Abstract
Background and Aims
Metabolic associated fatty liver disease (MAFLD) is a serious condition, and a simple method is needed for practitioners to identify patients with the disease and have a high risk of disease progression.
Methods
We developed and validated a nomogram for fatty liver disease and reclassified the risk factors for MAFLD. The development cohort had 335 patients who received bioelectrical impedance analysis and liver ultrasound attenuation measurements at Shenzhen People’s Hospital between September 2020 and June 2021. The validation cohort had 200 patients from other hospitals who received the same evaluation. A random forest procedure and binary logistic analysis were used to screen for risk factors, establish a fatty liver disease predictive model, and forecast the risk of MAFLD. The performance of the nomogram was evaluated by measurement of discrimination, calibration, and clinical usefulness.
Results
The nomogram provided good predictions in a model that included body mass index (BMI) and waist circumference. The areas under the curve of the nomogram were 0.793 in the development cohort and 0.774 in the validation cohort. The nomogram performed well for calibration, category-free net reclassification improvement, and integrated discrimination improvement. Decision curve analysis indicated the nomogram performed better than BMI for predicting net outcome.
Conclusions
The nomogram was an effective screening tool for fatty liver disease, and for those overweight individuals, may help physicians make appropriate decisions regarding treatment of MAFLD.
Keywords: MAFLD; Obesity; Nomogram; Body composition; Liver ultrasound attenuation, LiSA
Introduction
Metabolic associated fatty liver disease (MAFLD) has become more prevalent in recent decades.1 A recent systematic review and meta-analysis estimated that the prevalence of MAFLD in Asia was 29.62%.2 Overweight and obesity are the main risk factors for MAFLD,3 and obesity and obesity-related diseases are increasing worldwide. In the past four decades, the prevalence of overweight has increased by 47%,4 and more than 25% of adults worldwide are now overweight or obese,5 and consequently have a higher mortality rate than those of normal weight.6
The diagnosis of MAFLD requires assessment of liver histology, measurement of noninvasive biomarkers, or imaging evidence.3 A liver biopsy is an invasive procedure and is unsuitable for routine screening. Ultrasound is thus the most commonly method of diagnosing liver diseases in clinical practice. However, the subjectivity of a visual assessment of fatty liver on a gray-scale results in substantial interobserver variability7 and decreased sensitive for the detection of a mildly fatty liver.8
Acoustic attenuation parameters have been reported to quantify the fatty liver content.9,10 The controlled attenuation parameter (CAP) does not provide visualization, which may decrease reliability. CAP is also unsuitable for individuals who are overweight. To overcome the disadvantages of CAP, the Mindray diagnostic ultrasound system uses liver ultrasound attenuation (LiSA),11 that includes a visualization method that greatly improves stability and precision in detection of MAFLD in individuals with different body types.12 In particular, a comparison of the diagnostic performances of LiSA and CAP, with liver biopsy as the gold standard, indicated that LiSA had higher area under the curve values (0.883 vs. 0.778), sensitivity (76.08% vs. 72.10%), and specificity (96.23% vs. 91.53%) than CAP.12
It is also important to provide patients with rapid diagnosis and risk classification for MAFLD. This information is important feedback for individuals who want to lose weight, and it can also increase their motivation for weight management. However, ultrasound is not as convenient and effective as simple body weight monitoring for patients who have MAFLD and are receiving care at weight-management institutions or groups. For those patients, it is assumed that the pathologic stages of fatty liver and the risk of MALFD can be monitored by simple body weight measurement.
Bioelectrical impedance analysis (BIA) is a simple, fast, and noninvasive method of assessing body composition.13–15 The nomogram used to estimate body composition from BIA measurements is a reliable and convenient tool for quantifying risk, and is widely used for risk stratification.16 This study describes the development and validation of a risk-stratified nomogram that combines data from BIA and LiSA to provide a method for simple, real-time monitoring of MAFLD.
Methods
Development cohort
This study had a double-blind design, in that the participants and evaluators were unaware of group allocations. The study was approved by the Shenzhen People’s Hospital Ethics Committee, and participants provided written consent prior to all procedures. Between September 2020 and June 2021, 729 individuals received weight-health assessments in the Nutrition Department of the Shenzhen People’s Hospital (Guangdong, China). All received body composition analysis using BIA. The exclusion criteria were: (1) no LiSA assessment (n=459); (2) presence of a severe systemic disease, such as a severe infection, liver dysfunction, or kidney dysfunction (n=38); and (3) more than 1 week between the LiSA assessment and BAI measurement (n=172). The remaining development cohort included 335 participants (Supplementary Fig. 1). A comparison of included and excluded subjects found no significant differences in age, sex, body mass index (BMI), and waist circumference (WC).
Participant characteristics included age, height, sex, and weight, and intracellular water (ICW), extracellular water (ECW), total body water (TBW), protein, mineral, fat, soft lean mass (SLM), fat-free mass (FFM), skeletal muscle mass (SMM), percent body fat (PBF), BMI, ECW/TBW, body cell mass (BCM), bone mineral content (BMC), waist circumference (WC), visceral fat area (VFA), basal metabolic rate (BMR), and TBW/FFM were obtained with a InBody S10 instrument (Seoul, Korea). Fasting blood samples were collected for analysis of albumin, globulin, alanine aminotransferase (ALT), alanine aminotransferase (AST), gamma-glutamyl transpeptidase (GGT), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), triglycerides, total cholesterol, alkaline phosphatase (ALP), fasting plasma glucose, serum creatinine (SCr), homocysteine, and uric acid.
Validation cohort
Between September 2020 and June 2021, 475 consecutive patients at the Longhua and Bantian Branches of Shenzhen People’s Hospital received the same screening as the development cohort. This validation cohort included 200 participants (Supplementary Fig. 1). The Ethics Committees of each hospital branch approved the study.
Bioelectrical impedance analysis (BIA)
All participants were assessed with a BIA (InBody S10, Seoul, Korea) at an ambient temperature of 20°C to 25°C. All participants were required to remove metal objects, shoes and socks, wear light clothing. wipe their hands and feet with the InBody wet tissues, and then stand on the instrument while making sure their heels were flush with the electrodes. They then grasped the handle, placed a thumb on the oval electrode, kept their arms straight, and avoided touching other parts of the body. They were asked to stay relaxed until the test was complete.
Measurement of hepatic steatosis
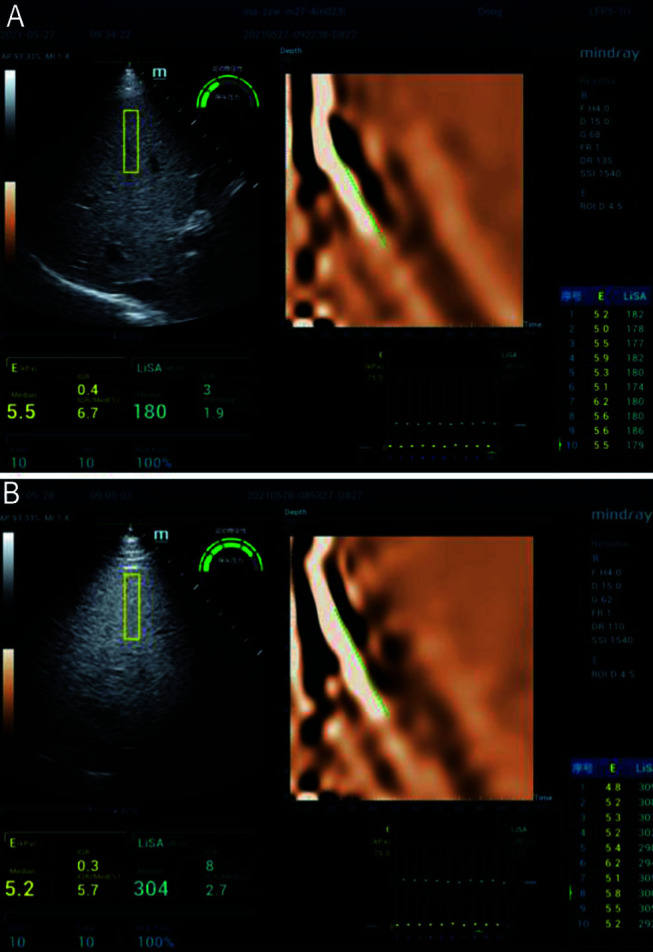
The LiSA measurements were performed with a Mindray noninvasive liver diagnostic ultrasound system and a Resona Hepatus fibrosis evaluation system (LFP5-1U; Mindray, Shenzhen, China) (Fig. 1).11,12 The probe was placed parallel to the optimal intercostal space and perpendicular to the liver capsule while the subject was in a supine position with the right arm lifted. Using real-time B-Mode imaging guidance, the technician located a liver segment that had no vessels larger than 3 mm in diameter, and then placed the rectangular region of interest away from the liver capsule. The patient was asked to breathe normally or to hold their breath for 5 to 6 s to avoid any influence of respiratory movements. The final LiSA value was the median of at least 10 measurements, and an interquartile range less than 40 dB/m was used as a quality-control criterion.12 The LiSA threshold for the discrimination of hepatic steatosis has been previously reported as 238 dB/m.
Fig. 1. Representative liver ultrasound attenuation (LiSA) measurements of a patient (A) without fatty liver and a patient (B) with fatty liver.
Statistical analysis
The Wilcoxon-Mann-Whitney test or Fisher’s exact test was used to assess differences in the distribution of variables within and between cohorts. A random forest procedure was used to select variables for constructing a nomogram for identification of fatty liver in the development cohort. Then, based on evaluations of clinical and statistical significance,16 a binary logistic model was used to select the final covariates.17 Calibration curves were plotted to assess the calibration of the fatty liver nomogram, and the Hosmer-Lemeshow test was used to evaluate goodness of model fit. Harrell’s C-statistic (concordance index), the net reclassification index (NRI), and the integrated discrimination improvement (IDI) were also determined.18–20 Decision curve analyses were applied to the validation cohort to compare nomogram performance with other methods of predicting fatty liver.21 The statistical analysis were performed with R version 3.6.2 (http://www.r-project.org/).
Results
Characteristics of the development and validation cohorts
Development and validation cohorts were similar in age, height, gender, fat, PBF, ECW/TBW, WC, VFA, and TBW/FFM (Table 1). The two groups were also similar in the presence of fatty liver, 179 in the development cohort (53.4%) and 98 (49.0%) in the validation cohort. The two groups differed in a number of other anthropometric measurements.
Table 1. Anthropometric characteristics of participants in the development cohort (n=335) and the validation cohort (n=200) based on biometrical impedance analysis.
| Characteristic | Development cohort | Validation cohort | p a |
|---|---|---|---|
| Age in year | 32.81 (10.76) | 33.16 (11.02) | 0.716 |
| Height in cm | 164.76 (8.11) | 164.26 (8.94) | 0.511 |
| Sex, n (%) | |||
| Female | 208 (62.1) | 138 (69.0) | 0.127 |
| Male | 127 (37.9) | 62 (31.0) | |
| Weight in kg | 80.58 (16.74) | 76.70 (15.14) | 0.007* |
| ICW in L | 23.03 (4.89) | 22.04 (4.66) | 0.02* |
| ECW in L | 13.94 (2.85) | 13.34 (2.67) | 0.016* |
| TBW in L | 36.97 (7.72) | 35.37 (7.32) | 0.018* |
| Protein in kg | 9.95 (2.11) | 9.52 (2.02) | 0.02* |
| Mineral in kg | 3.46 (0.72) | 3.28 (0.67) | 0.003* |
| Fat in kg | 30.18 (9.84) | 28.52 (9.14) | 0.053 |
| SLM in kg | 47.53 (9.95) | 45.47 (9.45) | 0.018* |
| FFM in kg | 50.40 (10.52) | 48.18 (9.99) | 0.016* |
| SMM in kg | 28.04 (6.38) | 26.74 (6.08) | 0.02* |
| PBF in % | 37.07 (6.97) | 36.89 (7.17) | 0.776 |
| BMI in kg/m2 | 29.52 (4.82) | 28.32 (4.50) | 0.005* |
| ECW/TBW | 0.38 (0.01) | 0.38 (0.01) | 0.802 |
| BCM in kg | 32.99 (7.00) | 31.56 (6.68) | 0.02* |
| BMC in kg | 2.86 (0.59) | 2.70 (0.56) | 0.003* |
| WC in cm | 96.71 (12.96) | 94.76 (11.86) | 0.084 |
| VFA in cm2 | 137.22 (46.32) | 132.38 (44.77) | 0.237 |
| BMR in kcal | 1,458.56 (227.14) | 1,410.69 (215.81) | 0.017* |
| TBW/FFM, % | 73.37 (0.31) | 73.42 (0.27) | 0.064 |
| Fatty liver, % | |||
| No | 156 (46.6) | 98 (49.0) | 0.649 |
| Yes | 179 (53.4) | 102 (51.0) |
Data are number (%) or mean±SD. *p<0.05. aWilcoxon-Mann-Whitney (continuous variables) or Fisher’s exact test (other categorical variables). BCM, body cell mass; BMC, bone mineral content; BMI, body mass index; BMR, basal metabolic rate. ECW, extracellular water; FFM, fat-free mass; ICW, intracellular water; PBF, percent body fat; SLM, soft lean mass; SMM, skeletal muscle mass; TBW, total body water; VFA, visceral fat area; WC, waist circumference.
Serological characteristics of fatty liver and nonfatty liver
The serological parameters of the fatty liver and the nonfatty liver groups in the development and validation cohorts are shown in Table 2. As expected, individuals in the fatty liver group had significantly higher ALT, AST, GGT, triglyceride, total cholesterol, homocysteine, and blood uric acid levels, a significantly lower HDL-C levels than the nonfatty liver group. Differences in albumin, globulin, LDL-C, ALP, fasting plasma glucose, and SCr were not significant.
Table 2. Serological characteristics of patients with fatty liver (n=281) and nonfatty liver (n=254).
| Characteristic | Fatty livera | Nonfatty livera | p |
|---|---|---|---|
| Albumin in g/L | 50.4±29.02 | 50.25±35.29 | 0.9661 |
| Globulin in g/L | 27.77±3.99 | 27.28±3.46 | 0.2977 |
| ALT in IU/L | 45.37±37.27 | 19.56±10.06 | <0.0001 |
| AST in IU/L | 29.49±19.27 | 19.15±10.18 | <0.0001 |
| GGT in IU/L | 40.05±29.65 | 20.26±11.81 | <0.0001 |
| HDL-C in mmol/L | 1.18±0.25 | 1.37±0.31 | <0.0001 |
| LDL-C in mmol/L | 3.21±0.78 | 3.16±2.6 | 0.8311 |
| Triglycerides in mmol/L | 1.71±1.24 | 1.09±0.53 | <0.0001 |
| Total cholesterol in mmol/L | 5.3±1.86 | 4.9±1.07 | 0.0176 |
| Homocysteine in µmol/L | 12±4.79 | 10.53±2.93 | 0.0028 |
| ALP in IU/L | 66.09±39.6 | 59.36±23.32 | 0.1508 |
| Fasting plasma glucose in mmol/L | 5.24±0.77 | 4.95±0.47 | 0.1669 |
| Blood uric acid in µmol/L | 428.65±110.35 | 361.23±86.14 | <0.0001 |
| SCr in µmol/L | 73.28±41.35 | 66±13.01 | 0.2789 |
aData are mean±SD. ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transpeptidase; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SD, standard deviation.
Factors predictive of fatty liver
We used a random forest procedure to select variables for the nomogram in the development cohort (Supplementary Fig. 2). The first six variables were WC, BMI, weight, age, VFA, and PBF. Multivariate analysis showed that WC (p=0.0023) and BMI (p=0.0879) were the best predictors of fatty liver (Table 3). In other words, increased WC and BMI were associated with an increased risk of fatty liver. We derived a nomogram for these factors from their β coefficients (Table 3).
Table 3. Risk factors for fatty liver.
| Intercept and variable | β | Odds ratio | 95% CI | p |
|---|---|---|---|---|
| Intercept | −9.898 | <0.001 | ||
| BMI | 0.109 | 1.116 | 0.985, 1.266 | 0.0870 |
| WC | 0.071 | 1.074 | 1.027, 1.126 | 0.0023 |
β, regression coefficient; BMI, body mass index; CI, confidence interval; WC, waist circumference.
Nomogram for fatty liver and MAFLD
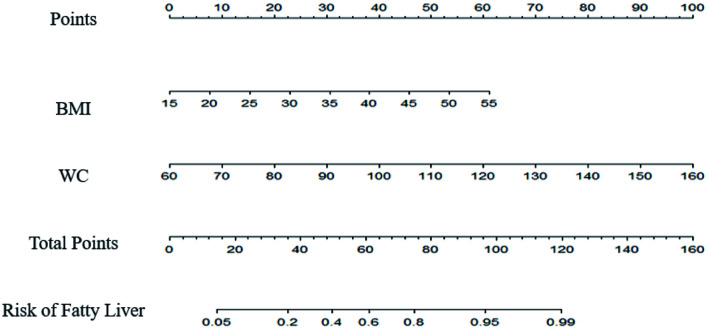
The BMI and WC data were used to establish a nomogram for prediction of fatty liver and MAFLD (Fig. 2). If a subject has a BMI of 24 or more and a WC of 90 cm or more for men and 80 cm or more for women, then the predicted probability of a fatty liver is the predicted probability of MAFLD.
Fig. 2. Nomogram for prediction of fatty liver and MAFLD.
Procedure: First, identify the waist circumference (WC), draw a vertical line to the points axis, and determine the score. Second, repeat this process for BMI. Third, sum the two scores, and identify this sum on the total points axis. Fourth, draw a vertical line from the total points” axis to the probability axis to determine the probability. When a subject has a BMI >24 and a WC ≥90 cm (men) or ≥80 cm (women), the predicted probability of fatty liver is the predicted probability of MAFLD. MAFLD, metabolic associated fatty liver disease.
Discrimination and calibration of the nomogram
The calibration curves of the nomogram indicated good consistency between the predicted and observed probabilities of fatty liver in the development cohort (Supplementary Fig. 3A). The Hosmer-Lemeshow test showed the data had a good fit to the model (p=0.514), indicating that the nomogram was well calibrated. The concordance index was 0.973, indicating the nomogram had good discriminative ability. Further analysis indicated the nomogram slightly improved the concordance index of the univariate models, but including both variables for development of the nomogram led to no significant difference (p=0.358 for BMI+WC vs. WC and p=0.370 for BMI+WC vs. BMI, Table 4). However, the category-free net reclassification improvement (cf-NRI) and the integrated discrimination improvement (IDI) were both >0. The results indicated the nomogram provided significantly better predictive performance than the univariate models (Table 4). The nomogram also had good calibration in the validation cohort (Supplementary Fig. 3B). In particular, the p-value was 0.362 in the Hosmer-Lemeshow test and the concordance index was 0.774, indicating good discriminative ability (Table 4).
Table 4. Discrimination and reclassification performance with 95% CIs for the nomogram (BMI+WC) and for univariable models of BMI and WC alone.
| Cohort | Model | C-statistic (95% CI) | cf-NRI (95% CI) | IDI (95% CI) |
|---|---|---|---|---|
| Development | BMI+WC | 0.793 (0.745, 0.840) | ||
| BMI | 0.784(0.735, 0.833) | 0.308 (0.096, 0.520)* | 0.0212 (0.005, 0.037)* | |
| WC | 0.787(0.739, 0.835) | 0.253 (0.040, 0.466)* | 0.0084 (−4×10−4, 0.017) | |
| Validation | BMI+WC | 0.775 (0.710, 0.837) | ||
| BMI | 0.752(0.686, 0.819) | 0.3617 (0.089, 0.634)* | 0.0367 (0.010, 0.063)* | |
| WC | 0.775(0.711, 0.839) | 0.160 (−0.116, 0.436) | 0.0018 (−0.005, 0.009) |
*p <0.05. BMI, body mass index; cf-NRI, category-free net reclassification improvement; CI, confidence interval; IDI, integrated discrimination improvement; WC, waist circumference.
Clinical use
We used the results of decision curve analyses to compare the performance of the nomogram with the performances of univariable models for each of the variables selected by the random forest procedure (Fig. 3). The results indicate that the net benefit of predicting fatty liver from the nomogram (BMI+WC) and a univariable model of WC alone were comparable in the validation cohort. However, each provided a greater net benefit than predicting fatty liver by BMI alone.
Fig. 3. Decision curve analysis of the validation cohort.
BMI, body mass index; WC, waist circumference.
Discussion
We used WC and BMI to develop and validate a risk-stratified nomogram for the diagnosis of fatty liver. The nomogram had good calibration based on comparison of the development and validation cohorts, good discriminative ability, and had general applicability. Decision curve analyses indicated that the nomogram had greater net benefit than the use of BMI alone at all threshold probabilities. The nomogram thus provided improved accuracy for the diagnosis of fatty liver. For overweight and obese individuals, a fatty liver is a necessary condition for the diagnosis of MAFLD.22 Therefore, for those with BMIs of 24 or more, the risk-stratified nomogram presented here can be used as a risk prediction model for MAFLD.
Previous studies showed that anthropometric parameters combined with noninvasive biochemical indicators can be used to provide instruments for the simple and early detection and evaluation of nonalcoholic fatty liver. The instruments include the hepatic steatosis index (HSI), the fatty liver index (FLI), the visceral adiposity index (VAI), and the Zhejiang University (ZJU) index.23,24 However, each of those methods has limitations. For example, the FLI, which is based on BMI, WC, triglycerides, and GGT, can diagnose fatty liver with an AUC of 0.84 (95% CI: 0.81, 0.87),25 but is not applicable to subjects with hepatitis B or C virus infections. The HSI, which is based on ALT/AST, BMI, sex, and diabetes, can diagnose fatty liver with an AUC of 0.812 (95% CI: 0.801, 0.824),26 but is not applicable to patients who have fatty liver caused by alcohol, drugs, or viral liver disease. The VAI is based on histological results and high viral loads in patients with chronic hepatitis C, but is not a predictor of NAFLD.27–29 Furthermore, the three models were developed in studies of Westerners and may not be suitable for Chinese people. Although the ZJU index is a powerful indicator for identifying nonalcoholic fatty liver in Chinese patients and severely obese North American women,24,30 it is not applicable to the newly defined condition of MAFLD.3
An international expert consensus statement recently recommended replacing the term nonalcoholic fatty liver disease (NAFLD) with MAFLD. MAFLD has affirmative diagnostic criteria that are not based on the exclusion of factors such as alcohol consumption or other concomitant liver diseases, and the key driver is metabolic dysfunction.3,22,31 A recent study found that MAFLD was more accurate in identifying subjects with fatty liver disease and a high risk of disease progression,32 and patients who have an increased risk of poor hepatic or cardiovascular outcomes.31 MAFLD can also help identify individuals who have more severe disease and would benefit most from early intervention.33 That is one of the significant findings of the present study.
The main results of this study are that simple anthropometric indicators can be used to predict fatty liver disease and that we reclassified the risk factors for MAFLD. However, our nomogram is only suitable for a those with a BMI of ≥24 and a WC of ≥ 90 cm or more in men or 80 cm or more in women. If these requirements are not met, and the risk of fatty liver is considered great, the patient can be reminded to check whether there are co-occurring metabolic diseases, such as diabetes, to rule out MAFLD. Even though the discriminability of our model was not as high as a previous model,23,24 it allows a preliminary assessment of MAFLD based on simple measurements of BMI and WC in people who are overweight. The nomogram presented here is a new, promising, and simple method for prediction of MAFLD.
There are several limitations in our study. First, the sample size was limited, even though this was a retrospective study of patients at three centers. Second, we did not use liver biopsy as the gold standard, and LiSA can only screen for significant steatosis (≥10%). Third, some patients were excluded because of missing data, and may have led to some bias. Finally, the subjects selected in our sample were overweight, so our results are not applicable to the general population. Application of this approach to the general population is a topic of our forthcoming research. Overall, overweight and obesity are major worldwide public health challenges for the 21st Century. Our study used WC and BMI to establish a risk-stratified nomogram that screens for the risk of MAFLD in overweight or obese populations. For high-risk subjects, early lifestyle interventions may help to reduce the risk of adverse outcomes.
Supporting information
BCM, body cell mass; BMC, bone mineral content; BMI, body mass index; BMR, basal metabolic rate. ECW, extracellular water; FFM, fat-free mass; ICW, intracellular water; PBF, percent body fat; SLM, soft lean mass; SMM, skeletal muscle mass; TBW, total body water; VFA, visceral fat area; WC, waist circumference.
(A) In the development and the (B) validation cohorts. Y-axis, actual fatty liver probability; X-axis, predicted fatty liver risk; black diagonal line, reference line (ideal nomogram).
Acknowledgments
Thanks for the support and help of our D.team of ultrasound department Shenzhen People’s Hospital.
Abbreviations
- ALP
alkaline phosphatase
- ALT
alanine aminotransferase
- AST
aspartate aminotransferase
- BCM
body cell mass
- BIA
bioelectrical impedance analysis
- BMC
bone mineral content
- BMI
body mass index
- BMR
basal metabolic rate
- CAP
controlled attenuation parameter
- cf-NRI
category-free net reclassification improvement
- CI
confidence interval
- ECW
extracellular water
- FFM
fat-free mass
- GGT
gamma-glutamyl transpeptidase
- HDL-C
high-density lipoprotein cholesterol
- ICW
intracellular water
- IDI
integrated discrimination improvement
- LDL-C
low-density lipoprotein cholesterol
- LiSA
liver ultrasound attenuation
- MAFLD
metabolic associated fatty liver disease
- PBF
percent body fat
- SLM
soft lean mass
- SMM
skeletal muscle mass
- TBW
total body water
- VFA
visceral fat area
- WC
waist circumference
Data sharing statement
No additional data are available.
References
- 1.Sarin SK, Kumar M, Eslam M, George J, Al Mahtab M, Akbar SMF, et al. Liver diseases in the Asia-Pacific region: a Lancet Gastroenterology & Hepatology Commission. Lancet Gastroenterol Hepatol. 2020;5(2):167–228. doi: 10.1016/S2468-1253(19)30342-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Li J, Zou B, Yeo YH, Feng Y, Xie X, Lee DH, et al. Prevalence, incidence, and outcome of non-alcoholic fatty liver disease in Asia, 1999-2019: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2019;4(5):389–398. doi: 10.1016/S2468-1253(19)30039-1. [DOI] [PubMed] [Google Scholar]
- 3.Eslam M, Newsome PN, Sarin SK, Anstee QM, Targher G, Romero-Gomez M, et al. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J Hepatol. 2020;73(1):202–209. doi: 10.1016/j.jhep.2020.03.039. [DOI] [PubMed] [Google Scholar]
- 4.Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384(9945):766–781. doi: 10.1016/S0140-6736(14)60460-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Luis J, Coronado-Cáceres, Rabadán-Chávez G, Mojica L, Stampa IJF. Cocoa (Theobroma cacao L.) Seed Proteins’ Anti-Obesity Potential through Lipase Inhibition Using In Silico, In Vitro and In Vivo Models. Foods. 2020;9(10):1359. doi: 10.3390/foods9101359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kramer CK, Zinman B, Retnakaran R. Are metabolically healthy overweight and obesity benign conditions?: A systematic review and meta-analysis. Ann Intern Med. 2013;159(11):758–769. doi: 10.7326/0003-4819-159-11-201312030-00008. [DOI] [PubMed] [Google Scholar]
- 7.Strauss S, Gavish E, Gottlieb P, Katsnelson L. Interobserver and intraobserver variability in the sonographic assessment of fatty liver. AJR Am J Roentgenol. 2007;189(6):W320–323. doi: 10.2214/AJR.07.2123. [DOI] [PubMed] [Google Scholar]
- 8.Tan CH, Venkatesh SK. Magnetic Resonance Elastography and Other Magnetic Resonance Imaging Techniques in Chronic Liver Disease: Current Status and Future Directions. Gut Liver. 2016;10(5):672–686. doi: 10.5009/gnl15492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dasarathy S, Dasarathy J, Khiyami A, Joseph R, Lopez R, McCullough AJ. Validity of real time ultrasound in the diagnosis of hepatic steatosis: a prospective study. J Hepatol. 2009;51(6):1061–1067. doi: 10.1016/j.jhep.2009.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sasso M, Beaugrand M, Ledinghen VD, Douvin C, Marcellin P, Poupon R, et al. Controlled Attenuation Parameter (CAP): A Novel VCTE™ Guided Ultrasonic Attenuation Measurement for the Evaluation of Hepatic Steatosis: Preliminary Study and Validation in a Cohort of Patients with Chronic Liver Disease from Various Causes. Ultrasound Med Biol. 2010;36(11):1825–1835. doi: 10.1016/j.ultrasmedbio.2010.07.005. [DOI] [PubMed] [Google Scholar]
- 11.Ren X, Zhang L, Xia S, Chen Z, Zhou W, Ji R, et al. A New Visual Transient Elastography Technique for Grading Liver Fibrosis in Patients With Chronic Hepatitis B. Ultrasound Q. 2020;37(2):105–110. doi: 10.1097/RUQ.0000000000000509. [DOI] [PubMed] [Google Scholar]
- 12.Ren X, Xia S, Zhang L, Li R, Zhou W, Ji R, et al. Analysis of liver steatosis analysis and controlled attenuation parameter for grading liver steatosis in patients with chronic hepatitis B. Quant Imaging Med Surg. 2021;11(2):571–578. doi: 10.21037/qims-19-1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dehghan M, Merchant ATJNJ. Is bioelectrical impedance accurate for use in large epidemiological studies? Nutr J. 2008;7(1):26–26. doi: 10.1186/1475-2891-7-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Böhm A, Heitmann BL. The use of bioelectrical impedance analysis for body composition in epidemiological studies. Eur J Clin Nutr. 2013;67:S79–S85. doi: 10.1038/ejcn.2012.168. [DOI] [PubMed] [Google Scholar]
- 15.Di Vincenzo O, Marra M, Scalfi L. Bioelectrical impedance phase angle in sport: a systematic review. J Int Soc Sports Nutr. 2019;16(1):49. doi: 10.1186/s12970-019-0319-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Iasonos A, Schrag D, Raj GV, Panageas KS. How to build and interpret a nomogram for cancer prognosis. J Clin Oncol. 2008;26(8):1364–1370. doi: 10.1200/JCO.2007.12.9791. [DOI] [PubMed] [Google Scholar]
- 17.Altmann A, Tolosi L, Sander O, Lengauer T. Permutation importance: a corrected feature importance measure. Bioinformatics. 2010;26(10):1340–1347. doi: 10.1093/bioinformatics/btq134. [DOI] [PubMed] [Google Scholar]
- 18.Harrell FE, Jr, Lee KL, Mark DB. Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med. 1996;15(4):361–387. doi: 10.1002/(SICI)1097-0258(19960229)15:4<361::AID-SIM168>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 19.Alba AC, Agoritsas T, Walsh M, Hanna S, Iorio A, Devereaux PJ, et al. Discrimination and Calibration of Clinical Prediction Models: Users’ Guides to the Medical Literature. JAMA. 2017;318(14):1377–1384. doi: 10.1001/jama.2017.12126. [DOI] [PubMed] [Google Scholar]
- 20.Esmeijer K, Schoe A, Ruhaak LR, Hoogeveen EK, Soonawala D, Romijn F, et al. The predictive value of TIMP-2 and IGFBP7 for kidney failure and 30-day mortality after elective cardiac surgery. Sci Rep. 2021;11(1):1071. doi: 10.1038/s41598-020-80196-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fitzgerald M, Saville BR, Lewis RJ. Decision curve analysis. JAMA. 2015;313(4):409–410. doi: 10.1001/jama.2015.37. [DOI] [PubMed] [Google Scholar]
- 22.Eslam M, Sanyal AJ, George J, International Consensus P. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology. 2020;158(7):1999–2014.e1. doi: 10.1053/j.gastro.2019.11.312. [DOI] [PubMed] [Google Scholar]
- 23.Machado MV, Cortez-Pinto H. Non-invasive diagnosis of non-alcoholic fatty liver disease. A critical appraisal. J Hepatol. 2013;58(5):1007–1019. doi: 10.1016/j.jhep.2012.11.021. [DOI] [PubMed] [Google Scholar]
- 24.Li L, You W, Ren W. The ZJU index is a powerful index for identifying NAFLD in the general Chinese population. Acta Diabetol. 2017;54(10):905–911. doi: 10.1007/s00592-017-1024-8. [DOI] [PubMed] [Google Scholar]
- 25.Bedogni G, Bellentani S, Miglioli L, Masutti F, Passalacqua M, Castiglione A, et al. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006;6:33. doi: 10.1186/1471-230X-6-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lee JH, Kim D, Kim HJ, Lee CH, Yang JI, Kim W, et al. Hepatic steatosis index: a simple screening tool reflecting nonalcoholic fatty liver disease. Dig Liver Dis. 2010;42(7):503–508. doi: 10.1016/j.dld.2009.08.002. [DOI] [PubMed] [Google Scholar]
- 27.Vongsuvanh R, George J, McLeod D, van der Poorten D. Visceral adiposity index is not a predictor of liver histology in patients with non-alcoholic fatty liver disease. J Hepatol. 2012;57(2):392–398. doi: 10.1016/j.jhep.2012.03.013. [DOI] [PubMed] [Google Scholar]
- 28.Amato MC, Giordano C, Galia M, Criscimanna A, Vitabile S, Midiri M, et al. Visceral Adiposity Index: a reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care. 2010;33(4):920–922. doi: 10.2337/dc09-1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Petta S, Amato M, Cabibi D, Cammà C, Di Marco V, Giordano C, et al. Visceral adiposity index is associated with histological findings and high viral load in patients with chronic hepatitis C due to genotype 1. Hepatology. 2010;52(5):1543–1552. doi: 10.1002/hep.23859. [DOI] [PubMed] [Google Scholar]
- 30.Fu CP, Ali H, Rachakonda VP, Oczypok EA, DeLany JP, Kershaw EE. The ZJU index is a powerful surrogate marker for NAFLD in severely obese North American women. PLoS One. 2019;14(11):e0224942. doi: 10.1371/journal.pone.0224942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kang SH, Cho Y, Jeong SW, Kim SU, Lee JW, Korean NSG. From nonalcoholic fatty liver disease to metabolic-associated fatty liver disease: Big wave or ripple? Clin Mol Hepatol. 2021;27(2):257–269. doi: 10.3350/cmh.2021.0067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lin S, Huang J, Wang M, Kumar R, Liu Y, Liu S, et al. Comparison of MAFLD and NAFLD diagnostic criteria in real world. Liver Int. 2020;40(9):2082–2089. doi: 10.1111/liv.14548. [DOI] [PubMed] [Google Scholar]
- 33.Huang SC, Su HJ, Kao JH, Tseng TC, Yang HC, Su TH, et al. Clinical and Histologic Features of Patients with Biopsy-Proven Metabolic Dysfunction-Associated Fatty Liver Disease. Gut Liver. 2021;15(3):451–458. doi: 10.5009/gnl20218. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
BCM, body cell mass; BMC, bone mineral content; BMI, body mass index; BMR, basal metabolic rate. ECW, extracellular water; FFM, fat-free mass; ICW, intracellular water; PBF, percent body fat; SLM, soft lean mass; SMM, skeletal muscle mass; TBW, total body water; VFA, visceral fat area; WC, waist circumference.
(A) In the development and the (B) validation cohorts. Y-axis, actual fatty liver probability; X-axis, predicted fatty liver risk; black diagonal line, reference line (ideal nomogram).