Abstract
Background
Children with underlying kidney diseases display a mild course of SARS-CoV-2 infection, but they only accounted for a minority of cases until the spread of the Omicron variant. Nonetheless, idiopathic nephrotic syndrome (INS) has been advocated as a predictor of worse outcome.
Methods
We investigated the spread, severity, and risk of relapse related to SARS-CoV-2 infection among children with INS. The incidence and characteristics of SARS-CoV-2 infections, immunosuppression, and vaccination status were retrospectively collected from the beginning of the pandemic to May 31, 2022.
Results
We enrolled 176 patients (73 females, median age 10.22 years); 28 had a steroid-resistant disease, and 108 (61.4%) were on immunosuppressive therapy. Sixty-one (34.7%) patients reported a SARS-CoV-2 infection, with incidence peaking between December 2021 and January 2022. No hospitalization or deaths were reported, and symptoms were absent or mild. The rate of SARS-CoV-2 infection was similar in children with and without immunosuppression (33.8% vs 35.2%; p = 0.85). None of the 38 immunosuppressed patients discontinued the therapy, but they had a longer time to negativization (13.31 vs. 10.04 days; p = 0.03). Proteinuria was detected in 7 patients, but only one had a relapse requiring steroid therapy, with prompt remission and a mild course.
Conclusions
After the spread of the Omicron variant, the rate of SARS-CoV-2 infection in children with INS was much higher than previously reported. In this large cohort, symptoms were mild, even in immunosuppressed patients and those with proteinuria. During the infection, transient proteinuria was common with a low rate of relapses.
Graphical abstract
A higher resolution version of the Graphical abstract is available as Supplementary information
Supplementary Information
The online version contains supplementary material available at 10.1007/s00467-022-05736-6.
Keywords: Idiopathic nephrotic syndrome, Children, SARS-CoV-2, COVID-19, Relapse, Vaccines, Proteinuria
Introduction
Idiopathic nephrotic syndrome (INS) is the most common glomerular disease in children, and it is usually characterized by a relapsing–remitting course, often requiring immunosuppressive therapy [1]. Infectious diseases are one of the major complications, with viral upper respiratory tract infections being the most common trigger for relapses [2].
The SARS-CoV2 pandemic rapidly spread from China at the end of 2019, causing more than 500 million cases and 6 million deaths worldwide, as of May 2022 [3].
Children display a milder clinical course as compared to adults, with a rate of asymptomatic infections approaching 50% [4]. A benign SARS-CoV-2 infection has been reported also in children with underlying kidney diseases, including INS [5–7]. According to most of the literature, during a SARS-CoV-2 infection, there is no need to preemptively hospitalize or discontinue immunosuppression in children with INS [8]. However, SARS-CoV-2-related disease onsets and relapses have been reported [8], and two studies have described a significant morbidity and mortality, particularly during relapses [9, 10]. Children represented only a minority of the reported SARS-CoV-2 cases until the spread of the Omicron variant in November 2021 [11], which caused a marked increase of cases, specifically in the pediatric population [12]. In Italy, new daily cases increased from 40,000 in November 2020 to a peak of 180,000 in January 2022, with pediatric cases rising in the same period from 1–5% up to 17.7% [13].
In light of the recent increase of SARS-CoV-2 cases in children and relentless spreading of the infection, we aimed to investigate the diffusion and severity of SARS-CoV-2 infection in pediatric patients with INS.
Methods
We performed a retrospective monocentric observational study to assess the incidence and severity of SARS-CoV-2 infection among pediatric patients affected by INS and to explore the risk of relapse associated with the infection.
The study was conducted at the Pediatric Nephrology, Dialysis and Transplant Unit of Milan, the reference center for Pediatric Nephrology in Lombardy, the most populous region of Italy.
Inclusion criteria were as follows: (1) clinical diagnosis of INS; (2) age between 1 and 20 years; (3) a follow-up of at least 6 months at the time of data collection. Data were retrospectively collected from June 1, 2022 to June 15, 2022 by a phone-based questionnaire performed by the same author (F.V.) and submitted to caregivers; patients were directly interviewed only if > 18 years old. Families were asked to report any previous SARS-CoV-2 infection documented either by a molecular or antigenic nasal swab from the beginning of the pandemic until May 31, 2022. SARS-CoV-2–related symptoms (fever, upper respiratory or gastrointestinal involvement, rash), the duration of infection, the need for hospitalization or respiratory support, and the occurrence of relapse or self-limiting proteinuria were collected. Anonymized patients’ demographics, disease characteristics, ongoing immunosuppressive treatment, and vaccination status were also recorded.
The incidence and severity of SARS-CoV-2 infection and the related relapse rates were correlated to ongoing immunosuppressive treatment and vaccination status. The risk of SARS-CoV-2 related relapse was assessed only in patients for whom a previous remission had been documented.
The study was approved by the local ethics committee, and informed consent was obtained from patients or legal guardians.
Definitions
INS was defined according to the international guidelines and classified as infrequent relapsing nephrotic syndrome (IRNS), frequent-relapsing nephrotic syndrome (FRNS), steroid-dependent nephrotic syndrome (SDNS), or steroid-resistant nephrotic syndrome (SRNS) [1, 14]. Relapse was defined as a persistent proteinuria (urinary dipstick ≥ 3 + or urinary protein/creatinine ratio (uPr/uCr) > 2 g/g) for 3 consecutive days requiring steroid therapy [15]. Self-limiting proteinuria was defined as a positive urinary dipstick (≥ 2 +) with spontaneous resolution within 7 days. Short-term steroid prophylaxis was defined as the prophylactic use of a low prednisone dose (0.5 mg/kg) for up to 5 days, during an upper respiratory infection. SARS-CoV-2 infection was defined as a positive molecular or antigenic nasal swab. Duration of infection was defined as the time elapsed until a negative nasal swab, as required to end the isolation period in Italy during the entire study period.
Statistical analysis
Categorical data are expressed as proportions. Continuous data are expressed as mean (SD) when normally distributed, otherwise as median (interquartile range [IQR]). Categorical variables were compared by using the χ2 test, and continuous variables were compared using the t-test (normally distributed) or the Mann–Whitney U test (not normally distributed). For all the statistical analyses, p-values < 0.05 were considered significant.
All statistical analyses were performed using the open source software R (R Core Team, 2014. R: A language and environment for statistical computing, R Foundation for Statistical Computing, Vienna, Austria).
Results
From the review of the digital medical records, 215 patients affected by INS were identified. Seventeen young adults and 6 patients with a clinical follow-up < 6 months were excluded, while 16 patients could not be reached for the analysis. A total of 176 children (73 females) with a median age of 10.22 years (IQR, 6.71–14.01) were enrolled in the study (Fig. 1).
Fig. 1.
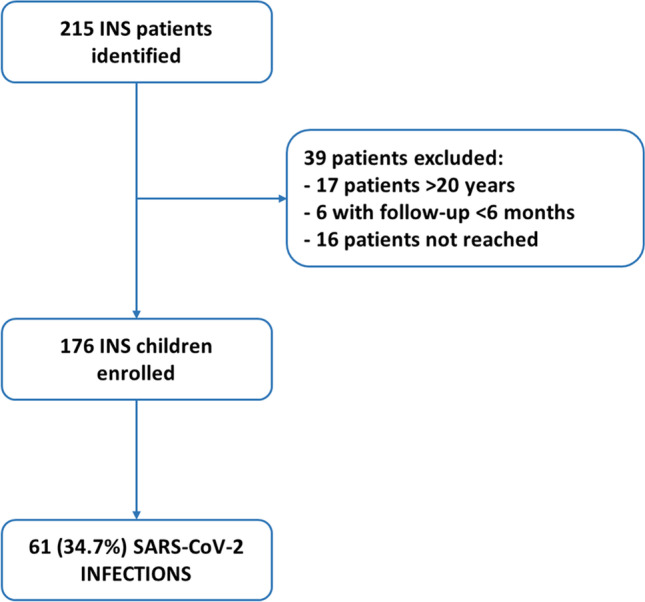
Study flow diagram. The figure summarizes the number of children and young adults identified, excluded and enrolled in the study. INS, idiopathic nephrotic syndrome
Forty-one enrolled patients (23.3%) were affected by IRNS, 107 (60.8%) by SDNS/FRNS, and 28 (15.9%) by SRNS. One hundred and eight (61.4%) patients were on immunosuppressive therapy: among them, 85 patients were receiving a single drug, while 23 patients were treated with a combined therapy. Among SRNS patients, 10 (35.7%) had a persistent proteinuria, 7 for a multidrug resistant INS (MDR), and 3 for a genetic form.
Eighty-two (46.6%) patients were vaccinated against SARS-CoV-2 with at least 2 doses of an mRNA vaccine, 5 of them after the infection, 7 (4%) patients had received the first dose only, and 87 were not vaccinated for parental choice (n = 61) or because they were below the age limit (n = 26). The main characteristics of the enrolled population are summarized in Table 1.
Table 1.
Clinical characteristics of enrolled children
| All patients n = 176 | SARS-CoV-2 Infection n = 61 | No infection n = 115 | p-value | |
|---|---|---|---|---|
| Age at enrollment in years, median (IQR) | 10.22 (IQR, 6.71–14.01) | 10.5 (IQR, 6.46–14.10) | 10.09 (IQR, 7.04–13.95) | 0.67 |
| Gender, N (%) | 0.58 | |||
| Males | 103 (58.5%) | 34 (55.7%) | 69 (60%) | |
| Females | 73 (41.5%) | 27 (44.3%) | 46 (40%) | |
| INS subtype, N (%) | 0.9 | |||
| IRNS | 41 (23.3%) | 13 (21.3%) | 28 (24.3%) | |
| SDNS | 107 (60.8%) | 38 (62.3%) | 69 (60%) | |
| SRNS | 28 (15.9%) | 10 (16.4%) | 18 (15.7%) | |
| Immunosuppressive treatment, N (%) | 0.85 | |||
| No | 68 (38.6%) | 23 (37.7%) | 45 (39%) | |
| Yes | 108 (61.4%) | 38 (62.3%) | 70 (61%) | |
| MMF | 96 | 31 | 65 | |
| CNI | 26 | 14 | 12 | |
| Steroid | 5 | 3 | 2 | |
| Rituximab | 8 | 4 | 4 | |
| Monotherapy | 85 | 24 | 61 | |
| Combined therapy | 23 | 14 | 9 | |
| SARS-CoV-2 vaccination, N (%) | 0.067 | |||
| No | 92 (52.3%) * | 37 (60.7%)* | 55 (47.8%) | |
| At least 2 doses | 77 (43.8%) | 20 (32.8%) | 57 (49.6%) | |
| Incomplete schedule | 7 (3.9%) | 4 (6.5%) | 3 (2.6%) | |
| Ethnicity | 0.02 | |||
| Caucasian | 122 (69.3%) | 50 (82%) | 72 (62.6%) | |
| African | 22 (12.6%) | 4 (6.6%) | 18 (15.7%) | |
| Hispanic | 9 (5.1%) | 4 (6.6%) | 5 (4.3%) | |
| Asian | 23 (13%) | 3 (4.8%) | 20 (17.4%) |
IRNS infrequent relapse nephrotic syndrome, SDNS steroid-dependent nephrotic syndrome, SRNS steroid-resistant nephrotic syndrome, MMF mycophenolate mofetil, CNI calcineurin inhibitors
*5 patients were vaccinated after the infection
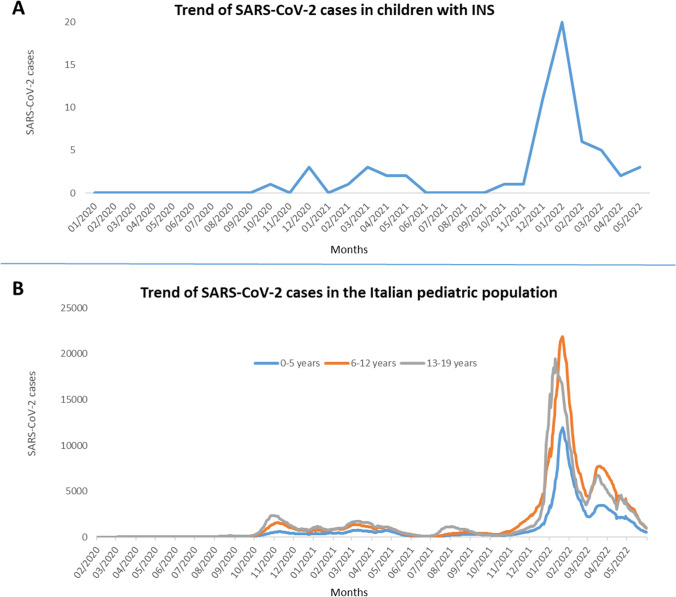
A total of 61/176 (34.7%) patients reported a previous SARS-CoV-2 infection documented by nasal swab at a median age of 10 (IQR, 6.08–13.22) years: 4 patients in 2020, 21 in 2021, and 36 in the first 5 months of 2022. The trend of SARS-CoV-2 infections is summarized in Fig. 2A. One patient reported a previous infection documented by seroconversion, and 1 patient developed mild symptoms suggestive for SARS-COV-2 after a close positive contact, but no diagnostic test was performed. These 2 patients were not included in the final analysis. The cumulative rate of SARS-CoV-2 infections was comparable to that reported in the healthy age-matched Italian population (34.7% vs. 35.8%; p = 0.82) with a similar trend over time (Fig. 2) [13, 16].
Fig. 2.
The figure shows the comparable trend of SARS-CoV-2 infections over the time in our population of children with INS (A) and in the general Italian pediatric population (B) [13, 16]. INS, idiopathic nephrotic syndrome
Table 2 summarizes the main characteristics of patients with SARS-CoV-2 infection. Among the 61 SARS-CoV-2-infected patients, 13 were affected by IRNS, 38 by SDNS/FRNS, and 10 by SRNS. Fifty-seven children were in remission at the time of infection, while 4 patients affected by either a MDR or genetic SRNS had a persistent proteinuria. The characteristics of the population with SARS-CoV-2 infection were comparable to those of non-infected children, except for the higher proportion of Caucasian patients (Table 2).
Table 2.
Clinical characteristics of INS children infected by SARS-CoV-2
| SARS-CoV2 infected patients (n = 61) | |
|---|---|
| Remission at the time of infection | |
| Yes | 57 (93.5%) |
| No | 4 (6.5%) |
| Immunosuppressive therapy | |
| Yes | 38 (62.3%) |
| No | 23 (37.7%) |
| Symptoms, N (%) | |
| Yes | 49 (80.3%) |
| No | 12 (19.3%) |
| Reported symptoms, N | |
| Fever | 28 |
| Flu-like symptoms | 10 |
| Rhinitis | 30 |
| Gastrointestinal symptoms | 4 |
| Skin rash | 0 |
| MIS-C | 0 |
| Dyspnea or respiratory failure | 0 |
| Hospitalization | 0 |
| Proteinuria | |
| Relapse | 1 |
| Transient proteinuria | 6 |
| Infection duration in days, mean (SD) | 12 (6.43) |
| Immunosuppressed | 13.31 |
| Not immunosuppressed | 10.04 |
MIS-C multisystem inflammatory syndrome in children
All patients had an indolent course of the infection: 12/61 (19.7%) were completely asymptomatic, while 49/61 (80.3%) had mild symptoms only. Upper respiratory tract symptoms and fever were the most prevalent complaints, reported respectively in 32/61 (52.4%) and 33/61 (54%) patients, while only 7 (11.4%) patients had gastrointestinal involvement. In no case was specific treatment, respiratory support, or hospitalization for SARS-CoV-2 infection required. The clinical course of the infection was mild also in the 4 patients with proteinuria affected by MDR or genetic SRNS and in the one experiencing a concomitant relapse (see below).
One patient affected by an IRNS and not undergoing any kind of chronic immunosuppression experienced a reinfection 7 months after the first episode: the first SARS-CoV-2 infection was characterized by flu-like symptoms, while the second had a completely asymptomatic course.
The rate of SARS-CoV-2 infection was similar in immunosuppressed and not immunosuppressed patients (33.8% vs. 35.2%; p = 0.85). Of all 61 children and young adults with SARS-CoV-2 infection, 38 (62.3%) were on immunosuppressive drugs at the time of SARS-CoV-2 infection, and 14 (22.9%) were treated with a combination therapy. Immunosuppression was not associated with a more severe course of the disease, 10 were completely asymptomatic, and 28 were paucisymptomatic, with no hospitalizations. No patient needed a modification or a temporary withdrawal of the ongoing immunosuppressive therapy during the infection. SARS-CoV-2 infections had a mean (SD) duration of 12 days (6.43) in the entire cohort, and it was slightly but significantly longer in patients receiving immunosuppressive drugs (13.31 vs. 10.04 days; p = 0.03).
Among the 77 patients previously vaccinated with at least 2 doses, 20 reported a SARS-CoV-2 infection after a median time of 73 days from the last dose. The rate of SARS-CoV-2 infection was lower but not significantly different in vaccinated vs. non-vaccinated children (25.9% vs. 40.2%; p = 0.067), with a similar time required to negativization (12.35 (7.36) vs. 12.78 (5.28) days; p = 0.82). All had an indolent course, and the rate of symptomatic patients was similar in vaccinated and non-vaccinated patients (85% vs. 78%; p = 0.41).
Among the 57 patients in clinical remission, proteinuria was detected in 7 (12.3%) by urine dipstick during the infection, but only one had a clinical relapse requiring prednisone therapy, with a prompt remission in 7 days. As mentioned before, also in this patient, the clinical course of SARS-CoV-2 infection was mild, and ongoing immunosuppressive therapy was not discontinued. In the remaining 6 patients, the proteinuria was transient and underwent spontaneous remission within the resolution of the infection. In two cases, a short-term low-dose steroid prophylaxis was performed. The risk of developing proteinuria during SARS-CoV-2 infection was not significantly different in children with or without immunosuppressive therapy (9% vs. 25%; p = 0.19) and in patients vaccinated or not (10% vs. 15.6%; p = 0.63).
Finally, from March 2020, 4 patients with a median age of 2.5 years had their INS onset during a SARS-CoV-2 infection. All of them had a typical course, with no complications and a rapid remission after steroid therapy.
Discussion
Here, we report the outcome and the detailed characteristics of the largest cohort of children and young adults with INS infected by SARS-CoV-2, during the whole course of the pandemic up to May 2022. Indeed, all previous studies were performed before the emergence of the Omicron variant, which is responsible for most of the pediatric SARS-CoV-2 cases [12].
While initial studies reported a limited number of cases and a mild course of the infection in this population [8], contradictory results have emerged from two recent reports, describing a worse outcome including 9 deaths [9, 10].
In a population of 176 children with INS, we identified a total of 61 SARS-CoV-2 infections documented by a swab test, with a rate of infection of 34.7%, much higher than previously reported [6, 7]. In the early stages of the pandemic, two Italian nationwide studies documented a low prevalence of SARS-CoV-2 infection in children with kidney diseases, with an infection rate of 0.19% and 2% by swab test or serology, respectively [6, 7]. In our population, the incidence had a growing trend through the 2 years of the pandemic, with 4 cases occurring between October and December 2020, 21 throughout 2021 and 36 between January and May 2022. This is in line with the spreading of the infection in the general pediatric population, with an incidence of pediatric cases 6–8 times higher during the Omicron-predominant period [12].
In our study, all patients had a benign course of the infection with no severe cases, no hospitalizations or deaths, even in patients with a relapse or new onset, or those with persistent proteinuria. This is consistent with what has been previously reported in most of the available literature. In our systematic review, the 43 cases of SARS-CoV-2 infections in children with INS, reported in high income countries up to August 2021, were characterized by a mild disease course, with low need of respiratory support and no deaths [8]. A retrospective study performed by Weinbrand and colleagues on children with kidney diseases including INS, in the relatively low-income area of Jerusalem, has confirmed the benign course of SARS-CoV-2 infection also in this subset of patients, with 37% asymptomatic infections and no severe cases [17]. Conversely, Krishnasamy et al. reported higher rates of severe COVID-19 in pediatric INS patients from the area of New Delhi, with 15% of patients requiring invasive ventilation and a mortality rate of 4.5%. Relapse was a risk factor for severity [9]. Moreover, in a Brazilian study, Vasconcelos and colleagues described a mortality rate of 28% in children with INS, hospitalized during the first year of the pandemic. Geographical origin was associated with the highest risk of death, overcoming any other clinical characteristic and supporting the hypothesis that socio-economic factors are extremely relevant in determining the natural history of SARS-CoV-2 infection in a vulnerable population, such as children with INS [10]. We were not able to systematically assess socio-economic status, but our population had a heterogeneous background and ethnicity. Anyway, the study was performed in a high-income country with universal health care. In addition to socio-economic factors, a different severity of the Omicron variant could have played a role in the better outcome of our patients. However, the completely benign course of the infection during all waves of pandemic and the large size of our cohort allow us to support the favorable evolution of SARS-CoV-2 infection in children and young adults with INS, reinforcing our published recommendations on the management of these patients indicating no need for additional restrictions, therapy discontinuation, or pre-emptive hospitalization [8].
Among 57 children who were in remission at the time of the infection, only 1 (1.7%) developed a clinically relevant relapse requiring steroid therapy, while 6 additional patients had a self-limiting proteinuria, which resolved with no steroid treatment. Different case reports have highlighted the association between SARS-CoV-2 infection and INS relapse [8], but this is the first study that could assess the real rate of relapse in a cohort of SARS-CoV-2-infected patients. According to our data, while a correlation between SARS-CoV-2 infection and relapse is possible, the rate seems to be low.
Immunosuppressed patients in our cohort (n = 108) were not at higher risk of developing SARS-CoV-2 infection; as compared to untreated patients, the infections were not associated with a more severe course and did not require the discontinuation of the ongoing immunosuppressive therapy. This finding is consistent with the global survey performed by Marlais and colleagues on SARS-CoV-2-infected children receiving immunosuppressive therapies for kidney diseases (30/113 affected by INS) until July 2020, describing a mild disease course in the majority of patients, with no association between the number of immunosuppressive medications and COVID-19 severity. This is also in line with a more recent review of the literature carried out by Connelly et al. on the impact of SARS-CoV-2 infection in pediatric patients undergoing immunosuppression for variable diseases: among them, immunosuppressed patients for kidney disease reported low rates of severe complications, while increased morbidity and mortality was documented in oncologic and rheumatologic patients, mostly due to decreased access to care [18]. Nonetheless, in our cohort, the infection had a slightly but significantly longer course in children undergoing immunosuppressive therapy (13 vs. 10 days). This is consistent with previous reports in immunocompromised adults and children, and a matter of concern for the emergence of more virulent variants [19, 20].
Despite a good compliance to the vaccination campaign, in our cohort, the rate of SARS-CoV-2 infection was lower but not significantly different in vaccinated children. Among the 77 patients who had received at least 2 doses, 20 (26%) reported a SARS-CoV-2 infection after a median time of 73 days from the last dose. Our study was neither planned nor powered to assess the impact of vaccination on SARS-CoV-2 infection in children with INS; however, two main factors may justify this finding. Firstly, a recent systematic review of the literature in the general population by Ssentongo et al. [21] has demonstrated how vaccine effectiveness against all SARS-CoV-2 infections declined from 83% in the first month after completion of the original vaccination series to 22% at 5 months, while effectiveness against severe COVID-19 remained as high as 90% at 5 months or longer at all ages. Moreover, SARS-CoV-2 infections in our cohort peaked between December 2021 and January 2022. A reduced effectiveness of mRNA vaccines against the Omicron variant, particularly in children, has been reported by different clinical and biological studies [22, 23]. The incidence of SARS-CoV-2 infections in our cohort could therefore be explained by both the incomplete and time-dependent effectiveness of vaccines in protecting against asymptomatic or mild infections and by the concomitant spread of higher virulence variants, namely, the Omicron variant, which increasingly hit the pediatric population. Further evidence is needed about the effectiveness of currently available vaccines against newly emerging SARS-CoV-2 variants; however, the high safety profile and efficacy against severe forms of SARS-CoV-2 [24] still justify large-scale vaccination in the pediatric population, including children with INS.
This study has some limitations. Firstly, the retrospective and monocentric nature of the study may have introduced unknown bias. Moreover, data accuracy could have been affected by self-reporting. Finally, the relatively small numbers may have not been able to identify rare events. Nonetheless, up to now, this is the largest case series of children and young adults affected by INS with SARS-CoV-2 infection during the whole course of the pandemic.
Conclusions
Considering the homogeneity of this cohort and the total number of collected cases, the results of our study support the generally benign course of SARS-CoV-2 infection in children and young adults with INS, with no severe complications, even in patients with immunosuppressive therapy or proteinuria. Self-limiting proteinuria is a common finding during the infection, but the risk of clinically relevant relapses appears to be low. Our results support our previous recommendations on the management of children with INS during the pandemic, unless different variants with unexpected behaviors will emerge.
Supplementary Information
Below is the link to the electronic supplementary material.
Declarations
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
William Morello and Federica Alessandra Vianello contributed equally to this paper.
References
- 1.Noone DG, Iijima K, Parekh R. Idiopathic nephrotic syndrome in children. Lancet. 2018;392:61–74. doi: 10.1016/S0140-6736(18)30536-1. [DOI] [PubMed] [Google Scholar]
- 2.Takahashi S, Wada N, Murakami H, Funaki S, Inagaki T, Harada K. Nagata M (2007) Triggers of relapse in steroid-dependent and frequently relapsing nephrotic syndrome. Pediatr Nephrol. 2006;222(22):232–236. doi: 10.1007/S00467-006-0316-Y. [DOI] [PubMed] [Google Scholar]
- 3.WHO Coronavirus (COVID-19) Dashboard | WHO Coronavirus (COVID-19) Dashboard With Vaccination Data. https://covid19.who.int/. Accessed 2 Jun 2022
- 4.Dawood FS, Porucznik CA, Veguilla V, Stanford JB, Duque J, Rolfes MA, Dixon A, Thind P, Hacker E, Castro MJE, Jeddy Z, Daugherty M, Altunkaynak K, Hunt DR, Kattel U, Meece J, Stockwell MS. Incidence rates, household infection risk, and clinical characteristics of SARS-CoV-2 infection among children and adults in Utah and New York City, New York. JAMA Pediatr. 2022;176:59–67. doi: 10.1001/JAMAPEDIATRICS.2021.4217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Marlais M, Wlodkowski T, Vivarelli M, Pape L, Tönshoff B, Schaefer F, Tullus K. The severity of COVID-19 in children on immunosuppressive medication. Lancet Child Adolesc Health. 2020;4:e17–e18. doi: 10.1016/S2352-4642(20)30145-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mastrangelo A, Morello W, Vidal E, Guzzo I, AnnicchiaricoPetruzzelli L, Benetti E, Materassi M, Giordano M, Pasini A, Corrado C, Puccio G, Chimenz R, Pecoraro C, Massella L, Peruzzi L, Montini G, COVID-19 Task Force of the Italian Society of Pediatric Nephrology Impact of COVID-19 pandemic in children with CKD or immunosuppression. Clin J Am Soc Nephrol. 2020;16:449–451. doi: 10.2215/CJN.13120820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Morello W, Mastrangelo A, Guzzo I, Cusinato L, AnnicchiaricoPetruzzelli L, Benevenuta C, Martelli L, Dall'Amico R, Vianello FA, Puccio G, Massella L, Benetti E, Pecoraro C, Peruzzi L, Montini G, COVID-19 Task Force of the Italian Society of Pediatric Nephrology Prevalence of SARS-CoV-2-IgG antibodies in children with CKD or immunosuppression. Clin J Am Soc Nephrol. 2021;16:1097–1099. doi: 10.2215/CJN.00330121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Morello W, Vianello FA, Proverbio E, Peruzzi L, Pasini A, Montini G. COVID-19 and idiopathic nephrotic syndrome in children: systematic review of the literature and recommendations from a highly affected area. Pediatr Nephrol. 2022;37:757–764. doi: 10.1007/s00467-021-05330-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Krishnasamy S, Mantan M, Mishra K, Kapoor K, Brijwal M, Kumar M, Sharma S, Swarnim S, Gaind R, Khandelwal P, Hari P, Sinha A, Bagga A. SARS-CoV-2 infection in children with chronic kidney disease. Pediatr Nephrol. 2021;37:849–857. doi: 10.1007/S00467-021-05218-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vasconcelos MA, Mendonça ACQ, Colosimo EA, Nourbakhsh N, Martelli-Júnior H, Silva LR, Oliveira MCL, Pinhati CC, Mak RH, Simões E Silva AC, Oliveira EA (2022) Outcomes and risk factors for death among hospitalized children and adolescents with kidney diseases and COVID-19: an analysis of a nationwide database. Pediatr Nephrol. 10.1007/s00467-022-05588-0 [DOI] [PMC free article] [PubMed]
- 11.Karim SSA, Karim QA. Omicron SARS-CoV-2 variant: a new chapter in the COVID-19 pandemic. Lancet. 2021;398:2126–2128. doi: 10.1016/S0140-6736(21)02758-6/ATTACHMENT/FB9D1AF8-6927-48BD-B7B7-A5055C3743D5/MMC1.PDF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang L, Berger NA, Kaelber DC, Davis PB, Volkow ND, Xu R. Incidence Rates and clinical outcomes of SARS-CoV-2 infection with the Omicron and Delta variants in children younger than 5 years in the US. JAMA Pediatr. 2022;176:811–813. doi: 10.1001/JAMAPEDIATRICS.2022.0945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sorveglianza integrata COVID-19: i principali dati nazionali. https://www.epicentro.iss.it/coronavirus/sars-cov-2-sorveglianza-dati. Accessed 18 Aug 2022
- 14.Pasini A, Benetti E, Conti G, Ghio L, Lepore M, Massella L, Molino D, Peruzzi L, Emma F, Fede C, Trivelli A, Maringhini S, Materassi M, Messina G, Montini G, Murer L, Pecoraro C, Pennesi M. The Italian Society for Pediatric Nephrology (SlNePe) consensus document on the management of nephrotic syndrome in children: part I –diagnosis and treatment of the first episode and the first relapse. Ital J Pediatr. 2017;43:41. doi: 10.1186/s13052-017-0356-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kidney Disease: Improving Global Outcomes (KDIGO) Glomerular Diseases Work Group KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney Int. 2021;100:S1–S276. doi: 10.1016/J.KINT.2021.05.021/ATTACHMENT/D54815F6-C088-4C3F-A044-CFC19E0C0544/MMC1.PDF. [DOI] [PubMed] [Google Scholar]
- 16.Popolazione residente al 1° gennaio : Per fascia di età. http://dati.istat.it/Index.aspx?QueryId=42869. Accessed 18 Aug 2022
- 17.Weinbrand-Goichberg J, Ben Shalom E, Rinat C, Choshen S, Tzvi-Behr S, Frishberg Y, Becker-Cohen R. COVID-19 in children and young adults with kidney disease: risk factors, clinical features and serological response. J Nephrol. 2022;35:121–129. doi: 10.1007/s40620-021-01171-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Connelly JA, Chong H, Esbenshade AJ, Frame D, Failing C, Secord E, Walkovich K. Impact of COVID-19 on pediatric immunocompromised patients. Pediatr Clin North Am. 2021;68:1029–1054. doi: 10.1016/j.pcl.2021.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Choi B, Choudhary MC, Regan J, Sparks JA, Padera RF, Qiu X, Solomon IH, Kuo HH, Boucau J, Bowman K, Adhikari UD, Winkler ML, Mueller AA, Hsu TY, Desjardins M, Baden LR, Chan BT, Walker BD, Lichterfeld M, Brigl M, Kwon DS, Kanjilal S, Richardson ET, Jonsson AH, Alter G, Barczak AK, Hanage WP, Yu XG, Gaiha GD, Seaman MS, Cernadas M, Li JZ. Persistence and evolution of SARS-CoV-2 in an immunocompromised host. N Engl J Med. 2020;383:2291–2293. doi: 10.1056/NEJMC2031364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Scherer EM, Babiker A, Adelman MW, Allman B, Key A, Kleinhenz JM, Langsjoen RM, Nguyen PV, Onyechi I, Sherman JD, Simon TW, Soloff H, Tarabay J, Varkey J, Webster AS, Weiskopf D, Weissman DB, Xu Y, Waggoner JJ, Koelle K, Rouphael N, Pouch SM, Piantadosi A. SARS-CoV-2 evolution and immune escape in immunocompromised patientS. N Engl J Med. 2022;386:2436–2438. doi: 10.1056/NEJMC2202861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ssentongo P, Ssentongo AE, Voleti N, Groff D, Sun A, Ba DM, Nunez J, Parent LJ, Chinchilli VM, Paules CI. SARS-CoV-2 vaccine effectiveness against infection, symptomatic and severe COVID-19: a systematic review and meta-analysis. BMC Infect Dis. 2022;22:439. doi: 10.1186/s12879-022-07418-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen LL, Chua GT, Lu L, Chan BP, Wong JS, Chow CC, Yu TC, Leung AS, Lam SY, Wong TW, Tsang HW, Wong IC, Chan KH, Yuen KY, Ip P, Kwan MY, To KK. Omicron variant susceptibility to neutralizing antibodies induced in children by natural SARS-CoV-2 infection or COVID-19 vaccine. Emerg Microbes Infect. 2022;11:543–547. doi: 10.1080/22221751.2022.2035195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Katz SE, Edwards K. Protecting children against omicron. JAMA. 2022 doi: 10.1001/JAMA.2022.7315. [DOI] [PubMed] [Google Scholar]
- 24.Frenck RW, Klein NP, Kitchin N, Gurtman A, Absalon J, Lockhart S, Perez JL, Walter EB, Senders S, Bailey R, Swanson KA, Ma H, Xu X, Koury K, Kalina WV, Cooper D, Jennings T, Brandon DM, Thomas SJ, Türeci Ö, Tresnan DB, Mather S, Dormitzer PR, Şahin U, Jansen KU, Gruber WC, C4591001 Clinical Trial Group Safety, immunogenicity, and efficacy of the BNT162b2 Covid-19 vaccine in adolescents. N Engl J Med. 2021;385:239–250. doi: 10.1056/nejmoa2107456. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.