Abstract
OBJECTIVE
Whether and how dietary protein intake is linked to type 2 diabetes (T2D) remains unclear. The aim of this study was to investigate the associations of protein intake with development of T2D and the potential mediating roles of T2D biomarkers.
RESEARCH DESIGN AND METHODS
We included 108,681 postmenopausal women without T2D at baseline from the Women’s Health Initiative (WHI) (primary cohort) and 34,616 adults without T2D from the U.K. Biobank (UKB) (replication cohort). Cox proportional hazard models were used for estimation of protein-T2D associations. Mediation analysis was performed to assess the mediating roles of biomarkers in case-control studies nested in the WHI.
RESULTS
In the WHI, 15,842 incident T2D cases were identified during a median follow-up of 15.8 years. Intake of animal protein was associated with increased T2D risk (hazard ratio in comparing the highest to the lowest quintile = 1.31 [95% CI 1.24–1.37]) and plant protein with decreased risk (0.82 [0.78–0.86]). Intakes of red meat, processed meat, poultry, and eggs were associated with increased T2D risk and whole grains with decreased risk. Findings from the UKB were similar. These findings were materially attenuated after additional adjustment for BMI. Substituting 5% energy from plant protein for animal protein was associated with 21% decreased T2D risk (0.79 [0.74–0.84]), which was mediated by levels of hs-CRP, interleukin-6, leptin, and SHBG.
CONCLUSIONS
Findings from these two large prospective cohorts support the notion that substituting plant protein for animal protein may decrease T2D risk mainly by reducing obesity-related inflammation.
Introduction
Dietary modification or medical nutrition therapy remains the cornerstone in preventing and managing type 2 diabetes (T2D) (1,2). Although much is known about the effects of fats versus carbohydrates on risk of T2D (3), fewer studies have directly investigated the role of dietary protein in the development of T2D (4). While recent work has shown a positive association between intake of total and animal proteins with risk of T2D, findings relating plant protein intake to T2D risk remain inconsistent (5–8). Moreover, few prospective studies (9,10) have assessed the relation of protein food sources intake to T2D risk and the beneficial associations between substituting different protein sources and T2D risk.
Although substituting plant protein for animal protein was associated with decreased risk of T2D, none have attempted to evaluate the mediating roles of known biochemical intermediaries, limiting our understanding of mechanistic pathways by which dietary proteins may affect T2D risk. Obesity (11), inflammation (12), endothelial dysfunction (13), sex steroids and sex hormone binding globulin (SHBG) (14–17), and telomere attrition (18,19) are well-characterized T2D risk factors or biomarkers, which are also associated with some protein sources intake (20–23). Therefore, these biomarkers may represent critical mediators for the beneficial association between substituting plant protein for animal protein and T2D risk.
To fill such gaps in the knowledge base for T2D prevention related to dietary modification, we conducted analyses for 1) investigation of the roles of different animal and plant protein sources in relation to incident T2D among participants enrolled in the Women’s Health Initiative (WHI) and the U.K. Biobank (UKB), 2) evaluation of the beneficial association between substituting different protein sources with T2D risk in the WHI and the UKB, and 3) examination of the extent to which known T2D-related biomarkers could explain the association of substituting plant protein for animal protein in case-control studies nested in the WHI.
Research Design and Methods
Study Populations
We included the WHI as the primary analytical cohort and the UKB as the replication cohort. Between 1993 and 1998, 161,808 postmenopausal women aged 50–79 years from 40 clinical centers in the U.S. were recruited into the WHI Observational Study (OS) and clinical trials (CT). The study design was previously described in detail (24). We excluded participants who were assigned to the treatment arms in the WHI CT or had self-reported diabetes at baseline, missing dietary information, or implausible daily energy intake (<600 or >5,000 kcal/day) from a validated food-frequency questionnaire (FFQ) (25). Finally, a total of 108,681 participants with a median follow-up duration of 15.8 years until 28 February 2020 were available for the analysis. In the UKB, we included 34,616 participants free of diabetes at baseline who then were followed up for a median of 11.4 years. Details of the UKB cohort and related methods can be found in Supplementary Methods.
Measurement of Dietary Protein Sources
In the WHI, dietary data were collected for each participant at enrollment with a validated self-administered FFQ at baseline (25). Daily nutrient intake was calculated with use of the University of Minnesota Nutrient Data System for Research (25). The energy-adjusted correlation coefficients between the FFQ and food records ranged from 0.2 to 0.7 for the estimated intakes of 30 nutrients, with 0.4 for protein (25). Food items in the FFQ were summarized into 32 food groups based on the MyPyramid Equivalents Database (MPED) 2.0 (26,27), among which were the dietary protein sources of interest. Based on a study in 10 countries in the European Prospective Investigation into Cancer and Nutrition (EPIC) (28), we modified the EPIC protein source list to include red meat, processed meat, poultry, high–n-3 seafood, low–n-3 seafood, cheese, yogurt, milk, and eggs as major animal protein sources and legumes, nuts, and whole grains as major plant protein sources. Specific food items in each protein source are listed in Supplementary Table 1.
Ascertainment of Incident T2D Cases
In the WHI, incident T2D cases were identified during annual follow-up with self-administered questionnaires. Participants were asked if “a doctor prescribed pills or insulin shots for diabetes for the first time” since their last medical update. Validation studies of self-reported diabetes with use of medical records and biomarkers indicated high accuracy and reliability (12,29).
Covariates
Demographic characteristics and lifestyle factors for each participant collected at baseline were included in our models as covariates, including age, race/ethnicity, smoking status, alcohol intake, total energy intake, education, family income, physical activity, region of residence, family history of diabetes, use of menopausal hormone therapy, multivitamin use, and antihypertension medicine and score of a modified Alternate Healthy Eating Index (AHEI)-2010 (30) with removal of red/processed meat, long-chain (n-3) fats including eicosapentaenoic acid and docosahexaenoic acid, nuts and legumes, and whole grains. The AHEI-2010 modified score ranged from 0 to 70 indicating a lower- to a higher-quality modified diet. Detailed descriptions of the validity and reproducibility of baseline measurements have previously been published (31). The proportions of missing data for covariates were <4%; mean or mode imputation was used for continuous and categorical covariates, respectively.
Nested Case-Control Studies Within the WHI
We completed a series of case-control studies nested in the WHI to investigate the associations between biomarkers and T2D risk (12,13,16,18). Using these nested case-control studies, we further examined the potential roles of specific T2D-related biomarkers that could mediate the protein-T2D associations. Details of these case-control studies (12,15,18) and quality control of specific biomarker measurements (24) have previously been published. After exclusion of participants with missing dietary data, 3,464 participants with five sets of biomarkers were included in the mediation analysis. These included 1) biomarkers for inflammation, including tumor necrosis factor-α receptor 2 (TNFα-R2), interleukin-6 (IL-6), and hs-CRP; 2) biomarkers for endothelial dysfunction (i.e., vascular cell adhesion molecule 1 [VCAM-1], E-selectin, and soluble intercellular adhesion molecule-1 [SICAM-1]); 3) sex steroids, including estradiol, testosterone, and SHBG; 4) leptin and soluble leptin receptor; and 5) leukocyte telomere length for aging.
Statistical Analyses
In the WHI, each participant’s follow-up time was defined as the duration between entry to the cohort and the date of occurrence of T2D or censor (death, lost to follow-up, or end of follow-up)—whichever occurred first. Cox proportional hazards model was used to evaluate the associations of energy-adjusted dietary protein and its food sources with risk of incident T2D. Dietary protein sources were entered into the models as quintiles, with the lowest quintile serving as the reference group.
Covariates were adjusted for in multivariable models. In model 1, we adjusted for age, study group indicator (OS, CT), self-identified race and ethnicity (White, Black, Hispanic, Asian), region of residence at baseline (Northeast, South, Midwest, West), family income (<$20,000, $20,000–49,999, $50,000–99,999, >$100,000), education (<high school, high school, college, postgraduate), and family history of diabetes (yes/no); in model 2, we further included smoking status (never, past, current smoker), alcohol intake (continuous), physical activity (continuous), hormone replacement therapy (never, past, current user), multivitamin use (yes/no), antihypertensive medication use (never, previously, currently treated), and total energy intake (continuous); in model 3 (full model), we additionally adjusted for modified AHEI-2010 score (continuous) and intake of other dietary protein sources; in model 4 (sensitivity model), we further adjusted for BMI and waist-to-hip ratio (WHR) given that BMI could be both a confounder and mediator in protein-T2D associations. We also included restricted cubic spline term for dietary protein and food sources with three knots at 10th, 50th, and 90th centiles in model 3 to explore potential nonlinear relations of specific dietary protein sources to T2D risk. The nonlinearity P value was estimated with a likelihood ratio test.
We used the leave-one-out model to evaluate the association between isocaloric substitution of plant protein for animal protein and T2D risk. We simultaneously included the percentage of energy from carbohydrate, fat, and plant protein and total energy intake with covariates in a multivariable model. The coefficient of plant protein from the model can be interpreted as the estimated effect of substituting a specific percentage of energy from plant protein for the same percentage of energy from animal protein.
In the mediation analysis, we examined whether the beneficial associations of substituting plant protein for animal protein with decreased risk of T2D were mediated by well-characterized T2D-related biochemical intermediaries. We constructed two regression models, a logistic model, to regress the outcome (T2D) on the exposure (protein intake) and the mediator (biomarker), and a weighted linear model, to regress the mediator on the exposure, with adjustment for potential confounders. We integrated these two regressions to obtain the estimates for direct and indirect effects using the regression-based approach proposed by VanderWeele (24,32,33). The proportion of mediating effect was estimated on an odds ratio (OR) scale; 95% CIs were obtained via bootstrapping. All analyses were performed with R (version 4.0.3). Two-sided P values and 95% CIs were calculated for statistical inference.
Results
Associations of Dietary Protein Food Sources Intake With Risk of Incident T2D
In the WHI, among the 108,681 postmenopausal women, mean age 63 years at baseline, 15,842 (14.6%) participants developed T2D during a median follow-up period of 15.8 years (Table 1). The postmenopausal women with higher energy-adjusted intake of total protein were more likely to be White, educated, physically active, and have a higher BMI and income, while they were less likely to be a smoker or drinker, compared with those with lower energy-adjusted intake of total protein (Table 1). In the fully adjusted model, participants with higher energy-adjusted intake of animal protein had increased risk of incident T2D (adjusted hazard ratio [HR] in comparing the highest with the lowest quintile: 1.31 [95% CI 1.24–1.37], P for trend <0.001). In contrast, participants with greater intake of energy-adjusted plant protein had lower risk of incident T2D (0.82 [0.78–0.86], P for trend <0.001) (Table 2).
Table 1.
Baseline characteristics according to quintiles of daily intake of total dietary protein sources among 108,681 postmenopausal women in the WHI
| Total | Quintiles of energy-adjusted total protein | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | ||
| N | 108,681 | 21,737 | 21,736 | 21,736 | 21,736 | 21,736 |
| Follow-up duration (person-years) | 1,654,022 | 321,986 | 328,762 | 334,337 | 336,959 | 331,979 |
| Incident diabetes | 15,842 (14.6) | 3,039 (14.0) | 2,985 (13.7) | 3,108 (14.3) | 3,193 (14.7) | 3,517 (16.2) |
| Age at baseline (years) | 63 ± 7 | 63 ± 7 | 63 ± 7 | 64 ± 7 | 63 ± 7 | 63 ± 7 |
| Self-identified ethnicity or race | ||||||
| White | 92,209 (84.8) | 17,248 (79.3) | 18,045 (83.0) | 18,605 (85.6) | 19,097 (87.9) | 19,214 (88.4) |
| Black | 8,018 (7.4) | 2,595 (11.9) | 1,789 (8.2) | 1,442 (6.6) | 1,123 (5.2) | 1,069 (4.9) |
| Hispanic | 3,768 (3.5) | 876 (4.0) | 805 (3.7) | 720 (3.3) | 667 (3.1) | 700 (3.2) |
| Asian | 2,818 (2.6) | 597 (2.7) | 721 (3.3) | 591 (2.7) | 494 (2.3) | 415 (1.9) |
| Region of residence at baseline | ||||||
| Northeast | 25,126 (23.1) | 5,212 (24.0) | 4,944 (22.7) | 4,951 (22.8) | 5,196 (23.9) | 4,823 (22.2) |
| South | 27,803 (25.6) | 6,088 (28.0) | 5,700 (26.2) | 5,501 (25.3) | 5,208 (24.0) | 5,306 (24.4) |
| Midwest | 23,823 (21.9) | 4,197 (19.3) | 4,345 (20.0) | 4,647 (21.4) | 5,015 (23.1) | 5,619 (25.9) |
| West | 31,929 (29.4) | 6,240 (28.7) | 6,747 (31.0) | 6,637 (30.5) | 6,317 (29.1) | 5,988 (27.5) |
| Education, n (%) | ||||||
| <High school | 4,878 (4.5) | 1,343 (6.2) | 1,071 (4.9) | 954 (4.4) | 763 (3.5) | 747 (3.4) |
| High school | 28,030 (25.8) | 6,212 (28.6) | 5,930 (27.3) | 5,571 (25.6) | 5,401 (24.8) | 4,916 (22.6) |
| College | 42,650 (39.2) | 8,236 (37.9) | 8,479 (39) | 8,555 (39.4) | 8,751 (40.3) | 8,629 (39.7) |
| Postgraduate | 33,123 (30.5) | 5,946 (27.4) | 6,256 (28.8) | 6,656 (30.6) | 6,821 (31.4) | 7,444 (34.2) |
| BMI (kg/m2) | 27.3 ± 5.7 | 27.3 ± 5.8 | 26.8 ± 5.5 | 27.1 ± 5.5 | 27.4 ± 5.7 | 28.1 ± 5.9 |
| Waist circumference (cm) | 84.9 ± 13.3 | 85.1 ± 13.5 | 83.8 ± 12.9 | 84.2 ± 12.8 | 84.9 ± 13.2 | 86.5 ± 13.7 |
| Recreational physical activity (MET h/week) | 13.4 ± 13.9 | 11.8 ± 13.7 | 13.0 ± 13.7 | 13.4 ± 13.7 | 13.9 ± 14 | 14.5 ± 14.2 |
| Alcohol intake (g/day) | 5.63 ± 11.0 | 8.3 ± 16.6 | 5.8 ± 10.2 | 5.1 ± 8.9 | 4.7 ± 8.3 | 4.3 ± 7.9 |
| Smoking status | ||||||
| Never | 55,871 (51.4) | 10,798 (49.7) | 11,311 (52.0) | 11,297 (52.0) | 11,269 (51.8) | 11,196 (51.5) |
| Past | 45,870 (42.2) | 9,048 (41.6) | 8,982 (41.3) | 9,144 (42.1) | 9,256 (42.6) | 9,440 (43.4) |
| Current | 6,940 (6.4) | 1,891 (8.7) | 1,443 (6.6) | 1,295 (6.0) | 1,211 (5.6) | 1,100 (5.1) |
| Income (USD) | ||||||
| <20,000 | 15,167 (14.0) | 3,844 (17.7) | 3,289 (15.1) | 2,902 (13.4) | 2,620 (12.1) | 2,512 (11.6) |
| 20,000–49,999 | 52,139 (48.0) | 10,728 (49.4) | 10,517 (48.4) | 10,434 (48.0) | 10,319 (47.5) | 10,141 (46.7) |
| 50,000–99,999 | 30,433 (28.0) | 5,406 (24.9) | 5,835 (26.8) | 6,209 (28.6) | 6,435 (29.6) | 6,548 (30.1) |
| >100,000 | 10,942 (10.1) | 1,759 (8.1) | 2,095 (9.6) | 2,191 (10.1) | 2,362 (10.9) | 2,535 (11.7) |
| Family history of diabetes | 32,612 (30.0) | 6,397 (29.4) | 6,334 (29.1) | 6,489 (29.9) | 6,505 (29.9) | 6,887 (31.7) |
| Hormone replacement therapy | ||||||
| Never | 35,252 (32.4) | 7,556 (34.8) | 7,109 (32.7) | 6,827 (31.4) | 6,848 (31.5) | 6,912 (31.8) |
| Past | 23,027 (21.2) | 4,790 (22.0) | 4,541 (20.9) | 4,651 (21.4) | 4,476 (20.6) | 4,569 (21.0) |
| Current | 50,402 (46.4) | 9,391 (43.2) | 10,086 (46.4) | 10,258 (47.2) | 10,412 (47.9) | 10,255 (47.2) |
| Antihypertension medication | ||||||
| Never treated | 75,334 (69.3) | 14,877 (68.4) | 15,252 (70.2) | 15,167 (69.8) | 15,131 (69.6) | 14,907 (68.6) |
| Previously treated | 8,270 (7.6) | 1,779 (8.2) | 1,640 (7.5) | 1,556 (7.2) | 1,537 (7.1) | 1,758 (8.1) |
| Currently treated | 25,077 (23.1) | 5,081 (23.4) | 4,844 (22.3) | 5,013 (23.1) | 5,068 (23.3) | 5,071 (23.3) |
| Total energy intake (kcal/day) | 1,610 ± 619 | 1,803 ± 706 | 1,453 ± 562 | 1,455 ± 542 | 1,531 ± 538 | 1,796 ± 626 |
| Total carbohydrate (g/day) | 203 ± 78.6 | 230.7 ± 89.5 | 188.0 ± 72.0 | 186.7 ± 70.2 | 193.5 ± 70 | 218.6 ± 79.3 |
| Total fat (g/day) | 57.5 ± 31.2 | 69.8 ± 37.6 | 52.6 ± 27.8 | 51.2 ± 26.6 | 52.8 ± 26.7 | 61.0 ± 31.7 |
| Saturated fat (g/day) | 19.3 ± 11.3 | 23.2 ± 13.7 | 17.5 ± 10.1 | 17.1 ± 9.6 | 17.7 ± 9.7 | 20.8 ± 11.6 |
| Monounsaturated fat (g/day) | 21.8 ± 12.1 | 26.6 ± 14.5 | 19.9 ± 10.8 | 19.4 ± 10.3 | 19.9 ± 10.5 | 23.0 ± 12.4 |
| Polyunsaturated fat (g/day) | 11.9 ± 6.7 | 14.9 ± 8.5 | 11.1 ± 6.0 | 10.6 ± 5.6 | 10.8 ± 5.6 | 12.1 ± 6.2 |
| Total protein (g/day) | 67.1 ± 27.8 | 49.9 ± 8.0 | 60.8 ± 1.8 | 66.5 ± 1.6 | 72.7 ± 2.1 | 85.7 ± 9.2 |
| Animal protein (g/day) | 46.7 ± 22.7 | 36.0 ± 19.1 | 35.7 ± 16.8 | 41.4 ± 16.2 | 49.7 ± 16.2 | 70.8 ± 23.7 |
| Plant protein (g/day) | 20.3 ± 8.7 | 21.5 ± 9.4 | 18.8 ± 8.2 | 19.0 ± 8.0 | 19.8 ± 8.1 | 22.3 ± 9.1 |
| Dietary protein sources (g/day) | ||||||
| Red meat | 41.1 ± 36.5 | 31.5 ± 27.8 | 31.4 ± 26.1 | 36.1 ± 28.1 | 43.5 ± 32.9 | 63.1 ± 51.4 |
| Processed meat | 11.2 ± 14.0 | 10.7 ± 14.3 | 9.3 ± 12 | 10.1 ± 12.3 | 11.3 ± 13.2 | 14.8 ± 17 |
| Eggs | 15.5 ± 18.4 | 15.9 ± 17.3 | 13.7 ± 15.3 | 14.1 ± 15.4 | 15.2 ± 17.5 | 18.8 ± 24.3 |
| Poultry | 25.6 ± 22.0 | 16.5 ± 15.9 | 18 ± 15.7 | 22.5 ± 16.8 | 29 ± 19.6 | 42.1 ± 28.7 |
| High–n-3 seafood | 9.4 ± 11.8 | 5.63 ± 7.35 | 6.76 ± 7.84 | 8.46 ± 9.08 | 10.81 ± 11.34 | 15.53 ± 17.7 |
| Low–n-3 seafood | 7.9 ± 8.8 | 6.71 ± 7.55 | 6.56 ± 6.88 | 7.26 ± 7.14 | 8.21 ± 8.25 | 10.99 ± 12.19 |
| Cheese | 111.0 ± 104 | 95.4 ± 91 | 89.8 ± 83 | 98.8 ± 85.1 | 114.4 ± 97.3 | 155.8 ± 140.7 |
| Milk | 235.9 ± 233.9 | 171.7 ± 161.7 | 172.9 ± 158.1 | 205.4 ± 178.8 | 252.3 ± 214.4 | 377 ± 339.7 |
| Yogurt | 41.0 ± 68.7 | 27.3 ± 55.4 | 32.9 ± 59.6 | 39.2 ± 63.9 | 44.6 ± 67.5 | 60.8 ± 87.5 |
| Whole grains | 34.4 ± 28.2 | 33.08 ± 29.9 | 31.32 ± 26.41 | 32.72 ± 26.38 | 35.07 ± 27.08 | 39.95 ± 30.12 |
| Legumes | 20.3 ± 26.4 | 18.4 ± 25.3 | 18.4 ± 23.9 | 19.2 ± 24.4 | 20.7 ± 26.4 | 24.7 ± 30.7 |
| Nuts | 10.1 ± 16.0 | 12.2 ± 19.1 | 9.6 ± 14.9 | 9.1 ± 14.8 | 9.2 ± 14.6 | 10.2 ± 15.9 |
| Modified AHEI-2010 score | 35.5 ± 8.0 | 33.3 ± 8.3 | 35.8 ± 8 | 36.3 ± 7.7 | 36.4 ± 7.7 | 35.8 ± 7.7 |
Data for continuous variables are presented as mean ± SD. Data for categorical variables are presented as n (%).
Table 2.
Adjusted HRs with 95% CIs of T2D according to quintiles of daily intake of dietary protein and food sources among 108,681 postmenopausal women in the WHI
| Quintiles of dietary protein and food sources | P for trend | |||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | ||
| N | 21,737 | 21,736 | 21,736 | 21,736 | 21,736 | |
| Total protein | ||||||
| Daily intake, g/day | 49.9 ± 8.0 | 60.8 ± 1.8 | 66.5 ± 1.6 | 72.7 ± 2.1 | 85.7 ± 9.2 | |
| Cases/person-years | 3,039/321,986 | 2,985/328,762 | 3,108/334,337 | 3,193/336,959 | 3,517/331,979 | |
| Model 1 | Ref | 1.01 (0.96–1.06) | 1.05 (1.00–1.11) | 1.09 (1.04–1.15) | 1.25 (1.19–1.31) | <0.001 |
| Model 2 | Ref | 1.05 (0.99–1.10) | 1.09 (1.03–1.14) | 1.11 (1.06–1.17) | 1.21 (1.16–1.28) | <0.001 |
| Model 3 | Ref | 1.06 (1.00–1.11) | 1.10 (1.05–1.16) | 1.13 (1.07–1.19) | 1.24 (1.18–1.30) | <0.001 |
| Model 4 | Ref | 1.06 (1.00–1.12) | 1.10 (1.04–1.18) | 1.12 (1.04–1.20) | 1.21 (1.10–1.33) | <0.001 |
| Animal protein | ||||||
| Daily intake, g/day | 28.3 ± 8.0 | 40.0 ± 2.0 | 46.1 ± 1.7 | 52.6 ± 2.2 | 66.6 ± 10.2 | |
| Cases/person-years | 2,911/329,625 | 3,017/330,174 | 3,064/332,403 | 3,192/334,318 | 3,658/327,503 | |
| Model 1 | Ref | 1.06 (1.01–1.12) | 1.09 (1.03–1.14) | 1.14 (1.09–1.20) | 1.35 (1.29–1.42) | <0.001 |
| Model 2 | Ref | 1.09 (1.04–1.15) | 1.11 (1.06–1.17) | 1.16 (1.10–1.22) | 1.31 (1.25–1.37) | <0.001 |
| Model 3 | Ref | 1.09 (1.03–1.14) | 1.11 (1.05–1.17) | 1.15 (1.09–1.21) | 1.31 (1.24–1.37) | <0.001 |
| Model 4 | Ref | 1.06 (1.00–1.13) | 1.06 (1.00–1.14) | 1.08 (1.00–1.17) | 1.20 (1.08–1.32) | 0.008 |
| Plant protein | ||||||
| Daily intake, g/day | 13.5 ± 2.8 | 17.5 ± 0.7 | 19.7 ± 0.6 | 22.3 ± 0.9 | 28.5 ± 5.0 | |
| Cases/person-years | 3,628/313,107 | 3,230/326,656 | 3,081/332,941 | 3,021/338,691 | 2,882/342,627 | |
| Model 1 | Ref | 0.88 (0.84–0.92) | 0.84 (0.80–0.88) | 0.82 (0.78–0.86) | 0.77 (0.73–0.81) | <0.001 |
| Model 2 | Ref | 0.92 (0.87–0.96) | 0.87 (0.83–0.91) | 0.84 (0.80–0.89) | 0.78 (0.75–0.83) | <0.001 |
| Model 3 | Ref | 0.93 (0.88–0.97) | 0.89 (0.84–0.93) | 0.87 (0.83–0.92) | 0.82 (0.78–0.86) | <0.001 |
| Model 4 | Ref | 1.00 (0.95–1.05) | 1.00 (0.94–1.05) | 1.02 (0.96–1.09) | 1.04 (0.96–1.13) | 0.34 |
| Red meat | ||||||
| Daily intake, g/day | 6.7 ± 3.8 | 18.6 ± 3.3 | 31.3 ± 4.3 | 50.5 ± 7.2 | 98.5 ± 37.4 | |
| Cases/person-years | 2,679/331,140 | 2,997/331,851 | 3,097/332,529 | 3,310/331,514 | 3,759/326,988 | |
| Model 1 | Ref | 1.13 (1.07–1.19) | 1.18 (1.12–1.24) | 1.27 (1.21–1.34) | 1.43 (1.36–1.50) | <0.001 |
| Model 2 | Ref | 1.09 (1.03–1.15) | 1.12 (1.06–1.18) | 1.17 (1.11–1.24) | 1.27 (1.20–1.34) | <0.001 |
| Model 3 | Ref | 1.05 (1.00–1.11) | 1.06 (1.01–1.12) | 1.10 (1.04–1.16) | 1.15 (1.09–1.22) | <0.001 |
| Model 4 | Ref | 1.03 (0.97–1.08) | 1.00 (0.94–1.06) | 1.00 (0.94–1.06) | 0.97 (0.89–1.06) | 0.59 |
| Processed meat | ||||||
| Daily intake, g/day | 0.5 ± 0.6 | 3.2 ± 0.9 | 6.6 ± 1.2 | 12.7 ± 2.6 | 33.3 ± 16.9 | |
| Cases/person-years | 2,687/335,320 | 2,908/333,408 | 3,163/335,418 | 3,434/326,240 | 3,650/323,637 | |
| Model 1 | Ref | 1.09 (1.03–1.14) | 1.17 (1.11–1.23) | 1.29 (1.23–1.36) | 1.37 (1.30–1.44) | <0.001 |
| Model 2 | Ref | 1.06 (1.00–1.12) | 1.11 (1.06–1.17) | 1.20 (1.13–1.26) | 1.23 (1.16–1.30) | <0.001 |
| Model 3 | Ref | 1.03 (0.97–1.08) | 1.06 (1.00–1.12) | 1.12 (1.06–1.18) | 1.14 (1.08–1.20) | <0.001 |
| Model 4 | Ref | 1.02 (0.97–1.07) | 1.01 (0.96–1.07) | 1.04 (0.98–1.10) | 0.99 (0.92–1.07) | 0.60 |
| Poultry | ||||||
| Daily intake, g/day | 4.7 ± 2.5 | 11.7 ± 1.8 | 19.9 ± 3.4 | 31.9 ± 5.6 | 59.9 ± 21.9 | |
| Cases/person-years | 2,751/322,365 | 3,110/330,299 | 3,166/333,001 | 3,315/337,557 | 3,500/330,800 | |
| Model 1 | Ref | 1.10 (1.05–1.16) | 1.12 (1.06–1.18) | 1.16 (1.10–1.22) | 1.21 (1.15–1.27) | <0.001 |
| Model 2 | Ref | 1.07 (1.02–1.13) | 1.07 (1.02–1.13) | 1.10 (1.04–1.16) | 1.11 (1.05–1.17) | <0.001 |
| Model 3 | Ref | 1.04 (0.99–1.10) | 1.03 (0.98–1.09) | 1.06 (1.01–1.12) | 1.09 (1.03–1.15) | 0.003 |
| Model 4 | Ref | 1.03 (0.98–1.09) | 1.00 (0.95–1.06) | 1.01 (0.94–1.07) | 0.97 (0.89–1.06) | 0.72 |
| Eggs | ||||||
| Daily intake, g/day | 1.5 ± 1.1 | 5.6 ± 1.3 | 9.8 ± 1.4 | 17.8 ± 3.5 | 42.8 ± 24.3 | |
| Cases/person-years | 2,844/336,357 | 2,872/336,758 | 3,146/336,639 | 3,224/330,310 | 3,756/313,959 | |
| Model 1 | Ref | 1.00 (0.95–1.06) | 1.09 (1.04–1.15) | 1.15 (1.09–1.21) | 1.38 (1.31–1.45) | <0.001 |
| Model 2 | Ref | 0.99 (0.94–1.04) | 1.05 (1.00–1.10) | 1.11 (1.05–1.16) | 1.27 (1.20–1.34) | <0.001 |
| Model 3 | Ref | 0.97 (0.92–1.02) | 1.01 (0.96–1.07) | 1.07 (1.01–1.13) | 1.21 (1.14–1.27) | <0.001 |
| Model 4 | Ref | 0.94 (0.89–0.99) | 0.95 (0.91–1.01) | 1.00 (0.94–1.05) | 1.03 (0.96–1.11) | 0.29 |
| High–n-3 seafood | ||||||
| Daily intake, g/day | 0.3 ± 0.3 | 2.6 ± 0.9 | 6.1 ± 1 | 11.1 ± 1.8 | 27.1 ± 15.5 | |
| Cases/person-years | 3,289/311,838 | 3,184/321,288 | 3,093/334,076 | 3,129/343,799 | 3,147/343,021 | |
| Model 1 | Ref | 0.98 (0.93–1.03) | 0.94 (0.90–0.99) | 0.95 (0.90–1.00) | 0.98 (0.94–1.04) | 0.26 |
| Model 2 | Ref | 0.99 (0.94–1.04) | 0.96 (0.91–1.01) | 0.97 (0.92–1.02) | 1.01 (0.96–1.06) | 0.86 |
| Model 3 | Ref | 0.99 (0.94–1.04) | 0.96 (0.91–1.01) | 0.97 (0.92–1.02) | 1.01 (0.96–1.07) | 0.87 |
| Model 4 | Ref | 0.99 (0.95–1.04) | 0.98 (0.93–1.03) | 0.99 (0.93–1.05) | 1.02 (0.95–1.11) | 0.95 |
| Low–n-3 seafood | ||||||
| Daily intake, g/day | 0.8 ± 0.7 | 3.2 ± 0.7 | 5.7 ± 0.9 | 9.4 ± 1.3 | 20.6 ± 11.8 | |
| Cases/person-years | 2,807/322,442 | 3,067/332,773 | 3,071/335,496 | 3,300/337,318 | 3,597/325,993 | |
| Model 1 | Ref | 1.08 (1.03–1.14) | 1.07 (1.02–1.13) | 1.14 (1.08–1.20) | 1.25 (1.19–1.31) | <0.001 |
| Model 2 | Ref | 1.08 (1.02–1.13) | 1.05 (1.00–1.10) | 1.10 (1.05–1.16) | 1.17 (1.11–1.24) | <0.001 |
| Model 3 | Ref | 1.07 (1.01–1.12) | 1.03 (0.98–1.08) | 1.07 (1.02–1.13) | 1.12 (1.06–1.18) | <0.001 |
| Model 4 | Ref | 1.06 (1.01–1.12) | 1.01 (0.96–1.06) | 1.03 (0.97–1.09) | 0.99 (0.93–1.07) | 0.75 |
| Cheese | ||||||
| Daily intake, g/day | 18 ± 10.7 | 49.1 ± 8.4 | 81.9 ± 10.8 | 131.9 ± 19.4 | 273.3 ± 117.3 | |
| Cases/person-years | 2,968/308,884 | 3,089/328,020 | 3,115/337,099 | 3,344/339,742 | 3,326/340,278 | |
| Model 1 | Ref | 1.04 (0.99–1.10) | 1.05 (1.00–1.11) | 1.15 (1.09–1.21) | 1.14 (1.09–1.20) | <0.001 |
| Model 2 | Ref | 1.02 (0.96–1.07) | 1.02 (0.96–1.07) | 1.09 (1.03–1.14) | 1.04 (0.99–1.11) | 0.02 |
| Model 3 | Ref | 1.00 (0.95–1.06) | 1.00 (0.95–1.05) | 1.06 (1.00–1.12) | 1.02 (0.97–1.09) | 0.11 |
| Model 4 | Ref | 0.99 (0.94–1.04) | 0.98 (0.92–1.03) | 1.04 (0.98–1.10) | 1.00 (0.92–1.10) | 0.41 |
| Milk | ||||||
| Daily intake, g/day | 35.1 ± 17.8 | 96.9 ± 18.8 | 167.2 ± 23.5 | 276.9 ± 40 | 603.3 ± 264.2 | |
| Cases/person-years | 3,179/320,510 | 3,202/329,166 | 3,109/333,262 | 3,211/334,186 | 3,141/336,897 | |
| Model 1 | Ref | 1.01 (0.96–1.06) | 0.99 (0.94–1.04) | 1.04 (0.99–1.09) | 1.04 (0.98–1.09) | 0.09 |
| Model 2 | Ref | 0.97 (0.92–1.02) | 0.93 (0.89–0.98) | 0.95 (0.90–1.00) | 0.91 (0.86–0.96) | 0.002 |
| Model 3 | Ref | 0.98 (0.93–1.03) | 0.96 (0.91–1.01) | 0.99 (0.94–1.04) | 0.98 (0.93–1.04) | 0.78 |
| Model 4 | Ref | 0.98 (0.93–1.03) | 0.96 (0.91–1.02) | 0.98 (0.92–1.04) | 0.98 (0.90–1.07) | 0.51 |
| Yogurt | ||||||
| Daily intake, g/day | 0 ± 0 | 0.4 ± 1.1 | 12.3 ± 4.7 | 42.4 ± 16.2 | 149.8 ± 85.5 | |
| Cases/person-years | 3,249/314,010 | 3,248/316,639 | 3,231/334,222 | 3,162/341,702 | 2,952/347,449 | |
| Model 1 | Ref | 0.99 (0.94–1.04) | 0.97 (0.92–1.02) | 0.95 (0.90–1.00) | 0.89 (0.85–0.94) | <0.001 |
| Model 2 | Ref | 1.00 (0.95–1.05) | 0.99 (0.94–1.04) | 0.97 (0.92–1.01) | 0.91 (0.86–0.95) | <0.001 |
| Model 3 | Ref | 1.00 (0.95–1.05) | 1.02 (0.97–1.07) | 1.00 (0.96–1.06) | 0.98 (0.93–1.03) | 0.63 |
| Model 4 | Ref | 0.99 (0.95–1.04) | 1.01 (0.96–1.06) | 1.00 (0.95–1.05) | 0.98 (0.90–1.06) | 0.95 |
| Whole grains | ||||||
| Daily intake, g/day | 6.3 ± 3.2 | 17.2 ± 3.1 | 28.2 ± 3.3 | 41.9 ± 4.9 | 78.5 ± 28.2 | |
| Cases/person-years | 3,372/308,523 | 3,193/326,973 | 3,160/334,239 | 3,139/340,953 | 2,978/343,334 | |
| Model 1 | Ref | 0.92 (0.88–0.97) | 0.92 (0.87–0.96) | 0.90 (0.86–0.95) | 0.84 (0.80–0.89) | <0.001 |
| Model 2 | Ref | 0.91 (0.86–0.95) | 0.90 (0.85–0.94) | 0.86 (0.82–0.90) | 0.77 (0.73–0.81) | <0.001 |
| Model 3 | Ref | 0.93 (0.88–0.97) | 0.93 (0.88–0.97) | 0.90 (0.86–0.95) | 0.84 (0.79–0.88) | <0.001 |
| Model 4 | Ref | 0.95 (0.90–1.00) | 0.96 (0.90–1.01) | 0.95 (0.89–1.02) | 0.92 (0.83–1.02) | 0.17 |
| Legumes | ||||||
| Daily intake, g/day | 1.2 ± 1.3 | 6.3 ± 1.6 | 12.7 ± 2.1 | 22.4 ± 3.9 | 58.9 ± 36.6 | |
| Cases/person-years | 3,191/319,639 | 3,158/326,702 | 3,142/331,320 | 3,161/338,060 | 3,190/338,301 | |
| Model 1 | Ref | 0.96 (0.91–1.00) | 0.96 (0.92–1.01) | 0.96 (0.91–1.01) | 0.97 (0.92–1.02) | 0.25 |
| Model 2 | Ref | 0.95 (0.91–1.00) | 0.95 (0.91–1.00) | 0.94 (0.89–0.99) | 0.93 (0.88–0.98) | 0.007 |
| Model 3 | Ref | 0.96 (0.91–1.01) | 0.97 (0.92–1.02) | 0.96 (0.92–1.01) | 0.98 (0.93–1.04) | 0.60 |
| Model 4 | Ref | 0.97 (0.92–1.02) | 0.98 (0.93–1.03) | 0.96 (0.91–1.02) | 0.95 (0.88–1.02) | 0.17 |
| Nuts | ||||||
| Daily intake, g/day | 0 ± 0 | 1.7 ± 0.5 | 4.3 ± 0.8 | 9.9 ± 3.1 | 34.5 ± 21.6 | |
| Cases/person-years | 3,315/317,036 | 3,099/328,827 | 3,266/336,047 | 3,097/337,094 | 3,065/335,019 | |
| Model 1 | Ref | 0.91 (0.87–0.96) | 0.95 (0.90–1.00) | 0.90 (0.86–0.95) | 0.90 (0.85–0.94) | <0.001 |
| Model 2 | Ref | 0.91 (0.86–0.95) | 0.93 (0.89–0.98) | 0.88 (0.83–0.92) | 0.85 (0.81–0.90) | <0.001 |
| Model 3 | Ref | 0.91 (0.87–0.95) | 0.94 (0.89–0.98) | 0.89 (0.85–0.94) | 0.90 (0.86–0.95) | <0.001 |
| Model 4 | Ref | 0.92 (0.87–0.96) | 0.95 (0.90–1.00) | 0.91 (0.86–0.96) | 0.90 (0.83–0.97) | 0.002 |
Data for daily intakes of protein and food sources are shown as mean ± SD. Model 1: adjustment for age, study group indicator, self-identified race/ethnicity, region of residence at baseline, family income, education, and family history of diabetes. Model 2: model 1 adjustments plus smoking status, alcohol intake, physical activity, hormone replacement therapy, multivitamin use, antihypertensive medication use, and total energy intake. Model 3 (full model): model 2 adjustments plus modified AHEI-2010 score and intake of other dietary protein sources. Model 4 (sensitivity model): model 3 adjustments plus BMI and WHR. Ref, reference.
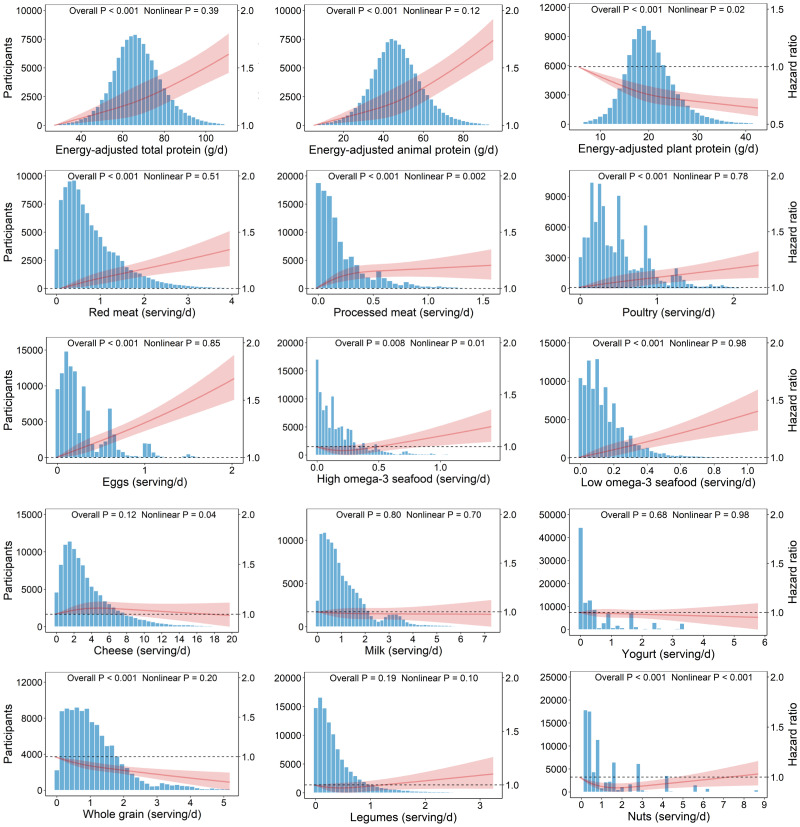
Among the 12 dietary protein sources, intake of red meat, processed meat, poultry, eggs, and low–n-3 seafood was associated with increased T2D risk in the fully adjusted model. Adjusted HRs of T2D for participants in the highest (vs. the lowest) quintile of these protein sources intake was 1.15 (95% CI 1.09–1.22) for red meat, 1.14 (1.08–1.20) for processed meat, 1.09 (1.03–1.15) for poultry, 1.21 (1.14–1.27) for eggs, and 1.12 (1.06–1.18) for low–n-3 seafood. In contrast, intake of whole grains and nuts was inversely associated with T2D risk, with HRs of 0.84 (0.79–0.88) for whole grains and 0.90 (0.86–0.95) for nuts in comparing the two extreme quintiles. Intakes of high–n-3 seafood, cheese, milk, yogurt, and legumes had neutral associations with T2D risk in the fully adjusted model (Table 2). Results of additional restricted cubic spline analyses in the WHI showed nonlinear relations for intake of plant protein, processed meat, high–n-3 seafood, and nuts to T2D risk (all P for overall <0.01 and all P for nonlinearity <0.05) and linear relations for intake of total protein, animal protein, red meat, poultry, eggs, low–n-3 seafood, and whole grains with T2D risk (all P for overall <0.001 and all P for nonlinearity >0.05) (Fig. 1).
Figure 1.
Associations of dietary protein and food sources with risk of incident T2D among 108,681 postmenopausal women in the WHI. Cox proportional hazards models were used for analysis, including restricted cubic spline term for each protein source, with adjustment for age, study group indicator, self-identified race/ethnicity, region of residence, family income, education, family history of diabetes, smoking status, alcohol intake, physical activity, hormone replacement therapy, multivitamin use, antihypertensive medication use, total energy intake, modified AHEI-2010 score, and intake of other dietary protein sources. d, day; serving/d, servings per day.
We further conducted a replication analysis in the UKB of 14,902 men and 19,714 women followed for a median of 11.4 years, during which 663 incident T2D cases were identified (Supplementary Table 3). The positive associations of intake of total protein, red meat, processed meat, eggs, and poultry and the inverse associations of whole grain intake with risk of incident T2D observed in the WHI were confirmed in the UKB. However, the intake of low–n-3 seafood and nuts had no association with risk of incident T2D in the UKB (Supplementary Table 4).
Associations Between Substituting Different Protein Sources and T2D Risk
In the WHI, substituting 5% energy from plant protein for 5% of energy from animal protein was associated with a 21% reduced risk of T2D (HR 0.79 [95% CI 0.74–0.84]). Specifically for protein sources, replacing 1 serving/day red meat (0.87 [0.85–0.90]), processed meat (0.79 [0.75–0.84]), eggs (0.78 [0.75–0.81]), or poultry (0.87 [0.84–0.91]) with plant protein sources, particularly whole grains, was associated with 13–26% decreased risk of T2D (Table 3).
Table 3.
Associations of substitution of different protein and food sources with risk of incident T2D in the WHI and the UKB
| Substituted protein | Equivalent amount of substituted protein | WHI | UKB |
|---|---|---|---|
| 5% of energy from animal protein | Plant protein | 0.79 (0.74–0.84) | NA |
| 1 serving/day red meat | Whole grains | 0.87 (0.85–0.90) | 0.80 (0.71–0.89) |
| Nuts | 0.92 (0.90–0.94) | 0.81 (0.71–0.91) | |
| Legumes | 0.95 (0.91–0.99) | 0.91 (0.75–1.10) | |
| Yogurt | 0.92 (0.89–0.94) | 0.80 (0.71–0.90) | |
| Milk | 0.92 (0.90–0.95) | 0.97 (0.83–1.15) | |
| Cheese | 0.92 (0.90–0.95) | 0.78 (0.65–0.92) | |
| High–n-3 seafood | 0.97 (0.90–1.04) | 0.82 (0.66–1.02) | |
| Low–n-3 seafood | 1.15 (1.06–1.25) | 0.82 (0.68–0.98) | |
| 1 serving/day processed meat | Whole grains | 0.79 (0.75–0.84) | 0.70 (0.59–0.83) |
| Nuts | 0.84 (0.79–0.89) | 0.71 (0.59–0.85) | |
| Legumes | 0.86 (0.81–0.92) | 0.80 (0.62–1.02) | |
| Yogurt | 0.83 (0.79–0.88) | 0.70 (0.59–0.84) | |
| Milk | 0.84 (0.79–0.89) | 0.86 (0.70–1.06) | |
| Cheese | 0.84 (0.80–0.89) | 0.68 (0.55–0.85) | |
| High–n-3 seafood | 0.88 (0.81–0.97) | 0.72 (0.56–0.93) | |
| Low–n-3 seafood | 1.05 (0.95–1.16) | 0.72 (0.57–0.91) | |
| 1 serving/day eggs | Whole grains | 0.78 (0.75–0.81) | 0.84 (0.73–0.96) |
| Nuts | 0.82 (0.79–0.86) | 0.85 (0.73–0.99) | |
| Legumes | 0.85 (0.81–0.89) | 0.96 (0.77–1.19) | |
| Yogurt | 0.82 (0.79–0.85) | 0.84 (0.73–0.97) | |
| Milk | 0.82 (0.79–0.86) | 1.03 (0.86–1.23) | |
| Cheese | 0.83 (0.80–0.86) | 0.82 (0.67–1.00) | |
| High–n-3 seafood | 0.87 (0.80–0.94) | 0.86 (0.67–1.10) | |
| Low–n-3 seafood | 1.03 (0.94–1.13) | 0.86 (0.70–1.07) | |
| 1 serving/day poultry | Whole grains | 0.87 (0.84–0.91) | 0.84 (0.75–0.93) |
| Nuts | 0.92 (0.89–0.96) | 0.84 (0.75–0.96) | |
| Legumes | 0.95 (0.91–1.00) | 0.95 (0.78–1.16) | |
| Yogurt | 0.92 (0.88–0.96) | 0.84 (0.74–0.94) | |
| Milk | 0.93 (0.89–0.96) | 1.02 (0.87–1.20) | |
| Cheese | 0.93 (0.89–0.96) | 0.81 (0.69–0.96) | |
| High–n-3 seafood | 0.97 (0.89–1.06) | 0.86 (0.69–1.07) | |
| Low–n-3 seafood | 1.16 (1.05–1.27) | 0.86 (0.71–1.04) |
Data are HR (95% CI). One serving size was defined as 50 g/day for red meat, processed meat, eggs, high–n-3 seafood, low–n-3 seafood, and legumes; 30 g/day for cheese and whole grains; 200 g/day for milk; 70 g/day for yogurt; and 10 g/day for nuts. In the WHI, the substitution analysis models included adjustment for age, study group indicator, self-identified race/ethnicity, region of residence, family income, education, and family history of diabetes, smoking status, alcohol intake, physical activity, hormone replacement therapy, multivitamin use, antihypertensive medication use, total energy intake, modified AHEI-2010 score, and intake of other protein sources. In the UKB, the substitution analyses included adjustment for age, sex, residence area, Townsend deprivation index at recruitment, smoking status, drinking status, physical activity, family history of diabetes, multivitamin use, antihypertensive medication treatment, total energy intake, and other protein sources.
NA: In the UKB, only total protein was calculated and released. The variables of animal protein and plant protein intake were not available in the UKB database; therefore, the according substitution analysis cannot be conducted.
We also examined the associations between replacing red meat, processed meat, eggs, and poultry with other animal protein sources and risk of T2D. Substituting milk, yogurt, or cheese for red meat, processed meat, eggs, or poultry was associated with decreased risk of T2D. For example, replacing 1 serving/day eggs with 1 serving/day yogurt (HR 0.82 [95% CI 0.79–0.85]), milk (0.82 [0.79–0.86]), or cheese (0.83 [0.80–0.86]) was associated with a 17–18% lower risk of T2D. Substituting high–n-3 seafood for processed meat or eggs was also associated with decreased risk of T2D. The HRs of T2D were 0.88 (95% CI 0.81–0.97) for substituting 1 serving/day high–n-3 seafood for processed meat and 0.87 (0.80–0.94) for substituting 1 serving/day high–n-3 seafood for eggs in the WHI. These findings were consistent with the results of substitution analysis in the UKB (Table 3).
In sensitivity analysis, additional adjustment for BMI and WHR attenuated associations of intakes of dietary protein and food sources with T2D risk, leading most associations toward neutral (model 4 in Table 2 and Supplementary Table 4). Similarly, additional adjustment for BMI and WHR also materially attenuated the beneficial associations of substituting different protein and food sources with T2D risk (Supplementary Table 5).
Mediating Roles of T2D-Related Biomarkers in the Associations Between Substituting Plant Protein for Animal Protein and T2D Risk
We included 1,445 incident T2D case and 2,019 control subjects in the case-control studies nested within the WHI. Compared with the control subjects, the T2D case subjects had higher BMI, waist circumference, HOMA of insulin resistance, inflammation factors (hs-CRP, IL-6, and TNFα-R2), endothelial dysfunction markers (E-selectin, SICAM-1, and VCAM-1), and leptin but had lower SHBG (Supplementary Tables 6 and 7).
The observed association of substituting plant protein for animal protein with decreased risk of T2D was mainly mediated by inflammation factors, leptin, endothelial dysfunction, and SHBG. Specifically, hs-CRP mediated 77% of the total relation of substituting 5% energy from plant protein for 5% energy from animal protein to decreased risk of T2D; IL-6 accounted for 47% of the total relation. We also constructed an inflammation score by summing up inflammation markers above the median value (34). The constructed inflammation score mediated 45% of the total relation. Leptin, a major hormone secreted by adipocytes, mediated 66% of the total relation. SHBG accounted for 46% of the total relation. E-selectin mediated 30% of the total relation (Table 4). After additional adjustment for BMI, almost all mediating effects due to biomarkers were eliminated (Supplementary Table 8).
Table 4.
Mediating effects of biomarkers on the association of isocaloric substitution of plant protein for animal protein with risk of T2D among postmenopausal women enrolled in nested case-control studies in the WHI
| Mediators | Substituting 5% energy from plant protein for 5% energy from animal protein → T2D | ||||
|---|---|---|---|---|---|
| N | Total effect | Indirect effect | Direct effect | Proportion mediated | |
| BMI | 3,151 | 0.78 (0.55–1.07) | 0.73 (0.65–0.81) | 1.06 (0.74–1.52) | NA |
| WHR | 3,175 | 0.80 (0.57–1.14) | 0.83 (0.72–0.95) | 0.97 (0.69–1.39) | 65 |
| Inflammation factors | |||||
| TNFα-R2 | 2,771 | 0.82 (0.59–1.15) | 0.97 (0.90–1.02) | 0.85 (0.61–1.21) | 10 |
| IL-6 | 2,780 | 0.81 (0.57–1.15) | 0.87 (0.79–0.95) | 0.93 (0.65–1.34) | 47 |
| hs-CRP | 2,786 | 0.77 (0.52–1.09) | 0.80 (0.72–0.89) | 0.97 (0.66–1.35) | 77 |
| Inflammation score | 2,769 | 0.79 (0.56–1.12) | 0.88 (0.79–0.97) | 0.91 (0.64–1.27) | 45 |
| Endothelial dysfunction | |||||
| VCAM-1 | 2,778 | 0.84 (0.60–1.19) | 1.01 (0.98–1.05) | 0.82 (0.59–1.17) | NA |
| E-selectin | 2,772 | 0.88 (0.62–1.26) | 0.92 (0.83–1.02) | 0.95 (0.68–1.37) | 30 |
| SICAM-1 | 2,752 | 0.89 (0.65–1.24) | 1.04 (0.95–1.14) | 0.85 (0.61–1.22) | NA |
| Sex steroids and SHBG | |||||
| Estradiol | 1,402 | 0.81 (0.52–1.22) | 0.99 (0.96–1.01) | 0.82 (0.53–1.23) | 2 |
| Testosterone | 1,402 | 0.80 (0.51–1.22) | 0.98 (0.93–1.03) | 0.81 (0.52–1.24) | 4 |
| SHBG | 1,402 | 0.81 (0.50–1.23) | 0.85 (0.74–0.98) | 0.94 (0.59–1.46) | 46 |
| Leptin and leptin receptor | |||||
| Leptin | 1,403 | 0.78 (0.51–1.20) | 0.80 (0.70–0.89) | 0.98 (0.65–1.48) | 66 |
| Soluble leptin receptor | 1,403 | 0.81 (0.54–1.23) | 0.97 (0.92–1.01) | 0.84 (0.55–1.27) | 7 |
| Cellular aging: leukocyte telomere length | 3,101 | 0.80 (0.58–1.09) | 1.00 (0.98–1.01) | 0.80 (0.58–1.10) | 1 |
Data are OR (95% CI) or % unless otherwise indicated. NA: proportion mediated was not calculated when the point estimate of the direct effect was in a direction oppositive to that of the indirect effect. The covariates adjusted for in the mediation analyses included age, ethnicity, education, smoking, drinking, and physical activity. We calculated inflammation score by summing up inflammation markers above the median value.
Conclusions
In two large prospective cohorts of the WHI and the UKB, we found that intake of animal protein sources from red meat, processed meat, poultry, or eggs was directly associated with increased T2D risk, while plant protein sources from whole grains or nuts were associated with decreased risk. Substituting plant protein sources for animal protein sources showed benefits in reducing T2D risk, which was mainly mediated by inflammation, followed by endothelial dysfunction and SHBG.
An accumulating body of evidence now indicates that red meat (9,35), processed meat (9,36), or eggs (10) may increase T2D risk, whereas whole grains (37,38) and dairy products (39) may decrease risk. In the current study, the inverse associations of milk or yogurt intake with T2D risk in the WHI became null after adjustment for the other dietary protein sources. A previous study in the WHI reported that a diet high in low-fat dairy products, but not high-fat dairy products, was associated with lower T2D risk in postmenopausal women, indicating that fat in dairy products may distort the dairy-T2D relation (40). Our analysis in the UKB, however, indicates that milk intake appeared to be associated with increased T2D risk. Further work is warranted to clarify these inconsistent findings, particularly in diverse populations where different T2D risk and dietary practices may be associated with dairy intake.
In both the WHI and the UKB, intake of poultry was associated with increased T2D risk, which appeared to be in contrast with null findings of a recent meta-analysis of 28 prospective studies regarding poultry and fish intake (41). In contrast, in the China Kadoorie Biobank (CKD) study of ∼0.5 million Chinese adults, published after the aforementioned meta-analysis, findings showed no association between poultry intake and T2D and a positive association between total fish intake and T2D (35). In our study, we found that intake of low–n-3 seafood, but not high–n-3 seafood, was associated with increased risk of T2D, suggesting that the association between seafood intake and T2D risk may be dependent on n-3 content in seafood, which may explain the discrepancy in reported associations with fish intake.
Additionally, our substitution analysis confirmed the reduction of T2D risk associated with replacing animal protein sources, such as red meat, processed meat, poultry, or eggs, with plant protein sources, milk, yogurt, cheese, or high–n-3 seafood. These findings are consistent with the previous studies showing beneficial associations of protein sources substitution with T2D risk, although which focused mainly on the replacement of red meat with other foods (42–44). Our findings are also consistent with previous research showing that adherence to a plant-based dietary pattern may lower T2D risk (45) and cardiovascular disease (46) and support the 2021 dietary guidelines for improving cardiovascular health from the American Heart Association (47). These observed protein-T2D associations were materially attenuated if we additionally adjusted for BMI and WHR. Dietary data collected with an FFQ reflect one’s long-term nutritional status. In our study, dietary intake and BMI were assessed at baseline simultaneously. The possibilities of BMI being a cause and effect of the measured dietary intake exist. Therefore, BMI could be both a confounder and mediator in the protein-T2D relation; hence, the additional adjustment for BMI and WHR that attenuated the protein-T2D association.
The mechanisms underlying the observed beneficial association between substituting plant protein sources for animal protein sources and T2D risk remain to be determined. Previous studies have implicated red meat intake in accumulation of abdominal fat (48,49) and higher serum ferritin, amino acids, and lipid metabolites (42). In mediation analysis, we examined well-characterized T2D-related biomarkers, including inflammation factors, endothelial dysfunction, sex steroids and SHBG, leptin and its soluble receptor, and cellular aging (24). Our isocaloric substitution analysis indicated T2D risk reduction to be associated with replacing 5% energy from animal protein with plant protein, which was mediated mainly by inflammation factors, followed by leptin, endothelial dysfunction biomarkers, and SHBG. Recent advances in gut microbiota also support the mediating role of inflammation in the dietary protein–T2D association. Dietary factors can modulate the gut microbial composition and then alter intestinal permeability, leading to lipopolysaccharides leakage and inflammatory activation through toll-like receptors (50). In addition, intake of red meat, which is particularly high in l-carnitine, was associated with elevated levels of inflammatory trimethylamine N-oxide, which may increase risk of cardiometabolic diseases (51).
The mediating effects of these biomarkers were decreased after additional adjustment for BMI in the mediation analyses. This is consistent with other reports indicating the mediating and confounding roles of BMI in the relations of inflammation and leptin to metabolic diseases (52,53). At a population level, BMI is a reliable measure that captures the biological actions of adipose tissues, including inflammatory processes and leptin’s action. Therefore, it is no surprise that the proportion mediated by inflammation and leptin diminished with additional adjustment for BMI in our study.
The current study has several strengths, including analysis of a large prospective national cohort of multiethnic postmenopausal women with long-term follow-up, with replication in a large independent cohort of men and women; a comprehensive investigation of 12 major sources of dietary protein, with possible substitution combinations—which is beneficial for identifying optimal T2D dietary intervention strategies; and nested case-control studies in the WHI allowing for the comprehensive examination of the potential mechanistic mediators that may explain the specific protein-T2D relations observed. Our findings revealed novel mechanisms underlying the beneficial association between substituting plant protein sources for animal protein sources and decreased risk of T2D, which is helpful for the development of a mechanism-based dietary intervention strategy for the prevention and management of T2D.
Nevertheless, there are limitations that should be considered in interpreting our findings. First, self-reported dietary data are prone to measurement error (both random and systematic) (54), even with extensive validation effort of FFQ with use of biomarkers in WHI and multiple records used in the UKB to reduce errors. One has to assume that participants’ dietary habits did not change materially during the follow-up; otherwise, the occurrence of such changes could introduce bias. Second, the observed beneficial associations of food substitution with T2D were simulated based on statistical modeling, which may be different from interventions with real foods in an experimental setting. Third, although our analyses included adjustment for known confounding factors, some residual confounding could still exist. For example, WHI dietary data were collected in the 1990s, while UKB’s were from the 2010s, which may lead to higher exposure to ultraprocessed foods (UPFs) in the more recent UKB as sales of UPFs have been increasing globally (55–57). Given lack of adjustment for UPFs in our analyses, there may have been differences in the estimated associations between the WHI and the UKB. Finally, the majority of both the WHI and UKB participants were White, which may limit the generalizability of our findings to other populations, although a relatively homogenous population does enhance the internal validity of the relations observed.
In conclusion, in these two large prospective cohort studies, intake of animal protein sources, such as red meat, processed meat, poultry, and eggs, was associated with increased T2D risk, whereas intake of plant protein sources, especially whole grains and nuts, was associated with decreased risk. Substituting the unfavorable animal protein sources as determined in this study with plant protein sources, milk, yogurt, cheese, or high–n-3 seafood was associated with lower risk of T2D. The beneficial association of isocaloric substitution of plant protein for animal protein was mechanistically mediated mainly by obesity-related inflammation. These findings support the recommendation that dietary protein sources should be given attention for the prevention of T2D.
Article Information
Acknowledgments. The authors appreciate all WHI and UKB participants and all staff for their contribution to these studies.
Funding. J.L. was supported by the National Natural Science Foundation of China (81673156, 82073528, and 81302417). S.L. was partly supported by the National Institutes of Health (NIH) grants R01DK125403, R01ES031391, and R01ES029082. J.B. was supported by NIH grant R01DK127916. A.J.G. was supported by the Canadian Institute of Health Research, CIHR Fellowship. The WHI program is funded by the National Heart, Lung, and Blood Institute, National Institutes of Health, U.S. Department of Health and Human Services through contracts 75N92021D00001, 75N92021D00002, 75N92021D00003, 75N92021D00004, and 75N92021D00005.
The funders had no role in the study design, data collection, data analysis, data interpretation, or writing of the manuscript.
Duality of Interest. A.J.G. has received consulting fees from Solo GI Nutrition and has received an honorarium from the Soy Nutrition Institute. No other potential conflicts of interest relevant to this article were reported.
Author Contributions. J.L. and S.L. designed the research. J.L., A.J.G., Q.Y., D.D. and L.Z. conducted statistical analysis. J.L. and S.L. led the writing of the manuscript. All authors contributed to the interpretation of the results and critical revision of the manuscript for important intellectual content and approved the final version of the manuscript. J.L. and S.L. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains supplementary material online at https://doi.org/10.2337/figshare.19860148.
This article is featured in a podcast available at diabetesjournals.org/journals/pages/diabetes-core-update-podcasts.
J.L. and A.J.G. contributed equally to this work.
References
- 1. Hu FB, Manson JE, Stampfer MJ, et al. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N Engl J Med 2001;345:790–797 [DOI] [PubMed] [Google Scholar]
- 2. Khazrai YM, Defeudis G, Pozzilli P. Effect of diet on type 2 diabetes mellitus: a review. Diabetes Metab Res Rev 2014;30(Suppl. 1):24–33 [DOI] [PubMed] [Google Scholar]
- 3. Ludwig DS, Willett WC, Volek JS, Neuhouser ML. Dietary fat: From foe to friend? Science 2018;362:764–770 [DOI] [PubMed] [Google Scholar]
- 4. Sluijs I, Beulens JW, van der A DL, Spijkerman AM, Grobbee DE, van der Schouw YT. Dietary intake of total, animal, and vegetable protein and risk of type 2 diabetes in the European Prospective Investigation into Cancer and Nutrition (EPIC)-NL study. Diabetes Care 2010;33:43–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Malik VS, Li Y, Tobias DK, Pan A, Hu FB. Dietary protein intake and risk of type 2 diabetes in US men and women. Am J Epidemiol 2016;183:715–728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Viguiliouk E, Glenn AJ, Nishi SK, et al. Associations between dietary pulses alone or with other legumes and cardiometabolic disease outcomes: an umbrella review and updated systematic review and meta-analysis of prospective cohort studies. Adv Nutr 2019;10(Suppl. 4):S308–S319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Pearce M, Fanidi A, Bishop TRP, et al.; EPIC-InterAct Consortium . Associations of total legume, pulse, and soy consumption with incident type 2 diabetes: federated meta-analysis of 27 studies from diverse world regions. J Nutr 2021;151:1231–1240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Zhao LG, Zhang QL, Liu XL, Wu H, Zheng JL, Xiang YB. Dietary protein intake and risk of type 2 diabetes: a dose-response meta-analysis of prospective studies. Eur J Nutr 2019;58:1351–1367 [DOI] [PubMed] [Google Scholar]
- 9. Pan A, Sun Q, Bernstein AM, Manson JE, Willett WC, Hu FB. Changes in red meat consumption and subsequent risk of type 2 diabetes mellitus: three cohorts of US men and women. JAMA Intern Med 2013;173:1328–1335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Drouin-Chartier JP, Schwab AL, Chen S, et al. Egg consumption and risk of type 2 diabetes: findings from 3 large US cohort studies of men and women and a systematic review and meta-analysis of prospective cohort studies. Am J Clin Nutr 2020;112:619–630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Ford ES, Williamson DF, Liu S. Weight change and diabetes incidence: findings from a national cohort of US adults. Am J Epidemiol 1997;146:214–222 [DOI] [PubMed] [Google Scholar]
- 12. Liu S, Tinker L, Song Y, et al. A prospective study of inflammatory cytokines and diabetes mellitus in a multiethnic cohort of postmenopausal women. Arch Intern Med 2007;167:1676–1685 [DOI] [PubMed] [Google Scholar]
- 13. Song Y, Manson JE, Tinker L, et al. Circulating levels of endothelial adhesion molecules and risk of diabetes in an ethnically diverse cohort of women. Diabetes 2007;56:1898–1904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ding EL, Song Y, Malik VS, Liu S. Sex differences of endogenous sex hormones and risk of type 2 diabetes: a systematic review and meta-analysis. JAMA 2006;295:1288–1299 [DOI] [PubMed] [Google Scholar]
- 15. Ding EL, Song Y, Manson JE, et al. Sex hormone-binding globulin and risk of type 2 diabetes in women and men. N Engl J Med 2009;361:1152–1163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Chen BH, Brennan K, Goto A, et al. Sex hormone-binding globulin and risk of clinical diabetes in American black, Hispanic, and Asian/Pacific Islander postmenopausal women. Clin Chem 2012;58:1457–1466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Liu S, Sun Q. Sex differences, endogenous sex-hormone hormones, sex-hormone binding globulin, and exogenous disruptors in diabetes and related metabolic outcomes. J Diabetes 2018;10:428–441 [DOI] [PubMed] [Google Scholar]
- 18. You NC, Chen BH, Song Y, et al. A prospective study of leukocyte telomere length and risk of type 2 diabetes in postmenopausal women. Diabetes 2012;61:2998–3004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Shen Q, Zhao X, Yu L, et al. Association of leukocyte telomere length with type 2 diabetes in mainland Chinese populations. J Clin Endocrinol Metab 2012;97:1371–1374 [DOI] [PubMed] [Google Scholar]
- 20. Azadbakht L, Kimiagar M, Mehrabi Y, Esmaillzadeh A, Hu FB, Willett WC. Soy consumption, markers of inflammation, and endothelial function: a cross-over study in postmenopausal women with the metabolic syndrome. Diabetes Care 2007;30:967–973 [DOI] [PubMed] [Google Scholar]
- 21. Barak F, Falahi E, Keshteli AH, Yazdannik A, Saneei P, Esmaillzadeh A. Red meat intake, insulin resistance, and markers of endothelial function among Iranian women. Mol Nutr Food Res 2015;59:315–322 [DOI] [PubMed] [Google Scholar]
- 22. Brinkman MT, Baglietto L, Krishnan K, et al. Consumption of animal products, their nutrient components and postmenopausal circulating steroid hormone concentrations. Eur J Clin Nutr 2010;64:176–183 [DOI] [PubMed] [Google Scholar]
- 23. Zhou M, Zhu L, Cui X, et al. Influence of diet on leukocyte telomere length, markers of inflammation and oxidative stress in individuals with varied glucose tolerance: a Chinese population study. Nutr J 2016;15:39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Song Y, Huang YT, Song Y, et al. Birthweight, mediating biomarkers and the development of type 2 diabetes later in life: a prospective study of multi-ethnic women. Diabetologia 2015;58:1220–1230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Patterson RE, Kristal AR, Tinker LF, Carter RA, Bolton MP, Agurs-Collins T. Measurement charac-teristics of the Women’s Health Initiative food frequency questionnaire. Ann Epidemiol 1999;9:178–187 [DOI] [PubMed] [Google Scholar]
- 26. Bowman SA, Friday JE, Moshfegh AJ. MyPyramid Equivalents Database, 2.0 for USDA Survey Foods, 2003-2004. U.S. Department of Agriculture, 2008. Accessed 13 June 2022. Available from https://www.ars.usda.gov/ARSUserFiles/80400530/pdf/mped/mped2_doc.pdf
- 27. For the WHI FFQ: My Pyramid Equivalents Database 2.0 (MPED 2.0). Accessed 13 June 2022. Available from https://www.whi.org/doc/f60_MPEDs_v2_ReadMe_inv-FINAL.pdf
- 28. Halkjaer J, Olsen A, Bjerregaard LJ, et al. Intake of total, animal and plant proteins, and their food sources in 10 countries in the European Prospective Investigation into Cancer and Nutrition. Eur J Clin Nutr 2009;63(Suppl. 4):S16–S36 [DOI] [PubMed] [Google Scholar]
- 29. Margolis KL, Lihong Qi, Brzyski R, et al.; Women Health Initiative Investigators . Validity of diabetes self-reports in the Women’s Health Initiative: comparison with medication inventories and fasting glucose measurements. Clin Trials 2008;5:240–247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Chiuve SE, Fung TT, Rimm EB, et al. Alternative dietary indices both strongly predict risk of chronic disease. J Nutr 2012;142:1009–1018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Langer RD, White E, Lewis CE, Kotchen JM, Hendrix SL, Trevisan M. The Women’s Health Initiative Observational Study: baseline characteristics of participants and reliability of baseline measures. Ann Epidemiol 2003;13(Suppl.):S107–S121 [DOI] [PubMed] [Google Scholar]
- 32. VanderWeele TJ. Mediation analysis: a practitioner’s guide. Annu Rev Public Health 2016;37:17–32 [DOI] [PubMed] [Google Scholar]
- 33. Gong JH, Lo K, Liu Q, et al. Dietary manganese, plasma markers of inflammation, and the development of type 2 diabetes in postmenopausal women: findings from the Women’s Health Initiative. Diabetes Care 2020;43:1344–1351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Duncan BB, Schmidt MI, Pankow JS, et al.; Atherosclerosis Risk in Communities Study . Low-grade systemic inflammation and the development of type 2 diabetes: the Atherosclerosis Risk in Communities study. Diabetes 2003;52:1799–1805 [DOI] [PubMed] [Google Scholar]
- 35. Du H, Guo Y, Bennett DA, et al.; China Kadoorie Biobank collaborative group . Red meat, poultry and fish consumption and risk of diabetes: a 9 year prospective cohort study of the China Kadoorie Biobank. Diabetologia 2020;63:767–779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Ericson U, Sonestedt E, Gullberg B, et al. High intakes of protein and processed meat associate with increased incidence of type 2 diabetes. Br J Nutr 2013;109:1143–1153 [DOI] [PubMed] [Google Scholar]
- 37. Aune D, Norat T, Romundstad P, Vatten LJ. Whole grain and refined grain consumption and the risk of type 2 diabetes: a systematic review and dose-response meta-analysis of cohort studies. Eur J Epidemiol 2013;28:845–858 [DOI] [PubMed] [Google Scholar]
- 38. Parker ED, Liu S, Van Horn L, et al. The association of whole grain consumption with incident type 2 diabetes: the Women’s Health Initiative Observational Study. Ann Epidemiol 2013;23:321–327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Gijsbers L, Ding EL, Malik VS, de Goede J, Geleijnse JM, Soedamah-Muthu SS. Consumption of dairy foods and diabetes incidence: a dose-response meta-analysis of observational studies. Am J Clin Nutr 2016;103:1111–1124 [DOI] [PubMed] [Google Scholar]
- 40. Margolis KL, Wei F, de Boer IH, et al.; Women’s Health Initiative Investigators . A diet high in low-fat dairy products lowers diabetes risk in postmenopausal women. J Nutr 2011;141:1969–1974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Yang X, Li Y, Wang C, et al. Meat and fish intake and type 2 diabetes: dose-response meta-analysis of prospective cohort studies. Diabetes Metab 2020;46:345–352 [DOI] [PubMed] [Google Scholar]
- 42. Ibsen DB, Steur M, Imamura F, et al. Replacement of red and processed meat with other food sources of protein and the risk of type 2 diabetes in European Populations: the EPIC-InterAct Study. Diabetes Care 2020;43:2660–2667 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Würtz AML, Jakobsen MU, Bertoia ML, et al. Replacing the consumption of red meat with other major dietary protein sources and risk of type 2 diabetes mellitus: a prospective cohort study. Am J Clin Nutr 2021;113:612–621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Ibsen DB, Jakobsen MU, Halkjær J, et al. Replacing red meat with other nonmeat food sources of protein is associated with a reduced risk of type 2 diabetes in a Danish cohort of middle-aged adults. J Nutr 2021;151:1241–1248 [DOI] [PubMed] [Google Scholar]
- 45. Chen Z, Drouin-Chartier JP, Li Y, et al. Changes in plant-based diet indices and subsequent risk of type 2 diabetes in women and men: three U.S. prospective cohorts. Diabetes Care 2021;44:663–671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Glenn AJ, Lo K, Jenkins DJA, et al. Relationship between a plant-based dietary portfolio and risk of cardiovascular disease: findings from the Women’s Health Initiative prospective cohort study. J Am Heart Assoc 2021;10:e021515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Lichtenstein AH, Appel LJ, Vadiveloo M, et al. 2021 dietary guidance to improve cardiovascular health: a scientific statement from the American Heart Association. Circulation 2021;144:e472–e487 [DOI] [PubMed] [Google Scholar]
- 48. Mazidi M, Kengne AP, George ES, Siervo M. The association of red meat intake with inflammation and circulating intermediate biomarkers of type 2 diabetes is mediated by central adiposity. Br J Nutr 2021;125:1043–1050 [DOI] [PubMed] [Google Scholar]
- 49. Chen Z, Franco OH, Lamballais S, et al. Associations of specific dietary protein with longitudinal insulin resistance, prediabetes and type 2 diabetes: The Rotterdam Study. Clin Nutr 2020;39:242–249 [DOI] [PubMed] [Google Scholar]
- 50. de Vos WM, Tilg H, Van Hul M, Cani PD. Gut microbiome and health: mechanistic insights. Gut 2022;71:1020–1032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Abbasi J. TMAO and heart disease: the new red meat risk? JAMA 2019;321:2149–2151 [DOI] [PubMed] [Google Scholar]
- 52. Duncan BB, Schmidt MI, Chambless LE, Folsom AR, Carpenter M, Heiss G. Fibrinogen, other putative markers of inflammation, and weight gain in middle-aged adults--the ARIC study. Atherosclerosis Risk in Communities. Obes Res 2000;8:279–286 [DOI] [PubMed] [Google Scholar]
- 53. Schmidt MI, Duncan BB, Vigo A, et al.; ARIC Investigators . Leptin and incident type 2 diabetes: risk or protection? Diabetologia 2006;49:2086–2096 [DOI] [PubMed] [Google Scholar]
- 54. Tinker LF, Sarto GE, Howard BV, et al. Biomarker-calibrated dietary energy and protein intake associations with diabetes risk among postmenopausal women from the Women’s Health Initiative. Am J Clin Nutr 2011;94:1600–1606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Levy RB, Rauber F, Chang K, et al. Ultra-processed food consumption and type 2 diabetes incidence: a prospective cohort study. Clin Nutr 2021;40:3608–3614 [DOI] [PubMed] [Google Scholar]
- 56. Llavero-Valero M, Escalada-San Martín J, Martínez-González MA, Basterra-Gortari FJ, de la Fuente-Arrillaga C, Bes-Rastrollo M. Ultra-processed foods and type-2 diabetes risk in the SUN project: a prospective cohort study. Clin Nutr 2021;40:2817–2824 [DOI] [PubMed] [Google Scholar]
- 57. Srour B, Fezeu LK, Kesse-Guyot E, et al. Ultraprocessed food consumption and risk of type 2 diabetes among participants of the NutriNet- Santé prospective cohort. JAMA Intern Med 2020;180:283–291 [DOI] [PMC free article] [PubMed] [Google Scholar]