Abstract
Neutrophils, the most abundant innate immune cells, play essential roles in the innate immune system. As key innate immune cells, neutrophils detect intrusion of pathogens and initiate immune cascades with their functions; swarming (arresting), cytokine production, degranulation, phagocytosis, and projection of neutrophil extracellular trap. Because of their short lifespan and consumption during immune response, neutrophils need to be generated consistently, and generation of newborn neutrophils (granulopoiesis) should fulfill the environmental/systemic demands for training in cases of infection. Accumulating evidence suggests that neutrophils also play important roles in the regulation of adaptive immunity. Neutrophil-mediated immune responses end with apoptosis of the cells, and proper phagocytosis of the apoptotic body (efferocytosis) is crucial for initial and post resolution by producing tolerogenic innate/adaptive immune cells. However, inflammatory cues can impair these cascades, resulting in systemic immune activation; necrotic/pyroptotic neutrophil bodies can aggravate the excessive inflammation, increasing inflammatory macrophage and dendritic cell activation and subsequent TH1/TH17 responses contributing to the regulation of the pathogenesis of autoimmune disease. In this review, we briefly introduce recent studies of neutrophil function as players of immune response.
Keywords: Efferocytosis, Granulopoiesis, Immune homeostasis, Neutrophil, Resolution
INTRODUCTION
Neutrophils, the first defenders of the immune response, recruited into inflamed sites according to infectious and/or inflammatory cues. Recruited neutrophils recognize/integrate the unique patterns of danger molecule secreted by pathogens or the host and initiate immune cascades (1, 2). Neutrophils can eliminate extracellular pathogens or debris with phagocytic activity while releasing pro/anti-inflammatory cytokines and redox/cytotoxic molecules (3), therefore can present a ‘blueprint’ of further immune responses. Although the functional roles of neutrophils have been investigated well in innate immunity, the heritage of immune response after neutrophil action is now getting attention to understand the following innate/adaptive immune activation (2). The excessive activation of neutrophils can threaten the homeostasis of the host immune/organ system and paradoxically induce immune paralysis during the progress of sepsis and tumors (1, 2). Moreover, recent studies demonstrated the possibility of neutrophil response and death in the pathogenesis of chronic inflammation and autoimmune disease (2, 4, 5), proving the notion that neutrophils are not just a part of innate immune system. In this review, we briefly overview the functions of neutrophils and their generation by focusing on the roles of neutrophils as modulators of the entire immune response.
NEUTROPHIL-TRIGGERED INFLAMMATORY CASCADES
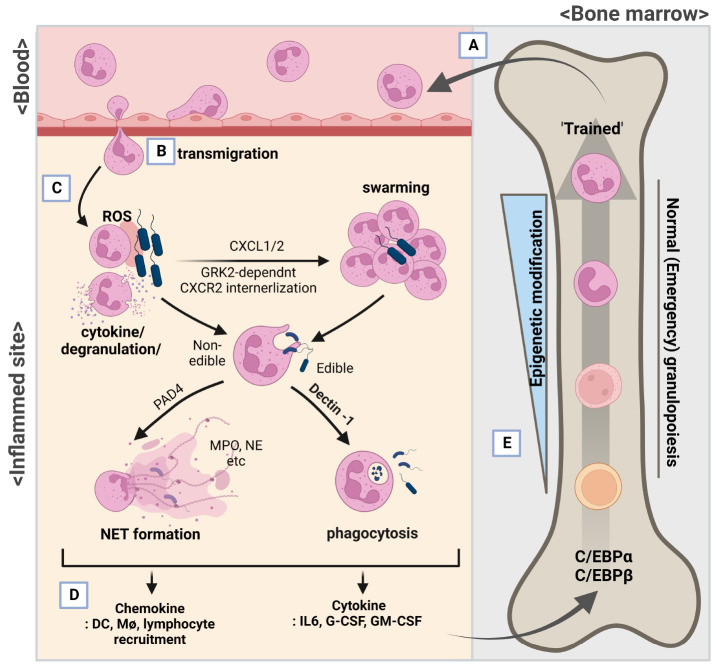
Neutrophils, the most abundant innate immune cells in blood stream patrol and surveil the inflammatory signs of the mammalian body (5-7). Depletion or defects of neutrophil function raises susceptibility to infection, especially opportunistic bacterial infection, demonstrating the important role of neutrophils for host defense (8). When epithelial cells or tissue-resident immune cells detect pathogen-associated molecular patterns (PAMPs) or host-derived danger-associated molecular patterns (DAMPs), they secrete alert signals and chemokines, making the inflammatory environment (2, 3). As the frontline unit of innate immune cells, neutrophils can recognize host- or bacteria-derived danger molecules and migrate into inflamed sites to block expansion of infection and inflammation (7, 9, 10) (Fig. 1A-D). When migrated neutrophils encounter pathogens, they may estimate the required number of neutrophils for pathogen exclusion with distinct reactive oxygen species (ROS) generation and secretion of IL-1β and chemokines (CXCL1 and CXCL2) (11). Recruited neutrophils, which undergo G protein-coupled receptor kinase 2 (GRK2)-dependent internalization of CXCR2, can surround and swarm around pathogens to prevent their escape, preparing initial immune responses (10, 12). Neutrophils can ingest (phagocytosis) and subsequently eliminate bacterial/fungal pathogens or host-derived particles, while selectively opening (closing) their azurophil, specific, or gelatinase granules and context-dependent cytokines based on complex signaling of pattern-recognition receptors (PRRs) and antibody-Fc receptor (2, 3, 13). During the process, neutrophils recognize and check the possibility of phagocytosis with dectin-1 (a non-TLR PRR), integrin Mac-1 (CD11b/CD18), and environmental cues (13-15). If the plan is frustrated, they are instructed to project lattice structures containing DNA and histone called a neutrophil extracellular trap (NET) and/or request the reinforcements of other immune cells, such as monocytes and macrophages for further immune response (4, 16). Whether there is neutrophil death (NETosis) or not (NET formation, surviving neutrophils), nuclear chromatin-based NET is not just a web-like DNA-histone complex; it is thickly covered with antimicrobial peptides and pro-coagulant molecules, like myeloperoxidase, cathepsin G, P-selectin glycoprotein ligand-1 (PSGL-1), neutrophil elastase (NE), defensins, and calprotectin (4, 17), carrying out neutrophil’s last mission even after death (NETosis). With the assistance of antimicrobial peptides from NET, reinforced macrophages can effectively kill the trapped pathogens, accelerating inflammatory cascades (16). Bacterial- or fungal-derived PAMPs and endogenous DAMPs (PSGL-1-platelets interaction, high-mobility group box 1 protein (HMGB1), immune complex) can act as triggers for NET formation; ROS/Ca2+ signaling can induce protein arginine deiminase type 4 (PAD4)-mediated citrullination (arginine to citrulline, positive charge to no net charge) of histone; meanwhile, NE from azurophilic granules translocates to the nucleus, and proteolytic activity of PAD4 and NE wind off condensed chromatins (4, 17), facilitating the projection of an ‘armed’ web. Not all neutrophils are programmed to project NET, and the other neutrophils (no NET-projected) still cluster around the pathogens, secreting IL-1β and chemokines for CXCR2 (11), and wait for their own destinies. In contrast to NET projection, when neutrophils carry out phagocytosis with dectin-1, recruitment of NE by activated phagosome attenuates translocation of NE to the nucleus and subsequently inhibits NET formation (14). Previously, we demonstrated the functional role of phospholipase D2 (PLD2), which catalyzes phosphatidylcholine-specific hydrolysis of phospholipids, in neutrophils for bacterial control during experimental sepsis. Inhibition of the PLD2 enzymatic activity or PLD2 knockout in neutrophils can attenuate GRK2-mediated CXCR2 internalization in an LPS-stimulated condition and an experimental mouse sepsis model (18). With GRK2-dependent CXCR2 internalization, neutrophils can self-limit and stand around the pathogens (swarming) and therefore can arrest pathogen movements (12). Damaged tissues and bacterial movement or swarming can cause changes in osmolarity, which can attract leukocytes to patrol to these sites (19, 20). Membrane tension increased by osmotic pressure can lead to the interaction of the PLD2-mammalian target of rapamycin complex 2 (mTORC2), and the PLD2-mTORC2 complex can inhibit actin assembly during neutrophil mobilization (21). PLD2 does not affect the phagocytic activity of neutrophils, but Pld2 deficiency significantly augmented NET and subsequently increased bactericidal effects with increased PAD activity (18), collectively showing the sequential and crucial roles of neutrophils in host defense.
Fig. 1.
Function of neutrophils and their generation (granulopoiesis). Neutrophils circulate and detect inflammatory cues. (A) Because of their short lifespan, neutrophils are continuously generated in the bone marrow of the hematopoietic system by granulopoiesis. (B) When they detect alert signals from inflamed tissue, neutrophils transmigrate into inflamed sites and initiate immune activation. (C) Sensing the size of pathogens by means of dectin-1, non-TLR pattern recognition receptor, and distinct generation of reactive oxygen species (ROS), neutrophils may surround pathogens (swarming), prey on them (phagocytosis), or project a sticky neutrophil extracellular trap (NET), while secreting context-dependent cytokines and granules (degranulation). (D, E) Self-immolation of neutrophils (D) and immune activation of other monocytes and macrophages (Mφ) increases production of IL-6, G-CSF, and GM-CSF, which in turn stimulate emergency neutrophil generation in the bone marrow (granulopoiesis) (E) and spleen (not shown). The context and signaling cues given for granulopoiesis affect the heterogeneity of newly generated neutrophils (trained granulopoiesis).
GENERATION OF ACQUIRED (TRAINED) NEUTROPHILS
While circulating neutrophils migrate into inflamed sites and ignite their short lives, new neutrophils are continually generated in bone marrow to replace their former fellows via granulopoiesis. Because of their relatively short lifespan (a few hours to a few days), neutrophils need to be generated from hematopoietic stem cells consistently at steady-state (normal granulopoiesis), and the hematopoietic system can rapidly adapt to hematopoietic stress and external environmental cues and produce the white blood cells needed urgently to deal with an call like infection (emergency granulopoiesis for neutrophils) (Fig. 1D, E) (22-24). Granulocyte colony-stimulating factor (G-CSF) is the main growth factor for granulopoiesis, and β-catenin-T-cell factor/lymphoid enhancer-binding factor-mediated signaling maintains neutrophil maturation during normal/emergency granulopoiesis by increasing G-CSF receptor expression (25). Pathogenic bacterial infection can interfere with the expression or stability of Wnt/β-catenin-mediated signaling, which can promote granulopoiesis, to avoid or use the host defense system (25, 26). On the other hand, inflammatory cascade can induce secretion of G-CSF, IL-6, and granulocyte-macrophage colony-stimulating factor, which can stimulate emergency granulopoiesis in bone-marrow (medullary) and spleen (extramedullary, in the emergency state) (1, 27). These results suggest that there is a competition between pathogens and innate immune cells for the host reinforcement system. When the hematopoietic system detects this pathogen-triggered hematopoietic stress and increased cytokines, the hematopoietic system of bone marrow and spleen rapidly switch the main transcription factor for granulopoiesis from CCAT/enhancer-binding protein (C/EBP)α to C/EBPβ, the master transcription factors for the steady and emergency states, respectively (1). Patterns of degraded or leaked proteins/peptides like N-formyl-peptides produced by the inflamed/damaged host cells or bacteria can be detected by formyl peptide receptor (FPR)2 and trigger emergency granulopoiesis. Blocking or deficiency of Fpr2 attenuates sepsis-induced neutrophil generation, and sole administration of an FPR2 ligand (WKYMVm) can be enough to induce granulopoiesis by increasing c-kit+sca-1− granulocyte-macrophage progenitor cells in a phospholipase C-dependent manner (28). Likewise, activation of FPR can prevent sepsis-induced mortality by increased H2O2 production of neutrophil and secretion of IFN-γ and IL-17a (29), the last of which can be secreted by IL-6/IL-23-exposed RORγt+ neutrophils, increase its bactericidal/anti-fungal activity (30, 31), and trigger IL-23/IL-17a-G-CSF axis-mediated granulopoiesis in bone marrow (32). Hence the hematopoietic system can detect molecular patterns and initiate generation of neutrophils. Several lines of study suggest that the properties of generated neutrophils are not constant; instead, the cells acquire lifelong functional modification, which is now called ‘trained immunity’ (33). The functions of trained neutrophil can be heterogeneous and context-dependent, which favor pro- or anti-inflammatory response in inflamed sites; for instance, β-glucan/type I interferon-trained neutrophils (N1 neutrophil) can drive anti-tumor activities with increased ROS production and T-cell stimulatory ligands (34, 35); meanwhile, prolonged G-CSF/GM-CSF-exposed (trained) neutrophils (N2 neutrophils) from bone marrow and spleen can drive pro-tumor immune responses (35-37) with increased angiogenetic molecules (VEGF, MMP-9) and T-cell suppressive ROS and arginase, the last two of which increase the ratio of Treg/cytotoxic CD8 T cells (35, 38, 39). Infection by bacteria (for example, M. tuberculosis) or change of microbiota can reprogram long-lasting myelopoiesis (40-43). Change of cytokine-sphingolipid signaling and subsequent lipid metabolism can affect the rate of myelopoiesis and differentiation of neutrophils with autophagy modulation (44-46). Likewise, Bacillus Calmette-Guérin (BCG) vaccination against tuberculosis can trigger epigenetic modification of neutrophils (genome-wide trimethylation at H3K4) and induce a phenotype change of generated neutrophils with increased maturation surface marker (CD10, CD15, and CD16) and activation marker (CD11b, CD66b) while decreasing CD62L (l-selectin) and PD-L1; these ‘trained’ neutrophil shows improved bactericidal and anti-fungal activity, but NET formation is not affected (47). Administration of 4-phenyl butyric acid, a peroxisomal stress-reducing agent and inhibitor of histone deacetylase, can potently educate a small subpopulation of CD200R+CD86+, but low CD177 (neutrophil exhaustion marker) pro-resolving (increased resolvin D1 (RvD1)/SerpinB1, reduced TNF-α) neutrophils with increased bactericidal activity (48). These studies indicate that, although the lifespan of neutrophils is relatively short, entrained by extrinsic cues with epigenetic modification from the immature stage (granulopoiesis) (23), neutrophils can be heterogeneous and ‘the giver’ of memory that guides the direction of further immune cascades. Interestingly, BCG vaccination of humans in the morning but not evening (circadian rhythm) can influence ‘long-term’ trained immunity of neutrophils (49). The interrelation between Bmal-1-dependent TH17 (not TH1 and Treg) development (in spleen and small intestine) and daily generation/oscillation of neutrophils (in bone marrow) (23, 49) suggests that trained granulopoiesis also can be affected by systemic TH17 activation and vice versa; that can explain the functional role of gut microbiota in regulating the generation/priming of neutrophils and why some neutrophils migrate into the intestine to control IL-23/IL-17-mediated G-CSF production (42, 50, 51). However, details of the immunological roles of trained granulopoiesis in generating TH17 and identification of specific gut microbiota involved in trained immunity need to be deeply explored to understand the patho-mechanism of chronic inflammatory disease.
PROGRAMED NEUTROPHIL APOPTOSIS AND INITIATION OF RESOLUTION
Neutrophil-mediated inflammatory responses end with apoptosis of the cells within inflamed sites, and some of the neutrophils reverse migrate to the lungs, the liver, the spleen, and the bone marrow, and then accept their programed cell death, which is critical for initiation of resolution (Fig. 2A-E) (32, 52, 53). Professional or non-professional phagocytic cells recognize the surface antigen (eat-me signal, phosphatidylserine) of apoptotic body and remove the debris of immune cascades via efferocytosis, restoring normal tissue/immune homeostasis (54). Macrophages are professional efferocytic cells that remove apoptotic neutrophils and neutrophil-derived NET (55). Engulfment of cellular debris from the apoptotic body or NET component can modulate intracellular machineries and metabolism of macrophages and regulate proliferation and phenotype change of efferocytes, accelerating tissue resolution (56, 57). During the efferocytic process, interaction between macrophage-derived developmental endothelial locus-1 and integrins (LFA-1, CD11a/CD18; and Mac-1) of the apoptotic body can increase the clearance of apoptotic neutrophils and subsequent immune resolution, which in turn induces production of specialized pro-resolving mediators, such as RvD1 and lipoxin A4 (LXA4) in macrophages (32, 58). As a positive feedback loop, RvD1 can limit LPS/arachidonic-acid-induced inflammatory cues while promoting the conversion of M2-macrophages (alternative activated) by switching production of proinflammatory leukotriene B4 to LXA4 and upregulating TGF-β (59-62). Produced LXA4 can sustain viability of macrophages against pathophysiological apoptotic cues by increasing Bcl2 via PI3K/Akt and ERK/Nrf-2 pathways and assist M2 macrophage polarization via the FPR2-IRF4/5 axis, accelerating the removal of apoptotic neutrophils (63-65). Likewise, complement protein C1q binds to apoptotic neutrophils and facilitates opsonization of NETs. Macrophages can also clear away apoptotic cells and C1q-opsonized NETs (55). Meanwhile, C1q can induce polarization of alternatively activated M2 macrophages in a MafB-dependent manner with increased type I IFN, IL-27, and IL-10 production, while attenuating inflammasome activation (66-68). Especially, efferocytosis of apoptotic (reverse migrated) neutrophils in bone marrow decreases IL-23/IL-17-G-CSF axis-mediated granulopoiesis in a β2 integrin (CD18)-dependent manner (32). Produced IL-10 can induce Jak-Stat3-mediated expression of the suppressor of cytokine signaling 3 (SOCS3), which can block G-CSF-mediated signaling and subsequent granulopoiesis; it can also block IL-6R/M-CSFR-mediated (WSXWS motif-containing, class I receptor) signaling but not IL-10R (no WSXWS motif, class II receptor) (69-72), thereby restraining the emergency preparedness and reinforcement of neutrophils. C1q bound to apoptotic cells also can modulate checkpoint ligand/receptor of dendritic cells (DC) (PD-L1, CD86) and macrophages (PD-L1/2, CD40) (73). These tolerogenic antigen-presenting cells (APCs) can migrate from inflamed sites into lymphatic drains and lymph nodes and induce Treg cells and T regulatory type 1 (Tr1) cells, which are crucial for initial and long-term peripheral tolerance (infectious tolerance), respectively (74, 75). Neutrophils can respond and adapt to the migrated circumstance with transcriptional modification (6, 76, 77). Treg/IL-10 educated neutrophils can become IL-10-producing and later apoptotic, assisting repair of damaged tissue by transferring preexisting matrix and fueling repair activities of other immune cells, such as monocytes, macrophages, and type 3 innate lymphoid cells (53, 76, 78, 79). Taken together, neutrophils are not limited to regulating inflammation in inflamed sites, but can also act as pioneers of systemic immune regulators.
Fig. 2.
Programmed neutrophil apoptosis and initiation of resolution. (A) When the role of neutrophils in inflamed sites ends, they remain or reverse migrate into other organs (bone marrow, lungs, and spleen; not shown) and undergo programmed cell death (apoptosis). (B) Apoptotic neutrophils expose phosphatidylserine as an eat-me signal, so that macrophages (Mφ), dendritic cells (DC), and monocytes can recognize an apoptotic body and initiate efferocytosis. (C) Clearance of an apoptotic body changes the phenotype of efferocytic cells and induces the expression of immune-modulatory lipids called specialized pro-resolving mediators (SPMs). The efferocytosis and subsequent activation with resolvin D1 (RvD1), lipoxin A4 (LXA4), and TGF-β accelerate removal of cellular debris and restore normal tissue/immune homeostasis. (D) Induction of immune modulatory T cells, Foxp3+ Treg and IL-10-producing CD4+Foxp3− Tr1. IL-10-educated neutrophils become apoptotic and help tissue repair. (E) The efferocytosis of apoptotic neutrophils in the bone marrow decreases the IL-23/IL-17a-G-CSF axis and restores normal state of granulopoiesis.
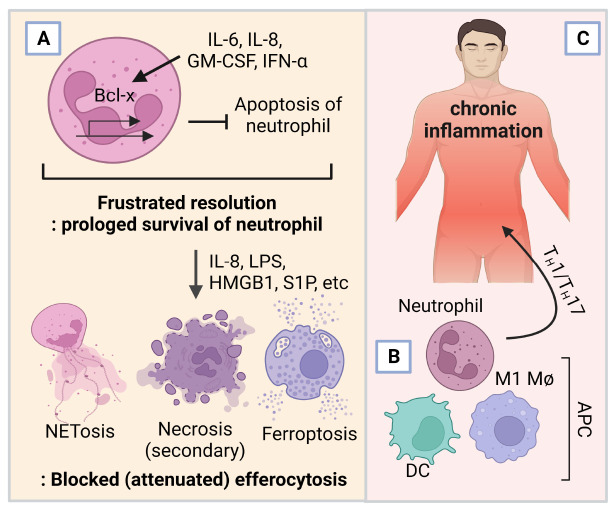
FRUSTRATED RESOLUTION AND NEUTROPHIL-MEDIATED CHRONIC INFLAMMATION
Recently, accumulating evidence suggests that defects in apoptotic progress of neutrophils and frustrated efferocytosis are closely related to chronic/systemic inflammation, and that neutrophils can carry phagocytic antigen and directly guide lymphocyte migration (as trail) and activation (Fig. 3A-C) (54, 80-84). Neutrophils can exhibit MHC and co-stimulatory molecules by localizing in peripheral tissue (lung) and being exposed to inflammatory cues like immune complex-mediated FcγR signaling, G-CSF, and GM-CSF (6, 83). The maturation state (CD10+) of neutrophils can present opposite effects on T cells, and trained granulopoiesis (after BCG vaccination) can tune the ratio of mature and immature neutrophils; mature CD10+ CD66b+ neutrophils display an activated phenotype, but inhibit proliferation and production of IFN-γ of T cells, whereas immature (CD10−) neutrophils sustain T-cell survival and increase proliferation and IFN-γ production (47, 82). Besides types of pathogens, developmental stage (immature, mature, or aged), activation state of neutrophils, and external cues of inflamed sites can shape the response of neutrophils with distinct transcriptional activities, and vice versa (23, 77). An interesting aspect of macrophage-mediated efferocytosis is that, if an apoptotic cell was not infected, this process does not load lysosomal particles to MHC and therefore can modulate antigen presentation to lymphocytes, removing inflammatory stimuli silently and attenuating systemic adaptive immune activation (54). However, failure to silence inflammatory cues (or evasion of pathogens from bactericidal action of neutrophils after phagocytosis) and/or defect of efferocytosis; subsequently neglected dead bodies can induce a form of programmed cell death called necrosis (secondary necrosis) (84, 85). Bursting out inflammatory molecules and bacterial components can trigger serial pro-inflammatory responses of inflamed sites, and inflammatory cytokines such as IL-6, IL-8, IFN-α, and GM-CSF, can prolong the lifespan of neutrophils that should have undergone ‘silent’ apoptosis, by modulating PI3K-Akt signaling and Bcl2 (Bcl-x for neutrophil); Bcl2 can block Bax-mediated release of cytochrome c and therefore attenuate caspase-dependent cell death (86, 87). Although induced Bcl2 in neutrophils does not affect the phagocytic activity of macrophages (88), exposure of pathogen- or host-derived inflammatory cues, such as IL-8, LPS, HMGB1, and S1P, can change the death of neutrophils from apoptosis to ferroptosis and NETosis, which are the main drivers of chronic and systemic autoimmunity (4, 17, 87, 89). HMGB1 released from ferroptotic cells can be taken by phagocytic macrophages to accumulate iron inside the cells, activating M1 macrophages, which then increase production of IL-6, TNF-α, and IL-1β (90). In addition, NET and its component HMGB1 can promote caspase-1-dependent macrophage pyroptosis, another form of cell death, which releases AIM2 inflammasome-mediated IL-1β and accelerates inflammatory cascades while blocking macrophage-mediated efferocytosis with opsonin-related defects (91, 92). The DNA of NET can be recognized by the TLR of macrophages, which phagocyte NETs and NETs do not transfer into phagosome but reside in cytosol; DNA and enzymatic activity of NE from NET stimulate the cyclic GMP-AMP synthase (GAS)-stimulator of interferon genes (STING) pathway that induces type I IFN production and subsequently necroptosis and senescence of macrophages (93, 94). Moreover, the cGAS-STING pathway can turn on an anti-proliferative program and induce Bax-mediated cell death of macrophages, which can counteract the proliferation and efferocytic activity of, but promote macrophage-mediated inflammation (57, 93, 95). Whereas the efferocytosis of DC leads to tolerogenic immature DC with low costimulatory checkpoint ligands, activated neutrophils can recall and directly cluster with DCs, the most potent APC for T lymphocytes, through DC-SIGN and Mac-1, and can mediate maturation of DC, providing TNF-α and other cytokines and granule components (73, 96, 97). Moreover, NET components can drive DC activation to produce type I interferon, and DC can take some NET components as antigens, which may lead to autologous lymphocyte activation (87, 98). Enriched neutrophils in synovial fluid and delayed neutrophil apoptosis in joints may explain the increase of double-stranded DNA and anti-citrullinated antibodies of rheumatoid disease patients (99). Collectively, these facts suggest that neutrophils are crucial immune modulators that affect overall immune response.
Fig. 3.
Impaired efferocytosis and neutrophil death trigger prolonged inflammation. An inflammatory milieu can disrupt apoptosis of neutrophils and make the immune response chronic. (A) A sustained neutrophil life exposed to inflammatory cytokines and other programmed neutrophil deaths, NETosis, secondary necrosis, and ferroptosis. (B) Activation of inflammatory macrophages (Mφ), dendritic cells (DC), and antigen-presenting cell (APC)-like neutrophils. (C) Activation of adaptive immune systems and chronic disease.
CONCLUSION
The functions of neutrophils, the most abundant in the circulation and crucial innate immune cells in host defense, are now getting attention for understanding their following innate/adaptive immune cascades. As a frontline unit of non-specific innate immune responses, the research of neutrophils was focused on migration, detection, and removal of pathogens and damaged host cells (1, 2). However, accumulating evidence suggests that the immunological functions of neutrophil are not limited to initial immune responses. Neutrophils can educate other innate immune cells, such as monocytes, macrophages, and DCs, guiding the direction of immune cascades with production of cytokines and granules and presenting their dead bodies as immune context (3, 32, 52, 53, 93, 94). Moreover, neutrophils directly/indirectly activate lymphocytes, which may aggravate the progress of chronic and autoimmune disease by presenting a source of auto-antigens (4, 17, 87, 89). On the other hand, programed apoptosis of neutrophils initiates immune modulatory phenotype changes of macrophages and DCs as efferocytosis, which can induce tolerogenic APCs that induce immune suppressive Treg and Tr1 (32, 54, 58). Therefore, it is now accepted that excessive activation, dysfunction, or malfunction of neutrophils is closely related to pathogenesis and progression of disease. Hence neutrophils are emerging therapeutic targets for human disease (2, 5). However, further investigations of the roles of trained granulopoiesis and epigenetically modified neutrophils in immune cascades are needed. We hope the gradual progress in the analysis of trained granulopoiesis and heterogeneous neutrophils may lead to further understanding of peripheral tolerance and immune activation.
ACKNOWLEDGEMENTS
This study was supported by the Basic Science Research Program Planning through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT, and Future Planning (NRF-2020M3A9D3038435, NRF-2021R1A2C3011228, NRF-2017R1A5A1014560), and by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI20C 0026). The figures were created with BioRender.com.
Footnotes
CONFLICTS OF INTEREST
The authors have no conflicting interests.
REFERENCES
- 1.Yvan-Charvet L, Ng LG. Granulopoiesis and neutrophil homeostasis: a metabolic, daily balancing act. Trends Immunol. 2019;40:598–612. doi: 10.1016/j.it.2019.05.004. [DOI] [PubMed] [Google Scholar]
- 2.Németh T, Sperandio M, Mócsai A. Neutrophils as emerging therapeutic targets. Nat Rev Drug Discov. 2020;19:253–275. doi: 10.1038/s41573-019-0054-z. [DOI] [PubMed] [Google Scholar]
- 3.Cassatella MA, Östberg NK, Tamassia N, Soehnlein O. Biological roles of neutrophil-derived granule proteins and cytokines. Trends Immunol. 2019;40:648–664. doi: 10.1016/j.it.2019.05.003. [DOI] [PubMed] [Google Scholar]
- 4.Papayannopoulos V. Neutrophil extracellular traps in immunity and disease. Nat Rev Immunol. 2018;18:134–147. doi: 10.1038/nri.2017.105. [DOI] [PubMed] [Google Scholar]
- 5.Liew PX, Kubes P. The neutrophil's role during health and disease. Physiol Rev. 2019;99:1223–1248. doi: 10.1152/physrev.00012.2018. [DOI] [PubMed] [Google Scholar]
- 6.Bae GH, Kim YS, Park JY, et al. Unique characteristics of lung resident neutrophils are maintained by PGE2/PKA/Tgm2-mediated signaling. Blood. 2022;140:889–899. doi: 10.1182/blood.2021014283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kolaczkowska E, Kubes P. Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol. 2013;13:159–175. doi: 10.1038/nri3399. [DOI] [PubMed] [Google Scholar]
- 8.Dinauer MC. Inflammatory consequences of inherited disorders affecting neutrophil function. Blood. 2019;133:2130–2139. doi: 10.1182/blood-2018-11-844563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Thanabalasuriar A, Scott BNV, Peiseler M, et al. Neutrophil extracellular traps confine pseudomonas aeruginosa ocular biofilms and restrict brain invasion. Cell Host Microbe. 2019;25:526–536.:e524. doi: 10.1016/j.chom.2019.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hopke A, Scherer A, Kreuzburg S, et al. Neutrophil swarming delays the growth of clusters of pathogenic fungi. Nat Commun. 2020;11:2031. doi: 10.1038/s41467-020-15834-4.5f5b9de83fd34c48ac2c106384e5e005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Warnatsch A, Tsourouktsoglou T-D, Branzk N, et al. Reactive oxygen species localization programs inflammation to clear microbes of different size. Immunity. 2017;46:421–432. doi: 10.1016/j.immuni.2017.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kienle K, Glaser KM, Eickhoff S, et al. Neutrophils self-limit swarming to contain bacterial growth in vivo. Science. 2021;372:eabe7729. doi: 10.1126/science.abe7729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Margaroli C, Moncada-Giraldo D, Gulick DA, et al. Transcriptional firing represses bactericidal activity in cystic fibrosis airway neutrophils. Cell Rep Med. 2021;2:100239. doi: 10.1016/j.xcrm.2021.100239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Branzk N, Lubojemska A, Hardison SE, et al. Neutrophils sense microbe size and selectively release neutrophil extracellular traps in response to large pathogens. Nat Immunol. 2014;15:1017–1025. doi: 10.1038/ni.2987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li X, Utomo A, Cullere X, et al. The β-glucan receptor Dectin-1 activates the integrin Mac-1 in neutrophils via vav protein signaling to promote Candida albicans clearance. Cell Host Microbe. 2011;10:603–615. doi: 10.1016/j.chom.2011.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Monteith AJ, Miller JM, Maxwell CN, Chazin WJ, Skaar EP. Neutrophil extracellular traps enhance macrophage killing of bacterial pathogens. Sci Adv. 2021;7:eabj2101. doi: 10.1126/sciadv.abj2101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Castanheira FVS, Kubes P. Neutrophils and NETs in modulating acute and chronic inflammation. Blood. 2019;133:2178–2185. doi: 10.1182/blood-2018-11-844530. [DOI] [PubMed] [Google Scholar]
- 18.Lee SK, Kim SD, Kook M, et al. Phospholipase D2 drives mortality in sepsis by inhibiting neutrophil extracellular trap formation and down-regulating CXCR2. J Exp Med. 2015;212:1381–1390. doi: 10.1084/jem.20141813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Enyedi B, Kala S, Nikolich-Zugich T, Niethammer P. Tissue damage detection by osmotic surveillance. Nat Cell Biol. 2013;15:1123–1130. doi: 10.1038/ncb2818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ping L, Wu Y, Hosu BG, Tang JX, Berg HC. Osmotic pressure in a bacterial swarm. Biophys J. 2014;107:871–878. doi: 10.1016/j.bpj.2014.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Diz-Muñoz A, Thurley K, Chintamen S, et al. Membrane tension acts through PLD2 and mTORC2 to limit actin network assembly during neutrophil migration. PLoS Biol. 2016;14:e1002474. doi: 10.1371/journal.pbio.1002474.62f6117bc8374b84a0d564ed0515e6cc [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Manz MG, Boettcher S. Emergency granulopoiesis. Nat Rev Immunol. 2014;14:302–314. doi: 10.1038/nri3660. [DOI] [PubMed] [Google Scholar]
- 23.Yvan-Charvet L, Ng LG. Granulopoiesis and neutrophil homeostasis: a metabolic, daily balancing act. Trends Immunol. 2019;40:598–612. doi: 10.1016/j.it.2019.05.004. [DOI] [PubMed] [Google Scholar]
- 24.Boettcher S, Gerosa RC, Radpour R, et al. Endothelial cells translate pathogen signals into G-CSF-driven emergency granulopoiesis. Blood. 2014;124:1393–1403. doi: 10.1182/blood-2014-04-570762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Danek P, Kardosova M, Janeckova L, et al. β-Catenin-TCF/LEF signaling promotes steady-state and emergency granulopoiesis via G-CSF receptor upregulation. Blood. 2020;136:2574–2587. doi: 10.1182/blood.2019004664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Silva-García O, Valdez-Alarcón JJ, Baizabal-Aguirre VM. Wnt/β-catenin signaling as a molecular target by pathogenic bacteria. Front Immunol. 2019;10:2135. doi: 10.3389/fimmu.2019.02135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Becher B, Tugues S, Greter M. GM-CSF: from growth factor to central mediator of tissue inflammation. Immunity. 2016;45:963–973. doi: 10.1016/j.immuni.2016.10.026. [DOI] [PubMed] [Google Scholar]
- 28.Kim HS, Park MY, Lee SK, Park JS, Lee HY, Bae YS. Activation of formyl peptide receptor 2 by WKYMVm enhances emergency granulopoiesis through phospholipase C activity. BMB Rep. 2018;51:418–423. doi: 10.5483/BMBRep.2018.51.8.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kim SD, Kim Y-K, Lee HY, et al. The agonists of formyl peptide receptors prevent development of severe sepsis after microbial infection. J Immunol Res. 2010;185:4302–4310. doi: 10.4049/jimmunol.1001310. [DOI] [PubMed] [Google Scholar]
- 30.Freitas A, Alves-Filho JC, Victoni T, et al. IL-17 receptor signaling is required to control polymicrobial sepsis. J Immunol. 2009;182:7846–7854. doi: 10.4049/jimmunol.0803039. [DOI] [PubMed] [Google Scholar]
- 31.Taylor PR, Roy S, Leal SM, et al. Activation of neutrophils by autocrine IL-17A-IL-17RC interactions during fungal infection is regulated by IL-6, IL-23, RORγt and dectin-2. Nat Immunol. 2014;15:143–151. doi: 10.1038/ni.2797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stark MA, Huo Y, Burcin TL, Morris MA, Olson TS, Ley K. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity. 2005;22:285–294. doi: 10.1016/j.immuni.2005.01.011. [DOI] [PubMed] [Google Scholar]
- 33.Netea MG, Domínguez-Andrés J, Barreiro LB, et al. Defining trained immunity and its role in health and disease. Nat Rev Immunol. 2020;20:375–388. doi: 10.1038/s41577-020-0285-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kalafati L, Kourtzelis I, Schulte-Schrepping J, et al. Innate immune training of granulopoiesis promotes anti-tumor activity. Cell. 2020;183:771–785. doi: 10.1016/j.cell.2020.09.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kalafati L, Mitroulis I, Verginis P, Chavakis T, Kourtzelis I. Neutrophils as orchestrators in tumor development and metastasis formation. Front Oncol. 2020;10:581457. doi: 10.3389/fonc.2020.581457.4ab3ff6d3e3e4f8e81d2ecbfa79f6f0e [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cortez-Retamozo V, Etzrodt M, Newton A, et al. Origins of tumor-associated macrophages and neutrophils. Proc Natl Acad Sci U S A. 2012;109:2491–2496. doi: 10.1073/pnas.1113744109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Casbon AJ, Reynaud D, Park C, et al. Invasive breast cancer reprograms early myeloid differentiation in the bone marrow to generate immunosuppressive neutrophils. Proc Natl Acad Sci U S A. 2015;112:E566–575. doi: 10.1073/pnas.1424927112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Grzywa TM, Sosnowska A, Matryba P, et al. Myeloid cell-derived arginase in cancer immune response. Front immunol. 2020;11:938. doi: 10.3389/fimmu.2020.00938.737b5bbdd4b6476491006f01b398fe56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Deryugina EI, Zajac E, Juncker-Jensen A, Kupriyanova TA, Welter L, Quigley JP. Tissue-infiltrating neutrophils constitute the major in vivo source of angiogenesis-inducing MMP-9 in the tumor microenvironment. Neoplasia. 2014;16:771–788. doi: 10.1016/j.neo.2014.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Khan N, Downey J, Sanz J, et al. M. tuberculosis reprograms hematopoietic stem cells to limit myelopoiesis and impair trained immunity. Cell. 2020;183:752–770.:e722. doi: 10.1016/j.cell.2020.09.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ebata KO, Hashimoto D, Takahashi S, Hayase E, Ogasawara R, Teshima T. Intestinal microbiota play a critical role in neutrophil engraftment posttransplant and recovery after chemotherapy by stimulating T cell Production of IL-17A. Blood. 2017;130:3166 [Google Scholar]
- 42.Balmer ML, Schürch CM, Saito Y, et al. Microbiota-derived compounds drive steady-state granulopoiesis via MyD88/TICAM signaling. J Immunol Res. 2014;193:5273–5283. doi: 10.4049/jimmunol.1400762. [DOI] [PubMed] [Google Scholar]
- 43.Khosravi A, Yáñez A, Price Jeremy G, et al. Gut microbiota promote hematopoiesis to control bacterial infection. Cell Host Microbe. 2014;15:374–381. doi: 10.1016/j.chom.2014.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Orsini M, Chateauvieux S, Rhim J, et al. Sphingolipid-mediated inflammatory signaling leading to autophagy inhibition converts erythropoiesis to myelopoiesis in human hematopoietic stem/progenitor cells. Cell Death Differ. 2019;26:1796–1812. doi: 10.1038/s41418-018-0245-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Riffelmacher T, Clarke A, Richter FC, et al. Autophagy-dependent generation of free fatty acids is critical for normal neutrophil differentiation. Immunity. 2017;47:466–480. doi: 10.1016/j.immuni.2017.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rožman S, Yousefi S, Oberson K, Kaufmann T, Benarafa C, Simon HU. The generation of neutrophils in the bone marrow is controlled by autophagy. Cell Death Differ. 2015;22:445–456. doi: 10.1038/cdd.2014.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Moorlag SJCFM, Rodriguez-Rosales YA, Gillard J, et al. BCG vaccination induces long-term functional reprogramming of human neutrophils. Cell Rep. 2020;33:108387. doi: 10.1016/j.celrep.2020.108387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lin R, Yi Z, Wang J, Geng S, Li L. Generation of resolving memory neutrophils through pharmacological training with 4-PBA or genetic deletion of TRAM. Cell Death Dis. 2022;13:345. doi: 10.1038/s41419-022-04809-6.7da9eaebe74c4136a4b0164777826c60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.de Bree LCJ, Mourits VP, Koeken VA, et al. Circadian rhythm influences induction of trained immunity by BCG vaccination. J Clin Invest. 2020;130:5603–5617. doi: 10.1172/JCI133934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Casanova-Acebes M, Nicolás-Ávila JA, Li JL, et al. Neutrophils instruct homeostatic and pathological states in naive tissues. J Exp Med. 2018;215:2778–2795. doi: 10.1084/jem.20181468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhang D, Frenette PS. Cross talk between neutrophils and the microbiota. Blood. 2019;133:2168–2177. doi: 10.1182/blood-2018-11-844555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Rankin SM. The bone marrow: a site of neutrophil clearance. J Leukoc Biol. 2010;88:241–251. doi: 10.1189/jlb.0210112. [DOI] [PubMed] [Google Scholar]
- 53.Wang J, Hossain M, Thanabalasuriar A, Gunzer M, Meininger C, Kubes P. Visualizing the function and fate of neutrophils in sterile injury and repair. Science. 2017;358:111–116. doi: 10.1126/science.aam9690. [DOI] [PubMed] [Google Scholar]
- 54.Doran AC, Yurdagul A, Tabas I. Efferocytosis in health and disease. Nat Rev Immunol. 2020;20:254–267. doi: 10.1038/s41577-019-0240-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Farrera C, Fadeel B. Macrophage clearance of neutrophil extracellular traps is a silent process. J Immunol. 2013;191:2647–2656. doi: 10.4049/jimmunol.1300436. [DOI] [PubMed] [Google Scholar]
- 56.Ampomah PB, Cai B, Sukka SR, et al. Macrophages use apoptotic cell-derived methionine and DNMT3A during efferocytosis to promote tissue resolution. Nat Metab. 2022;4:444–457. doi: 10.1038/s42255-022-00551-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gerlach BD, Ampomah PB, Yurdagul A, Jr, et al. Efferocytosis induces macrophage proliferation to help resolve tissue injury. Cell Metab. 2021;33:2445–2463. doi: 10.1016/j.cmet.2021.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kourtzelis I, Li X, Mitroulis I, et al. DEL-1 promotes macrophage efferocytosis and clearance of inflammation. Nat Immunol. 2019;20:40–49. doi: 10.1038/s41590-018-0249-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Schmid M, Gemperle C, Rimann N, Hersberger M. Resolvin D1 polarizes primary human macrophages toward a proresolution phenotype through GPR32. J Immunol. 2016;196:3429–3437. doi: 10.4049/jimmunol.1501701. [DOI] [PubMed] [Google Scholar]
- 60.Fredman G, Ozcan L, Spolitu S, et al. Resolvin D1 limits 5-lipoxygenase nuclear localization and leukotriene B4 synthesis by inhibiting a calcium-activated kinase pathway. Proc Natl Acad Sci U.S.A. 2014;111:14530–14535. doi: 10.1073/pnas.1410851111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Arnardottir H, Thul S, Pawelzik SC, et al. The resolvin D1 receptor GPR32 transduces inflammation resolution and atheroprotection. J Clin Invest. 2021;131:e142883. doi: 10.1172/JCI142883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Luo B, Han F, Xu K, et al. Resolvin D1 Programs inflammation resolution by increasing TGF-β expression induced by dying cell clearance in experimental autoimmune neuritis. J Neurosci. 2016;36:9590–9603. doi: 10.1523/JNEUROSCI.0020-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yuan J, Lin F, Chen L, et al. Lipoxin A4 regulates M1/M2 macrophage polarization via FPR2-IRF pathway. Inflammopharmacology. 2022;30:487–498. doi: 10.1007/s10787-022-00942-y. [DOI] [PubMed] [Google Scholar]
- 64.Prieto P, Cuenca J, Través PG, Fernández-Velasco M, Martín-Sanz P, Boscá L. Lipoxin A4 impairment of apoptotic signaling in macrophages: implication of the PI3K/Akt and the ERK/Nrf-2 defense pathways. Cell Death Differ. 2010;17:1179–1188. doi: 10.1038/cdd.2009.220. [DOI] [PubMed] [Google Scholar]
- 65.Godson C, Mitchell S, Harvey K, Petasis NA, Hogg N, Brady HR. Cutting edge: lipoxins rapidly stimulate nonphlogistic phagocytosis of apoptotic neutrophils by monocyte-derived macrophages. J Immunol. 2000;164:1663–1667. doi: 10.4049/jimmunol.164.4.1663. [DOI] [PubMed] [Google Scholar]
- 66.Son M, Porat A, He M, et al. C1q and HMGB1 reciprocally regulate human macrophage polarization. Blood. 2016;128:2218–2228. doi: 10.1182/blood-2016-05-719757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Benoit ME, Clarke EV, Morgado P, Fraser DA, Tenner AJ. Complement protein C1q directs macrophage polarization and limits inflammasome activity during the uptake of apoptotic cells. J Immunol. 2012;188:5682–5693. doi: 10.4049/jimmunol.1103760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Tran MTN, Hamada M, Jeon H, et al. MafB is a critical regulator of complement component C1q. Nat Commun. 2017;8:1700. doi: 10.1038/s41467-017-01711-0.8ad6548fe15640be8a3fcc672a1b03f2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Croker BA, Metcalf D, Robb L, et al. SOCS3 is a critical physiological negative regulator of G-CSF signaling and emergency granulopoiesis. Immunity. 2004;20:153–165. doi: 10.1016/S1074-7613(04)00022-6. [DOI] [PubMed] [Google Scholar]
- 70.Lee CK, Raz R, Gimeno R, et al. STAT3 is a negative regulator of granulopoiesis but is not required for G-CSF-dependent differentiation. Immunity. 2002;17:63–72. doi: 10.1016/S1074-7613(02)00336-9. [DOI] [PubMed] [Google Scholar]
- 71.Hutchins AP, Diez D, Miranda-Saavedra D. The IL-10/STAT3-mediated anti-inflammatory response: recent developments and future challenges. Brief Funct Genom. 2013;12:489–498. doi: 10.1093/bfgp/elt028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Morris R, Kershaw NJ, Babon JJ. The molecular details of cytokine signaling via the JAK/STAT pathway. Protein Sci. 2018;27:1984–2009. doi: 10.1002/pro.3519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Clarke EV, Weist BM, Walsh CM, Tenner AJ. Complement protein C1q bound to apoptotic cells suppresses human macrophage and dendritic cell-mediated Th17 and Th1 T cell subset proliferation. J Leukoc Biol. 2015;97:147–160. doi: 10.1189/jlb.3A0614-278R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Fullerton JN, Gilroy DW. Resolution of inflammation: a new therapeutic frontier. Nat Rev Drug Discov. 2016;15:551–567. doi: 10.1038/nrd.2016.39. [DOI] [PubMed] [Google Scholar]
- 75.Roncarolo MG, Gregori S, Bacchetta R, Battaglia M, Gagliani N. The biology of T regulatory type 1 cells and their therapeutic application in immune-mediated diseases. Immunity. 2018;49:1004–1019. doi: 10.1016/j.immuni.2018.12.001. [DOI] [PubMed] [Google Scholar]
- 76.Lewkowicz N, Mycko MP, Przygodzka P, et al. Induction of human IL-10-producing neutrophils by LPS-stimulated Treg cells and IL-10. Mucosal Immunol. 2016;9:364–378. doi: 10.1038/mi.2015.66. [DOI] [PubMed] [Google Scholar]
- 77.Khoyratty TE, Ai Z, Ballesteros I, et al. Distinct transcription factor networks control neutrophil-driven inflammation. Nat Immunol. 2021;22:1093–1106. doi: 10.1038/s41590-021-00968-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wang X, Cai J, Lin B, et al. GPR34-mediated sensing of lysophosphatidylserine released by apoptotic neutrophils activates type 3 innate lymphoid cells to mediate tissue repair. Immunity. 2021;54:1123–1136.:e1128. doi: 10.1016/j.immuni.2021.05.007. [DOI] [PubMed] [Google Scholar]
- 79.Fischer A, Wannemacher J, Christ S, et al. Neutrophils direct preexisting matrix to initiate repair in damaged tissues. Nat Immunol. 2022;23:518–531. doi: 10.1038/s41590-022-01166-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lim K, Hyun YM, Lambert-Emo K, et al. Neutrophil trails guide influenza-specific CD8⁺ T cells in the airways. Science. 2015;349:aaa4352. doi: 10.1126/science.aaa4352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Vono M, Lin A, Norrby-Teglund A, Koup RA, Liang F, Loré K. Neutrophils acquire the capacity for antigen presentation to memory CD4+ T cells in vitro and ex vivo. Blood. 2017;129:1991–2001. doi: 10.1182/blood-2016-10-744441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Marini O, Costa S, Bevilacqua D, et al. Mature CD10+ and immature CD10− neutrophils present in G-CSF-treated donors display opposite effects on T cells. Blood. 2017;129:1343–1356. doi: 10.1182/blood-2016-04-713206. [DOI] [PubMed] [Google Scholar]
- 83.Mysore V, Cullere X, Mears J, et al. FcγR engagement reprograms neutrophils into antigen cross-presenting cells that elicit acquired anti-tumor immunity. Nat Commun. 2021;12:4791. doi: 10.1038/s41467-021-24591-x.86a5a1c6c18f436081a1927c18c8de2d [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lu T, Kobayashi SD, Quinn MT, Deleo FR. A NET outcome. Front Immunol. 2012;3:365. doi: 10.3389/fimmu.2012.00365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bedoui S, Herold MJ, Strasser A. Emerging connectivity of programmed cell death pathways and its physiological implications. Nat Rev Mol Cell Biol. 2020;21:678–695. doi: 10.1038/s41580-020-0270-8. [DOI] [PubMed] [Google Scholar]
- 86.Brostjan C, Oehler R. The role of neutrophil death in chronic inflammation and cancer. Cell Death Discov. 2020;6:26. doi: 10.1038/s41420-020-0255-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Li P, Jiang M, Li K, et al. Glutathione peroxidase 4-regulated neutrophil ferroptosis induces systemic autoimmunity. Nat Immunol. 2021;22:1107–1117. doi: 10.1038/s41590-021-00993-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lagasse E, Weissman IL. bcl-2 inhibits apoptosis of neutrophils but not their engulfment by macrophages. J Exp Med. 1994;179:1047–1052. doi: 10.1084/jem.179.3.1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zhao X, Yang L, Chang N, et al. Neutrophils undergo switch of apoptosis to NETosis during murine fatty liver injury via S1P receptor 2 signaling. Cell Death Dis. 2020;11:379. doi: 10.1038/s41419-020-2582-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Yang Y, Wang Y, Guo L, Gao W, Tang T-L, Yan M. Interaction between macrophages and ferroptosis. Cell Death Dis. 2022;13:355. doi: 10.1038/s41419-022-04775-z.b26221cd93184cadbafc41cb6cac2324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Chen K, Murao A, Arif A, et al. Inhibition of efferocytosis by extracellular CIRP-Induced neutrophil extracellular traps. J Immunol. 2021;206:797–806. doi: 10.4049/jimmunol.2000091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Chen L, Zhao Y, Lai D, et al. Neutrophil extracellular traps promote macrophage pyroptosis in sepsis. Cell Death Dis. 2018;9:597. doi: 10.1038/s41419-018-0538-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Decout A, Katz JD, Venkatraman S, Ablasser A. The cGAS-STING pathway as a therapeutic target in inflammatory diseases. Nat Rev Immunol. 2021;21:548–569. doi: 10.1038/s41577-021-00524-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Apel F, Andreeva L, Knackstedt LS, et al. The cytosolic DNA sensor cGAS recognizes neutrophil extracellular traps. Sci Signal. 2021;14:eaax7942. doi: 10.1126/scisignal.aax7942. [DOI] [PubMed] [Google Scholar]
- 95.Boada-Romero E, Martinez J, Heckmann BL, Green DR. The clearance of dead cells by efferocytosis. Nat Rev Mol Cell Biol. 2020;21:398–414. doi: 10.1038/s41580-020-0232-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.van Gisbergen KP, Sanchez-Hernandez M, Geijtenbeek TB, van Kooyk Y. Neutrophils mediate immune modulation of dendritic cells through glycosylation-dependent interactions between Mac-1 and DC-SIGN. J Exp Med. 2005;201:1281–1292. doi: 10.1084/jem.20041276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Steinman RM, Turley S, Mellman I, Inaba K. The induction of tolerance by dendritic cells that have captured apoptotic cells. J Exp Med. 2000;191:411–416. doi: 10.1084/jem.191.3.411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Sangaletti S, Tripodo C, Chiodoni C, et al. Neutrophil extracellular traps mediate transfer of cytoplasmic neutrophil antigens to myeloid dendritic cells toward ANCA induction and associated autoimmunity. Blood. 2012;120:3007–3018. doi: 10.1182/blood-2012-03-416156. [DOI] [PubMed] [Google Scholar]
- 99.Fresneda Alarcon M, McLaren Z, Wright HL. Neutrophils in the pathogenesis of rheumatoid arthritis and systemic lupus erythematosus: same foe different M.O. Front Immunol. 2021;12:649693. doi: 10.3389/fimmu.2021.649693.f1b6b980c459419ba51babdb1a31b2af [DOI] [PMC free article] [PubMed] [Google Scholar]