Abstract
Simple Summary
Osteoarthritis (OA) is a common and disabling condition affecting half billion people worldwide. To date, management has been only palliative and regenerative approaches based on mesenchymal stromal cells (MSCs) have therefore gained interest in the scientific and medical communities. One of the main obstacles for their translation into everyday practice is the lack of a thorough characterization of these biological products in the context of the disease. In this study, we aimed at dissecting the molecular signals released by MSCs in an environment resembling OA joints. The main findings are that MSCs in contact with synovial fluid of OA patients are able to release factors with anti-inflammatory and regenerative properties. These results will lay the foundations for a wider use of MSCs based products for regenerative medicine and hopefully slow down or halt progression of the disease in patients that, when pharmacological, biologic or surgical treatments fail, have prosthesis implant as the elective therapeutic option.
Abstract
Bone marrow-derived mesenchymal stromal cells (BMSCs)-based therapies show a great potential to manage inflammation and tissue degeneration in osteoarthritis (OA) patients. Clinical trials showed the ability to manage pain and activation of immune cells and allowed restoration of damaged cartilage. To date, a molecular fingerprint of BMSC-secreted molecules in OA joint conditions able to support clinical outcomes is missing; the lack of that molecular bridge between BMSC activity and clinical results hampers clinical awareness and translation into practice. In this study, BMSCs were cultured in synovial fluid (SF) obtained from OA patients and, for the first time, a thorough characterization of soluble factors and extracellular vesicles (EVs)-embedded miRNAs was performed in this condition. Molecular data were sifted through the sieve of molecules and pathways characterizing the OA phenotype in immune cells and joint tissues. One-hundred and twenty-five secreted factors and one-hundred and ninety-two miRNAs were identified. The combined action of both types of molecules was shown to, first, foster BMSCs interaction with the most important OA immune cells, such as macrophages and T cells, driving their switch towards an anti-inflammatory phenotype and, second, promote cartilage homeostasis assisting chondrocyte proliferation and attenuating the imbalance between destructive and protective extracellular matrix-related players. Overall, molecular data give an understanding of the clinical results observed in OA patients and can enable a faster translation of BMSC-based products into everyday clinical practice.
Keywords: osteoarthritis, mesenchymal stromal cells, secretome, extracellular vesicles, miRNAs, immune cells, cartilage
1. Introduction
Osteoarthritis (OA) is a common and disabling condition affecting 250 million people worldwide [1]. It is a complex chronic disease characterized by structural alterations in the hyaline articular cartilage [2] and the synovium [3], with further involvement of the entire set of joint elements such as subchondral bone, ligaments, capsule, and periarticular muscles [4]. The crucial feature of OA is an imbalance between the repair and destruction of joint tissues, involving the presence of an immune response mainly mediated by macrophages [5] and T cells [6] in the synovial membrane and fluid. Regarding macrophages, high levels are detected in OA patients compared to healthy controls and correlate with clinical symptoms [7]. Furthermore, increases in inflammatory macrophage associated molecules in OA patient synovial fluid (SF) are linked with clinical outcomes in OA [8,9]. Similarly, T cells in the SF are associated with the pathogenesis of OA [10] and are the major constituents of synovial infiltrates in the membranes of OA patients [11]. To date, no drugs have a disease-modifying effect on OA, but rather their action is mostly related to relief of symptoms [12]. For this reason, treatments that may reduce disease manifestation in addition to slowing or stopping its progression are actively sought. In this frame, mesenchymal stromal cells (MSCs) have emerged as an attractive option, both as GMP-grade products [13] and as point-of-care products [14] such as bone marrow aspirate concentrate (BMAC) [15], micro-fragmented adipose tissue (MFAT) [16] or stromal vascular fraction (SVF) [17].
In all these applications, MSCs showed the potential of tissue restoration and inflammation management [18], with these features mainly ascribed to their secreted factors, either free [19] or conveyed within extracellular vesicles (EVs) [20], such as miRNAs, altogether defining the cell “secretome” [21]. Thus, a comprehensive fingerprint of the MSC-secretome is mandatory to give a molecular explanation of MSC therapeutic benefits and allow a faster translation of these innovative medicinal options. In this perspective, the main pitfall of traditional analyses of secreted factors and EV-miRNAs is the culturing conditions that are far from those which MSCs encounter when injected in the knee of OA patients. Mostly, MSC-secretome analysis was conducted cultivating cells in standard medium [22]. Although giving valuable hints, only a pathological environment may drive a more reliable secretome fingerprint, as demonstrated for bone marrow-MSCs (BMSCs) exposed to both healthy and degenerative human intervertebral disc conditions [23]. This pivotal experiment showed that a pathological environment induced in BMSCs a pronounced secretory phenotype able to promote immunomodulation, adjustment of ECM synthesis and degradation imbalance, and ECM reorganization.
To shed light on the secretory capacity of BMSCs as a treatment for OA, the aim of this work was to mimic an osteoarthritic joint environment by cultivating cells in the presence of SF from OA patients and evaluate the presence and amount of both soluble factors and EV-embedded miRNAs. Identified molecules were discussed within the framework of OA affected cell and tissue types including cartilage, synovium, macrophages and T cells.
2. Materials and Methods
2.1. Synovial Fluid Collection
An aliquot of synovial fluid (SF) (mean volume 2.7 mL ± 2.2) was collected from 13 patients (8 females, 5 males, mean age 69 ± 8 years; Kellgren and Lawrence III–IV grade) undergoing total knee arthroplasty. Floating cells and debris were removed by centrifugation (16,000× g, 10 min at RT) and supernatants stored at −80 °C. Before experiments were performed, single aliquots were pooled.
2.2. ELISA Characterization of Pooled SF
Two hundred-fifty μL pooled SF were treated with 12.5 μL of 40 mg/mL Hyaluronidase (Sigma-Aldrich, Milan, Italy) and 1.25 μL of Protease Inhibitor Cocktail (abcam, Cambridge, MA, USA). Incubation was performed for 30 min at 37 °C. Two-fold diluted digested OA-SF was used for soluble factors detection with the enzyme-linked immunosorbent assay (ELISA) Quantibody® Human Cytokine Array 4000 Kit (RayBiotech, Norcross, GA, USA, https://www.raybiotech.com/quantibody-human-cytokine-array-4000/, accessed on 3 October 2022) following manufacturer’s protocol and four technical replicates. Concentrations were determined by comparison with standard samples. The amount of each factor was determined as pg/mL.
2.3. Bone Marrow Collection, BMSCs Isolation and Expansion
Total bone marrow aspirate from 3 female donors (mean age 50 ± 2 years) was seeded at a concentration of 50,000 nucleated cells/cm2 in αMEM (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% FBS at 37 °C, 5% CO2, and 95% humidity. Colonies appearing after 2 weeks were detached and BMSCs seeded at 4000/cm2 and cultured up to passage 3 for all described experiments. For secretome collection, BMSCs at 70% confluence were cultured for 2 days in the presence of 50% pooled SF (1:1 diluted in complete cell culture medium). This concentration was used since in clinical trials BMSCs are usually suspended in a volume between 5 and 8 mL [24,25,26] and injected in the OA synovial cavity that was reported to contain between 3 and 11 mL of SF [27,28], depending on the cohort of patients, for a final 50% SF concentration after BMSCs administration. After the treatment, BMSCs were washed 3 times with PBS and serum-free αMEM added (0.07 mL/cm2). After 2 days, the secretome was collected and centrifuged at 376× g for 5 min at 4 °C, 1000× g for 15 min at 4 °C, 2000× g for 15 min at 4 °C and twice at 4000× g for 15 min at 4 °C. Clarified secretomes were used for ELISA and EVs analyses. After secretome removal, BMSCs were counted and viability assessed with a NucleoCounter NC-3000 (ChemoMetec, Allerod, Denmark).
2.4. Flow Cytometry Characterization of SF-Treated BMSCs
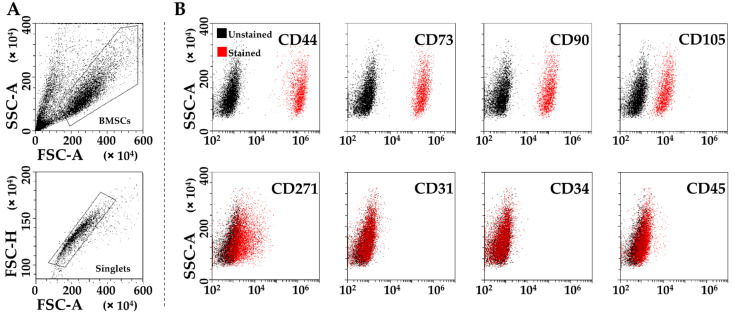
After 2 days in 50% pooled SF, BMSCs were detached and stained (30 min at 4 °C in the dark) with both hemato/endothelial (CD31-PerCP Vio700 clone REA730, CD34-FITC clone AC136, CD45-PE Vio770 clone REA747) and MSC (CD44-PE Vio770 clone REA690, CD73-PE clone REA804, CD90-FITC clone REA897, CD105-PerCP Vio700 clone REA794, CD271-PE clone REA844) markers (Miltenyi Biotec, Bergisch Gladbach, Germany). BMSCs were detected by flow cytometry with a CytoFLEX flow cytometer (Beckman Coulter, Fullerton, CA, USA), collecting a minimum of 30,000 events. The following combinations of antibodies were used: CD73/90/105/44 and CD34/271/31/45.
2.5. ELISA Characterization of SF-Treated BMSCs Secretome
The enzyme-linked immunosorbent assay (ELISA) Quantibody® Human Cytokine Array 4000 Kit was used, as previously described, to assay 2-fold diluted 250 μL secretomes from SF-treated BMSCs. The amount of each factor in pg/mL was converted into pg/million cells by multiplying the original value for the total volume in ml and dividing by the total number of cells. Values are shown as mean ± SD.
2.6. Protein-Protein Interaction Network
The online tool STRING (http://www.string-db.org, accessed on 14 June 2022) (database v11.5) was used to generate interactome maps of ELISA-identified proteins. The following settings were used: (i) organism, Homo sapiens; (ii) meaning of network edges, evidence; (iii) active interaction sources, experiments; (iv) minimum required interaction scores, low confidence (0.150).
2.7. Characterization of EVs in SF-Treated BMSCs Secretomes
All analyses were performed after 1:1 secretome dilution with PBS.
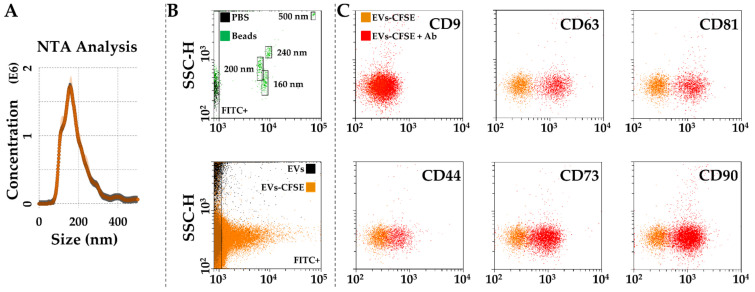
Nanoparticle tracking analysis (NTA): secretomes were run by Nanosight NS-300 system (NanoSight Ltd., Amesbury, UK) (5 recordings of 60 s) and EVs visualized with NTA software v3.4 providing both high-resolution particle size distribution profiles and concentration measurements.
Flow cytometry: 3 aliquots were analyzed. (i) Unstained, (ii) 5(6)-carboxyfluorescein diacetate succinimidyl ester (CFDA-SE, Sigma-Aldrich, St. Louis, MO, USA) stained (1 µM final concentration, 30 min at 37 °C) to visualize EVs after transformation into FITC-channel fluorescent carboxyfluorescein succinimidyl ester (CFSE), iii) after CFDA-SE supplementation, stained (30 min at 4 °C) with CD9-APC clone HI9A, CD63-APC clone H5C6, CD81-APC clone 5A6, CD44-APC clone BJ18, CD73-APC clone AD2, CD90-APC clone 5E10 (Biolegend, San Die-go, CA, USA). Samples were analyzed with a CytoFlex flow cytometer collecting at least 30,000 events. FITC-fluorescent nanobeads (160, 200, 240, and 500 nm, Biocytex, Marseille, France) were used as internal control for efficient detection in the nanometric range.
2.8. Total RNA Isolation from EVs and miRNAs Quantification
Secretomes were 1:1 diluted in PBS for a total volume of 10 mL and ultra centrifugated (100,000× g, 9 h at 4 °C) in an Optima L-90K Ultracentrifuge (Beckman Coulter) equipped with a Type 70.1 Ti Fixed-Angle Titanium Rotor (Beckman Coulter). To evaluate the efficiency of RNA recovery and cDNA synthesis, an exogenous Arabidopsis thaliana ath-miR-159a (30 pg) synthetic miRNA spike was added to EV pellets before total RNA extraction with miRNeasy and RNeasy Cleanup Kits (Qiagen, Hilden, Germany). The OpenArray system (Life Technologies, Foster City, CA, USA) equipped with the 384-well OpenArray plates was used to detect the presence of 754 miRNAs, according to the manufacturer’s instructions. Each single miRNA was considered as present and considered for further analyses only when amplification appeared in all three samples. The equalization of technical differences was performed scoring ath-miR-159 spike-in CRT. Eventually, the global mean method allowed normalization between samples.
2.9. Identification of miRNAs Target
The mRNA targets of detected miRNAs were identified with miRTarBase v8.0 (https://mirtarbase.cuhk.edu.cn/~miRTarBase/miRTarBase_2022/php/index.php, accessed on 14 March 2022) [29]. Only miRNA-mRNA interactions supported by strong experimental evidence were considered.
2.10. Statistical and Computational Analyses
Statistical analyses were performed with GraphPad Prism Software version 5 (GraphPad, San Diego, CA, USA). The Pearson correlation coefficient (R2) formula was used to calculate the linear association between samples. The outcome results were interpreted according to the degree of association [30].
Principal component analysis (PCA) and hierarchical clustering were obtained with ClustVis package (https://biit.cs.ut.ee/clustvis/, accessed on 15 June 2022) [31]. Maps were generated using the following settings for both rows and columns clustering distance and method: correlation and average, respectively.
3. Results
3.1. SF Characterization
The values of 190 detected soluble factors in pooled SF are shown in Supplementary Table S1. Three molecules were detected at >100,000 pg/mL: Insulin-like growth factor-binding protein 4 (IGFBP4, 248,050), Vascular cell adhesion protein 1 (VCAM1, 109,797) and Intercellular adhesion molecule 2 (ICAM2, 109,414). Other IGFBPs found at high levels, included IGFBP2 (44,358), IGFBP3 (42,150), IGFBP6 (6732) and IGFBP1 (5671). Out of classically described OA-related inflammatory cytokines, IL1B (99), IFNG (86) and IL6 (209) were found, while TNFA was absent. The recently described and synovia pro-inflammatory Interferon lambda-1 (IFNL1) was found at a high level (13,977), with Interferon lambda-2 (IFNL2) at a lower level (274). TGFB1, an OA-SF reported molecule, was amongst the most abundant molecules (29,154) as well as Plasminogen (PLG, 42,376) and its activator Urokinase plasminogen activator surface receptor (PLAUR, 11,597) along with PLAUR repressor Plasminogen activator inhibitor 1 (SERPINE1, 7097). Of note, 3 molecules interfering with IL1B signaling were detected: Interleukin-1 receptor antagonist protein (IL1RN, 459), Interleukin-1 receptor-like 1 (IL1RL1, 182) and Interleukin-1 receptor type 1 (IL1R1, 9). With a similar function on IL6, Interleukin-6 receptor subunit alpha (IL6R, 19,933) and Interleukin-6 receptor subunit beta (IL6ST, 15,002) were found at high levels. Although TNFA was not present, several receptors could be identified, including Tumor necrosis factor receptor superfamily member 1A (TNFRSF1A, 19,497), 17 (12,355), 1B (10,977), 14 (2201), 21 (1902), 10C (993), 18 (404), 10D (313), 9 (263), 8 (228) and 11B (104). Finally, 2 inhibitors of ECM-degrading enzymes were strongly detected: Metalloproteinase inhibitor 2 (TIMP2, 25,220) and 1 (TIMP1, 9879). The >10,000 pg/mL factors are shown in Table 1.
Table 1.
>10,000 pg/mL pooled SF factors.
| TYPE | FACTOR | (pg/mL) | |
|---|---|---|---|
| GF | IGFBP4 | 248,050 | Insulin-like growth factor-binding protein 4 |
| REC | VCAM1 | 109,797 | Vascular cell adhesion protein 1 |
| CYT | ICAM2 | 109,414 | Intercellular adhesion molecule 2 |
| GF | IGFBP2 | 44,358 | Insulin-like growth factor-binding protein 2 |
| CYT | PLG | 42,376 | Plasminogen |
| GF | IGFBP3 | 42,150 | Insulin-like growth factor-binding protein 3 |
| CYT | SIGLEC5 | 32,371 | Sialic acid-binding Ig-like lectin 5 |
| GF | TGFB1 | 29,154 | Transforming growth factor beta-1 |
| INF | TIMP2 | 25,220 | Metalloproteinase inhibitor 2 |
| REC | SELL | 20,525 | L-selectin |
| INF | IL6R | 19,933 | Interleukin-6 receptor subunit alpha |
| GF | BMP4 | 19,550 | Bone morphogenetic protein 4 |
| INF | TNFRSF1A | 19,497 | Tumor necrosis factor receptor superfamily member 1A |
| GF | CSF1R | 19,117 | Macrophage colony-stimulating factor 1 receptor |
| CYT | IL6ST | 15,002 | Interleukin-6 receptor subunit beta |
| CHE | IFNL1 | 13,977 | Interferon lambda-1 |
| REC | TNFRSF17 | 12,355 | Tumor necrosis factor receptor superfamily member 17 |
| REC | PLAUR | 11,597 | Urokinase plasminogen activator surface receptor |
| INF | TNFRSF1B | 10,977 | Tumor necrosis factor receptor superfamily member 1B |
CHE: Chemokine; CYT: Cytokine; GF: Growth factor; INF: Inflammation; REC: Receptor.
3.2. Immunophenotype of SF-Treated BMSCs
After 48 h in SF, BMSCs resulted strongly positive for MSC (CD44 100% ± 0, CD73 100% ± 0, CD 90 100% ± 0, CD105 95 % ± 4) and negative for hemato-endothelial (CD31 3% ± 0, CD34 0% ± 0, CD45 3% ± 1) markers (Figure 1). BMSCs were also positive to adult-MSC specific CD271 (19% ± 11), although at lower levels as previously reported [32] (Figure 1).
Figure 1.
Flow cytometry analysis of SF-treated BMSCs. (A), after exclusion of debris (upper panel), single cells were identified (lower panel). (B), staining of single cells for general mesenchymal (CD44, CD73, CD90 and CD105, positive), BMSC-specific (CD271, positive) and hemato-endothelial markers (CD31, CD34, and CD45, negative). Representative plots are shown.
3.3. Characterization of SF-Treated BMSCs Secreted Factors
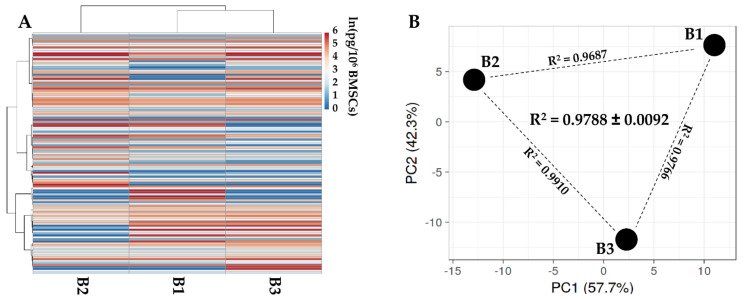
One-hundred and twenty-five secreted factors were detected in all 3 SF-treated BMSCs secretomes (Supplementary Table S2). Hierarchical clustering showed a closer relationship between BMSCs 1 and 3, although PCA analysis highlighted a conserved distance between donors, further confirmed by correlation analysis (Figure 2). For these reasons, an average value was calculated for each factor. The 2 most abundant (>100,000 pg/million BMSCs) factors were IGFBP4 and 3. Another 9 molecules had an amount between 100,000 and 10,000 pg/million, including IGFBP2, TIMP1 and TIMP2, along with another ECM protective factor (SERPINE1). In this group, Transforming growth factor beta-1 (TGFB1), IFNL1, Interleukin-9 (IL9), Bone morphogenetic protein 4 (BMP4) and Vascular endothelial growth factor A (VEGFA) were also found. Twenty-four factors had a concentration between 10,000 and 1000 pg/million, with Platelet factor 4 (PF4) and IGFBP6 being by far the most abundant ones. In this group, C-C and C-X-C motif chemokines appeared, including CCL21/26/27 and CXCL11/16. A few growth factors could be found, such as Hepatocyte growth factor-like protein (MST1), Hepatocyte growth factor (HGF) and Fibroblast growth factor 4 (FGF4). Forty-eight proteins populated the 1000 to 100 pg/million group, with 2 C-C motif chemokines being among the most abundant (CCL2/25) along with other C-C and C-X-C motif factors (CCL5 and CXCL9/10/12). In this group, several interleukins were also present including IL1A/6/16/17B/23A/31 and IL8 (also known as CXCL8). C-C/C-X-C chemokines and interleukins also populated the <100 pg/million group of 42 factors, in particular, CCL1/4/7/8/11/13/14/16/17/18/20/24 and CXCL5/13, with IL1B/2/4/7/12/15. In this group, many growth factors could also be found, including Protransforming growth factor alpha (TGFA), Placenta growth factor (PGF), Granulocyte/macrophage colony-stimulating factor (CSF1/2/3), Platelet-derived growth factor subunit A (PDGFA), Brain-derived neurotrophic factor (BDNF), Beta-nerve growth factor (NGF) and Pro-epidermal growth factor (EGF). IFNG was also in this group. The most abundant factors (>1000 pg/million BMSCs) are shown in Table 2.
Figure 2.
Comparison of secreted factor profiles between SF-treated BMSCs under study. (A), heat map of hierarchical clustering analysis of the ln(x) transformed pg/million BMSCs values of detected factors with sample clustering tree at the top. The color scale reflects the absolute expression levels: red shades = high expression levels and blue shades = low expression levels. B1, B2 and B3 stands for BMSC donors 1, 2 and 3. (B), principal component analysis of the ln(x) transformed pg/million BMSCs values of detected factors. X and Y axes show principal component 1 and principal component 2, which explain 57.7% and 42.3% of the total variance. B1, B2 and B3 stands for BMSC donors 1, 2 and 3.
Table 2.
Most abundant (>1000 pg/million cells) SF-treated BMSCs factors.
| pg/Million BMSCs | |||||||
|---|---|---|---|---|---|---|---|
| TYPE | FACTOR | B1 | B2 | B3 | MEAN | SD | FUNCTION |
| GF | IGFBP4 | 109,869 | 131,569 | 124,587 | 122,009 | 9045 | Insulin-like growth factor-binding protein 4 |
| GF | IGFBP3 | 84,091 | 117,198 | 102,025 | 101,105 | 13,532 | Insulin-like growth factor-binding protein 3 |
| GF | TGFB1 | 39,339 | 32,615 | 30,189 | 34,048 | 3870 | Transforming growth factor beta-1 |
| INF | TIMP2 | 22,096 | 28,331 | 30,462 | 26,963 | 3550 | Metalloproteinase inhibitor 2 |
| CHE | IFNL1 | 19,399 | 20,613 | 22,381 | 20,798 | 1225 | Interferon lambda-1 |
| CHE | IL9 | 1770 | 24,217 | 16,270 | 14,086 | 9293 | Interleukin-9 |
| INF | TIMP1 | 12,116 | 14,563 | 14,503 | 13,727 | 1140 | Metalloproteinase inhibitor 1 |
| GF | BMP4 | 7872 | 10,892 | 20,075 | 12,946 | 5190 | Bone morphogenetic protein 4 |
| CYT | SERPINE1 | 10,110 | 14,584 | 13,253 | 12,649 | 1876 | Plasminogen activator inhibitor 1 |
| GF | IGFBP2 | 6439 | 14,130 | 10,661 | 10,410 | 3145 | Insulin-like growth factor-binding protein 2 |
| GF | VEGFA | 7794 | 12,442 | 10,732 | 10,323 | 1919 | Vascular endothelial growth factor A |
| CHE | PF4 | 9652 | 10,111 | 8745 | 9503 | 567 | Platelet factor 4 |
| GF | IGFBP6 | 6466 | 10,314 | 9509 | 8763 | 1657 | Insulin-like growth factor-binding protein 6 |
| CHE | MIF | 3604 | 4526 | 4973 | 4368 | 570 | Macrophage migration inhibitory factor |
| REC | VCAM1 | 4131 | 5719 | 2852 | 4234 | 1173 | Vascular cell adhesion protein 1 |
| CYT | INHBA | 4344 | 4350 | 3770 | 4155 | 272 | Inhibin beta A chain |
| CHE | XCL1 | 2921 | 4047 | 5177 | 4048 | 921 | Lymphotactin |
| CHE | CCL27 | 4283 | 2370 | 3979 | 3544 | 840 | C-C motif chemokine 27 |
| CHE | CXCL16 | 2374 | 3787 | 3932 | 3364 | 702 | C-X-C motif chemokine 16 |
| INF | TNFRSF1A | 2640 | 3403 | 3410 | 3151 | 361 | Tumor necrosis factor receptor superfamily member 1A |
| CHE | MST1 | 3356 | 1822 | 2150 | 2443 | 659 | Hepatocyte growth factor-like protein |
| CHE | CCL26 | 2212 | 932 | 4108 | 2417 | 1305 | C-C motif chemokine 26 |
| CHE | CCL21 | 2552 | 3078 | 1594 | 2408 | 614 | C-C motif chemokine 21 |
| CHE | CXCL11 | 2165 | 3168 | 1466 | 2266 | 698 | C-X-C motif chemokine 11 |
| REC | PLAUR | 1515 | 2482 | 1641 | 1879 | 429 | Urokinase plasminogen activator surface receptor |
| CYT | ANGPT1 | 1553 | 2005 | 1837 | 1798 | 187 | Angiopoietin-1 |
| GF | BMP7 | 2239 | 327 | 1766 | 1444 | 813 | Bone morphogenetic protein 7 |
| CYT | IL6ST | 1239 | 1842 | 1090 | 1390 | 325 | Interleukin-6 receptor subunit beta |
| GF | HGF | 1058 | 1462 | 1443 | 1321 | 186 | Hepatocyte growth factor |
| GF | FGF4 | 1536 | 1484 | 729 | 1250 | 369 | Fibroblast growth factor 4 |
| CYT | ANG | 971 | 1427 | 1301 | 1233 | 193 | Angiogenin |
| REC | ALCAM | 956 | 1209 | 1239 | 1135 | 127 | CD166 antigen |
| CYT | FST | 690 | 1260 | 1284 | 1078 | 274 | Follistatin |
| CYT | CTSS | 808 | 1256 | 1150 | 1071 | 191 | Cathepsin S |
| GF | KDR | 1078 | 887 | 1102 | 1022 | 96 | Vascular endothelial growth factor receptor 2 |
CHE: Chemokine; CYT: Cytokine; GF: Growth factor; INF: Inflammation; REC: Receptor. B1, B2 and B3 stands for BMSC donors 1, 2 and 3.
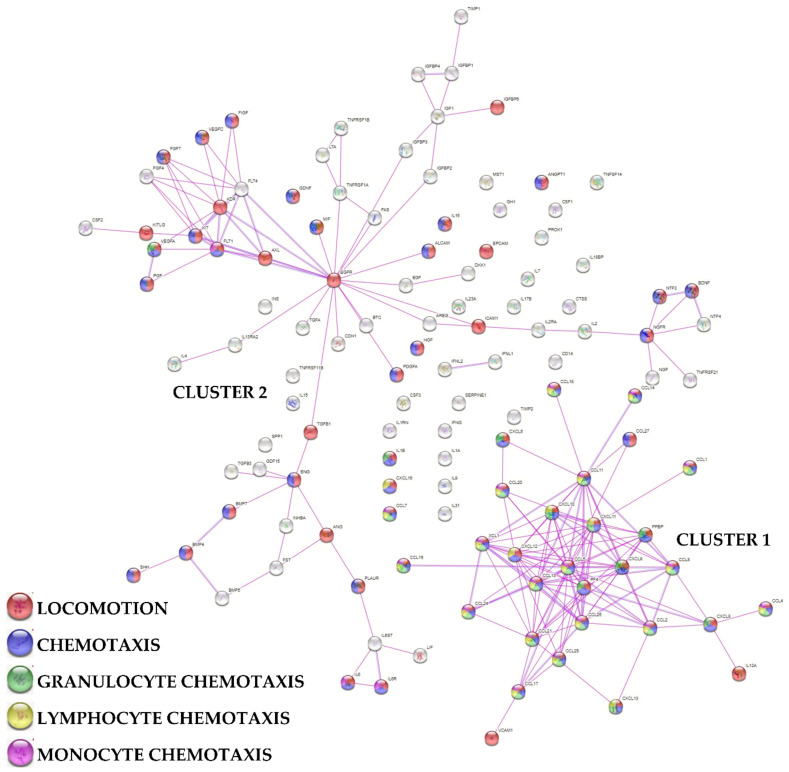
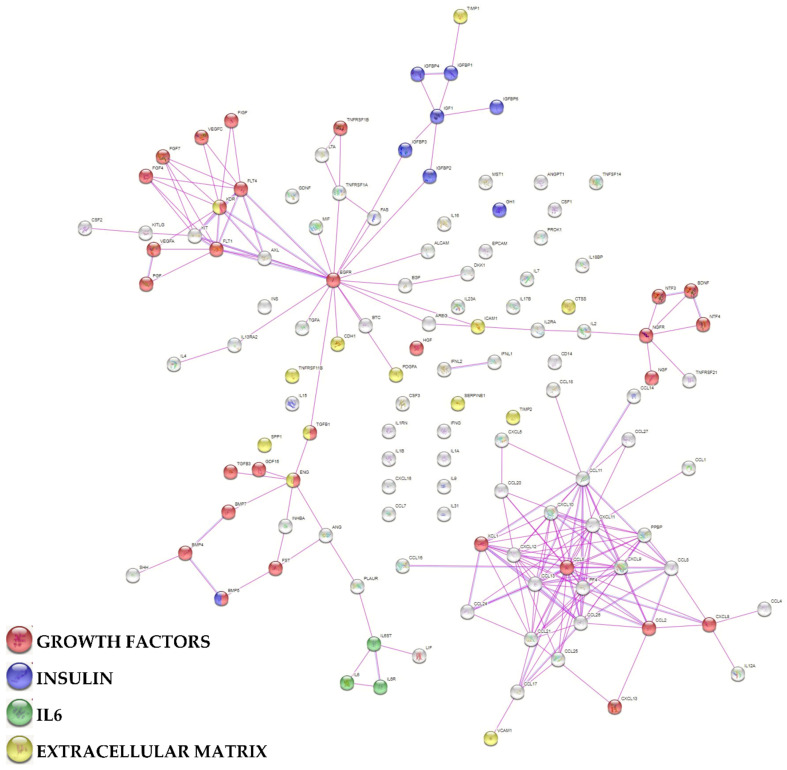
To assign an overall function to the detected factors, a protein association network analysis was performed (Figure 3 and Figure 4). Two main clusters emerged, one tighter (Cluster 1) and another spread along the interaction map. In particular, Cluster 1 was composed of 28 proteins, all belonging to the gene ontology (GO) groups Locomotion (GO:0040011; see Supplementary Table S3 for factors related to this term and others in the text below) and Chemotaxis (GO:0006935) with the exception of IL12A and VCAM1 for this term (Figure 3). After sifting through the different blood cell types, 24 factors were related to Granulocytes (GO:0071621), 20 to Lymphocytes (GO:0048247) and 16 to Monocytes (GO:0002548). Locomotion and Chemotaxis also defined a small subgroup within Cluster 2 irradiating from EGFR. In this case, no obvious distinction of further levels related to blood cell types was observed. Notably, in this subgroup, several growth factors defined the term Cellular response to growth factor stimulus (GO:0071363). They were shared with 2 other small subgroups connected with EGFR, one defined by neuro-related proteins and one more heterogeneous with preponderance of BMPs (Figure 4), from which a small cluster associated with IL6 and its receptors (Interleukin-6 mediated signaling pathway, GO:0070102) could be identified. From EGFR, another small subcluster could be framed and associated with insulin signaling (Regulation of insulin-like growth factor receptor signaling pathway, GO:0043567). Finally, 13 proteins were related to Extracellular Matrix Organization (GO:0030198), without the formation of an interconnected cluster or group.
Figure 3.
Functional association network for identified secreted factors released by SF-treated BMSCs. Using the online tool STRING, protein-protein interaction levels for 125 proteins of the BMSCs secretome were mined. The blue connections are for proteins with known interactions based on curated databases; violet connections for proteins with experimentally determined interactions. Empty nodes, proteins of unknown 3D structure; filled nodes, known or predicted 3D structure. Locomotion, chemotaxis, granulocyte chemotaxis, lymphocyte chemotaxis and monocyte chemotaxis related factors are shown.
Figure 4.
Functional association network for identified secreted factors released by SF-treated BMSCs. Using the online tool STRING, protein-protein interaction levels for 125 proteins of the BMSCs secretome were mined. The blue connections are for proteins with known interactions based on curated databases; violet connections for proteins with experimentally determined interactions. Empty nodes, proteins of unknown 3D structure; filled nodes, known or predicted 3D structure. Growth factors, insulin, IL6 and extracellular matrix related factors are shown.
3.4. Characterization of SF-Treated BMSC-EVs
After SF treatment, BMSCs released 537 ± 68 EVs per cell in 48 h. EVs had an average mode size of 155 nm ± 3, with 65 % of particles below 200 nm (Figure 5A). Flow cytometry analysis confirmed dimensional data after comparison with latex beads of nanometric size (Figure 5B). Particles were positive for CD63 (91% ± 0) and CD81 (90.3% ± 0.5) EV markers, while almost negative for CD9 (4.7% ± 0.5) as previously shown for BMSC-EVs [33] (Figure 5C). With respect to MSC-lineage markers, EVs were strongly positive for CD73 (82.3% ± 1.7) and CD90 (85.3% ± 0.5), while CD44 staining showed lower expression (46.0% ± 1) although the complete population shift suggested the homogeneous presence of the epitope (Figure 5C).
Figure 5.
Characterization of SF-treated BMSC-EVs. (A), EVs size analysis from NTA data. (B), flow cytometer was first calibrated to score FITC-fluorescent particles of nanometer scale (upper panel, starting from 160 nm). EVs were CFSE stained to allow their identification and gating in the FITC channel (lower panel). (C), after gating, CFSE+ EVs showed positive staining for extracellular vesicle defining molecules CD63 and CD81, and MSC markers CD44, CD73 and CD90. CD9, another EV postulated marker, was barely detectable. Representative cytograms are presented.
3.5. Identification of EV-Embedded miRNAs
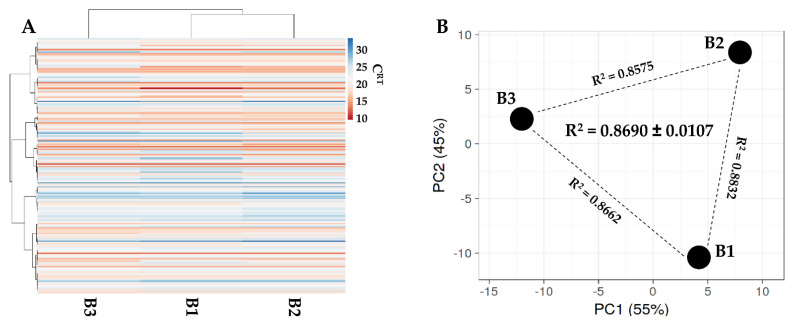
One-hundred and ninety-two miRNAs were identified at varying levels of intensity (Supplementary Table S4). Hierarchical clustering showed closer similitude for B1 and B2 samples (Figure 6A), although a pattern of overall similarity emerged by both PCA and correlation analyses (Figure 6B). For these reasons an average value for each miRNA was calculated. Moreover, to sharpen the genetic message, only miRNAs falling in the first quartile of expression were considered for further analyses. This choice is based on the knowledge that, even for the most abundant miRNAs, in MSC-EVs no more than one molecule per EV is present [34] and that to transfer one miRNA molecule to a recipient cell at least 100 EVs are needed [35]. Within this frame, 46 miRNAs emerged (Table 3) that were able to represent the 96.3% of the screened genetic weight of EVs. Further, to avoid possible misleading players, hsa-miR-720 and hsa-miR-1274A/B were excluded from analyses, being likely fragments of tRNA [36]. The most abundant miRNAs were hsa-miR-518f-3p (17.8% genetic weight), hsa-miR-24-3p (13.2%) and hsa-miR-193b-3p (8.39%). At the bottom of the quartile were hsa-miR-34a-5p (0.2%), hsa-miR-376a-3p (0.2%) and hsa-miR186-5p (0.2%). To evaluate EV-miRNAs’ effect in recipient cells, experimentally validated miRNA-mRNA interactions were sifted (Supplementary Table S5). miRNAs with the highest number of reported interactions were hsa-miR-145-5p (1.25%, 143 mRNA targets), hsa-miR-21-5p (0.88%, 135) and hsa-miR-34-5p (0.17%, 132). Analyzing the top 3 most abundant miRNAs, the interactions were: hsa-miR-518f-3p (17.76%, 0 targets), hsa-miR-24-3p (13.17%, 88) and hsa-miR-193b-3p (8.39%, 17). Considering all first quartile miRNAs, 1142 univocal mRNAs were validated (Supplementary Table S6).
Figure 6.
Comparison of EV-miRNA expression profiles between SF-treated BMSCs under study. (A), heat map of hierarchical clustering analysis of the normalized CRT values of detected miRNAs with sample clustering tree at the top. The color scale reflects the absolute expression levels: red shades = high expression levels (low CRT values) and blue shades = low expression levels (high CRT values). B1, B2 and B3 stands for BMSC donors 1, 2 and 3. (B), principal component analysis of the normalized CRT values of detected miRNAs. X and Y axis show principal component 1 and principal component 2 that explain 55% and 45% of the total variance. B1, B2 and B3 stands for BMSC donors 1, 2 and 3.
Table 3.
miRNAs detected in SF-treated BMSC-EVs and falling in the first quartile of expression.
| miRBase ID | Crt | |||||
|---|---|---|---|---|---|---|
| B1 | B2 | B3 | Mean | SD | Weight % | |
| hsa-miR-518f-3p | 9.26 | 12.46 | 13.80 | 11.84 | 1.90 | 17.75728 |
| hsa-miR-24-3p | 12.47 | 12.14 | 12.20 | 12.27 | 0.15 | 13.16838 |
| hsa-miR-193b-3p | 13.04 | 12.78 | 12.95 | 12.92 | 0.11 | 8.388070 |
| hsa-miR-222-3p | 13.26 | 13.12 | 12.81 | 13.06 | 0.19 | 7.591257 |
| hsa-miR-1274B | 13.12 | 13.18 | 13.33 | 13.21 | 0.09 | 6.863791 |
| hsa-miR-574-3p | 13.50 | 13.24 | 13.55 | 13.43 | 0.14 | 5.882132 |
| hsa-miR-191-5p | 13.43 | 13.53 | 13.56 | 13.51 | 0.06 | 5.582864 |
| hsa-miR-484 | 14.17 | 13.74 | 13.90 | 13.94 | 0.18 | 4.142033 |
| hsa-miR-320a-3p | 14.34 | 13.94 | 14.30 | 14.19 | 0.18 | 3.468564 |
| hsa-miR-197-3p | 15.20 | 14.51 | 14.89 | 14.86 | 0.28 | 2.181023 |
| hsa-miR-19b-3p | 15.44 | 15.55 | 15.68 | 15.55 | 0.10 | 1.352228 |
| hsa-miR-214-3p | 15.47 | 15.49 | 15.81 | 15.59 | 0.16 | 1.317989 |
| hsa-miR-99a-5p | 15.69 | 15.67 | 15.63 | 15.66 | 0.03 | 1.254117 |
| hsa-miR-145-5p | 15.61 | 15.59 | 15.80 | 15.67 | 0.09 | 1.250356 |
| hsa-miR-125b-5p | 15.76 | 15.67 | 15.65 | 15.69 | 0.05 | 1.226040 |
| hsa-miR-1274A | 15.80 | 15.93 | 16.14 | 15.96 | 0.14 | 1.021016 |
| hsa-miR-627-5p | 17.05 | 14.73 | 16.22 | 16.00 | 0.96 | 0.992408 |
| hsa-miR-342-3p | 16.22 | 15.80 | 16.38 | 16.13 | 0.25 | 0.904174 |
| hsa-miR-409-3p | 16.19 | 16.33 | 15.94 | 16.15 | 0.16 | 0.891932 |
| hsa-miR-21-5p | 15.80 | 16.24 | 16.46 | 16.17 | 0.28 | 0.884135 |
| hsa-miR-106a-5p | 16.02 | 16.42 | 16.33 | 16.25 | 0.17 | 0.832009 |
| hsa-miR-16-5p | 16.37 | 16.29 | 16.19 | 16.28 | 0.07 | 0.816961 |
| hsa-miR-17-5p | 16.10 | 16.36 | 16.39 | 16.28 | 0.13 | 0.815264 |
| hsa-let-7b-5p | 16.39 | 16.40 | 16.66 | 16.48 | 0.12 | 0.710056 |
| hsa-miR-29a-3p | 16.71 | 16.83 | 16.39 | 16.64 | 0.19 | 0.635518 |
| hsa-miR-30c-5p | 16.48 | 16.81 | 16.81 | 16.70 | 0.16 | 0.610052 |
| hsa-miR-221-3p | 16.79 | 16.75 | 16.86 | 16.80 | 0.05 | 0.569594 |
| hsa-miR-92a-3p | 17.12 | 16.93 | 17.10 | 17.05 | 0.08 | 0.478969 |
| hsa-miR-30b-5p | 16.93 | 17.20 | 17.35 | 17.16 | 0.17 | 0.444629 |
| hsa-miR-20a-5p | 16.92 | 17.28 | 17.43 | 17.21 | 0.21 | 0.428492 |
| hsa-miR-132-3p | 17.31 | 17.13 | 17.47 | 17.30 | 0.14 | 0.402206 |
| hsa-miR-618 | 13.41 | 14.78 | 24.32 | 17.50 | 4.85 | 0.350464 |
| hsa-miR-138-5p | 17.39 | 17.43 | 17.87 | 17.56 | 0.22 | 0.335722 |
| hsa-miR-382-5p | 18.55 | 17.53 | 17.41 | 17.83 | 0.51 | 0.279323 |
| hsa-miR-663b | 17.74 | 17.68 | 18.19 | 17.87 | 0.23 | 0.271308 |
| hsa-miR-483-5 | 18.29 | 17.46 | 17.99 | 17.91 | 0.34 | 0.264011 |
| hsa-miR-199a-3p | 18.14 | 18.30 | 17.86 | 18.10 | 0.18 | 0.231327 |
| hsa-miR-520e-3p | 15.88 | 17.10 | 21.67 | 18.22 | 2.49 | 0.213307 |
| hsa-miR-31-5p | 18.39 | 18.44 | 17.85 | 18.23 | 0.27 | 0.211883 |
| hsa-miR-28-3p | 18.39 | 18.51 | 18.18 | 18.36 | 0.14 | 0.193402 |
| hsa-miR-146a-5p | 18.47 | 18.77 | 18.02 | 18.42 | 0.31 | 0.185395 |
| hsa-miR-720 | 18.69 | 18.31 | 18.31 | 18.44 | 0.18 | 0.183435 |
| hsa-miR-193a-5p | 18.27 | 18.37 | 18.71 | 18.45 | 0.19 | 0.181957 |
| hsa-miR-34a-5p | 19.15 | 18.58 | 17.84 | 18.52 | 0.54 | 0.172740 |
| hsa-miR-376a-3p | 18.51 | 18.59 | 18.58 | 18.56 | 0.03 | 0.168094 |
| hsa-miR-186-5p | 19.01 | 18.74 | 18.40 | 18.72 | 0.25 | 0.151005 |
In italics miRNAs that are possibly tRNA fragments and therefore excluded from analyses. B1, B2 and B3 stands for BMSC donors 1, 2 and 3.
3.6. EV-miRNAs Effect on OA-Related Molecules and Cell Types
To envision the effect of SF-treated BMSC-EVs miRNAs for OA pathology, reported OA-regulators expressed in at least 1% of OA chondrocytes, synoviocytes, macrophages and T cells [37] were compared with the list of first quartile miRNA targets (Table 4). Of note, the majority of pro-inflammatory cytokines reported to induce and sustain OA were targeted, including IL1A/B, IL6 and TNF although this last one was not detected in the OA synovial fluid used in the study. The most targeted (>1% of the scored EV genetic weight) cytokine was IL1A, due to hsa-miR-191-5p (5.58% of the genetic weight), followed by TNF (main regulator hsa-miR-125-5p, 1.23%) and CCL5 (main hsa-miR-214-3p, 1.32%). Concerning growth factors, TGFB1 was by far the most targeted molecule, due to the second most abundant miRNA hsa-miR-24-3p (13.2%) and hsa-miR574-3p (5.88%). Other preferential (>1% EV weight) targets were VEGFA, regulated by 10 miRNAs leaded by hsa-miR-145-5p (1.25%), KITLG (hsa-miR-320a-3p, 3.47%), ANGPT2 (main hsa-miR-145-5p, 1.25%), TGFB2 (main hsa-miR-145-5p, 1.25%), CTGF (main hsa-miR-145-5p, 1.25%), BMP2 (main hsa-miR-106a-5p, 0.83%), IGF2 (main hsa-miR-125b-5p, 1.23%), BDNF (main hsa-miR-16-5p, 0.82%) and HGF (main hsa-miR-16-5p, 0.82%). Looking at proteases and related factors, EV-miRNAs target 11 proteins involved with ECM degradation and only 2 with protective features. In the first group, the top factors were MMP14 (mainly due to hsa-miR-24-3p, 13.17%), MMP1 (main hsa-miR-222-3p, 7.59%) and PLAU (hsa-miR-193b-3p, 8.39%). Other proteases preferentially inhibited by EV-miRNAs were MMP2 (main hsa-miR-125b-5p, 1.23%), ADAM17 (hsa-miR-145-5p, 1.25%) and MMP13 (hsa-miR-125b-5p, 1.23%). For ECM protective factors, TIMP3 is the most heavily targeted (main hsa-miR-222-3p, 7.59%), followed by TIMP2 (main hsa-miR-106a-5p, 0.83%). Further, due to its abundance, the most impactful miRNA was hsa-miR-24-3p (13.17%), able to target both TGFB1 and MMP14. Other miRNAs that can tip the protection/destruction balance were hsa-miR-193b-3p (8.39%) regulating PLAU and hsa-miR-222-3p (7.59%) having a dual role by acting on MMP1 and TIMP3. Other miRNAs able to strongly regulate OA factors were hsa-miR-574-3p (5.88%, targets TGFB1), hsa-miR-191-5p (5.58%, IL1A) and hsa-miR320a-3p (3.47%, KITLG). Lastly, synoviocytes were the most targeted cells, with 32 OA-regulators, followed by HLA-DR+ cells (including inflammatory macrophages) (19), chondrocytes (17) and T cells (2).
Table 4.
OA regulators expressed by at least 1% of indicated cell types and targeted by first quartile EV-miRNAs.
| CHO | SYN | HLA-DR+ | T CELL | % WEIGHT | MAIN miRNA | |
|---|---|---|---|---|---|---|
| CYTOKINES | ||||||
| TNF | X | X | 2.05 | hsa-miR-125-5p (1.23%) | ||
| IL6 | X | X | 0.19 | hsa-miR-146a-5p (0.19%) | ||
| IL1B | X | X | 0.88 | hsa-miR-21-5p (0.88%) | ||
| IL1A | X | X | 5.58 | hsa-miR-191-5p (5.58%) | ||
| CXCL12 | X | X | 0.97 | hsa-miR-221-3p (0.57%) | ||
| CCL5 | X | X | X | 1.51 | hsa-miR-214-3p (1.32%) | |
| IL11 | X | X | X | 0.61 | hsa-miR-30c-5p (0.61%) | |
|
GROWTH
FACTORS |
||||||
| TGFB1 | X | X | X | X | 19.24 | hsa-miR-24-3p (13.17%) |
| IGF1 | X | X | 0.87 | hsa-miR-29a-3p (0.64%) | ||
| FGF2 | X | X | 0.97 | hsa-miR-16-5p (0.82%) | ||
| BMP2 | X | X | X | 1.65 | hsa-miR-106a-5p (0.83%) | |
| VEGFA | X | X | X | 6.22 | hsa-miR-145-5p (1.25%) | |
| HGF | X | X | 1.05 | hsa-miR-16-5p (0.82%) | ||
| ANGPT2 | X | X | 2.48 | hsa-miR-145-5p (1.25%) | ||
| CTGF | X | X | X | 1.86 | hsa-miR-145-5p (1.25%) | |
| KITLG | X | X | X | 3.47 | hsa-miR-320a-3p (3.47%) | |
| TGFB2 | X | X | X | 2.13 | hsa-miR-145-5p (1.25%) | |
| INHBB | X | 0.17 | hsa-miR-34a-5p (0.17%) | |||
| IGF2 | X | X | 1.5 | hsa-miR-125b-5p (1.23%) | ||
| BDNF | X | 1.22 | hsa-miR-16-5p (0.82%) | |||
| PROTEASES | ||||||
| ADAM12 | X | X | 0.64 | hsa-miR-29a-3p (0.64%) | ||
| ADAM17 | X | X | X | 1.25 | hsa-miR-145-5p (1.25%) | |
| ADAMTS9 | X | 0.64 | hsa-miR-29a-3p (0.64%) | |||
| MMP1 | X | 8.84 | hsa-miR-222-3p (7.59%) | |||
| MMP2 | X | X | 3.26 | hsa-miR-125b-5p (1.23%) | ||
| MMP9 | X | X | 0.4 | hsa-miR-132-3p (0.40%) | ||
| MMP13 | 1.23 | hsa-miR-125b-5p (1.23%) | ||||
| MMP14 | X | X | 14.42 | hsa-miR-24-3p (13.17%) | ||
| PLAU | X | X | 8.39 | hsa-miR-193b-3p (8.39%) | ||
| PLAT | X | X | 0.88 | hsa-miR-21-5p (0.88%) | ||
| APC | X | X | 2.06 | hsa-miR-125b-5p (1.23%) | ||
| TIMP2 | X | X | 1.26 | hsa-miR-106a-5p (0.83%) | ||
| TIMP3 | X | X | 9.86 | hsa-miR-222-3p (7.59%) |
CHO stands for chondrocytes, SYN for synoviocytes.
Eventually, abundant miRNAs were sifted through available literature describing those miRNAs directly involved in homeostasis, inflammation, protection and destruction of the most important cell types driving OA (Table 5). Regarding cartilage [38], 9 miRNAs have protective functions and 7 have destructive functions. The overall genetic weight for the protective group is mostly due to 5 miRNAs with single weight >1% (hsa-miR-24-3p, 13.17%; hsa-miR-193b-3p, 8.39%; hsa-miR-222-3p, 7.59%, hsa-miR-320a-3p, 3.47%; hsa-miR-125b-5p, 1.23%), for a total of 35.94%. In contrast, none of the destructive miRNAs had a weight >1%, for a total of 3.10%. Therefore, overall, the protection vs. destruction ratio is 11.6 in favor of cartilage healing and maintenance. For synovia, to date, very little is known about the role of single miRNAs [39]. We identified one miRNA (hsa-miR-29a-3p, 0.64%) with protective and one (hsa-miR-34a-5p, 0.17%) with destructive properties, together with hsa-miR-146a-5p (0.19%) having a dual role. Thus, it is not possible to outline a clear picture. Concerning macrophages [40], 2 miRNAs were reported to promote a pro-inflammatory M1 while 5 miRNAs supported an anti-inflammatory M2 phenotype. The overall weight was 2.48% for inflammation (hsa-miR-145-5p, 1.25%; hsa-miR-125b-5p, 1.23%) vs. 21.83% for anti-inflammation (mainly due to hsa-miR24-3p, 13.17% and hsa-miR-222-3p, 7.59%), for a ratio of 8.8 in favor of M2 macrophages. Finally, miRNAs involved in T cells [41] were analyzed. Eight pro-activating and 4 anti-activating miRNAs were found. Due to hsa-miR-24-3p (13.17%), repressing miRNAs had a total weight of 15.48% vs. 6.88% of activating molecules, ending in a ratio of 2.3 in favor of inhibition of T cell activity. Thus, overall, protection and inflammation reduction signals far exceeded damaging and pro-inflammatory features for almost all OA-affected tissues under analysis.
Table 5.
EV-miRNAs involved in homeostasis of OA-affected tissues and cells.
| TISSUE/CELLS | % WEIGHT | ROLE |
|---|---|---|
| CARTILAGE | ||
| Protective | ||
| hsa-miR-24-3p | 13.17 | Prevents ECM degradation, increases chondrocyte viability |
| hsa-miR-193b-3p | 8.39 | Reduces cartilage degradation |
| hsa-miR-222-3p | 7.59 | Reduces cartilage degradation |
| hsa-miR-320a-3p | 3.47 | Increases chondrocyte viability |
| hsa-miR-125b-5p | 1.23 | Prevents aggrecan loss |
| hsa-miR-17-5p | 0.82 | Induces autophagy |
| hsa-miR-221-3p | 0.57 | Prevents ECM degradation |
| hsa-miR-92a-3p | 0.48 | Increases collagen deposition |
| hsa-miR-199a-3p | 0.23 | Anti-catabolic |
| Total | 35.94 | |
| Destructive | ||
| hsa-miR-21-5p | 0.88 | Negatively regulates chondrogenesis |
| hsa-miR-16-5p | 0.82 | Cartilage degradation |
| hsa-miR-30b-5p | 0.44 | Pro-apoptotic, ECM degradation |
| hsa-miR-138-5p | 0.34 | Cartilage degradation |
| hsa-miR-483-5 | 0.26 | Chondrocyte hypertrophy, ECM degradation and cartilage angiogenesis |
| hsa-miR-146a-5p | 0.19 | Activator in early OA |
| hsa-miR-34a-5p | 0.17 | Apoptosis |
| Total | 3.10 | |
| Dual | ||
| hsa-miR-145-5p | 1.25 | Regulates chondrocyte proliferation and fibrosis |
| SYNOVIUM | ||
| Protective | ||
| hsa-miR-29a-3p | 0.64 | Anti-fibrotic effects |
| Destructive | ||
| hsa-miR-34a-5p | 0.17 | Enhances synovial inflammation |
| Dual | ||
| hsa-miR-146a-5p | 0.19 | Enhances/Suppresses synovial inflammation |
| MACROPHAGE | ||
| M1 | ||
| hsa-miR-145-5p | 1.25 | Pro-M1 |
| hsa-miR-125b-5p | 1.23 | Pro-M1 |
| Total | 2.48 | |
| M2 | ||
| hsa-miR-24-3p | 13.17 | Pro M2, blocks M1 |
| hsa-miR-222-3p | 7.59 | Pro M2 |
| hsa-let-7b-5p | 0.71 | Pro M2 |
| hsa-miR-146a-5p | 0.19 | Pro M2, blocks M1 |
| hsa-miR-34a-5p | 0.17 | Pro M2 |
| Total | 21.83 | |
| T CELL | ||
| Pro-Activation | ||
| hsa-miR-19b-3p | 1.35 | Reduces PTEN repressor |
| hsa-miR-214-3p | 1.32 | Reduces PTEN repressor |
| hsa-miR-21-5p | 0.88 | Reduces PTEN repressor |
| hsa-miR-106a-5p | 0.83 | Represses IL10 |
| hsa-miR-17-5p | 0.82 | Reduces PTEN repressor and promotes IFNγ |
| hsa-let-7b-5p | 0.71 | Represses IL10 |
| hsa-miR-221-3p | 0.57 | Downregulates PIK3R1 |
| hsa-miR-132-3p | 0.40 | Downregulates PIK3R1 |
| Total | 6.88 | |
| Anti-activation | ||
| hsa-miR-24-3p | 13.17 | Represses IFNγ in activated CD4+ and CD8+ |
| hsa-miR-125b-5p | 1.23 | Maintains T cell naïve state |
| hsa-miR-342-3p | 0.90 | Downregulated upon activation |
| hsa-miR-146a-5p | 0.19 | Represses activation markers |
| Total | 15.48 | |
| Dual | ||
| hsa-miR-31-5p | 0.21 | Upregulates IL2, downregulated with activation |
4. Discussion
In this report the secreted factors and EV-embedded miRNAs released by BMSCs treated with synovial fluid of OA patients were analyzed for the first time. The main strength and innovative feature of this study lies in mimicking a closer therapeutic application of BMSC-derived products. In fact, the exposure of BMSC to the synovial fluid of patients with OA instead of single pro-inflammatory molecules makes the observations more reliable and similar to what happens in the clinical setting.
The main findings were that the soluble molecules released by BMSCs in the presence of OA synovial fluid are involved with locomotion and chemotaxis of immune cells while EV-miRNAs have protective and anti-inflammatory roles in the tissues and cells, promoting and maintaining the OA phenotype.
In recent years, several clinical studies have investigated BMSC-based therapies for OA [42], including both randomized control trials and observational studies. Most of the reports included patients with grade II–III of Kellgren–Lawrence, with some cases of grade IV suggesting that the therapy may be envisioned to be applied for both early and late stage OA. The main finding of these studies was the improvement of the function of the knee joint after BMSCs intra-articular injections, along with gain in tissue structure and overall pain reduction. Regarding cartilage, a few studies also reported a decrease of poor cartilage areas with tissue quality improvement quantified by radiographic and MRI measurements [24,25,43,44], although in small patient cohorts. BMSCs also reduced pain [26,44,45,46], with results, maintained up to several months, that allowed an increase in the walking time function [26]. BMSCs injections were also shown to relieve synovitis with a concomitant decrease of pro-inflammatory macrophages in synovial fluids [47]. For these reasons, BMSC-based therapies showed a sufficient effect to postpone or avoid knee replacement when studied in the contra-lateral joint in patients with bilateral osteoarthritis [48].
These anti-inflammatory and protective/regenerative results observed in OA patients may find a molecular background by the properties of the soluble factors and EV-miRNAs in the secretome of BMSCs exposed to OA synovial fluid, as described in this paper. Regarding inflammation, BMSCs were reported to interact with many kinds of immune cells, including macrophages [49,50] and T cells [50,51], with cell-cell contact being postulated to facilitate MSC-regulated immunosuppression [52]. In this view, SF-treated BMSCs secrete several molecules able to attract all immune cells, including macrophages and lymphocytes. Once in proximity, BMSCs may promote phenotype switch through factors and EV-miRNAs. In the first group, we identified IL1RN (alias IL1RA, found at 280 pg/mL), a well described molecule mediating the MSCs immunosuppressive effect at different levels. IL1RA was shown to inhibit the proliferation of T lymphocytes, to increase the amount of Tregs and to induce the macrophage polarization from M1 to M2 phenotype, in turn secreting IL10 and exerting an additional suppressive effect on T cells [53]. Moreover, in a collagen-induced OA murine model, IL1RA was a crucial factor in protection from OA progression by decreasing the percentage of activated T lymphocytes and increasing the percentage of Tregs [53]. A similar effect on Tregs was proposed for other two molecules present in SF-treated BMSCs secretome, CCL18 (46 pg/mL) and TGFB1 (34,048 pg/mL), with their neutralization leading to a significant reduction in MSC-induced Tregs formation from conventional T cells [54,55]. Of note, both molecules are among the drivers of the formation of FoxP3+ Tregs from naïve CD4+ T cell [54,56]. BMSC-secreted TGFB1 was also reported to suppress T cell proliferation [57] and, on a more general level, was shown to prevent their activation [58]. Similarly, BMSC-derived ICAM1 (178 pg/mL) was demonstrated to be critical for the MSC-mediated immunosuppression of T cells, contributing to the rapid suppression of TNF and IFNG in activated T cells [59]. These effects on immune cells for factors released after OA-SF treatment were supported by the function of the most abundant EV-embedded miRNAs, which target several well described OA inflammatory cytokines such as IL1A/B, IL6 and TNF. EV-miRNAs also target other important cytokines associated with OA, such as CXCL12, whose levels in SF were closely related to the radiographic severity of OA [60], CCL5, recruiting Th1/17/22 to the affected joint triggering the inflammation process [61], and IL11, recently shown to be upregulated with OA severity in the synovial fluid of OA patients [62]. Of importance, all these targeted inflammatory cytokines are expressed by OA HLA-DR+ cells, including M1 macrophages, and activated T cells for CCL5. Consistently, overall, the most abundant miRNAs have a preponderance for M2 polarization of macrophages and for reduction of activation of T cells. For both cell types, the balance towards immune suppression is driven by hsa-miR-24-3p (13.17 % of the EV genetic weight). This miRNA was shown to regulate macrophage polarization and plasticity, inhibiting M1 and supporting M2 phenotype when overexpressed [63], with basal hsa-miR-24-3p expression being higher in M2 macrophages [64]. For T cells, hsa-miR-24-3p targets IFNG in activated CD4+ [65] and CD8+ [66] cells, with its delivery through EVs being able to inhibit T-cell proliferation and Th1 and Th17 differentiation and induce Tregs [67]. Finally, another EV-miRNA with an important role in tipping the balance towards macrophage M2 polarization is hsa-miR-22-3p (7.59 %), whose expression is again opposed to IFNG [68]. Thus, overall, the combination of soluble factors and EV-miRNAs support the observed anti-inflammatory effects of BMSCs when injected in the OA joint through a combined action relying on SF immune cell attraction followed by phenotype polarization, especially for macrophages and T cells. The intrinsic capacity of secreted molecules and EVs to permeate the synovia [69] suggest that their modulatory properties can be envisioned also for tissue resident immune cells that greatly contribute to OA joint inflammatory status [70].
Together with immune cell interaction, secreted molecules and EV-miRNAs suggested a molecular background for tissue protection, with particular focus on cartilage. Among the most abundant secreted factors, TIMP1 (13,727 pg/mL) and TIMP2 (26,963 pg/mL) were widely reported as crucial molecules for cartilage protection. TIMPs are key regulators of the metalloproteinases (MMPs) that degrade the extracellular matrix (ECM) [71], this phenomenon being one of the main events leading to cartilage disruption in OA. Accordingly, the supplementation of bovine cartilage with TIMP1/2 prevented the release of collagen fragments [72], and new molecules altering pathological MMPs/TIMPs imbalance in OA joints are currently being tested. An example is Paeoniflorin, a pinane monoterpene glucoside, that was able to downregulate the expression of MMPs and increase the expression of TIMP1 mRNA and protein in rat chondrocytes [73]. A second example is the histone deacetylase inhibitor trichostatin A that in OA rats was able to reduce the imbalance of the TIMPs/MMPs ratio through the increase of TIMP1 and decrease of MMP1/3/13 [74]. In this frame, injection of BMSCs could be a way to tip the balance towards protection through the release of TIMP1 and 2. Further, among the most abundantly secreted molecules, TGFB1 (34,048 pg/mL) was also proposed as an OA therapeutic tool. In fact, TGFB1 is crucial for cartilage maintenance and its supplementation can enhance cartilage repair [75]. Nevertheless, it may also create problems in other tissues of the joint like fibrosis of the synovia and osteophyte formation. In this regard, SF-treated BMSCs might be an intriguing option, due to the concurrent release of both TGFB1 and EV-embedded miRNAs such as hsa-miR-24-3p that can locally inhibit TGFB synthesis at sites of unwanted side effects such as bone/cartilage and synovia. In the same paradigm of BMSCs secreted molecules as therapeutic tools, SerpinE1 (12,649 pg/mL) is a direct inhibitor of both tissue- and urokinase-type activators of plasminogen into plasmin, thereby regulating plasmin-related cleavage of ECM components such as fibronectin, glycoproteins and proteoglycans and direct activation of MMPs [76]. Consistently, SerpinE1 was able to protect against cartilage collagen breakdown in an ex vivo model of cartilage destruction through the inhibition of proteolytic activators of MMPs, with its levels decreasing in OA cartilage together with an overall increase of plasmin activity [77]. Again, under the paradigm of secretome molecules as part of the therapeutic tool, the abundant BMP4 (12,946 pg/mL) plays a crucial role in maintaining a chondrogenic phenotype and enhancing matrix production [78]. Consistently, BMP4 (12,946 pg/mL) in association with muscle MSCs, was able to efficiently regenerate cartilage when injected intra-articularly in a rat OA model [79], and BMP4-transfected adipose-MSCs significantly improved in vivo chondrogenesis in a rabbit OA model [80]. Thus, several SF-treated BMSCs secretome molecules have ECM and cartilage protective roles. Nevertheless, there are some abundant factors that might have a tricky effect on cartilage and ECM. Some falling in this category are IGFBPs, including the 2 most detected proteins (IGFBP4, 122,009 pg/mL and IGFBP3, 101,105 pg/mL) and other members of the family (IGFBP2, 10,410 pg/mL, IGFBP6, 8763 pg/mL and IGFBP1, 724 pg/mL). They bind IGFs, known to have potent anabolic actions on chondrocytes. Since they are increased in OA cartilage, IGFBPs’ binding and masking of IGF1 might underlie the reduced cartilage matrix synthesis in degenerated areas of OA cartilage [81], making IGFBPs detrimental factors. Nevertheless, in cartilage, ECM macromolecular complexes between IGFBPs and IGFs have been detected that, when degraded by MMPs acting on IGFBPs, lead to increased amounts of available IGFs around chondrocytes as part of an attempted repair process of the cartilage structure [82]. Of note, the ratio between cartilage ECM synthesis and degradation, that is strongly affected in OA, is not only regulated by inhibitors or other proteins influencing their amounts/activity but also by several EV-embedded miRNAs, making the picture far more complex. In fact, we observed that abundant miRNAs may target several MMPs, in particular MMP1 and 2, and other ECM degrading proteases such as ADAMs, PLAU/PLAT and APC, while only TIMP3 had strong targeting. In particular, the urokinase-type activator PLAU, that is increased in its levels in both OA-SF and cartilage [83], is heavily targeted by hsa-miR-193b-3p (8.39 %). This miRNA, together with hsa-miR-24-3p (13.17 %), miR-222-3p (7.59 %) and miR-320a-3p (3.47 %) is responsible for the overall cartilage protective role of EV-embedded miRNAs that go beyond the regulation of single proteins. hsa-miR-24-3p was reduced in OA cartilage; in chondrocytes, its overexpression downregulated apoptosis, inflammation and ECM degradation by targeting BCL2L12 [84]. hsa-miR-193b-3p expression was significantly reduced in OA cartilage and its overexpression strongly enhanced in vivo cartilage formation by directly targeting HDAC3 and promoting H3 acetylation [85]. hsa-miR-222-3p was found significantly downregulated in OA cartilage [86] and its over-expression significantly suppressed cartilage destruction by targeting HDAC4 [87]. Eventually, hsa-miR-320-3p expression was significantly reduced in OA cartilage and its overexpression was associated with increased collagen deposition and COL2A1 expression [88]. Therefore, overall, secreted factors and EV-miRNAs account for the cartilage protective features of BMSCs when injected intra-articularly in the joints of OA patients.
We are aware that this study has some limitations. First, the number of factors and miRNAs is limited to the techniques used, ELISA and qRT-PCR. Especially for miRNAs, we preferred to detect the presence of very well characterized players, being the vast majority of the, to date (September 2022), 38,589 identified molecules still lacking a proper characterization not only for OA but also in relation to other pathologies or regulated targets and pathways. Second, the secretome was obtained in starving conditions after culturing BMSCs in synovial fluid. This was necessary to avoid contamination of FBS and SF-derived factors and EVs. The OA-SF used in the study was obtained pooling several samples and using this pool on BMSCs obtained from a different set of patients. This choice was due to the reduced amount of SF that could be obtained from single patients. We are aware that the analyte quantities reported in Table 1 for the pooled SF are not necessarily reflective of the 13 single individual samples and that one sample could have been responsible for the majority of the presence of a particular molecule of interest. Nevertheless, we preferred to describe absolute values rather than relative abundance to give a general roadmap of the molecules that can be found in OA-SF, being conscious that this is of particular relevance for the herein described set of experiments and that with single SF both individual factors and BMSC general response could be at least in part differently modulated. Moreover, it was not possible to obtain bone marrow samples from the same patients undergoing SF collection before knee surgery for obvious ethical reasons, avoiding treatments not related with the surgical procedure. Nevertheless, the choice of a more “realistic” model to test the secretory ability of BMSCs when exposed to a trigger such as OA would make the findings very interesting and worthy of further investigation.
5. Conclusions
BMSCs treatment for OA patients has shown promising results for both inflammation management and cartilage restoration allowing for increased quality of life and a delay of total knee arthroplasty. Soluble factors and EV-miRNAs released from BMSCs cultured in the synovial fluid of OA patients had strong immunomodulatory and cartilage protecting potential. The combined action of both types of molecules is able to promote BMSC interaction with the most common immune cells involved in OA, such as macrophages and T cells, driving their switch towards an anti-inflammatory phenotype. Further, cartilage homeostasis is stimulated by assisting chondrocyte proliferation and attenuating the imbalance in destructive/protective extracellular matrix-related players. Altogether, these data give a molecular grounding to the clinical results and will be a fundamental milestone, allowing a faster translation of this cutting-edge approach into everyday clinical practice.
Acknowledgments
The authors wish to thank all healthcare workers of IRCCS Istituto Ortopedico Galeazzi for their exceptional work during this unexpected world pandemic challenge.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/biology11111632/s1, Table S1: Pooled SF factors; Table S2: SF-treated BMSCs secreted factors; Table S3: GO terms defined by SF-treated BMSCs secreted factors; Table S4: miRNAs detected in SF-treated BMSC-EVs; Table S5: First quartile BMSCs EV-miRNAs targets; Table S6: Univocal miRNA-mRNA targets.
Author Contributions
Conceptualization, E.R., C.P.O. and L.d.G.; methodology, E.R. and C.P.O.; software, C.P.O.; validation, E.R. and C.P.O.; formal analysis, E.R. and C.P.O.; investigation, E.R., C.P.O., F.V. and L.Z.; resources, L.d.G.; data curation, E.R.; writing—original draft preparation, E.R.; writing—review and editing, F.V. and L.d.G.; visualization, C.P.O.; supervision, L.d.G.; project administration, L.d.G.; funding acquisition, L.d.G. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was performed at IRCCS Istituto Ortopedico Galeazzi. Institutional Review Board approval (San Raffaele Hospital Ethics Committee approval on date 16 December 2020, registered under number 214/int/2020) was granted before the beginning of the study.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The raw data presented in this study are openly available in OSF at https://osf.io/62fdj/?view_only=bc28fe818dd5426eb31a2286f6253c48 (accessed on 24 June 2022).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Funding Statement
The work reported in this publication was funded by the Italian Ministry of Health, RCR-2021-23671217 project under the Italian Musculoskeletal Apparatus Network RAMS and Ricerca Corrente. The APC was funded by the Italian Ministry of Health, RCR-2021-23671217 project under the Italian Musculoskeletal Apparatus Network RAMS and Ricerca Corrente.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hunter D.J., Bierma-Zeinstra S. Osteoarthritis. Lancet. 2019;393:1745–1759. doi: 10.1016/S0140-6736(19)30417-9. [DOI] [PubMed] [Google Scholar]
- 2.Kleemann R.U., Krocker D., Cedraro A., Tuischer J., Duda G.N. Altered cartilage mechanics and histology in knee osteoarthritis: Relation to clinical assessment (ICRS Grade) Osteoarthr. Cartil. 2005;13:958–963. doi: 10.1016/j.joca.2005.06.008. [DOI] [PubMed] [Google Scholar]
- 3.Benito M.J., Veale D.J., FitzGerald O., van den Berg W.B., Bresnihan B. Synovial tissue inflammation in early and late osteoarthritis. Ann. Rheum. Dis. 2005;64:1263–1267. doi: 10.1136/ard.2004.025270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Martel-Pelletier J., Barr A.J., Cicuttini F.M., Conaghan P.G., Cooper C., Goldring M.B., Goldring S.R., Jones G., Teichtahl A.J., Pelletier J. Osteoarthritis. Nat. Rev. Dis. Primers. 2016;2:16072. doi: 10.1038/nrdp.2016.72. [DOI] [PubMed] [Google Scholar]
- 5.Thomson A., Hilkens C.M.U. Synovial Macrophages in Osteoarthritis: The Key to Understanding Pathogenesis? Front. Immunol. 2021;12:678757. doi: 10.3389/fimmu.2021.678757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li Y., Luo W., Zhu S., Lei G. T Cells in Osteoarthritis: Alterations and Beyond. Front. Immunol. 2017;8:356. doi: 10.3389/fimmu.2017.00356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kraus V.B., McDaniel G., Huebner J.L., Stabler T.V., Pieper C.F., Shipes S.W., Petry N.A., Low P.S., Shen J., McNearney T.A., et al. Direct in vivo evidence of activated macrophages in human osteoarthritis. Osteoarthr. Cartil. 2016;24:1613–1621. doi: 10.1016/j.joca.2016.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Daghestani H.N., Pieper C.F., Kraus V.B. Soluble macrophage biomarkers indicate inflammatory phenotypes in patients with knee osteoarthritis. Arthritis Rheumatol. 2015;67:956–965. doi: 10.1002/art.39006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huo L.W., Ye Y.L., Wang G.W., Ye Y.G. Fractalkine (CX3CL1): A biomarker reflecting symptomatic severity in patients with knee osteoarthritis. J. Investig. Med. 2015;63:626–631. doi: 10.1097/JIM.0000000000000158. [DOI] [PubMed] [Google Scholar]
- 10.Dolganiuc A., Stăvaru C., Anghel M., Georgescu E., Chichoş B., Olinescu A. Shift toward T lymphocytes with Th1 and Tc1 cytokine-secterion profile in the joints of patients with osteoarthritis. Roum. Arch. Microbiol. Immunol. 1999;58:249–258. [PubMed] [Google Scholar]
- 11.Haynes M.K., Hume E.L., Smith J.B. Phenotypic characterization of inflammatory cells from osteoarthritic synovium and synovial fluids. Clin. Immunol. 2002;105:315–325. doi: 10.1006/clim.2002.5283. [DOI] [PubMed] [Google Scholar]
- 12.Grässel S., Muschter D. Recent advances in the treatment of osteoarthritis. F1000Res. 2020;9:325. doi: 10.12688/f1000research.22115.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Song Y., Zhang J., Xu H., Lin Z., Chang H., Liu W., Kong L. Mesenchymal stem cells in knee osteoarthritis treatment: A systematic review and meta-analysis. J. Orthop. Translat. 2020;24:121–130. doi: 10.1016/j.jot.2020.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lopa S., Colombini A., Moretti M., de Girolamo L. Injective mesenchymal stem cell-based treatments for knee osteoarthritis: From mechanisms of action to current clinical evidences. Knee Surg. Sports Traumatol. Arthrosc. 2019;27:2003–2020. doi: 10.1007/s00167-018-5118-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Silva S., Andriolo L., Boffa A., Di Martino A., Reale D., Vara G., Miceli M., Cavallo C., Grigolo B., Zaffagnini S., et al. Prospective double-blind randomised controlled trial protocol comparing bone marrow aspirate concentrate intra-articular injection combined with subchondral injection versus intra-articular injection alone for the treatment of symptomatic knee osteoarthritis. BMJ Open. 2022;12:e062632. doi: 10.1136/bmjopen-2022-062632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Heidari N., Borg T., Olgiati S., Slevin M., Danovi A., Fish B., Wilson A., Noorani A. Microfragmented Adipose Tissue Injection (MFAT) May Be a Solution to the Rationing of Total Knee Replacement: A Prospective, Gender-Bias Mitigated, Reproducible Analysis at Two Years. Stem Cells Int. 2021;2021:9921015. doi: 10.1155/2021/9921015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang S., Xu H., He B., Fan M., Xiao M., Zhang J., Chen D., Tong P., Mao Q. Mid-term prognosis of the stromal vascular fraction for knee osteoarthritis: A minimum 5-year follow-up study. Stem Cell Res. Ther. 2022;13:105. doi: 10.1186/s13287-022-02788-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhu C., Wu W., Qu X. Mesenchymal stem cells in osteoarthritis therapy: A review. Am. J. Transl. Res. 2021;13:448–461. [PMC free article] [PubMed] [Google Scholar]
- 19.Van Buul G.M., Villafuertes E., Bos P.K., Waarsing J.H., Kops N., Narcisi R., Weinans H., Verhaar J.A.N., Bernsen M.R., van Osch G.J.V.M. Mesenchymal stem cells secrete factors that inhibit inflammatory processes in short-term osteoarthritic synovium and cartilage explant culture. Osteoarthr. Cartil. 2012;20:1186–1196. doi: 10.1016/j.joca.2012.06.003. [DOI] [PubMed] [Google Scholar]
- 20.Cosenza S., Ruiz M., Toupet K., Jorgensen C., Noël D. Mesenchymal stem cells derived exosomes and microparticles protect cartilage and bone from degradation in osteoarthritis. Sci. Rep. 2017;7:16214. doi: 10.1038/s41598-017-15376-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.González-González A., García-Sánchez D., Dotta M., Rodríguez-Rey J.C., Pérez-Campo F.M. Mesenchymal stem cells secretome: The cornerstone of cell-free regenerative medicine. World J. Stem Cells. 2020;12:1529–1552. doi: 10.4252/wjsc.v12.i12.1529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Baberg F., Geyh S., Waldera-Lupa D., Stefanski A., Zilkens C., Haas R., Schroeder T., Stühler K. Secretome analysis of human bone marrow derived mesenchymal stromal cells. Biochim. Biophys. Acta Proteins Proteom. 2019;1867:434–441. doi: 10.1016/j.bbapap.2019.01.013. [DOI] [PubMed] [Google Scholar]
- 23.Wangler S., Kamali A., Wapp C., Wuertz-Kozak K., Häckel S., Fortes C., Benneker L.M., Haglund L., Richards R.G., Alini M., et al. Uncovering the secretome of mesenchymal stromal cells exposed to healthy, traumatic, and degenerative intervertebral discs: A proteomic analysis. Stem Cell Res. Ther. 2021;12:11. doi: 10.1186/s13287-020-02062-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Vega A., Martín-Ferrero M.A., Del Canto F., Alberca M., García V., Munar A., Orozco L., Soler R., Fuertes J.J., Huguet M., et al. Treatment of Knee Osteoarthritis with Allogeneic Bone Marrow Mesenchymal Stem Cells: A Randomized Controlled Trial. Transplantation. 2015;99:1681–1690. doi: 10.1097/TP.0000000000000678. [DOI] [PubMed] [Google Scholar]
- 25.Al-Najar M., Khalil H., Al-Ajlouni J., Al-Antary E., Hamdan M., Rahmeh R., Alhattab D., Samara O., Yasin M., Al Abdullah A., et al. Intra-articular injection of expanded autologous bone marrow mesenchymal cells in moderate and severe knee osteoarthritis is safe: A phase I/II study. J. Orthop. Surg. Res. 2017;12:190. doi: 10.1186/s13018-017-0689-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Davatchi F., Abdollahi B.S., Mohyeddin M., Shahram F., Nikbin B. Mesenchymal stem cell therapy for knee osteoarthritis. Preliminary report of four patients. Int. J. Rheum. Dis. 2011;14:211–215. doi: 10.1111/j.1756-185X.2011.01599.x. [DOI] [PubMed] [Google Scholar]
- 27.Kraus V.B., Stabler T.V., Kong S.Y., Varju G., McDaniel G. Measurement of synovial fluid volume using urea. Osteoarthr. Cartil. 2007;15:1217–1220. doi: 10.1016/j.joca.2007.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li W., Abram F., Pelletier J., Raynauld J., Dorais M., d’Anjou M., Martel-Pelletier J. Fully automated system for the quantification of human osteoarthritic knee joint effusion volume using magnetic resonance imaging. Arthritis Res. Ther. 2010;12:R173. doi: 10.1186/ar3133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Huang H., Lin Y., Li J., Huang K., Shrestha S., Hong H., Tang Y., Chen Y., Jin C., Yu Y., et al. miRTarBase 2020: Updates to the experimentally validated microRNA-target interaction database. Nucleic Acids Res. 2020;48:D148–D154. doi: 10.1093/nar/gkz896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Akoglu H. User’s guide to correlation coefficients. Turk. J. Emerg. Med. 2018;18:91–93. doi: 10.1016/j.tjem.2018.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Metsalu T., Vilo J. ClustVis: A web tool for visualizing clustering of multivariate data using Principal Component Analysis and heatmap. Nucleic Acids Res. 2015;43:W566–W570. doi: 10.1093/nar/gkv468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Barilani M., Banfi F., Sironi S., Ragni E., Guillaumin S., Polveraccio F., Rosso L., Moro M., Astori G., Pozzobon M., et al. Low-affinity Nerve Growth Factor Receptor (CD271) Heterogeneous Expression in Adult and Fetal Mesenchymal Stromal Cells. Sci. Rep. 2018;8:9321. doi: 10.1038/s41598-018-27587-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Barilani M., Peli V., Cherubini A., Dossena M., Dolo V., Lazzari L. NG2 as an Identity and Quality Marker of Mesenchymal Stem Cell Extracellular Vesicles. Cells. 2019;8:1524. doi: 10.3390/cells8121524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Toh W.S., Lai R.C., Hui J.H.P., Lim S.K. MSC exosome as a cell-free MSC therapy for cartilage regeneration: Implications for osteoarthritis treatment. Semin. Cell Dev. Biol. 2017;67:56–64. doi: 10.1016/j.semcdb.2016.11.008. [DOI] [PubMed] [Google Scholar]
- 35.Chevillet J.R., Kang Q., Ruf I.K., Briggs H.A., Vojtech L.N., Hughes S.M., Cheng H.H., Arroyo J.D., Meredith E.K., Gallichotte E.N., et al. Quantitative and stoichiometric analysis of the microRNA content of exosomes. Proc. Natl. Acad. Sci. USA. 2014;111:14888–14893. doi: 10.1073/pnas.1408301111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Schopman N.C.T., Heynen S., Haasnoot J., Berkhout B. A miRNA-tRNA mix-up: tRNA origin of proposed miRNA. RNA Biol. 2010;7:573–576. doi: 10.4161/rna.7.5.13141. [DOI] [PubMed] [Google Scholar]
- 37.Chou C., Jain V., Gibson J., Attarian D.E., Haraden C.A., Yohn C.B., Laberge R., Gregory S., Kraus V.B. Synovial cell cross-talk with cartilage plays a major role in the pathogenesis of osteoarthritis. Sci. Rep. 2020;10:10868. doi: 10.1038/s41598-020-67730-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Endisha H., Rockel J., Jurisica I., Kapoor M. The complex landscape of microRNAs in articular cartilage: Biology, pathology, and therapeutic targets. JCI Insight. 2018;3:e121630. doi: 10.1172/jci.insight.121630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tavallaee G., Rockel J.S., Lively S., Kapoor M. MicroRNAs in Synovial Pathology Associated with Osteoarthritis. Front. Med. 2020;7:376. doi: 10.3389/fmed.2020.00376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xu S.J., Hu H.T., Li H.L., Chang S. The Role of miRNAs in Immune Cell Development, Immune Cell Activation, and Tumor Immunity: With a Focus on Macrophages and Natural Killer Cells. Cells. 2019;8:1140. doi: 10.3390/cells8101140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rodríguez-Galán A., Fernández-Messina L., Sánchez-Madrid F. Control of Immunoregulatory Molecules by miRNAs in T Cell Activation. Front. Immunol. 2018;9:2148. doi: 10.3389/fimmu.2018.02148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xiang X., Zhu S., He H., Yu X., Xu Y., He C. Mesenchymal stromal cell-based therapy for cartilage regeneration in knee osteoarthritis. Stem Cell Res. Ther. 2022;13:14. doi: 10.1186/s13287-021-02689-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Orozco L., Munar A., Soler R., Alberca M., Soler F., Huguet M., Sentís J., Sánchez A., García-Sancho J. Treatment of knee osteoarthritis with autologous mesenchymal stem cells: A pilot study. Transplantation. 2013;95:1535–1541. doi: 10.1097/TP.0b013e318291a2da. [DOI] [PubMed] [Google Scholar]
- 44.Soler R., Orozco L., Munar A., Huguet M., López R., Vives J., Coll R., Codinach M., Garcia-Lopez J. Final results of a phase I-II trial using ex vivo expanded autologous Mesenchymal Stromal Cells for the treatment of osteoarthritis of the knee confirming safety and suggesting cartilage regeneration. Knee. 2016;23:647–654. doi: 10.1016/j.knee.2015.08.013. [DOI] [PubMed] [Google Scholar]
- 45.Emadedin M., Labibzadeh N., Liastani M.G., Karimi A., Jaroughi N., Bolurieh T., Hosseini S., Baharvand H., Aghdami N. Intra-articular implantation of autologous bone marrow-derived mesenchymal stromal cells to treat knee osteoarthritis: A randomized, triple-blind, placebo-controlled phase 1/2 clinical trial. Cytotherapy. 2018;20:1238–1246. doi: 10.1016/j.jcyt.2018.08.005. [DOI] [PubMed] [Google Scholar]
- 46.Bastos R., Mathias M., Andrade R., Bastos R., Balduino A., Schott V., Rodeo S., Espregueira-Mendes J. Intra-articular injections of expanded mesenchymal stem cells with and without addition of platelet-rich plasma are safe and effective for knee osteoarthritis. Knee Surg. Sports Traumatol. Arthrosc. 2018;26:3342–3350. doi: 10.1007/s00167-018-4883-9. [DOI] [PubMed] [Google Scholar]
- 47.Chahal I., Gómez-Aristizábal A., Shestopaloff K., Bhatt S., Chaboureau A., Fazio A., Chisholm J., Weston A., Chiovitti J., Keating A., et al. Bone Marrow Mesenchymal Stromal Cell Treatment in Patients with Osteoarthritis Results in Overall Improvement in Pain and Symptoms and Reduces Synovial Inflammation. Stem Cells Transl. Med. 2019;8:746–757. doi: 10.1002/sctm.18-0183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hernigou P., Delambre J., Quiennec S., Poignard A. Human bone marrow mesenchymal stem cell injection in subchondral lesions of knee osteoarthritis: A prospective randomized study versus contralateral arthroplasty at a mean fifteen year follow-up. Int. Orthop. 2021;45:365–373. doi: 10.1007/s00264-020-04571-4. [DOI] [PubMed] [Google Scholar]
- 49.Zheng Y.H., Deng Y.Y., Lai W., Zheng S.Y., Bian H.N., Liu Z.A., Huang Z.H., Sun C.W., Li H.H., Luo H.M., et al. Effect of bone marrow mesenchymal stem cells on the polarization of macrophages. Mol. Med. Rep. 2018;17:4449–4459. doi: 10.3892/mmr.2018.8457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang Y., Chen X., Cao W., Shi Y. Plasticity of mesenchymal stem cells in immunomodulation: Pathological and therapeutic implications. Nat. Immunol. 2014;15:1009–1016. doi: 10.1038/ni.3002. [DOI] [PubMed] [Google Scholar]
- 51.Luz-Crawford P., Kurte M., Bravo-Alegría J., Contreras R., Nova-Lamperti E., Tejedor G., Noël D., Jorgensen C., Figueroa F., Djouad F., et al. Mesenchymal stem cells generate a CD4+CD25+Foxp3+ regulatory T cell population during the differentiation process of Th1 and Th17 cells. Stem Cell Res. Ther. 2013;4:65. doi: 10.1186/scrt216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kean T.J., Lin P., Caplan A.I., Dennis J.E. MSCs: Delivery Routes and Engraftment, Cell-Targeting Strategies, and Immune Modulation. Stem Cells Int. 2013;2013:732742. doi: 10.1155/2013/732742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Luz-Crawford P., Djouad F., Toupet K., Bony C., Franquesa M., Hoogduijn M.J., Jorgensen C., Noël D. Mesenchymal Stem Cell-Derived Interleukin 1 Receptor Antagonist Promotes Macrophage Polarization and Inhibits B Cell Differentiation. Stem Cells. 2016;34:483–492. doi: 10.1002/stem.2254. [DOI] [PubMed] [Google Scholar]
- 54.Melief S.M., Schrama E., Brugman M.H., Tiemessen M.M., Hoogduijn M.J., Fibbe W.E., Roelofs H. Multipotent stromal cells induce human regulatory T cells through a novel pathway involving skewing of monocytes toward anti-inflammatory macrophages. Stem Cells. 2013;31:1980–1991. doi: 10.1002/stem.1432. [DOI] [PubMed] [Google Scholar]
- 55.Wang D., Huang S., Yuan X., Liang J., Xu R., Yao G., Feng X., Sun L. The regulation of the Treg/Th17 balance by mesenchymal stem cells in human systemic lupus erythematosus. Cell Mol. Immunol. 2017;14:423–431. doi: 10.1038/cmi.2015.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Schmidt A., Zhang X.M., Joshi R.N., Iqbal S., Wahlund C., Gabrielsson S., Harris R.A., Tegnér J. Human macrophages induce CD4(+)Foxp3(+) regulatory T cells via binding and re-release of TGF-β. Immunol. Cell Biol. 2016;94:747–762. doi: 10.1038/icb.2016.34. [DOI] [PubMed] [Google Scholar]
- 57.Di Nicola M., Carlo-Stella C., Magni M., Milanesi M., Longoni P.D., Matteucci P., Grisanti S., Gianni A.M. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood. 2002;99:3838–3843. doi: 10.1182/blood.V99.10.3838. [DOI] [PubMed] [Google Scholar]
- 58.Aoki C.A., Borchers A.T., Li M., Flavell R.A., Bowlus C.L., Ansari A., Gershwin M.E. Transforming growth factor beta (TGF-beta) and autoimmunity. Autoimmun. Rev. 2005;4:450–459. doi: 10.1016/j.autrev.2005.03.006. [DOI] [PubMed] [Google Scholar]
- 59.Zheng S., Huang K., Xia W., Shi J., Liu Q., Zhang X., Li G., Chen J., Wang T., Chen X., et al. Mesenchymal Stromal Cells Rapidly Suppress TCR Signaling-Mediated Cytokine Transcription in Activated T Cells Through the ICAM-1/CD43 Interaction. Front. Immunol. 2021;12:609544. doi: 10.3389/fimmu.2021.609544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Xu Q., Sun X., Shang X., Jiang H. Association of CXCL12 levels in synovial fluid with the radiographic severity of knee osteoarthritis. J. Investig. Med. 2012;60:898–901. doi: 10.2310/JIM.0b013e31825f9f69. [DOI] [PubMed] [Google Scholar]
- 61.Monasterio G., Castillo F., Rojas L., Cafferata E.A., Alvarez C., Carvajal P., Núñez C., Flores G., Díaz W., Vernal R. Th1/Th17/Th22 immune response and their association with joint pain, imagenological bone loss, RANKL expression and osteoclast activity in temporomandibular joint osteoarthritis: A preliminary report. J. Oral Rehabil. 2018;45:589–597. doi: 10.1111/joor.12649. [DOI] [PubMed] [Google Scholar]
- 62.Bollmann M., Lokau J., Garbers C., Bertrand J. Interleukin-11—A new cytokine in osteoarthritis? Osteoarthr. Cartil. 2022;30:S168–S169. doi: 10.1016/j.joca.2022.02.219. [DOI] [Google Scholar]
- 63.Fordham J.B., Naqvi A.R., Nares S. miR-24 Regulates Macrophage Polarization and Plasticity. J. Clin. Cell Immunol. 2015;6:362. doi: 10.4172/2155-9899.1000362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sun X., Liu Y., Wang J., Zhang M., Wang M. Cardioprotection of M2 macrophages-derived exosomal microRNA-24-3p/Tnfsf10 axis against myocardial injury after sepsis. Mol. Immunol. 2022;141:309–317. doi: 10.1016/j.molimm.2021.11.003. [DOI] [PubMed] [Google Scholar]
- 65.Fayyad-Kazan H., Hamade E., Rouas R., Najar M., Fayyad-Kazan M., El Zein N., ElDirani R., Hussein N., Fakhry M., Al-Akoum C., et al. Downregulation of microRNA-24 and -181 parallels the upregulation of IFN-γ secreted by activated human CD4 lymphocytes. Hum. Immunol. 2014;75:677–685. doi: 10.1016/j.humimm.2014.01.007. [DOI] [PubMed] [Google Scholar]
- 66.Chandran P.A., Keller A., Weinmann L., Seida A.A., Braun M., Andreev K., Fischer B., Horn E., Schwinn S., Junker M., et al. The TGF-β-inducible miR-23a cluster attenuates IFN-γ levels and antigen-specific cytotoxicity in human CD8⁺ T cells. J. Leukoc. Biol. 2014;96:633–645. doi: 10.1189/jlb.3A0114-025R. [DOI] [PubMed] [Google Scholar]
- 67.Ye S.-B., Zhang H., Cai T.-T., Liu Y.-N., Ni J.-J., He J., Peng J.-Y., Chen Q.-Y., Mo H.-Y., Jun-Cui, et al. Exosomal miR-24-3p impedes T-cell function by targeting FGF11 and serves as a potential prognostic biomarker for nasopharyngeal carcinoma. J. Pathol. 2016;240:329–340. doi: 10.1002/path.4781. [DOI] [PubMed] [Google Scholar]
- 68.Graff J.W., Dickson A.M., Clay G., McCaffrey A.P., Wilson M.E. Identifying functional microRNAs in macrophages with polarized phenotypes. J. Biol. Chem. 2012;287:21816–21825. doi: 10.1074/jbc.M111.327031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ragni E., Perucca Orfei C., De Luca P., Lugano G., Viganò M., Colombini A., Valli F., Zacchetti D., Bollati V., de Girolamo L. Interaction with hyaluronan matrix and miRNA cargo as contributors for in vitro potential of mesenchymal stem cell-derived extracellular vesicles in a model of human osteoarthritic synoviocytes. Stem Cell Res. Ther. 2019;10:109. doi: 10.1186/s13287-019-1215-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.De Lange-Brokaar B.J.E., Ioan-Facsinay A., van Osch G.J.V.M., Zuurmond A., Schoones J., Toes R.E.M., Huizinga T.W.J., Kloppenburg M. Synovial inflammation, immune cells and their cytokines in osteoarthritis: A review. Osteoarthr. Cartil. 2012;20:1484–1499. doi: 10.1016/j.joca.2012.08.027. [DOI] [PubMed] [Google Scholar]
- 71.Brew K., Nagase H. The tissue inhibitors of metalloproteinases (TIMPs): An ancient family with structural and functional diversity. Biochim. Biophys. Acta. 2010;1803:55–71. doi: 10.1016/j.bbamcr.2010.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ellis A.J., Curry V.A., Powell E.K., Cawston T.E. The prevention of collagen breakdown in bovine nasal cartilage by TIMP, TIMP-2 and a low molecular weight synthetic inhibitor. Biochem. Biophys. Res. Commun. 1994;201:94–101. doi: 10.1006/bbrc.1994.1673. [DOI] [PubMed] [Google Scholar]
- 73.Hu P., Sun F., Jiang L., Bao J., Wu L. Paeoniflorin inhibits IL-1β-induced MMP secretion via the NF-κB pathway in chondrocytes. Exp. Ther. Med. 2018;16:1513–1519. doi: 10.3892/etm.2018.6325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Qu H., Li J., Wu L., Chen W. Trichostatin A increases the TIMP-1/MMP ratio to protect against osteoarthritis in an animal model of the disease. Mol. Med. Rep. 2016;14:2423–2430. doi: 10.3892/mmr.2016.5523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Blaney Davidson E.N., van der Kraan P.M., van den Berg W.B. TGF-beta and osteoarthritis. Osteoarthr. Cartil. 2007;15:597–604. doi: 10.1016/j.joca.2007.02.005. [DOI] [PubMed] [Google Scholar]
- 76.Sanrattana W., Maas C., de Maat S. SERPINs-From Trap to Treatment. Front. Med. 2019;6:25. doi: 10.3389/fmed.2019.00025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Martel-Pelletier J., Faure M.P., McCollum R., Mineau F., Cloutier J.M., Pelletier J.P. Plasmin, plasminogen activators and inhibitor in human osteoarthritic cartilage. J. Rheumatol. 1991;18:1863–1871. [PubMed] [Google Scholar]
- 78.Deng Z.H., Li Y.S., Gao X., Lei G.H., Huard J. Bone morphogenetic proteins for articular cartilage regeneration. Osteoarthr. Cartil. 2018;26:1153–1161. doi: 10.1016/j.joca.2018.03.007. [DOI] [PubMed] [Google Scholar]
- 79.Matsumoto T., Cooper G.M., Gharaibeh B., Meszaros L.B., Li G., Usas A., Fu F.H., Huard J. Cartilage repair in a rat model of osteoarthritis through intraarticular transplantation of muscle-derived stem cells expressing bone morphogenetic protein 4 and soluble Flt-1. Arthritis Rheum. 2009;60:1390–1405. doi: 10.1002/art.24443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Shi J., Zhang X., Zhu J., Pi Y., Hu X., Zhou C., Ao Y. Nanoparticle delivery of the bone morphogenetic protein 4 gene to adipose-derived stem cells promotes articular cartilage repair in vitro and in vivo. Arthroscopy. 2013;29:2001–2011.e2. doi: 10.1016/j.arthro.2013.09.076. [DOI] [PubMed] [Google Scholar]
- 81.Tanaka N., Tsuno H., Ohashi S., Iwasawa M., Furukawa H., Kato T., Fukui N. The attenuation of insulin-like growth factor signaling may be responsible for relative reduction in matrix synthesis in degenerated areas of osteoarthritic cartilage. BMC Musculoskelet Disord. 2021;22:231. doi: 10.1186/s12891-021-04096-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Martin J.A., Scherb M.B., Lembke L.A., Buckwalter J.A. Damage control mechanisms in articular cartilage: The role of the insulin-like growth factor I axis. Iowa Orthop. J. 2000;20:1–10. [PMC free article] [PubMed] [Google Scholar]
- 83.Pérez-García S., Carrión M., Gutiérrez-Cañas I., Villanueva-Romero R., Castro D., Martínez C., González-Álvaro I., Blanco F.J., Juarranz Y., Gomariz R.P. Profile of Matrix-Remodeling Proteinases in Osteoarthritis: Impact of Fibronectin. Cells. 2019;9:40. doi: 10.3390/cells9010040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Xu J., Qian X., Ding R. MiR-24-3p attenuates IL-1β-induced chondrocyte injury associated with osteoarthritis by targeting BCL2L12. J. Orthop. Surg. Res. 2021;16:371. doi: 10.1186/s13018-021-02378-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Meng F., Li Z., Zhang Z., Long D., He A., Liao W. Microrna-193B modulates chondrogenesis and cartilage degeneration via histone deacetylase 3. Osteoarthr. Cartil. 2018;26:S93–S94. doi: 10.1016/j.joca.2018.02.201. [DOI] [Google Scholar]
- 86.Kopańska M., Szala D., Czech J., Gabło N., Gargasz K., Trzeciak M., Zawlik I., Snela S. MiRNA expression in the cartilage of patients with osteoarthritis. J. Orthop. Surg. Res. 2017;12:51. doi: 10.1186/s13018-017-0542-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Song J., Jin E., Kim D., Kim K.Y., Chun C., Jin E. MicroRNA-222 regulates MMP-13 via targeting HDAC-4 during osteoarthritis pathogenesis. BBA Clin. 2014;3:79–89. doi: 10.1016/j.bbacli.2014.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Meng F., Zhang Z., Chen W., Huang G., He A., Hou C., Long Y., Yang Z., Zhang Z., Liao W. MicroRNA-320 regulates matrix metalloproteinase-13 expression in chondrogenesis and interleukin-1β-induced chondrocyte responses. Osteoarthr. Cartil. 2016;24:932–941. doi: 10.1016/j.joca.2015.12.012. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The raw data presented in this study are openly available in OSF at https://osf.io/62fdj/?view_only=bc28fe818dd5426eb31a2286f6253c48 (accessed on 24 June 2022).