Abstract
The release of cytokines from epithelial and stromal cells is critical for the initiation and maintenance of tissue immunity. Three such cytokines, TSLP, IL-33, and IL-25, are important regulators of type 2 immune responses triggered by parasitic worms and allergens. In particular, these cytokines activate group 2 innate lymphoid cells (ILC2s), Th2 cells, and myeloid cells that drive hallmarks of type 2 immunity. However, emerging data indicates that these tissue-associated cytokines are not only involved in canonical type 2 responses, but are also important in the context of viral infections, cancer, and even homeostasis. Here we provide a brief review of the roles of TSLP, IL-33, and IL-25 in diverse immune contexts, while highlighting their relative contributions in tissue-specific responses. We also emphasize a biologically-motivated framework for thinking about the integration of multiple immune signals, including the three featured in this review.
Keywords: cytokines, tissue immunity, allergy, helminth, cancer, viral infection
INTRODUCTION
Parasitic worms (helminths) and allergens elicit a type 2 immune response characterized by profound remodeling of barrier tissue composition and function. Examples of such changes include increased fluid and mucus production, smooth muscle hypercontractility, goblet and tuft cell hyperplasia, and fibroblast remodeling. Understanding and treating type 2 inflammatory diseases therefore requires dissecting the signals that initiate and regulate changes in barrier tissues. IL-25, IL-33, and TSLP were among the first cytokines shown to activate group 2 innate lymphoid cells (ILC2s) and they have been conceptually grouped together ever since. However, these cytokines are structurally and functionally unique and belong to distinct cytokine families. The importance of these cytokines in type 2 immunity is well established and extends beyond their role in ILC2 biology, as they are key regulators of T helper 2 (Th2) cells, mast cells, basophils, dendritic cells (DCs) and more. Yet the mechanisms that control the production and release of these cytokines are not unique to type 2 immunity. In particular, IL-33, which is released by tissue damage, and TSLP, which is induced by NFkB, broadly monitor and regulate tissue homeostasis. As such, it is perhaps unsurprising that TSLP, IL-33, and IL-25 have been implicated in contexts not classically associated with type 2 immunity, including viral infections and cancer. Recent reviews have comprehensively described this “holy trinity” of cytokines in type 2 immunity(1) (2, 3). Here, we emphasize their tissue specificity and function in type 2 immunity and in contexts not classically associated with type 2 inflammation.
TSLP
Background
Thymic stromal lymphopoietin (TSLP) is a member of the 4-helix bundle cytokine family and a distant paralog of interleukin 7 (IL-7)(4). Like other 4-helix bundle cytokines including the common γ chain cytokines, TSLP acts to shape the activities of adaptive and innate immune cells, notably T cells and DCs(5). As the name suggests, TSLP was first identified in the supernatant of a mouse thymic stromal cell line and was shown to support immature B cell proliferation and development(6–8). TSLP is expressed primarily by epithelial and stromal cells at barrier surfaces, such as the skin, gut, and lung, while myeloid cells express high levels of the receptor(9, 10). TSLP expression and release from epithelial cells, as well as other cell types such as fibroblasts and smooth muscle cells, is generally NFkB-dependent and increases in response to a broad array of stimuli including mechanical injury, infection, inflammatory cytokines, and proteases such as trypsin and papain(11–15).
In 2000, several groups isolated a low-affinity TSLP-binding protein in both humans and mice (referred to as TSLP receptor (TSLPR))(16–19). Subsequent studies showed that the functional, high-affinity TSLP receptor complex is a heterodimer of TSLPR and IL-7 receptor alpha (IL-7Rα) that signals through JAK1/2 and STAT5 (hematopoietic cells) or STAT3 (structural cells) (Figure 1)(16, 17). TSLP and TSLPR homologs have been identified in fish, but cross-species homology for both the cytokine and its receptor is relatively low (~40% between mice and humans), although functionally they appear to be quite similar(20–22). Thus, the role of TSLP is conserved at least between man and mouse despite limited sequence identity.
Figure 1. TSLP, IL-33, and IL-25 contribute to both canonical type 2 immunity and diseases not typically associated with type 2.
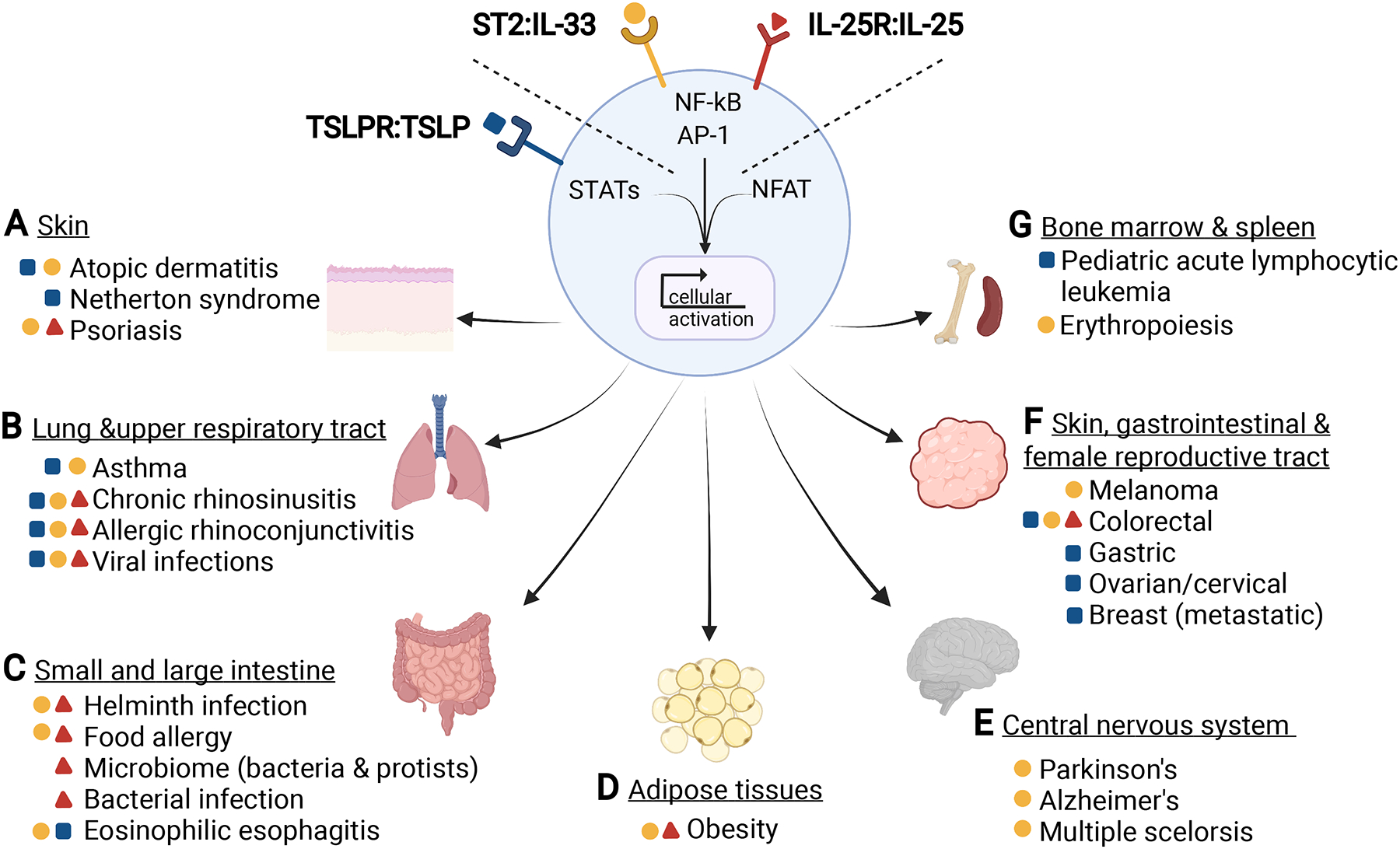
(A) – (G) Shown is a summary of TSLP (blue square), TSLP receptor (blue receptor), IL-33 (yellow circle), ST2 (yellow receptor), IL-25 (red triangle), IL-25R (red receptor) signaling pathways and the tissues and diseases in which these cytokines have been implicated. These cytokines and their receptors can be grouped based on the transcription factors they activate. When combined with NFAT-inducing signals, these cytokines can activate all the major transcription factors required for lymphocyte survival and function.
TSLP and type 2 immunity
TSLP has been broadly implicated in the pathogenesis of type 2 inflammatory diseases. Beginning with a seminal analysis of patients with atopic dermatitis, TSLP expression was found to be markedly increased in lesional skin biopsies, but not as highly expressed in adjacent healthy tissue (Figure 1A)(23, 24). Following this discovery, TSLP genetic variants and high levels of TSLP expression have been associated with other type 2 diseases such as asthma, chronic rhinosinusitis, allergic rhinoconjunctivitis, and eosinophilic esophagitis (Figure 1B and 1C)(25–29). For example, a single-nucleotide polymorphism in the TSLP promoter was linked to asthma in multiple cohorts; biopsy studies have revealed significantly increased TSLP expression in the airway epithelium and lamina propria from asthma patients(30). TSLP overexpression has also been reported in Netherton syndrome, a genetic disease caused by mutations in keratinocyte SPINK5 gene that manifests in type 2 inflammation at multiple sites (Figure 1A)(31).
Mouse studies have further demonstrated the importance of TSLP in models of type 2 inflammation, including OVA/alum immunization and challenge, eosinophilic esophagitis, and atopic dermatitis (Figure 1C)(32–35). However, there are also examples of type 2 inflammation in which the contribution of TSLP is minimal, particularly in the lung and intestine, underscoring the tissue- and context-specificity of innate type 2 cytokines(36–38). Overall, TSLP appears to be more important for the initiation of type 2 immune responses rather than for their maintenance in the tissue. Consistent with this idea, Khodoun et al. showed that prior administration of blocking antibodies to TSLP, as well as IL-25 or IL-33, could prevent onset of allergic responses to medium chain triglycerides plus egg white(39). However, blockade of single cytokines had no effect on established disease in this context. In addition, TSLP is critical for the priming of type 2 immunity. TSLP signaling in naïve CD4 T cells, in the presence of TCR stimulation, promotes Th2 differentiation and proliferation through induction of IL-4 gene transcription(40–42). Similarly, TSLP directly supports ILC2 proliferation.
TSLP and viral infections
The plethora of cell types that can produce or respond to TSLP demonstrates a potential role for this cytokine in inflammation beyond classical contexts of type 2 immunity. Indeed, in respiratory viral infections, TSLP functions across both type 1 and type 2 immunity. Several groups have shown that infection with respiratory syncytial virus (RSV) and rhinovirus (RV) induces TSLP secretion by bronchial epithelial cells, leading to a subsequent type 2 immune response(43–46). RSV or RV infection in infants is particularly likely to result in type 2 immunity, which has been implicated in the development of asthma in adolescents as well as asthma exacerbations in children and adults (Figure 1B)(46–48).
There are conflicting reports on the role of TSLP in the response to influenza virus infection. One study showed no effect of TSLP signaling, while subsequent work suggested TSLP signaling was important for CD8+ responses, both during acute infection and in memory responses(49–51). The effect of TSLP was shown to be both direct on CD8+ T cells and indirect through DCs. Surprisingly, type III interferon was shown to induce TSLP expression following influenza infection, leading to increased adaptive mucosal immunity (52). Consistent with this observation, inclusion of TSLP increased the efficacy of influenza subunit vaccines when administered at mucosal surfaces(53). Taken as a whole, these data support a role for TSLP in influencing the response to respiratory viral infection through both type 2- and non-type 2-mediated mechanisms.
TSLP and cancer
Immune responses in the tumor microenvironment are highly variable and are largely dictated by factors produced by the tumor and other tumor-associated cells(54). For example, tumors displaying type 2 responses generally have a worse prognosis relative to tumors where type 1 responses predominate, but there are also examples where type 2 immunity is protective(55–59). An early study demonstrating TSLP production by the tumor strroma showed that patients with higher Th2 cellular infiltrate also had an increase of TSLP-producing cancer associated fibroblasts (CAFs), and that their tumors and tumor-draining lymph nodes contained TSLPR+ DCs(57). Additionally, tumor-derived IL-1α and IL-1β are required for TSLP expression by the CAFs, indicating that the tumor itself can induce TSLP(60). Subsequent studies in ovarian, gastric, and cervical tumors similarly found that TSLP-mediated cross-talk between stromal tumor cells and infiltrating hematopoietic cells can promote tumorigenesis (Figure 1F)(61–64).
In another example, the role of TSLP in metastatic breast cancer has been studied but remains unclear. In tumor tissue from breast cancer patients, tumor cell-derived TSLP was shown to promote Th2 differentiation of naïve CD4+ T cells by regulating DC maturation(65). In addition, DC-derived IL-1β is required for TSLP production by the tumor cells, such that IL-1 blockade, using therapeutics such as Anakinra, blocked tumor growth(66). Consistent with these findings, mouse models using an orthotopic model of metastatic breast cancer supported a role for tumor-derived TSLP to skew the tumor microenvironment towards type 2(67, 68). In contrast, one study showed that human breast tumor cell lines, as well as several primary tumors, lack TSLP expression, suggesting that TSLP signaling is not an important pathway in human breast cancer(69). A recent report that may serve to reconcile these opposing views of TSLP and breast cancer found that TSLP acts as a survival factor for breast tumor cells through induction of Bcl-2 and Bcl-xL expression(70). Tumor cells from breast cancer patients expressed TSLPR while the circulating monocytes and neutrophils expressed TSLP(70). Thus, TSLP expression by the tumor was not critical for growth, but TSLPR signaling within the tumor could drive metastatic tumor growth.
The function of TSLP is similarly complex in the gastrointestinal tract. Loss of TSLP or its receptor is associated with increased inflammatory disease (e.g. DSS-induced colitis), suggesting that TSLP could normally help prevent inflammation-induced tumorigenesis (71, 72). Once tumors have developed however, TSLP exacerbates disease by supporting TSLPR+ T regulatory cells in the tumor (Figure 1C)(73). Further study in mouse models of esophageal, gastric, intestinal, and colon cancers will be required to determine the tissue- and context-specific roles of TSLP in gastrointestinal tumors.
Finally, TSLP has been implicated in blood cancers. Pediatric acute lymphocytic leukemia (B-ALL) is a heterogeneous disease associated with a variety of genetic lesions, including mutations in the TSLP signaling pathway (Figure 1G)(74). Approximately 50% of the Philadelphia chromosome positive-like (Ph-like) patients have chromosomal rearrangements involving the TSLPR gene (also referred to as CRLF2), leading to increased TSLPR expression(75–77). In addition, a gain-of-function mutation in the coding sequence of TSLPR has been found in Ph-like B-ALL(78). These data make a compelling case for enhanced TSLPR signaling and the development of B-ALL. From a therapeutic perspective, CAR-T cells directed against TSLPR were effective at eliminating TSLPR+ B-ALL cells in a xenograft setting, suggesting that this modality could be effective in patients(79). In sum, while the presence of TSLP/TSLPR activity in a variety of human cancers is provocative, future studies are needed to obtain a clear view of the context-specific role of TSLP in cancer.
Emerging functions of TSLP
Since its association with atopic dermatitis, TSLP has been considered a type 2 promoting factor. However, as described in this review, it is becoming clear that TSLP is involved in a broad spectrum of immune responses. These include a role in germinal center responses and humoral immunity through its expression by follicular dendritic cells, as well as its role in B cell development in the bone marrow(80). The recent finding of a critical role for TSLP signaling in wound-induced hair growth due to lack of hair follicle stem cell expansion is an area of interest, as the hair follicle is being recognized as an important source of immune cells in the skin(81). Overall, recent studies are beginning to reveal novel roles for TSLP outside of the type 2 inflammatory milieu, and further inquiry will no doubt expand our understanding of the tissue-specific function of TSLP in multiple disease states.
IL-33
Background
Interleukin-33 (IL-33) is a nuclear cytokine and a member of the IL-1 family, which is an evolutionarily conserved cluster of cytokines and associated receptors that regulate barrier homeostasis and inflammation (82–84). Orthologues of ST2 (or IL-1 receptor-like-1, IL1RL1), part of the IL-33 receptor, exist in birds, fish and reptiles, while IL-33 evolved in mammals(85). The gene that encodes canine IL-33 (DVS27) was first identified in 1999, and in 2003, Girard and colleagues reported gene and protein expression of an ortholog in human high endothelial venules in multiple lymphoid tissues(83, 86). In 2005, Schmitz and colleagues identified the same protein in a sequence database search for novel IL-1 family members(87). Many non-hematopoietic cells constitutively express IL-33, including endothelial cells, epithelial cells, mesenchymal cells, and fibroblasts(82).
The IL-33 protein has a basic N-terminal chromatin binding domain and an acidic C-terminal IL-1 like domain(82, 83). Despite its nuclear localization, IL-33 does not appear to have significant effects on gene transcription or regulation, although further studies in this area are warranted(82). Instead, nuclear localization provides regulatory control by sequestering IL-33 inside the cell to prevent constitutive secretion(82, 88, 89). IL-33 is generally thought to be released from damaged cells as an alarmin, though a recent report has shown that IL-33 is secreted from DCs via perforin 2(90). Mobilization of a RIPK1-caspase 8 “ripoptosome” in epithelial cells has also been identified as a mechanism for release of IL-33. While full-length IL-33 has biological function, ripoptosome-dependent cleavage by caspase 3/7 or by extracellular serine proteases generates a more potent “mature” form(82, 91–93). Of note, secreted IL-33 can remain bound to chromatin, which can synergistically activate receptor-mediated signaling, suggesting an important role for the chromatin-binding activity of IL-33 in modulating IL-33 signal strength(89). Consistent with constitutive IL-33 expression and release by dying cells, elevated IL-33 levels can be detected in a wide range of human diseases, including asthma, atopic dermatitis, ulcerative colitis, allergic rhinitis, and rheumatoid arthritis(94).
IL-33 signals via a heterodimer formed by the IL-1 family receptor ST2 (IL1RL1) and the co-receptor IL-1 receptor accessory protein (IL-1RAcP) that is used by other members of the IL-1 cytokine family (Figure 1) (87, 95). Cells that express ST2 include tissue resident cells in the epithelium and stroma, mast cells, basophils, eosinophils, DCs, ILC2s, and T cells (82, 87, 95–98). ST2 binding results in a conformational change and IL1RAcP recruitment, leading to downstream recruitment of MyD88 and IL-1R-associated kinase to form the myddosome complex. The myddosome interfaces with TRAF6 to activate MAPKs and NF-κB(95). In ILC2s and other type 2 inflammation-associated cell types, IL-33 signaling also mobilizes phosphoinositide-3-kinase (PI3K)/protein kinase B (AKT) and mTOR, suggesting that IL-33 signaling pathways can be cell type-specific(99, 100).
IL-33 and type 2 immunity
Given the distribution of ST2 expression, IL-33 is poised to promote many aspects of type 2 immunity(95). For example, IL-33 drives eosinophil survival, expression of adhesion molecules, and release of pro-inflammatory cytokines and chemokines such as CXCL8 and CCL2(101). For ILC2s, which reside at epithelial surfaces such as lung, skin, gut, and adipose tissues, IL-33 causes robust IL-5 and IL-13 production that in turn supports immune and epithelial type 2 inflammatory functions(102, 103). IL-33 also activates macrophages and DCs to elicit type 2 immunity(104, 105). In the case of DCs, IL-33 exposure leads to upregulation of cell surface co-stimulatory molecules, like CD86 and MHC-II, along with IL-6 production, thereby generally enhancing DC ability to prime naive T cells and elicit type 2 cytokine production(105). In mature Th2 cells, IL-33 directly leads to initiation of type 2 cytokine production, with ST2 serving as an important marker of effector and memory Th2 cells in certain tissues(95). In an interesting example of context-specific IL-33 function, IL-33 derived from the intestinal epithelium induced type 2 protective immunity to helminth infection, while CD11c+ DC-derived IL-33 led to the expansion of ST2+ Tregs that maintain tissue homeostasis(90).
With these cellular effects, IL-33 is centrally placed to mediate key functions during type 2 inflammation in parasitic helminth infection and allergic disease(95, 106). IL-33 is important in resistance to parasitic helminth infection, inducing type 2 cytokines and activating cells that are critical for worm expulsion, but also promoting fibrosis in the context of chronic inflammation(95, 106–108). During allergic disease, IL-33 can be pro-inflammatory, initiating and exacerbating disease by acting on multiple cell types(95, 106, 109–112). For example, IL-33 promotes the pro-allergic functions of ILC2s and basophils in atopic dermatitis, mast cell infiltration of the airway epithelium, key effector processes in TSLP-driven models of the allergic march, and OX40L expression on ILC2s to promote adaptive responses to allergens and helminths (Figure 1A–C) (109–112).
In the context of metabolic homeostasis, IL-33 also supports type 2 immunity, but in this case, a type 2 immune tone has been shown to underlie metabolic health and maintenance of a lean state. IL-33 drives browning of white adipose tissue (WAT) by activating ILC2s and eosinophils, a process that facilitates thermogenesis and other metabolic processes associated with protection from obesity(Figure 1D)(102, 113–115). Here, IL-33 is likely derived from adipose stromal cells(116, 117). Thus, IL-33 is a key factor in the development and maintenance of type 2 inflammation in a variety of scenarios to either promote inflammation or maintain homeostasis.
IL-33 and viral infection
Like TSLP, IL-33 also contributes to immune responses during viral infection, although its functions are complex and context-dependent. Elevations in IL-33 levels have been reported to occur in multiple viral infections, likely due to lytic cell death(118). IL-33 can promote expansion of NK cell, CD8+ T cell, and Th1 cell populations, all of which are associated with protection against viral replication(95, 118–120). In particular, IL-33 derived from radio-resistant cells has been shown to play a key role in cytotoxic T lymphocyte activation in response to RNA and DNA virus infection as well as during vaccination, and IL-33 can support Th1 function during LCMV infection(119, 120). IL-33 also has potent effects on ST2+ Tregs that can suppress inflammation during cytomegalovirus infection or drive Treg-mediated tissue repair during influenza virus infection(121). In addition, as in type 2 inflammation, IL-33 stimulates ILC2s during viral infection, supporting tissue homeostasis and repair in some cases(122) and promoting allergic airway hyperreactivity in others (Figure 1C)(123). Of note, recent studies have suggested that increased levels of IL-33 are a predictor for progression to severe COVID-19, although further studies are required to dissect the function of IL-33 in various disease stages of SARS-CoV-2 infection(124). Overall, the effects of IL-33 during viral infection appear to be virus- and tissue-specific. Further studies using ex vivo human tissue and in vitro models that utilize human-derived cells are required to dissect the role of IL-33 in the effector and memory stage of the antiviral immune response.
IL-33 and cancer
As is true for type 2 immunity generally, the role of IL-33 in anti-tumor immune responses is controversial and varies depending on the type and tissue location of the tumor(125). Increased expression of IL-33 is found in many cancers, including human colorectal cancer(125). IL-33 can promote a type 2 inflammatory tone in the tumor microenvironment and can activate ST2+ Tregs and dampen anti-tumor NK cell responses to support tumor growth(126, 127). In contrast, the ability of IL-33 to stimulate CD8+ T cells, NK cells, DCs, eosinophils and ILC2s can convey anti-tumor function, with IL-33 eliciting responses that can hamper tumor growth, particularly synergizing with PD-1 inhibition to promote anti-tumor ILC2 responses in a mouse model of melanoma (Figure 1F)(128–131). Thus, the same is true for the role of IL-33 in cancer as it is during viral infection, and indeed, even type 2 inflammation: the effects of IL-33 are influenced by the spatial and temporal nature of the immune response.
Emerging functions of IL-33
Recent studies have revealed novel roles for IL-33 in the central nervous system, in the regulation of erythropoiesis, and during fibrosis. As regards the CNS, numerous neurological conditions are associated with increased IL-33 or ST2 expression, including Parkinson’s disease, multiple sclerosis (MS), traumatic brain injury (TBI), ischemic stroke, and Alzheimer’s disease (Figure 1E)(132). Exogenous IL-33 treatment can reduce the severity of experimental autoimmune encephalomyelitis (EAE), a mouse model of MS, with IL-33 promoting the mobilization of anti-inflammatory M2-like macrophages and ST2+ Tregs(133). IL-33 derived from oligodendrocytes and astrocytes underlies resistance to the protozoan parasite Toxoplasma gondii(134). Recent studies have shown that IL-33 also plays a significant role in synapse and neural circuit development, opening new avenues to study CNS development and remodelling(135, 136). IL-33 also appears to play a role in hemostasis (Figure 1G). Erythrocyte expression of IL-33 is associated with the development of mature red pulp macrophages (RMPs) that are responsible for erythrocyte maintenance and iron recycling, though it remains unclear where IL-33, a nuclear protein, localizes in an enucleated cell type(137). In addition, ST2 is expressed on erythroid progenitor cells and exogenous IL-33 suppresses erythropoiesis, leading to anemia in chronic inflammation(138). Finally, IL-33 contributes to fibrotic processes in skin, lung and epithelial tissues of patients with pulmonary fibrosis, pancreatic fibrosis, or fibrotic colitis(139). In the gut, IL-33-dependant fibrosis is considered to be protective during IBD(140), but the multifacted role of IL-33 in fibrosis remains to be fully appreciated. Together, these studies reveal exciting new roles for IL-33 in a variety of physiological processes during inflammation and homeostasis.
IL-25
Background
The IL-17 cytokine family is evolutionarily conserved across kingdoms, ranging from lampreys to humans, and is most often associated with Th17/Type 3 immunity that regulates epithelial, stromal, and immune cells to tune tissue function and inflammation(141–143). One IL-17 family member, IL-17E, is also known as IL-25 and has a unique ability to elicit type 2 immune responses within barrier tissues(142, 143). Originally described as a Th2-derived cytokine, Il25 transcript is expressed by both hematopoietic and nonhematopoietic cells, although functionally the protein is best characterized when secreted by keratinocytes and epithelial tuft cells. Indeed, murine tuft cells are the only airway and intestinal cells marked by an Il25 reporter(144, 145). Ex vivo stimulation of bone marrow cells revealed that mast cells can secrete IL-25, whereas in the lung, inhalation of particles leads to IL-25 production by alveolar macrophages(146–148). However, transfer of T cells, mast cells, or bone marrow cells from Il25-deficient mice still induces eosinophilia, suggesting that IL-25 derived from airway epithelial cells is responsible for the initiation of asthmatic lung inflammation(147, 148). In both acute and chronic allergic skin inflammation, keratinocytes are the predominant source of IL-25, but mast cells may also be a source of IL-25 in models of contact hypersensitivity(149, 150).
IL-25 signals via a heterodimeric receptor (IL-25R) composed of IL-17RB and IL-17RA, the latter of which is shared with receptors for other IL-17 family members(151). In most cells, IL-25R signaling leads to TRAF6- and Act1-dependent activation of NF-kB and AP-1 transcription factors (Figure 1)(151). Expression of the IL-25 receptor is more restricted than that of ST2 and TSLPR. Some IL-17RB can be detected on myeloid cells, granulocytes and certain T cell subsets, but expression is much higher on ILC2s, particularly in the small intestine, and on certain thymic subsets(152).
IL-25 and type 2 immunity
Systemic IL-25 elicits a robust type 2 immune response that can be detrimental if dysregulated. For example, injection of exogenous IL-25 results in an expansion of Th2 cells, ILC2s, and multipotent progenitor type-2 cells(153). As a result, these cells then produce type 2 cytokines IL-4, IL-5, IL-9, and IL-13 and promote eosinophilia and mucus secretion(153, 154). Germline overexpression of Il25 using transgenic mouse models leads to growth retardation, jaundice, and increased serum levels of type 2 cytokines and antibodies, IgE and IgG1(155). Chronic overexpression of Il25 specifically by airway epithelial cells causes asthmatic symptoms, while constitutive overexpression of intestinal Il25 leads to enhanced IL-13 production by ILC2s in response to food allergens(156, 157). In sum, these mouse studies suggest that IL-25 acts predominantly as a type 2 cytokine, as has been confirmed by many studies of tissue immunity. IL-25 has also been broadly linked to type 2 immunity in humans, with elevated levels detected in blood and/or tissue of patients with food allergy, asthma, chronic rhinosinusitis with nasal polyps (CRSwNP) and atopic dermatitis (Figure 1A–B)(158–162).
The role of IL-25 in type 2 immunity is best defined in the small intestine (Figure 1C). During acute models of helminth infection, germline IL-25 knockout mice inefficiently expel worms and have a delayed cytokine response(163). Similarly, Il25−/− mice challenged with chronic or secondary helminth infection have a delayed type 2 cytokine response, a defect in tissue remodeling, and impaired worm clearance(164). IL17RB-deficient mice are also unable to expel parasites(165). Tuft cells are the primary IL-25 producers in the gut; complete loss of tuft cells or epithelium-specific deletion of Il25 leads to defective goblet cell hyperplasia and compromised worm expulsion(145, 166). In both acute and chronic helminth infection, administration of exogenous IL-25 early in infection leads to rapid worm expulsion, suggesting that IL-25 is sufficient to initiate and amplify type 2 immune responses to helminths. Compared to the skin and lung, intestinal ILC2s have high expression of IL17RB and express lower levels of ST2(167). During acute helminth infection, tuft cell derived-IL25 drives ILC2 expansion and secretion of IL-13, promoting tissue remodeling and worm clearance(145, 166). Whether tuft cell-derived IL-25 signals to Th2s and/or other cells in a similar way is unknown, but IL-25 enhances the expansion and function of human memory Th2 cells in vitro(162). Finally, Il25−/− mice chronically infected with Trichuris muris develop severe intestinal pathology characterized by high levels of IFN-γ and IL-17; however, whether this pathology is associated with dysregulated activation of Th1 or Th17 cells is unknown(168).
The role of IL-25 in models of food allergy is less clear. IL-25 is dispensable when allergy is induced using oral delivery of cholera toxin and peanut antigen, but in a model of cutaneous sensitization to peanut, IL-25 from tuft cells is required to regulate intestinal mast cell responses (Figure 1C)(169, 170). Similarly, mice epicutaneously sensitized with TSLP and OVA develop IL-25-dependent diarrhea and anaphylaxis in response to repeated exposure to antigen(171).
IL-25 has also been implicated in numerous models of allergic airway inflammation, including asthma, fibrosis, and CRSwNP (Figure 1B)(2). It is important to note, however, that in most contexts IL-25 is not the dominant innate type 2 cytokine in the lung(172). Airway ILC2s express far more ST2 than IL-17RB(167), and while both IL-33 and IL-25 are produced by nasal epithelial cells during house dust mite (HDM)-induced allergic rhinitis, defects in eosinophil and goblet cell counts are seen only in Il33−/− and not Il25−/− mice(173). Similarly, comparison of Il17rb-deficient and St2-deficient mice in models of allergic asthma revealed that IL-33 more potently induces expansion of IL-13-producing ILC2s, correlating with airway contraction(173).
In the skin, as in the lung, IL-25 is likely not the dominant cytokine driving type 2 immune responses (Figure 1A). Skin-derived ILC2s express Il17rb, but at much lower levels than receptors for IL-18 and IL-33, and in vitro stimulation with IL-25 minimally induces cytokine secretion from skin ILC2s(174). Nonetheless, deletion of Il17rb, using Rora-Cre, strongly attenuates IL-13 production by ILC2s and reduces allergic skin inflammation(175).
Lastly, like IL-33, IL-25 has been linked to maintenance of metabolic health by type 2 immunity (Figure 1D). Treating obese mice with IL-25 induces weight loss, improves glucose tolerance, promotes the browning of white adipose tissues, and is associated with an increase in alternatively activated macrophages(176–178). Mice lacking IL-25 gain more weight on a high-fat diet and have a decrease in energy expenditure(179). Like the lung and skin, adipose-associated ILC2s express ST2 at much higher levels than IL-17RB. Mice lacking IL-33 gain more weight and display defects in beiging of white adipose tissues, while adoptive transfer of ILC2s into Rag2−/−Il2rg−/− mice in the presence of exogenous IL-33 restored adipose beiging(113). Together, these studies indicate that both IL-25 and IL-33 are essential to maintaining homeostasis in adipose tissues.
IL-25 in viral infections and cancer
Of the cytokines discussed in this review, IL-25 is the least pleiotropic. Nonetheless, IL-25 has been implicated in diverse disease contexts. Viral infections are one of the most common causes of asthma exacerbation. In asthma models, lung IL-25 levels are augmented by RV infection, and IL-25R blockade inhibits RV-induced exacerbation(180). Conversely, RV infection drives an increase in epithelial production of IL-33 and TSLP as well as increases in IL-13-secreting ILC2s in neonatal but not mature mice(181). Similar to airway allergic responses, RV-induced disease was attenuated by anti-IL-33 treatment and in TSLPR-knockout mice, suggesting a complex interplay among IL-25, IL-33, and TSLP during viral infections in the lung (Figure 1C)(182).
Unlike IL-33 and TSLP, there are few documented links between IL-25 and cancer. That said, fibrosis is common in tumor microenvironments and IL-25 (and type 2 immunity in general) promotes fibrosis, so this may provide a general link. Also, in murine models of colitis-induced colon cancer, acute IL-25 blockade resulted in greater tumor burdens and increased colitis, but tumor burden was unchanged in Il25-deficient mice (Figure 1F)(183). Similarly,in colorectal cancer patients higher Il25 expression was correlated with ILC2 accumulation in tumors and reduced survival(184). In mice, IL-25-regulated ILC2s sustained tumor-infiltrating myeloid-derived suppressor cells to inhibit anti-tumor immunity, and this could be reversed by IL-25 blockade(184). IL25 and IL17RB expression have also been reported in breast cancers, but the in vivo function remains unclear(185, 186).
Emerging functions of IL-25
Although IL-25 is indeed predominantly a type 2 cytokine, there are interesting examples in which it contributes to responses not primarily associated with type 2 immunity. One example is psoriasis, a skin disease that is predominantly driven by IL-17. Nonetheless, in skin lesions from patients with psoriasis, as well as in imiquimod (IMQ)-induced psoriasis mouse models, keratinocytes express both IL-25 and IL-25R (Figure 1A). In the mouse model, IL-17 activates an autoregulatory circuit in which keratinocytes both secrete and respond to IL-25(187). Indeed, germline or keratinocyte-specific deletion of IL-25 leads to a significant reduction in IMQ-induced psoriasis, indicating a role for IL-25 in this type 3 disease(187). Another example is IL-25-dependent sensing of succinate in the small intestine. Tritrichomonas protists and bacteria release succinate as a metabolite and its detection by small intestinal tuft cells leads to IL-25 release and cytokine production by ILC2s (Figure 1C)(188, 189). Why protists and bacteria would trigger a type 2 response and whether this response benefits the host remains unknown. Acquisition of commensal bacteria also increases IL-25 levels in the colon(190). Conversely, infection with an intestinal pathogenic bacterium, such as Clostridium difficile, suppresses epithelial IL-25 expression, worsening disease outcome(191). Administration of exogenous IL-25 reduces mortality during C. difficile infection, suggesting IL-25 can mediate tissue repair during type 1 bacterial infections(191). Conversely, in the setting of chronic intestinal inflammation, IL-25 is likely detrimental. Production of type 2 cytokines has been reported in ulcerative colitis patients. This finding can be mirrored using oxazolone-induced colitis in mice, and IL-25 blocking antibodies are protective(192). Lastly, there are also examples of IL-25 regulating B1 B cell antibody production, though whether these effects are model-specific or represent a broader paradigm remains to be determined(193).
Therapeutic opportunities
TSLP, IL-33, and IL-25 have been linked to a broad array of inflammatory, infectious, and autoimmune diseases, and cancers making them attractive targets for therapeutic intervention. This notion has been validated by the many mouse studies cited above in which antibody blockade of one or more of these cytokines or their receptors reduced or prevented disease pathology. To date, TSLP and IL-33 but not IL-25 have been targeted in the clinic, with promising results in the treatment of a variety of type 2 allergic diseases and evolving prospects in other disease modalities.
TSLP blockade is a promising therapeutic strategy for asthma in particular. In an initial placebo-controlled trial in moderate-severe uncontrolled asthma, a TSLP blocking antibody (Tezepelumab) was associated with up to a 71% reduction in the annualized asthma exacerbation rate, with significant reductions observed in patients with type 2 as well as non-type 2 asthma(194). Importantly, a larger Phase III trial of Tezepelumab in asthmatics to inhibit exacerbations showed a clear benefit in steroid-resistant asthma and other types of asthma(195). Based on these trials, Tezepelumab has been approved for treatment of severe asthma. Tezepelumab has also been trialed for treatment of moderate to severe atopic dermatitis, with some beneficial effect(196), and it is likely that TSLP will be targeted to treat other type 2 inflammatory diseases. Thus far, TSLP has not been targeted in human cancers or infectious diseases, as we must better understand the context- and cancer-specific roles of TSLP before identifying potentially fruitful opportunities to target TSLP in these contexts.
Our expanding understanding of IL-33 has also made it possible to target the IL-33 pathway therapeutically. Clinical trials are underway or recently completed to treat patients with moderate to severe asthma, COPD, chronic bronchitis and AD with MEDI3506, a neutralizing anti-IL-33 mAb. An anti-IL-33 mAb that blocks IL-33 signaling, itepekimab, was recently tested in a Phase 2 trial for COPD and decreased exacerbation rates in former but not current smokers, leading to ongoing Phase 3 trials(197). Itepekimab also shows therapeutic effects in asthma, but trials in AD were discontinued due to a lack of efficacy(198). Another anti-IL-33 mAb, etokimab, is in ongoing or recently completed trials in patients with CRSwNP, severe eosinophilic asthma, and AD, where therapeutic efficacy was demonstrated(199). In patients with peanut allergy, a Phase 2 trial showed that etokimab treatment was associated with a decrease in adverse events following peanut challenge(200). Finally, astegolimab, and anti-ST2 mAb, is in trials in patients with AD; COPD, in which initial results did an improvement in health status but not exacerbation rate; and asthma, in which treatment was associated with a decrease in exacerbation rate(201, 202). Emerging studies are identifying novel potential applications for IL-33 pathway blockade in non-allergic diseases, including cancer, where IL-33 has been proposed to be a potential adjuvant for cancer vaccines, and infectious disease, though a recent trial showed that astegolimab was not effective in reducing time to improvement in patients with severe COVID-19 pneumonia(203). Ultimately, as with TSLP, additional preclinical studies dissecting the context-specific biology of IL-33 will be required to maximally leverage IL-33 pathway blockade in the clinic.
Beyond the “holy trinity”
In the context of type 2 inflammation, TSLP, IL-33, and IL-25 are routinely cited together, often to the exclusion of other important innate signals. Why these three cytokines are always grouped together is unclear, but is likely a matter of history and habit rather than being biologically motivated. We find that grouping signals based on the transcription factors that they mobilize provides a more informative framework. TSLP, for example, predominantly activates STATs, while IL-33 and IL-25 mobilize NFkB and AP-1. Indeed, these categorizations apply more broadly to each cytokine’s larger cytokine family: IL-2, IL-1, and IL-17, respectively. As would be predicted based on known functions of these transcription factors, TSLP is particularly important for cell survival while IL-33 and IL-25 induce effector functions(204). Notably, none of the cytokines in this “holy trinity” induce the calcium flux needed to activate NFAT, a critical transcription factor for lymphocyte activation. Indeed, ligands like cysteinyl leukotrienes and neuropeptides that signal through G protein coupled receptors to induce the calcium flux needed for NFAT translocation to the nucleus have been shown to provide non-redundant signals for ILC2 activation (Figure 1)(205, 206). Such non-redundant function is likely explained by the well-documented cooperative binding of AP-1 and NFAT to cis-regulatory regions of many immune genes(207). On the other hand, our proposed framework suggests redundancies between IL-33, IL-25, and IL-18, which also activates NFkB and AP-1. Indeed, ILC2s in different tissues predominantly express the receptor for just one of these cytokines(167). Redundancy is also predicted for IL-9R and TSLPR, and to some extent IL-2R and IL-7R, since they all activate STAT5, yet many type 2 immune cells express two or more of these receptors. It could be that regulation is instead achieved by in vivo availability of the respective ligands. The transcription factor framework could also provide insight into a central theme of this review: the role of IL-25, TSLP, and IL-33 in contexts beyond type 2 immunity. In these settings, the function of these cytokines is likely modified by interactions with cytokines the activate different transcription factors. For example, in viral infection IRF-inducing interferons will be abundant, while in cancer TGF-beta-induced Smads are often important. Lastly, while the transcription factor framework is a useful starting point, it is likely that even overlapping signals like IL-25 and IL-33 have nonredundant functions that will need to be carefully dissected. There are already several examples where blocking TSLP, IL-25, and IL-33 provides more benefit than targeting just one or two(39, 208, 209).
Given their redundant and non-redundant functions, understanding the in vivo source and regulation of tissue-derived signals controlling TSLP, IL-33, and IL-25 production is a critical task for future studies. For example, a better understanding is needed of IL-33 release. Also, what ligands drive IL-25 release from tuft cells? What other signals do neurons, fibroblasts, and epithelial cells provide to regulate tissue immunity and how do these differ between and within tissues? By thinking beyond TSLP, IL-33, and IL-25 to consider the full spectrum of tissue-derived signals, we can generate a more complete understanding of not just type 2 immunity, but also anti-viral immunity, cancer, metabolic disease, and more.
Abbreviations used:
- ILC2s
group 2 innate lymphoid cells
- Th2 cells
T helper 2 cells
- DCs
dendritic cells
- TSLP
thymic stromal lymphopoietin
- IL-7
interleukin 7
- IL-7Rα
IL-7 receptor α
- OVA
ovalbumin
- RSV
respiratory syncytial virus
- RV
rhinovirus
- IL-33
interleukin 33
- ST2
IL-1 receptor-like-1
- IL-1RAcP
IL-1 receptor accessory protein
- IL-25/IL-17E
interleukin 17E
- IL-25R
IL-25 receptor composed of IL-17RB and IL-17RA
- CRSwNP
chronic rhinosinusitis with nasal polyps
- AD
atopic dermatitis
- COPD
chronic obstructive pulmonary disease
- MS
multiple sclerosis
- TBI
traumatic brain injury
- EAE
experimental autoimmune encephalomyelitis
- RPM
red pulp macrophages
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosure of potential conflict of interest: The authors declare that they have no relevant conflicts of interest.
References:
- 1.Divekar R, Kita H. Recent advances in epithelium-derived cytokines (IL-33, IL-25, and thymic stromal lymphopoietin) and allergic inflammation. Curr Opin Allergy Clin Immunol. 2015;15(1):98–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Patel NN, Kohanski MA, Maina IW, Workman AD, Herbert DR, Cohen NA. Sentinels at the wall: epithelial-derived cytokines serve as triggers of upper airway type 2 inflammation. Int Forum Allergy Rhinol. 2019;9(1):93–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hasegawa T, Oka T, Demehri S. Alarmin Cytokines as Central Regulators of Cutaneous Immunity. Front Immunol. 2022;13:876515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sims JE, Williams DE, Morrissey PJ, Garka K, Foxworthe D, Price V, et al. Molecular cloning and biological characterization of a novel murine lymphoid growth factor. J Exp Med. 2000;192(5):671–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rochman Y, Spolski R, Leonard WJ. New insights into the regulation of T cells by gamma(c) family cytokines. Nat Rev Immunol. 2009;9(7):480–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Levin SD, Koelling RM, Friend SL, Isaksen DE, Ziegler SF, Perlmutter RM, et al. Thymic stromal lymphopoietin: a cytokine that promotes the development of IgM+ B cells in vitro and signals via a novel mechanism. J Immunol. 1999;162(2):677–83. [PubMed] [Google Scholar]
- 7.Friend SL, Hosier S, Nelson A, Foxworthe D, Williams DE, Farr A. A thymic stromal cell line supports in vitro development of surface IgM+ B cells and produces a novel growth factor affecting B and T lineage cells. Exp Hematol. 1994;22:321–8. [PubMed] [Google Scholar]
- 8.Ray RJ, Furlonger C, Williams DE, Paige CJ. Characterization of thymic stromal-derived lymphopoietin (TSLP) in murine B cell development in vitro. Eur J Immunol. 1996;26(1):10–6. [DOI] [PubMed] [Google Scholar]
- 9.Reche PA, Soumelis V, Gorman DM, Clifford T, Liu M, Travis M, et al. Human thymic stromal lymphopoietin preferentially stimulates myeloid cells. J Immunol. 2001;167(1):336–43. [DOI] [PubMed] [Google Scholar]
- 10.He R, Geha RS. Thymic stromal lymphopoietin. Ann N Y Acad Sci. 2010;1183:13–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kato A, Favoreto S Jr., Avila PC, Schleimer RP. TLR3- and Th2 cytokine-dependent production of thymic stromal lymphopoietin in human airway epithelial cells. J Immunol. 2007;179(2):1080–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Allakhverdi Z, Comeau MR, Jessup HK, Yoon BR, Brewer A, Chartier S, et al. Thymic stromal lymphopoietin is released by human epithelial cells in response to microbes, trauma, or inflammation and potently activates mast cells. J Exp Med. 2007;204(2):253–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Soumelis V, Reche PA, Kanzler H, Yuan W, Edward G, Homey B, et al. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nat Immunol. 2002;3(7):673–80. [DOI] [PubMed] [Google Scholar]
- 14.Allakhverdi Z, Comeau MR, Jessup HK, Delespesse G. Thymic stromal lymphopoietin as a mediator of crosstalk between bronchial smooth muscles and mast cells. J Allergy Clin Immunol. 2009;123(4):958–60. [DOI] [PubMed] [Google Scholar]
- 15.Kaur D, Doe C, Woodman L, Wan WY, Sutcliffe A, Hollins F, et al. Mast cell-airway smooth muscle crosstalk: the role of thymic stromal lymphopoietin. Chest. 2012;142(1):76–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Park LS, Martin U, Garka K, Gliniak B, Di Santo JP, Muller W, et al. Cloning of the murine thymic stromal lymphopoietin (TSLP) receptor: Formation of a functional heteromeric complex requires interleukin 7 receptor. J Exp Med. 2000;192(5):659–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pandey a, Ozaki K, Baumann H, Levin SD, Puel a, Farr aG, et al. Cloning of a receptor subunit required for signaling by thymic stromal lymphopoietin. Nat Immunol. 2000;1(1):59–64. [DOI] [PubMed] [Google Scholar]
- 18.Fujio K, Mosaka T, Kojima T, Kawashima T, Yahata T, Copeland NG, et al. Molecular cloning of a novel type 1 cytokine receptor similar to the common gamma chain. Blood. 2000;95(2204):2210. [PubMed] [Google Scholar]
- 19.Tonozuka Y, Fujio K, Sugiyama T, Nosaka T, Hirai M, Kitamura T. Molecular cloning of a human novel type I cytokine receptor related to delta1/TSLPR. Cytogenet Cell Genet. 2001;93:23–5. [DOI] [PubMed] [Google Scholar]
- 20.Kubick N, Klimovich P, Flournoy PH, Bienkowska I, Lazarczyk M, Sacharczuk M, et al. Interleukins and Interleukin Receptors Evolutionary History and Origin in Relation to CD4+ T Cell Evolution. Genes (Basel). 2021;12(6). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liongue C, Ward AC. Evolution of Class I cytokine receptors. BMC Evol Biol. 2007;7:120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Savino AM, Izraeli S. On mice and humans: the role of thymic stromal lymphopoietin in human B-cell development and leukemia. Haematologica. 2016;101(4):391–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ziegler SF. The role of thymic stromal lymphopoietin (TSLP) in allergic disorders. Curr Opin Immunol. 2010;22:795–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fang C, Siew LQ, Corrigan CJ, Ying S. The role of thymic stromal lymphopoietin in allergic inflammation and chronic obstructive pulmonary disease. Arch Immunol Ther Exp (Warsz ). 2010;58(2):81–90. [DOI] [PubMed] [Google Scholar]
- 25.Ying S, O’Connor B, Ratoff J, Meng Q, Mallett K, Cousins D, et al. Thymic stromal lymphopoietin expression is increased in asthmatic airways and correlates with expression of Th2-attracting chemokines and disease severity. J Immunol. 2005;174(12):8183–90. [DOI] [PubMed] [Google Scholar]
- 26.Rothenberg ME, Spergel JM, Sherrill JD, Annaiah K, Martin LJ, Cianferoni A, et al. Common variants at 5q22 associate with pediatric eosinophilic esophagitis. Nat Genet. 2010;42(4):289–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Miyake Y, Hitsumoto S, Tanaka K, Arakawa M. Association Between TSLP Polymorphisms and Eczema in Japanese Women: the Kyushu Okinawa Maternal and Child Health Study. Inflammation. 2015;38(4):1663–8. [DOI] [PubMed] [Google Scholar]
- 28.Harada M, Hirota T, Jodo AI, Hitomi Y, Sakashita M, Tsunoda T, et al. TSLP Promoter Polymorphisms are Associated with Susceptibility to Bronchial Asthma. Am J Respir Cell Mol Biol. 2010; 44(6):787–793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cianferoni A, Spergel J. The importance of TSLP in allergic disease and its role as a potential therapeutic target. Expert Rev Clin Immunol. 2014;10(11):1463–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shikotra A, Choy DF, Ohri CM, Doran E, Butler C, Hargadon B, et al. Increased expression of immunoreactive thymic stromal lymphopoietin in patients with severe asthma. J Allergy Clin Immunol. 2012;129(1):104–11. [DOI] [PubMed] [Google Scholar]
- 31.Briot A, Deraison C, Lacroix M, Bonnart C, Robin A, Besson C, et al. Kallikrein 5 induces atopic dermatitis-like lesions through PAR2-mediated thymic stromal lymphopoietin expression in Netherton syndrome. J Exp Med. 2009;206(5):1135–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Noti M, Wojno ED, Kim BS, Siracusa MC, Giacomin PR, Nair MG, et al. Thymic stromal lymphopoietin-elicited basophil responses promote eosinophilic esophagitis. Nat Med. 2013;19(8):1005–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Han H, Xu W, Headley MB, Jessup HK, Lee KS, Omori M, et al. Thymic stromal lymphopoietin (TSLP)-mediated dermal inflammation aggravates experimental asthma. Mucosal Immunol. 2012;5(3):342–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Al-Shami A, Spolski R, Kelly J, Keane-Myers A, Leonard WJ. A role for TSLP in the development of inflammation in an asthma model. J Exp Med. 2005;202(6):829–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhou B, Comeau MR, De Smedt T, Liggitt HD, Dahl ME, Lewis DB, et al. Thymic stromal lymphopoietin as a key initiator of allergic airway inflammation in mice. Nature Immunol. 2005;6(10):1047–53. [DOI] [PubMed] [Google Scholar]
- 36.Chu DK, Llop-Guevara A, Walker TD, Flader K, Goncharova S, Boudreau JE, et al. IL-33, but not thymic stromal lymphopoietin or IL-25, is central to mite and peanut allergic sensitization. J Allergy Clin Immunol. 2013;131(1):187–200 e1–8. [DOI] [PubMed] [Google Scholar]
- 37.Jang S, Morris S, Lukacs NW. TSLP promotes induction of Th2 differentiation but is not necessary during established allergen-induced pulmonary disease. PLoS One. 2013;8(2):e56433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Massacand JC, Stettler RC, Meier R, Humphreys NE, Grencis RK, Marsland BJ, et al. Helminth products bypass the need for TSLP in Th2 immune responses by directly modulating dendritic cell function. Proc Natl Acad Sci U S A. 2009;106(33):13968–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Khodoun MV, Tomar S, Tocker JE, Wang YH, Finkelman FD. Prevention of food allergy development and suppression of established food allergy by neutralization of thymic stromal lymphopoietin, IL-25, and IL-33. J Allergy Clin Immunol. 2018;141(1):171–9 e1. [DOI] [PubMed] [Google Scholar]
- 40.Omori M, Ziegler S. Induction of IL-4 expression in CD4+ T cells by thymic stromal lymphopoietin. J Immunol. 2007;178(3):1396–404 [DOI] [PubMed] [Google Scholar]
- 41.Rochman I, Watanabe N, Arima K, Liu YJ, Leonard WJ. Cutting edge: direct action of thymic stromal lymphopoietin on activated human CD4+ T cells. J Immunol. 2007;178(11):6720–4. [DOI] [PubMed] [Google Scholar]
- 42.Kitajima M, Lee HC, Nakayama T, Ziegler SF. TSLP enhances the function of helper type 2 cells. Eur J Immunol. 2011;41(7):1862–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lee HC, Headley MB, Loo YM, Berlin A, Gale M Jr., Debley JS, et al. Thymic stromal lymphopoietin is induced by respiratory syncytial virus-infected airway epithelial cells and promotes a type 2 response to infection. J Allergy Clin Immunol. 2012;130:1187–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Stier MT, Bloodworth MH, Toki S, Newcomb DC, Goleniewska K, Boyd KL, et al. Respiratory syncytial virus infection activates IL-13-producing group 2 innate lymphoid cells through thymic stromal lymphopoietin. J Allergy Clin Immunol. 2016;138(3):814–24 e11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fan P, Liu Z, Zheng M, Chen M, Xu Y, Zhao D. Respiratory syncytial virus nonstructural protein 1 breaks immune tolerance in mice by downregulating Tregs through TSLPOX40/OX40L-mTOR axis. Mol Immunol. 2021;138:20–30. [DOI] [PubMed] [Google Scholar]
- 46.Perez GF, Pancham K, Huseni S, Preciado D, Freishtat RJ, Colberg-Poley AM, et al. Rhinovirus infection in young children is associated with elevated airway TSLP levels. Eur Respir J. 2014;44(4):1075–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Jackson DJ, Gangnon RE, Evans MD, Roberg KA, Anderson EL, Pappas TE, et al. Wheezing rhinovirus illnesses in early life predict asthma development in high-risk children. Am J Respir Crit Care Med. 2008;178(7):667–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jartti T, Lehtinen P, Vuorinen T, Osterback R, van den Hoogen B, Osterhaus AD, et al. Respiratory picornaviruses and respiratory syncytial virus as causative agents of acute expiratory wheezing in children. Emerg Infect Dis. 2004;10(6):1095–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yadava K, Sichelstiel A, Luescher IF, Nicod LP, Harris NL, Marsland BJ. TSLP promotes influenza-specific CD8+ T-cell responses by augmenting local inflammatory dendritic cell function. Mucosal immunology. 2013; 6(1):83–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shane HL, Klonowski KD. A direct and nonredundant role for thymic stromal lymphopoietin on antiviral CD8 T cell responses in the respiratory mucosa. J Immunol. 2014;192(5):2261–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ebina-Shibuya R, West EE, Spolski R, Li P, Oh J, Kazemian M, et al. Thymic stromal lymphopoietin limits primary and recall CD8(+) T-cell anti-viral responses. Elife. 2021;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ye L, Schnepf D, Becker J, Ebert K, Tanriver Y, Bernasconi V, et al. Interferon-lambda enhances adaptive mucosal immunity by boosting release of thymic stromal lymphopoietin. Nat Immunol. 2019;20(5):593–601. [DOI] [PubMed] [Google Scholar]
- 53.Ye L, Schnepf D, Ohnemus A, Ong LC, Gad HH, Hartmann R, et al. Interferon-lambda Improves the Efficacy of Intranasally or Rectally Administered Influenza Subunit Vaccines by a Thymic Stromal Lymphopoietin-Dependent Mechanism. Front Immunol. 2021;12:749325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Binnewies M, Roberts EW, Kersten K, Chan V, Fearon DF, Merad M, et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med. 2018;24(5):541–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Loose D, Van de Wiele C. The immune system and cancer. Cancer Biother Radiopharm. 2009;24:369–76. [DOI] [PubMed] [Google Scholar]
- 56.Aspord C, Pedroza-Gonzalez A, Gallegos M, Tindle S, Burton EC, Su D, et al. Breast cancer instructs dendritic cells to prime interleukin 13-secreting CD4+ T cells that facilitate tumor development. J Exp Med. 2007;204:1037–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.De Monte L, Reni M, Tassi E, l Clavenna D, Papa I, Recalde H, Braga M, et al. Intratumor T helper type 2 cell infiltrate correlates with cancer-associated fibroblast thymic stromal lymphpoietin production and reduced survival in pancreatic cancer. J Exp Med. 2011;208:469–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Boieri M, Malishkevich A, Guennoun R, Marchese E, Kroon S, Trerice KE, et al. CD4+ T helper 2 cells suppress breast cancer by inducing terminal differentiation. J Exp Med. 2022;219(7). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Moral JA, Leung J, Rojas LA, Ruan J, Zhao J, Sethna Z, et al. ILC2s amplify PD-1 blockade by activating tissue-specific cancer immunity. Nature. 2020;579(7797):130–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Brunetto E, De Monte L, Balzano G, Camisa B, Laino V, Riba M, et al. The IL-1/IL-1 receptor axis and tumor cell released inflammasome adaptor ASC are key regulators of TSLP secretion by cancer associated fibroblasts in pancreatic cancer. J Immunother Cancer. 2019;7(1):45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Barooei R, Mahmoudian RA, Abbaszadegan MR, Mansouri A, Gholamin M. Evaluation of thymic stromal lymphopoietin (TSLP) and its correlation with lymphatic metastasis in human gastric cancer. Med Oncol. 2015;32(8):217. [DOI] [PubMed] [Google Scholar]
- 62.Xu L, Guo Y, Xu N, Chen L, Zhu J, Liu N, et al. Overexpression of thymic stromal lymphopoietin is correlated with poor prognosis in epithelial ovarian carcinoma. Biosci Rep. 2019;39(5). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Watanabe J, Saito H, Miyatani K, Ikeguchi M, Umekita Y. TSLP Expression and High Serum TSLP Level Indicate a Poor Prognosis in Gastric Cancer Patients. Yonago Acta Med. 2015;58(3):137–43. [PMC free article] [PubMed] [Google Scholar]
- 64.Xie F, Meng YH, Liu LB, Chang KK, LI H, Li MQ, et al. Cervical carcinoma cells stimulate the angiogenesis through TSLP promoting growth and activation of vascular endothelial cells. Am J Reprod Immunol. 2013; 70(1):69–79. [DOI] [PubMed] [Google Scholar]
- 65.Pedroza-Gonzalez A, Xu K, Wu TC, Aspord C, Tindle S, Marches F, et al. Thymic stromal lymphopoietin fosters human breast tumor growth by promoting type 2 inflammation. J Exp Med. 2011;208(3):479–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Wu TC, Xu K, Martinek J, Young RR, Banchereau R, George J, et al. IL1 Receptor Antagonist Controls Transcriptional Signature of Inflammation in Patients with Metastatic Breast Cancer. Cancer Res. 2018;78(18):5243–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Aslakson CJ, Miller FR. Selective events in the metastatic process defined by analysis of the sequential dissemination of subpopulations of a mouse mammary tumor. Cancer Res. 1992;52:1399–405. [PubMed] [Google Scholar]
- 68.Olkhanud PB, Rochman Y, Bodogai M, Malchinkhuu E, Wejksza K, Xu M, et al. Thymic stromal lymphopoietin is a key mediator of breast cancer progression. J Immunol. 2011;186(10):5656–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ghirelli C, Sadacca B, Reyal F, Zollinger R, Michea P, Sirven P, et al. No evidence for TSLP pathway activity in human breast cancer. Oncoimmunology. 2016;5(8):e1178438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kuan EL, Ziegler SF. A tumor-myeloid cell axis, mediated via the cytokines IL-1alpha and TSLP, promotes the progression of breast cancer. Nat Immunol. 2018;19(4):366–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Taylor BC, Zaph C, Troy AE, Du Y, Guild KJ, Comeau MR, et al. TSLP regulates intestinal immunity and inflammation in mouse models of helminth infection and colitis. J Exp Med. 2009;206(3):655–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Tahaghoghi-Hajghorbani S, Ajami A, Ghorbanalipoor S, Hosseini-Khah Z, Taghiloo S, Khaje-Enayati P, et al. Protective effect of TSLP and IL-33 cytokines in ulcerative colitis. Auto Immun Highlights. 2019;10(1):1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Obata-Ninomiya K, de Jesus Carrion S, Hu A, Ziegler SF. Emerging role for thymic stromal lymphopoietin-responsive regulatory T cells in colorectal cancer progression in humans and mice. Sci Transl Med. 2022;14(645):eabl6960. [DOI] [PubMed] [Google Scholar]
- 74.Mullighan CG. The molecular genetic makeup of acute lymphoblastic leukemia. Hematology Am Soc Hematol Educ Program. 2012;2012:389–96. [DOI] [PubMed] [Google Scholar]
- 75.Yoda A, Yoda Y, Chiaretti S, Bar-Natan M, Mani K, Rodig SJ, et al. Functional screening identifies CRLF2 in precursor B-cell acute lymphoblastic leukemia. Proc Natl Acad Sci U S A. 2010;107(1):252–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Mullighan CG, Collins-Underwood JR, Phillips LA, Loudin MG, Liu W, Zhang J, et al. Rearrangement of CRLF2 in B-progenitor- and Down syndrome-associated acute lymphoblastic leukemia. Nat Genet. 2009;41(11):1243–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Russell LJ, Capasso M, Vater I, Akasaka T, Bernard OA, Calasanz MJ, et al. Deregulated expression of cytokine receptor gene, CRLF2, is involved in lymphoid transformation in B-cell precursor acute lymphoblastic leukemia. Blood. 2009;114:2688–98. [DOI] [PubMed] [Google Scholar]
- 78.Chapiro E, Russell L, Lainey E, Kaltenbach S, Ragu C, Della-Valle V, et al. Activating mutation in the TSLPR gene in B-cell precursor lymphoblastic leukemia. Leukemia. 2010;24:642–5. [DOI] [PubMed] [Google Scholar]
- 79.Qin H, Cho M, Haso W, Zhang L, Tasian SK, Oo HZ, et al. Eradication of B-ALL using chimeric antigen receptor-expressing T cells targeting the TSLPR oncoprotein. Blood. 2015;126(5):629–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Scheeren FA, van Lent AU, Nagasawa M, Weijer K, Spits H, Legrand N, et al. Thymic stromal lymphopoietin induces early human B-cell proliferation and differentiation. Eur J Immunol. 2010;40(4):955–65. [DOI] [PubMed] [Google Scholar]
- 81.Shannon JL, Corcoran DL, Murray JC, Ziegler SF, MacLeod AS, Zhang JY. Thymic stromal lymphopoietin controls hair growth. Stem Cell Reports. 2022;17(3):649–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cayrol C, Girard JP. Interleukin-33 (IL-33): A nuclear cytokine from the IL-1 family. Immunol Rev. 2018;281(1):154–68. [DOI] [PubMed] [Google Scholar]
- 83.Baekkevold ES, Roussigne M, Yamanaka T, Johansen FE, Jahnsen FL, Amalric F, et al. Molecular characterization of NF-HEV, a nuclear factor preferentially expressed in human high endothelial venules. Am J Pathol. 2003;163(1):69–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Matarazzo L, Hernandez Santana YE, Walsh PT, Fallon PG. The IL-1 cytokine family as custodians of barrier immunity. Cytokine. 2022;154:155890. [DOI] [PubMed] [Google Scholar]
- 85.Rivers-Auty J, Daniels MJD, Colliver I, Robertson DL, Brough D. Redefining the ancestral origins of the interleukin-1 superfamily. Nat Commun. 2018;9(1):1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Onda H, Kasuya H, Takakura K, Hori T, Imaizumi T, Takeuchi T, et al. Identification of genes differentially expressed in canine vasospastic cerebral arteries after subarachnoid hemorrhage. J Cereb Blood Flow Metab. 1999;19(11):1279–88. [DOI] [PubMed] [Google Scholar]
- 87.Schmitz J, Owyang A, Oldham E, Song Y, Murphy E, McClanahan TK, et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity. 2005;23(5):479–90. [DOI] [PubMed] [Google Scholar]
- 88.Bessa J, Meyer CA, de Vera Mudry MC, Schlicht S, Smith SH, Iglesias A, et al. Altered subcellular localization of IL-33 leads to non-resolving lethal inflammation. J Autoimmun. 2014;55:33–41. [DOI] [PubMed] [Google Scholar]
- 89.Travers J, Rochman M, Miracle CE, Habel JE, Brusilovsky M, Caldwell JM, et al. Chromatin regulates IL-33 release and extracellular cytokine activity. Nat Commun. 2018;9(1):3244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hung LY, Tanaka Y, Herbine K, Pastore C, Singh B, Ferguson A, et al. Cellular context of IL-33 expression dictates impact on anti-helminth immunity. Sci Immunol. 2020;5(53). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Cayrol C, Girard JP. The IL-1-like cytokine IL-33 is inactivated after maturation by caspase-1. Proc Natl Acad Sci U S A 2009;106(22):9021–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Luthi AU, Cullen SP, McNeela EA, Duriez PJ, Afonina IS, Sheridan C, et al. Suppression of interleukin-33 bioactivity through proteolysis by apoptotic caspases. Immunity. 2009;31(1):84–98. [DOI] [PubMed] [Google Scholar]
- 93.Lefrancais E, Roga S, Gautier V, Gonzalez-de-Peredo A, Monsarrat B, Girard JP, et al. IL-33 is processed into mature bioactive forms by neutrophil elastase and cathepsin G. Proc Natl Acad Sci U S A. 2012;109(5):1673–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Palmer G, Gabay C. Interleukin-33 biology with potential insights into human diseases. Nat Rev Rheumatol. 2011;7(6):321–9. [DOI] [PubMed] [Google Scholar]
- 95.Liew FY, Girard JP, Turnquist HR. Interleukin-33 in health and disease. Nat Rev Immunol. 2016;16(11):676–89. [DOI] [PubMed] [Google Scholar]
- 96.Rank MA, Kobayashi T, Kozaki H, Bartemes KR, Squillace DL, Kita H. IL-33-activated dendritic cells induce an atypical TH2-type response. J Allergy Clin Immunol. 2009;123(5):1047–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Smithgall MD, Comeau MR, Yoon BR, Kaufman D, Armitage R, Smith DE. IL-33 amplifies both Th1- and Th2-type responses through its activity on human basophils, allergen-reactive Th2 cells, iNKT and NK cells. Int Immunol. 2008;20(8):1019–30. [DOI] [PubMed] [Google Scholar]
- 98.Neill DR, Wong SH, Bellosi A, Flynn RJ, Daly M, Langford TK, et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature. 2010;464(7293):1367–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Mun SH, Ko NY, Kim HS, Kim JW, Kim DK, Kim AR, et al. Interleukin-33 stimulates formation of functional osteoclasts from human CD14(+) monocytes. Cell Mol Life Sci. 2010;67(22):3883–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Salmond RJ, Mirchandani AS, Besnard AG, Bain CC, Thomson NC, Liew FY. IL-33 induces innate lymphoid cell-mediated airway inflammation by activating mammalian target of rapamycin. J Allergy Clin Immunol. 2012;130(5):1159–66 e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chow JY, Wong CK, Cheung PF, Lam CW. Intracellular signaling mechanisms regulating the activation of human eosinophils by the novel Th2 cytokine IL-33: implications for allergic inflammation. Cell Mol Immunol. 2010;7(1):26–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Molofsky AB, Nussbaum JC, Liang HE, Van Dyken SJ, Cheng LE, Mohapatra A, et al. Innate lymphoid type 2 cells sustain visceral adipose tissue eosinophils and alternatively activated macrophages. J Exp Med. 2013;210(3):535–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Tait Wojno ED, Artis D. Emerging concepts and future challenges in innate lymphoid cell biology. J Exp Med. 2016;213(11):2229–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Espinassous Q, Garcia-de-Paco E, Garcia-Verdugo I, Synguelakis M, von Aulock S, Sallenave JM, et al. IL-33 enhances lipopolysaccharide-induced inflammatory cytokine production from mouse macrophages by regulating lipopolysaccharide receptor complex. J Immunol. 2009;183(2):1446–55. [DOI] [PubMed] [Google Scholar]
- 105.Besnard AG, Togbe D, Guillou N, Erard F, Quesniaux V, Ryffel B. IL-33-activated dendritic cells are critical for allergic airway inflammation. Eur J Immunol. 2011;41(6):1675–86. [DOI] [PubMed] [Google Scholar]
- 106.Hammad H, Lambrecht BN. Barrier Epithelial Cells and the Control of Type 2 Immunity. Immunity. 2015;43(1):29–40. [DOI] [PubMed] [Google Scholar]
- 107.Hung LY, Lewkowich IP, Dawson LA, Downey J, Yang Y, Smith DE, et al. IL-33 drives biphasic IL-13 production for noncanonical Type 2 immunity against hookworms. Proc Natl Acad Sci U S A 2013;110(1):282–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Yasuda K, Muto T, Kawagoe T, Matsumoto M, Sasaki Y, Matsushita K, et al. Contribution of IL-33-activated type II innate lymphoid cells to pulmonary eosinophilia in intestinal nematode-infected mice. Proc Natl Acad Sci U S A. 2012;109(9):3451–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Imai Y, Yasuda K, Sakaguchi Y, Haneda T, Mizutani H, Yoshimoto T, et al. Skin-specific expression of IL-33 activates group 2 innate lymphoid cells and elicits atopic dermatitis-like inflammation in mice. Proc Natl Acad Sci U S A 2013;110(34):13921–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Altman MC, Lai Y, Nolin JD, Long S, Chen CC, Piliponsky AM, et al. Airway epithelium-shifted mast cell infiltration regulates asthmatic inflammation via IL-33 signaling. J Clin Invest. 2019;129(11):4979–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Han H, Roan F, Johnston LK, Smith DE, Bryce PJ, Ziegler SF. IL-33 promotes gastrointestinal allergy in a TSLP-independent manner. Mucosal Immunol. 2018;11(2):578. [DOI] [PubMed] [Google Scholar]
- 112.Halim TYF, Rana BMJ, Walker JA, Kerscher B, Knolle MD, Jolin HE, et al. Tissue-Restricted Adaptive Type 2 Immunity Is Orchestrated by Expression of the Costimulatory Molecule OX40L on Group 2 Innate Lymphoid Cells. Immunity. 2018;48(6):1195–207 e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Brestoff JR, Kim BS, Saenz SA, Stine RR, Monticelli LA, Sonnenberg GF, et al. Group 2 innate lymphoid cells promote beiging of white adipose tissue and limit obesity. Nature. 2015;519(7542):242–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Odegaard JI, Lee MW, Sogawa Y, Bertholet AM, Locksley RM, Weinberg DE, et al. Perinatal Licensing of Thermogenesis by IL-33 and ST2. Cell. 2016;166(4):841–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Lee MW, Odegaard JI, Mukundan L, Qiu Y, Molofsky AB, Nussbaum JC, et al. Activated type 2 innate lymphoid cells regulate beige fat biogenesis. Cell. 2015;160(1–2):74–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Meng X, Qian X, Ding X, Wang W, Yin X, Zhuang G, et al. Eosinophils regulate intra-adipose axonal plasticity. Proc Natl Acad Sci U S A 2022;119(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Mahlakoiv T, Flamar AL, Johnston LK, Moriyama S, Putzel GG, Bryce PJ, et al. Stromal cells maintain immune cell homeostasis in adipose tissue via production of interleukin-33. Sci Immunol. 2019;4(35). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Rostan O, Arshad MI, Piquet-Pellorce C, Robert-Gangneux F, Gangneux JP, Samson M. Crucial and diverse role of the interleukin-33/ST2 axis in infectious diseases. Infect Immun. 2015;83(5):1738–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Bonilla WV, Frohlich A, Senn K, Kallert S, Fernandez M, Johnson S, et al. The alarmin interleukin-33 drives protective antiviral CD8(+) T cell responses. Science. 2012;335(6071):984–9. [DOI] [PubMed] [Google Scholar]
- 120.Baumann C, Bonilla WV, Frohlich A, Helmstetter C, Peine M, Hegazy AN, et al. T-bet- and STAT4-dependent IL-33 receptor expression directly promotes antiviral Th1 cell responses. Proc Natl Acad Sci U S A. 2015;112(13):4056–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Arpaia N, Green JA, Moltedo B, Arvey A, Hemmers S, Yuan S, et al. A Distinct Function of Regulatory T Cells in Tissue Protection. Cell. 2015;162(5):1078–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Monticelli LA, Sonnenberg GF, Abt MC, Alenghat T, Ziegler CG, Doering TA, et al. Innate lymphoid cells promote lung-tissue homeostasis after infection with influenza virus. Nat Immunol. 2011;12(11):1045–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Chang YJ, Kim HY, Albacker LA, Baumgarth N, McKenzie AN, Smith DE, et al. Innate lymphoid cells mediate influenza-induced airway hyper-reactivity independently of adaptive immunity. Nat Immunol. 2011;12(7):631–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Markovic SS, Jovanovic M, Gajovic N, Jurisevic M, Arsenijevic N, Jovanovic M, et al. IL 33 Correlates With COVID-19 Severity, Radiographic and Clinical Finding. Front Med (Lausanne). 2021;8:749569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Afferni C, Buccione C, Andreone S, Galdiero MR, Varricchi G, Marone G, et al. The Pleiotropic Immunomodulatory Functions of IL-33 and Its Implications in Tumor Immunity. Front Immunol. 2018;9:2601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Pastille E, Wasmer MH, Adamczyk A, Vu VP, Mager LF, Phuong NNT, et al. The IL-33/ST2 pathway shapes the regulatory T cell phenotype to promote intestinal cancer. Mucosal Immunol. 2019;12(4):990–1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Schuijs MJ, Png S, Richard AC, Tsyben A, Hamm G, Stockis J, et al. ILC2-driven innate immune checkpoint mechanism antagonizes NK cell antimetastatic function in the lung. Nat Immunol. 2020;21(9):998–1009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Dominguez D, Ye C, Geng Z, Chen S, Fan J, Qin L, et al. Exogenous IL-33 Restores Dendritic Cell Activation and Maturation in Established Cancer. J Immunol. 2017;198(3):1365–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Gao X, Wang X, Yang Q, Zhao X, Wen W, Li G, et al. Tumoral expression of IL-33 inhibits tumor growth and modifies the tumor microenvironment through CD8+ T and NK cells. J Immunol. 2015;194(1):438–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Lucarini V, Ziccheddu G, Macchia I, La Sorsa V, Peschiaroli F, Buccione C, et al. IL-33 restricts tumor growth and inhibits pulmonary metastasis in melanoma-bearing mice through eosinophils. Oncoimmunology. 2017;6(6):e1317420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Jacquelot N, Seillet C, Wang M, Pizzolla A, Liao Y, Hediyeh-Zadeh S, et al. Blockade of the co-inhibitory molecule PD-1 unleashes ILC2-dependent antitumor immunity in melanoma. Nat Immunol. 2021;22(7):851–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Sun Y, Wen Y, Wang L, Wen L, You W, Wei S, et al. Therapeutic Opportunities of Interleukin-33 in the Central Nervous System. Front Immunol. 2021;12:654626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Gadani SP, Walsh JT, Smirnov I, Zheng J, Kipnis J. The glia-derived alarmin IL-33 orchestrates the immune response and promotes recovery following CNS injury. Neuron. 2015;85(4):703–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Still KM, Batista SJ, O’Brien CA, Oyesola OO, Fruh SP, Webb LM, et al. Astrocytes promote a protective immune response to brain Toxoplasma gondii infection via IL-33-ST2 signaling. PLoS Pathog. 2020;16(10):e1009027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Vainchtein ID, Chin G, Cho FS, Kelley KW, Miller JG, Chien EC, et al. Astrocyte-derived interleukin-33 promotes microglial synapse engulfment and neural circuit development. Science. 2018;359(6381):1269–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Nguyen PT, Dorman LC, Pan S, Vainchtein ID, Han RT, Nakao-Inoue H, et al. Microglial Remodeling of the Extracellular Matrix Promotes Synapse Plasticity. Cell. 2020;182(2):388–403 e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Lu Y, Basatemur G, Scott IC, Chiarugi D, Clement M, Harrison J, et al. Interleukin-33 Signaling Controls the Development of Iron-Recycling Macrophages. Immunity. 2020;52(5):782–93 e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Swann JW, Koneva LA, Regan-Komito D, Sansom SN, Powrie F, Griseri T. IL-33 promotes anemia during chronic inflammation by inhibiting differentiation of erythroid progenitors. J Exp Med. 2020;217(9). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Gieseck RL 3rd, Wilson MS, Wynn TA. Type 2 immunity in tissue repair and fibrosis. Nat Rev Immunol. 2018;18(1):62–76. [DOI] [PubMed] [Google Scholar]
- 140.Lopetuso LR, Scaldaferri F, Pizarro TT. Emerging role of the interleukin (IL)-33/ST2 axis in gut mucosal wound healing and fibrosis. Fibrogenesis Tissue Repair. 2012;5(1):18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Han Q, Das S, Hirano M, Holland SJ, McCurley N, Guo P, et al. Characterization of Lamprey IL-17 Family Members and Their Receptors. The Journal of Immunology. 2015;195(11):5440–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Hurst SD, Muchamuel T, Gorman DM, Gilbert JM, Clifford T, Kwan S, et al. New IL-17 Family Members Promote Th1 or Th2 Responses in the Lung: In Vivo Function of the Novel Cytokine IL-25. The Journal of Immunology. 2002;169(1):443–53. [DOI] [PubMed] [Google Scholar]
- 143.Rickel EA, Siegel LA, Yoon B-RP, Rottman JB, Kugler DG, Swart DA, et al. Identification of Functional Roles for Both IL-17RB and IL-17RA in Mediating IL-25-Induced Activities. The Journal of Immunology. 2008;181(6):4299–310. [DOI] [PubMed] [Google Scholar]
- 144.Bankova LG, Dwyer DF, Yoshimoto E, Ualiyeva S, John W, Raff H, et al. inflammation. 2019;3(28). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Von Moltke J, Ji M, Liang HE, Locksley RM. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature. 2016;529(7585):221–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Ikeda K, Nakajima H, Suzuki K, Kagami SI, Hirose K, Suto A, et al. Mast cells produce interleukin-25 upon FcεRI-mediated activation. Blood. 2003;101(9):3594–6. [DOI] [PubMed] [Google Scholar]
- 147.Kang CM, Jang AS, Ahn MH, Shin JA, Kim JH, Choi YS, et al. Interleukin-25 and interleukin-13 production by alveolar macrophages in response to particles. American Journal of Respiratory Cell and Molecular Biology. 2005;33(3):290–6. [DOI] [PubMed] [Google Scholar]
- 148.Suzukawa M, Morita H, Nambu A, Arae K, Shimura E, Shibui A, et al. Epithelial Cell-Derived IL-25, but Not Th17 Cell-Derived IL-17 or IL-17F, Is Crucial for Murine Asthma. The Journal of Immunology. 2012;189(7):3641–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Leyva-Castillo JM, Galand C, Mashiko S, Bissonnette R, McGurk A, Ziegler SF, et al. ILC2 activation by keratinocyte-derived IL-25 drives IL-13 production at sites of allergic skin inflammation. Journal of Allergy and Clinical Immunology. 2020;145(6):1606–14.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Suto H, Nambu A, Morita H, Yamaguchi S, Numata T, Yoshizaki T, et al. IL-25 enhances TH17 cell–mediated contact dermatitis by promoting IL-1β production by dermal dendritic cells. Journal of Allergy and Clinical Immunology. 2018;142(5):1500–9.e10. [DOI] [PubMed] [Google Scholar]
- 151.Borowczyk J, Shutova M, Brembilla NC, Boehncke WH. IL-25 (IL-17E) in epithelial immunology and pathophysiology. Journal of Allergy and Clinical Immunology. 2021;148(1):40–52. [DOI] [PubMed] [Google Scholar]
- 152.Heng TS, Painter MW, Immunological Genome Project C. The Immunological Genome Project: networks of gene expression in immune cells. Nat Immunol. 2008;9(10):1091–4. [DOI] [PubMed] [Google Scholar]
- 153.Huang Y, Guo L, Qiu J, Chen X, Hu-Li J, Siebenlist U, et al. IL-25-responsive, lineage-negative KLRG1 hi cells are multipotential ‘inflammatory’ type 2 innate lymphoid cells. Nature Immunology. 2015;16(2):161–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Saenz SA, Siracusa MC, Monticelli LA, Ziegler CGK, Kim BS, Brestoff JR, et al. IL-25 simultaneously elicits distinct populations of innate lymphoid cells and multipotent progenitor type 2 (MPPtype2) cells. Journal of Experimental Medicine. 2013;210(9):1823–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Pan G, French D, Mao W, Maruoka M, Risser P, Lee J, et al. Forced Expression of Murine IL-17E Induces Growth Retardation, Jaundice, a Th2-Biased Response, and Multiorgan Inflammation in Mice. The Journal of Immunology. 2001;167(11):6559–67. [DOI] [PubMed] [Google Scholar]
- 156.Angkasekwinai P, Park H, Wang YH, Wang YH, Seon HC, Corry DB, et al. Interleukin 25 promotes the initiation of proallergic type 2 responses. Journal of Experimental Medicine. 2007;204(7):1509–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Lee JB, Chen CY, Liu B, Mugge L, Angkasekwinai P, Facchinetti V, et al. IL-25 and CD4+ TH2 cells enhance type 2 innate lymphoid cell-derived IL-13 production, which promotes IgE-mediated experimental food allergy. Journal of Allergy and Clinical Immunology. 2016;137(4):1216–25.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Aalberse JA, van Thuijl AO, Meijer Y, de Jager W, van der Palen-Merkus T, Sprikkelman AB, et al. Plasma IL-25 is elevated in a subgroup of patients with clinical reactivity to peanut. Clin Transl Allergy. 2013;3(1):40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Corrigan CJ, Wang W, Meng Q, Fang C, Eid G, Caballero MR, et al. Allergen-induced expression of IL-25 and IL-25 receptor in atopic asthmatic airways and late-phase cutaneous responses. J Allergy Clin Immunol. 2011;128(1):116–24. [DOI] [PubMed] [Google Scholar]
- 160.Hvid M, Vestergaard C, Kemp K, Christensen GB, Deleuran B, Deleuran M. IL-25 in atopic dermatitis: a possible link between inflammation and skin barrier dysfunction? J Invest Dermatol. 2011;131(1):150–7. [DOI] [PubMed] [Google Scholar]
- 161.Chen F, Hong H, Sun Y, Hu X, Zhang J, Xu G, et al. Nasal interleukin 25 as a novel biomarker for patients with chronic rhinosinusitis with nasal polyps and airway hypersensitiveness: A pilot study. Ann Allergy Asthma Immunol. 2017;119(4):310–6 e2. [DOI] [PubMed] [Google Scholar]
- 162.Wang YH, Angkasekwinai P, Lu N, Voo KS, Arima K, Hanabuchi S, et al. IL-25 augments type 2 immune responses by enhancing the expansion and functions of TSLP-DC-activated Th2 memory cells. J Exp Med. 2007;204(8):1837–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Fallon PG, Ballantyne SJ, Mangan NE, Barlow JL, Dasvarma A, Hewett DR, et al. Identification of an interleukin (IL)-25-dependent cell population that provides IL-4, IL-5, and IL-13 at the onset of helminth expulsion. Journal of Experimental Medicine. 2006;203(4):1105–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Pei C, Zhao C, Wang AJ, Fan AX, Grinchuk V, Smith A, et al. Critical role for interleukin-25 in host protective Th2 memory response against Heligmosomoides polygyrus bakeri. Infection and Immunity. 2016;84(12):3328–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Smith KA, Löser S, Varyani F, Harcus Y, McSorley HJ, McKenzie ANJ, et al. Concerted IL-25R and IL-4Rα signaling drive innate type 2 effector immunity for optimal helminth expulsion. eLife. 2018;7:1–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Gerbe F, Sidot E, Smyth DJ, Ohmoto M, Matsumoto I, Dardalhon V, et al. Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature. 2016;529(7585):226–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Ricardo-Gonzalez RR, Van Dyken SJ, Schneider C, Lee J, Nussbaum JC, Liang HE, et al. Tissue signals imprint ILC2 identity with anticipatory function. Nat Immunol. 2018;19(10):1093–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Owyang AM, Zaph C, Wilson EH, Guild KJ, McClanahan T, Miller HRP, et al. Interleukin 25 regulates type 2 cytokine-dependent immunity and limits chronic inflammation in the gastrointestinal tract. Journal of Experimental Medicine. 2006;203(4):843–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Chu DK, Llop-Guevara A, Walker TD, Flader K, Goncharova S, Boudreau JE, et al. IL-33, but not thymic stromal lymphopoietin or IL-25, is central to mite and peanut allergic sensitization. Journal of Allergy and Clinical Immunology. 2013;131(1):187–200.e8. [DOI] [PubMed] [Google Scholar]
- 170.Leyva-Castillo JM, Galand C, Kam C, Burton O, Gurish M, Musser MA, et al. Mechanical Skin Injury Promotes Food Anaphylaxis by Driving Intestinal Mast Cell Expansion. Immunity. 2019;50(5):1262–75.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Han H, Thelen TD, Comeau MR, Ziegler SF. Thymic stromal lymphopoietin-mediated epicutaneous inflammation promotes acute diarrhea and anaphylaxis. Journal of Clinical Investigation. 2014;124(12):5442–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Barlow JL, Peel S, Fox J, Panova V, Hardman CS, Camelo A, et al. IL-33 is more potent than IL-25 in provoking IL-13-producing nuocytes (type 2 innate lymphoid cells) and airway contraction. Journal of Allergy and Clinical Immunology. 2013;132(4):933–41. [DOI] [PubMed] [Google Scholar]
- 173.Nakanishi W, Yamaguchi S, Matsuda A, Suzukawa M, Shibui A, Nambu A, et al. IL-33, but Not IL-25, Is Crucial for the Development of House Dust Mite Antigen-Induced Allergic Rhinitis. PLoS ONE. 2013;8(10):9–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Ricardo-Gonzalez RR, Van Dyken SJ, Schneider C, Lee J, Nussbaum JC, Liang HE, et al. Tissue signals imprint ILC2 identity with anticipatory function. Nature Immunology. 2018;19(10):1093–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Salimi M, Barlow JL, Saunders SP, Xue L, Gutowska-Owsiak D, Wang X, et al. A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. Journal of Experimental Medicine. 2013;210(13):2939–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Feng J, Li L, Ou Z, Li Q, Gong B, Zhao Z, et al. IL-25 stimulates M2 macrophage polarization and thereby promotes mitochondrial respiratory capacity and lipolysis in adipose tissues against obesity. Cellular and Molecular Immunology. 2018;15(5):493–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Hams E, Locksley RM, McKenzie ANJ, Fallon PG. Cutting Edge: IL-25 Elicits Innate Lymphoid Type 2 and Type II NKT Cells That Regulate Obesity in Mice. The Journal of Immunology. 2013;191(11):5349–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Li L, Ma L, Zhao Z, Luo S, Gong B, Li J, et al. IL-25-induced shifts in macrophage polarization promote development of beige fat and improve metabolic homeostasis in mice. PLoS Biology. 2021;19(8):1–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Wang A-J, Yang Z, Grinchuk V, Smith A, Qin B, Lu N, et al. IL-25 or IL-17E Protects against High-Fat Diet–Induced Hepatic Steatosis in Mice Dependent upon IL-13 Activation of STAT6. The Journal of Immunology. 2015;195(10):4771–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Beale J, Jayaraman A, Jackson DJ, Jonathan DR, Edwards MR, Walton RP, et al. Europe PMC Funders Group Rhinovirus induced IL-25 in asthma exacerbation drives type-2 immunity and allergic pulmonary inflammation. 2015;6(256):1–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Wilson MSM-KMM. Bone. 2012;23(1):1–7. [Google Scholar]
- 182.Han M, Rajput C, Hong JY, Lei J, Hinde JL, Wu Q, et al. The Innate Cytokines IL-25, IL-33, and TSLP Cooperate in the Induction of Type 2 Innate Lymphoid Cell Expansion and Mucous Metaplasia in Rhinovirus-Infected Immature Mice. The Journal of Immunology. 2017;199(4):1308–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Thelen TD, Green RM, Ziegler SF. Acute blockade of IL-25 in a colitis associated colon cancer model leads to increased tumor burden. Scientific Reports. 2016;6(May):1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Jou E, Rodriguez-Rodriguez N, Ferreira AF, Jolin HE, Clark PA, Sawmynaden K, et al. An innate IL-25-ILC2-MDSC axis creates a cancer-permissive microenvironment for Apc mutation-driven intestinal tumorigenesis. Sci Immunol. 2022;7(72):eabn0175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Furuta S, Jeng YM, Zhou L, Huang L, Kuhn I, Bissell MJ, et al. IL-25 causes apoptosis of IL-25R-expressing breast cancer cells without toxicity to nonmalignant cells. Science Translational Medicine. 2011;3(78). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Yin SY, Jian FY, Chen YH, Chien SC, Hsieh MC, Hsiao PW, et al. Induction of IL-25 secretion from tumour-associated fibroblasts suppresses mammary tumour metastasis. Nature Communications. 2016;7(11311). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Xu M, Lu H, Lee YH, Wu Y, Liu K, Shi Y, et al. An Interleukin-25-Mediated Autoregulatory Circuit in Keratinocytes Plays a Pivotal Role in Psoriatic Skin Inflammation. Immunity. 2018;48(4):787–98.e4. [DOI] [PubMed] [Google Scholar]
- 188.Nadjsombati MS, McGinty JW, Lyons-Cohen MR, Jaffe JB, DiPeso L, Schneider C, et al. Detection of Succinate by Intestinal Tuft Cells Triggers a Type 2 Innate Immune Circuit. Immunity. 2018;49(1):33–41.e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Schneider C, O’Leary CE, von Moltke J, Liang HE, Ang QY, Turnbaugh PJ, et al. A Metabolite-Triggered Tuft Cell-ILC2 Circuit Drives Small Intestinal Remodeling. Cell. 2018: 174(2):271–284.e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Zaph C, Du Y, Saenz SA, Nair MG, Perrigoue JG, Taylor BC, et al. Commensal-dependent expression of IL-25 regulates the IL-23-IL-17 axis in the intestine. Journal of Experimental Medicine. 2008;205(10):2191–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Buonomo EL, Cowardin CA, Wilson MG, Saleh MM, Pramoonjago P, Petri WA. Microbiota-Regulated IL-25 Increases Eosinophil Number to Provide Protection during Clostridium difficile Infection. Cell Reports. 2016;16(2):432–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Camelo A, Barlow JL, Drynan LF, Neill DR, Ballantyne SJ, Wong SH, et al. Blocking IL-25 signalling protects against gut inflammation in a type-2 model of colitis by suppressing nuocyte and NKT derived IL-13. J Gastroenterol. 2012;47(11):1198–211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Martin RK, Damle SR, Valentine YA, Zellner MP, James BN, Lownik JC, et al. B1 Cell IgE Impedes Mast Cell-Mediated Enhancement of Parasite Expulsion through B2 IgE Blockade. Cell Reports. 2018;22(7):1824–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Corren J, Parnes JR, Wang L, Mo M, Roseti SL, Griffiths JM, et al. Tezepelumab in Adults with Uncontrolled Asthma. N Engl J Med. 2017;377(10):936–46. [DOI] [PubMed] [Google Scholar]
- 195.Menzies-Gow A, Corren J, Bourdin A, Chupp G, Israel E, Wechsler ME, et al. Tezepelumab in Adults and Adolescents with Severe, Uncontrolled Asthma. N Engl J Med. 2021;384(19):1800–9. [DOI] [PubMed] [Google Scholar]
- 196.Simpson EL, Parnes JR, She D, Crouch S, Rees W, Mo M, et al. Tezepelumab, an anti-thymic stromal lymphopoietin monoclonal antibody, in the treatment of moderate to severe atopic dermatitis: A randomized phase 2a clinical trial. J Am Acad Dermatol. 2019;80(4):1013–21. [DOI] [PubMed] [Google Scholar]
- 197.Rabe KF, Celli BR, Wechsler ME, Abdulai RM, Luo X, Boomsma MM, et al. Safety and efficacy of itepekimab in patients with moderate-to-severe COPD: a genetic association study and randomised, double-blind, phase 2a trial. Lancet Respir Med. 2021;9(11):1288–98. [DOI] [PubMed] [Google Scholar]
- 198.Wechsler ME, Ruddy MK, Pavord ID, Israel E, Rabe KF, Ford LB, et al. Efficacy and Safety of Itepekimab in Patients with Moderate-to-Severe Asthma. N Engl J Med. 2021;385(18):1656–68. [DOI] [PubMed] [Google Scholar]
- 199.Chen YL, Gutowska-Owsiak D, Hardman CS, Westmoreland M, MacKenzie T, Cifuentes L, et al. Proof-of-concept clinical trial of etokimab shows a key role for IL-33 in atopic dermatitis pathogenesis. Sci Transl Med. 2019;11(515). [DOI] [PubMed] [Google Scholar]
- 200.Chinthrajah S, Cao S, Liu C, Lyu SC, Sindher SB, Long A, et al. Phase 2a randomized, placebo-controlled study of anti-IL-33 in peanut allergy. JCI Insight. 2019;4(22). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Yousuf AJ, Mohammed S, Carr L, Yavari Ramsheh M, Micieli C, Mistry V, et al. Astegolimab, an anti-ST2, in chronic obstructive pulmonary disease (COPD-ST2OP): a phase 2a, placebo-controlled trial. Lancet Respir Med. 2022;10(5):469–77. [DOI] [PubMed] [Google Scholar]
- 202.Kelsen SG, Agache IO, Soong W, Israel E, Chupp GL, Cheung DS, et al. Astegolimab (anti-ST2) efficacy and safety in adults with severe asthma: A randomized clinical trial. J Allergy Clin Immunol. 2021;148(3):790–8. [DOI] [PubMed] [Google Scholar]
- 203.Villarreal DO, Wise MC, Walters JN, Reuschel EL, Choi MJ, Obeng-Adjei N, et al. Alarmin IL-33 acts as an immunoadjuvant to enhance antigen-specific tumor immunity. Cancer Res. 2014;74(6):1789–800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Camelo A, Rosignoli G, Ohne Y, Stewart RA, Overed-Sayer C, Sleeman MA, et al. IL-33, IL-25, and TSLP induce a distinct phenotypic and activation profile in human type 2 innate lymphoid cells. Blood Adv. 2017;1(10):577–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.von Moltke J, O’Leary CE, Barrett NA, Kanaoka Y, Austen KF, Locksley RM. Leukotrienes provide an NFAT-dependent signal that synergizes with IL-33 to activate ILC2s. J Exp Med. 2017;214(1):27–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Cardoso V, Chesne J, Ribeiro H, Garcia-Cassani B, Carvalho T, Bouchery T, et al. Neuronal regulation of type 2 innate lymphoid cells via neuromedin U. Nature. 2017;549(7671):277–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Macian F, Lopez-Rodriguez C, Rao A. Partners in transcription: NFAT and AP-1. Oncogene. 2001;20(19):2476–89. [DOI] [PubMed] [Google Scholar]
- 208.Van Dyken SJ, Mohapatra A, Nussbaum JC, Molofsky AB, Thornton EE, Ziegler SF, et al. Chitin activates parallel immune modules that direct distinct inflammatory responses via innate lymphoid type 2 and gammadelta T cells. Immunity. 2014;40(3):414–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Vannella KM, Ramalingam TR, Borthwick LA, Barron L, Hart KM, Thompson RW, et al. Combinatorial targeting of TSLP, IL-25, and IL-33 in type 2 cytokine-driven inflammation and fibrosis. Sci Transl Med. 2016;8(337):337ra65. [DOI] [PubMed] [Google Scholar]


