Objective:
The aim of this study was to develop a classification system for pancreas-associated risk factors in pancreatoduodenectomy (PD).
Summary Background Data:
Postoperative pancreatic fistula (POPF) is the most relevant PD-associated complication. A simple standardized surgical reporting system based on pancreas-associated risk factors is lacking.
Methods:
A systematic literature search was conducted to identify studies investigating clinically relevant (CR) POPF (CR-POPF) and pancreas-associated risk factors after PD. A meta-analysis of CR-POPF rate for texture of the pancreas (soft vs not-soft) and main pancreatic duct (MPD) diameter was performed using the Mantel-Haenszel method. Based on the results, the International Study Group of Pancreatic Surgery (ISGPS) proposes the following classification: A, not-soft (hard) texture and MPD >3 mm; B, not-soft (hard) texture and MPD ≤3 mm; C, soft texture and MPD >3 mm; D, soft texture and MPD ≤3 mm. The classification was evaluated in a multi-institutional, international cohort.
Results:
Of the 2917 articles identified, 108 studies were included in the analyses. Soft pancreatic texture was significantly associated with the development of CR-POPF [odds ratio (OR) 4.24, 95% confidence interval (CI) 3.67-4.89, P < 0.01) following PD. Similarly, MPD diameter ≤3 mm significantly increased CR-POPF risk compared with >3 mm diameter MPDs (OR 3.66, 95% CI 2.62–5.12, P < 0.01). The proposed 4-stage system was confirmed in an independent cohort of 5533 patients with CR-POPF rates of 3.5%, 6.2%, 16.6%, and 23.2% for type A-D, respectively (P < 0.001).
Conclusion:
For future pancreatic surgical outcomes studies, the ISGPS recommends reporting these risk factors according to the proposed classification system for better comparability of results.
Keywords: pancreatic duct, pancreatic fistula, pancreatic texture, pancreaticoduodenectomy, pancreatoduodenectomy
Pancreatoduodenectomy (PD) is the treatment of choice for malignant and symptomatic benign disease of the pancreatic head. It offers the only potential curative option for patients with pancreatic ductal adenocarcinoma (PDAC), distal bile duct cancer, or pancreatic neuroendocrine tumors. In addition, it is the treatment of choice for a range of premalignant and benign lesions such as intraductal papillary mucinous neoplasms and chronic pancreatitis. 1
Although surgery-associated mortality after PD has decreased in specialized centers, postoperative complications are frequent and affect up to 50% of patients. 2–4 The benchmark from the International Study Group of Pancreatic Surgery (ISGPS) Evidence Map of Pancreatic Surgery 5 shows a postoperative mortality rate of 1% [99% confidence interval (CI) 0.01–0.02) in 155 randomized controlled trials (RCTs) and a clinically relevant postoperative pancreatic fistula (CR-POPF) 6,7 rate of 15% (99% CI 0.12-0.18) in 76 RCT after PD. 5
Multiple risk factors have been identified that are associated with CR-POPF development following PD. 8 These include: patient-associated risk factors such as body mass index (BMI) 9 and sex 10 ; perioperative risk factors such as volume management, 11 neoadjuvant chemotherapy, 12 and preoperative total bilirubin levels, 10 ;andsur-geon-associated risk factors such as experience in PD, anastomotic technique, frequency of pancreatic surgery, and blood loss. 13–16 Furthermore, several pancreas-associated risk factors have been proposed in the literature, including histology, 12 the localization, 17 and diameter of the main pancreatic duct (MPD), 9,11,18 and soft pancreatic texture. 3,19
These factors have been combined in numerous risk scores to calculate the individual CR-POPF risk for a specific patient. 9, 20–22 However, there is no uniform reporting classification enhancing the comparability of study results in pancreatic surgery. Therefore, the aims of this systematic review were to evaluate pancreatic texture and MPD diameter as the most prominent pancreas-specific risk factors for CR-POPF after PD, to develop a simple classification for reporting the pancreas-specific risk in future studies, and to validate this classification in a large cohort.
Methods
This systematic review was reported according to the PRISMA guidelines 23 (PRISMA checklist: Supplement 1, http://links.lww.com/SLA/D34). The resources and facilities of the Department of General, Visceral and Transplantation Surgery at the University Hospital of Heidelberg, the Study Center of the German Surgical Society and the 17-center, multinational Pancreas Fistula Study Group (PFSG) database were used to conduct this study.
Systematic Literature Search and Information Sources
The databases Medline (via PubMed), Web of Science, and Cochrane Central Register of controlled trials (CENTRAL) were searched 24 between 2006 and November 2020 without restriction of publication language. A combination of medical subject headings and free text words combined by Boolean connectors was used. An additional hand search of relevant articles was performed. According to the PICO scheme, search terms describing the following two population (P) characteristics were chosen: search terms for pancreatoduodenectomies and search terms for MPD size and pancreatic texture. The full search terms for Medline (via PubMed) were:
(pancreas[MeSH Terms] OR pancreas[tiab] OR pancreatic[-tiab]) AND (surgery[tiab] OR surgeries[tiab] OR surgical [tiab] OR removal[tiab] OR operation [tiab] OR resection* [tiab] OR laparos-cop*[tiab] OR ''surgical procedures, operative”[MeSH Terms] OR “general surgery”[MeSH Terms]) OR pancreaticoduodenec-tom*[tiab] OR pancreatoduodenectom*[tiab] OR duodenopancrea-tectom*[tiab] OR Whipple[tiab] OR ppWhipple[tiab] OR Kausch-Whipple [tiab] OR PPPD[tiab] OR “pancreatic head resection”[tiab] OR pancreatectom*[tiab] OR ''pancreatic resection“[tiab] ORpan-creaticoduodenectomy[MeSH Terms] OR pancreatectomy[MeSH Terms] OR “duodenum-preserving pancreatic head resection”[tiab] OR dpphr[tiab] OR “pancreatic enucleation”[tiab]
AND
((pancreas[tiab] OR pancreatic[tiab]) AND (duct[tiab] OR ducts[tiab]) AND (size[tiab] OR diameter*[tiab])) OR ”small pancreatic duct“ [tiab] OR ”large pancreatic duct”[tiab]
OR ((pancreas[tiab] ORpancreatic[tiab]) AND texture*[tiab]) OR ((“Pancreatic Ducts/diagnosis”[Mesh] OR “Pancreatic Ducts/diagnostic imaging”[Mesh]) AND (size[tiab] OR textur-e*[tiab]))
OR“soft pancreatic parenchyma” [tiab] OR “soft pancreas [tiab]
NOT (animals [mh] NOT humans [mh])
Study Selection
All studies providing data on the association of POPF with either pancreatic texture or MPD after PD were eligible, irrespective of disease. Only studies that used the ISGPS definition of POPF were included. Studies lacking the above-mentioned information, animal studies, and studies reporting or investigating conservative procedures or the placement of interventional drains without surgical resection of the pancreas were excluded, as were studies published before 2006, the time of the first ISGPS POPF definition. This last limitation was chosen because of the multiplicity of adjustments of definitions, therapeutic approaches, and types of surgical interventions reported in earlier publications. Furthermore, all letters, titles without abstract, and case reports, and study protocols were excluded.
All included studies were screened and extracted by two reviewers independently (F.S., P.P.). Differences that could not be resolved were discussed with a third reviewer (A.L.M.).
Data Extraction
The data extracted were: author, year of publication, the primary investigated organ characteristics (texture yes/no, MPD yes/no, texture radiologically measured, texture measured by durom-eter), other primary investigated risk factors for POPF (somatostatin administration, enzymes, different anastomosis, drains), MPD cutoff to differentiate between a narrow and a wide duct (in millimeters), classification of gland texture (soft, hard, firm, friable, and others), sample size, number of POPF in risk and non-risk populations (small duct or soft gland), surgical procedure (PD—resection/preservation of pylorus, different techniques of anastomosis), conclusions of trial, number of grade A/B/C fistulas according to original ISGPS defini-tion, 6 and biochemical leak and B/C POPF (CR-POPF) according to the updated ISGPS definition. 7
Risk of Bias
Risk of bias was assessed using the Quality Assessment of Diagnostic Accuracy Studies 2 tool (QUADAS-2). 25 Four domains were assessed: patient selection, index parameter, reference parameter, and flow and timing. For ''patient selection, the focus was to investigate whether a consecutive or random population was investigated. If the selection process was not random or if there were major differences concerning the surgical procedure and/or the preoperative treatments, the risk of bias was high. The domain “index parameter” concerned the influence of individual pancreatic characteristics such as pancreatic gland texture and MPD diameter on the rate of CR-POPF. Texture had to be reported as soft and not-soft/hard. The diameter of the MPD had to be given in millimeters. A study that fulfilled all these conditions was classified as having a low risk of bias. The domain ''reference parameter covered the influence of well-known con-founders on the rate of CR-POPF. At least, the texture of the pancreatic gland, the diameter of the MPD, and the BMI 9 had to be recorded and evaluated for a study to be classified as having a low risk of bias. Differences in the perioperative therapy, such as (neoadjuvant therapy, within the study population were also recorded as a high risk of bias. In the domain “flow and timing,” nonprospective study design and missing data were defined as high risk of bias.
The risk of publication bias was assessed by means of funnel plots for the association of the parenchymal characteristics (texture and MPD) with CR-POPF with/without biochemical leaks.
The Proposed Classification and Its Validation
The results of the systematic reviews were discussed by the members of the ISGPS on February 6, 2020 at the third World Pancreas Forum in Bern. Based on the odds ratios (ORs) of the meta-analyses the following classification system for MPD size andtexture was developed, where the category “not-soft” comprises any pancreatic texture (eg, hard, firm, sclerotic) other than soft, whereas “soft” also includes “friable” and “brittle” tissue (Fig. 1). Based on the results from the included studies, texture and MDP should be measured intraoperatively by the surgeon (see details in the Results section and Discussion).
Figure 1.
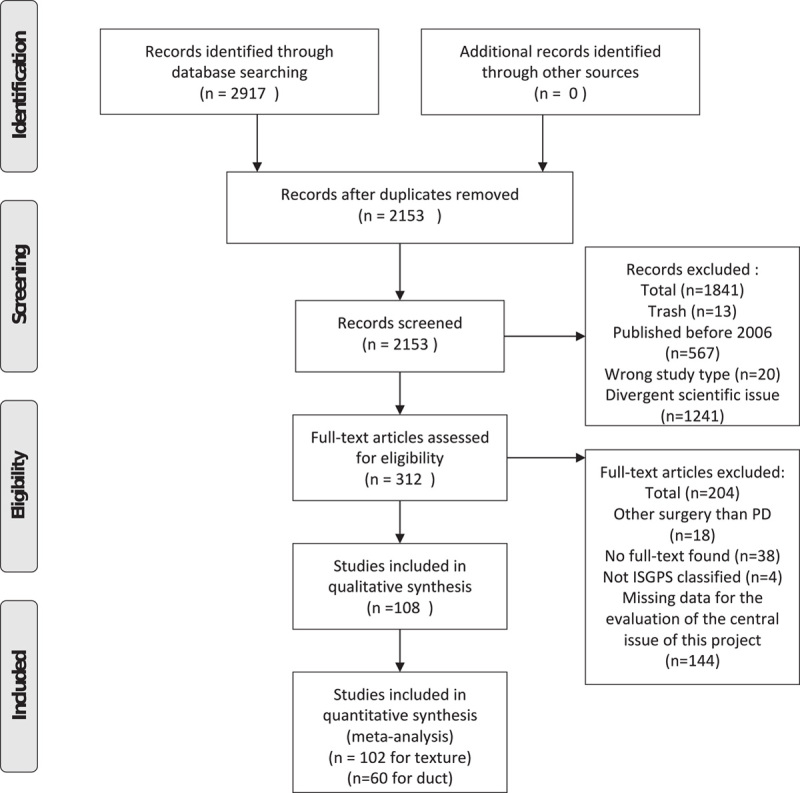
PRISMA flow chart.
| Type A: not-soft pancreatic texture | AND main pancreatic duct size >3mm |
| Type B: not-soft pancreatic texture | AND main pancreatic duct size ≤3mm |
| Type C: soft pancreatic texture | AND main pancreatic duct size > 3mm |
| Type D: soft pancreatictexture | AND main pancreatic duct size ≤ 3mm |
Thereafter, this classification was validated using the PFSG database, which includes 5533 pancreatoduodenectomies carried out between 2004 and 2019. Finally, the proposal was approved by all ISPGS members, whereupon the manuscript was prepared and peer-reviewed internally to establish the classification.
Statistics
Statistical analysis was performed with the program R. 26 The comparison of low- and high-risk factors was reported as OR with 95% CIs. The studies were pooled using the Mantel-Haenszel method with a random-effects model. A P value <0.05 was considered to show a statistically significant difference. Forest plots were created for graphic presentation of the results. To assess a potential publication bias, funnel plots were created to investigate the presence of graphical asymmetry.
The main analysis included CR-POPF (ISGPS grade B/C) only. Additionally, a sensitivity analysis including also grade A fistula or biochemical leaks was conducted. Furthermore, subgroup analyses were performed for different definitions of pancreatic texture and different cut-offs for MPD diameter.
Results
Literature Search Results
The systematic literature search identified 2153 articles, of which 1841 were excluded because of a publication date before the first ISGPS definition of POPF, inappropriate study type, or divergent research questions. The full texts of the remaining 312 articles were screened, and finally 108 studies were included in the qualitative analysis. For texture 102 studies and for duct size 60 studies were included in the quantitative analysis (Fig. 2). Details of the studies included can be found in supplement 2, http://links.lww.com/SLA/D35.
Figure 2.
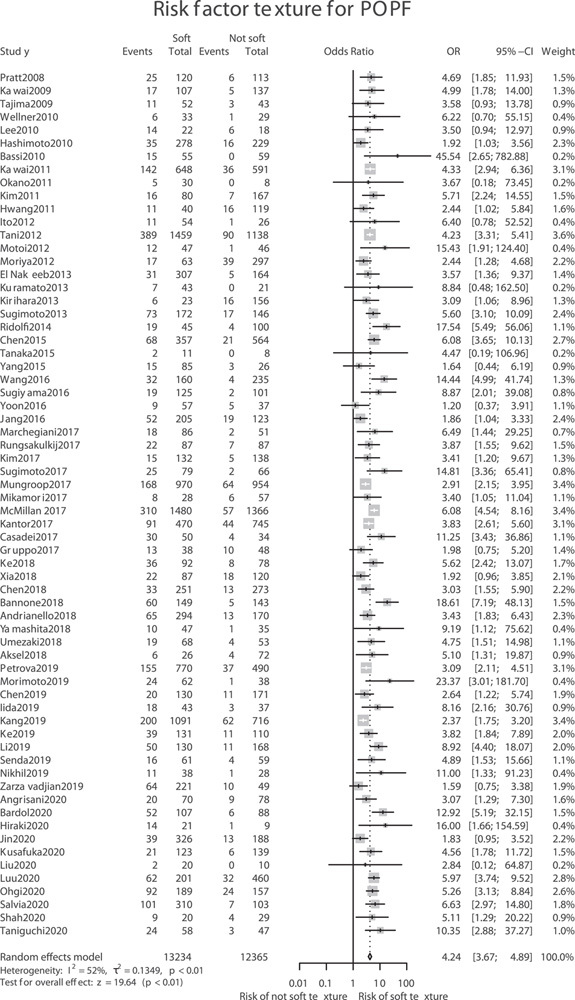
Meta-analysis of pancreatic texture (soft vs not-soft) and clinically relevant postoperative pancreatic fistula, defined as POPF B or C according to the ISGPS.
Association of Texture and POPF
A total of 66 studies9–12,18,20,27–86 with 25,599 patients investigated the association of soft pancreatic texture and CR-POPF development. The classifications used to grade pancreatic texture differed among the studies. The most frequent classification was description of the pancreatic gland as soft versus hard based on the impression of the operating surgeon. Other terms used were “firm,” “friable,” “sclerotic,” “medium,” and “intermediate.” Because of this discrepancy, differentiation between soft and not-soft was chosen for use in this systematic review and meta-analysis. Soft also includes “friable” and “brittle” tissue. “Not-soft” comprises any pancreatic texture (eg, hard, firm, sclerotic) other than soft. Soft texture was significantly associated with CR-POPF (B/C) (OR 4.24, 95% CI 3.67–4.89, P < 0.01 (Fig. 3).
Figure 3.
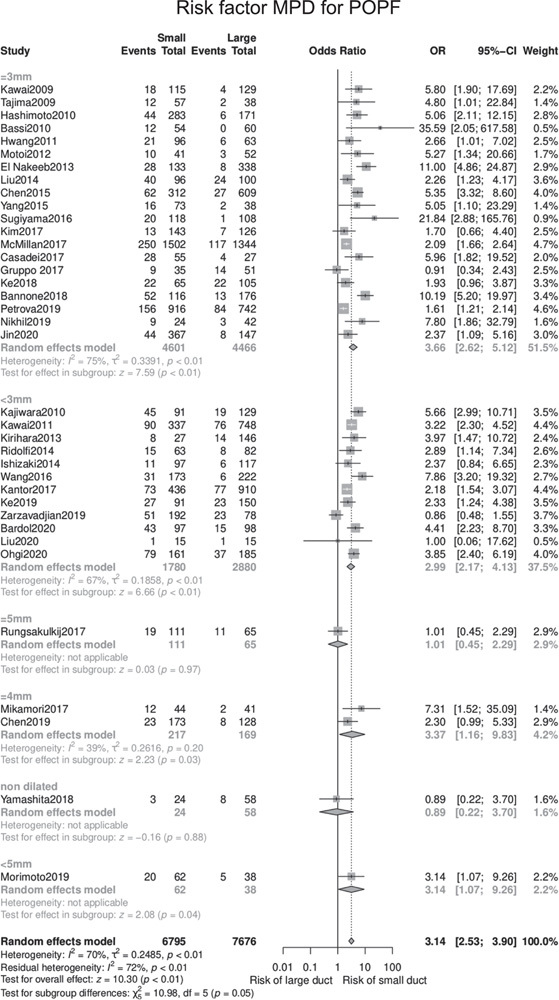
Meta-analysis of main pancreatic duct size and clinically relevant postoperative pancreatic fistula, defined as POPF B or C according to the ISGPS.
The sensitivity analysis including also biochemical leaks showed similar results in 102 studies9–12,18,20,27–122 for 37,259 patients with an OR of 4.28 (95% CI: 3.84–4.78, P < 0.01).
Association of MPD Size and POPF
A total of 37 studies10,11,28–30,33,34,37,39,44,45,48–53,55,58–64,72,74,75,77,81,83,87,87,123–125 with 14,471 patients investigated MPD diameter as a risk factor for CR-POPF, applying different cut-offs. Irrespective of the cut-off, duct diameter was associated with CR-POPF (OR 3.14, 95% CI 2.53–3.90, P < 0.01) (Fig. 4). Similar results were obtained if only studies were included that used intraoperative measurements of the MPD (29 studies; OR 3.25; 95% CI 2.52–4.17, P < 0.01)
Figure 4.
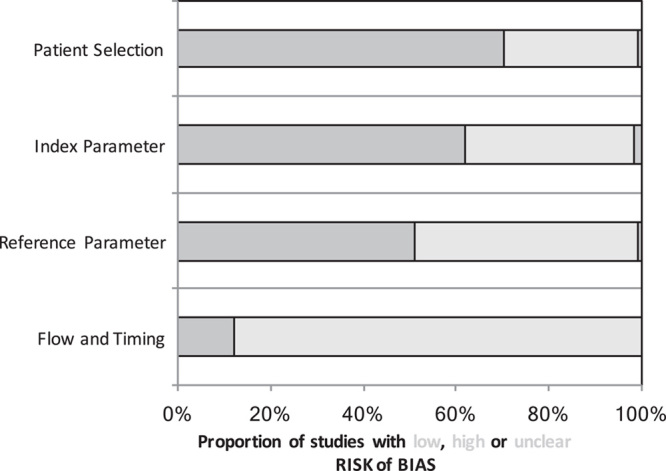
Risk of bias of the included studies according to QUADAS-2 analysis. 25
A sensitivity analysis including also biochemical leaks showed similar results irrespective of the chosen cut-off with an OR of 3.14 (95%CI 2.73–3.61, P < 0.01). The 60 studies ana-lyzed10,11,28–30,33,34,37,39,44,45,47–55,58–64,69,72,74,75,77,81,83,87–103,120,121,123–128 included 23,932 patients. Again, results were comparable when including only the 49 studies that assessed the MPD intraoperatively (OR 3.21, 95% CI 2.70–3.81, P < 0.01) The studies included for analysis used different cutoff values for MPD diameter. Twelve studies 10,44,55,59,63,72,75,77,81,83,123,124 including 4660 patients classified MPD diameter of ≤3mm as a high risk for CR-POPF development (OR 2.99, 95% CI 2.17–4.13, P < 0.01). Another 20 studies11,29,30,33,37,39,44,45,48– 51,58,61,62,64,74,79,87,125 with 9067 patients used MPD diameter of ≤3 mm as the high-risk cut-off, although the results were comparable (OR 3.66, 95% CI 2.62–5.12, P < 0.01).
One study used an MPD cut-off of exactly ≤5 mm, 34 without a significant association with CR-POPF (176 patients; OR 1.01, 95% CI 0.45–2.29, P = 0.97), whereas another study chose to classify glands with MPD diameter <5 mm as high risk, 53 with borderline significant results (100 patients; OR 3.14, 95% CI 1.07–9.26, P = 0.04).
One study 60 defined the duct as nondilated versus dilated to differentiate between a high-risk and a low-risk gland, with no significant association with CR-POPF (82 patients; OR 0.89, 95% CI 0.22-3.70, P = 0.88), whereas 2 studies 52,69 had the MPD cut-off at 4 mm, with a significant association between MPD <4mm and CR-POPF (386 patients; OR 3.37; 95% CI 1.16-9.83, P = 0.03).
Risk of Bias
The QUADAS-2 analysis shows a high risk of bias in all evaluated domains in a number of the studies included (Fig. 5; Supplement 3, http://links.lww.com/SLA/D36). Detailed assessment is shown in supplement 2, http://links.lww.com/SLA/D35. For “patient selection”, 31 of 108 studies (28.7%) were at high risk of bias due to differences in selection criteria. Thirty-nine of 108 studies (36.1%) were at high risk of bias due to missing data for pancreatic texture and/or the diameter of the MPD or to major differences in the classification of these characteristics. Another 52 of 108 studies (48.2%) did not consider relevant confounding factors or had major differences in the perioperative treatment. Finally, 95 of 108 studies (88.0%) were judged to be at high risk of bias due the retrospective study design.
Figure 5.
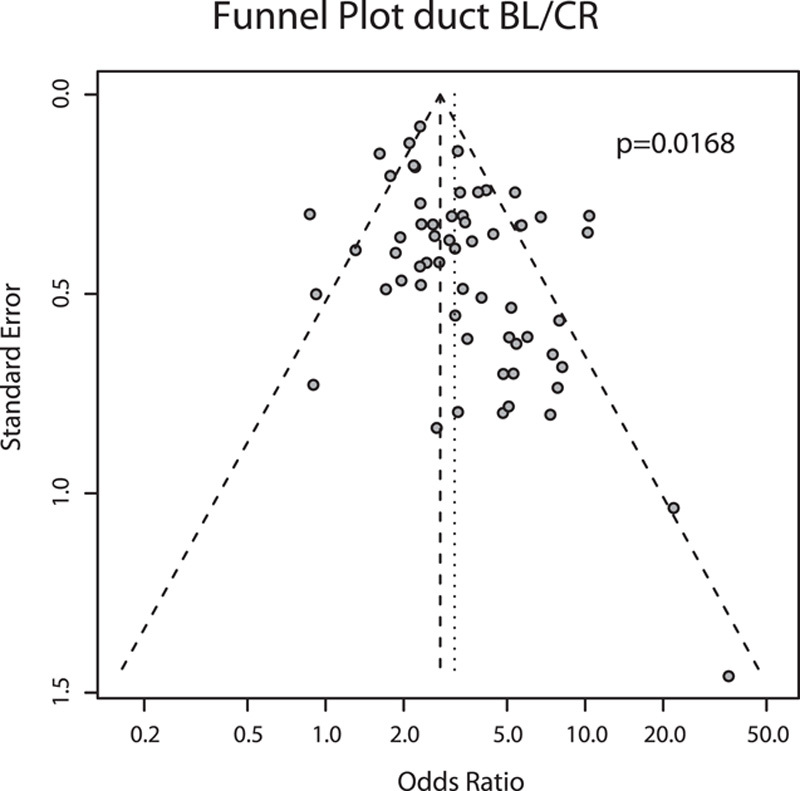
Funnel Plot for publications investigating the association between main pancreatic duct size and postoperative pancreatic fistula.
Additionally, the association of POPF including biochemical leaks and MPD size showed significant asymmetry (P = 0.0168). It is therefore very likely that studies without significant association were withheld, resulting in publication bias (Fig. 6). The funnel plots of the remaining metaanalyses can be found in supplement 4, http://link-s.lww.com/SLA/D37.
Figure 6.
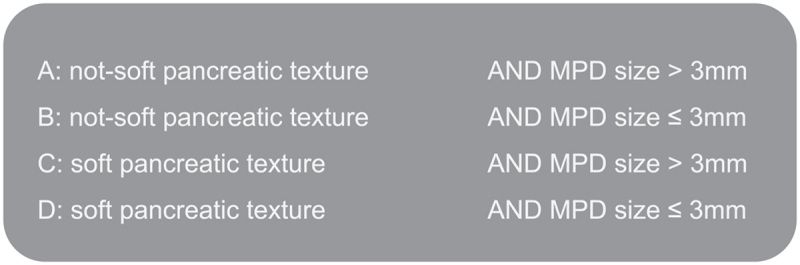
ISGPS consensus classification on risk of POPF based on pancreatic texture and main pancreatic duct size The category “soft” also includes brittle or friable tissue. The category “not-soft” contains any pancreatic texture (eg, hard, firm, or sclerotic) other than “soft, brittle or friable”.
ISGPS Proposal
The results of the meta-analyses were discussed with the ISGPS members on February 6, 2020 at the third World Pancreas Forum in Bern. Based on the ORs of CR-POPF for pancreatic texture and MPD size, the members proposed a simple, sensible classification (Fig. 1) with the goal of facilitating reporting and enabling the comparison of pancreas-associated fistula risk factors among studies in the future. The category “soft” also includes brittle or friable tissue. The category “not-soft” contains any pancreatic texture (eg, hard, firm or sclerotic) other than “soft, brittle, or friable.” As most studies in our meta-analysis used intraoperative evaluation of pancreatic texture via palpation by the surgeon (Supplement 2, http://links.lww.com/SLA/D35), we recommend applying this method for assessment of pancreatic texture. Furthermore, intraoperative palpation has been shown to correlate well with durometer measure-ments. 12,129 Similarly, MPD diameter should be measured intraoperatively at the transection site of the pancreatic remnant (site of anastomosis), as this was the method most frequently used in the included studies.
Validation of the ISGPS Proposal
Finally, the ISGPS proposal was applied to an independent cohort comprising 5533 patients of the PFSG. The rates of CR-POPF differed significantly among the grades: 3.5%, 6.2%, 16.6%, and 23.2% for grades A, B, C, and D, respectively (overall P < 0.001) (Table 1).
Table 1.
CR-POPF for Grade A-D Anastomoses in 5533 Patients of the Pancreatic Fistula Study Group
| No. of Patients | No. of Patients | ||||
|---|---|---|---|---|---|
| Without CR-POPF | With CR-POPF | Rates | P | ||
| A | Not-soft pancreatic texture and MPD > 3 mm | 1533 | 56 | 3.5% | 0.002 |
| B | Not-soft pancreatic texture and MPD ≤ 3 mm | 854 | 56 | 6.2% | <0.001 |
| C | Soft pancreatic texture and MPD >3 mm | 847 | 169 | 16.6% | <0.001 |
| D | Soft pancreatic texture and MPD ≤3 mm | 1547 | 471 | 23.2% | |
| 4781 | 752 | 15.7% | Overall P < 0.001 |
Discussion
The aims of this systematic review were to evaluate pancreatic texture and MPD size as risk factors for POPF after PD and to develop a consensus for standardized reporting of pancreas- associated risk factors. The results of the meta-analysis show a significant association of both of these factors with the development of CR-POPF with the association being stronger for soft pancreatic texture than for small MPD size. The association was stronger for soft pancreatic texture than for small MPD size. The quantitative results are limited by the inherent risk of bias due to retrospective designs and failure to include confounding factors in some of the included studies. To improve comparability of studies, the ISGPS herewith suggests a straightforward, 4-teir reporting classification (Fig. 1).
There are probably many different reasons why CR-POPF rates are higher following PD with soft pancreatic tissue, including the increased exocrine function of soft glands, 56 the association of soft glands with smaller MPD, and the higher number of side branches in soft glands. 130 Furthermore, soft pancreatic tissue—as well as friable/brittle glands, which were included in the soft texture group in this systematic review—results in a lack of suture-holding capacity, and even ischemic or necrotic processes due to compression of the suture, ultimately leading to anastomotic failure. 131 In addition, a lower degree of fibrosis, as present in soft pancreatic glands, is a risk factor for POPF development. 132,133 Eshmuminov et al published a systematic review concerning the impact of a soft pancreatic gland on the development of CR-POPF according to the updated ISGPS definitions and the results presented here are in line with their findings. 19
Similarly, the association of a narrow MPD with the incidence of POPF is multifactorial. First of all, pancreatic anastomosis creation is technically more challenging with a small MPD than with a more dilated duct. Second, small MPDs are associated with postoperative acute pancreatitis. 134 Most studies have used MPD diameter of ≤3 mm as a cut-off to differentiate between high-risk and low-risk glands. Considering the results of our meta-analysis, this cut-off seems reasonable for classification purposes, due to the clear results in comparison with higher cutoff values; however, it should be pointed out that MDP size is probably a continuous risk factor for CR-POPF development, as has been explored in previous stud-ies. 20,21,135
We recommend evaluating pancreatic texture intraoperatively via palpation of the gland by an experienced surgeon. This method was used most frequently in the included studies and has been shown to correlate well with durometer measurements. 12,129 Similarly, MPD diameter should be measured intraoperatively at the transection site of the pancreatic remnant (site of anastomosis), as this was the method most frequently used in the included studies. Probing of the duct should be avoided or limited to once, not to distort MPD diameter.
The proposed classification does not aim to calculate the individual CR-POPF risk for a specific patient. This is better done by using one of the many fistula risk scores which, besides pancreas-inherent factors, include nonpancreatic risk factors.9,10,20–22 However, few of these scores have been as extensively validated as the fistula risk score by Vollmer et al,20,21,136–38 and no consensus on the clinical consequences 135,138 of implementing these scores in everyday clinical practice has been reached because interventional efficacy trials are sparse in the literature so far. Therefore, the aim of this systematic review was not to establish yet another fistula risk score to evaluate the individual CR-POPF risk of a given patient, but rather to provide a simple reporting classification of organ-specific risks for CR-POPF following PD. This seems essential for several reasons. First, as was evident from the heterogeneous trials in our systematic review, studies investigating pancreatic surgery lack a standardized risk factor and reporting of confounders. The current proposal addresses this shortcoming with regard to 2 of the most prominent pancreas-inherent risk factors, thus enabling transparent comparison of future studies. The proposed system could be especially useful as a reporting tool for baseline characteristics in future clinical trials exploring the efficacy of surgical or perioperative mitigation strategies to address CR-POPF. Second, the proposal is useful for auditing, as it allows standardization and comparison between centers and can be easily implemented. Third, it can be used in everyday clinical practice as a simple tool to guide intraoperative management in high-risk anastomoses (groups C and D).
Palpation of the pancreas by the surgeon is the method most frequently used to determine the texture of the gland. However, other ways of measuring the texture of the parenchyma, such as CT measurement, pathologic staining, and direct measurement with a durometer, may also be used, as studies show good correlation between these measurements and surgeons' judgment. 52,129 Furthermore, texture and MPD size can also be determined at the resected PD specimen. Therefore, the proposal can readily be implemented in the minimally invasive era, once the pancreatic head specimen has been removed.
Limitations
Our study has several limitations. First, only studies that appeared after publication of the first ISGPS POPF definition were included. This restriction was necessary due to the myriad different POPF definitions before publication of the ISGPS consensus, 6,7 impeding comparison of results. Second, the methodological quality of some of the included studies was limited, as can be seen in the risk of bias analysis (Figs. 5 and 6; Supplement 2, http://links.lww.com/SLA/D35). However, as only studies with standardized ISGPS definition of POPF were included and results were consistent over time and across countries, the proposed classification is based on sound evidence. Third, the classification explores only the most prominent pancreas-inherent risk factors and focuses entirely on the pancreatic gland itself, neglecting numerous other risk factors. This simplification is inherent in the objective of the classification itself, that is, to provide a simple reporting tool for comparison and clinical decision-making.
In conclusion, the ISGPS recommends reporting MPD size and pancreatic texture according to the proposed classification system for better comparability of study results, clinical decision-making, and auditing.
Footnotes
Funding: No funding has been received for this work.
F.S. and A.L.M. have contributed equally to this study.
Author Contributions: F.S.: acquisition and analysis of data, drafting the manuscript, final approval of manuscript; A.L.M.: conception and design of study, acquisition of data and analysis and interpretation of data, drafting the manuscript, editing of manuscript, final approval of manuscript;
P.P.: design of study, acquisition and analysis and interpretation of data, critical revision of manuscript, final approval of manuscript; M.T.T.: design of study, acquisition and analysis of data, critical revision of manuscript, final approval of manuscript; P.C.M.: design of study, acquisition and analysis of data, critical revision of manuscript, final approval of manuscript; G.M.: conception and design of study, critical revision of manuscript, final approval of manuscript;
M.G.B.: conception and design of study, critical revision of manuscript, final approval of manuscript; F.U.: conception and design of study, critical revision of manuscript, final approval of manuscript;
J.R.I.: conception and design of study, critical revision of manuscript, final approval of manuscript; M.F.: Giovannconception and design of study, critical revision of manuscript, final approval of manuscript; C.F.C.: conception and design of study, critical revision of manuscript, final approval of manuscript.
M.A.: conception and design of study, critical revision of manuscript, final approval of manuscript; K.Z.: conception and design of study, critical revision of manuscript, final approval of manuscript; H.F.: conception and design of study, critical revision of manuscript, final approval of manuscript; J.W.: conception and design of study, critical revision of manuscript, final approval of manuscript; J.W.: conception and design of study, critical revision of manuscript, final approval of manuscript; O.S.: conception and design of study, critical revision of manuscript, final approval of manuscript; T.H.: conception and design of study, critical revision of manuscript, final approval of manuscript; D.R.: conception and design of study, critical revision of manuscript, final approval of manuscript; D.K.: conception and design of study, critical revision of manuscript, final approval of manuscript; C.W.: conception and design of study, critical revision of manuscript, final approval of manuscript; Y.I.M.: conception and design of study, critical revision of manuscript, final approval of manuscript; S.V.S.: Giovanconception and design of study, critical revision of manuscript, final approval of manuscript; K.D.L.: conception and design of study, critical revision of manuscript, final approval of manuscript; C.D.: conception and design of study, critical revision of manuscript, final approval of manuscript; C.B.: conception and design of study, critical revision of manuscript, final approval of manuscript; J.P.N.: conception and design of study, critical revision of manuscript, final approval of manuscript; M.K.D: conception and design of study, critical revision of manuscript, final approval of manuscript.
The authors report no conflicts of interest.
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal's website, www.annalsofsurgery.com.
REFERENCES
- 1. Mihaljevic AL, Kleeff J, Friess H. Adenocarcinoma of the pancreas. In: Poston GJ, D’Angelica M, Adam R, eds. Surgical Management of Hepatobiliary and Pancreatic Disorders. Informa Healthcare; 2011:380–400. [Google Scholar]
- 2. Witzigmann H, Diener MK, Kienkötter S, et al. No need for routine drainage after pancreatic head resection: the dual-center, randomized, controlled PANDRA trial (ISRCTN04937707). Ann Surg. 2016;264:528–537. [DOI] [PubMed] [Google Scholar]
- 3. Keck T, Wellner UF, Bahra M, et al. Pancreatogastrostomy versus pancrea-tojejunostomy for RECOnstruction After PANCreatoduodenectomy (REC0-PANC, DRKS 00000767): perioperative and long-term results of a multicenter randomized controlled trial. Ann Surg. 2016;263:440–449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Diener MK, Hüttner FJ, Kieser M, et al. Partial pancreatoduodenectomy versus duodenum-preserving pancreatic head resection in chronic pancreatitis: the multicentre, randomised, controlled, double-blind ChroPac trial. Lancet Lond Engl. 2017;390:1027–1037. [DOI] [PubMed] [Google Scholar]
- 5. Probst P, Hüttner FJ, Meydan O, et al. Evidence map of pancreatic surgery: protocol for a living systematic review and meta-analysis. BMJ Open. 2019;9:e032353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Bassi C, Dervenis C, Butturini G, et al. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery. 2005;138:8–13. [DOI] [PubMed] [Google Scholar]
- 7. Bassi C, Marchegiani G, Dervenis C, et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surgery. 2017;161:584–591. [DOI] [PubMed] [Google Scholar]
- 8. McMillan MT, Vollmer CM. Predictive factors for pancreatic fistula following pancreatectomy. Langenbecks Arch Surg. 2014;399:811–824. [DOI] [PubMed] [Google Scholar]
- 9. Mungroop TH, van Rijssen LB, van Klaveren D, et al. Alternative Fistula Risk Score for Pancreatoduodenectomy (a-FRS): Design and International External Validation. Ann Surg. 2019;269:937–943. [DOI] [PubMed] [Google Scholar]
- 10. Kantor O, Talamonti MS, Pitt HA, et al. Using the NSQIP pancreatic demonstration project to derive a modified fistula risk score for preoperative risk stratification in patients undergoing pancreaticoduodenectomy. J Am Coll Surg. 2017;224:816–825. [DOI] [PubMed] [Google Scholar]
- 11. Bannone E, Andrianello S, Marchegiani G, et al. Postoperative acute pancreatitis following pancreaticoduodenectomy: a determinant of fistula potentially driven by the intraoperative fluid management. Ann Surg. 2018;268:815–822. [DOI] [PubMed] [Google Scholar]
- 12. Marchegiani G, Ballarin R, Malleo G, et al. Quantitative assessment of pancreatic texture using a durometer: a new tool to predict the risk of developing a postoperative fistula. World J Surg. 2017;41:2876–2883. [DOI] [PubMed] [Google Scholar]
- 13. Krautz C, Haase E, Elshafei M, et al. The impact of surgical experience and frequency of practice on perioperative outcomes in pancreatic surgery. BMC Surg. 2019;19:108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Seykora TF, Ecker BL, McMillan MT, et al. The beneficial effects of minimizing blood loss in pancreatoduodenectomy. Ann Surg. 2019;270:147–157. [DOI] [PubMed] [Google Scholar]
- 15. Shrikhande SV, Sivasanker M, Vollmer CM, et al. Pancreatic anastomosis after pancreatoduodenectomy: A position statement by the International Study Group of Pancreatic Surgery (ISGPS). Surgery. 2017;161:1221–1234. [DOI] [PubMed] [Google Scholar]
- 16. Casciani F, Trudeau M, Asbun H, et al. Surgeon experience contributes to improved outcomes in pancreatoduodenectomies at high risk for fistula development. Surgery. 2021;169:708–720. [DOI] [PubMed] [Google Scholar]
- 17. El Nakeeb A, Sultan AM, Atef E, et al. Tailored pancreatic reconstruction after pancreaticoduodenectomy: a single-center experience of 892 cases. Hepatobiliary Pancreat Dis Int. 2017;16:528–536. [DOI] [PubMed] [Google Scholar]
- 18. Senda Y, Shimizu Y, Natsume S, et al. Randomized clinical trial of duct-to-mucosa versus invagination pancreaticojejunostomy after pancreatoduode-nectomy. Br J Surg. 2018;105:48–57. [DOI] [PubMed] [Google Scholar]
- 19. Eshmuminov D, Schneider MA, Tschuor C, et al. Systematic review and meta-analysis of postoperative pancreatic fistula rates using the updated 2016 International Study Group Pancreatic Fistula definition in patients undergoing pancreatic resection with soft and hard pancreatic texture. HPB. 2018;20:992–1003. [DOI] [PubMed] [Google Scholar]
- 20. Pratt WB, Callery MP, Vollmer CM. Risk prediction for development of pancreatic fistula using the ISGPF classification scheme. World J Surg. 2008;32:419–428. [DOI] [PubMed] [Google Scholar]
- 21. Callery MP, Pratt WB, Kent TS, et al. A prospectively validated clinical risk score accurately predicts pancreatic fistula after pancreatoduodenectomy. J Am Coll Surg. 2013;216:1–14. [DOI] [PubMed] [Google Scholar]
- 22. Mungroop TH, Klompmaker S, Wellner UF, et al. Updated alternative fistula risk score (ua-FRS) to include minimally invasive pancreatoduodenectomy: pan-european validation. Ann Surg. 2021;273:334–340. [DOI] [PubMed] [Google Scholar]
- 23. Moher D, Liberati A, Tetzlaff J, et al. , PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Goossen K, Tenckhoff S, Probst P, et al. Optimal literature search for systematic reviews in surgery. Langenbecks Arch Surg. 2018;403:119–129. [DOI] [PubMed] [Google Scholar]
- 25. Whiting PF, Rutjes AWS, Westwood ME, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155:529–536. [DOI] [PubMed] [Google Scholar]
- 26. R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Published online 2018. https://www.R-project.org/. [Google Scholar]
- 27. Ito Y, Kenmochi T, Irino T, et al. Strategies to prevent pancreatic fistula after pancreaticoduodenectomy. Hepatogastroenterology. 2012;59:2609–2613. [DOI] [PubMed] [Google Scholar]
- 28. Kawai M, Tani M, Hirono S, et al. How do we predict the clinically relevant pancreatic fistula after pancreaticoduodenectomy?-an analysis in 244 consecutive patients. World J Surg. 2009;33:2670–2678. [DOI] [PubMed] [Google Scholar]
- 29. Ke Z, Cui J, Hu N, et al. Risk factors for postoperative pancreatic fistula: analysis of 170 consecutive cases of pancreaticoduodenectomy based on the updated ISGPS classification and grading system. Medicine (Baltimore). 2018;97:e12151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Kim DH, Choi SH, Choi DW, et al. Division of surgeon workload in pancreaticoduodenectomy: striving to decrease post-operative pancreatic fistula. ANZ J Surg. 2017;87:569–575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Kim WS, Choi DW, Choi SH, et al. Clinical validation of the ISGPF classification and the risk factors of pancreatic fistula formation following duct-to-mucosa pancreaticojejunostomy by one surgeon at a single center. J Gastrointest Surg. 2011;15:2187–2192. [DOI] [PubMed] [Google Scholar]
- 32. Okano K, Kakinoki K, Suto H, et al. Persisting ratio of total amylase output in drain fluid can predict postoperative clinical pancreatic fistula. J Hepatobiliary Pancreat Sci. 2011;18:815–820. [DOI] [PubMed] [Google Scholar]
- 33. Petrova E, Lapshyn H, Bausch D, et al. Risk stratification for postoperative pancreatic fistula using the pancreatic surgery registry StuDoQ Pancreas of the German Society for General and Visceral Surgery. Pancreatology Al. 2019;19:17–25. [DOI] [PubMed] [Google Scholar]
- 34. Rungsakulkij N, Mingphruedhi S, Tangtawee P, et al. Risk factors for pancreatic fistula following pancreaticoduodenectomy: a retrospective study in a Thai tertiary center. World J Gastrointest Surg. 2017;9:270–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Wellner UF, Kayser G, Lapshyn H, et al. A simple scoring system based on clinical factors related to pancreatic texture predicts postoperative pancreatic fistula preoperatively. HPB 2010;12:696–702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Andrianello S, Marchegiani G, Malleo G, et al. Polyester sutures for pancreaticojejunostomy protect against postoperative pancreatic fistula: a case-control, risk-adjusted analysis. HPB 2018;20:977–983. [DOI] [PubMed] [Google Scholar]
- 37. Bassi C, Molinari E, Malleo G, et al. Early versus late drain removal after standard pancreatic resections: results of a prospective randomized trial. Ann Surg. 2010;252:207–214. [DOI] [PubMed] [Google Scholar]
- 38. Kuramoto M, Ikeshima S, Shimada S, et al. Pancreaticojejunostomy by reinforcing the pancreas without covering the anastomotic line reduces pancreatic fistula. Int J Surg. 2013;11:909–913. [DOI] [PubMed] [Google Scholar]
- 39. Tajima Y, Kuroki T, Tsuneoka N, et al. Anatomy-specific pancreatic stump management to reduce the risk of pancreatic fistula after pancreatic head resection. World J Surg. 2009;33:2166–2176. [DOI] [PubMed] [Google Scholar]
- 40. Tanaka K, Tomita H, Osada S, et al. Significance of histopathological evaluation of pancreatic fibrosis to predict postoperative course after pancreatic surgery. Anticancer Res. 2015;35:1749–1756. [PubMed] [Google Scholar]
- 41. Tani M, Kawai M, Hirono S, et al. Use of omentum or falciform ligament does not decrease complications after pancreaticoduodenectomy: nation wide survey of the Japanese Society of Pancreatic Surgery. Surgery. 2012;151:183–191. [DOI] [PubMed] [Google Scholar]
- 42. Umezaki N, Hashimoto D, Nakagawa S, et al. Number of acinar cells at the pancreatic stump predicts pancreatic fistula after pancreaticoduodenectomy. Surg Today. 2018;48:790–795. [DOI] [PubMed] [Google Scholar]
- 43. Yoon JH, Lee JM, Lee KB, et al. Pancreatic steatosis and fibrosis: quantitative assessment with preoperative multiparametric mr imaging. Radiology. 2016;279:140–150. [DOI] [PubMed] [Google Scholar]
- 44. Kawai M, Kondo S, Yamaue H, et al. Predictive risk factors for clinically relevant pancreatic fistula analyzed in 1,239 patients with pancreaticoduo-denectomy: multicenter data collection as a project study of pancreatic surgery by the Japanese Society of Hepato-Biliary-Pancreatic Surgery. J Hepatobiliary Pancreat Sci. 2011;18:601–608. [DOI] [PubMed] [Google Scholar]
- 45. Casadei R, Ricci C, Taffurelli G, et al. Prospective validation of a preoperative risk score model based on pancreatic texture to predict postoperative pancreatic fistula after pancreaticoduodenectomy. Int J Surg Lond Engl. 2017;48:189–194. [DOI] [PubMed] [Google Scholar]
- 46. Chen CB, McCall NS, Pucci MJ, et al. The combination of pancreas texture and postoperative serum amylase in predicting pancreatic fistula risk. Am Surg. 2018;84:889–896. [PubMed] [Google Scholar]
- 47. Chen J-Y, Feng J, Wang X-Q, et al. Risk scoring system and predictor for clinically relevant pancreatic fistula after pancreaticoduodenectomy. World J Gastroenterol. 2015;21:5926–5933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. El Nakeeb A, Salah T, Sultan A, et al. Pancreatic anastomotic leakage after pancreaticoduodenectomy. Risk factors, clinical predictors, and management (single center experience). World J Surg. 2013;37:1405–1418. [DOI] [PubMed] [Google Scholar]
- 49. Gruppo M, Angriman I, Martella B, et al. Perioperativealbuminratioisassociated with post-operative pancreatic fistula. ANZ J Surg. 2018;88:E602–E605. [DOI] [PubMed] [Google Scholar]
- 50. Hashimoto Y, Traverso LW. Incidence of pancreatic anastomotic failure and delayed gastric emptying after pancreatoduodenectomy in 507 consecutive patients: use of a web-based calculator to improve homogeneity of definition. Surgery. 2010;147:503–515. [DOI] [PubMed] [Google Scholar]
- 51. McMillan MT, Zureikat AH, Hogg ME, et al. A propensity score-matched analysis of robotic vs open pancreatoduodenectomy on incidence of pancreatic fistula. JAMA Surg. 2017;152:327–335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Mikamori M, Gotoh K, Takahashi H, et al. Novel intraoperative use of the “Tensipresser” to assess factors predictive of pancreatic fistula after pancreaticoduodenectomy. Surg Today. 2017;47:1201–1207. [DOI] [PubMed] [Google Scholar]
- 53. Morimoto M, Honjo S, Sakamoto T, et al. Bacterial smear test of drainage fluid after pancreaticoduodenectomy can predict postoperative pancreatic fistula. Pancreatology. 2019;19:274–279. [DOI] [PubMed] [Google Scholar]
- 54. Moriya T, Clark CJ, Kirihara Y, et al. Stenting and the rate of pancreatic fistula following pancreaticoduodenectomy. Arch Surg. 2012;147:35–40. [DOI] [PubMed] [Google Scholar]
- 55. Ridolfi C, Angiolini MR, Gavazzi F, et al. Morphohistological features of pancreatic stump are the main determinant of pancreatic fistula after pancreatoduodenectomy. Bio Med Res Int. 2014;2014:641239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Lee SE, Jang J-Y, Lim C-S, et al. Measurement of pancreatic fat by magnetic resonance imaging. Ann Surg. 2010;251:932–936. [DOI] [PubMed] [Google Scholar]
- 57. Sugimoto M, Takahashi S, Kojima M, et al. In patients with a soft pancreas, a thick parenchyma, a small duct, and fatty infiltration are significant risks for pancreatic fistula after pancreaticoduodenectomy. J Gastrointest Surg. 2017;21:846–854. [DOI] [PubMed] [Google Scholar]
- 58. Sugiyama M, Suzuki Y, Nakazato T, et al. Pancreatic duct holder and mucosa squeeze-out technique for duct-to-mucosa pancreatojejunostomy after pancreatoduodenectomy: propensity score matching analysis. World J Surg. 2016;40:3021–3028. [DOI] [PubMed] [Google Scholar]
- 59. Wang G, Li L, Ma Y, et al. External versus internal pancreatic duct drainage for the early efficacy after pancreaticoduodenectomy: a retrospectively comparative study. J Investig Surg. 2016;29:226–233. [DOI] [PubMed] [Google Scholar]
- 60. Yamashita K, Kato D, Sasaki T, et al. Contaminated drainage fluid and pancreatic fistula after pancreatoduodenectomy: a retrospective study. Int J Surg Lond Engl. 2018;52:314–319. [DOI] [PubMed] [Google Scholar]
- 61. Yang H, Lu X-F, Xu Y-F, et al. Application of air insufflation to prevent clinical pancreatic fistula after pancreaticoduodenectomy. World J Gastroenterol. 2015;21:1872–1879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Hwang HK, Park JS, Park CI, et al. The impact of body mass index on pancreatic fistula after pancreaticoduodenectomy in Asian patients on the basis of Asia-Pacific perspective of body mass index. JOP J Pancreas. 2011;12:586–592. [PubMed] [Google Scholar]
- 63. Kirihara Y, Takahashi N, Hashimoto Y, et al. Prediction of pancreatic anastomotic failure after pancreatoduodenectomy: the use of preoperative, quantitative computed tomography to measure remnant pancreatic volume and body composition. Ann Surg. 2013;257:512–519. [DOI] [PubMed] [Google Scholar]
- 64. Motoi F, Egawa S, Rikiyama T, et al. Randomized clinical trial of external stent drainage of the pancreatic duct to reduce postoperative pancreatic fistula after pancreaticojejunostomy. Br J Surg. 2012;99:524–531. [DOI] [PubMed] [Google Scholar]
- 65. Sugimoto M, Takahashi S, Gotohda N, et al. Schematic pancreatic configuration: a risk assessment for postoperative pancreatic fistula after pancrea-ticoduodenectomy. J Gastrointest Surg. 2013;17:1744–1751. [DOI] [PubMed] [Google Scholar]
- 66. Xia W, Zhou Y, Lin Y, et al. A predictive risk scoring system for clinically relevant pancreatic fistula after pancreaticoduodenectomy. Med Sci Monit Int Med J Exp Clin Res. 2018;24:5719–5728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Jang J-Y, Chang YR, Kim S-W, et al. Randomized multicentre trial comparing external and internal pancreatic stenting during pancreaticoduodenec-tomy. Br J Surg. 2016;103:668–675. [DOI] [PubMed] [Google Scholar]
- 68. Aksel B, Güven HE. Pancreatic fistula rates after internal and external stenting of the pancreatojejunostomy anastomosis following pancreatoduo-denectomy. Acta Chir Belg. 2020;120:16–22. [DOI] [PubMed] [Google Scholar]
- 69. Chen J-S, Liu G, Li T-R, et al. Pancreatic fistula after pancreaticoduode-nectomy: Risk factors and preventive strategies. J Cancer Res Ther. 2019;15:857–863. [DOI] [PubMed] [Google Scholar]
- 70. Iida H, Tani M, Maehira H, et al. Postoperative pancreatic swelling predicts pancreatic fistula after pancreaticoduodenectomy. Am Surg. 2019;85:321–326. [PubMed] [Google Scholar]
- 71. Kang JS, Park T, Han Y, et al. Clinical validation of scoring systems of postoperative pancreatic fistula after pancreatoduodenectomy: applicability to Eastern cohorts? Hepatobiliary Surg Nutr. 2019;8:211–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Ke Z-X, Xiong J-X, Hu J, et al. Risk factors and management of postoperative pancreatic fistula following pancreaticoduodenectomy: single-center experience. Curr Med Sci. 2019;39:1009–1018. [DOI] [PubMed] [Google Scholar]
- 73. Li Y, Zhou F, Zhu D.-M, et al. Novel risk scoring system for prediction of pancreatic fistula after pancreaticoduodenectomy. World J Gastroenterol. 2019;25:2650–2664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Nikhil S, Halder PJ, Santosh R, et al. Does the anatomy of the transected pancreatic neck influence post Whipple’s operation pancreatic fistula? Indian J Surg Oncol. 2019;10:31–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Zarzavadjian L.e Bian A, Fuks D, Montali F, et al. Predicting the severity of pancreatic fistula after pancreaticoduodenectomy: overweight and blood loss as independent risk factors: retrospective analysis of 277 patients. Surg Infect. 2019;20:486–491. [DOI] [PubMed] [Google Scholar]
- 76. Angrisani M, Sandini M, Cereda M, et al. Preoperative adiposity at bio-impedance vector analysis improves the ability of Fistula Risk Score (FRS) in predicting pancreatic fistula after pancreatoduodenectomy. Pancreatology. 2020;20:545–550. [DOI] [PubMed] [Google Scholar]
- 77. Bardol T, Delicque J, Hermida M, et al. Neck transection level and postoperative pancreatic fistula after pancreaticoduodenectomy: A retrospective cohort study of 195 patients. Int J Surg. 2020;82:43–50. [DOI] [PubMed] [Google Scholar]
- 78. Hiraki M, Miyoshi A, Sadashima E, et al. The novel early predictive marker presepsin for postoperative pancreatic fistula: A pilot study. Exp Ther Med. 2020;20:2298–2304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Jin K-M, Liu W, Wang K, et al. The individualized selection of Pancreaticoen-teric anastomosis in Pancreaticoduodenectomy. BMC Surg. 2020;20:140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Kusafuka T, Kato H, Iizawa Y, et al. Pancreas-visceral fat CT value ratio and serrated pancreatic contour are strong predictors of postoperative pancreatic fistula after pancreaticojejunostomy. BMC Surg. 2020;20:129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Liu Q, Zhao Z, Gao Y, et al. Novel single-layer continuous suture of pancreaticojejunostomy for robotic pancreaticoduodenectomy. J Hepatobiliary Pancreat Sci. 2020;27:56–63. [DOI] [PubMed] [Google Scholar]
- 82. Luu AM, Krasemann L, Fahlbusch T, et al. Facing the surgeon’s nightmare: Incidence and management of postoperative pancreatic fistulas grade C after pancreaticoduodenectomy based on the updated definition of the International Study Group of PancreaticSurgery(ISGPS). J Hepatobiliary Pancreat Sci. 2020;27:171–181. [DOI] [PubMed] [Google Scholar]
- 83. Ohgi K, Okamura Y, Sugiura T, et al. Pancreatic attenuation on computed tomography predicts pancreatic fistula after pancreaticoduodenectomy. HPB. 2020;22:67–74. [DOI] [PubMed] [Google Scholar]
- 84. Salvia R, Marchegiani G, Andrianello S, et al. Redefining the role of drain amylase value for a risk-based drain management after pancreaticoduode-nectomy: early drain removal still is beneficial. J Gastrointest Surg. 2020. [DOI] [PubMed]
- 85. Shah S, Ghimire B, Paudel S, et al. Pancreatic Configuration Index in Predicting postoperative pancreatic fistula in a tertiary care center in Nepal. J Nepal Health Res Counc. 2020;18:172–177. [DOI] [PubMed] [Google Scholar]
- 86. Taniguchi K, Matsuyama R, Yabushita Y, et al. Prophylactic drain management after pancreaticoduodenectomy without focusing on the drain fluid amylase level: A prospective validation study regarding criteria for early drain removal that do not include the drain fluid amylase level. J Hepatobiliary Pancreat Sci. 2020;27:950–961. [DOI] [PubMed] [Google Scholar]
- 87. Chen Y, Ke N, Tan C, et al. Continuous versus interrupted suture techniques of pancreaticojejunostomy after pancreaticoduodenectomy. J Surg Res. 2015;193:590–597. [DOI] [PubMed] [Google Scholar]
- 88. Ferla F, Di Sandro S, Giacomoni A, et al. Pancreatico-duodenectomy and postoperative pancreatic fistula: risk factors and technical considerations in a specialized HPB center. Updat Surg. 2014;66:145–150. [DOI] [PubMed] [Google Scholar]
- 89. Guilbaud T, Birnbaum DJ, Lemoine C, et al. C-reactive protein on postoperative day 1 is a reliable predictor of pancreas-specific complications after pancreaticoduodenectomy. J Gastrointest Surg. 2018;22:818–830. [DOI] [PubMed] [Google Scholar]
- 90. Hu B-Y, Wan T, Zhang W-Z, et al. Risk factors for postoperative pancreatic fistula: analysis of 539 successive cases of pancreaticoduodenectomy. World J Gastroenterol. 2016;22:7797–7805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Katuchova J, Bober J, Harbulak P, et al. Perioperative and follow-upresultsin chronic pancreatitis patients after pancreatic resection. Wien Klin Wochenschr. 2011;123:359–363. [DOI] [PubMed] [Google Scholar]
- 92. Martin AN, Narayanan S, Turrentine FE, et al. Pancreatic duct size and gland texture are associated with pancreatic fistula after pancreaticoduodenectomy but not after distal pancreatectomy. PloS One. 2018;13:e0203841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Rosso E, Casnedi S, Pessaux P, et al. The role of “fatty pancreas” and of BMI in the occurrence of pancreatic fistula after pancreaticoduodenectomy. J Gastrointest Surg. 2009;13:1845–1851. [DOI] [PubMed] [Google Scholar]
- 94. Tani M, Kawai M, Hirono S, et al. Randomized clinical trial of isolated Roux-en-Y versus conventional reconstruction after pancreaticoduodenectomy. Br J Surg. 2014;101:1084–1091. [DOI] [PubMed] [Google Scholar]
- 95. Kim JY, Park JS, Kim JK, et al. A model for predicting pancreatic leakage after pancreaticoduodenectomy based on the international study group of pancreatic surgery classification. Korean J Hepatobiliary Pancreat Surg. 2013;17:166–170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Uemura K, Murakami Y, Sudo T, et al. Elevation of urine trypsinogen 2 is an independent risk factor for pancreatic fistula after pancreaticoduodenectomy. Pancreas. 2012;41:876–881. [DOI] [PubMed] [Google Scholar]
- 97. Yin J, Lu Z, Wu P, et al. Afferent loop decompression technique is associated with a reduction in pancreatic fistula following pancreaticoduodenectomy. World J Surg. 2018;42:3726–3735. [DOI] [PubMed] [Google Scholar]
- 98. Zarzavadjian L.e Bian A, Fuks D, Chopinet S, et al. Consequences of metabolic syndrome on postoperative outcomes after pancreaticoduodenectomy. World J Gastroenterol. 2017;23:3142–3149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. You D, Jung K, Lee H, et al. Comparison of different pancreatic anastomosis techniques using the definitions of the International Study Group of Pancreatic Surgery: a single surgeon's experience. Pancreas. 2009;38:896–902. [DOI] [PubMed] [Google Scholar]
- 100. Barakat O, Ozaki CF, Wood RP. Topically applied 2-octyl cyanoacrylate (Dermabond) for prevention of postoperative pancreatic fistula after pancreaticoduodenectomy. J Gastrointest Surg Off. 2012;16:1499–1507. [DOI] [PubMed] [Google Scholar]
- 101. Shin HW, Kim JK, Park JS, et al. Can we predict postoperative pancreatic leakage after pancreaticoduodenectomy using preoperative fecal elastase-1 level? J Clin Lab Anal. 2013;27:379–383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Fu S-J, Shen S-L, Li S-Q, et al. Risk factors and outcomes of postoperative pancreatic fistula after pancreatico-duodenectomy: an audit of 532 consecutive cases. BMC Surg. 2015;15:34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Su A-P, Zhang Y, Ke N-W, et al. Triple-layer duct-to-mucosa pancreaticojejunostomy with resection of jejunal serosa decreased pancreatic fistula after pancreaticoduodenectomy. J Surg Res. 2014;186:184–191. [DOI] [PubMed] [Google Scholar]
- 104. Akgul O, Merath K, Mehta R, et al. Postoperative pancreatic fistula following pancreaticoduodenectomy—stratification of patient risk. J Gastrointest Surg. 2019;23:1817–1824. [DOI] [PubMed] [Google Scholar]
- 105. Addeo P, Delpero JR, Paye F, et al. Pancreatic fistula after a pancreaticoduodenectomy for ductal adenocarcinoma and its association with morbidity: a multicentre study of the French Surgical Association. HPB (Oxford). 2014;16:46–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Berger AC, Howard TJ, Kennedy EP, et al. Does type of pancreaticojejunostomy after pancreaticoduodenectomy decrease rate of pancreatic fistula? A randomized, prospective, dual-institution trial. J Am Coll Surg. 2009;208:738–747. [DOI] [PubMed] [Google Scholar]
- 107. Dinter DJ, Aramin N, Weiss C, et al. Prediction of anastomotic leakage after pancreatic head resections by dynamic magnetic resonance imaging (dMRI). J Gastrointest Surg. 2009;13:735–744. [DOI] [PubMed] [Google Scholar]
- 108. Nishida Y, Kato Y, Kudo M, et al. Preoperative sarcopenia strongly influences the risk of postoperative pancreatic fistula formation after pancreati-coduodenectomy. J Gastrointest Surg Off J Soc Surg Aliment Tract. 2016;20:1586–1594. [DOI] [PubMed] [Google Scholar]
- 109. Osman MM, Abd El Maksoud W.. Evaluation of a new modification of pancreaticogastrostomy after pancreaticoduodenectomy: anastomosis of the pancreatic duct to the gastric mucosa with invagination of the pancreatic remnant end into the posterior gastric wall for patients with cancer head of pancreas and periampullary carcinoma in terms of postoperative pancreatic fistula formation. Int J Surg Oncol. 2014;2014:490386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Palani Velu LK, Chandrabalan VV, Jabbar S, et al. Serum amylase on the night of surgery predicts clinically significant pancreatic fistula after pancreaticoduodenectomy. HPB. 2014;16:610–619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Shi Y, Liu Y, Gao F, et al. Pancreatic stiffness quantified with MR elastography: relationship to postoperative pancreatic fistula after pancreaticoenteric anastomosis. Radiology. 2018;288:476–484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Shimoda M, Katoh M, Yukihiro I, et al. Body mass index is a risk factor of pancreatic fistula after pancreaticoduodenectomy. Am Surg. 2012;78:190–194. [PubMed] [Google Scholar]
- 113. Suzuki S, Kaji S, Koike N, et al. Pancreaticojejunostomy of duct to mucosa anastomosis can be performed more safely without than with a stenting tube. Am J Surg. 2009;198:51–54. [DOI] [PubMed] [Google Scholar]
- 114. Zhu W, Li S, Zhang D, et al. Risk factors and outcome of pancreatic fistula after consecutive pancreaticoduodenectomy with pancreaticojejunostomy for patients with malignant tumor. Chin J Cancer Res. 2012;22:32–41. [Google Scholar]
- 115. Polanco PM, Zenati MS, Hogg ME, et al. An analysis of risk factors for pancreatic fistula after robotic pancreaticoduodenectomy: outcomes from a consecutive series of standardized pancreatic reconstructions. Surg Endosc. 2016;30:1523–1529. [DOI] [PubMed] [Google Scholar]
- 116. Casey P, Chaudhury MP, Khan A, et al. The impact of perio perative inotropes on the incidence of pancreatic leak following pancreaticoduodenectomy. Ann Hepatobiliary Pancreat Surg. 2019;23:392–396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Winter JM, Cameron JL, Campbell KA, et al. Does pancreatic ductstenting decrease the rate of pancreatic fistula following pancreaticoduodenectomy? Results of a prospective randomized trial. J Gastrointest Surg. 2006;10:1280–1290. [DOI] [PubMed] [Google Scholar]
- 118. Kollmar O, Moussavian MR, Bolli M, et al. Pancreatojejunal leakage after pancreas head resection: anatomic and surgeon-related factors. J Gastrointest Surg. 2007;11:1699–1703. [DOI] [PubMed] [Google Scholar]
- 119. Lee S-E, Yang S-H, Jang J-Y, et al. Pancreatic fistula after pancreaticoduo-denectomy: a comparison between the two pancreaticojejunostomy methods for approximating the pancreatic parenchyma to the jejunal seromuscular layer: interrupted vs continuous stitches. World J Gastroenterol. 2007;13:5351–5356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Liang T-B, Bai X-L, Zheng S-S. Pancreatic fistula after pancreaticoduode-nectomy: diagnosed according to International Study Group Pancreatic Fistula (ISGPF) definition. Pancreatology. 2007;7:325–331. [DOI] [PubMed] [Google Scholar]
- 121. Poon RTP, Fan ST, Lo CM, et al. External drainage of pancreatic duct with a stent to reduce leakage rate of pancreaticojejunostomy after pancreaticoduodenec-tomy: a prospective randomized trial. Ann Surg. 2007;246:425–433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Murakami Y, Uemura K, Hayasidani Y, et al. A soft pancreatic remnant is associated with increased drain fluid pancreatic amylase and serum CRP levels following pancreatoduodenectomy. J Gastrointest Surg. 2008;12:51–56. [DOI] [PubMed] [Google Scholar]
- 123. Ishizaki Y, Yoshimoto J, Sugo H, et al. Validation of mucosal sutureless pancreatojejunostomy after pancreatoduodenectomy. Am Surg. 2014;80:149–154. [PubMed] [Google Scholar]
- 124. Kajiwara T, Sakamoto Y, Morofuji N, et al. An analysis of risk factors for pancreatic fistula after pancreaticoduodenectomy: clinical impact of bile juice infection on day 1. Langenbecks Arch Surg. 2010;395:707–712. [DOI] [PubMed] [Google Scholar]
- 125. Liu Q-Y, Zhang W-Z, Xia H-T, et al. Analysis of risk factors for postoperative pancreatic fistula following pancreaticoduodenectomy. World J Gastroen-terol. 2014;20:17491–17497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Israel JS, Rettammel RJ, Leverson GE, et al. Does postoperative drain amylase predict pancreatic fistula after pancreatectomy? J Am Coll Surg. 2014;218:978–987. [DOI] [PubMed] [Google Scholar]
- 127. Jin S, Shi X-J, Wang S-Y, et al. Drainage fluid and serum amylase levels accurately predict development of postoperative pancreatic fistula. World J Gastroenterol. 2017;23:6357–6364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Graham JA, Johnson LB, Haddad N, et al. A prospective study of prophylactic long-acting octreotide in high-risk patients undergoing pancreatico-duodenectomy. Am J Surg. 2011;201:481–485. [DOI] [PubMed] [Google Scholar]
- 129. Belyaev O, Munding J, Herzog T, et al. Histomorphological features of the pancreatic remnant as independent risk factors for postoperative pancreatic fistula: a matched-pairs analysis. Pancreatology. 2011;11:516–524. [DOI] [PubMed] [Google Scholar]
- 130. Koga R, Yamamoto J, Saiura A, et al. Clamp-crushing pancreas transection in pancreatoduodenectomy. Hepatogastroenterology. 2009;56:89–93. [PubMed] [Google Scholar]
- 131. Belyaev O, Rosenkranz S, Munding J, et al. Quantitative assessment and determinants of suture-holding capacity of human pancreas. J Surg Res. 2013;184:807–812. [DOI] [PubMed] [Google Scholar]
- 132. Xingjun G, Feng Z, Meiwen Y, et al. A score model based on pancreatic steatosis and fibrosis and pancreatic duct diameter to predict postoperative pancreatic fistula after pancreatoduodenectomy. BMC Surg. 2019;19:75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Harrell KN, Jajja MR, Postlewait LM, et al. Influence of margin histology on development of pancreatic fistula following pancreatoduodenectomy. J Surg Res. 2020;246:315–324. [DOI] [PubMed] [Google Scholar]
- 134. Loos M, Strobel O, Dietrich M, et al. Hyperamylasemia and acute pancreatitis after pancreatoduodenectomy: two different entities. Surgery. 2021;169:369–376. [DOI] [PubMed] [Google Scholar]
- 135. Trudeau MT, Casciani F, Ecker BL, et al. The fistula risk score catalog: toward precision medicine for pancreatic fistula after pancreatoduodenectomy. Ann Surg. 2022;275:e463–e472. [DOI] [PubMed] [Google Scholar]
- 136. McMillan MT, Soi S, Asbun HJ, et al. Risk-adjusted outcomes of clinically relevant pancreatic fistula following pancreatoduodenectomy: a model for performance evaluation. Ann Surg. 2016;264:344–352. [DOI] [PubMed] [Google Scholar]
- 137. Miller BC, Christein JD, Behrman SW, et al. A multi-institutional external validation of the fistula risk score for pancreatoduodenectomy. J Gastrointest Surg Off J Soc Surg Aliment Tract. 2014;18:172–179. [DOI] [PubMed] [Google Scholar]
- 138. Ecker BL, McMillan MT, Asbun HJ, et al. Characterization and optimal management of high-risk pancreatic anastomoses during pancreatoduodenectomy. Ann Surg. 2018;267:608–616. [DOI] [PubMed] [Google Scholar]


