Abstract
Background
Language regression, broadly defined as the loss of acquired language skills in early childhood, is a distinctive feature of autism. Little is known about the factors underlying regression or the prognosis of children who exhibit regression. We examine potential predictors of language regression and test its association with language development in a prospective longitudinal sample of children with autism spectrum disorder (ASD) from diagnosis to age 10 years.
Methods
We analysed data from Pathways in ASD, a prospective longitudinal study of 421 children enrolled around the time of an autism diagnosis between 2 and 5 years. Autism Diagnostic Interview‐Revised data were available for 408 children, of whom 90 (22%) were classified as having language regression.
Results
Demographic and other health factors including caregiver education, family income, child sex, reported seizures, and age of enrolment did not differ between children with and without language regression. Children with language regression walked earlier and attained first words sooner than those without regression. However, both groups attained phrase speech at comparable ages. Those with regression exhibited greater delays in expressive and receptive communication over the follow‐up period, although this effect was attenuated when accounting for baseline differences in motor and cognitive ability. Overall, those with language regression continued to exhibit expressive but not receptive communication delay compared to those without regression. Communication trajectories were heterogeneous to age 10 years, irrespective of regression status.
Conclusions
Although language regression can be alarming, our findings confirm that its occurrence does not necessarily foreshadow worse developmental outcomes relative to those without regression. Although a discrepancy in age‐equivalent communication skills may persist, this can be expected to be of less practical importance with rising average levels of skills. Future studies need to account for the significant variability in language trajectories by considering factors beyond developmental regression.
Keywords: Language, autism, development
Introduction
The acquisition of language and communication milestones is highly variable in children with neurodevelopmental conditions like autism spectrum disorder (ASD). Delays in expressive language are among the signs for ASD that are amplified over time into widespread delays and atypicality in other domains (Elsabbagh, 2020; Lord, Luyster, Guthrie, & Pickles, 2012; Visser et al., 2017). Over time, children with ASD also exhibit highly variable language profiles – such that some develop fluent language and some never acquire phrase speech, while others who have delayed onset of words and phrase speech catch up later in childhood (Szatmari et al., 2000; Tager‐Flusberg, 2016).
Language regression is characterized as a loss of previously acquired words or a stagnation of language development (Barger, Campbell, & McDonough, 2013; Ozonoff & Iosif, 2019), and there is some evidence that regression defined as language loss is specific to ASD (Lord, Shulman, & DiLavore, 2004). Regression may be seen in other disorders, such as early‐onset acquired epileptic aphasia in children with rolandic epilepsy (Deonna & Roulet‐Perez, 2010), but overall the clinical manifestations of regression reported in epilepsy differs from that reported in ASD (Tuchman, 2017). Moreover, language loss is rarely reported on the Autism Diagnostic Interview (ADI) among those with specific language impairment (Pickles et al., 2009).
The prevalence of regression among children with ASD largely depends on the type of study conducted; more recent studies with at least partially prospective data suggest that regression may be more common in ASD than is retrospectively reported. Studies using parent surveys report an average prevalence of 41%, clinical samples report an average prevalence of 34%, and population samples report an average of 22% (Barger et al., 2013). Other forms of regression affecting social and adaptive functioning have also been reported in autism among other conditions (Barger et al., 2013). Yet, no specific associations have been identified between language regression and genetic factors or autism symptom severity (Jones & Campbell, 2010; Tammimies, 2019).
The association between regression and later development in various domains has been inconsistent across studies, in part attributed to a reliance on retrospective parent reports. As regression occurs early in life, often prior to diagnosis, the reliability of parent recall has been questioned. Hus and colleagues, comparing repeated Autism Diagnostic Interview‐Revised (ADI‐R) interviews for language milestones, reported ‘telescoping’ with the consequence that more children met language delay criteria as they grow older, in spite of the original parent report that their language was not delayed (Hus, Taylor, & Lord, 2011). Older studies tended to report that a regression profile is associated with worse functioning at the time of recall (Hoshino et al., 1987; Kurita, 1985; Rogers & DiLalla, 1990), slow and atypical speech development (Ballaban‐Gil, Rapin, Tuchman, & Shinnar, 1996; Gillberg & Steffenburg, 1987), or never reaching complete functional speech (Kurita, 1985; Wilson, Djukic, Shinnar, Dharmani, & Rapin, 2003). In contrast, more recent evidence points to considerable catch‐up among those with regression in their subsequent language development (Boterberg, Charman, Marschik, Bölte, & Roeyers, 2019; Fountain, Winter, & Bearman, 2012; Pickles, Anderson, & Lord, 2014; Williams, Brignell, Prior, Bartak, & Roberts, 2015; Wodka, Mathy, & Kalb, 2013).
Prospective longitudinal studies of infant siblings have addressed problems in parental recall and offered opportunities for direct comparisons between infants who later develop autism versus those who do not (Ozonoff & Iosif, 2019). They revealed a more complex picture, raising the possibility that the diverging trajectories in most infants who later develop ASD may be the same phenomenon that parents of older children retroactively label as ‘regression’. In both direct assessments and concurrent parent reports of social communication skills, infant siblings who later developed ASD and those who did not were indistinguishable in social communication skills at 6 months (e.g., gaze to face, smiles paired with eye contact, and/or vocalizations). The two groups displayed diverging trajectories thereafter, with the ASD group reaching a plateau or falling behind in social communication skills. By 12 months, the differences were significant, with the gap continuing to widen through to 36 months (Ozonoff & Iosif, 2019). In another study, measuring prospective trajectories of cognitive development, Brian et al. (2014) showed evidence for skill loss or plateau in infants with a family history of ASD that was not specific to those later diagnosed with ASD. Prospective studies suggest that parents may be more able to identify regression in cases in which the child has already developed some skills that are easy for parents to observe but not in cases where the child is experiencing delays in developmental milestones (Ozonoff & Iosif, 2019; Pickles et al., 2009).
As such, it is possible that language regression in ASD, defined as early language loss, may be more the rule than the exception, as one manifestation of atypical brain development impacting multiple domains, including early social communication and motor development. Moreover, operational definitions of regression in ASD may conflate potentially distinct phenomena, namely losing previously acquired skills, a plateau (or a stagnation) in skill acquisition, or a slower rate of skill acquisition. More studies are needed to address key knowledge gaps related to regression in ASD and how it interacts with the variable rates of language and communication development. In the current study, we analysed data from a large cohort of children with ASD followed up since diagnosis to address three objectives. First, we examined the prevalence, timing, and correlates of regression, defined as parent‐reported language loss occurring prior to 7 years of age. Second, we contrasted profiles of expressive and receptive communication in children with and without a regression profile from the time of diagnosis until around 10 years of age. Finally, we tested the potential influence of socio‐demographic predictors (biological sex, maternal education, family income) and developmental predictors (fine motor skills, cognitive skills), known to be associated with trajectories of development of expressive and receptive communication.
Methods
Participants
The Pathways in ASD study is a large (N = 421) inception cohort of children with ASD who were recruited at the time of diagnosis (between aged 2 and 5 years). The sample has been repeatedly assessed to track their developmental, cognitive, and socioemotional functioning across childhood and adolescence (see https://asdpathways.ca/). To determine eligibility for inclusion in the study, children and their caregivers participated in intake assessments at regional ASD assessment sites affiliated with academic institutions in five Canadian provinces (Nova Scotia, Quebec, Ontario, Alberta, and British Columbia). Approved by the Research Ethics Board at each site, eligible participants received a diagnosis of ASD by clinicians with ASD expertise using DSM‐IV‐TR criteria (American Psychiatric Association, 2000) and both the Autism Diagnostic Observation Scale (ADOS; Lord et al., 2000) and the ADI‐R (Lord, Rutter, & Le Couteur, 1994). Parents provided informed consent before completing any study measures or assessments. Children with a diagnosis of cerebral palsy or other neuromotor disorders, identified genetic or chromosomal abnormalities, or significantly impaired vision or hearing were excluded from the study. Caregivers were also required to be verbally proficient in English (or French, in Quebec). The data included in this report were collected at eight assessment points between recruitment (time 1) and age 10–11 years (time 8).
Measures
The ADI‐R provides a framework for systematic parent report of early milestones and regression of language and other skills. Parents completed the ADI‐R at times 1, 4, and 8, when the children were on average 3.33 years (SD = 0.77), 6.62 years (SD = 0.34), and 10.74 years (SD = 0.23), respectively. The time 1 interview was used for age of first walking (question 5), first words (question 9), and first phrases (question 10). For any children who had not attained phrase speech by the time 1 interview, the milestones were captured from the time 4 interview instead.
We defined language regression on the basis of questions 4 and 11 on the ADI‐R. This considers language regression as loss of a language skill, specifically communicative use of at least five words, which was previously mastered with words used on a daily basis for at least 3 months prior to the loss. Age at which regression occurred was taken as the earliest reported at any of the three available assessments (median age of loss 18 months, IQR 15–24 months).
The Vineland Adaptive Behavior Scales – Second Edition (VABS‐II; Sparrow, Balla, & Cicchetti, 2005) semi‐structured parent interview assesses adaptive functioning. To define trajectories of development across various domains of interest, we used the VABS‐II age‐equivalent and v‐scale subdomain scores for expressive communication, receptive communication, and socialization at times when the children were on average 3.3, 4.0, 4.5, 6.6, 8.7, and 10.8 years of age, respectively. Visual inspection of scatter plots of age‐equivalent scores against age showed clear evidence of arbitrary ceiling scores. We trimmed 17 (1%) of expressive and 55 (3%) of receptive scores to the upper bound of the main distribution of 1.6 times actual age. Scores from the VABS‐II taken at times when the language loss status of a child was uncertain – for example, when one ADI‐R reports loss with an age of onset before a previous ADI‐R that reported no loss as having occurred – were set to missing to avoid them contributing to the findings. This was the case for 8.9% (157/1,769) of the VABS‐II assessments. We also constructed a time‐varying variable for time since loss, equal to the difference in time between the reported age at loss and the current assessment age (coded zero for those without reported prior loss).
To examine potential factors associated with regression, a set of variables was extracted from the measures of the first assessment soon after diagnosis. We captured biological sex, child age at enrolment, family income, primary caregiver’s age and education, and history of seizures.
Fine motor and cognitive skills were assessed using the standard scores from the researcher‐administered Merrill–Palmer–Revised Scales of Development (M–P–R; Roid & Sampers, 2004) for use with preschoolers. Non‐zero raw scores that fell below the usual standard score floor (n = 34 fine motor and n = 24 cognitive) were assigned the minimum standard score of 10. The associations between M–P–R cognitive, M–P–R fine motor, VABS‐II expressive language, and VABS‐II receptive language at the baseline assessment are reported in Table S1.
Statistical analysis
Analysis was undertaken in Stata v17.1 (StataCorp., 2021). A logistic regression was used to examine variables associated with regression loss. The distribution of the ages of language milestone acquisition and walking was analysed using Kaplan–Meier survival plots with estimates of the hazard ratio (HR) associated with the regression group obtained from Cox models (sts and stcox commands). Growth curve models were fitted by maximum likelihood to the repeated measures of VABS‐II expressive communication and receptive communication age‐equivalent scores using an xtmixed model with correlated random effects for intercept and linear slope for age at assessment. The measure‐specific (level‐1) error variance was allowed to be heteroscedastic over six age categories. The fixed part of the model included linear and quadratic terms in age at assessment and additional variables associated with specific hypotheses and confounders. A dummy variable for language regression was used to assess the association with later communication level, and an interaction with age for an association with the rate of change or development of communication. All confidence intervals are 95% and p‐values are from 2‐tailed Wald tests. Maximum likelihood estimated coefficients are unbiased for selective attrition related to any model covariates and observed reports of communication skills. The STATA code for generating the Kaplan–Meier plot and growth curve model can be viewed in Appendices S1 and S2, respectively.
Results
Prevalence and correlates of regression
Of the 421 children recruited, ADI‐R parent interviews provided milestone and regression responses for 408 children, of whom 90 were reported to have loss of language skills, henceforth called the ‘regression subgroup’. Descriptive statistics are shown in Table 1. Of these regressions, 86 occurred before the family entered the study. Thus, variables that we examine for their association with regression were, for the most part, assessed after the putative age when loss occurred.
Table 1.
Descriptive measures from assessment when first recruited by regression loss and no‐loss groups
| Variable | No regression group | Regression group | p‐Value | ||||
|---|---|---|---|---|---|---|---|
| n | Mean (SD) | Min/Max | n | Mean (SD) | Min/Max | ||
| Biological sex (M:F) | 264:54 | – | 80:10 | – | .176 a | ||
| Family income | 289 | $70 k | $40 k/>$80 k | 79 | $70 k | $50 k/>$80 k | .802 a |
| Primary caregivers’ education | 297 | Diploma/Trade Certificate | Some Comm. College or Nursing /undergrad | 82 | Community College/Diploma | Some trade/undergrad | .430 b |
| Age at first assessment (months) | 311 | 40.1 (9.3) | 23/62 | 89 | 40.1 (7.7) | 25/61 | .903 c |
| History of seizures | 297 | 5.7% | – | 78 | 12.8% | .031 a | |
| Site | |||||||
| Halifax | 45 | 14.1% | – | 10 | 11.1% | – | .038 a |
| Montreal | 100 | 31.5% | 33 | 36.7% | |||
| Hamilton | 51 | 16.0% | 66 | 16.7% | |||
| Vancouver | 78 | 24.5% | 89 | 12.2% | |||
| Edmonton | 44 | 13.8% | 65 | 23.3% | |||
| Autism Diagnostic Interview‐Revised | |||||||
| n | Median | IQR | n | Median | IQR | ||
| Age at loss (months) | NA | NA | NA | 90 | 18 | 15/24 | |
| Age first walked (months) | 317 | 13 | 12/15 | 90 | 12 | 11/14 | <.001 c |
| Age first words (months) | 317 | 24 | 15/36 | 88 | 12.5 | 12/18 | <.001 c |
| Age phrase speech (months) | 318 | 39 | 30/66 | 90 | 39.5 | 24/75 | .568 |
| Merrill–Palmer–Revised | |||||||
| n | Mean (SD) | Min/Max | n | Mean (SD) | Min/Max | ||
| Merrill–Palmer–Revised Fine <Motor standard scores | 299 | 64.9 (28.8) | 10/143 | 90 | 53.1 (27.6) | 10/121 | <.001 d |
| Merrill–Palmer–Revised cognitive standard scores | 299 | 59.0 (27.2) | 10/154 | 90 | 46.8 (23.8) | 10/100 | <.001 d |
| Vineland Adaptive Behavior Scales‐II | |||||||
| n | Mean (SD) | Min/Max | n | Mean (SD) | Min/Max | ||
| 1st expressive communication age equivalent (months) | 311 | 21.5 (12.3) | 1/67 | 89 | 17.9 (9.4) | 2/47 | .012 d |
| 1st receptive communication age equivalent (months) | 311 | 21.8 (14.1) | 1/87 | 89 | 17.2 (9.0) | 1/43 | .004 d |
| 1st social interaction v‐scale score | 311 | 9.4 (2.0) | 3/15 | 89 | 9.0 (1.8) | 5/14 | .042 d |
Pearson chi‐square.
Ordinal logistic Wald test.
Wilcoxon test.
t‐test.
Reported loss was associated with few background demographic factors, though it did vary by site (see Table 1). Clearer differences emerged when we compared those with and without regression on the parent‐reported VABS‐II around the time of diagnosis. Those with regression had lower scores on expressive communication (effect size (ES) = −.36), receptive communication (ES = −.38), and had lower standard scores on the M–P–R fine motor (ES = −.45) and cognitive (ES = −.44) subscales relative to those without regression.
The Kaplan–Meier plots (Figure 1) show that the language regression group (n = 90) generally walked somewhat earlier (HR 1.50, p = .001), said their 1st word sooner (HR = 2.38, p < .001), and attained phrase speech at times comparable to those without regression (n = 318; HR = 1.02, p = .863).
Figure 1.
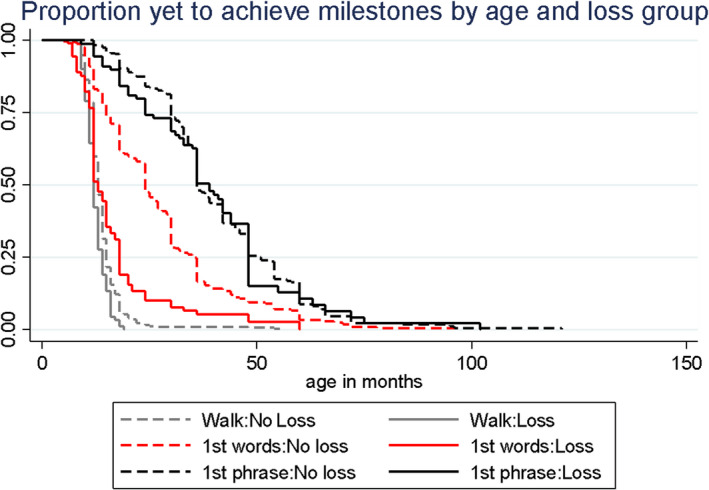
Proportion yet to achieve milestones by age and loss group
Association with regression
Growth curve models were fitted to 1,768 VABS‐II Communication scale assessments from 403 study participants. The model estimates shown in Table 2 included a dummy variable for language regression to estimate any persisting discrepancy in VABS‐II Communication. Language regression was significantly associated with the later level of expressive communication, the regression group estimated as having an overall expressive communication deficit of 3.28 months (CI 1.42–5.14, p < .001) compared to the remainder of the sample. The corresponding group difference for receptive communication was 3.20 months (CI 0.82, 5.82) and significant (p = .00916). Additional tests of the interaction of age and loss, examining whether the discrepancy changed with age, were both entirely non‐significant (p = .579 expressive; p = .990 receptive).
Table 2.
Mixed model estimates for effect of regression on expressive and receptive language
| Expressive communication (age equivalent in months) | Receptive communication (age equivalent in months) | |||||
|---|---|---|---|---|---|---|
| Coefficient | 95% CI | p‐Value | Coefficient | 95% CI | p‐Value | |
| Fixed part | ||||||
| Age (years) | 12.49 | 11.38, 13.59 | <.001 | 10.52 | 8.83, 12.20 | <.001 |
| Age‐sq | −0.41 | −0.50, −0.32 | <.001 | −0.15 | −0.29, 0.00 | .044 |
| Regression group | −3.28 | −5.14, −1.42 | .001 | −3.32 | −5.82, −0.82 | .009 |
| Random parts | ||||||
| Level 2 | ||||||
| Age (SD) | 4.33 | 3.94, 4.77 | 5.41 | 4.84, 6.03 | ||
| Intercept (SD) | 9.90 | 8.54, 11.48 | 15.10 | 13.00, 17.53 | ||
| Correlation | −0.80 | −0.85, −0.74 | −0.85 | −0.89, −0.79 | ||
| Level 1 | ||||||
| Age gp1 (SD) | 3.10 | 5.22 | ||||
| Age gp2 (SD) | 3.32 | 6.56 | ||||
| Age gp3 (SD) | 5.72 | 9.20 | ||||
| Age gp4 (SD) | 10.32 | 13.46 | ||||
| Age gp5 (SD) | 12.58 | 19.60 | ||||
| Age gp6 (SD) | 23.33 | 36.02 | ||||
Bold values represent significant at (p < .05).
Although Figure 2 demonstrates an overall deficit in the whole sample, there was considerable variability in the trajectories of communication skills of children, both with and without language regression. As a reflection of this variability in outcomes, by the time children reached 10 years of age, the model estimated 6% (24/403) had developed at or above average norms for expressive communication, with no difference between the regression group (8%; 7/90) and the non‐regression group (6%; 19/313). For receptive communication, 14% (58/403) had developed at or above average norms by age 10 years, 19% (17/90) for those who had experienced language regression, and 13% (41/313) for those who had not. Bolded values reperesent significance at p < .01
Figure 2.

Individual and group mean trajectories of parent‐reported expressive and receptive communication
Socio‐demographic confounders
Further model estimates for associations with communication level are shown in Tables S1 and S2. Additional terms involving interactions with age‐at‐assessment were used to examine effects on communication growth. Model 1 included socio‐demographic confounders. The level of expressive language was associated with site (4 df p < .001) and positively with household income (p < .001) but neither with biological sex nor maternal education. Higher income was additionally associated with growth in scores (p = .024). However, the inclusion of these potential confounders did not reduce the estimated deficit of the regression group (3.19 months, CI 1.27–5.10; p = .001).
For receptive communication, only family income (p ≤ .001) showed a significant association with the level of receptive communication (biological sex p = .443, maternal education p = .827, site 4 df p = .196) and none of the socio‐demographic predictors showed a significant association with rate of growth. The estimated deficit of the regression group remained significant (3.22, p = .016) on inclusion of these predictors. Higher income was additionally associated with growth in scores (p = .024).
Motor and cognitive predictors
Model 2 included early fine motor and cognitive skills at baseline. Fine motor skill was significantly associated with expressive communication level (p = .029) and additionally to growth (p = .035), while baseline cognitive skill was significantly associated only with level (p = .001 and .092). In contrast to socio‐demographic predictors, the inclusion in the model of cognitive predictors of communication level substantially reduced the expressive communication deficit of the regression group to a non‐significant 1.33 months (CI 0.33–3.00; p = .116). For receptive communication, early cognitive skills, but not fine motor skills, were related to the level of receptive communication (p = .001 and .267, respectively). Here too, their inclusion reduced the estimated deficit in receptive communication of the regression group to 0.00 months (CI −2.81 to 2.81; p = .999).
Effects of age
Model 3 showed that while age at recruitment to the cohort was associated with lower expressive and receptive language (p’s < .001) adjustment for age at recruitment did not change the estimates or significance of the deficits in language level in the regression group. The further addition of the interaction of regression with assessment age to allow the deficit to change with time proved non‐significant for both expressive (p = .298) and receptive communication (p = .597). In Model 4, in addition to age at recruitment and the main effect of regression, two interaction terms were examined that allowed the size of deficit in communication of the regression group to vary with the reported age at regression and with the time since the regression (these terms being set to zero for the non‐regression group). For expressive communication, Wald tests for the two interaction terms were neither individually nor jointly significant (2 df p = .195) indicating neither of these timing factors moderated the effect of language regression on the level of the deficit. This was also the case for receptive communication (2 df p = .621).
Discussion
Previous studies have characterized those children who experience language regression as being at the extreme end of a continuum of loss (masked in the subgroup with language delay, because language regression cannot be observed if the child is not yet speaking), a subtype of autism, or alternatively a distinct disorder (Grzadzinski, Huerta, & Lord, 2013; Ozonoff, Heung, Byrd, Hansen, & Hertz‐Picciotto, 2008; Pickles et al., 2009; Rogers, 2004). Moreover, factors associated with regression and/or those impacting how parents report it may include specific risk factors for ASD or may well overlap with the factors contributing to variation in typical language development. Finally, evidence is mixed with respect to outcomes of children with ASD who experience regression versus those who do not.
We explored the association between language regression and developmental outcomes in the Pathways in ASD cohort. We found that 22% of children in the sample were reported as having early language regression, a rate comparable to those reported in previous studies with population‐based samples (Barger et al., 2013). In line with previous findings (Pickles et al., 2009), regression was more commonly reported among the early developers of single‐word speech. In fact, the median age of first words in those with reported regression was almost 1 year earlier than those without reported regression. While a causal relationship between earlier speech and risk of loss cannot be ruled out, the findings are also consistent with an explanation involving censored observation, since regression loss requires speech prior to loss before it can be reported.
This relatively accelerated development was not specific to language; those with the regression profile also began to walk slightly earlier, approximately 1 month before those without regression. Yet, both groups with and without regression eventually attained phrase speech around the same time. Reported regression was associated with a history of seizures and with greater early fine motor and cognitive skills around the time of diagnosis but not with socio‐demographic predictors.
The salience of regression to parents may depend on the nature and timing of various developmental milestones; parents are more likely to observe and subsequently report loss for earlier emerging and more easily observed milestones like early words. Our findings corroborate previous studies and reviews with approximately 22% of our sample experiencing language regression, and this is less commonly reported in a subgroup with slow development in the first year. These findings are consistent with language regression being part of broader underlying biological changes impacting brain development across domains, where the onset of regression coincides with and may indeed reflect multiple atypicalities across different neural systems documented in infants who later develop ASD (Elsabbagh, 2020; Levin, Varcin, O’Leary, Tager‐Flusberg, & Nelson, 2017; Wilkinson et al., 2019).
Children with and without reported regression followed the same overall developmental trajectory of expressive and receptive communication. While some caution is required in the interpretation of age equivalents over extended age ranges, the regression profile was associated on average with a 3‐month deficit in both expressive and receptive communication that neither grew nor dissipated. The deficits were neither associated with the specific age at which regression was reported nor the time elapsed since the regression occurred. We further confirmed that reported regression had little impact on later attainment of typical levels of communication. By 11 years of age, while regression was still associated with a deficit, the practical relevance of a discrepancy of a few months declines as the average age‐equivalent communication level increases.
Cognitive and motor skills were tightly linked with receptive and expressive communication levels across the whole sample. Accounting for early motor skills removed the language deficit associated with language regression. This could be a measurement issue, as it is plausible that having low receptive language directly influences the assessment of a child’s motor skills, in terms of their ability to understand and follow instructions or their reduced use of communicative gestures. Their observed motor score may thus reflect, at least to some degree, underlying language ability. Motor and language correlations may also reflect a true shared general ability. In this case, the fact that controlling for motor skills removes the significant association with language suggests that language regression is indicative of domain general difficulties and is not specific to language outcomes. Nevertheless, while cautious interpretation is clearly important, these findings are consistent with previous studies where early motor development has been shown to predict rates of language development in children with autism (Bal et al., 2020; Bedford, Pickles, & Lord, 2016; Manelis et al., 2020) and language regression is often preceded or accompanied by delays in non‐speech social communication (Ozonoff, Williams, & Landa, 2005; Werner & Dawson, 2005).
Pathways in ASD is a comparatively large sample of children followed from the time of ASD diagnosis, providing rich prospective and repeated measurements. The method of sample ascertainment means that measurement describing pre‐diagnosis development, which typically includes language regression, must be obtained by retrospective reporting. It is possible that subtle regressions may have been missed, particularly for children who did not develop the requisite language required for a loss to be reported. However, unlike most studies of regression, the ADI‐R interviews were undertaken close in time to the reported regressions. Although the development of communication skills was from parent‐reported measures, such measures have been shown to correlate very highly with directly assessed measurement and to yield very similar growth curves (Taylor, Pickering, Lord, & Pickles, 1998). Significant attrition from the cohort has been unavoidable albeit lower than usually observed in other longitudinal studies. Moreover, attrition had minimal impact on reports of language loss that were essentially complete and the growth curve models took into account any selective attrition based on measured family demographics, early child motor and cognition skills, and trajectory of communication skills development.
Conclusion
In sum, although language regression can be alarming in the subgroup with otherwise near‐typical development, our findings confirm that its occurrence does not necessarily foreshadow worse long‐term developmental outcomes. Our trajectory analyses highlight the marked variability in communication trajectories in children with autism, both with and without regression. Variability in communication outcomes in our sample was associated with some of the factors known to impact rates of language development in the general population, such as income. Future research identifying modifiable factors linked to the external environment could prove especially useful in promoting early language as a protective mechanism for subsequent development. Our study focused on language regression because language loss has received the most interest in ASD, but future studies should assess possible loss in multiple domains using more refined measures.
Supporting information
Table S1. Expressive language growth curve model: fixed effect coefficient estimates.
Table S2. Receptive language growth curve model: fixed effect coefficient estimates.
Appendix S1. Code for generating Kaplan–Meier plot.
Appendix S2. Code for generating Table S1 expressive language analysis.
Acknowledgements
The authors acknowledge funding by The Canadian Institutes of Health Research, Fonds de Recherche du Québec, Kids Brain Health Network (formerly NeuroDevNet), Autism Speaks (US), Government of British Columbia, Alberta Innovates Health Solutions, and the Sinneave Family Foundation. The authors also thank all the families who participated in the Pathways in ASD study. A.P. received additional support from the NIHR KCL/South London and Maudsley NHS Foundation Trust Biomedical Research Centre and Senior Investigator Award NF‐SI‐0617‐10120. R.B. was supported by a King’s Prize Fellowship (204823/Z/16/Z). The authors also acknowledge the past and current members of the Pathways in ASD Study Team.
Data sharing: Due to ethical constraints supporting data cannot be made openly available. Supporting data are available to bona fide researchers on approval of an application for access. Further information about the data and conditions for access can be obtained by contacting the PI Dr. Peter Szatmari (peter.szatmari@utoronto.ca).
The authors have declared that they have no competing or potential conflicts of interest.
Key points.
Evidence on the predictors of and outcomes from language regression has been mixed.
In a prospective study of children with autism assessed from diagnosis (at age 2–5 years) to age 10 years, we showed that language regression had a lasting association with lower expressive but not receptive communication skills.
There was significant variability in language development in children both with and without regression, and both groups attained phrase speech at comparable ages.
Early cognitive and motor skills were associated with regression and with later communication skills.
We conclude that while language regression is associated with an age‐equivalent delay in expressive communication, this may be of less practical importance with rising average level of skills.
Conflict of interest statement: No conflicts declared.
References
- American Psychiatric Association . (2000). Diagnostic and Statistical Manual of Mental Disorders (DSM‐5) (4th edn). Washington, DC: American Psychiatric Association Publishing. [Google Scholar]
- Bal, V.H. , Fok, M. , Lord, C. , Smith, I.M. , Mirenda, P. , Szatmari, P. , … & Zaidman‐Zait, A. (2020). Predictors of longer‐term development of expressive language in two independent longitudinal cohorts of language‐delayed preschoolers with Autism Spectrum Disorder. Journal of Child Psychology and Psychiatry, 61, 826–835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballaban‐Gil, K. , Rapin, I. , Tuchman, R. , & Shinnar, S. (1996). Longitudinal examination of the behavioral, language, and social changes in a population of adolescents and young adults with autistic disorder. Pediatric Neurology, 15, 217–223. [DOI] [PubMed] [Google Scholar]
- Barger, B. , Campbell, J. , & McDonough, J.D. (2013). Prevalence and onset of regression within autism spectrum disorders: A meta‐analytic review. Journal of Autism and Developmental Disorders, 43, 817–828. [DOI] [PubMed] [Google Scholar]
- Bedford, R. , Pickles, A. , & Lord, C. (2016). Early gross motor skills predict the subsequent development of language in children with autism spectrum disorder. Autism Research, 9, 993–1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boterberg, S. , Charman, T. , Marschik, P. , Bölte, S. , & Roeyers, H. (2019). Regression in autism spectrum disorder: A critical overview of retrospective findings and recommendations for future research. Neuroscience and Biobehavioral Reviews, 102, 24–55. [DOI] [PubMed] [Google Scholar]
- Brian, A.J. , Roncadin, C. , Duku, E. , Bryson, S.E. , Smith, I.M. , Roberts, W. , … & Zwaigenbaum, L. (2014). Emerging cognitive profiles in high‐risk infants with and without autism spectrum disorder. Research in Autism Spectrum Disorders, 8(11), 1557–1566. 10.1016/j.rasd.2014.07.021 [DOI] [Google Scholar]
- Deonna, T. , & Roulet‐Perez, E. (2010). Early‐onset acquired epileptic aphasia (Landau–Kleffner syndrome, LKS) and regressive autistic disorders with epileptic EEG abnormalities: The continuing debate. Brain and Development, 32, 746–752. [DOI] [PubMed] [Google Scholar]
- Elsabbagh, M. (2020). Linking risk factors and outcomes in autism spectrum disorder: Is there evidence for resilience? BMJ, 368, l6880. [DOI] [PubMed] [Google Scholar]
- Fountain, C. , Winter, A. , & Bearman, P. (2012). Six developmental trajectories characterize children with autism. Pediatrics, 129(5), e1112–e1120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillberg, C. , & Steffenburg, S. (1987). Outcome and prognostic factors in infantile autism and similar conditions: A population‐based study of 46 cases followed through puberty. Journal of Autism and Developmental Disorders, 17, 273–287. [DOI] [PubMed] [Google Scholar]
- Grzadzinski, R. , Huerta, M. , & Lord, C. (2013). DSM‐5 and autism spectrum disorders (ASDs): An opportunity for identifying ASD subtypes. Molecular Autism, 4, 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoshino, Y. , Kaneko, M. , Yashima, Y. , Kumashiro, H. , Volkmar, F.R. , & Cohen, D.J. (1987). Clinical features of autistic children with setback course in their infancy. The Japanese Journal of Psychiatry and Neurology, 41, 237–245. [DOI] [PubMed] [Google Scholar]
- Hus, V. , Taylor, A. , & Lord, C. (2011). Telescoping of caregiver report on the Autism Diagnostic Interview – Revised. Journal of Child Psychology and Psychiatry, 52(7), 753–760. 10.1111/j.1469-7610.2011.02398.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones, L. , & Campbell, J. (2010). Clinical characteristics associated with language regression for children with autism spectrum disorders. Journal of Autism and Developmental Disorders, 40, 54–62. [DOI] [PubMed] [Google Scholar]
- Kurita, H. (1985). Infantile autism with speech loss before the age of thirty months. Journal of the American Academy of Child Psychiatry, 24, 191–196. [DOI] [PubMed] [Google Scholar]
- Levin, A. , Varcin, K. , O'Leary, H. , Tager‐Flusberg, H. , & Nelson, C.A. (2017). EEG power at 3 months in infants at high familial risk for autism. Journal of Neurodevelopmental Disorders, 9, 34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lord, C. , Luyster, R. , Guthrie, W. , & Pickles, A. (2012). Patterns of developmental trajectories in toddlers with autism spectrum disorder. Journal of Consulting and Clinical Psychology, 80, 477–489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lord, C. , Risi, S. , Lambrecht, L. , Cook, E.H., Jr , Leventhal, B.L. , DiLavore, P.C. , … & Rutter, M. (2000). The Autism Diagnostic Observation Schedule—Generic: A standard measure of social and communication deficits associated with the spectrum of autism. Journal of Autism and Developmental Disorders, 30, 205–223. [PubMed] [Google Scholar]
- Lord, C. , Rutter, M. , & Le Couteur, A. (1994). Autism Diagnostic Interview‐Revised: A revised version of a diagnostic interview for caregivers of individuals with possible pervasive developmental disorders. Journal of Autism and Developmental Disorders, 24, 659–685. [DOI] [PubMed] [Google Scholar]
- Lord, C. , Shulman, C. , & DiLavore, P. (2004). Regression and word loss in autistic spectrum disorders. Journal of Child Psychology and Psychiatry, 45, 936–955. [DOI] [PubMed] [Google Scholar]
- Manelis, L. , Meiri, G. , Ilan, M. , Flusser, H. , Michaelovski, A. , Faroy, M. , … & Menashe, I. (2020). Language regression is associated with faster early motor development in children with autism spectrum disorder. Autism Research, 13, 145–156. [DOI] [PubMed] [Google Scholar]
- Ozonoff, S. , Heung, K. , Byrd, R. , Hansen, R. , & Hertz‐Picciotto, I. (2008). The onset of autism: Patterns of symptom emergence in the first years of life. Autism Research, 1, 320–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozonoff, S. , & Iosif, A.‐M. (2019). Changing conceptualizations of regression: What prospective studies reveal about the onset of autism spectrum disorder. Neuroscience and Biobehavioral Reviews, 100, 296–304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozonoff, S. , Williams, B. , & Landa, R. (2005). Parental report of the early development of children with regressive autism: The delays‐plus‐regression phenotype. Autism, 9, 461–486. [DOI] [PubMed] [Google Scholar]
- Pickles, A. , Anderson, D.K. , & Lord, C. (2014). Heterogeneity and plasticity in the development of language: A 17‐year follow‐up of children referred early for possible autism. Journal of Child Psychology and Psychiatry, 55, 1354–1362. [DOI] [PubMed] [Google Scholar]
- Pickles, A. , Simonoff, E. , Conti‐Ramsden, G. , Falcaro, M. , Simkin, Z. , Charman, T. , … & Baird, G. (2009). Loss of language in early development of autism and specific language impairment. Journal of Child Psychology and Psychiatry, 50, 843–852. [DOI] [PubMed] [Google Scholar]
- Rogers, S. (2004). Developmental regression in autism spectrum disorders. Mental Retardation and Developmental Disabilities Research Reviews, 10, 139–143. [DOI] [PubMed] [Google Scholar]
- Rogers, S. , & DiLalla, D. (1990). Age of symptom onset in young children with pervasive developmental disorders. Journal of the American Academy of Child and Adolescent Psychiatry, 29, 863–872. [DOI] [PubMed] [Google Scholar]
- Roid, G. , & Sampers, J. (2004). Merrill‐Palmer Developmental Scales‐Revised manual. Wood Dale, IL: Stoelting. [Google Scholar]
- Sparrow, S.S. , Balla, R. , & Cicchetti, D. (2005). Vineland adaptive behavior scales (2nd edn). Circle Pines, MN. NCS Pearson, Inc. [Google Scholar]
- StataCorp. (2021). Stata statistical software: Release 17. College Station, TX: StataCorp LLC. [Google Scholar]
- Szatmari, P. , Bryson, S. , Streiner, D. , Wilson, F. , Archer, L. , & Ryerse, C. (2000). Two‐year outcome of preschool children with autism or Asperger’s syndrome. The American Journal of Psychiatry, 157, 1980–1987. [DOI] [PubMed] [Google Scholar]
- Tager‐Flusberg, H. (2016). Risk factors associated with language in autism spectrum disorder: Clues to underlying mechanisms. Journal of Speech, Language, and Hearing Research, 59, 143–154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tammimies, K. (2019). Genetic mechanisms of regression in autism spectrum disorder. Neuroscience and Biobehavioral Reviews, 102, 208–220. [DOI] [PubMed] [Google Scholar]
- Taylor, A. , Pickering, K. , Lord, C. , & Pickles, A. (1998). Mixed and multilevel models for longitudinal data: Growth curve analysis of language development. In Everitt B. & Dunn G. (Eds.), Recent advances in the statistical analysis of medical data (pp. 127–144). London: Arnold. [Google Scholar]
- Tuchman, R. (2017). What is the relationship between autism spectrum disorders and epilepsy ? Seminars in Pediatric Neurology, 24, 292–300. [DOI] [PubMed] [Google Scholar]
- Visser, J.C. , Rommelse, N.N.J. , Lappenschaar, M. , Servatius‐Oosterling, I.J. , Greven, C.U. , & Buitelaar, J.K. (2017). Variation in the early trajectories of autism symptoms is related to the development of language, cognition, and behavior problems. Journal of the American Academy of Child and Adolescent Psychiatry, 56, 659–568. [DOI] [PubMed] [Google Scholar]
- Werner, E. , & Dawson, G. (2005). Validation of the phenomenon of autistic regression using home videotapes. Archives of General Psychiatry, 62, 889–895. [DOI] [PubMed] [Google Scholar]
- Wilkinson, C. , Gabard‐Durnam, L. , Kapur, K. , Tager‐Flusberg, H. , Levin, A. , & Nelson, C.A. (2019). Use of longitudinal EEG measures in estimating language development in infants with and without familial risk for autism spectrum disorder. Neurobiology of Language, 1(1), 1–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams, K. , Brignell, A. , Prior, M. , Bartak, L. , & Roberts, J. (2015). Regression in autism spectrum disorders. Journal of Paediatrics and Child Health, 51, 61–64. [DOI] [PubMed] [Google Scholar]
- Wilson, S. , Djukic, A. , Shinnar, S. , Dharmani, C. , & Rapin, I. (2003). Clinical characteristics of language regression in children. Developmental Medicine and Child Neurology, 45, 508–514. [DOI] [PubMed] [Google Scholar]
- Wodka, E. , Mathy, P. , & Kalb, L. (2013). Predictors of phrase and fluent speech in children with autism and severe language delay. Pediatrics, 131, e1128–e1134. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Expressive language growth curve model: fixed effect coefficient estimates.
Table S2. Receptive language growth curve model: fixed effect coefficient estimates.
Appendix S1. Code for generating Kaplan–Meier plot.
Appendix S2. Code for generating Table S1 expressive language analysis.


