Abstract
Background
Treadmill training is used in rehabilitation and is described as improving gait parameters of patients with Parkinson's disease.
Objectives
To assess the effectiveness of treadmill training in improving the gait of patients with Parkinson's disease and the acceptability and safety of this type of therapy.
Search methods
We searched the Cochrane Movement Disorders Group Specialised Register (see Review Group details for more information) (last searched September 2014), Cochrane Central Register of Controlled Trials (The Cochrane Library 2014, Issue 10), MEDLINE (1950 to September 2014), and EMBASE (1980 to September 2014). We also handsearched relevant conference proceedings, searched trials and research registers, and checked reference lists (last searched September 2014). We contacted trialists, experts and researchers in the field and manufacturers of commercial devices.
Selection criteria
We included randomised controlled trials comparing treadmill training with no treadmill training in patients with Parkinson's disease.
Data collection and analysis
Two review authors independently selected trials for inclusion, assessed trial quality and extracted data. We contacted the trialists for additional information. We analysed the results as mean differences (MDs) for continuous variables and relative risk differences (RD) for dichotomous variables.
Main results
We included 18 trials (633 participants) in this update of this review. Treadmill training improved gait speed (MD = 0.09 m/s; 95% confidence interval (CI) 0.03 to 0.14; P = 0.001; I2 = 24%; moderate quality of evidence), stride length (MD = 0.05 metres; 95% CI 0.01 to 0.09; P = 0.01; I2 = 0%; low quality of evidence), but walking distance (MD = 48.9 metres; 95% CI ‐1.32 to 99.14; P = 0.06; I2 = 91%; very low quality of evidence) and cadence did not improve (MD = 2.16 steps/minute; 95% CI ‐0.13 to 4.46; P = 0.07; I2 = 28%; low quality of evidence) at the end of study. Treadmill training did not increase the risk of patients dropping out from intervention (RD = ‐0.02; 95% CI ‐0.06 to 0.02; P = 0.32; I2 = 13%; moderate quality of evidence). Adverse events were not reported in included studies.
Authors' conclusions
This update of our systematic review provides evidence from eighteen trials with moderate to low risk of bias that the use of treadmill training in patients with PD may improve clinically relevant gait parameters such as gait speed and stride length (moderate and low quality of evidence, respectively). This apparent benefit for patients is, however, not supported by all secondary variables (e.g. cadence and walking distance). Comparing physiotherapy and treadmill training against other alternatives in the treatment of gait hypokinesia such as physiotherapy without treadmill training this type of therapy seems to be more beneficial in practice without increased risk. The gain seems small to moderate clinically relevant. However, the results must be interpreted with caution because it is not known how long these improvements may last and some studies used no intervention in the control group and underlie some risk of bias. Additionally the results were heterogenous and we found variations between the trials in patient characteristics, the duration and amount of training, and types of treadmill training applied.
Plain language summary
Treadmill training for people with Parkinson's disease
Question: We assessed whether treadmill training and body weight support, individually or in combination, could improve walking in people with Parkinson's disease when compared with other gait training methods or no treatment.
Background:Slow walking is a common problem for people with Parkinson's disease. For people with mild to moderate Parkinsons disease it affects ability to do everyday things and their quality of life. Treadmill training uses specially designed machines to help gait rehabilitation. However, the role of treadmill training for people with Parkinson's disease in improving gait parameters is still unclear.
Study characteristics: We identified 18 relevant trials, involving 633 participants which evaluated this type of therapy, up to September 2014.
Key results and quality of the evidence: Treadmill training did improve gait speed, and stride length; but walking distance and cadence did not improve. Acceptability of treadmill training for study participants was good and adverse events were rare. It seems that such devices could be beneficial and could be applied in routine rehabilitation. However, it is still not clear when and how often they should be used and how long a benefit lasts.
The quality of this evidence for the primary outcomes was moderate to low. Adverse events were not reported in studies and drop outs did not occur more frequently in people receiving treadmill training. Also we investigated only gait parameters, improvements of activities and/or quality of life were not investigated.
Summary of findings
Summary of findings for the main comparison. Treadmill training versus no treadmill training or active control intervention or gait training for patients with Parkinson's disease.
| Treadmill training versus no treadmill training or active control intervention or gait training for patients with Parkinson's disease | ||||||
| Patient or population: patients with patients with Parkinson's disease Settings: Inpatient and outpatient setting Intervention: Treadmill training versus no treadmill training or active control intervention or gait training | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Control | Treadmill training versus no treadmill training or active control intervention or gait training | |||||
| Gait speed at the end of the study ‐ Active control group (co‐interventions were similar in both groups) Measures of timed gait. Scale from: 0 to inf. | The mean gait speed at the end of the study ‐ active control group (co‐interventions were similar in both groups) in the control groups was 1.17 m/s1 | The mean gait speed at the end of the study ‐ active control group (co‐interventions were similar in both groups) in the intervention groups was 0.07 higher (0.03 to 0.12 higher) | 434 (14 studies) | ⊕⊕⊕⊝ moderate2 | ||
| Gait speed at the end of the study ‐ No interventioncontrol group (co‐interventions were not similar in both groups) Measures of timed gait. Scale from: 0 to inf. | The mean gait speed at the end of the study ‐ no intervention control group (co‐interventions were not similar in both groups) in the control groups was 1.43 m/s1 | The mean gait speed at the end of the study ‐ no intervention control group (co‐interventions were not similar in both groups) in the intervention groups was 0.4 higher (0.06 lower to 0.87 higher) | 76 (3 studies) | ⊕⊝⊝⊝ very low3,4,5 | ||
| walking distance in m (at the end of study; all studies) ‐ Active control group (co‐interventions were similar in both groups) Measures of timed gait. Scale from: 0 to inf. | The mean walking distance in m (at the end of study; all studies) ‐ active control group (co‐interventions were similar in both groups) in the control groups was 441.2 m1 | The mean walking distance in m (at the end of study; all studies) ‐ active control group (co‐interventions were similar in both groups) in the intervention groups was 9.48 higher (0.47 lower to 19.42 higher) | 385 (9 studies) | ⊕⊕⊝⊝ low2,3,4 | ||
| walking distance in m (at the end of study; all studies) ‐ No intervention control group (co‐interventions were not similar in both groups) Measures of timed gait. Scale from: 0 to inf. | The mean walking distance in m (at the end of study; all studies) ‐ no intervention control group (co‐interventions were not similar in both groups) in the control groups was 362 m1 | The mean walking distance in m (at the end of study; all studies) ‐ no intervention control group (co‐interventions were not similar in both groups) in the intervention groups was 364 higher (294.45 to 433.55 higher) | 31 (1 study) | See comment | ||
| acceptability and safety of treadmill training ‐ Active control group (co‐interventions were similar in both groups) Number of adverse events and drop‐outs | Study population | See comment | 531 (15 studies) | ⊕⊕⊕⊝ moderate2 | Risks were calculated from pooled risk differences | |
| 131 per 1000 | 122 per 1000 (81 to 161) | |||||
| Moderate | ||||||
| 0 per 1000 | 0 per 1000 (0 to 0) | |||||
| acceptability and safety of treadmill training ‐ No intervention control group (co‐interventions were not similar in both groups) Number of adverse events and drop‐outs | Study population | See comment | 102 (3 studies) | ⊕⊕⊝⊝ low3,4,5,6 | Risks were calculated from pooled risk differences | |
| 392 per 1000 | 255 per 1000 (‐39 to 553) | |||||
| Moderate | ||||||
| 200 per 1000 | 130 per 1000 (‐20 to 282) | |||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk ratio; | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
1 Final values reported 2 Downgraded due to several ratings with "High Risk of Bias" 3 Downgraded due to 95% confidence interval includes no effect and the upper or lower confidence limit crosses the minimal clinical important difference (MCID) 4 Downgraded due to the total population size being less than 400 (as a rule‐of‐thumb threshold) 5 Downgraded due to funnel plot asymmetry (diagnosed by visual inspection) 6 Not downgraded due to explainable statistical heterogeneity
Background
Description of the condition
Parkinson's disease (PD) is a progressive and disabling degenerative disorder characterised clinically by bradykinesia, tremor, rigidity, and postural instability. Disability occurs at all stages of the disease and the severity of disabilities usually increases with disease duration. Patients frequently have gait impairments, difficulty in linking movements together smoothly, and episodes of freezing. These problems together with balance disturbances lead to an increased incidence of falls with the concomitant risk of fractures. In fact one study found that 27% of Parkinson's patients have had a hip fracture within 10 years of their diagnosis (Johnell 1992).
Gait hypokinesia is one of the primary movement disorders associated with PD (Morris 2000). It is an important contributor to disability and quality of life in mild to moderate Parkinson disease (Muslimovic 2008). Kinematic measures have occasionally been found to been altered in individual patients but slowness of gait is the only symptom that has been consistently reported in group comparisons between control patients and patients with idiopathic PD (Morris 2000). Cadence control remains unaffected throughout its entire range in PD and gait hypokinesia is directly attributable to an inability to internally generate sufficiently large steps. Therefore, improvements of walking speed and stride length are the primary goals of gait rehabilitation in patients with PD (Pohl 2003), and reducing gait freezing when it is present.
The current management of PD focuses on pharmacological therapy; at present levodopa is regarded as the most effective treatment. However, many patients show abnormal involuntary movements due to levodopa known as dyskinesias (Jankovic 2000). Drugs other than levodopa such as dopamine agonists may initially control symptoms for many patients but levodopa and polytherapy are often necessary in the treatment of PD, particularly in the advanced stages (Motto 2003).
Despite new pharmacological interventions, treatment becomes unsatisfactory in a large proportion of patients. After five years of levodopa treatment, many patients experience severe motor complications such as motor fluctuations and dyskinesias. Theseare difficult to manage with the available drug strategies. Complications cause functional disability and impact on the person's quality of life (Motto 2003).
In recent years, interest in functional neurosurgery of basal ganglia has increased. Patients who have developed severe motor complications that are resistant to the available pharmacological interventions could be considered surgical candidates (Motto 2003). Three major targets for functional neurosurgery are; the thalamus ventro‐intermediate nucleus, internal globus pallidus, and subthalamic nucleus. Two different techniques, radiofrequency lesioning or high frequency stimulation (Limousin 1998) have been proposed. However, there is still debate concerning risks and benefits of surgery. A Cochrane review team is evaluating theses issues (Motto 2003).
Description of the intervention
Despite optimal medical and surgical therapies for PD, patients develop progressive disability (Deane 2001). However, the effectiveness of non‐pharmacological options such as exercises have recently been demonstrated (Goodwin 2008). A good example for patient‐tailored exercises is physiotherapy (Ashburn 2004; Comella 1994; de Goede 2001; Tomlinson 2013). The aim of physiotherapy is to enable PD patients to maintain their maximum level of mobility, activity, and independence. This outcome can be attained through monitoring of the patient’s condition, implementation of appropriate physical treatments, and incorporating a range of approaches to movement rehabilitation (Tomlinson 2013). However, in spite of established pharmacological and conventional approaches there is still a need for new interventions to improve the gait of people with PD.
Recently, the use of electromechanical devices such as treadmill training has provided a promising new therapeutic approach in the rehabilitation of patients with hemiparesis and impaired gait (Mehrholz 2014). Augmenting conventional therapy with treadmill training as a supplement to conventional therapies may improve the results of other gait training therapies. With seriously afflicted hemiparetic patients who cannot walk under their own power, treadmill training with bodyweight support (BWS) might be recommended.
How the intervention might work
As described recently, treadmill training with BWS has also been used with PD patients. Results of single studies suggested better improvement in gait parameters when compared with conventional gait therapy (Miyai 2002; Pohl 2003).
Treadmill training can be used to give people with PD intensive practice (in terms of high repetitions) of complex gait cycles. Treadmill training can be used to train at higher gait speeds and to achieve greater step length compared to physiotherapy not using such devices (Cakit 2007; Pohl 2003).
However, the most effective combination of training parameters (for example, amount and timing of BWS during the gait cycle and belt speed and acceleration) is still unknown. There is, therefore, still a need for a systematic evaluation in the form of a systematic review of the available literature. The present review assesses the effectiveness and acceptability of treadmill training to augment conventional gait rehabilitation for patients with PD.
Why it is important to do this review
As the scientific evidence for the benefits of treadmill training may have changed since our Cochrane Review was first published in 2009 (Mehrholz 2010), an update of the review seems to be required in order to justify the large equipment and human resource cost needed to implement treadmill training devices as well as to confirm the safety and acceptance of this type of training. Therefore, it seems to be important that this version of our review provides an update of the best available evidence about the above‐mentioned approach.
Objectives
To assess the effectiveness of treadmill training in improving the gait function of patients with Parkinson's disease and the acceptability and safety of this type of therapy. A secondary objective of this review is to find the most effective combination of training parameters (for example belt speed and acceleration).
Methods
Criteria for considering studies for this review
Types of studies
We included randomised controlled trials (RCTs) and randomised controlled cross‐over trials where only the first period was analysed as a parallel group trial.
Types of participants
We included studies with participants of both genders and all ages who were diagnosed with PD using the UK Parkinson's Disease Brain Bank Criteria (or PD diagnostic criteria as defined by the study authors) regardless of drug therapy, duration of treatment, duration of PD, or level of initial impairment.
Types of interventions
We compared treadmill training versus no treadmill training (main analysis) for improving gait. We assumed that co‐ interventions such as other rehabilitation interventions and medication or treatment were comparable between groups. Because this can not be assumed we compared treadmill training with a variety of other interventions in the (control group) and described these in an additional table. If co‐ interventions were comparable between groups e.g. active versus no‐active control intervention we did a separate comparison.
No restriction was placed for the duration or characteristics of the intervention. We considered end‐of‐treatment assessments as provided by the studies.
Types of outcome measures
Primary outcomes
The primary outcomes were walking speed (continuous outcome) and stride length (continuous). According to Hass 2014 we defined the cut‐off value representing a minimal clinical important difference (CID) for walking speed at 0.06 m/s, a moderate CID at 0.14 m/s and a large CID at 0.22 m/s
Secondary outcomes
The secondary outcomes were cadence (continuous) and walking distance (continuous). Another secondary outcome was the acceptability and safety of treadmill training. We investigated the safety of treadmill training using the incidence of adverse events such as cardiovascular events, injuries, and pain, and any other reported adverse events. To measure the acceptance of treadmill training we used drop outs from the study due to any reason.
We provided all primary and secondary outcomes in a summary of findings table. If we had more than seven outcomes to present we prioritised them according to their relevance and presented the most important outcomes.
Search methods for identification of studies
Electronic searches
We used the search strategy developed for the Movement Disorders Group and identified relevant trials by searching the following electronic databases:
Cochrane Movement Disorders Group Specialised Register;
Cochrane Central Register of Controlled Trials (CENTRAL) (The Cochrane Library; last searched September 2014);
MEDLINE (1966 to September 2014);
EMBASE (1966 to September 2014);
Pedro (last search September 2014).
The MeEDLINE and EMBASE searches can be found in the Appendices
Searching other resources
In addition, we also
searched the reference lists for identified trials and review articles;
hand‐searched and screened reference lists of potentially relevant conference proceedings (1998 to September 2014;Appendix 5) searched ongoing trials and research registers; contacted trialists, other researchers, and manufacturers of commercial devices in our field of study to identify published, unpublished, and ongoing trials not available in the major databases; contacted trialists and other researchers to obtain additional information on trials published elsewhere and unpublished trials.
Publication status or language did not influence our decision to include.
Data collection and analysis
Selection of studies
Selection and identification of relevant trials Two authors (JM and MP) independently read titles and, when available, abstracts of identified references and eliminated obviously irrelevant studies. Two review authors (MP and BE) independently examined potentially relevant studies using the predetermined criteria for including studies. We obtained the full text for the remaining studies. Based on our inclusion criteria (types of studies, participants, aims of interventions, outcome measures) two review authors (BE and MP) independently ranked these studies as relevant, irrelevant, or possibly relevant. We excluded all trials ranked initially as irrelevant, but included all other trials at this stage. We resolved disagreement among authors through discussion. If further information was needed to reach consensus we contacted trialists in an effort to obtain missing information
Data extraction and management
Two review authors (JM and MP) independently extracted trial and outcome data from the selected trials. If any review author was involved in any of the selected studies another member of our author group who was not involved in the study was requested to review the study information. We established the characteristics of unpublished trials through correspondence with the trial co‐ordinator or principal investigator. We used checklists to independently record details of the:
methods of generating randomisation schedule;
methods of concealment of allocation;
blinding of assessors;
use of an intention‐to‐treat analysis (all participants initially randomised were included in the analyses as allocated to groups);
adverse events and drop outs for all reasons;
important imbalance in prognostic factors;
participants (country, number of participants, age, gender, stage of PD as assessed by Hoehn Yahr for entry to the study, inclusion and exclusion criteria);
comparison (details of the intervention in treatment and control groups; details of co‐intervention(s) in both groups; duration of treatment);
outcomes and time points of measures (number of participants in each group and outcome, regardless of compliance).
We checked all of the extracted data for agreement among review authors, with another review author (BE or JK) arbitrating any items where consensus was not reached. If necessary, we contacted trialists to request more information, clarification, or missing data. If data was still missing we analysed the available data, but did not impute data.
The primary outcome variables of interest were continuous data, entered as means and standard deviations. We calculated a pooled estimate of the mean differences (MD) with 95% confidence intervals (CI). If studies did not use the same outcome, we use the standardised mean difference (SMD) with 95% CI.
For all binary outcomes (such as the secondary outcome 'drop out, from all causes') we calculated risk differences (RD), again with 95% CI.
If necessary we combined the results of different treadmill training groups in one (collapsed, treadmill) group and compared this with the combined results of the control group. We combined continuous data for pooled arms using the implemented RevMan Calculator. We have built a summary of findings table using the software GRADEprofiler and conducted GRADE assessments according to the GRADEprofiler help.
Assessment of risk of bias in included studies
For this update of the review two authors (BE and JM) independently assessed the risk of bias in the included trials in accordance with the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). We described the agreement between authors during the assessment of risk of bias, and we resolved disagreement by reaching consensus through discussion. We contacted trialists for clarification and to request missing information.
We checked all methodological quality assessments for agreement among the review authors and resolved disagreements by discussion among authors. Two review authors (MP and JM) were co‐authors of one included trial (Pohl 2003); other review authors (BE and JK) did the quality assessment for this trial. We contacted study authors for clarification and to request missing information. We did test the robustness of the main results in a sensitivity analysis (Analysis 2.1).
2.1. Analysis.
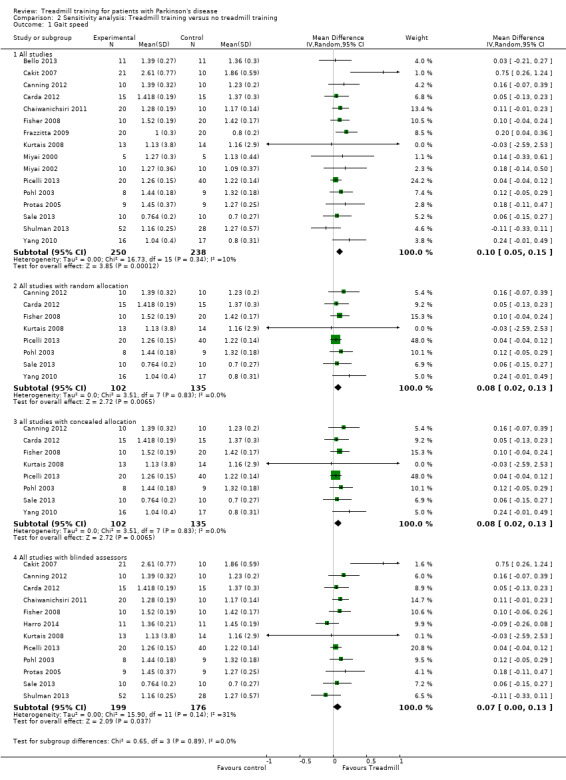
Comparison 2 Sensitivity analysis: Treadmill training versus no treadmill training, Outcome 1 Gait speed.
Measures of treatment effect
For all outcomes representing continuous data, we entered means and standard deviations. We calculated a pooled estimate of the mean difference (MD) with 95%confidence interval (CI). For all binary outcomes we calculated risk differences (RD) with 95% CI. For all analyses we used The Cochrane Collaboration’s Review Manager software, RevMan 5.2 and used a random‐effects model for all analyses.
Dealing with missing data
We contacted the relevant principal investigators to retrieve missing data.
Assessment of heterogeneity
We used the I² statistic to assess heterogeneity. We used a random effects model, regardless of the level of heterogeneity. Thus, in the case of heterogeneity we did not violate the preconditions of a fixed‐effect model approach. We visually examined publication bias using funnel plots.
Subgroup analysis and investigation of heterogeneity
To quantify for heterogeneity we used the I2 statistic for all comparisons. We always used random‐effects model regardless of the level of heterogeneity. We described variability in participants, interventions, and outcomes studied (clinical diversity) in an additional table (Table 2) and in the Description of studies. The variability of studies did not influence our intention to pool trials. For all statistical analyses we used the latest version of The Cochrane Collaboration's software Review Manager (RevMan).
1. Patient characteristics in studies.
| Study ID | Age, mean (SD) EXP | Age, mean (SD) CON | Hoehn & Yahr stages |
mean Duration of disease EXP |
mean Duration of disease CON |
female/male EXP | female/male CON | Duration of therapy | frequency of training | intensity of training in terms of minutes per session | intensity of training in terms of treadmill speed paradigm |
| Bello 2013 | 60 (11) | 58 (9) | 1 to 3 | 5 years | 5 years | 4/7 | 5/6 | 5 weeks | 3 times a week | 16' with increments of 4 ' per week | constant and as individually preferred speed |
| Cakit 2007 | 72 (6)* | 1 to 2 | 6 years* | 15/16* | 8 weeks | not described | 30 | relatively similar to so called speed dependent treadmill approach (Pohl 2002) | |||
| Canning 2012 | 61 (6) | 63 (10) | 1 to 2 | 6 years | 6 years | 5/5 | 4/6 | 6 weeks | 4 times a week | 20‐40 | gradually increased speed |
| Carda 2012 | 61 (6) | 63 (10) | 1 to 2 | 6 years | 5 years | not described | 6 weeks | 4 times a week | 30 | high, (80% of max), gradually increased | |
| Chaiwanichsiri 2011 | 68 (5) | 69 (5) | 2 to 3 | 6 years | 4 years | 0/10 | 0/10 | 4 weeks | 3 times a week | 20 | slightly higher than preferred |
| Fisher 2008 | 64 (15) | 62 (10) | 1 to 2 | 1 year | 1 year | 4/6 | 13/7 | 8 weeks | 3 times a week | 45 | progression of speed in high intensity group/ and low to moderate progression of speed in low intensity group |
| Frazzitta 2009 | 71 (8) | 71 (7) | 3 | 13 years | 13 years | 12/8 | 11/9 | 4 weeks | 7 times a week | 20 | 60% of max speed at start, then gradually increased |
| Harro 2014 | 65 (9) | 67 (11) | 1 to 3 | 4 years | 9 years | 5/5 | 2/8 | 6 weeks | 3 times a week | 30 | both groups received speed training relatively similar to so called speed dependent approach (Pohl 2002) |
| Kurtais 2008 | 64 (11) | 66 (5) | mean 2.2 to 2.5 | 5 years | 5 years | 7/5 | 5/7 | 6 weeks | 3 times a week | 40 | gradually increased speed |
| Miyai 2000 | 67 (2)* | 2.5 to 3 | 4 years* | 5/5* | 4 weeks | 3 times a week | 36‐45 | gradually increased speed | |||
| Miyai 2002 | 70 (2) | 70 (2) | 2.5 to 3 | 4 years | 4.5 years | 6/5 | 4/5 | 4 weeks | 3 times a week | 45 | gradually increased speed |
| Nadeau 2013 | 62 (7) | 64 (6) | 1 to 2 | Not reported | 2/9 | 5/18 | 24 weeks | 3 times a week | 60 | gradually increased speed | |
| Picelli 2013 | 69 (8) | 68 (9) | 3 | 7 years | 7 years | 14/6 | 23/17 | 4 weeks | 3 times a week | 30 | gradually increased speed |
| Pohl 2003 | 61 (9) | 61 (9) | 1 to 2.5 | 3 years | 3 years | 3/5 | 2/7 | 1 session | N.a. | 30 | similar to so called speed dependent treadmill approach (Pohl 2002) |
| Protas 2005 | 71 (7) | 74 (9) | 2 to 3 | 7 years | 8 years | not described | 8 weeks | 3 times a week | 30 | relatively similar to so called speed dependent treadmill approach (Pohl 2002) | |
| Sale 2013 | 68 (9) | 70 (10) | 2.5 to 3.5 | 9 years | 8 years | 5/5 | 6/4 | 4 weeks | 5 times a week | 45 | gradually increased speed |
| Shulman 2013 | 66 (10) | 65 (11) | 2 to 3 | 6 years | 6 years | 17/32 | 4/18 | 12 weeks | 3 times a week | 30‐50 | no clear speed increases but depending on maximal heart reserve speed was increased |
| Yang 2010 | 68 (8) | 66 (11) | 1 to 3 | 5 years | 5 years | 6/9 | 8/7 | 4 weeks | 3 times a week | 30 | constant, comfortable speed |
* information not available by group
Sensitivity analysis
We incorporated a post hoc sensitivity analysis for methodological quality to test the robustness of our results for the primary outcome gait speed. We analysed random sequence generation, allocation concealment, blinding of outcome assessors.
Results
Description of studies
See:Characteristics of included studies; Characteristics of excluded studies;Characteristics of studies awaiting classification; and Characteristics of ongoing studies.
Studies that were included compared treadmill training with a variety of other active interventions and none (Characteristics of included studies; Table 3; Additional tables).
2. Characteristics of control group in studies.
| Study ID | active treatment | no interventions | gait training | control group |
| Bello 2013 | yes | yes | overground gait training, 3 times a week for 5 weeks (72 min a week) | |
| Cakit 2007 | yes | not described further | ||
| Canning 2012 | yes | usual care including advice to maintain usual physical activity levels | ||
| Carda 2012 | yes | yes | robotic gait training, 3 times a week for 4 weeks (120 min a week) | |
| Chaiwanichsiri 2011 | yes | yes | home walking program, 6 times a week for 4 weeks (180 min a week) | |
| Fisher 2008 | yes | 1 | (2) low‐intensity group: general or traditional physiotherapy, for 24 sessions in 8 weeks (3) zero‐intensity (no‐exercise) group: six 1 hour education class over 8 weeks | |
| Frazzitta 2009 | yes | 1 | traditional rehabilitation with visual and auditory cues, 7 times a week for 4 weeks (140 min a week) | |
| Harro 2014 | yes | yes | 6 weeks rhythmic auditory‐cueing with incremental speed increases in small groups of five participants, 30 minutes a session, not described how often a week | |
| Kurtais 2008 | yes | not further described by the authors | ||
| Miyai 2000 | yes | 4 weeks conventional physiotherapy, 45 minutes a day, 3 days a week | ||
| Miyai 2002 | yes | 4 weeks conventional physiotherapy, 45 minutes a day, 3 days a week, with a total of 12 sessions | ||
| Nadeau 2013 | yes | low exercise intensity training in seated position, 3 times a week for 24 weeks (180 min a week) | ||
| Picelli 2013 | yes | yes | 3 arms:(1) robotic gait training group, twelve, 45‐min sessions, three days a week for 4 consecutive weeks (3) Physical Therapy group, twelve, 45‐min sessions, three days a week for 4 consecutive weeks | |
| Pohl 2003 | yes | yes | 4 arms(3) physiotherapy group: 1 session physiotherapy including gait training, 30 minutes (4) control group: resting in a chair for 30 minutes | |
| Protas 2005 | yes | no training | ||
| Sale 2013 | yes | yes | robot‐assisted gait training (device: G‐EO), 5 times a week for 4 weeks (225 min a week) | |
| Shulman 2013 | yes | stretching and resistance training, 3 times a week for 12 weeks (duration of sessions not described) | ||
| Yang 2010 | yes | conventional therapy, 3 times a week for 4 weeks (90 min a week) |
The age of participants was between 58 and 74 [BJ1] years and the disease severity was in most studies between Hoehn & Yahr stages 1 and 3.
13 out of 18 studies (72%) used UPDRS (total or subscales) at baseline for patient description but only 8 out of 18 included studies (44%) at study end (Table 4).
3. Use of UPDRS and QoL scales and follow‐up.
| Study ID | U PDRS at baseline | UPDRS at study end | QoL at baseline | Follow‐Up |
| Bello 2013 | UPDRS motor score | UPDRS motor score | ‐ | no |
| Cakit 2007 | UPDRS motor score | ‐ | ‐ | no |
| Canning 2012 | UPDRS motor score | UPDRS motor score | PDQ‐39 | after 6 weeks |
| Carda 2012 | UPDRS motor score | UPDRS motor score | SF‐12 PCS and MCS | after 3, 6 months |
| Chaiwanichsiri 2011 | ‐ | ‐ | ‐ | after 1 months |
| Fisher 2008 | UPDRS (total and subscales) | UPDRS (total and subscales) | ‐ | no |
| Frazzitta 2009 | UPDRS motor score | ‐ | ‐ | no |
| Harro 2014 | ‐ | ‐ | ‐ | 3mo |
| Kurtais 2008 | ‐ | ‐ | ‐ | no |
| Miyai 2000 | UPDRS (total and subscales) | UPDRS (total and subscales) | ‐ | no |
| Miyai 2002 | UPDRS (total and subscales) | UPDRS (total and subscales) | ‐ | after 2,3,4,5 and 6 months |
| Nadeau 2013 | UPDRS (total and subscales) | UPDRS (total and subscales) | PDQ‐39 | after 6 months |
| Picelli 2013 | UPDRS (total) | UPDRS (total) | ‐ | 3 months |
| Pohl 2003 | UPDRS (total and subscales) | ‐ | ‐ | no |
| Protas 2005 | ‐ | ‐ | ‐ | no |
| Sale 2013 | UPDRS (total and subscales) | ‐ | ‐ | no |
| Shulman 2013 | UPDRS (total and subscales) | ‐ | ‐ | no |
| Yang 2010 | ‐ | ‐ | ‐ | after 1 months |
Only 3 out of 18 studies (17%) assessed quality of life (2 studies used the PDQ‐39 and 1 study used the SF‐12 PCS and MCS (Table 4).
Eight out of 18 studies (44%) described a follow‐up assessment after study end (Table 4).
No adverse events were reported.
The trials were relatively comparable regarding patient’s characteristics (Table 2), but experimental and control interventions varied (Table 2; Table 3). E.g. some studies used a active control group doing time and dose matched gait exercises, but some did not described what was done.
Results of the search
Figure 1 shows the flow diagram for the selection of studies. The searches of the electronic databases and trials registers generated 925 unique references for screening.
1.
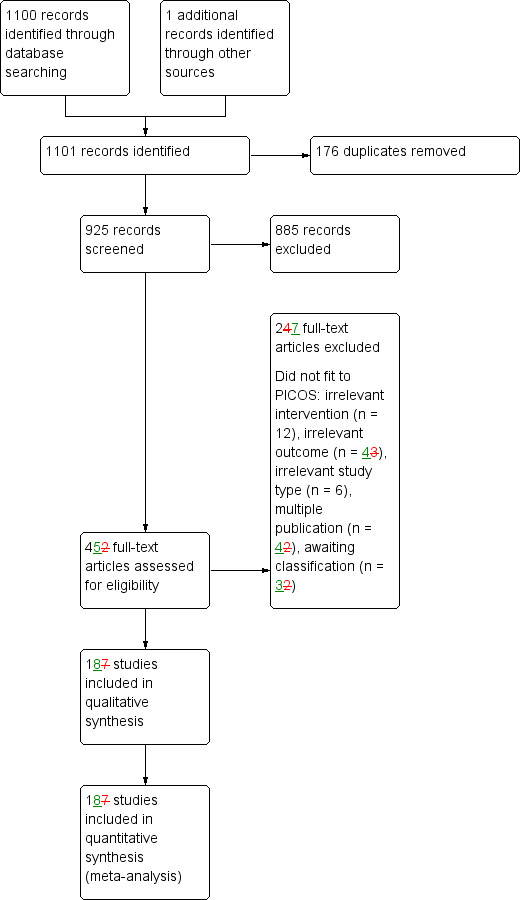
Study flow diagram.
After excluding non‐relevant citations we obtained the full texts of 45 papers; of these, we included 18 trials in the qualitative analysis and 18 trials in the quantitative analysis of the review.
Included studies
We included 18 trials involving a total of 623 participants in the quantitative analysis of this review (Bello 2013; Cakit 2007; Canning 2012; Carda 2012; Chaiwanichsiri 2011; Fisher 2008; Frazzitta 2009; Harro 2014; Kurtais 2008; Miyai 2000; Miyai 2002; Nadeau 2013; Picelli 2013; Pohl 2003; Protas 2005; Sale 2013; Shulman 2013; Yang 2010); see the Characteristics of included studies; Table 2;).
The characteristics of participants and the characteristics of the experimental interventions in the included studies are listed and described in detail in Table 2.
The included trials compared treadmill training with a variety of other interventions. We conducted a meta‐analysis of studies that measured the same treatment effect. Thus we combined treadmill training versus all other approaches as an estimate of the effect of treadmill training compared with a different treatment. However, we did not compare treadmill training type A with treadmill training type B as these are measuring different treatment effects.
Studies used a variety of primary outcomes, which are described in Characteristics of included studies. Because only 44% of studies reported follow‐up data we did not conduct a separate analysis of ‚end‐of‐treatment’ and ‚follow‐up’ data.
Excluded studies
Six studies were excluded (Bello 2008; Fisher 2013; Ganesan 2010; Gianfrancesco 2009; Diaz de la Fe 2008; Schenkman 2012). These trials were excluded for various reasons and the details are described in Characteristics of excluded studies. If there was any doubt whether the study should be excluded or not, we retrieved the full text of the article. In cases of disagreement between the review authors, another member of the author group reviewed the information to decide on inclusion or exclusion of a study.
One ongoing study was identified. Two studies (Horak 2011; Mezzarobba 2013) are still awaiting classification and are described in Characteristics of studies awaiting classification.
Risk of bias in included studies
All details about the methodological quality are provided for each included study in Figure 2.
2.
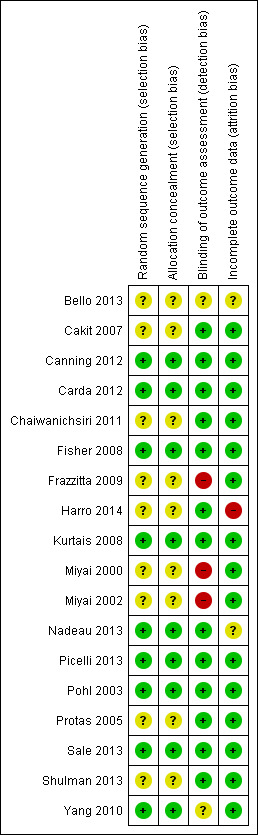
Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
We wrote to the authors of all the included studies requesting (if necessary) clarification of some design features or missing information in order to complete the quality ratings. The correspondence was via email and letter, and we wrote reminders every two weeks if we did not receive an answer. The risk of bias decisions are described in the (Characteristics of included studies and Figure 2).
Allocation
Nine out of 18 studies (50%) described an appropriate random sequence generation and some studies described allocation concealment appropriately (Figure 2). No included study described an inappropriate random sequence generation or allocation concealment.
Blinding
Twelve out of 18 studies (67%) used an appropriate blinding of outcome assessors and three out of 18 studies did not blind outcome assessors (17%) for two out of 18 studies (11%) this was unclear (Figure 2).
Incomplete outcome data
Fourteen studies (83%) described outcome data appropriately (Figure 2).
Effects of interventions
See: Table 1
See: Table 1 for the main comparison 'Treadmill training versus all other interventions'.
Treadmill training versus all other interventions (no treadmill training)
Comparison 1.1 Gait speed at the end of intervention phase (primary outcome measure)
Seventeen studies with a total of 520 participants compared treadmill training versus no treadmill training on gait speed. Treadmill training improved gait speed significantly. The pooled standardised mean difference (MD, random‐effect model) for gait speed was 0.09 m/s (95% CI 0.03 to 0.14; P = 0.001; level of heterogeneity I2 = 24%; moderate quality of evidence) at the end of the study (Analysis 1.1).
1.1. Analysis.
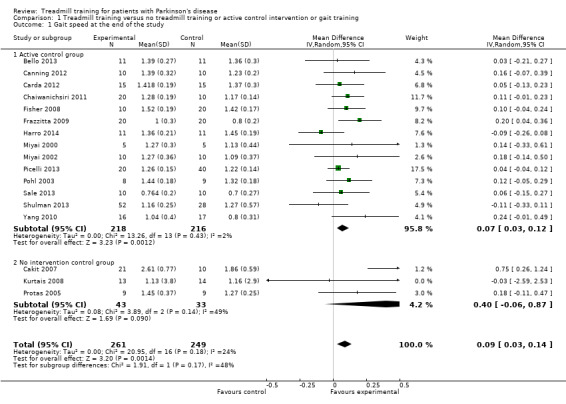
Comparison 1 Treadmill training versus no treadmill training or active control intervention or gait training, Outcome 1 Gait speed at the end of the study.
Comparison 1.1.1 Active control group
Fourteen studies with a total of 434 participants used an active control group and compared treadmill training versus no treadmill training on gait speed. Treadmill training improved gait speed significantly. The pooled standardised mean difference (MD, random‐effect model) for gait speed was 0.07 m/s (95% CI 0.03 to 0.12; P = 0.001; level of heterogeneity I2 = 2%) at the end of the study (Analysis 1.1).
Comparison 1.1.2 No intervention control group
Three studies with a total of 76 participants used no intervention in the control group and compared treadmill training versus no treadmill training on gait speed. Treadmill training did not improve gait speed significantly. The pooled standardised mean difference (MD, random‐effect model) for gait speed was 0.40 m/s (95% CI ‐0.06 to 0.87; P = 0.09; level of heterogeneity I2 = 49%) at the end of the study (Analysis 1.1).
Comparison 1.2 Stride length at the end of intervention phase (primary outcome measure)
Overall ten studies with a total of 333 participants compared treadmill training versus no treadmill training on stride length. Treadmill training improved stride length significantly. The MD (random‐effect model) for stride length was 0.05 metres (95% CI 0.01 to 0.09; P = 0.01; I2 = 0%; low quality of evidence) at the end of the study (Analysis 1.2).
1.2. Analysis.
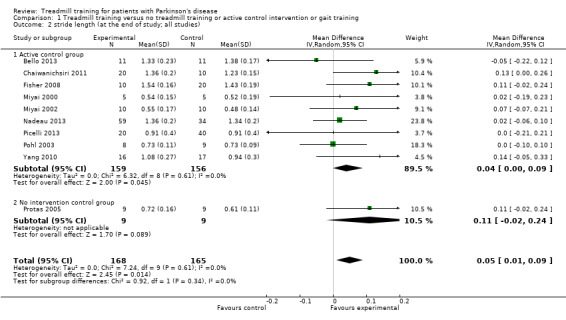
Comparison 1 Treadmill training versus no treadmill training or active control intervention or gait training, Outcome 2 stride length (at the end of study; all studies).
Comparison 1.2.1 Active control group (co‐interventions were similar in both groups)
Nine studies with a total of 315 participants used an active control group and compared treadmill training versus no treadmill training on stride length. Treadmill training did not improve stride length significantly. The MD (random‐effect model) for stride length was 0.04 metres (95% CI 0.00 to 0.09; P = 0.05; I2 = 0%) at the end of the study (Analysis 1.2).
Comparison 1.2.2 No intervention control group (co‐interventions were not similar in both groups)
One study (Protas 2005) with 18 participants used no intervention in the control group and compared treadmill training versus no treadmill training on stride length. Treadmill training did not improve stride length significantly. The MD (random‐effect model) for stride length was 0.11 metres (95% CI ‐0.02 to 0.24; P = 0.09; I2 = not applicable) at the end of the study (Analysis 1.2).
Comparison 1.3 Walking distance at the end of intervention phase
Overall ten studies with a total of 416 participants compared treadmill training versus no treadmill training on walking distance. Treadmill training did not improve walking distance significantly. The MD (random‐effect model) for walking distance was 48.9 metres (95% CI ‐1.32 to 99.1; P = 0.06; I2 = 91%; very low quality of evidence) at the end of the study (Analysis 1.3).
1.3. Analysis.
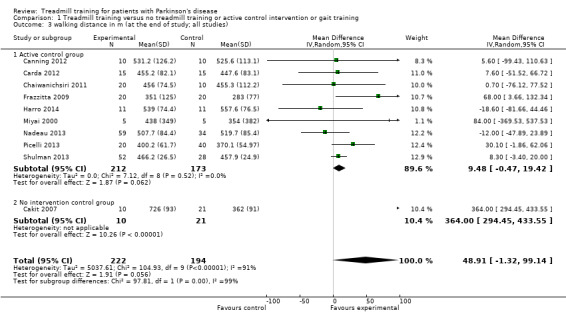
Comparison 1 Treadmill training versus no treadmill training or active control intervention or gait training, Outcome 3 walking distance in m (at the end of study; all studies).
It should be noted however that the described effect (treadmill training on walking distance) is mainly due to one trial (Cakit 2007), additionally the results for walking distance are very heterogeneous due to this trial (Cakit 2007). After leaving out the study by (Cakit 2007) there would be no effect: The MD (random‐effect model) for walking distance would be 8 metres (95% CI ‐3 to 20; P = 0.05; I2 = 0%).
Comparison 1.3.1 Active control group (co‐interventions were similar in both groups)
Nine studies with a total of 385 participants used an active control group and compared treadmill training versus no treadmill training on walking distance. Treadmill training did not improve walking distance significantly. The MD (random‐effect model) for walking distance was 9.48 metres (95% CI ‐0.47 to 19.42; P = 0.06; I2 = 0%) at the end of the study (Analysis 1.3).
Comparison 1.3.2 No intervention control group (co‐interventions were not similar in both groups)
One study (Cakit 2007) with 31 participants used no intervention in the control group and compared treadmill training versus no treadmill training on walking distance. Treadmill training improve walking distance significantly. The MD (random‐effect model) for walking distance was 364 metres (95% CI 294 to 434; P < 0.00001; I2 = not applicable) at the end of the study (Analysis 1.3).
Comparison 1.4 Cadence at the end of intervention phase
Overalls, ten studies with a total of 336 participants compared treadmill training versus no treadmill training on cadence. Treadmill training did not improve cadence significantly. The MD (random‐effect model) for cadence was 2.16 steps per minute (95% CI ‐0.13 to 4.46; P = 0.07; I2 = 17%; low quality of evidence) at the end of the study (Analysis 1.4).
1.4. Analysis.
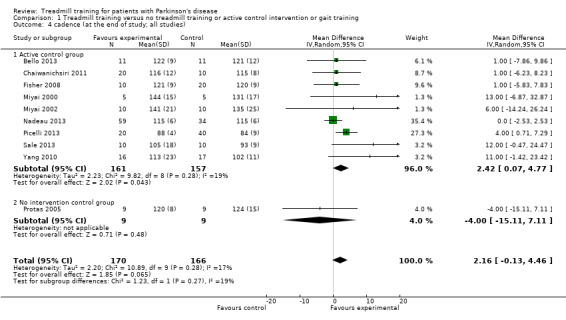
Comparison 1 Treadmill training versus no treadmill training or active control intervention or gait training, Outcome 4 cadence (at the end of study; all studies).
Comparison 1.4.1 Active control group (co‐interventions were similar in both groups)
Nine studies with a total of 318 participants used an active control group and compared treadmill training versus no treadmill training on cadence. Treadmill training did improve cadence significantly. The MD (random‐effect model) for cadence was 2.42 steps per minute (95% CI 0.07 to 4.77; P = 0.04; I2 = 19%) at the end of the study (Analysis 1.4).
Comparison 1.4.2 No intervention control group (co‐interventions were not similar in both groups)
One study (Protas 2005) with 18 participants used no intervention in the control group and compared treadmill training versus no treadmill training on cadence. Treadmill training did not improve cadence significantly. The MD (random‐effect model) for cadence was ‐4 steps per minute (95% CI ‐15.11 to 7.11; P = 0.48; I2 = not applicable) at the end of the study (Analysis 1.4).
Comparison 1.5 Acceptability and safety at the end of intervention phase
All 18 trials, with a total of 633 participants, reported drop‐out rates. We pooled the reported drop outs from all causes during the trial period. The use of treadmill training in patients with PD did not increase the risk of participants dropping out (risk difference (RD) (random‐effects model) ‐0.02; 95% CI ‐0.06 to 0.02; P = 0.32; I2 = 13%; moderate quality of evidence). No adverse events were reported in included studies (Analysis 1.5).
1.5. Analysis.
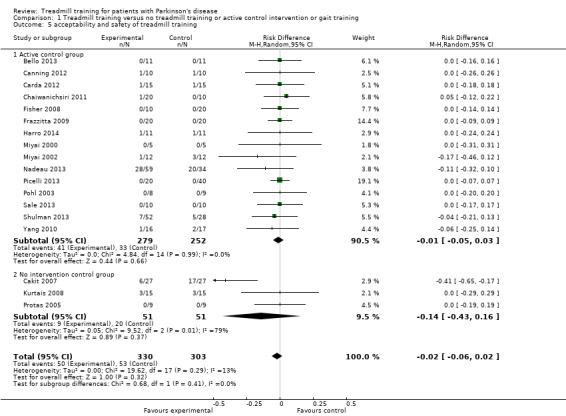
Comparison 1 Treadmill training versus no treadmill training or active control intervention or gait training, Outcome 5 acceptability and safety of treadmill training.
It should be noted that the acceptability might be influenced by one trial (Cakit 2007), however this study contributes to this analysis only by 2.9% (weight) (Analysis 1.5).
Comparison 1.5.1 Active control group (co‐interventions were similar in both groups)
15 trials, with a total of 531 participants used an active control group and reported drop‐out rates. We pooled the reported drop outs from all causes during the trial period. The use of treadmill training in patients with PD did not increase the risk of participants dropping out (risk difference (RD) (random‐effects model) ‐0.01; 95% CI ‐0.05 to 0.03; P = 0.66; I2 = 0%). No adverse events were reported in included studies (Analysis 1.5).
Comparison 1.5.2 No intervention control group (co‐interventions were not similar in both groups)
Three trials, with a total of 102 participants used no intervention in the control group and reported drop‐out rates. We pooled the reported drop outs from all causes during the trial period. The use of treadmill training in patients with PD did not increase the risk of participants dropping out (risk difference (RD) (random‐effects model) ‐0.14; 95% CI ‐0.43 to 0.16; P = 0.37; I2 = 79%). No adverse events were reported in included studies (Analysis 1.5).
Comparison 2.1: Sensitivity analysis by trial methodology
To test the robustness of the main results we used for our planned sensitivity analysis subgroups of the methodological features of randomisation, concealment of allocation, and blinding of assessors (Analysis 2.1).
To examine the robustness of results, we specified variables in a sensitivity analysis that we believed could influence the size of effect observed (method of randomisation, concealed allocation and blinding of assessors; Analysis 2.1).
Including only studies with described method of randomisation analysis
Eight trials with a total of 237 patients described a method of randomisation analysis. Treadmill training did improve gait speed. The pooled mean difference (MD, random‐effects model) for gait speed was 0.08; 95% confidence interval (CI) 0.02 to 0.13; P = 0.006; level of heterogeneity I2= 0%) at the end of study (Analysis 2.1).
Including only studies with adequate concealed allocation for the primary outcome gait speed
Eight trials with a total of 237 patients with adequate concealment of allocation were included. Treadmill training did improve gait speed. The pooled mean difference (MD, random‐effects model) for gait speed was 0.08 95% confidence interval (CI) 0.02 to 0.13; P = 0.006; level of heterogeneity I2= 0%) at the end of study (Analysis 2.1).
Including only studies with blinded assessors for the primary outcome gait speed
Twelve trials with a total of 375 patients described a blinded assessor for the primary outcome gait speed. Treadmill training did improve gait speed. The pooled mean difference (MD, random‐effects model) for gait speed was 0.07; 95% confidence interval (CI) 0.00 to 0.13; P = 0.04; level of heterogeneity I2= 31%) at the end of study (Analysis 2.1).
Comparison 2. 2: Sensitivity analysis by treadmill protocol used (gait speed increments)
To test the robustness of the main results we used for our second sensitivity analysis subgroups of the treadmill protocols used in studies (speed dependent approach, gradually increases of gait speed, constant gait speed or mixed) (Analysis 2.2).
2.2. Analysis.
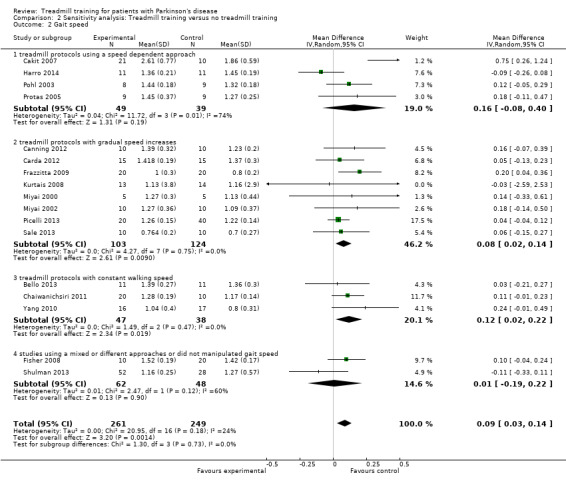
Comparison 2 Sensitivity analysis: Treadmill training versus no treadmill training, Outcome 2 Gait speed.
To examine the robustness of results, we categorised variables in this second sensitivity analysis that we believed could influence the size of effect observed (treadmill protocols used in studies Analysis 2.2).
Including only studies with speed dependent approach
Four trials with a total of 88 patients described a speed dependent approach. Treadmill training did not improve gait speed. The pooled mean difference (MD, random‐effects model) for gait speed was 0.16; 95% confidence interval (CI) ‐0.08 to 0.40; P = 0.19; level of heterogeneity I2= 74%) at the end of study (Analysis 2.2).
It should be noted however that the described effect might be affected by one trial (Harro 2014). This trial investigated a very small contrast between groups because in the experimental as in the control group a speed dependent approach was used (see Table 2 and Table 3). After leaving out the study by (Harro 2014) the effect would be (MD (random‐effect model) for gait speed would be higher (not significant) 0.27 m/s (95% CI ‐0.02 to 0.56; P = 0.06; and less heterogenous with I2 = 64%).
Including only studies with gradual gait speed increases
Eight trials with a total of 227 patients described a treadmill protocol with gradual increases of gait speed. Treadmill training did improve gait speed. The pooled mean difference (MD, random‐effects model) for gait speed was 0.08 95% confidence interval (CI) 0.02 to 0.14; P = 0.009; level of heterogeneity I2= 0%) at the end of study (Analysis 2.2).
Including only studies with constant gait speed
Three trials with a total of 85 patients described a treadmill protocol with constant gait speed. Treadmill training did improve gait speed. The pooled mean difference (MD, random‐effects model) for gait speed was 0.12; 95% confidence interval (CI) 0.02 to 0.22; P = 0.02; level of heterogeneity I2= 0%) at the end of study (Analysis 2.2).
Including only studies with a mixed or different gait speed approaches used
Two trials with a total of 110 patients described a treadmill protocol with mixed or different approaches. Treadmill training did not improve gait speed. The pooled mean difference (MD, random‐effects model) for gait speed was 0.01; 95% confidence interval (CI) ‐0.19 to 0.22; P = 0.90; level of heterogeneity I2= 60%) at the end of study (Analysis 2.2).
Subgroup analysis
Although initially planned, we decided to do only one sensitivity analysis (Analysis 2.2) instead of a formal subgroup analysis, due to limited number of studies and limited detailed information (Differences between protocol and review).
Discussion
Summary of main results
The aim of this review, which included 18 trials with a total of 623 participants, was to evaluate the effects of treadmill training on gait in patients with PD. We found evidence that the use of treadmill training may improve gait parameters, such as gait speed and stride length, of patients with PD at Hoehn Yahr stages one to three. However, walking distance and cadence did not improve [BJ1] significantly. Additionally, it is not known how long gait improvements after treadmill training may last. Adverse events and drop‐outs did not occur more frequently in people receiving treadmill training than control interventions and were not judged to be clinically serious adverse events.
Overall completeness and applicability of evidence
The results of this review seem to be quite generalisable to both in and outpatient settings in industrialised countries. More specifically our results may not apply to an assumed average older patient with Parkinson's disease or patients with Hoehn & Yahr stages higher than 3. The results may therefore not be broadly generalisable to more severe or older patients. There are factors producing uncertainty for generalisations: 1. The investigated study population was quite heterogeneous (e.g. stage of disease, age, duration of illness, and walking ability). 2. The investigated experimental and control conditions were heterogeneous (e.g. type of training, frequency and duration of training; some studies had no real 'active' control group and some compared treadmill training with no active therapy). Hence, the results may be of limited applicability for all people with PD.
One potential limitation could be that only gait parameters were considered in this update of our review. More general patient‐reported scales as UPDRS, quality of life scales (e.g. PDQ‐39) and health economics outcomes were not included neither in our protocol for this review (Mehrholz 2009) nor in this update of our review. The inclusion of such an analysis may be interesting, but would be beyond the scope of this update. Additionally the analysis of outcomes other than gait parameters was hardly possible because only a small amount of studies used such scales (e.g. only 4 out of 18 included studies described UPDRS total scores and subscale scores at baseline and at study end ; see Table 4). Eventually, the results of this review are only applicable to gait parameters of people with PD. However, gait hypokinesia is one of the primary movement disorders associated with PD(Morris 2000) and an important determinant of activities and quality of life in mild to moderate Parkinson disease (Muslimovic 2008).
We were not able to find any description of adverse events of treadmill training. It is not clear whether adverse events were not reported or did not occur.
The lack of long‐term follow‐up in more than the half of included trials might be a crucial point, but PD is a progressive condition and therefore benefits are not expected to be longlasting (Table 4). The small but clinical benefit may well have disappeared by after 3 to 12 months. However, from the results of our review it is unclear how much of the short term benefit will lasting for how long. Studies investigating the lasting of effects of treadmill training or studies of re‐intervention are therefore warranted.
Quality of the evidence
We presented the quality of evidence for our outcomes in Table 1.
We found heterogeneity between the trials in terms of trial design (two, three, or four arms; parallel group or cross‐over trial; duration of follow up; selection criteria for patients), characteristics of the therapy interventions (especially frequency and duration of intervention), and participant characteristics (Hoehn Yahr severity at baseline), but it is not clear whether this limited the quality of the evidence.
Although the methodological quality of the included trials seemed generally good to moderate (Figure 2), trials investigating treadmill training are subject to potential methodological limitations; for example;
inability to blind the therapist and participants, so‐called contamination (provision of the intervention to the control group) and co‐intervention (when the same therapist unintentionally provides additional care to either treatment or comparison group). All these potential methodological limitations introduce the possibility of performance bias, even though not supported by our sensitivity analyses of methodological quality (Analysis 2.1).
Potential biases in the review process
A risk of publication bias is present in all systematic reviews. However, we searched extensively for relevant literature in databases and trial registers and handsearched reference lists and conference abstracts. Additionally, we contacted and asked authors, trialists and experts in the field for information on other unpublished and ongoing trials. No statistical or graphical evidence for publication bias has been found (Figure 3 and Figure 4).
3.
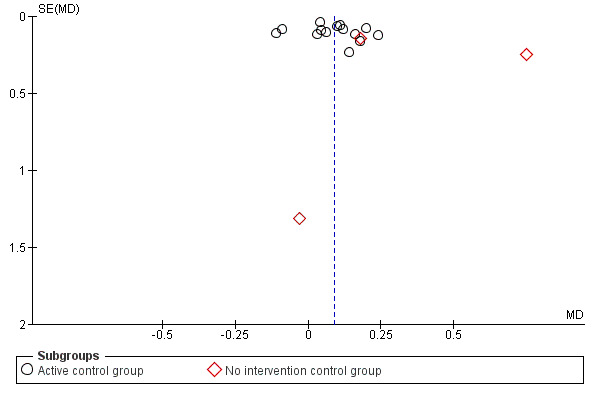
Funnel plot of comparison: 1 Treadmill training versus no treadmill training or active control intervention or gait training, outcome: 1.1 Gait speed at the end of the study.
4.
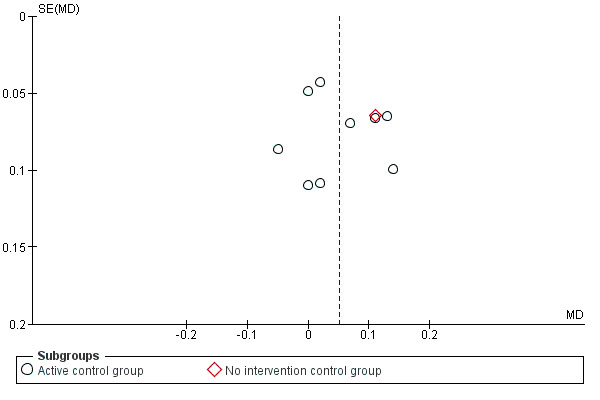
Funnel plot of comparison: 1 Treadmill training versus no treadmill training or active control intervention or gait training, outcome: 1.2 stride length (at the end of study; all studies).
One could argue that the clinical diversity of included trials with respect to duration and frequency of intervention and content of the control group could compromise a pooled analysis. The analyses of the primary outcome however did not reveal statistically heterogeneity (Analysis 1.1). Lastly, our aim was to provide a systematic overview about the current evidence and decided to pool the data of all available trials in a formal meta‐analysis.
The exclusion of patient groups, such as those with unstable cardiovascular conditions, cognitive and communication deficits and a limited range of joint motion at the start of the intervention may limit applicability of the findings to these groups.
However, using the results from the primary outcomes it is possible to explore the apparent effectiveness of treadmill training for improving gait in patients with PD. It might be important to consider that treadmill training might be just one way to apply many repetitions of gait cycles. However, one could argue that the gait training provided by a treadmill will lead to better results because people are forced to use higher gait speeds than over ground, as recently shown in one included study. In this study of Pohl and co‐workers, patients with PD were able to walk up to three times faster on a treadmill than over ground (Pohl 2003). Gait training on a treadmill could be seen as a 'forced‐use‐therapy, because patients are forced to use faster gait cycles and therefore higher velocities as they would self‐select over ground.
The trials included explored quite different training programs and used different intensities and doses of therapy (see Table 2). For example one could argue, that the studies of Pohl 2003 and Cakit 2007 and also Harro 2014 and Protas 2005 are somewhat different from all other included trials, because a rigorous and systematic speed increments approach was used (see Table 2).
These trials were therefore somewhat different in terms of duration of training and intensity of training and effect. For instance the investigators of the Cakit 2007 trial used a speed depended treadmill approach with increments of belt speed until the highest walking speed at which the patient could walk safely (similar to the 'speed‐depended walking' approach as described firstly in Pohl 2003) and trained patients for eight weeks. The study of Cakit 2007 is therefore, compared to all other studies very long and used a very intensive training paradigm. It is therefore that this at most lasting and very intensive training program results in the largest effects compared to all other studies(see Analysis 1.1; see Table 2 and Table 3)
However after excluding the studies of Pohl 2003 and Cakit 2007 from the pooled analysis (not figured), our main effects for gait speed were still present. According to our predefined inclusion and exclusion criteria (Mehrholz 2009), and in an effort to find all randomised controlled trials on treadmill training, we decided to include these studies.
We analysed only the type of treadmill protocol used in studies as part of an analysis to explore the influence of the intensity of treadmill training. The influence of specific training parameters such duration, frequency, and intensity of treadmill training on the gait parameters of patients with PD will be the subject of further evaluation in our next update, when more studies are available.
Treadmill training has the potential to increase the number of repetitions of practice. It is important to mention however that not all of the included studies had an active control group with matched number of repetitions of practice as in the experimental group. Also the co‐interventions varied greatly. In one study it was unclear what intervention the control group received (Cakit 2007). One could argue that these variations in the control interventions would lead to bias and may therefore overestimate the effect sizes, which seems clinically meaningful.
We were not aware of missing data and analysed the available data according to the Cochrane Handbook (chapter 16.1.2 General principles for dealing with missing data). We described the risk of bias due to missing data in Risk of bias in included studies (see also Figure 2). We assumed that if missing data have occurred that these data missing was at random. It is not clear how this can the results of our review biased.
Agreements and disagreements with other studies or reviews
At the time of writing the protocol for this Cochrane review we were not aware of any systematic reviews about the topic (Mehrholz 2009). However, we have found a review by Herman et al which included randomised controlled and non‐controlled studies on treadmill training in PD (Herman 2008). Although Herman et al gave a comprehensive overview of all the randomised studies we found, a pooled analysis for a possible treatment effect was not done. Additionally, descriptions of patient acceptance and side effects of treadmill training in PD were not conveniently provided. According to our protocol (Mehrholz 2009), and with the intention of reducing possible sources of bias, we only included randomised controlled trials.
The authors of the review of Herman 2008 reached in the end the conclusion that 'high quality randomized controlled studies are needed before TT can be recommended with evidence based support'. Our review from 2014 includes now 18 RCTs and more than 50% of them have a low risk of bias. We might conclude based on relatively precise estimators that there is evidence that the use of treadmill training in patients with PD may improve gait parameters such as gait speed, stride length and walking distance.
Another review about physiotherapy intervention in Parkinson's disease by Tomlinson 2012 found and described the effects of eight studies compared to 18 studies in our review. The authors included in their quantitative analysis (about the effects of treadmill training) three studies with only 56 patients and estimated the treatment effect of treadmill training on gait speed with a mean difference of 0.04 m/s (Tomlinson 2012). In our review we, however, included 18 trials with 623 patients and reached a more precise effect estimation compared to (Tomlinson 2012).
The authors of the review of Tomlinson 2012 concluded that most of the observed differences between the treatments were small or for some outcomes (e.g. velocity), the differences observed were at, or approaching, what are considered minimally clinical important changes. Our conclusions are in the same line: the effect of treadmill training on gait speed might be considered as minimally clinical important. For example the benefit in walking speed of treadmill training over no or no active control intervention was a large and clinical important difference of 0.4 m/s (mainly due to Cakit 2007), while the benefit in walking speed of treadmill training over 'conventional physiotherapy' was 0.07 m/s. The latter benefit is quite lower but still a minimal clinical important difference. Eventually, this benefit is observed both with and without gait training in the control group.
Whereas benefits of gait speed can be considered to be close to the minimally clinical important difference, it should be argued that such small change in gait speed would not be automatically be seen as relevant to the general public, administrators and policy makers though.
Another up to date Cochrane review about physical therapies versus active interventions (Tomlinson 2014) should also be mentioned here. This review investigated physiotherapy interventions and rated all interventions into one of the six categories (general physiotherapy, exercise, treadmill training, cueing, dance and martial arts). On the one hand the review of Tomlinson 2014 used compared to our review a greater gamut of outcome measures e.g. UPDRS and quality of life measures and described not just gait parameters as we did. On the other, we found and included seven randomised controlled trials more (Bello 2013; Canning 2012; Carda 2012; Harro 2014; Sale 2013; Shulman 2013; Pohl 2003) than the group of Tomlinson et al. about treadmill training (six out of these seven RCTs were not found with their search). We believe therefore that our review is more specific and used a more sensitive search.
This update of our Cochrane review seems therefore to our knowledge the most up to date systematic review about treadmill training in people with PD with a pooled estimate of treatment effects and patient acceptance.
Additionally our review seems to have the most robust and strongest recommendations for treadmill training for patients with PD so far.
Authors' conclusions
Implications for practice.
This systematic review provides evidence from a number of trials with moderate to good risk of bias that the use of treadmill training in patients with PD may improve clinically relevant gait parameters such as gait speed and stride length (high to moderate and moderate quality of evidence, respectively). This apparent benefit for patients is, however, not supported by all secondary variables (e.g. walking distance, cadence). In practice when treadmill training is available this technology might be used in relatively young and fit people with PD to improve gait speed as one specific parameter of gait hypokinesia.
Implications for research.
There is still a need for well‐designed large‐scale studies to evaluate benefits of different parameters and about the frequency of treadmill training in patients with PD. Further research should address specific questions about duration of effect, frequency, training parameters and duration of treadmill training. Future research should investigate the long‐term benefits of treadmill training, should investigate how often, how long and at which speed treadmill training should be done to establish a dose response relationship.
What's new
| Date | Event | Description |
|---|---|---|
| 7 August 2015 | New citation required and conclusions have changed | We have updated the searches to September 2014, and have revised the text as appropriate. We have included 18 trials with 633 participants in this major update compared with 8 trials with 203 participants in the last version of this review from 2009. The conclusion has been changed. Int his version we conclude that 'It seems that the use of treadmill training could be beneficial with comparable risk as conventional therapies.' |
Acknowledgements
We thank grateful Professor Jane Jane Burridge for her very helpful comments and for her great job on proofreading the manuscript of the review. We thank Gabi Voigt for providing us with many helpful studies. We thank all authors and investigators who provided us with additional or unpublished information or data about their trials.
Appendices
Appendix 1. Example for MEDLINE search through OVID gateway
Example for search in Medline through OVID Gateway: 1.Parkinson$.tw. 2.exp Parkinsonian Disorders/ 3.1 or 2 4.Treadmil$.tw. 5.Exercise Test/ 6.Exercise Therapy/ 7.Physical Therapy Modalities/ 8.Motor Activity/ 9.Walking/ 10.Periodicity/ 11.or/4‐10 12.randomized controlled trial.pt. 13.controlled clinical trial.pt. 14.randomized controlled trials/ 15.random allocation/ 16.double?blind method/ 17.single?blind method/ 18.clinical trial.pt. 19.clin$ with trial$.tw. 20.random$.tw. 21.exp research design/ 22. or/12‐21 23. 3 and 11 and 22 24.limit 23 to animal 25. 23 not 24
Number of hits retrieved: 579
Appendix 2. Example for Embase search through OVID gateway
Example for search in EMBASE through OVID Gateway:
1.Parkinson Disease/ 2.Parkinsonism/ 3.Parkinson$.tw. 4.1 or 2 or 3 5.Treadmill.tw. 6.Exercise adj5 test.tw. 7.((exercise or physical) adj5 (therapy)).tw. 8.5 or 6 or 7 9.clinical trial/ 10.multicenter study/ 11.phase 2 clinical trial/ 12.phase 3 clinical trial/ 13.phase 4 clinical trial/ 14.randomized controlled trial/ 15.controlled study/ 16.meta analysis/ 17.double blind procedure/ 18.single blind procedure/ 19.randomization/ 20.major clinical study/ 21.placebo/ 22.drug comparison/ 23.clinical study/ 24.(clin$ adj25 trial$).tw. 25.((singl$ or doubl$ or tripl$ or trebl$) adj25 (blind$ or mask$)).tw. 26.random$.tw. 27.control$.tw. 28.or/9‐27 29. 4 and 8 and 28 30. limit 29 to animal 31. 29 not 30
Number of hits retrieved: 361
Appendix 3. Example for CENTRAL search strategy
#1(PD or IPD) #2(Parkinson? next/5 Diseas?) #3MeSH descriptor: [Parkinson Disease] explode all trees #4#1 or #2 or #3 #5[mh ^exercise] or [mh ^"exercise test"] or [mh ^"exercise therapy"] or [mh ^"motion therapy, continuous passive"] #6[mh ^"body weight"] or [mh ^weight‐bearing] #7treadmill* or tread next mill* or running next wheel* or running next machine* #8(walking or walk or exercise) near/5 (machine* or device*) #9(walking or gait or locomotor or ambulation) near/5 (train* or re‐train* or retrain*) #10[mh ^walking] #11machine* or device* or train* or re‐train* or retrain* #12#10 and #11 #13(weight or "body‐weight" or bodyweight) near/5 (support* or suspen* or relief) #14(walk or walking or ambulat* or locomot* or gait or overhead) near/5 support* #15harness* #16#5 or #6 or #7 or #8 or #9 or #12 or #13 or #14 or #15 #17[mh ^walking] or [mh ^gait] or [mh ^"mobility limitation"] or [mh ^locomotion] #18walk* or gait* or ambulat* or mobil* or locomot* or stride #19#17 or #18 #20#4 and #16 and #19
Number of hits retrieved: 153
Appendix 4. Example for PEDro search
Abstract & Title: parkinson treadmill Therapy: fitness training Subdiscipline: neurology Method: clinical trial (Search terms matched with AND)
Number of records retrieved: 7
Appendix 5. List of conference proceedings searched
World Congress of NeuroRehabilitation;
World Congress of Physical Medicine and Rehabilitation;
World Congress of Physical Therapy ;
World Congress of Neurology;
World Congress on Parkinson's Disease and Related Disorders;
Deutsche Gesellschaft für Neurotraumatologie und Klinische Neurorehabilitation;
Deutsche Gesellschaft für Neurologie;
Deutsche Gesellschaft für Neurorehabilitation.
Data and analyses
Comparison 1. Treadmill training versus no treadmill training or active control intervention or gait training.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Gait speed at the end of the study | 17 | 510 | Mean Difference (IV, Random, 95% CI) | 0.09 [0.03, 0.14] |
| 1.1 Active control group | 14 | 434 | Mean Difference (IV, Random, 95% CI) | 0.07 [0.03, 0.12] |
| 1.2 No intervention control group | 3 | 76 | Mean Difference (IV, Random, 95% CI) | 0.40 [‐0.06, 0.87] |
| 2 stride length (at the end of study; all studies) | 10 | 333 | Mean Difference (IV, Random, 95% CI) | 0.05 [0.01, 0.09] |
| 2.1 Active control group | 9 | 315 | Mean Difference (IV, Random, 95% CI) | 0.04 [0.00, 0.09] |
| 2.2 No intervention control group | 1 | 18 | Mean Difference (IV, Random, 95% CI) | 0.11 [‐0.02, 0.24] |
| 3 walking distance in m (at the end of study; all studies) | 10 | 416 | Mean Difference (IV, Random, 95% CI) | 48.91 [‐1.32, 99.14] |
| 3.1 Active control group | 9 | 385 | Mean Difference (IV, Random, 95% CI) | 9.48 [‐0.47, 19.42] |
| 3.2 No intervention control group | 1 | 31 | Mean Difference (IV, Random, 95% CI) | 364.0 [294.45, 433.55] |
| 4 cadence (at the end of study; all studies) | 10 | 336 | Mean Difference (IV, Random, 95% CI) | 2.16 [‐0.13, 4.46] |
| 4.1 Active control group | 9 | 318 | Mean Difference (IV, Random, 95% CI) | 2.42 [0.07, 4.77] |
| 4.2 No intervention control group | 1 | 18 | Mean Difference (IV, Random, 95% CI) | ‐4.0 [‐15.11, 7.11] |
| 5 acceptability and safety of treadmill training | 18 | 633 | Risk Difference (M‐H, Random, 95% CI) | ‐0.02 [‐0.06, 0.02] |
| 5.1 Active control group | 15 | 531 | Risk Difference (M‐H, Random, 95% CI) | ‐0.01 [‐0.05, 0.03] |
| 5.2 No intervention control group | 3 | 102 | Risk Difference (M‐H, Random, 95% CI) | ‐0.14 [‐0.43, 0.16] |
Comparison 2. Sensitivity analysis: Treadmill training versus no treadmill training.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Gait speed | 17 | Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 1.1 All studies | 16 | 488 | Mean Difference (IV, Random, 95% CI) | 0.10 [0.05, 0.15] |
| 1.2 All studies with random allocation | 8 | 237 | Mean Difference (IV, Random, 95% CI) | 0.08 [0.02, 0.13] |
| 1.3 all studies with concealed allocation | 8 | 237 | Mean Difference (IV, Random, 95% CI) | 0.08 [0.02, 0.13] |
| 1.4 All studies with blinded assessors | 12 | 375 | Mean Difference (IV, Random, 95% CI) | 0.07 [0.00, 0.13] |
| 2 Gait speed | 17 | 510 | Mean Difference (IV, Random, 95% CI) | 0.09 [0.03, 0.14] |
| 2.1 treadmill protocols using a speed dependent approach | 4 | 88 | Mean Difference (IV, Random, 95% CI) | 0.16 [‐0.08, 0.40] |
| 2.2 treadmill protocols with gradual speed increases | 8 | 227 | Mean Difference (IV, Random, 95% CI) | 0.08 [0.02, 0.14] |
| 2.3 treadmill protocols with constant walking speed | 3 | 85 | Mean Difference (IV, Random, 95% CI) | 0.12 [0.02, 0.22] |
| 2.4 studies using a mixed or different approaches or did not manipulated gait speed | 2 | 110 | Mean Difference (IV, Random, 95% CI) | 0.01 [‐0.19, 0.22] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Bello 2013.
| Methods | Randomised controlled trial Method of randomisation: not described Blinding of outcome assessors: not described Adverse events: not described Deaths: not described Drop‐outs: not stated ITT: not stated | |
| Participants | Country: Spain
22 patients (11 in treatment group, 11 in control group)
Ambulatory at study onset: yes
Mean age: 58 to 59 years (control and treatment group respectively) Inclusion criteria: being able to walk for 10 min without stopping, walking aids or assistance (on medication) Exclusion criteria: history of neurological conditions other than PD, orthopedic, or visual disturbances which affected walking ability and signs of cardiovascular or autonomic dysfunction |
|
| Interventions | 2 arms: (1) control group used overground gait training, 3 times a week for 5 weeks (72 min a week) (2) experimental group received treadmill training without BWS, 3 times a week for 5 weeks (72 min a week) | |
| Outcomes | Outcomes were recorded at baseline and at the end of intervention phase Unified Parkinson’s Disease Ranking Scale (UPDRS) Motor Score Measures of timed gait (walking speed, cadence, stride length) at preferred and at maximal speed Timed Up‐and‐Go test (TUG) Posturography Knee extensor muscle strength | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | method of sequence generation not described by the authors |
| Allocation concealment (selection bias) | Unclear risk | method not described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | blinding not described by the authors |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | no missing outcome data described |
Cakit 2007.
| Methods | Randomised controlled trial Method of randomisation: not described | |
| Participants | Country: Turkey Sample size: 54 participants (27 in treatment group, 27 in control group) Inclusion criteria: medically stable; able to walk a 10m distance; able to give informed consent Exclusion criteria: neurological conditions other than PD; scored greater than 3 on the Hoehn and Yahr Disability Scale; scoring less than 20 Mini‐Mental State Examination; postural hypotension; cardiovascular or musculoskeletal disorder; visual or vestibular disturbance | |
| Interventions | 2 arms (1) training group: 8 weeks exercise programme including stretching, range of motion exercise and treadmill training with incrementally increasing belt speed (2) control group: 8 weeks not described further | |
| Outcomes | Outcomes were recorded at baseline and after 8 weeks of therapy and included
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | method of sequence generation not described by the authors |
| Allocation concealment (selection bias) | Unclear risk | method not described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | described as blinded to group assignment seemingly |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | reasons for loss to follow‐up apparently not related to the intervention |
Canning 2012.
| Methods | Randomised controlled trial Method of randomisation: sealed opaque envelopes Blinding of outcome assessors: yes Adverse events: none Deaths: none Drop‐outs: 3 (2 from the EXP group and 1 from the CTL group) ITT: yes | |
| Participants | Country: Australia
20 patients (10 in treatment group, 10 in control group)
Ambulatory at study onset: yes
Mean age: 61 to 63years (treatment and control group respectively) Inclusion criteria: Hoehn and Yahr stages 1 or 2, age between 30 and 80 years, <2 h of leisure activity per week, stable response to levodopa, subjective gait disturbance Exclusion criteria: disabling dyskinesias or motor fluctuations; freezing while ‘ON’ medication; or significant balance impairment, Mini‐Mental State Examination Score <24, history of falls or dizziness, other neurological/ musculoskeletal/cardiopulmonary or metabolic conditions that affected walking |
|
| Interventions | 2 arms (1) experimental group: 6 weeks home based treadmill walking, 30‐40 minutes a day, 4 times a week, 7 of 24 sessions supervised by physiotherapist (2) control group: 6 weeks usual care including maintaining usual physical activity levels | |
| Outcomes | Outcomes were recorded at baseline, after 6 weeks of therapy and after 12 weeks after baseline and included Primary outcome measure:
Secondary outcome measures:
Feasibility outcomes:
|
|
| Notes | This is the same study (now published as full text) as in our former review described as Canning 2008. The new reference is therefore Canning 2012 | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: “After baseline assessment, a staff member who was not involved in the trial randomly allocated participants to the treadmill training or control group using opaque envelopes pre‐prepared by one investigator” |
| Allocation concealment (selection bias) | Low risk | Quote: “After baseline assessment, a staff member who was not involved in the trial randomly allocated participants to the treadmill training or control group using opaque envelopes pre‐prepared by one investigator” |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote:”Efficacy outcome measures were made by an assessor blinded to group allocation” |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Missing data was balanced between groups and an intention‐to‐treat analysis has been performed by the authors |
Carda 2012.
| Methods | Randomised controlled trial Method of randomisation: software‐generated randomisation list Blinding of outcome assessors: yes Adverse events: none Deaths: none Drop‐outs: 2 (1 in EXP and 1 in CTL) ITT: yes | |
| Participants | Country: Italy 30 patients (15 in treatment group, 15 in control group) Ambulatory at study onset: yes Mean age: 67 to 68 years (treatment and control group respectively) Inclusion criteria: diagnosis of PD according to the UK Brain Bank Criteria, disease stage <III according to the classification of Hoehn and Yahr without motor fluctuations, being able to ambulate independently Exclusion criteria: treadmill training or other form of specific gait training for at least 6 months before the study, treadmill training or other form of specific gait training for at least 6 months before the study, body weight more than 100 kg; respiratory disease; other neurological diseases; dementia; depression; or uncorrected visual disturbances; undergone or planned deep brain stimulation in the following 6 months | |
| Interventions | 2 arms: (1) control group used robotic gait training, 3 times a week for 4 weeks (120 min a week) (2) experimental group received treadmill training, 3 times a week for 4 weeks (120 min a week) | |
| Outcomes | Outcomes were recorded at baseline and at the end of intervention phase Primary outcome: 6 Minute walk test Secondary outcome: 10‐m walk test Timed Up‐and‐Go test Unified Parkinson’s Disease Ranking Scale (UPDRS) Motor Score Global health status (SF‐12 questionnaire) | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | A software‐generated randomisation list was used |
| Allocation concealment (selection bias) | Low risk | A researcher not involved in the experiment checked for correct patient allocation prior and after the study |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: “A physical therapist who was not involved in the treatment of the enrolled patients and who was blinded to treatment allocation performed all outcome assessments.” |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Missing data was balanced between groups and an intention‐to‐treat analysis has been performed by the authors |
Chaiwanichsiri 2011.
| Methods | Randomised controlled trial Method of randomisation: not described Blinding of outcome assessors: yes Adverse events: none Deaths: none Drop‐outs: none ITT: yes | |
| Participants | Country: Thailand
30 patients (10 in treatment group 1, 10 in treatment group 2, 10 in control group)
Ambulatory at study onset: yes
Mean age: 68 to 69 years (treatment and control group respectively)
Inclusion criteria: Male sex, aged 60 to 80 years, diagnosed by neurologists as idiopathic PD, Hoehn and Yahr stage 2‐3, good cognitive function on Thai Mental State Examination (TMSE) score >23, stable symptoms with unmodified anti‐parkinsonian medication during the study, independent ambulation without using any gait Aids, good vision and hearing Exclusion criteria: other medical conditions that could interfere with the training program, participating in any other training program |
|
| Interventions | 3 arms: (1) control group used a home walking program, 6 times a week for 4 weeks (180 min a week) (2) experimental group 1 received a home walking program 3 times a week and treadmill training with music cues 3 times a week for 4 weeks (180 min a week) (3) experimental group 2 received a home walking program 3 times a week and treadmill training 3 times a week for 4 weeks (180 min a week) | |
| Outcomes | Outcomes were recorded at baseline and at the end of intervention phase Step length Stride length Cadence 6‐m walk test Walking speed Timed Up and Go Test (TUG) | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | method of sequence generation not described by the authors |
| Allocation concealment (selection bias) | Unclear risk | method not described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: “All participants were assessed by two physicians and one research assistant, who were blinded to group assignment.” |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | no missing outcome data |
Fisher 2008.
| Methods | Randomised controlled trial Method of randomisation: patients self selected a card with eyes closed | |
| Participants | Country: USA
Sample size: 30 participants (10 in high‐intensity exercise group, 10 in low‐intensity group, and 10 in zero‐intensity group)
Inclusion criteria: diagnosis of PD within 3 years of study participation; 18 years of age or older; medical clearance from the primary care physician to participate in an exercise program; and ability to walk Exclusion criteria: a score of less than 24 on the MMSE; physician‐determined major medical problems such as cardiac dysfunction; musculoskeletal impairments or excessive pain in any joint that could limit participation in an exercise program; and insufficient endurance and stamina to participate in exercise 3 times a week for a 1‐hour session |
|
| Interventions | 3 arms
(1) high‐intensity exercise group: body weight supported treadmill walking, up to 45 minutes a day, for 24 supervised sessions in 8 weeks
(2) low‐intensity group: general or traditional physiotherapy, for 24 sessions in 8 weeks (3) zero‐intensity (no‐exercise) group: six 1 hour education class over 8 weeks |
|
| Outcomes | Outcomes were recorded at baseline, after 8 weeks of therapy and included
|
|
| Notes | We analysed the high intensity group (1) with low‐intensity group (2) and zero‐intensity group (3) (we collapsed groups 2 and 3 to one pooled control group as in our former version of this review of Mehrholz 2009). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | patients self selected a card with eyes closed |
| Allocation concealment (selection bias) | Low risk | Patients were allocated to groups by self selecting a card with eyes closed |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | a blinded assessor was used |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | no missing outcome data |
Frazzitta 2009.
| Methods | Randomised controlled trial Method of randomisation: not described Blinding of outcome assessors: unclear Adverse events: not stated Deaths: none Drop‐outs: none ITT: yes | |
| Participants | Country: Italy 40 patients (20 in treatment group, 20 in control group) Ambulatory at study onset: yes Mean age: 71 years (control and treatment group) Inclusion criteria: being able to walk without any physical assistance, sufficient vision and hearing, freezing of gait during peak medication (confirmed by clinical examination), Hoehn & Yahr stage 3, Mini Mental State Examination Score >26), constant medication Exclusion criteria: neurological conditions other than idiopathic Parkinson’s disease, postural hypotension, cardiovascular, musculoskeletal, or vestibular disorders limiting locomotion or balance | |
| Interventions | 2 arms: (1) control group used traditional rehabilitation with visual and auditory cues, 7 times a week for 4 weeks (140 min a week) (2) experimental group received treadmill training with visual and auditory cues, 7 times a week for 4 weeks (140 min a week) | |
| Outcomes | Outcomes were recorded at baseline and at the end of intervention phase
Unified Parkinson’s Disease Ranking Scale (UPDRS) Motor Score
Freezing of Gait Questionnaire (FOGQ) 6‐min walk test (distance walked) Gait speed Stride length |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | method of sequence generation not described by the authors |
| Allocation concealment (selection bias) | Unclear risk | Not described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | all patients were assessed by same neurologist; no blinding described |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | no missing outcome data |
Harro 2014.
| Methods | Randomised controlled trial Method of randomisation: no further description in publication by the authors | |
| Participants | Country: USA
Sample size: 22 participants (11 in speed treadmill training group, and 11 in control group)
Inclusion criteria: age of 18–89 years, diagnosis of idiopathic PD, stage 1–3 on the Hoehn and Yahr scale, ability to walk continuously without physical assistance for five minutes with or without an assistive device, stable PD medication schedule and dosing over past month as reported by the participant’s neurologist and functional vision and hearing sufficient to perceive cues with or without aides/glasses Exclusion criteria: impaired cognitive functioning evidenced by a score of 20 or less on the Saint Louis Mental Status Examination, history of other neurologic or vestibular disorders, current orthopedic conditions that would affect the ability to walk, history of PD‐related deep brain stimulation, inability to speak and read English, and unstable medical status and inability to engage in moderate exercise |
|
| Interventions | 2 arms (1) treadmill training group: 6 weeks supervised speed dependent treadmill walking, 30 minutes a session, 3 times a week (2) control group: 6 weeks rhythmic auditory‐cueing in small groups of five participants, 30 minutes a session, not described how often a week | |
| Outcomes | Outcomes were recorded at baseline, after 6 weeks and after 3 months and included:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | not described |
| Allocation concealment (selection bias) | Unclear risk | not described |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | described as blinded assessors |
| Incomplete outcome data (attrition bias) All outcomes | High risk | not all included participants were analysed |
Kurtais 2008.
| Methods | Randomised controlled trial Method of randomisation: no further description in publication by the authors | |
| Participants | Country: Turkey
Sample size: 30 participants (15 in treadmill training group, and 15 in control group)
Inclusion criteria: stable medication, not participated in a rehabilitation programme in the previous 3 months Exclusion criteria: severe cognitive impairment; severe musculoskeletal cardiopulmonary or other systemic disorders |
|
| Interventions | 2 arms (1) treadmill training group: 6 weeks supervised treadmill walking, 40 minutes a session, 3 times a week (2) control group: not further described by the authors | |
| Outcomes | Outcomes were recorded at baseline, after 7 weeks and included:
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation list (personal communication) |
| Allocation concealment (selection bias) | Low risk | generated list was used by an independent person to allocate participants (personal communication) |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | assessed and tested during "on" phase by the authors who were blind to the randomization |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Missing data was balanced between groups and an intention‐to‐treat analysis has been performed by the authors |
Miyai 2000.
| Methods | Randomised cross‐over trial Method of randomisation: no further description | |
| Participants | Country: Japan Sample size: 10 participants (5 in treadmill training group, and 5 in control group, before first cross over) Inclusion criteria: Hoehn and Yahr stage 2.5 to 3, MMSE greater than 27 | |
| Interventions | 2 arms (1) treadmill training group: 4 weeks body weight supported treadmill training, 45 minutes a day, 3 days a week (2) control group: 4 weeks conventional physiotherapy, 45 minutes a day, 3 days a week | |
| Outcomes | Outcomes were recorded at baseline, after 4 weeks and included
|
|
| Notes | Raw data kindly provided by the authors were used for all analyses | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | method of sequence generation not described by the authors |
| Allocation concealment (selection bias) | Unclear risk | not described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | no blinding |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | no missing outcome data |
Miyai 2002.
| Methods | Randomised controlled trial Method of randomisation: not described by the authors | |
| Participants | Country: Japan
Sample size: 24 participants (12 in treadmill training group, and 12 in control group)
Inclusion criteria: diagnosis of PD, Hoehn and Yahr stage 2.5 to 3, MMSE greater than 27 Exclusion criteria: on‐off phenomenon |
|
| Interventions | 2 arms (1) treadmill training group: 4 weeks body weight supported treadmill training, 45 minutes a day, 3 days a week, with a total of 12 sessions (2) control group: 4 weeks conventional physiotherapy, 45 minutes a day, 3 days a week, with a total of 12 sessions | |
| Outcomes | Outcomes were recorded at baseline, after 1, 2, 3, 4, 5 and 6 months and included
|
|
| Notes | Raw data kindly provided by the authors were used for all analyses Because the details of the studies of Miyai 2000 and Mixai 2002 looks similar at a first look, we contacted the lead Author Prof. Miyai. He clearly stated that these trials are dissimilar and involve different patients. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | method of sequence generation not described by the authors |
| Allocation concealment (selection bias) | Unclear risk | not described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | no blinding |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | reasons form missing outcome data unlikely to be related to true outcomes |
Nadeau 2013.
| Methods | Randomised controlled trial Method of randomisation: computer‐generated randomisation sequence Blinding of outcome assessors: yes Adverse events: none Deaths: none Drop‐outs: 9 (7 in experimental groups, 2 in control group) ITT: no | |
| Participants | Country: Canada 93 patients (29 in treatment group I, 30 in treatment group II, 34 in control group) Ambulatory at study onset: not stated Mean age: 62 to 64 years (treatment and control group respectively) Inclusion criteria: not clearly stated except idiopathic PD and living up to 45 min away from the study centre Exclusion criteria: major health problem (cancer, heart/lung problems) | |
| Interventions | 3 arms: (1) control group used low exercise intensity training in seated position, 3 times a week for 24 weeks (180 min a week) (2) experimental group I received incremental speed treadmill training, 3 times a week for 24 weeks (180 min a week) (3) experimental group II received (mixed treadmill training) incremental speed treadmill training with additional incremental treadmill inclination, 3 times a week for 24 weeks (180 min a week) | |
| Outcomes | Outcomes were recorded at baseline, at halving interval at 3 months and at the end of intervention phase at 6 months
Walking speed (GAITRite)
Stride length (GAITRite)
Cadence (GAITRite)
Step width (GAITRite)
Gait capacity (6‐min walk test)
Unified Parkinson’s Disease Rating Scale (UPDRS)
Depression Beck (Depression Inventory – II (BDI‐II)) Parkinson’s Disease Questionnaire (PDQ) Balance Confidence Scale Exercise intensity Exercise adherence Exercise‐related adverse events |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation sequence |
| Allocation concealment (selection bias) | Low risk | Quote: “The allocation sequence was concealed from the project director who assigned participants to groups.” |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: “Participants and research assistants performing the assessments were blind to group assignment” |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | missing outcome data imbalanced between groups but not directly attributable to the intervention; no intention‐to‐treat analysis performed |
Picelli 2013.
| Methods | Randomised controlled trial Method of randomisation: randomisation list used Blinding of outcome assessors: done Adverse events: none during study period Deaths: none Drop‐outs: none ITT: yes | |
| Participants | Country: Italy
60 patients (20 in robotic gait training group, 20 in treadmill training group and 20 in Physical Therapy group)
Ambulatory at study onset: yes
Mean age: 68 years (control and treatment group)
Inclusion criteria: confirmed diagnosis of idiopathic PD according to the UK Brain Bank Criteria; Hoehn and Yahr stage 3 determined in the “on” phase; Mini Mental State Examination
>24. Exclusion criteria: severe dyskinesias or “on‐off” fluctuations; change of PD medication during the study; deficits of somatic sensation involving the lower limbs; vestibular disorders or paroxysmal vertigo; other neurological or orthopedic conditions involving the lower limbs (musculoskeletal diseases, severe osteoarthritis, peripheral neuropathy, joint replacement); cardiovascular comorbidity (recent myocardial infarction, heart failure, uncontrolled hypertension, orthostatic hypotension) |
|
| Interventions | 3 arms:
(1) robotic gait training group, twelve, 45‐min sessions, three days a week for 4 consecutive weeks
(2) treadmill training group, twelve, 45‐min sessions, three days a week for 4 consecutive weeks (3) Physical Therapy group, twelve, 45‐min sessions, three days a week for 4 consecutive weeks |
|
| Outcomes | Outcomes were recorded at baseline and at the end of intervention phase
Gait speed (10m walk test) 6‐min walk test (distance walked) Spatiotemporal gait parameters (e.g. Stride length, cadence) Unified Parkinson’s Disease Ranking Scale (UPDRS) Motor Score Berg Balance Scale Parkinson's Fatigue Scale |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | randomisation list used |
| Allocation concealment (selection bias) | Low risk | allocation concealment by masked investigator |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | blinded rater |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | no missing outcome data |
Pohl 2003.
| Methods | Randomised cross‐over trial Method of randomisation: sealed opaque envelopes | |
| Participants | Country: Germany
Sample size: 17 participants
Inclusion criteria: early PD, defined as Hoehn and Yahr stages I through III; subjective disturbances in gait; stable drug program, and in stable cardiovascular condition Exclusion criteria: paroxysmal motor fluctuations, such as on‐off and wearing‐off phenomena, class B, C, or D exercise risk by the ACSM criteria; cognitive deficits (defined as scores of less than 26 on the MMSE; moderate or severe depression (defined as scores of greater than 17 on the Beck Depression Inventory); and orthopedic and other gait‐influencing diseases such as arthrosis or total hip joint replacement |
|
| Interventions | 4 arms
(1) treadmill training group with incremental speed increase: 1 session treadmill training, 30 minutes (2) treadmill training group without increases of gait speed: 1 session treadmill training, 30 minutes (3) physiotherapy group: 1 session physiotherapy including gait training, 30 minutes (4) control group: resting in a chair for 30 minutes |
|
| Outcomes | Outcomes were recorded at baseline, after 1 session of 30 minutes and included
|
|
| Notes | Raw data of the authors used for all analyses, data of treadmill groups were collapsed in to one group (n=8) and data of physiotherapy and control group were also collapsed into one group (n=9) | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | computer generated list |
| Allocation concealment (selection bias) | Low risk | Sealed opaque envelopes were used for allocation procedure. They contained one of four sequences: 'A', 'B', 'C' and 'D' An assistant blinded to group assignment and not involved in patient recruitment allocated all participants by opening one sealed envelope. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | blinded assessor for gait speed and steps |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | no missing outcome data |
Protas 2005.
| Methods | Randomised cross‐over trial Method of randomisation: not stated by the authors | |
| Participants | Country: USA
Sample size: 18 participants (9 in the treadmill and 9 in the control group)
Inclusion criteria: postural instability‐gait difficulty predominant PD; experiences with freezing episodes, and/or
a history of falls; stable regimen of antiparkinsonian medications; ability to stand and walk without assistance; stage 2 or 3 of the Hoehn and Yahr staging; and scores of moderate or higher on all scales of the Neurobehavioral Cognitive StatusExamination (Cognistat) Exclusion criteria: not used/not described |
|
| Interventions | 2 arms
(1) treadmill training group: treadmill training to improve gait and standing abilities for approximately 30 minutes including forward and backward walking and side stepping, 3 times a week for 8 weeks, 24 sessions of treadmill walking and stepping training (2) control group: no training |
|
| Outcomes | Outcomes were recorded at baseline and after 8 weeks and included
|
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | method of sequence generation not described by the authors |
| Allocation concealment (selection bias) | Unclear risk | not described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "All testing except for the fall record was conducted by a physical therapist and a technician who were blinded to the subject’s group assignment." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | no missing outcome data |
Sale 2013.
| Methods | Randomised controlled trial Method of randomisation: custom computerized system with Lehmer’s algorithm Blinding of outcome assessors: yes Adverse events: not described Deaths: none Drop‐outs: none ITT: yes | |
| Participants | Country: Italy
20 patients (10 in treatment group, 10 in control group)
Ambulatory at study onset: yes
Mean age: 18 to 90 years (control and treatment group respectively)
Inclusion criteria: aged between 18 and 90 years, diagnosis of IPD by UK Brain Bank criteria, capability to walk unassisted or with little assistance for 25 feet walk, unassisted or with little assistance, for 25 feet. Exclusion criteria: other significant neurological or orthopedic conditions, not understanding instructions, primarily wheelchair bound, substance abuse, psychiatric disorders, atypical parkinsonian syndrome, deep brain stimulation |
|
| Interventions | 2 arms: (1) control group used robot‐assisted gait training (device: G‐EO), 5 times a week for 4 weeks (225 min a week) (2) experimental group received treadmill training, 5 times a week for 4 weeks (225 min a week) | |
| Outcomes | Outcomes were recorded at baseline and at the end of intervention phase Primary outcome: walking speed Secondary outcomes: cadence step length stride length step width stance time swing time duration of double support | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | software based sequence generation |
| Allocation concealment (selection bias) | Low risk | allocation concealment done by blinded professionals as described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | blinded professionals as described by the authors |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | no missing outcome data |
Shulman 2013.
| Methods | Randomised controlled trial Method of randomisation: random number generator Blinding of outcome assessors: yes Adverse events: none Deaths: none Drop‐outs: 13 (4 from experimental group I, 3 from experimental group II and 6 from control group) ITT: no | |
| Participants | Country: USA
80 patients (26 from experimental group I, 26 from experimental group II and 28 from control group)
Ambulatory at study onset: not described
Mean age: 65 to 66 years (control and treatment group respectively)
Inclusion criteria: aged 40 and above, diagnosis of PD characterized by asymmetrical onset of at least 2 of 3 cardinal signs, Hoehn & Yahr stage 1 to 3, presence of gait or balance disturbances, Mini‐Mental State Examination >23 Exclusion criteria: unstable medical or psychiatric conditions, aerobic training prior to study enrollment |
|
| Interventions | 3 arms: (1) control group used stretching and resistance training, 3 times a week for 12 weeks (duration of sessions not described) (2) experimental group I received lower intensity treadmill exercise, 3 times a week for 12 weeks (150 min a week) (3) experimental group II received higher intensity treadmill exercise, 3 times a week for 12 weeks (90 min a week) | |
| Outcomes | Outcomes were recorded at baseline and at the end of intervention phase Primary outcome measures: Gait speed (6‐min walk test, 10m walk test) cardiovascular fitness (ergospirometry) muscle strength (1‐repetition maximum strength) | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | method of sequence generation not described by the authors |
| Allocation concealment (selection bias) | Unclear risk | not described by the authors |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | physicians and staff were blinded as described by the authors |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | missing outcome data balanced between groups with similar reasons for missing data across groups |
Yang 2010.
| Methods | Randomised controlled trial Method of randomisation: Sealed opaque envelopes Blinding of outcome assessors: no Adverse events: none Deaths: none Drop‐outs: 3 (2 in the control group and 1 in the experimental group) ITT: no | |
| Participants | Country: Taiwan 33 patients (16 in treatment group, 17 in control group) Ambulatory at study onset: yes Mean age: 66 to 68 years (control and treatment group respectively) Inclusion criteria: diagnosed with IPD by a neurologist as defined by the UK Brain Bank criteria, Hoehn & Yahr stage 1 to 3, independent ambulation, constant medication, ability to understand instructions Exclusion criteria: other conditions limiting exercise | |
| Interventions | 2 arms: (1) control group used conventional therapy, 3 times a week for 4 weeks (90 min a week) (2) experimental group received downhill treadmill training, 3 times a week for 4 weeks (90 min a week) | |
| Outcomes | Outcomes were recorded at baseline and at the end of intervention phase Gait performance (GAITRite) Thoracic kyphosis (electronic goniometer) Muscle strength (handheld dynamometer) | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | sealed envelopes were drawn by an independent arbiter |
| Allocation concealment (selection bias) | Low risk | sealed envelopes were drawn by an independent arbiter |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | no blinding was done |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | missing outcome data balanced between groups with similar reasons for missing data across groups |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Bello 2008 | Not a randomised controlled trial |
| Diaz de la Fe 2008 | Not a randomised controlled trial (personal communication with the authors) |
| Fisher 2013 | Irrelevant outcome measures |
| Ganesan 2010 | Irrelevant outcome measures |
| Gianfrancesco 2009 | Not a randomised controlled trial |
| Schenkman 2012 | Experimental group received treadmill training together with training on a stationary bicycle or elliptical trainer |
Characteristics of studies awaiting assessment [ordered by study ID]
Horak 2011.
| Methods | Randomised controlled trial with parallel group assignment |
| Participants | Estimated enrollment: 40, aged between 50 and 80 years Inclusion criteria: Diagnosis of idiopathic Parkinson's Disease Exclusion criteria: Other neurological conditions, artificial joints |
| Interventions | 2 arms: (1) Treadmill training 4 times a week for 4 weeks with a physical therapist (2) Agility training 4 times a week for 4 weeks with a physical therapist |
| Outcomes | Primary Outcome Measures: Dynamic Posturography Secondary Outcome Measures: UPDRS |
| Notes | This study has been completed. No study results yet posted. |
Mezzarobba 2013.
| Methods | Design: randomised controlled trial Method of randomisation: computer‐generated Blinding of outcome assessors: yes Adverse events: not stated Deaths: not stated Drop‐outs: not stated ITT: unclear |
| Participants | Country: Italy 21 patients (10 in treatment group, 11 in control group) Ambulatory at study onset: unclear Median age: 75 years Inclusion criteria: Hoehn & Yahr stage 1‐3, Mini Mental State Examination Score >24 Exclusion criteria: Beck Depression Inventory score <16 |
| Interventions | 2 arms: (1) control group used motor imagery training for 20 sessions (duration not stated) (2) experimental group received treadmill training for 20 sessions (duration not stated) |
| Outcomes | Outcomes were recorded at baseline, at the end of intervention phase and at 4‐week and at 12‐week follow‐up Disease stage (Hoehn and Yahr scale, Unified Parkinson’s Disease Ranking Scale (UPDRS)) Freezing of Gait (Freezing of Gait Questionnaire) Quality of life (Parkinson’s Disease Questionnaire (PDQ‐39)) Locomotion (Timed Up and Go Test, 6‐minute walk test) Balance (Berg Balance‐scale) Disability (Modified Parkinson’s Activity scale (MPAS)) |
| Notes | Conference abstract |
Characteristics of ongoing studies [ordered by study ID]
NCT01768832.
| Trial name or title | Exercise and Parkinson's: Comparing Interventions and Exploring Neural Mechanisms |
| Methods | Randomised controlled trial with parallel group assignment |
| Participants | Estimated enrollment: 120, aged above 30 years Inclusion criteria: Diagnosis of Parkinson's Disease, at least grade 3/5 strength and normal joint ranges of motion in both legs, good vision, independent ambulation for 10 feet with or without assistive devices, normal gross somatosensory function in the feet Exclusion criteria: Other medical condition with exercise being a contraindication, abnormal brain imaging, evidence or history of other neurological or muscular conditions, failed to pass MRI procedure |
| Interventions | 3 arms: (1) Treadmill training 2 times a week (120 min per week) for 12 weeks (2) Tango dance training 2 times a week (120 min per week) for 12 weeks (3) Stretching 2 times a week (120 min per week) for 12 weeks |
| Outcomes | Primary Outcome Measures: Change in Walking Velocity from Baseline to 3 Months Secondary Outcome Measures: Change in Blood oxygen level dependent signal from baseline to 3 months, Change in Mini Balance Evaluation Systems Test (Mini‐BESTest) from baseline to 3 months, Change in PDQ‐39 from baseline to 3 months, Change in Movement Disorder Society Unified Parkinson Disease Rating Scale (UPDRS) Subscale III from baseline to 3 months, Change in Mini Balance Evaluation Systems Test (Mini‐BESTest) from 3 to 6 months, Change in UPDRS Subscale III from 3 months to 6 months, Change in PDQ‐39 from 3 months to 6 months, Change in walking velocity from 3 months to 6 months |
| Starting date | February 2013 |
| Contact information | Washington University School of Medicine St. Louis, Missouri, United States, 63108 Martha Hessler: hesslerm@wusm.wustl.edu Gammon M Earhart, PhD, PT: earhartg@wusm.wustl.edu |
| Notes |
Differences between protocol and review
There are some differences between the protocol and the review briefly described below. We planned to do a subgroup analysis comparing subgroups of similar interventions in terms of duration and frequency.
After introducing a sensitivity analysis with incorporating four subgroups, we decided to do not any further subgroup analysis due to the small number of studies and to avoid multiplicity.
However, since such a subgroup analyses by duration and frequency of intervention, and additionally by time of outcome assessment might be clinically relevant, these will be conducted when and if there is data available in future updates.
For primary and secondary outcomes, we did not separate analyses for data immediately after the end of the study and at follow up after the study end to look for any sustained effects. This was due to the small number of studies.
Contributions of authors
Jan Mehrholz (JM) contributed to the conception and design of the protocol and approved the final manuscript. He searched electronic databases and conference proceedings, screened titles and abstracts of references identified by the search, selected and assessed trials, extracted trial and outcome data, guided the analysis and the interpretation of the data, and contributed to and approved the final manuscript of the review.
Joachim Kugler (JK) assessed the methodological quality of selected trials, and contributed to and approved the final manuscript of the review.
Bernhard Elsner (BE) searched electronic databases and conference proceedings, screened titles and abstracts of references identified by the search, located, selected and assessed trials, extracted trial and outcome data, assessed the methodological quality of selected trials, and contributed to and approved the final manuscript of the review.
Kathleen Hirsch (KH) and Alexancer Storch (AS) contributed to and approved the final manuscript of the review.
Marcus Pohl (MP) contributed to the conception and design of the review, drafted the protocol, and assessed the methodological quality of selected trials. Together with JM, he contacted trialists about unpublished data and also entered the data, carried out statistical analysis, helped with the interpretation of the data, drafted the review and approved the final manuscript of the review.
Sources of support
Internal sources
Wissenschaftliches Institut, Klinik Bavaria Kreischa, Germany.
Department of Public Health, Medizinische Fakultät Carl Gustav Carus, Technische Universität Dresden, Germany.
External sources
No sources of support supplied
Declarations of interest
MP and JM were co‐authors of one included trial (Pohl 2003). They did not participate in the quality assessment and data extraction of this study.
Edited (conclusions changed)
References
References to studies included in this review
Bello 2013 {published data only}
Cakit 2007 {published data only}
Canning 2012 {published data only}
- Canning C, Allen N, Fung V, Morris J, Dean C. Home‐based treadmill walking for individuals with Parkinson’s disease: A pilot randomized controlled trial. Movement Disorders 2008; Vol. 23, issue Suppl. 1:S1‐6.
- Canning CG, Allen NE, Dean CM, Goh L, Fung VSC. Minimally‐supervised treadmill training for individuals with Parkinson's disease: A randomized controlled trial. Neurorehabilitation and Neural Repair 2012; Vol. 26 (6):703‐4. [1545‐9683]
Carda 2012 {published data only}
- Carda S, Invernizzi M, Baricich A, Comi C, Croquelois A, Cisari C. Robotic gait training is not superior to conventional treadmill training in parkinson disease: a single‐blind randomized controlled trial. Neurorehabilitation & Neural Repair 2012; Vol. 26, issue 9:1027‐34. [1552‐6844] [DOI] [PubMed]
Chaiwanichsiri 2011 {published data only}
- Chaiwanichsiri D, Kitisomprayoonkul W. Treadmill and Music Cueing for Gait Training in Mild to Moderate Parkinson's Disease. www.clinicaltrials.gov ClinicalTrials.gov Identifier: NCT00750945.
- Chaiwanichsiri D, Wangno W, Kitisomprayoonkul W, Bhidayasiri R. Treadmill training with music cueing: A new approach for Parkinson's gait facilitation. Asian Biomedicine 2011; Vol. 5, issue 5:649‐54. [1905‐7415]
Fisher 2008 {published data only}
- Fisher BE, Wu AD, Salem GJ, Song J, Lin CH, Yip J, et al. The effect of exercise training in improving motor performance and corticomotor excitability in people with early Parkinson's disease. Archives of Physical Medicine and Rehabilitation 2008; Vol. 89, issue 7:1221‐9. [DOI] [PMC free article] [PubMed]
Frazzitta 2009 {published data only}
- Frazzitta G, Maestri R, Uccellini D, Bertotti G, Abelli P. Rehabilitation treatment of gait in patients with Parkinson's disease with freezing: a comparison between two physical therapy protocols using visual and auditory cues with or without treadmill training. Movement disorders : official journal of the Movement Disorder Society 2009, issue 8:1139‐43. [DOI] [PubMed]
Harro 2014 {published data only}
- Harro CC, Shoemaker MJ, Frey OJ, Gamble AC, Harring KB, Karl KL, et al. The effects of speed‐dependent treadmill training and rhythmic auditory‐cued overground walking on gait function and fall risk in individuals with idiopathic Parkinson's disease: a randomized controlled trial. NeuroRehabilitation 2014;34(3):557‐72. [DOI] [PubMed] [Google Scholar]
Kurtais 2008 {published data only}
Miyai 2000 {published data only}
Miyai 2002 {published data only}
Nadeau 2013 {published data only}
- Nadeau A, Pourcher E, Corbeil P. Effects of 24 Weeks of Treadmill Training on Gait Performance in Parkinson Disease. Medicine & Science in Sports & Exercise. 2013/09/05 2013. [1530‐0315: (Electronic)] [DOI] [PubMed]
- Nadeau A, Pourcher E, Corbeil P. Effects of 24 wk of treadmill training on gait performance in Parkinson's disease. Medicine & Science in Sports & Exercise. 2013/09/05 2014; Vol. 46, issue 4:645‐55. [1530‐0315: (Electronic)] [DOI] [PubMed]
- Nadeau A, Pourcher E, Corbeil P. The effect of treadmill training on gait and quality of life in patients with early Parkinson's disease. Movement Disorders 2012; Vol. 27:S309. [0885‐3185]
Picelli 2013 {published data only}
- Picelli A, Melotti C, Origano F, Neri R, Waldner A, Smania N. Robot‐assisted gait training versus equal intensity treadmill training in patients with mild to moderate Parkinson's disease: a randomized controlled trial. Parkinsonism & Related Disorders 2013; Vol. 19, issue 6:605‐10. [1873‐5126] [DOI] [PubMed]
Pohl 2003 {published data only}
Protas 2005 {published data only}
- Protas EJ, Mitchell K, Williams A, Qureshy H, Caroline K, Lai EC. Gait and step training to reduce falls in Parkinson's disease. NeuroRehabilitation 2005; Vol. 20, issue 3:183‐90. [PubMed]
Sale 2013 {published data only}
- Sale P, Pandis MF, Domenica LP, Sova I, Cimolin V, Ancillao A, et al. Robot‐assisted walking training for individuals with Parkinson's disease: A pilot randomized controlled trial. BMC Neurology 2013; Vol. 13, issue 50. [DOI] [PMC free article] [PubMed]
Shulman 2013 {published data only}
- Shulman LM, Katzel LI, Ivey FM, Sorkin JD, Favors K, Anderson KE, et al. Randomized clinical trial of 3 types of physical exercise for patients with Parkinson disease. JAMA Neurology 2013; Vol. 70, issue 2:183‐90. [2168‐6157] [DOI] [PMC free article] [PubMed]
Yang 2010 {published data only}
References to studies excluded from this review
Bello 2008 {published data only}
Diaz de la Fe 2008 {published data only}
- Diaz De La Fe A, Alvarez Gonzalez L, Rodriguez Martinez A, Sentmanat Belison A, Martinez Hidalgo C, Alvarez M, et al. Use of the mat of march (Treadmill) and the partial support of weight of the body (BPWS) to treat the disorders of the march in Parkinson's disease. [Spanish]. Revista Mexicana de Neurociencia 2008; Vol. 9, issue 6:466‐70. [1665‐5044]
Fisher 2013 {published data only}
Ganesan 2010 {published data only}
- Ganesan M, Pal PK, Gupta A, Sathyaprabha TN. Treadmill gait training improves baroreflex sensitivity in Parkinson's disease. Clinical Autonomic Research 2014, issue 3:111‐8. [DOI] [PubMed]
- Ganesan M, Pal PK, Gupta A, Talakad S. Effect of partial weight supported treadmill gait training on balance in patients of Parkinson's disease. Parkinsonism and Related Disorders 2010; Vol. 16:S66. [1353‐8020]
- Ganesan M, Sathyaprabha TN, Gupta A, Pal PK. Effect of Partial Weight‐Supported Treadmill Gait Training on Balance in Patients With Parkinson Disease. PM and R 2014, issue 1:22‐33. [DOI] [PubMed]
- Gupta A, Ganesan M, Pal P, Talakkad S. Effect of partial weight‐supported treadmill gait training on balance in patients with parkinson disease. PM and R 2011; Vol. 1):S163‐4. [1934‐1482] [DOI] [PubMed]
Gianfrancesco 2009 {published data only}
- Gianfrancesco M, Benedicto D. Effect of Robot‐Assisted Gait Training on Freezing of Gait in Parkinson's Disease. www.clinicaltrials.gov January 7, 2009 :ClinicalTrials.gov Identifier: NCT00819949.
Schenkman 2012 {published data only}
- Schenkman M, Hall DA, Barón AE, Schwartz RS, Mettler P, Kohrt WM. Exercise for people in early‐ or mid‐stage Parkinson disease: a 16‐month randomized controlled trial. Physical Therapy 2012, issue 11:1395‐410. [DOI] [PMC free article] [PubMed]
References to studies awaiting assessment
Horak 2011 {published data only}
- Horak F. The Effects of Treadmill Versus Agility Training in Parkinson's Disease. http://ClinicalTrials.gov/show/NCT00982709 (accessed 18 February 2013).
Mezzarobba 2013 {published data only}
- Mezzarobba S, Pellegrini L, Giulia S, Gorian A, Banica M, Busan P, et al. Neurocognitive rehabilitation with motor imagery vs treadmill training for freezing of gait in Parkinson's disease: A randomized controlled study. Journal of Parkinson's Disease 2013; Vol. 3:152. [1877‐7171]
References to ongoing studies
NCT01768832 {published data only}
- NCT01768832. Exercise and Parkinson's: Comparing Interventions and Exploring Neural Mechanisms. http://ClinicalTrials.gov/show/NCT01768832 (accessed 18 February 2014).
Additional references
Ashburn 2004
- Ashburn A, Jones D, Plant R, Lovegreen B, Kinnear E, Handford F, et al. Physiotherapy for people with Parkinson’s disease in the UK: an exploration of practice. International Journal of Therapeutics and Rehabilitation 2004; Vol. 11, issue 4:167.
Comella 1994
de Goede 2001
Deane 2001
- Deane KH, Jones D, Playford ED, Ben‐Shlomo Y, Clarke CE. Physiotherapy for patients with Parkinson's Disease: a comparison of techniques. Cochrane Database of Systematic Reviews 2001, Issue 3. [DOI: 10.1002/14651858.CD002815] [DOI] [PubMed] [Google Scholar]
Goodwin 2008
Hass 2014
Herman 2008
Higgins 2011
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Jankovic 2000
- Jankovic J. Complications and limitations of drug therapy for Parkinson's disease. Neurology 2000; Vol. 55, issue Suppl 6 (12):S2‐6. [PubMed]
Johnell 1992
Limousin 1998
Mehrholz 2014
- Mehrholz J, Pohl M, Elsner B. Treadmill training and body weight support for walking after stroke. Cochrane Database of Systematic Reviews 2014, Issue 1. [DOI: 10.1002/14651858.CD002840.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Morris 2000
- Morris M. Movement disorders in people with Parkinson disease: a model for physical therapy. Physical Therapy 2000;80:578–97. [PubMed] [Google Scholar]
Motto 2003
- Motto C, Tamma F, Candelise L. Deep brain stimulation of subthalamic nucleus for Parkinson's disease. Cochrane Database of Systematic Reviews 2003, Issue 2. [DOI: 10.1002/14651858.CD004491] [DOI] [Google Scholar]
Muslimovic 2008
Pohl 2002
- Pohl M, Mehrholz J, Ritschel C, Rückriem S. Speed‐dependent treadmill training in ambulatory hemiparetic stroke patients: a randomized controlled trial. Stroke 2002;33(2):553‐8. [DOI] [PubMed] [Google Scholar]
Tomlinson 2012
- Tomlinson CL, Patel S, Meek C, Herd CP, Clarke CE, Stowe R, et al. Physiotherapy intervention in Parkinson's disease: systematic review and meta‐analysis. British medical journal 2012;345:e5004. [DOI] [PMC free article] [PubMed] [Google Scholar]
Tomlinson 2013
- Tomlinson CL, Patel S, Meek C, Herd CP, Clarke CE, Stowe R, et al. Physiotherapy versus placebo or no intervention in Parkinson's disease. Cochrane Database of Systematic Reviews 2013, Issue 9. [DOI: 10.1002/14651858.CD002817.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
Tomlinson 2014
- Tomlinson CL, Herd CP, Clarke CE, Meek C, Patel S, Stowe R, Deane KHO, Shah L, Sackley CM, Wheatley K, Ives N. Physiotherapy for Parkinson's disease: a comparison of techniques. Cochrane Database of Systematic Reviews 2014, Issue 6. [DOI: 10.1002/14651858.CD002815.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to other published versions of this review
Mehrholz 2009
- Mehrholz J, Friis R, Kugler J, Twork S, Storch A, Pohl M. Treadmill training for patients with Parkinson's disease. Cochrane Database of Systematic Reviews 2009, Issue 2. [DOI: 10.1002/14651858.CD007830] [DOI] [PubMed] [Google Scholar]
Mehrholz 2010
- Mehrholz J, Friis R, Kugler J, Twork S, Storch A, Pohl M. Treadmill training for patients with Parkinson's disease. Cochrane Database of Systematic Reviews 2010, Issue 1. [DOI: 10.1002/14651858.CD007830.pub2] [DOI] [PubMed] [Google Scholar]


