Abstract
Objective
Follistatin-like protein 1 (FSTL1) is widely recognised as a secreted glycoprotein, but its role in modulating macrophage-related inflammation during liver fibrosis has not been documented. Herein, we aimed to characterise the roles of macrophage FSTL1 in the development of liver fibrosis.
Design
Expression analysis was conducted with human liver samples obtained from 33 patients with liver fibrosis and 18 individuals without fibrosis serving as controls. Myeloid-specific FSTL1-knockout (FSTL1M-KO) mice were constructed to explore the function and mechanism of macrophage FSTL1 in 3 murine models of liver fibrosis induced by carbon tetrachloride injection, bile duct ligation or a methionine-deficient and choline-deficient diet.
Results
FSTL1 expression was significantly elevated in macrophages from fibrotic livers of both humans and mice. Myeloid-specific FSTL1 deficiency effectively attenuated the progression of liver fibrosis. In FSTL1M-KO mice, the microenvironment that developed during liver fibrosis showed relatively less inflammation, as demonstrated by attenuated infiltration of monocytes/macrophages and neutrophils and decreased expression of proinflammatory factors. FSTL1M-KO macrophages exhibited suppressed proinflammatory M1 polarisation and nuclear factor kappa B pathway activation in vivo and in vitro. Furthermore, this study showed that, through its FK domain, FSTL1 bound directly to the pyruvate kinase M2 (PKM2). Interestingly, FSTL1 promoted PKM2 phosphorylation and nuclear translocation, reduced PKM2 ubiquitination to enhance PKM2-dependent glycolysis and increased M1 polarisation. Pharmacological activation of PKM2 (DASA-58) partially countered FSTL1-mediated glycolysis and inflammation.
Conclusion
Macrophage FSTL1 promotes the progression of liver fibrosis by inducing M1 polarisation and inflammation based on the intracellular PKM2 reprogramming function of macrophages.
Keywords: inflammation, hepatic fibrosis, macrophages, signaling
Significance of this study.
What is already known on this subject?
Macrophage has been identified as key regulators of the liver inflammation, which is critical for the progression or resolution of liver fibrosis.
Follistatin-like protein 1 (FSTL1) is widely regarded as a secreted glycoprotein with regulatory functions in many diseases.
Secreted/Circulatory FSTL1 has been proposed as a promoting protein in inflammation or fibrosis-related diseases in a secretion-dependent manner.
What are the new findings?
FSTL1 expression in macrophages is increased and is positively correlated with histological stages of liver fibrosis in humans and mice.
Myeloid FSTL1 deficiency suppresses M1 polarisation, reduces proinflammatory mediator expression and macrophage/neutrophil recruitment and alleviates liver fibrosis.
FSTL1 directly binds to pyruvate kinase M2 (PKM2), inhibits ubiquitin-mediated degradation, enhances the stability of cytoplasmic PKM2 and promotes PKM2 phosphorylation and nuclear translocation in macrophages.
PKM2 is critical for FSTL1-mediated M1 polarisation, inflammation and glycolysis in macrophages.
How might it impact on clinical practice in the foreseeable future?
Our findings provide a rationale for novel therapeutic strategies to ameliorate macrophage-mediated liver inflammation and fibrosis.
Introduction
Liver fibrosis is the most common consequence of chronic liver diseases, and it causes >1 million deaths worldwide every year.1 Advanced liver fibrosis, especially its end-stage form, cirrhosis, is generally regarded as an incurable disease, and liver transplantation is the sole and definitive treatment option.2 Various aetiologies, such as viral hepatitis, alcohol abuse, cholestasis or, as recently discovered, non-alcoholic steatohepatitis (NASH), can cause chronic liver injury and subsequent liver fibrosis.3 However, this mechanism of precise regulation is problematic and uncontrollable, especially when liver injury persists. Currently, liver fibrosis is widely recognised as a dynamic cascade fuelled by persistent and excessive activation of hepatic inflammation that develops in response to repetitive or chronic hepatocyte injury irrespective of aetiology.4 5 Uncontrolled chronic inflammation is a non-negligible driving force transforming self-limited tissue repair processes into a vicious cycle that boosts the progression of liver fibrosis.6
Immune cells, especially liver-residing Kupffer cells and recruited macrophages, have been identified as key regulators of the liver inflammation that are key to the progression or resolution of liver fibrosis.7 Damaged hepatocytes release danger-associated molecular patterns (DAMPs) to activate Kupffer cells and infiltrated macrophages. Activated macrophages release various cytokines, causing direct damage to liver parenchymal cells, enhancing inflammatory cell infiltration and activating hepatic stellate cells (HSCs).8 9 Hepatic macrophages have reportedly promoted liver fibrosis by increasing the survival of activated HSCs in an nuclear factor kappa B (NF-κB)-dependent manner.10 In addition, macrophage-derived transforming growth factor (TGF)-β has been recognised as a key molecule initiating HSC activation.11 Furthermore, several studies have indicated that toll-like receptor (TLR)-4 and TLR-9 signalling pathways mediate crosstalk between inflammatory and fibrotic factors.12 13 Thus, a deeper understanding of the underlying mechanisms involved in macrophage-orchestrated chronic liver inflammation can provide valuable information that may help to halt and effectively reverse ongoing liver fibrosis.
Follistatin-like protein 1 (FSTL1) is widely known as a secreted glycoprotein with regulatory functions in many diseases and is indispensable during embryogenesis.14 FSTL1-deficient mice suffer from postnatal death as a result of respiratory failure, while hypomorphic mice develop spontaneous emphysema.15 16 In addition to its regulatory functions seen in embryonic development, the role of FSTL1 in inflammation has been widely studied in the past decade.17 18 However, whether FSTL1 plays a proinflammatory role is still debated.17–21 FSTL1 is widely regarded as a fibrosis-promoting protein both outside and inside the liver, but previous studies have mainly focused on the profibrotic functions of FSTL1 in mesenchymal cells such as fibroblasts, not in inflammatory cells such as macrophages.22–24 Pyruvate kinase M2 (PKM2) is directly involved in the metabolic reprogramming (aerobic glycolysis) associated with cancer and the inflammatory response.25 26 Importantly, PKM2 has been shown to control macrophage metabolic remodelling in inflammation. PKM2 interacts with hypoxia-inducible factor 1α (HIF-1α) and activates the HIF-1α-dependent transcription of enzymes necessary for aerobic glycolysis in macrophages.26
Herein, we aimed to characterise the roles and mechanisms of macrophage FSTL1 during liver fibrosis. First, macrophage FSTL1 expression was assessed in patients and mice with liver fibrosis/cirrhosis. Second, we created a myeloid-specific FSTL1-knockout (FSTL1M-KO) mouse to study the function of macrophage FSTL1 in different mouse models of liver fibrosis. Third, the functional effects and underlying mechanisms of FSTL1 on macrophage-related inflammation were thoroughly analysed in vivo and in vitro.
Materials and methods
Human liver samples
Liver samples were obtained from patients who underwent hepatectomy or percutaneous liver biopsy. Liver samples were collected from 18 patients with benign liver diseases (postoperative pathological diagnosis showed hemangioma with no liver fibrosis or obvious steatosis; 18 liquid nitrogen-frozen samples and 6 paraffin-embedded samples) and 33 patients with pathologically diagnosed liver fibrosis (33 liquid nitrogen-frozen samples and 16 paraffin-embedded slides). The control individuals that were included had no history of diabetes, alcohol abuse or viral hepatitis. Control samples were taken from normal liver tissues at the edge of resected hemangioma. Pathological diagnosis of liver fibrosis was determined by senior pathologists based on H&E and Masson’s staining. Samples from patients with liver fibrosis were graded according to the fibrosis scoring systems by two senior pathologists (online supplemental tables 1–4).
gutjnl-2021-325150supp001.pdf (126.1KB, pdf)
Animal experiments
All mice were housed in an SPF level animal facility with controlled temperature and humidity on a 12–12 hour light-dark cycle with food and water supplied ad libitum. For the carbon tetrachloride (CCl4) injection-induced liver fibrosis, male mice aged 6–8 weeks were randomly assigned to receive intraperitoneal injection of olive oil or CCl4 (10% v/v dissolved in olive oil, 2 mL/kg) twice per week for 8 weeks. For bile duct ligation (BDL)-induced liver fibrosis, male mice aged 6–8 weeks were subjected to surgical intervention. Briefly, mice were anaesthetised by isoflurane inhalation. After skin disinfection, a midline abdominal incision was made. The common bile ducts were exposed and ligated using 5–0 non-absorbable sutures. Samples were collected 4 weeks after surgery. To induce liver fibrosis by methionine-deficient and choline-deficient (MCD) diet, male mice aged 8–10 weeks were fed a normal chow (NC) or MCD diet (A02082002BR, Research Diets) for an indicated duration of up to 8 weeks.
Statistical analysis
Unpaired Student’s t-test or Mann-Whitney U test were performed in group comparisons by using GraphPad Prism (V.8.0, GraphPad Software). P values <0.05 were considered to be statistically significant.
For detailed methods and materials, please refer to online supplemental materials.
gutjnl-2021-325150supp002.pdf (151KB, pdf)
Results
FSTL1 expression is increased in macrophages in fibrotic liver tissues
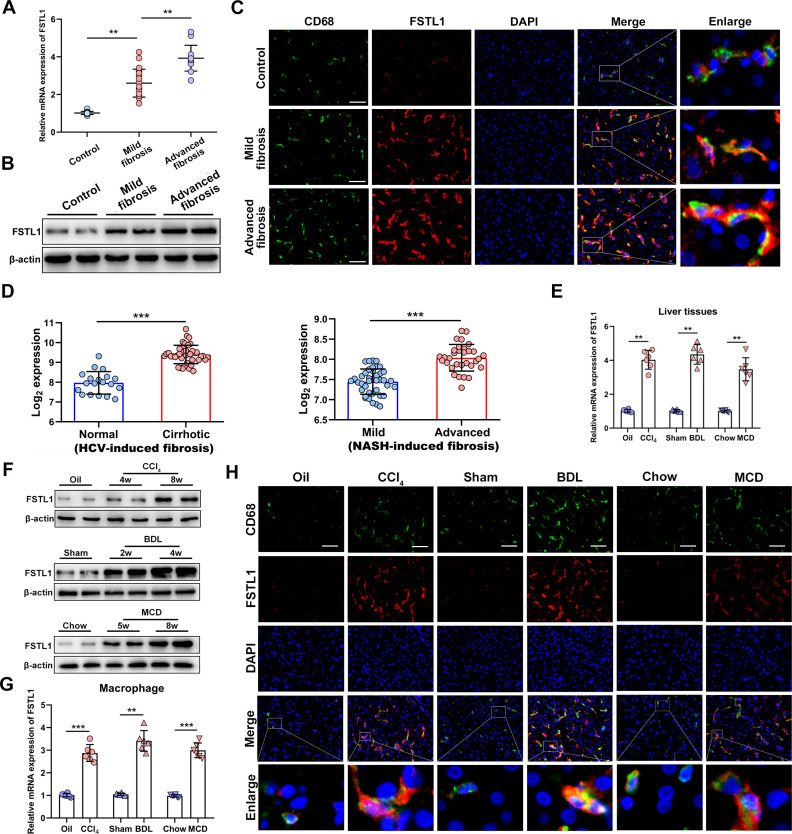
First, we aimed to characterise FSTL1 expression among fibrotic liver samples. We collected 51 human liver samples including normal and fibrosis liver tissues (online supplemental tables 1–4). As indicated by H&E staining, Masson’s staining and messenger RNA (mRNA) expression of tissue inhibitor of metalloproteinase 1 (TIMP-1) and collagen 1A1 (COL1A1), our liver samples showed different histological stages of fibrosis with fair between-group comparability (online supplemental figure 1A, B). Interestingly, the mRNA and protein expression of FSTL1 was markedly elevated in fibrotic livers compared with normal control livers (figure 1A, B). Next, we determined whether FSTL1 expression was mainly elevated in liver macrophages. Dual immunofluorescence staining showed that FSTL1 was primarily expressed on macrophages in human fibrotic livers (figure 1C and online supplemental figure 1C). Then, we used publicly available omics data on patient samples to validate these results. Virus-mediated and NASH-induced liver fibrosis are common pathological processes in liver fibrosis worldwide. By reanalysing a published dataset, we found that FSTL1 expression was significantly elevated in HCV-induced or NASH-induced liver fibrosis (figure 1D).27 28
Figure 1.
Follistatin-like protein 1 (FSTL1) expression is increased in macrophage of liver fibrosis tissues. Human dada (A–D) and mice data (E–H): (A) FSTL1 messenger RNA (mRNA) expression and (B) protein expression in human liver samples; (C) dual-immunofluorescence staining of FSTL1 and CD68 in liver samples; (D) liver FSTL1 expression among patients with HCV or non-alcoholic steatohepatitis (NASH)-induced liver fibrosis as determined by microarray data; (E) FSTL1 mRNA expression and (F) protein expression in murine livers; (G) FSTL1 mRNA expression in isolated mice liver macrophages; (H) dual-immunofluorescence staining of FSTL1 and CD68 in murine livers. Human samples: control (n=18), mild fibrosis (n=20), advanced fibrosis (n=13); mice samples (n=6–8 per group). Data were presented as mean±SEM; scale bars, 50 µm; **p<0.01, ***p<0.001. BDL, bile duct ligation; CCl4, carbon tetrachloride; MCD, methionine-deficient and choline-deficient diet.
gutjnl-2021-325150supp003.pdf (1.9MB, pdf)
To confirm these findings, we explored FSTL1 expression in three widely recognised animal models of liver fibrosis, namely, CCl4 injection-induced, BDL-induced and the MCD diet-induced fibrosis. FSTL1 expression was significantly increased in liver tissues obtained from all models (figure 1E, F). These results were validated using three publicly available omics datasets (online supplemental figure 1D).29–31 Then, we isolated liver macrophages from fibrotic livers of mice. Consistently, FSTL1 mRNA expression levels were significantly increased in macrophages isolated from fibrotic livers (figure 1G). Dual-immunofluorescence staining furthermore showed FSTL1+CD68+ macrophages were significantly more in fibrotic livers than control livers (figure 1H). In addition, we revealed FSTL1 was higher expression on macrophages, lower expression on HSCs and non-expression on hepatocytes in liver fibrosis tissues (online supplemental figure 2A, B). Taken together, these findings demonstrate that FSTL1 expression is significantly increased in liver tissues, especially in macrophages of fibrotic livers.
Myeloid FSTL1 deficiency alleviates liver fibrosis
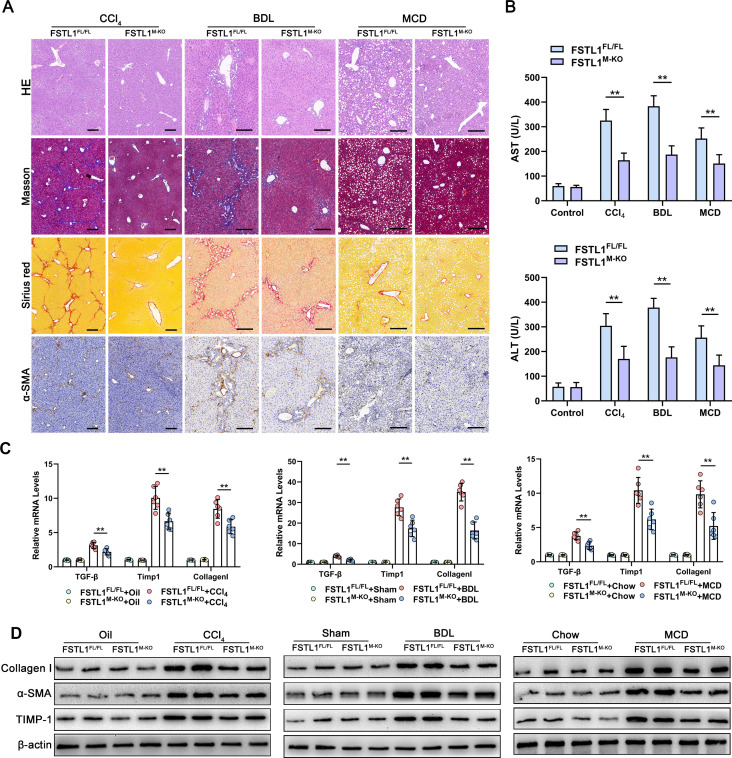
To test the function of FSTL1+ macrophages in liver fibrosis, we used the Cre-LoxP system to create myeloid-specific FSTL1-deficient (FSTL1M-KO) mice. Then, liver fibrosis was induced in FSTL1FL/FL and FSTL1M-KO mice with CCl4 injection, BDL or a MCD diet. Figure 2A shows that liver fibrosis was markedly lower in the FSTL1M-KO mice than in the FSTL1FL/FL mice, as revealed by Masson’s, Sirius Red and alfa-smooth muscle actin (α-SMA) staining. In addition, lower serum alanine aminotransferase and aspartate aminotransferase levels postchallenge were detected in FSTL1M-KO mice (figure 2B). Moreover, expressions of TGF-β, TIMP-1 and collagen I were markedly lower in liver tissues from the FSTLM-KO mice than that in their littermate controls (figure 2C). Western blot (WB) analysis further showed FSTL1 deficiency reduced the expression of collagen I, α-SMA and TIMP-1 in fibrotic liver tissues compared with that in the control tissues (figure 2D). Our results suggest that myeloid FSTL1 deficiency attenuates hepatic fibrosis in mice.
Figure 2.
Myeloid follistatin-like protein 1 (FSTL1) deficiency alleviates mice liver fibrosis. (A) The degree of fibrosis was evaluated by H&E, Masson’s, Sirius Red and alfa-smooth muscle actin (α-SMA) immunobiological staining; (B) serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) was measured. (C) Messenger RNA (mRNA) expression of transforming growth factor (TGF)-β, tissue inhibitor of metalloproteinase 1 (TIMP-1) and collagen I was quantified by quantitative reverse transcription-PCR and (D) protein expression of collagen I, α-SMA and TIMP-1 was analysed by western blot (WB) analysis in liver samples. Data were presented as mean±SEM; n=6–8 per group; scale bars, 50 µm; **p<0.01. BDL, bile duct ligation; CCl4, carbon tetrachloride; MCD, methionine-deficient and choline-deficient diet.
Myeloid FSTL1 deficiency attenuates inflammation and macrophage/neutrophil recruitment in fibrotic livers
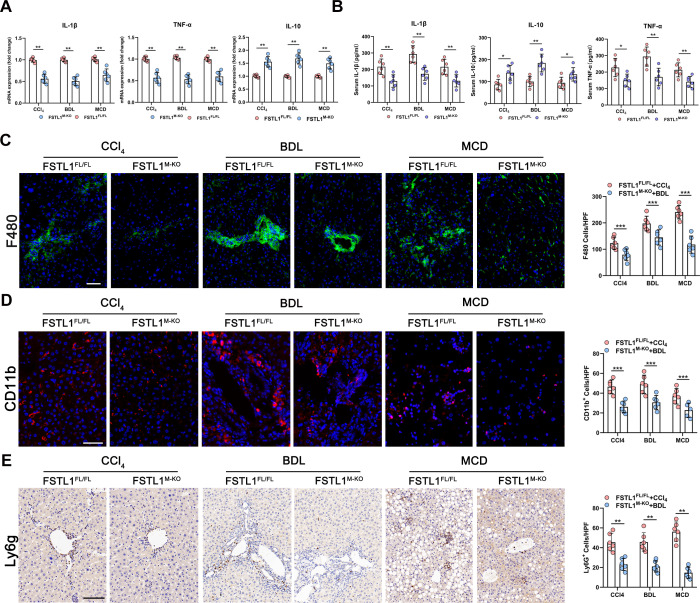
Next, we examined the impact of FSTL1+ macrophages on local inflammation in fibrotic liver tissues. Figure 3A shows lower expression of tumour necrosis factor (TNF)-α and interleukin (IL)-1β, and higher expression of IL-10 in fibrotic livers obtained from FSTL1M-KO mice than controls. Consistently, serum levels of TNF-α and IL-1β were lower, while IL-10 was higher in FSTL1M-KO mice than their littermates (figure 3B). Furthermore, we evaluated the accumulation of hepatic inflammatory cells by F4/80 and CD11b (macrophage markers) and Ly6G (a neutrophil marker) staining in fibrotic liver tissues. Compared with the FSTL1FL/FL mouse, myeloid-specific FSTL1 deficiency markedly decreased macrophage and neutrophil infiltration in fibrotic liver tissues (figure 3C–E).
Figure 3.
Myeloid follistatin-like protein 1 (FSTL1) deficiency attenuates inflammation in mice fibrotic livers. (A) Messenger RNA (mRNA) expression of tumour necrosis factor (TNF)-α, interleukin (IL)-1β and IL-10 was quantified in liver tissues; (B) serum TNF-α, IL-1β and IL-10 were measured by ELISA. (C) F4/80, (D) CD11b and (E) Ly6G were detected by immunofluorescence, and quantified as numbers of positive cells per high power field (HPF). Data were presented as mean±SEM; n=6–8 per group; scale bars, 50 µm; *p<0.05, **p<0.01, ***p<0.001. BDL, bile duct ligation; CCl4, carbon tetrachloride; MCD, methionine-deficient and choline-deficient diet.
Myeloid FSTL1 deficiency suppresses M1 polarisation and NF-κB pathway activation in fibrotic livers
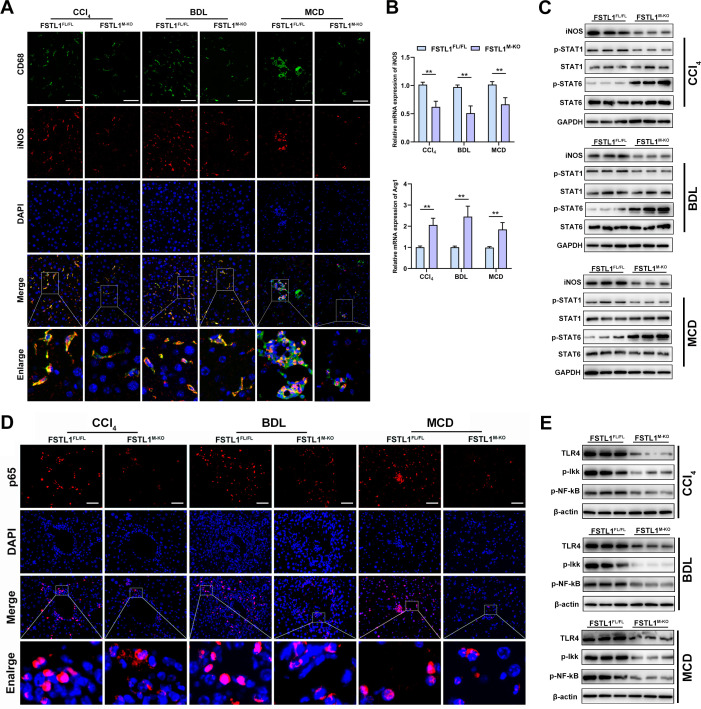
As mentioned above, myeloid-specific FSTL1 deficiency alleviated liver inflammation, suggesting that FSTL1 may promote M1 polarisation. First, we analysed the quantification of M1 macrophages in liver slices, and the results showed that fewer iNOS+CD68+ macrophages were observed in the FSTL1M-KO mice than in the FSTL1FL/FL mice (figure 4A). In addition, in the FSTL1M-KO mice, the mRNA expression of inducible nitric oxide synthase (iNOS) was downregulated but Arg1 was upregulated in the fibrotic livers (figure 4B). WB analysis showed that myeloid FSTL1-deficient mice exhibited decreased expression of iNOS and p-STAT1 and increased expression of p-STAT6 in fibrotic livers compared with the control livers (figure 4C). The TLR-4/NF-κB pathway is a well-characterised regulatory pathway intensively involved in macrophage-orchestrated acute/chronic liver inflammation.10 Herein, immunostaining and WB analysis of fibrotic livers also revealed that FSTL1M-KO mice exhibited depressed TLR-4/NF-κB pathway activation (figure 4D, E).
Figure 4.
Myeloid follistatin-like protein 1 (FSTL1) deficiency suppresses M1 polarisation and nuclear factor kappa B (NF-κB) pathway activation in mice fibrotic livers. (A) Dual-immunofluorescence staining of inducible nitric oxide synthase (iNOS) and CD68 in liver tissues; (B) messenger RNA (mRNA) expression of iNOS and Arg1 was quantified in liver tissues; (C) protein expression of iNOS, p-STAT1, p-STAT6 and GAPDH were analysed in liver samples; (D) immunofluorescence staining of p65 and diamidino-2-phenylindole (DAPI) in liver tissues; (E) protein expression of toll-like receptor (TLR)-4, phospho-IkappaB kinases (p-Ikk), p-NF-κBp65 and GAPDH were analysed in liver samples. Data were presented as mean±SEM; n=6–8 per group; scale bars, 50 µm; **p<0.01. BDL, bile duct ligation; CCl4, carbon tetrachloride; MCD, methionine-deficient and choline-deficient diet.
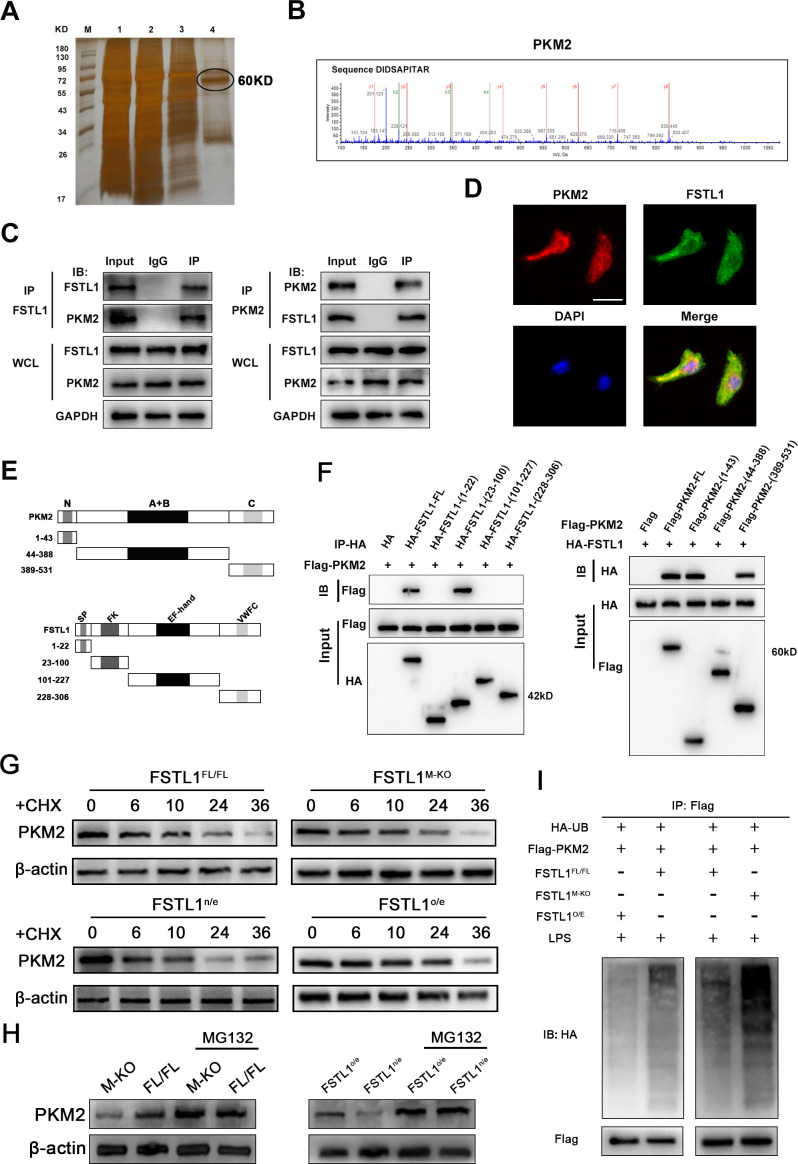
FSTL1 directly binds with PKM2 and enhances the stability of PKM2 in macrophages
To further investigate the mechanism underlying the regulatory role of macrophage FSTL1 during liver fibrosis, immunoprecipitation (IP) assays and LC-MS/MS screening were performed to explore FSTL1 binding to potential proteins in macrophages. We were particularly interested in determining whether PKM2 directly interacts with FSTL1 in macrophages (figure 5A, B). Therefore, cell lysates were immunoprecipitated using anti-FSTL1 or anti-PKM2 antibodies, and the Co-IP complex was then immunoblotted. The results showed that FSTL1 bound to PKM2 in macrophages (figure 5C). In addition, confocal immunofluorescence analysis revealed that FSTL1 colocalised with PKM2 in the cytoplasm and nucleus (figure 5D). Next, we analysed the domains critical for the FSTL1-PKM2 interaction. According to the previously reported structural features of FSTL1 and PKM2, plasmids encoding four truncated fragments of FSTL1 each with ahemagglutinin (HA) tag and three truncated fragments of PKM2 each with a FLAG tag were constructed and transfected into 293 T cells (figure 5E).32 33 Cell lysates were subjected to IP with an anti-HA antibody and immunoblotting with an anti-FLAG antibody. Our data showed that the FK domain of FSTL1 was critical for the interaction with the N-terminus or C-terminus of PKM2 (figure 5F). These results reveal that FSTL1 directly interacts with PKM2 through the FK domain of FSTL1 and the N-terminus or C-terminus of PKM2. Upregulation of the PKM2 protein in tissues was previously achieved by promoting transcription, inhibiting proteasome degradation or enhancing mRNA translation.34 However, in this study, there were no significant differences in the PKM2 mRNA levels in the FSTL1-knockout or FSTL1-overexpressing cells (online supplemental figure 3). Thus, we speculated that FSTL1 might regulate the protein stability of PKM2. The results from a cycloheximide chase assay demonstrated that PKM2 in FSTL1-downregulated cells had a shorter half-life, while the half-life of PKM2 was much longer in the FSTL1-overexpressing cells than in the control cells (figure 5G). These results suggest that FSTL1 might regulate PKM2 stability by inhibiting proteasome degradation. Notably, we observed that the reduction in protein level of PKM2 accompanied by FSTL1 downregulation was recovered by the addition of MG-132 to the FSTL1M-KO and FSTL1o/e bone marrow-derived macrophages (BMDMs) cultures (figure 5H). Then, using an in vitro ubiquitination assay, we tested whether FSTL1 was involved in ubiquitin-mediated PKM2 degradation. Downregulation of FSTL1 increased the ubiquitination level of PKM2 protein in the FSTL1M-KO BMDMs. The ubiquitin-mediated degradation effect of PKM2 was significantly abrogated in FSTL1-overexpressing BMDMs (figure 5I). PKM2 ubiquitination is a critical step in PKM2 phosphorylation and nuclear translocation.
Figure 5.
Follistatin-like protein 1 (FSTL1) directly binds with pyruvate kinase M2 (PKM2) and enhances the stability of PKM2 in macrophages. (A) The Co-immunoprecipitation (Co-IP) complex was subjected to silver-staining (line 4 represented the eluted protein); (B) the peptide sequences of the PKM2 protein as detected in the Co-IP complex by mass spectrometry; (C) western blot (WB) analysis of Co-IP complex confirmed that FSTL1 interacted with PKM2 in bone marrow-derived macrophages (BMDMs); (D) the immunofluorescence showed that FSTL1 colocalised with PKM2 and were expressed in both the cytoplasm and nucleus of BMDMs; (E) the structural compositions of FSTL1 and PKM2; (F) WB analysis of relationship between truncated FSTL1 and full-length PKM2 or truncated PKM2 and full-length FSTL1; (G) BMDMs extracted from FSTL1FL/FL and FSTL1M-KO mice and BMDMs extracted from FSTL1FL/FL transfected with LV-FSTL1 or lentivirus-negative control (LV-NC) were treated with cycloheximide (100 ng/mL) for the indicated periods of time; (H) BMDMs extracted from FSTL1FL/FL and FSTL1M-KO mice and BMDMs extracted from FSTL1FL/FL transfected with LV-FSTL1 or LV-NC were treated with or without MG132 (50 µM) for 6 hours. (I) In vitro ubiquitination assay of FSTL1FL/FL, FSTL1M-KO and FSTL1o/e BMDMs. All cells were co-transfected with FLAG-PKM2 and HA-UB plasmid. Cell lysates were immunoprecipitated with anti-FLAG antibody followed by immunoblotting analysis with anti-HA or anti-FLAG antibody. Scale bars, 50 µm.
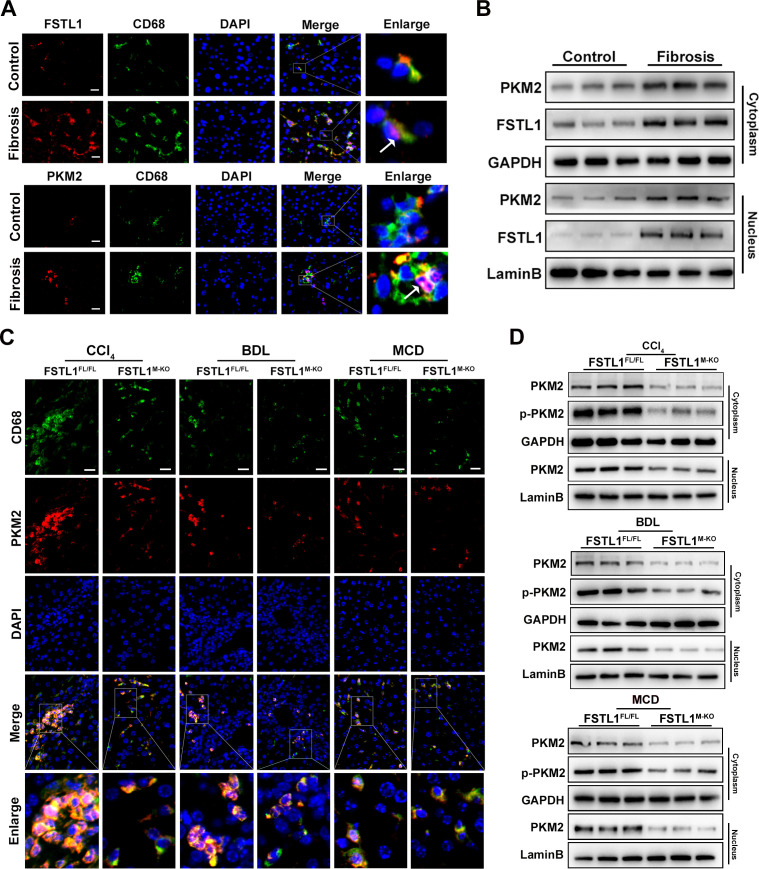
FSTL1 promotes PKM2 phosphorylation and nuclear translocation in macrophages of fibrotic liver tissues
To explore the role of PKM2 in FSTL1-mediated macrophage proinflammatory phenotype switching, we first examined whether PKM2 expression was altered in macrophages of fibrotic livers. Immunofluorescence staining showed that fibrotic livers had more FSTL1+ macrophages (figure 6A, upper panel) and PKM2+ macrophages (figure 6A, down panel) than control livers. Then, we analysed the protein expression of PKM2 and FSTL1 in the cytoplasm and nucleus of human liver samples, which showed the expression of PKM2 and FSTL1 was markedly increased in both the cytoplasm and nucleus of the fibrotic liver tissues (figure 6B and online supplemental figure 4). Immunofluorescence staining also displayed nuclear translocation of PKM2 and FSTL1 was significantly increased in the macrophages from patient livers compared with control livers (figure 6A, purple area). The results for the human fibrotic livers were similar to those for the mouse fibrotic livers. In contrast, disruption of myeloid FSTL1 clearly reduced PKM2+ macrophages and hindered the nuclear import of PKM2 in liver macrophages compared with the FSTL1FL/FL mice (figure 6C). PKM2 phosphorylation was previously shown to be an indicator of monomer or dimer configuration, which can be translocated into the nucleus.33 35 Therefore, we analysed the protein expression of total PKM2 and p-PKM2 in the cytoplasm and total PKM2 in the nucleus of isolated liver macrophages. Figure 6D showed that, compared with the FSTL1FL/FL controls, FSTL1 deficiency reduced the expression of total PKM2 and p-PKM2 in the cytoplasm and then decreased total PKM2 in the nucleus. Collectively, these results indicate that macrophage FSTL1 promotes the nuclear translocation of PKM2.
Figure 6.
Follistatin-like protein 1 (FSTL1) promotes pyruvate kinase M2 (PKM2) phosphorylation and nuclear translocation in macrophage of fibrotic liver tissues. Human data (A–B) and mice data (C–D): (A) immunofluorescence staining of FSTL1 (upper panels) or PKM2 (lower panels), CD68 and DAPI in human liver tissues; (B) expressions of PKM2 and FSTL1 were analysed by western blot (WB) analysis in cytoplasm and nucleus of human livers; (C) immunofluorescence staining of PKM2, CD68 and DAPI in mice fibrotic livers; (D) expression of PKM2 and p-PKM2 were determined by WB analysis in cytoplasm and nucleus of fibrotic livers. Scale bars, 20 μm. BDL, bile duct ligation; CCl4, carbon tetrachloride; MCD, methionine-deficient and choline-deficient diet.
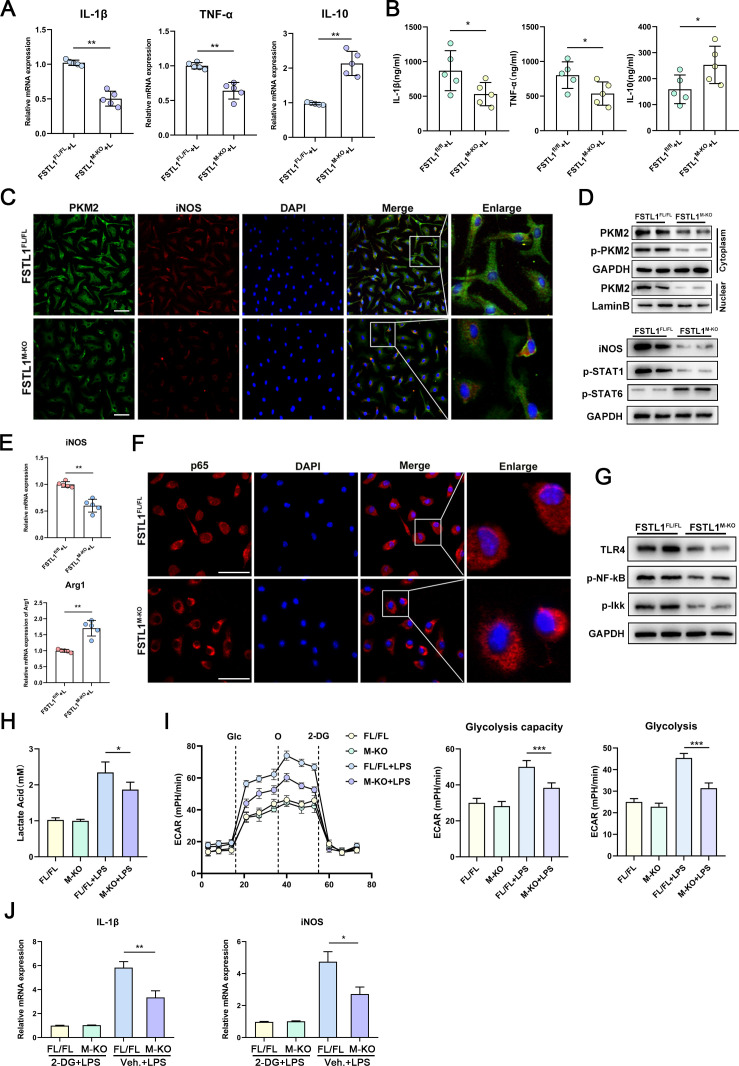
FSTL1 deficiency attenuates the inflammatory responses, M1 polarisation and glycolysis in macrophages
To further analyse how FSTL1 regulates macrophage function in vitro, BMDMs were differentiated from FSTL1FL/FL or FSTL1M-KO mouse cells and subjected to lipopolysaccharide (LPS) stimulation. FSTL1M-KO macrophages showed decreased expression of TNF-α and IL-1β and increased expression of IL-10 (figure 7A, B). Consistent with the in vivo experiments, FSTL1 deletion attenuated M1 polarisation and decreased TLR-4/NF-κB pathway activation in the BMDMs following LPS treatment (figure 7C–G). Next, we explored whether FSTL1 deficiency reduces PKM2 expression and nuclear translocation in vitro. PKM2 expression and its nuclear translocation were attenuated in the FSTL1M-KO BMDMs (figure 7C, D). PKM2 is crucial for the Warburg effect.36 To explore whether FSTL1 also regulates glycolysis, we examined the extracellular acidification rate (ECAR) and relative lactate release level in these macrophages. We found that ECAR and lactic acid were markedly decreased after LPS stimulation in the FSTL1M-KO BMDMs compared with FSTL1FL/FL BMDMs (figure 7H, I). We used the glycolysis inhibitor 2-deoxy-D-glucose (2-DG) to block the glucose supply and found that the decrease in the expression of IL-1β and iNOS in FSTL1M-KO BMDMs was inhibited (figure 7J). In addition, we cocultured HSCs with BMDMs in an LPS-induced inflammatory environment (online supplemental figure 5A). FSTL1-deficient BMDMs markedly reduced α-SMA expression and attenuated the activation of HSCs (online supplemental figure 5B, C).
Figure 7.
Follistatin-like protein 1 (FSTL1) deficiency attenuates inflammation and glycolysis in macrophages. Bone marrow-derived macrophages (BMDMs) treated with 100 ng/mL lipopolysaccharide (LPS) for 24 hours (A–G): (A–B) messenger RNA (mRNA) and protein expressions of tumour necrosis factor (TNF)-α, interleukin (IL)-1β and IL-10 were determined; (C) immunofluorescence staining of pyruvate kinase M2 (PKM2) and inducible nitric oxide synthase (iNOS); (D) protein expression of PKM2, p-PKM2, iNOS, p-STAT1 and p-STAT6; (E) mRNA expression of iNOS and Arg1 was measured; (F) immunofluorescence staining of p65 and DAPI; (G) protein expression of toll-like receptor (TLR)-4, p-nuclear factor kappa B (NF-κB) and p-Ikk; (H) lactate acid production was measured from BMDMs in response to LPS (100 ng/mL, 6 hours). (I) Glycolytic rate and compacities of BMDMs were measured by real-time recording of extracellular acidification rates (ECAR) after successively injection of glucose (Glc), oligomycin (O) and 2-DG. (J) FSTL1FL/FL and FSTL1M-KO BMDMs treated with LPS (100 ng/mL, 4 hours). These cells were washed twice and pretreated with 10 mM 2-DG for 1 hour, followed by treatment with or without LPS (100 ng/mL) for another 4 hours. mRNA expression of IL-1β and iNOS were determined. Data were presented as mean±SEM of at least three independent experiments; scale bars, 50 µm; *p<0.05, **p<0.01, ***p<0.001.
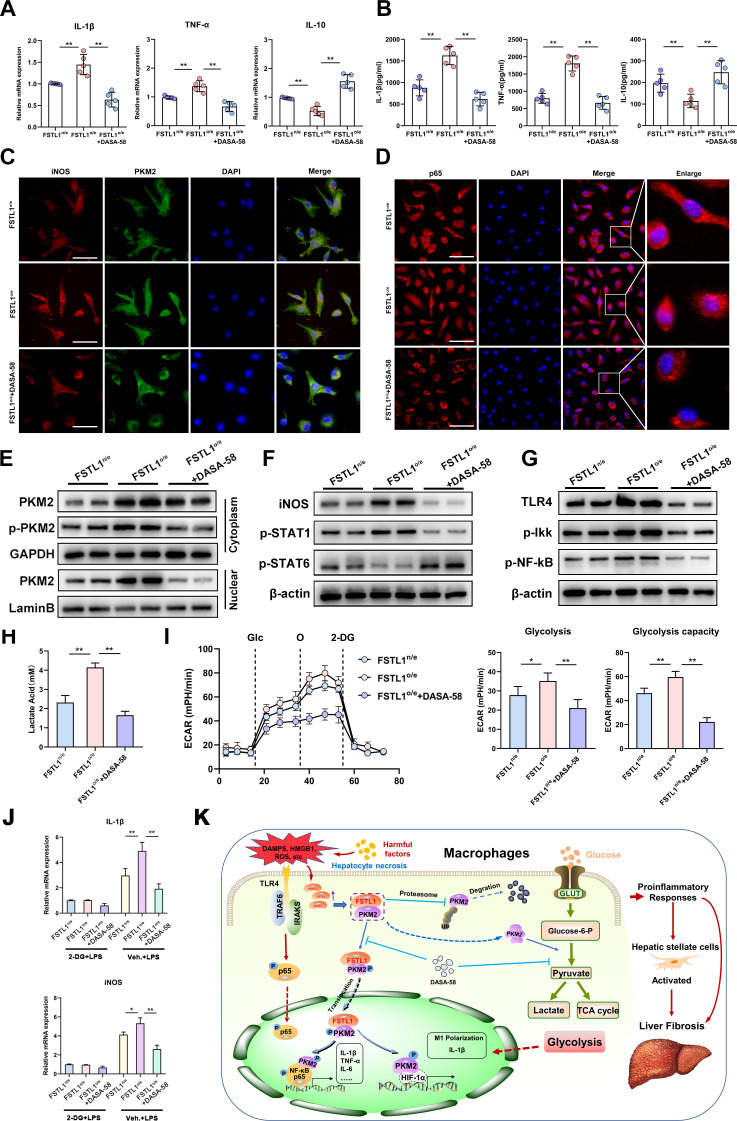
PKM2 activator inhibits FSTL1-mediated M1 polarisation and glycolysis in macrophages
To further determine the effect of PKM2 on FSTL1-induced macrophage polarisation and glycolysis, FSTL1-overexpressing (FSTL1o/e) BMDMs were treated with the small-molecule PKM2 activator DASA-58.37 38 DASA-58 alleviated FSTL1o/e-induced TNF-α and IL-1β while increasing IL-10 level (figure 8A, B). As indicated by immunofluorescence staining of PKM2, iNOS and p65, inhibition of PKM2 nuclear translocation reversed FSTL1-induced macrophage M1 polarisation and NF-κB activation (figure 8C, D). These results were confirmed by WB analysis of PKM2, p-PKM2, iNOS, p-STAT1, p-STAT6, TLR-4, p-Ikk and p-p65 expression in the BMDMs (figure 8E–G). To further verify the role of PKM2 in FSTL1-mediated glycolysis, we examined the ECAR and relative lactate release level. ECAR and lactic acid were significantly higher in the FSTL1o/e BMDMs than in the FSTL1n/e BMDMs, and DASA-58 effectively reversed the FSTL1o/e-induced increase in glycolysis (figure 8H, I). In addition, FSTL1o/e-related proinflammatory mediators (IL-1β and iNOS) were neutralised by 2-DG (figure 8J). These results validate that the nuclear and cytoplasmic functions of PKM2 are crucial to FSTL1-induced M1 polarisation, glycolysis and inflammation.
Figure 8.
Pyruvate kinase M2 (PKM2) activator inhibits follistatin-like protein 1 (FSTL1)-mediated inflammation and glycolysis in macrophages. Bone marrow-derived macrophages (BMDMs) with FSTL1 overexpression (FSTL1o/e) or normal expression (FSTL1n/e) and FSTL1o/e BMDMs treated with DASA-58 (50 μM, 1 hour) were treated by lipopolysaccharide (LPS) stimulation. BMDMs treated with 100 ng/mL LPS for 24 hours (A–G): (A–B) messenger RNA (mRNA) expression and protein expressions of tumour necrosis factor (TNF)-α, interleukin (IL)-1β and IL-10 was determined; (C) dual-immunofluorescence staining of PKM2 and inducible nitric oxide synthase (iNOS); (D) protein expression of PKM2, p-PKM2, iNOS, p-STAT1 and p-STAT6; (E) mRNA expression of iNOS and Arg1 was measured; (F) immunofluorescence staining of p65 and DAPI; (G) protein expression of toll-like receptor (TLR)-4, p-nuclear factor kappa B (NF-κB) and p-Ikk. (H) Lactate acid production was measured from BMDMs in response to LPS (100 ng/mL, 6 hours). (I) Glycolytic rate and compacities of BMDMs were measured by real-time recording of extracellular acidification rates (ECAR) after successively injection of glucose (Glc), oligomycin (O) and 2-DG. (J) FSTL1FL/FL and FSTL1M-KO BMDMs treated with LPS (100 ng/mL) for 4 hours. These cells were washed twice and pretreated with 10 mM 2-DG for 1 hour, followed by treatment with or without LPS (100 ng/mL) for another 4 hours. mRNA levels or IL-1β and iNOS were determined. (K) Schematic illustration how FSTL1 complexed with PKM2 to promote phosphorylation and nuclear import of PKM2 and enhance cytoplasmic PKM2 stability in reprograming macrophage function during liver fibrosis. Data were presented as mean±SEM of at least three independent experiments; scale bars, 50 µm; *p<0.05, **p<0.01.
Discussion
To the best of our knowledge, this study is the first to document that FSTL1-mediated PKM2 is crucial for modulating macrophage function during liver fibrosis. The principal findings are as follows: (i) FSTL1 expression in macrophages is increased and is positively correlated with histological stages of liver fibrosis; (ii) myeloid FSTL1 deficiency suppresses M1 polarisation, reduces proinflammatory mediator expression and macrophage/neutrophil recruitment and alleviates liver fibrosis; (iii) FSTL1 directly binds to PKM2, inhibits ubiquitin-mediated degradation, enhances the stability of PKM2 and promotes PKM2 phosphorylation and nuclear translocation in macrophages and (iv) PKM2 is critical for FSTL1-mediated M1 polarisation, inflammation and glycolysis in macrophages. Our findings highlight the importance of FSTL1 as a key regulator of PKM2 function and inflammation in macrophages during liver fibrosis (figure 8K).
It has been reported that uncontrolled inflammation is a major driving force that transforms self-limited tissue repair processes to a vicious cycle, boosting the progression of liver fibrosis.6 Macrophages are identified as pivotal regulators of liver inflammation that dominates the progression or resolution of liver fibrosis. Activated macrophages are generally divided into two categories, namely, M1 and M2.39 M1-polarised macrophages disrupt local homeostasis and facilitate fibrosis by interacting with hepatocytes and other fibrosis-related non-parenchymal cells, such as HSCs.8 39 40 FSTL1 is widely regarded as a proinflammatory gene possibly involved in macrophage polarisation.19–21 It is reported that FSTL1 expression is enhanced in liver tissue and serum of patients with advanced fibrosis and steatosis.41 42 Our data showed that FSTL1 expression is significantly increased in macrophages of human or mouse liver fibrotic tissues. In addition, we observed that myeloid FSTL1 deficiency inhibited macrophage TLR-4/NF-κB activation and M1 polarisation and limited hepatic inflammation and fibrosis.
FSTL1, as a secreted glycoproprotein, one widely acknowledged function of secreted FSTL1 is its modulatory role in inflammation, while the roles of FSTL1 in the cytoplasm and nucleus are not well understood. Circulatory FSTL1 levels are significantly elevated in patients with various autoimmune diseases.43 Secreted FSTL1 has been shown to play modulatory roles in metabolism and glycolysis. FSTL+/− mice showed impaired energy expenditure and developed glucose intolerance with decreased adaptive thermogenesis of brown adipose tissue.44 Furthermore, both acute and chronic infusion of FSTL1 impact oxygen consumption and energy substrate metabolism in cardiomyocytes through AMPK pathway activation.45 These findings highlighted the modulatory role of FSTL1 in the inflammatory response and cellular metabolism of macrophages in a secretion-dependent manner. However, whether intracellular FSTL1 functions as a regulator of inflammation and cellular metabolism remains unknown.
One striking finding was that, through its FK domain, intracellular FSTL1 directly binds with the N-terminus or C-terminus of PKM2, which is a master immune regulator and established metabolic enzyme. PKM2 primarily exists as an enzymatically inactive form of monomer or dimer, with phosphorylation being the key indicator of its state.33 Monomeric/dimeric PKM2 can translocate into the nucleus and stabilise the HIF-1α to promote the expression of glycolysis-related genes in tumours.35 Furthermore, PKM2 directly promotes proinflammatory reprogramming of macrophages; it is upregulated by LPS stimulation and promotes IL-1β production by binding to its promoter, which predominantly occurs during glycolysis in M1 macrophages.38 Herein, we found that the FSTL1 interacts with and stabilises the PKM2 protein by inhibiting PKM2 ubiquitin-mediated degradation. Our data further demonstrated that FSTL1 increases phosphorylation/nuclear PKM2, enhances PKM2-mediated M1 polarisation and promotes glycolysis in LPS-stimulated macrophages. It was reported that PKM2 can be directly bound to and phosphorylated by extracellular regulated kinase and translocated to the nucleus in a PIN1-dependent manner.46 However, how FSTL1 promotes the phosphorylation and nuclear translocation of PKM2 is still unknown.
IL-1β expression is closely linked to Warburg metabolism in LPS-activated macrophages.38 47 In addition, macrophage activation requires a metabolic switch from canonical oxidative phosphorylation towards glycolysis or the Warburg effect.48 PKM2 is a pyruvate kinase that is widely regarded as a rate-limiting enzyme during glycolysis that catalyses the transfer of phosphate groups from phosphoenolpyruvate to ADP and produces pyruvate in the cytoplasm. Interestingly, we found that FSTL1 increased the expression and stability of PKM2 in the cytoplasm of macrophages following LPS treatment. Logically, our data indicated that FSTL1 deficiency reduced glycolysis after LPS stimulation. DASA-58, a highly specific small-molecule PKM2 activator, leads to a decrease in glycolytic and pentose phosphate pathway intermediates by activating the enzyme.36 In addition, DASA-58 dramatically reverts this glycolytic phenotype in macrophages.38 We found DASA-58 effectively restored FSTL1o/e-mediated glycolysis, M1 polarisation and TLR-4/NF-κB pathway activation in LPS-stimulated macrophages. Thus, PKM2-mediated glycolysis is also critical for FSTL1-related M1 polarisation and inflammation. More importantly, the current study shows meticulously delineated FSTL1 regulation of glycolysis and glycolysis-dependent immune responses in macrophages, providing important insights into Warburg-like metabolic changes in macrophages.
In conclusion, we reveal the functional roles of macrophage FSTL1 in liver fibrosis. FSTL1 promotes M1 polarisation, glycolysis and the inflammatory response by binding to PKM2, promoting PKM2 phosphorylation and inhibiting PKM2 ubiquitination in macrophages. By identifying the molecular pathways by which FSTL1 mediates liver inflammation, our findings provide a rationale for novel therapeutic approaches to ameliorate macrophage-mediated liver inflammation and fibrosis.
Footnotes
Contributors: JR, HW and MN contributed equally. The study was designed by JR, FC and LL. The manuscript was written by JR, HW, FC and LL. The experiments and data analysis were performed by JR, HW, MN, ZW, ZW (5th author), SW, ML, PW, JQ, LZ, CW, HS, XW, FC and LL. JR, FC and LL supported and supervised the project. LL is the guarantor. All authors were involved in the critical revision of the manuscript.
Funding: The project was supported by the National Natural Science Foundation of China (81871259, 81971495, and 82070675), the Foundation of Jiangsu Collaborative Innovation Center of Biomedical Functional Materials, the Priority Academic Program Development of Jiangsu Higher Education Institutions, The Six talent peaks project in Jiangsu Province (2017-WSW-019) and the CAMS Innovation Fund for Medical Sciences (2019-I2M-5-035).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available in a public, open access repository. A generic lab email address or an individual’s ORCID identifier.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
The study design and sample collection protocols were approved by the Ethical Board of First Affiliated Hospital of Nanjing Medical University (Serial number: 2018-SRFA-197). All animal study protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of Nanjing Medical University (AP#:GPTAP002). All patients provided informed consent at the time of recruitment.
References
- 1. Mokdad AA, Lopez AD, Shahraz S, et al. Liver cirrhosis mortality in 187 countries between 1980 and 2010: a systematic analysis. BMC Med 2014;12:145. 10.1186/s12916-014-0145-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Tsochatzis EA, Bosch J, Burroughs AK. Liver cirrhosis. Lancet 2014;383:1749–61. 10.1016/S0140-6736(14)60121-5 [DOI] [PubMed] [Google Scholar]
- 3. Hernandez-Gea V, Friedman SL. Pathogenesis of liver fibrosis. Annu Rev Pathol 2011;6:425–56. 10.1146/annurev-pathol-011110-130246 [DOI] [PubMed] [Google Scholar]
- 4. Bataller R, Brenner DA. Liver fibrosis. J Clin Invest 2005;115:209–18. 10.1172/JCI24282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Parola M, Pinzani M. Liver fibrosis: pathophysiology, pathogenetic targets and clinical issues. Mol Aspects Med 2019;65:37–55. 10.1016/j.mam.2018.09.002 [DOI] [PubMed] [Google Scholar]
- 6. Pellicoro A, Ramachandran P, Iredale JP, et al. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat Rev Immunol 2014;14:181–94. 10.1038/nri3623 [DOI] [PubMed] [Google Scholar]
- 7. Koyama Y, Brenner DA. Liver inflammation and fibrosis. J Clin Invest 2017;127:55–64. 10.1172/JCI88881 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Tacke F, Zimmermann HW. Macrophage heterogeneity in liver injury and fibrosis. J Hepatol 2014;60:1090–6. 10.1016/j.jhep.2013.12.025 [DOI] [PubMed] [Google Scholar]
- 9. Tsuchida T, Friedman SL. Mechanisms of hepatic stellate cell activation. Nat Rev Gastroenterol Hepatol 2017;14:397–411. 10.1038/nrgastro.2017.38 [DOI] [PubMed] [Google Scholar]
- 10. Pradere J-P, Kluwe J, De Minicis S, et al. Hepatic macrophages but not dendritic cells contribute to liver fibrosis by promoting the survival of activated hepatic stellate cells in mice. Hepatology 2013;58:1461–73. 10.1002/hep.26429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hellerbrand C, Stefanovic B, Giordano F, et al. The role of TGFbeta1 in initiating hepatic stellate cell activation in vivo. J Hepatol 1999;30:77–87. 10.1016/s0168-8278(99)80010-5 [DOI] [PubMed] [Google Scholar]
- 12. Seki E, De Minicis S, Osterreicher CH, et al. Tlr4 enhances TGF-beta signaling and hepatic fibrosis. Nat Med 2007;13:1324–32. 10.1038/nm1663 [DOI] [PubMed] [Google Scholar]
- 13. Miura K, Kodama Y, Inokuchi S, et al. Toll-Like receptor 9 promotes steatohepatitis by induction of interleukin-1beta in mice. Gastroenterology 2010;139:323–34. 10.1053/j.gastro.2010.03.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Mattiotti A, Prakash S, Barnett P, et al. Follistatin-Like 1 in development and human diseases. Cell Mol Life Sci 2018;75:2339–54. 10.1007/s00018-018-2805-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Geng Y, Dong Y, Yu M, et al. Follistatin-Like 1 (FSTL1) is a bone morphogenetic protein (BMP) 4 signaling antagonist in controlling mouse lung development. Proc Natl Acad Sci U S A 2011;108:7058–63. 10.1073/pnas.1007293108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Henkel M, Partyka J, Gregory AD, et al. FSTL-1 attenuation causes spontaneous Smoke-Resistant pulmonary emphysema. Am J Respir Crit Care Med 2020;201:934–45. 10.1164/rccm.201905-0973OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Peters MMC, Meijs TA, Gathier W, et al. Follistatin-Like 1 in cardiovascular disease and inflammation. Mini Rev Med Chem 2019;19:1379–89. 10.2174/1389557519666190312161551 [DOI] [PubMed] [Google Scholar]
- 18. Chaly Y, Hostager B, Smith S, et al. Follistatin-Like protein 1 and its role in inflammation and inflammatory diseases. Immunol Res 2014;59:266–72. 10.1007/s12026-014-8526-z [DOI] [PubMed] [Google Scholar]
- 19. Liu Y, Xu J, Liu T, et al. Fstl1 aggravates cigarette smoke-induced airway inflammation and airway remodeling by regulating autophagy. BMC Pulm Med 2021;21:45. 10.1186/s12890-021-01409-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Li G, Ren H, Wu X, et al. Follistatin like protein-1 modulates macrophage polarization and aggravates dextran sodium sulfate-induced colitis. Int Immunopharmacol 2020;83:106456. 10.1016/j.intimp.2020.106456 [DOI] [PubMed] [Google Scholar]
- 21. Chaly Y, Fu Y, Marinov A, et al. Follistatin-Like protein 1 enhances NLRP3 inflammasome-mediated IL-1β secretion from monocytes and macrophages. Eur J Immunol 2014;44:1467–79. 10.1002/eji.201344063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Zheng X, Qi C, Zhang S, et al. TGF-β1 induces Fstl1 via the Smad3-c-Jun pathway in lung fibroblasts. Am J Physiol Lung Cell Mol Physiol 2017;313:L240–51. 10.1152/ajplung.00523.2016 [DOI] [PubMed] [Google Scholar]
- 23. Dong Y, Geng Y, Li L, et al. Blocking follistatin-like 1 attenuates bleomycin-induced pulmonary fibrosis in mice. J Exp Med 2015;212:235–52. 10.1084/jem.20121878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Liu T, Liu Y, Miller M, et al. Autophagy plays a role in FSTL1-induced epithelial mesenchymal transition and airway remodeling in asthma. Am J Physiol Lung Cell Mol Physiol 2017;313:L27–40. 10.1152/ajplung.00510.2016 [DOI] [PubMed] [Google Scholar]
- 25. Zhou X, Xie F, Wang L, et al. The function and clinical application of extracellular vesicles in innate immune regulation. Cell Mol Immunol 2020;17:323–34. 10.1038/s41423-020-0391-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Yang L, Xie M, Yang M, et al. Pkm2 regulates the Warburg effect and promotes HMGB1 release in sepsis. Nat Commun 2014;5:4436. 10.1038/ncomms5436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Moylan C, Diehl AM. Expression data for nonalcoholic fatty liver disease patients. gene expression omnibus, 2019. Available: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE49541
- 28. Mas VR, Maluf DG, Archer KJ. RMA expression data for liver samples from subjects with HCV, HCV-HCC, or normal liver. gene expression omnibus, 2018. Available: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE14323
- 29. Lalloyer F, Bauge E, Pawlak M. Transcriptomic analysis of CLL4-induced liver injury in WT and DpT KO mice. gene expression omnibus, 2019. Available: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE141821
- 30. Misu H, Takamura T. Expression data from mouse liver treated with metformin. gene expression omnibus, 2019. Available: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE35961
- 31. Gijbels E, Devisscher L, Vinken M. Robustness testing and optimization of an adverse outcome pathway on cholestatic liver injury. gene expression omnibus, 2020. Available: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE15249432 [DOI] [PubMed]
- 32. Li X, Li L, Chang Y, et al. Structural and functional study of FK domain of FSTL1. Protein Sci 2019;28:1819–29. 10.1002/pro.3696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Wang P, Sun C, Zhu T, et al. Structural insight into mechanisms for dynamic regulation of PKM2. Protein Cell 2015;6:275–87. 10.1007/s13238-015-0132-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Dayton TL, Jacks T, Vander Heiden MG. Pkm2, cancer metabolism, and the road ahead. EMBO Rep 2016;17:1721–30. 10.15252/embr.201643300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Luo W, Hu H, Chang R, et al. Pyruvate kinase M2 is a PHD3-stimulated coactivator for hypoxia-inducible factor 1. Cell 2011;145:732–44. 10.1016/j.cell.2011.03.054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Anastasiou D, Yu Y, Israelsen WJ, et al. Pyruvate kinase M2 activators promote tetramer formation and suppress tumorigenesis. Nat Chem Biol 2012;8:839–47. 10.1038/nchembio.1060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Guo J, Ren R, Yao X, et al. Pkm2 suppresses osteogenesis and facilitates adipogenesis by regulating β-catenin signaling and mitochondrial fusion and fission. Aging 2020;12:3976–92. 10.18632/aging.102866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Palsson-McDermott EM, Curtis AM, Goel G, et al. Pyruvate kinase M2 regulates HIF-1α activity and IL-1β induction and is a critical determinant of the Warburg effect in LPS-activated macrophages. Cell Metab 2015;21:65–80. 10.1016/j.cmet.2014.12.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Xu R, Zhang Z, Wang F-S. Liver fibrosis: mechanisms of immune-mediated liver injury. Cell Mol Immunol 2012;9:296–301. 10.1038/cmi.2011.53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Sica A, Invernizzi P, Mantovani A. Macrophage plasticity and polarization in liver homeostasis and pathology. Hepatology 2014;59:2034–42. 10.1002/hep.26754 [DOI] [PubMed] [Google Scholar]
- 41. Ramnath D, Irvine KM, Lukowski SW, et al. Hepatic expression profiling identifies steatosis-independent and steatosis-driven advanced fibrosis genes. JCI Insight 2018;3:e120274. 10.1172/jci.insight.120274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Ramachandran P, Dobie R, Wilson-Kanamori JR, et al. Resolving the fibrotic niche of human liver cirrhosis at single-cell level. Nature 2019;575): :512–8. 10.1038/s41586-019-1631-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Li D, Wang Y, Xu N, et al. Follistatin-Like protein 1 is elevated in systemic autoimmune diseases and correlated with disease activity in patients with rheumatoid arthritis. Arthritis Res Ther 2011;13:R17. 10.1186/ar3241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Fang D, Shi X, Lu T, et al. The glycoprotein follistatin-like 1 promotes brown adipose thermogenesis. Metabolism 2019;98:16–26. 10.1016/j.metabol.2019.05.008 [DOI] [PubMed] [Google Scholar]
- 45. Seki M, Powers JC, Maruyama S, et al. Acute and chronic increases of circulating FSTL1 normalize energy substrate metabolism in pacing-induced heart failure. Circ Heart Fail 2018;11:e004486. 10.1161/CIRCHEARTFAILURE.117.004486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Yang W, Zheng Y, Xia Y, et al. Erk1/2-Dependent phosphorylation and nuclear translocation of PKM2 promotes the Warburg effect. Nat Cell Biol 2012;14:1295–304. 10.1038/ncb2629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Tannahill GM, Curtis AM, Adamik J, et al. Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature 2013;496:238–42. 10.1038/nature11986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Kelly B, O'Neill LAJ. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res 2015;25:771–84. 10.1038/cr.2015.68 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
gutjnl-2021-325150supp001.pdf (126.1KB, pdf)
gutjnl-2021-325150supp002.pdf (151KB, pdf)
gutjnl-2021-325150supp003.pdf (1.9MB, pdf)
Data Availability Statement
Data are available in a public, open access repository. A generic lab email address or an individual’s ORCID identifier.