Abstract
Viruses that replicate in the human respiratory mucosa without infecting systemically, including influenza A, SARS-CoV-2, endemic coronaviruses, RSV, and many other “common cold” viruses, cause significant mortality and morbidity and are important public health concerns. Because these viruses generally do not elicit complete and durable protective immunity by themselves, they have not to date been effectively controlled by licensed or experimental vaccines. In this review, we examine challenges that have impeded development of effective mucosal respiratory vaccines, emphasizing that all of these viruses replicate extremely rapidly in the surface epithelium and are quickly transmitted to other hosts, within a narrow window of time before adaptive immune responses are fully marshaled. We discuss possible approaches to developing next-generation vaccines against these viruses, in consideration of several variables such as vaccine antigen configuration, dose and adjuventation, route and timing of vaccination, vaccine boosting, adjunctive therapies, and options for public health vaccination polices.
Despite the successful deployment of vaccines during the SARS-CoV2 pandemic, viruses replicating in the respiratory mucosal environment continue to present a particular challenge for developing effective vaccines with strong protection. Morens et al. discuss approaches that need to be prioritized for the development of next-generation vaccines against these viruses.
Introduction
Effective vaccines and vaccine prevention strategies against endemic and emerging respiratory viruses are of critical importance, as these pathogens kill as many as 5 million people worldwide every year. For example, over the past decade, influenza killed 12,000–52,000 people in the United States each year1 and ranks among the leading causes of years of productive life lost (YPLL). Endemic respiratory viruses such as respiratory syncytial virus (RSV) and the parainfluenzaviruses take many additional lives, and previously unrecognized respiratory viruses such as SARS-CoV-2, the cause of COVID-19, have emerged unexpectedly. SARS-CoV-2 thus far has killed more than one million people in the United States. The increasing frequency of emergences of such pandemic respiratory viruses may be a key feature of a new pandemic era,2 forcing us to consider anew the state of respiratory virus vaccinology (Figure 1).2 , 3
Figure 1.
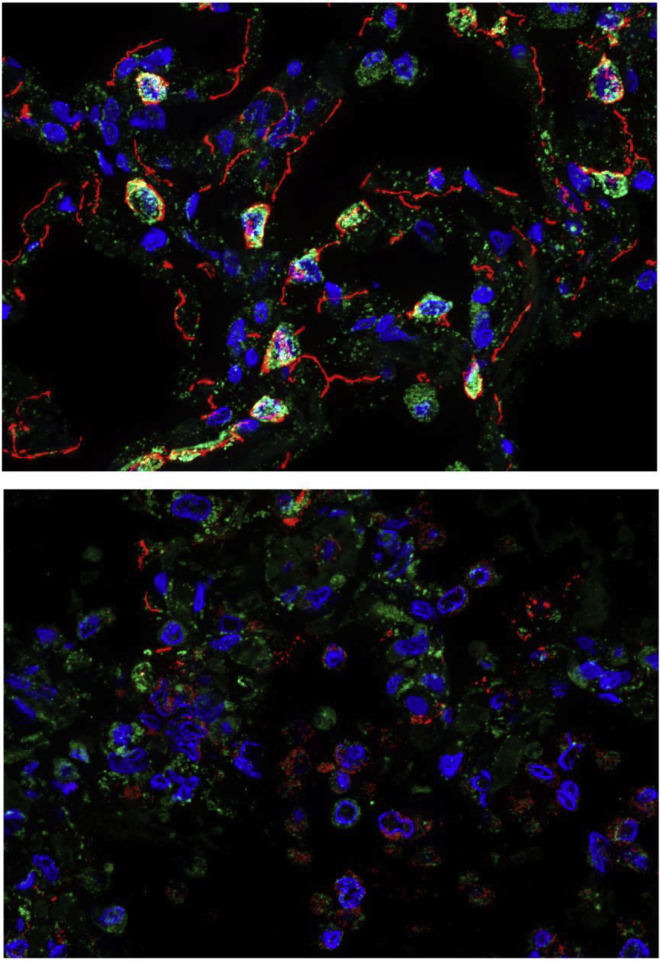
Alveolar damage in fatal COVID-19 lung autopsy samples
Multicolor immunofluorescence showing prosurfactant protein C (green) and E-cadherin (red) expression in normal lung tissue (top) and in a COVID-19 lung autopsy case (bottom). Nuclei are stained blue. Normal lung tissue alveolar septa show prominent prosurfactant protein C and E-cadherin expression in lung alveolar type 2 cells and epithelial junctions, respectively, compared to fatal COVID-19 lung tissue, which shows marked loss of alveolar septal prosurfactant protein C and E-cadherin staining and intra-alveolar accumulation of positive-stained epithelial debris.
The images demonstrate the extensive damage to the lung observed in a fatal COVID-19 case. New generations of vaccines against respiratory viruses like SARS-CoV-2 and influenza viruses are critically important for preventing pulmonary pathology, serious illness, and death.
Images of normal lung and a COVID-19 autopsy case are derived from D’Agnillo F, et al. Sci Transl Med. 2021;13(620):eabj7790. Images courtesy of Dr. Felice D’Agnillo of the US Food and Drug Administration.
Until the emergence of COVID-19, influenza had for many decades been the deadliest vaccine-preventable viral respiratory disease, one for which only less than suboptimal vaccines are available. Surprisingly, little has changed with influenza vaccines since 1957 when they were first administered in US national vaccination programs. Over the years, influenza vaccines have never been able to elicit durable protective immunity against seasonal influenza virus strains, even against non-drifted strains.4 , 5 , 6 , 7 Although current influenza vaccines reduce the risk of severe disease, hospitalization, and death to some degree, their effectiveness against clinically apparent infection is decidedly suboptimal, ranging from 14% to 60% over the past 15 influenza seasons.1 Furthermore, the duration of vaccine-elicited immunity is measured only in months. Current vaccines require annual re-vaccination with updated formulations that are frequently not precisely matched to circulating virus strains.8 Although annual influenza vaccinations are strongly recommended for most of the general public and especially for persons in high-risk groups, including the elderly, those with chronic diseases, and pregnant women, vaccine acceptance by the general public is not ideal.9
As of 2022, after more than 60 years of experience with influenza vaccines, very little improvement in vaccine prevention of infection has been noted. As pointed out decades ago, and still true today, the rates of effectiveness of our best approved influenza vaccines would be inadequate for licensure for most other vaccine-preventable diseases.7 Even decades-long efforts to develop better, so-called “universal” influenza vaccines—vaccines that would create more broadly protective immunity, preferably lasting over longer time periods8 , 10—have not yet resulted in next-generation, broadly protective vaccines, although a large number of experimental vaccines are in preclinical or early clinical development.11
During the COVID-19 pandemic, the rapid development and deployment of SARS-CoV-2 vaccines has saved innumerable lives and helped to achieve early partial pandemic control.12 However, as variant SARS-CoV-2 strains have emerged, deficiencies in these vaccines reminiscent of influenza vaccines have become apparent. The vaccines for these two very different viruses have common characteristics: they elicit incomplete and short-lived protection against evolving virus variants that escape population immunity.12 , 13 , 14 , 15 Considering that vaccine development and licensure is a long and complex process requiring years of preclinical and clinical safety and efficacy data, the limitations of influenza and SARS-CoV-2 vaccines remind us that candidate vaccines for most other respiratory viruses have to date been insufficiently protective for consideration of licensure, including candidate vaccines against RSV, a major killer of infants and the elderly,16 , 17 , 18 , 19 , 20 , 21 parainfluenzaviruses, endemic coronaviruses,22 and many other “common cold” viruses that cause significant morbidity and economic loss.
More than 50 years ago, the development of successful vaccines against some of the most important respiratory viruses, including measles, mumps, and rubella, led to the hope that vaccines could soon be developed for all other respiratory viruses. However, natural infections with these three vaccine-controlled respiratory viruses, as well as smallpox and varicella zoster virus (VZV), are not representative of infections caused by most respiratory viruses. They differ in at least three critically important ways that are related to their successful control with vaccines (Table 1 ):
(1) after first replicating mucosally, these systemic respiratory viruses all cause significant viremia that seeds an enormous number of infectious virions throughout the body, putting them in contact with multiple immune compartments and immune competent cell types,
(2) they have relatively long incubation periods that reflect initial mucosal replication and the subsequent systemic spread of infectious virions, which allows time for the induction of the full force of adaptive immunity, and
(3) they elicit long-term or lifetime protective immunity (Table 1).
Table 1.
Epidemiologic and immunologic parameters of selected human respiratory viruses and vaccines used to control them
| Virus | Incubation perioda | Marked viremia | Infection elicits long-term protective immunity | Re-infections are rare | Vaccines elicit long-term protective immunity | Vaccine type |
|---|---|---|---|---|---|---|
| Measles (to prodrome) | ≈10 days | yes | yes | yes | yes | replicating |
| Mumps | ≈16 days | yes | yes | yes | yes | replicating |
| Rubella | ≈16 days | yes | yes | yes | yes | replicating |
| Smallpoxb | ≈12 days | yes | yes | yes | yes | replicating |
| VZVc | ≈14 days | yes | yes | yes | yes | replicating |
| Endemic coronaviruses | ≈5 days | no | no | no | no | none |
| Influenza virus | ≈2 days | no | no | no | no | replicating, other |
| Parainfluenzaviruses | ≈4 days | no | no | no | no | none |
| RSV | ≈5 days | no | no | no | no | none |
| SARS-CoV-2 | ≈4 days | nod | no | no | no | non-replicating |
Viral incubation periods, especially shorter incubation periods, typically have very broad ranges; these estimates are taken from cross-sections of the literature.
Smallpox was eradicated from natural circulation in 1978.
Varicella-zoster virus (VZV) recrudescence (referred to as zoster, zona, or “shingles”) results from release of latent viruses from ganglia; second exogenous respiratory infections in normal persons are rare.
Although SARS-CoV-2 antigens have been detected in multiple tissues, the virus does not appear to be associated with significant “free” viremia, as evidenced by difficulty in culturing infectious virions from blood or tissues, and by weak elicitation of broad and durable protective systemic immunity.
In stark contrast, the non-systemic respiratory viruses such as influenza viruses, SARS-CoV-2, and RSV tend to have significantly shorter incubation periods (Table 1) and rapid courses of viral replication. They replicate predominantly in local mucosal tissue, without causing viremia, and do not significantly encounter the systemic immune system or the full force of adaptive immune responses, which take at least 5–7 days to mature, usually well after the peak of viral replication and onward transmission to others. SARS-CoV-2 “RNAemia” (circulation of viral RNA in the bloodstream, as is seen with most mucosal respiratory virus infections, as distinct from viremia, in which infectious viruses can be cultured from the blood), has been reported, and RT-PCR levels of viral RNA have been linked to severe disease,23 , 24 similar to studies of influenza RNAemia.25 , 26 As a result, the non-systemically replicating respiratory viruses, apparently including SARS-CoV-2,13 , 14 , 15 tend to repeatedly re-infect people over their lifetimes without ever eliciting complete and durable protection.27
Another important factor to consider is that although RNA viruses share a similar inherent RNA-dependent RNA polymerase error rate,28 different viruses (and different open reading frames within their genomes) differ in their tolerance for mutation. Mutational constraints can be related to frequent overlapping open reading frames28 or functional constraints on the acquisition of nonsynonymous mutations as is the case, for example, with measles virus.29 In contrast, the external influenza A virus hemagglutinin and neuraminidase proteins are comparatively plastic, and positively selected nonsynonymous mutations result in immunologically significant antigenic drift,30 , 31 by the acquisition of nonsynonymous mutations in antigenic epitopes, as well as by altering the N-linked glycosylation patterns.32 Rapid antigenic drift affects the control of annual influenza epidemics8 and complicates the effort to produce broadly protective, “universal” influenza vaccines. The SARS-CoV-2 spike protein has shown a similar plasticity, with the emergence of multiple variants with altered antigenicity33 that has complicated its control through current vaccination strategies.34
Although rapid evolution of antigenically variable mucosal viruses like influenza A viruses35 and SARS-CoV-2 complicate next-generation vaccine design, other mucosal-only respiratory viruses, such as RSV, have shown much less antigenic plasticity36 , 37 , 38; however, it still causes repeated infections over a lifetime without the development of long-term protective immunity.39 , 40 Thus, although genetic and antigenic variability of viruses like influenza and SARS-CoV-2 make vaccine design more challenging, these factors by themselves cannot fully explain the lack of elicitation of long-term protective immunity against other respiratory mucosal viruses like the more phenotypically stable RSV.
Taking all of these factors into account, it is not surprising that none of the predominantly mucosal respiratory viruses have ever been effectively controlled by vaccines. This observation raises a question of fundamental importance: if natural mucosal respiratory virus infections do not elicit complete and long-term protective immunity against reinfection, how can we expect vaccines, especially systemically administered non-replicating vaccines, to do so? This is a major challenge for future vaccine development, and overcoming it is critical as we work to develop “next-generation” vaccines.
Key challenges (summarized in Table 2 ) are discussed below, along with ways to confront them in the pursuit of new and improved vaccines for respiratory viruses.
Table 2.
Key challenges in developing next-generation vaccines against mucosal respiratory viruses including SARS-CoV-2, influenza A viruses, and emerging pandemic and other viruses of importance
| (1) Natural infections with mucosal respiratory viruses may not be fully controlled by human immune responses because the human immune system has evolved to tolerate them during very short intervals of mucosal viral replication |
| (2) Since mucosal and systemic immunity only partially protects against infection with mucosal respiratory viruses, we must take advantage of alternative host immune mechanisms |
| (3) Immune correlates of protection against mucosal respiratory viruses are incompletely understood, vary between viral strains and subtypes, with viral drift, and they exhibit inter-individual variation |
| (4) Vaccine-related questions of route of administration, antigen configuration, adjuventation, and association with adjunctive therapy are of great importance for current research |
| (5) Vaccinated hosts and host risk groups are many and heterogeneous |
| (6) Public health considerations relating to next-generation respiratory vaccines must contribute to shaping vaccine design, including vaccine schedule, role of boosting, frequency of vaccination and duration/completeness of protection, side effects, and public acceptance |
Natural infections with mucosal respiratory viruses may not be fully controlled by human immune responses because the human immune system has evolved to tolerate them during very short intervals of mucosal viral replication
The signs and symptoms of many different mucosal respiratory virus infections are remarkably similar: a short duration of illness and a typically uncomplicated course featuring rhinorrhea, sneezing, sore throat, variable cough, malaise, and in many cases low or absent fever.41 These commonalities strongly suggest similar pathogenic mechanisms for these viruses involving host inflammatory and innate immune responses.
The inability of mucosal respiratory viruses to elicit durable protective immunity reflects, among many other factors, evolutionary adaptation of virus to host, and host to virus. In this evolutionary relationship, viruses have two important advantages: (1) the human respiratory immune system has evolved to express complex tolerance regulation (see below) that may inhibit effective viral immune elimination during the first 5–7 post-inoculation days, when innate immune responses predominate; and (2) very short incubation periods (Table 1) allow substantial and unchallenged viral replication and onward viral spread before adaptive immune responses can be adequately mounted to eliminate them.42 , 43
Moreover, the several compartments of the respiratory immune system function in significantly different ways than do immune system compartments in other organ systems. In particular, the respiratory immune system resides in separate tissue-specific areas of the semi-organized, mucosal-associated lymphoid tissues (MALTs) of the tear/conjunctival associated lymphoid tissue (TALT), nasopharyngeal associated lymphoid tissue (NALT), bronchial associated lymphoid tissue (BALT),44 and in separate pulmonary compartments. Each of these compartments independently senses viral infection and antigen presentation, interacts with the other compartments and with the systemic immune system, initiates local effector responses, and maintains a variably tolerized immune state.
The terms “disease tolerance” and “immune tolerance” refer to the still-incompletely characterized but distinct category of mammalian immune defense mechanisms that allow hosts to “accept” infection and other antigenic stimuli to optimize survival (reviewed in Medzhitov et al.42 and Iwasaki et al.43). Because humans inhale and ingest enormous quantities of exogenous proteins with every breath and mouthful, the respiratory and gastrointestinal immune compartments have evolved to deal with continual and massive antigenic assaults from the outside world. (Immune responses to viral infection of the gastrointestinal mucosa have recently been reviewed,45 and are not discussed here.) Inhaled and ingested proteins must be identified and either tolerated or attacked and eliminated. This requires a highly evolved and complicated immune “decision-making” strategy that simultaneously accepts harmless foreign proteins while down-regulating immune responses to infectious agents and allergens that otherwise might cause bystander tissue damage.46 , 47 , 48 , 49 , 50
From the teleological point of view of the pathogen, mucosally replicating/non-systemic respiratory viruses have adapted over millennia to tolerized host immune environments to optimally infect, replicate, and spread quickly, before adaptive immune responses can be fully marshaled to control them. Among other tactics, they do so by inhibiting host interferon (IFN) responses (as do other systemically replicating RNA viruses), expressing decoy antigens, and inducing aberrant immune responses that promote viral survival without causing significant host tissue damage.16 The host respiratory microbiome contributes to this balance as well, on one hand by limiting infection (for example, by regulating IFN production) or on the other hand by promoting infection (for example, inducing antiviral states of immune tolerance).46 , 51 , 52 , 53 The gut microbiome has also been associated with individual variation in vaccine response, and this association also needs further in-depth investigation54 , 55
A better understanding of the afferent arm of these systems46 , 49 , 51 , 52 , 56 , 57 , 58 , 59 , 60 , 61 , 62 , 63 is needed, including the roles of upper airway epithelial cells (which play a key role in sensing foreign materials including viral pathogens). transepithelial antigen-sensing and -presenting cells such as M cells and intraepithelial dendritic cells, immune regulation, tolerance homeostasis, IgA control, and the expression and effects of class-switched IgA.64 The latter is locally stimulated by incoming mucosal antigens and, in addition to its viral neutralization and other effector functions, is more cross-protective than other immunoglobulins in its secretory form,65 , 66 initiates antibody-dependent cellular cytotxocity (ADCC), and is an immune regulator.67 , 68 , 69 , 70 The importance of mucosal secretory IgA (sIgA) in pathogen-specific responses against respiratory viral infections has long been appreciated for influenza viruses,65 , 71 RSV,72 , 73 and more recently SARS-CoV-2.74 , 75
The immunologic “Faustian bargain” between tolerance versus infection control, which permits transient, moderated infection by respiratory agents of low or intermediate pathogenicity to restrain the destructive forces of an immune elimination response,16 , 59 may be problematic for vaccine control of respiratory viruses, not only in the local and systemic sensing of vaccine antigens but also in eliciting optimal immune responses. Not surprisingly, immune tolerance is most pronounced in the upper respiratory tract, where respiratory viruses are inoculated, compared with the lower respiratory tract,49 , 57 , 76 where some respiratory viruses may spread if not contained (see below). This presents an additional challenge to developing vaccines that should ideally both dampen initiation of viral infection and control infection post inoculation.
The human tolerance/immune response paradigm is not unique. A more extreme version is found among some bat species: during long hibernations, bats conserve survival energy by tolerating enormous burdens of infection over long periods.77 This phenomenon is of more than passing interest because it may explain, in part, why bats are such important incubators of numerous viruses (coronaviruses, filoviruses, henipaviruses, rabies viruses, etc.) with cross-over potential to humans. Ironically, immune tolerance mechanisms in bats and other host species may be important determinants of the emergence of human epidemics and pandemics, just as they facilitate human-to-human spread of human-adapted viruses.
Since mucosal and systemic immunity only partially protects against infection with mucosal respiratory viruses, we must take advantage of alternative host immune mechanisms
For example, the recently appreciated phenomenon of “trained innate immunity”46 , 47 , 48 , 49 , 50 may offer promise that future vaccines might someday be able to boost innate immune responses either to specific pathogens or non-specifically. However, a key unanswered question remains how to control these responses to avoid harmful inflammatory consequences.78
Many studies in humans and experimental animals, some before sIgA had been recognized,22 , 58 , 79 , 80 , 81 indicate that secretory mucosal immunity is generally more effective than systemic immunity in controlling mucosal respiratory viruses18 , 79 , 82 and that tissue-resident memory T cells can be effective in rapidly responding to mucosal infection.83 The main humoral effectors at mucosal sites are sIgA expressed upon mucosal surfaces by local IgA-secreting plasma cells/plasmablasts and T cell memory and effector cells in the MALT.18 , 58 , 61 , 70 , 81 , 84 , 85 , 86 Nasal sIgA is the best correlate of protection in RSV challenge studies,18 even in the absence of systemic IgA-producing B cells. Similar results are seen with other viruses, including SARS-CoV-2.87 , 88 , 89 , 90 Although non-systemically replicating mucosal viruses elicit systemic effectors, including systemic IgA-producing plasma cells and in some cases high levels of serum IgA and IgG, neither circulating antibodies, plasmablasts, nor systemic B or T or T effector cells function optimally at mucosal sites. This is due in part to the dilution of transuded antibody and the fact that many such effector cells lack trafficking signals to these sites.85
It is therefore important to consider natural situations in which acquired antibody-mediated immunity does control non-systemic mucosal respiratory viruses. As long ago as 1918, it was shown that passively administered influenza immune plasmas could limit human influenza infection.91 In the 1940s, inhaled aerosolized influenza antibodies were also shown to have an effect against clinical influenza.92 Influenza infection generates durable systemic immune memory responses, as indicated by the detection of specific memory B cell clones 90 years after infection with the 1918 influenza virus93; however, low levels of circulating immunoglobulin and the time lag between infection and development of an anamnestic response may not offer neutralizing protection against a rapidly replicating influenza virus. Similar observations have been made with other mucosal respiratory viruses. For example, maternal IgG antibodies, especially antibodies against RSV F protein, protect against infant RSV16 , 17 , 18 , 19 and a parenterally administered humanized monoclonal antibody prevents RSV infection in at-risk infants.20
Nevertheless, the effectiveness of circulating Ig in these situations depends on transudation to mucosal surfaces of very high titers of antibody with specificity for key viral epitopes. It is not fully understood how such transudation is controlled, how antiviral IgA is regulated to function in both passive immunity and immune regulation,67 , 94 or how to elicit and sustain such high antibody levels with vaccination.
Apart from preventing initial infection, it is also important to consider the role of host immunity in limiting viral spread once infection has been established. Respiratory viruses usually first infect the mucosa of the upper respiratory tract, where IgA is most effective in antiviral control;95 however, some may spread contiguously to the lower respiratory tract, even to the pulmonary alveoli. This is of considerable significance for a subset of pneumogenic respiratory viruses such as influenza, RSV, and possibly SARS-CoV-2. These viruses are normally limited to the upper respiratory tract but can escape immune control and spread into the lungs to cause primary viral pneumonia with or without secondary bacterial pneumonia, often the principal causes of death from these infections.96 Moreover, evidence suggests that viral-bacterial coinfection may facilitate viral, and viral/bacterial airborne transmission97 and may even increase case-fatality, a phenomenon noted as long ago as 1917.98
The lung is functionally and immunologically different from the upper respiratory tract, containing both resident alveolar macrophages within the alveoli and resident immune and memory cells in peribronchiolar niches.85 The latter niches are composed of immune cells that are absent at birth but develop in response to post-natal antigenic exposures,86 which differ greatly among individuals and may expand in response to antigenic stimulation. Because the pulmonary immune system is semi-autonomous, it may be difficult for either systemically administered or upper respiratory-administered vaccines designed to prevent infection to additionally prevent pulmonary infection if upper respiratory infection spreads to the lungs. For instance, because IgA appears to be a better effector in the upper respiratory tract, whereas IgG is better in the lung,82 it may be difficult to develop single vaccines that protect against all respiratory tissues. The implications for vaccinology are clear: preventing viral upper respiratory infection and limiting post-infection viral spread to contiguous respiratory compartments are both critical but may not be easily achieved with single vaccines.
To understand how vaccines might protect against lower respiratory infections, we will need to learn how the “cross-talk” between upper respiratory, lower respiratory, and systemic immune systems is coordinated and controlled, at the level of cell receptors, antigen sensing, antigen presentation, and numerous effector functions.49 , 50 , 61 , 82 We also need to further investigate the development and maintenance of viral-specific lung resident memory B and T cells, how to increase their persistence in the lung, and how quickly they can be mobilized to infected mucosal sites.99 , 100
Immune correlates of protection against mucosal respiratory viruses are incompletely understood, vary between viral strains and subtypes, with viral drift, and they exhibit inter-individual variation
In developing next-generation vaccines, we will need to identify strong immunologic correlates of protection against each mucosal respiratory virus and agree about their relevance to public health vaccination goals.80 , 101 , 102 Additional immune correlate studies in humans are clearly needed and should be a research priority. Following influenza infection in humans, studies have long identified serum and mucosal immunoglobulin correlates103 , 104 , 105 and T cell immune correlates.104 , 106 , 107 In contrast, a human influenza challenge study after vaccination with inactivated vaccines or live-attenuated influenza vaccine (LAIV), followed by LAIV challenge, was unable to find any immunologic correlates of protection.108
The immune system is complex with many effectors. Serum antibody titers to various viral epitopes may only indirectly correlate with protection because of association with other more critical (but not usually measured) immune effectors, such as mucosal immunoglobulins65 or, for example, steric interactions of hemagglutinin stem antibodies resulting in neuraminidase inhibition.109 In recent human challenge studies, serum neuraminidase antibody titers were more strongly correlated with different measures of protection compared with hemagglutinin head or stem antibody titers.110 , 111 Neuraminidase remains and under-appreciated vaccine target for next-generation influenza vaccine design.112 In short, correlations between serum antibody titers and susceptibility to influenza infection may be statistically valid in large studies, but imperfect in the context of individual variation, rapid viral evolution, and waning titers.
For each mucosal virus, we must also arrive at consensus on desired levels of protection; for example, goals for protection might be:
-
•
preventing infection entirely, as vaccines for systemic respiratory viruses may do (Table 1);
-
•
limiting viral replication or preventing transmission as with influenza anti-neuraminidase immunity;
-
•
preventing disease; or
-
•
only preventing severe disease (e.g., requiring hospitalization), as appears to be the case with some influenza virus and SARS-CoV-2 vaccines.
Such consensus is needed if we are to develop the best vaccines and optimized vaccination strategies and policies for using them. With influenza, for example, vaccines historically have been designed to prevent upper respiratory infection, not secondary pulmonary infection associated with spread from the upper respiratory tract. This has proved problematic because current influenza vaccines are suboptimal at both preventing infection and eliciting pulmonary immunity.113 Although influenza and SARS-CoV-2 vaccines reduce disease severity when vaccines fail to prevent infection, significant numbers of fatalities still occur, resulting in tens of thousands of annual influenza deaths in the United States.114 , 115 , 116 With the imperfections of these vaccines, it seems a public health imperative to aggressively pursue better vaccines and vaccination strategies.
Vaccine-related questions of route of administration, antigen configuration, adjuventation, and association with adjunctive therapy are of great importance for current research
Human mucosal surfaces amount to 30–40 square feet,82 almost entirely underlaid with active lymphoid tissues. The predominant antibody at most of these sites is sIgA, representing an extraordinary 65%–70% of all human immunoglobulins.81 It is increasingly accepted that route of vaccine administration (e.g., intramuscular, intranasal, conjunctival, or aerosol routes) is a key determinant of mucosal respiratory response.
In general, and when feasible, mucosal immunization seems the optimal approach for respiratory viruses52 , 58 , 61 , 70 , 81 , 86 , 117; however, in contemplating next-generation vaccines we also may need optimized formulations, higher vaccine doses, greater frequency of vaccine administration, and overcoming immune tolerance challenges.
It is important for each virus to answer key questions such as:
-
(1)
Can non-replicating vaccines, which may be considerably less effective at eliciting IgA,82 be as efficacious as replicating vaccines, such as live-attenuated virus vaccines and live vaccine vectors expressing key viral proteins?
-
(2)
Can single- or pauci-antigen vaccines provide protection equivalent to more antigenically complex vaccines?
-
(3)
Can higher antigen doses or repeat vaccinations elicit better immunity?
-
(4)
What are the differential effects of soluble versus particulate antigens?82
-
(5)
What are ideal relationships between vaccine antigen load and systemic or mucosal adjuventation?118
-
(6)
What are the optimal strategies for routes and timing of vaccination: mucosal/systemic “prime-boost”? Newer strategies such as “prime-pull” and “prime-deploy” (vaccination strategies to elicit systemic T cell responses followed by recruitment of activated T cells via attractant or recruitment of resident memory T cells, respectively, to lung)119 , 120 , 121 , 122 and others?
It is noteworthy that all of the vaccines that are successful against the systemic respiratory viruses (Table 1)—those which generally elicit broader and longer lasting protective humoral and cell-mediated immunity—are systemically replicating live virus vaccines that fully encounter the host mucosal and systemic immune system. The imperfect protective properties of LAIV nasal spray represents an exception that might result from over-attenuation, pre-existing LAIV immunity, compositional balance, antigenic drift, or other yet unappreciated factors.123 , 124 In recent decades, live virus vaccines have been less commonly developed for reasons of time constraints and lack of effort due to expense, potential safety, and difficulties in quickly adapting to antigenic drift; however, we believe this potentially powerful approach must be energetically pursued.
With respect to vaccine antigen (immunogen) configuration, it is important to ask whether vaccines that elicit immunity to specific conserved epitopes can create complex protective immunity equivalent to that seen with more complex antigen presentations, such as multi-antigen cocktails, and whether vaccines that decouple antigen components, such as influenza HA/NA and HA/stem125 can reliably increase antigenicity. It is also of note that for some non-respiratory viruses, such as dermally inoculated (mosquito) systemically infecting flaviviruses—which include yellow fever virus, dengue virus, Zika virus, and Japanese encephalitis virus—complex cross-reactive immunity based on shared epitopes on related viruses and viral strains, when encountered sequentially, can elicit broad protection against somewhat distantly related viruses, associated with anamnestic antibody responses.126 Similarly, an experimental multi-subtype intranasal influenza vaccine elicits experimental protective immunity to influenza A virus subtypes absent in the vaccine itself.127 What are the viral and immunologic bases of these powerful, cross-protective phenomena, and can they be replicated with mucosal respiratory vaccines? How can we identify determinants of multi-antigen relatedness and complexity sufficient to make vaccines of greater breadth against different-but-related viral types?
A closely related question is whether vaccines that generate immune responses only against single critical epitopes conserved across virus strains and subtypes, or a limited number of such epitopes, can perform as well as vaccines that elicit broad humoral and cell-mediated responses against multiple epitopes. Although such conserved epitopes seem ideal candidates, vaccines based on this approach have not been particularly successful. The HIV conserved gp120 Muster-Katinger epitope, the influenza M2 protein, and conserved influenza virus stem epitopes, all have been considered promising candidates for vaccines but have not yet been shown to elicit strong protective immunity in clinical studies, although many candidates are at preclinical or only early clinical stages of development.128 , 129 , 130 , 131 , 132 , 133 These findings emphasize that host immune responses are complex and inter-related and that making broadly protective vaccines will be challenging.
Vaccine routes, doses, and re-vaccination schedules must be carefully considered, including routes of vaccination that maximize development of mucosal immune memory B and T cells as well as sIgA; the possible differential advantages of upper respiratory, lower respiratory tract, and systemic vaccination44 , 76 , 134 , 135 , 136 , 137 , 138 , 139 , 140 , 141; or optimized combinations of these. Attempting to control mucosal respiratory viruses with systemically administered non-replicating vaccines has thus far been largely unsuccessful, indicating that new approaches are needed. For example, during times of significant viral circulation within human populations, can better protection be achieved with repeat dosing, or by sequential mucosal and systemic vaccination? Is frequent re-boosting a viable vaccine option, e.g., via self-administered home nasal vaccines? Also to be considered are possible ancillary roles for prophylactic antivirals, including “programmable antivirals” targeting conserved structures,142 antibodies, including therapeutic IgA143 and innate immunity stimulators as adjuncts to imperfect vaccines. It remains to be seen if vaccination and prophylaxis can be effectively combined at the population level.
These provocative questions should all be considered as we work to optimize vaccination strategies. One intriguing approach to pursue, as noted above, would be making vaccines that stimulate innate immunity50 which might be ideally suited to “hit and run” infections by mucosal respiratory viruses, which characteristically infect, spread locally, and are transmitted to others before adaptive immune responses capable of controlling them can be mounted. Finally, there are currently limited options for mucosal vaccine adjuventation.82 , 144 If data support that mucosal vaccine adjuventation is needed and proves possible, we will need to assess potential pitfalls, including possible toxicity and side effects of repeated high-dose mucosal immunization/adjuventation, and how best to study it in humans.
Vaccinated hosts and host risk groups are many and heterogeneous
A key challenge for next-generation vaccines is determining if one-size-fits-all vaccines or vaccines targeted to key risk groups will be useful. It is inevitable that various human risk groups may require different vaccines or vaccine formulations. For example, we know that RSV infections are best prevented by humoral immunity in children but by cell-mediated immunity in the elderly.136 Moreover, children may require different vaccine doses than do young adults, who in turn may need different doses than do the elderly with narrowed B cell and especially T cell repertoires to viral antigens.136
Indeed, it seems likely that respiratory vaccines that fail to elicit robust cell-mediated immunity may be suboptimal for the elderly—but also that vaccines that elicit stronger cell-mediated responses could also increase the risk of immunopathogenic effects. If they do, can optimal trade-offs be found? The elderly are the most important litmus test risk group for vaccine prevention of influenza, SARS-CoV-2, RSV, and other respiratory viruses because they are most likely to suffer severe and fatal outcomes, and least likely to respond to vaccination, factors that make successful vaccination of older people a benchmark for other groups. It will also be important to learn more about genetic differences between individuals related to mucosal viral antigen sensing, predisposition to disease severity, and vaccine protection; for example, variations in IFITM3145 and other IFN pathway-associated genes, as much evidence suggests that severe viral respiratory disease often reflects host genetic susceptibility factors.16 , 51 , 146 , 147
Public health considerations relating to next-generation respiratory vaccines must contribute to shaping vaccine design, including vaccine schedule, role of boosting, frequency of vaccination and duration/completeness of protection, side effects, and public acceptance
Once improved vaccines are developed, vaccine recipes and schedules will need to be optimized to best elicit durable protective mucosal immunity, especially with multivalent or boosted vaccines, for which antigen immunodominance and balance between humoral and cell-mediated immune responses may be complex.
With regard to public health usefulness and acceptance, it will be important to consider roles for high dose or frequently boosted vaccine antigens, mixed-sequential vaccines (e.g., prime-boost with different vaccines), and whether these approaches will be accepted by providers, regulators, and the public. The observation that repeated infant exposures to RSV reduces severe disease upon subsequent infection,148 coupled with experimental data, suggest that respiratory vaccine timing and frequency can be important.19 Indeed, a recent controversial theory posits that the key determinant of immune/vaccine protection is not immune memory and recall but repeated antigenic exposures.149 This proposal seems to be contradicted by many observable phenomena but is at the same time consistent with the observation that maintenance of memory T cells in the lungs is associated with repeated antigenic exposures.82 Because of its implications for vaccinology, this question can and should be studied experimentally. It also prompts reconsideration of many accepted approaches, such as one-time annual influenza vaccination at the beginning of influenza seasons.
We also need to ask whether there are other vaccine approaches that should be considered, such as sequential seasonal vaccinations and supplemental mucosal vaccines to stimulate specific upper respiratory immunity, or non-specific innate immunity. Such approaches might include prime-boost approaches, for example, mixing elicitation of systemic and mucosal immunity, perhaps with prime systemic vaccination followed by a boost with intranasal vaccination or vice versa.35 , 82 , 150 Beyond intranasal vaccination, we will need to more fully explore responses to vaccination in other respiratory immune compartments, such as eye-drop conjunctival vaccination117 and particularly aerosol vaccination for certain respiratory viruses, as suggested by human and animal experimental studies for influenza and other respiratory viral diseases.44 , 135
Concluding remarks
Durably protective vaccines against non-systemic mucosal respiratory viruses with high mortality rates have thus far eluded vaccine development efforts.
Challenges to developing next-generation respiratory vaccines are many and complex (Table 2). We must better understand why multiple sequential mucosal infections with the same circulating respiratory viruses, spread out over decades of life, fail to elicit natural protective immunity, especially with viruses that lack significant antigenic drift (e.g., RSV and parainfluenzaviruses),17 , 18 , 19 , 22 if we are to rationally develop vaccines that prevent them. We must think outside the box to make next-generation vaccines that elicit immune protection against viruses that survive in human populations because of their ability to remain significantly outside of the full protective reach of human innate and adaptive immunity.
Past unsuccessful attempts to elicit solid protection against mucosal respiratory viruses and to control the deadly outbreaks and pandemics they cause have been a scientific and public health failure that must be urgently addressed. We are excited and invigorated that many investigators and collaborative groups are rethinking, from the ground up, all of our past assumptions and approaches to preventing important respiratory viral diseases and working to find bold new paths forward.
Acknowledgments
This work was supported by the Intramural Research Program of the NIH and NIAID.
Declaration of interests
The authors declare no competing interests.
References
- 1.CDC Past seasons’ vaccine effectiveness estimates. 2022. https://www.cdc.gov/flu/vaccines-work/past-seasons-estimates.html
- 2.Morens D.M., Fauci A.S. Emerging pandemic diseases: how we got to COVID-19. Cell. 2020;182:1077–1092. doi: 10.1016/j.cell.2020.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Graham B.S., Sullivan N.J. Emerging viral diseases from a vaccinology perspective: preparing for the next pandemic. Nat. Immunol. 2018;19:20–28. doi: 10.1038/s41590-017-0007-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Belongia E.A., Simpson M.D., King J.P., Sundaram M.E., Kelley N.S., Osterholm M.T., McLean H.Q. Variable influenza vaccine effectiveness by subtype: a systematic review and meta-analysis of test-negative design studies. Lancet Infect. Dis. 2016;16:942–951. doi: 10.1016/S1473-3099(16)00129-8. [DOI] [PubMed] [Google Scholar]
- 5.Memoli M.J., Han A., Walters K.A., Czajkowski L., Reed S., Athota R., Angela Rosas L., Cervantes-Medina A., Park J.K., Morens D.M., et al. Influenza A reinfection in sequential human challenge: implications for protective immunity and "universal" vaccine development. Clin. Infect. Dis. 2020;70:748–753. doi: 10.1093/cid/ciz281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tricco A.C., Chit A., Soobiah C., Hallett D., Meier G., Chen M.H., Tashkandi M., Bauch C.T., Loeb M. Comparing influenza vaccine efficacy against mismatched and matched strains: a systematic review and meta-analysis. BMC Med. 2013;11:153. doi: 10.1186/1741-7015-11-153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Langmuir A.D., Henderson D.A., Serfling R.E. The epidemiological basis for the control of influenza. Am. J. Public Health Nations Health. 1964;54:563–571. doi: 10.2105/ajph.54.4.563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Morens D.M., Taubenberger J.K. Making universal influenza vaccines: lessons from the 1918 pandemic. J. Infect. Dis. 2019;219:S5–S13. doi: 10.1093/infdis/jiy728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schmid P., Rauber D., Betsch C., Lidolt G., Denker M.L. Barriers of influenza vaccination intention and behavior - A systematic review of influenza vaccine hesitancy, 2005–2016. PLoS One. 2017;12:e0170550. doi: 10.1371/journal.pone.0170550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Erbelding E.J., Post D.J., Stemmy E.J., Roberts P.C., Augustine A.D., Ferguson S., Paules C.I., Graham B.S., Fauci A.S. A universal influenza vaccine: the strategic plan for the National Institute of Allergy and Infectious Diseases. J. Infect. Dis. 2018;218:347–354. doi: 10.1093/infdis/jiy103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.CIRDRAP The influenza vaccines roadmap: universal influenza vaccine technology landscape. 2022. https://ivr.cidrap.umn.edu/universal-influenza-vaccine-technology-landscape
- 12.Golob J.L., Lugogo N., Lauring A.S., Lok A.S. SARS-CoV-2 vaccines: a triumph of science and collaboration. JCI Insight. 2021;6:e149187. doi: 10.1172/jci.insight.149187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liu M., Li Y. Advances in COVID-19 vaccines and new coronavirus variants. Front. Med. 2022;9:888631. doi: 10.3389/fmed.2022.888631. Lausanne. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sette A., Crotty S. Immunological memory to SARS-CoV-2 infection and COVID-19 vaccines. Immunol. Rev. 2022;310:27–46. doi: 10.1111/imr.13089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shrestha L.B., Foster C., Rawlinson W., Tedla N., Bull R.A. Evolution of the SARS-CoV-2 omicron variants BA.1 to BA.5: implications for immune escape and transmission. Rev. Med. Virol. 2022;32:e2381. doi: 10.1002/rmv.2381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bergeron H.C., Tripp R.A. Immunopathology of RSV: an updated review. Viruses. 2021;13:2478. doi: 10.3390/v13122478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bueno S.M., González P.A., Pacheco R., Leiva E.D., Cautivo K.M., Tobar H.E., Mora J.E., Prado C.E., Zúñiga J.P., Jiménez J., et al. Host immunity during RSV pathogenesis. Int. Immunopharmacol. 2008;8:1320–1329. doi: 10.1016/j.intimp.2008.03.012. [DOI] [PubMed] [Google Scholar]
- 18.Habibi M.S., Jozwik A., Makris S., Dunning J., Paras A., DeVincenzo J.P., de Haan C.A., Wrammert J., Openshaw P.J., Chiu C., et al. Impaired antibody-mediated protection and defective IgA B-cell memory in experimental infection of adults with respiratory syncytial virus. Am. J. Respir. Crit. Care Med. 2015;191:1040–1049. doi: 10.1164/rccm.201412-2256OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lambert L., Sagfors A.M., Openshaw P.J., Culley F.J. Immunity to RSV in early-life. Front. Immunol. 2014;5:466. doi: 10.3389/fimmu.2014.00466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shadman K.A., Wald E.R. A review of palivizumab and emerging therapies for respiratory syncytial virus. Expert Opin. Biol. Ther. 2011;11:1455–1467. doi: 10.1517/14712598.2011.608062. [DOI] [PubMed] [Google Scholar]
- 21.Shi T., McAllister D.A., O'Brien K.L., Simoes E.A.F., Madhi S.A., Gessner B.D., Polack F.P., Balsells E., Acacio S., Aguayo C., et al. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in young children in 2015: a systematic review and modelling study. Lancet. 2017;390:946–958. doi: 10.1016/S0140-6736(17)30938-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Callow K.A., Parry H.F., Sergeant M., Tyrrell D.A. The time course of the immune response to experimental coronavirus infection of man. Epidemiol. Infect. 1990;105:435–446. doi: 10.1017/s0950268800048019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cardeñoso Domingo L., Roy Vallejo E., Zurita Cruz N.D., Chicot Llano M., Ávalos Pérez-Urria E., Barrios A., Hernando Santos J., Ortiz J., Rodríguez García S.C., Martín Ramírez A., et al. Relevant SARS-CoV-2 viremia is associated with COVID-19 severity: prospective cohort study and validation cohort. J. Med. Virol. 2022;94:5260–5270. doi: 10.1002/jmv.27989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jacobs J.L., Mellors J.W. Detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) RNA in blood of patients with coronavirus disease 2019 (COVID-19): what does it mean? Clin. Infect. Dis. 2021;73:e2898–e2900. doi: 10.1093/cid/ciaa1316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Campbell A.P., Jacob S.T., Kuypers J., Wald A., Englund J.A., Corey L., Boeckh M. Respiratory failure caused by 2009 novel influenza A/H1N1 in a hematopoietic stem-cell transplant recipient: detection of extrapulmonary H1N1 RNA and use of intravenous peramivir. Ann. Intern. Med. 2010;152:619–620. doi: 10.7326/0003-4819-152-9-201005040-00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lee N., Chan P.K., Wong C.K., Wong K.T., Choi K.W., Joynt G.M., Lam P., Chan M.C., Wong B.C., Lui G.C., et al. Viral clearance and inflammatory response patterns in adults hospitalized for pandemic 2009 influenza A(H1N1) virus pneumonia. Antivir. Ther. 2011;16:237–247. doi: 10.3851/IMP1722. [DOI] [PubMed] [Google Scholar]
- 27.Yewdell J.W. Individuals cannot rely on COVID-19 herd immunity: durable immunity to viral disease is limited to viruses with obligate viremic spread. PLoS Pathog. 2021;17:e1009509. doi: 10.1371/journal.ppat.1009509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Holmes E.C. Error thresholds and the constraints to RNA virus evolution. Trends Microbiol. 2003;11:543–546. doi: 10.1016/j.tim.2003.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fulton B.O., Sachs D., Beaty S.M., Won S.T., Lee B., Palese P., Heaton N.S. Mutational analysis of measles virus suggests constraints on antigenic variation of the glycoproteins. Cell Rep. 2015;11:1331–1338. doi: 10.1016/j.celrep.2015.04.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Russell C.A., Jones T.C., Barr I.G., Cox N.J., Garten R.J., Gregory V., Gust I.D., Hampson A.W., Hay A.J., Hurt A.C., et al. Influenza vaccine strain selection and recent studies on the global migration of seasonal influenza viruses. Vaccine. 2008;26:D31–D34. doi: 10.1016/j.vaccine.2008.07.078. [DOI] [PubMed] [Google Scholar]
- 31.Taubenberger J.K., Kash J.C. Influenza virus evolution, host adaptation, and pandemic formation. Cell Host Microbe. 2010;7:440–451. doi: 10.1016/j.chom.2010.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Altman M.O., Angel M., Košík I., Trovão N.S., Zost S.J., Gibbs J.S., Casalino L., Amaro R.E., Hensley S.E., Nelson M.I., Yewdell J.W. Human influenza A virus hemagglutinin glycan evolution follows a temporal pattern to a glycan limit. mBio. 2019;10:e. doi: 10.1128/mBio.00204-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.van der Straten K., Guerra D., van Gils M.J., Bontjer I., Caniels T.G., van Willigen H.D.G., Wynberg E., Poniman M., Burger J.A., Bouhuijs J.H., et al. Antigenic cartography using sera from sequence-confirmed SARS-CoV-2 variants of concern infections reveals antigenic divergence of Omicron. Immunity. 2022;55:1725–1731.e4. doi: 10.1016/j.immuni.2022.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Morens D.M., Taubenberger J.K., Fauci A.S. Universal coronavirus vaccines - an urgent need. N. Engl. J. Med. 2022;386:297–299. doi: 10.1056/NEJMp2118468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tang J., Zeng C., Cox T.M., Li C., Son Y.M., Cheon I.S., Wu Y., Behl S., Taylor J.J., Chakaraborty R., et al. Respiratory mucosal immunity against SARS-CoV-2 after mRNA vaccination. Sci. Immunol. 2022;7:eadd4853. doi: 10.1126/sciimmunol.add4853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Do L.A.H., Wilm A., van Doorn H.R., Lam H.M., Sim S., Sukumaran R., Tran A.T., Nguyen B.H., Tran T.T.L., Tran Q.H., et al. Direct whole-genome deep-sequencing of human respiratory syncytial virus A and B from Vietnamese children identifies distinct patterns of inter- and intra-host evolution. J. Gen. Virol. 2015;96:3470–3483. doi: 10.1099/jgv.0.000298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rios Guzman E., Hultquist J.F. Clinical and biological consequences of respiratory syncytial virus genetic diversity. Ther. Adv. Infect. Dis. 2022;9 doi: 10.1177/20499361221128091. 20499361221128091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tan L., Lemey P., Houspie L., Viveen M.C., Jansen N.J., van Loon A.M., Wiertz E., van Bleek G.M., Martin D.P., Coenjaerts F.E. Genetic variability among complete human respiratory syncytial virus subgroup A genomes: bridging molecular evolutionary dynamics and epidemiology. PLoS One. 2012;7:e51439. doi: 10.1371/journal.pone.0051439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bont L., Versteegh J., Swelsen W.T., Heijnen C.J., Kavelaars A., Brus F., Draaisma J.M., Pekelharing-Berghuis M., van Diemen-Steenvoorde R.A., Kimpen J.L. Natural reinfection with respiratory syncytial virus does not boost virus-specific T-cell immunity. Pediatr. Res. 2002;52:363–367. doi: 10.1203/00006450-200209000-00009. [DOI] [PubMed] [Google Scholar]
- 40.Yang K., Varga S.M. Mucosal vaccines against respiratory syncytial virus. Curr. Opin. Virol. 2014;6:78–84. doi: 10.1016/j.coviro.2014.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Newton A.H., Cardani A., Braciale T.J. The host immune response in respiratory virus infection: balancing virus clearance and immunopathology. Semin. Immunopathol. 2016;38:471–482. doi: 10.1007/s00281-016-0558-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Medzhitov R., Schneider D.S., Soares M.P. Disease tolerance as a defense strategy. Science. 2012;335:936–941. doi: 10.1126/science.1214935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Iwasaki A., Foxman E.F., Molony R.D. Early local immune defences in the respiratory tract. Nat. Rev. Immunol. 2017;17:7–20. doi: 10.1038/nri.2016.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Low N., Bavdekar A., Jeyaseelan L., Hirve S., Ramanathan K., Andrews N.J., Shaikh N., Jadi R.S., Rajagopal A., Brown K.E., et al. A randomized, controlled trial of an aerosolized vaccine against measles. N. Engl. J. Med. 2015;372:1519–1529. doi: 10.1056/NEJMoa1407417. [DOI] [PubMed] [Google Scholar]
- 45.James K.R., Elmentaite R., Teichmann S.A., Hold G.L. Redefining intestinal immunity with single-cell transcriptomics. Mucosal Immunol. 2022;15:531–541. doi: 10.1038/s41385-021-00470-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bekkering S., Domínguez-Andrés J., Joosten L.A.B., Riksen N.P., Netea M.G. Trained immunity: reprogramming innate immunity in health and disease. Annu. Rev. Immunol. 2021;39:667–693. doi: 10.1146/annurev-immunol-102119-073855. [DOI] [PubMed] [Google Scholar]
- 47.Netea M.G., Giamarellos-Bourboulis E.J., Domínguez-Andrés J., Curtis N., van Crevel R., van de Veerdonk F.L., Bonten M. Trained immunity: a tool for reducing susceptibility to and the severity of SARS-CoV-2 infection. Cell. 2020;181:969–977. doi: 10.1016/j.cell.2020.04.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Netea M.G., Quintin J., van der Meer J.W. Trained immunity: a memory for innate host defense. Cell Host Microbe. 2011;9:355–361. doi: 10.1016/j.chom.2011.04.006. [DOI] [PubMed] [Google Scholar]
- 49.Randall T.D., Mebius R.E. The development and function of mucosal lymphoid tissues: a balancing act with micro-organisms. Mucosal Immunol. 2014;7:455–466. doi: 10.1038/mi.2014.11. [DOI] [PubMed] [Google Scholar]
- 50.Xing Z., Afkhami S., Bavananthasivam J., Fritz D.K., D'Agostino M.R., Vaseghi-Shanjani M., Yao Y., Jeyanathan M. Innate immune memory of tissue-resident macrophages and trained innate immunity: re-vamping vaccine concept and strategies. J. Leukoc. Biol. 2020;108:825–834. doi: 10.1002/JLB.4MR0220-446R. [DOI] [PubMed] [Google Scholar]
- 51.Denney L., Ho L.P. The role of respiratory epithelium in host defence against influenza virus infection. Biomed J. 2018;41:218–233. doi: 10.1016/j.bj.2018.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zimmermann P., Curtis N. Factors that influence the immune response to vaccination. Clin. Microbiol. Rev. 2019;32 doi: 10.1128/CMR.00084-18. e00084–e00018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ichinohe T., Pang I.K., Kumamoto Y., Peaper D.R., Ho J.H., Murray T.S., Iwasaki A. Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc. Natl. Acad. Sci. USA. 2011;108:5354–5359. doi: 10.1073/pnas.1019378108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hagan T., Cortese M., Rouphael N., Boudreau C., Linde C., Maddur M.S., Das J., Wang H., Guthmiller J., Zheng N.Y., et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell. 2019;178:1313–1328.e13. doi: 10.1016/j.cell.2019.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Oh J.Z., Ravindran R., Chassaing B., Carvalho F.A., Maddur M.S., Bower M., Hakimpour P., Gill K.P., Nakaya H.I., Yarovinsky F., et al. TLR5-mediated sensing of gut microbiota is necessary for antibody responses to seasonal influenza vaccination. Immunity. 2014;41:478–492. doi: 10.1016/j.immuni.2014.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bradley K.C., Finsterbusch K., Schnepf D., Crotta S., Llorian M., Davidson S., Fuchs S.Y., Staeheli P., Wack A. Microbiota-driven tonic interferon signals in lung stromal cells protect from influenza virus infection. Cell Rep. 2019;28:245–256.e4. doi: 10.1016/j.celrep.2019.05.105. [DOI] [PubMed] [Google Scholar]
- 57.Crane M.J., Lee K.M., FitzGerald E.S., Jamieson A.M. Surviving deadly lung infections: innate host tolerance mechanisms in the pulmonary system. Front. Immunol. 2018;9:1421. doi: 10.3389/fimmu.2018.01421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gould V.M.W., Francis J.N., Anderson K.J., Georges B., Cope A.V., Tregoning J.S. Nasal IgA provides protection against human influenza challenge in volunteers with low serum influenza antibody titre. Front. Microbiol. 2017;8:900. doi: 10.3389/fmicb.2017.00900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hansen I.S., Baeten D.L.P., den Dunnen J. The inflammatory function of human IgA. Cell. Mol. Life Sci. 2019;76:1041–1055. doi: 10.1007/s00018-018-2976-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Holtzman M.J., Byers D.E., Alexander-Brett J., Wang X. The role of airway epithelial cells and innate immune cells in chronic respiratory disease. Nat. Rev. Immunol. 2014;14:686–698. doi: 10.1038/nri3739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lamichhane A., Azegamia T., Kiyonoa H. The mucosal immune system for vaccine development. Vaccine. 2014;32:6711–6723. doi: 10.1016/j.vaccine.2014.08.089. [DOI] [PubMed] [Google Scholar]
- 62.Whitsett J.A., Alenghat T. Respiratory epithelial cells orchestrate pulmonary innate immunity. Nat. Immunol. 2015;16:27–35. doi: 10.1038/ni.3045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zimmermann P., Curtis N. The influence of the intestinal microbiome on vaccine responses. Vaccine. 2018;36:4433–4439. doi: 10.1016/j.vaccine.2018.04.066. [DOI] [PubMed] [Google Scholar]
- 64.Sterlin D., Mathian A., Miyara M., Mohr A., Anna F., Claër L., Quentric P., Fadlallah J., Devilliers H., Ghillani P., et al. IgA dominates the early neutralizing antibody response to SARS-CoV-2. Sci. Transl. Med. 2021;13:eabd2223. doi: 10.1126/scitranslmed.abd2223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Taylor H.P., Dimmock N.J. Mechanism of neutralization of influenza virus by secretory IgA is different from that of monomeric IgA or IgG. J. Exp. Med. 1985;161:198–209. doi: 10.1084/jem.161.1.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Corthésy B. Multi-faceted functions of secretory IgA at mucosal surfaces. Front. Immunol. 2013;4:185. doi: 10.3389/fimmu.2013.00185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ben Mkaddem S., Rossato E., Heming N., Monteiro R.C. Anti-inflammatory role of the IgA Fc receptor (CD89): from autoimmunity to therapeutic perspectives. Autoimmun. Rev. 2013;12:666–669. doi: 10.1016/j.autrev.2012.10.011. [DOI] [PubMed] [Google Scholar]
- 68.Callow K.A. Effect of specific humoral immunity and some non-specific factors on resistance of volunteers to respiratory coronavirus infection. J. Hyg. 1985;95:173–189. doi: 10.1017/s0022172400062410. Lond. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Favre L., Spertini F., Corthésy B. Secretory IgA possesses intrinsic modulatory properties stimulating mucosal and systemic immune responses. J. Immunol. 2005;175:2793–2800. doi: 10.4049/jimmunol.175.5.2793. [DOI] [PubMed] [Google Scholar]
- 70.Rose M.A. Mucosal immunization in perspective. Hum. Vaccin. Immunother. 2014;10:2115–2117. doi: 10.4161/hv.29609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Suzuki T., Ainai A., Hasegawa H. Functional and structural characteristics of secretory IgA antibodies elicited by mucosal vaccines against influenza virus. Vaccine. 2017;35:5297–5302. doi: 10.1016/j.vaccine.2017.07.093. [DOI] [PubMed] [Google Scholar]
- 72.Wiseman D.J., Thwaites R.S., Drysdale S.B., Janet S., Donaldson G.C., Wedzicha J.A., Openshaw P.J., RESCEU Investigators Immunological and inflammatory biomarkers of susceptibility and severity in adult respiratory syncytial virus infections. J. Infect. Dis. 2020;222:S584–S591. doi: 10.1093/infdis/jiaa063. [DOI] [PubMed] [Google Scholar]
- 73.Russell C.D., Unger S.A., Walton M., Schwarze J. The human immune response to respiratory syncytial virus infection. Clin. Microbiol. Rev. 2017;30:481–502. doi: 10.1128/CMR.00090-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Takamatsu Y., Omata K., Shimizu Y., Kinoshita-Iwamoto N., Terada M., Suzuki T., Morioka S., Uemura Y., Ohmagari N., Maeda K., Mitsuya H. SARS-CoV-2-neutralizing humoral IgA response occurs earlier but is modest and diminishes faster than IgG response. Microbiol. Spectr. 2022:e0271622. doi: 10.1128/spectrum.02716-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ameratunga R., Leung E., Woon S.T., Lea E., Allan C., Chan L., Steele R., Lehnert K., Longhurst H. Selective IgA deficiency may be an under-recognized risk factor for severe COVID-19. J. Allergy Clin. Immunol. Pract. 2022 doi: 10.1016/j.jaip.2022.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Li M., Wang Y., Sun Y., Cui H., Zhu S.J., Qiu H.J. Mucosal vaccines: strategies and challenges. Immunol. Lett. 2020;217:116–125. doi: 10.1016/j.imlet.2019.10.013. [DOI] [PubMed] [Google Scholar]
- 77.Irving A.T., Ahn M., Goh G., Anderson D.E., Wang L.F. Lessons from the host defences of bats, a unique viral reservoir. Nature. 2021;589:363–370. doi: 10.1038/s41586-020-03128-0. [DOI] [PubMed] [Google Scholar]
- 78.Arneth B. Trained innate immunity. Immunol. Res. 2021;69:1–7. doi: 10.1007/s12026-021-09170-y. [DOI] [PubMed] [Google Scholar]
- 79.Smith C.B., Purcell R.H., Bellanti J.A., Chanock R.M. Protective effect of antibody to parainfluenza type 1 virus. N. Engl. J. Med. 1966;275:1145–1152. doi: 10.1056/NEJM196611242752101. [DOI] [PubMed] [Google Scholar]
- 80.Plotkin S.A. Updates on immunologic correlates of vaccine-induced protection. Vaccine. 2020;38:2250–2257. doi: 10.1016/j.vaccine.2019.10.046. [DOI] [PubMed] [Google Scholar]
- 81.Russell M.W., Moldoveanu Z., Ogra P.L., Mestecky J. Mucosal immunity in COVID-19: A neglected but critical aspect of SARS-CoV-2 infection. Front. Immunol. 2020;11:611337. doi: 10.3389/fimmu.2020.611337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lavelle E.C., Ward R.W. Mucosal vaccines - fortifying the frontiers. Nat. Rev. Immunol. 2022;22:236–250. doi: 10.1038/s41577-021-00583-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Schenkel J.M., Masopust D. Tissue-resident memory T cells. Immunity. 2014;41:886–897. doi: 10.1016/j.immuni.2014.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Allie S.R., Randall T.D. Pulmonary immunity to viruses. Clin. Sci. (Lond.) 2017;131:1737–1762. doi: 10.1042/CS20160259. [DOI] [PubMed] [Google Scholar]
- 85.Seong Y., Lazarus N.H., Sutherland L., Habtezion A., Abramson T., He X.S., Greenberg H.B., Butcher E.C. Trafficking receptor signatures define blood plasmablasts responding to tissue-specific immune challenge. JCI Insight. 2017;2:e90233. doi: 10.1172/jci.insight.90233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Turner D.L., Bickham K.L., Thome J.J., Kim C.Y., D'Ovidio F., Wherry E.J., Farber D.L. Lung niches for the generation and maintenance of tissue-resident memory T cells. Mucosal Immunol. 2014;7:501–510. doi: 10.1038/mi.2013.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Sheikh-Mohamed S., Sanders E.C., Gommerman J.L., Tal M.C. Guardians of the oral and nasopharyngeal galaxy: IgA and protection against SARS-CoV-2 infection. Immunol. Rev. 2022;309:75–85. doi: 10.1111/imr.13118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Primorac D., Vrdoljak K., Brlek P., Pavelić E., Molnar V., Matišić V., Erceg Ivkošić I., Parčina M. Adaptive immune responses and immunity to SARS-CoV-2. Front. Immunol. 2022;13:848582. doi: 10.3389/fimmu.2022.848582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Mouro V., Fischer A. Dealing with a mucosal viral pandemic: lessons from COVID-19 vaccines. Mucosal Immunol. 2022;15:584–594. doi: 10.1038/s41385-022-00517-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hennings V., Thörn K., Albinsson S., Lingblom C., Andersson K., Andersson C., Järbur K., Pullerits R., Idorn M., Paludan S.R., et al. The presence of serum anti-SARS-CoV-2 IgA appears to protect primary health care workers from COVID-19. Eur. J. Immunol. 2022;52:800–809. doi: 10.1002/eji.202149655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Luke T.C., Kilbane E.M., Jackson J.L., Hoffman S.L. Meta-analysis: convalescent blood products for Spanish influenza pneumonia: a future H5N1 treatment? Ann. Intern. Med. 2006;145:599–609. doi: 10.7326/0003-4819-145-8-200610170-00139. [DOI] [PubMed] [Google Scholar]
- 92.Smorodintzew A.A., Gulamow A.G., Tschalkina O.M. Über die spezifische Prophylaxe der epidemischen Grippe durch Inhalation antigrippösen Serums. Z. Klin. Med. 1940;138:687–806. [Google Scholar]
- 93.Yu X., Tsibane T., McGraw P.A., House F.S., Keefer C.J., Hicar M.D., Tumpey T.M., Pappas C., Perrone L.A., Martinez O., et al. Neutralizing antibodies derived from the B cells of 1918 influenza pandemic survivors. Nature. 2008;455:532–536. doi: 10.1038/nature07231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Englund J.A. Passive protection against respiratory syncytial virus disease in infants: the role of maternal antibody. Pediatr. Infect. Dis. J. 1994;13:449–453. doi: 10.1097/00006454-199405000-00037. [DOI] [PubMed] [Google Scholar]
- 95.Renegar K.B., Small P.A., Jr., Boykins L.G., Wright P.F. Role of IgA versus IgG in the control of influenza viral infection in the murine respiratory tract. J. Immunol. 2004;173:1978–1986. doi: 10.4049/jimmunol.173.3.1978. [DOI] [PubMed] [Google Scholar]
- 96.Morens D.M., Taubenberger J.K., Fauci A.S. Predominant role of bacterial pneumonia as a cause of death in pandemic influenza: implications for pandemic influenza preparedness. J. Infect. Dis. 2008;198:962–970. doi: 10.1086/591708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Rowe H.M., Livingston B., Margolis E., Davis A., Meliopoulos V.A., Echlin H., Schultz-Cherry S., Rosch J.W. Respiratory bacteria stabilize and promote airborne transmission of influenza A virus. mSystems. 2020;5 doi: 10.1128/mSystems.00762-20. e00762–e00720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Morens D.M., Taubenberger J.K. A forgotten epidemic that changed medicine: measles in the US Army, 1917–18. Lancet Infect. Dis. 2015;15:852–861. doi: 10.1016/S1473-3099(15)00109-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Szabo P.A., Miron M., Farber D.L. Location, location, location: tissue resident memory T cells in mice and humans. Sci. Immunol. 2019;4:eaas9673. doi: 10.1126/sciimmunol.aas9673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Gray J.I., Farber D.L. Tissue-resident immune cells in humans. Annu. Rev. Immunol. 2022;40:195–220. doi: 10.1146/annurev-immunol-093019-112809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Krammer F. Correlates of protection from SARS-CoV-2 infection. Lancet. 2021;397:1421–1423. doi: 10.1016/S0140-6736(21)00782-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hall V., Foulkes S., Insalata F., Saei A., Kirwan P., Atti A., Wellington E., Khawam J., Munro K., Cole M., et al. Effectiveness and durability of protection against future SARS-CoV-2 infection conferred by COVID-19 vaccination and previous infection; findings from the UK SIREN prospective cohort study of healthcare workers March 2020 to September 2021. Preprint at medRxiv. 2021 doi: 10.1101/2021.11.29.21267006. [DOI] [Google Scholar]
- 103.Brown T.A., Murphy B.R., Radl J., Haaijman J.J., Mestecky J. Subclass distribution and molecular form of immunoglobulin A hemagglutinin antibodies in sera and nasal secretions after experimental secondary infection with influenza A virus in humans. J. Clin. Microbiol. 1985;22:259–264. doi: 10.1128/jcm.22.2.259-264.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Dolin R., Richman D.D., Murphy B.R., Fauci A.S. Cell-mediated immune responses in humans after induced infection with influenza A virus. J. Infect. Dis. 1977;135:714–719. doi: 10.1093/infdis/135.5.714. [DOI] [PubMed] [Google Scholar]
- 105.Ng S., Nachbagauer R., Balmaseda A., Stadlbauer D., Ojeda S., Patel M., Rajabhathor A., Lopez R., Guglia A.F., Sanchez N., et al. Novel correlates of protection against pandemic H1N1 influenza A virus infection. Nat. Med. 2019;25:962–967. doi: 10.1038/s41591-019-0463-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hayward A.C., Wang L., Goonetilleke N., Fragaszy E.B., Bermingham A., Copas A., Dukes O., Millett E.R., Nazareth I., Nguyen-Van-Tam J.S., et al. Natural T cell-mediated protection against seasonal and pandemic influenza. Results of the flu watch cohort study. Am. J. Respir. Crit. Care Med. 2015;191:1422–1431. doi: 10.1164/rccm.201411-1988OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Sridhar S., Begom S., Bermingham A., Hoschler K., Adamson W., Carman W., Bean T., Barclay W., Deeks J.J., Lalvani A. Cellular immune correlates of protection against symptomatic pandemic influenza. Nat. Med. 2013;19:1305–1312. doi: 10.1038/nm.3350. [DOI] [PubMed] [Google Scholar]
- 108.Wright P.F., Hoen A.G., Ilyushina N.A., Brown E.P., Ackerman M.E., Wieland-Alter W., Connor R.I., Jegaskanda S., Rosenberg-Hasson Y., Haynes B.C., et al. Correlates of immunity to influenza as determined by challenge of children with live, attenuated influenza vaccine. Open Forum Infect. Dis. 2016;3:ofw108. doi: 10.1093/ofid/ofw108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Kosik I., Angeletti D., Gibbs J.S., Angel M., Takeda K., Kosikova M., Nair V., Hickman H.D., Xie H., Brooke C.B., Yewdell J.W. Neuraminidase inhibition contributes to influenza A virus neutralization by anti-hemagglutinin stem antibodies. J. Exp. Med. 2019;216:304–316. doi: 10.1084/jem.20181624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Memoli M.J., Shaw P.A., Han A., Czajkowski L., Reed S., Athota R., Bristol T., Fargis S., Risos K., Powers J.H., et al. Evaluation of antihemagglutinin and antineuraminidase antibodies as correlates of protection in an influenza A/H1N1 virus healthy human challenge model. mBio. 2016;7 doi: 10.1128/mBio.00417-16. e00417–e00416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Park J.K., Han A., Czajkowski L., Reed S., Athota R., Bristol T., Rosas L.A., Cervantes-Medina A., Taubenberger J.K., Memoli M.J. Evaluation of preexisting anti-hemagglutinin stalk antibody as a correlate of protection in a healthy volunteer challenge with influenza A/H1N1pdm virus. mBio. 2018;9 doi: 10.1128/mBio.02284-17. e02284–e02217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Eichelberger M.C., Morens D.M., Taubenberger J.K. Neuraminidase as an influenza vaccine antigen: a low hanging fruit, ready for picking to improve vaccine effectiveness. Curr. Opin. Immunol. 2018;53:38–44. doi: 10.1016/j.coi.2018.03.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Cox R.J., Brokstad K.A., Ogra P. Influenza virus: immunity and vaccination strategies. Comparison of the immune response to inactivated and live, attenuated influenza vaccines. Scand. J. Immunol. 2004;59:1–15. doi: 10.1111/j.0300-9475.2004.01382.x. [DOI] [PubMed] [Google Scholar]
- 114.Alcendor D.J., Matthews-Juarez P., Smoot D., Hildreth J.E.K., Lamar K., Tabatabai M., Wilus D., Juarez P.D. Breakthrough COVID-19 infections in the US: implications for prolonging the pandemic. Vaccines. 2022;10:755. doi: 10.3390/vaccines10050755. Basel. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Demicheli V., Jefferson T., Di Pietrantonj C., Ferroni E., Thorning S., Thomas R.E., Rivetti A. Vaccines for preventing influenza in the elderly. Cochrane Database Syst. Rev. 2018;2:CD004876. doi: 10.1002/14651858.CD004876.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Vardeny O., Kim K., Udell J.A., Joseph J., Desai A.S., Farkouh M.E., Hegde S.M., Hernandez A.F., McGeer A., Talbot H.K., et al. Effect of high-dose trivalent vs standard-dose quadrivalent influenza vaccine on mortality or cardiopulmonary hospitalization in patients with high-risk cardiovascular disease: A randomized clinical trial. JAMA. 2021;325:39–49. doi: 10.1001/jama.2020.23649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Lee J.S., Yoon S., Han S.J., Kim E.D., Kim J., Shin H.S., Seo K.Y. Eyedrop vaccination: an immunization route with promises for effective responses to pandemics. Expert Rev. Vaccines. 2022;21:91–101. doi: 10.1080/14760584.2022.2008246. [DOI] [PubMed] [Google Scholar]
- 118.Calzas C., Chevalier C. Innovative mucosal vaccine formulations against influenza A virus infections. Front. Immunol. 2019;10:1605. doi: 10.3389/fimmu.2019.01605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Hodgins B., Pillet S., Landry N., Ward B.J. Prime-pull vaccination with a plant-derived virus-like particle influenza vaccine elicits a broad immune response and protects aged mice from death and frailty after challenge. Immun. Ageing. 2019;16:27. doi: 10.1186/s12979-019-0167-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Chung H., Kim E.A., Chang J. A “prime and deploy” strategy for universal influenza vaccine targeting nucleoprotein induces lung-resident memory CD8 T cells. Immune Netw. 2021;21:e28. doi: 10.4110/in.2021.21.e28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Gary E.N., Tursi N.J., Warner B., Parzych E.M., Ali A.R., FRASE D., Moffat E., Embury-Hyatt C., Smith T.R.F., Broderick K.E., et al. Mucosal chemokine adjuvant enhances synDNA vaccine-mediated responses to SARS-CoV-2 and provides heterologous protection in vivo. Cell. Reprod. Med. 2022;3:100693. doi: 10.1016/j.xcrm.2022.100693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Bernstein D.I., Cardin R.D., Bravo F.J., Awasthi S., Lu P., Pullum D.A., Dixon D.A., Iwasaki A., Friedman H.M. Successful application of prime and pull strategy for a therapeutic HSV vaccine. npj Vaccines. 2019;4:33. doi: 10.1038/s41541-019-0129-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Poehling K.A., Caspard H., Peters T.R., Belongia E.A., Congeni B., Gaglani M., Griffin M.R., Irving S.A., Kavathekar P.K., McLean H.Q., et al. 2015–2016 vaccine effectiveness of live attenuated and inactivated influenza vaccines in children in the United States. Clin. Infect. Dis. 2018;66:665–672. doi: 10.1093/cid/cix869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Isakova-Sivak I., Matyushenko V., Kotomina T., Kiseleva I., Krutikova E., Donina S., Rekstin A., Larionova N., Mezhenskaya D., Sivak K., et al. Sequential immunization with universal live attenuated influenza vaccine candidates protects ferrets against a high-dose heterologous virus challenge. Vaccines. 2019;7:61. doi: 10.3390/vaccines7030061. Basel. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Angeletti D., Kosik I., Santos J.J.S., Yewdell W.T., Boudreau C.M., Mallajosyula V.V.A., Mankowski M.C., Chambers M., Prabhakaran M., Hickman H.D., et al. Outflanking immunodominance to target subdominant broadly neutralizing epitopes. Proc. Natl. Acad. Sci. USA. 2019;116:13474–13479. doi: 10.1073/pnas.1816300116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Price W.H., Thind I.S. The mechanism of cross-protection afforded by dengue virus against West Nile virus in hamsters. J. Hyg. 1972;70:611–617. doi: 10.1017/s0022172400022476. Lond. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Park J., Fong Legaspi S.L., Schwartzman L.M., Gygli S.M., Sheng Z.M., Freeman A.D., Matthews L.M., Xiao Y., Ramuta M.D., Batchenkova N.A., et al. An inactivated multivalent influenza A virus vaccine is broadly protective in mice and ferrets. Sci. Transl. Med. 2022;14:eabo2167. doi: 10.1126/scitranslmed.abo2167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Folschweiller N., Vanden Abeele C., Chu L., Van Damme P., García-Sastre A., Krammer F., Nachbagauer R., Palese P., Solórzano A., Bi D., et al. Reactogenicity, safety, and immunogenicity of chimeric haemagglutinin influenza split-virion vaccines, adjuvanted with AS01 or AS03 or non-adjuvanted: a phase 1–2 randomised controlled trial. Lancet Infect. Dis. 2022;22:1062–1075. doi: 10.1016/S1473-3099(22)00024-X. [DOI] [PubMed] [Google Scholar]
- 129.Darricarrère N., Qiu Y., Kanekiyo M., Creanga A., Gillespie R.A., Moin S.M., Saleh J., Sancho J., Chou T.H., Zhou Y., et al. Broad neutralization of H1 and H3 viruses by adjuvanted influenza HA stem vaccines in nonhuman primates. Sci. Transl. Med. 2021;13:eabe5449. doi: 10.1126/scitranslmed.abe5449. [DOI] [PubMed] [Google Scholar]
- 130.Trkola A., Purtscher M., Muster T., Ballaun C., Buchacher A., Sullivan N., Srinivasan K., Sodroski J., Moore J.P., Katinger H. Human monoclonal antibody 2G12 defines a distinctive neutralization epitope on the gp120 glycoprotein of human immunodeficiency virus type 1. J. Virol. 1996;70:1100–1108. doi: 10.1128/JVI.70.2.1100-1108.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Saelens X. The role of matrix protein 2 ectodomain in the development of universal influenza vaccines. J. Infect. Dis. 2019;219:S68–S74. doi: 10.1093/infdis/jiz003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Han A., Czajkowski L., Rosas L.A., Cervantes-Medina A., Xiao Y., Gouzoulis M., Lumbard K., Hunsberger S., Reed S., Athota R., et al. Safety and efficacy of CR6261 in an influenza A H1N1 healthy human challenge model. Clin. Infect. Dis. 2021;73:e4260–e4268. doi: 10.1093/cid/ciaa1725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Park J.K., Xiao Y., Ramuta M.D., Rosas L.A., Fong S., Matthews A.M., Freeman A.D., Gouzoulis M.A., Batchenkova N.A., Yang X., et al. Pre-existing immunity to influenza virus hemagglutinin stalk might drive selection for antibody-escape mutant viruses in a human challenge model. Nat. Med. 2020;26:1240–1246. doi: 10.1038/s41591-020-0937-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Tungatt K., Dolton G., Morgan S.B., Attaf M., Fuller A., Whalley T., Hemmink J.D., Porter E., Szomolay B., Montoya M., et al. Induction of influenza-specific local CD8 T-cells in the respiratory tract after aerosol delivery of vaccine antigen or virus in the Babraham inbred pig. PLoS Pathog. 2018;14:e1007017. doi: 10.1371/journal.ppat.1007017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Morgan S.B., Hemmink J.D., Porter E., Harley R., Shelton H., Aramouni M., Everett H.E., Brookes S.M., Bailey M., Townsend A.M., et al. Aerosol delivery of a candidate universal influenza vaccine reduces viral load in pigs challenged with pandemic H1N1 virus. J. Immunol. 2016;196:5014–5023. doi: 10.4049/jimmunol.1502632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Henry C., Zheng N.Y., Huang M., Cabanov A., Rojas K.T., Kaur K., Andrews S.F., Palm A.E., Chen Y.Q., Li Y., et al. Influenza virus vaccination elicits poorly adapted B cell responses in elderly individuals. Cell Host Microbe. 2019;25:357–366.e6. doi: 10.1016/j.chom.2019.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.King R.G., Silva-Sanchez A., Peel J.N., Botta D., Meza-Perez S., Allie R., Schultz M.D., Liu M., Bradley J.E., Qiu S., et al. 2020. Single-dose intranasal administration of AdCOVID elicits systemic and mucosal immunity against SARS-CoV-2 in mice. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Krishnaswamy J.K., Gowthaman U., Zhang B., Mattsson J., Szeponik L., Liu D., Wu R., White T., Calabro S., Xu L., et al. Migratory CD11b(+) conventional dendritic cells induce T follicular helper cell-dependent antibody responses. Sci. Immunol. 2017;2:eaam9169. doi: 10.1126/sciimmunol.aam9169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Lund F.E., Randall T.D. Scent of a vaccine. Science. 2021;373:397–399. doi: 10.1126/science.abg9857. [DOI] [PubMed] [Google Scholar]
- 140.van Doremalen N., Purushotham J.N., Schulz J.E., Holbrook M.G., Bushmaker T., Carmody A., Port J.R., Yinda C.K., Okumura A., Saturday G., et al. Intranasal ChAdOx1 nCoV-19/AZD1222 vaccination reduces viral shedding after SARS-CoV-2 D614G challenge in preclinical models. Sci. Transl. Med. 2021;13:eabh0755. doi: 10.1126/scitranslmed.abh0755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Ye Z.W., Ong C.P., Tang K., Fan Y., Luo C., Zhou R., Luo P., Cheng Y., Gray V.S., Wang P., et al. Intranasal administration of a single dose of a candidate live attenuated vaccine derived from an NSP16-deficient SARS-CoV-2 strain confers sterilizing immunity in animals. Cell. Mol. Immunol. 2022;19:588–601. doi: 10.1038/s41423-022-00855-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Hagey R.J., Elazar M., Pham E.A., Tian S., Ben-Avi L., Bernardin-Souibgui C., Yee M.F., Moreira F.R., Rabinovitch M.V., Meganck R.M., et al. Programmable antivirals targeting critical conserved viral RNA secondary structures from influenza A virus and SARS-CoV-2. Nat. Med. 2022;28:1944–1955. doi: 10.1038/s41591-022-01908-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Sterlin D., Gorochov G. When therapeutic IgA antibodies might come of age. Pharmacology. 2021;106:9–19. doi: 10.1159/000510251. [DOI] [PubMed] [Google Scholar]
- 144.van Splunter M., van Hoffen E., Floris-Vollenbroek E.G., Timmerman H., de Bos E.L., Meijer B., Ulfman L.H., Witteman B., Wells J.M., Brugman S., et al. Oral cholera vaccination promotes homing of IgA(+) memory B cells to the large intestine and the respiratory tract. Mucosal Immunol. 2018;11:1254–1264. doi: 10.1038/s41385-018-0006-7. [DOI] [PubMed] [Google Scholar]
- 145.Everitt A.R., Clare S., Pertel T., John S.P., Wash R.S., Smith S.E., Chin C.R., Feeley E.M., Sims J.S., Adams D.J., et al. IFITM3 restricts the morbidity and mortality associated with influenza. Nature. 2012;484:519–523. doi: 10.1038/nature10921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.D'Agnillo F., Walters K.A., Xiao Y., Sheng Z.M., Scherler K., Park J., Gygli S., Rosas L.A., Sadtler K., Kalish H., et al. Lung epithelial and endothelial damage, loss of tissue repair, inhibition of fibrinolysis, and cellular senescence in fatal COVID-19. Sci. Transl. Med. 2021;13:eabj7790. doi: 10.1126/scitranslmed.abj7790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Ziegler C.G.K., Miao V.N., Owings A.H., Navia A.W., Tang Y., Bromley J.D., Lotfy P., Sloan M., Laird H., Williams H.B., et al. Impaired local intrinsic immunity to SARS-CoV-2 infection in severe COVID-19. Cell. 2021;184:4713–4733.e22. doi: 10.1016/j.cell.2021.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Green C.A., Sande C.J., de Lara C., Thompson A.J., Silva-Reyes L., Napolitano F., Pierantoni A., Capone S., Vitelli A., Klenerman P., Pollard A.J. Humoral and cellular immunity to RSV in infants, children and adults. Vaccine. 2018;36:6183–6190. doi: 10.1016/j.vaccine.2018.08.056. [DOI] [PubMed] [Google Scholar]
- 149.Zinkernagel R.M. What if protective immunity is antigen-driven and not due to so-called "memory" B and T cells? Immunol. Rev. 2018;283:238–246. doi: 10.1111/imr.12648. [DOI] [PubMed] [Google Scholar]
- 150.Mao T., Israelow B., Suberi A., Zhou L., Reschke M., Peña-Hernández M.A., Dong H., Homer R.J., Saltzman W.M., Iwasaki A. 2022. Unadjuvanted intranasal spike vaccine booster elicits robust protective mucosal immunity against sarbecoviruses. [DOI] [PMC free article] [PubMed] [Google Scholar]