Abstract
Atherosclerosis is a major risk factor for myocardial infarction and ischemic stroke, which are the leading cause of death worldwide. All-trans retinoic acid (ATRA) is a natural derivative of essential vitamin A. Numerous studies have shown that ATRA plays an important role in cell proliferation, cell apoptosis, cell differentiation, and embryonic development. All-trans retinoic acid (ATRA) is a ligand of retinoic acid receptors that regulates various biological processes by activating retinoic acid signals. In this paper, the metabolic processes of ATRA were reviewed, with emphasis on the effects of ATRA on inflammatory cells involved in the process of atherosclerosis.
Keywords: ATRA, atherosclerosis, adipocytes, RARs, immunity
1. Introduction
Atherosclerosis is a progressive disease of large arteries and a leading cause of stroke and cardiovascular disease [1]. All in all, atherosclerosis can be considered to be a chronic inflammation caused by the interaction between monocyte-derived macrophages, T cells, and other immune response cells [2]. When hypertension, hypercholesterolemia, and other persistent effects or blood flow shear forces from vascular bifurcation diseases cause endothelial cell dysfunction or anatomical damage, vascular smooth muscle cells and fibroblast migrate and proliferate to the intimal layer wall [2]. This inflammatory process can eventually result in complex lesions or the development of plaques that can narrow blood vessels and interfere with normal blood flow to the heart and brain, leading to clinical symptoms [3]. According to statistics, the number of deaths caused by cardiovascular and cerebrovascular diseases accounts for nearly one-third of all the deaths in the world, and this number will increase year by year [4]. Although the common risk factors for atherosclerosis such as hyperlipidemia, hypertension, hyperglycemia, and smoking have been strictly controlled, the incidence of atherosclerotic diseases remains high. Thus, developing new strategies for prevention and treatment requires understanding other factors contributing to atherogenesis.
Vitamin A participates in a variety of physiological processes, such as embryonic development, energy metabolism, immune regulation, and other functions [5]. ATRA plays an important role in the cell growth, development, and differentiation of vertebrates [6]. ATRA has been implicated in several diseases, including inflammatory disorders [7] and cancer [8]. Currently, ATRA is the routine treatment for the management of acute promyelocytic leukemia (APL) [9]. Studies have reported that ATRA can significantly inhibit the formation of atherosclerotic lesions in rabbit models of atherosclerosis induced by a high-fat diet [10,11]. In addition, it was shown that ATRA can inhibit restenosis after balloon angioplasty in atherosclerotic rabbits [12]. A recent study showed that ATRA relieves coronary artery stenosis by regulating the function of smooth muscle cells in a mouse model of Kawasaki disease [13].
This article reviews the effects of ATRA and retinoic acid (RA) on the development of atherosclerotic cardiovascular disease (ASCVD). First, we focused on ATRA molecular signal transduction, and then we discussed the effects of ATRA on lipid metabolism and various inflammatory cells. The main focus of this review is the effect of ATRA on various types of inflammatory cells, and then it will discuss a summary of the current literature related to the physiology of ATRA and macrophages, T cells, and smooth muscle cells.
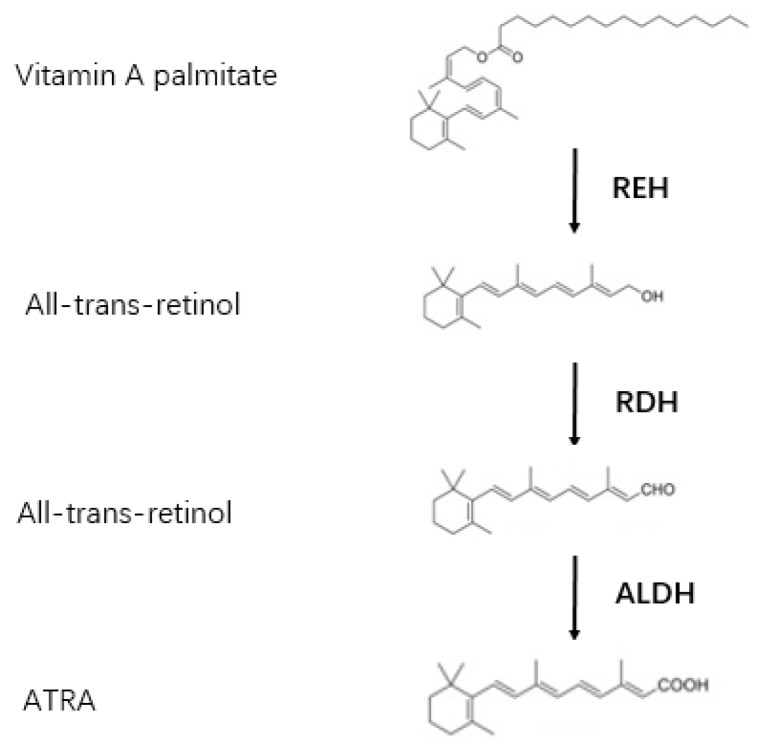
2. Metabolism of ATRA
Vitamin A achieves its effect on growth and development through the action of the biologically active metabolite RA, mainly including ATRA and 9-cis-retinoic acid (9-cis-retinoic acid) [14,15]. However, all-trans-RA is the primary ligand during development [16]. Mammals cannot synthesize vitamin A, they can only get vitamin A from food. Vitamin A mainly exists in the form of retinol ester, which is hydrolyzed to retinol by retinol ester hydrolase (REH). Retinol binds to the retinol binding protein (RBP4) in the plasma and enters the cell, where it is oxidized by retinol dehydrogenase (RDH) to all-trans retinal, and then by retinol dehydrogenase (ALDH) converted to ATRA (Figure 1). Cytochrome P450 superfamily enzymes (CYP26A1, CYP26B1, and CYP26C1) strictly regulate the intracellular concentration of ATRA and promote the conversion of intracellular ATRA into inactive metabolites [17]. The effects of ATRA were relayed by binding to nuclear retinoid receptors [18]. Two types of nuclear retinoic acid receptors (RARs) and retinoic X receptors (RXRs) have been discovered [19,20]. ATRA only binds to retinoic acid receptors (RARs) with high affinity, 9-cis retinoic acid is an isomer of ATRA which can be combined with RARs and retinoic X receptors (RXRs) [21]. 13-cis retinoic acid, another isomer of ATRA, is a high-affinity ligand that only binds to RXRs [22]. Among RA isomers, ATRA can bind to and activate RARs both in vivo and in vitro. 9-cis-RA can activate RXRs and RARs in vitro, but it does not work in the body [23]. 13-cis-RA makes up about 25% of circulating retinoic acid levels [24]. 9-cis RA has been detected at extremely low levels. Therefore, most of the in vivo activation of all RAR subtypes (α, β and γ) is considered to be ATRA-mediated [23].
Figure 1.
The process of converting of Vitamin A to ATRA.
RARs are members of the steroid/vitamin D/thyroid hormone receptor family, the pleiotropic effect of ATRA is mediated through its combination with RARs [18], and regulates many biological functions, such as embryonic development, organogenesis, homeostasis, vision, immune function, and reproduction [25]. Like most nuclear receptors (NRs), RARs have the function of the dimer. RARs cooperate with the retinoid receptor (RXR) to form heterodimers, which are the main regulators of human gene expression and an important drug target [26]. RARs and RXRs are modular proteins composed of several domains, the most notable being a DNA binding domain (DBD) and a C-terminal ligand-binding domain (LBD) [27]. The LBD contains the ligand-dependent activation function, AF-2 [27]. Before binding to the ligand, the RXR and RARα bind to specific regions of the retinoic acid response element (RARE) DNA as heterodimers. The RXR and RARα recruit silencing mediators of retinoic acid and thyroid hormone receptor (SMRT), nuclear receptor corepressor (NCoR), a corepressor related to histone deacetylase (HDAC), leading to transcriptional inhibition of target genes [28]. After binding to ATRA, the conformation of the RARα changes, triggering the release of co-inhibitors and the recruitment of co-activators (steroid receptor co-activator, SRC-1, -2, and -3) and the recruitment of histone acetyltransferases (HATs), leading to transcriptional activation of target genes [29] (Figure 2). ATRA binds to retinoid receptors and their heterodimers, which can affect cellular processes not only through genomic pathways but also through non-genomic mechanisms [30].
Figure 2.
The genomic molecular pathway by ATRA. In the absence of ligands, retinoic acid X receptors and RXR/RAR heterodimers bind to RAREs in the regulatory regions of target genes and inhibit transcription by recruiting HDAC or silencing mediators (SMRT and NCoR). After binding to ATRA, the corepressor complex is dissociated, and the coactivator binds to histone acetyltransferase (HAT) activity, resulting in transcriptional activation of target genes.
3. ATRA and Adipocytes
Adipose tissue accounts for 15 to 20 percent of the body’s total steroid reserves [31] and is a potential target for the effects of ATRA [32]. The differentiation of adipose precursor cells into adipose cells in white (WAT) or brown adipose tissue (BAT) is related to the accumulation of intracellular retinoids [33]. In addition to storing retinoids, adipocytes also synthesize and secrete RBP [34]. Vitamin A is a nutrient that has significant effects on adipose tissue biology and energy homeostasis [35]. Dietary vitamin A and provitamin A are stored in the form of retinol, or metabolized in cells to retinoic acid, the main active form of vitamin A [36]. All-trans retinoic acid treatment can reduce fat in mice and improve insulin sensitivity by promoting fat mobilization and catabolism [37]. In addition, ATRA can effectively inhibit the differentiation of clonal preadipocyte lines in vitro [38]. ATRA reduces the ability of adipogenesis, directly increases lipolysis, and reduces the triacylglycerol content in mature adipocytes [39]. Tourniaire et al. [40] showed that ATRA affects mitochondria in adipocytes, resulting in increased oxidative phosphorylation (OXPHOS) capacity and mitochondrial content in these cells. Exogenous ATRA acts mainly by reducing the proliferation and differentiation of preadipocytes, inducing apoptosis, and promoting adipocyte defatting [41]. Perivascular adipose tissue (PVAT) is composed of a mixture of two types of WAT and brown adipose tissue (BAT) [42]. Apolipoprotein E-deficient mice (Apo-E) treated with ATRA stimulated PVAT browning and increased adiponectin synthesis. Apo-E mice treated with ATRA stimulated PVAT browning and increased adiponectin synthesis while improving atherosclerosis in Apo-E mice [43]. However, further studies are needed to demonstrate the role of ATRA in regulating lipid metabolism in vivo. The molecular mechanism of ATRA’s influence on lipid metabolism is complex and not yet fully understood.
4. Effect of All-Trans Retinoic Acid on Atherosclerosis
The pathological basis of most cardiovascular and cerebrovascular diseases is atherosclerosis, which is the main cause of death worldwide. Atherosclerosis is a chronic disease involving processes such as lipid deposition, endothelial damage, and immune inflammation. Endothelial cells, macrophages, white blood cells, and intimal smooth muscle cells are the main players in the development of this disease [44]. So far, atherosclerosis is still the most common underlying cause of cardiovascular and cerebrovascular diseases. ATRA affects the development of atherosclerosis by regulating the biological processes of immune cells, such as cell proliferation, migration, and phenotypic transformation. ATRA is also involved in the regulation of blood glucose concentration, lipid metabolism, inflammation, and other risk factors for atherosclerosis [45].
4.1. ATRA and Macrophages
In Western society, atherosclerotic cardiovascular disease is the main cause of death. The hallmark of early-stage atherosclerosis is the accumulation of macrophage-derived foam cells [46,47]. Due to the uncontrolled uptake of modified low-density lipoprotein (LDL) by macrophages, excessive lipoprotein-derived cholesterol accumulates in the cells, leading to the formation of macrophage foam cells. The removal of cholesterol deposits in arteries by macrophages is beneficial in the early stages of atherosclerosis [48]. HDL (high-density lipoprotein) particles enhance the outflow of cholesterol from foam cells, which is the first step of reverse cholesterol transport (RCT) [49]. The endogenous production of lipid-poor Apo-E and the ATP-binding cassette transporters A1 (ABCA1) and G1 (ABCG1) are common pathways for cholesterol efflux [50,51]. Ayaori et al. [52] found that ATRA enhanced the expression of ABCA1 and ABCG1 in ApoA-I/HDL-mediated THP-1 macrophages and human monocyte-derived macrophages (HMDM), thereby promoting cholesterol efflux from macrophages. The liver X receptor (liver X receptor, LXR) is a ligand-dependent nuclear receptor, which plays an important role in the cells involved in the transcriptional regulation of cholesterol homeostasis. ABCA1 is one of the most well-known LXR target genes, and LXR activation increases ABCA1 expression, which in turn promotes the outflow of cholesterol and phospholipids to lipid-poor ApoA-I, resulting in the formation of nascent HDL particles. The transactivation of RXRs can enhance the expression of LXR target genes, thereby regulating lipid homeostasis, which is an attractive therapeutic target in macrophages. The coordinated tissue-specific role of the LXR pathway maintains cholesterol homeostasis throughout the body and regulates immune and inflammatory responses [53]. Therefore, LXR agonists have attracted widespread attention in the treatment of atherosclerosis. Unfortunately, due to its serious adverse reactions, this class of drugs has not yet been approved by the FDA. Fortunately, the use of nanoparticles (NPs) to target the delivery of LXR agonists can facilitate effective LXR-mediated therapy [54].
Macrophages are plastic cells with proinflammatory or anti-inflammatory properties. In response to microenvironmental signals, two classical modes of macrophages are activated, proinflammatory M1 macrophages or anti-inflammatory M2 macrophages [55]. During the progression of atherosclerosis, M1 macrophages have been shown to aggravate plaque and systemic inflammation, leading to plaque rupture, which dominates the progression of atherosclerosis. M2-like macrophages are abundant in stable plaques [56]. ATRA has been found to promote M1 and M2 phenotypic transformation in tumors [57], parasitic infections [58], and other diseases, but no studies have shown that ATRA can promote the phenotypic transformation of macrophages in atherosclerosis. However, recent studies have shown that ATRA can enhance the initiating signal of NOD-like receptor protein-3 (NLRP3) inflammasome in human macrophages, thereby improving the inflammatory response [59].
4.2. ATRA and SMC
Smooth muscle cells (SMCs) are the most important cells involved in the development of atherosclerosis, and they tend to occur in the thickened intimal layer of arteries. With the development of atherosclerotic plaque and intimal SMC phenotype changes, contractility is lost, SMC marker expression decreases, and it participates in the formation of foam cells [60]. The phenotype of SMC is converted to a poorly differentiated form and SMC markers are reduced or completely absent, leading to an underestimation of its role in the development of atherosclerosis in humans and animals. A large number of studies have shown that ATRA is involved in the active process of vascular injury sites, including SMC differentiation and proliferation. It is a potential candidate to prevent atherosclerosis, intimal hyperplasia, and restenosis.
4.2.1. ATRA and SMC Proliferation and Migration
The accumulation of smooth muscle cells (SMCs) in the intima is a characteristic of vascular lesions, including atherosclerotic plaque and restenosis after angioplasty. In the past few decades, there have been conflicting reports about the effects of retinoids on the proliferation of vascular smooth muscle cells. At the earliest, Peclo showed that ATRA has a mitogenic effect on smooth muscle cells [61]. Other studies have not found the effect of retinoic acid on SMC proliferation [62]. However, the vast majority of reports have reported the inhibitory effects of ATRA on the growth of vascular smooth muscle cells (VSMCs) of different species from rats to humans [63]. A number of studies have shown that several mitotic factors can inhibit the proliferation of SMCs, such as PDGF-BB (platelet-derived growth factor-BB) [63], endothelin-1, serum [64], serotonin [65], AngII (angiotensin II) [66], endothelin-1 [67], and bFGF (basic fibroblast growth factor) [68]. Krüppel-like zinc finger transcription factor 5 (KLF5; also known as BTEB2 and IKLF) was significantly induced in activated vascular smooth muscle cells and fibroblasts, whereas the arterial wall thickening of heterozygous KLF knockout (KLF5 +/−) mice was significantly lower than that of wild-type animals [69]. Shindo et al. [69] discovered that KLF5 interacts with the retinoic acid receptor (RAR), and the synthesis of RAR ligands regulates the transcriptional activity of KLF5 to play a protective effect on cardiovascular remodeling. In addition, Sakamoto and colleagues [70] showed that the growth of VSMCs in cultured human atherosclerotic specimens was associated with increased KLF5 expression. These studies clearly demonstrated the inhibitory effect of retinoic acid on the proliferation of SMC. Tran-Lundmark [71] demonstrated that ATRA inhibits the proliferation of SMCs by regulating the expression of perlecan in SMCs, depending on its heparin sulfate chain. In addition to its effect on perlecan, ATRA can also induce the expression of tumor suppressor a-kinase anchoring protein 12 in SMCs and block the growth of vascular SMCs [72], and has been shown to inhibit the expression of fibrin, an extracellular matrix component of human fetal palatal mesenchymal cells [73]. In SMCs, ATRA can downregulate urokinase plasminogen activator (uPA) [74]. uPA can activate a number of proteins, one of which is PDGF-D, an effective activator of SMC proliferation [75]. Recent studies on the response of VSMCs to vascular injury have shown that ATRA can inhibit intimal hyperplasia and smooth muscle cell proliferation and migration by activating the AMPK signaling pathway and inhibiting mTOR signaling [76].
In summary, retinoic acid can inhibit the proliferation and migration of VSMCs, thereby weakening the effect of smooth muscle cells on the progression of atherosclerosis, but the specific molecular mechanism is still unclear. Recently, Yu et al. [77] found that ATRA prevented vein grafts stenosis by inhibiting Rb-E2F-mediated cell cycle progression and KLF5-RARα interaction in human vein SMC. The mechanism of ATRA on smooth muscle cells should be further explored in atherosclerosis models in the future.
4.2.2. ATRA and SMC Differentiation
There are two phenotypes of VSMCs: contractile VSMCs and synthetic VSMCs. This phenotypic differentiation occurs in response to microenvironmental stimuli. The phenotypic transition of SMCs plays a central role in the development, progression, and stabilization of plaques. “Phenotypic switching” is a process of medial SMC dedifferentiating, proliferating, and migrating to intimal lesions in response to atherosclerosis stimulation. Human genomics studies have found that the RA signal is one of the main regulators of this process. Activation of RA signaling by ATRA prevents the phenotypic transition of SMCs, reduces the burden of atherosclerosis, and promotes the stability of the fiber cap [78]. Retinoic acid is a well-known phenotypic differentiation inducer in many cell types, including vascular SMCs [79]. ATRA can enhance the expression of SMC differentiation markers smooth muscle MHC (myosin heavy chain) [80], α-actin [12,80,81,82,83,84], and significantly inhibit the protein expression and activity of matrix metalloproteinases MMP-2 and MMP-9 [80]. Colbert et al. [85] showed that a RARE-lacZ transgene colocalizes with the expression of adult smooth muscle myosin heavy chain subtype SM2. This is the first in vivo evidence supporting the role of activated retinoic acid receptors in SMC differentiation. Many in vitro studies have demonstrated that ATRA can affect the differentiation process of SMCs. In vitro, compared with SMCs in a normal culture medium, the sensitivity of intimal cells to ATRA-induced apoptosis is increased, confirming the main role of phenotype in determining SMC behavior [63,74,86]. Hayashi et al. [64] found that ATRA stimulated the expression of tropoelastin mRNA, which in turn promoted the fine expression of elastin in vascular SMCs of chicken embryos. Haller et al. [83] evaluated the effect of ATRA on the differentiation of SMCs in primary rat aorta by increasing the expression of protein kinase C(PKC)-α and SM α-actin. Fully differentiated SMCs can activate the myofilament device, increase intracellular calcium ions, and produce responsiveness to contractile agonists. Blank et al. [87] showed that the activation of G protein-coupled receptors cloned from ATRA-derived SMCs in P19 embryonal carcinoma cells (P19s) resulted in a significant increase in intracellular calcium. Parental P19s (untreated with ATRA) showed virtually no such increase in intracellular calcium levels. Wright et al. [88] showed that ATRA could restore the contractility of SMCs in aortic rings. In addition, Rogers et al. [89] found that ATRA increased the level of calcification inhibitor MGP (matrix Gla protein) and decreased the activity of TNAP (tissue-nonspecific alkaline phosphatase) through RARα and its coordinated transcriptional regulation, thereby inhibiting calcification of SMCs and aortic VICs.
Through the complex interaction with many factors, ATRA plays a pleiotropic effect on the differentiation, proliferation, and migration of VSMCs, and VSMCs play an increasingly important role in the pathogenesis of atherosclerosis. However, the sometimes contradictory nature found in the field highlights the need for further investigation of the mechanisms involved.
4.3. ATRA and Neutrophils
Neutrophils are the main effector cells of natural immunity, which fight infection through phagocytosis and degranulation. Similar to microglia/macrophages, neutrophils show heterogeneous phenotypes. N2 neutrophils promoted microglial/macrophage phagocytic activity and contributed to post-stroke inflammation reduction, whereas the N1 phenotype intensified neuroinflammation [90]. Activated neutrophils also release neutrophilic extracellular traps (NETs) in response to various stimuli [91]. The development of atherosclerosis, beginning with the formation of fatty streaks and progressing to atherosclerosis and plaque formation, relies on a chronic inflammatory process driven by lipids that involves blood vessels and immune cells [92]. It is known that hyperlipidemia-induced neutrophils have been shown to be positively correlated with human atherosclerosis and atherosclerosis-related diseases in humans [93,94]. Megens et al. [95] detected NETs in human plaques obtained by endarterectomy. Warnatsch et al. [96] found in a mouse model of atherosclerosis that sterile inflammation drives the production of cytokines that subsequently trigger neutrophils to release extracellular traps. A recent study shows that neutrophils are polarized toward beneficial N2 phenotypes with ATRA treatment, whereas the harmful NETs formation was inhibited [97]. In conclusion, ATRA regulates neuroinflammation after stroke and modulates neutrophilic function. Because of its protective effect against ischemic stroke, further clinical studies of ATRA as a promising treatment for patients with ischemic stroke are necessary.
4.4. ATRA and T Cell
A growing body of evidence supports the critical role of T cells as drivers and modulators of atherosclerosis, a chronic inflammatory disease. In atherosclerosis, T cells of different subtypes regulate the progression or regression of atherosclerosis by secreting inflammatory signals. Regulatory T (Treg) cells have an anti-atherosclerotic function, whereas CD4 + T helper 1 (Th1) cells and natural killer T cells have pro-atherosclerotic properties. Each Th cell subtype expresses unique characteristics and performs unique functions in the immune response process.
Th1 cells exert pro-inflammatory effects through binding to M1 macrophages and secreting IFNγ [98]; Th1 cells are the dominant T cell population in human plaques. A large number of studies have shown that Th1 cells have an atherosclerotic effect [99]. Since the Th2 phenotype is opposite to the differentiation of Th1 cells that promote atherosclerosis, Th2 cells seem to have anti-atherosclerotic effects. Cytokines produced by Th2 cells include IL-4, IL-5, and IL-13, all of which are considered to have atherosclerotic protection [100,101]. The influence of RA on the balance of the Th1/Th2 response is controversial. A large number of research studies have shown that ATRA can promote the differentiation of naive T cells into Th2 cells by inducing gene expression of IL-4 [102]. In addition, ATRA regulates the production of IL-12 through APCs, inhibits Th1 cell differentiation [103], and induces the expression of GATA3 (GATA-binding protein 3), an activator of transcription 6 (STAT6), which is important for maintaining Th2 response [102]. However, retinoic acid does not always inhibit Th1-related mechanisms. It is essential for the stability and maintenance of Th1 cells. RA signal transduction is essential for limiting the conversion of Th1 cells to Th17 effectors and preventing Th17 pathogenic reactions in the body [104].
TGFβ and IL-6 or IL-23 mediate differentiation into Th17 cells [105]. Th17 cells release the cytokines IL-17, IL-21, IL-22, and IL-23 [106]. Once Th17 cells infiltrate the plaque, SMCs are induced to secrete pro-inflammatory cytokines and chemokines [107]. The role of Th17 in atherosclerosis is still under debate. Treg cells are negative regulators of the immune system [108] and inhibit atherosclerosis by secreting anti-inflammatory cytokines including IL-10 and IL-35 [109]. The mechanism of RA’s influence on Th17/Treg balance is well known. In the presence of TGF-β [110], small intestinal lamina propria dendritic cells can synthesize RA and produce Treg. Therefore, the increase in TGF-β level promotes naive CD4 T cells to produce Treg through an RA-dependent mechanism [111,112,113,114].
Antigen-presenting cells can recognize pathogens directly. T cell activation requires antigen-presenting cells to recognize the antigen before proliferating and differentiating into effector cells [115]. Macrophages direct the responses of T cells, such as Th1, Th2, Th17, and Treg cells. Macrophages in M1 suppression mode direct T cells to produce Th1-like cytokines (such as IFN-γ), which stimulate specific cytolytic T cells and activate more M1 macrophages. In contrast, M2 macrophages stimulate T cells to produce Th2-like cytokines (such as IL-4 and TGF-β), causing B cell proliferation and antibody production, and further amplifying M2 responses [116]. It is known that most macrophages in progressive plaques in mice and humans resemble the activated classic M1 phenotypic state. Lin et al. found that plaque regression is characterized not only by the reduction in classically activated M1 macrophages but also by the enrichment of cells expressing selective activation (M2 or M[IL-4]) macrophage markers. It has been shown that activated M2 macrophages are involved in inflammation regression and tissue damage repair, which is consistent with the characteristics of plaque regression [117]. Macrophages can complete phenotypic transformation between M1 and M2 types in an ATRA-dependent manner, resulting in increased numbers of Tregs, which are also abundant in regression plaques. There has been no evidence to prove the direct regulation of ATRA on T cells in atherosclerosis, and further exploration is needed.
4.5. ATRA and Endotheliocyte
The formation mechanism of atherosclerosis is complex and diverse. The dysfunction and injury of vascular endothelial cells is one of the important risk factors for the development of atherosclerosis [118,119] and affects the degree of atherosclerotic lesions [120,121]. In the presence of a variety of risk factors for atherosclerosis, atherosclerotic lesions develop from local lesions to systemic complications, such as coronary heart disease and stroke [122]. Endothelial dysfunction is usually used to refer to the abnormal production or bioavailability of endothelial nitric oxide (NO), and the resulting harmful changes in vascular reactivity [123]. Studies have reported that ATRA may change the level of endogenous NO synthase inhibitor asymmetric dimethylarginine (ADMA), thereby increasing NO synthesis [124]. In addition, ATRA can inhibit the expression of endothelin-1 (ET-1) mRNA in endothelin-1 (ET-1) mRNA in endothelial cells (ECs), leading to anti-atherosclerosis effects [125,126]. ATRA may alleviate atherosclerosis by reducing ET-1 expression and caveolin-1 (CAV-1) expression in atherosclerotic rabbits and increasing endothelial nitric oxide synthase (eNOS) phosphorylation [127,128].
4.6. ATRA and Other Pathologic Mechanisms Associated with Atherosclerosis
ATRA can not only regulate the biological processes of immune cells, but also participate in the regulation of blood glucose concentration, oxidative stress, and other risk factors related to atherosclerosis [45,129,130]. Antidiabetic effects of ATRA in pancreatic beta cells have been reported [129,131], and high-dose vitamin A supplementation has been shown in a randomized trial to significantly reduce fasting blood glucose and glycated hemoglobin in patients with type 2 diabetes, but the results of this study failed to show a significant effect on lipid metabolism [132]. Furthermore, Blaner et al. [130] showed that ATRA mediates the antioxidant effects of vitamin A in vivo by affecting the transcription of genes critical for mediating antioxidant responses. They showed that the antioxidant effects of ATRA were dose-related, with low doses of ATRA having antioxidant properties, but high doses of ATRA increasing free radical production, inducing lipid peroxidation, and reducing cell viability.
5. Conclusions
The beneficial effects of ATRA and its isomers on cardiovascular disease outcomes have also been confirmed in many articles [75,120,133,134,135,136]. ATRA is currently used clinically to treat a variety of human malignancies [137,138,139], and its therapeutic dose, side effects, and interaction with other drugs have been clarified, which obviously has advantages in future clinical trials of atherosclerosis treatment. The current dose of ATRA in clinical use is 0.1–1 μM, and high doses of ATRA may bring serious adverse reactions [140]. With the development of nanotechnology, the targeted delivery of ATRA in the treatment of atherosclerosis has greater benefits and fewer adverse reactions [141]. The pathological process of atherosclerosis is complicated. An in-depth understanding of the effects of ATRA on the various stages of atherosclerosis may bring new progress in the treatment of atherosclerosis and new breakthroughs in the treatment of atherosclerotic cardiovascular diseases. Finally, the influence of other isomers of ATRA or synthetic retinoic acid on atherosclerosis cannot be ignored. In the future, more animal models are still needed to study the role of ATRA in atherosclerosis, especially in the immune response to various inflammatory cells. Further exploration is needed in the future.
Author Contributions
Q.D. had the idea and performed the literature search. J.C. were involved in critically revising and editing the manuscript. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare that there is no conflict of interest.
Funding Statement
This research received no external funding.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Libby P. Inflammation in atherosclerosis. Nature. 2002;420:868–874. doi: 10.1038/nature01323. [DOI] [PubMed] [Google Scholar]
- 2.Hansson G.K., Hermansson A. The immune system in atherosclerosis. Nat. Immunol. 2011;12:204–212. doi: 10.1038/ni.2001. [DOI] [PubMed] [Google Scholar]
- 3.Ross R. Atherosclerosis is an inflammatory disease. N. Engl. J. Med. 1999;340:115–126. doi: 10.1056/NEJM199901143400207. [DOI] [PubMed] [Google Scholar]
- 4.Cannon B. Cardiovascular disease: Biochemistry to behaviour. Nature. 2013;493:S2–S3. doi: 10.1038/493S2a. [DOI] [PubMed] [Google Scholar]
- 5.Ghyselinck N.B., Duester G. Retinoic acid signaling pathways. Development. 2019;146:dev167502. doi: 10.1242/dev.167502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rhinn M., Dollé P. Retinoic acid signalling during development. Development. 2012;139:843–858. doi: 10.1242/dev.065938. [DOI] [PubMed] [Google Scholar]
- 7.Bécherel P.-A., Mossalayi M., Goff L., Francès C., Chosidow O., Debré P., Arock M. Mechanism of anti-inflammatory action of retinoids on keratinocytes. Lancet. 1994;344:1570–1571. doi: 10.1016/S0140-6736(94)90377-8. [DOI] [PubMed] [Google Scholar]
- 8.Soprano D.R., Qin P., Soprano K.J. Retinoic Acid Receptors and Cancers. Annu. Rev. Nutr. 2004;24:201–221. doi: 10.1146/annurev.nutr.24.012003.132407. [DOI] [PubMed] [Google Scholar]
- 9.Avvisati G., Tallman M.S. All-trans retinoic acid in acute promyelocytic leukaemia. Best Pract. Res. Clin. Haematol. 2003;16:419–432. doi: 10.1016/S1521-6926(03)00057-4. [DOI] [PubMed] [Google Scholar]
- 10.Zhou B., Pan Y., Hu Z., Wang X., Han J., Zhou Q., Zhai Z., Wang Y. All-trans-Retinoic Acid Ameliorated High Fat Diet-Induced Atherosclerosis in Rabbits by Inhibiting Platelet Activation and Inflammation. J. Biomed. Biotechnol. 2012;2012:259693. doi: 10.1155/2012/259693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zarei L., Bahrami M., Farhad N., Froushani S.M.A., Abbasi A. All-trans retinoic acid effectively reduces atheroma plaque size in a rabbit model of high-fat-induced atherosclerosis. Adv. Clin. Exp. Med. Off. Organ Wroc. Med. Univ. 2018;27:1631–1636. doi: 10.17219/acem/74552. [DOI] [PubMed] [Google Scholar]
- 12.Wiegman P.J., Barry W.L., McPherson J.A., McNamara C.A., Gimple L.W., Sanders J.M., Sarembock I.J. All-trans-retinoic acid limits restenosis after balloon angioplasty in the focally atherosclerotic rabbit: A favorable effect on vessel remodeling. Arterioscler. Thromb. Vasc. Biol. 2000;20:89–95. doi: 10.1161/01.ATV.20.1.89. [DOI] [PubMed] [Google Scholar]
- 13.Suganuma E., Sato S., Honda S., Nakazawa A. All trans retinoic acid alleviates coronary stenosis by regulating smooth muscle cell function in a mouse model of Kawasaki disease. Sci. Rep. 2021;11:13856. doi: 10.1038/s41598-021-93459-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ross S.A., McCaffery P., Dräger U.C., De Luca L.M. Retinoids in Embryonal Development. Physiol. Rev. 2000;80:1021–1054. doi: 10.1152/physrev.2000.80.3.1021. [DOI] [PubMed] [Google Scholar]
- 15.Petkovich P.M. Retinoic acid metabolism. J. Am. Acad. Dermatol. 2001;45:S136–S142. doi: 10.1067/mjd.2001.113715. [DOI] [PubMed] [Google Scholar]
- 16.Cunningham T., Duester G. Mechanisms of retinoic acid signalling and its roles in organ and limb development. Nat. Rev. Mol. Cell Biol. 2015;16:110–123. doi: 10.1038/nrm3932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fujii H., Sato T., Kaneko S., Gotoh O., Fujii-Kuriyama Y., Osawa K., Kato S., Hamada H. Metabolic inactivation of retinoic acid by a novel P450 differentially expressed in developing mouse embryos. EMBO J. 1997;16:4163–4173. doi: 10.1093/emboj/16.14.4163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gudas L.J. Retinoids and vertebrate development. J. Biol. Chem. 1994;269:15399–15402. doi: 10.1016/S0021-9258(17)40689-2. [DOI] [PubMed] [Google Scholar]
- 19.Mangelsdorf D.J., Thummel C., Beato M., Herrlich P., Schütz G., Umesono K., Blumberg B., Kastner P., Mark M., Chambon P., et al. The nuclear receptor superfamily: The second decade. Cell. 1995;83:835–839. doi: 10.1016/0092-8674(95)90199-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chambon P. A decade of molecular biology of retinoic acid receptors. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 1996;10:940–954. doi: 10.1096/fasebj.10.9.8801176. [DOI] [PubMed] [Google Scholar]
- 21.Heyman R.A., Mangelsdorf D., Dyck J.A., Stein R.B., Eichele G., Evans R., Thaller C. 9-cis retinoic acid is a high affinity ligand for the retinoid X receptor. Cell. 1992;68:397–406. doi: 10.1016/0092-8674(92)90479-V. [DOI] [PubMed] [Google Scholar]
- 22.Connolly R.M., Nguyen N.K., Sukumar S. Molecular Pathways: Current Role and Future Directions of the Retinoic Acid Pathway in Cancer Prevention and Treatment. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2013;19:1651–1659. doi: 10.1158/1078-0432.CCR-12-3175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ziouzenkova O., Plutzky J. Retinoid metabolism and nuclear receptor responses: New insights into coordinated regulation of the PPAR-RXR complex. FEBS Lett. 2007;582:32–38. doi: 10.1016/j.febslet.2007.11.081. [DOI] [PubMed] [Google Scholar]
- 24.Napoli J.L. [13] Quantification of physiological levels of retinoic acid. Methods Enzymol. 1986;123:112–124. doi: 10.1016/s0076-6879(86)23015-3. [DOI] [PubMed] [Google Scholar]
- 25.Tang X.H., Gudas L.J. Retinoids, retinoic acid receptors, and cancer. Annu. Rev. Pathol. 2011;6:345–364. doi: 10.1146/annurev-pathol-011110-130303. [DOI] [PubMed] [Google Scholar]
- 26.Rochel N., Moras D. Architecture of DNA Bound RAR Heterodimers. Biochem. Retin. Acid Recept. I Struct. Act. Funct. Mol. Level. 2014;70:21–36. doi: 10.1007/978-94-017-9050-5_2. [DOI] [PubMed] [Google Scholar]
- 27.Bastien J., Rochette-Egly C. Nuclear retinoid receptors and the transcription of retinoid-target genes. Gene. 2004;328:1–16. doi: 10.1016/j.gene.2003.12.005. [DOI] [PubMed] [Google Scholar]
- 28.Evans R.M., Mangelsdorf D.J. Nuclear Receptors, RXR, and the Big Bang. Cell. 2014;157:255–266. doi: 10.1016/j.cell.2014.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Glass C.K., Rosenfeld M.G. The coregulator exchange in transcriptional functions of nuclear receptors. Genes Dev. 2000;14:121–141. doi: 10.1101/gad.14.2.121. [DOI] [PubMed] [Google Scholar]
- 30.Landrier J.-F., Marcotorchino J., Tourniaire F. Lipophilic Micronutrients and Adipose Tissue Biology. Nutrients. 2012;4:1622–1649. doi: 10.3390/nu4111622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tsutsumi C., Okuno M., Tannous L., Piantedosi R., Allan M., Goodman D.S., Blaner W.S. Retinoids and retinoid-binding protein expression in rat adipocytes. J. Biol. Chem. 1992;267:1805–1810. doi: 10.1016/S0021-9258(18)46017-6. [DOI] [PubMed] [Google Scholar]
- 32.Theodosiou M., Laudet V., Schubert M. From carrot to clinic: An overview of the retinoic acid signaling pathway. Cell. Mol. Life Sci. CMLS. 2010;67:1423–1445. doi: 10.1007/s00018-010-0268-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Morikawa K., Hanada H., Hirota K., Nonaka M., Ikeda C. All-trans retinoic acid displays multiple effects on the growth, lipogenesis and adipokine gene expression of AML-I preadipocyte cell line. Cell Biol. Int. 2012;37:36–46. doi: 10.1002/cbin.10005. [DOI] [PubMed] [Google Scholar]
- 34.Zovich D., Orologa A., Okuno M., Kong L., Talmage D., Piantedosi R., Goodman D., Blaner W. Differentiation-dependent expression of retinoid-binding proteins in BFC-1 beta adipocytes. J. Biol. Chem. 1992;267:13884–13889. doi: 10.1016/S0021-9258(19)49651-8. [DOI] [PubMed] [Google Scholar]
- 35.Bonet M.L., Ribot J., Felipe F., Palou A. Vitamin A and the regulation of fat reserves. Cell. Mol. Life Sci. CMLS. 2003;60:1311–1321. doi: 10.1007/s00018-003-2290-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Blomhoff R., Blomhoff H.K. Overview of retinoid metabolism and function. J. Neurobiol. 2006;66:606–630. doi: 10.1002/neu.20242. [DOI] [PubMed] [Google Scholar]
- 37.Bonet M.L., Ribot J., Palou A. Lipid metabolism in mammalian tissues and its control by retinoic acid. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids. 2012;1821:177–189. doi: 10.1016/j.bbalip.2011.06.001. [DOI] [PubMed] [Google Scholar]
- 38.Brandebourg T.D., Hu C.Y. Regulation of differentiating pig preadipocytes by retinoic acid. J. Anim. Sci. 2005;83:98–107. doi: 10.2527/2005.83198x. [DOI] [PubMed] [Google Scholar]
- 39.Mercader J., Madsen L., Felipe F., Palou A., Kristiansen K., Bonet M.L. All-trans retinoic acid increases oxidative metabolism in mature adipocytes. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2007;20:1061–1072. doi: 10.1159/000110717. [DOI] [PubMed] [Google Scholar]
- 40.Tourniaire F., Musinovic H., Gouranton E., Astier J., Marcotorchino J., Arreguin A., Bernot D., Palou A., Bonet M.L., Ribot J., et al. All-trans retinoic acid induces oxidative phosphorylation and mitochondria biogenesis in adipocytes. J. Lipid Res. 2015;56:1100–1109. doi: 10.1194/jlr.M053652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xu Q., Fan Y., Loor J.J., Liang Y., Sun X., Jia H., Zhao C., Xu C. All-trans retinoic acid controls differentiation, proliferation, and lipolysis in isolated subcutaneous adipocytes from peripartal Holstein cows. J. Dairy Sci. 2021;104:4999–5008. doi: 10.3168/jds.2020-19408. [DOI] [PubMed] [Google Scholar]
- 42.Cinti S. Between brown and white: Novel aspects of adipocyte differentiation. Ann. Med. 2011;43:104–115. doi: 10.3109/07853890.2010.535557. [DOI] [PubMed] [Google Scholar]
- 43.Kalisz M., Chmielowska M., Martyńska L., Domańska A., Bik W., Litwiniuk A. All-trans-retinoic acid ameliorates atherosclerosis, promotes perivascular adipose tissue browning, and increases adiponectin production in Apo-E mice. Sci. Rep. 2021;11:4451. doi: 10.1038/s41598-021-83939-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Taleb S. Inflammation in atherosclerosis. Arch. Cardiovasc. Dis. 2016;109:708–715. doi: 10.1016/j.acvd.2016.04.002. [DOI] [PubMed] [Google Scholar]
- 45.Villarroya F., Iglesias R., Giralt M. Retinoids and Retinoid Receptors in the Control of Energy Balance: Novel Pharmacological Strategies in Obesity and Diabetes. Curr. Med. Chem. 2004;11:795–805. doi: 10.2174/0929867043455747. [DOI] [PubMed] [Google Scholar]
- 46.Glass C.K., Witztum J.L. Atherosclerosis: The road ahead. Cell. 2001;104:503–516. doi: 10.1016/S0092-8674(01)00238-0. [DOI] [PubMed] [Google Scholar]
- 47.Lusis A.J. Atherosclerosis. Nature. 2000;407:233–241. doi: 10.1038/35025203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Li A.C., Glass C.K. The macrophage foam cell as a target for therapeutic intervention. Nat. Med. 2002;8:1235–1242. doi: 10.1038/nm1102-1235. [DOI] [PubMed] [Google Scholar]
- 49.Ouimet M., Barrett T.J., Fisher E.A. HDL and Reverse Cholesterol Transport. Circ. Res. 2019;124:1505–1518. doi: 10.1161/CIRCRESAHA.119.312617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang N., Silver D., Thiele C., Tall A.R. ATP-binding Cassette Transporter A1 (ABCA1) Functions as a Cholesterol Efflux Regulatory Protein. J. Biol. Chem. 2001;276:23742–23747. doi: 10.1074/jbc.M102348200. [DOI] [PubMed] [Google Scholar]
- 51.Yvan-Charvet L., Ranalletta M., Wang N., Han S., Terasaka N., Li R., Welch C., Tall A.R. Combined deficiency of ABCA1 and ABCG1 promotes foam cell accumulation and accelerates atherosclerosis in mice. J. Clin. Investig. 2007;117:3900–3908. doi: 10.1172/JCI33372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ayaori M., Yakushiji E., Ogura M., Nakaya K., Hisada T., Uto-Kondo H., Takiguchi S., Terao Y., Sasaki M., Komatsu T., et al. Retinoic acid receptor agonists regulate expression of ATP-binding cassette transporter G1 in macrophages. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids. 2012;1821:561–572. doi: 10.1016/j.bbalip.2012.02.004. [DOI] [PubMed] [Google Scholar]
- 53.Wang B., Tontonoz P. Liver X receptors in lipid signalling and membrane homeostasis. Nat. Rev. Endocrinol. 2018;14:452–463. doi: 10.1038/s41574-018-0037-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yu M., Amengual J., Menon A., Kamaly N., Zhou F., Xu X., Farokhzad O.C. Targeted Nanotherapeutics Encapsulating Liver X Receptor Agonist GW3965 Enhance Antiatherogenic Effects without Adverse Effects on Hepatic Lipid Metabolism in Ldlr(−/−) Mice. Adv. Healthc. Mater. 2017;6:1700313. doi: 10.1002/adhm.201700313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mills C.D., Kincaid K., Alt J.M., Heilman M.J., Hill A.M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J. Immunol. 2000;164:6166–6173. doi: 10.4049/jimmunol.164.12.6166. [DOI] [PubMed] [Google Scholar]
- 56.Amengual J., Barrett T. Monocytes and macrophages in atherogenesis. Curr. Opin. Lipidol. 2019;30:401–408. doi: 10.1097/MOL.0000000000000634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Shao X.-J., Xiang S.-F., Chen Y.-Q., Zhang N., Cao J., Zhu H., Yang B., Zhou Q., Ying M.-D., He Q.-J. Inhibition of M2-like macrophages by all-trans retinoic acid prevents cancer initiation and stemness in osteosarcoma cells. Acta Pharmacol. Sin. 2019;40:1343–1350. doi: 10.1038/s41401-019-0262-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Vellozo N.S., Pereira-Marques S.T., Cabral-Piccin M.P., Filardy A.A., Ribeiro-Gomes F.L., Rigoni T.S., DosReis G.A., Lopes M.F. All-Trans Retinoic Acid Promotes an M1- to M2-Phenotype Shift and Inhibits Macrophage-Mediated Immunity to Leishmania major. Front. Immunol. 2017;8:1560. doi: 10.3389/fimmu.2017.01560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Alatshan A., Kovács G.E., Aladdin A., Czimmerer Z., Tar K., Benkő S. All-Trans Retinoic Acid Enhances Both the Signaling for Priming and the Glycolysis for Activation of NLRP3 Inflammasome in Human Macrophage. Cells. 2020;9:1591. doi: 10.3390/cells9071591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Dubland J.A., Francis G.A. So Much Cholesterol: The unrecognized importance of smooth muscle cells in atherosclerotic foam cell formation. Curr. Opin. Lipidol. 2016;27:155–161. doi: 10.1097/MOL.0000000000000279. [DOI] [PubMed] [Google Scholar]
- 61.Peclo M.M., Printseva O.Y. Retinoic acid enhances the proliferation of smooth muscle cells. Experientia. 1987;43:196–198. doi: 10.1007/BF01942850. [DOI] [PubMed] [Google Scholar]
- 62.Hagiwara H., Inoue A., Nakajo S., Nakaya K., Kojima S., Hirose S. Inhibition of Proliferation of Chondrocytes by Specific Receptors in Response to Retinoids. Biochem. Biophys. Res. Commun. 1996;222:220–224. doi: 10.1006/bbrc.1996.0725. [DOI] [PubMed] [Google Scholar]
- 63.Miano J.M., Topouzis S., Majesky M.W., Olson E.N. Retinoid Receptor Expression and all-trans Retinoic Acid–Mediated Growth Inhibition in Vascular Smooth Muscle Cells. Circulation. 1996;93:1886–1895. doi: 10.1161/01.CIR.93.10.1886. [DOI] [PubMed] [Google Scholar]
- 64.Hayashi A., Suzuki T., Tajima S. Modulations of Elastin Expression and Cell Proliferation by Retinoids in Cultured Vascular Smooth Muscle Cells. J. Biochem. 1995;117:132–136. doi: 10.1093/oxfordjournals.jbchem.a124699. [DOI] [PubMed] [Google Scholar]
- 65.Pakala R., Benedict C.R. RAR gamma agonists inhibit proliferation of vascular smooth muscle cells. J. Cardiovasc. Pharmacol. 2000;35:302–308. doi: 10.1097/00005344-200002000-00019. [DOI] [PubMed] [Google Scholar]
- 66.Takeda K., Ichiki T., Funakoshi Y., Ito K., Takeshita A. Downregulation of Angiotensin II Type 1 Receptor by All- trans Retinoic Acid in Vascular Smooth Muscle Cells. Hypertension. 2000;35:297–302. doi: 10.1161/01.HYP.35.1.297. [DOI] [PubMed] [Google Scholar]
- 67.Chen S., Gardner D.G. Retinoic acid uses divergent mechanisms to activate or suppress mitogenesis in rat aortic smooth muscle cells. J. Clin. Investig. 1998;102:653–662. doi: 10.1172/JCI3483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kosaka C., Sasaguri T., Komiyama Y., Takahashi H. All-trans Retinoic Acid Inhibits Vascular Smooth Muscle Cell Proliferation Targeting Multiple Genes for Cyclins and Cyclin-Dependent Kinases. Hypertens. Res. 2001;24:579–588. doi: 10.1291/hypres.24.579. [DOI] [PubMed] [Google Scholar]
- 69.Shindo T., Manabe I., Fukushima Y., Tobe K., Aizawa K., Miyamoto S., Kawai-Kowase K., Moriyama N., Imai Y., Kawakami H., et al. Krüppel-like zinc-finger transcription factor KLF5/BTEB2 is a target for angiotensin II signaling and an essential regulator of cardiovascular remodeling. Nat. Med. 2002;8:856–863. doi: 10.1038/nm738. [DOI] [PubMed] [Google Scholar]
- 70.Sakamoto H., Sakamaki T., Kanda T., Hoshino Y.I., Sawada Y., Sato M., Kurabayashi M. Smooth muscle cell outgrowth from coronary atherectomy specimens in vitro is associated with less time to restenosis and expression of a key Transcription factor KLF5/BTEB2. Cardiology. 2003;100:80–85. doi: 10.1159/000073043. [DOI] [PubMed] [Google Scholar]
- 71.Tran-Lundmark K., Tannenberg P., Rauch B.H., Ekstrand J., Tran P.-K., Hedin U., Kinsella M.G. Perlecan Heparan Sulfate Is Required for the Inhibition of Smooth Muscle Cell Proliferation by All-trans-Retinoic Acid. J. Cell. Physiol. 2014;230:482–487. doi: 10.1002/jcp.24731. [DOI] [PubMed] [Google Scholar]
- 72.Streb J.W., Long X., Lee T.-H., Sun Q., Kitchen C.M., Georger M.A., Slivano O.J., Blaner W.S., Carr D.W., Gelman I.H., et al. Retinoid-Induced Expression and Activity of an Immediate Early Tumor Suppressor Gene in Vascular Smooth Muscle Cells. PLoS ONE. 2011;6:e18538. doi: 10.1371/journal.pone.0018538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Li X., Zhang L., Yin X., Gao Z., Zhang H., Liu X., Pan X., Li N., Yu Z. Retinoic acid remodels extracellular matrix (ECM) of cultured human fetal palate mesenchymal cells (hFPMCs) through down-regulation of TGF-β/Smad signaling. Toxicol. Lett. 2014;225:208–215. doi: 10.1016/j.toxlet.2013.12.013. [DOI] [PubMed] [Google Scholar]
- 74.Neuville P., Yan Z., Gidlöf A., Pepper M.S., Hansson G.K., Gabbiani G., Sirsjo A. Retinoic acid regulates arterial smooth muscle cell proliferation and phenotypic features in vivo and in vitro through an RARalpha-dependent signaling pathway. Arterioscler. Thromb. Vasc. Biol. 1999;19:1430–1436. doi: 10.1161/01.ATV.19.6.1430. [DOI] [PubMed] [Google Scholar]
- 75.Gidlöf A.C., Ocaya P., Krivospitskaya O., Sirsjö A. Vitamin A: A drug for prevention of restenosis/reocclusion after percutaneous coronary intervention? Clin. Sci. 2008;114:19–25. doi: 10.1042/CS20070090. [DOI] [PubMed] [Google Scholar]
- 76.Zhang J., Deng B., Jiang X., Cai M., Liu N., Zhang S., Tan Y., Huang G., Jin W., Liu B., et al. All-Trans-Retinoic Acid Suppresses Neointimal Hyperplasia and Inhibits Vascular Smooth Muscle Cell Proliferation and Migration via Activation of AMPK Signaling Pathway. Front. Pharmacol. 2019;10:485. doi: 10.3389/fphar.2019.00485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yu Y., Wang Y., Fei X., Song Z., Xie F., Yang F., Liu X., Xu Z., Wang G. All-Trans Retinoic Acid Prevented Vein Grafts Stenosis by Inhibiting Rb-E2F Mediated Cell Cycle Progression and KLF5-RARα Interaction in Human Vein Smooth Muscle Cells. Cardiovasc. Drugs Ther. 2020;35:103–111. doi: 10.1007/s10557-020-07089-4. [DOI] [PubMed] [Google Scholar]
- 78.Pan H., Xue C., Auerbach B.J., Fan J., Bashore A.C., Cui J., Yang D.Y., Trignano S.B., Liu W., Shi J., et al. Single-Cell Genomics Reveals a Novel Cell State During Smooth Muscle Cell Phenotypic Switching and Potential Therapeutic Targets for Atherosclerosis in Mouse and Human. Circulation. 2020;142:2060–2075. doi: 10.1161/CIRCULATIONAHA.120.048378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Schwartz S.M., deBlois D., O’Brien E.R. The intima. Soil for atherosclerosis and restenosis. Circ. Res. 1995;77:445–465. doi: 10.1161/01.RES.77.3.445. [DOI] [PubMed] [Google Scholar]
- 80.Axel D.I., Frigge A., Dittmann J., Runge H., Spyridopoulos I., Riessen R., Viebahn R., Karsch K.R. All-trans retinoic acid regulates proliferation, migration, differentiation, and extracellular matrix turnover of human arterial smooth muscle cells. Cardiovasc. Res. 2001;49:851–862. doi: 10.1016/S0008-6363(00)00312-6. [DOI] [PubMed] [Google Scholar]
- 81.Herdeg C., Oberhoff M., Baumbach A., Schroeder S., Leitritz M., Blattner A., Siegel-Axel D.I., Meisner C., Karsch K.R. Effects of local all-trans-retinoic acid delivery on experimental atherosclerosis in the rabbit carotid artery. Cardiovasc. Res. 2003;57:544–553. doi: 10.1016/S0008-6363(02)00709-5. [DOI] [PubMed] [Google Scholar]
- 82.Johst U., Betsch A., Wiskirchen J., Schöber W., Vonthein R., Rinkert N., Kehlbach R., Claussen C.D., Duda S.H. All-Trans and 9-cis Retinoid Acids Inhibit Proliferation, Migration, and Synthesis of Extracellular Matrix of Human Vascular Smooth Muscle Cells by Inducing Differentiation In Vitro. J. Cardiovasc. Pharmacol. 2003;41:526–535. doi: 10.1097/00005344-200304000-00004. [DOI] [PubMed] [Google Scholar]
- 83.Haller H., Lindschau C., Quass P., Distler A., Luft F.C. Differentiation of Vascular Smooth Muscle Cells and the Regulation of Protein Kinase C-α. Circ. Res. 1995;76:21–29. doi: 10.1161/01.RES.76.1.21. [DOI] [PubMed] [Google Scholar]
- 84.Gollasch M., Haase H., Ried C., Lindschau C., Morano I., Luft F.C., Haller H. L-type calcium channel expression depends on the differentiated state of vascular smooth muscle cells. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 1998;12:593–601. doi: 10.1096/fasebj.12.7.593. [DOI] [PubMed] [Google Scholar]
- 85.Colbert M.C., Kirby M.L., Robbins J. Endogenous Retinoic Acid Signaling Colocalizes With Advanced Expression of the Adult Smooth Muscle Myosin Heavy Chain Isoform during Development of the Ductus Arteriosus. Circ. Res. 1996;78:790–798. doi: 10.1161/01.RES.78.5.790. [DOI] [PubMed] [Google Scholar]
- 86.Neuville P., Bochaton-Piallat M.-L., Gabbiani G. Retinoids and Arterial Smooth Muscle Cells. Arter. Thromb. Vasc. Biol. 2000;20:1882–1888. doi: 10.1161/01.ATV.20.8.1882. [DOI] [PubMed] [Google Scholar]
- 87.Blank R.S., Swartz E.A., Thompson M.M., Olson E.N., Owens G.K. A Retinoic Acid–Induced Clonal Cell Line Derived From Multipotential P19 Embryonal Carcinoma Cells Expresses Smooth Muscle Characteristics. Circ. Res. 1995;76:742–749. doi: 10.1161/01.RES.76.5.742. [DOI] [PubMed] [Google Scholar]
- 88.Wright G.L., Wang S., Bailey G., Reichenbecher V. Effect of retinoic acid on contractile competence of vascular smooth muscle. Am. J. Physiol. Circ. Physiol. 1996;270:H1363–H1370. doi: 10.1152/ajpheart.1996.270.4.H1363. [DOI] [PubMed] [Google Scholar]
- 89.Rogers M.A., Chen J., Nallamshetty S., Pham T., Goto S., Muehlschlegel J.D., Libby P., Aikawa M., Aikawa E., Plutzky J. Retinoids Repress Human Cardiovascular Cell Calcification with Evidence for Distinct Selective Retinoid Modulator Effects. Arter. Thromb. Vasc. Biol. 2020;40:656–669. doi: 10.1161/ATVBAHA.119.313366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Cuartero M.I., Ballesteros I., Moraga A., Nombela F., Vivancos J., Hamilton J.A., Corbí Á.L., Lizasoain I., Moro M.A. N2 neutrophils, novel players in brain inflammation after stroke: Modulation by the PPARγ agonist rosiglitazone. Stroke A J. Cereb. Circ. 2013;44:3498–3508. doi: 10.1161/STROKEAHA.113.002470. [DOI] [PubMed] [Google Scholar]
- 91.Brinkmann V., Reichard U., Goosmann C., Fauler B., Uhlemann Y., Weiss D.S., Weinrauch Y., Zychlinsky A. Neutrophil extracellular traps kill bacteria. Science. 2004;303:1532–1535. doi: 10.1126/science.1092385. [DOI] [PubMed] [Google Scholar]
- 92.Rafieian-Kopaei M., Setorki M., Doudi M., Baradaran A., Nasri H. Atherosclerosis: Process, indicators, risk factors and new hopes. Int. J. Prev. Med. 2014;5:927–946. [PMC free article] [PubMed] [Google Scholar]
- 93.Giugliano G., Brevetti G., Lanero S., Schiano V., Laurenzano E., Chiariello M. Leukocyte count in peripheral arterial disease: A simple, reliable, inexpensive approach to cardiovascular risk prediction. Atherosclerosis. 2010;210:288–293. doi: 10.1016/j.atherosclerosis.2009.11.009. [DOI] [PubMed] [Google Scholar]
- 94.Friedman G.D., Klatsky A.L., Siegelaub A.B. The Leukocyte Count as a Predictor of Myocardial Infarction. N. Engl. J. Med. 1974;290:1275–1278. doi: 10.1056/NEJM197406062902302. [DOI] [PubMed] [Google Scholar]
- 95.Megens R.T.A., Vijayan S., Lievens D., Döring Y., van Zandvoort M.A.M.J., Grommes J., Weber C., Soehnlein O. Presence of luminal neutrophil extracellular traps in atherosclerosis. Thromb. Haemost. 2012;107:597–598. doi: 10.1160/TH11-09-0650. [DOI] [PubMed] [Google Scholar]
- 96.Warnatsch A., Ioannou M., Wang Q., Papayannopoulos V. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science. 2015;349:316–320. doi: 10.1126/science.aaa8064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Cai W., Wang J., Hu M., Chen X., Lu Z., Bellanti J.A., Zheng S.G. All trans-retinoic acid protects against acute ischemic stroke by modulating neutrophil functions through STAT1 signaling. J. Neuroinflammation. 2019;16:175. doi: 10.1186/s12974-019-1557-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mallat Z., Taleb S., Ait-Oufella H., Tedgui A. The role of adaptive T cell immunity in atherosclerosis. J. Lipid Res. 2009;50:S364–S369. doi: 10.1194/jlr.R800092-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Chistiakov D.A., Orekhov A.N., Bobryshev Y.V. Immune-inflammatory responses in atherosclerosis: Role of an adaptive immunity mainly driven by T and B cells. Immunobiology. 2016;221:1014–1033. doi: 10.1016/j.imbio.2016.05.010. [DOI] [PubMed] [Google Scholar]
- 100.Cardilo-Reis L., Gruber S., Schreier S.M., Drechsler M., Papac-Milicevic N., Weber C., Wagner O., Stangl H., Soehnlein O., Binder C.J. Interleukin-13 protects from atherosclerosis and modulates plaque composition by skewing the macrophage phenotype. EMBO Mol. Med. 2012;4:1072–1086. doi: 10.1002/emmm.201201374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.King V.L., Szilvassy S.J., Daugherty A. Interleukin-4 deficiency decreases atherosclerotic lesion formation in a site-specific manner in female LDL receptor-/-mice. Arterioscler. Thromb. Vasc. Biol. 2002;22:456–461. doi: 10.1161/hq0302.104905. [DOI] [PubMed] [Google Scholar]
- 102.Mora J.R., Iwata M., von Andrian U.H. Vitamin effects on the immune system: Vitamins A and D take centre stage. Nat. Rev. Immunol. 2008;8:685–698. doi: 10.1038/nri2378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Iwata M., Eshima Y., Kagechika H. Retinoic acids exert direct effects on T cells to suppress Th1 development and enhance Th2 development via retinoic acid receptors. Int. Immunol. 2003;15:1017–1025. doi: 10.1093/intimm/dxg101. [DOI] [PubMed] [Google Scholar]
- 104.Brown C.C., Esterhazy D., Sarde A., London M., Pullabhatla V., Osma-Garcia I., Al-Bader R., Ortiz C., Elgueta R., Arno M., et al. Retinoic Acid Is Essential for Th1 Cell Lineage Stability and Prevents Transition to a Th17 Cell Program. Immunity. 2015;42:499–511. doi: 10.1016/j.immuni.2015.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Taleb S., Tedgui A., Mallat Z. IL-17 and Th17 cells in atherosclerosis: Subtle and contextual roles. Arterioscler. Thromb. Vasc. Biol. 2015;35:258–264. doi: 10.1161/ATVBAHA.114.303567. [DOI] [PubMed] [Google Scholar]
- 106.Korn T., Bettelli E., Oukka M., Kuchroo V.K. IL-17 and Th17 Cells. Annu. Rev. Immunol. 2009;27:485–517. doi: 10.1146/annurev.immunol.021908.132710. [DOI] [PubMed] [Google Scholar]
- 107.Eid R.E., Rao D.A., Zhou J., Lo S.F.L., Ranjbaran H., Gallo A., Tellides G. Interleukin-17 and interferon-gamma are produced concomitantly by human coronary artery-infiltrating T cells and act synergistically on vascular smooth muscle cells. Circulation. 2009;119:1424–1432. doi: 10.1161/CIRCULATIONAHA.108.827618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Proto J.D., Doran A.C., Gusarova G., Yurdagul A., Sozen E., Subramanian M., Islam M.N., Rymond C.C., Du J., Hook J., et al. Regulatory T Cells Promote Macrophage Efferocytosis during Inflammation Resolution. Immunity. 2018;49:666–677.e6. doi: 10.1016/j.immuni.2018.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Pastrana J.L., Sha X., Virtue A., Mai J., Cueto R., Lee I.A., Yang X.F. Regulatory T cells and Atherosclerosis. J. Clin. Exp. Cardiol. 2012;2012:2. doi: 10.4172/2155-9880.S12-002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sun C.-M., Hall J.A., Blank R.B., Bouladoux N., Oukka M., Mora J.R., Belkaid Y. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 2007;204:1775–1785. doi: 10.1084/jem.20070602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.von Boehmer H. Oral tolerance: Is it all retinoic acid? J. Exp. Med. 2007;204:1737–1739. doi: 10.1084/jem.20071251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Coombes J.L., Maloy K.J. Control of intestinal homeostasis by regulatory T cells and dendritic cells. Semin. Immunol. 2007;19:116–126. doi: 10.1016/j.smim.2007.01.001. [DOI] [PubMed] [Google Scholar]
- 113.Coombes J.L., Siddiqui K.R., Arancibia-Cárcamo C.V., Hall J., Sun C.M., Belkaid Y., Powrie F. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-beta and retinoic acid-dependent mechanism. J. Exp. Med. 2007;204:1757–1764. doi: 10.1084/jem.20070590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Pino-Lagos K., Guo Y., Brown C., Alexander M.P., Elgueta R., Bennett K.A., De Vries V., Nowak E., Blomhoff R., Sockanathan S., et al. A retinoic acid–dependent checkpoint in the development of CD4+ T cell–mediated immunity. J. Exp. Med. 2011;208:1767–1775. doi: 10.1084/jem.20102358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Shevach E.M., Rosenthal A.S. Function of macrophages in antigen recognition by guinea pig T lymphocytes. II. Role of the macrophage in the regulation of genetic control of the immune response. J. Exp. Med. 1973;138:1213–1229. doi: 10.1084/jem.138.5.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Mills C.D., Ley K. M1 and M2 Macrophages: The Chicken and the Egg of Immunity. J. Innate Immun. 2014;6:716–726. doi: 10.1159/000364945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Lin J.-D., Nishi H., Poles J., Niu X., McCauley C., Rahman K., Brown E.J., Yeung S.T., Vozhilla N., Weinstock A., et al. Single-cell analysis of fate-mapped macrophages reveals heterogeneity, including stem-like properties, during atherosclerosis progression and regression. JCI Insight. 2019;4:e124574. doi: 10.1172/jci.insight.124574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Hansson G.K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 2005;352:1685–1695. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- 119.Sugino I., Kuboki K., Matsumoto T., Murakami E., Nishimura C., Yoshino G. Influence of fatty liver on plasma small, dense LDL- cholesterol in subjects with and without metabolic syndrome. J. Atheroscler. Thromb. 2011;18:1–7. doi: 10.5551/jat.5447. [DOI] [PubMed] [Google Scholar]
- 120.Bilbija D., Elmabsout A.A., Sagave J., Haugen F., Bastani N., Dahl C.P., Gullestad L., Sirsjö A., Blomhoff R., Valen G. Expression of retinoic acid target genes in coronary artery disease. Int. J. Mol. Med. 2014;33:677–686. doi: 10.3892/ijmm.2014.1623. [DOI] [PubMed] [Google Scholar]
- 121.Grace V.B., Siddikuzzaman, Rimashree B. Liposome encapsulated all trans retinoic acid (ATRA) has enhanced immunomodulatory and inflammation reducing activities in mice model. Anti-Cancer Agents Med. Chem. 2015;15:196–205. doi: 10.2174/1871520615666150116104538. [DOI] [PubMed] [Google Scholar]
- 122.Reriani M., Raichlin E., Prasad A., Mathew V., Pumper G.M., Nelson R.E., Lennon R., Rihal C., Lerman L.O., Lerman A. Long-Term Administration of Endothelin Receptor Antagonist Improves Coronary Endothelial Function in Patients With Early Atherosclerosis. Circulation. 2010;122:958–966. doi: 10.1161/CIRCULATIONAHA.110.967406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Gimbrone M.A., Jr., García-Cardeña G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016;118:620–636. doi: 10.1161/CIRCRESAHA.115.306301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Achan V., Tran C.T., Arrigoni F., Whitley G.S., Leiper J.M., Vallance P. all-trans-Retinoic acid increases nitric oxide synthesis by endothelial cells: A role for the induction of dimethylarginine dimethylaminohydrolase. Circ. Res. 2002;90:764–769. doi: 10.1161/01.RES.0000014450.40853.2B. [DOI] [PubMed] [Google Scholar]
- 125.Rhee E.-J., Nallamshetty S., Plutzky J. Retinoid metabolism and its effects on the vasculature. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids. 2012;1821:230–240. doi: 10.1016/j.bbalip.2011.07.001. [DOI] [PubMed] [Google Scholar]
- 126.Yokota J., Kawana M., Hidai C., Aoka Y., Ichikawa K.-I., Iguchi N., Okada M., Kasanuki H. Retinoic acid suppresses endothelin-1 gene expression at the transcription level in endothelial cells. Atherosclerosis. 2001;159:491–496. doi: 10.1016/S0021-9150(01)00530-5. [DOI] [PubMed] [Google Scholar]
- 127.Tao L., Nie Y., Wang G., Ding Y., Ding J., Xiong F., Tang S., Wang Y., Zhou B., Zhu H. All-trans retinoic acid reduces endothelin-1 expression and increases endothelial nitric oxide synthase phosphorylation in rabbits with atherosclerosis. Mol. Med. Rep. 2017;17:2619–2625. doi: 10.3892/mmr.2017.8156. [DOI] [PubMed] [Google Scholar]
- 128.Wu Y., Wang X., Zhou Q., Wang Y., Zhou J., Jiang Q., Wang Y., Zhu H. ATRA improves endothelial dysfunction in atherosclerotic rabbits by decreasing CAV-1 expression and enhancing eNOS activity. Mol. Med. Rep. 2018;17:6796–6802. doi: 10.3892/mmr.2018.8647. [DOI] [PubMed] [Google Scholar]
- 129.Amisten S., Al-Amily I.M., Soni A., Hawkes R., Atanes P., Persaud S.J., Rorsman P., Salehi A. Anti-diabetic action of all-trans retinoic acid and the orphan G protein coupled receptor GPRC5C in pancreatic β-cells. Endocr. J. 2017;64:325–338. doi: 10.1507/endocrj.EJ16-0338. [DOI] [PubMed] [Google Scholar]
- 130.Blaner W.S., Shmarakov I.O., Traber M.G. Vitamin A and Vitamin E: Will the Real Antioxidant Please Stand Up? Annu. Rev. Nutr. 2021;41:105–131. doi: 10.1146/annurev-nutr-082018-124228. [DOI] [PubMed] [Google Scholar]
- 131.Cabrera-Valladares G., German M.S., Matschinsky F.M., Wang J., Fernandez-Mejia C. Effect of Retinoic Acid on Glucokinase Activity and Gene Expression and on Insulin Secretion in Primary Cultures of Pancreatic Islets. Endocrinology. 1999;140:3091–3096. doi: 10.1210/endo.140.7.6765. [DOI] [PubMed] [Google Scholar]
- 132.Said E., Mousa S., Fawzi M., Sabry N., Farid S. Combined effect of high-dose vitamin A, vitamin E supplementation, and zinc on adult patients with diabetes: A randomized trial. J. Adv. Res. 2020;28:27–33. doi: 10.1016/j.jare.2020.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Bilbija D., Haugen F., Sagave J., Baysa A., Bastani N., Levy F.O., Sirsjö A., Blomhoff R., Valen G. Retinoic Acid Signalling Is Activated in the Postischemic Heart and May Influence Remodelling. PLoS ONE. 2012;7:e44740. doi: 10.1371/journal.pone.0044740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Oliveira L.C., Azevedo P.S., Minicucci M.F., Rafacho B.P.M., Duarte D.R., Matsubara L.S., Matsubara B.B., Paiva S., Zornoff L.A.M. Retinoic acid prevents ventricular remodelling induced by tobacco smoke exposure in rats. Acta Cardiol. 2011;66:3–7. doi: 10.1080/AC.66.1.2064960. [DOI] [PubMed] [Google Scholar]
- 135.Subramanian U., Nagarajan D. All-Trans Retinoic Acid supplementation prevents cardiac fibrosis and cytokines induced by Methylglyoxal. Glycoconj. J. 2017;34:255–265. doi: 10.1007/s10719-016-9760-5. [DOI] [PubMed] [Google Scholar]
- 136.Danzl K., Messner B., Doppler C., Nebert C., Abfalterer A., Sakic A., Temml V., Heinz K., Streitwieser R., Edelmann T., et al. Early inhibition of endothelial retinoid uptake upon myocardial infarction restores cardiac function and prevents cell, tissue, and animal death. J. Mol. Cell. Cardiol. 2018;126:105–117. doi: 10.1016/j.yjmcc.2018.11.012. [DOI] [PubMed] [Google Scholar]
- 137.Koshiuka K., Elstner E., Williamson E., Said J.W., Tada Y., Koeffler H.P. Novel therapeutic approach: Organic arsenical (melarsoprol) alone or with all-trans-retinoic acid markedly inhibit growth of human breast and prostate cancer cells in vitro and in vivo. Br. J. Cancer. 2000;82:452–458. doi: 10.1054/bjoc.1999.0942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Lin L.-M., Li B.-X., Xiao J.-B., Lin D.-H., Yang B.-F. Synergistic effect of all-trans-retinoic acid and arsenic trioxide on growth inhibition and apoptosis in human hepatoma, breast cancer, and lung cancer cells in vitro. World J. Gastroenterol. 2005;11:5633–5637. doi: 10.3748/wjg.v11.i36.5633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Verhagen H.J.M.P., Smit M.A., Rutten A., Denkers F., Poddighe P.J., Merle P.A., Ossenkoppele G.J., Smit L. Primary acute myeloid leukemia cells with overexpression of EVI-1 are sensitive to all-trans retinoic acid. Blood. 2016;127:458–463. doi: 10.1182/blood-2015-07-653840. [DOI] [PubMed] [Google Scholar]
- 140.Ni X., Hu G., Cai X. The success and the challenge of all-trans retinoic acid in the treatment of cancer. Crit. Rev. Food Sci. Nutr. 2018;59:S71–S80. doi: 10.1080/10408398.2018.1509201. [DOI] [PubMed] [Google Scholar]
- 141.Yi X., Wang Y., Jia Z., Hiller S., Nakamura J., Luft J.C., Tian S., DeSimone J.M. Retinoic Acid-Loaded Poly(lactic-co-glycolic acid) Nanoparticle Formulation of ApoB-100-Derived Peptide 210 Attenuates Atherosclerosis. J. Biomed. Nanotechnol. 2020;16:467–480. doi: 10.1166/jbn.2020.2905. [DOI] [PMC free article] [PubMed] [Google Scholar]