An IDO-specific peptide vaccine is demonstrated to target IDO+ cells, enhancing vaccine-induced immune responses against non-IDO tumor antigens. These effects reduce tumor growth and inhibit immune-suppressive myeloid populations in multiple tumor models.
Abstract
The immunosuppressive tumor microenvironment (TME) does not allow generation and expansion of antitumor effector cells. One of the potent immunosuppressive factors present in the TME is the indoleamine-pyrrole 2,3-dioxygenase (IDO) enzyme, produced mainly by cancer cells and suppressive immune cells of myeloid origin. In fact, IDO+ myeloid-derived suppressor cells (MDSC) and dendritic cells (DC) tend to be more suppressive than their IDO− counterparts. Hence, therapeutic approaches that would target the IDO+ cells in the TME, while sparing the antigen-presenting functions of IDO− myeloid populations, are needed. Using an IDO-specific peptide vaccine (IDO vaccine), we explored the possibility of generating effector cells against IDO and non-IDO tumor-derived antigens. For this, IDO-secreting (B16F10 melanoma) and non–IDO-secreting (TC-1) mouse tumor models were employed. We showed that the IDO vaccine significantly reduced tumor growth and enhanced survival of mice in both the tumor models, which associated with a robust induction of IDO-specific effector cells in the TME. The IDO vaccine significantly enhanced the antitumor efficacy of non-IDO tumor antigen–specific vaccines, leading to an increase in the number of total and antigen-specific activated CD8+ T cells (IFNγ+ and granzyme B+). Treatment with the IDO vaccine significantly reduced the numbers of IDO+ MDSCs and DCs, and immunosuppressive regulatory T cells in both tumor models, resulting in enhanced therapeutic ratios. Together, we showed that vaccination against IDO is a promising therapeutic option for both IDO-producing and non–IDO-producing tumors. The IDO vaccine selectively ablates the IDO+ compartment in the TME, leading to a significant enhancement of the immune responses against other tumor antigen–specific vaccines.
Introduction
Tumor cells exploit tolerogenic and immunosuppressive mechanisms in the tumor microenvironment (TME) to support their growth and proliferation. Tumor immune escape and lack of response to immunotherapy have been shown to be associated with multiple immune-suppressive mechanisms in the TME (1, 2). Thus, it is important to develop therapeutic approaches that reduce the tumor-suppressive strategies, while simultaneously enhancing the antitumor effector CD8+ T cells.
Among many tumor-associated immune tolerance and suppressive mechanisms, indoleamine-pyrrole 2,3-dioxygenase (IDO) expression is a major contributor that functions by tryptophan catabolism in the TME. This leads to deprivation of tryptophan required for optimum immune cell growth and function and to the production of kynurenine, an immune inhibitory metabolite (3–5). Thus, the interruption of the IDO pathway can transform a tolerogenic/suppressive microenvironment into a proinflammatory environment that supports optimum T-cell responses (6). It is also known that IDO secretion results in the recruitment of myeloid-derived suppressor cells (MDSC) and M2-like macrophages to the TME and also enhances their suppressive function (7, 8). Furthermore, IDO may enhance the production of regulatory dendritic cells (DC) that, in turn, leads to immune suppression by inhibiting T-cell proliferation, inducing regulatory T cells (Treg), and inducing T-cell anergy (9, 10).
Importantly, besides tumors, the immune-suppressive compartment of myeloid cells, including DCs and macrophages, can also be a significant source of the IDO enzyme. Strategies to inhibit the IDO enzyme have been developed and have not been, so far, successful in clinical trials (11). Accordingly, other strategies to target IDO are being needed. Previously, using in vitro coculture experiments, we have shown that naturally occurring, IDO-specific T cells isolated from cancer patients are able to lyse IDO+ immune cells, as well as cancer cells (12–14). Here, we took a novel approach of generating IDO-specific CD8+ T cells with a peptide antigen-based IDO vaccine. We hypothesized that this approach leads to a two-prong immune targeting of the tumor: direct targeting of tumor cells expressing IDO and targeting of IDO+ immune-suppressive myeloid cells, while sparing the physiologically needed IDO− DCs and macrophages. Accordingly, this IDO vaccine would potentially also be effective against tumors that do not express IDO and would potentially be utilized as an immune-modulating approach to enhance other immune therapeutic strategies, including vaccines against an array of other antigens.
Hence, in the current study, we developed an IDO peptide vaccine strategy with the intention of generating IDO-specific CD8+ T-cell immune responses. We report that IDO vaccine significantly increases the number, activation, and function of CD8+ T cells in the TME, which was accompanied with decreased IDO-expressing suppressive myeloid cells and Tregs, independent of IDO expression in tumor cells. The immune response against a non-IDO tumor-specific vaccine was also enhanced when given in combination with IDO vaccine. This resulted in delayed tumor progression in both IDO-expressing and nonexpressing tumor models, B16F10 and TC-1, respectively. Hence, peptide-based IDO vaccine is a promising strategy to enhance the efficacy of tumor-specific vaccines in cancer therapy.
Materials and Methods
Mice and tumor cell lines
Female wild-type C57BL/6J mice (6–8 weeks old, The Jackson Laboratory) were housed under specific pathogen-free environment at Georgetown University Medical Center animal facility. All procedures were performed in accordance with and with the approval of Georgetown University Medical Center Institutional Animal Care and Use Committee protocols.
The B16F10 and CT26 cell lines were purchased from ATCC. TC-1 cells, derived by stable transfection of mouse lung epithelial cells with human papillomavirus strain 16 (HPV16) early proteins 6 and 7 (E6 and E7) and activated H-ras oncogene, were obtained from Dr. T-C Wu (Johns Hopkins University, Baltimore, MD; ref. 15). All tumor cell lines were routinely authenticated and tested for Mycoplasma by microscopic evaluation and PCR-based methods. Cells from passage numbers 10 to 12 were grown in RPMI1640 supplemented with 10% FBS, 2 mmol/L l-glutamine, penicillin (100 U/mL), and streptomycin (100 µg/mL; all from Gibco) at 37°C with 5% CO2 and maintained at a confluence of 70% to 80% as described previously (16).
IDO peptide identification and MHC binding prediction
SYFPEITHI (17), Rankpep (http://imed.med.ucm.es/Tools/rankpep.html), and CTLPred (https://bio.tools/ctlpred) databases for MHC ligands and peptide motifs were used to identify a set of 20 H2Db-specific nonamer epitopes in the mouse-IDO1 protein (18). The custom peptides were generated using PEPscreen peptide library synthesis at ProImmune Inc. The candidate peptides were assembled with MHC Class I mouse allele H-2Db and analyzed using the ProImmune REVEAL MHC-peptide binding assay to determine incorporation into MHC molecules (19). Binding to MHC molecules was compared with a positive control peptide, a known ProImmune-proprietary T-cell epitope. The binding score for each MHC-peptide complex was calculated by comparison with the binding of the positive control. Experimental standard error (SE) was obtained by triplicate positive control binding experiments. The SE for the positive control is reported as an illustration of the degree of error that can be obtained in a ProImmune REVEAL MHC-peptide Binding Assay.
Vaccine, reagents, and antibodies
The CTL epitope from human gp10025–33 (KVPRNQDWL; an altered peptide ligand vaccine for mice (100 µg/mouse; Celtek Bioscience) for the B16F10 tumor model or HPV16 E749–57 (RAHYNIVTF) for TC-1 tumor model [9-amino acid (aa) peptide, 100 µg/mouse] mixed with synthetic T-helper epitope PADRE (13 aa peptide, aK-Cha-VAAWTLKAAa, where “a” is D-alanine and Cha is L-cyclohexylalanine, 20 μg/mouse; both from Celtek Bioscience) and QuilA adjuvant (10 μg/mouse; Brenntag) were used as the model vaccine (subcutaneous injections, as described below), which contains both CD8 and CD4 epitopes in all studies (20). IDO vaccine (10-aa peptide, MTYENMDIL, 100 µg/mouse) for both the B16F10 and TC-1 tumor model was mixed with synthetic T-helper epitope PADRE (20 μg/mouse) and QuilA adjuvant (10 μg/mouse).
Recombinant IL2 (catalog no. 200-02; Peprotech), anti-CD3 (catalog no. 5 55273, clone 17A2), and anti-CD28 (553295, clone 37.51) were obtained from BD Biosciences. Live/Dead Fixable Near-IR Dead Cell staining kit was obtained from Invitrogen, Thermo Fisher Scientific (catalog no. L34976). Appropriately fluorochrome-labeled anti-mouse antibodies against CD45 (clone 30-F11), CD3 (clone 145-2C11), CD4 (clone RM4-5), CD8 (clone 53-6.7), CD40 L (clone MR1), IFNγ (clone XMG1.2), Foxp3 (clone MF23), CD11b (clone M170), CD11c (clone HL3), Gr1 (clone RB6-8C5), Ly-6G (clone 1A8), Ly-6C (clone AL-21), F4/80 (clone BM8), CD80 (clone 16-10A1), and CD206 (clone C068C2) were purchased from BD Biosciences, whereas granzyme B (GB; clone NGZB) and B220 (catalog no. 11-0452-82, clone RA3-6B2) were purchased from Thermo Fisher Scientific for flow cytometric measurements as described below. For IDO estimation, two different antibodies were used: anti-IDO1-AF647 (clone 2E2/IDO1, BioLegend, catalog no. 654004) and anti-IDO-1-PE (clone mIDO-48, eBioscience, catalog no. 12-9473-82). The intracellular Foxp3 staining kit (catalog no. 560409) and intracellular cytokine staining kit were obtained from BD Biosciences (catalog no. 51-2091KZ). gp100-APC (catalog no. JA3570; H2Db-KVPRNQDWL) and E7-FITC (catalog no. JA2195; H2Db-RAHYNIVTF) dextramers were from Immudex. For detection of IDO+ CD8+ T cells, biotin-labeled H2-Db-MTYENMDIL (catalog no. SP/7337-01) targeting pentamer or a nonspecific biotin-labeled H2Db-KAVYNFATC (catalog no. RP/6893-08) pentamer from ProImmune Inc. was used. Streptavidin-PE (catalog no. 349023) from BD Biosciences was used to detect the IDO-specific pentamer signal. The IDO antibody for Western blotting was obtained from AbCam (ab106134) and β-tubulin antibody was from Cell Signaling Technology (9F3; catalog no. 2128).
Tumor implantation, immunization, and tumor volume measurement
For therapeutic experiments, 1 × 105 B16F10 or 7 × 104 TC-1 cells per mouse were subcutaneously implanted into the flanks of C57BL/6J mice on day 0. On day 10 when tumors measured 5 to 6 mm in diameter, mice were randomly assigned to four groups according to the schedule shown in figures, and treated with a tumor-specific vaccine (gp10025–33 or HPV16 E749–57), IDO vaccine, or a combination of both (once a week, subcutaneously, total three doses). One group was left untreated and served as the control. Every 3 to 4 days, tumors were measured by a digital Vernier caliper, and the tumor volume was calculated using the formula: V = L × W2/2, where V is tumor volume, L is the length of tumor (longer diameter), and W is the width of the tumor (shorter diameter). Mice were observed for tumor growth and survival. Mice were euthanized once tumors reached a volume of 1.5 cm3, tumors were ulcerated, or when mice became moribund.
For immune response studies, mice were treated following the same schedule as for the therapy experiment, except only two doses of weekly vaccines were administered. Three days after the second vaccination, mice from appropriate groups were euthanized to harvest tumors (TC-1 and B16). The tumors were diced into small pieces of about 2 to 3 mm and were further processed using GentleMACS dissociator (catalog no. 130-093-235, Miltenyi Biotec) and the solid tumor homogenization protocol using tumor dissociation kit, mouse (catalog no. 103-096-730, Miltenyi Biotec), as suggested by the manufacturer (Miltenyi Biotec). Each experiment was repeated at least twice.
Flow cytometric analysis of tumor-infiltrating lymphocytes
One million cells isolated from tumor tissues were stained using the Live/Dead Fixable Near-IR Dead Cell staining kit (Invitrogen, Thermo Fisher Scientific), followed by cell fixation and permeabilization according to the manufacturer's (BD Pharmingen) protocol. For surface markers, cells were stained with appropriate antibodies in FACS buffer [1% FBS/BSA, 0.09% sodium azide, and 5 mmol/L EDTA (Sigma-Aldrich)] for 30 minutes at 4°C in the dark and washed twice in FACS buffer before acquisition. For IFNγ and IDO staining, BD Cytofix/Cytoperm (catalog no. 51-2090KZ) and BD Perm/Wash (catalog no. 51-2091KZ) buffer set were used as per manufacturer's instructions (BD Pharmingen). Data acquisition was performed on FACSCalibur or LSRII (BD Biosciences). Total numbers or frequency of CD3+, CD8+, CD8+E7+, E7+GB+, CD40L+, IFNγ+, CD4+, CD4+Foxp3+, CD11b+F4/80+, CD11c+, CD11b+Gr1+, CD80+, CD206+, B220+, and IDO+ cells were analyzed within the CD45+ hematopoietic cell population and represented on respective populations. For checking the expression of IDO, TC-1 and B16F10 cancer cell lines were stained with IDO antibody as described above. Results were analyzed with FlowJo (TreeStar) software.
Western blot analysis
TC-1 and CT26 tumor cells were either treated with IFNγ (100 ng/mL, catalog no. 315-05, Peprotech) for 48 hours or remained untreated. Cells were harvested and treated with cell lysis buffer cocktail (RIPA buffer, 1% phosphatase inhibitor, and 1% protease inhibitor; Thermo Fisher Scientific) to prepare cell lysates. Protein concentration in each lysate was quantified by Pierce BCA Protein Assay Kit (Thermo Fisher Scientific). A total of 40 μg protein was loaded onto Novex 4%–20% Tris-Glycin Mini Gels (Thermo Fisher Scientific), followed by transfer onto nitrocellulose membranes. Membranes were blocked with 3% BSA (Sigma-Aldrich) in Tris buffer (VWR Chemicals) for 2 hours at room temperature, followed by overnight probing of the proteins with antibodies directed against mouse IDO1 (1:500, AbCam, ab106134) and β-tubulin (1:2,000, Cell Signaling Technology, catalog no. 2128; 9F3 used as an internal control) in the blocking buffer at 4°C. The blots were washed in TBST [Tris buffer + Tween-20 (0.1%, Promega)] three times, each 10 minutes at room temperature on the shaker. Blots were incubated with anti-rabbit horseradish peroxidase–labeled secondary antibody (1:2,000, catalog no. 7074, Cell Signaling Technology) for 2 to 3 hours at room temperature. After washing the membrane for three times (each 10 minutes on the shaker at room temperature), blots were developed with Pierce enhanced chemiluminescence Western blotting substrate (Thermo Fisher Scientific) using iBright Imaging System (Thermo Fisher Scientific).
Statistical analysis
All statistical parameters (average values, SEM, significant differences between groups) were calculated using GraphPad Prism or Excel as appropriate. Statistical significance between groups was determined by Student t test or one-way ANOVA with Tukey multiple comparison post hoc test, and P ≤ 0.05 was considered as statistically significant. Survival in various groups was compared using GraphPad Prism using log-rank (Mantel–Cox) test. SK plots were generated by internally developed software (https://skylineplotter.shinyapps.io/SkyLinePlotter/). Contrary to the survival plots generated using GraphPad Prism, the SK plot gives dynamic simultaneous presentation of tumor volumes and mouse survival at a specific timepoint.
Results
Identification of IDO-MHC binding epitopes
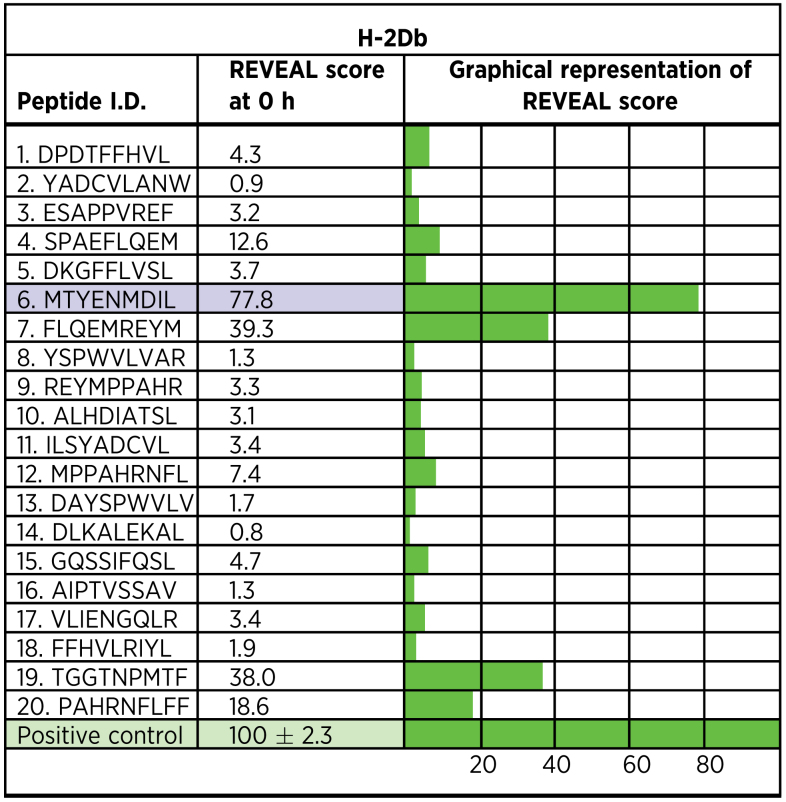
Some tumor cells express IDO enzyme as an immune-suppressive factor within the TME (21). Therefore, IDO protein may potentially function as a unique tumor-associated antigen. Hence, vaccination using an IDO-specific epitope may provide a viable immune therapeutic strategy. To test this hypothesis, we determined the predictable REVEAL binding score of 20 H2Db-specific nonamer-IDO candidate peptides (ProImmune) and found binding scores ranging from 0.8 (for the least binding peptide) to 77.8 (for the best binding peptide; Table 1). Three of 20 peptides had binding scores > 35% (Table 1; also shown as graphical representation). However, only one peptide (MTYENMDIL) had a score that was > 45% of the positive control, an internal standard set to depict high probability of specific binding. Hence, this IDO peptide was chosen for further experiments.
Table 1.
IDO-specific peptide candidates and their respective MHCI:Peptide binding scores.
IDO vaccine induces potent IDO-specific antitumor immune responses
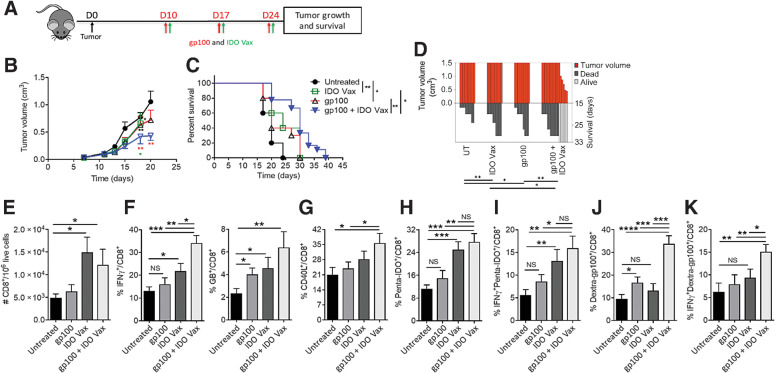
To determine the direct antitumor effect of IDO vaccine, we tested the vaccine in an IDO-expressing tumor model, B16F10, a tumor that expressed high IDO, as measured by flow cytometry (Supplementary Fig. S1A–S1C). TC-1 cells, on the other hand, did not express IDO and showed minimal induction when treated with IFNγ compared with CT26 cells (positive control; Supplementary Fig. S1). Mice carrying B16F10 tumors were treated with either IDO vaccine or in combination with a B16 tumor-specific vaccine (gp100) as a positive control. We found that treatment of B16F10 tumor-bearing mice with IDO vaccine (Fig. 1A) led to a significant reduction in tumor growth (P ≤ 0.05) and enhancement of mouse survival P ≤ 0.01) compared with untreated mice (Fig. 1B–D). The antitumor effects achieved after IDO vaccine were comparable with that of the gp100 peptide treatment (Fig. 1B–D). Furthermore, we found that combining IDO vaccine and gp100 peptide led to a significant enhancement of antitumor efficacy over either of the peptides administered alone because these mice continued to have significant reduction in the tumor volume, and 33% of them survived at day 33 (Fig. 1B–D). These results demonstrate that IDO vaccine induces effective antitumor responses in IDO-expressing tumors and can boost responses to a known tumor antigen–specific vaccine.
Figure 1.
IDO vaccine enhances immune-mediated antitumor effects of tumor antigen–specific vaccination and prolongs survival in the B16F10 tumor model. Gating strategy and IDO pentamer specificity in Supplementary Fig. S2. A, Schematic of the treatment schedule in the tumor model. On day 10 of tumor growth, B16F10 tumor-bearing mice were given IDO vaccine (IDO Vax) with the gp10025–33 peptide vaccine, along with PADRE (20 μg/mouse) and QuilA (10 μg/mouse) subcutaneously, every 7 days for a total of three doses. Tumor growth and survival were measured. B, Average tumor volume in mice following treatment (* vs. untreated; green * vs. IDO Vax; red * vs. gp100). C, Percent survival of mice depicted by the Kaplan–Meier plot. D, SK plot showing tumor volume and survival for each mouse at different days. Data are shown as an average of two independent experiments (n = 10–18 per group). Error bars indicate the SEM. For tumor growth, statistical analysis was performed by unpaired, one-tailed Student t test. Survival in various groups was compared using log-rank (Mantel–Cox) tests. *, P ≤ 0.05; and **, P ≤ 0.01. E–K, C57BL/6J mice (n = 5–8 per group) were treated as in A, except 3 days after second vaccination, mice were sacrificed, and tumors were harvested for immune response study. The frequency of B16F10 tumor-infiltrating cells was determined. Total (E), IFNγ+ and GB+ (F), CD40L+ (G), Penta-IDO+ (H), IFNγ+penta-IDO+ (I), Dextra-gp100+ (J), IFNγ+dextra-gp100+ (K) CD8+ T cells were measured by flow cytometry. Data are shown from one representative experiment of two independent experiments. Error bars indicate the SEM. Statistical analysis was performed by unpaired, one-tailed Student t test. NS: nonsignificant; *, P ≤ 0.05; **, P ≤ 0.01; ***, P ≤ 0.001; ****, P ≤ 0.0001.
IDO vaccine significantly enhances gp100-specific immune responses
To test the immune mechanisms by which IDO vaccine led to its therapeutic efficacy when given either as a single agent or in combination with another tumor-specific gp100 vaccine, we evaluated the immune profile in the TME. For this, B16F10 tumor-bearing mice were treated as mentioned above, and tumors were harvested 3 days after the second vaccination for evaluation of immune infiltrates by flow cytometry (Supplementary Fig. S2). We found that IDO vaccine significantly increased the number of total CD8+ T cells within the TME (Fig. 1E), as well as their functionality and activation as measured by IFNγ and granzyme B production and CD40 L expression (Fig. 1F and G). Furthermore, IDO vaccine increased the number of IDO antigen-specific CD8+ T cells measured with a pentamer against IDO (Penta-IDO+CD8+ T cells; Fig. 1H). To ascertain the identity of these IDO-specific CD8+ T cells, a nonspecific biotin-labeled pentamer was used as an internal control (Supplementary Fig. S2B). These IDO-specific CD8+ T cells showed robust activity (IFNγ+Penta-IDO+CD8+ T cells; Fig. 1I). We also found that when combined with the gp100 vaccine, IDO vaccine led to a significant enhancement of gp100-specific immune response as measured with a dextramer against gp100 (Dextra-gp100+CD8+; Fig. 1J). This also led to the enhancement of the functional activity of gp100-specific CD8+ T cells (IFNγ+Dextra-gp100+CD8+ T cells; Fig. 1K). On the other hand, as expected, the gp100 vaccine did not enhance immune responses of IDO vaccine (Fig. 1H and I). These data demonstrate that the generation of IDO-specific immune responses promotes development of immune responses against other tumor antigens.
IDO vaccine enhances antitumor immune responses in non–IDO-expressing TC-1 tumors
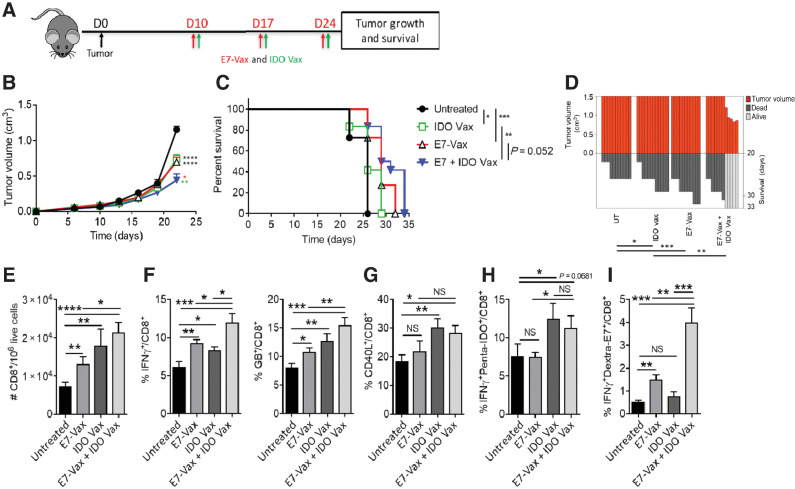
On the basis of data thus far and to test the mechanism by which IDO vaccine can enhance immune responses against a nonrelevant antigen (gp100; Fig. 1), we hypothesized that anti-IDO immune responses exert an additional bystander immune effect within the TME. To test this hypothesis, we treated mice carrying TC-1 tumors that did not express IDO protein (Supplementary Fig. S1; ref. 22) with IDO vaccine and monitored tumor responses (Fig. 2A). We found that despite the lack of IDO protein expression in TC-1 tumors, treatment with IDO vaccine led to a significant reduction in tumor growth and an increase in mouse survival (Fig. 2B–D). This clearly indicated that IDO vaccine could generate antitumor immune responses that were independent of IDO expression by tumor cells via bystander immune modulation. In line with the B16 model, in TC-1 tumors, the anti-tumor activity of IDO and tumor-specific E7 vaccines were comparable (Fig. 2B–D). We also found that IDO vaccine significantly enhanced the antitumor activity of the TC-1–specific E7 vaccine when given in combination (Fig. 2B–D).
Figure 2.
IDO vaccine enhances immune-mediated antitumor effects of tumor antigen–specific vaccination and prolongs survival in the TC-1 tumor model. Gating strategy and IDO pentamer specificity in Supplementary Fig. S3. A, Schematic of the treatment schedule in the tumor model. On day 10 of tumor growth, TC-1 tumor-bearing mice were given IDO vaccine with HPV17 E749–57 peptide vaccine, along with PADRE (20 μg/mouse) and QuilA (10 μg/mouse) subcutaneously, every 7 days for a total of three doses. Tumor growth and survival were measured. B, Average tumor volume in mice following treatment (* vs. untreated; green * vs. IDO Vax; red * vs. E7-Vax). C, Percent survival of mice depicted by the Kaplan–Meier plot. D, SK plot showing the tumor volume and survival for each mouse at different days. Data are shown as an average of two independent experiments (n = 10–12 per group). Error bars indicate SEM. For tumor growth, statistical analysis was performed by unpaired, one-tailed Student t test. Survival in various groups was compared using log-rank (Mantel–Cox) tests. *, P ≤ 0.05; **, P ≤ 0.01; ***, P ≤ 0.001; ****, P ≤ 0.0001. E–I, C57BL/6J mice (n = 5–7 per group) were treated as in A, except 3 days after second vaccination, mice were sacrificed, and tumors were harvested for immune response study. The frequency of TC-1 tumor-infiltrating cells was determined. Total (E), IFNγ+ and GB+ (F), CD40L+ (G), IFNγ+penta-IDO+ (H), IFNγ+dextra-E7+ (I) CD8+ T cells were measured by flow cytometry. Data are shown from one representative experiment of two independent experiments. Error bars indicate the SEM. Statistical analysis was performed by unpaired, one-tailed Student t test. NS, nonsignificant; *, P ≤ 0.05; **, P ≤ 0.01; ***, P ≤ 0.001; ****, P ≤ 0.0001.
To understand the mechanism by which IDO vaccine improved the therapeutic outcome in animals carrying tumors that did not express IDO, we evaluated immune responses in the TME (Supplementary Fig. S3). Consistent with the findings in B16F10 tumors, IDO vaccine significantly increased the number of total and IDO-specific CD8+ T cells in the TME and also increased their activation and functionality (Fig. 2E–H). Also consistent with the observation with the gp100 peptide, treatment with IDO vaccine significantly increased the number of E7-induced activated antigen-specific CD8+ T cells in the TME (IFNγ+Dextra-E7+CD8+; Fig. 2I). This further indicated that IDO vaccine exerted an immune-modulatory effect that enhanced specific antitumor immune responses beyond antigen-specific effects. Taken together, these data demonstrate that IDO vaccine exerts its IDO-specific CD8+ T cells effect through two separate mechanisms: (i) directly targeting tumors expressing IDO and (ii) the generation of an immune-modulatory effect that enhances tumor antigen–specific CD8+ T-cell responses independent of tumor cell IDO expression.
IDO vaccine reduces IDO-expressing myeloid populations in the TME
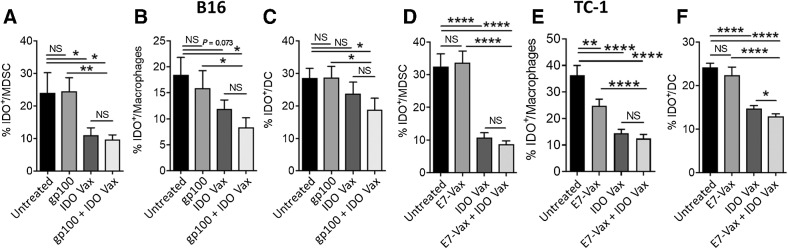
Next, we aimed to determine the underlying immune mechanism by which IDO vaccine enhanced antitumor immune responses independent of tumor cell IDO expression and its ability to enhance immune responses against other antigens. Myeloid cells are known to express IDO (23), and IDO-expressing myeloid cells are reported to be more suppressive compared with their non-IDO counterparts (24). Hence, we evaluated the effect of the IDO vaccine on immune-regulatory myeloid cells that expressed IDO. We found that IDO vaccine significantly reduced the frequency of IDO-expressing suppressive myeloid cells in the TME, including MDSCs (IDO+CD45+CD3–CD11b+Ly6G/C+; Fig. 3A and D), macrophages (IDO+CD45+CD3−CD11b+CD11c−F4/80+; Fig. 3B and E), and DCs (IDO+CD45+CD3–CD11b+CD11c+F4/80−; Fig. 3C and F) in both the IDO-expressing B16F10 and non–IDO-expressing TC-1 tumor models. These results demonstrate that IDO vaccine significantly increases the number and activity of CD8+ T cells, potentially by reducing the frequency of immunosuppressive myeloid cells, leading to enhanced immune-mediated antitumor effects.
Figure 3.
IDO vaccine reduces IDO-expressing suppressive myeloid cell populations in the TME. B16F10-bearing or TC-1–bearing mice were treated as in Fig. 1A and Fig. 2A, respectively, except 3 days after second vaccination, mice were sacrificed, and tumors were harvested for immune response evaluation. The frequency of B16F10 tumor-infiltrating IDO+ MDSCs (A), IDO+ macrophages (B), and IDO+DCs (C) were examined by flow cytometry. Data are shown from one representative experiment (n = 4–6 per group) of two independent experiments. The frequency of TC-1 tumor-infiltrating IDO+ MDSCs (D), IDO+ macrophages (E), IDO+DC (F) were also examined by flow cytometry. Data are shown as an average of two independent experiments (n = 10–15 per group). Error bars indicate the SEM. Statistical analysis was performed by unpaired, one-tailed Student t test. NS, nonsignificant; *, P ≤ 0.05; **, P ≤ 0.01; ***, P ≤ 0.001; ****, P ≤ 0.0001.
IDO vaccine enriches antitumor myeloid populations in the TME
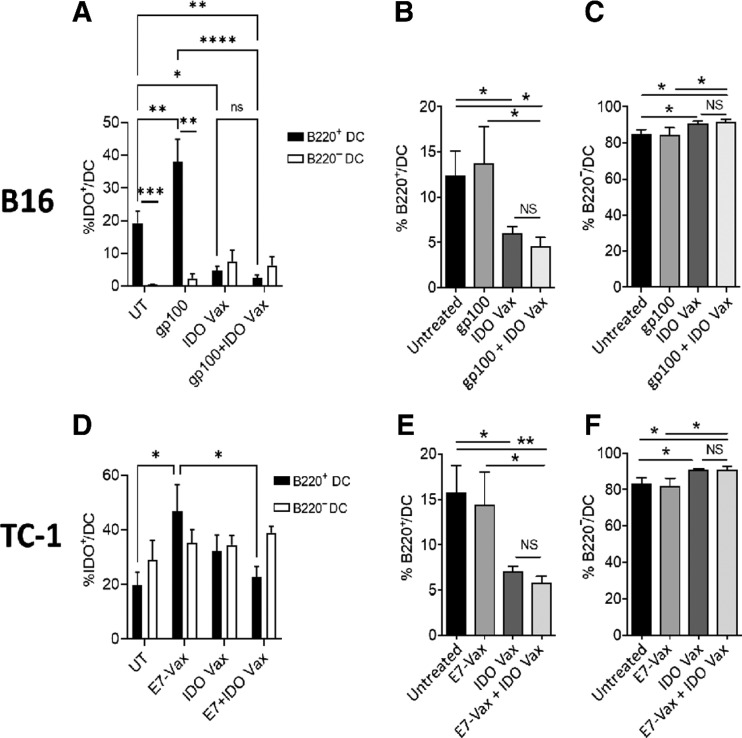
IDO is a suppressive enzyme and can skew immune cells toward a suppressive phenotype. Because we found that IDO vaccination resulted in a significant decrease in the numbers of IDO+ DCs (Fig. 3C and F), we asked whether IDO vaccination could also skew DCs, the most potent antigen-presenting cells (APC), from a protumorigenic phenotype (CD11b+CD11c+F4/80−B220+) to an antitumorigenic phenotype (CD11b+CD11c+F4/80−B220−; Supplementary Fig. S4). We, therefore, first checked the numbers of IDO+B220+ and IDO+B220− DCs after various treatments in B16F10 (Fig. 4A–C) and TC-1 (Fig. 4D–F) tumor-bearing mice. We found that the tumor-specific vaccine significantly increased the number of IDO+B220+ DCs in both tumor models (Fig. 4A and D). However, administration of IDO vaccine, along with the tumor-specific vaccine, prevented the induction of these protumorigenic cells. On the other hand, the number of IDO+B220− DCs remained unchanged in treated mice in both the tumor models (Fig. 4A and D). Furthermore, we found that the frequency of B220+ DCs was significantly reduced with IDO vaccine treatment alone or in combination with the tumor-specific vaccine in both B16F10 and TC-1 tumor models (Fig. 4B and E). This was accompanied by further enrichment of antitumor B220− DCs in the TME in both the tumors (Fig. 4C and F). Thus, these results clearly demonstrate that IDO-peptide vaccination removes IDO-expressing DCs from the TME and skews DCs toward antitumor phenotype.
Figure 4.
IDO vaccine modulates phenotypic characteristics of the myeloid population in the TME. Gating strategy in Supplementary Fig. S4. Phenotypic characteristics of myeloid populations were analyzed by flow cytometry in the B16F10 and TC-1 tumor models (see Fig. 1A and Fig. 2A, respectively, except 3 days after second vaccination, mice were sacrificed, and tumors were harvested for immune response evaluation. The frequency of B16F10 tumor-infiltrating IDO+B220+ DCs and IDO+B220− DCs (A), total B220+ DCs (B), B220− DCs (C) were determined. The frequency of TC-1 tumor-infiltrating IDO+B220+ DCs and IDO+B220− DCs (D), B220+ DCs (E), B220− DCs (F) were also assessed. Data are shown from one representative experiment (n = 4–7 per group) of two independent experiments. Error bars indicate the SEM. Statistical analysis was performed by unpaired, one-tailed Student t test. NS, nonsignificant; *, P ≤ 0.05; **, P ≤ 0.01; ***, P ≤ 0.001.
We further investigated the effects of the generation of IDO-specific CD8+ T cell responses on the protumor and antitumor macrophages in the TME after IDO vaccine treatment alone or in combination with tumor-specific vaccines. We found that the number of protumor M2 macrophages (CD45+CD3−CD11b+CD11c−F4/80+CD80loCD206hi) decreased significantly (Supplementary Fig. S5A and S5C), whereas the frequency of antitumor M1 macrophages (CD45+CD3−CD11b+CD11c−F4/80+CD80hiCD206lo; Supplementary Fig. S5B and S5D) significantly increased in B16F10 and TC-1 tumors. Together, these results suggest that IDO-specific vaccine enriches antitumor myeloid populations in the TME.
IDO vaccine results in significant increase in therapeutic ratio within the TME
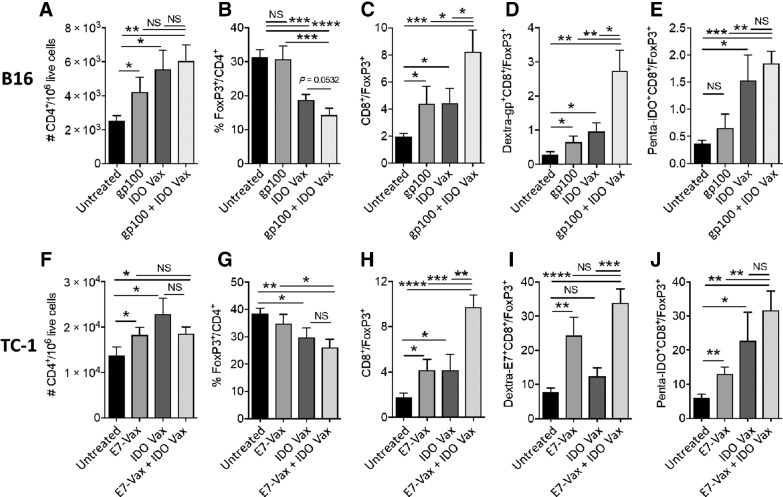
In line with the enhanced antitumor activity of IDO vaccine with and without tumor-specific antigens, we found a significant increase in the number of CD4+ T cells in the TME of both B16F10 and TC-1 tumors (Fig. 5A and F). Foxp3+CD4+ Tregs are an important element of the immunosuppressive lymphoid repertoire, which are known to be enhanced in the presence of the IDO enzyme (23). Moreover, IDO-expressing DCs are shown to increase the numbers and activation of Tregs in the TME (25). Because we found a significant decrease in the numbers of IDO+B220+ DCs, next we asked whether this treatment strategy also affected Tregs in the TME. We found that, indeed, the treatment with IDO vaccine, but not gp100- or E7-specific vaccines leads to reduction in the ratio of Tregs within CD4+ T cells in the TME (Fig. 5B and G). Furthermore, the combination of gp100- or E7-specific vaccines with IDO vaccine did not further influence the frequency of Tregs (Fig. 5B and G).
Figure 5.
IDO vaccine reduces frequency of Tregs and enhances therapeutic ratio in the TME. C57BL/6J mice were treated as in Fig. 1A and Fig. 2A, except 3 days after second vaccination, mice were sacrificed, and tumors were harvested for immune response study. The frequency of B16F10 tumor-infiltrating CD4+ (A), FoxP3+CD4+ (B), CD8+/Tregs (C), Dextra-gp100+ CD8+/Tregs (D), and Penta-IDO+CD8+/Tregs (E) was determined by flow cytometry. The frequency of TC-1 tumor-infiltrating CD4+ (F), FoxP3+CD4+ (G), CD8+/Tregs (H), Dextra-E7+ CD8+/Tregs (I), and Penta-IDO+CD8+/Tregs (J) were also measured by flow cytometry. Data are shown as an average of two independent experiments (n = 8–13 per group). Error bars indicate the SEM. Statistical analysis was performed by unpaired, one-tailed Student t test. NS, nonsignificant; *, P ≤ 0.05; **, P ≤ 0.01; ***, P ≤ 0.001; ****, P ≤ 0.0001.
On the basis of the data above, we found that IDO vaccine in combination with tumor-specific vaccines significantly increased the therapeutic ratios CD8+/Tregs, Penta-IDO+ CD8+/Tregs, Dextra-gp100+ CD8+/Tregs, and Dextra-E7+CD8+/Tregs compared with control groups in the two tumor models (Fig. 5C–E, H–J). Together, these data demonstrate that the enhanced antitumor efficacy caused by IDO vaccine is facilitated by the significant increase in therapeutic immune cell ratios. These results also show that combination of IDO and tumor antigen–specific vaccines has the potential to expand the immunologic response to tumor vaccines.
Discussion
One of the major reasons for the failure of immunotherapy, including cancer vaccines, is the presence of a highly immunosuppressive TME (26). Despite the fact that cancer vaccines have been shown to generate high frequencies of effective tumor-specific T cells, no major clinical benefit has been observed so far, and, hence, despite the many cancer vaccines under development, only one therapeutic cancer vaccine has been approved for human use in the past 20 years (27, 28). Thus, therapeutic strategies to downregulate the suppressive activity of the TME are necessary to enhance the outcome of immunotherapy, including therapeutic vaccines.
IDO is a known immune-suppressive enzyme secreted by some tumor cells and by suppressive myeloid cells, including APCs and MDSCs (29). It has been reported that myeloid cells expressing IDO have higher immunosuppressive activity (29). IDO-expressing cells enhance the accumulation of the tryptophan catabolite, kynurenine, which has been shown to mediate immunosuppression (30). IDO-mediated depletion of tryptophan, an essential growth factor, prevents activation of immune cells in the TME (30). A concerted effect of nutrient depletion and enhancement of myeloid and lymphoid immunosuppressive populations in the TME leads to establishment of an anti-inflammatory cytokine profile. Thus, inhibiting the IDO pathway can support the generation of a proinflammatory environment that also allows for immune-mediated antitumor responses (6). However, strategies to inhibit the IDO enzyme using specific inhibitors have not been successful in the clinic (11). Interestingly, in a small phase I trial, IDO-specific vaccination was found to induce durable responses in some patients, but the mechanisms underlying this effect were not explored (31, 32). Recently, results from a phase I/II trial in patients with metastatic melanoma show a very high objective response rate (80%) and a median progression-free survival of 26 months following treatment with an immunomodulatory IDO/PD-L1–targeting vaccine in combination with nivolumab (33). Under in vitro conditions, it has been shown that IDO-specific CD8+ T cells isolated from patients with cancer are able to eliminate IDO+ immune cells and cancer cells (12–14), raising the possibility that immunologic induction of these cells by appropriate IDO-specific vaccination can be a viable strategy to induce antitumor immune responses. In fact, in the above mentioned trial in patients with metastatic melanoma, we found that vaccine-reactive T cells have significant activity against cancer cells and immune cells that expressed IDO and PD-L1 (33).
Our results show that IDO-specific vaccination is a novel approach, the effects of which are not only limited to IDO-expressing tumors but also extend to non–IDO-expressing tumors, whereby anti-IDO effector cells induced by IDO vaccine potentially kill the IDO+ host myeloid cell populations, leading to their reduced numbers. Besides killing of the IDO+ cells, downregulation of IDO production in these cells could be another mechanism for their reduced numbers. In fact, it has been shown that soluble CD28 interaction with B7 (CD80/86) on DCs induces expression of IL6, which prevents IFNγ-mediated upregulation of IDO (34). We found an enrichment of CD80+ DCs with activated CD8+ T cells after IDO vaccine treatment, suggesting that indeed this could be one of the mechanisms of IDO vaccine–mediated reduction in the IDO+ host myeloid cells. IDO vaccine strategy also resulted in enrichment of antitumor IDO− myeloid cells in the TME. IDO is known to stabilize suppressive Tregs (3). Accordingly, treatment with IDO vaccine also resulted in a diminished Treg population within CD4+ T cells in the TME. On the basis of the above, collectively, this leads to downregulation of immunosuppressive populations and an increase in the proinflammatory immune cell populations, an indicator of good prognosis in cancer (35, 36).
Here, we made the observation that the generation of specific CD8+ T-cell responses against IDO provided a bystander immune-modulatory effect that enhanced immune responses against other tumor-specific antigens, such as gp100 in B16F10 and E7 in TC-1 tumors. Furthermore, the antitumor immune responses induced by IDO vaccine were observed independent of IDO expression by tumor cells, suggesting that IDO-specific CD8+ T cells could be effective against tumors regardless of the IDO expression status. Indeed, we found that IDO vaccine significantly reduced the number of IDO-expressing myeloid cells, including DCs, MDSCs, and macrophages, which are immunosuppressive in nature (29). The suppressive effects of MDSCs on innate and adaptive immune responses block immune surveillance and decrease the immune functions of effector cells, preventing the elimination of cancer cells (37). Importantly, earlier correlation between IDO expression and MDSC infiltration has been seen in human melanoma (38). Inhibition of IDO results in reduction of MDSCs and reversal of immune suppression (38). Our findings showed that IDO-expressing MDSCs were successfully decreased by the generation of IDO-specific CD8+ T cells in the TME.
IDO+ DCs have been shown to be suppressive and can induce Tregs in the TME (25). Recently, it was reported that IDO-expressing DCs can skew T-cell immune responses toward T-cell tolerance (39). Furthermore, IDO+ DCs help dormant tumor cells evade CD8+ T cells (40). In line with these observations, we demonstrate that IDO vaccine reduced the number of IDO+ DCs in the TME and was accompanied by reduced Treg numbers. This is consistent with earlier in vitro observations where it was found that activated IDO-specific CD8+ T cells decrease the frequency of Tregs and increase the levels of pro-inflammatory cytokines in peripheral blood mononuclear cells (13). In addition, it is known that tolerogenic B220+ DCs can activate resting Tregs, which suppress target immune cells in a PD-L1–dependent manner (40). Here, we found a decrease in tolerogenic B220+ DCs, with a concomitant increase in immune-activating B220−DCs in the TME after IDO vaccine therapy. Finally, it has been reported that IDO contributes to macrophage polarization through conversion of an antitumor M1 to a protumor M2 macrophages phenotype (8). We found that this macrophage polarization was reversed after treatment with IDO vaccine. Moreover, IDO-expressing macrophages suppress adaptive immunity by inhibiting inflammatory T-cell proliferation. We also found that IDO vaccine leads to a significant reduction in the number of IDO+ macrophages. Thus, this treatment strategy would skew the intratumoral environment, making it more conducive for generation of antitumor macrophages. Furthermore, these observations also highlight the fact that the IDO vaccination strategy primarily induces cell-mediated immune responses, although contribution of humoral immunity and associated antibody-dependent cellular cytotoxicity after IDO vaccine treatment cannot be ruled out and needs further investigation. Accordingly, to further elucidate the cellular mechanisms of this novel vaccination strategy, we are currently evaluating the cytotoxic/killing function of vaccine-induced CD8+ T cells ex vivo. In addition, to confirm that reduction in immunosuppressive cells is mediated by CD8+ and CD4+ T cells, in vivo T-cell depletion experiments are ongoing. We are also assessing the TME for the infiltration of CD8+ T cells and frequency of IDO+ MDSCs and macrophages following various treatments.
In conclusion, IDO vaccine enhances antigen-specific CD8+ T cells that can eliminate IDO-expressing tumor cells, APCs, and MDSCs, potentiating the effector arm of the immune system to induce robust antitumor immune responses. Importantly, IDO vaccine could also enhance responses against a tumor antigen–specific vaccine and showed significant antitumor activity even against non–IDO-expressing tumors. These preclinical findings together with the clinical data (31, 32) suggest that IDO vaccine has a significant promise for cancer immunotherapy.
Supplementary Material
Acknowledgments
We are grateful to Jeannie and Tony Loop for their generous support to SNK's laboratory. We acknowledge the Flow Cytometry & Cell Sorting Shared Resource in Georgetown University, which is partially supported by NIH/NCI grant P30-CA051008 and NIH S10 grant, S10OD016213.
This work is supported by IO Biotech through a sponsored nonclinical research agreement with Samir N. Khleif.
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
Footnotes
Note: Supplementary data for this article are available at Cancer Immunology Research Online (http://cancerimmunolres.aacrjournals.org/).
Authors' Disclosures
M.H. Andersen reports personal fees from IO Biotech during the conduct of the study; grants and personal fees from IO Biotech outside the submitted work; in addition, M.H. Andersen has a patent for INDOLEAMINE 2,3-DIOXYGENASE BASED IMMUNOTHERAPY issued. A.W. Pedersen reports other support from IO Biotech ApS during the conduct of the study; other support from IO Biotech ApS outside the submitted work. M.-B. Zocca reports personal fees and other support from IO Biotech during the conduct of the study; personal fees from Valo Therapeutics outside the submitted work. S.N. Khleif reports grants and other support from IOBiotech during the conduct of the study; grants and other support from Syndax, Lycera; other support from Northwest Biotherapeutics, Advaxis, EMD Serono, GSK, UbiVac, McKinsey, Georgiammune, KAHR Medical, CytomX, NewLink Genetics, AratingaBio, CanImGuide, aMoon, Aummune, Incyte, Adaptive Biotech, Israel Biotech Fund, Livzon Mabpharm USA, Tessa Therapeutics, AgonOx, Autolus, Bayer, and Rheo Medicine; and grants from MedImmune outside the submitted work. No disclosures were reported by the other authors.
Authors' Contributions
R. Nandre: Formal analysis, investigation. V. Verma: Conceptualization, formal analysis, supervision, validation, investigation, visualization, methodology, writing–original draft, writing–review and editing. P. Gaur: Investigation. V. Patil: Investigation. X. Yang: Investigation. Z. Ramlaoui: Investigation. N. Shobaki: Investigation. M.H. Andersen: Resources, writing–review and editing. A.W. Pedersen: Resources, writing–review and editing. M.-B. Zocca: Resources, writing–review and editing. M. Mkrtichyan: Supervision. S. Gupta: Conceptualization, data curation, formal analysis, supervision, validation, visualization, methodology, writing–original draft, writing–review and editing. S.N. Khleif: Conceptualization, resources, data curation, supervision, funding acquisition, validation, visualization, project administration, writing–review and editing.
References
- 1. Andersen MH. The balance players of the adaptive immune system. Cancer Res 2018;78:1379–82. [DOI] [PubMed] [Google Scholar]
- 2. Guerrouahen BS, Maccalli C, Cugno C, Rutella S, Akporiaye ET. Reverting immune suppression to enhance cancer immunotherapy. Front Oncol 2019;9:1554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Baban B, Chandler PR, Sharma MD, Pihkala J, Koni PA, Munn DH, et al. IDO activates regulatory T cells and blocks their conversion into Th17-like T cells. J Immunol 2009;183:2475–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Opitz CA, Somarribas Patterson LF, Mohapatra SR, Dewi DL, Sadik A, Platten M, et al. The therapeutic potential of targeting tryptophan catabolism in cancer. Br J Cancer 2020;122:30–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Liu XH, Zhai XY. Role of tryptophan metabolism in cancers and therapeutic implications. Biochimie 2021;182:131–9. [DOI] [PubMed] [Google Scholar]
- 6. Munn DH, Mellor AL. Indoleamine 2,3 dioxygenase and metabolic control of immune responses. Trends Immunol 2013;34:137–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Holmgaard RB, Zamarin D, Li Y, Gasmi B, Munn DH, Allison JP, et al. Tumor-expressed IDO recruits and activates MDSCs in a Treg-dependent manner. Cell Rep 2015;13:412–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Wang XF, Wang HS, Wang H, Zhang F, Wang KF, Guo Q, et al. The role of indoleamine 2,3-dioxygenase (IDO) in immune tolerance: focus on macrophage polarization of THP-1 cells. Cell Immunol 2014;289:42–8. [DOI] [PubMed] [Google Scholar]
- 9. Zhao ZG, Xu W, Sun L, Li WM, Li QB, Zou P. The characteristics and immunoregulatory functions of regulatory dendritic cells induced by mesenchymal stem cells derived from bone marrow of patient with chronic myeloid leukaemia. Eur J Cancer 2012;48:1884–95. [DOI] [PubMed] [Google Scholar]
- 10. Huang H, Dawicki W, Zhang X, Town J, Gordon JR. Tolerogenic dendritic cells induce CD4+CD25hiFoxp3+ regulatory T cell differentiation from CD4+CD25-/loFoxp3- effector T cells. J Immunol 2010;185:5003–10. [DOI] [PubMed] [Google Scholar]
- 11. Van den Eynde BJ, van Baren N, Baurain J-F. Is there a clinical future for IDO1 inhibitors after the failure of epacadostat in melanoma? Annu Rev Cancer Biol 2020;4:241–56. [Google Scholar]
- 12. Sorensen RB, Berge-Hansen L, Junker N, Hansen CA, Hadrup SR, Schumacher TN, et al. The immune system strikes back: cellular immune responses against indoleamine 2,3-dioxygenase. PLoS One 2009;4:e6910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Sorensen RB, Hadrup SR, Svane IM, Hjortso MC, Thor Straten P, Andersen MH. Indoleamine 2,3-dioxygenase specific, cytotoxic T cells as immune regulators. Blood 2011;117:2200–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Munir S, Larsen SK, Iversen TZ, Donia M, Klausen TW, Svane IM, et al. Natural CD4+ T-cell responses against indoleamine 2,3-dioxygenase. PLoS One 2012;7:e34568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Lin KY, Guarnieri FG, Staveley-O'Carroll KF, Levitsky HI, August JT, Pardoll DM, et al. Treatment of established tumors with a novel vaccine that enhances major histocompatibility class II presentation of tumor antigen. Cancer Res 1996;56:21–6. [PubMed] [Google Scholar]
- 16. Verma V, Shrimali RK, Ahmad S, Dai W, Wang H, Lu S, et al. PD-1 blockade in subprimed CD8 cells induces dysfunctional PD-1(+)CD38(hi) cells and anti-PD-1 resistance. Nat Immunol 2019;20:1231–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Rammensee H, Bachmann J, Emmerich NP, Bachor OA, Stevanovic S. SYFPEITHI: database for MHC ligands and peptide motifs. Immunogenetics 1999;50:213–9. [DOI] [PubMed] [Google Scholar]
- 18. Habara-Ohkubo A, Takikawa O, Yoshida R. Cloning and expression of a cDNA encoding mouse indoleamine 2,3-dioxygenase. Gene 1991;105:221–7. [DOI] [PubMed] [Google Scholar]
- 19. Milcent B, Josseaume N, Riller Q, Giglioli I, Rabia E, Deligne C, et al. Presence of T cells directed against CD20-derived peptides in healthy individuals and lymphoma patients. Cancer Immunol Immunother 2019;68:1561–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Verma V, Jafarzadeh N, Boi S, Kundu S, Jiang Z, Fan Y, et al. MEK inhibition reprograms CD8(+) T lymphocytes into memory stem cells with potent antitumor effects. Nat Immunol 2021;22:53–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hornyak L, Dobos N, Koncz G, Karanyi Z, Pall D, Szabo Z, et al. The role of indoleamine-2,3-dioxygenase in cancer development, diagnostics, and therapy. Front Immunol 2018;9:151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Berrong Z, Mkrtichyan M, Ahmad S, Webb M, Mohamed E, Okoev G, et al. Antigen-specific antitumor responses induced by OX40 agonist are enhanced by the IDO inhibitor indoximod. Cancer Immunol Res 2018;6:201–8. [DOI] [PubMed] [Google Scholar]
- 23. Zoso A, Mazza EM, Bicciato S, Mandruzzato S, Bronte V, Serafini P, et al. Human fibrocytic myeloid-derived suppressor cells express IDO and promote tolerance via Treg-cell expansion. Eur J Immunol 2014;44:3307–19. [DOI] [PubMed] [Google Scholar]
- 24. Yu J, Du W, Yan F, Wang Y, Li H, Cao S, et al. Myeloid-derived suppressor cells suppress antitumor immune responses through IDO expression and correlate with lymph node metastasis in patients with breast cancer. J Immunol 2013;190:3783–97. [DOI] [PubMed] [Google Scholar]
- 25. Veglia F, Gabrilovich DI. Dendritic cells in cancer: the role revisited. Curr Opin Immunol 2017;45:43–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Aranda F, Vacchelli E, Eggermont A, Galon J, Sautes-Fridman C, Tartour E, et al. Trial Watch: peptide vaccines in cancer therapy. Oncoimmunology 2013;2:e26621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Jou J, Harrington KJ, Zocca MB, Ehrnrooth E, Cohen EEW. The changing landscape of therapeutic cancer vaccines-novel platforms and neoantigen identification. Clin Cancer Res 2021;27:689–703. [DOI] [PubMed] [Google Scholar]
- 28. Sutherland SIM, Ju X, Horvath LG, Clark GJ. Moving on from sipuleucel-T: new dendritic cell vaccine strategies for prostate cancer. Front Immunol 2021;12:641307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Novitskiy SV, Moses HL. Turn off the IDO: will clinical trials be successful? Cancer Discov 2012;2:673–5. [DOI] [PubMed] [Google Scholar]
- 30. Zhai L, Bell A, Ladomersky E, Lauing KL, Bollu L, Sosman JA, et al. Immunosuppressive IDO in cancer: mechanisms of action, animal models, and targeting strategies. Front Immunol 2020;11:1185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Iversen TZ, Engell-Noerregaard L, Ellebaek E, Andersen R, Larsen SK, Bjoern J, et al. Long-lasting disease stabilization in the absence of toxicity in metastatic lung cancer patients vaccinated with an epitope derived from indoleamine 2,3 dioxygenase. Cancer Res 2014;20:221–32. [DOI] [PubMed] [Google Scholar]
- 32. Kjeldsen JW, Iversen TZ, Engell-Noerregaard L, Mellemgaard A, Andersen MH, Svane IM. Durable clinical responses and long-term follow-up of stage III-IV non-small-cell lung cancer (NSCLC) patients treated with IDO peptide vaccine in a phase I study-a brief research report. Front Immunol 2018;9:2145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Kjeldsen JW, Lorentzen CL, Martinenaite E, Ellebaek E, Donia M, Holmstroem RB, et al. A phase 1/2 trial of an immune-modulatory vaccine against IDO/PD-L1 in combination with nivolumab in metastatic melanoma. Nat Med 2021;27:2212–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Orabona C, Grohmann U, Belladonna ML, Fallarino F, Vacca C, Bianchi R, et al. CD28 induces immunostimulatory signals in dendritic cells via CD80 and CD86. Nat Immunol 2004;5:1134–42. [DOI] [PubMed] [Google Scholar]
- 35. Shen Z, Zhou S, Wang Y, Li RL, Zhong C, Liang C, et al. Higher intratumoral infiltrated Foxp3+ Treg numbers and Foxp3+/CD8+ ratio are associated with adverse prognosis in resectable gastric cancer. J Cancer Res Clin Oncol 2010;136:1585–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Curran MA, Montalvo W, Yagita H, Allison JP. PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Natl Acad Sci U S A 2010;107:4275–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Groth C, Hu X, Weber R, Fleming V, Altevogt P, Utikal J, et al. Immunosuppression mediated by myeloid-derived suppressor cells (MDSCs) during tumour progression. Br J Cancer 2019;120:16–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Kang C, SY J, Song SY, Choi EK. The emerging role of myeloid-derived suppressor cells in radiotherapy. Radiat Oncol J 2020;38:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Harden JL, Egilmez NK. Indoleamine 2,3-dioxygenase and dendritic cell tolerogenicity. Immunol Invest 2012;41:738–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Sharma MD, Baban B, Chandler P, Hou DY, Singh N, Yagita H, et al. Plasmacytoid dendritic cells from mouse tumor-draining lymph nodes directly activate mature Tregs via indoleamine 2,3-dioxygenase. J Clin Invest 2007;117:2570–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.