Abstract
Background
Arterial arteriosclerosis and atherosclerosis reflect vascular disease, the subclinical detection of which allows opportunity for cardiovascular disease (CVD) prevention. Larger cohort studies simultaneously quantifying anatomic thoracic and abdominal aortic pathologic abnormalities are lacking in the literature.
Purpose
To investigate the association of aortic wall area (AWA) and atherosclerotic plaque presence and burden as measured on MRI scans with incident CVD in a community sample.
Materials and Methods
In this prospective cohort study, participants in the Framingham Heart Study Offspring Cohort without prevalent CVD underwent 1.5-T MRI (between 2002–2005) of the descending thoracic and abdominal aorta with electrocardiogram-gated axial T2-weighted black-blood acquisitions. The wall thickness of the thoracic aorta was measured at the pulmonary bifurcation level and used to calculate the AWA as the difference between cross-sectional vessel area and lumen area. For primary or secondary analyses, multivariable Cox proportional hazards regression models were used to examine the association of aortic MRI measures with risk of first-incident CVD events or stroke and coronary heart disease, respectively.
Results
In 1513 study participants (mean age, 64 years ± 9 [SD]; 842 women [56%]), 223 CVD events occurred during follow-up (median, 13.1 years), of which 97 were major events (myocardial infarction, ischemic stroke, or CVD death). In multivariable analysis, thoracic AWA and prevalent thoracic plaque were associated with incident CVD (hazard ratio [HR], 1.20 per SD unit [95% CI: 1.05, 1.37] [P = .006] and HR, 1.63 [95% CI: 1.12, 2.35] [P = .01], respectively). AWA and prevalent thoracic plaque were associated with increased hazards: 1.32 (95% CI: 1.07, 1.62; P = .01) and 2.20 (95% CI: 1.28, 3.79; P = .005), for stroke and coronary heart disease, respectively.
Conclusion
In middle-aged community-dwelling adults, thoracic aortic wall area (AWA), plaque prevalence, and plaque volumes measured with MRI were independently associated with incident cardiovascular disease, with AWA associated in particular with stroke, and plaque associated with coronary heart disease.
Clinical trial registration no. NCT00041418
© RSNA, 2022
Online supplemental material is available for this article.
See also the editorial by Peshock in this issue.
Summary
In middle-aged community-dwelling adults without prevalent cardiovascular disease (CVD), aortic arteriosclerosis and atherosclerosis at MRI were associated with incident CVD events.
Key Results
■ In this prospective cohort study of 1513 participants without prevalent cardiovascular disease (CVD), aortic wall area (AWA) and prevalent thoracic aortic plaque measured on MRI scans were associated with incident CVD (HR, 1.20 per SD [P = .006] and 1.63 [P = .01], respectively).
■ Abdominal aortic plaque prevalence was associated with incident CVD (HR, 1.38 [P = .02]).
■ Elevated AWA was associated with a one-third increased risk of stroke, and prevalent plaque was associated with a greater than twofold risk of coronary heart disease.
Introduction
Atherosclerotic cardiovascular disease (CVD) is the leading cause of morbidity and mortality in the Western world. Identification and treatment of subclinical CVD at its earlier stages with use of cardiovascular imaging may help avoid adverse clinical outcomes (1).
Clinically overt vascular disease is preceded by a long subclinical phase characterized by widespread arterial wall thickening and plaque formation, two processes that may be pathophysiologically distinct. Aortic wall thickness (WT) and cross-sectional aortic wall area (AWA) are objective measures of the magnitude of the combined intima, media, and adventitia. WT increases with atherosclerotic intimal thickening (2), chronic renal disease–related arteriopathy (3), and hypertension-related medial hypertrophy (4). AWA reflects aortic vessel lumen size and WT, both of which are associated with risk factors, including age, sex, diabetes, and hypertension (5–8). AWA therefore captures not only atherosclerosis but also additional pathophysiologic processes leading to lumen diameter changes and arterial wall remodeling.
Measurements of subclinical vascular damage, including arterial plaque (2), merit consideration in CVD risk assessment (1,9). The relative associations of arterial WT and plaque with CVD outcome in large studies differ by artery location (2,5). Carotid plaque was a stronger predictor of CVD than WT at US in the Framingham Heart Study (FHS) Offspring Cohort (n = 2965) (2), while abdominal aortic WT (compared with abdominal aortic plaque) at MRI had a stronger positive association with outcomes in the Dallas Heart Study (5). The variation in association of WT and plaque, such as greater plaque prevalence in the abdominal compared with thoracic aorta (2,10,11), likely relates to differences in intrinsic regional aortic properties or altered arterial dynamics in these regions (12,13).
High–spatial resolution black-blood MRI is used to assess aortic WT (14) and diameter (7) and quantify aortic plaque (2,10,11), with excellent reproducibility (10,15) and good test-retest repeatability (16). Moreover, compared with US assessment, MRI assessment of carotid artery WT has been shown to be more consistently associated with incident CVD (17). Considering the variability of intrinsic aortic properties and function by location, we hypothesized in this study that (a) thoracic AWA, which captures several pathophysiologic processes in vascular remodeling, would be associated with incident CVD and that (b) the association of plaque with CVD varies by plaque location. Thus, we sought to investigate the associations of AWA and aortic plaque by thoracoabdominal location as measured at MRI with incident CVD events in a community-dwelling middle-aged population.
Materials and Methods
Participants
In this prospective cohort study, participants were recruited from the FHS Offspring Cohort (18). Briefly, starting in 1971, study participants were followed with serial examinations every 4–5 years. Examination 7 (1998–2001) included 3539 individuals. For MRI safety and compliance, participants with prior brain MRI scans (n = 1889) were invited to undergo cardiovascular MRI. After exclusion of participants with MRI contraindications, lack of sinus rhythm, and residency outside of Massachusetts or a contiguous state, 1776 individuals were consecutively scanned. Of those, 200 participants had CVD events before examination and 63 had thoracoabdominal MRI examinations that were insufficient for analysis, leading to 1513 eligible participants (Fig 1). CVD prevalence and incidence were based on the general CVD definition introduced by D’Agostino et al (19). Written informed consent was obtained from all participants, and the study was approved by institutional review boards of Beth Israel Deaconess Medical Center and Boston University Medical Center (ClinicalTrials.gov identifier: NCT00041418). The study was compliant with the Health Insurance Portability and Accountability Act.
Figure 1:
Sample selection flowchart. CVD = cardiovascular disease, Exam = examination.
MRI of the Aortic Valve
Between 2002 and 2005, participants underwent thoracoabdominal aortic imaging on a 1.5-T whole-body MRI system (Gyroscan ACS-NT; Philips Healthcare) using either a five-element cardiac phased-array (thorax) or body receiver coil (abdomen). A total of 36 transverse sections covering the aorta from the arch to the aortoiliac bifurcation were acquired using a free-breathing, electrocardiogram-gated, fat-suppressed, black-blood, two-dimensional, T2-weighted turbo spin-echo sequence (11) with in-plane spatial resolution of 1.03 × 0.64 mm and 5-mm section thickness. Section gaps of 10 or 5 mm, repetition time of 1.4 or 2.1 seconds, and echo time of 40 or 45 msec were applied in the thorax and abdomen, respectively.
Aortic Wall and Atherosclerotic Plaque Analysis
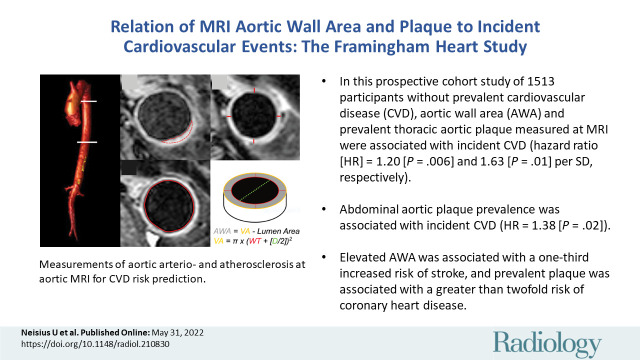
MRI examinations were evaluated using commercial software (MASS, version 6.1; QT-Medis) by one independent reviewer (N.O.M., with >6 years of cardiovascular MRI experience) blinded to clinical data, as previously described (10). Images (a) perpendicular to the descending aorta and (b) in which the inner circumference of the aortic wall could be contoured were considered appropriate for analysis. An interactive freehand manual drawing tool was applied for radial WT assessment and contour tracing (Fig 2). The radial WT of the descending thoracic aorta (level of the pulmonary artery bifurcation) was measured at 12-, 3-, 6-, and 9-o’clock orientations. The cross-sectional aortic lumen area was measured by manual delineation of the blood-to-wall barrier. AWA at this level was calculated as [π × (average WT + lumen diameter/2)2] – [π × (lumen diameter/2)2]. The descending aortic diameter was calculated based on the cross-sectional aortic lumen area at the level of the pulmonary artery bifurcation. In cases of insufficient image quality at the pulmonary artery bifurcation level (n = 24), the consecutive, more distal image was assessed. Atherosclerotic plaque was defined as characteristic luminal protrusions of 1 mm or greater in radial thickness (10,11,15) that could be visually distinguished from the minimal residual blood signal of each plaque. Luminal protrusions were measured based on the continuation of the intimal surface of the adjacent nondiseased vessel wall. Plaque location above or below the diaphragm was classified as thoracic or abdominal, respectively. The cross-sectional area of each plaque was quantified, and the total plaque volume was calculated by multiplying plaque area by (section thickness + gap), with summation of all sections (Fig 2). WT measurements were omitted at wall locations (12, 3, 6, and/or 9 o’clock positions) with plaque and insufficient delineation of the aortic wall (ie, merged with surrounding structures).
Figure 2:
Measurements of aortic arterio- and atherosclerosis at aortic MRI for cardiovascular disease (CVD) risk prediction. (A) Three-dimensional rendered MRI aortic angiogram depicts the aortic anatomy. The aortic wall area (AWA) was measured at the pulmonary artery bifurcation level (upper line), and the diaphragm (lower line) was used to separate the thoracic and abdominal aorta. (B–D) Axial fat-suppressed, black-blood, two-dimensional, T2-weighted MRI images of the thoracic aorta visualize aortic measurements. Aortic plaque was defined as characteristic luminal protrusions of 1 mm or greater in radial thickness (dotted line in B). Wall thickness (WT) (lines in C) and lumen area (circle in D) were used to calculate AWA. (E) Diagram shows AWA equation and cross-sectional depiction of lumen diameter (D) (dotted line) and cross-sectional vessel area (VA) (orange circle).
Participants were categorized in either the less than or equal to 80th percentile or greater than the 80th percentile of AWA among all FHS Offspring Cohort participants, where the sex-specific top quintile was labeled AWA greater than the 80th percentile. The fifth quintile was chosen to have a sufficient number of CVD events in the high-AWA category for Cox proportional hazards regression analysis.
Statistical Analysis
Time to incident of the first CVD event was computed as the difference between the event and MRI examination dates. Follow-up for events or mortality ended December 31, 2019, or December 31, 2018, respectively. Participants who died or were lost to follow-up before experiencing an incident CVD event were censored at the date of death or last FHS contact. A multivariable Cox proportional hazards regression model was used to examine the association of plaque and WT imaging markers, including AWA and AWA indexed for height, with the risk of CVD events. Natural-logarithmic transformation of aortic plaque volume was performed to normalize the distribution before fitting sex-specific analysis of covariance. Sex-specific tobit regression analysis was used to account for left-censoring caused by individuals with zero aortic plaque volume (20). We confirmed the proportional hazards assumptions by visually inspecting Kaplan-Meier plots and assessing the interaction terms of the predictor variables with time in Cox proportional hazards regression models.
Additional description of our statistical analysis is provided in Appendix E1 (online). All statistical analyses were conducted using SAS software (release 9.4, SAS Institute). P < .05 was considered indicative of statistically significant difference.
Results
Participant Characteristics
A study inclusion flow diagram is presented in Figure 1. FHS Offspring Cohort participants (mean age, 64 years ± 9 [SD]; 842 women [56%]) without prevalent CVD at the time of MRI were observed for a median of 13.1 years (range, 0–15.5 years). A total of 223 individuals had incident CVD. Myocardial infarction (n = 52), angina pectoris (n = 30), stroke (n = 31), and transient ischemic attack (n = 28) accounted for 141 of 223 (63%) of first-onset CVD events (Table E1 [online]). Summary demographic characteristics of participants with and without incident CVD are provided in Table 1.
Table 1:
Clinical Characteristics (Examination 7) of Framingham Heart Study Offspring Cohort Participants Who Underwent MRI according to CVD Status

AWA and Atherosclerotic Plaque Prevalence
AWA was greater in study participants with incident CVD compared with those without incident CVD (1.5 cm2 ± 0.4 vs 1.3 cm2 ± 0.3; P < .001). AWA greater than the 80th percentile was twice as prevalent in individuals with incident CVD compared with those without incident CVD (68 of 223 [30%] vs 219 of 1209 [17%]; P < .001) (Table 2). Individuals with incident CVD had a higher prevalence of plaque in the descending thoracic and abdominal aorta (35 of 223 [16%] vs 103 of 1290 [8%] [P < .001] and 116 of 223 [52%] vs 516 of 1290 [40%] [P < .001], respectively).
Table 2:
Aortic MRI Characteristics of Framingham Heart Study Offspring Cohort Participants Who Underwent MRI according to CVD Status

Association of AWA and Aortic Plaque with CVD Risk Factors
The crude associations of AWA and aortic plaque volume with age and CVD risk factors stratified by sex are shown in Tables E2A and E2B (online), respectively. Based on our linear regression analyses, AWA was associated with increased systolic blood pressure (men: β = 0.36 [95% CI: 0.20, 0.52], P < .001; women: β = 0.29 [95% CI: 0.17, 0.41], P < .001), lower total cholesterol in men (β = –0.10 [95% CI: –0.18, –0.02]; P = .01) and lower high-density lipoprotein cholesterol (β = –0.18 [95% CI: –0.32, –0.04]; P = .007) and diabetes (β = 15.6 [95% CI: 6.20, 25.18]; P = .001) in women. Based on our tobit regression analyses, natural-logarithmic–transformed aortic plaque volume was associated with low total (β = –0.26 [95% CI: –0.40, –0.12]; P < .001) and high-density lipoprotein cholesterol (β = –0.64 [95% CI: –1.03, –0.25]; P = .002) in men and diabetes (β = 17.49 [95% CI: 3.18, 31.80]; P = .02) in women.
Association of AWA and Aortic Plaque with Incident CVD
Figure 3 depicts the association of MRI aortic wall and plaque measures with incident CVD events assessed using multivariable Cox proportional hazards regression models. Continuous measures of thoracic AWA were associated with a 20% higher hazard of incident CVD per SD increase in AWA after multivariable adjustment (HR, 1.20 [95% CI: 1.05, 1.37]; P = .006). Linear component measures of AWA were associated with incident CVD and attenuated after multivariable adjustment (average aortic wall radius, per SD: HR, 1.15 [95% CI: 1.01, 1.31] [P = .04]; descending aortic diameter, per SD: HR, 1.14 [95% CI: 0.99, 1.31] [P = .08]). Indexing for height resulted in similar hazards for CVD (AWA/height, per SD; multivariable HR, 1.19 [95% CI: 1.04, 1.36]; P = .01) (Table E3 [online]).
Figure 3:
Risk of elevated aortic wall measures and plaque for incident cardiovascular disease (CVD). Forest plot shows the hazard ratios (HRs) of CVD events for each unit increase in aortic wall and plaque measures after age/sex or multivariable adjustments (age, sex, hypertension, hypercholesterolemia, diabetes mellitus, and smoking status). Continuous variables were calculated per 1 SD unit. CVD occurred in 223 of 1513 Framingham Heart Study Offspring Cohort participants free of disease at baseline (incidence rate, 12.4 [95% CI: 10.8, 14.1]). Ln = natural-logarithmic–transformed.
After multivariable adjustment, prevalent thoracic aortic plaque (HR, 1.63 [95% CI: 1.12, 2.35]; P = .01), prevalent abdominal aortic plaque (HR, 1.38 [95% CI: 1.05, 1.80]; P = .02), and prevalent total aortic plaque (HR, 1.40 [95% CI: 1.07, 1.84]; P = .02) were associated with incident CVD. Continuous plaque volume in the thoracic (HR, 1.17 [95% CI: 1.04, 1.31]; P = .009), abdominal (HR, 1.17 [95% CI: 1.04, 1.31]; P = .008), and total aorta (HR, 1.19 [95% CI: 1.06, 1.34]; P = .004) were also associated with incident CVD after multivariable adjustment (Fig 3, Table E3 [online]).
Both thoracic AWA and prevalent plaque remained associated with incident CVD when analyzed simultaneously in multivariable HR models (HR [AWA, per cm2], 1.65 [95% CI: 1.13, 2.40] [P = .009]; HR [prevalent thoracic plaque], 1.61 [95% CI: 1.11, 2.33] [P = .01]). Assessment of effect modification between AWA and thoracic plaque prevalence revealed that there was no statistically significant interaction between AWA and thoracic plaque prevalence (P = .74 for AWA by prevalent thoracic plaque interaction).
Secondary Outcome Analyses
Kaplan-Meier plot analysis showed that individuals with both AWA greater than the 80th percentile and prevalent thoracic plaque had lower CVD event–free survival compared with either marker alone or neither marker (log-rank P < .001) (Fig 4).
Figure 4:
Kaplan-Meier plots for event-free survival. Kaplan-Meier curves show survival free of cardiovascular disease for participants with and without (A) aortic wall area (AWA) greater than the 80th percentile, (B) prevalence of thoracic plaque, and (C) two (AWA >80th percentile and thoracic plaque), one (AWA >80th percentile or thoracic plaque), or no composite variables. P values reflect log-rank tests. perc. = percentile.
Tables E4A and E4B (online) present multivariable Cox proportional hazards regression models for incident CVD without heart failure and angina, respectively. In multivariable models of CVD not including heart failure, the association with AWA remained unchanged (HR, 1.20 per SD [95% CI: 1.03, 1.39]; P = .02). In multivariable models in which the definition of CVD excluded heart failure, prevalent thoracic plaque was associated with a greater than twofold risk (HR, 2.09 [95% CI: 1.40, 3.13]; P < .001). After exclusion of angina from the outcome definition, AWA and prevalent thoracic plaque were associated, with hazard ratios of 1.16 and 1.44 after multivariable adjustment (HR [AWA, per SD], 1.16 [95% CI: 1.01, 1.34] [P = .04]; HR [prevalent thoracic plaque], 1.44 [95% CI: 0.96, 2.16] [P = .08]).
Table 3 presents multivariable Cox proportional hazards regression estimates of AWA or total plaque prevalence and CVD subgroups, including coronary heart disease, stroke, and heart failure. Prevalent thoracic and abdominal aortic plaque were associated with approximately twofold hazards of incident coronary heart disease risk (multivariable HR, 2.20 [95% CI: 1.28, 3.79] [P = .007] and 1.95 [95% CI: 1.27, 3.01] [P = .003], respectively), whereas AWA and AWA greater than the 80th percentile were associated with an approximate one-third and two-thirds increase in the hazard for incident stroke (multivariable HR [AWA, per SD], 1.32 [95% CI: 1.07, 1.62] [P = .01] and HR [AWA >80th percentile], 1.64 [95% CI: 1.02, 2.63] [P = .04]).
Table 3:
Multivariable Cox Proportional Hazards Model of AWA, Aortic Diameter, and Aortic Plaque Prevalence for Risk of CHD, Stroke, and Heart Failure
Reproducibility of AWA Measurements
Intra- (intraclass correlation coefficient, 0.95 [95% CI: 0.90, 0.98]) and interreader agreement (intraclass correlation coefficient, 0.87 [95% CI: 0.71, 0.94]) for AWA were excellent. Bland-Altman plots showed mean intra- and interreader differences of 0.04 cm2 (range, 0.0–0.5 cm2) and 0.08 cm2 (range, 0.0–0.3 cm2), respectively (Fig 5).
Figure 5:
Reproducibility of aortic wall area (AWA) measurements. (A) Line graphs show intra- and interobserver correlation. Solid blue lines indicate the linear regression line, and dotted red lines show the 95% CI. (B) Bland-Altman plots show AWA measurements. Solid blue lines indicate the bias, and dotted red lines represent the limits of agreement (1.96 × SD).
Discussion
Cardiovascular imaging plays an important role in cardiovascular disease (CVD) risk assessment (1). In this MRI study of 1513 community-dwelling adults who were free of prevalent CVD at baseline and observed for 13.1 years, each SD increase in thoracic aortic wall area (AWA) and prevalent plaque was associated with a respective 20% and 63% higher risk of incident CVD events, even after adjustment for cardiovascular risk factors. Prevalent abdominal plaque and continuous plaque measures were also associated with incident CVD (multivariable hazard ratios: 1.17–1.40, P = .004 to P = .02). Each SD increase in AWA was associated with a 32% greater risk of incident stroke, while prevalent thoracic plaque was associated with greater than twofold hazard of coronary heart disease.
The differential associations of AWA and aortic plaque with CVD likely relate to distinct underlying pathophysiologic characteristics. AWA, a measure of aortic WT and aortic caliber, incorporates assessment of arteriosclerotic remodeling. Similar to linear measures of aortic WT (5,21) and vessel diameter (6), AWA was more closely related to blood pressure than aortic plaque volume was in our study. Such hemodynamic perturbations conferred by higher AWA may thus result in the observed association with stroke.
As demonstrated by transesophageal echocardiography (22) and MRI (10), the prevalence and severity of atherosclerotic plaque increase from the ascending to abdominal aorta. Similarly, aortic calcification at CT in the FHS Offspring Cohort was more prevalent in the abdominal aorta (23). Our results demonstrate that thoracic, more than abdominal, aortic plaque is a stronger predictor of CVD, highlighting the clinical significance of plaque location. Additionally, we observed that continuous quantification of thoracic or abdominal aortic plaque was associated with CVD events, contrary to observations in the Dallas Heart Study (5), which may be due to its shorter follow-up duration.
The evolution of aortic atherosclerosis has a long subclinical phase (24). Thus, earlier diagnosis and treatment of atherosclerosis in the preclinical stage may reduce occurrence of overt CVD. We observed a strong association of AWA with hypertension and stroke, implying that elevated AWA may warrant more aggressive management of blood pressure.
The high reproducibility of AWA and aortic plaque measures (10,11) suggests that they may be readily evaluated at routine cardiothoracic MRI ordered for a variety of clinical indications. The increasing use of machine learning applications in radiology image analysis, including automated segmentation similar to aortic diameter assessment established by Hepp et al (25), could facilitate measurements in routine clinical practice.
Limitations of our study merit consideration. First, we obtained higher-density imaging of the abdominal compared with the thoracic aorta due to greater plaque burden in the former. We attained excellent reproducibility of aortic measures, and the associations we detected at the achieved spatial resolution suggest the validity of the observations. However, future studies with higher spatial resolution may help confirm our findings. Second, due to the FHS participants’ single scan for these sequences, we were unable to evaluate aortic MRI interscan variability. Third, the selection of the 80th percentile of the AWA for Cox proportional hazards regression and Kaplan-Meier analyses was based on our cohort and thus may not be generalizable to other populations. Finally, the majority of individuals the FHS Offspring Cohort are of Western European descent.
In conclusion, in community-dwelling adults with a low to modest burden of cardiovascular disease (CVD) risk factors, thoracic aortic wall area (AWA) and aortic atherosclerotic plaque were predictive of incident CVD, with predominant associations of AWA with stroke and of plaque with coronary heart disease. Our study underscores the importance of subclinical pathologic thoracic aortic remodeling and supports further investigation of the impact of risk factor modification and treatment in adults with elevated thoracic AWA and aortic plaque on modulation of CVD risk. Our results support further studies to assess the impact of aggressive risk factor treatment on the presence and extent of abnormal thoracic AWA and plaque and their impact on primary prevention of CVD, including coronary heart disease and stroke.
Current address: Department of Radiology, Saitama Medical Center, Saitama Medical University, Saitama, Japan.
The Framingham Heart Study is supported by the National Heart, Lung, and Blood Institute (NHLBI) (NO1-HC-25195 and HHSN268201500001I). This work was also supported by NHLBI grants R01-HL70279 (W.J.M.), K23 HL118259 (C.W.T.), and R03 HL145195 (C.W.T.).
Data sharing: Data generated or analyzed during the study are available from the corresponding author by request.
Disclosures of conflicts of interest: U.N. No relevant relationships. P.N.G. No relevant relationships. N.O.M. Japan Society for the Promotion of Science KAKENHI grant (20K08042); consulting fees from Canon Medical Systems; honoraria from Canon Medical Systems, Daiichi-Sankyo, GE Healthcare, Philips Japan, Bayer, and Eisai. M.L.C. No relevant relationships. C.J.O. Employee of Novartis. W.J.M. No relevant relationships. C.W.T. Consulting fees from Calyx Medical Imaging and Abiomed.
Abbreviations:
- AWA
- aortic wall area
- CVD
- cardiovascular disease
- FHS
- Framingham Heart Study
- WT
- wall thickness
References
- 1. Arnett DK , Blumenthal RS , Albert MA , et al . 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines . Circulation 2019. ; 140 ( 11 ): e596 – e646 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Polak JF , Pencina MJ , Pencina KM , O’Donnell CJ , Wolf PA , D’Agostino RB Sr . Carotid-wall intima-media thickness and cardiovascular events . N Engl J Med 2011. ; 365 ( 3 ): 213 – 221 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Litwin M , Wühl E , Jourdan C , et al . Altered morphologic properties of large arteries in children with chronic renal failure and after renal transplantation . J Am Soc Nephrol 2005. ; 16 ( 5 ): 1494 – 1500 . [DOI] [PubMed] [Google Scholar]
- 4. Laurent S , Boutouyrie P . The structural factor of hypertension: large and small artery alterations . Circ Res 2015. ; 116 ( 6 ): 1007 – 1021 . [DOI] [PubMed] [Google Scholar]
- 5. Maroules CD , Rosero E , Ayers C , Peshock RM , Khera A . Abdominal aortic atherosclerosis at MR imaging is associated with cardiovascular events: the Dallas Heart Study . Radiology 2013. ; 269 ( 1 ): 84 – 91 . [DOI] [PubMed] [Google Scholar]
- 6. Rogers IS , Massaro JM , Truong QA , et al . Distribution, determinants, and normal reference values of thoracic and abdominal aortic diameters by computed tomography (from the Framingham Heart Study) . Am J Cardiol 2013. ; 111 ( 10 ): 1510 – 1516 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Turkbey EB , Jain A , Johnson C , et al . Determinants and normal values of ascending aortic diameter by age, gender, and race/ethnicity in the Multi-Ethnic Study of Atherosclerosis (MESA) . J Magn Reson Imaging 2014. ; 39 ( 2 ): 360 – 368 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Qazi S , Massaro JM , Chuang ML , D’Agostino RB Sr , Hoffmann U , O’Donnell CJ . Increased aortic diameters on multidetector computed tomographic scan are independent predictors of incident adverse cardiovascular events: the Framingham Heart Study . Circ Cardiovasc Imaging 2017. ; 10 ( 12 ): e006776 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Whelton PK , Carey RM , Aronow WS , et al . 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines . J Am Coll Cardiol 2018. ; 71 ( 19 ): e127 – e248 . [Published correction appears in J Am Coll Cardiol 2018;71(19):2275-2279.] [DOI] [PubMed] [Google Scholar]
- 10. Oyama N , Gona P , Salton CJ , et al . Differential impact of age, sex, and hypertension on aortic atherosclerosis: the Framingham Heart Study . Arterioscler Thromb Vasc Biol 2008. ; 28 ( 1 ): 155 – 159 . [DOI] [PubMed] [Google Scholar]
- 11. Jaffer FA , O’Donnell CJ , Larson MG , et al . Age and sex distribution of subclinical aortic atherosclerosis: a magnetic resonance imaging examination of the Framingham Heart Study . Arterioscler Thromb Vasc Biol 2002. ; 22 ( 5 ): 849 – 854 . [DOI] [PubMed] [Google Scholar]
- 12. Voges I , Jerosch-Herold M , Hedderich J , et al . Normal values of aortic dimensions, distensibility, and pulse wave velocity in children and young adults: a cross-sectional study . J Cardiovasc Magn Reson 2012. ; 14 ( 1 ): 77 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Engelen L , Bossuyt J , Ferreira I , et al . Reference values for local arterial stiffness . Part A: carotid artery. J Hypertens 2015. ; 33 ( 10 ): 1981 – 1996 . [DOI] [PubMed] [Google Scholar]
- 14. Liu CY , Chen D , Bluemke DA , et al . Evolution of aortic wall thickness and stiffness with atherosclerosis: long-term follow up from the multi-ethnic study of atherosclerosis . Hypertension 2015. ; 65 ( 5 ): 1015 – 1019 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Chan SK , Jaffer FA , Botnar RM , et al . Scan reproducibility of magnetic resonance imaging assessment of aortic atherosclerosis burden . J Cardiovasc Magn Reson 2001. ; 3 ( 4 ): 331 – 338 . [DOI] [PubMed] [Google Scholar]
- 16. Maroules CD , McColl R , Khera A , Peshock RM . Assessment and reproducibility of aortic atherosclerosis magnetic resonance imaging: impact of 3-tesla field strength and parallel imaging . Invest Radiol 2008. ; 43 ( 9 ): 656 – 662 . [DOI] [PubMed] [Google Scholar]
- 17. Zhang Y , Guallar E , Malhotra S , et al . Carotid artery wall thickness and incident cardiovascular events: a comparison between US and MRI in the Multi-Ethnic Study of Atherosclerosis (MESA) . Radiology 2018. ; 289 ( 3 ): 649 – 657 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Feinleib M , Kannel WB , Garrison RJ , McNamara PM , Castelli WP . The Framingham Offspring Study. Design and preliminary data . Prev Med 1975. ; 4 ( 4 ): 518 – 525 . [DOI] [PubMed] [Google Scholar]
- 19. D’Agostino RB Sr , Vasan RS , Pencina MJ , et al . General cardiovascular risk profile for use in primary care: the Framingham Heart Study . Circulation 2008. ; 117 ( 6 ): 743 – 753 . [DOI] [PubMed] [Google Scholar]
- 20. Tobin J . Estimation of relationships for limited dependent variables . Econometrica 1958. ; 26 ( 1 ): 24 – 36 . [Google Scholar]
- 21. Malayeri AA , Natori S , Bahrami H , et al . Relation of aortic wall thickness and distensibility to cardiovascular risk factors (from the Multi-Ethnic Study of Atherosclerosis [MESA]) . Am J Cardiol 2009. ; 102 ( 4 ): 491 – 496 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Goldstein SA , Evangelista A , Abbara S , et al . Multimodality imaging of diseases of the thoracic aorta in adults: from the American Society of Echocardiography and the European Association of Cardiovascular Imaging: endorsed by the Society of Cardiovascular Computed Tomography and Society for Cardiovascular Magnetic Resonance . J Am Soc Echocardiogr 2015. ; 28 ( 2 ): 119 – 182 . [DOI] [PubMed] [Google Scholar]
- 23. Hoffmann U , Massaro JM , D’Agostino RB Sr , Kathiresan S , Fox CS , O’Donnell CJ . Cardiovascular event prediction and risk reclassification by coronary, aortic, and valvular calcification in the Framingham Heart Study . J Am Heart Assoc 2016. ; 5 ( 2 ): e003144 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Holman RL , McGill HC Jr , Strong JP , Geer JC . The natural history of atherosclerosis: the early aortic lesions as seen in New Orleans in the middle of the of the 20th century . Am J Pathol 1958. ; 34 ( 2 ): 209 – 235 . [PMC free article] [PubMed] [Google Scholar]
- 25. Hepp T , Fischer M , Winkelmann MT , et al . Fully automated segmentation and shape analysis of the thoracic aorta in non-contrast-enhanced magnetic resonance images of the German National Cohort Study . J Thorac Imaging 2020. ; 35 ( 6 ): 389 – 398 . [DOI] [PubMed] [Google Scholar]





![Risk of elevated aortic wall measures and plaque for incident cardiovascular disease (CVD). Forest plot shows the hazard ratios (HRs) of CVD events for each unit increase in aortic wall and plaque measures after age/sex or multivariable adjustments (age, sex, hypertension, hypercholesterolemia, diabetes mellitus, and smoking status). Continuous variables were calculated per 1 SD unit. CVD occurred in 223 of 1513 Framingham Heart Study Offspring Cohort participants free of disease at baseline (incidence rate, 12.4 [95% CI: 10.8, 14.1]). Ln = natural-logarithmic–transformed.](https://cdn.ncbi.nlm.nih.gov/pmc/blobs/26a0/9434818/145397726e88/radiol.210830.fig3.jpg)


