Abstract
An increase in the number of vertebrae can significantly affect the meat production performance of livestock, thus increasing carcass weight, which is of great importance for livestock production. The homeobox gene C8 (HOXC8) has been identified as an essential candidate gene for regulating vertebral development. However, it has not been researched on the Dezhou donkey. This study aimed to verify the Dezhou donkey HOXC8 gene’s polymorphisms and assess their effects on multiple vertebral numbers and carcass weight. In this study, the entire HOXC8 gene of the Dezhou donkey was sequenced, SNPs at the whole gene level were identified, and typing was accomplished utilizing a targeted sequencing genotype detection technique (GBTS). Then, a general linear model was used to perform an association study of HOXC8 gene polymorphism loci, multiple vertebral numbers, and carcass weight for screening candidate markers that can be used for molecular breeding of Dezhou donkeys. These findings revealed that HOXC8 included 12 SNPs, all unique mutant loci. The HOXC8 g.15179224C>T was significantly negatively associated with carcass weight (CW) and lumbar vertebrae length (LL) (p < 0.05). The g.15179674G>A locus was shown to be significantly positively associated with the number of lumbar vertebrae (LN) (p < 0.05). The phylogenetic tree constructed for the Dezhou donkey HOXC8 gene and seven other species revealed that the HOXC8 gene was highly conserved during animal evolution but differed markedly among distantly related animals. The results suggest that HOXC8 is a vital gene affecting multiple vertebral numbers and carcass weight in Dezhou donkeys, and the two loci g.15179224C>T and g.15179674G>A may be potential genetic markers for screening and breeding of new strains of high-quality and high-yielding Dezhou donkeys.
Keywords: HOXC8, Dezhou donkey, genetic polymorphism, carcass weight, vertebrae number
1. Introduction
In recent years, people’s awareness of health care has improved, and the market for healthy food such as donkey meat, donkey skin, donkey bone, and donkey milk has expanded, leading to increasing demand for donkey products. Meanwhile, since donkeys experience long intergenerational intervals, most of them are single-birth breeding, which leads to the current situation of demand exceeding supply. It was essential to use molecular marker-assisted breeding to meet the market demand and speed up the selection process of Dezhou donkeys. The Dezhou donkey is one of China’s five premium donkey breeds with solid adaptability, rough feeding resistance, and disease resistance. It is an important livestock and poultry genetic resource and the primary source of donkey products [1,2]. We have assembled a high-quality reference genome for Dezhou donkeys [3], which provided a reference basis for exploring the effects of gene polymorphisms on phenotypic traits and the screening of molecularly assisted breeding markers.
The multi-vertebral number was mainly concentrated in the thoracic and lumbar vertebrae [4]. It was common in livestock populations and significantly impacted meat production [5,6]. Carcass weight is considered one of the most valuable economic traits in livestock production [7,8,9]. To date, studies investigating the effects of multi-vertebral number and carcass weight at the genetic level have focused on animals such as pigs [10,11], cattle [12,13], and sheep [6,14], while fewer studies have been conducted on Dezhou donkeys. Studies on pigs, yaks, and sheep have demonstrated that multi-vertebral numbers contribute to livestock body length [12,15,16]. The body length of livestock was positively correlated with carcass weight and hide production [17]. Based on our previous studies, it has been confirmed that gene single nucleotide polymorphisms do cause changes in body size, vertebrae number, and production performance in Dezhou donkeys. For example, the nuclear receptor subfamily 6 group A member 1 (NR6A1) g.18114954C>T can significantly increase the number of lumbar vertebrae, the total number of thoracolumbar vertebrae, and carcass weight in Dezhou donkeys, which can be a candidate gene to explore the genetic mutation affecting vertebral development [18]. However, the research is not sufficient and more genes need to be examined to screen for more molecular markers to accelerate the selection process of new strains of Dezhou donkeys.
Homeobox gene C8 (HOXC8), a member of the homeobox genes (HOX) family, is a highly conserved evolutionary transcription factor commonly found in vertebrates. It has been demonstrated that the HOX gene was a major regulatory gene in the development of vertebrate and mammalian body axis skeletons and was highly conserved during animal evolution [19,20]. Gene mutation experiments have confirmed that mutations in the mouse HOXC8 gene induce the addition of a thoracic vertebra and an extra pair of ribs [21]. Differential expression of the HOXC8 gene similarly caused vertebral changes in livestock, such as the appearance of multi-vertebral variants in pigs and Mongolian sheep [22,23]. Furthermore, HOXC8 also played a regulatory role in the muscle tissue of adult mice and adipose differentiation in sheep [24,25]. Therefore, it is hypothesized that the HOXC8 gene may be associated with the multi-vertebral number and carcass weight traits of Dezhou donkeys.
In keeping with the above views, this study aimed to detect mutations in the HOXC8 gene, investigate the correlation of its genetic variation with multi-vertebral number and carcass weight traits, and provide potential candidate markers for the selection and breeding of new strains of high-quality and high-yielding Dezhou donkey.
2. Materials and Methods
2.1. Moral Statement
The Animal Ethics Committee approved all experimental animals used in this study of Liaocheng University (LC2019-1), and all conformed to animal welfare requirements.
2.2. Animals and DNA Samples
Blood samples of 400 adult male Dezhou donkeys with phenotypic data were collected from the jugular vein by venipuncture in a donkey farm (Yucheng, Shandong, China). Also, all of them were 2 years old fattening donkeys. Phenotypic records included body height (BH), body length (BL), chest circumference (CC), number of thoracic vertebrae (TN), number of lumbar vertebrae (LN), total number of thoracic and lumbar vertebrae (TLN), thoracic vertebrae length (TL), lumbar vertebrae length (LL), carcass weight (CW) and hide weight (HW). The body size measurements were performed with the donkeys standing on a level ground and in a natural stance, while all measurements were performed by the same operator to reduce human errors. The specific measurement method was described in detail in the article by Zhang et al. [26]. Genomic DNA was extracted using the Blood Genomic DNA Extraction Kit, dissolved in Elution Buffer TB, and stored at 4 degrees.
2.3. Identification of HOXC8 Gene Polymorphism
The mass determination of genomic concentrations was performed using an ultra-micro spectrophotometer (B-500, METASH, Shanghai, China). Then, all 400 eligible genomic samples (n > 1000 ng) were sent to Shijiazhuang MOL BREEDING Biotechnology Co., Ltd. (Shijiazhuang, China) for high-depth resequencing of the HOXC8 gene using targeted sequencing genotype testing technology (GBTS) to examine genetic locus variants in the whole gene range precisely. Six samples were randomly selected to verify the accuracy of GBTS sequencing results, which were matched to three mutation sites by PCR amplification. Then, the PCR products were sent to Sangon Biotech (Shanghai) Co., Ltd. (Shanghai, China) for Sanger sequencing. Finally, the gene’s single nucleotide polymorphisms (SNPs) were identified in Dezhou donkeys and subsequently searched in the Ensembl database (https://asia.ensembl.org/Equus_asinus_asinus/Gene/Summary?db=core;g=ENSEASG00005010151;r=PSZQ01003681.1:11208231-11210397;t=ENSEAST00005015832, accessed on 24 May 2022), European Variation Archive (https://www.ebi.ac.uk/eva/?Home, accessed on 9 November 2022) and NCBI database (https://www.ncbi.nlm.nih.gov/search/all/?term=donkey, accessed on 9 November 2022) to check if these loci were novel mutations.
2.4. Validation of Polymorphic Loci
For better results, three pairs of primers were designed using Primer Premier 5.0 software (version 5.0, PREMIER Biosoft International, San Francisco, CA, USA) based on the HOXC8 gene (GenBank NC_052198.1). More information on the primers is shown in Table 1. And then, six DNA samples were randomly selected as templates for PCR amplification. The total volume of PCR was 25 μL, including 12.5 μL 2× Taq PCR Master Mix (TIANGEN, without-dye, MF002, Beijing, China), 8.5 μL ddH2O, 1 μL each of upstream and downstream (Sangon Biotech, Shanghai, China), and 2 μL of 50 ng/μL DNA template. The PCR conditions were as follows: pre-denaturation at 95 °C for 10 min; followed by 30 cycles of denaturation at 95 °C for 30 s, annealing for 30 s (primer annealing temperature as shown in Table 1), extension at 72 °C for 30 s; and then extension at 72 °C for 10 min to complete the reaction. After the reaction, the PCR products were subjected to 2% agarose electrophoresis in 1× TBE (Tris-borate-EDTA) buffer and visualized by ethidium bromide staining. The PCR products were sent to Sangon Biotech Co., Ltd. (Shanghai, China) for sanger sequencing.
Table 1.
Primer sequences used for single nucleotide polymorphisms (SNPs) validation.
| SNP Position | Primer Sequence (5′-3′) | PCR Size (bp) | Ta (°C) |
|---|---|---|---|
| g.15177692G>A | F:5′-TTGGACCAGGAACAGAGCTG-3′ | 539 | 58 |
| R:5′-GTCGCTCAGAACTCACCATA-3′ | |||
| g.15178184C>T | F:5′-GACAGCAAAGGGGAGGAAGG-3’ | 317 | 58 |
| R:5′-TGCTAGGGTTAGTGTATGAGATTGA-3′ | |||
| g.15179224C>T | F:5′-CACTTCATCCTTCGGTTCTGGA-3′ | 507 | 60 |
| R: 5′-GCCACTCTGCACTTGTAAACA-3′ |
Note: Ta, annealing temperature.
2.5. Statistical Analysis
Allele and genotype frequencies of mutant loci in Dezhou donkeys were calculated using SHEsis online software (http://analysis.bio-x.cn/myAnalysis.php, accessed on 24 May 2022) [27]. We also used the GDIcall Online Calculator (http://www.msrcall.com/Gdicall, accessed on 24 May 2022) to estimate population genetic parameters, including adequate allele number (Ne), population heterozygosity (H), observed heterozygosity (Ho), expected heterozygosity (He) [28] and the polymorphic information content (PIC) [29]. The distribution of genotypes at each locus was tested for compliance with Hardy-Weinberg equilibrium (HWE) using a chi-square test. Associative analysis of phenotypic traits with SNPs was performed using the general linear model of SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). The statistical model is shown as follows:
| Y = µ + G + TL + XG + e |
Y is the observed phenotypic values, µ is the population mean, G is the fixed effect of genotype, TL is the fixed effect of types of thoracolumbar vertebrae, XG is the genotype-related interactions and was removed from the models when found to be not significant (p > 0.05), and e is each observation’s random error effect. Since the experimental population was consistent in age, sex, season of birth, feeding environment and season of sample collection, their effects were not considered in the model. SAS version 9.4 software (SAS Institute Inc., Cary, NC, USA) was also used to correlate phenotypic traits with types of thoracolumbar. The analysis model is: Y = μ + e, where Y represents individual phenotypic records, the μ represents the population mean, and e represents the random error. All phenotypic data were tested for normal distribution using the Shapiro-Wilk test. Phenotypic data that did not conform to a normal distribution were transformed with Box-Cox or Johnson.
2.6. Phylogenetic Analysis
A phylogenetic tree of HOXC8 gene sequences in eight species (Dezhou donkey, pig, horse, mouse, cattle, goat, human, and zebrafish) was constructed by the maximum likelihood method with MEGA-X software 11.0 [30]. For Dezhou donkey, pig, horse, mouse, cattle, goat, human, and zebrafish, sequences were obtained from the NCBI (https://www.ncbi.nlm.nih.gov/, accessed on 25 May 2022) Reference Sequences (NC_052198.1, NC_010447.5, NC_009149.3, NC_000081.7, NC_037332.1, NC_030812.1, NC_052198.1, NC_000012.12, and NC_007134.7, respectively).
3. Results
3.1. Accuracy of GBTS
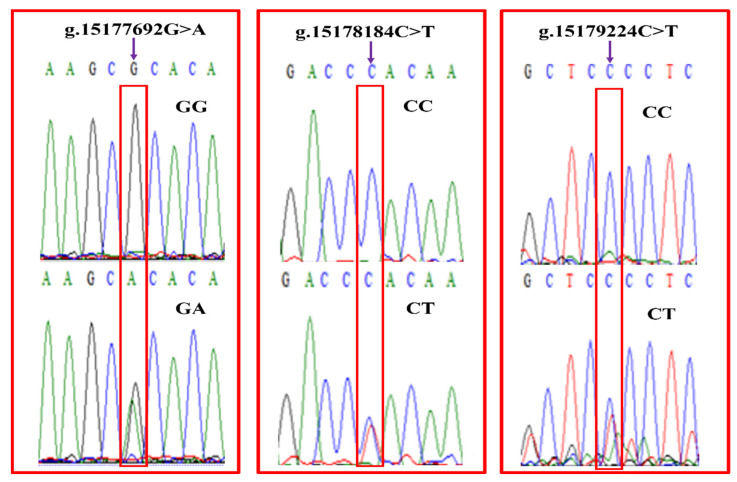
The sequencing profiles of the three mutant loci are shown in Figure 1. The results indicated that the sequencing results of the three mutant loci matched the sequencing results of GBTS, and the genotypes were consistent with the liquid-phase microarray typing results.
Figure 1.
Sequencing profiles around three mutation loci in the HOXC8 of Dezhou donkeys. The upper panels represent the reference genome, and the lower panels represent the variant.
3.2. Analysis of HOXC8 Gene Polymorphisms and Population Genetics in Dezhou Donkey
By mapping to the known Dezhou donkey gene sequence (GenBank NC_052198.1), 12 mutant loci were identified in the HOXC8 gene, and all loci were novel. The mutation information for the HOXC8 gene is summarized in Table 2. The alignment analysis of HOXC8 revealed 10 SNPs downstream and 2 in the introns. Among the 12 mutant loci, non-diallelic loci and those with genotypic typing failure were filtered out, leaving 4 mutant loci for subsequent analysis (g.15177692G>A, g.15178184C>T, g.15179224C>T, g.15179674G>A). Population genetic analysis was performed on four SNPs of the HOXC8 gene. The results revealed that all four loci (g.15177692G>A, g.15178184C>T, g.15179224C>T, g.15179674G>A) had low polymorphism (PIC < 0.25) in Dezhou donkeys (Table 3). In addition, the chi-square test indicated that the other three loci of HOXC8 were in Hardy-Weinberg equilibrium (p < 0.05), except for the g.15179674G>A locus (p > 0.05).
Table 2.
The information of 12 mutation loci in the HOXC8 gene of Dezhou donkeys.
| Region | Genomic Location | Wild-Type | Mutant | Mutation in Ensembl Database | Mutation in NCBI Database | Mutation in European Variation Archive | AA Coding Residue | Amino Acid Change |
|---|---|---|---|---|---|---|---|---|
| Downstream | 15177467 | G | A | NO | NO | NO | - | - |
| 15177692 | G | A | NO | NO | NO | - | - | |
| 15177975 | T | TATTA | NO | NO | NO | - | - | |
| 15178184 | C | T | NO | NO | NO | - | - | |
| 15178385 | C | G | NO | NO | NO | - | - | |
| 15178428 | C | T | NO | NO | NO | - | - | |
| 15178486 | C | T | NO | NO | NO | - | - | |
| 15178804 | A | G | NO | NO | NO | - | - | |
| 15178804 | A | GG | NO | NO | NO | - | - | |
| 15178806 | G | C | NO | NO | NO | - | - | |
| Intron | 15179224 | C | T | NO | NO | NO | - | - |
| 15179674 | G | A | NO | NO | NO | - | - |
Table 3.
Population genetic analysis of 4 SNPs of the HOXC8 mutation in Dezhou donkeys.
| Site | Location | Genotype Frequency |
Allele Frequency |
PIC | Ne | Ho | H | He | HWE | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chi-Square | p Value | ||||||||||||
| g.15177692G>A | Downstream (dist = 1137) |
GG (393) |
GA (7) |
AA (0) |
G | A | 0.018 | 1.018 | 0.983 | 0.050 | 0.017 | 0.031 | 0.860 |
| 0.983 | 0.017 | 0.000 | 0.991 | 0.009 | |||||||||
| g.15178184C>T | Downstream (dist = 645) |
CC (373) |
CT (27) |
TT (0) |
C | T | 0.064 | 1.070 | 0.935 | 0.148 | 0.065 | 0.488 | 0.485 |
| 0.933 | 0.067 | 0.000 | 0.966 | 0.034 | |||||||||
| g.15179224C>T | Intron | CC (394) |
CT (6) |
TT (0) |
C | T | 0.014 | 1.015 | 0.985 | 0.044 | 0.015 | 0.023 | 0.880 |
| 0.985 | 0.015 | 0.000 | 0.993 | 0.007 | |||||||||
| g.15179674G>A | Intron | GG (373) |
GA (25) |
AA (2) |
G | A | 0.067 | 1.075 | 0.930 | 0.156 | 0.070 | 4.453 | 0.035 |
| 0.932 | 0.063 | 0.005 | 0.964 | 0.036 | |||||||||
3.3. Association Analysis
Association analysis between the SNPs and phenotypes revealed that the g.15179224C>T locus was significantly associated with CW (p < 0.01). It was also significantly associated with LL (p < 0.05). The g.15179674G>A locus was significantly associated with LN (p < 0.05). More information is shown in Table 4. The g.15179224C>T locus was not observed as a mutant homozygote. At this locus, the CW of wild homozygotes of Dezhou donkeys was significantly higher than that of mutant heterozygotes (p < 0.01). Its LL was considerably higher than mutant heterozygous genotype individuals (p < 0.05). It indicates that mutation at this locus may be detrimental to the increase of CW and LL. There were three genotypes of the g.15179674G>A locus. The LN was significantly higher in individuals with mutant homozygotes than in wild homozygotes and mutant heterozygotes (p < 0.05). It indicates that mutations at this locus may be conducive to increased LN. Analysis of the association between thoracolumbar types and phenotypic traits showed that thoracolumbar types were significantly correlated with carcass weight. These individuals with T17L5 showed minimal values. The detailed information is shown in Table 5.
Table 4.
LSMs with standard errors of each phenotype for different genotypes of 4 SNPs in Dezhou donkeys.
| Site | Genotype | BH/cm | BL/cm | CC/cm | HW/kg | CW/kg | LN | LL/cm | TN | TL/cm | TLN |
|---|---|---|---|---|---|---|---|---|---|---|---|
| g.15177692G>A | GG (393) |
134.81 ± 0.26 | 132.55 ± 0.31 | 144.85 ± 0.26 | 24.15 ± 0.14 | 151.32 ± 0.97 | 5.21 ± 0.02 | 24.12 ± 0.11 | 17.86 ± 0.02 | 72.81 ± 0.18 | 23.07 ± 0.02 |
| GA (7) |
135.71 ± 1.91 | 132.43 ± 2.32 | 146.43 ± 1.92 | 24.04 ± 1.07 | 155.29 ± 7.25 | 5.14 ± 0.15 | 23.57 ± 0.81 | 18.00 ± 0.14 | 73.14 ± 1.36 | 23.14 ± 0.13 | |
| g.15178184C>T | CC (373) |
134.87 ± 0.26 | 132.60 ± 0.32 | 144.87 ± 0.26 | 24.12 ± 0.15 | 151.43 ± 0.99 | 5.21 ± 0.02 | 24.09 ± 0.11 | 17.86 ± 0.02 | 72.83 ± 0.19 | 23.07 ± 0.02 |
| CT (27) |
134.22 ± 0.97 | 131.89 ± 1.18 | 144.98 ± 0.98 | 24.51 ± 0.54 | 150.83 ± 3.69 | 5.22 ± 0.08 | 24.30 ± 0.41 | 17.85 ± 0.07 | 72.63 ± 0.69 | 23.07 ± 0.07 | |
| g.15179224C>T | CC (394) |
134.85 ± 0.25 | 132.59 ± 0.31 | 144.89 ± 0.26 | 24.17 ± 0.14 | 151.87 ± 0.95 A | 5.21 ± 0.02 | 24.14 ± 0.11 a | 17.86 ± 0.02 | 72.86 ± 0.18 | 23.07 ± 0.02 |
| CT (6) |
133.33 ± 2.06 | 129.83 ± 2.50 | 144.00 ± 2.08 | 22.57 ± 1.15 | 119.92 ± 7.67 B | 5.17 ± 0.17 | 22.25 ± 0.87 b | 17.83 ± 0.16 | 70.33 ± 1.47 | 23.00 ± 0.14 | |
| g.15179674G>A | GG (373) |
134.96 ± 0.26 | 132.72 ± 0.32 | 144.93 ± 0.26 | 24.15 ± 0.15 | 151.69 ± 0.99 | 5.21 ± 0.02 a | 24.10 ± 0.11 | 17.86 ± 0.02 | 72.81 ± 0.19 | 23.07 ± 0.02 |
| GA (25) |
133.16 ± 1.01 | 130.08 ± 1.22 | 144.38 ± 1.02 | 23.91 ± 0.57 | 146.78 ± 3.84 | 5.20 ± 0.08 a | 23.79 ± 0.42 | 17.88 ± 0.08 | 73.03 ± 0.74 | 23.08 ± 0.07 | |
| AA (2) |
131.00 ± 3.56 | 131.00 ± 4.33 | 142.00 ± 3.60 | 22.15 ± 2.00 | 152.50 ± 13.56 | 6.00 ± 0.29 b | 27.50 ± 1.50 | 17.50 ± 0.27 | 72.50 ± 2.55 | 23.50 ± 0.24 | |
| overall average of phenotypic data | 134.90 ± 0.25 | 132.59 ± 0.31 | 144.97 ± 0.26 | 24.18 ± 0.14 | 152.01 ± 0.89 | 5.21 ± 0.02 | 24.11 ± 0.11 | 17.86 ± 0.02 | 72.89 ± 0.18 | 23.07 ± 0.02 |
Note: Different letters for the groups indicate difference: (a, b) indicate differences (p < 0.05); (A, B) indicate differences (p < 0.01). BH, body height; BL, body length; CC, chest circumference; HW, hide weight; CW, carcass weight; LN, number of lumbar vertebrae; LL, lumbar vertebrae length; TN, number of thoracic vertebrae; TL, thoracic vertebrae length; TLN, total number of thoracic and lumbar vertebrae.
Table 5.
Means with standard error of each phenotype for different thoracolumbar types of Dezhou donkeys.
| Type of Thoracolumbar | Number | BH/cm | BL/cm | CC/cm | HW/kg | CW/kg | Proportion |
|---|---|---|---|---|---|---|---|
| T17L5 | 10 | 131.90 ± 1.72 | 129.30 ± 1.41 | 141.10 ± 1.56 | 23.45 ± 0.55 | 127.20 ± 14.56 a | 2.50% |
| T17L6 | 50 | 134.64 ± 0.64 | 132.10 ± 0.72 | 144.48 ± 0.68 | 23.77 ± 0.38 | 150.09 ± 2.02 b | 12.50% |
| T18L5 | 301 | 134.90 ± 0.30 | 132.56 ± 0.36 | 145.09 ± 0.30 | 24.22 ± 0.17 | 152.25 ± 1.07 b | 75.25% |
| T18L6 | 34 | 135.38 ± 0.79 | 134.15 ± 0.98 | 144.81 ± 0.64 | 24.34 ± 0.47 | 152.71 ± 2.36 b | 8.50% |
| T19L5 | 5 | 134.60 ± 3.12 | 132.20 ± 3.85 | 144.40 ± 2.50 | 23.68 ± 1.17 | 151.50 ± 6.27 b | 1.25% |
Note: Different letters for the groups indicate difference: (a, b) indicate differences (p < 0.05). BH, body height; BL, body length; CC, chest circumference; HW, hide weight; CW, carcass weight.
3.4. Construction of Phylogenetic Tree of HOXC8 Gene for Dezhou Donkey and Other Seven Species of Animals
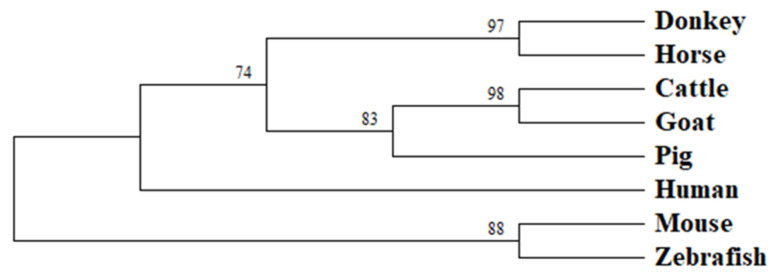
To investigate the variation of the HOXC8 gene in different species, a phylogenetic tree of the HOXC8 gene was constructed by the neighbor-joining method, and 1000 bootstrap replications were performed, showing that donkeys and horses were most closely related (Figure 2). The donkey, horse, cattle, goat, pig, and human sequences were clustered together and differed significantly from those of other breeds. The results showed that the HOXC8 gene was highly conserved in mammals. The phylogenetic tree indicated that the HOXC8 gene was highly conserved during animal evolution but differed considerably among distantly related species.
Figure 2.
The phylogenetic tree of the HOXC8 gene in different species. The number on the internal node represents support value and is used to represent the reliability of this branch structure.
4. Discussion
Numerous studies have shown that the HOXC8 gene was essential for skeletal morphogenesis, hematopoiesis, and cartilage differentiation during embryonic development [31,32]. Meanwhile, HOXC8 was also involved in regulating the development and progression of various human malignancies, such as gastric and breast cancer [33,34,35]. In addition, the HOXC8 gene has been associated with vertebral development in mice [36], myogenesis in Korean Hanwoo cattle [37], kyphosis and arthrogryposis multiplex congenital (AMC) in pigs [38,39], and the multi-vertebral number trait in Mongolian sheep [40]. HOXC8 was also widely expressed in the muscle, reproductive, and digestive systems in adult Dezhou donkeys [41]. However, no studies have been conducted on the HOXC8 polymorphism in donkeys. Therefore, we explored the association between the polymorphism of the HOXC8 gene and multi-vertebral number and carcass weight traits in donkeys.
In this study, we undertook an association study of four mutant loci in 400 individual Dezhou donkeys and identified two SNPs associated with CW, LL, and LN traits at the whole gene level (p < 0.05). Here, we presented four SNPs associated with each attribute (Table 4). The g.15179224C>T and g.15179674G>A were located in the introns of the HOXC8 gene. Only the genotype CC and CT were present at g.15179224C>T. Individuals with the CT genotype had significantly lower CW and LL than those with the CC genotype (p < 0.05), and BL, CC, HW, LN, and TL had the same downward trend but without reaching significant levels. Therefore, this locus was an adverse mutation for CW and LL. Consequently, it was speculated that the function of this locus would probably be to reduce BL by affecting LL and then to reduce CW by reducing BL. All three genotypes were present at g.15179674G>A, GG, GA, and AA, respectively. Individuals with the AA genotype had significantly higher LN than those with GG and GA genotypes (p < 0.05). In contrast, BH, CC, and HW had the opposite trend, though none reached a significant level. Therefore, the mutation at this locus was a favorable mutation for LN, which was unfavorable for BH, CC, and HW. Since the mutation sites affecting CW, LL and LN in Dezhou donkeys were all located on introns, their specific regulatory mechanisms were unclear. Studies have shown that mutations on introns cause alterations in splice variants [42,43], so HOXC8 gene expression might be regulated by the variable shear mechanism. The g.15179224C>T and g.15179674G>A sites may alter the binding of shear factor binding proteins to target sequences, leading to the appearance of phenotypic variants. Furthermore, the association of the HOXC8 gene with CW in Dezhou donkeys might be related to adipose differentiation and lipid deposition during livestock development and the regulation of LL and LN by the HOXC8 gene [18,44,45]. The correlation of the HOXC8 gene with LL and LN was possibly attributed to the differential expression of the HOXC8 gene during skeletal differentiation.
Analysis of the phylogenetic tree showed that the Dezhou donkey was most closely related to horses, with the sequences of donkey, horse, cattle, goat, pig, and humans all clustered together. According to the findings, the HOXC8 gene was highly conserved throughout animal evolution, which was in line with the previous conclusion of Krumlauf [46]. However, zebrafish did not cluster with them, probably due to the significant genetic sequence differences caused by the distant affinities.
Though the HOXC8 gene polymorphism of the Dezhou donkey was studied for the first time, some limitations to our study should be highlighted. Several studies have confirmed that HOXC8 was not a single gene that influenced the developmental process of the vertebra but most likely in combination with other genes or regulatory elements [47]. In contrast, we only investigated the effect of HOXC8 single gene polymorphism on multiple vertebral numbers in Dezhou donkeys, and the study of genetic mechanisms was not comprehensive enough. In addition, the results may have been underestimated due to sample size constraints. Therefore, more mutant loci, gene interactions, and experimental validation with larger sample sizes should be evaluated to understand better the effects of genetic polymorphisms on multi-vertebral numbers and carcass weight traits. In the meantime, it is worth exploring whether the increase in the number or length of thoracolumbar vertebrae may have some unintended negative consequences. Based on current studies in domestic animals, no direct clinical signs of increased number or length of thoracolumbar vertebrae have been reported, and specific welfare issues need to be explored.
5. Conclusions
In this study, we identified 12 mutant loci on the HOXC8 gene, and all of them were novel mutant loci. The types of thoracolumbar vertebrae in Dezhou donkeys were associated with carcass weight traits. The polymorphisms of the HOXC8 gene were associated with its multi-vertebral number and carcass weight traits. The g.15179224C>T and g.15179674G>A polymorphisms significantly affected CW, LL, and LN traits. In addition, the HOXC8 g.15179224C allele had a positive effect on CW and LL; the g.15179674G allele had a negative effect on LN. Therefore, g.15179224C>T was probably the restriction locus restricting the increment of CW and LL, and g.15179674G>A was a possible candidate gene for selection and breeding of Dezhou donkeys with multi-vertebra number. All these findings emphasized that the HOXC8 gene was an essential candidate gene impacting multi-vertebral number and carcass weight traits, providing helpful information for the marker-assisted breeding of Dezhou donkeys.
Author Contributions
Conceptualization, C.W.; methodology, Y.L.; software, T.W.; validation, T.W.; formal analysis, X.S.; investigation, T.W., W.R., B.H., X.W., Z.L., H.L., X.K., Y.C. and Y.W.; resources, C.W.; data curation, X.S. and W.R.; writing—original draft preparation, X.S. and Y.L.; writing—review and editing, Y.L. and F.A.; visualization, X.S.; supervision, C.W.; project administration, C.W.; funding acquisition, C.W. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Experiments and animal care in this study were approved by the Animal Welfare and Ethics Committee of Institute of Animal Sciences, Liaocheng University (No. LC2019-1).
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research was funded by National Natural Science Foundation of China (grant number 31671287), Taishan Leading Industry Talents-Agricultural Science of Shandong Province (grant number LJNY202022), Well-bred Program of Shandong Province (grant number 2017LZGC020), Shandong Province Modern Agricultural Technology System Donkey Industrial Innovation Team (grant number SDAIT-27) and National Natural Science Foundation of China (grant number 318051921).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Lai Z., Wu F., Zhou Z., Li M., Gao Y., Yin G., Yu J., Lei C., Dang R. Expression profiles and polymorphic identification of the ACSL1 gene and their association with body size traits in Dezhou donkeys. Arch. Anim. Breed. 2020;63:377–386. doi: 10.5194/aab-63-377-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lai Z., Li S., Wu F., Zhou Z., Gao Y., Yu J., Lei C., Dang R. Genotypes and haplotype combination of ACSL3 gene sequence variants is associated with growth traits in Dezhou donkey. Gene. 2020;743:144600. doi: 10.1016/j.gene.2020.144600. [DOI] [PubMed] [Google Scholar]
- 3.Wang C., Li H., Guo Y., Huang J., Sun Y., Min J., Wang J., Fang X., Zhao Z., Wang S., et al. Donkey genomes provide new insights into domestication and selection for coat color. Nat. Commun. 2020;11:6014. doi: 10.1038/s41467-020-19813-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.King J.W.B., Roberts R.C. Carcass length in the bacon pig; Its association with vertebrae numbers and prediction from radiographs of the young pig. Anim. Sci. 1960;2:59–65. doi: 10.1017/S0003356100033493. [DOI] [Google Scholar]
- 5.Huang J., Zhang M., Ye R., Ma Y., Lei C. Effects of increased vertebral number on carcass weight in PIC pigs. Anim. Sci. J. 2017;88:2057–2062. doi: 10.1111/asj.12881. [DOI] [PubMed] [Google Scholar]
- 6.Zhang Z., Sun Y., Du W., He S., Liu M., Tian C. Effects of vertebral number variations on carcass traits and genotyping of Vertnin candidate gene in Kazakh sheep. Asian-Australas. J. Anim. Sci. 2017;30:1234–1238. doi: 10.5713/ajas.16.0959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mehtiö T., Pitkänen T., Leino A.M., Mäntysaari E.A., Kempe R., Negussie E., Lindauer M.H. Genetic analyses of metabolic body weight, carcass weight and body conformation traits in Nordic dairy cattle. Animal. 2021;15:100398. doi: 10.1016/j.animal.2021.100398. [DOI] [PubMed] [Google Scholar]
- 8.Whaley J.R., Means W.J., Ritten J.P., Murphy T.W., Gifford C.L., Cunningham-Hollinger H.C., Woodruff K.L., McKibben H.N., Page C.M., Stewart W.C. Harvest season, carcass weight, and fat measurement effects on lamb carcass characteristics and economic comparison of moderate and heavyweight lamb carcasses in the Western lamb processing industry. Transl. Anim. Sci. 2020;4:S27–S31. doi: 10.1093/tas/txaa091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hwang Y.H., Lee S.J., Lee E.Y., Joo S.T. Effects of carcass weight increase on meat quality and sensory properties of pork loin. J. Anim. Sci. Technol. 2020;62:753–760. doi: 10.5187/jast.2020.62.5.753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rohrer G.A., Nonneman D.J., Wiedmann R.T., Schneider J.F. A study of vertebra number in pigs confirms the association of certain and reveals additional QTL. BMC Genet. 2015;16:129. doi: 10.1186/s12863-015-0286-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li J., Wang L., Yu D., Hao J., Zhang L., Adeola A.C., Mao B., Gao Y., Wu S., Zhu C., et al. Single-cell RNA Sequencing Reveals Thoracolumbar Vertebra Heterogeneity and Rib-genesis in Pigs. Genom. Proteom. Bioinform. 2021;19:423–436. doi: 10.1016/j.gpb.2021.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang Y., Cai H., Luo X., Ai Y., Jiang M., Wen Y. Insight into unique somitogenesis of yak (Bos grunniens) with one additional thoracic vertebra. BMC Genom. 2020;21:201. doi: 10.1186/s12864-020-6598-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jianmei Y., Yongli W., Zhengping F., Mingling K., Xiaoyun G., Deke A. Correlation Analysis for Beef Performance and Multi-vertebra Properties of Jinchuan Yak. Acta Ecol. Anim. Domastici. 2015;36:26–30. [Google Scholar]
- 14.Siwu G. Master’s Thesis. Hebei University of Engineering; Wuhan, China: 2021. Screening of Important Candidate SNPs Related to Carcass Traits in Sheep. [Google Scholar]
- 15.Mikawa S., Hayashi T., Nii M., Shimanuki S., Morozumi T., Awata T. Two quantitative trait loci on Sus scrofa chromosomes 1 and 7 affecting the number of vertebrae. J. Anim. Sci. 2005;83:2247–2254. doi: 10.2527/2005.83102247x. [DOI] [PubMed] [Google Scholar]
- 16.Li C., Zhang X., Cao Y., Wei J., You S., Jiang Y., Cai K., Wumaier W., Guo D., Qi J., et al. Multi-vertebrae variation potentially contribute to carcass length and weight of Kazakh sheep. Small Rumin. Res. 2017;150:8–10. doi: 10.1016/j.smallrumres.2017.02.021. [DOI] [Google Scholar]
- 17.Jamdar M.N., Ema A.N. A Note on the Vertebral Formula of the Donkey. Br. Vet. J. 1982;138:209–211. doi: 10.1016/S0007-1935(17)31084-9. [DOI] [PubMed] [Google Scholar]
- 18.Liu Z., Gao Q., Wang T., Chai W., Zhan Y., Akhtar F., Zhang Z., Li Y., Shi X., Wang C. Multi-Thoracolumbar Variations and NR6A1 Gene Polymorphisms Potentially Associated with Body Size and Carcass Traits of Dezhou Donkey. Animals. 2022;12:1349. doi: 10.3390/ani12111349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wellik D.M. Hox patterning of the vertebrate axial skeleton. Dev. Dyn. 2007;236:2454–2463. doi: 10.1002/dvdy.21286. [DOI] [PubMed] [Google Scholar]
- 20.Taotao L., Meilin J., Xiaojuan F., Huihua W., Jian L., Ran D., Caihong W. The Hox Gene Family and Its Effects on Spine Formation in Animals. Acta Vet. Zootech. Sin. 2022;53:999–1009. [Google Scholar]
- 21.Van Den Akker E., Fromental-Ramain C., de Graaff W., Le Mouellic H., Brûlet P., Chambon P., Deschamps J. Axial skeletal patterning in mice lacking all paralogous group 8 Hox genes. Development. 2001;128:1911–1921. doi: 10.1242/dev.128.10.1911. [DOI] [PubMed] [Google Scholar]
- 22.Jing Z., Liling Z., Qi C., Batu Relationship between methylation of Hoxc8 gene and the numbers of thoracic vertebrae in Mongolia sheep. Heilongjiang Anim. Sci. Vet. 2011;5:5–8. doi: 10.13881/j.cnki.hljxmsy.2011.05.015. [DOI] [Google Scholar]
- 23.Jiao T., Yili L., Shuqiong X., Shilong M., Mingfeng J. Advances in Candidate Genes on Vertebral Number Trait of Pig. Chin. J. Anim. Sci. 2022;58:92–98. doi: 10.19556/j.0258-7033.20210813-02. [DOI] [Google Scholar]
- 24.Zhao B., Pan Y., Qiao L., Liu J., Yang K., Liang Y., Liu W. miR-301a inhibits adipogenic differentiation of adipose-derived stromal vascular fractions by targeting HOXC8 in sheep. Anim. Sci. J. 2021;92:e13661. doi: 10.1111/asj.13661. [DOI] [PubMed] [Google Scholar]
- 25.Houghton L., Rosenthal N. Regulation of a muscle-specific transgene by persistent expression of Hox genes in postnatal murine limb muscle. Dev. Dyn. 1999;216:385–397. doi: 10.1002/(SICI)1097-0177(199912)216:4/5<385::AID-DVDY7>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 26.Zhang Z., Zhan Y., Han Y., Liu Z., Wang Y., Wang C. Estimation of Liveweight from Body Measurements through Best Fitted Regression Model in Dezhou Donkey Breed. J. Equine Vet. Sci. 2021;101:103457. doi: 10.1016/j.jevs.2021.103457. [DOI] [PubMed] [Google Scholar]
- 27.Shi Y.Y., He L. SHEsis, a powerful software platform for analyses of linkage disequilibrium, haplotype construction, and genetic association at polymorphism loci. Cell Res. 2005;15:97–98. doi: 10.1038/sj.cr.7310101. [DOI] [PubMed] [Google Scholar]
- 28.Nei M., Roychoudhury A.K. Sampling variances of heterozygosity and genetic distance. Genetics. 1974;76:379–390. doi: 10.1093/genetics/76.2.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Botstein D., White R.L., Skolnick M., Davis R.W. Construction of a genetic linkage map in man using restriction fragment length polymorphisms. Am. J. Hum. Genet. 1980;32:314–331. [PMC free article] [PubMed] [Google Scholar]
- 30.Kumar S., Stecher G., Li M., Knyaz C., Tamura K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018;35:1547–1549. doi: 10.1093/molbev/msy096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kruger C., Kappen C. Expression of cartilage developmental genes in Hoxc8- and Hoxd4-transgenic mice. PLoS ONE. 2010;5:e8978. doi: 10.1371/journal.pone.0008978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yueh Y.G., Gardner D.P., Kappen C. Evidence for regulation of cartilage differentiation by the homeobox gene Hox-8. Proc. Natl. Acad. Sci. USA. 1998;95:9956–9961. doi: 10.1073/pnas.95.17.9956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gu H., Zhong Y., Liu J., Shen Q., Wei R., Zhu H., Zhang X., Xia X., Yao M., Ni M. The Role of miR-4256/HOXC8 Signaling Axis in the Gastric Cancer Progression: Evidence From lncRNA-miRNA-mRNA Network Analysis. Front. Oncol. 2021;11:793678. doi: 10.3389/fonc.2021.793678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xu P., Zhang X., Ni W., Fan H., Xu J., Chen Y., Zhu J., Gu X., Yang L., Ni R., et al. Upregulated HOXC8 Expression Is Associated with Poor Prognosis and Oxaliplatin Resistance in Hepatocellular Carcinoma. Dig. Dis. Sci. 2015;60:3351–3363. doi: 10.1007/s10620-015-3774-x. [DOI] [PubMed] [Google Scholar]
- 35.Shah M., Cardenas R., Wang B., Persson J., Mongan N.P., Grabowska A., Allegrucci C. HOXC8 regulates self-renewal, differentiation and transformation of breast cancer stem cells. Mol. Cancer. 2017;16:38. doi: 10.1186/s12943-017-0605-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lei H., Juan A.H., Kim M.S., Ruddle F.H. Identification of a Hoxc8-regulated transcriptional network in mouse embryo fibroblast cells. Proc. Natl. Acad. Sci. USA. 2006;103:10305–10309. doi: 10.1073/pnas.0603552103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.De Las Heras-Saldana S., Chung K.Y., Lee S.H., Gondor C. Gene expression of Hanwoo satellite cell differentiation in longissimus dorsi and semimembranosus. BMC Genom. 2019;20:156. doi: 10.1186/s12864-019-5530-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lindholm-Perry A.K., Rohrer G.A., Kuehn L.A., Keele J.W., Holl J.W., Shackelford S.D., Wheeler T.L., Nonneman D.J. Genomic regions associated with kyphosis in swine. BMC Genet. 2010;11:112. doi: 10.1186/1471-2156-11-112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Genini S., Nguyen T.T., Malek M., Talbot R., Gebert S., Rohrer G., Nonneman D., Stranzinger G., Vögeli P. Radiation hybrid mapping of 18 positional and physiological candidate genes for arthrogryposis multiplex congenital on porcine chromosome 5. Anim. Genet. 2006;37:239–244. doi: 10.1111/j.1365-2052.2006.01447.x. [DOI] [PubMed] [Google Scholar]
- 40.Qi C., Jing Z., Ling Z., Yuehui M. Methylation analysis of Hoxc8 exon-1 in multi-vertebrate Mongolian sheep. Chin. J. Anim. Sci. 2009;45:10–14. [Google Scholar]
- 41.Wang Y., Miao X., Zhao Z., Wang Y., Li S., Wang C. Transcriptome Atlas of 16 Donkey Tissues. Front. Genet. 2021;12:682734. doi: 10.3389/fgene.2021.682734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yan S.Q., Hou J.N., Bai C.Y., Jiang Y., Zhang X.J., Ren H.L., Sun B.X., Zhao Z.H., Sun J.H. A base substitution in the donor site of intron 12 of KIT gene is responsible for the dominant white coat colour of blue fox (Alopex lagopus) Anim. Genet. 2014;45:293–296. doi: 10.1111/age.12105. [DOI] [PubMed] [Google Scholar]
- 43.Fang G., Guojing L., Zhihua J., Xiuge W., Jinming H., Yinxue X. Association of SPEF2 gene splices variant and functional SNP with semen quality traits in Chinese Holstein bulls. J. Nanjing Agric. Univ. 2014;37:119–125. doi: 10.7685/j.issn.1000-2030.2014.03.018. [DOI] [Google Scholar]
- 44.Karastergiou K., Fried S.K., Xie H., Lee M.J., Devaux A., Rosencrantz M.A., Chang R.J., Smith S.R. Distinct developmental signatures of human abdominal and gluteal subcutaneous adipose tissue depots. J. Clin. Endocrinol. Metab. 2013;98:362–371. doi: 10.1210/jc.2012-2953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yamamoto Y., Gesta S., Lee K.Y., Tran T.T., Saadatirad P., Kahn C.R. Adipose depots possess unique developmental gene signatures. Obesity. 2010;18:872–878. doi: 10.1038/oby.2009.512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Krumlauf R. Hox genes, clusters and collinearity. Int. J. Dev. Biol. 2018;62:659–663. doi: 10.1387/ijdb.180330rr. [DOI] [PubMed] [Google Scholar]
- 47.Juan A.H., Ruddle F.H. Enhancer timing of Hox gene expression: Deletion of the endogenous Hoxc8 early enhancer. Development. 2003;130:4823–4834. doi: 10.1242/dev.00672. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.