Abstract
We continue to increase our cognizance and recognition of the importance of healthy living (HL) behaviors and HL medicine (HLM) to prevent and treat chronic disease. The continually unfolding events precipitated by the coronavirus disease 2019 (COVID-19) pandemic have further highlighted the importance of HL behaviors, as indicated by the characteristics of those who have been hospitalized and died from this viral infection. There has already been recognition that leading a healthy lifestyle, prior to the COVID-19 pandemic, may have a substantial protective effect in those who become infected with the virus. Now more than ever, HL behaviors and HLM are essential and must be promoted with a renewed vigor across the globe. In response to the rapidly evolving world since the beginning of the COVID-19 pandemic, and the clear need to change lifestyle behaviors to promote human resilience and quality of life, the HL for Pandemic Event Protection (HL-PIVOT) network was established. The 4 major areas of focus for the network are: (1) knowledge discovery and dissemination; (2) education; (3) policy; (4) implementation. This HL-PIVOT network position statement provides a current synopsis of the major focus areas of the network, including leading research in the field of HL behaviors and HLM, examples of best practices in education, policy, and implementation, and recommendations for the future.
Abbreviations: ACA, Affordable Care Act; BMI, body mass index; COPD, chronic obstructive pulmonary disease; COVID-19, coronavirus disease 2019; CRF, cardiorespiratory fitness; HCPs, healthcare professionals; HL, healthy living; HLM, healthy living medicine; HL-PIVOT, healthy living for pandemic event protection; METs, metabolic equivalents; PA, physical activity; PAFiT, Physical Activity and FiTness; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; US, United States; VO2, oxygen consumption; WHO, World Health Organization
Introduction
We continue to increase our cognizance and recognition of the importance of healthy living (HL) behaviors and HL medicine (HLM) to prevent and treat chronic disease.1 The continually unfolding events precipitated by the coronavirus disease 2019 (COVID-19) pandemic have further highlighted the importance of adhering to HL behaviors, as indicated by the unhealthy lifestyle characteristics of those who have been hospitalized and died from this viral infection.2, 3, 4, 5 Specifically, those with unhealthy lifestyle characteristics (ie, increased risk for chronic disease) or diagnosed with 1 or more chronic diseases are at a significantly higher risk for poor health outcomes if infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).4 , 6, 7, 8 Oftentimes, unhealthy lifestyle characteristics and 1 or more chronic disease diagnoses coexist in those with the poorest outcomes associated with COVID-19 infection. There has already been recognition that adhering to HL behaviors, prior to the COVID-19 pandemic, may have a substantial protective effect in those who become infected with the virus.9 Moreover, necessary changes to daily life across the globe (eg, social distancing, stay at home orders, etc) have increased the prevalence of unhealthy lifestyle behaviors, such as an increase in sedentary behavior and poor dietary habits.10, 11, 12, 13, 14 The unhealthy lifestyle phenotype, was a global health crisis prior to COVID-19. This phenotype, along with the effects of a further deterioration in lifestyle choices precipitated by the COVID-19 pandemic, if not reversed, will further compound the already disastrous effects of unhealthy lifestyle characteristics on health outcomes.15 Now more than ever, HL behaviors and HLM are essential and must be studied, promoted and implemented with a renewed vigor across the globe. In response to the rapidly evolving world since the beginning of the COVID-19 pandemic, and the clear need to change lifestyle behaviors to promote human resilience and quality of life, the HL for Pandemic Event Protection (HL-PIVOT) network was established,16 , 17 now with more than 120 members with expertise in HL and HLM from around the world (See Appendix 1 for list of current network members). The 4 major areas of focus for the network are: (1) knowledge discovery and dissemination; (2) education; (3) policy; and (4) implementation. The term “pandemic event protection” was chosen as part of the network title given obesity,18 physical inactivity,19 type-2 diabetes,20 and other chronic diseases,21 in addition to COVID-19, have all been characterized as pandemics. Moreover, as we will demonstrate, increasing HL behaviors and HLM are essential for the prevention and treatment of all of the aforementioned pandemics. This HL-PIVOT network position statement provides a current synopsis of the major focus areas of the network, including leading research in the field of HL behaviors and HLM, examples of best practices in education, policy, and implementation, and recommendations for the future.
Knowledge Discovery and Knowledge Discovery and Dissemination
Knowledge discovery and dissemination of findings are the cornerstone of determining effective interventions and programs in all settings (ie, healthcare, school systems, workplace, etc). There is a wealth of knowledge on the detrimental impact of unhealthy lifestyle characteristics as well as the favorable impact of the adoption of and adherence to HL behaviors and practice of HLM. The following passages provide key scientific highlights related to these areas.
Physical Activity and Cardiorespiratory Fitness
Increased physical activity (PA) levels have been widely documented to provide multiple physiological adaptations leading to significant health benefits including reductions in morbidity and mortality.22 Specifically, higher levels of PA equate to a lower risk of all-cause mortality,23 , 24 cardiovascular mortality,25 cancer mortality,26 chronic obstructive pulmonary disease severity,27 mobility disability in the elderly,28 as well as multiple other benefits.29 Furthermore, PA levels have been shown to be closely associated with quality-of-life measures and improvements in psychologic affect.30 , 31
The effects of PA on both the lifespan (ie, from childhood to advanced age) and, more importantly, healthspan,a make habitual PA a necessary component of any effort to improve and promote HL behaviors. The importance of being physically active in the current COVID-19 pandemic environment is also gaining recognition, given the importance of PA in promoting healthy physiologic function, in particular immune function and reduced systemic inflammation, thereby likely playing a protective role with viral infection.8 , 9 , 32, 33, 34 Along these lines, Tavakol et al35 recently found higher levels of habitual PA were significantly associated with lower symptom severity in 206 patients infected with COVID-19. There is also previous evidence to indicate aerobic exercise training improves the influenza vaccine response. In 140 sedentary elderly adults randomized to either aerobic exercise training or balance/flexibility exercises, commenced 4 months prior to influenza vaccination, seroprotection 24 weeks after vaccination, peak oxygen consumption (VO2) and sleep quality were all significantly greater in the aerobic exercise training group compared to the balance and/or flexibility exercise group. The self-reported severity of respiratory track illnesses was also significantly lower in the aerobic exercise training group compared to the balance and/or flexibility exercise group.36 Similar studies are needed in individuals receiving the COVID-19 vaccine. From a broader population perspective, Lu et al37 reported that moving more and sitting less was associated with better sleep quality and mental health in Chinese adolescents during the COVID-19 pandemic, further demonstrating the numerous benefits of PA in the current environment.
The world was in the midst of a physical inactivity crisis prior to the COVID-19 pandemic,38 a crisis that now appears to be worsening due to the pandemic.10 , 39, 40, 41 If the new, lower, physical inactivity baseline is not reversed as the world recovers from the COVID-19 pandemic, the global health consequences are likely to be devastating.15 As such, urging/persuading individuals to move more and sit less on a daily basis is a global public health imperative moving forward.42 , 43 Members of the HL-PIVOT network have recently launched a global PA survey - PA and FiTness behaviors among adults during the COVID19 pandemic (PAFiT) (https://redcap.sctimst.ac.in/surveys/?s=J9PLJMNJTL), which will help to assess current PA trends and guide the development of interventions and programing moving forward.
A significant degree of the gained benefit from PA is linked to improvements in cardiorespiratory fitness (CRF). An individual's CRF, often expressed as peak metabolic equivalents or oxygen consumption (VO2), portends valuable information related to cross-sectional health status and longitudinal health trajectory.44 Simply stated, individuals who present with a higher CRF, irrespective of other negative health factors (eg, excess body mass, diabetes mellitus, diagnosis of 1 or more chronic diseases) have a better in health status45 and prognosis.44 There is a sound theoretical rational to hypothesize that a higher CRF, brought about by exercise training and PA, would have a protective effect in those who are infected with COVID-19 given the association with a higher CRF and both reduced systemic inflammation and enhanced immune function.46 In fact, recent evidence supports this hypothesis by demonstrating a higher CRF is associated with reduced risk of hospitalization in individuals infected with COVID-19.7 Longitudinal improvements in CRF have also been shown to cause significant reductions in cardiovascular risk markers such as hypertension, obesity, and diabetes47 as well as improve prognosis and mortality.48 The significant role of CRF on overall health can be highlighted by appreciating that oxygen transport is dependent upon multiple system collaboration to ensure efficient oxygen transfer from the atmosphere to the mitochondria of working muscle.22 This collaboration demonstrates whole-body physiology and can explain the powerful prognostic utility of CRF, which is now considered a vital sign.22 , 44 Unfortunately, there are already indications that the COVID-19 pandemic is having a detrimental impact on CRF. In a cohort of healthy children undergoing repeat cardiopulmonary exercise testing, Dayton et al49 demonstrated a significant reduction in peak VO2 during the COVID-19 pandemic compared to pre-pandemic levels. Moreover, Raman et al50 demonstrated a significantly lower peak VO2 following hospital discharge in patients infected with COVID-19 compared to healthy controls. Given the importance of CRF to health status and prognosis, assessment and, when indicated, improvement in CRF is a health imperative in all individuals.
Nutrition
Nutritional patterns have been linked to either an increased or decreased risk of chronic disease and poor health outcomes, depending on key characteristics of the diet consumed. Independent of a specific type of diet, consumption of fruits and vegetables are shown to decrease the risk of chronic disease.51 , 52 The Mediterranean diet has garnered significant attention as an approach that is healthy and reduces the risk of adverse health events.53, 54, 55 The Mediterranean diet is broadly characterized by a higher “intake of fruits, nuts and seeds, vegetables, fish, legumes, and cereals and limiting the intake of meat and dairy products.”55 Conversely, a high consumption of fried food, saturated fat, processed meats and sugar-sweetened beverages has been linked to an increased risk of chronic disease.56 There is no 1-size-fits-all approach to a healthy diet. Rather, certain types of food are preferable over others with respect to reducing the risk of chronic disease. The American Heart Association/American College of Cardiology recommends dietary patterns emphasize high intake of “vegetables, fruits, and whole grains; include low-fat or fat-free dairy products, poultry, fish, legumes, non-tropical (not coconut or palm kernel oil) vegetable oils, and nuts; and limit intake of sweets, sugar-sweetened beverages (SSBs), salty or highly processed foods, and fatty or processed meats (choose lean or extra-lean meats instead).”57 Because of the clear importance of dietary patterns to gauging risk for chronic disease, screening of dietary intake in the clinical setting is recommended by the American Heart Association.58
Nutritional patterns have received attention during the COVID-19 pandemic. Poor dietary patterns are clearly linked to a higher prevalence of chronic disease and obesity, which both portend poorer outcomes with COVID-19 infection.59 , 60 The inability to access healthy nutritional choices, leading to increased chronic disease and obesity prevalence, is particularly problematic in underserved communities, which is likely contributing to the disproportional negative impact of the COVID-19 pandemic on these communities.61 Moreover, healthy nutritional patterns have been linked to improved immune function which is critical to an individual's ability to successfully overcome a viral infection.62 Bousquet et al63 recently hypothesized that differences in dietary patterns across countries influence the observed differences in COVID-19 mortality rates. Specifically, dietary patterns in some European countries that have higher antioxidant qualities or promote reduced angiotensin-converting enzyme activity may contribute to the lower risk of death due to COVID-19 infection. More troubling is recent data indicating dietary patterns during the COVID-19 pandemic are worsening,11 , 12 , 40 a trend that, if not reversed, will further erode population health (ie, increased risk for chronic disease and susceptibility to severe viral infections) on a global scale.
Body Habitus
The prevalence of overweight and obesity are very significant in many countries around the world. More than 10% of the population at the global level and more than one third of the United States (US) population have body mass index (BMI) levels in the obesity range (ie, ≥30 kg/m2), with average adult BMIs constantly rising over time.64 Overweight and obesity are leading to an increased risk for developing chronic diseases such as cancer, diabetes, and cardiovascular diseases.64 Previous studies report that an increase of 2% in the average BMI of a society reduces average life expectancy by 1 year.65 Due to the enormous medical and economic impact, the prevention and treatment of overweight and obesity are primary public health goals globally.64 , 66 Obesity has rapidly become recognized as a primary contributor to adverse outcomes in patients infected with COVID-19.67 , 68 Obesity also significantly increases the cost of care in patients infected with COVID-19.69
A mild reduction in weight loss, such as 3%-5%, significantly reduces the risk for other chronic diseases.64 , 66 , 70 , 71 To achieve this goal, it is important to implement multidisciplinary, multilevel interventions, such as a healthy diet, PA, pharmacotherapy, surgery, and behavioral counseling. For optimal success, interventions need to be supported at the personal, community, organization, and government levels.64 , 66 , 70, 71, 72, 73 The primary goal is a weight reduction of 3%-10% over a 6-12-month timeframe, depending on the presence of comorbidities. However, if a 2.5% weight loss is not achieved during the first month of therapy, interventions should be intensified as early weight loss strongly indicates long‐term success.64 , 66 After target weight reduction is achieved, long‐term measures for weight maintenance are warranted.74 Multidisciplinary teams should include physicians, nurses, clinical exercise physiologists, dieticians, physical therapists, pharmacists, psychologists, social workers, administrators and decision-makers, in order to treat obesity as a chronic disease.66 , 70, 71, 72, 73, 74
HL behaviors are needed to create an environment that supports healthy energy balance endeavors and include personal efforts and community-based activities supported by government and public policies.66 , 72 , 73 There are many guidelines for lifestyle modifications recommending PA, diet, and behavioral interventions for weight‐reduction when BMI is ≥30 kg/m2, or ≥25 kg/m2 accompanied by weight‐related complications (eg, cardiovascular disease, hypertension, diabetes).64 , 66 , 70 , 71 A low‐calorie balanced diet, increased PA, and behavioral support are suggested for reduction and maintenance of weight.64 , 66 , 70 , 71 , 74 A daily energy deficit should be around 500-750 kcal, without favoring any particular composition or form of nutritional therapy, emphasizing that what matters most is a healthy and balanced diet that is personalized to the individual to achieve optimal adherence.64 , 66 , 70 , 71 Structured meal plans, portion control, and meal replacements are also recommended.64 , 66 , 70 , 71 In the case of need for faster weight loss, a very low-calorie diet (≤800 kcal/day) with medical supervision should be considered.64 Behavioral interventions are important additional tools for reaching weight loss goals and consist of various strategies such as stimulus control, motivational interviewing, and cognitive restructuring.64 , 66 , 70 , 71 Only well-organized implementation of targeted multidisciplinary and multilevel interventions may address excess body weight, in terms of maintenance of behaviors that are necessary for weight reduction and weight-loss maintenance.
Smoking Cessation
Although the global trend for smoking is declining, the current prevalence is estimated to be 19.9% of the world's adult population.75 Smoking-related illness remains a pertinent risk factor in the development of chronic diseases, including certain cancers, chronic obstructive pulmonary disease and cardiovascular disease.76 The economic burden of smoking is considerable; in 2012 smoking accounted for 5.7% of global health expenditures and ∼12% (4.9 million) of all adult deaths worldwide.77 Moreover, a history of smoking, both current and former, has been shown to be associated with increased COVID-19 severity as well as an increased risk of death.78 As such, smoking cessation remains an important recommendation for clinical practice guidelines worldwide.
Smoking cessation is directly associated with substantial reductions in health risks and mortality77 as well as significant cost-benefit to healthcare systems.79 While services for smoking cessation are based on psychological and pharmacological approaches that use face-to-face or online methods, there is little consistency and widespread variation and success in helping smokers to quit.80 The need to develop successful and scalable interventions that have increased efficacy will have global effects, especially in countries where the smoking epidemic is most rampant. At an individual level, improved patient outcomes and reduced mortality are important, but when considered alongside population-level change, the potential impact is magnified and considers broad societal and economic benefits for healthcare providers and policymakers globally.
Crucially, access to smoking cessation support and the required resources must be universal. Support systems are currently directed by local health authorities with evidence demonstrating that particular groups in society are less likely to be offered support, despite a higher incidence of smoking.81 In addition to increasing access to support services, future research must: (1) Utilize cross-disciplinary approaches that consider intrapersonal, societal, behavioral, and attitudinal approaches to increase effectiveness; (2) Adopt approaches that are informed by a strong theoretical underpinning that considers behavioral insights, addiction, readiness to change and possible ambivalence; (3) Improve understanding of the optimal method of cessation activities such as counselling, alongside behavioral insights to ensure optimal timing and frequency of support; and (4) Increase understanding of the role and importance of technology in the delivery and support of smoking cessation initiatives. The impact of global smoking cessation is of paramount importance, with widespread impacts on the international health and wellbeing agenda. Current localized approaches are not sufficient to promote lasting cessation. Unified approaches that are underpinned by a strong theoretical basis that is widely accessible is likely to result in sustainable and scalable changes in population-level health, alleviating pressure on healthcare systems worldwide.
Sleep and Health
A panel of 15 experts in sleep medicine developed a recommendation for sleep duration, or sleep duration range, that promotes optimal health in adults aged 18-60 years, suggesting that adults regularly sleep ≥7 hours per night for optimal health.82 Grandner's sleep review in 2012 indicated that insufficient sleep is common at the population level.83 This sleep insufficiency has been associated with many adverse health outcomes across the lifespan including obesity, cardiovascular disease (eg, hypertension, heart disease, stroke), metabolic disorders (eg, diabetes), and mortality.83 An extensive review by Matricciani et al83 suggests that children may sleep less today than previous generations. Asarnow et al84 demonstrated that earlier adolescent bedtime was associated with healthier food choices (eg, low glycemic index foods, fruit, and dairy) which over time may be protective of obesity. Gildner et al85 examined the effects of sleep duration and quality on cognition in older adults from 6 middle income countries across the globe and found short (<6 h/night) and long (>9 h/night) sleep duration was associated with reduced cognitive test performance. The importance of healthy sleep patterns in the COVID-19 pandemic era is gaining recognition, particularly given its link to healthy immune function.86 This is coupled with the troubling findings in a systematic review and meta-analysis by Jahrami et al87 indicating the prevalence of poor sleep is high in the general population, health care workers and patients infected with COVID-19. Recently, Zhang et al88 found poor sleep quality was associated with an increased need for intensive care unit admission in patients hospitalized with COVID-19. The potential negative impacts of poor sleep quality in the context of an increased risk for a more severe clinical course and complicated recovery in patients infected with COVID-19 have also been raised by other groups.89 , 90
The Future of Knowledge Discovery in Healthy Living Behaviors and Healthy Living Medicine
The significant body of evidence regarding the multiple health benefits of HL behaviors and HLM are beyond dispute. Increasing PA and CRF, consuming a healthy diet, maintaining a healthy body weight, not smoking, and good sleep quality, significantly reduces the risk of poor health outcomes associated with chronic diseases and viral infections, as well as increases the ability to perform activities of daily living and quality of life. While mechanistic studies related to HL practices are ongoing (ie, determining the physiologic effects of HL behaviors), there is a more urgent need to investigate and solidify approaches to increase the adoption of HL behaviors and the practice of HLM.91 In short, we know both HL behaviors and HLM work and a significantly greater percentage of the global population needs to adopt a healthier lifestyle.
The adoption of HL behaviors and delivery of HLM are not one-size-fits-all approaches and as such, it is critical that we gain a better understanding, through future knowledge discovery, of how to help individuals, communities, and populations, from diverse backgrounds, increase the adoption of HL behaviors. Only through this knowledge discovery, which will drive best practices, will we be able to reduce the devastating effects of chronic diseases and viral pandemics.
Education – The Passport to Healthy Living
Lifelong Education
Education is acknowledged as a fundamental cornerstone of adherence to HL behaviors, wellbeing and a key indicator of development.92 A life-course perspective for HL describes a constant companion for individuals, rather than focus on a particular life stage for a single intervention. Although an early exposure to HL behaviors followed by progressive development across subsequent life stages has proven to increase the likelihood of adoption of such behaviors93, 94, 95; to date, the adult years have received greatest attention regarding public policies and community-based interventions.96 However, at this stage of life, unhealthy lifestyle behaviors are more likely to be entrenched and are more difficult to reverse. The COVID-19 pandemic has brought refocused attention on unhealthy lifestyle behaviors and the detrimental consequences of such behaviors.3 , 8 , 67 , 68
The foundations of a healthy, productive and sustainable society are built on the healthy growth and development of children and youth.97 Unhealthy lifestyles result in developmental vulnerabilities and perpetuate cycles of disadvantage and health disparity for generations.98, 99, 100 In contrast, HL behaviors builds human capital.101 The Human Capital Model considers the relationships between PA and various aspects of human development and proposes that the outcomes of PA can be framed as differential “capitals.”102 A poor investment in these capitals increases the likelihood of suboptimal opportunities for many young people with potential long-term consequences for adulthood, and more importantly, the health and wellbeing of the next generation.98 , 101
A life-course perspective for HL behaviors describe an approach where targeted activities and initiatives are tailored, taking account of the key differences and unique developmental needs of individuals in each age group. The World Health Organization (WHO) Ending Obesity report103 provides a very good example of a framework for an international focus on HL behaviors at multiple life stages for chronic disease prevention. Strategies and models in HL behaviors could be stratified in early childhood, adolescent, adult, and HL ecosystems that focus on longitudinal efforts to support individuals (Fig 1 ). When individuals start participating in this HL model, the healthy lifestyles are caregiver-directed. As individuals mature in age and experience in HL behaviors, they become self-directed and the new habits and lifestyles are self-care motivated.
FIG 1.
Healthy living accompaniment through life stages.
An early childhood and primary school focus on HL behaviors is essential for a child to reach their full potential104 with key enablers being health-promoting environments and strong relationships early in life.105 Increasing evidence highlights the importance of the first 1000-2000 days of life for later health and wellbeing106; it has been described as a ‘blueprint for lifespan trajectories’ for later health and wellbeing.104 Exposure factors in early life include: (1) parental, for example, obesity, gestational diabetes mellitus, or smoking; (2) dietary, including breastfeeding and eating behaviors; (3) PA or sedentary behavior; and (4) sleep quality.107 Interestingly, childhood obesity and poor academic performance tend to cluster in school settings with a high percentage of children and young people from low socioeconomic backgrounds.108 For example, integrating strategies for higher levels of PA as part of the academic curriculum allow educational attainment and improved body composition.109
The adolescent period is critically important given the typical changes in body composition, dietary and PA behaviors, and often, increased social responsibilities during these years.100 Current research estimates that 35.8% female and 34.1% male adolescents are overweight or obese.110 A sub-optimal environment, commonly including limited opportunities for PA and limited access to healthy nutrition, increases the risk for chronic diseases including cardiovascular disease, type 2 diabetes, depression and overweight and obesity.98 , 111 Adolescence is a highly teachable period and an acknowledged gateway to future generations.100 , 108 , 112 , 113 Targeted approaches to promoting HL behaviors in adolescent girls have particular merit with a focus on minimizing the risk of entering the childbearing years in sub-optimal health.114 , 115
The adult focus is crucial for the current global obesogenic environment, with a significant and increasing proportion of individuals with overweight or obesity and associated co-morbidities. To maximize the chance of success, approaches require an extensive collaboration, a whole-of-government approach, and co-design of HL educational opportunities with educators, teachers, health care professionals (HCPs), families, and children in early education and school settings.106 , 116
HL ecosystem approaches propose that the ingredients for increased adherence to HL behaviors are gathered through the collective educational experiences across the life course, commencing with positive PA and nutrition experiences which are necessary to optimize the development of a broad range of skills and healthy behaviors. Habits such as promotion of PA and healthy nutrition in communities, such as family, school settings or religious groups, integrate individuals in different life stages. Approaches like these are effective as they develop not only skills for them to achieve their goals, but to create ambassadors that can further motivate others to engage in HL behaviors. Effective education models in HL for each approach are presented in Table .93 , 117, 118, 119, 120, 121, 122
TABLE.
Healthy living accompaniment examples
| Approach | Initiative | Setting | Strategy |
|---|---|---|---|
| Early childhood | Early Child Education and Care (ECEC)93 | Rural communities in South Africa: Buschbuckridge, Mpumalanga. Preschool-aged children in three preschools and 2 primary schools. |
Observation for assessing physical using the Observational System for Recording Physical Activity in Children – Preschool Version. |
| PreSchool @Healthy Weight117 |
Dutch preschool-based: Amsterdam Nieuw-West, Netherlands. Migration background |
Teachers promote practices about nutrition, physical activity and education. | |
| Munch & Move | New South Wales (NSW), Australia. Targeting children from 3 to 5 years age attending preschools. | Teachers participate in professional development training to implement fun, play-based approach to supporting health eating and physical activity habits. Based on 6 health messages: encourage breastfeeding, choose water as a drink, healthier snacks, eat more fruits and vegetables, get active each day, and turn off the screen and get active. | |
| Adolescent | Whole School, Whole Community, Whole Child (WSCC) model118 |
Alabama Champions for Healthy, Active Schools in United States. | A champion school provides at least 60 minutes of physical activity through the academic day. Provide nutritious meals, nutritious smart snacks, and nutrition education. Involve parents, staff and community. |
| Y Kids Academy119 | Canadian teens | Program designed for development of confidence and competence for cardiovascular, muscular and flexibility activities, generation of positive attitudes and behaviors for healthy living, and the removal of barriers for young adolescent participation in exercise program. | |
| Adult | i Week – Educational Tec21 Model120 |
Undergraduate students from different majors. Mexico | Immersion challenging activities focus in the development of transversal competences: self and personal skills. Intensive week-long activities where students participate in experiences outside the university. The focus is to motivate and develop self-care. |
| Healthy living ecosystem | Play Streets temporary street closures122 | Across United States | Temporary street closures creating safe spaces for play for a few hours. Family targeted: parents, adolescents and children. Urban areas during the summer. |
| Health and Wellness Academy Concept121 |
School-based multicenter wellness program (SBWP) that focuses on nutrition and physical activity. Illinois, United States. |
Health and wellness ambassadors and community connection. Integration of youth program in the community, and a college course where students participate in a 16-week course. |
Individuals inherit socioeconomic status, cultural norms, and associated family dietary patterns and PA behaviors, healthy and unhealthy alike. Every effort is required to maximize access to HL opportunities across individuals and populations. Education has proven to be a key social determinant of health, which results in significant economic benefits for a wider population. Too many homes are exposed to a sedentary, low energy expenditure environment, devoid of PA where other activities (eg, occupational, academic, social media) are prioritized over health and wellbeing.112 As inequalities in education and health cluster with a social gradient, increasing the number of HL educational initiatives contribute to provide equal opportunities amongst those who are most disadvantaged.123 More programs that create HL ecosystems are needed to develop individuals and communities that share HL behaviors that are self-care motivated.
Educating Health Care Professionals
Effective delivery of HLM requires HCPs who are competently trained to deliver this service effectively to the patients under their care. Unfortunately, most HCPs across the disciplines (ie, physicians, nurses, pharmacists, dentists, physical, and occupational therapists, etc) do not receive any training on how to assess HL behaviors, such as PA, CRF, nutrition, and subsequently counsel patients and facilitate referral to other professionals for more intensive interventions, as appropriate, when unhealthy lifestyle behaviors are identified. Competencies and educational models for HLM have recently been proposed.124, 125, 126 Unlike other practice areas that are uniquely the domain of a specific HCP, HLM should be practiced, to varying appropriate degrees, by all HCPs. Patients receiving a consistent message that PA, not smoking, a healthy body weight and heathy nutrition are essential during all HCP encounters will increase the likelihood of adoption and long-term adherence to HL behaviors.
Role of Policy and Legislation in Promoting Healthy Living
Policy can provide the opportunities to create environments that support HL behaviors. Public policy (ie, laws and regulation) and organizational or systems-level policies are critical contributors to health and wellbeing, influencing the physical, social, and economic environments that connect to behaviors from the population to individual level. They create the institutional and legal frameworks for needed for broader adoption of HL behaviors. Policies need to be carefully evaluated because the success of their implementation can have either positive or negative impacts. The policy development process and rigorous policy implementation and outcome evaluation must consider and account for unintended consequences with a special focus on health and equity impact.127 Organizational or systems level policy can begin to change a social norm and support broader public policy.
Four areas of particular importance wherein policy has had a direct effect on HL behaviors are tobacco use, nutrition, obesity prevention and treatment, and PA. Even though evidence has suggested benefits for various strategies, none are sustainable unless they are translated to policy and systems changes. Policy change has broad applicability, but implementation strategies vary considerably based on contextual and economic factors resulting in a pragmatic approach informed by local circumstances coupled with rigorous ongoing evaluation of policy implementation. This, however, requires a continuous system that works towards advocacy for a policy change while also supporting opportunities for progress learned through implementation and outcome evaluation to promote HL behaviors (Fig 2 ).
FIG 2.
Pillars of advocacy to achieve policy change.
This process underscores the importance of public policies as levers to foster HL behaviors. These public policies have been viewed as either regressive or an overreach of government (eg, taxes on tobacco, labeling or limitation of food constituents that may have adverse health effects) or positive and facilitating (eg, policies providing access to healthy foods or government appropriations for public parks and walkways). Continued policy implementation and outcome evaluation will determine which approaches have the greatest health and equity impact. The mixed responses to various policies are influenced by the ambivalent attitudes of people towards them. To better elucidate the policies around HL behaviors, this section will review existing policy achievements, issues related to vigilance for direct and indirect effects of policies, and opportunities for future policy goals and actions that will favor HL-PIVOT.
Tobacco Cessation
Among the various unhealthy behaviors, tobacco control and prevention efforts have probably gained most traction around the world and are now evolving toward a tobacco endgame.128 These have been the result of public policy and organizational and systems-wide policy. In a public health context, these policy changes at all levels are often interrelated and synergistic.
Tackling the burden of tobacco has been a priority for various organizations and governments globally. These decades of experience have led to evidence-based strategies that provide a road map for the most impactful policy change. Key strategies that, globally, have been instrumental in tobacco control, regulation and prevention, include: (1) excise taxes; (2) comprehensive smoke-free air laws; (3) robust appropriations for tobacco control and prevention programs; (4) government regulation of tobacco products; (5) counter-marketing efforts; (6) restricting the sale of tobacco products in pharmacies and health-related institutions; (7) comprehensive coverage for tobacco cessation therapy in private and public health plans; and (8) retail licensure laws. Importantly, each of these strategies has been found to be effective in reducing tobacco consumption with robust implementation science and reaffirmed by ministries of health and the US Surgeon General. While most of the current evidenced-based policy interventions are identified as negative levers, implementation and accessibility of low-cost and free access to tobacco cessation therapies are examples of positive levers that have facilitated successful movement toward a tobacco endgame. Nonetheless, from the data, it would appear that a negative lever related to policies on increased taxation, is most effective.129
Obesity
Curbing the problem of obesity has been a major concern around the globe and requires a comprehensive set of interrelated strategies addressing both prevention and treatment.130 Policy actions that broadly support routine screening and the development and delivery of effective, empirically tested obesity prevention and management programs (eg, early care and education) should be implemented across numerous settings including, schools, health care systems and similar clinical environments, communities and workplaces. Lifestyle-related treatment of obesity and obesity-related health comorbidities through intensive behavior counseling on: (1) adoption of a healthy diet; (2) a structured exercise program; and (3) an increase in PA throughout the day to promote and sustain weight loss is regarded as the first line of therapy by leading advisory organizations and should therefore be adequately covered by health care plans and be made accessible by health care systems.131, 132, 133, 134
Stigma, which is pervasive and negatively affects persons with obesity in a myriad of ways, should be systematically addressed. Stigma directly influences adoption of HL behaviors through possible delayed care and treatment and erodes prevention strategies. Policy solutions in this area should focus on separating the disease of obesity from the people who are affected. Failure to diagnose obesity as a chronic disease is a primary cause of stigma and exacerbates bias towards people with obesity when seeking medical care and advice.135 From a policy perspective, failure to adopt obesity as a non-communicable chronic disease only reinforces stigma and reduces the effectiveness of potential solutions by disconnecting them from a more global cohesive strategy. Organizational health system policy should recognize the detrimental impact of obesity stigma, bias, and discrimination on the health and well-being of patients affected by the disease and focus in on training and supportive resources that create a respectful, inclusive, and safe environment for care and prevention.136
Yet, the need for policies to adopt public health, population-based strategies to better facilitate screening, prevention, and control of obesity, is crucial. In the United States, the Affordable Care Act, has required Medicare to cover annual wellness visits and personalized prevention plan services to all beneficiaries, including screening for chronic disease risk factors and furnishing referrals to health education and prevention counseling services137 such as face-to-face intensive behavior therapy for obesity prevention and control.138 Despite policies like this in place, there remain numerous barriers to implementation of obesity programs in health care systems, presenting new and much needed opportunities to develop and/or strengthen policies facilitating the integration of various health promotion, and/or obesity-directed programs within clinical and community-based settings that reach a larger segment of the overall population, particularly those most vulnerable to obesity and are least likely to have access to adequate care
Nutrition and Access to Healthy Foods
Nutrition and access to healthy foods have a significant impact on health and well-being. The WHO developed the “Global Strategy on Diet, Physical Activity and Health” to recommend that individuals achieve a healthy weight through an appropriate energy balance, limiting intake of fats, salt, and sugar, especially refined carbohydrates, while increasing intake of fruits, vegetables, legumes, whole grains, fiber and nuts and seeds.139 To facilitate this nutritional recommendation, countries all over the world have developed various policies and strategies to promote healthy eating. Strategies to tackle healthy food and beverage consumption are categorized as mandates, restrictions, economic incentives, marketing limits, information provision, and environmental defaults.140 Similar to interventions for smoking, pricing interventions such as increased taxes on sweetened beverages141, 142, 143 (ie, sugar-sweetened beverage taxes144) and subsidies for fruit and vegetable purchases145 have shown a positive benefit on purchasing behavior and health outcomes. Additionally, restrictions on the use of trans-fatty acids in restaurants and processed foods are an important initiative that benefits cardiovascular health.146 Continued implementation evaluation will further elucidate the equity and health impact of these incentive and/or disincentive approaches.
Curbs on advertising and marketing of unhealthy foods and beverages have also been enforced in certain countries in an effort to lower the burden of poor nutrition and obesity.147 Enhancing healthy food options has been advocated across countries in different settings including schools, the workplace, and away-from home eating as well as through government nutrition assistance programs.141
Physical Activity and Sedentary Behavior
The rising burden of physical inactivity and sedentary behavior is a major concern around the world. Governments have developed various guidelines on promoting PA with the goal to reduce sedentary behavior and increase movement at the population level. These PA guidelines have made recommendations for PA across the lifespan.148 However, these guidelines do not necessarily translate into implementation. From the available evidence, we know that PA-based interventions are behavioral interventions, which introduce complexity. PA policy may also create the settings or environments where people can be physically active on a regular basis. Policy interventions focusing on PA are challenging and often require multiple stakeholders across various sectors and levels of government.149
A comprehensive analysis of policy on PA identified various sectors such as healthcare, sport, recreation and leisure, education, transport, environment, urban/rural planning and design, tourism, work and employment, public finance and research sectors influenced by PA policy.150 In addition, data-driven initiatives like Healthy People 2030 (https://health.gov/healthypeople) and the US National Physical Activity Plan (https://paamovewithus.org/national-physical-activity-plan/), are now in place to promote PA and reduce sedentary behavior. Some of the policies around PA have focused on: (1) programs for children in schools, including physical education and after-school programs151 , 152; (2) worksite-based programs153 , 154; and (3) the built environment, including active transport155 and activity-supportive environments.156, 157, 158 The WHO also released guidelines on PA and sedentary behavior, further reinforcing the importance of increasing the former and decreasing the latter.159
Despite various policies and mandates in place across the globe at different levels of government, there have not been adequate implementation studies. Incentive and disincentive approaches span all areas of HL policy and further implementation and outcome evaluation is needed to identify the most impactful approaches, especially in obesity, healthy food access and PA. In addition to assessing the best practice approaches, identifying the challenges and barriers to implementation as well as any potential unintended consequence that arises from non-targeted population-based interventions that may lead to widening of health inequities is also important.
Challenges to Successful Policy Implementation
The case for health policies toward promoting HL behaviors is well-established. Effectiveness of implementation varies by issue, geography, level of government, socioeconomic status, and race/ethnicity. Various factors that could be responsible for disparate impact should be elucidated with more implementation research. Factors that should be considered in implementation evaluation include population reach, whether there is sustainable funding, barriers, value on investment, equity impact, and feasibility.160 Furthermore, individual attitudes and behavior toward HL could also contribute since ultimately people decide to engage with policies that target the systems and environments in which they live. Psychological responses should also be considered when planning effective policy campaigns.
In summary, even though policies facilitating HL behaviors have been successful in certain areas, implementation of policies targeting unhealthy behaviors are still evolving. Continuous evaluation is necessary to ensure effectiveness of policies and evaluate the impact on pre-defined health outcomes that inform strategies to ensure equitable opportunities to adopt and adhere to HL behaviors across different populations.
Healthy Living Program Implementation
Implementation of HL programs grounded in sound research has consistently reestablished the strong correlation between adoption of and adherence to HL behaviors and reductions in mortality and morbidity as well as improvements in quality of life. However, the transfer of best practice recommendations to successful implementation has proved challenging. Of the programs which have had success implementing good scientifically sound methods, most fall into 4 distinct categories; (1) the community; (2) schools; (3) the workplace; and (4) clinics. The following sections provide implementation examples in different settings.
Community-based health programs are generally aimed at education and dissemination of information targeted at large scale prevention of disease in target populations, especially those populations which may be more difficult to access through existing mechanisms. For example, populations at a lower socioeconomic status are more likely to receive medical care and, when they receive care, it is often later in the disease process due to limitations in health education or financial resources (eg, to pay a co-pay or take time off from work to see a health care professional [HCP]). Family, school-based, and other community interventions aim to reach out to these populations for primary and secondary prevention.
Largely due to increases in childhood obesity, children have been the focus of several types of HL interventions. Family-based lifestyle interventions can be especially successful in the delivery of education and training in HL behaviors, especially with community engagement and applying a feedback system to the participants.161 As reported in a recent review article by Arnason et al,161 factors which supported successful family-based programming included: (1) 6-12-month duration; (2) community involvement; (3) the inclusion of a multidisciplinary team; (4) the inclusion of a mentor/role model; and (5) reinforcement with technology. These are themes which are consistent with other HL programming. Another model of HL programming focusing on children is school based. School-based interventions tend to focus on cognitive health as well as traditional health metrics and are largely focused on primary prevention. In a review of childhood obesity prevention as part of the Feel4Diabetes-intervention,162 Lambrinou et al163 included 27 programs, of which 15 were effective in influencing weight status or clinical indices of weight gain, 3 were effective in improving energy-balance behaviors and 9 were at least partially effective in improving some of these behaviors. After applying these best practices, the authors observed that there were major limitations which reduced the potential for intervention efficacy and suggested that face-to-face contact and collaborating with the target groups during development may improve the outcomes of such programs.162 These findings highlight the difficulties observed in attempting to create successful HL programs in children. In a recent Cochrane Systematic Review article from Martin et al164 the authors reviewed 18 studies with over 2300 overweight and obese children, assessing different types of interventions, including: (1) PA; (2) combined PA/HL education; and (3) dietary interventions. Most of these studies were focused on weight reduction/control and those which were effective seemed to improve cognition in children who were overweight and obese. These findings suggest that the benefits of HL programs may also help improve scholastic achievement in elementary school students.
Beyond childhood, HL programs usually target adult populations with specific health risks. For example, Luque et al165 reviewed the literature on barbershop based health programs, which are seen as exemplary as programs which aim to use culturally appropriate venues to reach target populations. After filtering the results of their literature search to 16 studies, the authors were able to demonstrate interventions targeting primarily prostate cancer and hypertension were effective in disseminating materials and training HCPs to conduct education and screening. Although there were some promising data supporting the transfer of health knowledge to customers, these were not conclusive.165 Other novel approaches to HL programs in the community are diverse, including: (1) increasing health care access for adults with intellectual and developmental disabilities166; (2) improving access and quality of community mental health services for women in ethnic minority settings;167 and (3) building an environment which supports increases in PA,168 among many others. The workplace is another viable setting for HL program implementation in the adult population.169 , 170 A significant percentage of the workforce presents with unhealthy lifestyle characteristics, including excess body weight, physical inactivity, and poor dietary patterns.171 , 172 Programs centered on screening lifestyle behaviors and risk factors, promotion of PA and healthy nutrition, weight loss, and smoking cessation have the potential to meaningfully impact HL behaviors and improve health outcomes if designed appropriately and follow evidence-based recommendations.169 , 170 , 173, 174, 175, 176 Given the percentage of adults who spend a significant portion of their day in the workplace around the world, implementing HL programs in this setting is a tremendous opportunity not to be missed. The American Heart Association has developed a “Workplace Health Achievement Index” for employers to “evaluate the comprehensiveness of their workplace health promotion program, identify opportunities for improvement, and benchmark their progress over time against their peer organizations.”177 This type of index will greatly assist in allowing employers to assess and optimize their worksite wellness programs; moving forward the potential positive impacts of worksite wellness programs will heavily depend on standardizing the field in a way that uses scientific evidence to guide program development and implementation.
The difference between HL behavior goals and current trends is readily apparent when population level PA data are considered, and, while trends are promising, there is clearly a continued need for an upscaling of effective implementation strategies. While PA remains inadequate on a population level since the first edition of the PA Guidelines for Americans in 2008, the percentage of adults meeting the recommended amounts of combined aerobic exercise and strength training have increased from 18.2% to 23.2%, and aerobic exercise alone from 43.4% to 53.3%. Additionally, the percentage of patients receiving advice to begin exercising has increased by 40%.178 , 179 Importantly, physically active HCPs are more likely to provide PA counseling to their patients and serve as PA role models. HCPs who are less knowledgeable about PA counseling are less likely to promote it in the clinic. PA levels of patients increase when recommended by HCPs who are viewed as credible by patients.180 , 181 The effect of different brief interventions applicable to the primary care setting was tested by Pears et al182 in a randomized controlled trial. The trial tested efficacy, feasibility, acceptability, and cost of very brief interventions including: (1) motivational; (2) pedometers; and (3) combined motivational and pedometers to increase PA in 394 adults. The authors found that although all interventions were at least somewhat effective, the least effective was the combined intervention, likely due to the inclusion of too many behavior change strategies at 1 time, which may have been overwhelming to the participants. The intervention which had the highest efficiency (speed of delivery and effectiveness) was the pedometer alone, which cost 25 UD dollars per participant, took less than 5 minutes to deliver and resulted in a 73% chance of having a positive effect on participants significantly increasing daily step count.182
There have also been attempts made at creating recommendations for primary care providers to help coach individuals to perform regular PA based on PA guidelines. For example, the National Institute for Health and Care Excellence in England released a statement on advising adults in primary care which stresses: (1) identification of those who may be inactive; (2) delivering and following up on brief advice; (3) incorporating brief advice in commissioning services to prevent or treat disease; (4) ensuring resources and information to access opportunities to be physically active are available and conditioned to the individual if possible; and (5) providing up to date information.183 The American Heart Association has previously published a scientific statement that comprehensively describes different, evidence-based approaches to assessing PA.184 To summarize, PA encouraged by HCPs in the clinical setting may be an effective approach to increasing adherence to this essential HL behavior. However, implementation has been inadequate despite various efforts to encourage PA promotion by HCPs in the clinical setting. Because the clinical environment seems to offer an ideal opportunity to discuss PA as a positive health behavior, the focus should remain on how to best utilize hospitals and clinics to help individuals increase PA to improve health outcomes.
The Impact of Unhealthy Lifestyle Behaviors on the Current Syndemic and the Role of Healthy Living in Syndemic Uncoupling
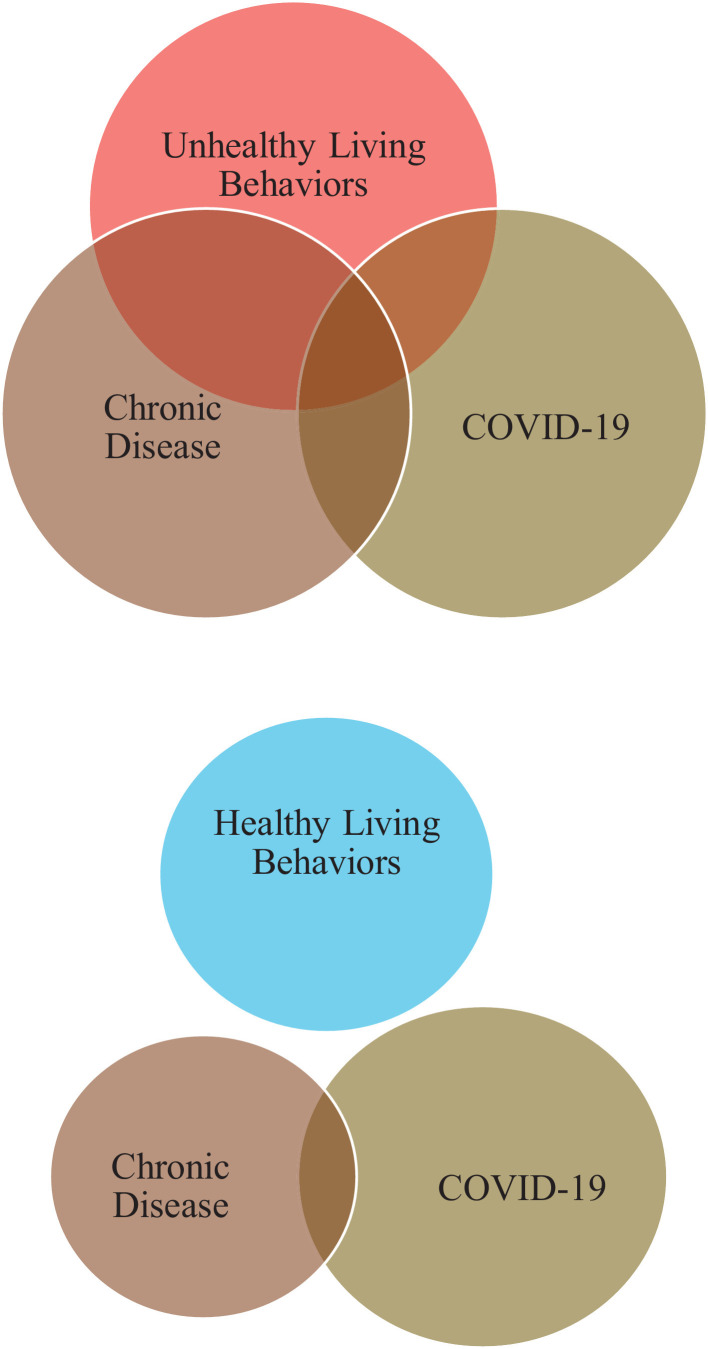
The current interrelationships amongst unhealthy lifestyle behaviors (eg, sedentary lifestyle, poor diet, excess body mass, smoking), chronic disease and the COVID-19 pandemic may be more appropriately characterized as a syndemic, defined as 2 or more health conditions or diseases that cluster by person, place or time which adversely interact with and negatively affect the outcomes of one another.176 , 185 , 186 Figure 3A illustrates the proposed unhealthy lifestyle behaviors – chronic disease – COVID-19 syndemic.186 In Figure 3A, unhealthy lifestyle behaviors are shifted towards chronic disease where its influence is clearly established; an independent interaction between unhealthy lifestyle behaviors and COVID-19, independent of chronic disease, appears highly likely although further research is needed to clarify the significance of this interaction.33 , 59 , 63 , 90 , 187 , 188 Even with the current knowledge gaps, the poorest outcomes are significantly increased when unhealthy lifestyle behaviors, 1 or more chronic disease diagnoses, and COVID-19 infection are simultaneously present on a global population level.4 , 8 , 9 , 68 , 186 , 187 Syndemics can also entail a social context, which is clearly relevant to the unhealthy lifestyle behaviors – chronic disease – COVID-19 syndemic given the disproportionate impact of this cluster of conditions in underserved communities.189, 190, 191, 192, 193 As such, while the unhealthy lifestyle behaviors – chronic disease – COVID-19 syndemic is a leading health concern on a global population level, the impact this cluster of conditions is having on underserved communities is immeasurably worse and requires immediate action. Conversely, Figure 3B illustrates the syndemic uncoupling that can be expected through an increased prevalence of HL behaviors (ie, physically active lifestyle, heathy diet, normal body weight, and not smoking); chronic disease prevalence would undoubtably shrink with a significant decrease in unhealthy lifestyle behaviors, as would the overlap between chronic disease and COVID-19 infection, an overlap which increases risk for a poor outcome. Cases of viral infection would also likely be less severe in those individuals not diagnosed with 1 or more chronic diseases if HL behaviors were more prevalent given the improved ability to successfully overcome a viral infection.3 , 8 , 9 , 34 , 36 , 63 , 187 , 194 The world will eventually move past the COVID-19 pandemic, hospitalizations and deaths associated with SARS-CoV-2 infection will subside and society will return to normal activities. Even so, the circle associated with COVID-19 in Fig 3A and B will remain a relevant component of this syndemic construct and syndemic uncoupling. Recent evidence links a higher prevalence of severe influenza cases in patients with co-existing chronic diseases.195 Seroprotection from the influenza vaccine has also been shown to be significantly higher in individuals who adhere to HL behaviors, such as PA.36 , 196 Therefore, the COVID-19 circle in Fig 3A and B will transition to viral infections/future pandemics once we overcome the COVID-19 pandemic. In this context, improving HL behaviors must be viewed as a global imperative to uncouple this syndemic construct.6 , 9 As mentioned previously, while promotion of HL behaviors and the expanded practice of HLM is essential on a population level, a particular focus is desperately needed in underserved communities.191 , 192 , 197 , 198
FIG 3.
A The Syndemic of Unhealthy Living Behaviors, Chronic Disease and COVID-19.
Conclusions
It has been well established that adherence of HL behaviors are essential to optimizing quality of life, the ability to perform activities of daily living, and realizing positive health outcomes. The COVID-19 pandemic has further demonstrated the importance of HL behaviors on minimizing the risk for detrimental health events. Prior to the COVID-19 pandemic, the importance of preventing and treating chronic conditions, such as cardiovascular disease, diabetes, obesity and certain cancers, through the promotion of HL behaviors and the practice of HLM has steadily gained acceptance, although implementation continues to lag behind acceptance. The literature rapidly emerging during the COVID-19 pandemic strongly indicates individuals that adhere to HL behaviors have advanced immune system resiliency and therefore a reduced risk of poor outcomes if infected with the SARS-CoV-2 virus. The importance of HL behaviors in the context of the COVID-19 pandemic is 2-fold: 1) HL behaviors enhance immune function, thereby improving the body's ability to prevent and fight a viral infection; and 2) HL behaviors reduce the likelihood of the chronic conditions (ie, obesity, 1 or more chronic disease diagnoses, smoking) that have been associated with poor outcomes in those infected with the SARS-CoV-2 virus. Now more than ever, promotion of HL behaviors and HLM are essential to combat both chronic disease and viral pandemics. Ongoing HL and HLM research (ie, knowledge discovery and dissemination) are essential to shape effective education, policy, and implementation strategies. The current HL-PIVOT position statement serves as a basis for this 4-pillar approach moving forward.
Appendix 1
Members of the HL-PIVOT Network
Antonio Abbate, MD, PhD, Virginia Commonwealth University, Richmond, Virginia, USA
Gopala Krishna Alaparthi, PhD, PT, University of Sharjah, United Arab Emirates
Sampath Kumar Amaravadi MPT, PhD, CRFC, Gulf Medical University, Ajman, United Arab Emirates
Ross Arena, PhD, PT, University of Illinois at Chicago, Chicago, Illinois, USA
Leslie D. Austford, MN, MBA, CMPE, TotalCardiology Research Network, and TotalCardiologyTM, Calgary, Alberta, Canada
Robert S. Axtell, PhD, Southern Connecticut State University, New Haven, Connecticut, USA
Abraham Samuel Babu, PhD, PT, Manipal College of Health Professions, Manipal Academy of Higher Education, Manipal, India
James R. Bagley, PhD, San Francisco State University, San Francisco, California, USA
Lauren Bates, MA, University of North Carolina, Chapel Hill, North Carolina, USA
Kathy Berra, MSN, NP-BC, FAANP, FPCNA, FAHA, FAAN, The LifeCare Company, Redwood City, California and Stanford University, Menlo Park, California, USA
Jarett D. Berry, MD, UT Southwestern Medical Center, Dallas, TX, USA
Samantha Bond, MS, University of Illinois at Chicago, Chicago, Illinois, USA
Sandra A Billinger, PhD, University of Kansas Medical Center, Kansas City, Kansas, USA
W. Scott Black, MD, University of Kentucky, Lexington, Kentucky, USA
Audrey Borghi-Silva, PhD, PT, Federal University of Sao Carlos, Sao Paulo, Brazil
Harry Brar, POWERbreathe International Ltd, Warwickshire, UK
Patrice Brassard, PhD, Université Laval, Québec, Canada
Peter H. Brubaker PhD, Wake Forest University, Winston Salem, North Carolina, USA
Paolo Brugnoli, CosMed, Inc, Rome, Italy
Marco Brugnoli, CosMed, Inc, Rome, Italy
Tavis S. Campbell, PhD, University of Calgary, Calgary, Alberta, Canada
Salvatore Carbone, PhD, MS, Virginia Commonwealth University, Richmond, Virginia, USA
Brian Carlin, MD, Sleep Medicine and Lung Health Consultants LLC, and Pittsburgh Critical Care Associates, Pittsburgh, Pennsylvania, USA
Kalyana Chakravarthy B, PhD, PT, University of Sharjah, United Arab Emirates
Michael J. Chapman, ND, Genova Diagnostics, Asheville, North Carolina USA
Paul J. Chase, PhD, Ohio University, Athens, Ohio, USA
Sandeep Chaudhry, MD, MET-TEST, Roswell, Georgia, USA
Jeffrey W Christle, PhD, CEP, Stanford University, Stanford, CA, USA
Andrew J Stewart Coats, Monash University, Melbourne, Australia and University of Warwick, Coventry, United Kingdom
Naama W. Constantini, MD, DFM, FACSM, Heidi Rothberg Sport Medicine Center, Shaare Zedek Medical Center, Jerusalem, Israel
Jeff S. Coombes, PhD, University of Queensland, Brisbane, Queensland, Australia.
Andréa Lúcia Gonçalves da Silva, PhD, PT, University of Santa Cruz do Sul, Santa Cruz do Sul, Brazil
Luiz Carlos Soares de Carvalho Junior, PhD, PT, Federal University of Recôncavo of Bahia, Cruz das Almas, Bahia, Brazil
Mildred Vanessa López Cabrera, PhD, Monterrey Institute of Technology, Monterrey, Mexico.
Patricia M. Devers, DO, Genova Diagnostics, Asheville, North Carolina USA
Victor Zuniga Dourado, PhD, PT, Federal University of São Paulo, Santos, São Paulo, Brazil
Daniela Bassi Dibai, PhD, PT, Ceuma University, São Luís, Brazil
Snehil Dixit, PhD, PT, King Khalid University, Abha, Saudi Arabia
Devon Dorbrosielski, PhD, Towson University, Towson, Maryland, USA
Victor Zuniga Dourado, PhD, PT, Federal University of São Paulo, Santos, SP, Brazil.
Brandon Dykstra, PhD, Taylor University, Upland, Indiana, USA
Patrick Dunn, PhD, American Heart Association and Walden University, Dallas, Texas, USA
Diane Eastabrook, Freelance Journalist, Chicago, Illinois, USA
Mark Faghy, PhD, University of Derby, Derby, United Kingdom
Barry A. Franklin, PhD, Beaumont Health, Royal Oak, Michigan, USA
Nina Cherie Franklin, PhD, Synthycity, LLC, Atlanta, Georgia, USA
Ross Goglia, Cubii, Chicago, Illinois, USA
Rochelle L. Goldsmith, MS, Columbia University, New York, New York, USA
Sherry L. Grace, PhD, York University & University Health Network, Toronto, Canada and International Council of Cardiovascular Prevention and Rehabilitation, Markham, Ontario, Canada
Giovanni Grazzi, MD, Department of Neuroscience and Rehabilitation, University of Ferrara, Ferrara, Italy.
Gregory J. Grosicki, PhD, Georgia Southern University (Armstrong Campus), Savannah, Georgia, USA
Solange Guizilini, PhD, PT, Federal University of Sao Paulo, São Paulo, Brazil.
Grenita Hall, PhD, University of Illinois at Chicago, Chicago, Illinois, USA
Rebecca H. Haraf, MD, University Hospitals Cleveland Medical Center/Case Western Reserve University, Louis Stokes Cleveland VAMC, Cleveland, Ohio, USA
Matthew P. Harber, PhD, Ball State University, Muncie, Indiana, USA
Erik Hayes, PhD, Taylor University, Upland, Indiana, USA
Samuel. A. E. Headley, PhD, FACSM, Springfield College, Springfield, Massachusetts, USA
Kevin S. Heffernan, PhD, Syracuse University, Syracuse, New York, USA
Sylvia Herbozo, PhD, University of Illinois at Chicago, Chicago, Illinois, USA
John P. Heybach, PhD, Altus Academy, Chicago, Illinois, USA
Andrew P. Hills, PhD, University of Tasmania, Tasmania, Australia
Anne E Holland PhD, PT, Monash University and Alfred Health, Melbourne, Australia
Martha I. Huizar, MS, RDN, University of Illinois at Chicago, Chicago, Illinois, USA
Reed Humphrey, PhD, PT, University of Montana, Missoula, Montana, USA
Lee Ingle, PhD, University of Hull, Hull, UK
Panniyammakal Jeemon, Ph.D, MPH, FESC, Sree Chitra Tirunal Institute for Medical Sciences and Technology, Trivandrum, Kerala, India
Richard A Josephson, MS, MD, Case Western Reserve University and University Hospitals of Cleveland, Cleveland, Ohio, USA
Leonard A. Kaminsky, PhD, Ball State University, Muncie, Indiana, USA
Duncan Kerr, POWERbreathe International Ltd, Warwickshire, UK
Bradley Kendall, PhD, CSCS, Taylor University, Upland, Indiana, USA
Aditya Khetan, MD, McMaster University, Hamilton, Ontario, Canada
Peter Kokkinos, PhD, Rutgers University, New Brunswick, NJ, and Veterans Affairs Medical Center Washington, DC, USA
Sundar Kumar, PhD, Ramaiah Medical College, Bangalore, India
Deepika R. Laddu, PhD, University of Illinois at Chicago, Chicago, Illinois, USA
Jari Laukkanen, MD, PhD, Institute of Clinical Medicine, University of Eastern Finland, Kuopio, Finland
Carl J Lavie, MD, John Ochsner Heart and Vascular Institute, Ochsner
Clinical School-University of Queensland School of Medicine, New Orleans, LA, USA
C. Matthew Lee, PhD, San Francisco State University, San Francisco, California, USA
Gregory D. Lewis, MD, Massachusetts General Hospital, Boston, Massachusetts, USA
Andrew T. Lovering, PhD, University of Oregon, Eugene, Oregon, USA
Steven Loy, PhD, Department of Kinesiology, California State University Northridge, Northridge, California, USA
Sara Maldonado-Martín, PhD, Department of Physical Education and Sports, University of the Basque Country (UPV/EHU), Basque Country, Spain
Stephen Maris, PhD, Department of Exercise Science and Athletic Training, Springfield College, Springfield Massachusetts, USA
Susan Marzolini, RKin, PhD, KITE Research Institute, Toronto Rehabilitation and University Health Network, Cardiovascular Prevention and Rehabilitation Program, Toronto, Canada
Supriya D. Mehta, MHS, PhD, University of Illinois at Chicago, Chicago, Illinois, USA
Norman Morris, PhD, PT, Griffith University and Metro North Hospital and Health Service, The Prince Charles Hospital, Queensland, Australia
Jonathan Myers, PhD, VA Palo Alto Health Care System and Stanford University, Palo Alto, California, USA
Jagat Narula, MD, PhD, Mount Sinai Heart, New York, New York, USA
Javaid Nauman, PhD, United Arab Emirates University, Al-Ain, UAE. Norwegian University of Science and Technology, Trondheim, Norway.
Alan M. Nevill, PhD, University of Wolverhampton, Walsall, United Kingdom
Josef Niebauer, MD, PhD, MBA, Institute of Sports Medicine, Prevention and Rehabilitation, Paracelsus Medical University, Salzburg, Austria.
James H. O'Keefe, MD, University of Missouri, Kansas City, Kansas, USA
Silvia Lizett Olivares Olivars, PhD, Monterrey Institute of Technology, Monterrey, Mexico
Francisco B. Ortega, PhD, University of Granada, Granada, Spain and Karolinska Institute, Stockholm, Sweden.
Cemal Ozemek, PhD, University of Illinois at Chicago, Chicago, Illinois, USA
Ambarish Pandey, MD, MSCS, University of Texas Southwestern Medical Center Dallas, Texas, USA
Edward Parsley, DO, Medical Consultant, San Diego, California, USA
James E. Peterman, PhD, Ball State University, Muncie, Indiana, USA
Shane A. Phillips, PhD, PT, University of Illinois at Chicago, Chicago, Illinois, USA
Dejana Popovic, MD, PhD, Clinic for Cardiology, Clinical Center of Serbia, University of Belgrade, Belgrade, Serbia
Axel Pressler, MD, PhD, FESC, Private Center of Sports Cardiology & Prevention, Munich, Germany
Nicolaas P. Pronk, PhD, MA, HealthPartners Institute, Bloomington, Minnesota, and Harvard TH Chan School of Public Health, Boston, Massachusetts, USA
Chathuranga Ranasinghe MBBS, D.Sp.Med,PhD, Faculty of Medicine, University of Colombo, Colombo, Sri Lanka
Kyle Rausch, EdD, University of Illinois at Chicago, Chicago, Illinois, USA
Robert Ross, PhD, RKin, Queen's University, Kingston, Ontario, Canada.
Michel Silva Reis, PhD, PT, Federal University of Rio de Janeiro, Rio de Janeiro, Brazil
Fabrizio Ricci, MD, PhD, FEACVI, G.d'Annunzio University, Chieti, Italy
Daniel S. Rubin, M.D., M.S, University of Chicago, Chicago, Illinois, USA
Ahmad Sabbahi, PT, PhD, University of Illinois at Chicago, Chicago, Illinois, USA and Cairo University, Giza, Egypt
James F. Sallis, PhD, University of California, San Diego, La Jolla, California, USA
Patrick Savage, MS, University of Vermont Medical Center, Cardiac Rehabilitation Program, South Burlington, Vermont, USA
Richard Severin, PT, DPT, CCS, University of Illinois at Chicago, Chicago, Illinois and Baylor University, Waco, Texas, USA
Bruno P. C. Smirmaul, PhD, Yolife GmbH, Berlin, Germany (Rio Claro, São Paulo, Brazil)
Dorthe Stensvold, PhD, Norwegian University of Science and Technology, Trondheim, Norway
Lee Stoner, PhD, MPH, FACSM, University of North Carolina, Chapel Hill, North Carolina, USA
Lindsey Strieter, MS, University of Illinois at Chicago, Chicago, Illinois, USA
Xing Guo Sun, MD, Beijing Fuwai Hospital National Center for Cardiovascular Diseases, Beijing, China
Renata Trimer, PhD, PT, University of Santa Cruz do Sul, Santa Cruz do Sul, Brazil
Jean-Luc Vachiery, MD, Cliniques Universitaires de Bruxelles-Hôpital, Brussels, Belgium
Amber Vermeesch, PhD, University of Portland, Portland, Oregon, USA
Laurie P. Whitsel, PhD, Ligonier, Pennsylvania, USA
Mark A. Williams, PhD, Creighton University, Omaha, Nebraska, USA
Ulrik Wisløff, PhD, Norwegian University of Science and Technology, Trondheim, Norway, and School of Human Movement and Nutrition Science, University of Queensland, Queensland, Australia.
Abebaw M Yohannes PhD, MSc, FCCP, Azusa Pacific University, Azusa, California, USA
Minoru Yamada, PT, PhD, University of Tsukuba, Otsuka, Bunkyo-ku, Tokyo, Japan
Gabriel Zieff, MA, University of North Carolina, Chapel Hill, North Carolina, USA
Footnotes
The authors have no conflicts of interest to disclose.
References
- 1.Arena R, McNeil A, Sagner M, Lavie CJ. Healthy living: the universal and timeless medicine for healthspan. Prog Cardiovasc Dis. 2017;59:419–421. doi: 10.1016/j.pcad.2017.01.007. [DOI] [PubMed] [Google Scholar]
- 2.Cheng KJG, Sun Y, Monnat SM. COVID-19 death rates are higher in rural counties with larger shares of Blacks and Hispanics. J Rural Health. 2020;36:602–608. doi: 10.1111/jrh.12511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hamer M, Kivimäki M, Gale CR, Batty GD. Lifestyle risk factors, inflammatory mechanisms, and COVID-19 hospitalization: a community-based cohort study of 387,109 adults in UK. Brain Behav Immun. 2020;87:184–187. doi: 10.1016/j.bbi.2020.05.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lavie CJ, Sanchis-Gomar F, Henry BM, Lippi G. COVID-19 and obesity: links and risks. Expert Rev Endocrinol Metab. 2020;15:215–216. doi: 10.1080/17446651.2020.1767589. [DOI] [PubMed] [Google Scholar]
- 5.Maroko AR, Nash D, Pavilonis BT. COVID-19 and inequity: a comparative spatial analysis of new york city and chicago hot spots. J Urban Health. 2020;97:461–470. doi: 10.1007/s11524-020-00468-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nieman DC. Coronavirus disease-2019: a tocsin to our aging, unfit, corpulent, and immunodeficient society. J Sport Health Sci. 2020;9:293–301. doi: 10.1016/j.jshs.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brawner CA, Ehrman JK, Bole S, et al. Inverse relationship of maximal exercise capacity to hospitalization secondary to coronavirus disease 2019. Mayo Clin Proc. 2021;96:32–39. doi: 10.1016/j.mayocp.2020.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Burtscher J, Millet GP, Burtscher M. Low cardiorespiratory and mitochondrial fitness as risk factors in viral infections: implications for COVID-19. Br J Sports Med. 2020 doi: 10.1136/bjsports-2020-103572. https://doi.org/10.1136/bjsports-2020-103572 bjsports-2020-103572. Online ahead of print. [DOI] [PubMed] [Google Scholar]
- 9.Chesnut WM, MacDonald S, Wambier CG. Could diet and exercise reduce risk of COVID-19 syndemic? Med Hypotheses. 2021;148 doi: 10.1016/j.mehy.2021.110502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dunton GF, Do B, Wang SD. Early effects of the COVID-19 pandemic on physical activity and sedentary behavior in children living in the U.S. BMC Public Health. 2020;20:1351. doi: 10.1186/s12889-020-09429-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ferrante G, Camussi E, Piccinelli C, et al. Did social isolation during the SARS-CoV-2 epidemic have an impact on the lifestyles of citizens? Epidemiol Prev. 2020;44:353–362. doi: 10.19191/EP20.5-6.S2.137. [DOI] [PubMed] [Google Scholar]
- 12.Giacalone D, Frøst MB, Rodríguez-Pérez C. Reported changes in dietary habits during the COVID-19 lockdown in the Danish population: the Danish COVIDiet study. Front Nutr. 2020;7 doi: 10.3389/fnut.2020.592112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Meyer J, McDowell C, Lansing J, et al. Changes in physical activity and sedentary behavior in response to COVID-19 and their associations with mental health in 3052 US adults. Int J Environ Res Public Health. 2020;17:6469. doi: 10.3390/ijerph17186469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zheng C, Huang WY, Sheridan S, Sit CH, Chen XK, Wong SH. COVID-19 pandemic brings a sedentary lifestyle in young adults: a cross-sectional and longitudinal study. Int J Environ Res Public Health. 2020;17:6035. doi: 10.3390/ijerph17176035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.The L. Global health: time for radical change? Lancet. 2020;396:1129. doi: 10.1016/S0140-6736(20)32131-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Arena R, Lavie CJ. The global path forward - healthy living for pandemic event protection (HL - PIVOT) Prog Cardiovasc Dis. 2021;64:96–101. doi: 10.1016/j.pcad.2020.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bond S, Arena R, Berra K, Popovic D, Smirmaul BPC, Ortega FB. Introducing the HL-PIVOT network: promoting human resilience and quality of life through healthy living initiatives. J Cardiopulm Rehabil Prev. 2020;40:356–358. doi: 10.1097/HCR.0000000000000555. [DOI] [PubMed] [Google Scholar]
- 18.Carbone S, Lee PJH, Mauro AG, et al. Interleukin-18 mediates cardiac dysfunction induced by western diet independent of obesity and hyperglycemia in the mouse. Nutr Diabetes. 2017;7 doi: 10.1038/nutd.2017.1. e258-e258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wen H, Gris D, Lei Y, et al. Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat Immunol. 2011;12:408–415. doi: 10.1038/ni.2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.L'Homme L, Esser N, Riva L, et al. Unsaturated fatty acids prevent activation of NLRP3 inflammasome in human monocytes/macrophages. J Lipid Res. 2013;54:2998–3008. doi: 10.1194/jlr.M037861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sui Y-H, Luo W-J, Xu Q-Y, Hua J. Dietary saturated fatty acid and polyunsaturated fatty acid oppositely affect hepatic NOD-like receptor protein 3 inflammasome through regulating nuclear factor-kappa B activation. World J Gastroentero. 2016;22:2533–2544. doi: 10.3748/wjg.v22.i8.2533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lavie CJ, Ozemek C, Carbone S, Katzmarzyk PT, Blair SN. Sedentary behavior, exercise, and cardiovascular health. Circ Res. 2019;124:799–815. doi: 10.1161/CIRCRESAHA.118.312669. [DOI] [PubMed] [Google Scholar]
- 23.Löllgen H, Böckenhoff A, Knapp G. Physical activity and all-cause mortality: an updated meta-analysis with different intensity categories. Int J Sports Med. 2009;30:213–224. doi: 10.1055/s-0028-1128150. [DOI] [PubMed] [Google Scholar]
- 24.Landi F, Russo A, Cesari M, et al. Walking one hour or more per day prevented mortality among older persons: results from ilSIRENTE study. Prev Med. 2008;47:422–426. doi: 10.1016/j.ypmed.2008.06.020. [DOI] [PubMed] [Google Scholar]
- 25.Stewart RAH, Held C, Hadziosmanovic N, et al. Physical activity and mortality in patients with stable coronary heart disease. J Am Coll Cardiol. 2017;70:1689–1700. doi: 10.1016/j.jacc.2017.08.017. [DOI] [PubMed] [Google Scholar]
- 26.O'Donovan G, Lee IM, Hamer M, Stamatakis E. Association of “Weekend Warrior” and other leisure time physical activity patterns with risks for all-cause, cardiovascular disease, and cancer mortality. JAMA Intern. Med. 2017;177:335–342. doi: 10.1001/jamainternmed.2016.8014. [DOI] [PubMed] [Google Scholar]
- 27.Hill K, Gardiner PA, Cavalheri V, Jenkins SC, Healy GN. Physical activity and sedentary behaviour: applying lessons to chronic obstructive pulmonary disease. Intern Med J. 2015;45:474–482. doi: 10.1111/imj.12570. [DOI] [PubMed] [Google Scholar]
- 28.Pahor M, Guralnik JM, Ambrosius WT, et al. Effect of structured physical activity on prevention of major mobility disability in older adults: the LIFE study randomized clinical trial. JAMA. 2014;311:2387–2396. doi: 10.1001/jama.2014.5616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Füzéki E, Engeroff T, Banzer W. Health Benefits of light-intensity physical activity: a systematic review of accelerometer data of the national health and nutrition examination survey (NHANES) Sports Med (Auckland, NZ) 2017;47:1769–1793. doi: 10.1007/s40279-017-0724-0. [DOI] [PubMed] [Google Scholar]
- 30.Geneen LJ, Moore RA, Clarke C, Martin D, Colvin LA, Smith BH. Physical activity and exercise for chronic pain in adults: an overview of Cochrane Reviews. Cochrane Database Syst Rev. 2017;1 doi: 10.1002/14651858.CD011279.pub2. CD011279-CD011279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Buman MP, Hekler EB, Haskell WL, et al. Objective light-intensity physical activity associations with rated health in older adults. Am J Epidemiol. 2010;172:1155–1165. doi: 10.1093/aje/kwq249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nunn AVW, Guy GW, Brysch W, et al. SARS-CoV-2 and mitochondrial health: implications of lifestyle and ageing. Immun Ageing. 2020;17:33. doi: 10.1186/s12979-020-00204-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Simpson RJ, Katsanis E. The immunological case for staying active during the COVID-19 pandemic. Brain Behav Immun. 2020;87:6–7. doi: 10.1016/j.bbi.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Filgueira TO, Castoldi A, Santos LER, et al. The relevance of a physical active lifestyle and physical fitness on immune defense: mitigating disease burden, with focus on COVID-19 consequences. Front Immunol. 2021;12:587146. doi: 10.3389/fimmu.2021.587146. eCollection 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tavakol Z, Ghannadi S, Tabesh MR, et al. Relationship between physical activity, healthy lifestyle and COVID-19 disease severity; a cross-sectional study. J Public Health. 2021:1–9. doi: 10.1007/s10389-020-01468-9. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Woods JA, Keylock KT, Lowder T, et al. Cardiovascular exercise training extends influenza vaccine seroprotection in sedentary older adults: the immune function intervention trial. J Am Geriatr Soc. 2009;57:2183–2191. doi: 10.1111/j.1532-5415.2009.02563.x. [DOI] [PubMed] [Google Scholar]
- 37.Lu C, Chi X, Liang K, et al. Moving more and sitting less as healthy lifestyle behaviors are protective factors for insomnia, depression, and anxiety among adolescents during the COVID-19 pandemic. Psychol Res Behav Manag. 2020;13:1223–1233. doi: 10.2147/PRBM.S284103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kohl HW, 3rd, Craig CL, Lambert EV, et al. The pandemic of physical inactivity: global action for public health. Lancet. 2012;380:294–305. doi: 10.1016/S0140-6736(12)60898-8. [DOI] [PubMed] [Google Scholar]
- 39.Hall G, Laddu DR, Phillips SA, Lavie CJ, Arena R. A tale of two pandemics: How will COVID-19 and global trends in physical inactivity and sedentary behavior affect one another? Prog Cardiovasc Dis. 2021;64:108–110. doi: 10.1016/j.pcad.2020.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ammar A, Brach M, Trabelsi K, et al. Hoekelmann A and consortium OBotE-C. Effects of COVID-19 home confinement on eating behaviour and physical activity: results of the ECLB-COVID19 international online survey. Nutrients. 2020;12:1583. doi: 10.3390/nu12061583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sassone B, Mandini S, Grazzi G, Mazzoni G, Myers J, Pasanisi G. Impact of COVID-19 pandemic on physical activity in patients with implantable cardioverter-defibrillators. J Cardiopulm Rehabil. 2020;40:285–286. doi: 10.1097/HCR.0000000000000539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Arena R, McNeil A, Street S, et al. Let us talk about moving: reframing the exercise and physical activity discussion. Curr Probl Cardiol. 2018;43:154–179. doi: 10.1016/j.cpcardiol.2017.06.002. [DOI] [PubMed] [Google Scholar]
- 43.Smirmaul BPC, Arena R. The urgent need to sit less and move more during the COVID-19 pandemic. J Cardiopulm Rehabil. 2020;40:287–289. doi: 10.1097/HCR.0000000000000538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ross R, Blair SN, Arena R, et al. Importance of assessing cardiorespiratory fitness in clinical practice: a case for fitness as a clinical vital sign: a scientific statement from the American Heart Association. Circulation. 2016;134:e653–e699. doi: 10.1161/CIR.0000000000000461. [DOI] [PubMed] [Google Scholar]
- 45.Jae SY, Heffernan KS, Kurl S, et al. Cardiorespiratory fitness, inflammation, and the incident risk of pneumonia. J Cardiopulm Rehabil. 2021 doi: 10.1097/HCR.0000000000000581. Online ahead of print. [DOI] [PubMed] [Google Scholar]
- 46.Zbinden-Foncea H, Francaux M, Deldicque L, Hawley JA. Does high cardiorespiratory fitness confer some protection against proinflammatory responses after infection by SARS-CoV-2? Obesity (Silver Spring) 2020;28:1378–1381. doi: 10.1002/oby.22849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Harber MP, Kaminsky LA, Arena R, et al. Impact of cardiorespiratory fitness on all-cause and disease-specific mortality: advances since 2009. Prog Cardiovasc Dis. 2017;60:11–20. doi: 10.1016/j.pcad.2017.03.001. [DOI] [PubMed] [Google Scholar]
- 48.Martin BJ, Arena R, Haykowsky M, et al. Cardiovascular fitness and mortality after contemporary cardiac rehabilitation. Mayo Clin Proc. 2013;88:455–463. doi: 10.1016/j.mayocp.2013.02.013. [DOI] [PubMed] [Google Scholar]
- 49.Dayton JD, Ford K, Carroll SJ, Flynn PA, Kourtidou S, Holzer RJ. The deconditioning effect of the COVID-19 pandemic on unaffected healthy children. Pediatr Cardiol. 2021:1–6. doi: 10.1007/s00246-020-02513-w. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Raman B, Cassar MP, Tunnicliffe EM, et al. Medium-term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge. EClinicalMedicine. 2021;31 doi: 10.1016/j.eclinm.2020.100683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ansai N, Wambogo EA. Fruit and vegetable consumption among adults in the United States, 2015-2018. NCHS Data Brief. 2021;397:1–8. [PubMed] [Google Scholar]
- 52.Miller V, Mente A, Dehghan M, et al. Fruit, vegetable, and legume intake, and cardiovascular disease and deaths in 18 countries (PURE): a prospective cohort study. Lancet. 2017;390:2037–2049. doi: 10.1016/S0140-6736(17)32253-5. [DOI] [PubMed] [Google Scholar]
- 53.De Filippis F, Pellegrini N, Vannini L, et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut. 2016;65:1812–1821. doi: 10.1136/gutjnl-2015-309957. [DOI] [PubMed] [Google Scholar]
- 54.Estruch R, Ros E, Salas-Salvado J, et al. Primary prevention of cardiovascular disease with a mediterranean diet. N Engl J Med. 2013;368:1279–1290. doi: 10.1056/NEJMoa1200303. [DOI] [PubMed] [Google Scholar]
- 55.Schulze MB, Martínez-González MA, Fung TT, Lichtenstein AH, Forouhi NG. Food based dietary patterns and chronic disease prevention. BMJ. 2018;361:k2396. doi: 10.1136/bmj.k2396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Shikany JM, Safford MM, Newby PK, Durant RW, Brown TM, Judd SE. Southern dietary pattern is associated with hazard of acute coronary heart disease in the reasons for geographic and racial differences in stroke (REGARDS) study. Circulation. 2015;132:804–814. doi: 10.1161/CIRCULATIONAHA.114.014421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Van Horn L, Carson JAS, Appel LJ, et al. Recommended dietary pattern to achieve adherence to the American Heart Association/American College of Cardiology (AHA/ACC) guidelines: a scientific statement from the American Heart Association. Circulation. 2016;134:e505–e529. doi: 10.1161/CIR.0000000000000462. [DOI] [PubMed] [Google Scholar]
- 58.Vadiveloo M, Lichtenstein AH, Anderson C, et al. Rapid diet assessment screening tools for cardiovascular disease risk reduction across healthcare settings: a scientific statement from the American Heart Association. Circulation. 2020;13 doi: 10.1161/HCQ.0000000000000094. [DOI] [PubMed] [Google Scholar]
- 59.Butler MJ, Barrientos RM. The impact of nutrition on COVID-19 susceptibility and long-term consequences. Brain Behav Immun. 2020;87:53–54. doi: 10.1016/j.bbi.2020.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Huizar MI, Arena R, Laddu DR. The global food syndemic: the impact of food insecurity, malnutrition and obesity on the healthspan amid the COVID-19 pandemic. Prog Cardiovasc Dis. 2021;64:105–107. doi: 10.1016/j.pcad.2020.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Belanger MJ, Hill MA, Angelidi AM, Dalamaga M, Sowers JR, Mantzoros CS. Covid-19 and disparities in nutrition and obesity. New Engl J Med. 2020;383:e69. doi: 10.1056/NEJMp2021264. [DOI] [PubMed] [Google Scholar]
- 62.Naja F, Hamadeh R. Nutrition amid the COVID-19 pandemic: a multi-level framework for action. Eur J Clin Nutr. 2020;74:1117–1121. doi: 10.1038/s41430-020-0634-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bousquet J, Anto JM, Iaccarino G, et al. Is diet partly responsible for differences in COVID-19 death rates between and within countries? Clin Transl Allergy. 2020;10:16. doi: 10.1186/s13601-020-00323-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ryan DH, Kahan S. Guideline recommendations for obesity management. Med Clin North Am. 2018;102:49–63. doi: 10.1016/j.mcna.2017.08.006. [DOI] [PubMed] [Google Scholar]
- 65.Peto R, Whitlock G, Jha P. Effects of obesity and smoking on U.S. Life Expectancy. 2010;362:855–857. doi: 10.1056/NEJMc1000079. [DOI] [PubMed] [Google Scholar]
- 66.Raynor HA, Champagne CM. Position of the academy of nutrition and dietetics: interventions for the treatment of overweight and obesity in adults. J Acad Nutr Diet. 2016;116:129–147. doi: 10.1016/j.jand.2015.10.031. [DOI] [PubMed] [Google Scholar]
- 67.Finer N, Garnett SP and Bruun JM. COVID-19 and obesity. 2020;10:e12365-e12365. [DOI] [PMC free article] [PubMed]
- 68.Cedano J, Fabian Corona E, Gonzalez-Lara M, et al. Characteristics and outcomes of patients with COVID-19 in an intensive care unit of a community hospital; retrospective cohort study. J Commun Hosp Intern Med Perspect. 2021;11:27–32. doi: 10.1080/20009666.2020.1830516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Czernichow S, Bain SC, Capehorn M, et al. Costs of the COVID-19 pandemic associated with obesity in Europe: a health-care cost model. Clin Obes. 2021;11:e12442. doi: 10.1111/cob.12442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Semlitsch T, Stigler FL, Jeitler K, Horvath K, Siebenhofer A. Management of overweight and obesity in primary care-a systematic overview of international evidence-based guidelines. Obes Rev. 2019;20:1218–1230. doi: 10.1111/obr.12889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gonzalez-Campoy JM, St Jeor ST, Castorino K, et al. Clinical practice guidelines for healthy eating for the prevention and treatment of metabolic and endocrine diseases in adults: cosponsored by the American Association of Clinical Endocrinologists/the American College of Endocrinology and the Obesity Soc. Endocr Pract. 2013;19(Suppl 3):1–82. doi: 10.4158/EP13155.GL. [DOI] [PubMed] [Google Scholar]
- 72.Fernández-Ruiz VE, Armero-Barranco D, Paniagua-Urbano JA, Sole-Agusti M, Ruiz-Sánchez A, Gómez-Marín J. Short-medium-long-term efficacy of interdisciplinary intervention against overweight and obesity: randomized controlled clinical trial. Int J Nurs Pract. 2018;24 doi: 10.1111/ijn.12690. e12690-e12690. [DOI] [PubMed] [Google Scholar]
- 73.Cochrane AJ, Dick B, King NA, Hills AP, Kavanagh DJ. Developing dimensions for a multicomponent multidisciplinary approach to obesity management: a qualitative study. BMC Public Health. 2017;17:814. doi: 10.1186/s12889-017-4834-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Montesi L, El Ghoch M, Brodosi L, Calugi S, Marchesini G, Dalle Grave R. Long-term weight loss maintenance for obesity: a multidisciplinary approach. Diabetes Metab Syndr Obes. 2016;9:37–46. doi: 10.2147/DMSO.S89836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.World Health O. World Health Statistics 2019: Monitoring health for the SDGs. 2019:5-10.
- 76.Sharpe T, Alsahlanee A, Ward KD, Doyle F. Systematic review of clinician-reported barriers to provision of smoking cessation interventions in hospital inpatient settings. J Smok Cessat. 2018;13:233–243. [Google Scholar]
- 77.Goodchild M, Nargis N, Tursan d'Espaignet E. Global economic cost of smoking-attributable diseases. Tob Control. 2018;27:58–64. doi: 10.1136/tobaccocontrol-2016-053305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Umnuaypornlert A, Kanchanasurakit S, Lucero-Prisno DEI, Saokaew S. Smoking and risk of negative outcomes among COVID-19 patients: A systematic review and meta-analysis. Tob Induc Dis. 2021;19:09. doi: 10.18332/tid/132411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Selby P, Hunter K, Rogers J, et al. How to adapt existing evidence-based clinical practice guidelines: a case example with smoking cessation guidelines in Canada. BMJ Open. 2017;7 doi: 10.1136/bmjopen-2017-016124. e016124-e016124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lancaster T, Stead LF. Individual behavioural counselling for smoking cessation. Cochrane Database Syst Rev. 2017;3 doi: 10.1002/14651858.CD001292.pub3. CD001292-CD001292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Kathuria H, Detterbeck FC, Fathi JT, et al. Stakeholder research priorities for smoking cessation interventions within lung cancer screening programs. an official American Thoracic Society research statement. Am J Respir Crit Care Med. 2017;196:1202–1212. doi: 10.1164/rccm.201709-1858ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: a joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38:843–844. doi: 10.5665/sleep.4716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Grandner MA. Sleep duration across the lifespan: implications for health. 2012;16:199-201. [DOI] [PMC free article] [PubMed]
- 84.Asarnow LD, Greer SM, Walker MP, Harvey AG. The impact of sleep improvement on food choices in adolescents with late bedtimes. J Adolesc Health. 2017;60:570–576. doi: 10.1016/j.jadohealth.2016.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Gildner TE, Liebert MA, Kowal P, Chatterji S, Snodgrass JJ. Associations between sleep duration, sleep quality, and cognitive test performance among older adults from six middle income countries: results from the Study on Global Ageing and Adult Health (SAGE) J Clin Sleep Med. 2014;10:613–621. doi: 10.5664/jcsm.3782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Besedovsky L, Lange T, Haack M. The sleep-immune crosstalk in health and disease. Physiol Rev. 2019;99:1325–1380. doi: 10.1152/physrev.00010.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Jahrami H, BaHammam AS, Bragazzi NL, Saif Z, Faris M, Vitiello MV. Sleep problems during the COVID-19 pandemic by population: a systematic review and meta-analysis. J Clin Sleep Med. 2021;17:299–313. doi: 10.5664/jcsm.8930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhang J, Xu D, Xie B, et al. Poor-sleep is associated with slow recovery from lymphopenia and an increased need for ICU care in hospitalized patients with COVID-19: A retrospective cohort study. Brain Behav Immun. 2020;88:50–58. doi: 10.1016/j.bbi.2020.05.075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Barone MTU, Ngongo B, Menna-Barreto L. Sleep-wake cycle impairment adding on the risk for COVID-19 severity in people with diabetes. Sleep Sci (Sao Paulo, Brazil) 2020;13:191–194. doi: 10.5935/1984-0063.20200038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Vitale JA, Perazzo P, Silingardi M, Biffi M, Banfi G, Negrini F. Is disruption of sleep quality a consequence of severe Covid-19 infection? A case-series examination. Chronobiol Int. 2020;37:1110–1114. doi: 10.1080/07420528.2020.1775241. [DOI] [PubMed] [Google Scholar]
- 91.Mazzucca S, Arredondo EM, Hoelscher DM, et al. Expanding implementation research to prevent chronic diseases in community settings. Annu Rev Public Health. 2021 doi: 10.1146/annurev-publhealth-090419-102547. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Friedman J, York H, Graetz N, et al. Measuring and forecasting progress towards the education-related SDG targets. Nature. 2020;580:636–639. doi: 10.1038/s41586-020-2198-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Tomaz SA, Jones RA, Hinkley T, et al. Physical activity in early childhood education and care settings in a low-income, rural South African community: an observational study. Rural Remote Health. 2019;19:5249. doi: 10.22605/RRH5249. [DOI] [PubMed] [Google Scholar]
- 94.Tonge KL, Jones RA, Hagenbuchner M, Nguyen TV, Okely AD. Educator engagement and interaction and children's physical activity in early childhood education and care settings: an observational study protocol. BMJ Open. 2017;7 doi: 10.1136/bmjopen-2016-014423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ward S, Bélanger M, Donovan D, Carrier N. Systematic review of the relationship beHween childcare educators' practices and preschoolers' physical activity and eating behaviours. Obes Rev. 2015;16:1055–1070. doi: 10.1111/obr.12315. [DOI] [PubMed] [Google Scholar]
- 96.Mikkelsen BE, Novotny R, Gittelsohn J. Multi-level, multi-component approaches to community based interventions for healthy living-a three case comparison. Int J Environ Res Public Health. 2016;13:1023. doi: 10.3390/ijerph13101023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Hills A, Gibson M, Brown T. In: Harnessing the Transformative Power of Education. Shelley B., Brown N., Crellin T., editors. Brill Publishers; 2019. Passport to better health and education outcomes for Tasmania's children.’ Innovations and Controversies: Interrogating Educational Change (ICIE), Vol. 9; pp. 197–209. [Google Scholar]
- 98.Campbell F, Conti G, Heckman JJ, et al. Early childhood investments substantially boost adult health. Science. 2014;343:1478–1485. doi: 10.1126/science.1248429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Medicine Io . 2015. Exploring Opportunities For Collaboration Between Health and Education to Improve Population Health: Workshop Summary. [PubMed] [Google Scholar]
- 100.Patton GC, Olsson CA, Skirbekk V, et al. Adolescence and the next generation. Nature. 2018;554:458–466. doi: 10.1038/nature25759. [DOI] [PubMed] [Google Scholar]
- 101.Daelmans B, Black MM, Lombardi J, et al. Effective interventions and strategies for improving early child development. BMJ. 2015;351:h4029. doi: 10.1136/bmj.h4029. [DOI] [PubMed] [Google Scholar]
- 102.Bailey R, Hillman C, Arent S, Petitpas A. Physical activity: an underestimated investment in human capital? J Phys Act Health. 2013;10:289–308. doi: 10.1123/jpah.10.3.289. [DOI] [PubMed] [Google Scholar]
- 103.World Health Organization . Implementation Plan: Executive Summary. 2017. Report of the commission on ending childhood obesity. [Google Scholar]
- 104.Black MM, Hurley KM. Investment in early childhood development. Lancet. 2014;384:1244–1245. doi: 10.1016/S0140-6736(14)60607-3. [DOI] [PubMed] [Google Scholar]
- 105.Developement OfEC-oa . 2006. Starting Strong II: Early Childhood Education and Care Policy. [Google Scholar]
- 106.Blake-Lamb TL, Locks LM, Perkins ME, Woo Baidal JA, Cheng ER, Taveras EM. Interventions for childhood obesity in the first 1,000 days a systematic review. Am J Prev Med. 2016;50:780–789. doi: 10.1016/j.amepre.2015.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Mihrshahi S, Baur LA. What exposures in early life are risk factors for childhood obesity? J Paediatr Child Health. 2018;54:1294–1298. doi: 10.1111/jpc.14195. [DOI] [PubMed] [Google Scholar]
- 108.Lubans DR, Morgan PJ, Aguiar EJ, Callister R. Randomized controlled trial of the Physical Activity Leaders (PALs) program for adolescent boys from disadvantaged secondary schools. Prev Med. 2011;52:239–246. doi: 10.1016/j.ypmed.2011.01.009. [DOI] [PubMed] [Google Scholar]
- 109.Donnelly JE, Greene JL, Gibson CA, et al. Physical activity and academic achievement across the curriculum (A + PAAC): rationale and design of a 3-year, cluster-randomized trial. BMC Public Health. 2013;13:307. doi: 10.1186/1471-2458-13-307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Hernández-Cordero S, Cuevas-Nasu L, Morales-Ruán MC, Humarán IM, Ávila-Arcos MA, Rivera-Dommarco JA. Overweight and obesity in Mexican children and adolescents during the last 25 years. Nutr Diabetes. 2017;7:e280. doi: 10.1038/nutd.2017.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Shonkoff JP, Boyce WT, McEwen BS. Neuroscience, molecular biology, and the childhood roots of health disparities: building a new framework for health promotion and disease prevention. JAMA. 2009;301:2252–2259. doi: 10.1001/jama.2009.754. [DOI] [PubMed] [Google Scholar]
- 112.Street SJ, Wells JC, Hills AP. Windows of opportunity for physical activity in the prevention of obesity. Obes Rev. 2015;16:857–870. doi: 10.1111/obr.12306. [DOI] [PubMed] [Google Scholar]
- 113.Morgan PJ, Young MD, Smith JJ, Lubans DR. Targeted health behavior interventions promoting physical activity: a conceptual model. Exerc Sport Sci Rev. 2016;44:71–80. doi: 10.1249/JES.0000000000000075. [DOI] [PubMed] [Google Scholar]
- 114.Todd AS, Street SJ, Ziviani J, Byrne NM, Hills AP. Overweight and obese adolescent girls: the importance of promoting sensible eating and activity behaviors from the start of the adolescent period. Int J Environ Res Public Health. 2015;12:2306–2329. doi: 10.3390/ijerph120202306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Bay JL, Vickers MH, Mora HA, Sloboda DM, Morton SM. Adolescents as agents of healthful change through scientific literacy development: A school-university partnership program in New Zealand. Int J STEM Educ. 2017;4:15. doi: 10.1186/s40594-017-0077-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Institute of Medicine and National Research Council of The National Academies . 2015. Transforming the Workforce for Children Birth Through Age 8: A Unifying Foundation. [PubMed] [Google Scholar]
- 117.Toussaint N, Streppel MT, Mul S, et al. A preschool-based intervention for Early Childhood Education and Care (ECEC) teachers in promoting healthy eating and physical activity in toddlers: study protocol of the cluster randomized controlled trial PreSchool@HealthyWeight. BMC Public Health. 2019;19:278. doi: 10.1186/s12889-019-6611-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Bryan CL, Sims SK, Dunaway DL, Hester DJ. Become a champion for healthy, active schools. Strategies. 2019;32:24–31. [Google Scholar]
- 119.Lee JG, Alpous A, Yusuf W, et al. 2018. Y Kids Academy Program Increases Knowledge of Healthy Living in Young Adolescents. [Google Scholar]
- 120.Tecnologico de Monterrey . 2021. Mondel Tec21.https://tec.mx/en/model-tec21 Available at: Accessed October 6, 2020. [Google Scholar]
- 121.Strieter L, Laddu DR, Sainsbury J, Arena R. The importance of school-based healthy living initiatives: introducing the health and wellness academy concept. Prog Cardiovasc Dis. 2019;62:68–73. doi: 10.1016/j.pcad.2018.08.005. [DOI] [PubMed] [Google Scholar]
- 122.Umstattd Meyer MR, Bridges Hamilton CN, Prochnow T, et al. Come together, play, be active: physical activity engagement of school-age children at Play Streets in four diverse rural communities in the U.S. Prev Med. 2019;129 doi: 10.1016/j.ypmed.2019.105869. [DOI] [PubMed] [Google Scholar]
- 123.De Vogli R. Status syndrome: how your social standing directly affects your health and life expectancy. BMJ. 2004;329:408. [Google Scholar]
- 124.Hivert MF, Arena R, Forman DE, et al. Medical training to achieve competency in lifestyle counseling: an essential foundation for prevention and treatment of cardiovascular diseases and other chronic medical conditions: a scientific statement from the American Heart Association. Circulation. 2016;134:e308–e327. doi: 10.1161/CIR.0000000000000442. [DOI] [PubMed] [Google Scholar]
- 125.Hivert MF, McNeil A, Lavie CJ, Arena R. Training health professionals to deliver healthy living medicine. Prog Cardiovasc Dis. 2017;59:471–478. doi: 10.1016/j.pcad.2017.02.004. [DOI] [PubMed] [Google Scholar]
- 126.Thompson WR, Sallis R, Joy E, Jaworski CA, Stuhr RM, Trilk JL. Exercise is medicine. Am J Lifestyle Med. 2020;14:511–523. doi: 10.1177/1559827620912192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.LAW AND POLICY AS DETERMINANTS OF HEALTH AND WELL-BEING. Issue briefs to inform development and implementation of healthy people 2030. 2018:57-64.
- 128.Bhatnagar A, Whitsel LP, Blaha MJ, et al. New and emerging tobacco products and the nicotine endgame: the role of robust regulation and comprehensive tobacco control and prevention: a presidential advisory from the American Heart Association. Circulation. 2019;139:e937–e958. doi: 10.1161/CIR.0000000000000669. [DOI] [PubMed] [Google Scholar]
- 129.Golden SD, Smith MH, Feighery EC, Roeseler A, Rogers T, Ribisl KM. Beyond excise taxes: a systematic review of literature on non-tax policy approaches to raising tobacco product prices. Tob Control. 2016;25:377–385. doi: 10.1136/tobaccocontrol-2015-052294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Wang Y, Cai L, Wu Y, et al. What childhood obesity prevention programmes work? A systematic review and meta-analysis. Obes Rev. 2015;16:547–565. doi: 10.1111/obr.12277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Sacks G, Swinburn B, Lawrence M. Obesity policy action framework and analysis grids for a comprehensive policy approach to reducing obesity. Obes Rev. 2009;10:76–86. doi: 10.1111/j.1467-789X.2008.00524.x. [DOI] [PubMed] [Google Scholar]
- 132.Mechanick JI, Youdim A, Jones DB, et al. American Association of Clinical E, Obesity S, American Society for M and Bariatric S. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient–2013 update: cosponsored by American Association of Clinical Endocrinologists, The Obesity Society, and American Society for Metabolic & Bariatric Surgery. Obesity (Silver Spring) 2013;21:S1–27. doi: 10.1002/oby.20461. Suppl 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Curry SJ, Krist AH, Owens DK, et al. Behavioral weight loss interventions to prevent obesity-related morbidity and mortality in adults: us preventive services task force recommendation statement. JAMA. 2018;320:1163–1171. doi: 10.1001/jama.2018.13022. [DOI] [PubMed] [Google Scholar]
- 134.LeBlanc ES, Patnode CD, Webber EM, Redmond N, Rushkin M, O'Connor EA. Behavioral and pharmacotherapy weight loss interventions to prevent obesity-related morbidity and mortality in adults: updated evidence report and systematic review for the US preventive services task force. JAMA. 2018;320:1172–1191. doi: 10.1001/jama.2018.7777. [DOI] [PubMed] [Google Scholar]
- 135.Phelan SM, Burgess DJ, Yeazel MW, Hellerstedt WL, Griffin JM, van Ryn M. Impact of weight bias and stigma on quality of care and outcomes for patients with obesity. Obes Rev. 2015;16:319–326. doi: 10.1111/obr.12266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Alberga AS, Edache IY, Forhan M, Russell-Mayhew S. Weight bias and health care utilization: a scoping review. Prim Health Care Res Dev. 2019;20:e116. doi: 10.1017/S1463423619000227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Gable L. The patient protection and affordable care act, public health, and the elusive target of human rights. J Law Med Ethics. 2011;39:340–354. doi: 10.1111/j.1748-720X.2011.00604.x. [DOI] [PubMed] [Google Scholar]
- 138.Marincic P, Gray C. CMS coverage for obesity management. JAAPA. 2014;27:47–50. doi: 10.1097/01.JAA.0000438539.99762.50. [DOI] [PubMed] [Google Scholar]
- 139.World Health Organization. Global strategy on diet, Physical Activity and Health. Accessed 15 September, 2021.
- 140.Gorski MT, Roberto CA. Public health policies to encourage healthy eating habits: recent perspectives. J Healthc Leadersh. 2015;7:81090. doi: 10.2147/JHL.S69188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.WHO . 2015. Using Price Policies to Promote Healthier Diets. [Google Scholar]
- 142.Basu S, Madsen K. Effectiveness and equity of sugar-sweetened beverage taxation. PLos Med. 2017;14 doi: 10.1371/journal.pmed.1002327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Lee Y, Mozaffarian D, Sy S, et al. Health impact and cost-effectiveness of volume, tiered, and absolute sugar content sugar-sweetened beverage tax policies in the united states: a microsimulation study. Circulation. 2020;142:523–534. doi: 10.1161/CIRCULATIONAHA.119.042956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Teng AM, Jones AC, Mizdrak A, Signal L, Genç M, Wilson N. Impact of sugar-sweetened beverage taxes on purchases and dietary intake: Systematic review and meta-analysis. Obes Rev. 2019;20:1187–1204. doi: 10.1111/obr.12868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Afshin A, Peñalvo JL, Del Gobbo L, et al. The prospective impact of food pricing on improving dietary consumption: A systematic review and meta-analysis. PLoS One. 2017;12 doi: 10.1371/journal.pone.0172277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Brandt EJ, Myerson R, Perraillon MC, Polonsky TS. Hospital admissions for myocardial infarction and stroke before and after the trans-fatty acid restrictions in New York. JAMA Cardiol. 2017;2:627. doi: 10.1001/jamacardio.2017.0491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.WHO . 2004. Marketing Food to Children: The Global Regulatory Environment. [Google Scholar]
- 148.U.S . Physical Activity Guidelines for Americans. 2018. Department of health and human services. [Google Scholar]
- 149.Rütten A, Abu-Omar K, Gelius P, Schow D. Physical inactivity as a policy problem: applying a concept from policy analysis to a public health issue. Health Res Policy Syst. 2013;11:9. doi: 10.1186/1478-4505-11-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Klepac Pogrmilovic B, O'Sullivan G, Milton K, et al. The development of the comprehensive analysis of policy on physical activity (CAPPA) framework. Int J Behav Nutr Phys Act. 2019;16:60. doi: 10.1186/s12966-019-0822-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Barroso CS, Kelder SH, Springer AE, et al. Senate Bill 42: implementation and impact on physical activity in middle schools. J Adolesc Health. 2009;45:S82–S90. doi: 10.1016/j.jadohealth.2009.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Chriqui JF, Eyler A, Carnoske C, Slater S. State and district policy influences on district-wide elementary and middle school physical education practices. J Public Health Manag Pract. 2013;19:S41–S48. doi: 10.1097/PHH.0b013e31828a8bce. [DOI] [PubMed] [Google Scholar]
- 153.Sawchuk CN, Russo J, Ferguson G, et al. Health promotion programs and policies in the workplace: an exploratory study with Alaska businesses. Prev Chronic Dis. 2020;17:E125. doi: 10.5888/pcd17.200111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Mulder L, Belay B, Mukhtar Q, Lang JE, Harris D, Onufrak S. Prevalence of workplace health practices and policies in hospitals: results from the workplace health in America study. Am J Health Promot. 2020;34:867–875. doi: 10.1177/0890117120905232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Young DR, Cradock AL, Eyler AA, et al. Creating built environments that expand active transportation and active living across the United States: a policy statement from the American Heart Association. Circulation. 2020;142:e167–e183. doi: 10.1161/CIR.0000000000000878. [DOI] [PubMed] [Google Scholar]
- 156.Sallis JF, Bull F, Burdett R, et al. Use of science to guide city planning policy and practice: how to achieve healthy and sustainable future cities. Lancet North Am Ed. 2016;388:2936–2947. doi: 10.1016/S0140-6736(16)30068-X. [DOI] [PubMed] [Google Scholar]
- 157.Giles-Corti B, Vernez-Moudon A, Reis R, et al. City planning and population health: a global challenge. Lancet North Am Ed. 2016;388:2912–2924. doi: 10.1016/S0140-6736(16)30066-6. [DOI] [PubMed] [Google Scholar]
- 158.Omura JD, Carlson SA, Brown DR, et al. Built environment approaches to increase physical activity: a science advisory from the American Heart Association. Circulation. 2020;142:e160–e166. doi: 10.1161/CIR.0000000000000884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Bull FC, Al-Ansari SS, Biddle S, et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br J Sports Med. 2020;54:1451–1462. doi: 10.1136/bjsports-2020-102955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Howie EK, Stevick ED. The “Ins” and “Outs” of physical activity policy implementation: inadequate capacity, inappropriate outcome measures, and insufficient funds. J Sch Health. 2014;84:581–585. doi: 10.1111/josh.12182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Arnason E. 2014. A Woman of the Iron People: Open Road Media. [Google Scholar]
- 162.Huys N, Van Stappen V, Shadid S, et al. Effectiveness of a family-, school- and community-based intervention on physical activity and its correlates in Belgian families with an increased risk for type 2 diabetes mellitus: the Feel4Diabetes-study. BMC Public Health. 2020;20:1231. doi: 10.1186/s12889-020-09336-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Lambrinou C-P, Androutsos O, Karaglani E, et al. Effective strategies for childhood obesity prevention via school based, family involved interventions: a critical review for the development of the Feel4Diabetes-study school based component. BMC Endocr Disord. 2020;20:52. doi: 10.1186/s12902-020-0526-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Martin A, Booth JN, Laird Y, Sproule J, Reilly JJ, Saunders DH. Physical activity, diet and other behavioural interventions for improving cognition and school achievement in children and adolescents with obesity or overweight. Cochrane Database Syst Rev. 2018;3:CD009728. doi: 10.1002/14651858.CD009728.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Luque JS, Ross L, Gwede CK. Qualitative systematic review of barber-administered health education, promotion, screening and outreach programs in African-American communities. J Community Health. 2014;39:181–190. doi: 10.1007/s10900-013-9744-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Lin E, Balogh R, Chung H, et al. Looking across health and healthcare outcomes for people with intellectual and developmental disabilities and psychiatric disorders: population-based longitudinal study. Br J Psychiatry. 2021;218:51–57. doi: 10.1192/bjp.2020.202. [DOI] [PubMed] [Google Scholar]
- 167.Jankovic J, Parsons J, Jovanović N, et al. Differences in access and utilisation of mental health services in the perinatal period for women from ethnic minorities-a population-based study. BMC Med. 2020;18:245. doi: 10.1186/s12916-020-01711-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Peterman JE, Loy S, Carlos J, Arena R, Kaminsky LA. Increasing physical activity in the community setting. Prog Cardiovasc Dis. 2021;64:27–32. doi: 10.1016/j.pcad.2020.10.008. [DOI] [PubMed] [Google Scholar]
- 169.Arena R, Arnett DK, Terry PE, et al. The role of worksite health screening: a policy statement from the American Heart Association. Circulation. 2014;130:719–734. doi: 10.1161/CIR.0000000000000079. [DOI] [PubMed] [Google Scholar]
- 170.Arena R, Guazzi M, Briggs PD, et al. Promoting health and wellness in the workplace: a unique opportunity to establish primary and extended secondary cardiovascular risk reduction programs. Mayo Clin Proc. 2013;88:605–617. doi: 10.1016/j.mayocp.2013.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Goetzel RZ, Henke RM, Head MA, Benevent R, Calitz C. Workplace programs, policies, and environmental supports to prevent cardiovascular disease. Health Aff (Millwood) 2017;36:229–236. doi: 10.1377/hlthaff.2016.1273. [DOI] [PubMed] [Google Scholar]
- 172.Idris IB, Azit NA, Abdul Ghani SR, Syed Nor SF, Nawi M. Industrial health; 2021. A systematic review on noncommunicable diseases among working women. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Whitsel LP, Arena R, Kaminsky LA, et al. Assessing physical activity, sedentary behavior, and cardiorespiratory fitness in worksite health promotion. Am J Health Promot. 2019;33:318–326. doi: 10.1177/0890117118816750e. [DOI] [PubMed] [Google Scholar]
- 174.Cahalin LP, Kaminsky L, Lavie CJ, et al. Development and implementation of worksite health and wellness programs: a focus on non-communicable disease. Prog Cardiovasc Dis. 2015;58:94–101. doi: 10.1016/j.pcad.2015.04.001. [DOI] [PubMed] [Google Scholar]
- 175.Pronk NP. Implementing movement at the workplace: approaches to increase physical activity and reduce sedentary behavior in the context of work. Prog Cardiovasc Dis. 2021;64:17–21. doi: 10.1016/j.pcad.2020.10.004. [DOI] [PubMed] [Google Scholar]
- 176.Pronk N, Arena R, Bond S. Work, health, and COVID-19: the role of healthy living in pandemic preparedness for Business. ACSM Health Fit J. 2021;25:55–60. [Google Scholar]
- 177.Calitz C, Pham K. American Heart Association's Workplace Health Achievement Index. Am J Health Promot. 2020;34:317–318. doi: 10.1177/0890117119898026b. [DOI] [PubMed] [Google Scholar]
- 178.Whitfield GP, Carlson SA, Ussery EN, Fulton JE, Galuska DA, Petersen R. Trends in meeting physical activity guidelines among urban and rural dwelling adults - United States, 2008-2017. MMWR Morb Mortal Wkly Rep. 2019;68:513–518. doi: 10.15585/mmwr.mm6823a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Barnes PM, Schoenborn CA. Trends in adults receiving a recommendation for exercise or other physical activity from a physician or other health professional. NCHS Data Brief. 2012;86:1–8. [PubMed] [Google Scholar]
- 180.Lewis BS, Lynch WD. The effect of physician advice on exercise behavior. Prev Med. 1993;22:110–121. doi: 10.1006/pmed.1993.1008. [DOI] [PubMed] [Google Scholar]
- 181.Lobelo F, de Quevedo IG. The evidence in support of physicians and health care providers as physical activity role models. Am J Lifestyle Med. 2016;10:36–52. doi: 10.1177/1559827613520120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Pears S, Bijker M, Morton K, et al. A randomised controlled trial of three very brief interventions for physical activity in primary care. BMC Public Health. 2016;16:1033. doi: 10.1186/s12889-016-3684-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.NICE. Physical activity: brief advice for adults in primary care. Available at: https://www.nice.org.uk/guidance/ph44/chapter/1-Recommendations. Accessed November 2, 2021.
- 184.Strath SJ, Kaminsky LA, Ainsworth BE, et al. Guide to the assessment of physical activity: clinical and research applications: a scientific statement from the American Heart Association. Circulation. 2013;128:2259–2279. doi: 10.1161/01.cir.0000435708.67487.da. [DOI] [PubMed] [Google Scholar]
- 185.Hart L, Horton R. Syndemics: committing to a healthier future. Lancet. 2017;389:888–889. doi: 10.1016/S0140-6736(17)30599-8. [DOI] [PubMed] [Google Scholar]
- 186.Singer M, Clair S. Syndemics and public health: reconceptualizing disease in bio-social context. Med Anthropol Q. 2003;17:423–441. doi: 10.1525/maq.2003.17.4.423. [DOI] [PubMed] [Google Scholar]
- 187.Laddu DR, Lavie CJ, Phillips SA, Arena R. Physical activity for immunity protection: Inoculating populations with healthy living medicine in preparation for the next pandemic. Prog Cardiovasc Dis. 2021;64:102–104. doi: 10.1016/j.pcad.2020.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Cardinal BJ. Toward a greater understanding of the syndemic nature of hypokinetic diseases. Journal of exercise science and fitness. 2016;14:54–59. doi: 10.1016/j.jesf.2016.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Rentsch CT, Kidwai-Khan F, Tate JP, et al. medRxiv: The Preprint Server for Health Sciences. 2020. Covid-19 by race and ethnicity: a national cohort study of 6 million united states veterans. [Google Scholar]
- 190.El-Mohandes A, Ratzan SC, Rauh L, et al. COVID-19: a barometer for social justice in New York city. Am J Public Health. 2020;110:1656–1658. doi: 10.2105/AJPH.2020.305939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Krishnaswami J, Sardana J, Daxini A. Community-engaged lifestyle medicine as a framework for health equity: principles for lifestyle medicine in low-resource settings. Am J Lifestyle Med. 2019;13:443–450. doi: 10.1177/1559827619838469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Bantham A, Taverno Ross SE, Sebastião E, Hall G. Overcoming barriers to physical activity in underserved populations. Prog Cardiovasc Dis. 2021;64:64–71. doi: 10.1016/j.pcad.2020.11.002. [DOI] [PubMed] [Google Scholar]
- 193.Lee IJ, Ahmed NU. The devastating cost of racial and ethnic health inequity in the COVID-19 pandemic. J Natl Med Assoc. 2021;113:114–117. doi: 10.1016/j.jnma.2020.11.015. [DOI] [PubMed] [Google Scholar]
- 194.Leandro CG, Ferreira E, Silva WT, Lima-Silva AE. Covid-19 and exercise-induced immunomodulation. NeuroImmunoModulation. 2020;27:75–78. doi: 10.1159/000508951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Walker TA, Waite B, Thompson MG, et al. Risk of severe influenza among adults with chronic medical conditions. J Infect Dis. 2020;221:183–190. doi: 10.1093/infdis/jiz570. [DOI] [PubMed] [Google Scholar]
- 196.Kohut ML, Cooper MM, Nickolaus MS, Russell DR, Cunnick JE. Exercise and psychosocial factors modulate immunity to influenza vaccine in elderly individuals. J Gerontol A Biol Sci Med Sci. 2002;57:M557–M562. doi: 10.1093/gerona/57.9.m557. [DOI] [PubMed] [Google Scholar]
- 197.Fernandez-Jimenez R, Jaslow R, Bansilal S, et al. Different lifestyle interventions in adults from underserved communities: the FAMILIA trial. J Am Coll Cardiol. 2020;75:42–56. doi: 10.1016/j.jacc.2019.10.021. [DOI] [PubMed] [Google Scholar]
- 198.Sebastiao E, Chodzko-Zajko W, Schwingel A. The need to modify physical activity messages to better speak to older African American women: a pilot study. BMC Public Health. 2015;15:962. doi: 10.1186/s12889-015-2317-x. [DOI] [PMC free article] [PubMed] [Google Scholar]