Abstract
Objective
To determine the association of quantitative infrapatellar fat pad (IPFP) signal intensity alteration with knee osteoarthritis (OA) progression.
Method
This study was performed based on the Foundation for the National Institutes of Health OA Biomarkers Consortium study, a nested case–control study consisting of 600 participants. The IPFP signal intensity alterations were quantitatively measured at baseline, 12 months and 24 months. The associations of baseline and time-integrated values over 12 and 24 months of IPFP signal intensity measures with knee OA progression over 48 months were evaluated with adjustment for baseline confounders.
Results
The baseline level of clustering effect of high signal intensity (Clustering factor (H)) was predictive of clinically relevant progression (both radiographic and pain progression) (OR 1.22). The time-integrated values of all IPFP signal intensity measures, except for mean value of IPFP signal intensity (Mean (IPFP)) over 24 months (ORs ranging from 1.23 to 1.39) as well was all except for Mean (IPFP) and mean value of IPFP high signal intensity (Mean (H)) over 12 months (ORs ranging from 1.20 to 1.31), were positively associated with clinically relevant progression. When the associations of quantitative IPFP signal intensity measures with radiographic and pain progression were examined separately, more IPFP signal intensity measures with stronger effect sizes were associated with radiographic progression compared with pain progression.
Conclusion
The associations of short-term alteration in quantitative IPFP signal intensity measures with long-term knee OA progression suggest that these measures might serve as efficacy of intervention biomarkers of knee OA.
Keywords: osteoarthritis, knee; magnetic resonance imaging; epidemiology
WHAT IS ALREADY KNOWN ON THIS TOPIC
Based on the semi-automated quantitative method developed by our group to measure infrapatellar fat pad (IPFP) signal intensity alteration with multiple continuous measures sensitive to change, we previously demonstrated that baseline quantitative IPFP signal intensity measures could predict the occurrence of radiographic knee osteoarthritis (OA), knee osteoarthritic structural progression and total knee replacement.
WHAT THIS STUDY ADDS
Our present study demonstrated that time-integrated values over 12 and 24 months of quantitative IPFP signal intensity alteration were associated with knee OA progression over a 4-year follow-up.
More IPFP signal intensity measures with stronger effect sizes were associated with radiographic progression compared with pain progression.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
Quantitative IPFP signal intensity measures may serve as surrogate efficacy measures of interventions in knee OA clinical trials.
Introduction
As the most prevalent joint disease and a leading cause of chronic pain and disability among elderly population, no disease-modifying drug for osteoarthritis (OA) has been approved.1 One of the many barriers to the successful development of OA therapeutic drugs is the lack of valid and responsive biomarkers to ascertain efficacy.2 In order to facilitate the translational success of anti-OA therapies, we urgently need to identify biomarkers showing short-term alteration associated with long-term clinically relevant outcomes, which hold the potential to serve as surrogate clinical trial endpoints to evaluate the efficacy of interventions for OA.
Recently, a local adipose tissue structure in the knee joint, infrapatellar fat pad (IPFP), has become a focus of increasing interest due to its strong involvement in knee OA.3 4 IPFP is a major source of pro-inflammatory mediators, including cytokines, chemokines and adipokines as well as inflammatory lipid mediators, and the recognition of the involvement of these mediators in the knee joint tissue haemostasis has resulted in considerable attention to IPFP in the field of knee OA research.5–15 The inflammation status of IPFP could be assessed based on signal alterations on non-contrast-enhanced fat-suppressed MRI,3 4 and the high signal alterations within IPFP assessed semi-quantitatively has long been used as a surrogate for synovitis, which has shown associations with knee structural16–20 and symptomatic abnormalities.21–23 However, in view of the shortcomings of semi-quantitative methods such as output insensitive to change, being time-consuming and with low reproducibility, our group has developed an efficient and reproducible method to measure the IPFP signal alteration quantitatively with acceptable concurrent and clinical construct validity.24 Applying this method, we previously demonstrated that baseline quantitative IPFP signal intensity measures could predict the occurrence of radiographic knee OA,25 knee osteoarthritic structural progression26 and total knee replacement (TKR).27 Moreover, our recent study found that quantitative IPFP signal intensity measures were longitudinally associated with biochemical biomarkers reflecting knee joint tissue turnover in knee OA.28 Nevertheless, the association of short-term change in quantitative IPFP signal measures with long-term knee OA progression, especially clinically relevant progression, and the predictive validity of quantitative IPFP signal measures in pain progression has not been examined previously. Based on the Foundation for the National Institutes of Health (FNIH) OA Biomarkers Consortium study, we aimed to investigate the associations of baseline levels and time-integrated values over 12 and 24 months of quantitative IPFP signal intensity alteration with knee OA progression.
Methods
Study design
This study was performed based on the data of 600 participants (one index knee per subject) in FNIH study, a nested case–control study within the Osteoarthritis Initiative (OAI). The details of the FNIH study design were published elsewhere.2 Participants eligible for the FNIH study were those with at least one knee with a Kellgren/Lawrence (K/L) grade of 1–3 at baseline and with knee radiographs, knee MRI, biological specimens and clinical data available at baseline and 24 months. Briefly, the FNIH study consisted of four mutually exclusive groups of participants based on the radiographic and/or pain progression status in an index knee. Radiographic progression was defined as a decrease of ≥0.7 mm in minimum joint space width (minJSW) of medial femorotibial compartment from baseline to 24, 36 or 48 months. Pain progression was defined as a Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain increase of ≥9 points at two or more time points from 24 to 60 months. The following subjects were excluded from the FNIH study: subjects having a total knee or hip replacement or metal implants in bone from baseline to 24 months; knees unable to meet criteria for outcome progression due to ceiling effects at baseline (minJSW<1.0 mm and/or WOMAC pain >91 on 0–100 scale); knees having poor and/or inconsistent positioning on knee radiographs at one or more visits; knees with predominantly lateral compartment joint space narrowing at baseline or during follow-up; knees without enough follow-up time points to determine persistent pain progression; subjects with a knee that already met the criteria for radiographic and pain progression at 12 months; subjects with outcomes inconsistent between knees.
For better covariate balance, the four groups of knees were frequency matched based on K/L grade strata (grade 1, 2, 3) and BMI strata (<25, 25 to <27.5, 27.5 to <30, 30 to <35 and ≥35 kg/m2). Finally, four groups of knees were included: group 1 as the primary case with clinically relevant progression (knee with both radiographic and pain progression, n=194), and the comparator knees lacking the combination of radiographic and pain progression consisted of group 2 (knees with radiographic progression but not pain progression, n=103), group 3 (knees with pain progression but not radiographic progression, n=103) and group 4 (knees with neither radiographic progression nor pain progression, n=200).
Measurements of IPFP signal intensity
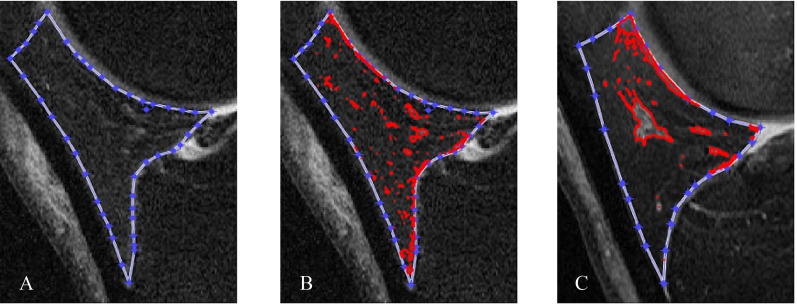
All IPFP measurements were performed by investigators blinded to all clinical characteristics, including progression status. Our semi-automatic method was applied to quantitatively measure the IPFP signal intensity alteration on the three time points (baseline, 12 months and 24 months) based on sagittal planes of fat-saturated T2-weighted images acquired from 3.0T MRI, which were obtained by four identical Siemens 3T MRI scanners at each clinic visit. The details of this new algorithm were introduced in detail previously.24 Briefly, an initial lasso consisting of a series of points was created manually close to the outer contour of IPFP and then it contracted inward to approach the actual boundary of IPFP automatically (figure 1A). The new algorithm was designed to segment IPFP semi-automatically, insensitive to noise, easy to distinguish fake edges from real edges and more accurately identify the IPFP boundary. The high signal intensity regions were captured automatically based on a method to examine neighbouring pixels of initial seed points and to determine whether the pixel neighbours should be added to the region (figure 1B). The following IPFP signal intensity measures were calculated and output automatically: the mean value (Mean (IPFP)) and SD (sDev (IPFP)) of IPFP signal intensity, mean value (Mean (H)) and SD (sDev (H)) of IPFP high signal intensity, median value (Median (H)) and upper quartile value (UQ (H)) of high signal intensity, and the ratio of volume of high signal intensity to volume of whole IPFP (Percentage (H)) and the clustering effect of high signal intensity (Clustering factor (H)). Among these measures, Mean (IPFP) and sDev (IPFP) reflect the average level and heterogeneity of the whole IPFP signal intensity, and Mean (H) and sDev (H) denote the average level and heterogeneity of high signal intensity, respectively. Median (H) and UQ (H) are the median value and upper quantile value of high signal intensity, respectively. The volume of high signal intensity was calculated according to the slice thickness and the area on each slice, and Percentage (H) represents the adjusted quantity of high signal regions. The clustering regions with high signal intensity in the IPFP varied in participants, which might possess different clinical significance. Therefore, Clustering factor (H) was used to reflect this clustering effect. The bigger the clustering effects, the greater aggregation of the high signal intensity (figure 1C). Collectively, these measures could be classified into four categories: signal alteration of the whole IPFP, high signal intensity alteration of IPFP, adjusted quantity and clustering effect of high signal intensity. The intraclass correlation coefficients and interobserver correlation coefficients for all measures are high (>0.90).24
Figure 1.
Segmentation and signal intensity calculation of the whole infrapatellar fat pad and high signal intensity regions on sagittal planes of fat-saturated T2-weighted images using MATLAB.
Quantitative and qualitative variables were presented as mean±SD and frequencies (percentages), respectively. Student’s t-test and χ2 test were employed to compare the differences in means and percentages when appropriate. Following the data analysis methods established by the FNIH study 2, our primary analysis focused on the comparison between primary case in group 1 and all comparators of the other three groups combined together. For the secondary analysis, four methods were applied, and method 1 compared each of groups 1, 2 and 3 with group 4 separately. Method 2 compared all progressors (groups 1, 2, 3 combined) with non-progressors (group 4). Method 3 compared all radiographic progressors (groups 1 and 2 combined) with no radiographic progressors (groups 3 and 4 combined). Method 4 compared all pain progressors (groups 1 and 3 combined) with no pain progressors (groups 2 and 4 combined).
Statistical analysis
The associations of baseline levels and time-integrated values over 12 and 24 months of IPFP signal intensity measures with knee OA progression were examined by logistic regression with the exception of method 1 in secondary analysis, which was performed by multinomial logistic regression with generalised logits, with all data analysis methods adjusted for baseline age, sex, body mass index (BMI), race, K/L grade, WOMAC pain score, minJSW and pain medication use. Time-integrated values were selected over absolute change owing to its significance of providing the longitudinal information of the focused biomarkers, and these values are equal to the area under the curve defined by the individual values for the specific time interval.29 The baseline and time-integrated values over 12 and 24 months of IPFP signal intensity measures were transposed to z values (created by subtracting the original value from the mean and divided by the SD). Associations were expressed as the increase in odds of being a progressor knee for each 1 SD increases.
All statistical analysis was carried out using Stata V.15.0 for Windows (StataCorp.), and a two-tailed p value less than 0.05 was considered statistically significant.
Results
The baseline characteristics are shown in table 1, with detailed description of baseline characteristics of participants in each group shown in online supplemental table 1. The participants were aged between 45 and 79 years (mean±SD, 61.55±8.88 years) and 353 (58.83%) were female. The primary cases and all comparators were well matched for the baseline characteristics except for K/L grade and WOMAC pain score. The proportion of participants with baseline K/L grade 3 was relatively higher, while the baseline WOMAC pain score was relatively lower in primary cases compared with all other groups.
Table 1.
Baseline characteristics and infrapatellar fat pad signal intensity measures of participants*
| Variable | Primary cases (n=194) | All comparators (n=406) |
| Age, years | 62.04±8.80 | 61.31±8.92 |
| Female, number (%) | 110 (56.70) | 243 (59.85) |
| BMI, kg/m2 | 30.73±4.77 | 30.71±4.79 |
| Race, white, % | 155 (79.90) | 320 (78.82) |
| K/L grade 1/2/3, number (%) | 24/84/86 (12.37/43.30/44.33) | 51/222/133 (12.56/54.68/32.76) |
| WOMAC pain score | 10.15±12.98 | 13.00±16.66 |
| Pain medication use, number (%) | 63 (32.47) | 114 (28.08) |
| Minimum medial radiographic JSW, mm | 3.79±1.39 | 3.85±1.06 |
| Mean (IPFP) | 0.19±0.04 | 0.19±0.04 |
| sDev (IPFP) | 0.09±0.02 | 0.09±0.02 |
| Mean (H) | 0.36±0.06 | 0.36±0.06 |
| sDev (H) | 0.09±0.01 | 0.09±0.02 |
| Median (H) | 0.35±0.06 | 0.34±0.06 |
| UQ (H) | 0.41±0.06 | 0.40±0.07 |
| Percentage (H) | 0.13±0.01 | 0.13±0.01 |
| Clustering factor (H) | 0.74±0.05 | 0.73±0.05 |
*Values are the mean±SD except for numbers and percentages. Primary cases were knees in group 1, namely the knees with both radiographic and pain progression. All comparators consisted of group 2 (knees with radiographic progression but not pain progression), group 3 (knees with pain progression but not radiographic progression) and group 4 (knees with neither radiographic progression nor pain progression).
BMI, body mass index; JSW, joint space width; K/L, Kellgren/Lawrence; OA, osteoarthritis; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
rmdopen-2022-002565supp001.pdf (86.4KB, pdf)
Primary analysis
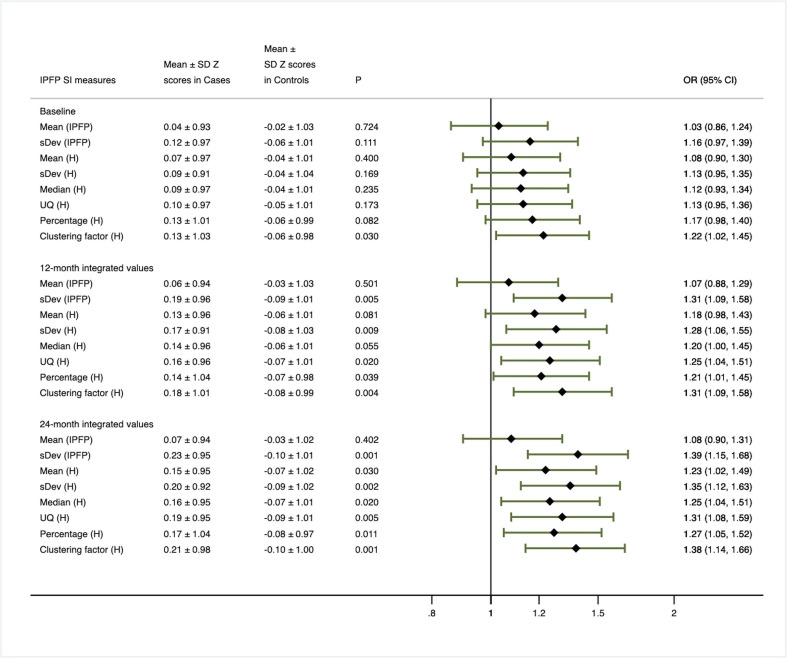
A total of 600, 582 and 600 MRI of suitable quality for IPFP signal intensity measurement were available at baseline, 12 months and 24 months, respectively. The associations of baseline levels, 12-month and 24-month time-integrated values of IPFP signal measures with the combination of radiographic and pain progression are shown in figure 2.
Figure 2.
Primary analysis based on baseline, 12-month and 24-month integrated value of infrapatellar fat pad signal intensity measure Z scores.
When the baseline levels of IPFP signal measures were compared between primary cases and all comparators, only Clustering factor (H) was found to be positively associated with primary case status. Over 12 months, the time-integrated values for all IPFP signal measures except for Mean (IPFP) and Mean (H) were positively associated with primary case status with the ORs ranging from 1.20 to 1.31. Over 24 months, the time-integrated values for all IPFP signal measures with the exception of Mean (IPFP) were significantly and positively associated with primary case status with the ORs ranging from 1.23 to 1.39, with the strength of association being much stronger than that of 12 months.
Secondary analysis
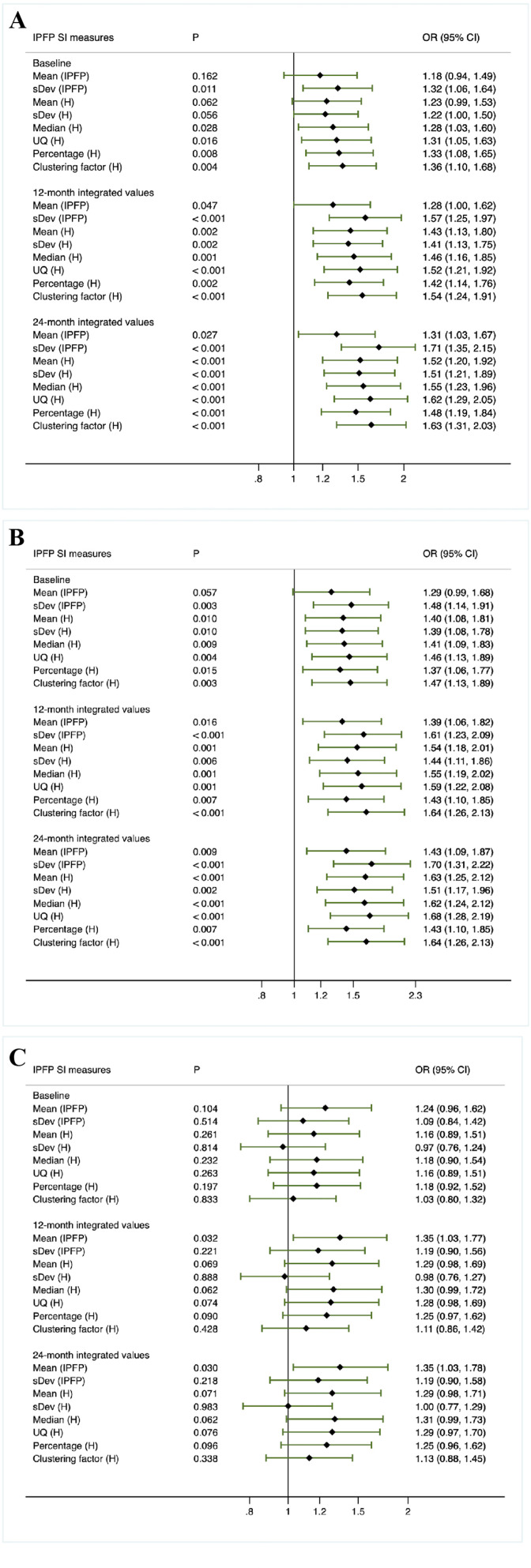
When knees with neither radiographic nor pain progression (group 4) were employed as the control group to determine the associations of IPFP signal measures with both radiographic and pain progression (group 4 vs group 1), the baseline levels of sDev (IPFP), Median (H), UQ (H), Percentage (H) and Clustering factor (H) were significantly and positively associated with both radiographic and pain progression, with the ORs ranging from 1.28 to 1.36. Moreover, the 12-month and 24-month time-integrated values of all IPFP signal measures were significantly and positively associated with both radiographic and pain progression (figure 3A), with the OR values being greater than that in figure 2.
Figure 3.
Secondary analysis based on baseline, 12-month and 24-month integrated value of infrapatellar fat pad signal intensity measures Z scores (method 1).
When the associations of IPFP signal measures with any progression were evaluated (figure 4A, method 2), significant evidence was found for the baseline levels of all IPFP measures except for Mean (IPFP) and sDev (H), with the ORs varying from 1.25 to 1.30. Furthermore, significant evidence was detected for the time-integrated values of all IPFP signal measures over 12 months (ORs varied from 1.28 to 1.47) and 24 months (ORs varied from 1.34 to 1.55).
Figure 4.
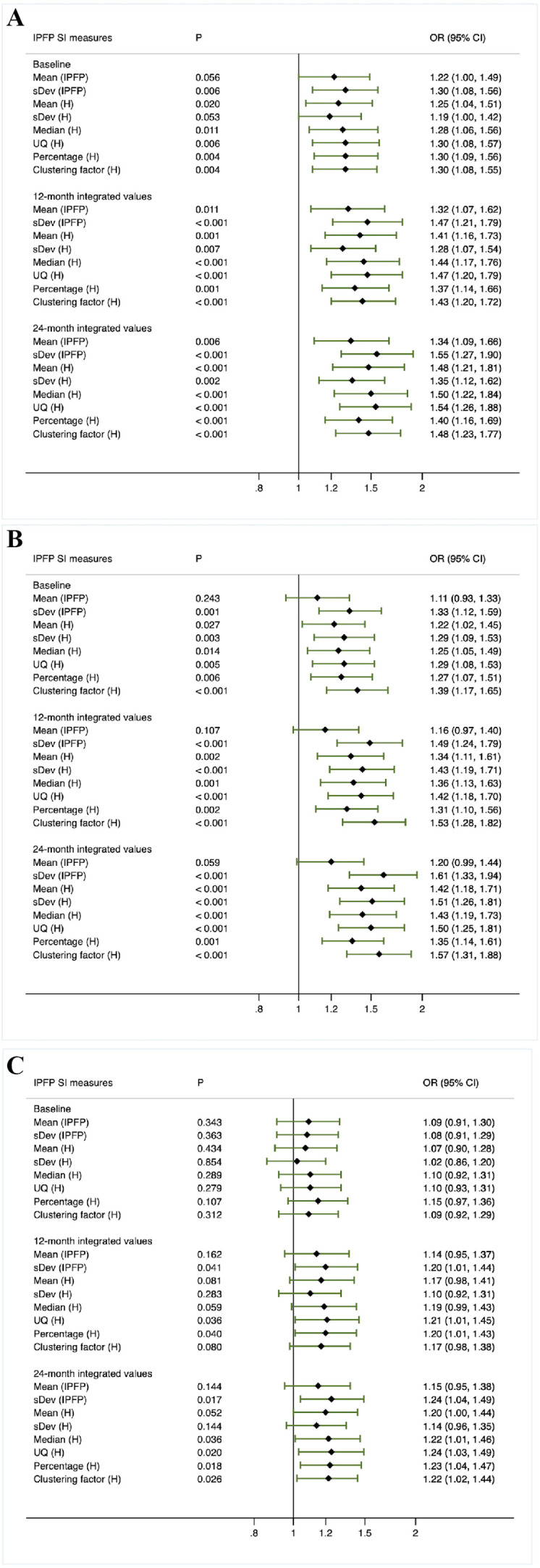
Secondary analysis based on baseline, 12-month and 24-month integrated value of infrapatellar fat pad signal intensity measures Z scores (method 2–4).
When knees with radiographic progression (group 1 and 2 combined) were compared with knees without radiographic progression (group 3 and 4 combined) (figure 4B, method 3), the baseline levels, 12-month and 24-month time-integrated values of all IPFP signal measures except for Mean (IPFP) were found to be significantly different, with the ORs falling into the range of 1.22–1.39, 1.31–1.53 and 1.35–1.61, respectively. Similarly, results derived from the comparison made between group 2 and group 4 indicated that the baseline levels of all but Mean (IPFP), 12-month and 24-month time-integrated values of all IPFP measures were significantly and positively associated with radiographic progression (figure 3B). When knees with pain progression (group 1 and 3 combined) were compared with knees without pain progression (group 2 and 4 combined) (figure 4C, method 4), there was a significant difference in 12-month time-integrated values of sDev (IPFP), UQ (H) and Percentage (H) (ORs varied from 1.20 to 1.21), and 24-month time-integrated values of sDev (IPFP), Median (H), UQ (H), Percentage (H) and Clustering factor (H) (ORs varied from 1.22 to 1.24).
Discussion
To our knowledge, this is the first study focusing on the association of short-term alteration in quantitative IPFP signal measures with knee OA progression and the predictive ability of quantitative IPFP signal measures for pain progression. We found that the time-integrated values of all IPFP signal intensity measures except for Mean (IPFP) over 24 months as well as all IPFP signal intensity measures except for Mean (IPFP) and Mean (H) over 12 months were positively associated with clinically relevant progression. Besides, more IPFP signal intensity measures were associated with radiographic progression showing greater effect sizes compared with pain progression.
With the main purpose of examining the potential of quantitative IPFP signal intensity measures as candidate surrogate endpoints to evaluate the efficacy of knee OA interventions in clinical trials, we determined to analyse the relationships between alteration of quantitative IPFP signal intensity measures over 24 months and clinical relevant progression of knee OA over 48 months based on the data of FNIH study. In addition to finding that baseline levels of Clustering factor (H) was predictive of primary case status, the time-integrated values of all IPFP measures except for Mean (IPFP) over 24 months were positively associated with clinically relevant progression of knee OA. Besides, similar results were also observed for the time-integrated values of IPFP measures over 12 months. Thus, the near-term alteration of IPFP signal intensity were associated with long-term clinically relevant progression significantly, suggesting that the quantitative IPFP signal alteration measures might serve as candidate surrogate endpoints to evaluate the efficacy of knee OA interventions in clinical trials. It is worth mentioning that Collins et al reported that worsening in Hoffa-synovitis over 24 months was also associated with an increased risk of developing both radiographic and pain progression.30 Different from that study, our study used quantitative method to measure IPFP signal alteration with detailed measures sensitive to change, which could be ideal surrogate endpoints.
During the past decade, multiple studies have investigated the role of IPFP signal alteration evaluated by semi-quantitative method in regard to knee structural and symptomatic changes separately with inconsistent results. Commonly, IPFP signal alteration is semi-quantitatively scored from 0 to 3, with a score of ≥1 defined as Hoffa-synovitis.31 Several studies demonstrated that baseline Hoffa-synovitis was associated with elevated risk of radiographic knee OA development,16 17 knee replacement18 and accelerated knee OA occurrence.19 20 Our previous study showed that the baseline semi-quantitatively measured IPFP high signal intensity was positively associated with increase in tibiofemoral cartilage defects over 2.6 years among older adults.32 In contrast, Roemer et al reported that baseline synovitis was not associated with increased risk of cartilage loss.33 Similarly, another prospective study showed that change in synovitis scored semi-quantitatively was not associated with cartilage loss.21 The discrepancy could be attributed to differences in the definition of IPFP signal alteration, sample size and the length of follow-up. Using the OAI data, we found that the baseline quantitative IPFP signal measures could predict the incidence of radiographic OA25 and TKR.27 Moreover, we have analysed the relationships between four quantitative IPFP signal measures (sDev (IPFP), UQ (H), Percentage (H) and Clustering factor (H)) and changes in knee osteoarthritic structural abnormalities, and found that baseline levels of sDev (IPFP), UQ (H) and Clustering factor (H) were positively associated with an increase in tibiofemoral cartilage defects and tibial cartilage loss over 2 years.26 Consistent with this study, we found that the baseline levels as well as time-integrated values over 12 and 24 months of all quantitative IPFP signal intensity measures except for Mean (IPFP) were positively associated with radiographic progression. The underlying mechanism of IPFP signal alteration in radiographic progression could be due to the pro-inflammatory mediators secreted by IPFP and synovium, which is involved in the catabolism of cartilage and other knee joint tissues.4 34 To date, a few studies have examined the relation of IPFP signal alteration to knee pain. Hill et al reported that the change in synovitis assessed in IPFP was strongly associated with change in pain as evaluated using a visual analogue scale.21 Zhang et al reported that changes in synovitis, scored semi-quantitatively in the infrapatellar and intercondylar regions of IPFP, were associated with the fluctuation of frequent knee pain and pain severity.22 A cross-sectional study found that Hoffa-synovitis and the severity of inflammation within IPFP was significantly associated with the Knee injury and Osteoarthritis Outcome Score pain.23 Nonetheless, Crema et al showed that the maximum score of signal alteration within the infrapatellar and intercondylar regions of IPFP was not associated with pain on walking up or down stairs.35 Steidle-Kloc et al reported that the signal intensity and heterogeneity of IPFP was not associated with knee pain.36 The controversy might result from the difference in the IPFP signal alteration and pain measurement methods. Our previous study found that the baseline IPFP high signal intensity assessed semi-quantitatively was positively associated with increases in knee pain when going upstairs/downstairs.32 However, the relationships between quantitative IPFP signal intensity measures and knee pain have not been examined previously. In the present study, we found that 12-month time-integrated values of sDev (IPFP), UQ (H) and Percentage (H) and 24-month time-integrated values of sDev (IPFP), Median (H), UQ (H), Percentage (H) and Clustering factor (H) were positively associated with pain progression. This is consistent with a recent review highlighting the contribution of the inflammation of IPFP and synovium to peripheral and central sensitisation, and the possible involved mediators include neuropeptides and peptide hormones, growth factors and cytokines.37
The main strength of our present study is that the novel quantitative approach was applied to measure IPFP signal alteration with continuous measures. However, potential limitations should be noted. First, the time period of the measurement of IPFP signal alteration from baseline to 24 months overlapped with that of the definition of knee OA progression which was determined from baseline to 24–48 months, thus the 24-month time-integrated values should be interpreted with both predictive validity and concurrent validity. In addition to finding prognostic biomarkers, identifying the efficacy of intervention biomarkers is also the main purpose of FNIH study, thus the association of 24-month time-integrated values with clinically relevant progression support the use of quantitative IPFP signal alteration measures as surrogate efficacy measures for knee OA interventions. Second, variations in MRI machine and sequence would impact the measured IPFP SI values over the study period. However, four identical Siemens 3T MRI scanners were applied to acquire state-of-the-art MRI of knees in all OAI participants at each clinic visit, so our IPFP SI measures at three visits were not affected by MRI machine and sequence. Third, there is no direct comparison between IPFP signal alteration and histological examination results, the pathological significance of IPFP signal alteration remains to be determined. Notably, one recent study has indicated inflammatory pathogenesis of high signal intensity within IPFP existed only in knee OA.38 Finally, the current study mainly focused on high signal intensity, whereas IPFP low signal intensity might also be implicated in the knee OA progression.39 Thus, future studies with low signal intensity measures should be taken into account.
Taken together, the time-integrated values of IPFP signal intensity measures except for Mean (IPFP) over 24 months as well as all IPFP signal intensity measures except for Mean (IPFP) and Mean (H) over 12 months were positively associated with clinically relevant progression, indicating that the associations of short-term alteration in quantitative IPFP signal measures with long-term knee OA progression suggest that these measures might serve as efficacy of intervention biomarkers of knee OA.
Acknowledgments
We thank the Osteoarthritis Research Society International (OARSI) for their leadership and expertise on the FNIH project. Data provided from the FNIH OA Biomarkers Consortium Project made possible through grants and direct or in-kind contributions by: AbbVie; Amgen; Arthritis Foundation; Artialis; Bioiberica; BioVendor; DePuy; Flexion Therapeutics; GSK; IBEX; IDS; Merck Serono; Quidel; Rottapharm | Madaus; Sanofi; Stryker; the Pivotal OAI MRI Analyses (POMA) study, NIH HHSN2682010000 21C; and the Osteoarthritis Research Society International. The OAI is a public–private partnership comprised of five contracts (N01-AR-2-2258; N01-AR-2-2259; N01-AR-2-2260; N01-AR-2-2261; N01-AR-2-2262) funded by the National Institutes of Health. Funding partners include Merck Research Laboratories; Novartis Pharmaceuticals Corporation, GlaxoSmithKline; and Pfizer, Inc. Private sector funding for the Consortium and OAI is managed by the Foundation for the National Institutes of Health.
Footnotes
HC and QY contributed equally.
Contributors: CD had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Conception and design: HC, QY and CD. Acquisition of data: HC, QY, TM, ZC, DH and CD. Analysis and interpretation of data: HC, QY and CD. All authors were involved in drafting the article or revising it critically for important intellectual content, and all authors approved the final version to be submitted for publication.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: DH provides consulting advice on scientific advisory boards for Pfizer, Lilly, TLCBio, Novartis, Tissuegene, Biobone. All other authors declare no other competing interests.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study involves human participants and was approved by the Foundation for the National Institutes of Health (FNIH) OA Biomarkers Consortium study is a nested case–control study within the Osteoarthritis Initiative (OAI). The OAI study has received ethics board approval by the institutional review board at the University of California, San Francisco (OAI Coordinating Center; Approval Number: 10-00532), and written informed consent was obtained from all participants. Participants gave informed consent to participate in the study before taking part.
References
- 1.Hunter DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet 2019;393:1745–59. 10.1016/S0140-6736(19)30417-9 [DOI] [PubMed] [Google Scholar]
- 2.Hunter DJ, Nevitt M, Losina E, et al. Biomarkers for osteoarthritis: current position and steps towards further validation. Best Pract Res Clin Rheumatol 2014;28:61–71. 10.1016/j.berh.2014.01.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chang J, Liao Z, Lu M, et al. Systemic and local adipose tissue in knee osteoarthritis. Osteoarthritis Cartilage 2018;26:864–71. 10.1016/j.joca.2018.03.004 [DOI] [PubMed] [Google Scholar]
- 4.Ioan-Facsinay A, Kloppenburg M. An emerging player in knee osteoarthritis: the infrapatellar fat pad. Arthritis Res Ther 2013;15:225. 10.1186/ar4422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ushiyama T, Chano T, Inoue K, et al. Cytokine production in the infrapatellar fat pad: another source of cytokines in knee synovial fluids. Ann Rheum Dis 2003;62:108–12. 10.1136/ard.62.2.108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Presle N, Pottie P, Dumond H, et al. Differential distribution of adipokines between serum and synovial fluid in patients with osteoarthritis. contribution of joint tissues to their articular production. Osteoarthritis Cartilage 2006;14:690–5. 10.1016/j.joca.2006.01.009 [DOI] [PubMed] [Google Scholar]
- 7.Distel E, Cadoudal T, Durant S, et al. The infrapatellar fat pad in knee osteoarthritis: an important source of interleukin-6 and its soluble receptor. Arthritis Rheum 2009;60:3374–7. 10.1002/art.24881 [DOI] [PubMed] [Google Scholar]
- 8.Chen W-P, Bao J-P, Feng J, et al. Increased serum concentrations of visfatin and its production by different joint tissues in patients with osteoarthritis. Clin Chem Lab Med 2010;48:1141–5. 10.1515/CCLM.2010.230 [DOI] [PubMed] [Google Scholar]
- 9.Klein-Wieringa IR, Kloppenburg M, Bastiaansen-Jenniskens YM, et al. The infrapatellar fat pad of patients with osteoarthritis has an inflammatory phenotype. Ann Rheum Dis 2011;70:851–7. 10.1136/ard.2010.140046 [DOI] [PubMed] [Google Scholar]
- 10.Clockaerts S, Bastiaansen-Jenniskens YM, Feijt C, et al. Cytokine production by infrapatellar fat pad can be stimulated by interleukin 1β and inhibited by peroxisome proliferator activated receptor α agonist. Ann Rheum Dis 2012;71:1012–8. 10.1136/annrheumdis-2011-200688 [DOI] [PubMed] [Google Scholar]
- 11.Conde J, Scotece M, López V, et al. Differential expression of adipokines in infrapatellar fat pad (IPFP) and synovium of osteoarthritis patients and healthy individuals. Ann Rheum Dis 2014;73:631–3. 10.1136/annrheumdis-2013-204189 [DOI] [PubMed] [Google Scholar]
- 12.Gross J-B, Guillaume C, Gegout-Pottie P, et al. The infrapatellar fat pad induces inflammatory and degradative effects in articular cells but not through leptin or adiponectin. Clin Exp Rheumatol 2017;35:53–60. [PubMed] [Google Scholar]
- 13.Eymard F, Pigenet A, Citadelle D, et al. Knee and hip intra-articular adipose tissues (IAATs) compared with autologous subcutaneous adipose tissue: a specific phenotype for a central player in osteoarthritis. Ann Rheum Dis 2017;76:1142–8. 10.1136/annrheumdis-2016-210478 [DOI] [PubMed] [Google Scholar]
- 14.Gierman LM, Wopereis S, van El B, et al. Metabolic profiling reveals differences in concentrations of oxylipins and fatty acids secreted by the infrapatellar fat pad of donors with end-stage osteoarthritis and normal donors. Arthritis Rheum 2013;65:n/a–2614. 10.1002/art.38081 [DOI] [PubMed] [Google Scholar]
- 15.Mustonen A-M, Käkelä R, Lehenkari P, et al. Distinct fatty acid signatures in infrapatellar fat pad and synovial fluid of patients with osteoarthritis versus rheumatoid arthritis. Arthritis Res Ther 2019;21:124. 10.1186/s13075-019-1914-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Roemer FW, Kwoh CK, Hannon MJ, et al. What comes first? Multitissue involvement leading to radiographic osteoarthritis: magnetic resonance imaging-based trajectory analysis over four years in the osteoarthritis initiative. Arthritis Rheumatol 2015;67:2085–96. 10.1002/art.39176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Atukorala I, Kwoh CK, Guermazi A, et al. Synovitis in knee osteoarthritis: a precursor of disease? Ann Rheum Dis 2016;75:390–5. 10.1136/annrheumdis-2014-205894 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roemer FW, Kwoh CK, Hannon MJ, et al. Can structural joint damage measured with MR imaging be used to predict knee replacement in the following year? Radiology 2015;274:810–20. 10.1148/radiol.14140991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Harkey MS, Davis JE, Lu B, et al. Early pre-radiographic structural pathology precedes the onset of accelerated knee osteoarthritis. BMC Musculoskelet Disord 2019;20:241. 10.1186/s12891-019-2624-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Davis JE, Ward RJ, MacKay JW, et al. Effusion-synovitis and infrapatellar fat pad signal intensity alteration differentiate accelerated knee osteoarthritis. Rheumatology 2019;58:418–26. 10.1093/rheumatology/key305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hill CL, Hunter DJ, Niu J, et al. Synovitis detected on magnetic resonance imaging and its relation to pain and cartilage loss in knee osteoarthritis. Ann Rheum Dis 2007;66:1599–603. 10.1136/ard.2006.067470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang Y, Nevitt M, Niu J, et al. Fluctuation of knee pain and changes in bone marrow lesions, effusions, and synovitis on magnetic resonance imaging. Arthritis Rheum 2011;63:691–9. 10.1002/art.30148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ballegaard C, Riis RGC, Bliddal H, et al. Knee pain and inflammation in the infrapatellar fat pad estimated by conventional and dynamic contrast-enhanced magnetic resonance imaging in obese patients with osteoarthritis: a cross-sectional study. Osteoarthritis Cartilage 2014;22:933–40. 10.1016/j.joca.2014.04.018 [DOI] [PubMed] [Google Scholar]
- 24.Lu M, Chen Z, Han W, et al. A novel method for assessing signal intensity within infrapatellar fat pad on MR images in patients with knee osteoarthritis. Osteoarthritis Cartilage 2016;24:1883–9. 10.1016/j.joca.2016.06.008 [DOI] [PubMed] [Google Scholar]
- 25.Wang K, Ding C, Hannon MJ, et al. Quantitative signal intensity alteration in infrapatellar fat pad predicts incident radiographic osteoarthritis: the osteoarthritis initiative. Arthritis Care Res 2019;71:30–8. 10.1002/acr.23577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Han W, Aitken D, Zheng S, et al. Association between quantitatively measured infrapatellar fat pad high Signal-Intensity alteration and magnetic resonance Imaging-Assessed progression of knee osteoarthritis. Arthritis Care Res 2019;71:638–46. 10.1002/acr.23713 [DOI] [PubMed] [Google Scholar]
- 27.Wang K, Ding C, Hannon MJ, et al. Signal intensity alteration within infrapatellar fat pad predicts knee replacement within 5 years: data from the Osteoarthritis Initiative. Osteoarthritis Cartilage 2018;26:1345–50. 10.1016/j.joca.2018.05.015 [DOI] [PubMed] [Google Scholar]
- 28.Cen H, Yan Q, Han W, et al. Longitudinal association of infrapatellar fat pad signal intensity alteration with biochemical biomarkers in knee osteoarthritis. Rheumatology 2022;62:keac214:439–49. 10.1093/rheumatology/keac214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Matthews JN, Altman DG, Campbell MJ, et al. Analysis of serial measurements in medical research. BMJ 1990;300:230–5. 10.1136/bmj.300.6719.230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Collins JE, Losina E, Nevitt MC, et al. Semiquantitative imaging biomarkers of knee osteoarthritis progression: data from the foundation for the National Institutes of health osteoarthritis biomarkers Consortium. Arthritis Rheumatol 2016;68:2422–31. 10.1002/art.39731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hunter DJ, Guermazi A, Lo GH, et al. Evolution of semi-quantitative whole joint assessment of knee OA: MOAKS (MRI osteoarthritis knee score). Osteoarthritis Cartilage 2011;19:990–1002. 10.1016/j.joca.2011.05.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Han W, Aitken D, Zhu Z, et al. Signal intensity alteration in the infrapatellar fat pad at baseline for the prediction of knee symptoms and structure in older adults: a cohort study. Ann Rheum Dis 2016;75:1783–8. 10.1136/annrheumdis-2015-208360 [DOI] [PubMed] [Google Scholar]
- 33.Roemer FW, Guermazi A, Felson DT, et al. Presence of MRI-detected joint effusion and synovitis increases the risk of cartilage loss in knees without osteoarthritis at 30-month follow-up: the MOST study. Ann Rheum Dis 2011;70:1804–9. 10.1136/ard.2011.150243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sellam J, Berenbaum F. The role of synovitis in pathophysiology and clinical symptoms of osteoarthritis. Nat Rev Rheumatol 2010;6:625–35. 10.1038/nrrheum.2010.159 [DOI] [PubMed] [Google Scholar]
- 35.Crema MD, Felson DT, Roemer FW, et al. Peripatellar synovitis: comparison between non-contrast-enhanced and contrast-enhanced MRI and association with pain. The most study. Osteoarthritis Cartilage 2013;21:413–8. 10.1016/j.joca.2012.12.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Steidle-Kloc E, Culvenor AG, Dörrenberg J, et al. Relationship between knee pain and infrapatellar fat pad morphology: a within- and Between-Person analysis from the osteoarthritis initiative. Arthritis Care Res 2018;70:550–7. 10.1002/acr.23326 [DOI] [PubMed] [Google Scholar]
- 37.Belluzzi E, Stocco E, Pozzuoli A, et al. Contribution of infrapatellar fat pad and synovial membrane to knee osteoarthritis pain. Biomed Res Int 2019;2019:1–18. 10.1155/2019/6390182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.de Vries BA, van der Heijden RA, Poot DHJ, et al. Quantitative DCE-MRI demonstrates increased blood perfusion in Hoffa's fat pad signal abnormalities in knee osteoarthritis, but not in patellofemoral pain. Eur Radiol 2020;30:3401–8. 10.1007/s00330-020-06671-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Han W, Aitken D, Zhu Z, et al. Hypointense signals in the infrapatellar fat pad assessed by magnetic resonance imaging are associated with knee symptoms and structure in older adults: a cohort study. Arthritis Res Ther 2016;18:234. 10.1186/s13075-016-1130-y [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
rmdopen-2022-002565supp001.pdf (86.4KB, pdf)
Data Availability Statement
Data are available upon reasonable request.