Abstract
Twenty of the last one hundred years of vitamin D research have involved investigations of the brain as a target organ for this hormone. Our group was one of the first to investigate brain outcomes resulting from primarily restricting dietary vitamin D during brain development. With the advent of new molecular and neurochemical techniques in neuroscience, there has been increasing interest in the potential neuroprotective actions of vitamin D in response to a variety of adverse exposures and how this hormone could affect brain development and function. Rather than provide an exhaustive summary of this data and a listing of neurological or psychiatric conditions that vitamin D deficiency has been associated with, here, we provide an update on the actions of this vitamin in the brain and cellular processes vitamin D may be targeting in psychiatry and neurology.
Keywords: brain, development, vitamin D deficiency, neuroprotection, disease mechanisms
1. Introduction
The vitamin D receptor is part of the nuclear receptor super family containing members such as testosterone, estradiol, cortisol, progesterone, vitamin A derivative all-trans retinoic acid and the thyroid hormones [1]. These factors have important roles in the differentiation of all organs including the brain. It is now 21 years since we first suggested that vitamin D was a “possible” neurosteroid [2]. At that time, all we knew from existing research was that immunohistochemical evidences for the vitamin D receptor (VDR) had been shown in non-neuronal glial cells and that vitamin D may affect neurotrophic factor expression in vitro. There was very little knowledge about how vitamin D could exert its genomic effects in brain cells and a complete absence of any understanding about whether, like other neurosteroids, vitamin D would also have more rapid non-genomic actions. Over the last two decades, research on neurons, non-neuronal brain cells, in both human and animal brains, has confirmed not only does the brain possess the molecular machinery for vitamin D’s actions, but also that VDRs are functional, that liganded VDRs directly regulate the expression of target genes and that vitamin D in the form of 1,25-hydroxyvitamin D3, 1,25(OH)2D3 can rapidly alter ion channel function [3]. Moreover, only very recently have new epigenetic mechanisms been revealed as gene regulatory pathways that vitamin D targets to affect gene expression in the developing brain [4,5]. As a result of all this research, vitamin D can now be considered an important steroid in brain development [6]. More recently the neuroprotective functions of vitamin D have been highlighted in models of adult brain disorders. Here we integrate these data and outline the molecules and processes vitamin D appears to target in both developing and mature brains and how such actions shape behaviour and brain function.
We are now aware of the large number of non-skeletal targets for vitamin D. In brain cells, changing vitamin D status alters cytokine regulation and has been shown to affect cell differentiation, neurotrophin expression, intracellular calcium signalling, neurotransmitter release, anti-oxidant activity, anti-inflammatory actions, stress responsivity and the expression of genes/proteins important to neuron physiology [7,8]. The purpose of this short update is not to exhaustively summarise all such associations, but to bring the reader up to speed with recent findings using the very latest techniques in molecular manipulation/quantitation and cell visualisation.
With respect to brain disorders, there is now reliable epidemiology linking embryonic or neonatal vitamin D deficiency with an increased risk of neurodevelopmental disorders, such as schizophrenia [9,10], autism [11,12,13,14] and, more recently, attention deficit hyperactivity disorder (ADHD) [15,16,17]. There are also numerous studies suggesting adult vitamin D deficiency can be correlated with certain neurodegenerative conditions. This clinical epidemiological literature has been reviewed elsewhere [18,19], and we will return to this in the final section of this article. Our purpose here will be to focus on the latest preclinical studies modelling these epidemiological links and to discuss plausible biological mechanisms behind disease-relevant phenotypes.
Although we were pioneers in this area, works from a number of laboratories have firmly established the biological plausibility for how low levels of vitamin D could adversely affect how brains form and how this could lead to subtle changes in brain function and behaviour. Our task now is to discover exactly how low levels of vitamin D impair the function of specific brain cells/circuits leading to adult brain disorders and whether correcting vitamin levels and/or the processes affected by impaired vitamin D signalling can diminish phenotype/symptom severity.
2. Vitamin D Signalling in the Brain, the Basics and Controversies
Early studies reporting levels of vitamin D metabolite levels in the brain produced widely varied results. This has been to a large extent due to technical difficulties in extraction and quantification methods. The major circulatory form of vitamin D, 25-hydroxyvitamin D3, (25(OH)D3), and its active hormonal form, 1,25-hydroxyvitamin D3, 1,25(OH)2D3, have been reported to be present in brain [20,21]. Whilst the exact concentrations are debatable, they are routinely reported as far lower than blood levels. The later use of LC/MS/MS with isotope dilution provides absolute chemical identification due to its selectivity and greater sensitivity. Using this technology, one group shows 25(OH)D3 to be around (4 ng/g tissue) in rodent brain [22,23]. This group used atmospheric pressure, photoionisation which may avoid ion-suppression artifacts which may have affected earlier methods. Very recently, Fu and colleagues developed a method to measure vitamin D metabolites in the human brain and found 25(OH)D3 was measurable, but levels were far lower than those found in the blood around 0.2–0.3 ng/g tissue [24].
With respect to the VDR, despite immunohistochemical studies confirming its presence in human, chick, rat, mouse and zebrafish brains, [25,26,27,28,29,30] claims that some of the antibodies used may have been less-than-specific and have invited dispute [31,32]. Now unambiguous evidence has been provided using mass-spectrophotometry to electrophoretically resolve proteins from adult rodent brains to identify five unique VDR peptides with a confidence interval greater than 99% [33]. There is also close anatomical overlap between brain regions in rats and humans, with respect to VDR location [25,26,27,29,34,35]. Consistent close cross-species overlap in VDR distribution validates the use of rodents in modelling vitamin D-related brain outcomes.
Immunohistochemistry, mRNA and protein analyses all reveal a gradual expression of the VDR in the developing brain [33,36,37,38]. In the developing brain, the VDR is concentrated in differentiating fields, such as the ependymal surface of the lateral ventricles [39] (the greatest source of cell division in the brain), consistent with vitamin D actions as a differentiation agent. In particular, VDR expression in the nucleus of dopamine neurons which appear very early in brain development correlates tightly with the ontogeny of these neurons [35]. New genomic technologies have now allowed VDR location and activity to be assessed in animal brains. Using clustered regularly interspaced short palindromic repeats (CRISPR), Liu et al. inserted a Cre-expression sequence driven by the endogenous VDR promotor. When such animals were mated with a tdTomato reporter mouse ((Ai14; B6.Cg-Gt(ROSA)26Sor tm14 (CAG-tdTomato)Hze/J), intense tdTomato fluorescence was detected in multiple brain regions, including caudate putamen, amygdala, reticular thalamic nucleus, cortex and less intense tdTomato fluorescence in hippocampus, hypothalamus, paraventricular thalamic nucleus (PVH), dorsal raphe and bed nucleus of the stria terminalis (BNST). RNAscope confirmed the presence of VDR RNA in these same tdTomato positive neurons. However, perhaps most importantly, 1,25(OH)2D3 (5 μM) selectively depolarised the tdTomato positive but not negative neurons in the PVH, indicating vitamin D directly regulates neuronal activity, but only in VDR-expressing neurons [40].
Immunohistochemical evidences for the enzymes that convert 25(OH)D3 to the active hormonal form of vitamin D, 1,25(OH)2D3, CYP27B1, and enzymes responsible for its breakdown, CYP24A1, have been provided in the foetal [41] and adult human brain [25,42], suggesting the active hormone can be made or removed locally in human brains. The most thorough investigation of expression of the major vitamin D metabolising enzymes CYP24A1, CYP271B and CYP271A in brain cell types was undertaken by Landel and colleagues [43]. Using primary cultures of neurons, astrocytes, microglia and oligodendrocytes (encompassing all major brain cell types), this group showed a broad distribution across all cell types but at a lower level than the kidney and liver. Strikingly, however, the addition of 1,25(OH)2D3 induced a profound upregulation of CYP24A1 only in astrocytes and microglia, a finding also previously observed [44]. Again, genomic technologies have been employed to study the activity of these enzymes. Hendrix et al. created a transgenic mouse that expresses the luciferase reporter under the control of a full-length human CYP27B1 promoter. The organ distribution of CYP27B1 luciferase activity is similar to endogenous CYP27B1 with highest activity in the kidney, testis, brain, skin and bone [45]. Unfortunately, this useful model apparently has not yet been utilised to study what factors upregulate CYP27B1 brain activity.
3. Vitamin D and Normal Brain Development
A wealth of experimental evidence exists regarding the plausibility for how altering vitamin D signalling could affect critical events in brain development, such as axonal elongation, neurotransmitter synthesis, neurotrophin production and later brain function. Although there are some comprehensive reviews in this space [46,47], here, we bring this up to date.
3.1. Vitamin D and Neurite Growth
The addition of 1,25(OH)2D3 to embryonic hippocampal neurons in culture increases neurite outgrowth possibly via an increased nerve growth factor (NGF) [48,49]. We also now have new unpublished data replicating the neurite-promoting potential of 1,25(OH)2D3 in developing dopamine neurons differentiated from (a) a neuroblastoma cell line and (b) dopamine neurons in explant mesencephalic cultures. This action is in line with most other neurosteroids. Now, we are exploring the molecular mechanisms behind these effects. Others have chosen to lesion peripheral neurons and to show that vitamin D enhances axonal repair, myelination and functional recovery [50,51]. The 1,25(OH)2D3 treatment also restores neurite outgrowth in models where it is impeded. The knockdown of the important neuronal epigenetic regulator, MeCP2, in cortical neurons blunts neurite extension potentially through reducing the activity of NFκB pathways. The 1,25(OH)2D3 restores normal outgrowth in this model [52]. Vitamin D deficiency has also been shown to reduce peripheral nerve fibre density [53], and vitamin D has been considered a potential therapeutic in spinal cord repair [54].
3.2. Vitamin D and Neurotrophic Factors
The first evidence that vitamin D had any action on brain cells came from the early studies from Didier Wion’s group in glioblastoma cells. Early studies showed that vitamin D promoted the expression of NT-3, NT-4 and nerve growth factor (NGF) [48,49,55,56,57]. Vitamin D-mediated increases in NGF were shown to be highly relevant to neuronal survival in vitro [48,58,59,60].
Given its prominent role in dopaminergic neuron differentiation (see below) and survival in earlier studies, there was a strong focus on vitamin D and neurotrophic factors important for dopaminergic neurons, such as glial cell line-derived neurotrophic factor (GDNF) [61,62] and now brain-derived neurotrophic factor (BDNF) [63]. Blocking vitamin D-mediated increase in GDNF synthesis prevents vitamin D’s trophic effects on these neurons [64]. We have described the genomic proof that vitamin D regulates the transcription of both receptors for GDNF. The 1,25(OH)2D3 suppresses GDNF family receptor alpha 1 (GFRa1), but ligand bound VDR binds to the promoter of the other major receptor for this neurotrophin, the proto-oncogene tyrosine-protein kinase receptor Ret (C-Ret), to upregulate C-Ret expression. Accordingly, the maternal absence of vitamin D decreases C-Ret expression in the developing rat mesencephalon [65].
The effect of vitamin D on neurotrophic factors in the developing brain has been far less explored. One study showed DVD-deficiency in rats reduced NGF and GDNF protein in neonatal brains [66]. Reductions in BDNF and transforming growth factor-β1 (Tgf-β1) in DVD-deficient embryonic mouse brains have also been described [67].
Most recently, there have been numerous studies showing a convincing link between hippocampal BDNF levels and vitamin D status in adult animals. Consistent with findings in development where the absence of vitamin D leads to a BDNF deficit, very recent studies show chronic treatment with 1,25(OH)2D3 induces a profound upregulation in BDNF in rat hippocampus [68]. Others show stress- or drug-induced memory deficits correspond with decreased hippocampal BDNF expression that can be restored via chronic supplementation with high doses of cholecalciferol [69,70]. Another study revealed age-induced memory deficits could be reversed with cholecalciferol supplementation with corresponding increases in hippocampal BDNF and NGF expression [71]. In a model of type 2 diabetes in mice, BDNF and phosphorylation of its downstream effector CREB are reduced in the brain, and cholecalciferol restores these and associated behavioural deficits [72].
In summarising this data, 1,25(OH)2D3 increases, and the developmental dietary absence of vitamin D reduces the expression of these crucial neurotrophic factors in neurons [73] and glia in developing and adult brains. Vitamin D’s regulation of neurotrophic factors remains a central feature in brain ontogeny.
3.3. Vitamin D Regulates the Development of Dopamine Neurons
Since we first reported the VDR within the human substantia nigra [25], we have gone on to confirm the VDR is prominent in neuromelanin containing Tyrosine Hydroxylase (TH) positive human nigral neurons [35]. Vitamin D deficiency during development leads to a reduction in specification factors crucial for dopamine neurons [74,75], alters dopamine turnover [76] and leads to decreased lateral positioning of dopamine neurons that will form the substantia nigra [75]. Importantly, similar developmental anomalies in dopamine neuron ontogeny created by other adverse developmental exposures are rescued by maternal treatment with 1,25(OH)2D3 [77]. To better understand the molecular processes involved, we have used neuroblast cells in which the VDR has been over-expressed. In such a model, we have shown 1,25(OH)2D3 increases TH mRNA [78], drives cells down a dopaminergic lineage [79] and directly targets the promoter of Catechol-o-methyl transferase (COMT), an important enzyme in brain dopamine turnover [79].
One very recent study has examined microRNAs in developing dopamine neurons as epigenetic factors in neuronal maturation. This study showed that in flow cytometry-sorted E14 dopamine neurons from DVD-deficient rat embryos, the expression of a number of microRNAs was increased and that gene-ontology analysis indicated these microRNAs were involved in neuronal differentiation. These authors then examined the functional consequences of this in developing dopamine neurons and showed that some of these microRNAs decreased neurite outgrowth [4]. This represents the first evidence in the brain indicating how vitamin D may epigenetically differentiate dopamine neurons.
Taken together, the decreased expression of dopamine-promoting neurotrophic and specification agents and delayed dopamine neuron maturation in DVD-deficient embryonic brains along with the abundant in vitro evidences for 1,25(OH)2D3 as a maturation agent for dopamine neurons, all confirm a central role for vitamin D in dopamine ontogeny. Given the links between DVD-deficiency and schizophrenia [9,10] and that dopamine abnormalities are perhaps causal in this disease, further studies examining this possible mechanistic link are needed.
4. Developmental Vitamin D Deficiency Effect on Brain Function and Behaviour
The long-term effects of DVD-deficiency on offspring behaviour have also been extensively studied in both rats and mice. Outcomes vary based on the species used/strain and duration and degree of vitamin D deficiency. Notably, there are alterations in critical early dam/pup interactions, such as maternal licking and grooming [80] and pup ultrasonic vocalisations [5,80], along with signs of delayed motor development [5]. As juveniles or adults, offspring have social behavioural deficits and alterations in stereotyped behavioural phenotypes of relevance to autism [5,80]. Interestingly, vitamin D deficiency leads to subtle increases in testosterone levels in maternal blood [81], a finding which has long-been considered a risk-modifying factor in autism [82]. Additionally, foetal male brains from vitamin D deficient dams have increased testosterone compared to control males. This study went on to show silencing of the major enzyme involved in testosterone breakdown, aromatase, by hypermethylation of its promoter in the brain may be the mechanism [81].
DVD-deficiency also leads to long-term changes in adult behaviours. Novelty [83], or exposure to psychomimetics, such as the N-methyl-D-aspartic acid receptor (NMDA-R) antagonist, MK-801 [84,85,86] or amphetamine [87], all increase locomotor activity in the adult offspring of DVD-deficient dams. Along with sensitivity to these exposures/agents that release dopamine, DVD-deficient rats have a greater response to antipsychotics that block dopamine 2 receptors [84,88]. Brain functional measures are also abnormal in these animals. Long-term potentiation, a cellular correlate of learning and memory and latent inhibition, a measure of attentional processing are also abnormal in DVD-deficient adults [89,90]. With respect to cognition, DVD-deficient offspring have impaired response inhibition, a deficit normalised by the antipsychotic clozapine [91]. Associative learning is also impaired in DVD-deficient adults [92].
Only recently have investigators begun to assess the impact of vitamin D treatment in dams exposed to either DVD-deficiency or another developmental animal model that induces brain changes, maternal immune activation. Such interventions reverse behavioural phenotypes produced by these models related to anxiety, depression cognition, stereotyped and social behaviours [93,94].
As discussed previously, given the stark differences in the effects of DVD-deficiency on brain development between rats and mice, it is not surprising that the behavioural phenotypes in adult offspring also vary widely [95]. For instance, although there are some similarities in novelty-induced hyperlocomotion [84] and perseverative responses [96], outcomes vary widely in tests of exploratory behaviour [88,97] and locomotor response to psychomimetics [98].
Whether DVD-deficiency affects cognitive performance in children is not clear. Two studies show DVD-deficient children have delayed cognitive development [99,100]. However, these findings were not replicated in much larger studies [15]. A recent meta-analysis that summarised 31 studies could find no conclusive evidence for the association between maternal or child vitamin D status and behavioural or cognitive outcomes in children and adolescents. A summary of twelve mother–child studies (n = 17,136) and five studies just in children (n = 1091) showed low maternal or child 25(OH)D3 levels led to impaired behavioural outcomes in children. In contrast, fifteen mother–child studies (n = 20,778) and eight studies in children (n = 7496) showed no association [101]. A large Finnish nested case-control study (1607 children with learning difficulties with matched controls) also showed no link between maternal 25(OH)D3 levels and behaviour [102].
One small randomised clinical trial (55 infants) in healthy term infants showed a low dose of vitamin D supplementation might benefit gross motor function compared to a higher dose of vitamin D supplementation, but this study lacked a placebo control [103]. There is also a not-insubstantial number of studies that link maternal or childhood vitamin D deficiency to neurodevelopmental psychiatric disorders such as autism, ADHD or schizophrenia [9,10,11,13,14,100,104,105]. This association is not universally replicated [106,107]; however, the mean population 25(OH)D3 levels in these later studies were much higher, making the association difficult to test. Randomised controlled trials of cholecalciferol supplementation in children with ASD or ADHD show diverse outcomes, some showed no beneficial effects and others reported some symptom relief [108,109,110,111,112,113,114]. In the future, trials examining vitamin D levels in mothers/children prior to supplementation are required as supplementing vitamin D-sufficient mothers or children is likely to have minimal effect, as shown in other clinical trials with vitamin D [115].
5. Vitamin D Is Neuroprotective in Neurons and Adult Brain
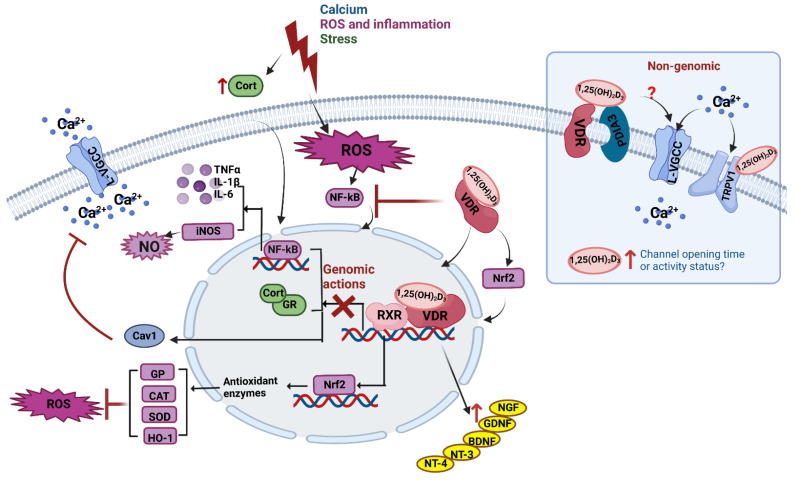
There are now sufficient epidemiological studies also linking low levels of 25OHD3 with various neurodegenerative conditions such as Alzheimer’s disease, Parkinson’s disease and multiple sclerosis. Though many such studies suffer from reverse causality (i.e., low levels of vitamin D are a consequence of disease-induced behavioural changes rather than causal), there has been sufficient interest to launch numerous clinical supplementation trials [116,117,118]. There have also been studies linking exposure to various toxins with low 25OHD3 levels. Such links have stimulated researchers to initiate mechanistic studies in neuronal cell systems and animal models. Along with the wealth of studies showing vitamin D is neuroprotective via neurotrophin production, here, we have chosen not to focus on the clinical epidemiology, but rather on vitamin D’s regulatory actions on calcium in the brain and its neuroprotective actions against reactive oxygen species (ROS) and inflammation as well as its actions in mitigating stress as plausible prophylactic/therapeutic mechanisms. We summarised the potential mechanisms underlying the action of vitamin D (Figure 1). Much of this work is very recent.
Figure 1.
Neuroprotective actions of vitamin D in brain.
Calcium-related mechanisms (in blue). Non-genomic actions. The 1,25(OH)2D3 acutely facilitates calcium influx via L-VGCC. The 1,25(OH)2D3; can also directly bind to TRPV1 to induce calcium influx. Both actions facilitate neuronal function. Genomic actions. Chronic vitamin D treatment decreases Cav1 (a L-VGCC subunit) expression which will reduce calcium influx in response to ROS/inflammation or stress.
ROS/inflammation-related mechanisms (in purple). ROS enhances NF-κB nuclear translocation to promote proinflammatory cytokine production. The 1,25(OH)2D3 acts to inhibit this nuclear translocation. By also inhibiting NF-kB expression, 1,25(OH)2D3 reduces iNOS expression and thus, reduces NO production. Finally, 1,25(OH)2D3 increases Nrf2 to enhance the expression of anti-oxidant enzymes including GP, CAT, SOD and HO-1, thus countering ROS toxicity. The 1,25(OH)2D3 also directly facilitates Nrf2 nuclear translocation.
Stress-related mechanisms (in green). Stress increases corticosterone synthesis. In the brain, corticosterone increases inflammatory cytokine production via its receptor (GR). The 1,25(OH)2D3 is neuroprotective by antagonising GR expression.
Neurotrophin-related mechanisms (in yellow). A variety of neurotrophic factors have been shown to counter the effects of stress- or toxin-induced neuronal damage. Historically, 1,25(OH)2D3 has been shown to increase NGF, GDNF, BDNF, NT-3 and NT-4 under such conditions.
5.1. Calcium Regulation
Calcium transients are required for normal neuronal function, but if calcium is unbuffered, it is toxic to brain cells. For more than 30 years, we have known how vitamin D regulates calcium uptake in bone cells [119,120] and similar mechanisms appear to act in neurons and the brain. In cultured neurons, 1,25(OH)2D3 retards calcium influx via the downregulation of L-type voltage-sensitive calcium channels, thus potentially preventing toxic outcomes [59,121,122,123]. In contrast, the rapid non-genomic actions of vitamin D produce the opposite effect, increasing calcium influx in cortical slices, a process that is again dependent on L-type calcium channels [124]. In a seminal study, the non-genomic rapid actions of 1,25(OH)2D3 were investigated in cortical neurons using calcium imaging, electrophysiology and molecular biological techniques. This study confirmed that physiological concentrations of 1,25(OH)2D3 lead to rapid calcium influx, but only in some neurons. Somatic nucleated patch recordings revealed a rapid, 1,25(OH)2D3-evoked increase in high-voltage-activated calcium currents mediated by L-type voltage-gated calcium channels [125]. Whether any of these actions are caused by the putative membrane VDR, Protein disulfide-isomerase A3 (PDIA3), in the brain remains unknown. Genetic variants in L-type voltage-gated calcium channels continue to be implicated in schizophrenia [126]. Given the epidemiological links between DVD-deficiency and schizophrenia [9,10], continued studies in this area are warranted.
Recent research also discovered that 25OHD3 and 1,25(OH)2D3 directly bind to the transient receptor potential vanilloid subfamily member 1 (TRPV1) channel [127]. Binding to the same region as the TRPV1 agonist capsaicin, 25OHD3 can weakly activate TRPV1 and inhibit capsaicin-induced TRPV1 activity. TRVP1 activity modulates immune cell activation and cytokine production through regulating intracellular calcium to mediate nociceptive signals. This, therefore, may be one mechanism for how vitamin D may modulate nociceptive pain pathways, as vitamin D deficiency has been linked to chronic pain [128], although oxidative mechanisms have also been proposed [129].
5.2. ROS and Inflammation
Vitamin D increases anti-oxidants, such as glutathione and cytochrome c, to mediate anti-oxidant actions in cultured neurons [121,130] and the brain [131,132]. Along with extracellular calcium, vitamin D deficiency in the adult brain increases ROS along with producing impairments in gamma-aminobutyric acid (GABA) and glutamate release. Importantly, reintroducing dietary vitamin D normalises all deficits [133].
In the brain, microglia are the immunologically responsive cells responsible for the production of inflammatory regulators such as nitric oxide (NO). Early studies showed 1,25(OH)2D3 blocks inducible nitric oxide synthetase in the rat brain in response to autoimmune or inflammatory factors [134,135,136]. Later studies suggested that oxidative stress may upregulate CYP27B1 in microglia to induce 1,25(OH)2D3 production locally at the site of NO or ROS production to mediate vitamin D’s anti-oxidant effects in the brain [133,137,138].
In primary cultured neurons, hypoxia induces apoptotic cell death and interferes with normal calcium signalling. One study, that chose to use cholecalciferol rather than calcitriol, showed that when added to cultured primary neurons exposed to hypoxia, cholecalciferol (10 nM) was anti-apoptotic and preserved calcium signalling though this effect was reversed at high doses. Upregulation of hypoxia-induced factor (HIF)-1α and/or BDNF were considered as the possible protective mechanisms [139], though conversion to calcitriol was not assessed.
DVD-deficiency would also appear to render the foetal environment more prone to oxidative stress. Ali and colleagues reported that DVD-deficient rat placenta produces more of the inflammatory cytokines IL-6 and 1L-1β upon challenge with a viral inflammatory agent [140]. Separately, when microglia were cultured from DVD-deficient mouse brains, they were increased in number, were hyperproliferative and had increased ROS production. Culturing these cells in the presence of calcitriol reversed these changes [141].
In recent years, there appears to have been intense interest in vitamin D’s protective actions against ROS and inflammation in the brain induced by numerous disease models. For instance, in hypothyroid juvenile rats, cholecalciferol supplementation (100 or 500 IU/kg/day) prevented hypothyroidism-induced cognitive and learning memory impairments [142]. Plausible mechanisms included elevations in the anti-oxidant enzyme superoxide dismutase (SOD) and thiol content in hippocampus and cortical tissue and reductions in malondialdehyde (MDA), a marker of oxidative stress. Similar restorative outcomes on cognition have been achieved in models of acute inflammation and again these same anti-oxidant processes in the brain were invoked by vitamin D supplementation [143].
In spontaneously hypertensive (SH) rats, infusion of 1,25(OH)2D3 into the hypothalamic paraventricular nucleus (PVN), a brain region maintaining baroreflex and autonomic function, prevented the elevation of ROS stress-related proteins such as NOX2, NOX4 and p22phox. Chronic calcitriol infusion also attenuated microglial activation and reduced tumour necrosis factor (TNF)-α, IL-1β and IL-6 inflammatory cytokine production. Likely, mechanisms involved the inhibition of the high-mobility group box-1(HMGB1)-receptor for advanced glycation end products (RAGE)/Toll-like receptor (TLR4) and NF-κB in the PVN of SH rats [144].
The effect of vitamin D on NO is also detected in a rat model of cerebral ischemia reperfusion model. Seven days of calcitriol administration prior to ischemic surgery reduced stroke-induced elevation of MDA and NO and increased total anti-oxidant capacity. These effects could be attributed to vitamin D-increasing nuclear factor erythroid 2-related factor 2(Nrf2), a transcription factor that decreases oxidative stress following stroke and/or heme oxygenase (HO-1), a major cytoprotective enzyme with anti-oxidative, and anti-inflammatory properties [145,146].
In a traumatic brain injury model, calcitriol treatment reduced MDA production and promoted autophagic flux and activated Nrf2 pathways. Autophagy reduces oxidative stress by a timely removal of damaged substances [147]. Nrf2 is a redox-sensitive transcription factor that binds to anti-oxidant response elements to promote the expression of enzymes/proteins for detoxication and anti-oxidation [148]. Confirmation that these two processes were central to calcitriols protective actions in traumatic brain injury was shown by inhibiting the autophagy using chloroquine or deleting Nrf2 as either action blocked calcitriol’s protective effects [149].
In a model of lead-induced neurotoxicity and oxidative stress, decreases in anti-oxidant molecules GSH, SOD and catalase and increased ROS production are observed in rat cortex. Cholecalciferol reversed these changes possibly via Nrf2 and/or NF-κB mechanism [150]. In a model of experimentally induced epilepsy in young male rats, cholecalciferol not only reduced seizure severity, but corrected associated memory deficits, reduced extracellular calcium, restored anti-oxidative enzymes SOD and glutathione-related enzymes and reduced inflammatory cytokine production in the hippocampus [151].
One neurological disorder commonly associated with ROS-mediated brain damage is Parkinsons’s disease (PD). The motor deficits produced by PD are believed to be caused by dopamine neurons dying selectively in the substantia nigra and the associated reduced dopamine release in the dorsal striatum. This is modelled in animals via the intracranial delivery of relatively selective dopaminergic terminal toxins, such as 6-hydroxy dopamine (6-OHDA), or nigral toxins such as 1-methyl-4-phenyltetrahydropyrine (MPTP). In a 6-OHDA-treated mouse, cholecalciferol treatment two weeks after surgical lesion attenuated 6-OHDA-induced increases in the microglia marker CD11b, IL-1β and the oxidative stress marker p47phox, a primary modulator of NADPH oxidase activity and restored some motor functional deficits [152]. Another 6-OHDA study showed that cholecalciferol prevented characteristic losses in dopamine synthetic enzymes and transporters, preserved motor function as well as reduced lipid peroxidation [153]. Another study using MPTP showed co-administration with calcitriol improved motor deficits, reduced dopamine neuron toxicity and reduced ROS production [154]. The mechanism proposed was via an interaction between the liganded VDR and poly(ADP-ribose) polymerase-1(PARP1) to reduce its contribution to ROS-induced cell death. PARP1 pathways have been proposed as one mechanism for dopamine cell death in PD.
ROS production is also linked with other degenerative processes such as Alzheimer’s disease. Animal models for Alzheimer’s disease frequently employ genetic models that over-express the Tau or amyloid (Aβ) proteins that are closely linked with disease pathology. In an amyloid presenilin model, 13 weeks of vitamin D deficiency exacerbated the ROS production in this model by downregulating superoxide dismutase 1 (SOD1), glutathione peroxidase 4 and enhanced the expression of IL-1β, IL-6 and TNFα, along with increased Aβ production and Tau phosphorylation [155].
In another amyloid model where rats are injected with Aβ1-40, cholecalciferol supplementation reduced Aβ-induced MDA levels, increased SOD activity and improved hippocampal neuronal survival [156]. Prolonged vitamin D hypovitaminosis in mice (from 6-weeks-old to 6-months-old) altered the expression of genes involved in amyloid precursor protein homeostasis (Snca, Nep, Psmb5), oxidative stress (Park7), inflammation (Casp4), lipid metabolism (Abca1), signal transduction (Gnb5) and neurogenesis (Plat) [157]. In another distinct amyloid protein mutant mouse model (3xtg-AD), vitamin D levels are dramatically reduced at 9 and 12 months of age. Vitamin D supplementation in this model improves memory possibly via the suppression of collapsin response mediator protein-2 (CRMP2) phosphorylation [158]. An in vitro study has also shown 1,25(OH)2D3 alleviates Tau hyperphosphorylation and reduces ROS in a neuronal cell model treated with Aβ possibly via vitamin D’s role in enhancing GDNF [159].
Experimentally induced autoimmune encephalitis (EAE) is a mouse model mimicking the autoimmune reaction to myelin proteins inducing multiple sclerosis-like pathology. In this model, calcitriol administration decreased the severity of EAE by attenuating inflammation and demyelination at the spinal cord [160]. This same study also showed calcitriol treatment reduces lymphocyte, macrophage and activated microglia infiltration into the brain. EAE mice have increased blood brain barrier permeability thought to be due at least in part to a reduction in endothelial tight-junction proteins such as ZO-1. This same study showed calcitriol increases this proteins expression. Calcitriol also reduced EAE-induced lipid hydroxylation and enhanced the anti-oxidant enzymes glutathione peroxidase, catalase and SOD.
The 1,25(OH)2D3 not only suppresses inflammation in models of demyelination, but also enhances the differentiation or survival of oligodendrocyte progenitor cells in the spinal cord of a MOG35–55-induced EAE mouse model [161]. The promotion of oligodendrocyte maturation by 1,25(OH)2D3 is also observed in a cuprizone-induced EAE mouse model [162]. Krabbe disease is an inherited leukodystrophy. This demyelinating condition is caused by a galactocerebrosidase (GALC) deficit that results in loss of oligodendrocytes and demyelination. In an animal model of this disorder (GALCtwi/twi; twitcher mouse), supplementing the heterozygous GALC+/− dam from birth to weaning with cholecalciferol delayed onset of disease-induced locomotor deficits and tremors and extend the life span of offspring [163].
5.3. Glucocorticoids and Stress
Glucocorticoid release is the classic endocrine response to stress, and protracted exposure induces neuronal shrinkage then cell death [164]. The effects of 1,25(OH)2D3 and glucocorticoids in the body can be considered antagonistic [165,166,167,168,169]. Similarly, in the brain, 1,25(OH)2D3 antagonises the effects of dexamethasone (a corticosterone agonist) on hippocampal neuron differentiation and glucocorticoid receptor function [170]. Interestingly, dexamethasone can decrease 1,25(OH)2D3 synthesis in the hippocampus and prefrontal cortex, indicating this process is reversible [171].
Behaviourally, vitamin D antagonises the depression-like phenotypes induced by chronic cortisol administration in animals [172,173,174,175]. Possible mechanisms include regulation of hippocampal glucocorticoid receptors or the restoration of dopamine levels in the reward centres in the brain [175]. Chronic mild stress in rats leads to increased corticosteroids, inflammatory markers and decreased anti-oxidant enzymes SOD and glutathione peroxidase and catalase in the hippocampus and prefrontal cortex. Simultaneous cholecalciferol treatment reverses these stress-mediated effects [69,176].
Chronic unpredictable stress increases immobility in a widely used test of behavioural despair (tail suspension test). Cholecalciferol treatment reduces this stress-induced immobility. Inhibiting the synthesis of the neurotransmitter serotonin abolishes cholecalciferol’s actions in this test, suggesting vitamin D might act via increased serotonin synthesis [177]. Support for this idea comes from studies in glioblastoma cells showing functional VDREs are localised at −7 kb and −10 kb upstream of the serotonin, synthesising enzyme tryptophan hydroxylase [178]. Other studies chronically administering corticosterone replicate the cholecalciferol’s reversal of these stress-induced behaviours and suggest either alterations to glucocorticoid signalling in the brain or reductions in stress-associated ROS production in the brain are the protective mechanisms [172,179]. Stress can be measured in animals using other behavioural paradigms such as immobility in a forced swim test. Chronic unpredictable stress increases immobility in this test. Cholecalciferol supplementation reverses this immobility as well as reduces serum corticosterone/ACTH levels and increases BDNF and NT-3/NT-4 levels in the hippocampus [174].
DVD-deficiency may also alter maternal response to stress in rats [180] and mice [181] and can adversely affect maternal care [80], which is well-known to induce permanent changes in offspring stress-response [182]. The translational potential of these later findings is enhanced given that a randomised clinical trial showed that 50,000 IU of cholecalciferol alone every 2 weeks, or in combination with Omega 3 fatty acids (1 g/day), for 8 weeks significantly reduced anxiety and improved sleep quality in women of reproductive age with pre-diabetes and hypovitaminosis D [183].
6. Hypervitaminosis D and Adverse CNS Outcomes
While the role of hypovitaminosis D in brain function has been extensively investigated, the effect of hypervitaminosis D has received less attention. This is despite an inverted U-shaped cellular response to 1,25(OH)2D3 being frequently reported, i.e., both high and low levels of 1,25(OH)2D3 induce adverse outcomes. Hypervitaminosis D is rare in humans, generally resulting from excess vitamin D supplementation or diseases such as sarcoidosis that produces excess 1,25(OH)2D3 due to activated macrophages [184]. Though not focused on neurological conditions, an older review concluded “There is accumulating evidence that both high and low serum calcidiol concentrations are associated with an increased risk of chronic diseases” [185]. Hypervitaminosis D always results in hypercalcaemia, which may be toxic to brain function. Animal studies showed that high cholecalciferol intake, 25,000 IU/kg, for four consecutive days reduces brainwave activity [186]. Senescence-accelerated-mouse-phenotype (SAMP) strain-8 mice, an animal model of accelerated human ageing, showed a progressive increase in serum 25OHD3, which coexists with reduced cognitive function and increased capillary permeability [187]. Deleting Fibroblast growth factor (FGF-23) in mice increased CYP27B1 levels, leading to increased 1,25(OH)2D3 synthesis. FGF-23 null mice display early ageing, and deleting CYP27B1 delayed premature ageing [188]. Considering the risk of hypervitaminosis D, serum vitamin D levels should be monitored in clinical trials of vitamin D supplementation.
7. Conclusions and Future Challenges
Here we have provided an up-to-date summary of research detailing vitamin D turnover, synthesis genomic and non-genomic actions in the brain, neurons and non-neuronal cells. We advise caution when considering much of the prior literature employing either dietary restrictions which often produced hypo-calcaemia or the use of constitutive knock out models permanently reducing 1,25(OH)2D3 synthesis or impairing VDR signalling, as these models produce too many non-CNS effects to make brain-related outcomes interpretable. Although there are still strain and species differences when choosing a model organism, contemporary DVD- or AVD-deficient models in rodents do not produce hypocalcaemic offspring and continue to produce findings of apparent relevance to the fields of psychiatry and neurology.
Increasingly, epidemiological studies associate low levels of vitamin D either prenatally or at birth with psychiatric conditions such as autism and schizophrenia. The epidemiology for degenerative diseases as diverse as Alzheimer’s disease, Parkinson’s disease and multiple sclerosis all continue to indicate a role of optimal vitamin D status throughout life. This raises the possibility of simple dietary supplementation as an adjunct to current therapies. Although not practicably possible for developmental conditions with adult onset such as schizophrenia, this could eventually be considered in early onset psychiatric disorders, such as autism or ADHD, or for maintaining neural integrity in degenerative conditions via large placebo-controlled, randomised clinical supplementation trials.
We would also like to take this opportunity to highlight design issues in many observational studies opportunistically linking early-life vitamin D deficiency with psychiatric disorders. Most of the published observational epidemiological studies linking early-life vitamin D status with a psychiatric diagnosis never address reverse causality (the condition changes behaviours that lead to less sun exposure). By way of illustration, a very high-profile recent report in the New England Journal of Medicine showed all mental illnesses were associated with an increased risk in a general medical condition [189]. In other words, patients with psychiatric conditions are generally suffering from other conditions that will curtail behaviour perhaps altering diet, exercise and exposure to sunshine. We urge all future epidemiological studies that seek to examine the relationship between vitamin D and psychiatric or neurological conditions to rigorously control for the often-poor general health of patients. This, of course, is less of an issue for gestational (DVD-deficiency) exposures in otherwise healthy mothers.
Important data have emerged from a recent mendelian randomisation study examining gene pathways related to 25(OH)D3 blood concentrations. This study found no evidence that genetic factors involved in the synthesis of 25(OH)D3 were causal for psychiatric disorders [190]. We have interpreted this to mean any link between 25(OH)D3 levels and brain-related outcomes are likely to be solely driven by environmental factors.
Autism is perhaps the developmental brain disorder most regularly linked with DVD-deficiency. However, well-conducted studies refuting this link are now emerging. For the studies describing an inverse relationship between maternal vitamin D levels and autism, they all had mean 25(OH)D3 levels of <50 nM which is considered by some authors to represent a cut off for vitamin D deficiency [11,12,13,191]. Two recent studies have failed to find this inverse association [106,192]. So, at face value, this appears a failure to replicate previous studies. However, it is crucial to note that in these last two studies, the mean levels of 25(OH)D3 were actually very high (>70–80 nM) and there were very few individuals that were actually vitamin D-deficient, meaning the association could not be properly tested. These same six studies all used the same laboratory to analyse samples, so technical bias (so common amongst vitamin D studies in different populations) could be ruled out. This suggests a threshold effect rather than any continuous relationship between DVD-deficiency and autism. We highlight this particular relationship to illustrate some of the confusion regarding statements regarding potential causality between vitamin D and various brain-related clinical disorders.
The incidence of hypovitaminosis D in both pregnant women and their newborns and the general population remains concerning [193]. Clearly, more rigorous study design is required taking into consideration such issues to bring clarity to the future epidemiological studies. Better quality studies in the future are needed given the substantial emotional and financial burden psychiatric and neurological disorders place on the patient and community. The opportunity to use such a simple, safe and inexpensive intervention as vitamin D supplementation as treatment or as an adjunct to existing therapies in such disorders remains extremely attractive from a public health perspective.
Acknowledgments
We thank Suzy Alexander for assistance creating the figure.
Abbreviations
VDR, vitamin D receptor; PDIA3, Protein disulfide-isomerase A3; RXR, retinoid X receptor; L-VGCC, L-type voltage-gated calcium channel; Cav1, L-type voltage-gated calcium subunit 1; TRPV1, transient receptor potential vanilloid subfamily member 1; ROS, reactive oxygen species; iNOS, inducible nitric oxide synthetase; NO, nitric oxide; NF-kB, nuclear factor kappa-B; Nrf2, nuclear factor erythroid 2-related factor 2; GP, glutathione peroxidase; CAT, catalase; SOD, superoxide dismutase; HO-1, heme oxygenase; NGF, nerve growth factor; BDNF, brain-derived neurotrophic factor; GDNF, glial cell line-derived neurotrophic factor; NT-3, neurotrophin 3; NT-4, neurotrophin 4; GR, glucocorticoid receptor; CORT, corticosterone; TNFα, tumour necrosis factor α; IL-6, interlukin-6; IL-1B, interlukin-1 Beta. (Figure created from BioRender.com).
Author Contributions
D.W.E. and X.C. conceived and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors have no conflicts of interest.
Funding Statement
This research was funded by the National Health and Medical Research Council (APPS 1124721 and 1141699) and the Queensland State Government.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Evans R.M. The nuclear receptor superfamily: A rosetta stone for physiology. Mol. Endocrinol. 2005;19:1429–1438. doi: 10.1210/me.2005-0046. [DOI] [PubMed] [Google Scholar]
- 2.McGrath J., Feron F., Eyles D. Vitamin D: The neglected neurosteroid? Trends Neurosci. 2001;24:570–572. doi: 10.1016/S0166-2236(00)01949-4. [DOI] [PubMed] [Google Scholar]
- 3.Cui X., Gooch H., Petty A., McGrath J.J., Eyles D. Vitamin D and the brain: Genomic and non-genomic actions. Mol. Cell. Endocrinol. 2017;453:131–143. doi: 10.1016/j.mce.2017.05.035. [DOI] [PubMed] [Google Scholar]
- 4.Pertile R., Kiltschewskij D.G., Geaghan M., Barnett M., Cui X., Cairns M.J., Eyles D.W. Developmental vitamin D-deficiency increases the expression of microRNAs involved in dopamine neuron development. Brain Res. 2022;1789:147953. doi: 10.1016/j.brainres.2022.147953. [DOI] [PubMed] [Google Scholar]
- 5.Ali A., Vasileva S., Langguth M., Alexander S., Cui X., Whitehouse A., McGrath J.J., Eyles D. Deveopmental vitamin D deficiency produces behavioral phenotypes of relevance to autism in an animal model. Nutrients. 2019;11:1187. doi: 10.3390/nu11051187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Melcangi R.C., Panzica G. Neuroactive steroids: An update of their roles in central and peripheral nervous system. Psychoneuroendocrinology. 2009;34:S1–S8. doi: 10.1016/j.psyneuen.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 7.Eyles D.W., Burne T.H., McGrath J.J. Vitamin D, effects on brain development, adult brain function and the links between low levels of vitamin D and neuropsychiatric disease. Front. Neuroendocrinol. 2013;34:47–64. doi: 10.1016/j.yfrne.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 8.McCann J.C., Ames B.N. Is there convincing biological or behavioral evidence linking vitamin D deficiency to brain dysfunction? FASEB J. 2008;22:982–1001. doi: 10.1096/fj.07-9326rev. [DOI] [PubMed] [Google Scholar]
- 9.McGrath J.J., Eyles D.W., Pedersen C.B., Anderson C., Ko P., Burne T.H., Norgaard-Pedersen B., Hougaard D.M., Mortensen P.B. Neonatal vitamin D status and risk of schizophrenia: A population-based case-control study. Arch. Gen. Psychiatry. 2010;67:889–894. doi: 10.1001/archgenpsychiatry.2010.110. [DOI] [PubMed] [Google Scholar]
- 10.Eyles D.W., Trzaskowski M., Vinkhuyzen A.A.E., Mattheisen M., Meier S., Gooch H., Anggono V., Cui X., Tan M.C., Burne T.H.J., et al. The association between neonatal vitamin D status and risk of schizophrenia. Sci. Rep. 2018;8:17692. doi: 10.1038/s41598-018-35418-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vinkhuyzen A.A.E., Eyles D.W., Burne T.H.J., Blanken L.M.E., Kruithof C.J., Verhulst F., Jaddoe V.W., Tiemeier H., McGrath J.J. Gestational vitamin D deficiency and autism-related traits: The Generation R study. Mol. Psychiatry. 2016;23:240–246. doi: 10.1038/mp.2016.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vinkhuyzen A., Eyles D., Burne T., Blanken L., Kruithof C., Verhulst F., White T., Jaddoe V.W., Tiemeier H., McGrath J. Gestational Vitamin D Deficiency and Autism Spectrum Disorder. Br. J. Psychiatry Open. 2017;3:85–90. doi: 10.1192/bjpo.bp.116.004077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee B.K., Eyles D.W., Magnusson C., Newschaffer C.J., McGrath J.J., Kvaskoff D., Ko P., Dalman C., Karlsson H., Gardner R.M. Developmental vitamin D and autism spectrum disorders: Findings from the Stockholm Youth Cohort. Mol. Psychiatry. 2019;26:1578–1588. doi: 10.1038/s41380-019-0578-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sourander A., Upadhyaya S., Surcel H.M., Hinkka-Yli-Salomaki S., Cheslack-Postava K., Silwal S., Sucksdorff M., McKeague I.W., Brown A.S. Maternal Vitamin D Levels During Pregnancy and Offspring Autism Spectrum Disorder. Biol. Psychiatry. 2021;90:790–797. doi: 10.1016/j.biopsych.2021.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Strøm M., Halldorsson T., Hansen S., Granström C., Maslova E., Petersen S.B., Cohen A.S., Olsen S.F. Vitamin D measured in maternal serum and offspring neurodevelopmental outcomes: A prospective study with long-term follow-up. Ann. Nutr. Metab. 2014;64:254–261. doi: 10.1159/000365030. [DOI] [PubMed] [Google Scholar]
- 16.Morales E., Julvez J., Torrent M., Ballester F., Rodríguez-Bernal C.L., Andiarena A., Vegas O., Castilla A.M., Rodriguez-Dehli C., Tardón A., et al. Vitamin D in Pregnancy and Attention Deficit Hyperactivity Disorder-like Symptoms in Childhood. Epidemiology. 2015;26:458–465. doi: 10.1097/EDE.0000000000000292. [DOI] [PubMed] [Google Scholar]
- 17.Arns M., van der Heijden K.B., Arnold L.E., Kenemans J.L. Geographic variation in the prevalence of ateention-deficit/hyperactivity disorder: The sunny perspective. Biol. Psychiatry. 2013;74:585–590. doi: 10.1016/j.biopsych.2013.02.010. [DOI] [PubMed] [Google Scholar]
- 18.Groves N., McGrath J., Burne T. Adult vitamin D deficiency and adverse Brain Outcomes. In: Feldman D., editor. Vitamin D Vol 2 Health, Disease and Therapeutics. 4th ed. Volume 1. Elsevier; London, UK: 2018. pp. 1147–1158. [Google Scholar]
- 19.Cui X., McGrath J.J., Burne T.H.J., Eyles D.W. Vitamin D and schizophrenia: 20 years on. Mol. Psychiatry. 2021;26:2708–2720. doi: 10.1038/s41380-021-01025-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Holmoy T., Moen S.M., Gundersen T.A., Holick M.F., Fainardi E., Castellazzi M., Casetta I. 25-hydroxyvitamin D in cerebrospinal fluid during relapse and remission of multiple sclerosis. Mult. Scler. 2009;15:1280–1285. doi: 10.1177/1352458509107008. [DOI] [PubMed] [Google Scholar]
- 21.Balabanova S., Richter H.P., Antoniadis G., Homoki J., Kremmer N., Hanle J., Teller W.M. 25-Hydroxyvitamin D, 24, 25-dihydroxyvitamin D and 1,25-dihydroxyvitamin D in human cerebrospinal fluid. Klin. Wochenschr. 1984;62:1086–1090. doi: 10.1007/BF01711378. [DOI] [PubMed] [Google Scholar]
- 22.Ahonena L., Maireb F.B.R., Savolainenc S., Koprac J., Vreekenb R.J., Hankemeierb T., Myöhänenc T., Kyllia P., Kostiainena R. Analysis of oxysterols and vitamin D metabolites in mouse brain and cell line samples by ultra-high-performance liquid chromatography-atmospheric pressure photoionization–mass spectrometry. J. Chromatogr. A. 2014;1364:214–222. doi: 10.1016/j.chroma.2014.08.088. [DOI] [PubMed] [Google Scholar]
- 23.Xue Y., He X., Li H.-D., Deng Y., Yan M., Cai H.-L., Tang M.-M., Dang R.-L., Jiang P. Simultaneous quantification of 25-Hydroxyvitamin D3 and 24,25-dihydroxyvitamin D3 in rats shows strong correlations between serum and brain tissue levels. Int. J. Endocrinol. 2015;2015:296531. doi: 10.1155/2015/296531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fu X., Dolnikowski G.G., Patterson W.B., Dawson-Hughes B., Zheng T., Morris M.C., Holland T.M., Booth S.L. Determination of Vitamin D and Its Metabolites in Human Brain Using an Ultra-Pressure LC-Tandem Mass Spectra Method. Curr. Dev. Nutr. 2019;3:nzz074. doi: 10.1093/cdn/nzz074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Eyles D.W., Smith S., Kinobe R., Hewison M., McGrath J.J. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat. 2005;29:21–30. doi: 10.1016/j.jchemneu.2004.08.006. [DOI] [PubMed] [Google Scholar]
- 26.Prufer K., Jirikowski G.F. 1.25-Dihydroxyvitamin D3 receptor is partly colocalized with oxytocin immunoreactivity in neurons of the male rat hypothalamus. Cell. Mol. Biol. 1997;43:543–548. [PubMed] [Google Scholar]
- 27.Clemens T.L., McGlade S.A., Garrett K.P., Horiuchi N., Hendy G.N. Tissue-specific regulation of avian vitamin D-dependent calcium-binding protein 28-kDa mRNA by 1,25-dihydroxyvitamin D3. J. Biol. Chem. 1988;263:13112–13116. doi: 10.1016/S0021-9258(18)37678-6. [DOI] [PubMed] [Google Scholar]
- 28.Prufer K., Veenstra T.D., Jirikowski G.F., Kumar R. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the rat brain and spinal cord. J. Chem. Neuroanat. 1999;16:135–145. doi: 10.1016/S0891-0618(99)00002-2. [DOI] [PubMed] [Google Scholar]
- 29.Walbert T., Jirikowski G.F., Prufer K. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the limbic system of the rat. Horm. Metab. Res. 2001;33:525–531. doi: 10.1055/s-2001-17210. [DOI] [PubMed] [Google Scholar]
- 30.Craig T.A., Sommer S., Sussman C.R., Grande J.P., Kumar R. Expression and regulation of the vitamin D receptor in the zebrafish, Danio rerio. J. Bone Miner. Res. 2008;23:1486–1496. doi: 10.1359/jbmr.080403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang Y., Becklund B.R., DeLuca H.F. Identification of a highly specific and versatile vitamin D receptor antibody. Arch. Biochem. Biophys. 2010;494:166–177. doi: 10.1016/j.abb.2009.11.029. [DOI] [PubMed] [Google Scholar]
- 32.Wang Y., DeLuca H.F. Is the vitamin D receptor found in muscle? Endocrinology. 2011;152:354–363. doi: 10.1210/en.2010-1109. [DOI] [PubMed] [Google Scholar]
- 33.Eyles D.W., Liu P.Y., Josh P., Cui X. Intracellular distribution of the vitamin D receptor in the brain: Comparison with classic target tissues and redistribution with development. Neuroscience. 2014;268:1–9. doi: 10.1016/j.neuroscience.2014.02.042. [DOI] [PubMed] [Google Scholar]
- 34.Stumpf W.E., O’Brien L.P. 1,25 (OH)2 vitamin D3 sites of action in the brain. An autoradiographic study. Histochemistry. 1987;87:393–406. doi: 10.1007/BF00496810. [DOI] [PubMed] [Google Scholar]
- 35.Cui X., Pelekanos M., Liu P.Y., Burne T.H.J., McGrath J.J., Eyles D. The vitamin D receptor in dopamine neurons; its presence in human substantia nigra and its ortogenesis in rat midbrain. Neuroscience. 2013;236:77–87. doi: 10.1016/j.neuroscience.2013.01.035. [DOI] [PubMed] [Google Scholar]
- 36.Veenstra T.D., Prufer K., Koenigsberger C., Brimijoin S.W., Grande J.P., Kumar R. 1,25-Dihydroxyvitamin D3 receptors in the central nervous system of the rat embryo. Brain Res. 1998;804:193–205. doi: 10.1016/S0006-8993(98)00565-4. [DOI] [PubMed] [Google Scholar]
- 37.Burkert R., McGrath J., Eyles D. Vitamin D receptor expression in the embryonic rat brain. Neurosci. Res. Commun. 2003;33:63–71. doi: 10.1002/nrc.10081. [DOI] [Google Scholar]
- 38.Erben R.G., Soegiarto D.W., Weber K., Zeitz U., Lieberherr M., Gniadecki R., Moller G., Adamski J., Balling R. Deletion of deoxyribonucleic acid binding domain of the vitamin D receptor abrogates genomic and nongenomic functions of vitamin D. Mol. Endocrinol. 2002;16:1524–1537. doi: 10.1210/mend.16.7.0866. [DOI] [PubMed] [Google Scholar]
- 39.Cui X., McGrath J.J., Burne T.H., Mackay-Sim A., Eyles D.W. Maternal vitamin D depletion alters neurogenesis in the developing rat brain. Int. J. Dev. Neurosci. 2007;25:227–232. doi: 10.1016/j.ijdevneu.2007.03.006. [DOI] [PubMed] [Google Scholar]
- 40.Liu H., He Y., Beck J., da Silva Teixeira S., Harrison K., Xu Y., Sisley S. Defining vitamin D receptor expression in the brain using a novel VDR(Cre) mouse. J. Comp. Neurol. 2021;529:2362–2375. doi: 10.1002/cne.25100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fu G.K., Lin D., Zhang M.Y., Bikle D.D., Shackleton C.H., Miller W.L., Portale A.A. Cloning of human 25-hydroxyvitamin D-1 alpha-hydroxylase and mutations causing vitamin D-dependent rickets type 1. Mol. Endocrinol. 1997;11:1961–1970. doi: 10.1210/mend.11.13.0035. [DOI] [PubMed] [Google Scholar]
- 42.Zehnder D., Bland R., Williams M.C., McNinch R.W., Howie A.J., Stewart P.M., Hewison M. Extrarenal expression of 25-hydroxyvitamin d(3)-1 alpha-hydroxylase. J. Clin. Endocrinol. Metab. 2001;86:888–894. doi: 10.1210/jcem.86.2.7220. [DOI] [PubMed] [Google Scholar]
- 43.Landel V., Stephan D., Cui X., Eyles D., Feron F. Differential expression of vitamin D-associated enzymes and receptors in brain cell subtypes. J. Steroid Biochem. Mol. Biol. 2018;177:129–134. doi: 10.1016/j.jsbmb.2017.09.008. [DOI] [PubMed] [Google Scholar]
- 44.Naveilhan P., Neveu I., Baudet C., Ohyama K.Y., Brachet P., Wion D. Expression of 25(OH) vitamin D3 24-hydroxylase gene in glial cells. Neuroreport. 1993;5:255–257. doi: 10.1097/00001756-199312000-00018. [DOI] [PubMed] [Google Scholar]
- 45.Hendrix I., Anderson P., May B., Morris H. Regulation of gene expression by the CYP27B1 promoter-study of a transgenic mouse model. J. Steroid Biochem. Mol. Biol. 2004;89:139–142. doi: 10.1016/j.jsbmb.2004.03.093. [DOI] [PubMed] [Google Scholar]
- 46.Eyles D.W., McGrath J.J. Vitamin D brain development and function. In: Feldman D., editor. Vitamin D Vol 1 Biochemistry, Physiology and Diagnostics. Volume 1. Elsevier; London, UK: 2018. pp. 563–581. [Google Scholar]
- 47.Eyles D.W. Vitamin D: Brain and Behavior. JBMR Plus. 2021;5:e10419. doi: 10.1002/jbm4.10419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Brown J., Bianco J.I., McGrath J.J., Eyles D.W. 1,25-dihydroxyvitamin D3 induces nerve growth factor, promotes neurite outgrowth and inhibits mitosis in embryonic rat hippocampal neurons. Neurosci. Lett. 2003;343:139–143. doi: 10.1016/S0304-3940(03)00303-3. [DOI] [PubMed] [Google Scholar]
- 49.Marini F., Bartoccini E., Cascianelli G., Voccoli V., Baviglia M.G., Magni M.V., Garcia-Gil M., Albi E. Effect of 1alpha,25-dihydroxyvitamin D3 in embryonic hippocampal cells. Hippocampus. 2010;20:696–705. doi: 10.1002/hipo.20670. [DOI] [PubMed] [Google Scholar]
- 50.Horst R.L., Napoli J.L., Littledike E.T. Discrimination in the metabolism of orally dosed ergocalciferol and cholecalciferol by the pig, rat and chick. Biochem. J. 1982;204:185–189. doi: 10.1042/bj2040185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chabas J.F., Alluin O., Rao G., Garcia S., Lavaut M.N., Risso J.J., Legre R., Magalon G., Khrestchatisky M., Marqueste T., et al. Vitamin D2 potentiates axon regeneration. J. Neurotrauma. 2008;25:1247–1256. doi: 10.1089/neu.2008.0593. [DOI] [PubMed] [Google Scholar]
- 52.Ribeiro M.C., Moore S.M., Kishi N., Macklis J.D., MacDonald J.L. Vitamin D Supplementation Rescues Aberrant NF-kappaB Pathway Activation and Partially Ameliorates Rett Syndrome Phenotypes in Mecp2 Mutant Mice. eNeuro. 2020;7 doi: 10.1523/ENEURO.0167-20.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tague S.E., Smith P.G. Vitamin D deficiency leads to sensory and sympathetic denervation of the rat synovium. Neuroscience. 2014;279:77–93. doi: 10.1016/j.neuroscience.2014.08.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Feron F., Marqueste T., Bianco J., Gueye Y., Chabas J.F., Decherchi P. Repairing the spinal cord with vitamin D: A promising strategy. Biol. Aujourdhui. 2014;208:69–75. doi: 10.1051/jbio/2014008. [DOI] [PubMed] [Google Scholar]
- 55.Neveu I., Naveilhan P., Jehan F., Baudet C., Wion D., De Luca H.F., Brachet P. 1,25-dihydroxyvitamin D3 regulates the synthesis of nerve growth factor in primary cultures of glial cells. Brain Res. Mol. Brain Res. 1994;24:70–76. doi: 10.1016/0169-328X(94)90119-8. [DOI] [PubMed] [Google Scholar]
- 56.Wion D., MacGrogan D., Neveu I., Jehan F., Houlgatte R., Brachet P. 1,25-Dihydroxyvitamin D3 is a potent inducer of nerve growth factor synthesis. J. Neurosci. Res. 1991;28:110–114. doi: 10.1002/jnr.490280111. [DOI] [PubMed] [Google Scholar]
- 57.Neveu I., Naveilhan P., Baudet C., Brachet P., Metsis M. 1,25-dihydroxyvitamin D3 regulates NT-3, NT-4 but not BDNF mRNA in astrocytes. Neuroreport. 1994;6:124–126. doi: 10.1097/00001756-199412300-00032. [DOI] [PubMed] [Google Scholar]
- 58.Dursun E., Gezen-Ak D., Yilmazer S. A novel perspective for Alzheimer’s disease: Vitamin D receptor suppression by amyloid-beta and preventing the amyloid-beta induced alterations by vitamin D in cortical neurons. J. Alzheimers. Dis. 2011;23:207–219. doi: 10.3233/JAD-2010-101377. [DOI] [PubMed] [Google Scholar]
- 59.Gezen-Ak D., Dursun E., Yilmazer S. The Effects of Vitamin D Receptor Silencing on the Expression of LVSCC-A1C and LVSCC-A1D and the Release of NGF in Cortical Neurons. PLoS ONE. 2011;6:e17553. doi: 10.1371/journal.pone.0017553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Saporito M.S., Wilcox H.M., Hartpence K.C., Lewis M.E., Vaught J.L., Carswell S. Pharmacological induction of nerve growth factor mRNA in adult rat brain. Exp. Neurol. 1993;123:295–302. doi: 10.1006/exnr.1993.1162. [DOI] [PubMed] [Google Scholar]
- 61.Granholm A.C., Reyland M., Albeck D., Sanders L., Gerhardt G., Hoernig G., Shen L., Westphal H., Hoffer B. Glial cell line-derived neurotrophic factor is essential for postnatal survival of midbrain dopamine neurons. J. Neurosci. 2000;20:3182–3190. doi: 10.1523/JNEUROSCI.20-09-03182.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Oo T.F., Burke R.E. The time course of developmental cell death in phenotypically defined dopaminergic neurons of the substantia nigra. Brain Res. Dev. Brain Res. 1997;98:191–196. doi: 10.1016/S0165-3806(96)00173-3. [DOI] [PubMed] [Google Scholar]
- 63.Shirazi H.A., Rasouli J., Ciric B., Rostami A., Zhang G.X. 1,25-Dihydroxyvitamin D3 enhances neural stem cell proliferation and oligodendrocyte differentiation. Exp. Mol. Pathol. 2015;98:240–245. doi: 10.1016/j.yexmp.2015.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Orme R.P., Bhangal M.S., Fricker R.A. Calcitriol imparts neuroprotection in vitro to midbrain dopaminergic neurons by upregulating GDNF expression. PLoS ONE. 2013;23:e62040. doi: 10.1371/journal.pone.0062040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Pertile R., Cui X., Hammond L.A., Eyles D.W. Vitamin D regulation of GDNF/Ret signaling in dopaminergic neurons. FASEB J. 2018;32:819–828. doi: 10.1096/fj.201700713R. [DOI] [PubMed] [Google Scholar]
- 66.Eyles D., Brown J., Mackay-Sim A., McGrath J., Feron F. Vitamin D3 and brain development. Neuroscience. 2003;118:641–653. doi: 10.1016/S0306-4522(03)00040-X. [DOI] [PubMed] [Google Scholar]
- 67.Hawes J.E., Tesic D., Whitehouse A.J., Zosky G.R., Smith J.T., Wyrwoll C.S. Maternal vitamin D deficiency alters fetal brain development in the BALB/c mouse. Behav. Brain Res. 2015;286:192–200. doi: 10.1016/j.bbr.2015.03.008. [DOI] [PubMed] [Google Scholar]
- 68.Abdollahzadeh M., Panahpour H., Ghaheri S., Saadati H. Calcitriol supplementation attenuates cisplatin-induced behavioral and cognitive impairments through up-regulation of BDNF in male rats. Brain Res. Bull. 2022;181:21–29. doi: 10.1016/j.brainresbull.2022.01.006. [DOI] [PubMed] [Google Scholar]
- 69.Bakhtiari-Dovvombaygi H., Izadi S., Zare M., Asgari Hassanlouei E., Dinpanah H., Ahmadi-Soleimani S.M., Beheshti F. Vitamin D3 administration prevents memory deficit and alteration of biochemical parameters induced by unpredictable chronic mild stress in rats. Sci. Rep. 2021;11:16271. doi: 10.1038/s41598-021-95850-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Mansouri F., Ghanbari H., Marefati N., Arab Z., Salmani H., Beheshti F., Hosseini M. Protective effects of vitamin D on learning and memory deficit induced by scopolamine in male rats: The roles of brain-derived neurotrophic factor and oxidative stress. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2021;394:1451–1466. doi: 10.1007/s00210-021-02062-w. [DOI] [PubMed] [Google Scholar]
- 71.Bayat M., Kohlmeier K.A., Haghani M., Haghighi A.B., Khalili A., Bayat G., Hooshmandi E., Shabani M. Co-treatment of vitamin D supplementation with enriched environment improves synaptic plasticity and spatial learning and memory in aged rats. Psychopharmacology. 2021;238:2297–2312. doi: 10.1007/s00213-021-05853-4. [DOI] [PubMed] [Google Scholar]
- 72.Tan X., Gao L., Cai X., Zhang M., Huang D., Dang Q., Bao L. Vitamin D3 alleviates cognitive impairment through regulating inflammatory stress in db/db mice. Food Sci. Nutr. 2021;9:4803–4814. doi: 10.1002/fsn3.2397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Manjari S.K.V., Maity S., Poornima R., Yau S.Y., Vaishali K., Stellwagen D., Komal P. Restorative Action of Vitamin D3 on Motor Dysfunction Through Enhancement of Neurotrophins and Antioxidant Expression in the Striatum. Neuroscience. 2022;492:67–81. doi: 10.1016/j.neuroscience.2022.03.039. [DOI] [PubMed] [Google Scholar]
- 74.Cui X., Pelekanos M., Burne T.H., McGrath J.J., Eyles D.W. Maternal vitamin D deficiency alters the expression of genes involved in dopamine specification in the developing rat mesencephalon. Neurosci. Lett. 2010;486:220–223. doi: 10.1016/j.neulet.2010.09.057. [DOI] [PubMed] [Google Scholar]
- 75.Luan W., Hammond L.A., Cotter E., Osborne G.W., Alexander S.A., Nink V., Cui X., Eyles D.W. Developmental Vitamin D (DVD) Deficiency Reduces Nurr1 and TH Expression in Post-mitotic Dopamine Neurons in Rat Mesencephalon. Mol. Neurobiol. 2018;55:2243–2453. doi: 10.1007/s12035-017-0497-3. [DOI] [PubMed] [Google Scholar]
- 76.Kesby J.P., Cui X., Ko P., McGrath J.J., Burne T.H., Eyles D.W. Developmental vitamin D deficiency alters dopamine turnover in neonatal rat forebrain. Neurosci. Lett. 2009;461:155–158. doi: 10.1016/j.neulet.2009.05.070. [DOI] [PubMed] [Google Scholar]
- 77.Luan W., Hammond L.A., Vuillermot S., Meyer U., Eyles D.W. Maternal vitamin D prevents abnormal dopaminergic development and function in a mouse model of prenatal immune activation. Sci. Rep. 2018;8:9741. doi: 10.1038/s41598-018-28090-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Cui X., Pertile R., Liu P., Eyles D.W. Vitamin D regulates tyrosine hydroxylase expression: N-cadherin a possible mediator. Neuroscience. 2015;304:90–100. doi: 10.1016/j.neuroscience.2015.07.048. [DOI] [PubMed] [Google Scholar]
- 79.Pertile R.A.N., Cui X., Eyles D.W. Vitamin D signalling and the differentiation of developing dopamine systems. Neuroscience. 2016;333:193–203. doi: 10.1016/j.neuroscience.2016.07.020. [DOI] [PubMed] [Google Scholar]
- 80.Yates N.J., Tesic D., Feindel K.W., Smith J.T., Clarke M.W., Wale C., Crew R.C., Wharfe M., Whitehouse A.J., Wyrwoll C.S. Vitamin D is crucial for maternal care and offspring social behaviour in rats. J. Endocrinol. 2018;237:73–85. doi: 10.1530/JOE-18-0008. [DOI] [PubMed] [Google Scholar]
- 81.Ali A.A., Cui X., Pertile R.A.N., Li X., Medley G., Alexander S.A., Whitehouse A.J.O., McGrath J.J., Eyles D.W. Developmental vitamin D deficiency increases foetal exposure to testosterone. Mol. Autism. 2020;11:96. doi: 10.1186/s13229-020-00399-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Xu X.J., Shou X.J., Li J., Jia M.X., Zhang J.S., Guo Y., Wei Q.Y., Zhang X.T., Han S.P., Zhang R., et al. Mothers of autistic children: Lower plasma levels of oxytocin and Arg-vasopressin and a higher level of testosterone. PLoS ONE. 2013;8:e74849. doi: 10.1371/journal.pone.0074849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Burne T.H., Becker A., Brown J., Eyles D.W., Mackay-Sim A., McGrath J.J. Transient prenatal Vitamin D deficiency is associated with hyperlocomotion in adult rats. Behav. Brain Res. 2004;154:549–555. doi: 10.1016/j.bbr.2004.03.023. [DOI] [PubMed] [Google Scholar]
- 84.Kesby J.P., Burne T.H., McGrath J.J., Eyles D.W. Developmental vitamin D deficiency alters MK 801-induced hyperlocomotion in the adult rat: An animal model of schizophrenia. Biol. Psychiatry. 2006;60:591–596. doi: 10.1016/j.biopsych.2006.02.033. [DOI] [PubMed] [Google Scholar]
- 85.Kesby J.P., O’Loan J.C., Alexander S., Deng C., Huang X.F., McGrath J.J., Eyles D.W., Burne T.H. Developmental vitamin D deficiency alters MK-801-induced behaviours in adult offspring. Psychopharmacology. 2012;220:455–463. doi: 10.1007/s00213-011-2492-0. [DOI] [PubMed] [Google Scholar]
- 86.O’Loan J., Eyles D.W., Kesby J., Ko P., McGrath J.J., Burne T.H. Vitamin D deficiency during various stages of pregnancy in the rat; its impact on development and behaviour in adult offspring. Psychoneuroendocrinology. 2007;32:227–234. doi: 10.1016/j.psyneuen.2006.12.006. [DOI] [PubMed] [Google Scholar]
- 87.Kesby J.P., Cui X., O’Loan J., McGrath J.J., Burne T.H., Eyles D.W. Developmental vitamin D deficiency alters dopamine-mediated behaviors and dopamine transporter function in adult female rats. Psychopharmacology. 2010;208:159–168. doi: 10.1007/s00213-009-1717-y. [DOI] [PubMed] [Google Scholar]
- 88.Becker A., Grecksch G. Pharmacological treatment to augment hole board habituation in prenatal Vitamin D-deficient rats. Behav. Brain Res. 2006;166:177–183. doi: 10.1016/j.bbr.2005.07.010. [DOI] [PubMed] [Google Scholar]
- 89.Grecksch G., Ruthrich H., Hollt V., Becker A. Transient prenatal vitamin D deficiency is associated with changes of synaptic plasticity in the dentate gyrus in adult rats. Psychoneuroendocrinology. 2009;34:S258–S264. doi: 10.1016/j.psyneuen.2009.07.004. [DOI] [PubMed] [Google Scholar]
- 90.Becker A., Eyles D.W., McGrath J.J., Grecksch G. Transient prenatal vitamin D deficiency is associated with subtle alterations in learning and memory functions in adult rats. Behav. Brain Res. 2005;161:306–312. doi: 10.1016/j.bbr.2005.02.015. [DOI] [PubMed] [Google Scholar]
- 91.Turner K.M., Young J.W., McGrath J.J., Eyles D.W., Burne T.H.J. Cognitive performance and response inhibition in developmentally vitamin D (DVD)-deficient rats. Behav. Brain Res. 2013;242:47–53. doi: 10.1016/j.bbr.2012.12.029. [DOI] [PubMed] [Google Scholar]
- 92.Overeem K., Alexander S., Burne T.H.J., Ko P., Eyles D.W. Developmental Vitamin D Deficiency in the Rat Impairs Recognition Memory, but Has No Effect on Social Approach or Hedonia. Nutrients. 2019;11:2713. doi: 10.3390/nu11112713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Vuillermot S., Luan W., Meyer U., Eyles D. Vitamin D treatment during pregnancy prevents autism-related phenotypes in a mouse model of maternal immune activation. Mol. Autism. 2017;8:9. doi: 10.1186/s13229-017-0125-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Kazemi F., Babri S., Keyhanmehr P., Farid-Habibi M., Rad S.N., Farajdokht F. Maternal vitamin D supplementation and treadmill exercise attenuated vitamin D deficiency-induced anxiety-and depressive-like behaviors in adult male offspring rats. Nutr. Neurosci. 2022:1–13. doi: 10.1080/1028415X.2022.2059203. [DOI] [PubMed] [Google Scholar]
- 95.Schoenrock S.A., Tarantino L.M. Deveopmental vitamin D deficiency and schizophrenia: The role of animal models. Genes Brain Behav. 2016;15:45–61. doi: 10.1111/gbb.12271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Harms L.H., Turner K.M., Eyles D.W., Young J.W., McGrath J.J., BUrne T.H.J. Attentional processing in C57BL/6J mice exposed to developmental vitamin D deficiency. PLoS ONE. 2012;7:e35896. doi: 10.1371/journal.pone.0035896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Harms L.R., Eyles D.W., McGrath J.J., Mackay-Sim A., Burne T.H. Developmental vitamin D deficiency alters adult behaviour in 129/SvJ and C57BL/6J mice. Behav. Brain Res. 2008;187:343–350. doi: 10.1016/j.bbr.2007.09.032. [DOI] [PubMed] [Google Scholar]
- 98.Harms L.H., Cowin G., Eyles D.W., Kurniawan N., McGrath J.J., Burne T.H.J. Neuroanatomy and psychomimetic-induced locomotion in C57BL/6J and 129/X1SvJ mice exposed to developmental vitamin D deficiency. Behav. Brain Res. 2012;230:125–131. doi: 10.1016/j.bbr.2012.02.007. [DOI] [PubMed] [Google Scholar]
- 99.Hart P.H., Lucas R.M., Walsh J.P., Zosky G.R., Whitehouse A.J., Zhu K., Allen K.L., Kusel M.M., Anderson D., Mountain J.A. Vitamin D in fetal development: Findings from a birth cohort study. Pediatrics. 2015;135:e167–e173. doi: 10.1542/peds.2014-1860. [DOI] [PubMed] [Google Scholar]
- 100.Voltas N., Canals J., Hernandez-Martinez C., Serrat N., Basora J., Arija V. Effect of Vitamin D Status during Pregnancy on Infant Neurodevelopment: The ECLIPSES Study. Nutrients. 2020;12:3196. doi: 10.3390/nu12103196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Mutua A.M., Mogire R.M., Elliott A.M., Williams T.N., Webb E.L., Abubakar A., Atkinson S.H. Effects of vitamin D deficiency on neurobehavioural outcomes in children: A systematic review. Wellcome Open Res. 2020;5:28. doi: 10.12688/wellcomeopenres.15730.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Arrhenius B., Upadhyaya S., Hinkka-Yli-Salomaki S., Brown A.S., Cheslack-Postava K., Ohman H., Sourander A. Prenatal Vitamin D Levels in Maternal Sera and Offspring Specific Learning Disorders. Nutrients. 2021;13:3321. doi: 10.3390/nu13103321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wicklow B., Gallo S., Majnemer A., Vanstone C., Comeau K., Jones G., L’Abbe M., Khamessan A., Sharma A., Weiler H., et al. Impact of Vitamin D Supplementation on Gross Motor Development of Healthy Term Infants: A Randomized Dose-Response Trial. Phys. Occup. Ther. Pediatr. 2016;36:330–342. doi: 10.3109/01942638.2015.1050150. [DOI] [PubMed] [Google Scholar]
- 104.Whitehouse A.J., Holt B.J., Serralha M., Holt P.G., Kusel M.M., Hart P.H. Maternal serum vitamin D levels during pregnancy and offspring neurocognitive development. Pediatrics. 2012;129:485–493. doi: 10.1542/peds.2011-2644. [DOI] [PubMed] [Google Scholar]
- 105.Hanieh S., Ha T.T., Simpson J.A., Thuy T.T., Khuong N.C., Thoang D.D., Tran T.D., Tuan T., Fisher J., Biggs B.A. Maternal vitamin D status and infant outcomes in rural Vietnam: A prospective cohort study. PLoS ONE. 2014;9:e99005. doi: 10.1371/journal.pone.0099005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Schmidt R.J., Niu Q., Eyles D.W., Hansen R.L., Iosif A.M. Neonatal vitamin D status in relation to autism spectrum disorder and developmental delay in the CHARGE case-control study. Autism. Res. 2019;12:976–988. doi: 10.1002/aur.2118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lopez-Vicente M., Sunyer J., Lertxundi N., Gonzalez L., Rodriguez-Dehli C., Espada Saenz-Torre M., Vrijheid M., Tardon A., Llop S., Torrent M., et al. Maternal circulating Vitamin D3 levels during pregnancy and behaviour across childhood. Sci. Rep. 2019;9:14792. doi: 10.1038/s41598-019-51325-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Sass L., Vinding R.K., Stokholm J., Bjarnadottir E., Noergaard S., Thorsen J., Sunde R.B., McGrath J., Bonnelykke K., Chawes B., et al. High-Dose Vitamin D Supplementation in Pregnancy and Neurodevelopment in Childhood: A Prespecified Secondary Analysis of a Randomized Clinical Trial. JAMA Netw. Open. 2020;3:e2026018. doi: 10.1001/jamanetworkopen.2020.26018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Li B., Xu Y., Zhang X., Zhang L., Wu Y., Wang X., Zhu C. The effect of vitamin D supplementation in treatment of children with autism spectrum disorder: A systematic review and meta-analysis of randomized controlled trials. Nutr. Neurosci. 2022;25:835–845. doi: 10.1080/1028415X.2020.1815332. [DOI] [PubMed] [Google Scholar]
- 110.Dehbokri N., Noorazar G., Ghaffari A., Mehdizadeh G., Sarbakhsh P., Ghaffary S. Effect of vitamin D treatment in children with attention-deficit hyperactivity disorder. World J. Pediatr. 2019;15:78–84. doi: 10.1007/s12519-018-0209-8. [DOI] [PubMed] [Google Scholar]
- 111.Elshorbagy H.H., Barseem N.F., Abdelghani W.E., Suliman H.A.I., Al-Shokary A.H., Abdulsamea S.E., Elsadek A.E., Abdel Maksoud Y.H., Nour El Din D. Impact of Vitamin D Supplementation on Attention-Deficit Hyperactivity Disorder in Children. Ann. Pharmacother. 2018;52:623–631. doi: 10.1177/1060028018759471. [DOI] [PubMed] [Google Scholar]
- 112.Miller M.C., Pan X., Eugene Arnold L., Mulligan A., Connor S., Bergman R., deBeus R., Roley-Roberts M.E. Vitamin D levels in children with attention deficit hyperactivity disorder: Association with seasonal and geographical variation, supplementation, inattention severity, and theta:beta ratio. Biol. Psychol. 2021;162:108099. doi: 10.1016/j.biopsycho.2021.108099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Mohammadpour N., Jazayeri S., Tehrani-Doost M., Djalali M., Hosseini M., Effatpanah M., Davari-Ashtiani R., Karami E. Effect of vitamin D supplementation as adjunctive therapy to methylphenidate on ADHD symptoms: A randomized, double blind, placebo-controlled trial. Nutr. Neurosci. 2018;21:202–209. doi: 10.1080/1028415X.2016.1262097. [DOI] [PubMed] [Google Scholar]
- 114.Zhou P., Wolraich M.L., Cao A.H., Jia F.Y., Liu B., Zhu L., Liu Y., Li X., Li C., Peng B., et al. Adjuvant effects of vitamin A and vitamin D supplementation on treatment of children with attention-deficit/hyperactivity disorder: A study protocol for a randomised, double-blinded, placebo-controlled, multicentric trial in China. BMJ Open. 2021;11:e050541. doi: 10.1136/bmjopen-2021-050541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Pilz S., Trummer C., Theiler-Schwetz V., Grubler M.R., Verheyen N.D., Odler B., Karras S.N., Zittermann A., Marz W. Critical Appraisal of Large Vitamin D Randomized Controlled Trials. Nutrients. 2022;14:303. doi: 10.3390/nu14020303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Suzuki M., Yoshioka M., Hashimoto M., Murakami M., Noya M., Takahashi D., Urashima M. Randomized, double-blind, placebo-controlled trial of vitamin D supplementation in Parkinson disease. Am. J. Clin. Nutr. 2013;97:1004–1013. doi: 10.3945/ajcn.112.051664. [DOI] [PubMed] [Google Scholar]
- 117.Kang J.H., Vyas C.M., Okereke O.I., Ogata S., Albert M., Lee I.M., D’Agostino D., Buring J.E., Cook N.R., Grodstein F., et al. Effect of vitamin D on cognitive decline: Results from two ancillary studies of the VITAL randomized trial. Sci. Rep. 2021;11:23253. doi: 10.1038/s41598-021-02485-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Feige J., Moser T., Bieler L., Schwenker K., Hauer L., Sellner J. Vitamin D Supplementation in Multiple Sclerosis: A Critical Analysis of Potentials and Threats. Nutrients. 2020;12:783. doi: 10.3390/nu12030783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Lieberherr M. Effects of vitamin D3 metabolites on cytosolic free calcium in confluent mouse osteoblasts. J. Biol. Chem. 1987;262:13168–13173. doi: 10.1016/S0021-9258(18)45183-6. [DOI] [PubMed] [Google Scholar]
- 120.Caffrey J.M., Farach-Carson M.C. Vitamin D3 metabolites modulate dihydropyridine-sensitive calcium currents in clonal rat osteosarcoma cells. J. Biol. Chem. 1989;264:20265–20274. doi: 10.1016/S0021-9258(19)47057-9. [DOI] [PubMed] [Google Scholar]
- 121.Ibi M., Sawada H., Nakanishi M., Kume T., Katsuki H., Kaneko S., Shimohama S., Akaike A. Protective effects of 1 alpha,25-(OH)(2)D-3 against the neurotoxicity of glutamate and reactive oxygen species in mesencephalic culture. Neuropharmacology. 2001;40:761–771. doi: 10.1016/S0028-3908(01)00009-0. [DOI] [PubMed] [Google Scholar]
- 122.Brewer L.D., Thibault V., Chen K.C., Langub M.C., Landfield P.W., Porter N.M. Vitamin D hormone confers neuroprotection in parallel with downregulation of L-type calcium channel expression in hippocampal neurons. J. Neurosci. 2001;21:98–108. doi: 10.1523/JNEUROSCI.21-01-00098.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Gezen-Ak D., Dursun E., Yilmazer S. Vitamin D inquiry in hippocampal neurons: Consequences of vitamin D-VDR pathway disruption on calcium channel and the vitamin D requirement. Neurol. Sci. 2013;34:1453–1458. doi: 10.1007/s10072-012-1268-6. [DOI] [PubMed] [Google Scholar]
- 124.Zanatta L., Goulart P.B., Gonçalves R., Pierozan P., Winkelmann-Duarte E.C., Woehl V.M., Pessoa-Pureur R., Silva F.R., Zamoner A. 1α,25-dihydroxyvitamin D(3) mechanism of action: Modulation of L-type calcium channels leading to calcium uptake and intermediate filament phosphorylation in cerebral cortex of young rats. Biochim. Biophys. Acta. 2012;1823:1708–1719. doi: 10.1016/j.bbamcr.2012.06.023. [DOI] [PubMed] [Google Scholar]
- 125.Gooch H., Cui X., Anggono V., Trzaskowski M., Tan M.C., Eyles D.W., Burne T.H.J., Jang S.E., Mattheisen M., Hougaard D.M., et al. 1,25-Dihydroxyvitamin D modulates L-type voltage-gated calcium channels in a subset of neurons in the developing mouse prefrontal cortex. Transl. Psychiatry. 2019;9:281. doi: 10.1038/s41398-019-0626-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Schizophrenia Working Group of the Psychiatric Genomics Consortium Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421–427. doi: 10.1038/nature13595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Long W., Fatehi M., Soni S., Panigrahi R., Philippaert K., Yu Y., Kelly R., Boonen B., Barr A., Golec D., et al. Vitamin D is an endogenous partial agonist of the transient receptor potential vanilloid 1 channel. J. Physiol. 2020;598:4321–4338. doi: 10.1113/JP279961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Holick M.F. Vitamin D deficiency. N. Engl. J. Med. 2007;357:266–281. doi: 10.1056/NEJMra070553. [DOI] [PubMed] [Google Scholar]
- 129.Almeida Moreira Leal L.K., Lima L.A., Alexandre de Aquino P.E., Costa de Sousa J.A., Jatai Gadelha C.V., Felicio Calou I.B., Pereira Lopes M.J., Viana Lima F.A., Tavares Neves K.R., Matos de Andrade G., et al. Vitamin D (VD3) antioxidative and anti-inflammatory activities: Peripheral and central effects. Eur. J. Pharmacol. 2020;879:173099. doi: 10.1016/j.ejphar.2020.173099. [DOI] [PubMed] [Google Scholar]
- 130.Uberti F., Morsanuto V., Bardelli C., Molinari C. Protective effects of 1α,25-Dihydroxyvitamin D3 on cultured neural cells exposed to catalytic iron. Physiol. Rep. 2016;4:e12769. doi: 10.14814/phy2.12769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Chen K.B., Lin A.M., Chiu T.H. Systemic vitamin D3 attenuated oxidative injuries in the locus coeruleus of rat brain. Ann. N. Y. Acad. Sci. 2003;993:313–324; discussion 345–349. doi: 10.1111/j.1749-6632.2003.tb07539.x. [DOI] [PubMed] [Google Scholar]
- 132.Lin A.M., Fan S.F., Yang D.M., Hsu L.L., Yang C.H. Zinc-induced apoptosis in substantia nigra of rat brain: Neuroprotection by vitamin D3. Free Radic. Biol. Med. 2003;34:1416–1425. doi: 10.1016/S0891-5849(03)00105-9. [DOI] [PubMed] [Google Scholar]
- 133.Kasatkina L.A., Tarasenko A.S., Krupko O.O., Kuchmerovska T.M., Lisakovska O.O., Trikash I.O. Vitamin D deficiency induces the excitation/inhibition brain imbalance and the proinflammatory shift. Int. J. Biochem. Cell Biol. 2020;119:105665. doi: 10.1016/j.biocel.2019.105665. [DOI] [PubMed] [Google Scholar]
- 134.Garcion E., Nataf S., Berod A., Darcy F., Brachet P. 1,25-Dihydroxyvitamin D3 inhibits the expression of inducible nitric oxide synthase in rat central nervous system during experimental allergic encephalomyelitis. Brain Res. Mol. Brain Res. 1997;45:255–267. doi: 10.1016/S0169-328X(96)00260-4. [DOI] [PubMed] [Google Scholar]
- 135.Garcion E., Sindji L., Montero-Menei C., Andre C., Brachet P., Darcy F. Expression of inducible nitric oxide synthase during rat brain inflammation: Regulation by 1,25-dihydroxyvitamin D3. Glia. 1998;22:282–294. doi: 10.1002/(SICI)1098-1136(199803)22:3<282::AID-GLIA7>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 136.Lefebvre d’Hellencourt C., Montero-Menei C.N., Bernard R., Couez D. Vitamin D3 inhibits proinflammatory cytokines and nitric oxide production by the EOC13 microglial cell line. J. Neurosci. Res. 2003;71:575–582. doi: 10.1002/jnr.10491. [DOI] [PubMed] [Google Scholar]
- 137.Hur J., Lee P.H., Kim M.J., Cho Y.-W. Regulatory effect of 25-hydroxyvitamin D3 on nitric oxide production in activated microglia. Korean J. Physiol. Pharmacol. 2014;18:397–402. doi: 10.4196/kjpp.2014.18.5.397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Huang Y., Ho Y., Lai C., Chiu C., Wang Y. 1,25-dihydroxyvitamin D3 attenuates endotoxin-induced production of inflammatory mediators by inhibiting MAPK activation in primary cortical neuron-glia cultures. J. Neuroinflammation. 2015;12:147. doi: 10.1186/s12974-015-0370-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Loginova M., Mishchenko T., Savyuk M., Guseva S., Gavrish M., Krivonosov M., Ivanchenko M., Fedotova J., Vedunova M. Double-Edged Sword of Vitamin D3 Effects on Primary Neuronal Cultures in Hypoxic States. Int. J. Mol. Sci. 2021;22:5417. doi: 10.3390/ijms22115417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Ali A., Cui X., Alexander S., Eyles D. The placental immune response is dysregulated developmentally vitamin D deficient rats: Relevance to autism. J. Steroid. Biochem. Mol. Biol. 2018;180:73–80. doi: 10.1016/j.jsbmb.2018.01.015. [DOI] [PubMed] [Google Scholar]
- 141.Alessio N., Belardo C., Trotta M.C., Paino S., Boccella S., Gargano F., Pieretti G., Ricciardi F., Marabese I., Luongo L., et al. Vitamin D Deficiency Induces Chronic Pain and Microglial Phenotypic Changes in Mice. Int. J. Mol. Sci. 2021;22:3604. doi: 10.3390/ijms22073604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Rastegar-Moghaddam S.H., Hosseini M., Alipour F., Rajabian A., Ebrahimzadeh Bideskan A. The effects of vitamin D on learning and memory of hypothyroid juvenile rats and brain tissue acetylcholinesterase activity and oxidative stress indicators. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2022;395:337–351. doi: 10.1007/s00210-021-02195-y. [DOI] [PubMed] [Google Scholar]
- 143.Mokhtari-Zaer A., Hosseini M., Salmani H., Arab Z., Zareian P. Vitamin D3 attenuates lipopolysaccharide-induced cognitive impairment in rats by inhibiting inflammation and oxidative stress. Life Sci. 2020;253:117703. doi: 10.1016/j.lfs.2020.117703. [DOI] [PubMed] [Google Scholar]
- 144.Xu M.L., Yu X.J., Zhao J.Q., Du Y., Xia W.J., Su Q., Du M.M., Yang Q., Qi J., Li Y., et al. Calcitriol ameliorated autonomic dysfunction and hypertension by down-regulating inflammation and oxidative stress in the paraventricular nucleus of SHR. Toxicol. Appl. Pharmacol. 2020;394:114950. doi: 10.1016/j.taap.2020.114950. [DOI] [PubMed] [Google Scholar]
- 145.Khassafi N., Zahraei Z., Vahidinia Z., Karimian M., Azami Tameh A. Calcitriol Pretreatment Attenuates Glutamate Neurotoxicity by Regulating NMDAR and CYP46A1 Gene Expression in Rats Subjected to Transient Middle Cerebral Artery Occlusion. J. Neuropathol. Exp. Neurol. 2022;81:252–259. doi: 10.1093/jnen/nlac011. [DOI] [PubMed] [Google Scholar]
- 146.Vahidinia Z., Khassafi N., Tameh A.A., Karimian M., Zare-Dehghanani Z., Moradi F., Joghataei M.T. Calcitriol Ameliorates Brain Injury in the Rat Model of Cerebral Ischemia-Reperfusion Through Nrf2/HO-1 Signalling Axis: An in Silico and in Vivo Study. J. Stroke Cerebrovasc. Dis. 2022;31:106331. doi: 10.1016/j.jstrokecerebrovasdis.2022.106331. [DOI] [PubMed] [Google Scholar]
- 147.Filomeni G., De Zio D., Cecconi F. Oxidative stress and autophagy: The clash between damage and metabolic needs. Cell Death Differ. 2015;22:377–388. doi: 10.1038/cdd.2014.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Bhowmick S., D’Mello V., Caruso D., Abdul-Muneer P.M. Traumatic brain injury-induced downregulation of Nrf2 activates inflammatory response and apoptotic cell death. J. Mol. Med. 2019;97:1627–1641. doi: 10.1007/s00109-019-01851-4. [DOI] [PubMed] [Google Scholar]
- 149.Cui C., Wang C., Jin F., Yang M., Kong L., Han W., Jiang P. Calcitriol confers neuroprotective effects in traumatic brain injury by activating Nrf2 signaling through an autophagy-mediated mechanism. Mol. Med. 2021;27:118. doi: 10.1186/s10020-021-00377-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Hosseinirad H., Shahrestanaki J.K., Moosazadeh Moghaddam M., Mousazadeh A., Yadegari P., Afsharzadeh N. Protective Effect of Vitamin D3 Against Pb-Induced Neurotoxicity by Regulating the Nrf2 and NF-kappaB Pathways. Neurotox. Res. 2021;39:687–696. doi: 10.1007/s12640-020-00322-w. [DOI] [PubMed] [Google Scholar]
- 151.Jiang H., Zhang S. Therapeutic effect of acute and chronic use of different doses of vitamin D3 on seizure responses and cognitive impairments induced by pentylenetetrazole in immature male rats. Iran. J. Basic Med. Sci. 2022;25:84–95. doi: 10.22038/IJBMS.2021.60123.13328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Bayo-Olugbami A., Nafiu A.B., Amin A., Ogundele O.M., Lee C.C., Owoyele B.V. Vitamin D attenuated 6-OHDA-induced behavioural deficits, dopamine dysmetabolism, oxidative stress, and neuro-inflammation in mice. Nutr. Neurosci. 2022;25:823–834. doi: 10.1080/1028415X.2020.1815331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Lima L.A.R., Lopes M.J.P., Costa R.O., Lima F.A.V., Neves K.R.T., Calou I.B.F., Andrade G.M., Viana G.S.B. Vitamin D protects dopaminergic neurons against neuroinflammation and oxidative stress in hemiparkinsonian rats. J. Neuroinflammation. 2018;15:249. doi: 10.1186/s12974-018-1266-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Hu J., Wu J., Wan F., Kou L., Yin S., Sun Y., Li Y., Zhou Q., Wang T. Calcitriol Alleviates MPP(+)- and MPTP-Induced Parthanatos Through the VDR/PARP1 Pathway in the Model of Parkinson’s Disease. Front. Aging Neurosci. 2021;13:657095. doi: 10.3389/fnagi.2021.657095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Fan Y.G., Pang Z.Q., Wu T.Y., Zhang Y.H., Xuan W.Q., Wang Z., Yu X., Li Y.C., Guo C., Wang Z.Y. Vitamin D deficiency exacerbates Alzheimer-like pathologies by reducing antioxidant capacity. Free Radic. Biol. Med. 2020;161:139–149. doi: 10.1016/j.freeradbiomed.2020.10.007. [DOI] [PubMed] [Google Scholar]
- 156.Mehrabadi S., Sadr S.S. Administration of Vitamin D3 and E supplements reduces neuronal loss and oxidative stress in a model of rats with Alzheimer’s disease. Neurol. Res. 2020;42:862–868. doi: 10.1080/01616412.2020.1787624. [DOI] [PubMed] [Google Scholar]
- 157.Grimm M.O.W., Lauer A.A., Grosgen S., Thiel A., Lehmann J., Winkler J., Janitschke D., Herr C., Beisswenger C., Bals R., et al. Profiling of Alzheimer’s disease related genes in mild to moderate vitamin D hypovitaminosis. J. Nutr. Biochem. 2019;67:123–137. doi: 10.1016/j.jnutbio.2019.01.015. [DOI] [PubMed] [Google Scholar]
- 158.Lin F.Y., Lin Y.F., Lin Y.S., Yang C.M., Wang C.C., Hsiao Y.H. Relative D3 vitamin deficiency and consequent cognitive impairment in an animal model of Alzheimer’s disease: Potential involvement of collapsin response mediator protein-2. Neuropharmacology. 2020;164:107910. doi: 10.1016/j.neuropharm.2019.107910. [DOI] [PubMed] [Google Scholar]
- 159.Lin C.I., Chang Y.C., Kao N.J., Lee W.J., Cross T.W., Lin S.H. 1,25(OH)2D3 Alleviates Abeta(25-35)-Induced Tau Hyperphosphorylation, Excessive Reactive Oxygen Species, and Apoptosis Through Interplay with Glial Cell Line-Derived Neurotrophic Factor Signaling in SH-SY5Y Cells. Int. J. Mol. Sci. 2020;21:4215. doi: 10.3390/ijms21124215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.De Oliveira L.R.C., Mimura L.A.N., Fraga-Silva T.F.C., Ishikawa L.L.W., Fernandes A.A.H., Zorzella-Pezavento S.F.G., Sartori A. Calcitriol Prevents Neuroinflammation and Reduces Blood-Brain Barrier Disruption and Local Macrophage/Microglia Activation. Front. Pharmacol. 2020;11:161. doi: 10.3389/fphar.2020.00161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Shirazi H.A., Rasouli J., Ciric B., Wei D., Rostami A., Zhang G.X. 1,25-Dihydroxyvitamin D3 suppressed experimental autoimmune encephalomyelitis through both immunomodulation and oligodendrocyte maturation. Exp. Mol. Pathol. 2017;102:515–521. doi: 10.1016/j.yexmp.2017.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Nystad A.E., Wergeland S., Aksnes L., Myhr K.M., Bo L., Torkildsen O. Effect of high-dose 1.25 dihydroxyvitamin D3 on remyelination in the cuprizone model. APMIS. 2014;122:1178–1186. doi: 10.1111/apm.12281. [DOI] [PubMed] [Google Scholar]
- 163.Paintlia M.K., Singh I., Singh A.K. Effect of vitamin D3 intake on the onset of disease in a murine model of human Krabbe disease. J. Neurosci. Res. 2015;93:28–42. doi: 10.1002/jnr.23476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Sapolsky R.M. Stress, Glucocorticoids, and Damage to the Nervous System: The Current State of Confusion. Stress. 1996;1:1–19. doi: 10.3109/10253899609001092. [DOI] [PubMed] [Google Scholar]
- 165.Chen T.L., Cone C.M., Morey-Holton E., Feldman D. Glucocorticoid regulation of 1,25(OH)2-vitamin D3 receptors in cultured mouse bone cells. J. Biol. Chem. 1982;257:13564–13569. doi: 10.1016/S0021-9258(18)33484-7. [DOI] [PubMed] [Google Scholar]
- 166.Chen T.L., Cone C.M., Morey-Holton E., Feldman D. 1 alpha,25-dihydroxyvitamin D3 receptors in cultured rat osteoblast-like cells. Glucocorticoid treatment increases receptor content. J. Biol. Chem. 1983;258:4350–4355. doi: 10.1016/S0021-9258(18)32630-9. [DOI] [PubMed] [Google Scholar]
- 167.Neveu I., Barbot N., Jehan F., Wion D., Brachet P. Antagonistic effects of dexamethasone and 1,25-dihydroxyvitamin D3 on the synthesis of nerve growth factor. Mol. Cell. Endocrinol. 1991;78:R1–R6. doi: 10.1016/0303-7207(91)90129-G. [DOI] [PubMed] [Google Scholar]
- 168.Neveu I., Jehan F., Wion D. Alteration in the levels of 1,25-(OH)2D3 and corticosterone found in experimental diabetes reduces nerve growth factor (NGF) gene expression in vitro. Life Sci. 1992;50:1769–1772. doi: 10.1016/0024-3205(92)90060-3. [DOI] [PubMed] [Google Scholar]
- 169.Lundqvist J., Norlin M., Wikvall K. 1alpha,25-Dihydroxyvitamin D3 affects hormone production and expression of steroidogenic enzymes in human adrenocortical NCI-H295R cells. Biochim. Biophys. Acta. 2010;1801:1056–1062. doi: 10.1016/j.bbalip.2010.04.009. [DOI] [PubMed] [Google Scholar]
- 170.Obradovic D., Gronemeyer H., Lutz B., Rein T. Cross-talk of vitamin D and glucocorticoids in hippocampal cells. J. Neurochem. 2006;96:500–509. doi: 10.1111/j.1471-4159.2005.03579.x. [DOI] [PubMed] [Google Scholar]
- 171.Jiang P., Xue Y., Li H.D., Liu Y.P., Cai H.L., Tang M.M., Zhang L.H. Dysregulation of vitamin D metabolism in the brain and myocardium of rats following prolonged exposure to dexamethasone. Psychopharmacology. 2014;231:3345–3351. doi: 10.1007/s00213-014-3440-6. [DOI] [PubMed] [Google Scholar]
- 172.Camargo A., Dalmagro A.P., Platt N., Rosado A.F., Neis V.B., Zeni A.L.B., Kaster M.P., Rodrigues A.L.S. Cholecalciferol abolishes depressive-like behavior and hippocampal glucocorticoid receptor impairment induced by chronic corticosterone administration in mice. Pharmacol. Biochem. Behav. 2020;196:172971. doi: 10.1016/j.pbb.2020.172971. [DOI] [PubMed] [Google Scholar]
- 173.Camargo A., Dalmagro A.P., Rikel L., da Silva E.B., Simao da Silva K.A.B., Zeni A.L.B. Cholecalciferol counteracts depressive-like behavior and oxidative stress induced by repeated corticosterone treatment in mice. Eur. J. Pharmacol. 2018;833:451–461. doi: 10.1016/j.ejphar.2018.07.002. [DOI] [PubMed] [Google Scholar]
- 174.Koshkina A., Dudnichenko T., Baranenko D., Fedotova J., Drago F. Effects of Vitamin D3 in Long-Term Ovariectomized Rats Subjected to Chronic Unpredictable Mild Stress: BDNF, NT-3, and NT-4 Implications. Nutrients. 2019;11:1726. doi: 10.3390/nu11081726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Sedaghat K., Yousefian Z., Vafaei A.A., Rashidy-Pour A., Parsaei H., Khaleghian A., Choobdar S. Mesolimbic dopamine system and its modulation by vitamin D in a chronic mild stress model of depression in the rat. Behav. Brain Res. 2019;356:156–169. doi: 10.1016/j.bbr.2018.08.020. [DOI] [PubMed] [Google Scholar]
- 176.Sedaghat K., Naderian R., Pakdel R., Bandegi A.R., Ghods Z. Regulatory effect of vitamin D on pro-inflammatory cytokines and anti-oxidative enzymes dysregulations due to chronic mild stress in the rat hippocampus and prefrontal cortical area. Mol. Biol. Rep. 2021;48:7865–7873. doi: 10.1007/s11033-021-06810-2. [DOI] [PubMed] [Google Scholar]
- 177.Neis V.B., Werle I., Moretti M., Rosa P.B., Camargo A., de O. Dalsenter, Y.; Platt, N.; Rosado, A.F.; Engel, W.D.; de Almeida, G.R.L.; et al. Involvement of serotonergic neurotransmission in the antidepressant-like effect elicited by cholecalciferol in the chronic unpredictable stress model in mice. Metab. Brain Dis. 2022;37:1597–1608. doi: 10.1007/s11011-022-00979-6. [DOI] [PubMed] [Google Scholar]
- 178.Kaneko I., Sabir M.S., Dussik C.M., Whitfield G.K., Karrys A., Hsieh J.C., Haussler M.R., Meyer M.B., Pike J.W., Jurutka P.W. 1,25-Dihydroxyvitamin D regulates expression of the tryptophan hydroxylase 2 and leptin genes: Implication for behavioral influences of vitamin D. FASEB J. 2015;29:4023–4035. doi: 10.1096/fj.14-269811. [DOI] [PubMed] [Google Scholar]
- 179.Da Silva Souza S.V., da Rosa P.B., Neis V.B., Moreira J.D., Rodrigues A.L.S., Moretti M. Effects of cholecalciferol on behavior and production of reactive oxygen species in female mice subjected to corticosterone-induced model of depression. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2020;393:111–120. doi: 10.1007/s00210-019-01714-2. [DOI] [PubMed] [Google Scholar]
- 180.Eyles D.W., Rogers F., Buller K., McGrath J.J., Ko P., French K., Burne T.H. Developmental vitamin D (DVD) deficiency in the rat alters adult behaviour independently of HPA function. Psychoneuroendocrinology. 2006;31:958–964. doi: 10.1016/j.psyneuen.2006.05.006. [DOI] [PubMed] [Google Scholar]
- 181.Tesic D., Hawes J.E., Zosky G.R., Wyrwoll C.S. Vitamin D Deficiency in BALB/c Mouse Pregnancy Increases Placental Transfer of Glucocorticoids. Endocrinology. 2015;156:3673–3679. doi: 10.1210/en.2015-1377. [DOI] [PubMed] [Google Scholar]
- 182.Meaney M.J. Maternal care, gene expression, and the transmission of individual differences in stress reactivity across generations. Annu. Rev. Neurosci. 2001;24:1161–1192. doi: 10.1146/annurev.neuro.24.1.1161. [DOI] [PubMed] [Google Scholar]
- 183.Rajabi-Naeeni M., Dolatian M., Qorbani M., Vaezi A.A. Effect of omega-3 and vitamin D co-supplementation on psychological distress in reproductive-aged women with pre-diabetes and hypovitaminosis D: A randomized controlled trial. Brain Behav. 2021;11:e2342. doi: 10.1002/brb3.2342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Tebben P.J., Singh R.J., Kumar R. Vitamin D-Mediated Hypercalcemia: Mechanisms, Diagnosis, and Treatment. Endocr. Rev. 2016;37:521–547. doi: 10.1210/er.2016-1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Tuohimaa P., Keisala T., Minasyan A., Cachat J., Kalueff A. Vitamin D, nervous system and aging. Psychoneuroendocrinology. 2009;34:S278–S286. doi: 10.1016/j.psyneuen.2009.07.003. [DOI] [PubMed] [Google Scholar]
- 186.Lima G.O., Menezes da Silva A.L., Azevedo J.E.C., Nascimento C.P., Vieira L.R., Hamoy A.O., Oliveira Ferreira L., Bahia V., Muto N.A., Lopes D.C.F., et al. 100 YEARS of VITAMIN D: Supraphysiological doses of vitamin D changes brainwave activity patterns in rats. Endocr. Connect. 2022;11:e210457. doi: 10.1530/EC-21-0457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Lam V., Takechi R., Mano J. Vitamin D, Cerebrocapillary Integrity and Cognition in Murine Model of Accelerated Ageing. Alzheimer’s Dement. 2017;13:1304. doi: 10.1016/j.jalz.2017.06.1990. [DOI] [Google Scholar]
- 188.Razzaque M.S., Sitara D., Taguchi T., St-Arnaud R., Lanske B. Premature aging-like phenotype in fibroblast growth factor 23 null mice is a vitamin D-mediated process. FASEB J. 2006;20:720–722. doi: 10.1096/fj.05-5432fje. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Momen N.C., Plana-Ripoll O., Agerbo E., Benros M.E., Borglum A.D., Christensen M.K., Dalsgaard S., Degenhardt L., de Jonge P., Debost J.P.G., et al. Association between Mental Disorders and Subsequent Medical Conditions. N. Engl. J. Med. 2020;382:1721–1731. doi: 10.1056/NEJMoa1915784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Revez J.A., Lin T., Qiao Z., Xue A., Holtz Y., Zhu Z., Zeng J., Wang H., Sidorenko J., Kemper K.E., et al. Genome-wide association study identifies 143 loci associated with 25 hydroxyvitamin D concentration. Nat. Commun. 2020;11:1647. doi: 10.1038/s41467-020-15421-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Wu D.M., Wen X., Han X.R., Wang S., Wang Y.J., Shen M., Fan S.H., Zhuang J., Li M.Q., Hu B., et al. Relationship between Neonatal Vitamin D at Birth and Risk of Autism Spectrum Disorders: The NBSIB Study. J. Bone Miner. Res. 2018;33:458–466. doi: 10.1002/jbmr.3326. [DOI] [PubMed] [Google Scholar]
- 192.Windham G.C., Pearl M., Anderson M.C., Poon V., Eyles D., Jones K.L., Lyall K., Kharrazi M., Croen L.A. Newborn vitamin D levels in relation to autism spectrum disorders and intellectual disability: A case-control study in california. Autism. Res. 2019;12:989–998. doi: 10.1002/aur.2092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Saraf R., Morton S.M.B., Camargo C.A.J., Grant C.C. Global summary of maternal and newborn vitamin D status—A systematic review. Matern. Child Nutr. 2016;12:647–668. doi: 10.1111/mcn.12210. [DOI] [PMC free article] [PubMed] [Google Scholar]