ABSTRACT
Despite an important link between the gut and ocular health, the role of the gut-eye axis remains elusive in ocular infections. In this study, we investigated the role of butyrate, a gut microbial metabolite, in the pathobiology of intraocular bacterial (Staphylococcus aureus) infection, endophthalmitis. We found that intravitreal administration of butyrate derivatives, sodium butyrate (NaB), or phenylbutyrate (PBA) reduced intraocular bacterial growth and retinal inflammatory response. The ocular tissue architecture and retinal function were preserved in butyrate-treated eyes. In cultured mouse bone marrow-derived macrophages (BMDMs) and human retinal Müller glia, NaB or PBA treatment reduced S. aureus-induced inflammatory response by inhibiting NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome. However, in vivo data showed NLRP3-independent effects of butyrate. The butyrate-treated mouse retina and cells exhibited induced expression of antimicrobial molecules CRAMP (LL37) and S100A7/A8, resulting in increased bacterial phagocytosis and killing. Moreover, butyrate treatment enhanced AMP-activated protein kinase (AMPK)-dependent autophagy and promoted the co-localization of CRAMP in autophagosomes, indicating autophagy-mediated bacterial killing. Furthermore, pharmacological inhibition of autophagy in mice revealed its role in butyrate-mediated protection. Finally, butyrate exhibited synergy with antibiotic in promoting endophthalmitis resolution. Collectively, our study demonstrated the protective mechanisms of butyrate in ameliorating bacterial endophthalmitis. Therefore, butyrate derivatives could be explored as immunomodulatory and anti-bacterial therapeutics to improve visual outcomes in ocular bacterial infections.
KEYWORDS: eye, endophthalmitis, Staphylococcus aureus, butyrate, inflammation, autophagy, AMPK, innate immunity, retina
INTRODUCTION
Short-chain fatty acids (SCFAs) such as butyrate are metabolites produced by anaerobic bacterial fermentation of undigested or partially digested dietary fibers in the colonic lumen (1). Many SCFAs, including butyrate, promote intestinal epithelial barrier function and regulate the host mucosal immune system (2). Butyrate has been shown to play a beneficial role in the regulation of inflammation (3–5), adaptive immunity (6–8), and bacterial infection (9, 10). Butyrate also promotes the generation of regulatory T cells directly in the gut by inhibiting histone deacetylase activity at the Foxp3 gene and indirectly by promoting anti-inflammatory properties in macrophages and dendritic cells (11, 12). Butyrate influences the disease pathology in patients with inflammatory bowel disease by modulating T regulatory cells (13). Furthermore, exposure of peripheral blood mononuclear cells such as neutrophils and macrophages to SCFAs inhibits inflammatory cytokine production (14). Recently, in rheumatoid arthritis patients and arthritic mice, butyrate supplementation was shown to suppress arthritis by modulating B regulatory functions (15). Butyrate promotes cellular metabolism and the memory potential of activated CD8+ T cells and is required for optimal recall responses upon antigen reencounter (16). Besides, recent studies on neurological diseases have shown the novel gut-brain axis in the disease pathology, suggesting the influence of gut metabolites on distant organs (17, 18). Overall, recent literature indicates that SCFAs induce metabolic and transcriptional changes in immune cells, influencing health and disease outcomes. Although some recent studies have demonstrated the protective role of SCFAs in experimental noninfectious eye diseases (19–21), to our knowledge, their effects have not been investigated in ocular infections.
One blinding ocular infection is bacterial endophthalmitis characterized by severe intraocular inflammation (22–25). Most cases of bacterial endophthalmitis are caused by Staphylococci, notably, Staphylococcus aureus, which is responsible for less than 15% of cases but has poor disease outcomes (26). The bacteria primarily enter the eye during ocular surgeries and releases several toxins and cell wall components, e.g., peptidoglycan and lipoteichoic acids (27). Host residential immune cells detect these bacterial components through Toll-like receptor 2, and a cascade of inflammatory events ensues, leading to the recruitment of innate immune cells (24, 28). The infiltration of immune cells results in aberrant inflammation and, if it remains unchecked, destroys the retina, resulting in permanent vision loss (27). There is currently no universal therapeutic regimen available which can prevent uncontrolled inflammation and vision loss. In addition, the emergence of antibiotic resistance among ocular pathogens poses a significant threat to the management of ocular infections (29). Therefore, there is an urgent need to identify novel compounds with immunomodulatory and antimicrobial potential to prevent and treat bacterial endophthalmitis.
In our search for new immunomodulatory agents to treat bacterial endophthalmitis, we recently used a global metabolomics approach to understand the complex interplay of host-pathogen interaction. This innovative metabolomics approach led to the identification of several immunoregulatory molecules and pathways associated with S. aureus endophthalmitis (30). Our metabolomics data revealed that among the SCFAs, butyrate is one of the highly downregulated metabolites in S. aureus-induced endophthalmitis. In the current study, we investigated whether butyrate can be used to treat S. aureus endophthalmitis and determine the underlying protective mechanisms.
RESULTS
Butyrate treatment protects the eye and ameliorates bacterial endophthalmitis.
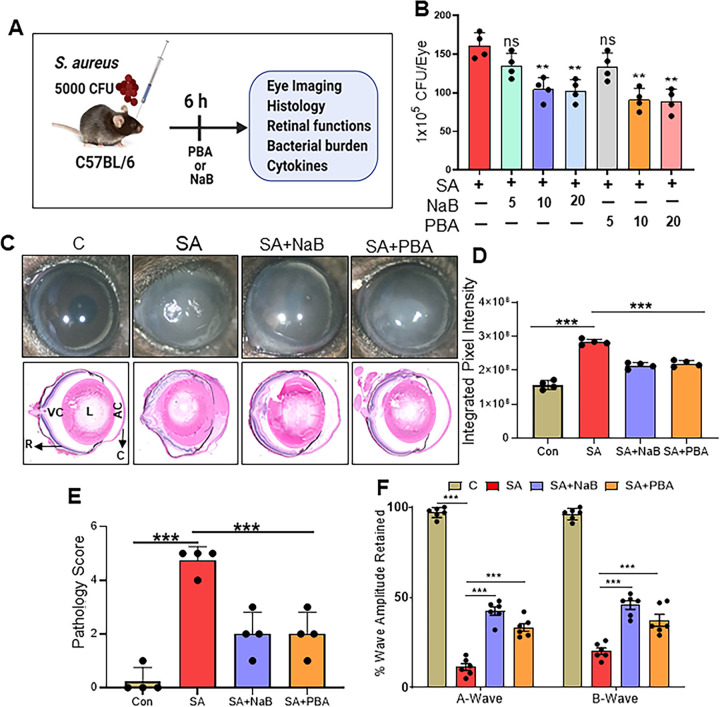
Our interest in investigating the role of butyrate in the pathobiology of bacterial endophthalmitis emerged from our recent untargeted metabolomics study (30, 31). Of the various metabolites, we found temporal declines in the levels of butyrate derivatives, 3-hydroxybutyrate, and 3-hydroxyisobutyrate in S. aureus-infected retinal tissue (Fig. S1 in the supplemental material). Next, we assessed the expression of potential butyrate transporters in retinal residential (human Müller glia) and infiltrating immune (mouse macrophages) cells. Our data showed that the transcription of several G-protein coupled receptor (GPCR) genes, such as Gpr41, Gpr109A, and Slc5A8, was induced by S. aureus infection, and their expression was potentiated by NaB or PBA treatment in both cell types (Fig. S1). Because butyrate has been shown to downregulate inflammation, we hypothesized that butyrate supplementation would exert protective effects during bacterial endophthalmitis. To test this, we used two commercially available butyrate derivatives (NaB and PBA) and performed a dose-response study using intravitreal injections of various dosages (5, 10, or 20 μg/eye) for each metabolite, as shown in Fig. 1A. First, we assessed the antibacterial effects and observed that both NaB and PBA treatments significantly reduced bacterial burdens in the eye both at 10- and 20-μg dosages (Fig. 1B). Thus, we decided to use the 10-μg dose for the rest of the study. Next, we tested the therapeutic efficacy of butyrate by a gross eye examination and found that both the NaB- and PBA-treated groups had diminished corneal haze, opacity, and hypopyon compared to the untreated groups (Fig. 1C, upper panel; Fig. 1D). The protective effect of butyrate was also confirmed by histological analysis which showed reduced retinal tissue damage in butyrate-treated eyes (Fig. 1C, lower panel; Fig. 1E). The preservation of retinal architecture by butyrate treatment coincided with improved retinal function, as indicated by significant retention of a- and b-wave amplitudes (Fig. 1F).
FIG 1.
Butyrate ameliorates bacterial endophthalmitis in mice. Endophthalmitis was induced in C57BL/6 mice eyes (n = 4) by intravitreal injection of Staphylococcus aureus. Eyes were treated with various doses (5, 10, 20 μg/eye) of sodium butyrate (NaB) or phenylbutyrate (PBA) via intravitreal injection at 6 h postinfection. (A) Timeline of induction of endophthalmitis, butyrate treatment, and assays used to monitor disease progression. (B) Quantitation of intraocular bacterial burden by serial dilution and plate count, represented as CFU/eye. (C) Upper panel: representative slit-lamp images showing corneal haze/opacity. Lower panel: hematoxylin and eosin (H&E) staining of eyes enucleated at 30 h postinfection. (D) Corneal opacity was measured using ImageJ and represented as integrated pixel intensity. (E) H&E was quantified by pathology score, with 4 being the worst and 0 being the best. (E) Scotopic electroretinography (ERG) response as measured by the percentage of a- and b-wave amplitudes retained in comparison to uninfected control eyes, with values kept at 100%. Data represent the culmination of two independent experiments and are shown as mean ± standard deviation (SD). Statistical analysis was performed using one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons (panels B and D). **, P < 0.001; ***, P < 0.0001; ns, not significant.
Because increased inflammation is a hallmark of endophthalmitis, we evaluated the effect of butyrate treatment on intraocular inflammation. Our data show that eyes treated with NaB or PBA had significantly less inflammation, as indicated by reduced production of inflammatory mediators such as interleukin (IL)-1β, IL-6, tumor necrosis factor α (TNF-α), and C-X-C motif chemokine ligand 2 (CXCL2; Fig. 2A) and decreased neutrophil infiltration (Fig. 2B). Although we observed butyrate-mediated protection, some eyes still exhibited considerable corneal haze and opacity at 30 h postinfection. Thus, we monitored the animals until 72 h post-treatment for disease resolution. Our data showed that inflammatory response and bacterial burden were lower in butyrate-treated eyes, while corneal haze and opacity still persisted at later time points (Fig. S2). We also tested the effect of delayed butyrate treatment by injecting eyes with NaB or PBA at 12 h postinfection instead of at 6 h postinfection. Here, we found that butyrate was still effective in reducing the bacterial burden and inflammatory cytokines, but the magnitude of this protection was comparatively less than that of 6-h postinfection treatment (Fig. S3). However, when butyrate was administered along with an antibiotic (sub-MIC levels of vancomycin), it exerted a synergistic effect in reducing inflammation and bacterial burden, resulting in better protection (Fig. S3). These results support the notion that butyrate can be used as an adjunct therapy to treat bacterial endophthalmitis.
FIG 2.
Butyrate treatment reduces the inflammatory response and neutrophil infiltration in the eyes. Endophthalmitis was induced in C57BL/6 mouse eyes by intravitreal inoculation of 5,000 CFU of S. aureus strain RN6390. After 6 h, eyes were treated with butyrate derivatives (10 μg/eye) NaB or PBA via intravitreal injection. (A) Enzyme-linked immunosorbent assay (ELISA) was performed to measure the levels of indicated inflammatory cytokines or chemokines in whole eye lysates (n = 6). (B) Flow cytometry was performed using retinal single-cell suspensions to determine neutrophil infiltration (CD45+Ly6G+ cells). Bar graph represents the percentage of neutrophil infiltration with or without butyrate derivative treatment. Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test (panels A and B). **, P < 0.001; ***, P < 0.0001; ns = not significant.
Butyrate attenuates bacterial-induced inflammatory response in cultured cells.
In endophthalmitis, both residential (e.g., Müller glia) (32) and infiltrating immune cells (e.g., macrophages) play critical roles in initiating the innate inflammatory response. However, aberrant inflammation by immune cells often causes retinal tissue damage (24, 30). To test the effect of butyrate treatment in vitro, we used mouse bone marrow-derived macrophages (BMDMs) and human retinal Müller glia (MIO-M1 cell line). We observed that S. aureus induced the expression of inflammatory cytokines, and that butyrate treatment attenuated this response in both cell types. Our data showed that both NaB- and PBA-treated cells exhibited significantly reduced mRNA expression of Il1β, Il6, and Tnfα in mouse macrophages and human Müller glia, respectively (Fig. 3A and C). Similar reductions were observed in the protein levels of these inflammatory mediators, as assessed by ELISA (Fig. 3B and D). These results indicate that butyrate treatment diminishes the inflammatory response in retinal residential and infiltrating innate immune cells during intraocular bacterial infection.
FIG 3.
Butyrate attenuates S. aureus-induced inflammatory response in cultured cells. (A and B) Mouse bone marrow-derived macrophages (BMDMs) from C57BL/6 mice (n = 4) and (C and D) human retinal Müller glia (MIO-M1 cell line) were pretreated with NaB or PBA (3 mM) for 2 h followed by infection with S. aureus (MOI = 10:1) for 6h. The transcript levels of inflammatory cytokines were measured by quantitative PCR (qPCR) (A and C), and ELISA was used to estimate protein levels (B and D). Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test by comparing S. aureus-infected samples with or without butyrate treatment. **, P < 0.001, ***, P < 0.0001.
Butyrate treatment inhibits NLRP3 inflammasome.
The observed anti-inflammatory effects of butyrate may be due to the direct antimicrobial properties of butyrate (33). To confirm this, we challenged BMDMs with heat-killed S. aureus (HKSA) and assessed the inflammatory response. Our data show that HKSA-induced expression of various inflammatory mediators and that butyrate pretreatment significantly reduced their levels at both the transcript and protein levels (Fig. S4). We recently reported that during endophthalmitis, S. aureus triggers an inflammatory response by activating the NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome pathway (34). The activation of the NLRP3 inflammasome is primarily responsible for the cleavage of pro-IL-1β into active IL-1β via activation of caspase 1. Therefore, we sought to determine the effect of butyrate on the NLRP3 inflammasome pathway. We found that both NaB or PBA treatment downregulated Nlrp3 and Caspase1 transcription (Fig. 4A and B). Moreover, a Western blot analysis revealed decreased levels of NLRP3 protein in butyrate-treated cells (Fig. 4C and D). These results indicate that butyrate treatment may exert anti-inflammatory effects by inhibiting NLRP3 signaling.
FIG 4.

Butyrate inhibits S. aureus-induced NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome. BMDMs from C57BL/6 mice (n = 4/condition) were either left untreated or pretreated with NaB or PBA at 3 mM for 2 h, followed by S. aureus (MOI = 10:1) challenge for 6 h. (A and B) qPCR was performed to determine Nlrp3 and Caspase1 expression. Data are expressed as relative fold change by normalizing gene expression to the endogenous β-actin gene. (C) Western blot detection of NLRP3 and (D) densitometry analysis was performed using ImageJ; data are expressed as relative fold changes normalized to the loading control HSP90. Data represent the culmination of two independent experiments and are shown as mean ± SD. Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test by comparing S. aureus-infected samples with or without NaB or PBA treatment. ***, P < 0.0001; ns = not significant.
In our recent study, we reported that NLRP3 deficiency increases disease severity in mice during endophthalmitis, indicating an essential role of NLRP3 signaling (34). Paradoxically, here, our in vitro data show that butyrate reduces NLRP3 and inflammatory cytokines. To investigate the involvement of NLRP3 in butyrate treatment, we used a potent pharmacological inhibitor of NLRP3, MCCP50, alone or in combination with butyrate. First, we confirmed NLRP3 inhibition by MCCP50 (Fig. S5). Afterwards, we injected eyes with MCCP50 for 12 h to inhibit NLRP3, followed by butyrate treatment at 6 h postinfection. Our results showed that NLRP3 inhibition alone increased the corneal haze, opacity, hypopyon (Fig. 5A and B), and intraocular bacterial burden (Fig. 5C), as well as inflammatory mediators except for IL-1β and TNF-α (Fig. 5D). Interestingly, butyrate treatment in NLRP3-inhibited mice still produced significant reductions in corneal haze, opacity, bacterial burden, and inflammatory mediators (Fig. 5A to D). Collectively, these results indicate that although butyrate antagonizes NLRP3, it may exert NLRP3-independent anti-inflammatory effects.
FIG 5.
NLRP3 inhibition exacerbates the disease pathology and diminishes the anti-inflammatory properties of butyrate. C57BL/6 mice eyes (n = 4) were injected with the selective NLRP3 inhibitor MCC950 (50 μM/eye) 12 h before induction of endophthalmitis. After 6 h postinfection, eyes were treated with butyrate derivatives (10 μg/eye) NaB or PBA. (A) Representative slit-lamp images showing corneal haze/opacity, with increased corneal opacity (B) represented as integrated pixel intensity by ImageJ quantification. (C) Quantitation of intraocular bacterial burden by serial dilution and plate count represented as CFU/eye (n = 4). (D) ELISA was performed to measure levels of indicated inflammatory cytokines or chemokines in whole-eye lysates (n = 4). Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test. *, P < 0.05; **, P < 0.001; ***, P < 0.0001; ns = not significant.
Butyrate treatment promotes bacterial phagocytosis and killing.
Because we observed reduced intraocular bacterial burden in mouse eyes, we sought to determine whether butyrate derivatives possess direct antibacterial activity. We performed a MIC assay and found that the MICs of both butyrate derivatives were ~25 mg/mL (Fig. S5). However, the butyrate dose (10 μg) used for the treatment was lower than the MIC, indicating other antimicrobial mechanisms of butyrate. We postulated that butyrate increases the phagocytic activity of innate immune cells to restrict bacterial growth. In support, our data show that butyrate-pretreated BMDMs had increased intracellular S. aureus at 1 h post-challenge, indicating enhanced phagocytic uptake of bacteria (Fig. 6A). At the later time points, i.e., 3 and 6 h, the recovery of viable intracellular bacteria began to decline in butyrate-treated cells, indicating intracellular killing. We have previously shown that human Müller glia can also phagocytize and kill S. aureus (32). Similarly, butyrate-treated Müller glia also showed enhanced phagocytic uptake at early time points and reduced intracellular bacterial burdens at later time points (Fig. 6B). Consistent with enhanced antibacterial activity, butyrate treatment induced the expression of antimicrobial molecules, such as LL-37, Cramp (murine homolog of human LL37), S100A7, and S100A8 in both BMDMs (Fig. 7A) and Müller glia (Fig. 7B). Because activated Müller glia exhibit robust bactericidal activity against S. aureus (28, 35), we determined S100A8 and LL37 expression at the protein level using immunofluorescence staining. Our results showed induced expression of S100A8 and LL37 in butyrate-treated Müller glia (Fig. 7C). Most importantly, butyrate-treated mouse eyes also exhibited induced expression of S100a7, S100a8, and Cramp in the retina (Fig. 7D). Collectively, our data indicate that butyrate exerts antibacterial effects by enhancing phagocytic killing and inducing expression of antimicrobial molecules.
FIG 6.

Butyrate treatment promotes bacterial phagocytosis and killing. (A) BMDMs from C57BL/6 mice and (B) human retinal Müller glia (MIO-M1 cell line) were pretreated with NaB (3 mM) for 2 h followed by challenge with S. aureus (MOI = 10:1). After 2 h of infection, cells were rinsed to remove extracellular bacteria and incubated with fresh medium containing gentamicin (200 μg/mL) for the indicated time points. At the desired time points, cells were lysed, and viable bacterial counts were quantitated via serial dilution and plate count. Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test (A and B). Data represent mean ± SD from three independent experiments. *, P < 0.05; **, P < 0.001; ns = not significant.
FIG 7.
Butyrate treatment induced the expression of antimicrobial molecules both in vitro and in vivo. (A) BMDMs from C57BL/6 mice (n = 6/condition) and (B and C) human retinal Müller glia (MIO-M1 cell line) were either left untreated or pretreated with NaB or PBA (3 mM) for 2 h followed by infection with green fluorescent protein (GFP)-expressing S. aureus (green) at 6 h for qPCR or 3 h for immunostaining. The expression of genes (e.g., S100a7, S100a8, Cramp, LL37) with antimicrobial functions was assessed by qPCR (A and B) or immunostaining for S100A7 (red) or LL37 (red) expression, and images were captured at ×60 original magnification (C). To assess the expression of these genes in vivo, mouse retinal tissue was used for qPCR (D). Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test (panels A, B, and D). Data represent mean ± SD from three independent experiments. *, P < 0.05; **, P < 0.001; ***, P < 0.0001; ns = not significant.
Butyrate potentiates autophagy and autophagosome-mediated bacterial killing.
One potential mechanism for intracellular pathogen killing is the induction of autophagy. We observed that butyrate treatment activated autophagy in BMDMs, as indicated by the degradation of p62 and subsequently increased accumulation of the lipidated form of LC3BII (Fig. 8A and B). Concomitant with autophagy, our data showed that both NaB and PBA treatment induced AMP-activated protein kinase (AMPK) activation, as evidenced by increased levels of p-AMPK and its downstream target p-ACC (Fig. 8C and D). This is consistent with our prior study showing perturbation in AMPK-mediated energy metabolism in ocular infections (22, 36). Similar to that in BMDMs, immunostaining of S. aureus-infected Müller glia showed the induction of autophagy markers, p62 and LC3BII, in butyrate-treated cells (Fig. 8E). Collectively, these results indicate that butyrate treatment potentiates autophagy activation by augmenting AMPK signaling pathways. Among the antimicrobial peptides, LL37 has been shown to mediate bacterial killing via autophagosomes (37, 38). Thus, we used immunofluorescence staining to determine whether LL37 co-localizes in the autophagosome, using LC3BII as an autophagosome marker. Our results show that butyrate treatment significantly increased the co-localization of LL37 and LC3BII puncta in Müller glia cells, implicating autophagosome-mediated bacterial killing (Fig. 9A and B).
FIG 8.
Butyrate potentiates autophagy and autophagosome-mediated bacterial killing. (A to C) BMDMs from C57BL/6 mice (n = 3) were pretreated with 3-methyladenine (3-MA) (1 mM), NaB, or PBA (3 mM) for 2 h, followed by infection with S. aureus (MOI = 10:1) for 3 or 6 h. (A and C) Western blot analysis was performed to detect p62, LC3BII, pAMPK, total AMP-activated protein kinase (AMPK), pACC, total ACC, β-actin, and HSP90. (B and D) Densitometric analysis of immunoblots were performed using ImageJ. Data are expressed as relative fold change by normalizing the expression of proteins with respect to the housekeeping controls β-actin or HSP-90. (E) Protein expression of autophagy markers p62 and LC3BII was determined in human retinal Müller glia (MIO-M1 cell line) by immunostaining. Images were captured at ×60 magnification. Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test (B and D). *, P < 0.01; **, P < 0.001; ***, P < 0.0001; ns = not significant.
FIG 9.
Butyrate treatment potentiates the co-localization of LL37 and LC3BII. Human retinal Müller glia (MIO-M1 cell line) were pretreated with NaB or PBA (3 mM) for 2 h followed by challenge with S. aureus (MOI = 10:1) for 3 h. (A) Representative images showing immunostaining for LC3BII (green), LL37 (red), and DAPI (4′,6-diamidino-2-phenylindole; blue, a cell nuclear stain). All images were captured at ×60 magnification. (B) Bar graph showing the fraction of LL37 and LC3BII co-localization cells/field. Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test. Data represent mean ± SD from two independent experiments. **, P < 0.001.
Because we observed that butyrate promotes autophagy and autophagosome-mediated killing of S. aureus in cultured cells, we sought to determine the effect of autophagy in vivo. We used an autophagy inhibitor, 3-methyladenine (3-MA), which blocks autophagy at initiation and autophagosome formation, as confirmed by reduced p62 and LC3BII levels (Fig. 8A). Our data show that autophagy inhibition during endophthalmitis exacerbates intraocular bacterial burden and inflammation and that the protective effects of butyrate were diminished (Fig. 10). These results support the role of autophagy in butyrate-mediated protection in bacterial endophthalmitis.
FIG 10.
Autophagy inhibition exacerbates endophthalmitis and diminishes the protective effects of butyrate. C57BL/6 mice eyes (n = 4) were injected with selective autophagy inhibitor 3-MA (500 μM/eye) 12 h before induction of endophthalmitis. At 6 h postinfection, eyes were treated with NaB or PBA (10 μg/eye) via intravitreal injections. (A) Representative slit-lamp images showing corneal haze/opacity and (B) increased corneal opacity represented as integrated pixel intensity by ImageJ quantification. (C) Quantitation of intraocular bacterial burden by serial dilution and plate count represented as CFU/eye (n = 4). (D) ELISA was performed to measure the levels of indicated inflammatory cytokines or chemokines in whole-eye lysates (n = 4). Statistical analysis was performed using one-way ANOVA with Tukey’s multiple-comparison test. *, P < 0.01; **, P < 0.001; ***, P < 0.0001; ns = not significant.
DISCUSSION
Intravitreal antibiotic injections remain the main treatment option for bacterial endophthalmitis. Antibiotic treatment clears the bacteria, but dead bacterial cell wall components and toxins contribute to host-induced aberrant inflammation (27, 39). To control ocular inflammation, intravitreal injections of steroids are often considered, but their role in infectious endophthalmitis remains controversial due to side effects and immunosuppression (40–42). Therefore, newer anti-inflammatory therapeutic modalities are needed as adjunct therapy (30). In this study, we show that butyrate treatment ameliorates bacterial endophthalmitis and that butyrate exerted both anti-bacterial and anti-inflammatory properties when injected into the eye. Moreover, butyrate exhibited synergistic effects along with antibiotics in treating bacterial endophthalmitis.
Butyrate is primarily produced in the gut through the bacterial fermentation of dietary fibers (1). In the gut, butyrate promotes homeostasis by rewiring the host mucosal immune system via epigenetic regulation of resident intestinal immune cells (11). In our recent study, we used a global metabolomics approach to identify metabolic pathways which are altered during endophthalmitis, and our data showed that butyrate levels are decreased in S. aureus-infected mouse retina. These observations indicated that the gut-eye axis may play a role in orchestrating the innate retinal response during infection. Indeed, a recent study demonstrated that short-chain fatty acids from the gut can reach the eye by crossing the blood-retina barrier, ameliorate endotoxin-induced uveitis, and attenuate retinal astrocyte inflammatory responses (19). Therefore, we began this study to investigate the role of butyrate in bacterial endophthalmitis. Given the anti-inflammatory role of butyrate in various inflammatory diseases (3, 16, 37), we hypothesized that butyrate promotes inflammation resolution during ocular infections.
Here, we demonstrated for the first time that intravitreal injection of butyrate derivatives (NaB or PBA) protects mice from endophthalmitis by reducing the bacterial burden and inflammatory response. Butyrate has been shown to protect the intestinal epithelium from Clostridium difficile toxins by stabilizing HIF-1α and increasing tight junctions, resulting in reduced intestinal epithelial permeability and bacterial translocation (43). Several cell surface receptors, mainly GPCRs, including Gpr41, Gpr109A, and Slc5a8, have been identified for butyrate. They are primarily expressed in immune cells, the colonic epithelium, and adipose and corneal tissues (44, 45). Butyrate activates these receptors to suppress colonic inflammation and carcinogenesis (46). Here, we report the expression of these receptors on human Müller glia and infiltrating macrophages. Moreover, their expression was induced by S. aureus infection and potentiated by NaB or PBA treatment, indicating the involvement of these receptors in exerting the effects of butyrate in bacterial endophthalmitis.
We observed that both butyrate derivatives inhibit bacterial growth, mitigated the S. aureus-induced inflammatory response, and improved disease outcomes. In all experiments, we administered butyrate once at 6 h postinfection. However, a single intravitreal injection of butyrate may not be sufficient to control ocular infection as opposed to the repeated injections in human endophthalmitis. Moreover, we observed that delayed (12 h postinfection) butyrate treatment was not as effective as early treatment (6 h postinfection). We postulate that by 12 h bacteria continue to grow inside the eye, coinciding with massive intraocular inflammation, and at this stage, therapeutic modalities could fail to reverse the damage. These observations are consistent with clinical management of endophthalmitis, in which early intervention is essential to prevent irreversible vision loss in patients (47, 48). However, consistent with our prior study (30), butyrate can be administered along with antibiotics to reduce inflammation even in the later stages of endophthalmitis. In this proof-of-concept study, we primarily focused on elucidating the mechanisms underlying butyrate-mediated protection. Future studies are needed to fully evaluate butyrate for the potential treatment of ocular infections alone or in combination with antibiotics.
NLRP3 inflammasome is implicated in the pathology of many infectious diseases (49–51). NLRP3 regulates IL-1β and IL-18 cytokines via activation of caspase-1. NLRP3 inflammasome is a double-edged sword, where its lack or excessive activation can be detrimental during infections. Indeed, in our recent study, we found that NLRP3–/– mice had increased endophthalmitis severity due to increased bacterial burden (34). However, at later stages of infection, NLRP3 activation can lead to uncontrolled inflammation, resulting in retinal tissue damage. Here, we show that butyrate derivatives can inhibit the S. aureus-induced NLRP3, resulting in an attenuated inflammatory response. Moreover, butyrate treatment reduced neutrophil infiltration in S. aureus-infected eyes. This is most likely due to reduced levels of CXCL2, a key chemokine known to recruit neutrophils during bacterial (Bacillus) endophthalmitis (52). To determine the role of NLRP3 in butyrate-mediated effects, we used a pharmacological approach to inhibit NLRP3. We observed that NLRP3 inhibition alone increased disease severity, but butyrate treatment still exerted protection in NLRP3-inhibited mice. These findings indicate that butyrate antagonizes NLRP3 and that its protective effects may also be NLRP3-independent. Histone deacetylation and the modulation of regulatory T cell functions have been reported as underlying mechanisms of the anti-inflammatory effects of butyrate (13, 53, 54).
Our data showed that butyrate treatment reduced bacterial burdens in infected mouse eyes. However, the MIC of butyrate against S. aureus was ~25 mg/mL, which is higher than used for in vivo treatments. This led us to hypothesize that butyrate exerts its antibacterial effects indirectly by influencing the bactericidal activity of retinal residential and infiltrating immune cells. Indeed, the indirect antibacterial activity of butyrate has been demonstrated in Mycobacterium tuberculosis infection and colitis disease models via the production of antimicrobial peptides (10, 38). Our data also showed that butyrate treatment induced the production of many antimicrobial peptides such as cathelicidins (i.e., CRAMP, LL37), S100A7, and S100A8 in the mouse retina. Moreover, BMDMs and retinal Müller glia exposed to butyrate exhibited higher levels of these antimicrobial molecules in response to S. aureus infection. Similarly, butyrate was shown to induce LL-37 expression in colonic epithelial cells (55). The induced expression of antimicrobial molecules coincided with enhanced uptake and intracellular killing of S. aureus by Müller glia and BMDMs. These findings support the indirect effect of butyrate treatment in reducing bacterial load in the eye.
Host cells employ several mechanisms, including autophagy, to restrict the growth of bacteria and kill them intracellularly (56, 57). In contrast, pathogens can induce autophagy in a nonselective manner to evade the host-mediated immune response (58–60). Our data showed that butyrate treatment activated autophagy pathways in S. aureus-infected cells, as evidenced by p62 degradation and increased lipidated LC3BII puncta. Moreover, we observed that butyrate treatment enhanced the co-localization of LL37 in autophagosomes, indicating autophagosome-mediated killing of S. aureus. While LL37 has been shown to mediate bacterial killing via autophagosomes in macrophages (37, 38), our study is the first to demonstrate this phenomenon in retinal Müller glia. This observation also provides a mechanistic basis for the antibacterial properties of retinal Müller glia as reported in our prior studies (28, 32). To investigate the role of autophagy in butyrate-mediated protection in mice, we used an autophagy inhibitor, 3-MA. Our findings indicated that autophagy inhibition exacerbated disease outcomes, and butyrate provided partial protection when used along with an autophagy inhibitor. These results indicate that autophagy plays a protective role in endophthalmitis, likely by promoting bacterial killing. Thus, butyrate exerts protective effects, in part by enhancing autophagy. However, further studies using autophagy-deficient mice are warranted to delineate the role of autophagy in bacterial endophthalmitis.
Previously, we reported that energy metabolism is perturbed during bacterial endophthalmitis, with increased glycolysis (22, 61). In energy metabolism, AMPK signaling regulates various pathological processes during normal and disease conditions. We found that AMPK deficiency increases endophthalmitis severity, whereas pharmacological activation of AMPK promotes disease resolution (22). Because AMPK is known to regulate autophagy, we sought to determine this link. In support, our data showed that butyrate treatment induced the activation (i.e., phosphorylation) of AMPK and its downstream target ACC in retinal Müller glia cells. Butyrate-mediated AMPK activation has also been reported in intestinal epithelial and adipocytes (62, 63). Given the protective role of AMPK in ocular bacterial (22) and viral infections (36), we postulate that butyrate potentiates autophagy by augmenting AMPK, therefore resulting in reduced bacterial burdens and inflammation (Fig. 11).
FIG 11.

Mechanisms underlying butyrate treatment mediated protection in the eye. Butyrate derivatives (NaB or PBA) exert both antibacterial and anti-inflammatory properties in mouse eyes and cultured cells (macrophages and Müller glia). Butyrate treatment activates AMPK, promotes selective autophagy, induces antimicrobial peptide expression, and inhibits NLRP3 expression. This ameliorates bacterial endophthalmitis by increasing bacterial clearance and reducing intraocular inflammation.
In conclusion, our study demonstrates that butyrate treatment reduced bacterial burdens and attenuated bacterial-induced intraocular inflammation, resulting in the preservation of vision. Therefore, butyrate could be explored as an adjunct antimicrobial and anti-inflammatory therapy to treat ocular bacterial infections.
MATERIALS AND METHODS
Mice and ethical statement.
C57BL/6 (B6) mice (male and female, aged 6 to 8 weeks) were purchased from the Jackson Laboratory (Bar Harbor, ME). Mice were housed in a restricted-access Division of Laboratory Animal Resources (DLAR) facility at the Kresge Eye Institute, maintained on a 12-h light 12-h dark cycle at 22°C temperature, and fed on LabDiet rodent chow (PicoLab; LabDiet, St. Louis, MO) and water ad libitum. All procedures were performed in compliance with the Animals in Ophthalmic and Vision Research (ARVO) statement for the use of animals and were approved by the institutional animal care and use committee (IACUC) of Wayne State University.
Bacterial strains and induction of endophthalmitis.
S. aureus strain RN6390 and the green fluorescent protein (GFP)-expressing strain AL1743 were maintained in tryptic soy broth and agar plates (TSB/TSA; Sigma-Aldrich, St. Louis, MO). Endophthalmitis was induced in B6 mice as described previously (24, 64). Briefly, mice were anesthetized and intravitreally injected with S. aureus (5,000 CFU/eye) using a 34G needle under a dissecting microscope inside a biosafety cabinet. Contralateral eyes were injected with sterile phosphate-buffered saline (PBS) and used as controls. For treatment groups, the butyrate derivatives sodium butyrate or phenylbutyrate (Cayman Chemicals, Ann Arbor, MI) were injected intravitreally (10 μg/eye) at 6 h post-bacterial infection. For inhibition studies, mice were intravitreally injected with NLRP3 inhibitor MCC950 (50 μM/eye) or autophagy inhibitor 3-methyladenine (500 μM/eye; Cayman Chemicals, Ann Arbor, MI).
Isolation of bone marrow-derived macrophages.
Mouse bone marrow-derived macrophages were isolated as described previously (65). Briefly, mice were euthanized, and bone marrow was flushed from the tibia and femur with RPMI medium containing 10% fetal bovine serum (FBS) and 0.2 mM EDTA. Cells were pelleted by centrifugation at 400 × g for 5 min at 4°C. Red blood cells were lysed by adding 0.2% NaCl solution for 20 sec, followed by the addition of 1.6% NaCl and centrifugation. Afterward, cell pellets were rinsed with RPMI medium and resuspended and seeded in RPMI medium supplemented with 10% FBS, 100 U/mL penicillin, 100 mg/mL streptomycin, and 10 ng/mL macrophage colony-stimulating factor for macrophage differentiation at 37°C in 5% CO2. Six days post-differentiation, 4 × 106 BMDMs/mL cells were seeded in 65-mm petri dishes for in vitro experiments. For drug treatments, BMDMs were pretreated with NaB or PBA (3 mM each) for 2 h, followed by challenge with S. aureus at an MOI of 10:1 for 6 h. To produce heat-killed S. aureus, a similar MOI was boiled for 5 min and used for in vitro experiments.
Müller glia culture.
The immortalized human Müller glia cell line MIO-M1 was cultured in Dulbecco’s modified Eagle medium (DMEM) supplemented with 10% FBS, 1% penicillin-streptomycin, and 10 μg/mL l-glutamine. Before infection, cells were grown overnight in serum- and antibiotic-free medium and infected with S. aureus at an MOI of 10:1 for the desired time points. PBS-treated cells were used as a vehicle control. For drug treatments, MIO-M1 cells were pretreated with PBA (3 mM) for 2 h, followed by S. aureus infection.
Electroretinography.
Scotopic electroretinography (ERG) was used to determine retinal function in S. aureus-infected and butyrate-treated mouse eyes using the Celeries ERG system (Diagnosis LLC, Lowell, MA), per the manufacturer’s recommendations.
Bacterial burden estimation.
Eyes were enucleated following S. aureus infection and 24 h post-drug treatment. Whole-eye lysates were prepared in 250 μL PBS using stainless steel beads and a tissue lyser (Qiagen, Valencia, CA). A small portion (50 μL) of the tissue homogenate was serially diluted and plated on TSA to enumerate the bacterial burden. The following day, the colonies were counted, and the results were expressed as the mean number of CFU/eye.
Cytokine ELISA.
For in vivo samples, eyes were enucleated, and lysates were prepared using a tissue lyser as described above. The tissue homogenates were centrifuged at 10,000 × g for 20 min at 4°C, and the supernatant was used for cytokine estimation. For in vitro studies, the conditioned media from the S. aureus-infected and drug-treated groups and the vehicle control were preserved for cytokine measurements. A mouse enzyme-linked immunosorbent assay (ELISA) was performed for the inflammatory cytokines IL-1β, IL-6, TNF-α, and CXCL2 per the manufacturer’s instructions (R&D Systems, Minneapolis, MN). ELISA was performed for the inflammatory cytokines IL-1β, IL-8, and TNF-α per the manufacturer’s instructions (BD Biosciences, San Jose, CA).
Histology.
Eyes were enucleated and fixed in modified Davidson’s fixative for histopathological examination. The embedding, sectioning, and hematoxylin and eosin (H&E) staining was performed by Excalibur Pathology, Inc. (Oklahoma City, OK).
RNA extraction and qPCR analysis.
Total RNA was extracted from the neural retina and BMDM or MIO-M1 cells by TRIzol per the manufacturer’s instructions (Thermo Fisher Scientific, Rockford, IL). RNA was reverse-transcribed using a Maxima First Strand cDNA Synthesis kit per the manufacturer’s protocol (Thermo Fisher Scientific). Quantitative assessment of gene expression was carried out by SYBR Green-based quantitative PCR (qPCR) using gene-specific primers on a Step One Plus Real-Time PCR system (Applied Biosystems, Foster City, CA). The data were analyzed using the comparative threshold cycle (ΔΔCT) method as described previously (66). Gene expression in the test samples was normalized to the endogenous β-actin controls.
MIC.
A micro broth-dilution method was used to determine the MICs for NaB and PBA as described previously (64, 67). Briefly, bacterial cultures (105 CFU/well) were exposed to a 2-fold serial dilution of the test compound in a 96-well plate. Following overnight incubation, the optical density (A600) of each microplate well was recorded using a spectrophotometer. MICs were determined based on the optical density of the growth control and the lowest NaB or PBA concentrations which resulted in the inhibition of S. aureus growth compared with the negative-medium control.
Phagocytosis assay.
BMDMs and MIO-M1 cells (106 cells/dish) were grown in 60-mm petri dishes in RPMI 1640 or DMEM media, respectively. The cells were infected with S. aureus at an MOI of 10:1 for 2 h. Following incubation, the cells were rinsed and treated with gentamicin (200 μg/mL) for 2 h to kill extracellular bacteria. The absence of extracellular bacteria was confirmed via dilution plating and CFU enumeration. Two hours after gentamicin addition, cells were washed with RPMI 1640 or DMEM and incubated with fresh medium containing gentamicin (200 μg/mL) for 1, 3, and 6 h. For the enumeration of phagocytized bacteria, following incubation, the cells were washed three times with PBS and lysed with 0.01% Triton X-100. The lysed cells were scraped and centrifuged at 7,500 × g for 5 min. The cell pellets were washed twice with PBS by centrifugation at 7,500 × g for 5 min. The pellets were resuspended in 1 mL sterile PBS, serially diluted, and plated on TSA plates for bacterial count enumeration.
Flowcytometry.
Flow cytometry was performed to estimate neutrophil infiltration in the eyes as described previously (24). Briefly, retinas from euthanized mice were isolated and digested with Accumax (MilliporeSigma, Burlington, MA) for 10 min at 37°C. Single-cell suspension was prepared by triturating the cell mixture with a 22G needle, followed by filtration with a 40-μm cell strainer (BD Falcon, San Jose, CA). The cells were then blocked using Fc Block (BD Biosciences) for 30 min followed by three washes with PBS containing 1% bovine serum albumin (BSA). The cells were stained by incubating with CD45-APC and Ly6G-FITC (fluorescein isothiocyanate)-conjugated antibodies (BD Biosciences) on ice for 20 min in the dark. Following incubation, cells were washed and suspended in PBS with 1% BSA. The cells were acquired using an Accuri C6 flow cytometer and software, respectively (BD Biosciences). The flow cytometry results were analyzed using FlowJo v8 Software (BD Life Sciences).
Immunostaining.
Müller glia and MIO-M1 cells were cultured in a four-well chamber slide (Thermo Fisher Scientific, Rochester, NY) and pretreated with NaB or PBA for 2 h. Following treatment, cells were infected with the GFP-expressing S. aureus strain AL1743 at an MOI of 10:1 for 3 h. At the desired time points, infected cells were fixed overnight with 4% paraformaldehyde in PBS at 4°C. After washing with PBS, the cells were permeabilized and blocked with 1% (wt/vol) BSA and 0.4% Triton X-100 made in PBS for 1 h at room temperature (RT). Cells were incubated with primary antibodies; anti-LC3B II, anti-p62 (1:100; Cell Signaling Technology, Danvers, MA), anti-LL37, and anti-S100A8 (1:100; Invitrogen, Waltham, MA) overnight at 4°C. Following removal of the primary antibody, the cells were washed extensively with PBS and incubated with anti-mouse/rabbit Alexa Fluor 485/594-conjugated antibody (1:200) for 1 h at RT. Finally, the cells were extensively washed with PBS and the slides were mounted in Vectashield Antifade Mounting Medium (Vector Laboratories, Burlingame, CA) and visualized using a Keyence microscope (Keyence, Itasca, IL).
Western blotting.
Protein samples were prepared by direct lysis of cells in radioimmunoprecipitation assay (RIPA) buffer containing protease and phosphatase inhibitor cocktails. The total protein concentration of the cell lysates was determined using a Micro BCA protein assay kit (Thermo Fisher Scientific, Rockford, IL). Protein samples (30 to 40 μg) were resolved on SDS-PAGE (8% or 16%) and transferred onto a nitrocellulose membrane (0.45 or 0.20 μm) using a wet-transfer system. Membranes were blocked in 5% (wt/vol) dried milk in Tris-buffered saline (TBS)-Tween (TBST) for 1 h at room temperature. Membranes were incubated with the following primary antibodies: anti-pAMPK, anti-pACC, anti-tAPMK, anti-tACC, anti-NLRP-3, anti-p62, anti-LC3BII, and anti-HSP-90 (Cell Signaling Technology, Danvers, MA) per the manufacturer’s instructions, followed by incubation with the appropriate horseradish peroxidase-conjugated secondary antibody. After four TBST washes, membranes were exposed to Super Signal West Femto chemiluminescent substrate and imaged using an iBright fluorescence imager (Thermo Scientific, Rockford, IL). Densitometry analysis was performed using ImageJ software (Rasband, W.S., ImageJ; NIH, Bethesda, MD [https://imagej.nih.gov/ij/]).
Statistical analysis.
Statistical analyses were performed using Prism v9.2 (GraphPad, San Diego, CA). A one-way analysis of variance was used for group comparisons followed by Tukey’s multiple-comparisons post hoc test, and data were expressed as mean ± standard deviation. A confidence interval of 95% was used for all statistical tests. A P value of <0.05 was considered statistically significant. All experiments were performed at least three times unless indicated otherwise.
ACKNOWLEDGMENTS
This study is supported by the NIH grants R01EY026964, R01EY027381, R21AI140033 (A.K.), and an unrestricted grant from Research to Prevent Blindness (RPB) to the Department of Ophthalmology, Visual, and Anatomical Sciences. P.K.S. is supported by NIH grant R01EY032495. The immunology resource core is supported by NIH center grant P30EY004068.
The authors are also grateful to other lab members for their helpful discussion and editing of the final manuscript.
The funders had no role in study design, data collection, interpretation, or the decision to submit the work for publication.
We declare no competing interests.
Footnotes
Supplemental material is available online only.
Contributor Information
Ashok Kumar, Email: akuma@med.wayne.edu.
Nancy E. Freitag, University of Illinois at Chicago
REFERENCES
- 1.Rooks MG, Garrett WS. 2016. Gut microbiota, metabolites and host immunity. Nat Rev Immunol 16:341–352. 10.1038/nri.2016.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tan J, McKenzie C, Potamitis M, Thorburn AN, Mackay CR, Macia L. 2014. The role of short-chain fatty acids in health and disease. Adv Immunol 121:91–119. 10.1016/B978-0-12-800100-4.00003-9. [DOI] [PubMed] [Google Scholar]
- 3.Ohira H, Tsutsui W, Fujioka Y. 2017. Are short chain fatty acids in gut microbiota defensive players for inflammation and atherosclerosis? J Atheroscler Thromb 24:660–672. 10.5551/jat.RV17006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van der Beek CM, Dejong CHC, Troost FJ, Masclee AAM, Lenaerts K. 2017. Role of short-chain fatty acids in colonic inflammation, carcinogenesis, and mucosal protection and healing. Nutr Rev 75:286–305. 10.1093/nutrit/nuw067. [DOI] [PubMed] [Google Scholar]
- 5.Vinolo MA, Rodrigues HG, Nachbar RT, Curi R. 2011. Regulation of inflammation by short chain fatty acids. Nutrients 3:858–876. 10.3390/nu3100858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Haase S, Haghikia A, Gold R, Linker RA. 2018. Dietary fatty acids and susceptibility to multiple sclerosis. Mult Scler 24:12–16. 10.1177/1352458517737372. [DOI] [PubMed] [Google Scholar]
- 7.Haghikia A, Jorg S, Duscha A, Berg J, Manzel A, Waschbisch A, Hammer A, Lee DH, May C, Wilck N, Balogh A, Ostermann AI, Schebb NH, Akkad DA, Grohme DA, Kleinewietfeld M, Kempa S, Thone J, Demir S, Muller DN, Gold R, Linker RA. 2015. Dietary fatty acids directly impact central nervous system autoimmunity via the small intestine. Immunity 43:817–829. 10.1016/j.immuni.2015.09.007. [DOI] [PubMed] [Google Scholar]
- 8.Perrigoue J, Das A, Mora JR. 2014. Interplay of nutrients and microbial metabolites in intestinal immune homeostasis: distinct and common mechanisms of immune regulation in the small bowel and colon. Nestle Nutr Inst Workshop Ser 79:57–71. 10.1159/000360685. [DOI] [PubMed] [Google Scholar]
- 9.Buffie CG, Bucci V, Stein RR, McKenney PT, Ling L, Gobourne A, No D, Liu H, Kinnebrew M, Viale A, Littmann E, van den Brink MR, Jenq RR, Taur Y, Sander C, Cross JR, Toussaint NC, Xavier JB, Pamer EG. 2015. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nature 517:205–208. 10.1038/nature13828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schulthess J, Pandey S, Capitani M, Rue-Albrecht KC, Arnold I, Franchini F, Chomka A, Ilott NE, Johnston DGW, Pires E, McCullagh J, Sansom SN, Arancibia-Carcamo CV, Uhlig HH, Powrie F. 2019. The short chain fatty acid butyrate imprints an antimicrobial program in macrophages. Immunity 50:432–445.e7. 10.1016/j.immuni.2018.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, deRoos P, Liu H, Cross JR, Pfeffer K, Coffer PJ, Rudensky AY. 2013. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 504:451–455. 10.1038/nature12726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Smith PM, Howitt MR, Panikov N, Michaud M, Gallini CA, Bohlooly YM, Glickman JN, Garrett WS. 2013. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341:569–573. 10.1126/science.1241165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Frank DN, St Amand AL, Feldman RA, Boedeker EC, Harpaz N, Pace NR. 2007. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci USA 104:13780–13785. 10.1073/pnas.0706625104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chang PV, Hao L, Offermanns S, Medzhitov R. 2014. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. Proc Natl Acad Sci USA 111:2247–2252. 10.1073/pnas.1322269111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rosser EC, Piper CJM, Matei DE, Blair PA, Rendeiro AF, Orford M, Alber DG, Krausgruber T, Catalan D, Klein N, Manson JJ, Drozdov I, Bock C, Wedderburn LR, Eaton S, Mauri C. 2020. Microbiota-derived metabolites suppress arthritis by amplifying aryl-hydrocarbon receptor activation in regulatory B cells. Cell Metab 31:837–851 e10. 10.1016/j.cmet.2020.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bachem A, Makhlouf C, Binger KJ, de Souza DP, Tull D, Hochheiser K, Whitney PG, Fernandez-Ruiz D, Dahling S, Kastenmuller W, Jonsson J, Gressier E, Lew AM, Perdomo C, Kupz A, Figgett W, Mackay F, Oleshansky M, Russ BE, Parish IA, Kallies A, McConville MJ, Turner SJ, Gebhardt T, Bedoui S. 2019. Microbiota-derived short-chain fatty acids promote the memory potential of antigen-activated CD8+ T cells. Immunity 51:285–297.e5. 10.1016/j.immuni.2019.06.002. [DOI] [PubMed] [Google Scholar]
- 17.Chang CH, Lin CH, Lane HY. 2020. d-Glutamate and gut microbiota in Alzheimer’s disease. Int J Mol Sci 21:2676. 10.3390/ijms21082676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cryan JF, O’Riordan KJ, Cowan CSM, Sandhu KV, Bastiaanssen TFS, Boehme M, Codagnone MG, Cussotto S, Fulling C, Golubeva AV, Guzzetta KE, Jaggar M, Long-Smith CM, Lyte JM, Martin JA, Molinero-Perez A, Moloney G, Morelli E, Morillas E, O'Connor R, Cruz-Pereira JS, Peterson VL, Rea K, Ritz NL, Sherwin E, Spichak S, Teichman EM, van de Wouw M, Ventura-Silva AP, Wallace-Fitzsimons SE, Hyland N, Clarke G, Dinan TG. 2019. The microbiota-gut-brain axis. Physiol Rev 99:1877–2013. 10.1152/physrev.00018.2018. [DOI] [PubMed] [Google Scholar]
- 19.Chen N, Wu J, Wang J, Piri N, Chen F, Xiao T, Zhao Y, Sun D, Kaplan HJ, Shao H. 2021. Short chain fatty acids inhibit endotoxin-induced uveitis and inflammatory responses of retinal astrocytes. Exp Eye Res 206:108520. 10.1016/j.exer.2021.108520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nakamura YK, Janowitz C, Metea C, Asquith M, Karstens L, Rosenbaum JT, Lin P. 2017. Short chain fatty acids ameliorate immune-mediated uveitis partially by altering migration of lymphocytes from the intestine. Sci Rep 7:11745. 10.1038/s41598-017-12163-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Skrzypecki J, Niewęgłowska K, Samborowska E. 2020. Valeric acid, a gut microbiota product, penetrates to the eye and lowers intraocular pressure in rats. Nutrients 12:387. 10.3390/nu12020387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kumar A, Giri S, Kumar A. 2016. 5-Aminoimidazole-4-carboxamide ribonucleoside-mediated adenosine monophosphate-activated protein kinase activation induces protective innate responses in bacterial endophthalmitis. Cell Microbiol 18:1815–1830. 10.1111/cmi.12625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kumar A, Kumar A. 2015. Role of Staphylococcus aureus virulence factors in inducing inflammation and vascular permeability in a mouse model of bacterial endophthalmitis. PLoS One 10:e0128423. 10.1371/journal.pone.0128423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Talreja D, Singh PK, Kumar A. 2015. In vivo role of TLR2 and MyD88 signaling in eliciting innate immune responses in staphylococcal endophthalmitis. Invest Ophthalmol Vis Sci 56:1719–1732. 10.1167/iovs.14-16087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Durand ML. 2013. Endophthalmitis. Clin Microbiol Infect 19:227–234. 10.1111/1469-0691.12118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Livingston ET, Mursalin MH, Callegan MC. 2019. A pyrrhic victory: the PMN response to ocular bacterial infections. Microorganisms 7:537. 10.3390/microorganisms7110537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Miller FC, Coburn PS, Huzzatul MM, LaGrow AL, Livingston E, Callegan MC. 2019. Targets of immunomodulation in bacterial endophthalmitis. Prog Retin Eye Res 73:100763. 10.1016/j.preteyeres.2019.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shamsuddin N, Kumar A. 2011. TLR2 mediates the innate response of retinal Muller glia to Staphylococcus aureus. J Immunol 186:7089–7097. 10.4049/jimmunol.1100565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shivaramaiah HS, Relhan N, Pathengay A, Mohan N, Flynn HW, Jr. 2018. Endophthalmitis caused by gram-positive bacteria resistant to vancomycin: clinical settings, causative organisms, antimicrobial susceptibilities, and treatment outcomes. Am J Ophthalmol Case Rep 10:211–214. 10.1016/j.ajoc.2018.02.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Singh S, Singh PK, Jha A, Naik P, Joseph J, Giri S, Kumar A. 2021. Integrative metabolomics and transcriptomics identifies itaconate as an adjunct therapy to treat ocular bacterial infection. Cell Rep Med 2:100277. 10.1016/j.xcrm.2021.100277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rajamani D, Singh PK, Rottmann BG, Singh N, Bhasin MK, Kumar A. 2016. Temporal retinal transcriptome and systems biology analysis identifies key pathways and hub genes in Staphylococcus aureus endophthalmitis. Sci Rep 6:21502. 10.1038/srep21502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Singh PK, Shiha MJ, Kumar A. 2014. Antibacterial responses of retinal Muller glia: production of antimicrobial peptides, oxidative burst and phagocytosis. J Neuroinflammation 11:33. 10.1186/1742-2094-11-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Du K, Bereswill S, Heimesaat MM. 2021. A literature survey on antimicrobial and immune-modulatory effects of butyrate revealing non-antibiotic approaches to tackle bacterial infections. Eur J Microbiol Immunol (Bp) 11:1–9. 10.1556/1886.2021.00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kumar A, Singh PK, Ahmed Z, Singh S, Kumar A. 2022. Essential role of NLRP3 inflammasome in mediating IL-1β production and the pathobiology of Staphylococcus aureus endophthalmitis. Infect Immun 90:e00103-22. 10.1128/iai.00103-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kumar A, Pandey RK, Miller LJ, Singh PK, Kanwar M. 2013. Muller glia in retinal innate immunity: a perspective on their roles in endophthalmitis. Crit Rev Immunol 33:119–135. 10.1615/critrevimmunol.2013006618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Singh S, Singh PK, Suhail H, Arumugaswami V, Pellett PE, Giri S, Kumar A. 2020. AMP-activated protein kinase restricts Zika virus replication in endothelial cells by potentiating innate antiviral responses and inhibiting glycolysis. J Immunol 204:1810–1824. 10.4049/jimmunol.1901310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Raqib R, Sarker P, Bergman P, Ara G, Lindh M, Sack DA, Nasirul Islam KM, Gudmundsson GH, Andersson J, Agerberth B. 2006. Improved outcome in shigellosis associated with butyrate induction of an endogenous peptide antibiotic. Proc Natl Acad Sci USA 103:9178–9183. 10.1073/pnas.0602888103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rekha RS, Rao Muvva SS, Wan M, Raqib R, Bergman P, Brighenti S, Gudmundsson GH, Agerberth B. 2015. Phenylbutyrate induces LL-37-dependent autophagy and intracellular killing of Mycobacterium tuberculosis in human macrophages. Autophagy 11:1688–1699. 10.1080/15548627.2015.1075110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhou R, Caspi RR. 2010. Ocular immune privilege. F1000 Biol Rep 2:3. 10.3410/B2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bui DK, Carvounis PE. 2014. Evidence for and against intravitreous corticosteroids in addition to intravitreous antibiotics for acute endophthalmitis. Int Ophthalmol Clin 54:215–224. 10.1097/IIO.0000000000000020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shah GK, Stein JD, Sharma S, Sivalingam A, Benson WE, Regillo CD, Brown GC, Tasman W. 2000. Visual outcomes following the use of intravitreal steroids in the treatment of postoperative endophthalmitis. Ophthalmology 107:486–489. 10.1016/s0161-6420(99)00139-6. [DOI] [PubMed] [Google Scholar]
- 42.Renfro L, Snow JS. 1992. Ocular effects of topical and systemic steroids. Dermatol Clin 10:505–512. 10.1016/S0733-8635(18)30318-8. [DOI] [PubMed] [Google Scholar]
- 43.Fachi JL, Felipe JS, Pral LP, da Silva BK, Correa RO, de Andrade MCP, da Fonseca DM, Basso PJ, Camara NOS, de Sales ESEL, Dos Santos Martins F, Guima SES, Thomas AM, Setubal JC, Magalhaes YT, Forti FL, Candreva T, Rodrigues HG, de Jesus MB, Consonni SR, Farias ADS, Varga-Weisz P, Vinolo MAR. 2019. Butyrate protects mice from Clostridium difficile-induced colitis through an HIF-1-dependent mechanism. Cell Rep 27:750–761.e7. 10.1016/j.celrep.2019.03.054. [DOI] [PubMed] [Google Scholar]
- 44.Ganapathy V, Thangaraju M, Prasad PD, Martin PM, Singh N. 2013. Transporters and receptors for short-chain fatty acids as the molecular link between colonic bacteria and the host. Curr Opin Pharmacol 13:869–874. 10.1016/j.coph.2013.08.006. [DOI] [PubMed] [Google Scholar]
- 45.Schaefer L, Hernandez H, Coats RA, Yu Z, Pflugfelder SC, Britton RA, de Paiva CS. 2022. Gut-derived butyrate suppresses ocular surface inflammation. Sci Rep 12:4512. 10.1038/s41598-022-08442-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Singh N, Gurav A, Sivaprakasam S, Brady E, Padia R, Shi H, Thangaraju M, Prasad PD, Manicassamy S, Munn DH, Lee JR, Offermanns S, Ganapathy V. 2014. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity 40:128–139. 10.1016/j.immuni.2013.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bari A, Chawla R, Mishra D, Das U, Hasan N, Satpathy G, Velpandian T, Azad SV, Venkatesh P, Vohra R, Kumar A. 2022. Real-life comparison of three intravitreal antibiotic drug regimens in endophthalmitis. Indian J Ophthalmol 70:1696–1700. 10.4103/ijo.IJO_2640_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Feng HL, Robbins CB, Fekrat S. 2020. A nine-year analysis of practice patterns, microbiologic yield, and clinical outcomes in cases of presumed infectious endophthalmitis. Ophthalmol Retina 4:555–559. 10.1016/j.oret.2020.03.009. [DOI] [PubMed] [Google Scholar]
- 49.Anand PK, Malireddi RK, Kanneganti TD. 2011. Role of the nlrp3 inflammasome in microbial infection. Front Microbiol 2:12. 10.3389/fmicb.2011.00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Coll RC, Schroder K, Pelegrin P. 2022. NLRP3 and pyroptosis blockers for treating inflammatory diseases. Trends Pharmacol Sci 43:653–668. 10.1016/j.tips.2022.04.003. [DOI] [PubMed] [Google Scholar]
- 51.Zhao C, Zhao W. 2020. NLRP3 inflammasome: a key player in antiviral responses. Front Immunol 11:211. 10.3389/fimmu.2020.00211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mursalin MH, Coburn PS, Miller FC, Livingston ET, Astley R, Callegan MC. 2021. C-X-C chemokines influence intraocular inflammation during bacillus endophthalmitis. Invest Ophthalmol Vis Sci 62:14. 10.1167/iovs.62.14.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bian F, Xiao Y, Zaheer M, Volpe EA, Pflugfelder SC, Li DQ, de Paiva CS. 2017. Inhibition of NLRP3 inflammasome pathway by butyrate improves corneal wound healing in corneal alkali burn. Int J Mol Sci 18:562. 10.3390/ijms18030562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Youm YH, Nguyen KY, Grant RW, Goldberg EL, Bodogai M, Kim D, D’Agostino D, Planavsky N, Lupfer C, Kanneganti TD, Kang S, Horvath TL, Fahmy TM, Crawford PA, Biragyn A, Alnemri E, Dixit VD. 2015. The ketone metabolite beta-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat Med 21:263–269. 10.1038/nm.3804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Schauber J, Svanholm C, Termen S, Iffland K, Menzel T, Scheppach W, Melcher R, Agerberth B, Luhrs H, Gudmundsson GH. 2003. Expression of the cathelicidin LL-37 is modulated by short chain fatty acids in colonocytes: relevance of signalling pathways. Gut 52:735–741. 10.1136/gut.52.5.735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Huang J, Brumell JH. 2014. Bacteria-autophagy interplay: a battle for survival. Nat Rev Microbiol 12:101–114. 10.1038/nrmicro3160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Siqueira MDS, Ribeiro RM, Travassos LH. 2018. Autophagy and its interaction with intracellular bacterial pathogens. Front Immunol 9:935. 10.3389/fimmu.2018.00935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cui L, Zhao H, Yin Y, Liang C, Mao X, Liu Y, Yu Q, Li M. 2019. Function of Atg11 in non-selective autophagy and selective autophagy of Candida albicans. Biochem Biophys Res Commun 516:1152–1158. 10.1016/j.bbrc.2019.06.148. [DOI] [PubMed] [Google Scholar]
- 59.Awuh JA, Flo TH. 2017. Molecular basis of mycobacterial survival in macrophages. Cell Mol Life Sci 74:1625–1648. 10.1007/s00018-016-2422-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Leymarie O, Lepont L, Berlioz-Torrent C. 2017. Canonical and non-canonical autophagy in HIV-1 replication cycle. Viruses 9:270. 10.3390/v9100270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Francis R, Singh PK, Singh S, Giri S, Kumar A. 2020. Glycolytic inhibitor 2-deoxyglucose suppresses inflammatory response in innate immune cells and experimental staphylococcal endophthalmitis. Exp Eye Res 197:108079. 10.1016/j.exer.2020.108079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Peng L, Li ZR, Green RS, Holzman IR, Lin J. 2009. Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. J Nutr 139:1619–1625. 10.3945/jn.109.104638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yan H, Ajuwon KM. 2015. Mechanism of butyrate stimulation of triglyceride storage and adipokine expression during adipogenic differentiation of porcine stromovascular cells. PLoS One 10:e0145940. 10.1371/journal.pone.0145940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Singh PK, Donovan DM, Kumar A. 2014. Intravitreal injection of the chimeric phage endolysin Ply187 protects mice from Staphylococcus aureus endophthalmitis. Antimicrob Agents Chemother 58:4621–4629. 10.1128/AAC.00126-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Swamydas M, Lionakis MS. 2013. Isolation, purification and labeling of mouse bone marrow neutrophils for functional studies and adoptive transfer experiments. J Vis Exp 10:e50586. 10.3791/50586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Livak KJ, Schmittgen TD. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408. 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 67.Becker SC, Dong S, Baker JR, Foster-Frey J, Pritchard DG, Donovan DM. 2009. LysK CHAP endopeptidase domain is required for lysis of live staphylococcal cells. FEMS Microbiol Lett 294:52–60. 10.1111/j.1574-6968.2009.01541.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Fig. S1 to S5. Download iai.00252-22-s0001.pdf, PDF file, 0.8 MB (815.8KB, pdf)