Abstract
Craniofacial microsomia (CFM; also known as Goldenhar syndrome), is a craniofacial developmental disorder of variable expressivity and severity with a recognizable set of abnormalities. These birth defects are associated with structures derived from the first and second pharyngeal arches, can occur unilaterally and include ear dysplasia, microtia, preauricular tags and pits, facial asymmetry and other malformations. The inheritance pattern is controversial, and the molecular etiology of this syndrome is largely unknown. A total of 670 patients belonging to unrelated pedigrees with European and Chinese ancestry with CFM, are investigated. We identify 18 likely pathogenic variants in 21 probands (3.1%) in FOXI3. Biochemical experiments on transcriptional activity and subcellular localization of the likely pathogenic FOXI3 variants, and knock-in mouse studies strongly support the involvement of FOXI3 in CFM. Our findings indicate autosomal dominant inheritance with reduced penetrance, and/or autosomal recessive inheritance. The phenotypic expression of the FOXI3 variants is variable. The penetrance of the likely pathogenic variants in the seemingly dominant form is reduced, since a considerable number of such variants in affected individuals were inherited from non-affected parents. Here we provide suggestive evidence that common variation in the FOXI3 allele in trans with the pathogenic variant could modify the phenotypic severity and accounts for the incomplete penetrance.
Subject terms: Genetics, Molecular medicine, Development
Most cases of craniofacial microsomia are sporadic but familial cases have been reported. Here the authors report that variants in FOXI3 can cause a small fraction of cases with different modes of inheritance including autosomal dominant with reduced penetrance.
Introduction
In 1952, Maurice Goldenhar in Geneva described a disorder of craniofacial morphogenesis known as Goldenhar syndrome (OMIM 164210)1,2. The syndrome is also known as craniofacial microsomia (CFM) and other names such as Oculo-Auriculo-Vertebral spectrum3 or hemifacial microsomia. The craniofacial anomalies include ear dysplasia, microtia, and preauricular tags, and originate from the first and second pharyngeal arch-derived tissues (a comprehensive phenotypic description is given here4). Most cases are sporadic; however, reports of familial cases suggest either an autosomal dominant (AD) or autosomal recessive (AR) mode of inheritance4. The incidence of CFM ranges from 1:3500 to 1:5600 live births5. A pharmacological phenocopy of microtia is known to be caused by isotretinoin exposure during pregnancy6. In addition, there is a recent hypothesis that cases of malformation syndromes (including CFM) termed “recurrent constellations of embryonic malformations” (RCEM) may also be caused by environmental factors7.
A twin study has provided support for a genetic etiology8; 60% concordance in monozygotic twins, but only 5% in dizygotic twins. Various chromosomal abnormalities and structural variants have been described in some sporadic cases of CFM4. In addition, variants in genes MYT1, VWA1, SF3B2, and some others have been proposed as causative9–14. SF3B2 is the most common causative gene to date, haploinsufficiency of which accounts for ~3% of sporadic CFM14. However, the molecular basis for the majority of CFM remains elusive. Genome-wide association studies on CFM have identified 15 predisposing genomic signals15,16, explaining 8% of the phenotypic variation. Recently, one of the GWAS signals, a ∼10-kb interval in the intergenic region between ROBO1 and ROBO2 genes has been strongly associated with an increased risk of microtia in Amerindigenous populations. The risk locus contains a polymorphic complex repeat element that is expanded in affected individuals17.
Here we show that FOXI3 pathogenic alleles cause a small but significant fraction of CFM, particularly that with types II and III microtia or ear deformity and preauricular tags. Mouse and cellular models support this contention. The mode of inheritance of FOXI3-related CFM can be either AD with reduced penetrance or AR. Further, we provide evidence to support the hypothesis that a combination of rare pathogenic FOXI3 alleles in trans with a common FOXI3 haplotype could explain both the penetrance and the variable expressivity of CFM in a compound inheritance gene dosage model.
Results
FOXI3 likely pathogenic variants in a Pakistani CFM family
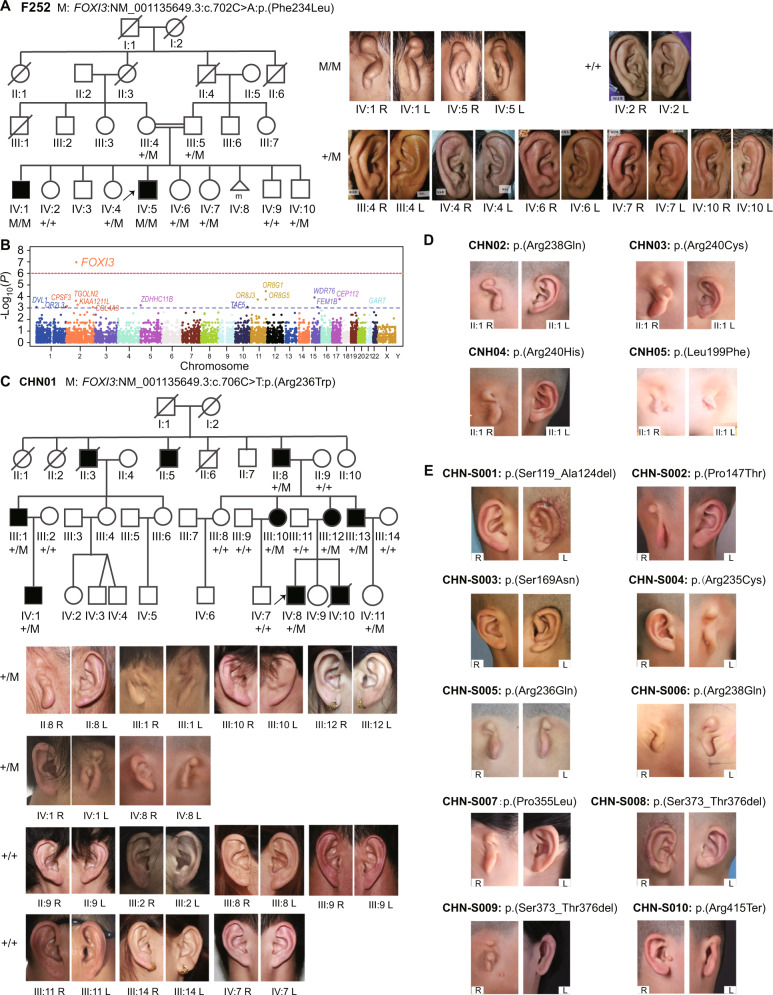
As part of an autozygosity project18, a Pakistani family F252 was ascertained with two affected siblings with type III microtia and deafness; the parents were first cousins (Fig. 1A and Supplementary Table 1). Whole exome sequencing (WES) identified a homozygous FOXI3 variant (NM_001135649.3):c.702C>A p.(Phe234Leu) as likely causative (Supplementary Fig. 1A). The unaffected parents, and 4 of 6 non-affected siblings, were heterozygous carriers. This family provided evidence for an AR inheritance for this form of CFM.
Fig. 1. Identification of FOXI3 as the pathogenic gene for CFM.
A The Pakistani consanguineous pedigree F252 with the likely pathogenic FOXI3: p.(Phe234Leu) variant. Photographs of the microtia type III of both ears of affected individuals IV:1 and IV:5. The bottom panel depicts the ears of additional members of the F252 family. The Sanger sequences are shown in Supplementary Fig. 1. M depicts the mutant allele, and + the normal allele. B Family-based gene-level association on rare LoF (Loss-of-Function) variants from 48 CFM Chinese families. The blue dashed line indicates the significance threshold of 1E-03, and the red dashed line indicates the Bonferroni correction threshold of 1.03E-06. Source data are provided as a Source Data file. C The pedigree, genotype, and external ear phenotype for the Chinese CFM family CHN01. M depicts the mutant allele, and + indicates the normal allele. D The genotype and external auricular phenotype for the four Chinese CFM families. E The genotype and external auricular phenotype of the sporadic Chinese patients.
FOXI3 likely pathogenic variants in Chinese CFM families
The laboratory in Beihang University, independently recruited 159 individuals in 48 Chinese families with CFM and performed WES on all of them; clinical phenotypes are summarized in Supplementary Table 1 and Supplementary Table 2. Family-based gene burden tests on the rare non-synonymous and Loss-of-Function (LoF) variants found the FOXI3 gene was the only genome-wide significant one (P = 1.08E-07) (Fig. 1B and Supplementary Table 3). The largest Chinese CFM family CHN01 shows an AD inheritance pattern with a deleterious heterozygous variant of FOXI3:c.706C>T p.(Arg236Trp) co-segregating with CFM (Fig. 1C and Supplementary Fig. 1B). The phenotypes vary greatly, and range from an absence of the posterior crus of the antihelix to bilateral type III microtia and micrognathia in CHN01 (Fig. 1C and Supplementary Table 1).
There were additional FOXI3 likely pathogenic variants in 4 of the 48 Chinese CFM families: FOXI3:p.(Leu199Phe), p.(Arg238Gln), p.(Arg240Cys), and p.(Arg240His) (Fig. 1D, Supplementary Fig. 2A, and Table 1). A further 9 FOXI3 variants were found by targeted-capture sequencing in a cohort of 498 additional sporadic CFM patients: 3 variants p.(Arg235Cys), p.(Arg236Gln), and p.(Arg238Gln) are located in the nuclear localization signal (NLS); 2 variants p.(Pro147Thr) and p.(Ser169Asn), are located in the Forkhead domain (FHD); 2 inframe deletions of p.(Ser119_Ala124del) and p.(Ser373_Thr376del), and 2 variants of p.(Pro355Leu) and p.(Arg415Ter) map in the intrinsically disordered regions (IDRs) of FOXI3 (Fig. 1E, Fig. 2A, and Table 1). Supplementary Table 4 lists the variants in known candidate genes related to syndromes with microtia found in this cohort.
Table 1.
Likely pathogenic rare variants within the FOXI3 gene identified from patients (CFM or Goldenhar syndrome)
| Family ID | Proband ID | Chromosome Position (hg38) | Reference | Alternative | Consequence | FOXI3 cHGVS NM_001135649.3 | FOXI3 pHGVS | In silico predictions | gnomAD_Allele_Frequency (v3.1.2) |
|---|---|---|---|---|---|---|---|---|---|
| EUR04 | EUR04 (SEA15423) | chr2:88452445 | G | GG | frameshift | c.92dupG | p.(Ala32GlyfsTer147) | CADD: 23.0 | absent |
| EUR05 | EUR05 (SEA15566) | chr2:88452231 | A | T | missense | c.305T>A | p.(Phe102Tyr) | CADD: 14.07 SIFT: 0.96 Polyphen2: 0 | absent |
| CHN-S001 | CHN-S001 | chr2:88452164 | CGCGGGCGCGGCGGGCGAG | C | inframe deletion | c.354_371del | p.(Ser119_Ala124del) | CADD: 19.88 | 0.0000199 |
| CHN-S002 | CHN-S002 | chr2:88452097 | G | T | missense | c.439C>A | p.(Pro147Thr) | CADD: 28.3 SIFT: 0 Polyphen2: 1 | absent |
| CHN-S003 | CHN-S003 | chr2:88452030 | C | T | missense | c.506G>A | p.(Ser169Asn) | CADD: 27.4 SIFT: 0 Polyphen2: 0.994 | absent |
| CHN05 | CHN05 | chr2:88451941 | G | A | missense | c.595C>T | p.(Leu199Phe) | CADD: 29.8 SIFT: 0 Polyphen2: 1 | absent |
| EUR03 | EUR03 (SEA15422) | chr2:88448797 | A | G | missense | c.673T>C | p.(Cys225Arg) | CADD: 26.4 SIFT: 0.02 Polyphen2:0.599 | absent |
| EUR02 | EUR02(SEA15533) | chr2:88448770 | A | C | missense | c.700T>G | p.(Phe234Val) | CADD: 29.1 SIFT: 0 Polyphen2: 0.992 | absent |
| F252 | IV: 5 | chr2:88448768 | G | T | missense | c.702C>A | p.(Phe234Leu) | CADD: 26.4 SIFT: 0 Polyphen2: 0.896 | 0.00001314 |
| CHN-S004 | CHN-S004 | chr2:88448767 | G | A | missense | c.703C>T | p.(Arg235Cys) | CADD: 32 SIFT: 0 Polyphen2: 0.998 | absent |
| CHN01 | IV: 8 | chr2:88448764 | G | A | missense | c.706C>T | p.(Arg236Trp) | CADD: 23.9 SIFT: 0.03 Polyphen2: 0.899 | absent |
| CHN-S005 | CHN-S005 | chr2:88448763 | C | T | missense | c.707G>A | p.(Arg236Gln) | CADD: 28.7 SIFT: 0 Polyphen2: 0.899 | 0.00000657 |
| CHN02 | II: 2 | chr2:88448757 | C | T | missense | c.713G>A | p.(Arg238Gln) | CADD: 28.8 SIFT: 0.04 Polyphen2: 1 | absent |
| CHN-S006 | CHN-S006 | chr2:88448757 | C | T | missense | c.713G>A | p.(Arg238Gln) | CADD: 28.8 SIFT: 0.04 Polyphen2: 1 | absent |
| CHN03 | II: 2 | chr2:88448752 | G | A | missense | c.718C>T | p.(Arg240Cys) | CADD: 29.1 SIFT: 0 Polyphen2: 0.993 | absent |
| EUR01 | EUR01 (SEA9213) | chr2:88448752 | G | A | missense | c.718C>T | p.(Arg240Cys) | CADD: 29.1 SIFT: 0 Polyphen2: 0.993 | absent |
| CHN04 | II: 2 | chr2:88448751 | C | T | missense | c.719G>A | p.(Arg240His) | CADD: 28.2 SIFT: 0 Polyphen2: 0.993 | 0.00002628 |
| CHN-S007 | CHN-S007 | chr2:88448406 | G | A | missense | c.1064C>T | p.(Pro355Leu) | CADD: 12.73 SIFT: 0.09 Polyphen2: 0.003 | 0.00002094 |
| CHN-S008 | CHN-S008 | chr2:88448341 | CGGTGCTATTGCT | C | inframe deletion | c.1117_1128del | p.(Ser373_Thr376del) | CADD: 14.11 | 0.00003245 |
| CHN-S009 | CHN-S009 | chr2:88448341 | CGGTGCTATTGCT | C | inframe deletion | c.1117_1128del | p.(Ser373_Thr376del) | CADD: 14.11 | 0.00003245 |
| CHN-S010 | CHN-S010 | chr2:88448227 | G | A | stop gained | c.1243C>T | p.(Arg415Ter) | CADD:38 | 0.000006574 |
| EUR06 | EUR06 (SEA15421) | chr2:87317229-89306982 | + | - | SV deletion | chr2: 87317229-89306982 deletion | / | / | / |
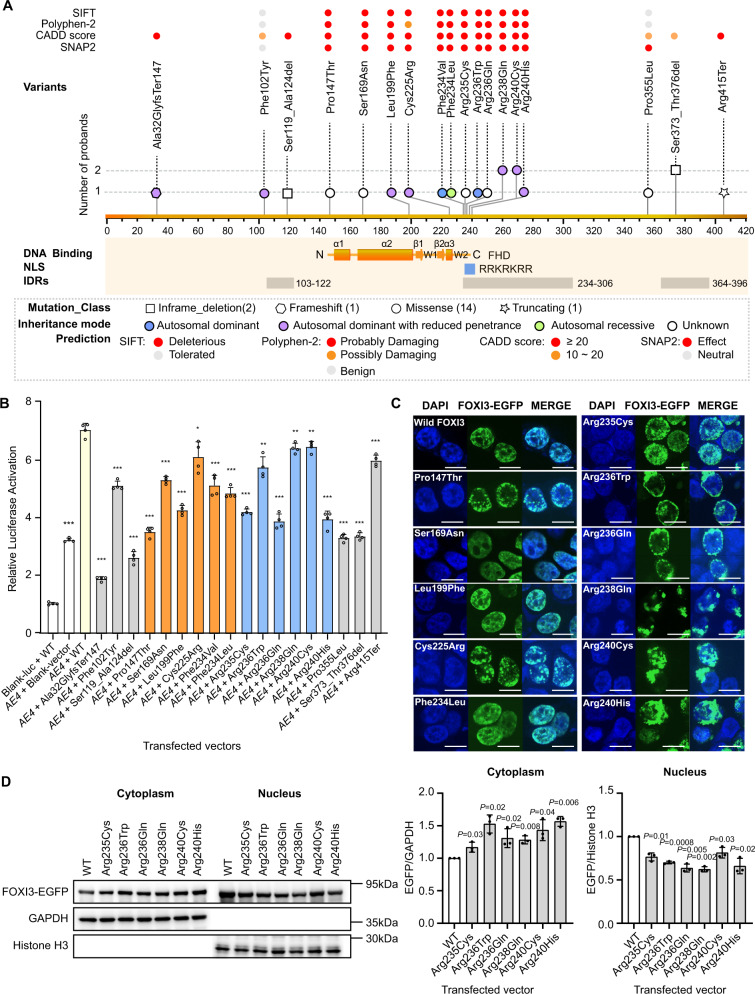
Fig. 2. The effect of FOXI3 variants with in silico prediction and in vitro experiments.
A The 18 identified variants on FOXI3. The predictions of the variants are classified by bins and shown on the top of each variant. The variants are classified by the type of inframe_deletion (square), frameshift (hexagon), missense (circle), and truncating (star), and the inheritance mode of autosomal dominant (blue) or with reduced penetrance (purple), autosomal recessive (green), and unknown (white). The variants of p.(Arg238Gln), p.(Arg240Cys), and p.(Ser373_Thr376del) are detected in two probands and the rest are in one proband. B Effects of the 18 variants on the transactivation of FOXI3 in HEK293T cells (n = 4/group). Gray Bars: intrinsically disordered regions (IDRs), orange bars: forkhead domain (FHD), blue bar: nuclear localization signal (NLS). *P < 0.05, **P < 0.01, ***P < 0.001. Each experiment was repeated at least three times. C The localization and distribution of wild type or mutant FOXI3-EGFP fusion proteins in HEK-293T cells. DAPI counterstain (blue) shows the location of the nucleus. EGFP (green fluorescent protein) shows the subcellular localization of the FOXI3-EGFP fusion protein. Wild-type FOXI3 is almost completely transferred into the nucleus and uniformly distributed. A majority of FOXI3 with mutant NLS is blocked outside the nucleus; FOXI3 with mutant FHD tends to form intranuclear aggregation; most of FOXI3 with mutant IDR is similar to Wild-type FOXI3. Scale bar = 10 μm. Each experiment was performed in triplicate and repeated at least three times. D Representative western blots of FOXI3-EGFP with mutant NLS. Immunoprecipitation on the EGFP-tagged FOXI3 using antibody of EGFP and detect the amount of FOXI3-EGFP in the nucleus and cytoplasmic fractions of HEK293T cells (n = 3 biologically independent experiments). Compared to the wild type, the mutant FOXI3-EGFP fusion protein is significantly trapped in the cytoplasm and the amount of mutant FOXI3-EGFP is significantly reduced in the nucleus. Significant differences between two groups (wild type and each mutant) were determined by unpaired Student’s t-test (two-tailed). Data are presented as the means ± s.d. Source data are provided as a Source Data file.
FOXI3 likely pathogenic variants in European CFM families
The Geneva laboratory screened the FOXI3 gene by Sanger sequencing or WES in 124 CFM probands of European ancestry; 6 probands with heterozygous rare non-synonymous or LoF, likely pathogenic variants were identified (Table 1, Supplementary Table 1, and Supplementary Fig. 2B). The phenotype of 5 European CFM families was CFM with isolated microtia type II or type III (family EUR01 is shown in Supplementary Fig. 2C). The variants identified were p.(Ala32GlyfsTer147), p.(Phe102Tyr), p.(Cys225Arg), p.(Phe234Val), and p.(Arg240Cys) were heterozygous in the five probands, respectively (Table 1). Notably, these variants were inherited in each case from an asymptomatic individual (EUR01, EUR03, EUR04, and EUR05) or transmitted from a mildly affected parent (EUR02) (Supplementary Fig. 2B), indicating autosomal dominant inheritance with reduced penetrance. In family EUR01, two asymptomatic brothers of the proband also inherited the mutant p.(Arg240Cys) allele.
In order to test the hypothesis that a modifier FOXI3-related variation in trans with a pathogenic FOXI3 allele contributes to the phenotype in the seemingly autosomal dominant pedigrees presented above, we studied samples from a proband of family EUR06 previously described with type III microtia, and a 2.06 Mb deletion that included FOXI3 inherited from the asymptomatic father19. We used primary cultured fibroblasts (FFF0062013 cell line) from this case to identify a potentially contributing variant in the remaining FOXI3 allele. WGS with short reads, did not identify any candidate non-coding variants within the FOXI3-containing TAD. WGS long-read Nanopore did not reveal any structural variant at the remaining trans FOXI3 locus. Whole-genome optical mapping also validated the large 2.06 Mb deletion (Supplementary Fig. 3) and did not reveal any abnormality in the remaining copy of the FOXI3 locus. We were able to determine the haplotype of the polymorphic sites of the remaining FOXI3 allele. This haplotype contains two linkage disequilibrium (LD) blocks, and is common in Europeans (12.9%) and East Asians (19.7%) (data from LDlink 5.1 https://ldlink.nci.nih.gov/) (Supplementary Fig. 4). No quantitative trait loci (QTLs) were identified in GTEx due to undetectable expression of FOXI3 in adult tissues20–22. This haplotype is discussed further below.
The contribution of FOXI3 mutations to CFM
The frequency of the FOXI3 causative variants in CFM patients of European ancestry is 5 out of 124 (4.0%); the case of EUR06 was not counted since it has been described in the literature and the total number of CFM patients studied was not mentioned. In the 48 unrelated CFM families of Chinese ancestry, 5 had FOXI3 pathogenic variants (10.4%). In the 498 additional Chinese CFM cases, there were ten patients with pathogenic FOXI3 variants (2.0%). Thus, FOXI3 pathogenic variants account for 3.1% of CFM cases of European and Chinese ancestry combined (21 of 670 cases). The frequency of all FOXI3 variants with a CADD score of >20 in the BRAVO database23 (https://bravo.sph.umich.edu/freeze8/hg38) is 0.34% (456 heterozygous in 132,345 samples) which is 10 times less frequent than in CFM (χ2 test P value 10−5).
In silico analysis of FOXI3 deleterious variants
Figure 2A depicts the mapping of the 18 identified likely pathogenic variants including the 14 non-synonymous variants in FOXI3; 6 map within the FHD, further 6 within the NLS, and 2 within the IDR domains. These variants were predicted to be damaging to FOXI3 according to the scores of SIFT (Sorting Intolerant From Tolerant), Polyphen (Polymorphism Phenotyping v2), CADD (Combined Annotation Dependent Depletion), or SNAP2 (Fig. 2A).
We performed 3D modeling based on the structures of the related human FOXC2 (PDB ID: 6AKO)24 or Foxk1A (PDB ID: 2C6Y)25 (Supplementary Fig. 5A). The modeled FOXI3:DNA complex covers the FHD and NLS which harbor 12 variants distributed at 9 sites (Supplementary Fig. 5B, C). The p.(Pro147Thr), p.(Cys225Arg), and p.(Phe234Val) would decrease free energy by more than 1.6 kcal/mol and, using Foldx software26, are predicted to affect the stability of FOXI3 (Supplementary Fig. 5D). In the 3D modeled FOXI3, the Cys225 was predicted to form a disulfide bond with Cys202, while Arg225 could not. Phe234 was on the C-terminal end of the Forkhead domain and was predicted to interact with Ala152 and Ala155 of alpha-helix 2 and with Phe229, forming a hydrophobic cluster. The mutant Val234 would change the hydrophobic cluster and destabilize the structure of FOXI3 (Supplementary Fig. 5E). The p.(Ser169Asn) and p.(Leu199Phe) are both in the alpha-helix and cannot form hydrogen bonds with Thr167 and His195, respectively, which may affect the stability of FOXI3 (Supplementary Fig. 5E). The positively charged residues of Arg235, Arg236, Arg238, and Arg240 are all within the conserved NLS domain (Supplementary Fig. 6A) and were predicted to affect the ability of FOXI3 to enter the nucleus (Supplementary Fig. 6B). Each of these variants also conforms to the CG to TG hypermutation rule27. As with variants in other NLS sequences, these variants were also predicted to perturb binding DNA as the substitutions would break the hydrogen bonds with phosphate moieties of the DNA backbone; this is consistent with the free energy calculation prediction (ΔGmut − ΔGwt < 1.6 kcal/mol and ΔΔG(monomer) − ΔΔG(complex) > 0.25; Supplementary Fig. 5D).
In vitro analysis of FOXI3 deleterious variants
We investigated the effects of all 18 likely pathogenic variants on the transcriptional activity of FOXI3. We co-transfected wild-type or mutant FOXI3 with the AE4 promoter luciferase reporter28,29 in HEK-293T cells and found that the luciferase activity was reduced for all mutations compared to wild-type FOXI3 (Fig. 2B).
We transfected EGFP-tagged FOXI3 into HEK-293T cells followed by immunostaining to visualize the subcellular localization of mutant FOXI3. The wild-type FOXI3 was predominantly localized in the nucleus; the mutant FOXI3 were (i) barely or partially entering the nucleus, and aggregating in the cytoplasm and nucleus (p.(Arg235Cys), p.(Arg236Trp), p.(Arg236Gln), p.(Arg238Gln), p.(Arg240Cys), p.(Arg240His); (ii) mostly entering the nucleus but aggregating into granular foci (p.(Pro147Thr), p.(Cys225Arg), p.(Phe234Val); (iii) no difference from the wild-type (all other FOXI3 variants) (Fig. 2C and Supplementary Fig. 7). Western blot analysis on EGFP-tagged FOXI3 from nuclear and cytoplasmic fractions again demonstrated that the NLS variants significantly reduce the amount of FOXI3 in the nucleus (Fig. 2D).
Mouse models with FOXI3 variants
We produced mouse lines with Foxi3 mutations considering the following criteria: 1) mutations should be located in conserved regions with the highest mouse-human homology, thus those within the FHD domain and NLS (97.9% between human and mouse) were preferred (Supplementary Fig. 8A). 2) mutations in regions such as the NLS of FOXI3 were preferred (P = 9.358E-8, Chi-square test). 3) mutations observed in the largest CFM pedigrees (Fig. 1A, C, E, and Supplementary Fig. 8B). 4) mutations that severely affected the transcriptional activity or that severely disturb FOXI3 entering the nucleus. Finally, we produced two mouse lines within the C-terminal end of FHD and the NLS: Foxi3R224H equivalent to human p.(Arg240His) and Foxi3F218L equivalent to human p.(Phe234Leu) (Fig. 3A, Supplementary Fig. 9A and 10A).
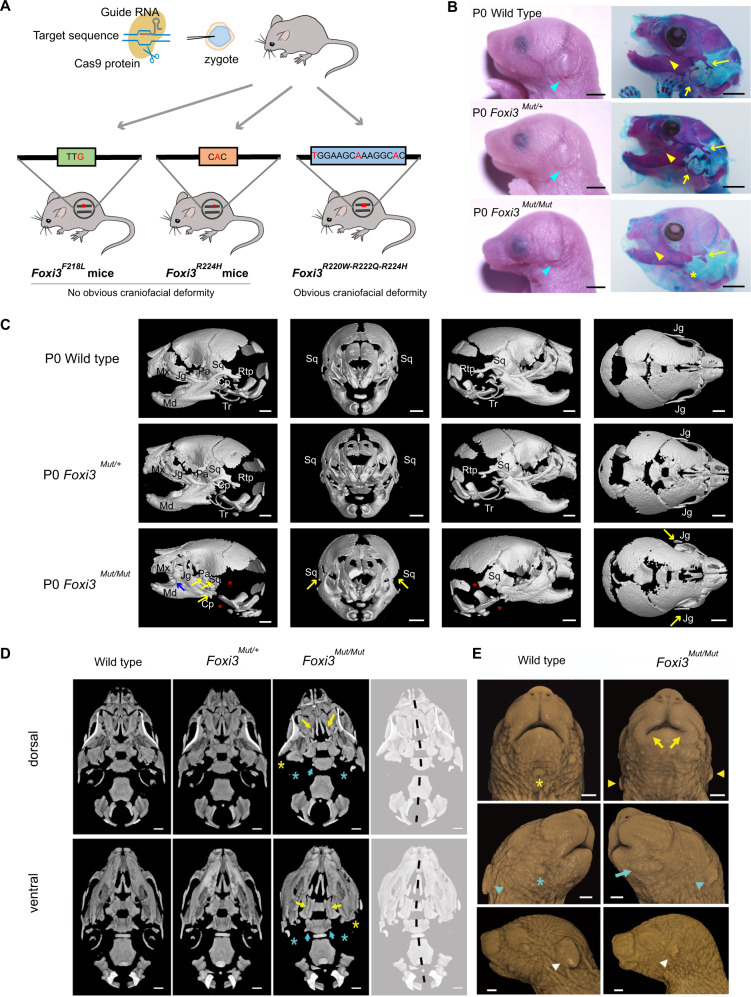
Fig. 3. Generation and characterization of FOXI3 mutant mice.
A A schematic illustration of the generation of Foxi3 mutant mice. Three mouse lines are created including Foxi3F218L, Foxi3R224H, and Foxi3R220W-R222Q-R224H. B Craniofacial differences of wild-type, heterozygous, and homozygous Foxi3R220W-R222Q-R224H neonatal mice (n = 3 for each genotype). Comparing with wild type or heterozygotes, the cyan arrows indicate homozygotes with small pinnae, and various overt cranioskeletal defects, including abnormal middle and inner ear bones, syngnathia (mandible and maxilla fusion), and anomalous squamosal development (yellow arrows). The left panel was photographed with bright field; the right panel of mice were stained with Alizarin Red and Alcian Blue. Scale bar = 2 mm. C Asymmetric presentation of cranioskeletal features in the neonatal Foxi3R220W-R222Q-R224H homozygous mice (n = 4 for each genotype). Micro-CT images show variable development of the jugal bone, and almost complete loss of the squamous bone in one or both sides in homozygous neonates. The blue arrow indicates the fusion of the mandible (Md) to the maxilla (Mx) in a homozygote. The yellow arrow indicates the poorly developed squamosal (Sq), jugal (Jg), palatine (Pt), and condylar process (Cp). The red asterisk (*) indicates the retrotympanic process (Rtp) and the tympanic ring (Tr) that are absent or severely hypoplastic in homozygotes. D Dorsal and ventral views of the cranial base show the absent or hypoplastic bony ear structures (noted in (C); asterisked) as well as hypoplasia of the lateral pterygoids of the basisphenoid (cyan arrows) and cleft palate (yellow arrows, dorsal view), with ventrally protruding palatine bones (yellow arrows, ventral view) of newborn triple-mutant mice. Dashed black line denotes the asymmetry of the skull centered on the cranial base. E Surface rendering of the facial soft tissue from the same micro-CT scans highlights the severe left-side microtia in the homozygote (white and cyan arrowheads) and asymmetry in positions of the pinnae (yellow arrowheads). Homozygotes exhibit microstomia that is asymmetric in presentation (yellow arrows) and, variably, a row of whisker follicles on the ‘mandible’ (blue arrow) for the newborn triple-mutant mice. Scale bar = 1 mm in (C), (D), and (E). P0: Postnatal day 0.
Foxi3F218L/F218L and Foxi3R224H/R224H mice did not show any obvious pathological phenotypes (Supplementary Fig. 10D and Supplementary Fig. 9C). We crossed Foxi3F218L/F218L to previously produced Foxi3+/- mice (without abnormalities)30,31 to create Foxi3F218L/- animals to investigate the effect of the mutation. These mice failed to thrive after birth; were underweight, exhibited respiratory abnormalities, and died between birth and day 5. Foxi3F218L/- mice did not show defects in the external, middle, or inner ears, the mandible, or the maxilla. However, Foxi3F218L/- mice had absent thymus previously observed in Foxi3–/– mice32 (Supplementary Fig. 10D).
Although we had shown that the human FOXI3 p.(Arg240His) variant is within the NLS and disturbs FOXI3 entering the nucleus (Fig. 2 C, D), we tested whether its homologous variant in Foxi3 p.(Arg224His), as well as all the other five amino acid sites in the NLS, also had the same effect. Surprisingly, transfected EGFP-tagged mutant Foxi3 predominantly entered the nucleus (Supplementary Fig. 11), which differs from the human homologous mutant FOXI3 (Fig. 2C). This phenomenon can be explained by either an artifact of the HEK-293T transfection assay, or a real difference between the nuclear localization requirements of the mouse and human mutant proteins. The latter was considered the likely reason since the Foxi3R224H/R224H mice did not show any obvious pathological phenotypes (Supplementary Fig. 9C).
Mouse model with NLS dysfunction
We found that 6 of 18 identified mutations were in the NLS, which represents a significant enrichment (P = 9.358E-8, Chi-square test) since the NLS is comprised of only 7 amino acids (the full length of FOXI3 is 420 AA). Thus, we tried to produce a mouse model with NLS dysfunction to recapitulate the CFM phenotype. We introduced additional identified FOXI3 variants in the NLS and found that two-point mutant Foxi3 still has the ability to enter the nucleus but that triple-point mutant Foxi3 was unable to enter the nucleus (Supplementary Fig. 11). We, therefore, produced a triple-mutant knock-in mouse line carrying p.(Arg220Trp), p.(Arg222Gln), and p.(Arg224His) (equivalent to human FOXI3 p.(Arg236Trp), p.(Arg238Gln), and p.(Arg240His)), named the triple-mutant model (Fig. 3A and Supplementary Fig. 9B, D).
The birth weight and length of the homozygous neonatal (P0) triple-mutant mice (n = 9) were significantly smaller than wild-type (n = 7) or heterozygous (n = 15) mice (Supplementary Fig. 9E). Homozygous triple-mutant mice die immediately after birth. They show an underdeveloped mandible and a complete absence of the external ear (pinna), replaced by small pre-auricular tags (Fig. 3B and Supplementary Fig. 9F). The heterozygous P0 mice only show a hyperplastic bone between the unfused frontal bones which was also observed in P0 homozygous mice (Fig. 3C). Neonatal homozygous mice revealed a partially truncated mandible and fusion of the upper and lower jaws (syngnathia) and anomalies of various bones of the ear (Fig. 3B); the tympanic ring, malleus, and incus were missing and the stapes were partially developed (Supplementary Fig. 9F).
Micro-CT scans show asymmetric craniofacial defects in homozygous Foxi3R220W-R222Q-R224H mice. Bony anomalies include poorly developed squamosal (Sq) and jugal (Jg) bones, absent or severely hypoplastic retrotympanic process and tympanic ring (Fig. 3C), hypoplasia of the lateral pterygoids of the basisphenoid and cleft palate in 50% of homozygotes (n = 4) (Fig. 3D). All homozygous neonates showed asymmetry of the skull. The pinna varied from severe (grade 4) to grade 2 microtia (Supplementary Fig. 12). Auricular size in homozygotes ranged between 35 and 70% of the wildtype. All homozygotes also exhibited asymmetric microstomia (Fig. 3E). A row of whisker follicles on the “mandible” was evident in one in four homozygotes strongly suggesting the syngnathia is a result of a partial mandibular to maxilla transformation, similar to that in Auriculocondylar syndrome33.
A FOXI3 region trans haplotype as a determinant of reduced penetrance
The study of the pedigrees described above demonstrated a reduced penetrance for CFM with microtia types II or III. One molecular mechanism of the penetrance could be the nature of the FOXI3 allele in trans with the pathogenic allele. We propose the following hypothesis (Fig. 4A): (i) homozygotes for a pathogenic variant allele show microtia (pedigree F252). We term this genotype combination FOXI3 Mut/FOXI3 Mut; (ii) heterozygotes for a pathogenic variant and a modifier trans allele also show microtia (pedigree EUR01). This genotype combination is FOXI3 Mut / FOXI3 modifier; (iii) heterozygotes for a pathogenic variant and a non-modifier trans allele: there is no microtia in these cases; instead, the external ear could be normal or mildly dysmorphic. This genotype combination is FOXI3 Mut / FOXI3 wild type or normal. To test this hypothesis, we sequenced the genome of selected individuals, or genotyped selected polymorphic variants.
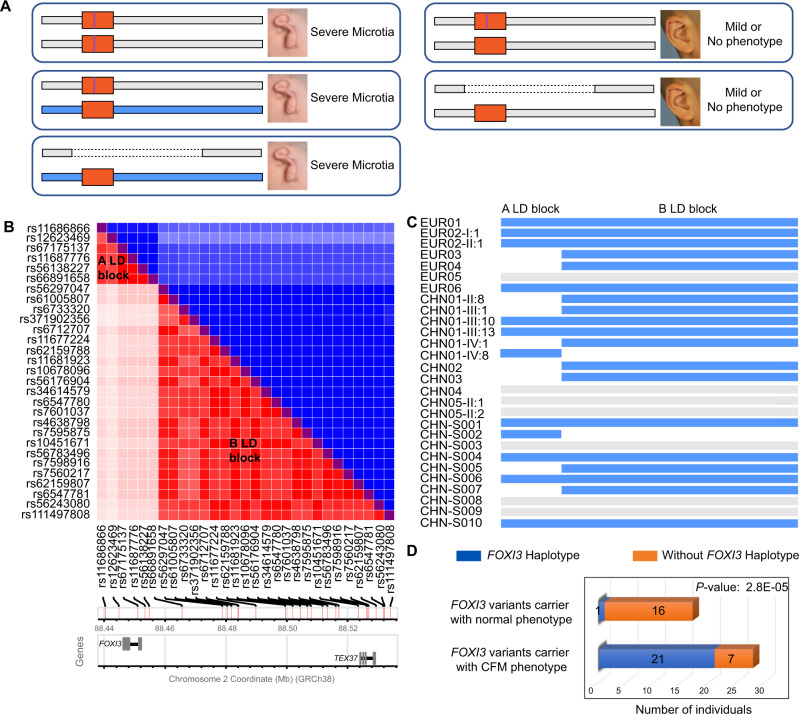
Fig. 4. Presumed FOXI3 haplotype that modifies the clinical severity of pathogenic FOXI3 variants.
A The hypothesis of FOXI3 haplotype for the reduced penetrance of CFM with microtia types II or III. The graph shows three possible genotype combination for CFM with microtia. The orange rectangular box indicates FOXI3 genes in the human genome, the purple line means a pathogenic variant allele. The blue rectangular depicts the presumed modifier trans FOXI3 haplotype while the gray rectangular is the common haplotype. The dashed line indicates the missing haplotype. B Linkage disequilibrium (LD) blocks A and B (see text) of polymorphic sites shown in the y- and x-axis (data from Europeans and Chinese). The x-axis also depicts a schematic representation of the map position of the polymorphic sites along chromosome 2 (GRCh38). The blue part is the D’ and the red is the r2 statistics. C Presence of the presumed modifier haplotype of A LD block and/or B or both in the DNA of individuals with microtia type III or preauricular tags described in this study. D Chi-square test for the trans FOXI3 haplotype in CFM and normal individuals. The blue column square indicates the individual who carried FOXI3 variants, as well as trans FOXI3 haplotype, and the orange column square, indicates the individual who carried FOXI3 variants but without trans FOXI3 haplotype.
The sequence of the FOXI3 deletion case EUR06 provided a candidate trans FOXI3 modifier haplotype. This 94.83 kb haplotype (from rs11686866 to rs111497808 including FOXI3) was present in heterozygosity in 7 of 25 (28%) CFM cases with types II or III microtia. There are two Linkage Disequilibrium (LD) blocks (A and B) in this haplotype (Fig. 4B). The A LD block extends for 25.24 kb from rs11686866 to rs66891658; the B LD block spans 59.1 kb 5′ to the FOXI3 gene, from rs56297047 to rs111497808. A total of 21 cases with microtia have the A LD and/or B LD blocks in heterozygosity (10 cases with A + B, 2 with A, and 9 with B) suggesting that there are at least two different sites contributing to the phenotypic variation, while 7 cases did not have this haplotype (Fig. 4C). Furthermore, there were 16 cases from different pedigrees with a likely pathogenic FOXI3 variant without the modifier haplotype in which there was no microtia. The P value of the χ2 test of this comparison is 2.8E-05 (Fig. 4D). Family-based transmission disequilibrium testing showed that the modifier haplotype is also significantly (P = 0.0325) associated with CFM phenotype. The alleles of the polymorphic sites for the A LD and the B LD blocks are listed in Supplementary Fig. 3.
The trans FOXI3 modifier haplotype is absent in the unaffected carrier parents and siblings of the Pakistani family. In addition, the trans FOXI3 modifier haplotype is not present in the unaffected carrier father and brothers of family EUR01, while the proband with the combination FOXI3 p.Arg240Cys / FOXI3 modifier has microtia. There was a similar configuration in family CHN03. In pedigree CHN01, the phenotypic expression is variable and only 4 individuals showed microtia type III, while the other carriers have milder manifestations of the external ear. Three of these 4 individuals have the FOXI3 modifier B LD block and one the A LD block. However, there are also individuals who carry the FOXI3 modifier haplotype without severe microtia. Thus, the presence of the proposed FOXI3 modifier haplotype(s) could not explain all the cases of incomplete penetrance. The affected individuals have the FOXI3 modifier haplotype, while the carrier asymptomatic mothers lack this haplotype.
Discussion
We describe the identification of pathogenic variants in FOXI3 that cause one form of CFM. Approximately 3.1% of 670 CFM cases are associated with pathogenic variants of FOXI3. There is a possible genotype–phenotype correlation; the majority of FOXI3 pathogenic variants result in CFM with isolated type III microtia.
The FOXI3 protein belongs to the forkhead transcription factor family involved in cell fate decisions during embryonic development34,35. The forkhead domain proteins bind DNA as monomers, dimers, or in a complex with other proteins36. This family of proteins in humans contains 50 members classified into 19 subfamilies, one of which contains FOXI1, FOXI2, and FOXI334,37,38. Pathogenic variants in several members of this gene family cause various Mendelian disorders34,39, and are also involved in cancer. Some of these include FOXC1 and FOXE3 in anterior segment dysgenesis syndrome type 3 and type 2, respectively, FOXF1 CNV, SNV, and lncRNA in lethal lung developmental defects, FOXG1 in an atypical Rett-like syndrome, FOXJ1 in one form of Ciliary Dyskinesia syndrome, FOXL2 in blepharophimosis-epicanthus inversus and ptosis syndrome, FOXE1 in the Bamforth-Lazarus syndrome, FOXP1 in intellectual disability with language impairment, FOXP2 in speech and language disorder, FOXN1 in T cell immunodeficiency, alopecia and nail dystrophy, and FOXP3 in immune regulation polyendocrinopathy syndrome34. The gene FOXI1, the closest relative of FOXI3 is involved in Pendred / Enlarged Vestibular Aqueduct syndrome, an autosomal recessive disorder characterized by the combination of goiter and inner ear anomalies40.
The molecular cause of the reduced penetrance is elusive and requires a better understanding of the regulation of FOXI3 expression during development. Potential causes of the reduced penetrance include: (1) Stochastic monoallelic expression41 of the mutant allele. (2) The nature of the trans FOXI3 allelic region. The hypothesis that a trans haplotype harbors a regulatory variant that modifies the expression of its cis-FOXI3 gene is discussed in the “Results”, and has been described as a Compound Inheritance Gene Dosage model42. Examples include the Thrombocytopenia Absent Radius (TAR)43 and the TBX6-related congenital scoliosis42. (3) The presence of cis or trans eQTLs associated with the expression levels of FOXI3, as observed for FOXF1 in a lethal lung developmental disorder44. The expression level of FOXI3 is very low (https://gtexportal.org/home/ and ref. 45) and no eQTLs have been identified. (4) Digenic inheritance similar to some families with Retinitis Pigmentosa46 and Bardet-Biedl syndrome47. (5) Somatic mutations (a second hit hypothesis). This two-hit phenomenon is common in cancer48, and has been shown to occur in developmental disorders such as in Hereditary Hemorrhagic Telangiectasia49. (6) Epigenetic modifications other than imprinting as in the case of DNMT3B variants that modify epigenetic repression of the D4Z4 repeat and the penetrance of facioscapulohumeral dystrophy50. (7) Non-genetic factors including environmental effects.
The potential mechanism of the modifier FOXI3 containing haplotype described here could be due to common variants of an enhancer sequence that regulate the level of FOXI3 expression. One testable hypothesis is that the modifier haplotype is linked to a reduced expression of the normal FOXI3 and therefore the effect of the mutant FOXI3 allele is stronger. In contrast, all the other FOXI3 haplotypes are linked to higher expression of the normal FOXI3 gene and thus the effect of the mutant FOXI3 is reduced or it is negligible. An analogous example is that of a hypomorphic allele of the FECH gene that increases the penetrance of the autosomal dominant erythropoietic protoporphyria when it occurs in trans to a pathogenic FECH variant51.
Heterozygous FOXI3 mutations have been observed in some hairless dog strains; such dogs have not only hair defects but also dental defects. The FOXI3 variant in hairless dogs is a dominant negative neomorphic mutation52. The hairless dog phenotype is always inherited in an autosomal semidominant fashion. The mutation in these dogs occurs early in the coding regions and produces a new protein as a result of a 7 bp duplication that shifts the reading frame. In two previous collaborative studies, conditional mouse knockouts of Foxi3 were created in teeth and hair, and although tooth and hair defects were present in the conditional null mice, these defects were significantly less strong than those observed in Foxi3 mutant dog breeds53,54.
The severity of phenotypic expression of a causative variant is related to the extensive genomic variation of each individual; the severity of sickle cell anemia in different populations provides an excellent example55. The possibility of imprinting of the FOXI3 locus could be excluded based on the pedigrees studied. In addition, FOXI3 has not been identified as an imprinted gene; however, transient or tissue-specific imprinting could not be excluded56,57.
The phenotypic expression of the pathogenic FOXI3 variants is variable. This is evident in family CHN01, where the p.(Arg236Trp) results in various ear and facial phenotypes. Potential explanations may include: the random monoallelic expression of FOXI3 during development41; the randomness of a developmental process when a key protein is dysfunctional or reduced as in Kartagener syndrome where heart looping is randomized between the left and right sides58. In almost all syndromes of abnormal development of fingers and toes, the phenotype varies between the two sides as for example in the GLI3-related Pallister-Hall syndrome59.
Previously reported Foxi3–/– mice show abnormalities in the inner ear, the jaw, external, and middle ear, heart and artery development, and thymus27–29, while the severity of organs involved is quite different from CFM. However, our single-point mutation mice did not have visible malformations. This may be explained by genetic, developmental and physiological differences between mice and humans60, or the poor sequence homology (69.74%) between mice and humans for FOXI3; in addition, the timing of facial development and the rate of growth of embryonic facial precursors is quite different between mice and humans, which may underlie the differences in sensitivity to changes in the activity levels of transcription factors including FOXI3. However, the triple-mutation mice show many similar anomalies to human CFM phenotypes.
Different tissues in mice and humans have differing sensitivities to a reduction in Foxi3 function. Foxi3 is expressed in the ectoderm and endoderm of the pharyngeal arches but not in the migrating crest cells that populate these arches30. The thymus, originating from the third pharyngeal pouch, is the most sensitive. Foxi3 heterozygous mice and patients with a loss of one allele of FOXI3 show thymus defects and T-cell lymphopenia61. The patients described in ref. 48 all have large deletions that include the FOXI3 gene, and one had craniofacial abnormalities. The external ear is the second most sensitive structure, with microtia associated with many of the human FOXI3 variants. In the homozygous triple-mutant mice, microtia is fully penetrant, and is associated with perturbed development of the inner ear and middle ear ossicles, anomalies of the cranial base and palate, and syngnathia. The inner ear defects are derived from Foxi3-expressing preplacodal tissue30,31,62, while the middle ear and mandibular structures are derived from neural crest cells migrating into the first and second pharyngeal arches.
Studies on the gene network of FGF signaling (particularly FGF19) during otic induction in chick embryos have placed Foxi3 in the first step of otic induction i.e., the formation of the otic-epibranchial progenitors in the posterior pre-placodal region63,64. Additional proteins besides Foxi3, involved in this FGF signaling are Mapk, Etv5, Gbx2, Pax2, Six1, Six4, and Eya263,64. Thus, all of these proteins could also be candidates for pathogenicity in CFM cases. Previously studies found that Foxi3-Gata3 and Foxi3-Fgf8 signaling is essential for facial morphogenesis30,31,65, and genome-wide association studies also found 15 CFM-related genes15,16. Variation in these genes could also be candidates of modifiers for the CFM phenotypes. However, we did not find any coding variants in them in the cohort of patients described here.
In conclusion, we provide genetic, genomic, and biological experimental evidence that likely pathogenic FOXI3 variant alleles cause one form of Craniofacial Microsomia (CFM).
Methods
Ethics statement
All procedures involving human samples were performed with the approval of the Swiss Bioethics Committee of the University Hospitals and the Canton of Geneva (CER 11-036) and the Ethics Committees of the School of Biological Science and Medical Engineering, Beihang University (BM20210057), the Beijing Tongren Hospital, Capital Medical University, and the Plastic Surgery Hospital of the Peking Union Medical College and conducted under the Principles of Helsinki or an IRB approved protocol from USA. The study has been registered and approved by China’s Ministry of Science and Technology (project 2023-CJ0849). All participants or their guardians signed informed consent forms for biological investigation. All the European samples were collected after informed consent from the patients or their parents. Consent for the publication of photographs was given after a separate explanation and request. The FFF0062013 fibroblasts cell line was obtained from the Galliera Genetic Bank in Genova, Italy under the appropriate ethics approval for the distribution of samples (https://openbioresources.metajnl.com/articles/10.5334/ojb.15/). All animal experiments were performed under the approval of the Animal Care Committee at Beihang University (BM20210057) and the Institutional Animal Care and Use Committee at Baylor College of Medicine (D16-00475).
Patient samples
The Chinese subjects were collected from the Beijing Tongren Hospital and the Plastic Surgery Hospital of the Peking Union Medical College. For the Chinese cohort, exome sequencing and target-capture sequencing were performed on 159 enrolled individuals from 48 CFM families and 498 sporadic CFM patients (Supplementary Table 2). The 48 probands include 36 males and 12 females and the 498 sporadic CFM patients include 396 males and 102 females. In the Pakistani family, ten individuals were enrolled including five males and five females. In the European patients, 124 probands (including 83 males and 41 females) were included in the project. The clinical characteristics of the Chinese CFM patients are shown in Supplementary Table 2. Ages range from 5 to 18 years in probands from CFM families and from 5 to 41 years in sporadic CFM patients. Clinical diagnosis for CFM is based on the OMENS classification system.
Exome sequence and computational analyses
Exome sequencing
Genomic DNA from blood was extracted using standard procedures. DNA libraries for exome sequencing were prepared from 2 μg of qualified genomic DNA, which was sheared to 180–200 base pairs. The targeted coding regions of more than 20,000 genes were captured by Agilent SureSelect Human All Exon kit (V6) (5190-8883, Agilent Technologies, Inc., CA, USA) and Twist according to the manufacturer’s protocol. Paired-end sequencing (2 × 150 bp) was performed on the Illumina Xten or NovaSeq platforms (Illumina Inc., CA, USA) at Compass Biotechnology company (Whole-exome sequencing) or IGeneTech Biotechnology company (Target-capture sequencing) (Beijing, China).
Dataset processing and family-based burden test
Burrows–Wheeler aligner tool (v0.7.17)66, SAMtools (v1.9)66, PICARD (v2.0.1), and the Genome Analysis Toolkit (v4)67 were used to analyze the exome/target-capture sequencing data. Sequenced reads were aligned to the GRCh38/hg38 reference human genome, and the filtering of variants was performed as described in previous studies68–70.
To prioritize the potential causal genes, family-based gene burden tests were performed on the rare loss-of-function variants of the 48 CFM families, and the promising variants were screened in the prioritized genes of each family according to inheritance patterns. The detailed steps were as follows: Firstly, the 273,164 high-quality variants were annotated by ANNOVAR software (Version: 2018Apr16)71, and the loss-of-function variants including splicing-site variants, missense variants, stop-gain- and stop-loss- variants, and frameshift variants were retained. Secondly, the variants with minor allele frequency less than 0.001 in the Genome Aggregation Database (gnomAD v3.1.2) were kept considering CFM is a rare congenital disorder. Thirdly, family-based burden tests were performed on the rare loss-of-function variants of the enrolled 48 families using Pedgene software (v3.6)72. The genes with burden P-values less than 0.001 were selected as candidates for subsequent analysis, and the detailed burden test statistics were shown in Supplementary Table 2. Fourthly, the variants within candidate genes were screened according to the inheritance pattern including autosomal dominant, autosomal recessive, de novo, or compound heterozygous. Fifthly, the effect of each variant identified from the above step was evaluated by SIFT (http://provean.jcvi.org/index.php), Polyphen-2 (http://genetics.bwh.harvard.edu/pph2/), and CADD (https://cadd.gs.washington.edu/) software. The variants predicted as “deleterious” or “probably damaging” or with CADD score larger than 10 were considered pathogenic ones. In addition, the functional effects on protein of the remained candidate single amino acid substitutions were predicted by SNAP2 software (v3.0, https://rostlab.org/services/snap2web/).
In silico analysis
To assess the conservation of the mutated sites, multiple alignments were performed on sequences of FOXI3 among different species. Then the nuclear localization sequence (NLS) of FOXI3 was predicted by NucPred software (https://nucpred.bioinfo.se/cgi-bin/single.cgi) and the effect of the variants within NLS was evaluated by NLStradamus software (version: r.9, http://www.moseslab.csb.utoronto.ca/NLStradamus/) with default parameters.
To further predict the effect of the missense variants located in the conserved FHD or NLS on the three-dimensional (3D) structure, the sequence from 135 to 243 amino acid residues of human FOXI3 was primarily mapped to the other members of FOX family with known crystal structure referred to PDB database (https://www.rcsb.org/), including HNF-3 (1VTN), FOXA2 (5×07), FOXC2 (6AKO), Foxk1A (2C6Y), and FOXO1 (4LG0); and a phylogenetic tree of these proteins was then constructed by MEGA-X software (Version:10.0.5) (Supplementary Fig. 5A). Based on the phylogenetic tree and homologous evaluation, the crystal structures of 6AKO and 2C6Y were used to model the homologous sequence of FOXI3 by Modeller software (Version:9.25) (Supplementary Fig. 5B, 5C). The modeled 3D-structure of mutant proteins was presented through Mutagenesis function in UCSF Chimera software (Version:1.16) (Supplementary Fig. 5E). In addition, the crystal structures of the monomer and protein-DNA complex of 6AKO were used to mimic the effect of variants on DNA-binding for FOXI3 by FoldX software (Version:5.0). The prediction of free energy changes of each variant on monomer and protein-DNA complex was replicated 10 times with default parameters. The consequence of the variant on the protein was classified as “destabilize fold” when the difference of free energy (ΔΔG) > 1.6 kcal/mol for the monomer protein between wild type and mutant. When the monomer ΔΔG ≤ 1.6 kcal/mol, and the difference between the ΔΔG for the protein-DNA complex and the monomer >0.25 kcal/mol, the consequence of variants on the protein was classified as “perturb protein-DNA interaction” (Supplementary Fig. 5D).
Sanger sequencing
Sanger sequencing was performed to validate or to discover variants in FOXI3. The primer design for the two exons of FOXI3 was done using Primer3Web (Version: 4.1.0). PCR amplification of FOXI3 exon 1 was performed on each sample in 50 µl reaction using 100 ng DNA, PCR primers (FOXI3_exon1_F1: ATGGAAATCCCAGCTCACAC; FOXI3_exon1_R1: TCATTCGTACTGTTCGGGTTA), the KAPA2G Robust HotStart PCR Kit (KK5522, Sigma-Aldrich, St. Louis, MO, USA) using KAPA Enhancer 1 and KAPA2G GC Buffer. The exon1 PCR reaction conditions were: 5 min at 95 °C, 40 cycles (15 s at 95 °C, 10 s at 60 °C, 3 min at 72 °C) and 2 min at 72 °C. PCR amplification of FOXI3 exon 2 was performed on each sample in a 50 µl reaction using 50 ng DNA and the JumpStart™ REDTaq® DNA-Polymerase Kit (D8187-50UN, Merck, Kenilworth, NJ, USA) and PCR primers (FOXI3_exon2_F2: CAGGTGTGCATACTCAAGTCT; FOXI3_exon2_R2: AGGTCCTTCTCTAACTTGGTCTT). The exon 2 PCR reaction conditions were: 2 min at 94 °C, 35 cycles (60 s at 95 °C, 30 s at 58 °C, 5 min at 72 °C) and 2 min at 72 °C.
PCR was performed in 96-well plates (Biometra, Jena, Germany). Primers and dNTPs were digested prior to sequencing by enzymatic treatment using Shrimp Alkaline Phosphatase (M9910, Promega, WI, USA) and Exonuclease I (M0293, New England Biolabs, Ipswich, Madison, MA, USA). About 1.55 µl dH2O, 3U Exonuclease I and 6U SAP were added to 10 µl PCR product, incubated at 37 °C for 30 min, then at 80 °C for 15 min.
Sequencing reactions were conducted using Applied Biosystems Big Dye Terminator chemistry (4337455, Applied Biosystems, Carlsbad, CA, USA), and the products were resolved on the ABI Prism 3730XL DNA Analyzers (Applied Biosystems, Carlsbad, CA, USA). All the results were analyzed and visualized by Chromas software (Version: 2.6.5).
Genome sequence
The genome sequence was performed in the accredited laboratory of Novagene (https://en.novogene.com/). A total of 0.5 μg of genomic DNA per sample was used for library preparation. Sequencing libraries were generated using the TruSeq Nano DNA HT Sample Prep Kit (20015965, Illumina Inc., CA, USA) following manufacturer’s recommendations. Genomic DNA was fragmented by sonication to a median size of 350 bp. Whole Genome Sequence libraries were sequenced on the Illumina HiSeq X TEN platform (2 × 150-bp paired-end reads) to a depth of 30X. Data were analyzed using the Saphetor computational platform (https://saphetor.com).
Bionano optical mapping
Fibroblasts of the FFF006-2013 case19 have been obtained from the biorepository of the Istituto Giannina Gaslini (www.biobanknetwork.telethon.it) in Genova and cultured in RPMI 1640 (72400047, Gibco, Thermo Fisher, Paisley, Scotland, UK) supplemented with 2% L-Glutamine (25030149, Gibco, Thermo Fisher, Paisley, Scotland, UK) 15% fetal bovine serum (10100147, Gibco, Thermo Fisher, Paisley, Scotland, UK) and 1% penicillin/streptomycin/fungizone mix (Amimed, BioConcept, Allschwill, Switzerland) at 37 °C in a 5% CO2 atmosphere to reach 1.5 million cells. High molecular weight was prepared by the Bionano genomics (www.bionanogenomics.com). Labeling of the DNA was done using the DNA Bionano labeling kit; linearization and imaging were done using the manufacturer’s procedure of the Saphyr Chip in one of the flowcells. The computational analysis and visualization of the data were done with the Analysis Bionano Solve software (Version: 3.3.1).
PacBio long-read sequence
PacBio circular consensus sequencing (CCS) was used for long-read sequencing targeted to the FOXI3 locus in 20 CFM probands. Briefly, a ~7 kb region encompassing the entirety of the FOXI3 locus was targeted for PCR amplification, followed by ligation to SMRTbell adapters to create circularized molecules from dsDNA. DNA polymerase and primers are annealed to circularized molecules to generate 1000 s of reads from a single molecule. Adapters are removed from reads to generate subreads, and subreads are used to discern consensus sequence of the region with >99% accuracy for further analysis. Bam files were viewed in IGV and cross-referenced with variant call files to analyze SNV and indel variants found by PacBio CCS. The 9 bp duplication found in BAB9863 from Baylor College of Medicine was Sanger sequenced for orthogonal validation. Cloning was performed using TOP10 competent cells and standard cloning procedure to isolate alleles and confirm in cis vs in trans inheritance of the indel variant with non-coding variants, as well as to confirm 9-bp in-frame duplication.
Cell culture
HEK-293T cell line (1101HUM-PUMC000091) was obtained from the Cell Resource Center, Peking Union Medical College (which is the headquarter of National Science & Technology infrastructure—National BioMedical Cell-Line Resource, NSTI-BMCR). The HEK-293T cells were maintained in DMEM (Gibco Laboratories, NY, USA) supplemented with 10% FBS (Gibco Laboratories, NY, USA) in a humidified incubator at 37 °C with 5% CO2.
Plasmid constructs
The entire coding sequence of FOXI3 was cloned into the pIRES2-EGFP eukaryotic overexpression vector (GV146, Genechem Co., Ltd., Shanghai, China). The mutants of FOXI3 were generated using the Q5 Site-Directed Mutagenesis Kit (E0554, New England BioLabs, Beverly, MA, USA) and verified by Sanger sequencing. The AE4 promoter sequence was cloned into the luciferase vector (GV238, Genechem Co., Ltd., Shanghai, China). The wild type and mutant variants of FOXI3 were cloned into the pEGFP-N1 vector (Clontech Laboratories, Inc., Palo Alto, CA, USA) to construct a C-terminus EGFP fused FOXI3 expression vector, respectively. Endotoxin-free plasmid was prepared using EndoFree Maxi Plasmid Kit (4992194, Tiangen, Shanghai, China) according to the manufacturer’s instructions.
Luciferase assay
To perform luciferase assays, HEK-293T cells were seeded in 24-well plates and allowed to reach 80% confluence. Then, the cells were transfected with 0.25 μg of FOXI3 expression vectors, 0.25 μg of AE4 promoter vectors, and 0.01 μg of pRL-TK vectors using Lipofectamine 3000 transfection reagent (L3000008, Thermo Fisher, Paisley, Scotland, UK). Twenty-four hours after transfection, the cells were harvested and lysed, and the fluorescence intensity was measured by the Dual-Luciferase assay kit (E1910, Promega Corporation, Madison, WI, USA) according to the manufacturer’s instructions.
Confocal analysis
The HEK-293T cells were cultured in 20 mm glass bottom confocal dishes (801001, Nest Scientific, Wuxi, Jiangsu, China) until they reached 80% confluence. Then, the cells were transfected with 1.5 μg FOXI3-EGFP or mutant FOXI3-EGFP expression vectors using Lipofectamine 3000 transfection reagent (L3000008, Thermo Fisher, Paisley, Scotland, UK) according to the transfection protocol. Twenty-four hours after transfection, the Hoechst 33342 (Beyotime Institute of Biotechnology, Haimen, China) was added to the medium. After incubation for 20 min, the cells were washed once with PBS. The high-speed confocal platform Dragonfly 200 (Andor Technology, Belfast, UK) and Fusion v2.3.0.36 (Andor Technology, Belfast, UK) were used to acquire and analyze the images.
Western-blot assay
Twenty-four hours after transfection, the HEK-293T cells were harvested to separate the proteins into cytoplasmic and nuclear proteins by the Nuclear and Cytoplasmic Protein Extraction Kit (P0027, Beyotime Institute of Biotechnology, Haimen, China) according to the manufacturer’s instructions. The protein concentrations of the cytoplasmic and nuclear constituents were determined by the BCA Protein Assay Kit (P0010, Beyotime Institute of Biotechnology, Haimen, China). The cytoplasmic and nuclear proteins were then separated by 4–20% Tris-Gly gel (GSG2001-420F, WSHT, Shanghai, China) and transferred to PVDF membranes (IPVH0010, Millipore, Billerica, MA, USA), respectively. After blocking with 5% w/v nonfat milk, the membranes were incubated with specific primary antibodies overnight at 4 °C. Subsequently, the membranes were incubated with HRP-conjugated secondary antibodies at room temperature for 1 h. Blots were visualized by chemiluminescence reagent (WBKLS0100, Millipore, Billerica, MA, USA). The primary antibodies is anti-EGFP (ab184601, Abcam, Cambridge, UK; 1:1000 dilution), and secondary antibody is Goat ani mouse lgG(H + L)-HRP (P03S01L, Gene-Protein Link Biotech,Beijing, China; 1:10,000 dilution); nuclear protein internal control is anti-H3 (ab1791, Abcam, Cambridge, UK; 1:5000 dilution) and secondary antibody is Goat anti-Rabbit lgG(H + L)-HRP (P03S02L, Gene-Protein Link Biotech,Beijing, China ;1:10,000 dilution); cytoplasmic protein internal control is anti-GAPDH (ab8245, Abcam, Cambridge, UK; 1:5000 dilution) and secondary antibody is Goat ani mouse lgG(H + L)-HRP (P03S01L, Gene-Protein Link Biotech,Beijing, China ;1:10,000 dilution). The band density was quantified by ImageJ software v1.8.0.345 (National Institutes of Health, Bethesda, Maryland, USA). The uncropped and unprocessed scans of the western blots were provided in the Source Data file.
Constructing Foxi3 variant mouse models using CRISPR
Generation of triple variant mice
C57BL/6 mice carrying the three variants p.(Arg220Trp), p.(Arg222Gln), and p.(Arg224His), orthologous to FOXI3:p.(Arg236Trp), p.(Arg238Gln) and p.(Arg240His) in one allele were generated by Cas9/CRISPR-mediated genome editing (Cyagen Biosciences, Guangzhou, China). Briefly, a gRNA (TCGGAAGCGAAGGCGCCGCG-CGG) to the mouse Foxi3 gene, a donor oligo with a targeting sequence containing p.(Arg220Trp) (codon from CGG to TGG), p.(Arg222Gln) (codon from CGA to CAA) and p.(Arg224His) (codon from CGC to CAC) mutations sites, and Cas9 protein were co-injected into fertilized mouse eggs to generate targeted knock-in offspring. The genotype of pups was determined by Sanger sequencing using 5′-CATGCACTAAAGTGTCTTGGAACAT-3′ and 5′-TTAAAGGTGCTGAGGAAAGTGTTG-3′ as PCR primer and 5′-GTTGAGACACGGAGTGGAGG-3′ as sequencing primer. Heterozygous mice Foxi3R220W-R222Q-R224H/+ were mated with Foxi3+/+ C57BL/6 mice to acquire heterozygous progenies, which were then intercrossed to generate homozygous Foxi3R220W-R222Q-R224H/R220W-R222Q-R224H mice.
Generation of Foxi3R224H/+ mice
The generation and genotyping of Foxi3R224H/+ mice were consistent with the above methods except for the donor oligonucleotide containing the variant of p.(Arg224His) (codon from CGC to CAC).
Generation of Foxi3F218L/+ mice
C57BL/6 mice carrying the variant p.(Phe218Leu), orthologous to FOXI3:p.(Phe233Leu) were generated by Cas9/CRISPR-mediated genome editing. A gRNA (CGCGGCGCCTTCGCTTCCGA-CGG) to the mouse Foxi3 gene, a 135 bp donor oligonucleotide flanking the targeted site and containing p.(Phe218Leu) (codon from TTC to CTG), and Cas9 protein were co-injected into fertilized mouse eggs to generate targeted knock-in offspring. Correct targeting and absence of indels were verified by Sanger sequencing of exon 2 of Foxi3 containing the modified allele. Genotyping was performed using 5′-CAGAGAAACCCTGCCTCGAA-3′ and 5′-AGGAGGCAGTACTCTTGGTG-3′ to generate a 368 bp product. The presence of the modified Foxi3 allele was revealed by digesting the amplified PCR band with MwoI to produce two bands of 187 and 181 bp; the wild-type band was not cut by MwoI and ran at 368 bp. Heterozygous Foxi3F218L/+ mice were crossed with wild type mice for several generations, and the heterozygous offspring intercrossed to generate homozygous Foxi3F218L/F218L mice. Homozygous mice were mated with Foxi3 null mice20,22 (Foxi3+/-) to generate Foxi3F218L/- mice.
All animals used in this study were bred and maintained under specific pathogen-free conditions with a temperature of 18–24 °C and humidity of 35–60% and 12-h light/dark cycle and free access to food and water.
Phenotypic analysis of mouse models
Skeletal staining
Newborn mice were euthanized, stored in PBS on ice, and then scalded the fetuses in hot tap water (70 °C) for approximately 15 s. The skin was carefully peeled off and all the internal organs were cut away with forceps. The bodies were fixed in 95% ethanol for 24 h and stained with Alizarin Red and Alcian Blue as described in previous studies73 and photographed with bright field microscopy.
Micro-computed tomography (Micro-CT) analysis of skull tissue
Newborn mice were euthanized, fixed in 4% PFA and scanned with a photon counting detector-based spectral Micro-CT developed by the Institute of High Energy Physics, Chinese Academy of Sciences. The parameters are used as follows: 20 μm resolution, 50kVp, 144mAs without a filter. Raw images from all scans were reconstructed using the FDK algorithm. Reconstructed scan data were imported into VG Studio software version 3.1(Volume Graphics, Inc., NC, USA) and Drishti Volume Exploration Software74 for 3D rendering of bone and soft tissue.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Supplementary information
Acknowledgements
We thank the patients and their families for their participation in the study. The study was partially supported by the National Natural Science Foundation of China (82171844 and 81970898 to Y.-B.Z., 81701930 to B.Q.W.); the ChildCare foundation and an ERC grant 249968 to S.E.A.; the United States National Institutes of Health: NINDS R35NS105078; NHGRI/NHLBI UM1HG006542; NHGRI U01HG011758 to J.R.L.; RO1 DC013072 to A.K.G; the Italian Ministry of Health to G.M. We thank Domenico Coviello, Eleonora Di Venere, Annalisa Paparella, Fritz Sedlazeck, Georg Papoutsoglou, Andy Pang, Jill Lai, Diogo Ribeiro, Olivier Delaneau for providing material and assistance in this study.
Source data
Author contributions
S.E.A. and Y-B.Z. designed and supervised the study. B.W., Q.L., Q.C., S.Z., and P.C. recruited case samples. Ke.M., C.B., M.A., T.C.C., X.X., X.W., A.J., and F.J. planned and conducted laboratory experiments. A.K.G., A.J., H.A., S.S., Ho.Z., H.S., Ke.M. performed experiments related to Foxi3–/– and Foxi3218/+ mice. Ke.M., Ha.Z., C.B, J.I., V.Z., X.B., and M.A., analyzed data, and performed bioinformatics, statistical, and 3D structural analyses. S.E.A., Ke.M., Y-B.Z., A.K.G., T.C.C., J.R.L., Q.Z. drafted and revised the manuscript. P.M., C.F., E.R., S.P., S.F.S., V.G., F.P.-S., D.M., B. D., M.D., A.N., Mo.M., M.F., Ker.M., R.A.L., G.S., P.P., Gio.M., G.G., Mi.M., A.S., S.P., H.F., S.D., S.M., S.B., A.D.L., Giu.M., and J.R.L. ascertained and diagnosed cases, collected samples, and made critical comments on the manuscript. All authors have reviewed and contributed to the manuscript.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Data availability
The likely pathogenic FOXI3 variants described in this study have been submitted to the ClinVar database (https://www.ncbi.nlm.nih.gov/clinvar/) and their accession numbers are SCV003803092-SCV003803100 and SCV003806728-SCV003806732. The raw datasets (FASTQ files) supporting this study have not been deposited in a public repository because of confidentiality of medical tests; however, VCF files for selected genes are available under restricted access from S.E.A. (stylianos.antonarakis@unige.ch) or Y.-B.Z. (zhangyongbiao@buaa.edu.cn) upon request. The access to these data requires a short description of the project, and signing a data-use agreement that includes restrictions on downstream data, and use of the data only by the requesting investigator. There is no authorship requirement for a reanalysis of the data unless there is an extensive follow-up collaboration and input from the authors of this paper. The response timeframe will be less than 3 weeks. The mouse models described here are also available upon request from Y.-B.Z. (zhangyongbiao@buaa.edu.cn) or A.K.G. (akgroves@bcm.edu). Frequency data of selected FOXI3 variants in different human populations have been obtained from gnomAD (https://gnomad.broadinstitute.org/). Additional datasets required to reproduce the analysis and figures are in Source data. Source data are provided with this paper. GRCh38/hg38 reference human genome (https://hgdownload.soe.ucsc.edu/goldenPath/hs1/vsHg38/); Publicly available protein structures were obtained from the Protein Data Bank (https://www.rcsb.org/). All data supporting the findings described in this manuscript are available in the article and its Supplementary Information files, and from the corresponding authors upon request. Source data are provided with this paper.
Competing interests
S.E.A. is a cofounder and CEO of Medigenome, and serves on the Scientific Advisory Board of the “Imaging” institute in Paris; J.R.L. has stock ownership in 23andMe, is a paid consultant for Regeneron Genetics Center, is a co-inventor on multiple United States and European patents related to molecular diagnostics for inherited neuropathies, eye diseases, and bacterial genomic fingerprinting, and serves on the Scientific Advisory Board of BG. The Department of Molecular and Human Genetics at Baylor College of Medicine receives revenue from clinical genetic and genomic testing conducted at Baylor Genetics (BG) Laboratories. The remaining authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Ke Mao, Christelle Borel, Muhammad Ansar.
Contributor Information
Qingguo Zhang, Email: plastic135@126.com.
Yong-Biao Zhang, Email: zhangyongbiao@buaa.edu.cn.
Stylianos E. Antonarakis, Email: stylianos.antonarakis@unige.ch
Supplementary information
The online version contains supplementary material available at 10.1038/s41467-023-37703-6.
References
- 1.Klein D. Living history–autobiography: genetics and environment from a personal perspective. Am. J. Med. Genet. 1990;37:325–335. doi: 10.1002/ajmg.1320370307. [DOI] [PubMed] [Google Scholar]
- 2.Goldenhar M. Associations malformatives de l’oeil et de l’oreille, en particulier le svndrome dermoide epibulbaire- appendices auriculaires-fistula auris congenita et ses relations avec la dysostose mandibulo-faciale. J. Genet. Hum. 1952;1:243–282. [Google Scholar]
- 3.Cohen MM, Jr., Rollnick BR, Kaye CI. Oculoauriculovertebral spectrum: an updated critique. Cleft Palate J. 1989;26:276–286. [PubMed] [Google Scholar]
- 4.Beleza-Meireles A, Clayton-Smith J, Saraiva JM, Tassabehji M. Oculo-auriculo-vertebral spectrum: a review of the literature and genetic update. J. Med. Genet. 2014;51:635–645. doi: 10.1136/jmedgenet-2014-102476. [DOI] [PubMed] [Google Scholar]
- 5.Mastroiacovo P, et al. Epidemiology and genetics of microtia-anotia: a registry based study on over one million births. J. Med. Genet. 1995;32:453–457. doi: 10.1136/jmg.32.6.453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shirazi M, Abbariki E, Pirjani R, Akhavan S, Dastgerdy E. Congenital microtia in a neonate due to maternal isotretinoin exposure 1 month before pregnancy: case report. J. Obstet. Gynaecol. Res. 2015;41:975–978. doi: 10.1111/jog.12644. [DOI] [PubMed] [Google Scholar]
- 7.Adam AP, et al. Recurrent constellations of embryonic malformations re-conceptualized as an overlapping group of disorders with shared pathogenesis. Am. J. Med. Genet. A. 2020;182:2646–2661. doi: 10.1002/ajmg.a.61847. [DOI] [PubMed] [Google Scholar]
- 8.Artunduaga MA, et al. A classic twin study of external ear malformations, including microtia. N. Engl. J. Med. 2009;361:1216–1218. doi: 10.1056/NEJMc0902556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tingaud-Sequeira A, et al. A recurrent missense variant in EYA3 gene is associated with oculo-auriculo-vertebral spectrum. Hum. Genet. 2021;140:933–944. doi: 10.1007/s00439-021-02255-6. [DOI] [PubMed] [Google Scholar]
- 10.Lopez E, et al. Mutations in MYT1, encoding the myelin transcription factor 1, are a rare cause of OAVS. J. Med. Genet. 2016;53:752–760. doi: 10.1136/jmedgenet-2016-103774. [DOI] [PubMed] [Google Scholar]
- 11.Tingaud-Sequeira A, et al. Functional and genetic analyses of ZYG11B provide evidences for its involvement in OAVS. Mol. Genet. Genom. Med. 2020;8:e1375. doi: 10.1002/mgg3.1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rengasamy Venugopalan S, et al. A novel nonsense substitution identified in the AMIGO2 gene in an Occulo-Auriculo-Vertebral spectrum patient. Orthod. Craniofac. Res. 2019;22 Suppl 1:163–167. doi: 10.1111/ocr.12259. [DOI] [PubMed] [Google Scholar]
- 13.Wang Y, et al. A mutation in VWA1, encoding von willebrand factor a domain-containing protein 1, is associated with hemifacial microsomia. Front. Cell Dev. Biol. 2020;8:571004. doi: 10.3389/fcell.2020.571004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Timberlake AT, et al. Haploinsufficiency of SF3B2 causes craniofacial microsomia. Nat. Commun. 2021;12:4680. doi: 10.1038/s41467-021-24852-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Xu X, et al. Novel risk factors for craniofacial microsomia and assessment of their utility in clinic diagnosis. Hum. Mol. Genet. 2021;30:1045–1056. doi: 10.1093/hmg/ddab055. [DOI] [PubMed] [Google Scholar]
- 16.Zhang YB, et al. Genome-wide association study identifies multiple susceptibility loci for craniofacial microsomia. Nat. Commun. 2016;7:10605. doi: 10.1038/ncomms10605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Quiat D, et al. An ancient founder mutation located between ROBO1 and ROBO2 is responsible for increased microtia risk in Amerindigenous populations. Proc. Natl Acad. Sci. USA. 2022;119:e2203928119. doi: 10.1073/pnas.2203928119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ansar M, et al. Bi-allelic loss-of-function variants in DNMBP cause infantile cataracts. Am. J. Hum. Genet. 2018;103:568–578. doi: 10.1016/j.ajhg.2018.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tassano E, et al. Congenital aural atresia associated with agenesis of internal carotid artery in a girl with a FOXI3 deletion. Am. J. Med. Genet. A. 2015;167A:537–544. doi: 10.1002/ajmg.a.36895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gutierrez-Arcelus M, et al. Passive and active DNA methylation and the interplay with genetic variation in gene regulation. Elife. 2013;2:e00523. doi: 10.7554/eLife.00523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lappalainen T, et al. Transcriptome and genome sequencing uncovers functional variation in humans. Nature. 2013;501:506–511. doi: 10.1038/nature12531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Consortium GT, et al. Genetic effects on gene expression across human tissues. Nature. 2017;550:204–213. doi: 10.1038/nature24277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Taliun D, et al. Sequencing of 53,831 diverse genomes from the NHLBI TOPMed Program. Nature. 2021;590:290–299. doi: 10.1038/s41586-021-03205-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen X, et al. Structural basis for DNA recognition by FOXC2. Nucleic Acids Res. 2019;47:3752–3764. doi: 10.1093/nar/gkz077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tsai KL, et al. Crystal structure of the human FOXK1a-DNA complex and its implications on the diverse binding specificity of winged helix/forkhead proteins. J. Biol. Chem. 2006;281:17400–17409. doi: 10.1074/jbc.M600478200. [DOI] [PubMed] [Google Scholar]
- 26.Schymkowitz J, et al. The FoldX web server: an online force field. Nucleic Acids Res. 2005;33:W382–W388. doi: 10.1093/nar/gki387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Youssoufian H, et al. Recurrent mutations in haemophilia A give evidence for CpG mutation hotspots. Nature. 1986;324:380–382. doi: 10.1038/324380a0. [DOI] [PubMed] [Google Scholar]
- 28.Kurth I, et al. The forkhead transcription factor Foxi1 directly activates the AE4 promoter. Biochem J. 2006;393:277–283. doi: 10.1042/BJ20051094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Singh S, Jangid RK, Crowder A, Groves AK. Foxi3 transcription factor activity is mediated by a C-terminal transactivation domain and regulated by the protein phosphatase 2A (PP2A) complex. Sci. Rep. 2018;8:17249. doi: 10.1038/s41598-018-35390-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Edlund RK, Ohyama T, Kantarci H, Riley BB, Groves AK. Foxi transcription factors promote pharyngeal arch development by regulating formation of FGF signaling centers. Dev. Biol. 2014;390:1–13. doi: 10.1016/j.ydbio.2014.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Birol O, et al. The mouse Foxi3 transcription factor is necessary for the development of posterior placodes. Dev. Biol. 2016;409:139–151. doi: 10.1016/j.ydbio.2015.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hasten E, Morrow BE. Tbx1 and Foxi3 genetically interact in the pharyngeal pouch endoderm in a mouse model for 22q11.2 deletion syndrome. PLoS Genet. 2019;15:e1008301. doi: 10.1371/journal.pgen.1008301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rieder MJ, et al. A human homeotic transformation resulting from mutations in PLCB4 and GNAI3 causes auriculocondylar syndrome. Am. J. Hum. Genet. 2012;90:907–914. doi: 10.1016/j.ajhg.2012.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Herman L, Todeschini AL, Veitia RA. Forkhead transcription factors in health and disease. Trends Genet. 2021;37:460–475. doi: 10.1016/j.tig.2020.11.003. [DOI] [PubMed] [Google Scholar]
- 35.Golson ML, Kaestner KH. Fox transcription factors: from development to disease. Development. 2016;143:4558–4570. doi: 10.1242/dev.112672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dai S, Qu L, Li J, Chen Y. Toward a mechanistic understanding of DNA binding by forkhead transcription factors and its perturbation by pathogenic mutations. Nucleic Acids Res. 2021;49:10235–10249. doi: 10.1093/nar/gkab807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hannenhalli S, Kaestner KH. The evolution of Fox genes and their role in development and disease. Nat. Rev. Genet. 2009;10:233–240. doi: 10.1038/nrg2523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jackson BC, Carpenter C, Nebert DW, Vasiliou V. Update of human and mouse forkhead box (FOX) gene families. Hum. Genom. 2010;4:345–352. doi: 10.1186/1479-7364-4-5-345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Benayoun BA, Caburet S, Veitia RA. Forkhead transcription factors: key players in health and disease. Trends Genet. 2011;27:224–232. doi: 10.1016/j.tig.2011.03.003. [DOI] [PubMed] [Google Scholar]
- 40.Yang T, et al. Transcriptional control of SLC26A4 is involved in Pendred syndrome and nonsyndromic enlargement of vestibular aqueduct (DFNB4) Am. J. Hum. Genet. 2007;80:1055–1063. doi: 10.1086/518314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Borel C, et al. Biased allelic expression in human primary fibroblast single cells. Am. J. Hum. Genet. 2015;96:70–80. doi: 10.1016/j.ajhg.2014.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wu N, et al. TBX6 null variants and a common hypomorphic allele in congenital scoliosis. N. Engl. J. Med. 2015;372:341–350. doi: 10.1056/NEJMoa1406829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Albers CA, et al. Compound inheritance of a low-frequency regulatory SNP and a rare null mutation in exon-junction complex subunit RBM8A causes TAR syndrome. Nat. Genet. 2012;44:435–439. doi: 10.1038/ng.1083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Szafranski P, et al. Small noncoding differentially methylated copy-number variants, including lncRNA genes, cause a lethal lung developmental disorder. Genome Res. 2013;23:23–33. doi: 10.1101/gr.141887.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Consortium GT. The GTEx Consortium atlas of genetic regulatory effects across human tissues. Science. 2020;369:1318–1330. doi: 10.1126/science.aaz1776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kajiwara K, Berson EL, Dryja TP. Digenic retinitis pigmentosa due to mutations at the unlinked peripherin/RDS and ROM1 loci. Science. 1994;264:1604–1608. doi: 10.1126/science.8202715. [DOI] [PubMed] [Google Scholar]
- 47.Katsanis N, et al. Triallelic inheritance in Bardet-Biedl syndrome, a Mendelian recessive disorder. Science. 2001;293:2256–2259. doi: 10.1126/science.1063525. [DOI] [PubMed] [Google Scholar]
- 48.Cavenee WK, et al. Expression of recessive alleles by chromosomal mechanisms in retinoblastoma. Nature. 1983;305:779–784. doi: 10.1038/305779a0. [DOI] [PubMed] [Google Scholar]
- 49.Snellings DA, et al. Somatic mutations in vascular malformations of hereditary hemorrhagic telangiectasia result in bi-allelic loss of ENG or ACVRL1. Am. J. Hum. Genet. 2019;105:894–906. doi: 10.1016/j.ajhg.2019.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.van den Boogaard ML, et al. Mutations in DNMT3B modify epigenetic repression of the D4Z4 repeat and the penetrance of facioscapulohumeral dystrophy. Am. J. Hum. Genet. 2016;98:1020–1029. doi: 10.1016/j.ajhg.2016.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gouya L, et al. The penetrance of dominant erythropoietic protoporphyria is modulated by expression of wildtype FECH. Nat. Genet. 2002;30:27–28. doi: 10.1038/ng809. [DOI] [PubMed] [Google Scholar]
- 52.Drogemuller C, et al. A mutation in hairless dogs implicates FOXI3 in ectodermal development. Science. 2008;321:1462. doi: 10.1126/science.1162525. [DOI] [PubMed] [Google Scholar]
- 53.Jussila M, et al. Suppression of epithelial differentiation by Foxi3 is essential for molar crown patterning. Development. 2015;142:3954–3963. doi: 10.1242/dev.124172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Shirokova V, et al. Foxi3 deficiency compromises hair follicle stem cell specification and activation. Stem Cells. 2016;34:1896–1908. doi: 10.1002/stem.2363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nagel RL, et al. Hematologically and genetically distinct forms of sickle cell anemia in Africa. The Senegal type and the Benin type. N. Engl. J. Med. 1985;312:880–884. doi: 10.1056/NEJM198504043121403. [DOI] [PubMed] [Google Scholar]
- 56.Monk D, Mackay DJG, Eggermann T, Maher ER, Riccio A. Genomic imprinting disorders: lessons on how genome, epigenome and environment interact. Nat. Rev. Genet. 2019;20:235–248. doi: 10.1038/s41576-018-0092-0. [DOI] [PubMed] [Google Scholar]
- 57.Santoni FA, et al. Detection of imprinted genes by single-cell allele-specific gene expression. Am. J. Hum. Genet. 2017;100:444–453. doi: 10.1016/j.ajhg.2017.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Afzelius BA. A human syndrome caused by immotile cilia. Science. 1976;193:317–319. doi: 10.1126/science.1084576. [DOI] [PubMed] [Google Scholar]
- 59.Al-Qattan MM, Shamseldin HE, Salih MA, Alkuraya FS. GLI3-related polydactyly: a review. Clin. Genet. 2017;92:457–466. doi: 10.1111/cge.12952. [DOI] [PubMed] [Google Scholar]
- 60.Cacheiro P, Haendel MA, Smedley D. International mouse phenotyping, C. & the Monarch, I. New models for human disease from the International Mouse Phenotyping Consortium. Mamm. Genome. 2019;30:143–150. doi: 10.1007/s00335-019-09804-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bernstock JD, et al. Recurrent microdeletions at chromosome 2p11.2 are associated with thymic hypoplasia and features resembling DiGeorge syndrome. J. Allergy Clin. Immunol. 2020;145:358–367.e2. doi: 10.1016/j.jaci.2019.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Khatri SB, Groves AK. Expression of the Foxi2 and Foxi3 transcription factors during development of chicken sensory placodes and pharyngeal arches. Gene Expr. Patterns. 2013;13:38–42. doi: 10.1016/j.gep.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Tambalo M, Anwar M, Ahmed M, Streit A. Enhancer activation by FGF signalling during otic induction. Dev. Biol. 2020;457:69–82. doi: 10.1016/j.ydbio.2019.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Anwar M, Tambalo M, Ranganathan R, Grocott T, Streit A. A gene network regulated by FGF signalling during ear development. Sci. Rep. 2017;7:6162. doi: 10.1038/s41598-017-05472-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Abe M, et al. GATA3 is essential for separating patterning domains during facial morphogenesis. Development. 2021;148:dev199534. doi: 10.1242/dev.199534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Li H, Durbin R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.DePristo MA, et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 2011;43:491–498. doi: 10.1038/ng.806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Makrythanasis P, et al. Diagnostic exome sequencing to elucidate the genetic basis of likely recessive disorders in consanguineous families. Hum. Mutat. 2014;35:1203–1210. doi: 10.1002/humu.22617. [DOI] [PubMed] [Google Scholar]
- 69.Ansar M, et al. Visual impairment and progressive phthisis bulbi caused by recessive pathogenic variant in MARK3. Hum. Mol. Genet. 2018;27:2703–2711. doi: 10.1093/hmg/ddy180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ansar M, et al. Biallelic variants in LINGO1 are associated with autosomal recessive intellectual disability, microcephaly, speech and motor delay. Genet. Med. 2018;20:778–784. doi: 10.1038/gim.2017.113. [DOI] [PubMed] [Google Scholar]
- 71.Wang K, Li M, Hakonarson H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010;38:e164. doi: 10.1093/nar/gkq603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Schaid DJ, McDonnell SK, Sinnwell JP, Thibodeau SN. Multiple genetic variant association testing by collapsing and kernel methods with pedigree or population structured data. Genet. Epidemiol. 2013;37:409–418. doi: 10.1002/gepi.21727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ovchinnikov D. Alcian blue/alizarin red staining of cartilage and bone in mouse. Cold Spring Harb. Protoc. 2009;2009:pdb prot5170. doi: 10.1101/pdb.prot5170. [DOI] [PubMed] [Google Scholar]
- 74.Hu Y, Limaye A, Lu J. Three-dimensional segmentation of computed tomography data using Drishti Paint: new tools and developments. R. Soc. Open Sci. 2020;7:201033. doi: 10.1098/rsos.201033. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The likely pathogenic FOXI3 variants described in this study have been submitted to the ClinVar database (https://www.ncbi.nlm.nih.gov/clinvar/) and their accession numbers are SCV003803092-SCV003803100 and SCV003806728-SCV003806732. The raw datasets (FASTQ files) supporting this study have not been deposited in a public repository because of confidentiality of medical tests; however, VCF files for selected genes are available under restricted access from S.E.A. (stylianos.antonarakis@unige.ch) or Y.-B.Z. (zhangyongbiao@buaa.edu.cn) upon request. The access to these data requires a short description of the project, and signing a data-use agreement that includes restrictions on downstream data, and use of the data only by the requesting investigator. There is no authorship requirement for a reanalysis of the data unless there is an extensive follow-up collaboration and input from the authors of this paper. The response timeframe will be less than 3 weeks. The mouse models described here are also available upon request from Y.-B.Z. (zhangyongbiao@buaa.edu.cn) or A.K.G. (akgroves@bcm.edu). Frequency data of selected FOXI3 variants in different human populations have been obtained from gnomAD (https://gnomad.broadinstitute.org/). Additional datasets required to reproduce the analysis and figures are in Source data. Source data are provided with this paper. GRCh38/hg38 reference human genome (https://hgdownload.soe.ucsc.edu/goldenPath/hs1/vsHg38/); Publicly available protein structures were obtained from the Protein Data Bank (https://www.rcsb.org/). All data supporting the findings described in this manuscript are available in the article and its Supplementary Information files, and from the corresponding authors upon request. Source data are provided with this paper.