Abstract
Background
The highly complex pathogenesis of Type 1 Diabetes is driven by several immune cell types with both effector and regulatory characteristics, which ultimately ends in the destruction of the insulin-producing beta cells. There are multiple layers of interaction between these immune cell populations and the pancreatic islets.
Scope of review
In this review article, we aim to discuss important recent insights into the multiple layers of interaction between immune cell populations and the pancreatic islets. Specifically, we discuss the environment where immune and beta cell interactions occur, the key cell types and molecules involved, and the outcomes of these interactions.
Major conclusions
Most of the molecular mechanisms underlying aberrant immune cell activation and impaired immune tolerance remain insufficiently understood, which hinders the development of efficient prevention and treatment strategies. In order to overcome this knowledge gap, a better understanding of the complex interactions of immune cells and beta cells, including both the underlying protective and pathogenic mechanisms is urgently required.
Keywords: Type 1 diabetes, Autoimmunity, Autoimmune diabetes, Beta cells, Immune cells, Immune Regulation
Graphical abstract
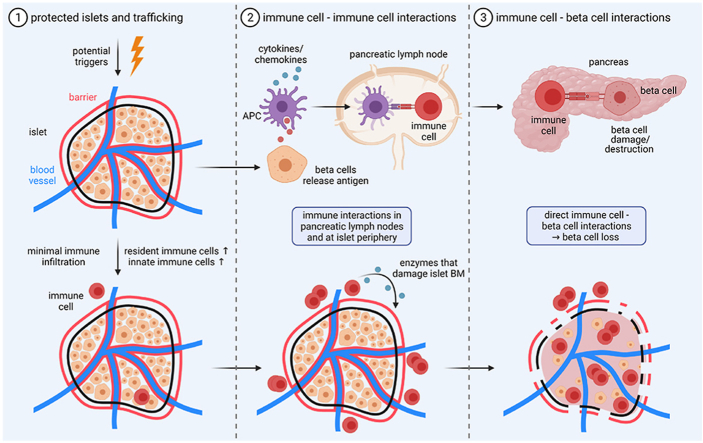
1. Introduction
To dissect beta cell and immune cell interactions in type 1 diabetes (T1D), it is important to first consider under which circumstances beta cells and immune cells can either physically interact or be exposed to soluble factors secreted by either cell type. Beta cells are uniquely localized within the islets of Langerhans in the pancreas, which also contain glucagon-producing alpha cells, somatostatin-producing delta cells, and, in fewer numbers, ghrelin-producing epsilon cells and pancreatic polypeptide (PP) cells. Islets constitute around 1–2% of the pancreas volume, with the remainder of the pancreas consisting predominantly of acinar cells, which produce digestive enzymes. These mini-organs are physically separated from the surrounding acinar tissue by a connective tissue network and a mixture of extracellular matrix proteins, the composition of which can differ between species. This barrier gives islets “immune privilege” and, under normal circumstances, prevents immune cells present within the exocrine pancreas from physically reaching the beta cells. To penetrate this barrier, immune cells must first be recruited to the islets, a process mediated by a range of different chemokines. Following extravasation, immune cells accumulate at the islet periphery and breakdown of the barrier is required before they can infiltrate and directly interact with the endocrine cells. This review expands on each of these areas exploring the environment where immune and beta cell interactions occur, the key cell types and molecules involved, and the outcomes of these interactions. It should be noted that the undoubtedly important topic of T1D genetics has been addressed in several recent reviews and is beyond the scope of this review. Similarly, a plethora of phase I-III clinical studies have been completed in the last decade that target immune cells either directly, or indirectly. These have been expertly reviewed in [7]. Moving forward it will be important to map these success and failures on to the lessons we are learning regarding the communication be-tween immune and beta cells in T1D.
2. The Islet environment
2.1. Endocrine cells and innervation
The cytoarchitecture of islets differs between rodents and humans: the former has a central core of beta cells, surrounded by a mantle of alpha and delta, while the latter has a more heterogeneous distribution of these three endocrine cells [95]. Sympathetic nerves surround the islet in rodents, while acetylcholine is supplied to beta cells through parasympathetic fibers, distributed throughout the core. In contrast, in humans, sympathetic fibers target the islet vasculature while parasympathetic fibers are rare: alpha cells control the supply of acetylcholine to beta and delta cells [156,157,191].
2.2. Vasculature
Islets receive between 10 and 15% of the pancreatic blood flow and intra-islet endothelia are a conduit for the delivery of glucose and transport of insulin and carriage of oxygen and removal of waste products. Interestingly, however, there is also evidence showing that endothelial cells have a role in stimulating insulin transcription and secretion and beta cell proliferation. This is thought to be via secretion of soluble factors, production of extracellular matrix proteins and direct contact of cell-surface molecules [142,143]. Migration of T cells across blood vessel walls and the ECM of islets requires T cell activation and is mediated primarily by β1-integrins [51]. Extravasation is most likely to occur from post-capillary venules surrounding the outside of the islet periphery, with extravasated cells tracking back along the vessels to reach the islet.
2.3. Islet barrier
Islets are surrounded by a layer of extra-cellular matrix (ECM), comprised of a basement membrane (BM) and an interstitial matrix (IM), which separates the endocrine tissue from the exocrine and functions a critical barrier for immune cells to overcome to infiltrate an islet and destroy beta cells. Studies into the development of T1D in the non-obese diabetic (NOD) mouse model and human patients have demonstrated a global loss of the peri-islet BM and IM components in these subjects, but only at sites of leukocyte infiltration [98]. At these sites, cathepsin S, W and C activity has been identified, suggesting that these proteases are important for allowing penetration of the protective barrier of islets [98]. Differences in the composition of the BM and IM between mice and man have been reported [98,141], with humans reportedly having a double basement membrane [197]. This added barrier could provide a strengthened physical defense to immune cells, preventing their extravasation within the confines of the islet. As such, the islets in humans are likely to have a degree of immune privilege, which can be lost following sufficient immune cell recruitment to the peri-islet region and release of degradative enzymes resulting in the breakdown of the barrier [98]. These differences in the composition of the protective layer surrounding the islet could explain both the disparities between the infiltration seen in mice and men, and the apparent protracted destruction of beta cells in humans.
2.4. Islet resident lymphocytes
As with many organs, tissue-resident immune cells, which are of great importance for understanding the immune regulation of these tissues, are also found within islets. Regarding pancreatic immune cells, a first key study used flow cytometry analysis of islets isolated from 38 perfused pancreases (diabetes free, islet autoantibody negative) to quantify alpha-, beta-, T-, natural killer (NK-) and B cells [150]. Up to 80% of lymphocytes were T cells, (the majority of which were CD8+), which also had a pronounced memory phenotype. There were, however, low frequencies of B and NK-cells in most islet preparations [150]. In order to improve our understanding of the cellular and molecular events that precede and lead to beta cell loss in T1D, including pancreas-resident immune cells, the Human Pancreas Analysis Program (HPAP) investigates the human pancreas using deep phenotyping methods such as imaging mass cytometry. These efforts resulted in several recent publications revealing important insights into the human pancreas such as changes in immune cell populations including novel cellular states during the progression of T1D [42,52,200,208].
3. Immune cells involved in type 1 diabetes
3.1. T cells
Although multiple immune cell populations contribute to the full clinical manifestation of T1D, it is well established that T cells play an essential role in the development of islet autoimmunity and progression to overt diabetes in both human and animal models. Indeed, CD8+ T cells are the most common islet tissue-resident and infiltrating immune cells [150,204]. However, the specific contribution of T cell subsets and their interplay with other immune and islet cells, remains insufficiently understood. Several studies revealed T cell infiltration within the islets of individuals with T1D and prediabetic and diabetic NOD mice. In NOD mice, very early onset of T cell infiltration in the pancreatic islets was detected at only 10–15 days of age [137]. Furthermore, transfer experiments using NOD mice showed that the destruction of pancreatic beta cells depends on a combination of CD4+ and CD8+ T cells. The transfer of both T cell subsets from NOD mice to immunodeficient recipients induced T1D, while CD4+ or CD8+ T cells alone failed to do so [147]. In humans, segmental pancreas transplants from non-diabetic, HLA-identical donors resulted in the recurrence of T1D, caused by T cells, in particular the CD8+ subset [188].
The exact mechanisms triggering and driving T cell infiltration into the islets remain to be elucidated. However, several partially redundant pathways seem to be involved. ‘Recent studies suggest both antigen-specific and non-antigen specific mechanisms are involved, as well as a role of vascular adhesion molecules [164] and chemokines.
In line with their critical contribution to T1D pathogenesis, T cells are among the first immune cells to infiltrate the pancreas: in NOD islets CD4+ T cells could be detected as early as four weeks, CD8+ T cells and B cells after six weeks and all other immune cell subsets after eight weeks [26]. The first infiltrating CD4+ T cells are specific for islet antigens with the vast majority recognizing an IAg7-restricted insulin epitope of the B chain comprising residues 9–23 (B:9–23) [41,43,201]. Other antigens recognized by pancreas infiltrating T cells include glutamic acid decarboxylase (GAD), insulinoma-associated protein 2 (IA-2), zinc transporter 8 (ZnT8) and heat shock protein 60 (Hsp60), all of which are derived from pancreatic islets and correlate with the presence of autoantibodies [107]. Infiltrating CD4+ T cells interact with islet-resident antigen-presenting cells (APCs) [20], resulting in changes in the islet microenvironment, such as upregulation of adhesion molecules on the endothelium of islet blood vessels (VCAM1) and beta cells (ICAM1), which attracts more T cells, including non-antigen-specific cells [19]. While the majority of T cells infiltrate the pancreas during the progression of islet autoimmunity, CD8+ T cells specific to preproinsulin were found in the healthy exocrine pancreas [9]. In addition to these very early infiltrates, islet antigen-specific CD8+ T cells were found in islets from patients with both recent onset and long duration T1D [40]. Additional murine studies analyzing dynamics of islet infiltration in an antigen-specific manner revealed that antigen-specific CD8+ T cells infiltrate the islets as early as CD4+ T cells, but in lower numbers [108,206] and the infiltration of these cells depends on MHCI expression in the pancreas [2,167].
The exact mechanisms of beta cell killing in T1D have been investigated in several studies. Activated autoreactive CD8+ T cells can induce beta cell death in a contact-dependent manner through cytotoxic degranulation [97] as well as perforin and granzyme molecules [193]. In addition, pro-inflammatory cytokines such as TNF-α and IFN-γ are produced by both pancreas-resident CD4+ and CD8+ T cells [32,187]. These cytokines are toxic to beta cells, and can additionally create a feedback loop of cytokine production and beta cell killing by activating pro-inflammatory macrophages [22].
The functional interaction of infiltrating T cells with the islet microenvironment are, among others, highlighted by the induction of an effector memory phenotype [31,69] and the upregulation of KLRG1 and CD127 [125] in CD8+ T cells which are located in islets with high levels of IFNγ and granzyme B.
Intervention studies in humans and mice confirmed the important role of T cells in the development of T1D. In NOD mice, an anti-CD3 antibody could induce long-term remission of overt autoimmunity [29]. A similar approach in humans revealed evidence that an anti-CD3 antibody could reduce beta cell destruction in patients with recent onset of T1D [28]. Encouragingly, recently published clinical trials in individuals at high-risk of developing T1D have demonstrated that treatment with the anti-CD3 antibody, Teplizumab, significantly delays progression to clinical disease implying a beta cell protective effect [79,180]. Taken together these findings support the conclusion that T cells are key effector cells in T1D and have a significant impact on beta cell viability.
3.2. Regulatory T cells
Although T cells play an essential pathological role in T1D development and progression, one T cell subset, regulatory T cells (Tregs), are a major contributor to interfere with autoimmune activation and progression. In both humans and mice, mutations in the FOXP3 gene, which is the master regulator of Treg development, maintenance and function, result in the development of fatal autoimmunity, including T1D [10,94].
The first human studies indicating that Tregs are impaired in T1D showed that CD4+CD25+ T cells from patients with T1D have a reduced ability to suppress T cell proliferation in vitro [110]. Later studies showed that Tregs from T1D patients have impaired IL-2 receptor signaling [113].
Furthermore, there is accumulating evidence that impairments in Treg induction, stability and function play a role as key drivers of islet autoimmune activation and the progression to clinical T1D [168,171,173]. Specifically, during the onset of human and murine islet autoimmunity insulin-specific Tregs are significantly reduced and Treg induction and stability are impaired. Mechanistically, a set of differentially expressed T cell specific microRNAs (miRNAs) was identified as important mediators of Treg dysfunction, plasticity, and instability during islet autoimmunity. A miR181a-mediated increase in signal strength of stimulation and costimulation links Nuclear factor of activated T cells 5 (NFAT5) with impaired Treg induction and autoimmune activation [173]. Furthermore, during islet autoimmunity a miR142–3p/Tet2/Foxp3 axis in CD4+ T cells leads to impaired epigenetic remodeling and consequently interferes with the efficient induction of Tregs and results in impaired Treg stability [168]. Of note, pilot studies on a specific targeting of these aberrantly expressed miRNAs resulted in reduced islet autoimmunity in mouse models, supporting the important role of Tregs for T1D pathogenesis and suggesting these pathways as potential targets for future prevention strategies [168,172,173].
The important role of Tregs for the prevention of T1D is in line with an accelerated disease progression in NOD mice lacking Tregs [163] and the finding that induced Treg deficiency resulted in increased T cell and NK cell infiltration in the pancreas [56]. Importantly, the inhibitory effect of Tregs was not limited to CD4+ T cells. Increased Treg numbers in the pancreas resulted in reduced numbers of CD8+ T cells, and the IFNγ production of these cells was decreased [117]. The first successful intervention strategies in the murine system support the importance of these findings. The injection of low dose IL-2 for five consecutive days in prediabetic NOD mice increased Treg numbers in the pancreas 1.5-fold. It was able to stop T1D progression and normalized blood glucose levels in 60% of treated animals [72]. Importantly, due to the high expression of the IL-2R alpha chain (CD25), Tregs require less IL-2 to boost their function and expansion than their effector counterparts [215]. Therefore, low-dose IL-2 administration is a promising strategy to specifically promote Tregs in therapeutic approaches. In the human system, the first dose-defining trial of low-dose IL-2 administration in patients with established T1D was conducted in 2013 [76]. The daily administration of low-dose IL-2 for five consecutive days effectively expanded Tregs in peripheral blood, with almost no effect on effector T cells or NK cells, which was not the case with administration of higher doses. An ongoing trial (Interleukin-2 Therapy of Autoimmunity in Diabetes (ITAD)) is now assessing the effect of low-dose IL-2 on endogenous beta cell function in newly diagnosed children and adolescents (aged 6-18 y) [118].
Despite these studies suggesting that Tregs of T1D patients can be enhanced by IL-2 treatment, an approach of combining IL-2 with rapamycin resulted in transient beta cell dysfunction despite an increase in Tregs [114]. While no differences were observed in effector T cell subsets, the frequencies of natural killer cells and eosinophils increased.
Another therapeutic approach was based on the discovery that human Tregs can be expanded ex vivo on a clinical scale using beads coated with antibodies for CD3 and CD28 in the presence of recombinant IL-2. The first clinical trial transferring ex vivo-expanded autologous Treg to children with recent-onset T1D showed that administration of Tregs was safe and tolerable and resulted in a reduced insulin requirement, prolonged the survival of β-cells and higher C-peptide levels [119,120]. A second trial investigated the transfer of expanded Tregs in adult patients with recent-onset T1D [14]. Treg levels in the blood peaked in the first 2 weeks, followed by a loss of 75% of the peak level during the first 3 months, before numbers stabilized for more than 1 year. Furthermore, the expanded Tregs retained their T cell receptor diversity and demonstrated enhanced functional activity. Of note, several patients had stable C-peptide levels and insulin use for up to 2 years after Treg injection.
Despite these promising results, a phase II clinical trial (ClinicalTrials.gov Identifier: NCT02691247) investigating the safety and efficacy of CLBS03, a cell product comprised of autologous, ex vivo expanded Tregs, did not meet its primary endpoint of preserving C-peptide levels at one year versus placebo. However, several aspects should be considered when classifying this failed study. 1) One particular problem in T1D is that by the time of diagnosis patients might not have enough viable beta cells for the pancreas to function properly. This means that the treatment of recent-onset T1D patients (<100 days after diagnosis) with expanded Tregs might already be too late. Here, studies in individuals in an earlier disease phase or ideally during islet autoimmunity could potentially increase the efficacy of such approaches. 2) The use of polyclonally expanded Tregs might critically limit their therapeutic potential in T1D. Here, approaches using antigen-specific Tregs might have a greater potential to suppress autoreactive T cells in T1D and therefore stop or delay the disease progression [49,89,140,170]. However, so far these approaches have shown only limited success in humans and will require further research in the future. Therefore, several approaches are under way to develop and optimize islet-specific Tregs using genetic engineering, either with chimeric antigen receptors or transgenic T cell receptors [151]. Here, the goal is an off-the-shelf product for adoptive therapy that would overcome the need of isolating and expanding patient-specific Tregs.
Taken together these findings support the conclusion that Tregs play a key role in the maintenance of immune tolerance and consequently the prevention of islet autoimmunity and T1D. Treg targeting could be a promising approach for the development of future intervention strategies in autoimmune diseases such as T1D.
3.3. T follicular helper cells
A counterpart of Tregs, which could play a crucial role in the development of autoimmunity alongside effector T cells, are T follicular helper (TFH) cells. These cells are a subset of CD4+ T cells that play an integral part in humoral immunity by providing help to B cells to undergo maturation and class-switching to produce high-affinity antibodies. TFH cells are characterized by the expression of their master transcription factor B cell lymphoma 6 (Bcl6) together with high expression of the chemokine-receptor CXCR5 and low expression of CCR7. The differential expression of these two chemokine receptors enables TFH cells to leave the T cell zone and enter the B cell follicle in secondary lymphoid organs, where they can interact with B cells.
Due to their essential contribution to humoral immunity, TFH cells have been studied in the context of autoimmune diseases that are characterized by the presence of high-affinity autoantibodies, such as T1D. Several studies indicate a functional role of TFH cells in T1D: one study, employing a transgenic TCR model indicated that antigen-specific CXCR5+CD4+ T cells can induce diabetes after transfer into recipient mice expressing the same antigen under the insulin promoter in the beta cells [92]. Additionally, more descriptive studies show enhanced secretion of IL-21 (the signature cytokine of TFH cells) by CD4+ T cells of T1D patients [55] and enhanced frequencies of circulating TFH precursors in the peripheral blood of children who recently developed islet autoantibodies but have not progressed to clinical T1D yet [171]. Similarly, gene expression studies reveal gene signatures resembling TFH/TH17 cell responses in infants prior to the development of islet autoantibodies [77].
Of note, targeting the TFH cytokine IL-21 to halt the progression of T1D recently showed considerable success in mice and humans. In diabetic NOD mice, treatment with an anti-IL-21 monoclonal antibody combined with liraglutide could effectively reverse hyperglycemia [162]. In a phase II clinical trial, the same combination could preserve beta cell function in patients with recently diagnosed T1D [199]. Furthermore, it has been shown in NOD mice and patients with recent-onset T1D, that TFH cells can be efficiently decreased by costimulation blockade using a CTLA-4–Ig fusion protein [47]. The sensitivity to this treatment was linked to the expression of the inducible costimulatory molecule (ICOS), indicating that TFH subtypes could have the potential to predict clinical response to an immunotherapy.
Importantly, several studies indicate that the Ig isotype and even the IgG subtype of islet autoantibodies correlate with disease progression [81,82,146]. TFH cells can be divided into different subsets according to their chemokine receptor expression (Th1-, Th2- and Th17-like TFH cells) and differ in their ability to provide help to B cells [11,112,135,189]. These different TFH subsets can also influence the antibody isotype production by B cells [135,189]. Even though data on TFH cell subsets in T1D is limited, a recent study indicates that Th2-like TFH cells are enhanced in children with recent development of islet autoantibodies or with newly diagnosed T1D, while Th1-like and Th17-like TFH subset frequencies were unaltered [171]. These findings highlight the need for more detailed analysis of TFH cells in T1D, since significant differences in subsets might be missed in an analysis that focuses solely on global TFH cell markers.
Importantly, TFH/B cell interactions are not limited to the secondary lymphoid organs. Tertiary lymphoid structures (TLS) form at sites of inflammation, can contain active germinal centers and contribute to the clearance of infections and anti-tumor immune responses [63]. TLS have been associated with many autoimmune diseases, such as rheumatoid arthritis [84], Sjögren's syndrome [103] and Hashimoto Thyroiditis [5]. Likewise, studies in NOD mice have demonstrated the presence of peri-islet TLS that seem to develop within the disease course and recent studies have highlighted the presence of TLS in human T1D with structural similarities to those in NOD mice [99]. Data from both human and murine studies indicate the presence of TLS only in the early phase of the disease, while they disappear once the beta cells are destroyed and the immune response subsides [99]. Importantly, these TLS in the NOD pancreas were shown to contain active germinal centers that enable antigen-driven somatic hypermutation of B cells directly at the site of inflammation [91]. Interestingly, analysis of the BCR repertoire in NOD mice showed a polyclonal repertoire in the pancreas, while single islets showed oligoclonality [91]. These findings indicate TLS as a site of somatic maturation of specific B cells enabling a more diverse autoimmune attack, and are in line with findings from other autoimmune diseases [84,136].
3.4. B cells
In line with the proposed contribution of TFH/B cell interactions to the development of T1D, the latter have also increasingly become the focus of recent research projects. Although dogma states that T1D is a T cell-mediated disease, there is a growing body of evidence suggesting that B cells may also play an important role in the development of this condition. In 2009, a detailed analysis of the immune cells infiltrating islets in recent-onset (<2 y duration) T1D revealed that CD20+ B cells were one of the largest populations present within and/or around endocrine tissue, second only to cytotoxic CD8+ T cells. This was particularly true in islets with evidence of established beta cell loss [204]. NOD mice models also support the hypothesis that B cells function to promote the survival of cytotoxic T cells within inflamed islets. For example, genetic or antibody driven depletion of B cells arrests diabetes development at the pre-insulitis stage [174], which is abrogated when treating them with NOD B cells [175]. Furthermore, depletion of B cells with anti-CD20 [83,212] or anti-CD22 therapies [57] also has protective effects. Despite a reasonable wealth of evidence in mouse models, the role of these lymphocytes in humans is still relatively unknown and continues to be contested [13]. Indeed, a case report of T1D development in an individual with X-linked agammaglobulinemia (B-lymphocyte deficiency) [124] is often cited to argue against these cells playing a critical role in the pathogenesis of T1D [13].
It is now clear, however, that the extent of B cell infiltration of islets is directly associated with age at diabetes onset. Immune infiltration of islets is more aggressive in individuals diagnosed <7 y and includes significant numbers of CD20+ B cells (CD20Hi), compared with patients diagnosed 13 y, in whom a more pauci-immune response is observed, with very few CD20+ B cells (CD20Lo) [4,105]. Examination of beta cell destruction in <7 y versus 13 y reveals that individuals diagnosed <7 y have significantly fewer insulin-positive islets and reduced beta cell area compared to those diagnosed 13 y [25,105]. This implies that the presence of B cells within the inflammatory infiltrate is associated with a more aggressive and rapid destruction of beta cells. Based on age at diagnosis, we could predict that the patient with X-linked agammaglobulinemia, who developed diabetes aged 14 y [124], likely would have been a CD20Lo donor, in whom B cells appear to play less of a role in disease development.
Evidence of B cell involvement in T1D pathogenesis is not limited to the pancreas. Dufort et al. found that B cell and neutrophil immune phenotypes in peripheral blood samples were strongly dependent on age, as B cell levels were able to predict the rate of progression in young subjects [46]. Smith et al. have also reported a loss of insulin-binding, anergic B cells in the periphery of some first-degree relatives and all prediabetic and new-onset (<1 y) T1D patients tested [182]. They speculate too, that this might reflect a relocalization of these autoreactive B cells to other anatomical sites, such as the pancreas.
The B cell depletion therapy Rituximab has also been utilized with some success to preserve beta cell function in recent-onset T1D, as measured by serum C-peptide decline over 18mo [144,145]. Interestingly, post-analysis work also showed a split between ‘responders’ and ‘non-responders’ of the therapy [111], which may be linked with patients having CD20Hi vs CD20Lo phenotypes [145], as the former may benefit more from B cell depletion.
3.4.1. B cell function in T1D
It is widely thought that the antigen-presenting capacity of B cells, via interactions with CD4+ and CD8+ T cells and the secretion of pro-inflammatory cytokines (including TGF-β and IFNγ) [75,192], rather than the production of autoantibodies, potentiates the autoimmune process in T1D. Indeed, strategies to deplete autoantibodies in humans failed to abrogate disease [115,169], while studies in the NOD mouse indicate activation of autoreactive T cells is driven by interactions with antigen-presenting B cells [121,207]. For example, B cell-specific deletion of the MHC class II I–Ag7 (homolog to human DQ molecules) prevented disease development in NOD mice [139].
Additional studies into the NOD mouse found that B cells could interact with autoreactive CD8+ T cells in the pancreatic lymph node, which led to proliferative expansion and differentiation into effector cells. Knocking out MHC class I in B cells or restricting B cell receptor specificity to an irrelevant antigen diminishes the expansion of autoreactive CD8+ T cells and suppresses disease development, suggesting that antigen capture and presentation by B cells are critical to this process [121,178].
Supporting this, CD20 depletion led to functional modulation of macrophages and splenic dendritic cells (DCs) in NOD mice, accompanied by a reduction in the production of cytokines IFN and IL-17 by CD4+ and CD8+ T cells [83]. Furthermore, BCR L chain genes from B cells isolated from lymphocytic aggregates attacking islets in NOD mice had a complex pancreatic repertoire, indicating that antigen specificity is important to the pathogenesis of disease [91].
3.5. Antigen-presenting cells
Until now, this review has discussed the role of the adaptive immune system in T1D. While both T- and B cells play a crucial role in disease pathogenesis, the innate immune system, in particular myeloid antigen-presenting cells (APCs), may provide a key link between stressed beta cells and the adaptive immune response: facilitating islet infiltration. Macrophages and dendritic cells (DCs) are professional APCs, which both circulate in the periphery and reside in islets [195].
Macrophages can have an anti-inflammatory or pro-inflammatory phenotype [128] and play an important role in tissue homeostasis and antigen presentation to T cells. Islet resident macrophages can also contribute to tissue development and remodeling [211]. They sense their microenvironment by detecting extracellular ATP and constantly probe their surroundings by engulfing vesicles released by beta cells [21,203]. These vesicles may contain immunogenic peptides, which could be presented to and recognized by auto-reactive T cells. Immunohistochemical analysis of pancreas samples from individuals with recent-onset T1D showed that macrophages contribute to the insulitic milieu, though these are likely to have been recruited rather than tissue-resident [204].
Conversely, after uptake of immunogenic antigens, DCs migrate to draining lymph nodes to activate antigen-specific T cells. However, the frequency in which DCs reside in islets remains unclear.
Although there is evidence of these APCs displaying genetic, qualitative or quantitative changes in NOD mice and human T1D, their contribution to disease pathogenesis remains unclear. It is conceivable, however, that a failure in these cells results in them connecting with, priming and propagating the adaptive immune system against beta cells [217].
For example, in NOD mice, CD11c expressing mononuclear phagocytes (MNPs) stimulated the trafficking of lymphocytes to islets [127]. The inflammatory milieu in infiltrated islets leads to the recruitment of these cells [19,26,130], which then produced more than 20 different chemokines. The binding of these chemokines to respective receptors on T- and B cells facilitated their recruitment and extravasation into the islets [164]. This is in line with the finding that T cell extravasation occurs close to islet resident CD11c+ MNPs [165].
On the other hand, single-cell multi-omics of human pancreatic islets revealed a novel subset of ductal cells in T1D donors but not in nondiabetic or islet autoantibody positive donors [52]. These cells express high levels of MHC class II and interferon pathways, are surrounded by pancreas-resident immune cells and transcriptionally resemble tolerogenic DCs. They exhibit a tolerogenic response to chronic T cell infiltration of the islets and seem to be an ultimately unsuccessful attempt to limit the T cell response responsible for destroying beta cells.
One additional aspect to consider, regardless of the cell type presenting the antigens, is the role of neoantigens for the activation of immune cells. Several recent studies suggested that islet autoimmunity also targets neoantigens expressed by the beta cells, and which are therefore not available for thymic tolerance induction [155]. Multiple factors such as the pro-inflammatory environment in the diabetic pancreas can contribute to cellular stress, affecting splicing, degradation and modification of proteins. This mechanism consequently results in the generation of beta cell derived neoepitopes and can contribute to autoimmunity.
4. Type 1 diabetes endotypes
Importantly, many features of T1D, including the multitude of cell types described above, exhibit a high degree of heterogeneity. When considering interactions between beta cells and immune cells, it is important to factor this heterogeneity as well as disease endotypes which can be defined based on it. As mentioned previously, the degree of immune-cell infiltration of islets is directly linked with age of diabetes onset: individuals aged <7 y have a hyperimmune phenotype (with significant B cell [CD20Hi] inflammation), while individuals aged 13 y have a less aggressive, pauci-immune phenotype (with only a minor contribution from B cells [CD20Lo]) [4,105]. Added to this, beta cell expression and localization of proinsulin is also associated with age at onset and, critically, aligns with patient immunophenotypes [104]. This adds weight to the suggestion that two type 1 diabetes endotypes (T1DE) exist – one which is defined by significant levels of inflammation and widespread beta cell dysfunction (T1DE 1) and a second which has a more moderate immune response and appears to have less beta cell distress (T1DE 2). Although the term ‘endotype’ is beginning to become common parlance for discussing T1D pathogenesis [8], there are still many questions to consider. For example, are islets in individuals with T1DE 1 more vulnerable to immune attack? Or is the less mature immune system of children <7 y responsible for the more aggressive assault in these individuals? Moreover, the NOD mouse is often criticized for the overwhelming level of infiltration observed in islets, which tends not to reflect what is seen in humans [86]. Perhaps this is because the model is more reflective of young-onset T1DE1, which may be important for the design and testing of future therapeutics. However, it is important to remember that the onset of T1D in the NOD mouse occurs in adults, not juveniles. The implications of this are unclear. Finally, if we are to use the information regarding pancreatic endotypes to help guide treatment in the future, it will be necessary to predict an individual's endotype at, or ideally before, onset of T1D. As such we must identify easily accessible biomarkers, that can be monitored in at-risk individuals, that correlate with pancreatic endotype. One such biomarker, could be islet autoantibodies (directed against insulin, GAD65, IA2 and ZnT8) which have been measured routinely in natural history studies of T1D and are increasingly being used clinically to help with diabetes disease classification. Recent modelling studies examining age-at-seroconversion and islet autoantibody type have revealed that an early age of seroconversion (<2 y) with insulin autoantibodies or multiple islet autoantibodies are associated with a high probability of developing diabetes with 5 y, so diabetes is diagnosed <7 y [50,100]. Whereas, those with a later age of seroconversion and GAD65 autoantibodies are associated with a lower risk of progression to T1D within 5 y, so likely diagnosed >13 y. Perhaps islet autoantibodies will provide us with a tool to determine pancreatic endotype and focus treatment accordingly, but again this requires further investigation. It is important to note that for this strategy to be successful, a population screening program would have to be initiated and strides are being made towards this [12,90,216].
5. Immune-immune-cell interactions within the pancreas
Pancreatic lymph nodes are thought to be critical contributors to the pathogenesis of T1D, as they are the site for the initial priming of autoreactive T cells. In BDC2.5 T cell receptor (TCR) transgenic mice, which recognize a natural beta cell antigen, activated T cells were restricted to the pancreatic islets and the pancreas draining lymph nodes [80]. Naïve BDC2.5 T cells, transferred into non-transgenic recipients, proliferated only in pancreatic lymph nodes, and this was observed before T cells infiltrated the islets. These observations indicate that beta cell antigens are specifically transported to the pancreas draining lymph nodes, where they activate T cells and trigger islet infiltration.
Studies in the NOD mouse model have further highlighted the importance of the pancreatic lymph nodes for priming of beta cell reactive T cells. The excision of pancreatic lymph nodes in 3 weeks old mice almost completely prevented the development of insulin autoantibodies, insulitis and T1D onset, while removing the spleen or mesenteric lymph nodes had no effect on T1D development [62]. Interestingly, the effect was not observed in 10w old mice, indicating that the priming of autoreactive T cells occurs in NOD mice from 3w of age.
In addition to the critical priming of autoreactive lymphocytes in the pancreatic lymph nodes which leads to the upregulation of cytotoxic effector markers, there is a concurrent upregulation of these markers in the inflamed islets independent of beta cells [69]. This suggests that the primed autoreactive lymphocytes are trafficking to the islets. Indeed, altered gene expression profiles in pancreatic lymph nodes have been reported in NOD mice, and occurred before the detection of T cells in the islet periphery [6]. These early alterations were characterized by increased expression of genes related to lymphocyte homing, DC attraction, angiogenesis and apoptosis.
Furthermore, the importance of pancreatic lymph nodes in the emergence and maintenance of autoimmune cell populations has recently been highlighted in NOD mice [66]. Here, a stem-like autoimmune progenitor population has the ability to self-renew and gives rise to CD8+ autoimmune mediators which migrate to the pancreas, and destroy beta cells. Since these pancreatic autoimmune mediators are short-lived, a continuous seeding of progenitors to the pancreas is crucial for sustained beta cell destruction and T1D pathogenesis. The presence of such stem-like immune cell populations is one possible explanation for the failure of several treatment approaches and strategies aimed at targeting these progenitors could emerge as promising immunotherapeutic interventions for T1D.
Antigen presentation in the lymph nodes can be mediated by hematopoietic and non-hematopoietic antigen-presenting cells such as lymph node stromal cells (LNSCs). LNSCs in pancreatic lymph nodes of T1D patients and diabetic NOD mice show phenotypic alterations. They exhibit an increased antigen-presentation potential and a more tolerogenic phenotype, which may indicate an attempt to compensate DC-related tolerance defects during the development of islet autoimmunity [149]. However, recent-onset T1D donors exhibited a decreased B cell follicle frequency and reduced follicular dendritic cell networks within their pancreatic lymph node germinal centers. A phenomenon not seen in longer duration T1D donors, suggesting that this defect could be related to an inability to control autoimmunity close to the onset of disease [205].
Another layer of complexity is revealed when examining pancreatic lymph nodes located at the head and the tail of the pancreas. These differ regarding their microvasculature, extracellular matrix, and the abundance of B cells and APCs - which are more abundant in the pancreatic lymph nodes located in the head of the pancreas [106].
T cells isolated from pancreatic lymph nodes of long-term T1D patients showed a high degree of clonal expansion when compared to non-diabetic controls [93]. These T cells recognized the insulin A 1–15 epitope, indicating that insulin may indeed be a target antigen priming autoreactive T cells in the pancreatic lymph nodes and contributing to T1D development.
The critical role of pancreatic lymph nodes for the development of T1D by priming autoreactive T cells has also been successfully targeted by prevention strategies in NOD mice. α-galactosylceramide, which stimulates NKT cells to produce a soluble factor that induces DC maturation and accumulation in the pancreatic lymph nodes [33]. These accumulated DCs attract and tolerize autoreactive T cells and inhibit the development of T1D.
6. Chemokines
In addition to the multitude of cell types contributing to the development of T1D, how these cells are regulated and how they interact with each other is another layer of complexity. Here, chemokines are one important means of regulation.
6.1. The role of chemokines in guiding immune–immune cell interactions
In addition to their function in guiding immune cell trafficking, chemokines are essential mediators of immune cell interactions. Various studies have demonstrated that chemokines are directly involved in shaping the immunological synapse and contribute to the fine tuning of T cell activation [194]. Similar to the migration of lymphocytes into tissues, the basis for migration of T cells within the tissue towards APCs is likewise dependent on cell polarization. This polarization of cells so they have a leading edge and a trailing edge, is accompanied by a redistribution of receptors in the cell membrane mediated by chemokines [44]. This redistribution of molecules increases the sensitivity of the leading edge of the T cell to peptide-MHC or anti-CD3 stimulation when compared with the trailing edge [138,202]. While naïve T cells can form immunological synapses in the absence of chemokines, studies demonstrated that the presence of chemokines (specifically CXCL12 and CCL21, ligands for the chemokine receptors CXCR4 and CCR7 respectively) on the DC surface can increase the number of T cells that are capable of responding to antigens presented by APCs [17,60,102].
Additionally, chemokines can act as T cell costimulatory molecules [183]. Specifically, chemokine secretion by APCs leads to the accumulation of CCR5 and CXCR4 at the immunological synapse. Importantly, this redistribution of chemokine receptors to the immunological synapse affects T cell-APC interaction, T cell responsiveness to chemotactic gradients, T cell proliferation and pro-inflammatory cytokine production [27,38,53,131].
The ability of these chemokine receptors to modulate the sensitivity of T cell responsiveness to antigenic stimulation has direct implications for autoimmunity. Accordingly, an enrichment of CCR5 expressing T cells can be observed in the target tissues of various autoimmune diseases (e.g. multiple sclerosis [179,185] and rheumatoid arthritis [65,67]) and homozygosity for the inactive variant of CCR5 (Δ32) was associated either with delayed disease onset or a milder disease course. Furthermore, mice lacking the CCR5 receptor showed a threefold increase in cardiac allograft survival time in the absence of additional immunosuppression [64]. Although studies on the role of CCR5 in T1D are rare, a cohort study from Poland indicated a negative association between the Δ32 variant and T1D [181]. Furthermore, findings from NOD mice treated with IL-4 indicate that a reduction in CCR5 positive T cell frequency in the spleen and pancreas might contribute to the beneficial effect of IL-4 treatment [23].
In addition to T cell activation, chemokines are likewise involved in the regulation of immune tolerance. In this regard, recent studies have demonstrated that CCR4 expression on Tregs contributes to their suppressive function [85]. CCL22, one of the two known ligands for CCR4, is constitutively and highly expressed by DCs in the lymph nodes, helping to mediate stable DC-Treg contacts in lymphoid organs, outcompeting naïve T cells for DC contacts and thereby inhibiting their activation. While neither CCR4 deficient nor CCL22 deficient mice show spontaneous autoimmunity, both animal models show strongly enhanced T cell activation after vaccination, excessive inflammation and enhanced tumor clearance [152]. Furthermore, lymph node DCs are able to contribute to the induction of Tregs and their retention in the lymph nodes [198], again highlighting the importance of CCL22 in maintaining self-tolerance and preventing autoimmunity.
Studies on the role of CCL22 in T1D are limited and have shown divergent results. Montane et al. show the recruitment of Tregs to the pancreas after adenovirus-mediated overexpression of CCL22 in islets and consequently reduced signs of inflammation and protection from T1D in NOD mice [132]. Furthermore, adenoviral CCL22-overexpression in islet allografts was shown to attract Treg and prevent diabetes recurrence [133]. We could show recently that the cytokine GM-CSF strongly induces CCL22 in vitro and in vivo [148]. Interestingly, administration of GM-CSF was previously shown to delay diabetes onset in NOD mice and to enhance Treg suppression [30]. These findings indicate a potential role of the GM-CSF CCL22 pathway in diabetes development. Contradictory to these findings, Kim et al. identified a population of CCR4 expressing autoimmune memory CD4+ T cells present during the progression of T1D in NOD mice. In their hands, the systemic neutralization of CCL22 (also known as macrophage-derived chemokine [MDC]) by a polyclonal antibody inhibited the development of the disease and reduced the numbers of CCR4+ T cells within the pancreas [96]. The divergence in the two studies may result, in part, from the difference in manipulation of CCL22 expression namely specifically in the pancreas versus systemically. These findings also highlight the complexity of chemokine involvement in autoimmunity due to their diverse actions based on the location and, most likely, the cell type secreting the chemokine. These apparent discrepancies owing to the location of chemokine expression may likely represent their functional differences concerning the guidance of immune cell interaction and priming on the one hand, and mediating immune cell migration on the other.
6.2. Chemokine mediated immune cell infiltration into the pancreas
The infiltration of the pancreatic islets by motile immune cells is a precondition for initiating the autoimmune attack in T1D and the destruction of the beta cells, indicating the importance of immune cell migration for the pathogenesis of T1D. In general, the directed migration and extravasation of lymphocytes to sites of inflammation is a key feature of the immune system which is particularly enabled and controlled by chemokine signaling [158]. Consequently, several chemokines and their receptors contribute to the development of various autoimmune diseases [159]. Indeed, more than half of the about 50 members of the human chemokine superfamily and their murine homologs, are involved in T1D pathogenesis [166]. The expression of many chemokines expressed in pancreatic islets during autoimmunity is driven by IFNγ [18]. The direct contribution of chemokine signaling to the onset of T1D has been demonstrated using the γHV68 protein M3, which efficiently blocks the action of several chemokines [196]. The islet-specific expression of M3 reduced islet infiltration and destruction and prevented the development of T1D in NOD mice [122]. The importance of chemokine signaling for the development and progression of T1D is also highlighted by reported associations of chemokine ligand and receptor genes within IDD risk loci. For example, CXCR6 is located in the human IDDM22 risk locus [164], and its ligand CXCL16 shows an association with the murine Idd4 risk locus [88]. However, CXCR6 deficiency was not sufficient to prevent islet infiltration in NOD mice, again indicating a high degree of redundancy in chemokine signaling [164].
Based on these findings, several studies investigated the potential of targeting chemokine signaling to prevent islet infiltration. These approaches could efficiently prevent the immune cell migration to the islets before infiltration, but not once immune cells were present in the islets [39,134,213].
Several chemokines are involved in the recruitment of T cells to the pancreatic islets. Some play redundant roles or can exhibit both tolerogenic and pathogenic roles in the development of T1D [164].
The ligands of the chemokine receptor CCR7, CCL19 and CCL21, which are in a steady state involved in the migration of immune cells to the lymph nodes [58], are abundantly expressed around prediabetic islets in NOD mice during the development of T1D [16]. This has been shown to drive the infiltration of the islets by islet antigen-specific T cells [167]. Consequently, CCR7 deficiency in NOD mice prevents the development of T1D [123].
Studies on CXCR4, a chemokine receptor that is abundantly expressed on immune cells, were less conclusive. While expression of its ligand CXCL12 on islet endothelial cells was shown to limit diabetogenic T cell binding in vitro [176] and to enhance beta cell survival in STZ treated mice [214], inhibition of CXCL12 showed divergent results [1,126]. These divergent results most likely stem from differences in the used mouse models and cell types expressing the chemokine. E.g. the STZ model of beta cell destruction might be more prone to reveal effects on beta cell survival while underappreciating opposing effects related to immune regulation.
The strongest evidence for chemokine involvement in T1D is coming from the chemokine CXCL10 and its receptor CXCR3. CXCR3 is mainly expressed on CD8+ effector T cells and CD4+ Th1 cells. This drives their migration to sites of inflammation [73], such as CXCL9 and CXCL10 producing islets [61]. While studies on the expression of CXCL10 in serum of pre-diabetic at risk individuals or patients with T1D showed divergent results, the vast majority of studies both in humans and mouse models showed enhanced CXCL10 levels related to T1D (summarized in [34. In line with these results, the beta cell-specific expression of CXCL10 caused islet infiltration in T1D-resistant mice [153], while its blockade delayed the disease in NOD mice [134] and the combination therapy with anti-CD3 antibody lead to persistent remission in different mouse models of T1D [101]. Similarly, CXCR3 deficiency had a protective effect in mice upon viral induction of T1D [61]. Another study showed that CXCR3 is involved in early T cell migration to the pancreas, but CXCR3 deficiency in T cells in this study did not prevent islet infiltration in a murine T1D model, most likely due to redundancies in chemokine signaling [164]. In these studies, differences in the models used could account for the divergent results. While Frigerio et al. [61] used an inducible model with the LCMV glycoprotein expressed under the rat insulin promoter which leads to the immune-mediated destruction of insulin-producing cells after LCMV infection, Sandor et al. [164] used wildtype NOD mice with spontaneous disease development. The differences in disease severity and progression in the two models highlights, that different mechanisms might be involved in the immune responses of both models. Importantly, CXCR3 is not only expressed on Th1 cells but can also be expressed on Tregs, mediating their migration to the pancreas [213]. Accordingly, Tan et al. identified a prominent CXCR3+Tbet+ Treg population in the islets of NOD mice and showed that ablation of this cell population enhanced disease severity and onset and was even able to overcome the usually present sex-bias in disease development in NOD mice [190].
The role of chemokine signaling in T1D is not limited to T cells but also drives the migration of MNPs and B cells. MNP recruitment is mainly mediated via the chemokine receptors CCR2 and CCR5 and their ligands CCL2 and CCL3, CCL4 and CCL5, respectively [71] while migration of B cells into the islets is thought to be mediated by CXCR5 and its ligand CXCL13 [3]. Studies on the blockade of CCR2, CCR5 and CXCL13 are limited and have shown divergent results in mouse models of T1D [78,116,184].
Combined, these studies highlight the difficulties in studying effects of chemokines on T1D, including local versus systemic effects and differences in disease models and emphasize the necessity to evaluate effects on different T cell subtypes as well as immune cell populations apart from T cells.
7. Mediators of direct beta cell – immune cell interactions
7.1. The role of classical HLA-I
Increased islet expression of HLA class I was one of the earliest reported histological features of T1D, first noted in 1985 [15] and further characterized in 1987, when evidence of aberrant MHC class II expression was also highlighted [59]. Over 30 years later, it is now widely accepted that hyperexpression of HLA class I is a key pathological feature of T1D and is thought to be critical in early disease progression, facilitating the effective engagement of autoreactive CD8+ cytotoxic T cells, which recognize specific islet autoantigens [87,154] (Figure 1). HLA-I expression is dramatically elevated in all endocrine cell types within the insulin-containing islets of T1D donors, but expression is dramatically reduced in the insulin-deficient islets suggesting the presence of beta cells is required for the hyperexpression [154]. Islet endothelial cell expression may also be key to facilitating CD8+ cytotoxic T cell recruitment. Studies in mouse models, have demonstrated that cognate MHC/peptide complexes are presented by pancreatic endothelial cells, which acquire the antigen (insulin) from nearby beta cells [167]. Homing of the insulin-specific CD8+ T cells to islets was abrogated in mice that lacked MHC class I expression, had impaired insulin secretion or were unable to present the specific insulin peptide. Triggering of the TCR and chemokine receptors activated integrin expression, further facilitating the adhesion of T cells to the pancreatic endothelium and, as such could, contribute to homing of activated T cells to the islets [167]. The endothelial homing receptor VCAM-1 has also been demonstrated to be important for the infiltration of T cells expressing A4β1 integrin into pancreatic islets of transgenic mice [74].
Figure 1.
Role of classical and non-classical HLA-I expression in beta cell – immune cell interactions in T1D. High expression of HLA-I can have both protective and detrimental effects. In T1D hyperexpression of classical and non-classical HLA class I in beta cells is thought to be critical in early disease progression, facilitating the effective engagement of autoreactive CD8+ cytotoxic T cells, which recognize specific islet autoantigens. (created with BioRender).
7.2. The role of non-classical HLA-I – HLA-E, HLA-F and HLA-G
7.2.1. HLA-E
HLA-E is constitutively expressed in most cell types and interacts with β2-microglobulin to present peptides from the leader sequence of HLA-I molecules. This peptide-bound HLA-E molecule can then interact with CD94/NKG2A/B/C present on the surface of NK cells. Engagement of either CD94/NKG2A and CD94/NKG2B transduces an inhibitory signal to the NK cells reducing cytolytic activity. In contrast, CD94/NKG2C causes activation of NK cells. Levels of HLA-E expression are normally low within pancreatic islets. However, upregulation of HLA-E has been demonstrated in insulin-containing islets of individuals with T1D (Fig. 1) [37,209]. However, despite a global increase in expression across these islets, alpha cells, in particular, seem to express a higher level of HLA-E when compared to beta cells [209]. It was proposed that this could preferentially protect alpha cells from immune attack. However, histopathological studies of insulitis in donors with T1D have suggested that there are very few NK cells present in or around the islets [204]. Although one study did report the presence of NK cells within the infiltrate in a subset of T1D donors [45]. It is conceivable that NK cells may have a role before clinical onset of T1D, and support for this comes from emerging genetic studies that indicate a strong NK cell signature that tracks with rate of progression to T1D [210]. This brings into question the role HLA-E plays at the islet site as it is unclear whether it functions to protect cells from infiltrating NK cells expressing the HLA-E ligands, CD94/NKG2A and CD94/NKG2B, or if it could prime them for destruction by CD94/NKG2C+ NK cells. This can only be uncovered by studying pancreatic material from individuals who are at-risk of T1D and have evidence of insulitis, which unfortunately, is extremely rare
7.2.2. HLA-F
Relatively little is known about HLA-F, and this particular non-classical HLA-I molecule, can be present in multiple different forms within the cytosol or at the cell surface. HLA-F can form a complex with β2-microglobulin and peptide; it can be present as an open conformation (OC) (without bound peptide) either alone, or as a homodimer; or it can form a heterodimer with classical HLA-I molecules [109]. The OC HLA-F molecules are capable of binding peptides between 7 and 30 residues long, and these can be derived from the exogenous environment [68]. These exogenous peptides can be internalized and processed before being presented on classical HLA-I molecules. As such, this pathway has been suggested to play a role in the cross-presentation of peptides.
A range of different ligand partners are associated with these different HLA-F conformations. These include both inhibitory (KIR3DL1/2; ILT2 and ILT4) and activating ligand pairs ((KIR3DS1; KIR2DS4). These ligands are predominantly localized to NK cells and T cells. The OC of HLA-F has also been demonstrated to have the ability to bind TCRs. The expression of HLA-F is tightly regulated and is typically localized within the cytoplasm, with very few cell types exhibiting surface expression. Like HLA-E, HLA-F is also increased in the insulin-containing islets of individuals with T1D. Intriguingly, in these islets there is evidence of very clear surface localization. A phenomenon not seen in donors without diabetes [154,209]. Other cell types with surface HLA-F expression include activated B, T and NK cells; EBV-transformed B cells; virus-infected cells and first and second trimester migratory and extravillous cytotrophoblasts [109]. In these latter cells, HLA-F has been shown to have an important immunomodulatory role. Indeed, HLA-F expression has been associated with a negative prognosis in several different types of cancer, which suggests that cancerous cells can in part negatively regulate anti-tumour immune responses through this pathway. Functional evidence to determine if HLA-F has an immunomodulatory role in T1D is lacking, and it will also be important to determine if any, or all, of these ligands are present on infiltrating immune cells. The outcome of these interactions would determine if the balance of signals is in favour of inactivation or activation of the cognate immune cell and the consequence of this for the beta cells remains to be determined.
7.2.3. HLA-G
HLA-G expression is restricted to limited cell types including cytotrophoblasts in the placenta, corneal embryonic and nail matrix mesenchymal stem cells and pancreatic beta cells [24,35]. Ligands of this molecule include ILT-2, ILT4, KIR2DL4 and CD160, found on NK, DC, B and T cells. Binding of HLA-G to these ligands can inhibit NK and T cell proliferation, DC maturation and the cytolytic and proliferative responses of antigen-specific CD4+ and CD8+ T cells. HLA-G essentially acts to conceal the cell it is expressed on from immune surveillance. Insulin-containing islets of T1D donors have elevated HLA-G expression when compared to donors without diabetes [209]. The increased expression of HLA-G on islet cells in donors with T1D suggests that the islets are attempting to reduce their visibility to the infiltrating immune cells. It remains to be determined whether HLA-G expression varies amongst the different endocrine cell types and if this can explain their differential susceptibility to immune attack.
7.3. HLA Class-II
A comprehensive study of islet beta cells from donors with recent-onset T1D demonstrated that there was increased mRNA and protein expression of MHC Class II and Class II antigen presentation pathway components. Specifically, immunohistological studies of donor pancreas from three independent cohorts showed Class II HLA protein and its transcriptional regulator Class II MHC trans-activator protein to be expressed by a subset of insulin+ beta cells found within inflamed islets [161]. This supported earlier studies from the 1980's reporting this phenomenon [15,59]. Surface expression of Class II is suggestive of an antigen-presenting roll for beta cells, and this may directly play a role in the autoimmune response [161]. It is speculated that secretion of IFNγ by CD8+ T cells may promote this beta cell Class II expression. It is unclear, however, whether Class II-expressing beta cells take up and process exogenous proteins and then present peptides via Class II to CD4+ T cells in the same way as professional APCs. Indeed, evidence of expression of CD80 or CD86 costimulatory molecules was not detected in Class II-expressing beta cells [161].
7.4. Expression of the checkpoint inhibitor PDL1 in islets
The emergence of checkpoint inhibitors, particularly PD1 and PDL1 blocking antibodies, as effective treatments for various cancers has also led to a series of important insights into T1D pathogenesis. Monitoring a clinical cohort of individuals treated with checkpoint inhibitor therapies revealed that 0.2–11% of patients present with diabetes following PDL1/PD1 antibody treatment [186]. Of the patients presenting with diabetic ketoacidosis (DKA), with very low c-peptide levels, >50% had evidence of islet autoantibodies, and a large proportion were shown to have the T1D genetic risk allele HLA-DR4 [186]. As such the clinical features of this form of diabetes demonstrate that checkpoint inhibitor-induced diabetes shows a greater similarity with T1D versus T2D. Efforts from the community to investigate the expression of PD1 and PDL1 in the pancreas post-mortem or in pancreas biopsies of individuals with T1D revealed that PDL1 expression is increased in the insulin-containing islets (ICIs) of donors with diabetes [36]. In contrast, donors without diabetes and the islets devoid of beta cells in donors with T1D, did not exhibit detectable PDL1 expression. The expression of PDL1 in the ICIs was associated with the presence of infiltrating immune cells, implying that a factor released from these cells (e.g. IFNγ) could be responsible for the increased PDL1 expression. Indeed, in vitro studies confirmed that treatment of human beta cell lines with IFNs induced expression of PDL1 at both the mRNA and protein level [36]. As inhibition of activation and induction of exhaustion/apoptosis pathways in T cells occurs following cross-linking of PDL1 and PD1, it could be hypothesized that increased PDL1 expression on stressed beta cells helps to defend against infiltrating PD1+ T cells. In keeping with this, upregulation of PDL1 was observed in beta cells of NOD mice that were more resistant to immune attack [160]. However, the observation that PD1 expression is reduced on T cells at the onset of childhood T1D may suggest that dysregulation of this pathway could predispose these children to beta cell destruction [70].
Furthermore, inhibition of this defense mechanism by treatment with PD1/PDL1 blocking antibodies would accelerate beta cell loss by releasing the brakes on the PD1+ T cell. The observation that patients with checkpoint-related diabetes have elevated genetic risk of T1D and some individuals were shown to be islet autoantibody positive before treatment, suggest that pre-screening of individuals for biomarkers of T1D (such as the T1D genetic risk score [177] and islet autoantibodies [54] before treatment with checkpoint inhibitors may be a sensible precaution moving forward.
In keeping with this hypothesis, findings from mouse models revealed that injection of 8w NOD mice with a beta cell targeting adeno-associated virus (AAV) construct containing both PDL1 and abatacept (PDL1-CTLA-4-Ag) prevented the development of autoimmune diabetes and preserved beta cell mass [48]. The PDL1-CTLA4Ig AAV was, however, unable to prevent diabetes in early onset diabetic NOD mice with >200 mg/dl blood glucose levels for 2 consecutive weeks. It did, however, protect islet allographs [48].
On balance the majority of the non-classical HLA-I receptor–ligand interactions appear to transduce inhibitory signals to their respective immune cells. The increased expression of non-classical HLA-I molecules and PD-L1 in the islets of T1D donors implies that the islets are attempting to defend themselves against non-specific cytotoxicity from infiltrating immune cells.
So, in a scenario where the beta cell is recognized by specific autoreactive CD8+ T cells via the classical HLAI; does this detrimental pathway win out over these defense pathways? This is a difficult question to address in humans but is an important one to consider. The recent development of pancreatic islet microtissues by InSphero [129] may provide one platform in which this could be assessed. Islet microtissues pretreated with factors that enhance non-classical HLAI expression (and likely also classical HLAI) could be co-cultured with CD8+ T cell subsets with varying specificities to assess the impact this has on islet viability. The outcome of such studies may inform strategies to help protect beta cells from islet-specific CD8+ T cell attack in the future.
Of note, the exact mechanisms of how immune ligands on beta cells, including checkpoint inhibitors such as PDL1, are involved in T1D pathogenesis remain insufficiently understood and require further examination in the future.
8. Conclusion
The highly complex pathogenesis of T1D is driven by several immune cell types with both effector and regulatory characteristics, which ultimately ends in the destruction of the insulin-producing beta cells (Figure 2). There are multiple layers of interaction between these immune cell populations and the pancreatic islets. However, most of the molecular mechanisms underlying aberrant immune cell activation and impaired immune tolerance remain largely unexplored, complicating the development of efficient prevention and treatment strategies. In order to overcome this knowledge gap, a better understanding of the complex interactions of immune cells and beta cells, including both the underlying protective and pathogenic mechanisms is urgently required. The trafficking of immune cells to the pancreas is regulated by multiple pathways, but might serve as promising target for strategies preventing immune infiltration to the islets in the first place. However, while this might be a promising prevention approach, the high level of redundancy in these pathways, and the potential of induction of unwanted broad immunosuppression, are major challenges. Another important aspect is the heterogeneity of islets within the pancreas, in particular with regard to the varying degree of immune infiltration, and the phenotypes of the infiltrating immune cells. A better understanding of these differences could allow the identification of pathogenic and, perhaps even more importantly protective pathways, which could be targeted for future strategies aiming at the prevention of islet autoimmunity and T1D.
Figure 2.
Cellular and molecular mechanisms contributing to the prevention or development of T1D. Beta cell antigens are released by damaged beta cells. These antigens are processed by pancreas-resident antigen-presenting cells (APC). Following activation APCs migrate to the pancreatic lymph nodes where they prime pathogenic beta cell antigen-specific T cells. On the other hand, tolerogenic APCs can prime and expand Tregs which can directly suppress autoreactive T cells, including but not limited to the release of IL10 and TGFβ. Beta cell killing in the pancreas happens by autoreactive T cells, NK cells and macrophages, besides other through the release of cytokines such as IL1β, IFNγ and TNF. Furthermore, dendritic cells (DC) release IL12 to sustain effector functions of activated T cells and NK cells. Lastly, changes in the islet microenvironment as well as the release of chemokines by beta cells can recruit additional lymphocytes and macrophages, further driving the autoimmune process. (created with BioRender).
Conflict of interest
None
References
- 1.Aboumrad E., Madec A.M., Thivolet C. The CXCR4/CXCL12 (SDF-1) signalling pathway protects non-obese diabetic mouse from autoimmune diabetes. Clinical and Experimental Immunology. 2007;148(3):432–439. doi: 10.1111/j.1365-2249.2007.03370.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alkemade G.M., Clemente-Casares X., Yu Z., Xu B.Y., Wang J., Tsai S., et al. Local autoantigen expression as essential gatekeeper of memory T-cell recruitment to islet grafts in diabetic hosts. Diabetes. 2013;62(3):905–911. doi: 10.2337/db12-0600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ansel K.M., Harris R.B.S., Cyster J.G. CXCL13 is required for B1 cell homing, natural antibody production, and body cavity immunity. Immunity. 2002;16(1):67–76. doi: 10.1016/s1074-7613(01)00257-6. [DOI] [PubMed] [Google Scholar]
- 4.Arif S., Leete P., Nguyen V., Marks K., Nor N.M., Estorninho M., et al. Blood and islet phenotypes indicate immunological heterogeneity in type 1 diabetes. Diabetes. 2014;63(11):3835–3845. doi: 10.2337/db14-0365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Armengol M.P., Juan M., Lucas-Martin A., Fernandez-Figueras M.T., Jaraquemada D., Gallart T., et al. Thyroid autoimmune disease: demonstration of thyroid antigen-specific B cells and recombination-activating gene expression in chemokine-containing active intrathyroidal germinal centers. American Journal Of Pathology. 2001;159(3):861–873. doi: 10.1016/S0002-9440(10)61762-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Aspord C., Rome S., Thivolet C. Early events in islets and pancreatic lymph nodes in autoimmune diabetes. Journal of Autoimmunity. 2004;23(1):27–35. doi: 10.1016/j.jaut.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 7.Atkinson M.A., Roep B.O., Posgai A., Wheeler D.C.S., Peakman M. The challenge of modulating beta-cell autoimmunity in type 1 diabetes. Lancet Diabetes & Endocrinology. 2019;7(1):52–64. doi: 10.1016/S2213-8587(18)30112-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Battaglia M., Ahmed S., Anderson M.S., Atkinson M.A., Becker D., Bingley P.J., et al. Introducing the endotype concept to address the challenge of disease heterogeneity in type 1 diabetes. Diabetes Care. 2020;43(1):5–12. doi: 10.2337/dc19-0880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bender C., Rodriguez-Calvo T., Amirian N., Coppieters K.T., von Herrath M.G. The healthy exocrine pancreas contains preproinsulin-specific CD8 T cells that attack islets in type 1 diabetes. Science Advances. 2020;6(42) doi: 10.1126/sciadv.abc5586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bennett C.L., Christie J., Ramsdell F., Brunkow M.E., Ferguson P.J., Whitesell L., et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nature Genetics. 2001;27(1):20–21. doi: 10.1038/83713. [DOI] [PubMed] [Google Scholar]
- 11.Bentebibel S.E., Lopez S., Obermoser G., Schmitt N., Mueller C., Harrod C., et al. Induction of ICOS+CXCR3+CXCR5+ TH cells correlates with antibody responses to influenza vaccination. Science Translational Medicine. 2013;5(176):176ra132. doi: 10.1126/scitranslmed.3005191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Besser R.E.J., Ng S.M., Gregory J.W., Dayan C.M., Randell T., Barrett T. General population screening for childhood type 1 diabetes: is it time for a UK strategy? Archives of Disease in Childhood. 2021 doi: 10.1136/archdischild-2021-321864. [DOI] [PubMed] [Google Scholar]
- 13.Bloem S.J., Roep B.O. The elusive role of B lymphocytes and islet autoantibodies in (human) type 1 diabetes. Diabetologia. 2017;60(7):1185–1189. doi: 10.1007/s00125-017-4284-5. [DOI] [PubMed] [Google Scholar]
- 14.Bluestone J.A., Buckner J.H., Fitch M., Gitelman S.E., Gupta S., Hellerstein M.K., et al. Type 1 diabetes immunotherapy using polyclonal regulatory T cells. Science Translational Medicine. 2015;7(315):315ra189. doi: 10.1126/scitranslmed.aad4134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bottazzo G.F., Dean B.M., McNally J.M., MacKay E.H., Swift P.G., Gamble D.R. In situ characterization of autoimmune phenomena and expression of HLA molecules in the pancreas in diabetic insulitis. New England Journal of Medicine. 1985;313(6):353–360. doi: 10.1056/NEJM198508083130604. [DOI] [PubMed] [Google Scholar]
- 16.Bouma G., Coppens J.M., Mourits S., Nikolic T., Sozzani S., Drexhage H.A., et al. Evidence for an enhanced adhesion of DC to fibronectin and a role of CCL19 and CCL21 in the accumulation of DC around the pre-diabetic islets in NOD mice. European Journal of Immunology. 2005;35(8):2386–2396. doi: 10.1002/eji.200526251. [DOI] [PubMed] [Google Scholar]
- 17.Bromley S.K., Dustin M.L. Stimulation of naive T-cell adhesion and immunological synapse formation by chemokine-dependent and -independent mechanisms. Immunology. 2002;106(3):289–298. doi: 10.1046/j.1365-2567.2002.01441.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Burke S.J., Collier J.J. Transcriptional regulation of chemokine genes: a link to pancreatic islet inflammation? Biomolecules. 2015;5(2):1020–1034. doi: 10.3390/biom5021020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Calderon B., Carrero J.A., Miller M.J., Unanue E.R. Cellular and molecular events in the localization of diabetogenic T cells to islets of Langerhans. Proceedings of the National Academy of Sciences of the United States of America. 2011;108(4):1561–1566. doi: 10.1073/pnas.1018973108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Calderon B., Carrero J.A., Miller M.J., Unanue E.R. Entry of diabetogenic T cells into islets induces changes that lead to amplification of the cellular response. Proceedings of the National Academy of Sciences of the United States of America. 2011;108(4):1567–1572. doi: 10.1073/pnas.1018975108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Calderon B., Suri A., Miller M.J., Unanue E.R. Dendritic cells in islets of Langerhans constitutively present beta cell-derived peptides bound to their class II MHC molecules. Proceedings of the National Academy of Sciences of the United States of America. 2008;105(16):6121–6126. doi: 10.1073/pnas.0801973105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Calderon B., Suri A., Pan X.O., Mills J.C., Unanue E.R. IFN-gamma-dependent regulatory circuits in immune inflammation highlighted in diabetes. The Journal of Immunology. 2008;181(10):6964–6974. doi: 10.4049/jimmunol.181.10.6964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cameron M.J., Arreaza G.A., Grattan M., Meagher C., Sharif S., Burdick M.D., et al. Differential expression of CC chemokines and the CCR5 receptor in the pancreas is associated with progression to type I diabetes. The Journal of Immunology. 2000;165(2):1102–1110. doi: 10.4049/jimmunol.165.2.1102. [DOI] [PubMed] [Google Scholar]
- 24.Carosella E.D., Moreau P., Le Maoult J., Le Discorde M., Dausset J., Rouas-Freiss N. HLA-G molecules: from maternal-fetal tolerance to tissue acceptance. Advances in Immunology. 2003;81:199–252. doi: 10.1016/s0065-2776(03)81006-4. [DOI] [PubMed] [Google Scholar]
- 25.Carr A.L.J., Inshaw J.R.J., Flaxman C.S., Leete P., Wyatt R.C., Russell L.A., et al. Circulating C-peptide levels in living children and young people and pancreatic beta-cell loss in pancreas donors across type 1 diabetes disease duration. Diabetes. 2022;71(7):1591–1596. doi: 10.2337/db22-0097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Carrero J.A., Calderon B., Towfic F., Artyomov M.N., Unanue E.R. Defining the transcriptional and cellular landscape of type 1 diabetes in the NOD mouse. PLoS One. 2013;8(3) doi: 10.1371/journal.pone.0059701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cascio G., Martin-Cofreces N.B., Rodriguez-Frade J.M., Lopez-Cotarelo P., Criado G., Pablos J.L., et al. CXCL12 regulates through JAK1 and JAK2 formation of productive immunological synapses. The Journal of Immunology. 2015;194(11):5509–5519. doi: 10.4049/jimmunol.1402419. [DOI] [PubMed] [Google Scholar]
- 28.Chatenoud L. Immune therapy for type 1 diabetes mellitus-what is unique about anti-CD3 antibodies? Nature Reviews Endocrinology. 2010;6(3):149–157. doi: 10.1038/nrendo.2009.275. [DOI] [PubMed] [Google Scholar]
- 29.Chatenoud L., Thervet E., Primo J., Bach J.F. Anti-CD3 antibody induces long-term remission of overt autoimmunity in nonobese diabetic mice. Proceedings of the National Academy of Sciences of the United States of America. 1994;91(1):123–127. doi: 10.1073/pnas.91.1.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cheatem D., Ganesh B.B., Gangi E., Vasu C., Prabhakar B.S. Modulation of dendritic cells using granulocyte-macrophage colony-stimulating factor (GM-CSF) delays type 1 diabetes by enhancing CD4+CD25+ regulatory T cell function. Clinical Immunology. 2009;131(2):260–270. doi: 10.1016/j.clim.2008.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chee J., Ko H.J., Skowera A., Jhala G., Catterall T., Graham K.L., et al. Effector-memory T cells develop in islets and report islet pathology in type 1 diabetes. The Journal of Immunology. 2014;192(2):572–580. doi: 10.4049/jimmunol.1302100. [DOI] [PubMed] [Google Scholar]
- 32.Chen J., Grieshaber S., Mathews C.E. Methods to assess beta cell death mediated by cytotoxic T lymphocytes. Journal of Visualized Experiments. 2011;52 doi: 10.3791/2724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen Y.G., Choisy-Rossi C.M., Holl T.M., Chapman H.D., Besra G.S., Porcelli S.A., et al. Activated NKT cells inhibit autoimmune diabetes through tolerogenic recruitment of dendritic cells to pancreatic lymph nodes. The Journal of Immunology. 2005;174(3):1196–1204. doi: 10.4049/jimmunol.174.3.1196. [DOI] [PubMed] [Google Scholar]
- 34.Christen U., Kimmel R. Chemokines as drivers of the autoimmune destruction in type 1 diabetes: opportunity for therapeutic intervention in consideration of an optimal treatment schedule. Frontiers in Endocrinology. 2020;11 doi: 10.3389/fendo.2020.591083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cirulli V., Zalatan J., McMaster M., Prinsen R., Salomon D.R., Ricordi C., et al. The class I HLA repertoire of pancreatic islets comprises the nonclassical class Ib antigen HLA-G. Diabetes. 2006;55(5):1214–1222. doi: 10.2337/db05-0731. [DOI] [PubMed] [Google Scholar]
- 36.Colli M.L., Hill J.L.E., Marroqui L., Chaffey J., Dos Santos R.S., Leete P., et al. PDL1 is expressed in the islets of people with type 1 diabetes and is up-regulated by interferons-alpha and-gamma via IRF1 induction. EBioMedicine. 2018;36:367–375. doi: 10.1016/j.ebiom.2018.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Colli M.L., Ramos-Rodriguez M., Nakayasu E.S., Alvelos M.I., Lopes M., Hill J.L.E., et al. An integrated multi-omics approach identifies the landscape of interferon-alpha-mediated responses of human pancreatic beta cells. Nature Communications. 2020;11(1):2584. doi: 10.1038/s41467-020-16327-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Contento R.L., Molon B., Boularan C., Pozzan T., Manes S., Marullo S., et al. CXCR4-CCR5: a couple modulating T cell functions. Proceedings of the National Academy of Sciences of the United States of America. 2008;105(29):10101–10106. doi: 10.1073/pnas.0804286105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Coppieters K.T., Amirian N., Pagni P.P., Baca Jones C., Wiberg A., Lasch S., et al. Functional redundancy of CXCR3/CXCL10 signaling in the recruitment of diabetogenic cytotoxic T lymphocytes to pancreatic islets in a virally induced autoimmune diabetes model. Diabetes. 2013;62(7):2492–2499. doi: 10.2337/db12-1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Coppieters K.T., Dotta F., Amirian N., Campbell P.D., Kay T.W., Atkinson M.A., et al. Demonstration of islet-autoreactive CD8 T cells in insulitic lesions from recent onset and long-term type 1 diabetes patients. Journal of Experimental Medicine. 2012;209(1):51–60. doi: 10.1084/jem.20111187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Crawford F., Stadinski B., Jin N., Michels A., Nakayama M., Pratt P., et al. Specificity and detection of insulin-reactive CD4+ T cells in type 1 diabetes in the nonobese diabetic (NOD) mouse. Proceedings of the National Academy of Sciences of the United States of America. 2011;108(40):16729–16734. doi: 10.1073/pnas.1113954108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Damond N., Engler S., Zanotelli V.R.T., Schapiro D., Wasserfall C.H., Kusmartseva I., et al. A map of human type 1 diabetes progression by imaging mass cytometry. Cell Metabolism. 2019;29(3):755–768. doi: 10.1016/j.cmet.2018.11.014. e755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Daniel D., Gill R.G., Schloot N., Wegmann D. Epitope specificity, cytokine production profile and diabetogenic activity of insulin-specific T cell clones isolated from NOD mice. European Journal of Immunology. 1995;25(4):1056–1062. doi: 10.1002/eji.1830250430. [DOI] [PubMed] [Google Scholar]
- 44.del Pozo M.A., Cabanas C., Montoya M.C., Ager A., Sanchez-Mateos P., Sanchez-Madrid F. ICAMs redistributed by chemokines to cellular uropods as a mechanism for recruitment of T lymphocytes. The Journal of Cell Biology. 1997;137(2):493–508. doi: 10.1083/jcb.137.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dotta F., Fondelli C., Falorni A. Can NK cells be a therapeutic target in human type 1 diabetes? European Journal of Immunology. 2008;38(11):2961–2963. doi: 10.1002/eji.200838851. [DOI] [PubMed] [Google Scholar]
- 46.Dufort M.J., Greenbaum C.J., Speake C., Linsley P.S. Cell type-specific immune phenotypes predict loss of insulin secretion in new-onset type 1 diabetes. JCI Insight. 2019;4(4) doi: 10.1172/jci.insight.125556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Edner N.M., Heuts F., Thomas N., Wang C.J., Petersone L., Kenefeck R., et al. Follicular helper T cell profiles predict response to costimulation blockade in type 1 diabetes. Nature Immunology. 2020;21(10):1244–1255. doi: 10.1038/s41590-020-0744-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.El Khatib M.M., Sakuma T., Tonne J.M., Mohamed M.S., Holditch S.J., Lu B., et al. beta-Cell-targeted blockage of PD1 and CTLA4 pathways prevents development of autoimmune diabetes and acute allogeneic islets rejection. Gene Therapy. 2015;22(5):430–438. doi: 10.1038/gt.2015.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Elding Larsson H., Lundgren M., Jonsdottir B., Cuthbertson D., Krischer J., Di A.-I.T.S.G. Safety and efficacy of autoantigen-specific therapy with 2 doses of alum-formulated glutamate decarboxylase in children with multiple islet autoantibodies and risk for type 1 diabetes: a randomized clinical trial. Pediatric Diabetes. 2018;19(3):410–419. doi: 10.1111/pedi.12611. [DOI] [PubMed] [Google Scholar]
- 50.Endesfelder D., Zu Castell W., Bonifacio E., Rewers M., Hagopian W.A., She J.X., et al. Time-resolved autoantibody profiling facilitates stratification of preclinical type 1 diabetes in children. Diabetes. 2019;68(1):119–130. doi: 10.2337/db18-0594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Espinosa-Carrasco G., Le Saout C., Fontanaud P., Michau A., Mollard P., Hernandez J., et al. Integrin beta1 optimizes diabetogenic T cell migration and function in the pancreas. Frontiers in Immunology. 2018;9:1156. doi: 10.3389/fimmu.2018.01156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fasolino M., Schwartz G.W., Patil A.R., Mongia A., Golson M.L., Wang Y.J., et al. Single-cell multi-omics analysis of human pancreatic islets reveals novel cellular states in type 1 diabetes. Nat Metab. 2022;4(2):284–299. doi: 10.1038/s42255-022-00531-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Felce J.H., Parolini L., Sezgin E., Cespedes P.F., Korobchevskaya K., Jones M., et al. Single-molecule, super-resolution, and functional analysis of G protein-coupled receptor behavior within the T cell immunological synapse. Frontiers in Cell and Developmental Biology. 2020;8 doi: 10.3389/fcell.2020.608484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ferrat L.A., Vehik K., Sharp S.A., Lernmark A., Rewers M.J., She J.X., et al. A combined risk score enhances prediction of type 1 diabetes among susceptible children. Nature Medicine. 2020;26(8):1247–1255. doi: 10.1038/s41591-020-0930-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ferreira R.C., Simons H.Z., Thompson W.S., Cutler A.J., Dopico X.C., Smyth D.J., et al. IL-21 production by CD4+ effector T cells and frequency of circulating follicular helper T cells are increased in type 1 diabetes patients. Diabetologia. 2015;58(4):781–790. doi: 10.1007/s00125-015-3509-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Feuerer M., Shen Y., Littman D.R., Benoist C., Mathis D. How punctual ablation of regulatory T cells unleashes an autoimmune lesion within the pancreatic islets. Immunity. 2009;31(4):654–664. doi: 10.1016/j.immuni.2009.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Fiorina P., Vergani A., Dada S., Jurewicz M., Wong M., Law K., et al. Targeting CD22 reprograms B-cells and reverses autoimmune diabetes. Diabetes. 2008;57(11):3013–3024. doi: 10.2337/db08-0420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Forster R., Schubel A., Breitfeld D., Kremmer E., Renner-Muller I., Wolf E., et al. CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs. Cell. 1999;99(1):23–33. doi: 10.1016/s0092-8674(00)80059-8. [DOI] [PubMed] [Google Scholar]
- 59.Foulis A.K., Farquharson M.A., Hardman R. Aberrant expression of class II major histocompatibility complex molecules by B cells and hyperexpression of class I major histocompatibility complex molecules by insulin containing islets in type 1 (insulin-dependent) diabetes mellitus. Diabetologia. 1987;30(5):333–343. doi: 10.1007/BF00299027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Friedman R.S., Jacobelli J., Krummel M.F. Surface-bound chemokines capture and prime T cells for synapse formation. Nature Immunology. 2006;7(10):1101–1108. doi: 10.1038/ni1384. [DOI] [PubMed] [Google Scholar]
- 61.Frigerio S., Junt T., Lu B., Gerard C., Zumsteg U., Hollander G.A., et al. Beta cells are responsible for CXCR3-mediated T-cell infiltration in insulitis. Nature Medicine. 2002;8(12):1414–1420. doi: 10.1038/nm1202-792. [DOI] [PubMed] [Google Scholar]
- 62.Gagnerault M.C., Luan J.J., Lotton C., Lepault F. Pancreatic lymph nodes are required for priming of beta cell reactive T cells in NOD mice. Journal of Experimental Medicine. 2002;196(3):369–377. doi: 10.1084/jem.20011353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gago da Graca C., van Baarsen L.G.M., Mebius R.E. Tertiary lymphoid structures: diversity in their development, composition, and role. The Journal of Immunology. 2021;206(2):273–281. doi: 10.4049/jimmunol.2000873. [DOI] [PubMed] [Google Scholar]
- 64.Gao W., Faia K.L., Csizmadia V., Smiley S.T., Soler D., King J.A., et al. Beneficial effects of targeting CCR5 in allograft recipients. Transplantation. 2001;72(7):1199–1205. doi: 10.1097/00007890-200110150-00003. [DOI] [PubMed] [Google Scholar]
- 65.Garred P., Madsen H.O., Petersen J., Marquart H., Hansen T.M., Freiesleben Sorensen S., et al. CC chemokine receptor 5 polymorphism in rheumatoid arthritis. Journal of Rheumatology. 1998;25(8):1462–1465. [PubMed] [Google Scholar]
- 66.Gearty S.V., Dundar F., Zumbo P., Espinosa-Carrasco G., Shakiba M., Sanchez-Rivera F.J., et al. An autoimmune stem-like CD8 T cell population drives type 1 diabetes. Nature. 2022;602(7895):156–161. doi: 10.1038/s41586-021-04248-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Gomez-Reino J.J., Pablos J.L., Carreira P.E., Santiago B., Serrano L., Vicario J.L., et al. Association of rheumatoid arthritis with a functional chemokine receptor, CCR5. Arthritis & Rheumatism. 1999;42(5):989–992. doi: 10.1002/1529-0131(199905)42:5<989::AID-ANR18>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- 68.Goodridge J.P., Lee N., Burian A., Pyo C.W., Tykodi S.S., Warren E.H., et al. HLA-F and MHC-I open conformers cooperate in a MHC-I antigen cross-presentation pathway. The Journal of Immunology. 2013;191(4):1567–1577. doi: 10.4049/jimmunol.1300080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Graham K.L., Krishnamurthy B., Fynch S., Mollah Z.U., Slattery R., Santamaria P., et al. Autoreactive cytotoxic T lymphocytes acquire higher expression of cytotoxic effector markers in the islets of NOD mice after priming in pancreatic lymph nodes. American Journal Of Pathology. 2011;178(6):2716–2725. doi: 10.1016/j.ajpath.2011.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Granados H.M., Draghi A., 2nd, Tsurutani N., Wright K., Fernandez M.L., Sylvester F.A., et al. Programmed cell death-1, PD-1, is dysregulated in T cells from children with new onset type 1 diabetes. PLoS One. 2017;12(9) doi: 10.1371/journal.pone.0183887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Griffith J.W., Sokol C.L., Luster A.D. Chemokines and chemokine receptors: positioning cells for host defense and immunity. Annual Review of Immunology. 2014;32:659–702. doi: 10.1146/annurev-immunol-032713-120145. [DOI] [PubMed] [Google Scholar]
- 72.Grinberg-Bleyer Y., Baeyens A., You S., Elhage R., Fourcade G., Gregoire S., et al. IL-2 reverses established type 1 diabetes in NOD mice by a local effect on pancreatic regulatory T cells. Journal of Experimental Medicine. 2010;207(9):1871–1878. doi: 10.1084/jem.20100209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Groom J.R., Luster A.D. CXCR3 in T cell function. Experimental Cell Research. 2011;317(5):620–631. doi: 10.1016/j.yexcr.2010.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Hanninen A., Nurmela R., Maksimow M., Heino J., Jalkanen S., Kurts C. Islet beta-cell-specific T cells can use different homing mechanisms to infiltrate and destroy pancreatic islets. American Journal Of Pathology. 2007;170(1):240–250. doi: 10.2353/ajpath.2007.060142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Harris D.P., Haynes L., Sayles P.C., Duso D.K., Eaton S.M., Lepak N.M., et al. Reciprocal regulation of polarized cytokine production by effector B and T cells. Nature Immunology. 2000;1(6):475–482. doi: 10.1038/82717. [DOI] [PubMed] [Google Scholar]
- 76.Hartemann A., Bensimon G., Payan C.A., Jacqueminet S., Bourron O., Nicolas N., et al. Low-dose interleukin 2 in patients with type 1 diabetes: a phase 1/2 randomised, double-blind, placebo-controlled trial. Lancet Diabetes & Endocrinology. 2013;1(4):295–305. doi: 10.1016/S2213-8587(13)70113-X. [DOI] [PubMed] [Google Scholar]
- 77.Heninger A.K., Eugster A., Kuehn D., Buettner F., Kuhn M., Lindner A., et al. A divergent population of autoantigen-responsive CD4(+) T cells in infants prior to beta cell autoimmunity. Science Translational Medicine. 2017;9(378) doi: 10.1126/scitranslmed.aaf8848. [DOI] [PubMed] [Google Scholar]
- 78.Henry R.A., Kendall P.L. CXCL13 blockade disrupts B lymphocyte organization in tertiary lymphoid structures without altering B cell receptor bias or preventing diabetes in nonobese diabetic mice. The Journal of Immunology. 2010;185(3):1460–1465. doi: 10.4049/jimmunol.0903710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Herold K.C., Bundy B.N., Long S.A., Bluestone J.A., DiMeglio L.A., Dufort M.J., et al. An anti-CD3 antibody, Teplizumab, in relatives at risk for type 1 diabetes. New England Journal of Medicine. 2019;381(7):603–613. doi: 10.1056/NEJMoa1902226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hoglund P., Mintern J., Waltzinger C., Heath W., Benoist C., Mathis D. Initiation of autoimmune diabetes by developmentally regulated presentation of islet cell antigens in the pancreatic lymph nodes. Journal of Experimental Medicine. 1999;189(2):331–339. doi: 10.1084/jem.189.2.331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hoppu S., Harkonen T., Ronkainen M.S., Simell S., Hekkala A., Toivonen A., et al. IA-2 antibody isotypes and epitope specificity during the prediabetic process in children with HLA-conferred susceptibility to type I diabetes. Clinical and Experimental Immunology. 2006;144(1):59–66. doi: 10.1111/j.1365-2249.2006.03033.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hoppu S., Ronkainen M.S., Kimpimaki T., Simell S., Korhonen S., Ilonen J., et al. Insulin autoantibody isotypes during the prediabetic process in young children with increased genetic risk of type 1 diabetes. Pediatric Research. 2004;55(2):236–242. doi: 10.1203/01.PDR.0000100905.41131.3F. [DOI] [PubMed] [Google Scholar]
- 83.Hu C.Y., Rodriguez-Pinto D., Du W., Ahuja A., Henegariu O., Wong F.S., et al. Treatment with CD20-specific antibody prevents and reverses autoimmune diabetes in mice. Journal of Clinical Investigation. 2007;117(12):3857–3867. doi: 10.1172/JCI32405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Humby F., Bombardieri M., Manzo A., Kelly S., Blades M.C., Kirkham B., et al. Ectopic lymphoid structures support ongoing production of class-switched autoantibodies in rheumatoid synovium. PLoS Medicine. 2009;6(1):e1. doi: 10.1371/journal.pmed.0060001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Iellem A., Mariani M., Lang R., Recalde H., Panina-Bordignon P., Sinigaglia F., et al. Unique chemotactic response profile and specific expression of chemokine receptors CCR4 and CCR8 by CD4(+)CD25(+) regulatory T cells. Journal of Experimental Medicine. 2001;194(6):847–853. doi: 10.1084/jem.194.6.847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.In't Veld P. Insulitis in human type 1 diabetes: a comparison between patients and animal models. Seminars in Immunopathology. 2014;36(5):569–579. doi: 10.1007/s00281-014-0438-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Itoh N., Hanafusa T., Miyazaki A., Miyagawa J., Yamagata K., Yamamoto K., et al. Mononuclear cell infiltration and its relation to the expression of major histocompatibility complex antigens and adhesion molecules in pancreas biopsy specimens from newly diagnosed insulin-dependent diabetes mellitus patients. Journal of Clinical Investigation. 1993;92(5):2313–2322. doi: 10.1172/JCI116835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ivakine E.A., Gulban O.M., Mortin-Toth S.M., Wankiewicz E., Scott C., Spurrell D., et al. Molecular genetic analysis of the Idd4 locus implicates the IFN response in type 1 diabetes susceptibility in nonobese diabetic mice. The Journal of Immunology. 2006;176(5):2976–2990. doi: 10.4049/jimmunol.176.5.2976. [DOI] [PubMed] [Google Scholar]
- 89.Izquierdo C., Ortiz A.Z., Presa M., Malo S., Montoya A., Garabatos N., et al. Treatment of T1D via optimized expansion of antigen-specific Tregs induced by IL-2/anti-IL-2 monoclonal antibody complexes and peptide/MHC tetramers. Scientific Reports. 2018;8(1):8106. doi: 10.1038/s41598-018-26161-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Karl F.M., Winkler C., Ziegler A.G., Laxy M., Achenbach P. Costs of public health screening of children for presymptomatic type 1 diabetes in bavaria, Germany. Diabetes Care. 2022;45(4):837–844. doi: 10.2337/dc21-1648. [DOI] [PubMed] [Google Scholar]
- 91.Kendall P.L., Yu G., Woodward E.J., Thomas J.W. Tertiary lymphoid structures in the pancreas promote selection of B lymphocytes in autoimmune diabetes. The Journal of Immunology. 2007;178(9):5643–5651. doi: 10.4049/jimmunol.178.9.5643. [DOI] [PubMed] [Google Scholar]
- 92.Kenefeck R., Wang C.J., Kapadi T., Wardzinski L., Attridge K., Clough L.E., et al. Follicular helper T cell signature in type 1 diabetes. Journal of Clinical Investigation. 2015;125(1):292–303. doi: 10.1172/JCI76238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kent S.C., Chen Y., Bregoli L., Clemmings S.M., Kenyon N.S., Ricordi C., et al. Expanded T cells from pancreatic lymph nodes of type 1 diabetic subjects recognize an insulin epitope. Nature. 2005;435(7039):224–228. doi: 10.1038/nature03625. [DOI] [PubMed] [Google Scholar]
- 94.Khattri R., Cox T., Yasayko S.A., Ramsdell F. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nature Immunology. 2003;4(4):337–342. doi: 10.1038/ni909. [DOI] [PubMed] [Google Scholar]
- 95.Kim A., Miller K., Jo J., Kilimnik G., Wojcik P., Hara M. Islet architecture: a comparative study. Islets. 2009;1(2):129–136. doi: 10.4161/isl.1.2.9480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kim S.H., Cleary M.M., Fox H.S., Chantry D., Sarvetnick N. CCR4-bearing T cells participate in autoimmune diabetes. Journal of Clinical Investigation. 2002;110(11):1675–1686. doi: 10.1172/JCI15547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Knight R.R., Kronenberg D., Zhao M., Huang G.C., Eichmann M., Bulek A., et al. Human beta-cell killing by autoreactive preproinsulin-specific CD8 T cells is predominantly granule-mediated with the potency dependent upon T-cell receptor avidity. Diabetes. 2013;62(1):205–213. doi: 10.2337/db12-0315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Korpos E., Kadri N., Kappelhoff R., Wegner J., Overall C.M., Weber E., et al. The peri-islet basement membrane, a barrier to infiltrating leukocytes in type 1 diabetes in mouse and human. Diabetes. 2013;62(2):531–542. doi: 10.2337/db12-0432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Korpos E., Kadri N., Loismann S., Findeisen C.R., Arfuso F., Burke G.W., 3rd, et al. Identification and characterisation of tertiary lymphoid organs in human type 1 diabetes. Diabetologia. 2021;64(7):1626–1641. doi: 10.1007/s00125-021-05453-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Kwon B.C., Anand V., Achenbach P., Dunne J.L., Hagopian W., Hu J., et al. Progression of type 1 diabetes from latency to symptomatic disease is predicted by distinct autoimmune trajectories. Nature Communications. 2022;13(1):1514. doi: 10.1038/s41467-022-28909-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lasch S., Muller P., Bayer M., Pfeilschifter J.M., Luster A.D., Hintermann E., et al. Anti-CD3/Anti-CXCL10 antibody combination therapy induces a persistent remission of type 1 diabetes in two mouse models. Diabetes. 2015;64(12):4198–4211. doi: 10.2337/db15-0479. [DOI] [PubMed] [Google Scholar]
- 102.Laufer J.M., Kindinger I., Artinger M., Pauli A., Legler D.F. CCR7 is recruited to the immunological synapse, acts as Co-stimulatory molecule and drives LFA-1 clustering for efficient T cell adhesion through ZAP70. Frontiers in Immunology. 2018;9:3115. doi: 10.3389/fimmu.2018.03115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Le Pottier L., Devauchelle V., Fautrel A., Daridon C., Saraux A., Youinou P., et al. Ectopic germinal centers are rare in Sjogren's syndrome salivary glands and do not exclude autoreactive B cells. The Journal of Immunology. 2009;182(6):3540–3547. doi: 10.4049/jimmunol.0803588. [DOI] [PubMed] [Google Scholar]
- 104.Leete P., Oram R.A., McDonald T.J., Shields B.M., Ziller C., s. team T., et al. Studies of insulin and proinsulin in pancreas and serum support the existence of aetiopathological endotypes of type 1 diabetes associated with age at diagnosis. Diabetologia. 2020;63(6):1258–1267. doi: 10.1007/s00125-020-05115-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Leete P., Willcox A., Krogvold L., Dahl-Jorgensen K., Foulis A.K., Richardson S.J., et al. Differential insulitic profiles determine the extent of beta-cell destruction and the age at onset of type 1 diabetes. Diabetes. 2016;65(5):1362–1369. doi: 10.2337/db15-1615. [DOI] [PubMed] [Google Scholar]
- 106.Li X., Bean A., Uehara M., Banouni N., Ben Nasr M., Kasinath V., et al. Immune heterogeneity of head and tail pancreatic lymph nodes in non-obese diabetic mice. Scientific Reports. 2019;9(1):9778. doi: 10.1038/s41598-019-45899-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lieberman S.M., DiLorenzo T.P. A comprehensive guide to antibody and T-cell responses in type 1 diabetes. Tissue Antigens. 2003;62(5):359–377. doi: 10.1034/j.1399-0039.2003.00152.x. [DOI] [PubMed] [Google Scholar]
- 108.Lieberman S.M., Takaki T., Han B., Santamaria P., Serreze D.V., DiLorenzo T.P. Individual nonobese diabetic mice exhibit unique patterns of CD8+ T cell reactivity to three islet antigens, including the newly identified widely expressed dystrophia myotonica kinase. The Journal of Immunology. 2004;173(11):6727–6734. doi: 10.4049/jimmunol.173.11.6727. [DOI] [PubMed] [Google Scholar]
- 109.Lin A., Yan W.H. The emerging roles of human leukocyte antigen-F in immune modulation and viral infection. Frontiers in Immunology. 2019;10:964. doi: 10.3389/fimmu.2019.00964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lindley S., Dayan C.M., Bishop A., Roep B.O., Peakman M., Tree T.I. Defective suppressor function in CD4(+)CD25(+) T-cells from patients with type 1 diabetes. Diabetes. 2005;54(1):92–99. doi: 10.2337/diabetes.54.1.92. [DOI] [PubMed] [Google Scholar]
- 111.Linsley P.S., Greenbaum C.J., Rosasco M., Presnell S., Herold K.C., Dufort M.J. Elevated T cell levels in peripheral blood predict poor clinical response following rituximab treatment in new-onset type 1 diabetes. Genes and Immunity. 2019;20(4):293–307. doi: 10.1038/s41435-018-0032-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Locci M., Havenar-Daughton C., Landais E., Wu J., Kroenke M.A., Arlehamn C.L., et al. Human circulating PD-1+CXCR3-CXCR5+ memory Tfh cells are highly functional and correlate with broadly neutralizing HIV antibody responses. Immunity. 2013;39(4):758–769. doi: 10.1016/j.immuni.2013.08.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Long S.A., Cerosaletti K., Bollyky P.L., Tatum M., Shilling H., Zhang S., et al. Defects in IL-2R signaling contribute to diminished maintenance of FOXP3 expression in CD4(+)CD25(+) regulatory T-cells of type 1 diabetic subjects. Diabetes. 2010;59(2):407–415. doi: 10.2337/db09-0694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Long S.A., Rieck M., Sanda S., Bollyky J.B., Samuels P.L., Goland R., et al. Rapamycin/IL-2 combination therapy in patients with type 1 diabetes augments Tregs yet transiently impairs beta-cell function. Diabetes. 2012;61(9):2340–2348. doi: 10.2337/db12-0049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ludvigsson J., Heding L., Lieden G., Marner B., Lernmark A. Plasmapheresis in the initial treatment of insulin-dependent diabetes mellitus in children. BMJ. 1983;286(6360):176–178. doi: 10.1136/bmj.286.6360.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Luther S.A., Lopez T., Bai W., Hanahan D., Cyster J.G. BLC expression in pancreatic islets causes B cell recruitment and lymphotoxin-dependent lymphoid neogenesis. Immunity. 2000;12(5):471–481. doi: 10.1016/s1074-7613(00)80199-5. [DOI] [PubMed] [Google Scholar]
- 117.Mahne A.E., Klementowicz J.E., Chou A., Nguyen V., Tang Q. Therapeutic regulatory T cells subvert effector T cell function in inflamed islets to halt autoimmune diabetes. The Journal of Immunology. 2015;194(7):3147–3155. doi: 10.4049/jimmunol.1402739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Marcovecchio M.L., Wicker L.S., Dunger D.B., Dutton S.J., Kopijasz S., Scudder C., et al. Interleukin-2 Therapy of Autoimmunity in Diabetes (ITAD): a phase 2, multicentre, double-blind, randomized, placebo-controlled trial. Wellcome Open Res. 2020;5:49. doi: 10.12688/wellcomeopenres.15697.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Marek-Trzonkowska N., Mysliwiec M., Dobyszuk A., Grabowska M., Derkowska I., Juscinska J., et al. Therapy of type 1 diabetes with CD4(+)CD25(high)CD127-regulatory T cells prolongs survival of pancreatic islets - results of one year follow-up. Clinical Immunology. 2014;153(1):23–30. doi: 10.1016/j.clim.2014.03.016. [DOI] [PubMed] [Google Scholar]
- 120.Marek-Trzonkowska N., Mysliwiec M., Dobyszuk A., Grabowska M., Techmanska I., Juscinska J., et al. Administration of CD4+CD25highCD127- regulatory T cells preserves beta-cell function in type 1 diabetes in children. Diabetes Care. 2012;35(9):1817–1820. doi: 10.2337/dc12-0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Marino E., Tan B., Binge L., Mackay C.R., Grey S.T. B-cell cross-presentation of autologous antigen precipitates diabetes. Diabetes. 2012;61(11):2893–2905. doi: 10.2337/db12-0006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Martin A.P., Grisotto M.G., Canasto-Chibuque C., Kunkel S.L., Bromberg J.S., Furtado G.C., et al. Islet expression of M3 uncovers a key role for chemokines in the development and recruitment of diabetogenic cells in NOD mice. Diabetes. 2008;57(2):387–394. doi: 10.2337/db07-1309. [DOI] [PubMed] [Google Scholar]
- 123.Martin A.P., Marinkovic T., Canasto-Chibuque C., Latif R., Unkeless J.C., Davies T.F., et al. CCR7 deficiency in NOD mice leads to thyroiditis and primary hypothyroidism. The Journal of Immunology. 2009;183(5):3073–3080. doi: 10.4049/jimmunol.0900275. [DOI] [PubMed] [Google Scholar]
- 124.Martin S., Wolf-Eichbaum D., Duinkerken G., Scherbaum W.A., Kolb H., Noordzij J.G., et al. Development of type 1 diabetes despite severe hereditary B-cell deficiency. New England Journal of Medicine. 2001;345(14):1036–1040. doi: 10.1056/NEJMoa010465. [DOI] [PubMed] [Google Scholar]
- 125.Masopust D., Ha S.J., Vezys V., Ahmed R. Stimulation history dictates memory CD8 T cell phenotype: implications for prime-boost vaccination. The Journal of Immunology. 2006;177(2):831–839. doi: 10.4049/jimmunol.177.2.831. [DOI] [PubMed] [Google Scholar]
- 126.Matin K., Salam M.A., Akhter J., Hanada N., Senpuku H. Role of stromal-cell derived factor-1 in the development of autoimmune diseases in non-obese diabetic mice. Immunology. 2002;107(2):222–232. doi: 10.1046/j.1365-2567.2002.01478.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Melli K., Friedman R.S., Martin A.E., Finger E.B., Miao G., Szot G.L., et al. Amplification of autoimmune response through induction of dendritic cell maturation in inflamed tissues. The Journal of Immunology. 2009;182(5):2590–2600. doi: 10.4049/jimmunol.0803543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Mills C.D., Kincaid K., Alt J.M., Heilman M.J., Hill A.M. M-1/M-2 macrophages and the Th1/Th2 paradigm. The Journal of Immunology. 2000;164(12):6166–6173. doi: 10.4049/jimmunol.164.12.6166. [DOI] [PubMed] [Google Scholar]
- 129.Mir-Coll J., Moede T., Paschen M., Neelakandhan A., Valladolid-Acebes I., Leibiger B., et al. Human islet microtissues as an in vitro and an in vivo model system for diabetes. International Journal of Molecular Sciences. 2021;22(4) doi: 10.3390/ijms22041813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Mohan J.F., Kohler R.H., Hill J.A., Weissleder R., Mathis D., Benoist C. Imaging the emergence and natural progression of spontaneous autoimmune diabetes. Proceedings of the National Academy of Sciences of the United States of America. 2017;114(37):E7776–E7785. doi: 10.1073/pnas.1707381114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Molon B., Gri G., Bettella M., Gomez-Mouton C., Lanzavecchia A., Martinez A.C., et al. T cell costimulation by chemokine receptors. Nature Immunology. 2005;6(5):465–471. doi: 10.1038/ni1191. [DOI] [PubMed] [Google Scholar]
- 132.Montane J., Bischoff L., Soukhatcheva G., Dai D.L., Hardenberg G., Levings M.K., et al. Prevention of murine autoimmune diabetes by CCL22-mediated Treg recruitment to the pancreatic islets. Journal of Clinical Investigation. 2011;121(8):3024–3028. doi: 10.1172/JCI43048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Montane J., Obach M., Alvarez S., Bischoff L., Dai D.L., Soukhatcheva G., et al. CCL22 prevents rejection of mouse islet allografts and induces donor-specific tolerance. Cell Transplantation. 2015;24(10):2143–2154. doi: 10.3727/096368914X685249. [DOI] [PubMed] [Google Scholar]
- 134.Morimoto J., Yoneyama H., Shimada A., Shigihara T., Yamada S., Oikawa Y., et al. CXC chemokine ligand 10 neutralization suppresses the occurrence of diabetes in nonobese diabetic mice through enhanced beta cell proliferation without affecting insulitis. The Journal of Immunology. 2004;173(11):7017–7024. doi: 10.4049/jimmunol.173.11.7017. [DOI] [PubMed] [Google Scholar]
- 135.Morita R., Schmitt N., Bentebibel S.E., Ranganathan R., Bourdery L., Zurawski G., et al. Human blood CXCR5(+)CD4(+) T cells are counterparts of T follicular cells and contain specific subsets that differentially support antibody secretion. Immunity. 2011;34(1):108–121. doi: 10.1016/j.immuni.2010.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Nacionales D.C., Weinstein J.S., Yan X.J., Albesiano E., Lee P.Y., Kelly-Scumpia K.M., et al. B cell proliferation, somatic hypermutation, class switch recombination, and autoantibody production in ectopic lymphoid tissue in murine lupus. The Journal of Immunology. 2009;182(7):4226–4236. doi: 10.4049/jimmunol.0800771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Nakayama M., Abiru N., Moriyama H., Babaya N., Liu E., Miao D., et al. Prime role for an insulin epitope in the development of type 1 diabetes in NOD mice. Nature. 2005;435(7039):220–223. doi: 10.1038/nature03523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Negulescu P.A., Krasieva T.B., Khan A., Kerschbaum H.H., Cahalan M.D. Polarity of T cell shape, motility, and sensitivity to antigen. Immunity. 1996;4(5):421–430. doi: 10.1016/s1074-7613(00)80409-4. [DOI] [PubMed] [Google Scholar]
- 139.Noorchashm H., Lieu Y.K., Noorchashm N., Rostami S.Y., Greeley S.A.S., Schlachterman A., et al. I-A(g7)-mediated antigen presentation by B lymphocytes is critical in overcoming a checkpoint in T cell tolerance to islet beta cells of nonobese diabetic mice. Journal of Immunology. 1999;163(2):743–750. [PubMed] [Google Scholar]
- 140.Orban T., Farkas K., Jalahej H., Kis J., Treszl A., Falk B., et al. Autoantigen-specific regulatory T cells induced in patients with type 1 diabetes mellitus by insulin B-chain immunotherapy. Journal of Autoimmunity. 2010;34(4):408–415. doi: 10.1016/j.jaut.2009.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Otonkoski T., Banerjee M., Korsgren O., Thornell L.E., Virtanen I. Unique basement membrane structure of human pancreatic islets: implications for beta-cell growth and differentiation. Diabetes, Obesity and Metabolism. 2008;10(Suppl 4):119–127. doi: 10.1111/j.1463-1326.2008.00955.x. [DOI] [PubMed] [Google Scholar]
- 142.Peiris H., Bonder C.S., Coates P.T., Keating D.J., Jessup C.F. The beta-cell/EC axis: how do islet cells talk to each other? Diabetes. 2014;63(1):3–11. doi: 10.2337/db13-0617. [DOI] [PubMed] [Google Scholar]
- 143.Penko D., Rojas-Canales D., Mohanasundaram D., Peiris H.S., Sun W.Y., Drogemuller C.J., et al. Endothelial progenitor cells enhance islet engraftment, influence beta-cell function, and modulate islet connexin 36 expression. Cell Transplantation. 2015;24(1):37–48. doi: 10.3727/096368913X673423. [DOI] [PubMed] [Google Scholar]
- 144.Pescovitz M.D., Greenbaum C.J., Bundy B., Becker D.J., Gitelman S.E., Goland R., et al. B-lymphocyte depletion with rituximab and beta-cell function: two-year results. Diabetes Care. 2014;37(2):453–459. doi: 10.2337/dc13-0626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Pescovitz M.D., Greenbaum C.J., Krause-Steinrauf H., Becker D.J., Gitelman S.E., Goland R., et al. Rituximab, B-lymphocyte depletion, and preservation of beta-cell function. New England Journal of Medicine. 2009;361(22):2143–2152. doi: 10.1056/NEJMoa0904452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Petersen J.S., Kulmala P., Clausen J.T., Knip M., Dyrberg T. Progression to type 1 diabetes is associated with a change in the immunoglobulin isotype profile of autoantibodies to glutamic acid decarboxylase (GAD65). Childhood Diabetes in Finland Study Group. Clinical Immunology. 1999;90(2):276–281. doi: 10.1006/clim.1998.4641. [DOI] [PubMed] [Google Scholar]
- 147.Phillips J.M., Parish N.M., Raine T., Bland C., Sawyer Y., De La Pena H., et al. Type 1 diabetes development requires both CD4+ and CD8+ T cells and can be reversed by non-depleting antibodies targeting both T cell populations. The Review of Diabetic Studies. 2009;6(2):97–103. doi: 10.1900/RDS.2009.6.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Piseddu I., Rohrle N., Knott M.M.L., Moder S., Eiber S., Schnell K., et al. Constitutive expression of CCL22 is mediated by T cell-derived GM-CSF. The Journal of Immunology. 2020;205(8):2056–2065. doi: 10.4049/jimmunol.2000004. [DOI] [PubMed] [Google Scholar]
- 149.Postigo-Fernandez J., Farber D.L., Creusot R.J. Phenotypic alterations in pancreatic lymph node stromal cells from human donors with type 1 diabetes and NOD mice. Diabetologia. 2019;62(11):2040–2051. doi: 10.1007/s00125-019-04984-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Radenkovic M., Uvebrant K., Skog O., Sarmiento L., Avartsson J., Storm P., et al. Characterization of resident lymphocytes in human pancreatic islets. Clinical and Experimental Immunology. 2017;187(3):418–427. doi: 10.1111/cei.12892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Raffin C., Vo L.T., Bluestone J.A. Treg cell-based therapies: challenges and perspectives. Nature Reviews Immunology. 2020;20(3):158–172. doi: 10.1038/s41577-019-0232-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Rapp M., Wintergerst M.W.M., Kunz W.G., Vetter V.K., Knott M.M.L., Lisowski D., et al. CCL22 controls immunity by promoting regulatory T cell communication with dendritic cells in lymph nodes. Journal of Experimental Medicine. 2019;216(5):1170–1181. doi: 10.1084/jem.20170277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Rhode A., Pauza M.E., Barral A.M., Rodrigo E., Oldstone M.B., von Herrath M.G., et al. Islet-specific expression of CXCL10 causes spontaneous islet infiltration and accelerates diabetes development. The Journal of Immunology. 2005;175(6):3516–3524. doi: 10.4049/jimmunol.175.6.3516. [DOI] [PubMed] [Google Scholar]
- 154.Richardson S.J., Rodriguez-Calvo T., Gerling I.C., Mathews C.E., Kaddis J.S., Russell M.A., et al. Islet cell hyperexpression of HLA class I antigens: a defining feature in type 1 diabetes. Diabetologia. 2016;59(11):2448–2458. doi: 10.1007/s00125-016-4067-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Rodriguez-Calvo T., Johnson J.D., Overbergh L., Dunne J.L. Neoepitopes in type 1 diabetes: etiological insights, biomarkers and therapeutic targets. Frontiers in Immunology. 2021;12 doi: 10.3389/fimmu.2021.667989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Rodriguez-Diaz R., Abdulreda M.H., Formoso A.L., Gans I., Ricordi C., Berggren P.O., et al. Innervation patterns of autonomic axons in the human endocrine pancreas. Cell Metabolism. 2011;14(1):45–54. doi: 10.1016/j.cmet.2011.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Rodriguez-Diaz R., Dando R., Jacques-Silva M.C., Fachado A., Molina J., Abdulreda M.H., et al. Alpha cells secrete acetylcholine as a non-neuronal paracrine signal priming beta cell function in humans. Nature Medicine. 2011;17(7):888–892. doi: 10.1038/nm.2371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Rot A., von Andrian U.H. Chemokines in innate and adaptive host defense: basic chemokinese grammar for immune cells. Annual Review of Immunology. 2004;22:891–928. doi: 10.1146/annurev.immunol.22.012703.104543. [DOI] [PubMed] [Google Scholar]
- 159.Rotondi M., Chiovato L., Romagnani S., Serio M., Romagnani P. Role of chemokines in endocrine autoimmune diseases. Endocrine Reviews. 2007;28(5):492–520. doi: 10.1210/er.2006-0044. [DOI] [PubMed] [Google Scholar]
- 160.Rui J., Deng S., Arazi A., Perdigoto A.L., Liu Z., Herold K.C. Beta cells that resist immunological attack develop during progression of autoimmune diabetes in NOD mice. Cell Metabolism. 2017;25(3):727–738. doi: 10.1016/j.cmet.2017.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Russell M.A., Redick S.D., Blodgett D.M., Richardson S.J., Leete P., Krogvold L., et al. HLA class II antigen processing and presentation pathway components demonstrated by transcriptome and protein analyses of islet beta-cells from donors with type 1 diabetes. Diabetes. 2019;68(5):988–1001. doi: 10.2337/db18-0686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Ryden A.K., Perdue N.R., Pagni P.P., Gibson C.B., Ratliff S.S., Kirk R.K., et al. Anti-IL-21 monoclonal antibody combined with liraglutide effectively reverses established hyperglycemia in mouse models of type 1 diabetes. Journal of Autoimmunity. 2017;84:65–74. doi: 10.1016/j.jaut.2017.07.006. [DOI] [PubMed] [Google Scholar]
- 163.Salomon B., Lenschow D.J., Rhee L., Ashourian N., Singh B., Sharpe A., et al. B7/CD28 costimulation is essential for the homeostasis of the CD4(+)CD25(+) immunoregulatory T cells that control autoimmune diabetes. Immunity. 2000;12(4):431–440. doi: 10.1016/s1074-7613(00)80195-8. [DOI] [PubMed] [Google Scholar]
- 164.Sandor A.M., Jacobelli J., Friedman R.S. Immune cell trafficking to the islets during type 1 diabetes. Clinical and Experimental Immunology. 2019;198(3):314–325. doi: 10.1111/cei.13353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Sandor A.M., Lindsay R.S., Dyjack N., Whitesell J.C., Rios C., Bradley B.J., et al. CD11c(+) cells are gatekeepers for lymphocyte trafficking to infiltrated islets during type 1 diabetes. Frontiers in Immunology. 2019;10:99. doi: 10.3389/fimmu.2019.00099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Sarkar S.A., Lee C.E., Victorino F., Nguyen T.T., Walters J.A., Burrack A., et al. Expression and regulation of chemokines in murine and human type 1 diabetes. Diabetes. 2012;61(2):436–446. doi: 10.2337/db11-0853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Savinov A.Y., Wong F.S., Stonebraker A.C., Chervonsky A.V. Presentation of antigen by endothelial cells and chemoattraction are required for homing of insulin-specific CD8+ T cells. Journal of Experimental Medicine. 2003;197(5):643–656. doi: 10.1084/jem.20021378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Scherm M.G., Serr I., Zahm A.M., Schug J., Bellusci S., Manfredini R., et al. miRNA142-3p targets Tet2 and impairs Treg differentiation and stability in models of type 1 diabetes. Nature Communications. 2019;10(1):5697. doi: 10.1038/s41467-019-13587-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Seidel D.K., Geiss H.C., Donner M.G., Ritter M.M., Schwandt P., Koll R.A., et al. Course of islet autoantibody titers during Ig-immunoadsorption in a patient with newly diagnosed type 1 diabetes. Journal of Autoimmunity. 1998;11(3):273–277. doi: 10.1006/jaut.1998.0195. [DOI] [PubMed] [Google Scholar]
- 170.Serr I., Drost F., Schubert B., Daniel C. Antigen-specific Treg therapy in type 1 diabetes - challenges and opportunities. Frontiers in Immunology. 2021;12 doi: 10.3389/fimmu.2021.712870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Serr I., Furst R.W., Achenbach P., Scherm M.G., Gokmen F., Haupt F., et al. Type 1 diabetes vaccine candidates promote human Foxp3(+)Treg induction in humanized mice. Nature Communications. 2016;7 doi: 10.1038/ncomms10991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Serr I., Furst R.W., Ott V.B., Scherm M.G., Nikolaev A., Gokmen F., et al. miRNA92a targets KLF2 and the phosphatase PTEN signaling to promote human T follicular helper precursors in T1D islet autoimmunity. Proceedings of the National Academy of Sciences of the United States of America. 2016;113(43):E6659–E6668. doi: 10.1073/pnas.1606646113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Serr I., Scherm M.G., Zahm A.M., Schug J., Flynn V.K., Hippich M., et al. A miRNA181a/NFAT5 axis links impaired T cell tolerance induction with autoimmune type 1 diabetes. Science Translational Medicine. 2018;10(422) doi: 10.1126/scitranslmed.aag1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Serreze D.V., Chapman H.D., Varnum D.S., Hanson M.S., Reifsnyder P.C., Richard S.D., et al. B lymphocytes are essential for the initiation of T cell-mediated autoimmune diabetes: analysis of a new "speed congenic" stock of NOD.Ig mu null mice. Journal of Experimental Medicine. 1996;184(5):2049–2053. doi: 10.1084/jem.184.5.2049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Serreze D.V., Fleming S.A., Chapman H.D., Richard S.D., Leiter E.H., Tisch R.M. B lymphocytes are critical antigen-presenting cells for the initiation of T cell-mediated autoimmune diabetes in nonobese diabetic mice. The Journal of Immunology. 1998;161(8):3912–3918. [PubMed] [Google Scholar]
- 176.Sharp C.D., Huang M., Glawe J., Patrick D.R., Pardue S., Barlow S.C., et al. Stromal cell-derived factor-1/CXCL12 stimulates chemorepulsion of NOD/LtJ T-cell adhesion to islet microvascular endothelium. Diabetes. 2008;57(1):102–112. doi: 10.2337/db07-0494. [DOI] [PubMed] [Google Scholar]
- 177.Sharp S.A., Rich S.S., Wood A.R., Jones S.E., Beaumont R.N., Harrison J.W., et al. Development and standardization of an improved type 1 diabetes genetic risk score for use in newborn screening and incident diagnosis. Diabetes Care. 2019;42(2):200–207. doi: 10.2337/dc18-1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Silveira P.A., Johnson E., Chapman H.D., Bui T., Tisch R.M., Serreze D.V. The preferential ability of B lymphocytes to act as diabetogenic APC in NOD mice depends on expression of self-antigen-specific immunoglobulin receptors. European Journal of Immunology. 2002;32(12):3657–3666. doi: 10.1002/1521-4141(200212)32:12<3657::AID-IMMU3657>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 179.Simpson J., Rezaie P., Newcombe J., Cuzner M.L., Male D., Woodroofe M.N. Expression of the beta-chemokine receptors CCR2, CCR3 and CCR5 in multiple sclerosis central nervous system tissue. Journal of Neuroimmunology. 2000;108(1–2):192–200. doi: 10.1016/s0165-5728(00)00274-5. [DOI] [PubMed] [Google Scholar]
- 180.Sims E.K., Bundy B.N., Stier K., Serti E., Lim N., Long S.A., et al. Teplizumab improves and stabilizes beta cell function in antibody-positive high-risk individuals. Science Translational Medicine. 2021;13(583) doi: 10.1126/scitranslmed.abc8980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Slominski B., Lawrynowicz U., Mysliwska J., Ryba-Stanislawowska M., Skrzypkowska M., Mysliwiec M., et al. CCR5-Delta32 gene polymorphism is related to celiac disease and autoimmune thyroiditis coincidence in patients with type 1 diabetes. Journal of Diabetic Complications. 2017;31(3):615–618. doi: 10.1016/j.jdiacomp.2016.10.031. [DOI] [PubMed] [Google Scholar]
- 182.Smith M.J., Packard T.A., O'Neill S.K., Henry Dunand C.J., Huang M., Fitzgerald-Miller L., et al. Loss of anergic B cells in prediabetic and new-onset type 1 diabetic patients. Diabetes. 2015;64(5):1703–1712. doi: 10.2337/db13-1798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Smith X., Schneider H., Kohler K., Liu H., Lu Y., Rudd C.E. The chemokine CXCL12 generates costimulatory signals in T cells to enhance phosphorylation and clustering of the adaptor protein SLP-76. Science Signaling. 2013;6(286):ra65. doi: 10.1126/scisignal.2004018. [DOI] [PubMed] [Google Scholar]
- 184.Solomon M., Balasa B., Sarvetnick N. CCR2 and CCR5 chemokine receptors differentially influence the development of autoimmune diabetes in the NOD mouse. Autoimmunity. 2010;43(2):156–163. doi: 10.3109/08916930903246464. [DOI] [PubMed] [Google Scholar]
- 185.Sorensen T.L., Tani M., Jensen J., Pierce V., Lucchinetti C., Folcik V.A., et al. Expression of specific chemokines and chemokine receptors in the central nervous system of multiple sclerosis patients. Journal of Clinical Investigation. 1999;103(6):807–815. doi: 10.1172/JCI5150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Stamatouli A.M., Quandt Z., Perdigoto A.L., Clark P.L., Kluger H., Weiss S.A., et al. Collateral damage: insulin-dependent diabetes induced with checkpoint inhibitors. Diabetes. 2018;67(8):1471–1480. doi: 10.2337/dbi18-0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Suk K., Kim S., Kim Y.H., Kim K.A., Chang I., Yagita H., et al. IFN-gamma/TNF-alpha synergism as the final effector in autoimmune diabetes: a key role for STAT1/IFN regulatory factor-1 pathway in pancreatic beta cell death. The Journal of Immunology. 2001;166(7):4481–4489. doi: 10.4049/jimmunol.166.7.4481. [DOI] [PubMed] [Google Scholar]
- 188.Sutherland D.E., Goetz F.C., Sibley R.K. Recurrence of disease in pancreas transplants. Diabetes. 1989;38(Suppl 1):85–87. doi: 10.2337/diab.38.1.s85. [DOI] [PubMed] [Google Scholar]
- 189.Tan J., Jin X., Zhao R., Wei X., Liu Y., Kong X. Beneficial effect of T follicular helper cells on antibody class switching of B cells in prostate cancer. Oncology Reports. 2015;33(3):1512–1518. doi: 10.3892/or.2014.3684. [DOI] [PubMed] [Google Scholar]
- 190.Tan T.G., Mathis D., Benoist C. Singular role for T-BET+CXCR3+ regulatory T cells in protection from autoimmune diabetes. Proceedings of the National Academy of Sciences of the United States of America. 2016;113(49):14103–14108. doi: 10.1073/pnas.1616710113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Tang S.C., Baeyens L., Shen C.N., Peng S.J., Chien H.J., Scheel D.W., et al. Human pancreatic neuro-insular network in health and fatty infiltration. Diabetologia. 2018;61(1):168–181. doi: 10.1007/s00125-017-4409-x. [DOI] [PubMed] [Google Scholar]
- 192.Tian J., Zekzer D., Hanssen L., Lu Y., Olcott A., Kaufman D.L. Lipopolysaccharide-activated B cells down-regulate Th1 immunity and prevent autoimmune diabetes in nonobese diabetic mice. The Journal of Immunology. 2001;167(2):1081–1089. doi: 10.4049/jimmunol.167.2.1081. [DOI] [PubMed] [Google Scholar]
- 193.Trivedi P., Graham K.L., Krishnamurthy B., Fynch S., Slattery R.M., Kay T.W., et al. Perforin facilitates beta cell killing and regulates autoreactive CD8+ T-cell responses to antigen in mouse models of type 1 diabetes. Immunology & Cell Biology. 2016;94(4):334–341. doi: 10.1038/icb.2015.89. [DOI] [PubMed] [Google Scholar]
- 194.Tybulewicz V.L. Chemokines and the immunological synapse. Immunology. 2002;106(3):287–288. doi: 10.1046/j.1365-2567.2002.01467.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Uno S., Imagawa A., Okita K., Sayama K., Moriwaki M., Iwahashi H., et al. Macrophages and dendritic cells infiltrating islets with or without beta cells produce tumour necrosis factor-alpha in patients with recent-onset type 1 diabetes. Diabetologia. 2007;50(3):596–601. doi: 10.1007/s00125-006-0569-9. [DOI] [PubMed] [Google Scholar]
- 196.van Berkel V., Barrett J., Tiffany H.L., Fremont D.H., Murphy P.M., McFadden G., et al. Identification of a gammaherpesvirus selective chemokine binding protein that inhibits chemokine action. Journal of Virology. 2000;74(15):6741–6747. doi: 10.1128/jvi.74.15.6741-6747.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Virtanen I., Banerjee M., Palgi J., Korsgren O., Lukinius A., Thornell L.E., et al. Blood vessels of human islets of Langerhans are surrounded by a double basement membrane. Diabetologia. 2008;51(7):1181–1191. doi: 10.1007/s00125-008-0997-9. [DOI] [PubMed] [Google Scholar]
- 198.Vitali C., Mingozzi F., Broggi A., Barresi S., Zolezzi F., Bayry J., et al. Migratory, and not lymphoid-resident, dendritic cells maintain peripheral self-tolerance and prevent autoimmunity via induction of iTreg cells. Blood. 2012;120(6):1237–1245. doi: 10.1182/blood-2011-09-379776. [DOI] [PubMed] [Google Scholar]
- 199.von Herrath M., Bain S.C., Bode B., Clausen J.O., Coppieters K., Gaysina L., et al. Anti-interleukin-21 antibody and liraglutide for the preservation of beta-cell function in adults with recent-onset type 1 diabetes: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Diabetes & Endocrinology. 2021;9(4):212–224. doi: 10.1016/S2213-8587(21)00019-X. [DOI] [PubMed] [Google Scholar]
- 200.Wang Y.J., Golson M.L., Schug J., Traum D., Liu C., Vivek K., et al. Single-cell mass cytometry analysis of the human endocrine pancreas. Cell Metabolism. 2016;24(4):616–626. doi: 10.1016/j.cmet.2016.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Wegmann D.R., Norbury-Glaser M., Daniel D. Insulin-specific T cells are a predominant component of islet infiltrates in pre-diabetic NOD mice. European Journal of Immunology. 1994;24(8):1853–1857. doi: 10.1002/eji.1830240820. [DOI] [PubMed] [Google Scholar]
- 202.Wei X., Tromberg B.J., Cahalan M.D. Mapping the sensitivity of T cells with an optical trap: polarity and minimal number of receptors for Ca(2+) signaling. Proceedings of the National Academy of Sciences of the United States of America. 1999;96(15):8471–8476. doi: 10.1073/pnas.96.15.8471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Weitz J.R., Jacques-Silva C., Qadir M.M.F., Umland O., Pereira E., Qureshi F., et al. Secretory functions of macrophages in the human pancreatic islet are regulated by endogenous purinergic signaling. Diabetes. 2020;69(6):1206–1218. doi: 10.2337/db19-0687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Willcox A., Richardson S.J., Bone A.J., Foulis A.K., Morgan N.G. Analysis of islet inflammation in human type 1 diabetes. Clinical and Experimental Immunology. 2009;155(2):173–181. doi: 10.1111/j.1365-2249.2008.03860.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Willcox A., Richardson S.J., Walker L.S.K., Kent S.C., Morgan N.G., Gillespie K.M. Germinal centre frequency is decreased in pancreatic lymph nodes from individuals with recent-onset type 1 diabetes. Diabetologia. 2017;60(7):1294–1303. doi: 10.1007/s00125-017-4221-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Wong F.S., Karttunen J., Dumont C., Wen L., Visintin I., Pilip I.M., et al. Identification of an MHC class I-restricted autoantigen in type 1 diabetes by screening an organ-specific cDNA library. Nature Medicine. 1999;5(9):1026–1031. doi: 10.1038/12465. [DOI] [PubMed] [Google Scholar]
- 207.Wong F.S., Wen L., Tang M., Ramanathan M., Visintin I., Daugherty J., et al. Investigation of the role of B-cells in type 1 diabetes in the NOD mouse. Diabetes. 2004;53(10):2581–2587. doi: 10.2337/diabetes.53.10.2581. [DOI] [PubMed] [Google Scholar]
- 208.Wu M., Lee M.Y.Y., Bahl V., Traum D., Schug J., Kusmartseva I., et al. Single-cell analysis of the human pancreas in type 2 diabetes using multi-spectral imaging mass cytometry. Cell Reports. 2021;37(5) doi: 10.1016/j.celrep.2021.109919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Wyatt R.C., Lanzoni G., Russell M.A., Gerling I., Richardson S.J. What the HLA-I!-Classical and non-classical HLA class I and their potential roles in type 1 diabetes. Current Diabetes Reports. 2019;19(12):159. doi: 10.1007/s11892-019-1245-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Xhonneux L.P., Knight O., Lernmark A., Bonifacio E., Hagopian W.A., Rewers M.J., et al. Transcriptional networks in at-risk individuals identify signatures of type 1 diabetes progression. Science Translational Medicine. 2021;13(587) doi: 10.1126/scitranslmed.abd5666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Xiao X., Gaffar I., Guo P., Wiersch J., Fischbach S., Peirish L., et al. M2 macrophages promote beta-cell proliferation by up-regulation of SMAD7. Proceedings of the National Academy of Sciences of the United States of America. 2014;111(13):E1211–E1220. doi: 10.1073/pnas.1321347111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Xiu Y., Wong C.P., Bouaziz J.D., Hamaguchi Y., Wang Y., Pop S.M., et al. B lymphocyte depletion by CD20 monoclonal antibody prevents diabetes in nonobese diabetic mice despite isotype-specific differences in Fc gamma R effector functions. The Journal of Immunology. 2008;180(5):2863–2875. doi: 10.4049/jimmunol.180.5.2863. [DOI] [PubMed] [Google Scholar]
- 213.Yamada Y., Okubo Y., Shimada A., Oikawa Y., Yamada S., Narumi S., et al. Acceleration of diabetes development in CXC chemokine receptor 3 (CXCR3)-deficient NOD mice. Diabetologia. 2012;55(8):2238–2245. doi: 10.1007/s00125-012-2547-8. [DOI] [PubMed] [Google Scholar]
- 214.Yano T., Liu Z., Donovan J., Thomas M.K., Habener J.F. Stromal cell derived factor-1 (SDF-1)/CXCL12 attenuates diabetes in mice and promotes pancreatic beta-cell survival by activation of the prosurvival kinase Akt. Diabetes. 2007;56(12):2946–2957. doi: 10.2337/db07-0291. [DOI] [PubMed] [Google Scholar]
- 215.Yu A., Zhu L., Altman N.H., Malek T.R. A low interleukin-2 receptor signaling threshold supports the development and homeostasis of T regulatory cells. Immunity. 2009;30(2):204–217. doi: 10.1016/j.immuni.2008.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Ziegler A.G., Kick K., Bonifacio E., Haupt F., Hippich M., Dunstheimer D., et al. Yield of a public health screening of children for islet autoantibodies in bavaria, Germany. JAMA. 2020;323(4):339–351. doi: 10.1001/jama.2019.21565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Zirpel H., Roep B.O. Islet-resident dendritic cells and macrophages in type 1 diabetes: in search of bigfoot's print. Frontiers in Endocrinology. 2021;12 doi: 10.3389/fendo.2021.666795. [DOI] [PMC free article] [PubMed] [Google Scholar]