Abstract
The twenty-first century has seen the emergence of many epidemic and pandemic viruses, with the most recent being the SARS-CoV-2-driven COVID-19 pandemic. As obligate intracellular parasites, viruses rely on host cells to replicate and produce progeny, resulting in complex virus and host dynamics during an infection. Single-cell RNA sequencing (scRNA-seq), by enabling broad and simultaneous profiling of both host and virus transcripts, represents a powerful technology to unravel the delicate balance between host and virus. In this review, we summarize technological and methodological advances in scRNA-seq and their applications to antiviral immunity. We highlight key scRNA-seq applications that have enabled the understanding of viral genomic and host response heterogeneity, differential responses of infected versus bystander cells, and intercellular communication networks. We expect further development of scRNA-seq technologies and analytical methods, combined with measurements of additional multi-omic modalities and increased availability of publicly accessible scRNA-seq datasets, to enable a better understanding of viral pathogenesis and enhance the development of antiviral therapeutics strategies.
Keywords: Single-cell RNA sequencing, Antiviral immunity, Virus, Transcriptomics
Introduction
Viral infectious diseases perennially threaten global health. The most recent pandemic, COVID-19, has globally accounted for more than 584 million SARS-CoV-2 infections and 6.4 million deaths as of August 2022 [1]. Each new viral species, strain, and mutation can influence disease severity through complex host-virus interactions [2–4]. As obligate intracellular parasites, viruses require host cellular machinery for replication. In response, host cells employ antiviral mechanisms to recognize and restrict viral replication [5]. The arms race between virus and host at the single-cell level collectively drives variability within virus and host cell populations, impacting disease pathogenesis and epidemiological dynamics. Untangling intra- and inter-individual heterogeneity in responses to viral infection requires a high-resolution analysis of viral dynamics and the ensuing host response.
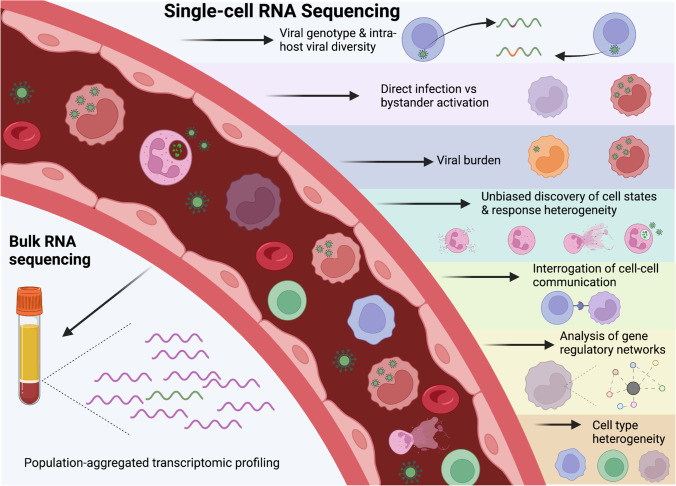
While bulk RNA-seq studies have been instrumental to our understanding of cellular antiviral responses [6–9], these methods average over a population of cells, thereby obscuring underlying heterogeneity. In contrast, single-cell RNA sequencing (scRNA-seq) allows for transcriptome-wide profiling at the resolution of the individual cell, providing a powerful method to interrogate the transcriptomic heterogeneity of cellular responses. This single-cell resolution enables understanding of variations in host responses that can be driven by factors that include direct viral infection versus activation of bystander cells [10–13], viral genotype and intra-host viral diversity [14–17], cell type heterogeneity in disease severity and responses [11, 18–21], and impacts of viral burden on host responses [11, 22, 23] (Fig. 1). scRNA-seq has previously been used to investigate infection by diverse viruses including herpes simplex virus [24, 25], reovirus [26], dengue virus [11, 19, 27], influenza virus [28, 29], HIV [20, 30], hepatitis B virus [10], and SARS-CoV-2 [18, 31–33].
Fig. 1.
Transcriptomic approaches to profile antiviral immunity
In this review, we aim to provide researchers with tools to inform scRNA-seq study design for understanding virus-host interactions, as well as encourage increased studies across diverse viral species. We highlight various scRNA-seq technologies and methods, from sample processing to computational analyses, and the power and nuances of each in examining antiviral immunology. Throughout, we will present important contributions of previous scRNA-seq studies to our understanding of viral immunology while highlighting areas of scRNA-seq development that show promise. With continued scRNA-seq studies and data sharing, together, this work will expand our understanding of immunological responses across all viruses and drive innovation towards antiviral interventions to combat current and emerging viruses to enhance pandemic preparedness [34].
Technologies for deep transcriptional profiling of antiviral immunity
Overview of scRNA-seq workflow
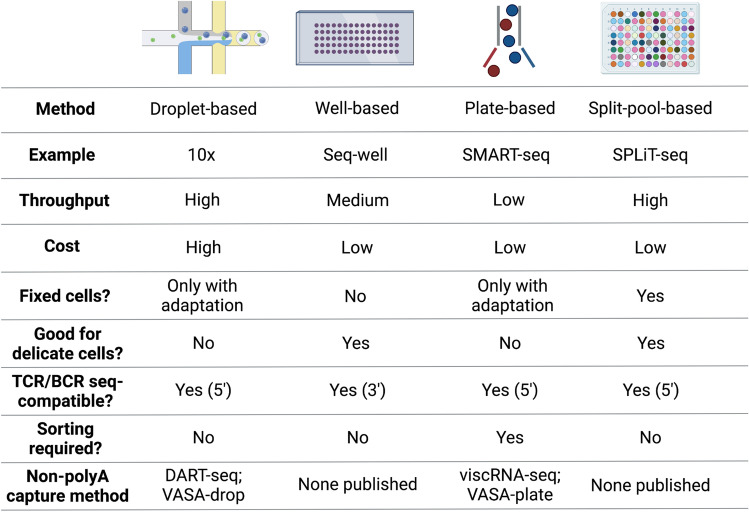
The last several years have seen a proliferation of scRNA-seq platforms, each with distinct advantages in scalability, flexibility, applications, and cost. Fundamentally, all scRNA-seq platforms involve the same basic steps: (1) nucleotide barcoding of single cells; (2) cell lysis; (3) capture of mRNA; (4) generation of cDNA through reverse transcription; (5) cDNA amplification by PCR; (6) cDNA library preparation; and (7) sequencing. These methods utilize unique barcoding of each cell to identify each transcript’s cell of origin, with some methods also including unique molecular identifiers (UMIs) which can be added to each transcript prior to library amplification to reduce amplification bias. However, there are important distinctions between the different strategies of performing scRNA-seq which impact each method’s ability to capture transcripts from particular cell types and viruses. Here, we discuss four classes of scRNA-seq technologies that are distinguished by their strategies for single-cell nucleotide barcoding: droplet-based (e.g., 10X), well-based (e.g., Seq-Well), plate-based (e.g., Smart-Seq), and split-pool-based (e.g., SPLiT-seq). In the following sections, we will describe the advantages and limitations of each technology, with a focus on how they can be applied to the study of viral infectious diseases (Fig. 2).
Fig. 2.
Table summarizing the advantages and disadvantages of different scRNA-seq methods in viral immunology research
Droplet-based methods
Droplet-based sequencing methods rely on microfluidic mechanisms to encapsulate individual cells with beads bearing cell barcodes in oil droplet emulsions [35]. The use of barcoded beads allows for the identification of transcripts from the same cell during downstream analysis, as each cell co-localizes with one bead bearing a unique oligonucleotide barcode that labels all transcripts in a given droplet. These cells are lysed within these droplets, allowing transcripts to hybridize into the mRNA capture beads. Next, depending upon the platform, reverse transcription can happen either within the droplet or after the demulsification of the droplet. After transcript capture and reverse transcription, standard library preparation procedures occur during which UMI barcodes are added to each transcript prior to PCR amplification. The standard droplet-based platforms include inDrop and Drop-Seq [36, 37], both open-source methods, and the commercially available methods from 10X Genomics Chromium. Differences between these platforms include bead composition, timing of cDNA synthesis, barcode design, and sequence processing. These methods’ power lies in their ease of use and flexibility, with 10X methods particularly increasing the capacity to profile thousands of cells. Recently, 10X Genomics developed the Chromium X controller that pushes high-throughput scRNA-seq sequencing from 10 to 60 K cells per sample by adding additional sensors and more accurate pressure and temperature control during droplet formation. This depth in cellular sequencing enables the profiling of rarer cell states and cell types that may be missed otherwise.
The flexibility and modularity of bead construction have enabled the development and commercialization of multiple different kits on the same platform. For example, beads can be designed to hybridize either the 5’ or the 3’ ends of mRNAs and can include additional primers to enrich for particular transcripts, like B cell receptor (BCR) and T cell receptor (TCR) sequences. Beads can also include oligonucleotides to capture transposed accessible chromatin fragments or antibody-conjugated oligonucleotides, thereby performing multimodal epigenetic or proteomic profiling, respectively. However, this system has difficulty capturing usable transcripts from neutrophils and other granulocytes, even when these cells are freshly isolated, in part due to their high RNAse levels and sensitivity to degranulation during microfluidics processing [38, 39]. While 10X recommends the removal of granulocytes from samples, for those that want to profile these cells, 10X provides recommendations [40, 41] for capturing granulocyte sequences and these have successfully been shown to work on neutrophils [42]. A direct comparison of 10X, inDrop, and Drop-seq found that 10X has the highest molecular sensitivity, highest precision, and least technical noise, though 10X is more expensive and dependent on proprietary reagents [43]. Many studies have leveraged droplet-based methods to analyze thousands of cells across various virus infections [44–48]. Without the constraints of array size, droplet-based methods are highly scalable.
Well-based methods
Well-based scRNA-seq platforms, such as Seq-Well [49, 50] or the BD Rhapsody platform [51, 52], achieve single-cell barcoding by loading cell suspensions onto microarrays pre-loaded with oligonucleotide-barcoded mRNA capture beads. In Seq-Well, cells are lysed within microarrays sealed with a semipermeable membrane, allowing the RNA from each cell to be captured on an individual barcoded bead and preventing mRNA contamination from neighboring wells. Following capture, the bead-bound RNAs can be released from the arrays and processed for reverse transcription, library preparation, and sequencing.
In addition to being much cheaper than droplet-based scRNA-seq methods, well-based methods have the advantage of being gentler on the cells they process. Importantly, this allows well-based methods to capture sensitive cell types, such as neutrophils, that may be lost during a microfluidic-based workflow. The ability of well-based methods to successfully preserve the transcriptomes of neutrophils has proven invaluable in the study of viral infections, particularly COVID-19 [18, 31, 45]. Seq-Well-based studies have identified key features of neutrophil activation and dysfunction in severe COVID-19 that appear to contribute to disease pathology [18]. Moreover, these studies discovered a population of immature neutrophils that emerges in the peripheral blood of COVID-19 patients that predicts 28-day mortality [18, 31, 45].
The portability and economy of well-based methods have also made it possible to perform scRNA-seq in extraordinary conditions. In a 2020 study, researchers performed Seq-Well on peripheral blood mononuclear cells (PBMC) from non-human primates infected with the Ebola virus under BSL4 conditions [23]. However, the scalability of well-based methods is relatively limited. Additionally, well-based methods are currently limited to use on unfixed cells.
Plate-based methods
Plate-based methods, such as Smart-seq [53] and MARS-seq [54], rely on cell sorters to place individual cells into individual wells across 96- or 396-well plates. These wells contain lysis buffer and cell barcodes, and can include additional UMIs and plate barcodes. These protocols are amenable to automation with liquid-handling robots and do not require specialized equipment besides a cell sorter and a PCR machine. The Smart-seq protocols are able to capture full-length transcriptome coverage, with methodology improvements made in Smart-seq2 [55] and Smart-seq3 [56] protocols allowing for higher coverage and sensitivity of detected transcripts. The per-plate nature of processing tends to lower throughput (96 or 364 per plate) and technical variability can be introduced in processing steps (e.g., thermocycler used, pipetting steps) that may contribute to batch effects between plate and experiment.
The cell sorting step can be made specific to the inclusion/exclusion of particular cell types (e.g., removal of erythrocytes and doublets, inclusion of specific populations) to thereby be well suited for profiling rarer cell populations. Additionally, using fluorescence-activated cell sorting (FACS) of cells prior to plating allows for cell types to be identified prior to their sequencing, which can help distinguish cell types that are difficult to identify using transcriptomics alone (e.g., intermediate monocytes, NK cells, T cells) and/or gain better sequencing depth into populations that might not be evenly sampled in a pooled scRNA-seq method. A key advantage of these methods is their usefulness in investigating the mechanism and/or transcriptomics of specific cell subsets (e.g., rare cell type, functionally responding cell type, viral-protein expressing cells).
An example of the usefulness of pre-sorting populations comes from Steuerman et al. [29] who FACS-sorted CD45 + (immune) and CD45- (non-immune) populations from the lungs of influenza-infected mice and performed MARS-seq to identify virus and host cell transcripts [29]. This approach enabled sufficient depth of sequencing into both populations to identify multiple immune and non-immune cell types associating with varying proportions of infected cells. While different cell types carried varying levels of influenza transcript load, all infected cells—independent of cell type—demonstrated a conserved transcriptional response marked by repression of mitochondrial-related transcripts. While its strengths lie in pre-sorting to better understand the input cell population prior to sequencing, the sorting associated with plate-based methods can add time and manipulation into sample processing and, paired with the use of fixed plates, can make this method lower-throughput in comparison to droplet-based methods.
Split-pool methods
Split-pool sequencing is a relatively new technique that leverages combinatorial barcoding to identify individual cells rather than using a physical partition such as a droplet or a microwell [57]. Split-pool scRNA-seq begins by performing reverse transcription on fixed and permeabilized cells to add a sample-specific oligonucleotide barcode to all mRNAs in each sample. The cells are then pooled, redistributed, and barcoded multiple times such that each cell receives a unique combination of barcodes. When performed on a 96-well plate, four rounds of barcoding are sufficient to create over 20 million unique barcode combinations, which can label up to an estimated 1 million cells without creating a significant number of multiplets. Once the four barcoding steps are completed, the cells are lysed and libraries are prepared for sequencing [57].
Split-pool sequencing has the major advantage of needing no specialized equipment. Moreover, split-pool-based platforms are designed to be compatible with paraformaldehyde-fixed samples, making it particularly attractive for researchers working with virus-infected samples that require a high level of biosafety or have cells that can’t be processed immediately (e.g., when thawed for use across multiple experiments). Like well-based platforms, split-pool sequencing technologies are gentle enough for use on delicate cells such as neutrophils that are sensitive to microfluidics. Finally, these methods allow for easy multiplexing of a large number of samples in one experiment.
Single-cell TCR/BCR sequencing
Technologies relying solely on short-read sequencing of mRNA, while generally appropriate for measuring the abundance levels of most genes, are insufficient for the capture and reassembly of the more complex transcripts encoding the exquisitely specific receptors expressed on T cells (TCRs) and B cells (BCRs). In order to create the extraordinary diversity of TCR and BCR sequences found in humans, DNA segments of the genes encoding these receptors are rearranged in a process called V(D)J recombination. Additional diversity is introduced post-transcriptionally, when the transcripts undergo alternative splicing to produce unique genes that give rise to uniquely specific receptors. Therefore, in order to successfully capture the full diversity of a given V(D)J region, long-read sequencing techniques are necessary. Moreover, while traditional single-cell RNA sequencing approaches capture the 3’ ends of transcripts, the rearranged V(D)J region of a BCR or TCR is located at the 5’ end of the transcript, making it difficult to capture with short-read approaches [58].
In 2019, Repertoire And Gene Expression by Sequencing (RAGE-seq) was developed to pair full-transcript sequencing with 5’ capture in order to robustly resolve TCR and BCR sequences in a high-throughput manner [58]. This method is most commonly used in conjunction with microfluidics-based platforms such as those offered by 10X Genomics, though similar methods have now been developed for compatibility with well-based platforms, including BD Rhapsody and Seq-Well, which use a unique 3’ approach to capture TCR sequences [59]. The advent of techniques that allow for simultaneous study of the whole transcriptome and TCR/BCR sequences provides the opportunity to analyze the transcriptomes of cells with specific TCR or BCRs, for example, clonally expanded populations [60–64].
A limitation of studying antigen-specific T cells is that these populations can be too small to serve as inputs into many scRNA-seq methods. SELECT-seq was developed as a method to address this limitation and gather both TCR sequencing and cellular transcriptomic information for rare/specific cell populations of interest. SELECT-seq does this by using a modified Smart-Seq2 protocol to generate cDNA libraries from single T cells, then taking an aliquot of each library for nested PCR to amplify CDR3 regions of both TCRɑ and TCRβ chains [65]. T cell libraries were selected for further high-coverage in-depth whole transcriptome sequencing based upon whether there were duplicated CDR3 regions present (assumed to be clonally expanded). The authors utilize this method to identify transcriptional differences between CD8 + T cell populations with high versus low clonal expansions. They show that they are able to select for CMV-reactive CD8 + T cell populations (by activating T cells with CMV peptide) and identified that clones with low expansion had increased IL2RA (T cell activation signaling receptor) and CD27 and CD28 (costimulatory markers) while highly expanded clones had increased expression of TIGIT (coinhibitory receptor) and KLRG1 (senescent marker). The advantage of this method is that it reduces costs associated with whole transcriptome sequencing by allowing users to select for cell populations of interest as well as for specific TCR clones for limited scRNA-seq.
LInking B-cell Receptor to Antigen specificity through Sequencing (LIBRA-seq) can be used to interrogate the antigen specificity of BCRs while simultaneously collecting BCR sequences and whole transcriptome data at the single-cell level. Briefly, LIBRA-seq involves exposing B-cells to a pool of oligonucleotide-barcoded antigens. Antigens bound by a B-cell are then captured within a droplet alongside the cell so that the antigen barcode can be sequenced in conjunction with the B-cell’s mRNA, thereby revealing the specificity of that cell’s BCR [66]. LIBRA-seq has also been leveraged for the discovery of novel broadly neutralizing antibody lineages in HIV-infected patients [66] and the identification of novel neutralizing antibodies against SARS-CoV-2 [67]. It can also be used to interrogate the transcriptomes of viral antigen-specific B-cells [66].
Adaptations to established scRNA-seq methods for measuring viral transcripts
Capture of non-polyadenylated viral transcripts
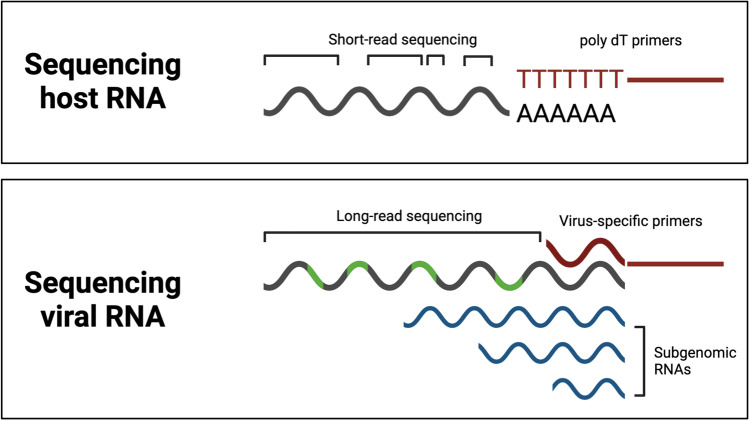
Most scRNA-sequencing methods that perform 3’ mRNA capture (e.g., Seq-Well, BD Rhapsody, Drop-Seq, inDrop, and several 10X Genomics kits) utilize poly-T oligonucleotide (oligo(dT)) primers to capture polyadenylated transcripts associated with human mRNA, while limiting the measurement of highly abundant ribosomal RNAs [68]. Because many viruses are polyadenylated, they can be detected through standard methods: for example, SARS-CoV-2 transcripts can be measured using standard 10X 3’ methods [69, 70] and Ebola virus can be measured using the standard Seq-Well protocol [23].
However, a limitation of many scRNA-seq methods is that they cannot capture non-polyadenylated transcripts. Some viruses, such as those in the Flaviviridae family (e.g., dengue virus, Zika virus, yellow fever virus, and hepatitis C virus), generate non-polyadenylated transcripts. Hence, if the study goal is to understand host responses in relation to viral infection dynamics, it can be important to choose a sequencing method that will capture non-polyadenylated reads. There are numerous methods that have been developed to measure non-polyadenylated transcripts, some of which have been utilized in the context of viruses. For example, virus-inclusive single-cell RNA-Seq (viscRNA-Seq) adapts the Smart-seq2 plate-based scRNA-seq protocol to pair standard oligo(dT) primers with virus-specific primers prior to cDNA generation (Fig. 3). As part of viscRNA-seq, amplified cDNA is split into aliquots for two purposes: (1) qPCR for viral RNA that can be associated with (2) sequencing for host transcripts [11]. This method measured viral RNA transcripts from the dengue virus and Zika virus from in vitro infections and recovered dengue viral transcripts from the PBMCs of dengue patients to identify viral tropism [11].
Fig. 3.
Schematic demonstrating key differences between host and viral RNA that can require alternative approaches for sequencing virus transcripts
Droplet-assisted RNA targeting by single-cell sequencing (DART-seq), which is an adaption of Drop-seq to include multiplexed RNA amplicon sequencing, captured the non-polyadenylated viral transcripts in an in vitro reovirus infection [26]. Other methods that utilize intentional primer design to generally capture both polyadenylated and non-polyadenylated transcripts include vast transcriptome analysis of single cells by dA-tailing (VASA-seq) which can be adapted in droplet workflows (VASA-drop) and plate-based workflows (VASA-plate) [71]; single-cell universal poly(A)-independent RNA sequencing (SUPeR-seq) [72]; and multiple annealing and dC-tailing-based quantitative single-cell RNA-seq (MATQ-seq) [73].
Long-read sequencing for viral genomes
Many RNA viruses have low-fidelity, error-prone polymerases that introduce mutations into the viral genome upon replication. For example, estimates for HIV-1’s RNA-dependent DNA polymerase predict around ~ 5–10 errors per HIV-1 genome per replication round [74]. Thus, during an in vitro or in vivo viral infection, viruses can exist as a viral quasispecies—a population of viruses differing in genetic variation leading to competent and/or defective viral particles that can differentially infect and drive host pathogenesis [73]. scRNA-seq studies that examine host antiviral responses without measuring viral genetic diversity are studying the combined effect of various viral mutations and defects. Most scRNA-seq methods incorporate library fragmentation for downstream short-read sequencing, but viral genomes span from 1–2 kb (Circoviruses) [75] to ~ 30 kb (SARS-CoV-2) and past 1000 kb (Mimiviruses) [76], allowing these methods to only capture a fraction of most viral genomes. Thus, methods that incorporate long-read sequencing empower an understanding of viral genomic diversity in relation to cellular responses in addition to viral transcript abundance (Fig. 3). One such method was developed by Russell et al. which utilizes 10X technology to generate cell-barcoded cDNA from influenza virus (IAV)–infected cells and split the cDNA for two measurements: (1) standard downstream fragmentation and short-read sequencing and (2) enrichment of IAV-specific transcripts through PCR amplification and full-length sequencing using PacBio methods [14]. The group demonstrated that two-thirds of IAV-infected cells had mutations or defects in one or more of IAV’s 7 genomic segments and were able to use this integrated data to identify four IAV defects that correlated with increased cellular IFN induction and further validate these in vitro. An alternative approach, Single-cell Nanopore sequencing with UMIs (ScNaUmi-seq), leverages the 10X Genomics Chromium system to generate cell-barcoded cDNA products that are prepared downstream for Oxford Nanopore long-read sequencing [77].
Insights from single-cell transcriptional profiling of antiviral immunity
Unbiased discovery of cell type
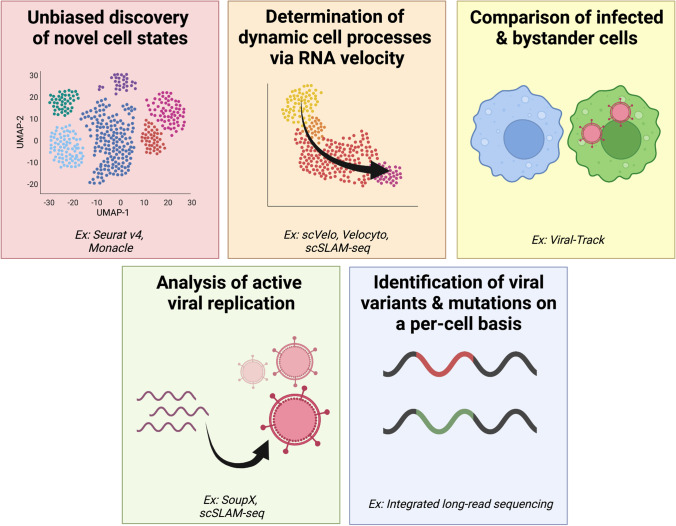
By profiling cellular phenotype at the transcriptome-wide scale, scRNA-seq enables the discovery of cell type and state that is not biased by the selection of marker panels, as in flow or mass cytometry (Fig. 4). Unbiased discovery of cell type generally relies on unsupervised clustering analysis, which groups transcriptionally similar cells into “clusters” based on gene expression. Most toolkits for scRNA-seq computational analysis, including Seurat, scanpy, and Monocle, include implementations of several graph-based unsupervised clustering algorithms; the strengths and weaknesses of these various algorithms have been extensively assessed and reviewed elsewhere [78–81]. The use of unsupervised clustering to assess cell type composition in scRNA-seq data is advantageous because it does not require a priori knowledge, allowing for easy discovery of unexpected populations.
Fig. 4.
Overview of ways in which scRNA-seq can enable a deeper understanding of cellular antiviral responses and viral dynamics
Analyses of COVID-19 patient samples exemplified the utility of leveraging unsupervised clustering approaches for cell type discovery. Early evidence of emergency myelopoiesis in severe COVID-19 came from scRNA-seq datasets that unexpectedly found immature neutrophils in the peripheral blood of patients with severe COVID-19 [31], which was directly enabled by performing transcriptome-wide measurements. Broadly, cell type annotation in scRNA-seq can be accomplished by manually assigning identities to cell clusters, or by using automated tools to annotate individual cells. In the absence of a suitable transcriptomic reference dataset, differentially expressed genes (DEGs) for each cluster can be compared to known or expected transcriptomic profiles from the literature to call cell types for each cluster [82].
Reference datasets can greatly simplify this task, and a variety of automated tools have been developed to leverage transcriptomic references to annotate query datasets (reviewed by [83]). Generally, these tools operate either by correlating the transcriptomic profile of each single query cell to a reference bulk or single-cell profile (scmap [84], SingleR [85], scMatch [86]), by identifying mutual nearest neighbors between reference and query cells and transferring cell type labels (Seurat v4 [87], scArches [88]), or by performing supervised classification (SingleCellNet [89], scClassify [90], LAmbDA [91], scPred [92]).
Choosing a suitable reference is a critical and challenging step in any automated cell annotation process. The most robust reference-based automated cell type annotations would ideally use references that contain the same cell types as the query and from the same tissue niche. Most reference datasets are derived from homeostatic tissue niches, and care must be taken when interpreting reference-based cell type annotations in a highly perturbed query dataset. For example, there are frequent situations where unexpected cell types are present outside of their typical tissue niche. For example, when emergency myelopoiesis results in immature myeloid cells being present in the peripheral blood [31, 45], applying peripheral blood references to peripheral blood datasets with emergency hematopoiesis [93] will lead to incorrect and misleading cell type annotations [93]. Thus, reference-based cell type annotation should always be paired with manual verification based on biological knowledge and expectations.
In addition to generating and annotating new references, there is an ongoing effort in cell type annotation to incorporate additional data modalities into assigning cell type identity. For instance, T cells contain low RNA content that makes biologically distinct T cell subtypes difficult to distinguish based on transcriptomic data alone [87]. However, these subtypes can be readily dissected by their cell surface proteome. Data from single-cell multimodal methods that capture both transcriptome and cell surface protein data [94] thus have the potential to solve this problem. Recently, Hao et al. [87] introduced weighted nearest neighbor (WNN) analysis to leverage multiple data modalities for neighbor graph generation, clustering, integration, and data transfer. WNN analysis learns, for each cell, which data modality best predicts that cell’s identity and uses these single-cell modality weights for downstream analytical tasks. This approach allows highly accurate (r = 0.91) quantification of difficult-to-identify T cell subsets like mucosal-associated invariant T (MAIT) cells [87]. We anticipate that the continued publication of additional multimodal references, by integrating additional dimensions of a cell’s biological identity, will enable even more robust cell type annotations.
Analysis of dynamic cellular processes
Viral infection can drive cells towards different states with diverse functional consequences. scRNA-seq powerfully reveals RNA abundances of cells at the time of sample collection; however, transcript abundances alone are not enough to determine drivers of infection-altered cell trajectories, which are important to elucidating disease pathogenesis. RNA velocity seeks to derive cell trajectory dynamics by leveraging explicit measurements of newly transcribed pre-mRNAs (unspliced) and mature mRNAs (spliced) in order to estimate gene splicing and degradation rates. These measurements can help infer the continuous, dynamic spectra of cell states and estimate an individual cell’s position in that spectrum as a pseudotime value (Fig. 4). Both scVelo [95] and Velocyto [96] are both tools that enable an analysis of this information. RNA velocity has limitations, including its high dependency on the k-NN graph built on the data, the cells included in the collected data, and its strong dependence on two-dimensional representations for visualization built on observed transcriptional data that do not fully capture cell-state transitions [97]. The recently developed veloViz addresses some of these limitations by incorporating RNA velocity information into 2D and 3D embeddings to better capture cellular trajectories even when intermediate cell types are missing [98].
RNA velocity has been leveraged to understand drivers of myelopoiesis and lymphopenia seen in severe COVID-19 patients. To do so, Wang et al. [99] profiled bone marrow mononuclear cells of COVID-19 patients and utilized RNA velocity to identify differences in hematopoiesis. The study found that hematopoietic stem cells from patients with severe COVID-19 demonstrated preferential differentiation trajectories towards granulocyte-monocyte progenitors and away from lymphoid progenitors, potentially underlying differences in myeloid and lymphoid cell proportions in the blood of severe COVID-19 patients.
While RNA velocity can be inferred from general scRNA-seq methods, metabolic labeling combined with scRNA-seq can enable time-resolved scRNA-seq, or tscRNA-seq, through direct experimental measurements of “new” and “old” RNA molecules to more accurately measure RNA turnover rates and infer cell-state transitions. The method, scSLAM-seq (single-cell, thiol-(SH)-linked alkylation of RNA for metabolic labeling sequencing), integrates metabolic RNA labeling and biochemical nucleoside conversion with scRNA-seq to directly measure RNA turnover on the basis of U-to-C conversion rates at the single-cell level [100]. Proof-of-concept scSLAM-seq experiments on mouse fibroblast cells infected with mouse cytomegalovirus (MCMV) showed the robustness of this experimental method to identify intermittent “bursting” kinetics (periods of transcription separated by transcriptional inactivity) of genes in response to MCMV infection. An open-source software, dynamo, robustly integrates RNA metabolically labeled data with scRNA-seq splicing kinetics to show an increased accuracy in RNA velocity estimates in comparison to RNA velocity analysis on solely scRNA-seq splicing data when used on a metabolically labeled human hematopoiesis scRNA-seq dataset [101].
Analysis of viral dynamics
Impact of viral transcript abundance on cellular responses
Using an scRNA-seq method optimized to measure virus transcripts of interest can elucidate the impact of viral presence on cell function (Fig. 4). Identification of cells containing viral reads versus “bystander” (exposed but uninfected) cells can be accomplished by including the viral genomes of interest into the genome to which scRNA-seq transcripts are aligned. Additionally, Viral-Track introduced a reference genome (curated by Stano et al. [102]) that includes over 1000 virus genomes which can be used to detect both expected and unexpected viral infections [10, 32]. This viral reference genome has been used to identify a putative SARS-CoV-2 and metapneumovirus co-infection in a severe COVID-19 patient [10]. Annotating infected versus bystander cells allows the identification of differentially expressed genes (DEGs) that correlate with viral gene presence, and has been used to identify host gene correlates of lymphocytic choriomeningitis virus (LCMV) infection of mice in vivo [10].
Unlike in typical bulk RNA-seq workflows, UMI labeling of unique transcripts prior to amplification in scRNA-seq methods allows for better estimation of the interconnectedness between viral abundance and cellular responses. Correlation methods, such as Pearson’s or Spearman’s rank correlation, can be utilized to connect intracellular viral abundance to cellular host responses. Utilizing viscRNA-seq of cells infected in vitro with either dengue or Zika virus, Zanini et al. [19] performed Spearman’s rank correlations of all host genes against viral RNA abundance to identify a conserved positive correlation between host transcripts involved in the endoplasmic reticulum (ER) unfolded protein response (UPR) and the abundance of Zika and dengue viral transcripts, relevant as both viruses’ replication and translation processes are largely restricted to the ER. In another example, Shnayder et al. [21] used scRNA-seq to show that lytic and latent human cytomegalovirus (HCMV) infections were distinguished by viral transcript abundance, but had similar host expression programs associated with a viral infection. Although the ability to correlate viral transcript abundance with host transcriptional programs is a powerful tool, it is important to keep in mind the challenges in capturing and accurately quantifying viral transcripts as discussed throughout this review.
Additionally, there can exist heterogeneity in which particular viral genes are expressed or not expressed that can impact host responses. Sun et al. [103] infected cells in vitro with influenza A virus (IAV), performed 10X-based scRNA-seq, and aligned transcriptomic data to a combined reference of both human and influenza virus (IAV) genomes. Among cells infected with IAV, the authors found substantial heterogeneity in host and viral gene expression [103]. With the IAV genome composed of 8 segments, while a majority of infected cells expressed genes from all 8 segments, others expressed transcripts from different subsets of these segments with few cells expressing transcripts from only 1–2 segments. The authors demonstrated that this heterogeneity was the result of cells expressing viral genes from a variable combination and number of IAV genome segments. For example, cells that did not express the IAV nonstructural segment (NS) demonstrated increased ISG and IFN-related transcripts—an expected outcome as IAV NS1 is known to suppress antiviral responses [2]. The identification of variable viral gene expression dynamics and their differential impact on host immune responses was directly enabled by a combination of single-cell resolution profiling and unbiased mapping of host and viral transcripts.
Determining active replication by scRNA-seq
A challenge in scRNA-seq studies is the ability to determine whether measured viral transcripts are associated with actively replicating viral infection rather than non-replicating internalized, extracellularly bound, or ambient extracellular viral RNAs (e.g., those from cell supernatants, bound infection inoculum, virus from lysis of an infected cell, internalized via phagocytosis). Analytic methods to detect and remove contaminating ambient RNA transcripts have been developed by Kotliar et al. [23] to identify intracellular Ebola virus transcripts and by Young and Behjait [104], who developed SoupX (Fig. 4). These methods follow a similar workflow: (1) estimate the ambient RNA profile for empty droplets, (2) estimate the fraction of each cell’s transcript associated with the ambient RNA profile, (3) determine the level of ambient RNA contamination and correct the expression profile, which helps direct analyses towards cells with actively replicating RNA.
Additionally, methods including scSLAM-seq that utilize metabolic labeling of new and old RNA transcripts to infer active versus ambient transcripts [100] can help understand viral dynamics as well. Erhard et al. [100] showed that among cells infected with mouse cytomegalovirus (MCMV), scSLAM-seq was able to distinguish between “older” viral transcripts (hypothesized to be virion-associated RNA delivered to the cell) versus “newer” viral transcripts (hypothesized to be associated with actively replicating virus) to more accurately identify infected cells.
Additionally, a priori knowledge of the virus of interest can inform analyses of viral replication dynamics. In the context of DNA viruses and/or latent viruses, sequenced viral transcripts could indicate replicating and/or reactivating DNA viruses as scRNA-seq processing selects for RNA, therefore not measuring DNA viral genomes and genomically integrated virus. Authors studying herpes simplex virus (HSV)-1-infected cells, a virus that can exist in either a latent (quiescent) or lytic (active) state, were able to use measured HSV-1 transcripts as a correlate of lytic infection to identify the stepwise progression of viral gene program transcription as infection progressed and host transcripts associated with restricting viral infection [24]. In another example, utilizing the method Viral-Track to detect viral transcripts present in scRNA-seq data, researchers were able to identify human metapneumovirus (hMPV) present in a patient with severe SARS-CoV-2 infection [10]. Utilizing coverage analysis and hMPV virology, authors identified a biased nature to the transcripts (higher abundance of N, P, M, F, M2, SH, G, and lower abundance of L hMPV genes) suggesting the presence of actively replicating virus in samples at the time of collection. Another method, scCoVseq, can measure subgenomic RNA transcripts which are generally only present during active viral replication, thus having the potential to identify cells hosting actively replicating virus across coronavirus or nidovirus infections more generally [105]. Furthermore, analysis of strandedness of viral transcripts can identify potential for replicative infection: for example, if analyzing an infection by a positive-sense RNA virus, detection of negative-stranded RNA transcripts (needed as a template to build positive-sense genomic RNA to package into progeny virus particles) could indicate active replication. However, read alignment softwares such as Cell Ranger ignore antisense transcripts, so intentional design of alignment genome and processing pipelines is important to detect this information. These current analytical tools generally require a strong understanding of viral replication and viral gene expression dynamics in order to make conclusions about infection type, with the gold-standard confirmation method being plaque assay validation to prove active infection. More work is necessary to build tools across virus families to identify active versus ambient and extracellular viral RNA in scRNA-seq data.
Deriving the differential impact of viruses within a viral quasispecies
Viruses can exist as a heterogeneous quasispecies during infection, with virions that can include a range of genomic mutations and defects. This viral genetic diversity can heterogeneously impact the responses of individual infected host cells, necessitating single-cell resolution profiling to capture the full phenotypic structure of virus-host interactions. Leitch and McLauchlan sequenced individual Huh7 cells infected with the hepatitis C virus (HCV) to demonstrate the heterogeneity in HCV quasispecies within a cell. Authors showed a cell could range from harboring only wild-type HCV sequences to containing up to four different HCV viral sequences with diverse mutations from one another, with a population of cells containing 32 different HCV sequences [15]. Authors identify differences in fitness of three HCV variants identified, which further highlights the functional impact of these viral quasispecies on the host. More work is necessary to understand the impact of viral quasispecies composition and individual variants on host antiviral responses, which may be particularly important in studying viral escape, and scRNA-seq provides a strong tool to start to answer these questions.
Integrated views of cell signaling and communication in antiviral immunity
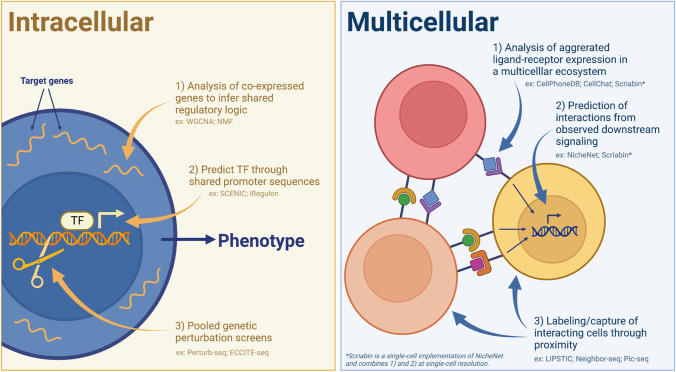
Analysis of intracellular regulatory systems
At its most basic level, scRNA-seq data provides a single transcriptional snapshot of cell state and identity, but it is possible to go beyond this to provide insights into intercellular regulatory logic and signaling (Fig. 5). For example, identifying groups, or modules, of genes that are co-expressed can imply orthogonal biological functionality. One of the most widespread approaches to this analytical question is weighted gene correlation network analysis (WGCNA) [106, 107]. Originally developed for bulk transcriptomic datasets, WGCNA identifies clusters of genes with a high degree of topological overlap, a measure of gene interconnectedness, between samples. WGCNA has recently been adapted to single-cell transcriptomic datasets by discovering modules of highly connected genes between individual cells rather than between separate samples [30, 108]. This approach has recently been applied to longitudinal scRNA-seq profiling of hyperacute HIV infection, where it revealed temporally coordinated and prolonged expression of gene modules associated with NK cell cytolytic activity as potentially associated with future viral control [30, 109]. Another complementary approach to WGCNA involves non-negative matrix factorization (NMF). NMF-based approaches, including consensus NMF (cNMF) [110], have recently been adapted for scRNA-seq data, where they have been applied to disentangle highly interconnected gene programs that define cell state rather than cell type. For example, cNMF has been used to identify a gene program that is associated with bacterial sepsis [111]. This same gene program has now been shown to be strongly associated with severe COVID-19 [18], and is directly inducible in hematopoietic progenitors treated with plasma from severe COVID-19 patients [112].
Fig. 5.
Overview of scRNA-seq analytic approaches to understanding intracellular and multicellular signaling
In addition to implying shared biological functionality, strongly co-expressed gene modules can also reflect the activity of a shared intercellular regulatory network. Many recent methods have sought to leverage genetic and epigenetic data on gene regulatory pathways to infer what transcription factors underlie observed gene modules or DEGs, thereby yielding a more integrated view of cellular phenotype. For example, SCENIC [113] and companion tool iRegulon [114] use promoter and enhancer sequences associated with each gene to predict which motifs and transcription factors are most likely to underlie observed transcriptional profiles. This approach has been used to identify STAT1/2/3 as a key putative driver of neutrophil activation in severe COVID-19 [18]. Alternatively, databases of transcriptional regulation have also been assembled from perturbation experiments, enabling the prediction of transcription factor activity from empirical measurements [115–118]. These databases have been applied to identify the activity of IRF9 in the alveolar epithelial cells of patients with severe COVID-19 [119, 120].
Recent developments in gene editing technology have enabled multiplexed genetic perturbation screens to be combined with deep single-cell transcriptomic readouts, providing approaches to directly uncover gene regulatory networks at a single-cell resolution [121–124]. These methods have recently been applied to uncover several host dependency factors and pathways for SARS-CoV-2, including the NF-κB inhibitor IκBα (NFKBIA) [125] and RAB7A, which prevents sequestration of the ACE2 receptor [126, 127]. In addition to performing functional genetic mapping of host factors, these techniques can be used to simultaneously perturb viral genetic elements. Hein and Weissman [12] have recently used Perturb-seq to map both host and viral factors that are protective or detrimental to Epstein-Barr virus (EBV) infection. They leveraged the single-cell resolution of Perturb-seq to describe a stereotyped trajectory of EBV infection that could be altered by the deletion of viral factors, but slowed or accelerated by the deletion of host factors [12]. These experiments highlight the power of integrating pooled genetic perturbations with single-cell transcriptomic readouts, providing a deep view of host and viral interactions.
Analysis of multicellular ecosystems
In the setting of antiviral immunity, immune cells do not exert their functions in a solitary void but are rather involved in finely balanced communication networks with their microenvironment and other immune cells in order to limit viral disease. By providing a deep view of cellular phenotype at single-cell resolution, scRNA-seq datasets are well suited for the prediction of how individual cells may communicate with each other in a tissue niche (Fig. 5). The curation of ligand-receptor interaction databases has enabled the development and application of many tools to infer patterns of cell–cell communication (CCC) from scRNA-seq data [128, 129].
The most common approach to inferring CCC in scRNA-seq data is to average ligand and receptor expression values for a given cluster or cell type, use these aggregated values to predict which cells are most capable of communicating, and identify which ligand-receptor edges are most specific to communicative pathways between given cell types. These methods include CellPhoneDB [130, 131], CellChat [132], Connectome [133], NATMI [134], SingleCellSignalR [135], and iTALK [136] (reviewed by [137]). These methods have been applied to many scRNA-seq datasets profiling antiviral immune responses. For example, a recent preprint identified SARS-CoV-2-mediated induction of CCL2 in activated interstitial macrophages as a potential mechanism to recruit specific dendritic cell (DC) subtypes through the expression of CCR2 [138].
However, the expression of a cognate ligand-receptor pair by two cells does not demonstrate that those cells are indeed interacting or that the putative interaction impacts downstream cellular phenotype. To address this issue, NicheNet introduced a curated database linking ligand activities to target gene expression and developed a method to infer ligand activity from a set of DEGs [139]. In the setting of COVID-19, NicheNet has been applied to identify persistent IFN-α signaling in NK cells from patients with severe COVID-19 [140], IFN-γ and TNF-α as ligands driving monocyte dysfunction in post-acute sequelae of COVID-19 (PASC) [141], and IL-15 and IL-18 as macrophage-expressed ligands predicted to enhance functional activity of SARS-CoV-2 antigen-reactive CD4 and CD8 T cells [63].
A major limitation of these tools is that they operate at the level of the cell type or cell cluster and thus can obscure biologically-important heterogeneity and specificity. Our lab has demonstrated that CCC analysis methods that aggregate at the level of the cell type or cluster lose > 50% of unique CCC phenotypes in the process of agglomeration, highlighting the importance of maintaining single-cell resolution [142]. Two recent methods, NICHES [143] and Scriabin [142], present techniques to analyze CCC at near single-cell resolution. The fundamental advancement in both of these methods is the encoding of CCC information in a cell–cell matrix that measures the interaction potential of cell–cell pairs along each possible ligand-receptor edge [142, 143]. In applying Scriabin to a longitudinal dataset of SARS-CoV-2 infection [144], Scriabin revealed that uninfected bystander epithelial cells may initiate downstream inflammatory pathways through the production of IL1B which can act on infected cells to upregulate acute-phase reactant encoding genes involved in tissue remodeling processes [142].
Another complementary set of techniques for CCC inference are computational methods that infer which cells are communicating by identifying putative multiplets in the dataset (e.g., Neighbor-seq [145]), or by directly sequencing interacting cells (e.g., PIC-seq [146]). While this provides an additional layer of evidence for biologically-meaningful interactions, cells that have previously interacted but are no longer associated will not be detected. This latter problem has been addressed by techniques such as LIPSTIC [147] that permanently label cells that have interacted using particular ligands or receptors. However, these methods remain poorly scalable and require prior cell engineering. We anticipate that future technological developments will enable the synergy of these complementary approaches towards more comprehensive solutions for CCC analysis.
Multimodal profiling of viral infections
Integrated transcriptomic and genomic single-cell methods
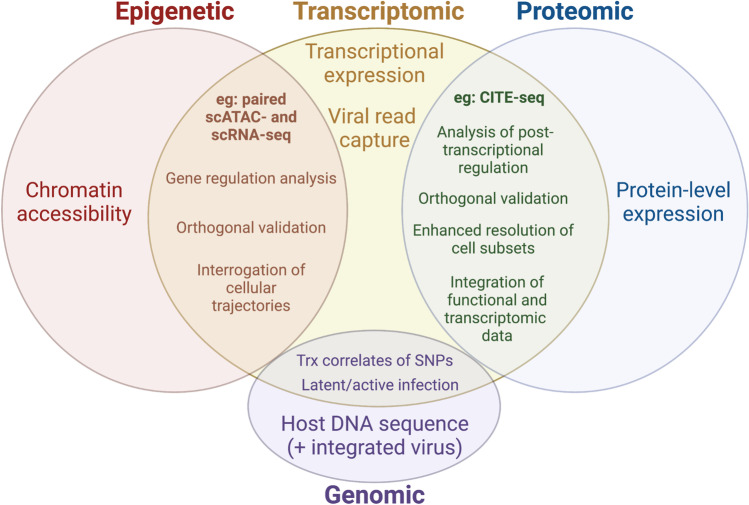
Having genomic information to underlie transcriptomic changes can highlight the role of inherent genetic differences that may drive different antiviral responses and viral susceptibility (Fig. 6). Mutations in host proteins required for viral replication can alter disease susceptibility. For example, individuals homozygous for the CCR5 delta32 allele are resistant to HIV infection [148, 149], while polymorphisms of ACE2, a cellular entry receptor for SARS-CoV-2, may impact ACE2 protein expression and SARS-CoV-2 binding to potentially affecting COVID-19 pathogenesis [150]. Furthermore, associations between the HLA genotype and SARS-CoV-2 susceptibility and disease progression have been reported [151]. To simultaneously study the complexities of genomic variation on transcriptional profiles, G&T-seq (genome and transcriptome sequencing) is a method able to measure both genomic and transcriptomic information from the same cell [152]. After single-cell plating and cell lysis to release mRNA and genomic RNA, polyadenylated mRNA transcripts are physically separated from the DNA with the use of biotinylated oligo-dT primers, and both the RNA and DNA libraries are processed in parallel. Other methods for paired transcriptomic and genomic measurements of single cells include TARGET-seq [153], SIDR [154], and DR-seq [155]. DNA measurements in combination with transcriptomics can also enable an analysis of DNA virus genomic abundance as well as capture any integrated viral reads (e.g., HIV) or detect latent versus lytic viral infection (e.g., HSV1, HSV2) that may drive virus expression dynamics downstream.
Fig. 6.
Venn diagram demonstrating the information that can be derived from epigenetic, transcriptomic, and proteomic measurements. Overlapping regions contain unique information that can be derived from integrative analyses of multiple omics in the same cell. CITE-seq, cellular indexing of transcriptomes and epitopes by sequencing; trx, transcription; scATAC-seq, single-cell resolution in assay for transposase-accessible chromatin using sequencing; SNP, single nucleotide polymorphism
Integrated transcriptomic and proteomic single-cell methods
Technical methods have been developed to integrate scRNA-seq measurements with proteomic measurements of the same cells, such as CITE-seq [94] and REAP-seq [156]. The power of this integrated approach is the ability to correlate transcript abundance with translated protein expression—important for understanding downstream functional responses. Additionally, CITE-seq also empowers the analysis of transcriptomes within cell types defined by surface proteins, which is important as some cell types can be difficult to distinguish on transcriptomes alone (e.g., NK cells versus T cells, intermediate monocytes) (Fig. 6). A study looking at differences between hospitalized patients across moderate, severe, critical, and fatal COVID-19 severities utilized CITE-seq to measure PBMC transcriptomes and paired protein measurements across 188 cell surface proteins [157]. The paired transcriptome-protein analysis empowered the study’s ability to distinguish NK cell subtypes (CD56hiCD16lo NK, CD56dimCD16hi NK, CD56loCD16lo NK) and further identify IL-15-linked fatty metabolism and attenuated inflammation in CD56dimCD16hi NK cells as a primary correlate of disease severity across the study group. While unpaired at the cellular level, CyTOF and scRNA-seq profiling of whole blood from the same COVID-19 patients was able to identify significantly decreased expression of activating receptors DNAM-1 and NKG2D protein in activated NK cells from severe COVID-19 samples compared with healthy, but no changes in levels of their transcripts [18], which provides an example for the importance of multimodal analyses as different conclusions could be had when focusing on one measurement for analyses.
Integrated transcriptomic and epigenomic single-cell methods
Investigating the association between transcriptional and epigenetic variation can elucidate underlying mechanisms and regulatory features that drive immune responses to viral infections (Fig. 6). Previous research has demonstrated that exposure to different environments and vaccines can drive persistent epigenetic changes and downstream cellular responsiveness to infection and viral susceptibility. For example, work from Wimmers et al. [158] demonstrated that an adjuvanted H5N1 influenza vaccine can change the histone modification landscape of monocytes, with changes to chromatin accessibility that associate with increase expression of antiviral- and interferon-related genes and resistance to dengue and Zika virus infection [158]. There are multiple methods that can characterize both the transcriptomics and the epigenomics of a single cell and vary at what epigenomic and transcriptomic layers they can capture. One method, the 10X Chromium Single-Cell Multiome ATAC + Gene Expression kit, works by incubating nuclei suspensions with Transposases to fragment open region fragments and the kit’s Gel Beads include (a) a poly(dT) sequence to build barcoded cDNA libraries from polyadenylated nucleic mRNA as well as (b) a Spacer sequence to attached to transposed DNA fragments for the ATAC library. However, it is important to note that methods using nuclei rather than the whole cell as input into RNA-seq would be unable to detect viral transcripts from viruses that don’t use the nucleus for its life cycle (e.g., flaviviruses, coronaviruses). Another method called scM&T-seq (single-cell methylome and transcription sequencing) [159] utilizes the previously mentioned scG&T method but instead of DNA isolation for genomic sequencing, scBS-seq (single-cell bisulfite sequencing) is applied to isolated DNA to generate methylomes from the same single cells that transcriptomes are generated. A further adaptation to scM&T-seq is called scNMT-seq (single-cell nucleosome, methylation, and transcription sequencing) which adds measurement of chromatin accessibility by adapting Nucleosome Occupancy and Methylation sequencing (NOMe-seq) methods prior to BS-seq [160]. Together, all these methods can begin to bring together a deeper understanding of regulatory mechanisms and cellular trajectories driving immune responses to viral infections and the differential drivers that contribute to protective versus pathogenic responses.
Integration and reanalysis of public scRNA-seq datasets
The expensive nature of generating scRNA-seq datasets in comparison to methods like bulk RNA-seq and flow cytometry tends to limit the number of samples run via scRNA-seq. Therefore, studies can vary in the number of samples profiled and further limits the heterogeneity of experimental conditions and sampled populations that are included. The advent of public repositories to submit scRNA-seq data and the accessibility to these resources, such as NCBI’s Gene Expression Omnibus (for processed data) and the Sequence Read Archive repositories (for raw sequencing data), make it possible for scientists around the world to download and reanalyze collected data. Xu et al. [161] reanalyzed data collected by Zanini et al. [11] of scRNA-seq of PBMCs from dengue-infected patients and employed new tools such as CellChat [132], which was developed in 2021 after the data was published in 2018, to identify cell–cell communication rewiring of PBMCs from severe dengue disease compared to control and mild dengue patients.
Additionally, a comparative understanding of infection across heterogeneity-associated disease manifestations, experimental conditions, and sampling population differences can drive a robust understanding of viral responses across included contexts. Additionally, data integration can boost sample sizes across multiple axes to power analyses. For example, a review by Tian et al. [162] integrated high-quality cells from 21 publically deposited COVID-19 scRNA-seq profiling studies of mainly PBMC and whole blood. In the end, authors collectively analyzed 3.2 million cells from COVID-19 patients from various demographics and disease severities to identify cell type correlates of COVID-19 pathogenesis [162]. Integrated analyses with non-viral diseases are also powerful: Reyes et al. [112] comparatively analyzed scRNA-seq of PBMCs they collected from patients with bacterial sepsis and public COVID-19 PBMC scRNA-seq data to identify shared monocyte transcriptional responses during severe disease, marked by reduced MHC-II transcripts (important for antigen presentation) and increased expression of S100A8 (implicated in the development of myeloid-derived suppressor cells). Further understanding viral infection responses comparatively with other diseases and conditions (including vaccination) is an important avenue for exploration to better understand protective/pathogenic mechanisms.
Conclusions and future directions
scRNA-seq overcomes some of the limits of bulk sequencing methods to measure the heterogeneity of viral dynamics and cellular responses in relation to one another (Fig. 1). With new scRNA-seq methods able to integrate additional measurement technologies, an incredible amount of information can be uncovered from a single sample. Multimodal methods that integrate transcriptomics with other biological information are an important frontier to gaining a deeper understanding of the complexities of human antiviral responses. Additionally, integrated analyses of data, not just within one type of viral infection, but also across various other viral infections, vaccine responses, and conditions (e.g., sepsis) might also add to our understanding of viral pathogenesis through an understanding of the complexities of cellular responses. Public availability of scRNA-seq data paired with constantly developing data analysis tools allows a deeper understanding of viral-host dynamics, thereby allowing these data to continue contributing scientific knowledge far beyond data collection. It remains important to continue scRNA-seq studies across diverse viruses in the context of diverse cell types and populations to gather a holistic understanding of disease pathogenesis. Importantly, all methods discussed have their various pros and cons: when designing an scRNA-seq study uncovering virus-host responses, it is crucial to understand the study’s goals and limitations (e.g., virus type, cell types of interest, sample number, budget) in order to decide on a method that can optimally measure the information of interest. For example, while many studies focus on blood, a number of viruses target specific tissues and may not be present in the blood, thus limiting the power of discussed methods to optimize virus quantification and dynamics. Therefore, utilizing prior information regarding viral tropism and disease dynamics to inform scRNA-seq study and method design can go a long way to obtaining high-information data.
There also remain broad limitations on the use of scRNA-seq technology. For example, the techniques discussed in this review require the sample input to be a single-cell suspension; therefore, no spatial information is captured, as solid tissues must be dissociated prior to analysis. This results in the omission of important information in solid tissue infections (e.g., lung infection in SARS-CoV-2) though is less of an issue for blood-borne pathogens. Newer methods such as spatial barcoding and high-plex RNA imaging seek to remedy this limitation [163, 164]. Additionally, although the throughput of scRNA-seq methods has vastly improved since the advent of these technologies, the maximum cell number that can be used in these workflows is still far below the number of cells that can be processed by single-cell proteomic methods such as flow cytometry and CyTOF. This is due to both technological limitations of scRNA-seq methods as well as cost prohibition, as high-throughput sequencing is quite expensive.
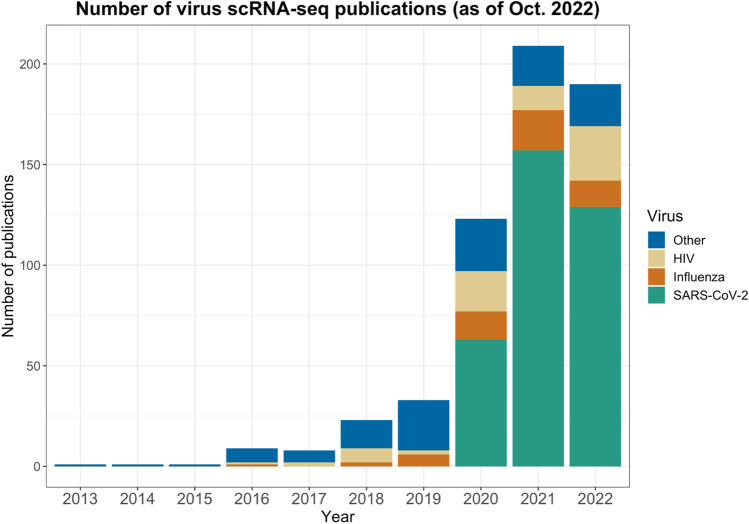
We have learned a lot about antiviral immunity with the advent of scRNA-seq technology—and there is still more to learn. There is a wealth of scRNA-seq publications on SARS-CoV-2 (Fig. 7), and we must extend scRNA-seq applications across other viral diseases, including those identified by WHO as priority diseases [165]. A proactive rather than reactive application of scRNA-seq methods to a broader range of viral infections will allow us to better understand protective and pathogenic cellular responses to viruses to be better equipped to manage current and emerging viral diseases.
Fig. 7.
Virus-related scRNA-seq publications from 2013 to October 2022. SCOPUS database search for publications associated with “scRNA-seq” and “viruses,” with publications relating to HIV, influenza, and SARS-CoV-2 highlighted within the identified total
Acknowledgements
Figures were created with BioRender.com.
Funding
This work was supported by the following fellowships: Bio-X graduate fellowship (K. R. and A. J. W), T32 AI00729037 (M. J. L.), National Science Foundation Graduate Research Fellowship 2019282939 (K. R.), F31 AI172311 (M. J. L.), T32 GM007364 (A. J. W.). Additional support was provided by NIH DP1 DA046089 (C. A. B.) and a 2019 Sentinel Pilot Project from the Bill and Melinda Gates Foundation and OP113682 from the Bill and Melinda Gates Foundation (P. K. and C. A. B.), U19AI109662 (P. K.), U19AI057229 (P. K. and C. A. B.), R01AI125197 (P. K.), Department of Defense contract W81XWH-18–1-0253 and W81XWH1910235 (P. K.), and the Ralph and Marian Falk Medical Research Trust (P. K.). C. A. B. is an Investigator of the Chan Zuckerberg Biohub.
Declarations
Conflict of interest
PK is a shareholder in Inflammatix.
Footnotes
This article is a contribution to the special issue on: Single-cell and spatial multi-omics in clinical outcomes studies - Guest Editor: Brice Gaudillière
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Kalani Ratnasiri, Aaron J. Wilk and Madeline J. Lee have equal contributions
Contributor Information
Purvesh Khatri, Email: pkhatri@stanford.edu.
Catherine A. Blish, Email: cblish@stanford.edu
References
- 1.COVID-19 map. In: Johns Hopkins Coronavirus Resource Center. https://coronavirus.jhu.edu/map.html. Accessed 27 Jul 2022
- 2.de Vries W, Haasnoot J, Fouchier R, et al. Differential RNA silencing suppression activity of NS1 proteins from different influenza A virus strains. J Gen Virol. 2009;90:1916–1922. doi: 10.1099/vir.0.008284-0. [DOI] [PubMed] [Google Scholar]
- 3.Lin L, Liu Y, Tang X, He D. The disease severity and clinical outcomes of the SARS-CoV-2 variants of concern. Front Public Health. 2021;9:775224. doi: 10.3389/fpubh.2021.775224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hayward AC, Fragaszy EB, Bermingham A, et al. Comparative community burden and severity of seasonal and pandemic influenza: results of the Flu Watch cohort study. Lancet Respir Med. 2014;2:445–454. doi: 10.1016/S2213-2600(14)70034-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kawai T, Akira S. Antiviral signaling through pattern recognition receptors. J Biochem. 2007;141:137–145. doi: 10.1093/jb/mvm032. [DOI] [PubMed] [Google Scholar]
- 6.Andres-Terre M, McGuire HM, Pouliot Y, et al. Integrated, multi-cohort analysis identifies conserved transcriptional signatures across multiple respiratory viruses. Immunity. 2015;43:1199–1211. doi: 10.1016/j.immuni.2015.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mick E, Kamm J, Pisco AO, et al. Upper airway gene expression reveals suppressed immune responses to SARS-CoV-2 compared with other respiratory viruses. Nat Commun. 2020;11:5854. doi: 10.1038/s41467-020-19587-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rossetto CC, Tarrant-Elorza M, Verma S, et al. Regulation of viral and cellular gene expression by Kaposi’s sarcoma-associated herpesvirus polyadenylated nuclear RNA. J Virol. 2013;87:5540–5553. doi: 10.1128/JVI.03111-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zheng H, Rao AM, Dermadi D, et al. Multi-cohort analysis of host immune response identifies conserved protective and detrimental modules associated with severity across viruses. Immunity. 2021;54:753–768.e5. doi: 10.1016/j.immuni.2021.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bost P, Giladi A, Liu Y, et al. Host-viral infection maps reveal signatures of severe COVID-19 patients. Cell. 2020;181:1475–1488.e12. doi: 10.1016/j.cell.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zanini F, Robinson ML, Croote D, et al. Virus-inclusive single-cell RNA sequencing reveals the molecular signature of progression to severe dengue. Proc Natl Acad Sci U S A. 2018;115:E12363–E12369. doi: 10.1073/pnas.1813819115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hein MY, Weissman JS. Functional single-cell genomics of human cytomegalovirus infection. Nat Biotechnol. 2021;40:391–401. doi: 10.1038/s41587-021-01059-3. [DOI] [PubMed] [Google Scholar]
- 13.Martínez-Colón GJ, Ratnasiri K, Chen H et al (2022) SARS-CoV-2 infection drives an inflammatory response in human adipose tissue through infection of adipocytes and macrophages. Sci Transl Med eabm9151. 10.1126/scitranslmed.abm9151 [DOI] [PMC free article] [PubMed]
- 14.Russell AB, Elshina E, Kowalsky JR, et al (2019) Single-cell virus sequencing of influenza infections that trigger innate immunity. J Virol 93: 10.1128/JVI.00500-19 [DOI] [PMC free article] [PubMed]
- 15.McWilliam Leitch EC, McLauchlan J. Determining the cellular diversity of hepatitis C virus quasispecies by single-cell viral sequencing. J Virol. 2013;87:12648–12655. doi: 10.1128/JVI.01602-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wu L, Zhang X, Zhao Z, et al. Full-length single-cell RNA-seq applied to a viral human cancer: applications to HPV expression and splicing analysis in HeLa S3 cells. Gigascience. 2015;4:51. doi: 10.1186/s13742-015-0091-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fabozzi G, Oler AJ, Liu P, et al (2018) Strand-specific dual RNA sequencing of bronchial epithelial cells infected with influenza A/H3N2 viruses reveals splicing of gene segment 6 and novel host-virus interactions. J Virol 92: 10.1128/JVI.00518-18 [DOI] [PMC free article] [PubMed]
- 18.Wilk AJ, Lee MJ, Wei B, et al (2021) Multi-omic profiling reveals widespread dysregulation of innate immunity and hematopoiesis in COVID-19. J Exp Med 218: 10.1084/jem.20210582 [DOI] [PMC free article] [PubMed]
- 19.Zanini F, Pu S-Y, Bekerman E, et al (2018) Single-cell transcriptional dynamics of flavivirus infection. Elife 7: 10.7554/eLife.32942 [DOI] [PMC free article] [PubMed]
- 20.Golumbeanu M, Cristinelli S, Rato S, et al. Single-cell RNA-Seq reveals transcriptional heterogeneity in latent and reactivated HIV-infected cells. Cell Rep. 2018;23:942–950. doi: 10.1016/j.celrep.2018.03.102. [DOI] [PubMed] [Google Scholar]
- 21.Shnayder M, Nachshon A, Krishna B, et al (2018) Defining the transcriptional landscape during cytomegalovirus latency with single-cell RNA sequencing. MBio 9: 10.1128/mBio.00013-18 [DOI] [PMC free article] [PubMed]
- 22.O’Neal JT, Upadhyay AA, Wolabaugh A, et al (2019) West Nile virus-inclusive single-cell RNA sequencing reveals heterogeneity in the type I interferon response within single cells. J Virol 93: 10.1128/JVI.01778-18 [DOI] [PMC free article] [PubMed]
- 23.Kotliar D, Lin AE, Logue J, et al. Single-cell profiling of Ebola virus disease in vivo reveals viral and host dynamics. Cell. 2020;183:1383–1401.e19. doi: 10.1016/j.cell.2020.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wyler E, Franke V, Menegatti J, et al. Single-cell RNA-sequencing of herpes simplex virus 1-infected cells connects NRF2 activation to an antiviral program. Nat Commun. 2019;10:4878. doi: 10.1038/s41467-019-12894-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hu H-L, Srinivas KP, Wang S, et al. Single-cell transcriptomics identifies Gadd45b as a regulator of herpesvirus-reactivating neurons. EMBO Rep. 2022;23:e53543. doi: 10.15252/embr.202153543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Saikia M, Burnham P, Keshavjee SH, et al. Simultaneous multiplexed amplicon sequencing and transcriptome profiling in single cells. Nat Methods. 2019;16:59–62. doi: 10.1038/s41592-018-0259-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sanborn MA, Li T, Victor K, et al. Analysis of cell-associated DENV RNA by oligo(dT) primed 5’ capture scRNAseq. Sci Rep. 2020;10:9047. doi: 10.1038/s41598-020-65939-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gao KM, Derr AG, Guo Z, et al (2021) Human nasal wash RNA-Seq reveals distinct cell-specific innate immune responses in influenza versus SARS-CoV-2. JCI Insight 6: 10.1172/jci.insight.152288 [DOI] [PMC free article] [PubMed]
- 29.Steuerman Y, Cohen M, Peshes-Yaloz N, et al. Dissection of influenza infection in vivo by single-cell RNA sequencing. Cell Syst. 2018;6:679–691.e4. doi: 10.1016/j.cels.2018.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kazer SW, Aicher TP, Muema DM, et al. Integrated single-cell analysis of multicellular immune dynamics during hyperacute HIV-1 infection. Nat Med. 2020;26:511–518. doi: 10.1038/s41591-020-0799-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wilk AJ, Rustagi A, Zhao NQ, et al. A single-cell atlas of the peripheral immune response in patients with severe COVID-19. Nat Med. 2020;26:1070–1076. doi: 10.1038/s41591-020-0944-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bost P, De Sanctis F, Canè S, et al. Deciphering the state of immune silence in fatal COVID-19 patients. Nat Commun. 2021;12:1428. doi: 10.1038/s41467-021-21702-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Delorey TM, Ziegler CGK, Heimberg G, et al. COVID-19 tissue atlases reveal SARS-CoV-2 pathology and cellular targets. Nature. 2021;595:107–113. doi: 10.1038/s41586-021-03570-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bekerman E, Einav S. Infectious disease. Combating emerging viral threats Science. 2015;348:282–283. doi: 10.1126/science.aaa3778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhou W-M, Yan Y-Y, Guo Q-R, et al. Microfluidics applications for high-throughput single cell sequencing. J Nanobiotechnology. 2021;19:312. doi: 10.1186/s12951-021-01045-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Macosko EZ, Basu A, Satija R, et al. Highly parallel genome-wide expression profiling of individual cells using nanoliter droplets. Cell. 2015;161:1202–1214. doi: 10.1016/j.cell.2015.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zilionis R, Nainys J, Veres A, et al. Single-cell barcoding and sequencing using droplet microfluidics. Nat Protoc. 2017;12:44–73. doi: 10.1038/nprot.2016.154. [DOI] [PubMed] [Google Scholar]
- 38.Ekpenyong AE, Toepfner N, Chilvers ER, Guck J. Mechanotransduction in neutrophil activation and deactivation. Biochim Biophys Acta. 2015;1853:3105–3116. doi: 10.1016/j.bbamcr.2015.07.015. [DOI] [PubMed] [Google Scholar]
- 39.Yap B, Kamm RD. Mechanical deformation of neutrophils into narrow channels induces pseudopod projection and changes in biomechanical properties. J Appl Physiol. 2005;98:1930–1939. doi: 10.1152/japplphysiol.01226.2004. [DOI] [PubMed] [Google Scholar]
- 40.Can I process neutrophils (or other granulocytes) using 10x Single Cell applications? In: 10X Genomics. https://kb.10xgenomics.com/hc/en-us/articles/360004024032-Can-I-process-neutrophils-or-other-granulocytes-using-10x-Single-Cell-applications-. Accessed 11 Oct 2022
- 41.Capturing Neutrophils in 10x Single Cell Gene Expression Data -Software -Single Cell Gene Expression -Official 10x Genomics Support. https://support.10xgenomics.com/single-cell-gene-expression/software/pipelines/latest/tutorials/neutrophils. Accessed 11 Oct 2022
- 42.Wigerblad G, Cao Q, Brooks S, et al (2022) Single-cell analysis reveals the range of transcriptional states of circulating human neutrophils. bioRxiv 2022.02.22.481522 [DOI] [PMC free article] [PubMed]
- 43.Zhang X, Li T, Liu F, et al. Comparative analysis of droplet-based ultra-high-throughput single-Cell RNA-Seq systems. Mol Cell. 2019;73:130–142.e5. doi: 10.1016/j.molcel.2018.10.020. [DOI] [PubMed] [Google Scholar]
- 44.Saichi M, Ladjemi MZ, Korniotis S, et al. Single-cell RNA sequencing of blood antigen-presenting cells in severe COVID-19 reveals multi-process defects in antiviral immunity. Nat Cell Biol. 2021;23:538–551. doi: 10.1038/s41556-021-00681-2. [DOI] [PubMed] [Google Scholar]
- 45.Schulte-Schrepping J, Reusch N, Paclik D, et al. Severe COVID-19 is marked by a dysregulated myeloid cell compartment. Cell. 2020 doi: 10.1016/j.cell.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Garcia-Flores V, Romero R, Xu Y, et al. Maternal-fetal immune responses in pregnant women infected with SARS-CoV-2. Nat Commun. 2022;13:320. doi: 10.1038/s41467-021-27745-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Pique-Regi R, Romero R, Tarca AL, et al (2020) Does the human placenta express the canonical cell entry mediators for SARS-CoV-2? Elife 9: 10.7554/eLife.58716 [DOI] [PMC free article] [PubMed]
- 48.Lu-Culligan A, Chavan AR, Vijayakumar P, et al. Maternal respiratory SARS-CoV-2 infection in pregnancy is associated with a robust inflammatory response at the maternal-fetal interface. Med (N Y) 2021;2:591–610.e10. doi: 10.1016/j.medj.2021.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gierahn TM, Wadsworth MH, 2nd, Hughes TK, et al. Seq-Well: portable, low-cost RNA sequencing of single cells at high throughput. Nat Methods. 2017;14:395–398. doi: 10.1038/nmeth.4179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hughes TK, Wadsworth MH, Gierahn TM, et al. Second-strand synthesis-based massively parallel scRNA-Seq reveals cellular states and molecular features of human inflammatory skin pathologies. Immunity. 2020;53:878–894.e7. doi: 10.1016/j.immuni.2020.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fan HC, Fu GK, Fodor SPA. Expression profiling. Combinatorial labeling of single cells for gene expression cytometry. Science. 2015;347:1258367. doi: 10.1126/science.1258367. [DOI] [PubMed] [Google Scholar]
- 52.Shum EY, Walczak EM, Chang C, Christina Fan H (2019) Quantitation of mRNA transcripts and proteins using the BD RhapsodyTM single-cell analysis system. In: Suzuki Y (ed) Single Molecule and Single Cell Sequencing. Springer Singapore, Singapore, pp 63–79
- 53.Goetz JJ, Trimarchi JM. Transcriptome sequencing of single cells with Smart-Seq. Nat Biotechnol. 2012;30:763–765. doi: 10.1038/nbt.2325. [DOI] [PubMed] [Google Scholar]
- 54.Jaitin DA, Kenigsberg E, Keren-Shaul H, et al. Massively parallel single-cell RNA-seq for marker-free decomposition of tissues into cell types. Science. 2014;343:776–779. doi: 10.1126/science.1247651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Picelli S, Björklund ÅK, Faridani OR, et al. Smart-seq2 for sensitive full-length transcriptome profiling in single cells. Nat Methods. 2013;10:1096–1098. doi: 10.1038/nmeth.2639. [DOI] [PubMed] [Google Scholar]
- 56.Hagemann-Jensen M, Ziegenhain C, Chen P, et al. Single-cell RNA counting at allele and isoform resolution using Smart-seq3. Nat Biotechnol. 2020;38:708–714. doi: 10.1038/s41587-020-0497-0. [DOI] [PubMed] [Google Scholar]
- 57.Rosenberg AB, Roco CM, Muscat RA, et al. Single-cell profiling of the developing mouse brain and spinal cord with split-pool barcoding. Science. 2018;360:176–182. doi: 10.1126/science.aam8999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Singh M, Al-Eryani G, Carswell S, et al. High-throughput targeted long-read single cell sequencing reveals the clonal and transcriptional landscape of lymphocytes. Nat Commun. 2019;10:3120. doi: 10.1038/s41467-019-11049-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Tu AA, Gierahn TM, Monian B, et al. TCR sequencing paired with massively parallel 3′ RNA-seq reveals clonotypic T cell signatures. Nat Immunol. 2019;20:1692–1699. doi: 10.1038/s41590-019-0544-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Luo L, Liang W, Pang J, et al. Dynamics of TCR repertoire and T cell function in COVID-19 convalescent individuals. Cell Discov. 2021;7:89. doi: 10.1038/s41421-021-00321-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Mathew NR, Jayanthan JK, Smirnov IV, et al. Single-cell BCR and transcriptome analysis after influenza infection reveals spatiotemporal dynamics of antigen-specific B cells. Cell Rep. 2021;35:109286. doi: 10.1016/j.celrep.2021.109286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gao S, Wu Z, Arnold B, et al. Single-cell RNA sequencing coupled to TCR profiling of large granular lymphocyte leukemia T cells. Nat Commun. 2022;13:1982. doi: 10.1038/s41467-022-29175-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Fischer DS, Ansari M, Wagner KI, et al. Single-cell RNA sequencing reveals ex vivo signatures of SARS-CoV-2-reactive T cells through “reverse phenotyping”. Nat Commun. 2021;12:1–14. doi: 10.1038/s41467-021-24730-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Yang H-Q, Wang Y-S, Zhai K, Tong Z-H. Single-cell TCR sequencing reveals the dynamics of T cell repertoire profiling during pneumocystis infection. Front Microbiol. 2021;12:637500. doi: 10.3389/fmicb.2021.637500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Huang H, Sikora MJ, Islam S, et al. Select sequencing of clonally expanded CD8+ T cells reveals limits to clonal expansion. Proc Natl Acad Sci U S A. 2019;116:8995–9001. doi: 10.1073/pnas.1902649116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Setliff I, Shiakolas AR, Pilewski KA, et al. High-throughput mapping of B cell receptor sequences to antigen specificity. Cell. 2019;179:1636–1646.e15. doi: 10.1016/j.cell.2019.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Shiakolas AR, Kramer KJ, Johnson NV, et al. Efficient discovery of SARS-CoV-2-neutralizing antibodies via B cell receptor sequencing and ligand blocking. Nat Biotechnol. 2022 doi: 10.1038/s41587-022-01232-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Svoboda M, Frost HR, Bosco G. Internal oligo(dT) priming introduces systematic bias in bulk and single-cell RNA sequencing count data. NAR Genom Bioinform. 2022;4:lqac035. doi: 10.1093/nargab/lqac035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ziegler CGK, Allon SJ, Nyquist SK, et al. SARS-CoV-2 receptor ACE2 is an interferon-stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell. 2020;181:1016–1035.e19. doi: 10.1016/j.cell.2020.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Martínez-Colón GJ, Ratnasiri K, Chen H, et al (2021) SARS-CoV-2 infects human adipose tissue and elicits an inflammatory response consistent with severe COVID-19. bioRxiv 2021.10.24.465626
- 71.Salmen F, De Jonghe J, Kaminski TS, et al. High-throughput total RNA sequencing in single cells using VASA-seq. Nat Biotechnol. 2022 doi: 10.1038/s41587-022-01361-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Fan X, Zhang X, Wu X, et al. Single-cell RNA-seq transcriptome analysis of linear and circular RNAs in mouse preimplantation embryos. Genome Biol. 2015;16:148. doi: 10.1186/s13059-015-0706-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sheng K, Cao W, Niu Y, et al. Effective detection of variation in single-cell transcriptomes using MATQ-seq. Nat Methods. 2017;14:267–270. doi: 10.1038/nmeth.4145. [DOI] [PubMed] [Google Scholar]
- 74.Preston BD, Poiesz BJ, Loeb LA. Fidelity of HIV-1 reverse transcriptase. Science. 1988;242:1168–1171. doi: 10.1126/science.2460924. [DOI] [PubMed] [Google Scholar]
- 75.Campillo-Balderas JA, Lazcano A, Becerra A (2015) Viral genome size distribution does not correlate with the antiquity of the host lineages. Front Ecol Evol 3: 10.3389/fevo.2015.00143
- 76.Chaitanya KV (2019) Structure and organization of virus genomes. In: Chaitanya KV (ed) Genome and genomics: from archaea to eukaryotes. Springer Singapore, Singapore, pp 1–30
- 77.Lebrigand K, Magnone V, Barbry P, Waldmann R. High throughput error corrected nanopore single cell transcriptome sequencing. Nat Commun. 2020;11:4025. doi: 10.1038/s41467-020-17800-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Stassen SV, Siu DMD, Lee KCM, et al. PARC: ultrafast and accurate clustering of phenotypic data of millions of single cells. Bioinformatics. 2020;36:2778–2786. doi: 10.1093/bioinformatics/btaa042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yu L, Cao Y, Yang JYH, Yang P. Benchmarking clustering algorithms on estimating the number of cell types from single-cell RNA-sequencing data. Genome Biol. 2022;23:49. doi: 10.1186/s13059-022-02622-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Krzak M, Raykov Y, Boukouvalas A, et al. Benchmark and parameter sensitivity analysis of single-cell RNA sequencing clustering methods. Front Genet. 2019;10:1253. doi: 10.3389/fgene.2019.01253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Duò A, Robinson MD, Soneson C. A systematic performance evaluation of clustering methods for single-cell RNA-seq data. F1000Res. 2018;7:1141. doi: 10.12688/f1000research.15666.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Travaglini KJ, Nabhan AN, Penland L, et al. A molecular cell atlas of the human lung from single-cell RNA sequencing. Nature. 2020;587:619–625. doi: 10.1038/s41586-020-2922-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Pasquini G, Rojo Arias JE, Schäfer P, Busskamp V. Automated methods for cell type annotation on scRNA-seq data. Comput Struct Biotechnol J. 2021;19:961–969. doi: 10.1016/j.csbj.2021.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kiselev VY, Yiu A, Hemberg M. scmap: projection of single-cell RNA-seq data across data sets. Nat Methods. 2018;15:359–362. doi: 10.1038/nmeth.4644. [DOI] [PubMed] [Google Scholar]
- 85.Aran D, Looney AP, Liu L, et al. Reference-based analysis of lung single-cell sequencing reveals a transitional profibrotic macrophage. Nat Immunol. 2019;20:163–172. doi: 10.1038/s41590-018-0276-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hou R, Denisenko E, Forrest ARR. scMatch: a single-cell gene expression profile annotation tool using reference datasets. Bioinformatics. 2019;35:4688–4695. doi: 10.1093/bioinformatics/btz292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hao Y, Hao S, Andersen-Nissen E, et al. Integrated analysis of multimodal single-cell data. Cell. 2021 doi: 10.1016/j.cell.2021.04.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lotfollahi M, Naghipourfar M, Luecken MD, et al. Mapping single-cell data to reference atlases by transfer learning. Nat Biotechnol. 2021;40:121–130. doi: 10.1038/s41587-021-01001-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Tan Y, Cahan P. SingleCellNet: a computational tool to classify single cell RNA-Seq data across platforms and across species. Cell Syst. 2019;9:207–213.e2. doi: 10.1016/j.cels.2019.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Lin Y, Cao Y, Kim HJ, et al. scClassify: sample size estimation and multiscale classification of cells using single and multiple reference. Mol Syst Biol. 2020;16:e9389. doi: 10.15252/msb.20199389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Johnson TS, Wang T, Huang Z, et al. LAmbDA: label ambiguous domain adaptation dataset integration reduces batch effects and improves subtype detection. Bioinformatics. 2019;35:4696–4706. doi: 10.1093/bioinformatics/btz295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Alquicira-Hernandez J, Sathe A, Ji HP, et al. scPred: accurate supervised method for cell-type classification from single-cell RNA-seq data. Genome Biol. 2019;20:264. doi: 10.1186/s13059-019-1862-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Schmidt F, Ranjan B, Lin QXX, et al. RCA2: a scalable supervised clustering algorithm that reduces batch effects in scRNA-seq data. Nucleic Acids Res. 2021;49:8505–8519. doi: 10.1093/nar/gkab632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Stoeckius M, Hafemeister C, Stephenson W, et al. Simultaneous epitope and transcriptome measurement in single cells. Nat Methods. 2017;14:865–868. doi: 10.1038/nmeth.4380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Bergen V, Lange M, Peidli S, et al. Generalizing RNA velocity to transient cell states through dynamical modeling. Nat Biotechnol. 2020;38:1408–1414. doi: 10.1038/s41587-020-0591-3. [DOI] [PubMed] [Google Scholar]
- 96.La Manno G, Soldatov R, Zeisel A, et al. RNA velocity of single cells. Nature. 2018;560:494–498. doi: 10.1038/s41586-018-0414-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Zheng SC, Stein-O’Brien G, Boukas L, et al (2022) Pumping the brakes on RNA velocity – understanding and interpreting RNA velocity estimates. bioRxiv 2022.06.19.494717 [DOI] [PMC free article] [PubMed]
- 98.Atta L, Sahoo A, Fan J. VeloViz: RNA velocity informed embeddings for visualizing cellular trajectories. Bioinformatics. 2021 doi: 10.1093/bioinformatics/btab653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wang X, Wen Y, Xie X, et al. Dysregulated hematopoiesis in bone marrow marks severe COVID-19. Cell Discov. 2021;7:60. doi: 10.1038/s41421-021-00296-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Erhard F, Baptista MAP, Krammer T, et al. scSLAM-seq reveals core features of transcription dynamics in single cells. Nature. 2019;571:419–423. doi: 10.1038/s41586-019-1369-y. [DOI] [PubMed] [Google Scholar]
- 101.Qiu X, Zhang Y, Martin-Rufino JD, et al. Mapping transcriptomic vector fields of single cells. Cell. 2022;185:690–711.e45. doi: 10.1016/j.cell.2021.12.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Stano M, Beke G, Klucar L (2016) viruSITE-integrated database for viral genomics. Database 2016: 10.1093/database/baw162 [DOI] [PMC free article] [PubMed]
- 103.Sun J, Vera JC, Drnevich J, et al. Single cell heterogeneity in influenza A virus gene expression shapes the innate antiviral response to infection. PLoS Pathog. 2020;16:e1008671. doi: 10.1371/journal.ppat.1008671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Young MD, Behjati S (2020) SoupX removes ambient RNA contamination from droplet-based single-cell RNA sequencing data. Gigascience 9.: 10.1093/gigascience/giaa151 [DOI] [PMC free article] [PubMed]
- 105.Cohen P, DeGrace EJ, Danziger O, et al (2021) Unambiguous detection of SARS-CoV-2 subgenomic mRNAs with single cell RNA sequencing. bioRxiv. 10.1101/2021.11.22.469642 [DOI] [PMC free article] [PubMed]
- 106.Langfelder P, Horvath S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics. 2008;9:559. doi: 10.1186/1471-2105-9-559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Yip AM, Horvath S. Gene network interconnectedness and the generalized topological overlap measure. BMC Bioinformatics. 2007;8:22. doi: 10.1186/1471-2105-8-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Morabito S, Miyoshi E, Michael N, et al. Single-nucleus chromatin accessibility and transcriptomic characterization of Alzheimer’s disease. Nat Genet. 2021;53:1143–1155. doi: 10.1038/s41588-021-00894-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Kazer SW, Walker BD, Shalek AK. Evolution and diversity of immune responses during acute HIV infection. Immunity. 2020;53:908–924. doi: 10.1016/j.immuni.2020.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Kotliar D, Veres A, Nagy MA, et al. Identifying gene expression programs of cell-type identity and cellular activity with single-cell RNA-Seq. Elife. 2019;8:e43803. doi: 10.7554/eLife.43803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Reyes M, Filbin MR, Bhattacharyya RP, et al. An immune-cell signature of bacterial sepsis. Nat Med. 2020;26:333–340. doi: 10.1038/s41591-020-0752-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Reyes M, Filbin MR, Bhattacharyya RP, et al. Plasma from patients with bacterial sepsis or severe COVID-19 induces suppressive myeloid cell production from hematopoietic progenitors in vitro. Sci Transl Med. 2021;13:eabe9599. doi: 10.1126/scitranslmed.abe9599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Aibar S, González-Blas CB, Moerman T, et al. SCENIC: single-cell regulatory network inference and clustering. Nat Methods. 2017;14:1083–1086. doi: 10.1038/nmeth.4463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Janky RS, Verfaillie A, Imrichová H, et al. iRegulon: from a gene list to a gene regulatory network using large motif and track collections. PLoS Comput Biol. 2014;10:e1003731. doi: 10.1371/journal.pcbi.1003731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Garcia-Alonso L, Holland CH, Ibrahim MM, et al. Benchmark and integration of resources for the estimation of human transcription factor activities. Genome Res. 2019;29:1363–1375. doi: 10.1101/gr.240663.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Türei D, Korcsmáros T, Saez-Rodriguez J. OmniPath: guidelines and gateway for literature-curated signaling pathway resources. Nat Methods. 2016;13:966–967. doi: 10.1038/nmeth.4077. [DOI] [PubMed] [Google Scholar]
- 117.Türei D, Valdeolivas A, Gul L, et al. Integrated intra- and intercellular signaling knowledge for multicellular omics analysis. Mol Syst Biol. 2021;17:e9923. doi: 10.15252/msb.20209923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Ceccarelli F, Turei D, Gabor A, Saez-Rodriguez J. Bringing data from curated pathway resources to Cytoscape with OmniPath. Bioinformatics. 2020;36:2632–2633. doi: 10.1093/bioinformatics/btz968. [DOI] [PubMed] [Google Scholar]
- 119.Vázquez-Jiménez A, León UEA-PD, Matadamas-Guzman M et al (2021) On deep landscape exploration of COVID-19 patients cells and severity markers. Front Immunol 12:705646. 10.3389/fimmu.2021.705646 [DOI] [PMC free article] [PubMed]
- 120.Liao M, Liu Y, Yuan J, et al. Single-cell landscape of bronchoalveolar immune cells in patients with COVID-19. Nat Med. 2020;26:842–844. doi: 10.1038/s41591-020-0901-9. [DOI] [PubMed] [Google Scholar]
- 121.Adamson B, Norman TM, Jost M, et al. A multiplexed single-cell CRISPR screening platform enables systematic dissection of the unfolded protein response. Cell. 2016;167:1867–1882.e21. doi: 10.1016/j.cell.2016.11.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Dixit A, Parnas O, Li B, et al. Perturb-Seq: dissecting molecular circuits with scalable single-cell RNA profiling of pooled genetic screens. Cell. 2016;167:1853–1866.e17. doi: 10.1016/j.cell.2016.11.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Jaitin DA, Weiner A, Yofe I, et al. Dissecting immune circuits by linking CRISPR-pooled screens with single-cell RNA-Seq. Cell. 2016;167:1883–1896.e15. doi: 10.1016/j.cell.2016.11.039. [DOI] [PubMed] [Google Scholar]
- 124.Replogle JM, Saunders RA, Pogson AN, et al. Mapping information-rich genotype-phenotype landscapes with genome-scale Perturb-seq. Cell. 2022;185:2559–2575.e28. doi: 10.1016/j.cell.2022.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Sunshine S, Puschnik AS, Replogle JM, et al (2022) Systematic functional interrogation of SARS-CoV-2 host factors using Perturb-seq. bioRxiv 2022.07.15.500120 [DOI] [PMC free article] [PubMed]
- 126.Mimitou EP, Cheng A, Montalbano A, et al. Multiplexed detection of proteins, transcriptomes, clonotypes and CRISPR perturbations in single cells. Nat Methods. 2019;16:409–412. doi: 10.1038/s41592-019-0392-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Daniloski Z, Jordan TX, Wessels H-H, et al. Identification of required host factors for SARS-CoV-2 infection in human cells. Cell. 2021;184:92–105.e16. doi: 10.1016/j.cell.2020.10.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ramilowski JA, Goldberg T, Harshbarger J, et al. A draft network of ligand–receptor-mediated multicellular signalling in human. Nat Commun. 2015;6:7866. doi: 10.1038/ncomms8866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Dimitrov D, Türei D, Garrido-Rodriguez M, et al. Comparison of methods and resources for cell-cell communication inference from single-cell RNA-Seq data. Nat Commun. 2022;13:1–13. doi: 10.1038/s41467-022-30755-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Efremova M, Vento-Tormo M, Teichmann SA, Vento-Tormo R. Cell PhoneDB: inferring cell–cell communication from combined expression of multi-subunit ligand–receptor complexes. Nat Protoc. 2020;15:1484–1506. doi: 10.1038/s41596-020-0292-x. [DOI] [PubMed] [Google Scholar]
- 131.Vento-Tormo R, Efremova M, Botting RA, et al. Single-cell reconstruction of the early maternal-fetal interface in humans. Nature. 2018;563:347–353. doi: 10.1038/s41586-018-0698-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Jin S, Guerrero-Juarez CF, Zhang L, et al. Inference and analysis of cell-cell communication using Cell Chat. Nat Commun. 2021;12:1–20. doi: 10.1038/s41467-021-21246-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Raredon MSB, Yang J, Garritano J, et al (2021) Connectome: computation and visualization of cell-cell signaling topologies in single-cell systems data. bioRxiv 2021.01.21.427529 [DOI] [PMC free article] [PubMed]
- 134.Hou R, Denisenko E, Ong HT, et al. Predicting cell-to-cell communication networks using NATMI. Nat Commun. 2020;11:5011. doi: 10.1038/s41467-020-18873-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Cabello-Aguilar S, Alame M, Kon-Sun-Tack F, et al. SingleCellSignalR: inference of intercellular networks from single-cell transcriptomics. Nucleic Acids Res. 2020;48:e55. doi: 10.1093/nar/gkaa183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Wang Y, Wang R, Zhang S et al (2019) iTALK: an R package to characterize and illustrate intercellular communication. Biorxiv 507871. 10.1101/507871
- 137.Armingol E, Officer A, Harismendy O, Lewis NE. Deciphering cell–cell interactions and communication from gene expression. Nat Rev Genet. 2020;22:71–88. doi: 10.1038/s41576-020-00292-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Wu TT-H, Travaglini KJ, Rustagi A, et al (2022) Activated interstitial macrophages are a predominant target of viral takeover and focus of inflammation in COVID-19 initiation in human lung. bioRxiv 2022.05.10.491266
- 139.Browaeys R, Saelens W, Saeys Y. NicheNet: modeling intercellular communication by linking ligands to target genes. Nat Methods. 2020;17:159–162. doi: 10.1038/s41592-019-0667-5. [DOI] [PubMed] [Google Scholar]
- 140.Krämer B, Knoll R, Bonaguro L, et al. Early IFN-α signatures and persistent dysfunction are distinguishing features of NK cells in severe COVID-19. Immunity. 2021;54:2650–2669.e14. doi: 10.1016/j.immuni.2021.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Talla A, Vasaikar SV, Lemos MP, et al (2021) Longitudinal immune dynamics of mild COVID-19 define signatures of recovery and persistence. bioRxiv 2021.05.26.442666
- 142.Wilk AJ, Shalek AK, Holmes S, Blish CA (2022) Comparative analysis of cell-cell communication at single-cell resolution. bioRxiv 2022.02.04.479209 [DOI] [PMC free article] [PubMed]
- 143.Raredon MSB, Yang J, Kothapalli N, et al (2022) Comprehensive visualization of cell-cell interactions in single-cell and spatial transcriptomics with NICHES. bioRxiv 2022.01.23.477401 [DOI] [PMC free article] [PubMed]
- 144.Ravindra NG, Alfajaro MM, Gasque V, et al. Single-cell longitudinal analysis of SARS-CoV-2 infection in human airway epithelium identifies target cells, alterations in gene expression, and cell state changes. PLoS Biol. 2021;19:e3001143. doi: 10.1371/journal.pbio.3001143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Ghaddar B, De S (2022) Reconstructing physical cell interaction networks from single-cell data using neighbor-seq. Nucleic Acids Res 50:e82–e82. 10.1093/nar/gkac333 [DOI] [PMC free article] [PubMed]
- 146.Giladi A, Cohen M, Medaglia C, et al. Dissecting cellular crosstalk by sequencing physically interacting cells. Nat Biotechnol. 2020;38:629–637. doi: 10.1038/s41587-020-0442-2. [DOI] [PubMed] [Google Scholar]
- 147.Pasqual G, Chudnovskiy A, Tas JMJ, et al. Monitoring T cell-dendritic cell interactions in vivo by intercellular enzymatic labelling. Nature. 2018;553:496–500. doi: 10.1038/nature25442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Hütter G, Nowak D, Mossner M, et al. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. N Engl J Med. 2009;360:692–698. doi: 10.1056/NEJMoa0802905. [DOI] [PubMed] [Google Scholar]
- 149.Liu R, Paxton WA, Choe S, et al. Homozygous defect in HIV-1 coreceptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection. Cell. 1996;86:367–377. doi: 10.1016/S0092-8674(00)80110-5. [DOI] [PubMed] [Google Scholar]
- 150.Hou Y, Zhao J, Martin W, et al. New insights into genetic susceptibility of COVID-19: an ACE2 and TMPRSS2 polymorphism analysis. BMC Med. 2020;18:216. doi: 10.1186/s12916-020-01673-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Toyoshima Y, Nemoto K, Matsumoto S, et al. SARS-CoV-2 genomic variations associated with mortality rate of COVID-19. J Hum Genet. 2020;65:1075–1082. doi: 10.1038/s10038-020-0808-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Macaulay IC, Haerty W, Kumar P, et al. G&T-seq: parallel sequencing of single-cell genomes and transcriptomes. Nat Methods. 2015;12:519–522. doi: 10.1038/nmeth.3370. [DOI] [PubMed] [Google Scholar]
- 153.Rodriguez-Meira A, O’Sullivan J, Rahman H, Mead AJ. TARGET-Seq: a protocol for high-sensitivity single-cell mutational analysis and parallel RNA sequencing. STAR Protoc. 2020;1:100125. doi: 10.1016/j.xpro.2020.100125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Han KY, Kim K-T, Joung J-G, et al. SIDR: simultaneous isolation and parallel sequencing of genomic DNA and total RNA from single cells. Genome Res. 2018;28:75–87. doi: 10.1101/gr.223263.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Dey SS, Kester L, Spanjaard B, et al. Integrated genome and transcriptome sequencing of the same cell. Nat Biotechnol. 2015;33:285–289. doi: 10.1038/nbt.3129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Peterson VM, Zhang KX, Kumar N, et al. Multiplexed quantification of proteins and transcripts in single cells. Nat Biotechnol. 2017;35:936–939. doi: 10.1038/nbt.3973. [DOI] [PubMed] [Google Scholar]
- 157.Liu C, Martins AJ, Lau WW, et al. Time-resolved systems immunology reveals a late juncture linked to fatal COVID-19. Cell. 2021;184:1836–1857.e22. doi: 10.1016/j.cell.2021.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Wimmers F, Donato M, Kuo A, et al. The single-cell epigenomic and transcriptional landscape of immunity to influenza vaccination. Cell. 2021;184:3915–3935.e21. doi: 10.1016/j.cell.2021.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Angermueller C, Clark SJ, Lee HJ, et al. Parallel single-cell sequencing links transcriptional and epigenetic heterogeneity. Nat Methods. 2016;13:229–232. doi: 10.1038/nmeth.3728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Clark SJ, Argelaguet R, Kapourani C-A, et al. scNMT-seq enables joint profiling of chromatin accessibility DNA methylation and transcription in single cells. Nat Commun. 2018;9:781. doi: 10.1038/s41467-018-03149-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Xu G, Gao Y, Pan T, et al. Dynamic immune ecosystem of dengue infection revealed by single-cell sequencing. J Leukoc Biol. 2022 doi: 10.1002/JLB.6MA0622-738RR. [DOI] [PubMed] [Google Scholar]
- 162.Tian Y, Carpp LN, Miller HER, et al. Single-cell immunology of SARS-CoV-2 infection. Nat Biotechnol. 2022;40:30–41. doi: 10.1038/s41587-021-01131-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Longo SK, Guo MG, Ji AL, Khavari PA. Integrating single-cell and spatial transcriptomics to elucidate intercellular tissue dynamics. Nat Rev Genet. 2021;22:627–644. doi: 10.1038/s41576-021-00370-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Marx V. Method of the year: spatially resolved transcriptomics. Nat Methods. 2021;18:9–14. doi: 10.1038/s41592-020-01033-y. [DOI] [PubMed] [Google Scholar]
- 165.Prioritizing diseases for research and development in emergency contexts. https://www.who.int/activities/prioritizing-diseases-for-research-and-development-in-emergency-contexts. Accessed 10 Aug 2022