Abstract
Clinically significant sleep problems affect up to 86% of the autistic population in school-age. Sleep problems can have negative impacts on child cognition, behavior, and health. However, sex differences in the prevalence and types of sleep problems are not well understood in autism. To evaluate sex differences in sleep problems in the school-age autistic population, we obtained parent-report of sleep problems on the Children’s Sleep Habits Questionnaire and conducted direct assessments to establish diagnosis and intellectual ability in 6–12-year-old children (autism n=250; typical development [TD] n=114). Almost 85% of autistic females demonstrated sleep problems compared to 65.8% of autistic males, 44.8% of TD females, and 42.4% of TD males; a statistically significant increase for autistic females. Autistic females demonstrated increased bedtime resistance, sleep anxiety, and sleepiness, and decreased sleep duration, but did not differ in sleep onset delay, night wakings, parasomnias, or disordered breathing compared with autistic males. Intellectual ability was not related to increased sleep problems. Higher anxiety scores were associated with more sleep problems for males but not females. In one of the first studies to evaluate sex differences in sleep in the school-age, autistic population, autistic females demonstrated increased sleep problems compared to autistic males, TD females, and TD males. Current autism assessment and intervention practices may benefit from increased attention to sleep problems in autistic school-age females and to anxiety in autistic males to enhance well-being and behavioral and health outcomes.
Keywords: autism, sleep, sex, school-age
Lay Abstract
Sleep problems are common in autism, but little is known about sex differences in sleep problems in autism. In one of the first studies to evaluate sex differences in sleep in school-age (6- to 12-year-old) autistic children, we found that autistic girls demonstrated more sleep problems than autistic boys or typically developing (TD) girls and boys. Almost 85% of autistic girls demonstrated sleep problems compared to approximately 66% of autistic boys, 45% of TD girls, and 42% of TD boys. Autistic girls demonstrated different types of sleep problems than autistic males; specifically, more bedtime resistance, sleep anxiety, and daytime sleepiness, and less sleep overall. Higher anxiety scores were associated with more sleep problems for boys but not girls. This information could help improve assessment and intervention by alerting practitioners to the importance of sleep problems and highlights the importance of research in this area to improve outcomes for school-age children.
Sleep problems, a broad term used to describe a variety of challenges in sleep duration, timing, and quality, affect 50 to 86% of children diagnosed with an Autism Spectrum Disorder (ASD; Liu et al., 2006; Krakowiak et al., 2008). Sleep problems have significant, pervasive, health and behavioral impacts and inadequate sleep is an urgent public health concern (Dinges, 2006). Parents report that their autistic children have difficulty falling asleep, staying asleep, and waking too early, leading to insufficient and lower-quality sleep (Goldman et al., 2011; Sikora et al., 2012). Sleep problems have well-established associations with behavioral characteristics in autism (Veatch, Maxwell-Horn, Malow, 2015) and increased autism severity (Mayes & Calhoun, 2009; Johnson et al, 2018; Malow et al., 2006). Sleep problems have been associated with increased repetitive behaviors, communication problems, and social skills deficits; fundamental characteristics of autism (Goldman et al 2009; MacDuffie et al., 2020a; Park et al., 2012). However, the relationship between sleep problems and intellectual disability in autistic children is inconsistent; evident in some studies (e.g., Bernier, et al., 2014) but not others (Johnson et al., 2018, Anders et al., 2011; Krakowiak et al., 2008). In the school-age period, sleep problems may be particularly salient as compared with earlier in development. Sleep problems tend to resolve by school-age in typical development, with the sharpest decrease occurring from 4 to 7 years of age (Gregory & O’Connor, 2002). In contrast, autistic children continue to suffer from sleep problems at high rates through school-age and into adulthood (Goldman et al., 2011; Sare & Smith, 2020; Sivertsen et al., 2012). Very little is known about whether there are sex differences in sleep problems in autistic children.
Sex differences in children’s sleep problems
In TD children, sex differences in sleep begin as early as 6 months of age (Bach, Telliez, Leke, Libert, 2000). From birth through adolescence, TD females sleep longer and demonstrate less sleep fragmentation than TD males. Females begin to show higher rates of insomnia than males in adolescence (Franco et al., 2020). Extrapolating from the polygenic multiple threshold model, autistic females might be expected to demonstrate a higher prevalence of sleep problems than autistic males. This model asserts that the sex ratio in autism may be due to females requiring an increased number of causal factors as compared with males to exhibit the characteristics of autism (Eme, 1992; Robinson et al., 2013). There is emerging evidence that autistic females may have increased prevalence of sleep problems and different types of sleep problems than autistic males. In toddlers with autism, sleep problems have been reported to be more prevalent in females than males (Hartley & Sikora, 2009). Evidence from a large healthcare database showed increased odds of sleep disorders in autistic females 1 to 21 years of age, compared with autistic males (Angell et al, 2021). An online study reported no sex differences in prevalence of sleep problems in autistic individuals birth to 18 years of age but found a stronger association between repetitive behavior and sleep problems in females compared with males (Sare & Smith, 2020). The only report of sex differences in sleep in autistic school-age children of which we are aware is a small study of children 7–13 years of age in which autistic females demonstrated higher levels of daytime sleepiness than autistic males (D’Agati et al., 2020).
However, the majority of prior studies of sleep in autism report sex incidentally, as a control variable or a secondary consideration. These studies tend to report no sex differences in parent-reported sleep problems (e.g., Liu et al., 2006; Wiggs & Stores, 2004; Mayes & Calhoun, 2009; Horovitz et al., 2011), or fewer sleep problems in autistic female than autistic male children (Sivertsen et al., 2012). The existing literature is limited by the lack of comparison of autistic females to typically developing (TD) females. The lack of TD female comparison groups in previous research makes it difficult to determine whether there is an overall effect of sex on sleep or a disorder-specific interaction between autism and sex (Jamison, Bishop, Huerta, Halladay, 2017). The minimal amount of research to date, samples with wide age-ranges, and limited comparison groups, contributes to inconsistent results and prevents definitive conclusions about sex differences in sleep problems in autism. Adequately powered studies that employ standardized measures of sleep and directly assess diagnosis and intellectual ability are needed to establish the prevalence and types of sex differences in sleep problems in the school-age autistic population.
The paucity of published research on sex difference in sleep in autism is surprising given robust findings of sex differences in autism itself (Christensen et al., 2012). Autism is 3–4 times more prevalent in males than females, with more females represented in lower IQ ranges (Maenner, Shaw, Baio, 2020). There is not yet consensus about why more males have autism. Sex differences in autism may be due to direct effects of sex-linked biological factors related to genetics and sex hormones (Jacquemont et al 2014; Werling et al., 2016; Ferri et al., 2018). But differential presentation in males versus females may also contribute to the greater proportion of males diagnosed with ASD (Hiller et al., 2016; Hartley & Sikora, 2009). There also may be an under-recognition of autism in females, ascertainment bias, or bias in the criteria used to identify autism (Lai et al., 2015).
Autistic males and females may show different profiles of core and co-occurring symptoms which may affect sleep. It is now widely recognized that autistic individuals are at increased risk for anxiety disorders (Kent & Simonoff, 2017). Although relatively few studies to date examine autistic gender differences in anxiety, some emerging data suggest that females may be particularly susceptible (Kirsch et al., 2020; Wodka et al., 2021). These data are consistent with decades of research establishing increased risk for anxiety disorder among females generally (Kessler et al., 2005; McLean et al., 2011). Given that sleep disturbances are a common consequence of anxiety disorders, it follows that autistic females may experience these disturbances at higher rates than autistic males.
The aim of the current study was to examine sex differences in parent-reported sleep problems in a large sample of 6- to 12-year-old children diagnosed with autism and TD. We evaluated the degree to which the prevalence and types of sleep problems varied between autistic and TD females and males. Informed by the emerging evidence of sex differences in the prevalence of sleep problems, we predicted sleep would be more impaired in autistic females. We explored whether intellectual ability and anxiety contributed to sex differences in sleep problems. Lastly, we evaluated whether the types of sleep problems observed in (1) autistic females versus autistic males and (2) autistic females vs TD females and males suggested a disorder-specific interaction between sex and autism or an overall effect of sex.
Methods
Participants
A total of 364 participants (Autism, n = 250 and TD, n = 114), 6 to 12 years of age, were included. Children were from a larger, community-based cohort of children with autism at the Center for Autism Research (CAR) of the Children’s Hospital of Philadelphia (CHOP) enrolled between 2009 and 2015. This cohort was originally recruited via EHR records, listservs, and advertisements, and all families within a 1-hour radius of CAR who agreed to be contacted were offered an opportunity to participate. All participants provided informed consent, which was approved by CHOP’s Institutional Review Board.
Expert clinical judgment confirmed that all children in the autism group met the Diagnostic and Statistical Manual of Mental Disorders (4th ed. text rev.; DSM-IV-TR) criteria for either autistic disorder, Asperger’s syndrome, or pervasive developmental disorder-not otherwise specified. Clinical best estimates were informed by the Autism Diagnostic Interview-Revised (ADI-R; Rutter, LeCouteur, Lord, 2003), Autism Diagnostic Observation Schedule-WPS (ADOS-WPS; Lord, Rutter, DiLavore, Risi, 1999), and clinical impressions. Based on these criteria, children could have been included in the autism cohort who did not meet criteria on the ADOS. This method was utilized from the start of the project to align with community diagnostic procedures and increase the representativeness of the sample.
Exclusion criteria were extreme prematurity (gestational age <32 weeks), seizures or a seizure disorder, tic disorder, presence of an acquired head injury that interfered with neurological functioning, sensory impairment that would impact testing participation, and parent report of a genetic variation with known clinical significance (e.g. Fragile X and Down syndrome). For the TD group, additional exclusions were taking psychoactive medication or evidence of ADHD.
Data on sleep were available for 364 children (n= 250 Autism; n= 114 TD). No significant difference in age was found for the Autism vs TD group. As expected, the Autism group had significantly lower Full Scale IQ scores than the TD group (t=9.73; p<0.001; See Table 1).
Table 1.
Sample description
| Autism Male | Autism Female | TD Male | TD Female | |||||
|---|---|---|---|---|---|---|---|---|
| N=220 | N=30 | N=85 | N=29 | |||||
| Variable | M | SD | M | SD | M | SD | M | SD |
| Age (years) | 8.67 | 1.57 | 9.66 | 1.35 | 9.05 | 1.53 | 9.00 | 1.57 |
| Full scale IQ | 91.73 | 25.61 | 95.13 | 24.30 | 112.13 | 13.53 | 111.24 | 12.61 |
| CSHQ Total score | 45.54 | 8.32 | 50.03 | 7.37 | 40.36 | 5.21 | 40.55 | 5.62 |
| Above CSHQ cutoff | 147 | 66.8% | 25 | 83.3% | 36 | 42.4% | 13 | 44.8% |
| Race | ||||||||
| Asian | 3 | 1.4% | 1 | 3.3% | 1 | 1.2% | 1 | 3.4% |
| Black | 20 | 9.1% | 2 | 6.7% | 10 | 11.8% | 2 | 6.9% |
| White | 179 | 81.4% | 26 | 86.7% | 70 | 82.4% | 23 | 79.3% |
| Biracial/Other | 15 | 6.8% | 1 | 3.3% | 4 | 4.7% | 3 | 10.3% |
| Unknown/Race not specified | 3 | 1.4% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% |
| Ethnicity | ||||||||
| Hispanic | 16 | 7.3% | 2 | 6.7% | 3 | 3.5% | 0 | 0.0% |
| Not Hispanic | 192 | 87.3% | 26 | 86.7% | 74 | 87.1% | 26 | 89.7% |
| Unknown/Missing | 12 | 5.5% | 2 | 6.7% | 8 | 9.4% | 3 | 10.3% |
Measures
The Children’s Sleep Habits Questionnaire (CSHQ; Owens, Spirito, McGuinn, 2000) is a 33-item scale using a 3-point Likert scale to screen for sleep disorders. The original scale was normed on a neurotypical sample of children aged 4–10 years, but it has been subsequently implemented and evaluated with multiple cohorts of children, including those with autism, up to age 17 (e.g., Goldman et al., 2012; Shui, Richdale, Katz, 2021). Two studies have evaluated the psychometric properties of the CSHQ in autistic cohorts whose age overlaps with our sample of 6–12-year-olds (Katz et al., 2018; Shui et al., 2021), however the current cohort was finalized in 2015, prior to these studies. Thus, we employ the clinical cut-off total score of 41 or higher, a total score (maximum 123), and eight subscales: 1) bedtime resistance, 2) sleep onset delay, 3) sleep duration, 4) sleep anxiety, 5) night wakings, 6) parasomnias, 7) sleep-disordered breathing, 8) daytime sleepiness, as defined in Owen et al., 2000.
The Differential Ability Scales – Second Edition (DAS; Elliott, 2007) Early Years (n=43, Autism) and School Age Levels (Autism, n=205; TD, n=113) were used to measure child intellectual ability in the majority of children in this sample. The DAS-II is a standardized cognitive assessment tool for children 2-years-6-months to 17-years-11-months old. Standard scores on General Conceptual Ability, which reflects conceptual and reasoning ability in verbal and nonverbal domains, were included in analyses. An additional 3 children (Autism, n=2; TD, n=1) were assessed with the Wechsler Intelligence Scale for Children, Third Edition (Wechsler, 1997), which also provides a comprehensive assessment of conceptual and reasoning ability in verbal and nonverbal domains, and standard scores.
The Screen for Child Anxiety Related Emotional Disorders (SCARED; Birmaher et al., 1997) is a 41-item questionnaire collected to assess anxiety. The SCARED yields 5 anxiety factors: 1) panic/somatic, 2) GAD, 3) separation anxiety, 4) social phobia, and 5) simple phobia. A cut-off total score of greater than or equal to 25 is indicative of clinically significant anxiety problems.
Results
Data analyses were conducted in R. Two-tailed significance tests were used throughout with an alpha level of .05. Unstandardized model coefficients are reported unless otherwise noted.
Sex differences in sleep problems
Sleep problems (CSHQ total score >= 41) were more common in the Autism group (68.1%) than in the TD group (43.0%, fisher exact test, p < .001). Autistic females demonstrated the highest level of sleep problems (84.4%) followed by autistic males (65.8%), TD females (44.8%) and TD males (42.4%, fisher exact test, p <.001).
A linear regression model was used to explore differences in CSHQ total scores as a function of diagnostic group (Autism versus TD) and sex (males versus females). The diagnostic group main effect was significant; the Autism group showed higher total sleep problems (Table 2). There was also a significant interaction between group and sex, with autistic females demonstrating the highest level of sleep problems (p=0.047).
Table 2.
Predicting CSHQ Total Score by Group and Sex
| CSHQ Total Score | |||||
|---|---|---|---|---|---|
| Predictors | Estimates | std. Error | CI | Statistic | p |
| (Intercept) | 40.365 | 0.806 | 38.779 – 41.950 | 50.071 | <0.001 |
| Group [Autism] | 5.176 | 0.949 | 3.310 – 7.043 | 5.453 | <0.001 |
| Sex [Female] | 0.187 | 1.598 | −2.956 – 3.330 | 0.117 | 0.907 |
| Group [Autism] * Sex [Female] | 4.305 | 2.156 | 0.066 – 8.545 | 1.997 | 0.047 |
| Observations | 364 | ||||
| R2 / R2 adjusted | 0.133 / 0.126 | ||||
A second regression model was run in which total anxiety score, full scale IQ, and their interactions with sex were added as predictors of CSHQ total sleep problems (Table 3). Given the mean difference in IQ and anxiety scores between the groups, these variables were standardized as a function of their group means (individual score minus group mean divided by pooled standard deviation). This was done to keep the main effect of the diagnostic group variable independent of the anxiety and IQ predictors while equating their variance, making the magnitude of model estimates more directly comparable. Including the sex by anxiety and sex by IQ terms allowed us to explore the degree to which the relationship between sleep problems and anxiety and IQ might differ between males and females. The main effect for group and the group by sex interaction seen in the first analysis remained significant. The main effect for anxiety and the sex by anxiety interaction (estimate = −2.59) terms were both significant. Considered together, we see that higher anxiety scores were associated with more sleep problems for males but not females. For each unit change in anxiety (one-unit equivalent to the sample standard deviation), sleep problems changed 3.16 for boys, however for females the change in sleep problems is only 0.57 points. Neither the main effect for IQ, nor the sex by IQ interaction were significant, however, removing these terms from the whole model did result in a significant reduction in the overall variance explained (overall model R2 reduced from 0.252 to 0.237, F(2,337)=3.27, p=.039).
Table 3.
Predicting CSHQ Total Score by Group, Sex, Anxiety, and IQ
| CSHQ Total Score | |||||
|---|---|---|---|---|---|
| Predictors | Estimates | SE | CI | Statistic | p |
| (Intercept) | 40.30 | 0.76 | 38.81 – 41.78 | 53.27 | <0.001 |
| Group [Autism] | 5.32 | 0.90 | 3.56 – 7.08 | 5.94 | <0.001 |
| Sex [Female] | 0.24 | 1.49 | −2.69 – 3.18 | 0.16 | 0.870 |
| Group [Autism] * Sex [Female] | 4.77 | 2.26 | 0.32 – 9.22 | 2.11 | 0.036 |
| Total anxiety scorea | 3.16 | 0.45 | 2.27 – 4.04 | 7.01 | <0.001 |
| Full scale IQa | −0.77 | 0.45 | v1.65 – 0.11 | −1.71 | 0.088 |
| Sex [Female] * Total anxiety scorea | −2.59 | 1.22 | −5.00 – −0.18 | −2.11 | 0.035 |
| Sex [Female] * Full scale IQa | −1.70 | 1.37 | −4.40 – 1.00 | −1.24 | 0.216 |
| Observations | 345 | ||||
| R2 / R2 adjusted | 0.252 / 0.236 | ||||
variable was group-mean centered and standardized using the pooled standard deviation.
Autistic females were more likely to be above the cutoff in anxiety than autistic males (females 41.7% (10 of 24), males 25.4% (53 or 156)), however, this was not a significant difference (Fisher exact test, p = 0.095). The typical group had only 2 children above the cutoff in anxiety (males 2.4% (2 of 83), females 0% (0 of 29), Fisher exact test, p = 1.00).
We explored the relationship between having clinically elevated sleep problems and anxiety in the Autism group. Among autistic females, there was no association between being above the cutoff in anxiety and above the cutoff in sleep problems. Nine of 10 (90.0%) of autistic females with high anxiety also were above the sleep problems cutoff as were 12 of 14 (85.7%) of autistic females with low anxiety (Fisher exact test, p = 1.00). However, among autistic males there was a relationship between anxiety and sleep problems as 43 of 53 (81.1%) of autistic males with high anxiety also had sleep problems, however only 96 of 156 (61.5%) of autistic males with low anxiety also had sleep problems (Fisher exact text, p = 0.011).
Sex differences in CSHQ sleep problem subscales
Figure 1 displays the wide variety of sleep problems in this sample and shows the percentage of each group (autistic female, autistic male, TD female, TD male) whose subscale scores on the CSHQ are greater than 1 SD above the normative means reported in previous research (Owens et al., 2000). We used this threshold approach to address the different number of items across the CSHQ subscales (Sleepiness, Disordered Breathing, Parasomnias, Night Wakings, Sleep Anxiety, Sleep Duration, Sleep Onset Delay, Bedtime Resistance). The subscales were evaluated in regression models (see Table 4). Autistic females were set as the reference group in regression models to compare with each group on each CSHQ subscale using dummy coded variables representing each other group (autistic males, TD females, and TD males). This yields a total of 24 comparisons (3 group indicator variables for the 8 subscales). We adjusted the p-values using the Benjamini-Hockberg procedure to control the familywise error rate (Benjamini & Hochberg, 1995). Autistic females demonstrated significantly more impaired sleep than TD females across 7 of 8 subscales (all except Sleep Disordered Breathing) and on 7 of 8 subscales compared to TD males (all except Night Wakings). Autistic females demonstrated significantly more impaired sleep than autistic males on 4 out of 8 subscales (Bedtime Resistance, Sleep Duration, Sleep Anxiety, and Sleepiness), but did not differ in the remaining 4 (Sleep Onset Delay, Night Wakings, Parasomnias, or Sleep Disordered Breathing).
Figure 1.
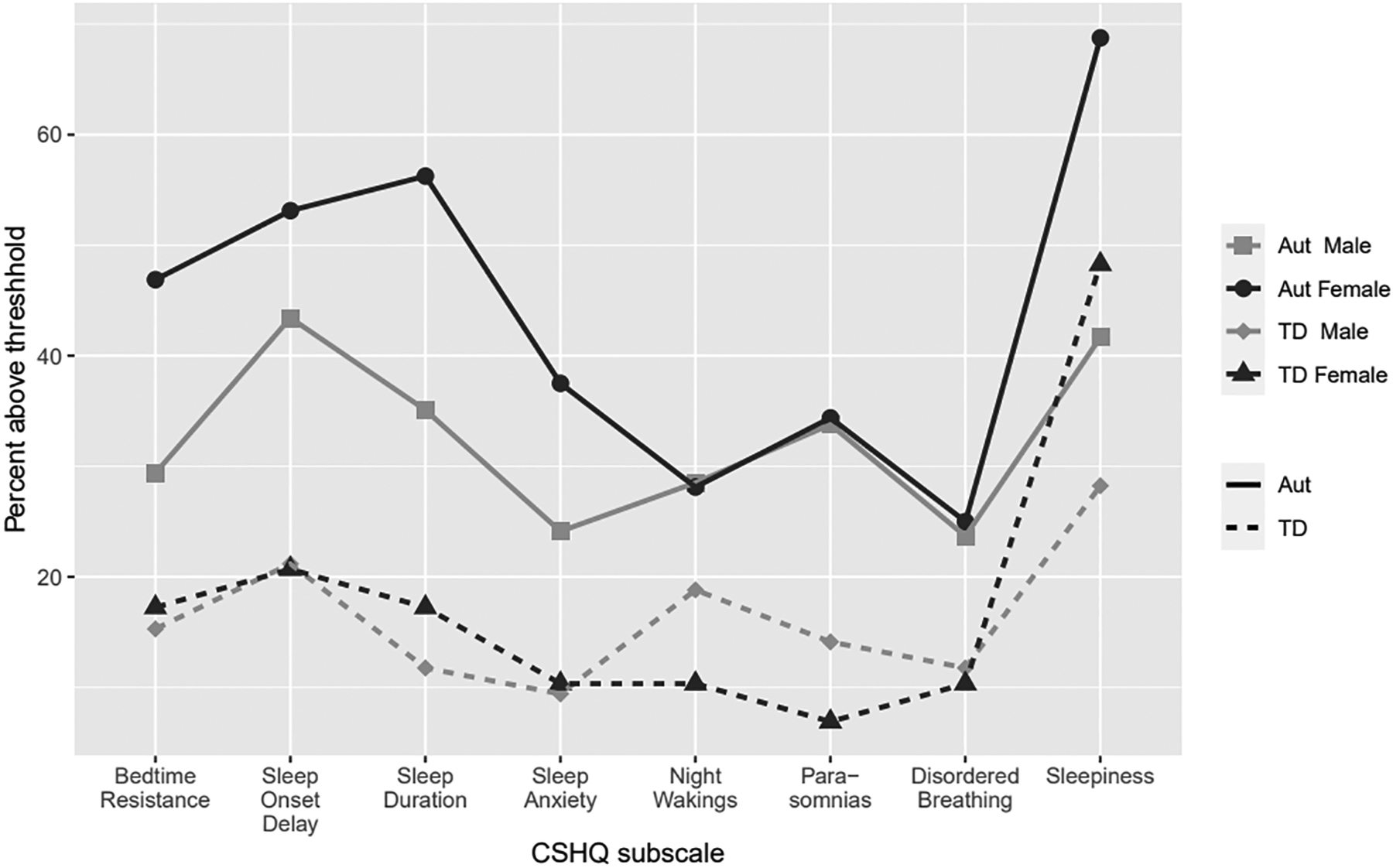
Percentage of sample above thresholda by Group and Sex
a +1 SD above previously published sample (Owen et al., 2000)
Table 4.
CSHQ Subscales
| Bedtime resistance | Sleep onset delay | |||||||
|---|---|---|---|---|---|---|---|---|
| Predictor | Est | SE | t | p BH | Est | SE | t | p BH |
| (Intercept) | 9.1 | 0.46 | 19.65 | < 0.001 | 1.77 | 0.13 | 13.13 | < 0.001 |
| Autism Male | −1.02 | 0.49 | −2.07 | 0.048 | −0.15 | 0.14 | −1.04 | 0.321 |
| TD Male | −1.94 | 0.54 | −3.59 | < 0.001 | −0.44 | 0.16 | −2.79 | 0.01 |
| TD Female | −2.1 | 0.66 | −3.18 | 0.003 | −0.53 | 0.19 | −2.74 | 0.011 |
| Observations / R2 | 364 / 0.049 | 364 / 0.045 | ||||||
| Sleep duration | Sleep anxiety | |||||||
| Predictor | Est | SE | t | p BH | Est | SE | t | p BH |
| (Intercept) | 4.87 | 0.27 | 17.82 | < 0.001 | 6.57 | 0.35 | 18.96 | < 0.001 |
| Autism Male | −0.61 | 0.29 | −2.09 | 0.048 | −0.78 | 0.37 | −2.11 | 0.047 |
| TD Male | −1.37 | 0.32 | −4.32 | < 0.001 | −1.73 | 0.4 | −4.3 | < 0.001 |
| TD Female | −1.21 | 0.39 | −3.11 | 0.004 | −1.6 | 0.49 | −3.24 | 0.003 |
| Observations / R2 | 364 / 0.069 | 364 / 0.069 | ||||||
| Night wakings | Parasomnias | |||||||
| Predictor | Est | SE | t | p BH | Est | SE | t | p BH |
| (Intercept) | 4.1 | 0.27 | 15.26 | < 0.001 | 8.9 | 0.31 | 28.82 | < 0.001 |
| Autism Male | 0.02 | 0.29 | 0.08 | 0.937 | 0.09 | 0.33 | 0.26 | 0.819 |
| TD Male | −0.46 | 0.31 | −1.49 | 0.158 | −0.76 | 0.36 | −2.11 | 0.047 |
| TD Female | −0.82 | 0.38 | −2.15 | 0.047 | −1.07 | 0.44 | −2.43 | 0.025 |
| Observations / R2 | 364 / 0.036 | 364 / 0.062 | ||||||
| Disordered breathing | Sleepiness | |||||||
| Predictor | Est | SE | t | p BH | Est | SE | t | p BH |
| (Intercept) | 3.47 | 0.13 | 27.63 | < 0.001 | 14.67 | 0.57 | 25.62 | < 0.001 |
| Autism Male | −0.14 | 0.13 | −1.08 | 0.312 | −2.54 | 0.61 | −4.17 | < 0.001 |
| TD Male | −0.31 | 0.15 | −2.15 | 0.047 | −3.65 | 0.67 | −5.49 | < 0.001 |
| TD Female | −0.33 | 0.18 | −1.84 | 0.079 | −2.87 | 0.82 | −3.52 | 0.001 |
| Observations / R2 | 364 / 0.02 | 364 / 0.078 | ||||||
pBH = p-values adjusted via Benjamini-Hochberg procedure
Discussion
This study investigated sleep problems in school-aged children, all of whom were directly evaluated for autism and intellectual ability and whose parents completed a widely used measure of pediatric sleep problems. The overall prevalence of sleep problems (i.e., CSHQ total scores above the clinical cutoff) in this cohort was consistent with other school-aged autism (e.g., Malow et al., 2006; Goldman et al, 2009) and TD cohorts (e.g., Bhargava, 2011). Autistic females demonstrated significantly increased sleep problems compared to autistic males, TD females, and TD males; almost 85% of autistic females demonstrated sleep problems compared to 65.8% of autistic males, 44.8% of TD females, and 42.4% of TD males. Interestingly, anxiety scores were more strongly associated with total sleep problem scores for males than for females. Intellectual ability was not related to sleep problem scores or sex differences. This study also revealed a different pattern of types of sleep problems for autistic females compared to autistic males, with significantly more problems on the Bedtime Resistance, Sleep Duration, Sleep Anxiety, and Sleepiness subscales. This is one of the first studies to investigate sex differences in sleep problems in school-age children diagnosed with ASD and the first of which we are aware to compare a group of well-characterized autistic females with same-age TD females.
The finding that autistic females have higher prevalence of sleep problems than autistic males or TD females was consistent with our hypothesis and with emerging evidence of sex differences in sleep problems in autistic children (Hartley & Sikora, 2009; Sare & Smith, 2020; D’Agati et al., 2020). However, these findings are based on a relatively small sample of females and differ from previous studies that report sex effects incidentally (e.g., Liu et al., 2006; Wiggs & Stores, 2004; Mayes & Calhoun, 2009; Horovitz et al., 2011; Sivertsen et al., 2012). Thus, replication in larger, independent samples are needed to determine whether the current findings are robust or might be attributed to sampling error.
The polygenic multiple threshold (PMT) model posits that females need a greater etiologic load (e.g., increased numbers or forms of autism risk-genes, environmental factors that occur early in development) to manifest the disorder (Eme, 1992; Robinson et al., 2013). Extending this logic to sleep problems, we might expect autistic females, who theoretically have a greater number of factors that disrupt typical development, to experience greater disruption to the underlying mechanisms that support adequate sleep compared to autistic males. In this interpretation, sleep problems may be more prevalent in autistic females because females require more causal factors to manifest autism.
It is also possible that sleep problems are part and parcel of autism, stemming from shared etiologic factors. In fact, some genetic factors and downstream neurobiological alterations may be shared in common between autism and sleep problems (Takumi et al., 2020; Ruzzo et al., 2019; Carmassi et al, 2019). Our previous research suggests that parent-reported sleep-onset problems in the first year of life (6 and 12 months of age) are related to atypical hippocampal growth trajectories in the group of children who go on to develop autism at 24 months of age (MacDuffie et al., 2020b). Altered sleep, prior to the detection of autism, is consistent with the possibility of a shared etiology. Although it is unclear how males and females differ with regard to sleep in the first 2 years of life, sex differences in sleep have been reported in autistic toddlers, leaving open the question of when sex differences in sleep first emerge (Hartley & Sikora 2009).
We found evidence of sex differences in the relationship between anxiety and sleep problem scores in autism. However, in contrast to our expectations, anxiety was more strongly related to sleep problems in males than in females. This was unexpected and warrants replication. If future research finds that anxiety plays a greater role in sleep problems for autistic males than autistic females, this could have clinical implications. Emerging evidence that anxiety is increased in autistic females, led us to hypothesize that anxiety might play a key role in sleep problems in autistic females. Researchers and clinicians should be aware that anxiety should be assessed and evaluated in autistic males with sleep problems, even though anxiety may be more prevalent in females in general. Future studies are needed to investigate other co-morbidities beyond anxiety that may contribute to sex differences in sleep problems, such as ADHD, and to investigate putative mechanisms that might drive sex differences in sleep in autism.
This is one of the first studies to evaluate sex differences in the types of sleep problems in school-age autistic children. Importantly, compared with TD females, autistic females had increased scores across all the types of sleep problems reported on the CSHQ except Disordered Breathing. This finding, combined with the overall pattern of sex differences in total sleep problems (autistic female > autistic male > TD female = TD male) suggests that sex per se is not driving increased challenges faced by autistic females. Within the Autism group, females demonstrated increased bedtime resistance, sleep anxiety, and sleepiness and decreased sleep duration compared with males. Future research is needed to evaluate the mechanisms driving sex differences in the types of sleep problems in autism. For example, many different factors could lead to increased bedtime resistance; from biological factors related to later chronotype to behavioral factors, such as how females react to certain parenting approaches. Despite anxiety not being related to sleep problems in autistic females, sleep anxiety was elevated in this group. Increased sleep anxiety could lead to increased bedtime resistance which could delay sleep onset and lead to decreased sleep duration and increased daytime sleepiness. Future studies utilizing objective sleep measures are need to replicate these findings and evaluate whether these 4 types of sleep problems are interrelated or independent.
If replicated, findings of increased sleep problems for autistic females may also have clinical implications. Researchers are considering whether there may be an under-recognition of autism in females, ascertainment bias, or bias in the instruments or criteria used to identify autism (Lai et al., 2015). Sleep problems have the potential to be assessed objectively, and, if sleep is less subject to bias, this may help increase recognition of autistic females. Our findings suggest that sleep problems should be carefully considered in autistic females. Reduced sleep duration and increased daytime sleepiness, as seen in the autistic female group, can have pervasive effects on health, behavior, and learning. Importantly, sleep problems are usually treatable with relatively low intensity interventions, although more work is needed to validate the use of existing approaches in this population.
Limitations of our design include the fact that sleep was assessed only via parent-report, rather than with objective measures (e.g., actigraphy, polysomnography). Although the CSHQ has been validated against objective measures, it demonstrated low to moderate concordance with sleep problems as detected by polysomnography and actigraphy (Markovich, Gendron, Corkum, 2015). In addition, this study utilized an existing dataset completed in 2015, prior to the work of Katz and colleagues (Katz et al., 2018; Shui, Richdale & Katz, 2021) in which autism-specific CSHQ cutoff scores and factors were identified. Thus, objective measures and independent samples are needed to validate and expand the results of this study. Parent-report is limited by the fact that parents are usually asleep, and often in different rooms, while their child is in bed. It may be difficult for parents to know whether a child is asleep or awake, or is showing signs of sleep disordered breathing. Parents of autistic children often have high levels of stress which might exacerbate the impact of children’s sleep problems and alter parent report of the severity of milder sleep challenges. Despite being the most widely used tool to study sleep in autistic children, the CSHQ relies on a single item to characterize sleep onset delay, a common sleep problem faced by autistic children. However, polysomnography cannot capture naturalistic sleep patterns in the home environment, bedtime behavior that might interfere with sleep (bedtime resistance, problematic sleep associations), or daytime sleepiness. Future research needs to take a biopsychosocial measurement approach, including use of objective sleep measures (e.g., actigraphy) and measures of environmental factors (e.g., parental approaches to bedtime routines, bedroom temperature, light and sound level) that could contribute to the types of sleep problems impacting autistic females in school-age.
Another limitation is that this study was cross sectional. Longitudinal studies are needed to understand potentially differing trajectories of sleep problems for females vs males from infancy through adolescence. Longitudinal designs can also help determine whether there is a relationship between school-age sleep problems in autistic females and anxiety and mood dysregulation in adolescence. A common perception is that characteristics and behaviors associated with autism lead to increased sleep problems; for example, repetitive interests might interfere with transitioning to bedtime and sleep. However, due to the transactional relationship between sleep and behavior, and the limitations of cross-sectional data, a strong causal interpretation is not warranted.
It is important to note that these findings may have limited generalizability to minoritized populations. The study population included a lower proportion of BIPOC and a higher proportion of White participants than the community from which it was drawn. Furthermore, the current study utilized clear inclusion and exclusion criteria based on gold-standard assessment tools, which is a strength of the study. However, there are questions about whether current diagnostic measures adequately capture autism in females (Lai et al., 2015). If future innovations change the diagnostic criteria for females, it will be important to determine whether these findings generalize to this newly identified population. We did not find an association of sleep problems with IQ. The lack of correlation between IQ and sleep problems in autism in our study and previous studies (Krakowiak et al., 2008) is somewhat at odds with reports of high rates of sleep problems in children with genetic variants and intellectual disability (Bernier et al., 2014). It is possible that rare genetic variants are associated with autism, low IQ and sleep, but this may be difficult to detect in large samples or samples that exclude children with lower IQ. However, some previous studies have found that IQ correlated with sleep problems in TD but not autistic children (Gunes et al., 2019). The PMT model also leads to the prediction that relatives of female probands might demonstrate increased impairment since females require a larger genetic “dose” to develop autism. We did not have family pedigree information, and it is possible that our findings would differ if we evaluated whether autistic males were part of female-containing families.
Sleep problems were highly prevalent in autistic females in this school-age sample. This is one of the first studies to evaluate sex differences in sleep in an autistic cohort with a relatively narrow age-range, utilizing a standard measure of sleep and direct assessment of autism. Sleep problems that have not resolved by school-age may have high clinical significance. Current assessment and intervention practices may benefit from increased attention to sleep problems in autistic females to reduce negative impacts on behavior and health. Autism diagnosis occurs less often and later in females without intellectual disability (Hiller, Young, Weber, 2016); increased awareness of sleep problems could improve identification of females on the autism spectrum. Furthermore, anxiety may play an under-recognized role in sleep problems for autistic males. Effective interventions exist to improve sleep and increased awareness of how sleep problems manifest in autism holds promise for increasing the health and well-being of autistic school-aged children.
Grant sponsor:
NICHD; Grant number P50HD105354; U54HD083091
Contributor Information
Annette Estes, Department of Speech and Hearing Science, Box 357920, UW Autism Center, University of Washington, Seattle, WA, 98195;.
Jeffrey Munson, Department of Psychiatry and Behavioral Sciences, Box 357920, UW Autism Center, University of Washington, Seattle, WA, 98195;.
Tanya St. John, Department of Speech and Hearing Science, Box 357920, UW Autism Center, University of Washington, Seattle, WA, 98195;.
Robin Finlayson, College of Education, Box 357920, UW Autism Center, University of Washington, Seattle, WA, 98195;.
Juhi Pandey, Center for Autism Research, Children’s Hospital of Philadelphia, Department of Psychiatry, University of Pennsylvania; 2716 South St. #5, Philadelphia, PA, 19104;.
Bridget Gottlieb, Center for Autism Research, Children’s Hospital of Philadelphia; 2716 South St. #5, Philadelphia, PA, 19104;.
John Herrington, Center for Autism Research, Children’s Hospital of Philadelphia, Department of Psychiatry, University of Pennsylvania; 2716 South St. #5, Philadelphia, PA, 19104;.
Robert T Schultz, Center for Autism Research, Children’s Hospital of Philadelphia, Department of Psychiatry, University of Pennsylvania; 2716 South St. #5, Philadelphia, PA, 19104.
References
- Anders TF, Iosif AM, Schwichtenberg AJ, Tang K, Goodlin-Jones BL. Six-month sleep–wake organization and stability in preschool-age children with autism, developmental delay, and typical development. Behavioral Sleep Medicine, 2011; 9(2), 92–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angell AM, Deavenport-Saman A, Yin L, Zou B, Bai C, Varma D, Solomon O. Sex differences in co-occurring conditions among autistic children and youth in Florida: A retrospective cohort study (2012–2019). Journal of Autism and Developmental Disorders, 2021; 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bach V, Telliez F, Leke A, Libert JP. Gender‐related sleep differences in neonates in thermoneutral and cool environments. Journal of Sleep Research, 2000; 9(3), 249–254. [DOI] [PubMed] [Google Scholar]
- Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal statistical society: series B (Methodological), 1995; 57(1), 289–300. [Google Scholar]
- Bernier R, Golzio C, Xiong B, Stessman HA, Coe BP, Penn O, Witherspoon K, Gerdts J, Baker C, Vulto-van Silfhout AT, Schuurs-Hoeijmakers JH, Fichera M, Bosco P, Buono S, Alberti A, Failla P, Peeters H, Steyaert J, Vissers LELM, Francescatto L, Mefford HC, Rosenfeld JA, Bakken T, O’Roak BJ, Pawlus M, Moon R, Shendure J, Amaral DG, Lein E, Rankin J, Romano C, de Vries BBA, Katsanis N, Eichler EE. Disruptive CHD8 mutations define a subtype of autism early in development. Cell. 2014; 17;158(2):263–276. doi: 10.1016/j.cell.2014.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhargava S Diagnosis and management of common sleep problems in children. Pediatrics in Review-Elk Grove, 2011; 32(3), 91. [DOI] [PubMed] [Google Scholar]
- Birmaher B, Khetarpal S, Brent D, Cully M, Balach L, Kaufman J, McKenzie Neer S. The Screen for Child Anxiety Related Emotional Disorders (SCARED): Scale Construction and Psychometric Characteristics, Journal of the American Academy of Child & Adolescent Psychiatry. 1997; 36(4), 545–553 [DOI] [PubMed] [Google Scholar]
- Carmassi C, Palagini L, Caruso D, Masci I, Nobili L, Vita A, & Dell’Osso L (2019). Systematic review of sleep disturbances and circadian sleep desynchronization in autism spectrum disorder: toward an integrative model of a self-reinforcing loop. Frontiers in Psychiatry, 10, 366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christensen DL, Braun KVN, Baio J, Bilder D, Charles J, Constantino JN, … Yeargin-Allsopp M. Prevalence and characteristics of autism spectrum disorder among children aged 8 years—autism and developmental disabilities monitoring network, 11 sites, United States, 2012. MMWR Surveillance Summaries. 2018; 65(13), doi.org/ 10.15585/mmwr.ss6503a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Agati E, Abate R, Gialloreti L, Napolitano C, Postorino V, Curatolo P, Mazzone L. Sleep problems in attention-deficit/hyperactivity disorder and autism spectrum disorder: Sex differences and parental stress, Psychiatry Research. 2020; 291, 10.1016/j.psychres.2020.113099. [DOI] [PubMed] [Google Scholar]
- Dinges DF (2006). The state of sleep deprivation: From functional biology to functional consequences. Sleep medicine reviews, 10(5), 303–305. 10.1016/j.smrv.2006.07.001 [DOI] [PubMed] [Google Scholar]
- Elliott CD. The Differential Ability Scales-Second Edition. 2007. San Antonio, Texas. [Google Scholar]
- Eme RF. Selective female affliction in the developmental disorders of childhood: A literature review. Journal of Clinical Child Psychology. 1992. 21(4), 354–364. [Google Scholar]
- Ferri SL, Abel T, Brodkin ES. Sex differences in autism spectrum disorder: a review. Current psychiatry reports. 2018; 20(2), 1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franco P, Putois B, Guyon A, Raoux A, Papadopoulou M, Guignard-Perret A. ..Plancoulaine S. Sleep during development: Sex and gender differences. Sleep medicine reviews. 2020; 51, 101276. [DOI] [PubMed] [Google Scholar]
- Goldman SE, Surdyka K, Cuevas R, Adkins K, Wang L, & Malow BA (2009). Defining the Sleep Phenotype in Children With Autism. Developmental Neuropsychology, 34(5), 560–573. 10.1080/87565640903133509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman SE, McGrew S, Johnson KP, Richdale AL, Clemons T, Malow BA. Sleep is associated with problem behaviors in children and adolescents with autism spectrum disorders. Research in Autism Spectrum Disorders. 2011; 5(3), 1223–1229. [Google Scholar]
- Goldman SE, Richdale AL, Clemons T, Malow BA. Parental sleep concerns in autism spectrum disorders: variations from childhood to adolescence. J Autism Dev Disord. 2012;42:531–8. [DOI] [PubMed] [Google Scholar]
- Gregory AM, O’Connor TG. Sleep problems in childhood: a longitudinal study of developmental change and association with behavioral problems. Journal of the American Academy of Child & Adolescent Psychiatry. 2002;41(8), 964–971. [DOI] [PubMed] [Google Scholar]
- Gunes S, Ekinci O, Feyzioglu A, Ekinci N, Kalinli M. Sleep problems in children with autism spectrum disorder: clinical correlates and the impact of attention deficit hyperactivity disorder. Neuropsychiatric disease and treatment. 2019;15, 763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartley SL, & Sikora DM Sex differences in autism spectrum disorder: an examination of developmental functioning, autistic symptoms, and coexisting behavior problems in toddlers. Journal of Autism and Developmental Disorders. 2009;39(12), 1715–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiller RM, Young RL, Weber N. Sex differences in pre-diagnosis concerns for children later diagnosed with autism spectrum disorder. Autism. 2016;20(1), 75–84. [DOI] [PubMed] [Google Scholar]
- Jacquemont S, Coe BP, Hersch M, Duyzend MH, Krumm N, Bergmann S, … Eichler EE. A higher mutational burden in females supports a “female protective model” in neurodevelopmental disorders. The American Journal of Human Genetics. 2014;94(3), 415–425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamison R, Bishop SL, Huerta M, Halladay AK. The clinician perspective on sex differences in autism spectrum disorders. Autism: the International Journal of Research and Practice. 2017;21(6), 772–784. 10.1177/1362361316681481. [DOI] [PubMed] [Google Scholar]
- Johnson CR, Smith T, DeMand A, Lecavalier L, Evans V, Gurka M, … Scahill L. Exploring sleep quality of young children with autism spectrum disorder and disruptive behaviors. Sleep Medicine. 2018;44, 61–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kent R, Simonoff E. Prevalence of Anxiety in Autism Spectrum Disorders. In Kerns CM, Renno P, Storch EA, Kendall PC & Wood JJ (Eds.), Anxiety in Children and Adolescents with Autism Spectrum Disorder. 2017; 5–32. Academic Press. 10.1016/B978-0-12-805122-1.00002-8 [DOI] [Google Scholar]
- Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Archives of General Psychiatry. 2005;62(6), 593–602. 10.1001/archpsyc.62.6.593 [DOI] [PubMed] [Google Scholar]
- Kirsch AC, Huebner A, Mehta S, Howie F, Weaver A, Myers S, Voigt R, Katusic S. Association of comorbid mood and anxiety disorders with autism spectrum disorder. JAMA Pediatrics. 2020;174(1), 63–70. 10.1001/jamapediatrics.2019.4368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krakowiak P, Goodlin‐Jones B, Hertz‐Picciotto I, Croen L, Hansen R. Sleep problems in children with autism spectrum disorders, developmental delays, and typical development: A population‐based study. Journal of sleep research, 2008;17(2), 197–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai MC, Lombardo MV, Auyeung B, Chakrabarti B, Baron-Cohen S. Sex/gender differences and autism: setting the scene for future research. Journal of the American Academy of Child & Adolescent Psychiatry. 2015;54(1), 11–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X, Hubbard JA, Fabes RA, Adam JB. Sleep disturbances and correlates of children with autism spectrum disorders. Child Psychiatry Hum Dev 2006;37:179–91. [DOI] [PubMed] [Google Scholar]
- Lord C, Rutter M, DiLavore PC, Risi S. Autism diagnostic observation schedule-WPS (ADOS-WPS). 1999; Los Angeles, CA: Western Psychological Services. [Google Scholar]
- Malow BA, Marzec ML, McGrew SG, Wang L, Henderson LM, Stone WL. Characterizing sleep in children with autism spectrum disorders: a multidimensional approach. Sleep. 2006;29(12), 1563–1571. [DOI] [PubMed] [Google Scholar]
- MacDuffie KE, Munson J, Greenson J, Ward TM, Rogers SJ, Dawson G, Estes A. Sleep problems and trajectories of restricted and repetitive behaviors in children with neurodevelopmental disabilities. Journal of Autism and Developmental Disorders. 2020a;50(11), 3844–3856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacDuffie KE, Shen MD, Dager SR, Styner MA, Kim SH, Paterson S, Pandey J, St. John T, Elison JT, Wolff JJ, Swanson MR, Botteron KN, Zwaigenbaum L, Piven J, Estes AM, IBIS Network. Sleep-onset problems and subcortical development in infants later diagnosed with autism spectrum disorder. The American Journal of Psychiatry. 2020b;177(6):518–525. doi: 10.1176/appi.ajp.2019.19060666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maenner MJ, Shaw KA, Baio J. Prevalence of autism spectrum disorder among children aged 8 years—autism and developmental disabilities monitoring network, 11 sites, United States, 2016. MMWR Surveillance Summaries. 2020;69(4), 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markovich AN, Gendron MA, Corkum PV. Validating the Children’s Sleep Habits Questionnaire against polysomnography and actigraphy in school-aged children. Frontiers in Psychiatry. 2015;5, 188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayes SD, Calhoun SL. Variables related to sleep problems in children with autism. Research in Autism Spectrum Disorders. 2009;3(4), 931–941. [Google Scholar]
- McLean CP, Asnaani A, Litz BT, Hofmann SG. Gender differences in anxiety disorders: Prevalence, course of illness, comorbidity and burden of illness. Journal of Psychiatric Research. 2011;45(8), 1027–1035. 10.1016/j.jpsychires.2011.03.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owens JA, Spirito A, McGuinn M. The Children’s Sleep Habits Questionnaire (CSHQ): psychometric properties of a survey instrument for school-aged children. Sleep. 2000;23(8), 1043–1051. [PubMed] [Google Scholar]
- Park S, Cho S-C, Cho IH, Kim B-N, Kim J-W, Shin M-S, et al. (2012). Sleep problems and their correlates and comorbid psychopathology of children with autism spectrum disorders. Research in Autism Spectrum Disorders, 6(3), 1068–1072. [Google Scholar]
- Philbrook LE, Hinnant JB, Elmore-Staton L, Buckhalt JA, El-Sheikh M. Sleep and cognitive functioning in childhood: Ethnicity, socioeconomic status, and sex as moderators. Developmental Psychology. 2017;53(7), 1276–1285. 10.1037/dev0000319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson EB, Lichtenstein P, Anckarsäter H, Happé F, Ronald A. Examining and interpreting the female protective effect against autistic behavior. Proceedings of the National Academy of Sciences. 2013;110(13), 5258–5262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rutter M, Le Couteur A, Lord C. Autism diagnostic interview-revised. Los Angeles, CA: Western Psychological Services, 2003;29, 30. [Google Scholar]
- Ruzzo EK, Pérez-Cano L, Jung JY, Wang LK, Kashef-Haghighi D, Hartl C, …Wall DP. Inherited and de novo genetic risk for autism impacts shared networks. Cell. 2019;178(4), 850–866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saré RM, Smith CB. Association Between Sleep Deficiencies with Behavioral Problems in Autism Spectrum Disorder: Subtle Sex Differences. Autism Research. 2020;13(10), 1802–1810. [DOI] [PubMed] [Google Scholar]
- Shui AM, Richdale AL, Katz T. Evaluating sleep quality using the CSHQ-Autism. Sleep Medicine. 2021;87, 69–76. 10.1016/j.sleep.2021.08.022 [DOI] [PubMed] [Google Scholar]
- Sikora DM, Johnson K, Clemons T, Katz T. The relationship between sleep problems and daytime behavior in children of different ages with autism spectrum disorders. Pediatrics. 2012;130(Supplement 2), S83–S90. 10.1542/peds.2012-0900F [DOI] [PubMed] [Google Scholar]
- Sivertsen B, Posserud MB, Gillberg C, Lundervold AJ, Hysing M. Sleep problems in children with autism spectrum problems: a longitudinal population-based study. Autism. 2012;16(2), 139–150. [DOI] [PubMed] [Google Scholar]
- Takumi T, Tamada K, Hatanaka F, Nakai N, Bolton PF. Behavioral neuroscience of autism. Neuroscience & Biobehavioral Reviews, 2020;110, 60–76. [DOI] [PubMed] [Google Scholar]
- Veatch OJ, Maxwell-Horn AC, Malow BA. Sleep in Autism Spectrum Disorders. Current Sleep Medicine Reports, 2015. 1(2), 131–140. 10.1007/s40675-015-0012-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wechsler D (1997). WAIS-iii. San Antonio, TX: Psychological Corporation. [Google Scholar]
- Werling DM, Parikshak NN, Geschwind DH. Gene expression in human brain implicates sexually dimorphic pathways in autism spectrum disorders. Nature Communications. 2016;7(1), 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wodka EL, Parish-Morris J, Annett RD, Carpenter L, Dillon E, Michaelson J, Kim SH, Landa R, SPARK Consortium, Kanne S. Co-occurring attention-deficit/hyperactivity disorder and anxiety disorders differentially affect males and females with autism. The Clinical Neuropsychologist. 2021;1–25. 10.1080/13854046.2021.1942554 [DOI] [PMC free article] [PubMed] [Google Scholar]


