Abstract
Background
Capillary refill time (CRT) is a valuable tool for triage and to guide resuscitation. However, little is known about CRT kinetics after fluid infusion.
Methods
We conducted a prospective observational study in a tertiary teaching hospital. First, we analyzed the intra-observer variability of CRT. Next, we monitored fingertip CRT in sepsis patients during volume expansion within the first 24 h of ICU admission. Fingertip CRT was measured every 2 min during 30 min following crystalloid infusion (500 mL over 15 min).
Results
First, the accuracy of repetitive fingertip CRT measurements was evaluated on 40 critically ill patients. Reproducibility was excellent, with an intra-class correlation coefficient of 99.5% (CI 95% [99.3, 99.8]). A CRT variation larger than 0.2 s was considered as significant. Next, variations of CRT during volume expansion were evaluated on 29 septic patients; median SOFA score was 7 [5–9], median SAPS II was 57 [45–72], and ICU mortality rate was 24%. Twenty-three patients were responders as defined by a CRT decrease > 0.2 s at 30 min after volume expansion, and 6 were non-responders. Among responders, we observed that fingertip CRT quickly improved with a significant decrease at 6–8 min after start of crystalloid infusion, the maximal improvement being observed after 10–12 min (−0.7 [−0.3;−0.9] s) and maintained at 30 min. CRT variations significantly correlated with baseline CRT measurements (R = 0.39, P = 0.05).
Conclusions
CRT quickly improved during volume expansion with a significant decrease 6–8 min after start of fluid infusion and a maximal drop at 10–12 min.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13613-022-01049-x.
Keywords: Sepsis, Kinetics, Fluid challenge, Capillary time refill, Intensive care medicine
Introduction
Peripheral tissue hypoperfusion has been identified as a powerful predictor of poor outcomes in patients suffering from severe conditions, including sepsis [1], cardiac arrest [2] or cardiogenic shock [3]. International guidelines highlighted that the evaluation of peripheral tissue perfusion is of paramount importance to identify shock patients [4]. Peripheral tissue perfusion could be evaluated at the bedside with easy-to-use and easy-to-learn tools, either semi-quantitative such as the mottling score, or quantitative, including skin temperature and the capillary refill time (CRT) [5].
The capillary refill time (CRT) measures the time necessary for the skin to return to baseline color after applying pressure on soft tissue (generally the fingertip). Interrater variability of CRT was weak in non-trained physicians [6] but is very good after standardization in centers expert in peripheral tissue perfusion evaluation [1, 7]. CRT is a valuable tool to assess the severity of an acute illness at both early and late stages. In the emergency ward, persistent prolonged fingertip CRT (> 3 s) is associated with more severe organ failure, more frequent use of organ support therapy and ultimately, a higher in-ICU mortality [8]. In the intensive care unit, in a mixed critically ill population, Lima et al. have reported that a prolonged CRT (> 4.5 s on the index finger) was associated with hyperlactatemia and high SOFA score [9]. Finally, in septic shock patients, persistent prolonged finger CRT after resuscitation is predictive of 14-day mortality, with an Area Under Curve of 84%. A 2.4 s threshold value predicted mortality with good sensitivity (82%, 95% CI [60–95]) and specificity (73%, 95% CI [56–86]) [1]. More recently, the ANDROMEDA-SHOCK trial provided convincing evidence that CRT can be used to guide treatment and resuscitation [10]. In septic shock patients, a strategy based on CRT monitoring led to more important organ failure recovery than an approach based on lactate clearance associated with improved survival. In this trial, the CRT was measured every 30 min but the precise kinetics of CRT after therapeutic intervention is not known. This study aimed to analyze the kinetics of CRT variations after a fluid challenge in sepsis patients.
Methods
Study design and measurements
We conducted a prospective observational study in an 18-bed ICU in a tertiary teaching hospital. During 3 months, all consecutive patients, older than 18 years, to whom the attending intensivist decided to administer a volume expansion were screened. Patients with a prolonged CRT (> 2.5 s) within the first 24 h of ICU admission were included. In addition, to limit confounders, we focused on patients with sepsis with or without shock according to the Third International Consensus Definitions [11]. Patients with dark skin for whom accurate clinical evaluation of CRT was not possible were excluded. During the kinetic study, fingertip CRT was measured twice and the mean value was recorded. CRT was measured every 2 min for 20 min and finally at 25 and 30 min by one single physician. CRT measurements started at the same time that crystalloid infusion was initiated. As previously reported and standardized by our group, CRT was measured by applying firm pressure to the distal phalanx of the index finger for 15 s. The pressure applied was just enough to remove the blood at the tip of the physician’s nail, which was illustrated by the appearance of a thin white distal crescent (blanching) under the nail. A chronometer recorded the time for the return to the baseline color [1]. Volume expansion was standardized with the infusion of 500 mL of crystalloids (saline or ringer lactate) over 15 min.
Patient management and data collection
Patients were admitted directly from the emergency department or medical wards. Circulatory support was guided by our local protocol, adapted from international guidelines [12]. Initial therapeutic management includes antibiotic administration, fluid infusion (30 mL/Kg), norepinephrine infusion to maintain a mean arterial pressure (MAP) > 65 mmHg and infection source control when available. All patients were investigated with transthoracic echocardiography (Vivid 7 Dimension’06, GE Healthcare®) to assess left ventricular function, volemia and cardiac output. Repetitive transthoracic echocardiography was performed routinely during acute circulatory failure management. A fluid infusion was decided by the physician in charge of the patient and was based on several parameters as indicated by international guidelines [13].
General characteristics of the patients were recorded: demographic data, diagnoses, severity of illness evaluated by the Sequential Organ Failure Assessment (SOFA) score [14] and Simplified Acute Physiology Score II (SAPS II) [15]. We collected MAP, heart rate (HR) and cardiac index. Tissue and organ perfusion were assessed through arterial lactate level, urinary output, mottling score, skin temperature, and fingertip CRT.
Statistical analysis
Patient characteristics were summarized as mean ± standard deviation, median (25th–75th percentiles) for skewed distributions and percentages as appropriate. Differences between groups were compared using the Mann–Whitney test or Wilcoxon’s test. Correlations were computed using Pearson’s formula.
CRT was measured to the nearest tenth of a second. Two measurements were obtained for each patient. Reproducibility (variation due to measurement method) was assessed by the intra-class correlation coefficient (ICC), defined as the ratio between-individual variance to the sum of between- and within-individual variance. An ICC close to 1 shows that all variance is explained by between-individual variation and not by within-individual the total variance. ICC was obtained from random-effect ANOVA (R package multilevel). Confidence intervals were obtained by (patient-level) bootstrap.
Statistical significance was set at P < 0.05. All analyses were made using the R software (v 2.12.0; http://cran.r-project.org).
The ethical committee of the French Intensive Care Society (FICS) approved the protocol (CE SRLF 22-009). (CE SRLF 22-009). This is an observational study without any specific intervention. Volume expansion was decided by the physician in charge of the patient and repetitive CRT measurements were recorded. Repetitive CRT measurements during fluid infusion are routinely performed in our ICU.
Results
Intra-reader reproducibility of the Capillary Refill Time
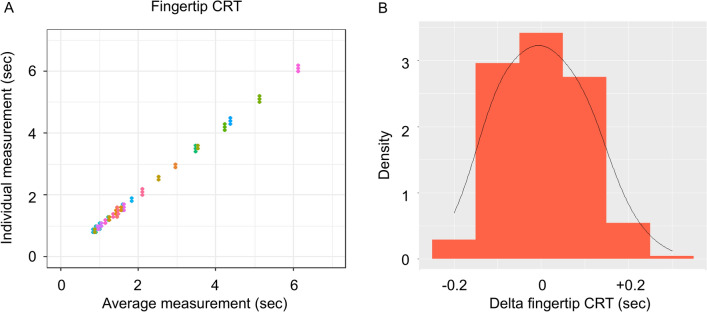
We have previously reported an excellent inter-reader reproducibility of CRT measurements [1]. Given that one physician monitored CRT kinetics for each patient, we first analyzed the intra-reader reproducibility of CRT with two physicians who participated in this study, both having experience in CRT measurements and peripheral tissue evaluation. Forty critically ill patients were prospectively included in this first study and 3 fingertip CRT measurements were done by each physician. The reproducibility of fingertip CRT was high (Fig. 1A). The ICC was 99.5% (CI 95% [99.3, 99.8]), suggesting that 0.5% of the total variance was due to measurement and the rest to patient peripheral perfusion change. Intra-reader standard deviation of the fingertip CRT was 0.07 s. Therefore, we assumed that a CRT variation larger than 0.2 s was significant [16] (Fig. 1B).
Fig. 1.
A Individual and patient-averaged repeated fingertip CRT measurements at a single timepoint (seconds). Data are color-coded by patient. B Distribution of variations of fingertip CRT measurements (seconds)
Studied population
Next, we investigated prospectively kinetics of fingertip CRT variations induced by a fluid challenge. Over 3 months, 68 patients received fluid expansion during the first 24 h of ICU admission. 37 patients were excluded, 18 patients with no sepsis, 8 patients, because accurate CRT measurements were not possible (dark skin), 11 patients with CRT < 2.5 s, leaving 31 patients for the study. Among them, 25 patients were responders as defined by a CRT decrease > 0.2 s at 30 min after fluid challenge, and 6 were non-responders. Finally, 2 responders were excluded, because vasopressor dose was increased during the fluid challenge leaving 23 responders for analysis (Additional file 1). Baseline characteristics are summarized in Table 1. All patients had sepsis without (N = 15/29) or with shock (N = 14/29). The main causes were pneumonia (41%) and abdominal infection (27%). Disease severity was more pronounced in responders with higher SOFA score at admission (5 [4–6] versus 7 [5–10], P = 0.03) and more frequent use of invasive mechanical ventilation (17% versus 61%, P = 0.05). Baseline fingertip CRT was longer in responders (2.8 [2.5–3.4] versus 3.8 [2.6–4.1] sec, P = 0.02). In ICU mortality rate was 24%.
Table 1.
Baseline characteristics of studied population. Parameters and SOFA (Sequential Organ Failure Assessment) were reported at inclusion
| Total N = 29 | Non-responders N = 6 | Responders N = 23 | P value | |
|---|---|---|---|---|
| Age, years | 67 [57–71] | 67 [57–68] | 67 [57–71] | 0.85 |
| Male gender | 21 (72.4) | 3 (50) | 18 (78) | 0.30 |
| Body mass index, kg/m2 | 25 [22–28] | 25 [23–28] | 25 [19–29] | 0.92 |
| SAPS II | 57 [45–72] | 46 [44–47] | 61 [50–78] | 0.10 |
| SOFA | 7 [5–9] | 5 [4–6] | 7 [5–10] | 0.03 |
| Comordibities | ||||
| Cardiovascular disease | 5 (17) | 1 (17) | 4 (17) | > 0.99 |
| Hypertension | 18 (62) | 2 (33) | 16 (69) | 0.24 |
| Cirrhosis | 0 | 0 | 0 | – |
| Diabetes mellitus | 12 (41) | 1 (17) | 11 (48) | 0.36 |
| Chronic kidney disease | 5 (17) | 0 (0) | 4 (17) | 0.27 |
| Malignancy | 12 (41) | 3 (67) | 9 (39) | 0.63 |
| Sepsis | ||||
| Proven | 21 (72) | 4 (67) | 17 (74) | |
| Suspected | 8 (35) | 2 (33) | 6 (26) | 0.72 |
| Heart rate, bpm | 109 [95–127] | 124 [89–152] | 106 [95–125] | 0.34 |
| MAP, mmHg | 71 [67–82] | 74 [70–79] | 71 [68–83] | 0.94 |
| Cardiac index, mL/min/m2 | 3.4 [2.7–4.5] | 3.4 [2.6–4.4] | 3.4 [2.6–4.6] | 0.84 |
| CRT fingertip, sec | 3.4 [2.5–4.0] | 2.8 [2.5–3.4] | 3.8 [2.6–4.1] | 0.02 |
| Mottling score | 2 [1–3] | 1.5 [0.75–3.25] | 2 [1–3] | 0.51 |
| Skin temperature, °C | 29.8 [29.2–31.6] | 29.8 [28.8–31] | 29.9 [29.2–31.9] | 0.89 |
| Mechanical ventilation, n (%) | 15 (52) | 1 (17) | 14 (61) | 0.05 |
| Crystalloid infused before inclusion (mL) | 1500 [1000–2500] | 1500 [1000–1800] | 1500 [1000–2200] | 0.67 |
| Norepinephrine | ||||
| Yes, n (%) | 14 (48) | 2 (33) | 12 (52) | 0.41 |
| Dose, μg/kg/min | 0 [0–0.40] | 0 [0–0.38] | 0.10 [0–0.40] | 0.53 |
| Biological parameters | ||||
| Arterial pH | 7.31 [7.26–7.44] | 7.29 [7.10–7.40] | 7.32 [7.27–7.44] | 0.36 |
| Arterial lactate, mmol/L | 2.4 [1.4–3.3] | 2.3 [1.3–3.4] | 2.4 [1.3–3.8] | 0.80 |
| Leukocyte count, G/L | 13.4 [6.6–18.8] | 10.6 [5.5–18.6] | 13.4 [7.0–19.3] | 0.72 |
| Hemoglobin, g/dL | 13.1 [10.3–13.8] | 11.7 [9.2–13.7] | 13.3 [11–13.9] | 0.29 |
| Platelet count, G/L | 172 [105–233] | 161 [107–289] | 172 [105–233] | 0.84 |
| Serum creatinin, μmol/L | 127 [96–189] | 104 [99–117] | 131 [84–241] | 0.18 |
| Sodium, mmol/L | 136 [130–139] | 137 [130–140] | 136 [129–139] | 0.94 |
| Protidemia, g/L | 58 [49–62] | 59 [49–62] | 55 [49–63] | 0.98 |
| Bilirubin, μmol/L | 14 [10–18] | 15 [9–27] | 14 [10–19] | 0.85 |
| C reactive protein, mg/L | 137 [30–390] | 161 [75–282] | 100 [4–398] | 0.76 |
| Procalcitonin, ng/L | 10.3 [2.9–65] | 13.5 [5.7–87.8] | 10.5 [1.2–63.6] | 0.48 |
| Death in ICU, n (%) | 7 (24) | 0 (0) | 7 (30) | 0.12 |
SAPS II (Simplified Acute Physiology Score) was calculated within 24 h of admission. Values are given as median (25th–75th percentiles) and percentage according to data distribution. Comparisons were done using non-parametric Mann Whitney test. Patients’ characteristics
ICU intensive care unit, MAP mean arterial pressure, SAPS Simplified Acute Physiology Score, SOFA Score SOFA (Sequential Organ Failure Assessment), CRT capillary refill time
Bold values indicate statistically differences between groups
In responders, 30 min after infusion the heart rate significantly decreased, while systolic blood pressure increased, with a trend to an increase in cardiac index. No difference in mottling score was observed between the two timepoints (Table 2).
Table 2.
Global hemodynamic and tissue parameters at baseline and 30 min after fluid infusion parameters
| Parameters | Baseline | 30 min | P value |
|---|---|---|---|
| Heart rate, bpm | 106 [95–125] | 103 [90–124] | 0.01 |
| Blood pressure | |||
| Systolic, mmHg | 100 [94–123] | 109 [95–131] | 0.05 |
| Diastolic, mmHg | 59 [55–69] | 62 [56–67] | 0.51 |
| Mean, mmHg | 71 [68–83] | 78 [69–86] | 0.11 |
| Norepinephrine, μg/kg/min | 0.10 [0–0.40] | 0.10 [0–0.40] | 1 |
| Cardiac index, L/min/m2 | 2.8 [2.2–3.7] | 3.4 [2.3–4.3] | 0.12 |
| Capillary refill time | 3.8 [2.6–4.1] | 2.5 [1.5–3.4] | < 0.0001 |
| Mottling score | 2 [1–3] | 2 [0–3] | 0.13 |
Values are given as median [25th–75th percentiles]. Comparisons were done using paired non-parametric test. Global hemodynamic and tissue perfusion parameters baseline and 30 min after fluid infusion in responders (N = 23)
Bold values indicate statistically differences between groups
Fingertip CRT kinetics
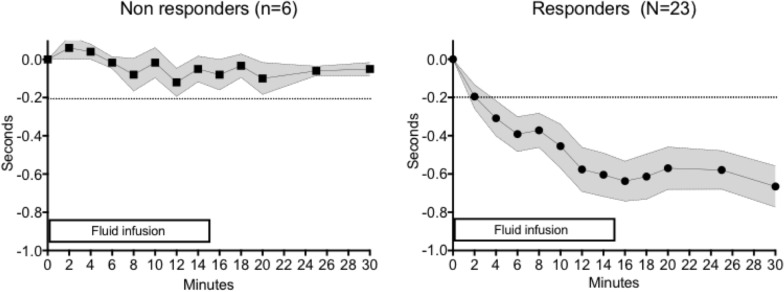
Fingertip CRT monitoring was started when the crystalloid infusion was initiated during a total 30 min period. We observed that fingertip CRT significantly decreased very quickly. Indeed, variations larger than 0.2 s were observed 6–8 min after the initiation of crystalloid infusion. Maximal fingertip CRT drop was observed 10–12 min after the start of infusion (−0.7 [−0.3;−0.9] sec) and this drop persisted until the end of the 30 min monitoring (Fig. 2A, B). The variations of the fingertip CRT correlated positively with the baseline values (R = 0.39, P = 0.05).
Fig. 2.
Kinetics of the variations of fingertip CRT overtime in non-responders (left) and in responders (right). Measurements started upon start of crystalloid infusion. Data were expressed as mean ± SD
Discussion
In sepsis patients we showed that fingertip CRT quickly improved after crystalloid infusion was initiated, with a significant decrease at 6–8 min, the maximal improvement being observed after 10–12 min and persisting at 30 min. CRT variations significantly correlated with baseline CRT measurements.
In pediatric wards, CRT has been mainly used as a triage tool to identify the most severe children suffering from infectious diseases, such as pneumonia, gastroenteritis and malaria [17, 18]. After initial resuscitation, a prolonged CRT identified sepsis patients with poor outcomes in both the emergency ward [8] and ICU [1]. More recently, Hernandez et al. reported that index tip CRT “normalization” at H6 after resuscitation was associated with a good prognosis [19]. Our group also found that improvement in peripheral tissue hypoperfusion is strongly associated with 14-day survival in septic shock patients [20]. Finally, in the ANDROMEDA-SHOCK trial, monitoring CRT every 30 min was used to guide early therapeutic strategy in septic shock patients, and suggested a benefit when compared to a lactate clearance guided approach [10]. Choosing a 30 min timepoint was mainly based on physician clinical experience, but the accurate timing of CRT variations during volume expansion in critically ill patients remained unknown. Despite no detailed timing for measurements, Jacquet-Lagrèze et al. reported a decrease in fingertip CRT after passive leg raising [21], suggesting that variations of CRT during fluid infusion may be rapid. We found that the CRT quickly improved with maximal response observed at 10–12 min after fluid expansion initiation.
Decreased CRT following fluid infusion may reflect improved microvascular perfusion as previously reported by Ospina-Tascon with videomicroscopy in the sublingual area during the early phase of resuscitation [22]. Several mechanisms may be responsible for improved microvascular perfusion, such as an attenuation of sympathetic-induced vasoconstriction or increased cardiac output. Here, we observed lower heart rate after fluid challenge supporting a reduction of sympathetic activation after volume expansion and a trend to increased cardiac index. Variability in cardiac index measurements using echocardiography [23] may explain, at least in part, the absence of significant difference between baseline and 30 min timepoints. Finally, the normalization of peripheral tissue hypoperfusion despite the absence of cardiac index increase following fluid infusion could also be due to blood dilution and improved microvascular rheology [24].
Different thresholds have been previously proposed to define prolonged CRT. Schriger et al. proposed 4.5 s while comparing subjects before and after fingertip exposure to cold water, which is far from the clinical setting [25]. We selected patients with fingertip CRT > 2.5 s based on our previous study on selected septic shock patients reporting that the index CRT cutoff at 2.4 s was a strong predictor of 14-day mortality[1] [5]. We report in this study a positive correlation between baseline CRT and variations after fluid challenge. In other words, the higher the baseline CRT, the higher the decrease after volume expansion.
In previous works, our group found that a decrease in mottling score 6 h after resuscitation is associated with better 14-day survival [20, 26]. Here, in responders, mottling score did not significantly change 30 min after fluid infusion, whereas CRT dropped. Such observation suggests that the mottling score is an appropriate triage tool but could not be used as a monitoring tool to guide rapid therapeutic intervention.
Our study has several limitations. It is a monocentric study, and results need to be confirmed in a larger population. CRT was monitored during a total of 30 min but only 15 min after the end of volume expansion and we cannot excluded delayed CRT improvement in the non-responders. In our study, criteria for volume expansion were not standardized and the intervention was decided by the physician in charge of the patient. Therefore, the evaluation of fluid responsiveness before the challenge was not recorded. In addition, CRT kinetics were measured by physicians with expertise in the evaluation of peripheral tissue perfusion and training is of paramount importance to perform accurate monitoring of CRT.
Conclusions
Fingertip CRT quickly improved during volume expansion in over 80% of septic patients, with a significant decrease as soon as 6–8 min after start of fluid expansion, and a maximal improvement at 10–12 min.
Supplementary Information
Additional file 1. Flow chart of studied population.
Acknowledgements
We are indebted to Rozenn Leboursicaud for their help in data extraction and research organization. We are indebted to Assistance Publique-Hôpitaux de Paris staff for his support.
Abbreviations
- CRT
Capillary refill time
- ICC
Intra-class correlation coefficient
- ICU
Intensive care unit
- MAP
Mean arterial pressure
- SOFA
Sequential organ failure assessment
- SAPS II
Simplified acute physiologic score II
Author contributions
Study concept and design, all authors. Acquisitions of data, all authors. Drafting of the manuscript, LR, PG, JJ and HAO. Critical revision of manuscript, all authors. Statistical analysis, LR, JJ and HAO. All authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethical approval and consent to participate
The protocol was approved by the ethical committee of the French Intensive Care Society (FICS) (CE SRLF 22-009). This is an observational study without any specific intervention. Volume expansion was decided by the physician in charge of the patient and repetitive capillary refill time measurements were recorded. Repetitive capillary refill time measurements during fluid infusion is routinely done in our ICU. All patients and families were informed through the admission leaflet that anonymous data could be used for academic research and gave their consent.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ait-Oufella H, Bige N, Boelle PY, Pichereau C, Alves M, Bertinchamp R, Baudel JL, et al. Capillary refill time exploration during septic shock. Intensive Care Med. 2014;7:958–964. doi: 10.1007/s00134-014-3326-4. [DOI] [PubMed] [Google Scholar]
- 2.van Genderen ME, Lima A, Akkerhuis M, Bakker J, van Bommel J. Persistent peripheral and microcirculatory perfusion alterations after out-of-hospital cardiac arrest are associated with poor survival. Crit Care Med. 2012;8:2287–2294. doi: 10.1097/CCM.0b013e31825333b2. [DOI] [PubMed] [Google Scholar]
- 3.Hasdai D, Holmes DR, Jr, Califf RM, Thompson TD, Hochman JS, Pfisterer M, Topol EJ. Cardiogenic shock complicating acute myocardial infarction: predictors of death. GUSTO investigators. Global Utilization of Streptokinase and Tissue-Plasminogen Activator for Occluded Coronary Arteries. Am Heart J. 1999;138:21–31. doi: 10.1016/S0002-8703(99)70241-3. [DOI] [PubMed] [Google Scholar]
- 4.Cecconi M, De Backer D, Antonelli M, Beale R, Bakker J, Hofer C, Jaeschke R, et al. Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2014;12:1795–1815. doi: 10.1007/s00134-014-3525-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hariri G, Joffre J, Leblanc G, Bonsey M, Lavillegrand JR, Urbina T, Guidet B, et al. Narrative review: clinical assessment of peripheral tissue perfusion in septic shock. Ann Intensive Care. 2019;1:37. doi: 10.1186/s13613-019-0511-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Alsma J, van Saase J, Nanayakkara PWB, Schouten W, Baten A, Bauer MP, Holleman F, et al. The power of flash mob research: conducting a nationwide observational clinical study on capillary refill time in a single day. Chest. 2017;5:1106–1113. doi: 10.1016/j.chest.2016.11.035. [DOI] [PubMed] [Google Scholar]
- 7.van Genderen ME, Paauwe J, de Jonge J, van der Valk RJ, Lima A, Bakker J, van Bommel J. Clinical assessment of peripheral perfusion to predict postoperative complications after major abdominal surgery early: a prospective observational study in adults. Crit Care. 2014;3:R114. doi: 10.1186/cc13905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lara B, Enberg L, Ortega M, Leon P, Kripper C, Aguilera P, Kattan E, et al. Capillary refill time during fluid resuscitation in patients with sepsis-related hyperlactatemia at the emergency department is related to mortality. PLoS ONE. 2017;11:e0188548. doi: 10.1371/journal.pone.0188548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lima A, Jansen TC, van Bommel J, Ince C, Bakker J. The prognostic value of the subjective assessment of peripheral perfusion in critically ill patients. Crit Care Med. 2009;3:934–938. doi: 10.1097/CCM.0b013e31819869db. [DOI] [PubMed] [Google Scholar]
- 10.Hernandez G, Ospina-Tascon GA, Damiani LP, Estenssoro E, Dubin A, Hurtado J, Friedman G, et al. Effect of a resuscitation strategy targeting peripheral perfusion status vs serum lactate levels on 28-Day mortality among patients with septic shock: the ANDROMEDA-SHOCK randomized clinical trial. JAMA. 2019;7:654–664. doi: 10.1001/jama.2019.0071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3) JAMA. 2016;8:801–810. doi: 10.1001/jama.2016.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, Sevransky JE, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Med. 2013;2:165–228. doi: 10.1007/s00134-012-2769-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, Machado FR, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Crit Care Med. 2021;11:e1063–e1143. doi: 10.1097/CCM.0000000000005337. [DOI] [PubMed] [Google Scholar]
- 14.Moreno R, Vincent JL, Matos R, Mendonca A, Cantraine F, Thijs L, Takala J, et al. The use of maximum SOFA score to quantify organ dysfunction/failure in intensive care. Results of a prospective, multicentre study. Working group on sepsis related problems of the ESICM. Intensive Care Med. 1999;7:686–696. doi: 10.1007/s001340050931. [DOI] [PubMed] [Google Scholar]
- 15.Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA. 1993;24:2957–2963. doi: 10.1001/jama.1993.03510240069035. [DOI] [PubMed] [Google Scholar]
- 16.Sandler SF, Zelefsky JR, Dorairaj S, Arthur SN, Ritch R, Liebmann JM. Intra-observer and inter-observer reliability and reproducibility of slit-lamp-adapted optical coherence tomography for evaluation of anterior chamber depth and central corneal thickness. Ophthalmic Surg Lasers Imaging. 2008;4:299–303. doi: 10.3928/15428877-20080701-15. [DOI] [PubMed] [Google Scholar]
- 17.Evans JA, May J, Ansong D, Antwi S, Asafo-Adjei E, Nguah SB, Osei-Kwakye K, et al. Capillary refill time as an independent prognostic indicator in severe and complicated malaria. J Pediatr. 2006;5:676–681. doi: 10.1016/j.jpeds.2006.07.040. [DOI] [PubMed] [Google Scholar]
- 18.Gove S, Tamburlini G, Molyneux E, Whitesell P, Campbell H. Development and technical basis of simplified guidelines for emergency triage assessment and treatment in developing countries. WHO Integrated Management of Childhood Illness (IMCI) referral care project. Arch Dis Child. 1999;6:473–477. doi: 10.1136/adc.81.6.473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hernandez G, Pedreros C, Veas E, Bruhn A, Romero C, Rovegno M, Neira R, et al. Evolution of peripheral vs metabolic perfusion parameters during septic shock resuscitation. A clinical-physiologic study. J Crit Care. 2012;3:283–288. doi: 10.1016/j.jcrc.2011.05.024. [DOI] [PubMed] [Google Scholar]
- 20.Ait-Oufella H, Lemoinne S, Boelle PY, Galbois A, Baudel JL, Lemant J, Joffre J, et al. Mottling score predicts survival in septic shock. Intensive Care Med. 2011;5:801–807. doi: 10.1007/s00134-011-2163-y. [DOI] [PubMed] [Google Scholar]
- 21.Jacquet-Lagreze M, Bouhamri N, Portran P, Schweizer R, Baudin F, Lilot M, Fornier W, et al. Capillary refill time variation induced by passive leg raising predicts capillary refill time response to volume expansion. Crit Care. 2019;1:281. doi: 10.1186/s13054-019-2560-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ospina-Tascon G, Neves AP, Occhipinti G, Donadello K, Buchele G, Simion D, Chierego ML, et al. Effects of fluids on microvascular perfusion in patients with severe sepsis. Intensive Care Med. 2010;6:949–955. doi: 10.1007/s00134-010-1843-3. [DOI] [PubMed] [Google Scholar]
- 23.Jozwiak M, Mercado P, Teboul JL, Benmalek A, Gimenez J, Depret F, Richard C, et al. What is the lowest change in cardiac output that transthoracic echocardiography can detect? Crit Care. 2019;1:116. doi: 10.1186/s13054-019-2413-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pranskunas A, Koopmans M, Koetsier PM, Pilvinis V, Boerma EC. Microcirculatory blood flow as a tool to select ICU patients eligible for fluid therapy. Intensive Care Med. 2013;4:612–619. doi: 10.1007/s00134-012-2793-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schriger DL, Baraff LJ. Capillary refill–is it a useful predictor of hypovolemic states? Ann Emerg Med. 1991;6:601–605. doi: 10.1016/S0196-0644(05)82375-3. [DOI] [PubMed] [Google Scholar]
- 26.Dumas G, Lavillegrand JR, Joffre J, Bige N, de Moura EB, Baudel JL, Chevret S, et al. Mottling score is a strong predictor of 14-day mortality in septic patients whatever vasopressor doses and other tissue perfusion parameters. Crit Care. 2019;1:211. doi: 10.1186/s13054-019-2496-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Flow chart of studied population.
Data Availability Statement
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.