. Technical outcomes of sentinel-lymph-node resection and conventional axillary-lymph-node dissection in patients with clinically node-negative breast cancer: results from the NSABP B-32 randomised phase III trial. Lancet Oncol. 2007;8:881–888. doi: 10.1016/S1470-2045(07)70278-4. [DOI] [PubMed] [Google Scholar]
- 17.Boughey J.C., Suman V.J., Mittendorf E.A., Ahrendt G.M., Wilke L.G., Taback B., et al. Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the American college of surgeons oncology group (ACOSOG) Z1071 clinical trial. J Am Med Assoc. 2013;310:1455–1461. doi: 10.1001/jama.2013.278932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Martin R.C., 2nd, Chagpar A., Scoggins C.R., Edwards M.J., Hagendoorn L., Stromberg A.J., et al. Clinicopathologic factors associated with false-negative sentinel lymph-node biopsy in breast cancer. Ann Surg. 2005;241:1005–1012. doi: 10.1097/01.sla.0000165200.32722.02. discussion 12-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schwartz G.F. Clinical practice guidelines for the use of axillary sentinel lymph node biopsy in carcinoma of the breast: current update. Breast J. 2004;10:85–88. doi: 10.1111/j.1075-122x.2004.21439.x. [DOI] [PubMed] [Google Scholar]
- 20.Yen T.W., Laud P.W., Sparapani R.A., Nattinger A.B. Surgeon specialization and use of sentinel lymph node biopsy for breast cancer. JAMA Surg. 2014;149:185–192. doi: 10.1001/jamasurg.2013.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Smidt M.L., Strobbe L.J., Groenewoud H.M., der Wilt G.J., Van Zee K.J., Wobbes T. Can surgical oncologists reliably predict the likelihood for non-SLN metastases in breast cancer patients? Ann Surg Oncol. 2007;14:615–620. doi: 10.1245/s10434-006-9150-5. [DOI] [PubMed] [Google Scholar]
- 22.Caudle A.S., Kuerer H.M., Le-Petross H.T., Yang W., Yi M., Bedrosian I., et al. Predicting the extent of nodal disease in early-stage breast cancer. Ann Surg Oncol. 2014;21:3440–3447. doi: 10.1245/s10434-014-3813-4. [DOI] [PubMed] [Google Scholar]
- 23.Pilewskie M., Mautner S.K., Stempel M., Eaton A., Morrow M. Does a positive axillary lymph node needle biopsy result predict the need for an axillary lymph node dissection in clinically node-negative breast cancer patients in the ACOSOG Z0011 era? Ann Surg Oncol. 2016;23:1123–1128. doi: 10.1245/s10434-015-4944-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yu J.C., Hsu G.C., Hsieh C.B., Yu C.P., Chao T.Y. Role of sentinel lymphadenoectomy combined with intraoperative ultrasound in the assessment of locally advanced breast cancer after neoadjuvant chemotherapy. Ann Surg Oncol. 2007;14:174–180. doi: 10.1245/s10434-006-9132-7. [DOI] [PubMed] [Google Scholar]
- 25.Hill A.D., Tran K.N., Akhurst T., Yeung H., Yeh S.D., Rosen P.P., et al. Lessons learned from 500 cases of lymphatic mapping for breast cancer. Ann Surg. 1999;229:528–535. doi: 10.1097/00000658-199904000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Povoski S.P., Olsen J.O., Young D.C., Clarke J., Burak W.E., Walker M.J., et al. Prospective randomized clinical trial comparing intradermal, intraparenchymal, and subareolar injection routes for sentinel lymph node mapping and biopsy in breast cancer. Ann Surg Oncol. 2006;13:1412–1421. doi: 10.1245/s10434-006-9022-z. [DOI] [PubMed] [Google Scholar]
- 27.Yang J., Tang S., Zhou Y., Qiu J., Zhang J., Zhu S., et al. Prognostic implication of the primary tumor location in early-stage breast cancer: focus on lower inner zone. Breast Cancer. 2018;25:100–107. doi: 10.1007/s12282-017-0797-5. [DOI] [PubMed] [Google Scholar]
- 28.Sohn V.Y., Arthurs Z.M., Sebesta J.A., Brown T.A. Primary tumor location impacts breast cancer survival. Am J Surg. 2008;195:641–644. doi: 10.1016/j.amjsurg.2007.12.039. [DOI] [PubMed] [Google Scholar]
- 29.Bao J., Yu K.D., Jiang Y.Z., Shao Z.M., Di G.H. The effect of laterality and primary tumor site on cancer-specific mortality in breast cancer: a SEER population-based study. PLoS One. 2014;9 doi: 10.1371/journal.pone.0094815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shao J., Rodrigues M., Baxter N.N. Multidisciplinary care of breast cancer patients: a scoping review of multidisciplinary styles, processes and outcomes. Curr Oncol. 2019;26:e385–e397. doi: 10.3747/co.26.4713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Corradini S., Reitz D., Pazos M., Schönecker S., Braun M., Harbeck N., et al. Mastectomy or breast-conserving therapy for early breast cancer in real-life clinical practice: outcome comparison of 7565 cases. Cancers. 2019;11:160. doi: 10.3390/cancers11020160. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.