Abstract
Expression of CD28 is highly regulated during thymic development, with CD28 levels extremely low on immature thymocytes but increasing dramatically as CD4−CD8− cells initiate expression of TCRβ. B7-1 and B7-2, the ligands for CD28, have a restricted distribution in the thymic cortex where immature thymocytes reside, and are more highly expressed in the medulla where the most mature thymocytes are located. To determine the importance of this regulated CD28/B7 expression for T cell development, we examined the effect of induced CD28 signaling of immature thymocytes in CD28/B7-2 double transgenic mice. Strikingly, we found that differentiation to the CD4+CD8+ stage in CD28/B7-2 transgenics proceeds independent of the requirement for TCRβ expression manifest in wildtype thymocytes, occurring even in Rag- or CD3ɛ- knockouts. These findings indicate that signaling of immature thymocytes through CD28 in the absence of TCR- or pre-TCR-derived signals can promote an aberrant pathway of T cell differentiation, and highlight the importance of finely regulated physiologic expression of CD28 and B7 in maintaining integrity of the “β” checkpoint for pre-TCR/TCR-dependent thymic differentiation.
Keywords: costimulation, thymus, cell differentiation, T cells
Introduction
CD28 is expressed constitutively on peripheral T cells where its role in promoting the T cell activation program is well established. In the thymus, CD28 is expressed at the highest levels on CD4+CD8+ double positive (DP) cells and at somewhat lower levels on CD4+ and CD8+ single positive (SP) cells. CD28 expression is extremely low on TCRneg CD4−CD8− double negative (DN) cells which do not express TCRβ but is substantially increased on more mature DN which express TCRβ ((1) and this report). Under normal conditions, expression of the CD28 ligands B7-1 (CD80) and B7-2 (CD86) is also highly regulated within the thymus, where expression of these ligands is restricted in its distribution on cortical epithelial cells (2) and on medullary epithelial and dendritic cells (3, 4). The restricted expression of CD28 during early thymic development should reduce the likelihood that CD28-B7 interactions would occur prior to TCRβ expression and suggests that the absence of costimulatory signaling during early T cell differentiation may be critical for normal T cell development.
In order to determine whether the induction of CD28/B7 signaling during early thymic development would have consequences for the thymic differentiation processes, we have generated CD28/B7-2 double transgenic mice in which CD28 is expressed on immature DN thymocytes and in which expression of high levels of CD28 on DN cells is no longer coupled to expression of TCRβ and the pre-TCR. In addition, the CD28 ligand B7-2 is expressed throughout the thymus. We have found that exposure of immature DN thymocytes to CD28 engagement by B7-2 results in profound alterations in thymic development, allowing thymocytes to bypass the β-checkpoint and to progress from the DN to DP stage without rearranging the TCRβ chain. Neither a CD28 truncation mutant lacking the cytoplasmic domain nor a CD28 mutant in which the PI3K/Grb2 binding domain is disrupted is capable of mediating this β-checkpoint bypass, indicating that the intracellular motifs previously demonstrated to mediate CD28 signaling in peripheral T cells are also critical in driving differentiation of DN thymocytes. When expressed on either a Rag-2- or CD3ɛ–deficient background, full length CD28 (CD28.FL) and B7-2 reverse the developmental block that occurs at the DN stage in both of these knockouts. The bypass of the β-checkpoint observed in the CD28/B7-2 double transgenics provides, to our knowledge, the first evidence that signaling through CD28 in the absence of pre-TCR and therefore, independent of pre-TCR-derived signals, can promote T cell differentiation. Furthermore, our results may offer insight into the finely regulated normal expression patterns of CD28 and B7 within the thymus. By restricting B7 expression and permitting only low levels of CD28 expression on early TCRneg DN thymocytes, the developmental program of the thymus ensures that the abortive thymic differentiation pathway resulting from early CD28-B7 interactions does not occur, and that DN thymocytes are driven to differentiation uniquely through a productive pathway that is dependent upon pre-TCR and TCR signals.
Material and Methods
Mice
C57BL/6 (B6) mice were obtained from Frederick Cancer Research Facility (Frederick, MD). C57BL/6JIghaThy1aGpi1a mice were obtained from The Jackson Laboratory (Bar Harbor, ME) and maintained at the Frederick Cancer Research Facility. B7-2 Line 7, CD28.FL, CD28.TM and CD28.Y189F transgenic mice and TCRβ, CD3ɛ and Rag-2 knockout mice were generated as previously described (5–8) and maintained in our breeding facility at Bioqual Inc. (Rockville, MD). All mice were used at between 8 and 12 weeks of age.
Antibodies
Anti-CD4, CD8, TCRβ (H57-597), CD45.1 (Ly5.2), CD45.2 (Ly5.1), CD90.1 (Thy1.1), B220, TCRγδ(GL3), Mac-1, NK1.1, GR-1, CD11c, TER-119, and CD28 Abs were purchased from BD PharMingen (San Diego, CA). Anti-B7-2 (GL1) Ab was generated as described previously (9).
Flow cytometric analysis
Single cell suspensions were prepared from thymus and resuspended in FACS buffer (0.2% BSA, 0.01% sodium azide in HBSS without phenol red). Anti-FcR mAb 24G2 (blocks FcγII and FcγIII) was added to prevent FcR-mediated binding of mAb, and cells were then incubated with directly conjugated FITC- and PE-labeled mAbs for 30 min. Following extensive washing, cells were incubated with biotin-labeled mAb and strepavidin-Cy-Chrome conjugate sequentially. Viable cells were analyzed by FACScan (Becton Dickinson, San Jose, CA) using CellQuest software. Four color staining was performed using FITC-labeled mAb, PE-labeled mAb, Cy-5-labeled mAb, biotinylated-mAb, and strepavidin-Alexa 594 conjugate (Molecular Probes, Eugene, OR). Viable cells, as determined by PI exclusion, were then analyzed on a dual-laser FACStar Plus (Becton Dickinson). Detection of intracellular TCRβ was performed as described by manufacturer’s instructions (BD Biosciences). Briefly, cells were fixed in Cytofix/Cytoperm solution for 20 min at room temperature, washed in PermWash buffer, then incubated with FITC-labeled anti-TCRβ (H57-597) mAb or an isotype control for 30 min, followed by extensive washes in PermWash buffer before analysis on a FACScan.
Bone marrow chimeras
Radiation bone marrow chimeras were prepared as described previously (10). CD28.FL/CD3ɛ KO → B7-2 Line 7/CD3ɛ KO chimeras were generated by reconstituting lethally irradiated (950 rad) B7-2 Line 7 transgenic mice on a CD3ɛ KO background with 107 T-depleted bone marrow cells from CD28 FL transgenics on a CD3ɛ KO background.
Immunohistology
Sections (6 μm) of OCT-embedded frozen tissue were air dried for 30 min and then incubated at least 2 hr with optimal dilutions of the primary antibodies rat anti-B7-2 (clone GL1, BD Biosciences PharMingen, San Diego, CA) and rabbit polyclonal anti-keratin 14 (Covance Research Products, Berkeley, CA). Tissues were washed and, after an amplification step for B7-2 with goat anti-rat IgG, immunoreactivity was detected with fluorochrome-conjugated donkey anti-rabbit IgG FITC and donkey anti-goat IgG Texas Red (Jackson ImmunoResearch Laboratories, West Grove, PA). Analysis was performed with an Olympus Provis AX70 microscope (Olympus, Melville, NY) and images were taken with a SPOT RT Color Camera and SPOT Imaging Software (Diagnostic Instruments, Sterling Heights, MI).
Results
Thymic expression patterns of CD28 and B7-2 in wildtype mice
In order to understand the role that regulated expression of costimulatory molecules may play during thymic development, we first examined CD28 levels on thymic subpopulations from wild type B6 mice. As has previously been described (1), we find that CD28 is expressed at the highest levels on DP cells and lower on CD4+ and CD8+ SP cells (Fig 1A). Analysis of CD28 expression on DN thymocytes revealed that CD28 levels are low on DN1, DN2 and the majority of DN3 cells as compared to an isotype-matched control antibody but are elevated on a subpopulation of DN3 cells and are uniformly high on DN4 thymocytes (Fig. 1B). Interestingly, when the CD25+ DN thymocytes (DN2 and DN3) were examined for expression of CD28 and TCRβ, we find that those cells expressing high levels of CD28 also express TCRβ (Fig. 2A) suggesting that there may be an important developmental link between expression of the pre-TCR and CD28. To determine whether the increase in CD28 expression levels observed in DN thymocytes which express TCRβ could result from signaling through the nascent pre-TCR, we tested the ability of anti-CD3 treatment to induce CD28 up-regulation in Rag KO mice. Treatment of Rag KO mice with anti-CD3 rescues development of DP thymocytes and is thus thought to reproduce critical aspects of pre-TCR signaling (11, 12). Levels of CD28 on untreated Rag KO thymocytes were low and were similar to those observed in wildtype DN3 cells lacking expression of TCRβ. However, when Rag KO mice were treated with anti-CD3, the majority of DN thymocytes expressed increased levels of CD28 comparable to levels on TCRβ+ wildtype DN cells (Fig. 2B). Similarly, increased levels of CD28 were observed on the DP cells which develop in anti-CD3-treated Rag KOs (Fig. 2B). These findings indicate that increased expression of CD28 in developing thymocytes may be coupled to expression of a signaling-competent pre-TCR.
Figure 1.

CD28 expression during thymocyte development A. Thymocytes from B6 mice were stained with biotin-conjugated anti-CD4, FITC anti-CD8 and PE anti-CD28. Histograms show CD28 staining on CD4−CD8− DN, CD4+CD8+ DP, CD4+SP and CD8+SP thymocytes (black). Staining with an isotype-matched control antibody is also shown (gray). For the CD28 staining, the mean fluorescence intensity (MFI) for bracketed regions is indicated to the right of each figure. B. Thymocytes were labeled with anti-CD3, -TCRβ, -CD4, -CD8, -B220, -TCRγ, -MAC-1, -NK1.1, -GR.1, TER-119 and –CD11c (all biotinylated and detected with strepavidin-PerCP), FITC anti-CD25, PE-anti-CD28 and APC anti-CD44. PerCP-negative cells were analyzed for expression of CD25 versus CD44. Histograms show CD28 staining on CD44+CD25− (DN1), CD44+CD25+ (DN2), CD44−CD25+ (DN3) and CD44−CD25− (DN4) thymocytes. Staining with an isotype-matched control antibody is also shown (gray). For the CD28 staining, the percent population (when other than 100%) and MFI for bracketed regions are indicated to the right of each figure.
Figure 2.

Coordinate expression of TCRβ and CD28 in DN thymocytes. A. Thymocytes were labeled with anti-CD3, -TCRβ, -CD4, -CD8, -B220, -TCRγ, -MAC-1, -NK1.1, -GR.1, TER-119 and –CD11c (all biotinylated and detected with strepavidin-PerCP), PE-anti-CD28 and APC-anti-CD25 followed by intracellular staining with FITC anti-TCRβ. PerCP-negative, CD25+ cells were analyzed for expression of CD28 and intracellular TCRβ. Histograms show CD28 and intracellular TCRβ staining on TCRβneg(gray) and TCRβhi (black) cells. B. Increased expression of CD28 induced on Rag-2 KO thymocytes by in vivo anti-CD3 treatment. 12-week old Rag-2 KO mice were injected with 300 μg of anti-CD3 (2C11) i.p. Thymocytes were labeled with FITC anti-CD8, APC anti-CD4 and PE anti-CD28. Histograms in the left panel show CD28 staining on CD4−CD8− DN thymocytes from a Rag-2 KO 8 days after injection of anti-CD3 (solid black line), a control Rag-2 KO (dotted black line) and a wildtype B6 (gray filled). Staining with an isotype-matched PE control antibody is shown for each mouse (dashed, stippled and solid gray lines). Histograms in the right panel show CD28 staining on CD4+CD8+ DP thymocytes from a Rag-2 KO 8 days after injection of anti-CD3 (solid black line) and a control B6 (solid gray line). Staining with an isotype-matched PE control antibody is shown for each mouse (dashed and solid gray lines).
The pattern of B7-2 expression was assessed in wildtype B6 mice and, as has been described previously (1), revealed that B7-2 staining is observed in both the thymic cortex and medulla, though staining is substantially more intense in the medulla (see Fig. 3). Taken together with the finding that CD28 levels are low in the earliest stages of DN thymocyte development, the paucity of B7-2+ cells in the cortex where DN thymocyte maturation occurs suggests that regulated expression of CD28 and B7 ensures that costimulatory signaling does not occur in early developing thymocytes.
Figure 3.
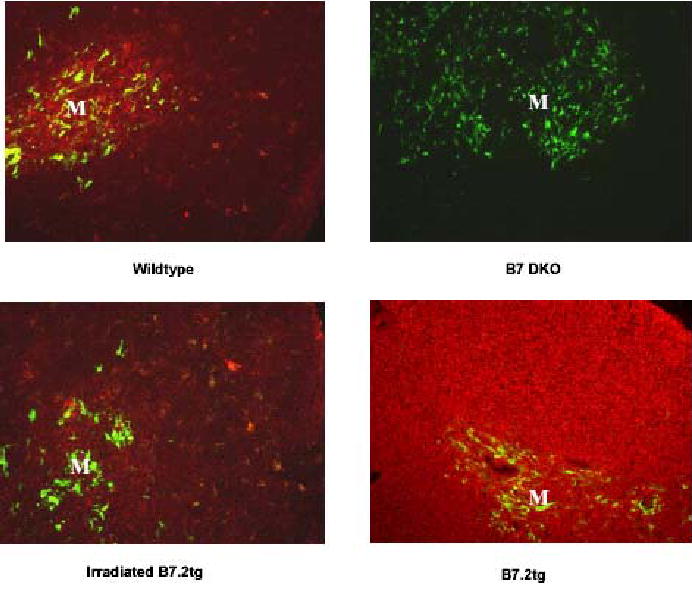
Thymic B7-2 expression patterns in wildtype, B7-1/B7-2 knockout, irradiated B7-2 tg and B7-2 tg mice. Sections of frozen thymus tissue from 7–14 week old mice or from an irradiated B7-2 tg mouse 7–14 days after reconstitution with CD28.FL tg bone marrow were stained with anti-B7-2. Medullary regions of the thymus are identified by staining with anti-keratin 14 and are identified in the figure with an “M”. B7-2 reactivity was detected with Texas Red conjugated anti-Ig and keratin 14 reactivity was detected with FITC conjugated anti Ig as described in Materials and Methods
Generating CD28 FL/B7-2 double transgenic mice
To determine whether altered expression of CD28 and B7-2 would have effects on thymic differentiation we studied B7-2 Line 7 mice (13), which express B7-2 under the control of the H-2Kb promoter and Ig μ-chain enhancer, and mice expressing a full length CD28 (CD28.FL) under control of the human CD2 promoter/enhancer (5), as well as CD28.FL/B7-2 double transgenic mice. In contrast to wildtype mice in which staining for B7-2 is more intense in the medulla, B7-2 in Line 7 transgenic mice is homogeneously expressed at high levels throughout the thymus (Fig.3), and is expressed on all thymocyte subpopulations as assessed by flow cytometry (data not shown). The CD2 promoter used to drive expression of the CD28 transgene restricts CD28 expression to T cells. However, CD28 expression levels are dramatically increased at all stages of thymocyte development relative to wildtype mice. More specifically, the increase in CD28 expression which occurs as thymocytes progress from the DN3 to DN4 stage in wildtype mice is no longer apparent in the CD28.FL transgenics which express high levels of CD28 as early as the DN2 stage and maintain high levels throughout thymocyte development (Fig. 4A and B).
Figure 4.
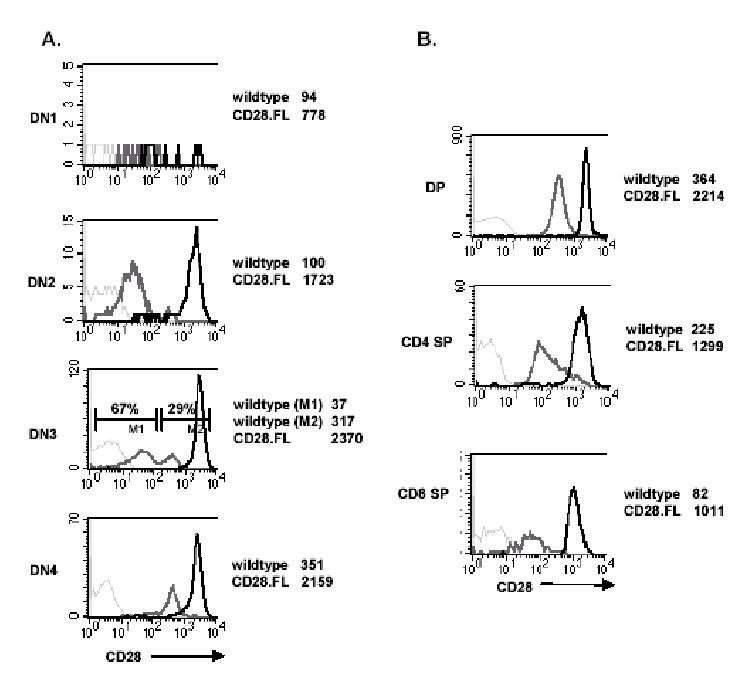
CD28 expression on thymocytes from CD28.FL transgenic mice. A. Thymocytes were labeled as described in Fig. 1B. Histograms show CD28 staining on CD44+CD25− (DN1), CD44+CD25+ (DN2), CD44−CD25+ (DN3) and CD44−CD25− (DN4) thymocytes from wildtype (solid gray line) and CD28.FL (solid black line) mice. Staining with an isotype-matched PE control antibody is shown for the CD28.FL thymocytes (dashed gray lines). For CD28 staining, the mean fluorescent intensity (MFI) of wildtype and CD28.FL transgenic thymocytes is indicated to the right of each figure. B. Thymocytes from wildtype and CD28.FL transgenic mice were stained with biotin-conjugated anti-CD4, FITC anti-CD8 and PE anti-CD28. Histograms show CD28 staining on CD4+CD8+ DP, CD4+SP and CD8+SP thymocytes from wildtype (solid gray line) and CD28.FL (solid black line) mice. The MFI for CD28-stained cells are indicated to the right of each figure. Staining with an isotype-matched PE control antibody is shown for the CD28.FL thymocytes (dashed gray lines).
Analysis of thymocyte subpopulations from CD28.FL/B7-2 double transgenics revealed a significant reduction in the numbers of SP CD4 and CD8 thymocytes and an increase in the numbers of DN thymocytes compared to wildtype littermates (Fig. 5 and 6A). Similar alterations in thymocyte profiles were observed in crosses of the B7-2 transgenic mice to two additional independently derived CD28 transgenic lines (data not shown). In contrast, thymocyte profiles of either B7-2 Line 7 or CD28.FL single transgenics were similar to those of wildtype mice, consistent with the need to introduce altered expression of both CD28 and its ligand in order to alter thymocyte development.
Figure 5.

Altered thymocyte profile in CD28.FL/B7-2 double transgenic mice. Thymocytes from wildtype, CD28.FL, B7-2 Line 7, and CD28.FL/B7-2, double transgenic mice were stained with PE anti-CD4 and FITC anti-CD8 mAb. Results are representative of four independent experiments.
Figure 6.

Altered frequency of thymocyte subpopulations in CD28.FL/B7-2 double transgenic mice. A. Analysis of thymus cellularity and frequency of DN, DP, CD4 SP, and CD8 SP thymocytes in wildtype (striped), B7.2 tg (white), CD28.WT tg (gray) and CD28.FL/B7.2 double transgenic mice (black). Data shown are derived from analysis of 3–5 mice per group and represent the mean ± s.e.m. B. Increased proportion of DN 4 thymocytes in CD28.FL/B7.2 transgenic mice. Thymocytes from wildtype, B7 tg, CD28.FL tg, and CD28.FL/B7.2 double transgenic mice were labeled with anti-CD3, -TCRβ, -CD4, -CD8, -B220, -TCRγ, -MAC-1, -NK1.1, -GR.1, TER-119 and –CD11c (all biotinylated and detected with strepavidin-PerCP), PE anti-CD25, and APC anti-CD44. PerCP-negative cells were analyzed for expression of CD25 versus CD44.
To determine whether the increase in DN thymocytes observed in the CD28.FL/B7.2 mice reflects an expansion of a particular subset of DN cells, we examined the DN1 through DN4 subset distribution in these mice. Figure 6B illustrates that there is a significant increase in the proportion of DN 4 thymocytes in the CD28.FL/B7.2 transgenic relative to that observed in the wildtype control thymus (87% vs. 24%), with a reciprocal decrease in the proportion of DN3 thymocytes (9% vs. 66%).
The majority of CD28.FL/B7-2 DP thymocytes do not express TCRβ
We considered the possibility that the reduction in CD4 and CD8 SP thymocytes observed in CD28.FL/B7-2 double transgenics might derive from abnormalities in early thymic development related to expression of transgenic CD28. Staining for TCRβ demonstrated that a substantial proportion of DP thymocytes from CD28.FL/B7-2 mice express no detectable cell surface TCRβ, in contrast to expression of TCRβ on essentially all DP thymocytes from wildtype mice (Fig. 7A). To determine whether the reduction in DP thymocytes expressing cell-surface TCRβ requires an intact CD28 cytoplasmic domain, B7-2 Line 7 transgenics were crossed with mice expressing a CD28 truncation mutant lacking the cytoplasmic sequence (CD28.TM) (5) or with mice expressing a CD28 Y189→F mutant (CD28.Y189F) (5) which no longer binds PI3K and Grb-2 (14–16). Expression of transgenic CD28 in the CD28.FL, CD28.TM and CD28.Y189F lines is comparable ((5) and data not shown). In contrast to the results from CD28.FL/B7.2 transgenics, cell-surface TCRβ is expressed on essentially all DP thymocytes from CD28.TM/B7-2 and CD28.Y189F/B7-2 mice (Fig. 7A). This finding suggests that the presence of cell-surface TCRβneg DP thymocytes in mice expressing both CD28 and B7.2 transgenes requires CD28 signaling through the CD28 cytoplasmic domain.
Figure 7.

The majority of CD28.FL/B7-2 DP thymocytes do not express TCRβ. A. Thymocytes from wildtype, CD28.FL/B7-2, CD28.TM/B7-2, and CD28.Y198F/B7.2 double transgenic mice were surface stained with antibodies specific for CD4, CD8 and TCRβ. TCRβ expression on CD4+CD8+ gated cells is shown. The gray histogram shows staining with an isotype-matched biotinylated control antibody. Results are representative of four independent experiments. B. Thymocytes from wild type, TCRβ KO, and CD28.FL/B7-2 double transgenic mice were stained with antibodies specific for CD4 and CD8 followed by intracellular staining for TCRβ. Total TCRβ expression in CD4+CD8+ gated cells is shown. Results are representative of three independent experiments.
During normal thymic development, thymocytes transiting from the DN to DP stage are interrogated at the β-checkpoint. Only cells that have rearranged and can express TCRβ proceed through this checkpoint to become DP thymocytes. Thus, essentially all wildtype DP thymocytes should stain positive for intracellular TCRβ. Fig. 7B compares total (cell surface + intracellular) TCRβ staining for wildtype, TCRβ KO and CD28.FL/B7-2 DP thymocytes. The small numbers of DP thymocytes present in the TCRβ KOs derive from TCRγδ-driven DP development (17). Surprisingly, in contrast to wildtype thymocytes, the majority of DP thymocytes from CD28.FL/B7-2 mice lack expression of surface or intracellular TCRβ. This suggests that differentiation to the DP stage of development in these mice might be occurring independent of the requirement for TCRβ expression that is manifest in wildtype thymocytes.
CD28.FL/B7-2 transgenes rescue development of DP thymocytes in Rag-2 and CD3ɛ KO mice
The presence of CD28.FL/B7-2 DP thymocytes lacking detectable expression of TCRβ led us to hypothesize that CD28-B7-2 interactions may permit cells to transit from the DN to DP stage in the absence of TCRβ. We tested this hypothesis by generating CD28.FL/B7-2 double transgenic mice on a Rag-2 KO background and examining these mice for the presence of DP thymocytes. As shown in Fig. 8A, Rag-2 KO mice have a profound defect in thymic development, with extremely low total numbers of thymocytes, consisting exclusively of immature DN cells. Notably, thymic cellularity in CD28.FL/B7-2 Rag-2 KO mice is increased approximately 100-fold, to wildtype numbers, and thymic differentiation to the DP stage occurs efficiently (86.1 ± 0.9% DP in wildtype and 85.5 ± 4% DP in CD28.FL/B7-2 Rag-2 KO, versus <1% in Rag-2 KO), though differentiation to the CD4 or CD8 SP stage was not observed. As demonstrated above (Fig. 2B), it is known that Rag-2 KO thymocytes can differentiate to the DP stage when treated with anti-CD3ɛ, indicating that signaling through a CD3ɛ- containing receptor can drive differentiation in the absence of TCRα or TCRβ (11, 12). We therefore considered the possibility that the rescue of thymic differentiation we observed in the CD28.FL/B7-2 Rag-2 KO mice might require intact CD3 signaling machinery. To test whether this was the case, we generated CD28.FL/B7-2 mice on a CD3ɛ KO background. As shown in Fig. 8B, similar to what was observed in CD28.FL/B7-2 Rag-2 KO mice, thymic cellularity in CD28.FL/B7-2 CD3ɛ KO mice is recovered to wild type numbers, and thymic differentiation to the DP stage occurs efficiently (86.1 ± 0.9% DP in wildtype and 78.7 ± 0.9% DP in CD28.FL/B7-2 CD3ɛ KO, versus <1% in CD3ɛ KO). Thus, CD28-B7-2 interactions appear able to direct thymic differentiation from the DN to DP stage in the absence of contributions from known components of the pre-TCR.
Figure 8.

CD28.FL/B7-2 transgenes rescue development to the DP stage in Rag-2 and CD3ɛ KO mice. A. CD8 versus CD4 thymic profiles from Rag-2 KO and CD28.FL/B7-2 double transgenic mice on a Rag-2 KO background. B. CD8 versus CD4 thymic profiles from CD3ɛ KO and CD28.FL/B7-2 double transgenic mice on a CD3ɛ KO background. Total thymocyte numbers are indicated for each mouse. Results are representative of three independent experiments for each KO background.
Transgenic CD28 expressed by thymocytes interacts with B7-2 expressed by radiation-resistant thymic elements to promote DN to DP differentiation
To probe further the nature of interactions required for promoting the pre-TCR-independent DN to DP transition in the presence of B7-2 and CD28 FL transgenes, we generated chimeric mice in which developing thymocytes expressed the CD28.FL transgene, while expression of the B7-2 transgene was restricted to radioresistant host cells (CD28.FL tg donor→B7.2 tg host). Fig. 3A shows B7-2 expression patterns in thymuses from irradiated and non-irradiated B7-2 transgenic mice contrasted with those of wildtype and B7 KOs (lacking both B7-1 and B7-2). Levels of B7-2 are dramatically reduced in the irradiated versus non-irradiated B7-2 transgenic thymus but differ from those found in the wildtype by displaying similar intensity of staining in both the cortex and medulla. To determine if the radiation-resistant B7-2 expression in B7-2 tg hosts would be sufficient to promote the TCRβ-independent DN to DP differentiation observed in the CD28.FL/B7-2 double transgenics, where B7-2 and CD28 transgenes are expressed by the same cells, radiation bone marrow chimeras were generated by reconstituting lethally irradiated transgenic B7-2 transgenic mice with bone marrow cells from CD28.FL transgenic mice. Both donor and recipient mice were CD3ɛ KOs, thus permitting the most sensitive assessment of pre-TCR-independent progression from DN to DP stage of thymic differentiation. Thymic differentiation to the DP stage occurred efficiently in CD28 FL/CD3ɛ KO → B7-2 Line 7/CD3ɛ KO chimeras (Fig. 9A), and thymic cellularity was substantially increased over that observed in CD3ɛ KO → CD3ɛ KO control chimeras. These findings make two critical points: first, that the CD28.FL and B7.2 transgenes need not be expressed on the same cell to promote aberrant thymocyte differentiation, and, second, that transgenic B7-2 expressed by radiation-resistant host cells is sufficient to perturb thymic differentiation in the presence of the CD28.FL transgene.
Figure 9.

A. Development to the DP stage is rescued in CD28.FL/CD3ɛ KO→B7-2/CD3ɛ KO chimeras. Chimeras were generated by reconstituting B7-2/CD3e KO mice with CD28.FL/CD3ɛ KO bone marrow. Controls chimeras were generated by reconstituting CD3ɛ KO mice with CD3ɛ KO bone marrow. Results are representative of two independent experiments. B. Development to the DP stage in CD28.FL/CD3ɛ KO mice. Results are representative of 4 independent experiments.
Although expression of the B7.2 transgene in CD28 FL/CD3ɛ KO → B7-2 Line 7/CD3ɛ KO chimeras is restricted relative to that observed in non-irradiated B7.2 transgenic animals, it differs from the wildtype expression pattern in that similar levels of B7.2 are observed in the cortex and medulla. To determine whether endogenous wild-type expression of B7 is sufficient to promote TCRβ-independent differentiation of thymocytes expressing the CD28.FL tg, we assessed thymocyte subpopulations in CD28.FL/CD3ɛ KO mice. As shown in Figure 9B, significant rescue of DP differentiation is observed in the CD28.FL/CD3ɛ KO mice expressing only endogenous B7 molecules. The average percentage of DP thymocytes in CD28.FL/CD3ɛ KO mice was 10.2 ± 1% (n=6) compared to 0.05 ± 0.02% (n=5) in littermate CD3ɛ KO control mice, with minimal increase in total thymocyte numbers (2.1 × 106 ± 0.3 × 106 in CD28.FL/CD3ɛ KO mice; 1.0 × 106 ± 0.1 × 106 in CD3ɛ KO control mice). While the recovery of DP thymocytes in the CD28.FL/CD3ɛ KO mice was significantly less than that observed in either the CD28.FL/B7-2 CD3ɛ KO mice or the CD28 FL/CD3ɛ KO → B7-2 Line 7/CD3ɛ KO chimeras, these findings demonstrate that elevated CD28 expression can drive aberrant thymocyte development in the presence of a spectrum of B7 expression levels ranging from overt overexpression of the B7.2 transgene to physiologic endogenous B7 levels, and further highlight the importance of finely regulating CD28-B7 interactions during thymic development.
Discussion
In the normal thymus, expression of both CD28 and B7-2 is notably low in the thymic compartments in which the earliest stages of thymic differentiation occur. This finding led us to hypothesize that regulated expression of the costimulatory molecules CD28 and B7, by specifically preventing costimulatory signaling of immature early DN thymocytes, might be important for normal thymic development. We have demonstrated that CD28/B7 signaling of immature thymocytes can, surprisingly, result in differentiation from the DN to DP stage of T cell development in the absence of pre-TCR signaling. This pre-TCR-independent differentiation is an abortive pathway which generates DP thymocytes that lack TCR and are unable to progress to the SP stage of development, similar to the recently described TCRαγ-driven abortive pathway in TCRα transgenic mice (18). These findings illustrate that finely regulated physiologic expression of thymic CD28 and B7 may be critical in ensuring that thymocyte development does not proceed via an abortive costimulatory signaling pathway independent of TCR.
Interestingly, a recent report from Zheng et al. (2) provides evidence that endogenous B7-CD28 interactions are critical to the later stages of DN thymocyte differentiation. They found an increase in the proportion of DN4 thymocytes in CD28 or B7 deficient mice which they argue derives, in part, from a more rapid DN3 to DN4 transition coupled with a delayed DN4 to DP transition. Zheng et al. conclude that the accelerated DN3 to DN4 transition is the result of elevated RAG expression in DN3 cells, which promotes TCRβ rearrangements and allows for an increased transition through the β-checkpoint. Paradoxically, we also observe an increase in DN4 thymocytes in CD28.FL/B7.2 double transgenics. However, expression of TCRβ in these DN4 thymocytes is dramatically decreased relative to wildtype (<1% vs. 81%; data not shown). We propose that this is the result of elevated CD28 expression levels prior to the β-checkpoint, thus allowing CD28-driven development to proceed independent of the pre-TCR. Taken together, these results demonstrate that CD28-B7 interactions have the potential to profoundly affect the DN to DP differentiation step during thymocyte development, and thus regulated expression of these molecules is critical to ensure normal thymic development.
The pre-TCR-independent DN to DP maturation observed in CD28.FL/B7-2 double transgenics is, to our knowledge, the first indication that a costimulatory signal can substitute for signal generated by the pre-TCR to drive T cell differentiation. These findings suggest that CD28 is able to initiate and propagate functionally effective signals independent of signals generated by the pre-TCR, consistent with several recent reports suggesting that engagement of CD28 can activate discrete signaling pathways in mature T cells (19–21). Interestingly, however, signaling through CD28 was not sufficient to promote TCR-independent signaling in the context of thymocyte development as evidenced by the failure to observe development of CD4 SP or CD8 SP thymocytes in CD28.FL/B7-2 Rag-2 KO or CD28.FL/B7-2 CD3ɛ KO mice. Furthermore, the decrease in CD4 SP and CD8 SP thymocytes observed in CD28.FL/B7.2 double transgenic mice on a wildtype background is likely due, at least in part, to the failure of TRCβ-negative DP thymocytes to further differentiate.
The intracellular portion of the CD28 molecule lacks known direct enzymatic activity but has several conserved motifs which mediate association with a number of molecules known to be involved in signal transduction. In particular, in mature T cells, phosphorylation of tyrosine-170 in the cytoplasmic YMNM motif of CD28 has been shown to promote binding to PI3K and Grb-2 (14–16), while a cytoplasmic PXXP motif has been demonstrated to be critical for activation of Lck (22). The profound effect of CD28-B7 interactions on thymocyte DN to DP differentiation observed in our CD28.FL/B7-2 double transgenics provides a platform for examining the roles of individual CD28 motifs. We have demonstrated that the CD28.TM/B7-2 double transgenic is not effective in driving a DN to DP transition in the absence of pre-TCR signaling, thereby establishing a requirement for the cytoplasmic domain of the CD28 molecule in promoting this transition. To examine in greater detail the requirement for the CD28 cytoplasmic tail, we assessed the capacity of the CD28.Y189F mutant (note that Y170 and Y189 are used interchangeably in the literature to refer to the same CD28 tyrosine residue; the Y189 designation reflects enumeration that includes the signal peptide), in which the YMNM motif required for PI3K and Grb2 association is disrupted, to promote the DN to DP transition. CD28.Y189F/B7-2 double transgenics were also unable to support TCRβ-independent DN to DP differentiation. This finding suggests that associations between CD28 and PI3K and/or Grb2 may be critical to the process of CD28-driven DN to DP differentiation. Intriguing in this regard is the recent report that inactivation of PTEN, a phosphatase that regulates signaling mediated by PI3K, allows thymic development to bypass requirements for pre-TCR signaling (23). It is of interest that the YMNM motif is also required for induction of Bcl-xL in response to CD28 engagement in peripheral T cells (24, 25), and that Bcl-xL is implicated in the survival of DP thymocytes (26, 27), raising the possibility that induction of Bcl-xL is involved in the CD28-driven DN to DP transition observed in the present studies. It remains to be determined whether other CD28 cytoplasmic domains, such as the lck-binding PXXP motif, also play a role in this differentiative stimulus. In this regard, it is of interest that a constitutively active lck transgene has been demonstrated to drive the DN to DP transition in the absence of pre-TCR (28, 29). Manipulations of several other intracellular signaling pathways including the Wnt-β-catenin and Ras-Raf-MAPK pathways (30–32) as well as of cell survival pathways involving p53 and FADD (33, 34) have also allowed the DN to DP transition to proceed in the absence of a TCR signal. Whether or not the CD28 signals which permit pre-TCR–independent DN to DP transition to occur involve these pathways is yet to be determined.
In peripheral T cells, engagement of the TCR alone is not sufficient to promote full activation of the responding T cell, full activation requiring the participation of coreceptor molecules CD4 or CD8 and costimulatory molecules such as CD28 (reviewed in (35–37)). It has been suggested that the threshold for activation/differentiation in DN thymocytes may be lower than for more mature T cells (38). In this regard it is perhaps significant that normal pre-TCR-dependent differentiation of DN T cells occurs in the absence of CD4 and CD8, and that CD28 expression is initiated at low levels as immature thymocytes traverse through the DN1 to DN3 stages of development, only increasing once DN cells have rearranged and expressed TCRβ (1). In addition, expression of the CD28 ligands, B7-1 and B7-2, is limited in the thymic cortex (3, 4), thus reducing the likelihood that CD28 on immature DN thymocytes would be engaged. The potential importance of limiting coreceptor and costimulatory signaling events during pre-TCR-dependent differentiation is demonstrated by the finding that ectopic expression of transgenic CD4 (39) and now of CD28 and B7 (this report) results in differentiation from the DN to DP stage in the absence of TCRβ. Moreover, it is notable that the ability of CD28 engagement to induce differentiation of CD3ɛ-deficient cells to the DP stage is, to our knowledge, a unique instance of T cell differentiation independent of these core components of the TCR and pre-TCR receptors. Thus, the signals generated by the pre-TCR during thymic development may not be unique in their ability to drive thymic differentiation. Rather, the physiologic requirement for a pre-TCR “β checkpoint” is dependent upon preventing activation of alternative pre-TCR-independent pathways capable of driving competing and non-productive pathways of T cell development. The finely regulated physiologic expression of CD28 and B7 within thymic compartments may be critical to ensuring that T cell differentiation is driven by pre-TCR and TCR signals, and not through an alternative and abortive pathway resulting from costimulatory signaling in the absence of TCR.
Acknowledgments
We thank Xuguang Tai and Alfred Singer for generously providing additional lines of transgenic mice expressing full length CD28, Anthony Adams for expert assistance with FACS analysis, and Genevieve Sanchez-Howard and staff at Bioqual for excellent animal care and husbandry. We would like to give special thanks to Alfred Singer and Avinash Bhandoola for helpful discussions and suggestions throughout the course of this work. We also thank Jeffrey Bluestone, Remy Bosselut, and Melanie Vacchio for comments and critical reading of the manuscript.
References
- 1.Gross JA, Callas E, Allison JP. Identification and distribution of the costimulatory receptor CD28 in the mouse. J Immunol. 1992;149:380. [PubMed] [Google Scholar]
- 2.Zheng X, Gao JX, Chang X, Wang Y, Liu Y, Wen J, Zhang H, Zhang J, Zheng P. B7-CD28 interaction promotes proliferation and survival but suppresses differentiation of CD4-CD8- T cells in the thymus. J Immunol. 2004;173:2253. doi: 10.4049/jimmunol.173.4.2253. [DOI] [PubMed] [Google Scholar]
- 3.Inaba K, Inaba M, Witmer-Pack M, Hatchcock K, Hodes R, Steinman RM. Expression of B7 costimulator molecules on mouse dendritic cells. Adv Exp Med Biol. 1995;378:65. doi: 10.1007/978-1-4615-1971-3_13. [DOI] [PubMed] [Google Scholar]
- 4.Nelson AJ, Hosier S, Brady W, Linsley PS, Farr AG. Medullary thymic epithelium expresses a ligand for CTLA4 in situ and in vitro. J Immunol. 1993;151:2453. [PubMed] [Google Scholar]
- 5.Harada Y, Tokushima M, Matsumoto Y, Ogawa S, Otsuka M, Hayashi K, Weiss BD, June CH, Abe R. Critical requirement for the membrane-proximal cytosolic tyrosine residue for CD28-mediated costimulation in vivo. J Immunol. 2001;166:3797. doi: 10.4049/jimmunol.166.6.3797. [DOI] [PubMed] [Google Scholar]
- 6.Mombaerts P, Clarke AR, Rudnicki MA, Iacomini J, Itohara S, Lafaille JJ, Wang L, Ichikawa Y, Jaenisch R, Hooper ML, et al. Mutations in T-cell antigen receptor genes alpha and beta block thymocyte development at different stages. Nature. 1992;360:225. doi: 10.1038/360225a0. [DOI] [PubMed] [Google Scholar]
- 7.DeJarnette JB, Sommers CL, Huang K, Woodside KJ, Emmons R, Katz K, Shores EW, Love PE. Specific requirement for CD3epsilon in T cell development. Proc Natl Acad Sci U S A. 1998;95:14909. doi: 10.1073/pnas.95.25.14909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shinkai Y, Rathbun G, Lam KP, Oltz EM, Stewart V, Mendelsohn M, Charron J, Datta M, Young F, Stall AM, et al. RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell. 1992;68:855. doi: 10.1016/0092-8674(92)90029-c. [DOI] [PubMed] [Google Scholar]
- 9.Hathcock KS, Laszlo G, Dickler HB, Bradshaw J, Linsley P, Hodes RJ. Identification of an alternative CTLA-4 ligand costimulatory for T cell activation. Science. 1993;262:905. doi: 10.1126/science.7694361. [DOI] [PubMed] [Google Scholar]
- 10.Singer A, Hathcock KS, Hodes RJ. Cellular and genetic control of antibody responses. V. Helper T-cell recognition of H-2 determinants on accessory cells but not B cells. J Exp Med. 1979;149:1208. doi: 10.1084/jem.149.5.1208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Levelt CN, Mombaerts P, Iglesias A, Tonegawa S, Eichmann K. Restoration of early thymocyte differentiation in T-cell receptor beta-chain-deficient mutant mice by transmembrane signaling through CD3 epsilon. Proc Natl Acad Sci U S A. 1993;90:11401. doi: 10.1073/pnas.90.23.11401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shinkai Y, Alt FW. CD3 epsilon-mediated signals rescue the development of CD4+CD8+ thymocytes in RAG-2−/− mice in the absence of TCR beta chain expression. Int Immunol. 1994;6:995. doi: 10.1093/intimm/6.7.995. [DOI] [PubMed] [Google Scholar]
- 13.Fournier S, Rathmell JC, Goodnow CC, Allison JP. T cell-mediated elimination of B7.2 transgenic B cells. Immunity. 1997;6:327. doi: 10.1016/s1074-7613(00)80335-0. [DOI] [PubMed] [Google Scholar]
- 14.Cai YC, Cefai D, Schneider H, Raab M, Nabavi N, Rudd CE. Selective CD28pYMNM mutations implicate phosphatidylinositol 3-kinase in CD86-CD28-mediated costimulation. Immunity. 1995;3:417. doi: 10.1016/1074-7613(95)90171-x. [DOI] [PubMed] [Google Scholar]
- 15.Pages F, Ragueneau M, Rottapel R, Truneh A, Nunes J, Imbert J, Olive D. Binding of phosphatidylinositol-3-OH kinase to CD28 is required for T-cell signalling. Nature. 1994;369:327. doi: 10.1038/369327a0. [DOI] [PubMed] [Google Scholar]
- 16.Prasad KV, Cai YC, Raab M, Duckworth B, Cantley L, Shoelson SE, Rudd CE. T-cell antigen CD28 interacts with the lipid kinase phosphatidylinositol 3-kinase by a cytoplasmic Tyr(P)-Met-Xaa-Met motif. Proc Natl Acad Sci U S A. 1994;91:2834. doi: 10.1073/pnas.91.7.2834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Passoni L, Hoffman ES, Kim S, Crompton T, Pao W, Dong MQ, Owen MJ, Hayday AC. Intrathymic delta selection events in gammadelta cell development. Immunity. 1997;7:83. doi: 10.1016/s1074-7613(00)80512-9. [DOI] [PubMed] [Google Scholar]
- 18.Erman B, Feigenbaum L, Coligan JE, Singer A. Early TCRalpha expression generates TCRalphagamma complexes that signal the DN-to-DP transition and impair development. Nat Immunol. 2002;3:564. doi: 10.1038/ni800. [DOI] [PubMed] [Google Scholar]
- 19.Raab M, Pfister S, Rudd CE. CD28 signaling via VAV/SLP-76 adaptors: regulation of cytokine transcription independent of TCR ligation. Immunity. 2001;15:921. doi: 10.1016/s1074-7613(01)00248-5. [DOI] [PubMed] [Google Scholar]
- 20.Tacke M, Hanke G, Hanke T, Hunig T. CD28-mediated induction of proliferation in resting T cells in vitro and in vivo without engagement of the T cell receptor: evidence for functionally distinct forms of CD28. Eur J Immunol. 1997;27:239. doi: 10.1002/eji.1830270136. [DOI] [PubMed] [Google Scholar]
- 21.Piccolella E, Spadaro F, Ramoni C, Marinari B, Costanzo A, Levrero M, Thomson L, Abraham RT, Tuosto L. Vav-1 and the IKK alpha subunit of I kappa B kinase functionally associate to induce NF-kappa B activation in response to CD28 engagement. J Immunol. 2003;170:2895. doi: 10.4049/jimmunol.170.6.2895. [DOI] [PubMed] [Google Scholar]
- 22.Holdorf AD, Green JM, Levin SD, Denny MF, Straus DB, Link V, Changelian PS, Allen PM, Shaw AS. Proline residues in CD28 and the Src homology (SH)3 domain of Lck are required for T cell costimulation. J Exp Med. 1999;190:375. doi: 10.1084/jem.190.3.375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hagenbeek TJ, Naspetti M, Malergue F, Garcon F, Nunes JA, Cleutjens KB, Trapman J, Krimpenfort P, Spits H. The loss of PTEN allows TCR alphabeta lineage thymocytes to bypass IL-7 and Pre-TCR-mediated signaling. J Exp Med. 2004;200:883. doi: 10.1084/jem.20040495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Burr JS, Savage ND, Messah GE, Kimzey SL, Shaw AS, Arch RH, Green JM. Cutting edge: distinct motifs within CD28 regulate T cell proliferation and induction of Bcl-XL. J Immunol. 2001;166:5331. doi: 10.4049/jimmunol.166.9.5331. [DOI] [PubMed] [Google Scholar]
- 25.Okkenhaug K, Wu L, Garza KM, La Rose J, Khoo W, Odermatt B, Mak TW, Ohashi PS, Rottapel R. A point mutation in CD28 distinguishes proliferative signals from survival signals. Nat Immunol. 2001;2:325. doi: 10.1038/86327. [DOI] [PubMed] [Google Scholar]
- 26.Ma A, Pena JC, Chang B, Margosian E, Davidson L, Alt FW, Thompson CB. Bclx regulates the survival of double-positive thymocytes. Proc Natl Acad Sci U S A. 1995;92:4763. doi: 10.1073/pnas.92.11.4763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Grillot DA, Merino R, Nunez G. Bcl-XL displays restricted distribution during T cell development and inhibits multiple forms of apoptosis but not clonal deletion in transgenic mice. J Exp Med. 1995;182:1973. doi: 10.1084/jem.182.6.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mombaerts P, Anderson SJ, Perlmutter RM, Mak TW, Tonegawa S. An activated lck transgene promotes thymocyte development in RAG-1 mutant mice. Immunity. 1994;1:261. doi: 10.1016/1074-7613(94)90077-9. [DOI] [PubMed] [Google Scholar]
- 29.Fehling HJ, Iritani BM, Krotkova A, Forbush KA, Laplace C, Perlmutter RM, von Boehmer H. Restoration of thymopoiesis in pT alpha-/- mice by anti-CD3epsilon antibody treatment or with transgenes encoding activated Lck or tailless pT alpha. Immunity. 1997;6:703. doi: 10.1016/s1074-7613(00)80446-x. [DOI] [PubMed] [Google Scholar]
- 30.Gounari F, Aifantis I, Khazaie K, Hoeflinger S, Harada N, Taketo MM, von Boehmer H. Somatic activation of beta-catenin bypasses pre-TCR signaling and TCR selection in thymocyte development. Nat Immunol. 2001;2:863. doi: 10.1038/ni0901-863. [DOI] [PubMed] [Google Scholar]
- 31.Gartner F, Alt FW, Monroe R, Chu M, Sleckman BP, Davidson L, Swat W. Immature thymocytes employ distinct signaling pathways for allelic exclusion versus differentiation and expansion. Immunity. 1999;10:537. doi: 10.1016/s1074-7613(00)80053-9. [DOI] [PubMed] [Google Scholar]
- 32.Iritani BM, Alberola-Ila J, Forbush KA, Perimutter RM. Distinct signals mediate maturation and allelic exclusion in lymphocyte progenitors. Immunity. 1999;10:713. doi: 10.1016/s1074-7613(00)80070-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Haks MC, Krimpenfort P, van den Brakel JH, Kruisbeek AM. Pre-TCR signaling and inactivation of p53 induces crucial cell survival pathways in pre-T cells. Immunity. 1999;11:91. doi: 10.1016/s1074-7613(00)80084-9. [DOI] [PubMed] [Google Scholar]
- 34.Newton K, Harris AW, Strasser A. FADD/MORT1 regulates the pre-TCR checkpoint and can function as a tumour suppressor. Embo J. 2000;19:931. doi: 10.1093/emboj/19.5.931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Alegre ML, Frauwirth KA, Thompson CB. T-cell regulation by CD28 and CTLA-4. Nat Rev Immunol. 2001;1:220. doi: 10.1038/35105024. [DOI] [PubMed] [Google Scholar]
- 36.Sharpe AH, Freeman GJ. The B7-CD28 superfamily. Nat Rev Immunol. 2002;2:116. doi: 10.1038/nri727. [DOI] [PubMed] [Google Scholar]
- 37.Salomon B, Bluestone JA. Complexities of CD28/B7: CTLA-4 costimulatory pathways in autoimmunity and transplantation. Annu Rev Immunol. 2001;19:225. doi: 10.1146/annurev.immunol.19.1.225. [DOI] [PubMed] [Google Scholar]
- 38.Haks MC, Belkowski SM, Ciofani M, Rhodes M, Lefebvre JM, Trop S, Hugo P, Zuniga-Pflucker JC, Wiest DL. Low activation threshold as a mechanism for ligand-independent signaling in pre-T cells. J Immunol. 2003;170:2853. doi: 10.4049/jimmunol.170.6.2853. [DOI] [PubMed] [Google Scholar]
- 39.Norment AM, Forbush KA, Nguyen N, Malissen M, Perlmutter RM. Replacement of pre-T cell receptor signaling functions by the CD4 coreceptor. J Exp Med. 1997;185:121. doi: 10.1084/jem.185.1.121. [DOI] [PMC free article] [PubMed] [Google Scholar]


