Abstract
The three-dimensional morphology of the left atrial appendage provides the substrate for thrombus generation, and is a harbinger for embolic material due to its direct connection to the left-sided circulation. Appreciating the development of the appendage from mesodermal layer to its adult form provides the basis to improve exclusion from the atrial circulation, and thereby can lead to a significant reduction in stroke risk. This process also provides insight into the role of the left atrial appendage as an endocrine organ, its involvement in fluid homeostasis, and its connection to the autonomic nervous system. Knowledge of the surrounding structural arrangement is critical to identify landmarks from both an endocardial and epicardial perspective to improve targeted device placement. Furthermore, correlation of the left atrial appendage body, neck, and ostium to the surrounding anatomy can also improve both procedural efficacy and safety. In addition, a working knowledge of the regional anatomy adds a prudent degree of awareness for procedural complications, and allows for early identification and timely intervention as these situations arise. A detailed understanding of the left atrial appendage embryology, histology, and gross anatomy is imperative to identify the correct device and approach for each individual patient. In addition, this increased awareness can identify areas that are in need of further innovation, and thus provide the ability to adapt and refine existing technologies to overcome pitfalls currently facing catheter-based approaches.
Keywords: left atrial appendage, cardiac embryology, ostium, stroke, embolism, atrial fibrillation, vein of marshall, endocardial ridge, mesenchymal, pulmonary vein, phrenic nerve, left circumflex artery
Introduction
The left atrial appendage (LAA) itself is one of the most “lethal” structures that exists in the human body [1]. The anatomical landscape of the pectinate muscles and crevices of the LAA, in the presence of blood stasis when the atrium is in fibrillation, produces an optimal site for the generation of thrombi [2]. The data regarding its pertinence to stroke risk are impressive, where its involvement occurs in approximately 90% of cases with a cardioembolic etiology in non-valvular atrial fibrillation (AF) patients [3]. Thus, this is one of the most critical cardiac structures where it is absolutely essential to have a detailed understanding of the embryology, histopathology, and anatomy for proceduralists of the LAA who wish to improve efficacy while establishing and maintaining safety [4].
Understanding the left atrial appendage anatomy is becoming of ever increasing importance as the toolkit of LAA exclusion devices continue to grow and more options for targeting this structure are generated [5]. Irrespective of whether a surgical or percutaneous appendage procedure is employed, the goal is to achieve LAA exclusion without damage to the LAA itself or the major structures that lie in its close proximity [6]. Because of its baseline complex structure and the tremendous amount of variation amongst individuals, difficulty still remains when attempting to match the correct approach, device, and patient so that the procedure is both therapeutic and safe [4]. Moreover, the same can be said not only pre-procedurally, but also post-procedurally in order to maintain successful exclusion from the left atrial circulation, as well as to monitor for potential consequences during long term follow-up [7]. Furthermore, this may provide a foundation for solving issues where complete exclusion is difficult, and provide a better understanding of the surrounding landscape, which can make catheter based approaches increasingly complicated [6].
In this review, we provide the proceduralist with a background of LAA embryology, endocrinology, anatomy, and place the critically related spatio-temporal structures in context, in order to provide a roadmap for safer and more effective exclusion therapies.
Embryologic and Histopathologic Overview of the Left Atrial Appendage
II-A. Development from Mesenchymal Tissue
Cardiac structures are ultimately derived from the mesodermal tissue layer during the third week of embryonal development [8]. The genesis and ultimate location of the left atrial appendage and its surrounding structures begins from a simple connection of paired cardiac mesoderm that fuses to form a two-cell thick tubular heart [9]. The initiation of this anatomical development involves a pro-myocardial plate of cells, which slowly develops into a three-dimensional tubular formation [8, 10]. This growth continues on towards a more seasoned cardiac formation via folding in an area that ultimately develops into a heart residing in the eventual pericardial space [8, 10]. It is within these early stages of embryonic growth that the left atrial appendage begins to emerge [11].
It is during the third week of gestation that the left atrial appendage originates and develops from the wall of the left side of the primary left atrium [12]. However, prior to formation of the semblance of a recognizable heart structure, a primary cardiac tube is formed which contains two caudally oriented, asymmetric, inverted “Y-shaped” structures which eventually develop into the primary right and left atrial horns; these horns are continuous with the venous system at approximately week four of embryologic development [8, 10]. At around week six of embryologic life, further development of the left atrium occurs, but it is dependent on the growth and development of the pulmonary system, which connects to the heart via the pulmonary vein-left sinus horn [8, 10].
The right and left atrial appendages are both formed from the superolateral wall of the primary atrium, with the left appendage specifically arising from the superior and left side of the primary atrial tube [8]. The appendage further matures with the formation of trabeculae, secondary to cellular protrusion into the lumen and surrounding vasculature which engages this substrate from the epicardial aspect, in order to solidify the basal layer [9]. These trabeculations start to form in the atria at around week five in humans [9]. The left atrial appendage is significantly smaller than its counterpart structure on the right side, and is heavily trabeculated, with fibers running in parallel orientation [12].
II-B. Relevant Histology of the Left Atrial Appendage
The majority of developing thrombi which are ultimately responsible for ischemic events, including those that form during rhythms of AF, are generated in the left atrial appendage [13]. In fact, approximately 90% of the thrombi that are found in patients with AF are discovered in the LAA [14]. A clear reason for this occurrence is due to the loss of the appendages’ contractile properties during AF, which leads to increased pressure causing expansion and dilation of the LAA, as well as a resultant stagnancy of blood in the trabeculated muscle [14]. Indeed, when a slower velocity of blood flow is found in AF, patients are more likely to have thrombi in the LAA [15]. The issue of stasis can be facilitated by a smaller size of the LAA (diameter of the LAA being 10–40 mm in some patients), in addition to significantly reduced contractibility, leading to an increasing risk of thrombus formation [11]. Although exclusion of the LAA from the atrial circulation can prevent thrombus formation, undesirable changes may occur after disrupting the structure and histology with device therapy [16].
The underlying histology of the left atrial appendage gives rise to multiple aspects of anatomic and physiologic relevance in the human body [17]. These functions are usually overlooked or poorly understood, although intervention involving the LAA can have multiple downstream effects post-procedurally due to alterations in cellular physiology [17]. Furthermore, the cellular composition of the appendage is quite variable both in composition and compaction. It contains both endocardial and epicardial layers, and these structures are complicated by a disarray of myocyte orientation [18]. Additionally, the myocytes of the LAA are more akin to ventricular than atrial cells in physical structure [17]. From an endocardial aspect, the LAA has a variable thickness across the area between the muscle bundles [19].
The characteristic shape of the epicardial aspect of the left appendage is due in part to the intervening conducting bundles of fibers that alter its architecture [19], and adds to the complexity of its function. In addition to Bachman’s bundle, the epicardial layer of the LAA is comprised of myocytes [18]. Bachman’s bundle is a sub-epicardial structure that provides facilitation of conduction through circumferential muscle bundles running from the right atrial appendage and coursing across to feed into the left appendage [19]. These bundles reach the LAA from the right atrial appendage after traversing the inter-atrial septum; the course of which requires the bundles themselves to branch both superior and inferiorly to encircle the neck of the LAA [20].
In addition to increased coagulability and stasis in the left appendage, histo-morphologic changes can occur over time especially in patients with chronic AF, and these micro-structural changes can further alter the endocardial substrate and may also play a role in thrombus formation [21]. A post-mortem study found that patients with AF had appendages that were triple the volume of those without (5.4 ± 3.7 cc3 vs. 1.7 ± 1.1 cc3), and had a lower volume of pectinate muscles likely due to increased distension of this structure [21]. Furthermore, thickening of the endocardial surface due to fibrous and elastic changes occurred more often in patients with AF, and these changes to the endocardial landscape also included embedding of the pectinate muscles by this infiltration [21]. Another cause of stasis in the LAA may be related to its role in atrial amyloidosis and possibly in “atrial myopathy,” because amyloid often collects in the LAA more than the right [22]. The amyloid deposition in the LAA is especially found in patients with chronic AF where significant structural remodeling and infiltration occurs over time [22].
II-C. Endocrine Function of the Left Atrial Appendage
A greatly underappreciated aspect of the appendage is its unique role as an endocrine organ [17]. Interestingly, the LAA has been found to contain a variety of cardiac progenitor cells (CPC) [23], and this attribute is fundamental to many of its underlying functions, including its impressive role in endocrine regulation [23]. It is of critical importance to cardiac physiology and homeostasis, as this structure contains almost 30% of the heart’s pool of atrial natriuretic factor [11, 24]. The release of this potent endocrine modulator leads to a myriad of effects including change in heart rate, natriuresis, and urination, all underscoring the importance of the LAA’s role in modulating body volume status [11]. Furthermore, atrial natriuretic factor release is also stimulated by low oxygen, myocyte stretch, and oxytocin, which incrementally improves the tight regulation of pressure overload and volume status sensed via the cells of the appendage [11, 17].
Distension of the wall of the appendage can occur during procedural interventions as well as certain pathologic states, and the tension on the wall is amplified because of the physical properties of its shape [17]. Increased distention of the LAA in an animal study has shown to generate an increase in heart rate, urination, and facilitation of sodium excretion [25]. These data support the potential for the LAA neuroendocrine response via release of atrial natriuretic factor from stretch receptor activated cells that reside in the LAA [11]. Therefore, there was early controversy that the critical role of the LAA in normal physiological functioning may be eliminated with LAA procedures. For example, one potential concern was in regards to LAA exclusion in precluding the ability to maintain this critical pressure-induced, stretch receptor endocrine response [25]. However, the right atrial appendage is likely critical in overcoming this dilemma, as it is able to execute similar endocrine effects as the LAA [11]. Although there is the possibility that tight endocrine regulation via both appendage structures are required, and exclusion may eventually lead to long-term adverse results, no studies to date have shown any adverse effects post-exclusion [17].
In addition to its role in natriuresis and urination, the left appendage has additional capabilities to function in tightly controlling overall volume [26]. For example, it may play a role in facilitating or mediating thirst [27]. An in vivo study in sheep showed results in support of this notion, as the sheep with non-intact versus intact LAA had a differential response to water consumption [22, 26, 27]. The sheep that did not have an intact LAA did not increase the amount of water consumption despite dehydration, however the sheep with intact LAA did [22, 26]. Furthermore, in addition to thirst, the closely connected volume status of an individual may depend on an intact and functioning LAA to regulate blood pressure via innervation with both sympathetic and parasympathetic fibers [11]. A study performed in canines found that destroying the base of the LAA did in fact decrease the heart rate, which can likely be ascribed to the reflex response from the vagus nerve, and further implicates the role of the autonomic system and its interaction with the LAA [11]. Therefore, this structure plays a magnificent role in the physiology of the human body and its appreciation is necessary when choosing which approach is best on a per-patient basis.
Anatomical Variations that Influence Appendage Exclusion Procedures
III-A. Pectinate Muscles of the Left Atrial Appendage
In contrast to the rest of the smooth left atrium, the appendage is comprised of rigid pectinate muscles that are orientated in a “whorl-like” fashion throughout, with thin-walled myocardium interdigitating these raised regions [28, 29]. These pectinate structures are almost exclusively found in the appendage, in comparison to the remainder of the left atrium [20]. The majority of hearts have variable appendageal wall thickness, and post-mortem studies have shown that the majority of pectinate muscle thickness to be of at least 1 mm in size [18, 30]. Moreover, a retrospective study found a more extensively trabeculated LAA to be a strong independent predictor of thromboembolic risk [31]. In addition to these rigid pectinate muscles, additional complexities associated with the LAA such as the three-dimensional morphology and the shape and size of the appendage ostium can create difficulty when attempting to interface opposing surfaces during LAA exclusion procedures (FIGURE 1) [32].
Figure 1. Complexity of Left Atrial Appendage Morphology.
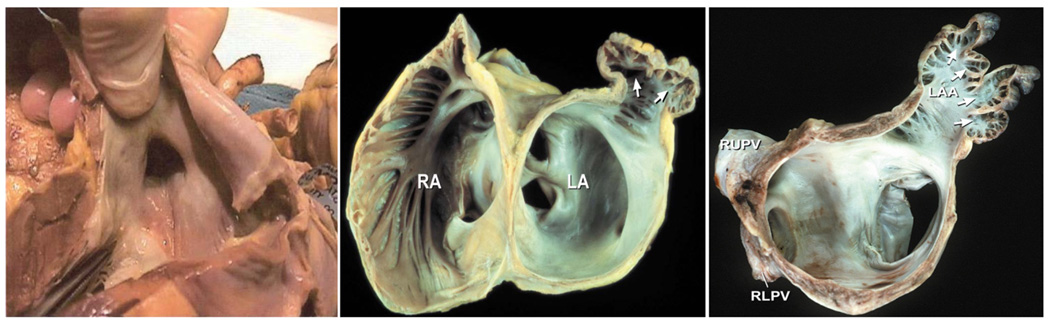
Shown are endocardial views of the left atrial appendage from post-mortem specimens of human hearts. Left panel: The left atrial appendage ostia have more of an elliptical, rather than round shape. Middle panel: An endocardial view of that shows the relationship of the left atrial appendage ostia, neck, pectinate muscles, and outpouching structure is shown. The smooth left atrium and orifices of pulmonary veins are seen in the figure in relation to the appendage. RA; right atrium. LA; left atrium. White arrows; outpouching structure of the appendage where the neck extends outwards to a 3D body. Right panel: An endocardial view of the left atrial appendage showing the complexity of a multi-lobed structure, 3D shape, and prominent pectinate ridges. LAA; left atrial appendage. White arrows; point towards multiple lobes and pectinate muscles.
III-B. Variable Morphology of the Left Atrial Appendage Ostium Shape and Dimensions
The three-dimensional shapes of the left atrial appendage are varied and complex [4]. The gross structure of the appendage usually consists of a uni- or multi-lobed, finger or stump-like extension of the left atrium [28, 29]. The distal most lobular structures of the appendage transition towards the left atrium proper by assuming a tubular shape, and further tapers down in size to form a “waist-like” structure [28]. The connecting structure from the base of this outpouching appendage to the opening of the left atrium is via the LAA ostium, which is of particular importance especially with respect to LAA procedures involving endocardially placed occlusion devices. This is because the entry for endocardial delivery of an occlusion device first engages the appendage through accessing this ostial region [18].
The ostium of the left atrial appendage has great variation in shape and is more elliptical as opposed to the circular structure associated with many appendage exclusion devices [6]. Because the LAA ostium has been noted to have great variation in shape, but falls into certain categories of distinctive morphology, these structures have been given terms for easier classification. Both the nomenclature and the prevalence of these shapes have been displayed in an elegant study to which the ostium was termed to be “oval” (68.9%), “foot-like” (10%), “triangular” (7.7%), “water drop-like”, or round (5.7%) [20] (FIGURE 2). This hollowed structure is separated from the left superior pulmonary vein both superiorly and posteriorly by the presence of the left lateral ridge of the atrium; an endocardial invagination of the lateral left atrial wall [18, 28]. The prominent ridge that separates the ostium of the LAA and the left superior pulmonary vein can be seen as a “Q-tip sign” on echocardiographic images, allowing for easier identification of this structure [29].
Figure 2. Morphologic Variation in Left Atrial Appendage Ostia.
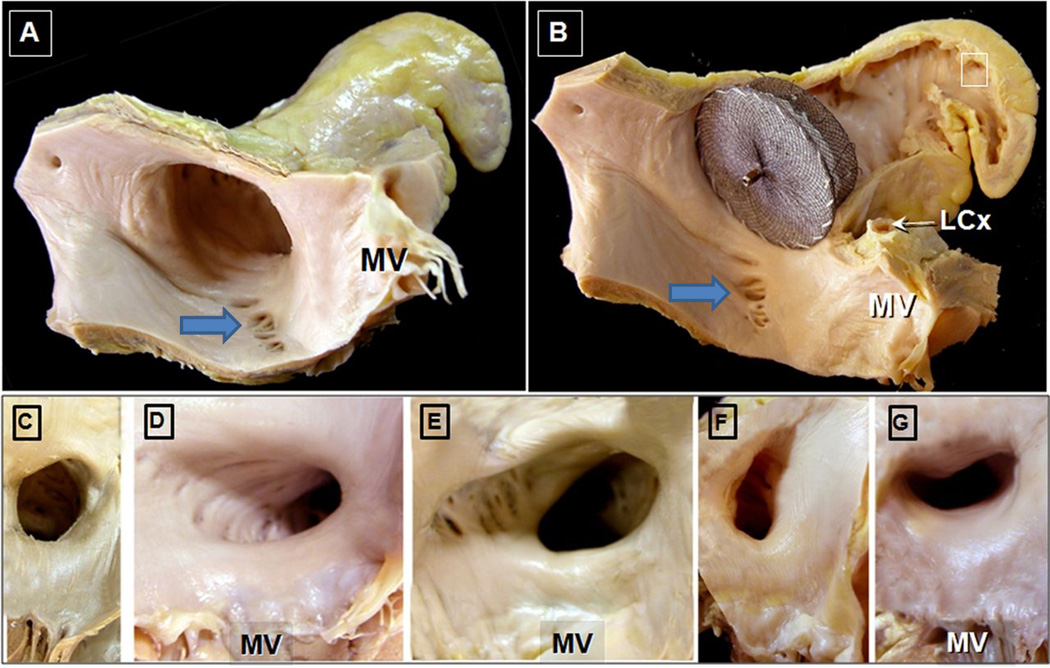
Adapted with permission from Cabrera; Heart 2014
Panel A. The ostium of the left atrial appendage with a “chicken wing” morphology is shown. Notice the slightly elliptical morphology of the os. In addition, the proximity to the mitral valve (MV) is seen. Also of note are the crevices/pits surrounding the ostium. Panel B. Amplatzer Cardiac Plug in situ 10 mm from the ostium to occlude the appendage. Notice that after appendage occlusion, there the divots are “unprotected”. Also note the proximity of the device to the circumflex artery (LCx) and mitral valve (MV). Panels C–D: Variant ostial morphology; these are classified as C) “round” ostial variant, D) “elliptical: variant”, E) triangular, F) “water-drop like”, G) “foot-like”. MV; mitral valve.
A large study performed by Ernst et al was integral in demonstrating an appreciation for the various left atrial appendage morphologies, as well as connection of these shapes to potential underlying pathologic conditions [33]. This study involved 220 hearts and the numerical dimensions of the anatomy was achieved by making resin casts to delicately study this structure [33]. In these hearts, the LAA ostium was found to have an average minimum to maximum diameter of 15–21 mm, an average size from bottom to top of 30 mm, a 31 mm distance at a right angle from the “bottom to top” measurement, and an average appendageal volume of 5220 mm3 [33]. Important clinical associations were found between AF and LAA volume, as well as correlation to appendage orifice diameter [33]. In addition, fewer branches of the LAA structure were likely to be found in patients with AF [33]. The authors also found that the larger the volume of both the LAA and ostium were more likely to have thrombus detected [33]. Interestingly, a retrospective study found an independent association with thromboembolic risk in patients who had a smaller sized diameter of LAA os [31].
III-C. Variable Morphology of the Left Atrial Appendage and Stroke Risk
There are several variations in the left atrial appendage anatomy with respect to shape, volume, length, and width [33]. The variation in LAA morphology has been found to be related to the likelihood of thrombus formation, especially with respect to larger LAA volume, depth, and number of lobes [34]. One study reported the occurrence of the number of lobes in a study of fifty hearts of chronic AF patients; the LAA was comprised of a single lobe in 68%, two lobes in 24%, three lobes in 6%, and four lobes in 2% in their studied cohort [4]. Moreover, there has been a classification of appendageal variation with respect to similarity in three-dimensional shapes of the LAA. One study noted their prevalence in a cohort, and utilized the terms “chicken wing” (48%), “cactus” (30%), “windsock” (19%), and the least common “cauliflower” (3%) [18, 35] (Figure 3).
Figure 3. Morphologic Complexity and Variation in Left Atrial Appendage 3D structure.
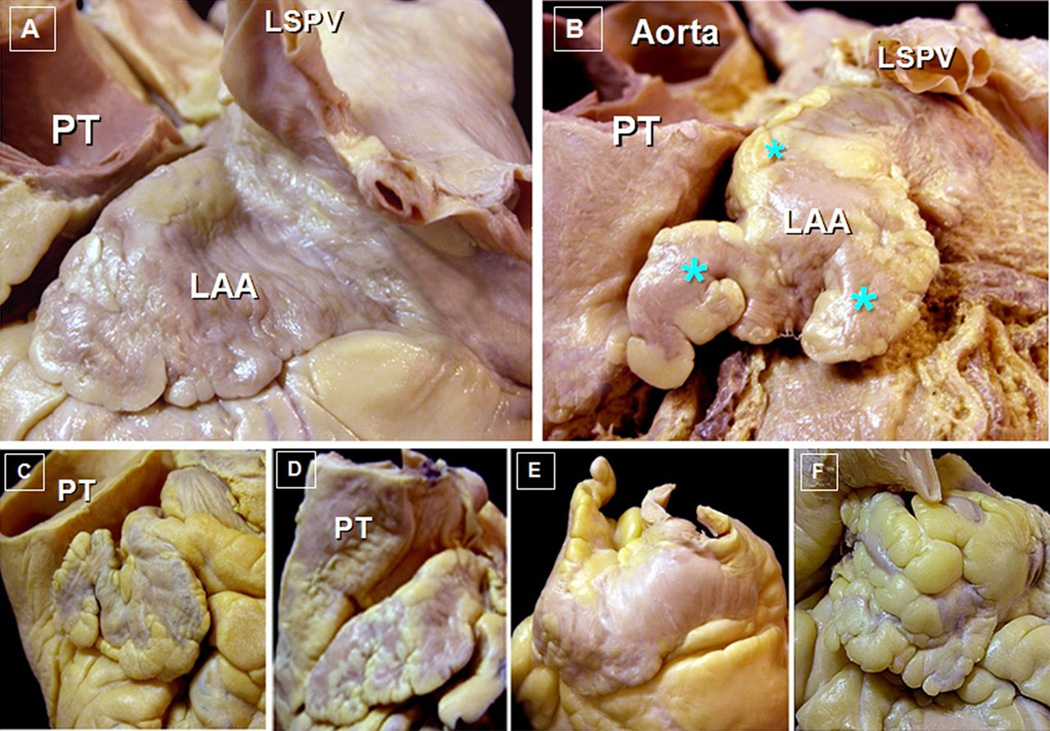
Adapted with permission from Cabrera; Heart 2014
Panel A: Post-mortem specimens showing a single lobed left atrial appendage (LAA). The closely related pulmonary trunk (PT) and left superior pulmonary vein (LSPV) are also shown. Panel B: Example of a multi-lobed left atrial appendage (asteric showing distinct lobes). The aorta, as well as the pulmonary trunk and LSPV are seen in this view. Panels C-F: Examples of variant 3D morphology of left atrial appendage shape; Panel C: “Chicken Wing”, Panel D: “Windsock”, Panel E: “Cactus”, Panel F: “Cauliflower”.
The left atrial appendage shape may also have prognostic significance in terms of likelihood of stroke. This association was shown in a retrospective study where “chicken wing” was least likely and “cauliflower” morphology was most likely to be associated with a cardioembolic event [35]. Furthermore, in a study with a cohort of low CHADS2 scores and non-valvular AF, the presence of a LAA with a “cauliflower” morphology was found to be a strong independent predictor for stroke risk [36]. However, a separate retrospective study conducted in patients with AF, showed data to support that a smaller LAA ostium and more extensive trabeculations may trump this “cauliflower” morphology in terms of stroke risk [31].
III-D. Critical Problems Associated with Left Atrial Appendage Exclusion Procedures
Although there are multiple devices and approaches, not every patient is able to achieve a complete exclusion of their left atrial appendage. This can be attributed to the wide variation in shape, number of lobes, and ostia for each unique LAA encountered, and is further complicated by potential limitations with certain imaging modalities such as transesophageal echocardiography, which is used during surgical procedures [15]. In addition, a major problem is that the failure to achieve full exclusion may lead to a more dangerous situation because of the potentially increased thrombogenicity with “residual stumps” [23]. The incomplete exclusion of the LAA can even occur with external clipping or excision procedures because of residual stump formation, as well as with percutaneous measures; though it seems that the actual type of approach used itself is not the determining factor of this inability for complete exclusion [37].
The issue of incomplete occlusion of the appendage poses a major threat to the viability of this procedural option, as well as presenting an uncertain and controversial risk of persistent thrombogenesis in these patients [15]. This problem can be borne out during LAA occlusion procedures when proper planning is not carefully performed or is lackluster. For example, it is essential for correct tailoring of the size of the device to match the variant morphology of the LAA in order to ensure a secure fit to prevent collateral blood pooling into unwanted sites that promote thrombi formation [4]. In addition to the variant morphology of the body, the “neck” of the LAA can be variable in patients as well. A retrospective study has shown that the “neck” of the LAA (which connects the body to the left atrium) had an average volume (17.3 ± 6.7 cc3), long axis diameter (20.0 ±5.3 mm), and short axis diameter (14.1 ± 4.7 mm), with associated ranges of volume (5–35.7 cc3), long axis diameter (9.5–29.9), and short axis diameter (3.1 – 24.1 mm) [4].
The finding of “divots” or diverticular pockets surrounding the endocardial surface of the appendage ostia can further complicate the ability to achieve complete exclusion, and these sites may be optimal for thrombus propagation [6, 29]. A post-mortem examination of human hearts showed these “pitted” structures to range from 0.5–10.3 mm in diameter, and found a range of distance from the LAA ostia to vary from 1.4 – 20.9 mm [6]. Because these diverticular-like pouch structures can vary in size and depth around the appendage ostium [19], these areas may not be excluded from the left atrial circulation during exclusion procedures regardless of whether the approach is occlusion or ligation, and regardless of whether an endocardial or epicardial approach is employed. The major issue with these pitted areas is that even after LAA device exclusion these divots may remain unprotected and in contact with the left atrial blood (Figure 4). Therefore, these structures can potentially house thrombi that would still pose a thromboembolic risk to the patient. This may indeed prove to be the “Achilles heel” of LAA exclusion in certain patients. Furthermore, if large enough, these pitted regions can pose difficulty during endocardial manipulation and may lead to potential damage of surrounding structures during procedures [6].
Figure 4. Diverticular Structures Surrounding the Left Atrial Appendage Ostium.
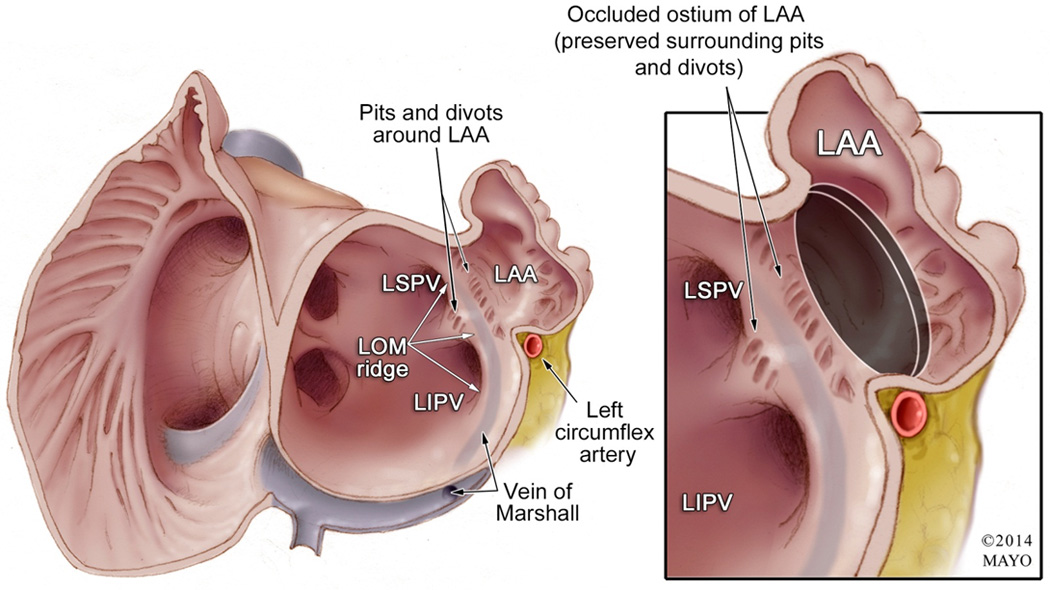
Left panel: Endocardial view of the atria showing the ostium of the left atrial appendage (LAA) and its surrounding structures. Of special note are the diverticular structures that are proximal to the LAA ostium. The relationship of the left superior and inferior pulmonary veins (LSPV and LIPV) is shown, as well as their separation by the vein/ligament of Marshal (LOM) ridge; which is an endocardial marker for the epicardial vein of Marshall (note that the course of the vein/ligament of Marshall running in between the Left pulmonary veins and the LAA, on its route to empty into the coronary sinus. The relationship of the LAA and the left circumflex artery is also illustrated. Right panel: After the left atrial appendage is occluded with a device (black circle represents the proximal end of the occlusion device), the surrounding pits/divots that are proximal to the ostium remain “unprotected” and in contact with the left atrial circulation. In addition, note the relationship of the proximal end of the occlusion device with the left circumflex artery.
The various morphologies of the left atrial appendage can pose great difficulty for procedural exclusion from the cardiac circulation because of the shape of the device itself [15]. For example, the “chicken-wing” morphology is of particular difficulty for LAA occlusion because of its shape; this is due to the course of this structure taking a very sharp bend (<180 degrees) and having this turn occur extremely close to the ostium (<20 mm) [32]. An insightful approach was taken by Freixa et al., and proved to solve the difficultly associated with certain cases with varying morphologies by implanting the Amplatzer Amulet or Plug (St. Jude Medical, MN) [32]. The key to the successful procedural outcomes stemmed from the clever use of placing a slightly oversized device (3–6 mm) and deploying the distal end of the device towards the dominant appendage lobe, in order to seal off the ostium with the proximal end of the device [32]. Innovative approaches and studies such as these, as well as their publication will aid in the future application of these ideas and techniques for procedural safety.
Relevant Spatial Relationships for Left Atrial Appendage Procedures
The left atrial appendage is a blind-ended pouch emanating from the left atria and is situated within the pericardium [28, 29]. The anatomical relations of the left appendage are complex and the critical structures surrounding the LAA include: 1) a superiorly directed pulmonary artery, 2) the appendage tip pointing in an inferomedial direction towards the free wall of the left ventricle, 3) the left phrenic nerve coursing overtop of the appendage, 4) fibers of Bachmann’s bundle that approach the LAA from the medial aspect of the atrial roof, 5) a posteriorly situated left superior pulmonary vein, and 6) an inferiorly related mitral valve [6, 12, 18, 28, 38]. However, of utmost importance to note when performing any type of appendage procedure is the critical relationship of the LAA covering the area above the left atrioventricular groove, which houses both the left circumflex artery and great cardiac vein (Figure 5) [18, 28].
Figure 5. Relevant Anatomy Surrounding the Left Atrial Appendage.
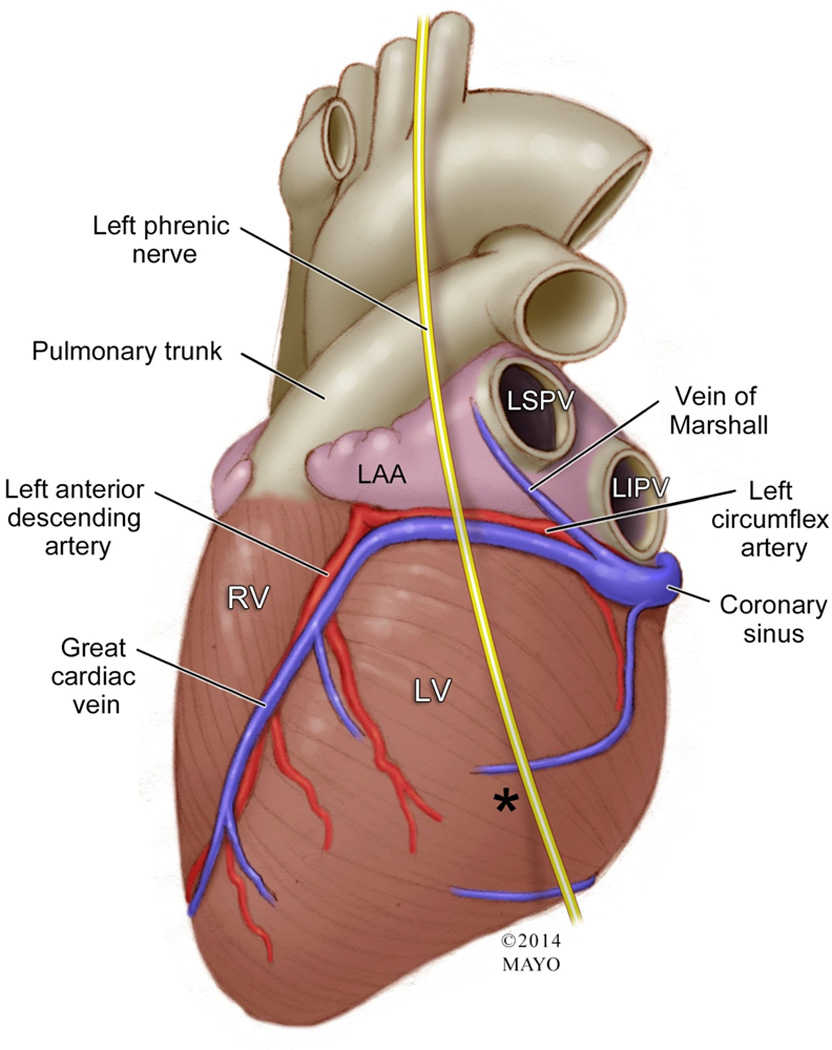
Illustration of the human heart showing the left atrial appendage (LAA) and its closely related structures. Notice that the tip of the appendage is pointing towards, as well as the body of the LAA, covering the left circumflex artery and great cardiac vein. Also seen is the overlying left phrenic nerve coursing on top of the LAA. The vein of Marshall traverses through the LAA and left superior pulmonary vein. The closely related pulmonary trunk also is shown abutting the LAA.
IV-A. Relationship of the Left Circumflex Artery to the Left Atrial Appendage Ostium
The proximity of the left circumflex artery is of particular importance when performing either endocardial or epicardial procedures of the left appendage. The left circumflex artery courses along the epicardial surface which overlies the approximate endocardial location of the LAA ostium, and was found to be in close relation to this structure in a study of postmortem hearts [6]. In this study, the authors sought to measure the actual distance of the ostium to the coronary artery [6]. This was performed by measuring the distance from the epicardial location, which correlated to the underlying the location of the endocardial ostium, to the epicardial distance to the coronary artery and on average was separated by a small distance of only 11.3 ± 5.2 mm [6]. The importance of this knowledge becomes readily apparent when taking into context situations where an endocardially placed occlusion device can: 1) be too large for the ostium and pose a risk for compression of the device on the circumflex artery, or 2) cause overdistention of the LAA and poses a risk for perforation [6, 7].
IV-B. Epicardial/Endocardial Relationship of the Left Atrial Appendage to the Left Superior Vena Cava and Left Superior Pulmonary Vein
In early embryologic life, the mere presence of blood flowing through the left superior vena cava results in an indentation at a fairly distinct location in between the left atrial appendage and the left superior pulmonary vein [38]. As a result, in adult life, where most patients have a regressed left superior vena cava, an indentation persists between the left atrial appendage and the left superior pulmonary vein which houses the vein/ligament of Marshall [38]. This provides two demarcations that can serve as landmarks from both an epicardial and endocardial perspective (Figure 6). From an epicardial perspective, a persistent indentation remains that courses between the LAA and left superior pulmonary vein [39]. From an endocardial aspect, the left lateral endocardial ridge is present and is consistently located endocardially to the vein of Marshall; this structure separates the appendage ostium from both the left superior and inferior pulmonary vein ostia [38, 39]. Furthermore, potential damage to the left superior pulmonary vein exists with intervention of the LAA during exclusion procedures. This is due to the close proximity of the pulmonary vein to the LAA, which lies directly anterior to this structure [38]. The distance found between the LAA and the left superior pulmonary vein was found in a study of post-mortem human hearts to be 11.1 ± 4.1 mm [6].
Figure 6. Endocardial and Epicardial Landmarks of the Left Atrial Appendage Ostium.
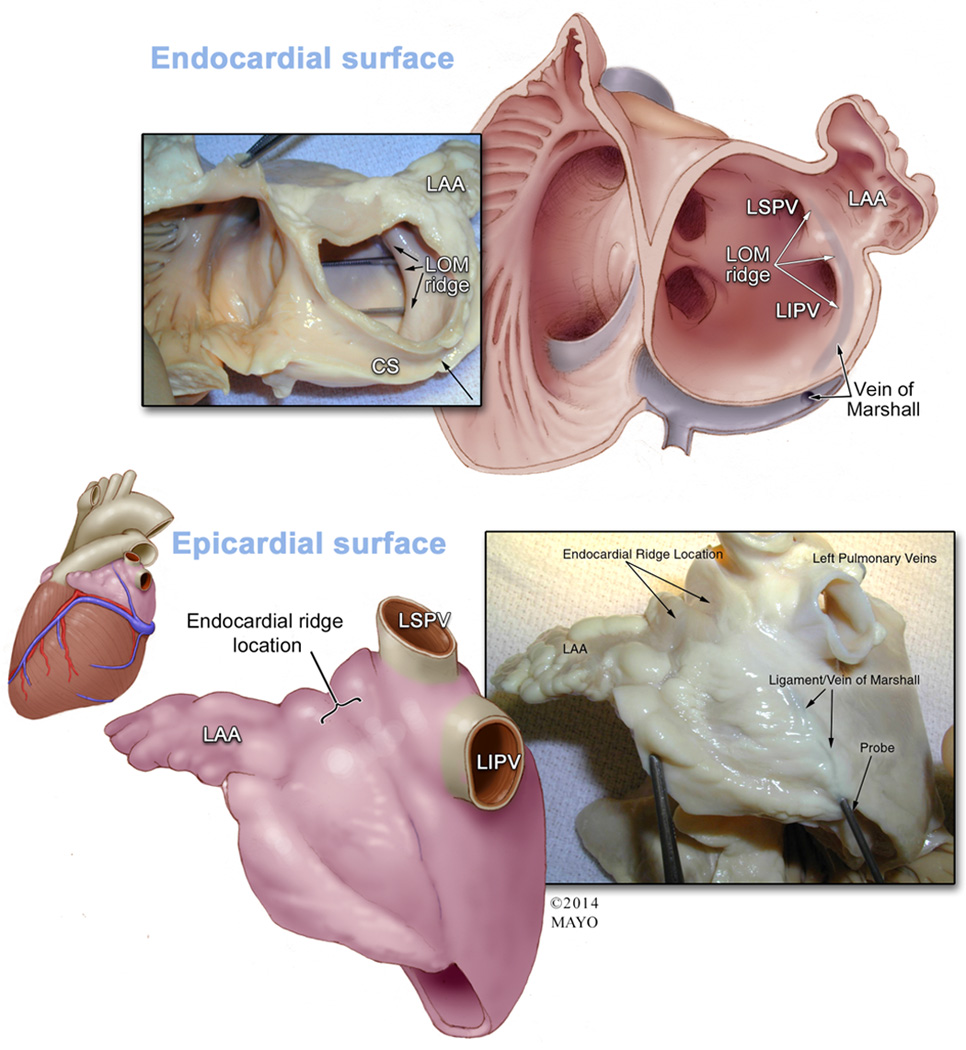
Top Panel: The left inset shows a gross anatomical specimen of an endocardial view of the right and left atrium transected at the annulus to show the relationship between the left atrial appendage (LAA), ligament/vein of Marshall (LOM) ridge, and coronary sinus (CS). Two probes are shown marking where the ostia of the left sided pulmonary veins are in relation to this ridge. The accompanying Illustration on the right shows a similar view of the gross anatomy after opening the LAA to provide a view the endocardial surface of the appendage. This image shows the endocardial relationship of the LOM ridge which is a marker for the overlying epicardial ligament of Marshall. This ridge separates the ostia of the left superior and left inferior pulmonary veins (LSPV and LIPV) and the ostium of the LAA. The ligament/vein of Marshall course is shown (blue-grey shadowing), as well as its connection to the CS. Bottom Panel: An illustration of an epicardial view showing the invagination of the epicardial surface which contains the vein/ligament of Marshall is shown in the left inset. The invagination forms a boundary between the LAA and the LSPV/LIPV. The location of the epicardial invagination reflects the location of the endocardial ridge and approximation of the ostium of the LAA. The accompanying right inset shows a gross anatomical specimen depicting this view with a probe inside of the LOM.
IV-C. Relationship of the Left Atrial Appendage Ostium and Mitral Valve
The mitral valve lies inferior to the ostium of the left atrial appendage, along with an interspaced vestibule between these two structures [28]. The mitral valve is therefore at potential risk for damage during any endocardially based procedure as it can be compressed from an oversized device, or simply because of the tight spacing that may exist between these two structures [6]. One study has shown that the average distance between the mitral valve and ostium of the appendage was 10.7 ± 2.4 mm [6]. It is therefore a critical structure for visualization during procedures and should be monitored for dysfunction or device interaction prior to final deployment as well as post-procedurally for surveillance [40].
Conclusions
In this review we focused on providing the invasive electrophysiologist with an appreciation of the LAA, with special emphasis on its magnificent abilities ranging from its unique cellular properties that are critical in development and endocrine regulation, difficulties that face proceduralists when attempting to match the correct approach and device to highly variable individual patient LAA morphology, and to highlight the extremely important spatial anatomy surrounding the LAA that can be damaged during procedures. Though many advances have been made in this field and innovations continue to abound at a tremendous pace, a well-developed knowledge of the anatomy and physiology of the LAA, including the variations that must be assessed on a case-by-case basis, can increase the safety and efficacy of LAA exclusion procedures.
References
- 1.Johnson WD, et al. The left atrial appendage: our most lethal human attachment! Surgical implications. European Journal of Cardio-Thoracic Surgery. 2000;17(6):718–722. doi: 10.1016/s1010-7940(00)00419-x. [DOI] [PubMed] [Google Scholar]
- 2.Singh I, Holmes D., Jr Left Atrial Appendage Closure. Current Cardiology Reports. 2010;12(5):413–421. doi: 10.1007/s11886-010-0122-9. [DOI] [PubMed] [Google Scholar]
- 3.Blackshear JL, Odell JA. Appendage obliteration to reduce stroke in cardiac surgical patients with atrial fibrillation. The Annals of Thoracic Surgery. 1996;61(2):755–759. doi: 10.1016/0003-4975(95)00887-X. [DOI] [PubMed] [Google Scholar]
- 4.Heist EK, et al. Analysis of the left atrial appendage by magnetic resonance angiography in patients with atrial fibrillation. Heart Rhythm. 2006;3(11):1313–1318. doi: 10.1016/j.hrthm.2006.07.022. [DOI] [PubMed] [Google Scholar]
- 5.Hussain S, Malhotra R, DiMarco J. Left Atrial Appendage Devices for Stroke Prevention in Atrial Fibrillation. Journal of Cardiovascular Translational Research. 2014;7(4):458–464. doi: 10.1007/s12265-014-9565-5. [DOI] [PubMed] [Google Scholar]
- 6.Su P, McCarthy KP, Ho SY. Occluding the left atrial appendage: anatomical considerations. Heart. 2008;94(9):1166–1170. doi: 10.1136/hrt.2006.111989. [DOI] [PubMed] [Google Scholar]
- 7.Alli O, Holmes DR., Jr Left Atrial Appendage Occlusion for Stroke Prevention. Current Problems in Cardiology. 2012;37(10):405–441. doi: 10.1016/j.cpcardiol.2012.06.001. [DOI] [PubMed] [Google Scholar]
- 8.Moorman A, et al. Development of the Heart: (1) Formation of the Cardiac Chambers And Arterial Trunks. Heart. 2003;89(7):806–814. doi: 10.1136/heart.89.7.806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sedmera D, P T, Vuillemin M, Thompson RP, Anderson RH. Developmental Patterning of the Myocardium. Anatomic Rec. 2000;258:319–337. doi: 10.1002/(SICI)1097-0185(20000401)258:4<319::AID-AR1>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- 10.Sherif HMF. The developing pulmonary veins and left atrium: implications for ablation strategy for atrial fibrillation. European Journal of Cardio-Thoracic Surgery. 2013;44(5):792–799. doi: 10.1093/ejcts/ezt098. [DOI] [PubMed] [Google Scholar]
- 11.Al-Saady NM, Obel OA, Camm AJ. Left atrial appendage: structure, function, and role in thromboembolism. Heart. 1999;82(5):547–554. doi: 10.1136/hrt.82.5.547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Al-Saady NM, Obel OA, Camm AJ. Left atrial appendage: structure, function, and role in thromboembolism. Heart. 1999;82(5):547–554. doi: 10.1136/hrt.82.5.547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lerakis S, Synetos A. Left atrial appendage exclusion system for stroke prevention in atrial fibrillation: a percutaneous device delivery approach. Minerva Cardioangiol. 2008;56(6):667–670. [PubMed] [Google Scholar]
- 14.Aryana A, Saad EB, d'Avila A. Left atrial appendage occlusion and ligation devices: what is available, how to implement them, and how to manage and avoid complications. Curr Treat Options Cardiovasc Med. 2012;14(5):503–519. doi: 10.1007/s11936-012-0203-8. [DOI] [PubMed] [Google Scholar]
- 15.Fuller C, Reisman M. Stroke Prevention in Atrial Fibrillation: Atrial Appendage Closure. Current Cardiology Reports. 2011;13(2):159–166. doi: 10.1007/s11886-010-0162-1. [DOI] [PubMed] [Google Scholar]
- 16.Holmes DR, Jr, et al. Left Atrial Appendage Occlusion: Opportunities and Challenges. Journal of the American College of Cardiology. 2014;63(4):291–298. doi: 10.1016/j.jacc.2013.08.1631. [DOI] [PubMed] [Google Scholar]
- 17.Hara H, et al. Is the left atrial appendage more than a simple appendage? Catheterization and Cardiovascular Interventions. 2009;74(2):234–242. doi: 10.1002/ccd.21983. [DOI] [PubMed] [Google Scholar]
- 18.Cabrera JA, Saremi F, Sánchez-Quintana D. Left atrial appendage: anatomy and imaging landmarks pertinent to percutaneous transcatheter occlusion. Heart. 2014 doi: 10.1136/heartjnl-2013-304464. [DOI] [PubMed] [Google Scholar]
- 19.Ho SY, Cabrera JA, Sanchez-Quintana D. Left Atrial Anatomy Revisited. Circulation: Arrhythmia and Electrophysiology. 2012;5(1):220–228. doi: 10.1161/CIRCEP.111.962720. [DOI] [PubMed] [Google Scholar]
- 20.Wang K, H S, Gibson DG, Anderson RH. Archietecture of Atrial Musculature in Humans. Br Heart J. 1995;73:559–565. doi: 10.1136/hrt.73.6.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shirani J, Alaeddini J. Structural remodeling of the left atrial appendage in patients with chronic non-valvular atrial fibrillation: Implications for thrombus formation, systemic embolism, and assessment by transesophageal echocardiography. Cardiovasc Pathol. 2000;9(2):95–101. doi: 10.1016/s1054-8807(00)00030-2. [DOI] [PubMed] [Google Scholar]
- 22.Leone O, et al. Amyloid deposition as a cause of atrial remodelling in persistent valvular atrial fibrillation. Eur Heart J. 2004;25(14):1237–1241. doi: 10.1016/j.ehj.2004.04.007. [DOI] [PubMed] [Google Scholar]
- 23.Sakellaridis T, et al. Left atrial appendage exclusion-Where do we stand? J Thorac Dis. 2014;6(Suppl 1):S70–S77. doi: 10.3978/j.issn.2072-1439.2013.10.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sakellaridis T, et al. Left atrial appendage exclusion—Where do we stand? Journal of Thoracic Disease. 2014:S70–S77. doi: 10.3978/j.issn.2072-1439.2013.10.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kappagoda CT, Linden RJ, Saunders DA. The effect on heart rate of distending the atrial appendages in the dog. The Journal of Physiology. 1972;225(3):705–719. doi: 10.1113/jphysiol.1972.sp009965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zimmerman M, Blaine EH, Stricker EM. Water intake in hypovolemic sheep: effects of crushing the left atrial appendage. Science. 1981;(211):489–491. doi: 10.1126/science.7455689. [DOI] [PubMed] [Google Scholar]
- 27.Stöllberger C, Schneider B, Finsterer J. Elimination of the left atrial appendage to prevent stroke or embolism?*: Anatomic, physiologic, and pathophysiologic considerations. CHEST Journal. 2003;124(6):2356–2362. doi: 10.1378/chest.124.6.2356. [DOI] [PubMed] [Google Scholar]
- 28.Ho SY, McCarthy KP. Anatomy of the Left Atrium for Interventional Electrophysiologists. Pacing and Clinical Electrophysiology. 2010;33(5):620–627. doi: 10.1111/j.1540-8159.2009.02659.x. [DOI] [PubMed] [Google Scholar]
- 29.Ho SY, McCarthy KP, Faletra FF. Anatomy of the left atrium for interventional echocardiography. European Journal of Echocardiography. 2011;12(10):i11–i15. doi: 10.1093/ejechocard/jer093. [DOI] [PubMed] [Google Scholar]
- 30.Veinot JP, et al. Anatomy of the Normal Left Atrial Appendage: A Quantitative Study of Age-Related Changes in 500 Autopsy Hearts: Implications for Echocardiographic Examination. Circulation. 1997;96(9):3112–3115. doi: 10.1161/01.cir.96.9.3112. [DOI] [PubMed] [Google Scholar]
- 31.Khurram IM, et al. Relationship between left atrial appendage morphology and stroke in patients with atrial fibrillation. Heart Rhythm. 2013;10(12):1843–1849. doi: 10.1016/j.hrthm.2013.09.065. [DOI] [PubMed] [Google Scholar]
- 32.Freixa X, et al. The Chicken-Wing Morphology: An Anatomical Challenge for Left Atrial Appendage Occlusion. Journal of Interventional Cardiology. 2013;26(5):509–514. doi: 10.1111/joic.12055. [DOI] [PubMed] [Google Scholar]
- 33.Ernst G, S C, Abzeiher F, Veit-Dirscerl W, Bonner E, Bibus B, Schneider B, Slany J. Morphology of the Left Atrial Appendage. Anatomic Rec. 1995;242(4):553–561. doi: 10.1002/ar.1092420411. [DOI] [PubMed] [Google Scholar]
- 34.Yamamoto M, et al. Complex Left Atrial Appendage Morphology and Left Atrial Appendage Thrombus Formation in Patients With Atrial Fibrillation. Circulation: Cardiovascular Imaging. 2014;7(2):337–343. doi: 10.1161/CIRCIMAGING.113.001317. [DOI] [PubMed] [Google Scholar]
- 35.Di Biase L, et al. Does the Left Atrial Appendage Morphology Correlate With the Risk of Stroke in Patients With Atrial Fibrillation?: Results From a Multicenter Study. Journal of the American College of Cardiology. 2012;60(6):531–538. doi: 10.1016/j.jacc.2012.04.032. [DOI] [PubMed] [Google Scholar]
- 36.Kimura T, et al. Anatomical characteristics of the left atrial appendage in cardiogenic stroke with low CHADS2 scores. Heart Rhythm. 2013;10(6):921–925. doi: 10.1016/j.hrthm.2013.01.036. [DOI] [PubMed] [Google Scholar]
- 37.Apostolakis E, et al. Surgical Strategies and Devices for Surgical Exclusion of the Left Atrial Appendage: A Word of Caution. Journal of Cardiac Surgery. 2013;28(2):199–206. doi: 10.1111/jocs.12055. [DOI] [PubMed] [Google Scholar]
- 38.Macedo PG, et al. Correlative Anatomy for the Electrophysiologist: Ablation for Atrial Fibrillation. Part I: Pulmonary Vein Ostia, Superior Vena Cava, Vein of Marshall. Journal of Cardiovascular Electrophysiology. 2010;21(6):721–730. doi: 10.1111/j.1540-8167.2010.01728.x. [DOI] [PubMed] [Google Scholar]
- 39.DeSimone CV, et al. Myocardium of the Superior Vena Cava, Coronary Sinus, Vein of Marshall, and the Pulmonary Vein Ostia: Gross Anatomic Studies in 620 Hearts. Journal of Cardiovascular Electrophysiology. 2012;23(12):1304–1309. doi: 10.1111/j.1540-8167.2012.02403.x. [DOI] [PubMed] [Google Scholar]
- 40.Palios J, P I. Thromboembolism Prevention via Transcatheter Left Atrial Appendage Closure with Transesophageal Echocardiography Guidance. Thrombosis. 2014 doi: 10.1155/2014/832752. [DOI] [PMC free article] [PubMed] [Google Scholar]


