Abstract
Introduction
Limited evidence has been reported for surgical site infections (SSIs) in patients undergoing surgery who are carriers of extended-spectrum cephalosporin-resistant Enterobacterales (ESCR-E). A systematic review and meta-analysis were conducted to evaluate the risk of postoperative infections in adult inpatients colonised with ESCR-E before surgery.
Methods
The Medline, Embase and Cochrane databases were searched between January 2011 and April 2022, following PRISMA indications. Random effects meta-analysis was used to quantify the association between ESCR-E colonisation and infection.
Results
Among the 467 articles reviewed, 9 observational studies encompassing 7219 adult patients undergoing surgery were included. The ESCR-E colonisation rate was 13.7% (95% CI 7.7–19.7). The most commonly reported surgeries included abdominal surgery (44%) and liver transplantation (LT; 33%). The SSI rate was 23.2% (95% CI 13.2–33.1). Pooled incidence risk was 0.36 (95% CI 0.22–0.50) vs 0.13 (95% CI 0.02–0.24) for any postoperative infection and 0.28 (95% CI 0.18–0.38) vs 0.17 (95% CI 0.07–0.26) for SSIs in ESCR-E carriers vs noncarriers, respectively. In ESCR-E carriers, the ESCR-E infection ratio was 7 times higher than noncarriers. Postoperative infection risk was higher in carriers versus noncarriers following LT. Sources of detected heterogeneity between studies included ESCR-E colonisation and the geographic region of origin.
Conclusions
Patients colonised with ESCR-E before surgery had increased incidence rates of post-surgical infections and SSIs compared to noncarriers. Our results suggest considering the implementation of pre-surgical screening for detecting ESCR-E colonisation status according to the type of surgery and the local epidemiology.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40121-022-00756-z.
Keywords: Extended-spectrum cephalosporin-resistant Enterobacterales (ESCR-E), Rectal colonisation, Postoperative infections, Surgical site infections, Meta-analysis
Key Summary Points
| There is limited evidence to recommend rectal screening in patients colonised with ESCR-E before surgery. |
| We observed an increased postoperative risk of infections in ESCR-E carriers vs noncarriers, including surgical site infections. |
| Infections caused by ESCR-E after surgery were more common in ESCR-E carriers vs noncarriers. |
| Postoperative infection risk following liver transplant surgery was higher in carriers versus noncarriers. |
| High heterogeneity was noted among the studies in terms of type of surgery and prophylaxis. |
Introduction
Extended-spectrum cephalosporin-resistant Enterobacterales (ESCR-E) have been classified as a global urgent threat by scientific communities [1–3]. Over the last decades, these pathogens have been recognised as a main multidrug-resistant (MDR) causative agent in numerous infections, including surgical site infections (SSIs) [4]. SSIs are the most frequent postoperative complications, representing up to 31% of all hospital-acquired infections (HAIs) and showing high morbidity, leading to associated healthcare costs [5]. Recent studies report an increased burden of SSIs caused by MDR organisms, such as ESCR-E, and these infections are often associated with worse outcomes due to the limited therapeutic options available [6–8]. There is also emerging evidence that colonisation with ESCR-E is associated with a higher incidence of SSIs compared with a noncolonised status [9, 10]. The efficacy of perioperative antibiotic prophylaxis (PAP) in reducing SSIs has been clearly established, making it the standard of care in most surgical procedures [11]. Standard PAP in colorectal surgery, however, usually includes a cephalosporin combined with metronidazole (to provide coverage of the aerobic and anaerobic flora) and does not specifically target ESCR-E [11]. On the other hand, the use of culture-based PAP (e.g. based on the result of rectal cultures and susceptibility tests) to target MDR according to the results of rectal colonisation has raised concerns about the risk of increased carbapenem use following the detection of ESCR-E colonisation [12].
We conducted a systematic review and meta-analysis to evaluate the risk of postoperative infections, including SSIs, among adult hospitalised surgical patients who were ESCR-E carriers before surgery.
Methods
This systematic review and meta-analysis were conducted according to the PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) guidelines. This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors [13].
Search Strategy and Selection Criteria
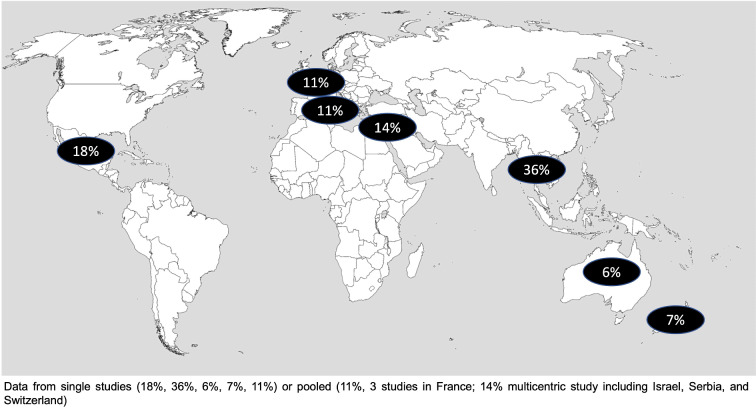
The study protocol is reported in the PROSPERO register of systematic reviews (CRD42021170244, http://www.crd.york.ac.uk/PROSPERO/) and is part of a wider review aimed at gathering the evidence for the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) recommendations for PAP in patients colonised with MDR-GNB bacteria before surgery. We searched the Medline, Embase and Cochrane databases for publications in any language between 1 January 2011 and 30 April 2022, documenting the occurrence of any type of infection caused by any pathogen following surgery in ESCR-E carriers vs noncarriers. Studies including adult (18 years of age and older) patients who were ESCR-E rectal carriers before surgery were retrieved. Bibliographies of reviews and original publications were also hand searched for further studies. All search strings were discussed with a qualified librarian. Details of the bibliographic search strategy are reported in Fig. 1.
Fig. 1.
Rectal colonisation rates by extended-spectrum cephalosporin-resistant Enterobacterales (ESCR-E) in different geographic regions
Data Extraction
All types of studies, except case reports and reviews, were considered for inclusion if they reported the rates of postoperative infections in hospitalised adult patients undergoing any type of surgery who were ESCR-E carriers and noncarriers before surgery. Two investigators independently assessed each potentially relevant study for eligibility. Disagreements were resolved by consultation with a third party. If eligibility could not be determined, the full article was retrieved. Bacterial isolates were considered ESCR-E if they complied with the local interpretive criteria. A standardised data extraction method was used to record the relevant characteristics of each study in an electronic database including country/WHO area, year of publication, study design, percentage of rectal colonisation [reported as (number of patients with a rectal swab that grew an ESCR-E)/(total number of patients screened for ESCR-E) × 100] before surgery, type and site of surgery, method of antimicrobial susceptibility testing, type of PAP used, outcome, and follow-up time. Patients were considered ESCR-E carriers if the bacteria were isolated from rectal or faecal samples without evidence of gastrointestinal infection. The outcomes assessed were the all-type postoperative infection incidence risk [measured as (number of patients developing any postoperative infection after surgery)/(total number of patients undergoing surgery)] as well as that of SSIs [measured as (number of patients developing an SSI)/(total number of patients undergoing surgery)] caused by any type of bacteria. Postoperative infections caused by ESCR-E were specifically searched for and analysed separately. Only articles reporting the outcomes in both ESCR-E carriers and noncarriers were included. Studies that did not allow data retrieval on ESCR-E colonisation, paediatric studies, nonhuman studies, or those including outpatient procedures were excluded. Duplicates were removed prior to study selection. Abstracts from conference proceedings were not included in the analysis.
Quality Assessment
The Newcastle–Ottawa Scale was used to evaluate the quality of each included study. Scores of 0–3, 4–6 and 7–8 were considered as indicative of low, moderate and high quality, respectively [14]. The criteria for the Newcastle–Ottawa scale regarding star allocation to assess the quality of studies (out of a total of eight stars) are reported in the Supplementary Material.
Data Analysis
Studies analysing the infection risk in ESCR-E carriers versus noncarriers were included in the meta-analysis. Infections were categorised as (1) postoperative infections, defined as any type of infection, including SSIs and other hospital-acquired infections (HAIs) such as urinary tract infections, abdominal infections, pneumonia, bacteremia, etc.; (2) SSIs; and (3) ESCR-E related infections after surgery. Because of the differences that were expected between studies, the results were combined using a random effects model [15]. Pooled incidence risk was measured using random-effects meta-analysis and was reported as the unadjusted risk ratio (RR) estimates and the 95% confidence intervals (95% CIs) for the noncolonised (nonexposed) and colonised (exposed) groups. Exposed to nonexposed ratio was reported for comparing group incidence. The odds ratio (OR) was also calculated to directly compare postoperative infections and SSIs in colonised and noncolonised patients, and is reported in the Supplementary Material. We prefer, however, to display the results separately for the exposed and nonexposed groups since the studies were all observational, and the formal meta-analysis of observational studies should not be a prominent component of these systematic reviews [16, 17].
If a study directly compared two different PAPs, each regimen was considered an independent study for the meta-analysis. A forest plot was generated to show the rates of ESCR-E colonisation according to the geographic area. Heterogeneity between studies was assessed using I2 tests (0–40% no heterogeneity, 30–60% moderate, 50–90% substantial, and 75–100% considerable heterogeneity) [18].
Four subgroup analyses were performed, excluding (1) non-liver-transplant (LT) patients, (2) transrectal ultrasound-guided prostate biopsy (TRUSPB) patients, (3) SSIs, and (4) patients not receiving PAP with potential ESCR-E activity.
Meta-regression analysis was implemented to account for potential sources of heterogeneity and confounding factors, including year of publication, geographic area, ESCR-E rectal colonisation, and study quality. Stata Statistical Software release 16 (StataCorp LLC, College Station, TX, USA) was used for analysis. Funnel plots are reported as supplementary material.
Results
A total of 467 studies were retrieved by literature search. After removing 126 duplicates, 341 studies were assessed by title and abstract. Of these, 283 were excluded and 58 were eligible for full-text reading. Among these, 25 did not address appropriate surgical populations, 21 did not include an outcome of interest, and 1 had only in vitro data. Eleven articles, including three retrieved by a bibliographic search of published papers, were included [6, 9, 10, 19–26]; of these, two were excluded (since a relevant number of carriers were colonised not only before but also after surgery) [22, 24] and nine were included, as reported in the PRISMA flowchart (Supplementary Fig. 1). All studies were observational and published between 2012 and 2021, providing data on 7219 adult surgical patients (Table 1). All studies except one were prospective [25] and conducted at a single centre: four in Europe (three in France [6, 19, 25], one in Italy [20]), one in Mexico [5] and one in Thailand [10]. Two studies were performed in the South Pacific area (one in Australia [26] and one in New Zealand [23]). Only one study was multicentric and included patients from Israel, Switzerland and Serbia [21]. Perioperative screening was performed through rectal swabs in all studies from 3 weeks to 1 hour before surgery. ESCR-E detection included the use of VITEK2, single disk diffusion (SDD), double disk diffusion (DDD), and chromogenic agar medium. The overall ESCR-E colonisation rate was 13.7% (95% CI 7.7–19.7). Figure 1 shows the prevalence (pooled or single-study reported prevalence) of ESCR-E colonisation per region, highlighting a considerable geographical variability. The type of surgery was highly heterogeneous and included LT recipients in three studies [6, 19, 25], abdominal or colorectal surgery in three cases [10, 19, 20], TRUSPB in two cases [2, 23], and gynaecologic/gastrointestinal surgery in one case [9]. The assessment of outcomes based on the type of PAP was reported by two authors. Bloomfield et al. [23] analysed patients undergoing TRUSPB and receiving perioperative ertapenem, while De Pastena et al. [20] compared the rate of infections between baseline and intervention periods in which ampicillin/sulbactam and piperacillin/tazobactam were used for prophylaxis in pancreatic surgery, respectively. In this study, postoperative infections during the baseline period were higher compared with the intervention period (43% versus 33%; p = 0.004). No differences were found in the occurrence of total SSIs (32% versus 28%; p = 0.14), while the superficial SSI rate significantly differed in the two periods (11% versus 2%; p < 0.001).
Table 1.
Characteristics of the included studies
| Author (publication year) | Study period | Study design | Country | Type of surgery | Number of patients | Outcome |
|---|---|---|---|---|---|---|
| Bert (2012) | 2001–2010 | Prospective | France | LT | 710 | Postoperative infections |
| Bert (2014) | 2009–2011 | Prospective | France | LT | 317 | Postoperative infections |
| Bloomfield (2017) | 2014–2016 | Prospective | New Zealand | TRUSPB | 281 | Postoperative infections |
| Apisarnthanarak (2019) | 2017–2019 | Prospective | Thailand | Abdominal | 360 | SSIs |
| Dubinsky-Pertzov (2019) | 2012–2017 | Prospective | Israel, Serbia, Switzerland | Colorectal | 3600 | SSIs |
| Golzarri (2019) | 2014–2015 | Prospective | Mexico | GI and GYN | 171 | Postoperative infections; SSIs |
| Sewell (2019) | 2012–2016 | Prospective | Australia | TRUSPB | 352 | Postoperative infections |
| De Pastena (2021) | 2015–2018 | Prospective, interventional, nonrandomised | Italy | Pancreatic | 679 | Postoperative infections; SSIs |
| Logre (2021) | 2010–2016 | Retrospective | France | LT | 749 | Postoperative infections; SSIs |
SSIs surgical site infections, GI gastrointestinal, GYN gynaecological, LT liver transplant, TRUSPB trans-rectal ultrasound-guided prostate biopsy
In three studies [9, 20, 25], both SSIs and postoperative non-SSIs were reported, while four studies showed the overall rate of postoperative infections (Table 1). The average rate of SSIs was 23.2% (95% CI 13.2–33.1) and was reduced, as expected, in prostate surgery (Table 2). Only four studies specifically reported the rates of ESCR-E infection in ESCR-E carriers vs noncarriers [10, 20, 21, 25]. Follow-up times to assess postoperative infections varied across studies (Table 2). Most studies were of moderate or low quality. A detailed quality assessment is presented in the Supplementary Material.
Table 2.
Infections in colonised and noncolonised surgical patients
| Author (publication year) | ESCR-E carriers (%) | Prophylaxis type | Postoperative infections in ESCR-E carriers vs noncarriers | Follow-up time (days) |
|---|---|---|---|---|
| Bert (2012) | 4 | Cefoxitin | 45% vs 4% (p < 0.0001) | 120 |
| Bert (2014) | 16 | Cefoxitin | 48% vs 7% (p < 0.001) | 120 |
| Bloomfield (2017) | 6 | Ertapenem | 11% vs 0.4% | 30 |
| Dubinsky-Pertzov (2019) | 14 | Cephalosporin + metronidazole |
SSIs 25% vs 11% (p < 0.001) ESCR-E SSIs 7% vs 2% |
30 |
| Sewell (2019) | 7 | Ciprofloxacin | 4% vs 1% | 30 |
| Golzarri (2019) | 18a | Cefuroxime (23%), metronidazole (12%), othersb |
SSIs 27% vs 11% ( p = 0.016) ESCR-E SSIs 11% vs 4% |
30 |
| Apisarnthanarak (2019)c | 36 | 2G Ceph (48%), 3G Ceph (25%), BLBLI (14%), carbapenems (12%) |
SSIs 31% vs 5% ESCR-E SSIs 6% vs 0% |
28 |
| De Pastena (2021)d | 11 | Ampicillin/sulbactam (56%), piperacillin/tazobactam (44%) |
54% vs 37% SSIs 42% vs 28% |
NA |
| Logre (2021)e | 13 | ESCR-E inactive (16%) and active (84%) | ESCR-E infections 45% vs 4% | 30 and 90 |
Ceph cephalosporin, BLBLI beta-lactam beta-lactamase inhibitor, SSIs surgical site infections, ESCR-E extended-spectrum cephalosporin-resistant Enterobacterales
aSSI results also included 7 post-surgical carriers
bCeftriaxone (10%), ciprofloxacin (5%), clindamycin (4%), cephalothin (1%)
cApisarnthanarak et al.: outcome data for carbapenems vs other PAP were not reported
dDe Pastena et al.: two study periods with two different PAPs were reported
eLogre et al.: among ESCR-E infections, 39% were caused by the same ESCR-E strains; only a subgroup of 57 patients received active PAP (cefoxitin 40%, a carbapenem 31%, piperacillin/tazobactam 29%)
Meta-analysis
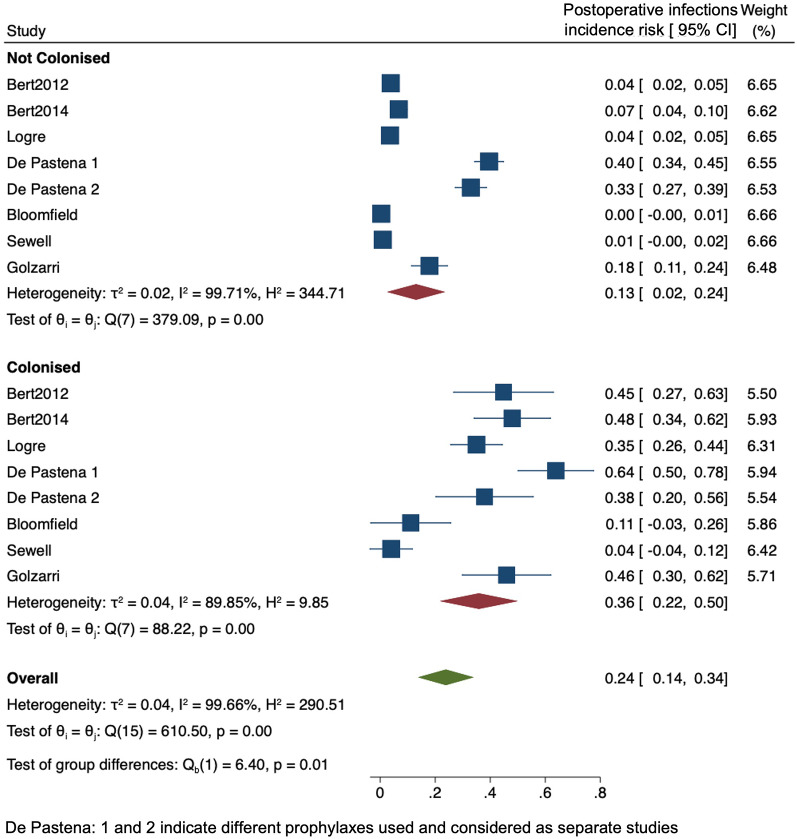
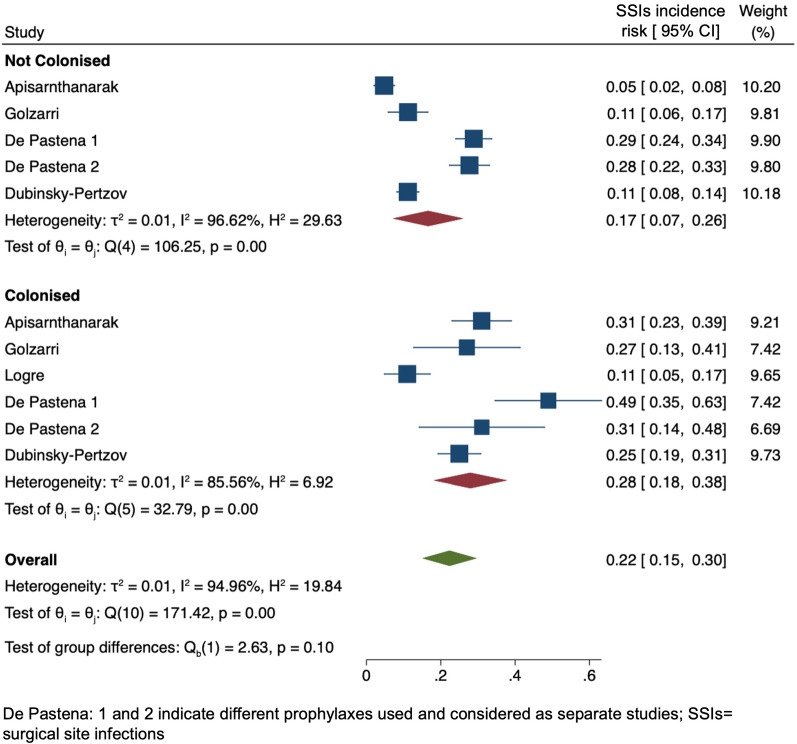
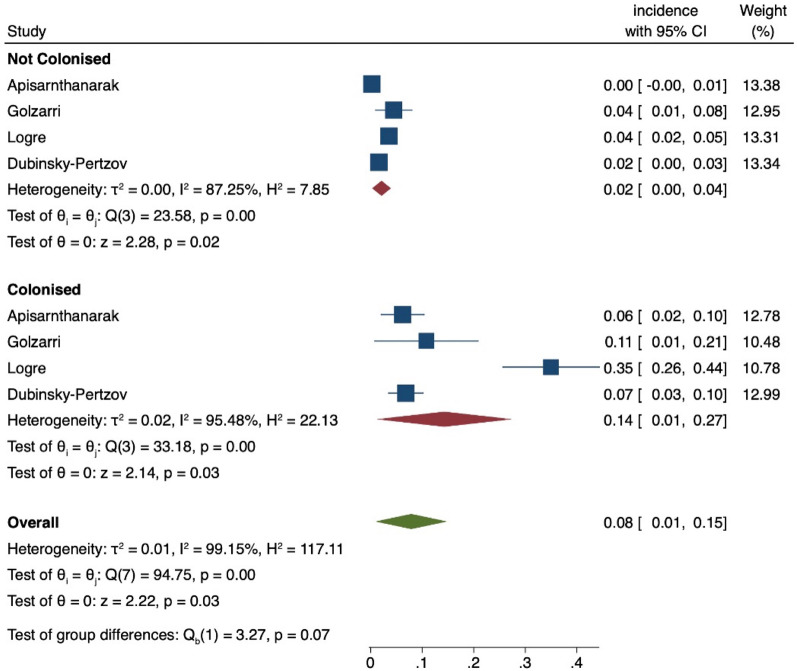
The distribution of postoperative infections according to the geographic area is reported in Supplementary Fig. 2. It could not be clearly estimated since most studies were performed in Europe and there is a known lower rate of infections following TRUSPB (as reported by two studies from the Western Pacific region) compared to other surgeries. Substantial or considerable heterogeneity is mainly shown. Figure 2 shows the forest plot for the studies reporting all postoperative infections, and Fig. 3 reports SSIs in ESCR-E-colonised and -noncolonised patients. ESCR-E carriers vs noncarriers had 2.8-fold and 1.6-fold higher rates of postoperative infections (0.36, 95% CI 0.22–0.50 vs 0.13, 95% CI 0.02–0.24) and SSIs (0.28, 95% CI 0.18–0.38 vs 0.17, 95% CI 0.07–0.26), respectively. The pooled OR confirmed an increase in postoperative infections and SSIs in colonised vs noncolonised patients (6.63, 95% CI 3.02–14.54 and 2.90, 95% CI 1.56–5.38, respectively), as reported in Supplementary Fig. 3. Figure 4 includes only the studies reporting the infections specifically caused by ESCR-E after surgery in colonised and noncolonised patients. ESCR-E infections were 7 times more common in ESCR-E carriers (0.14, 95% CI 0.01–0.27) vs noncarriers (0.02, 95% CI 0.02–0.04).
Fig. 2.
Forest plot of the included studies according to postoperative infection risk in extended-spectrum cephalosporin-resistant Enterobacterales (ESCR-E)-colonised and -noncolonised surgical patients. CI confidence interval
Fig. 3.
Forest plot of the included studies according to surgical site infection (SSI) infection risk in extended-spectrum cephalosporin-resistant Enterobacterales (ESCR-E)-colonised and -noncolonised surgical patients. CI confidence interval
Fig. 4.
Forest plot of the included studies according to the postoperative risk of infections due to ESCR-E among ESCR-E carriers and noncarriers
Subgroup Analyses
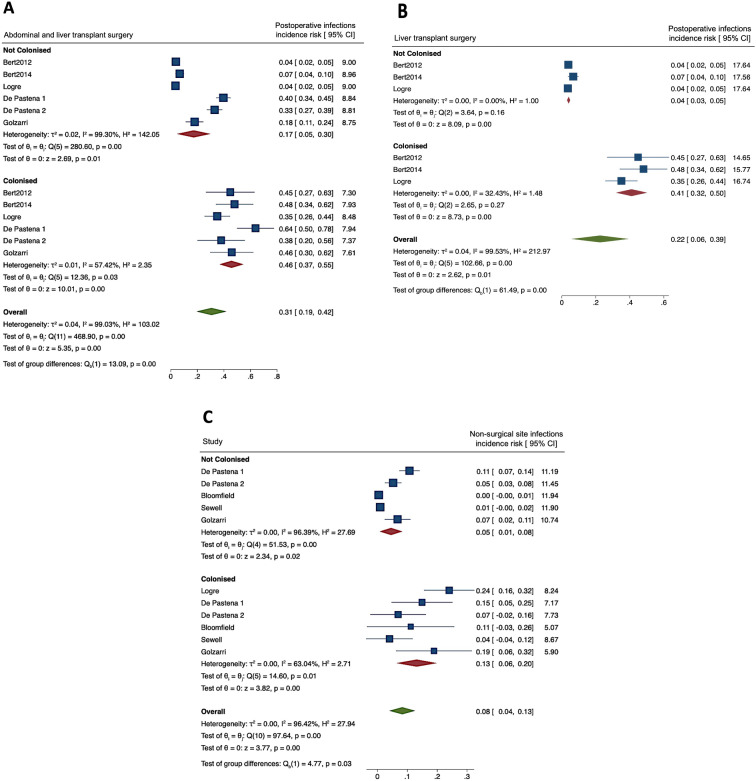
To identify differences according to the type of surgery, we excluded from the meta-analysis the two studies on TRUSBP, confirming 2.7 higher rates in ESCR-E carriers vs noncarriers (0.46, 95% CI 0.37–0.55 vs 0.17, 95% CI 0.05–0.30, Fig. 5A). To understand the impact of colonisation before LT, a subgroup analysis was performed excluding the studies in non-transplanted patients (Fig. 5B). Postoperative infections could only be analysed in three studies including LT recipients. In this group, the pooled incidence risk for postoperative infections was 0.41 (95% CI 0.32–0.50) vs 0.04 (95% CI 0.03–0.05) in ESCR-E carriers and noncarriers, respectively. SSIs were also subtracted, when possible, from the overall rate of postoperative infections, and a sensitivity analysis including only non-SSIs was performed (Fig. 5C). In this analysis, the difference between noncolonised and colonised patients was similar to the one reported in Fig. 1, although the overall incidence risk was reduced.
Fig. 5.
A Subgroup analysis including abdominal and liver transplant surgery. CI confidence interval. B Subgroup analysis including only studies performed in patients receiving liver transplant surgery. CI confidence interval. C Subgroup analysis that only included non-SSIs
A subgroup analysis was also performed that excluded patients receiving PAP regimens with potential ESCR-E activity. When only patients receiving routine PAP were included, we observed higher rates of postoperative infections in ESCR-E carriers (0.40, 95% CI 0.14–0.65) vs noncarriers (0.10, 95% CI −0.04–0.24) (Supplementary Fig. 4) rates. For SSIs, only two studies reported the data, so the results were not considered informative.
The meta-regression analysis showed that ESCR-E colonisation was associated with an increased incidence risk of postoperative infections (0.23, 95% CI 0.09–0.37, p = 0.001), while studies deriving from the Western Pacific area were associated with a decreased risk (− 0.26, 95% CI − 0.42 to − 0.10, p = 0.001). No other sources of heterogeneity were identified (e.g. year of publication, study quality); the same was confirmed for SSIs and ESCR-E postoperative infections.
Funnel plots for all postoperative infections and for SSIs are reported in Supplementary Fig. 5 and reflect the high heterogeneity of the studies included.
Discussion
Infections are a common complication following surgery. Among these, SSIs represent a threat, especially in patients undergoing colorectal surgery [11]. A meta-analysis assessing the efficacy of surgical prophylaxis in preventing postoperative infections showed that SSIs in colorectal surgery steadily increased between 1980 and 2005, irrespective of the type of antibiotic prophylaxis used and the type of surgical technique [27]. Due to an increase in the global circulation of ESCR-E in the past decades, it can be assumed that the current PAP may be suboptimal, especially in areas with a high burden of ESCR-E [27, 28]. Although the relationship between ESCR-E colonisation and increased risk of infection has been shown in patients with malignancies [29], very few studies have been conducted among surgical patients, and no meta-analyses were performed comparing infections between ESCR carriers and noncarriers. The evidence that ESCR-E colonisation precedes ESCR-E SSIs, however, remains limited due to the scarce number of studies specifically addressing this question.
Our review showed an increased proportion of SSIs and—in general—of postoperative infections in ESCR-E carriers compared to noncarriers. The fact that not only SSIs, but any HAIs may increase in patients with rectal colonisation by resistant bacteria has been previously reported in other patient populations [30]. These data support ESCR-E screening in surgical patients, especially in areas with high ESCR-E prevalence, and suggest that culture-based PAP may have an impact in reducing postoperative infections in ESCR-E carriers. The subgroup analysis excluding PAP with a potential ESCR-E effect showed an increased difference in the infection rate between carriers and noncarriers, suggesting a potential mitigating effect of personalised PAP on postoperative infections; nevertheless, there were not enough studies to quantitatively assess SSIs or postoperative infections according to the different types of PAP.
The local epidemiology (e.g. colonisation prevalence) should be always taken into consideration if screening policies are implemented or PAP changes are planned. As shown, certain areas may have low rates of ESCR-E colonisation and a reduced impact on subsequent infections. Data for analysing ESCR-E infection risk according to different geographic areas were limited, however, since most studies were performed in Europe, while those from the Western Pacific area included only TRUSBP, which is known to have lower rates of postoperative infections (around 1%) compared to other surgeries. In contexts such as LT, ESCR-E screening and, in general, a knowledge of MDR-GNB carrier status may be useful in enhancing infection control measures and/or directing empiric therapy in cases of severe infections [31]. In our study, when patients undergoing LT were analysed, the difference between ESCR-E carriers and noncarriers appeared remarkable (e.g. 10 times higher rates of infections in carriers vs noncarriers). These results, however, should be interpreted with caution, as only three reports (two from the same author) were included.
The rationale for targeted PAP appears more obvious in abdominal surgery, and specifically in colorectal surgery, due to the extensive procedures involving the opening of the intestinal lumen, which contains faecal flora. One prospective, non-randomised interventional study compared routine with ESCR-targeted prophylaxis using ertapenem in 478 patients undergoing elective colorectal surgery. The rates of SSIs caused by any pathogen were 22.7% vs 15.8% and the rates of those caused by ESCR-E were 6.5% vs 0.9% in patients receiving standard compared to ertapenem PAP, respectively, with an adjusted risk difference of − 7.7% (95% CI − 14.6% to − 0.8%) and − 5.6% (95% CI − 8.9% to − 2.3%), respectively. During the intervention phase, however, the adherence to the study protocol was suboptimal, as 21% of the ESCR-E carriers did not receive ertapenem [32].
Our study has several limitations. Firstly, only observational studies were available for inclusion. Moreover, there was high variability between studies according to the type of surgery (e.g. postoperative infections are more commonly reported in pancreatic surgery and reduced in TRUSBP), which makes pooled estimates difficult to generalise to all surgical adult populations. When only abdominal surgery and LT were analysed, the results were in line with the previous ones showing increased postoperative infections in ESCR-E carriers vs noncarriers. Secondly, the studies included varied by timing of ESCR-E screening, type of infections assessed, and follow-up duration. We could not clearly compare SSIs vs other HAIs due to the limited number of studies specifically dissecting the type of postoperative infections. Finally, only inpatient surgery was considered, potentially limiting the generalisability of the results, particularly to TRUSBP, which can be also performed as an ambulatory procedure.
Nevertheless, our findings are relevant in showing that a substantial burden of infection is associated with ESCR-E colonisation in surgical patients. This patient population is more likely to develop infections when rectal colonisation by ESCR-E is detected before surgery compared to its noncolonised counterpart, and these figures may further increase according to the type of surgery (e.g. LT) and if ESCR-E infections are considered.
Conclusions
In conclusion, we conducted a systematic review and meta-analysis to evaluate the effect of ESCR-E colonisation on the development of subsequent infections in surgical patients. This study adds to the evidence base since, to our knowledge, no meta-analyses have been previously performed in this group. The increased burden of infection in colonised vs noncolonised patients may support the need for ESCR-E colonisation detection among patients undergoing surgery, especially in areas with high ESCR-E prevalence and in LT recipients. Further research and well-designed studies, including randomised clinical trials, are needed to understand the benefits and impact of targeted antimicrobial prophylaxis in ESCR-E-colonised patients undergoing surgery to reduce the burden of postoperative MDR infections.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
We thank Valeria Scotti (IRCCS Policlinico San Matteo) and Burcu Isler (University of Queensland Centre for Clinical Research) for their help with the data search. They did not receive funding for their support.
Funding
No funding or sponsorship was received for this study or the publication of this article.
Author Contributions
Elda Righi and Evelina Tacconelli conceived the study; Alessandro Visentin, Marco Meroi, Anna Schwabe, Anna Erbogasto, Gianluca Vantini and Elizabeth Cross contributed to the data search and data extraction; Luigia Scudeller and Massimo Mirandola performed statistical analysis; Nico T. Mutters, Maddalena Giannella and Xavier Guirao critically reviewed the paper. Elda Righi drafted the manuscript.
Prior Presentation
Part of this work was presented as a poster at the 32nd European Congress of Clinical Microbiology and Infectious Diseases (ECCMID), Lisbon, 23–26 April 2022.
Disclosures
Elda Righi, Luigia Scudeller, Massimo Mirandola, Alessandro Visentin, Nico T. Mutters, Marco Meroi, Anna Schwabe, Anna Erbogasto, Gianluca Vantini, Elizabeth Cross, Maddalena Giannella, Xavier Guirao and Evelina Tacconelli have nothing to disclose.
Compliances with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
All data generated or analysed during this study are included in this published article and as supplementary information files.
References
- 1.Tacconelli E, Carrara E, Savoldi A, Harbarth S, Mendelson M, Monnet DL, et al. Discovery, research, and development of new antibiotics: the WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect Dis. 2018;18(3):318–327. doi: 10.1016/S1473-3099(17)30753-3. [DOI] [PubMed] [Google Scholar]
- 2.Williams DN. Antimicrobial resistance: are we at the dawn of the post-antibiotic era? J R Coll Physicians Edinb. 2016;46(3):150–156. doi: 10.4997/jrcpe.2016.302. [DOI] [PubMed] [Google Scholar]
- 3.Bajaj P, Singh NS, Virdi JS. Escherichia coli beta-lactamases: what really matters. Front Microbiol. 2016;7:417. doi: 10.3389/fmicb.2016.00417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rodriguez-Bano J, Pascual A. Clinical significance of extended-spectrum beta-lactamases. Expert Rev Anti Infect Ther. 2008;6(5):671–683. doi: 10.1586/14787210.6.5.671. [DOI] [PubMed] [Google Scholar]
- 5.Badia JM, Casey AL, Petrosillo N, Hudson PM, Mitchell SA, Crosby C. Impact of surgical site infection on healthcare costs and patient outcomes: a systematic review in six European countries. J Hosp Infect. 2017;96(1):1–15. doi: 10.1016/j.jhin.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 6.Bert F, Larroque B, Dondero F, Durand F, Paugam-Burtz C, Belghiti J, et al. Risk factors associated with preoperative fecal carriage of extended-spectrum beta-lactamase-producing Enterobacteriaceae in liver transplant recipients. Transpl Infect Dis. 2014;16(1):84–89. doi: 10.1111/tid.12169. [DOI] [PubMed] [Google Scholar]
- 7.Linares L, Cervera C, Cofan F, Ricart MJ, Esforzado N, Torregrosa V, et al. Epidemiology and outcomes of multiple antibiotic-resistant bacterial infection in renal transplantation. Transplant Proc. 2007;39(7):2222–2224. doi: 10.1016/j.transproceed.2007.06.061. [DOI] [PubMed] [Google Scholar]
- 8.Patel G, Huprikar S, Factor SH, Jenkins SG, Calfee DP. Outcomes of carbapenem-resistant Klebsiella pneumoniae infection and the impact of antimicrobial and adjunctive therapies. Infect Control Hosp Epidemiol. 2008;29(12):1099–1106. doi: 10.1086/592412. [DOI] [PubMed] [Google Scholar]
- 9.Golzarri MF, Silva-Sanchez J, Cornejo-Juarez P, Barrios-Camacho H, Chora-Hernandez LD, Velazquez-Acosta C, et al. Colonization by fecal extended-spectrum beta-lactamase-producing Enterobacteriaceae and surgical site infections in patients with cancer undergoing gastrointestinal and gynecologic surgery. Am J Infect Control. 2019;47(8):916–921. doi: 10.1016/j.ajic.2019.01.020. [DOI] [PubMed] [Google Scholar]
- 10.Apisarnthanarak A, Kondo S, Mingmalairak C, Mahawongkajit P, Juntong J, Limpavitayaporn P, et al. Outcomes of extended-spectrum beta-lactamases producing Enterobacteriaceae colonization among patients abdominal surgery patients. Infect Control Hosp Epidemiol. 2019;40(11):1290–1293. doi: 10.1017/ice.2019.254. [DOI] [PubMed] [Google Scholar]
- 11.Bratzler DW, Dellinger EP, Olsen KM, Perl TM, Auwaerter PG, Bolon MK, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Surg Infect (Larchmt) 2013;14(1):73–156. doi: 10.1089/sur.2013.9999. [DOI] [PubMed] [Google Scholar]
- 12.Allegranzi B, Zayed B, Bischoff P, Kubilay NZ, de Jonge S, de Vries F, et al. New WHO recommendations on intraoperative and postoperative measures for surgical site infection prevention: an evidence-based global perspective. Lancet Infect Dis. 2016;16(12):e288–e303. doi: 10.1016/S1473-3099(16)30402-9. [DOI] [PubMed] [Google Scholar]
- 13.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol. 2009;62(10):1006–12. [DOI] [PubMed]
- 14.Wells B, Saskin R, Wright F, McCready D, Quan ML. Measuring the quality of sentinel lymph node biopsy (SLNB) for breast cancer: a population-based evaluation. Ann Surg Oncol. 2013;20(2):615–619. doi: 10.1245/s10434-012-2626-6. [DOI] [PubMed] [Google Scholar]
- 15.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 16.Metelli S, Chaimani A. Challenges in meta-analyses with observational studies. Evid Based Ment Health. 2020;23(2):83–87. doi: 10.1136/ebmental-2019-300129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Egger M, Schneider M, Davey SG. Spurious precision? Meta-analysis of observational studies. BMJ. 1998;316(7125):140–144. doi: 10.1136/bmj.316.7125.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–1558. doi: 10.1002/sim.1186. [DOI] [PubMed] [Google Scholar]
- 19.Bert F, Larroque B, Paugam-Burtz C, Dondero F, Durand F, Marcon E, et al. Pretransplant fecal carriage of extended-spectrum beta-lactamase-producing Enterobacteriaceae and infection after liver transplant, France. Emerg Infect Dis. 2012;18(6):908–916. doi: 10.3201/eid1806.110139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.De Pastena M, Paiella S, Azzini AM, Zaffagnini A, Scarlini L, Montagnini G, et al. Antibiotic prophylaxis with piperacillin-tazobactam reduces post-operative infectious complication after pancreatic surgery: an interventional, non-randomized study. Surg Infect (Larchmt) 2021;22(5):536–542. doi: 10.1089/sur.2020.260. [DOI] [PubMed] [Google Scholar]
- 21.Dubinsky-Pertzov B, Temkin E, Harbarth S, Fankhauser-Rodriguez C, Carevic B, Radovanovic I, et al. Carriage of extended-spectrum beta-lactamase-producing Enterobacteriaceae and the risk of surgical site infection after colorectal surgery: a prospective cohort study. Clin Infect Dis. 2019;68(10):1699–1704. doi: 10.1093/cid/ciy768. [DOI] [PubMed] [Google Scholar]
- 22.Freire MP, Villela Soares Oshiro IC, Bonazzi PR, Pierrotti LC, de Oliveira LM, Machado AS, et al. Surveillance culture for multidrug-resistant gram-negative bacteria: performance in liver transplant recipients. Am J Infect Control. 2017;45(3):e40–e44. [DOI] [PubMed]
- 23.Bloomfield MG, Page MJ, McLachlan AG, Studd RC, Blackmore TK. Routine ertapenem prophylaxis for transrectal ultrasound guided prostate biopsy does not select for carbapenem resistant organisms: a prospective cohort study. J Urol. 2017;198(2):362–368. doi: 10.1016/j.juro.2017.03.077. [DOI] [PubMed] [Google Scholar]
- 24.Macesic N, Gomez-Simmonds A, Sullivan SB, Giddins MJ, Ferguson SA, Korakavi G, et al. Genomic surveillance reveals diversity of multidrug-resistant organism colonization and infection: a prospective cohort study in liver transplant recipients. Clin Infect Dis. 2018;67(6):905–912. doi: 10.1093/cid/ciy199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Logre E, Bert F, Khoy-Ear L, Janny S, Giabicani M, Grigoresco B, et al. Risk factors and impact of perioperative prophylaxis on the risk of extended-spectrum beta-lactamase-producing enterobacteriaceae-related infection among carriers following liver transplantation. Transplantation. 2021;105(2):338–345. doi: 10.1097/TP.0000000000003231. [DOI] [PubMed] [Google Scholar]
- 26.Sewell J, Chow K, De Sousa A, Sapre N, Norris B, Namdarian B, et al. Extended-spectrum beta-lactamase in the rectal flora of patients undergoing transrectal prostate biopsy: a study of the prevalence in a major metropolitan hospital. BJU Int. 2019;123(Suppl 5):43–46. doi: 10.1111/bju.14745. [DOI] [PubMed] [Google Scholar]
- 27.Gandra S, Trett A, Alvarez-Uria G, Solomkin JS, Laxminarayan R. Is the efficacy of antibiotic prophylaxis for surgical procedures decreasing? Systematic review and meta-analysis of randomized control trials. Infect Control Hosp Epidemiol. 2019;40(2):133–141. doi: 10.1017/ice.2018.295. [DOI] [PubMed] [Google Scholar]
- 28.Pfeffer I, Zemel M, Kariv Y, Mishali H, Adler A, Braun T, et al. Prevalence and risk factors for carriage of extended-spectrum beta-lactamase-producing Enterobacteriaceae among patients prior to bowel surgery. Diagn Microbiol Infect Dis. 2016;85(3):377–380. doi: 10.1016/j.diagmicrobio.2016.04.002. [DOI] [PubMed] [Google Scholar]
- 29.Alevizakos M, Karanika S, Detsis M, Mylonakis E. Colonisation with extended-spectrum beta-lactamase-producing Enterobacteriaceae and risk for infection among patients with solid or haematological malignancy: a systematic review and meta-analysis. Int J Antimicrob Agents. 2016;48(6):647–654. doi: 10.1016/j.ijantimicag.2016.08.021. [DOI] [PubMed] [Google Scholar]
- 30.Grasselli G, Scaravilli V, Alagna L, et al. Gastrointestinal colonization with multidrug-resistant Gram-negative bacteria during extracorporeal membrane oxygenation: effect on the risk of subsequent infections and impact on patient outcome. Ann Intensive Care. 2019;9(1):141. doi: 10.1186/s13613-019-0615-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Aguado JM, Silva JT, Fernandez-Ruiz M, Cordero E, Fortun J, Gudiol C, et al. Management of multidrug resistant Gram-negative bacilli infections in solid organ transplant recipients: SET/GESITRA-SEIMC/REIPI recommendations. Transplant Rev (Orlando) 2018;32(1):36–57. doi: 10.1016/j.trre.2017.07.001. [DOI] [PubMed] [Google Scholar]
- 32.Nutman A, Temkin E, Harbarth S, Carevic B, Ris F, Fankhauser-Rodriguez C, et al. Personalized ertapenem prophylaxis for carriers of extended-spectrum beta-lactamase-producing Enterobacteriaceae undergoing colorectal surgery. Clin Infect Dis. 2020;70(9):1891–7. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analysed during this study are included in this published article and as supplementary information files.