Abstract
Genetic vaccination is predicated on the underlying principle that diseases can be prevented by the controlled introduction of genetic material encoding antigenic proteins from pathogenic organisms to elicit the formation of protective immune responses. Driving this process is the choice of carrier that is responsible for navigating the obstacles associated with gene delivery. In this work, we expand upon a novel class of hybrid biosynthetic gene delivery vectors that are composed of a biomaterial outer coating and a bacterial (Escherichia coli) inner core. Specifically, a series of newly developed biodegradable cationic polylactides (CPLAs) and their PEGylated variants were selected to investigate the role of low polydispersity index (PDI), charge density, and PEGylation upon hybrid vector assembly and gene delivery efficacy. Upon assembly, hybrid vectors mediated increased gene delivery beyond that of the individual bacterial vector in isolation, including assays with increasing medium protein content to highlight shielding properties afforded by the PEG-functionalized CPLA component. Furthermore, after extensive characterization of surface deposition of the polymer, results prompted a new model for describing hybrid vector assembly that includes cellular coating and penetration of the CPLA component. In summary, these results provide new options and insight toward the assembly and application of next-generation hybrid biosynthetic gene delivery vectors.
Keywords: cationic polymer, polylactide, gene therapy, gene delivery, biosynthetic hybrid
Graphical Abstract
INTRODUCTION
Recently, the World Health Organization expressed concern over increasing antibiotic resistance and the specter of living in a “post-antibiotic era”1. Such concerns have prompted the search for other effective means of addressing infectious diseases with the equally powerful theme of preventative vaccination providing an alternative option. However, to date, only 27 human diseases are recognized by the US Center of Disease Control as preventable by vaccination despite centuries of development.2 Furthermore, current approaches often rely on technology that is limited in rapid production capability and associated engineering parameters to influence the type, duration, and potency of an immune response.
In this context, gene therapy offers a vaccination strategy based upon nucleic acid delivery to antigen presenting cells (APCs) as an initial step toward immune activation. Several delivery vectors have been used in this capacity including both biological and biomaterial options. Cationic polymers represent a common biomaterial vector; whereas bacteria have served as useful biological vectors in the context of APC delivery.3–5 Within the cationic polymer category, numerous synthetic schemes coupled to a wealth of fundamental polymer chemistry knowledge have given rise to various classes of polymers that feature facile synthesis, innate gene packaging properties, low cytotoxicity, and toolsets to permit rapid tailoring for specific applications. Examples include poly(beta-amino esters) (PBAEs) and cationic polylactides (CPLAs).6–12 Bacterial vectors such as Escherichia coli provide additional and alternative means to influence gene delivery with similarly low cytotoxicity and immunogenicity profiles.13–17 In particular, E. coli natively promotes phagocytic uptake by APCs, and upon internalization, lysosomal escape can be engineered through the heterologous expression of a pore-forming listeriolysin O (LLO) protein13,14,16,18,19 for cytoplasmic release of genetic cargo. Conversely, cationic polymers are internalized by generalized endocytosis or through specialized receptors grafted as targeting ligands.5 Postinternalization, cationic polymer mediates lysosomal escape by the “proton-sponge effect”.3
These innate and engineered features of bacteria and cationic polymers prompted the development of a hybrid biological–biomaterial antigen delivery vector.20 This new class of vector was composed of a cationic polymer outer coating and a bacterial (E. coli) inner core. The design provides an engineering-enhanced alternative to current gene delivery vectors and their production practices. Specifically, the hybrid vector allows for synergistic gene delivery as a function of combining the capabilities of the biological and biomaterial components of the overall vector. In addition, the engineering features of the hybrid device offer the potential to tune subsequent immune responses. Finally, the components of the hybrid vector were chosen for their proven safety profiles and ease of synthesis, which permits a future production process that is rapid, scalable, flexible, and economical in meeting emerging vaccination demands.
However, our previously selected cationic polymer component, PBAEs, despite their documented effectiveness, possessed high polydispersity indexes (PDIs), which complicate comparisons of effectiveness with each new PBAE tested in the hybrid format. Thus, in this study, we report the application of well-defined CPLAs and their PEGylated variants as a next-generation hybrid vector constituent. Effects of charge density and PEGylation on hybrid characteristics and gene delivery efficacy were systematically studied as was the effect of polymer addition upon hemolysis and immunogenicity. Furthermore, polymer-mediated bacterial extracellular release of protein and DNA was observed in a dose-dependent manner, suggesting revised structural features of the final hybrid device. Transfection with PEGylated hybrids demonstrated statistical improvements when compared to unaltered bacterial controls. In addition, gene delivery maintained in elevated levels of serum highlighted the positive effects of CPLA PEGylation when compared to unPEGylated hybrid vectors and points to the potential of utilizing shielding agents for future hybrid vector-mediated delivery applications.
EXPERIMENTAL SECTION
Measurements.
All 1H NMR spectra were measured at 500 MHz in CDCl3 using a Varian INOVA-500 spectrometer maintained at 25 °C with tetramethylsilane (TMS) as an internal reference standard. Gel permeation chromatography (GPC) data were acquired from a Viscotek GPC system equipped with a VE-3580 refractive index (RI) detector, a VE 1122 pump, and two mixed-bed organic columns (PAS-103 M and PAS-105M). Dimethylformamide (DMF; HPLC) containing 0.01 M LiBr was used as mobile phase with a flow rate of 0.5 mL/min at 55 °C. The GPC instrument was calibrated using narrowly dispersed linear polystyrene standards purchased from Varian. Zeta potential of hybrid vectors was obtained using dynamic light scattering (DLS) on a Zetasizer nano-ZS90 (Malvern, Inc.) in water at 25 °C. All experiments were conducted using a 4 mW 633 nm HeNe laser as the light source at a fixed measuring angle of 90° to the incident laser beam. The correlation decay functions were analyzed by cumulants method coupled with Mie theory to obtain volume distribution.
Materials.
4-Dimethylaminopyridine (DMAP; 99+%) and l-lactide (L-LA, 98%) were purchased from Sigma-Aldrich. 2,2′-Dimethoxy-2-phenylacetophenone (DMPA; 98%) was purchased from Acros Organics. Dichloromethane (DCM; HPLC), acetone (HPLC), ethyl acetate (HPLC), n-hexadecane (HPLC), DMF (HPLC), and diethyl ether (HPLC) were purchased from Fisher Chemical. α-Methoxy-ω-hydroxyl polyethylene glycol (mPEG-OH; MW, 2000 Da) was purchased from RAPP Polymere. 2-(Diethylamino)ethanethiol hydrochloride (DEAET, >98%) was purchased from Amfinecom Inc. DCM, DMF, and ethyl acetate were dried by distillation over CaH2. LA was recrystallized from dry ethyl acetate four times prior to use. mPEG-OH was dried as follows prior to use: mPEG-OH was dissolved in 1 mL of dried DCM, followed by complete solvent removal, and the cycle was repeated five times; toluene was used as a solvent to treat mPEG-OH for another five cycles. Allyl-functionalized LA monomer 1 was prepared through the method reported previously.8 All other chemicals were used without further purification.
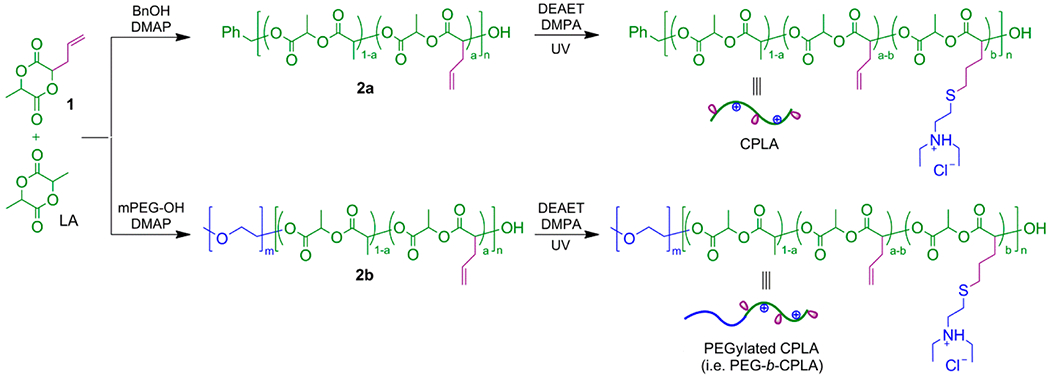
Synthesis of Allyl-Functionalized PLA (2a) and CPLA-26 and CPLA-54.
Allyl-functional PLA (2) was synthesized according to previous methodologies.6,8,21 Briefly, 1 (1440 mg; 10 mmol), L-LA (1700 mg; 10 mmol), and DCM (16.3 mL) were added to a 25 mL reaction flask with a magnetic stirring bar under nitrogen atmosphere. Upon reaching a solution temperature of 35 °C, BnOH (21.6 mg; 0.2 mmol; in 0.5 mL DCM) and DMAP (97.7 mg; 0.8 mmol; in 0.5 mL DCM) were added to initiate the polymerization. Synthesis was allowed to continue for 3 weeks at 35 °C, before being manually stopped at a comonomer conversion of ~80%. Comonomer conversion was calculated by 1H NMR based on the resonance intensities of the CH3 protons of remaining comonomers at 1.67–1.73 ppm relative to the CH3 protons of the resulting polymer at 1.49–1.61 ppm. Next, allyl-functionalized 2a was purified by precipitation in ice-cold methanol (50 mL).
1H NMR (500 MHz, CDCl3, ppm) of 2a: δ 1.49–1.61 (br m, CH3 units from LA and 1), 2.66–2.73 (br m, CH2CH=CH2 units from 1), 5.14–5.23 (br m, CHCH3 units from LA; CHCH3, CHCH2CH=CH2, and CH2CH=CH2 units from 1), 5.77–5.79 (m, CH2CH=CH2 units from 1), 7.33–7.39 (m, Ar–H from BnOH). ; ; PDIGPC = 1.12. Mole fraction of 1 was 54% based upon the 1H NMR resonance intensities of 1H from units of 1 at 5.77–5.79 ppm relative to 4H from units of 1 and 2H from units of LA at 5.14–5.23 ppm.
To synthesize CPLA-26, 2a (200 mg), DEAET (57.3 mg), and photoinitiator DMPA (34.8 mg) were dissolved in CDCl3 (5 mL) in a 10 mL flask, resulting in the molar ratio of [allyl of 2a]0/[SH of DEAET]0/[DMPA]0 = 1:0.5:0.2. The freeze–pump–thaw procedure was conducted for three cycles to deoxygenate the solution. Then, the thiol-ene reaction was induced by UV irradiation (λmax = 365 nm) for 30 min. Subsequently, to remove the unreacted DEAET and DMPA, dialysis of the resulting solution was conducted against acetone for 10 days using molecular porous membrane tubing (Spectra/Por Dialysis Membrane, Spectrum Laboratories Inc.) with an approximate molecular weight cut off (MWCO) at 3500 Da. Following dialysis, the solution was completely dried by vacuum to give CPLA-26 at 90% yield. Using a different feed ratio of reactants ([allyl of 2a]0/[SH of DETA]0/[DMPA]0 = 1:3:0.4), CPLA-54 was prepared using the same method applied to CPLA-26
1H NMR (500 MHz, CDCl3, ppm) of CPLA-26: δ 1.39–1.43 (br m, (CH3CH2)2NH+Cl− from amine-functionalized units), 1.49–1.61 (br m, CH3 from LA, 1, and amine-functionalized units), 1.76–1.79 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.00–2.10 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.49–3.23 (br m, CH2CH=CH2 units from 1; CH2CH2CH2SCH2 and SCH2CH2NH+Cl−(CH2CH3)2 from amine-functionalized units), 5.14–5.20 (br m, CHCH3 units from LA; CHCH3, CHCH2CH=CH2, and CH2CH=CH2 units from 1; CHCH3 units from 2a; and CHCH2CH2CH2SCH2 from amine-functionalized units), 5.77–5.79 (br m, CH2CH=CH2 units from 1), 7.33–7.39 (m, Ar–H from BnOH). ; ; PDIGPC = 1.37. 1H NMR (500 MHz, CDCl3, ppm) of CPLA-54: δ 1.17–1.26 (br m, (CH3CH2)2NH+Cl− from amine-functionalized units), 1.49–1.61 (br m, CH3 from LA and amine-functionalized units), 1.74–1.77 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.00–2.08 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.49–3.20 (br m CH2CH2CH2SCH2 and SCH2CH2NH+Cl−(CH2CH3)2 from amine-functionalized units), 5.14–5.20 (br m, CHCH3 units from LA; CHCH3 and CHCH2CH2CH2SCH2 from amine-functionalized units), 7.33–7.39 (m, Ar–H from BnOH). ; ; PDIGPC = 1.35.
Synthesis of PEG-b-allyl-Functionalized PLA (2b) and PEG-b-CPLAs.
In a 10 mL reaction flask with a magnetic stirring bar under nitrogen atmosphere, 1 (544 mg; 3.78 mmol), L-LA (643 mg; 3.78 mmol), and dried mPEG-OH (189 mg; 0.09 mmol) were added with dried DCM (4 mL). The solution was heated to 35 °C using an oil bath for 1 h, followed by the addition of a solution of DMAP (44 mg; 0.36 mmol) in 0.5 mL of dried DCM. After incubation for 1 week at 35 °C, the reaction was stopped at comonomer conversion of ~80% as determined by 1H NMR analysis of an aliquot of polymerization solution, based on the resonance intensities of the CH3 protons of remaining comonomers at 1.67–1.71 ppm relative to the CH3 protons of the resulting polymer at 1.49–1.59 ppm. The reaction mixture was precipitated by cold diethyl ether three times. Then the precipitate was collected and dried in a vacuum to give 2b as a white solid powder in 30% isolated yield. 1H NMR (500 MHz, CDCl3, ppm): δ 1.49–1.59 (br m, CH3 units from LA and 1), 2.67–2.73 (br m, CH2CH=CH2 units from 1), 3.38 (s, terminal CH3O units of mPEG-OH), 3.54–3.68 (br m, CH2O units of mPEG-OH), 5.14–5.30 (br m, CHCH3 units from LA; CHCH3, CHCH2CH=CH2 and CH2CH=CH2 units from 1), 5.77–5.79 (m, CH2CH=CH2 units from 1). , , PDIGPC = 1.05. The mole fraction of 1 in the PLA-based block was 50% based upon the 1H NMR resonance intensities of 1H from units of 1 at 5.77–5.79 ppm relative to 4H from units of 1 and 2H from units of LA at 5.14–5.30 ppm.
For the synthesis of PEG-b-CPLA-20, in a 10 mL flask, 2b (100 mg), DEAET (16.7 mg), and photoinitiator DMPA (10.15 mg) were dissolved in CDCl3 (5 mL) with molar ratio of [allyl of 2b]0/[SH of DEAET]0/[DMPA]0 = 1:0.5:0.2. To remove oxygen in the reaction solution, a freeze–pump–thaw procedure was carried out for three cycles. Subsequently, the thiol-ene reaction was induced by UV irradiation (λmax = 365 nm) for 30 min.
The reaction solution was dialyzed against acetone for 5 days using molecular porous membrane tubing (as described above for non-PEGylated CPLAs). Drying of the resulting solution in vacuum gave PEG-b-CPLA-20 with 87% yield. 1H NMR (500 MHz, CDCl3, ppm) of PEG-b-CPLA-20: δ 1.28–1.36 (br m, (CH3CH2)2NH+Cl− from amine-functionalized units), 1.51–1.63 (br m, CH3 from LA, 1, and amine-functionalized units), 1.76–1.81 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.00–2.04 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.64–3.20 (br m, CH2CH=CH2 units from 1; CH2CH2CH2SCH2 and SCH2CH2NH+Cl−(CH2CH3)2 from amine-functionalized units), 3.40 (s, terminal CH3O of mPEG-OH), 3.53–3.68 (br m, CH2O of mPEG-OH), 5.13–5.30 (br m, CHCH3 units from LA; CHCH3, CHCH2CH=CH2, and CH2CH=CH2 units from 1; CHCH3 units from 2b; CHCH2CH2CH2S from amine-functionalized units), 5.79–5.83 (br m, CH2CH=CH2 of units from 1). , , PDIGPC = 1.06.
Using a different feed ratio of reactants ([allyl of 2b]0/[SH of DETA]0/[DMPA]0 = 1:3:0.4), PEG-b-CPLA-50 was prepared using the same method applied to PEG-b-CPLA-20. 1H NMR (500 MHz, CDCl3, ppm) of PEG-b-CPLA-50: δ 1.28–1.37 (br m, (CH3CH2)2NH+Cl− from amine-functionalized units), 1.53–1.63 (br m, CH3 from LA and amine-functionalized units), 1.76–1.82 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.00–2.18 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.60–2.72 (br m, CH2CH2CH2SCH2 from amine-functionalized units), 2.80–3.20 (SCH2CH2NH+Cl−(CH2CH3)2 from amine-functionalized units), 3.40 (s, terminal CH3O of mPEG-OH), 3.53–3.68 (br m, CH2O of mPEG-OH), 5.12–5.25 (br m, CHCH3 units from LA; CHCH3 units from 2b; CHCH2CH2CH2S from amine-functionalized units). , , PDIGPC = 1.06.
Cell Lines and Reporter Plasmids.
A RAW264.7 (murine macrophage) cell line was provided by Dr. Terry Connell (Department of Microbiology and Immunology, University at Buffalo, SUNY). The cell line was maintained in medium prepared as follows: 50 mL of FBS (heat inactivated), 5 mL of 100 mM MEM sodium pyruvate, 5 mL of 1 M HEPES buffer, 5 mL of penicillin/streptomycin solution, and 1.25 g of d-(+)-glucose added to 500 mL of RPMI-1640 and filter sterilized. Cells were housed in T75 flasks and cultured at 37 °C/5% CO2.
The BL21(DE3) E. coli cell line (Novagen) was used as the parent strain (Strain 1 [S1]) containing a chromosomal insertion of listeriolysin O (LLO; hly gene designation) driven by a lac-inducible T7 promoter for all gene delivery bacterial vectors. Genetic manipulations were described previously.14,16 To determine in vitro transfection efficacy, we employed a luciferase reporter plasmid with expression driven by a cytomegalovirus promoter within pCMV-Luc (Elim Biopharmaceuticals). The pCMV-Luc plasmids was transformed into and isolated from an E. coli cloning host (GeneHogs, Invitrogen) using a PureYield Plasmid Midiprep System (Promega) prior to being used in the experiments outlined below.
Preparation of Gene Delivery Vectors.
Bacterial and hybrid vectors were prepared from bacterial cultures inoculated at 2% (v/v) from overnight starter cultures. Plasmid selection antibiotics were used as needed during bacterial culture within lysogeny broth (LB) medium. Following incubation at 36 °C and 250 rpm until 0.4 to 0.5 OD600, samples were induced with 0.1 mM isopropyl β-d-1-thiogalactopyranoside (IPTG) at 30 °C for 1 h. Bacterial vectors were then washed once and standardized to 0.5 OD600 in PBS; whereas bacterial strains to be used in hybrid vector formation were washed once and standardized to 1.0 OD600 in 25 mM NaOAc (pH 5.15). Polymer doses dissolved in chloroform were desiccated and resuspended in 25 mM NaOAc (pH 5.15) prior to equal volume addition to 1.0 OD600 bacterial strains. Hybrid vectors (final 0.5 OD600) and bacterial vectors in PBS were allowed to incubate at 22 °C for 15 min before being diluted into RPMI medium to produce desired multiplicity of infections (MOIs; ratio of the number of hybrid vectors to APCs).
Transfection Studies.
For gene delivery experiments, RAW264.7 cells were seeded into two different types of 96-well plates at 3 × 104 cells/well in 100 μL of antibiotic-free media and incubated for 24 h to allow attachment. A tissue culture-treated, flat-bottom, sterile, white, polystyrene 96-well plate was used for luciferase assays; whereas a tissue culture-treated, sterile, polystyrene 96-well plate was used for bicinchoninic acid (BCA) assessment, the 3-(4,5-dimethylthiazol-2-yl)-diphenyltetrazolium bromide (MTT) assay, and nitric oxide (NO) production.
Hybrid and bacterial vectors were diluted in antibiotic-free RPMI-1640 to desired MOIs. Following cellular attachment, macrophage medium was replaced with 50 μL of each respective vector and allowed to incubate for an hour. After incubation, 50 μL of gentamicin containing RPMI-1640 was added to each well to eliminate external/nonphagocytized vectors. Following an additional 24 h incubation (48 h after initial seeding), plates were analyzed for luciferase expression using the Bright Glo assay (Promega) and protein content using the Micro BCA Protein Assay Kit (Pierce) according to each manufacturer’s instructions. Gene delivery was calculated by normalizing luciferase expression by protein content for each well/plate. In addition, hybrid gene delivery is presented as a percentage of transfection of the bacterial control (S1) in 10% FBS.
Serum Inhibition to Transfection.
To determine if transfection was affected by the presence of increasing levels of serum, hybrid vectors were prepared and incubated with RAW264.7 cells (100 ng/well) in RPMI-1640 medium with 10, 20, 30, 40, 50, and 60% FBS for 24 h. Gene delivery was quantified as described above.
MTT Assay and Nitric Oxide Production.
Cytotoxicity resulting from hybrid vectors was determined by the MTT colorimetric assay. RAW264.7 cells were seeded and transfected as described above. Following a 24 h incubation after vector addition, cells were assayed with MTT solution (5 mg/mL), added at 10% v/v, for 3 h at 37 °C/5% CO2. Medium plus MTT solution was then aspirated and replaced by DMSO to dissolve the formazan reaction products. Following agitated incubation for 1 h, the formazan solution was analyzed using a microplate reader at 570 nm with 630 nm serving as the reference wavelength. Results are presented as a percentage of untreated cells (100% viability). NO production was measured using a Griess reagent kit (Promega, Madison, WI) according to the manufacturer’s instructions.
Characterization of Hybrid Devices.
Zeta potential of bacterial and hybrid vectors was measured by DLS. To measure surface hydrophobicity of bacteria before and after polymer additions, samples were analyzed using a modified microbial adhesion to hydrocarbon (MATH) assay.22,23 Briefly, bacterial and hybrid vectors were prepared and resuspended in PBS to a final 1.0 OD600. One milliliter of bacterial or hybrid vector was added to a clean glass tube in addition to 110 μL of n-hexadecane (10% v/v). Each sample was then vortexed for 1 min at setting 10 (Analog Vortex Mixer, Fisher Scientific) and allowed 15 min for phase separation. Using a clean Pasteur pipet, bacterial/hybrid vector solution was retrieved, taking care to avoid the hydrocarbon layer, and transferred to a cuvette for a final OD600 measurement. The percentage change of hydrophobicity is calculated using ΔHydrophobicity % = ((A600,initial – A600,final)/A600,initial). In addition, results were standardized by subtracting the untreated bacterial control (~20% hydrophobicity).
Hemolytic Activity Assay.
The hemolysis assay was modified from that previously described.24 Briefly, a 5% RBC (red blood cell)/PBS solution was prepared by washing sheep blood (HemoStat Laboratories) with PBS until the supernatant became clear of red color. Next, 100 μL of purified 5% RBC solution was incubated with 900 μL of polymer or hybrids in PBS at various concentrations and doses for 1 h at 37 °C. Triton-X 100 (1% solution) was used to construct a % blood lysis standard curve by altering the amount of blood added to each respective sample. For example, for 50 and 100% lysis, 50 and 100 μL of purified 5% RBC solution was mixed with 1% Triton-X (to 1 mL). PBS was used to generate negative controls. Samples were centrifuged and hemolysis quantified by measuring supernatant at 541 nm and comparing to the % blood lysis standard curve.
Bacterial Membrane Shear Studies.
Induced bacterial culture and hybrid vector samples (200 μL) were washed and resuspended in PBS, before being sonicated at 20% capacity for 5 s using a Branson 450D Sonifier (400 W, tapered microtip). Sonicated samples were then plated on LB agar plates and allowed to incubate for 24 h before counting colony forming units.
Protein and DNA Release.
To quantify the amount of protein and DNA released from the supernatant of CPLA-coated bacterial vectors, hybrid and bacterial vectors were prepared as described above. However, after the 15 min incubation, samples were pelleted and supernatants were measured for absorbance at 260 and 280 nm for DNA and protein quantification, respectively. Polymixin B (Krackeler Scientific) was added as a control to bacterial vectors at 0.5 mg/mL.
Statistical Evaluation.
Unless otherwise indicated, data presented were generated from three independent experiments. Error bars represent standard deviation values. All statistical significance comparisons were performed using a one-way ANOVA with Dunnett (to compare within groups) or Bonferroni (to compare across groups) post-tests.
RESULTS AND DISCUSSION
Polymer Synthesis and Strain Generation.
In our initial report of a hybrid biosynthetic gene delivery vector, we utilized PBAEs for surface modification. This class of polymers is recognized for ease of synthesis and significant transfection levels.9–12,25 However, such polymer classes possess relatively large PDI values (>1.4) that result in potential batch-to-batch variation of transfection and cytotoxicity responses. Also in our initial reports we observed that conjugation of targeting moieties (i.e., mannose) to polymer backbones reduced coalescence at elevated polymer doses (data not shown). However, the previous study did not investigate the effect of the most commonly used shielding agent, poly(ethylene glycol) (PEG). The current work thus investigates the utilization of a class of well-defined PEGylated and unPEGylated cationic polylactides (PEG-b-CPLA and CPLA) with varying charge densities.
Polymers were synthesized through living ring-opening polymerization and click functionalization (Scheme 1 and Table 1) in accordance with our previous reports.7,8 Our previous studies demonstrated that amine mol % of CPLAs and PEG-b-CPLAs was the most important structural factor to govern their hydrolysis rate, complexation ability with genetic material, and the transfection efficiency of the corresponding polymer–genetic material nanoplexes.7,8 Therefore, CPLAs and their PEG-b-CPLA counterparts with similar amine mol % were selected as the cationic polymer components in the current study. In addition, a singular E. coli strain was selected as the optimal choice to deliver a mammalian expression luciferase reporter plasmid based upon optimization studies conducted previously.15–17,20 The selected strain, YWT7-hly/pCMV-Luc (S1), contains an inducible LLO expression cassette (T7 promoter driven) chromosomally integrated into BL21(DE3) at the clpP gene location.16
Scheme 1.
Synthesis of Cationic Polylactides (CPLAs) and PEGylated Variants via Ring-Opening Polymerization and Thiol-ene Functionalization
Table 1.
Synthesis of CPLA and PEGylated Variants
| polymer | [ene]0/[SH]0/[DMPA]0 | amine (mol %)a | (kDa) | PDIGPCb | ref |
|---|---|---|---|---|---|
| CPLA-26 | 1:0.5:0.2 | 26 | 18.5 | 1.37 | 6, 8 |
| PEG-b-CPLA-20 | 1:0.5:0.2 | 20 | 6.2 | 1.06 | 7 |
| CPLA-54 | 1:3:0.4 | 54 | 22.8 | 1.35 | 6, 8 |
| PEG-b-CPLA-50 | 1:3:0.4 | 50 | 7.3 | 1.06 | 7 |
Determined by 1H NMR spectroscopy relative to repeat units of the CPLA block.
Relative to linear polystyrene standards.
Hybrid Vector Formation and Characterization.
The formation of hybrid biosynthetic gene delivery vectors is presumably driven by electrostatic interactions between positively charged polymers and the negatively charged outer membrane of E. coli, which permits the use of simple mixing schemes. This facile method of formulation is advantageous for future scalability studies as it eliminates complex formulation protocols and can be accomplished without the use of expensive equipment. Furthermore, for all presented studies involving hybrid vectors, formulations were prepared over a range of polymer doses (0.25, 0.5, 0.75, and 1.00 mg/mL) to assess the degree of impact of coating upon subsequent results.
Surface deposition of cationic polymers to the bacterial core results in a beneficial attenuation phenomenon that is driven by (1) a mild disruption of the outer bacterial membrane, (2) neutralization of excess charge of the polymer constituent, and (3) reduced exposure of immunogenic molecules such as LPS.20 To further test these initial observations, hemolysis was investigated in the context of the hybrid device and vectors in isolation (Figure 1A). As previously observed, incubation of polymers with RBCs resulted in dose-dependent hemolysis; whereas PEGylated polymers possessed the same trend but at statistically significant lower values.7 Conversely, incubation with the background strain of bacteria (BL21[DE3]) in isolation resulted in no apparent hemolysis (<2%). Furthermore, upon hybrid vector formation, statistically significant reductions of hemolysis for hybrids formulated with CPLA-26 and −54 (S1:C26 and S1:C54) and PEG-b-CPLA-20 and −50 (S1:PC20 and S1:P50) were observed for all concentrations. Reduction in hybrid-mediated hemolysis is presumably caused by polymer charge neutralization and suggests that higher dosages of hybrid vectors can be tolerated in future in vivo applications.
Figure 1.
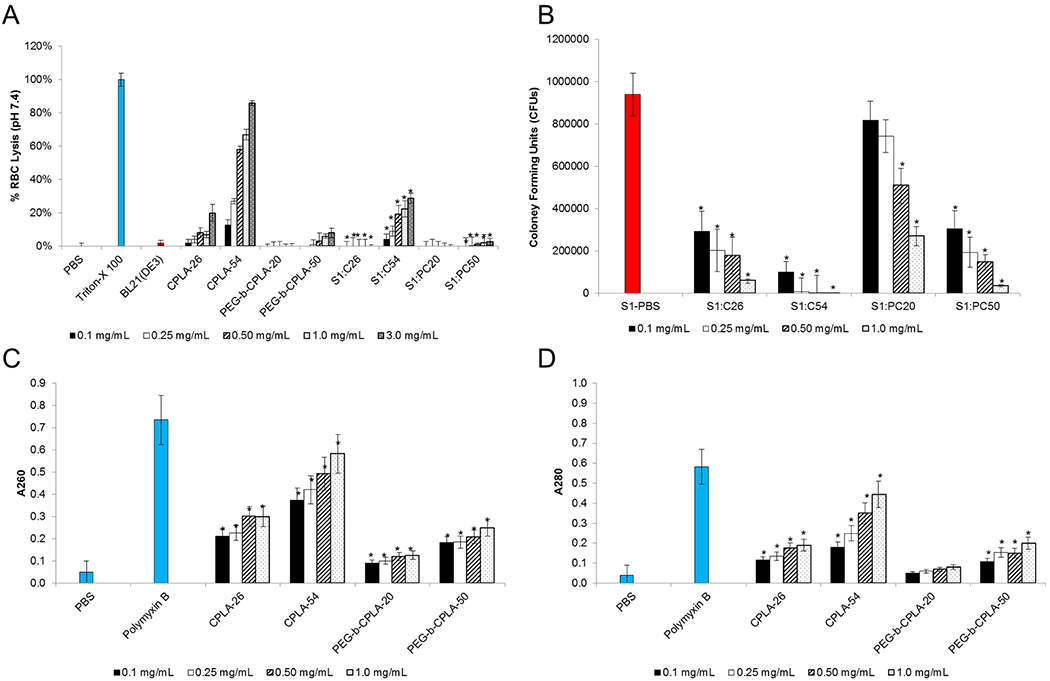
Biophysical characterization of hybrid vectors at various polymer doses. (A) Sheep red blood cell (RBC) hemolysis. (B) Hybrid membrane shear studies with 5 s sonication. (C) DNA and (D) protein release. *Statistical significance (95% confidence) compared to respective polymers (panel A) or bacterial (panel B) and PBS (panels C and D) controls. Abbreviations: CPLA-26, C26; CPLA-54, C54; PEG-b-CPLA-20, PC20; PEG-b-CPLA-50, PC50.
Aside from charge neutralization, cationic polymer surface deposition to the bacterial outer membrane may weakly disrupt the phospholipid bilayer in a mechanism similar to poreforming antibiotics (e.g., polymyxin B).15 Thus, to test this possibility and expand upon a potential reduction of bacterial viability driven by polymer surface additions, shear disruption studies were conducted by briefly sonicating (5 s) strain 1 in PBS (S1-PBS) and respective hybrid vectors (Figure 1B). With the exception of S1:PC20 at lower polymer doses, all hybrid vectors were significantly attenuated in a dose-dependent manner when compared to S1-PBS. Attenuation increased linearly with respect to total charge density (C54 > C26 > PC50 > PC20). Higher doses of CPLA-54 or PEG-b-CPLA-50 may potentially result in two opposing effects. Specifically, because of the intrinsic properties of the uncoated bacteria (i.e., uncoated bacteria mediate moderate APC cytotoxicity), polymer-mediated membrane destabilization of the bacterial cell wall has been previously linked to improvements upon APC gene delivery and cell viability;15 alternatively, the increased fragility of the bacterial membrane in the hybrid device may prompt premature clearance and/or vector decomposition. Furthermore, improvements to APC gene delivery and cytotoxicity resulting from bacterial membrane disruption are linked to the leakage of intracellular material. For instance, upon APC internalization, increased leakage of protein, specifically LLO, and plasmid DNA (pDNA) may further improve gene delivery by enhancing phagosomal escape and the concentration of genetic cargo available for transfection, respectively. Thus, pDNA and protein release studies were completed using hybrid vectors prepared as described above and compared to analogous bacterial treatments with PBS (negative control) and polymyxin B (positive control) (Figure 1C,D). Hybrid vectors demonstrated a statistically significant CPLA-mediated dose-dependent increase in release for both pDNA (A260) and protein (A280) for all polymers with the exception of PEG-b-CPLA-20 for protein release. Notably, S1:PC20 demonstrated the least attenuation potential, which may be correlated by the small molecular weight and/or the lack of increased charge density. Given the lack of bacterial core attenuation, this vector should only be utilized in a context where a stronger immunological response is required.
To further quantify the degree of surface modification, hybrid vectors were assessed for net surface charge using DLS (Figure S1A, Supporting Information). Bacterial surface charge transitioned to increasingly positively charged states in a dose- and charge density-dependent manner. Furthermore, increasing charge density, C54 > PC50 > C26 > PC20, mediated an increasing net surface charge trend. Analogously, surface modification of bacterial vectors with cationic polymers that possess large stretches of hydrophobic domains were expected to affect resulting hybrid vector polarity, noting that similar vector modifications have been associated with increases in gene delivery.3,5,12,26 Thus, using a MATH assay, hybrid vectors were assessed for increased relative hydrophobicity as compared to untreated bacterial controls (Figure S1B, Supporting Information). All polymers resulted in a general dose-dependent increase upon surface hydrophobicity. Interestingly, no statistically significant trends emerged related to charge density except at the lowest dose (0.25 mg/mL). At this dose, bacterial surface modification by CPLAs is driven predominately by charge difference between hybrid constituents. Hence, polymers with higher charge density are expected to provide better coverage at these polymer doses, leading to greater hydrophobicity measurements of the hybrid devices as a result. As coating increases with greater polymer dosing, the hydrophobicity levels between hybrid vectors are not statistically different.
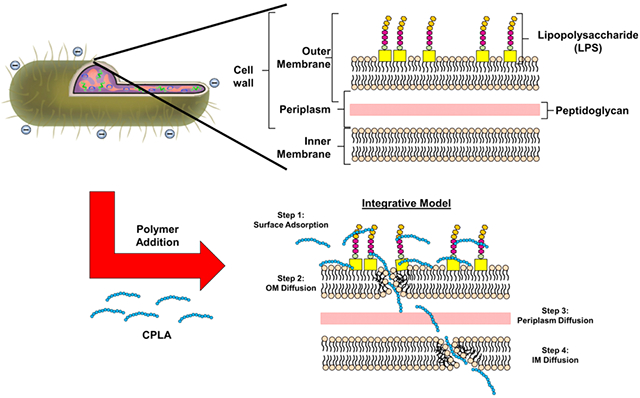
These studies and previous observations when taken together suggest that surface addition of cationic polymers results in the permeation of bacteria without causing gross bactericidal effects. Presumably the initial interaction between the bacterial surface and polymers is driven by electrostatic interactions before being replaced by membrane integration mechanisms. This “integrative” hypothesis stems from observations that polymers have both surface and membrane-spanning effects (Scheme 2). Surface coverage is supported by visual hybrid vector coalescence to form a larger biofilm-like structure upon increased doses of polymer20 and that NO production mediated by binding of LPS to external receptors of macrophages is significantly reduced upon polymer addition (to be presented later). Conversely, membrane-spanning effects include the extracellular release of protein and pDNA upon polymer addition, which would require double membrane permeation of the outer and inner phospholipid bilayers. Furthermore, given the estimated thickness of 30–40 nm for an intact Gram-negative bacterial surface (7.5–10 nm for each inner and outer membrane and 13–25 nm for the periplasmic space), it is unlikely that polymer molecules can span the entire space with the consideration of their molecular sizes.27 In addition, the mechanism of disruption most-likely resembles cationic antimicrobial polymers that act through a sequential series of steps.28 Accordingly, the first step presumably involves the initial surface adsorption (mediated primarily through charge–charge interactions; Scheme 1, Step 1), followed by diffusion and mild disruption of the outer membrane (Step 2).28 Lastly, upon diffusion through the outer membrane and the peptidoglycan layer, the polymer may again adsorb on the inner membrane (Step 3) before diffusing and disrupting (as before) the inner membrane (Step 4).
Scheme 2. Proposed Model of Hybrid Vector Formulation and Assemblya.
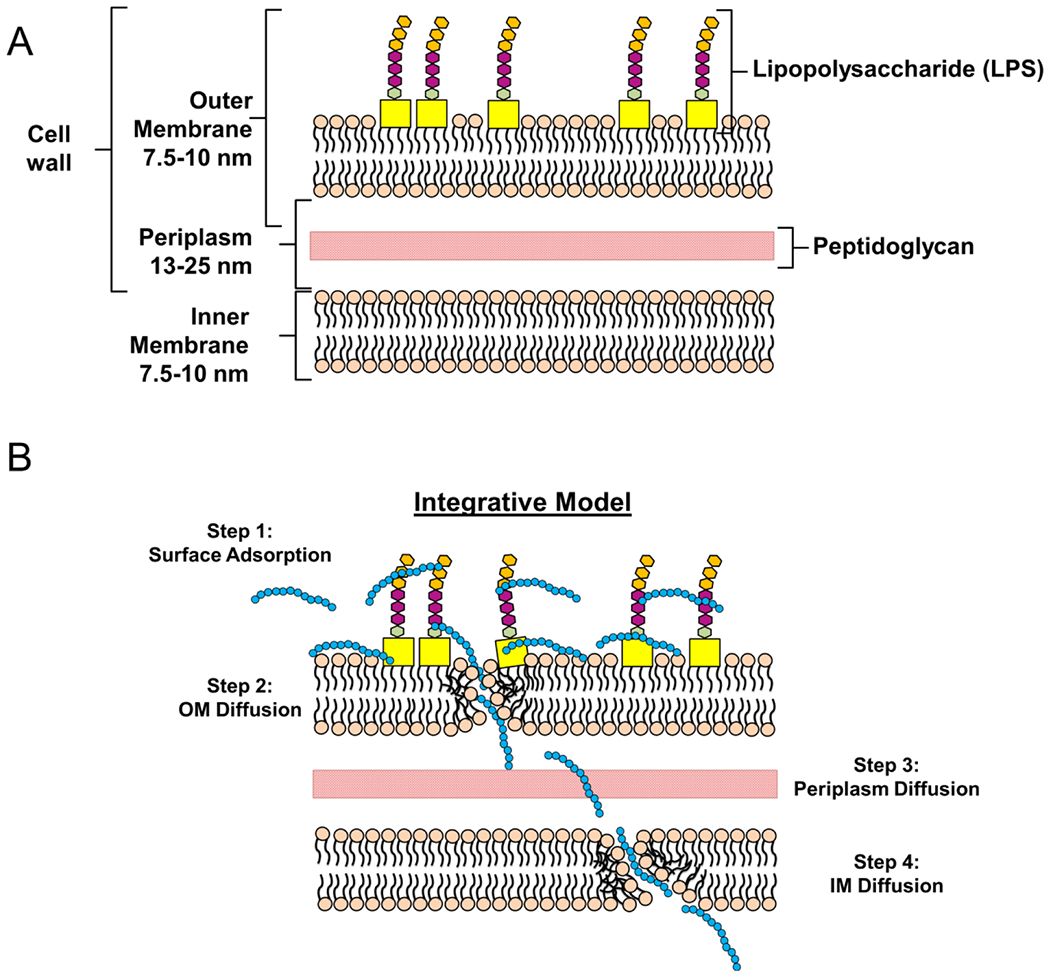
a(A) Layout of a normal cell wall of a Gram-negative bacterium. (B) The proposed hybrid formation model proceeds in four steps. First, polymer is adsorbed to the bacterial surface through charge–charge interaction (Step 1). Afterwards, the polymer diffuses slowly through the outer membrane (OM) while simultaneously compromising the structural integrity (Step 2). In the latter steps, the polymer chains diffuse slowly through the periplasmic space (Step 3) before subsequent integration and diffusion through the bacterial inner membrane (IM; Step 4). Taken together, these membrane disruptive steps are hypothesized to release internal bacterial cargo (i.e., DNA and protein) into the external periphery.
Transfection Studies.
Upon confirming bacterial surface modification and possible polymer–membrane integration, hybrid vectors were evaluated for gene delivery capabilities using a luciferase reporter model. Hybrid vectors composed of all four polymers at four doses were incubated with a murine macrophage cell line, RAW264.7, and assessed for luminescence (Figure 2). Gene delivery is reported as quotient of luminescence to total protein content of each respective sample. In addition, these values are further standardized by gene delivery values of S1. As such, values exceeding 100% represent improvements upon gene delivery as compared to the bacterial control in isolation.
Figure 2.
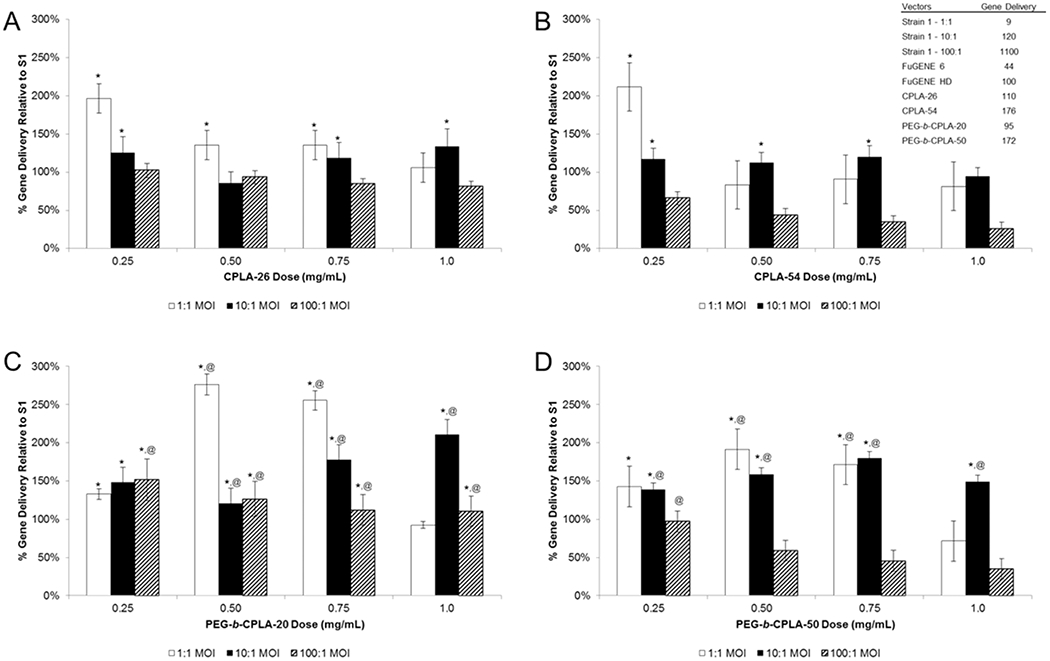
Gene delivery of hybrid vectors at various polymer doses formulated using (A) CPLA-26 and (B) CPLA-54 or (C) PEG-b-CPLA-20 and (D) PEG-b-CPLA-50. *Statistical significance (95% confidence) compared to Strain 1 (i.e., the 100% value). @Statistical significance (95% confidence) compared to hybrid vector prepared using respective nonPEGylated CPLA polymer. The table presented in panel B provides raw gene delivery values (luminescence per μg protein) of bacterial vectors (at various MOIs), commercial controls, and CPLA polyplexes (polymer complexed to pDNA).
Hybrids composed of unPEGylated CPLAs (Figure 2A,B) demonstrated a generally negative gene delivery correlation with respect to dose increases. In contrast, PEGylated hybrids showed less of an overall trend but demonstrated improved gene delivery with increased polymer dose for 1:1 and 10:1 MOI samples (Figure 2C,D). As a group, PEGylated hybrids resulted in gene delivery values that were improved in comparison to their unPEGylated counterparts. Interestingly, the S1:PC20 hybrid demonstrates the greatest gene delivery values. In comparison to the trends of Figure 1, we believe this is due to specific biophysical properties of the S1:PC20 hybrid enabling improved gene delivery at the indicated polymer dose levels. In addition, our previous studies reported reductions of hybrid-mediated gene delivery at doses higher than 0.5 mg/mL.20 This was believed to be caused by coalescence of hybrid vectors into conglomerates that were unable to be phagocytized and/or processed. The results presented here suggest that the addition of a shielding functionality prevents undesired coalescence, thus increasing the number of viable vectors per unit volume with increased polymer doses.
Serum Inhibition of Transfection.
For PEG-b-CPLA-based hybrid vectors to be relevant in in vivo applications, gene delivery must maintain efficacy in the presence of high concentrations of protein. The marked decrease of efficacy of transfection agents when shifting from in vitro models to an in vivo application is due to the deposition of negatively charged serum proteins to positively charged complexes, which in turn results in aggregation and clearance.5 As such, CPLA-54 and PEG-b-CPLA-50 hybrids were prepared and transfected with RAW264.7 cells with increasing v/v percentages of FBS (Figure 3). Values were standardized by gene delivery values from transfection using S1 in medium containing 10% FBS (normal transfection conditions). For both polymers, gene delivery was negatively correlated with increased levels of FBS. In addition, CPLA-54 hybrids demonstrated a dose-dependent decrease in gene delivery at all FBS levels except 10% FBS, but gene delivery remained statistically improved until 30% FBS. Higher doses of polymer decreased at a faster rate as compared to the lower doses due to increased deposition of FBS and aggregation. Conversely, PEG-b-CPLA-50 hybrids demonstrated statistically significant improvements in gene delivery until 50% FBS. Interestingly, at 60% FBS, polymer doses 0.50 and 0.75 mg/mL performed comparable to the S1 control in 10% FBS. This is significant because it is generally accepted that physiological serum levels range from 45 to 60% of volume.29 Unlike the unPEGylated hybrids, PEGylation resulted in a dose-dependent increase of gene delivery, with the exception of 1.0 mg/mL, across all FBS levels. Presumably this is associated with the innate properties of PEG to prevent coalescence and aggregation of particles resulting from serum deposition. Taken together, this is the first report indicating the importance of PEGylation (or any shielding molecules) and well-defined structural characteristics of the hybrid vector polymer constituent in preventing drastic reductions of gene delivery that is normally accompanied by increased levels of serum.
Figure 3.
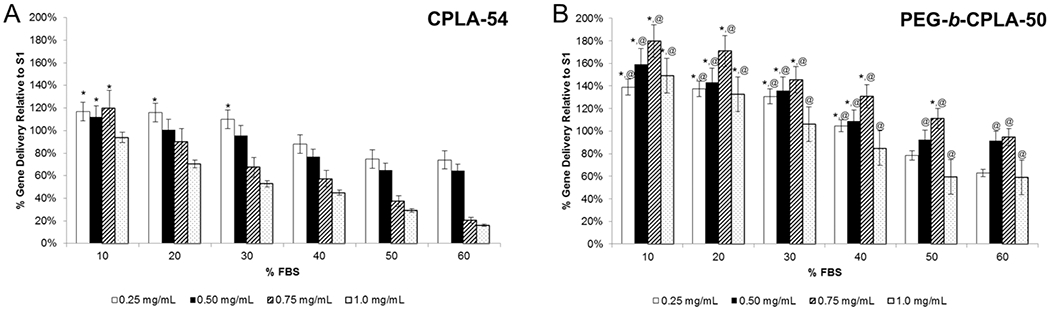
Serum inhibition study of gene delivery of hybrid vectors at various polymer doses formulated using (A) CPLA-54 or (B) PEG-b-CPLA-50. *Statistical significance (95% confidence) compared to S1 transfection in 10% FBS. @Statistical significance (95% confidence) compared to hybrid vector prepared using respective nonPEGylated CPLA polymer.
Cytotoxicity and NO Production.
For eventual translation, the hybrid technology must possess a safe cytotoxicity and immunogenicity profile. Thus, CPLA hybrids were examined for their cytotoxicity at four polymer doses and three MOIs (1:1, 10:1, and 100:1) (Figures 4 and S3, Supporting Information). At the lowest MOI, CPLA-26 hybrids demonstrated no cytotoxicity; whereas increased MOI levels were associated with increased cytotoxicity (Figure 4A). Conversely, use of a higher charge density polymer, CPLA-54 (>2 times the amine content of CPLA-26), resulted in a statistically significant dose-dependent increase in cytotoxicity at higher MOIs and doses when compared to the CPLA-26 hybrid (Figure 4B). This is likely the result of innate cytotoxicity associated with high-charge density polymers.6,7 Interestingly, formation of hybrids with PEGylated CPLAs resulted in a dose-dependent decrease in cytotoxicity for both polymers across all MOIs. The largest dose-dependent decrease of cytotoxicity occurred with the use of PEG-b-CPLA-20; however, use of PEG-b-CPLA-50 resulted in a significant decrease of cytotoxicity when compared to CPLA-54. Increased cell viability from PEGylated hybrids supports the claim that nontoxic polymers can provide an additional form of attenuation that has been tied with increased bacterial-mediated gene delivery.15
Figure 4.
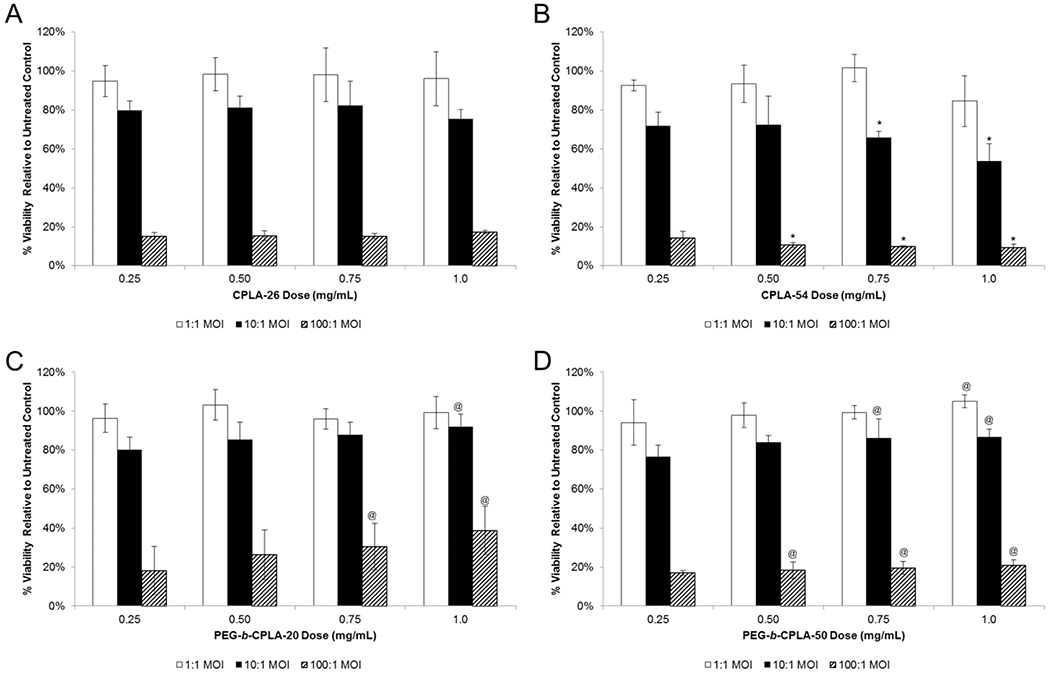
Cytotoxicity of RAW264.7 incubated with hybrid vectors at various polymer doses formulated using (A) CPLA-26 and (B) CPLA-54 or (C) PEG-b-CPLA-20 and (D) PEG-b-CPLA-50. *Statistical significant (95% confidence) decreases in viability of CPLA-54 hybrids compared to CPLA-26 hybrids. @Statistical significance (95% confidence) compared to hybrid vector prepared using respective nonPEGylated CPLA polymer.
Aside from cytotoxicity, a hybrid vector must reduce unwanted immunogenicity associated with the use of Gramnegative bacteria. As such, a Griess reagent assay was used to assess macrophage activation via lipopolysaccharide (LPS)-mediated NO production. Incubation of LPS with RAW264.7 cells is linked to concomitant release of TNF-α and NO via binding and activation of toll-like receptor 4 (TLR4). Although APC activation is required to elicit an effective immune response, excessive bacterial-mediated activation can result in systemic shock and potential death.4 Thus, we investigated NO production resulting from the incubation with hybrid vectors composed of CPLA-54 and PEG-b-CPLA-50 (Figure S2, Supporting Information). In both cases, polymer coating resulted in significant reduction of NO as compared to bacterial controls across all polymer doses and vector MOIs. Reduction in NO production presumably occurs by physical masking of LPS20 and/or competitive TLR4 binding.30,31 Additional confirmation of polymer coating improvements of cytotoxicity and immunogenicity highlights another advantage of the hybrid vector.
CONCLUSION
To elucidate the underlying properties that are responsible for the observed efficacy of a recently developed hybrid biosynthetic gene delivery vector, a set of well-defined PEGylated and unPEGylated cationic polylactides with tunable charge densities were utilized as the polymer constituent. Results indicate that this class of polymers effectively complements hybrid vector design and function and alters the previous model proposed for vector assembly. Accordingly, a new “integrative model” has been developed and presented to better align with experimental observations. In addition, PEGylation prevents coalescence of hybrid particles, thus providing a means to confer serum resistance and hemolysis reduction.
Supplementary Material
ACKNOWLEDGMENTS
The authors recognize support from NSF grants CBET-1019227 and DMR-1133737 (to C.C.), NIH award AI088485 (to B.A.P.), and a SUNY-Buffalo Schomburg fellowship (to C.H.J.).
Footnotes
Supporting Information
Physical characterization of hybrid vectors; NO production of RAW264.7; different presentation of data in Figure 4. This material is available free of charge via the Internet at http://pubs.acs.org.
The authors declare no competing financial interest.
REFERENCES
- (1).WHO’s First Global Report on Antibiotic Resistance Reveals Serious, Worldwide Threat to Public Health. http://www.who.int/mediacentre/news/releases/2014/amr-report/en/.
- (2).List of Vaccine-Preventable Diseases. http://www.cdc.gov/vaccines/vpd-vac/vpd-list.htm.
- (3).Pack DW; Hoffman AS; Pun S; Stayton PS Design and development of polymers for gene delivery. Nat. Rev. Drug Discovery 2005, 4 (7), 581–93. [DOI] [PubMed] [Google Scholar]
- (4).Parsa S; Pfeifer B Engineering bacterial vectors for delivery of genes and proteins to antigen-presenting cells. Mol. Pharmaceutics 2007, 4, 4–17. [DOI] [PubMed] [Google Scholar]
- (5).Jones CH; Chen CK; Ravikrishnan A; Rane S; Pfeifer BA Overcoming nonviral gene delivery barriers: perspective and future. Mol. Pharmaceutics 2013, 10 (11), 4082–4098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Jones CH; Chen CK; Jiang M; Fang L; Cheng C; Pfeifer BA Synthesis of cationic polylactides with tunable charge densities as nanocarriers for effective gene delivery. Mol. Pharmaceutics 2013, 10 (3), 1138–45. [DOI] [PubMed] [Google Scholar]
- (7).Chen CK; Jones CH; Mistriotis P; Yu Y; Ma X; Ravikrishnan A; Jiang M; Andreadis ST; Pfeifer BA; Cheng C Poly(ethylene glycol)-block-cationic polylactide nanocomplexes of differing charge density for gene delivery. Biomaterials 2013, 34 (37), 9688–99. [DOI] [PubMed] [Google Scholar]
- (8).Chen CK; Law WC; Aalinkeel R; Nair B; Kopwitthaya A; Mahajan SD; Reynolds JL; Zou J; Schwartz SA; Prasad PN; Cheng C Well-defined degradable cationic polylactide as nanocarrier for the delivery of siRNA to silence angiogenesis in prostate cancer. Adv. Healthcare Mater 2012, 1 (6), 751–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (9).Lynn DM; Langer R Degradable poly(β-amino esters): synthesis, characterization, and self-assembly with plasmid dna. J. Am. Chem. Soc 2000, 122, 10761–10768. [Google Scholar]
- (10).Sunshine JC; Sunshine SB; Bhutto I; Handa JT; Green JJ Poly(beta-amino ester)-nanoparticle mediated transfection of retinal pigment epithelial cells in vitro and in vivo. PLoS One 2012, 7 (5), e37543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (11).Sunshine JC; Peng DY; Green JJ Uptake and transfection with polymeric nanoparticles are dependent on polymer end-group structure, but largely independent of nanoparticle physical and chemical properties. Mol. Pharmaceutics 2012, 9 (11), 3375–3383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (12).Sunshine JC; Akanda MI; Li D; Kozielski KL; Green JJ Effects of base polymer hydrophobicity and end-group modification on polymeric gene delivery. Biomacromolecules 2011, 12 (10), 3592–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (13).Radford KJ; Higgins DE; Pasquini S; Cheadle EJ; Carta L; Jackson AM; Lemoine NR; Vassaux G A recombinant E. coli vaccine to promote MHC class I-dependent antigen presentation: application to cancer immunotherapy. Gene Ther. 2002, 9, 1455–1463. [DOI] [PubMed] [Google Scholar]
- (14).Higgins DE; Shastri N; Portnoy DA Delivery of protein to the cytosol of macrophages using Escherichia coli K-12. Mol. Microbiol 1999, 31, 1631–41. [DOI] [PubMed] [Google Scholar]
- (15).Jones CH; Rane S; Patt E; Ravikrishnan A; Chen CK; Cheng C; Pfeifer BA Polymyxin B Treatment Improves Bactofection Efficacy and Reduces Cytotoxicity. Mol. Pharmaceutics 2013, 10 (11), 4301–4308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (16).Parsa S; Wang Y; Rines K; Pfeifer BA A high-throughput comparison of recombinant gene expression parameters for E. coli-mediated gene transfer to P388D1 macrophage cells. J. Biotechnol 2008, 137 (1–4), 59–64. [DOI] [PubMed] [Google Scholar]
- (17).Parsa S; Wang Y; Fuller J; Langer R; Pfeifer BA A comparison between polymeric microsphere and bacterial vectors for macrophage p388d1 gene delivery. Pharm. Res 2008, 25, 1202–1208. [DOI] [PubMed] [Google Scholar]
- (18).Critchley RJ; Jezzard S; Radford KJ; Goussard S; Lemoine NR; Grillot-Courvalin C; Vassaux G Potential therapeutic applications of recombinant, invasive E. coli. Gene Ther. 2004, 11 (15), 1224–33. [DOI] [PubMed] [Google Scholar]
- (19).Grillot-Courvalin C; Goussard S; Courvalin P Wild-type intracellular bacteria deliver DNA into mammalian cells. Cell. Microbiol 2002, 4 (3), 177–86. [DOI] [PubMed] [Google Scholar]
- (20).Jones CH; Ravikrishnan A; Chen M; Reddinger R; Kamal Ahmadi M; Rane S; Hakansson AP; Pfeifer BA Hybrid biosynthetic gene therapy vector development and dual engineering capacity. Proc. Natl. Acad. Sci U.S.A 2014, 111 (34), 12360–12365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (21).Zou J; Hew CC; Themistou E; Li Y; Chen CK; Alexandridis P; Cheng C Clicking well-defined biodegradable nanoparticles and nanocapsules by UV-induced thiol-ene cross-linking in transparent miniemulsions. Adv. Mater 2011, 23 (37), 4274–7. [DOI] [PubMed] [Google Scholar]
- (22).Rosenberg M Microbial adhesion to hydrocarbons: twenty-five years of doing MATH. FEMS Microbiol. Lett 2006, 262 (2), 129–34. [DOI] [PubMed] [Google Scholar]
- (23).Geertsemadoornbusch GI; Vandermei HC; Busscher HJ Microbial cell-surface hydrophobicity - the involvement of electrostatic interactions in microbial adhesion to hydrocarbons (math). J. Microbiol. Methods 1993, 18 (1), 61–68. [Google Scholar]
- (24).Higgins DE; Shastri N; Portnoy DA Delivery of protein to the cytosol of macrophages using Escherichia coli K-12. Mol. Microbiol 1999, 31, 1631–41. [DOI] [PubMed] [Google Scholar]
- (25).Anderson DG; Lynn DM; Langer R Semi-automated synthesis and screening of a large library of degradable cationic polymers for gene delivery. Angew. Chem 2003, 42 (27), 3153–8. [DOI] [PubMed] [Google Scholar]
- (26).Liu MA DNA vaccines: an historical perspective and view to the future. Immunol. Rev 2010, 239, 62–84. [DOI] [PubMed] [Google Scholar]
- (27).Salton MRJ; Kim KS Structure. In Medical Microbiology, 4th ed.; Baron S, Ed.; University of Texas Medical Branch: Galveston, TX, 1996. [PubMed] [Google Scholar]
- (28).Kenawy el R; Worley SD; Broughton R The chemistry and applications of antimicrobial polymers: a state-of-the-art review. Biomacromolecules 2007, 8 (5), 1359–84. [DOI] [PubMed] [Google Scholar]
- (29).He XM; Carter DC Atomic structure and chemistry of human serum albumin. Nature 1992, 358 (6383), 209–15. [DOI] [PubMed] [Google Scholar]
- (30).Lee J; Sohn JW; Zhang Y; Leong KW; Pisetsky D; Sullenger BA Nucleic acid-binding polymers as anti-inflammatory agents. Proc. Natl. Acad. Sci U.S.A 2011, 108 (34), 14055–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (31).Chen H; Li P; Yin Y; Cai X; Huang Z; Chen J; Dong L; Zhang J The promotion of type 1 T helper cell responses to cationic polymers in vivo via toll-like receptor-4 mediated IL-12 secretion. Biomaterials 2010, 31 (32), 8172–80. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


