Summary
Humans harbour large quantities of microbes, including bacteria, fungi, viruses and archaea, in the gut. Patients with liver disease exhibit changes in the intestinal microbiota and gut barrier dysfunction. Preclinical models demonstrate the importance of the gut microbiota in the pathogenesis of various liver diseases. In this review, we discuss how manipulation of the gut microbiota can be used as a novel treatment approach for liver disease. We summarise current data on untargeted approaches, including probiotics and faecal microbiota transplantation, and precision microbiome-centred therapies, including engineered bacteria, postbiotics and phages, for the treatment of liver diseases.
Keywords: mycobiome, fecal microbiota transplantation, cirrhosis, gut-liver-axis
Introduction
The gastrointestinal microbiome is extraordinarily complex, containing organisms that span several kingdoms. The composition and functions of the gut microbiota are affected by several intrinsic and extrinsic factors such as host genetics, disease state, immune health, diet, socio-economic status, location, and medications [1]. These factors have the potential to impact liver health directly or indirectly since most liver diseases lie at the crossroads of addiction, psychosocial and medical disorders [2, 3]. Therefore, it is important to contextualise human microbial data within these constraints.
Most research to date has been focused on bacteria given that they are the easiest to culture or characterise and are closely linked with several infectious and chronic diseases. There is a gradient in the number of bacteria present along the gastrointestinal tract, with the highest number being in the colon and stool. Firmicutes, Bacteroidetes and Proteobacteria constitute the major bacterial phyla in the gastrointestinal tract, the relative balance of which contribute towards the development of enterotypes [4]. The presence, function and interaction of bacteria with the host, diet, and other kingdoms present in the gut can influence their impact on disease progression in chronic liver disease and cirrhosis [5, 6]. The presence of specific microbial taxa such as Lachnospiraceae and Ruminococcaceae members have been consistently associated with relative health while some members of Enterobacteriaceae are associated with poor outcomes in liver disease [7, 8]. However, given the redundancies across pathways, the functions and interactions of the microbiota, rather than their mere presence, are more likely the major determinants of their impact on the human host. Microbial therapies targeting bacteria using relatively blunt instruments, such as antibiotics and faecal microbiota transplantation (FMT), are already standard of care. However, a nuanced and targeted approach may be necessary, which will require knowledge of the other kingdoms (fungi, viruses, and archaea) that co-exist and affect bacterial populations.
Although, compared to bacteria, there are relatively far fewer fungi, they can have a major impact on bacterial populations and humans through direct competition, commensalism, or production of metabolites. The major fungal phyla are Ascomycetes, Basidiomycetes and Zygomycetes. Fungi, especially Candida spp, have been evaluated as potential contributors to poor outcomes in pre-cirrhotic and cirrhotic liver disease [9, 10].
There is an increasing recognition of the “dark matter” or the currently unidentified components of the gut microbiota [11]. This is most relevant for the virome, which is the assemblage of viruses that is often described using metagenomic sequencing of viral nucleic acids. Viruses are the most abundant biological entities with large heterogeneity in the environment and within the human host [12]. While there has been an exponential rise in the number of viral sequences available, there is still scope for improvement in recognition of these viral-like particles [13]. The virome consists of eukaryotic viruses and bacteriophages (phages) and other viruses that have manifold effects on hosts directly or through their impact on bacteria [14]. Several clinical and translational analyses are beginning to shed light on these trans-kingdom interactions in liver disease [15, 16]. Archaea are a relatively rare but metabolically active component of the microbiome [17]. They consist of few taxa that were initially thought to be methanogens, but their metabolic function might be beyond the production of methane.
In addition to analysis of the presence, absence or relative abundance of microbial species, functional analysis is an important concept in microbial research. Functional changes are essential to modulate the impact of gut microbiota on outcomes and generate therapeutic responses. Exciting new microbiome-based approaches are being developed, which have the potential to favourably impact all aspects of liver disease. The basis for microbiome-centred therapies for liver disease is the intimate relationship between the gut and the liver. Almost all venous blood from the small and large intestine reaches the liver as the first organ in our body via the portal vein. The liver entertains a bidirectional communication via secretion of bile and its constituents, such as bile acids, into the small intestine. Thus, the gut microbiota is known to modulate the severity of different liver diseases. Preclinical models have also demonstrated the transmissibility of liver disease by FMT[18].
Traditional therapies for cirrhosis include absorbable and non-absorbable antibiotics. The major indication for absorbable antibiotics beyond the traditional treatment of infection is prophylaxis for spontaneous bacterial peritonitis (with predominantly gram-negative organism coverage [19]). Other aspects of antibiotic use are for hepatic encephalopathy (HE) treatment and prevention of recurrence. Antibiotics that have been studied for HE treatment include vancomycin, neomycin, metronidazole and rifaximin [20, 21]. Of these rifaximin is the only one that is widely used due to its efficacy, favourable adverse event profile and tolerability compared to the others listed [21, 22]. The mechanism of action of rifaximin is also interesting and could go beyond a traditional “antibiotic” [23]. However, there remain questions regarding the specificity of antibiotic use in an era when the frequency of gram-positive infections, fungal infections and antibiotic resistance is increasing [5, 24–27].
In this review, we summarise data on untargeted approaches, including probiotics and FMT, and precision microbiome-centred therapies, including engineered bacteria, postbiotics and phages, for the treatment of liver diseases (Fig. 1).
Fig. 1. Microbiome-centred therapies for patients with liver disease.
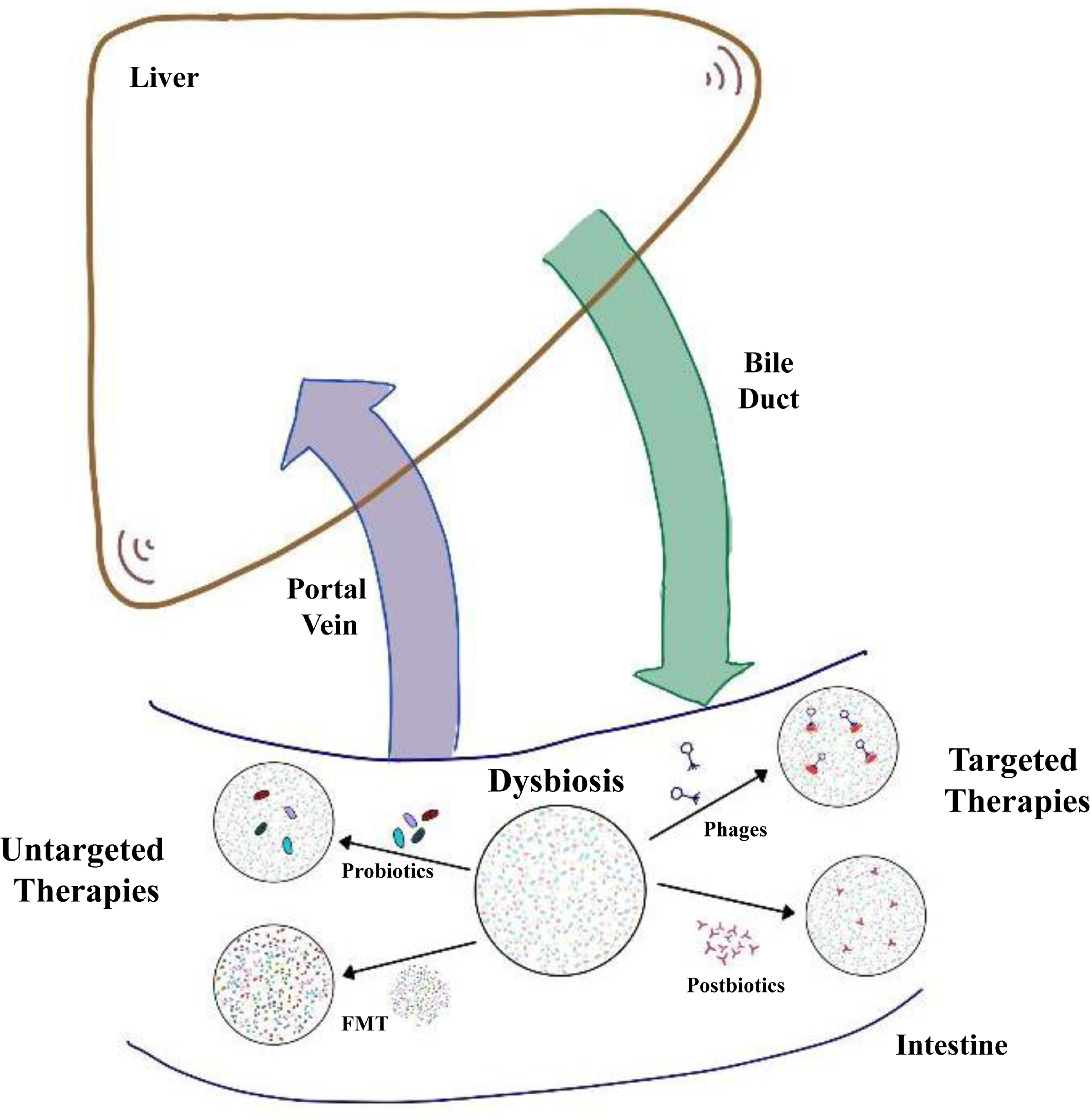
Patients with liver disease show quantitative and compositional changes in the intestinal microbiota, also called dysbiosis. Intestinal homeostasis can be restored by untargeted therapies, which include oral administration of probiotics or FMT. The aim of FMT is to replace the entire dysbiotic microbiota using stool from a healthy donor. Targeted therapies include phages and bacterial derived metabolites, also called postbiotics. Selective targeting of bacterial strains by phage therapy can modify liver disease progression in preclinical models. FMT, faecal microbiota transplant.
Probiotics and engineered bacteria
Probiotics are live bacteria that promote health in the host and are considered safe. Although the beneficial effect of traditional probiotics, such as Lactobacillus and Bifidobacterium, have been well documented for decades, mechanisms of action have only recently begun to be elucidated. For example, Lactobacillus salivarius Li01 reduced acute toxin-induced liver injury and hyperammonaemia by stabilising the gut barrier and restoring bacterial eubiosis in the intestine of mice [28, 29]. Increased intestinal aryl hydrocarbon receptor activation by probiotics can increase intestinal expression of antimicrobial molecules, which contributes to gut barrier protection in preclinical models [30]. Probiotics can also reduce hepatic bile acid synthesis by enhancing the negative intestinal feedback mediated by fibroblast growth factor 15 [31].
Several randomised, placebo-controlled clinical trials showed benefits of probiotics in various liver diseases [32, 33]. Selection criteria for a Cochrane analysis (21 trials) were randomised clinical trials that compared probiotics in any dosage with placebo or no intervention, or with any other treatment in patients with cirrhosis of any aetiology and HE [34]. Probiotics improved recovery and may lead to improvements in the development of overt HE, quality of life, and plasma ammonia concentrations, but no difference in mortality was observed [34]. The benefit of probiotics compared with lactulose was less evident [34]. Two meta analyses of 28 clinical trials [35] and 22 randomised-controlled trials [36] included patients with non-alcoholic fatty liver disease (NAFLD). Selection criteria included randomised and controlled clinical trials testing the effect of probiotics in the treatment of patients with NAFLD diagnosed on the basis of radiological/histological evidence of fatty liver and in the absence of alcohol abuse [35, 36]. Probiotics lowered body mass index, liver enzyme levels and inflammation, and improved diabetes and dyslipidaemia [35, 36].
A common shortcoming is that different clinical trials have used a variety of probiotics, sometimes in combination with other agents, and the duration of treatment can range from a few days to years. Few clinical trials have been reproduced with the same probiotic regimen.
Next-generation probiotics have been engineered to further enhance their beneficial effects. Whole-genome sequencing and molecular characterisation enable the selection of health-promoting genes that are associated with improved colonisation in the gut, host-bacteria interaction, immunomodulation, antimicrobial activity or pathogen control [37]. Early generation of engineered bacteria focused on the production of the anti-inflammatory cytokine interleukin (IL)-10 (Fig. 2A), which was beneficial in models of preclinical colitis [38]. IL-22 is a member of the IL-10 cytokine family that exerts beneficial effects on epithelial cells such as hepatocytes and gut enterocytes [39]. Engineered Lactobacillus reuteri-secreting mouse IL-22 were administered in a mouse model of ethanol-induced liver disease with a dose and frequency that did not increase systemic levels of IL-22. IL-22 restricted to the intestinal environment stimulated expression of antimicrobial molecules, such as Reg3g (regenerating islet-derived 3-gamma) in intestinal epithelial cells, which prevented translocation of bacteria from the intestinal lumen to the liver and reduced ethanol-induced liver injury, steatosis and inflammation [40]. This exemplifies the beneficial effect of IL-22-secreting L. reuteri on increasing antimicrobial activity and improving host-bacteria interactions in preclinical models (Fig. 2A). Bacterial delivery of the peptide hormone Glp1 (glucagon-like peptide 1) to the gut can induce insulin production in intestinal epithelial cells and increase systemic insulin levels [41] (Fig. 2A).
Fig. 2. Engineered bacteria.
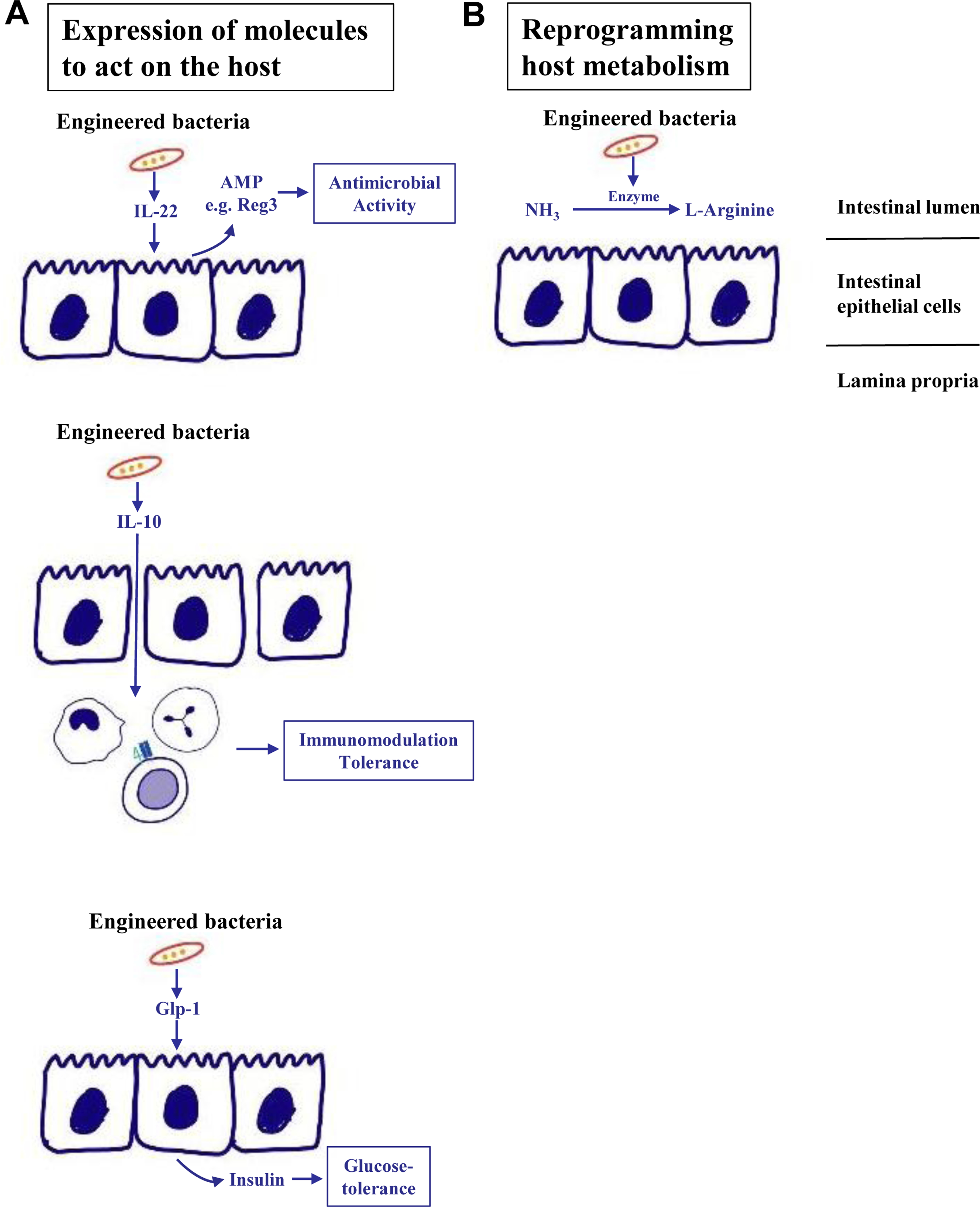
(A) Synthetic bacteria can express molecules that exert beneficial effects for the host. Examples include secretion of IL-22, which can induce AMP secretion such as Reg3 from intestinal epithelial cells to increase luminal antimicrobial activity, or IL-10, which exerts immunomodulatory and tolerogenic properties on immune cells in the lamina propria. Bacterial delivery of Glp1 induces secretion of insulin in intestinal epithelial cells to the systemic circulation and improves glucose tolerance in preclinical models. (B) In addition, engineered bacteria can metabolise toxic metabolites such as ammonia, which is converted into L-arginine. AMP, antimicrobial protein; Glp1, glucagon-like peptide 1; IL, interleukin; Reg3, regenerating islet-derived 3.
Engineered bacteria are used to consume toxic metabolites in metabolic diseases. Ammonia is produced by gut bacteria, reaches the liver via the portal vein and is metabolised into urea. Systemic ammonia levels increase in patients with cirrhosis and are associated with HE. Probiotic Escherichia coli (E. coli) Nissle 1917 were engineered to metabolise ammonia and convert it to L-arginine in the gut [42]. Oral administration showed ammonia-lowering effects and improved survival in preclinical models of hyperammonaemia [42] (Fig. 2B). However, a recent randomised, double-blind, placebo-controlled phase Ib/IIa study was not successful. Oral SYNB1020 treatment did not lower blood ammonia or change other exploratory endpoints relative to placebo in patients with cirrhosis and elevated blood ammonia, despite evidence of the synthetic strain being active and well tolerated (Synlogic website https://www.synlogictx.com).
Overall, synthetic bacteria are promising microbes for microbiota-centred therapies. They can be utilised as precision medicine tools to restore intestinal and/or systemic homeostasis. Although preclinical models show benefit, the effect of these microbes in clinical trials has not been confirmed. Several issues need to be further addressed such as pretreatment, dose, frequency and duration of therapy. Although a stable colonisation of synthetic bacteria is desirable for host interaction, a continued and long-term colonisation might not be needed or even desirable. In addition, heterogeneity of the microbiota in individual patients with chronic liver disease requires a personalised treatment approach. Screening of patients prior to initiating therapy might guide the use of engineered bacteria.
Postbiotics
Microbially generated metabolites influence microbiota-host, trans-kingdom, and inter-bacterial interactions. These metabolites can be harnessed as therapeutic and or prognostic factors in liver disease. There remains controversy as to which metabolites are specifically microbially derived, host-generated or both[43]. Microbially generated metabolites that are of particular interest are i) bile acids, ii) tri-methylamine oxide (TMAO), iii) tryptophan derivatives and iv) short-chain fatty acids (SCFAs) (Table 1). A deeper overview of the role of metabolites and gut microbiota is published elsewhere[43, 44], especially focused on NAFLD and non-alcoholic steatohepatitis (NASH) [45].
Table 1.
Examples of postbiotics.
| Microbial-metabolite pathways | Overview of bacterial transformation | Examples of clinical and translational investigations in liver disease |
|---|---|---|
| Bile acids | • Deconjugation: bile salt hydrolases • Secondary bile acid formation: 7α dehydroxylation • Epimerisation • Desulphation |
• Significant changes during alcohol-associated liver disease progression and alcohol cessation • Changes with use of FXR modulators, FMT or rifaximin |
| Choline compounds (TMAO) | • Degraded by several taxa to TMA then oxidised in the liver to TMAO | • Associated with metabolic syndrome • Linked with minimal hepatic encephalopathy • Reduced pre-transplant but rebounds post-liver transplant |
| Indole derivatives | • Balance between human and microbial tryptophan degradation • Specific taxa are unclear • Metabolites: tryptamine, indole-3-pyruvate, indole, indole-3-acetamide, indole-3-acetaldehyde, indole-3-aldehyde, indole-3-acetic acid, indole-3-lactic acid, indole acrylic acid and indole-3-propionic acid • Indole sulfation in the liver |
• Preclinical evidence of strengthening gut barrier with selected metabolites • High production of oxindole in patients with hepatic encephalopathy • Associated with mood changes that co-exist with liver disease • Higher in those who developed negative outcomes in cirrhosis in outpatient and inpatient setting |
| Short-chain fatty acids | • Fermentation of dietary fibre and colonic mucus | • Varying impact on obesity and high-fat diet response • Butyrate levels reduce with advancing liver disease and alcohol-associated liver disease • Varies with lactulose administration • Restored/enhanced after FMT |
FMT, faecal microbiota transplant; FXR, farnesoid X receptor; TMAO, tri-methylamine oxide.
While microbial metabolites were previously included in the broad spectrum of “postbiotics”, this has recently been changed by the International Scientific Association of Probiotics and Prebiotics [46]. The current definition of a postbiotic is “preparation of inanimate microorganisms and/or their components that confers a health benefit on the host”. This includes purposely inactivated microbial cells with/without metabolites or cell components that contribute to health benefits and does not include purified microbial metabolites or vaccines. Examples of postbiotics used in clinical trials in liver disease are bile acid modulators and SCFAs.
Bile acids are pleiotropic molecules that are synthesised from cholesterol in a series of steps before being secreted in conjugated forms into the biliary tract and intestine[47]. Multiple bacterially driven bio-transformations lead to deconjugation, epimerisation, formation of secondary, desulfated, oxo- and iso-bile acids[47]. Several taxa have the capability to deconjugate, de-sulfate and form oxo-bile acids, but few can mediate 7α-dehydroxylation and iso-bile acid formation[48]. Bile acid profiles in the serum, stool, and urine vary in pre-cirrhotic stages of liver disease in those with cholestatic (primary biliary cholangitis and primary sclerosing cholangitis [PSC]) and non-cholestatic forms of liver disease. Alcohol-associated liver disease induces a reduction in bile flow which reaches its nadir during alcohol-associated hepatitis[49]. As liver disease progresses to cirrhosis, the total bile acid content reduces with continued deconjugation but relatively lower formation of secondary bile acids [50]. Secondary bile acids have a U-shaped relationship with health, with levels in the very low and very high range associated with inflammation and intestinal barrier damage. Direct bile acid-related approaches in the intestine (ASBT [apical sodium-dependent bile acid transporter] inhibitors, bile acid sequestrants) and liver (ursodeoxycholic acid [UDCA]) have been studied in pre-cirrhotic liver diseases [51–53]. There is emerging data regarding the UDCA analogue Nor-UDCA in several cholestatic diseases and NAFLD [54, 55], but its direct impact on the microbiome needs to be evaluated. However, agents that indirectly modulate the impact of bile acids, such as farnesoid X receptor agonists, are being increasingly tested in NAFLD and other liver diseases with some success, owing to their ability to modulate the gut-liver axis (partly through changes in bile acid moieties[56–58]). A recent study on a bile acid analogue, aldafermin, showed that Veillonella spp could serve as a biomarker for treatment success [59]. Changes in secondary/primary bile acid ratios in faeces and circulation also determine response to FMT and can indicate a return to baseline after antibiotic therapy in patients with cirrhosis [60, 61].
SCFA production is a major bacterial contributor towards intestinal homeostasis. SCFAs activate G-protein-coupled receptors, modulate the intestinal immune system and reduce oxidative stress through the inhibition of histone deacetylases [44]. Acetate, propionate, and butyrate are produced by several species that vary by individual in their response to dietary fibre. They have varying impacts on obesity and NAFLD, which are being explored in the preclinical setting. However, the capability to produce SCFAs is generally considered to be beneficial to the human host and some studies have specifically selected patients with stools enriched in SCFA producers as donors for FMT. SCFA levels reduce with advancing liver disease severity, and interestingly, only propionate and butyrate are extracted by the liver [62]. Moreover, individual variations in SCFA generation have been linked to differences in the success of lactulose therapy [63, 64].
Since most butyrate is assimilated by the colonocytes, stool and serum SCFA levels may not reflect its true bioavailability [65]. Despite this, studies pre- and post-FMT have shown changes in SCFAs in patients with liver disease and alcohol-associated liver disease. SCFA enemas and intravenous formulations of butyrate have been studied in conditions other than liver disease with borderline success [46, 66].
Tryptophan is a complex amino acid whose degradation can occur through human or microbial pathways with target engagement in widespread organs [40, 43, 44]. Tryptophan can be directly converted to indole and its derivatives by the microbiota or can enter host kynurerine or serotonergic metabolic pathways [43, 44]. The balance between these pathways is being explored but by and large the intestinal barrier is strengthened by indole-related metabolites in alcohol-associated liver disease [39, 67]. On the other hand, oxindoles, kynurerine-related and other tryptophan metabolites are associated with poor outcomes in cirrhosis with and without HE [68–72].
Choline degradation by a wide range of gut microbes results in tri-methylamine production[73], which is modified by hepatic flavin-containing monooxygenase enzymes to form TMAO, which has important patho-physiological and prognostic value in atherosclerosis and potentially in the metabolic syndrome [74, 75]. In cirrhosis, TMAO is associated with disease severity and is affected by liver transplantation [72, 76]. Despite holding promise in heart disease, using rifaximin or probiotics to improve gut health did not improve outcomes in patients with heart failure in a recent trial [77]. In cirrhosis, TMAO generation is likely a functional test for the gut-liver axis.
Phages
The human virome consists of approximately 1013 particles per individual and is dominated by phages [14]. Phages are viruses infecting bacteria that are usually classified based on their structure (morphology) and sequence [78]. Phages are incredibly diverse and can have host ranges from extremely specific (such as particular strains of a species) to extremely broad, encompassing multiple genera [79]. Phages are categorised as virulent or temperate based on their functional behaviour. Virulent phages strictly follow a lytic life cycle. Lytic phages recognise the receptor on the bacterial cell wall surface and inject their DNA via their tail into the bacteria. Phage genomic nucleic acids are replicated inside the bacterium by hijacking the bacterial cell machinery. After progeny virions are assembled, phage enzymes lyse the bacterial cell wall from the inside resulting in a release of the newly assembled phages to the environment. Released phages continue entertaining this self-perpetuating prey-predator relationship [80]. Temperate phages can similarly inject DNA (possibly containing drug resistance and virulence factors) into bacterial cells, which then become integrated into the host’s chromosome as prophages [14]. Temperate phages can enter a lytic life cycle under certain circumstances.
Changes in the faecal virome and phageome have been described in alcohol-associated liver disease, NAFLD and cirrhosis. While viral diversity was increased in patients with alcohol-associated liver disease and alcohol-associated hepatitis, compared with non-alcoholic controls [81], more advanced NASH was associated with decreased viral diversity compared with patients with less advanced NAFLD or control individuals [82]. Patients with cirrhosis (mixed aetiology) showed a similar phage diversity as healthy controls [16]. Faecal Escherichia-, Enterobacteria-, and Enterococcus phages were higher in patients with alcohol-associated hepatitis, together with a significant increase in mammalian viruses such as Parvoviridae and Herpesviridae [81]. Escherichia, Enterobacteria, and Lactobacillus phages were more abundant in patients with advanced NAFLD [82]. Whether changes in the intestinal virome translate into modifications of the bacterial microbiota (or vice versa) and eventually affect liver disease progression is currently not known.
Phages were used as anti-bacterial drugs for a long time, but with increasing use of antibiotics their importance rapidly declined. With the emergence of multidrug-resistant bacteria, a better understanding of the human microbiota and the advent of sequencing technologies, phage-based therapies and their use in clinical trials has seen something of a resurgence. Phage therapy is considered safe even if phage cocktails are administered intravenously [83]. Efficient treatment of multidrug-resistant bacteria using phages has been documented in several case reports [83, 84]. A comprehensive overview of studies using phage therapy in patients with gastrointestinal diseases has recently been published [85].
Phages might be useful in treating diseases other than classic bacterial or pathogenic infections. A case report showed that ethanol-producing Klebsiella pneumoniae (K. pneumoniae) was associated with NASH and is present in 60% of a Chinese cohort of patients with NAFLD [86]. Oral gavage of ethanol-producing K. pneumoniae induced steatohepatitis in mice [86]. Similarly, FMT of ethanol-producing K. pneumoniae from a patient with NASH into germ-free mice resulted in steatohepatitis [86]. Selective elimination of the ethanol-producing K. pneumoniae strain using phages prior to FMT into mice prevented development of steatohepatitis [86], indicating that ex vivo phage therapy reduces liver disease.
The presence of the virulence factor and secreted toxin, cytolysin, in Enterococcus faecalis (E. faecalis) correlated with liver disease severity and mortality in patients with alcohol-associated hepatitis [15]. Gnotobiotic mouse studies confirmed the importance of cytolysin for promoting ethanol-induced liver disease in mice [15]. To demonstrate that cytolytic E. faecalis are necessary for the development of ethanol-induced steatohepatitis, microbiota humanised mice were treated orally with phages. Phages targeting cytolysin-positive E. faecalis reduced ethanol-induced liver injury, steatosis and inflammation, indicating that lytic bacteriophage treatment can selectively attenuate ethanol-induced liver disease caused by cytolysin-positive E. faecalis in humanised mice [15].
These studies are excellent examples of how pathobionts contribute to the pathogenesis of fatty liver diseases. Elimination of these bacteria using phages can decrease liver disease in preclinical models. Challenges of applying phage therapy in clinical practice include the narrow host range of some phages, route of administration, the development of bacterial host resistance and kinetics in the gastrointestinal tract.
Faecal microbiota transplantation
FMT from healthy donor(s) to patients has been used to alter the gut microbiota and represents an untargeted and novel therapeutic approach in liver diseases[87]. Initially reported for its ability to cure recurrent Clostridiodes difficile infection [88], FMT is increasingly being tested in liver diseases. Clinical trials on FMT have demonstrated safety and favourable changes in gut microbiota composition and function in alcohol-associated hepatitis and HE (Table 2 and Table S1). Preclinical and pilot data suggest that FMT may also have a role in the treatment of NAFLD [89, 90], PSC [91], chronic hepatitis B [92, 93] and hepatocellular carcinoma [94].
Table 2.
Summary of published clinical studies of IMT on liver diseases
| Year | Author | Disease | Study Type | FMT (number) | Control (number) | FMT Frequency | FMT methods | Follow-up duration | Outcome/ Main findings |
|---|---|---|---|---|---|---|---|---|---|
| 2018 | Mehta et al. | Recurrent overt hepatic encephalopathy | Case Series | 10 | - | Single | Colonoscopy | 5 months | Sustained clinical response with single FMT treatment in 6 of 10 (60%) patients |
| 2019 | Bajaj et al | Cirrhosis & Hepatic Encephalopathy | RCT | 10 | 10 (Placebo) | Single | 15 FMT capsules | 5 months | Improvement in short-term cognitive function and hospitalizations with favorable changes in mucosal and stool microbial composition and enhancement of intestinal barrier |
| 2019 | Bajaj et al. | Cirrhosis & Hepatic Encephalopathy | RCT | 10 | 10 (Placebo) | Single | 15 FMT capsules | 15 months | FMT after antibiotics is safe and potentially effective in preventing long-term recurrence of HE. |
| 2018 | Bajaj et al. | Cirrhosis & Hepatic Encephalopathy | RCT | 10 (antibiotics and then FMT) | 10 (standard-of-care, no antibiotics/FMT) | Single | Enema | 6 months | Restoration of antibiotic-associated disruption in microbial diversity and function |
| 2017 | Bajaj et al | Cirrhosis & Hepatic Encephalopathy | RCT | 10 (antibiotics and then FMT) | 10 (standard-of-care, no antibiotics/FMT) | Single | Enema | 5 months | Reduced hospitalizations, improved cognition, and dysbiosis in patients with cirrhosis with recurrent HE following FMT |
| 2018 | Philips et al. | Severe Alcohol-Associated Hepatitis | Pilot Study | 16 | 17 (nutritional support), 10 (pentoxifylline) | Single | NA | 3 months | Improved survival with lower relative risk and hazard ratios for death in FMT than steroids, nutrition and pentoxifylline arms. |
| 2017 | Philips et al. | Alcohol-Associated Hepatitis | Pilot Study | 8 | 18 (Historical control, standard of care) | Seven (daily) | Nasoduodenal tube | 12 months | Indices of liver disease severity improved significantly within the first week after FMT compared with historical controls. |
| 2021 | Chauhan et al. | Hepatitis B | Pilot Study | 14 | 15 (using antiviral drugs) | Six (four weeks interval) | Third part of the duodenum via gastroscope | 6 months | 16.7% patients had HBeAg clearance in FMT arm and none in the antiviral therapy arm. None of the patients in either arm had HBsAg loss. |
| 2017 | Ren et al. | Hepatitis B | Pilot Study | 5 | 13 (No FMT) | One to seven | NA | 10 months | A significant decline in HBeAg titer rather than HBsAg titer was observed in FMT arm compared to that at baseline |
| 2019 | Allegretti et al. | Primary sclerosing cholangitis with IBD | Pilot Study | 10 (9UC; 1CD) | - | Single | Colonoscopy | 6 months | 30% of patients experienced a 50% decrease in alkaline phosphatase levels. |
| 2020 | Craven et al | Non-alcoholic Fatty Liver Disease | RCT | 15 | 6 (FMT using autologous stool) | Single | Endoscope to the distal duodenum | 6 months | FMT did not improve insulin resistance and hepatic proton density fat fraction (PDFF) but reduced small intestinal permeability |
| 2020 | Witjes et al. | Obesity With Steatohepatitis | RCT | 11 | 12 FMT using autologous stool) | Three (eight weeks interval) | Colonoscopy | 6 months | A trend toward improved necro-inflammatory histology and significant changes in expression of hepatic genes involved in inflammation and lipid metabolism following allogenic FMT. |
#RCT: randomized controlled trial; UC: ulcerative colitis; CD: Crohn’s disease; HE: hepatic encephalopathy; IBD: inflammatory bowel disease; SAH, severe alcohol-associated hepatitis
Mechanistic and experimental studies
Although the molecular mechanism(s) underlying the action of FMT remains unclear, it is believed to work by restoring the patient’s microbiome with diverse microorganisms, restoring the normal gut microbiota. As the liver and gut are connected via the enterohepatic circulation, any alteration in gut microbiota leads to altered metabolism which indirectly affects the liver. FMT was found to have a positive effect on liver function parameters and inflammatory mediators known to be an important driver of cirrhosis-associated complications. FMT may attenuate bacterial translocation by restoring gut barrier function and/or promoting SCFA synthesis by gut bacteria [95]. Interestingly, germ-free mice receiving gut microbiota from a patient with severe alcohol-associated hepatitis developed more severe liver inflammation, worse liver necrosis, increased intestinal permeability, and bacterial translocation than mice receiving gut microbiota from patients without alcohol-associated hepatitis [96]. In addition, FMT prevents the development of ethanol-induced liver lesions and FMT-protected mice recovered a gut microbiota comparable to that of alcohol-resistant mice [97]. A study in mice demonstrated that insulin resistance and the fatty liver phenotype could be transmitted via FMT [90]. To date, FMT studies in NAFLD have mostly been performed in murine models. Mice fed a high-fat diet had a significant decrease in intrahepatic lipid accumulation and intrahepatic inflammatory cytokines after 8 weeks of FMT compared with controls [98]. Similarly, mice transplanted with faeces from patients with NASH showed increased hepatic steatosis and inflammatory cell infiltration compared to those transplanted with faeces from healthy controls [99]. Although promising, further FMT studies are needed in earlier stages of NAFLD to evaluate its effectiveness on altering liver histology and slowing disease progression in humans.
Clinical studies
Among all liver diseases, patients with severe alcohol-associated hepatitis caused by chronic and acute alcohol abuse may benefit most from FMT due to limited therapeutic options. Survival rates in patients with severe alcohol-associated hepatitis are poor (with mortality rates of up to 30%) and 60% of patients will need a liver transplant [100]. Two small studies using FMT combined with standard of care showed significant improvements in 1-year survival rate in FMT-treated patients compared with historical controls (87.5% vs. 33.3%) [101, 102]. In one of the first pilot studies by Philips et al., 8 male patients with severe alcohol-associated hepatitis who received 7 days of FMT were compared with 18 matched historical controls with severe alcohol-associated hepatitis who received standard of care during the same period. Indices of liver disease severity within the first week of FMT improved significantly in the FMT arm compared to the standard care arm, and survival was also significantly better in the FMT arm [102]. These results suggest that FMT improves survival beyond what is offered by current therapies and may be a cost-effective bridge to liver transplant. Microbial analysis revealed a reduction in Proteobacteria and an increase in Actinobacteria 1 year after FMT. Importantly, co-existence of donor and recipient species was seen up to 12 months post-FMT [102]. A randomised-controlled trial comparing FMT vs. corticosteroid therapy in patients with severe alcohol-associated hepatitis (NCT03091010 at www.clinicaltrials.gov) is in progress. A recent study also reported that alcohol craving or consumption and long-term alcohol-related hospitalisations can potentially be reduced by FMT in patients with alcohol use disorders [103].
In patients with HE whereby bacterial metabolic products can trigger cognitive impairment after passing the blood-brain barrier [104, 105], FMT represents a promising approach for modulation of the gut microbiota in association with standard therapy (lactulose or rifaximin). Pilot studies have demonstrated that FMT enema and oral capsule FMT can achieve good safety and efficacy in patients with HE [106–108]. In an open-label randomised trial, 20 outpatients with cirrhosis and recurrent HE were randomised to FMT preceded by 5 days of broad-spectrum antibiotics or standard of care with lactulose or rifaximin [109]. HE recurred in 50% of patients in the standard of care arm but no recurrence was reported in those who received FMT, which was also associated with lower hospitalisation rates and greater improvement in cognitive tests. Safety and efficacy were maintained for up to 1 year, with significantly less HE and reduced hospitalisation episodes in the FMT compared to standard care arm [110]. Improvement after FMT was accompanied by an increase in faecal microbial alpha diversity and abundance of the beneficial bacteria Lactobacillaceae and Bifidobacteriaceae [109]. A recent systematic review consisting of 4 human studies and 2 rodent studies reported that FMT overall led to an improvement in neurocognitive function and a reduction in severe adverse events in patients with HE [111].
In patients with PSC and inflammatory bowel disease, a single dose of FMT was safe and feasible and was associated with a decrease in alkaline phosphatase levels when patients were followed for up to 6 months [91]. However, the use of alkaline phosphatase as a predictor of outcomes in PSC remains controversial and the lack of validated surrogate biomarkers in PSC makes it difficult to draw definitive conclusions [112]. Two pilot studies reported that FMT was associated with a significant reduction in HBeAg titre compared to that at baseline, but a similar trend was not observed in HBsAg [92, 93]. In a randomised pilot study of patients with NAFLD, insulin resistance and hepatic steatosis were not improved, but small intestinal permeability was reduced after FMT. Since elevated intestinal permeability has been implicated in liver disease, further investigation of FMT in this population is warranted [89]. Several randomised-controlled trials addressing the therapeutic modulation of gut microbiota in HE, outcome of severe alcohol-associated hepatitis, metabolic consequences of liver disease, NASH and fibrosis progression are currently underway (trials number NCT02485106, NCT02862249, NCT01069133, NCT02400216, NCT02496390, NCT02424175, NCT01968382 and NCT02469272 at www.clinicaltrials.gov)
One major concern with FMT in patients with liver disease is the risk of infections. Patients with cirrhosis are susceptible to infections due to cirrhosis-associated immune dysfunction. Studies have reported a risk of spontaneous bacterial peritonitis and transfer of multidrug-resistant bacteria after FMT. Furthermore, not all FMT studies provided specific causes of deaths for patients [108, 113]. The methods for determining donor eligibility based on gut microbiota composition varied among studies, ranging from clinical questionnaires and screening for transmittable pathogens to metagenomic sequencing.
Challenges and future directions
One of the largest gaps in microbiota modulation is the ability to translate these discoveries into humans and assess their clinical relevance. Better characterisation of the gut microbiome, metabolome and host response using robust preclinical models, various stages of liver disease and bigger and longitudinal cohorts of patients will allow us to determine subtypes of patients with liver disease who would benefit most from microbiota modulation. Additional research is required to uncover determinants and mechanisms by which gut microbiome composition would render an individual susceptible or resistant to a specific intervention. Since several studies showed that modulations of the gut microbiome might be transient, with reversal back to baseline after a few weeks to months, longitudinal and longer term studies to identify which therapeutics result in long-lasting microbiota changes and effective clinical outcomes will be needed. We envision that developing functional assays to test how individual microbes respond to a particular intervention may unlock the potential of the gut microbiota as a predictor of clinical outcome.
To date, most studies have relied on analysing microbiota composition using 16S rRNA gene sequencing, but accurately predicting responses may require analysis of metagenomes (all the genomic content of a microbiota) and/or meta-transcriptomes (all the genes actively expressed by a microbiota) to identify responders and non-responders to different types of intervention. In addition to the bacterial species level, identification of microbiota communities at the strain level, including those that are present in low abundance yet have an important role in outcomes, as well as the role of viruses and fungi along with bacteria may need to be investigated following FMT [114]. Determining which microbiota subpopulation should be used to properly stratify patient populations – e.g. the luminal microbiota, faecal microbiota, mucosa-associated microbiota, or inner mucus-associated microbiota – will be important, as these microbiota subtypes communicate with the host or respond to dietary interventions in different ways. Recently, studies have revealed personalised diet-microbiome associations in humans [115] and used the prediction of glycaemic responses to guide personalised dietary interventions [116]. Nearly all human diet studies to date have shown large inter-individual variations in the gut microbiome and metabolome. Ultimately, the impact of diet on microbiota in liver disease needs to be controlled for before comparing across populations.
Probiotics, prebiotics, synbiotics, postbiotics, antibiotics and FMT have demonstrated efficacy and mostly been well tolerated in randomised clinical trials. These untargeted therapies will likely be replaced by personalised and precision medicine approaches including bio-engineered bacterial strains or phages that modulate specific bacterial enzymes and metabolic pathways, which – if successfully tested in clinical trials – will transform the way liver diseases such as alcohol-associated liver disease and NAFLD/NASH are managed in clinical practice. With more advanced technologies, more precise interventions with a better side-effect profile can be developed. It appears likely that in the future, before initiating any targeted or untargeted microbiome therapies, a detailed analysis of the patient’s microbiome including metabolite assessment will be performed to determine which precision medicine approach will be the most likely to improve liver disease.
Currently, the risk to benefit ratio of using FMT to treat chronic liver disease, cirrhosis and HE remains unclear. Given that patients with cirrhosis have compromised intestinal barrier integrity and are immunocompromised, there is potential of transmission of unwanted pathogenic organisms that are not routinely screened for. Transmission of drug-resistant E. coli from a donor following FMT has been reported and can result in death, hence careful selection of donor and recipient is necessary to minimise the risks [113]. In 2019, the FDA updated their protocol on FMT to mandate that donors be screened for multidrug-resistant organisms and, based on the detection of SAR-CoV-2 in faecal samples of asymptomatic individuals, donor stool will also need to be screened for SARS-CoV-2 [117]. Openbiome in the United States demonstrated that less than 2% of individuals who applied to be donors were ultimately selected[118], and a recent study in Hong Kong found that less than 1% of potential donors were successfully recruited as regular donors because of the high prevalence of extended-spectrum beta lactamase-producing organisms in stool [119]. Rigorous screening is crucial as sicker patients and those with more advanced liver disease are candidates for FMT. Once FMT is administered, patients need to be monitored closely for unwanted effects in the short- and longer term.
The complexity of chronic liver disease and cirrhosis require a multi-faceted treatment approach and the possibility of long-lasting cure from FMT alone is highly unlikely given the multi-factorial nature of chronic liver disease. Unlike Clostridioides difficile, cirrhosis is the result of decades of liver injury; thus, a realistic expectation of what can be achieved following microbiota modulation needs to be reinforced. It is clear that microbiota modulation may be more effective as an adjuvant to existing therapies for liver disease, rather than as a sole therapy. Uncertainty also exists regarding the duration and number of FMT administrations required. For example, it is likely that NAFLD stemming from a long-standing but slow process will require multiple FMT over time compared to a rapidly progressing disorder such as alcohol-associated hepatitis. Currently, most studies have utilised a single donor. It is unclear whether positive results can be reproduced using a different donor, and to what degree the specific composition of the donor microbiome affects outcomes. It has been shown in patients with ulcerative colitis and obesity/metabolic diseases that donor and recipient microbiome profile influenced FMT success [120, 121]. Data on the dose, frequency, route of modulation of gut microbiota, including small bowel microbiota, are needed for different liver diseases. A PROspective, randomised placebo-controlled feasibility trial of Faecal mIcrobiota Transplantation (PROFIT) will assess infusion of FMT or placebo directly into the small bowel instead of the colon, directly targeting the small bowel bacterial overgrowth observed in patients with cirrhosis [122], while another randomised-controlled trial is assessing the effect of multiple doses and concomitant oral capsule and enema routes in patients with HE in the United States (Clinicaltrials.gov NCT03796598). FMT needs to be universally available, and patients need to be accepting of this therapy.
In summary, the human gut microbiota potentially plays a major role in the causation and progression of chronic liver disease, although our understanding of the interaction between the gut and the liver is still incomplete. There are immense clinical implications for defining the microbial basis of liver disease, not least for the development of microbiome-centred interventions that can effectively reduce disease severity and slow progression toward cirrhosis and its complications. Targeted therapies such as engineered bacteria, postbiotics and phages have mostly been tested in preclinical models. Ultimately, the efficacy and safety of microbiota-centred therapeutics need to be evaluated from a rigorous pharmacologic perspective and in larger randomised-controlled trials in patients with liver disease.
Supplementary Material
Financial support
This review was supported in part by NIH grants R01 AA24726, R01 AA020703, U01 AA026939, by Award Number BX004594 from the Biomedical Laboratory Research & Development Service of the VA Office of Research and Development, and a Biocodex Microbiota Foundation Grant (to B.S.) and services provided by NIH centers P30 DK120515 and P50 AA011999. The review was also supported by NIH grants R21TR003095 and VA merit review CX001076 (to J.S.B.).
Abbreviations
- E. faecalis
Enterococcus faecalis
- FMT
faecal microbiota transplantation
- HE
hepatic encephalopathy
- IL
interleukin
- K. pneumoniae
Klebsiella pneumoniae
- NAFLD
non-alcoholic fatty liver disease
- NASH
non-alcoholic steatohepatitis
- PSC
primary sclerosing cholangitis
- SCFAs
short-chain fatty acids
- TMAO
tri-methylamine oxide
Footnotes
Conflicts of interest
B.S. has been consulting for Ferring Research Institute, HOST Therabiomics, Intercept Pharmaceuticals, Mabwell Therapeutics, Patara Pharmaceuticals and Takeda. B.S.’s institution UC San Diego has received research support from Axial Biotherapeutics, BiomX, CymaBay Therapeutics, NGM Biopharmaceuticals, Prodigy Biotech and Synlogic Operating Company. B.S. is founder of Nterica Bio. UC San Diego has filed several patents with B.S. as inventor related to this work. J.S.B.’s institution has received research support from Bausch health, Mallinckrodt Pharmaceuticals, Grifols Pharmaceuticals, Kaleido, Enterome, and Sequana Pharmaceuticals. Virginia Commonwealth University has filed patents with J.S.B. as inventor. S.C.N. has served as speaker for Janssen, Abbvie, Takeda, Ferring Tillotts and Menarini. S.C.N.’s institution The Chinese University of Hong Kong has received research support from Olympus, Ferring and Abbvie. S.C.N. is scientific cofounder for GenieBiome Limited. The Chinese University of Hong Kong has filed several patents with S.C.N. as inventor relating to microbiome therapeutics in Clos. difficile infections.
Please refer to the accompanying ICMJE disclosure forms for further details.
References
- [1].Bajaj JS, Khoruts A. Microbiota changes and intestinal microbiota transplantation in liver diseases and cirrhosis. J Hepatol 2020;72:1003–1027. [DOI] [PubMed] [Google Scholar]
- [2].Collaborators GBDC. The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol 2020;5:245–266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Julien J, Ayer T, Bethea ED, Tapper EB, Chhatwal J. Projected prevalence and mortality associated with alcohol-related liver disease in the USA, 2019–40: a modelling study. Lancet Public Health 2020;5:e316–e323. [DOI] [PubMed] [Google Scholar]
- [4].Arumugam M, Raes J, Pelletier E, Le Paslier D, Yamada T, Mende DR, et al. Enterotypes of the human gut microbiome. Nature 2011;473:174–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Bajaj JS, Kamath PS, Reddy KR. The Evolving Challenge of Infections in Cirrhosis. Reply. N Engl J Med 2021;385:1151–1152. [DOI] [PubMed] [Google Scholar]
- [6].Tilg H, Cani PD, Mayer EA. Gut microbiome and liver diseases. Gut 2016;65:2035–2044. [DOI] [PubMed] [Google Scholar]
- [7].Allaband C, McDonald D, Vazquez-Baeza Y, Minich JJ, Tripathi A, Brenner DA, et al. Microbiome 101: Studying, Analyzing, and Interpreting Gut Microbiome Data for Clinicians. Clin Gastroenterol Hepatol 2019;17:218–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Trebicka J, Bork P, Krag A, Arumugam M. Utilizing the gut microbiome in decompensated cirrhosis and acute-on-chronic liver failure. Nat Rev Gastroenterol Hepatol 2021;18:167–180. [DOI] [PubMed] [Google Scholar]
- [9].Yang AM, Inamine T, Hochrath K, Chen P, Wang L, Llorente C, et al. Intestinal fungi contribute to development of alcoholic liver disease. J Clin Invest 2017;127:2829–2841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Bajaj JS, Liu EJ, Kheradman R, Fagan A, Heuman DM, White M, et al. Fungal dysbiosis in cirrhosis. Gut 2018;67:1146–1154. [DOI] [PubMed] [Google Scholar]
- [11].de Cena JA, Zhang J, Deng D, Dame-Teixeira N, Do T. Low-Abundant Microorganisms: The Human Microbiome’s Dark Matter, a Scoping Review. Front Cell Infect Microbiol 2021;11:689197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Zuo T, Sun Y, Wan Y, Yeoh YK, Zhang F, Cheung CP, et al. Human-Gut-DNA Virome Variations across Geography, Ethnicity, and Urbanization. Cell Host Microbe 2020;28:741–751 e744. [DOI] [PubMed] [Google Scholar]
- [13].Roux S, Paez-Espino D, Chen IA, Palaniappan K, Ratner A, Chu K, et al. IMG/VR v3: an integrated ecological and evolutionary framework for interrogating genomes of uncultivated viruses. Nucleic Acids Res 2021;49:D764–D775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Liang G, Bushman FD. The human virome: assembly, composition and host interactions. Nat Rev Microbiol 2021;19:514–527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Duan Y, Llorente C, Lang S, Brandl K, Chu H, Jiang L, et al. Bacteriophage targeting of gut bacterium attenuates alcoholic liver disease. Nature 2019;575:505–511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Bajaj JS, Sikaroodi M, Shamsaddini A, Henseler Z, Santiago-Rodriguez T, Acharya C, et al. Interaction of bacterial metagenome and virome in patients with cirrhosis and hepatic encephalopathy. Gut 2021;70:1162–1173. [DOI] [PubMed] [Google Scholar]
- [17].Borrel G, Brugere JF, Gribaldo S, Schmitz RA, Moissl-Eichinger C. The host-associated archaeome. Nat Rev Microbiol 2020;18:622–636. [DOI] [PubMed] [Google Scholar]
- [18].Lang S, Schnabl B. Microbiota and Fatty Liver Disease-the Known, the Unknown, and the Future. Cell Host Microbe 2020;28:233–244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Biggins SW, Angeli P, Garcia-Tsao G, Gines P, Ling SC, Nadim MK, et al. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 2021;74:1014–1048. [DOI] [PubMed] [Google Scholar]
- [20].Patidar KR, Bajaj JS. Antibiotics for the treatment of hepatic encephalopathy. Metab Brain Dis 2013;28:307–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Caraceni P, Vargas V, Sola E, Alessandria C, de Wit K, Trebicka J, et al. The Use of Rifaximin in Patients With Cirrhosis. Hepatology 2021;74:1660–1673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Bass NM, Mullen KD, Sanyal A, Poordad F, Neff G, Leevy CB, et al. Rifaximin treatment in hepatic encephalopathy. N Engl J Med 2010;362:1071–1081. [DOI] [PubMed] [Google Scholar]
- [23].Patel V, Lee S, McPhail M, Da Silva K, Guilly S, Zamalloa A, et al. Rifaximin reduces gut-derived inflammation and mucin degradation in cirrhosis and encephalopathy: RIFSYS randomised controlled trial. J Hepatol 2021. [DOI] [PubMed] [Google Scholar]
- [24].Shamsaddini A, Gillevet PM, Acharya C, Fagan A, Gavis E, Sikaroodi M, et al. Impact of Antibiotic Resistance Genes in Gut Microbiome of Patients With Cirrhosis. Gastroenterology 2021;161:508–521 e507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Bajaj JS, Reddy RK, Tandon P, Wong F, Kamath PS, Biggins SW, et al. Prediction of Fungal Infection Development and Their Impact on Survival Using the NACSELD Cohort. Am J Gastroenterol 2018;113:556–563. [DOI] [PubMed] [Google Scholar]
- [26].Fernandez J, Piano S, Bartoletti M, Wey EQ. Management of bacterial and fungal infections in cirrhosis: The MDRO challenge. J Hepatol 2021;75 Suppl 1:S101–S117. [DOI] [PubMed] [Google Scholar]
- [27].Piano S, Singh V, Caraceni P, Maiwall R, Alessandria C, Fernandez J, et al. Epidemiology and Effects of Bacterial Infections in Patients With Cirrhosis Worldwide. Gastroenterology 2019;156:1368–1380 e1310. [DOI] [PubMed] [Google Scholar]
- [28].Yang L, Bian X, Wu W, Lv L, Li Y, Ye J, et al. Protective effect of Lactobacillus salivarius Li01 on thioacetamide-induced acute liver injury and hyperammonaemia. Microb Biotechnol 2020;13:1860–1876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Zhuge A, Li B, Yuan Y, Lv L, Li Y, Wu J, et al. Lactobacillus salivarius LI01 encapsulated in alginate-pectin microgels ameliorates D-galactosamine-induced acute liver injury in rats. Appl Microbiol Biotechnol 2020;104:7437–7455. [DOI] [PubMed] [Google Scholar]
- [30].Gu Z, Li F, Liu Y, Jiang M, Zhang L, He L, et al. Exosome-Like Nanoparticles From Lactobacillus rhamnosusGG Protect Against Alcohol-Associated Liver Disease Through Intestinal Aryl Hydrocarbon Receptor in Mice. Hepatol Commun 2021;5:846–864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Liu Y, Chen K, Li F, Gu Z, Liu Q, He L, et al. Probiotic Lactobacillus rhamnosus GG Prevents Liver Fibrosis Through Inhibiting Hepatic Bile Acid Synthesis and Enhancing Bile Acid Excretion in Mice. Hepatology 2020;71:2050–2066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Kirpich IA, Solovieva NV, Leikhter SN, Shidakova NA, Lebedeva OV, Sidorov PI, et al. Probiotics restore bowel flora and improve liver enzymes in human alcohol-induced liver injury: a pilot study. Alcohol 2008;42:675–682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Dhiman RK, Rana B, Agrawal S, Garg A, Chopra M, Thumburu KK, et al. Probiotic VSL#3 reduces liver disease severity and hospitalization in patients with cirrhosis: a randomized, controlled trial. Gastroenterology 2014;147:1327–1337 e1323. [DOI] [PubMed] [Google Scholar]
- [34].Dalal R, McGee RG, Riordan SM, Webster AC. Probiotics for people with hepatic encephalopathy. Cochrane Database Syst Rev 2017;2:CD008716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Xiao MW, Lin SX, Shen ZH, Luo WW, Wang XY. Systematic Review with Meta-Analysis: The Effects of Probiotics in Nonalcoholic Fatty Liver Disease. Gastroenterol Res Pract 2019;2019:1484598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Tang Y, Huang J, Zhang WY, Qin S, Yang YX, Ren H, et al. Effects of probiotics on nonalcoholic fatty liver disease: a systematic review and meta-analysis. Therap Adv Gastroenterol 2019;12:1756284819878046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Douillard FP, de Vos WM. Biotechnology of health-promoting bacteria. Biotechnol Adv 2019;37:107369. [DOI] [PubMed] [Google Scholar]
- [38].Steidler L, Hans W, Schotte L, Neirynck S, Obermeier F, Falk W, et al. Treatment of murine colitis by Lactococcus lactis secreting interleukin-10. Science 2000;289:1352–1355. [DOI] [PubMed] [Google Scholar]
- [39].Hendrikx T, Schnabl B. Indoles: metabolites produced by intestinal bacteria capable of controlling liver disease manifestation. J Intern Med 2019;286:32–40. [DOI] [PubMed] [Google Scholar]
- [40].Hendrikx T, Duan Y, Wang Y, Oh JH, Alexander LM, Huang W, et al. Bacteria engineered to produce IL-22 in intestine induce expression of REG3G to reduce ethanol-induced liver disease in mice. Gut 2019;68:1504–1515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Duan FF, Liu JH, March JC. Engineered commensal bacteria reprogram intestinal cells into glucose-responsive insulin-secreting cells for the treatment of diabetes. Diabetes 2015;64:1794–1803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Kurtz CB, Millet YA, Puurunen MK, Perreault M, Charbonneau MR, Isabella VM, et al. An engineered E. coli Nissle improves hyperammonemia and survival in mice and shows dose-dependent exposure in healthy humans. Sci Transl Med 2019;11. [DOI] [PubMed] [Google Scholar]
- [43].Krautkramer KA, Fan J, Backhed F. Gut microbial metabolites as multi-kingdom intermediates. Nat Rev Microbiol 2021;19:77–94. [DOI] [PubMed] [Google Scholar]
- [44].Agus A, Clement K, Sokol H. Gut microbiota-derived metabolites as central regulators in metabolic disorders. Gut 2021;70:1174–1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Chu H, Duan Y, Yang L, Schnabl B. Small metabolites, possible big changes: a microbiota-centered view of non-alcoholic fatty liver disease. Gut 2019;68:359–370. [DOI] [PubMed] [Google Scholar]
- [46].Salminen S, Collado MC, Endo A, Hill C, Lebeer S, Quigley EMM, et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat Rev Gastroenterol Hepatol 2021;18:649–667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Ridlon JM, Harris SC, Bhowmik S, Kang DJ, Hylemon PB. Consequences of bile salt biotransformations by intestinal bacteria. Gut Microbes 2016;7:22–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Wahlstrom A, Sayin SI, Marschall HU, Backhed F. Intestinal Crosstalk between Bile Acids and Microbiota and Its Impact on Host Metabolism. Cell Metab 2016;24:41–50. [DOI] [PubMed] [Google Scholar]
- [49].Hartmann P, Hochrath K, Horvath A, Chen P, Seebauer CT, Llorente C, et al. Modulation of the intestinal bile acid-FXR-FGF15 axis improves alcoholic liver disease in mice. Hepatology 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Kakiyama G, Pandak WM, Gillevet PM, Hylemon PB, Heuman DM, Daita K, et al. Modulation of the fecal bile acid profile by gut microbiota in cirrhosis. J Hepatol 2013;58:949–955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Al-Dury S, Wahlstrom A, Wahlin S, Langedijk J, Elferink RO, Stahlman M, et al. Pilot study with IBAT inhibitor A4250 for the treatment of cholestatic pruritus in primary biliary cholangitis. Sci Rep 2018;8:6658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [52].Al-Dury S, Marschall HU. Ileal Bile Acid Transporter Inhibition for the Treatment of Chronic Constipation, Cholestatic Pruritus, and NASH. Front Pharmacol 2018;9:931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Le TA, Chen J, Changchien C, Peterson MR, Kono Y, Patton H, et al. Effect of colesevelam on liver fat quantified by magnetic resonance in nonalcoholic steatohepatitis: a randomized controlled trial. Hepatology 2012;56:922–932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Traussnigg S, Schattenberg JM, Demir M, Wiegand J, Geier A, Teuber G, et al. Norursodeoxycholic acid versus placebo in the treatment of non-alcoholic fatty liver disease: a double-blind, randomised, placebo-controlled, phase 2 dose-finding trial. Lancet Gastroenterol Hepatol 2019;4:781–793. [DOI] [PubMed] [Google Scholar]
- [55].Fickert P, Hirschfield GM, Denk G, Marschall HU, Altorjay I, Farkkila M, et al. norUrsodeoxycholic acid improves cholestasis in primary sclerosing cholangitis. J Hepatol 2017;67:549–558. [DOI] [PubMed] [Google Scholar]
- [56].Schwabl P, Hambruch E, Seeland BA, Hayden H, Wagner M, Garnys L, et al. The FXR agonist PX20606 ameliorates portal hypertension by targeting vascular remodelling and sinusoidal dysfunction. J Hepatol 2017;66:724–733. [DOI] [PubMed] [Google Scholar]
- [57].Kremoser C. FXR agonists for NASH: How are they different and what difference do they make? J Hepatol 2021;75:12–15. [DOI] [PubMed] [Google Scholar]
- [58].Friedman ES, Li Y, Shen TD, Jiang J, Chau L, Adorini L, et al. FXR-Dependent Modulation of the Human Small Intestinal Microbiome by the Bile Acid Derivative Obeticholic Acid. Gastroenterology 2018;155:1741–1752 e1745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Loomba R, Ling L, Dinh DM, DePaoli AM, Lieu HD, Harrison SA, et al. The Commensal Microbe Veillonella as a Marker for Response to an FGF19 Analog in NASH. Hepatology 2021;73:126–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Bajaj JS, Kakiyama G, Savidge T, Takei H, Kassam ZA, Fagan A, et al. Antibiotic-Associated Disruption of Microbiota Composition and Function in Cirrhosis is Restored by Fecal Transplant. Hepatology 2018. [DOI] [PubMed] [Google Scholar]
- [61].Bajaj JS, Salzman N, Acharya C, Takei H, Kakiyama G, Fagan A, et al. Microbial Functional Change is Linked with Clinical Outcomes after Capsular Fecal Transplant in Cirrhosis. JCI Insight 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Juanola O, Ferrusquia-Acosta J, Garcia-Villalba R, Zapater P, Magaz M, Marin A, et al. Circulating levels of butyrate are inversely related to portal hypertension, endotoxemia, and systemic inflammation in patients with cirrhosis. FASEB J 2019;33:11595–11605. [DOI] [PubMed] [Google Scholar]
- [63].Jin M, Kalainy S, Baskota N, Chiang D, Deehan EC, McDougall C, et al. Faecal microbiota from patients with cirrhosis has a low capacity to ferment non-digestible carbohydrates into short-chain fatty acids. Liver Int 2019;39:1437–1447. [DOI] [PubMed] [Google Scholar]
- [64].Bloemen JG, Olde Damink SW, Venema K, Buurman WA, Jalan R, Dejong CH. Short chain fatty acids exchange: Is the cirrhotic, dysfunctional liver still able to clear them? Clin Nutr 2010;29:365–369. [DOI] [PubMed] [Google Scholar]
- [65].van der Beek CM, Bloemen JG, van den Broek MA, Lenaerts K, Venema K, Buurman WA, et al. Hepatic Uptake of Rectally Administered Butyrate Prevents an Increase in Systemic Butyrate Concentrations in Humans. J Nutr 2015;145:2019–2024. [DOI] [PubMed] [Google Scholar]
- [66].Canfora EE, van der Beek CM, Jocken JWE, Goossens GH, Holst JJ, Olde Damink SWM, et al. Colonic infusions of short-chain fatty acid mixtures promote energy metabolism in overweight/obese men: a randomized crossover trial. Sci Rep 2017;7:2360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [67].Taleb S. Tryptophan Dietary Impacts Gut Barrier and Metabolic Diseases. Front Immunol 2019;10:2113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Riggio O, Mannaioni G, Ridola L, Angeloni S, Merli M, Carla V, et al. Peripheral and splanchnic indole and oxindole levels in cirrhotic patients: a study on the pathophysiology of hepatic encephalopathy. Am J Gastroenterol 2010;105:1374–1381. [DOI] [PubMed] [Google Scholar]
- [69].Claria J, Moreau R, Fenaille F, Amoros A, Junot C, Gronbaek H, et al. Orchestration of Tryptophan-Kynurenine Pathway, Acute Decompensation, and Acute-on-Chronic Liver Failure in Cirrhosis. Hepatology 2019;69:1686–1701. [DOI] [PubMed] [Google Scholar]
- [70].Bajaj JS, Reddy KR, O’Leary JG, Vargas HE, Lai JC, Kamath PS, et al. Serum Levels of Metabolites Produced by Intestinal Microbes and Lipid Moieties Independently Associated With Acute-on-Chronic Liver Failure and Death in Patients With Cirrhosis. Gastroenterology 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [71].Bajaj JS, Fan S, Thacker LR, Fagan A, Gavis E, White MB, et al. Serum and urinary metabolomics and outcomes in cirrhosis. PLoS One 2019;14:e0223061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Jimenez B, Montoliu C, MacIntyre DA, Serra MA, Wassel A, Jover M, et al. Serum metabolic signature of minimal hepatic encephalopathy by (1)H-nuclear magnetic resonance. J Proteome Res 2010;9:5180–5187. [DOI] [PubMed] [Google Scholar]
- [73].Hosseinkhani F, Heinken A, Thiele I, Lindenburg PW, Harms AC, Hankemeier T. The contribution of gut bacterial metabolites in the human immune signaling pathway of non-communicable diseases. Gut Microbes 2021;13:1–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011;472:57–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [75].Barrea L, Annunziata G, Muscogiuri G, Di Somma C, Laudisio D, Maisto M, et al. Trimethylamine-N-oxide (TMAO) as Novel Potential Biomarker of Early Predictors of Metabolic Syndrome. Nutrients 2018;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Bajaj JS, Kakiyama G, Cox IJ, Nittono H, Takei H, White M, et al. Alterations in gut microbial function following liver transplant. Liver Transpl 2018;24:752–761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Awoyemi A, Mayerhofer C, Felix AS, Hov JR, Moscavitch SD, Lappegard KT, et al. Rifaximin or Saccharomyces boulardii in heart failure with reduced ejection fraction: Results from the randomized GutHeart trial. EBioMedicine 2021;70:103511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [78].Ackermann HW, Prangishvili D. Prokaryote viruses studied by electron microscopy. Arch Virol 2012;157:1843–1849. [DOI] [PubMed] [Google Scholar]
- [79].Ross A, Ward S, Hyman P. More Is Better: Selecting for Broad Host Range Bacteriophages. Front Microbiol 2016;7:1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [80].Duan Y, Young R, Schnabl B. Bacteriophages and their potential for treatment of gastrointestinal diseases. Nat Rev Gastroenterol Hepatol 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [81].Jiang L, Lang S, Duan Y, Zhang X, Gao B, Chopyk J, et al. Intestinal virome in patients with alcoholic hepatitis. Hepatology 2020;72:2182–2196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [82].Lang S, Demir M, Martin A, Jiang L, Zhang X, Duan Y, et al. Intestinal Virome Signature Associated With Severity of Nonalcoholic Fatty Liver Disease. Gastroenterology 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [83].Schooley RT, Biswas B, Gill JJ, Hernandez-Morales A, Lancaster J, Lessor L, et al. Development and Use of Personalized Bacteriophage-Based Therapeutic Cocktails To Treat a Patient with a Disseminated Resistant Acinetobacter baumannii Infection. Antimicrob Agents Chemother 2017;61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [84].Jennes S, Merabishvili M, Soentjens P, Pang KW, Rose T, Keersebilck E, et al. Use of bacteriophages in the treatment of colistin-only-sensitive Pseudomonas aeruginosa septicaemia in a patient with acute kidney injury-a case report. Crit Care 2017;21:129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [85].Hsu CL, Duan Y, Fouts DE, Schnabl B. Intestinal virome and therapeutic potential of bacteriophages in liver disease. J Hepatol 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [86].Yuan J, Chen C, Cui J, Lu J, Yan C, Wei X, et al. Fatty Liver Disease Caused by High-Alcohol-Producing Klebsiella pneumoniae. Cell Metab 2019;30:675–688 e677. [DOI] [PubMed] [Google Scholar]
- [87].Simone S. Li AZ, Vladimir Benes, Costea Paul I., RajnaHercog, Hildebrand Falk, Huerta-Cepas Jaime, Nieuwdorp Max, Salojärvi Jarkko, Voigt Anita Y., Zeller Georg, Sunagawa Shinichi, de Vos Willem M., Bork Peer. Durable coexistence of donor and recipient strains after fecal microbiota transplantation. Science 2016;352:586–589. [DOI] [PubMed] [Google Scholar]
- [88].van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med 2013;368:407–415. [DOI] [PubMed] [Google Scholar]
- [89].Craven L, Rahman A, Nair Parvathy S, Beaton M, Silverman J, Qumosani K, et al. Allogenic Fecal Microbiota Transplantation in Patients With Nonalcoholic Fatty Liver Disease Improves Abnormal Small Intestinal Permeability: A Randomized Control Trial. Am J Gastroenterol 2020;115:1055–1065. [DOI] [PubMed] [Google Scholar]
- [90].Le Roy T, Llopis M, Lepage P, Bruneau A, Rabot S, Bevilacqua C, et al. Intestinal microbiota determines development of non-alcoholic fatty liver disease in mice. Gut 2013;62:1787–1794. [DOI] [PubMed] [Google Scholar]
- [91].Allegretti JR, Kassam Z, Carrellas M, Mullish BH, Marchesi JR, Pechlivanis A, et al. Fecal Microbiota Transplantation in Patients With Primary Sclerosing Cholangitis: A Pilot Clinical Trial. Am J Gastroenterol 2019;114:1071–1079. [DOI] [PubMed] [Google Scholar]
- [92].Chauhan A, Kumar R, Sharma S, Mahanta M, Vayuuru SK, Nayak B, et al. Fecal Microbiota Transplantation in Hepatitis B e Antigen-Positive Chronic Hepatitis B Patients: A Pilot Study. Dig Dis Sci 2021;66:873–880. [DOI] [PubMed] [Google Scholar]
- [93].Ren YD, Ye ZS, Yang LZ, Jin LX, Wei WJ, Deng YY, et al. Fecal microbiota transplantation induces hepatitis B virus e-antigen (HBeAg) clearance in patients with positive HBeAg after long-term antiviral therapy. Hepatology 2017;65:1765–1768. [DOI] [PubMed] [Google Scholar]
- [94].Ponziani FR, Bhoori S, Castelli C, Putignani L, Rivoltini L, Del Chierico F, et al. Hepatocellular Carcinoma Is Associated With Gut Microbiota Profile and Inflammation in Nonalcoholic Fatty Liver Disease. Hepatology 2019;69:107–120. [DOI] [PubMed] [Google Scholar]
- [95].Sheng L, Jena PK, Hu Y, Liu HX, Nagar N, Kalanetra KM, et al. Hepatic inflammation caused by dysregulated bile acid synthesis is reversible by butyrate supplementation. J Pathol 2017;243:431–441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [96].Llopis M, Cassard AM, Wrzosek L, Boschat L, Bruneau A, Ferrere G, et al. Intestinal microbiota contributes to individual susceptibility to alcoholic liver disease. Gut 2016;65:830–839. [DOI] [PubMed] [Google Scholar]
- [97].Ferrere G, Wrzosek L, Cailleux F, Turpin W, Puchois V, Spatz M, et al. Fecal microbiota manipulation prevents dysbiosis and alcohol-induced liver injury in mice. J Hepatol 2017;66:806–815. [DOI] [PubMed] [Google Scholar]
- [98].Zhou D, Pan Q, Shen F, Cao HX, Ding WJ, Chen YW, et al. Total fecal microbiota transplantation alleviates high-fat diet-induced steatohepatitis in mice via beneficial regulation of gut microbiota. Sci Rep 2017;7:1529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [99].Chiu CC, Ching YH, Li YP, Liu JY, Huang YT, Huang YW, et al. Nonalcoholic Fatty Liver Disease Is Exacerbated in High-Fat Diet-Fed Gnotobiotic Mice by Colonization with the Gut Microbiota from Patients with Nonalcoholic Steatohepatitis. Nutrients 2017;9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [100].Thursz MR, Richardson P, Allison M, Austin A, Bowers M, Day CP, et al. Prednisolone or pentoxifylline for alcoholic hepatitis. N Engl J Med 2015;372:1619–1628. [DOI] [PubMed] [Google Scholar]
- [101].Philips CA, Phadke N, Ganesan K, Ranade S, Augustine P. Corticosteroids, nutrition, pentoxifylline, or fecal microbiota transplantation for severe alcoholic hepatitis. Indian J Gastroenterol 2018;37:215–225. [DOI] [PubMed] [Google Scholar]
- [102].Philips CA, Pande A, Shasthry SM, Jamwal KD, Khillan V, Chandel SS, et al. Healthy Donor Fecal Microbiota Transplantation in Steroid-Ineligible Severe Alcoholic Hepatitis: A Pilot Study. Clin Gastroenterol Hepatol 2017;15:600–602. [DOI] [PubMed] [Google Scholar]
- [103].Bajaj JS, Gavis EA, Fagan A, Wade JB, Thacker LR, Fuchs M, et al. A Randomized Clinical Trial of Fecal Microbiota Transplant for Alcohol Use Disorder. Hepatology 2021;73:1688–1700. [DOI] [PubMed] [Google Scholar]
- [104].Wijdicks EF. Hepatic Encephalopathy. N Engl J Med 2016;375:1660–1670. [DOI] [PubMed] [Google Scholar]
- [105].Rai R, Saraswat VA, Dhiman RK. Gut microbiota: its role in hepatic encephalopathy. J Clin Exp Hepatol 2015;5:S29–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [106].Bajaj JS, Kakiyama G, Savidge T, Takei H, Kassam ZA, Fagan A, et al. Antibiotic-Associated Disruption of Microbiota Composition and Function in Cirrhosis Is Restored by Fecal Transplant. Hepatology 2018;68:1549–1558. [DOI] [PubMed] [Google Scholar]
- [107].Bajaj JS, Salzman NH, Acharya C, Sterling RK, White MB, Gavis EA, et al. Fecal Microbial Transplant Capsules Are Safe in Hepatic Encephalopathy: A Phase 1, Randomized, Placebo-Controlled Trial. Hepatology 2019;70:1690–1703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [108].Mehta R, Kabrawala M, Nandwani S, Kalra P, Patel C, Desai P, et al. Preliminary experience with single fecal microbiota transplant for treatment of recurrent overt hepatic encephalopathy-A case series. Indian J Gastroenterol 2018;37:559–562. [DOI] [PubMed] [Google Scholar]
- [109].Bajaj JS, Kassam Z, Fagan A, Gavis EA, Liu E, Cox IJ, et al. Fecal microbiota transplant from a rational stool donor improves hepatic encephalopathy: A randomized clinical trial. Hepatology 2017;66:1727–1738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [110].Bajaj JS, Fagan A, Gavis EA, Kassam Z, Sikaroodi M, Gillevet PM. Long-term Outcomes of Fecal Microbiota Transplantation in Patients With Cirrhosis. Gastroenterology 2019;156:1921–1923 e1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [111].Madsen M, Kimer N, Bendtsen F, Petersen AM. Fecal microbiota transplantation in hepatic encephalopathy: a systematic review. Scand J Gastroenterol 2021;56:560–569. [DOI] [PubMed] [Google Scholar]
- [112].Ponsioen CY, Chapman RW, Chazouilleres O, Hirschfield GM, Karlsen TH, Lohse AW, et al. Surrogate endpoints for clinical trials in primary sclerosing cholangitis: Review and results from an International PSC Study Group consensus process. Hepatology 2016;63:1357–1367. [DOI] [PubMed] [Google Scholar]
- [113].DeFilipp Z, Bloom PP, Torres Soto M, Mansour MK, Sater MRA, Huntley MH, et al. Drug-Resistant E. coli Bacteremia Transmitted by Fecal Microbiota Transplant. N Engl J Med 2019;381:2043–2050. [DOI] [PubMed] [Google Scholar]
- [114].Greenblum S, Carr R, Borenstein E. Extensive strain-level copy-number variation across human gut microbiome species. Cell 2015;160:583–594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [115].Johnson AJ, Vangay P, Al-Ghalith GA, Hillmann BM, Ward TL, Shields-Cutler RR, et al. Daily Sampling Reveals Personalized Diet-Microbiome Associations in Humans. Cell Host Microbe 2019;25:789–802 e785. [DOI] [PubMed] [Google Scholar]
- [116].Zeevi D, Korem T, Zmora N, Israeli D, Rothschild D, Weinberger A, et al. Personalized Nutrition by Prediction of Glycemic Responses. Cell 2015;163:1079–1094. [DOI] [PubMed] [Google Scholar]
- [117].Ng SC, Chan FKL, Chan PKS. Screening FMT donors during the COVID-19 pandemic: a protocol for stool SARS-CoV-2 viral quantification. The Lancet Gastroenterology & Hepatology 2020;5:642–643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [118].Kassam Z, Dubois N, Ramakrishna B, Ling K, Qazi T, Smith M, et al. Donor Screening for Fecal Microbiota Transplantation. N Engl J Med 2019;381:2070–2072. [DOI] [PubMed] [Google Scholar]
- [119].Yau YK Mak, Wing Yan Joyce Lui, Nok Shun Rashid Ng, Wai Yin Rita Cheung, Choi Yan Kitty Li, Ying Lee Amy Ching, YL Jessica Chin, Miu Ling Lau, Ho Shing Louis Chan, Ka Leung Francis Chan, Kay Sheung Paul, Ng Siew Chien. High prevalence of ESBL organisms and the COVID-19 pandemic impact on donor recruitment for Fecal Microbiota Transplantation in Hong Kong. United European Gastroenterology Journal 2021;In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [120].Paramsothy S, Nielsen S, Kamm MA, Deshpande NP, Faith JJ, Clemente JC, et al. Specific Bacteria and Metabolites Associated With Response to Fecal Microbiota Transplantation in Patients With Ulcerative Colitis. Gastroenterology 2019;156:1440–1454 e1442. [DOI] [PubMed] [Google Scholar]
- [121].Ng SC, Xu Z, Mak JWY, Yang K, Liu Q, Zuo T, et al. Microbiota engraftment after faecal microbiota transplantation in obese subjects with type 2 diabetes: a 24-week, double-blind, randomised controlled trial. Gut 2021. [DOI] [PubMed] [Google Scholar]
- [122].Woodhouse CA, Patel VC, Goldenberg S, Sanchez-Fueyo A, China L, O’Brien A, et al. PROFIT, a PROspective, randomised placebo controlled feasibility trial of Faecal mIcrobiota Transplantation in cirrhosis: study protocol for a single-blinded trial. BMJ Open 2019;9:e023518. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


