Abstract
Pregnancy is a critical period for medical care, during which the well-being of woman and fetus must be considered. This is particularly relevant in managing non-psychotic mental disorders since treatment with central nervous system-active drugs and untreated NMDs may have negative effects. Some well-known herbal preparations (phytopharmaceuticals), including St. Johnʼs wort, California poppy, valerian, lavender, and hops, possess antidepressant, sedative, anxiolytic, or antidepressant properties and could be used to treat mental diseases such as depression, restlessness, and anxiety in pregnancy. Our goal was to assess their safety in vitro , focusing on cytotoxicity, induction of apoptosis, genotoxicity, and effects on metabolic properties and differentiation in cells widely used as a placental cell model (BeWo b30 placenta choriocarcinoma cells). The lavender essential oil was inconspicuous in all experiments and showed no detrimental effects. At low-to-high concentrations, no extract markedly affected the chosen safety parameters. At an artificially high concentration of 100 µg/mL, extracts from St. Johnʼs wort, California poppy, valerian, and hops had minimal cytotoxic effects. None of the extracts resulted in genotoxic effects or altered glucose consumption or lactate production, nor did they induce or inhibit BeWo b30 cell differentiation. This study suggests that all tested preparations from St. Johnʼs wort, California poppy, valerian, lavender, and hops, in concentrations up to 30 µg/mL, do not possess any cytotoxic or genotoxic potential and do not compromise placental cell viability, metabolic activity, and differentiation. Empirical and clinical studies during pregnancy are needed to support these in vitro data.
Key words: non-psychotic mental disorders, pregnancy, BeWo cell line, toxicity, Hypericum perforatum, Hypericaceae, Eschscholzia californica, Papaveraceae, Valeriana officinalis, Caprifoliaceae, Lavandula angustifolia, Lamiaceae, Humulus lupulus, Cannabaceae
Abbreviations
- CHMP
Committee on Herbal Medicinal Products
- CNS
central nervous system
- CPT
camptothecin
- EMA
European Medicines Agency
- EMS
ethyl methanesulfonate
- FACS
fluorescence-activated cell sorting
- FITC
fluorescein isothiocyanate
- FSK
forskolin
- LMA
low-melting agarose
- NMA
normal-melting agarose
- NMDs
non-psychotic mental disorders
- SSRI
selective serotonin reuptake inhibitor
- TX
Triton-X-100
- β- hCG
beta-human chorionic gonadotropin
Introduction
NMDs, such as depression and anxiety disorders, are common issues during pregnancy and the postpartum period. A recent prevalence estimate in Switzerland revealed that 17% of women receive mental health care during pregnancy and the first postpartum year 1 . For women with pre-existing psychiatric conditions, this period is prone to recurrence or even worsening of NMDs. About 10 – 13% of fetuses are exposed to a psychotropic drug 2 , leading to side effects for both the mother and the fetus or newborn 3 . The most commonly used psychotropic medications include antidepressants, with the use of SSRIs such as citalopram being preferred 4 . Exposure to these medications and untreated depression in pregnancy have been associated with poor birth outcomes and increased risks, and the treatment of depression in pregnancy remains challenging 5 , 6 . Benzodiazepines, particularly diazepam, are used to treat anxiety, sleep, and mood disorders. However, their use in the weeks before childbirth may provoke neonatal withdrawal syndrome, floppy infant syndrome, or various acute toxic effects in the newborn 7 . Since treating mental diseases during pregnancy with conventional medications has several drawbacks, and since an untreated NMD ifself should be avoided, safe herbal preparations may be a treatment option. Several herbal candidates could be considered to treat mild NMDs during pregnancy, namely hops, valerian, lavender, California poppy, and St. Johnʼs wort.
Preparations of hops ( Humulus lupulus L., Cannabaceae) and valerian ( Valeriana officinalis L., Caprifoliaceae) have a long history of traditional use for the treatment of sleeping disorders 8 . A noninterventional study of a combination of hops and valerian improved sleep latency and quality in adults with primary insomnia 8 . The combination also led to sleep-improving effects in a randomized, placebo-controlled sleep-EEG study 9 . However, divergent results have been reported regarding the clinical effectiveness of hop-valerian combinations (for a review, see 10 ). In rodents, valerian showed anxiolytic and antidepressant-like activity, which may account for the sleep-enhancing effects of valerian 11 . Data from a Swedish birth register from 1995 to 2004 with 860′215 women show that valerian preparations are among the most frequently used medicines during pregnancy, and no unfavorable effects on pregnancy outcomes were reported 12 . A multinational study also showed that valerian is among the most frequently used herbal preparations in pregnancy 13 . In a recent retrospective observational study performed in the South of Italy, no influence of valerian ingestion on pregnancy and neonatal outcomes was detected (n = 9) 14 . However, to the best of our knowledge, no clinical or in vivo data are available on the safety of hops preparations in pregnancy.
The essential oil of lavender ( Lavandula angustifolia Mill., Lamiaceae) is used in various applications and must contain 25 – 46% linalyl acetate and 20 – 45% linalool for therapeutic use 15 . It effectively treats anxiety 16 , general restlessness, and difficulty falling asleep 15 . In adults with generalized anxiety disorder, the anxiolytic effect was comparable to that of a benzodiazepine (low-dose lorazepam 17 and the SSRI paroxetine 18 ), and fewer adverse effects were reported than with the synthetic drugs.
California poppy ( Eschscholzia californica Cham., Papaveraceae) is traditionally used to relieve mild symptoms of mental stress and as a sleeping aid 19 . Although herbal products have been on the market for almost 40 years (since 1982), there are very few clinical studies on their efficacy 20 . Anxiolytic and sedative effects have been reported in rodents 21 , which could be explained by high affinity to benzodiazepine receptors of yet unknown compounds 22 . Various alkaloids have been identified in the aerial parts of California poppy but do not seem to bind to GABA A receptors and thus to modulate chloride currents 23 .
Phytomedicines containing St. Johnʼs wort ( Hypericum perforatum L., Hypericaceae) are widely used for the treatment of mild to moderate depression 24 , nervous unrest, anxiety, and, to some extent, insomnia 15 . A meta-analysis of 27 studies revealed that the herbal antidepressant did not differ in efficacy from SSRIs 25 . Treatment of mild-to-moderate depression with St. Johnʼs wort extract resulted in fewer adverse events and thus fewer treatment discontinuations 25 . However, several relevant drug interactions with St. Johnʼs wort extract must be considered in clinical practice 26 . The potential risk for pregnant women is still unclear due to the current lack of clinical data and equivocal results in animal studies. Data on the prevalence of fetal malformations in pregnant women exposed to St. Johnʼs wort preparations are not extensive. A study using data from the Danish National Birth Cohort reported a nonsignificantly higher prevalence of malformations (8.1%; 3/38) in the group exposed to St. Johnʼs wort than in the comparator group (3.3%, 2′891/90′128, p = 0.13). However, the difference was based on 3 cases only and did not follow a specific pattern 27 . Another prospective study compared the rate of major malformations in subjects taking St. Johnʼs wort (5.3%; 2/38) with pregnant women treated with another antidepressant (4.2%; 2/48) or healthy women (0%; 0/56). Similarly, the number of preterm and live births was comparable in all 3 groups 28 .
A considerable proportion of healthcare professionals who deal with pregnant women daily recommend herbal medications 29 . Pregnant women themselves often turn to phytomedicines 30 and tend to perceive herbal products as safe 31 . However, the CHMP of the EMA does not recommend the use of hops 32 , valerian 33 , lavender 34 , California poppy 19 , and St. Johnʼs wort 35 during pregnancy because of a lack of sufficient safety data. Specifically, it is unknown to what extent phytochemicals cross the placenta barrier or interfere with placenta function and, thereby, may interfere with the development of the fetus.
Assessing the safety of phytopharmaceuticals is a significant challenge. The active ingredients of herbal extracts are multi-compound mixtures, many of which may be metabolized by the intestinal microbiota upon oral administration or by the liver once they have been absorbed. An interdisciplinary project is underway to fill this gap and address some of these issues, ranging from metabolizing phytochemicals by intestinal microbiota to intestinal absorption, liver metabolism, and passage across the placental barrier 36 . We performed an in vitro assessment in BeWo cells of the safety profile of medicinal plants that are used to treat mild NMDs. This human choriocarcinoma cell line (clone b30) is a widely used in vitro model for investigating placental metabolism, villous trophoblast fusion, syncytium formation, and monolayer permeability. The undifferentiated, mononuclear cells (villous cytotrophoblasts) can undergo fusion and morphological differentiation into a layer of syncytiotrophoblasts with the addition of FSK 37 . The formation of a layer of syncytiotrophoblasts and the concomitant production of β- hCG are essential for the function of the human placenta and the preservation of a healthy pregnancy 37 . We assessed the cytotoxicity and genotoxicity of the extracts in BeWo b30 cells, their effects on critical metabolic properties such as glucose consumption and lactate production, and the ability to induce or inhibit cell differentiation.
Results
In herbal extracts, defining a concentration range for in vitro testing that reflects clinically attainable tissue concentrations is challenging. Maximum daily recommended doses for phytomedicines containing St. Johnʼs wort, hops, valerian, California poppy, and lavender essential oil range between 80 and 1200 mg/day (highest value corresponding to maximal dose of Somnofor; see 38 ). Assuming a daily dose of 1200 mg extract dissolved in body-water (ca. 30.6 L in 15 – 24-year-old women, calculated for an average body weight of 60 kg 39 ), an absence of metabolization by the intestinal microbiota, and a 100% bioavailability of all compounds, a maximal concentration of 39 µg/mL of extract would be reached in the body fluid. Based on this calculation, a concentration range of 0.03 to 100 µg/mL was used in the assays, whereby the higher test concentrations were significantly above possible clinically achieved tissue concentrations.
First, the effects of the extracts on cell viability and apoptosis induction were assessed via the turnover of WST-1 in viable cells. At concentrations of 30 µg/mL, all extracts showed no or only minimal cytotoxicity after 72 h of incubation, and pronounced cytotoxic effects for 4 out of 5 extracts were only observed at a concentration of 100 µg/mL ( Fig. 1 ). At this concentration, St. Johnʼs wort, California poppy, and valerian extracts reduced cell viability by 25 – 40% compared to untreated control cells, while hops extract lowered cell viability by 75%. Apoptosis in BeWo b30 cells was assessed via flow cytometric analysis. The extracts of California poppy and lavender did not induce apoptosis at concentrations up to 100 µg/mL ( Fig. 2 ). Extracts of St. Johnʼs wort, valerian, and hops only increased apoptotic cell death at the highest concentration of 100 µg/mL. Diazepam and citalopram were also tested for comparison and did not show cytotoxicity in this concentration range ( Fig. 8S , Supporting Information).
Fig. 1.
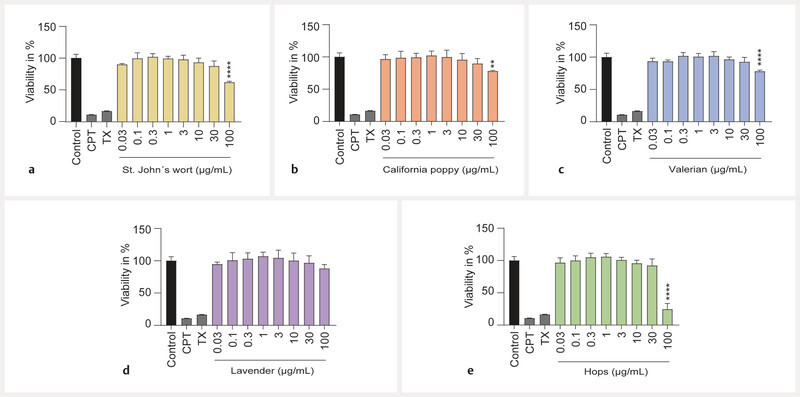
Effects of extracts on cell viability of undifferentiated BeWo b30 cells. Cell viability was assessed with a WST-1 assay after 72 h of treatment. None of the extracts showed a significant effect at concentrations up to 30 µg/mL. Only the highest concentration of 100 µg/mL led to effects for 4 extracts St. Johnʼs wort ( a ), California poppy ( b ), valerian ( c ), and hops ( e ). Lavender oil did not lead to any significant effect ( d ). The effects are shown as fold change compared to the untreated control. Treatments with 300 µM CPT and 0.5% Triton-X-100 (TX) served as toxicity controls. Results were normalised to untreated control signal = 100% (n = 3).
Fig. 2.
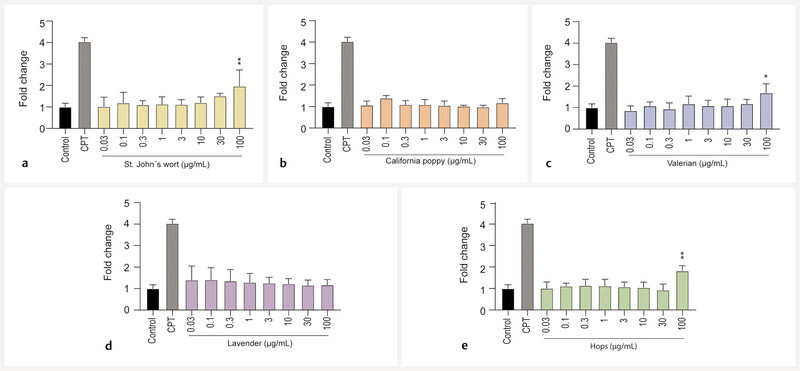
Effects of extracts on cell death of undifferentiated BeWo b30 cells after treatment for 72 h. Apoptosis only significantly increased for the highest concentrations of St. Johnʼs wort ( a ), valerian ( c ), and hops ( e ). California poppy ( b ) and lavender ( d ) did not induce apoptosis at concentrations up to 100 µg/mL. Results were calculated as fold change compared to the untreated control. Camptothecin (CPT, 300 µM) was used as a positive control for apoptosis (n = 3).
Possible genotoxicity of extracts was assessed with the aid of the comet assay, whereby a noncytotoxic concentration range (3, 10, and 30 µg/mL) was tested. None of the extracts induced notable increases in tail DNA, and mostly intact nuclear DNA could be detected after treatment of the cells with extracts for 3 h ( Fig. 3 ). Diazepam and citalopram also showed no genotoxicity in this assay ( Fig. 8S , Supporting Information).
Fig. 3.
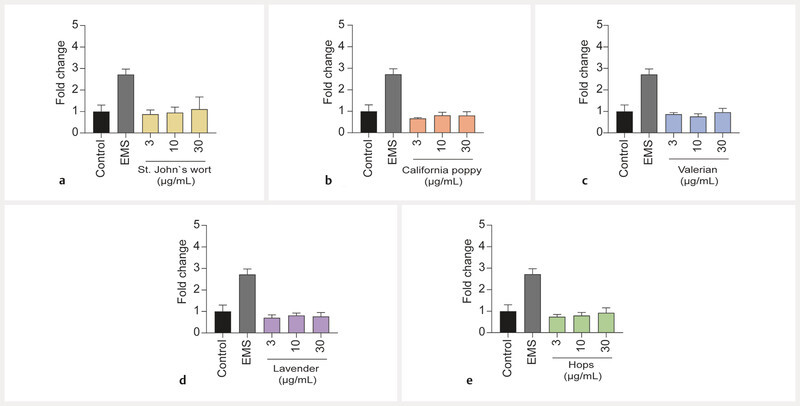
Effects of extracts of St. Johnʼs wort ( a ), California poppy ( b ), valerian ( c ), lavender ( d ), and hops ( e ) on tail DNA in undifferentiated BeWo b30 cells after exposure for 3 h. No significant genotoxic effects were observed at extract concentrations ranging from 3 to 30 µg/mL. Results were calculated as fold change compared to the untreated control. Ethyl methanesulfonate (EMS, 3 mM) was used as a positive control (n = 3).
In the next step, the effect of the herbal extracts on glucose consumption and lactate production was examined. None of the extracts affected the metabolic activity of viable BeWo b30 cells when tested at concentrations up to 100 µg/mL. Glucose and lactate concentrations of cell supernatants were not statistically different from the untreated control ( Fig. 4 ). Data were normalized to the protein content. Without the normalization, valerian extract at a 100 µg/mL concentration led to a decrease in glucose consumption and concomitant reduction in lactate production (data not shown). Diazepam and citalopram did not lead to changes in glycolytic metabolism ( Fig. 8S , Supporting Information).
Fig. 4.
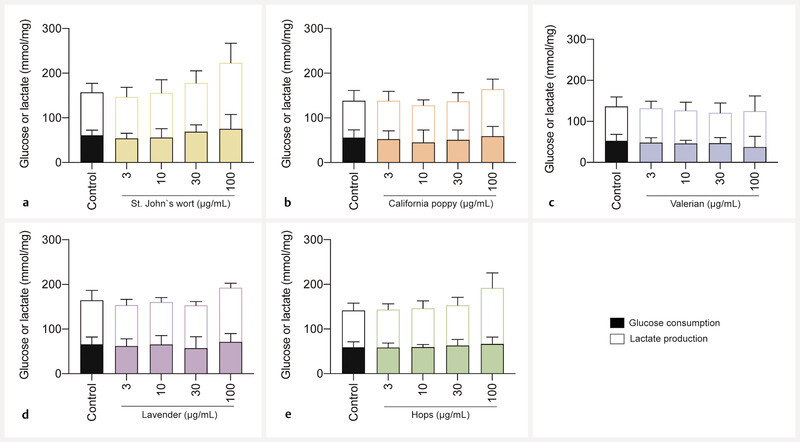
Effects of extracts on glucose consumption and lactate production in undifferentiated BeWo b30 cells after treatment for 48 h. Data were normalized per amount of protein (mg). Statistically significant impairment of metabolic activity could not be detected at all test concentrations (3, 10, 30, 100 µg/mL) of extracts of St. Johnʼs wort ( a ), California poppy ( b ), valerian ( c ), lavender ( d ), and hops ( e ). The control consisted of cell culture media containing 0.2% of DMSO. Data were obtained from 3 independent experiments (n = 3; in triplicate) and are shown as mean ± SD.
Finally, the impact of the extracts on β- hCG secretion was investigated. Upon adding 50 µM FSK as a positive control, a 100-fold increase of β -hCG levels was observed, which is characteristic of differentiation of BeWo b30 cells. In contrast, none of the 5 herbal extracts triggered an increase in β- hCG production ( Fig. 5 a ). Also, no significant decrease of β- hCG levels was observed for cells treated with the extracts compared to untreated control. Only the valerian extractʼs highest test concentration (100 µg/mL) led to a slight (nonsignificant) reduction. Only nontoxic extract concentrations were chosen to inhibit placental cell differentiation by herbal preparations (≤ 30 µg/mL). The addition of 5 µM FSK resulted in 5-fold increased β- hCG levels, showing that differentiation of BeWo b30 cells was successful (as shown in Fig. 5 b ) even after using 10-fold lower FSK concentrations and cell exposure for 24 h only. A 24 h pre-incubation with any of the 5 herbal preparations (in concentrations of 1, 3, 10, and 30 µg/mL) did not have a statistically significant inhibitory effect on the FSK-induced placenta cell differentiation ( Fig. 5 b ).
Fig. 5.
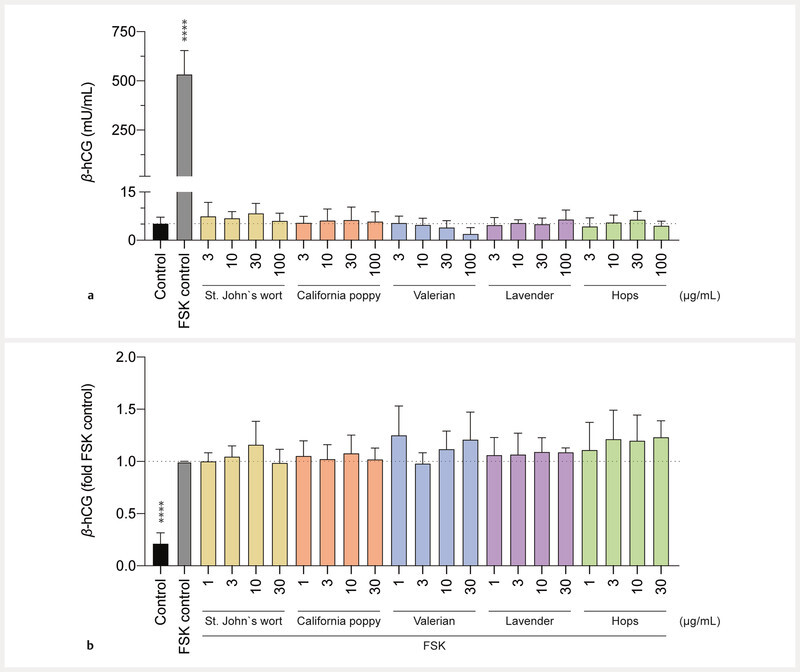
Effects of extracts on the production of β- hCG in BeWo b30 cells. The control consisted of cell culture media containing 0.2% of DMSO. Data are presented as mean ± SD of at least 3 independent experiments (n = 3 – 4; in triplicate). a Comparison of β- hCG secretion of BeWo b30 cells upon 48 h treatment with increasing concentrations of test compounds vs. 50 µM FSK control. b Effects on inhibition of FSK-induced differentiation of BeWo b30 cells (detected by measuring β- hCG). Treatment with 5 µM FSK led to increased β- hCG levels in all test compounds after exposure to different concentrations (1, 3, 10, 30 µg/mL) after an incubation of 48 h. Cells were pre-treated with the test compounds (or cell culture medium) for 24 h, before adding 5 µM FSK for another 24 h.
Discussion
Hydroalcoholic extracts from St. Johnʼs wort, California poppy, valerian and hops, and lavender essential oil did not induce in vitro cytotoxicity, apoptosis, or genotoxicity in BeWo b30 cells at concentrations up to 30 µg/mL. Moreover, there were no abnormalities in metabolic properties and no impact on cell differentiation. No changes were observed when normalizing the glucose and lactate concentrations (mmol) to the amount of protein (mg). This indicates that viable cells had a normal glycolytic metabolism under a 48 h exposure to extract concentrations up to 100 µg/mL. No significant β -hCG release was observed for all extracts, suggesting that they did not induce the cell fusion process. No significant decrease in β- hCG supernatant concentrations was seen when BeWo b30 cells were pre-incubated for 24 h with noncytotoxic concentrations (≤ 30 µg/mL) of the extracts. These results showed that the extracts could not inhibit the cell differentiation of cytotrophoblasts into syncytiotrophoblasts. Reduced cell viability and induction of apoptosis were seen at the highest extract concentration of 100 µg/mL, except for lavender essential oil, which was inconspicuous in all experiments. To summarize, all herbal extracts of St. Johnʼs wort, California poppy, valerian, lavender, and hops showed no toxicological abnormalities in a relevant concentration range.
The main strength of this in vitro study is the combination of different and well-recognized assays to assess the safety of commonly used herbal preparations. A wide range of concentrations and various time exposures of up to 3 days were tested using the cell line most frequently used as a cellular model of placenta, which closely reflects the biological environment. The constraint to in vitro model is, at the same time, a limitation, which is quite difficult to overcome and translate into the clinical situation. For instance, intestinal and hepatic metabolism, clearance, allergic reactions, and pregnancy-related specificities could not be considered.
In our study, St. Johnʼs wort showed no significant effects at concentrations up to 30 µg/mL in all experiments. The viability assay and the apoptosis markers showed an impairment of the BeWo b30 cells only at the highest concentration of 100 µg/mL. This is in good agreement with other published in vitro data, where the toxicity of St. Johnʼs wort was seen at high concentrations (≥ 150 µg/mL) 40 . Moreover, no effect on the β- hCG production could be detected at 25 µg/mL of the extract 40 . Hyperforin reportedly inhibited the growth of embryonic stem cells and induced apoptosis in fibroblasts and thus may, at high concentrations, pose embryotoxic and teratogenic risks 41 . It should be added that the hyperforin content in phytomedicines varies widely, depending on the specific product 42 .
We only found minimal cytotoxic potential for California poppy, as only the viability assay showed a slight significance at 100 µg/mL. Currently, there is a lack of sufficient clinical data on the safety of California poppy, as preparations have not been tested for reproductive toxicity, genotoxicity, and carcinogenicity. Furthermore, no data on in vitro cytotoxicity, genotoxicity, or influence on glycolytic metabolism and placental cell differentiation could be found. With our data, we have managed to provide an insight into in vitro safety that no one has found before to the best of our knowledge.
Valerian has been repeatedly discussed regarding a possible genotoxic potential. In a human endothelial cell line, 5 – 60 µg/mL of a dichloromethane extract of valerian show no significant cytotoxic effects. Discrete DNA damage occurred after in vitro exposure for 48 h to 40 or 60 µg/mL extract, but not at concentrations ≤ 40 µg/mL 43 . Valepotriates were considered responsible for the DNA damage 43 . However, valepotriates are unstable and degrade during the drying and heating of roots, and the valerian extract used in our study was devoid of valepotriates (see Fig. 1S, Table 1S , Supporting Information). In vivo , chromosomal aberrations, spermatozoa abnormalities, and a decrease in nucleic acids in testicular cells were reported to occur in mice after 7 days of oral administration of valerian by gavage (capsules containing 800 mg valerian root and 220 mg valerian root dried extract; dosage 500 – 2000 mg/kg/day) 44 . Valerian showed significant cytotoxic events in our setup for the same assays as St. Johnʼs wort but slightly less pronounced. It was also the only extract that had any significant effects on the metabolic properties of the BeWo b30 cells before normalizing per amount of protein available (data not shown). Valerian also showed a tendency (nonsignificant) to reduce β- hCG hormone concentrations in cell supernatants, which is most certainly due to cell toxicity and, therefore, decreased cell viability. Nevertheless, in all assays, the effects were only seen for the artificial nonphysiological concentration of 100 µg/mL. No significant effects could be detected at concentrations up to 30 µg/mL.
The essential oil of lavender did not show any impairment of the BeWo b30 cells at concentrations up to 100 µg/mL. How much of the volatile components of the oil were still present at the end of our experiments (48 – 72 h at 37 °C) is unknown and requires further clarification.
The hops extract affected cell viability and induced apoptosis only at a physiologically irrelevant concentration of 100 µg/mL. Only a few toxicity data on hops (compounds) are available. This is the case of prenylated flavonoids that revealed antiproliferative and cytotoxic effects in human cancer cell lines 45 . It must be noted that hop extracts are often combined with valerian or other extracts, and mono-preparations are not available in Switzerland.
Extracts of St. Johnʼs wort, California poppy, valerian, lavender, and hops do not appear to affect the functionality of placenta cells at concentrations that can be expected upon ingestion of recommended daily doses of phytomedicines. Only at very high test concentrations, particularly in the case of St. Johnʼs wort, a decrease of cell viability and induction of apoptosis was observed. Most importantly, no indications for genotoxic effects and no alterations in important metabolic parameters or cell differentiation were detected.
Materials and Methods
Plant material and extraction
All plant material was of Ph. Eur. grade. V. officinalis roots, H. lupulus flowers, and H. perforatum herb were purchased from Dixa (lot numbers 180 084, 191 241, 192 140, respectively). E. californica herb was obtained from Galke (lot number 811 502). Voucher specimens (numbers 1029, 1167, 1166, and 1234, respectively) have been deposited at the Division of Pharmaceutical Biology, University of Basel. The powdered plant material was extracted with 70% EtOH by pressurized liquid extraction in a Dionex ASE 200 Accelerated Solvent Extractor. Three cycles of extraction of 5 min each were performed at a temperature of 70 °C and a pressure of 120 bar. L. angustifolia essential oil Ph. Eur. was purchased from Hänseler (lot number 2018.01.0274).
HPLC-PDA-ESI-MS analysis of herbal extracts
HPLC-PDA-ESI-MS analysis of V. officinalis ( Fig. 1S, Table 1S , Supporting Information), H. lupulus ( Fig. 2S, Table 2S , Supporting Information), E. californica ( Fig. 3S, Table 3S , Supporting Information), and H. perforatum ( Fig. 4S, Table 4S , Supporting Information) 70% EtOH extracts were performed on an LC-MS system consisting of an 8030 triple quadrupole MS connected to an HPLC system consisting of a DGU-20A degasser, an LC-20AD binary high-pressure mixing pump, a SIL-20 ACHT autosampler, a CTO-20AC column oven, and an SPD-M20A diode array detector (all Shimadzu). The mobile phase consisted of water (A) and acetonitrile (B), both containing 0.1% formic acid. Analyses were performed at 25 °C on a SunFire C18 column (3.5 µm; 150 × 3 mm i. d., Waters). V. officinalis and H. lupulus extracts were analyzed with a 5 – 100% B gradient in 30 min at flow rates of 0.4 mL/min or 0.5 mL/min, respectively. E. californica extract was analyzed with a 5 – 40% B gradient in 30 min at a 0.5 mL/min flow rate. H. perforatum extract was analyzed with a gradient of 10 – 23% B in 20 min, then 23 – 70% B in 10 min at a flow rate of 0.4 mL/min. Extracts were dissolved in DMSO at a concentration of 10 mg/mL. The injection volume was 10 µL. Compounds were identified based on UV and MS spectroscopic data. The identity of compounds was further confirmed by chromatographic comparison with reference compounds when available.
HPLC-UV analysis of hyperforin, adhyperforin, hypericin, and pseudohypericin in H. perforatum
HPLC-UV analysis was performed on an Alliance 2690 chromatographic system coupled to a PDA996 detector (Waters). The mobile phase consisted of water (A) and acetonitrile (B), both containing 0.1% trifluoroacetic acid (hyperforin and adhyperforin), or 0.5% trifluoroacetic acid (hypericin and pseudohypericin). Separation of hyperforin and adhyperforin was achieved on a Zorbax Eclipse XDB-C8 column (3.5 µm; 150 × 2.1 mm i. d., Agilent) with a gradient of 50 – 100% B in 20 min at a flow rate of 0.4 mL/min ( Fig. 5S , Supporting Information). Hypericin and pseudohypericin were analyzed on an Atlantis dC18 column (3 µm; 150 × 4.6 mm i. d., Waters) with a 45 – 100% B gradient in 15 min ( Fig. 6S , Supporting Information). The extract was dissolved in DMSO at a concentration of 10 mg/mL. The injection volume was 10 µL. The identity of hyperforin, hypericin, and pseudohypericin was confirmed by chromatographic comparison with reference compounds.
GC-MS analysis of L. angustifolia essential oil
GC-MS analysis was performed on a Hewlett-Packard G1503A GC/MS system equipped with a 5973 Mass Selective Detector and a 59 864B Ion Gauge Controller (all Agilent). A J&W 122 – 5536 GC column (0.5 µm; 30 m × 0.25 mm i. d., Agilent) was used, with helium (1.4 mL/min) as a carrier gas. The injector temperature was set at 280 °C, and the transfer line temperature was 240 °C. The following temperature program was applied: 60 °C hold for 1 min, linear increase to 240 °C at 10 °C/min, followed by 5 min at 240 °C. EI ionization was in positive ion mode (electron energy: 2040 V; Full Scan: m/z: 50 – 700). Linalool and linalyl acetate were identified by comparing their MS spectra with the NIST database ( Fig 7S, Table 5S , Supporting Information).
Cell culture
The human choriocarcinoma BeWo cell line (clone b30) was obtained from Dr. Tina Buerki-Thurnherr (Empa – Swiss Federal Laboratories for Materials Science and Technology, St. Gallen, Switzerland), with permission from Dr. Alan L. Schwartz (Washington University School of Medicine, MO, USA). Cells were maintained in modified F-12K Nut Mix (Gibco). The cell culture medium was supplemented with 10% heat-inactivated FBS, penicillin (100 U/mL) and streptavidin (100 µg/mL), and 1% L-glutamine (all from Gibco). Cells were cultured in monolayers at 37 °C in a humidified atmosphere of 5% CO 2 balance air, and the medium was changed every 2 – 3 days.
Cell treatments
Plant extracts and lavender essential oil were dissolved in sterile DMSO (Sigma-Aldrich) to obtain a final concentration of 50 mg/mL for the stock solutions. In each experiment, treatments and controls consisted of cells exposed to a final concentration of 0.2% (vol/vol) DMSO to not decrease the cell viability ( Fig. 9S , Supporting Information). If required, BeWo b30 cells were stimulated with 5 or 50 µM FSK (Lucerna-Chem). FSK was added to the medium directly from a DMSO stock solution (30 mM). For all assays, cells were treated with increasing concentrations of extracts (up to 100 µg/mL) in cell culture medium. Samples were always protected from light while using St. Johnʼs wort extract.
Cytotoxicity assay
The in vitro cytotoxicity of the different concentrations of herbal extracts (0.03, 0.1, 0.3, 1, 3, 10, 30, and 100 µg/mL) was tested using a WST-1 viability assay. BeWo b30 cells were seeded with 100 µL/well (2 × 10 5 cells/mL) in a 96-well flat-bottom plate the day before, followed by exposure to extract dilutions in a fresh culture medium. Cells treated with 300 µM CPT (apoptosis control; Tocris Bioscience) or 0.5% TX (necrosis control; Sigma-Aldrich) were used as positive controls. After an incubation period of 72 h at 37 °C and 5% CO 2 , the culture supernatant was aspirated and replaced by a medium without phenol red, and 5 µL Cell Proliferation Reagent WST-1 (Roche) was added. After 75 min of incubation at 37 °C and 5% CO 2 , a spectrophotometric measurement was taken at 450 nm using a plate reader (Tecan Reader Infinite M 200).
Apoptosis assay
To assess the level of apoptosis after application of the test substances, BeWo b30 cells were subjected to the same treatment described for the WST-1 assay. Each extract was prediluted in a 96-well V-bottom plate and then added to the BeWo b30 cell culture to achieve final concentrations. After 72 h, the cells were washed with PBS and detached using Accutase (Sigma-Aldrich). According to the manufacturerʼs instructions, all liquids were pooled to collect living cells and fragments, centrifuged at 300 g for 5 min, and then stained with AnnexinV-FITC (eBioscience). A FACS readout was obtained using a fluorescence-activated cell analysis (BD FACScalibur, BD Biosciences).
Comet assay
The genotoxic potential of the selected extracts was examined by conducting a comet assay. A short incubation time of 3 h was chosen to avoid the onset of cellular DNA-repair mechanisms. Before the experiment, microscopic slides were coated with 1.0% NMA, and 0.7% LMA (both by SERVA Electrophoresis GmbH) was prepared and stored at − 20 °C until use. The cell suspension (100 µL/well) was seeded a day before the experiment with a 4 × 10 5 cells/mL cell concentration. On the experiment day, different concentrations of herbal preparations (3, 10, and 30 µg/mL) or 3 mM EMS (positive control; Sigma-Aldrich) were added for 3 h. Then, 200 µL of a completely dissolved and slightly boiling 0.7% NMA solution was applied to the precoated slides, which were cooled on metal plates in the fridge for later use. After 3 h, the cells were washed once with PBS and dissolved using Accutase (Sigma-Aldrich). The cells were then resuspended in 30 µL complete medium. The previously prepared LMA was rapidly heated to 100 °C and then kept at 38 °C. The cells were then gently mixed with 90 µL 0.7% LMA and added as a final layer to the slides. After the slides were cooled on the metal plates for 15 min, the slides were placed in a lysis solution in the refrigerator for 1 h, after which the DNA was exposed. The slides were then placed in the electrophoresis chamber and submerged with electrophoresis buffer. After 20 min accommodation time, electrophoresis was performed for 20 min at 25 V/300 mA. Finally, the slides were washed with ddH 2 O and PBS and fixed with 99% EtOH. For the microscopic measurement, the fixed samples were stained with ethidium bromide solution (5 µg/mL; Carl Roth GmbH). Pictures were taken for later analysis with CometScore software (version 2.0.038 for Windows; TriTek Corp., USA).
Glucose and lactate concentration measurements
Cells were seeded into transparent 24-well plates at a density of 2.5 × 10 4 cells/1000 µL/well. After overnight incubation, they were exposed to different concentrations (3, 10, 30, and 100 µg/mL) of plant extracts, lavender essential oil, and untreated control (0.2% DMSO) for 48 h. Cell culture supernatants and pellets were collected and immediately frozen at − 80 °C for subsequent analysis. All experiments were performed 3 times independently (in triplicate). The metabolic parameters–glucose and lactate–were determined using an automated blood gas analyzer (ABL800 Flex, Radiometer Medical ApS) based on amperometric measuring principles.
Placental cell differentiation and β -hCG production
Two different setups were used to test the influence of plant extracts and lavender essential oil on the induction or inhibition of placental cell differentiation. For the induction setup, cells were seeded into transparent 24-well plates (2.5 × 10 4 cells/1000 µL/well) before exposure to different concentrations (3, 10, 30, and 100 µg/mL) of test compounds and control (0.2% DMSO) for 48 h the next day. FSK (50 µM) was used as the positive control. For the inhibition setup, cells were seeded with 100 µL/well (1 × 10 5 cells/mL) into transparent 96-well flat-bottom plates on the day before the stimulation with test compounds and cell culture medium. After 24 h incubation, cells were differentiated with 5 µM FSK for another 24 h. All experiments were performed at least 3 times independently (in triplicate). Analysis of β -hCG concentrations–a marker of placenta cell differentiation–was performed using cell culture supernatants by standard ELISA (see 36 for additional references). Transparent 96-well flat-bottom microplates were used for all analyses. Samples treated with 50 or 5 µM FSK were diluted 1 : 50 or 1 : 10, respectively, before analysis. Rabbit polyclonal anti-hCG antibody (Agilent Dako) was used at a 1 : 1′000 dilution; mouse monoclonal anti-hCG (abcam) and goat anti-mouse-IgG-horseradish peroxidase conjugate (abcam) antibodies were both used at a 1 : 5′000 dilution. The peptide hormone hCG (Lucerna-Chem) was used as reference standard.
Protein concentration
BeWo b30 cells were lysed and extracted in the radioimmunoprecipitation assay buffer (Thermo Scientific) solution with 0.1% protease and phosphatase inhibitor single-use cocktail and 0.1% EDTA solution (Thermo Scientific). The cellular protein concentration was determined in transparent 96-well flat-bottom microplates by spectrophotometric quantification at 562 nm using the bicinchoninic acid protein assay kit (Pierce) with BSA (Thermo Scientific) as reference standard.
Statistical data analysis
Statistical data analyses were performed using GraphPad Prism (version 8.4.3 for Windows or macOS; GraphPad Software, La Jolla CA, USA). Shapiro-Wilk test was used to check for normal distribution. Multiple group comparisons were performed using the Brown-Forsythe and Welch ANOVA tests, followed by the Dunnettʼs T3 multiple comparisons posthoc test (with individual variances computed for each comparison). Probability values *p ≤ 0.05 were considered statistically significant. The asterisks represent significant differences from the control group (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001). All results are expressed as means ± SD of at least 3 independent experiments.
Contributorsʼ Statement
APSW, CG, MH and OP designed the study. DS and MW planned and conducted the experiments, performed the data analysis and prepared the original manuscript. DS focused on the metabolic properties and differentiation of placenta cells, and wrote the first version of the manuscript. MW conducted experiments on the viability, apoptosis and genotoxicity of placenta cells. AC performed the HPLC-UV-MS and GC-MS analyses. VA prepared all extracts. All authors agreed with the final version.
Acknowledgements
Alexandra Dolder is gratefully acknowledged for her support in executing cell experiments and measuring β -hCG, glucose, and lactate content in samples. The authors thank Dr. Tina Buerki-Thurnherr (EMPA, Switzerland) for kindly providing the BeWo b30 cells. Financial support was provided by the Swiss National Science Foundation (Sinergia, CRSII5_177260).
Conflict of Interest The authors declare that they have no conflict of interest.
These authors contributed equally to the work and should be considered as joint first authors.
These authors contributed equally to the work and should be considered as joint last authors.
Supporting Information
HPLC-UV-MS chromatograms of EtOH extracts of V. officinalis, H. lupulus, E. californica, H. perforatum ; HPLC-UV chromatograms of H. perforatum extract; GC-MS chromatogram of L. angustifolia essential oil; all data obtained with diazepam and citalopram; and in vitro cytotoxicity of different concentrations of DMSO on the BeWo b30 cells are available as Supporting Information.
References
- 1.Berger A, Bachmann N, Signorell A, Erdin R, Oelhafen S, Reich O, Cignacco E. Perinatal mental disorders in Switzerland: prevalence estimates and use of mental-health services. Swiss Med Wkly. 2017;147:w14417. doi: 10.4414/smw.2017.14417. [DOI] [PubMed] [Google Scholar]
- 2.Chisolm M S, Payne J L. Management of psychotropic drugs during pregnancy. BMJ. 2015;351:h5918. doi: 10.1136/bmj.h5918. [DOI] [PubMed] [Google Scholar]
- 3.Stewart D, Vigod S.Antenatal use of antidepressants and risk of teratogenicity and adverse pregnancy outcomes: selective serotonin reuptake inhibitors (SSRIs). (October 9, 2020)Accessed February 11, 2021 at:https://www.uptodate.com/contents/antenatal-use-of-antidepressants-and-risk-of-teratogenicity-and-adverse-pregnancy-outcomes-selective-serotonin-reuptake-inhibitors-ssris
- 4.Molenaar N M, Kamperman A M, Boyce P, Bergink V. Guidelines on treatment of perinatal depression with antidepressants: an international review. Aust N Z J Psychiatry. 2018;52:320–327. doi: 10.1177/0004867418762057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hanley G E, Oberlander T F. The effect of perinatal exposures on the infant: antidepressants and depression. Best Pract Res Clin Obstet Gynaecol. 2014;28:37–48. doi: 10.1016/j.bpobgyn.2013.09.001. [DOI] [PubMed] [Google Scholar]
- 6.Grote N K, Bridge J A, Gavin A R, Melville J L, Iyengar S, Katon W J. A meta-analysis of depression during pregnancy and the risk of preterm birth, low birth weight, and intrauterine growth restriction. Arch Gen Psychiatry. 2010;67:1012–1024. doi: 10.1001/archgenpsychiatry.2010.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Iqbal M M, Sobhan T, Ryals T. Effects of commonly used benzodiazepines on the fetus, the neonate, and the nursing infant. Psychiat Serv. 2002;53:39–49. doi: 10.1176/appi.ps.53.1.39. [DOI] [PubMed] [Google Scholar]
- 8.Salter S, Brownie S. Treating primary insomnia: the efficacy of valerian and hops. Aust Fam Physician. 2010;39:433–437. [PubMed] [Google Scholar]
- 9.Dimpfel W, Suter A. Sleep improving effects of a single dose administration of a valerian/hops fluid extract. A double-blind, randomized, placebo-controlled sleep-EEG study in a parallel design using electrohypnograms. Eur J Med Res. 2008;13:1–5. [PubMed] [Google Scholar]
- 10.Zanoli P, Zavatti M. Pharmacognostic and pharmacological profile of Humulus lupulus L . J Ethnopharmacol. 2008;116:383–396. doi: 10.1016/j.jep.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 11.Hattesohl M, Feistel B, Sievers H, Lehnfeld R, Hegger M, Winterhoff H. Extracts of Valeriana officinalis L. s.l. show anxiolytic and antidepressant effects but neither sedative nor myorelaxant properties . Phytomedicine. 2008;15:2–15. doi: 10.1016/j.phymed.2007.11.027. [DOI] [PubMed] [Google Scholar]
- 12.Holst L, Nordeng H, Haavik S. Use of herbal drugs during early pregnancy in relation to maternal characteristics and pregnancy outcome. Pharmacoepidemiol Drug Saf. 2008;17:151–159. doi: 10.1002/pds.1527. [DOI] [PubMed] [Google Scholar]
- 13.Kennedy D A, Lupattelli A, Koren G, Nordeng H. Herbal medicine use in pregnancy: results of a multinational study. BMC Complem Altern M. 2013;13:355. doi: 10.1186/1472-6882-13-355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Trabace L, Tucci P, Ciuffreda L, Matteo M, Fortunato F, Campolongo P, Trezza V, Cuomo V. “Natural” relief of pregnancy-related symptoms and neonatal outcomes: above all do no harm. J Ethnopharmacol. 2015;174:396–402. doi: 10.1016/j.jep.2015.08.046. [DOI] [PubMed] [Google Scholar]
- 15.Schilcher H, Kammerer S, Wegener T. München: Urban & Fischer; 2016. Leitfaden Phytotherapie. [Google Scholar]
- 16.Donelli D, Antonelli M, Bellinazzi C, Gensini G F, Firenzuoli F. Effects of lavender on anxiety: a systematic review and meta-analysis. Phytomedicine. 2019;65:153099. doi: 10.1016/j.phymed.2019.153099. [DOI] [PubMed] [Google Scholar]
- 17.Woelk H, Schläfke S. A multi-center, double-blind, randomised study of the Lavender oil preparation Silexan in comparison to Lorazepam for generalized anxiety disorder. Phytomedicine. 2010;17:94–99. doi: 10.1016/j.phymed.2009.10.006. [DOI] [PubMed] [Google Scholar]
- 18.Kasper S, Gastpar M, Müller W E, Volz H P, Möller H J, Schläfke S, Dienel A. Lavender oil preparation Silexan is effective in generalized anxiety disorder–a randomized, double-blind comparison to placebo and paroxetine. Int J Neuropsychopharmacol. 2014;17:859–869. doi: 10.1017/S1461145714000017. [DOI] [PubMed] [Google Scholar]
- 19.European Medicines Agency EMA/HMPC/680372/2013 – European Union herbal monograph on Eschscholzia californica Cham., herba. (January 28, 2015)Accessed June 2, 2021 at:https://www.ema.europa.eu/en/documents/herbal-monograph/final-european-union-herbal-monograph-eschscholzia-californica-cham-herba_en.pdf
- 20.European Medicines Agency EMA/HMPC/680375/2013–Assessment report on Eschscholzia californica Cham., herba. (January 28, 2015)Accessed June 2, 2021 at:https://www.ema.europa.eu/en/documents/herbal-report/final-assessment-report-eschscholzia-californica-cham-herba_en.pdf
- 21.Rolland A, Fleurentin J, Lanhers M C, Younos C, Misslin R, Mortier F, Pelt J M. Behavioural effects of the American traditional plant Eschscholzia californica : sedative and anxiolytic properties . Planta Med. 1991;57:212–216. doi: 10.1055/s-2006-960076. [DOI] [PubMed] [Google Scholar]
- 22.Rolland A, Fleurentin J, Lanhers M, Misslin R, Mortier F. Neurophysiological effects of an extract of Eschscholzia californica Cham. (Papaveraceae) . Phytother Res. 2001;15:377–381. doi: 10.1002/ptr.884. [DOI] [PubMed] [Google Scholar]
- 23.Fedurco M, Gregorová J, Šebrlová K, Kantorová J, Peš O, Baur R, Sigel E, Táborská E. Modulatory effects of Eschscholzia californica alkaloids on recombinant GABA A receptors . Biochem Res Int. 2015;2015:617620. doi: 10.1155/2015/617620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dingermann T, Loew D. Stuttgart: Wissenschaftliche Verlagsgesellschaft mbH; 2003. Phytopharmakologie: experimentelle und klinische Pharmakologie pflanzlicher Arzneimittel. [Google Scholar]
- 25.Cui Y H, Zheng Y. A meta-analysis on the efficacy and safety of St Johnʼs wort extract in depression therapy in comparison with selective serotonin reuptake inhibitors in adults. Neuropsychiatr Dis Treat. 2016;12:1715–1723. doi: 10.2147/NDT.S106752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nicolussi S, Drewe J, Butterweck V, Meyer zu Schwabedissen H E. Clinical relevance of St. Johnʼs wort drug interactions revisited. Br J Pharmacol. 2020;177:1212–1226. doi: 10.1111/bph.14936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kolding L, Pedersen L H, Henriksen T B, Olsen J, Grzeskowiak L E. Hypericum perforatum use during pregnancy and pregnancy outcome . Reprod Toxicol. 2015;58:234–237. doi: 10.1016/j.reprotox.2015.10.003. [DOI] [PubMed] [Google Scholar]
- 28.Moretti M E, Maxson A, Hanna F, Koren G. Evaluating the safety of St. Johnʼs Wort in human pregnancy. Reprod Toxicol. 2009;28:96–99. doi: 10.1016/j.reprotox.2009.02.003. [DOI] [PubMed] [Google Scholar]
- 29.Stewart D, Pallivalappila A, Shetty A, Pande B, McLay J. Healthcare professional views and experiences of complementary and alternative therapies in obstetric practice in North East Scotland: a prospective questionnaire survey. BJOG. 2014;121:1015–1019. doi: 10.1111/1471-0528.12618. [DOI] [PubMed] [Google Scholar]
- 30.Pallivalappila A R, Stewart D, Shetty A, Pande B, McLay J S. Complementary and alternative medicines use during pregnancy: A systematic review of pregnant women and healthcare professional views and experiences. Evid Based Complement Alternat Med. 2013;2013:1–10. doi: 10.1155/2013/205639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Petersen I, McCrea R L, Lupattelli A, Nordeng H. Womenʼs perception of risks of adverse fetal pregnancy outcomes: A large-scale multinational survey. BMJ Open. 2015;5:e007390. doi: 10.1136/bmjopen-2014-007390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.European Medicines Agency EMA/HMPC/682384/2013–Community herbal monograph on Humulus lupulus L., flos. (May 6, 2014)Accessed June 2, 2021 at:https://www.ema.europa.eu/en/documents/herbal-monograph/final-community-herbal-monograph-humulus-lupulus-l-flos_en.pdf
- 33.European Medicines Agency EMA/HMPC/150848/2015–European Union herbal monograph on Valeriana officinalis L., radix. (February 2, 2016)Accessed June 2, 2021 at:https://www.ema.europa.eu/en/documents/herbal-monograph/final-european-union-herbal-monograph-valeriana-officinalis-l-radix_en.pdf
- 34.European Medicines Agency EMA/HMPC/143181/2010–Community herbal monograph on Lavandula angustifolia Miller, aetheroleum. (March 27, 2012)Accessed June 2, 2021 at:https://www.ema.europa.eu/en/documents/herbal-monograph/final-community-herbal-monograph-lavandula-angustifolia-miller-aetheroleum_en.pdf
- 35.European Medicines Agency EMA/HMPC/101304/2008–Community herbal monograph on Hypericum perforatum L., herba (well-established medicinal use). (November 12, 2009)Accessed June 2, 2021 at:https://www.ema.europa.eu/en/documents/herbal-monograph/final-community-herbal-monograph-hypericum-perforatum-l-herba-well-established-medicinal-use_en.pdf
- 36.Spiess D, Abegg V F, Chauveau A, Treyer A, Reinehr M, Oufir M, Duong E, Potterat O, Hamburger M, Simões-Wüst A P. Placental passage of humulone and protopine in an ex vivo human perfusion system . Planta Med. 2021 doi: 10.1055/a-1578-3803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Orendi K, Gauster M, Moser G, Meiri H, Huppertz B. The choriocarcinoma cell line BeWo: Syncytial fusion and expression of syncytium-specific proteins. Reproduction. 2010;140:759–766. doi: 10.1530/REP-10-0221. [DOI] [PubMed] [Google Scholar]
- 38.Medicinal product information search platform (AIPS) Accessed April 9, 2021 at:https://www.swissmedicinfo.ch
- 39.Swiss Federal Statistical Office Durchschnittliches Körpergewicht (in kg). (February 28, 2019)Accessed February 11, 2021 at:https://www.bfs.admin.ch/bfs/de/home/statistiken/gesundheit.assetdetail.7586025.html
- 40.Da Conceicao A O, Takser L, Lafond J. Effect of St. Johnʼs wort standardized extract and hypericin on in vitro placental calcium transport . J Med Food. 2010;13:934–942. doi: 10.1089/jmf.2009.0161. [DOI] [PubMed] [Google Scholar]
- 41.Nakamura K, Aizawa K, Yamauchi J, Tanoue A. Hyperforin inhibits cell proliferation and differentiation in mouse embryonic stem cells. Cell Prolif. 2013;46:529–537. doi: 10.1111/cpr.12060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schäfer A M, Potterat O, Seibert I, Fertig O, Meyer zu Schwabedissen H E. Hyperforin-induced activation of the pregnane X receptor is influenced by the organic anion-transporting polypeptide 2B1. Mol Pharmacol. 2019;95:313–323. doi: 10.1124/mol.118.114066. [DOI] [PubMed] [Google Scholar]
- 43.Hui-Lian W, Dong-Fang Z, Zhao-Feng L, Yang L, Qian-Rong L, Yu-Zhen W. In vitro study on the genotoxicity of dichloromethane extracts of valerian (DEV) in human endothelial ECV304 cells and the effect of vitamins E and C in attenuating the DEV-induced DNA damages . Toxicol Appl Pharmacol. 2003;188:36–41. doi: 10.1016/s0041-008x(03)00017-6. [DOI] [PubMed] [Google Scholar]
- 44.Al-Majed A A, Al-Yahya A A, Al-Bekairi A, Al-Shabanah O, Qureshi S. Studies on the cytological and biochemical effects of valerian in somatic and germ cells of Swiss albino mice. Food Chem Toxicol. 2006;44:1830–1837. doi: 10.1016/j.fct.2006.06.001. [DOI] [PubMed] [Google Scholar]
- 45.Miranda C, Stevens J, Helmrich A, Henderson M, Rodriguez R, Yang Y H, Deinzer M, Barnes D, Buhler D. Antiproliferative and cytotoxic effects of prenylated flavonoids from hops ( Humulus lupulus ) in human cancer cell lines . Food Chem Toxicol. 1999;37:271–285. doi: 10.1016/s0278-6915(99)00019-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
HPLC-UV-MS chromatograms of EtOH extracts of V. officinalis, H. lupulus, E. californica, H. perforatum ; HPLC-UV chromatograms of H. perforatum extract; GC-MS chromatogram of L. angustifolia essential oil; all data obtained with diazepam and citalopram; and in vitro cytotoxicity of different concentrations of DMSO on the BeWo b30 cells are available as Supporting Information.


