Abstract
Oxidative stress, driven by reactive oxygen, nitrogen, and sulphur species (ROS, RNS, RSS), poses a significant threat to cellular integrity and human health. Generated during mitochondrial respiration, inflammation, UV exposure and pollution, these species damage cells and contribute to pathologies like cardiovascular issues, neurodegeneration, cancer, and metabolic syndromes. Lifestyle factors exert a substantial influence on oxidative stress levels, with mitochondria emerging as pivotal players in ROS generation and cellular equilibrium. Phytochemicals, abundant in plants, such as carotenoids, ascorbic acid, tocopherols and polyphenols, offer diverse antioxidant mechanisms. They scavenge free radicals, chelate metal ions, and modulate cellular signalling pathways to mitigate oxidative damage. Furthermore, plants thriving in high-altitude regions are adapted to extreme conditions, and synthesize secondary metabolites, like flavonoids and phenolic compounds in bulk quantities, which act to form a robust antioxidant defence against oxidative stress, including UV radiation and temperature fluctuations. These plants are promising sources for drug development, offering innovative strategies by which to manage oxidative stress-related ailments and enhance human health. Understanding and harnessing the antioxidant potential of phytochemicals from high-altitude plants represent crucial steps in combating oxidative stress-induced disorders and promoting overall wellbeing. This study offers a comprehensive summary of the production and physio-pathological aspects of lifestyle-induced oxidative stress disorders and explores the potential of phytochemicals as promising antioxidants. Additionally, it presents an appraisal of high-altitude medicinal plants as significant sources of antioxidants, highlighting their potential for drug development and the creation of innovative antioxidant therapeutic approaches.
Keywords: antioxidant, high-altitude medicinal plants, lifestyle-associated disorders, oxidative stress, phytochemicals, ROS
1. Introduction
Oxidative stress (OS) within organisms arises when there is an imbalance between the production of reactive oxygen species (ROS) and the body’s ability to neutralize them [1]. ROS are generated during cellular metabolism, particularly in processes like the respiratory chain and tricarboxylic acid (TCA) cycle within mitochondria. ROS, including hydrogen peroxide (H2O2) and superoxide anion (O2•−), play essential roles in physiological functions such as cellular defence and signalling [2]. However, disproportion between ROS production and neutralization can lead to oxidative stress, which is implicated in various pathological conditions [3]. This imbalance, where an excess of reactive molecules overwhelms the body’s innate defence mechanisms, damages cellular structures and essential molecules like lipids, proteins and DNA. As a result, this leads to the development and progression of multiple diseases [4]. While ROS, when present in controlled, low concentrations, serve as signalling molecules facilitating cellular functions and offering cellular protection, their excessive production, as seen in conditions like inflammation, can spur the generation of additional highly reactive species, such as superoxide radical (O2•−), hydroperoxyl radical (HO2), singlet oxygen (1O2), ozone (O3), nitric oxide (NO), nitrogen dioxide (NO2), sulphur dioxide (SO2), and sulphur trioxide (SO3) [5]. These reactive species react with cellular components, modifying their normal structure and function. Notably, the oxidative modification of essential enzymes or regulatory sites is critical, changing their redox potential, that trigger alterations in cell signalling pathways and induce programmed cell death [6]. Evidently, oxidative stress and inflammation are closely linked. Oxidative stress can trigger inflammation, while inflammation can, in turn, amplify OS. This creates a harmful cycle that promotes cell damage and a pro-inflammatory environment [7].
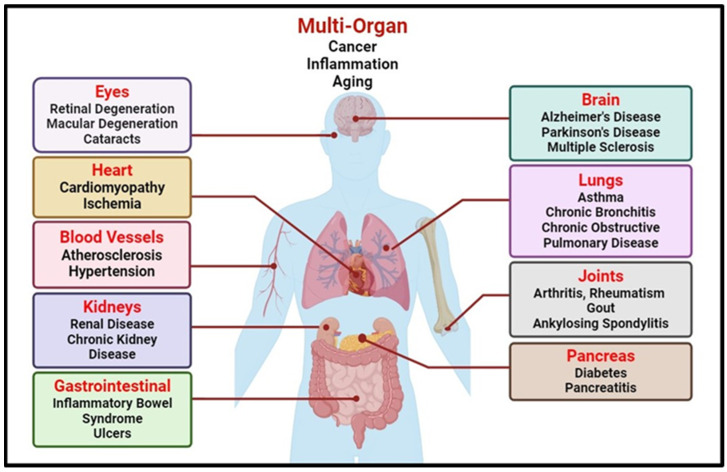
Oxidative stress stands as a central mechanism in the pathogenesis of a spectrum of health disorders, spanning cardiovascular, neurodegenerative, and metabolic conditions such as obesity, diabetes and many others [8] (Figure 1). Its pivotal role is evident in the disruption of cell membrane integrity through induced lipid peroxidation, contributing significantly to the progression of cardiovascular complications like atherosclerosis, endothelial dysfunction, and plaque formation, as well as neuronal membrane damage, which underlies various neurodegenerative diseases [9].
Figure 1.
Oxidative stress-induced health modalities (The illustration was created using BioRender at www.biorender.com).
Moreover, the impact of ROS extends beyond membrane disruption, influencing critical proteins and enzymes and thereby compromising essential cellular functions and signalling pathways. This includes the matrix metalloproteinases (MMPs) activation in cardiovascular ailments and the initiation of protein misfolding and aggregation, characteristic of neurodegenerative disorders like Parkinson’s and Alzheimer’s diseases [10]. Furthermore, oxidative stress triggers an inflammatory cascade, marked by the release of pro-inflammatory mediators, perpetuating a cycle that exacerbates cellular damage and disease progression. Notably, oxidative stress causes adipose tissue inflammation and dysfunction, increasing pro-inflammatory cytokines and adipokines, which lead to insulin resistance and disrupted lipid metabolism leading to obesity [11]. In diabetes, oxidative stress impairs insulin signalling and damages pancreatic β-cells, reducing glucose uptake and insulin secretion, thereby worsening the disease [12]. Understanding the intricate interplay between oxidative stress and its associated inflammatory responses is paramount, as it not only elucidates the underlying mechanisms of disease but also offers promising avenues for therapeutic intervention in combating these debilitating health conditions.
Phytochemicals, particularly those derived from high-altitude medicinal plants, have emerged as potent antioxidants with the potential to counteract oxidative stress and its associated health disorders by scavenging harmful free radicals in the body [13]. Their diverse mechanisms of action also include anti-inflammatory effects, modulation of cellular signalling pathways, and enhancement of immune function. High altitude medicinal plants have adapted to extreme environmental conditions such as low oxygen levels, intense ultraviolet radiation and temperature fluctuations. These harsh conditions stimulate the production of bioactive compounds within these plants, making them rich sources of phytochemicals with unique properties [14]. The exploration of high-altitude medicinal plants not only preserves cultural traditions but also harnesses their therapeutic potential for modern medicine, particularly in combating oxidative stress-related diseases and discovering novel pharmaceutical compounds. Though high-altitude regions harbour a vast array of plant species and genetic diversity, much of this biodiversity remains unexplored and underutilized [15]. This untapped reservoir of biological diversity offers immense potential for discovering new bioactive compounds and understanding evolutionary adaptations to extreme environments. Therefore, exploring high-altitude medicinal plants as sources of potent antioxidants not only advances our understanding of natural defence mechanisms but also paves the way for developing innovative therapeutic strategies to overcome oxidative stress-related diseases [16]. This study highlights and summarizes the production and physio-pathological aspects of oxidative imbalance and emphasizes the role of phytochemicals in mitigating these effects. Further, this study provides a comprehensive tabulation of more than 160 high-altitude medicinal plants along with their reported phytochemicals, which could be very useful in harnessing their potential to combat lifestyle-associated, oxidative stress-induced disorders and could serve as a starting point for the exploration of alternate medicine for combating these diseases.
1.1. Oxidative Stress: Source, Mechanism and Lifestyle-Related Diseases
1.1.1. Source of Oxidative Stress
Oxidative stress occurs when highly reactive species, such as superoxide radical (O2•−), hydroperoxyl radical (HO2), singlet oxygen (1O2), and ozone (O3); reactive nitrogen species (RNS) like nitric oxide (NO) and nitrogen dioxide (NO2); and reactive sulphur species (RSS) like sulphur dioxide (SO2) and sulphur trioxide (SO3), overwhelm the natural antioxidant defence system of a body. This leads to cellular damage and dysfunction, which can contribute to a wide array of diseases [17]. These reactive species are continuously produced within cells at low levels during normal metabolic processes, which are safely neutralized by cellular machinery, but can also stem from contact to external factors such as radiation (such as X-rays and UV), air pollutants, ozone, cigarette smoke, bacteria, viruses, drugs and various forms of cellular stress, whether acute or chronic [18].
These reactive species include both non-radicals and free radical oxidants. Free radicals are particularly unstable due to having unpaired electrons in their outer electron orbit. This instability drives them to react with other molecules, causing oxidation and subsequent harm to crucial biological molecules such as nucleic acids (DNA, RNA), lipids and proteins [19].
The key intercellular origin of these reactive species includes endoplasmic reticulum, mitochondria, peroxisomes, lysosomes, plasma membrane and, cytosol [20]. ROS, formed from the chemical reactions involving molecular oxygen, encompass free radicals such as superoxide anions (O2•−) and hydroxyl radicals (OH⁻), alongside non-radical oxidants like hydrogen peroxide (H2O2) and hypochlorous acid (HOCl). Reactive nitrogen species, on the other hand, include peroxynitrite radical (ONOO⁻), and nitric oxide (NO). Recently identified reactive sulphur species (RSS) include thiol radical (RS•) and RSS formed through reactions between ROS and thiols. RSS exhibit both radical and non-radical properties, and they have a particular affinity for sulphur-containing molecules, such as peptides and proteins, triggering oxidation and reduction reactions [20].
Enzymes of the mitochondrial electron transport respiratory chain are major contributors to ROS production [21]. Furthermore, various other enzymes catalyse chemical reactions that contribute to ROS formation. These include homologs of phospholipase A2 (PLA2), nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, cyclooxygenase (COX), uncoupled nitric oxide (NOS), xanthine oxidase (XO), glucose oxidase (GOXs), myeloperoxidase (MPO) and, lipoxygenases (LOXs) [22].
NADPH oxidase (NOX), initially identified in the phagosomes of immune cells, has several homologs with diverse intracellular localizations. Some homologs, like DUOX2 and NOX1, play major roles in various inflammatory conditions and tumours. Xanthine oxidase, primarily expressed in the small intestinal mucosa and liver, catalyses ROS production both on the outer surface of the plasma membrane and in the cytoplasm [23]. Lipoxygenases are non-heme iron enzymes that accumulate ROS by oxidizing arachidonic acid (AA), whereas myeloperoxidase, a heme protein that localizes lysosomes, contributes to ROS production in immune cells [24].
1.1.2. Mechanism of ROS Production
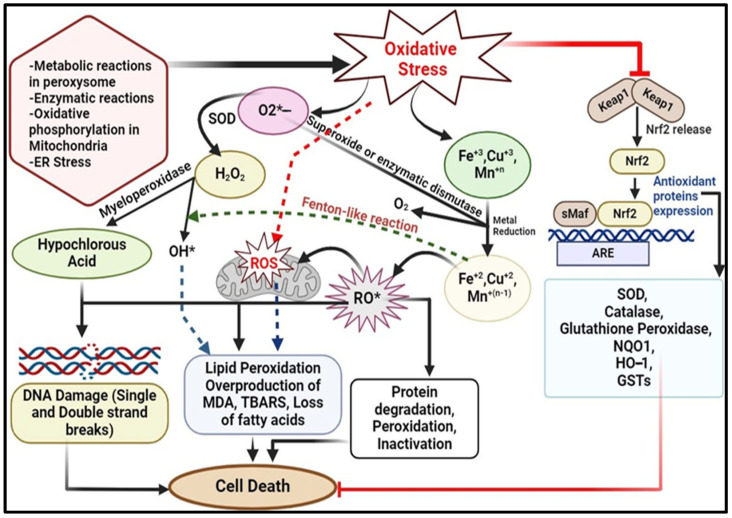
Mitochondria is the main endogenous source of ROS because of its involvement in ATP synthesis through oxidative phosphorylation. This process involves the reduction of molecular oxygen (O2) to water (H2O) in the electron transport chain (ETC) [10]. Superoxide (O2•−) production within mitochondria is a significant contributor to cellular ROS. Seven primary sites of superoxide production have been identified in mammalian cells [6]. Ranked by their highest capacity, these include the ubiquinone binding sites in complex I (site IQ) and complex III (site IIIQo); glycerol 3-phosphate dehydrogenase (GPDH); the flavin in complex I (site IF); electron transferring flavoprotein: Q oxidoreductase (ETFQOR), involved in fatty acid beta-oxidation; and pyruvate and 2-oxoglutarate dehydrogenases. Most of these complexes release O2•− into the mitochondrial matrix, except for complex III site and GPDH. Within the mitochondrial membrane, three types of superoxide dismutase (SOD) exist: copper superoxide dismutase (Cu-SOD), manganese superoxide dismutase (Mn-SOD), and zinc superoxide dismutase (Zn-SOD). Mn-SOD catalyses the conversion of O2•− into H2O2. Hydrogen peroxide can then be converted into a hydroxyl radical by the enzyme aconitase through the Fenton reaction. Copper and zinc SODs function primarily in the inter-membrane space to convert superoxide into less ROS [25] (Figure 2).
Figure 2.
Source, mechanism of production of ROS leading to oxidative stress and its repercussions along with cellular antioxidant defence. (The illustration was created using BioRender www.biorender.com.) [*: Free radical; Nrf2: Nuclear factor erythroid 2-related factor 2; ARE: Antioxidant Response Element; Mn: Manganese; Cu: Copper; Fe: Iron; OH: Hydroxyl radical; SOD: Superoxide Dismutase; NQO1: NAD(P)H quinone dehydrogenase 1; HO-1: Heme Oxygenase-1; GSTs: Glutathione S-transferases; MDA: Malondialdehyde; TBARS: Thiobarbituric Acid Reactive Substances; ROS: Reactive Oxygen Species; RO: Reactive Oxygen; O2: Oxygen; Keap1: Kelch-like ECH-associated protein 1; sMaf: Small Maf proteins; H2O2: Hydrogen Peroxide].
An alternative pathway for generating ROS involves the mitochondrial cytochrome catalytic cycle, which includes enzymes like cytochrome P450. These enzymes process a broad variety of organic compounds, such as steroids, lipids and, xenobiotics, leading to the production of different reactive byproducts, including hydrogen peroxide and superoxide radicals [2]. Additionally, in mammals, various protein complexes, such as nicotinamide adenine dinucleotide (NADH)-cytochrome b5 reductase (b5R), dihydroorotate dehydrogenase (DHODH), succinate dehydrogenase (SDH) from complex II, and monoamine oxidases (MAO), generate ROS [5]. Numerous antioxidant defence systems safeguard mitochondria from the detrimental effects of ROS. These include endogenous antioxidants like glutathione peroxidases (GPXs), thioredoxin peroxidases (TRXPs), SODs, peroxiredoxins (PRDXs), glutathione (GSH), thioredoxin-2 (TRX2), glutaredoxin-2 (GRX2), cytochrome C oxidase (complex IV), and coenzyme Q. Additionally, exogenous antioxidants, such as ascorbic acid, vitamin E, and phytochemicals (carotenes, phenols, etc.), play crucial roles in this protective mechanism [5] (Figure 2). Excessive production of ROS is associated with numerous human disorders. These include myocardial dysfunction, inflammation, diabetes, neurodegenerative disease, aging, chronic kidney disease and DNA damage leading to cancer. ROS can cause damage to genomic and mitochondrial DNA, leading to mutations in somatic cells, genomic instability, activation of oncogenes, suppression of tumour suppressor genes, and disruptions in various metabolic and signalling pathways. Compensatory mechanisms may initially be activated but can ultimately contribute to cellular damage and the development of various pathological conditions [2].
1.1.3. Lifestyle-Associated Oxidative Stress-Induced Disorders
Besides many other external factors, lifestyle factors, such as lack of physical activity, smoking, poor dietary habits, and excessive alcohol intake, predominately sponsors the onset of oxidative stress-related disorders (Figure 1). These behaviours result in the overproduction of ROS, overwhelming the body’s antioxidant defences and leading to oxidative stress [26]. In cardiovascular diseases, oxidative stress damages blood vessels and promotes atherosclerosis. In neurodegenerative disorders, ROS-induced neuronal damage accelerates conditions like Parkinson’s and Alzheimer’s diseases [27]. For metabolic disorders, oxidative stress disrupts insulin signalling and lipid metabolism, fostering obesity and diabetes [28]. Addressing these lifestyle factors is crucial for preventing and managing these oxidative stress-related diseases.
Cardiovascular Diseases
Atherosclerosis
Oxidative stress stages the oxidation process of LDL cholesterol, giving rise to oxidized LDL (oxLDL). Within the arterial wall, macrophages ingest oxLDL, which triggers foam cell formation and initiates an inflammatory reaction. This response triggers the release of chemokines, and cytokines, which recruit additional immune cells to the site of inflammation. Further, oxidative stress enhances endothelial dysfunction, promoting vasoconstriction and platelet aggregation, which contribute to plaque formation and narrowing of arteries [29].
-
2.
Hypertension
Oxidative stress diminishes the availability of nitric oxide (NO), a powerful vasodilator, by scavenging it and promoting its inactivation. This results in endothelial dysfunction and impaired vasodilation, contributing to increased peripheral vascular resistance and hypertension. Moreover, ROS can activate the renin—angiotensin—aldosterone system (RAAS), that leads to vasoconstriction and sodium retention, further exacerbating hypertension [30].
-
3.
Myocardial Infarction
Oxidative stress stages the development and progression of plaque and atherosclerosis instability, thereby increasing the risk of plaque rupture and thrombosis. ROS can directly damage cardiomyocytes and impair myocardial contractility. Additionally, oxidative stress activates inflammatory pathways, promoting myocardial inflammation and fibrosis, which can lead to cardiac remodelling and dysfunction [31].
Neurodegenerative Diseases
Alzheimer’s Disease (AD)
Oxidative stress induces the accumulation of hyperphosphorylated tau proteins and, β-amyloid (Aβ) peptides, leading to the formation of senile plaques and neurofibrillary tangles, respectively. ROS also disrupts calcium homeostasis, mitochondrial function, and synaptic transmission, contributing to neuronal dysfunction and cell death. Inflammatory mediators, including cytokines and microglial activation, further exacerbate neuroinflammation and neuronal damage in AD [32].
-
2.
Parkinson’s Disease (PD)
Oxidative stress promotes the misfolding and accumulation of α-synuclein protein, forming Lewy bodies, the pathological hallmark of PD. ROS-induced mitochondrial dysfunction leads to impaired energy production, increased oxidative damage, and neuronal cell death, particularly in dopaminergic neurons of the substantia nigra. Additionally, oxidative stress activates microglia and astrocytes, triggering neuroinflammation and neurodegeneration in PD [33].
Cancer
DNA Damage and Mutation
Oxidative stress induces DNA lesions, including strand breaks, base modifications and DNA—protein cross-links. Unrepaired DNA damage can lead to mutations in tumour suppressor genes and oncogenes, promoting the initiation and progression of tumours. Additionally, ROS-mediated activation of signalling pathways, such as nuclear factor-kappa B (NF-κB) and mitogen-activated protein kinases (MAPKs), further drives tumour growth, invasion, and metastasis [34].
-
2.
Tumour Angiogenesis
Oxidative stress promotes the production of angiogenic factors, such as vascular endothelial growth factor (VEGF) and hypoxia-inducible factor 1-alpha (HIF-1α), which stimulate the formation of new blood vessels to support tumour growth and metastasis. ROS-mediated activation of pro-angiogenic pathways and inhibition of anti-angiogenic factors contribute to tumour angiogenesis and neovascularization [35].
Metabolic Disorders
Insulin Resistance
Oxidative stress impairs insulin signalling pathways by promoting serine phosphorylation of insulin receptor substrate 1 (IRS-1), inhibiting its association with the insulin receptor and downstream activation of phosphatidylinositol 3-kinase (PI3K) and Akt. This leads to decreased glucose uptake and glycogen synthesis, and increased gluconeogenesis and lipolysis, leading to insulin resistance and hyperglycaemia in type 2 diabetes [36].
-
2.
Obesity
Oxidative stress promotes adipocyte dysfunction and inflammation by activating pro-inflammatory pathways, such as NF-κB and c-Jun N-terminal kinase (JNK). ROS induce the secretion of pro-inflammatory cytokines, such as tumour necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6), from adipose tissue macrophages and adipocytes, causing a persistent inflammatory condition that leads to insulin resistance, dyslipidemia, and overall metabolic dysfunction [37].
1.2. Antioxidant Defence Systems
Antioxidants play a pivotal part in preventing or delaying the oxidation of target molecules caused by ROS, which in turn leads to oxidative stress. These compounds act as defenders by donating electrons to free radicals, neutralizing their harmful effects on lipids, proteins, DNA, and other biomolecules [38]. They serve as scavengers within biological systems and are essential defence mechanisms against oxidative stress [4].
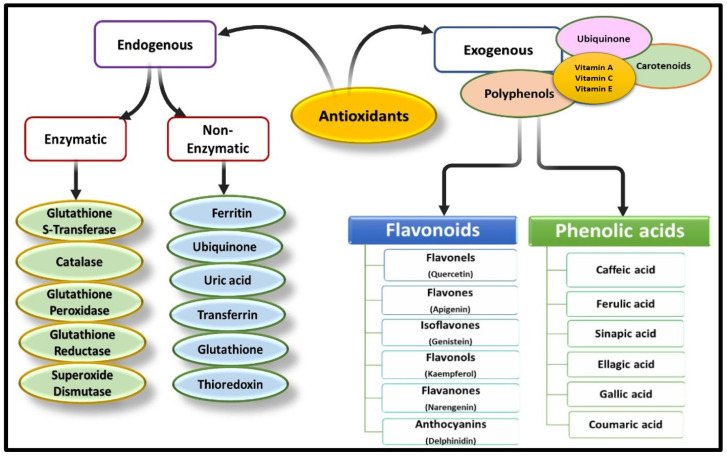
Antioxidants can originate from external sources, known as exogenous antioxidants, which are mainly obtained through food, as well as from internal sources, referred to as endogenous antioxidants, which are produced within the body [39]. Endogenous antioxidants can be enzymatic or non-enzymatic in nature [40]. Enzymatic antioxidants are a specific category of antioxidant systems present in the human body. These enzymes possess antioxidant activity and are capable of acquiring different valences, allowing them to transfer electrons to neighbouring free radicals, thereby facilitating their breakdown and neutralization [41]. Some examples of enzymatic antioxidants include glutathione reductase (GR), superoxide dismutase, catalase (CAT), and glutathione peroxidase (GPx) [42]. Glutathione reductase aids in the production of reduced glutathione, which helps counteract the oxidative damage caused by ROS [43]. Similarly, SOD plays a crucial role in neutralizing free radical species by converting superoxide radicals into hydrogen peroxide [25,44]. Non-enzymatic endogenous antioxidants are produced within the body through various metabolic pathways and physiological processes. Therefore, these antioxidants are essential for neutralizing ROS and protecting cells from oxidative damage [45]. Some examples of non-enzymatic endogenous antioxidants are glutathione (GSH), uric acid, bilirubin, melatonin and alpha-lipoic acid (Figure 3).
Figure 3.
Endogenous and exogenous sources of antioxidants. (The illustration was created using BioRender www.biorender.com).
Exogenous antioxidants refer to the types of antioxidants that originate outside the body and can be supplied to the body primarily through diet or supplements. These antioxidants encompass various essential nutrients like vitamin C, vitamin E, omega-3 and omega-6 fatty acids [46]. Additionally, they may include certain plant-derived phytochemicals such as polyphenols, including flavonoids, as well as trace elements like zinc and manganese [16]. Synthetic antioxidants like butyl hydroxyanisole may also be classified as exogenous antioxidants, as they aid in preventing lipid oxidation [40].
Phytochemicals are low molecular weight non-enzymatic compounds produced by plants and possess numerous medicinal and therapeutic properties [47,48]. Certain phytochemicals possess antioxidant properties and actively engage with oxidative radicals, neutralizing their harmful effects through various mechanisms. These include scavenging free radicals by electron transfer and chelating metal ions that trigger ROS production. Different groups of phytochemicals such as flavonoids, ascorbic acid and carotenoids, exhibit diverse antioxidant activities against different ROS.
Medicinal plants that thrive at high altitudes possess inherent protective processes against the detrimental results of ROS [49]. They produce enzymatic antioxidants like SOD and CAT, as well as non-enzymatic antioxidants such as tannins, flavonoids, and ascorbic acid in bulk quantities to mitigate harsh environmental stress factors [50]. However, due to challenges associated with their isolation and the risk of denaturation, plant-derived enzymatic antioxidants are typically not employed for therapeutic purposes [51]. Some plants possess genetic capabilities to synthesize phytochemicals that effectively neutralize toxic ROS [47]. Additionally, exposure to various environmental stresses stimulates the production of phytochemicals, which act as countermeasures against ROS [50]. These secondary metabolites, derived from essential metabolic pathways, exert protective effects by preventing the oxidation of plant proteins, lipids, and DNA through passive or active resistance mechanisms [52].
This study provides a summary of major oxidative stress-induced health disorders and mechanistic details of phytochemicals being used as antioxidants. This study also aims to focus upon high altitude medicinal plants as the bulk producers of antioxidants and as a potential source of plant-derived therapeutic agents against lifestyle-induced oxidative stress-related diseases.
2. Phytochemicals as Antioxidants
Phytochemicals are non-enzymatic compounds, with low molecular weight, that abundantly exist in plants [53]. These biologically active substances have gained recognition for their medicinal and therapeutic properties. The World Health Organization (WHO) has acknowledged the use of these plant-derived compounds in the treatment of various human diseases, highlighting their significance in healthcare [48]. Numerous phytochemicals possess antioxidant properties and actively engage with oxidative radicals such as ROS, neutralizing their harmful effects by scavenging free radicals by electron transfer and chelating metal ions that trigger ROS production [47]. Many phytochemicals, such as flavonoids, ascorbic acid and carotenoids, show diverse mechanisms by which to counter the effects of ROS and to therefore mitigate OS [13]. These phytochemicals offer immense potential for inhibiting and treating oxidative stress, contributing to the overall wellbeing and health of individuals.
2.1. Carotenoids
Carotenoids are lipophilic pigments found in plant plastids. They are responsible for the vibrant colours seen in various fruits and vegetables [54]. Carotenes, having a beta-ionone ring, also serve as a crucial source for the synthesis of vitamin A [55]. Almost 1200 natural carotenoids have been identified and characterized so far, along with their structures and biological sources (http://carotenoiddb.jp; accessed on 7 June 2024), with beta-carotene being the most extensively studied among them [56]. The chemical structure of carotenoids consists of 40 carbon atoms arranged in a specific pattern of double bonds, which contributes to their antioxidant properties [57].
Carotenoids can be broadly classified into two categories: carotenes, which contain carbon and hydrogen atoms, and xanthophylls, which contain at least one oxygen atom [58]. Carotenes include alpha-carotene, beta-carotene, lutein, and lycopene, while xanthophylls encompass canthaxanthin, antheraxanthin, zeaxanthin, and others [59].
The antioxidant action of carotenoids primarily involves their ability to react with peroxyl radicals and singlet oxygen species, thereby preventing oxidative damage to lipid membranes [60]. Singlet oxygen species transfer their energy to nearby carotenoid molecules, allowing the oxygen molecule to return to its non-toxic state. The excited carotenoid molecule then dissipates its energy to the surrounding solvent, returning to its ground state and enabling it to react with other free radicals [61].
Carotenoids have demonstrated effectiveness against various diseases associated with oxidative stress, including Alzheimer’s disease [62]. Certain carotenoids, such as beta-carotene, have been found to bind efficiently to receptors associated with Alzheimer’s disease, such as histone and p53 receptors [63]. Carotenoids also play a protective role against photo-oxidative damage to the skin caused by UV radiation. By leveraging their antioxidant properties, carotenoids, like lycopene and beta-carotene, can help suppress and inhibit skin diseases, mitigating the risk of dermatoses and cutaneous malignancy [60]. Additionally, carotenoids show potential in inhibiting the progression of health abnormalities such as rheumatoid arthritis and have cardiovascular protective effects [64]. Lutein and zeaxanthin, key carotenoids concentrated in the macula of the eye, play critical roles in eye health by acting as antioxidants and blue light filters. These compounds protect retinal cells by neutralizing ROS and reducing oxidative stress, which are known contributors to age-related macular degeneration (AMD). Mechanistically, lutein and zeaxanthin absorb blue light wavelengths, particularly those most damaging to the retina (400—500 nm), thereby preventing phototoxicity and subsequent cellular damage. Their presence in the macular pigment also enhances visual performance by improving contrast sensitivity and by reducing glare. Scientific evidence supports their effectiveness in maintaining retinal integrity and potentially slowing the progression of AMD, underscoring their importance in preserving long-term eye function and vision [65].
Overall, carotenoids serve as valuable antioxidants, contributing to the prevention and management of various diseases linked to oxidative stress.
2.2. Ascorbic Acid (AsA)
Ascorbic acid (AsA), popularly known as vitamin C, plays an important role in the non-enzymatic defence mechanisms against ROS [66]. This class of antioxidant compounds consists of low molecular weight substances that act as reducing agents [67]. Plants produce ascorbic acid through the Smirnoff-Wheeler pathway, involving the conversion of mannose and lactose in their D and L forms. Additionally, the Wolucka—Van pathway serves as an alternative route for synthesizing ascorbic acid in plants. Mitochondria, particularly in the photosynthetic tissues of plants, serve as key sites for the production of ascorbic acid, which exists in two forms: semi-dehydroascorbyl radical and dehydroascorbate [68].
Ascorbic acid (vitamin C) plays a pivotal role in the ascorbate—glutathione cycle in plants, serving as a primary antioxidant by scavenging ROS such as hydrogen peroxide [69]. It undergoes oxidation to monodehydroascorbate (MDHA) and dehydroascorbate (DHA) during ROS detoxification. DHA is then reduced back to ascorbic acid by dehydroascorbate reductase (DHAR), with the assistance of glutathione, thereby replenishing the cellular pool of active ascorbate. Additionally, ascorbic acid regenerates oxidized vitamin E (tocopherol and tocotrienol) by reducing tocopheroxyl radicals (vitamin E), prolonging vitamin E’s antioxidant function in protecting cellular membranes from oxidative damage. This cycle ensures effective antioxidant defence and redox homeostasis, essential for plant resilience against environmental stressors [70].
Within plants, free radicals are generated as a result of metabolic activities in the presence of oxygen or exposure to UV radiation [19]. Ascorbic acid acts as an antioxidant by scavenging ROS, including hydrogen peroxide, superoxide anion, and hydroxyl radical, forming monodehydroascorbate. By doing so, it protects essential biomolecules such as unsaturated fatty acids, proteins, and DNA from damage [71]. The antioxidant activity of ascorbic acid contributes to the prevention of various cardiovascular disorders and gastric problems. It enhances the concentration of nitric oxide in the vascular endothelium, thus aiding in the prevention of hypertension. Moreover, ascorbic acid promotes the absorption of iron in the small intestine, offering potential inhibition of gastric issues associated with Helicobacter pylori infection [72].
Overall, ascorbic acid serves as a vital antioxidant in plants, safeguarding against oxidative damage and contributing to the prevention of cardiovascular and gastric ailments in humans [73].
2.3. Tocopherols and Tocotrienols
Tocopherols and tocotrienols are isoforms of vitamin E, consisting of four types: alpha, beta, gamma, and delta [74]. These phytochemicals possess a hydrophobic nature and contain a prenyl group [75]. They exhibit significant antioxidant activity and play a crucial role in preventing various cardiovascular diseases, neurodegenerative diseases, like Alzheimer’s, and aging [76]. The antioxidant characteristics of tocopherols and tocotrienols are attributed to the occurrence of a chromanol ring in their structure. This ring contains a hydroxyl group that combats free radicals by donating hydrogen atoms [77].
Among the various forms of vitamin E, both alpha tocopherols and tocotrienols are particularly active in preventing lipid peroxidation caused by free radicals, thereby protecting cell membranes from damage [78]. The alpha forms of tocopherols and tocotrienols work by inhibiting the generation of free radicals, while the gamma forms are effective in capturing and neutralizing the impacts of ROS. Collectively, these vitamin E isoforms contribute to the body’s defence against oxidative stress and its detrimental effects [77].
2.4. Polyphenols
Polyphenols are a prominent class of phytochemicals, which play a major role as antioxidants [79]. They are synthesized by plants as a result of shikimic acid pathway from amino acids phenylalanine or tyrosine [80]. Polyphenols exhibit varying molecular weights depending upon the degree of polymerization (small molecules such as quercetin have a molecular weight of 302.24 Da, while, as tannins, they can reach several thousand kDa due to their polymeric nature) and exert antioxidant effects by acting as reducing agents [81]. They donate hydrogen atoms to the ROS produced, thus, scavenging the free radical species [82]. Important polyphenols present in the plants, which perform antioxidant activity, are flavonoids, phenolic acids and lignans [83,84,85].
Flavonoids are a major group of plant phenolic compounds, characterized by the presence of a flavan nucleus in their chemical structure [86]. They have 2 benzene rings denoted by ring A and ring B connected to a third pyran ring that is ring C [87]. These phytochemicals play a major role in preventing the peroxidation of lipids by using processes such as electron transfer or chelation of metal ions [88]. The B ring that is present in the molecular structure of flavonoids engage a major role in the scavenging of free radicals. The B ring contains hydroxyl groups, which stabilize the free radical species, such as hydroxyl or peroxynitrite, by transferring either electrons or hydrogen atoms to them [89]. Flavonoids further prevent oxidative stress by chelating metal ions such as copper or ferric ions which stimulate the production of ROS in the body [89]. Different types of flavonoids exhibiting antioxidant activity include flavonols, flavones, isoflavone, and anthocyanidin and are found mainly in citrus fruits, tea, onion, berries, broccoli and soybean [86].
Stilbenes are a major sub-class of polyphenols present in the plants which also show antioxidant activity [90]. Stilbenes such as resveratrol help in preventing the oxidative stress to proteins and lipids and it also increases the activity of antioxidant enzymes such as GPx and SOD [91]. Phenolic acids such as salicylic acid, vanillic acid, caffeic acid also show significant antioxidant activity [85].
2.5. Polysterols
Polysterols are a subclass of sterols, which are a type of lipid characterized by a specific chemical structure containing a steroid nucleus [92]. These compounds naturally occur in plants and have gained recognition for their potential health benefits, particularly due to their antioxidant properties [93]. Polysterols possess the ability to scavenge free radicals and reduce oxidative stress within the body, thereby contributing to overall health and wellbeing [82]. One example of a polysterol compound with potent antioxidant activity is beta-sitosterol, which has been studied for its potential role in promoting cardiovascular health and supporting the immune system [94]. Another example is campesterol, which also exhibits antioxidant effects and may contribute to the prevention of chronic diseases associated with oxidative damage [95].
A comprehensive list of phytochemical classes and their representative antioxidant molecules, along with their high-altitude plant sources and their associated health benefits is shown in Table 1.
Table 1.
List of phytochemical classes, along with representative molecules within each class having antioxidant property, their sources from high-altitude plants and their therapeutic properties against various oxidative stress associated diseases.
| Phytochemical Class | Sub-Class | Representative Compounds | Chemical Formulae | PubChem ID | High Altitude Plant Source | Preventive Activity Against | Reference |
|---|---|---|---|---|---|---|---|
| Carotenoids | Carotenes | Alpha-carotene | C40H56 | 6419725 |
Gentiana algida Pall., Rhododendron ferrugineum L., Ranunculus glacialis L., Saxifraga oppositifolia L., Primula hirsuta All. |
Cardiovascular diseases, type 2 diabetes, cancer, skin and eye diseases, ageing, inflammation | [96,97] |
| Beta-carotene | C40H56 | 5280489 | |||||
| Lycopene | C40H56 | 446925 | |||||
| Phytoene | C40H64 | 5280784 | |||||
| Phytofluene | C40H62 | 6436722 | |||||
| Xanthophylls | Lutein | C40H56O2 | 5281243 | ||||
| Canthaxanthin | C40H52O2 | 5281227 | |||||
| Antheraxanthin | C40H56O3 | 5281223 | |||||
| Zeaxanthin | C40H56O2 | 5280899 | |||||
| β-cryptoxanthin | C40H56O | 5281235 | |||||
| Astaxanthin | C40H52O4 | 5281224 | |||||
| Fucoxanthin | C42H58O6 | 5281239 | |||||
| Rubixanthin | C40H56O | 5281252 | |||||
| Violaxanthin | C40H56O4 | 448438 | |||||
| Vitamins | Ascorbic Acid | C6H8O6 | 54670067 |
Vaccinium macrocarpon Aiton. (Mountain cranberry), Sorbus aucuparia Poir., Sorbus scopulina Greene, Juniperus recurva Buch. -Ham. ex D. Don. |
Age-related muscular degeneration, cataract, cardiovascular diseases, immunosuppression | [98,99] | |
| Tocopherols | Alpha-tocopherol | C29H50O2 | 14985 | Cardiovascular diseases, cancer, obesity, diabetes | |||
| Beta-tocopherol | C28H48O2 | 6857447 | |||||
| Gama-tocopherol | C28H48O2 | 92729 | |||||
| Delta-tocopherol | C27H46O2 | 92094 | |||||
| Tocotrienols | Alpha-tocotrienol | C29H44O2 | 5282347 | ||||
| Polyphenols | Flavonoids | Quercetin | C15H10O7 | 5280343 |
Rhodiola rosea L., Vaccinium vitis-idaea L., Dipsacus fullonum L., Dipsacus sylvestris Huds., Juniperus recurva Buch. -Ham. ex D. Don. |
Obesity, neurodegenerative diseases, type 2 diabetes, and cardiovascular diseases | [100,101] |
| Kaempferol | C15H10O6 | 5280863 | |||||
| Fisetin | C15H10O6 | 5281614 | |||||
| Isorhamnetin | C16H12O7 | 5281654 | |||||
| Myricetin | C15H10O8 | 5281672 | |||||
| Luteolin | C15H10O6 | 5280445 | |||||
| Apigenin | C15H10O5 | 5280443 | |||||
| Sinensetin | C20H20O7 | 145659 | |||||
| Isosinensetin | C20H20O7 | 632135 | |||||
| Nobiletin | C21H22O8 | 72344 | |||||
| Tangeretin | C20H20O7 | 68077 | |||||
| Galangin | C15H10O5 | 5281616 | |||||
| Chrysin | C15H10O4 | 5281607 | |||||
| Baicalin | C21H18O11 | 64982 | |||||
| Catechin | C15H14O6 | 9064 | |||||
| Epicatechin | C15H14O6 | 72276 | |||||
| Epicatechin gallate | C22H18O10 | 107905 | |||||
| Gallocatechin | C15H14O7 | 65084 | |||||
| Epigallocatechin | C15H14O7 | 72277 | |||||
| Epigallocatechin gallate | C22H18O11 | 65064 | |||||
| Daidzein | C15H10O4 | 5281708 | |||||
| Genistein | C15H10O5 | 5280961 | |||||
| Daidzin | C21H20O9 | 107971 | |||||
| Naringenin | C15H12O5 | 439246 | |||||
| Naringin | C27H32O14 | 442428 | |||||
| Hesperidin | C28H34O15 | 10621 | |||||
| Hesperetin | C16H14O6 | 72281 | |||||
| Eriodicytol | C15H12O6 | 11095 | |||||
| Pelargonidin | C15H11O5⁺ | 440832 | |||||
| Cyanidin | C15H11O6⁺ | 128861 | |||||
| Delphinidin | C15H11ClO7 | 68245 | |||||
| Peonidin | C16H13O6⁺ | 441773 | |||||
| Petunidin | C16H13O7⁺ | 441774 | |||||
| Malvidin | C17H15O7⁺ | 159287 | |||||
| Stilbenes | Resveratrol | C14H12O3 | 445154 | ||||
| Pinosylvin | C14H12O2 | 5280457 | |||||
| Piceatannol | C14H12O4 | 667639 | |||||
| Pterostilbene | C16H16O3 | 5281727 | |||||
| Rhapontigenin | C15H14O4 | 5320954 | |||||
| Isorhapontigenin | C15H14O4 | 5318650 | |||||
| Phenolic acids | Salicylic acid | C7H6O3 | 338 | ||||
| Hydroxybenzoic acid | C7H6O3 | 135 | |||||
| Protocatechuic acid | C7H6O4 | 72 | |||||
| Gallic acid | C7H6O5 | 370 | |||||
| Syringic acid | C9H10O5 | 10742 | |||||
| Vanillic acid | C8H8O4 | 8468 | |||||
| Gentisic acid | C7H6O4 | 3469 | |||||
| Coumaric acid | C9H6O2 | 323 | |||||
| Phytosterols | Campesterol | C28H48O | 173183 |
Rhodiola spp., Dipsacus spp., Juniperus spp. |
Elevated cholesterol level, inflammation, oxidative stress, immunosuppression. | [102,103] | |
| Sitosterol | C29H50O | 222284 | |||||
| Stigmasterol | C29H48O | 5280794 | |||||
| Campestanol | C28H50O | 119394 | |||||
| Stigmastanol | C29H52O | 241572 | |||||
3. Role of Phytochemical Antioxidants in Mitigating Major Lifestyle-Associated Oxidative Stress-Induced Health Disorders
3.1. Cardiovascular Diseases
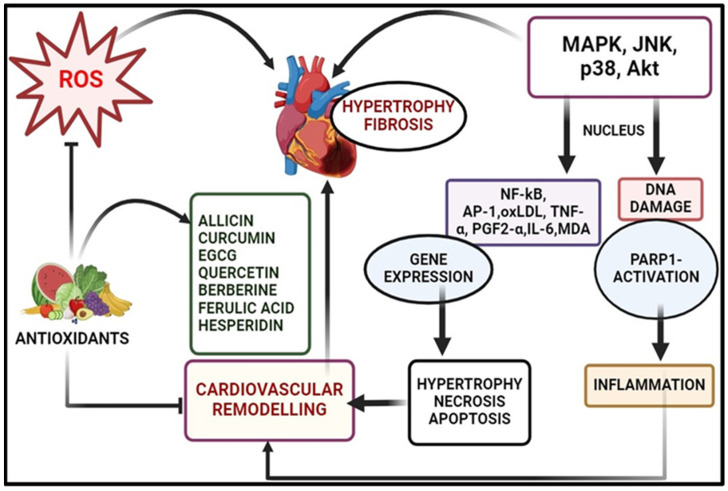
Cardiovascular disease (CVD), the leading cause of global mortality, is intricately linked to oxidative damage, with ROS orchestrating various deleterious effects [104]. As discussed in the section regarding cardiovascular diseases, elevated ROS levels diminish nitric oxide availability, inducing vasoconstriction and hypertension, while also disrupting myocardial calcium handling, leading to arrhythmias and cardiac remodelling via hypertrophic signalling and apoptosis [105,106] (Figure 4). Chronic oxidative stress in heart failure triggers cardio myocyte apoptosis, fibrosis, and mitochondrial dysfunction, perpetuating myocardial damage and dysfunction through pro-inflammatory cytokine activation, fibrotic growth factor release, and impaired calcium homeostasis. Atrial fibrillation (AF) is the most common cardiac arrhythmia, fuelled by oxidative stress-induced atrial remodelling and inflammation, promoting structural changes and fibrosis, which create a substrate for atrial fibrillation [107].
Figure 4.
Oxidative stress-induced cardiovascular diseases and modulation via phytochemical antioxidants (the illustration was created using BioRender at www.biorender.com). [MAPK: mitogen-activated protein kinase; ROS: reactive oxygen species; JNK: c-Jun N-terminal kinase; p38: p38 mitogen-activated protein kinase; Akt: protein kinase B (PKB); NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; AP-1: activator protein 1; oxLDL: oxidized low-density lipoprotein; TBF-α: tumour necrosis factor alpha; PGF2-α: prostaglandin F2 alpha; IL-6: interleukin 6; MDA: malondialdehyde; PARP-1: poly (ADP-ribose) polymerase 1].
In the relentless pursuit to mitigate oxidative damage in cardiovascular tissue, there has arisen a growing interest in the utilization of medicinal plants as natural antioxidants [108]. The bioactive components derived from these botanical sources, encompassing polyphenols and polysaccharides commonly found in traditional herbal medicine, hold promise in combatting oxidative stress and its associated cardiovascular disorders [109]. Table 2 delineates the myriad plant bioactive compounds targeting oxidative stress pathways and related cardiovascular diseases. As free radicals instigate a chain reaction of oxidative damage within cardiovascular tissues [19], the active constituents found in medicinal plants serve as potent scavengers, blocking this detrimental process through both direct and indirect mechanisms [110].
One important example is curcumin, which is derived from the turmeric plant and is renowned for its anti-inflammatory and antioxidant properties. Curcumin exerts antioxidant effects by directly scavenging free radicals and upregulating endogenous antioxidant enzymes [111]. It also inhibits inflammatory pathways, such as NF-κB pathway, thereby mitigating inflammation and oxidative stress in cardiovascular tissues [111]. Epigallocatechin gallate (EGCG), found in tea, is renowned for its potent antioxidant and cardioprotective effects. EGCG modulates signalling pathways involved in oxidative stress and inflammation, such as the MAPK and PI3K/Akt pathways, thereby protecting against cardiovascular diseases [112]. Quercetin, abundant in various fruits, vegetables, and teas, functions as a free radical scavenger, inhibits lipid peroxidation, and enhances the activity of antioxidant enzymes like SOD and CAT. Furthermore, it modulates inflammatory pathways, including NF-κB and COX, thereby mitigating oxidative stress and inflammation in cardiovascular tissues [113].
Given their favourable safety profile and multifaceted antioxidative properties, the exploration and integration of plant-derived phytochemical antioxidants into clinical practice hold tremendous potential for ameliorating oxidative stress in the management of cardiovascular disorders [114].
Table 2.
Phytochemicals, along with their high-altitude plant sources, are reported to mitigate oxidative stress-induced cardiovascular diseases [108].
| Phytochemical | Plant | Chemical Structure | Treatment | Mechanism of Action | Reference |
|---|---|---|---|---|---|
| Allicin | Allium humile Kunth |

|
Hypertension | Inhibits the formation of LPO and MDA | [108,115] |
| Berberine | Berberis aristata DC. |

|
Hypertension | Reduces O2 and H2O2 levels | [116] |
| Delphinidin-3-glucoside | Vaccinium myrtillus L. |

|
Coronary heart disease, ischemia-reperfusion injury | Inhibits caspase-3, bax, and ap-JNK expression | [117,118] |
| Gastrodin | Gastrodia elata Blume. |

|
Heart failure | Regulates AMPK, Akt, mTOR, and Bcl-2 | [119] |
| Gypenoside | Gynostemma pentaphyllum Thunb. |

|
Acute myocardial infarction | Regulates the PI3K/Akt/mTOR signalling pathway | [120,121] |
| Matrine |
Sophora flavescens Aiton. |

|
Arrhythmia | Increases production of SOD | [122,123,124] |
| Orientin | Millettia nitida Benth. |

|
Coronary heart disease, atherosclerosis | Reduces ROS | [125,126,127] |
| Paeonol |
Paeonia suffruticosa Andrews |

|
Arrhythmia, coronary heart disease | Inhibits free radical reaction | [122,128] |
| Polysaccharides | Astragalus propinquus Schischk. | Coronary heart disease, acute myocardial infarction | Inhibits the expression of NOX | [129] | |
| Quercetin | Dendrobium nobile Lindl. |

|
Acute myocardial infarction, ischemia Reperfusion |
Reduce ROS | [130] |
| Tanshinone II-A |
Salvia miltiorrhiza Bunge. |

|
Coronary heart disease, acute myocardial infarction | Regulates Nrf2/ARE/HO-1 and TGF-beta1/signal transduction | [131,132] |
| Tetramethylpyrazine | Ligusticum chuanxiong |

|
Heart failure, coronary heart disease | Increases the activity of SOD, CAT and GSH-Px | [133,134] |
[LPO: lipid peroxidation; MDA: malondialdehyde; O2: oxygen; H2O2: hydrogen peroxide; bax: Bcl-2-associated X protein; ap-JNK: activator protein-1 c-Jun N-terminal kinase; AMPK: AMP-activated protein kinase; Akt: protein kinase B; mTOR: mechanistic target of rapamycin; Bcl-2: B-cell lymphoma 2; Bad: Bcl-2-associated death promoter; PI3K: phosphoinositide 3-kinase; SOD: superoxide dismutase; ROS: reactive oxygen species; NOX: NADPH oxidase; Nrf2: nuclear factor erythroid 2-related factor 2; ARE: antioxidant response element; HO-1: heme oxygenase 1; TGF-beta1: transforming growth factor beta 1; CAT: catalase; GSH-Px: glutathione peroxidase].
3.2. Neurodegenerative Disorders
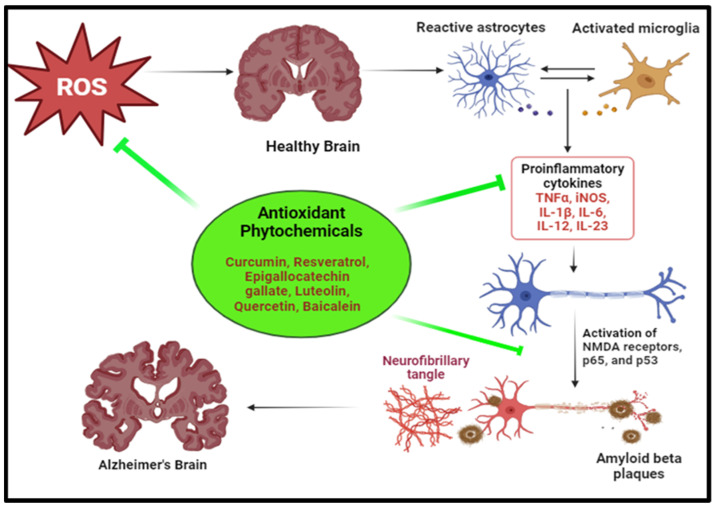
Neurodegenerative disorders involve the loss of functional capacity and eventual dysfunction or death of neuronal cells in the brain [135]. Diseases like Parkinson’s and Alzheimer’s are characterized by neurodegeneration, and oxidative stress plays a major role in their pathogenesis [136]. The high level of ROS generation and low antioxidant levels in brain cells make them susceptible to oxidative damage, which alters the function of lipids, DNA and proteins, contributing to neurodegeneration (Figure 5) [137,138].
Figure 5.
Oxidative stress-induced neurodegenerative disease pathology and modulation by antioxidant phytochemicals. (the illustration was created using BioRender at www.biorender.com). [TNFα: tumour necrosis factor alpha, iNOS: inducible nitric oxide synthase, IL-1β: interleukin-1 beta, IL-6: interleukin-6, IL-12: interleukin-12, IL-23: interleukin-23, NMDA: N-methyl-D-aspartate, p65: RelA (a subunit of the NF-κB transcription factor), p53: tumour protein p53].
In Alzheimer’s disease, ROS stimulate the cleavage of amyloid precursor protein (APP), enhancing the production of Aβ peptides which aggregates to form toxic Aβ plaques. [139]. During oxidative stress, ROS induces activation of kinases and inhibition of phosphatases leading dysregulate tau phosphorylation dynamics which destabilizes microtubules and leads to their aggregation into neurofibrillary tangles [140]. ROS overwhelm the endogenous antioxidant defence system, which amplifies oxidative damage and potentiates neuronal vulnerability. The activation of microglia initiates an inflammatory cascade, which starts a pro-inflammatory cytokine release and causes exacerbate neuroinflammation, contributing to neuronal dysfunction and degeneration [32]. Sequential lipid peroxidation generates breakdown products like 4-hydroxy-2,3-nonenal (HNE), elevated levels of which, in brain tissues, is indicative of Alzheimer’s disease [141,142].
Parkinson’s disease, the second most common neurodegenerative disorder in elderly individuals, on the other hand, primarily affects the motor functions of the body, leading to noticeable movement disorders. OS promotes the misfolding of α-synuclein protein, which aggregates to form Lewy bodies. PD is linked with increased levels of HNE in brain tissues. Increased levels of 8-hydroxyguanine and 8-hydroxy-2-deoxyguanosine, resulting from oxidative damage to DNA base pairs, are also indicative of Parkinson’s disease [33,139].
Several potent phytochemicals have shown potential in combating neurodegenerative diseases, offering avenues for novel therapeutic interventions [143] (Table 3). For Alzheimer’s disease, compounds like curcumin, found in turmeric, exhibit anti-inflammatory and antioxidant properties, inhibiting the formation of beta-amyloid plaques and reducing neuroinflammation [144].
Resveratrol, abundant in red grapes and berries, demonstrates neuroprotective effects by modulating signalling pathways involved in neuronal survival and reducing oxidative stress [145]. Similarly, flavonoids, such as EGCG found in green tea, and quercetin, which is abundant in onions and apples, possess neuroprotective properties by scavenging free radicals and inhibiting neuroinflammation [146].
In Parkinson’s disease, phytochemicals, like polyphenols, particularly found in berries, cocoa, and grapes, exhibit neuroprotective effects by enhancing mitochondrial function, reducing oxidative stress, and inhibiting alpha-synuclein aggregation [147]. Additionally, compounds, like sulforaphane, present in cruciferous vegetables, activate cellular defence mechanisms against oxidative stress and inflammation, potentially mitigating neuronal damage in Parkinson’s disease [148].
Phytochemical compounds have also been found to decrease the risk of 4-hydroxy-2,3-nonenal (HNE) aggregation, a reactive aldehyde produced during oxidative stress and implicated in various neurodegenerative diseases. For instance, polyphenolic compounds, such as curcumin, found in turmeric, and resveratrol, abundant in red grapes and berries, have been shown to inhibit HNE-induced protein aggregation and lipid peroxidation [149]. Additionally, flavonoids, like EGCG from tea and quercetin from onions and apples, have demonstrated protective effects against HNE-induced toxicity by modulating cellular signalling pathways and enhancing antioxidant defences. These compounds possess strong antioxidant properties, scavenging free radicals and mitigating oxidative damage and thereby reducing the formation of HNE adducts and subsequent aggregation [150].
Table 3.
Phytochemicals, along with their high-altitude plant sources, reported to mitigate oxidative stress-induced neurodegenerative disorders [151].
| Phytochemicals | Plant | Structure | Mode of Action | Reference |
|---|---|---|---|---|
| 1,8-Cineole | Salvia officinalis L. |

|
Selectively suppresses NF- κB and activation of pro-inflammatory gene expression and cytokine production, enhances neurogenesis | [152] |
| Asiatic acid | Centella asiatica (L.) urban |

|
Inhibits pro-inflammatory cytokines and inflammatory pathway and promotes neurogenesis | [153,154] |
| Asiaticoside | Centella asiatica (L.) urban |

|
Inhibits pro-inflammatory cytokines | [155,156] |
| Bacoside A | Bacopa monniera (L.) Pennel |

|
Reduces oxidative stress-induced neuronal damage, enhances cholinergic neurotransmission, improves cognitive function, inhibits pro-inflammatory cytokines, inhibits amyloid-beta (Aβ) peptide aggregation, and promotes synaptic remodelling | [157,158] |
| Baohuoside I | Centella asiatica (L.) urban |

|
Promotes the antioxidant activity of essential enzyme such as SOD, CAT and GSH-Px. | [159] |
| Betulic acid | Centella asiatica (L.) urban |

|
Inhibiting pro-inflammatory cytokines and signalling pathways and promotes neurotrophic factor BDNF expression contributing to overall brain health | [160] |
| Borneol | Salvia officinalis L. |

|
Exhibits antioxidant properties and suppresses pro-inflammatory cytokine production | [161] |
| Brahmic acid | Centella asiatica (L.) urban |

|
Promotes neurogenesis; modulates neurotransmitter levels, including acetylcholine, serotonin, and dopamine; and reduces the production of pro-inflammatory cytokines | [155] |
| Camphor | Salvia officinalis L. |

|
Exhibits antioxidant properties and suppresses NF-κB activation and pro-inflammatory cytokine production | [162] |
| Caryophyllene | Salvia officinalis L. |

|
Demonstrates anti-inflammatory activity, modulates neurotransmitter systems and enhances neurogenesis | [163] |
| Herpestine | Bacopa monniera (L.) Pennel |

|
Enhances neuronal synthesis, increases kinase activity, and restores synaptic activity and nerve impulse transmission | [164] |
| Linalool | Salvia officinalis L. |

|
Scavenges free radicals, suppresses NF-κB activation and pro-inflammatory cytokine production, modulates neurotransmitter systems and enhances neurogenesis |
[152] |
| Luteolin | Picrorhiza scrophulariiflora Pennell. |

|
Reduces neuroinflammation, promotes expression of brain-derived neurotrophic factor (BNDF) and modulates neurotransmitter systems, such as dopamine and serotonin | [165] |
| Madecassic acid | Centella asiatica (L.) urban |

|
Inhibits pro-inflammatory cytokines and signalling pathways and promotes neurotrophic factors’ BDNF expression | [160,166] |
| Picroside II | Picrorhiza scrophulariiflora Pennell. |

|
Inhibits neuronal apoptosis | [167] |
[NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; SOD: superoxide dismutase; CAT: catalase; GSH-Px: glutathione peroxidase; BDNF: brain-derived neurotrophic factor].
3.3. Metabolic Disorders: Diabetes and Obesity
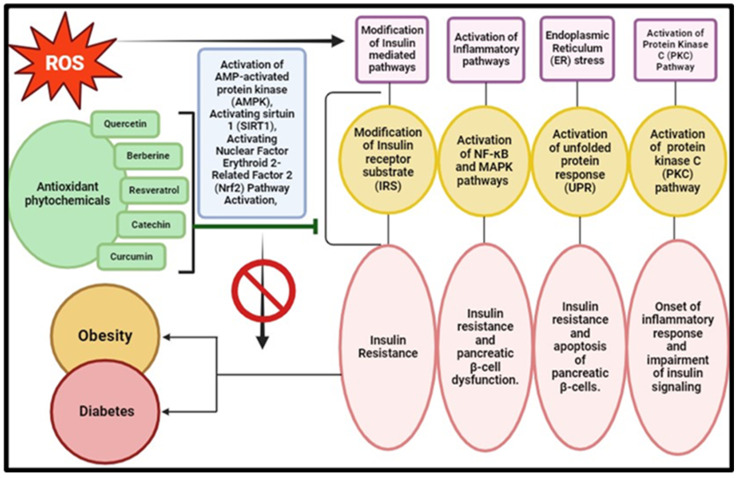
Metabolic disorders, including obesity and diabetes, are closely linked with the generation of ROS in the body [8]. Studies have shown a positive correlation between decreased levels of high-density lipoproteins (HDLs) and increased levels of low-density lipoproteins (LDLs) with oxidative stress. Lower levels of HDLs result in dysfunctional antioxidant defence mechanisms, leading to elevated oxidative stress (Figure 6) [168,169]. OS is also implicated in obesity as excessive ROS production acts as a trigger for abnormal amplification and enlargement of pre-adipocytes and adipocytes. This abnormal adipose cell growth leads to adipogenesis, a fundamental factor in obesity [37]. Improper dietary patterns, including high carbohydrate and high-fat diets, can increase oxidative stress in the body, contributing to obesity [170].
Figure 6.
Antioxidant phytochemicals and modulation of oxidative stress-induced metabolic disorders (obesity and diabetes) (the illustration was created using BioRender at www.biorender.com). [AMPK: AMP-activated protein kinase; SIRT1: sirtuin 1; Nrf2: nuclear factor erythroid 2-related factor; 2NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; MAPK: mitogen-activated protein kinase; IRS: insulin receptor substrate; UPR: unfolded protein response; PKC: protein kinase C].
Diabetes, on the other hand, characterized by high glucose levels and decreased insulin sensitivity, is another metabolic disorder linked to oxidative stress [36]. Mitochondrial dysfunction resulting from oxidative stress contributes significantly to insulin resistance, impairing insulin responses and leading to abnormal glucose levels [171]. Oxidative radicals also promote apoptosis in pancreatic beta-cells, modifying cell cycle regulators and contributing to the diabetes development [12]. In type 2 diabetes, islet inflammation causing pancreatic β cell dysfunction underscores inflammation’s significance [172]. Concurrently, oxidative stress in people with diabetes and obesity plays a major role in causing cardiovascular associated complications as well [173].
Plant products have been gaining attention for the potential mitigation of metabolic disorders by modulating proinflammatory cytokines and ROS. Methanolic extracts from Capparis spinosa L. leaves show in vitro anti-inflammatory effects, inhibiting membrane destabilization, and exerting anti-inflammatory effects in murine models [174]. Plant secondary metabolites like carotenoids and alkaloids induce an anti-inflammatory response by suppressing IL-17 and inducing IL-4 gene expression [175]. With concerns about synthetic antioxidants’ long-term safety, there is rising demand for natural antioxidants to mitigate oxidative stress-related diseases [176]. Recognized as rich in essential antioxidants, plants are increasingly viewed as functional ingredients promoting health. Plant-derived products, including phytochemicals, emerge as a valuable natural source of anti-inflammatory agents with potential therapeutic implications for metabolic disorders (Table 4). For instance, curcumin, resveratrol, quercetin, epigallocatechin gallate, berberine, and alpha-lipoic acid have garnered significant attention for their potential in mitigating oxidative stress-related metabolic dysregulations [177]. Curcumin exerts its effects through NF-κB pathway inhibition, activation of the Nrf2 pathway, and modulation of insulin signalling, thereby offering therapeutic benefits in diabetes, obesity, and cardiovascular diseases [178]. Similarly, resveratrol activates sirtuin 1 (SIRT1), possesses antioxidant activity, and activates AMP-activated protein kinase (AMPK), contributing to its medicinal properties against metabolic disorders [179]. Quercetin scavenges free radicals, modulates inflammatory pathways, and enhances mitochondrial function, making it beneficial for metabolic health. EGCG exhibits antioxidant activity, regulates insulin signalling, and modulates adipocyte function, thereby improving metabolic parameters in various disorders [180]. Berberine activates AMPK, modulates gut microbiota, and inhibits inflammatory pathways, offering therapeutic potential in metabolic disorders [181]. Alpha-lipoic acid exerts antioxidant effects, regulates mitochondrial function, and modulates insulin signalling, contributing to its efficacy against metabolic dysregulations [182]. Overall, understanding the mechanistic insights into these plant bioactive compounds is crucial for developing targeted strategies to combat oxidative stress-related metabolic disorders and improve public health outcomes.
Table 4.
Phytochemicals, along with their high-altitude plant sources, as treatment options against metabolic disorders [183].
| Phytochemical | Plant | Chemical Structure | Mode of Action | Reference |
|---|---|---|---|---|
| Anthocyanin | Aristotelia chilensis (Molina) Stuntz |

|
Inhibits synthesis of the pro-inflammatory cytokines, TNF-α and IL-6, further reducing inflammation associated with diabetes and obesity, and modulates the NF-κB signalling pathway, leading to decreased expression of inflammatory mediators | [184] |
| Ascorbic acid | Rosehips produced by Rosa pendulina L. |

|
Enhances insulin sensitivity, facilitating the uptake of glucose into cells; reduces risk of hyperglycaemia; and modulates lipid metabolism by reducing lipid peroxidation and inhibiting fatty acid synthesis, which prevents dyslipidemia | [185,186] |
| Caffeine | Ilex guayusa Loes. |

|
Stimulates lipolysis and thermogenesis, caffeine may help reduce circulating levels of LDL cholesterol and triglycerides, thereby preventing the development of atherosclerotic plaques | [187] |
| Niazirin | Moringa oleifera Lam. |

|
Helps regulate lipid metabolism, reducing the level of triglyceride and LDL cholesterol while increasing the production of HDL cholesterol; modulates lipid metabolism and helps prevent the formation of atherosclerotic plaques; and maintains vascular health in diabetic individuals. | [188,189] |
| Proanthocyanidins | Vitis vinifera L. |

|
Promotes endothelial NO production, leading to vasodilation and improved blood flow; inhibits endothelial cell apoptosis and preserve vascular homeostasis; prevents formation of atherosclerotic plaques; and maintains cardiovascular health | [190] |
| Phenolic acids (Protocatechuic acid) and saponins | Androsace umbellata (Lour.) Merr. |

|
Promotes the production of serum antioxidant enzymes, upregulates the expression of hepatic antioxidant genes, and inhibits the NF-κB signalling pathway, leading to the decreased expression of inflammatory mediators | [191,192] |
[TNF-α: tumour necrosis factor alpha; IL-6: interleukin 6; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; NO: nitric oxide; LDL: low-density lipoprotein; HDL: high-density lipoprotein].
4. High-Altitude Medicinal Plants: Bulk Producers of Antioxidants
Plants that thrive in high-altitude environments face numerous challenges due to extreme environmental conditions, such as low carbon dioxide and oxygen levels, intense mutagenic radiation, and drastic temperature fluctuations. These factors create a harsh survival environment for plants. ROS production is heightened in these plants, leading to cellular damage and impairing photosynthesis. In response to these conditions, plants have developed adaptive mechanisms to counteract the negative effects of oxidative stress caused by ROS [193].
High-altitude plants have evolved the ability to synthesize secondary metabolites in large quantities. These metabolites, including flavonoids, phenols, tannins, and other compounds, serve as antioxidants within the plants [47]. By accumulating these secondary metabolites, plants can adapt to the extreme environmental conditions and mitigate the harmful effects of ROS-induced oxidative damage. This defence mechanism helps these plants maintain their cellular integrity and sustain their growth and survival in such harsh environments [194].
4.1. Environmental Factors Influencing Antioxidant Production in High-Altitude Medicinal Plants
High-altitude regions, characterized by unique environmental conditions, present challenges as well as opportunities for plant life. The synthesis of antioxidant phytochemicals in high-altitude medicinal plants is influenced by several environmental factors:
4.1.1. Solar Radiation Intensity and Ultraviolet (UV) Exposure
High-altitude regions often experience increased solar radiation due to reduced atmospheric filtration. Elevated UV radiation levels can lead to oxidative stress in plant tissues by generating ROS. Plants respond by activating antioxidant defence mechanisms modulated by flavonoids such as the production of quercetin, kaempferol, catechins and others to scavenge ROS and protect cellular components [195].
4.1.2. Temperature Fluctuations
High-altitude environments exhibit significant diurnal temperature variations, including cold nights and warm days. Temperature fluctuations can disrupt cellular homeostasis and induce oxidative stress. High-altitude plants adapt by synthesizing antioxidant compounds such as chlorogenic acid, a phenolic compound, to mitigate temperature-induced oxidative damage and maintain cellular integrity [96].
4.1.3. Low Oxygen Levels (Hypoxia)
Reduced atmospheric pressure at higher elevations results in lower oxygen levels, leading to hypoxic conditions. Hypoxia-induced oxidative stress can occur due to impaired mitochondrial function and increased ROS production. High-altitude plants enhance the production of alkaloid antioxidants such as berberine to counteract hypoxia-induced oxidative damage [96].
4.1.4. Water Scarcity and Drought Stress
Water availability in high-altitude regions can be limited, particularly in arid or semi-arid environments. Drought stress disrupts cellular hydration and photosynthetic processes, triggering oxidative stress. High-altitude plants accumulate osmo-protectants, such as proline and antioxidants, such as flavonoids to mitigate water stress-induced oxidative damage and maintain cellular hydration [196].
4.1.5. Soil Composition and Nutrient Availability
High-altitude soils often exhibit low nutrient availability, high acidity, and metal-rich compositions. Adverse soil conditions can exacerbate oxidative stress in plants by limiting nutrient uptake and promoting metal-induced ROS generation. High-altitude plants produce metal chelators that bind and detoxify heavy metals present in the soil, reducing metal-induced oxidative stress. Polyphenols scavenge ROS and regulate nutrient uptake, contributing to antioxidant defence and nutrient homeostasis [197].
4.1.6. Altitude-Dependent Factors
Altitude-specific variables, including atmospheric pressure, humidity, and air pollution, influence antioxidant production in high-altitude plants. Changes in atmospheric pressure and humidity modulate plant metabolism and ROS production, while air pollutants like ozone and nitrogen oxides contribute to oxidative stress. High-altitude plants adjust their antioxidant defences, such as the production of terpenoids, which exhibit adaptogenic properties, enhancing plant resilience to altitude-dependent stressors like changes in atmospheric pressure and humidity. Anthocyanins act as antioxidants and UV protectants, shielding plant tissues from oxidative damage and UV radiation at high altitudes [96].
4.2. High-Altitude Plants and Their Antioxidant Potential
Plants have held a significant role in the field of medicine since ancient times [198]. Various plant species, such as Tulsi and Neem, have been recognized for their beneficial effects on human health, functioning as antibacterial, anti-inflammatory, and antioxidant agents [199]. In particular, certain plants found in the high-altitude regions possess unique properties and produce phytochemicals and essential oils, rich in phenolic compounds and flavonoids etc. These phytochemicals have the ability to scavenge free radicals through various mechanisms, such as electron donation, hydrogen atom donation, acting as reducing agents, or chelation of metal ions [82]. By employing these strategies, they effectively neutralize harmful free radicals, thereby providing antioxidant protection. These natural compounds hold great promise in the field of drug discovery, as they serve as botanical leads for the development of novel therapeutic agents. The following section describes selected high-altitude medicinal plants along with their antioxidant potential.
4.2.1. Saussurea lappa (Decne.) C. B. Clarke
Saussurea lappa (Decne.) C. B. Clarke is a medicinal plant that belongs to the Asteraceae family and is predominantly found at high altitudes, ranging from 2500 to 3500 m above mean sea level, primarily in the Himalayan region [200]. It is commonly referred to as ‘Costus’ and has garnered significant attention due to its extensive medicinal applications. Notably, this plant is enriched with essential vitamins, including vitamin B12, vitamin B2, vitamin A, as well as vital minerals such as calcium, iron, and zinc [201].
A distinctive feature of Costus is the presence of a phytochemical called costunolide, which is primarily found in its roots [201]. Costunolide exhibits remarkable antioxidant activity, which has been attributed to its ability to counteract the development of cancer [202]. The compound contains N-acetylcysteine, which plays a pivotal role in neutralizing ROS by facilitating the production of key enzymes like SOD and CAT [203]. These aid in the detoxification of harmful free radicals, thereby contributing to the plant’s antioxidant defence system. Through its antioxidant properties, Costus holds potential as a therapeutic agent in the prevention and management of various diseases [204].
4.2.2. Arnebia benthamii (Wall. ex G. Don) I. M. Johnst.
Arnebia, scientifically known as Arnebia benthamii (Wall. ex G. Don) I. M. Johnst., is a highly valued medicinal plant belonging to the Boraginaceae family. It thrives in high-altitude Himalayan regions, specifically ranging from 3000 to 3900 m above mean sea level [205]. However, it is important to note that this plant has been classified as a critically endangered species in the Northwestern Himalayas by the International Union for Conservation of Nature (IUCN) [206]. Himalayan Arnebia possess various phytochemicals, including a prominent compound called shikonin [207]. Shikonin plays a vital role in preventing oxidative DNA damage through its free radical scavenging mechanism. As a quinone derivative, shikonin acts as a potent antioxidant, effectively thwarting lipid peroxidation and DNA damage by neutralizing free radicals and reducing ferrous ions [82].
The antioxidative properties of shikonin contribute to the overall preservation of cellular integrity, providing a protective shield against oxidative stress. The presence of shikonin in Himalayan Arnebia underscores its medicinal significance and potential therapeutic applications [208]. Studies have highlighted the antioxidant capabilities of this plant, shedding light on its role in preventing oxidative damage and maintaining cellular health [209,210].
4.2.3. Pinus nigra Aiton, Hort. Kew. [W. Aiton]
Belonging to the Pinaceae family, this particular plant species thrives in the high-altitude regions (2000 m above mean sea level) of the Toros mountains and holds a great significance in combatting oxidative stress-induced damage [211]. This plant abundantly produces phenols and flavonoids, which are extremely efficient in neutralizing several free radical species, including hydrogen peroxides and superoxide free radical species, including superoxide radicals and hydrogen peroxide. This antioxidant activity is facilitated through multiple mechanisms, such as chelation of metal ions, free radical scavenging and the reduction of ferrous ions [212].
The presence of phenols and flavonoids in this plant demonstrates its adaptation to cope with the challenging environmental conditions it encounters. By effectively neutralizing free radicals, these compounds help protect the plant’s cellular components from oxidative damage and maintain their functionality. Studies have highlighted the antioxidative properties of this plant, shedding light on its potential role in preventing oxidative stress-related disorders [213].
4.2.4. Cedrus deodara (Roxb. ex D. Don) G. Don
Cedrus deodara, also known as the Deodar cedar, is a significant plant that has been used in Ayurveda for its medicinal benefits [214]. It is an evergreen plant found at high altitudes, specifically around 3000—3300 m above mean sea level [214]. Belonging to the family Pinaceae, this plant contains phytochemicals, such as ‘Metairesinol,’ which exhibit antioxidant activity [215]. These phytochemicals help in inhibiting oxidative stress by chelating metal ions or transferring hydrogen atoms to free radical species [212].
4.2.5. Podophyllum hexandrum Royle
Podophyllum hexandrum, also known as Himalayan May apple, is found at an altitude of around 3000—3500 m above mean sea level [216]. It belongs to the Berberidaceae family and exhibits high antioxidant activity due to phytochemicals, such as podophyllotoxin, present in its rhizome, leaves, and other parts [216]. The extracts of this plant are capable of neutralizing hydrogen peroxide and superoxide radicals, thus preventing lipid peroxidation, and also stimulate the activity of antioxidant enzymes [217].
4.2.6. Valeriana jatamansi D. Don
Valeriana jatamansi, known as Mushkibala in Hindi, is a high-altitude medicinal plant found at an altitude of around 3000 m above mean sea level [218]. It belongs to the Valerianaceae family and possesses antiseptic and antioxidant properties [218]. It contains a class of terpenoids called valepotriates, which are responsible for its medicinal applications [219]. The rhizome of Valeriana jatamansi contains phenols and flavonoids, which exhibit antioxidant activity by donating hydrogen atoms or quenching singlet oxygen species. It can also chelate certain metal ions, thereby inhibiting the generation of ROS [220].
4.2.7. Berberis aristata DC.
Berberis aristata, also known as Daru Haldhi, is a Himalayan shrub found at an altitude of around 2000—3000 m above sea level. It is primarily found in the areas of Himachal Pradesh, Nepal, and Sri Lanka [221]. Berberis aristata possesses antioxidant potential attributed to certain protoberberines present in its root and shoot extracts. These compounds contribute to the neutralization of ROS, reducing the risk of oxidative stress-related issues such as hepatic damage [222].
4.2.8. Pedicularis longiflora Rudolph
Pedicularis longiflora is a plant widely found in the Himalayan regions of Ladakh, Jammu and Kashmir, at an altitude of approximately 2700 m above mean sea level [223]. This plant is valued for its medicinal properties, particularly its antioxidant and anti-inflammatory effects [224]. It contains phytochemicals such as flavonoids and phenols, which reduce lipid peroxidation by scavenging superoxide radicals. Moreover, Pedicularis longiflora enhances the activity of CAT and SOD, further contributing to its antioxidant activity [224].
4.2.9. Aconitum heterophyllum Wall. ex Royle
Aconitum heterophyllum, also known as Indian aconite or Atees, is an Ayurvedic medicinal plant native to the Himalayan region, including Jammu and Kashmir, Nepal, Sikkim, and Uttarakhand, at altitudes ranging from 2500 to 4000 m above mean sea level [225]. It belongs to the Ranunculaceae family. The roots, stems, and leaves of this plant contain alkaloids and flavonoids, which play a crucial role in detoxifying ROS within the body [226]. These compounds contribute to the prevention of gastrointestinal problems such as liver inflammation [227].
4.3. Underutilization of High-Altitude Medicinal Plants
High-altitude regions, defined as areas above 1500 m (4900 feet) elevation, encompass diverse ecosystems ranging from alpine meadows to snow-capped peaks. These regions are home to a rich array of medicinal plants that have been traditionally used by indigenous communities for centuries to treat various ailments [228]. The harsh environmental conditions of high-altitude environments, including intense solar radiation, extreme temperatures, and oxidative stress, have driven the evolution of plants towards unique biochemical compositions and pharmacological properties. For instance, one example of a high-altitude medicinal plant with potent antioxidant properties is Rhodiola rosea, also known as golden root or arctic root. Indigenous to mountainous regions of Europe and Asia, Rhodiola rosea has been traditionally used to increase resistance to physical and environmental stress, enhance mental performance, and promote longevity. Studies have attributed its adaptogenic and antioxidant effects to bioactive compounds, including salidroside, rosavin, and flavonoids, which scavenge free radicals, reduce oxidative damage, and modulate stress-responsive pathways [14]. Similarly, Berberis aristata, a high-altitude plant native to the Himalayas, is valued for its medicinal properties, including its antioxidant, anti-inflammatory, and hepatoprotective effects. Berberis aristata contains bioactive alkaloids, such as berberine, palmatine, and berbamine, which exhibit potent antioxidant activity by neutralizing ROS, inhibiting lipid peroxidation, and enhancing cellular antioxidant enzymes [229].
However, despite their immense potential therapeutic benefits, a large variety of these plants, and the products they produce, remain largely underutilized in modern medicine. Several factors contribute to this underutilization [9]. Firstly, there is a lack of comprehensive scientific research exploring the antioxidant potential of high-altitude medicinal plants. Limited funding and resources are allocated to studying plants in remote mountainous regions, making it difficult to gather robust scientific evidence to support their medicinal properties. As a result, many of these plants remain overlooked in pharmaceutical and nutraceutical industries. Additionally, challenges in accessing high-altitude environments pose logistical difficulties for researchers. Harsh terrain, extreme weather conditions, and limited infrastructure make it challenging to conduct field studies and collect plant samples. This impedes efforts to characterize the bioactive compounds and pharmacological activities of high-altitude medicinal plants [230].
Furthermore, traditional knowledge of these plants is at risk of being lost as the young population inhabiting mountainous regions tends to migrate to urban areas and adopt modern lifestyles. The decline of indigenous knowledge and traditional healing practices contributes to the under appreciation of high-altitude medicinal plants in mainstream healthcare systems [231]. This necessitates the compilation of knowledge of important medicinal plants thriving in high-altitude regions, along with their reported bioactive compounds and their reported medicinal applications. Table 5 provides a comprehensive compilation of 168 such plants of medicinal value, which are able to survive in high-altitude regions, along with their ethnopharmacological applications. A large majority of these plants are unexplored and have not been utilized to their full potential. The comprehensive detailed analysis of their phytochemicals could act as starting point for the exploration of their potential to mitigate oxidative stress-related disorders.
Table 5.
List of high-altitude medicinal plants, along with their reported bioactive compounds and their pharmacological properties.
| S. No. | Plant Name | Plant Family | Altitude (m above m.s.l.) | Parts Used | Principle Bioactive Compound | Pharmacological Activity | Reference |
|---|---|---|---|---|---|---|---|
|
Allium humile Kunth | Amaryllidaceae | 3200–4500 | Whole plant | Allicin | Antioxidant | [232] |
|
Allium semenovii Regel. | 2000–3000 | Whole plant | Alliin | Antioxidant | [233] | |
|
Allium stoliczki Regel | 3200–3700 | Bulbs | S-Allyl-L-cysteine sulfoxide | Antioxidant, Cardiovascular health benefits | [234] | |
|
Pistacia integerrima L. | Anacardiaceae | 800–2200 | Fruits | Gallic acid, Quercetin | Antioxidant, Anti-inflammatory |
[235] |
|
Angelica glauca Edgew. | Apiaceae | 2000–3800 | Roots | Angelicin, Umbelliferone |
Antioxidant, Hepatoprotective |
[236] |
|
Bupleurum falcatum L | 2130–3500 | Roots | Saikosaponins | Anti-inflammatory, Hepatoprotective |
[237] | |
|
Chaerophyllum aromaticum L. | 2800–3200 | Roots | Coumarin, Umbelliferone |
Antioxidant, Anti-inflammatory |
[238] | |
|
Ferula jaeschkeana Vatke | 2600–3000 | Rhizomes | Ferutinin, Ferulenol | Antioxidant | [239] | |
|
Heracleum candicans L. | 1800–4000 | Leaves, Stem Roots | Bergapten, Psoralen | Antioxidant, Anti-inflammatory |
[240] | |
|
Pleurospermum brunonis Benth. ex C.B Clarke | 3000–4000 | Leaves | Psoralen, Isopsoralen | Antioxidant, Anti-inflammatory |
[241] | |
|
Selinum vaginatum C.B. Clarke | 2700–3800 | Roots Bhutkeshi | Selinidin, Selinidiol | Antioxidant, Anti-inflammatory |
[242] | |
|
Arisaema flavum (Forsk.) Schott. | Araceae | 2000–3400 | Rhizome | Arisarumol | Antioxidant, Anti-inflammatory |
[243] |
|
Hedera nepalensis C. Koch | Araliaceae | 1500–3000 | Leaves, Stems | Hederacoside C, Hederagenin | Antioxidant, Anti-inflammatory |
[244] |
|
Achillea millefolium L. | Asteraceae | 3200–3700 | Leaves, Flowers |
Apigenin, Luteolin | Antioxidant, Anti-inflammatory |
[245] |
|
Artemisia absinthium L. | 2000–3660 | Whole plant | Absinthin, Anabsinthin |
Antioxidant | [246] | |
|
Artemisia macrocephala Jacq. ex Bess | 3400–5500 | Aerial parts | Artemisinin, Dihydroartemisinin |
Antioxidant, Anticancer | [247] | |
|
Carduus nutans L. | 2600–3000 | Leaves, Roots | Silymarin | Hepatoprotective, Antioxidant |
[248] | |
|
Cichorium intybus L. | 2600–3000 | Leaves, Roots | Inulin, Lactucin | Hepatoprotective, Hypoglycemic |
[249] | |
|
Erigeron acris L. | 2600–3400 | Roots | Quercetin, Kaempferol | Anti-inflammatory, Antioxidant |
[250] | |
|
Inula cappa DC. | 2600–3500 | Roots | Alantolactone, Isoalantolactone |
Antioxidant, Anti-inflammatory |
[251] | |
|
Inula racemosa Hook. f. | 2000–3100 | Roots | Alantolactone, Isoalantolactone |
Antioxidant, Anti-inflammatory |
[252] | |
|
Jurinea dolomiaea Boiss. | 3000–4000 | Roots | Jurineol, Jurineol acetate |
Antioxidant, Anti-inflammatory |
[253] | |
|
Jurinea macrocephala DC. | 3000–4000 | Roots Leaves | Jurineol, Jurineol acetate |
Antioxidant, Anti-inflammatory |
[254] | |
|
Saussurea albescens Hook. f. et. Thomson | 2000–3600 | Leaves | Costunolide, Eupatilin | Antioxidant, Anti-inflammatory |
[255] | |
|
Saussurea costus (Falc.) Lipsch. |
2600–4000 | Roots | Costunolide, Dehydrocostus lactone | Antioxidant, Anti-inflammatory |
[256] | |
|
Saussurea gossypiphora D. Don | 4500–5300 | Flowers | Saussureamine | Antioxidant, Anti-inflammatory |
[257] | |
|
Scorzonera virgata DC. | 2700–4200 | Leaves | Inulin, Scorzodioside B |
Hepatoprotective, Hypoglycemic |
[258] | |
|
Waldhemia glabra (Decne.) Regel. | 4000–5000 | Aerial parts | Waldhemiol, Waldhemidin |
Antioxidant, Anti-inflammatory |
[259] | |
|
Waldhemia tomentosa (Decne.) Regel. | 3800–4500 | Whole plant | Waldhemiol, Waldhemidin |
Antioxidant, Anti-inflammatory |
[260] | |
|
Impatiens sulcata Wall. | Balsaminaceae | 2000–3900 | Whole plant | Lawsone | Antioxidant, Anti-inflammatory |
[261] |
|
Berberis lycium Royle | Berberidaceae | 1200–3000 | Roots, stems | Berberine, Palmatine | Antioxidant, Antidiabetic | [262] |
|
Betula utilis D. Don | Betulaceae | 2900–4000 | Bark | Betulin, Betulinic acid | Antioxidant, Anti-inflammatory |
[263] |
|
Biebersteinia odora Steph. ex Fish | Biebersteiniaceae | 4200–5030 | Rootstocks | Coumarin, Umbelliferone |
Antioxidant, Anti-inflammatory |
[264] |
|
Arnebia benthamii (Wall. ex G. Don.) Johnston | Boraginaceae | 3000–3900 | Roots | Alkannin, Shikonin | Antioxidant, Anti-inflammatory |
[265] |
|
Cynoglossum wallichii G. Don | 2600–3700 | Leaves | Shikonin, Deoxyshikonin |
Antioxidant, Anti-inflammatory |
[266] | |
|
Cynoglossum zeylanicum Thunb. ex Lehm. Brand. | 2600–3350 | Roots | Shikonin, Deoxyshikonin |
Antioxidant, Anti-inflammatory |
[266] | |
|
Myosotis silvatica Ehrh. ex Hoffm. | 3200–4200 | Whole plant | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[267] | |
|
Onosma hispida Wall. ex G. Don | 2000–3400 | Roots, Leaves | Alkannin, Shikonin | Antioxidant, Anti-inflammatory |
[268] | |
|
Arabidopsis mollissma (C. May.) N. Busch | Brassicaceae | 3800–4300 | Leaves | Sinapine, Sinapic acid | Antioxidant, Anti-inflammatory |
[269] |
|
Arabis nova Vill. | 3500–3900 | Fruits | Glucosinolates | Antioxidant, Anticancer | [270] | |
|
Brassica rapa L. ssp. | 3200–4500 | Whole plant | Glucosinolates | Antioxidant, Anticancer | [271] | |
|
Descurainia sophia (L.) Webb. ex Prantl | 2600–3500 | Whole plant | Linalool, Thymoquinone |
Antioxidant, Anti-inflammatory |
[272] | |
|
Lepidium latifolium L. | 2500–4300 | Aerial parts | Glucosinolates | Antioxidant | [273] | |
|
Nasturtium officinale W.T. Ait. Hort. | 2600–3500 | Whole plant | Glucosinolates | Antioxidant | [274] | |
|
Sisymbrium orientale L. | 2600–3600 | Seeds | Glucosinolates | Antioxidant | [275] | |
|
Sarcococca saligna (D. Don) Muell.-Arg. | Buxaceae | 1500–2300 | Leaves, Stem | Sarcococcin | Antioxidant, Anti-inflammatory |
[276] |
|
Codonopsis clematidea (Schrenk) C.B. Clarke | Campanulaceae | 3000–3800 | Flowers | Codonopsin, Codonopsidic acid |
Antioxidant, Immunomodulatory |
[277] |
|
Codonopsis ovata Benth. | 2700–3200 | Whole plant | Codonopsin, Codonopsidic acid |
Antioxidant, Immunomodulatory |
[278] | |
|
Cyananthus lobatus Wall. ex Benth | 3000–4000 | Leaves, flowers | Cyanolobatolide | Antioxidant, Anti-inflammatory |
[279] | |
|
Capparis himalayensis Jafri | Capparaceae | 2800–3300 | Leaves | Flavonoids, Glucosinolates |
Antioxidant | [280] |
|
Lonicera hypoleuca Decne. | Caprifoliaceae | 2900–3100 | Stem | Chlorogenic acid, Luteolin |
Antioxidant, Anti-inflammatory |
[281] |
|
Lonicera quinquelocularis Hardw. | 2600–3500 | Stems, Leaves, Fruit | Chlorogenic acid, Luteolin |
Antioxidant, Anti-inflammatory |
[282] | |
|
Viburnum cotinifolium D. Don | 2300–2600 | Fruits | Iridoids, Flavonoids | Antioxidant, Anti-inflammatory |
[283] | |
|
Viburnum grandiflorum Buch-Ham. ex D. Don | 2800–4300 | Fruits, seeds | Iridoids, Flavonoids | Antioxidant, Anti-inflammatory |
[283] | |
|
Cerastium cerastoides (L.) Britt. | Caryophyllaceae | 2000–4000 | Whole plant | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[86] |
|
Myosoton aquaticum (L.) Moench | 2000–2800 | Leaves, Stem | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[284] | |
|
Silene vulgaris (Moench) Garcke | 2740–3450 | Leaves, Twigs | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[285] | |
|
Stellaria media (L.) Vill. | 2600–3000 | Leaves | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[286] | |
|
Chenopodium album L. | Chenopodiaceae | 350–4300 | Leaves, Seeds | Saponins, Flavonoids | Antioxidant, Anti-inflammatory |
[287] |
|
Chenopodium foliosum Wall. | 2000–4000 | Fruits | Saponins, Flavonoids | Antioxidant, Anti-inflammatory |
[288] | |
|
Convolvulus arvensis L. | Convolvulaceae | 3000–4000 | Flower buds | Alkaloids, Flavonoids | Antioxidant, Neuroprotective |
[289] |
|
Corylus jacquemontii Decne. | Corylaceae | 2000–3300 | Seeds | Catechins, Quercetin | Antioxidant, Anti-inflammatory |
[290] |
|
Rosularia alpestris (Kar. and Kir.) Boriss. | Crassulaceae | 3000–4300 | Whole plant | Phenolic compounds, Flavonoids | Antioxidant, Anti-inflammatory |
[102] |
|
Juniperus communis L. | Cupressaceae | 3000–4200 | Needles | Monoterpenes, Flavonoids |
Antioxidant | [291] |
|
Juniperus indica Bertol. | 3500–4500 | Wood | Monoterpenes, Flavonoids |
Antioxidant | [292] | |
|
Cuscuta reflexa Roxb. | Cuscutaceae | 800–2500 | Whole plant | Flavonoids, Alkaloids | Antioxidant, Hepatoprotective |
[293] |
|
Datisca cannabina L. | Datiscaceae | 2800–3200 | Leaves, Roots | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[294] |
|
Dioscorea deltoidea Wall. ex Kunth | Dioscoreaceae | 2000–2800 | Tuber | Diosgenin, Dioscin | Antioxidant, Anti-inflammatory |
[15] |
|
Elaeagnus conferta Roxb. | Elaeagnaceae | 1500–2200 | Fruits | Triterpenoids, Flavonoids |
Antioxidant, Anti-inflammatory |
[295] |
|
Hippophae rhamnoides L. | 2600–3500 | Fruits, Stem | Flavonoids, Vitamin C | Antioxidant, Immunomodulatory |
[296] | |
|
Hippophae salicifolia D. Don | 2800–3500 | Fruits | Flavonoids, Vitamin C | Antioxidant, Immunomodulatory |
[297] | |
|
Cassiope fastigiata (Wall.) D. Don | Ericaceae | 3800–4600 | Leaves | Polyphenols, Flavonoids |
Antioxidant, Anti-inflammatory |
[298] |
|
Rhododendron anthopogon D. Don | 3200–4500 | Leaves, Flowers | Rhododendrin, Ursolic acid |
Antioxidant, Anti-inflammatory |
[299] | |
|
Rhododendron arboretum Sm. | 2000–4000 | Leaves, Flowers | Arbutin, Quercetin | Antioxidant, Anti-inflammatory |
[300] | |
|
Rhododendron campanulatum D. Don | 3000–4300 | Leaves | Arbutin, Quercetin | Antioxidant, Anti-inflammatory |
[301] | |
|
Gentiana kurroo Royle | Gentianaceae | 1800–4200 | Roots | Gentisin, Swertiamarin |
Antioxidant, Hepatoprotective |
[302] |
|
Gentiana leucomelaena Maxim. ex Kusn. | 2500–5000 | Whole plant | Gentisin, Swertiamarin |
Antioxidant, Hepatoprotective |
[303] | |
|
Gentiana moorcroftiana | 2700–5000 | Leaves | Gentisin, Swertiamarin |
Antioxidant, Hepatoprotective |
[304] | |
|
Gentiana tianshanica Rupr. | 3900–3900 | Whole plant | Gentisin, Swertiamarin |
Antioxidant, Hepatoprotective |
[305] | |
|
Gentiana tubiflora (G. Don) Grirseb. | 4000–5300 | Whole plant | Gentisin, Swertiamarin |
Antioxidant, Hepatoprotective |
[306] | |
|
Gentianopsis detonsa (Rottb.) Ma | 2700–4200 | Whole plant | Gentisin, Swertiamarin |
Antioxidant, Hepatoprotective |
[303] | |
|
Gentianopsis paludosa (Hook.) Ma | 3000–4000 | Whole plant | Gentisin, Swertiamarin |
Antioxidant, Hepatoprotective |
[307] | |
|
Swertia chirayita (Roxb. ex Fleming) Karst. | 1500–3000 | Whole plant | Amarogentin, Swertiamarin |
Antioxidant, Hepatoprotective |
[308] | |
|
Geranium pratense L. | Geraniaceae | 2680–3900 | Whole plant | Geraniin, Tannins | Antioxidant, Anti-inflammatory |
[309] |
|
Geranium wallichianum D. Don ex Sweet | 2600–3980 | Whole plant | Geraniin, Tannins | Antioxidant, Anti-inflammatory |
[310] | |
|
Juglans regia L. | Juglandaceae | 1000–3300 | Leaves, seeds | Juglone, Quercetin | Antioxidant, Anti-inflammatory |
[311] |
|
Lamium album L. | Lamiaceae | 1500–2400 | Roots, Rhizomes |
Rosmarinic acid, Flavonoids |
Antioxidant, Anti-inflammatory |
[312] |
|
Origanum vulgare L | 1800–3600 | Leaves, Stems | Carvacrol, Thymol | Antioxidant | [313] | |
|
Phlomis bracteosa Royle ex Benth. | 3200–4400 | Whole plant | Ursolic acid | Antioxidant, Anti-inflammatory |
[314] | |
|
Salvia nubicola Wall. ex Sweet | 2000–2700 | Roots, Leaves | Salvianolic acid, Rosmarinic acid |
Antioxidant, Anti-inflammatory |
[315] | |
|
Astragalus bicuspis Fischer | Leguminosae | 3100–3500 | Whole plant | Astragaloside IV | Antioxidant, Immunomodulatory |
[316] |
|
Astragalus candolleanus Royle | 3000–4000 | Roots | Astragaloside IV | Antioxidant, Immunomodulatory |
[317] | |
|
Astragalus grahamianus Royle ex Benth. | 3000–3500 | Whole plant | Astragaloside IV | Antioxidant, Immunomodulatory |
[318] | |
|
Astragalus himalayanus Klotzsch | 3200–4400 | Flowers Seeds | Astragaloside IV | Antioxidant, Immunomodulatory |
[319] | |
|
Astragalus strobiliferus Royle | 3000–4000 | Roots | Astragaloside IV | Antioxidant, Immunomodulatory |
[320] | |
|
Astragalus zanskarensis Benth. ex Bunge | 3200–4600 | Roots | Astragaloside IV | Antioxidant, Immunomodulatory |
[321] | |
|
Cicer microphyllum Benth. | 3200–4600 | Aerial parts, | Flavonoids, Saponins | Antioxidant, Anti-inflammatory |
[322] | |
|
Desmodium elegans DC. | 2000–4000 | Leaves | Flavonoids, Alkaloids | Anti-inflammatory | [323] | |
|
Lotus corniculatus L. | 2500–3400 | Whole plant | Rutin, Quercetin | Antioxidant, Anti-inflammatory |
[324] | |
|
Medicago falcata L. | 2700–3500 | Aerial parts | Isoflavones, Saponins | Antioxidant, Anti-inflammatory |
[325] | |
|
Trifolium pratense L. | 2600–3800 | Whole plant | Formononetin, Biochanin A |
Antioxidant | [326] | |
|
Trifolium repens L. | 2600–3200 | Whole plant | Trifoside, Genistein | Antioxidant, Anti-inflammatory |
[327] | |
|
Trigonella emodi Benth. | 2600–3800 | Whole plant | Trigonelline, Diosgenin |
Antioxidant, Antidiabetic, Hypolipidemic | [328] | |
|
Vicia sativa L. | 2600–3000 | Whole plant | Vicine, Convicine | Antioxidant, Antidiabetic | [329] | |
|
Eremurus himalaicus Baker | Liliaceae | 3200–4500 | Fruits | Steroidal saponins | Anti-inflammatory, Immunomodulatory |
[330] |
|
Viscum album L. | Loranthaceae | 2000–3000 | Bark | Viscotoxins, Lectins | Antioxidant, Immunomodulatory |
[331] |
|
Malva neglecta Wallr. | Malvaceae | 2600–4500 | Whole plant | Mucilage | Antioxidant, Anti-inflammatory |
[332] |
|
Malva verticillata L. | 2500–3800 | Seeds | Mucilage | Antioxidant, Anti-inflammatory |
[333] | |
|
Morus serrata Roxb. | Moraceae | 2000–2300 | Leaves, Fruits | Morin, Resveratrol | Antioxidant, Anti-inflammatory |
[334] |
|
Morina coulteriana Royle | Morinaceae | 3000–3700 | Flowers | Morin | Antioxidant, Anti-inflammatory |
[335] |
|
Morina longifolia Wall. ex DC. | 3000–4300 | Roots, Flowers | Morin | Antioxidant, Anti-inflammatory |
[336] | |
|
Jasminum officinale L. | Oleaceae | 1800–4000 | Leaves Stems | Jasmonic acid, Quercetin |
Antioxidant, Anti-inflammatory |
[337] |
|
Epilobium angustifolium L. | Onagraceae | 3000–4700 | Roots | Oenothein B, Quercetin |
Antioxidant, Anti-inflammatory |
[338] |
|
Oenothera glazioviana Micheli | 2000–2700 | Whole plant | Linoleic acid, Gamma-linolenic acid |
Antioxidant, Anti-inflammatory |
[339] | |
|
Dactylorhiza hatagirea D. Don | Orchidaceae | 3000–3800 | Rhizome | Phenanthrenes | Antioxidant, Anti-inflammatory |
[340] |
|
Meconopsis aculeata Royle | Papaveraceae | 2400–4200 | Whole plant | Alkaloids, Flavonoids | Antioxidant, Anti-inflammatory |
[341] |
|
Parnassia nubicola Hook. f. | Parnassiaceae | 1900–3400 | Roots | Parnassiol | Antioxidant, Hepatoprotective, Anti-inflammatory | [342] |
|
Cedrus deodara (Royle ex D. Don) | Pinaceae | 1600–3000 | Wood | Deodarone, Cedrol | Antioxidant | [343] |
|
Pinus gerardiana Wall. ex Lambert. | 2500–3000 | Fruits/Kernels | Pinene, Pinenes | Antioxidant, Anti-inflammatory |
[344] | |
|
Pinus nigra Aiton, Hort. Kew. [W. Aiton] | 1300–2200 | Fruits/Kernels | Pinene, limonene borneol | Antioxidant, Anti-inflammatory |
[345] | |
|
Plantago depressa Willd. | Plantaginaceae | 2000–4500 | Whole plant | Glycosides, Flavonoids |
Antioxidant, Anti-inflammatory |
[346] |
|
Plantago major L. | 2000–2800 | Leaves, Roots, | Aucubin, Ursolic acid | Antioxidant, Anti-inflammatory |
[347] | |
|
Bistorta vaccinifolia (Wall. ex Meisn.) Greene | Polygonaceae | 3000–4600 | Whole plant | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[348] |
|
Koenigia delicatula (Meisn.) H. Hara | 3000–4500 | Stems | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[349] | |
|
Oxyria digyna Hill | 2600–5300 | Whole plant | Oxycoumarins | Antioxidant, Anti-inflammatory |
[350] | |
|
Polygonum alpinum Allioni. | 1500–2400 | Stems, Leaves | Rutin, Quercetin | Antioxidant, Anti-inflammatory |
[351] | |
|
Polygonum aviculare L. | 2000–4200 | Flower buds | Polyphenols, Flavonoids |
Antioxidant, Anti-inflammatory |
[352] | |
|
Polygonum plebejum R.Br. | 1000–4000 | Whole plant | Polyphenols, Flavonoids |
Antioxidant, Anti-inflammatory |
[353] | |
|
Polygonum pubescens Blume | 1500–3700 | Roots | Polyphenols, Flavonoids |
Antioxidant, Anti-inflammatory |
[354] | |
|
Polygonum tortuosum D. Don | 3600–4900 | Young peduncle | Polyphenols, Flavonoids |
Antioxidant, Anti-inflammatory |
[352] | |
|
Rheum australe D. Don | 3300–5200 | Roots | Anthraquinones, Tannins |
Antioxidant | [355] | |
|
Rheum spiciforme Royle | 4000–5000 | Peduncle | Anthraquinones, Tannins |
Antioxidant | [356] | |
|
Rumex acetosa L. | 1500–4000 | Leaves | Anthraquinones, Tannins |
Antioxidant | [357] | |
|
Rumex hestatus D. Don | 1500–3700 | Leaves, Stem | Anthraquinones, Tannins |
Antioxidant | [358] | |
|
Rumex nepalensis Spreng. | 1200–4000 | Roots | Anthraquinones, Tannins |
Antioxidant | [359] | |
|
Aconitum heterophyllum Wall. ex Royle |
Ranunculaceae | 3200–4500 | Roots | Aconitine, Pseudoaconitine |
Antioxidant, Anti-inflammatory |
[360] |
|
Aconitum rotundifolium Kar. and Kir. | 3500–4800 | Stem | Aconitine, Pseudoaconitine |
Antioxidant, Anti-inflammatory |
[361] | |
|
Aconitum violaceum Jacq. ex Stapf | 3200–4400 | Roots | Aconitine, Pseudoaconitine |
Antioxidant, Anti-inflammatory |
[362] | |
|
Aconitum heterophyllum Wall. ex Royle. | 2000–4000 | Roots | Aconitine, atisine, heteratisine, hetisine | Antioxidant, Anti-inflammatory |
[363] | |
|
Anemone rivularis Buch. Ham. ex DC. | 2400–3300 | Leaves, Roots | Saponins, Tannins | Antioxidant, Anti-inflammatory |
[364] | |
|
Aquilegia fragrans Benth. | 2900–3500 | Whole plant | Alkaloids, Flavonoids | Antioxidant, Anti-inflammatory |
[365] | |
|
Aquilegia moorcroftiana Wall. ex Royle | 3300–3700 | Twigs | Alkaloids, Flavonoids | Antioxidant, Anti-inflammatory |
[366] | |
|
Caltha palustris L. | 3020–3500 | Leaves, Roots | Protoanemonin | Antioxidant, Anti-inflammatory |
[367] | |
|
Clematis grata Wall. | 2000–2600 | Leaves | Clematichinenoside | Antioxidant, Anti-inflammatory |
[368] | |
|
Clematis ladakhiana C. Grey-Wilson | 3200–3900 | Roots Shoots | Clematichinenoside | Antioxidant, Anti-inflammatory |
[369] | |
|
Clematis orientalis L. | 3400–5200 | Whole plant | Clematichinenoside | Antioxidant, Anti-inflammatory |
[370] | |
|
Crataegus songarica K. Koch | 1500–2000 | Fruits, Leaves | Flavonoids, Triterpenes |
Antioxidant, Cardioprotective |
[371] | |
|
Fragaria nubicola Lindl. | 2500–3900 | Fruit, Roots | Anthocyanins, Ellagic acid |
Antioxidant | [372] | |
|
Geum elatum Wall. ex G. Don | 3500–4500 | Roots | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[373] | |
|
Potentilla atrisanguinea Lodd. var. argyrophylla (Wall. ex Lehm.) Griers. and Long | 3000–4500 | Roots | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[374] | |
|
Potentilla eriocarpa Wall. ex Lehm. | 3000–5000 | Whole plant | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[375] | |
|
Potentilla fulgens Wall. | 2000–3200 | Roots | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[376] | |
|
Potentilla nubicola Lindl. ex Lacaita | 2900–4000 | Fruits | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[377] | |
|
Prinsepia utilis Royle | 1800–3000 | Seeds, Roots | Triterpenes, Flavonoids |
Antioxidant, Hepatoprotectiv | [378] | |
|
Pyracantha crenulata (D. Don) Roemer | 1000–2600 | Fruits | Flavonoids, Triterpenes |
Antioxidant, Anti-inflammatory |
[379] | |
|
Pyrus lanata D. Don. | 2700–3400 | Fruits | Triterpenes, Flavonoids |
Antioxidant, Hepatoprotective | [380] | |
|
Rosa brunonii Lindl. | 2100–4500 | Flowers | Anthocyanins, Flavonoids |
Antioxidant, Anti-inflammatory |
[381] | |
|
Rosa webbiana Wall. ex Royle | 3000–3800 | Fruits, Stem, Flowers | Anthocyanins, Flavonoids |
Antioxidant, Anti-inflammatory |
[382] | |
|
Rubus ellipticus Sm. | 1800–2600 | Fruits | Anthocyanins, Ellagic acid |
Antioxidant | [383] | |
|
Rubus niveus Thunb. | 2000–2800 | Fruits | Anthocyanins, Ellagic acid |
Antioxidant | [384] | |
|
Spiraea canescens D. Don | 2600–4000 | Stem | Tannins, Flavonoids | Antioxidant, Anti-inflammatory |
[385] | |
|
Rubia cordifolia L. | Rubiaceae | 1800–3000 | Leaves, Stem, Roots | Anthraquinones, Tannins |
Antioxidant, Anti-inflammatory |
[386] |
|
Euphrasia flabellate Pennell | Scrophulariaceae | 3000–4000 | Whole plant | Iridoid glycosides, Flavonoids | Antioxidant, Anti-inflammatory |
[387] |
|
Euphrasia paucifolia Wettst. | 3000–4300 | Leaves | Iridoid glycosides, Flavonoids | Antioxidant, Anti-inflammatory |
[388] | |
|
Picrorhiza kurroa Royle ex Benth. | 3000–4000 | Roots | Picroside I, Picroside II |
Antioxidant, Hepatoprotective |
[389] | |
|
Scrophularia calycina Benth. | 3000–4000 | Whole plant | Iridoid glycosides, Flavonoids | Antioxidant, Anti-inflammatory |
[390] | |
|
Scrophularia decomposita Royle ex Benth. | 3000–4200 | Leaves | Iridoid glycosides, Flavonoids | Antioxidant, Anti-inflammatory |
[391] | |
|
Urtica dioica Jacq. ex Wedd. | Urticaceae | 2000–3000 | Leaves | Acetylcholine, Histamine |
Anti-inflammatory | [392] |
5. Challenges of Using High-Altitude Phytochemicals in Medicine
5.1. Challenges in Extraction and Utilization
Extracting and utilizing compounds from high-altitude medicinal plants hold immense promise for various practical applications, yet it also presents formidable challenges and limitations that demand thoughtful consideration and innovative solutions [393]. These plants, thriving in extreme environments that are characterized by harsh climatic conditions and often rare or endemic species, offer unique bioactive compounds that could revolutionize pharmaceutical, nutraceutical, and cosmetic industries. However, factors such as environmental harshness, slow growth rates, seasonal variability in compound production, and the complexity of chemical profiles pose significant hurdles. The limiting factors when utilizing high-altitude-plant-derived compounds in modern health care are discussed as follows [394].
5.1.1. Harsh Environmental Conditions
High-altitude environments are characterized by extreme temperatures, strong UV radiation, and variable weather conditions. Accessing and cultivating these plants can be challenging due to these harsh conditions, which can affect plant growth, availability, and quality of the compounds extracted [395].
5.1.2. Low Biomass and Slow Growth
Many high-altitude plants have slow growth rates and produce low biomass. This makes it difficult to obtain sufficient quantities of the desired compounds for large-scale extraction and commercial applications [396].
5.1.3. Species Rarity and Endemism
Some high-altitude plants are rare, endemic to specific regions, or protected due to conservation concerns. Harvesting these plants for medicinal compounds may raise ethical and sustainability issues if not managed carefully [397].
5.1.4. Seasonal Variability
The growth and production of bioactive compounds in high-altitude plants can be highly seasonal and dependent on weather patterns. This variability makes it challenging to maintain a consistent quality and quantity of extracts throughout the year [398].
5.1.5. Complex Chemistry
High-altitude plants often contain complex chemical profiles with multiple bioactive compounds. Identifying, isolating, and characterizing the specific compounds responsible for medicinal properties can be difficult and require sophisticated analytical techniques [395].
5.1.6. Extraction Efficiency
Extracting bioactive compounds from high-altitude plants can be inefficient due to factors such as low compound concentrations, the presence of interfering substances, and extraction methods that may not be optimized for these specific plant materials [395].
5.1.7. Cultural and Traditional Knowledge
Utilizing medicinal plants from high-altitude regions often requires understanding traditional knowledge and practices. Integrating modern extraction techniques with traditional wisdom can be challenging but is essential for sustainable use [399].
Addressing these challenges requires interdisciplinary approaches that blend botanical expertise, ecological understanding, advanced extraction technologies, and respect for local traditions and environmental sustainability. Research and development efforts aimed at overcoming these limitations are crucial for harnessing the potential of high-altitude medicinal plants for practical applications in healthcare and beyond.
5.2. Regulatory Challenges
Regulatory challenges encompass navigating diverse international regulations governing the use of natural products in healthcare, which vary widely in terms of safety standards, labelling requirements, and permissible claims. Compliance with these regulations is crucial to establish the credibility and legality of medicinal products derived from high-altitude plants. Moreover, rigorous quality control measures are indispensable for guaranteeing the batch-to-batch consistency, purity, and potency of phytochemical antioxidants. Variations in environmental factors, such as altitude, climate, and soil composition, can significantly influence the composition and concentration of bioactive compounds in these plants, underscoring the need for robust analytical methods, standardized protocols and regulatory bodies, stringent quality control, rigorous clinical trials, protected intellectual property, and traditional knowledge that is integrated with scientific advancements [400].
To establish robust safety assessment plans and regulatory frameworks for manufacturing herbal medicines, various global regulatory bodies have issued guidance documents. These include the International Life Sciences Institute, Washington, DC, USA; the International Union of Pure and Applied Chemistry, North Carolina, USA; the European Medicines Agency, Amsterdam, The Netherlands and the European Food Safety Authority in Parma, Italy [401]. In the USA, the sale and purchase of herbal medicines falls under the regulation of the Dietary Supplement Health and Education Act of 1994 [402]. In the European Union, national regulatory bodies like the Committee on Herbal Medicinal Products (HMPC), part of the European Medicines Agency, oversee the production and marketing of herbal drugs. Similarly, Canada operates under the Natural Health Products Regulations (NHPR), and Australia relies on the Therapeutic Goods Administration (TGA) to regulate the manufacturing and marketing of herbal drugs [403,404]. In India, the Ministry of AYUSH serves as the regulatory authority responsible for issuing licenses for the manufacturing and marketing of herbal drugs [405].
By addressing these regulatory and quality control challenges proactively, researchers and practitioners can ensure that phytochemical antioxidants from high-altitude plants meet the stringent safety and efficacy standards required for medical use. This approach not only enhances the credibility of natural products but also fosters trust among healthcare providers and patients, facilitating their broader adoption and contribution to improving health outcomes. Therefore, future studies should prioritize these critical considerations in order to accelerate the translation of research findings into impactful medical applications.
6. Future Prospects
6.1. Dietary Phytochemicals as Antioxidants
Dietary phytochemicals, encompassing a diverse array of compounds, like flavonoids, polyphenols, and carotenoids, play a pivotal role as antioxidants in human health [406]. These compounds are specifically derived from plant-based foods consumed as part of a diet, distinguishing them from phytochemicals in general, which include those found in plants but may not necessarily be consumed through dietary sources. By scavenging free radicals, chelating metal ions, and activating endogenous antioxidant enzymes, dietary phytochemicals mitigate oxidative stress and prevent the cellular damage associated with chronic diseases such as cancer [407], cardiovascular diseases [2], neurodegenerative disorders [2], gastrointestinal disorders [2], metabolic disorders and ageing [408]. The following examples highlight the antioxidant prowess of dietary phytochemicals like curcumin, resveratrol, quercetin, and epigallocatechin gallate. Curcumin, abundant in turmeric, not only scavenges free radicals but also exhibits potent anti-inflammatory effects, making it a promising candidate for conditions like Alzheimer’s disease and cancer [409]. Resveratrol, found in red grapes and wine, not only scavenges free radicals but also improves endothelial function and reduces oxidative damage in cardiovascular tissues, potentially reducing the risk of heart disease [410]. Quercetin, present in various fruits and vegetables, not only scavenges free radicals but also modulates signalling pathways involved in oxidative stress and inflammation, offering protection against chronic inflammatory diseases [180]. EGCG, abundant in green tea, not only scavenges free radicals but also induces antioxidant enzymes and inhibits oxidative damage to DNA and proteins, contributing to cancer prevention and overall longevity [411]. These compounds exhibit multifaceted antioxidant mechanisms, thereby contributing to overall health and longevity.
Incorporating a variety of dietary phytochemical-rich plant foods into the diet offers a promising strategy for disease prevention and promoting optimal wellbeing. Ongoing research into novel dietary phytochemicals continues to expand our understanding of their antioxidant properties, fuelling the development of dietary interventions for combating oxidative stress-related disorders. Biotechnological advances hold the promise of enhancing production and bioavailability, while personalized nutrition approaches will tailor antioxidant interventions to individual needs. Overall, the future of plant-based antioxidants is promising, offering innovative solutions for improving health and wellbeing on a global scale.
6.2. Novel Delivery Systems for Sustained Release
Innovative delivery systems for the sustained release of phytochemical antioxidants are poised to revolutionize the field of nutraceuticals and pharmaceuticals. These systems, ranging from nanoparticles to encapsulation techniques, offer enhanced stability, bioavailability, and targeted delivery of antioxidant compounds [412]. Nanoparticle-based carriers, such as liposomes, polymeric nanoparticles, and solid lipid nanoparticles, enable the controlled release and protection of phytochemicals during digestion, ensuring optimal absorption and efficacy. Encapsulation methods, including microencapsulation and nano-emulsions, provide a protective matrix for phytochemicals, preventing degradation and enabling sustained release in the gastrointestinal tract. These delivery systems not only improve the therapeutic potential of phytochemical antioxidants but also offer opportunities for functional food fortification and personalized supplementation strategies [413]. As research in this area continues to advance, novel delivery systems hold promise for addressing complex health challenges and optimizing the preventive and therapeutic effects of phytochemical antioxidants.
One prominent innovative delivery system used for phytochemical antioxidants is nanoemulsions. Nanoemulsions are colloidal dispersions of oil and water stabilized by surfactants or emulsifiers, with droplet sizes typically in the nanometre range. These delivery systems offer several advantages for phytochemical encapsulation and controlled release, including enhanced solubility, stability, and bioavailability. By encapsulating phytochemicals within nanoemulsion droplets, their exposure to harsh environmental conditions, such as pH changes and enzymatic degradation in the gastrointestinal tract, can be minimized, leading to improved absorption and efficacy [414]. Nanoemulsions have been successfully employed to deliver various antioxidant compounds from plant sources, such as polyphenols and carotenoids, in functional foods, dietary supplements, and pharmaceutical formulations [415]. Their versatility and effectiveness make nanoemulsions a promising strategy for harnessing the potential of phytochemical antioxidants in combating oxidative stress-related diseases.
Nanoparticles represent another prominent and versatile delivery system for phytochemical antioxidants. Nanoparticles, including liposomes, polymeric nanoparticles, and solid lipid nanoparticles, offer numerous advantages, such as controlled release, enhanced stability, and the targeted delivery of antioxidants [416]. Liposomes encapsulate hydrophilic and hydrophobic phytochemicals within their aqueous core or lipid bilayer. Polymeric nanoparticles, on the other hand, provide a customizable platform for encapsulating phytochemicals through techniques like nanoprecipitation or emulsion solvent evaporation. Additionally, solid lipid nanoparticles offer improved stability and sustained release of antioxidants due to their lipid matrix [417]. These nanoparticle-based delivery systems enable the efficient protection of phytochemicals from degradation, prolonged circulation time, and enhanced cellular uptake, making them promising candidates for pharmaceutical and nutraceutical applications in combating oxidative stress and related diseases.
Innovative delivery systems represent a promising avenue for enhancing the efficacy and applicability of phytochemical antioxidants. Microencapsulation, for instance, offers a means by which to encapsulate antioxidants within protective matrices, safeguarding them from degradation while enabling controlled release. This approach ensures sustained antioxidant activity, addressing issues of stability and bioavailability [418]. Additionally, smart delivery systems, incorporating molecular sensors and precision targeting mechanisms, hold potential for the precise and efficient delivery of antioxidants to sites of oxidative stress. Nanoparticle-based carriers, such as liposomes and polymeric nanoparticles, provide further opportunities for enhanced delivery and cellular uptake of antioxidants. These systems offer controlled release kinetics and the ability to tailor delivery to specific tissues or cells, optimizing therapeutic outcomes. Furthermore, the development of nanogel-based delivery systems presents a promising approach for topical antioxidant delivery, offering improved skin penetration and localized antioxidant activity [419]. As research in this field progresses, these innovative delivery systems hold significant promise for advancing the therapeutic utility of phytochemical antioxidants in combating oxidative stress-related diseases.
7. Methodology
In this study, two databases, namely PubMed and DOAJ, were searched particularly by using the following specific keywords: “Oxidative stress and health disorders”, “High altitude medicinal plants” and “plant antioxidants.” Collectively, a total number of 30,924 articles, both reviews as well as original research articles, published from the year 1930 to 2023, were identified. The selection was meticulously undertaken as per the focus of the present study and only those articles published in peer-reviewed journals were included in the study to ensure the quality of the work.
Moreover, the chemical structures and formulae of phytocompounds were sourced from PubChem by using their common as well as IUPAC names (wherever necessary). The botanical names of the plants mentioned in the study have been cross verified with the International Plant Names Index (IPNI).
8. Conclusions
As a prominent contributing factor, oxidative stress poses serious health hazards, including cardiovascular diseases; cancers; neurological disorders, such as Parkinson’s and Alzheimer’s diseases; and metabolic disorders, which ultimately lead to mortality. This phenomenon arises from the generation of reactive oxygen and nitrogen species within the human body, causing damage to crucial biomolecules such as DNA, proteins, and lipids. Moreover, oxidative stress disrupts gene expression levels, resulting in chronic health conditions. The rise in oxidative stress levels can be attributed to environmental and lifestyle changes prevalent today. Factors like high pollution levels in the environment, along with unhealthy habits such as alcohol consumption, cigarette smoking, and poor dietary patterns, contribute significantly to oxidative stress and its associated health disorders.
Given the detrimental effects of oxidative stress, its prevention and inhibition have become increasingly crucial. Antioxidants play a key role in combating oxidative stress and mitigating its detrimental effects. They can be produced naturally within the body or obtained through dietary or other supplements. Natural sources of antioxidants, particularly those derived from plants and their bioactive compounds, have emerged as a beneficial approach due to their immense versatility, bioavailability and minimal side effects. Phytochemicals, including polyphenols, flavonoids, ascorbic acid, tocopherols, and tocotrienols, possess inherent abilities to neutralize toxic oxygen species such as hydrogen peroxide and singlet oxygen radicals, thus counteracting oxidative stress.
Plants produce a plethora of secondary metabolites, with a range of biological effects, including antioxidants. Further, plants thriving in high-altitude regions experience numerous environmental stresses that stimulate the production of these phytochemicals in substantial quantities. However, despite advances in research, much of the available biodiversity among the plants and their phytocompounds is still unexplored. This necessitates further exploration of the antioxidant properties of high-altitude medicinal plants to unlock their full therapeutic potential. By investing in interdisciplinary research initiatives, the wealth of natural antioxidants present in these plants could be harnessed with the development of innovative approaches in preventive healthcare and drug discovery. Collaboration between scientists, indigenous communities, and policymakers is essential to promote the sustainable utilization of plant resources and improve global health outcomes. It is imperative that we prioritize the exploration of high-altitude medicinal plants in order to address the growing burden of oxidative stress-related diseases and contribute to the preservation of biodiversity and traditional knowledge for the improvement of human health.
Acknowledgments
The authors extend their appreciation to the Dean, Academic Affairs, Baba Ghulam Shah Badshah University for Academic and Institutional support. The authors extend their gratitude to their fellow colleagues, who helped in improving the contents of this manuscript.
Author Contributions
Conceptualization, M.V.A., S.A. and S.M; writing—original draft preparation, M.V.A., S.K., S.M., M.A. and S.A.; writing—review and editing, M.V.A., S.K., M.A.H.K., A.A.S., M.A., K.S.G., B.R. and S.A.; visualization, S.R. and M.A.H.K.; supervision, A.A.S., M.A. and S.A.; project administration, S.A.; funding acquisition, S.A. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This work was supported by University Grants Commission, New Delhi, India, grant number F 30-459/2019(BSR) to S.A.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Liguori I., Russo G., Curcio F., Bulli G., Aran L., Della-Morte D., Gargiulo G., Testa G., Cacciatore F., Bonaduce D., et al. Oxidative Stress, Aging, and Diseases. Clin. Interv. Aging. 2018;13:757–772. doi: 10.2147/CIA.S158513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hajam Y.A., Rani R., Ganie S.Y., Sheikh T.A., Javaid D., Qadri S.S., Pramodh S., Alsulimani A., Alkhanani M.F., Harakeh S., et al. Oxidative Stress in Human Pathology and Aging: Molecular Mechanisms and Perspectives. Cells. 2022;11:552. doi: 10.3390/cells11030552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Trachootham D., Lu W., Ogasawara M.A., Valle N.R.-D., Huang P. Redox Regulation of Cell Survival. Antioxid. Redox Signal. 2008;10:1343–1374. doi: 10.1089/ars.2007.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Birben E., Sahiner U.M., Sackesen C., Erzurum S., Kalayci O. Oxidative Stress and Antioxidant Defense. World Allergy Organ. J. 2012;5:9–19. doi: 10.1097/WOX.0b013e3182439613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.de Almeida A.J.P.O., de Oliveira J.C.P.L., da Silva Pontes L.V., de Souza Júnior J.F., Gonçalves T.A.F., Dantas S.H., de Almeida Feitosa M.S., Silva A.O., de Medeiros I.A. ROS: Basic Concepts, Sources, Cellular Signaling, and Its Implications in Aging Pathways. Oxid. Med. Cell. Longev. 2022;2022:1225578. doi: 10.1155/2022/1225578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fujii J., Homma T., Osaki T. Superoxide Radicals in the Execution of Cell Death. Antioxidants. 2022;11:501. doi: 10.3390/antiox11030501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Forcados G.E., Muhammad A., Oladipo O.O., Makama S., Meseko C.A. Metabolic Implications of Oxidative Stress and Inflammatory Process in SARS-CoV-2 Pathogenesis: Therapeutic Potential of Natural Antioxidants. Front. Cell. Infect. Microbiol. 2021;11:654813. doi: 10.3389/fcimb.2021.654813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Masenga S.K., Kabwe L.S., Chakulya M., Kirabo A. Mechanisms of Oxidative Stress in Metabolic Syndrome. Int. J. Mol. Sci. 2023;24:7898. doi: 10.3390/ijms24097898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mudau F.N., Chimonyo V.G.P., Modi A.T., Mabhaudhi T. Neglected and Underutilised Crops: A Systematic Review of Their Potential as Food and Herbal Medicinal Crops in South Africa. Front. Pharmacol. 2022;12:809866. doi: 10.3389/fphar.2021.809866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Juan C.A., Pérez de la Lastra J.M., Plou F.J., Pérez-Lebeña E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021;22:4642. doi: 10.3390/ijms22094642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Panday A., Sahoo M.K., Osorio D., Batra S. NADPH Oxidases: An Overview from Structure to Innate Immunity-Associated Pathologies. Cell. Mol. Immunol. 2015;12:5–23. doi: 10.1038/cmi.2014.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eguchi N., Vaziri N.D., Dafoe D.C., Ichii H. The Role of Oxidative Stress in Pancreatic β Cell Dysfunction in Diabetes. Int. J. Mol. Sci. 2021;22:1509. doi: 10.3390/ijms22041509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Muscolo A., Mariateresa O., Giulio T., Mariateresa R. Oxidative Stress: The Role of Antioxidant Phytochemicals in the Prevention and Treatment of Diseases. Int. J. Mol. Sci. 2024;25:3264. doi: 10.3390/ijms25063264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhu H., Liu C., Qian H. Pharmaceutical Potential of High-Altitude Plants for Fatigue-Related Disorders: A Review. Plants. 2022;11:2004. doi: 10.3390/plants11152004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Semwal P., Painuli S., Cruz-Martins N. Dioscorea Deltoidea Wall. Ex Griseb: A Review of Traditional Uses, Bioactive Compounds and Biological Activities. Food Biosci. 2021;41:100969. doi: 10.1016/j.fbio.2021.100969. [DOI] [Google Scholar]
- 16.Llauradó Maury G., Méndez Rodríguez D., Hendrix S., Escalona Arranz J.C., Fung Boix Y., Pacheco A.O., García Díaz J., Morris-Quevedo H.J., Ferrer Dubois A., Aleman E.I., et al. Antioxidants in Plants: A Valorization Potential Emphasizing the Need for the Conservation of Plant Biodiversity in Cuba. Antioxidants. 2020;9:1048. doi: 10.3390/antiox9111048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pizzino G., Irrera N., Cucinotta M., Pallio G., Mannino F., Arcoraci V., Squadrito F., Altavilla D., Bitto A. Oxidative Stress: Harms and Benefits for Human Health. Oxid. Med. Cell. Longev. 2017;2017:8416763. doi: 10.1155/2017/8416763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bhattacharyya A., Chattopadhyay R., Mitra S., Crowe S.E. Oxidative Stress: An Essential Factor in the Pathogenesis of Gastrointestinal Mucosal Diseases. Physiol. Rev. 2014;94:329–354. doi: 10.1152/physrev.00040.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Phaniendra A., Jestadi D.B., Periyasamy L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J. Clin. Biochem. 2015;30:11–26. doi: 10.1007/s12291-014-0446-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Checa J., Aran J.M. Reactive Oxygen Species: Drivers of Physiological and Pathological Processes. J. Inflamm. Res. 2020;13:1057–1073. doi: 10.2147/JIR.S275595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bhatti J.S., Bhatti G.K., Reddy P.H. Mitochondrial Dysfunction and Oxidative Stress in Metabolic Disorders—A Step towards Mitochondria Based Therapeutic Strategies. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2017;1863:1066–1077. doi: 10.1016/j.bbadis.2016.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vona R., Pallotta L., Cappelletti M., Severi C., Matarrese P. The Impact of Oxidative Stress in Human Pathology: Focus on Gastrointestinal Disorders. Antioxidants. 2021;10:201. doi: 10.3390/antiox10020201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nagarkatti P., Pandey R., Rieder S.A., Hegde V.L., Nagarkatti M. Cannabinoids as Novel Anti-Inflammatory Drugs. Future Med. Chem. 2009;1:1333–1349. doi: 10.4155/fmc.09.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sharma P., Jha A.B., Dubey R.S., Pessarakli M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J. Bot. 2012;2012:217037. doi: 10.1155/2012/217037. [DOI] [Google Scholar]
- 25.Wang Y., Branicky R., Noë A., Hekimi S. Superoxide Dismutases: Dual Roles in Controlling ROS Damage and Regulating ROS Signaling. J. Cell Biol. 2018;217:1915–1928. doi: 10.1083/jcb.201708007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Carraro E., Schilirò T., Biorci F., Romanazzi V., Degan R., Buonocore D., Verri M., Dossena M., Bonetta S., Gilli G. Physical Activity, Lifestyle Factors and Oxidative Stress in Middle Age Healthy Subjects. Int. J. Environ. Res. Public Health. 2018;15:1152. doi: 10.3390/ijerph15061152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Halliwell B. Oxidative Stress and Neurodegeneration: Where Are We Now? J. Neurochem. 2006;97:1634–1658. doi: 10.1111/j.1471-4159.2006.03907.x. [DOI] [PubMed] [Google Scholar]
- 28.Rani V., Deep G., Singh R.K., Palle K., Yadav U.C.S. Oxidative Stress and Metabolic Disorders: Pathogenesis and Therapeutic Strategies. Life Sci. 2016;148:183–193. doi: 10.1016/j.lfs.2016.02.002. [DOI] [PubMed] [Google Scholar]
- 29.Dubois-deruy E., Peugnet V., Turkieh A., Pinet F. Oxidative Stress in Cardiovascular Diseases. Antioxidants. 2020;9:864. doi: 10.3390/antiox9090864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Griendling K.K., Camargo L.L., Rios F.J., Alves-Lopes R., Montezano A.C., Touyz R.M. Oxidative Stress and Hypertension. Circ. Res. 2021;128:993–1020. doi: 10.1161/CIRCRESAHA.121.318063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Murphy E., Liu J.C. Mitochondrial Calcium and Reactive Oxygen Species in Cardiovascular Disease. Cardiovasc. Res. 2023;119:1105–1116. doi: 10.1093/cvr/cvac134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.DeTure M.A., Dickson D.W. The Neuropathological Diagnosis of Alzheimer’s Disease. Mol. Neurodegener. 2019;14:32. doi: 10.1186/s13024-019-0333-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wei Z., Li X., Li X., Liu Q., Cheng Y. Oxidative Stress in Parkinson’s Disease: A Systematic Review and Meta-Analysis. Front. Mol. Neurosci. 2018;11:236. doi: 10.3389/fnmol.2018.00236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Davalli P., Marverti G., Lauriola A., D’Arca D. Targeting Oxidatively Induced DNA Damage Response in Cancer: Opportunities for Novel Cancer Therapies. Oxid. Med. Cell. Longev. 2018;2018:2389523. doi: 10.1155/2018/2389523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Klaunig J.E. Oxidative Stress and Cancer. Curr. Pharm. Des. 2019;24:4771–4778. doi: 10.2174/1381612825666190215121712. [DOI] [PubMed] [Google Scholar]
- 36.Tangvarasittichai S. Oxidative Stress, Insulin Resistance, Dyslipidemia and Type 2 Diabetes Mellitus. World J. Diabetes. 2015;6:456. doi: 10.4239/wjd.v6.i3.456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Manna P., Jain S.K. Obesity, Oxidative Stress, Adipose Tissue Dysfunction, and the Associated Health Risks: Causes and Therapeutic Strategies. Metab. Syndr. Relat. Disord. 2015;13:423–444. doi: 10.1089/met.2015.0095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tan B.L., Norhaizan M.E., Liew W.-P.-P., Sulaiman Rahman H. Antioxidant and Oxidative Stress: A Mutual Interplay in Age-Related Diseases. Front. Pharmacol. 2018;9:01162. doi: 10.3389/fphar.2018.01162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dias T.R., Martin-Hidalgo D., Silva B.M., Oliveira P.F., Alves M.G. Endogenous and Exogenous Antioxidants As a Tool to Ameliorate Male Infertility Induced by Reactive Oxygen Species. Antioxid. Redox Signal. 2020;33:767–785. doi: 10.1089/ars.2019.7977. [DOI] [PubMed] [Google Scholar]
- 40.Moussa Z., Judeh Z.M.A., Ahmed S.A. Free Radical Medicine and Biology. IntechOpen; London, UK: 2020. Nonenzymatic Exogenous and Endogenous Antioxidants. [DOI] [Google Scholar]
- 41.Krishnamurthy P., Wadhwani A. Antioxidant Enzyme. IntechOpen; London, UK: 2012. Antioxidant Enzymes and Human Health. [DOI] [Google Scholar]
- 42.Irato P., Santovito G. Enzymatic and Non-Enzymatic Molecules with Antioxidant Function. Antioxidants. 2021;10:579. doi: 10.3390/antiox10040579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Couto N., Wood J., Barber J. The Role of Glutathione Reductase and Related Enzymes on Cellular Redox Homoeostasis Network. Free Radic. Biol. Med. 2016;95:27–42. doi: 10.1016/j.freeradbiomed.2016.02.028. [DOI] [PubMed] [Google Scholar]
- 44.Cox G.M., Harrison T.S., McDade H.C., Taborda C.P., Heinrich G., Casadevall A., Perfect J.R. Superoxide Dismutase Influences the Virulence of Cryptococcus neoformans by Affecting Growth within Macrophages. Infect. Immun. 2003;71:173–180. doi: 10.1128/IAI.71.1.173-180.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Niki E. Oxidative Stress and Antioxidants: Distress or Eustress? Arch. Biochem. Biophys. 2016;595:19–24. doi: 10.1016/j.abb.2015.11.017. [DOI] [PubMed] [Google Scholar]
- 46.Bouayed J., Bohn T. Exogenous Antioxidants—Double-Edged Swords in Cellular Redox State: Health Beneficial Effects at Physiologic Doses versus Deleterious Effects at High Doses. Oxid. Med. Cell. Longev. 2010;3:228–237. doi: 10.4161/oxim.3.4.12858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kasote D.M., Katyare S.S., Hegde M.V., Bae H. Significance of Antioxidant Potential of Plants and Its Relevance to Therapeutic Applications. Int. J. Biol. Sci. 2015;11:982–991. doi: 10.7150/ijbs.12096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sofowora A., Ogunbodede E., Onayade A. The Role and Place of Medicinal Plants in the Strategies for Disease Prevention. Afr. J. Tradit. Complement. Altern. Med. 2013;10:210–229. doi: 10.4314/ajtcam.v10i5.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kumar A., Guleria S., Ghosh D., Dogra V., Kumar S. Managing Reactive Oxygen Species—Some Learnings from High Altitude Extremophytes. Environ. Exp. Bot. 2021;189:104525. doi: 10.1016/j.envexpbot.2021.104525. [DOI] [Google Scholar]
- 50.Das K., Roychoudhury A. Reactive Oxygen Species (ROS) and Response of Antioxidants as ROS-Scavengers during Environmental Stress in Plants. Front. Environ. Sci. 2014;2:53. doi: 10.3389/fenvs.2014.00053. [DOI] [Google Scholar]
- 51.Lee M.T., Lin W.C., Yu B., Lee T.T. Antioxidant Capacity of Phytochemicals and Their Potential Effects on Oxidative Status in Animals—A Review. Asian-Australas. J. Anim. Sci. 2016;30:299–308. doi: 10.5713/ajas.16.0438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ponnampalam E.N., Kiani A., Santhiravel S., Holman B.W.B., Lauridsen C., Dunshea F.R. The Importance of Dietary Antioxidants on Oxidative Stress, Meat and Milk Production, and Their Preservative Aspects in Farm Animals: Antioxidant Action, Animal Health, and Product Quality—Invited Review. Animals. 2022;12:3279. doi: 10.3390/ani12233279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kumar A., P N., Kumar M., Jose A., Tomer V., Oz E., Proestos C., Zeng M., Elobeid T., K S., et al. Major Phytochemicals: Recent Advances in Health Benefits and Extraction Method. Molecules. 2023;28:887. doi: 10.3390/molecules28020887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.González-Peña M.A., Ortega-Regules A.E., Anaya de Parrodi C., Lozada-Ramírez J.D. Chemistry, Occurrence, Properties, Applications, and Encapsulation of Carotenoids—A Review. Plants. 2023;12:313. doi: 10.3390/plants12020313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Carazo A., Macáková K., Matoušová K., Krčmová L.K., Protti M., Mladěnka P. Vitamin A Update: Forms, Sources, Kinetics, Detection, Function, Deficiency, Therapeutic Use and Toxicity. Nutrients. 2021;13:1703. doi: 10.3390/nu13051703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yabuzaki J. Carotenoids Database: Structures, chemical fingerprints and distribution among organisms. Database J. Biol. Databases Curation. 2017;2017:bax004. doi: 10.1093/database/bax004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Merhan O. Carotenoids. IntechOpen; London, UK: 2017. The Biochemistry and Antioxidant Properties of Carotenoids. [DOI] [Google Scholar]
- 58.Sindhu E.R., Kavya A.K., Binitha P.P. Handbook of Oxidative Stress in Cancer: Therapeutic Aspects. Springer; Singapore: 2022. Role of Carotenoids in Preventing Oxidative Stress—Induced Cancer; pp. 351–363. [DOI] [Google Scholar]
- 59.Khoo H.-E., Prasad K.N., Kong K.-W., Jiang Y., Ismail A. Carotenoids and Their Isomers: Color Pigments in Fruits and Vegetables. Molecules. 2011;16:1710–1738. doi: 10.3390/molecules16021710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tapiero H., Townsend D.M., Tew K.D. The Role of Carotenoids in the Prevention of Human Pathologies. Biomed. Pharmacother. 2004;58:100–110. doi: 10.1016/j.biopha.2003.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ramel F., Birtic S., Cuiné S., Triantaphylidès C., Ravanat J.-L., Havaux M. Chemical Quenching of Singlet Oxygen by Carotenoids in Plants. Plant Physiol. 2012;158:1267–1278. doi: 10.1104/pp.111.182394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Park H.-A., Hayden M.M., Bannerman S., Jansen J., Crowe-White K.M. Anti-Apoptotic Effects of Carotenoids in Neurodegeneration. Molecules. 2020;25:3453. doi: 10.3390/molecules25153453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tan B.L., Norhaizan M.E. Carotenoids: How Effective Are They to Prevent Age-Related Diseases? Molecules. 2019;24:1801. doi: 10.3390/molecules24091801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Maria A.G., Graziano R., Nicolantonio D. Carotenoids: Potential Allies of Cardiovascular Health? Food Nutr. Res. 2015;59:26762. doi: 10.3402/fnr.v59.26762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Mrowicka M., Mrowicki J., Kucharska E., Majsterek I. Lutein and Zeaxanthin and Their Roles in Age-Related Macular Degeneration—Neurodegenerative Disease. Nutrients. 2022;14:827. doi: 10.3390/nu14040827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Akram N.A., Shafiq F., Ashraf M. Ascorbic Acid-A Potential Oxidant Scavenger and Its Role in Plant Development and Abiotic Stress Tolerance. Front. Plant Sci. 2017;8:613. doi: 10.3389/fpls.2017.00613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Flieger J., Flieger W., Baj J., Maciejewski R. Antioxidants: Classification, Natural Sources, Activity/Capacity Measurements, and Usefulness for the Synthesis of Nanoparticles. Materials. 2021;14:4135. doi: 10.3390/ma14154135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gallie D.R. L-Ascorbic Acid: A Multifunctional Molecule Supporting Plant Growth and Development. Scientifica. 2013;2013:795964. doi: 10.1155/2013/795964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hasanuzzaman M., Nahar K., Anee T.I., Fujita M. Glutathione in Plants: Biosynthesis and Physiological Role in Environmental Stress Tolerance. Physiol. Mol. Biol. Plants. 2017;23:249–268. doi: 10.1007/s12298-017-0422-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Carr A., Maggini S. Vitamin C and Immune Function. Nutrients. 2017;9:1211. doi: 10.3390/nu9111211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gęgotek A., Skrzydlewska E. Antioxidative and Anti-Inflammatory Activity of Ascorbic Acid. Antioxidants. 2022;11:1993. doi: 10.3390/antiox11101993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Morelli M.B., Gambardella J., Castellanos V., Trimarco V., Santulli G. Vitamin C and Cardiovascular Disease: An Update. Antioxidants. 2020;9:1227. doi: 10.3390/antiox9121227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chambial S., Dwivedi S., Shukla K.K., John P.J., Sharma P. Vitamin C in Disease Prevention and Cure: An Overview. Indian J. Clin. Biochem. 2013;28:314–328. doi: 10.1007/s12291-013-0375-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Jiang Q. Natural Forms of Vitamin E: Metabolism, Antioxidant, and Anti-Inflammatory Activities and Their Role in Disease Prevention and Therapy. Free Radic. Biol. Med. 2014;72:76–90. doi: 10.1016/j.freeradbiomed.2014.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Almagro L., Sabater-Jara A.B., Belchí-Navarro S., Pedreño M.Á. Recent Trends in the Biotechnological Production of Tocopherols Using in Vitro Cultures. Phytochem. Rev. 2021;20:1193–1207. doi: 10.1007/s11101-021-09742-8. [DOI] [Google Scholar]
- 76.Pritam P., Deka R., Bhardwaj A., Srivastava R., Kumar D., Jha A.K., Jha N.K., Villa C., Jha S.K. Antioxidants in Alzheimer’s Disease: Current Therapeutic Significance and Future Prospects. Biology. 2022;11:212. doi: 10.3390/biology11020212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Szewczyk K., Chojnacka A., Górnicka M. Tocopherols and Tocotrienols—Bioactive Dietary Compounds; What Is Certain, What Is Doubt? Int. J. Mol. Sci. 2021;22:6222. doi: 10.3390/ijms22126222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Rizvi S., Raza S.T., Ahmed F., Ahmad A., Abbas S., Mahdi F. The Role of Vitamin e in Human Health and Some Diseases. Sultan Qaboos Univ. Med. J. 2014;14:e157-65. [PMC free article] [PubMed] [Google Scholar]
- 79.Rudrapal M., Khairnar S.J., Khan J., Dukhyil A.B., Ansari M.A., Alomary M.N., Alshabrmi F.M., Palai S., Deb P.K., Devi R. Dietary Polyphenols and Their Role in Oxidative Stress-Induced Human Diseases: Insights Into Protective Effects, Antioxidant Potentials and Mechanism(s) of Action. Front. Pharmacol. 2022;13:806470. doi: 10.3389/fphar.2022.806470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wu S., Chen W., Lu S., Zhang H., Yin L. Metabolic Engineering of Shikimic Acid Biosynthesis Pathway for the Production of Shikimic Acid and Its Branched Products in Microorganisms: Advances and Prospects. Molecules. 2022;27:4779. doi: 10.3390/molecules27154779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Kanner J. Food Polyphenols as Preventive Medicine. Antioxidants. 2023;12:2103. doi: 10.3390/antiox12122103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lobo V., Patil A., Phatak A., Chandra N. Free Radicals, Antioxidants and Functional Foods: Impact on Human Health. Pharmacogn. Rev. 2010;4:118. doi: 10.4103/0973-7847.70902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ullah A., Munir S., Badshah S.L., Khan N., Ghani L., Poulson B.G., Emwas A.-H., Jaremko M. Important Flavonoids and Their Role as a Therapeutic Agent. Molecules. 2020;25:5243. doi: 10.3390/molecules25225243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hassanein E.H.M., Althagafy H.S., Baraka M.A., Abd-alhameed E.K., Ibrahim I.M., Abd El-Maksoud M.S., Mohamed N.M., Ross S.A. The Promising Antioxidant Effects of Lignans: Nrf2 Activation Comes into View. Naunyn Schmiedebergs Arch. Pharmacol. 2024 doi: 10.1007/s00210-024-03102-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kumar N., Goel N. Phenolic Acids: Natural Versatile Molecules with Promising Therapeutic Applications. Biotechnol. Rep. 2019;24:e00370. doi: 10.1016/j.btre.2019.e00370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Panche A.N., Diwan A.D., Chandra S.R. Flavonoids: An Overview. J. Nutr. Sci. 2016;5:e47. doi: 10.1017/jns.2016.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Dias M.C., Pinto D.C.G.A., Silva A.M.S. Plant Flavonoids: Chemical Characteristics and Biological Activity. Molecules. 2021;26:5377. doi: 10.3390/molecules26175377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lü J., Lin P.H., Yao Q., Chen C. Chemical and Molecular Mechanisms of Antioxidants: Experimental Approaches and Model Systems. J. Cell. Mol. Med. 2010;14:840–860. doi: 10.1111/j.1582-4934.2009.00897.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kumar S., Pandey A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013;2013:162750. doi: 10.1155/2013/162750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Reinisalo M., Kårlund A., Koskela A., Kaarniranta K., Karjalainen R.O. Polyphenol Stilbenes: Molecular Mechanisms of Defence against Oxidative Stress and Aging-Related Diseases. Oxid. Med. Cell. Longev. 2015;2015:340520. doi: 10.1155/2015/340520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Pinyaev S.I., Kuzmenko T.P., Revina N.V., Parchaykina M.V., Pronin A.S., Syusin I.V., Novozhilova O.S., Revin V.V., Chudaikina E.V., Revina E.S. Influence of Resveratrol on Oxidation Processes and Lipid Phase Characteristics in Damaged Somatic Nerves. BioMed Res. Int. 2019;2019:2381907. doi: 10.1155/2019/2381907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang H.H., Liu M., Portincasa P., Wang D.Q.-H. Lipid Transfer in Lipoprotein Metabolism and Cardiovascular Disease. Advances in Experimental Medicine and Biology. Spinger; Berlin/Heidelberg, Germany: 2020. Recent Advances in the Critical Role of the Sterol Efflux Transporters ABCG5/G8 in Health and Disease; pp. 105–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Bakrim S., Benkhaira N., Bourais I., Benali T., Lee L.-H., El Omari N., Sheikh R.A., Goh K.W., Ming L.C., Bouyahya A. Health Benefits and Pharmacological Properties of Stigmasterol. Antioxidants. 2022;11:1912. doi: 10.3390/antiox11101912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Arivarasu L. In-Vitro Antioxidant Potential of Beta-Sitosterol: A Preface. Cureus. 2023;15:e45617. doi: 10.7759/cureus.45617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Li X., Xin Y., Mo Y., Marozik P., He T., Guo H. The Bioavailability and Biological Activities of Phytosterols as Modulators of Cholesterol Metabolism. Molecules. 2022;27:523. doi: 10.3390/molecules27020523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Hashim A.M., Alharbi B.M., Abdulmajeed A.M., Elkelish A., Hozzein W.N., Hassan H.M. Oxidative Stress Responses of Some Endemic Plants to High Altitudes by Intensifying Antioxidants and Secondary Metabolites Content. Plants. 2020;9:869. doi: 10.3390/plants9070869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Crupi P., Faienza M.F., Naeem M.Y., Corbo F., Clodoveo M.L., Muraglia M. Overview of the Potential Beneficial Effects of Carotenoids on Consumer Health and Well-Being. Antioxidants. 2023;12:1069. doi: 10.3390/antiox12051069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mannino G., Di Stefano V., Lauria A., Pitonzo R., Gentile C. Vaccinium macrocarpon (Cranberry)-Based Dietary Supplements: Variation in Mass Uniformity, Proanthocyanidin Dosage and Anthocyanin Profile Demonstrates Quality Control Standard Needed. Nutrients. 2020;12:992. doi: 10.3390/nu12040992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Fagbohun O.F., Gillies C.R., Murphy K.P.J., Rupasinghe H.P.V. Role of Antioxidant Vitamins and Other Micronutrients on Regulations of Specific Genes and Signaling Pathways in the Prevention and Treatment of Cancer. Int. J. Mol. Sci. 2023;24:6092. doi: 10.3390/ijms24076092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cory H., Passarelli S., Szeto J., Tamez M., Mattei J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018;5:370438. doi: 10.3389/fnut.2018.00087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chiang H.-M., Chen H.-C., Wu C.-S., Wu P.-Y., Wen K.-C. Rhodiola Plants: Chemistry and Biological Activity. J. Food Drug Anal. 2015;23:359–369. doi: 10.1016/j.jfda.2015.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Vezza T., Canet F., de Marañón A.M., Bañuls C., Rocha M., Víctor V.M. Phytosterols: Nutritional Health Players in the Management of Obesity and Its Related Disorders. Antioxidants. 2020;9:1266. doi: 10.3390/antiox9121266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Radomska-Leśniewska D.M., Skopiński P., Bałan B.J., Białoszewska A., Jóźwiak J., Rokicki D., Skopińska-Różewska E., Borecka A., Hevelke A. Review Paper Angiomodulatory Properties of Rhodiola spp. and other Natural Antioxidants. Cent. Eur. J. Immunol. 2015;2:249–262. doi: 10.5114/ceji.2015.52839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Panth N., Paudel K.R., Parajuli K. Reactive Oxygen Species: A Key Hallmark of Cardiovascular Disease. Adv. Med. 2016;2016:9152732. doi: 10.1155/2016/9152732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Münzel T., Camici G.G., Maack C., Bonetti N.R., Fuster V., Kovacic J.C. Impact of Oxidative Stress on the Heart and Vasculature. J. Am. Coll. Cardiol. 2017;70:212–229. doi: 10.1016/j.jacc.2017.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Padovan J.C., Dourado T.M.H., Pimenta G.F., Bruder-Nascimento T., Tirapelli C.R. Reactive Oxygen Species Are Central Mediators of Vascular Dysfunction and Hypertension Induced by Ethanol Consumption. Antioxidants. 2023;12:1813. doi: 10.3390/antiox12101813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Leventopoulos G., Koros R., Travlos C., Perperis A., Chronopoulos P., Tsoni E., Koufou E.-E., Papageorgiou A., Apostolos A., Kaouris P., et al. Mechanisms of Atrial Fibrillation: How Our Knowledge Affects Clinical Practice. Life. 2023;13:1260. doi: 10.3390/life13061260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Chang X., Zhang T., Zhang W., Zhao Z., Sun J. Natural Drugs as a Treatment Strategy for Cardiovascular Disease through the Regulation of Oxidative Stress. Oxid. Med. Cell. Longev. 2020;2020:5430407. doi: 10.1155/2020/5430407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Tungmunnithum D., Thongboonyou A., Pholboon A., Yangsabai A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines. 2018;5:93. doi: 10.3390/medicines5030093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Parham S., Kharazi A.Z., Bakhsheshi-Rad H.R., Nur H., Ismail A.F., Sharif S., RamaKrishna S., Berto F. Antioxidant, Antimicrobial and Antiviral Properties of Herbal Materials. Antioxidants. 2020;9:1309. doi: 10.3390/antiox9121309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Wilken R., Veena M.S., Wang M.B., Srivatsan E.S. Curcumin: A Review of Anti-Cancer Properties and Therapeutic Activity in Head and Neck Squamous Cell Carcinoma. Mol. Cancer. 2011;10:12. doi: 10.1186/1476-4598-10-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Mokra D., Joskova M., Mokry J. Therapeutic Effects of Green Tea Polyphenol (—)-Epigallocatechin-3-Gallate (EGCG) in Relation to Molecular Pathways Controlling Inflammation, Oxidative Stress, and Apoptosis. Int. J. Mol. Sci. 2022;24:340. doi: 10.3390/ijms24010340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Yang D., Wang T., Long M., Li P. Quercetin: Its Main Pharmacological Activity and Potential Application in Clinical Medicine. Oxid. Med. Cell. Longev. 2020;2020:8825387. doi: 10.1155/2020/8825387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Zhang Y.J., Gan R.Y., Li S., Zhou Y., Li A.N., Xu D.P., Li H.B., Kitts D.D. Antioxidant Phytochemicals for the Prevention and Treatment of Chronic Diseases. Molecules. 2015;20:21138–21156. doi: 10.3390/molecules201219753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Dobhal Y., Parcha V., Dhasmana D.C. Characterization of New Cardioprotective Principle Isolated from Methanolic Extract of Allium humile Leaves from Himalayan Region. Bangladesh J. Pharmacol. 2016;11:383. doi: 10.3329/bjp.v11i2.25991. [DOI] [Google Scholar]
- 116.Giuseppe D., Angela D., Davide R., Pamela M. Effects of a Combination of Berberis aristata, Silybum marianum and Monacolin on Lipid Profile in Subjects at Low Cardiovascular Risk; A Double-Blind, Randomized, Placebo-Controlled Trial. Int. J. Mol. Sci. 2017;18:343. doi: 10.3390/ijms18020343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Habanova M., Saraiva J.A., Haban M., Schwarzova M., Chlebo P., Predna L., Gažo J., Wyka J. Intake of Bilberries (Vaccinium myrtillus L.) Reduced Risk Factors for Cardiovascular Disease by Inducing Favorable Changes in Lipoprotein Profiles. Nutr. Res. 2016;36:1415–1422. doi: 10.1016/j.nutres.2016.11.010. [DOI] [PubMed] [Google Scholar]
- 118.Karcheva-Bahchevanska D., Nikolova M., Iliev I. Inhibitory Potential of Different Bilberry (Vaccinium myrtillus L.) Extracts on Human Salivary α-Amylase. Molecules. 2023;28:5820. doi: 10.3390/molecules28155820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Sun X., Jia B., Sun J., Lin J., Lu B., Duan J., Li C., Wang Q., Zhang X., Tan M., et al. Gastrodia elata Blume: A Review of Its Mechanisms and Functions on Cardiovascular Systems. Fitoterapia. 2023;167:105511. doi: 10.1016/j.fitote.2023.105511. [DOI] [PubMed] [Google Scholar]
- 120.Chen F., Zhang H.-Y., He D., Rao C.-M., Xu B. Cardioprotective Effect of Gynostemma pentaphyllum against Streptozotocin Induced Cardiac Toxicity in Rats via Alteration of AMPK/Nrf2/HO-1 Pathway. J. Oleo Sci. 2022;71:ess21281. doi: 10.5650/jos.ess21281. [DOI] [PubMed] [Google Scholar]
- 121.Dai N., Zhao F., Fang M., Pu F., Kong L., Liu J. Gynostemma pentaphyllum for Dyslipidemia: A Systematic Review of Randomized Controlled Trials. Front. Pharmacol. 2022;13:917521. doi: 10.3389/fphar.2022.917521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Wu M., Yu Z., Li X., Zhang X., Wang S., Yang S., Hu L., Liu L. Paeonol for the Treatment of Atherosclerotic Cardiovascular Disease: A Pharmacological and Mechanistic Overview. Front. Cardiovasc. Med. 2021;8:690116. doi: 10.3389/fcvm.2021.690116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Zhang H., Chen L., Sun X., Yang Q., Wan L., Guo C. Matrine: A Promising Natural Product with Various Pharmacological Activities. Front. Pharmacol. 2020;11:588. doi: 10.3389/fphar.2020.00588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Jung Y.A., Wan X., Yan H., Row K.H. Determination of Matrine and Oxymatrine in Sophora flavescens Ait. via High Performance Liquid Chromatography. J. Liq. Chromatogr. Relat. Technol. 2008;31:2752–2761. doi: 10.1080/10826070802388326. [DOI] [Google Scholar]
- 125.Janda K., Wojtkowska K., Jakubczyk K., Antoniewicz J., Skonieczna-Żydecka K. Passiflora incarnata in Neuropsychiatric Disorders—A Systematic Review. Nutrients. 2020;12:3894. doi: 10.3390/nu12123894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Achika J.I., Yusuf A.J., Ayo R.G., Liman D.U. Flavonoids from Nigerian Indigenous Medicinal Plants and Their Bioactivities: A Review. Phytomed. Plus. 2023;3:100405. doi: 10.1016/j.phyplu.2023.100405. [DOI] [Google Scholar]
- 127.Lam K.Y., Ling A.P.K., Koh R.Y., Wong Y.P., Say Y.H. A Review on Medicinal Properties of Orientin. Adv. Pharmacol. Sci. 2016;2016:4104595. doi: 10.1155/2016/4104595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ding L., Liu Z., Zhao F., Bai G., Chen L., Yao X., Qiu F. Isolation and Identification of the Metabolites of Paeonol in Human Urine. Xenobiotica. 2012;42:1206–1212. doi: 10.3109/00498254.2012.694492. [DOI] [PubMed] [Google Scholar]
- 129.Zhang M.-X., Huang X.-Y., Song Y., Xu W.-L., Li Y.-L., Li C. Astragalus propinquus Schischkin and Salvia miltiorrhiza Bunge Promote Angiogenesis to Treat Myocardial Ischemia via Ang-1/Tie-2/FAK Pathway. Front. Pharmacol. 2023;13:1103557. doi: 10.3389/fphar.2022.1103557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Fan C., Sun X., Wang X., Yu H. Therapeutic Potential of the Chemical Composition of Dendrobium nobile Lindl. Front. Pharmacol. 2023;14:1163830. doi: 10.3389/fphar.2023.1163830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Ren J., Fu L., Nile S.H., Zhang J., Kai G. Salvia miltiorrhiza in Treating Cardiovascular Diseases: A Review on Its Pharmacological and Clinical Applications. Front. Pharmacol. 2019;10:753. doi: 10.3389/fphar.2019.00753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Jiang Z., Gao W., Huang L. Tanshinones, Critical Pharmacological Components in Salvia miltiorrhiza. Front. Pharmacol. 2019;10:202. doi: 10.3389/fphar.2019.00202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Li D., Long Y., Yu S., Shi A., Wan J., Wen J., Li X., Liu S., Zhang Y., Li N., et al. Research Advances in Cardio-Cerebrovascular Diseases of Ligusticum Chuanxiong Hort. Front. Pharmacol. 2022;12:832673. doi: 10.3389/fphar.2021.832673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Lin J., Wang Q., Zhou S., Xu S., Yao K. Tetramethylpyrazine: A Review on Its Mechanisms and Functions. Biomed. Pharmacother. 2022;150:113005. doi: 10.1016/j.biopha.2022.113005. [DOI] [PubMed] [Google Scholar]
- 135.Jellinger K.A. Basic Mechanisms of Neurodegeneration: A Critical Update. J. Cell. Mol. Med. 2010;10:457–487. doi: 10.1111/j.1582-4934.2010.01010.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Niedzielska E., Smaga I., Gawlik M., Moniczewski A., Stankowicz P., Pera J., Filip M. Oxidative Stress in Neurodegenerative Diseases. Mol. Neurobiol. 2016;53:4094–4125. doi: 10.1007/s12035-015-9337-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Barnham K.J., Masters C.L., Bush A.I. Neurodegenerative Diseases and Oxidative Stress. Nat. Rev. Drug Discov. 2004;3:205–214. doi: 10.1038/nrd1330. [DOI] [PubMed] [Google Scholar]
- 138.Halliwell B. Reactive Species and Antioxidants. Redox Biology Is a Fundamental Theme of Aerobic Life. Plant Physiol. 2006;141:312–322. doi: 10.1104/pp.106.077073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Singh A., Kukreti R., Saso L., Kukreti S. Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules. 2019;24:1583. doi: 10.3390/molecules24081583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Alavi Naini S.M., Soussi-Yanicostas N. Tau Hyperphosphorylation and Oxidative Stress, a Critical Vicious Circle in Neurodegenerative Tauopathies? Oxid. Med. Cell. Longev. 2015;2015:151979. doi: 10.1155/2015/151979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Csala M., Kardon T., Legeza B., Lizák B., Mandl J., Margittai É., Puskás F., Száraz P., Szelényi P., Bánhegyi G. On the Role of 4-Hydroxynonenal in Health and Disease. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2015;1852:826–838. doi: 10.1016/j.bbadis.2015.01.015. [DOI] [PubMed] [Google Scholar]
- 142.Brand-Yavin A., Yavin E. Handbook of Neurochemistry and Molecular Neurobiology. Springer; Boston, MA, USA: 2009. Brain Oxidative Stress from a Phospholipid Perspective; pp. 603–630. [DOI] [Google Scholar]
- 143.Piccialli I., Tedeschi V., Caputo L., D’Errico S., Ciccone R., De Feo V., Secondo A., Pannaccione A. Exploring the Therapeutic Potential of Phytochemicals in Alzheimer’s Disease: Focus on Polyphenols and Monoterpenes. Front. Pharmacol. 2022;13:876614. doi: 10.3389/fphar.2022.876614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Mishra S., Palanivelu K. The Effect of Curcumin (Turmeric) on Alzheimer′s Disease: An Overview. Ann. Indian Acad. Neurol. 2008;11:13. doi: 10.4103/0972-2327.40220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Rahman M.H., Akter R., Bhattacharya T., Abdel-Daim M.M., Alkahtani S., Arafah M.W., Al-Johani N.S., Alhoshani N.M., Alkeraishan N., Alhenaky A., et al. Resveratrol and Neuroprotection: Impact and Its Therapeutic Potential in Alzheimer’s Disease. Front. Pharmacol. 2020;11:619024. doi: 10.3389/fphar.2020.619024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Minocha T., Birla H., Obaid A.A., Rai V., Sushma P., Shivamallu C., Moustafa M., Al-Shehri M., Al-Emam A., Tikhonova M.A., et al. Flavonoids as Promising Neuroprotectants and Their Therapeutic Potential against Alzheimer’s Disease. Oxid. Med. Cell. Longev. 2022;2022:6038996. doi: 10.1155/2022/6038996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Kujawska M., Jodynis-Liebert J. Polyphenols in Parkinson’s Disease: A Systematic Review of In Vivo Studies. Nutrients. 2018;10:642. doi: 10.3390/nu10050642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Klomparens E., Ding Y. The Neuroprotective Mechanisms and Effects of Sulforaphane. Brain Circ. 2019;5:74. doi: 10.4103/bc.bc_7_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Wang J., Sun B.-L., Xiang Y., Tian D.-Y., Zhu C., Li W.-W., Liu Y.-H., Bu X.-L., Shen L.-L., Jin W.-S., et al. Capsaicin Consumption Reduces Brain Amyloid-Beta Generation and Attenuates Alzheimer’s Disease-Type Pathology and Cognitive Deficits in APP/PS1 Mice. Transl. Psychiatry. 2020;10:230. doi: 10.1038/s41398-020-00918-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Magalingam K.B., Radhakrishnan A.K., Haleagrahara N. Protective Mechanisms of Flavonoids in Parkinson’s Disease. Oxid. Med. Cell. Longev. 2015;2015:314560. doi: 10.1155/2015/314560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Chandran R., Abrahamse H. Identifying Plant-Based Natural Medicine against Oxidative Stress and Neurodegenerative Disorders. Oxid. Med. Cell. Longev. 2020;2020:8648742. doi: 10.1155/2020/8648742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Uță G., Manolescu D.Ș., Avram S. Therapeutic Properties of Several Chemical Compounds of Salvia officinalis L. in Alzheimer’s Disease. Mini-Rev. Med. Chem. 2021;21:1421–1430. doi: 10.2174/1389557521999201230200209. [DOI] [PubMed] [Google Scholar]
- 153.Ariani A., Ghofar I., Khotimah H., Nurdiana N., Rahayu M. Asiatic Acid in Centella asiatica Extract towards Morphological Development in an Intermittent Hypoxia Intrauterine Embryo Model and Molecular Prediction Pathway of Insulin-like Growth Factor-1 (IGF-1) Receptor Signalling. Open Vet. J. 2023;13:629. doi: 10.5455/OVJ.2023.v13.i5.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Mushtaq Z., Imran M., Hussain M., Saeed F., Imran A., Umar M., Abdelgawad M.A., El-Ghorab A.H., Ahmed A., Alsagaby S.A., et al. Asiatic Acid: A Review on Its Polypharmacological Properties and Therapeutic Potential against Various Maladies. Int. J. Food Prop. 2023;26:1244–1263. doi: 10.1080/10942912.2023.2209702. [DOI] [Google Scholar]
- 155.Wong J.H., Barron A.M., Abdullah J.M. Mitoprotective Effects of Centella asiatica (L.) Urb.: Anti-Inflammatory and Neuroprotective Opportunities in Neurodegenerative Disease. Front. Pharmacol. 2021;12:687935. doi: 10.3389/fphar.2021.687935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Bandopadhyay S., Mandal S., Ghorai M., Jha N.K., Kumar M., Radha. Ghosh A., Proćków J., Pérez de la Lastra J.M., Dey A. Therapeutic Properties and Pharmacological Activities of Asiaticoside and Madecassoside: A Review. J. Cell. Mol. Med. 2023;27:593–608. doi: 10.1111/jcmm.17635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Fatima U., Roy S., Ahmad S., Al-Keridis L.A., Alshammari N., Adnan M., Islam A., Hassan M.I. Investigating Neuroprotective Roles of Bacopa Monnieri Extracts: Mechanistic Insights and Therapeutic Implications. Biomed. Pharmacother. 2022;153:113469. doi: 10.1016/j.biopha.2022.113469. [DOI] [PubMed] [Google Scholar]
- 158.Gubbannavar J., Chandola H., Harisha C., Khanpara K., Shukla V. A Comparative Pharmacognostical and Preliminary Physico-Chemical Analysis of Stem and Leaf of Bacopa monnieri (L.) Pennel and Bacopa Floribunda (R.BR.) Wettst. AYU. 2013;34:95. doi: 10.4103/0974-8520.115441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Gray N.E., Alcazar Magana A., Lak P., Wright K.M., Quinn J., Stevens J.F., Maier C.S., Soumyanath A. Centella asiatica: Phytochemistry and Mechanisms of Neuroprotection and Cognitive Enhancement. Phytochem. Rev. 2018;17:161–194. doi: 10.1007/s11101-017-9528-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Orhan I.E. Centella Asiatica (L.) Urban: From Traditional Medicine to Modern Medicine with Neuroprotective Potential. Evid.-Based Complement. Altern. Med. 2012;2012:946259. doi: 10.1155/2012/946259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Ehrnhöfer-Ressler M.M., Fricke K., Pignitter M., Walker J.M., Walker J., Rychlik M., Somoza V. Identification of 1,8-Cineole, Borneol, Camphor, and Thujone as Anti-Inflammatory Compounds in a Salvia officinalis L. Infusion Using Human Gingival Fibroblasts. J. Agric. Food Chem. 2013;61:3451–3459. doi: 10.1021/jf305472t. [DOI] [PubMed] [Google Scholar]
- 162.Ghorbani A., Esmaeilizadeh M. Pharmacological Properties of Salvia officinalis and Its Components. J. Tradit. Complement. Med. 2017;7:433–440. doi: 10.1016/j.jtcme.2016.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Tundis R., Leporini M., Bonesi M., Rovito S., Passalacqua N.G. Salvia officinalis L. from Italy: A Comparative Chemical and Biological Study of Its Essential Oil in the Mediterranean Context. Molecules. 2020;25:5826. doi: 10.3390/molecules25245826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Shoukat S., Zia M.A., Uzair M., Attia K.A., Abushady A.M., Fiaz S., Ali S., Yang S.H., Ali G.M. Bacopa monnieri: A Promising Herbal Approach for Neurodegenerative Disease Treatment Supported by in Silico and in Vitro Research. Heliyon. 2023;9:e21161. doi: 10.1016/j.heliyon.2023.e21161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Wu P., Chang C., Zhu G., Zhai L., Zhang X., Huan Q., Gao Z., Deng H., Liang Y., Xiao H. Network Pharmacology Study of Bioactive Components and Molecular Mechanisms of the Glycoside Fraction from Picrorhiza scrophulariiflora Against Experimental Colitis. Drug Des. Dev. Ther. 2023;17:1531–1546. doi: 10.2147/DDDT.S407339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.He Z., Hu Y., Niu Z., Zhong K., Liu T., Yang M., Ji L., Hu W. A Review of Pharmacokinetic and Pharmacological Properties of Asiaticoside, a Major Active Constituent of Centella asiatica (L.) Urb. J. Ethnopharmacol. 2023;302:115865. doi: 10.1016/j.jep.2022.115865. [DOI] [PubMed] [Google Scholar]
- 167.Agnihotri V., Walia M., Pathania V., Singh B., Kant K. Evaluation of Antioxidant Activity of Picrorhiza kurroa (Leaves) Extracts. Indian J. Pharm. Sci. 2013;75:324. doi: 10.4103/0250-474X.117438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Burchardt P., Żurawski J., Zuchowski B., Kubacki T., Murawa D., Wiktorowicz K., Wysocki H. State of the Art Paper Low-Density Lipoprotein, Its Susceptibility to Oxidation and the Role of Lipoprotein-Associated Phospholipase A2 and Carboxyl Ester Lipase Lipases in Atherosclerotic Plaque Formation. Arch. Med. Sci. 2013;1:151–158. doi: 10.5114/aoms.2013.33176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Leopold J.A., Loscalzo J. Oxidative Mechanisms and Atherothrombotic Cardiovascular Disease. Drug Discov. Today Ther. Strateg. 2008;5:5–13. doi: 10.1016/j.ddstr.2008.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Savini I., Catani M., Evangelista D., Gasperi V., Avigliano L. Obesity-Associated Oxidative Stress: Strategies Finalized to Improve Redox State. Int. J. Mol. Sci. 2013;14:10497–10538. doi: 10.3390/ijms140510497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Blagov A., Nedosugova L., Kirichenko T., Sukhorukov V., Melnichenko A., Orekhov A. Mitochondrial Dysfunction as a Factor of Energy Metabolism Disorders in Type 2 Diabetes Mellitus. Front. Biosci. Sch. 2024;16:5. doi: 10.31083/j.fbs1601005. [DOI] [PubMed] [Google Scholar]
- 172.Dludla P.V., Mabhida S.E., Ziqubu K., Nkambule B.B., Mazibuko-Mbeje S.E., Hanser S., Basson A.K., Pheiffer C., Kengne A.P. Pancreatic β-Cell Dysfunction in Type 2 Diabetes: Implications of Inflammation and Oxidative Stress. World J. Diabetes. 2023;14:130–146. doi: 10.4239/wjd.v14.i3.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Holvoet P. Stress in Obesity and Associated Metabolic and Cardiovascular Disorders. Scientifica. 2012;2012:205027. doi: 10.6064/2012/205027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Moutia M., El Azhary K., Elouaddari A., Al Jahid A., Jamal Eddine J., Seghrouchni F., Habti N., Badou A. Capparis spinosa L. Promotes Anti-Inflammatory Response in Vitro through the Control of Cytokine Gene Expression in Human Peripheral Blood Mononuclear Cells. BMC Immunol. 2016;17:26. doi: 10.1186/s12865-016-0164-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Rocha D.H.A., Pinto D.C.G.A., Silva A.M.S. Macroalgae Specialized Metabolites: Evidence for Their Anti-Inflammatory Health Benefits. Mar. Drugs. 2022;20:789. doi: 10.3390/md20120789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Lourenço S.C., Moldão-Martins M., Alves V.D. Antioxidants of Natural Plant Origins: From Sources to Food Industry Applications. Molecules. 2019;24:4132. doi: 10.3390/molecules24224132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Cione E., La Torre C., Cannataro R., Caroleo M.C., Plastina P., Gallelli L. Quercetin, Epigallocatechin Gallate, Curcumin, and Resveratrol: From Dietary Sources to Human MicroRNA Modulation. Molecules. 2019;25:63. doi: 10.3390/molecules25010063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Ghafouri-Fard S., Shoorei H., Bahroudi Z., Hussen B.M., Talebi S.F., Taheri M., Ayatollahi S.A. Nrf2-Related Therapeutic Effects of Curcumin in Different Disorders. Biomolecules. 2022;12:82. doi: 10.3390/biom12010082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.García-Martínez B.I., Ruiz-Ramos M., Pedraza-Chaverri J., Santiago-Osorio E., Mendoza-Núñez V.M. Effect of Resveratrol on Markers of Oxidative Stress and Sirtuin 1 in Elderly Adults with Type 2 Diabetes. Int. J. Mol. Sci. 2023;24:7422. doi: 10.3390/ijms24087422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Xu D., Hu M.-J., Wang Y.-Q., Cui Y.-L. Antioxidant Activities of Quercetin and Its Complexes for Medicinal Application. Molecules. 2019;24:1123. doi: 10.3390/molecules24061123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Wang H., Zhang H., Gao Z., Zhang Q., Gu C. The Mechanism of Berberine Alleviating Metabolic Disorder Based on Gut Microbiome. Front. Cell. Infect. Microbiol. 2022;12:854885. doi: 10.3389/fcimb.2022.854885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Capece U., Moffa S., Improta I., Di Giuseppe G., Nista E.C., Cefalo C.M.A., Cinti F., Pontecorvi A., Gasbarrini A., Giaccari A., et al. Alpha-Lipoic Acid and Glucose Metabolism: A Comprehensive Update on Biochemical and Therapeutic Features. Nutrients. 2022;15:18. doi: 10.3390/nu15010018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Yang S., Tao G., Yang L., Wu X., Liu J., Dagher F., Ou S., Song Y., Huang J. Dietary Phytochemical and Metabolic Disease Prevention: Focus on Plant Proteins. Front. Nutr. 2023;10:1089487. doi: 10.3389/fnut.2023.1089487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Martchenko A., Papaelias A., Bolz S.-S. Physiologic Effects of the Maqui Berry (Aristotelia chilensis): A Focus on Metabolic Homeostasis. Food Funct. 2024;15:4724–4740. doi: 10.1039/D3FO02524A. [DOI] [PubMed] [Google Scholar]
- 185.Kunc N., Hudina M., Osterc G., Bavcon J., Ravnjak B., Mikulič-Petkovšek M. Phenolic Compounds of Rose Hips of Some Rosa Species and Their Hybrids Native Grown in the South-West of Slovenia during a Two-Year Period (2020–2021) Foods. 2023;12:1952. doi: 10.3390/foods12101952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Oprica L., Bucsa C., Zamfirache M.M. Ascorbic Acid Content of Rose Hip Fruit Depending on Altitude. Iran. J. Public Health. 2015;44:138–139. [PMC free article] [PubMed] [Google Scholar]
- 187.Gan R.-Y., Zhang D., Wang M., Corke H. Health Benefits of Bioactive Compounds from the Genus Ilex, a Source of Traditional Caffeinated Beverages. Nutrients. 2018;10:1682. doi: 10.3390/nu10111682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Louisa M., Patintingan C.G.H., Wardhani B.W.K. Moringa Oleifera Lam. in Cardiometabolic Disorders: A Systematic Review of Recent Studies and Possible Mechanism of Actions. Front. Pharmacol. 2022;13:792794. doi: 10.3389/fphar.2022.792794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Wang F., Bao Y., Shen X., Zengin G., Lyu Y., Xiao J., Weng Z. Niazirin from Moringa oleifera Lam. Attenuates High Glucose-Induced Oxidative Stress through PKCζ/Nox4 Pathway. Phytomedicine. 2021;86:153066. doi: 10.1016/j.phymed.2019.153066. [DOI] [PubMed] [Google Scholar]
- 190.Akaberi M., Hosseinzadeh H. Grapes (Vitis vinifera) as a Potential Candidate for the Therapy of the Metabolic Syndrome. Phytother. Res. 2016;30:540–556. doi: 10.1002/ptr.5570. [DOI] [PubMed] [Google Scholar]
- 191.Graf B.L., Raskin I., Cefalu W.T., Ribnicky D.M. Plant-Derived Therapeutics for the Treatment of Metabolic Syndrome. Curr. Opin. Investig. Drugs. 2010;11:1107–1115. [PMC free article] [PubMed] [Google Scholar]
- 192.Wang Y., Zhang D., Ye W., Yin Z., Fung K.-P., Zhao S., Yao X. Triterpenoid Saponins from Androsace umbellata and Their Anti-Proliferative Activities in Human Hepatoma Cells. Planta Med. 2008;74:1280–1284. doi: 10.1055/s-2008-1081291. [DOI] [PubMed] [Google Scholar]
- 193.Xu D.-P., Li Y., Meng X., Zhou T., Zhou Y., Zheng J., Zhang J.-J., Li H.-B. Natural Antioxidants in Foods and Medicinal Plants: Extraction, Assessment and Resources. Int. J. Mol. Sci. 2017;18:96. doi: 10.3390/ijms18010096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Dumanović J., Nepovimova E., Natić M., Kuča K., Jaćević V. The Significance of Reactive Oxygen Species and Antioxidant Defense System in Plants: A Concise Overview. Front. Plant Sci. 2021;11:552969. doi: 10.3389/fpls.2020.552969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Askew E.W. Work at High Altitude and Oxidative Stress: Antioxidant Nutrients. Toxicology. 2002;180:107–119. doi: 10.1016/S0300-483X(02)00385-2. [DOI] [PubMed] [Google Scholar]
- 196.Seleiman M.F., Al-Suhaibani N., Ali N., Akmal M., Alotaibi M., Refay Y., Dindaroglu T., Abdul-Wajid H.H., Battaglia M.L. Drought Stress Impacts on Plants and Different Approaches to Alleviate Its Adverse Effects. Plants. 2021;10:259. doi: 10.3390/plants10020259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Siyar S., Sami S., Majeed A. Cellular and Molecular Phytotoxicity of Heavy Metals. Springer; Berlin/Heidelberg, Germany: 2020. Heavy Metal Stress in Plants: Effects on Nutrients and Water Uptake; pp. 89–98. [DOI] [Google Scholar]
- 198.Petrovska B. Historical Review of Medicinal Plants’ Usage. Pharmacogn. Rev. 2012;6:1. doi: 10.4103/0973-7847.95849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Kaushik P., Ahlawat P., Singh K., Singh R. Chemical Constituents, Pharmacological Activities, and Uses of Common Ayurvedic Medicinal Plants: A Future Source of New Drugs. Adv. Tradit. Med. 2021;23:673–714. doi: 10.1007/s13596-021-00621-3. [DOI] [Google Scholar]
- 200.Butola J.S., Samant S.S. Saussurea Species in Indian Himalayan Region: Diversity, Distribution and Indigenous Uses. Int. J. Plant Biol. 2010;1:e9. doi: 10.4081/pb.2010.e9. [DOI] [Google Scholar]
- 201.Pandey M.M., Rastogi S., Rawat A.K.S. Saussurea costus: Botanical, Chemical and Pharmacological Review of an Ayurvedic Medicinal Plant. J. Ethnopharmacol. 2007;110:379–390. doi: 10.1016/j.jep.2006.12.033. [DOI] [PubMed] [Google Scholar]
- 202.Xing W., Wen C., Wang D., Shao H., Liu C., He C., Olatunji O.J. Cardiorenal Protective Effect of Costunolide against Doxorubicin-Induced Toxicity in Rats by Modulating Oxidative Stress, Inflammation and Apoptosis. Molecules. 2022;27:2122. doi: 10.3390/molecules27072122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Mlejnek P., Dolezel P., Kriegova E., Pastvova N. N-Acetylcysteine Can Induce Massive Oxidative Stress, Resulting in Cell Death with Apoptotic Features in Human Leukemia Cells. Int. J. Mol. Sci. 2021;22:12635. doi: 10.3390/ijms222312635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Elshaer S.E., Hamad G.M., Hafez E.E., Baghdadi H.H., El-Demerdash F.M., Simal-Gandara J. Root Extracts of Saussurea costus as Prospective Detoxifying Food Additive against Sodium Nitrite Toxicity in Male Rats. Food Chem. Toxicol. 2022;166:113225. doi: 10.1016/j.fct.2022.113225. [DOI] [PubMed] [Google Scholar]
- 205.Jain R., Bhardwaj P., Pandey S.S., Kumar S. Arnebia euchroma, a Plant Species of Cold Desert in the Himalayas, Harbors Beneficial Cultivable Endophytes in Roots and Leaves. Front. Microbiol. 2021;12:696667. doi: 10.3389/fmicb.2021.696667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Parray J.A., Kamili A.N., Jan S., Mir M.Y., Shameem N., Ganai B.A., Abd_Allah E.F., Hashem A., Alqarawi A.A. Manipulation of Plant Growth Regulators on Phytochemical Constituents and DNA Protection Potential of the Medicinal Plant Arnebia benthamii. BioMed Res. Int. 2018;2018:6870139. doi: 10.1155/2018/6870139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Kumar A., Shashni S., Kumar P., Pant D., Singh A., Verma R.K. Phytochemical Constituents, Distributions and Traditional Usages of Arnebia euchroma: A Review. J. Ethnopharmacol. 2021;271:113896. doi: 10.1016/j.jep.2021.113896. [DOI] [PubMed] [Google Scholar]
- 208.Su L., Yan G., Guan B., Xu W., Hao Y., Wang Y., Zhang Y., Liu L. Shikonin Derivatives Protect Immune Organs from Damage and Promote Immune Responses In Vivo in Tumour-Bearing Mice. Phytother. Res. 2012;26:26–33. doi: 10.1002/ptr.3503. [DOI] [PubMed] [Google Scholar]
- 209.Ganie S.A., Dar T.A., Hamid R., Zargar O., Abeer S.U., Masood A., Amin S., Zargar M.A. In Vitro Antioxidant and Cytotoxic Activities of Arnebia benthamii (Wall Ex. G. Don): A Critically Endangered Medicinal Plant of Kashmir Valley. Oxid. Med. Cell. Longev. 2014;2014:792574. doi: 10.1155/2014/792574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Shameem N., Kamili A.N., Parray J.A., Hamid R., Bandh S.A. Antimicrobial and Antioxidant Activity of Methanol Extracts of Arnebia benthamii (Wall Ex. G. Don) Johnston—A Critically Endangered Medicinal Plant of North Western Himalaya. J. Anal. Sci. Technol. 2015;6:36. doi: 10.1186/s40543-015-0076-z. [DOI] [Google Scholar]
- 211.Papp N., Purger D., Czigle S., Czégényi D., Stranczinger S., Tóth M., Dénes T., Kocsis M., Takácsi-Nagy A., Filep R. The Importance of Pine Species in the Ethnomedicine of Transylvania (Romania) Plants. 2022;11:2331. doi: 10.3390/plants11182331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Gülçin İ., Büyükokuroǧlu M.E., Oktay M., Küfrevioǧlu Ö.İ. Antioxidant and Analgesic Activities of Turpentine of Pinus nigra Arn. subsp. pallsiana (Lamb.) Holmboe. J. Ethnopharmacol. 2003;86:51–58. doi: 10.1016/S0378-8741(03)00036-9. [DOI] [PubMed] [Google Scholar]
- 213.Kurti F., Giorgi A., Beretta G., Mustafa B., Gelmini F., Testa C., Angioletti S., Giupponi L., Zilio E., Pentimalli D., et al. Chemical Composition, Antioxidant and Antimicrobial Activities of Essential Oils of Different Pinus Species from Kosovo. J. Essent. Oil Res. 2019;31:263–275. doi: 10.1080/10412905.2019.1584591. [DOI] [Google Scholar]
- 214.Bisht A., Jain S., Misra A., Dwivedi J., Paliwal S., Sharma S. Cedrus deodara (Roxb. Ex D.Don) G.Don: A Review of Traditional Use, Phytochemical Composition and Pharmacology. J. Ethnopharmacol. 2021;279:114361. doi: 10.1016/j.jep.2021.114361. [DOI] [PubMed] [Google Scholar]
- 215.Bhardwaj K., Silva A.S., Atanassova M., Sharma R., Nepovimova E., Musilek K., Sharma R., Alghuthaymi M.A., Dhanjal D.S., Nicoletti M., et al. Conifers Phytochemicals: A Valuable Forest with Therapeutic Potential. Molecules. 2021;26:3005. doi: 10.3390/molecules26103005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Samant S.S., Pant S. Diversity, Distribution Pattern and Conservation Status of the Plants Used in Liver Diseases/Ailments in Indian Himalayan Region. J. Mt. Sci. 2006;3:28–47. doi: 10.1007/s11629-006-0028-6. [DOI] [Google Scholar]
- 217.Anand U., Biswas P., Kumar V., Ray D., Ray P., Loake V.I.P., Kandimalla R., Chaudhary A., Singh B., Routhu N.K., et al. Podophyllum hexandrum and Its Active Constituents: Novel Radioprotectants. Biomed. Pharmacother. 2022;146:112555. doi: 10.1016/j.biopha.2021.112555. [DOI] [PubMed] [Google Scholar]
- 218.Cornara L., Ambu G., Trombetta D., Denaro M., Alloisio S., Frigerio J., Labra M., Ghimire G., Valussi M., Smeriglio A. Comparative and Functional Screening of Three Species Traditionally Used as Antidepressants: Valeriana officinalis L., Valeriana jatamansi Jones Ex Roxb. and Nardostachys jatamansi (D. Don) DC. Plants. 2020;9:994. doi: 10.3390/plants9080994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Raina A., Negi K. Essential Oil Composition of Valeriana jatamansi Jones from Himalayan Regions of India. Indian J. Pharm. Sci. 2015;77:218. doi: 10.4103/0250-474X.156614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Thusoo S., Gupta S., Sudan R., Kour J., Bhagat S., Hussain R., Bhagat M. Antioxidant Activity of Essential Oil and Extracts of Valeriana jatamansi Roots. BioMed Res. Int. 2014;2014:614187. doi: 10.1155/2014/614187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Keet J.-H., Cindi D.D., du Preez P.J. Assessing the Invasiveness of Berberis aristata and B. julianae (Berberidaceae) in South Africa: Management Options and Legal Recommendations. S. Afr. J. Bot. 2016;105:288–298. doi: 10.1016/j.sajb.2016.04.012. [DOI] [Google Scholar]
- 222.Singh J., Kakkar P. Antihyperglycemic and Antioxidant Effect of Berberis aristata Root Extract and Its Role in Regulating Carbohydrate Metabolism in Diabetic Rats. J. Ethnopharmacol. 2009;123:22–26. doi: 10.1016/j.jep.2009.02.038. [DOI] [PubMed] [Google Scholar]
- 223.Frezza C., Venditti A., Toniolo C., De Vita D., Serafini I., Ciccòla A., Franceschin M., Ventrone A., Tomassini L., Foddai S., et al. Pedicularis L. Genus: Systematics, Botany, Phytochemistry, Chemotaxonomy, Ethnopharmacology, and Other. Plants. 2019;8:306. doi: 10.3390/plants8090306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Yatoo M.I., Dimri U., Gopalakrishnan A., Karthik K., Gopi M., Khandia R., Saminathan M., Saxena A., Alagawany M., Farag M.R., et al. Beneficial Health Applications and Medicinal Values of Pedicularis Plants: A Review. Biomed. Pharmacother. 2017;95:1301–1313. doi: 10.1016/j.biopha.2017.09.041. [DOI] [PubMed] [Google Scholar]
- 225.Jeelani S.M., Siddique M.A.A., Rani S. Variations of Morphology, Ecology and Chromosomes of Aconitum heterophyllum Wall., an Endangered Alpine Medicinal Plant in Himalayas. Caryologia. 2015;68:294–305. doi: 10.1080/00087114.2015.1109933. [DOI] [Google Scholar]
- 226.Konda V.G.R. Antioxidant and Nephroprotective Activities of Aconitum heterophyllum Root in Glycerol Induced Acute Renal Failure in Rats. J. Clin. Diagn. Res. 2016;10:FF01. doi: 10.7860/JCDR/2016/10798.7388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Nengroo Z.R., Ganie A.S., Azeem M. Aconitum heterophylum from Kashmir: Evaluation of Fatty Acid Profile, Antibacterial, Antioxidant Activities and Functional Group Analysis. Carbohydr. Polym. Technol. Appl. 2021;2:100105. doi: 10.1016/j.carpta.2021.100105. [DOI] [Google Scholar]
- 228.Manzoor M., Ahmad M., Zafar M., Gillani S.W., Shaheen H., Pieroni A., Al-Ghamdi A.A., Elshikh M.S., Saqib S., Makhkamov T., et al. The Local Medicinal Plant Knowledge in Kashmir Western Himalaya: A Way to Foster Ecological Transition via Community-Centred Health Seeking Strategies. J. Ethnobiol. Ethnomed. 2023;19:56. doi: 10.1186/s13002-023-00631-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Jahan F., Alvi S.S., Islam M.H. Berberis aristata and Its Secondary Metabolites: Insights into Nutraceutical and Therapeutical Applications. Pharmacol. Res. Mod. Chin. Med. 2022;5:100184. doi: 10.1016/j.prmcm.2022.100184. [DOI] [Google Scholar]
- 230.Zhang H., Wei Y., Yue J., Wang Z., Zou H., Ji X., Zhang S., Liu Z. Prediction of Potential Suitable Areas and Priority Protection for Cupressus gigantea on the Tibetan Plateau. Plants. 2024;13:896. doi: 10.3390/plants13060896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Saha M., Sarker D., Kar P., Gupta P., Sen A. Indigenous Knowledge of Plants in Local Healthcare Management Practices by Tribal People of Malda District, India. J. Intercult. Ethnopharmacol. 2014;3:179. doi: 10.5455/jice.20140630022609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Ravindra J., UG Y., Pandyanda Nanjappa D., Kalladka K., Dhakal R., Chakraborty A., Chakraborty G. Allicin Extracted from Allium sativum Shows Potent Anti-Cancer and Antioxidant Properties in Zebrafish. Biomed. Pharmacother. 2023;169:115854. doi: 10.1016/j.biopha.2023.115854. [DOI] [PubMed] [Google Scholar]
- 233.Barbu I.A., Ciorîță A., Carpa R., Moț A.C., Butiuc-Keul A., Pârvu M. Phytochemical Characterization and Antimicrobial Activity of Several Allium Extracts. Molecules. 2023;28:3980. doi: 10.3390/molecules28103980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Yudhistira B., Punthi F., Lin J., Sulaimana A.S., Chang C., Hsieh C. S-Allyl Cysteine in Garlic (Allium sativum): Formation, Biofunction, and Resistance to Food Processing for Value-added Product Development. Compr. Rev. Food Sci. Food Saf. 2022;21:2665–2687. doi: 10.1111/1541-4337.12937. [DOI] [PubMed] [Google Scholar]
- 235.Zahoor M., Zafar R., Rahman N.U. Isolation and Identification of Phenolic Antioxidants from Pistacia integerrima Gall and Their Anticholine Esterase Activities. Heliyon. 2018;4:e01007. doi: 10.1016/j.heliyon.2018.e01007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Yeh M.-L., Liu C.-F., Huang C.-L., Huang T.-C. Hepatoprotective Effect of Angelica archangelica in Chronically Ethanol-Treated Mice. Pharmacology. 2003;68:70–73. doi: 10.1159/000069530. [DOI] [PubMed] [Google Scholar]
- 237.Park W.H., Kang S., Piao Y., Pak C.J., Oh M.S., Kim J., Kang M.S., Pak Y.K. Ethanol Extract of Bupleurum falcatum and Saikosaponins Inhibit Neuroinflammation via Inhibition of NF-ΚB. J. Ethnopharmacol. 2015;174:37–44. doi: 10.1016/j.jep.2015.07.039. [DOI] [PubMed] [Google Scholar]
- 238.Witaicenis A., Seito L.N., da Silveira Chagas A., de Almeida L.D., Luchini A.C., Rodrigues-Orsi P., Cestari S.H., Di Stasi L.C. Antioxidant and Intestinal Anti-Inflammatory Effects of Plant-Derived Coumarin Derivatives. Phytomedicine. 2014;21:240–246. doi: 10.1016/j.phymed.2013.09.001. [DOI] [PubMed] [Google Scholar]
- 239.Yaqoob U., Nawchoo I.A., Ahmad M. Phytochemical Evaluation of Various Solvent Extracts of the Leaves, Fruits and Shoots of Ferula jaeschkeana Vatke. Herb. Med. Open Access. 2016;2:2472-0151. doi: 10.21767/2472-0151.100019. [DOI] [Google Scholar]
- 240.Bahadori M.B., Dinparast L., Zengin G. The Genus Heracleum: A Comprehensive Review on Its Phytochemistry, Pharmacology, and Ethnobotanical Values as a Useful Herb. Compr. Rev. Food Sci. Food Saf. 2016;15:1018–1039. doi: 10.1111/1541-4337.12222. [DOI] [PubMed] [Google Scholar]
- 241.Ren Y., Song X., Tan L., Guo C., Wang M., Liu H., Cao Z., Li Y., Peng C. A Review of the Pharmacological Properties of Psoralen. Front. Pharmacol. 2020;11:571535. doi: 10.3389/fphar.2020.571535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Srivastava R.P., Dixit P., Singh L., Verma P.C., Saxena G. Status of Selinum spp. L. a Himalayan Medicinal Plant in India: A Review of Its Pharmacology, Phytochemistry and Traditional Uses. Curr. Pharm. Biotechnol. 2019;19:1122–1134. doi: 10.2174/1389201020666181227150829. [DOI] [PubMed] [Google Scholar]
- 243.Nisa S., Bibi Y., Masood S., Ali A., Alam S., Sabir M., Qayyum A., Ahmed W., Alharthi S., Santali E.Y., et al. Isolation, Characterization and Anticancer Activity of Two Bioactive Compounds from Arisaema flavum (Forssk.) Schott. Molecules. 2022;27:7932. doi: 10.3390/molecules27227932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Akhtar M., Shaukat A., Zahoor A., Chen Y., Wang Y., Yang M., Umar T., Guo M., Deng G. Anti-Inflammatory Effects of Hederacoside-C on Staphylococcus aureus Induced Inflammation via TLRs and Their Downstream Signal Pathway in Vivo and in Vitro. Microb. Pathog. 2019;137:103767. doi: 10.1016/j.micpath.2019.103767. [DOI] [PubMed] [Google Scholar]
- 245.Farasati Far B., Behzad G., Khalili H. Achillea Millefolium: Mechanism of Action, Pharmacokinetic, Clinical Drug-Drug Interactions and Tolerability. Heliyon. 2023;9:e22841. doi: 10.1016/j.heliyon.2023.e22841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Mohammed H.A. Phytochemical Analysis, Antioxidant Potential, and Cytotoxicity Evaluation of Traditionally Used Artemisia absinthium L. (Wormwood) Growing in the Central Region of Saudi Arabia. Plants. 2022;11:1028. doi: 10.3390/plants11081028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Li Z., Li Q., Wu J., Wang M., Yu J. Artemisinin and Its Derivatives as a Repurposing Anticancer Agent: What Else Do We Need to Do? Molecules. 2016;21:1331. doi: 10.3390/molecules21101331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Kozyra M., Kukula-Koch W., Szymański M. Phenolic Composition of Inflorescences of Carduus nutans L. Chem. Biodivers. 2022;19:e202100827. doi: 10.1002/cbdv.202100827. [DOI] [PubMed] [Google Scholar]
- 249.shaikh T., Rub R.A., Sasikumar S. Antimicrobial Screening of Cichorium intybus Seed Extracts. Arab. J. Chem. 2016;9:S1569–S1573. doi: 10.1016/j.arabjc.2012.04.012. [DOI] [Google Scholar]
- 250.Jan R., Khan M., Asaf S., Lubna. Asif S., Kim K.-M. Bioactivity and Therapeutic Potential of Kaempferol and Quercetin: New Insights for Plant and Human Health. Plants. 2022;11:2623. doi: 10.3390/plants11192623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Kalola J., Shah R., Patel A., Lahiri S.K., Shah M.B. Anti-Inflammatory and Immunomodulatory Activities of Inula cappa Roots (Compositae) J. Complement. Integr. Med. 2017;14:20160083. doi: 10.1515/jcim-2016-0083. [DOI] [PubMed] [Google Scholar]
- 252.Arumugam P., Murugan M. Antimutagenic and Antiapoptotic Effects of Aqueous Root Extract of Inula racemosa Hook. f. on 4-NQO-Induced Genetic Damage in Mice. ISRN Pharmacol. 2013;2013:768359. doi: 10.1155/2013/768359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Shah N.A., Khan M.R., Naz K., Khan M.A. Antioxidant Potential, DNA Protection, and HPLC-DAD Analysis of Neglected Medicinal Jurinea dolomiaea Roots. BioMed Res. Int. 2014;2014:726241. doi: 10.1155/2014/726241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Kumar A., Agnihotri V.K. Phytochemical Studies of Jurinea macrocephala Roots from Western Himalaya. Nat. Prod. Res. 2020;34:421–424. doi: 10.1080/14786419.2018.1530233. [DOI] [PubMed] [Google Scholar]
- 255.Chang K.-M., Choi S.-I., Kim G.-H. Anti-Oxidant Activity of Saussurea lappa C.B. Clarke Roots. Prev. Nutr. Food Sci. 2012;17:306–309. doi: 10.3746/pnf.2012.17.4.306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Rathore S., Debnath P., Kumar R. Kuth Saussurea costus (Falc.) Lipsch.: A Critically Endangered Medicinal Plant from Himalaya. J. Appl. Res. Med. Aromat. Plants. 2021;20:100277. doi: 10.1016/j.jarmap.2020.100277. [DOI] [Google Scholar]
- 257.Mishra A.P., Saklani S., Parcha V., Nigam M., Coutinho H.D.M. Antibacterial Activity and Phytochemical Characterisation of Saussurea gossypiphora D. Don. Arch. Microbiol. 2021;203:5055–5065. doi: 10.1007/s00203-021-02494-1. [DOI] [PubMed] [Google Scholar]
- 258.Jaghthmi O., Zeid I. Hypoglycemic and Hepatoprotective Effect of Rhizophora mucronata and Avicennia marina against Streptozotocin-Induced Diabetes in Male Rats. J. Adv. Vet. Anim. Res. 2020;7:177. doi: 10.5455/javar.2020.g408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Manzo A., Musso L., Panseri S., Iriti M., Dallavalle S., Catalano E., Scarì G., Giorgi A. Screening of the Chemical Composition and Bioactivity of Waldheimia glabra (Decne.) Regel Essential Oil. J. Sci. Food Agric. 2016;96:3195–3201. doi: 10.1002/jsfa.7499. [DOI] [PubMed] [Google Scholar]
- 260.Hemeg H.A., Moussa I.M., Ibrahim S., Dawoud T.M., Alhaji J.H., Mubarak A.S., Kabli S.A., Alsubki R.A., Tawfik A.M., Marouf S.A. Antimicrobial Effect of Different Herbal Plant Extracts against Different Microbial Population. Saudi J. Biol. Sci. 2020;27:3221–3227. doi: 10.1016/j.sjbs.2020.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Singh P., Singh R., Sati N., Sati O.P. Antioxidant, Antibacterial and Antifungal Activity of Impatiens sulcata Wallich in Roxb. Extracts. Int. J. Life Sci. Sci. Res. 2016;2:671–677. doi: 10.21276/ijlssr.2016.2.6.5. [DOI] [Google Scholar]
- 262.Gulfraz M., Mehmood S., Ahmad A., Fatima N., Praveen Z., Williamson E.M. Comparison of the Antidiabetic Activity of Berberis lyceum Root Extract and Berberine in Alloxan-induced Diabetic Rats. Phytother. Res. 2008;22:1208–1212. doi: 10.1002/ptr.2438. [DOI] [PubMed] [Google Scholar]
- 263.Oliveira-Costa J.F., Meira C.S., das Neves M.V.G., Dos Reis B.P.Z.C., Soares M.B.P. Anti-Inflammatory Activities of Betulinic Acid: A Review. Front. Pharmacol. 2022;13:883857. doi: 10.3389/fphar.2022.883857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Monsef-Esfahani H.R., Amini M., Goodarzi N., Saiedmohammadi F., Hajiaghaee R., Faramarzi M.A., Tofighi Z., Ghahremani M.H. Coumarin Compounds of Biebersteinia multifida Roots Show Potential Anxiolytic Effects in Mice. DARU J. Pharm. Sci. 2013;21:51. doi: 10.1186/2008-2231-21-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Parray J.A., Hamid R., Kamili A.N., Shameem N., Jan S., Ganai B.A. Biological Efficacy and Radical Scavenging Potential of Shikonin in Arnebia benthamii (Wall Ex. G Don) Johnston. Ind. Crops Prod. 2015;74:434–439. doi: 10.1016/j.indcrop.2015.04.040. [DOI] [Google Scholar]
- 266.Yadav S., Sharma A., Nayik G.A., Cooper R., Bhardwaj G., Sohal H.S., Mutreja V., Kaur R., Areche F.O., AlOudat M., et al. Review of Shikonin and Derivatives: Isolation, Chemistry, Biosynthesis, Pharmacology and Toxicology. Front. Pharmacol. 2022;13:905755. doi: 10.3389/fphar.2022.905755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Chouhan H.S., Singh S.K. Phytochemical Analysis, Antioxidant and Anti-Inflammatory Activities of Phyllanthus simplex. J. Ethnopharmacol. 2011;137:1337–1344. doi: 10.1016/j.jep.2011.07.069. [DOI] [PubMed] [Google Scholar]
- 268.Kumar N., Kumar R., Kishore K. Onosma L.: A Review of Phytochemistry and Ethnopharmacology. Pharmacogn. Rev. 2013;7:140. doi: 10.4103/0973-7847.120513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Mattioli R., Francioso A., d’Erme M., Trovato M., Mancini P., Piacentini L., Casale A., Wessjohann L., Gazzino R., Costantino P., et al. Anti-Inflammatory Activity of A Polyphenolic Extract from Arabidopsis thaliana in In Vitro and In Vivo Models of Alzheimer’s Disease. Int. J. Mol. Sci. 2019;20:708. doi: 10.3390/ijms20030708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Melrose J. The Glucosinolates: A Sulphur Glucoside Family of Mustard Anti-Tumour and Antimicrobial Phytochemicals of Potential Therapeutic Application. Biomedicines. 2019;7:62. doi: 10.3390/biomedicines7030062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Kim J.S., Han S., Kim H., Won S.Y., Park H.W., Choi H., Choi M., Lee M.Y., Ha I.J., Lee S.-G. Anticancer Effects of High Glucosinolate Synthesis Lines of Brassica rapa on Colorectal Cancer Cells. Antioxidants. 2022;11:2463. doi: 10.3390/antiox11122463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Hsieh P.-C., Kuo C.-Y., Lee Y.-H., Wu Y.-K., Yang M.-C., Tzeng I.-S., Lan C.-C. Therapeutic Effects and Mechanisms of Actions of Descurainia sophia. Int. J. Med. Sci. 2020;17:2163–2170. doi: 10.7150/ijms.47357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Kaur T., Hussain K., Koul S., Vishwakarma R., Vyas D. Evaluation of Nutritional and Antioxidant Status of Lepidium latifolium Linn.: A Novel Phytofood from Ladakh. PLoS ONE. 2013;8:e69112. doi: 10.1371/journal.pone.0069112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Bahramikia S., Yazdanparast R. Antioxidant Efficacy of Nasturtium officinale Extracts Using Various In Vitro Assay Systems. J. Acupunct. Meridian Stud. 2010;3:283–290. doi: 10.1016/S2005-2901(10)60049-0. [DOI] [PubMed] [Google Scholar]
- 275.Đulović A., Popović M., Burčul F., Čikeš Čulić V., Marijan S., Ruščić M., Anđelković N., Blažević I. Glucosinolates of Sisymbrium officinale and S. orientale. Molecules. 2022;27:8431. doi: 10.3390/molecules27238431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Farrukh M., Saleem U., Ahmad B., Chauhdary Z., Alsharif I., Manan M., Qasim M., Alhasani R.H., Shah G.M., Shah M.A. Sarcococca saligna Hydroalcoholic Extract Ameliorates Arthritis in Complete Freund’s Adjuvant-Induced Arthritic Rats via Modulation of Inflammatory Biomarkers and Suppression of Oxidative Stress Markers. ACS Omega. 2022;7:13164–13177. doi: 10.1021/acsomega.2c00619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Jan H.A., Hussain W., Bussmann R.W., Paniagua-Zambrana N.Y. Ethnobotany of the Himalayas. Ethnobotany of Mountain Regions. Springer; Cham, Switzerland: 2021. Codonopsis clematidea (Schrenk Ex Fisch. & C.A. Mey.) C.B. Clarke Campanulaceae; pp. 591–593. [DOI] [Google Scholar]
- 278.He J.-Y., Ma N., Zhu S., Komatsu K., Li Z.-Y., Fu W.-M. The Genus Codonopsis (Campanulaceae): A Review of Phytochemistry, Bioactivity and Quality Control. J. Nat. Med. 2015;69:1–21. doi: 10.1007/s11418-014-0861-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Huang S.K.-H., Bueno P.R.P., Garcia P.J.B., Lee M.-J., De Castro-Cruz K.A., Leron R.B., Tsai P.-W. Antioxidant, Anti-Inflammatory and Antiproliferative Effects of Osmanthus fragrans (Thunb.) Lour. Flower Extracts. Plants. 2023;12:3168. doi: 10.3390/plants12173168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Conforti F., Marcotullio M.C., Menichini F., Statti G.A., Vannutelli L., Burini G., Menichini F., Curini M. The Influence of Collection Zone on Glucosinolates, Polyphenols and Flavonoids Contents and Biological Profiles of Capparis sicula ssp. Sicula. Food Sci. Technol. Int. 2011;17:87–97. doi: 10.1177/1082013210368583. [DOI] [PubMed] [Google Scholar]
- 281.Hsu H.-F., Hsiao P.-C., Kuo T.-C., Chiang S.-T., Chen S.-L., Chiou S.-J., Ling X.-H., Liang M.-T., Cheng W.-Y., Houng J.-Y. Antioxidant and Anti-Inflammatory Activities of Lonicera japonica Thunb. Var. Sempervillosa hayata Flower Bud Extracts Prepared by Water, Ethanol and Supercritical Fluid Extraction Techniques. Ind. Crops Prod. 2016;89:543–549. doi: 10.1016/j.indcrop.2016.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Yuan Y., Wang Z., Jiang C., Wang X., Huang L. Exploiting Genes and Functional Diversity of Chlorogenic Acid and Luteolin Biosyntheses in Lonicera japonica and Their Substitutes. Gene. 2014;534:408–416. doi: 10.1016/j.gene.2012.09.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Sharifi-Rad J., Quispe C., Vergara C.V., Kitic D., Kostic M., Armstrong L., Shinwari Z.K., Khalil A.T., Brdar-Jokanović M., Ljevnaić-Mašić B., et al. Genus Viburnum: Therapeutic Potentialities and Agro-Food-Pharma Applications. Oxid. Med. Cell. Longev. 2021;2021:3095514. doi: 10.1155/2021/3095514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Belahcene S., Kebsa W., Akingbade T.V., Umar H.I., Omoboyowa D.A., Alshihri A.A., Abo Mansour A., Alhasaniah A.H., Oraig M.A., Bakkour Y., et al. Chemical Composition Antioxidant and Anti-Inflammatory Activities of Myrtus communis L. Leaf Extract: Forecasting ADMET Profiling and Anti-Inflammatory Targets Using Molecular Docking Tools. Molecules. 2024;29:849. doi: 10.3390/molecules29040849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Boukhira S., El Mansouri L., Bouarfa M., Ouhammou A., Achour S., Khadhr M., Bousta D. Phytochemical Screening, Anti-Inflammatory and Analgesic Activities Of Formulation Cream of Silene vulgaris. Res. J. Med. Plant. 2016;10:150–158. doi: 10.3923/rjmp.2016.150.158. [DOI] [Google Scholar]
- 286.Oladeji O.S., Oyebamiji A.K. Stellaria media (L.) Vill.-A Plant with Immense Therapeutic Potentials: Phytochemistry and Pharmacology. Heliyon. 2020;6:e04150. doi: 10.1016/j.heliyon.2020.e04150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Poonia A., Upadhayay A. Chenopodium album Linn: Review of Nutritive Value and Biological Properties. J. Food Sci. Technol. 2015;52:3977–3985. doi: 10.1007/s13197-014-1553-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Ajayi A., Tanayen J., Magomere A., Ezeonwumelu J. Antinociceptive and Anti-Inflammatory Effects of Aqueous Extract of Chenopodium opulifolium Schrad Leaves. J. Intercult. Ethnopharmacol. 2017;6:14. doi: 10.5455/jice.20161229055924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Salamatullah A.M. Convolvulus Arvensis: Antioxidant, Antibacterial, and Antifungal Properties of Chemically Profiled Essential Oils: An Approach against Nosocomial Infections. Life. 2022;12:2138. doi: 10.3390/life12122138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Kumar A., Kumar P., Koundal R., Agnihotri V.K. Antioxidant Properties and UPLC-MS/MS Profiling of Phenolics in Jacquemont’s Hazelnut Kernels (Corylus Jacquemontii) and Its Byproducts from Western Himalaya. J. Food Sci. Technol. 2016;53:3522–3531. doi: 10.1007/s13197-016-2329-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Bais S., Gill N.S., Rana N., Shandil S. A Phytopharmacological Review on a Medicinal Plant: Juniperus communis. Int. Sch. Res. Notices. 2014;2014:634723. doi: 10.1155/2014/634723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Miceli N., Trovato A., Dugo P., Cacciola F., Donato P., Marino A., Bellinghieri V., La Barbera T.M., Güvenç A., Taviano M.F. Comparative Analysis of Flavonoid Profile, Antioxidant and Antimicrobial Activity of the Berries of Juniperus communis L. Var. Communis and Juniperus communis L. Var. Saxatilis Pall. from Turkey. J. Agric. Food Chem. 2009;57:6570–6577. doi: 10.1021/jf9012295. [DOI] [PubMed] [Google Scholar]
- 293.Mishra S., Alhodieb F.S., Barkat M.A., Hassan M.Z., Barkat H.A., Ali R., Alam P., Alam O. Antitumor and Hepatoprotective Effect of Cuscuta reflexa Roxb. in a Murine Model of Colon Cancer. J. Ethnopharmacol. 2022;282:114597. doi: 10.1016/j.jep.2021.114597. [DOI] [PubMed] [Google Scholar]
- 294.Deveoglu O., Cakmakcı E., Taskopru T., Torgan E., Karadag R. Identification by RP-HPLC-DAD, FTIR, TGA and FESEM-EDAX of natural pigments prepared from Datisca cannabina L. Dye. Pigment. 2012;94:437–442. doi: 10.1016/j.dyepig.2012.02.002. [DOI] [Google Scholar]
- 295.Lee Y.-S., Chang Z.-Q., Oh B.-C., Park S.-C., Shin S.-R., Kim N.-W. Antioxidant Activity, Anti-Inflammatory Activity, and Whitening Effects of Extracts of Elaeagnus multiflora Thunb. J. Med. Food. 2007;10:126–133. doi: 10.1089/jmf.2006.145. [DOI] [PubMed] [Google Scholar]
- 296.Ji M., Gong X., Li X., Wang C., Li M. Advanced Research on the Antioxidant Activity and Mechanism of Polyphenols from Hippophae Species—A Review. Molecules. 2020;25:917. doi: 10.3390/molecules25040917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Goyal A.K., Basistha B.C., Sen A., Middha S.K. Antioxidant Profiling of Hippophae salicifolia Growing in Sacred Forests of Sikkim, India. Funct. Plant Biol. 2011;38:697. doi: 10.1071/FP11016. [DOI] [PubMed] [Google Scholar]
- 298.Rosero S., Del Pozo F., Simbaña W., Álvarez M., Quinteros M.F., Carrillo W., Morales D. Polyphenols and Flavonoids Composition, Anti-Inflammatory and Antioxidant Properties of Andean Baccharis macrantha Extracts. Plants. 2022;11:1555. doi: 10.3390/plants11121555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Baral B., Shrestha Vaidya G., Laxmi Maharjan B., Teixeira Da Silva J.A. Phytochemical And Antimicrobial Characterization Of Rhododendron anthopogon From High Nepalese Himalaya. Bot. Lith. 2015;20:142–152. doi: 10.2478/botlit-2014-0009. [DOI] [Google Scholar]
- 300.Ahmad A., Wali A.F., Rehman M.U., Khan A., Raish M., Kazi M., Alnemer O., Rao P.G.M. Therapeutic Potential of Rhododendron arboreum Polysaccharides in an Animal Model of Lipopolysaccharide-Inflicted Oxidative Stress and Systemic Inflammation. Molecules. 2020;25:6045. doi: 10.3390/molecules25246045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Zong L., Zhang J., Dai L., Liu J., Yang Y., Xie J., Luo X. The Anti-Inflammatory Properties of Rhododendron molle Leaf Extract in LPS-Induced RAW264.7. Chem. Biodivers. 2020;17:e2000477. doi: 10.1002/cbdv.202000477. [DOI] [PubMed] [Google Scholar]
- 302.Ghazanfar K., Mubashir K., Dar S.A., Nazir T., Hameed I., Ganai B.A., Akbar S., Masood A. Gentiana kurroo Royle Attenuates the Metabolic Aberrations in Diabetic Rats; Swertiamarin, Swertisin and Lupeol Being the Possible Bioactive Principles. J. Complement. Integr. Med. 2017;14 doi: 10.1515/jcim-2017-0002. [DOI] [PubMed] [Google Scholar]
- 303.Jaishree V., Badami S. Antioxidant and Hepatoprotective Effect of Swertiamarin from Enicostemma axillare against D-Galactosamine Induced Acute Liver Damage in Rats. J. Ethnopharmacol. 2010;130:103–106. doi: 10.1016/j.jep.2010.04.019. [DOI] [PubMed] [Google Scholar]
- 304.Mihailović V., Mihailović M., Uskoković A., Arambašić J., Mišić D., Stanković V., Katanić J., Mladenović M., Solujić S., Matić S. Hepatoprotective Effects of Gentiana asclepiadea L. Extracts against Carbon Tetrachloride Induced Liver Injury in Rats. Food Chem. Toxicol. 2013;52:83–90. doi: 10.1016/j.fct.2012.10.034. [DOI] [PubMed] [Google Scholar]
- 305.Pasdaran A., Naychov Z., Batovska D., Kerr P., Favre A., Dimitrov V., Aneva I., Hamedi A., Kozuharova E. Some European Gentiana Species Are Used Traditionally to Cure Wounds: Bioactivity and Conservation Issues. Diversity. 2023;15:467. doi: 10.3390/d15030467. [DOI] [Google Scholar]
- 306.Mirzaee F., Hosseini A., Jouybari H.B., Davoodi A., Azadbakht M. Medicinal, Biological and Phytochemical Properties of Gentiana Species. J. Tradit. Complement. Med. 2017;7:400–408. doi: 10.1016/j.jtcme.2016.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Nagalekshmi R., Menon A., Chandrasekharan D.K., Nair C.K.K. Hepatoprotective Activity of Andrographis paniculata and Swertia chirayita. Food Chem. Toxicol. 2011;49:3367–3373. doi: 10.1016/j.fct.2011.09.026. [DOI] [PubMed] [Google Scholar]
- 308.Kumar V., Van Staden J. A Review of Swertia chirayita (Gentianaceae) as a Traditional Medicinal Plant. Front. Pharmacol. 2016;6:308. doi: 10.3389/fphar.2015.00308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Velázquez-González C., Cariño-Cortés R., Gayosso de Lucio J.A., Ortiz M.I., De la O Arciniega M., Altamirano-Báez D.A., Ángeles L.J., Bautista-Ávila M. Antinociceptive and Anti-Inflammatory Activities of Geranium bellum and Its Isolated Compounds. BMC Complement. Altern. Med. 2014;14:506. doi: 10.1186/1472-6882-14-506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Jabeen Q., Haider S.I., Asif A., Rasheed R., Gul S., Arshad S. Geranium wallichianum D. Don Ex Sweet Ameliorates Rheumatoid Arthritis by Curtailing the Expression of COX-II and Inflammatory Cytokines as Well as by Alleviating the Oxidative Stress. Dose-Response. 2022;20:155932582211126. doi: 10.1177/15593258221112649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Vieira V., Pereira C., Pires T.C.S.P., Calhelha R.C., Alves M.J., Ferreira O., Barros L., Ferreira I.C.F.R. Phenolic Profile, Antioxidant and Antibacterial Properties of Juglans regia L. (Walnut) Leaves from the Northeast of Portugal. Ind. Crops Prod. 2019;134:347–355. doi: 10.1016/j.indcrop.2019.04.020. [DOI] [Google Scholar]
- 312.Czerwińska M., Świerczewska A., Granica S. Bioactive Constituents of Lamium album L. as Inhibitors of Cytokine Secretion in Human Neutrophils. Molecules. 2018;23:2770. doi: 10.3390/molecules23112770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Simirgiotis M.J., Burton D., Parra F., López J., Muñoz P., Escobar H., Parra C. Antioxidant and Antibacterial Capacities of Origanum vulgare L. Essential Oil from the Arid Andean Region of Chile and Its Chemical Characterization by GC-MS. Metabolites. 2020;10:414. doi: 10.3390/metabo10100414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Zhao M., Wu F., Tang Z., Yang X., Liu Y., Wang F., Chen B. Anti-Inflammatory and Antioxidant Activity of Ursolic Acid: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2023;14:1256946. doi: 10.3389/fphar.2023.1256946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Brindisi M., Bouzidi C., Frattaruolo L., Loizzo M.R., Cappello M.S., Dugay A., Deguin B., Lauria G., Cappello A.R., Tundis R. New Insights into the Antioxidant and Anti-Inflammatory Effects of Italian Salvia officinalis Leaf and Flower Extracts in Lipopolysaccharide and Tumor-Mediated Inflammation Models. Antioxidants. 2021;10:311. doi: 10.3390/antiox10020311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Zhang J., Wu C., Gao L., Du G., Qin X. Astragaloside IV Derived from Astragalus Membranaceus: A Research Review on the Pharmacological Effects. Adv. Pharmacol. 2020;87:89–112. doi: 10.1016/bs.apha.2019.08.002. [DOI] [PubMed] [Google Scholar]
- 317.Stępnik K., Kukula-Koch W., Plazinski W., Gawel K., Gaweł-Bęben K., Khurelbat D., Boguszewska-Czubara A. Significance of Astragaloside IV from the Roots of Astragalus mongholicus as an Acetylcholinesterase Inhibitor—From the Computational and Biomimetic Analyses to the In Vitro and In Vivo Studies of Safety. Int. J. Mol. Sci. 2023;24:9152. doi: 10.3390/ijms24119152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Liang Y., Chen B., Liang D., Quan X., Gu R., Meng Z., Gan H., Wu Z., Sun Y., Liu S., et al. Pharmacological Effects of Astragaloside IV: A Review. Molecules. 2023;28:6118. doi: 10.3390/molecules28166118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Gong G., Yu H., Zheng Y., Qi B., He H., Yin T., Dong T.T.X., Tsim K.W.K. Astragaloside IV, a Saponin from Astragalus membranaceus Var. Mongholicus, Induces Expressions of Heme Recycle Proteins via Signaling of Nrf2/ARE in Cultured Macrophages. J. Ethnopharmacol. 2021;265:113389. doi: 10.1016/j.jep.2020.113389. [DOI] [PubMed] [Google Scholar]
- 320.Zhang W.-D., Chen H., Zhang C., Liu R.-H., Li H.-L., Chen H.-Z. Astragaloside IV from Astragalus membranaceus Shows Cardioprotection during Myocardial Ischemia in Vivo and in Vitro. Planta Med. 2006;72:4–8. doi: 10.1055/s-2005-873126. [DOI] [PubMed] [Google Scholar]
- 321.Li L., Hou X., Xu R., Liu C., Tu M. Research Review on the Pharmacological Effects of Astragaloside. Fundam. Clin. Pharmacol. 2017;31:17–36. doi: 10.1111/fcp.12232. [DOI] [PubMed] [Google Scholar]
- 322.Agour A., Mssillou I., Es-safi I., Conte R., Mechchate H., Slighoua M., Amrati F.E.-Z., Parvez M.K., Numan O., Bari A., et al. The Antioxidant, Analgesic, Anti-Inflammatory, and Wound Healing Activities of Haplophyllum tuberculatum (Forsskal) A. Juss Aqueous and Ethanolic Extract. Life. 2022;12:1553. doi: 10.3390/life12101553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Zhi K.-K., Yang Z.-D., Shi D.-F., Yao X.-J., Wang M.-G. Desmodeleganine, a New Alkaloid from the Leaves of Desmodium elegans as a Potential Monoamine Oxidase Inhibitor. Fitoterapia. 2014;98:160–165. doi: 10.1016/j.fitote.2014.07.022. [DOI] [PubMed] [Google Scholar]
- 324.Baali N., Mezrag A., Bouheroum M., Benayache F., Benayache S., Souad A. Anti-Inflammatory and Antioxidant Effects of Lotus corniculatus on Paracetamol-Induced Hepatitis in Rats. Antiinflamm. Antiallergy Agents Med. Chem. 2020;19:128–139. doi: 10.2174/1871523018666190222120752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Quiñones-Muñoz T.A., Villanueva-Rodríguez S.J., Torruco-Uco J.G. Nutraceutical Properties of Medicago sativa L., Agave Spp., Zea Mays L. and Avena Sativa L.: A Review of Metabolites and Mechanisms. Metabolites. 2022;12:806. doi: 10.3390/metabo12090806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Mu H., Bai Y.-H., Wang S.-T., Zhu Z.-M., Zhang Y.-W. Research on Antioxidant Effects and Estrogenic Effect of Formononetin from Trifolium pratense (Red Clover) Phytomedicine. 2009;16:314–319. doi: 10.1016/j.phymed.2008.07.005. [DOI] [PubMed] [Google Scholar]
- 327.Ahmad S., Zeb A. Phytochemical Profile and Pharmacological Properties of Trifolium repens. J. Basic. Clin. Physiol. Pharmacol. 2021;32:20200015. doi: 10.1515/jbcpp-2020-0015. [DOI] [PubMed] [Google Scholar]
- 328.Zhou J., Chan L., Zhou S. Trigonelline: A Plant Alkaloid with Therapeutic Potential for Diabetes and Central Nervous System Disease. Curr. Med. Chem. 2012;19:3523–3531. doi: 10.2174/092986712801323171. [DOI] [PubMed] [Google Scholar]
- 329.Liu L.-F., Li W.-H., Li M.-Y., Wu X.-Z., Yang F., Xu J.-N., Yuan C.-S. Chemical Constituents from Common Vetch ( Vicia sativa L.) and Their Antioxidant and Cytotoxic Activities. Nat. Prod. Res. 2020;34:3205–3211. doi: 10.1080/14786419.2018.1560282. [DOI] [PubMed] [Google Scholar]
- 330.Passos F.R.S., Araújo-Filho H.G., Monteiro B.S., Shanmugam S., de Souza Araújo A.A., da Silva Almeida J.R., Thangaraj P., Júnior L.J.Q., Quintans J.D. Anti-Inflammatory and Modulatory Effects of Steroidal Saponins and Sapogenins on Cytokines: A Review of Pre-Clinical Research. Phytomedicine. 2022;96:153842. doi: 10.1016/j.phymed.2021.153842. [DOI] [PubMed] [Google Scholar]
- 331.de Oliveira Melo M.N., Oliveira A.P., Wiecikowski A.F., Carvalho R.S., de Lima Castro J., de Oliveira F.A.G., Pereira H.M.G., da Veiga V.F., Capella M.M.A., Rocha L., et al. Phenolic Compounds from Viscum album Tinctures Enhanced Antitumor Activity in Melanoma Murine Cancer Cells. Saudi Pharm. J. 2018;26:311–322. doi: 10.1016/j.jsps.2018.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Akkol E.K., Karpuz B., Türkcanoğlu G., Coşgunçelebi F.G., Taştan H., Aschner M., Khatkar A., Sobarzo-Sánchez E. The Phytochemical Profile and Biological Activity of Malva neglecta Wallr. in Surgically Induced Endometriosis Model in Rats. Molecules. 2022;27:7869. doi: 10.3390/molecules27227869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Altyar A.E., Munir A., Ishtiaq S., Rizwan M., Abbas K., Kensara O., Elhady S.S., Rizg W.Y., Youssef F.S., Ashour M.L. Malva parviflora Leaves and Fruits Mucilage as Natural Sources of Anti-Inflammatory, Antitussive and Gastro-Protective Agents: A Comparative Study Using Rat Models and Gas Chromatography. Pharmaceuticals. 2022;15:427. doi: 10.3390/ph15040427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 334.Suriyaprom S., Srisai P., Intachaisri V., Kaewkod T., Pekkoh J., Desvaux M., Tragoolpua Y. Antioxidant and Anti-Inflammatory Activity on LPS-Stimulated RAW 264.7 Macrophage Cells of White Mulberry (Morus alba L.) Leaf Extracts. Molecules. 2023;28:4395. doi: 10.3390/molecules28114395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 335.Alhakmani F., Kumar S., Khan S.A. Estimation of Total Phenolic Content, in-Vitro Antioxidant and Anti—Inflammatory Activity of Flowers of Moringa oleifera. Asian Pac. J. Trop. Biomed. 2013;3:623–627. doi: 10.1016/S2221-1691(13)60126-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 336.Chauhan R.S., Nautiyal M.C., Tava A., Cecotti R. Essential Oil Composition of Morina longifolia Wall. Ex DC. from the Himalayan Region. J. Essent. Oil Res. 2012;24:461–463. doi: 10.1080/10412905.2012.703500. [DOI] [Google Scholar]
- 337.El-Hawary S.S., EL-Hefnawy H.M., Osman S.M., El-Raey M.A., Mokhtar F.A., Ibrahim H.A. Antioxidant, Anti-Inflammatory and Cytotoxic Activities of Jasminum multiflorum (Burm. F.) Andrews Leaves towards MCF-7 Breast Cancer and HCT 116 Colorectal Cell Lines and Identification of Bioactive Metabolites. Anticancer. Agents Med. Chem. 2021;21:2572–2582. doi: 10.2174/1871520621666210901103440. [DOI] [PubMed] [Google Scholar]
- 338.Kiss A.K., Bazylko A., Filipek A., Granica S., Jaszewska E., Kiarszys U., Kośmider A., Piwowarski J. Oenothein B’s Contribution to the Anti-Inflammatory and Antioxidant Activity of Epilobium sp. Phytomedicine. 2011;18:557–560. doi: 10.1016/j.phymed.2010.10.016. [DOI] [PubMed] [Google Scholar]
- 339.Timoszuk M., Bielawska K., Skrzydlewska E. Evening Primrose (Oenothera biennis) Biological Activity Dependent on Chemical Composition. Antioxidants. 2018;7:108. doi: 10.3390/antiox7080108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Sharma S., Kumar V., Seth C.A., Sourirajan A., El-Shazly M., Dev K. A Comprehensive Review on the Phytochemistry, Pharmacological Properties, and in Vitro Propagation of an Endemic Medicinal Orchid, Dactylorhiza hatagirea. Naunyn Schmiedebergs Arch. Pharmacol. 2024;397:2621–2635. doi: 10.1007/s00210-023-02827-5. [DOI] [PubMed] [Google Scholar]
- 341.Ahmad M., Kaloo Z.A., Ganai B.A., Ganaie H.A., Singh S. Phytochemical Screening of Meconopsis aculeata Royle an Important Medicinal Plant of Kashmir Himalaya: A Perspective. Res. J. Phytochem. 2016;10:1–9. doi: 10.3923/rjphyto.2016.1.9. [DOI] [Google Scholar]
- 342.Jaydeokar A.V., Bandawane D.D., Bibave K.H., Patil T.V. Hepatoprotective Potential of Cassia auriculata Roots on Ethanol and Antitubercular Drug-Induced Hepatotoxicity in Experimental Models. Pharm. Biol. 2014;52:344–355. doi: 10.3109/13880209.2013.837075. [DOI] [PubMed] [Google Scholar]
- 343.Charalambous D., Eliades N.-G.H., Christoforou M., Kakouri E., Kanakis C., Tarantilis P.A., Pantelidou M. Chemical Characterization, Antioxidant and Antimicrobial Properties of Different Types of Tissue of Cedrus brevifolia Henry Extracts. Molecules. 2022;27:2717. doi: 10.3390/molecules27092717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Bhardwaj K., Sharma R., Cruz-Martins N., Valko M., Upadhyay N.K., Kuča K., Bhardwaj P. Studies of Phytochemicals, Antioxidant, and Antibacterial Activities of Pinus gerardiana and Pinus roxburghii Seed Extracts. BioMed Res. Int. 2022;2022:5938610. doi: 10.1155/2022/5938610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Salehi B., Upadhyay S., Erdogan Orhan I., Kumar Jugran A., Jayaweera S.L.D., Dias D.A., Sharopov F., Taheri Y., Martins N., Baghalpour N., et al. Therapeutic Potential of α- and β-Pinene: A Miracle Gift of Nature. Biomolecules. 2019;9:738. doi: 10.3390/biom9110738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Zhakipbekov K., Turgumbayeva A., Issayeva R., Kipchakbayeva A., Kadyrbayeva G., Tleubayeva M., Akhayeva T., Tastambek K., Sainova G., Serikbayeva E., et al. Antimicrobial and Other Biomedical Properties of Extracts from Plantago major, Plantaginaceae. Pharmaceuticals. 2023;16:1092. doi: 10.3390/ph16081092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Samuelsen A.B. The Traditional Uses, Chemical Constituents and Biological Activities of Plantago major L. A Review. J. Ethnopharmacol. 2000;71:1–21. doi: 10.1016/S0378-8741(00)00212-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Pawłowska K.A., Hałasa R., Dudek M.K., Majdan M., Jankowska K., Granica S. Antibacterial and Anti-Inflammatory Activity of Bistort (Bistorta officinalis) Aqueous Extract and Its Major Components. Justification of the Usage of the Medicinal Plant Material as a Traditional Topical Agent. J. Ethnopharmacol. 2020;260:113077. doi: 10.1016/j.jep.2020.113077. [DOI] [PubMed] [Google Scholar]
- 349.Manivannan V., Johnson M. Total Phenolic, Tannin, Triterpenoid, Flavonoid and Sterol Contents, Anti-Diabetic, Anti-Inflammatory and Cytotoxic Activities of Tectaria paradoxa (Fee.) Sledge. Toxicol. Rep. 2020;7:1465–1468. doi: 10.1016/j.toxrep.2020.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 350.Nakamura T., Kodama N., Arai Y., Kumamoto T., Higuchi Y., Chaichantipyuth C., Ishikawa T., Ueno K., Yano S. Inhibitory Effect of Oxycoumarins Isolated from the Thai Medicinal Plant Clausena guillauminii on the Inflammation Mediators, INOS, TNF-α, and COX-2 Expression in Mouse Macrophage RAW 264.7. J. Nat. Med. 2009;63:21–27. doi: 10.1007/s11418-008-0277-5. [DOI] [PubMed] [Google Scholar]
- 351.Shanmugasundaram D., Roza J.M. Assessment of Anti-Inflammatory and Antioxidant Activity of Quercetin—Rutin Blend (SophorOxTM)—An Invitro Cell Based Assay. J. Complement. Integr. Med. 2022;19:637–644. doi: 10.1515/jcim-2021-0568. [DOI] [PubMed] [Google Scholar]
- 352.Granica S., Czerwińska M.E., Żyżyńska-Granica B., Kiss A.K. Antioxidant and Anti-Inflammatory Flavonol Glucuronides from Polygonum aviculare L. Fitoterapia. 2013;91:180–188. doi: 10.1016/j.fitote.2013.08.026. [DOI] [PubMed] [Google Scholar]
- 353.Wang K.-J., Zhang Y.-J., Yang C.-R. Antioxidant Phenolic Compounds from Rhizomes of Polygonum paleaceum. J. Ethnopharmacol. 2005;96:483–487. doi: 10.1016/j.jep.2004.09.036. [DOI] [PubMed] [Google Scholar]
- 354.Pukhrambam P.D., Devi K.K., Maibam C., Mutum R.D., Devi M.L., Das S. Phenolics and Flavonoids from Polygonum posumbu and Comparision of Flavonoid Compounds Content in Different Tissues (Leaves, Stems and Roots) Fitoterapia. 2024;174:105864. doi: 10.1016/j.fitote.2024.105864. [DOI] [PubMed] [Google Scholar]
- 355.Pandith S.A., Dar R.A., Lattoo S.K., Shah M.A., Reshi Z.A. Rheum. australe, an Endangered High-Value Medicinal Herb of North Western Himalayas: A Review of Its Botany, Ethnomedical Uses, Phytochemistry and Pharmacology. Phytochem. Rev. 2018;17:573–609. doi: 10.1007/s11101-018-9551-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 356.Zhumashova G., Kukula-Koch W., Koch W., Baj T., Sayakova G., Shukirbekova A., Głowniak K., Sakipova Z. Phytochemical and Antioxidant Studies on a Rare Rheum. cordatum Losinsk. Species from Kazakhstan. Oxid. Med. Cell. Longev. 2019;2019:5465463. doi: 10.1155/2019/5465463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 357.Khaliq T., Akhter S., Sultan P., Hassan Q.P. Critical Review on Rumex dentatus L. a Strong Pharmacophore and the Future Medicine: Pharmacology, Phytochemical Analysis and Traditional Uses. Heliyon. 2023;9:e14159. doi: 10.1016/j.heliyon.2023.e14159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 358.Ahmad S., Ullah F., Ayaz M., Sadiq A., Imran M. Antioxidant and Anticholinesterase Investigations of Rumex hastatus D. Don: Potential Effectiveness in Oxidative Stress and Neurological Disorders. Biol. Res. 2015;48:20. doi: 10.1186/s40659-015-0010-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Demirezer L.Ö., Kuruüzüm-Uz A., Bergere I., Schiewe H.-J., Zeeck A. The Structures of Antioxidant and Cytotoxic Agents from Natural Source: Anthraquinones and Tannins from Roots of Rumex patientia. Phytochemistry. 2001;58:1213–1217. doi: 10.1016/S0031-9422(01)00337-5. [DOI] [PubMed] [Google Scholar]
- 360.Wani T.A., Kaloo Z.A., Dangroo N.A. Aconitum heterophyllum Wall. Ex Royle: A Critically Endangered Medicinal Herb with Rich Potential for Use in Medicine. J. Integr. Med. 2022;20:104–113. doi: 10.1016/j.joim.2021.12.004. [DOI] [PubMed] [Google Scholar]
- 361.Zhang L., Siyiti M., Zhang J., Yao M., Zhao F. Anti-inflammatory and Anti-rheumatic Activities in Vitro of Alkaloids Separated from Aconitum soongoricum Stapf. Exp. Ther. Med. 2021;21:493. doi: 10.3892/etm.2021.9924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 362.Safdar A., Bibi Y. Medicinal Uses and Conservation Status of Aconitum violaceum. J. Plant Environ. 2020;2:19–23. doi: 10.33687/jpe.002.01.3475. [DOI] [Google Scholar]
- 363.Wani Z.A., Pant S. Aconitum heterophyllum Wall. Ex Royle: An Endemic, Highly Medicinal and Critically Endangered Plant Species of Northwestern Himalaya in Peril. Curr. Tradit. Med. 2021;7:2–7. doi: 10.2174/2215083807666210924162204. [DOI] [Google Scholar]
- 364.Anh Minh C.T., Khoi N.M., Thuong P.T., Hwang I.H., Kim D.W., Na M. A New Saponin and Other Constituents from Anemone rivularis Buch.-Ham. Biochem. Syst. Ecol. 2012;44:270–274. doi: 10.1016/j.bse.2012.03.017. [DOI] [Google Scholar]
- 365.Shrivastava A.K., Keshari M., Neupane M., Chaudhary S., Dhakal P.K., Shrestha L., Palikhey A., Yadav C.K., Lamichhane G., Shekh M.U., et al. Evaluation of Antioxidant and Anti-Inflammatory Activities, and Metabolite Profiling of Selected Medicinal Plants of Nepal. J. Trop. Med. 2023;2023:6641018. doi: 10.1155/2023/6641018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Jan H., Usman H., Shah M., Zaman G., Mushtaq S., Drouet S., Hano C., Abbasi B.H. Phytochemical Analysis and Versatile in Vitro Evaluation of Antimicrobial, Cytotoxic and Enzyme Inhibition Potential of Different Extracts of Traditionally Used Aquilegia pubiflora Wall. Ex Royle. BMC Complement. Med. Ther. 2021;21:165. doi: 10.1186/s12906-021-03333-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 367.Mubashir S., Dar M.Y., Lone B.A., Zargar M.I., Shah W.A. Anthelmintic, Antimicrobial, Antioxidant and Cytotoxic Activity of Caltha palustris Var. Alba Kashmir, India. Chin. J. Nat. Med. 2014;12:567–572. doi: 10.1016/S1875-5364(14)60087-X. [DOI] [PubMed] [Google Scholar]
- 368.Mostafa M., Appidi J.R., Yakubu M.T., Afolayan A.J. Anti-Inflammatory, Antinociceptive and Antipyretic Properties of the Aqueous Extract of Clematis brachiata Leaf in Male Rats. Pharm. Biol. 2010;48:682–689. doi: 10.3109/13880200903257966. [DOI] [PubMed] [Google Scholar]
- 369.Yesilada E., Küpeli E. Clematis vitalba L. Aerial Part Exhibits Potent Anti-Inflammatory, Antinociceptive and Antipyretic Effects. J. Ethnopharmacol. 2007;110:504–515. doi: 10.1016/j.jep.2006.10.016. [DOI] [PubMed] [Google Scholar]
- 370.Hasan U.H., Alamgeer. Shahzad M., Jahan S., Niazi Z.R., Bukhari I.A., Assiri A.M., Riaz H. Inhibitory Effects of Clematis orientalis Aqueous Ethanol Extract and Fractions on Inflammatory Markers in Complete Freund’s Adjuvant-Induced Arthritis in Sprague—Dawley Rats. Inflammopharmacology. 2019;27:781–797. doi: 10.1007/s10787-018-0543-4. [DOI] [PubMed] [Google Scholar]
- 371.Khan J., Deb P.K., Priya S., Medina K.D., Devi R., Walode S.G., Rudrapal M. Dietary Flavonoids: Cardioprotective Potential with Antioxidant Effects and Their Pharmacokinetic, Toxicological and Therapeutic Concerns. Molecules. 2021;26:4021. doi: 10.3390/molecules26134021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 372.Bahukhandi A., Barola A., Sekar K.C. Antioxidant Activity and Polyphenolics of Fragaria nubicola: A Wild Edible Fruit Species of Himalaya. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2020;90:761–767. doi: 10.1007/s40011-019-01142-5. [DOI] [Google Scholar]
- 373.Orlova A., Kysil E., Tsvetkova E., Meshalkina D., Whaley A., Whaley A.O., Laub A., Francioso A., Babich O., Wessjohann L.A., et al. Phytochemical Characterization of Water Avens (Geum rivale L.) Extracts: Structure Assignment and Biological Activity of the Major Phenolic Constituents. Plants. 2022;11:2859. doi: 10.3390/plants11212859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 374.Tangpu V., Deori K., Yadav A. Evaluation of Safety and Protective Effects of Potentilla fulgens Root Extract in Experimentally Induced Diarrhoea in Mice. J. Intercult. Ethnopharmacol. 2014;3:103. doi: 10.5455/jice.20140416104844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 375.Wölfle U., Hoffmann J., Haarhaus B., Rao Mittapalli V., Schempp C.M. Anti-Inflammatory and Vasoconstrictive Properties of Potentilla erecta—A Traditional Medicinal Plant from the Northern Hemisphere. J. Ethnopharmacol. 2017;204:86–94. doi: 10.1016/j.jep.2017.03.058. [DOI] [PubMed] [Google Scholar]
- 376.Radhika M., Ghoshal N., Chatterjee A. Comparison of Effectiveness in Antitumor Activity between Flavonoids and Polyphenols of the Methanolic Extract of Roots of Potentilla fulgens in Breast Cancer Cells. J. Complement. Integr. Med. 2012;9 doi: 10.1515/1553-3840.1644. [DOI] [PubMed] [Google Scholar]
- 377.Mashaal K., Shabbir A., Shahzad M., Mobashar A., Akhtar T., Fatima T., Riaz B., Alharbi R., Fatima A., Alanezi A.A., et al. Amelioration of Rheumatoid Arthritis by Fragaria nubicola (Wild Strawberry) via Attenuation of Inflammatory Mediators in Sprague Dawley Rats. Medicina. 2023;59:1917. doi: 10.3390/medicina59111917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 378.Bagale R., Acharya S., Gupta A., Chaudhary P., Chaudhary G.P., Pandey J. Antibacterial and Antioxidant Activities of Prinsepia utilis Royle Leaf and Seed Extracts. J. Trop. Med. 2022;2022:3898939. doi: 10.1155/2022/3898939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Tewari D., Bawari S., Sah A.N., Sharma H., Joshi B.C., Gupta P., Sharma V.K. Himalayan Pyracantha crenulata (D.Don) M.Roem. Leaf and Fruit Extracts Alleviate Algesia through COX-2 and Mu-Opioid Receptor Mediated Pathways. J. Ethnopharmacol. 2024;318:117004. doi: 10.1016/j.jep.2023.117004. [DOI] [PubMed] [Google Scholar]
- 380.Gogoi N., Gogoi A., Neog B., Baruah D., Singh K. Evaluation of Antioxidant and Hepatoprotective Activity of Fruit Rind Extract of Garcinia dulcis (Roxburgh) Kurz. Pharmacogn. Res. 2017;9:266. doi: 10.4103/0974-8490.210330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Ishaque M., BiBi Y., Valant-Vetschera K.M., Schinnerl J., Bacher M. Fruits of Rosa brunonii—A Source of Antioxidant Phenolic Compounds. Nat. Prod. Commun. 2017;12:1934578X1701201. doi: 10.1177/1934578X1701201106. [DOI] [Google Scholar]
- 382.Abla M., Cai Y., Gao L., Wu J., Yang L. Changes in the Antioxidant and Anti-Inflammatory Activities of Rosa rugosa ‘Mohong’ during Fermentation. Heliyon. 2024;10:e25982. doi: 10.1016/j.heliyon.2024.e25982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Dhatwalia J., Kumari A., Chauhan A., Mansi K., Thakur S., Saini R.V., Guleria I., Lal S., Kumar A., Batoo K.M., et al. Rubus ellipticus Sm. Fruit Extract Mediated Zinc Oxide Nanoparticles: A Green Approach for Dye Degradation and Biomedical Applications. Materials. 2022;15:3470. doi: 10.3390/ma15103470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Krauze-Baranowska M., Majdan M., Hałasa R., Głód D., Kula M., Fecka I., Orzeł A. The Antimicrobial Activity of Fruits from Some Cultivar Varieties of Rubus idaeus and Rubus occidentalis. Food Funct. 2014;5:2536–2541. doi: 10.1039/C4FO00129J. [DOI] [PubMed] [Google Scholar]
- 385.Kostikova V.A., Petrova N.V. Phytoconstituents and Bioactivity of Plants of the Genus Spiraea L. (Rosaceae): A Review. Int. J. Mol. Sci. 2021;22:11163. doi: 10.3390/ijms222011163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 386.Chandrashekar B., Prabhakara S., Mohan T., Shabeer D., Bhandare B., Nalini M., Sharmila P., Meghana D., Reddy B., Hanumantha Rao H., et al. Characterization of Rubia cordifolia L. Root Extract and Its Evaluation of Cardioprotective Effect in Wistar Rat Model. Indian J. Pharmacol. 2018;50:12. doi: 10.4103/ijp.IJP_418_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Viljoen A., Mncwangi N., Vermaak I. Anti-Inflammatory Iridoids of Botanical Origin. Curr. Med. Chem. 2012;19:2104–2127. doi: 10.2174/092986712800229005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Zhu W., Pang M., Dong L., Huang X., Wang S., Zhou L. Anti-Inflammatory and Immunomodulatory Effects of Iridoid Glycosides from Paederia scandens (LOUR.) MERRILL (Rubiaceae) on Uric Acid Nephropathy Rats. Life Sci. 2012;91:369–376. doi: 10.1016/j.lfs.2012.08.013. [DOI] [PubMed] [Google Scholar]
- 389.Ganeshkumar Y., Ramarao A., Veeresham C. Picroside I and Picroside II from Tissue Cultures of Picrorhiza kurroa. Pharmacognosy Res. 2017;9:53. doi: 10.4103/pr.pr_89_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Zhang J.-H., Shangguan Z.-S., Chen C., Zhang H.-J., Lin Y. Anti-Inflammatory Effects of Guggulsterone on Murine Macrophage by Inhibiting LPS-Induced Inflammatory Cytokines in NF-κB Signaling Pathway. Drug Des. Devel Ther. 2016;10:1829–1835. doi: 10.2147/DDDT.S104602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 391.Guzzo F., Russo R., Sanna C., Celaj O., Caredda A., Corona A., Tramontano E., Fiorentino A., Esposito F., D’Abrosca B. Chemical Characterization and Anti-HIV-1 Activity Assessment of Iridoids and Flavonols from Scrophularia trifoliata. Molecules. 2021;26:4777. doi: 10.3390/molecules26164777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Taheri Y., Quispe C., Herrera-Bravo J., Sharifi-Rad J., Ezzat S.M., Merghany R.M., Shaheen S., Azmi L., Prakash Mishra A., Sener B., et al. Urtica dioica-Derived Phytochemicals for Pharmacological and Therapeutic Applications. Evid.-Based Complement. Altern. Med. 2022;2022:4024331. doi: 10.1155/2022/4024331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 393.Raina R., Gautam K. Conservation and Utilization of Threatened Medicinal Plants. Springer International Publishing; Cham, Switzerland: 2020. Conservation and Utilization of High-Altitude Threatened Medicinal Plants; pp. 369–387. [DOI] [Google Scholar]
- 394.Giordano D. Bioactive Molecules from Extreme Environments. Mar. Drugs. 2020;18:640. doi: 10.3390/md18120640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.Alonso-Amelot M.E. High Altitude Plants, Chemistry of Acclimation and Adaptation. Stud. Nat. Prod. Chem. 2008;34:883–982. doi: 10.1016/S1572-5995(08)80036-1. [DOI] [Google Scholar]
- 396.Zhang X., Wang Y., Wang J., Yu M., Zhang R., Mi Y., Xu J., Jiang R., Gao J. Elevation Influences Belowground Biomass Proportion in Forests by Affecting Climatic Factors, Soil Nutrients and Key Leaf Traits. Plants. 2024;13:674. doi: 10.3390/plants13050674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 397.Sekar K.C., Thapliyal N., Pandey A., Joshi B., Mukherjee S., Bhojak P., Bisht M., Bhatt D., Singh S., Bahukhandi A. Plant Species Diversity and Density Patterns along Altitude Gradient Covering High-Altitude Alpine Regions of West Himalaya, India. Geol. Ecol. Landsc. 2023:1–15. doi: 10.1080/24749508.2022.2163606. [DOI] [Google Scholar]
- 398.Rathore N., Thakur D., Chawla A. Seasonal Variations Coupled with Elevation Gradient Drives Significant Changes in Eco-Physiological and Biogeochemical Traits of a High Altitude Evergreen Broadleaf Shrub, Rhododendron anthopogon. Plant Physiol. Biochem. 2018;132:708–719. doi: 10.1016/j.plaphy.2018.08.009. [DOI] [PubMed] [Google Scholar]
- 399.Arjona-García C., Blancas J., Beltrán-Rodríguez L., López Binnqüist C., Colín Bahena H., Moreno-Calles A.I., Sierra-Huelsz J.A., López-Medellín X. How Does Urbanization Affect Perceptions and Traditional Knowledge of Medicinal Plants? J. Ethnobiol. Ethnomed. 2021;17:48. doi: 10.1186/s13002-021-00473-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Roufogalis B.D. Challenges in Integrating Herbal Medicine in Healthcare Systems. Focus. Altern. Complement. Ther. 2015;20:34–35. doi: 10.1111/fct.12165. [DOI] [Google Scholar]
- 401.Ekor M. The Growing Use of Herbal Medicines: Issues Relating to Adverse Reactions and Challenges in Monitoring Safety. Front. Pharmacol. 2014;4:177. doi: 10.3389/fphar.2013.00177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Sharma S. Current Status of Herbal Product: Regulatory Overview. J. Pharm. Bioallied Sci. 2015;7:293. doi: 10.4103/0975-7406.168030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 403.Smith A., Jogalekar S., Gibson A. Regulation of Natural Health Products in Canada. J. Ethnopharmacol. 2014;158:507–510. doi: 10.1016/j.jep.2014.06.037. [DOI] [PubMed] [Google Scholar]
- 404.Harnett J., McIntyre E., Adams J., Addison T., Bannerman H., Egelton L., Ma J., Zabakly L., Steel A. Prevalence and Characteristics of Australians’ Complementary Medicine Product Use, and Concurrent Use with Prescription and Over-the-Counter Medications—A Cross Sectional Study. Nutrients. 2023;15:327. doi: 10.3390/nu15020327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 405.Sahoo N., Manchikanti P. Herbal Drug Regulation and Commercialization: An Indian Industry Perspective. J. Altern. Complement. Med. 2013;19:957–963. doi: 10.1089/acm.2012.0275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Pandey K.B., Rizvi S.I. Plant Polyphenols as Dietary Antioxidants in Human Health and Disease. Oxid. Med. Cell. Longev. 2009;2:270–278. doi: 10.4161/oxim.2.5.9498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Lavecchia T., Rea G., Antonacci A., Giardi M.T. Healthy and Adverse Effects of Plant-Derived Functional Metabolites: The Need of Revealing Their Content and Bioactivity in a Complex Food Matrix. Crit. Rev. Food Sci. Nutr. 2013;53:198–213. doi: 10.1080/10408398.2010.520829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Shahidi F., Ambigaipalan P. Phenolics and Polyphenolics in Foods, Beverages and Spices: Antioxidant Activity and Health Effects—A Review. J. Funct. Foods. 2015;18:820–897. doi: 10.1016/j.jff.2015.06.018. [DOI] [Google Scholar]
- 409.Sharifi-Rad M., Anil Kumar N.V., Zucca P., Varoni E.M., Dini L., Panzarini E., Rajkovic J., Tsouh Fokou P.V., Azzini E., Peluso I., et al. Lifestyle, Oxidative Stress, and Antioxidants: Back and Forth in the Pathophysiology of Chronic Diseases. Front. Physiol. 2020;11:694. doi: 10.3389/fphys.2020.00694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.Gligorijević N., Stanić-Vučinić D., Radomirović M., Stojadinović M., Khulal U., Nedić O., Ćirković Veličković T. Role of Resveratrol in Prevention and Control of Cardiovascular Disorders and Cardiovascular Complications Related to COVID-19 Disease: Mode of Action and Approaches Explored to Increase Its Bioavailability. Molecules. 2021;26:2834. doi: 10.3390/molecules26102834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 411.Ouyang J., Zhu K., Liu Z., Huang J. Prooxidant Effects of Epigallocatechin-3-Gallate in Health Benefits and Potential Adverse Effect. Oxid. Med. Cell. Longev. 2020;2020:9723686. doi: 10.1155/2020/9723686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.Chauhan D., Yadav P.K., Sultana N., Agarwal A., Verma S., Chourasia M.K., Gayen J.R. Advancements in Nanotechnology for the Delivery of Phytochemicals. J. Integr. Med. 2024:385–398. doi: 10.1016/j.joim.2024.04.005. [DOI] [PubMed] [Google Scholar]
- 413.Lu H., Zhang S., Wang J., Chen Q. A Review on Polymer and Lipid-Based Nanocarriers and Its Application to Nano-Pharmaceutical and Food-Based Systems. Front. Nutr. 2021;8:783831. doi: 10.3389/fnut.2021.783831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.El-Hefny M., Mohamed A.A., Abdelkhalek A., Salem M.Z.M. Productivity and Phytochemicals of Asclepias curassavica in Response to Compost and Silver Nanoparticles Application: HPLC Analysis and Antibacterial Activity of Extracts. Plants. 2023;12:2274. doi: 10.3390/plants12122274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Ashaolu T.J. Nanoemulsions for Health, Food, and Cosmetics: A Review. Environ. Chem. Lett. 2021;19:3381–3395. doi: 10.1007/s10311-021-01216-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Martínez-Ballesta M., Gil-Izquierdo Á., García-Viguera C., Domínguez-Perles R. Nanoparticles and Controlled Delivery for Bioactive Compounds: Outlining Challenges for New “Smart-Foods” for Health. Foods. 2018;7:72. doi: 10.3390/foods7050072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 417.Parveen S., Gupta P., Kumar S., Banerjee M. Lipid Polymer Hybrid Nanoparticles as Potent Vehicles for Drug Delivery in Cancer Therapeutics. Med. Drug Discov. 2023;20:100165. doi: 10.1016/j.medidd.2023.100165. [DOI] [Google Scholar]
- 418.Marisa Ribeiro A., Estevinho B.N., Rocha F. Microencapsulation of Polyphenols—The Specific Case of the Microencapsulation of Sambucus nigra L. Extracts—A Review. Trends Food Sci. Technol. 2020;105:454–467. doi: 10.1016/j.tifs.2019.03.011. [DOI] [Google Scholar]
- 419.Thang N.H., Chien T.B., Cuong D.X. Polymer-Based Hydrogels Applied in Drug Delivery: An Overview. Gels. 2023;9:523. doi: 10.3390/gels9070523. [DOI] [PMC free article] [PubMed] [Google Scholar]