Abstract
Background
Adults with Down syndrome are at an ultra-high risk of developing early-onset Alzheimer’s disease. Episodic memory deficits are one of the earliest signs of the disease, but their association with regional brain atrophy in the population with Down syndrome has not been explored. We aimed to investigate the neuroanatomical correlates of episodic memory in adults with Down syndrome and symptomatic Alzheimer’s disease.
Methods
Single-center, cross-sectional study. A total of 139 adults with Down syndrome (85 asymptomatic and 54 with symptomatic Alzheimer’s disease) were included in the study (mean age 43.6 ± 10.9 years, 46% female). Episodic memory was assessed using the modified Cued Recall Test. Immediate (trial 1 free immediate recall, trial 3 free immediate recall, total free immediate recall score, and total immediate score) and delayed scores (free delayed recall score and total delayed score) were examined. Cortical thickness from magnetic resonance imaging was determined with surface-based morphometry using the FreeSurfer 6.0 software package. The clusters of reduced cortical thickness were compared between symptomatic and asymptomatic participants to create a cortical atrophy map. Then, the correlation between cortical thickness and the modified Cued Recall Test subscores were separately assessed in symptomatic and asymptomatic subjects, controlling for age, sex, and severity of intellectual disability.
Results
Compared with asymptomatic participants, those with symptomatic Alzheimer’s disease showed a pattern of cortical atrophy in posterior parieto-temporo-occipital cortices. In symptomatic subjects, trial 1 immediate free recall significantly correlated with cortical atrophy in lateral prefrontal regions. Trial 3 free immediate recall and total free immediate recall were associated with the most widespread cortical atrophy. Total immediate score was related to posterior cortical atrophy, including lateral parietal and temporal cortex, posterior cingulate cortex, precuneus, and medial temporal lobe areas. Delayed memory scores were associated with cortical atrophy in temporoparietal and medial temporal lobe regions. No significant relationships were observed between episodic memory measures and cortical atrophy in asymptomatic subjects.
Conclusions
Different episodic memory measures were associated with cortical atrophy in specific brain regions in adults with Down syndrome and Alzheimer’s disease. These results overlap with those described in sporadic Alzheimer’s disease and further support the similarities between Down syndrome-associated Alzheimer’s disease and that in the general population.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13195-022-01064-x.
Keywords: Down syndrome, Alzheimer’s disease, Dementia, Episodic memory, MRI, Cortical thickness
Background
Down syndrome (DS) is the most frequent genetic form of intellectual disability [1] and is associated to an increased risk of developing early Alzheimer’s disease (AD) dementia due to the presence of an extra copy of the amyloid β precursor protein gene, which is coded on chromosome 21, and is thus overexpressed in people with DS [2].
Episodic memory refers to the ability to learn, store, and retrieve information about unique personal experiences that occur in daily life [3]. Episodic memory impairment is the hallmark symptom of typical Alzheimer’s disease (AD) in the general population and has been associated with early atrophy of medial temporal lobe structures [4]. As the disease progresses, AD pathology spreads to other cortical regions, including ventral and lateral temporal cortex, followed by parietal and frontal cortices [4, 5], affecting other cognitive domains. In DS, neurodegenerative changes due to AD follow a similar pattern as those described in sporadic AD, although it occurs at a younger age [6–8]. Early longitudinal studies suggested executive dysfunction and behavioral and psychological symptoms of dementia as commonly observed symptoms during the early stages of AD in DS [9], although the identification of imaging and fluid biomarkers in the past 5 years has provided new opportunities to characterize the sequence of cognitive decline during the preclinical and prodromal stages of the disease in this population [10]. Recent research now indicates that individuals with DS have a similar clinical presentation to that of people with sporadic or autosomal dominant AD, more specifically, declines in episodic memory and attention measures early in the course of the disease [11, 12].
Although medial temporal lobe regions play a central role in episodic memory, other cortical regions are also necessary to accurately remember new information. More specifically, three different processes or stages have been described in episodic memory that are sustained by different brain networks: encoding, retrieval, and storage [13]. In the general population, these memory processes have been assessed with the Free and Cued Selective Reminding Test (FCSRT) through different subscores and have been associated with different patterns of brain atrophy in patients with mild AD. Specifically, encoding has been associated with atrophy in parietal and temporal regions, storage with atrophy in entorhinal and parahippocampal regions, and retrieval with atrophy in a widespread network encompassing frontal regions [13]. Similarly, Wolk et al. investigated the relationship between performance on different stages of the Rey Auditory Verbal Learning Test (RAVLT), a verbal episodic memory test, and structural brain measures in patients with mild AD from the general population, including immediate and delayed memory recall [14]. This study also found dissociable relationships between regional brain atrophy and different aspects of memory function, indicating that different cognitive processes support different stages of episodic memory performance.
In DS, episodic memory has been evaluated using adapted tests such as the modified Cued Recall Test (mCRT), which has demonstrated to be useful to detect memory decline due to early-stage AD [15] and, more recently, for the diagnosis of prodromal AD [16]. The mCRT has emerged as a promising cognitive indicator of transition to the preclinical and prodromal stage of AD in subjects with DS [11]. Furthermore, poor performance on this episodic memory test has recently been related to white matter degeneration prior to the onset of dementia in adults with DS [17]. The association between regional cortical atrophy and different stages of episodic memory performance in patients with DS and symptomatic AD has not yet been studied.
The aim of this study was to investigate the neural correlates of different episodic memory processes measured with the mCRT in a large cohort of adults with DS, with and without symptomatic AD. Understanding the anatomic basis of the processes underlying episodic memory has potential practical implications, as it may facilitate the integration of clinical and radiological information, helping increase the diagnostic accuracy of AD in this population.
Methods
Study design and participants
In this single-center cross-sectional study, adults with DS aged 18 years or older were recruited from a population-based health plan designed to screen for AD dementia. This health plan includes an annual neurological and neuropsychological assessment, undertaken at the Alzheimer-Down Unit from the Catalan Down Syndrome Foundation and Hospital of Sant Pau, in Barcelona, Spain. Individuals who are also interested in participating in research studies are included in the Down Alzheimer Barcelona Neuroimaging Initiative (DABNI) cohort. The aim of the DABNI project is to improve our understanding of the natural history and mechanisms that drive AD in DS. It includes multimodal biomarker assessments, comprising magnetic resonance imaging (MRI), positron emission tomography, lumbar puncture, sleep studies, and blood and genetic analyses.
We included adults with DS (mild or moderate intellectual disability) with available MRI and a complete neuropsychological assessment. Patients showing any psychiatric or medical disorder that could affect cognition and/or functionality were excluded. We also excluded those patients not able to complete the mCRT due to severe cognitive decline or due to severe or profound intellectual disability [16].
The study was approved by the Sant Pau Ethics Committee following the standards for medical research in humans recommended by the Declaration of Helsinki and reported to the Ministry of Justice, according to the Spanish law for research in people with intellectual disabilities. All participants or their legally authorized representative gave written informed consent before enrollment.
Neuropsychological assessment
Caregivers completed a semi-structured adapted health questionnaire, the Cambridge Examination for Mental Disorders of Older People with Down syndrome and others with intellectual disabilities (CAMDEX-DS), aimed to evaluate cognitive decline in people with intellectual disability [18].
The neuropsychological test battery included the Cambridge Cognitive Examination for Older Adults with Down’s syndrome (CAMCOG-DS), a neuropsychological test battery included in the CAMDEX-DS, comprising subscales for the assessment of the following cognitive domains: orientation, language, memory, attention, praxis, abstract thinking, and perception. For the assessment of episodic memory, the mCRT was used (see below).
Level of intellectual disability
The level of intellectual disability was categorized according to the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition [19] as mild, moderate, severe, or profound intellectual disability, based on caregiver’s reports of the individuals’ best-ever level of functioning.
Assessment of episodic memory
The mCRT is an adapted test to assess episodic memory in people with intellectual disability [15]. This test was selected because it is based on semantic cueing, which allows controlled encoding processes and facilitates retrieval from stored information. The mCRT was administered according to the procedure described by Devenny et al. [15]. Briefly, participants are asked to memorize 12 stimuli (black-and-white line drawings of objects representing a distinct semantic category) presented on three, 4-item cards. The test consists of three trials of free and cued recall performed immediately after the learning phase to compute a total free immediate recall score (FIRS) and a total immediate score (FIRS + cued recall; TIS). The maximum score for both measures is 36. The same recall procedure (free and cued) is done after a 20-min delay to compute a free delayed recall score (FDRS) and a total delayed score (FDRS + cued recall; TDS). The maximum score for both delayed scores is 12. In the general population, some authors found that early immediate learning trials rely on different brain regions than later immediate learning trials [14]; therefore, we also analyzed the data for trial 1 free immediate recall (T1) and trial 3 free immediate recall (T3), representing early and late immediate learning, respectively.
Diagnostic categories
As in previous studies [6, 20], participants with DS were classified by the neurologists and neuropsychologists who assessed them in a consensus meeting into the following groups: asymptomatic, in those with no clinical or neuropsychological suspicion of AD; prodromal AD, in those who had cognitive decline, but symptoms did not fulfill criteria for dementia; or AD dementia, in those who had cognitive decline that significantly interfered in subject’s activities of daily living. In this study, participants were divided into symptomatic (including prodromal AD and AD dementia) and asymptomatic groups.
Structural MRI
All participants underwent MRI in a 3-T Philips Achieva (Philips Healthcare) or a 3-T Siemens Prisma (Siemens Healthcare) MR scanner (protocols detailed in the supplements). Cortical thickness was assessed with surface-based analysis in FreeSurfer 6.0 (https://surfer.nmr.mgh.harvard.edu/) using an automated standard pipeline detailed elsewhere [21–23]. Briefly, this pipeline normalizes T1-weighted images to a standard space (MNI305), removes non-brain tissue, classifies each brain voxel as gray of white matter, and defines interfaces between gray and white matter (GM and WM, respectively) and between GM and cerebrospinal fluid (CSF), generating WM and pial surfaces, respectively. To correct for eventual defects on these surfaces, manual edits were performed when needed. The cortical thickness is given by the distance between WM and pial surfaces at each point. Finally, each surface was smoothed with a Gaussian filter of 15mm before statistical analysis.
Statistical analysis
Demographic data
Differences between asymptomatic and symptomatic subjects in demographic characteristics and between the episodic memory scores were assessed using Student’s t-test. The chi-squared test was used for testing relationships on categorical variables (sex and level of intellectual disability). A p-value of ≤ 0.05 was considered significant. The statistical analyses were performed using IBM SPSS Statistics Software (version 22).
Neuroimaging data
Initially, cortical thickness was compared between asymptomatic and symptomatic individuals with DS using FreeSurfer’s QDEC (Query, Design, Estimate, Contrast) interface in order to identify regions where cortical thickness is significantly reduced in symptomatic subjects and create a cortical atrophy map. Sex, age, and degree of intellectual disability were included as covariates. We then performed correlation analyses between the different memory performance scores and cortical thickness using a whole-brain vertex-wise analysis separately in asymptomatic and symptomatic participants with QDEC. To this purpose, a general linear model was applied, controlling for sex, age, and degree of intellectual disability. The results were corrected for multiple comparisons using Monte Carlo null-Z simulation (cluster analysis) as implemented in QDEC (p corrected = 0.05) [24].
Results
Demographic characteristics
Between May 1, 2013, and Jun 7, 2021, 148 participants met the inclusion criteria and were included in this study (see study flowchart in Supplementary Fig. 1). From this group, 9 participants with scan artifacts impairing automated image processing had to be excluded. From the remaining sample (139 participants), 85 were asymptomatic and 54 met criteria for prodromal AD or AD dementia (28 and 26 subjects, respectively. These subjects constituted the symptomatic AD group).
Demographic characteristics and episodic memory scores for these groups are shown in Table 1. As expected, symptomatic participants were significantly older and had lower performance on all mCRT scores than the asymptomatic participants (p < 0.001). There were no differences in sex (p = 0.46) or severity of intellectual disability (p = 0.29) between the asymptomatic and symptomatic groups.
Table 1.
Demographic characteristics and episodic memory scores
| All participants | Asymptomatic | Symptomatic | p-values | |
|---|---|---|---|---|
| (N = 139) | (N = 85) | (N = 54) | ||
| Age (mean, sd) | 43.6 (10.9) | 38.5 (10.1) | 52.2 (4.6) | <0.001 |
| Female sex, n (%) | 64 (46) | 37 (43.5) | 27 (50) | 0.456 |
| Severity of ID, n (%) | 0.288 | |||
| Mild | 46 (33.1) | 31 (36.5) | 15 (27.8) | |
| Moderate | 93 (66.9) | 54 (63.5) | 39 (72.2) | |
| Time between MRI and mCRT, days (mean, sd) | 108.2 (81.4) | 116.2 (79.0) | 95.5 (84.2) | 0.088 |
| mCRT scores (mean, sd) | ||||
| Trial 1 free recall (0–12) | 4.3 (2.3) | 5.4 (1.9) | 2.5 (1.7) | <0.001 |
| Trial 3 free recall (0–12) | 5.8 (2.9) | 7.7 (1.7) | 2.7 (2.6) | <0.001 |
| Free immediate recall score (0–36) | 15.3 (7.4) | 19.8 (4.7) | 7.7 (5.3) | <0.001 |
| Total immediate score (0–36) | 29.5 (9.3) | 35 (1.6) | 20.7 (9.9) | <0.001 |
| Free delayed recall score (0–12) | 5 (3.5) | 6.8 (2.7) | 1.7 (2.4) | <0.001 |
| Total delayed score (0–12) | 9.7 (3.3) | 11.5 (1.0) | 6.3 (3.6) | <0.001 |
p values are given for comparison between asymptomatic and symptomatic subjects. Age refers to the age recorded on the day of the mCRT administration. Brackets next to each episodic memory measure reflect the total possible range of scores
ID Intellectual disability, mCRT Modified Cued Recall Test
Pattern of cortical atrophy in participants with DS and symptomatic AD
Compared with asymptomatic adults with DS, individuals with DS and symptomatic AD showed cortical atrophy in the temporoparietal, precuneus-posterior cingulate, and occipital areas (Fig. 1).
Fig. 1.
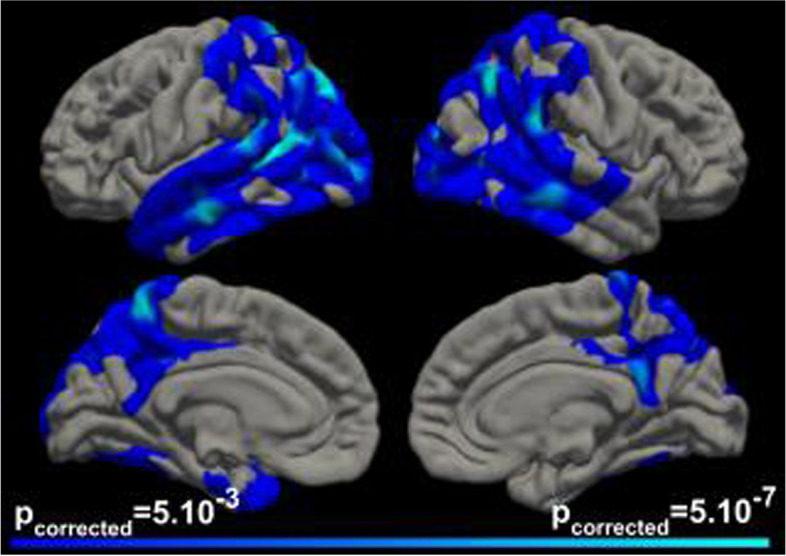
Pattern of cortical atrophy in symptomatic DS compared to asymptomatic DS participants. The clusters in blue-cyan scale represent the brain regions of statistically significant cortical atrophy in subjects with symptomatic Alzheimer’s disease (AD) compared to asymptomatic participants, after correction for multiple comparisons (threshold for significance p corrected = 0.05)
Cortical thickness and memory performance
Immediate recall
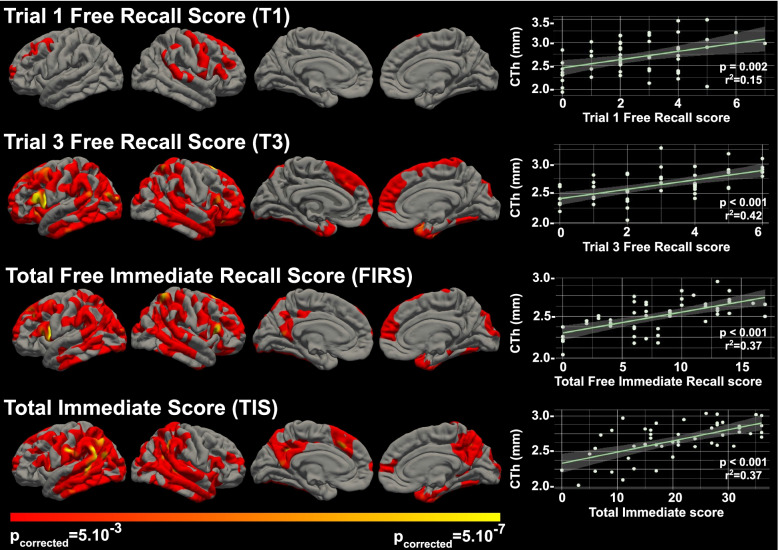
Figure 2 shows the correlation analyses between cortical thickness and the immediate memory scores (T1, T3, FIRS, and TIS) in symptomatic participants. T1 scores showed a significantly positive relationship with cortical thickness in the lateral prefrontal regions of both hemispheres. Performance on T3 correlated positively with cortical thickness in a more widespread network encompassing lateral regions in frontal, temporal, parietal, and occipital lobes, bilaterally, with the most significant clusters located in the left middle frontal, inferior frontal, supramarginal, and lingual gyri, and in the right temporal pole and superior frontal gyrus. T3 in addition also correlated with a small cluster located in the right parahippocampal cortex.
Fig. 2.
Correlation analyses between cortical thickness and immediate memory scores in symptomatic participants. The clusters in red-yellow scale represent the brain regions where each score was significantly related to cortical thickness after correction for multiple comparisons (threshold for significance p corrected = 0.05). Scatter plots on the right show the correlation between each score and the cortical thickness at the most significant vertex. CTh, cortical thickness (in millimeters)
The distribution of the clusters with significant association between cortical thickness and FIRS resembled that observed for T3, but with more widespread cortical atrophy also involving the right inferior frontal gyrus and superior parietal lobule, as well as the left posterior cingulate gyrus and precuneus.
TIS was mostly associated with cortical thickness in bilateral parieto-temporal regions, involving both precuneus and posterior cingulate gyri, and the right medial temporal lobe structures, namely the entorhinal and parahippocampal cortex (with p ≤ 0.05) (Fig. 2).
In asymptomatic participants, no clusters of significant association between immediate memory scores and cortical thickness were observed (Supplementary Fig. 2).
Delayed recall
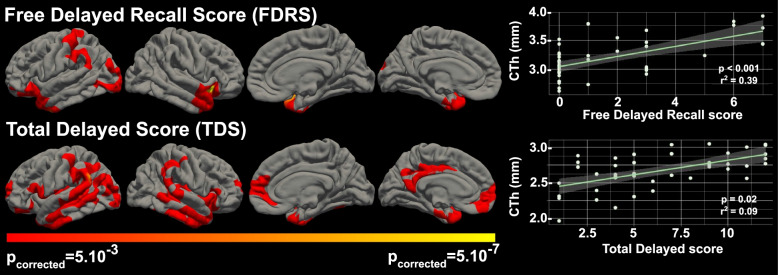
Figure 3 shows the correlation between cortical thickness and delayed memory scores (FDRS and TDS) in symptomatic participants. FDRS was associated with cortical thickness in areas mainly localized in anterior lateral and medial temporal regions, including both temporal poles, left entorhinal cortex, and right lateral superior and middle temporal gyri. We also found significant associations with cortical thickness in both inferior frontal gyri and in left postcentral and supramarginal gyri (p ≤ 0.05).
Fig. 3.
Correlation analyses between cortical thickness and delayed memory scores in symptomatic participants. The clusters in red-yellow scale represent the brain regions where each score was significantly related to cortical thickness after correction for multiple comparisons (threshold for significance p corrected = 0.05). Scatter plots on the right show the correlation between each score and the cortical thickness at the most significant vertex. CTh, cortical thickness (in millimeters)
TDS was significantly associated with cortical thickness in medial and lateral temporal lobe structures of both hemispheres. Clusters of significant correlation were also found in both supramarginal gyri, in the medial face of both superior frontal gyri, and in the left posterior cingulate gyrus (p≤0.05).
We did not find clusters of significant correlations between cortical thickness and delayed memory measures in asymptomatic participants (Supplementary Fig. 2).
Discussion
To our knowledge, this is the first study that investigates the neuroanatomical correlates of different processes involved in episodic memory in a cohort of adults with DS. Using structural MRI and an adapted episodic memory test, the mCRT, we found different correlations between the subtests assessing the different episodic memory processes and cortical thickness in symptomatic AD, suggesting that different anatomical regions play unique roles at different stages of episodic memory processing.
Adults with DS and symptomatic AD showed a pattern of cortical atrophy in lateral parieto-temporo-occipital cortices and in the posterior cingulate and precuneus. This pattern of widespread posterior cortical thinning has been previously described in amyloid-positive individuals with DS [25] and is very similar to that observed in sporadic AD [26].
Relationships in symptomatic individuals are detailed hereafter. The correlations between episodic memory measures and cortical thickness extended well beyond the medial temporal lobe structures. The different mCRT subscores were associated with cortical atrophy in different regions. T1 immediate free recall, a score that reflects initial encoding processes [13], mainly involved frontal lobe areas. FIRS, a score used to assess immediate retrieval [13], was associated with a widespread cortical network encompassing lateral frontal, parietal, temporal, and occipital regions, bilaterally, while TIS, which reflects immediate storage [13], was associated with more posterior cortical atrophy, including regions in the lateral parietal, temporal and occipital cortices, precuneus, posterior cingulate cortex, and medial temporal lobe cortices. In contrast to the immediate memory trials, delayed memory performance was related to a less distributed pattern of cortical atrophy, predominantly involving lateral and medial temporal lobe regions. Importantly, these associations were only seen in symptomatic participants and not in asymptomatic, possibly reflecting the smaller dynamic range of the mCRT scores and the milder atrophy in asymptomatic individuals [16]. To our knowledge, no other studies have related cortical thickness to different episodic memory measures in asymptomatic individuals with DS. In the general population, some authors have attempted to correlate FCSRT performance with volumetric measures of the hippocampal formation or entorhinal cortex in healthy aging subjects, without finding significant associations [27, 28].
Episodic memory impairment is a hallmark symptom of sporadic AD and DS-related AD during the early and middle stages of the disease [29–31] and has been traditionally associated with medial temporal lobe damage [32–34]. In the last decades, a growing number of neuroimaging studies have demonstrated that episodic memory does not depend solely on medial temporal structures. Several cognitive processes, like language, working memory, semantic processing, attention, and executive functions, which rely upon distinct brain networks, are necessary for successful episodic memory [13, 14, 35, 36]. We found that performance on T1 of the mCRT was associated with cortical thickness in frontal lobe regions, but not in medial temporal lobe structures in subjects with symptomatic AD, thus indicating that initial encoding was more dependent on regions supporting cognitive functions such as attention, working memory, or executive functions. Ventrolateral and dorsolateral prefrontal cortex have been described to implement control processes that support memory encoding (e.g., selection processes that direct attention toward goal-relevant information or organizational processes necessary for optimal memory encoding, respectively) [37, 38]. Our results are in line with those reported by Wolk et al. in sporadic AD, who also found a lack of influence of medial temporal structures during early list learning trials in patients with mild AD from the general population [14].
The temporal pole consistently appeared to be related to immediate and delayed memory measures (with the exception of T1 immediate recall) in our study. In this sense, functional neuroimaging studies have related the anterior temporal lobes to semantic processing (mental representations of the meaning of words, objects, people, and factual information about the world) [39, 40]. The semantic organization of the information that has to be encoded allows for a deeper processing and thus facilitates posterior retrieval processes. Our results are again consistent with previous studies relating the atrophy of the temporal pole with poor immediate recall performance in patients with early-stage AD in the general population [14, 35].
In addition, episodic memory performance (particularly T3, FIRS and TIS immediate recall scores, and TDS) was related to cortical thickness in the lateral posterior parietal cortex (PPC). These areas have been implicated in episodic memory retrieval because of their role in attentional processes (attention to internal representations). The dorsal PPC is implicated in the voluntary orienting of attention to relevant stimuli based on retrieval goals (top-down attentional processes), while the ventral PPC responds to the detection of new behaviorally relevant stimuli which are outside the focus of attention defined by the dorsal PPC (bottom-up attentional processes) [41, 42]. Moreover, inferior parietal regions (together with regions within the inferior frontal gyrus) have been associated with the storage function of the phonological loop and the visuospatial sketchpad supporting speech-based and visuospatial working memory, respectively [43]. Working memory, which temporarily maintains and stores information, is crucial for the transfer of information into a long-term store. Other regions also reported to be supportive of working memory are the posterior superior temporal gyrus, the premotor cortex, and the dorsolateral prefrontal cortex [44], regions found to be strongly associated with immediate memory measures in our study.
Atrophy in medial temporal lobe (MTL) structures, in particular the entorhinal and parahippocampal cortices, was found to be related to TIS and TDS. Low scores on both measures reflect storage deficits (immediate and delayed storage, respectively). Many studies support the role of MTL regions in the acquisition and maintenance of episodic memory [3, 13, 36]. The association between TIS and atrophy of extrahippocampal MTL regions in the right hemisphere in our study is not in line with previous research using the FCSRT or the RAVLT in patients with sporadic AD [35, 36]. One possible explanation for these discrepancies could be the visual nature of the stimuli used in the present study, unlike the FCSRT used in the general population, which is based on the learning of verbal stimuli. Similarly, we found that immediate and delayed recall measures were also associated with atrophy in occipital regions. Ventral occipito-temporal regions have been related to visual object processing [45]. Poor performance on some mCRT measures, which is based on the recall of familiar line drawings, could partly be explained by damage to this visual processing pathway.
Performance on the TIS, and to a lesser extent the TDS, was also associated with posteromedial regions, namely the precuneus and the posterior cingulate cortex. Recent functional imaging studies have suggested that memory function is subserved by a set of distributed networks which include the medial temporal lobe system and a set of cortical regions collectively referred to as the default mode network (DMN). The regions of the DMN include the anteromedial prefrontal cortex, the posterior cingulate cortex, the precuneus, the angular gyrus, and the medial temporal lobe [46]. Some authors have demonstrated that early amyloid deposition in sporadic AD occurs in areas mainly located within the DMN [47] and that this network plays a key role orienting the focus of attention to stored representational knowledge [48]. In DS subjects, recent research found that reductions in DMN connectivity to posterior brain regions were linked to the presence of AD neuropathology in a pattern substantially similar to that seen in sporadic AD [49, 50], even in asymptomatic individuals. Nevertheless, the association between the effects of AD neuropathology on the DMN and performance on episodic memory measures in the DS population has not been described.
Our results have several implications. First, the pattern of cortical atrophy observed in individuals with DS and AD in our study is remarkably similar to that described in the general population with AD [26], indicating that AD in individuals with DS targets the same cortical regions affected in sporadic AD. Second, the cortical areas found to be related to episodic memory in our study are in line with those previously described in sporadic AD. Our results thus reinforce the similarities between AD in DS and sporadic AD. Third, our results also support the use of the mCRT as a potential neuropsychological marker for the diagnosis of AD in people with DS, and particularly the TIS and the TDS (measures reflecting storage deficits), as these scores correlated well with medial temporal lobe regions, areas considered to be the earliest affected by AD neuropathology.
Our study has several strengths, including the large sample size of subjects included in our study with symptomatic AD, as well as the fact that our participants come from a large population-based cohort of adults with DS.
Limitations
We also acknowledge some limitations. First, this is a single-center study and did not include an age-matched control group of euploid individuals to test the neurodevelopmental abnormalities. Thus, our results need to be replicated in other cohorts to confirm the generalizability of our results. Second, as patients with prodromal AD and AD dementia were analyzed together, it has not been possible to characterize AD in its earliest stages. Future studies should include a more homogeneous sample of subjects with prodromal AD to better characterize the early stages of AD in this population.
Conclusions
Our results provide new evidence about the neuroanatomical correlates of the episodic memory deficits in adults with DS and symptomatic AD that largely corresponds with the findings described in sporadic AD. This study further supports the similarities of the AD pathophysiological process in sporadic AD and in DS.
Supplementary Information
Additional file 1: Supplementary Figure 1. Study flow chart. Description of data: flow chart of included and excluded subjects. Footnote: ID, intellectual disability; mCRT, modified Cued Recall Test; MRI, magnetic resonance imaging; OCD, obsessive compulsive disorder.
Additional file 2. MRI acquisition protocols.
Additional file 3: Supplementary Figure 2. Correlation analyses between cortical thickness and immediate and delayed memory scores in asymptomatic participants. Legend: Brain regions where each score was significantly related to cortical thickness (threshold for significance p corrected = 0.05, after correction for multiple comparisons). No clusters of significant correlation between cortical thickness and immediate or delayed memory scores were observed in asymptomatic participants.
Acknowledgements
The authors would like to thank all the participants with Down’s syndrome, their families, and their carers for their support of and dedication to this research. We also acknowledge the Fundació Catalana Síndrome de Down for global support, Tania Martínez and Cecilia Mota for administrative support, and Concepción Escolá and Diana Garzón for nursing handling.
Abbreviations
- AD
Alzheimer’s disease
- CAMCOG-DS
Cambridge Cognitive Examination for Older Adults with Down’s syndrome
- CAMDEX-DS
Cambridge Examination for Mental Disorders of Older People with Down syndrome and others with intellectual disabilities
- CSF
Cerebrospinal fluid
- DABNI
Down Alzheimer Barcelona Neuroimaging Initiative
- DMN
Default mode network
- DS
Down’s syndrome
- FCSRT
Free and Cued Selective Reminding Test
- FDRS
Free delayed recall score
- FIRS
Free immediate recall score
- GM
Gray matter
- ID
Intellectual disability
- mCRT
Modified Cued Recall Test
- MRI
Magnetic resonance imaging
- MTL
Medial temporal lobe
- PPC
Posterior parietal cortex
- QDEC
Query, Design, Estimate, Contrast
- RAVLT
Rey Auditory Verbal Learning Test
- T1
Trial 1 free immediate recall
- T3
Trial 3 free immediate recall
- TDS
Total delayed score
- TIS
Total immediate score
- WM
White matter
Authors’ contributions
JF and BB designed the study. Acquisition of the data was performed by BB, MRA, LV, CP, SV, SF, MA, MCI, IB, VM, JP, SGO, and JF. BB, MRA, and JF performed the data analysis. BB, MRA, and JF interpreted the data and wrote the manuscript. All authors reviewed the full paper providing important intellectual content and approved the final manuscript.
Funding
This study was supported by the Fondo de Investigaciones Sanitario (FIS), Instituto de Salud Carlos III (PI14/01126, PI17/01019, and PI20/01473 to Juan Fortea; PI13/01532 and PI16/01825 to Rafael Blesa; PI18/00335 to Maria Carmona-Iragui; INT19/00016 and PI18/00435 to Daniel Alcolea; and PI14/1561 and PI17/01896 to Alberto Lleó) and the CIBERNED program (Program 1, Alzheimer Disease to Alberto Lleó and SIGNAL study, www.signalstudy.es), partly jointly funded by Fondo Europeo de Desarrollo Regional, Unión Europea, Una manera de hacer Europa. This work was also supported by the National Institutes of Health (NIA grants 1R01AG056850–01A1; R21AG056974 and R01AG061566 to Juan Fortea), Fundació La Marató de TV3 (20141210 to Juan Fortea and 044412 to Rafael Blesa), Fundació Catalana Síndrome de Down, and Fundació Víctor Grífols i Lucas partially supported this work. This work was also supported by Generalitat de Catalunya (SLT006/17/00119 to Juan Fortea, SLT002/16/00408 to Alberto Lleó, and SLT006/17/00125 to Daniel Alcolea) and a grant from the Fundació Bancaria La Caixa to Rafael Blesa. Horizon 21 Consortium is partly funded by Jérôme Lejeune Foundation (clinical and trial outcome measures for dementia in individuals with Down syndrome). Maria Florencia Iulita acknowledges financial support from the Jerome Lejeune Foundation. Dr Bejanin was supported by a Miguel Servet I grant (CP20/00038) from the Carlos III Health Institute.
The article describes independent research, and the views expressed are those of the authors and not necessarily those of the funders. The sponsors of the study did not take part in the design and conduct of the study; collection, management, analysis, and interpretation of the data; writing and review of the report; or the decision to submit the article for publication.
Availability of data and materials
The anonymized data used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declarations
Ethics approval and consent to participate
The study was approved by the Sant Pau Ethics Committee following the standards for medical research in humans recommended by the Declaration of Helsinki and reported to the Ministry of Justice according to the Spanish law for research in people with intellectual disabilities. All participants or their legally authorized representative gave written informed consent before enrollment.
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Bessy Benejam and Mateus Rozalem Aranha have equally contributed to this manuscript.
References
- 1.Ballard C, Mobley W, Hardy J, Williams G, Corbett A. Dementia in Down’s syndrome. Lancet Neurol. 2016;15:622–636. doi: 10.1016/S1474-4422(16)00063-6. [DOI] [PubMed] [Google Scholar]
- 2.Wiseman FK, Al-Janabi T, Hardy J, Karmiloff-Smith A, Nizetic D, Tybulewicz VLJ, et al. A genetic cause of Alzheimer disease: mechanistic insights from Down syndrome. Nat Rev Neurosci. 2015;16:564–574. doi: 10.1038/nrn3983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dickerson BC, Eichenbaum H. The episodic memory system: neurocircuitry and disorders. Neuropsychopharmacology. 2010;35:86–104. doi: 10.1038/npp.2009.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Arnold SE, Hyman BT, Flory J, Damasio AR, Van Hoesen GW. The topographical and neuroanatomical distribution of neurofibrillary tangles and neuritic plaques in the cerebral cortex of patients with Alzheimer’s disease. Cerebral cortex (New York, NY : 1991) 1991;1:103–116. doi: 10.1093/cercor/1.1.103. [DOI] [PubMed] [Google Scholar]
- 5.Braak H, Braak E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991;82:239–259. doi: 10.1007/BF00308809. [DOI] [PubMed] [Google Scholar]
- 6.Fortea J, Vilaplana E, Carmona-Iragui M, Benejam B, Videla L, Barroeta I, et al. Clinical and biomarker changes of Alzheimer’s disease in adults with Down syndrome: a cross-sectional study. Lancet (London, England). 2020;395:1988–1997. doi: 10.1016/S0140-6736(20)30689-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Annus T, Wilson LR, Hong YT, Acosta-Cabronero J, Fryer TD, Cardenas-Blanco A, et al. The pattern of amyloid accumulation in the brains of adults with Down syndrome. Alzheimers Dement. 2016;12:538–545. doi: 10.1016/j.jalz.2015.07.490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Neale N, Padilla C, Fonseca LM, Holland T, Zaman S. Neuroimaging and other modalities to assess Alzheimer’s disease in Down syndrome. NeuroImage Clin. 2018;17:263–271. doi: 10.1016/j.nicl.2017.10.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lautarescu BA, Holland AJ, Zaman SH. The early presentation of dementia in people with Down syndrome: a systematic review of longitudinal studies. Neuropsychol Rev. 2017;27:31–45. doi: 10.1007/s11065-017-9341-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fortea J, Zaman SH, Hartley S, Rafii MS, Head E, Carmona-Iragui M. Alzheimer’s disease associated with Down syndrome: a genetic form of dementia. Lancet Neurol. 2021;20:930-42. [DOI] [PMC free article] [PubMed]
- 11.Hartley SL, Handen BL, Devenny D, Tudorascu D, Piro-Gambetti B, Zammit MD, et al. Cognitive indicators of transition to preclinical and prodromal stages of Alzheimer’s disease in Down syndrome. Alzheimers Dement. 2020;12:e12096. doi: 10.1002/dad2.12096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Startin CM, Hamburg S, Hithersay R, Al-Janabi T, Mok KY, Hardy J, et al. Cognitive markers of preclinical and prodromal Alzheimer’s disease in Down syndrome. Alzheimers Dementia. 2019;15:245–257. doi: 10.1016/j.jalz.2018.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Epelbaum S, Bouteloup V, Mangin JF, La Corte V, Migliaccio R, Bertin H, et al. Neural correlates of episodic memory in the Memento cohort. Alzheimers Dementia (New York, N Y). 2018;4:224–233. doi: 10.1016/j.trci.2018.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wolk DA, Dickerson BC. Fractionating verbal episodic memory in Alzheimer’s disease. NeuroImage. 2011;54:1530–1539. doi: 10.1016/j.neuroimage.2010.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Devenny DA, Zimmerli EJ, Kittler P, Krinsky-McHale SJ. Cued recall in early-stage dementia in adults with Down’s syndrome. J Intellect Disabil Res. 2002;46:472–483. doi: 10.1046/j.1365-2788.2002.00417.x. [DOI] [PubMed] [Google Scholar]
- 16.Benejam B, Videla L, Vilaplana E, Barroeta I, Carmona-Iragui M, Altuna M, et al. Diagnosis of prodromal and Alzheimer’s disease dementia in adults with Down syndrome using neuropsychological tests. Alzheimers Dement. 2020;12:e12047. [DOI] [PMC free article] [PubMed]
- 17.Bazydlo A, Zammit M, Wu M, Dean D, Johnson S, Tudorascu D, et al. White matter microstructure associations with episodic memory in adults with Down syndrome: a tract-based spatial statistics study. J Neurodevelopment Disord. 2021;13:17. doi: 10.1186/s11689-021-09366-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Esteba-Castillo S, Dalmau-Bueno A, Ribas-Vidal N, Vilà-Alsina M, Novell-Alsina R, García-Alba J. Adaptation and validation of CAMDEX-DS (Cambridge Examination for Mental Disorders of Older People with Down’s Syndrome and others with intellectual disabilities) in Spanish population with intellectual disabilities. Rev Neurol. 2013;57:337–346. [PubMed] [Google Scholar]
- 19.American Psychiatric Association . Diagnostic and statistical manual of mental disorders, fifth edition. Arlington: American Psychiatric Association; 2013. [Google Scholar]
- 20.Fortea J, Carmona-Iragui M, Benejam B, Fernández S, Videla L, Barroeta I, et al. Plasma and CSF biomarkers for the diagnosis of Alzheimer’s disease in adults with Down syndrome: a cross-sectional study. Lancet Neurol. 2018;17:860–869. doi: 10.1016/S1474-4422(18)30285-0. [DOI] [PubMed] [Google Scholar]
- 21.Fischl B, Sereno MI, Dale AM. Cortical surface-based analysis. II: inflation, flattening, and a surface-based coordinate system. NeuroImage. 1999;9:195–207. doi: 10.1006/nimg.1998.0396. [DOI] [PubMed] [Google Scholar]
- 22.Fischl B, Dale AM. Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proc National Acad Sci U S A. 2000;97:11050–11055. doi: 10.1073/pnas.200033797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fischl B, Salat DH, van der Kouwe AJW, Makris N, Ségonne F, Quinn BT, et al. Sequence-independent segmentation of magnetic resonance images. NeuroImage. 2004;23(Suppl 1):S69–S84. doi: 10.1016/j.neuroimage.2004.07.016. [DOI] [PubMed] [Google Scholar]
- 24.Hagler DJJ, Saygin AP, Sereno MI. Smoothing and cluster thresholding for cortical surface-based group analysis of fMRI data. NeuroImage. 2006;33:1093–1103. doi: 10.1016/j.neuroimage.2006.07.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Annus T, Wilson LR, Acosta-Cabronero J, Cardenas-Blanco A, Hong YT, Fryer TD, et al. The Down syndrome brain in the presence and absence of fibrillar β-amyloidosis. Neurobiol Aging. 2017;53:11–19. doi: 10.1016/j.neurobiolaging.2017.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dickerson BC, Bakkour A, Salat DH, Feczko E, Pacheco J, Greve DN, et al. The cortical signature of Alzheimer’s disease: regionally specific cortical thinning relates to symptom severity in very mild to mild AD dementia and is detectable in asymptomatic amyloid-positive individuals. Cerebral Cortex. 2009;19:497–510. [DOI] [PMC free article] [PubMed]
- 27.Sánchez-Benavides G, Gómez-Ansón B, Molinuevo JL, Blesa R, Monte GC, Buschke H, et al. Medial temporal lobe correlates of memory screening measures in normal aging, MCI, and AD. J Geriatr Psychiatry Neurol. 2010;23:100–108. doi: 10.1177/0891988709355271. [DOI] [PubMed] [Google Scholar]
- 28.Rizzolo L, Narbutas J, Van Egroo M, Chylinski D, Besson G, Baillet M, et al. Relationship between brain AD biomarkers and episodic memory performance in healthy aging. Brain Cogn. 2021;148:105680. doi: 10.1016/j.bandc.2020.105680. [DOI] [PubMed] [Google Scholar]
- 29.Benejam B, Fortea J, Molina-López R, Videla S. Patterns of performance on the modified Cued Recall Test in Spanish adults with down syndrome with and without dementia. Am J Intellect Dev Disabil. 2015;120:481-9. [DOI] [PubMed]
- 30.Blok JB, Scheirs JGM, Thijm NS. Personality and behavioural changes do not precede memory problems as possible signs of dementia in ageing people with Down syndrome. Int J Geriatr Psychiatry. 2017;32:1257–1263. doi: 10.1002/gps.4606. [DOI] [PubMed] [Google Scholar]
- 31.Krinsky-McHale SJ, Devenny DA, Silverman WP. Changes in explicit memory associated with early dementia in adults with Down’s syndrome. J Intellect Disabil Res. 2002;46:198–208. doi: 10.1046/j.1365-2788.2002.00365.x. [DOI] [PubMed] [Google Scholar]
- 32.Aylward EH, Li Q, Honeycutt NA, Warren AC, Pulsifer MB, Barta PE, et al. MRI volumes of the hippocampus and amygdala in adults with Down’s syndrome with and without dementia. Am J Psychiatry. 1999;156:564–568. doi: 10.1176/ajp.156.4.564. [DOI] [PubMed] [Google Scholar]
- 33.Krasuski JS, Alexander GE, Horwitz B, Rapoport SI, Schapiro MB. Relation of medial temporal lobe volumes to age and memory function in nondemented adults with Down’s syndrome: implications for the prodromal phase of Alzheimer’s disease. Am J Psychiatry. 2002;159:74–81. doi: 10.1176/appi.ajp.159.1.74. [DOI] [PubMed] [Google Scholar]
- 34.Pujol J, Fenoll R, Ribas-Vidal N, Martínez-Vilavella G, Blanco-Hinojo L, García-Alba J, et al. A longitudinal study of brain anatomy changes preceding dementia in Down syndrome. NeuroImage Clin. 2018;18:160–166. doi: 10.1016/j.nicl.2018.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Putcha D, Brickhouse M, Wolk DA, Dickerson BC. Fractionating the Rey Auditory Verbal Learning Test: distinct roles of large-scale cortical networks in prodromal Alzheimer’s disease. Neuropsychologia. 2019;129:83–92. doi: 10.1016/j.neuropsychologia.2019.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sarazin M, Chauviré V, Gerardin E, Colliot O, Kinkingnéhun S, de Souza LC, et al. The amnestic syndrome of hippocampal type in Alzheimer’s disease: an MRI study. J Alzheimer’s Dis. 2010;22:285–294. doi: 10.3233/JAD-2010-091150. [DOI] [PubMed] [Google Scholar]
- 37.Blumenfeld RS, Ranganath C. Prefrontal cortex and long-term memory encoding: an integrative review of findings from neuropsychology and neuroimaging. Neuroscientist. 2007;13:280–291. doi: 10.1177/1073858407299290. [DOI] [PubMed] [Google Scholar]
- 38.Fletcher PC, Shallice T, Dolan RJ. The functional roles of prefrontal cortex in episodic memory. I. Encoding. Brain. 1998;121(Pt 7):1239–1248. doi: 10.1093/brain/121.7.1239. [DOI] [PubMed] [Google Scholar]
- 39.Patterson K, Nestor PJ, Rogers TT. Where do you know what you know? The representation of semantic knowledge in the human brain. Nat Rev Neurosci. 2007;8:976–987. doi: 10.1038/nrn2277. [DOI] [PubMed] [Google Scholar]
- 40.Visser M, Jefferies E, Lambon Ralph MA. Semantic processing in the anterior temporal lobes: a meta-analysis of the functional neuroimaging literature. J Cogn Neurosci. 2010;22:1083–1094. doi: 10.1162/jocn.2009.21309. [DOI] [PubMed] [Google Scholar]
- 41.Cabeza R. Role of parietal regions in episodic memory retrieval: the dual attentional processes hypothesis. Neuropsychologia. 2008;46:1813–1827. doi: 10.1016/j.neuropsychologia.2008.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Corbetta M, Shulman GL. Control of goal-directed and stimulus-driven attention in the brain. Nat Rev Neurosci. 2002;3:201–215. doi: 10.1038/nrn755. [DOI] [PubMed] [Google Scholar]
- 43.Baddeley A. Working memory: looking back and looking forward. Nat Rev Neurosci. 2003;4:829–839. doi: 10.1038/nrn1201. [DOI] [PubMed] [Google Scholar]
- 44.Buchsbaum BR, D’Esposito M. The search for the phonological store: from loop to convolution. J Cogn Neurosci. 2008;20:762–778. doi: 10.1162/jocn.2008.20501. [DOI] [PubMed] [Google Scholar]
- 45.Cabeza R, Nyberg L. Imaging cognition II: an empirical review of 275 PET and fMRI studies. J Cogn Neurosci. 2000;12:1–47. doi: 10.1162/08989290051137585. [DOI] [PubMed] [Google Scholar]
- 46.Jeong W, Chung CK, Kim JS. Episodic memory in aspects of large-scale brain networks. Front Human Neurosci. 2015;9:454. doi: 10.3389/fnhum.2015.00454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Palmqvist S, Schöll M, Strandberg O, Mattsson N, Stomrud E, Zetterberg H, et al. Earliest accumulation of β-amyloid occurs within the default-mode network and concurrently affects brain connectivity. Nat Commun. 2017;8:1214. doi: 10.1038/s41467-017-01150-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Spreng RN, Grady CL. Patterns of brain activity supporting autobiographical memory, prospection, and theory of mind, and their relationship to the default mode network. J Cogn Neurosci. 2010;22:1112–1123. doi: 10.1162/jocn.2009.21282. [DOI] [PubMed] [Google Scholar]
- 49.Wilson LR, Vatansever D, Annus T, Williams GB, Hong YT, Fryer TD, et al. Differential effects of Down’s syndrome and Alzheimer’s neuropathology on default mode connectivity. Human Brain Mapping. 2019;40:4551–4563. doi: 10.1002/hbm.24720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rosas HD, Lewis LR, Mercaldo ND, Nasr S, Brickman AM, Siless V, et al. Altered connectivity of the default mode network in cognitively stable adults with Down syndrome: “Accelerated aging” or a prelude to Alzheimer’s disease? Alzheimers Dement. 2021;13:e12105. doi: 10.1002/dad2.12105. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Supplementary Figure 1. Study flow chart. Description of data: flow chart of included and excluded subjects. Footnote: ID, intellectual disability; mCRT, modified Cued Recall Test; MRI, magnetic resonance imaging; OCD, obsessive compulsive disorder.
Additional file 2. MRI acquisition protocols.
Additional file 3: Supplementary Figure 2. Correlation analyses between cortical thickness and immediate and delayed memory scores in asymptomatic participants. Legend: Brain regions where each score was significantly related to cortical thickness (threshold for significance p corrected = 0.05, after correction for multiple comparisons). No clusters of significant correlation between cortical thickness and immediate or delayed memory scores were observed in asymptomatic participants.
Data Availability Statement
The anonymized data used and/or analyzed during the current study are available from the corresponding author upon reasonable request.