Abstract
The rapid development of such research field as multi-omics and artificial intelligence (AI) has made it possible to acquire and analyze the multi-dimensional big data of human phenomes. Increasing evidence has indicated that phenomics can provide a revolutionary strategy and approach for discovering new risk factors, diagnostic biomarkers and precision therapies of diseases, which holds profound advantages over conventional approaches for realizing precision medicine: first, the big data of patients' phenomes can provide remarkably richer information than that of the genomes; second, phenomic studies on diseases may expose the correlations among cross-scale and multi-dimensional phenomic parameters as well as the mechanisms underlying the correlations; and third, phenomics-based studies are big data-driven studies, which can significantly enhance the possibility and efficiency for generating novel discoveries. However, phenomic studies on human diseases are still in early developmental stage, which are facing multiple major challenges and tasks: first, there is significant deficiency in analytical and modeling approaches for analyzing the multi-dimensional data of human phenomes; second, it is crucial to establish universal standards for acquirement and management of phenomic data of patients; third, new methods and devices for acquirement of phenomic data of patients under clinical settings should be developed; fourth, it is of significance to establish the regulatory and ethical guidelines for phenomic studies on diseases; and fifth, it is important to develop effective international cooperation. It is expected that phenomic studies on diseases would profoundly and comprehensively enhance our capacity in prevention, diagnosis and treatment of diseases.
Keywords: Phenomics, Diseases, Big data, Precision medicine, Diagnosis
Introduction
The societies in many countries around the world are undergoing rapid acceleration of aging. As reported by the seventh national census of China in 2020, there were up to 264 million people ages 60 and older, accounting for 18.7% of the total population of China, while there were up to 190 million people ages 65 and older, accounting for 13.5% of the total population of China (Tu et al. 2022). Since aging is a key risk factor for a number of major diseases, the aging of the society is leading to significant increases in the patients of age-related diseases (Campisi et al. 2019; Hou et al. 2019; North and Sinclair 2012; Partridge et al. 2018). In 2019, there were 3.94 million new stroke cases in China, with the incidence rate of stroke increasing by 86% from 1990 to 2019; and there were 2.19 million stroke-caused deaths in China in 2019, with the mortality rate increasing by 32.3% from 1990 to 2019 (Ma et al. 2021; Wang et al. 2022). The causes of these severe conditions are multifactorial, including limitations of effective drugs (Malik et al. 2021; Peña et al. 2017) and significant deficiency in medical imaging equipment in many areas, particularly in rural areas (Hricak et al. 2021; Khatib et al. 2018). To overcome these severe challenges, novel preventive strategies, diagnostic approaches and therapeutic strategies are urgently and critically needed. Increasing evidence has suggested that phenomics has provided a revolutionary strategy and approach for discovering new risk factors (Andrews et al. 2021; Nikpay and Mohammadzadeh 2020; Went et al. 2020; Yang et al. 2022; You et al. 2022), diagnostic biomarkers (Nicholson 2021; Xu et al. 2022; Zhang et al. 2022a) and precision therapies (Delude 2015; Nicholson 2021; Robinson 2012; Zhang et al. 2021) for human diseases.
The term ‘phenomics’ was coined by Dr. Steven A. Garan in his speech in 1996 (Jin 2021). Phenomics describes the measurement of the phenome, which is a series of measurable traits including physical, chemical, and biological traits of individuals and populations. The traits result from complex interactions among genetic factors, environmental factors, diet and symbiotic microorganisms (Jin 2021).
The data from phenomic studies are not merely regular big data of biomedicine, or an accumulation of biomedical data using various biomedical technologies. Compared with conventional studies on diseases, phenomics-based studies on diseases have the following distinct properties: first, standardization in all aspects of the studies, including the methods of measurements, data storage, data distribution and data analyses, which are required by artificial intelligence (AI)-based analyses; second, big data-driven research; and third, a majority of the data from phenomic studies are massive, multi-dimensional and structured, while such non-structured information as medical images and questionnaire also plays critical roles in phenomic studies. It is noteworthy that the data from phenomic studies on diseases belong to critical big data of biomedicine, which have the distinct characteristics as mentioned above.
After the completion of the Human Genome Project, it has become increasingly crucial to conduct comprehensive studies on human phenome, which may provide a fundamental basis for deciphering the codes for human health. International scientific community has recognized that phenomic studies can lead to elucidation of the mechanisms underlying the relationships among phenome, genome and environmental impact. An article published in Nature Review Genetics stated that there is significant deficiency in not only the information about phenome, but also the capability of studying phenome, which indicates urgent need for phenomic studies (Houle et al. 2010). It is reasonable to expect that phenomic studies, like genomic studies, would provide a revolutionary driving force for the development of new strategies and methods for biomedicine.
Phenomics has been established on the basis of the following development: first, there has been rapid development of multi-omics technologies that include genomics, proteomics, metabolomics, radiomics, epigenomics, transcriptomics and microbiomics (Hasin et al. 2017; Leon-Mimila et al. 2019; Olivier et al. 2019; Panunzio et al. 2021). Development of numerous new technologies including multi-omics technologies has enabled us to conduct large scale, multi-dimensional determinations on the biological properties of human subjects; second, development of AI and big data science has made it possible to analyze the big data from phenomic studies (Biswas and Chakrabarti 2020); third, development of modern biology and medicine has exposed significant relationships among various biological properties of human, indicating the potential and importance of not only determining the properties of individual units of life, including individual organs, tissues, cells and molecules, but also investigating the relationships among the biological properties of these units; fourth, it is required by precision medicine that multi-dimensional data of patients are collected and analyzed (Delude 2015; Houle et al. 2010; Newgard and Attie 2010; Nicholson 2021; Zhang et al. 2021); and fifth, cumulative evidence has indicated that the information from genetic studies on patients is far from sufficient for elucidating the mechanisms of diseases and establishing novel strategies for diagnosis and treatment of diseases (Delude 2015; Hasin et al. 2017; Wu et al. 2016).
Increasing evidence has indicated the significance and urgency for applications of phenomics in studying diseases (Delude 2015; Houle et al. 2010; Newgard and Attie 2010; Nicholson 2021; Zhang et al. 2021). However, since it is still in early developmental stage, phenomic studies on disease are facing multiple major challenges and tasks. The major aim of this article is to provide an overview of the significance and potential of phenomic studies on diseases, the current status of the research field, as well as the major challenges and tasks facing the research field.
Significance of Phenomic Studies on Diseases
With the increase in the life span of the human populations, there have been significant increases in the incidence of age-associated diseases (Hou et al. 2019; North and Sinclair 2012; Partridge et al. 2018). While there has been remarkable progress in our understanding on major diseases, cumulating evidence has indicated urgency to apply novel strategies to discover new risk factors, and to establish new diagnostic approaches and therapeutic strategies for diseases. For example, there has been only one drug approved by US Food and Drug Administration (FDA) for treating ischemic stroke—Recombinant Tissue-Type Plasminogen Activator (r-tPA), which was approved by FDA in 1996 (Marko et al. 2020; Zivin 2009). Due to its very limited therapeutic window and toxic side effects, only a small percentage of acute ischemic stroke (AIS) patients can be treated by the drug (Peña et al. 2017). During the last three decades, a number of clinical trials on the drugs for treating stroke have failed (Kaur et al. 2013; Matur et al. 2022). These pieces of information have indicated that human being is still far from conquering major diseases, which also indicates the critical significance of applying novel and revolutionary approaches to prevent, diagnose and treat diseases.
Increasing evidence has also indicated that it is insufficient to realize precision medicine by conducting solely genetic studies on major diseases: first, environmental factors may play crucial roles in the pathogenesis of numerous diseases, e.g., an article published in Nature reported that external risk factors play key roles in carcinogenesis, while internal risk factors contributed only 10–30% risk of developing cancer (Wu et al. 2016); second, as stated by Hasin et al., ‘given that most common, complex diseases develop over time and involve both environmental and genetic factors, full mechanistic insight will require coordinated sets of several omics data at multiple time points, collected from many disease relevant tissues’ (Hasin et al. 2017); third, as stated by Prof. Alan Attie, 'There are many steps between casual gene and phenotype at the level of body weight and blood sugar. Each step is subject to genetic variation, which can weaken links between gene and phenotype' (Delude 2015); and fourth, most major diseases result from long-term, complex interactions among numerous genetic and environmental factors, which would significantly decrease the precision for genomics-based diagnosis of diseases (Delude 2015).
There is evidence indicating that phenomic approach is a novel and revolutionary approach for accelerating profoundly the discoveries of novel risk factors (Andrews et al. 2021; Nikpay and Mohammadzadeh 2020; Went et al. 2020; Yang et al. 2022; You et al. 2022), diagnostic methods (Rahman and Rahman 2019; Viganò et al. 2012; Xu et al. 2022; Zhang et al. 2021, 2022a, e), and therapeutic strategies (Delude 2015; Robinson 2012; Zhang et al. 2021). Compared to genomic approach for studying diseases, phenomic studies on diseases determine a number of cross-scale phenomic parameters on several levels, including molecular level, cellular level, tissue and organ level, and systemic level (Table 1).
Table 1.
Representative technologies and measured phenomic parameters of human subjects in phenomic studies on diseases
| Levels of phenomic parameters | Representative technologies and measured phenomic parameters of human subjects in phenomic studies on diseases |
|---|---|
| Molecular level | (1) Routine blood assays (Moravej Aleali et al. 2019) |
| (2) Routine urine assays (Mishriki et al. 2013) | |
| (3) Routine stool assays (Kasırga 2019) | |
| (4) Genetic analyses (Gorcenco et al. 2020) | |
| (5) Epigenetic assays (Heyn and Esteller 2012) | |
| (6) Proteomic assays (D’Alessandro and Zolla 2017) | |
| (7) Metabolomic assays (Nordström and Lewensohn 2010) | |
| (8) Assays on body fluids (Boukouris and Mathivanan 2015) | |
| (9) Tumor biomarker assays (Lianidou et al. 2015) | |
| (10) Cytokine assays (Ueland et al. 2015) | |
| (11) Blood gas analysis (Thorp and Rushing 1999) | |
| Cellular level | (1) Routine blood assays (Moravej Aleali et al. 2019) |
| (2) Flow cytometry (Monneret et al. 2019) | |
| (3) Single-cell analysis (Gohil et al. 2021) | |
| (4) Immunophenotyping (Dermawan et al. 2021) | |
| Tissue and organ level | (1) Magnetic Resonance Imaging (MRI) (Choyke et al. 2003; González 2012; Muccilli et al. 2018) |
| (2) Computerized Tomography (CT) (Furukawa et al. 2004; Zeman et al. 1995) | |
| (3) Positron Emission Tomography (PET) (Deri et al. 2013; Tian et al. 2004) | |
| (4) X-rays (Jaeger et al. 2014) | |
| (5) Endoscope (Tajiri et al. 2010) | |
| (6) Bronchoscope (Ishiwata et al. 2020) | |
| (7) Ultrasound (Deshpande et al. 2010; Jensen 2007) | |
| (8) Radionuclide scanning (Datz and Taylor 1985; Pattou et al. 1998) | |
| (9) Electrocardiogram (Rudy and Burnes 1999) | |
| (10) Electroencephalogram (Michel and Murray 2012) | |
| (11) Electromyogram (Tankisi et al. 2020) | |
| (12) Cervical biopsy (Drezek et al. 2003) | |
| (13) Determinations of psychology and mental health (Cheng et al. 2019) | |
| Systemic level (Hellmann et al. 2018) | (1) Weight |
| (2) Height | |
| (3) Sex | |
| (4) Body temperature | |
| (5) Heart rate | |
| (6) Blood pressure | |
| (7) Behavior | |
| Other information (Hellmann et al. 2018) | (1) Age |
| (2) Personal history of diseases | |
| (3) Family history of diseases | |
| (4) Occupation | |
| (5) Educational level | |
| (6) Marriage status | |
| (7) Status of smoking and drinking |
The difference between conventional studies on diseases and phenomic studies on diseases may be analogous to the difference between western blot assays and proteomics. Based on scientific principles, it is highly reasonable to use the changes of human phenomes as the biomarkers for incidence, diagnosis and therapeutic effects of diseases: since human body is a system consisting of numerous closely associated components, theoretically, it is impossible that the pathological changes of a certain component of the system is a fully independent event. Instead, the pathological changes could be associated with the changes of certain other components in the system. Therefore, through comprehensive determinations and analyses of the phenomic information of human body, there should be significant potential to expose novel risk factors, diagnostic biomarkers and prognostic biomarkers.
Major Scientific Questions and Technical Questions for Phenomic Studies on Diseases
The major scientific questions for phenomics-based studies on diseases include at least the following unanswered questions:
What are the phenomic maps of diseases?
What are the major differences between the phenomic maps of the patients of certain diseases and the phenomic map of healthy subjects?
What are the major differences among the phenomic maps of the patients of various diseases?
What are the phenomic parameters that are key risk factors for diseases? What are the genes and proteins that are closely associated with the risk factors?
What are the phenomic parameters that can be used as novel diagnostic biomarkers of diseases? What are the genes and proteins that are closely associated with the new diagnostic biomarkers?
What are the phenomic parameters that can be used as novel prognostic biomarkers for diseases? How to establish protocols and models of precision therapies for diseases on the basis of phenomic information?
The following unanswered questions belong to the major technical questions for phenomic studies on diseases:
What are the standards for the measurements and data analyses in phenomic studies on diseases?
What are the novel and valuable phenomic parameters that should be measured in phenomic studies on diseases? How to develop novel methods and equipment for phenomic studies on diseases under clinical settings?
Significance and Theoretical Basis for Phenomics-Based Studies on Novel Diagnostic Biomarkers for Major Diseases
Early, non-invasive and precise diagnosis of major diseases is of pivotal significance for effective treatment of the diseases. However, there has been significant deficiency in this type of diagnostic biomarkers and methods, which has severely limited the therapeutic efficacy of the diseases. There have been numerous findings indicating that certain phenomic parameters of human body can become valuable biomarkers for diagnosis: first, the pathological changes of eye’s micro-vessels and the fiber layer of retina's nerves can become the biomarkers for diagnosis of early-stage Alzheimer’s disease (Berisha et al. 2007; Lu et al. 2010; Patton et al. 2005). Second, there have been studies indicating that the autofluorescence (AF) of the skin and the eyes can become diagnostic biomarkers for diabetes (Sparrow et al. 1992), which originates from the advanced glycation products (AGEs) of collagen (Bos et al. 2011; Mulder et al. 2006). The increased AF can also indicate the complications of diabetes such as micro-vessel damage (Gerrits et al. 2008) and kidney failure (Mulder et al. 2006). Third, our recent study has indicated that decreased green AF of lung parenchyma is a biomarker of lung cancer tissues (Zhang et al. 2022d); our latest study has indicated that the green AF of the Index Fingernails is a novel biomarker for non-invasive, rapid and economic determinations on the status of tobacco smoking (Zhang et al. 2022b); and our study has also indicated that ultraviolet (UV) radiation-induced skin's green AF is a biomarker for both non-invasive prediction of UV-induced skin damage and non-invasive evaluations of the dosages of UV exposures of the skin (Zhang and Ying 2022). Fourth, several reports have indicated that the pathological changes of the nails may become diagnostic biomarkers for diseases: the patients of Systemic Lupus Erythematosus have significant morphological changes in their nail's micro-vessels (Ciołkiewicz et al. 2010); pathological changes of the micro-vessels for nail fold microcirculation are a diagnostic biomarker for Systemic Sclerosis (Koenig et al. 2008; Hoogen et al. 2013); and the levels of Cd, Mn, Ni and Pb in the nails of prostate cancer patients are significantly higher than those of healthy subjects (Qayyum and Shah 2014). Fifth, the porphyrin's AF in the blood can become biomarkers for both cancer diagnosis (Masilamani et al. 2004) and differentiation between benign and malignant tumors (Koenig et al. 1996). Sixth, multiple studies have indicated that the pathological changes of the hair may become diagnostic biomarkers for multiple diseases (D’Anna-Hernandez et al. 2011; Denny et al. 2013; Price et al. 1980; Russell et al. 2012; Yamada et al. 2007).
During the last several years, our study has indicated that epidermal green AF can become novel biomarkers for non-invasive, label-free, economic and rapid evaluations of the levels of inflammation and oxidative stress in the body, which is generated from keratin 1 and/or keratin 10 in the keratinocytes at the spinous layer of the epidermis (Zhang et al. 2020). We have also found significant differences between the green AF intensity of healthy subjects and that of AIS patients (Wu et al. 2018b), lung cancer (Zhang et al. 2022a), and Parkinson’s disease (Wu et al. 2018a) at multiple skin’s locations examined. The AF properties have shown excellent promise for becoming new diagnostic biomarkers for the diseases, e.g., solely based on the skin's green AF intensity at the Dorsal Index Fingers of AIS patients, the area under curve (AUC) for differentiating AIS patients from the healthy subjects is 0.88 (Wu et al. 2018b). Based on these findings, we have proposed a novel theory for diagnosis of diseases—‘Pattern of AF Theory’: ‘Several properties of the keratin-based green AF at multiple locations of the skin, including AF intensity, AF asymmetry, locations of AF increase, and structure of the AF images, form the ‘Pattern of AF’. Each major disease has its characteristic ‘Pattern of AF’, based on which major diseases may be diagnosed (Wu et al. 2018b)’.
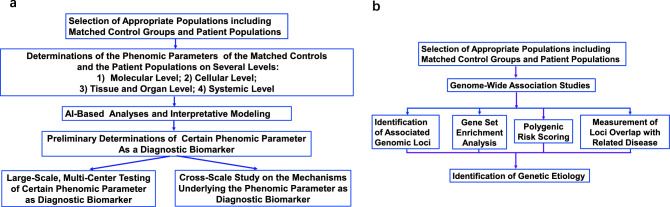
It is expected that phenomic studies on diagnostic biomarkers may profoundly enhance our capacity to discover novel diagnostic biomarkers and methods. Compared with conventional approaches, phenomics-based studies on diagnostic biomarkers have the following distinct merits: first, in the phenomics-based studies, the associations between the disease incidence and numerous phenomic parameters can be analyzed by AI, which may enhance greatly the efficiency for discovering new diagnostic biomarkers; second, the big data from phenomics-based studies enable us to elucidate the relationships among the diagnostic biomarkers and various phenomic parameters of patients; and third, the phenomics-based studies do not require findings from previous animal studies or cell culture studies. A proposed diagram for phenomic studies on diagnostic biomarkers of diseases is shown as Fig. 1a. For comparisons, a diagram for genetic studies on diagnostic biomarkers of diseases is shown as Fig. 1b.
Fig. 1.
a A proposed diagram for phenomic studies on diagnostic biomarkers of diseases. b A diagram for genetic studies on diagnostic biomarkers of diseases
Significance and Theoretical Basis for Phenomic Studies on the Risk Factors of Major Diseases
Disease prevention is a crucial strategy for overcoming the challenges from major diseases. Discovery of risk factors is required for not only prevention of major diseases, but also establishment of novel strategies for elucidating the mechanisms underlying the pathogenesis of the diseases. Conventional approaches for discovering risk factors of diseases have three major limitations: first, these studies usually focus on a single hypothesis or a single factor, which leads to relatively low efficiency in discovering new risk factors; second, the data from these studies are usually not rich enough to expose the relationships between the risk factor and other biological parameters of the subjects; and third, previous findings from biological experiments are usually required for the design of these studies.
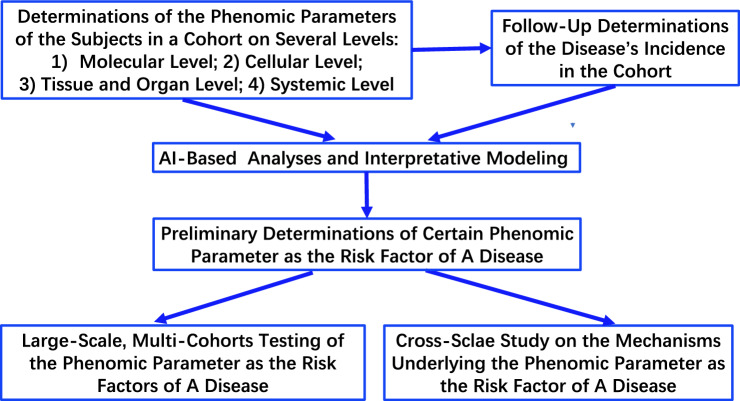
Applications of phenomics may enhance revolutionarily our capacity to discover new risk factors of diseases, which have the following distinct merits compared to conventional approaches: first, phenomics-based studies can search for multiple new risk factors in a single study by investigating the associations between the disease incidence and a number of phenomic parameters of the subjects (Andrews et al. 2021; Nikpay and Mohammadzadeh 2020; Went et al. 2020; Yang et al. 2022; You et al. 2022); second, the rich and multi-dimensional data from the phenomics-based studies enable us to investigate the relationships between the risk factors and other phenomic parameters of the subjects, which can profoundly enhance our understanding on the mechanisms underlying the associations between the risk factors and disease incidence; and third, the phenomics-based studies are big data-driven studies that do not require the findings from previous biological experiments. A proposed diagram for phenomic studies on risk factors of diseases is shown as Fig. 2.
Fig. 2.
A proposed diagram for phenomic studies on risk factors of diseases
Significance and Theoretical Basis for Phenomic Studies on Precision Therapies for Major Diseases
A number of clinical studies have found that under many circumstances, a specific therapy is effective in treating only one specific group of the patients of a certain disease (Jones and Baldwin 2018). Therefore, it is critical to establish personalized therapeutical protocols so as to conduct precision therapies. However, there has been significant deficiency in the strategies and methods for developing precision therapies. Genetic analysis has been a major strategy for establishing personalized therapies, which has shown significant limitations due to both complexity of diseases and significant impact of environmental factors on the development of diseases (Delude 2015; Hennekam and Biesecker 2012; Robinson 2012).
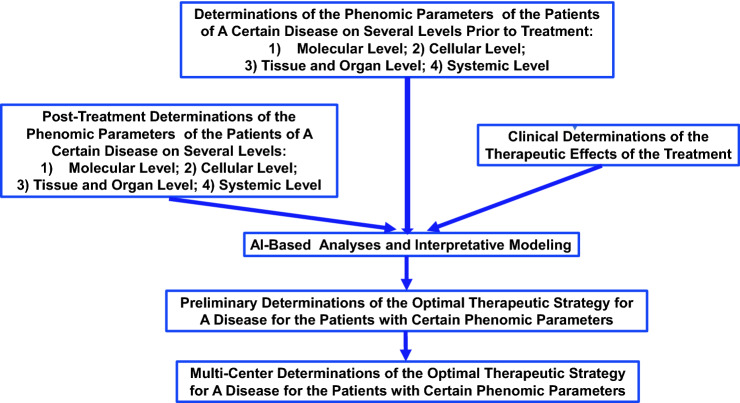
Phenomics appears to be one of the key areas for future development of precision medicine (Denny and Collins 2021). It is expected that by combining phenomic studies and genomic studies, the protocols of precision therapies may be established: Through AI-based investigation of the massive, multi-dimensional phenomic data collected before, during and after a certain therapy, there is a significant possibility to discover that certain phenomic parameters of patients are strongly associated with excellent, satisfactory or poor clinical outcomes. Based on these studies, a certain therapy may be applied only for the patients who have certain characteristic phenomic parameters and genomic properties, which may lead to establishment of precision therapies for major diseases. A proposed diagram for phenomic studies on precision therapies is shown as Fig. 3.
Fig. 3.
A proposed diagram for phenomic studies on precision therapies of diseases
Phenomic Studies on Diseases may Significantly Improve both Western Medicine and Traditional Chinese Medicine
Phenomic studies on diseases may profoundly transform both Western medicine and traditional Chinese medicine. One distinct property of Western medicine is that under most circumstances, only the key disease-related organ is the major target for medical examinations, which largely ignores the significance of the other phenomic changes of the patients. Phenomic studies on diseases may profoundly enhance the capacity of Western medicine by examining not only the key disease-related organ, but also other phenomic changes of the patients, which would significantly enhance the possibility of discovering new diagnostic biomarkers and establishing precision therapies.
Compared to Western medicine, traditional Chinese medicine relies highly on the observations of phenomic changes of the body as a system. However, traditional Chinese medicine lacks both precise measurements on the biological properties of the body and precise data-based elucidation of the theories of traditional Chinese medicine. It is expected that phenomic studies on diseases may provide an important basis for systemic and quantifiable explanations for the theories of traditional Chinese medicine.
Potential of Phenomic Studies for Development of Medical Instrumentation
The development of medical instrumentation such as medical imaging equipment and medical robotics has played an important role in the advancement of modern medicine, e.g., Magnetic Resonance Imaging (MRI) and Computerized Tomography (CT) imaging plays pivotal roles in AIS diagnosis (El-Koussy et al. 2014; Vilela and Rowley 2017); and Positron Emission Tomography (PET) imaging plays a critical role in cancer diagnosis (Zhang et al. 2022e). However, due to such factors as the cost of the medical instrumentation, there is significant deficiency in the medical equipment in many areas around the world, particularly in rural areas (Hricak et al. 2021; Khatib et al. 2018). Even in developed countries, MRI imaging resource is deficient in acute settings (Wintermark et al. 2013). This deficiency has severely limited the quality and efficiency of diagnosis and treatment of major diseases such as AIS. Phenomic studies on diseases have established a novel pathway for determining the association between incidence of a disease and a certain phenomic parameter that is usually overlooked in previous studies. Novel, economic and efficient diagnostic equipment may be established, based on phenomics-based discovery of novel diagnostic biomarkers, for example, a Portable Human Fluorescence Imaging Device has been invented in recent years for diagnosis of such diseases as AIS and lung cancer (Wu et al. 2018b; Zhang et al. 2022a).
Significance and Theoretical Basis for Phenomic Studies on Human Wellness
In addition to its wide applications to the studies on diseases, phenomics has extensive applications on studies on human wellness. One example for this type of study is phenomic study on human aging: human aging is highly complex and systemic alterations of the biological properties of human body, which can be affected by multiple mechanisms (Campisi et al. 2019). To obtain comprehensive picture of aging so as to elucidate the essential mechanisms of aging and discover hallmarks of aging, it is critical to conduct comprehensive determinations on the multi-dimensional and cross-scale phenomic parameters of both young and aging subjects. Rapid increases in applications of Omics approaches and AI in aging studies have established a strong basis for phenomics-based studies on aging (Mkrtchyan et al. 2020). It is expected that phenomic studies on aging can not only provide remarkably more comprehensive information regarding the biological alterations in the aging process, but also enable us to elucidate the mechanisms of aging by conducting AI-based, cross-scale determinations of the relationships among the microscopic and macroscopic properties of human body in the aging process.
Significance of Medical Imaging in Phenomic Studies on Diseases
Medical imaging, including MRI, CT and PET, has become indispensable tools for diagnosis of numerous diseases such as stroke (El-Koussy et al. 2014; Vilela and Rowley 2017) and cancer (Zhang et al. 2022e). As stated below, cumulating evidence has indicated that medical imaging techniques will play a critical role in phenomic studies on diseases, which is established on the basis of rapid technical development of medical imaging technologies, increased integration between medical imaging technologies and multi-omics technologies, and accelerated applications of AI technology in medical imaging.
AI technology has played an increasingly important role in MRI image analyses (Sheth and Giger 2020). Cumulating evidence has also indicated the promise that MRI datasets can be integrated with Omics datasets (Huang and Shih 2014; Leithner et al. 2018; Li et al. 2021b). To integrate MRI images with omics datasets, imaging techniques should be fast, accurate, safe and convenient. As a critical technical advance of MRI, the speed of MRI equipment has been significantly increased by the development of parallel computing technology and Dynamic Acceleration technology (Hamilton et al. 2017; Huang and Shih 2014). Collectively, these major advances of MRI technique have established strong basis for MRI to become a key technology in phenomic studies on diseases.
AI technology has played an increasingly important role in CT image analyses (Shi et al. 2021). Cumulative evidence has also indicated the promise that CT datasets can be integrated with Omics datasets (Huang and Shih 2014; Leithner et al. 2018; Li et al. 2021b). As a major technical advance of CT, radiation dosages of CT have been significantly reduced by several technologies (Huang and Shih 2014). Collectively, these major advances of CT technique have established solid basis for CT to become an important technology in phenomics-based studies on diseases.
PET is a major molecular imaging technology that can provide non-invasive whole-body phenotyping, which plays critical roles in diagnosis of such major diseases as cancer (Jin et al. 2022; Zhang et al. 2022e) and Alzheimer’s disease (Chapleau et al. 2022; Cook et al. 2014). PET is capable of assessing precisely multiple biological processes in the body, including signal transduction, protein expression, receptor availability, transporter systems, and gene mutation (Zhang et al. 2022e). The major current advances of PET technology have enabled PET to play significantly greater role in phenomic studies on diseases: first, the key deficiency of PET imaging is its relatively low spatial resolution (Surti 2015; Wollenweber et al. 2016). To overcome this shortcoming, PET imaging has been integrated with CT imaging or MRI imaging to form hybrid imaging equipment, including PET/CT and PET/MRI (Tian et al. 2021). The hybrid imaging equipment can achieve the distinct technical merits of both imaging techniques: it can obtain patients' medical images in both microscopic and macroscopic scale in the same platform, which can indicate the pathological state of patients in molecular, structural and functional aspects (Tian et al. 2021); second, novel PET tracers other than β-2-[18F]-Fluoro-2-deoxy-D-glucose (18F-FDG) are expected to further enhance its capacity to decode phenotypes of diseases (Zhang et al. 2022e); and third, there have been rapid increases in the applications of AI in PET studies (Arabi et al. 2021; Sitek et al. 2021).
Due to the rapid progresses of medical imaging technologies, Tian et al. has proposed a novel concept for the emerging new pathology, named ‘Transpathology’ (Tian et al. 2021). There are three major concepts in ‘Transpathology’, including trans-scale, transparency, and translation (Tian et al. 2021): a trans-scale imaging mode is applied for evaluations of the pathological changes of patients; multimodal probe technology can make the whole body of patients ‘transparent’, leading to markedly increased understanding on the pathological changes of patients; and translation of the discoveries from basic research to clinical practice would accelerate the realization of precision medicine. 'With the development of genomics, proteomics, and metabolomics, the organic combination of macro and micro imaging technology, and the progress in information and AI technology, clinical pathology will be promoted toward the pattern of cross-scale, multi-mode 'transparent pathology' (Tian et al. 2021)'.
Current Status of Phenomic Studies on Diseases
Twenty-seven clinical research centers from four countries including US and Canada established a joint research team in 2007 to initiate the Epilepsy Phenome/Genome Project (EPGP) (Abou-Khalil et al. 2013). They have established detailed procedures for cohort study and conducted determinations of the variety of phenomes (Abou-Khalil et al. 2013). Their study has indicated that the white people in the Caucasian region have high probability of developing Generalized Epilepsy compared with other races (Friedman and Fahlstrom 2013). They have also found that cerebellar gyrus can be used as a phenotype for predicting and examining the incidence of epilepsy (Shain et al. 2013). An Endometriosis Phenome and Biobanking Harmonisation Project was established, which applies both phenomic and genomic approaches to study endometriosis (Zondervan et al. 2016). The scientific goal of this project is to enhance classifications of endometriosis on the basis of both phenomic and genomic information of the patients (Zondervan et al. 2016).
A research team from Denmark proposed that based on the analyses on human protein complex, virtual prediction of familial diseases can be conducted (Lage et al. 2007). It was also reported that by applying machine learning technology to study Autism spectrum disorder, thousands of phenotypes that indicate cognitive ability, movement capacity and biorhythm can be analyzed (Bruining et al. 2014). Multiple phenomic studies on neurological diseases using animal models have been reported, e.g., it was reported that by machine learning-based analyses on numerous phenotypes that indicate cognitive ability and movement capacity in an animal model of Huntington disease, the number of CAG repeats and the age of mice could be predicted precisely (Alexandrov et al. 2016).
There have also been certain progresses on the methods for analyzing phenomic data. Based on machine learning, Genome-wide association study (GWAS) and phenome-wide association study (PheWAS) have been used jointly to study the relationships between the genotypes and the phenotypes of Type II diabetes (Zheng et al. 2017). As a tool for analyzing the phenomic data of cardiovascular diseases, PheWAS has been developed to discover new diagnostic and prognostic biomarkers (Han et al. 2015). PheWAS has also been used to analyze the data from epidemiological studies, clinical trials and electronic medical records (Pendergrass and Ritchie 2015; Roden 2017).
The development of the China-based International Human Phenome Project, led by Prof. Li Jin, has provided a major driving force for the progress of phenomic research. One distinct progress of phenomic studies has also originated from the founding of the first international journal that focuses at studies on human phenomes –Phenomics (Jin 2021).
A recent study has applied the strategies and methods of phenomic study to investigate the underlying genetic basis for differences in facial morphology in East Asian and European populations (Zhang et al. 2022c): they were the first group to apply Polygenic Shape Analysis (PSA) to conduct simulation of facial morphology; their PheWAS analysis showed that the facial morphology-influencing loci also influence other parameters of human phenome; and they also discovered that the locus rs6843082 is strongly associated with the risk of developing atrial fibrillation. Their findings have implicated the associations between facial morphology and development of cardiovascular and cerebrovascular diseases.
One recent phenomic study has indicated a key role of limb development genes in influencing the outcome of fingerprint patterning (Li et al. 2022): by applying genome-wide scans of Han Chinese cohorts, the authors identified 18 loci that are associated with the fingerprint patterns across the digits; they established a role of ecotropic viral integration site 1 (EVI1) in dermotoglyph patterning in mice; they found that EVI1 plays a role in shaping the digits and limbs; 43 fingerprints-associated loci were identified by transethnic meta-analysis, with nearby genes being enriched for limb development pathways; and hand proportions were genetically correlated with fingerprint patterns. In general, the study has found a strong association between fingerprint patterns and the phenotypes of limbs, the mechanism underlying which is the finding that EVI1 is the common gene that influences development of both limbs and fingerprint patterns.
A study by Li et al. reported their findings on high‑altitude acclimatization in a Chinese Han longitudinal cohort (Li et al. 2021a): they defined ‘composite phenotypes’ by developing a strategy that combines partial least squares path modeling and spectral clustering, which can be used to reveal hidden population's physiological heterogeneity in high-altitude acclimatization. In the evaluations of model fitness performance, a model based on composite phenotypes fit better than single trait-based model in modeling oxygen saturation changes in high-altitude acclimatization. The authors proposed that the new composite phenotype-based strategy may become a general strategy for studies on complex traits such as analyses of phenomics.
It has also been reported that cancer can be detected up to four years before current standard of care (Chen et al. 2020): a non-invasive blood test based on circulating tumor DNA methylation detected five common types of cancer in 88% of post-diagnosis patients with a specificity of 96%. Another study has also shown that higher serum levels of glutamate, tyrosine, acetate, glycine, and phenylalanine were negatively related to incidence of dementia, while higher serum levels of glutamine and O-acetyl glycoproteins were associated with increased risk of dementia (Cui et al. 2020).
Based on their PheWAS analysis, a recent study by Liu et al. has discovered novel links between genetically determined levels of liver enzymes and disease phenotypes (Liu et al. 2022): genetically determined alanine aminotransferase (ALT) levels were associated with liver-related diseases; genetically predicted alkaline phosphatase (ALP) levels were associated with pulmonary heart disease, hypercholesterolemia, phlebitis, and thrombophlebitis of lower extremities; genetically determined λ-glutamyl transferase (GGT) levels were associated with such diseases as chronic non-alcoholic liver disease and cholelithiasis; and genetically determined aspartate aminotransferase (AST) levels were associated with the diseases of a wide range of phenotypic categories.
By studying the Biobank cohort, the latest study by Yang et al. examined systematically the women-specific trajectory of the disease network, including blood and urine biomarkers as well as the role of baseline physical examination indexes (Yang et al. 2022): among 301 diseases, 82 diseases in women had odds ratios (ORs) > 1.2 and p < 0.00017 when compared to men, mainly including diseases in the endocrine, digestive and skeletal systems. Diseases with the highest ORs included breast diseases, hyperthyroidism, osteoporosis, and deformity of the toes.
The review of Nicolson stated the importance of molecular phenomics in deconvolving the systemic effects of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) infection and post-acute Corona Virus Disease 2019 (COVID-19) syndrome (Nicholson 2021): molecular phenomics mainly studies the chemical and biochemical signatures of cells and biofluids, as well as the characteristic changes of these signatures during the onset, development, and recovery phase of the disease. It is critical to obtain these pieces of information so as to provide molecular characterization of COVID-19 and elucidate the systemic effects of the disease.
Major Challenges and Tasks Facing Phenomic Studies on Diseases
The major challenges and tasks facing phenomic studies on diseases include:
Since phenomic studies on diseases can generate multi-dimensional big data in the size that is remarkably larger than that obtained by conventional studies on diseases, effective analyses of the data are essential for establishing valuable models. While there has been certain progress on this research direction (Han et al. 2015; Yang et al. 2017), the methods for the data analyses are still deficient. It is critical to establish the strategies and methods for analyzing phenomic data.
It is necessary to establish standardized protocols and methods for the data acquirement, data storage, data distribution and data usages in the phenomic studies on diseases, which is required for effective analyses, sharing and development of unified phenomic data banks on the basis of the phenomic data obtained by various organizations.
It is necessary to discover novel phenomic parameters that should be included as the fundamental parameters for phenomic studies on diseases.
Numerous analytic equipment for measuring biological parameters has been designed mainly for laboratory use. It is necessary to develop equipment that is used for measurements of phenomic parameters of patients under clinical settings.
It is essential to realize the potential of phenomic data by effective data analyses and data sharing, so as to promote the establishment of novel technology and equipment for enhancing our capacity of diagnosis and treatment of patients.
It is necessary to conduct persistent searches for the developmental strategies for phenomic studies on diseases. It is also important to conduct studies on the ethical and regulatory guidelines for phenomics-based studies on diseases.
It is important to establish large-scale phenomic research centers that can conduct multidisciplinary research. The centers also hold the responsibility of education and training of talented people for this research field.
It is critical to conduct international cooperation, in which each country can use its unique strength and potential to promote the development of this important field.
Conclusions
Systemic phenomics-based studies on diseases are critically needed, due to the profound potential of the studies. It is expected that phenomics will profoundly enhance human being's capacity in prevention, diagnosis and treatment of diseases. The multi-dimensional and muti-layer biomedical big data from phenomic studies on diseases would be an invaluable resource for both understanding the mechanisms of diseases and development of new biomedical technology and devices. It is expected that phenomics-based studies on diseases would become a key strategy for realizing precision medicine. With the development of the International Human Phenome Project, it is reasonable to expect that the ‘Golden Age of Phenomics’ is arriving.
Acknowledgements
The author would like to acknowledge the financial support by two research grants from Shanghai Municipal Science and Technology Major Project (No. 2017SHZDZX01) (to W.Y.). The author also would like to thank Dr. Mingchao Zhang, Yinghui Meng and Youjun Zhang for assisting literature search.
Author Contributions
WY conceived the ideas, performed the literature search, and wrote the manuscript.
Data Availability
Not applicable.
Code Availability
Not applicable.
Declarations
Conflict of Interest
The author claims no conflicts of interests.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
References
- Abou-Khalil B, Alldredge B, Bautista J, Berkovic S, Bluvstein J, Boro A, Cascino G, Consalvo D, Cristofaro S, Crumrine P, Devinsky O, Dlugos D, Epstein M, Fahlstrom R, Fiol M, Fountain N, Fox K, French J, Freyer Karn C, Friedman D, Geller E, Glauser T, Glynn S, Haas K, Haut S, Hayward J, Helmers S, Joshi S, Kanner A, Kirsch H, Knowlton R, Kossoff E, Kuperman R, Kuzniecky R, Lowenstein D, McGuire S, Motika P, Nesbitt G, Novotny E, Ottman R, Paolicchi J, Parent J, Park K, Poduri A, Risch N, Sadleir L, Scheffer I, Shellhaas R, Sherr E, Shih JJ, Shinnar S, Singh R, Sirven J, Smith M, Sullivan J, Thio LL, Venkat A, Vining E, von Allmen G, Weisenberg J, Widdess-Walsh P, Winawer M. The epilepsy phenome/genome project. Clin Trials. 2013;10(4):568–586. doi: 10.1177/1740774513484392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexandrov V, Brunner D, Menalled LB, Kudwa A, Watson-Johnson J, Mazzella M, Russell I, Ruiz MC, Torello J, Sabath E, Sanchez A, Gomez M, Filipov I, Cox K, Kwan M, Ghavami A, Ramboz S, Lager B, Wheeler VC, Aaronson J, Rosinski J, Gusella JF, MacDonald ME, Howland D, Kwak S. Large-scale phenome analysis defines a behavioral signature for Huntington's disease genotype in mice. Nat Biotechnol. 2016;34(8):838–844. doi: 10.1038/nbt.3587. [DOI] [PubMed] [Google Scholar]
- Andrews SJ, Fulton-Howard B, O'Reilly P, Marcora E, Goate AM. Causal associations between modifiable risk factors and the Alzheimer's Phenome. Ann Neurol. 2021;89(1):54–65. doi: 10.1002/ana.25918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arabi H, AkhavanAllaf A, Sanaat A, Shiri I, Zaidi H. The promise of artificial intelligence and deep learning in PET and SPECT imaging. Phys Med. 2021;83:122–137. doi: 10.1016/j.ejmp.2021.03.008. [DOI] [PubMed] [Google Scholar]
- Berisha F, Feke GT, Trempe CL, McMeel JW, Schepens CL. Retinal abnormalities in early Alzheimer's disease. Invest Ophthalmol Vis Sci. 2007;48(5):2285–2289. doi: 10.1167/iovs.06-1029. [DOI] [PubMed] [Google Scholar]
- Biswas N, Chakrabarti S. Artificial Intelligence (AI)-based systems biology approaches in multi-omics data analysis of cancer. Front Oncol. 2020;10:588221. doi: 10.3389/fonc.2020.588221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bos DC, de Ranitz-Greven WL, de Valk HW. Advanced glycation end products, measured as skin autofluorescence and diabetes complications: a systematic review. Diabetes Technol Ther. 2011;13(7):773–779. doi: 10.1089/dia.2011.0034. [DOI] [PubMed] [Google Scholar]
- Boukouris S, Mathivanan S. Exosomes in bodily fluids are a highly stable resource of disease biomarkers. Proteomics Clin Appl. 2015;9(3–4):358–367. doi: 10.1002/prca.201400114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruining H, Eijkemans MJ, Kas MJ, Curran SR, Vorstman JA, Bolton PF. Behavioral signatures related to genetic disorders in autism. Mol Autism. 2014;5(1):11. doi: 10.1186/2040-2392-5-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campisi J, Kapahi P, Lithgow GJ, Melov S, Newman JC, Verdin E. From discoveries in ageing research to therapeutics for healthy ageing. Nature. 2019;571(7764):183–192. doi: 10.1038/s41586-019-1365-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapleau M, Iaccarino L, Soleimani-Meigooni D, Rabinovici GD. The role of amyloid PET in imaging neurodegenerative disorders: a review. J Nucl Med. 2022;63(Suppl 1):13s–19s. doi: 10.2967/jnumed.121.263195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X, Gole J, Gore A, He Q, Lu M, Min J, Yuan Z, Yang X, Jiang Y, Zhang T, Suo C, Li X, Cheng L, Zhang Z, Niu H, Li Z, Xie Z, Shi H, Zhang X, Fan M, Wang X, Yang Y, Dang J, McConnell C, Zhang J, Wang J, Yu S, Ye W, Gao Y, Zhang K, Liu R, Jin L. Non-invasive early detection of cancer four years before conventional diagnosis using a blood test. Nat Commun. 2020;11(1):3475. doi: 10.1038/s41467-020-17316-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng LH, Liu YW, Wu CC, Wang S, Tsai YC. Psychobiotics in mental health, neurodegenerative and neurodevelopmental disorders. J Food Drug Anal. 2019;27(3):632–648. doi: 10.1016/j.jfda.2019.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choyke PL, Dwyer AJ, Knopp MV. Functional tumor imaging with dynamic contrast-enhanced magnetic resonance imaging. J Magn Reson Imaging JMRI. 2003;17(5):509. doi: 10.1002/jmri.10304. [DOI] [PubMed] [Google Scholar]
- Ciołkiewicz M, Kuryliszyn-Moskal A, Klimiuk PA. Analysis of correlations between selected endothelial cell activation markers, disease activity, and nailfold capillaroscopy microvascular changes in systemic lupus erythematosus patients. Clin Rheumatol. 2010;29(2):175–180. doi: 10.1007/s10067-009-1308-7. [DOI] [PubMed] [Google Scholar]
- Cook GJR, Siddique M, Taylor BP, Yip C, Chicklore S, Goh V. Radiomics in PET: principles and applications. Clin Transl Imaging. 2014;2(3):269–276. doi: 10.1007/s40336-014-0064-0. [DOI] [Google Scholar]
- Cui M, Jiang Y, Zhao Q, Zhu Z, Liang X, Zhang K, Wu W, Dong Q, An Y, Tang H, Ding D, Chen X. Metabolomics and incident dementia in older Chinese adults: the shanghai aging study. Alzheimers Dement. 2020;16(5):779–788. doi: 10.1002/alz.12074. [DOI] [PubMed] [Google Scholar]
- D’Alessandro A, Zolla L. Proteomic analysis of red blood cells and the potential for the clinic: what have we learned so far? Expert Rev Proteomics. 2017;14(3):243–252. doi: 10.1080/14789450.2017.1291347. [DOI] [PubMed] [Google Scholar]
- D'Anna-Hernandez KL, Ross RG, Natvig CL, Laudenslager ML. Hair cortisol levels as a retrospective marker of hypothalamic-pituitary axis activity throughout pregnancy: comparison to salivary cortisol. Physiol Behav. 2011;104(2):348–353. doi: 10.1016/j.physbeh.2011.02.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Datz FL, Taylor A. The clinical use of radionuclide bone marrow imaging. Semin Nucl Med. 1985;15(3):239–259. doi: 10.1016/S0001-2998(85)80003-9. [DOI] [PubMed] [Google Scholar]
- Delude CM. Deep phenotyping: the details of disease. Nature. 2015;527(7576):S14–S15. doi: 10.1038/527S14a. [DOI] [PubMed] [Google Scholar]
- Denny JC, Collins FS. Precision medicine in 2030-seven ways to transform healthcare. Cell. 2021;184(6):1415–1419. doi: 10.1016/j.cell.2021.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denny JC, Bastarache L, Ritchie MD, Carroll RJ, Zink R, Mosley JD, Field JR, Pulley JM, Ramirez AH, Bowton E, Basford MA, Carrell DS, Peissig PL, Kho AN, Pacheco JA, Rasmussen LV, Crosslin DR, Crane PK, Pathak J, Bielinski SJ, Pendergrass SA, Xu H, Hindorff LA, Li R, Manolio TA, Chute CG, Chisholm RL, Larson EB, Jarvik GP, Brilliant MH, McCarty CA, Kullo IJ, Haines JL, Crawford DC, Masys DR, Roden DM. Systematic comparison of phenome-wide association study of electronic medical record data and genome-wide association study data. Nat Biotechnol. 2013;31(12):1102–1110. doi: 10.1038/nbt.2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deri MA, Zeglis BM, Francesconi LC, Lewis JS. PET imaging with 89Zr: from radiochemistry to the clinic. Nucl Med Biol. 2013;40(1):3–14. doi: 10.1016/j.nucmedbio.2012.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dermawan JK, Azzato EM, Goldblum JR, Rubin BP, Billings SD, Ko JS. Superficial ALK-rearranged myxoid spindle cell neoplasm: a cutaneous soft tissue tumor with distinctive morphology and immunophenotypic profile. Mod Pathol. 2021;34(9):1710–1718. doi: 10.1038/s41379-021-00830-w. [DOI] [PubMed] [Google Scholar]
- Deshpande N, Needles A, Willmann JK. Molecular ultrasound imaging: current status and future directions. Clin Radiol. 2010;65(7):567–581. doi: 10.1016/j.crad.2010.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drezek RA, Richards-Kortum R, Brewer MA, Feld MS, Pitris C, Ferenczy A, Faupel ML, Follen M. Optical imaging of the cervix. Cancer. 2003;98(9 Suppl):2015–2027. doi: 10.1002/cncr.11678. [DOI] [PubMed] [Google Scholar]
- El-Koussy M, Schroth G, Brekenfeld C, Arnold M. Imaging of acute ischemic stroke. Eur Neurol. 2014;72(5–6):309–316. doi: 10.1159/000362719. [DOI] [PubMed] [Google Scholar]
- Friedman D, Fahlstrom R. Racial and ethnic differences in epilepsy classification among probands in the epilepsy phenome/genome project (EPGP) Epilepsy Res. 2013;107(3):306–310. doi: 10.1016/j.eplepsyres.2013.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furukawa A, Saotome T, Yamasaki M, Maeda K, Nitta N, Takahashi M, Tsujikawa T, Fujiyama Y, Murata K, Sakamoto T. Cross-sectional imaging in Crohn disease. Radiographics. 2004;24(3):689–702. doi: 10.1148/rg.243035120. [DOI] [PubMed] [Google Scholar]
- Gerrits EG, Lutgers HL, Kleefstra N, Graaff R, Groenier KH, Smit AJ, Gans RO, Bilo HJ. Skin autofluorescence: a tool to identify type 2 diabetic patients at risk for developing microvascular complications. Diabetes Care. 2008;31(3):517–521. doi: 10.2337/dc07-1755. [DOI] [PubMed] [Google Scholar]
- Gohil SH, Iorgulescu JB, Braun DA, Keskin DB, Livak KJ. Applying high-dimensional single-cell technologies to the analysis of cancer immunotherapy. Nat Rev Clin Oncol. 2021;18(4):244–256. doi: 10.1038/s41571-020-00449-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González RG. Clinical MRI of acute ischemic stroke. J Magn Reson Imaging. 2012;36(2):259–271. doi: 10.1002/jmri.23595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorcenco S, Ilinca A, Almasoudi W, Kafantari E, Lindgren AG, Puschmann A. New generation genetic testing entering the clinic. Parkinsonism Relat Disord. 2020;73:72–84. doi: 10.1016/j.parkreldis.2020.02.015. [DOI] [PubMed] [Google Scholar]
- Hamilton J, Franson D, Seiberlich N. Recent advances in parallel imaging for MRI. Prog Nucl Magn Reson Spectrosc. 2017;101:71–95. doi: 10.1016/j.pnmrs.2017.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han Y, Li L, Zhang Y, Yuan H, Ye L, Zhao J, Duan DD. Phenomics of vascular disease: the systematic approach to the combination therapy. Curr Vasc Pharmacol. 2015;13(4):433–440. doi: 10.2174/1570161112666141014144829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasin Y, Seldin M, Lusis A. Multi-omics approaches to disease. Genome Biol. 2017;18(1):83. doi: 10.1186/s13059-017-1215-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellmann MD, Nathanson T, Rizvi H, Creelan BC, Sanchez-Vega F, Ahuja A, Ni A, Novik JB, Mangarin LMB, Abu-Akeel M, Liu C, Sauter JL, Rekhtman N, Chang E, Callahan MK, Chaft JE, Voss MH, Tenet M, Li X-M, Covello K, Renninger A, Vitazka P, Geese WJ, Borghaei H, Rudin CM, Antonia SJ, Swanton C, Hammerbacher J, Merghoub T, McGranahan N, Snyder A, Wolchok JD. Genomic features of response to combination immunotherapy in patients with advanced non-small-cell lung cancer. Cancer Cell. 2018;33(5):843–852.e844. doi: 10.1016/j.ccell.2018.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hennekam RC, Biesecker LG. Next-generation sequencing demands next-generation phenotyping. Hum Mutat. 2012;33(5):884–886. doi: 10.1002/humu.22048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heyn H, Esteller M. DNA methylation profiling in the clinic: applications and challenges. Nat Rev Genet. 2012;13(10):679. doi: 10.1038/nrg3270. [DOI] [PubMed] [Google Scholar]
- Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL, Bohr VA. Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol. 2019;15(10):565–581. doi: 10.1038/s41582-019-0244-7. [DOI] [PubMed] [Google Scholar]
- Houle D, Govindaraju DR, Omholt S. Phenomics: the next challenge. Nat Rev Genet. 2010;11(12):855–866. doi: 10.1038/nrg2897. [DOI] [PubMed] [Google Scholar]
- Hricak H, Abdel-Wahab M, Atun R, Lette MM, Paez D, Brink JA, Donoso-Bach L, Frija G, Hierath M, Holmberg O, Khong PL, Lewis JS, McGinty G, Oyen WJG, Shulman LN, Ward ZJ, Scott AM. Medical imaging and nuclear medicine: a Lancet Oncology Commission. Lancet Oncol. 2021;22(4):e136–e172. doi: 10.1016/s1470-2045(20)30751-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang HM, Shih YY. Pushing CT and MR imaging to the molecular level for studying the "omics": current challenges and advancements. Biomed Res Int. 2014;2014:365812. doi: 10.1155/2014/365812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishiwata T, Gregor A, Inage T, Yasufuku K. Bronchoscopic navigation and tissue diagnosis. Gen Thorac Cardiovasc Surg. 2020;68(7):672–678. doi: 10.1007/s11748-019-01241-0. [DOI] [PubMed] [Google Scholar]
- Jaeger S, Candemir S, Antani S, Wáng Y, Thoma G. Two public chest X-ray datasets for computer-aided screening of pulmonary diseases. Quant Imaging Med Surg. 2014;4(6):475–477. doi: 10.3978/j.issn.2223-4292.2014.11.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen JA. Medical ultrasound imaging. Prog Biophys Mol Biol. 2007;93(1):153–165. doi: 10.1016/j.pbiomolbio.2006.07.025. [DOI] [PubMed] [Google Scholar]
- Jin L. Welcome to the phenomics journal. Phenomics. 2021;1(1):1–2. doi: 10.1007/s43657-020-00009-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin C, Luo X, Li X, Zhou R, Zhong Y, Xu Z, Cui C, Xing X, Zhang H, Tian M. Positron emission tomography molecular imaging-based cancer phenotyping. Cancer. 2022;128(14):2704–2716. doi: 10.1002/cncr.34228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones GS, Baldwin DR. Recent advances in the management of lung cancer. Clin Med (Lond) 2018;18(Suppl 2):s41–s46. doi: 10.7861/clinmedicine.18-2-s41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasırga E. The importance of stool tests in diagnosis and follow-up of gastrointestinal disorders in children. Turk Pediatri Ars. 2019;54(3):141–148. doi: 10.14744/TurkPediatriArs.2018.00483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaur H, Prakash A, Medhi B. Drug therapy in stroke: from preclinical to clinical studies. Pharmacology. 2013;92(5–6):324–334. doi: 10.1159/000356320. [DOI] [PubMed] [Google Scholar]
- Khatib R, Arevalo YA, Berendsen MA, Prabhakaran S, Huffman MD. Presentation, evaluation, management, and outcomes of acute stroke in low- and middle-income countries: a systematic review and meta-analysis. Neuroepidemiology. 2018;51(1–2):104–112. doi: 10.1159/000491442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koenig F, McGovern FJ, Althausen AF, Deutsch TF, Schomacker KT. Laser induced autofluorescence diagnosis of bladder cancer. J Urol. 1996;156(5):1597–1601. doi: 10.1016/S0022-5347(01)65456-9. [DOI] [PubMed] [Google Scholar]
- Koenig M, Joyal F, Fritzler MJ, Roussin A, Abrahamowicz M, Boire G, Goulet JR, Rich E, Grodzicky T, Raymond Y, Senécal JL. Autoantibodies and microvascular damage are independent predictive factors for the progression of Raynaud's phenomenon to systemic sclerosis: a twenty-year prospective study of 586 patients, with validation of proposed criteria for early systemic sclerosis. Arthritis Rheum. 2008;58(12):3902–3912. doi: 10.1002/art.24038. [DOI] [PubMed] [Google Scholar]
- Lage K, Karlberg EO, Størling ZM, Olason PI, Pedersen AG, Rigina O, Hinsby AM, Tümer Z, Pociot F, Tommerup N, Moreau Y, Brunak S. A human phenome-interactome network of protein complexes implicated in genetic disorders. Nat Biotechnol. 2007;25(3):309–316. doi: 10.1038/nbt1295. [DOI] [PubMed] [Google Scholar]
- Leithner D, Horvat JV, Ochoa-Albiztegui RE, Thakur S, Wengert G, Morris EA, Helbich TH, Pinker K. Imaging and the completion of the omics paradigm in breast cancer. Radiologe. 2018;58(Suppl 1):7–13. doi: 10.1007/s00117-018-0409-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leon-Mimila P, Wang J, Huertas-Vazquez A. Relevance of multi-omics studies in cardiovascular diseases. Front Cardiovasc Med. 2019;6:91. doi: 10.3389/fcvm.2019.00091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Ma Y, Wang K, Zhang M, Wang Y, Liu X, Hao M, Yin X, Liang M, Zhang H, Wang X, Chen X, Zhang Y, Duan W, Kang L, Qiao B, Wang J, Jin L. Using composite phenotypes to reveal hidden physiological heterogeneity in high-altitude acclimatization in a Chinese Han Longitudinal Cohort. Phenomics. 2021;1(1):3–14. doi: 10.1007/s43657-020-00005-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Jiang X, Wang Y, Kim Y. Applied machine learning in Alzheimer's disease research: omics, imaging, and clinical data. Emerg Top Life Sci. 2021;5(6):765–777. doi: 10.1042/etls20210249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Glover JD, Zhang H, Peng M, Tan J, Mallick CB, Hou D, Yang Y, Wu S, Liu Y, Peng Q, Zheng SC, Crosse EI, Medvinsky A, Anderson RA, Brown H, Yuan Z, Zhou S, Xu Y, Kemp JP, Ho YYW, Loesch DZ, Wang L, Li Y, Tang S, Wu X, Walters RG, Lin K, Meng R, Lv J, Chernus JM, Neiswanger K, Feingold E, Evans DM, Medland SE, Martin NG, Weinberg SM, Marazita ML, Chen G, Chen Z, Zhou Y, Cheeseman M, Wang L, Jin L, Headon DJ, Wang S. Limb development genes underlie variation in human fingerprint patterns. Cell. 2022;185(1):95–112.e118. doi: 10.1016/j.cell.2021.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lianidou ES, Markou A, Strati A. The role of CTCs as tumor biomarkers. Adv Exp Med Biol. 2015;867:341–367. doi: 10.1007/978-94-017-7215-0_21. [DOI] [PubMed] [Google Scholar]
- Liu Z, Suo C, Jiang Y, Zhao R, Zhang T, Jin L, Chen X. Phenome-wide association analysis reveals novel links between genetically determined levels of liver enzymes and disease phenotypes. Phenomics. 2022 doi: 10.1007/s43657-021-00033-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Y, Li Z, Zhang X, Ming B, Jia J, Wang R, Ma D. Retinal nerve fiber layer structure abnormalities in early Alzheimer's disease: evidence in optical coherence tomography. Neurosci Lett. 2010;480(1):69–72. doi: 10.1016/j.neulet.2010.06.006. [DOI] [PubMed] [Google Scholar]
- Ma Q, Li R, Wang L, Yin P, Wang Y, Yan C, Ren Y, Qian Z, Vaughn MG, McMillin SE, Hay SI, Naghavi M, Cai M, Wang C, Zhang Z, Zhou M, Lin H, Yang Y. Temporal trend and attributable risk factors of stroke burden in China, 1990–2019: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. 2021;6(12):e897–e906. doi: 10.1016/s2468-2667(21)00228-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malik P, Anwar A, Patel R, Patel U. Expansion of the dimensions in the current management of acute ischemic stroke. J Neurol. 2021;268(9):3185–3202. doi: 10.1007/s00415-020-09873-6. [DOI] [PubMed] [Google Scholar]
- Marko M, Posekany A, Szabo S, Scharer S, Kiechl S, Knoflach M, Serles W, Ferrari J, Lang W, Sommer P, Greisenegger S. Trends of r-tPA (recombinant tissue-type plasminogen activator) treatment and treatment-influencing factors in acute ischemic stroke. Stroke. 2020;51(4):1240–1247. doi: 10.1161/strokeaha.119.027921. [DOI] [PubMed] [Google Scholar]
- Masilamani V, Al-Zhrani K, Al-Salhi M, Al-Diab A, Al-Ageily M. Cancer diagnosis by autofluorescence of blood components. J Lumin. 2004;109(3–4):143–154. doi: 10.1016/j.jlumin.2004.02.001. [DOI] [Google Scholar]
- Matur AV, Candelario-Jalil E, Paul S, Karamyan VT, Lee JD, Pennypacker K, Fraser JF. Translating animal models of ischemic stroke to the human condition. Transl Stroke Res. 2022 doi: 10.1007/s12975-022-01082-9. [DOI] [PubMed] [Google Scholar]
- Michel CM, Murray MM. Towards the utilization of EEG as a brain imaging tool. Neuroimage. 2012;61(2):371–385. doi: 10.1016/j.neuroimage.2011.12.039. [DOI] [PubMed] [Google Scholar]
- Mishriki SF, Aboumarzouk O, Vint R, Grimsley SJ, Lam T, Somani B. Routine urine cytology has no role in hematuria investigations. J Urol. 2013;189(4):1255–1259. doi: 10.1016/j.juro.2012.10.022. [DOI] [PubMed] [Google Scholar]
- Mkrtchyan GV, Abdelmohsen K, Andreux P, Bagdonaite I, Barzilai N, Brunak S, Cabreiro F, de Cabo R, Campisi J, Cuervo AM, Demaria M, Ewald CY, Fang EF, Faragher R, Ferrucci L, Freund A, Silva-García CG, Georgievskaya A, Gladyshev VN, Glass DJ, Gorbunova V, de Grey A, He WW, Hoeijmakers J, Hoffmann E, Horvath S, Houtkooper RH, Jensen MK, Jensen MB, Kane A, Kassem M, de Keizer P, Kennedy B, Karsenty G, Lamming DW, Lee KF, MacAulay N, Mamoshina P, Mellon J, Molenaars M, Moskalev A, Mund A, Niedernhofer L, Osborne B, Pak HH, Parkhitko A, Raimundo N, Rando TA, Rasmussen LJ, Reis C, Riedel CG, Franco-Romero A, Schumacher B, Sinclair DA, Suh Y, Taub PR, Toiber D, Treebak JT, Valenzano DR, Verdin E, Vijg J, Young S, Zhang L, Bakula D, Zhavoronkov A, Scheibye-Knudsen M. ARDD 2020: from aging mechanisms to interventions. Aging (Albany NY) 2020;12(24):24484–24503. doi: 10.18632/aging.202454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monneret G, Gossez M, Aghaeepour N, Gaudilliere B, Venet F. How clinical flow cytometry rebooted sepsis immunology. Cytometry A. 2019;95(4):431–441. doi: 10.1002/cyto.a.23749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moravej Aleali A, Amani R, Shahbazian H, Namjooyan F, Latifi SM, Cheraghian B. The effect of hydroalcoholic Saffron (Crocus sativus L.) extract on fasting plasma glucose, HbA1c, lipid profile, liver, and renal function tests in patients with type 2 diabetes mellitus: a randomized double-blind clinical trial. Phytother Res. 2019;33(6):1648–1657. doi: 10.1002/ptr.6351. [DOI] [PubMed] [Google Scholar]
- Muccilli A, Seyman E, Oh J. Spinal cord MRI in multiple sclerosis. Neurol Clin. 2018;36(1):35–57. doi: 10.1016/j.ncl.2017.08.009. [DOI] [PubMed] [Google Scholar]
- Mulder DJ, Water TV, Lutgers HL, Graaff R, Gans RO, Zijlstra F, Smit AJ. Skin autofluorescence, a novel marker for glycemic and oxidative stress-derived advanced glycation endproducts: an overview of current clinical studies, evidence, and limitations. Diabetes Technol Ther. 2006;8(5):523–535. doi: 10.1089/dia.2006.8.523. [DOI] [PubMed] [Google Scholar]
- Newgard CB, Attie AD. Getting biological about the genetics of diabetes. Nat Med. 2010;16(4):388–391. doi: 10.1038/nm0410-388. [DOI] [PubMed] [Google Scholar]
- Nicholson JK. Molecular phenomic approaches to deconvolving the systemic effects of SARS-CoV-2 infection and post-acute COVID-19 syndrome. Phenomics. 2021;1(4):143–150. doi: 10.1007/s43657-021-00020-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikpay M, Mohammadzadeh S. Phenome-wide screening for traits causally associated with the risk of coronary artery disease. J Hum Genet. 2020;65(4):371–380. doi: 10.1038/s10038-019-0716-z. [DOI] [PubMed] [Google Scholar]
- Nordström A, Lewensohn R. Metabolomics: moving to the clinic. J Neuroimmune Pharmacol. 2010;5(1):4–17. doi: 10.1007/s11481-009-9156-4. [DOI] [PubMed] [Google Scholar]
- North BJ, Sinclair DA. The intersection between aging and cardiovascular disease. Circ Res. 2012;110(8):1097–1108. doi: 10.1161/circresaha.111.246876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olivier M, Asmis R, Hawkins GA, Howard TD, Cox LA. The need for multi-omics biomarker signatures in precision medicine. Int J Mol Sci. 2019 doi: 10.3390/ijms20194781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panunzio A, Tafuri A, Princiotta A, Gentile I, Mazzucato G, Trabacchin N, Antonelli A, Cerruto MA. Omics in urology: an overview on concepts, current status and future perspectives. Urologia. 2021;88(4):270–279. doi: 10.1177/03915603211022960. [DOI] [PubMed] [Google Scholar]
- Partridge L, Deelen J, Slagboom PE. Facing up to the global challenges of ageing. Nature. 2018;561(7721):45–56. doi: 10.1038/s41586-018-0457-8. [DOI] [PubMed] [Google Scholar]
- Patton N, Aslam T, Macgillivray T, Pattie A, Deary IJ, Dhillon B. Retinal vascular image analysis as a potential screening tool for cerebrovascular disease: a rationale based on homology between cerebral and retinal microvasculatures. J Anat. 2005;206(4):319–348. doi: 10.1111/j.1469-7580.2005.00395.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pattou F, Huglo D, Proye C. Radionuclide scanning in parathyroid diseases. Br J Surg. 1998;85(12):1605–1616. doi: 10.1046/j.1365-2168.1998.00942.x. [DOI] [PubMed] [Google Scholar]
- Peña ID, Borlongan C, Shen G, Davis W. Strategies to extend thrombolytic time window for ischemic stroke treatment: an unmet clinical need. J Stroke. 2017;19(1):50–60. doi: 10.5853/jos.2016.01515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pendergrass SA, Ritchie MD. Phenome-wide association studies: leveraging comprehensive phenotypic and genotypic data for discovery. Curr Genet Med Rep. 2015;3(2):92–100. doi: 10.1007/s40142-015-0067-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Price VH, Odom RB, Ward WH, Jones FT. Trichothiodystrophy: sulfur-deficient brittle hair as a marker for a neuroectodermal symptom complex. Arch Dermatol. 1980;116(12):1375–1384. doi: 10.1001/archderm.116.12.1375. [DOI] [PubMed] [Google Scholar]
- Qayyum MA, Shah MH. Comparative study of trace elements in blood, scalp hair and nails of prostate cancer patients in relation to healthy donors. Biol Trace Elem Res. 2014;162(1–3):46–57. doi: 10.1007/s12011-014-0123-4. [DOI] [PubMed] [Google Scholar]
- Rahman J, Rahman S. The utility of phenomics in diagnosis of inherited metabolic disorders. Clin Med (Lond) 2019;19(1):30–36. doi: 10.7861/clinmedicine.19-1-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson PN. Deep phenotyping for precision medicine. Hum Mutat. 2012;33(5):777–780. doi: 10.1002/humu.22080. [DOI] [PubMed] [Google Scholar]
- Roden DM. Phenome-wide association studies: a new method for functional genomics in humans. J Physiol. 2017;595(12):4109–4115. doi: 10.1113/jp273122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudy Y, Burnes JE. Noninvasive electrocardiographic imaging. Ann Noninvasive Electrocardiol. 1999;4(3):340–359. doi: 10.1111/j.1542-474X.1999.tb00220.x. [DOI] [Google Scholar]
- Russell E, Koren G, Rieder M, Van Uum S. Hair cortisol as a biological marker of chronic stress: current status, future directions and unanswered questions. Psychoneuroendocrinology. 2012;37(5):589–601. doi: 10.1016/j.psyneuen.2011.09.009. [DOI] [PubMed] [Google Scholar]
- Shain C, Ramgopal S, Fallil Z, Parulkar I, Alongi R, Knowlton R, Poduri A. Polymicrogyria-associated epilepsy: a multicenter phenotypic study from the epilepsy phenome/genome project. Epilepsia. 2013;54(8):1368–1375. doi: 10.1111/epi.12238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheth D, Giger ML. Artificial intelligence in the interpretation of breast cancer on MRI. J Magn Reson Imaging. 2020;51(5):1310–1324. doi: 10.1002/jmri.26878. [DOI] [PubMed] [Google Scholar]
- Shi F, Wang J, Shi J, Wu Z, Wang Q, Tang Z, He K, Shi Y, Shen D. Review of artificial intelligence techniques in imaging data acquisition, segmentation, and diagnosis for COVID-19. IEEE Rev Biomed Eng. 2021;14:4–15. doi: 10.1109/rbme.2020.2987975. [DOI] [PubMed] [Google Scholar]
- Sitek A, Ahn S, Asma E, Chandler A, Ihsani A, Prevrhal S, Rahmim A, Saboury B, Thielemans K. Artificial intelligence in PET: an industry perspective. PET Clin. 2021;16(4):483–492. doi: 10.1016/j.cpet.2021.06.006. [DOI] [PubMed] [Google Scholar]
- Sparrow JM, Bron AJ, Brown NA, Neil HA. Autofluorescence of the crystalline lens in early and late onset diabetes. Br J Ophthalmol. 1992;76(1):25–31. doi: 10.1136/bjo.76.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Surti S. Update on time-of-flight PET imaging. J Nucl Med. 2015;56(1):98–105. doi: 10.2967/jnumed.114.145029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tajiri H, Matsuda K, Fujisaki J. What can we see with the endoscope? Present status and future perspectives. Dig Endosc. 2010;14(4):131–137. doi: 10.1046/j.0915-5635.2002.00191.x. [DOI] [Google Scholar]
- Tankisi H, Burke D, Cui L, de Carvalho M, Kuwabara S, Nandedkar SD, Rutkove S, Stålberg E, van Putten M, Fuglsang-Frederiksen A. Standards of instrumentation of EMG. Clin Neurophysiol. 2020;131(1):243–258. doi: 10.1016/j.clinph.2019.07.025. [DOI] [PubMed] [Google Scholar]
- Thorp JA, Rushing RS. Umbilical cord blood gas analysis. Obstet Gynecol Clin North Am. 1999;26(4):695–709. doi: 10.1016/s0889-8545(05)70107-8. [DOI] [PubMed] [Google Scholar]
- Tian M, Zhang H, Oriuchi N, Higuchi T, Endo K. Comparison of 11C-choline PET and FDG PET for the differential diagnosis of malignant tumors. Eur J Nucl Med Mol Imaging. 2004;31(8):1064–1072. doi: 10.1007/s00259-004-1496-y. [DOI] [PubMed] [Google Scholar]
- Tian M, He X, Jin C, He X, Wu S, Zhou R, Zhang X, Zhang K, Gu W, Wang J, Zhang H. Transpathology: molecular imaging-based pathology. Eur J Nucl Med Mol Imaging. 2021;48(8):2338–2350. doi: 10.1007/s00259-021-05234-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tu WJ, Zeng X, Liu Q. Aging tsunami coming: the main finding from China's seventh national population census. Aging Clin Exp Res. 2022;34(5):1159–1163. doi: 10.1007/s40520-021-02017-4. [DOI] [PubMed] [Google Scholar]
- Ueland T, Gullestad L, Nymo SH, Yndestad A, Aukrust P, Askevold ET. Inflammatory cytokines as biomarkers in heart failure. Clin Chim Acta. 2015;443:71–77. doi: 10.1016/j.cca.2014.09.001. [DOI] [PubMed] [Google Scholar]
- van den Hoogen F, Khanna D, Fransen J, Johnson SR, Baron M, Tyndall A, Matucci-Cerinic M, Naden RP, Medsger TA, Jr, Carreira PE, Riemekasten G, Clements PJ, Denton CP, Distler O, Allanore Y, Furst DE, Gabrielli A, Mayes MD, van Laar JM, Seibold JR, Czirjak L, Steen VD, Inanc M, Kowal-Bielecka O, Müller-Ladner U, Valentini G, Veale DJ, Vonk MC, Walker UA, Chung L, Collier DH, Csuka ME, Fessler BJ, Guiducci S, Herrick A, Hsu VM, Jimenez S, Kahaleh B, Merkel PA, Sierakowski S, Silver RM, Simms RW, Varga J, Pope JE. 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 2013;65(11):2737–2747. doi: 10.1002/art.38098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viganò P, Somigliana E, Panina P, Rabellotti E, Vercellini P, Candiani M. Principles of phenomics in endometriosis. Hum Reprod Update. 2012;18(3):248–259. doi: 10.1093/humupd/dms001. [DOI] [PubMed] [Google Scholar]
- Vilela P, Rowley HA. Brain ischemia: CT and MRI techniques in acute ischemic stroke. Eur J Radiol. 2017;96:162–172. doi: 10.1016/j.ejrad.2017.08.014. [DOI] [PubMed] [Google Scholar]
- Wang YJ, Li ZX, Gu HQ, Zhai Y, Zhou Q, Jiang Y, Zhao XQ, Wang YL, Yang X, Wang CJ, Meng X, Li H, Liu LP, Jing J, Wu J, Xu AD, Dong Q, Wang D, Wang WZ, Ma XD, Zhao JZ. China Stroke Statistics: an update on the 2019 report from the National Center for Healthcare Quality Management in Neurological Diseases, China National Clinical Research Center for Neurological Diseases, the Chinese Stroke Association, National Center for Chronic and Non-communicable Disease Control and Prevention, Chinese Center for Disease Control and Prevention and Institute for Global Neuroscience and Stroke Collaboration. Stroke Vasc Neurol. 2022 doi: 10.1136/svn-2021-001374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Went M, Cornish AJ, Law PJ, Kinnersley B, van Duin M, Weinhold N, Försti A, Hansson M, Sonneveld P, Goldschmidt H, Morgan GJ, Hemminki K, Nilsson B, Kaiser M, Houlston RS. Search for multiple myeloma risk factors using Mendelian randomization. Blood Adv. 2020;4(10):2172–2179. doi: 10.1182/bloodadvances.2020001502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wintermark M, Sanelli PC, Albers GW, Bello J, Derdeyn C, Hetts SW, Johnson MH, Kidwell C, Lev MH, Liebeskind DS, Rowley H, Schaefer PW, Sunshine JL, Zaharchuk G, Meltzer CC. Imaging recommendations for acute stroke and transient ischemic attack patients: a joint statement by the American Society of Neuroradiology, the American College of Radiology, and the Society of NeuroInterventional Surgery. AJNR Am J Neuroradiol. 2013;34(11):E117–127. doi: 10.3174/ajnr.A3690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wollenweber SD, Alessio AM, Kinahan PE. A phantom design for assessment of detectability in PET imaging. Med Phys. 2016;43(9):5051. doi: 10.1118/1.4960365. [DOI] [PubMed] [Google Scholar]
- Wu S, Powers S, Zhu W, Hannun YA. Substantial contribution of extrinsic risk factors to cancer development. Nature. 2016;529(7584):43–47. doi: 10.1038/nature16166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu D, Tao Y, Zhang M, Li Y, Shen L, Li Y, Ying W. Selectively increased autofluorescence at fingernails and certain regions of skin: a potential novel diagnostic biomarker for Parkinson’s disease. bioRxiv. 2018 doi: 10.1101/322222. [DOI] [Google Scholar]
- Wu D, Zhang M, Tao Y, Li Y, Zhang S, Chen X, Ying W. Distinct ‘pattern of autofluorescence’ of acute ischemic stroke patients’ skin and fingernails: a novel diagnostic biomarker for acute ischemic stroke. bioRxiv. 2018 doi: 10.1101/310904. [DOI] [Google Scholar]
- Xu Z, Shen B, Tang Y, Wu J, Wang J. Deep clinical phenotyping of Parkinson’s Disease: towards a new era of research and clinical care. Phenomics. 2022 doi: 10.1007/s43657-022-00051-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada J, Stevens B, de Silva N, Gibbins S, Beyene J, Taddio A, Newman C, Koren G. Hair cortisol as a potential biologic marker of chronic stress in hospitalized neonates. Neonatology. 2007;92(1):42–49. doi: 10.1159/000100085. [DOI] [PubMed] [Google Scholar]
- Yang Y, Xu L, Feng Z, Cruz JA, Savage LJ, Kramer DM, Chen J. PhenoCurve: capturing dynamic phenotype-environment relationships using phenomics data. Bioinformatics. 2017;33(9):1370–1378. doi: 10.1093/bioinformatics/btw673. [DOI] [PubMed] [Google Scholar]
- Yang H, Pawitan Y, Fang F, Czene K, Ye W. Biomarkers and disease trajectories influencing women's health: Results from the UK Biobank Cohort. Phenomics. 2022;2(3):184–193. doi: 10.1007/s43657-022-00054-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- You J, Zhang Y-R, Wang H-F, Yang M, Feng J-F, Yu J-T, Cheng W. Development of a novel dementia risk prediction model in the general population: a large, longitudinal, population-based machine-learning study. eClinicalMedicine. 2022;53:101665. doi: 10.1016/j.eclinm.2022.101665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeman RK, Silverman PM, Vieco PT, Costello P. CT angiography. Am J Roentgenol. 1995;165(5):1079–1088. doi: 10.2214/ajr.165.5.7572481. [DOI] [PubMed] [Google Scholar]
- Zhang M, Ying W. UV-induced skin's green autofluorescence is a biomarker for both non-invasive evaluations of the dosages of UV exposures of the skin and non-invasive prediction of UV-induced skin damage. Photochem Photobiol Sci. 2022 doi: 10.1007/s43630-022-00306-z. [DOI] [PubMed] [Google Scholar]
- Zhang M, Li Y, Wang J, Tang H, Yang Z, Wu D, Tao Y, He H, Wang S, Chen X, Yin S, Shi H, Wei X, Chu T, Tang W, Maharjan DT, Yang Z, Wang Y, Jin L, Ying W. Keratin-based epidermal green autofluorescence is a common biomarker of organ injury. bioRxiv. 2020 doi: 10.1101/564112. [DOI] [Google Scholar]
- Zhang H, Hua X, Song J. Phenotypes of cardiovascular diseases: current status and future perspectives. Phenomics. 2021;1(5):229–241. doi: 10.1007/s43657-021-00022-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang M, Tao Y, Chang Q, Wang K, Chu T, Ying W. Skin's green autofluorescence at dorsal centremetacarpus may become a novel biomarker for diagnosis of lung cancer. J Biophotonics. 2022;15(5):e202100389. doi: 10.1002/jbio.202100389. [DOI] [PubMed] [Google Scholar]
- Zhang M, Tao Y, Yu H, Wu D, Liao B, Qiu JG, Jiang BH, Ying W. Green autofluorescence of the index fingernails is a novel biomarker for noninvasive determinations on the status of tobacco smoking. J Biophotonics. 2022 doi: 10.1002/jbio.202200195. [DOI] [PubMed] [Google Scholar]
- Zhang M, Wu S, Du S, Qian W, Chen J, Qiao L, Yang Y, Tan J, Yuan Z, Peng Q, Liu Y, Navarro N, Tang K, Ruiz-Linares A, Wang J, Claes P, Jin L, Li J, Wang S. Genetic variants underlying differences in facial morphology in East Asian and European populations. Nat Genet. 2022;54(4):403–411. doi: 10.1038/s41588-022-01038-7. [DOI] [PubMed] [Google Scholar]
- Zhang M, Zhang J, Teng H, Zhang Q, Xiang Z, Chang Q, Tao Y, Chu T, Ying W. Decreased green autofluorescence of lung parenchyma is a biomarker for lung cancer tissues. J Biophotonics. 2022;15(8):e202200072. doi: 10.1002/jbio.202200072. [DOI] [PubMed] [Google Scholar]
- Zhang X, Jiang H, Wu S, Wang J, Zhou R, He X, Qian S, Zhao S, Zhang H, Civelek AC, Tian M. Positron emission tomography molecular imaging for phenotyping and management of lymphoma. Phenomics. 2022;2(2):102–118. doi: 10.1007/s43657-021-00042-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng T, Xie W, Xu L, He X, Zhang Y, You M, Yang G, Chen Y. A machine learning-based framework to identify type 2 diabetes through electronic health records. Int J Med Inform. 2017;97:120–127. doi: 10.1016/j.ijmedinf.2016.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zivin JA. Acute stroke therapy with tissue plasminogen activator (tPA) since it was approved by the U.S. Food and Drug Administration (FDA) Ann Neurol. 2009;66(1):6–10. doi: 10.1002/ana.21750. [DOI] [PubMed] [Google Scholar]
- Zondervan KT, Rahmioglu N, Morris AP, Nyholt DR, Montgomery GW, Becker CM, Missmer SA. Beyond endometriosis genome-wide association study: from genomics to phenomics to the patient. Semin Reprod Med. 2016;34(4):242–254. doi: 10.1055/s-0036-1585408. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.
Not applicable.