Abstract
CD8+ T cells that respond to chronic viral infections or cancer are characterized by the expression of inhibitory receptors such as programmed cell death protein 1 (PD-1) and by the impaired production of cytokines. This state of restrained functionality–which is referred to as T cell exhaustion1,2–is maintained by precursors of exhausted T (TPEX) cells that express the transcription factor T cell factor 1 (TCF1), self-renew and give rise to TCF1− exhausted effector T cells3–6. Here we show that the long-term proliferative potential, multipotency and repopulation capacity of exhausted T cells during chronic infection are selectively preserved in a small population of transcriptionally distinct CD62L+ TPEX cells. The transcription factor MYB is not only essential for the development of CD62L+ TPEX cells and maintenance of the antiviral CD8+ T cell response, but also induces functional exhaustion and thereby prevents lethal immunopathology. Furthermore, the proliferative burst in response to PD-1 checkpoint inhibition originates exclusively from CD62L+ TPEX cells and depends on MYB. Our findings identify CD62L+ TPEX cells as a stem-like population that is central to the maintenance of long-term antiviral immunity and responsiveness to immunotherapy. Moreover, they show that MYB is a transcriptional orchestrator of two fundamental aspects of exhausted T cell responses: the downregulation of effector function and the long-term preservation of self-renewal capacity.
T cell exhaustion is an important physiological adaptation to continuous antigen stimulation in chronic infection and cancer, and although it protects against excessive immune-mediated tissue damage, it also contributes to viral or tumour persistence1,2,4,7. TPEX cells have the ability to continuously self-renew and give rise to functionally restrained effector cells, and therefore have an essential role in maintaining chronically antigen-stimulated T cells and their exhausted phenotype3–5,8,9. TPEX cells also mediate the response to therapeutic checkpoint inhibition3,5,10,11, which can reinvigorate exhausted CD8+ T cell responses and has revolutionized cancer therapy12. In mice, TPEX cells are defined by the co-expression of PD-1, the transcriptional regulators TCF1 and ID3 and the surface molecules CXCR5 and Ly108. By contrast, exhausted effector T (TEX) cells co-express PD-1 and TIM-3 but lack the expression of TCF1, ID3, CXCR5 and Ly108 (refs. 3–6,8,9,13). Thus, exhausted CD8+ T cells constitute a dynamic network of phenotypically and functionally distinct populations that ultimately depend on the functionality of TPEX cells. We and others have shown that TPEX and TEX cells are controlled by specific transcriptional and metabolic networks that support their differentiation and maintenance13–18. It remains, however, unclear how precisely longevity, self-renewal and responsiveness to checkpoint inhibition are orchestrated within the TPEX cell compartment.
CD62L+ TPEX cells have stem-like potential
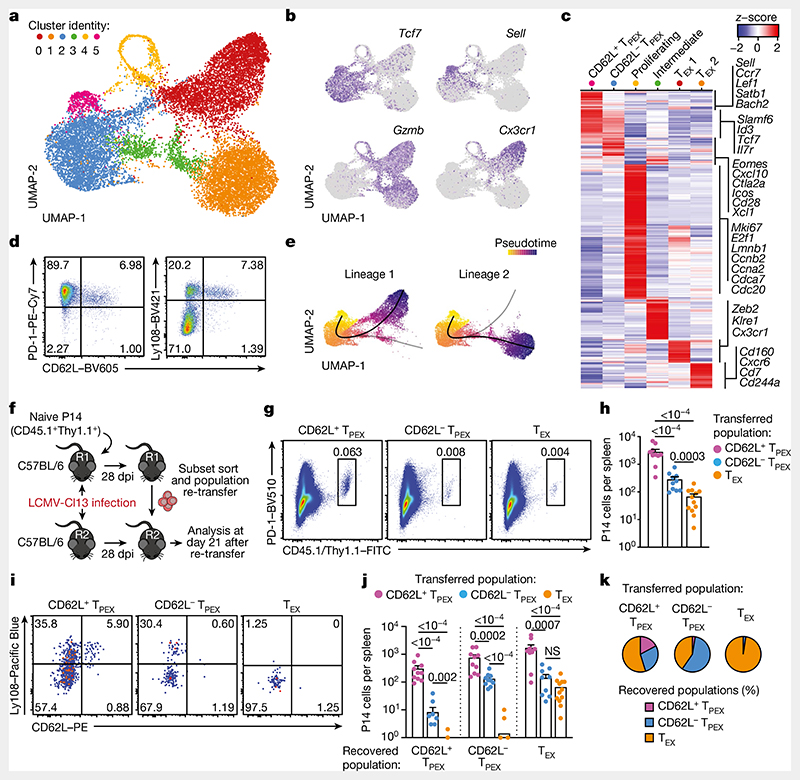
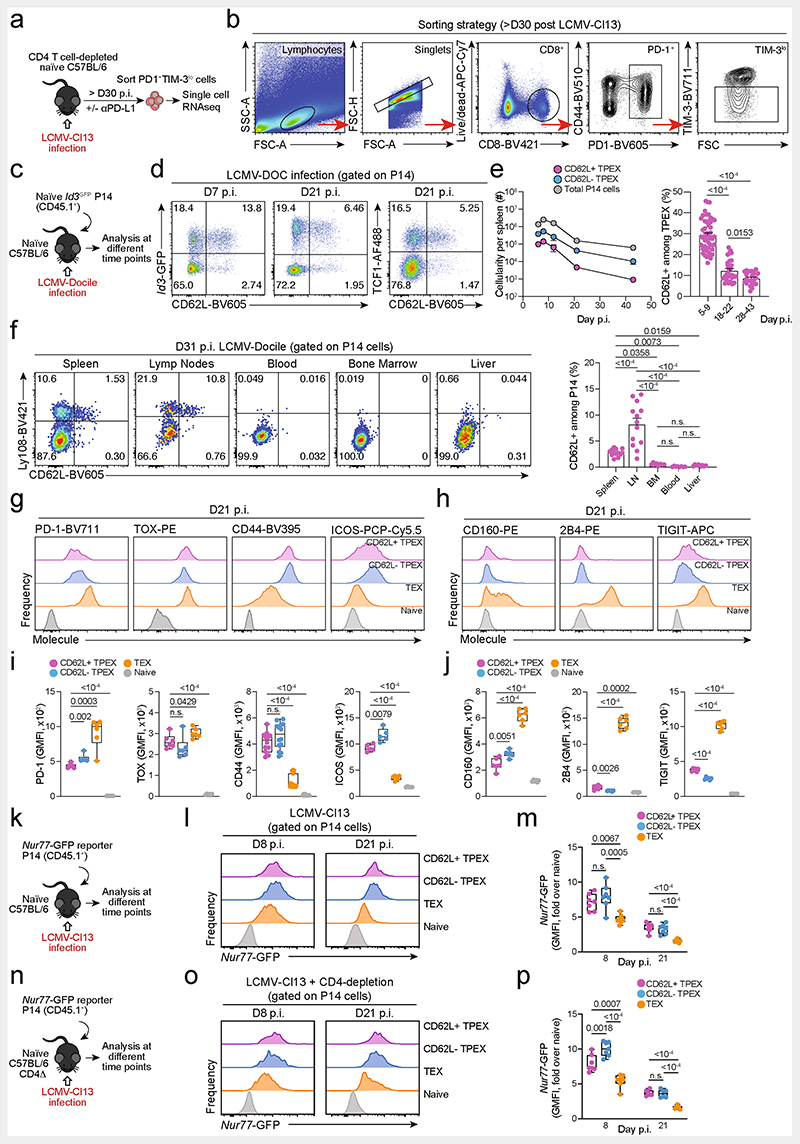
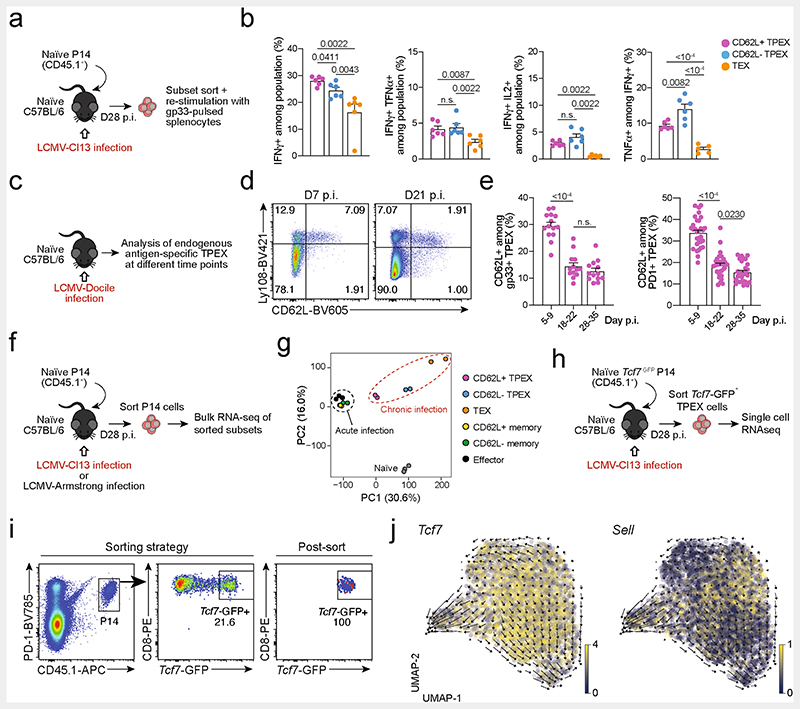
To identify factors that promote the self-renewal and multipotency of TPEX cells, we performed single-cell RNA sequencing (scRNA-seq) of TPEX-cell-enriched (PD-1+TIM-3−) CD8+ T cells sorted at 30 days post-infection (dpi) from mice chronically infected with lymphocytic choriomeningitis virus (LCMV) clone-13 (Cl13). Combined analysis of our data and publicly available scRNA-seq datasets11,19 (Fig. 1a and Extended Data Fig. 1a,b) identified two distinct TPEX cell clusters, both marked by high expression of Tcf7 and Id3 (Fig. 1a–c). The smaller of these clusters was characterized by high expression of transcripts that are typically associated with naive or central memory T cells, including Sell (encoding CD62L), Ccr7, S1pr1, Lef1, Satb1 and Bach2 (referred to as CD62L+ TPEX cells; Fig. 1a–c and Supplementary Table 1). By contrast, the larger TPEX cell cluster showed low expression of Sell but was enriched for other TPEX-cell-associated transcripts, including Icos, Xcl1, Cxcl10, Cd28 and Eomes(CD62L− TPEX cells; Fig. 1a–c and Supplementary Table 1). In line with previous findings20,21, we identified two TEX cell clusters, both marked by the expression of Gzmb and lack of Tcf7, but distinguished by the differential expression of Cx3cr1 (Fig. 1a–c and Supplementary Table 1). The two remaining clusters expressed intermediate levels of both TPEX and TEX cell marker genes (cluster 3) or cell-cycle-related genes such as Mki67, Ccnb2 and E2f1 (cluster 4) (Fig. 1a–c and Supplementary Table 1). To examine the heterogeneity of TPEX cells experimentally, we used CD8+ Id3GFP P14 T cells, which express a transgenic T cell receptor (TCR) specific for the LCMV epitope gp33, and GFP under the control of Id3, specific to TPEX cells13. Id3GFP P14 cells were adoptively transferred into naive mice, which were subsequently inoculated with LCMV-Docile, which causes chronic infection (Fig. 1d and Extended Data Fig. 1c–j). Both early TPEX and TEX cells were readily detectable during the acute phase (5–9 dpi) of the immune response (Extended Data Fig. 1d), and about 30% of TPEX cells expressed CD62L, which gradually declined and stabilized at around 10% by three weeks after infection (Fig. 1d and Extended Data Fig. 1d,e). CD62L+ TPEX cells were enriched in the spleen and lymph nodes, but largely absent from the blood, bone marrow and liver (Extended Data Fig. 1f). CD62L+ and CD62L− TPEX cells expressed high levels of PD-1, the activation marker CD44, the exhaustion-associated transcription factor TOX and the co-stimulatory molecule ICOS (Extended Data Fig. 1g,i), indicating that both populations were chronically stimulated, and both expressed low amounts of CD160, 2B4 and TIGIT (Extended Data Fig. 1h,j). Consistent with the notion that TPEX cells are particularly dependent on strong TCR signals13,15, both CD62L+ and CD62L− TPEX cells expressed higher levels of the TCR-induced transcriptional regulator NUR77 than TEX cells (Extended Data Fig. 1k–p). There were no major differences in cytokine production between the two TPEX subsets, but IFNγ+ cells were enriched among CD62L+ TPEX cells (Extended Data Fig. 2a,b). CD62L+ TPEX cells were also found among endogenous gp33-specific and among polyclonal antigen-responsive PD-1+CD8+ T cells in LCMV-Docile-infected mice (Extended Data Fig. 2c–e). Notably, CD62L+ TPEX cells were transcriptionally distinct from both naive and memory T cells derived from acute LCMV infection (Extended Data Fig. 2f,g).
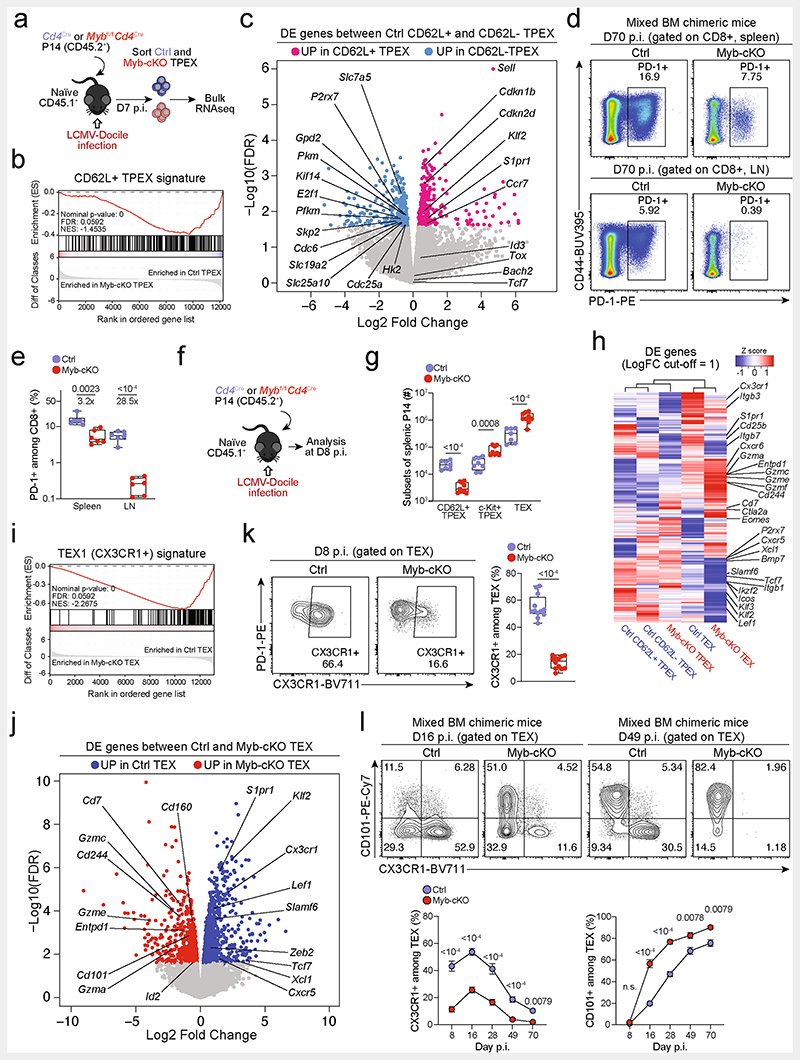
Fig. 1. CD62L marks transcriptionally distinct and functionally superior TPEX cells during chronic infection.
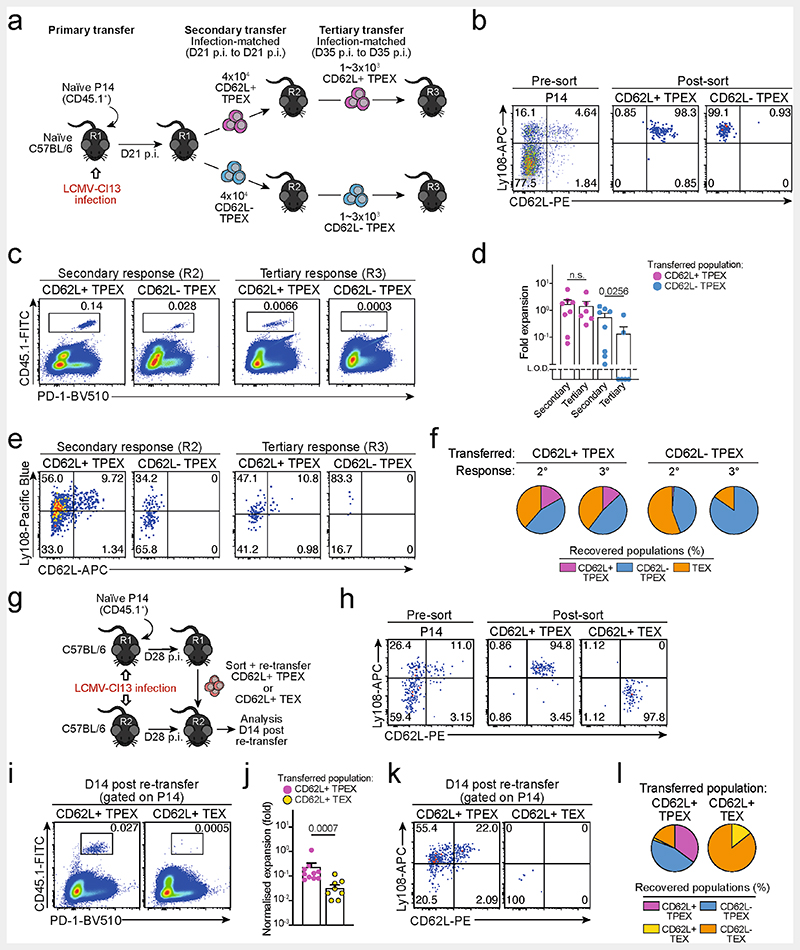
a-c, Naive wild-type mice were infected with LCMV-Cl13 and TPEX-cell-enriched (PD-1+TIM-3lo) CD8+ T cells were sorted and subjected to scRNA-seq at 30 dpi. The resulting data were combined with publicly available scRNA-seq datasets from mouse exhausted CD8+ T cells11,19 and analysed. a, Uniform manifold approximation and projection (UMAP) plot of 15,743 single exhausted T cells coloured according to cluster classification. b, Normalized gene expression of Tcf7, Sell, Gzmb and Cx3cr1 projected onto the UMAP. c, Heat map showing the expression of all identified cluster signature transcripts. d, Congenically marked naive P14 cells were transferred into recipient mice, which were subsequently infected with LCMV-Docile and analysed at 21dpi. Flow cytometry plots show the expression of PD-1, Ly108 and CD62L in splenic P14 T cells. e, UMAP plot showing two predicted developmental trajectories generated using Slingshot analysis. Cells are colour-coded on the basis of pseudotime prediction. f–k, Congenically marked naive P14 T cells were transferred into primary recipient (R1) mice, which were then infected with LCMV-Cl13. The indicated subsets of P14 T cells were sorted at 28 dpi and 3 × 103–15 × 103 cells were re-transferred to infection-matched secondary recipient (R2) mice. Splenic P14 T cells of R2 mice were analysed at day 21 after re-transfer. f, Schematic of the experimental set-up. g,h, Flow cytometry plots (g) and cell numbers (h) of recovered progenies at day 21 after re-transfer (gated on CD4−CD19− cells). i–k, Flow cytometry plots (i), numbers (j) and average percentages (k) of recovered CD62L+ TPEX, CD62L− TPEX and TEX cells per spleen in R2 mice. Cells were gated on P14 cells (day 21 after re-transfer). Dots in graphs represent individual mice (h,j); horizontal lines and error bars of bar graphs indicate mean and s.e.m., respectively. Data are representative of at least two independent experiments. P values are from Mann–Whitney tests (h,j); P > 0.05, not significant (NS).
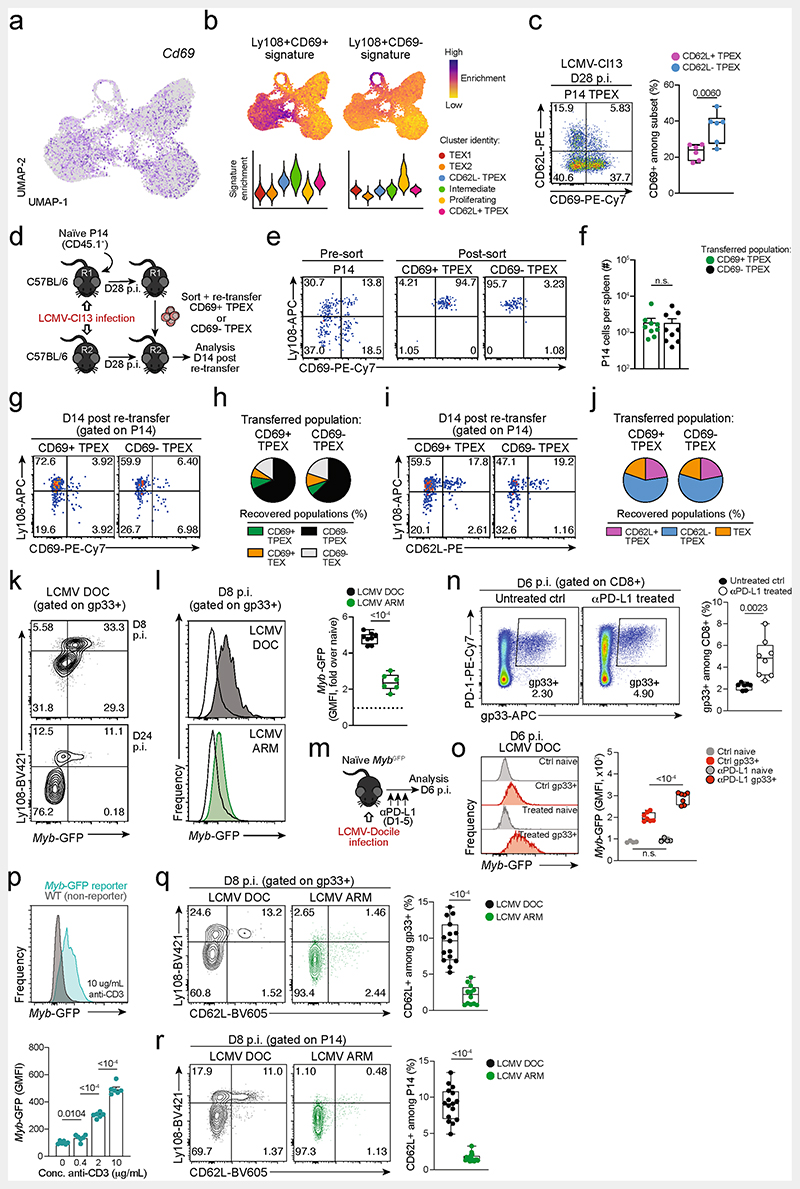
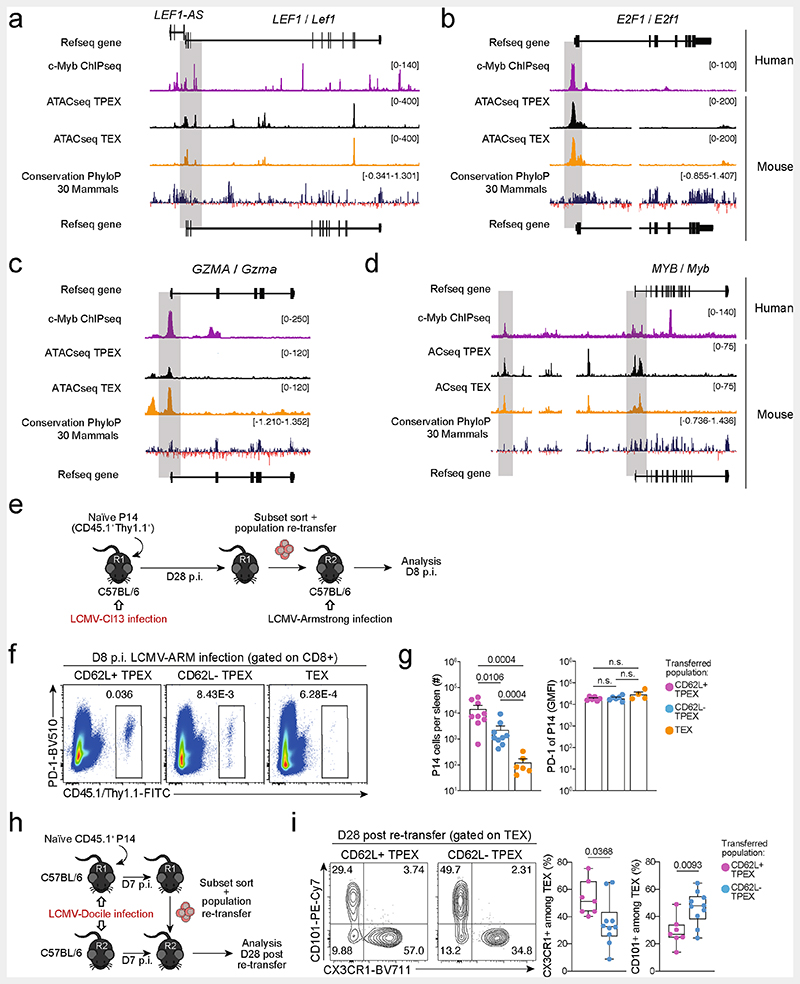
Slingshot analysis of our scRNA-seq data revealed a developmental trajectory that began with CD62L+ TPEX cells and progressed into CD62L− TPEX cells, from which it bifurcated into either CX3CR1+ or CX3CR1− TEX cells (Fig. 1e). Similar results were obtained when we sorted P14 TPEX cells based on a Tcf7GFP reporter from LCMV-Cl13-infected mice and performed scRNA-seq followed by RNA velocity analysis (Extended Data Fig. 2h–j). Overall, these data suggest a one-way developmental trajectory that originates from CD62L+ TPEX cells. To test this model experimentally, we sorted CD62L+ TPEX, CD62L- TPEX and TEX P14 cells on day 28 after infection with LCMV-Cl13, separately re-transferred them into congenically marked infection-matched hosts and analysed three weeks later (Fig. 1f–k). Compared with CD62L− TPEX and TEX cells, CD62L+ TPEX cells showed a superior repopulation capacity (Fig. 1g,h) and were able to efficiently self-renew and give rise to both CD62L- TPEX and TEX cells (Fig. 1i–k). These characteristics were maintained even after repetitive adoptive transfers (Extended Data Fig. 3a–f). By contrast, the few CD62L+ TEX cells that were detected in the P14 compartment (around 1–2%) did not expand or generate progeny efficiently (Extended Data Fig. 3g–l). We confirmed the superior developmental properties of CD62L+ TPEX cells using single T cell transfer and fate-mapping via retrogenic colour barcoding22–25 (Extended Data Fig. 4). Notably, single CD62L+ TPEX cells exhibited self-renewal and multipotent repopulation capacity, akin to single naive T cells (Extended Data Fig. 4a–h). In line with the epigenetic imprint of exhaustion26–28, progeny derived from single CD62L+ TPEX cells maintained high levels of PD-1 expression compared to their naive-derived counterparts (Extended Data Fig. 4d,g,i). The CD62L-linked developmental hierarchy uncovered here is unrelated to previously proposed TPEX cell subsets based on differential CD69 expression29 (Extended Data Fig. 5a–j). Together, these results show that CD62L+ TPEX cells represent a transcriptionally distinct population with stem-like developmental capacity that maintains the responses of exhausted CD8+ T cells during chronic infection.
MYB governs exhausted T cell function and longevity
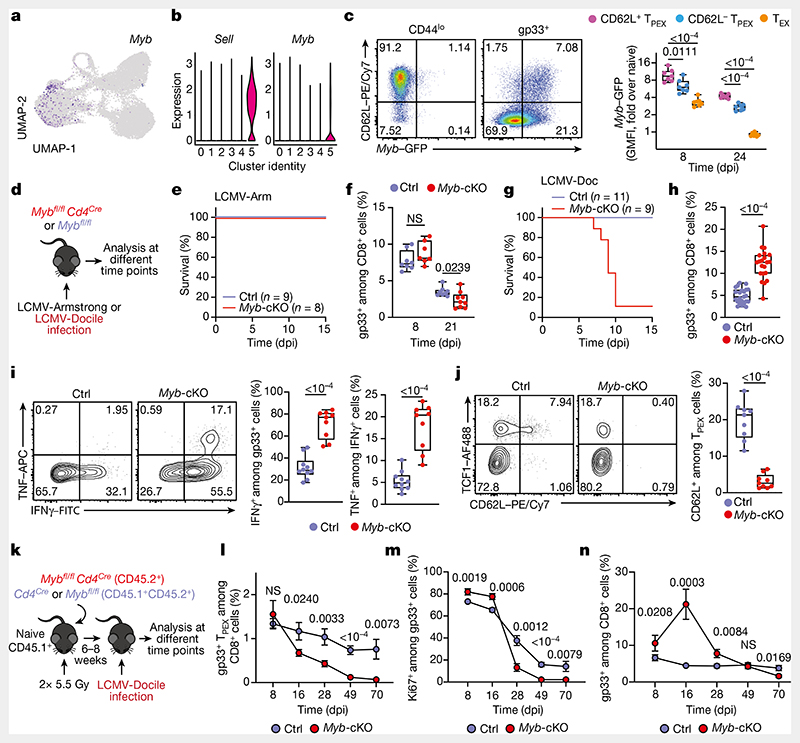
Functional annotation of our scRNA-seq data identified Myb, encoding the transcription factor MYB (also called c-Myb), as specifically enriched among CD62L+ TPEX cells (Fig. 2a,b and Supplementary Table 1). MYB has important roles in the self-renewal of haematopoietic stem cells and cancer cells30, T cell leukaemia31 and CD8+ memory T cells32,33. To characterize the dynamics of Myb expression in chronic infection, we infected MybGFP reporter mice34 with LCMV-Docile (Fig. 2c and Extended Data Fig. 5k), and found that the expression of Myb was highest in CD62L+ TPEX cells (Fig. 2c and Extended Data Fig. 5k). Myb expression in CD8+ T cells responding to LCMV-Docile infection was significantly higher than in those responding to LCMV-Armstrong infection (Extended Data Fig. 5l), and was further enhanced by the inhibition of PD-1 signalling in vivo (Extended Data Fig. 5m–o). Moreover, in vitro TCR stimulation induced the expression of Myb in a dose-dependent manner (Extended Data Fig. 5p). Finally, the proportions of CD62L+ antigen-specific CD8+ T cells were 10-fold higher in LCMV-Docile versus LCMV-Armstrong infection (Extended Data Fig. 5q,r). Together, these data indicate that strong and persistent TCR stimulation favours the sustained expression of MYB and retention of CD62L+ TPEX cells during chronic infection.
Fig. 2. The transcription factor MYB is required for the generation of CD62L+ TPEX cells and the functional exhaustion ofT cells during chronic infection.
a, Normalized gene expression of Myb projected onto the UMAP plot. b, Violin plots showing normalized expression of Sell and Myb. c, MybGFP reporter mice were infected with LCMV-Docile and splenic CD8+ T cells were analysed at the indicated time points after infection. Left, representative flow cytometry plots showing the expression of CD62L and Myb–GFP among naive (CD44lo) and gp33+CD8+ T cells. Right, quantification showing the geometric mean fluorescence intensity (GMFI) of Myb–GFP among CD62L+ TPEX, CD62L− TPEX and TEX cells as fold change over naive CD8+ T cells. d–j, Mybfl/flCd4Cre (Myb-cKO) and littermate Mybfl/fl control (Ctrl) mice were infected with either LCMV-Armstrong (LCMV-Arm) or LCMV-Docile (LCMV-Doc). d, Schematic of the experimental set-up. e–h, Survival curves of Myb-cKO and control mice and box plots showing the frequencies of gp33+CD8+ T cells at the indicated time points after infection with LCMV-Armstrong (e,f) or LCMV-Docile (g,h). i,j, Flow cytometry plots and quantification showing the expression of IFNγ and TNF after gp33 peptide restimulation (i) and the frequencies of CD62L+ TPEX cells (j). Cells were gated on gp33+cells; 8dpi. k–n, Mixed bone marrow chimeric mice containing Myb-cKO and Cd4Cre control T cells were infected with LCMV-Docile and analysed at the indicated time points. k, Schematic of the experimental set-up. l–n, Quantifications show the frequencies of gp33+ TPEX cells (l), Ki67+ cells (m) and gp33+ cells (n). Dots represent individual mice; symbols and error bars represent mean and s.e.m., respectively; box plots indicate minimum and maximum values (whiskers), interquartile range (box limits) and median (centre line). Data are representative of all analysed mice (e,g), two (c,f,i,j,l–n) or three independent experiments (h). P values are from two-tailed unpaired t-tests (c,f,h–j) and Mann–Whitney tests (l–n).
To study the role of MYB in CD8+ T cells during viral infection, we infected Mybfl/flCd4Cre mice35 (which lack MYB specifically in T cells) and Mybfl/fl (control) littermates with LCMV-Docile or LCMV-Armstrong (Fig. 2d). Before infection, Mybfl/flCd4Cre mice showed no major abnormalities in the thymic and mature CD8+ T cell compartments (Extended Data Fig. 6). LCMV-Armstrong-infected Mybfl/flCd4Cre mice mounted CD8+ T cell responses that were similar to those of controls, and showed no overt signs of disease (Fig. 2e,f and Extended Data Fig. 7a–d). By contrast, LCMV-Docile-infected Mybfl/flCd4Cre but not control mice exhibited signs of severe immunopathology and most became moribund within 10 dpi (Fig. 2g and Extended Data Fig. 7e–i). Depletion of CD8+ T cells averted these symptoms and protected LCMV-Docile-infected Mybfl/fl Cd4Cre mice (Extended Data Fig. 7j,k), indicating that MYB-deficient CD8+ T cells mediated the fatal immunopathology in chronic LCMV infection. In line with these findings, splenic gp33+CD8+ T cells accumulated at increased frequencies in Mybfl/flCd4Cre mice at 8 dpi (Fig. 2h). MYB-deficient TPEX and TEX cells expressed significantly higher levels of IFNγ and TNF, whereas TEX cells also expressed more granzyme B and underwent increased proliferation (measured by the expression of Ki67) compared to controls (Fig. 2i and Extended Data Fig. 7l-o), despite the viral titres being similar in both groups of mice (Extended Data Fig. 7p). MYB-deficient gp33+CD8+ T cells showed increased expression of the inhibitory receptors PD-1 and TIM-3 compared to control cells (Extended Data Fig. 7q), which suggests that the increased function and proliferation of effector cells was not due to impaired expression of inhibitory receptors. Notably, CD62L+ TPEX cells were specifically lost in the absence of MYB (Fig. 2j and Extended Data Fig. 7r,s).
To longitudinally examine the cell-intrinsic role of MYB in the absence of potentially confounding immune pathology, we generated mixed bone marrow chimeric mice that contained small numbers of Mybfl/flCd4Cre (10–20%) and congenically marked Cd4Cre control CD8+ T cells and infected them with LCMV-Docile (Fig. 2k–n and Extended Data Fig. 8). Consistent with our observations in non-chimeric mice, MYB-deficient antigen-specific CD8+ T cells proliferated more and exhibited increased expression of effector molecules compared to controls (Extended Data Fig. 8a–e). Similarly, the MYB-deficient CD8+ T cell compartment was devoid of CD62L+ TPEX cells (Extended Data Fig. 8f,g). Although MYB-deficient TCF1+ TPEX cells initially developed, they were poorly maintained (Fig. 2l and Extended Data Fig. 8h–j). This observation concurred with a premature termination of cell-cycle activity in MYB-deficient TPEX and TEX cells and a marked contraction of the entire antigen-specific compartment (Fig. 2m,n and Extended Data Fig. 8k–n). Similar results were obtained from adoptively transferred MYB-deficient and control P14 T cells (Extended Data Fig. 9a–g). Thus, MYB mediates the development of CD62L+ TPEX cells and functional exhaustion of CD8+ T cells during the acute phase, sustains long-term proliferative capacity and prevents the attrition of antigen-specific T cells during the chronic phase of infection.
MYB orchestrates exhausted T cell transcription
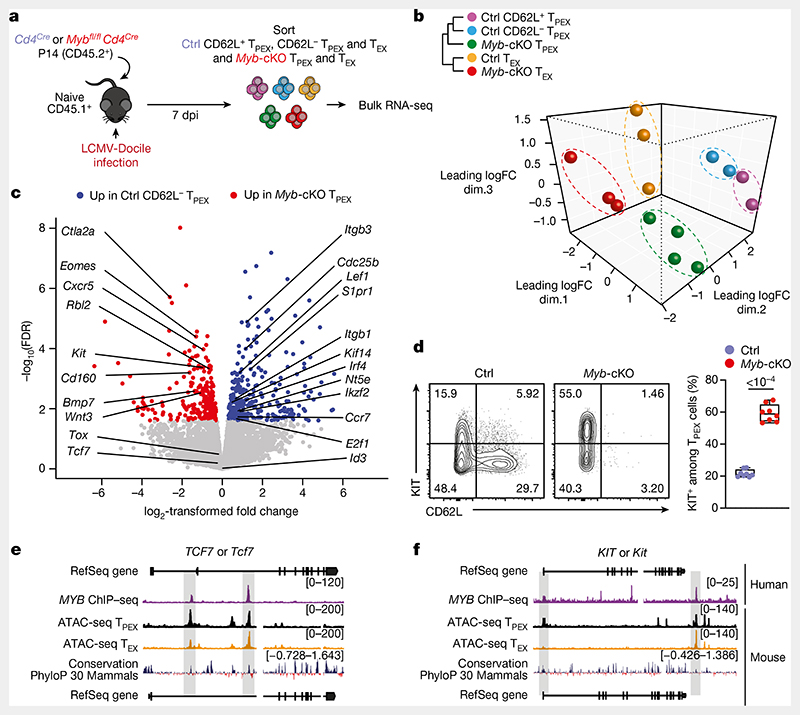
We next sorted MYB-deficient and control P14 TPEX cells from LCMV-Docile-infected mice and performed transcriptional profiling by RNA-seq (Extended Data Fig. 10a). The analysis showed that there was a loss of the CD62L+ TPEX cell signature in MYB-deficient compared with control TPEX cells (Extended Data Fig. 10b), confirming that MYB deficiency resulted in the loss of CD62L+ TPEX cells and not merely CD62L expression. We next performed RNA-seq of control and MYB-deficient P14 TEX cells and TPEX cells sorted for differential expression of CD62L (Fig. 3a). The analysis revealed transcriptional divergence between all subsets and identified 584 differentially expressed genes (P < 0.05) between control CD62L+ and CD62L− TPEX cells (Fig. 3b, Extended Data Fig. 10c and Supplementary Table 2). CD62L+ TPEX cells expressed higher levels of transcripts that encode molecules related to lymph node homing (for example, Sell, Ccr7 and S1pr1), and higher levels of the cell-cycle inhibitors Cdkn1b and Cdkn2d and the quiescence factors Klf2 and Klf3, compared with CD62L− TPEX cells. Genes that were upregulated in CD62L− TPEX cells included those that encode positive cell-cycle regulators (E2f1, Cdc6, Skp2, Cdc25a and Kif14), metabolic enzymes (P2rx7, Hk2, Pfkm, Pkm and Gpd2) and nutrient transporters (Slc7a5, Slc19a2 and Slc25a10) (Extended Data Fig. 10c and Supplementary Table 2). A comparison of MYB-deficient and control CD62L− TPEX cells identified 580 differentially expressed genes (Supplementary Table 2), including genes that encode molecules related to T cell exhaustion and TPEX cell identity (Lef1, Eomes, Ctla2a, Irf4, Ikzf2, Nt5e and Cd160), cell-cycle regulation and stem cell renewal (E2f1, Rbl2, Kif14, Cdc25b, Bmp7 and Wnt3) (Fig. 3c). Consistent with the impaired expression of transcripts related to cell migration and lymph node homing (Ccr7, Cxcr5, S1pr1, Itgb1 and Itgb3), MYB-deficient antigen-specific CD8+ T cells were largely excluded from the lymph nodes (Extended Data Fig. 10d,e). We also observed increased expression of Kit–encoding KIT, which is involved in haematopoiesis and T cell activation36,37–in MYB-deficient versus wild-type TPEX cells (Fig. 3c). Indeed, KIT was exclusively expressed in CD62L− TPEX cells and was highly upregulated in MYB-deficient TPEX cells (Fig. 3d and Extended Data Fig. 10f,g). A comparison of MYB-deficient and control TEX cells revealed further significant transcriptional changes (1,532 differentially expressed genes), including the upregulation of transcripts that encode cytotoxic molecules(Gzma, Gzmc and Gzme) or that are related to terminally exhausted TEX cells (Cd7, Cd244a, Cd160, Entpd1, Id2 and Cd101), and the downregulation of transcripts related to CX3CR1+ TEX cells, which have been shown to be more effective in controlling viral burden compared to their CX3CR1- counterparts20,21(Cx3cr1, Zeb2, Klf2 and S1pr1) (Extended Data Fig. 10h–j and Supplementary Table 2). Flow cytometric analysis revealed a lack of CX3CR1+ cells and an increase in terminally exhausted CD101+ cells among MYB-deficient TEX cells compared to controls (Extended Data Fig. 10k–l). Consistent with accelerated differentiation into terminally differentiated cells, TPEX-cell-related transcripts, including Tcf7, Slamf6, Lef1 and Xcl1, were more strongly downregulated in MYB-deficient than in control TEX cells (Extended Data Fig. 10h,j). Many of the genes that were dysregulated in the absence of MYB, including Tcf7, Kit, Slamf6, Lef1, Klf2, S1pr1, Icos, E2f1, Gzma, Gzmc and Myb itself, contained MYB-binding regions in human T cells38 (Supplementary Table 3), which were conserved and aligned with open chromatin regions in mouse exhausted T cells13 (Fig. 3e,f, Extended Data Fig. 11a–d and Supplementary Table 3). Together, our results show that MYB is a central transcriptional orchestrator of T cell exhaustion that mediates the development of CD62L+ TPEX cells and restrains the terminal differentiation of exhausted T cells.
Fig. 3. MYB regulates the expression of genes that are critical for the function and maintenance of exhausted T cells.
a–c, Congenically marked Mybfl/flCd4Cre (Myb-cKO) and Cd4Cre (control) P14 T cells were adoptively transferred into naive recipient mice, which were then infected with LCMV-Docile. Splenic P14 subsets were sorted at 7 dpi and processed for bulk RNA-seq. a, Schematic of the experimental set-up. b, Sample dendrogram and three-dimensional scaling plot of all the samples. logFC, log-transformed fold change. c, Volcano plot highlighting genes that are differentially expressed (false discovery rate (FDR)< 0.15) between Myb-cKO TPEX and control CD62L- TPEX cells, with genes of interest annotated. d, Flow cytometry plots and quantification show the frequencies of KIT+ cells among control and Myb-cKO TPEX P14 T cells at day 8 after infection with LCMV-Docile (gated on TPEX cells). e,f, Tracks show MYB chromatin immunoprecipitation followed by sequencing (ChIP–seq) peaks in the TCF7 (e) and KIT (f) gene loci of humanJurkat T cells and assay for transposase-accessible chromatin using sequencing (ATAC-seq) peaks of TPEX and TEX cells in the corresponding mouse gene loci aligned according to sequence conservation. Dots in graph represent individual mice; box plots indicate minimum and maximum values (whiskers), interquartile range (box limits) and median (centre line). Data are representative of two independent experiments (d). P values are from two-tailed unpaired t-tests (d).
CD62L+ TPEX cells fuel therapeutic reinvigoration
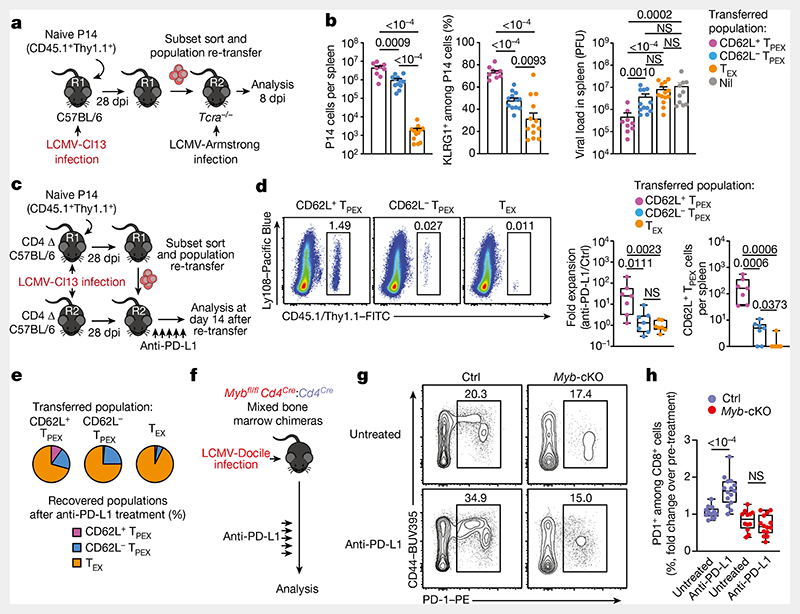
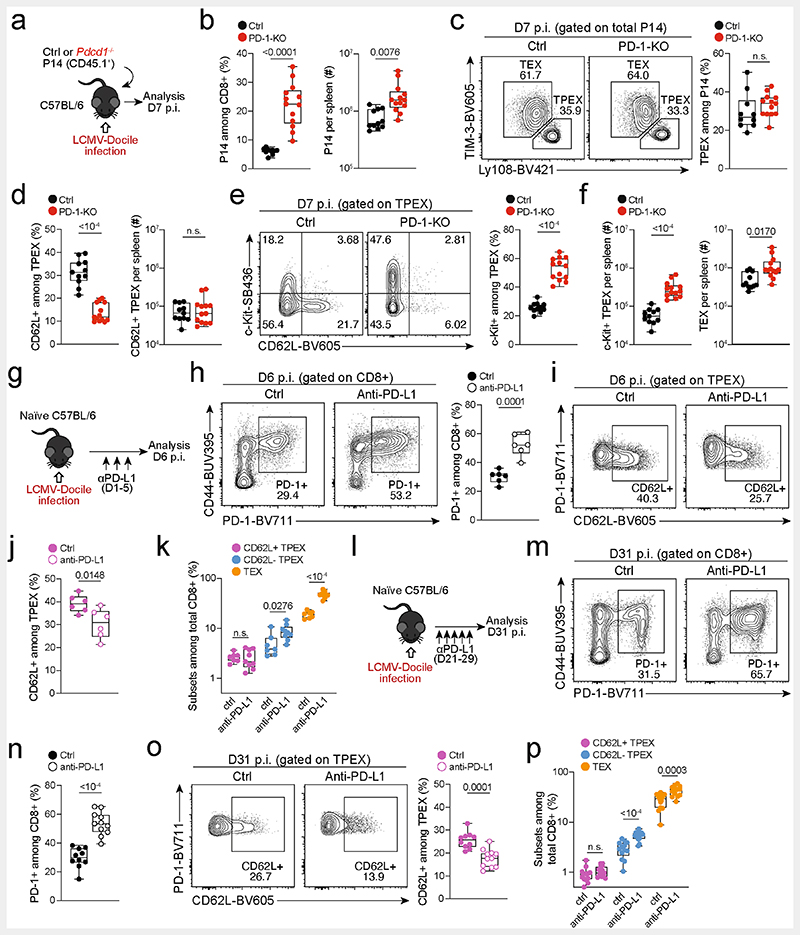
To test the functional potential of MYB-dependent CD62L+ TPEX cells, we separately transferred CD62L+ and CD62L− TPEX cells as well as TEX cells into either wild-type (Extended Data Fig. 11e–g) or T-cell-deficient Tcra−/− mice (Fig. 4a,b), which were then infected with LCMV-Armstrong. Although all subsets maintained high expression of PD-1 (Extended Data Fig. 11g), progeny that were derived from CD62L+ TPEX cells expanded more efficiently (Fig. 4b and Extended Data Fig. 11f,g), contained more KLRG1+ effector cells and provided significantly enhanced viral control compared to the other exhausted T cell subsets (Fig. 4b). CD62L+ TPEX cells also gave rise to more CX3CR1+ TEX cells (Extended Data Fig. 11h,i), altogether indicating that they have a superior potential to generate functional effector cells as compared to their CD62L− counterparts. We next tested the role of PD-1 and therapeutic PD-1 checkpoint blockade in the generation and function of CD62L+ TPEX cells. To this end, we generated P14 T cells that lack functional Pdcd1 (encoding PD-1) using CRISPR–Cas9 (Extended Data Fig. 12a–f). Similar to previous studies39–41, PD-1-deficient P14 T cells exhibited increased clonal expansion in response to LCMV-Docile, as compared with control cells (Extended Data Fig. 12b). Although the frequencies of TPEX and TEX cells were unaffected by the loss of PD-1 (Extended Data Fig. 12c), the frequencies–but not the numbers–of CD62L+ TPEX cells were markedly decreased compared to control P14 T cells (Extended Data Fig. 12d). This was due to a concurrent increase in the proportions and the absolute numbers of KIT+ TPEX cells and TEX cells (Extended Data Fig. 12e,f). These results indicate that PD-1 signalling does not affect the development or maintenance of CD62L+ TPEX cells but limits their differentiation into CD62L− TPEX and TEX cells. In line with this conclusion, the numbers of CD62L+ TPEX cells remained stable during PD-1 checkpoint inhibition, whereas the overall population of antigen-responsive PD-1+CD8+ T cells expanded robustly (Extended Data Fig. 12g–p). To directly test the role of CD62L+ TPEX cells in checkpoint blockade, we performed adoptive transfer experiments in the context of therapeutic PD-1 inhibition (Fig. 4c–e). Re-transferred CD62L+ TPEX cells proliferated strongly in response to PD-1 checkpoint inhibition and generated larger progenies compared with untreated controls, while undergoing concurrent self-renewal (Fig. 4d,e). In stark contrast, both CD62L− TPEX and TEX cells showed no apparent proliferative response (Fig. 4d,e), which indicates that CD62L+ but not CD62L− TPEX cells fuel the generation of effector cells in response to checkpoint blockade. Consistent with this conclusion, MYB-deficient antigen-responsive PD-1+CD8+ T cells, which lack CD62L+ TPEX cells, did not expand in response to PD-1 checkpoint inhibition (Fig. 4f–h). Together, our results reveal that MYB-dependent CD62L+ TPEX cells exclusively fuel the proliferative burst in response to PD-1 checkpoint inhibition and therefore dictate the success of therapeutic checkpoint blockade.
Fig. 4. CD62L+ TPEX cells show enhanced potential for effector cell generation and selectively mediate responsiveness to PD-1 checkpoint blocking therapy.
a,b, Congenically marked naive P14 T cells were transferred into primary recipient (R1) mice, which were subsequently infected with LCMV-Cl13. Exhausted T cell subsets were sorted at 28 dpi and 1.0 × 104–2.5 × 104 cells or no cells (Nil) were re-transferred into secondary Tcra-/- recipient (R2) mice. Splenic P14 T cells of R2 mice were analysed 8 days after infection with LCMV-Armstrong. a, Schematic of the experimental set-up. b, Numbers of recovered P14 T cells (left), percentages of KLRG1+ (middle) and splenic viral loads (right). PFU, plaque-forming units. c-e, Congenically marked naive P14 T cells were transferred into CD4-depleted R1 mice, which were subsequently infected with LCMV-Cl13. Exhausted T cell subsets were sorted at 28 dpi and re-transferred to infection-matched CD4-depleted (CD4 Δ) R2 mice, treated with anti-PD-L1 antibodies or phosphate-buffered saline (PBS) on days 1, 4, 7, 10 and 13 and analysed at day 14 after re-transfer. c, Schematic of the experimental set-up. d, Representative flow cytometry plots of splenic progeny derived from transferred T cell subsets after treatment with anti-PD-L1, at day 14 after re-transfer (cells were gated on CD4−CD19−PD-1+ cells). Box plots show the relative progeny expansion in anti-PD-L1-treated versus PBS-treated mice (left) and the numbers of CD62L+ TPEX cells among progeny after anti-PD-L1 treatment (right). e, Average subset distribution. f–h, Mixed bone marrow chimeric mice containing congenically marked Myb-cKO and Cd4Cre (control) T cells, infected with LCMV-Docile, were treated with anti-PD-L1 on days 33, 36, 39, 42 and 45 and analysed at 49 dpi. f, Schematic of the experimental set-up. g,h, Representative flow cytometry plots (g) and box plot (h) showing the fold change of frequencies of splenic polyclonal PD1+CD8+ T cells in anti-PD-L1-treated versus PBS-treated mice. Cells were gated on CD8+ cells; 49 dpi. Dots in graphs represent individual mice; box plots indicate minimum and maximum values (whiskers), interquartile range (box limits) and median (centre line); horizontal lines and error bars of bar graphs indicate mean and s.e.m., respectively. Data are representative of at least two independent experiments (b,d–e,g–h). P values are from two-tailed unpaired t-tests (b (middle), h) and Mann–Whitney tests (b (left, right, d).
Overall, our data show that the CD8+ T cell response in chronic infection is maintained by a small population of distinct TPEX cells that co-express TCF1, CD62L and the transcription factor MYB. These cells, which we term stem-like exhausted T (TSLEX) cells here, possess superior self-renewal, multipotency and long-term proliferative capacity compared to their TCF1+ but CD62L− descendants. Loss of MYB abrogated the differentiation of TSLEX cells and severely impaired the the persistence of the entire TCF1+ TPEX cell compartment, ultimately resulting in the collapse of the complete CD8+ T cell response. MYB also mediates functional exhaustion during chronic infection by restricting the initial expansion and effector function of antigen-responsive CD8+ effector T cells. As a result, mice that lacked MYB in their T cells succumbed to chronic but not acute viral infection, highlighting that T cell exhaustion is an essential adaptation to chronic infection. Thus, MYB represents a transcriptional checkpoint that instructs the differentiation and function of CD8+ T cells in response to severe or chronic infection. Our data also show that TSLEX cells are exclusively required to mediate the response to PD-1 checkpoint inhibition. These findings not only advance our understanding of the mechanisms of T cell re-invigoration in the context of checkpoint inhibition, but also emphasize the need for new therapeutic strategies that target TSLEX cells to harness the full potential of T cell-mediated immunotherapy. Furthermore, the superior proliferative and developmental potential of TSLEX cells makes them prime targets of adoptive T cell transfer and chimeric antigen receptor (CAR) T cell therapies. Finally, our results show that two central but seemingly unrelated properties of exhausted T cells–limited function and longevity–are intimately linked by a single transcription factor MYB; this is a notable example of evolutionary parsimony, which ensures ongoing T cell immunity during chronic infection while preventing collateral damage to the host.
Methods
Mice and generation of mixed bone marrow chimeric mice
All mice used in this study were on a C57BL/6J background. Age- and sex-matched mice were used for experiments and allocated to experimental groups without further randomization or blinding. CD45.1 or CD45.2 mice were obtained from the Australian Resources Centre or were purchased from Envigo at 6–8 weeks of age. Id3GFP mice42 expressing the P14 TCR transgene (JAX: Tg(TcrLCMV)327Sdz) were used in some experiments as described13. MybGFP mice and Mybfl/flCd4Cre mice were described previously34. Mybfl/flCd4Cre mice were crossed to include the P14 TCR transgene for some experiments. Littermate Mybfl/fl mice were used as controls. Mixed bone marrow chimeric mice were generated by irradiating CD45.1 host mice (2× 5.5 Gy), before reconstitution with a mix of CD45.1/CD45.2 Cd4Cre bone marrow and CD45.2 Mybfl/flCd4Cre bone marrow. Mice were left to recover for six to eight weeks before further experiments. P14 transgenic Tcf7GFP mice on a CD45.1 background were generated in the laboratory of D.Z. by inserting a GFP expression construct into the Tcf7 gene locus and will be described in detail elsewhere. P14 mice expressing diverse combinations of the congenic markers CD45.1/.2 and Thy1.1/1.2, as well as Tcra−/− mice were bred under specific-pathogen-free conditions at the mouse facility of the Institute for Medical Microbiology, Immunology and Hygiene at the Technical University of Munich. All mice were maintained and used in accordance with the guide-lines of the University of Melbourne Animal Ethics Committee or the district government of upper Bavaria (Department 5–Environment, Health and Consumer Protection).
Generation of colour-barcoded P14 cells
Retrogenic colour-barcoding was used to heritably label individual P14 cells and their progeny for in vivo single-cell fate-mapping experiments, as previously described24,25. In brief, bone marrow was collected from congenically marked (CD45.1+ or CD90.1+) P14 donor mice and stained with Ly6A/E (Sca-1), anti-mouse CD3 and CD19 antibodies, together with propidium iodide for live or dead discrimination. Haematopoietic stem cells (HSCs) were then sorted as live CD3−CD19−Sca-1+ cells and cultured at 37 °C in cDMEM (DMEM (Life Technologies), supplemented with 10% FCS, 0.025% l-glutamine, 0.1% HEPES, 0.1% gentamycin and 1% penicillin/streptomycin), supplemented with 20 ng ml−1 mouse IL-3, 50 ng ml−1 mouse IL-6 and 50 ng ml−1 mouse SCF, for three to four days in tissue-culture-treated 48-well-plates. Expanded stem cells were then retrovirally transduced with constructs encoding the fluorescent proteins GFP, YFP, BFP, CFP and T-Sapphire by spinocula-tion. After two days in culture, the transduced HSCs were suspended in fetal calf serum (FCS) and injected intravenously into irradiated C57BL/6 recipient mice (2 × 4.5 Gy, with a resting period of 4 h). After several weeks, colour-barcoded naive (CD8+CD44low) P14 cells were sorted from the peripheral blood of retrogenic mice and transferred into C57BL/6 recipients.
Organ preparation and adoptive T cell transfer
Single-cell suspensions were obtained by mashing total spleens, lymph nodes or bone marrow through a 70-μm nylon cell strainer (BD). For liver samples, lymphocytes were obtained by density gradient centrifugation. Red blood cells were lysed with a hypotonic ammonium chloride-potassium bicarbonate (ACK) or ammonium chloride-Tris (ACT) buffer. For isolating naive CD8+ or transgenic P14 T cells, the mouse CD8+ T cell enrichment kit (Miltenyi Biotech) was used, or cells were sorted as live CD8+CD44low cells.
For primary population transfer experiments, 2,000–10,000 naive P14 T cells were injected into naive congenically marked primary recipients. For adoptive re-transfer experiments, P14 cells were first enriched from the spleens and lymph nodes of primary or secondary recipients by sorting CD45.1+Thy1.1+ cells, followed by staining with anti-mouse CD62L, anti-mouse Ly108 and the eBioscience Fixable Viability Dye eFluor 780 or propidium iodide for live or dead discrimination. The indicated subsets were then sorted according to their expression profile of CD62L and Ly108 (note: the anti-mouse CD62L antibody was titrated to a dilution that precludes functional blocking of the molecule). Unless specified otherwise, equal numbers of cells of each subset were injected, ranging between 3,000 and 40,000 for secondary transfers and between 1,000 and 3,000 for tertiary transfers. In cases in which the numbers of transferred cells differed between experimental groups (Extended Data Fig. 3), a fold expansion factor was calculated by dividing the number of recovered P14 cells by the number of transferred cells. A 10% take rate was assumed for these calculations, based on our measurements in Extended Data Fig. 4f.
For primary single-cell transfer experiments, naive P14 cells were isolated from the peripheral blood or spleens of naive retrogenic P14 donor mice by staining with anti-mouse CD8, anti-mouse CD44, anti-mouse CD45.1 and anti-mouse Thy1.1. For secondary single-cell re-transfers, anti-mouse CD45.1, anti-mouse CD62L and anti-mouse Ly108 were used, together with the eBioscience Fixable Viability Dye eFluor 780 or propidium iodide for live or dead discrimination. Single P14 cells were then isolated by successively sorting individual cells according to their unique congenic or retrogenic colour barcode and their CD62L/Ly108 phenotype into a 96-well V-bottom plate containing a pellet of 4 × 105 C57BL/6 splenocytes. The unique congenic and retrogenic colour barcodes of sorted cells enabled the simultaneous transfers of multiple individual cells for fate-mapping. After sorting, the whole content of each well was injected into separate C57BL/6 recipients.
Gene deletion by CRISPR –Cas9–sgRNA complex electroporation
Pdcd1 gene deletion was conducted as reported previously41. In brief, P14 cells were purified using an EasySep mouse CD8+ T cell isolation kit (STEMCELL Technologies) according to the manufacturer’s instructions, after which cells were electroporated (Pulse DN100) with a complex of Alt-R S.p. Cas9 Nuclease (Integrated DNA Technologies) and a previously described Pdcd1-targeting sgRNA (Synthego)41 using the P3 primary cell 4D-Nucleofector X kit S electroporation kit (Lonza) and Lonza 4D-Nucleofector Core Unit (Lonza). Cells were rested in fully supplemented RPMI medium (see above) at 37°C for 10 min, after which P14 cells were counted, and 5,000 P14 cells were injected intravenously into recipient mice before infection with LCMV.
LCMV infections and checkpoint blockade
LCMV-Docile, LCMV-Cl13 and LCMV-Armstrong were propagated and quantified as previously described26. For LCMV-Docile and LCMV-Cl13 infection, frozen stocks were diluted in PBS and 2 × 106 PFU were injected intravenously. For LCMV-Armstrong infection, frozen stocks were diluted in PBS and 2 × 105 PFU were injected intraperitoneally. For infection of Tcra−/− mice, a dosage of 2 × 103 PFU was used. For CD4+ T cell depletion, mice were injected twice intraperitoneally with 200 μg per mouse of anti-CD4 monoclonal antibody (GK1.5, BioXCell) one day before and one day after infection with LCMV-Cl13. For CD8+ T cell depletion, mice were injected intraperitoneally with 100 μg per mouse of anti-CD8 monoclonal antibody (YTS-169, BioXCell) on days 1, 3 and 5 of infection. For PD-1 blockade, monoclonal anti-PD-L1 antibodies (B7-H1, BioXCell) were injected intraperitoneally at 200 μg per mouse at the specified days after infection.
In vitro culture of naive CD8+ T cells
Cell-culture 48-well or 96-well plates were prepared by coating with anti-CD3 at various concentrations for at least 2 h at 4 °C. Control wells were coated with PBS for the same duration. The wells were washed twice using PBS. Enriched naive CD8+ T cells were seeded in the wells and were cultured in RPMI medium supplemented with 10% FCS, 55 μM β-mercaptoethanol, 2 mM Glutamax, 25 mM HEPES buffer and 100 U ml−1 penicillin and 10 μg ml−1 streptomycin for three days in a humidified incubator at 37°C with 5% CO2.
Surface and intracellular antibody staining of mouse cells
Surface staining was performed for 30 min at 4°C in PBS supplemented with 2% FCS (FACS buffer) with the following antibodies: CD8a (53-6.7, BD), CD44 (IM7, BD), CD45.1 (A20, BD or Biolegend), CD45.2 (104, BD), CD90.1 (HIS52, Thermo Fisher Scientific) CX3CR1 (SA011F11, Biolegend), PD-1 (RMP1-30 or 29F.1A12, Biolegend), CD62L (MEL-14, Biolegend), TIM-3 (RMT3-23, Biolegend), CD101 (Moushi101, Thermo Fisher Scientific), Ly108 (eBio13G3-18D, BD), CD117 (KIT) (ACK2, Thermo Fisher Scientific), CD244 (2B4) (eBio244F4, Thermo Fisher Scientific), CD160 (eBioCNX46-3, eBioscience), TIGIT (GIGD7, Thermo Fisher Scientific) and KLRG1 (2F1, Biolegend). LCMV-derived Db/gp33-41 tetramers were obtained from the NIH Tetramer Facility; tetramer staining was performed for 30–60 min at 4 °C in FACS buffer. Each cell staining reaction was preceded by a 10-min incubation with purified anti-mouse CD16/32 Ab (FcgRII/III block; 2.4G2) and (fixable) viability dye (Thermo Fisher Scientific). For intracellular cytokine staining, splenocytes were ex vivo restimulated with gp33-41 (gp33) peptide (5 mM) for 5 h in the presence of brefeldin A (Sigma) for the last 4.5 h, fixed and permeabilized using the Cytofix/Cytoperm (BD) or transcription factor staining kit (eBioscience) and stained with anti-IFNγ (XMG1.2, Thermo Fisher Scientific), TNF (MP6-XT22, Thermo Fisher Scientific). Other intracellular staining was performed with the Foxp3 transcription factor staining kit (eBioscience) and the following antibodies: TCF1 (C63D9, Cell Signaling), GZMB (MHGB04, Thermo Fisher Scientific) and Ki67 (FM264G, BD).
In vitro activation of T cells
CD8+ T cells were isolated using the CD8+ T cell enrichment kit (Miltenyi Biotech) and, in some instances, CTV labelled. Wild-type cells were stimulated with plate-bound anti-CD3 at the indicated concentration and in fully supplemented tissue-culture medium (RPMI plus 10% FCS, 2 mM Glutamax, 1 mM pyruvate, 55 μM mercaptoethanol, 100 U ml−1 penicillin, 10 μg ml−1 streptomycin) and 100 U ml−1 IL-2.
Histology
For immunofluorescence, spleens were embedded and frozen in OCT, sectioned at 15 μm and mounted on SuperFrostPlus Adhesion glass (Thermo Fisher Scientific). Sections were dehydrated using silica beads, fixed with 4% paraformaldehyde for 10 min and washed with PBS. Samples were blocked using 5% normal goat serum for 2 h before staining. Samples were incubated with antibodies against B220 (RA3-6B2, eBioscience), CD3 (17A2, eBioscience) and F4/80 (BM8, Biolegend) diluted in 5% NGS for 2 h at room temperature in the dark. After staining, samples were washed with PBS at least three times. Samples were then mounted using ProLong Gold Antifade Mountant (Invitrogen) and imaged using an inverted LSM780 microscope (Carl Zeiss) and a plan apochromat 63× NA 1.40 oil-immersion objective (Carl Zeiss). For haematoxylin and eosin (H&E) staining, organs were collected and fixed in 10% formalin. Fixed samples were embedded in paraffin and sectioned at 10 μm, mounted on SuperFrostPlus Adhesion glass and stained using H&E. Mounted samples were imaged using a Nikon SMZ1270 Stereo Microscope. Imaging data were analysed using Fiji (ImageJ) software (NIH).
scRNA-seq and analysis
Relating to the dataset introduced in Fig. 1: TPEX-cell-enriched CD8+ T cells were sorted as CD8+PD-1+TIM-3low from the spleens of chronically infected mice (LCMV-Cl13) using a FACSAria III (BD Biosciences). Afterwards, the single cells were encapsulated into droplets with the ChromiumTM Controller (10X Genomics) and processed following the manufacturer’s specifications. Bead captured transcripts in all encapsulated cells were uniquely barcoded using a combination of a 16-bp 10X barcode and a 10-bp unique molecular identifier (UMI). The Chromium Single Cell 3’ Library & Gel Bead Kit v2 for the wild-type untreated sample or v3 for wild type treated with 200 μg of anti-PD-L1 antibody (10F.9G2, BioXCell) for 24 h were used to generate cDNA libraries (10X Genomics) following the protocol provided by the manufacturer. Libraries were quantified by QubitTM 3.0 Fluometer (Thermo Fisher Scientific) and quality was checked using 2100 Bioanalyzer with High Sensitivity DNA kit (Agilent). For library sequencing the NovaSeq 6000 platform (S1 Cartridge, Illumina) in 50-bp paired-end mode was used. The sequencing data were demultiplexed using Cell-Ranger software (v.2.0.2) and the reads were aligned to the mouse mm10 reference genome using STAR aligner. Aligned reads were used to quantify the expression level of mouse genes and generate the gene-barcode matrix. Subsequent data analysis was performed using Seurat R package (v.3.2)43. The sequencing data are available at the National Center for Biotechnology Information (NCBI) Gene Expression Omnibus (GEO) (http://www.ncbi.nlm.nih.gov/geo) under the accession number GSE168282 (ref. 19). For further analysis in this study, datasets GSM5135522 and GSM5135523 (ref. 19) were combined with another publicly available scRNA-seq dataset of mouse exhausted CD8+ T cells, accessed from GSE122712 (ref. 11) and analysed using Seurat R package (v.3.2)43. The 2,000 most variable genes were included for the anchoring process and used for downstream analysis to calculate principal components of log-normalized and scaled expression data. On the basis of the principal component analysis (PCA), a UMAP of the identified clusters was visualized. Cluster-specific genes were identified with the FindAllMarker function in Seurat with parameters min. pct = 0.25, logfc.threshold = 0.25. Trajectories were predicted using the Slingshot 1.4.0 package44, using the function slingshot with default settings and starting with the CD62L+ TPEX cell cluster. The functional annotation tool DAVID (LHRI) was used to interrogate gene sets to identify transcription factors of interest. Selected lists of genes were then further explored using enrichment analyses against existing RNA-seq datasets13,20.
For the dataset used in Extended Data Fig. 2h-j, TPEX cells were sorted as live CD8+PD1+CD45.1+Tcf7–GFP+ cells from the spleens of chronically infected mice (LCMV-Cl13, 28 dpi) using a MoFlo Astrios cell sorter (Beckman Coulter) and processed using the 10X Genomics technology, according to the manufacturer’s protocol (Chromium Single Cell 3’ GEM v3 kit). Quality control was performed with a High Sensitivity DNA Kit (Agilent 5067-4626) on a Bioanalyzer 2100, as recommended in the protocol. Libraries were quantified with the Qubit dsDNA HS Assay Kit (Life Technologies Q32851). All steps were performed using RPT filter tips (Starlab) and LoBind tubes (Sigma). The library was sequenced with 20,000 reads per cell. Illumina paired-end sequencing was performed with 150 cycles on a Novaseq 6000. Annotation of the sequencing data was performed using CellRanger software (v.5.0.0) against the mouse reference genome GRCm38 (mm10-2020-A). All subsequent analysis was performed using SCANPY (v.1.6)45. After general pre-processing (less than 15% mitochondrial genes, regressing out cell cycle, filtering mitochondrial genes and total counts), the data were count-normalized per cell and logarithmized. RNA velocities were calculated using Velo-cyto46 and analysed with scVelo47. The sequencing data are available at the NCBI GEO (http://www.ncbi.nlm.nih.gov/geo) under the accession number GSE205608.
Bulk RNA extraction, sequencing and analysis
Relating to the dataset in Extended Data Fig. 2f,g, the indicated subsets of exhausted P14 cells (CD62L+ TPEX, CD62L− TPEX, TEX) were sorted from the spleen of chronically infected mice (LCMV-Cl13) at 28 dpi. As a comparison, memory P14 cells were sorted from the spleen of LCMV-Armstrong-infected mice at 28 dpi according to the following phenotypes: CD62L+Ly108+ (CD62L+ memory), CD62L−Ly108+ (CD62L− memory) and CD62L−Ly108− (effector). In addition, naive P14 cells were included for the analysis. RNA extraction from sorted P14 T cells was performed using the RNeasy Plus Mini Kit (Qiagen) according to the manufacturer’s instructions. Each sample group consisted of two to three biological replicates. Sequencing was performed on an Illumina Novaseq by Novogene, generating 150-bp paired-end reads. RNA-seq reads were aligned to the mouse reference genome GRCm38/mm10 using STAR (v.2.5.4)48. Read counts per gene locus were obtained with htseq-count (v.0.11.4)49. Statistical analysis was performed in R (v.3.6.3). Genes with total reads lower than 200 across all samples were excluded. Normalization and differential gene expression analysis was performed using DESeq2 (v.1.26.0). Batch effects were identified using sva (v.3.34.0) and subsequently modelled in the DESeq2 design formula. Genes were considered differentially expressed when they achieved anFDR of less than 0.05 and a log2-transformed fold change of greater than 1. The sequencing data are available at the NCBI GEO (http://www.ncbi.nlm.nih.gov/geo) under the accession number GSE205608.
Relating to the data in Fig. 3 and Extended Data Fig. 10, RNA extraction from sorted P14 T cells was performed using the RNeasy Plus Mini Kit (Qiagen) according to the manufacturer’s instructions. Each sample group consisted of two experimental replicates. All samples were sequenced on an Illumina NextSeq500 generating 80-bp paired-end reads. RNA-seq reads were aligned to the mouse reference genome GRCm38/mm10 using the Subread aligner (Rsubread v.2.2.6)50. Gene-level read counts were obtained by running featureCounts51, a read count summarization program within the Rsubread package52 and the inbuilt Rsubread annotation that is a modified version of the NCBI RefSeq mouse (mm10) genome annotation build 38.1. Pseudogenes, or genes that did not meet a counts per million reads (CPM) cut-off of 0.5 in at least two libraries were excluded from further analysis. Read counts were converted to log2-CPM, quantile normalized and precision weighted with the voom function of the limma package53,54 after accounting for batch effects. A linear model was fitted to each gene, and the empirical Bayes moderated t-statistic was used to assess differences in expression55,56. Raw P values were adjusted to control the global FDR across all comparisons using the ‘global’ option in the decideTests function in the limma package. Genes were called differentially expressed if they achieved an FDR of 15% or less. Enrichment analysis of Gene Ontology (GO) terms on the differentially expressed genes was performed using the goana function within the limma package57. Pathway enrichment against the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways on the differentially expressed genes was performed using the kegga function also implemented in the limma package. Gene set enrichment analysis was performed using Gene Set Enrichment Analysis (GSEA) software (v.4.0.3)58.
ChIP–seq analysis
Previously published raw ChIP–seq data for the MYB transcription factor38 were downloaded from the NCBI GEO with accession number GSE59657. Reads were mapped to the human genome (GRCh38) using the align function in Rsubread (refs. 50,52). Peak calling was performed using Homer (v.4.11)59 with an FDR set to 1 × 10−8. In brief, tags for the aligned libraries were first created using the makeTagsDirectory function within Homer then followed by peak calling using the ‘style’ factor parameter with called peaks annotated to the nearest genes. Overlap between differentially expressed genes from the RNA-seq data (mouse) and ChIP–seq data (human) was performed by first transforming the human genes associated with each annotated peak to their corresponding mouse homologues using information available in the Ensembl database through the biomaRt Bioconductor package60. The two sets of genes were then compared for common genes.
Analysis of evolutionary conservation
Genomic conservation data for the human and mouse genomes were obtained from UCSC Genome Browser (https://genome.ucsc.edu). Annotated tracks of human ChIP data were manually aligned to annotated tracks of mouse ATAC data using conserved loci of 100 vertebrates against the human genome and conserved loci of 30 mammals against the mouse genome as reference points.
Flow cytometry
Flow cytometry was performed using a Fortessa or Cytoflex LX (Beckman Coulter) and sort purification was performed on a BD FACSAria Fusion or MoFlo Astrios (Beckman Coulter). All data were analysed using FlowJo 10 (Tree Star). Graphs and statistical analyses were done with Prism 7 (GraphPad Software).
Statistics
A paired or unpaired Student’s t-test (two-tailed), Welch’s t-test, Mann–Whitney U test or one-way ANOVA was used to assess significance. Statistical methods to predetermine sample size were not used.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Extended Data
Extended Data Fig. 1. Isolation of polyclonal exhausted T cells for scRNA-seq and phenotypic characterization of CD62L+ TPEX cells in chronic infection.
(a, b) CD4+ T cell-depleted naive mice were infected with LCMV-Cl13, treated with or without anti-PD-L1, and exhausted PD-1+TIM-3loT cells were sorted at >day 30 post-infection as described19. (a) Schematic of the experimental set-up. (b) Flow cytometry plots showing the sorting strategy. (c–j) Naive congenically marked (CD45.1+) Id3-GFP P14 cells were transferred to naive recipients (Ly.5.2), which were then infected with LCMV-Docile. Splenic P14 T cells were analysed at the indicated time points after infection. (c) Schematic of the experimental set-up. (d) Flow cytometry plots showing the expression of Id3-GFP, TCF1 and CD62L among splenic P14 T cells at 7 and 21 dpi. (e) Quantification showing absolute numbers of splenic CD62L+ TPEX, CD62L− TPEX and total P14 cells (left) and frequencies of CD62L+ cells among TPEX cells (right) at the indicated time points after infection (f) Flow cytometry plots showing the expression of Ly108 and CD62L and quantification of CD62L+ TPEX cells among P14 T cells in the spleen, lymph nodes, blood, bone marrow and liver at day 31 post LCMV-Docile infection. (g–j) Histograms (g, h) and quantification (i, j) of expression of molecules as indicated in P14 T cell subsets and naive CD8+ T cells. (k–p) Congenically marked naive Nur77-GFP reporter P14 T cells were transferred into naive (k–m) or CD4-T-cell-depleted (n–p) recipient mice, which were subsequently infected with LCMV-Cl13. Nur77-GFP expression was analysed at indicated time points post-infection. (k, n) Schematics of the experimental set-up. Histograms (l, o) and quantifications (m, p) showing Nur77-GFP expression in the indicated P14 T cell subsets at 8 and 21 dpi. GMFI, geometric mean fluorescence intensity. Dots in graphs represent individual mice; box plots indicate range, interquartile and median; horizontal lines and error bars of bar graphs indicate mean and s.e.m. Data are representative of two independent experiments (e, f, i,j) and all analysed mice (m, p). P values are from two-tailed unpaired t-tests (e, i,j), two-way ANOVA (f), and one-way ANOVA (m, p); P > 0.05, not significant (n.s.).
Extended Data Fig. 2. Functional and transcriptional profiling of exhausted T cell subsets and RNA velocity analysis showing that differentiation streams originate from CD62L+ TPEX cells.
(a, b) Congenically marked naive P14 T cells were adoptively transferred into naive recipient mice, which were then infected with LCMV-Cl13. Splenic P14 T cells from each group were sorted at day 28 post-infection and restimulated independently using gp33-pulsed splenocytes in vitro. (a) Schematic of the experimental set-up. (b) Quantifications showing cytokine production of each subset after restimulation. (c–e) Wild-type mice were infected with LCMV-Docile and splenic CD8+ T cells were analysed at the indicated time points after infection. (c) Schematic of the experimental set-up. (d) Flow cytometry plots showing the expression of CD62L in TPEX (Ly108hi) and TEX (Ly108lo) cells among endogenous gp33-specific CD8+ T cells. (e) Quantification showing the proportions of CD62L-expressing cells among gp33+ TPEX cells (left) and polyclonal PD-1+ TPEX cells (right) at the indicated time points after infection (f, g) Congenically marked naive P14 T cells were adoptively transferred into naive recipient mice, which were then infected with LCMV-Cl13 or LCMV-Armstrong. Splenic P14 compartments from each group were sorted at 28 dpi and processed for bulk RNA-seq. (f) Schematic of the experimental set-up. (g) Principal component plot showing the transcriptional landscapes of sorted populations as indicated. (h–j) Congenically marked naive Tcf7-GFP P14 T cells were adoptively transferred into naive mice, which were then infected with LCMV-Cl13. P14 TPEX cells were sorted at day 28 post-infection based on the expression of Tcf7-GFP. (h) Schematic of the experimental set-up. (i) Flow cytometry plots showing the sorting strategy and post-sort purity. (j) RNA velocity analysis showing developmental trajectories of TPEX cells, together with the expression of Tcf7 (left) and Sell (right). Horizontal lines and error bars of bar graphs indicate mean and s.e.m., respectively. Data are representative of two independent experiments (b) and all analysed mice (e). P values are from Mann–Whitney tests (b) and two-tailed unpaired t-tests (e); P > 0.05, not significant (n.s.).
Extended Data Fig. 3. Capacity for self-renewal and multipotent differentiation is restricted to the CD62L+ TPEX cell compartment.
(a–f) Congenically marked naive P14 T cells were transferred into primary recipient mice (R1), which were then infected with LCMV-Cl13. The indicated subsets of P14 T cells were sorted at 21 dpi and 4×104 cells were re-transferred to infection-matched secondary recipient mice (R2). The indicated subsets of P14 T cells were sorted from R2 mice at 35 dpi and 1~3×103 cells were re-transferred to infection-matched tertiary recipient mice (R3). Splenic P14 T cells of R3 mice were analysed at day 14 post re-transfer. (a) Schematic of the experimental set-up. (b) Representative flow cytometry plots showing the sorting strategy and post-sort purities. Flow cytometry plots (c) and calculated fold expansion (d) of recovered P14 progenies at day 14 after secondary and tertiary re-transfers. Flow cytometry plots and quantifications showing expression of Ly108 and CD62L of splenic P14 cells in R2 and R3 mice (e) and average percentages of recovered CD62L+ TPEX, CD62L− TPEX and TEX cells per spleen in R2 and R3 mice (f) at day 14 post re-transfer, respectively. (g–l) Congenically marked naive P14 T cells were transferred into primary recipient mice (R1), which were then infected with LCMV-Cl13. The indicated subsets of P14 T cells were sorted at 28 dpi and 3–30 x 103 cells were re-transferred to infection-matched secondary recipient mice (R2). Splenic P14 T cells of R2 mice were analysed at day 14 post re-transfer. Of note, maximum cell numbers attainable for each subset were transferred to allow for reliable evaluation of phenotypic diversification in expanded progenies. Fold expansion of recovered progenies was then normalized to distinct input numbers. (g) Schematic of the experimental set-up. (h) Representative flow cytometry plots showing the sorting strategy and post-sort purities. Flow cytometry plots (i) and fold expansion (j) of recovered progenies at day 14 post re-transfer. Flow cytometry plots and quantifications showing expression of Ly108 and CD62L of splenic P14 cells of R2 mice (k) and average percentages of recovered CD62L+ TPEX, CD62L− TPEX, CD62L+ TEX and CD62L− TEX cells per spleen (l) in R2 mice at day 14 post re-transfer. Dots in graphs represent individual mice; horizontal lines and error bars of bar graphs indicate mean and s.e.m., respectively. Data are representative of two independent experiments (b, c, e, h, i, k) and all analysed mice (d, f, j, l). P values are from Mann–Whitney tests (d,j); P > 0.05, not significant (n.s.).
Extended Data Fig. 4. Single CD62L+ TPEX cells show a stem-like capacity for self-renewal and multipotent differentiation.
(a–d) Single naive colour-barcoded P14 T cells were transferred to primary recipient mice, which were then infected with LCMV-Armstrong. Splenic P14 T cells were analysed at day 8 post LCMV-Armstrong infection. (a) Schematic of the experimental set-up for the naive P14 single-cell transfer. (b) Flow cytometry plots showing expression of GFP and YFP (left) or BFP/CFP and CFP/T-Sap (right) in peripheral blood of retrogenic P14 donor mice (pre-gated on CD8+CD44loCD45.1+). (c) Tracking of colour-barcoded single-cell-derived progenies at 8 dpi in the spleens of three representative recipient mice. Recovered progenies were distinguished according to their combinatorial expression of GFP and YFP into populations I, II, III, IV and V, which were further subdivided by their expression of T-Sapphire, CFP, and BFP into progenies characterized by their unique combinatorial colour barcode. Note: in the display used, CFP emission appears on the diagonal between the BFP (x-axis) and T-Sapphire signal (y-axis) and is therefore indicated on both axes. (d) Flow cytometry plots depicting combined staining of CD45.1 and Thy1.1 with KLRG1 (upper row), or CD62L with PD-1 (lower row) for three progenies derived from adoptively transferred single naive P14 cells (grey: endogenous CD4−CD19− cells). (e–i) Colour-barcoded naive P14 T cells were transferred into primary recipient mice (R1), which were subsequently infected with LCMV-Cl13. P14 T cells were sorted at 28 dpi and single CD62L+ or CD62L− TPEX cells were re-transferred into naive secondary recipient mice (R2), which were subsequently infected with LCMV-Armstrong. Splenic P14 T cells were analysed at day 8 post LCMV-Armstrong infection. (e) Schematic of the experimental set-up. (f) Percentages of transferred single cells of CD62L+ TPEX, CD62L− TPEX cell or naive phenotype from which progenies were recovered at 8 dpi. (g) As in (d), but for adoptively re-transferred single CD62L+ TPEX cells. (h) Size of single-T-cell-derived progenies and frequencies of CD62L+, KLRG1+ and (i) PD-1+ cells therein. Dots in graphs represent individual clones derived from a single transferred cell. Horizontal lines and error bars of bar graphs indicate mean and s.e.m., respectively. Data show all analysed mice (h, i). P values are from Mann–Whitney tests (h–i); P > 0.05, not significant (n.s.).
Extended Data Fig. 5. CD69 expression in TPEX cells does not correlate with CD62L expression and does not predict developmental and repopulation potential; chronic LCMV infection and strong TCR stimuli favour MYB expression and the formation of stem-like CD62L+ TPEX cells.
(a) Normalized expression of Cd69 projected on the UMAP of scRNA-seq data as in Fig. 1. (b) Enrichment of Ly108+CD69+ (“TEX prog1”) and Ly108+CD69− (“TEX prog2”) signatures29 at single-cell and cluster levels. (c) Flow cytometry plots and quantification showing CD69 expression in CD62L+ and CD62L− P14 TPEX cells on day 28 post LCMV-Cl13 infection. (d–j) Congenically marked naive P14 T cells were transferred into primary recipient mice (R1), which were then infected with LCMV-Cl13. The indicated subsets of P14 T cells were sorted at 28 dpi and re-transferred to infection-matched secondary recipient mice (R2). Splenic P14 T cells of R2 mice were analysed at day 14 post re-transfer. (d) Schematic of the experimental set-up. (e) Representative flow cytometry plots showing the sorting strategy and post-sort purities. (f) Quantification of recovered P14 cells at day 14 post re-transfer. (g) Flow cytometry plots and quantifications showing expression of Ly108 and CD69 of splenic P14 cells of R2 mice and (h) average percentages of recovered CD69+ TPEX, CD69− TPEX, CD69+ TEX and CD69− TEX cells per spleen in R2 mice at day 14 post re-transfer. (i) Flow cytometry plots and quantifications showing expression of Ly108 and CD62L of splenic P14 cells of R2 mice and (j) average percentages of recovered CD62L+ TPEX, CD62L− TPEX and TEX cells per spleen in R2 mice at day 14 post re-transfer. (k–l) Naive MybGFP reporter mice were infected with either LCMV-Docile or LCMV-Armstrong and CD8+ T cells were analysed at the indicated time points after infection. (k) Representative flow cytometry plots depict Ly108 and Myb-GFP expression among antigen-specific (gp33+) CD8+ T cells. (l) Histograms (filled) show Myb-GFP expression of gp33+ CD8+ T cells in mice infected with LCMV-Docile (top) and LCMV-Armstrong (bottom). Empty histograms depict Myb-GFP expression in naive CD8+ T cells in the same samples. Corresponding quantification show the fold change of geometric mean fluorescence intensity (GMFI) of Myb-GFP in the indicated populations. (m–o) LCMV-Docile-infected MybCFP reporter mice were treated with or without anti-PD-L1. Splenic CD8+ T cells were analysed at 6 dpi. (m) Schematic of the experimental set-up. (n) Flow cytometry plots and quantification showing frequencies of splenic gp33+CD8+ T cells in anti-PD-L1-treated and untreated control mice at 6 dpi. (o) Histograms showing Myb-GFP expression of gp33+ and naive (gated on CD62L+CD44−) CD8+ T cells in the same mice. (p) Naive MybGFP and wild-type (non-reporter, control) CD8+ T cells were stimulated and cultured in vitro using plate-bound anti-CD3. Representative histogram and normalized quantification show GMFI of Myb-GFP expression in CD8+ T cells stimulated with plate-bound anti-CD3 at the indicated concentrations. (q) Flow cytometry plots and quantification show the frequencies of Ly108+ and CD62L+ cells among splenic antigen-specific (gp33+) T cells in wild-type mice at day 8 post LCMV-Docile or LCMV-Armstrong infection. (r) Congenically marked P14 T cells were adoptively transferred into naive recipient mice, which were then infected with either LCMV-Docile or LCMV-Armstrong. Splenic P14 T cells were analysed at 8 dpi. Flow cytometry plots and quantification show the frequencies of Ly108+and CD62L+ cells among splenic P14 T cells. Dots in graphs represent individual mice (c, f, l, o, q, r) and individual wells (p); box plots indicate range, interquartile and median; horizontal lines and error bars in indicate mean and s.e.m., respectively. Data are representative of two independent experiments (c, l, o, p) and all analysed mice (f, q, r). P values are from two-tailed unpaired t-tests (c, l, o–r) and Mann–Whitney tests (f); P > 0.05, not significant (n.s.).
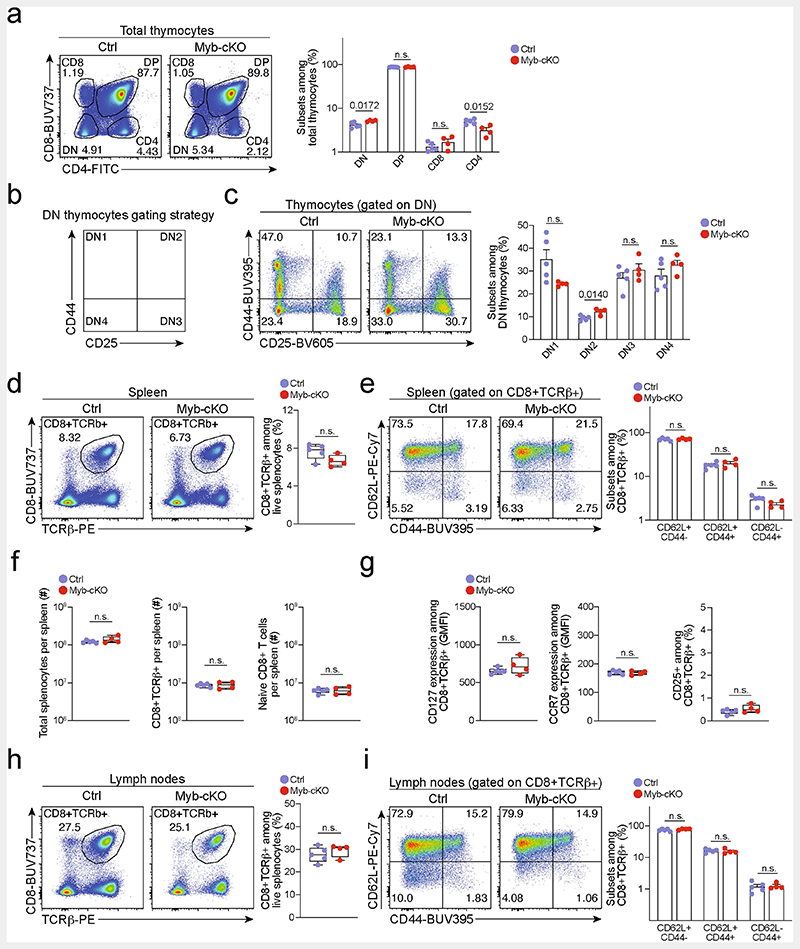
Extended Data Fig. 6. Development of mature CD8+ T cells is largely normal in Mybfl/flCd4Cre mice.
Adult 8-12 weeks Mybfl/flCd4Cre (Myb-cKO) and littermate Mybfl/fl control (Ctrl) mice were euthanized, and T cell populations were analysed in the thymus, spleen and lymph nodes. Flow cytometry and quantifications showing (a–c) frequencies of thymocyte subsets, (d–f) frequencies and abundance of mature splenic CD8+ T cells, (g) surface expression of CD127 (IL-7R), CCR7 and CD25 (IL-2R) of splenic CD8+ T cells and (h, i) frequencies of mature CD8+ T cells residing in lymph nodes. (h). Dots in graphs represent individual mice; box plots indicate range, interquartile and median. All data are representative of two independent experiments. P values are from two-tailed unpaired t-tests (d, f–h) and Mann–Whitney tests (a, c, e, i); P >0.05, not significant (n.s.).
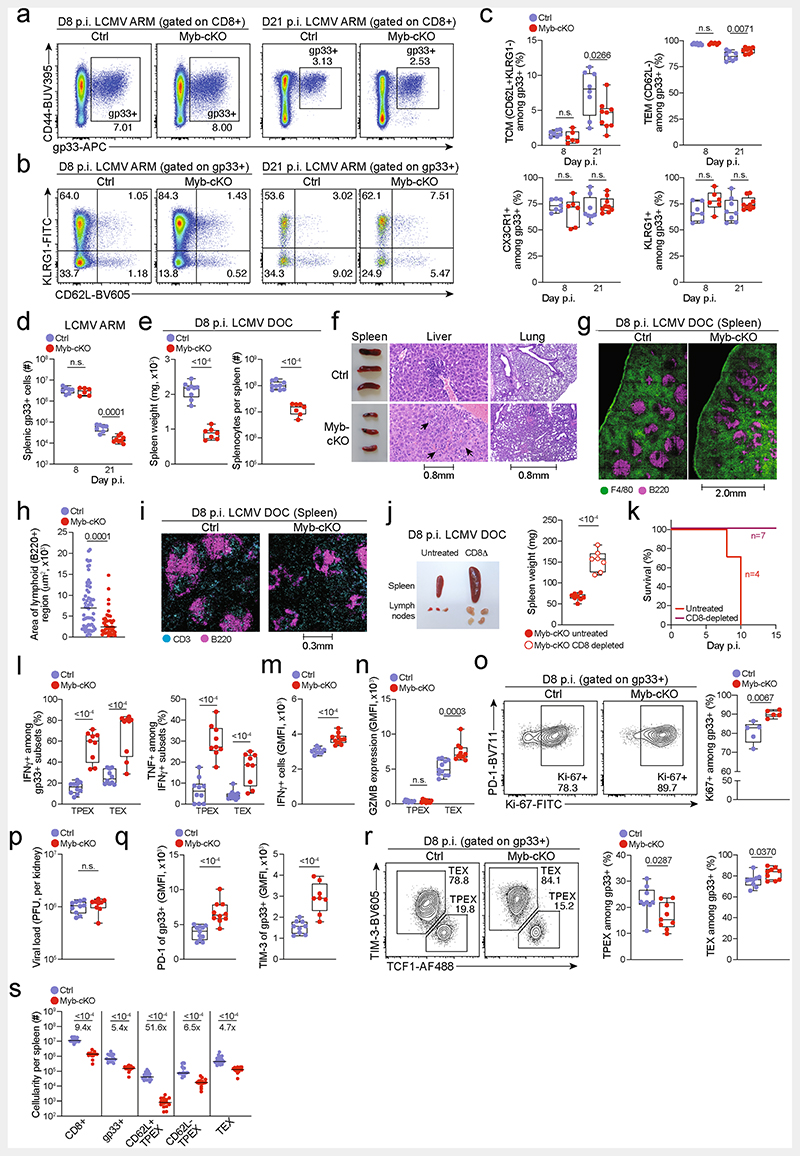
Extended Data Fig. 7. MYB is required to limit CD8+ T cell expansion and cytotoxicity in response to chronic infection.
(a–s) MybfvflCd4Cre (Myb-cKO) mice and littermate Mybfl/fl control mice (Ctrl) were infected with either LCMV-Armstrong (a–d) or LCMV-Docile (e–s). (a–b) Flow cytometry plots showing (a) splenic antigen-specific (gp33+) CD8+ cells and (b) expression of CD62L and KLRG1 among antigen-specific cells in Myb-cKO and control mice at indicated time points post LCMV-Armstrong infection. (c) Quantification of central memory (TCM), effector memory (TEM), CX3CR1+ and KLRG1+ cells among gp33+ CD8+cells in Myb-cKO and control mice at indicated time points post LCMV-Armstrong infection. (d) Numbers of splenic gp33+CD8+ T cells in Myb-cKO and control mice at indicated time points post LCMV-Armstrong infection. (e) Box plots showing the weights of spleens (left) and the total numbers of splenocytes (right) in Myb-cKO and control mice at day 8 post LCMV-Docile infection. (f) Spleen size (left) and haematoxylin and eosin staining of sections showing infiltration of immune cells (arrows) in livers (middle) and lungs (right) in Myb-cKO and control mice at 8 dpi. (g) Confocal images of F4/80 and B220 expression in frozen spleen sections and (h) quantification showing the cellular organization and area of lymphoid regions in Myb-cKO and control mice at day 8 post LCMV-Docile infection. (i) Confocal images of CD3 and B220 expression in frozen spleen sections showing the distribution of B and T cells in the spleens of Myb-cKO and control mice at day 8 post LCMV-Docile infection. (j) Image and box plot showing the size and weights of spleens in untreated and CD8+ T-cell-depleted Myb-cKO mice at day 8 post LCMV-Docile infection. (k) Survival curve of CD8-depleted Myb-cKO mice post LCMV-Docile infection. (l) Proportion of cytokine-producing antigen-specific TPEX and TEX cell subsets after gp33 peptide restimulation of Myb-cKO and control mice at day 8 post LCMV-Docile infection. (m, n) Quantification of IFNγ expression (m), and granzyme B (GZMB) expression in TPEX and TEX cells (n) in Myb-cKO and control mice at day 8 post LCMV-Docile infection. (o) Flow cytometry plots and quantification showing the proportions of Ki67+ within the gp33+compartment in Myb-cKO and control mice at day 8 post LCMV-Docile infection. (p) Box plots showing viral titres in the kidneys of Myb-cKO and control mice at day 8 post LCMV-Docile infection. (q) Box plots showing the expression of PD-1 (left) and TIM-3 (right) among gp33+CD8+ T cells of control and Myb-cKO mice at day 8 post LCMV-Docile infection. (r) Flow cytometry plots and quantification show the frequencies of TPEX cells (TCF1+TIM-3−) and TEX cells (TCF1TIM-3+) among splenic gp33+CD8+ T cells of Myb-cKO and control mice. (s) Quantification showing the absolute numbers of splenic CD8+, gp33+, CD62L+ TPEX, CD62L− TPEX and TEX cells in Myb-cKO and control mice at day 8 post LCMV-Docile infection. GMFI, geometric mean fluorescence intensity. Dots in graphs represent individual mice; box plots indicate range, interquartile and median; horizontal lines in (h) indicate median. Data are representative of two independent experiments (c–e, k–r) or all mice (j, s) and images (h) analysed; P > 0.05, not significant (n.s.).
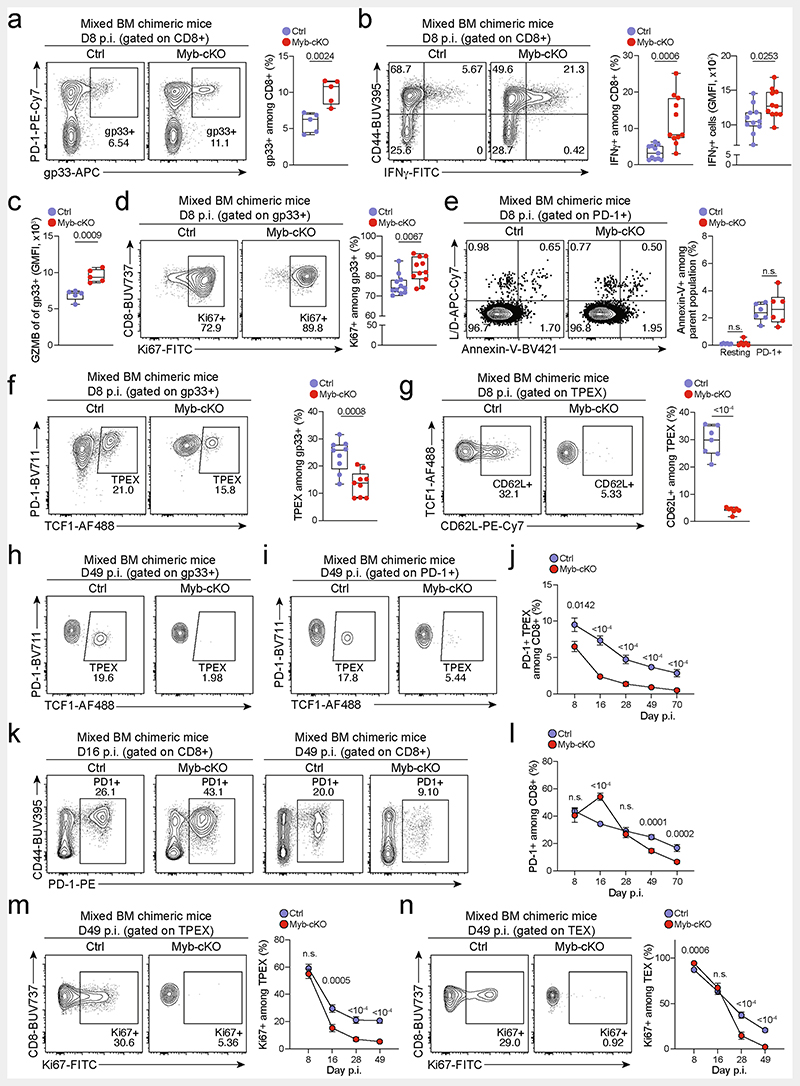
Extended Data Fig. 8. MYB limits CD8+T cell expansion and cytotoxicity during chronic infection in a cell-intrinsic manner.
(a–n) Naive CD45.1 mice were lethally irradiated and reconstituted using a mixture of Mybfl/flCd4Cre (Myb-cKO) and Cd4Cre or littermate Mybfl/fl control (Ctrl) bone marrow. Chimeric mice were subsequently infected with LCMV-Docile and analysed at the indicated time points after infection. Quantification showing the frequencies of (a) polyclonal antigen-specific gp33+cells among Myb-cKO and control CD8+ T cells at 8 dpi. (b) Flow cytometry plots and quantification of IFNγ+cells among Myb-cKO and control CD8+ T cells after peptide restimulation in vitro at 8 dpi. (c) Quantification of GZMB expression among gp33+ cells of the indicated genotypes. (d, e) Flow cytometry plots and quantification showing the frequencies of (d) Ki67+ cells and (e) annexin-V+ cells among Myb-cKO and control antigen-responsive CD8+ T cells. (f, g) Flow cytometry plots and quantification showing the frequencies of TCF1+ TPEX cells among antigen-specific T cells (f) and CD62L+ cells among TPEX cells (g) in Myb-cKO and control compartments at 8 dpi. (h) Flow cytometry plots showing the frequencies of TPEX cells among gp33+cells at 49 dpi. (i–j) Flow cytometry plots (i) and quantification (j) showing kinetics of splenic polyclonal PD-1+ TPEX cells among Myb-cKO and control CD8+ T cells after infection. (k–l) Flow cytometry plots (k) and quantification (l) showing the frequencies of the entire antigen-responsive PD-1+ cell compartment among Myb-cKO and control CD8+ T cells at indicated time points after infection. (m, n) Flow cytometry plots and quantifications showing the frequencies of Ki67+ cells among Myb-cKO and control polyclonal TPEX (m) and TEX (n) cells at indicated time points after infection. GMFI, geometric mean fluorescence intensity. Dots in graphs represent individual mice; box plots indicate range, interquartile and median. Symbols and error bars represent mean and s.e.m., respectively. All data are representative of two independent experiments. P values are from two-tailed unpaired t-tests (a–g) and Mann–Whitney tests (j–n); P >0.05, not significant (n.s.).
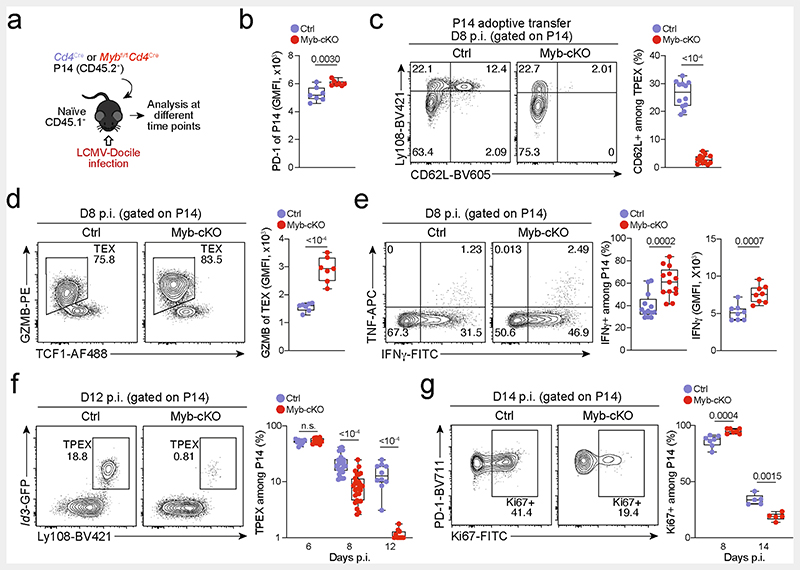
Extended Data Fig. 9. MYB limits proliferation and cytotoxicity and sustains the long-term self-renewal of exhausted CD8+ T cells.
(a–g) Congenically marked naive control(Cd4Cre) and Mybfl/flCd4Cre (Myb-cKO) P14 T cells were adoptively transferred into naive recipient mice, which were subsequently infected with LCMV-Docile. Splenic P14 T cells were analysed at indicated time points post-infection (p.i). (a) Schematic of the experimental set-up. (b) Box plot showing PD-1 expression of transferred P14 cells at 8 dpi. (c) Flow cytometry plots and quantification showing frequencies of CD62L+ cells among Myb-cKO and control P14 TPEX cells. (d) Flow cytometry plots and quantification showing the expression of granzyme B (GZMB) in Myb-cKO and control TEX P14 cells at 8 dpi. (e) Flow cytometry plots and quantifications showing the production of cytokines as indicated from Myb-cKO and control P14 T cells after gp33 peptide restimulation at 8 dpi. (f) Flow cytometry plots and quantification showing the frequencies of TPEX cells among Myb-cKO and control P14 T cells at the indicated time points after infection. (g) Flow cytometry plots and quantification showing the frequencies of Ki67+ cells among Myb-cKO and control P14 T cells at indicated time points after infection. GMFI, geometric mean fluorescence intensity. Dots in graphs represent individual mice; box plots indicate range, interquartile and median; Data are representative of two independent experiments (b–g). P values are from two-tailed unpaired t-tests; P > 0.05, not significant (n.s.).
Extended Data Fig. 10. MYB regulates the expression of multiple genes that are critical to exhausted T cell function and maintenance.
(a, b) Congenically marked Mybfl/flCd4Cre (Myb-cKO, CD45.2+) and Cd4Cre (Ctrl, CD45.2+ or CD45.2+ CD45.1+) P14 T cells were adoptively transferred into separate naive recipient (CD45.1) mice, which were then infected with LCMV-Docile. Splenic P14 TPEX cells were sorted at day 7 post-infection and processed for bulk RNA-seq. (a) Schematic of the experimental set-up. (b) Gene set enrichment analysis showing loss of CD62L+ TPEX transcriptional signature in Myb-cKO TPEX cells compared to control TPEX cells. (c) Volcano plots highlighting genes differentially expressed (FDR < 0.15) between control CD62L+ TPEX and CD62L− TPEX cells. (d–e) Mixed bone marrow chimeric mice containing congenically marked Myb-cKO and control CD8+ T cells were infected with LCMV-Docile. Flow cytometry plots (d) and quantification (e) showing the frequencies of the entire antigen-responsive PD-1+ cell compartment among Myb-cKO and control CD8+ T cells in the spleen and lymph nodes at day 70 post-infection. (f, g) Congenically marked Myb-cKO and Ctrl P14 T cells were adoptively transferred into separate naive recipient mice, which were then infected with LCMV-Docile. Splenic P14 TPEX cells were analysed at day 8 post-infection. (f) Schematic of the experimental set-up. (g) Quantification showing the abundances of the indicated P14 subsets per spleen. (h) Heat map depicting genes differentially expressed (FDR < 0.15, FC > 1) between control CD62L+ TPEX and CD62L− TPEX cell or Myb-cKO and control TPEX and TEX cells, with genes of interest annotated. (i) Gene set enrichment analysis showing loss of CX3CR1+ TEX transcriptional signature in P14 Myb-cKO TEX cells compared to control TEX cells. (j) Volcano plot highlighting genes differentially expressed (FDR < 0.15) between control and Myb-cKO TEX cells with genes of interested annotated. (k) Flow cytometry plots and quantification show the frequencies of CX3CR1+ cells among control and Myb-cKO TEX P14 T cells at day 8 post LCMV-Docile infection. (l) Flow cytometry plots and quantifications showing CX3CR1 and CD101 expression in Myb-cKO and control TEX cells at the indicated time points after infection. Dots in graph represent individual mice; box plots indicate range, interquartile and median. Symbols and error bars in (l) represent mean and s.e.m., respectively Data are representative of two independent experiments (e, g, k, l). P values are from two-tailed unpaired t-tests; P > 0.05, not significant (n.s.).
Extended Data Fig. 11. MYB directly regulates target gene expression, and CD62L+ TPEX cells have a superior potential to give rise to CX3CR1+ TEX cells.
(a–d) Representative tracks showing MYB ChIP–seq peaks in the LEF1 (a), E2F1 (b), GZMA (c), and MYB (d) gene loci of human Jurkat T cells and ATAC-seq peaks of TPEX and TEX cells in the corresponding mouse gene loci aligned according to the sequence conservation. (e–g) Congenically marked naive P14 cells were transferred to primary recipient mice (R1), which were subsequently infected with LCMV-Cl13. The indicated subsets of P14 cells were sorted at 28 dpi and re-transferred to naive secondary recipient mice (R2), which were then infected with LCMV-Armstrong. Splenic P14 T cells in R2 mice were analysed at 8 dpi. (e) Schematic of the experimental set-up. (f) Flow cytometry plots of progenies recovered at 8 dpi. (g) Cell numbers (left) and quantification of PD-1 expression (right) in P14 T cell populations derived from the indicated transferred subsets at 8 dpi. (h, i) Congenically marked naive P14 T cells were transferred into primary recipient mice (R1), which were then infected with LCMV-Docile. The indicated subsets of P14 T cells were sorted at 7 dpi and 7.5×104 cells were re-transferred to infection-matched (LCMV-Docile) secondary recipient mice (R2). Splenic P14 T cells of R2 mice were analysed at day 28 post re-transfer. (h) Schematic of the experimental set-up. (i) Flow cytometry plots and box plots showing the frequencies of CX3CR1+ and CD101+ cells among recovered TEX cells derived from the indicated re-transferred TPEX subsets at day 28 post re-transfer. Data are representative of two independent experiments. P values are from Mann–Whitney tests (g) and two-tailed unpaired t-test (i); P > 0.05, not significant (n.s.).
Extended Data Fig. 12. Effect of PD-1 signalling on CD62L+ TPEX cells.
(a–f) Congenically marked PD-1-deficient (Pdcd1−/−) and control P14 T cells were transferred to naive mice, which were subsequently infected with LCMV-Docile. Splenic P14 T cells were analysed at 7 dpi. (a) Schematic of the experimental set-up. (b) P14 T cell frequencies and numbers of indicated genotypes. (c) Flow cytometry plots and frequencies of TPEX (Ly108hiTIM-3lo) and TEX (Ly108loTIM-3hi) cells. (d) Box plots show frequencies and numbers of CD62L+ TPEX cells among control and PD-1-deficient P14 cells. Flow cytometry plots and box plots show (e) frequencies of KIT+ TPEX cells and (f) numbers of KIT+ TPEX and TEX cells per spleen. (g–k) Wild-type mice were infected with LCMV-Docile and treated with anti-PD-L1 at 200 μg/mouse at 1, 3 and 5 dpi. Splenic CD8+ T cells were analysed at 6 dpi. (g) Schematic of the experimental set-up. (h) Flow cytometry plots and quantification showing the frequencies of PD-1+ cells among splenic CD8+ T cells. (i–j) Flow cytometry plots (i) and quantification (j) showing expression of CD62L among polyclonal TPEX cells (Ly108hiTIM-3lo). (k) Quantification showing the population sizes of CD62L+ TPEX, CD62L− TPEX and TEX cells among total CD8+ T cells in untreated and anti-PD-L1-treated mice. (l–p) Wild-type mice were infected with LCMV-Docile and treated with anti-PD-L1 at 200 μg/mouse at 21, 23, 25, 27 and 29 dpi. Splenic CD8+ T cells were analysed at 31 dpi. (l) Schematic of the experimental set-up. (m–n) Flow cytometry plots (m) and quantification (n) showing the frequencies of the PD-1+ cells among splenic CD8+ T cells. (o) Flow cytometry plots and quantification showing the expression of CD62L among polyclonal TPEX cells (Ly108hiTIM-3lo). (p) Quantification showing the population sizes of CD62L+ TPEX, CD62L− TPEX and TEX cells among total CD8+ T cells in untreated and anti-PD-L1-treated mice. Dots in graphs represent individual mice; box plots indicate range, interquartile and median. Data are representative of at least two independent experiments. P values are from two-tailed unpaired t-tests; P > 0.05, not significant (n.s.).
Supplementary Material
Acknowledgements
We acknowledge I. Andrä and M. Schiemann for cell sorting, and R. Gloury and I. Hensel for technical help. This work was supported by the National Health and Medical Research Council of Australia (NHMRC) Research Fellowship (to A.K.) and Ideas Grants (APP2004333 to A.K. and C.T.; APP2001719 to I.A.P.); the European Research Council (starting grant 949719 SCIMAP to V.R.B.); the Else Kröner-Fresenius-Stiftung (EKFS 2019_A91 to V.R.B.); the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation; SFB-TRR 338/1 2021–452881907, SFB 1054–210592381 to V.R.B.); and the Deutsche Krebshilfe (DKH 70113918 to V.R.B). A.K. is a Senior Research Fellow of the NHMRC; D.T.U. is a Special Fellow of The Leukemia & Lymphoma Society and is supported by an NHMRC fellowship (1194779). We acknowledge the Melbourne Cytometry Platform for provision of flow cytometry services and the NIH Tetramer Facility for providing tetramers.
Footnotes
Author contributions C.T., L.K., V.R.B. and A.K. designed the study and wrote the manuscript. C.T., L.K. and S.R. performed core experimental work and data analysis. S.S.G. performed initial key experiments. D.C., K. Knöpper, Y.L., W.S., L.K. and S.J. performed computational analyses. K. Kanev, W.K., D.T.U., S.N., T.M., S.V.T., S.A.W., I.A.P., S.J. and J.L. performed supporting experimental work and data analysis. S.L.N., K. Kanev and D.Z. contributed new reagents or analytic tools.
Competing interests The authors declare no competing interests.
Peer review information Nature thanks Evan Newell and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Reprints and permissions information is available at http://www.nature.com/reprints.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Data availability
Most of the sequencing data generated for this study have been deposited in the NCBI GEO database with accession number GSE188526. The sequencing data shown in Extended Data Fig. 2f–j have been deposited with accession number GSE205608. Source data are provided with this paper.
References
- 1.Hashimoto M, et al. CD8 T cell exhaustion in chronic infection and cancer: opportunities for interventions. Annu Rev Med. 2018;69:301–318. doi: 10.1146/annurev-med-012017-043208. [DOI] [PubMed] [Google Scholar]
- 2.McLane LM, Abdel-Hakeem MS, Wherry EJ. CD8 T cell exhaustion during chronic viral infection and cancer. Annu Rev Immunol. 2019;37:457–495. doi: 10.1146/annurev-immunol-041015-055318. [DOI] [PubMed] [Google Scholar]
- 3.Im SJ, et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature. 2016;537:417–421. doi: 10.1038/nature19330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kallies A, Zehn D, Utzschneider DT. Precursor exhausted T cells: key to successful immunotherapy? Nat Rev Immunol. 2020;20:128–136. doi: 10.1038/s41577-019-0223-7. [DOI] [PubMed] [Google Scholar]
- 5.Utzschneider DT, et al. T Cell factor 1-expressing memory-like CD8+ T cells sustain the immune response to chronic viral infections. Immunity. 2016;45:415–427. doi: 10.1016/j.immuni.2016.07.021. [DOI] [PubMed] [Google Scholar]
- 6.Wu T, et al. The TCF1-Bcl6 axis counteracts type I interferon to repress exhaustion and maintain T cell stemness. Sci Immunol. 2016;1:eaai8593. doi: 10.1126/sciimmunol.aai8593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Speiser DE, et al. T cell differentiation in chronic infection and cancer: functional adaptation or exhaustion? Nat Rev Immunol. 2014;14:768–774. doi: 10.1038/nri3740. [DOI] [PubMed] [Google Scholar]
- 8.He R, et al. Follicular CXCR5-expressing CD8+T cells curtail chronic viral infection. Nature. 2016;537:412–416. doi: 10.1038/nature19317. [DOI] [PubMed] [Google Scholar]
- 9.Leong YA, et al. CXCR5+ follicular cytotoxic T cells control viral infection in B cell follicles. Nat Immunol. 2016;17:1187–1196. doi: 10.1038/ni.3543. [DOI] [PubMed] [Google Scholar]
- 10.Siddiqui I, et al. Intratumoral Tcf1+PD-1+CD8+ T cells with stem-like properties promote tumor control in response to vaccination and checkpoint blockade immunotherapy. Immunity. 2019;50:195–211. doi: 10.1016/j.immuni.2018.12.021. [DOI] [PubMed] [Google Scholar]
- 11.Miller BC, et al. Subsets of exhausted CD8+T cells differentially mediate tumor control and respond to checkpoint blockade. Nat Immunol. 2019;20:326–336. doi: 10.1038/s41590-019-0312-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol. 2020;20:651–668. doi: 10.1038/s41577-020-0306-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Utzschneider DT, et al. Early precursor T cells establish and propagate T cell exhaustion in chronic infection. Nat Immunol. 2020;21:1256–1266. doi: 10.1038/s41590-020-0760-z. [DOI] [PubMed] [Google Scholar]
- 14.Alfei F, et al. TOX reinforces the phenotype and longevity of exhausted T cells in chronic viral infection. Nature. 2019;571:265. doi: 10.1038/s41586-019-1326-9. [DOI] [PubMed] [Google Scholar]
- 15.Chen Z, et al. TCF-1-centered transcriptional network drives an effector versus exhausted CD8 T cell-fate decision. Immunity. 2019;51:840–855. doi: 10.1016/j.immuni.2019.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gabriel SS, et al. Transforming growth factor-β-regulated mTOR activity preserves cellular metabolism to maintain long-term T cell responses in chronic infection. Immunity. 2021;54:1698–1714. doi: 10.1016/j.immuni.2021.06.007. [DOI] [PubMed] [Google Scholar]
- 17.Khan O, et al. TOX transcriptionally and epigenetically programs CD8+T cell exhaustion. Nature. 2019;571:211–218. doi: 10.1038/s41586-019-1325-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Man K, et al. Transcription factor IRF4 promotes CD8+T cell exhaustion and limits the development of memory-like T cells during chronic infection. Immunity. 2017;47:1129–1141. doi: 10.1016/j.immuni.2017.11.021. [DOI] [PubMed] [Google Scholar]
- 19.Dähling S, et al. Type 1 conventional dendritic cells maintain and guide the differentiation of precursors of exhausted T cells in distinct cellular niches. Immunity. 2022;55:656–670. doi: 10.1016/j.immuni.2022.03.006. [DOI] [PubMed] [Google Scholar]
- 20.Hudson WH, et al. Proliferating transitory T cells with an effector-like transcriptional signature emerge from PD-1+stem-like CD8+T cells during chronic infection. Immunity. 2019;51:1043–1058. doi: 10.1016/j.immuni.2019.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zander R, et al. CD4+ T cell help is required for the formation of a cytolytic CD8+T cell subset that protects against chronic infection and cancer. Immunity. 2019;51:1028–1042. doi: 10.1016/j.immuni.2019.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Buchholz VR, et al. Disparate individual fates compose robust CD8+T cell immunity. Science. 2013;340:630–635. doi: 10.1126/science.1235454. [DOI] [PubMed] [Google Scholar]
- 23.Graef P, et al. Serial transfer of single-cell-derived immunocompetence reveals stemness of CD8+central memory T cells. Immunity. 2014;41:116–126. doi: 10.1016/j.immuni.2014.05.018. [DOI] [PubMed] [Google Scholar]
- 24.Grassmann S, et al. Distinct surface expression of activating receptor Ly49H drives differential expansion of NK cell clones upon murine cytomegalovirus infection. Immunity. 2019;50:1391–1400. doi: 10.1016/j.immuni.2019.04.015. [DOI] [PubMed] [Google Scholar]
- 25.Grassmann S, et al. Early emergence of T central memory precursors programs clonal dominance during chronic viral infection. Nat Immunol. 2020;21:1563–1573. doi: 10.1038/s41590-020-00807-y. [DOI] [PubMed] [Google Scholar]
- 26.Utzschneider DT, et al. T cells maintain an exhausted phenotype after antigen withdrawal and population reexpansion. Nat Immunol. 2013;14:603–610. doi: 10.1038/ni.2606. [DOI] [PubMed] [Google Scholar]
- 27.Abdel-Hakeem MS, et al. Epigenetic scarring of exhausted T cells hinders memory differentiation upon eliminating chronic antigenic stimulation. Nat Immunol. 2021;22:1008–1019. doi: 10.1038/s41590-021-00975-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yates KB, et al. Epigenetic scars of CD8+T cell exhaustion persist after cure of chronic infection in humans. Nat Immunol. 2021;22:1020–1029. doi: 10.1038/s41590-021-00979-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Beltra J-C, et al. Developmental relationships of four exhausted CD8+T cell subsets reveals underlying transcriptional and epigenetic landscape control mechanisms. Immunity. 2020;52:825–841.:e8. doi: 10.1016/j.immuni.2020.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ramsay RG, Gonda TJ. MYB function in normal and cancer cells. Nat Rev Cancer. 2008;8:523–534. doi: 10.1038/nrc2439. [DOI] [PubMed] [Google Scholar]
- 31.Lahortiga I, et al. Duplication of the MYB oncogene in T cell acute lymphoblastic leukemia. Nat Genet. 2007;39:593–595. doi: 10.1038/ng2025. [DOI] [PubMed] [Google Scholar]
- 32.Chen Z, et al. miR-150 regulates memory CD8 T cell differentiation via c-Myb. Cell Rep. 2017;20:2584–2597. doi: 10.1016/j.celrep.2017.08.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gautam S, et al. The transcription factor c-Myb regulates CD8+T cell stemness and antitumor immunity. Nat Immunol. 2019;20:337–349. doi: 10.1038/s41590-018-0311-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Dias S, et al. Effector regulatory T cell differentiation and immune homeostasis depend on the transcription factor Myb. Immunity. 2017;46:78–91. doi: 10.1016/j.immuni.2016.12.017. [DOI] [PubMed] [Google Scholar]
- 35.Emambokus N, et al. Progression through key stages of haemopoiesis is dependent on distinct threshold levels of c-Myb. EMBO J. 2003;22:4478–4488. doi: 10.1093/emboj/cdg434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Edling CE, Hallberg B. c-Kit—a hematopoietic cell essential receptor tyrosine kinase. Int J Biochem Cell Biol. 2007;39:1995–1998. doi: 10.1016/j.biocel.2006.12.005. [DOI] [PubMed] [Google Scholar]
- 37.Frumento G, et al. CD117 (c-Kit) is expressed during CD8+T cell priming and stratifies sensitivity to apoptosis according to strength of TCR engagement. Front Immunol. 2019;10:468. doi: 10.3389/fimmu.2019.00468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mansour MR, et al. An oncogenic super-enhancer formed through somatic mutation of a noncoding intergenic element. Science. 2014;346:1373–1377. doi: 10.1126/science.1259037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Frebel H, et al. Programmed death 1 protects from fatal circulatory failure during systemic virus infection of mice. J Exp Med. 2012;209:2485–2499. doi: 10.1084/jem.20121015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Odorizzi PM, Pauken KE, Paley MA, Sharpe A, Wherry EJ. Genetic absence of PD-1 promotes accumulation of terminally differentiated exhausted CD8+ T cells. J Exp Med. 2015;212:1125–1137. doi: 10.1084/jem.20142237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nüssing S, et al. Efficient CRISPR/Cas9 gene editing in uncultured naive mouse T Cells for in vivo studies. J Immunol. 2020;204:2308–2315. doi: 10.4049/jimmunol.1901396. [DOI] [PubMed] [Google Scholar]
- 42.Miyazaki M, et al. The opposing roles of the transcription factor E2A and its antagonist Id3 that orchestrate and enforce the naive fate of T cells. Nat Immunol. 2011;12:992–1001. doi: 10.1038/ni.2086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Stuart T, et al. Comprehensive integration of single-cell data. Cell. 2019;177:1888–1902. doi: 10.1016/j.cell.2019.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Street K, et al. Slingshot: cell lineage and pseudotime inference for single-cell transcriptomics. BMC Genomics. 2018;19:477. doi: 10.1186/s12864-018-4772-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wolf FA, Angerer P, Theis FJ. SCANPY: large-scale single-cell gene expression data analysis. Genome Biol. 2018;19:15. doi: 10.1186/s13059-017-1382-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.La Manno G, et al. RNA velocity of single cells. Nature. 2018;560:494–498. doi: 10.1038/s41586-018-0414-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bergen V, Lange M, Peidli S, Wolf FA, Theis FJ. Generalizing RNA velocity to transient cell states through dynamical modeling. Nat Biotechnol. 2020;38:1408–1414. doi: 10.1038/s41587-020-0591-3. [DOI] [PubMed] [Google Scholar]
- 48.Dobin A, et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics. 2013;29:15–21. doi: 10.1093/bioinformatics/bts635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Anders S, Pyl PT, Huber W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics. 2015;31:166–169. doi: 10.1093/bioinformatics/btu638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Liao Y, Smyth GK, Shi W. The Subread aligner: fast, accurate and scalable read mapping by seed-and-vote. Nucleic Acids Res. 2013;41:e108. doi: 10.1093/nar/gkt214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liao Y, Smyth GK, Shi W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics. 2014;30:923–930. doi: 10.1093/bioinformatics/btt656. [DOI] [PubMed] [Google Scholar]
- 52.Liao Y, Smyth GK, Shi W. The R package Rsubread is easier, faster, cheaper and better for alignment and quantification of RNA sequencing reads. Nucleic Acids Res. 2019;47:e47. doi: 10.1093/nar/gkz114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Law CW, Chen Y, Shi W, Smyth GK. voom: precision weights unlock linear model analysis tools for RNA-seq read counts. Genome Biol. 2014;15:R29. doi: 10.1186/gb-2014-15-2-r29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ritchie ME, et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015;43:e47. doi: 10.1093/nar/gkv007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Smyth GK. Linear models and empirical Bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol. 2004;3 doi: 10.2202/1544-6115.1027. [DOI] [PubMed] [Google Scholar]
- 56.McCarthy DJ, Smyth GK. Testing significance relative to a fold-change threshold is a TREAT. Bioinformatics. 2009;25:765–771. doi: 10.1093/bioinformatics/btp053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Young MD, Wakefield MJ, Smyth GK, Oshlack A. Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol. 2010;11:R14. doi: 10.1186/gb-2010-11-2-r14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Subramanian A, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005;102:15545–15550. doi: 10.1073/pnas.0506580102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Heinz S, et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol Cell. 2010;38:576–589. doi: 10.1016/j.molcel.2010.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Durinck S, Spellman PT, Birney E, Huber W. Mapping identifiers for the integration of genomic datasets with the R/Bioconductor package biomaRt. Nat Protoc. 2009;4:1184–1191. doi: 10.1038/nprot.2009.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Most of the sequencing data generated for this study have been deposited in the NCBI GEO database with accession number GSE188526. The sequencing data shown in Extended Data Fig. 2f–j have been deposited with accession number GSE205608. Source data are provided with this paper.