Abstract
Antibiotic resistance is a major public health threat of the twenty-first century and represents an important risk to the global economy. Healthcare-associated infections mainly caused by drug-resistant bacteria are wreaking havoc in patient care worldwide. The spread of such pathogens limits the utility of available drugs and complicates the treatment of bacterial diseases. As a result, there is an urgent need for new drugs with mechanisms of action capable of curbing resistance. Plants synthesize and utilize various metabolic compounds to deter pathogens and predators. Utilizing these plant-based metabolites is a promising option in identifying novel bioactive compounds that could be harnessed to develop new potent antimicrobial drugs to treat multidrug-resistant pathogens. The purpose of this review is to highlight medicinal plants as important sources of novel antimicrobial agents that could be developed to help combat antimicrobial resistance.
Keywords: Antimicrobial resistance, Plant metabolites, Plant secondary metabolites, Multidrug-resistant pathogens, Plant-based medical compounds, Anti-infective agents, Antibacterial drug screening
Introduction
Bacterial resistance to antibiotics constitutes one of the most important and urgent public health threats of the twenty-first century [1]. Infections caused by multidrug-resistant (MDR) pathogens are associated with increased mortality compared to those caused by drug-susceptible bacteria. The U.S. Center for Disease Control and Prevention (CDC) has designated antibiotic resistance as an important burden on the U.S. healthcare system, and over $20 billion are spent on treatment cost every year [2]. MDR pathogens are projected to cause about 300 million premature deaths worldwide and up to $100 trillion loss to the global economy by 2050 [3, 4].
Given the threat posed by drug-resistant bacteria, there is an urgent need for novel compounds with diverse mechanisms of action capable of limiting antimicrobial resistance. Secondary plant metabolites are one of the unexplored sources of antimicrobial agents in nature. It is estimated that less than 1% of the global tropical plant species have been screened for pharmaceutical applications [5] and investigated phytochemically [6]. Given the spread of multidrug-resistant pathogens and the dwindling number of available antibiotics, there is renewed interest in utilizing plant-based sources to identify potent novel antimicrobial agents.
The declining potency of antibiotics
Antibiotics are among the most frequently prescribed drugs in modern medicine and have been used to treat bacterial infections since 1940s [7–9]. Bacterial resistance to antibiotics was first predicted by Alexander Fleming in 1945 during his Nobel Prize acceptance speech: “The time may come when penicillin can be bought by anyone in the shops. Then there is the danger that the ignorant man may easily under-dose himself and by exposing his microbes to nonlethal quantities of the drug make them resistant”. Many other factors may lead to resistance, including overuse of broad-spectrum antibiotics, and lack of early identification of causative pathogens and their antimicrobial susceptibility patterns. Additionally, heavy use of antibiotics in agriculture and intensive animal farming promote development of antibiotic resistance [10, 11]. These factors, together with poor infection control, are the leading culprits in the increasing spread of resistance [7, 12, 13]. While antibiotic resistance has mainly been a clinical problem in healthcare settings, recent studies show existence of resistant pathogens in both primary care patients and community settings [2]. This has been exacerbated by easy access to antibiotics in many developing countries, where one can go to any pharmacy and obtain any form of drug without prescription. Such practices either lead to overuse or underuse of broad-spectrum drugs, thereby increasing the risk of resistance and turn-over rates. Alarmingly, this has contributed to rapid development of resistance and rapid loss of effectiveness of new antibiotics, usually within five years of introduction into the market [14]. Figure 1 demonstrates the turn-over rates of various antibiotics from 1940 to 2015.
Fig. 1.

Timeline of antibiotics discovery and year of first observed resistance [15–33]
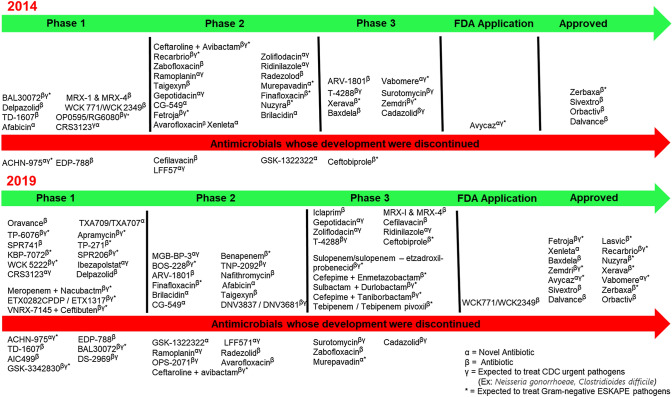
Most bacterial pathogens utilize various resistance mechanisms to render antibiotics ineffective. These include the use of efflux pumps, inactivating enzymes, target modification, and microenvironment modifications [34]. These antibiotic resistance mechanisms pose serious challenges to the pharmaceutical industry in developing new drugs. The process of developing new antibiotics is time-consuming and extremely costly. As of December 2019, a total of 41 antibiotics were in development (15 in Phase 1 clinical trials, 12 in Phase 2, 13 in Phase 3, 1 submitted for FDA application), and 14 approved. It is estimated that only 60% of drugs that enter Phase 3 clinical trials will be approved. Figure 2 shows the list of antibiotics in the pipeline between 2014 and 2019 as well as those that have been discontinued. Given the mismatch between the rate at which bacteria develop resistance and the slow pace of new drug development, the world may soon run out of effective antibiotics. As a result, there is renewed interest in identifying potent new bioactive compounds with the hope to develop novel antibiotics that are less amenable to bacterial resistance.
Fig. 2.
Antibiotic development pipeline from 2014 to 2019. As of December 2019, a total of 41 antibiotics were in development (15 in Phase 1 clinical trials, 12 in Phase 2, 13 in Phase 3, 1 submitted for FDA application), and 14 approved. It is estimated that only 60% of drugs that enter Phase 3 will be approved for treatment (pewtrusts.org). New antibiotic development involves time and resources and there are very few novel antibiotics under development. The declining number of antibiotics in the development pipeline, in part, reflects the challenges associated with its development. At the same time, bacteria that survives antibiotic treatment are spreading
Developing potent novel drugs from plants sources
The use of medicinal plants in controlling diseases has been documented throughout the history of man. Traditionally, different parts of plants (leaf, stem, bark, root, fruit) have been used to treat, prevent, and control several diseases [35]. The World Health Organization (WHO) has prioritized the search for new antibacterial agents against multidrug-resistant ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species) [36]. These rapidly evolving pathogens are responsible for most of the cases of hospital-acquired infections globally [36]. Over the years, several medicinal plant extracts and secondary metabolites have been explored for their efficacy against these pathogens [37]. Some of these are:
Different parts of Adiantum capillus-veneris and leaf extract of Artemisia absinthium have shown inhibitory effects against E. faecium and S. aureus [38].
Leaf extracts of Aloe ferox, Cynodon dactylon, Acacia nilotica, bud of Syzygium aromaticum, and seed and leaf of Theobroma cacao were active against Klebsiella pneumoniae [39–41].
Leaf extracts of Mentha sp. and Aloe vera and root of Zingiber officinale significantly inhibited P. aeruginosa growth [42].
Root of Piper longum, stem of Kalanchoe fedtschenkoi, and fruit extract of Martynia annua were all found to be active against A. baumannii, [43–45].
Leaf and seed extracts of Dacryodes edulis have activity against E. cloacae [46].
Leaf extracts of Ipomoea batatas and Hibiscus esculentus, leaf and seed extracts of Dacryodes edulis, bark of Azadirachta indica have inhibitory effects against E. aerogenes [46].
In addition to their antibacterial properties, medicinal plants have also been used in traditional medicine for the treatment of both human and animal fungal diseases [47]. The increased use of antifungal agents in addition to the spread of multidrug-resistant fungi, and limited number of drugs available has precipitated an interest in new classes of antifungal drugs. Recent reports showed anti-fungal activities of several medicinal plants against different fungal species, including Candida albicans, Aspergillus species, Trichophyton species, Microscopium species, penicillium species, Fusarium species, Epidermophyton species, and Rhodotorula ruba [48]. Some of these plants are:
Leaf extracts of Eugenia uniflora, Psidium guajava, Curcuma longa, Piptadenia colubrina, Persea americana showed activity against C. albicans, C. dubliniensis, C. glabrata, and C. krusei [49].
Leaf extract of Alibertia macrophylla exhibits inhibitory effects against Cladosporium sphaerospermum, C. cladosporioides, A. niger, and Colletotrichum gloeosporioides [49].
Leaf extract of Piper regnellii inhibits growth of Trichophyton rubrum, Trichophyton mentagrophytes, and Microsporum canis [50].
Root extract of Rubia tinctorum was active against A. niger, Alternaria alternaria, P. verrucosum, and Mucor mucedo [51].
Different parts of Tithonia diversifolia were active against Microbotryum violaceum and Chlorella fusca [52].
Seed of Cassia tora showed inhibitory activity against Botrytis cinerea, Erysiphe graminis, Phytophthora infestans, Puccinia recondita, and Pyricularia grisea [53].
Leaves and twigs of Chamaecyparis pisifera showed activity against P. oryzae [54].
The antiviral activities of medicinal plants have also been evaluated. The toxic side effects and ineffective response to the available antiviral drugs, especially in the wake of the coronavirus pandemic, has prioritized the development of potent agents to control deadly viral infections. Medicinal plants have been shown to possess potent antiviral agents with various activities against HIV, HBV, and several other viruses [55–59]. Exploring these plants and their bioactive metabolites will be a cost effective and secure way to develop new potent antiviral agents to combat viral diseases. Interestingly, 80% of the chronic Hepatitis B patients in China still rely on medicinal plants as primary treatment [60]. Some of these plants with antiviral activity showed similar or better efficacy against viruses than the available treatment options [60]. Among the reported medicinal plants with antiviral properties include:
Bulb of Allium sativum L. has demonstrated potent antiviral activities against ADV-3, ADV-41, DENV, SARS-CoV-2, HSV-I and II, HCMV, H9N2, IBV, H1N1, CBV-3, ECHO, EV-71, HRV-2, HAV, MeV, PIV-3, VV, [61].
Leaf of Justicia adhatoda L. was active against SARS-CoV-2, influenza virus, and HSV [60, 62].
Rhizome of Cyperus rotundus L. inhibited SARS-CoV-2, HAV, HSV-I, and CVB [63, 64].
Leaf of Ocimum basilicum L. was active against HIV-I, HSV, ADV-3, 8, 11, HVB, EV, and CVB-I [65–67].
Many of these plant extracts act by inhibiting viral replication, enhancing cellular immunity, inhibiting virus-cell attachment, inducing apoptosis of viral-infected cells, disrupting viral envelopes, inhibiting viral RNA and DNA synthesis, downregulating the expression of important host proteins, and inhibiting viral attachment to host cell surface [68].
Plants are rich in secondary metabolites and are a major source of chemical diversity, thus, may be promising sources of untapped potent antibacterial agents. Phytochemical analyses of some of these medicinal plants show different active groups, such as flavonoids, quinones, lignans, stilbenes, tannins, alkaloids, terpenes, polyphenolics, and coumarins [69], most of which are antibacterial in nature. For instance, phenol derivatives inhibit bacterial growth by either reducing the pH, increasing membrane permeability, or altering efflux pumping [70]. Phenolic compounds, one of the important secondary metabolites, have shown to act on many bacterial targets including cytoplasmic membrane damage, topoisomerase inhibition, NADH-reductase and ATP synthase inhibition [71]. Tannins have also been shown to induce bacterial membrane damage and metabolism inactivation [72]. Flavonoids, in turn may promote formation of extracellular complex soluble proteins and inhibit cell wall proteins as well as metabolism and DNA synthesis [73]. These mechanisms of action associated with plant secondary metabolic compounds make them promising agents to be harnessed to develop novel drugs to combat the growing problem of antimicrobial resistance.
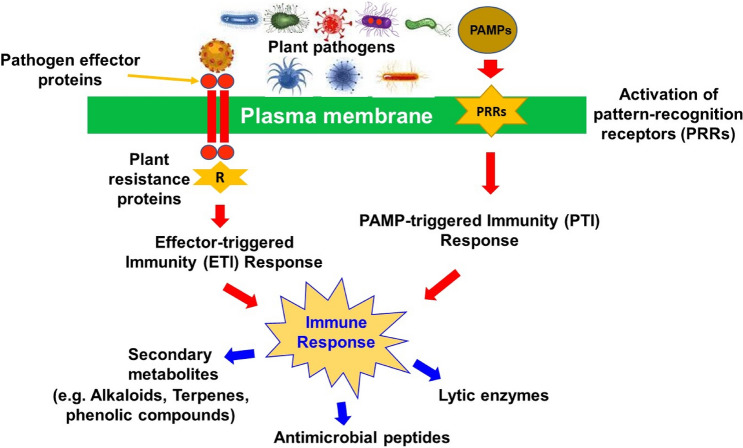
Plant secondary metabolites are usually produced as defensive mechanisms against predators, plant pathogens, insects, and animals. During response to pathogens, surface receptors present on plants detect infecting agents by recognizing specific patterns and chemical motifs [74]. Plants detect bacteria using either pathogen associated molecular patterns (PAMPs) or pathogen effectors (Fig. 3). The PAMPS are sensed by pattern-recognition receptors present on plant cell surfaces, which in turn activates a signaling cascade leading to PAMP-triggered immunity, the primary immune response in plants [75]. Bacteria can, however, interfere with PAMP-triggered immunity by injecting effector molecules into the plant cell. These effectors are recognized by plant intracellular protein complexes such as the nucleotide-binding leucine-rich repeat receptors, resulting in a hypersensitive response known as effector-triggered immunity, the secondary immune response in plants [76, 77]. These mechanisms either limit pathogen entry, restrict pathogen propagation, or kill pathogens within the host plant cells. Once a pathogen is identified, plant cells also protect themselves by either reinforcing cell wall biosynthesis of lytic enzymes, producing secondary metabolites, or other pathogenesis related proteins [78].
Fig. 3.
Plant immune response to pathogens. Bacteria are detected by either pathogen associated molecular patterns (PAMPs) or pathogen effectors: I The PAMPS activates the pattern-recognition receptors (PRRs) on the plant cell surface, which in turn activates a signaling cascade leading to PAMP-triggered immunity (PTI). II Pathogen effectors are recognized by plant resistance proteins, resulting in a hypersensitive response known as effector-triggered immunity (ETI). Together, these defense mechanisms result in the release of various secondary metabolites that ultimately kill the infecting pathogen. Given their novelty to human pathogens, these plant-derived antimicrobial secondary compounds can be harnessed to combat multidrug-resistant pathogens
Several bioactive compounds and their derivatives have been used as drugs for the treatment of different diseases, including cancer, hypertension, immuno-suppression, neurological diseases, fungal, viral and bacterial infections; some of which are either currently under clinical trials or already in the market [79]. Importantly, these compounds have demonstrated promising results in fighting the emergence of antibiotic resistant bacteria [72] and increasing the potency of old antibiotics through synergistic association, thus, preventing the development of resistance [80]. Some examples are:
Berberine: an isoquinolone isolated from plants such as Rhizoma coptidis. Berberine is known to possess activity against methicillin-resistant Staphylococcus aureus (MRSA) by inhibiting adhesion to human gingival fibroblasts, an important step during biofilm development [81]. In addition to its ability to inhibit biofilm formation, several studies have reported a positive synergistic activity of berberine when combined with other antibiotics. For instance, addition of berberine to azithromycin and levofloxacin lowers its minimum inhibitory concentration by 50.0–96.9% [82], as well as decreases adhesion and intracellular invasion of MRSA [83].
Piperine: a piperidine-type alkaloid was isolated from the Piper species (Piper nigrum, Piper longum). This compound has strong antimicrobial activity against both Gram positive and negative bacteria (S. aureus, Bacillus subtilis, Salmonella sp and Escherichia coli) [84] and acts as an efflux pump inhibitor in S. aureus when combined with ciprofloxacin [85].
Allicin: a sulfur-containing compound that is obtained from raw garlic (Allium sativum). Allicin has been shown to exhibit broad-spectrum antimicrobial activity against both Gram-positive and negative bacteria, including MRSA, Streptococcus spp., E. coli, and Salmonella enterica serovar Typhimurium [86]. Allicin acts through S-allylmercapto modification of thiol-containing proteins in bacteria, leading to reduction of glutathione levels, induction of protein aggregation, and inactivation of essential enzymes [86–88].
Ajoene: another organosulfur found abundantly in oil-macerated garlic. Ajoene exhibits antibacterial activity against several Gram-positive and Gram-negative bacteria, including H. pylori, Mycobacterium species, however, its antimicrobial property was more observed in Gram-positives [89]. The mechanism of action of this compound is similar to that of allicin. The use of this compound to treat antibiotic resistant organisms is promising as it is now produced by total synthesis [90].
Eugenol (4-allyl-2-methoxyphenol): a hydroxyphenyl propene, naturally occurring in essential oils from several plants belonging to the Lamiaceae, Lauraceae, Myrtaceae, and Myristicaceae families [91]. Several mechanisms of action of Eugenol has been reported, including inhibition of Streptococci biofilm and enterotoxin formation, disruption of Salmonella typhi cell membrane, and reduction of S. aureus toxin gene expression [92]. In addition, Eugenol has also been reported to inhibit production of bacterial virulence factors, such as violacein, elastase, pyocyanin [93].
Resveratrol (3,5,4′-trihydroxystilbene) is a naturally occurring polyphenolic antioxidant that has received massive attention for its potential health benefits. It can be extracted from different plant species, such as grapevines, pines, bananas, beans, pomegranates, peanuts, and soybeans, The antimicrobial activity of resveratrol has not been fully studied. However, it exhibits antibacterial activity against several Gram-positive and Gram-negative foodborne bacteria by inhibiting gene expression [94]. Resveratrol also inhibits toxin production, biofilm formation, motility and interferes with quorum sensing in a wide range of bacterial, viral and fungal species [95].
These plant-derived metabolites could potentially be harnessed as novel drugs to combat antibiotic-resistant bacteria due to their natural origin with no history of prolonged exposure to human pathogens. To our knowledge, no resistance to plant-based compounds has been recorded to date. Moreover, secondary metabolites from plants have different active moieties and offers a repertoire of different activities that may be utilized against different bacterial targets [96]. Many cost-effective approaches are available to identify, quantify and characterize the bioactive plant compounds for further investigation as potential new drug molecules [97]. These include the use of spectroscopy, gas chromatography, high-pressure liquid chromatography, and thin-layer chromatography to provide improved extraction efficiency, yield, extraction time, selectivity, and sensitivity in quantitation [98, 99].
Conclusion
Antimicrobial resistance is a major global health problem. This has been precipitated by rapid development and spread of resistant mechanisms resulting in loss of effectiveness of new antibiotics, usually within five years of introduction into the market [14]. As of today, no effective drug is available to reverse antibiotic resistance in bacteria. Several approaches have been undertaken to control bacterial resistance, including controlling antibiotic prescription, enhanced antimicrobial stewardship programs to improve antibiotic therapy, and developing new drugs. Another important approach, but less studied is to harness plant-based compounds. Plants are rich in several antimicrobial secondary metabolites and may be a rich source of potent drugs with a variety of chemical moieties that could target different resistant mechanisms in bacteria. Several plant species have already been reported to show potential antimicrobial effects against multidrug-resistant bacteria (Table 1). A deeper understanding of the mechanisms of action of these plant-derived compounds is needed. Harnessing secondary plants metabolites would be a cost-effective and innovative strategy to develop next generation novel antimicrobials and/or improve current antimicrobials to combat the emerging threat of antibiotic resistance, develop databases for plant metabolites, and their possible antimicrobial targets.
Table 1.
Examples of plants with known activity against multidrug-resistant pathogens. These plant-based metabolites provide promising option to develop novel drugs against multidrug-resistant pathogens
| Plant name and part | Extract type | Resistant bacteria | Evaluation method | Source, geographical location |
|---|---|---|---|---|
|
Moringa oleifera (Leaves) Metricaria recutita (Flowers) |
Water, Ethanol, Methanol |
Clinical MDR, XDR, PDR isolates: Escherichia coli Klebsiella spp P. aeruginosa Proteus mirabilis S. aureus S. epidermidis |
Microbroth dilution Disc diffusion | Farm in El-Fayoum governorate, Egypt [100] |
| Scutellaria barbata (Herbs) | Water extracts | Clinical MDR Acinetobacter baumannii | Disc diffusion, time-kill assays, murine lung infection model | Herb store in Kaohsiung City, Taiwan [101] |
|
Allexis cauliflora (leaves) Persea Americana (Stones) Entada Africana (bark) Pentaclethra macrophylla (Bark) |
CH2Cl2/MeOH MeOH C4H8O2 Extracts | Kanamycin-resistant E. coli AG100A | Microbroth dilution | Different regions of Cameroon [102] |
| Entada abyssinica (Leaves and roots) | Clinical MDR Klebsiella pneumoniae Kp55 | |||
| Pentaclethra macrophylla (Bark) | Clinical MDR Providencia stuartii-NAE16 | |||
|
Alkanna tentoria (leaves) |
Aqueous, chloroform, ethanol and hexane extracts | A. baumannii, E. coli, P. aeruginosa S. aureus | Well diffusion | Charsadda region, Pakistan [103] |
| Artemisia absinthium (Bark) | Aqueous Ethanol Extracts | Enterococcus faecium, Staphylococcus aureus | Disc diffusion, time-kill assays | Sudhnoti district, Northern Pakistan [104] |
| Martynia annua (bulk) | Enterococcus faecium,Staphylococcus aureus, Acinetobacter baumannii | Disc diffusion, time-kill assays | ||
| Adiantum capillus-venaris (Bark) | Enterococcus faecium, Staphylococcus aureus | Disc diffusion, time-kill assays | ||
| Zanthoxylum armatum (Bark) | Enterococcus faecium, Staphylococcus aureus | Time-kill assays | ||
| Swertia chirata (Bark) | Staphylococcus aureus | Disc diffusion, time-kill assays |
Funding
This work was supported by NIH R01 Grants R01AI116914, R01AI150685, Molecular Basis of Infectious Diseases Training Grant from the NIH Institute of Allergy and Infectious Diseases (T32AI055449) and Evans-Allen Grant from the USDA National Institute of Food and Agriculture (200094-20116 XX-140/ALAX-011-0816).
Declarations
Conflict of Interest
None of the authors have any conflict to declare.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.World Health Organization (2013) Traditional medicine strategy 2014–2023. World Health Organization (WHO), 1–76. https://www.who.int/medicines/publications/traditional/trm_strategy14_23/en/
- 2.Munita JM, Arias CA. Mechanisms of antibiotic resistance. Microbiol Spectrum. 2016 doi: 10.1128/microbiolspec.VMBF-0016-2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Center for Disease Control & Prevention (2015) Healthcare-associated infections (HAIs). www.cdc.gov/hai/data/archive/2015-HAI-data-report.html
- 4.Huixue J, Liuyi L, Weiguang L, Tieying H, Hongqiu M, Yun Y, Anhua W, Yunxi L, Jianguo W, Huai Y, Xiaoli L, Yawei X, Weihong Z, Yinghong W, Lili D, Weiping L, Ling L, Ying L, Meilian C. Impact of healthcare-associated infections on length of stay: a study in 68 hospitals in China. BioMed Res Int. 2019;2019(2590563):7. doi: 10.1155/2019/2590563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Roberson E. Medicinal plants at risk. Nat Pharm. 2008;1(1):1–16. [Google Scholar]
- 6.Mahesh B, Satish S. Antimicrobial activity of some important medicinal plants against plant and human pathogens. World J Agric Sci. 2008;4:839–843. [Google Scholar]
- 7.Kirchhelle C. Pharming animals: a global history of antibiotics in food production. Palgrave Commun. 2018 doi: 10.1057/s41599-018-0152-2. [DOI] [Google Scholar]
- 8.Sengupta S, Chattopadhyay MK, Grossart H. The multifaceted roles of antibiotics and antibiotic resistance in nature. Front Microbiol. 2013;4:47–47. doi: 10.3389/fmicb.2013.00047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ventola CL. The antibiotic resistance crisis part 1: causes and threats. Pharm Ther. 2015;40(4):277–283. [PMC free article] [PubMed] [Google Scholar]
- 10.Havenga B, Ndlovu T, Clements T, Reyneke B, Waso M, Khan W. Exploring the antimicrobial resistance profiles of WHO critical priority list bacterial strains. BMC Microbiol. 2019;19:303. doi: 10.1186/s12866-019-1687-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Manyi-Loh C, Mamphweli S, Meyer E, Okoh A. Antibiotic use in agriculture and its consequential resistance in environmental sources: potential public health implications. Molecules. 2018;23(4):795. doi: 10.3390/molecules23040795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Akova M. Epidemiology of antimicrobial resistance in bloodstream infections. Virulence. 2016;7(3):252–266. doi: 10.1080/21505594.2016.1159366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ayukekbong JA, Ntemgwa M, Atabe AN. The threat of antimicrobial resistance in developing countries: causes and control strategies. Antimicrob Resist Infect Control. 2017;6(47):1–8. doi: 10.1186/s13756-017-0208-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chandra H, Bishnoi P, Yadav A, Patni B, Mishra A, Nautiyal A. Antimicrobial resistance and the alternative resources with special emphasis on plant-based antimicrobials—a review. Plants. 2017;6(4):16. doi: 10.3390/plants6020016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Abdelbary MMH, Basset P, Blanc DS, Feil EJ. The evolution and dynamics of methicillin-resistant Staphylococcus aureus. In: Tibayrenc M, editor. Genetics and evolution of infectious diseases. 2. Elsevier; 2017. pp. 553–572. [Google Scholar]
- 16.Aghamali M, Sedighi M, Bialvaei AZ, Mohammadzadeh N, Abbasian S, Ghafouri Z, Kouhsari E. Fosfomycin: mechanisms and the increasing prevalence of resistance. J Med Microbiol. 2019;68(1):11–25. doi: 10.1099/jmm.0.000874. [DOI] [PubMed] [Google Scholar]
- 17.Bowling JE, Owens AE, McElmeel ML, Fulcher LC, Herrera ML, Wickes BL, Jorgensen JH. Detection of inducible clindamycin resistance in beta-hemolytic streptococci by using the CLSI broth microdilution test and erythromycin- clindamycin combinations. J Clin Microbiol. 2010;48(6):2275–2277. doi: 10.1128/jcm.00663-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Center for Disease Control & Prevention. (2020). Antibiotic /antimicrobial resistance (AR/AMR). www.cdc.gov/drugresistance/biggest-threats.html
- 19.Damrosch DS. Chemoprophylaxis and sulfonamide resistant Streptococci. J Am Med Assoc. 1946;130(3):214–128. doi: 10.1001/jama.1946.02870030004002. [DOI] [PubMed] [Google Scholar]
- 20.Donabedian SM, Thal LA, Hershberger E, Perri MB, Chow JW, Bartlett P, Zervos MJ. Molecular characterization of gentamicin-resistant Enterococci in the United States: evidence of spread from animals to humans through food. J Clin Microbiol. 2003;41(3):1109–1113. doi: 10.1128/jcm.41.3.1109-1113.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gniadkowski M. Evolution and epidemiology of extended-spectrum β-lactamases (ESBLs) and ESBL-producing microorganisms. Clin Microbiol Infect. 2001;7:597–608. doi: 10.1046/j.1198-743x.2001.00330.x. [DOI] [PubMed] [Google Scholar]
- 22.Goldstein BP. Resistance to rifampicin: a review. J Antibiot. 2014;67(9):625–630. doi: 10.1038/ja.2014.107. [DOI] [PubMed] [Google Scholar]
- 23.Hayden MK, Rezai K, Hayes RA, Lolans K, Quinn JP, Weinstein RA. Development of daptomycin resistance in vivo in methicillin-resistant Staphylococcus aureus. J Clin Microbiol. 2005;43(10):5285–5287. doi: 10.1128/jcm.43.10.5285-5287.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hedges RW, Datta N, Fleming MP. R factors conferring resistance to trimethoprim but not sulphonamides. J Gen Microbiol. 1972;73(3):573–575. doi: 10.1099/00221287-73-3-573. [DOI] [PubMed] [Google Scholar]
- 25.Humphries RM, Yang S, Hemarajata P, Ward KW, Hindler JA, Miller SA, Gregson A. First report of ceftazidime-avibactam resistance in a KPC-3-expressing Klebsiella pneumoniae isolate. Antimicrob Agents Chemother. 2015;59(10):6605–6607. doi: 10.1128/aac.01165-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kalach N, Benhamou PH, Campeotto F, Bergeret M, Dupont C, Raymond J. Clarithromycin resistance and eradication of Helicobacter pylori in children. Antimicrob Agents Chemother. 2001;45(7):2134–2135. doi: 10.1128/aac.45.7.2134-2135.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.MacVane SH, Pandey R, Steed LL, Kreiswirth BN, Chen L. Emergence of ceftolozane-tazobactam-resistant Pseudomonas aeruginosa during treatment is mediated by a single AmpC structural mutation. Antimicrob Agents Chemother. 2017;61(12):2015–2018. doi: 10.1128/aac.01183-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Okamoto S, Tamaru A, Nakajima C, Nishimura K, Tanaka Y, Tokuyama S, Suzuki Y, Ochi K. Loss of a conserved 7-methylguanosine modification in 16S rRNA confers low-level streptomycin resistance in bacteria. Mol Microbiol. 2007;63(4):1096–1106. doi: 10.1111/j.1365-2958.2006.05585.x. [DOI] [PubMed] [Google Scholar]
- 29.Rotimi VO, Khoursheed M, Brazier JS, Jamal WY, Khodakhast FB. Bacteroides species highly resistant to metronidazole: an emerging clinical problem? Clin Microbiol Infect. 1999;5(3):166–169. doi: 10.1111/j.1469-0691.1999.tb00531.x. [DOI] [PubMed] [Google Scholar]
- 30.Sanders CC, Watanakunakorn C. Emergence of resistance to β-lactams, aminoglycosides, and quinolones during combination therapy for infection due to Serratia marcescens. J Infect Dis. 1986;153(3):617–619. doi: 10.1093/infdis/153.3.617. [DOI] [PubMed] [Google Scholar]
- 31.Tristram S, Jacobs MR, Appelbaum PC. Antimicrobial resistance in Haemophilus influenzae. Clin Microbiol Rev. 2007;20(2):368–389. doi: 10.1128/cmr.00040-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vilcheze C, Jacobs WR. Resistance to isoniazid and ethionamide in Mycobacterium tuberculosis: genes, mutations, and causalities. Microbiol Spectr. 2014 doi: 10.1128/microbiolspec.MGM2-0014-2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yigit H, Queenan AM, Anderson GJ, Domenech-Sanchez A, Biddle JW, Steward CD. Novel carbapenem-hydrolyzing β-Lactamase, KPC-1, from a carbapenem-resistant strain. Society. 2001;45(4):1151–1161. doi: 10.1128/aac.45.4.1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Reygaert WC. An overview of the antimicrobial resistance mechanisms of bacteria. AIMS Microbiol. 2018;4(3):482–501. doi: 10.3934/microbiol.2018.3.482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mintah SO, Asafo-Agyei T, Archer M-A, Junior PA-A, Boamah D, Kumadoh D, Agyare C. Medicinal plants for treatment of prevalent diseases. Intechopen. 2019 doi: 10.5772/intechopen.82049. [DOI] [Google Scholar]
- 36.Mulani MS, Kamble EE, Kumkar SN, Tawre MS, Pardesi KR. Emerging strategies to combat ESKAPE pathogens in the era of antimicrobial resistance: a review. Front Microbiol. 2019;10:539. doi: 10.3389/fmicb.2019.00539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bhatia P, Sharma A, George AJ, Anvitha D, Kumar P, Dwivedi VP, Chandra NS. Antibacterial activity of medicinal plants against ESKAPE: an update. Heliyon. 2021;7(2):e06310. doi: 10.1016/j.heliyon.2021.e06310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Khan MF, Tang H, Lyles JT, Pineau R, Mashwani ZU, Quave CL. Antibacterial properties of medicinal plants from Pakistan against multidrug-resistant ESKAPE pathogens. Front Pharmacol. 2018;2018(9):815. doi: 10.3389/fphar.2018.00815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ghuman S. Antimicrobial activity, phenolic content, and cytotoxicity of medicinal plant extracts used for treating dermatological diseases and wound healing in KwaZulu-Natal, South Africa. Front Pharmacol. 2016;2016(7):320. doi: 10.3389/fphar.2016.00320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Harvey AL, Edrada-Ebel R, Quinn RJ. The re-emergence of natural products for drug discovery in the genomics era. Nat Rev Drug Discov. 2015;14(2):111–129. doi: 10.1038/nrd4510. [DOI] [PubMed] [Google Scholar]
- 41.Khan R, Islam B, Akram M, Shakil S, Ahmad A, Ali SM, Siddiqui M, Khan AU. Antimicrobial activity of five herbal extracts against multi drug resistant (MDR) strains of bacteria and fungus of clinical origin. Molecules. 2009;14(2):586–597. doi: 10.3390/molecules14020586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Masoumian M, Zandi M. Antimicrobial activity of some medicinal plant extracts against multidrug resistant bacteria. Zahedan J Res Med Sci. 2017 doi: 10.5812/zjrms.10080. [DOI] [Google Scholar]
- 43.Richwagen N, Lyles JT, Dale BLF, Quave CL. Fedtschenkoi against ESKAPE pathogens. Front Pharmacol. 2019;10:67. doi: 10.3389/fphar.2019.00067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Khan MF, Tang H, Lyles JT, Pineau R, Mashwani ZU, Quave CL. Antibacterial properties of medicinal plants from Pakistan against multidrug-resistant ESKAPE pathogens. Front Pharmacol. 2018;9:815. doi: 10.3389/fphar.2018.00815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chandrasekharan D, Ballal R, Ballal BB, Khetmalas MB. Evaluation of selected medicinal plants for their potential antimicrobial activities against ESKAPE pathogens and the study of P-glycoprotein related antibiosis; an indirect approach to assess efflux mechanism. Int J Rec Sci Res. 2018;9(11A):29461–29466. [Google Scholar]
- 46.Nayim P, Mbaveng AT, Wamba BEN, Fankam AG, Dzotam JK, Kuete V. Antibacterial and antibiotic-potentiating activities of thirteen Cameroonian edible plants against gram-negative resistant phenotypes. Sci World J. 2018;2018:4020294. doi: 10.1155/2018/4020294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mishra KK, Kaur CD, Sahu AK, Panik R, Kashyap P, Mishra SP, Dutta S (2020) Medicinal plants having antifungal properties. In (Ed.), Medicinal plants - use in prevention and treatment of diseases. IntechOpen. 10.5772/intechopen.90674
- 48.Dereje N, Gail D, Beyene TT, Malcolm B, Adefris LB, Abebaw F, Eyasu M. Antibacterial and antifungal activities of Ethiopian medicinal plants: a systematic review. Front Pharmacol. 2021;12:1663–9812. doi: 10.3389/fphar.2021.633921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ferreira MRA, Santiago RR, Langassner SMZ, de Mello JCP, Svidzinski TIE, Soares LAL. Antifungal activity of medicinal plants from northeastern Brazil. J Med Plant Res. 2013;7(40):3008–3013. doi: 10.5897/JMPR2013.5035. [DOI] [Google Scholar]
- 50.Koroishi AM, Foss SR, Cortez DAG, Nakamura TU, Nakamura CV, Filho BPD. In vitro antifungal activity of extracts and neolignans from Piper regnellii against dermatophytes. J Ethnopharmacol. 2008;117:270–277. doi: 10.1016/j.jep.2008.01.039. [DOI] [PubMed] [Google Scholar]
- 51.Manojlovic NT, Solujic S, Sukdolak S, Milosev M. Antifungal activity of Rubia tinctorum, Rhamnus frangula and Caloplaca cerina. Fitoterapia. 2005;76:244–246. doi: 10.1016/j.fitote.2004.12.002. [DOI] [PubMed] [Google Scholar]
- 52.Yemele-Bouberte M, Krohn K, Hussain H, Dongo E, Schulz B, Hu Q. Tithoniamarin and tithoniamide: a structurally unique isocoumarin dimer and a new ceramide from Tithonia diversifolia. Nat Prod Res. 2006;20:842–849. doi: 10.1080/14786410500462892. [DOI] [PubMed] [Google Scholar]
- 53.Kim KY, Davidson PM, Chung HJ. Antibacterial activity in extracts of Camellia japonica L. petals and its application to a model food system. J Food Protect. 2001;64:1255–1260. doi: 10.4315/0362-028X-64.8.1255. [DOI] [PubMed] [Google Scholar]
- 54.Kobayashi K, Nishino C, Tomita H, Fukushima M. Antifungal activity of pisiferic acid derivatives against the rice blast fungus. Phytochemistry. 1987;26:3175–3179. doi: 10.1016/S0031-9422(00)82465-6. [DOI] [Google Scholar]
- 55.Chou SC, Huang TJ, Lin EH, Huang CH, Chou CH. Antihepatitis B virus constituents of Solanum Erianthum. Nat Prod Commun. 2012;7:153–156. doi: 10.1177/1934578x1200700205. [DOI] [PubMed] [Google Scholar]
- 56.Qiu LP, Chen KP. Anti-HBV agents derived from botanical origin. Fitoterapia. 2013;84:140–157. doi: 10.1016/j.fitote.2012.11.003. [DOI] [PubMed] [Google Scholar]
- 57.Parvez MK, Arbab AH, Al-Dosari MS, Al-Rehaily AJ. Antiviral natural products against chronic hepatitis B: recent developments. Curr Pharm Des. 2016;22:286–293. doi: 10.2174/1381612822666151112152733. [DOI] [PubMed] [Google Scholar]
- 58.Wu YH. Naturally derived anti-hepatitis B virus agents and their mechanism of action. World J Gastroenterol. 2016;22:188–204. doi: 10.3748/wjg.v22.i1.188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kaur R, Sharma P, Gupta GK, Ntie-Kang F, Kumar D. Structure-activity-relationship and mechanistic insights for anti-HIV natural products. Molecules. 2020;25:2070. doi: 10.3390/molecules25092070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chavan R, Chowdhary A. In vitro inhibitory activity of Justicia Adhatoda extracts against influenza virus infection and hemagglutination. Int J Pharm Sci Rev Res. 2014;25:231–236. doi: 10.1002/14651858. [DOI] [Google Scholar]
- 61.Rouf R, Uddin SJ, Sarker DK, Islam MT, Ali ES, Shilpi JA, et al. Antiviral potential of garlic (Allium Sativum) and its Organosulfur compounds: a systematic update of pre-clinical and clinical data. Trends Food Sci Technol. 2020;104:219–234. doi: 10.1016/j.tifs.2020.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ghosh R, Chakraborty A, Biswas A, Chowdhuri S. Identification of alkaloids from Justicia Adhatoda as Potent SARS CoV-2 main protease inhibitors: an in silico perspective. J Mol Struct. 2021;1229:129489. doi: 10.1016/j.molstruc.2020.129489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Samra RM, Soliman AF, Zaki AA, El-Gendy AN, Hassan MA, Zaghloul AM. Chemical composition, antiviral and cytotoxic activities of essential oil from Cyperus rotundus growing in Egypt: evidence from chemometrics analysis. J Essent Oil Bear Plants. 2020;23:648–659. doi: 10.1080/0972060x.2020.1823892. [DOI] [Google Scholar]
- 64.Amparo TR, Seibert JB, Silveira BM, Costa FSF, Almeida TC, Braga SFP, et al. Brazilian essential oils as source for the discovery of new anti-COVID-19 drug: a review guided by in silico study. Phytochem Rev. 2021 doi: 10.1007/s11101-021-09754-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Behbahani M, Mohabatkar H, Soltani M. Anti-HIV-1 activities of aerial parts of Ocimum basilicum and its Parasite Cuscuta campestris. J Antivir Antiretrovir. 2013;5:57–61. doi: 10.4172/jaa.1000064. [DOI] [Google Scholar]
- 66.Kubiça TF, Alves SH, Weiblen R, Lovato LT. In vitro inhibition of the bovine viral diarrhoea virus by the essential oil of Ocimum Basilicum (Basil) and monoterpenes. Braz J Microbiol. 2014;45:209–214. doi: 10.1590/S1517-83822014005000030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tshilanda DD, Ngoyi EM, Kabengele CN, Matondo A, Bongo GN, Inkoto CL, et al. Ocimum Species as potential bioresources against COVID-19: a review of their phytochemistry and antiviral activity. Ijpr. 2020 doi: 10.9734/ijpr/2020/v5i430143. [DOI] [Google Scholar]
- 68.Bachar SC, Kishor M, Ritesh B, Asma A, Mamun AM. A review of medicinal plants with antiviral activity available in bangladesh and mechanistic insight into their bioactive metabolites on SARS-CoV-2, HIV and HBV. Front Pharmacol. 2021;12:1663–9812. doi: 10.3389/fphar.2021.732891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Savoia D. Plant-derived antimicrobial compounds: alternatives to antibiotics. Future Microbiol. 2012;7(8):979–990. doi: 10.2217/fmb.12.68. [DOI] [PubMed] [Google Scholar]
- 70.Srivastava J, Chandra H, Nautiyal AR, Kalra SJS. Antimicrobial resistance and plant-derived antimicrobials as an alternative drug line to control infections. Biotech. 2013;4(5):451–460. doi: 10.1007/s13205-013-0180-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Rempe CS, Burris KP, Lenaghan SC, Stewart CNS., Jr The Potential of systems biology to discover antibacterial mechanisms of plant phenolics. Front Microbiol. 2017;8:422. doi: 10.3389/fmicb.2017.00422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Khameneh B, Iranshahy M, Soheili V, Fazly-Bazzaz BS. Review on plant antimicrobials: a mechanistic viewpoint. Antimicrob Resist Infect Control. 2019 doi: 10.1186/s13756-019-0559-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Bouarab-chibane L, Forquet V, Lantéri P, Clément Y. Antibacterial properties of polyphenols: characterization and qsar (quantitative structure – activity relationship) models. Front Microbiol. 2019;10:829. doi: 10.3389/fmicb.2019.00829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Grennan AK. Plant response to bacterial pathogens. Overlap between innate and gene-for-gene defense response. Plant Physiol. 2006;142(3):809–811. doi: 10.1104/pp.106.900207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Li L, Yu Y, Zhou Z, Zhou JM. Plant pattern-recognition receptors controlling innate immunity. Sci China Life Sci. 2016;59(9):878–888. doi: 10.1007/s11427-016-0115-2. [DOI] [PubMed] [Google Scholar]
- 76.Bernouxa M, Jeffrey EG, Dodds PN. New insights in plant immunity signaling activation. Curr Opin Plant Biol. 2012;59(8):3485–3498. doi: 10.1021/jf104517j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ezzat A, Szabó Z, Nyéki J. Induce the plant resistance to pathogen infection. Int J Hortic Sci. 2014 doi: 10.31421/ijhs/20/1-2/1123. [DOI] [Google Scholar]
- 78.González-lamothe R, Mitchell G, Gattuso M, Diarra MS. Plant antimicrobial agents and their effects on plant and human pathogens. Int J Mol Sci. 2009;10(8):3400–3419. doi: 10.3390/ijms10083400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lahlou M. The success of natural products in drug discovery. Pharmacol Pharm. 2013;2013(4):17–31. doi: 10.4236/pp.2013.43A003. [DOI] [Google Scholar]
- 80.Barbieri RCE, Marchese A, Daglia M, Sobarzo-Sanchez E, Nabavi SF, et al. Phytochemicals for human disease: an update on plant-derived compounds antibacterial activity. Microbiol Res. 2017;196:44–68. doi: 10.1016/j.micres.2016.12.003. [DOI] [PubMed] [Google Scholar]
- 81.Zhang Xiujuan SX, Wu Jiaxin Wu, Yue WY, Xiaoqing Hu, Xiaoyuan W. Berberine damages the cell surface of methicillin-resistant Staphylococcus aureus. Front Microbiol. 2020;11:621. doi: 10.3389/fmicb.2020.00621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Zuo G-Y, Li Y, Han J, Wang G-C, Zhang Y-L, Bian Z-Q. Antibacterial and synergy of berberines with antibacterial agents against clinical multi-drug resistant isolates of methicillin-resistant Staphylococcus aureus (MRSA) Molecules. 2012;17(9):10322–10330. doi: 10.3390/molecules170910322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Chu M, Zhang Mb, Liu Yc, et al. Role of berberine in the treatment of methicillin-resistant Staphylococcus aureus Infections. Sci Rep. 2016;6:24748. doi: 10.1038/srep24748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hikal DM. Antibacterial activity of piperine and black pepper oil. Biosci Biotech Res Asia. 2018;15(4):877–880. doi: 10.13005/bbra/2697. [DOI] [Google Scholar]
- 85.Khan IA, Mirza ZM, Kumar A, Verma V, Qazi GN. Piperine, a phytochemical potentiator of ciprofloxacin against Staphylococcus aureus. Antimicrob Agents Chemother. 2006;50:810–812. doi: 10.1128/AAC.50.2.810-812.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Müller AEJ, Albrecht F, Prochnow P, Kuhlmann K, Bandow JE, Slusarenko AJ, Leichert LI. Allicin induces thiol stress in bacteria through s-allylmercapto modification of protein cysteines. J Biol Chem. 2016;291:11477–11490. doi: 10.1074/jbc.M115.702308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Nakamoto M, Kunimura K, Suzuki JI, Kodera Y. Antimicrobial properties of hydrophobic compounds in garlic: Allicin, vinyldithiin, ajoene and diallyl polysulfides. Exp Ther Med. 2020;19:1550–1553. doi: 10.3892/etm.2019.8388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Wallock-Richards DDC, Doherty L, Clarke DJ, Place M, Govan JR, Campopiano DJ. Garlic revisited: antimicrobial activity of allicin-containing garlic extracts against Burkholderia cepacia complex. PLoS ONE. 2014;1:e112726. doi: 10.1371/journal.pone.0112726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Bhatwalkar SB, Mondal R, Krishna S, Adam JK, Govender P, Anupam R. Antibacterial properties of organosulfur compounds of garlic (Allium sativum) Front Microbiol. 2021 doi: 10.3389/fmicb.2021.613077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Han CYKS, Kim YW, Noh K, Lee DY, Kang B, Ryu JH, Jeon R, Kim EH, Hwang SJ, Kim SG. Ajoene, a stable garlic by-product, inhibits high fat diet-induced hepatic steatosis and oxidative injury through LKB1-dependent AMPK activation. Antioxid Redox Signal. 2011;15:187–202. doi: 10.1089/ars.2010.3190. [DOI] [PubMed] [Google Scholar]
- 91.Marchese A, Barbieri R, Coppo E, Orhan IE, Daglia M, Nabavi SF, Izadi M, Abdollahi M, Nabavi SM, Ajami M. Antimicrobial activity of eugenol and essential oils containing eugenol: a mechanistic viewpoint. Crit Rev Microbiol. 2017;43:668–689. doi: 10.1080/1040841x.2017.1295225. [DOI] [PubMed] [Google Scholar]
- 92.Yadav MK, Chae S-W, Im GJ, Chung J-W, Song J-J. Eugenol: a phyto-compound effective against methicillin-resistant and methicillin-sensitive Staphylococcus aureus clinical strain biofilms. PLoS ONE. 2015;10(3):e0119564. doi: 10.1371/journal.pone.0119564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kit-Kay Mak MBK, Ayuba SB, Sakirolla R, Kang Y-B, Mohandas K, Balijepalli MK, Ahmad SH, Pichika MR. A comprehensive review on Eugenol's antimicrobial properties and industry applications: a transformation from ethnomedicine to industry. Pharmacogn Rev. 2019;19:1–9. doi: 10.4103/phrev.phrev_46_18. [DOI] [Google Scholar]
- 94.Ma DSL, Tan LT-H, Chan K-G, Yap WH, Pusparajah P, Chuah L-H, Ming LC, Khan TM, Lee L-H, Goh B-H. Resveratrol—potential antibacterial agent against foodborne pathogens. Front Pharmacol. 2018 doi: 10.3389/fphar.2018.00102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Vestergaard MIH. Antibacterial and antifungal properties of resveratrol. Antimicrob Agents. 2019;53:716–723. doi: 10.1016/j.ijantimicag.2019.02.015. [DOI] [PubMed] [Google Scholar]
- 96.Assob JC, Kamga HL, Nsagha DS, Njunda AL, Nde PF, Asongalem EA, Njouendou AJ, Sandjon B, Penlap VB. Antimicrobial and toxicological activities of five medicinal plant species from Cameroon traditional medicine. BMC Complement Altern Med. 2011;11:70. doi: 10.1186/1472-6882-11-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Agatonovic-Kustrin S, David WM. Hyphenated TLC as a tool in the effect-directed discovery of bioactive natural products. Appl Sci. 2020;10(3):1123. doi: 10.3390/app10031123. [DOI] [Google Scholar]
- 98.Zhang Y, Cai P, Cheng G, Zhang Y. A brief review of phenolic compounds identified from plants: their extraction, analysis, and biological activity. Nat Prod Commun. 2022 doi: 10.1177/1934578X211069721. [DOI] [Google Scholar]
- 99.Matam P, Dariusz K, Piotr K, Dibyendu M, Gregory F. Uncovering the phytochemical basis and the mechanism of plant extract-mediated eco-friendly synthesis of silver nanoparticles using ultra-performance liquid chromatography coupled with a photodiode array and high-resolution mass spectrometry. ACS Sustain Chem Eng. 2022;10(1):562–571. doi: 10.1021/acssuschemeng.1c06960. [DOI] [Google Scholar]
- 100.Atef NM, Shanab SM, Negm SI, et al. Evaluation of antimicrobial activity of some plant extracts against antibiotic susceptible and resistant bacterial strains causing wound infection. Bull Natl Res Cent. 2019;43:144. doi: 10.1186/s42269-019-0184-9. [DOI] [Google Scholar]
- 101.Tsai C-C, Lin C-S, Hsu C-R, Chang C-M, Chang IW, Lin L-W, Hung C-H, Wang J-L. Using the Chinese herb Scutellaria barbata against extensively drug-resistant Acinetobacter baumannii infections: in vitro and in vivo studies. BMC Complement Altern Med. 2018;18(1):96. doi: 10.1186/s12906-018-2151-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Tchana MES, Fankam AG, Mbaveng AT, Nkwengoua ET, Seukep JA, Tchouani K, Nyassé B, Kuete V, Benth P, Pierre O, Americana P, Lauraceae M. Activities of selected medicinal plants against multi-drug resistant Gram-negative bacteria in Cameroon. Afr Health Sci. 2014;14(1):167–172. doi: 10.4314/ahs.v14i1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Khan UA, Rahman H, Qasim M, et al. Alkanna tinctoria leaves extracts: a prospective remedy against multidrug resistant human pathogenic bacteria. BMC Complement Altern Med. 2015;15:127. doi: 10.1186/s12906-015-0646-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Khan MF, Tang H, Lyles JT, Pineau R, Mashwani ZU, Quave CL. Antibacterial properties of medicinal plants from pakistan against multidrug-resistant ESKAPE. Front Pharmacol. 2018;9:1–17. doi: 10.3389/fphar.2018.00815. [DOI] [PMC free article] [PubMed] [Google Scholar]