Abstract
Androgen/androgen receptor (AR) signaling pathways are essential for prostate tumorigenesis. However, the fundamental mechanisms underlying the AR functioning as a tumor promoter in inducing prostatic oncogenesis still remain elusive. Here, we demonstrate that a subpopulation of prostatic Osr1 (odd skipped-related 1)-lineage cells functions as tumor progenitors in prostate tumorigenesis. Single cell transcriptomic analyses reveal that aberrant AR activation in these cells elevates insulin-like growth factor 1 (IGF1) signaling pathways and initiates oncogenic transformation. Elevating IGF1 signaling further cumulates Wnt/β-catenin pathways in transformed cells to promote prostate tumor development. Correlations between altered androgen, IGF1, and Wnt/β-catenin signaling are also identified in human prostate cancer samples, uncovering a dynamic regulatory loop initiated by the AR through prostate cancer development. Co-inhibition of androgen and Wnt-signaling pathways significantly represses the growth of AR-positive tumor cells in both ex-vivo and in-vivo, implicating co-targeting therapeutic strategies for these pathways to treat advanced prostate cancer.
Subject terms: Cancer models, Oncogenes, Prostate cancer
Activation of the androgen receptor (AR) through androgen binding is essential for prostate tumorigenesis. Here the authors show that AR activation in a subpopulation of prostatic progenitor cells can initiate prostatic intraepithelial neoplasia formation and promotes prostate cancer development through activation of IGF1 and Wnt/β-catenin signalling pathways.
Introduction
Prostate cancer is one of the most common malignancies in men and claims about 250,000 deaths annually worldwide1. The activation of AR through binding of androgens is essential for prostate tumorigenesis2,3. Almost all primary prostate cancer cells express the AR and are dependent on androgens for their oncogenic growth and survival4. However, the fundamental mechanisms by which the AR, a steroid hormone receptor, functions as a tumor promoter to initiate prostatic oncogenesis and promote tumor progression still remains elusive. Although androgen deprivation therapy (ADT) directly targeting AR-expressing tumor cells is still the first-line treatment for advanced prostate cancer5, it eventually fails in most patients who consequently develop castration resistant prostate cancer (CRPC), an incurable disease2,6. Therefore, oncogenic AR action in prostate cancer development and progression needs to be understood for designing more effective therapies.
Currently, one of the significant challenges in the field of prostate cancer research is still the lack of appropriate animal models that can recapitulate the oncogenic role of AR during prostate cancer initiation, progression, and hormone refractoriness. While a variety of AR transgenic mouse models have been developed for assessing AR action in prostate oncogenesis in the past decades7, these models all used the mouse probasin (PB) promoter to induce transgenic AR expression and only showed minor pathological changes and failed to develop prostate tumor lesions as observed in human prostate cancer8,9. However, activation of the “floxed” human AR transgene (hARtg) expression in a subpopulation of prostatic progenitors through the Osr1 (odd skipped related1) promoter-driven Cre recombinases developed high-grade prostatic intraepithelial neoplasia (HGPIN) and invasive prostatic adenocarcinoma (PCa) lesions in mice10. The expression of transgenic AR was identified specifically in both prostatic atypical and tumor cells of the transgenic mice10, directly demonstrating a critical role of hARtg expression in HGPIN and prostate tumor development. Additionally, development of both HGPIN and prostatic adenocarcinoma lesions in the AR transgenic mice regulated through Osr1-Cre but not PB-Cre implicates the critical cellular properties of prostatic Osr1-expressing cells and their derivatives, termed Osr1-lineage cells, in initiating oncogenesis and tumor development10.
In this study, using single-cell RNA sequencing (scRNA-seq), genetic tracing, and other experimental approaches, we uncover an underlying mechanism by which aberrant AR activation elevates IGF1 signaling pathways in prostatic Osr1-lineage cells to initiate PIN formation. Subsequently, activated IGF1 signaling further cumulates Wnt/β-catenin activation in atypical PIN cells to promote tumor development. The correlations in co-occurrences of altered androgen, IGF1, and Wnt/β-catenin signaling pathways were further identified in human prostate cancer samples. Co-inhibition of androgen and Wnt signaling pathways showed more potent repressive effects on the growth of hARtg+ prostate tumor cells in ex-vivo and xenograft models than either reagent alone, elucidating the promotional role of Wnt activation in hARtg-induced prostate tumorigenesis. These findings uncover a dynamic regulatory loop initiated by aberrant AR expression in prostatic progenitors through altering IGF1 and Wnt signaling pathways in inducing prostate oncogenic transformation and tumor development. They also provide scientific evidence leading to the development of future therapeutic strategies by co-targeting these signaling pathways for achieving better clinical outcomes for advanced prostate cancer.
Results
Prostatic Osr1-lineage cells possess progenitor properties in expansion of prostatic epithelia through embryonic and pubescent development
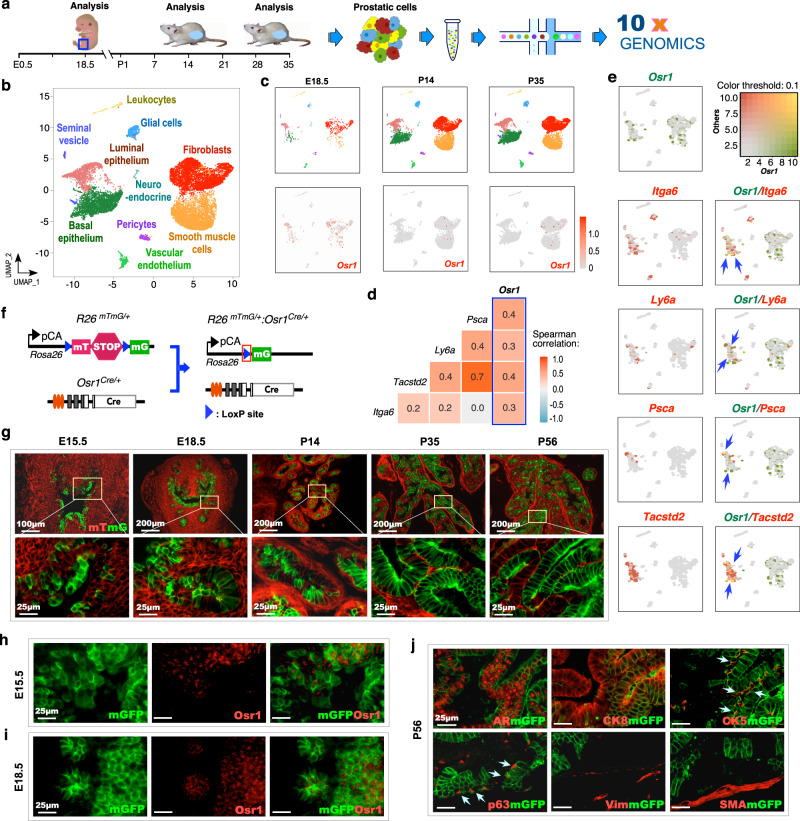
Development of HGPIN and prostatic adenocarcinoma lesions in Osr1-Cre-driven hARtg transgenic mice but not in previous AR transgenic models regulated by the PB promoter implicates the critical role of prostatic Osr1-lineage cells in prostate tumorigenesis10. To assess the cellular properties of prostatic Osr1-lineage cells, we examined the molecular characterization of these cells using scRNA-seq analyses with urogenital sinus (UGS), and prepubescent and pubertal prostate tissues (Fig. 1a). Approximately 766, 5676, and 8253 cells from embryonic day E18.5 UGS tissues, and postnatal day P14 and P35 prostate tissues were obtained after filtering, respectively (Supplementary Fig. 1), and then merged and clustered after performing cell cycle regression. Both epithelial and stromal cell subsets were identified in the merged Uniform Manifold Approximation and Projection (UMAP) plots (Fig. 1b), aligning similar cell types based on their transcriptomic profiles using Seurat’s integrated method11,12 (Supplementary Fig. 2a–c). These cell subsets also appeared in individual UMAP plots, suggesting comparable cellular properties from UGS and prostate tissue samples (Fig. 1c, top panel). Osr1 expression appeared in both mesenchymal and epithelial cells at E18.5 UGS samples but significantly decreased in both total cells and total epithelial cells of P14 (p = 0.004 and p = 0.002, respectively) and P35 (p = 0.003 and p = 0.002, respectively) samples (Fig. 1c, bottom panel; Supplementary Fig. 2d, e). A significant positive correlation between expression of Osr1 with prostatic stem/progenitor cell markers including Itga6, Ly6a and Tacstd213, as well as Psca14, a prostate cancer stem cell marker, (Spearman r = 0.3, 0.31, 0.4, and 0.41, respectively), was identified in urogenital sinus epithelium (UGE) but not urogenital sinus mesenchyme (UGM) cells (Fig. 1d vs. Supplementary Fig. 2f), suggesting the progenitor properties of Osr1-lineage cells in prostatic epithelial development. Co-expression UMAP plots also showed overlaying Osr1 with the above cellular markers in epithelial cell clusters of E18.5 samples (arrows, Fig. 1e). To assess the differential fates of embryonic prostatic Osr1-expressing cells, we traced them through embryonic and postnatal prostate developmental stages using Osr1-Cre activated membrane-bound green fluorescent protein (mGFP) expression in Rosa26 mTmG-LoxP/+:Osr1Cre/+ (R26 mTmG/+:Osr1Cre/+) reporter mice (Fig. 1f). The expression of mGFP activated by Osr1-Cre mainly appeared in urogenital epithelium and prostatic buds at E15.5 and 18.5, respectively, and mGFP+ cells were expanded robustly in the epithelium of prostatic glands at P14, 35, and 56, through and after puberty (Fig. 1g). Co-immunofluorescence analyses (Co-IF) showed very limited Osr1+mGFP+ cells but many more mGFP+ cells in both E15.5 and E18.5 UGS samples (Fig. 1h, i). Robust mGFP+ cells also appear in postnatal prostate epithelia (Fig. 1g), demonstrating the ability of Osr1-expressing cells to expand prostatic epithelia through pubertal prostate development. Co-IF analysis of P56 prostate tissues showed the majority of mGFP+ cells co-stained with endogenous AR or CK8 (Fig. 1j; Supplementary Fig. 2g1–h4). Intriguingly, a portion of mGFP+ cells also showed positive staining for CK5 and p63, basal epithelial cell markers, in the above prostate sections (blue arrows, Fig. 1j; Supplementary Fig. 2i1–j4). No overlay between mGFP with vimentin (Vim) or smooth muscle actin (SMA) staining was observed (Fig. 1j; Supplementary Fig. 2k1–l4). These data consistently demonstrate the regulatory role of Osr1-lineage cells in prostatic epithelial development and growth. Accordingly, more mGFP+ epithelial cells were observed in prostate tissues, particularly in the anterior and dorsal lobes, isolated from R26 mTmG/+:Osr1Cre/+ mice at both P28 and P56 days than those from R26 mTmG/+:PBCre/+ counterparts (Supplementary Fig. 3a–f4). Moreover, TP63 + mGFP+ basal epithelial cells were detected in prostate tissues of R26 mTmG/+:Osr1Cre/+ mice but not in those of R26 mTmG/+:PB Cre/+ mice (yellow or blue arrows, Supplementary Fig. 3g1-1’ vs 3g2–2’). Taken together, these data demonstrate that prostatic Osr1-lineage cells possess basal progenitor properties and are able to expand prostatic epithelial cell populations during prostate development.
Fig. 1. Prostatic Osr1-expressing cells possess progenitor properties in expansion of prostatic epithelia through embryonic and pubescent development.
a Schematic of the single cell RNA-sequencing experiment performed. b Uniform Manifold Approximation and Projection (UMAP) plot showing the cell type cluster identities based on gene expression patterns. c Three individual UMAP plots of single cells at the indicated time points after separation from the original clustering. Gene expression UMAP plots displaying the expression pattern of the Osr1 gene at the indicated time points. Color intensity indicates the scaled expression level in each cell. d Heatmap of pairwise Spearman correlation between the indicated gene expression in epithelial cells from E18.5 male urogenital sinus (UGS). Colors reflect the level of correlation (positive correlation in red and negative correlation in blue); numbers show the correlation coefficient. The blue box highlights the correlation between Osr1 and other progenitor cell markers. e Blended expression UMAP plots displaying the expression of Osr1 and the indicated genes in the E18.5 male UGS. Blue arrows show the overlay (yellow color) of Osr1 and the indicated genes in the E18.5 male UGS. f Schematics of the R26mTmG/+ and Osr1Cre/+ alleles, shown in relation to the mating strategy for this experiment. g Representative fluorescence images of mouse UGS or prostate tissues at the indicated timepoints expressing mGFP reporter fluorescence (green) controlled by Cre recombination. Scale bars, 100 µm; 200 µm; 25 µm. h–j Representative fluorescence images of co-immunofluoresence (IF) staining for the indicated proteins using mouse UGS or prostate tissues at the indicated timepoints. Blue arrows indicate the overlay between mGFP and cytokeratin 5 (CK5) or p63, basal epithelial cell markers j. AR, androgen receptor; CK8, cytokeratin 8; Vim, vimentin; SMA, smooth muscle actin. Scale bars, 25 µm. Representative images from three independent experiments with similar results are displayed for each micrograph. See also Supplementary Figs. 1–3.
Aberrant expression of hARtg regulates transcriptome of prostatic Osr1-lineage cells and induces PIN and prostatic adenocarcinoma development
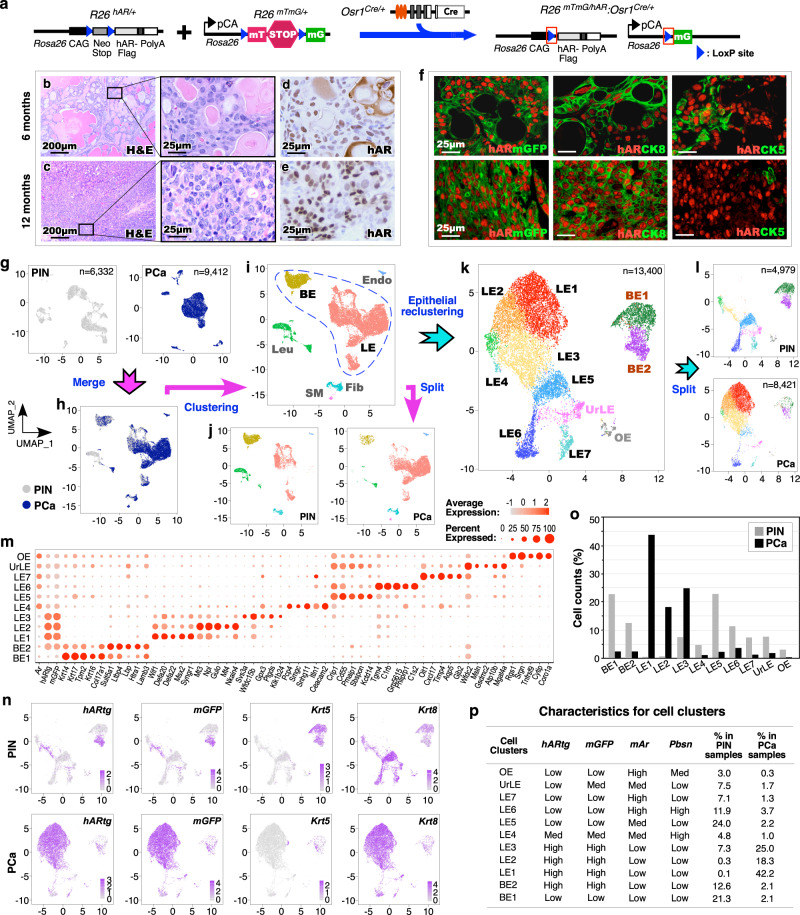
Development of HGPIN and PCa lesions in Osr1-Cre-driven hARtg mice suggests the promotional role of hARtg in Osr1-lineage cells in prostate tumorigenesis10. To better understand the oncogenic role of hARtg in Osr1-lineage cells, we generated R26 mTmG-LoxP/hAR-LoxP:Osr1Cre/+ (R26mTmG/hAR:Osr1Cre/+) mice, in which the expression of mGFP and transgenic AR co-occurs through Osr1-Cre mediated activation (Fig. 2a). Both HGPIN and prostatic PCa lesions developed in R26mTmG/hAR:Osr1Cre/+ mice starting at 4 and 10 months, respectively (Supplementary Table 1) but not in control littermates, which is similar as observed in R26hAR/+:Osr1Cre/+ mice10. Prostate tissues isolated from 6- and 12-month-old R26mTmG/hAR:Osr1Cre/+ mice showed typical HGPIN and PCa lesions (Fig. 2b, c), respectively, which were used for preparing scRNA-seq analyses (see below). A uniform nuclear staining for hARtg appeared in both atypical and tumor cells within PIN and PCa lesions (Fig. 2d, e). Co-staining of hARtg and mGFP were also revealed in both atypical and tumor cells (Fig. 2f), demonstrating their origin deriving from Osr1-expressing cells. Interestingly, while the majority of atypical and tumor cells showed positive staining for both hARtg and CK8, a portion of atypical cells showed a clear overlay of hARtg and CK5 expression within PIN lesions (Fig. 2f). The expression of both hARtg and mGFP in atypical and tumor cells within HGPIN and PCa lesions demonstrate the critical role of transgenic AR in Osr1-lineage cells in inducing prostatic oncogenesis and promoting PIN and PCa development.
Fig. 2. Single cell transcriptomic analyses of prostatic intraepithelial neoplasia and adenocarcinoma tissues originating from Osr1-expressing cells with hARtg expression.
a Schematic of the floxed human androgen receptor (hAR) transgene (R26hAR/+) and R26mTmG/+ alleles, as well as the corresponding recombined Osr1Cre/+ alleles, shown in relation to the mating strategy for this experiment. b–f Representative images of hematoxylin-eosin (H&E), immunohistochemistry (IHC), and co-IF staining using the indicated antibodies on adjacent prostate tissue sections from R26mTmG/hAR:Osr1Cre/+ mice. Scale bars, 100 µm; 25 µm. g Two individual UMAP plots of single cells isolated from prostatic intraepithelial neoplasia (PIN; gray) and prostate adenocarcinoma (PCa; dark blue) tissues, respectively. h UMAP visualization of the single cells following integration and clustering of the PIN (gray) and PCa (dark blue) cells. i Cell type cluster identities shown on the UMAP of the single cells based on their gene expression profiles. BE, basal epithelium; LE, luminal epithelium; Fib, fibroblasts; SM, smooth muscle cells; Endo, vascular endothelium; Leu, leukocytes. j UMAP plot from i is split by PIN or PCa cells colored by cell type. k UMAP plot of epithelial cells from PIN and PCa tissues. Epithelial cells were sub-clustered, re-clustered and colored by cell cluster. UrLE, urethral epithelium; OE, other epithelium. l UMAP plot from k is separated by PIN or PCa samples colored by cell type. m Dot plot of Ar, human AR transgene (hARtg), mGFP, as well as five cluster-specific genes for each epithelial cell cluster. Dot size indicates the percentage of cells in a cluster expressing each gene; color shows expression level. n Gene expression UMAP plots for the indicated genes after separating PIN (top) and PCa (bottom) samples. Color intensity indicates the scaled expression level. o Bar chart showing the cell counts within individual clusters from epithelial cells of PIN (gray) or PCa (black) tissues. Source data are provided as a Source Data file. p Table summarizing the characteristics for each cell cluster from PIN or PCa samples. Representative images with consistent results from three replicates are shown. See also Supplementary Fig. 4.
To gain in-depth mechanistic insight into the oncogenic role of hARtg, we performed scRNA-seq analyses using the above pathologically confirmed HGPIN and PCa tissues (see Methods). Both scRNA-seq samples prepared from PIN and PCa tissues underwent multiple steps of filtering following sequencing and alignment to the mm10 reference genome with addition of mGFP and hARtg sequences15. Post-filtering, 6332 or 9412 cells from PIN or PCa samples, with an average of 3751 or 3532 genes and 21,297 or 19,899 UMI counts per cell, respectively, were used in the study (Supplementary Fig. 4a, b). Both PIN and PCa samples were initially visualized individually using UMAP (Fig. 2g), and then merged and clustered, aligning similar cell subsets based on their transcriptomic profiles using Seurat’s integrated method11 (Fig. 2h). A total of 6 cell subsets were identified in both merged and individual PIN and PCa plots (Fig. 2i, j), demonstrating comparable cellular properties between these two samples. Specifically, the expression of hARtg and mGFP are comparable and restricted within the luminal and basal epithelial cell subsets in combined UMAP plots (Supplementary Fig. 4c). Those epithelial cell subsets were further validated with representative epithelial markers (Supplementary Fig. 4d). To gain higher resolution of hARtg + cells in PIN and PCa samples, epithelial cells were separated from other non-epithelial cells, and re-clustered following cell cycle regression (Supplementary Fig. 4e)16. Eleven cell clusters were yielded, including two basal, seven luminal, a urethral epithelial (UrLE)17, and another epithelial cluster, deemed other epithelia (OE) (Fig. 2k). Five highly expressed genes in each cell cluster were identified to represent their cellular properties (Fig. 2m). Prostatic basal epithelial cells showed high expression of Krt5, Krt14, and Trp63, while luminal cell clusters displayed elevated expression of Krt18, Krt19, and Pbsn, respectively18. The expression of hARtg and mGFP appears mainly in BE2 and LE1-3 clusters, and the expression of endogenous Ar is restricted within LE4-7 clusters. All cell clusters appeared in both individual PIN and PCa UMAP plots. However, luminal epithelial cell clusters, LE1 to 3, appeared predominantly in PCa samples, and the rest of the cell clusters showed abundantly in PIN samples (Fig. 2l). UMAP expression plots showed Krt5 and Krt8 expression selectively in basal and luminal epithelial cell clusters of both PIN and PCa samples while hARtg and mGFP expression was mainly revealed within the BE2 cluster in PIN, and LE1-3 clusters in PCa samples (Fig. 2n). Cell distribution analyses further confirmed luminal clusters (LE1-3) being mostly in PCa samples and other cell clusters being abundant in PIN samples (Fig. 2o). LE1-2 clusters showed relatively high expression of hARtg and mGFP but little or no expression of mouse Ar and Pbsn (Fig. 2p), suggesting their transformed and un-differentiated cellular properties. These data provide high-resolution insight into the cellular properties of prostatic hARtg+ atypical and tumor cells derived from Osr1-lineage, implicating the direct role of transgenic AR activation in PIN and prostate tumor development in R26mTmG/hAR:Osr1Cre/+ mice.
Transgenic AR expression elevates IGF1 signaling in atypical Osr1-lineage basal epithelial cells within PIN lesions
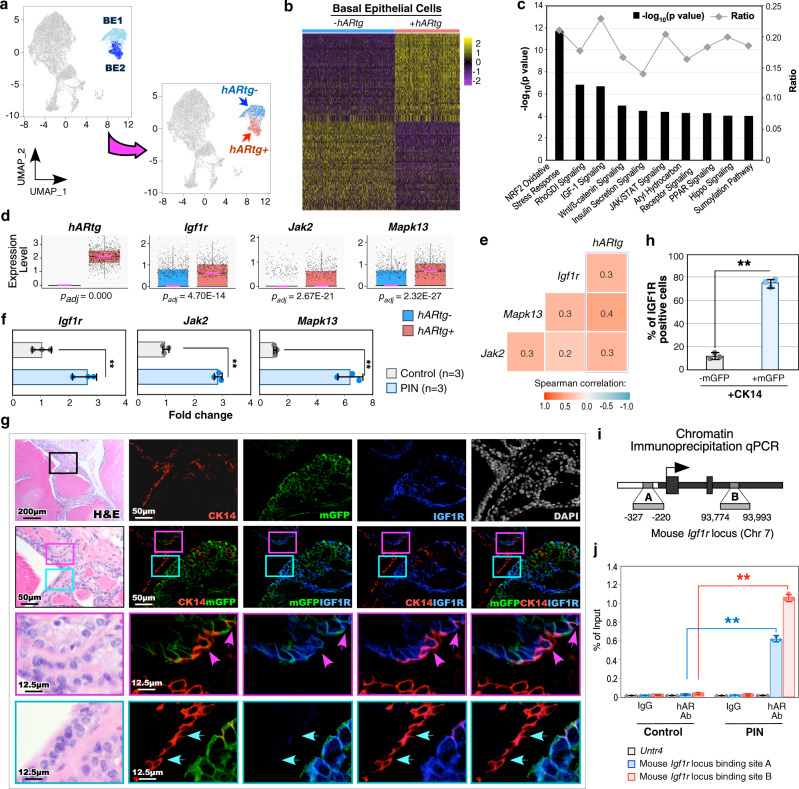
Identifying specific expression of hARtg in a subpopulation of atypical basal epithelial cells within BE2 cluster in R26mTmG/hAR:Osr1Cre/+ samples (Fig. 2m, n, p) suggests the important role of these hARtg+ basal epithelial cells in PIN initiation (Fig. 3a). Additionally, robust mGFP+TP63+ prostatic basal epithelial cells revealed in prostatic tissues of R26mTmG/+:Osr1Cre/+ mice but not in those of R26mTmG/+:PB Cre/+ counterparts (Supplementary Fig. 3g1, 2’). Moreover, only R26mTmG/hAR:Osr1Cre/+ mice developed HGPIN and PCa lesions but not in AR transgenic mice driven by PB promoters8–10,19. These data consistently demonstrate the significance of the hARtg+ atypical basal cells in initiating prostate oncogenesis. To gain direct insight into the regulatory role of hARtg in basal epithelial cells, we identified the differentially expressed genes (DEGs) between hARtg+ and hARtg−basal epithelial cells of PIN and PCa samples using a Wilcoxon Rank Sum test, which showed more than 5% of cells with adjusted p values < 0.05 by the Benjamini-Hochberg procedure and average log fold change >0.1 (Fig. 3a, b; Supplementary Data 1). Ingenuity pathway analysis (IPA) of the above DEGs identified significant enrichment in IGF1, Insulin secretion, Wnt/β-catenin, and JAK/STAT signaling pathways (Fig. 3c), which directly regulate prostate tumorigenesis20. Specifically, identifying enriched IGF1 and Insulin related signaling pathways in hARtg+ basal cells is intriguing. Multiple lines of evidence have shown that IGF1 signaling directly induces and promotes PIN development in both mouse prostate cancer models and human prostate cancers21–23. A significant increase in hARtg and Igf1r, as well as IGF1 signaling downstream genes, Jak2 and Mapk13 expression was further identified in hARtg+ compared to hARtg- basal epithelial cells using box plots (Fig. 3d). A correlation between hARtg with Igf1r or its downstream targets, Jak2 and Mapk13, was further identified using Spearman gene-gene correlation analysis (Fig. 3e). Quantitative reverse transcription-PCR (qRT-PCR) analyses showed the higher expression of IGF1 signaling effectors, including Igf1r, Jak2, and Mapk13, in PIN tissue samples of R26mTmG/hAR:Osr1Cre/+ mice than prostate tissues of R26mTmG/+:Osr1Cre/+ controls (Fig. 3f). Using triple-IF analyses, the regulatory role of transgenic AR in activating IGF1R axis was further assessed in PIN tissues of R26mTmG/hAR:Osr1Cre/+ mice. Whereas CK14 expression revealed in basal epithelial cells in both abnormal PIN prostatic glands and adjacent normal glands (Fig. 3g), co-expression of mGFP, representing hARtg-expressing cells, and IGF1R appeared mainly in atypical cells within PIN areas (Fig. 3g). Overlay of CK14 and mGFP, mGFP and IGF1R, or CK14 and IGF1R appeared selectively in atypical cells within the above PIN lesions (pink boxes and arrows, Fig. 3g) but no or very few cells with the above overlays were observed within normal glandular areas (blue boxes and arrows, Fig. 3g). Triple positive cells of CK14, mGFP, and IGF1R revealed only in atypical cells within abnormal prostatic glands (pink boxes pink arrows, Fig. 3g, right panels), but not in normal glandular cells (blue boxes and arrows, Fig. 3g, right panels). Quantified IGF1R expression in approximately a total of 500 to 800 CK14 + mGFP+ or CK14 + mGFP− cells from five different areas in each sample showed significantly more IGF1R expression in CK14 + mGFP+ cells than CK14 + mGFP− cells in three different experiments with three tissue samples prepared from three different R26mTmG/hAR:Osr1Cre/+ mice (p = 0.00001) (Fig. 3h).
Fig. 3. Transgenic AR expression elevates IGF1 signaling in atypical Osr1-driven prostatic basal epithelial cells within PIN lesions.
a UMAP plot indicating basal epithelial (BE) cells from PIN and PCa tissues of R26mTmG/hAR:Osr1Cre/+ mice (left), colored by hARtg expression (right). b Heatmap showing top 50 differentially expressed genes (DEGs) between hARtg + and hARtg- BE cells. c Ingenuity pathway analysis (IPA) pathway analysis of DEGs comparing hARtg + and hARtg- BE cells. Fisher’s exact test (two-sided). Ratio denotes the number of DEGs compared with the total number of genes associated with the canonical pathway. d Box-plots representing scaled expression for the indicated genes between hARtg + (n = 749) and hARtg- (n = 933) BE cells. Pink lines mark the median; top and bottom lines of boxes indicate the boundaries of the first and third quartiles, respectively; the top and bottom whiskers show the maximum and minimum values, respectively, excluding outliers. Wilcoxon rank-sum test (two-sided) followed by Benjamini-Hochberg correction. e Heatmap of pairwise Spearman correlation between indicated gene expression in BE cells. Numbers indicate correlation coefficient. f RT-qPCR analysis of the indicated genes shown as fold change in the indicated tissues. g Representative images of H&E and triple-IF staining for the indicated antibodies on adjacent prostate tissues from R26mTmG/hAR:Osr1Cre/+ mice. Pink and blue boxes indicate abnormal and normal glandular areas, respectively, and pink and blue arrows indicate atypical and normal cells, respectively. Nuclei were stained with DAPI. Scale bars, 200 µm; 50 µm; 12.5 µm. h Bar chart for the percentage of IGF1R + cells in mGFP− (black) or mGFP + (gray) CK14-expressing BE cells. i Diagram showing the mouse Igf1r locus containing two AR-binding sites. j hAR ChIP-qPCR analysis shown as percent input of Igf1r gene on its binding sites A and B in the indicated tissues. In f, h, and j, data are represented as mean ± SD of three biological replicates. Two-sided t-test, **p < 0.01. Representative images with consistent results from three replicates are shown. See also Supplementary Fig. 5 and Supplementary Data 1, 2. Source data and the exact p-values are provided in the Source Data file.
Identifying aberrant activation of IGF1 signaling pathways specifically in hARtg+ atypical basal cells within PIN lesions implicates a regulatory mechanism for hARtg in initiating prostatic oncogenesis through the activation of IGF1R axes. Given the AR functioning as a ligand-dependent transcriptional factor, we then performed chromatin immunoprecipitation sequencing (ChIPseq) analysis to examine if hARtg regulates IGF1R transcription. A specific enrichment of transgenic AR was identified within the Igf1r promoter region on mouse chromosome 7 in the human AR antibody immunoprecipitated samples prepared from PIN tissues of R26mTmG/hAR:Osr1Cre/+ mice (Supplementary Fig. 5; Supplementary Data 2). ChIP-quantitative polymerase chain reaction (ChIP-qPCR) analyses further showed the specific occupancy of the transgenic AR in both the promoter and enhancer regions of the Igf1r gene with the androgen response element sequences24, but not in the Untr4 locus, used as a negative control, in the same immunoprecipitated PIN samples (Fig. 3i, j). Using prostatic organoids derived from R26mTmG/hAR:Osr1Cre/+ mice, we also identified the higher expression of Igf1r transcripts in samples cultured with dihydrotestosterone (DHT) than those without DHT or treated with a combination of DHT and Enzalutamide (Enz), an antiandrogen (Supplementary Fig. 5c). Taken together, these lines of experimental evidence demonstrate a direct role of hARtg in regulating IGF1R expression, implicating an underlying mechanism by which aberrant AR induces PIN development via activating IGF1R signaling.
Activating Wnt signaling in hARtg positive prostatic tumor cells
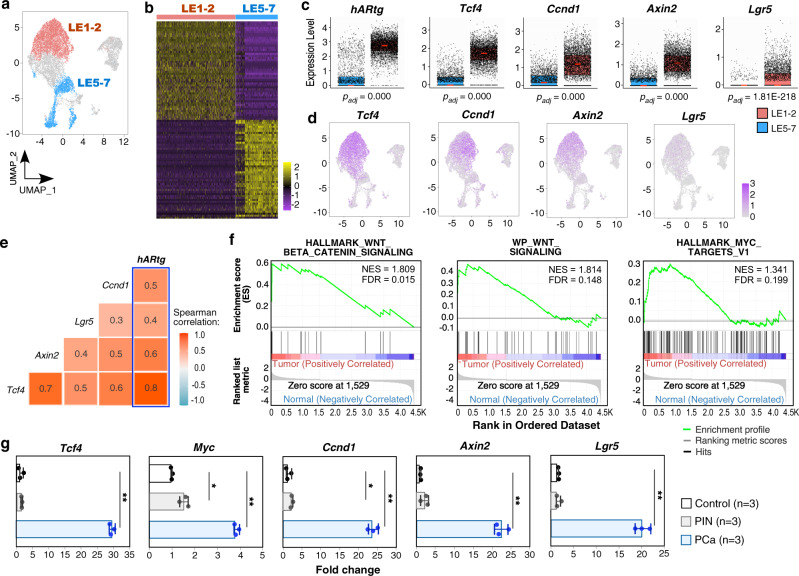
Our finding of hARtg to activate IGF1R axes in atypical basal cells explores a mechanism for AR's oncogenic role in prostate oncogenesis. Prostatic basal epithelial cells have been shown to possess the ability to initiate oncogenic transformation and to further transdifferentiate to luminal tumor cells in the presence of androgens25–27. To assess the underlying mechanisms for hARtg+ atypical cells to progress to prostate tumors, we analyzed the transcriptomic changes between LE1-2 tumor cell clusters possessing enriched hARtg+ tumor cells and normal LE5-7 clusters containing hARtg- cells (Fig. 4a, b). 1834 up-regulated and 3156 down-regulated DEGs were identified (Fig. 4b; Supplementary Data 3). A significant increase in the expression of Wnt/β-catenin target genes, such as Tcf4, Ccnd1, Axin2 and Lgr5, revealed in LE1-2 in comparison to LE5-7 clusters using either box plots (Fig. 4c) or UMAP expression plots (Fig. 4d). Strong correlations between hARtg and these Wnt downstream target genes were further confirmed in LE1-2 and LE5-7 cell clusters (Fig. 4e). GSEA using pre-ranked gene lists of LE1-2 versus LE5-7 further showed a significant enrichment in both Wnt and Myc target signaling pathways (Fig. 4f). Increased expression of Wnt/β-catenin downstream target genes, including Tcf4, c-Myc, Ccnd1, Axin2, and Lgr5, was also identified in RNA samples isolated from pathologically confirmed PCa samples in comparison to PIN and prostate tissues from age and sex-matched wild type (WT) mice using qRT-PCR (Fig. 4g). These data consistently demonstrate the activation of Wnt/β-catenin signaling pathways in hARtg+ prostate tumor cells of R26mTmG/hAR:Osr1Cre/+ mice.
Fig. 4. Activating Wnt signaling pathways in hARtg positive prostatic tumor cells.
a UMAP plot highlighting LE1-2 and LE5-7 clusters identified as hARtg + tumor cell clusters and hARtg-normal cell clusters, respectively. b Heatmap showing top 50 differentially expressed genes between LE1-2 (n = 5122) and LE5-7 (n = 2743) cell clusters. Yellow and purple indicate high and low expression, respectively. c Box plots representing scaled expression data for hARtg and Wnt downstream target genes applied to LE1-2 and LE5-7 cells. Pink lines mark the median; top and bottom lines of boxes indicate the boundaries of the first and third quartiles, respectively; the top and bottom whiskers show the maximum and minimum values, respectively, excluding outliers. P-values were computed using Wilcoxon rank-sum test (two-sided) and adjusted using Benjamini-Hochberg correction. d Gene expression UMAP plots displaying the expression patterns for the indicated Wnt downstream genes. Color intensity indicates the scaled expression level in each cell. e Heatmap of pairwise Spearman correlation between the indicated gene expression in LE1-2 and LE5-7 cells. Colors reflect the level of correlation (positive correlation in red and negative correlation in blue); numbers show the correlation coefficient. The blue box highlights the correlation between hARtg and other Wnt downstream genes. f GSEA enrichment plots highlighting the positive enrichment of Wnt signaling pathway with multiple gene sets comparing LE1-2 to LE5-7 cells. g RT-qPCR analysis of the indicated genes shown as fold change in normal prostate tissues from R26mTmG/+:Osr1Cre/+ control mice and PIN or PCa tissues from R26mTmG/hAR:Osr1Cre/+ mice. Data are represented as mean ± SD of three biological replicates. Two-sided t-test for PIN or PCa versus control, *p < 0.05, **p < 0.01. See also Supplementary Data 3. Source data and the exact p-values are provided in the Source Data file.
Activation of transgenic AR expression in Osr1-lineage cells upregulates Wnt signaling pathways in prostate tumor tissues
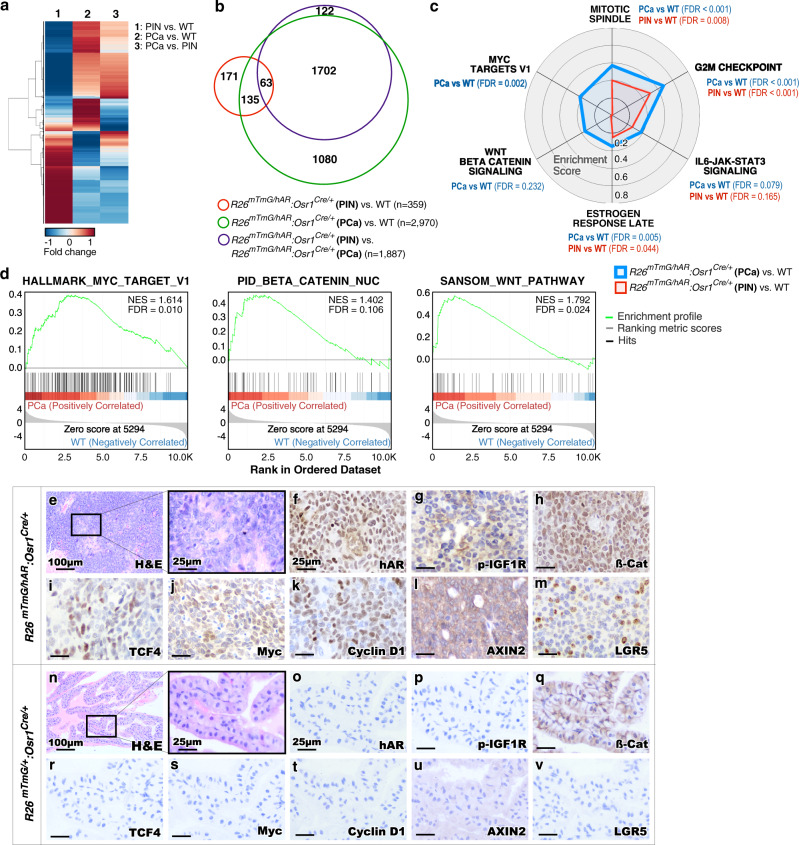
To gain more in-depth insight into transcriptomic changes in hARtg regulated prostatic oncogenesis, we performed bulk RNA-sequencing (RNA-seq) analyses using RNA samples isolated from microscopically confirmed PIN or PCa tissues with more than 80% atypical or tumor cells, respectively, from R26mTmG/hAR:Osr1Cre/+ mice, as well as prostate tissues from sex and age-matched WT mice. Using a median fold difference test (see the Methods), we identified 359, 2970 and 1887 DEGs with adjusted p value < 0.05 and fold change ≥ 2 by comparing RNA-seq samples between PIN or PCa versus WT controls, and PCa versus PIN samples, respectively (Fig. 5a; Supplementary Data 4–6). Crossing analyses from the above datasets identified common DEGs between each of the two or three groups (Fig. 5b). A group of common DEGs (n = 198) overlapping between PIN or PCa versus WT samples was identified (Fig. 5b; Supplementary Data 4–6). GSEA using pre-ranked DEG lists from PIN or PCa versus WT samples revealed four common significantly enriched signaling pathways, including the mitotic spindle, G2/M checkpoint, IL6-JAK-STAT3 signaling, and estrogen response pathways (red line, Fig. 5c). Much higher enrichment scores (ES) appeared in the samples of PCa versus WT than those of PIN versus WT, suggesting the promotional roles of these signaling pathways during disease progression. Interestingly, the significant enrichment of Wnt/β-catenin and Myc targets V1 signaling pathways only appeared in the gene list of PCa versus WT samples (Fig. 5c). GSEA with a pre-ranked DEG list by comparing PCa to WT samples further showed the significant enrichment (FDR < 0.25) in both Wnt/β-catenin signaling and Myc targets (Fig. 5d). IHC analyses using adjacent PCa tissue sections revealed positive nuclear staining for transgenic AR and β-catenin, as well as positive staining for phosphorylated IGF1R and for β-catenin downstream targets, including TCF4, c-Myc, Cyclin D1, AXIN2, and Lgr5 (Fig. 5f–m). In contrast, no specific staining was detected in normal prostate tissues isolated from age- and sex-matched R26mTmG/+:Osr1Cre/+ controls (Fig. 5o–v). These data consistently demonstrate aberrant activation of Wnt/β-catenin signaling pathways in prostate tumor cells of R26mTmG/hAR:Osr1Cre/+ mice, implicating the regulatory role of hARtg in activating Wnt/β-catenin to promote prostate tumor development and growth.
Fig. 5. Activation of transgenic AR expression in Osr1-expressing cells upregulates Wnt signaling pathways in prostate tumor tissues.
a Heatmap showing the expression patterns of differentially expressed genes from the three comparisons of bulk RNAseq data from normal prostate tissues from R26mTmG/+:Osr1Cre/+ wild type (WT) mice and PIN or PCa tissues from R26mTmG/hAR:Osr1Cre/+ mice as indicated in the figure. Red and blue colors indicate up- and down-regulation, respectively. b Venn diagram depicting the number of differentially expressed genes from the different comparisons as labeled. c Radar chart of the enrichment score displaying differential enrichment of R26mTmG/hAR:Osr1Cre/+ (PIN or PCa) in comparison with wild type (WT) control samples. Enrichment score and false discovery rate (FDR) were calculated using a hypergeometric test from GSEA with DEGs (|Log2 fold-change| >1 and adjusted p value < 0.05). P values were adjusted for multiple testing with Benjamini-Hochberg correction. Red and blue lines indicate enriched hallmark gene sets in PIN and PCa tissues, respectively. d GSEA enrichment plots of pre-ranked gene list from differentially expressed genes comparing PCa to wild type samples, highlighting positive enrichment of Wnt signaling pathway with multiple gene sets. e-v Representative images of H&E and IHC staining using the indicated antibodies on adjacent PCa tissue sections from R26mTmG/hAR:Osr1Cre/+ mice and wild type mice. Scale bars, 100 µm; 25 µm. Images representative of consistent results from three independent experiments are shown. See also Supplementary Data 4–6.
Activation of human AR transgene expression initiates PIN formation and promotes tumor development through aberrant elevation of IGF1 and Wnt/β-catenin signaling pathways
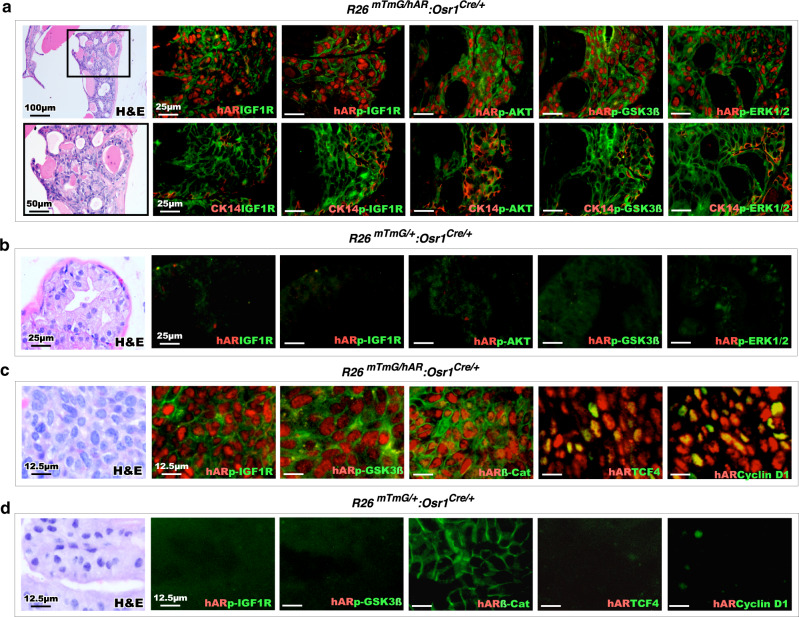
It has been shown that aberrant activation of IGF1 signaling can either directly or via activating AKT and GSK3β axes stabilize cellular β-catenin to augment its activity for prostate tumor growth28–31. We then assessed the effect of IGF1 signaling in activating Wnt/β-catenin axes in hARtg + atypical basal cells. Co-IF analyses identified positive staining for IGF1R or phosphorylated IGF1R overlaid with transgenic AR staining in atypical HGPIN cells (Fig. 6a). Positive staining for phosphorylated forms of AKT, GSK3β, and ERK1/2 were also detected and overlaid with transgenic AR in those HGPIN cells on adjacent tissue sections (Fig. 6a). Positive staining for IGF1R, phosphorylated IGF1R, AKT, GSK3β, and ERK1/2 also overlaid with CK14 staining, demonstrating the basal cell properties of those double positive atypical cells (Fig. 6a). In contrast, there is no or very weak staining with transgenic AR, IGF1R, and phosphorylated IGF1R, AKT, GSK3β, and ERK1/2 in prostate tissues of age- and sex-matched R26mTmG/+:Osr1Cre/+ controls (Fig. 6b). These data suggest up-regulated IGF1R expression and activation of IGF1R and its downstream target, AKT, leading to the phosphorylation of GSK3β, and ERK1/2 in atypical basal cells of HGPIN lesions. Using co-IF approaches, we further assessed the activation of IGF1R and Wnt/β-catenin axes in hARtg+ tumor cells. Co-staining for phosphorylated IGF1R and GSK3β, as well as nuclear β-catenin and its downstream targets, TCF4 and Cyclin D1, with transgenic AR staining appeared specifically in prostate tumor cells (Fig. 6c) in comparison to normal prostatic epithelial cells of WT mouse prostate tissues (Fig. 6d), suggesting the regulatory loop of hARtg expression in elevating IGF1 signaling and activating Wnt/β-catenin axis during PIN and tumor development in R26mTmG/hAR:Osr1Cre/+ mice. These lines of scientific evidence are consistent with previous in vitro studies and provide more relevant data to demonstrate the co-regulation of androgen, IGF1, and Wnt/β-catenin axes in prostate tumor development and growth28,29,31.
Fig. 6. Activation of hAR transgene expression initiates PIN formation and tumor development through aberrant elevation of IGF1 and Wnt/ß-catenin signaling pathways.
Representative images of H&E and co-IF staining for hAR or CK14, a basal cell marker, and IGF1 signaling downstream mediators, including IGF1R, phosphorylated-IGF1R (p-IGF1R), p-AKT, p-GSK3ß, and p-ERK1/2 in PIN tissue sections from R26mTmG/hAR:Osr1Cre/+ mice at 6 months age (a) and normal prostate tissue sections from age-matched R26mTmG/+:Osr1Cre/+ wild type mice (b). Scale bars, 100 µm; 50 µm; 25 µm. Representative images of H&E and co-IF staining for hAR and IGF1 downstream (p-IGF1R, p- GSK3ß) or Wnt downstream (ß-catenin, TCF4, and Cyclin D1) in PCa tissue sections from R26mTmG/hAR:Osr1Cre/+ mice at 12-month age (c) and normal prostate tissue sections from age-matched R26mTmG/+:Osr1Cre/+ wild type mice (d). Scale bars, 12.5 µm. Representative images with consistent results from three independent experiments are shown.
Aberrant transgenic AR induces Wnt/β-catenin signaling activation to promote prostate cancer development
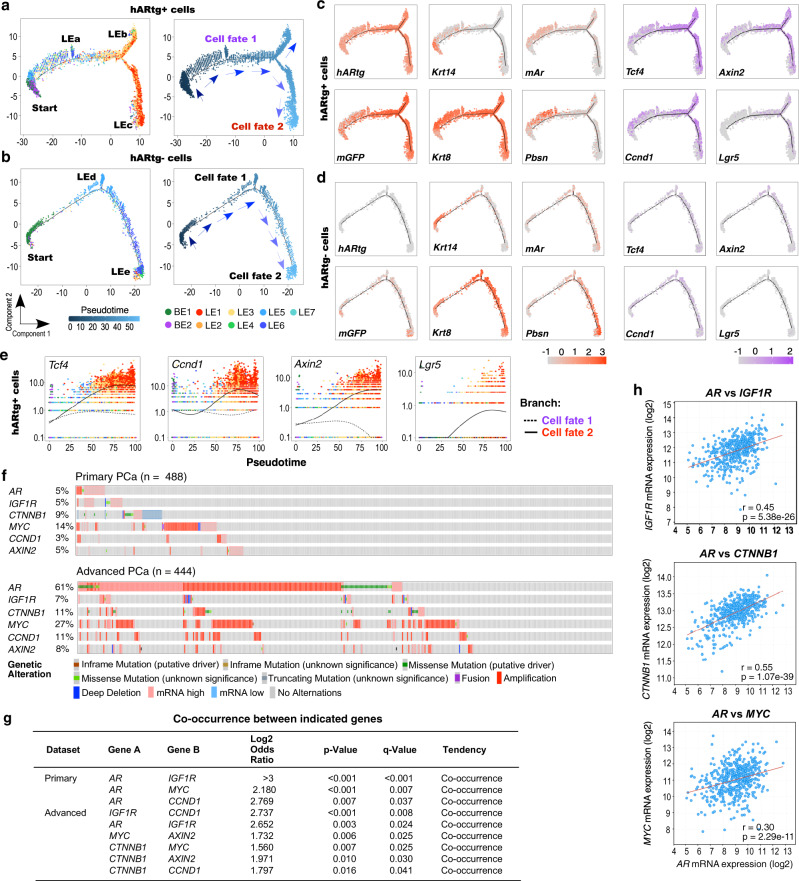
To gain dynamic and deep insight into the oncogenic role of hARtg in prostate tumorigenesis, we conducted single-cell trajectory analyses to unveil transcriptomic changes that were specifically induced by hARtg expression and governed cell fate decisions through PIN initiation and progression to prostatic adenocarcinomas32. Using Monocle232, we observed that the pseudotime trajectory plots of hARtg + epithelial cells from merged PIN and PCa samples displayed a starting point composed mainly of BE2, a PIN cell cluster, that differentiated to the cell trend with luminal cell properties, and then further progressed to two main luminal cell branches mainly possessing LE1-2 tumor cells (Fig. 7a). In contrast, hARtg- epithelial cells showed a different trajectory fate, which started with BE1 cells and differentiated into a single luminal cell branch containing normal LE5-7 cells (Fig. 7b). The cellular properties of different cell branches were further assessed using trajectory expression plots with different cellular markers. The expression of hARtg in mGFP+ Osr1-lineage cells was only observed in hARtg+ cell samples (Fig. 7c). Whereas the expression of Krt14 mainly appeared in the start of the branches, composing BE2 or BE1 cells, in hARtg+ or hARtg− cell plots, respectively, Krt8 expression showed at later cell branches in pseudotime in both hARtg+ and hARtg−samples, representing their luminal cell properties (Fig. 7c, d). Intense expression of endogenous Ar and Pbsn appeared at the later trend of the luminal cell branch in hARtg- cells (Fig. 7d). In contrast, in hARtg+ cell plots, both endogenous Ar and Pbsn expression only appeared earlier in pseudotime and decreased as cells progressed toward the two luminal tumor cell branches, indicating their undifferentiated and transformed cellular properties (Fig. 7c). Importantly, the expression of Wnt/β-catenin target genes, including Tcf4, Ccnd1, Axin2, and Lgr5, was observed in later luminal cell branches of hARtg+ cells through cell trajectory plots in comparison to those of hARtg- cells (Fig. 7c vs d), implicating the activation of Wnt/β-catenin signaling during the course of tumor development. Using pseudotemporal kinetic analysis, we further demonstrated increased expression of Tcf4, Ccnd1, Axin2, and Lgr5 in the later trend of tumor cells (fate 2) in comparison to the middle trend of cells (fate 1) in hARtg+ cell trajectory plots (Fig. 7e), showing the activation of Wnt/β-catenin signaling correlating with the course of tumor development. Taken together, the above analyses provide a dynamic and high-resolution image for transgenic AR induced Wnt signaling activation through prostate cancer development.
Fig. 7. Aberrant IGF1 and Wnt/β-catenin signaling pathways regulate prostate cancer development.
Pseudotime trajectory plots displaying a predicted directional path of hARtg + (a) or hARtg- epithelial cells (b) colored by cluster identity (left) and by pseudotime (right). Expression of the indicated genes projected onto the pseudotime trajectory in hARtg + (c) and hARtg- epithelial cells (d). Red and purple color intensities indicate the scaled expression levels of cellular markers, including hARtg, mGFP, Krt14, Krt8, endogenous Ar (mAr), Pbsn, and Wnt downstream target genes, including Tcf4, Axin2, Ccnd1, and Lgr5, respectively, in each cell. e Linear pseudotime expression plots for indicated Wnt downstream target genes in hARtg + epithelial cells. Lines on each plot correspond to the path of tumor progression moving from the start point (left) to indicated branch tips (right) as in a (right). f Oncoprint outlining genetic alterations and expression of the indicated genes in 488 human primary prostate cancer samples and 444 metastatic castration-resistant prostate cancer samples. Datasets of Prostate Adenocarcinoma (TCGA, PanCancer Atlas) and Metastatic Prostate Adenocarcinoma (SU2C/PCF Dream Team) for primary and advanced prostate cancer, respectively, were extracted from cBioPortal. Colors show genetic alteration as indicated in legend. See also Methods section. g Mutual exclusivity panel analysis depicting the co-occurrence of alterations of the indicated genes in human primary PCa and metastatic castration-resistant prostate cancer. P values were computed using Fisher’s exact test (one-sided) and adjusted using Benjamini-Hochberg correction. h Scatter plots showing the Spearman correlation of co-expression between AR and IGF1R, CTNNB1, or MYC. r = spearman’s correlation coefficient, p = p value. Two-sided t-test. See also Supplementary Fig. 8.
Correlative activation of androgen, IGF1, and β-catenin signaling occurs in human prostate cancer samples
Next, we assessed aberrant AR, IGF1, and Wnt/β-catenin signaling pathways in both human primary PCa samples from the TCGA Pan-Cancer Atlas and advanced PCa samples in cBioPortal33,34. Aberrant activation of AR signaling through AR gene amplification or increasing its expression was detected in 5% of 488 primary PCa samples. Within these samples, about 42% and 55% of them also co-existed with aberrant alterations in IGF1R and Wnt/β-catenin downstream target genes, MYC and CCND1, respectively (Fig. 7f, top panel). Intriguingly, similar alterations in AR amplification and increasing expression were identified in 61% of 444 total advanced PCa samples. Increased co-occurrence of altered IGF1R and gain-of-function alterations and mutations in Wnt/β-catenin pathways also appeared (Fig. 7f, bottom panel). Odds ratio testing showed significant co-occurrences of alterations of AR and IGF1R, MYC, or CCND1 genes and their transcripts in both primary and advanced PCa samples (Fig. 7g). Specifically, significantly positive correlations were observed between the transcripts of AR with IGF1R, CTNNB1, or MYC in primary PCa samples (Fig. 7h). Enrichment of AR on the IGF1R gene locus was also observed in AR ChIP-seq datasets of human PCa samples in comparison to controls with normal prostate tissues (Supplementary Fig. 6), further supporting our mouse ChIP-seq analyses (Supplementary Fig. 5). These lines of scientific evidence provide the importance and clinical relevance of aberrant AR activation in altering IGF1 and Wnt/β-catenin signaling pathways in human prostate tumorigenesis.
Aberrant activation of Wnt/β-catenin signaling pathways enhances AR-mediated prostate tumor growth
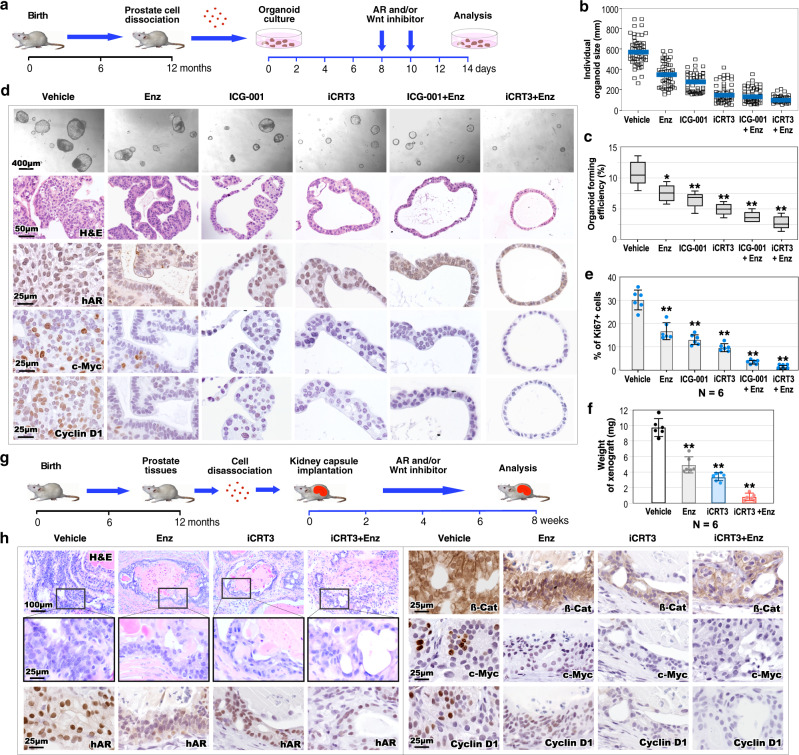
Multiple lines of evidence have shown a promotional role of Wnt/β-catenin in PCa growth and progression35,36. Specifically, an interaction between the AR and β-catenin has been identified in PCa cells, directly augmenting tumor cell growth37–39. Using organoid cultures derived from prostatic tumor cells of R26mTmG/hAR:Osr1Cre/+ mice, we directly assessed the role of Wnt/β-catenin in hARtg+ tumor cell growth. The developed prostatic organoids were treated with the anti-androgen, enzalutamide (Enz), and Wnt inhibitors, ICG-00140 and iCRT341 alone or in combination (Fig. 8a). It has been shown that iCRT3 can disrupt the interaction between AR and/β-catenin and inhibit AR-mediated transcription and cell growth in PCa cells41. Measuring average sizes of individual organoids and the organoid forming efficiency showed significant reduction in samples treated with Enz, ICG-001, and iCRT3 alone or in combination in comparison with vehicle-treated controls (Fig. 8b, c). Accordingly, more and larger organoids developed in vehicle-treated samples than in those treated with different inhibitors in the brightfield images (Fig. 8d). Specifically, far fewer and smaller organoids revealed in samples treated with Enz combined with Wnt inhibitors, ICG-001 or iCRT3. Histological analyses recapitulated similar prostatic adenocarcinoma lesions in vehicle-treated organoids as observed in tumor tissues of R26mTmG/hAR:Osr1Cre/+ mice (Fig. 8d). Abnormal glandular structures resembling PIN lesions represented in samples treated with Enz, or Wnt inhibitors alone. Appearances of minor pathological changes to normal glandular structures were observed in samples treated with both Enz and Wnt inhibitors. IHC showed CK5 and CK8 staining in organoid cells, indicting their epithelial properties (Supplementary Fig. 7a1–b6). Positive nuclear staining for transgenic AR appeared in organoids treated with vehicle, ICG-001, and iCRT, whereas samples treated with Enz alone and in combination with Wnt inhibitors showed both nuclear and cytoplasmic staining for transgenic AR (Fig. 8d). Positive staining for c-Myc and Cyclin D1, the downstream targets of β-catenin, appeared in vehicle-treated samples and slightly in Enz-treated samples, but not in those treated with Wnt inhibitors alone or in combination with Enz (Fig. 8d). Measuring Ki67 positive cells in the organoid samples revealed the inhibitory effects with Enz and Wnt inhibitors. Among them, the most robust inhibition appeared in samples treated with Enz and iCRT3 (Fig. 8e). Taken together, these data demonstrate the inhibitory effects of both antiandrogen and Wnt inhibitors on the growth of hARtg+ tumor organoids.
Fig. 8. Co-inhibition of androgen and Wnt/β-catenin signaling pathways in prostate tumor growth.
a Schematic representation of the experimental design for the ex vivo organoid culture performed. Organoids derived from PCa cells of R26mTmG/hAR:Osr1Cre/+ mice were treated with vehicle, antiandrogen (10 µM Enzalutamide; Enz), Wnt inhibitor (10 µM ICG-001 or 10 µM iCRT3), or combination of Enz and ICG-001 or iCRT3 two times for 6 days. See also Methods section. b Quantification of individual organoid size. Organoids per treatment group (n = 50) examined over three independent experiments. The center blue bar indicates the median value in each group. c Quantification of organoid forming efficiency showing the percentage of organoids above 50 μm diameter per total cells seeded at day 0 in a well. The center line represents the median value; the box borders represent the lower and upper quartiles (25% and 75% percentiles, respectively); the ends of the bottom and top whiskers represent the minimum and maximum values, respectively. d Representative images of brightfield, H&E and IHC staining for the indicated antibodies of the organoids with the indicated treatments. Scale bars, 400 µm; 50 µm; 25 µm. e Quantification of Ki67 positive cells per total cells in the organoids with the indicated treatments. f Weights of xenografts (n = 6) from groups treated as indicated. Data are represented as mean ± SD (n = 6 replicates per data point). Student’s t-test, **p < 0.01. g Schematic representation of the experimental design using in vivo kidney capsule transplantation. See also Methods section. h Histological and IHC analysis of graft tissues with the indicated treatment. Representative images of H&E and IHC images for indicated antibodies on adjacent sections from graft tissues with the indicated treatments. Scale bars, 100 µm; 25 µm. In c, e, and f data are represented as mean ± SD of six independent samples over three biological replicates. Two-sided t-test, *p < 0.05, **p < 0.01. Representative images with consistent results from three independent experiments are shown. Source data and the exact p-values are provided in the Source Data file.
Using in vivo tissue grafting assays, we further examined co-inhibition of AR and Wnt signaling in hARtg+ tumor growth. Specifically, we tested iCRT341, a Wnt inhibitor that also showed inhibition of AR signaling in PCa cells, alone or with Enz. Prostate tumor cells isolated from R26mTmG/hAR:Osr1Cre/+ mice were implanted under the kidney capsule of SCID mice (Fig. 8g). Four weeks after implantation, SCID mice were administered Enz, iCRT3, or both, as well as vehicle, and analyzed four weeks post treatment. Although grafts treated with Enz or iCRT3 alone appeared significantly smaller in size and less in weight than vehicle-treated group, samples treated with both Enz and iCRT3 showed the lowest weights and smallest size among the groups (Fig. 8f). Histologically, vehicle-treated samples retained abnormal pathological characteristics of prostate adenocarcinomas as observed in prostate tumor tissues of R26mTmG/hAR:Osr1Cre/+ mice whereas Enz or iCRT3-treated samples displayed minor pathological changes. Samples treated with both Enz and iCRT3 showed much less pathological changes than other controls (Fig. 8h). Positive staining for CK5 and CK8 was observed in all grafted samples (Supplementary Fig. 7g1–i4). While robust nuclear staining for hAR was observed in vehicle treated samples, much less staining appeared in the other treated samples (Fig. 8h). Positive nuclear staining for β-catenin revealed in the vehicle and Enz-treated grafts. In contrast, only cellular membrane staining of β-catenin appeared in samples treated with iCRT alone and in combination with Enz (Fig. 8h). Positive staining for c-Myc and Cyclin D1, the downstream targets of β-catenin, also only revealed in vehicle treated samples (Fig. 8h). These data reaffirm that the co-inhibition of AR and Wnt signaling pathways represses the growth of hARtg+ tumor cells.
Discussion
Although the promotional role of androgen-signaling in prostate tumorigenesis has been implicated for many decades2,6, the fundamental mechanisms by which the AR induces oncogenic transformation and initiates PIN and prostate tumor development are still unclear. Conditional expression of human AR transgene in mouse prostatic Osr1-lineage cells results in HGPIN and prostatic adenocarcinomas as mice progressed in age10, recapitulating the oncogenic role of the AR as observed in human prostate cancer. Here, we directly evaluated the cellular properties and transcriptomic changes of prostatic Osr1-lineage cells through prostatic embryonic and pubertal development. The expression of Osr1 revealed transiently in UGE tissues at E15.5 and E18.5 days, overlaying with other prostatic stem and progenitor cell markers. A robust increase in mGFP+ cells, the descendants of Osr1-expressing cells, was observed in UGE as well as prepubescent and pubertal prostatic epithelia. Genetically tracing labeled Osr1-expressing cells further showed their ability in expanding prostatic epithelia through embryonic to pubertal stages of prostate development. Identifying the basal epithelial cellular properties of mGFP+ cells in prepubescent prostatic epithelia further evidences Osr1-lineage cells functioning as prostatic progenitors. These observations are consistent with early studies showing the expression of Osr1-driven Cre starting at E11.5 in urogenital tissues that the prostate is derived from42. The expression of OSR1 was further identified in urogenital epithelial cells with the AR in human male embryonic tissues at 18 weeks (Supplementary Fig. 7j), implicating the biological importance and relevance of OSR1-lineage cells in human prostate development. These findings identify the cellular properties of prostatic Osr1-expressing cells and their descendants through prostate early, prepubescent, and pubertal development, elucidating a cell population of prostate epithelial progenitors.
Development of HGPIN and prostate adenocarcinomas in R26mTmG/hAR:Osr1Cre/+ mice but not in R26 mTmG/+:PB Cre/+ counterparts, aligning with the identification of hARtg+ atypical and tumor cells specifically within those HGPIN and prostate adenocarcinomas lesions, demonstrates the direct role of transgenic AR expression in inducing PIN and PCa development10. Data from our scRNA-seq analyses further identified elevated IGF1 and insulin related signaling pathways in hARtg+ basal epithelial cells in R26mTmG/hAR:Osr1Cre/+ mice, suggesting a regulatory mechanism underlying aberrant AR action in stimulating IGF1 signaling to promote PCa initiation. Early studies have shown that individuals with high circulating levels of IGF1 have an increased risk of PCa, particularly advanced disease43,44. Activated IGF1R through a ligand-receptor interaction can promote mitogenic signaling events through increasing cell proliferation and inhibiting apoptosis45. Aberrant IGF1R and insulin receptor expression has been frequently observed in human PCa samples46. Additionally, overexpression of the Igf1 in mouse prostatic basal epithelia induced oncogenic transformation and mouse PIN development21. Moreover, the AR has been shown to directly regulate the transcription of the IGF1R in PCa cells24. Our current findings are consistent with the previous results, demonstrating a critical role of hARtg in elevating aberrant IGF1 axis in Osr1-lineage basal cells to induce prostate oncogenic transformation and PIN formation. Specifically, our ChIPseq and ChIP-qPCR data revealed an enrichment of transgenic AR in the regulatory regions of the mouse Igf1r gene locus in the hARtg+ cells. These lines of evidence implicate a regulatory mechanism underlying aberrant AR activation through IGF1-mediated signaling pathways in PCa development. These findings also suggest an unknown interaction between AR and IGF1 signaling pathways that manipulate PCa metabolism and contribute to the pathogenesis of PCa.
In R26mTmG/hAR:Osr1Cre/+ mice, HGPIN lesions enable to continue to progress to prostatic adenocarcinomas, resembling what occurs in human PCa. This intriguing pathologic characteristic has not been observed in previous AR transgenic mice using the PB promoter7,47,48. In this study, we identified aberrant Wnt/β-catenin signaling activation in hARtg+ prostatic tumor cells, uncovering the regulatory mechanisms underlying AR action in atypical cells to progress to prostatic adenocarcinomas. The activation of aberrant Wnt/β-catenin axis has been shown to directly promote prostate cancer cell proliferation as well as tumor development and progression30,49. β-catenin is a co-regulator of the AR and synergistically promotes prostate tumor cell growth through direct interaction with the AR50. Moreover, IGF1 signaling can enhance the stability of cellular β-catenin and augment the AR-mediated transcription in PCa cells28,29. In this study, we provided multiple lines of scientific evidence demonstrating a regulatory loop through aberrant activation of androgen signaling in stimulating IGF1 and β-catenin activation to promote PCa initiation and development. The above data also suggest that the current AR transgenic model is a biologically significant and clinically relevant tool for the future investigation of AR action in prostate tumorigenesis.
Using single-cell trajectory analyses, we evaluated the effect of hARtg expression on cell fates and differentiation during the course of PIN initiation and progression to prostatic adenocarcinomas. In hARtg+ cell pseudotime trajectory plots, BE2, a PIN cell cluster, appeared at the starting branch, gradually progressing towards cells that possess endogenous Ar and Pbsn expression and are mainly composed of LE3-4 luminal cells, and finally progressed to two luminal tumor cell branches comprising LE1-2 clusters. In contrast, hARtg- cells showed a very different cell trajectory trend, which started with BE1 cell cluster and progressed to the cell trend and branch with normal luminal cell properties. Using Slingshot tools51, we further validated the above results and observed similar trajectory fates of hARtg+ and hARtg- cells as observed in our Monocle2 analyses (Supplementary Fig. 8a, b). These data provide a dynamic image for the different cell fates initiated from hARtg+ or hARtg- cells. Selective up-regulation of Wnt/β-catenin signaling downstream target gene expression, such as Tcf4, Ccnd1, Axin2, and Lgr5, only revealed in hARtg+ luminal tumor cell branches in comparison to both the earlier trend of hARtg+ cells (cell fate 1) in pseudotime and the cell branch of hARtg− cells in trajectory expression plots, demonstrating the activation of Wnt/β-catenin signaling induced by hAR aligning with PCa development and progression. Using newly developed package, PseudotimeDE, we further addressed uncertainty of pseudotime DEG inference, validating the expression of hARtg and Wnt/β-catenin downstream target genes along the established trajectory branches52 (Supplementary Fig. 8c).
In this study, we also identified the significant correlation in co-occurrence of altered AR, IGF1R, and Wnt/β-catenin signaling pathways in both primary and advanced human prostate cancer samples. Notably, these alterations and mutations appeared significantly higher in the advanced tumor samples than in primary tumor samples. To gain direct evidence on the regulatory role of AR action in IGF1R and β-catenin activation, we specifically analyzed the expression of AR, IGF1R, and β-catenin using human primary PCa samples. We observed the significant correlation between the expression of the AR with IGF1R, CTNNB1, or MYC gene. Analyses of human ChIP-seq datasets also showed increased enrichment of AR on the IGF1R gene locus in human PCa samples in comparison to normal prostate tissues. These data are consistent with our observations in R26mTmG/hAR:Osr1Cre/+ mice, and further support the regulatory role of aberrant AR activation in initiating prostatic tumorigenesis and promoting tumor growth and progression through elevating IGF1 and Wnt/β-catenin signaling pathways.
Using both antiandrogens and Wnt inhibitors, we evaluated the effect of co-targeting androgen and Wnt/β-catenin signaling pathways on the growth of hARtg+ prostatic tumors in organoid cultures and in-vivo tissue grafting models. Whereas the antiandrogen, Enz, and Wnt inhibitors, ICG-001 or iCRT3 all showed the inhibitory effect on the growth of organoids derived from hARtg+ tumor cells, iCRT3 revealed more potent effect than Enz or ICG-001. Additionally, iCRT3 in combination with Enz showed the most inhibitory effect among all different treated groups. In tissue grafting assays, the similarly robust inhibition was also observed in prostate tumor grafts treated with iCRT3 and Enz combined. It has been shown that iCRT3, a Wnt inhibitor, can also disrupt the interaction between AR and β-catenin and repress AR-mediated transcription and growth in prostate cancer cells41. These data provide relevant and important evidence for developing future therapeutic strategies to co-target AR and Wnt/β-catenin signaling pathways for treating advanced prostate cancer. They also support this AR transgenic model being a biologically significant and clinically relevant tool for future investigation.
Methods
Ethics statement
All experimental procedures and care of animals in this study were carried out according to the Institutional Animal Care and Use Committee (IACUC) at Beckman Research Institute at City of Hope (California, US) and approved by the IACUC. Euthanasia was performed by CO2 inhalation followed by cervical dislocation.
Mouse generation, mating and genotyping
Rosa26mTmG/+ (R26mTmG/+) reporter53, Osr1-Cre42, and Rosa26hARLoxP/wt, also named R26hAR/+ mice10 were obtained and used in this study, respectively. The PBCre/+ mice were obtained from the NCI mouse repository (strain #: 01XF5)54. R26mTmG/hAR, R26mTmG/+:Osr1Cre/+, or R26mTmG/+:PbCre/+ mice were first produced and then used to generate R26mTmG/+:Osr1Cre/+ and R26mTmG/hAR:Osr1Cre/+ mice. Experimental mice generated in this study were mixed from C57BL/6 and 129S1/SvImJ backgrounds. Mice were genotyped after collecting mouse tail tips by PCR approaches using specific primers (Supplementary Table 2). All mice bearing tumors were closely monitored during the entire course of the study according to the guidelines of IACUC in our institution, and the maximal tumor size and burden in mice was never exceeded in this study.
Isolation of normal, PIN, and PCa cells from mice
Cells were isolated and dissected from the mouse UGS or prostate tissues at the indicated timepoint55. Once trimmed, prostate tissues were cut into small pieces and digested in Gibco™ Dulbecco’s Modified Eagle’s Medium (DMEM)/Ham’s F-12 50/50 Mix (DMEM/F12) (11320033, Fisher Scientific) supplemented with 10% Gibco™ Fetal Bovine Serum, 1 nM dihydrotestosterone (DHT) (A8380, Sigma Aldrich), 10 µM Y-27632 dihydrochloride (M1817, Abmole Bioscience), and 1 mg/ml type II collagenase (17101-015, Life Technologies) at 37 °C for 135 min with rotation at 150 rpm, and then in Gibco™ TrypLE (12605-028, Fisher Scientific) supplemented with 1 nM DHT, 10 µM Y-27632 dihydrochloride and 0.5 U/µl DNase I (D5025, Sigma Aldrich) at 37 °C for 30 min with rotation. Cells were filtered through a 37 µm cell strainer (27215, StemCell technologies) and resuspended in PBS with 0.05% Bovine Serum Albumin. Cell viability and number were detected using a TC Automated Cell Counter (Bio-Rad Laboratories), and only cells at least 80% viability were processed.
Histological, IHC and IF analyses
Mouse tissues were cryo-protected in 30% sucrose in phosphate-buffered saline (PBS) and then embedded in OCT (Tissue–Tek®, Sakura Finetek) or processed in paraffin following fixation in 10% neutral-buffered formalin (American Master Tech Scientific). Membrane-bound Tomato (mT) and membrane-bound green fluorescent protein (mGFP) signal was detected by mTmG assays56. Briefly, OCT tissue blocks were cut to 5 μm and sections were washed with PBS, pH 7.3, and then were mounted with Vectashield Mounting Medium with DAPI (H-1200, Vector Laboratories). 4 μm serial sections from paraffin-embedded tissues were used for histological analyses using Hematoxylin-eosin (H&E) staining, immunohistochemistry (IHC) staining, or immunofluorescence (IF) staining27. Pathological analyses were conducted in accordance with the guidelines suggested by The Mouse Models of Human Cancers Consortium Prostate Pathology Committee in 20137. For IHC staining, tissue slides were boiled in antigen retrieval buffer, 0.01 M citrate buffer (pH 6.0) or Tris/EDTA (pH 9.0). Following rehydration, slides were placed in 0.3% H2O2 in methanol for 15 min, blocked in 5% goat serum for 1 hr, and incubated with primary antibodies in 1% goat serum in PBS overnight at 4 °C. Tissues were then incubated with biotinylated secondary antibodies for 1 hr and with horseradish peroxidase (HRP)-conjugated streptavidin (SA-5004, Vector Laboratories) followed by visualization using DAB kit (SK-4100, Vector Laboratories). Sections were counterstained with hematoxylin and mounted with Permount Mounting Medium (SP15–500, Fisher Scientific). For IF staining, tissues sections underwent heat-induced epitope retrieval, blocked in 5% normal goat serum, and incubated with primary antibodies at 4 °C overnight and with fluorescent-conjugated secondary antibodies, and then mounted with Vectashield Mounting Medium with DAPI (H-1200-10, Vector Laboratories). All antibodies used in this study are detailed in Supplementary Table 3.
Microscope image acquisition
Images of H&E and IHC staining were taken by an Axio Lab A1 microscope using ×10, ×20, and ×40 Zeiss A-Plan objectives and were captured using a Canon EOS 1000D camera and AxioVision software (Carl Zeiss). Images of mTmG signals and IF staining were acquired on a Nikon ECLIPSE E800 epi-fluorescence microscope using ×10, ×20, and ×40 Nikon Plan Fluor objectives using an QImaging RETGA EXi camera and QCapture software (QImaging).
Single-cell RNA-seq analyses
Upon isolation, cells were loaded on the Chromium Controller (10X Genomics) targeting 7000–10,000 cells per line. Single cell RNA-seq libraries were prepared using the Chromium v2 single cell 3’ RNA-seq reagent kit (10X Genomics) according to the manufacturer’s protocol. The purity and size of cDNA were validated by capillary electrophoresis on Agilent 2100 Bioanalyzer using Agilent High Sensitivity DNA Kit (#5067-4626, Agilent Technologies). Libraries were sequenced on a NovaSeq 6000 instrument (Illumina) with a depth of 50K–100 K reads per cell. Processing of raw sequencing data, including generation of FASTQ files and counting of Unique Molecular Identifiers (UMI), was conducted using the 10x Genomics Cell Ranger pipeline (3.1.0). Reads were aligned to the mm10 genome with mGFP and hARtg sequences15 for gene expression count. The Seurat package (3.2.1) in R (3.6.3) was used for the subsequent data analysis following upload of a filtered feature bar coded matrix including 1031, 5899, 8983, 7225, and 11,084 cells from E18.5 male UGS, P14, P35, PIN, and PCa prostate tissues, respectively. For a quality-control step, potential empty droplets and multiplets were filtered out based on dead cells or low-quality cells with a fraction of mitochondrial RNA higher than 10–15% were eliminated (Supplementary Figs. 1 and 4). Following this final filtering, 6332 PIN cells with an average of 3751 genes per cell and 21297 UMI counts per cell, and 9412 PCa cells with an average of 3532 genes per cell and 19,899 UMI counts per cell were conserved for future analyses. Integrating datasets were performed on Seurat using FindIntegrationAncors and IntegrateData functions with twenty dimensions. Normalized and scaled data were clustered by Graph-based clustering approach using Seurat functions including RunPCA with 30 principal components, FindNeighbors with twenty dimensions, and FindClusters with 0.5 resolutions. Data was visualized by a nonlinear dimensionality reduction UMAP technique using the Seurat RunUMAP function.
For analysis of signaling pathways, differential expression analysis was conducted on Seurat using FindMarkers or FindAllMarkers functions. p values were adjusted for multiple testing with Benjamini-Hochberg correction. Genes with |average Log2 fold-change| > 1 and adjusted p value < 0.05 were considered as differentially expressed genes (DEGs). We used canonical pathway analysis in Ingenuity Pathway Analysis (IPA) (v01-20-04) and Gene Set Enrichment Analysis (GSEA) (4.1.0) analysis with hallmark and curated gene sets. Spearman pairwise correlation matrices as the measure of association between hARtg and other genes were analyzed and plotted using the ggcorr function from the GGally package R (https://www.rdocumentation.org/packages/GGally/versions/1.5.0/topics/ggcorr) in R.
For pseudotime generation and trajectory analysis, Seurat objects were converted into CellDataSet format and the DDRTree algorithm32 applied to reduce expression profiles into two dimensions using the Monocle2 package (2.12.0)32 in R. Using BEs as the starting point, Monocle reconstructed differentiation trajectories and defined pseudotime. Branch expression analysis modeling (BEAM) was used to detect genes significantly branching between cell fate 1 and cell fate 2 and to identify genes that change after fate decisions along the hARtg in PCa development. Next, we generated aligned kinetic curves for Wnt target genes, which showed significant difference in the BEAM test, using Monocle’s plot_genes_branched_pseudotime function. Pseudotime trajectories were also identified using the Bioconductor package Slingshot (1.8.0)51 by default, and visualized on a 2-dimensional UMAP embedding. For pseudotime-based differential expression testing, Pseudotime DE package (1.0.0)52 was used with SingleCellExperiment objects, processed with Slingshot51, as input for the pseudotime inference method. After subsampling by default, differentially expressed gene testing was performed using the run PsuedotimeDE function with Negative Binomial Model. The gene trajectories estimated by the fitted Generalized Additive Model were visualized.
RNA isolation and RT-qPCR
Total RNA was extracted from fresh mouse prostate tissues or organoids derived from prostate tissues of R26mTmG/+:Osr1Cre/+ or R26mTmG/hAR:Osr1Cre/+ mice using RNeasy Mini Kit (#74104, Qiagen) and reverse-transcribed using SuperScript IV First-Strand Synthesis System (#18091050, Fisher Scientific) according to the manufacturer’s protocol. RT-qPCR were performed on the 7500 Real-Time PCR system (Fisher Scientific) using Power-SYBR Green PCR Master Mix (4367659, Applied Biosystems) with specific primers (Supplementary Table 3). Relative quantification was normalized to the level of peptidylprolyl isomerase A (Ppia) and was calculated using the comparative CT method57.
ChIP-seq and ChIP-qPCR
ChIP-DNA was obtained through ChIP assay58. Briefly, mouse prostate tissues were minced, cross-linked with 1% formaldehyde for 25 min at room temperature (RT) and quenched with 150 mM glycine for 10 min. Following wash with cold PBS, samples were homogenized in cell lysis buffer (140 mM NaCl, 50 mM Tris-HCl [pH 8.0], 1 mM EDTA, 10% Glycerol, 0.5% NP-40, and 0.25% Triton X-100). The chromatin was resuspended in nuclear lysis buffer (0.2% SDS, 10 mM Tris-HCl [pH 8.0], 1 mM EDTA, and 0.5 mM EGTA) and fragmented to an average size of 200–500 bp with a Sonic Dismembrator Model 100 (Fisher Scientific) at 4 °C. After centrifugation, the cell lysate was diluted with ChIP dilution buffer (0.01% SDS, 167 mM NaCl, 16.7 mM Tris-HCl [pH 8.1], 1.1% Triton X-100, and 1.2 mM EDTA), pre-cleared using Dynabead Protein G (10003D, Invitrogen) and then was subjected to immunoprecipitation with Dynabead Protein G conjugated with AR antibody (sc-7305X, Santa Cruz Biotechnology) with rotation for 4 hr at 4 °C. Beads were washed sequentially with washing buffer 1 (0.1% SDS, 150 mM NaCl, 20 mM Tris-HCl [pH 8.0], 1% Triton X-100, and 2 mM EDTA), washing buffer 2 (0.1% SDS, 500 mM NaCl, 20 mM Tris-HCl [pH 8.0], 1% Triton X-100, and 2 mM EDTA), washing buffer 3 (250 mM LiCl, 10 mM Tris-HCl [pH 8.0], 1% NP-40, 1% deoxycholate, and 1 mM EDTA), TE buffer (50 mM Tris-HCl [pH 8.0] and 10 mM EDTA). DNA was eluted with elution buffer (1% SDS, 50 mM Tris-HCl [pH 8.0] and 10 mM EDTA), incubated with 0.2 M NaCl and 1 mg/mL Protease K at 65 °C for 6 hr, purified with phenol/chloroform/isoamylalcohol (Invitrogen) and was precipitated with glycogen and cold EtOH. ChIP DNA samples were subjected to amplification using MicroPlex Library Preparation kit (AB-004-0012, Diagenode) on a StepOnePlus Real-Time PCR System (Applied Biosystems). The libraries were quantified by Qubit dsDNA HS Assay Kit and validated with Agilent Bioanlyzer DNA high Sensitivity Kit. Following library generation, DNA was purified twice with Agencourt AMPure XP beads (A63880, Beckman Coulter) and sequenced on Illumina Hiseq 2500 Sequencer. For ChIP-seq data analysis, reads were mapped to mouse genome assembly mm9 using Novoalign v3.02.07. MACS v2.059 was used to call Peaks using the corresponding negative control from R26mTmG/+:Osr1Cre/+ and to generate bed graph files for visualization of peaks. Gene annotations of mm9 genome were downloaded from the RefSeq database. Binding profiles were visualized using Integrated Genome Viewer (IGV v.6.3)60. ChIP-qPCR was performed with specific primers (Supplementary Table 2) using Power-SYBR Green PCR Master Mix (Applied Biosystems) on the 7500 Real-Time PCR Detection System (Fisher Scientific).
RNAseq and data analysis
RNA integrity and quality were evaluated using Agilent RNA 6000 Nano Kit (#5067-1511, Agilent Technologies). cDNA libraries were prepared using Kapa RNA HyperPrep Kit with RiboErase (KR1351, Kapa Biosystems) followed by sequencing on Illumina Hiseq 2500 at the City of Hope Integrative Genomics Core. Adapter sequences and other redundant sequences were removed from raw paired-end reads using cutadapt (v1.9.1)61. The filtered reads were aligned to the mm9 mouse reference genome using HISAT2 (2.1.0)62. Read counts per gene were detected using HTSeq (v0.6.1)63 and were further normalized using the trimmed mean of M-value method64 or the upper-quartile method65 in edgeR (v2.26.7)66 to obtain counts per million-mapped reads (CPM) and transcripts per million (TPM). Only genes with a CPM > 1 in at least two samples were retained for differential analysis. The fold change, p-value and FDR were calculated using quasi-likelihood (QL) F-test in edgeR settings with default using normalized expression values, TPM. The p values were adjusted for multiple testing with Benjamini-Hochberg correction. DEGs were defined as those having a |Log2 fold-change| > 1 and adjusted p-value < 0.05 and were preranked based on the Log2 fold-change and adjusted p-value. Clipper tools was also used with logCPM as input at the target FDR threshold q = 10%, for controlling false positive data67. GSEA (4.1.0) using preranked gene lists were performed for annotation, integration, and visualization of database in order to identify network and biological functions of DEGs68. Concordant results between two normalization methods were considered significant and results from the trimmed mean of M-value method normalization were presented as representative data.
Human dataset analysis
An integrative analysis of AR with IGF1/Wnt-related genes and clinical characteristics was conducted using cBio Cancer Genomics Portal (https://cbioportal.org), which is an open-access resource for the interactive analysis and visualization of multidimensional cancer genomics datasets34. The gene alteration frequency, co-occurrence, and co-expression were analyzed using prostate cancer datasets from Prostate Adenocarcinoma (TCGA, PanCancer Atlas; 488 samples) (https://www.cell.com/pb-assets/consortium/pancanceratlas/pancani3/index.html) for primay PCa and Metastatic Prostate Adenocarcinoma (SU2C/PCF Dream Team, 444 samples) for advanced PCa33. Co-expression analysis was performed using a log-rank test to identify the significance of the Spearman’s correlation coefficient between the mRNA expression z-scores (RNASeq V2 RSEM). We obtained AR ChIPseq datasets from both human primary PCa tissues, including “GSE130408”, “GSE114737”, and “GSE56288”, and normal prostate tissues (GSE130408) that were extracted from the Gene Expression Omnibus (GEO)69. Those data were visualized with ENCODE70 and RefSeq tracks on UCSC genome browser71. Slides from paraffin-embedded tissues of human UGS at 18 weeks were used for IHC analyses72.
Organoid culture and kidney capsule implantation
Prostatic tumor tissue was isolated from R26mTmG/hAR:Osr1Cre/+ mice at 12 months of age and dissociated into single cells by digestion in DMEM/F12/FBS/Collagenase for 2 hr and then TrypLE for 30 min at 37 °C. Digested cells were passed through 37 µm cell strainers and resuspended in 1:1 (v/v) PBS:Matrigel (#356231, BD Biosciences). For organoid culture, approximately 2000 cells per well were seeded in 24 well-plate and cultured in DMEM/F12 containing 0.25 μM A83-01 (R&D Systems), 1× B27 (Life Technologies), 1 nM DHT, 10 ng/mL EGF (PeproTech), 100 ng/mL N-acetylcysteine (PeproTech), 100 ng/mL Noggin (PeproTech), 2.5 ng/mL R-spondin1 (R&D Systems) and 100 μM Y-27632, as reported in the previous study55 but reduced concentration for EGF and R-spondin1. Eight days after culture, organoids were treated with vehicle, dimethyl sulfoxide (DMSO), antiandrogen, 10 µM Enzalutamide (Enz), Wnt inhibitors, 10 µM ICG-001 (HY-14428, MedChemExpress), 10 µM iCRT3 (HY-103705, MedChemExpress), or combination of Enz and ICG-001 or iCRT3 two times for 6 days and were fixed in 10% neutral-buffered formalin. Fixed cells were subjected to Histogel (HS-4000-012, Fisher Scientific) and paraffin embedding for histological analysis. Individual organoid size was quantified with the Image J (NIH) using at least 100 organoids per group. Organoid forming efficiency (%) was determined by quantifying the percentage of organoid structure above 50 μm diameter per total cells seeded at day 0 in a well. Quantification of the percentage of Ki67+ cells per total cells were performed from five different areas in each sample. All experiments were replicated with two different mice in triplicate wells.
For in vivo kidney capsule implantation, approximately 1 × 105 PCa cells were transplanted under the renal capsule of 6 to 8-week-old male NOD/SCID mice. 4 weeks after implantation, mice were randomized into vehicle control and treatment groups and were administered with vehicle (10% DMSO in corn oil), oral Enz at 20 mg/kg body weight, intraperitoneal iCRT3 at 30 mg/kg body weight41,73,74, or combination of Enz and iCRT3. The compound was administered three times per week for 28 days. Xenografts were excised, weighed and fixed for histological analysis.
Quantification and statistical analysis
Data are presented as the mean values ± SD for the indicated number of independently performed experiments. All data are representative of the results of at least three independent experiments. The significance of the differences between data (*p < 0.05, **p < 0.01) was measured using 2-tailed Student’s t-test. Adjusted p values were corrected for multiple testing using the Benjamini–Hochberg’s procedure. Differentially expressed gene (DEG) lists were determined using a Wilcoxon Rank Sum test, with genes showing adjusted p-value < 0.05 defined to be significant. Spearman’s correlation coefficient > 0.3 and p value < 0.05 was considered to indicate a statistical significance. Enrichment scores (ES) for each gene set in the ranked list of genes were calculated by a running-sum statistic using GSEA68. Nominal p values of ES were estimated using an empirical phenotype-based permutation test and corrected for multiple hypothesis testing using FDR. As recommended by the GSEA User Guide, pathways with FDR < 0.25 were considered significant in exploratory GSEA pathway analysis.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Supplementary information
Description of Additional Supplementary Files
Acknowledgements
This work was supported by NIH grants R01CA070297, R01CA166894, R01DK104941, and R01CA233664. The authors wish to thank Dr. Gerald Cunha and Dr. Laurence Baskin for their help of our IHC experiments.
Source data
Author contributions
Z.S. and W.K.K. conceived the project and designed the experiments. W.K.K., J.M., D.H.L., C.H.N., A.J.B, and Z.S. generated mouse colonies and performed genotyping and tissue collection experiments. W.K.K., A.W.O., J.M., D.H.L., V.L., A.H., J.A., and Z.S. performed related mouse works and collected RNA, DNA and single cell samples for the analyses. J.W., S.Y., W.K.K., and A.W.O. performed sequence experiments and analyzed sequencing data. W.K.K., V.L., D.H.L., A.H., and R.C. conducted ex vivo and in vivo experiments and performed staining and analyses. All authors analyzed and confirmed data. W.K.K, A.W.O., and Z.S. wrote the manuscript.
Peer review
Peer review information
Nature Communications thanks Zoran Culig, Jingyi Jessica Li, Shancheng Ren and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Data availability
Raw data of RNA-seq, single cell RNA-seq, ChIP-seq have been deposited in Gene Expression Omnibus database under accession number GSE164971. Source Data for this study are provided with this paper. All relevant data in this study are available within the article, Supplementary information, or Source Data. Source data are provided with this paper.
Code availability
The bioinformatics analyses were conducted using open-source software, including Cell Ranger version 3.1.0, Seurat version 3.2.111, R version 3.6.3, Novoalign version 3.02.07, MACS version 2.059, IGV version 6.360, cutadapt version 1.9.161, HISAT2 version 2.1.062, HTSeq version 0.6.163, edgeR version 2.26.766, GSEA version 4.1.068, and IPA version 01-20-04. R scripts used to process sequencing data are available in “GitHub repository [https://github.com/wk-kim/hAR-OSR1_Prostate_Tumorigenesis]”75.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Won Kyung Kim, Adam W. Olson.
Supplementary information
The online version contains supplementary material available at 10.1038/s41467-022-32119-0.
References
- 1.Rawla P. Epidemiology of Prostate Cancer. World J. Oncol. 2019;10:63–89. doi: 10.14740/wjon1191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Culig Z, Klocker H, Bartsch G, Steiner H, Hobisch A. Androgen receptors in prostate cancer. J. Urol. 2003;170:1363–1369. doi: 10.1097/01.ju.0000075099.20662.7f. [DOI] [PubMed] [Google Scholar]
- 3.Jenster G. The role of the androgen receptor in the development and progression of prostate cancer. Semin Oncol. 1999;26:407–421. [PubMed] [Google Scholar]
- 4.Abate-Shen C, Shen MM. Molecular genetics of prostate cancer. Genes Dev. 2000;14:2410–2434. doi: 10.1101/gad.819500. [DOI] [PubMed] [Google Scholar]
- 5.Huggins C, Hodges CV. Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. 1941. J. Urol. 2002;168:9–12. doi: 10.1016/S0022-5347(05)64820-3. [DOI] [PubMed] [Google Scholar]
- 6.Gelmann EP. Molecular biology of the androgen receptor. J. Clin. Oncol. 2002;20:3001–3015. doi: 10.1200/JCO.2002.10.018. [DOI] [PubMed] [Google Scholar]
- 7.Ittmann M, et al. Animal models of human prostate cancer: the consensus report of the New York meeting of the Mouse Models of Human Cancers Consortium Prostate Pathology Committee. Cancer Res. 2013;73:2718–2736. doi: 10.1158/0008-5472.CAN-12-4213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stanbrough M, Leav I, Kwan PW, Bubley GJ, Balk SP. Prostatic intraepithelial neoplasia in mice expressing an androgen receptor transgene in prostate epithelium. Proc. Natl Acad. Sci. 2001;98:10823–10828. doi: 10.1073/pnas.191235898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sun F, et al. Androgen receptor splice variant AR3 promotes prostate cancer via modulating expression of autocrine/paracrine factors. J. Biol. Chem. 2014;289:1529–1539. doi: 10.1074/jbc.M113.492140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhu C, et al. Conditional expression of the androgen receptor induces oncogenic transformation of the mouse prostate. J. Biol. Chem. 2011;286:33478–33488. doi: 10.1074/jbc.M111.269894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stuart T, et al. Comprehensive Integration of Single- Cell Data. Cell. 2019;177:1888–1902.e1821. doi: 10.1016/j.cell.2019.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee, D. H. et al. Androgen action in cell fate and communication during prostate development at single-cell resolution. Development (Cambridge, England)148, 10.1242/dev.196048 (2021). [DOI] [PMC free article] [PubMed]
- 13.Li, J. J. & Shen, M. M. Prostate Stem Cells and Cancer Stem Cells. Cold Spring Harbor Perspectives in Medicine9, 10.1101/cshperspect.a030395 (2019). [DOI] [PMC free article] [PubMed]
- 14.Reiter RE, et al. Prostate stem cell antigen: a cell surface marker overexpressed in prostate cancer. Proc. Natl Acad. Sci. 1998;95:1735–1740. doi: 10.1073/pnas.95.4.1735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang G, Gurtu V, Kain SR. An enhanced green fluorescent protein allows sensitive detection of gene transfer in mammalian cells. Biochem Biophys. Res Commun. 1996;227:707–711. doi: 10.1006/bbrc.1996.1573. [DOI] [PubMed] [Google Scholar]
- 16.Lee DH, et al. Deletion of the p16INK4a tumor suppressor and expression of the androgen receptor induce sarcomatoid carcinomas with signet ring cells in the mouse prostate. PLoS One. 2019;14:e0211153. doi: 10.1371/journal.pone.0211153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Joseph DB, et al. Urethral luminal epithelia are castration-insensitive cells of the proximal prostate. Prostate. 2020;80:872–884. doi: 10.1002/pros.24020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Downer JB, et al. Comparison of animal models for the evaluation of radiolabeled androgens. Nucl. Med. Biol. 2001;28:613–626. doi: 10.1016/S0969-8051(01)00229-3. [DOI] [PubMed] [Google Scholar]
- 19.Albertelli MA, Scheller A, Brogley M, Robins DM. Replacing the mouse androgen receptor with human alleles demonstrates glutamine tract length-dependent effects on physiology and tumorigenesis in mice. Mol. Endocrinol. 2006;20:1248–1260. doi: 10.1210/me.2006-0021. [DOI] [PubMed] [Google Scholar]
- 20.Baca SC, et al. Punctuated evolution of prostate cancer genomes. Cell. 2013;153:666–677. doi: 10.1016/j.cell.2013.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.DiGiovanni J, et al. Deregulated expression of insulin-like growth factor 1 in prostate epithelium leads to neoplasia in transgenic mice. Proc. Natl Acad. Sci. 2000;97:3455–3460. doi: 10.1073/pnas.97.7.3455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Figueroa JA, De Raad S, Speights VO, Rinehart JJ. Gene expression of insulin-like growth factors and receptors in neoplastic prostate tissues: correlation with clinico-pathological parameters. Cancer Invest. 2001;19:28–34. doi: 10.1081/CNV-100000072. [DOI] [PubMed] [Google Scholar]
- 23.Liao Y, et al. Up-regulation of insulin-like growth factor axis components in human primary prostate cancer correlates with tumor grade. Hum. Pathol. 2005;36:1186–1196. doi: 10.1016/j.humpath.2005.07.023. [DOI] [PubMed] [Google Scholar]
- 24.Schayek H, et al. Differential regulation of insulin-like growth factor-I receptor gene expression by wild type and mutant androgen receptor in prostate cancer cells. Mol. Cell Endocrinol. 2010;323:239–245. doi: 10.1016/j.mce.2010.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Saha A, Blando J, Fernandez I, Kiguchi K, DiGiovanni J. Linneg Sca-1high CD49fhigh prostate cancer cells derived from the Hi-Myc mouse model are tumor-initiating cells with basal-epithelial characteristics and differentiation potential in vitro and in vivo. Oncotarget. 2016;7:25194–25207. doi: 10.18632/oncotarget.7535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Toivanen R, Shen MM. Prostate organogenesis: tissue induction, hormonal regulation and cell type specification. Dev. (Camb., Engl.) 2017;144:1382–1398. doi: 10.1242/dev.148270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.He Y, et al. Androgen signaling is essential for development of prostate cancer initiated from prostatic basal cells. Oncogene. 2018;38:2337–2350. doi: 10.1038/s41388-018-0583-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Playford MP, Bicknell D, Bodmer WF, Macaulay VM. Insulin-like growth factor 1 regulates the location, stability, and transcriptional activity of beta-catenin. Proc. Natl Acad. Sci. 2000;97:12103–12108. doi: 10.1073/pnas.210394297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Verras M, Sun Z. Beta-catenin is involved in insulin-like growth factor 1-mediated transactivation of the androgen receptor. Mol. Endocrinol. 2005;19:391–398. doi: 10.1210/me.2004-0208. [DOI] [PubMed] [Google Scholar]
- 30.Verras M, Sun Z. Roles and regulation of Wnt signaling and beta-catenin in prostate cancer. Cancer Lett. 2006;237:22–32. doi: 10.1016/j.canlet.2005.06.004. [DOI] [PubMed] [Google Scholar]
- 31.Sharma M, Chuang WW, Sun Z. Phosphatidylinositol 3-kinase/Akt stimulates androgen pathway through GSK3beta inhibition and nuclear beta-catenin accumulation. J. Biol. Chem. 2002;277:30935–30941. doi: 10.1074/jbc.M201919200. [DOI] [PubMed] [Google Scholar]
- 32.Qiu X, et al. Reversed graph embedding resolves complex single-cell trajectories. Nat. Methods. 2017;14:979–982. doi: 10.1038/nmeth.4402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Abida W, et al. Genomic correlates of clinical outcome in advanced prostate cancer. Proc. Natl Acad. Sci. 2019;116:11428–11436. doi: 10.1073/pnas.1902651116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cerami E, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Disco. 2012;2:401–404. doi: 10.1158/2159-8290.CD-12-0095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Robinson D, et al. Integrative clinical genomics of advanced prostate cancer. Cell. 2015;161:1215–1228. doi: 10.1016/j.cell.2015.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Weischenfeldt J, et al. Integrative genomic analyses reveal an androgen-driven somatic alteration landscape in early-onset prostate cancer. Cancer Cell. 2013;23:159–170. doi: 10.1016/j.ccr.2013.01.002. [DOI] [PubMed] [Google Scholar]
- 37.Mulholland DJ, Cheng H, Reid K, Rennie PS, Nelson CC. The androgen receptor can promote beta-catenin nuclear translocation independently of adenomatous polyposis coli. J. Biol. Chem. 2002;277:17933–17943. doi: 10.1074/jbc.M200135200. [DOI] [PubMed] [Google Scholar]
- 38.Truica CI, Byers S, Gelmann EP. Beta-catenin affects androgen receptor transcriptional activity and ligand specificity. Cancer Res. 2000;60:4709–4713. [PubMed] [Google Scholar]
- 39.Yang F, et al. Linking beta-catenin to androgen-signaling pathway. J. Biol. Chem. 2002;277:11336–11344. doi: 10.1074/jbc.M111962200. [DOI] [PubMed] [Google Scholar]
- 40.Eguchi M, Nguyen C, Lee SC, Kahn M. ICG-001, a novel small molecule regulator of TCF/beta-catenin transcription. Med Chem. 2005;1:467–472. doi: 10.2174/1573406054864098. [DOI] [PubMed] [Google Scholar]
- 41.Lee E, et al. Inhibition of androgen receptor and beta-catenin activity in prostate cancer. Proc. Natl Acad. Sci. 2013;110:15710–15715. doi: 10.1073/pnas.1218168110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Grieshammer U, Agarwal P, Martin GR. A Cre transgene active in developing endodermal organs, heart, limb, and extra-ocular muscle. Genesis. 2008;46:69–73. doi: 10.1002/dvg.20366. [DOI] [PubMed] [Google Scholar]
- 43.Ma J, et al. Prediagnostic body-mass index, plasma C-peptide concentration, and prostate cancer-specific mortality in men with prostate cancer: a long-term survival analysis. Lancet Oncol. 2008;9:1039–1047. doi: 10.1016/S1470-2045(08)70235-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Price AJ, et al. Insulin-like growth factor-I concentration and risk of prostate cancer: results from the European Prospective Investigation into Cancer and Nutrition. Cancer Epidemiol. Biomark. Prev. 2012;21:1531–1541. doi: 10.1158/1055-9965.EPI-12-0481-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pollak M. Insulin and insulin-like growth factor signalling in neoplasia. Nat. Rev. Cancer. 2008;8:915–928. doi: 10.1038/nrc2536. [DOI] [PubMed] [Google Scholar]
- 46.Cox ME, et al. Insulin receptor expression by human prostate cancers. Prostate. 2009;69:33–40. doi: 10.1002/pros.20852. [DOI] [PubMed] [Google Scholar]
- 47.Arriaga, J. M. & Abate-Shen, C. Genetically Engineered Mouse Models of Prostate Cancer in the Postgenomic Era. Cold Spring Harbor Perspectives in Medicine9, 10.1101/cshperspect.a030528 (2019). [DOI] [PMC free article] [PubMed]
- 48.Brzezinska EA, Nixon C, Patel R, Leung HY. Genetically engineered mouse models to study prostate cancer. Methods Mol. Biol. (Clifton, NJ) 2015;1267:73–91. doi: 10.1007/978-1-4939-2297-0_4. [DOI] [PubMed] [Google Scholar]
- 49.Chesire DR, Isaacs WB. Ligand-dependent inhibition of beta-catenin/TCF signaling by androgen receptor. Oncogene. 2002;21:8453–8469. doi: 10.1038/sj.onc.1206049. [DOI] [PubMed] [Google Scholar]
- 50.Yang F, et al. Linking beta-catenin to androgen signaling pathway. J. Biol. Chem. 2002;277:11336–11344. doi: 10.1074/jbc.M111962200. [DOI] [PubMed] [Google Scholar]
- 51.Street K, et al. Slingshot: cell lineage and pseudotime inference for single-cell transcriptomics. BMC Genomics. 2018;19:477. doi: 10.1186/s12864-018-4772-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Song D, Li JJ. PseudotimeDE: inference of differential gene expression along cell pseudotime with well-calibrated p-values from single-cell RNA sequencing data. Genome Biol. 2021;22:124. doi: 10.1186/s13059-021-02341-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Muzumdar MD, Tasic B, Miyamichi K, Li L, Luo L. A global double-fluorescent Cre reporter mouse. Genesis. 2007;45:593–605. doi: 10.1002/dvg.20335. [DOI] [PubMed] [Google Scholar]
- 54.Wu X, et al. Generation of a prostate epithelial cell-specific Cre transgenic mouse model for tissue-specific gene ablation. Mech. Dev. 2001;101:61–69. doi: 10.1016/S0925-4773(00)00551-7. [DOI] [PubMed] [Google Scholar]
- 55.Drost J, et al. Organoid culture systems for prostate epithelial and cancer tissue. Nat. Protoc. 2016;11:347–358. doi: 10.1038/nprot.2016.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lee SH, Johnson D, Luong R, Sun Z. Crosstalking between androgen and PI3K/AKT signaling pathways in prostate cancer cells. J. Biol. Chem. 2015;290:2759–2768. doi: 10.1074/jbc.M114.607846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 58.He Y, et al. Loss of the tumor suppressor, Tp53, enhances the androgen receptor-mediated oncogenic transformation and tumor development in the mouse prostate. Oncogene. 2019;38:6507–6520. doi: 10.1038/s41388-019-0901-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang Y, et al. Model-based analysis of ChIP-Seq (MACS) Genome Biol. 2008;9:R137. doi: 10.1186/gb-2008-9-9-r137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Thorvaldsdóttir H, Robinson JT, Mesirov JP. Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief. Bioinforma. 2013;14:178–192. doi: 10.1093/bib/bbs017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kechin A, Boyarskikh U, Kel A, Filipenko M. cutPrimers: A New Tool for Accurate Cutting of Primers from Reads of Targeted Next Generation Sequencing. J. computational Biol.: a J. Computational Mol. Cell Biol. 2017;24:1138–1143. doi: 10.1089/cmb.2017.0096. [DOI] [PubMed] [Google Scholar]
- 62.Kim D, et al. Digenome-seq: genome-wide profiling of CRISPR-Cas9 off-target effects in human cells. Nat. Methods. 2015;12:237–243. doi: 10.1038/nmeth.3284. [DOI] [PubMed] [Google Scholar]
- 63.Anders S, Pyl PT, Huber W. HTSeq–a Python framework to work with high-throughput sequencing data. Bioinformatics. 2015;31:166–169. doi: 10.1093/bioinformatics/btu638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Robinson MD, Oshlack A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biol. 2010;11:R25. doi: 10.1186/gb-2010-11-3-r25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Bullard JH, Purdom E, Hansen KD, Dudoit S. Evaluation of statistical methods for normalization and differential expression in mRNA-Seq experiments. BMC Bioinforma. 2010;11:94. doi: 10.1186/1471-2105-11-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chen Y, Lun AT, Smyth GK. From reads to genes to pathways: differential expression analysis of RNA-Seq experiments using Rsubread and the edgeR quasi-likelihood pipeline. F1000Res. 2016;5:1438. doi: 10.12688/f1000research.8987.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ge X, et al. Clipper: p-value-free FDR control on high-throughput data from two conditions. Genome Biol. 2021;22:288. doi: 10.1186/s13059-021-02506-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Subramanian A, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. 2005;102:15545–15550. doi: 10.1073/pnas.0506580102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Barrett T, et al. NCBI GEO: archive for functional genomics data sets–update. Nucl. Acids Res. 2013;41:D991–995. doi: 10.1093/nar/gks1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.An integrated encyclopedia of DNA elements in the human genome. Nature489, 57–74 (2012). [DOI] [PMC free article] [PubMed]
- 71.Karolchik D, et al. The UCSC Genome Browser Database. Nucl.Acids Res. 2003;31:51–54. doi: 10.1093/nar/gkg129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cunha GR, Li Y, Mei C, Derpinghaus A, Baskin LS. Ontogeny of estrogen receptors in human male and female fetal reproductive tracts. Differentiation. 2021;118:107–131. doi: 10.1016/j.diff.2020.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sharma A, Yang WL, Ochani M, Wang P. Mitigation of sepsis-induced inflammatory responses and organ injury through targeting Wnt/beta-catenin signaling. Sci. Rep. 2017;7:9235. doi: 10.1038/s41598-017-08711-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Xiao H, Zeng Y, Wang Q, Wei S, Zhu X. A Novel Positive Feedback Loop Between NTSR1 and Wnt/beta-Catenin Contributes to Tumor Growth of Glioblastoma. Cell Physiol. Biochem. 2017;43:2133–2142. doi: 10.1159/000484232. [DOI] [PubMed] [Google Scholar]
- 75.Kim WK, et al. Aberrant Androgen Action in Prostatic Progenitor Cells Induces Oncogenesis and Tumor Development through IGF1 and Wnt Axes. Zenondo10.5281/zenodo.6829659 (2022). [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Description of Additional Supplementary Files
Data Availability Statement
Raw data of RNA-seq, single cell RNA-seq, ChIP-seq have been deposited in Gene Expression Omnibus database under accession number GSE164971. Source Data for this study are provided with this paper. All relevant data in this study are available within the article, Supplementary information, or Source Data. Source data are provided with this paper.
The bioinformatics analyses were conducted using open-source software, including Cell Ranger version 3.1.0, Seurat version 3.2.111, R version 3.6.3, Novoalign version 3.02.07, MACS version 2.059, IGV version 6.360, cutadapt version 1.9.161, HISAT2 version 2.1.062, HTSeq version 0.6.163, edgeR version 2.26.766, GSEA version 4.1.068, and IPA version 01-20-04. R scripts used to process sequencing data are available in “GitHub repository [https://github.com/wk-kim/hAR-OSR1_Prostate_Tumorigenesis]”75.