ABSTRACT
The development of tissue engineering has led to new strategies for mitigating clinical problems; however, the design of the tissue engineering materials remains a challenge. The limited sources and inadequate function, potential risk of microbial or pathogen contamination, and high cost of cell expansion impair the efficacy and limit the application of exogenous cells in tissue engineering. However, endogenous cells in native tissues have been reported to be capable of spontaneous repair of the damaged tissue. These cells exhibit remarkable plasticity, and thus can differentiate or be reprogrammed to alter their phenotype and function after stimulation. After a comprehensive review, we found that the plasticity of these cells plays a major role in establishing the cell source in the mechanism involved in tissue regeneration. Tissue engineering materials that focus on assisting and promoting the natural self-repair function of endogenous cells may break through the limitations of exogenous seed cells and further expand the applications of tissue engineering materials in tissue repair. This review discusses the effects of endogenous cells, especially stem cells, on injured tissue repairing, and highlights the potential utilisation of endogenous repair in orthopaedic biomaterial constructions for bone, cartilage, and intervertebral disc regeneration.
Key Words: biomaterials, cell plasticity, endogenous repair, stem cells, tissue engineering
Introduction
The fast-developing field of materials science has been integrated with the field of life sciences to give rise to tissue engineering. The concept of tissue engineering was first proposed by Yuan-Cheng Fung in 1985 and was clearly defined by the National Science Foundation in 1994.1 Tissue engineering is an area of study that develops biological substitutes for the repair, maintenance, and promotion of the functional and morphological properties of damaged tissues or organs, based on an understanding of the relationship between an organism’s tissue structure and function.2 To date, tissue engineering has been widely applied to the repair and regeneration of tissues including cartilage, bone, intervertebral disc, nerve tissue, blood vessels, corneal tissue, and skin.3-6
The three fundamental elements of tissue engineering are seed cells, growth factors, and scaffolds. Sourcing seed cells has been found to be a bottleneck, restricting the development of tissue engineering.7 Most primary tissue cells (such as chondrocytes, nerve cells, and endothelial cells) have limited donor sources and poor growth ability, meaning that a small number of these cells cannot multiply in vitro to produce an adequate number of cells for in vivo repair.8 Consequently, stem cells with good proliferation and directional differentiation ability to develop into corresponding native cells have become an important source of seed cells.7, 9 Materials loaded with stem/progenitor cells can achieve ideal tissue regeneration. For example, compared with the scaffold alone, a porous β-tricalcium phosphate scaffold loaded with autologous bone marrow mesenchymal stem cells (BMSCs), significantly increased the bone mass and proportion of lamellar bone when used for maxillary sinus floor elevation.10 Spraying autologous BMSCs onto chronic, unhealed skin wounds using a fibrin spray effectively repaired the wounds, and the number of cells sprayed positively correlated with the decrease in the area of the skin lesion.11
Many clinical trials of treatments for various diseases have investigated the use of stem cells for their regenerative potential,12-14 such as in intervertebral disc degeneration (IDD), joint injury, and spinal cord injury. In addition, stem cells have demonstrated immunomodulatory capabilities and modest efficacy in many animal models of tissue injury. Exogenous stem cells are categorized into three classes according to their source: autologous stem cells (from the stem cells of a person, which are produced independently), allogeneic stem cells (from a donor, whose human leukocyte antigens are acceptable matches to those of the patient), and xenogeneic stem cells (from a donor of another species).15 Although stem cell transplantation has shown great potential in tissue regeneration, many challenges associated with the usage of exogenous stem cells as seed cells are yet to be tackled. At present, the main sources of stem cells for clinical treatment are autologous sources, such as BMSCs or adipose stem cells extracted from the bone marrow of the patient or their adipose tissue, which is cultured and amplified in vitro and then re-administered to the patient by infusion.16 However, it is difficult to obtain approval from the Food and Drug Administration of the United States for this method, and there are many issues that are unresolved, including the limited availability of donor tissues, highly expensive and time consuming in vitro culture, potential infection of the cells by pathogens, and the need for additional invasive procedures in the case of the donors.8, 17 In addition, the age and basic pathology of the donors, in vitro preservation of the cells, and cell processing during surgery greatly affect the success of auto-transplantation. In the case of xenogeneic or allogeneic stem cells, the challenges are not confined to those mentioned above and include unexpected graft-versus-host reactions.18 In addition, for the repair of certain tissues, such as the bone-cartilage interface, different types of cell aggregation and extracellular matrix (ECM) deposition are required at the corresponding interfaces. Moreover, it is difficult to resolve the problem of cell spatial distribution using exogenous seed cells.
Endogenous repair involves local tissue repair that relies on the naturally available endogenous cells in situ. Further, it provides a feasible solution to the limitations of seed cells in tissue engineering.19 When developing tissue engineering materials, the ability of an organism to self-renew or self-repair is a significant aspect that should not be ignored. When patients, without underlying diseases, suffering from stable fractures are bandaged and immobilized, the fractures are completely healed within 1-6 months, indicating that they are capable of self-repair.20 Fracture healing and remodelling mainly depend on a variety of endogenous cells, including endothelial cells, mesenchymal stem cells (MSCs), and osteoblasts.21 Cell programming, that is, the plasticity of the endogenous stem/progenitor cells, has been identified as a major pathway to differentiate and supplement the residual adult cells in response to tissue injury.22 Therefore, focusing on the activity of the endogenous cells and fully elucidating their latent regeneration capacity may provide a new, effective strategy for developing tissue engineering materials. This review mainly discusses the challenges and key applications of endogenous repair in orthopaedic biomaterials for bone, cartilage, and intervertebral disc regeneration.
Search Strategy
The articles about the endogenous repair in bone, cartilage or intervertebral disc were retrieved by the search terms: (endogenous pair (Title/Abstract Terms) AND stem cells (MeSH Terms) OR progenitor cells (MeSH Terms) OR cell plasticity (MesH Terms)) AND (bone (MeSH Terms) OR cartilage (MeSH Terms) OR intervertebral disc (MeSH Terms)). All these searches were performed on PubMed and Web of Science prior to Feburary, 2021.The results were further screened by title and abstract. Irrelevant articles were excluded. In the end, 231 articles were included in this review (Figure 1).
Figure 1. The flow diagram of enrolling articles.
Plasticity of Endogenous Cells
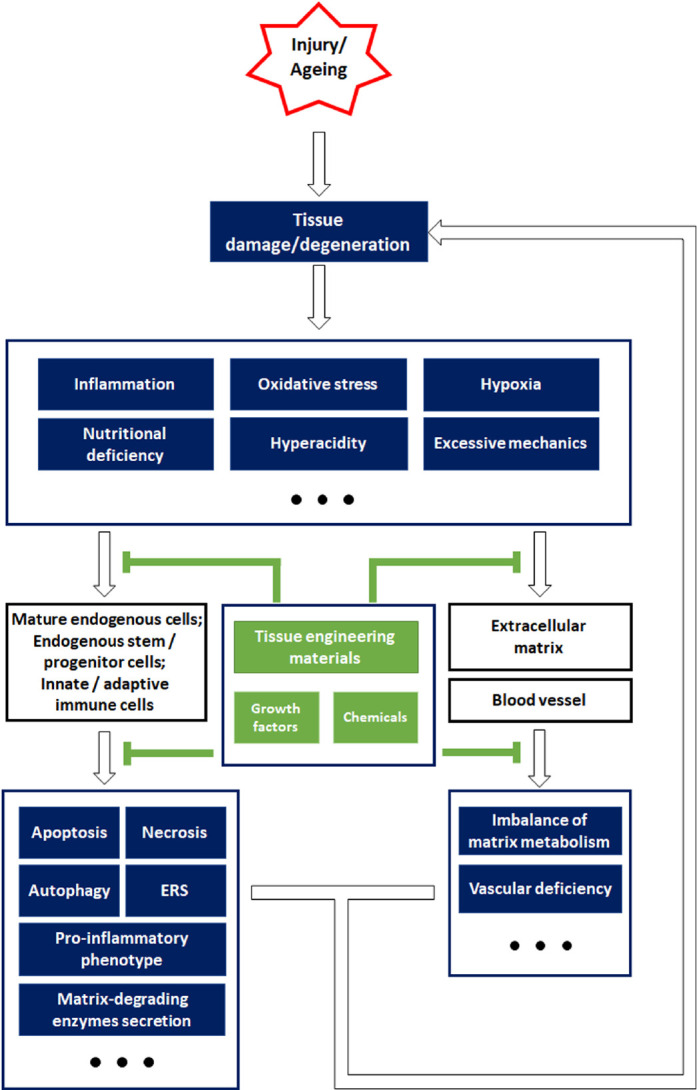
Endogenous repair depends on the natural endogenous cells that are responsible for the self-repair of local tissues, the endogenous stem/progenitor cells being the main functional cells.19 Endogenous stem/progenitor cells possess three main characteristics: self-renewal, pluripotent differentiation, and settlement in specific tissue sites. Most endogenous stem/progenitor cells are dormant under physiological conditions but can be activated by pathological stimuli or other inductive factors, following which they display regenerative ability that involves replacing the damaged or dead cells while producing tissue-specific ECM.23 Cell plasticity, which is a change in behaviour and functional contribution, is a characteristic of endogenous cells that can result in a phenotypic alteration and reprogramming after tissue injury.24, 25 Although intracellular defects and environmental factors may impair the biofunction of the endogenous cells and hinder the self-repair of degenerated/injured tissue, the remarkable plasticity of these cells offers feasible strategies for functional modifications (Figure 2).
Figure 2. Schematic overview of the strategies for constructing biomaterials, inspired by endogenous repair failure that occurs owing to injury or ageing-related pathophysiological changes. Aberrant external impacts cause tissue damage, while ageing often leads to tissue degeneration. After tissue damage or degeneration, the resulting unfavourable microenvironment is characterized by inflammation, oxidative stress, hypoxia, insufficient nutrition, hyperacidity, and abnormal mechanical properties, which impose a great burden on the endogenous cells and non-cellular components. Specifically, mature endogenous cells and stem/progenitor cells typically suffer from cell death and endoplasmic reticulum stress (ERS), and secrete pro-inflammatory factors (interleukin 1β, interleukin 6, tumour necrosis factor α, etc.), while immune cells are also involved in aggravating the inflammation. In addition, the harsh environment also leads to an imbalance in the matrix metabolism and impairs the endothelial cells that are essential for angiogenesis. Cellular and non-cellular alterations in unfavourable environments contribute to endogenous repair failure. However, tissue engineering materials and other bioactive agents are efficient in relieving the pathological changes and their damaging impact on cells and extracellular components, which may help re-establish endogenous repair mechanisms and alleviate tissue damage or degeneration.
Endogenous repair in bone reconstruction
Cell plasticity after bone injury
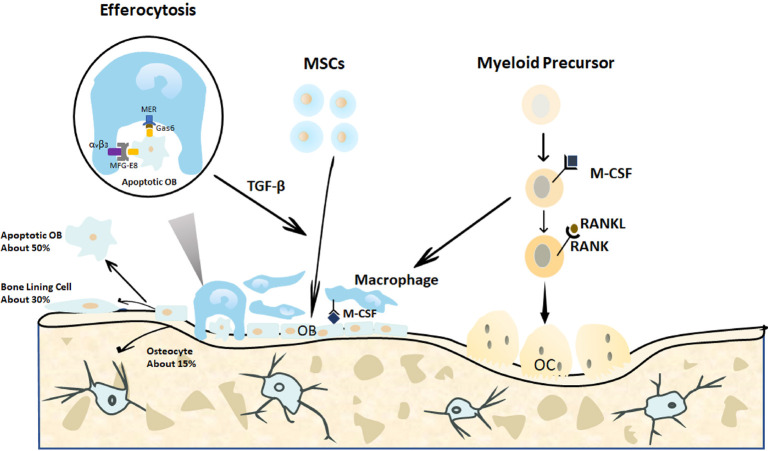
After fracture, the process of bone repair depends on a variety of endogenous cells. Osteoblasts and osteoclasts are key endogenous cells for bone tissue regeneration and reconstruction. After an injury, the MSCs are programmed and differentiated into osteoblasts that are responsible for bone formation by directing the synthesis, secretion, and mineralization of the bone matrix. Osteoblasts not only affect the growth and metabolism of the bone tissue under normal physiological conditions but also influence its reconstruction in the injured state.26 Osteoclasts are multinucleated cells that originate from bone marrow monocytes. Myeloid precursors enter the blood circulation after being activated by chemokines,27 reach the bone tissue defect site, and differentiate into osteoclasts in the presence of macrophage colony-stimulating factor and receptor activator of nuclear factor-κB (NF-κB) ligand (RANKL)28, 29 (Figure 3). Therefore, the plasticity of endogenous cells contributes to tissue regeneration when a bone is injured and its repair capability is impaired.
Bone homeostasis is regulated by a balance between bone formation by osteoblasts and bone resorption regulated by osteoclasts.30 On the one hand, the osteoblasts construct a non-mineralized bone matrix, which is the basis of bone formation at the site of bone resorption, by synthesizing and secreting collagen and glycoproteins. Calcium and phosphorus then form crystalline deposits in the bone matrix to generate new bone tissue, that is, the formation of new bone.31 In addition, the osteoblasts can secrete alkaline phosphatase to mediate bone mineralization, while simultaneously, the osteoblasts also interact with osteoclasts to regulate bone resorption.32 The glycoprotein osteopontin, which is synthesized and secreted by osteoblasts, is an important chemotactic factor for osteoclasts, inducing them to attach to the bone surface and initiate bone resorption. Furthermore, bone sialoprotein secreted by osteoclasts is directly involved in bone resorption. Normally, once osteoclasts leave the bone defect surface, osteoblasts start to cluster and mediate osteogenic activity.33, 34
Endogenous repair failure
In the initial stage of bone tissue injury, bleeding causes a hematoma at the fracture site; consequently, cytokines are released, which enlarge the gaps between endothelial cells and increase vascular permeability. A variety of chemokines induce the migration of leukocytes, monocytes, macrophages, and endogenous mesenchymal cells to the fracture site.35 Simultaneously, the blood supply to both sides of the fracture site is temporarily interrupted, resulting in local anoxic necrosis.36 Necrosis also leads to the release of growth factors such as bone morphogenetic proteins (BMPs), which promote the differentiation of peripheral mesenchymal cells into osteoblasts.37 The local blood supply is an important factor that affects fracture healing. After bone injury, insufficient penetration and vascularisation of the endothelial cells result in a limited supply of nutrients and oxygen to the osteoblasts and osteoclasts, thereby impairing bone regeneration.38
The adipose tissue of the bone marrow significantly affects the function of osteoclasts and osteoblasts. When BMSCs differentiate into preadipocytes in the bone marrow adipose tissue, the secretion of RANKL increases concomitantly, which induces the BMSCs to differentiate toward osteoclasts, promotes osteoclast activation, and reduce the bone mass.39 Similarly, adiponectin secreted by adipocytes reduces the activity of Forkhead box protein O1 in a PI3 kinase-dependent manner, which inhibits the proliferation of osteoblasts and promotes their apoptosis.40 Hence, inhibiting adiponectin secretion by adipocytes may be favourable for bone formation. However, this needs to be verified and supported by extensive and robust studies.
Endoplasmic reticulum stress (ERS) plays a key role in maintaining protein homeostasis and intracellular environment stability.41 However, when intracellular and extracellular stresses (including Ca2+ overload, hypoxia, abnormal glycosylation, and viral infection) become overwhelming after a fracture, the excessive ERS causes cellular dysfunction.42 For instance, the inflammatory microenvironment formed as a result of bone injury causes excessive ERS in osteoblasts, as implied by the significant increase in intracellular activated transcription factor 3, which inhibits the expression of alkaline phosphatase and other osteogenic marker proteins induced by BMP-2.43 Oxidative stress in the inflammatory microenvironment also inhibits osteoblast differentiation and proliferation and induces osteoblast apoptosis, resulting in bone defects.44
Endogenous repair modification
Intramembranous and endochondral ossification are the main processes involved in the formation of new bones. The formation of a primary bone is followed by extensive remodelling until the damaged bone recovers to its original shape and size.45 In this process, with remarkable cell plasticity, the regulation of certain signal pathways significantly alters the cellular behaviour and functional contribution, and affects bone repair. For instance, activation of the Wnt-β-catenin pathway increases the binding of β-catenin to transcription factors and promotes osteogenic gene expression.46 Related to this, the activation of BMP expression in endogenous MSCs by prostaglandins activates β-catenin to accelerate bone repair.47, 48 In addition, expression of vascular endothelial growth factor (VEGF) mediated by hypoxia-inducible factor-1α is a key regulator of endothelial cell vascularization.49 Human umbilical cord mesenchymal-derived exosomes have been used to promote hypoxia-inducible factor-1α-mediated endothelial cell vascularisation. As a result, extensive angiogenesis and ideal fracture healing were observed in a rat fracture model.38 Interfering with ERS and pro-inflammatory factors are also feasible approaches that promote osteoblast survival and bone regeneration.50, 51 Antagonists against critical inflammatory signals, including NOD-, LRR- and pyrin domain-containing protein 3 inflammasomes, NF-κB, and toll-like receptor pathways, reduce osteoblast and BMSC death and the pro-inflammatory phenotype, thus favouring endogenous repair.52-54
In summary, the endogenous cells in bone tissue are directly involved in bone regeneration after tissue injury. The survival and biofunction of these cells can be modified using appropriate agents or stimuli, to achieve ideal tissue regeneration.
Biomaterials for endogenous bone repair
Based on the remarkable plasticity and environmental dependence of the endogenous cells, tissue engineering materials with bionic structures and biological activity have been constructed to provide a suitable microenvironment for the endogenous cells, which may be a promising strategy for tissue regeneration.55, 56 Furthermore, recruiting and activating endogenous stem/progenitor cells can replace the traditional strategy of exogenous cell delivery in tissue biomaterials, which helps resolve the aforementioned problems associated with seed cells, such as insufficient sources and tissue rejection. Therefore, endogenous repair provides a highly inspiring and promising strategy for the construction of tissue engineering materials.
A decellularised bone matrix contains abundant endogenous pro-osteogenic proteins, including bone sialoprotein, osteopontin, fibroblast growth factor, BMP-2, BMP-4, BMP-7, insulin-like growth factor 1 (IGF-1), transforming growth factor β (TGF-β), VEGF, and platelet growth factor (PDGF).57-60 These endogenous bioactive substances endow the decellularised bone matrix with a satisfactory regeneration capacity. By decalcifying three-dimensional (3D) decellularised bone scaffolds for a specific time, a 3D bone scaffold with a bionic microstructure and suitable matrix stiffness can be established. Without supplementary agents or cytokines, this scaffold effectively promotes the osteogenic differentiation of endogenous stem cells into osteoblasts.61, 62 However, the content of endogenous active factors in the decellularised matrix is greatly affected by the age and species of the donors.63 In addition, the delayed release of encoded bioactive substances may reduce efficiency. In one study, diluted recombinant BMP-induced osteoblast markers began to increase in number on day 5 after application, while the number of osteoblast markers started to increase after 14 days in the decellularised bone matrix rich in endogenous BMP, indicating that the release rate of endogenous cytokines in the decellularised bone matrix was relatively low.64 Thus, different batches of decellularised bone matrix may cause variations in the reparative effect owing to the unstable release of endogenous cytokines.57, 58 Since the application of the decellularised bone matrix alone may encompass the characteristics of unstable clinical efficacy, loading exogenous agents to enhance and stabilize the efficacy of the decellularised matrix may be a feasible material construction strategy.
Many decellularised matrices and polymer materials have achieved satisfactory outcomes with the assistance of bioactive substances. Decellularised bone matrix loaded with TGF-β1 released 80-90% of incorporated TGF-β1 within 25 days, and the release kinetics were adjustable by varying the composition of poly(DL-lactic-co-glycolic acid).65 When a composite matrix was applied to skull defects, the defects were filled with new bone after 4 weeks.66 Three forms of poly(DL-lactic-co-glycolic acid) scaffold materials containing BMP-2 showed similar sustained release kinetics of BMP-2, and all of them achieved a complete repair of bone defects in vivo.67 Therefore, bioactive molecules can enhance the pro-osteogenic differentiation activity of bone tissue-engineering materials. At the same time, the combination of appropriate materials and bioactive substances is also a reliable approach to preserve the activity of these substances. For example, the N-terminal domain of BMP-2 has a collagen-binding region. Compared with native BMP-2, BMP-2 conjugated with collagen has a longer half-life, with better bone formation being observed in mandibular defects in vivo.68 Agents targeting specific signalling pathways are also a potential strategy for the construction of functional materials. Gelatine hydrogel loaded with arginine-glycine-aspartate tripeptide effectively improved the viability of MSCs and the release of VEGF by activating the integrin pathway, which significantly promoted mineralisation and bone tissue regeneration in vivo.69
Tissue engineering also provides an effective solution to improve the living environment of the endogenous cells. A 3D-printed alginate hydrogel and calcium peroxide material can increase the oxygen supply to the endogenous stem/progenitor cells in adipose tissue, thus reducing hypoxia-induced apoptosis and ensuring cell proliferation activity.70 Melatonin significantly affects the release of various antioxidant enzymes, thereby playing a key role in alleviating the cytotoxicity of reactive oxygen species (ROS) and free-radical intermediates.71 After being coated with gels and loaded onto a titanium dioxide (TiO2) nanotube matrix, continuously-released melatonin promoted the cell proliferation and osteogenic differentiation of MSCs.72 Chitosan-glycerol phosphate/blood implants in a subchondral bone defect site appropriately activated the macrophages that released more arginase-1 and VEGF without aggravating the inflammation, which induced ideal angiogenesis and bone tissue remodelling within 8 weeks.73 In addition, the combination of 3D-printed poly(DL-lactic-co-glycolic acid) scaffolds and decellularised matrix helped achieve a successful polarisation of the macrophages to M2 and promoted local bone tissue remodelling.74
Endogenous repair in cartilage reconstruction
Cell plasticity after cartilage damage
Articular cartilage comprises chondrocytes and abundant cartilage ECM. Collagens and proteoglycans provide mechanical support, enabling the articular cartilage to withstand hydrostatic pressure at rest and shear force during movement.75, 76 However, when osteoarthritis occurs, the inflammation of the ECM is obvious, causing a significant decrease in collagen, proteoglycan, and glycosaminoglycan.77, 78 As a result, the articular cartilage is progressively destroyed. As the main cellular component in articular cartilage, chondrocytes play a critical role in maintaining homeostasis of the ECM anabolic and catabolic metabolism.79 Barbero et al.80 found that adult dedifferentiated chondrocytes have the ability to form clones and induce osteogenesis, adipogenesis, and chondrogenic differentiation. Alsalameh et al.81 reported the presence of mesenchymal progenitor cells in normal and osteoarthritic cartilage. These cells share the same cell surface markers - CD105 and CD166 - and have the same capacity for proliferation and multi-differentiation as BMSCs, with the ability to differentiate into osteocytes and adipocytes.82, 83 Thus far, cartilage stem/progenitor cells have been confirmed to have the potential for self-renewal, multi-lineage differentiation, and the common phenotype of mesenchymal cells (CD9, C29, CD44, CD49e, CD54, CD73, CD90, CD105, CD166, and Notch-1).83-86
The presence of chondrocytes and cartilage stem/progenitor cells enables the self-repair of cartilage. A case series study suggested that after full-thickness chondral fractures, internal fixation of the fragments back to the bone using headless metal compression screws achieved stable fixation and ideal healing after 8 weeks, as suggested by the fibrous tissue at the small interface between the fragment and surrounding articular cartilage as well as the obvious covering of cartilage tissue on the surface of the screw.87 Cartilage stem/progenitor cells usually appear in the ECM around the injury after 7-14 days in cases where the cartilage damage was caused by impact.83 Soluble molecules, including high-mobility group protein B1, IGF-1, and PDGF, which are released after a cartilage injury, can act as chemotactic factors that induce cartilage stem/progenitor cells to aggregate in the injured area.88-90
Endogenous repair failure
When the articular cartilage is injured, chondrocytes often cannot achieve self-renewal and cartilage repair because of their weak proliferation and limited secretory capacity in vivo.91 Moreover, inflammation increases the apoptosis rate of chondrocytes. Consequently, chondrocyte death releases damage-associated molecular patterns, which further promote the secretion of inflammatory factors and aggravate the local inflammatory microenvironment.92 Therefore, reducing chondrocyte death after an injury, maintaining their phenotype, and increasing the secretory activity of chondrocytes after an injury are important factors to be considered in the reconstruction of articular cartilage.
Macrophages, which exhibit high plasticity and can be extensively modified, are the primary innate immune cells that induce and maintain the pro-inflammatory microenvironment in osteoarthritis. When osteoarthritis occurs, macrophages are activated by TNF-α, interferon γ, or pathogen-related molecular models and are polarised into the M1 phenotype, which secretes a large amount of pro-inflammatory factors such as IL-1, IL-6, and IL-12.93 The establishment of an inflammatory cascade induces the local activation of macrophages and accelerates the removal of pathogens and tissue fragments. Moreover, the M1 inflammatory phenotype of macrophages indicates a clinical manifestation of osteoarthritis.94 Specifically, the M1 phenotype is positively correlated with knee pain and arthritis severity scores (Kellgren-Lawrence grade).94 Therefore, the M1 phenotype may act as an indicator of the progression of osteoarthritis. Adaptive immune cells are also involved in the development of osteoarthritis.94 The number of T and B cells in the synovium and synovial fluid of patients with osteoarthritis is significantly higher than that in healthy controls.95 CD4+ and CD8+ T cells infiltrate into articular tissues, scavenge pathogens, and secrete cytokines such as IL-2, interferon γ, and TNF-α, causing the activation of macrophages and secretion of catabolic cytokines, which accelerates the destruction of the articular cartilage matrix.96, 97
In the induced inflammatory microenvironment, IL-1, IL-6, and TNF-α are vital pro-inflammatory cytokines.98 Chondrocytes and cartilage stem/progenitor cells living in unfavourable inflammatory conditions often fail to produce an ECM. Instead, they tend to aggravate the inflammatory reaction and tissue damage by secreting pro-inflammatory factors and matrix-degrading enzymes, including metalloproteinases and a disintegrin, and metalloproteinase with thrombospondin-like motifs.99 Moreover, chondrocyte death, autophagy, and senescence are induced by IL-1β.100 ROS play an important role in the cellular responses to inflammatory environments.101 Pro-inflammatory stimuli often induce ROS production and decrease antioxidant enzyme production.102 ROS accumulation then induces chondrocyte senescence, apoptosis, imbalance of ECM metabolism, synovitis, and subchondral bone dysfunction, which leads to endogenous failure.103, 104
Endogenous repair modification
The release of IL-4 and IL-13 can induce macrophage transformation into the M2 type, which is beneficial for tissue regeneration in osteoarthritis.105 M2 macrophages with an anti-inflammatory phenotype secrete TGF-β, epidermal growth factor, and VEGF, which promote fibre remodelling in impaired tissues.106 As a result, cells tend to exert anti-inflammatory effects and enhance ECM deposition.107 Some studies have attempted to regulate the inflammatory microenvironment and have achieved reasonable tissue regeneration. Squid collagen type II and exosomes extracted from MSCs employed an increased number of CD163+ M2 macrophages to infiltrate into cartilage defects and downregulate pro-inflammatory cytokines, which enhanced the proliferation and migration of chondrocytes and achieved ideal cartilage regeneration.108, 109
ROS scavengers and anti-inflammatory agents have been developed to reduce chondrocyte death and matrix imbalance. For example, casticin, a flavonoid isolated from Vitex trifolia, regulated oxidative stress and reduced inflammation in the cartilage of mice with osteoarthritis through the NF-κB signalling pathway.110 Loureirin A, a traditional Chinese medicinal plant extract with strong antioxidant activity, inhibited the release of inflammatory mediators and ROS induced by IL-1β through inhibition of the phosphorylation of protein kinase B (AKT) and activation of the NF-κB and nuclear factor E2-related factor 2 pathways.111 Lubricin is an important articular cartilage surface protein that can prevent the excessive proliferation of synovial cells and the apoptosis of chondrocytes. Injecting lubricin into a cartilage defect effectively attracted cartilage stem/progenitor cells located in the defect and alleviated joint degeneration.112, 113 Although the excessive inflammatory response caused by macrophages and lymphocytes in the progression of osteoarthritis often leads to repair failure, chondrocytes and cartilage stem/progenitor cells still have the potential to repair cartilage defects after appropriate modification. In view of the high plasticity of these endogenous cells, the construction of multifunctional materials to regulate inflammation and promote the biofunction of chondrocytes and cartilage stem/progenitor cells is a feasible strategy for treating cartilage injury and inflammation.
Biomaterials for endogenous chondrogenic repair
Collagen type II accounts for 90% of the collagen in the cartilage ECM. Other types of collagen combine with collagen type II to form a collagen fibre network that entraps proteoglycans.114 The most common proteoglycan is aggrecan, which is cross-linked with hyaluronic acid to form a stable protein structure. Many glycosaminoglycans, such as chondroitin sulphate and keratan sulphate, attach to the aggrecan-hyaluronan protein complex, which endows the cartilage with hydration and pressure-resisting capacity.115 Therefore, many biomaterials have utilised these basic cartilage components to form biomimetic materials and have achieved good biocompatibility in vitro.116-118 However, biomaterials comprising only collagen type II cannot induce cartilage formation in vivo,119 and only by loading exogenous stem cells or exogenous chondrocytes into the materials can we achieve ideal repair of cartilage defects.120, 121 Synthetic materials provide excellent mechanical and cross-linking properties while potentially supporting the biofunction of natural components when used to construct composite materials.122 In addition, compared to natural components alone, the combination of natural components and synthetic polymers in appropriate proportions can greatly promote chondrogenic differentiation.123 However, these composite materials fail to induce endogenous cell assembly at the injured site. That is to say, for these biomaterials, exogenous stem cells are required to enhance cartilage regeneration in vivo.124, 125
It has been reported that tissue engineering materials can activate endogenous stem cells and achieve in vivo regeneration without using exogenous stem cells.55, 126 For example, a decellularized matrix prepared by removing the cellular components from tissues and retaining the ECM components minimised the immunogenicity of the materials and maximally retained the matrix composition, microstructure, and biological properties of the natural tissues.55 With the provision of a bionic natural microenvironment, decellularized matrix hydrogels do not require an additional design of binding sites for cell adhesion; this also reduces dependence on exogenous bioactive molecules that induce cell migration and differentiation. A decellularized cartilage matrix is proven to induce chondrocyte migration and promote chondrogenesis of cartilage stem/progenitor cells.127-129 However, further studies are essential to prove the feasibility and effectiveness of such a material in clinical applications. Interestingly, polymeric scaffolds have been found to help enhance the mechanical properties of decellularized cartilage matrix and improve its ability to facilitate chondrogenic differentiation.130, 131
Certain growth factors and cytokines efficiently support cartilage maturation, including BMP-2, BMP-7, IGF-1, and fibroblast growth factor-2.132-135 A biodegradable polylactide-co-glycolide/poly-ε-caprolactone mesh loaded with IGF-1 effectively induced chondrocytes around a defect to break through the host/graft interface and induced the chondrocytes to synthesize ECM that was similar to the structure of natural cartilage.136 Platelets are also highly functional, active ingredients. After being activated by thrombin, platelets release alpha-granule contents that include mitotic factor and chemokines, such as PDGF, IGF-1, TGF-β, VEGF, and epidermal growth factor, which play a critical role in wound healing.137, 138 Therefore, platelets, as a growth factor-rich component, are a natural material with the purpose of preserving the activity of growth factors. The issues of short half-life and high cost of purification are resolved to a greater extent by platelets than by addition of a single growth factor.139 The application of a hydrogel loaded with autologous platelet-rich plasma into a cartilage defect effectively promoted the repair of bone-cartilage interface defects; conspicuous chondrocyte aggregation and ECM synthesis were observed on the cartilage surface, and the bone surface exhibited adequate bone calcification.140 These materials combine the excellent properties of bioactive molecules and scaffolds, thereby supplementing the ECM of endogenous cells or stem/progenitor cells and directly inducing their migration, proliferation, and differentiation.
Inflammation is an important factor in the development of osteoarthritis. In addition, the response of the immune system to an implant determines whether a material can achieve the desired effect after implantation.141 If the size of the degradation particles released from implants is > 100 μm, macrophages are activated to promote cell phagocytosis. The activation of macrophages further aggravates local inflammation and severely affects the biological activity of the materials. The lack of binding sites for cell adhesion in some synthetic materials, such as polyethylene oxide, polyethylene glycol, and polycaprolactone, reduces the local aggregation and activation of inflammatory cells.142, 143 Natural materials of a high molecular weight, such as hyaluronic acid and chitosan, promote the scavenging of oxygen radicals, thus relieving inflammation.144 Once injected into a cartilage defect, hyaluronic acid hydrogel loaded with platelet-rich plasma releases anti-inflammatory factors, including TGF-β, which significantly reduce the local inflammatory response and induce the accumulation of endogenous chondrocytes in the defect to promote tissue repair.145 In addition, a drug delivery system that achieves regional enrichment of anti-inflammatory agents and bioactive molecules provides an effective means to alleviate local inflammation. A light-responsive drug delivery system for the sustained release of dexamethasone maintained its local therapeutic effect after irradiation, which ameliorated the inflammatory environment of the articular cavity and alleviated the progression of osteoarthritis.146
Endogenous repair in intervertebral disc regeneration
Cell plasticity in intervertebral disc regeneration
Intervertebral discs are complex and multi-tissue organs composed of the nucleus pulposus (located in the central area), peripheral annulus fibrosus, and cartilage endplate (on both cranial and caudal sides). For many mammals, such as humans, goats, and dogs, the quantity of intervertebral disc cells and ECM decreases gradually with age. Further, the height and elasticity of the intervertebral discs progressively declines147, 148 and age-related degeneration occurs. In addition, excessive labour and abnormal spinal force lead to excessive pressure on the intervertebral discs, resulting in damage to the annulus fibrosus, prolapse of the nucleus pulposus, and eventually IDD.149, 150 Therefore, the occurrence of IDD is an age-related pathological change in humans. The self-repair capacity of mature intervertebral disc cells is also quite limited, especially in elderly people.151
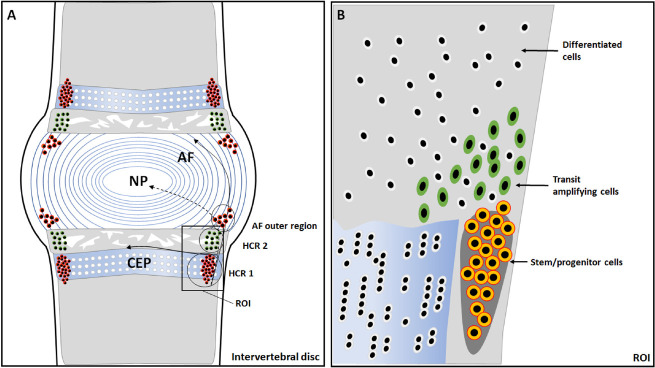
Recently, a group of intervertebral disc cells has been found to express MSC series markers, including OCT3/4, CD90, and stromal cell antigen 1, and they are highly homologous.152 A study on the distribution of 5′-bromodeoxyuridine-labeled cells in intervertebral discs revealed a possible stem cell niche in the perichondrium region adjacent to the epiphyseal plate and outer layers of the annulus fibrosus153 (Figure 4). Henriksson et al.154 showed that cartilage formation markers SRY-box transcription factor-9, growth differentiation factor-5, cell migration markers (snail family transcriptional repressor 1, SLUG, integrin β1), and other genes related to cell proliferation, migration, and differentiation were found in the stem cell niches, peripheral annulus fibrosus, and the central nucleus pulposus of rabbits of different ages. Some studies have revealed the migration path from the stem/progenitor cell niche to the intervertebral disc155, 156 (Figure 4). The 5′-bromodeoxyuridine-labeled cells appeared in the area around the annulus fibrous and the central nucleus pulposus when stimulated by controllable axial pressure, whereas stem/progenitor cell migration decreased under increased pressure.154 Therefore, when an injury is caused by microenvironmental factors, such as pressure, the stem/progenitor cells can be activated, and migrate into the intervertebral disc to perform self-repair.
Figure 3. Endogenous cellular changes after bone fracture. When a bone is fractured, the MSCs migrate to the bone defect area and differentiate into osteoblasts to form and remodel the bone matrix. In the end, approximately 15% of the osteoblasts become embedded in the bone matrix as osteocytes, 30% of the osteoblasts become quiescent bone lining cells, and the remaining 40-70% of the osteoblasts are likely to undergo death by apoptosis. The apoptotic osteoblasts expressing certain signals are efficiently cleared by macrophages in a process called efferocytosis. This process is initiated by the expression of the apoptotic signals on osteoblasts and is activated by the binding of linking proteins, including MFG-E8 or Gas6, and macrophage proteins, such as αvβ3 or Mer. The efferocytosis-induced production of specific proteins, such as TGF-β, may promote continuous bone modelling by recruiting osteoblasts from progenitor cells.29 Gas6: growth arrest-specific 6; M-CSF: macrophage colony-stimulating factor; MER (tk): receptor tyrosine kinase MerTK; MFG-8: milk fat globule-epidermal growth factor 8; MSCs: mesenchymal stromal cells; OB: osteoblasts; OC: osteoclasts; RANK: receptor activator of nuclear factor-κB; RANKL: receptor activator of nuclear factor-κB ligand; TGF-β: transforming growth factor β; αvβ3: alpha-V beta-3 integrin.
Figure 4. Anatomical structure of an intervertebral disc and identification of the stem cell niche and hypothetical migration paths. (A) The potential stem cell niche is in the perichondrium region adjacent to the epiphyseal plate and outer layers of the annulus fibrosus (AF). In the hypercellular region 1 (HCR 1), the cells are densely distributed, and this is where the stem cell niche is located; while in the hypercellular region 2 (HCR 2), the cells are relatively dispersed and morphologically mature. (B) A magnification of the region of interest shows slow cycling stem/progenitor cells (outlined in orange), the transit amplifying cells (outlined in green), and differentiated cells (outlined in white).153 CEP: cartilage endplate; NP: nucleus pulposus.
Endogenous repair failure
The microenvironment of the intervertebral disc is quite different from that of other tissues. Being the largest non-vascular structure in the human body, both oxygen and nutrients are mainly supplied by blood vessels in the adjacent vertebrae through diffusion. Owing to the problems of pressure caused by body weight and exercise, limited supply of oxygen and nutrients, and gradual accumulation of cell metabolites, the microenvironment of the intervertebral disc is characterized by high hydrostatic pressure, hypoxia, hyperacidity, and lack of nutrients.157-159 In such an unfavourable microenvironment, oxidative stress and release of inflammatory factors aggravate cell death and matrix decline, causing excessive oxidative stress, autophagy, apoptosis, and necrosis of stem/progenitor cells, thereby inhibiting endogenous repair.160, 161
Endogenous repair modification
The accumulation of senescent disc cells in degenerative intervertebral discs suggests the detrimental role of cell senescence in the pathogenesis of IDD.162 Moreover, senescent disc cells are thought to accelerate the process of IDD via their aberrant paracrine effects, by which these cells cause the senescence of neighbouring cells and enhance matrix catabolism and inflammation in the intervertebral discs.163 Therefore, anti-senescence-based therapies are essential to restore endogenous cell biofunction. Inhibiting the senescent phenotype of nucleus pulpous cells, which is induced by excessive disc compression, has been found to be effective in decreasing cell apoptosis and disc degeneration.164 Metformin has also been found to be protective against oxidative stress-induced senescence in nucleus pulposus cells, but its clinical applications in IDD therapy require further investigation.165
When IDD occurs, inflammatory factors aggravate the deterioration of the microenvironment. Consequently, endogenous repair is disturbed or deactivated by environmental factors. Some studies have attempted to regulate these harmful pathophysiological factors, and found that downregulating oxidative stress and interfering with inflammation led to improved proliferation of intervertebral disc cells, which alleviates IDD.166-169 For example, the injection of exosomes derived from MSCs into the nucleus pulposus of intervertebral discs promoted cell proliferation and prevented IDD by reducing activation of the inflammasome and ROS levels.166 The homeobox protein Mohawk is an important transcription factor in the outer annulus fibrosus. Overexpression of the Mohawk protein significantly increased collagen fibre synthesis and restored the functional structure of the annulus fibrosus defect.167 In addition, nucleus pulposus cells extracted from degenerative intervertebral discs continued to exhibit good proliferation ability in a nutritious medium.168 Annulus fibrosus cells can also dedifferentiate into an MSC-like phenotype and be reprogrammed to a state of increased plasticity so that additional stimuli, such as cell-matrix and cell-cell interactions, can promote differentiation at a high frequency after reversing treatment.169 Therefore, endogenous intervertebral disc cells exhibit remarkable plasticity and can be modified for tissue regeneration. Regulating cell apoptosis and autophagy may improve their survival rate and biological activity and promote intervertebral disc regeneration.
Biomaterials for intervertebral disc endogenous repair
The nucleus pulposus and annulus fibrosus constitute the basic components of the intervertebral disc that can withstand mechanical loading caused by body weight and shear force during movement.115 The isotropic and gelatinous nature of the nucleus pulposus in the centre of the intervertebral disc endows it with the ability to resist axial pressure and torque.170 The hydration structure of the nucleus pulposus mainly comprises collagen fibres together with various proteoglycans that assemble on the fibre network. Collagen type II accounts for 80% of the total collagen in the nucleus pulposus.171 Proteoglycans (e.g., aggrecan, hyaluronic acid, and decorin) and glycosaminoglycans (e.g., chondroitin sulphate and keratan sulphate) provide copious hydrophilic groups in the fibre network to attract water and maintain hydration of the nucleus pulposus.172 Collagen fibres are arranged into layers that are superimposed to form the annulus fibrosus. The composition of the annulus fibrosus is area-specific. The ratio of collagen type I to type II in the annulus fibrosus gradually decreases from the outer to the inner layers.173 In addition, the amount of proteoglycan gradually increases from the outer to the inner layers along with the water content, while the mechanical strength decreases.174 While the outer annulus fibrosus resists external tensile force, the inner part helps the nucleus pulposus to withstand hydrostatic pressure.175, 176 The endplate is a thin layer of hyaline cartilage covering the interface of the vertebral body, which is also composed of collagen and proteoglycan. Blood vessels in the adult vertebral body cannot pass through the endplate.177 Oxygen and nutrients can only permeate into the intervertebral disc through the endplate; therefore, the endplate plays a key role in controlling the infiltration of soluble materials into the annulus fibrosus and nucleus pulposus.177, 178
Because of the marked impact of ECM components on maintaining intervertebral disc function, biomaterials composed of natural polymers, synthetic polymers, or composite materials have been developed to supplement ECM components.179-182 However, simply supplementing the ECM cannot achieve an ideal therapeutic effect without restoring cell function. Therefore, the delivery of exogenous stem cells into the intervertebral disc has been widely applied, where they can differentiate into local mature cells and secrete ECM. Most cell-conjugated scaffolds or nanoparticles have succeeded in restoring the composition and physiological function of intervertebral discs.183-185 However, there are still some problems to be solved, which hinder the effectiveness of exogenous stem cells in intervertebral disc repair. For instance, BMSCs tend to undergo osteogenic differentiation in unfavourable microenvironments186, 187 and adipose-derived MSCs have tumorigenic potential.188, 189 Moreover, MSCs may still retain the differentiation trend related to tissue specificity, that is, MSCs are pluripotent progenitor cells that retain the source tissue tag.190 Therefore, the discovery of endogenous intervertebral disc stem/progenitor cells provides potential therapeutic targets for biomaterials based on endogenous repair.
The decellularized matrix of the intervertebral disc retains the microstructure of the ECM, cytokines, and other bioactive components, and exhibits the ideal effects of inducing endogenous cell migration and differentiation, resulting in local tissue repair in vivo.55, 191, 192 Therefore, a decellularized matrix that can motivate endogenous cells is a promising strategy for intervertebral disc regeneration. Moreover, the degenerative intervertebral disc tissue can recruit stem cells by releasing chemotactic cytokines, such as IGF-1, TGF-β, stromal cell-derived factor-1 (SDF-1) and chemokine ligand 5.193-195 These cytokines provide cues which activate the resting endogenous stem/progenitor cells. Albumin/heparin nanoparticles, as an injectable carrier of SDF-1α, achieved sustained release of SDF-1α in intervertebral discs. It was also observed in vivo that SDF-1α induced the migration of endogenous stem cells into degenerative intervertebral discs and restored the disc height and water content.196 Similarly, a compound system, based on the combination of pullulan microbeads, was found to release a variety of cytokines.197 After injection into the intervertebral disc, the sustained release of IGF-1 promoted the migration of endogenous stem cells to the injection site, while TGF-β1 and growth differentiation factor-5 induced stem cells to differentiate into nucleus pulposus cells and replenish local ECM components.197 Biomaterials designed with cytokines can quickly and effectively exert the biological function of endogenous stem/progenitor cells and have broad prospects for application in intervertebral disc regeneration.
It is noteworthy that the intervertebral disc naturally has a microenvironment of persistent pressure, low pH, and hypoxia. When IDD occurs, inflammatory factors aggravate the deterioration of the microenvironment, causing excessive oxidative stress, autophagy, apoptosis, and necrosis of cells,160, 161 which severely impairs the number and function of intervertebral disc stem/progenitor cells and inhibits endogenous repair. Additional regulatory reagents may promote the endogenous repair function of intervertebral disc stem cells by improving the microenvironment of the intervertebral disc. Puerarin relieved the compression-induced apoptosis of the intervertebral disc endogenous stem cells and improved their proliferative activity, which alleviated IDD in a rat model.198 Local release of dexamethasone effectively alleviated inflammation in the intervertebral disc tissue, contributing to the accumulation of MSCs in the intervertebral disc.199 Diuretic amiloride may help endogenous cells survive in acidic environments by blocking acid-sensing channels.200 Small leucine-rich proteoglycans may relieve intervertebral disc stem/progenitor cell apoptosis induced by hypoxia (3.5% oxygen).201 Interestingly, other studies found that a hypoxic environment (2-5% oxygen) may significantly promote cell proliferation and the differentiation of stem cells into nucleus pulposus-like cells, when compared with that in a normoxic environment (20% oxygen).202, 203 The effect of hypoxia on intervertebral disc tissue needs to be further investigated in vivo.
Summary
Endogenous repair, a concept of tissue regeneration depending on the natural endogenous cells in the original tissue, considers the biofunction and living environment of endogenous cells as the critical factors that determine the outcomes of tissue repair. The activation of endogenous cells would eliminate the limitations of exogenous seed cells and provide new strategies for the construction of tissue engineering materials. Therefore, tissue engineering materials that are based on endogenous repair require that considerable attention is paid to bionic microstructures, improved cell survival rates, and modified cell bioactivity.
However, there are many challenges that need to be tackled (Table 1). The quantity and quality of the endogenous cells largely rely on the regional microenvironment. If an injury is too severe or degeneration develops too drastically, overwhelming inflammation and unfavourable environmental factors may lead to irreversible cell death and deactivation,204 particularly in tissues that are naturally cell-depleted, such as intervertebral discs. In addition, many factors (e.g., ageing, inflammation, and mechanical loading) impair the differentiation potential of stem cells. A shortening of the telomere length may explain the failure of aged MSCs to differentiate. Liu et al.205 reported that telomerase knockout MSCs (mTR−/−MSCs) demonstrated a complete failure in terms of the differentiation of mTR−/−MSCs into chondrocytes. Inflammatory cytokines, such as TNF-α and IL-1, inhibit the differentiation of growth plate chondrocytes and longitudinal growth in a mouse model overexpressing TNF-α.206 Excessive mechanical loading on the nucleus pulposus stem cells also leads to a reduced capacity for osteogenic, adipogenic, and chondrogenic differentiation.207 Therefore, strategies should be properly designed to drive the migration of endogenous regenerative cells in order to achieve targeted differentiation and to generate the necessary matrix components and chemokines for ideal regeneration.
Table 1. Challenges in endogenous repair.
| How to maintain the viability and multi-lineage differentiation potential of endogenous stem cells in an injured tissue? |
| How to mobilise the endogenous stem cells to sufficiently proliferate and restore the decreased cell number? |
| How to enable the targeted migration of endogenous stem cells to damaged areas? |
| How to induce the targeted differentiation of endogenous stem cells into progenitors capable of regenerating desired cell types in vivo? |
| How to ensure that newly-generated cells integrate into the surrounding tissues and establish functional connectivity? |
The immunity of biomaterial scaffolds is also an essential issue for tissue regeneration. The interactions between scaffolds and the host are bound to alter the local microenvironment of endogenous cells. For example, degraded scaffolds may provoke monocytes and be harmful to resident cells.208 Thus, the immune reaction after implantation must be considered. The main concern revolves around the question of how to avoid pro-inflammatory reactions and induce a regenerative immune response.209
Finally, although growth factors may be powerful in activating resting cells and promoting endogenous repair, these agents suffer from fragile structural stability and a short in vivo half-life, which limits their therapeutic potential.210 Platelets are natural preservers of many cytokines (PDGF, IGF-1, TGF-β, VEGF, and epidermal growth factor).137 Thus, platelets may be suitable for the maintenance and delivery of cytokines and bioactive substances, but some limitations remain, including the uncontrollable cytokine concentration, insufficient autologous platelets, immune reactions associated with allogeneic proteins, and the lack of standardisation owing to variations in the individual platelet quantity and growth factor composition.211 Some stable chemicals have been found to possess the functions of certain growth factors. For example, kartogenin is a chondrogenic and chondroprotective agent. A kartogenin-conjugated chitosan-hyaluronic acid hydrogel achieved controllable release of kartogenin, promoting stem cell survival and nucleus pulposus regeneration.212 Therefore, developing controllable preservers or chemically/biologically stable agents may be a potential strategy for delivering or substituting growth factors.
Funding Statement
This work was supported by the National Key Research and Development Program of China (No. 2016YFC1100100), the Major Research Plan of the National Natural Science Foundation of China (No. 91649204), China Postdoctoral Science Foundation (No. 2021M701331) and the Scientific Research Training Program for Young Talents from Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, China.
Footnotes
Acknowledgement: None.
Conflicts of interest statement: The authors declare no conflict of interest.
Editor note: Zengwu Shao is an Editorial Board members of Biomaterials Translational. He was blinded from reviewing or making decisions on the manuscript. The article was subject to the journal’s standard procedures, with peer review handled independently of this Editorial Board member and his research groups.
References
- 1.National Science Foundation. The emergence of tissue engineering as a research field. http://www.nsf.gov/pubs/2004/nsf0450/emergence.htm. Accessed May 15, 2021.
- 2.Langer R., Vacanti J. P. Tissue engineering. Science. 1993;260:920–926. doi: 10.1126/science.8493529. [DOI] [PubMed] [Google Scholar]
- 3.Jahromi M., Razavi S., Bakhtiari A. The advances in nerve tissue engineering: From fabrication of nerve conduit to in vivo nerve regeneration assays. J Tissue Eng Regen Med. 2019;13:2077–2100. doi: 10.1002/term.2945. [DOI] [PubMed] [Google Scholar]
- 4.Qasim M., Chae D. S., Lee N. Y. Advancements and frontiers in nano-based 3D and 4D scaffolds for bone and cartilage tissue engineering. Int J Nanomedicine. 2019;14:4333–4351. doi: 10.2147/IJN.S209431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Frueh F. S., Menger M. D., Lindenblatt N., Giovanoli P., Laschke M. W. Current and emerging vascularization strategies in skin tissue engineering. Crit Rev Biotechnol. 2017;37:613–625. doi: 10.1080/07388551.2016.1209157. [DOI] [PubMed] [Google Scholar]
- 6.Goodarzi H., Jadidi K., Pourmotabed S., Sharifi E., Aghamollaei H. Preparation and in vitro characterization of cross-linked collagen-gelatin hydrogel using EDC/NHS for corneal tissue engineering applications. Int J Biol Macromol. 2019;126:620–632. doi: 10.1016/j.ijbiomac.2018.12.125. [DOI] [PubMed] [Google Scholar]
- 7.Berthiaume F., Maguire T. J., Yarmush M. L. Tissue engineering and regenerative medicine: history, progress, and challenges. Annu Rev Chem Biomol Eng. 2011;2:403–430. doi: 10.1146/annurev-chembioeng-061010-114257. [DOI] [PubMed] [Google Scholar]
- 8.Song H. G., Rumma R. T., Ozaki C. K., Edelman E. R., Chen C. S. Vascular tissue engineering: progress, challenges, and clinical promise. Cell Stem Cell. 2018;22:340–354. doi: 10.1016/j.stem.2018.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim H. D., Amirthalingam S., Kim S. L., Lee S. S., Rangasamy J., Hwang N. S. Biomimetic materials and fabrication approaches for bone tissue engineering. Adv Healthc Mater. 2017;6:1700612. doi: 10.1002/adhm.201700612. [DOI] [PubMed] [Google Scholar]
- 10.Katagiri W., Watanabe J., Toyama N., Osugi M., Sakaguchi K., Hibi H. Clinical study of bone regeneration by conditioned medium from mesenchymal stem cells after maxillary sinus floor elevation. Implant Dent. 2017;26:607–612. doi: 10.1097/ID.0000000000000618. [DOI] [PubMed] [Google Scholar]
- 11.Falanga V., Iwamoto S., Chartier M., Yufit T., Butmarc J., Kouttab N., Shrayer D., Carson P. Autologous bone marrow-derived cultured mesenchymal stem cells delivered in a fibrin spray accelerate healing in murine and human cutaneous wounds. Tissue Eng. 2007;13:1299–1312. doi: 10.1089/ten.2006.0278. [DOI] [PubMed] [Google Scholar]
- 12.Yoshikawa T., Ueda Y., Miyazaki K., Koizumi M., Takakura Y. Disc regeneration therapy using marrow mesenchymal cell transplantation: a report of two case studies. Spine (Phila Pa 1976) 2010;35:E475–480. doi: 10.1097/BRS.0b013e3181cd2cf4. [DOI] [PubMed] [Google Scholar]
- 13.Mendonça M. V., Larocca T. F., de Freitas Souza B. S., Villarreal C. F., Silva L. F., Matos A. C., Novaes M. A., Bahia C. M., de Oliveira Melo Martinez A. C., Kaneto C. M., Furtado S. B., Sampaio G. P., Soares M. B., dos Santos R. R. Safety and neurological assessments after autologous transplantation of bone marrow mesenchymal stem cells in subjects with chronic spinal cord injury. Stem Cell Res Ther. 2014;5:126. doi: 10.1186/scrt516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Al-Najar M., Khalil H., Al-Ajlouni J., Al-Antary E., Hamdan M., Rahmeh R., Alhattab D., Samara O., Yasin M., Abdullah A. A., Al-Jabbari E., Hmaid D., Jafar H., Awidi A. Intra-articular injection of expanded autologous bone marrow mesenchymal cells in moderate and severe knee osteoarthritis is safe: a phase I/II study. J Orthop Surg Res. 2017;12:190. doi: 10.1186/s13018-017-0689-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pigott J. H., Ishihara A., Wellman M. L., Russell D. S., Bertone A. L. Investigation of the immune response to autologous, allogeneic, and xenogeneic mesenchymal stem cells after intra-articular injection in horses. Vet Immunol Immunopathol. 2013;156:99–106. doi: 10.1016/j.vetimm.2013.09.003. [DOI] [PubMed] [Google Scholar]
- 16.Pas H. I., Winters M., Haisma H. J., Koenis M. J., Tol J. L., Moen M. H. Stem cell injections in knee osteoarthritis: a systematic review of the literature. Br J Sports Med. 2017;51:1125–1133. doi: 10.1136/bjsports-2016-096793. [DOI] [PubMed] [Google Scholar]
- 17.Jin J. Stem cell treatments. JAMA. 2017;317:330. doi: 10.1001/jama.2016.17822. [DOI] [PubMed] [Google Scholar]
- 18.Sackett S. D., Brown M. E., Tremmel D. M., Ellis T., Burlingham W. J., Odorico J. S. Modulation of human allogeneic and syngeneic pluripotent stem cells and immunological implications for transplantation. Transplant Rev (Orlando) 2016;30:61–70. doi: 10.1016/j.trre.2016.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wells J. M., Watt F. M. Diverse mechanisms for endogenous regeneration and repair in mammalian organs. Nature. 2018;557:322–328. doi: 10.1038/s41586-018-0073-7. [DOI] [PubMed] [Google Scholar]
- 20.Du S. H., Feng Y. Z., Huang Y. X., Guo X. S., Xia D. D. Comparison of pediatric forearm fracture fixation between single- and double-elastic stable intramedullary nailing. Am J Ther. 2016;23:e730–736. doi: 10.1097/MJT.0000000000000031. [DOI] [PubMed] [Google Scholar]
- 21.Majidinia M., Sadeghpour A., Yousefi B. The roles of signaling pathways in bone repair and regeneration. J Cell Physiol. 2018;233:2937–2948. doi: 10.1002/jcp.26042. [DOI] [PubMed] [Google Scholar]
- 22.Dekoninck S., Blanpain C. Stem cell dynamics, migration and plasticity during wound healing. Nat Cell Biol. 2019;21:18–24. doi: 10.1038/s41556-018-0237-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Stenudd M., Sabelström H., Frisén J. Role of endogenous neural stem cells in spinal cord injury and repair. JAMA Neurol. 2015;72:235–237. doi: 10.1001/jamaneurol.2014.2927. [DOI] [PubMed] [Google Scholar]
- 24.Nocera G., Jacob C. Mechanisms of Schwann cell plasticity involved in peripheral nerve repair after injury. Cell Mol Life Sci. 2020;77:3977–3989. doi: 10.1007/s00018-020-03516-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li W., Li L., Hui L. Cell plasticity in liver regeneration. Trends Cell Biol. 2020;30:329–338. doi: 10.1016/j.tcb.2020.01.007. [DOI] [PubMed] [Google Scholar]
- 26.Li C. J., Cheng P., Liang M. K., Chen Y. S., Lu Q., Wang J. Y., Xia Z. Y., Zhou H. D., Cao X., Xie H., Liao E. Y., Luo X. H. MicroRNA-188 regulates age-related switch between osteoblast and adipocyte differentiation. J Clin Invest. 2015;125:1509–1522. doi: 10.1172/JCI77716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Adachi J. D., Lyles K. W., Colón-Emeric C. S., Boonen S., Pieper C. F., Mautalen C., Hyldstrup L., Recknor C., Nordsletten L., Moore K. A., Bucci-Rechtweg C., Su G., Eriksen E. F., Magaziner J. S. Zoledronic acid results in better health-related quality of life following hip fracture: the HORIZON-recurrent fracture trial. Osteoporos Int. 2011;22:2539–2549. doi: 10.1007/s00198-010-1514-9. [DOI] [PubMed] [Google Scholar]
- 28.Kalbasi Anaraki P., Patecki M., Tkachuk S., Kiyan Y., Haller H., Dumler I. Urokinase receptor mediates osteoclastogenesis via M-CSF release from osteoblasts and the c-Fms/PI3K/Akt/NF-κB pathway in osteoclasts. J Bone Miner Res. 2015;30:379–388. doi: 10.1002/jbmr.2350. [DOI] [PubMed] [Google Scholar]
- 29.Sinder B. P., Pettit A. R., McCauley L. K. Macrophages: their emerging roles in bone. J Bone Miner Res. 2015;30:2140–2149. doi: 10.1002/jbmr.2735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alippe Y., Wang C., Ricci B., Xiao J., Qu C., Zou W., Novack D. V., Abu-Amer Y., Civitelli R., Mbalaviele G. Bone matrix components activate the NLRP3 inflammasome and promote osteoclast differentiation. Sci Rep. 2017;7:6630. doi: 10.1038/s41598-017-07014-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Omari S., Makareeva E., Roberts-Pilgrim A., Mirigian L., Jarnik M., Ott C., Lippincott-Schwartz J., Leikin S. Noncanonical autophagy at ER exit sites regulates procollagen turnover. Proc Natl Acad Sci U S A. 2018;115:E10099–E10108. doi: 10.1073/pnas.1814552115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Odkhuu E., Koide N., Haque A., Tsolmongyn B., Naiki Y., Hashimoto S., Komatsu T., Yoshida T., Yokochi T. Inhibition of receptor activator of nuclear factor-κB ligand (RANKL)-induced osteoclast formation by pyrroloquinoline quinine (PQQ) Immunol Lett. 2012;142:34–40. doi: 10.1016/j.imlet.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 33.Roohani-Esfahani S. I., No Y. J., Lu Z., Ng P. Y., Chen Y., Shi J., Pavlos N. J., Zreiqat H. A bioceramic with enhanced osteogenic properties to regulate the function of osteoblastic and osteocalastic cells for bone tissue regeneration. Biomed Mater. 2016;11:035018. doi: 10.1088/1748-6041/11/3/035018. [DOI] [PubMed] [Google Scholar]
- 34.Atanga E., Dolder S., Dauwalder T., Wetterwald A., Hofstetter W. TNFα inhibits the development of osteoclasts through osteoblast-derived GM-CSF. Bone. 2011;49:1090–1100. doi: 10.1016/j.bone.2011.08.003. [DOI] [PubMed] [Google Scholar]
- 35.Ozaki A., Tsunoda M., Kinoshita S., Saura R. Role of fracture hematoma and periosteum during fracture healing in rats: interaction of fracture hematoma and the periosteum in the initial step of the healing process. J Orthop Sci. 2000;5:64–70. doi: 10.1007/s007760050010. [DOI] [PubMed] [Google Scholar]
- 36.Marcucci G., Beltrami G., Tamburini A., Body J. J., Confavreux C. B., Hadji P., Holzer G., Kendler D., Napoli N., Pierroz D. D., Rizzoli R., Brandi M. L. Bone health in childhood cancer: review of the literature and recommendations for the management of bone health in childhood cancer survivors. Ann Oncol. 2019;30:908–920. doi: 10.1093/annonc/mdz120. [DOI] [PubMed] [Google Scholar]
- 37.Alman B. A., Kelley S. P., Nam D. Heal thyself: using endogenous regeneration to repair bone. Tissue Eng Part B Rev. 2011;17:431–436. doi: 10.1089/ten.TEB.2011.0189. [DOI] [PubMed] [Google Scholar]
- 38.Zhang Y., Hao Z., Wang P., Xia Y., Wu J., Xia D., Fang S., Xu S. Exosomes from human umbilical cord mesenchymal stem cells enhance fracture healing through HIF-1α-mediated promotion of angiogenesis in a rat model of stabilized fracture. Cell Prolif. 2019;52:e12570. doi: 10.1111/cpr.12570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fan Y., Hanai J. I., Le P. T., Bi R., Maridas D., DeMambro V., Figueroa C. A., Kir S., Zhou X., Mannstadt M., Baron R., Bronson R. T., Horowitz M. C., Wu J. Y., Bilezikian J. P., Dempster D. W., Rosen C. J., Lanske B. Parathyroid hormone directs bone marrow mesenchymal cell fate. Cell Metab. 2017;25:661–672. doi: 10.1016/j.cmet.2017.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kajimura D., Lee H. W., Riley K. J., Arteaga-Solis E., Ferron M., Zhou B., Clarke C. J., Hannun Y. A., DePinho R. A., Guo X. E., Mann J. J., Karsenty G. Adiponectin regulates bone mass via opposite central and peripheral mechanisms through FoxO1. Cell Metab. 2013;17:901–915. doi: 10.1016/j.cmet.2013.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Funamoto T., Sekimoto T., Murakami T., Kurogi S., Imaizumi K., Chosa E. Roles of the endoplasmic reticulum stress transducer OASIS in fracture healing. Bone. 2011;49:724–732. doi: 10.1016/j.bone.2011.06.012. [DOI] [PubMed] [Google Scholar]
- 42.Wang J., Yang J., Cheng X., Xiao R., Zhao Y., Xu H., Zhu Y., Yan Z., Ommati M. M., Manthari R. K., Wang J. Calcium alleviates fluoride-induced bone damage by inhibiting endoplasmic reticulum stress and mitochondrial dysfunction. J Agric Food Chem. 2019;67:10832–10843. doi: 10.1021/acs.jafc.9b04295. [DOI] [PubMed] [Google Scholar]
- 43.Park J. K., Jang H., Hwang S., Kim E. J., Kim D. E., Oh K. B., Kwon D. J., Koh J. T., Kimura K., Inoue H., Jang W. G., Lee J. W. ER stress-inducible ATF3 suppresses BMP2-induced ALP expression and activation in MC3T3-E1 cells. Biochem Biophys Res Commun. 2014;443:333–338. doi: 10.1016/j.bbrc.2013.11.121. [DOI] [PubMed] [Google Scholar]
- 44.Dai P., Mao Y., Sun X., Li X., Muhammad I., Gu W., Zhang D., Zhou Y., Ni Z., Ma J., Huang S. Attenuation of oxidative stress-induced osteoblast apoptosis by curcumin is associated with preservation of mitochondrial functions and increased Akt-GSK3β signaling. Cell Physiol Biochem. 2017;41:661–677. doi: 10.1159/000457945. [DOI] [PubMed] [Google Scholar]
- 45.Dimitriou R., Tsiridis E., Giannoudis P. V. Current concepts of molecular aspects of bone healing. Injury. 2005;36:1392–1404. doi: 10.1016/j.injury.2005.07.019. [DOI] [PubMed] [Google Scholar]
- 46.Li L., Yang S., Xu L., Li Y., Fu Y., Zhang H., Song J. Nanotopography on titanium promotes osteogenesis via autophagy-mediated signaling between YAP and β-catenin. Acta Biomater. 2019;96:674–685. doi: 10.1016/j.actbio.2019.07.007. [DOI] [PubMed] [Google Scholar]
- 47.Chen X. J., Shen Y. S., He M. C., Yang F., Yang P., Pang F. X., He W., Cao Y. M., Wei Q. S. Polydatin promotes the osteogenic differentiation of human bone mesenchymal stem cells by activating the BMP2-Wnt/β-catenin signaling pathway. Biomed Pharmacother. 2019;112:108746. doi: 10.1016/j.biopha.2019.108746. [DOI] [PubMed] [Google Scholar]
- 48.zur Nieden N. I., Price F. D., Davis L. A., Everitt R. E., Rancourt D. E. Gene profiling on mixed embryonic stem cell populations reveals a biphasic role for beta-catenin in osteogenic differentiation. Mol Endocrinol. 2007;21:674–685. doi: 10.1210/me.2005-0438. [DOI] [PubMed] [Google Scholar]
- 49.Yang M., Li C. J., Sun X., Guo Q., Xiao Y., Su T., Tu M. L., Peng H., Lu Q., Liu Q., He H. B., Jiang T. J., Lei M. X., Wan M., Cao X., Luo X. H. MiR-497∼195 cluster regulates angiogenesis during coupling with osteogenesis by maintaining endothelial Notch and HIF-1α activity. Nat Commun. 2017;8:16003. doi: 10.1038/ncomms16003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fasolino I., Raucci M. G., Soriente A., Demitri C., Madaghiele M., Sannino A., Ambrosio L. Osteoinductive and anti-inflammatory properties of chitosan-based scaffolds for bone regeneration. Mater Sci Eng C Mater Biol Appl. 2019;105:110046. doi: 10.1016/j.msec.2019.110046. [DOI] [PubMed] [Google Scholar]
- 51.Wang Y., Cao L., Liu X. Ghrelin alleviates endoplasmic reticulum stress and inflammation-mediated reproductive dysfunction induced by stress. J Assist Reprod Genet. 2019;36:2357–2366. doi: 10.1007/s10815-019-01589-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ohashi E., Kohno K., Arai N., Harashima A., Ariyasu T., Ushio S. Adenosine N1-oxide exerts anti-inflammatory effects through the PI3K/Akt/GSK-3β signaling pathway and promotes osteogenic and adipocyte differentiation. Biol Pharm Bull. 2019;42:968–976. doi: 10.1248/bpb.b18-00988. [DOI] [PubMed] [Google Scholar]
- 53.He Y. Q., Yang H., Shen Y., Zhang J. H., Zhang Z. G., Liu L. L., Song H. T., Lin B., Hsu H. Y., Qin L. P., Han T., Xin H. L., Zhang Q. Y. Monotropein attenuates ovariectomy and LPS-induced bone loss in mice and decreases inflammatory impairment on osteoblast through blocking activation of NF-κB pathway. Chem Biol Interact. 2018;291:128–136. doi: 10.1016/j.cbi.2018.06.015. [DOI] [PubMed] [Google Scholar]
- 54.Xu L., Zhang L., Wang Z., Li C., Li S., Li L., Fan Q., Zheng L. Melatonin suppresses estrogen deficiency-induced osteoporosis and promotes osteoblastogenesis by inactivating the NLRP3 inflammasome. Calcif Tissue Int. 2018;103:400–410. doi: 10.1007/s00223-018-0428-y. [DOI] [PubMed] [Google Scholar]
- 55.Peng Y., Huang D., Li J., Liu S., Qing X., Shao Z. Genipin-crosslinked decellularized annulus fibrosus hydrogels induces tissue-specific differentiation of bone mesenchymal stem cells and intervertebral disc regeneration. J Tissue Eng Regen Med. 2020;14:497–509. doi: 10.1002/term.3014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Duan H., Song W., Zhao W., Gao Y., Yang Z., Li X. Endogenous neurogenesis in adult mammals after spinal cord injury. Sci China Life Sci. 2016;59:1313–1318. doi: 10.1007/s11427-016-0205-2. [DOI] [PubMed] [Google Scholar]
- 57.Salih E., Wang J., Mah J., Fluckiger R. Natural variation in the extent of phosphorylation of bone phosphoproteins as a function of in vivo new bone formation induced by demineralized bone matrix in soft tissue and bony environments. Biochem J. 2002;364:465–474. doi: 10.1042/BJ20011272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wildemann B., Kadow-Romacker A., Haas N. P., Schmidmaier G. Quantification of various growth factors in different demineralized bone matrix preparations. J Biomed Mater Res A. 2007;81:437–442. doi: 10.1002/jbm.a.31085. [DOI] [PubMed] [Google Scholar]
- 59.Holt D. J., Grainger D. W. Demineralized bone matrix as a vehicle for delivering endogenous and exogenous therapeutics in bone repair. Adv Drug Deliv Rev. 2012;64:1123–1128. doi: 10.1016/j.addr.2012.04.002. [DOI] [PubMed] [Google Scholar]
- 60.Pietrzak W. S., Woodell-May J., McDonald N. Assay of bone morphogenetic protein-2, -4, and -7 in human demineralized bone matrix. J Craniofac Surg. 2006;17:84–90. doi: 10.1097/01.scs.0000179745.91165.73. [DOI] [PubMed] [Google Scholar]
- 61.Hu Q., Liu M., Chen G., Xu Z., Lv Y. Demineralized bone scaffolds with tunable matrix stiffness for efficient bone integration. ACS Appl Mater Interfaces. 2018;10:27669–27680. doi: 10.1021/acsami.8b08668. [DOI] [PubMed] [Google Scholar]
- 62.Chen G., Dong C., Yang L., Lv Y. 3D scaffolds with different stiffness but the same microstructure for bone tissue engineering. ACS Appl Mater Interfaces. 2015;7:15790–15802. doi: 10.1021/acsami.5b02662. [DOI] [PubMed] [Google Scholar]
- 63.Ranly D. M., McMillan J., Keller T., Lohmann C. H., Meunch T., Cochran D. L., Schwartz Z., Boyan B. D. Platelet-derived growth factor inhibits demineralized bone matrix-induced intramuscular cartilage and bone formation. A study of immunocompromised mice. J Bone Joint Surg Am. 2005;87:2052–2064. doi: 10.2106/JBJS.D.02752. [DOI] [PubMed] [Google Scholar]
- 64.Peel S. A., Hu Z. M., Clokie C. M. In search of the ideal bone morphogenetic protein delivery system: in vitro studies on demineralized bone matrix, purified, and recombinant bone morphogenetic protein. J Craniofac Surg. 2003;14:284–291. doi: 10.1097/00001665-200305000-00005. [DOI] [PubMed] [Google Scholar]
- 65.Gombotz W. R., Pankey S. C., Bouchard L. S., Ranchalis J., Puolakkainen P. Controlled release of TGF-beta 1 from a biodegradable matrix for bone regeneration. J Biomater Sci Polym Ed. 1993;5:49–63. doi: 10.1163/156856294x00644. [DOI] [PubMed] [Google Scholar]
- 66.Moxham J. P., Kibblewhite D. J., Bruce A. G., Rigley T., Gillespy T., 3rd, Lane J. Transforming growth factor-beta 1 in a guanidine-extracted demineralized bone matrix carrier rapidly closes a rabbit critical calvarial defect. J Otolaryngol. 1996;25:82–87. [PubMed] [Google Scholar]
- 67.Del Rosario C., Rodríguez-Evora M., Reyes R., González-Orive A., Hernández-Creus A., Shakesheff K. M., White L. J., Delgado A., Evora C. Evaluation of nanostructure and microstructure of bone regenerated by BMP-2-porous scaffolds. J Biomed Mater Res A. 2015;103:2998–3011. doi: 10.1002/jbm.a.35436. [DOI] [PubMed] [Google Scholar]
- 68.Chen B., Lin H., Wang J., Zhao Y., Wang B., Zhao W., Sun W., Dai J. Homogeneous osteogenesis and bone regeneration by demineralized bone matrix loading with collagen-targeting bone morphogenetic protein-2. Biomaterials. 2007;28:1027–1035. doi: 10.1016/j.biomaterials.2006.10.013. [DOI] [PubMed] [Google Scholar]
- 69.Ho S. S., Murphy K. C., Binder B. Y., Vissers C. B., Leach J. K. Increased survival and function of mesenchymal stem cell spheroids entrapped in instructive alginate hydrogels. Stem Cells Transl Med. 2016;5:773–781. doi: 10.5966/sctm.2015-0211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lu Z., Jiang X., Chen M., Feng L., Kang Y. J. An oxygen-releasing device to improve the survival of mesenchymal stem cells in tissue engineering. Biofabrication. 2019;11:045012. doi: 10.1088/1758-5090/ab332a. [DOI] [PubMed] [Google Scholar]
- 71.Hosseinzadeh A., Kamrava S. K., Joghataei M. T., Darabi R., Shakeri-Zadeh A., Shahriari M., Reiter R. J., Ghaznavi H., Mehrzadi S. Apoptosis signaling pathways in osteoarthritis and possible protective role of melatonin. J Pineal Res. 2016;61:411–425. doi: 10.1111/jpi.12362. [DOI] [PubMed] [Google Scholar]
- 72.Lai M., Jin Z., Tang Q., Lu M. Sustained release of melatonin from TiO(2) nanotubes for modulating osteogenic differentiation of mesenchymal stem cells in vitro. J Biomater Sci Polym Ed. 2017;28:1651–1664. doi: 10.1080/09205063.2017.1342334. [DOI] [PubMed] [Google Scholar]
- 73.Hoemann C. D., Chen G., Marchand C., Tran-Khanh N., Thibault M., Chevrier A., Sun J., Shive M. S., Fernandes M. J., Poubelle P. E., Centola M., El-Gabalawy H. Scaffold-guided subchondral bone repair: implication of neutrophils and alternatively activated arginase-1+ macrophages. Am J Sports Med. 2010;38:1845–1856. doi: 10.1177/0363546510369547. [DOI] [PubMed] [Google Scholar]
- 74.Deng M., Tan J., Hu C., Hou T., Peng W., Liu J., Yu B., Dai Q., Zhou J., Yang Y., Dong R., Ruan C., Dong S., Xu J. Modification of PLGA scaffold by MSC-derived extracellular matrix combats macrophage inflammation to initiate bone regeneration via TGF-β-induced protein. Adv Healthc Mater. 2020;9:e2000353. doi: 10.1002/adhm.202000353. [DOI] [PubMed] [Google Scholar]
- 75.Knudson C. B., Knudson W. Cartilage proteoglycans. Semin Cell Dev Biol. 2001;12:69–78. doi: 10.1006/scdb.2000.0243. [DOI] [PubMed] [Google Scholar]
- 76.Pacifici M., Koyama E., Iwamoto M., Gentili C. Development of articular cartilage: what do we know about it and how may it occur? Connect Tissue Res. 2000;41:175–184. doi: 10.3109/03008200009005288. [DOI] [PubMed] [Google Scholar]
- 77.Huang C. C., Chiou C. H., Liu S. C., Hu S. L., Su C. M., Tsai C. H., Tang C. H. Melatonin attenuates TNF-α and IL-1β expression in synovial fibroblasts and diminishes cartilage degradation: Implications for the treatment of rheumatoid arthritis. J Pineal Res. 2019;66:e12560. doi: 10.1111/jpi.12560. [DOI] [PubMed] [Google Scholar]
- 78.Kapoor M., Martel-Pelletier J., Lajeunesse D., Pelletier J. P., Fahmi H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011;7:33–42. doi: 10.1038/nrrheum.2010.196. [DOI] [PubMed] [Google Scholar]
- 79.Liu Y., Zou R., Wang Z., Wen C., Zhang F., Lin F. Exosomal KLF3-AS1 from hMSCs promoted cartilage repair and chondrocyte proliferation in osteoarthritis. Biochem J. 2018;475:3629–3638. doi: 10.1042/BCJ20180675. [DOI] [PubMed] [Google Scholar]
- 80.Barbero A., Ploegert S., Heberer M., Martin I. Plasticity of clonal populations of dedifferentiated adult human articular chondrocytes. Arthritis Rheum. 2003;48:1315–1325. doi: 10.1002/art.10950. [DOI] [PubMed] [Google Scholar]
- 81.Alsalameh S., Amin R., Gemba T., Lotz M. Identification of mesenchymal progenitor cells in normal and osteoarthritic human articular cartilage. Arthritis Rheum. 2004;50:1522–1532. doi: 10.1002/art.20269. [DOI] [PubMed] [Google Scholar]
- 82.Hiraoka K., Grogan S., Olee T., Lotz M. Mesenchymal progenitor cells in adult human articular cartilage. Biorh. 2006;43:447–454. [PubMed] [Google Scholar]
- 83.Seol D., McCabe D. J., Choe H., Zheng H., Yu Y., Jang K., Walter M. W., Lehman A. D., Ding L., Buckwalter J. A., Martin J. A. Chondrogenic progenitor cells respond to cartilage injury. Arthritis Rheum. 2012;64:3626–3637. doi: 10.1002/art.34613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.McCarthy H. E., Bara J. J., Brakspear K., Singhrao S. K., Archer C. W. The comparison of equine articular cartilage progenitor cells and bone marrow-derived stromal cells as potential cell sources for cartilage repair in the horse. Vet J. 2012;192:345–351. doi: 10.1016/j.tvjl.2011.08.036. [DOI] [PubMed] [Google Scholar]
- 85.Ozbey O., Sahin Z., Acar N., Ustunel I. Distribution of CD105 and CD166 positive cells in the proximal epiphysis of developing rat humerus. Histol Histopathol. 2010;25:1437–1445. doi: 10.14670/HH-25.1437. [DOI] [PubMed] [Google Scholar]
- 86.Fickert S., Fiedler J., Brenner R. E. Identification of subpopulations with characteristics of mesenchymal progenitor cells from human osteoarthritic cartilage using triple staining for cell surface markers. Arthritis Res Ther. 2004;6:R422–432. doi: 10.1186/ar1210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Churchill J. L., Krych A. J., Lemos M. J., Redd M., Bonner K. F. A case series of successful repair of articular cartilage fragments in the knee. Am J Sports Med. 2019;47:2589–2595. doi: 10.1177/0363546519865497. [DOI] [PubMed] [Google Scholar]
- 88.García-Arnandis I., Guillén M. I., Castejón M. A., Gomar F., Alcaraz M. J. Haem oxygenase-1 down-regulates high mobility group box 1 and matrix metalloproteinases in osteoarthritic synoviocytes. Rheumatology (Oxford) 2010;49:854–861. doi: 10.1093/rheumatology/kep463. [DOI] [PubMed] [Google Scholar]
- 89.Joos H., Wildner A., Hogrefe C., Reichel H., Brenner R. E. Interleukin-1 beta and tumor necrosis factor alpha inhibit migration activity of chondrogenic progenitor cells from non-fibrillated osteoarthritic cartilage. Arthritis Res Ther. 2013;15:R119. doi: 10.1186/ar4299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Mishima Y., Lotz M. Chemotaxis of human articular chondrocytes and mesenchymal stem cells. J Orthop Res. 2008;26:1407–1412. doi: 10.1002/jor.20668. [DOI] [PubMed] [Google Scholar]
- 91.Schnabel M., Marlovits S., Eckhoff G., Fichtel I., Gotzen L., Vécsei V., Schlegel J. Dedifferentiation-associated changes in morphology and gene expression in primary human articular chondrocytes in cell culture. Osteoarthritis Cartilage. 2002;10:62–70. doi: 10.1053/joca.2001.0482. [DOI] [PubMed] [Google Scholar]
- 92.Miller R. E., Scanzello C. R., Malfait A. M. An emerging role for Toll-like receptors at the neuroimmune interface in osteoarthritis. Semin Immunopathol. 2019;41:583–594. doi: 10.1007/s00281-019-00762-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Mills C. D., Kincaid K., Alt J. M., Heilman M. J., Hill A. M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J Immunol. 2000;164:6166–6173. doi: 10.4049/jimmunol.1701141. [DOI] [PubMed] [Google Scholar]
- 94.Kraus V. B., McDaniel G., Huebner J. L., Stabler T. V., Pieper C. F., Shipes S. W., Petry N. A., Low P. S., Shen J., McNearney T. A., Mitchell P. Direct in vivo evidence of activated macrophages in human osteoarthritis. Osteoarthritis Cartilage. 2016;24:1613–1621. doi: 10.1016/j.joca.2016.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Scanzello C. R., Goldring S. R. The role of synovitis in osteoarthritis pathogenesis. Bone. 2012;51:249–257. doi: 10.1016/j.bone.2012.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wang T., He C. Pro-inflammatory cytokines: The link between obesity and osteoarthritis. Cytokine Growth Factor Rev. 2018;44:38–50. doi: 10.1016/j.cytogfr.2018.10.002. [DOI] [PubMed] [Google Scholar]
- 97.Li Y. S., Luo W., Zhu S. A., Lei G. H. T cells in osteoarthritis: alterations and beyond. Front Immunol. 2017;8:356. doi: 10.3389/fimmu.2017.00356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Glyn-Jones S., Palmer A. J., Agricola R., Price A. J., Vincent T. L., Weinans H., Carr A. J. Osteoarthritis. Lancet. 2015;386:376–387. doi: 10.1016/S0140-6736(14)60802-3. [DOI] [PubMed] [Google Scholar]
- 99.Imada K., Oka H., Kawasaki D., Miura N., Sato T., Ito A. Anti-arthritic action mechanisms of natural chondroitin sulfate in human articular chondrocytes and synovial fibroblasts. Biol Pharm Bull. 2010;33:410–414. doi: 10.1248/bpb.33.410. [DOI] [PubMed] [Google Scholar]
- 100.Cheleschi S., Fioravanti A., De Palma A., Corallo C., Franci D., Volpi N., Bedogni G., Giannotti S., Giordano N. Methylsulfonylmethane and mobilee prevent negative effect of IL-1β in human chondrocyte cultures via NF-κB signaling pathway. Int Immunopharmacol. 2018;65:129–139. doi: 10.1016/j.intimp.2018.10.004. [DOI] [PubMed] [Google Scholar]
- 101.Zhuang C., Wang Y., Zhang Y., Xu N. Oxidative stress in osteoarthritis and antioxidant effect of polysaccharide from angelica sinensis. Int J Biol Macromol. 2018;115:281–286. doi: 10.1016/j.ijbiomac.2018.04.083. [DOI] [PubMed] [Google Scholar]
- 102.Ertürk C., Altay M. A., Selek S., Koçyiğit A. Paraoxonase-1 activity and oxidative status in patients with knee osteoarthritis and their relationship with radiological and clinical parameters. Scand J Clin Lab Invest. 2012;72:433–439. doi: 10.3109/00365513.2012.687116. [DOI] [PubMed] [Google Scholar]
- 103.Courties A., Gualillo O., Berenbaum F., Sellam J. Metabolic stress-induced joint inflammation and osteoarthritis. Osteoarthritis Cartilage. 2015;23:1955–1965. doi: 10.1016/j.joca.2015.05.016. [DOI] [PubMed] [Google Scholar]
- 104.Lepetsos P., Papavassiliou K. A., Papavassiliou A. G. Redox and NF-κB signaling in osteoarthritis. Free Radic Biol Med. 2019;132:90–100. doi: 10.1016/j.freeradbiomed.2018.09.025. [DOI] [PubMed] [Google Scholar]
- 105.Shapouri-Moghaddam A., Mohammadian S., Vazini H., Taghadosi M., Esmaeili S. A., Mardani F., Seifi B., Mohammadi A., Afshari J. T., Sahebkar A. Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol. 2018;233:6425–6440. doi: 10.1002/jcp.26429. [DOI] [PubMed] [Google Scholar]
- 106.Laskin D. L., Sunil V. R., Gardner C. R., Laskin J. D. Macrophages and tissue injury: agents of defense or destruction? Annu Rev Pharmacol Toxicol. 2011;51:267–288. doi: 10.1146/annurev.pharmtox.010909.105812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Ackerman J. E., Geary M. B., Orner C. A., Bawany F., Loiselle A. E. Obesity/Type II diabetes alters macrophage polarization resulting in a fibrotic tendon healing response. PLoS One. 2017;12:e0181127. doi: 10.1371/journal.pone.0181127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Zhang S., Chuah S. J., Lai R. C., Hui J. H. P., Lim S. K., Toh W. S. MSC exosomes mediate cartilage repair by enhancing proliferation, attenuating apoptosis and modulating immune reactivity. Biomaterials. 2018;156:16–27. doi: 10.1016/j.biomaterials.2017.11.028. [DOI] [PubMed] [Google Scholar]
- 109.Dai M., Sui B., Xue Y., Liu X., Sun J. Cartilage repair in degenerative osteoarthritis mediated by squid type II collagen via immunomodulating activation of M2 macrophages, inhibiting apoptosis and hypertrophy of chondrocytes. Biomaterials. 2018;180:91–103. doi: 10.1016/j.biomaterials.2018.07.011. [DOI] [PubMed] [Google Scholar]
- 110.Chu J., Yan B., Zhang J., Peng L., Ao X., Zheng Z., Jiang T., Zhang Z. Casticin attenuates osteoarthritis-related cartilage degeneration by inhibiting the ROS-mediated NF-κB signaling pathway in vitro and in vivo. Inflammation. 2020;43:810–820. doi: 10.1007/s10753-019-01167-y. [DOI] [PubMed] [Google Scholar]
- 111.Hu S. L., Wang K., Shi Y. F., Shao Z. X., Zhang C. X., Sheng K. W., Ge Z. D., Chen J. X., Wang X. Y. Downregulating Akt/NF-κB signaling and its antioxidant activity with Loureirin A for alleviating the progression of osteoarthritis: In vitro and vivo studies. Int Immunopharmacol. 2020;78:105953. doi: 10.1016/j.intimp.2019.105953. [DOI] [PubMed] [Google Scholar]
- 112.Gleghorn J. P., Jones A. R., Flannery C. R., Bonassar L. J. Boundary mode lubrication of articular cartilage by recombinant human lubricin. J Orthop Res. 2009;27:771–777. doi: 10.1002/jor.20798. [DOI] [PubMed] [Google Scholar]
- 113.Flannery C. R., Zollner R., Corcoran C., Jones A. R., Root A., Rivera-Bermúdez M. A., Blanchet T., Gleghorn J. P., Bonassar L. J., Bendele A. M., Morris E. A., Glasson S. S. Prevention of cartilage degeneration in a rat model of osteoarthritis by intraarticular treatment with recombinant lubricin. Arthritis Rheum. 2009;60:840–847. doi: 10.1002/art.24304. [DOI] [PubMed] [Google Scholar]
- 114.Sophia Fox A. J., Bedi A., Rodeo S. A. The basic science of articular cartilage: structure, composition, and function. Sports Health. 2009;1:461–468. doi: 10.1177/1941738109350438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chuah Y. J., Peck Y., Lau J. E., Hee H. T., Wang D. A. Hydrogel based cartilaginous tissue regeneration: recent insights and technologies. Biomater Sci. 2017;5:613–631. doi: 10.1039/c6bm00863a. [DOI] [PubMed] [Google Scholar]
- 116.Shields K. J., Beckman M. J., Bowlin G. L., Wayne J. S. Mechanical properties and cellular proliferation of electrospun collagen type II. Tissue Eng. 2004;10:1510–1517. doi: 10.1089/ten.2004.10.1510. [DOI] [PubMed] [Google Scholar]
- 117.Liu S., Wu J., Liu X., Chen D., Bowlin G. L., Cao L., Lu J., Li F., Mo X., Fan C. Osteochondral regeneration using an oriented nanofiber yarn-collagen type I/hyaluronate hybrid/TCP biphasic scaffold. J Biomed Mater Res A. 2015;103:581–592. doi: 10.1002/jbm.a.35206. [DOI] [PubMed] [Google Scholar]
- 118.Dong C., Lv Y. Application of collagen scaffold in tissue engineering: recent advances and new perspectives. Polymers (Basel) 2016;8:42. doi: 10.3390/polym8020042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Mohan N., Mohanan P. V., Sabareeswaran A., Nair P. Chitosan-hyaluronic acid hydrogel for cartilage repair. Int J Biol Macromol. 2017;104:1936–1945. doi: 10.1016/j.ijbiomac.2017.03.142. [DOI] [PubMed] [Google Scholar]
- 120.Pulkkinen H. J., Tiitu V., Valonen P., Jurvelin J. S., Lammi M. J., Kiviranta I. Engineering of cartilage in recombinant human type II collagen gel in nude mouse model in vivo. Osteoarthritis Cartilage. 2010;18:1077–1087. doi: 10.1016/j.joca.2010.05.004. [DOI] [PubMed] [Google Scholar]
- 121.Marquass B., Somerson J. S., Hepp P., Aigner T., Schwan S., Bader A., Josten C., Zscharnack M., Schulz R. M. A novel MSC-seeded triphasic construct for the repair of osteochondral defects. J Orthop Res. 2010;28:1586–1599. doi: 10.1002/jor.21173. [DOI] [PubMed] [Google Scholar]
- 122.Leone G., Volpato M. D., Nelli N., Lamponi S., Boanini E., Bigi A., Magnani A. Continuous multilayered composite hydrogel as osteochondral substitute. J Biomed Mater Res A. 2015;103:2521–2530. doi: 10.1002/jbm.a.35389. [DOI] [PubMed] [Google Scholar]
- 123.Mallick S. P., Singh B. N., Rastogi A., Srivastava P. Design and evaluation of chitosan/poly(l-lactide)/pectin based composite scaffolds for cartilage tissue regeneration. Int J Biol Macromol. 2018;112:909–920. doi: 10.1016/j.ijbiomac.2018.02.049. [DOI] [PubMed] [Google Scholar]
- 124.Duan P., Pan Z., Cao L., He Y., Wang H., Qu Z., Dong J., Ding J. The effects of pore size in bilayered poly(lactide-co-glycolide) scaffolds on restoring osteochondral defects in rabbits. J Biomed Mater Res A. 2014;102:180–192. doi: 10.1002/jbm.a.34683. [DOI] [PubMed] [Google Scholar]
- 125.Yang J., Zhang Y. S., Yue K., Khademhosseini A. Cell-laden hydrogels for osteochondral and cartilage tissue engineering. Acta Biomater. 2017;57:1–25. doi: 10.1016/j.actbio.2017.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Peng Y., Qing X., Lin H., Huang D., Li J., Tian S., Liu S., Lv X., Ma K., Li R., Rao Z., Bai Y., Chen S., Lei M., Quan D., Shao Z. Decellularized disc hydrogels for hBMSCs tissue-specific differentiation and tissue regeneration. Bioact Mater. 2021;6:3541–3556. doi: 10.1016/j.bioactmat.2021.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Antons J., Marascio M. G., Aeberhard P., Weissenberger G., Hirt-Burri N., Applegate L. A., Bourban P. E., Pioletti D. P. Decellularised tissues obtained by a CO(2)-philic detergent and supercritical CO(2) Eur Cell Mater. 2018;36:81–95. doi: 10.22203/eCM.v036a07. [DOI] [PubMed] [Google Scholar]
- 128.Sun Y., Yan L., Chen S., Pei M. Functionality of decellularized matrix in cartilage regeneration: A comparison of tissue versus cell sources. Acta Biomater. 2018;74:56–73. doi: 10.1016/j.actbio.2018.04.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Pati F., Jang J., Ha D. H., Won Kim S., Rhie J. W., Shim J. H., Kim D. H., Cho D. W. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat Commun. 2014;5:3935. doi: 10.1038/ncomms4935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Sutherland A. J., Beck E. C., Dennis S. C., Converse G. L., Hopkins R. A., Berkland C. J., Detamore M. S. Decellularized cartilage may be a chondroinductive material for osteochondral tissue engineering. PLoS One. 2015;10:e0121966. doi: 10.1371/journal.pone.0121966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Almeida H. V., Eswaramoorthy R., Cunniffe G. M., Buckley C. T., O’Brien F. J., Kelly D. J. Fibrin hydrogels functionalized with cartilage extracellular matrix and incorporating freshly isolated stromal cells as an injectable for cartilage regeneration. Acta Biomater. 2016;36:55–62. doi: 10.1016/j.actbio.2016.03.008. [DOI] [PubMed] [Google Scholar]
- 132.Shintani N., Hunziker E. B. Chondrogenic differentiation of bovine synovium: bone morphogenetic proteins 2 and 7 and transforming growth factor beta1 induce the formation of different types of cartilaginous tissue. Arthritis Rheum. 2007;56:1869–1879. doi: 10.1002/art.22701. [DOI] [PubMed] [Google Scholar]
- 133.Holland T. A., Bodde E. W., Cuijpers V. M., Baggett L. S., Tabata Y., Mikos A. G., Jansen J. A. Degradable hydrogel scaffolds for in vivo delivery of single and dual growth factors in cartilage repair. Osteoarthritis Cartilage. 2007;15:187–197. doi: 10.1016/j.joca.2006.07.006. [DOI] [PubMed] [Google Scholar]
- 134.Patil A. S., Sable R. B., Kothari R. M. Role of insulin-like growth factors (IGFs), their receptors and genetic regulation in the chondrogenesis and growth of the mandibular condylar cartilage. J Cell Physiol. 2012;227:1796–1804. doi: 10.1002/jcp.22905. [DOI] [PubMed] [Google Scholar]
- 135.Fischer J., Knoch N., Sims T., Rosshirt N., Richter W. Time-dependent contribution of BMP, FGF, IGF, and HH signaling to the proliferation of mesenchymal stroma cells during chondrogenesis. J Cell Physiol. 2018;233:8962–8970. doi: 10.1002/jcp.26832. [DOI] [PubMed] [Google Scholar]
- 136.Boushell M. K., Mosher C. Z., Suri G. K., Doty S. B., Strauss E. J., Hunziker E. B., Lu H. H. Polymeric mesh and insulin-like growth factor 1 delivery enhance cell homing and graft-cartilage integration. Ann N Y Acad Sci. 2019;1442:138–152. doi: 10.1111/nyas.14054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Lacci K. M., Dardik A. Platelet-rich plasma: support for its use in wound healing. Yale J Biol Med. 2010;83:1–9. [PMC free article] [PubMed] [Google Scholar]
- 138.Lindeboom J. A., Mathura K. R., Aartman I. H., Kroon F. H., Milstein D. M., Ince C. Influence of the application of platelet-enriched plasma in oral mucosal wound healing. Clin Oral Implants Res. 2007;18:133–139. doi: 10.1111/j.1600-0501.2006.01288.x. [DOI] [PubMed] [Google Scholar]
- 139.Barbon S., Stocco E., Macchi V., Contran M., Grandi F., Borean A., Parnigotto P. P., Porzionato A., De Caro R. Platelet-rich fibrin scaffolds for cartilage and tendon regenerative medicine: from bench to bedside. Int J Mol Sci. 2019;20:1701. doi: 10.3390/ijms20071701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Chang N. J., Erdenekhuyag Y., Chou P. H., Chu C. J., Lin C. C., Shie M. Y. Therapeutic effects of the addition of platelet-rich plasma to bioimplants and early rehabilitation exercise on articular cartilage repair. Am J Sports Med. 2018;46:2232–2241. doi: 10.1177/0363546518780955. [DOI] [PubMed] [Google Scholar]
- 141.Anderson J. M., Rodriguez A., Chang D. T. Foreign body reaction to biomaterials. Semin Immunol. 2008;20:86–100. doi: 10.1016/j.smim.2007.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Vasconcelos D. M., Gonçalves R. M., Almeida C. R., Pereira I. O., Oliveira M. I., Neves N., Silva A. M., Ribeiro A. C., Cunha C., Almeida A. R., Ribeiro C. C., Gil A. M., Seebach E., Kynast K. L., Richter W., Lamghari M., Santos S. G., Barbosa M. A. Fibrinogen scaffolds with immunomodulatory properties promote in vivo bone regeneration. Biomaterials. 2016;111:163–178. doi: 10.1016/j.biomaterials.2016.10.004. [DOI] [PubMed] [Google Scholar]
- 143.Revati R., Abdul Majid M. S., Ridzuan M. J. M., Normahira M., Mohd Nasir N. F., Rahman Y. M., Gibson A. G. Mechanical, thermal and morphological characterisation of 3D porous Pennisetum purpureum/PLA biocomposites scaffold. Mater Sci Eng C Mater Biol Appl. 2017;75:752–759. doi: 10.1016/j.msec.2017.02.127. [DOI] [PubMed] [Google Scholar]
- 144.Harrington S., Williams J., Rawal S., Ramachandran K., Stehno-Bittel L. Hyaluronic acid/collagen hydrogel as an alternative to alginate for long-term immunoprotected islet transplantation. Tissue Eng Part A. 2017;23:1088–1099. doi: 10.1089/ten.tea.2016.0477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Lu H. T., Chang W. T., Tsai M. L., Chen C. H., Chen W. Y., Mi F. L. Development of injectable fucoidan and biological macromolecules hybrid hydrogels for intra-articular delivery of platelet-rich plasma. Mar Drugs. 2019;17:236. doi: 10.3390/md17040236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Zhao Y., Wei C., Chen X., Liu J., Yu Q., Liu Y., Liu J. Drug delivery system based on near-infrared light-responsive molybdenum disulfide nanosheets controls the high-efficiency release of dexamethasone to inhibit inflammation and treat osteoarthritis. ACS Appl Mater Interfaces. 2019;11:11587–11601. doi: 10.1021/acsami.8b20372. [DOI] [PubMed] [Google Scholar]
- 147.Alini M., Eisenstein S. M., Ito K., Little C., Kettler A. A., Masuda K., Melrose J., Ralphs J., Stokes I., Wilke H. J. Are animal models useful for studying human disc disorders/degeneration? Eur Spine J. 2008;17:2–19. doi: 10.1007/s00586-007-0414-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Daly C., Ghosh P., Jenkin G., Oehme D., Goldschlager T. A review of animal models of intervertebral disc degeneration: pathophysiology, regeneration, and translation to the clinic. Biomed Res Int. 2016;2016:5952165. doi: 10.1155/2016/5952165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Ao X., Wang L., Shao Y., Chen X., Zhang J., Chu J., Jiang T., Zhang Z., Huang M. Development and characterization of a novel bipedal standing mouse model of intervertebral disc and facet joint degeneration. Clin Orthop Relat Res. 2019;477:1492–1504. doi: 10.1097/CORR.0000000000000712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Walter B. A., Korecki C. L., Purmessur D., Roughley P. J., Michalek A. J., Iatridis J. C. Complex loading affects intervertebral disc mechanics and biology. Osteoarthritis Cartilage. 2011;19:1011–1018. doi: 10.1016/j.joca.2011.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Hartman R., Patil P., Tisherman R., St Croix C., Niedernhofer L. J., Robbins P. D., Ambrosio F., Van Houten B., Sowa G., Vo N. Age-dependent changes in intervertebral disc cell mitochondria and bioenergetics. Eur Cell Mater. 2018;36:171–183. doi: 10.22203/eCM.v036a13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Lyu F. J., Cheung K. M., Zheng Z., Wang H., Sakai D., Leung V. Y. IVD progenitor cells: a new horizon for understanding disc homeostasis and repair. Nat Rev Rheumatol. 2019;15:102–112. doi: 10.1038/s41584-018-0154-x. [DOI] [PubMed] [Google Scholar]
- 153.Henriksson H., Thornemo M., Karlsson C., Hägg O., Junevik K., Lindahl A., Brisby H. Identification of cell proliferation zones, progenitor cells and a potential stem cell niche in the intervertebral disc region: a study in four species. Spine (Phila Pa 1976) 2009;34:2278–2287. doi: 10.1097/BRS.0b013e3181a95ad2. [DOI] [PubMed] [Google Scholar]
- 154.Henriksson H. B., Svala E., Skioldebrand E., Lindahl A., Brisby H. Support of concept that migrating progenitor cells from stem cell niches contribute to normal regeneration of the adult mammal intervertebral disc: a descriptive study in the New Zealand white rabbit. Spine (Phila Pa 1976) 2012;37:722–732. doi: 10.1097/BRS.0b013e318231c2f7. [DOI] [PubMed] [Google Scholar]
- 155.Sasaki N., Henriksson H. B., Runesson E., Larsson K., Sekiguchi M., Kikuchi S., Konno S., Rydevik B., Brisby H. Physical exercise affects cell proliferation in lumbar intervertebral disc regions in rats. Spine (Phila Pa 1976) 2012;37:1440–1447. doi: 10.1097/BRS.0b013e31824ff87d. [DOI] [PubMed] [Google Scholar]
- 156.Shi R., Wang F., Hong X., Wang Y. T., Bao J. P., Cai F., Wu X. T. The presence of stem cells in potential stem cell niches of the intervertebral disc region: an in vitro study on rats. Eur Spine J. 2015;24:2411–2424. doi: 10.1007/s00586-015-4168-7. [DOI] [PubMed] [Google Scholar]
- 157.Grunhagen T., Shirazi-Adl A., Fairbank J. C., Urban J. P. Intervertebral disk nutrition: a review of factors influencing concentrations of nutrients and metabolites. Orthop Clin North Am. 2011;42:465–477. doi: 10.1016/j.ocl.2011.07.010. vii. [DOI] [PubMed] [Google Scholar]
- 158.Holm S., Maroudas A., Urban J. P., Selstam G., Nachemson A. Nutrition of the intervertebral disc: solute transport and metabolism. Connect Tissue Res. 1981;8:101–119. doi: 10.3109/03008208109152130. [DOI] [PubMed] [Google Scholar]
- 159.Risbud M. V., Shapiro I. M. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat Rev Rheumatol. 2014;10:44–56. doi: 10.1038/nrrheum.2013.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Li Z., Chen S., Ma K., Lv X., Lin H., Hu B., He R., Shao Z. CsA attenuates compression-induced nucleus pulposus mesenchymal stem cells apoptosis via alleviating mitochondrial dysfunction and oxidative stress. Life Sci. 2018;205:26–37. doi: 10.1016/j.lfs.2018.05.014. [DOI] [PubMed] [Google Scholar]
- 161.Chen S., Deng X., Ma K., Zhao L., Huang D., Li Z., Shao Z. Icariin improves the viability and function of cryopreserved human nucleus pulposus-derived mesenchymal stem cells. Oxid Med Cell Longev. 2018;2018:3459612. doi: 10.1155/2018/3459612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Patil P., Falabella M., Saeed A., Lee D., Kaufman B., Shiva S., Croix C. S., Van Houten B., Niedernhofer L. J., Robbins P. D., Lee J., Gwendolyn S., Vo N. V. Oxidative stress-induced senescence markedly increases disc cell bioenergetics. Mech Ageing Dev. 2019;180:97–106. doi: 10.1016/j.mad.2019.04.006. [DOI] [PubMed] [Google Scholar]
- 163.Childs B. G., Gluscevic M., Baker D. J., Laberge R. M., Marquess D., Dananberg J., van Deursen J. M. Senescent cells: an emerging target for diseases of ageing. Nat Rev Drug Discov. 2017;16:718–735. doi: 10.1038/nrd.2017.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Huang D., Peng Y., Li Z., Chen S., Deng X., Shao Z., Ma K. Compression-induced senescence of nucleus pulposus cells by promoting mitophagy activation via the PINK1/PARKIN pathway. J Cell Mol Med. 2020;24:5850–5864. doi: 10.1111/jcmm.15256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Chen D., Xia D., Pan Z., Xu D., Zhou Y., Wu Y., Cai N., Tang Q., Wang C., Yan M., Zhang J. J., Zhou K., Wang Q., Feng Y., Wang X., Xu H., Zhang X., Tian N. Metformin protects against apoptosis and senescence in nucleus pulposus cells and ameliorates disc degeneration in vivo. Cell Death Dis. 2016;7:e2441. doi: 10.1038/cddis.2016.334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Xia C., Zeng Z., Fang B., Tao M., Gu C., Zheng L., Wang Y., Shi Y., Fang C., Mei S., Chen Q., Zhao J., Lin X., Fan S., Jin Y., Chen P. Mesenchymal stem cell-derived exosomes ameliorate intervertebral disc degeneration via anti-oxidant and anti-inflammatory effects. Free Radic Biol Med. 2019;143:1–15. doi: 10.1016/j.freeradbiomed.2019.07.026. [DOI] [PubMed] [Google Scholar]
- 167.Nakamichi R., Ito Y., Inui M., Onizuka N., Kayama T., Kataoka K., Suzuki H., Mori M., Inagawa M., Ichinose S., Lotz M. K., Sakai D., Masuda K., Ozaki T., Asahara H. Mohawk promotes the maintenance and regeneration of the outer annulus fibrosus of intervertebral discs. Nat Commun. 2016;7:12503. doi: 10.1038/ncomms12503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Yao M., Zhang J., Li Z., Guo S., Zhou X., Zhang W. Marein protects human nucleus pulposus cells against high glucose-induced injury and extracellular matrix degradation at least partly by inhibition of ROS/NF-κB pathway. Int Immunopharmacol. 2020;80:106126. doi: 10.1016/j.intimp.2019.106126. [DOI] [PubMed] [Google Scholar]
- 169.Saraiya M., Nasser R., Zeng Y., Addya S., Ponnappan R. K., Fortina P., Anderson D. G., Albert T. J., Shapiro I. M., Risbud M. V. Reversine enhances generation of progenitor-like cells by dedifferentiation of annulus fibrosus cells. Tissue Eng Part A. 2010;16:1443–1455. doi: 10.1089/ten.tea.2009.0343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Tendulkar G., Chen T., Ehnert S., Kaps H. P., Nüssler A. K. Intervertebral disc nucleus repair: hype or hope? Int J Mol Sci. 2019;20:3622. doi: 10.3390/ijms20153622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Roberts S., Ayad S., Menage P. J. Immunolocalisation of type VI collagen in the intervertebral disc. Ann Rheum Dis. 1991;50:787–791. doi: 10.1136/ard.50.11.787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Newell N., Little J. P., Christou A., Adams M. A., Adam C. J., Masouros S. D. Biomechanics of the human intervertebral disc: A review of testing techniques and results. J Mech Behav Biomed Mater. 2017;69:420–434. doi: 10.1016/j.jmbbm.2017.01.037. [DOI] [PubMed] [Google Scholar]
- 173.Clouet J., Grimandi G., Pot-Vaucel M., Masson M., Fellah H. B., Guigand L., Cherel Y., Bord E., Rannou F., Weiss P., Guicheux J., Vinatier C. Identification of phenotypic discriminating markers for intervertebral disc cells and articular chondrocytes. Rheumatology (Oxford) 2009;48:1447–1450. doi: 10.1093/rheumatology/kep262. [DOI] [PubMed] [Google Scholar]
- 174.Li J., Liu C., Guo Q., Yang H., Li B. Regional variations in the cellular, biochemical, and biomechanical characteristics of rabbit annulus fibrosus. PLoS One. 2014;9:e91799. doi: 10.1371/journal.pone.0091799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Guerin H. A., Elliott D. M. Degeneration affects the fiber reorientation of human annulus fibrosus under tensile load. J Biomech. 2006;39:1410–1418. doi: 10.1016/j.jbiomech.2005.04.007. [DOI] [PubMed] [Google Scholar]
- 176.Klein J. A., Hukins D. W. Collagen fibre orientation in the annulus fibrosus of intervertebral disc during bending and torsion measured by x-ray diffraction. Biochim Biophys Acta. 1982;719:98–101. doi: 10.1016/0304-4165(82)90312-9. [DOI] [PubMed] [Google Scholar]
- 177.Pearce R. H., Grimmer B. J., Adams M. E. Degeneration and the chemical composition of the human lumbar intervertebral disc. J Orthop Res. 1987;5:198–205. doi: 10.1002/jor.1100050206. [DOI] [PubMed] [Google Scholar]
- 178.Hee H. T., Chuah Y. J., Tan B. H., Setiobudi T., Wong H. K. Vascularization and morphological changes of the endplate after axial compression and distraction of the intervertebral disc. Spine (Phila Pa 1976) 2011;36:505–511. doi: 10.1097/BRS.0b013e3181d32410. [DOI] [PubMed] [Google Scholar]
- 179.Growney Kalaf E. A., Flores R., Bledsoe J. G., Sell S. A. Characterization of slow-gelling alginate hydrogels for intervertebral disc tissue-engineering applications. Mater Sci Eng C Mater Biol Appl. 2016;63:198–210. doi: 10.1016/j.msec.2016.02.067. [DOI] [PubMed] [Google Scholar]
- 180.Li Z., Lang G., Chen X., Sacks H., Mantzur C., Tropp U., Mader K. T., Smallwood T. C., Sammon C., Richards R. G., Alini M., Grad S. Polyurethane scaffold with in situ swelling capacity for nucleus pulposus replacement. Biomaterials. 2016;84:196–209. doi: 10.1016/j.biomaterials.2016.01.040. [DOI] [PubMed] [Google Scholar]
- 181.Woiciechowsky C., Abbushi A., Zenclussen M. L., Casalis P., Krüger J. P., Freymann U., Endres M., Kaps C. Regeneration of nucleus pulposus tissue in an ovine intervertebral disc degeneration model by cell-free resorbable polymer scaffolds. J Tissue Eng Regen Med. 2014;8:811–820. doi: 10.1002/term.1582. [DOI] [PubMed] [Google Scholar]
- 182.Priyadarshani P., Li Y., Yang S., Yao L. Injectable hydrogel provides growth-permissive environment for human nucleus pulposus cells. J Biomed Mater Res A. 2016;104:419–426. doi: 10.1002/jbm.a.35580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Feng G., Jin X., Hu J., Ma H., Gupte M. J., Liu H., Ma P. X. Effects of hypoxias and scaffold architecture on rabbit mesenchymal stem cell differentiation towards a nucleus pulposus-like phenotype. Biomaterials. 2011;32:8182–8189. doi: 10.1016/j.biomaterials.2011.07.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Zhou X., Wang J., Fang W., Tao Y., Zhao T., Xia K., Liang C., Hua J., Li F., Chen Q. Genipin cross-linked type II collagen/chondroitin sulfate composite hydrogel-like cell delivery system induces differentiation of adipose-derived stem cells and regenerates degenerated nucleus pulposus. Acta Biomater. 2018;71:496–509. doi: 10.1016/j.actbio.2018.03.019. [DOI] [PubMed] [Google Scholar]
- 185.Zhou X., Wang J., Huang X., Fang W., Tao Y., Zhao T., Liang C., Hua J., Chen Q., Li F. Injectable decellularized nucleus pulposus-based cell delivery system for differentiation of adipose-derived stem cells and nucleus pulposus regeneration. Acta Biomater. 2018;81:115–128. doi: 10.1016/j.actbio.2018.09.044. [DOI] [PubMed] [Google Scholar]
- 186.Huang Y. Z., Cai J. Q., Lv F. J., Xie H. L., Yang Z. M., Huang Y. C., Deng L. Species variation in the spontaneous calcification of bone marrow-derived mesenchymal stem cells. Cytotherapy. 2013;15:323–329. doi: 10.1016/j.jcyt.2012.11.011. [DOI] [PubMed] [Google Scholar]
- 187.Vadalè G., Sowa G., Hubert M., Gilbertson L. G., Denaro V., Kang J. D. Mesenchymal stem cells injection in degenerated intervertebral disc: cell leakage may induce osteophyte formation. J Tissue Eng Regen Med. 2012;6:348–355. doi: 10.1002/term.433. [DOI] [PubMed] [Google Scholar]
- 188.Liu C., Zhu C., Li J., Zhou P., Chen M., Yang H., Li B. The effect of the fibre orientation of electrospun scaffolds on the matrix production of rabbit annulus fibrosus-derived stem cells. Bone Res. 2015;3:15012. doi: 10.1038/boneres.2015.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Yuan C., Wang J., Zhu X., Zheng Y., Huang B., Li C., Zhou Y. Stress regulating osteogenic differentiation of human intervertebral disc cartilage endplate-derived stem cells. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2015;29:351–355. [PubMed] [Google Scholar]
- 190.Lv F., Lu M., Cheung K. M., Leung V. Y., Zhou G. Intrinsic properties of mesemchymal stem cells from human bone marrow, umbilical cord and umbilical cord blood comparing the different sources of MSC. Curr Stem Cell Res Ther. 2012;7:389–399. doi: 10.2174/157488812804484611. [DOI] [PubMed] [Google Scholar]
- 191.Liu C., Jin Z., Ge X., Zhang Y., Xu H. Decellularized annulus fibrosus matrix/chitosan hybrid hydrogels with basic fibroblast growth factor for annulus fibrosus tissue engineering. Tissue Eng Part A. 2019;25:1605–1613. doi: 10.1089/ten.tea.2018.0297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Xu J., Liu S., Wang S., Qiu P., Chen P., Lin X., Fang X. Decellularised nucleus pulposus as a potential biologic scaffold for disc tissue engineering. Mater Sci Eng C Mater Biol Appl. 2019;99:1213–1225. doi: 10.1016/j.msec.2019.02.045. [DOI] [PubMed] [Google Scholar]
- 193.Illien-Jünger S., Pattappa G., Peroglio M., Benneker L. M., Stoddart M. J., Sakai D., Mochida J., Grad S., Alini M. Homing of mesenchymal stem cells in induced degenerative intervertebral discs in a whole organ culture system. Spine (Phila Pa 1976) 2012;37:1865–1873. doi: 10.1097/BRS.0b013e3182544a8a. [DOI] [PubMed] [Google Scholar]
- 194.Baek S. J., Kang S. K., Ra J. C. In vitro migration capacity of human adipose tissue-derived mesenchymal stem cells reflects their expression of receptors for chemokines and growth factors. Exp Mol Med. 2011;43:596–603. doi: 10.3858/emm.2011.43.10.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Lee M. J., Kim J., Kim M. Y., Bae Y. S., Ryu S. H., Lee T. G., Kim J. H. Proteomic analysis of tumor necrosis factor-alpha-induced secretome of human adipose tissue-derived mesenchymal stem cells. J Proteome Res. 2010;9:1754–1762. doi: 10.1021/pr900898n. [DOI] [PubMed] [Google Scholar]
- 196.Zhang H., Yu S., Zhao X., Mao Z., Gao C. Stromal cell-derived factor-1α-encapsulated albumin/heparin nanoparticles for induced stem cell migration and intervertebral disc regeneration in vivo. Acta Biomater. 2018;72:217–227. doi: 10.1016/j.actbio.2018.03.032. [DOI] [PubMed] [Google Scholar]
- 197.Frapin L., Clouet J., Chédeville C., Moraru C., Samarut E., Henry N., André M., Bord E., Halgand B., Lesoeur J., Fusellier M., Guicheux J., Le Visage C. Controlled release of biological factors for endogenous progenitor cell migration and intervertebral disc extracellular matrix remodelling. Biomaterials. 2020;253:120107. doi: 10.1016/j.biomaterials.2020.120107. [DOI] [PubMed] [Google Scholar]
- 198.Huang D., Peng Y., Ma K., Qing X., Deng X., Li Z., Shao Z. Puerarin relieved compression-induced apoptosis and mitochondrial dysfunction in human nucleus pulposus mesenchymal stem cells via the PI3K/Akt pathway. Stem Cells Int. 2020;2020:7126914. doi: 10.1155/2020/7126914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Liang C. Z., Li H., Tao Y. Q., Peng L. H., Gao J. Q., Wu J. J., Li F. C., Hua J. M., Chen Q. X. Dual release of dexamethasone and TGF-β3 from polymeric microspheres for stem cell matrix accumulation in a rat disc degeneration model. Acta Biomater. 2013;9:9423–9433. doi: 10.1016/j.actbio.2013.08.019. [DOI] [PubMed] [Google Scholar]
- 200.Liu J., Tao H., Wang H., Dong F., Zhang R., Li J., Ge P., Song P., Zhang H., Xu P., Liu X., Shen C. Biological behavior of human nucleus pulposus mesenchymal stem cells in response to changes in the acidic environment during intervertebral disc degeneration. Stem Cells Dev. 2017;26:901–911. doi: 10.1089/scd.2016.0314. [DOI] [PubMed] [Google Scholar]
- 201.Huang S., Leung V. Y., Long D., Chan D., Lu W. W., Cheung K. M., Zhou G. Coupling of small leucine-rich proteoglycans to hypoxic survival of a progenitor cell-like subpopulation in Rhesus Macaque intervertebral disc. Biomaterials. 2013;34:6548–6558. doi: 10.1016/j.biomaterials.2013.05.027. [DOI] [PubMed] [Google Scholar]
- 202.Ni L., Liu X., Sochacki K. R., Ebraheim M., Fahrenkopf M., Shi Q., Liu J., Yang H. Effects of hypoxia on differentiation from human placenta-derived mesenchymal stem cells to nucleus pulposus-like cells. Spine J. 2014;14:2451–2458. doi: 10.1016/j.spinee.2014.03.028. [DOI] [PubMed] [Google Scholar]
- 203.Zhang Z., Li F., Tian H., Guan K., Zhao G., Shan J., Ren D. Differentiation of adipose-derived stem cells toward nucleus pulposus-like cells induced by hypoxia and a three-dimensional chitosan-alginate gel scaffold in vitro. Chin Med J (Engl) 2014;127:314–321. [PubMed] [Google Scholar]
- 204.Ma K., Chen S., Li Z., Deng X., Huang D., Xiong L., Shao Z. Mechanisms of endogenous repair failure during intervertebral disc degeneration. Osteoarthritis Cartilage. 2019;27:41–48. doi: 10.1016/j.joca.2018.08.021. [DOI] [PubMed] [Google Scholar]
- 205.Liu L., DiGirolamo C. M., Navarro P. A., Blasco M. A., Keefe D. L. Telomerase deficiency impairs differentiation of mesenchymal stem cells. Exp Cell Res. 2004;294:1–8. doi: 10.1016/j.yexcr.2003.10.031. [DOI] [PubMed] [Google Scholar]
- 206.Mårtensson K., Chrysis D., Sävendahl L. Interleukin-1beta and TNF-alpha act in synergy to inhibit longitudinal growth in fetal rat metatarsal bones. J Bone Miner Res. 2004;19:1805–1812. doi: 10.1359/JBMR.040805. [DOI] [PubMed] [Google Scholar]
- 207.Liang H., Chen S., Huang D., Deng X., Ma K., Shao Z. Effect of compression loading on human nucleus pulposus-derived mesenchymal stem cells. Stem Cells Int. 2018;2018:1481243. doi: 10.1155/2018/1481243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Dahms K., Sharkova Y., Heitland P., Pankuweit S., Schaefer J. R. Cobalt intoxication diagnosed with the help of Dr House. Lancet. 2014;383:574. doi: 10.1016/S0140-6736(14)60037-4. [DOI] [PubMed] [Google Scholar]
- 209.Goodman S. B., Pajarinen J., Yao Z., Lin T. Inflammation and bone repair: from particle disease to tissue regeneration. Front Bioeng Biotechnol. 2019;7:230. doi: 10.3389/fbioe.2019.00230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Johnson K. E., Makanji Y., Temple-Smith P., Kelly E. K., Barton P. A., Al-Musawi S. L., Mueller T. D., Walton K. L., Harrison C. A. Biological activity and in vivo half-life of pro-activin A in male rats. Mol Cell Endocrinol. 2016;422:84–92. doi: 10.1016/j.mce.2015.12.007. [DOI] [PubMed] [Google Scholar]
- 211.Etulain J. Platelets in wound healing and regenerative medicine. Platelets. 2018;29:556–568. doi: 10.1080/09537104.2018.1430357. [DOI] [PubMed] [Google Scholar]
- 212.Zhu Y., Tan J., Zhu H., Lin G., Yin F., Wang L., Song K., Wang Y., Zhou G., Yi W. Development of kartogenin-conjugated chitosan-hyaluronic acid hydrogel for nucleus pulposus regeneration. Biomater Sci. 2017;5:784–791. doi: 10.1039/c7bm00001d. [DOI] [PubMed] [Google Scholar]