Abstract
BACKGROUND
Individuals with heart failure (HF) frequently experience limitations in mobility, but specific aspects of these limitations are not well understood. This study investigated the association of HF severity, based on the New York Heart Association (NYHA) classes, with digital mobility outcomes (DMOs) and handgrip strength in older inpatients with HF.
METHODS
For this explorative analysis, hospital admission and discharge data from an ongoing, prospective cohort study were used. The sample included older participants with HF and a sub-sample of heart-healthy individuals. Participants were equipped with a wearable inertial measurement unit (IMU) system during mobility performance (balancing, sit-to-stand transfer, walking). We analyzed the association between 17 DMOs and HF severity with multiple linear regression models.
RESULTS
The total sample included 61 older participants (65–97 years of age, 55.7% female). Of all DMOs, only sway path in a semi-tandem stance position (m/s²) showed a relevant association with NYHA classes (admission: β = −0.28, P = 0.09; discharge: β = −0.39, P = 0.02). Handgrip strength showed a trend towards a significant association (admission: β = −0.15, P = 0.10; discharge: β = −0.15, P = 0.19).
CONCLUSIONS
This is to our best knowledge the first analysis on the association of HF severity and IMU-based DMOs. Sway path and handgrip strength may be the most promising parameters for monitoring mobility aspects in treatment of HF.
Heart failure (HF, with preserved and reduced ejection fraction[1]) is one of the most common, costly and disabling chronic conditions worldwide. HF primarily affects older people and the prevalence increases with age.[2] Approximately 80% of patients with HF are ≥ 65 years old[3,4] and in people ≥ 70 years, the prevalence is ≥ 10%.[5,6] The prognosis of HF is threatening and does not differ significantly from that of common malignant diseases. Therapy of HF is essentially based on the adjustment of risk factors and fluid intake, stage-appropriate pharmacotherapy and cause-dependent device therapy.[7]
HF is commonly classified using the New York Heart Association (NYHA) classification.[8,9] In stage I, there are no limitations of physical activity, while a person in stage IV is unable to carry on any physical activity without discomfort and may experience symptoms of HF or the anginal syndrome even at rest.
HF can affect mobility in several ways. On one hand, chronic HF leads to a variable state of malnutrition and consequently to sarcopenia. On the other hand, the reduced cardiopulmonary reserve allows only reduced activity, which promotes the process of sarcopenia.[10] Almost 60% of patients with HF have mobility disability, indicated by the inability to walk 2 to 3 blocks or walk up 10 steps.[11] Muscle wasting and sarcopenia[12,13,10] and reduced transfer, balance and walking abilities[12,11,14] may be the main reasons for this limitation, which conversely is the most consistently reported cause for reduced quality of life in this patient cohort.[15] Thus, interventions to increase mobility in HF are highly recommended.[16,17]
For this purpose, objective and robust outcome parameters are required. Mobility is usually assessed either with patient-reported outcomes (perception measures), or with clinical outcome assessments, such as the 6-min walk test, the Short Physical Performance Battery (SPPB), the Timed Up and Go Test (TUG) and timed walking tests.[18-21] For example, a study reported that SPPB scores differed significantly across NYHA classes in a sample of older hospital patients with HF (patients with less severe HF performed better).[22] However, these assessments were not designed to capture specific parameters of mobility. Recent developments in the area of digital mobility outcomes (DMOs), mainly with wearable inertial measurement units (IMUs), allow a more granular and detailed mobility assessment.[23-25] IMUs commonly consist of accelerometers, gyroscopes and magnetometers, and provide the opportunity to assess mobility aspects without the need for a dedicated laboratory.[26] The use of IMUs for the assessment of mobility parameters is feasible in a clinical and home setting and extracted parameters might reveal more information about mobility impairment than established clinical outcome assessments.[26,27]
To our knowledge, IMUs have never been used to quantify specific mobility parameters in patients with HF and it is unclear if DMOs are associated with disease severity in this population. Thus, this exploratory study aimed to assess the association between disease severity and DMOs as well as handgrip strength in patients with HF.[28]
Methods
Study Design and Setting
Data has been obtained from the ‘Cognitive and Motor Interactions in the Older Population’ (ComOn) trial,[28] an ongoing, prospective, explorative observational multi-center cohort study. Participants are geriatric inpatients recruited in hospitals in Europe. Details of the ComOn study have been reported recently.[28]
This report includes a sub-sample of geriatric inpatients admitted to the internal medicine ward of the Universitätsklinikum Schleswig-Holstein (UKSH), Campus Kiel, Germany. Ethical approval was provided from the ethics committee of the medical faculty of the University of Kiel (D427/17). The investigation conforms with the principles outlined in the Declaration of Helsinki (2013 version) and all participants provided written informed consent. Reporting is informed by the STROBE reporting guideline for cross-sectional studies.[29]
Participants
Inclusion criteria of the ComOn study are:[28] (a) inpatients aged ≥ 60 years with at least two chronic conditions; (b) the ability to stand without personal assistance for at least 10 s; and (c) to walk at least 3 m (walking aids permitted). Exclusion criteria are: (a) severe deficits in consciousness (clinical diagnosis); (b) > 2 falls during the previous week (risk of falling too high during the examination); (c) score of ≤ 5 points in the Montreal Cognitive Assessment (MoCA) test;[30] (d) history of or current drug abuse (except nicotine); and (e) (corrected) visual acuity below 60%. This study is based on data from inpatients submitted to the hospital between 11 January 2018 and 29 May 2019 (504 days, approximately 17 months). Participants received standard procedure for older inpatients in Germany, the so called “early rehabilitation in geriatric medicine” (“Geriatrische Frührehabilitative Komplexbehandlung”, GFK).[31] The GFK procedure includes, among others, the following aspects: care by a multidisciplinary team, comprehensive geriatric assessment, activating-therapeutic nursing care, regular physiotherapy, occupational therapy, and speech and language therapy. We included all participants with HF based on the medical diagnosis reported in the hospital discharge report. Participants with HF were classified according to the NYHA classes (class I to IV).[32,9] The clinical diagnosis of HF was made by experienced doctors with special training in internal medicine, cardiology and geriatric medicine. An additional convenience sample of participants without any known structural heart disease (e.g., coronary heart disease) and inconspicuous amino-terminal pro-brain natriuretic peptide (NT-proBNP) levels < 125 pg/mL was included in this analysis, to represent a group that has no HF-related limitation of physical activity. For every participant, the number of diagnoses according to the 10th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10), was recorded from the hospital report. In addition, the patients’ exact ICD-10 codes were grouped into the categories of diagnoses coded by the first three characters of the ICD-10 code to illustrate the multimorbidity of the present sample.
Procedures
Participants were recruited at hospital admission and assessed within the first two days and during the last two days before discharge of their inpatient stay (approximately 14–21 days). Participants received multidisciplinary care according to GFK procedures.
Outcome Measures
Outcome measures were derived from an extensive comprehensive geriatric assessment.[33] In addition, a detailed assessment of cognition and physical functioning was conducted. Clinical and demographical data were taken from the participants’ hospital records and additional semi-standardized clinical interviews. Gait and balance were assessed with two standardized performance-based clinical outcome assessments, the SPPB[34] and the TUG.[19] Both assessments were performed in a supervised environment (i.e., the hospital ward) on level ground. Participants wore solid footwear and the use of walking aids, such as canes or wheeled walkers, was permitted during the walking tasks if the participant was dependent on the walking aid in daily life. The SPPB is a measure of mobility and physical functioning, including objective tests of lower body motor functions: a hierarchical test of standing balance in 3 positions (feet closed, semi-tandem stance, tandem stance), a 4-m walk test (usual gait speed), and a 5-times chair rise test.[34] The SPPB has a scoring from 0 (unable) to 4 points for every sub-test, with a maximum of 12 points in total. Sufficient validity and reliability of the SPPB have been described for older people with and without cardiopulmonary diseases.[34,18,35,22] The TUG measures basic mobility functions. The participants are asked to stand up from a chair, walk 3 m, turn around, walk back, and return to the chair.[19] In the present study, participants were instructed to stand up without using their arms, if possible. In the first trial, the participant’s first step was with the left leg. In the second trial, the first step was with the right leg. The mean score of both trials in seconds was used as the final score. There was no familiarization trial. Shorter times indicate higher mobility capacity. There is evidence of sufficient reliability and construct validity of the TUG in people with HF.[36,37]
During the administration of the SPPB and the TUG, participants were equipped with a wearable IMU system (Rehagait®, Hasomed, Magdeburg, Germany) consisting of three IMUs worn at both ankles (lateral fixation) and at the lower back (L5). Each IMU contains a 3D accelerometer (± 16 g), a 3D gyroscope (± 2000 °/s), and a magnetometer (± 1.3 Gs), resulting in nine degrees of freedom. Based on IMU data recorded during SPPB and TUG administration, 17 different DMOs were calculated. An overview of those outcomes, according to the mobility-related activities, is given in Table 2. The selection of DMOs was based on the requirement to reflect mobility aspects of sit-to-stand, balance, and walking as broadly as possible, and to use parameters that have already been used for such measurements in other cohorts.[24,25,38]
Table 2. Handgrip strength and digital mobility outcome values at hospital admission and discharge.
| Admission | Discharge | |||||
| n | Mean ± SD | n | Mean ± SD | |||
| SD: standard deviation; SPPB: short physical performance battery. | ||||||
| Handgrip strength, kg Mobility-related activity |
Digital mobility outcome [unit] | 59 | 19.6 ± 7.8 | 47 | 19.4 ± 8.6 | |
| Static balance (based on SPPB semi-tandem stance position test performance) |
Area, m2/s5 | 46 | 0.0023 ± 0.0017 | 42 | 0.0027 ± 0.0035 | |
| Path, m/s2 | 46 | 2.30 ± 1.09 | 42 | 2.42 ± 1.27 | ||
| Acceleration, cm/s2 | 46 | 1.61 ± 0.56 | 42 | 1.68 ± 0.97 | ||
| Jerk, cm/s³ | 46 | 0.30 ± 0.31 | 42 | 0.32 ± 0.35 | ||
| Mean frequency, Hz | 46 | 2.03 + 0.35 | 42 | 2.04 ± 0.30 | ||
| Sit-to-stand (based on Timed Up and Go test performance) |
Total duration, s | 22 | 2.49 ± 0.77 | 26 | 2.57 ± 0.88 | |
| Total trunk flexion, ° | 22 | 40.93 ± 13.39 | 26 | 36.80 ± 12.55 | ||
| Maximum trunk flexion velocity, °/s | 22 | 129.91 ± 31.17 | 26 | 120.18 ± 43.75 | ||
| Maximum trunk extension velocity, °/s | 22 | 43.55 ± 15.02 | 26 | 42.00 ± 15.37 | ||
| Walking (based on SPPB 4-meter walk test performance) |
Gait speed, m/s | 52 | 0.48 ± 0.14 | 47 | 0.48 ± 0.11 | |
| Number of steps, n | 52 | 9.23 ± 3.25 | 47 | 9.02 ± 2.41 | ||
| Step-time, s | 52 | 0.72 ± 0.20 | 47 | 0.71 ± 0.16 | ||
| Step-time variability, SD (step time)/(m/s) | 52 | 0.25 ± 0.45 | 47 | 0.23 ± 0.30 | ||
Stance time/swing time ratio, 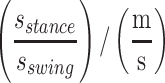
|
52 | 3.90 ± 1.62 | 47 | 3.66 ± 1.00 | ||
| Double support time, s/(m/s) | 52 | 0.46 ± 0.44 | 47 | 0.40 ± 0.20 | ||
| Double support time variability, SD (double support time)/(m/s) | 52 | 0.14 ± 0.31 | 47 | 0.09 ± 0.11 | ||
| Asymmetry index α, α/(m/s) | 47 | 0.20 ± 0.30 | 41 | 0.15 ± 0.17 | ||
The IMU data was processed by custom-written scripts using MATLAB (MathWorks, Nantick, MA) based on Mancini, et al.[39,40] For static balance, the collected outcomes provided information about the sway area [cm2/s5], path [m/s2], acceleration [cm/s2], jerk [cm/s3], and frequency [Hz] in a semi-tandem stance position.[39] Acceleration and jerk were expressed as the root mean square value and computed in both anterior-posterior (AP) and medio-lateral (ML) directions, as there is evidence that differences in both directions can represent different pathologies or compensation strategies of the body.[39,41] For sit-to-stand, we calculated total duration [s], total trunk flexion [°], maximum trunk flexion velocity [°/s], and maximum trunk extension velocity [°/s].[42] In addition, we calculated 8 different walking-related mobility outcomes: gait speed [m/s], number of steps [n], step-time [s] step-time variability [SD (step time)/(speed)], stance time/swing time ratio 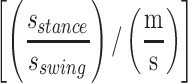 , double support time [s/(m/s)], double support time variability [SD (double support time)/(m/s)], asymmetry index α [α/(m/s)].[43]
, double support time [s/(m/s)], double support time variability [SD (double support time)/(m/s)], asymmetry index α [α/(m/s)].[43]
Handgrip strength in kg was measured with an hydraulic dynamometer (Saehan® Corporation Korea, Hydraulic Hand Dynamometer, Model SH5001) according to the protocol proposed by Roberts et al.[44] The highest score of 4 measures (each side two times) was used as the final score.
Statistical Analyses
For continuous data, descriptive statistics are presented as means and standard deviations. Categorical data are presented as frequencies (percentages). Data were analysed using SPSS 23.0 (IBM Corp., Armonk, New York, USA), JASP (University of Amsterdam, version 0.16), and MATLAB (MathWorks, Nantick, MA, USA). Sample characteristics are presented descriptively. Missing values are due to unavailability of patients or assessors, unplanned discharges, physical impairment/fatigue, and recording errors. Significance was set to P ≤ 0.05 in all analyses.
Multiple linear regression was used to estimate the association between the predictors (NYHA class, age, sex, walking aid use) and the outcome variables (DMOs, handgrip strength). The potential confounders age, sex, and walking aid use were included in the models since these variables are associated with the degree of physical impairment in this population.[45,46,12]
We assessed the following assumptions prior to model calculation:[47] normality (Shapiro-Wilk-test), homogeneity (Levene-test), and multicollinearity (Spearman’s rho). All assumptions were met. For each overall model, we report the coefficient of determination (R2), adjusted by the number of predictors.[48] Standardized beta coefficients (β), i.e., the regression coefficients referring to how many standard deviations the outcome variable will change, per standard deviation increase in the predictor variable, were reported to allow comparison of the strength of the effect of each individual predictor. We did not correct any P-values due to the exploratory character of the analyses.
RESULTS
During the study period, 61 older patients of the internal geriatric ward of the hospital were recruited and included in this analysis. Of those, 49 individuals were diagnosed with HF and 12 individuals presented without any known structural heart disease (heart-healthy sample). The mean age of participants was 82.2 ± 6.3 years (range: 65–97 years) and 34 participants (55.7%) were female.
Participants’ baseline characteristics are reported in Table 1. The medical diagnoses of the sample according to ICD-10 classification are listed in the Supplementary file 1. DMOs and handgrip strength at admission and discharge are presented in Table 2. The associations between the predictors (NYHA class, age, sex, walking aid) and the outcome variables (DMOs, handgrip strength) are reported in Table 3 for admission and discharge.
Table 1. Participant characteristics at hospital admission.
| Characteristic | Participants with HF (n = 49) |
Participants without any diagnosis of a structural heart disease (n = 12) |
Total sample (n = 61) |
|
| Data are presented as n (%) unless other indicated. *New York Heart Association classification system [The Criteria Committee of the New York Heart Association 1994].[11] Class I. Patients with cardiac disease but without resulting limitation of physical activity. Ordinary physical activity does not cause undue fatigue, palpitation, dyspnoea, or anginal pain. Class II. Patients with cardiac disease resulting in slight limitation of physical activity. They are comfortable at rest. Ordinary physical activity results in fatigue, palpitation, dyspnoea, or anginal pain. Class III. Patients with cardiac disease resulting in marked limitation of physical activity. They are comfortable at rest. Less than ordinary activity causes fatigue, palpitation, dyspnoea, or anginal pain. Class IV. Patients with cardiac disease resulting in inability to carry on any physical activity without discomfort. Symptoms of heart failure or the anginal syndrome may be present even at rest. If any physical activity is undertaken, discomfort increases. HF: heart failure; ICD-10: 10th revision of the International Statistical Classification of Diseases and Related Health Problems; IQR: interquartile range. | ||||
| Age in years, mean ± SD (range) | 83.1 ± 5.9 (65–97) | 78.6 ± 6.9 (70–88) | 82.2 ± 6.3 (65–97) | |
| Sex, females | 31 (63.3%) | 3 (25.0%) | 34 (55.7%) | |
| Number of ICD-10 diagnoses per participant, median (IQR) |
19 (14 – 22) | 18 (12 – 19) | 19 (16 – 25) | |
| *New York Heart Association class | ||||
| No diagnosis of HF | 0 | 12 (100%) | 12 (19.7%) | |
| I | 0 | 0 | 0 | |
| II | 26 (53.1%) | 0 | 26 (42.6%) | |
| III | 13 (26.5%) | 0 | 13 (21.3%) | |
| IV | 10 (20.4%) | 0 | 10 (16.4%) | |
| Walking aid | ||||
| None | 10 (20.4%) | 4 (33.3%) | 14 (23.0%) | |
| Cane | 5 (10.2%) | 0 | 5 (8.2%) | |
| Other | 27 (55.1%) | 6 (50.0%) | 33 (54.1%) | |
| Missing | 7 (14.3%) | 2 (16.7%) | 9 (14.8%) | |
Table 3. Results of the multiple linear regression models on the association between the independent variables (NYHA class, age, sex, walking aid use) and the dependent variables (grip strengths, digital mobility outcomes).
| Time | Overall model |
NYHA class |
Age | Sex | Walking aid use |
|||||||||||||
| n | R2
adjusted |
Regression equation F(df, residual) |
P−value | β | P−value | β | P−value | β | P−value | β | P−value | |||||||
| Age in years; sex: 0 for male and 1 for female; walking aid use: 0 for none and 1 for any walking aid use; β: standardized beta coefficient per one unit of the respective variable. R2: coefficient of determination; SPPB: Short Physical Performance Battery; NYHA = New York Heart Association. | ||||||||||||||||||
| Hand grip strength [kg] | Admission | 50 | 0.62 | F(4, 46) = 21.098 | < 0.01 | −0.15 | 0.10 | −0.21 | 0.03 | −0.68 | < 0.01 | −0.08 | 0.36 | |||||
| Discharge | 37 | 0.62 | F(4, 33) = 16.306 | < 0.01 | −0.15 | 0.19 | −0.30 | 0.01 | −0.55 | < 0.01 | −0.03 | 0.78 | ||||||
| Mobility-related activity |
Digital mobility outcome | |||||||||||||||||
| Static balance (based on SPPB semi-tandem stance position test performance) |
Area, m2/s5 | Admission | 45 | −0.03 | F(4, 41) = 0.633 | 0.64 | −0.05 | 0.75 | 0.25 | 0.14 | −0.04 | 0.81 | −0.06 | 0.71 | ||||
| Discharge | 41 | −0.09 | F(4, 37) = 0.132 | 0.97 | −0.07 | 0.70 | −0.01 | 0.96 | 0.12 | 0.52 | 0.00 | 0.98 | ||||||
| Path, m/s2 | Admission | 45 | 0.01 | F(4, 41) = 0.921 | 0.46 | −0.28 | 0.09 | 0.11 | 0.49 | 0.10 | 0.52 | −0.07 | 0.66 | |||||
| Discharge | 41 | 0.09 | F(4, 37) = 1.958 | 0.12 | −0.39 | 0.02 | 0.00 | 1.00 | −0.08 | 0.64 | 0.14 | 0.37 | ||||||
| Acceleration, m/s2 | Admission | 45 | 0.00 | F(4, 41) = 1.002 | 0.42 | −0.07 | 0.65 | 0.27 | 0.10 | −0.06 | 0.72 | −0.15 | 0.34 | |||||
| Discharge | 41 | −0.08 | F(4, 37) = 0.213 | 0.93 | −0.10 | 0.57 | 0.00 | 1.00 | 0.13 | 0.48 | −0.08 | 0.65 | ||||||
| Jerk, cm/s³ | Admission | 45 | −0.03 | F(4, 41) = 0.706 | 0.59 | −0.15 | 0.37 | 0.25 | 0.13 | 0.00 | 0.99 | −0.03 | 0.84 | |||||
| Discharge | 41 | −0.06 | F(4, 37) = 0.379 | 0.82 | −0.20 | 0.24 | 0.02 | 0.91 | 0.01 | 0.94 | 0.05 | 0.77 | ||||||
| Mean frequency, Hz | Admission | 45 | −0.03 | F(4, 41) = 0.721 | 0.58 | −0.01 | 0.97 | 0.11 | 0.51 | −0.26 | 0.12 | 0.05 | 0.74 | |||||
| Discharge | 41 | −0.05 | F(4, 37) = 0.496 | 0.74 | 0.05 | 0.75 | 0.10 | 0.57 | −0.23 | 0.20 | −0.03 | 0.86 | ||||||
| Sit-to-stand (based on Timed Up and Go test performance) |
Total duration, s | Admission | 21 | 0.06 | F(4,17) = 1.339 | 0.30 | 0.11 | 0.64 | −0.20 | 0.37 | 0.22 | 0.36 | 0.35 | 0.12 | ||||
| Discharge | 25 | 0.25 | F(4, 21) = 3.124 | 0.04 | 0.03 | 0.86 | 0.17 | 0.37 | 0.44 | 0.03 | 0.31 | 0.09 | ||||||
| Total trunk flexion, ° | Admission | 21 | 0.41 | F(4,17) = 4.623 | 0.01 | −0.19 | 0.30 | −0.15 | 0.40 | 0.48 | 0.02 | −0.48 | 0.01 | |||||
| Discharge | 25 | 0.02 | F(4, 21) = 1.128 | 0.37 | 0.02 | 0.92 | 0.32 | 0.14 | 0.06 | 0.79 | −0.24 | 0.24 | ||||||
| Maximum trunk flexion velocity, °/s | Admission | 21 | 0.03 | F(4,17) = 1.139 | 0.37 | −0.23 | 0.34 | 0.16 | 0.48 | 0.44 | 0.08 | −0.06 | 0.80 | |||||
| Discharge | 25 | −0.08 | F(4, 21) = 0.522 | 0.72 | 0.03 | 0.90 | 0.28 | 0.23 | −0.14 | 0.53 | −0.11 | 0.60 | ||||||
| Maximum trunk extension velocity, °/s | Admission | 21 | −0.25 | F(4,17) = 2.719 | 0.06 | 0.24 | 0.26 | −0.13 | 0.50 | 0.02 | 0.93 | −0.60 | < 0.01 | |||||
| Discharge | 25 | −0.19 | F(4, 21) = 2.429 | 0.08 | 0.06 | 0.77 | −0.23 | 0.24 | −0.01 | 0.69 | −0.49 | 0.01 | ||||||
| Walking (based on SPPB 4-meter walk test performance) |
Gait speed, m/s | Admission | 51 | 0.22 | F(4, 47) = 4.602 | < 0.01 | 0.19 | 0.16 | 0.07 | 0.59 | −0.24 | 0.07 | −0.49 | < 0.01 | ||||
| Discharge | 46 | 0.13 | F(4, 42) = 2.648 | 0.05 | 0.04 | 0.79 | −0.13 | 0.40 | −0.17 | 0.28 | −0.37 | 0.01 | ||||||
| Number of steps | Admission | 51 | −0.01 | F(4, 47) =0.925 | 0.46 | −0.22 | 0.14 | 0.02 | 0.89 | 0.13 | 0.40 | 0.19 | 0.19 | |||||
| Discharge | 46 | −0.01 | F(4, 42) = 0.892 | 0.48 | −0.07 | 0.66 | 0.19 | 0.24 | 0.13 | 0.44 | 0.11 | 0.45 | ||||||
| Step-time, s | Admission | 51 | 0.09 | F(4, 47) = 2.289 | 0.07 | 0.04 | 0.78 | −0.22 | 0.13 | 0.01 | 0.96 | 0.33 | 0.02 | |||||
| Discharge | 46 | 0.07 | F(4, 42) = 1.864 | 0.14 | 0.02 | 0.88 | −0.20 | 0.21 | 0.07 | 0.64 | 0.33 | 0.03 | ||||||
| Step-time variability, SD (step time)/(m/s) | Admission | 51 | −0.06 | F(4, 47) = 0.286 | 0.89 | −0.02 | 0.92 | −0.05 | 0.75 | −0.06 | 0.70 | 0.12 | 0.41 | |||||
| Discharge | 46 | −0.02 | F(4, 42) = 0.777 | 0.55 | −0.04 | 0.79 | −0.06 | 0.69 | 0.24 | 0.15 | 0.12 | 0.45 | ||||||
Stance time/swing time ratio, 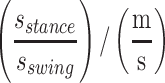
|
Admission | 51 | 0.05 | F(4, 47) = 1.653 | 0.18 | −0.13 | 0.38 | −0.07 | 0.64 | 0.10 | 0.48 | 0.33 | 0.02 | |||||
| Discharge | 46 | 0.04 | F(4, 42) = 1.526 | 0.21 | 0.00 | 1.00 | 0.05 | 0.77 | 0.14 | 0.40 | 0.30 | 0.05 | ||||||
| Double support time, s/(m/s) |
Admission | 51 | −0.02 | F(4, 47) = 0.732 | 0.58 | −0.03 | 0.85 | −0.07 | 0.64 | −0.02 | 0.92 | 0.23 | 0.12 | |||||
| Discharge | 46 | 0.03 | F(4, 42) = 1.315 | 0.28 | 0.02 | 0.92 | −0.08 | 0.59 | 0.11 | 0.51 | 0.30 | 0.05 | ||||||
| Double support time variability, SD (double support time)/(m/s) |
Admission | 51 | −0.05 | F(4, 47) = 0.365 | 0.83 | −0.04 | 0.78 | −0.03 | 0.86 | −0.15 | 0.34 | 0.04 | 0.80 | |||||
| Discharge | 46 | −0.08 | F(4, 42) = 0.161 | 0.96 | −0.06 | 0.73 | −0.02 | 0.90 | 0.13 | 0.45 | 0.02 | 0.92 | ||||||
| Asymmetry index α, α/(m/s) |
Admission | 46 | −0.03 | F(4, 42) = 0.705 | 0.59 | −0.13 | 0.43 | −0.04 | 0.81 | −0.09 | 0.57 | 0.18 | 0.26 | |||||
| Discharge | 40 | 0.02 | F(4, 36) = 1.188 | 0.33 | 0.11 | 0.50 | −0.03 | 0.86 | 0.24 | 0.18 | 0.15 | 0.36 | ||||||
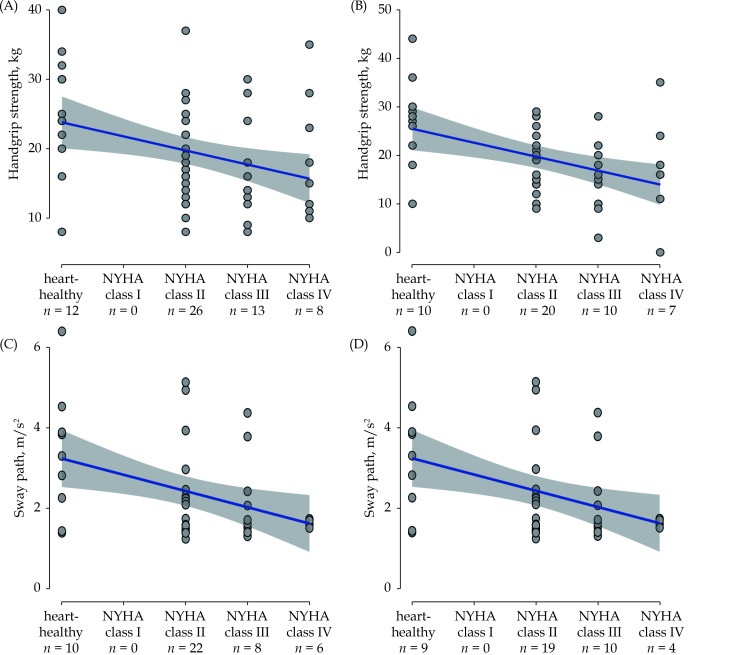
The following overall regression models including DMOs/hand grip strength were significant (Table 3): gait speed at admission and discharge, sit-to-stand total duration at discharge, and total trunk flexion at admission. However, for none of these parameters did the NYHA class show a significant association with the respective DMO. Rather, sex (for sit-to-stand total duration and total trunk flexion, and tendentially also for gait speed) and use of a walking aid (for gait speed) were found to be significantly contributing variables to the overall model. Use of a walking aid had an influence on the overall model for some DMOs. For handgrip strength, the overall model was also statistically significant at admission and discharge, with the highest variance resolutions observed in our analyses, respectively. The NYHA classes tended to contribute to this effect, especially at admission. Moreover, sex and age contributed relevantly to the overall model. Associations of handgrip strength with NYHA classes at admission and discharge are presented in Figure 1A and Figure 1B, respectively. Although not significant in the overall model, a notable association was shown between the following DMO and NYHA class. At admission, sway path showed a trend towards significance with NYHA classes. This association was significant at discharge (Figure 1C and Figure 1D, respectively). All other dependent outcome variables did not show relevant associations with NYHA classes (Table 3).
Figure 1.
Associations between handgrip strength/sway path and New York Heart Association (NYHA) classes.
(A): Handgrip strength and NYHA classes at admission (β = –0.15, P = 0.10); (B): handgrip strength and NYHA classes at discharge (β = –0.15, P = 0.19); (C): sway path and NYHA classes at admission (β = –0.28, P = 0.09); and (D): sway path and NYHA classes at discharge (β = –0.39, P = 0.02).
DISCUSSION
The main finding of this exploratory study in geriatric patients with HF is that regression models including NYHA class, age, sex, and use of walking aid showed significant associations with some DMOs and hand grip strength, suggesting that the (digital) assessment of mobility aspects could aid relevant information to presence and severity of HF in older adults. Moreover, static sway (more specifically, sway path) and hand grip strength turned out to be the most promising quantitative parameters associated with HF in our cohort.
To our knowledge, this work is the first to examine the above associations both before and after inpatient optimization of therapy. The results show that the existing associations between NYHA and DMOs hold for both assessment times. We are not aware of any other study that used IMUs to quantify specific mobility aspects in individual with HF.[49] Thus, comparability with existing evidence is limited. However, some studies have used at least partially comparable methods[50] which will be discussed in the following.
Based on the multiple linear regression models, sway path [m/s2] was the DMO most strongly associated with NYHA classes at admission and discharge, decreasing with higher NYHA classes. Postural sway is the seemingly random movement around the center of mass of a body during stance. Typically, less postural sway is considered to indicate higher stability and better balance ability,[40,51] and increased sway is considered a risk factor for falls in older adults.[52] Therefore, our result is surprising at first view. However, it could be that severely affected patients with HF, similar to severely affected patients with Parkinson’s disease,[53] reduce their movements around the center of mass, therefore reducing the risk of moving the body to the limit of stability and thus avoiding fall risk.[54] In line with our findings, a previous study found that patients with HF showed reduced maximum excursion in forward and backward directions compared with healthy controls.[14]
Interestingly, a recent meta-analysis showed a calculated decrease of falls and fall-related injuries by > 40% in controlled trials that performed gait and balance training at the limit of stability.[55,56] Although there is evidence from a systematic review that the fall rate in individuals with HF is higher than in community-dwelling older adults,[57] there are unfortunately only a few studies focusing on the balance abilities in patients with HF.[14] Our results may be a cautionary signal that in HF, as in many other conditions, balance training at the limit of stability is effective in reducing disease-related mobility deficits.
Besides static sway performance, sit-to-stand movements are inevitable prerequisites for successful performance of the activities of daily living[58] and chair-rise impairment is frequently reported by individuals with HF.[10] There was no statistically significant association between any of the four transfer-related DMOs and NYHA classes to observe in our study cohort. This is a surprising result as it can be assumed that increasing HF is associated with decreasing muscle strength,[59] and this limitation becomes relevant especially during sit-to-stand transfer. We collected the movement in the context of the TUG (at self-selected speed), and assume that by increased demand (e.g., as fast as possible, or repeated standing up) the expected effect would rather have been observed. For that purpose, the application of a repeated instrumented sit-to-stand test, for example the five times chair rise test,[60] seems particularly promising. This is supported by a recent study, which compared the durations recorded with an instrumented repeated sit-to-stand performance with manually recorded test durations in a sample of 63 older participants.[61] The instrumented sit-to-stand durations were more strongly associated with participant health status, functional status, and physical activity, indicating added value in clinical testing of older adults.
In our regression analyses, no walking-related DMO was significantly influenced by NHYA class, sex or age. This result is surprising since evidence from systematic reviews suggests that walking capacity, as measured with the 6-min walk test, is influenced by the NYHA classes in patients with HF, that older females walk significantly slower than men, and that gait speed decreases with age.[62-64] The following reasons might explain why our findings deviate from the results of those studies. Firstly, mobility capacity and walking speed was not assessed with IMUs in the other studies and DMOs are not directly comparable with mobility outcomes of “classical” clinical outcome assessments.[25] Secondly, walking was measured over a short distance in our study; longer distances for test administration can impact mobility parameters, such as gait speed.[65] Thirdly, about two third of our HF patients used a walking aid for ambulation. This rate is much higher than in other studies (e.g., Lee, et al., 2020)[66], indicating relatively severe physical impairment in our participants. We addressed this issue by including the ‘walking aid use’ predictor in the regression models since a significant impact of walking aids on gait parameters in various clinical populations has been described.[67,68] Gait speed has been used frequently to measure mobility performance in older adults and in patients with HF.[69] This parameter has prognostic value for various health outcomes in patients with HF, including disability, hospitalizations, and mortality.[70-72,50] For example, a recent meta-analysis found that patients with HF who showed a slower gait speed had a higher risk of all-cause mortality than those patients who showed a faster gait speed [hazard ratio = 1.49, 95% CI: 1.24–1.79].[50] In another review, gait speed was reported consistently different between known groups, such as patients with Parkinson’s disease or Multiple Sclerosis and healthy controls [25]. In our sample, mean gait speed was 0.48 m/s, which is substantially slower than gait speeds of healthy adults aged ≥ 80 years (0.95 m/s)[63] and patients with acute HF in other studies (e.g., 0.74 m/s at admission and 0.98 m/s at discharge).[73] Thus, the cohort presented here might be more severely affected than other cohorts. Severely affected (sub)cohorts are often excluded in studies and trials, which should be avoided if possible due to the often-different results (and then also different therapy options). Future research should thus examine mobility aspects, as measured with DMOs, in relation to NYHA classes in an as diverse as possible cohort of patients with HF. These studies should report key information on test administration and data collection in detail, including the measurement method (e.g., wearable sensor), setting (e.g., clinic or home), walking bout lengths (e.g., short distance or real-world), and other aspects (e.g., habitual, fast or real-world walking performance).[25,65,74]
Handgrip strength has prognostic value for negative health outcomes in patients with HF, including postoperative complications, cardiac adverse events, and mortality.[75-77] Moreover, handgrip strength plays a key role in the assessment and diagnosis of frailty and sarcopenia.[78,79] Concerning the association between handgrip strength and HF severity, two previous studies reported that handgrip strength decreased with increases in NYHA classes in patients with HF.[59,80] A further recent study with 500 inpatients with HF showed that handgrip strength was independently associated with HF severity, even after adjustment for sex and age (risk ratio per 1 kg increase for NYHA class III/IV: 0.98, 95% CI: 0.97–0.99).[81] We found comparable results. Since handgrip strength has a predictive validity for decline in cognition, mobility and functional status as well as for mortality,[82] the routine assessment of handgrip strength in older patients with HF is recommended to identify individuals at high risk, to identify those patients who may benefit from interventions to improve muscle strength and function, and to monitor such interventions.[83] More research will help to better understand how handgrip strength is influenced by disease severity in hospitalized patients with HF. We suggest to further explore this parameter with digital assessment tools. In addition, repeated and rapidly performed handgrip strength assessments may be useful to assess, for example, muscular fatigability aspects.[84]
Strengths and Limitations
The study sample included inpatients with HF and heart-healthy individuals from a geriatric ward.[28] Our sampling strategy, including very broad inclusion and only a few exclusion criteria, aims to produce a representative sample of geriatric inpatients. To our knowledge, although the sample is relatively small, this is the first analysis on the association of disease severity and IMU-based DMOs in patients with HF. A broad set of 17 different DMOs was extracted in this study, including some of the most frequently assessed parameters in common medical conditions of older people such as gait speed, step time, step time variability, and step count.[25] These mobility outcomes were derived from the performance of two standardized clinical outcomes assessments (SPPB, TUG) and based on three central mobility activities (balancing, sit-to-stand transfer, walking). This is an explanatory study aimed to generate hypotheses for further research. There might be selection bias since participants with (very advanced) HF might have been excluded due to the requirement of walking ability of at least 10 m. The measurement of DMOs based on standardized clinical outcome assessments (e.g., TUG, SPPB) is very difficult in individuals with pronounced physical and/or cognitive impairment,[85,86] who are thus excluded from the ComOn study. However, 38% of participants were classified with NYHA III or IV, gait speed was very low, and most participants used a walking aid, indicating a relatively impaired cohort sample. Furthermore, it is possible that some participants in the heart-healthy sample might suffer from undiagnosed HF. Many patients with HF of NYHA class I do not exhibit symptoms attributable to heart disease and therefore remain undiagnosed until symptoms occur.[7] However, we carefully checked hospital patient records and NT-proBNP levels to ensure that no patients with HF were included in the heart-healthy sample. This study focused on the general impact of HF on mobility. Future studies may also investigate the impact of preserved versus reduced ejection fraction.
Another reason for caution when interpreting the present results is the instruction on test performance used in this study. Participants were allowed to use their arms and the arm rests or a walking aid to rise up from the chair, subjected to their physical abilities and individual preferences. This procedure is not in agreement with the instruction of a standardized repeated chair rise test (e.g., 5-times or 30-s chair rise test),[60,87] but closer to the performance in everyday life. Since joint movements and shift in the center-of-gravity during sit-to-stand movement is highly affected by the use of the upper limbs,[88,89] the present findings might be biased and need further investigation.
Another aspect that should be considered when interpreting our results is the selection of DMOs. Although we included 17 parameters, some other mobility parameters that have been used in other conditions might be informative as well in patients with HF, such as stride length, cadence, turn duration and turn velocity.[25,38] Balance-related outcomes were derived from the semi-tandem stance position only and outcomes from other positions, such as a more challenging tandem-stance, might be used in further studies to better understand the association of balance abilities and NYHA class. Walking-related outcomes were based on a short distance walk. Real-world walking bouts or data from longer distances might provide different information.[25] The outcomes used in this study are most probably also influenced by factors not included in the regression models, such as nutritional status, comorbidities, cognition, medication, daytime of the examination, fatigue, motivation, or exercise capacity of the participants, and from factors that we simply do not know according to the current state of knowledge.
Another limitation may be the analysis approach. The measurement properties of most DMOs, including validity, reliability and responsiveness to change, are insufficiently examined and mostly unclear.[38,25] This is especially true for patients with HF. To our knowledge, the key measurement properties of IMU-based mobility parameters have never been reported for this population. A recent review concluded that some walking-related DMOs, including gait speed and step length, exhibited consistent evidence of validity and responsiveness in multiple conditions, but evidence was inconsistent or lacking for other DMOs.[25] Our group has recently assessed the day-to-day reliability of eight static balance parameters using IMUs in a neurogeriatric cohort.[90] We found only moderate to poor relative and absolute reliability. More research is needed to establish the measurement properties of DMOs in various clinical conditions before we can encourage an uptake into clinical practice or clinical trials.[25,91,38] Extensive attempt to develop, validate and approve DMOs in various health conditions, including Parkinson’s disease and HF, are currently performed by the Mobilise-D and IDEA-FAST projects, both being multidisciplinary consortia that aim to stimulate widespread adoption of DMOs.[38,92]
Conclusions
This explorative study suggests that particularly sway path and handgrip strength are associated with HF disease severity in geriatric patients. Particularly DMOs measuring capacity aspects might be examined as potential biomarkers for the presence, severity, progression and treatability of HF.[91] Future studies should also consider an evaluation of mobility in the place where it is really needed by patients: the usual environment.
SUPPLEMENTARY DATA
Supplementary data to this article can be found online.
ACKNOWLEDGMENTS
We would like to thank the participants for their valuable time and the administrative and technical support of the university.
References
- 1.Edelmann F Facts and numbers on epidemiology and pharmacological treatment of heart failure with preserved ejection fraction. ESC Heart Fail. 2015;2:41–45. doi: 10.1002/ehf2.12037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Störk S, Handrock R, Jacob J, et al Epidemiology of heart failure in Germany: a retrospective database study. Clin Res Cardiol. 2017;106:913–922. doi: 10.1007/s00392-017-1137-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ho KKL, Pinsky JL, Kannel WB, Levy D The epidemiology of heart failure: the Framingham Study. J Am Coll Cardiol. 1993;22(4S1):A6–A13. doi: 10.1016/0735-1097(93)90455-a. [DOI] [PubMed] [Google Scholar]
- 4.Benjamin EJ, Muntner P, Alonso A, et al Heart disease and stroke statistics-2019 update: a report from the american heart association. Circulation. 2019;139:e56–e528. doi: 10.1161/CIR.0000000000000659. [DOI] [PubMed] [Google Scholar]
- 5.Roger VL Epidemiology of heart failure. Circ Res. 2013;113:646–659. doi: 10.1161/CIRCRESAHA.113.300268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mosterd A, Hoes AW Clinical epidemiology of heart failure. Heart. 2007;93:1137–1146. doi: 10.1136/hrt.2003.025270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ponikowski P, Voors AA, Anker SD, et al 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37:2129–2200. doi: 10.1093/eurheartj/ehw128. [DOI] [PubMed] [Google Scholar]
- 8.Criteria Committee of the New York Heart Association. Nomenclature and criteria for diagnosis of diseases of the heart and great vessels; 9th Edition; Brown and Company; Boston, MA, USA; 1994.
- 9.Miller-Davis C, Marden S, Leidy NK. The New York Heart Association Classes and functional status: what are we really measuring? Heart Lung 2006; 35: 217-224.
- 10.Zhang Y, Zhang J, Ni W, et al Sarcopenia in heart failure: a systematic review and meta-analysis. ESC Heart Fail. 2021;8:1007–1017. doi: 10.1002/ehf2.13255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wong CY, Chaudhry SI, Desai MM, Krumholz HM Trends in comorbidity, disability, and polypharmacy in heart failure. Am J Med. 2011;124(2):136–143. doi: 10.1016/j.amjmed.2010.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Churilla JR, Richardson MR, Pinkstaff SO, et al Associations between heart failure and physical function in US adults. QJM. 2016;109:669–674. doi: 10.1093/qjmed/hcw042. [DOI] [PubMed] [Google Scholar]
- 13.Fülster S, Tacke M, Sandek A, et al Muscle wasting in patients with chronic heart failure: results from the studies investigating co-morbidities aggravating heart failure (SICA-HF) Eur Heart J. 2013;34:512–519. doi: 10.1093/eurheartj/ehs381. [DOI] [PubMed] [Google Scholar]
- 14.Tanriverdi A, Kahraman BO, Ozsoy I, et al Balance performance in patients with heart failure. Heart Lung. 2020;49:458–462. doi: 10.1016/j.hrtlng.2020.04.004. [DOI] [PubMed] [Google Scholar]
- 15.Jaarsma T, Johansson P, Agren S, Strömberg A Quality of life and symptoms of depression in advanced heart failure patients and their partners. Curr Opin Support Palliat Care. 2010;4:233–237. doi: 10.1097/SPC.0b013e328340744d. [DOI] [PubMed] [Google Scholar]
- 16.Sagar VA, Davies EJ, Briscoe S, et al Exercise-based rehabilitation for heart failure: systematic review and meta-analysis. Open Heart. 2015;2:e000163. doi: 10.1136/openhrt-2014-000163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Knosalla C, Gummert J, Edelmann F, et al Nationale Versorgungs Leitlinie Chronische Herzinsuffizienz. Zeitschrift für Herz-, Thorax-und Gefä ß chirurgie. 2018;32:381–390. [Google Scholar]
- 18.Guralnik JM, Ferrucci L, Simonsick EM, et al Lower-extremity function in persons over the age of 70 years as a predictor of subsequent disability. N Engl J Med. 1995;332:556–561. doi: 10.1056/NEJM199503023320902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Podsiadlo D, Richardson S The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39:142–148. doi: 10.1111/j.1532-5415.1991.tb01616.x. [DOI] [PubMed] [Google Scholar]
- 20.Abellan van Kan G, Rolland Y, Andrieu S, et al Gait speed at usual pace as a predictor of adverse outcomes in community-dwelling older people an International Academy on Nutrition and Aging (IANA) Task Force. J Nutr Health Aging. 2009;13:881–889. doi: 10.1007/s12603-009-0246-z. [DOI] [PubMed] [Google Scholar]
- 21.Yamamoto S, Matsunaga A, Wang G, et al Effect of balance training on walking speed and cardiac events in elderly patients with ischemic heart disease. Int Heart J. 2014;55:397–403. doi: 10.1536/ihj.14-017. [DOI] [PubMed] [Google Scholar]
- 22.Chiarantini D, Volpato S, Sioulis F, et al Lower extremity performance measures predict long-term prognosis in older patients hospitalized for heart failure. J Card Fail. 2010;16:390–395. doi: 10.1016/j.cardfail.2010.01.004. [DOI] [PubMed] [Google Scholar]
- 23.Kluge F, Del Din S, Cereatti A, et al Consensus based framework for digital mobility monitoring. PLoS One. 2021;16:e0256541. doi: 10.1371/journal.pone.0256541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Polhemus AM, Bergquist R, Bosch de Basea M, et al Walking-related digital mobility outcomes as clinical trial endpoint measures: protocol for a scoping review. BMJ Open. 2020;10:e038704. doi: 10.1136/bmjopen-2020-038704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Polhemus A, Ortiz LD, Brittain G, et al Walking on common ground: a cross-disciplinary scoping review on the clinical utility of digital mobility outcomes. NPJ Digit Med. 2021;4:1–14. doi: 10.1038/s41746-020-00373-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bernhard FP, Sartor J, Bettecken K, et al Wearables for gait and balance assessment in the neurological ward - study design and first results of a prospective cross-sectional feasibility study with 384 inpatients. BMC Neurol. 2018;18:114. doi: 10.1186/s12883-018-1111-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Maetzler W, Rochester L, Bhidayasiri R, et al Modernizing daily function assessment in parkinson’s disease using capacity, perception, and performance measures. Mov Disord. 2021;36:76–82. doi: 10.1002/mds.28377. [DOI] [PubMed] [Google Scholar]
- 28.Geritz J, Maetzold S, Steffen M, et al Motor, cognitive and mobility deficits in 1000 geriatric patients: protocol of a quantitative observational study before and after routine clinical geriatric treatment - the ComOn-study. BMC Geriatr. 2020;20:45. doi: 10.1186/s12877-020-1445-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Elm E von, Altman DG, Egger M, et al The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61:344–349. doi: 10.1016/j.jclinepi.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 30.Nasreddine ZS, Phillips NA, Bédirian V, et al The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695–699. doi: 10.1111/j.1532-5415.2005.53221.x. [DOI] [PubMed] [Google Scholar]
- 31.Kolb G, Breuninger K, Gronemeyer S, et al Jahre geriatrische frührehabilitative Komplexbehandlung im DRG-System. Z Gerontol Geriatr. 2014;47:6–12. doi: 10.1007/s00391-013-0592-7. [DOI] [PubMed] [Google Scholar]
- 32.Arroll B, Doughty R, Andersen V Investigation and management of congestive heart failure. BMJ. 2010;341:c3657. doi: 10.1136/bmj.c3657. [DOI] [PubMed] [Google Scholar]
- 33.Ellis G, Gardner M, Tsiachristas A, et al Comprehensive geriatric assessment for older adults admitted to hospital. Cochrane Database Syst Rev. 2017;9:CD006211. doi: 10.1002/14651858.CD006211.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guralnik JM, Simonsick EM, Ferrucci L, et al A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:85–94. doi: 10.1093/geronj/49.2.m85. [DOI] [PubMed] [Google Scholar]
- 35.Puthoff ML Outcome measures in cardiopulmonary physical therapy: short physical performance battery. Cardiopulm Phys Ther J. 2008;19:17–22. doi: 10.1097/01823246-200819010-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chun S, Shin DW, Han K, et al The timed up and go test and the ageing heart: Findings from a national health screening of 1, 084, 875 community-dwelling older adults. Eur J Prev Cardiol. 2021;28:213–219. doi: 10.1177/2047487319882118. [DOI] [PubMed] [Google Scholar]
- 37.Hwang R, Morris NR, Mandrusiak A, et al Timed up and go test: a reliable and valid test in patients with chronic heart failure. J Card Fail. 2016;22:646–650. doi: 10.1016/j.cardfail.2015.09.018. [DOI] [PubMed] [Google Scholar]
- 38.Rochester L, Mazzà C, Mueller A, et al. A roadmap to inform development, validation and approval of digital mobility outcomes: the mobilise-d approach. Digit Biomark 2020; 4(Suppl 1): S13-S27.
- 39.Mancini M, Horak FB, Zampieri C, et al Trunk accelerometry reveals postural instability in untreated Parkinson’s disease. Parkinsonism & Related Disorders. 2011;17:557–562. doi: 10.1016/j.parkreldis.2011.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mancini M, Salarian A, Carlson-Kuhta P, et al ISway: a sensitive, valid and reliable measure of postural control. J NeuroEngineering Rehabil. 2012;9:59. doi: 10.1186/1743-0003-9-59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Spain RI, St George RJ, Salarian A, et al Body-worn motion sensors detect balance and gait deficits in people with multiple sclerosis who have normal walking speed. Gait Posture. 2012;35:573–578. doi: 10.1016/j.gaitpost.2011.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pham MH, Warmerdam E, Elshehabi M, et al Validation of a Lower Back “Wearable”-based sit-to-stand and stand-to-sit algorithm for patients with Parkinson’s disease and older adults in a home-like environment. Front Neurol. 2018;9:652. doi: 10.3389/fneur.2018.00652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pham MH, Elshehabi M, Haertner L, et al Validation of a step detection algorithm during straight walking and turning in patients with Parkinson’s disease and older adults using an inertial measurement unit at the lower back. Front Neurol. 2017;8:457. doi: 10.3389/fneur.2017.00457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Roberts H, Denison H, Martin H, et al A review of the measurement of grip strength in clinical and epidemiological studies: towards a standardised approach. Age Ageing. 2011;40:423–429. doi: 10.1093/ageing/afr051. [DOI] [PubMed] [Google Scholar]
- 45.Dontje ML, van der Wal MHL, Stolk RP, et al Daily physical activity in stable heart failure patients. J Cardiovasc Nurs. 2014;29:218–226. doi: 10.1097/JCN.0b013e318283ba14. [DOI] [PubMed] [Google Scholar]
- 46.Duscha BD, Annex BH, Green HJ, et al Deconditioning fails to explain peripheral skeletal muscle alterations in men with chronic heart failure. J Am Coll Cardiol. 2002;39:1170–1174. doi: 10.1016/S0735-1097(02)01740-0. [DOI] [PubMed] [Google Scholar]
- 47.Hickey GL, Dunning J, Seifert B, et al Statistical and data reporting guidelines for the European Journal of Cardio-Thoracic Surgery and the Interactive CardioVascular and Thoracic Surgery. Eur J Cardiothorac Surg. 2015;48:180–193. doi: 10.1093/ejcts/ezv168. [DOI] [PubMed] [Google Scholar]
- 48.Wherry RJ A new formula for predicting the shrinkage of the coefficient of multiple correlation. The annals of mathematical statistics. 1931;2:440–457. doi: 10.1214/aoms/1177732951. [DOI] [Google Scholar]
- 49.Keogh A, Argent R, Anderson A, et al Assessing the usability of wearable devices to measure gait and physical activity in chronic conditions: a systematic review. J Neuroeng Rehabil. 2021;18:138. doi: 10.1186/s12984-021-00931-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fuentes-Abolafio IJ, Stubbs B, Pérez-Belmonte LM, et al Physical functional performance and prognosis in patients with heart failure: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2020;20:512. doi: 10.1186/s12872-020-01725-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Qiu H, Xiong S Center-of-pressure based postural sway measures: Reliability and ability to distinguish between age, fear of falling and fall history. Int J Ind Ergon. 2015;47:37–44. doi: 10.1016/j.ergon.2015.02.004. [DOI] [Google Scholar]
- 52.Johansson J, Nordström A, Gustafson Y, et al Increased postural sway during quiet stance as a risk factor for prospective falls in community-dwelling elderly individuals. Age Ageing. 2017;46:964–970. doi: 10.1093/ageing/afx083. [DOI] [PubMed] [Google Scholar]
- 53.Mancini M, Carlson-Kuhta P, Zampieri C, et al Postural sway as a marker of progression in Parkinson’s disease: a pilot longitudinal study. Gait Posture. 2012;36:471–476. doi: 10.1016/j.gaitpost.2012.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Creath R, Kiemel T, Horak F, et al A unified view of quiet and perturbed stance: simultaneous co-existing excitable modes. Neurosci Lett. 2005;377:75–80. doi: 10.1016/j.neulet.2004.11.071. [DOI] [PubMed] [Google Scholar]
- 55.Nørgaard JE, Jorgensen MG, Ryg J, et al Effects of gait adaptability training on falls and fall-related fractures in older adults: a systematic review and meta-analysis. Age Ageing. 2021;50:1914–1924. doi: 10.1093/ageing/afab105. [DOI] [PubMed] [Google Scholar]
- 56.Becker C Shake, rattle and get moving again. Age Ageing. 2021;50:1889–1890. doi: 10.1093/ageing/afab187. [DOI] [PubMed] [Google Scholar]
- 57.Lee K, Pressler SJ, Titler M Falls in patients with heart failure: a systematic review. J Cardiovasc Nurs. 2016;31:555–561. doi: 10.1097/JCN.0000000000000292. [DOI] [PubMed] [Google Scholar]
- 58.Gill TM. Assessment of function and disability in longitudinal studies. J Am Geriatr Soc 2010; 58(Suppl 2): S308-312.
- 59.Izawa KP, Watanabe S, Yokoyama H, et al Muscle strength in relation to disease severity in patients with congestive heart failure. Am J Phys Med Rehabil. 2007;86:893–900. doi: 10.1097/PHM.0b013e318154b592. [DOI] [PubMed] [Google Scholar]
- 60.Silva PF, Quintino LF, Franco J, Faria C Measurement properties and feasibility of clinical tests to assess sit-to-stand/stand-to-sit tasks in subjects with neurological disease: a systematic review. Braz J Phys Ther. 2014;18:99–110. doi: 10.1590/S1413-35552012005000155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.van Lummel RC, Walgaard S, Maier AB, et al The Instrumented Sit-to-Stand Test (iSTS) has greater clinical relevance than the manually recorded sit-to-stand test in older adults. PLoS One. 2016;11:e0157968. doi: 10.1371/journal.pone.0157968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yap J, Lim FY, Gao F, et al Correlation of the New York Heart Association Classification and the 6-minute walk distance: a systematic review. Clin Cardiol. 2015;38:621–628. doi: 10.1002/clc.22468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bohannon RW, Williams Andrews A Normal walking speed: a descriptive meta-analysis. Physiotherapy. 2011;97:182–189. doi: 10.1016/j.physio.2010.12.004. [DOI] [PubMed] [Google Scholar]
- 64.Salbach NM, O’Brien KK, Brooks D, et al Reference values for standardized tests of walking speed and distance: A systematic review. Gait Posture. 2015;41:341–360. doi: 10.1016/j.gaitpost.2014.10.002. [DOI] [PubMed] [Google Scholar]
- 65.Sustakoski A, Perera S, Vanswearingen JM, et al The impact of testing protocol on recorded gait speed. Gait Posture. 2015;41:329–331. doi: 10.1016/j.gaitpost.2014.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lee K, Davis MA, Marcotte JE, et al Falls in community-dwelling older adults with heart failure: A retrospective cohort study. Heart Lung. 2020;49:238–250. doi: 10.1016/j.hrtlng.2019.12.005. [DOI] [PubMed] [Google Scholar]
- 67.Schwenk M, Schmidt M, Pfisterer M, et al Rollator use adversely impacts on assessment of gait and mobility during geriatric rehabilitation. J Rehabil Med. 2011;43:424–429. doi: 10.2340/16501977-0791. [DOI] [PubMed] [Google Scholar]
- 68.Jayakaran P, DeSouza L, Cossar J, Gilhooly K Influence of a walking aid on temporal and spatial parameters of gait in healthy adults. PM R. 2014;6:796–801. doi: 10.1016/j.pmrj.2014.02.005. [DOI] [PubMed] [Google Scholar]
- 69.Peel NM, Kuys SS, Klein K Gait speed as a measure in geriatric assessment in clinical settings: a systematic review. J Gerontol A Biol Sci Med Sci. 2013;68:39–46. doi: 10.1093/gerona/gls174. [DOI] [PubMed] [Google Scholar]
- 70.Chaudhry SI, McAvay G, Ning Y, et al Risk factors for onset of disability among older persons newly diagnosed with heart failure: the Cardiovascular Health Study. J Card Fail. 2011;17:764–770. doi: 10.1016/j.cardfail.2011.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lo AX, Donnelly JP, McGwin G, et al Impact of gait speed and instrumental activities of daily living on all-cause mortality in adults ≥ 65 years with heart failure. Am J Cardiol. 2015;115:797–801. doi: 10.1016/j.amjcard.2014.12.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Pulignano G, Del Sindaco D, Di Lenarda A, et al. Incremental value of gait speed in predicting prognosis of older adults with heart failure: insights from the IMAGE-HF study. JACC Heart Fail. 2016; 4: 289-298.
- 73.Tanaka S, Kamiya K, Hamazaki N, et al Short-term change in gait speed and clinical outcomes in older patients with acute heart failure. Circ J. 2019;83:1860–1867. doi: 10.1253/circj.CJ-19-0136. [DOI] [PubMed] [Google Scholar]
- 74.Lord S, Howe T, Greenland J, et al Gait variability in older adults: A structured review of testing protocol and clinimetric properties. Gait Posture. 2011;34:443–450. doi: 10.1016/j.gaitpost.2011.07.010. [DOI] [PubMed] [Google Scholar]
- 75.Joyce E, Howell EH, Senapati A, et al Prospective assessment of combined handgrip strength and Mini-Cog identifies hospitalized heart failure patients at increased post-hospitalization risk. ESC Heart Fail. 2018;5:948–952. doi: 10.1002/ehf2.12300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pavasini R, Serenelli M, Celis-Morales CA, et al. Grip strength predicts cardiac adverse events in patients with cardiac disorders: an individual patient pooled meta-analysis. Heart 2019; 105: 834-841.
- 77.Larcher B, Zanolin-Purin D, Vonbank A, et al Usefulness of handgrip strength to predict mortality in patients with coronary artery disease. Am J Cardiol. 2020;129:5–9. doi: 10.1016/j.amjcard.2020.05.006. [DOI] [PubMed] [Google Scholar]
- 78.Fried LP, Tangen CM, Walston J, et al Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56:146–156. doi: 10.1093/gerona/56.3.M146. [DOI] [PubMed] [Google Scholar]
- 79.Cruz-Jentoft AJ, Bahat G, Bauer J, et al Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing. 2019;48:16–31. doi: 10.1093/ageing/afy169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cadena-Sanabria M, Velandia-Carrillo C Relationship between hand grip strength and functional class in elderly patients with heart failure. Innovation in Aging. 2017;1(suppl_1):S245–S246. [Google Scholar]
- 81.Duarte RRP, Gonzalez MC, Oliveira JF, et al. Is there an association between the nutritional and functional parameters and congestive heart failure severity? Clin Nutr 2021; 40: 3354-3359.
- 82.Rijk JM, Roos PR, Deckx L, et al Prognostic value of handgrip strength in people aged 60 years and older: A systematic review and meta-analysis. Geriatr Gerontol Int. 2016;16:5–20. doi: 10.1111/ggi.12508. [DOI] [PubMed] [Google Scholar]
- 83.Alley DE, Shardell MD, Peters KW, et al Grip strength cutpoints for the identification of clinically relevant weakness. J Gerontol A Biol Sci Med Sci. 2014;69:559–566. doi: 10.1093/gerona/glu011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Jäkel B, Kedor C, Grabowski P, et al Hand grip strength and fatigability: correlation with clinical parameters and diagnostic suitability in ME/CFS. J Transl Med. 2021;19:159. doi: 10.1186/s12967-021-02774-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Rockwood K, Awalt E, Carver D, MacKnight C Feasibility and measurement properties of the functional reach and the timed up and go tests in the Canadian study of health and aging. J Gerontol A Biol Sci Med Sci. 2000;55:70–73. doi: 10.1093/gerona/55.2.M70. [DOI] [PubMed] [Google Scholar]
- 86.Hauer K, Oster P. Measuring functional performance in persons with dementia. J Am Geriatr Soc 2008; 56: 949-950.
- 87.Mehmet H, Yang AWH, Robinson SR. What is the optimal chair stand test protocol for older adults? A systematic review. Disabil Rehabil 2020; 42: 2828-2835.
- 88.Kinoshita S, Kiyama R, Yoshimoto Y Effect of handrail height on sit-to-stand movement. PLoS One. 2015;10:e0133747. doi: 10.1371/journal.pone.0133747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Lindemann U, van Oosten L, Evers J, et al Effect of bed height and use of hands on trunk angular velocity during the sit-to-stand transfer. Ergonomics. 2014;57:1536–1540. doi: 10.1080/00140139.2014.933889. [DOI] [PubMed] [Google Scholar]
- 90.Hansen C, Beckbauer M, Romijnders R, et al Reliability of IMU-Derived Static Balance Parameters in Neurological Diseases. Int J Environ Res Public Health. 2021;18:3644. doi: 10.3390/ijerph18073644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Viceconti M, Hernandez Penna S, Dartee W, et al Toward a regulatory qualification of real-world mobility performance biomarkers in Parkinson’s patients using digital mobility outcomes. Sensors (Basel) 2020;20:5920. doi: 10.3390/s20205920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Maetzler W, Pilotto A Digital assessment at home - mPower against Parkinson disease. Nat Rev Neurol. 2021;17:661–662. doi: 10.1038/s41582-021-00567-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary data to this article can be found online.