Abstract
Uncertainty permeates decisions from the trivial to the profound. Integrating brain and behavioral evidence, we discuss how probabilistic (varied outcomes) and temporal (delayed outcomes) uncertainty differ across age and individuals; how critical tests adjudicate between theories of uncertainty (prospect theory and fuzzy-trace theory); and how these mechanisms might be represented in the brain. The same categorical gist representations of gains and losses account for choices and eye-tracking data in both value-allocation (add money to gambles) and risky-choice tasks, disconfirming prospect theory and confirming predictions of fuzzy-trace theory. The analysis is extended to delay discounting and disambiguated choices, explaining hidden-zero effects that similarly turn on categorical distinctions between some gain and no gain, certain gain and uncertain gain, gain and loss, and now and later. Bold activation implicates dorsolateral prefrontal and posterior parietal cortices in gist strategies that are not just one tool in a grab-bag of cognitive options but rather are general strategies that systematically predict behaviors across many different tasks involving probabilistic and temporal uncertainty. High valuation (e.g., ventral striatum; ventromedial prefrontal cortex) and low executive control (e.g., lateral prefrontal cortex) contribute to risky and impatient choices, especially in youth. However, valuation in ventral striatum supports reward-maximizing and gist strategies in adulthood. Indeed, processing becomes less “rational” in the sense of maximizing gains and more noncompensatory (eye movements indicate fewer tradeoffs) as development progresses from adolescence to adulthood, as predicted. Implications for theoretically predicted “public-health paradoxes” are discussed, including gist versus verbatim thinking in drug experimentation and addiction.
Keywords: Uncertainty, Temporal discounting, Ambiguity, Risk, Adolescent brain, Decision neuroscience
“Our new constitution is now established, and has an appearance that promises permanency; but in this world nothing can be said to be certain, except death and taxes.” Benjamin Franklin, November 13, 1789
Uncertainty permeates our lives and influences decisions ranging from the trivial (whether to order seafood in a steak restaurant) to the profound (whether voting for one candidate rather than another will increase the likelihood that the U.S. constitution endures). Uncertainty comes in different varieties, and we focus on two of them: uncertainty about outcomes because they are probabilistic (i.e., outcomes fluctuate), and uncertainty about outcomes because they occur in the future. We discuss how tolerance for uncertainty of these two types varies across individuals and across the lifespan; how two types of critical tests adjudicate between major theories of decision making under uncertainty (prospect theory and fuzzy-trace theory); and how different mechanisms underlying decision-making might be represented in the brain.
Implications for risk reduction and addiction are also briefly discussed, including what we call a “public health paradox” (Reyna and Farley, 2006). That is, precise tradeoffs of risk and reward often promote risk-taking (when probability of harm is low and magnitude of reward is high for single acts, a common scenario in public health), but healthy adults typically avoid such tradeoffs. This theoretically predicted public health paradox applies to trying opioids, HIV-AIDS transmission from unprotected sex, driving without a seat belt, and not taking risk-reduction measures during the COVID-19 pandemic, among other behaviors (Table 1). For example, among individuals who experimented with opioids for nonmedical use, only approximately 7% progressed to addiction (Boyd et al., 2009; McCabe et al., 2007). Accordingly, the probability of becoming addicted from trying opioids is relatively low (although greater at earlier onset; McCabe et al., 2007) while the pleasure is high, paradoxically favoring a decision to try opioids from a rational choice (risk-reward tradeoff) perspective. However, healthy adults typically do not engage in this trade-off thinking but apply gist principles which protects them from unlikely but substantial harm. Thus, the “right” choice from a rational choice perspective can be the “wrong” choice from a policy or psychological perspective, as delineated in fuzzy-trace theory, a position supported by the surprising (but predicted) direction of certain developmental differences and by health and medical outcomes, as explained below.
Table 1.
Fuzzy trace theory predicts a common public health paradox: examples of precise risk-reward tradeoffs that promote risk-taking and gist thinking that avoids them
| Verbatim: trading-off favors taking the risk | Gist: not trading-off favors avoiding the risk | ||||
|---|---|---|---|---|---|
| Scenario | Low probability of high harm | High probability of high reward | Trading-off favors… | Categorical gist | Therefore, I will avoid risk and… |
| Opioids | Approximately 0.7% chance of dying from overdose and/or 7% chance of getting addicted1 | Feeling high | Trying opioids | It only takes once to overdose or get addicted from trying opioids. | Not try opioids |
| HIV-AIDS transmission | Approximately 0.08% chance of contracting the virus per encounter2 | Pleasure from unprotected sex | Having unprotected sex | There is no cure for the virus so avoid risk. | Use a condom |
| Driving safety | Approximately 0.0001% chance of being in a fatal crash per 100 miles driven3 | Freedom and convenience of not using a seatbelt | Driving without a seatbelt | Why risk dying in a car accident? | Put on my seatbelt |
| Prevaccine COVID-19 transmission | Approximately 2.4% chance of dying from the virus4 | Having fun being social without masks | Not socially distance and not wear a mask | Before vaccine development: Social distancing and wearing masks are the only protection against a “real possibility” of dying. | Socially distance and wear a mask |
Scientific estimates are included to illustrate the paradox that trading off ironically often promotes unhealthy risk-taking in common public health contexts. Laypeople may not know exact risks but they usually know that bad outcomes are rare and pleasure (reward) is virtually certain for a single decision to take a risk. For these examples, precise (verbatim) thinking involves taking the amount of risk (low) into account and trading it off against the amount of reward (high) in deciding whether to take a risk. Categorical gist thinking, in contrast, emphasizes meaningful qualitative distinctions, such as between curable and incurable, alive and dead (e.g., from a car accident or overdose), and addicted or not. Thus, effective risk reduction involves searching for categorical pivot points in decisions and communicating these “bottom lines.” Because information about precise probabilities and magnitudes of harm/reward is hard to find in real life, fuzzy gist thinking makes it possible to make decisions despite uncertainty
1Chance of dying from overdose is calculated by the number of people who died from a drug overdose in 2019 out of the number of people who misused opioids in 2019 (U.S. Department of Health and Human Services, 2022). See Boyd et al. (2009) and McCabe et al. (2007) for chance of getting addicted
2Male to female transmission in high income countries (Boily et al., 2009)
3Insurance Institute for Highway Safety (2022)
4Calculated as the number of deaths divided by the number of cases as of November 1, 2020 (The COVID Tracking Project, n.d.)
Other behavioral theories take the opposite perspective, that magnitudes of risk and reward should determine risk-reduction behavior (e.g., for a review, see Reyna, Broniatowski, and Edelson, 2021b). Fuzzy-trace theory does not suggest that magnitudes are irrelevant to decision making (see Pirrone et al., 2022), but they are only one input and what matters more is the qualitative bottom-line gist that integrates inputs. In particular, the focus of this article is evidence for mental representations of the simplest gist that governs decision-making in brain and behavior alongside precise inputs: categorical representations.
Background: Varieties of uncertainty
The first type of uncertainty—probabilistic—is tapped when people decide between options that vary in risk, such as choosing between receiving $1,000 with certainty or a 50% chance of $2,000 (otherwise nothing). The second type of uncertainty—temporal—is tapped when people decide between options that vary in time, such as choosing between receiving $1,000 now or $2,000 in a year. A third type of uncertainty undergirds the first two types: ambiguity. Although experimenters often present choices with known probabilities (e.g., fully specified lotteries), probabilities in the real world are typically suffused with uncertainty (Levy and Schiller, 2021). For example, despite an “infodemic” of readily accessible information about COVID-19 (World Health Organization, 2020), individuals do not know the probability that they will die from COVID-19 if they choose to not wear a mask (e.g., Sinclair et al., 2021). Instead, these probabilities are fuzzy, which matters for decision making, and theories of brain and behavior must take this fuzziness into account.
Decisions that involve temporal delays also are uncertain. The fuzzy ambiguity of time is illustrated in maps that forecast the potential paths of a hurricane (with varying success; Padilla et al., 2020). As shown in Fig. 1, the current position of a hurricane is the point of origin, but as simulated time passes, the possible paths of the hurricane diverge. As these maps illustrate, time itself introduces uncertainty, because the future can play out in different ways. Similarly, the promised $2,000 prize mentioned above can fail to materialize in a year, not just because payers might have nefarious intentions (i.e., they are untrustworthy), but because events intervene (e.g., bankruptcy of the payer). Rather than converging on a preordained future, uncertainty grows with distance from the present, particularly when time periods span developmental stages, such as childhood to adulthood, because the self and the self’s preferences change (Bartels and Rips, 2010; Hershfield, 2011; Reyna and Farley, 2006).
Fig. 1.
Tracking forecast for Hurricane Sandy from the U.S. National Oceanic and Atmospheric Administration depicting growing uncertainty over time in the potential path of the hurricane, generated by mathematical models (see also Padilla et al., 2020). Because neither the size nor severity of the hurricane is depicted, this map can only be used to determine whether one is at-risk (and therefore need to evacuate) or not, consistent with categorical gist. This image is in the public domain because it contains materials that originally came from the U.S. National Oceanic and Atmospheric Administration, taken or made as part of an employee's official duties (forecast for October 28, 2012)
Hence, because of inherent ambiguity, a wise decision-maker should embrace vagueness in mental representations of probabilistic or temporal choices, such as those described above. Outcomes might not turn out precisely on the nose of predictions, probabilities fluctuate as conditions change, and changing contexts are not simply sources of bias, but, rather, they allow the meaning of information about probabilities and outcomes to be interpreted and revised (see discussion of the contextual relativity of gist in Reyna, Brainerd et al., 2021a). Moreover, in our approach, useful mental representations are not vague simply to cut mental corners, but because they distill what matters, which often is the qualitative even categorical (i.e., the simplest) gist of decision options. As fuzzy-trace theory outlines descriptively and prescriptively, adaptive decision making emphasizes getting the big picture of decision options (integrating facts and details into a meaningful bottom-line whole) and applying core values to that mental representation of the big picture: those values that are simple, fundamental, and enduring.
Preferences for risk and time
Reasons beyond ambiguity also support advantages of using fuzzy “gist” representations in decision making, including that memory for gist endures longer than verbatim memory for details, an important consideration evolutionarily before the invention of writing (Reyna and Mills, 2007). Gist processing is generally easier under stress, high arousal, or disruptive interference compared with verbatim processing (Rivers et al., 2008; Venkatraman et al., 2011). Most important, gist representations capture essential meaning rather than arbitrary details (the latter are captured in verbatim representations), scaffolding decision-making that consequently reflects an appreciation of the meaning of facts. Preferences involving time and probability derive from mental representations of meaning grounded in different backgrounds and experiences (Croote et al., 2020; Edelson and Reyna, 2021). This approach motivates such questions as what does “waiting” or “a long time” or “rewards” mean in a given cultural context, and how do different scenarios translate into these meanings?
Despite the fluidity of meaning in context, once decision scenarios are translated, core values can be remarkably similar across people. Thinking about probabilistic and temporal decisions in their starkest “gisty” terms, all else equal, most people prefer receiving certain rather than uncertain positive outcomes. They prefer rewards now and for sure, not later and maybe. Psychologically, these are categorical distinctions. However, these preferences are traditionally assessed by using tasks in which continuous trading off of the magnitude of outcomes against either time or probability is assumed theoretically (Glimcher, 2022). For example, decision-makers are described as discounting later, larger rewards relative to sooner, smaller rewards, yielding a discount rate. This is traditionally a smooth function over outcome magnitudes and time intervals. Correspondingly, risk aversion is characterized by using continuous nonlinear functions (e.g., negatively accelerated subjective valuation of objective outcome magnitudes) in traditional expectancy value theories, such as expected utility or prospect theory (Kahneman and Tversky, 1979; Trepel et al., 2005). These functions can be flatter due to emotion or other factors (Rottenstreich and Hsee, 2001; but see Levine, 2019), but decision dimensions still trade off and discounting or risk aversion are generally treated as a matter of degree (quantitative differences) not of kind (qualitative differences).
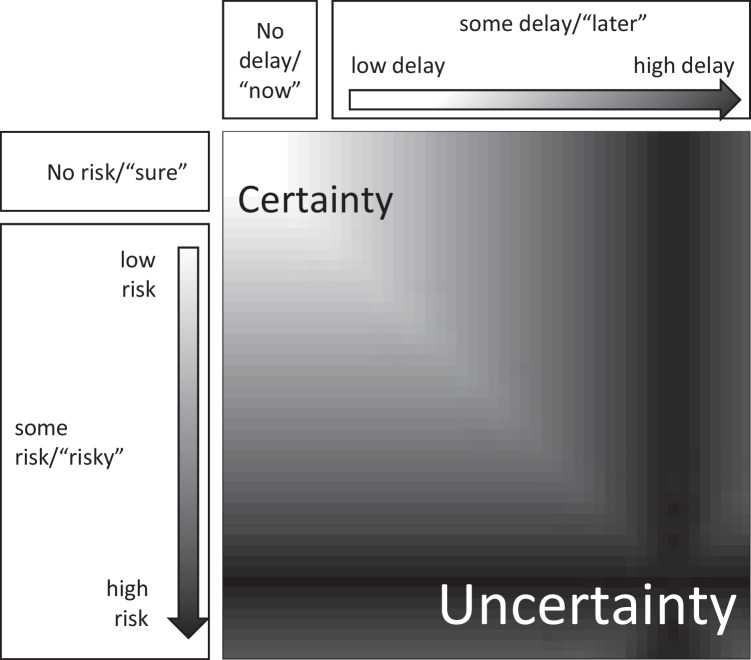
However, in both probabilistic and temporal decisions, there are psychological discontinuities at zero risk and zero delay, referred to as the certainty effect and present bias (immediacy effect), respectively (as well as at other categorical boundaries; Kahneman and Tversky, 1979; Laibson and Maxted, 2022). Receiving a reward now is psychologically distinct from receiving it very, very soon, although the literal difference between now and very, very soon (what we call “verbatim” differences) could be miniscule. An “instantaneous-gratification agent” is inside all of us. Analogously, “soon” can occur over a fuzzy horizon of minutes to days, but it is psychologically distinct from “later” (e.g., McClure et al., 2007; Augenblick and Rabin, 2019). Figure 2 illustrates these categorical discontinuities between no risk and some risk and between no delay and some delay, as well as continuous changes in probabilistic and temporal dimensions.
Fig. 2.
Uncertainty resulting from (increasing) delay and (growing) variability of outcomes or risk
According to fuzzy-trace theory, people encode these categorical distinctions (categorical gist), more fine-grained ordinal distinctions, and continuous or precise distinctions in parallel (cf. DeKay, Rubinchik et al., 2022), but they rely on the simplest distinctions that allow them to accomplish a task (e.g., the task of making a choice between two options or of providing a monetary value that they are willing to pay for an option; Reyna, 2012). Naturally, making a choice often requires lower resolution in mental representations than providing an exact monetary value, a principle called task calibration in fuzzy-trace theory that explains classic preference reversals (Corbin et al., 2015; Fischer and Hawkins, 1993).
Fuzzy-trace theory does not make ad hoc assumptions about differences in attention or weighting, although it is compatible with such claims. Instead, it explains why those phenomena are observed by using underlying psychological and neural mechanisms that have been independently verified. For instance, the theory explains that distinguishing between no risk and some risk (or low risk and high risk) is not arbitrary but must reflect the qualitative meaning of this distinction embedded in understanding what differences matter, regardless of their objective size. A seemingly tiny 1% prevalence of COVID-19 in the early stages of the pandemic was “high” (Reyna, Broniatowski et al. 2021b). The theory explains known effects that other theories describe, such as certainty effects and present bias effect, but as we discuss in the next section, it predicts effects that disconfirm those theories, too. In this connection, we focus on two tasks that provide critical tests of prospect theory and fuzzy-trace theory.
Critical tests of theories: Truncation and value allocation
Truncation in framing tasks
In 1991, Reyna and Brainerd introduced a manipulation with three variations, all presenting choices, such as those described above with a sure option versus a gamble, as tests of prospect theory and fuzzy-trace theory (for reviews of independent replications of predicted effects, see Reyna, 2012, and Kühberger and Tanner, 2010). One variation is just the standard version of these choices with fully specified gambles (50% chance of $2,000 and 50% chance of $0). The prospect theory explanation of preference for the sure option when options are phrased as gains—switching to preference for the gamble when options are phrased as losses—turns on posited nonlinear perceptions of outcomes and probabilities: For example, a 50% chance of a discounted $2,000 is less than a solid $1,000 for sure, favoring the sure option for gains (greater gains are better) and the gamble for losses (lower losses are better) (Tversky and Kahneman, 1986; Wakker, 2010). Fuzzy-trace theory, in contrast, predicts that multiple literal (verbatim) and gist representations are extracted and the simplest categorical gist determines choices because of (a) a gist-processing preference observed and predicted in many cognitive tasks, and (b) both ordinal gist (less money with higher chance ≈ more money with lower chance) and literal expected value are indeterminate (expected value: $1,000 x 1.0 = 0.5 x $2,000 + 0.5 x $0).
Therefore, in another variation designed to emphasize the categorical gist representation, the gamble was truncated by deleting the part concerning a 50% chance of $2,000 from the body of the problem. That deleted information was given to decision-makers as background information that preceded the options. Thus, for gains, the decision boiled down to a categorical contrast between gaining something for sure versus possibly gaining nothing, which accentuated preference for the sure option for gains. For losses, the same manipulation of deleting the nonzero part of the gamble accentuated preference for the gamble. Thus, this manipulation induced a categorical gist perspective on the decision that produced classic gain-loss framing effects, indeed, exaggerated them, consistent with the explanation that categorical gist causes framing effects. (Similar effects were obtained by substituting the qualitative categories “some” and “none” for all numerical outcomes in decision problems, another test of the categorical gist hypothesis.)
Conversely, in yet another variation designed to emphasize verbatim representations, deleting the part of the gamble concerning a 50% chance of gaining or losing nothing eliminated framing effects, because there was no longer a categorical contrast between options. Note that prospect theory predicts identical framing effects when the zero part is deleted, because that part multiplies out to be zero in the theory; all of the action in the theory involves nonlinear perceptions of the nonzero part of the gamble (Kühberger and Tanner, 2010). In sum, prospect theory’s predictions have been disconfirmed and fuzzy-trace theory’s predictions have been confirmed using deletion (aka truncation) manipulations, the latter through assuming simple representations of something versus nothing and preferences that respect simple differences in affective valence (something good is better than nothing and something bad is worse than nothing).
Other simplifying approaches have been offered for these tasks, but their motivation is often empirical rather than mechanism-driven; they make predictions that have been repeatedly falsified; and the claims are different from those of fuzzy-trace theory (e.g., simple-minded mental shortcuts due to working capacity limitations). Specifically, we do not discuss the priority heuristic (Brandstätter et al., 2006) and other accounts that involve simplifying representations, because data from the tasks that we review disconfirm those explanations (Venkatraman et al., 2014). However, it is possible that these accounts could be integrated using fuzzy-trace theory’s parallel cognitive architecture. For example, coding efficiency in the brain (Glimcher, 2022) is a simplification process that is likely to influence ordinal gist representations because they are inherently comparative (i.e., less-more; Broniatowski and Reyna, 2018). In this connection, we note that fuzzy-trace theory reconciles prior contradictory findings (e.g., Glöckner and Herbold, 2011; Johnson et al., 2008) in that it predicts that people engage in noncompensatory and compensatory automatic processes roughly simultaneously but rely more on the former.
Notably, “simplification” strategies differ in fuzzy-trace theory (they are meaningful and a cognitively advanced type of processing) compared with other simplification approaches (e.g., impulsive or lazy processing, Pennycook and Rand, 2019, which are separate parameters in fuzzy-trace theory); also see Gaissmaier and Schooler (2008); Raoelison, Thompson, and De Neys (2020). Strategy selection, or more precisely strategy emphasis, is driven by predicted individual and developmental differences (Broniatowski and Reyna, 2018; Reyna and Brainerd, 2011; Reyna and Brust-Renck, 2020). The stimulus is the source of both verbatim and gist representations and they both influence responses, but they operate in parallel as demonstrated in double-dissociation effects and other strong model tests that go beyond automatic-controlled, reason-emotion, or bad-good reasoning dichotomies (e.g., Keren and Schul, 2009; Li et al., 2017; Melnikoff and Bargh, 2018). Thus, fuzzy-trace theory and Sloman’s (1996) dual-processes model are precursors of current parallel processing approaches to cognitive biases, such as framing effects (De Neys, 2018; Pennycook, in press; Trippas, Thompson, and Handley, 2017).
Ambiguity in framing tasks
Some theorists have explained framing (Kühberger, 1995) and truncation effects (Mandel, 2014) as due to ambiguity about outcomes. Consider the classic dread-disease problem in which people choose between saving 200 people (of 600 expected to die) versus a one-third chance that 600 people will be saved; otherwise none will be saved (losses are phrased as the number who die). The gain frame that mentions 200 (of the 600 people) saved might raise the question—what happened to the other 400 people? The sure option could be ambiguous. To account for framing effects, the ambiguity has to be resolved optimistically; at least 200 people will be saved and possibly more, so the sure option is preferred. For the loss frame, the ambiguity has to be resolved pessimistically to account for effects; at least 400 die and maybe more, so the risky option is preferred. Thus, the ambiguity hypothesis requires two different assumptions to account for two data points, which is not parsimonious. Contrary to some arguments, these ambiguous readings of framing problems are not predicted by conversational implicatures but, rather, violate them; for example, if more than 200 people could be saved, the normal pragmatic assumption is that the speaker would have said so. The ambiguity hypothesis also requires that people ignore wording and instructions, such as “exact estimate” and “exactly” 200 people are saved.
However, Mandel (2014) and Kühberger and Tanner (2010) among others have generated evidence suggesting that ambiguity accounts for variance in framing effects. Mandel found, for example, that introducing the word “exactly” eliminated framing effects and adding “at least” to the sure option produced framing effects. But the effects of adding the word “exactly” have not replicated consistently (Chick et al., 2016; Simmons and Nelson, 2013). For example, framing effects are routinely observed when the words “exact” or “exactly” are used in problem information (for meta-analyses, see Broniatowski and Reyna, 2018, and Steiger and Kühberger, 2018). Moreover, experiments that provide instructions to avoid these hypothesized interpretations and explicit tests of how decision makers then interpret framing problems have shown that most pass ambiguity tests (i.e., they do not make the posited inferences about missing numbers, not conforming to the ambiguity hypothesis in any of several versions) and yet show framing effects (Chick et al., 2016; Reyna et al., 2021a).
One broad rationale for the ambiguity hypothesis is that exact numbers are unrealistic, an argument we also made earlier in this article (Geurts, 2013; Kühberger, 1995; Mandel, 2001; Teigen, 2011). However, this does not imply that numbers are interpreted as meaning “at least” as opposed to “at most” or other asymmetric interpretations. Mandel (2014) asserts that “‘at least n’ and ‘exactly n’ should both have the same gist (viz., some),” but this is not true. Fuzzy means that saving 200 people is construed as “about” 200 and that, in one type of representation (the least precise one and thus the favored representation), this amounts to the qualitative category of saving “some people” when it contrasts with the possibility of saving none (Reyna et al., 2021a). “At least” (e.g., 200 or more) or “at most” (e.g., 200 or less) each change the balance of expected values (the literal representations), but they also change meaning in context. This meaning has been distinguished from some versus none in research on numerical quantities in fuzzy-trace theory (Brainerd and Gordon, 1994; Reyna and Brust-Renck, 2020; Thompson and Siegler, 2010).
Part of the difficulty could be misinterpreting the explanation that framing effects depend crucially on categorical gist to mean that only categorical gist is processed. At least three levels of representation are processed—categorical, ordinal, and interval for numbers and inferences, paraphrases, and literal words for sentences—and they each “vote” in determining responses. This assumption explains why responses change predictably when the wording of options changes (e.g., as in the Allais paradox), namely, because different representations discriminate options differently (see Table 1 in Broniatowski and Reyna, 2018, and Figs. 1 and 2 in Reyna, 2012). When categorical representations do not discriminate options, decision makers “revert to” more precise representations in the sense that they then become determinative (Mar and Liu, 2022; Reyna, 2012).
Thus, it is not the case that either verbatim (e.g., numerical) or gist (e.g., nonnumerical) processing occurs. Kühberger and Luger-Bazinger (2016) point out that “In framed gambles, all relevant information is presented—outcomes and their respective probabilities—leaving no ambiguities” and that “everybody knows that exact numerical outcomes are the essence of gambl[ing for money]” (p. 948). They concluded that this implies that gist extraction is unlikely for gambles. However, gist extraction occurs routinely in numerical tasks, as demonstrated in many experiments on the independence of verbatim and gist representations (for a review, Reyna, 2012).
However, there is another important manipulation that has been interpreted as resolving ambiguity that changes framing effects, which concerns us. That “disambiguation” manipulation involves filling in the complementary amount in the sure option (Kühberger, 1995; Kühberger and Tanner, 2010; Mandel, 2001). “Saving 200 people” becomes “saving 200 people and not saving 400 people”; “400 dying” becomes “400 dying and 200 not dying.” As other research suggests, this information is often not unknown (the definition of ambiguity); most people pass tests indicating that they realize that 200 saved implies that 400 are not saved. The “implied” information also is sometimes explicitly given in instructions; thus, it is not uncertain (Chick et al., 2016). Framing becomes attenuated, because this manipulation changes the gist (see Table 1 in Broniatowski and Reyna, 2018).
That is, instead of saving some people (good news), the “sure” option becomes a wishy-washy “good news” and “bad news” gist: saving some people and not saving some people. This option now stacks up as pretty much equivalent to the gamble, which also contains good news and bad news. Naturally, these assumptions apply analogously to decision problems with losses. The roots of fuzzy-trace theory go back to classic findings in psycholinguistics demonstrating similar effects of verbal descriptions (Clark and Clark, 1977). The effects of descriptions also demonstrate the value of distinguishing between literal facts, which do not change across the various transformations of problems that we have discussed (extensional or informational equivalence), and gist representations of those facts that involve interpretation (cf. Fisher, 2022; that construal of semantic features shifts falls out of this approach; Fisher, 2021). Memory research shows that people extract independent verbatim and gist representations of the same stimulus, the ultimate example of informational equivalence (Reyna, 2012).
The value allocation task
Pwin = Pmax = gist
With framing effects accounted for, and several variations that manipulate the expression of verbatim and gist representations in responses, we now turn to another task that was invented independently (Payne, 2005) and yet elicits the same categorical gist processing. To preview, although gist and affective valence have been mentioned in publications on this task, this article is the first in which these publications are brought together and shown to be critical tests not only of prospect theory (as previously argued) but of fuzzy-trace theory. Indeed, fuzzy-trace theory predicts value-allocation results for choices, ratings, and eye movements and developmental differences between adults and adolescents. We then integrate behavioral results into a preliminary neural model and explore its implications for addiction and other kinds of unhealthy risk-taking.
In the value-allocation task, decision-makers are presented with a series of multiple-outcome gambles (e.g., Venkatraman et al., 2014). Each gamble consists of a large gain outcome, a large loss outcome, and an intermediate outcome that is one of the following: a small gain, $0, or a small loss. In some variations, the probabilities of the outcomes are equal (e.g., +$45, 0.33; $0, 0.33; −$65, 0.33) and in other variations they are unequal (e.g., +$65, 0.38; −$15, 0.26; −$80, 0.38). In the latter, adding money to the intermediate outcome has lower expected value than adding money to one of the extreme outcomes (because the intermediate outcome has lower probability). Before value allocation (e.g., before adding money), participants rate the desirability of the gambles (from “least preferred” to “most preferred”), which ensures that they are able to process differences in expected value.
Value allocation occurs in a subsequent phase in which participants choose how to allocate different fixed amounts (e.g., $15) to one of the outcomes. Allocating $15 to the intermediate outcome above (to the $0 outcome in +$45, 0.33; $0, 0.33; −$65, 0.33) increases the probability of winning some money as opposed to no money, which differ categorically, but it does not maximize the amount of gains (Gmax: allocating the $15 to the +$45 option) or minimize the amount of losses (Lmin: allocating the $15 to the −$65 option). The former strategy of winning some money rather than none is noncompensatory and is called Pwin or Pmax, whereas the other two strategies are compensatory, as assumed in prospect and other utility theories. Consistent with fuzzy-trace theory, adults across different experiments preferred the Pwin strategy (e.g., 69% in Venkatraman et al., 2014). The greatest preference for the Pwin strategy occurred when allocations created categorical changes in outcomes from no gain to gain, from loss to no loss, and, most dramatically, from loss to gain. As Venkatraman et al. (2014) explain, “The Pwin heuristic represents a computational simplification for complex gambles that ignores payoff (value) magnitude information and focuses on the ‘gist’ (gain versus loss) of an outcome value” (p. 75), but the connection to theory that predicts this is not made.
As further predicted by fuzzy-trace theory (and described above for framing problems), when the Pwin strategy was not available—when adding money to the intermediate outcome did not increase the chance of winning “something”—choices of that outcome for allocation declined (e.g., from 69% to 41%) and Lmin increased (e.g., from 23% to 44%) (Venkatraman et al., 2014). That is, strategy selection reverted to more precise processing, as exemplified in the compensatory Lmin strategy in which the amount of losses mattered. As in framing problems, when expected values were unequal and thus verbatim representations competed with the Pwin gist strategy, choices of the intermediate outcome declined (e.g., to 52%) but remained substantially higher (favoring gist processing) than the next preferred Lmin strategy (e.g., 36%). (See Table 2 for detailed examples and predictions.) In addition, there were individual differences in gist and verbatim processing that echo those in fuzzy-trace theory studies of decision-making (Reyna and Brainerd, 2011; Reyna and Brust-Renck, 2020; Reyna et al., 2018).
Table 2.
Examples of problems in the value allocation task when expected values of allocation choices are equal
| I: Core problem pitting fuzzy trace theory’s prediction (Pwin) against cumulative prospect theory’s prediction (Lmin) | |||||
| Outcome | Probability | Choose one of two allocations | Strategy | Categorical gist representation | Categorical gist principle |
| $75 | 0.20 | ||||
| $35 | 0.20 | ||||
| $0 | 0.20 | +$15 = $15 | Pwin | No gain to some gain. | Some gain is better than no gain. |
| −$25 | 0.20 | ||||
| −$75 | 0.20 | +$15 = −$60 | Lmin | Some loss to some loss. | No categorical improvement: not preferred. |
| II: Core problem pitting fuzzy trace theory’s prediction (Pwin) against expected utility theory’s prediction (Gmax) | |||||
| Outcome | Probability | Choose one of two allocations | Strategy |
Categorical gist representation |
Categorical gist principle |
| $75 | 0.20 | +$15 = $90 | Gmax | Some gain to some gain. | No categorical improvement: not preferred. |
| $35 | 0.20 | ||||
| $0 | 0.20 | +$15 = $15 | Pwin | No gain to some gain. | Some gain is better than no gain. |
| −$25 | 0.20 | ||||
| −$75 | 0.20 | ||||
| III: Pwin unavailable problem indicating fuzzy trace theory’s predictions (Lmin and problem I differs from III) and cumulative prospect theory’s predictions (Lmin and problem I is virtually identical to III) | |||||
| Outcome | Probability | Choose one of two allocations | Strategy |
Categorical gist representation |
Categorical gist principle |
| $75 | 0.20 | ||||
| $35 | 0.20 | ||||
| $5 | 0.20 | +$15 = $20 | Reference outcome (former Pwin) | Some gain to some gain. | No categorical improvement: not preferred. |
| −$25 | 0.20 | ||||
| −$75 | 0.20 | +$15 = −$60 |
Lmin; Problem I differed from Problem III |
Some loss to some loss: Revert to more precise representations; see Allais Problem 2 in Reyna & Brainerd, 2011; Reyna & Brust-Renck, 2020. | Losses hurt more than gains feel good (CPT). |
| IV: Pwin exaggerated problem indicating fuzzy trace theory’s prediction (exaggerated Pwin) and cumulative prospect theory’s prediction (Lmin) | |||||
| Outcome (only total shown) | Probability | Choose one of two allocations | Strategy | Categorical gist representation | Categorical gist principle |
| $75 + $70 = $145 | 0.20 | ||||
| $35 + $70 = $105 | 0.20 | ||||
| $0 + $70 = $70 | 0.20 | +$15 = $85 | Reference outcome (former Pwin) | Some gain to some gain. | No categorical improvement: not preferred. |
| −$25 + $70 = $50 | 0.20 | ||||
| −$75 + $70 = −$5 | 0.20 | +$15 = $10 |
FTT: Pwin+ CPT: Lmin |
Some loss to some gain: Gamble becomes certain gain (possible loss eliminated). |
Some gain is better than some loss. Categorical improvement: preferred. |
Modal observed preference indicated by bolding (see text). According to fuzzy-trace theory (FTT), uncertainty at the categorical gist level of representation means that an option presents more than one qualitatively different outcome: no gain vs. gain, no loss vs. loss, and gain vs. loss. Contrary to Venkatraman et al. (2009, p. 9), the categorical gist explanation here does “apply to risky choice problems that involve only ‘pure’ gain or loss options,” introduced in Reyna and Brainerd (1991). Pwin unavailable problems also rule out expected value and expected utility theory because a small amount added or subtracted from the intermediate outcome does not appreciably change overall value or utility, and yet large differences in preference are observed (per FTT). Additionally, Cumulative Prospect Theory (CPT) predicts that preferences should not change as a function of whether adding money to the intermediate outcome changes its valence (e.g., improving from −10 to $10 or improving from −$30 to −$10) due to the rank-dependent transformations applied to the extreme outcomes (as long as intermediate outcomes are not higher in probability; Venkatraman et al., 2014, p. 75). However, when the intermediate probability is lowered (not shown), and thus the expected value of the allocation is lower compared with the other option (Gmax or Lmin), Pwin should be avoided according to expected value, expected utility, and CPT, whereas FTT predicts that Pwin remains most preferred though at a smaller margin because of the competing expected value (verbatim) representation (see Allais Problem 1 in Broniatowski & Reyna, 2018; Reyna & Brust-Renck, 2020). Eye tracking and developmental differences further differentiate FTT’s and CPT’s predictions, supporting FTT and ruling out CPT (e.g., Kwak et al., 2015; Payne, 2005; Venkatraman et al., 2014)
The results we have described were comparable to those of a second and third experiment reported by Venkatraman et al. (2014), as well as those of Payne (2005), Venkatraman et al. (2009, 2011), and Kwak et al. (2015), and are reflected in multiple outcome measures used to diagnose strategies, including eye movements. The “huge bias” (p. 85, Venkatraman et al., 2014) toward Pwin choices in Pwin-available trials that was not present in the Pwin-unavailable trials cannot be accounted for by prospect theory, even by tinkering with parameter values to capture individual differences. Evidence from eye tracking also ruled out prospect theory, regardless of assumed parameter values, as well as other attribute-based, noncompensatory, or similar simplification approaches (e.g., the priority heuristic). Together, these results provide evidence of a gist-processing preference that is central to fuzzy-trace theory but which is otherwise a mysterious empirically observed violation of extant decision theories.
Developmental reversals: Compensatory and noncompensatory decision strategies
The prior section summarized results indicating that most adults prefer a gist processing strategy in the value allocation task, extending fuzzy-trace theory to three-outcome and five-outcome gambles. Not only do adults apparently rely on gist in these tasks but they rely on the same types of categorical gist representations and principles as implicated in critical tests of framing effects and the Allais paradox (some gain is better than no gain, no loss is better than some loss, and some gain is better than some loss; Broniatowski and Reyna, 2018; Reyna, 2012). Kwak et al. (2015) administered the valuation allocation task discussed above to a group of adolescents and a group of young adults. Developmental theories, ranging from the neoPiagetian tradition to contemporary information processing and dual-process approaches, generally predict that the ability to engage in compensatory processing (e.g., computing expected value in which probability and outcomes trade off) increases from childhood to adulthood (Reyna and Farley, 2006). Therefore, most theories suggest that adults would be more (or perhaps equally) likely to engage in compensatory processing (e.g., Lmin) than adolescents but certainly not less likely. Kwak et al. (2015) found the opposite.
Adolescents were significantly more likely to choose in accordance with the compensatory Lmin strategy than adults. Although the Pwin categorical gist strategy was the most popular for both age groups, it was more popular among adults than adolescents. Contrary to most developmental theories, adolescents with higher cognitive capacity were more likely to choose in accordance with Pwin (see Kogut and Slovic, 2016, for a similar result). Moreover, analyses of eye movements confirmed and expanded this account. Consistent with predictions and prior findings of fuzzy-trace theory (e.g., comparing children to early adolescents, Reyna and Ellis, 1994, and adolescents to adults, Reyna et al., 2011), Kwak et al. (2015) found that adolescents processed more information and processed it more deliberatively (e.g., greater number, more evenly distributed, and longer fixations). The patterns of fixations for adolescents—but not adults—were compatible with prospect theory and other utility theories (i.e., adolescents had a higher “Payne index” indicative of expected value processing). This developmental finding of objectively superior performance in younger (children and adolescents) compared with older samples (young and middle-aged adults) has been dubbed “developmental reversal” in fuzzy-trace theory, because it reverses typical developmental expectations, reflecting the growth in gist processing with development (Reyna et al., 2014). Thus, adolescents are likely sensitive to rewards, but their risk-taking in real life is not attributable to overvaluing rewards by itself (Edelson and Reyna, 2021) but rather the all-too-rational combination of risk (which is in fact low in many common circumstances) and reward (which is in fact high in those circumstances). Indeed, as we discuss below, brain and behavioral results buttress the hypothesis that adolescents are more sensitive to these risk-reward tradeoffs in decision-making than adults are.
Mechanisms of decision-making in the brain
We begin this section by briefly reviewing the neural substrates of the distinct types of processing that we have just discussed in adults and adolescents. We then integrate these findings with those concerning decision-making generally into a preliminary neural framework and develop its implications for addiction and other problem behaviors. To be sure, our conclusions oversimplify a more complex picture. Although it has been widely acknowledged that differential activation takes a snapshot of a dynamic system and this neural characterization is incomplete, an often-overlooked shortcoming of neuroscientific data is the inability to connect specific strategies to specific patterns of activation because of task confounds (see Edelson and Reyna, 2021). Shortcomings go beyond adding process measures, such as eye tracking or EEG, although they help. Too much inference from brain to behavior is a problem but so is not enough inference. We attempted to rectify that imbalance with a provisional framework, because understanding the behavioral functions of the brain is indispensable for basic science and applied goals, such as addressing addiction. We dwell on process assumptions, because they are the key to making scientific progress in this area.
Strategic assumptions
In traditional approaches to decision neuroscience, the focus is generally on tradeoffs because the expected utility model—that defines preferences in terms of tradeoffs—is assumed: For probabilistic uncertainty, a safer lower-value option is typically pitted against a riskier higher-value option. For temporal uncertainty, a sooner lower-value option is typically pitted against a later higher-value option. Activation in brain regions is associated with choices in probabilistic paradigms. As examples, increased activation in the anterior insula follows risk-averse choices (Paulus et al., 2003; Preuschoff et al., 2008) and increased activation in the ventromedial prefrontal cortex (VMPFC) and striatum predicts risk-seeking choices (Kuhnen and Knutson, 2005; Reyna and Huettel, 2014; Tobler et al., 2007). Risk-seeking choices necessarily involve higher reward values in tradeoff designs, which implicates reward processing. VMPFC and ventral striatum (VS) encode positive values, such as reward magnitude, with increasing activation, and VMPFC, orbitofrontal cortex (OFC) and posterior parietal cortex (PPC) encode negative values, more aversive values, with decreasing activation. Lateral prefrontal and parietal regions support executive control processes associated with risky decisions, risk evaluation, risk propensity, and judgments about probability and value (Barraclough et al., 2004; Glimcher, 2022; Huettel et al., 2005; Paulus et al., 2001; Sanfey et al., 2003). Individual differences in risk preferences vary with resting-state EEG during wakefulness (Gianotti et al., 2009; Studer et al., 2013) and with slow-wave activation during sleep, both over the right prefrontal cortex (Studler et al., 2022).
Using functional magnetic resonance imaging (fMRI), Venkatraman et al. (2009) studied an incentive-compatible version of the value allocation task, which as we noted above, identifies three types of decision strategies: maximizing the magnitude of gains (Gmax), minimizing the magnitude of losses (Lmin), and changing losses or nothing to wins (Pwin). Gmax and Lmin choices reflect compensatory choices, as they increase the magnitude of the largest possible gain or decrease the magnitude of the largest possible loss, respectively, increasing overall subjective value. In cumulative prospect theory, a further wrinkle is added in that extreme outcomes (those that rank best and worst) are given greater weight and extreme losses are especially aversive, favoring the Lmin strategy in this task. (Rank dependence matters when probabilities of each potential outcome are equal.) Rank dependence is consistent with neural activation data. High activation for both high rewards and high punishments is thought to be encoded in “salience” representations. The salience network includes the ventral striatum, dorsal anterior cingulate cortex, anterior insula, temporoparietal junction, and the amygdala (Kahnt et al., 2014; Levy and Schiller, 2021). However, crucially, cumulative prospect theory does not accommodate the Pwin strategy.
In contrast to compensatory tradeoffs, as discussed, Pwin choices follow a gist-based simplifying strategy to increase the chances of a gain compared to a loss irrespective of reward magnitude. In addition, some problems—Pwin-exaggerated—allow for conversion of uncertain wins to certain wins (and certain losses to uncertain losses). Following the same gist-based rationale per our earlier discussion of uncertainty, a certain win is categorically better than an uncertain win and an uncertain loss is categorically better than a certain loss. Ergo, most adults are predicted to forgo maximizing utility in order to achieve these gist-based categorical conversions (as observed). Expected utility (or expected value) is computed in parallel and, especially for individuals high in numeracy and in literal processing, can offset gist strategies as options become more disparate (but the size of numerical differences is generally not the ultimate arbiter; Broniatowski and Reyna, 2018; Reyna and Brainerd, 2011; Reyna and Brust-Renck, 2020).
Ad hoc (auxiliary) assumptions about regret are not required to explain the prevalence of gist-based phenomena, such as the Pwin strategy (memory, reasoning, and psycholinguistic research support it). Assumptions about aspiration levels are not required, but hypotheses about aspiration level might be profitably combined with gist representations to produce novel predictions (e.g., Coricelli et al., 2018). Contrary to Payne (2005), Venkatraman et al. (2009), and others, the Pwin strategy is an important part of “a general-purpose model of risky choice that is consistently applied across all conditions and contexts” (p. 75). The order of emphasis of strategies—emphasizing the least precise (categorical) distinctions and relying on more precise distinctions if needed (e.g., in Pwin-unavailable trials)—is the opposite of other cognitive theories (i.e., they assume precise processing as a default, which breaks down under cognitive load). This fuzzy-processing principle explains the otherwise theoretically unmotivated Pwin strategy: decision-makers focus on categorical gist and “if no option improves the overall probability of winning or not losing, individuals should shift to other [more precise] strategies to decide between the alternatives.” (Venkatraman et al., 2014, p. 75).
Supporting this process analysis, Venkatraman et al. (2009) found greater activation for compensatory choices (Gmax and Lmin) in the anterior insula (greater for Lmin) and VMPFC (greater for Gmax), areas associated with affective evaluation of decision outcomes. In contrast, Pwin choices resulted in increased activation in the dorsolateral prefrontal cortex (DLPFC) and PPC, which were significantly greater than those for Gmax and Lmin. These regions (DLPFC and PPC) are typically associated with executive functions and decision making. Interestingly, the behavioral data showed that Pwin choices were associated with faster response times (Venkatraman et al., 2009), which contradicts classical assumptions of effortful prefrontal control. Also arguing against a cognitive effort explanation, sleep deprivation did not increase Pwin strategies but rather increased Gmax, consistent with other studies showing increased risk taking (and reward chasing) after sleep deprivation (Venkatraman et al., 2011). Interestingly, consistent with a less-sleep-less-gist hypothesis, other research on gist representations in memory show that gist processes are reduced after sleep deprivation (i.e., sleep increased false recall of meaning-consistent words; Payne et al., 2009; Pardilla-Delgado and Payne, 2017).
Using functional connectivity analyses, Venkatraman et al. (2009) also showed that, when individuals made Pwin choices, connectivity with DMPFC increased in DLPFC and PPC, whereas when individuals made compensatory choices, connectivity with DMPFC increased in the anterioir insular (and amygdala), although not in VMPFC. Moreover, the relative strength of the connectivity between DMPFC and these regions was significantly associated with individual differences in strategy preferences across individuals. Overall, activation in the DMPFC did not predict either type of choice, but it predicted decisions that were inconsistent with the preferred decision strategy, typically shifts from a noncompensatory Pwin strategy to a compensatory Lmin strategy.
Venkatraman et al. (2009) also examined whether a neural substrate of reward processing could predict strategic preferences. At the end of the scanning session, each participant viewed some of their “improved” gambles, which were resolved to actual monetary gains or losses. While participants were anticipating the outcome of each gamble, there was increased activation in the ventral striatum (VS)—a brain region commonly implicated in expectation and processing of positive and negative rewards (e.g., Speer et al., 2021). Then, when the outcome was experienced, VS activation increased to gains but decreased to losses. Paradoxically from traditional perspectives, those individuals who showed the greatest VS increases to gains and decreases to losses preferred the simplifying Pwin strategy. Their brains indicated that they were more sensitive to magnitudes of rewards, compared with others, and yet they eschewed choices that maximized rewards. These results are consistent with fuzzy-trace theory’s tenet that the use of gist-based simplifying strategies is accompanied by application of values (social, moral, and monetary) that reflect affective valence (Broniatowski and Reyna, 2018; Reyna, 2021). That is, the gist representation of options—gaining some money versus either gaining some money or gaining nothing—requires applying simple values, such as “gaining money is good” to elicit a preference, apparently a response supported by the VS.
Developmental differences in neural substrates
As discussed earlier, developmental differences also offer an opportunity to examine strategic variations in mental representations—verbatim and gist—as well as changes in reward processing over time. Table 3 presents illustrative findings concerning adolescent decision and reward processing, and comparisons to children and adults in some studies. The studies vary in methodology and analyses, thus making conclusions preliminary (see also Defoe and Romer, 2022, summarizing a recent special issue on developmental differences in risky decision making). Many studies echo the results that we have reviewed thus far with respect to the brain, including sensitivity to risk (e.g., activation in DMPFC and insula), sensitivity to overall subjective value (e.g., activation in VMPFC and OFC), and heightened sensitivity among adolescents to reward (e.g., activation in VS or a subarea of VS, the nucleus accumbens, or caudate, but see Bjork and Paldini, 2015). DLPFC seems again to reflect executive function, including learning, and subjective value (although inconsistently).
Table 3.
Illustrative developmental neuroscience findings about decision and reward processing under uncertainty
| Study | Sample | Task | Main findings |
|---|---|---|---|
| van Duijvenvoorde et al. (2015) | n = 72 after exclusions, ages 8-35 yr |
fMRI version of Columbia Card Task: in each round, 6 to 16 cards shown face-down. Participants explicitly told total number of cards, number of loss cards (from 1-8), gain amount for each gain card (from 1-30), and loss amount for turning over loss card (from −5 to −100). Running total of points shown. Each round participant makes a series of binary decisions to turn over another card or not. Game ends when participant decides to stop turning over cards or turns over a loss card. Probability and amounts vary per round. Task performed in and out of scanner. Return (EV) = gain probability*gain amount + loss probability*loss amount Risk (SD) = sqrt(gain probability*[gain amount – EV] 2 + loss probability*[loss amount - EV]2) |
Included risk and return as parametric regressors and gain and loss outcomes and discrete regressors. Age-related changes in neural activation with parametric gains also evaluated. Behavioral results: Adolescents and adults took fewer cards as risk increased. Children, adolescents, and adults took more cards as returns increased. fMRI whole brain results: For risk, adolescent peak in activation in right anterior insula into inferior frontal gyrus and DMPFC; also bilateral caudate. Risk-related activation in thalamus and superior frontal gyrus for all age groups and de-activation in left prefrontal and bilateral parietal areas. Adolescents behavioral risk sensitivity negatively associated with neural activation in DMPFC and insula. Return-related activation in VMPFC increased linearly with age, as did medial OFC and subcallosal ACC and posterior cingulate cortex (PCC). Adolescent-specific activation in visual cortex and right superior parietal cortex. Higher return-sensitivity from behavioral session associated with greater VMPFC and PCC activation as well as superior parietal cortex. For gain > baseline, thalamus and striatal activation increased with increasing gains. |
| Barkley-Levenson & Galván (2017) | n = 51, 25 adolescents and 26 young adults |
Eye blink rate as proxy for striatal dopamine receptor function. Roulette Game, a novel version of value allocation task. “Wheel” (pie chart) gambles with a 1/3 probability of gaining money (ranging from +$3.50 to +$8), a 1/3 probability of losing money (ranging from −$4 to −$8.50) and a 1/3 probability of receiving $0. After viewing the gamble, participants were presented with an amount of money (ranging from $1 to $2.50) and instructed to add that amount of money to one of the three spaces on the wheel, changing the value of that gamble. |
Increased blink rate was associated with increased use of Gmax (reward seeking) strategy for adolescents but not for adults. Consistent with the adolescent ventral striatum’s hypersensitivity to reward observed with neuroimaging. Frequencies of Pwin and Gmax strategies did not differ from each other but were lower than Lmin strategy; age groups did not differ significantly. (Task modification makes distinguishing strategies more difficult.) Adolescents show more value sensitivity (to increased gain amounts) than adults. |
| van Duijvenvoorde et al. (2016) | n = 269 after exclusions, aged 8-25 yr (part of “larger-scale study”) |
Resting state connectivity. Used seed-based approach: mask of functional activation in DLPFC based on learning task and anatomical mask of NAcc based on reward-processing task. Feedback learning task: 3 empty squares with stimulus below; task is to sort stimuli into correct square. Feedback = “+” for correct sort or “−” for incorrect sort. Sequence ended after 12 trials or placing correct item twice for each stimulus. 15 sequences total. Learning phase and application phase (trials where stimulus previously sorted correctly and continued to be sorted correctly). Learning rate = % trials in learning phase where feedback successfully used on next trial. Reward task: heads or tails task (similar to above); used ratings of how much participants liked winning and losing in the task (only 8-17-yr olds, n = 235 had these data) |
Age-related changes in functional connectivity for DLPFC: Linear increase in connectivity with subcortical regions (e.g., bilateral caudate, putamen, and thalamus) and cortical regions (bilateral insula, inferior frontal gyrus). Linear age-related decrease in functional connectivity with pre-SMA, motor cortex, and precuneus. Individuals with higher learning rate had higher connectivity between DLPFC and thalamus and lower connectivity between DLPFC and motor regions. Older participants performed learning task more effectively; effect of age on learning partially mediated by functional connectivity with DLPFC and thalamus (and premotor cortex and SMA but primarily driven by thalamus). Age-related changes in functional connectivity with NAcc: Linear increase with age in functional connectivity with other subcortical regions (bilateral hippocampus, thalamus, caudate) and cortical regions (ACC, left insula, precuneus). Linear age-related decrease with medial OFC/subgenual ACC (VMPFC). Adolescent-specific heightened connectivity with hippocampus. Winning pleasure positively correlated with connectivity with VMPFC (regardless of age). Older age associated with decreased pleasure from winning; partly mediated by functional connectivity between NAcc and VMPFC. |
| Blankenstein et al. (2018) |
n = 198 (after exclusions) 11-24 yr (part of “Braintime” longitudinal study) |
Wheel of Fortune: choice between two wheels—safe option (always 3€) or gamble. Gamble could involve known or unknown (ambiguous) probability. For known probability, 30 trials were 50% gain probability, 8 were 25%, and 8 were 75%. Gain amount was 31€, 32€, 33€, or 34€. For ambiguous, the whole gamble wheel was covered with a question mark. Total of 46 risky and ambiguous trials each, intermixed. Participants had 3 seconds to make the choice. Choice and outcome phases measured separately. Incentive compatible. For choice, looked at Risk Gamble, Ambiguity Gamble, Risk Safe, and Ambiguity Safe (collapsing over levels of probability). For outcome, looked at Risk Gain, Risk No Gain, Ambiguity Gain, Ambiguity No Gain, Risk Gain Safe, Ambiguity Gain Safe (again collapsing over probability). Included Risky and Ambiguous gambling in GLM (controlling for other). Risky option always higher in EV (at least double, sometimes much more). |
No correlation between task behavior and self-report measures (ARQ and BIS/BAS). Participants took gamble ~75% of the time under risk and ambiguity (r = 0.686). Choice Risky Gamble > Ambiguous Gamble: greater activation in bilateral precentral gyrus, right VLPFC, posterior parietal cortex. Ambiguous Gamble > Risky Gamble: greater activation in DLPFC, bilateral temporal lobe, inferior parietal cortex, and precuneus. Activation in superior parietal lobe increased with age. From supplement: Gamble > Safe (in risky and ambiguous condition): increased activation in supplementary motor area, anterior cingulate cortex, right vl and DLPFC Safe > Gamble: right anterior insula Risk > Ambiguity (for safe or gamble choice): increased activation in right DLPFC Ambiguity > Risk: increased activation in left DLPFC and precuneus In regressions with task behavior, positive effect of gambling in Risky Gamble > Ambiguous Gamble in VS (controlling for gambling under ambiguity). Negative effect of ambiguous gambling in bilateral insula, DMPFC, dorsal ACC/SMA for Ambiguous Gamble > Risk Gamble. Outcome Gain > No gain: increased activation in bilateral striatum, VMPFC, PPC, angular gyrus. Greater activation in superior parietal and motor cortex for younger ages. Bigger differences for ambiguity condition in MPFC. In regressions with task behavior, negative effect of ambiguous gambling in right DLPFC and right superior temporal gyrus in Gain > No Gain. In regressions with self-reported measures, negative effect of ARQ rebellious in bilateral DLPFC in Gain > No Gain contrast. Similar results for left DLPFC with BAS drive. |
| Insel & Somerville (2018) | n = 74 (after exclusions), 13-20 yr |
“Magnitude tracking” = increase in striatal activity with reward magnitude, increase in anterior insula activity with increasing loss magnitude – specific to outcomes. Participants see a card with a question mark and told there is a number between 1 and 9 on the other side (but not 5). They have to guess whether the number is higher or lower than 5. If they are correct, they won money, and if they are wrong, they lost money. Low stakes condition: +$0.20 / −$0.10 High stakes condition: +$1.00 / −$0.50 Participants told earnings would be paid in full (actually everyone got $15). 24 blocks (each block was either high or low stakes) presented randomly; for each choice, 1.5 s to decide and feedback for 1 s. |
fMRI analyses: • Gain magnitude tracking: high vs. low (+$1 vs. +$0.20): bilateral VS (nucleus accumbens), dorsal striatum (caudate). For age trend—no regions significant for linear or quadratic predictors but for exploratory analyses using striatum mask, linear trend revelated—greater magnitude tracking in younger participants within right caudate, decreased with age. • Loss magnitude tracking: high vs. low (−$0.50 vs. −$0.10): right insula, right frontal pole, right middle frontal gyrus, bilateral cingulate, bilateral thalamus. For age trend, inverted-U (though in mid-adolescence)—significant activity in right anterior insula. • Reward reactivity: gains vs. losses (collapsing over magnitude): ventral striatum (nucleus accumbens), dorsal striatum (caudate and putamen), cingulate, OFC, precuneus. No regions significant for age-related trends. |
| Schreuders, Braams et al. (2018) |
n = 287, 8-29 yr (part of “Braintime”) Looked at adolescents younger than aged 16 yr and aged 16 yr and older |
Heads-or-tails gambling game Guessed heads or tails on each trial; correct guess = win coins, incorrect guess = lose coins. Always 50% chance of winning. Incentive compatible. From supplement: participants start with 10 coins. Could win 3 or lose 3; win 5 or lose 3; win 2 or lose 5. No sure option. Other scales: BIS/BAS and pleasure from winning/losing. Used anatomical mask for NAcc |
Age-related trends: NAcc activation for win > lose peaked around age 15. Pleasure from winning – losing declined linearly with age. For BAS subscales, mainly cubic trend (except linear increase for females with BAS drive). For younger adolescents (under 16), stronger BAS drive scores associated with greater activation in NAcc in Win > Lose. For older adolescents (16 and older), more pleasure from winning vs. losing associated with greater activation in NAcc in Win > Lose. |
| Blankenstein & van Duijvenvoorde (2019) |
n = 188 (after exclusions) [part of “Braintime”] |
fMRI: Wheel of Fortune task same as above. Behavioral: version of wheel of fortune task but more probabilities (0.125, 0.25, 0.375, 0.5, 0.625, 0.75) and amounts (5€, 8€, 20€, 50€) and different ambiguity levels (0%, 25%, 50%, 75%, 100%). Used model-based estimation of risk and ambiguity attitudes. No decision outcomes provided. 24 risk trials, 16 ambiguity trials (both presented twice = 80 total trials). Power utility function used to estimate risk and ambiguity attitudes. Participants did fMRI task first, then behavioral task. Subjective value under risk and ambiguity in fMRI task calculated based on risk and ambiguity attitudes from behavioral task. |
Model-based risk and ambiguity attitudes: risk seeking increased with age, no age effects observed for ambiguity. Risk and ambiguity attitudes not correlated. Behavior in fMRI task: no age effects for gambling in risky or ambiguous situations. Risk seeking attitude (from behavioral task) positively correlated with gambling under risk and ambiguity in fMRI task. Ambiguity seeking attitude positively correlated with gambling under ambiguity in fMRI task. fMRI results: • Subjective value under risk: Increased activation in bilateral VS, bilateral superior parietal cortex, postcentral gyrus, mid-cingulate cortex, and SMA with increasing subjective value. Decreased activation with DMPFC and right inferior parietal lobe with increasing subjective value. • Subjective value under ambiguity: decreased activation in DMPFC, bilateral DLPFC, right superior temporal gyrus, and bilateral IPL with increasing subjective value. • Conjunction analysis – decreasing activation in DMPFC with subjective value (both risk and ambiguity). • Greater activation in DLPFC and parietal cortex for subjective value under risk than ambiguity. • No effects of age re: parametric tracking of subjective expected value under risk and ambiguity. Compare with Levy et al. (2010)—striatum and medial PFC represented subjective value under risk and ambiguity. (n = 22, ages 19-35 yr); different task. |
| Korucuoglu et al. (2020) | n = 256 (after exclusions) adolescents (aged 12-15 yr) |
Adjusted BART • Out-of-scanner version: earn $0.01 per pump, can cash-out at any time. 30 balloons (trials) with 64-pump breakpoint. Outcomes: adjusted pumps = average number of pumps for cashed-out balloons; total pumps = total pumps in exploded and cashed-out balloons; number of cashed-out balloons; number of explosions; average number of pumps for exploded balloons. • In-scanner version: max of 12 pumps per balloon, probability of explosion and earnings increased monotonically (e.g., for second pump, 2.1% chance of exploding and reward was $0.05; for 12th pump, 89.6% chance of exploding and $5.15 reward). “Risk” defined as increasing probability of balloon popping AND increasing potential reward (probability of loss and reward confounded). |
Choosing to inflate balloon (“risky decisions”) associated with regions in salience, dorsal attention, and frontoparietal networks but only insula sensitive to increasing “risks” (increasing probability of popping and increasing magnitude of reward) in parametric analyses. Similar networks covaried with level of risk for risk-averse decisions (cashing out). Dorsal striatum engaged when choosing to inflate and cash out; not sensitive to level of “risk.” ROI, not whole brain, approach used for fMRI analyses. |
| Blankenstein, Telzer et al. (2020) |
n = 210 at T3, 8-29 yr (part of “Braintime”) |
Structural MRI (no task). ROIs: NACC and MPFC (superior frontal, rostral anterior cingulate, caudal anterior cingulate) Other measures: ARQ rebelliousness subscale, Prosocial behavior, BIS/BAS, Interpersonal Reactivity Index, Intention to Comfort |
Prosocial behavior and ARQ rebellious behavior at T3 positively correlated. BAS fun seeking predicted both behaviors. Greater reductions in MPFC volume associated with lower levels of ARQ rebellious behavior at T3 (effect remained significant when prosocial behavior added to model). |
| Braams, Davidow, & Somerville (2021) | n = 65 after exclusions, 14-22 yr |
Choice between $5 for sure or a risky lottery that was either low or high magnitude (chance of $0 or $3 to $76). Chance of winning in lottery varied from 20 to 80%. Ambiguity by covering party of lottery bar; varied between 0 and 80% covered proportionally based on change of winning. Risk tolerance and ambiguity aversion estimates for each participant derived through computational modeling framework. Choices presented in solo or peer condition (social safe or social risky). 108 trials presented in scanner (ambiguity levels 0%, 40%, and 80%); 72 additional trials presented outside scanner (ambiguity levels 20% and 60%). On some trials participants saw choices of “peers.” Risky peer made 75% risky choices and 25% safe; Safe peer made 75% safe choices and 25% risky. Unequal expected value; lottery had higher EV in 70% of trials. |
Behavioral results: participants chose risk option more in social risky compared with solo and less in social safe compared with solo. Computational models: increased risk tolerance in social risky condition and decreased risk tolerance in social safe condition; changes in risk tolerance not related to age. For ambiguity aversion, not impacted by social condition and no age-related differences. fMRI results: Social > solo whole brain: activation in temporoparietal junction and precuneus. • Increased activation with increased riskiness in VS and ACC • Increased activation with decreased riskiness in occipital cortex. • Increased activation with increased ambiguity in precuneus, superior frontal gyrus. • Increased activation with decreased ambiguity in temporal pole and supramarginal gyrus. Social risky > solo: activation in left MPFC higher for participants who showed higher difference in risk tolerance between social risk and solo. |
Publications are ordered chronologically. fMRI = functional magnetic resonance imaging; VS = ventral striatum; NAcc = nucleus accumbens; ACC = anterior cingulate cortex; PCC = posterior cingulate cortex; PPC = posterior parietal cortex; IPL = inferior parietal lobe; VMPFC = ventromedial prefrontal cortex; DLPFC = dorsolateral prefrontal cortex; SMA = supplemental motor area; ARQ = Adolescent Risk Questionnaire; BIS = Behavioral Inhibition Scale; BAS = Behavioral Activation Scale
Summarized in Table 3, Barkley-Levenson and Galván (2017) used a modified version of the critical value allocation task, which allows some differentiation of strategies. Eye blink rate was used as a proxy for striatal dopamine receptor function (but see Dang et al., 2017). Consistent with the adolescent ventral striatum’s hypersensitivity to reward observed with neuroimaging, eye blink rate was associated with increased use of the compensatory Gmax (reward seeking) strategy for adolescents—but not for adults. Indeed, adolescents were more sensitive to increases in gains than adults were, replicating findings for expected value in neuroimaging research (Barkley-Levenson and Galván, 2014) as well as the behavioral results reviewed earlier (e.g., Kwak et al., 2015).
Although sensitivity to reward is a hallmark of neural imbalance theories of adolescent risky decision making, the theory does not predict that adults would be less deliberative and less likely to use compensatory strategies compared with adolescents. Fuzzy-trace theory predicts the latter effects (see also Niebaum et al., 2022) and accounts for linear developmental trends in risk preference observed in meta-analyses (Defoe et al., 2015). In this view, VS “provide[s] cost and benefit signals necessary for verbatim-based processing engaged more heavily in adolescence; greater EBR [eye-blink rate] in this model could reflect greater reliance on verbatim-based relative to gist-based processing and would therefore correlate with increased focus on reward magnitude” (Barkley-Levenson and Galván, 2017, p. 8). In this role, VS would reflect reward valuation as opposed to salience.
Temporal uncertainty
As van den Bos et al. (2015) argue, impatient or impulsive choices in the delay discounting task can result from valuing immediacy of rewards (present bias) or discounting the future. The VS seems to reflect what is valued: in this instance, not the larger reward as in probabilistic decision making but the immediate reward. In this connection, van den Bos et al. (2015) report that adolescents are more impatient than adults, but this was not tied to reward valuation directly (“hedonism”). As development proceeded from adolescence to adulthood, falling discount rates reflected increased structural connectivity with DLPFC and increased functional connectivity (negative coupling) between DLPFC and VS. Thus, developmental differences were attributed to maturation of executive functions, such as cognitive control and greater future orientation.
More generally, when rewards are offered immediately, a single value-related signal has been identified at two locations: the ventral striatum (VS) and the ventromedial prefrontal cortex (VMPFC) (McClure et al., 2004). Later rewards do not easily elicit this reward-related signal. When decision-makers take longer to respond, thought to reflect patient choosing, activation has been observed in the DLPFC and the PPC. This work has been interpreted as evidence for a beta (impatient) module and an independent delta (patient) module predicted by theory. However, independence has been challenged by findings that increasing rewards in the later option elicited activation in regions partial to immediacy, VS and the VMPFC (Kable and Glimcher, 2007).
As we argued at the outset, and as suggested by the brain activations associated with each, probabilistic uncertainty and temporal uncertainty seem to share processing components. The effects of manipulating the presence or absence of zero (or “nothing”) to induce shifts in gist (categorical “something vs. nothing” contrasts in decision problems) or verbatim processing (computing expected value or utility) has been used in both probabilistic decision making and temporal discounting tasks (Reyna et al., 2014, 2021; Zhang and Slovic, 2018). For example, in temporal discounting tasks, making hidden zeros explicit increases patience: a sooner smaller reward but nothing later versus a larger later reward but nothing now (Rahimi-Golkhandan et al., 2017). This good-news/bad-news explication seems to create more decision conflict for impulsive choosers than an objectively identical formulation without the explicit zeros that may be processed at a more fine-grained level.
Consistent with this framework, repeated Transcranial Magnetic Stimulation (rTMS) to the DLPFC (which takes that region offline temporarily) in a delay discounting task had two different kinds of effects (Ballard et al., 2018). Large magnitudes (presumably because they make the delayed option differentially attractive relative to the immediate option) and explicit zero manipulations (presumably because they change the gist) reduced discounting (choosing the sooner option) relative to control groups. However, rTMS to DLPFC reduced both the magnitude effect relative to baseline and reduced the hidden zero effect relative to baseline; the latter effect was “unanticipated” by Ballard et al. (2018). These effects are consistent with the idea that DLPFC encompasses both gist representations as found by Venkatraman et al. (2009) and other higher order mental functions, such as cognitive control and associated inhibition (Ballard et al., 2018; Figner et al., 2010).
An integrated framework: Implications for addiction
Figure 3 brings together many of the components that we have reviewed (and others we have not). A major novel aspect of the framework is combining fuzzy-trace theory with research from alternative theories, including major decision theories, such as prospect theory and similar expected utility approaches. The implications of the framework can be illustrated by the example of addiction. Addiction often is initiated in adolescence or young adulthood and is thought to reflect heightened attraction to reward and immature cognitive control systems (including impulsivity and delay discounting, Amlung et al., 2017). As shown in the figure, these factors are likely to play a role in initiation that has been documented, although confounds plague this literature due to the difficulties of pulling apart factors, such as risk opportunity, risk preference, reward valuation, impatience/impulsivity, and underdevelopment of cognitive control and inhibition (Romer, Reyna, and Satterthwaite, 2017).
Fig. 3.
Provisional integrative framework for probabilistic and temporal uncertainty. Black text = probabilistic uncertainty and valuation tasks. Blue text = temporal uncertainty tasks. Red text = tasks that discriminate gist strategies Badre & Wagner, 2004; Ballard & Knutson, 2009; Canessa et al., 2013; Cooper et al., 2013; Cox et al., 2019; Dixon & Christoff, 2014; Dorris & Glimcher, 2004; Droutman et al., 2015; Figner et al., 2010; Frost & McNaughton, 2017; Hare et al., 2009; He et al., 2016; Jocham et al., 2014; Kable & Glimcher, 2007, 2010; Kahnt et al., 2011; Knoch et al., 2006; Kolling et al., 2012; Liston et al., 2006; Ludwig et al., 2015; Mahler et al., 2009; Manuel et al., 2019; Massar et al., 2015; McClure et al., 2004, 2007; Monterosso et al., 2007; Peters & Büchel, 2009, 2010, 2011; Pochon et al., 2008; Reyna & Huettel, 2014; Rodriguez et al., 2015; Rushworth et al., 2012; Schonberg et al., 2011; Schüller et al., 2019; Sellitto et al., 2016; Singer et al., 2009; Steinbeis et al., 2012; Venkatraman et al., 2009; Wittmann et al., 2007; Wood et al., 2014; Zalocusky et al., 2016
The core brain areas associated with probabilistic and intertemporal decisions largely overlap with those assumed to play a crucial role in the development and maintenance of addictive disorders (Brand, 2022; Volkow and Baler, 2015). Volkow and Baler (2015) proposed the so-called “NOW vs. LATER circuits” crucially involved in addiction and other pathologies. Consistent with our framework, cortical regions, especially DLPFC, MPFC, VMPFC, OFC, and ACC, and subcortical regions, especially dorsal and ventral striatum are assumed to be involved in NOW vs. LATER decisions. Main processes are driven by tonic dopamine signals in the DLPFC (favoring choices for later rewards) and phasic dopamine signals triggering VMPFC and ventral striatum leading to choices of immediate rewards (but see Kable and Glimcher, 2010). Several signals from other regions, including OFC, insula, and amygdala, interact with the circuits and mediate through the processing of internal states, experiences, and information/estimation about possible alternative options and their associated levels of uncertainty. It is assumed that repeatedly choosing immediate gratifications (e.g., drugs, instant lotteries, or highly processed food) can reinforce preexisting vulnerabilities in the two circuits.
Research on substance related and nonsubstance related addictions as well as animal models suggest that, during the development of addiction, there is a shift from involvement of ventral to dorsal striatum related circuits (Brand et al., 2019; Fineberg et al., 2010; Zhou et al., 2018). Brand (2022) assumes the ventral striatum to be especially involved in early stages of the addiction process as part of the so-called “feels better” path characterized by positive and negative reinforcement mechanisms and related reward expectancies. In later stages, the dorsal striatum is assumed to be more and more involved as part of the additional “must do” path associated with compulsive, habitual behaviors. The DLPFC and other regions associated with self-control are engaged in the “stop now” process, which is assumed to counteract the two paths (Brand, 2022).
Clinical implications mostly point to the importance to reduce discounting by improving self-control and reducing impulsivity. However, there might be other routes to reduce vulnerability to addiction (addressing similar networks, but less effortful strategies) as we discuss below. The DLPFC may play a crucial role here. As stated earlier, this region is not only involved in inhibition and other higher-order cognitive control, but it also is associated with decisions reflecting gist representations and affectively valenced principles. Accordingly, simple gist-based strategies could counteract the driving paths to addiction (also on a neural level) and support health-promoting choices.
From this fuzzy-trace-theory based foundation, various practical implications can be derived, including implications for interventions and public health communications (e.g., Reyna and Farley, 2006; Reyna, 2018; Wolfe et al., 2015). Principles of fuzzy-trace theory can be used to design the content of prevention and intervention programs by presenting (additional) information in a gist-promoting way (supporting avoidance of unhealthy risks) that is consistent with people’s preexisting values, thereby reducing reliance on policy mandates. For example, following up on Reyna and Mills (2014) on interventions for reducing sexual risk, addiction prevention programs could add information on the basic bottom-line meaning of decision options in addition to the exact numbers on risks. At the policy level, it can be inferred that instead of dictating “right” choices or trying to change people’s values, another path can be taken to promote healthy choices. Presenting the gist of possible options in an accurate and understandable way can enable individuals to link the decision to their own relevant values, which in turn promotes (self-generated) advantageous decisions and sustainable positive impact on behavioral outcomes.
An important factor that we have not discussed in detail is that adolescence and young adulthood are periods of exploration in which risky behaviors are initiated despite ambiguity (Tymula et al., 2012). That is, adolescents are primed to approach ambiguity rather than to be averse to it, an attraction to uncertainty that is not typical of other stages of life. Initiation of substance use seems to be a function of such willingness to approach despite uncertainty, plus reward and control factors, but also because of the public health paradox to which young people are more susceptible. As we have discussed, adolescents are more likely to weigh risk against reward and to process them using compensatory strategies. This “rationality bias” makes them vulnerable to normatively rare but individually catastrophic outcomes, such as addiction.
Additionally, in adolescence there is a peak in sensation seeking and reward sensitivity (Galván, 2013; Reyna et al., 2011; but see Insel and Somerville, 2018; van Duijvenvoorde et al., 2014), which may lead to (the subjective value of) the experience of immediate reward outweighing the uncertainty caused by potential risks. Ambiguity seeking also could trigger experimenting with drugs, because one does not know what (possibly very rewarding event) one is missing. However, neither reward seeking nor ambiguity seeking is sufficient by themselves to explain developmental differences in risk preference. Major developmental trends and prediction of risky behavior are linked to increasing emphasis with development on categorical gist in risky decision making—such as it only takes once to get HIV-AIDS. What begins in experimentation can progress to addiction much like a bullet fired in Russian roulette, a low probability event. It is low probability, because most adolescents escape the phase of experimentation, especially if they have mitigating factors, such as higher levels of cognitive control or lower levels of attraction to reward.
Our framework suggests that there is another System—a System 3 approach to reducing vulnerability to addiction, including inducing categorical gist thinking about risk, that is distinct from the difficult tasks of trying to modify either System 1 unthinking attraction to reward or System 2 deliberation and cognitive control (Reyna and Mills, 2014). This approach harnesses gist-based intuition as advanced but automatic cognition; grounded in research on learning and memory, it suggests methods for recognizing gist and encouraging retrieval of relevant values in context, processes that can operate largely unconsciously. We see enormous potential in bringing together the process mechanisms of fuzzy-trace theory with etiological process models of addiction for deriving more effective prevention and intervention programs. This approach would not necessarily seek to change the level of perceived risk in a verbatim sense, but it should decrease risk-taking when probabilities of adverse events are low and uncertain but outcomes are catastrophic—bridging a predictable gap in risk communication between policymakers and the public.
In this view, the processes of the DLPFC may consist of more than white-knuckled self-control but, rather, reappraising risky situations and connecting them more clearly to salient affective valence (simple good-bad signals) and social values. Results also suggest that what is rewarding undergoes reappraisal with development and experience, so that what is patient and in the future becomes more highly valued.
More generally, it is time to reevaluate traditional ways of thinking about uncertainty whether probabilistic or temporal. Evidence about anomalies in choice and valuation need to be integrated with major theories of decision-making to better develop interventions that preserve well-being. The meaningful categorical gist of decisions is not a one-off strategy for solving toy problems but is how mature decision-makers approach most decision problems by default. Communicating effectively with those decision makers requires understanding their perspective.
Footnotes
Support for the preparation of this manuscript was provided in part by grants from the National Science Foundation (SES-2029420) and from the National Institute of Standards and Technology (60NANB22D052).
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Valerie F. Reyna, Email: vr53@cornell.edu
Silke M. Müller, Email: silke.m.mueller@uni-due.de
Sarah M. Edelson, Email: sme28@cornell.edu
References
- Amlung M, Vedelago L, Acker J, Balodis I, MacKillop J. Steep delay discounting and addictive behavior: A metaanalysis of continuous associations. Addiction. 2017;112(1):51–62. doi: 10.1111/add.13535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Augenblick N, Rabin M. An experiment on time preference and misprediction in unpleasant tasks. Review of Economic Studies. 2019;86(3):941–975. doi: 10.1093/restud/rdy019. [DOI] [Google Scholar]
- Badre D, Wagner AD. Selection integration and conflict monitoring. Neuron. 2004;41(3):473–487. doi: 10.1016/S0896-6273(03)00851-1. [DOI] [PubMed] [Google Scholar]
- Ballard K, Knutson B. Dissociable neural representations of future reward magnitude and delay during temporal discounting. Neuroimage. 2009;45(1):143–150. doi: 10.1016/j.neuroimage.2008.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballard IC, Aydogan G, Kim B, et al. Causal evidence for the dependence of the magnitude effect on dorsolateral prefrontal cortex. Scientific Reports. 2018;8:16545. doi: 10.1038/s41598-018-34900-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barkley-Levenson E, Galván A. Neural representation of expected value in the adolescent brain. Proceedings of the National Academy of Sciences. 2014;111(4):1646–1651. doi: 10.1073/pnas.1319762111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barkley-Levenson E, Galván A. Eye blink rate predicts reward decisions in adolescents. Developmental Science. 2017;20(3):e12412. doi: 10.1111/desc.12412. [DOI] [PubMed] [Google Scholar]
- Barraclough DJ, Conroy ML, Lee D. Prefrontal cortex and decision making in a mixed-strategy game. Nature Neuroscience. 2004;7(4):404–410. doi: 10.1038/nn1209. [DOI] [PubMed] [Google Scholar]
- Bartels DM, Rips LJ. Psychological connectedness and intertemporal choice. Journal of Experimental Psychology. General. 2010;139:49–69. doi: 10.1037/a0018062. [DOI] [PubMed] [Google Scholar]
- Bjork JM, Pardini DA. Who are those “risk-taking adolescents”? Individual differences in developmental neuroimaging research. Developmental Cognitive Neuroscience. 2015;11:56–64. doi: 10.1016/j.dcn.2014.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blankenstein NE, van Duijvenvoorde AC. Neural tracking of subjective value under risk and ambiguity in adolescence. Cognitive, Affective, & Behavioral Neuroscience. 2019;19(6):1364–1378. doi: 10.3758/s13415-019-00749-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blankenstein NE, Schreuders E, Peper JS, Crone EA, van Duijvenvoorde AC. Individual differences in risk-taking tendencies modulate the neural processing of risky and ambiguous decision-making in adolescence. NeuroImage. 2018;172:663–673. doi: 10.1016/j.neuroimage.2018.01.085. [DOI] [PubMed] [Google Scholar]
- Blankenstein NE, Telzer EH, Do KT, Van Duijvenvoorde AC, Crone EA. Behavioral and neural pathways supporting the development of prosocial and risk-taking behavior across adolescence. Child Development. 2020;91(3):e665–e681. doi: 10.1111/cdev.13292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boily MC, Baggaley RF, Wang L, Masse B, White RG, Hayes RJ, Alary M. Heterosexual risk of HIV-1 infection per sexual act: Systematic review and meta-analysis of observational studies. The Lancet. Infectious diseases. 2009;9(2):118–129. doi: 10.1016/S1473-3099(09)70021-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyd CJ, Teter CJ, West BT, Morales M, McCabe SE. Non-medical use of prescription analgesics: A three-year national longitudinal study. Journal of Addictive Diseases. 2009;28(3):232–242. doi: 10.1080/10550880903028452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braams BR, Davidow JY, Somerville LH. Information about others’ choices selectively alters risk tolerance and medial prefrontal cortex activation across adolescence and young adulthood. Developmental Cognitive Neuroscience. 2021;52:101039. doi: 10.1016/j.dcn.2021.101039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brainerd CJ, Gordon LL. Development of verbatim and gist memory for numbers. Developmental Psychology. 1994;30(2):163–177. doi: 10.1037/0012-1649.30.2.163. [DOI] [Google Scholar]
- Brand M. Can internet use become addictive? Science. 2022;376(6595):798–799. doi: 10.1126/science.abn4189. [DOI] [PubMed] [Google Scholar]
- Brand M, Wegmann E, Stark R, Müller A, Wölfling K, Robbins TW, Potenza MN. The interaction of person affect-cognition-execution (I-PACE) model for addictive behaviors: Update, generalization to addictive behaviors beyond internet-use disorders, and specification of the process character of addictive behaviors. Neuroscience & Biobehavioral Reviews. 2019;104:1–10. doi: 10.1016/j.neubiorev.2019.06.032. [DOI] [PubMed] [Google Scholar]
- Brandstätter E, Gigerenzer G, Hertwig R. The priority heuristic: Making choices without trade-offs. Psychological Review. 2006;113(2):409–432. doi: 10.1037/0033-295X.113.2.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broniatowski DA, Reyna VF. A formal model of fuzzy-trace theory: Variations on framing effects and the Allais paradox. Decision. 2018;5(4):205–252. doi: 10.1037/dec0000083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canessa N, Crespi C, Motterlini M, Baud-Bovy G, Chierchia G, Pantaleo G, Cappa SF. The functional and structural neural basis of individual differences in loss aversion. Journal of Neuroscience. 2013;33(36):14307–14317. doi: 10.1523/JNEUROSCI.0497-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chick CF, Reyna VF, Corbin JC. Framing effects are robust to linguistic disambiguation: A critical test of contemporary theory. Journal of Experimental Psychology: Learning, Memory, and Cognition. 2016;42(2):238–256. doi: 10.1037/xlm0000158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark HH, Clark EV. Psychology and language. Journal of Child Language. 1977;4(2):b1–b3. doi: 10.1017/S0305000900001562. [DOI] [Google Scholar]
- Cooper N, Kable JW, Kim BK, Zauberman G. Brain activity in valuation regions while thinking about the future predicts individual discount rates. Journal of Neuroscience. 2013;33(32):13150–13156. doi: 10.1523/JNEUROSCI.0400-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corbin JC, Reyna VF, Weldon RB, Brainerd CJ. How reasoning, judgment, and decision making are colored by gistbased intuition: A fuzzy-trace theory approach. Journal of Applied Research in Memory and Cognition. 2015;4(4):344–355. doi: 10.1016/j.jarmac.2015.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coricelli G, Diecidue E, Zaffuto FD. Evidence for multiple strategies in choice under risk. Journal of Risk and Uncertainty. 2018;56:193–210. doi: 10.1007/s11166-018-9278-2. [DOI] [Google Scholar]
- Cox J, Witten IB. Striatal circuits for reward learning and decision-making. Nature Reviews Neuroscience. 2019;20(8):482–494. doi: 10.1038/s41583-019-0189-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Croote DE, Lai B, Hu J, et al. Delay discounting decisions are linked to temporal distance representations of world events across cultures. Scientific Reports. 2020;10:12913. doi: 10.1038/s41598-020-69700-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang LC, Samanez-Larkin GR, Castrellon JJ, Perkins SF, Cowan RL, Newhouse PA, Zald DH. Spontaneous eye blink rate (EBR) is uncorrelated with dopamine D2 receptor availability and unmodulated by dopamine agonism in healthy adults. eNeuro. 2017;4(5):ENEURO.0211-17.2017. doi: 10.1523/ENEURO.0211-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Neys W, editor. Dual process theory 2.0. Routledge/Taylor & Francis Group; 2018. [Google Scholar]
- Defoe, I. N., & Romer, D. (2022). Theoretical advances in research on the development of risk taking. Developmental Review, 63, Article 101001. 10.1016/j.dr.2021.101001
- Defoe IN, Dubas JS, Figner B, van Aken MAG. A meta-analysis on age differences in risky decision making: Adolescents versus children and adults. Psychological Bulletin. 2015;141(1):48–84. doi: 10.1037/a0038088. [DOI] [PubMed] [Google Scholar]
- DeKay, M. L., Rubinchik, N., Li, Z., & De Boeck, P. (2022). Accelerating psychological science with metastudies: A demonstration using the risky-choice framing effect. Perspectives on Psychological Science, 17456916221079611. 10.1177/17456916221079611 [DOI] [PubMed]
- Dixon ML, Christoff K. The lateral prefrontal cortex and complex value-based learning and decision making. Neuroscience & Biobehavioral Reviews. 2014;45:9–18. doi: 10.1016/j.neubiorev.2014.04.011. [DOI] [PubMed] [Google Scholar]
- Dorris MC, Glimcher PW. Activity in posterior parietal cortex is correlated with the relative subjective desirability of action. Neuron. 2004;44(2):365–378. doi: 10.1016/j.neuron.2004.09.009. [DOI] [PubMed] [Google Scholar]
- Droutman V, Bechara A, Read SJ. Roles of the different sub-regions of the insular cortex in various phases of the decision-making process. Frontiers in Behavioral Neuroscience. 2015;9:309. doi: 10.3389/fnbeh.2015.00309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edelson S, Reyna V. How fuzzy-trace theory predicts development of risky decision making, with novel extensions to culture and reward sensitivity. Developmental Review. 2021;62:100986. doi: 10.1016/j.dr.2021.100986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figner B, Knoch D, Johnson EJ, Krosch AR, Lisanby SH, Fehr E, Weber EU. Lateral prefrontal cortex and selfcontrol in intertemporal choice. Nature Neuroscience. 2010;13(5):538–539. doi: 10.1038/nn.2516. [DOI] [PubMed] [Google Scholar]
- Fineberg NA, Potenza MN, Chamberlain SR, Berlin HA, Menzies L, Bechara A, Hollander E. Probing compulsive and impulsive behaviors, from animal models to endophenotypes: A narrative review. Neuropsychopharmacology. 2010;35(3):591–604. doi: 10.1038/npp.2009.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer GW, Hawkins SA. Strategy compatibility, scale compatibility, and the prominence effect. Journal of Experimental Psychology: Human Perception and Performance. 1993;19(3):580–597. doi: 10.1037/0096-1523.19.3.580. [DOI] [Google Scholar]
- Fisher, S. A. (2021). Framing effects and fuzzy traces: ‘Some’ observations. Review of Philosophy and Psychology.10.1007/s13164-021-00556-3
- Fisher, S. A. (2022). A critical notice of: Frame it again: New tools for rational decision-making. International Journal of Philosophical Studies.10.1080/09672559.2022.2057685
- Frost R, McNaughton N. The neural basis of delay discounting: A review and preliminary model. Neuroscience & Biobehavioral Reviews. 2017;79:48–65. doi: 10.1016/j.neubiorev.2017.04.022. [DOI] [PubMed] [Google Scholar]
- Gaissmaier W, Schooler LJ. The smart potential behind probability matching. Cognition. 2008;109(3):416–422. doi: 10.1016/j.cognition.2008.09.007. [DOI] [PubMed] [Google Scholar]
- Galván A. The teenage brain: Sensitivity to rewards. Current Directions in Psychological Science. 2013;22(2):88–93. doi: 10.1177/0963721413480859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geurts B. Alternatives in framing and decision making. Mind & Language. 2013;28:1–19. doi: 10.1111/mila.12005. [DOI] [Google Scholar]
- Gianotti LRR, Knoch D, Faber PL, Lehmann D, Pascual-Marqui RD, Diezi C, Schoch C, Eisenegger C, Fehr E. Tonic activity level in the right prefrontal cortex predicts individuals’ risk taking. Psychological Science. 2009;20(1):33–38. doi: 10.1111/j.1467-9280.2008.02260.x. [DOI] [PubMed] [Google Scholar]
- Glimcher PW. Efficiently irrational: Deciphering the riddle of human choice. Trends in Cognitive Sciences. 2022;26(8):669–687. doi: 10.1016/j.tics.2022.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glöckner A, Herbold AK. An eye-tracking study on information processing in risky decisions: Evidence for compensatory strategies based on automatic processes. Journal of Behavioral Decision Making. 2011;24(1):71–98. doi: 10.1002/bdm.684. [DOI] [Google Scholar]
- Hare TA, Camerer CF, Rangel A. Self-control in decision-making involves modulation of the vmPFC valuation system. Science. 2009;324(5927):646–648. doi: 10.1126/science.1168450. [DOI] [PubMed] [Google Scholar]
- He Q, Chen M, Chen C, Xue G, Feng T, Bechara A. Anodal stimulation of the left DLPFC increases IGT scores and decreases delay discounting rate in healthy males. Frontiers in Psychology. 2016;7:1421. doi: 10.3389/fpsyg.2016.01421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershfield HE. Future self-continuity: How conceptions of the future self transform intertemporal choice. Annals of the New York Academy of Sciences. 2011;1235:30–43. doi: 10.1111/j.1749-6632.2011.06201.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huettel SA, Song AW, McCarthy G. Decisions under uncertainty: Probabilistic context influences activation of prefrontal and parietal cortices. Journal of Neuroscience. 2005;25(13):3304–3311. doi: 10.1523/JNEUROSCI.5070-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Insel C, Somerville LH. Asymmetric neural tracking of gain and loss magnitude during adolescence. Social Cognitive and Affective Neuroscience. 2018;13(8):785–796. doi: 10.1093/scan/nsy058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Insurance Institute for Highway Safety (2022). Fatality facts 2020: State by State. Retrieved December, 2022, from https://www.iihs.org/topics/fatality-statistics/detail/state-by-state.
- Jocham G, Furlong PM, Kröger IL, Kahn MC, Hunt LT, Behrens TE. Dissociable contributions of ventromedial prefrontal and posterior parietal cortex to value-guided choice. NeuroImage. 2014;100:498–506. doi: 10.1016/j.neuroimage.2014.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson EJ, Schulte-Mecklenbeck M, Willemsen MC. Process models deserve process data: Comment on Brandstätter, Gigerenzer, and Hertwig (2006) Psychological Review. 2008;115(1):263–272. doi: 10.1037/0033-295X.115.1.263. [DOI] [PubMed] [Google Scholar]
- Kable JW, Glimcher PW. The neural correlates of subjective value during intertemporal choice. Nature Neuroscience. 2007;10(12):1625–1633. doi: 10.1038/nn2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kable JW, Glimcher PW. An "as soon as possible" effect in human intertemporal decision making: Behavioral evidence and neural mechanisms. Journal of Neurophysiology. 2010;103(5):2513–2531. doi: 10.1152/jn.00177.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahneman D, Tversky A. Prospect theory: An analysis of decision under risk. Econometrica. 1979;47(2):263–292. doi: 10.2307/1914185. [DOI] [Google Scholar]
- Kahnt T, Heinzle J, Park SQ, Haynes JD. Decoding different roles for vmPFC and dlPFC in multi-attribute decision making. Neuroimage. 2011;56(2):709–715. doi: 10.1016/j.neuroimage.2010.05.058. [DOI] [PubMed] [Google Scholar]
- Kahnt T, Park SQ, Haynes J-D, Tobler PN. Disentangling neural representations of value and salience in the human brain. PNAS proceedings of the National Academy of Sciences of the United States of America. 2014;111(13):5000–5005. doi: 10.1073/pnas.1320189111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keren G, Schul Y. Two is not always better than one: A critical evaluation of two-system theories. Perspectives on Psychological Science. 2009;4(6):533–550. doi: 10.1111/2Fj.1745-6924.2009.01164.x. [DOI] [PubMed] [Google Scholar]
- Knoch D, Pascual-Leone A, Meyer K, Treyer V, Fehr E. Diminishing reciprocal fairness by disrupting the right prefrontal cortex. Science. 2006;314(5800):829–832. doi: 10.1126/science.1129156. [DOI] [PubMed] [Google Scholar]
- Kogut T, Slovic P. The development of scope insensitivity in sharing behavior. Journal of Experimental Psychology: Learning, Memory, and Cognition. 2016;42(12):1972–1981. doi: 10.1037/xlm0000296. [DOI] [PubMed] [Google Scholar]
- Kolling N, Behrens TE, Mars RB, Rushworth MF. Neural mechanisms of foraging. Science. 2012;336(6077):95–98. doi: 10.1126/science.1216930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korucuoglu O, Harms MP, Kennedy JT, Golosheykin S, Astafiev SV, Barch DM, Anokhin AP. Adolescent decision-making under risk: Neural correlates and sex differences. Cerebral Cortex. 2020;30(4):2691–2707. doi: 10.1093/cercor/bhz269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kühberger A. The framing of decisions: A new look at old problems. Organizational Behavior and Human Decision Processes. 1995;62(2):230–240. doi: 10.1006/obhd.1995.1046. [DOI] [Google Scholar]
- Kühberger A, Luger-Bazinger C. Predicting framed decisions: Simulation or theory? Psychology. 2016;7(6):941–953. doi: 10.4236/psych.2016.76095. [DOI] [Google Scholar]
- Kühberger A, Tanner C. Risky choice framing: Task versions and a comparison of prospect theory and fuzzy-trace theory. Journal of Behavioral Decision Making. 2010;23(3):314–329. doi: 10.1002/bdm.656. [DOI] [Google Scholar]
- Kuhnen CM, Knutson B. The neural basis of financial risk taking. Neuron. 2005;47(5):763–770. doi: 10.1016/j.neuron.2005.08.008. [DOI] [PubMed] [Google Scholar]
- Kwak Y, Payne JW, Cohen AL, Huettel SA. The rational adolescent: Strategic information processing during decision making revealed by eye tracking. Cognitive Development. 2015;36:20–30. doi: 10.1016/j.cogdev.2015.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laibson, D., & Maxted, (2022). The beta-delta DELTA sweet spot (August 4, 2022). Retrieved August 27, 2022 from https://static1.squarespace.com/static/6186b3b155561c2ab5fe4957/t/62ec891071319e27e0250046/1659668753724/Beta_Delta_DELTA.pdf
- Levine DS. One or two minds? Neural network modeling of decision making by the unified self. Neural Networks. 2019;120:74–85. doi: 10.1016/j.neunet.2019.08.008. [DOI] [PubMed] [Google Scholar]
- Levy I, Schiller D. Neural computations of threat. Trends in Cognitive Sciences. 2021;25(2):151–171. doi: 10.1016/j.tics.2020.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy I, Snell J, Nelson AJ, Rustichini A, Glimcher PW. Neural representation of subjective value under risk and ambiguity. Journal of Neurophysiology. 2010;103(2):1036–1047. doi: 10.1152/jn.00853.2009. [DOI] [PubMed] [Google Scholar]
- Li R, Smith DV, Clithero JA, Venkatraman V, Carter RM, Huettel SA. Reason's enemy is not emotion: Engagement of cognitive control networks explains biases in gain/loss framing. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience. 2017;37(13):3588–3598. doi: 10.1523/JNEUROSCI.3486-16.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liston C, Matalon S, Hare TA, Davidson MC, Casey BJ. Anterior cingulate and posterior parietal cortices are sensitive to dissociable forms of conflict in a task-switching paradigm. Neuron. 2006;50(4):643–653. doi: 10.1016/j.neuron.2006.04.015. [DOI] [PubMed] [Google Scholar]
- Ludwig VU, Nüsser C, Goschke T, Wittfoth-Schardt D, Wiers CE, Erk S, Walter H. Delay discounting without decision-making: medial prefrontal cortex and amygdala activations reflect immediacy processing and correlate with impulsivity and anxious-depressive traits. Frontiers in Behavioral Neuroscience. 2015;9:280. doi: 10.3389/fnbeh.2015.00280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahler SV, Berridge KC. Which cue to “want?” Central amygdala opioid activation enhances and focuses incentive salience on a prepotent reward cue. Journal of Neuroscience. 2009;29(20):6500–6513. doi: 10.1523/JNEUROSCI.3875-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandel DR. Gain-loss framing and choice: Separating outcome formulations from descriptor formulations. Organizational Behavior and Human Decision Processes. 2001;85(1):56–76. doi: 10.1006/obhd.2000.2932. [DOI] [PubMed] [Google Scholar]
- Mandel DR. Do framing effects reveal irrational choice? Journal of Experimental Psychology: General. 2014;143(3):1185–1198. doi: 10.1037/a0034207. [DOI] [PubMed] [Google Scholar]
- Manuel AL, Murray NW, Piguet O. Transcranial direct current stimulation (tDCS) over vmPFC modulates interactions between reward and emotion in delay discounting. Scientific Reports. 2019;9(1):1–9. doi: 10.1038/s41598-019-55157-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mar J, Liu J. Findings of the Association for Computational Linguistics: NAACL 2022. Association for Computational Linguistics; 2022. From cognitive to computational modeling: Text-based risky decision-making guided by fuzzy trace theory; pp. 391–409. [Google Scholar]
- Massar SA, Libedinsky C, Weiyan C, Huettel SA, Chee MW. Separate and overlapping brain areas encode subjective value during delay and effort discounting. Neuroimage. 2015;120:104–113. doi: 10.1016/j.neuroimage.2015.06.080. [DOI] [PubMed] [Google Scholar]
- McCabe SE, West BT, Morales M, Cranford JA, Boyd CJ. Does early onset of non-medical use of prescription drugs predict subsequent prescription drug abuse and dependence? Results from a national study. Addiction. 2007;102(12):1920–1930. doi: 10.1111/j.1360-0443.2007.02015.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClure SM, Laibson DI, Loewenstein G, Cohen JD. Separate neural systems value immediate and delayed monetary rewards. Science. 2004;306(5695):503–507. doi: 10.1126/science.1100907. [DOI] [PubMed] [Google Scholar]
- McClure SM, Ericson KM, Laibson DI, Loewenstein G, Cohen JD. Time discounting for primary rewards. Journal of Neuroscience. 2007;27(21):5796–5804. doi: 10.1523/JNEUROSCI.4246-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melnikoff DE, Bargh JA. The mythical number two. Trends in Cognitive Sciences. 2018;22(4):280–293. doi: 10.1016/j.tics.2018.02.001. [DOI] [PubMed] [Google Scholar]
- Monterosso JR, Ainslie G, Xu J, Cordova X, Domier CP, London ED. Frontoparietal cortical activity of methamphetamine-dependent and comparison subjects performing a delay discounting task. Human Brain Mapping. 2007;28(5):383–393. doi: 10.1002/hbm.20281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niebaum, J. C., Kramer, A.-W., Huizenga, H. M., & van den Bos, W. (2022). Adolescents sample more information prior to decisions than adults when effort costs increase. Developmental Psychology.10.1037/dev0001397 [DOI] [PubMed]
- Padilla LMK, Creem-Regehr SH, Thompson W. The powerful influence of marks: Visual and knowledge-driven processing in hurricane track displays. Journal of Experimental Psychology: Applied. 2020;26(1):1–15. doi: 10.1037/xap0000245. [DOI] [PubMed] [Google Scholar]
- Pardilla-Delgado E, Payne JD. The impact of sleep on true and false memory across long delays. Neurobiology of Learning and Memory. 2017;137(123-133):2017. doi: 10.1016/j.nlm.2016.11.016. [DOI] [PubMed] [Google Scholar]
- Paulus MP, Hozack N, Zauscher B, McDowell JE, Frank L, Brown GG, Braff DL. Prefrontal, parietal, and temporal cortex networks underlie decision-making in the presence of uncertainty. Neuroimage. 2001;13(1):91–100. doi: 10.1006/nimg.2000.0667. [DOI] [PubMed] [Google Scholar]
- Paulus MP, Rogalsky C, Simmons A, Feinstein JS, Stein MB. Increased activation in the right insula during risk taking decision making is related to harm avoidance and neuroticism. Neuroimage. 2003;19(4):1439–1448. doi: 10.1016/S1053-8119(03)00251-9. [DOI] [PubMed] [Google Scholar]
- Payne JW. It is whether you win or lose: The importance of the overall probabilities of winning or losing in risky choice. Journal of Risk and Uncertainty. 2005;30(1):5–19. doi: 10.1007/s11166-005-5831-x. [DOI] [Google Scholar]
- Payne JD, Schacter DL, Propper RE, Huang LW, Wamsley EJ, Tucker MA, Stickgold R. The role of sleep in false memory formation. Neurobiology of Learning and Memory. 2009;92(3):327–334. doi: 10.1016/j.nlm.2009.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pennycook, G. (in press). A framework for understanding reasoning errors: From fake news to climate change and beyond. Advances in Experimental Social Psychology.
- Pennycook G, Rand DG. Lazy, not biased: Susceptibility to partisan fake news is better explained by lack of reasoning than by motivated reasoning. Cognition. 2019;188:39–50. doi: 10.1016/j.cognition.2018.06.011. [DOI] [PubMed] [Google Scholar]
- Peters J, Büchel C. Overlapping and distinct neural systems code for subjective value during intertemporal and risky decision making. Journal of Neuroscience. 2009;29(50):15727–15734. doi: 10.1523/JNEUROSCI.3489-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters J, Büchel C. Episodic future thinking reduces reward delay discounting through an enhancement of prefrontalmediotemporal interactions. Neuron. 2010;66(1):138–148. doi: 10.1016/j.neuron.2010.03.026. [DOI] [PubMed] [Google Scholar]
- Peters J, Büchel C. The neural mechanisms of inter-temporal decision-making: understanding variability. Trends in Cognitive Sciences. 2011;15(5):227–239. doi: 10.1016/j.tics.2011.03.002. [DOI] [PubMed] [Google Scholar]
- Pirrone A, Reina A, Stafford T, Marshall JAR, Gobet F. Magnitude-sensitivity: Rethinking decision-making. Trends in Cognitive Sciences. 2022;26(1):66–80. doi: 10.1016/j.tics.2021.10.006. [DOI] [PubMed] [Google Scholar]
- Pochon JB, Riis J, Sanfey AG, Nystrom LE, Cohen JD. Functional imaging of decision conflict. Journal of Neuroscience. 2008;28(13):3468–3473. doi: 10.1523/JNEUROSCI.4195-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preuschoff K, Quartz SR, Bossaerts P. Human insula activation reflects risk prediction errors as well as risk. Journal of Neuroscience. 2008;28(11):2745–2752. doi: 10.1523/JNEUROSCI.4286-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahimi-Golkhandan S, Garavito DMN, Reyna-Brainerd BB, Reyna VF. A fuzzy-trace theory of risk and time preferences in decision making: Integrating cognition and motivation. In: Stevens J, editor. Impulsivity, Nebraska symposium on motivation 64. Springer; 2017. pp. 115–144. [PubMed] [Google Scholar]
- Raoelison M, Thompson VA, De Neys W. The smart intuitor: Cognitive capacity predicts intuitive rather than deliberate thinking. Cognition. 2020;204:104381. doi: 10.1016/j.cognition.2020.104381. [DOI] [PubMed] [Google Scholar]
- Reyna VF. A new intuitionism: Meaning, memory, and development in fuzzy-trace theory. Judgment and Decision making. 2012;7(3):332–359. doi: 10.1017/S1930297500002291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyna V. When irrational biases are smart: a fuzzy-trace theory of complex decision making. Journal of Intelligence. 2018;6(2):29. doi: 10.3390/jintelligence6020029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyna, V. F. (2021). A scientific theory of gist communication and misinformation resistance, with implications for health, education, and policy. PNAS Proceedings of the National Academy of Sciences of the United States of America 118(5). 10.1073/pnas.1912441117 [DOI] [PMC free article] [PubMed]
- Reyna VF, Brainerd CJ. Fuzzy-trace theory and framing effects in choice: Gist extraction, truncation, and conversion. Journal of Behavioral Decision Making. 1991;4(4):249–262. doi: 10.1002/bdm.3960040403. [DOI] [Google Scholar]
- Reyna VF, Brainerd CJ. Dual processes in decision making and developmental neuroscience: A fuzzy-trace model. Developmental Review. 2011;31(2-3):180–206. doi: 10.1016/j.dr.2011.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyna VF, Brainerd CJ, Chen Z, Bookbinder SH. Explaining risky choices with judgments: Framing, the zero effect, and the contextual relativity of gist. Journal of Experimental Psychology: Learning, Memory, and Cognition. 2021;47(7):1037–1053. doi: 10.1037/xlm0001016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyna VF, Broniatowski DA, Edelson SM. Viruses, vaccines, and COVID-19: Explaining and improving risky decision-making. Journal of Applied Research in Memory and Cognition. 2021;10(4):491–509. doi: 10.1016/j.jarmac.2021.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyna VF, Brust-Renck PG. How representations of number and numeracy predict decision paradoxes: A fuzzy-trace theory approach. Journal of Behavioral Decision Making. 2020;33(5):606–628. doi: 10.1002/bdm.2179. [DOI] [Google Scholar]
- Reyna VF, Ellis SC. Fuzzy-trace theory and framing effects in children's risky decision making. Psychological Science. 1994;5(5):275–279. doi: 10.1111/j.1467-9280.1994.tb00625.x. [DOI] [Google Scholar]
- Reyna VF, Farley F. Risk and rationality in adolescent decision making: Implications for theory, practice, and public policy. Psychological Science in the Public Interest. 2006;7(1):1–44. doi: 10.1111/j.1529-1006.2006.00026.x. [DOI] [PubMed] [Google Scholar]
- Reyna VF, Huettel SA. Reward, representation, and impulsivity: A theoretical framework for the neuroscience of risky decision making. In: Reyna VF, Zayas V, editors. The neuroscience of risky decision making. American Psychological Association; 2014. pp. 11–42. [Google Scholar]
- Reyna VF, Mills B. Converging evidence supports fuzzy-trace theory's nested sets hypothesis, but not the frequency hypothesis. Behavioral and Brain Sciences. 2007;30(3):278–280. doi: 10.1017/S0140525X07001872. [DOI] [Google Scholar]
- Reyna VF, Mills BA. Theoretically motivated interventions for reducing sexual risk taking in adolescence: A randomized controlled experiment applying fuzzy-trace theory. Journal of Experimental Psychology: General. 2014;143(4):1627–1648. doi: 10.1037/a0036717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyna VF, Estrada SM, DeMarinis JA, Myers RM, Stanisz JM, Mills BA. Neurobiological and memory models of risky decision making in adolescents versus young adults. Journal of Experimental Psychology: Learning, Memory, and Cognition. 2011;37(5):1125–1142. doi: 10.1037/a0023943. [DOI] [PubMed] [Google Scholar]
- Reyna VF, Chick CF, Corbin JC, Hsia AN. Developmental reversals in risky decision making: Intelligence agents show larger decision biases than college students. Psychological Science. 2014;25(1):76–84. doi: 10.1177/0956797613497022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyna VF, Helm RK, Weldon RB, Shah PD, Turpin AG, Govindgari S. Brain activation covaries with reported criminal behaviors when making risky choices: A fuzzy-trace theory approach. Journal of Experimental Psychology: General. 2018;147(7):1094–1109. doi: 10.1037/xge0000434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivers SE, Reyna VF, Mills B. Risk taking under the influence: A fuzzy-trace theory of emotion in adolescence. Developmental Review. 2008;28(1):107–144. doi: 10.1016/j.dr.2007.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez CA, Turner BM, Van Zandt T, McClure SM. The neural basis of value accumulation in intertemporal choice. European Journal of Neuroscience. 2015;42(5):2179–2189. doi: 10.1111/ejn.12997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romer D, Reyna VF, Satterthwaite TD. Beyond stereotypes of adolescent risk taking: Placing the adolescent brain in developmental context. Developmental Cognitive Neuroscience. 2017;27:19–34. doi: 10.1016/j.dcn.2017.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rottenstreich Y, Hsee CK. Money, kisses, and electric shocks: On the affective psychology of risk. Psychological Science. 2001;12(3):185–190. doi: 10.1111/1467-9280.00334. [DOI] [PubMed] [Google Scholar]
- Rushworth MF, Kolling N, Sallet J, Mars RB. Valuation and decision-making in frontal cortex: one or many serial or parallel systems? Current Opinion in Neurobiology. 2012;22(6):946–955. doi: 10.1016/j.conb.2012.04.011. [DOI] [PubMed] [Google Scholar]
- Sanfey AG, Rilling JK, Aronson JA, Nystrom LE, Cohen JD. The neural basis of economic decision-making in the ultimatum game. Science. 2003;300(5626):1755–1758. doi: 10.1126/science.1082976. [DOI] [PubMed] [Google Scholar]
- Schonberg T, Fox CR, Poldrack RA. Mind the gap: bridging economic and naturalistic risk-taking with cognitive neuroscience. Trends in Cognitive Sciences. 2011;15(1):11–19. doi: 10.1016/j.tics.2010.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreuders E, Braams BR, Blankenstein NE, Peper JS, Güroğlu B, Crone EA. Contributions of reward sensitivity to ventral striatum activity across adolescence and early adulthood. Child Development. 2018;89(3):797–810. doi: 10.1111/cdev.13056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schüller CB, Kuhn J, Jessen F, Hu X. Neuronal correlates of delay discounting in healthy subjects and its implication for addiction: An ALE meta-analysis study. The American Journal of Drug and Alcohol Abuse. 2019;45(1):51–66. doi: 10.1080/00952990.2018.1557675. [DOI] [PubMed] [Google Scholar]
- Sellitto M, Ciaramelli E, Mattioli F, Di Pellegrino G. Reduced sensitivity to sooner reward during intertemporal decisionmaking following insula damage in humans. Frontiers in Behavioral Neuroscience. 2016;9:367. doi: 10.3389/fnbeh.2015.00367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simmons, J., & Nelson, L. (2013). "Exactly": The most famous framing effect is robust to precise wording. Retrieved from http://datacolada.org/11.
- Sinclair AH, Hakimi S, Stanley ML, Adcock RA, Samanez-Larkin GR. Pairing facts with imagined consequences improves pandemic-related risk perception. PNAS Proceedings of the National Academy of Sciences of the United States of America. 2021;118(32):e2100970118. doi: 10.1073/pnas.2100970118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singer T, Critchley HD, Preuschoff K. A common role of insula in feelings, empathy and uncertainty. Trends in Cognitive Sciences. 2009;13(8):334–340. doi: 10.1016/j.tics.2009.05.001. [DOI] [PubMed] [Google Scholar]
- Sloman SA. The empirical case for two systems of reasoning. Psychological Bulletin. 1996;119(1):3–22. doi: 10.1037/0033-2909.119.1.3. [DOI] [Google Scholar]
- Speer ME, Ibrahim S, Schiller D, et al. Finding positive meaning in memories of negative events adaptively updates memory. Nature Communications. 2021;12:6601. doi: 10.1038/s41467-021-26906-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steiger A, Kühberger A. A meta-analytic re-appraisal of the framing effect. Zeitschrift für Psychologie. 2018;226(1):45–55. doi: 10.1027/2151-2604/a000321. [DOI] [Google Scholar]
- Steinbeis N, Bernhardt BC, Singer T. Impulse control and underlying functions of the left DLPFC mediate age-related and age-independent individual differences in strategic social behavior. Neuron. 2012;73(5):1040–1051. doi: 10.1016/j.neuron.2011.12.027. [DOI] [PubMed] [Google Scholar]
- Studer B, Pedroni A, Rieskamp J. Predicting risk-taking behavior from prefrontal resting-state activity and personality. PLoS One. 2013;8(10):1–8. doi: 10.1371/journal.pone.0076861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Studler M, Gianotti LRR, Koch K, Hausfeld J, Tarokh L, Maric A, Knoch D. Local slow-wave activity over the right prefrontal cortex reveals individual risk preferences. NeuroImage. 2022;253:1–7. doi: 10.1016/j.neuroimage.2022.119086. [DOI] [PubMed] [Google Scholar]
- Teigen KH. When frames meet realities: On the perceived correctness of inaccurate estimates. In: Keren G, editor. Perspectives on framing. Psychology Press; 2011. pp. 197–217. [Google Scholar]
- The COVID Tracking Project. (n.d.). Totals for the U.S.. Retrieved December, 2022, from https://covidtracking.com/data/national#reach-skip-nav
- Thompson CA, Siegler RS. Linear numerical-magnitude representations aid children’s memory for numbers. Psychological Science. 2010;21(9):1274–1281. doi: 10.1177/0956797610378309. [DOI] [PubMed] [Google Scholar]
- Tobler PN, O'Doherty JP, Dolan RJ, Schultz W. Reward value coding distinct from risk attitude-related uncertainty coding in human reward systems. Journal of Neurophysiology. 2007;97(2):1621–1632. doi: 10.1152/jn.00745.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trepel C, Fox CR, Poldrack RA. Prospect theory on the brain? Toward a cognitive neuroscience of decision under risk. Cognitive Brain Research. 2005;23(1):34–50. doi: 10.1016/j.cogbrainres.2005.01.016. [DOI] [PubMed] [Google Scholar]
- Trippas D, Thompson VA, Handley SJ. When fast logic meets slow belief: Evidence for a parallel-processing model of belief bias. Memory & Cognition. 2017;45(4):539–552. doi: 10.3758/s13421-016-0680-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tversky A, Kahneman D. Rational choice and the framing of decisions. The Journal of Business. 1986;59:S251–S278. doi: 10.1086/296365. [DOI] [Google Scholar]
- Tymula A, Rosenberg Belmaker LA, Roy AK, Ruderman L, Manson K, Glimcher PW, Levy I. Adolescents’ risktaking behavior is driven by tolerance to ambiguity. Proceedings of the National Academy of Sciences. 2012;109(42):17135–17140. doi: 10.1073/pnas.1207144109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- U.S. Department of Health and Human Services. (2022, December 16). Opioid facts and statistics. Retrieved January 25, 2023, from https://www.hhs.gov/opioids/statistics/index.html
- Van Den Bos W, Rodriguez CA, Schweitzer JB, McClure SM. Adolescent impatience decreases with increased frontostriatal connectivity. Proceedings of the National Academy of Sciences. 2015;112(29):E3765–E3774. doi: 10.1073/pnas.1423095112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Duijvenvoorde AC, de Macks ZAO, Overgaauw S, Moor BG, Dahl RE, Crone EA. A cross-sectional and longitudinal analysis of reward-related brain activation: effects of age, pubertal stage, and reward sensitivity. Brain and Cognition. 2014;89:3–14. doi: 10.1016/j.bandc.2013.10.005. [DOI] [PubMed] [Google Scholar]
- van Duijvenvoorde AC, Huizenga HM, Somerville LH, Delgado MR, Powers A, Weeda WD, Figner B. Neural correlates of expected risks and returns in risky choice across development. Journal of Neuroscience. 2015;35(4):1549–1560. doi: 10.1523/JNEUROSCI.1924-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Duijvenvoorde AC, Achterberg M, Braams BR, Peters S, Crone EA. Testing a dual-systems model of adolescent brain development using resting-state connectivity analyses. Neuroimage. 2016;124:409–404. doi: 10.1016/j.neuroimage.2015.04.069. [DOI] [PubMed] [Google Scholar]
- Venkatraman V, Payne JW, Bettman JR, Luce MF, Huettel SA. Separate neural mechanisms underlie choices and strategic preferences in risky decision making. Neuron. 2009;62:593–602. doi: 10.1016/j.neuron.2009.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venkatraman V, Huettel SA, Chuah LY, Payne JW, Chee MW. Sleep deprivation biases the neural mechanisms underlying economic preferences. Journal of Neuroscience. 2011;31:3712–3718. doi: 10.1523/JNEUROSCI.4407-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venkatraman V, Payne JW, Huettel SA. An overall probability of winning heuristic for complex risky decisions: Choice and eye fixation evidence. Organizational Behavior and Human Decision Processes. 2014;125(2):73–87. doi: 10.1016/j.obhdp.2014.06.003. [DOI] [Google Scholar]
- Volkow ND, Baler RD. NOW vs LATER brain circuits: Implications for obesity and addiction. Trends in Neurosciences. 2015;38(6):345–352. doi: 10.1016/j.tins.2015.04.002. [DOI] [PubMed] [Google Scholar]
- Wakker PP. Prospect theory: For risk and ambiguity. Cambridge University Press; 2010. [Google Scholar]
- Wittmann M, Leland DS, Paulus MP. Time and decision making: differential contribution of the posterior insular cortex and the striatum during a delay discounting task. Experimental Brain Research. 2007;179(4):643–653. doi: 10.1007/s00221-006-0822-y. [DOI] [PubMed] [Google Scholar]
- Wolfe CR, Reyna VF, Widmer CL, Cedillos EM, Fisher CR, Brust-Renck PG, Weil AM. Efficacy of a webbased intelligent tutoring system for communicating genetic risk of breast cancer: A fuzzy-trace theory approach. Medical Decision Making. 2015;35(1):46–59. doi: 10.1177/0272989X14535983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wood SM, Bechara A. The neuroscience of dual (and triple) systems in decision making. In: Reyna VF, Zayas V, editors. The neuroscience of risky decision making. American Psychological Association; 2014. pp. 177–202. [Google Scholar]
- World Health Organization (2020). Managing the COVID-19 infodemic: Promoting healthy behaviours and mitigating the harm from misinformation and disinformation. Retrieved August 25, 2022 from https://www.who.int/news/item/23-09-2020-managing-the-covid-19-infodemic-promoting-healthy-behaviours-and-mitigating-the-harm-from-misinformation-and-disinformation
- Zalocusky KA, Ramakrishnan C, Lerner TN, Davidson TJ, Knutson B, Deisseroth K. Nucleus accumbens D2R cells signal prior outcomes and control risky decision-making. Nature. 2016;531(7596):642–646. doi: 10.1038/nature17400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Slovic P. Much ado about nothing: The zero effect in life-saving decisions. Journal of Behavioral Decision Making. 2018;32(1):30–37. doi: 10.1002/bdm.2089. [DOI] [Google Scholar]
- Zhou F, Zimmermann K, Xin F, Scheele D, Dau W, Banger M, et al. Shifted balance of dorsal versus ventral striatal communication with frontal reward and regulatory regions in cannabis-dependent males. Human Brain Mapping. 2018;39(12):5062–5073. doi: 10.1002/hbm.24345. [DOI] [PMC free article] [PubMed] [Google Scholar]