Abstract
Background
Combining pathogen reduction and automated separation of whole blood (WB), together with the use of improved additive solutions, may increase reproducibility and extend shelf-life of blood components.
Materials and methods
Forty WB units were collected from volunteer donors and randomised 1:1 into two groups: 1) pathogen reduction with riboflavin and ultraviolet light (PRT); or 2) no treatment (Control). After two hours (h) at room temperature, all units underwent fully automated separation into red blood cell concentrate (RBCC), plasma and leukopack components. RBCCs were leukoreduced and stored in phosphate-adenine-glucose-guanosine-saline-mannitol (PAGGSM) solution while plasma units were shock frozen within 8 h of collection and stored at ≤ −25°C. RBCCs were sampled on day 1 and weekly thereafter until day 42, while plasma was sampled on days 1 and 30. The main study objective was to assess the in vitro quality of separated RBCCs using biochemical and haematological parameters. Plasma protein content after one cycle of freeze-thaw was also analysed.
Results
The quality of RBCCs was largely comparable between the PRT and Control groups, except for a significantly higher degree of haemolysis and extracellular potassium levels in the PRT group after 35 days of storage. While potassium concentration was significantly higher in the PRT group at all timepoints, the degree of haemolysis exceeded the accepted European threshold (i.e., <0.8% of red cell mass in ≥ 90.0% of tested units) after day 35. Most plasma protein levels were significantly lower in the PRT than the Control group at both day 1 and day 30.
Discussion
Pathogen reduction with riboflavin and ultraviolet light treatment of WB can be combined with fully automated separation to obtain RBCCs that may be stored for up to 35 days in PAGGSM solution with acceptable quality, comparable to that of RBCCs from untreated blood. The relative differences between factor concentrations in plasma from the PRT and the Control groups were similar during the 30-day storage.
Keywords: blood component safety, red blood cells, plasma, pathogen-reduced
INTRODUCTION
The last decades have seen a significant increase in the safety of blood transfusion. Examples of developments contributing to blood safety are closed system disposables with sample pouch for blood collection and processing, improved arm disinfection methods, and increasingly sensitive and specific screening tests for common pathogens (such as hepatitis B and C virus, human immunodeficiency virus, and syphilis)1–4. However, challenges to ensuring safe blood supplies remain, and these show how preventing transmission of infectious diseases through blood transfusion needs to be sustained by co-ordinated reactive and proactive measures1,2. These challenges include the limitations of detection methods for infectious disease markers (e.g., a window during which a pathogen’s nucleic acids or antigens are undetectable), the (re-) emergence of blood-borne pathogens, and the continuous risk of bacterial contamination, especially in platelet concentrates1,2. The outbreaks of emerging pathogens seen in recent decades, culminating with the recent global pandemic of coronavirus disease caused by the severe acute respiratory syndrome-coronavirus 2, exemplify the need for a reliable supply of safe blood products5. In addition, contamination of red blood cell (RBC) and platelet components with leukocytes may result in serious adverse reactions, such as transfusion-associated graft-versus-host disease (TA-GvHD)1.
Consequently, pathogen reduction and leukocyte inactivation methods may significantly contribute to blood transfusion safety6–9. In the first decade of the 21st century, pathogen reduction technologies (PRT) for plasma and platelet concentrates gradually became part of routine blood component processing10. In both pre-clinical and clinical studies, pathogen-reduced blood components have been demonstrated to offer adequate safety standards1,2,11–17. In 2015, a whole blood (WB) treatment based on riboflavin (RF) and UV light (Mirasol® PRT, Terumo Blood and Cell Technologies, Lakewood, CO, USA) was granted a European Union certification of conformity. The combination of RF and UV light (most in the range of 280 nm-350 nm, with a 313 nm peak) induces irreversible DNA damage sufficient for the inactivation of pathogens and leukocytes, while largely preserving the integrity of RBC concentrates ( RBCCs)18. RBCCs are the most often transfused blood component19 and the development of several generations of additive solutions has enabled their prolonged storage20,21.
Whole blood is rarely transfused in countries with a medium or high development index, except in strictly specified clinical indications (e.g., resuscitation of severe traumatic haemorrhage)18,22. PRT-treated WB can be used for WB transfusion, and some in vitro and in vivo studies employing PRT-treated WB showed acceptable quality of components, also when compared to gamma-irradiated or untreated components1,23–28. Many of these studies were based on combinations of PRT and manual or semi-automated systems for WB separation. Laboratories employing automated blood processing have reported time savings, without any significant impact on the blood component quality compared to manual processing7,8,29–31. The main aim of this study was to evaluate the in vitro quality of RBCCs obtained from WB subjected to pathogen inactivation with the Mirasol® PRT system and subjected to fully automated separation (FAS), compared to RBCCs from untreated WB separated in the same way. For this purpose, several in vitro parameters, including those defined by the European Directorate for the Quality of Medicines and Healthcare (EDQM) standards32, as well as Polish national guidelines33 were used to assess the quality of the RBCCs produced. Furthermore, in vitro properties of plasma derived from PRT-treated and untreated WB were descriptively evaluated. The quality of the resulting blood components is discussed and compared to published data using different blood processing methods.
MATERIALS AND METHODS
Study objectives
The primary objective of this study was to evaluate the quality of RBCC units resulting from the pathogen reduction treatment of WB and separation using FAS. A secondary objective was to describe the quality of plasma units after a cycle of freezing and thawing.
Blood donations
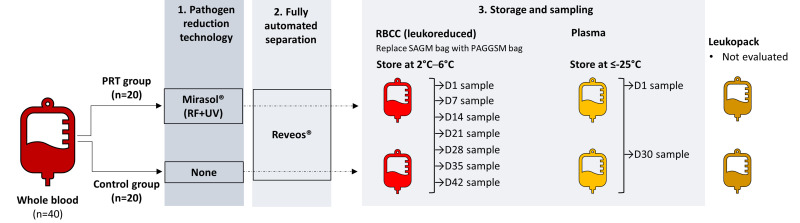
This study was conducted in the Regional Blood Transfusion Centre in Łódź, Poland. Forty units of WB with an approximate volume of 450 mL were collected from healthy adult volunteer donors, who were eligible according to Polish regulations based on EDQM recommendations33. All WB donations were collected into FAS leukoreduction (LR) sets (Reveos®, Terumo Blood and Cell Technologies) containing 63 mL of the anticoagulant citrate-phosphate-dextrose (CPD). The donations were randomly assigned to two groups at a 1:1 ratio: 20 WB donations were subjected to PRT treatment (PRT group), and 20 WB donations were left untreated (Control group) (Figure 1). The study received ethical approval from the Bioethics Commission at the Institute of Haematology and Transfusion Medicine in Warsaw, Poland.
Figure 1.
Study design
D1, D7, D14, D21, D28, D30, D35, and D42: sampling points after indicated days of storage; n: number of whole blood units processed in each group; PAGGSM: phosphate-adenine-glucose-guanosine-saline-mannitol; PRT: pathogen reduction technology; RBCC: red blood cell concentrate; RF: riboflavin; SAGM: saline-adenine-glucose-mannitol; UV: ultraviolet light.
Blood processing: pathogen reduction with riboflavin and ultraviolet light, and separation procedures
All WB units were left to rest for 2 h at room temperature (22–24°C) before automated separation. After the resting period, the Control group units were kept in the same FAS LR sets, while the PRT group WB units underwent pathogen reduction as follows: the units were transferred into an illumination bag (Mirasol® WB disposable kit, Terumo Blood and Cell Technologies), 35 mL of 500 μM RF solution (Terumo Blood and Cell Technologies) was added, and the units were subjected to pathogen and leukocyte inactivation according to the manufacturer’s instructions (Mirasol® Whole Blood Disposable Kit Instructions for Use Part n. 777714-015B, 11/2019; Terumo Blood and Cell Technologies). The average illumination lasted for 47.0±4.0 minutes (min), during which time each unit was illuminated with a UV light dose adjusted per volume of RBCC units to 80 J/mLRBC.
Subsequently, the PRT-treated units were transferred into new FAS LR sets, and all (PRT and Control) units were subjected to separation by the FAS LR system (Reveos® LR Set, Terumo Blood and Cell Technologies; 2 Components [2C] protocol), yielding RBCC, plasma and l eukopack components. All RBCC units were leukoreduced with in-line filters included in the FAS LR sets and the bag containing saline-adenine-glucose-mannitol (SAGM) was replaced by a bag with 100 mL of the phosphate-adenine-glucose-guanosine-saline-mannitol (PAGGSM) solution (Macopharma, Tourcoing, France), in which RBCCs were stored at 2–6°C for 42 days. The plasma units were shock frozen (MABAG, Barsbüttel, Germany) within 8 h from blood separation and stored at ≤ −25°C for 30 days. The leukopack fractions were not analysed in this study.
Red blood cell concentrate and plasma parameters
Blood groups (A, B, O) were determined as part of baseline donor characteristics by ABO typing. Two methods were simultaneously used for ABO typing: the microcolumn agglutination method (IHA-1000 analyser, Diahem, Kraków, Poland), and the microplate blood testing method (PK-7300 analyser, Beckman Coulter, Brea, CA, USA) according to the manufacturer’s instructions. The quality of all RBCC units was evaluated with respect to the following variables (quality requirement threshold defined by EDQM32 or Polish national guidelines33 indicated in brackets): pH, mean corpuscular volume (MCV), haemolysis ( <0.8% haemolysis of RBC mass in ≥ 90.0% of tested RBCCs), haemoglobin concentration (≥ 40 g/unit in ≥ 90.0% of tested RBCCs), haematocrit (50.0–70.0% in ≥ 90.0% of tested RBCCs), extracellular potassium, 2,3-diphosphoglycerate (DPG), and glucose concentration. Samples to evaluate haematological and biochemical RBCC parameters were collected at seven sequential weekly timepoints, starting from one day after FAS (day 1, day 7, day 14, day 21, day 28, day 35, and day 42).
Plasma samples were collected on day 1 and day 30 and were assessed after one freeze/thaw cycle using the following parameters: prothrombin time (PT [sec]), International Normalised Ratio (INR), activated partial thromboplastin time (aPTT), and fibrinogen, Factor VIII and Factor IX, von Willebrand factor (vWF) and Protein S concentration.
Measurements of in vitro parameters of leukoreduced red blood cell concentrates
The pH value was measured using a pH meter (744 pH meter, Metrohm, Herisau, Switzerland). A haematology analyser (XN-550, Sysmex, Kobe, Japan) was used to measure haemoglobin concentration, haematocrit value, and MCV (femtoliter [fL]). Haemolysis was measured in supernatants obtained after centrifugation of leukoreduced RBCCs, using the HemoCue®Plasma/Low Hb system (Hemocue, Ängelholm, Sweden). Potassium and glucose concentrations were determined using the ABL90 FLEX PLUS analyser (Radiometer, Copenhagen, Denmark). The 2,3-DPG concentration was measured with a commercial spectrometer-based diagnostic test (2,3-Diphosphoglycerate test kit; cat. n. 10148334001, Sigma-Aldrich, Darmstadt, Germany).
Measurements of in vitro parameters of plasma units
All plasma-associated parameters were measured using the following kits of the ACL TOP 500 analyser (Werfen, Barcelona, Spain), according to the manufacturer’s instructions: PT (sec) was determined with the HemosIL RecombiPlasTin 2G kit and was used to calculate the INR using the following conversion parameters: reference plasma value of −11.5 seconds and International Sensitivity Index of −0.970; aPTT was analysed with an aPTT-SP (liquid) kit; fibrinogen activity was measured with the HemosIL Q.F.A Thrombin (Bovine) kit (Instrumentation Laboratory, Bedford MA, USA), based on the Clauss method; Factor VIII and IX concentrations were determined with the one-stage HemosIL Factor VIII and Factor IX deficient plasma kits; vWF levels were analysed with the automated, latex-enhanced immunoassay (HemosIL vonWillebrand Factor Antigen kit); Protein S concentration was measured using the HemosIL Protein S Activity Kit.
Statistical analyses
For each parameter, actual values and changes from baseline (relative to day 1 values) were described as mean with standard deviation (SD), median, interquartile range (IQR, 25th and 7 5th percentile), range (minimum, maximum), and 95% confidence intervals (CIs) assuming normality. Differences between the treatments were analysed using 95% CIs based on a Student’s t-test with unequal variances.
For RBCC parameters, the change from baseline was modelled using a Mixed Model for Repeated Measures (MMRM), with the baseline value of the parameter, timepoint, treatment group, and interaction effect of treatment by timepoint as fixed effects, and the RBCC pack as subject effect. Least Square (LS) means were calculated from this model by timepoint and treatment, and for the differences between the treatment by timepoint, with 95% CIs.
For plasma parameters, as there was only one post-treatment assessment timepoint, the MMRM methodology was replaced with an analysis of co-variance (ANCOVA) model for the change from baseline, with baseline value of the parameter and treatment arm as fixed effects. LS means estimates with 95% CIs were derived from this model.
RESULTS
Blood units
Twenty units of WB were collected in each group, with a mean volume of 505±7 mL in PRT and 504±7 mL in the Control group (volumes include CPD; Table I). Blood groups A, B, and O were equally represented in both groups (Table I). In the PRT group, the mean (±SD) RBCC volume was significantly lower (259±11 mL vs 283±16 mL; p<0.0001), while the plasma volume was on average higher (301±22 mL vs 279±29 mL; p=0.0036) than in the Control group (Table I).
Table I.
Baseline characteristics of whole blood (WB) units subjected to combined pathogen reduction and FAS (PRT) or only FAS (Control)
| Characteristics | PRT | Control |
|---|---|---|
| Units, n | 20 | 20 |
| WB volume (mL), mean±SD | 505.4±7.4 | 503.9±7.3 mL |
| Blood type (RhD antigen), n (%) | ||
| O (+) | 6 (30.0%) | 6 (30.0%) |
| A (+) | 10 (50.0%) | 10 (50.0%) |
| B (+) | 4 (20.0%) | 4 (20.0%) |
| RBCC volume (mL), mean±SD * | 259.4±10.9 | 283.0±16.0 |
| Plasma volume (mL), mean±SD ** | 301.0±21.8 | 279.4±29.3 |
p<0.0001,
p=0.0036;
Wilcoxon Rank Sum test for continuous variables and Cochran-Mantel-Haenszel test for categorical variables. FAS: fully automated separation; n, number of samples; PRT: pathogen reduction technology; RBCC: red blood cell concentrate; RhD antigen +: rhesus factor positive; SD: standard deviation.
Red blood cell concentrate parameters
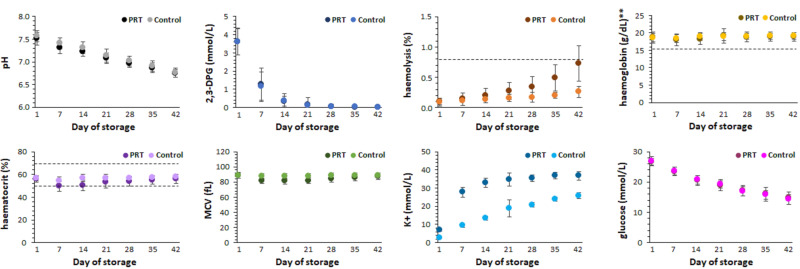
On day 1, PRT and Control units were comparable in terms of mean values for all RBCC parameters except extracellular potassium concentration (Figure 2 and Online Supplementary Table SI): pH (7.5±0.1 vs 7.6±0.1), 2,3-DPG concentration (3.6±0.8 mmol/L vs 3.6±0.7 mmol/L), haemolysis (0.1±0.1% vs 0.1±0.1%), haemoglobin (18.7±1.3 g/dL vs 18.6±1.6 g/dL; 48.2±4.0 g/unit and 52.4±5.4 g/unit), haematocrit (56.1±2.7% vs 56.5±1.9%), MCV (88.8±3.7 fL vs 88.3±3.4 fL), and glucose levels (26.6±1.1 mmol/L vs 26.9±1.5 mmol/L). Day 1 potassium levels were significantly higher in the PRT compared to the Control group (6.9±0.6 mmol/L vs 2.7±0.3 mmol/L; p<0.0001) (Figure 2 and Online Supplementary Table SI).
Figure 2.
Characteristics* of red blood cell concentrates (RBCC) obtained from pathogen-reduced (PRT) or untreated (Control) whole blood units subjected to fully automated separation
*Dashed lines represent thresholds as defined by European (EDQM)32 or Polish33 guidelines for acceptable quality of red blood cell concentrates. The threshold values are either upper (haemolysis) or lower (haemoglobin) cut-off values or range (haematocrit). **Threshold for haemoglobin (15.4g/dL) was manually calculated as a ratio of the EDQM-required concentration (40g/unit) and the average RBCC unit volume for the PRT group (259.4 mL or 2.6 dL). Error bars represent standard deviations from the mean; 2,3-DPG: 2,3-diphosphoglycerate concentration; fL: femtolitre; K+: potassium ion concentration; MCV: mean corpuscular volume; PRT: pathogen reduction technology.
The pH values, 2,3-DPG levels, and glucose concentration decreased over the observed storage time (Figure 2 and Online Supplementary Table SI). pH values were significantly lower in the PRT compared to the Control group only on days 7 and 14 (p=0.0349 and p=0.0172), but overall the differences were minimal. 2,3-DPG and glucose concentrations were comparable between both groups at each timepoint. The 2,3-DPG decrease was most pronounced between days 1 and 7.
The extracellular potassium concentration was significantly higher in the PRT compared to the Control group at all timepoints (p<0.0001). In addition, the increase in potassium levels over 42 days of RBCC storage followed a significantly different dynamic between groups, as reflected in change from baseline analyses (p<0.0100 for differences in change from baseline between groups at all timepoints) (Figure 2 and Online Supplementary Table SI). In the PRT group, the potassium concentration sharply increased between days 1 and 14, and gradually thereafter until day 42, while in the Control group it followed a linear increase over all timepoints.
Prominent differences between the PRT and Control groups were also observed in terms of the degree of haemolysis and haematocrit values (Figure 2 and Online Supplementary Table SI). Degree of haemolysis increased during storage in both PRT and Control RBCCs, as illustrated by the change from baseline within each group. Of note, haemolysis was more pronounced in the PRT compared to the Control group in terms of overall value after day 14 (p≤0.0157) and dynamics of increase (significant inter-group differences in change from baseline after day 21 [p≤0.0200]). The degree of haemolysis remained within the EDQM requirements (i.e., <0.8% of RBC mass in ≥ 90.0% of tested RBCCs) until day 35 for the PRT group (0.5±0.2% in the PRT group vs 0.2±0.1% in the Control group) and until day 42 of storage for the Control group RBCCs (0.7±0.3% in the PRT group vs 0.3±0.1% in the Control group). A more in-depth data analysis showed that two samples in the PRT group were “outliers”, with much higher haemolysis rates compared to the rest of the PRT RBCC samples, further increasing the shift in haemolysis in this group (Online Supplementary Figure S1). Haematocrit values were comparable between groups on day 42 (56.3±4.0% and 57.7±2.6%) but not at intermediate timepoints (days 7–35; p≤0.0204) (Figure 2 and Online Supplementary Table SI). The mean haemoglobin concentrations remained comparable between the groups at all timepoints and, for the most part, did not change during storage. On day 42, mean (±SD) haemoglobin content values were 48.7 (±3.1) g/unit in the PRT group and 53.9 (±4.7) g/unit in the Control group. Both haemoglobin and haematocrit were within the EDQM-defined quality requirements32 (i.e., ≥ 40 g/unit and 50.0–70.0% in ≥ 90.0% of tested RBCCs, respectively) at the end of the 42-day storage period.
There was a significant difference in MCV measured in RBCCs between PRT and Control samples at intermediate timepoints (days 7–35; p≤0.0403), although these differences were minimal.
Plasma parameters
Plasma variables were descriptively analysed in this study. Except for the vWF antigen, there was a significant difference in all plasma parameters between the PRT and Control groups both on day 1 and after 30 days of storage (Table II). PT (in seconds), aPTT, and INR were higher in the PRT group than the Control group (p<0.0001). Mean INR values were within the normal range for both groups (0.8–1.2)1. Concentrations of fibrinogen, Factor VIII, Factor IX, and Protein S were lower in the PRT group (p≤0.0102). At day 30, a subtle yet statistically significant change from baseline (relative to day 1 values) in the PRT but not the Control group was observed for PT (in seconds) (p<0.0100 vs p=0.1700), INR (p<0.0100 vs p=0.2500), aPTT (p=0.0200 vs p=0.3800), and Protein S concentration (p=0.0100 vs p=0.1200).
Table II.
Clotting variables and protein concentrations in plasma obtained from pathogen-reduced (PRT) or untreated (Control) whole blood units subjected to FAS
| Parameter, mean±SD | Group | Day | |
|---|---|---|---|
| 1 | 30 | ||
| PT (sec) | PRT | 13.7±1.4* | 13.9±0.9* |
| Control | 11.9±1.0 | 12.0±1.1 | |
| INR | PRT | 1.2±0.1* | 1.2±0.1* |
| Control | 1.0±0.1 | 1.1±0.1 | |
| aPTT (sec) | PRT | 40.1±2.9* | 41.2±4.5* |
| Control | 31.2±6.1 | 31.7±5.3 | |
| Fibrinogen (mg/dL) | PRT | 150.1±29.7* | 144.8±35.9* |
| Control | 216.0±40.0 | 212.5±54.4 | |
| Factor VIII (%) | PRT | 53.9±20.0* | 46.9±21.1* |
| Control | 94.2±32.4 | 89.8±42.5 | |
| Factor IX (%) | PRT | 70.0±13.7* | 65.7±13.7* |
| Control | 95.9±22.0 | 92.6±27.3 | |
| vWf (%) | PRT | 83.2±40.5 | 77.1±39.1 |
| Control | 101.5±38.4 | 95.7±42.7 | |
| Protein S (%) | PRT | 85.1±17.7* | 80.0±16.3* |
| Control | 100.1±17.3 | 102.5±18.7 | |
p<0.02;
Student’s t-test, comparison between PRT and Control groups. aPTT: activated partial thromboplastin time; FAS: fully automated separation; INR: International Normalised Ratio; PRT: pathogen reduction technology; PT: prothrombin time; SD: standard deviation; vWF: von Willebrand factor.
DISCUSSION
Red blood cell concentrates are the most commonly transfused blood components. Their quality, and consequently their therapeutic value, is influenced by multiple factors, including storage time, the additive solutions used, and methods of WB separation, leukoreduction, and pathogen reduction19,21. Storage of RBCCs negatively impacts the activity of several enzymes and results in the loss of different chemical compounds responsible for the maintenance of the cell metabolism, a phenomenon called “storage lesion”19,21.
The aim of this study was to assess to what extent the RF+UV PRT, followed by FAS, impacts the quality of blood components during storage. Currently, this PRT has a CE certification that allows for the transfusion of WB but not of the components derived after fractionation of the treated blood.
Randomisation of WB units was well balanced, since the relative proportions of A, B, and O blood groups were the same in both the PRT and the Control group. The differences in RBCC (lower) and plasma (higher) volumes between the PRT and the Control group were likely due to multiple transfers of the WB units, first from the FAS LR to the PRT illumination bag (addition of 35 mL of RF solution leading to increased plasma volume), and then to the new FAS LR bags before centrifugation.
The quality of RBCCs obtained from pathogen- and leuko-reduced and separated WB was evaluated through several variables, such as haemoglobin, haematocrit, 2,3-DPG concentration, potassium concentration, degree of haemolysis, and glucose concentration, some of which are also recommended by the EDQM32. Besides the degree of haemolysis, two other RBCC variables (haemoglobin content and haematocrit value) met the EDQM criteria for acceptable RBCC quality at the end of the 42-day storage period32.
The degree of haemolysis was higher in the PRT compared to the Control group RBCCs. It increased at a higher rate in RBCCs from the PRT group, exceeding the threshold recommended by the European (EDQM) and Polish guidelines (i.e., <0.8% haemolysis of RBC mass in ≥ 90.0% of tested RBCCs) after 35 days of storage. In contrast, Control RBCCs retained an acceptable degree of haemolysis throughout the entire 42-day storage period. An increased degree of haemolysis in RBCC fractions from PRT-treated versus untreated or gamma-irradiated WB had been previously reported after either 14 and 21 days of storage in additive solution 3 (AS-3) or SAGM solution24,26,28. The present study explored the use of PAGGSM as a storage solution, aiming to compare data to a recent study by Kumukova and colleagues that evaluated the quality of RBCCs derived from PRT-treated WB and stored in SAGM26. Kumukova and colleagues concluded that RBCCs obtained and stored in this solution should be stored no longer than 14 days, due to the observed higher degree of haemolysis26. The results of the present study suggest, however, that the use of PAGGSM and automated separation of PRT-treated WB resulted in RBCC components with a longer shelf-life than previously reported26. Higher haemolysis rates in RBCC units obtained after FAS processing of WB were initially reported, but recently published experience based on routine use of this technology attests rather to significantly lower degrees of haemolysis in RBCCs than with the conventional semi-automated process8,34–36. A more in-depth analysis of the current results revealed a subset of outliers within the PRT group with consistently and substantially increased haemolysis levels compared to the other donors. The accumulated damage to RBCCs in these specific samples might also be explained by donor-related factors, such as the health conditions as well as the genetic and metabolic profiles of these individuals19,37. Thus, the shelf-life of RBCCs derived from WB treated with PRT is potentially limited to 35 days, which is shorter than that of non-PRT-treated RBCCs.
It is noteworthy that extracellular potassium levels were significantly higher in the PRT than the Control group at all timepoints tested, in accordance with previous reports of elevated potassium concentrations in PRT-treated RBCC units. In the present study, the maximum value of potassium in the supernatant of stored RBCCs in the PRT group at day 42 was lower than that described in previous studies for gamma-irradiated or PRT-derived RBCCs stored in AS-3 or SAGM at day 14 or 2125,26,38. An elevated potassium concentration is likely to have a negligible clinical effect in patients25. Furthermore, the sharp increase in extracellular potassium levels with a gradually increasing degree of haemolysis is consistent with previous findings on RBCCs derived from PRT-treated WB26, and indicates that erythrocyte integrity is largely maintained over 35 days of storage despite potassium ion loss.
Significant differences in haematocrit values were transient between the analysed groups, while haemoglobin values remained comparable between the PRT and Control conditions at all timepoints tested, which is a positive outcome of this study, since haemoglobin is the key therapeutic constituent of the RBCC component. The observed haematocrit levels are in line with haematocrit trends reported previously for RBCC fractions derived from PRT-treated blood23, 26.
The pH values, glucose, and 2,3-DPG concentrations were mostly comparable between the PRT and Control groups and were within previously reported ranges24,25,27. There was a significant difference in MCV values between the groups, but these differences were small.
The data obtained on the plasma fractions were only descriptively analysed and cannot support a standard EDQM validation due to study design limitations (plasma information not collected before PRT procedure, as required by the EDQM32). More data are needed to conclusively evaluate plasma quality obtained after the combined PRT and FAS procedure. Except for the vWF values, there was a significant difference in all other variables between the PRT and Control plasma fractions. The relative differences in plasma factor concentrations between the PRT and Control groups remained similar over the 30-day storage period. Similar trends for plasma variable changes (prolonged PT and aPTT, decreased fibrinogen and Factor VIII concentrations, and comparable vWF concentrations) were described for plasma fractions obtained after RF+UV-based pathogen reduction compared to control treatments27,39. The differences in Protein S concentrations observed between the PRT and the Control group are also consistent with previous data showing lower Protein S concentrations in PRT-derived plasma fractions compared to untreated control27. Of note, addition of 35 mL RF solution to the WB before treatment also contributed to a certain dilution of plasma proteins, as observed elsewhere40.
The key strength of this study was its successful proof-of-principle approach, showing that the combination of PRT on WB followed by FAS can be used to successfully produce pathogen-reduced components of acceptable quality. As such, this protocol holds promise for the increased safety of individual blood components, although further development will be necessary to enable the process to be scaled up. Furthermore, the present work provides valuable data on possible storage solution effects on RBCC shelf-life; however, a direct comparison of the effect of the additive solution on shelf-life of RBCCs was not pursued to limit the number of WB units directed away from available transfusion stock for exploratory analyses. For the moment, comparisons can only be made with historical data from a study performed with semi-automated fractionation of WB treated with RF+UV light and employing SAGM for storage of PRT-treated RBCCs26. While PAGGSM belongs to the latest generation of additive solutions, further research is necessary to determine the optimal storage for blood components derived from PRT-treated blood. The present study reported a longer achievable RBCC storage time (35 days) compared to that previously reported for SAGM26. The longer RBCC shelf-life of 35 days is consistent with other reports on PAGGSM20,21 and is important given the universal interest in keeping a sustainable inventory of the RBCCs. A longer shelf-life means fewer blood collections and/or donors are needed, which may be crucial given that donor and patient populations are ageing.
Apart from the above-mentioned suboptimal design (lack of plasma information prior to the PRT procedure), another limitation was the lack of data on adenosine-triphosphate (ATP) levels in RBCCs. The ATP data were not collected due to the limited operational capacity and lack of available reagents; instead 2,3-DPG was measured. Finally, the in vitro character and the low number of samples limited the analytical power of the study.
CONCLUSIONS
Based on the present in vitro results, RBCCs obtained from pathogen-reduced WB subjected to fully automated separation could be stored for up to 35 days in PAGGSM additive solution. The quality of plasma fractions obtained from the same WB units is consistent with previous publications. The benefit of the approach presented here and blood component quality are to be corroborated in future studies.
Supplementary Information
ACKNOWLEDGEMENTS
The Authors would like to thank Modis for providing writing (Irena Zurnic Bönisch) and manuscript co-ordination (Sophie Timmery) assistance, Erik Spaepen for statistical analyses, and BB Łódź Edyta Wcisło for directing the study during its development.
Footnotes
AUTHORSHIP CONTRIBUTIONS
MB, PS, AR, and AW: performed the research study. MB, AG, SS, MC and EL: designed the research study. AR: contributed to essential reagents and tools. MB, AG, MC, and EL: analysed and interpretated the data. All Authors reviewed and approved the final draft and its submission in the journal.
DISCLOSURE OF CONFLICTS OF INTEREST
SS and MC are employees of Terumo Blood and Cell Technologies. The other Authors declare no conflicts of interest.
Trademark statement
Mirasol® PRT System and Reveos® Automated Blood Processing System are trademarks of Terumo Blood and Cell Technologies, USA. The Mirasol® PRT system is available for sale in the European Union (EU) and in non-EU countries that accept the CE mark. The Mirasol® PRT system currently is not cleared for sale in USA. The Reveos® Automated Blood Processing System is not cleared for sale in USA. It is available in select markets.
FUNDING AND RESOURCES
Terumo Blood and Cell Technologies funded the equipment, disposables and reagents used in this study. In addition, the company financed the statistical analyses, writing and editorial support, and was involved in the study design.
REFERENCES
- 1.Marschner S, Goodrich R. Pathogen reduction technology treatment of platelets, plasma and whole blood using riboflavin and UV light. Transfus Med Hemother. 2011;38:8–18. doi: 10.1159/000324160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Picker SM. Current methods for the reduction of blood-borne pathogens: a comprehensive literature review. Blood Transfus. 2013;11:343–8. doi: 10.2450/2013.0218-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hardwick J. Blood processing. ISBT Science Series. 2008;3:148–76. [Google Scholar]
- 4.McDonald CP. Interventions implemented to reduce the risk of transmission of bacteria by transfusion in the English National Blood Service. Transfus Med Hemother. 2011;38:255–58. doi: 10.1159/000330474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dodd RY. Emerging pathogens and their implications for the blood supply and transfusion transmitted infections. Br J Haematol. 2012;159:135–42. doi: 10.1111/bjh.12031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Allain J-P, Owusu-Ofori AK, Assennato SM, et al. Effect of Plasmodium inactivation in whole blood on the incidence of blood transfusion-transmitted malaria in endemic regions: the African Investigation of the Mirasol System (AIMS) randomised controlled trial. Lancet. 2016;387:1753–61. doi: 10.1016/S0140-6736(16)00581-X. [DOI] [PubMed] [Google Scholar]
- 7.Malvaux N, Schuhmacher A, Defraigne F, et al. Remodelling whole blood processing through automation and pathogen reduction technology at the Luxembourg Red Cross. Transfus Apher Sci. 2021:103195. doi: 10.1016/j.transci.2021.103195. [DOI] [PubMed] [Google Scholar]
- 8.Pérez Aliaga AI, Labata G, Aranda A, et al. Improvement of blood processing and safety by automation and pathogen reduction technology. Transfus Med Hemother. 2021;48:290–7. doi: 10.1159/000516696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Seltsam A. Pathogen inactivation of cellular blood products-an additional safety layer in transfusion medicine. Front Med (Lausanne) 2017;4:219. doi: 10.3389/fmed.2017.00219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mundt JM, Rouse L, Van den Bossche J, Goodrich RP. Chemical and biological mechanisms of pathogen reduction technologies. Photochem Photobiol. 2014;90:957–64. doi: 10.1111/php.12311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Goodrich RP, Edrich RA, Li J, Seghatchian J. The Mirasol PRT system for pathogen reduction of platelets and plasma: an overview of current status and future trends. Transfus Apher Sci. 2006;35:5–17. doi: 10.1016/j.transci.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 12.Seghatchian J, de Sousa G. Pathogen-reduction systems for blood components: the current position and future trends. Transfus Apher Sci. 2006;35:189–96. doi: 10.1016/j.transci.2006.10.002. [DOI] [PubMed] [Google Scholar]
- 13.Garban F, Guyard A, Labussiere H, et al. Comparison of the hemostatic efficacy of pathogen-reduced platelets vs untreated platelets in patients with thrombocytopenia and malignant hematologic diseases: a randomized clinical trial. JAMA Oncol. 2018;4:468–75. doi: 10.1001/jamaoncol.2017.5123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McCullough J, Vesole DH, Benjamin RJ, et al. Therapeutic efficacy and safety of platelets treated with a photochemical process for pathogen inactivation: the SPRINT Trial. Blood. 2004;104:1534–41. doi: 10.1182/blood-2003-12-4443. [DOI] [PubMed] [Google Scholar]
- 15.Mirasol Clinical Evaluation Study Group. . A randomized controlled clinical trial evaluating the performance and safety of platelets treated with MIRASOL pathogen reduction technology. Transfusion. 2010;50:2362–75. doi: 10.1111/j.1537-2995.2010.02694.x. [DOI] [PubMed] [Google Scholar]
- 16.Rebulla P, Vaglio S, Beccaria F, et al. Clinical effectiveness of platelets in additive solution treated with two commercial pathogen-reduction technologies. Transfusion. 2017;57:1171–83. doi: 10.1111/trf.14042. [DOI] [PubMed] [Google Scholar]
- 17.van der Meer PF, Ypma PF, van Geloven N, et al. Hemostatic efficacy of pathogen-inactivated vs untreated platelets: a randomized controlled trial. Blood. 2018;132:223–31. doi: 10.1182/blood-2018-02-831289. [DOI] [PubMed] [Google Scholar]
- 18.Goodrich RP, Doane S, Reddy HL. Design and development of a method for the reduction of infectious pathogen load and inactivation of white blood cells in whole blood products. Biologicals. 2010;38:20–30. doi: 10.1016/j.biologicals.2009.10.016. [DOI] [PubMed] [Google Scholar]
- 19.D’Alessandro A, Zimring JC, Busch M. Chronological storage age and metabolic age of stored red blood cells: are they the same? Transfusion. 2019;59:1620–23. doi: 10.1111/trf.15248. [DOI] [PubMed] [Google Scholar]
- 20.de Korte D. New additive solutions for red cells. ISBT Science Series. 2016;11:165–70. [Google Scholar]
- 21.Lagerberg JW, Korsten H, van der Meer PF, de Korte D. Prevention of red cell storage lesion: a comparison of five different additive solutions. Blood Transfus. 2017;15:456–62. doi: 10.2450/2017.0371-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lozano M, Badawi M. Indications for transfusion of blood components. ISBT Science Series. 2020;15:320–30. [Google Scholar]
- 23.Cancelas JA, Rugg N, Fletcher D, et al. In vivo viability of stored red blood cells derived from riboflavin plus ultraviolet light-treated whole blood. Transfusion. 2011;51:1460–8. doi: 10.1111/j.1537-2995.2010.03027.x. [DOI] [PubMed] [Google Scholar]
- 24.Cancelas JA, Slichter SJ, Rugg N, et al. Red blood cells derived from whole blood treated with riboflavin and ultraviolet light maintain adequate survival in vivo after 21 days of storage. Transfusion. 2017;57:1218–25. doi: 10.1111/trf.14084. [DOI] [PubMed] [Google Scholar]
- 25.Dimberg LY, Doane SK, Yonemura S, et al. Red blood cells derived from whole blood treated with riboflavin and UV Light maintain adequate cell quality through 21 days of storage. Transfus Med Hemother. 2019;46:240–47. doi: 10.1159/000495257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kumukova I, Trakhtman P, Starostin N, et al. Quality assessment of red blood cell suspensions derived from pathogen-reduced whole blood. Vox Sang. 2020;116:547–56. doi: 10.1111/vox.13039. [DOI] [PubMed] [Google Scholar]
- 27.Schubert P, Culibrk B, Karwal S, et al. Whole blood treated with riboflavin and ultraviolet light: quality assessment of all blood components produced by the buffy coat method. Transfusion. 2015;55:815–23. doi: 10.1111/trf.12895. [DOI] [PubMed] [Google Scholar]
- 28.Trakhtman P, Kumukova I, Starostin N, et al. The pathogen-reduced red blood cell suspension: single centre study of clinical safety and efficacy in children with oncological and haematological diseases. Vox Sang. 2019;114:223–31. doi: 10.1111/vox.12757. [DOI] [PubMed] [Google Scholar]
- 29.Janetzko K, Kluter H, van Waeg G, Eichler H. Fully automated processing of buffy-coat-derived pooled platelet concentrates. Transfusion. 2004;44:1052–8. doi: 10.1111/j.1537-2995.2004.03296.x. [DOI] [PubMed] [Google Scholar]
- 30.Lotens A, Najdovski T, Cellier N, et al. New approach to ‘top-and-bottom’ whole blood separation using the multiunit TACSI WB system: quality of blood components. Vox Sang. 2014;107:261–8. doi: 10.1111/vox.12159. [DOI] [PubMed] [Google Scholar]
- 31.Vetlesen A, Mirlashari MR, Ezligini F, Kjeldsen-Kragh J. Evaluation of platelet activation and cytokine release during storage of platelet concentrates processed from buffy coats either manually or by the automated OrbiSac system. Transfusion. 2007;47:126–32. doi: 10.1111/j.1537-2995.2007.01075.x. [DOI] [PubMed] [Google Scholar]
- 32.European Directorate for the Quality of Medicines and HealthCare (EDQM) Guide to the preparation, use and quality assurance of blood components. 20 ed. Strasbourg, France: 2020. [Google Scholar]
- 33.Polish Ministry of Health. [Accessed on 7/09/2021]. Available at: http://isap.sejm.gov.pl/isap.nsf/download.xsp/WDU20170000646/O/D20170646.pdf, [In Polish.]
- 34.Hansen AL, Kurach JD, Turner TR, et al. The effect of processing method on the in vitro characteristics of red blood cell products. Vox Sang. 2015;108:350–8. doi: 10.1111/vox.12233. [DOI] [PubMed] [Google Scholar]
- 35.Lagerberg JW, Salado-Jimena JA, Löf H, et al. Evaluation of the quality of blood components obtained after automated separation of whole blood by a new multiunit processor. Transfusion. 2013;53:1798–807. doi: 10.1111/trf.12010. [DOI] [PubMed] [Google Scholar]
- 36.Shih AW, Apelseth TO, Cardigan R, et al. Not all red cell concentrate units are equivalent: international survey of processing and in vitro quality data. Vox Sang. 2019;114:783–94. doi: 10.1111/vox.12836. [DOI] [PubMed] [Google Scholar]
- 37.Sparrow RL. Red blood cell components: time to revisit the sources of variability. Blood Transfus. 2017;15:116–25. doi: 10.2450/2017.0326-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Maia GAS, Cortes VF, de A, Ribeiro RIM, et al. Could Na, K-ATPase play a role in potassium leakage from irradiated erythrocytes? Clin Chim Acta. 2014;433:58–61. doi: 10.1016/j.cca.2014.02.025. [DOI] [PubMed] [Google Scholar]
- 39.Pidcoke HF, McFaul SJ, Ramasubramanian AK, et al. Primary hemostatic capacity of whole blood: a comprehensive analysis of pathogen reduction and refrigeration effects over time. Transfusion. 2013;53(Suppl 1):137S–49S. doi: 10.1111/trf.12048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Larrea L, Ortiz-de-Salazar MI, Martinez P, Roig R. Quantitative analysis of plasma proteins in whole blood-derived fresh frozen plasma prepared with three pathogen reduction technologies. Transfus Apher Sci. 2015;52:305–10. doi: 10.1016/j.transci.2015.01.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.