Abstract
Background
Most women living in low‐ and middle‐income countries (LMICs) present with advanced‐stage breast cancer. Limitations of poor serviceable health systems, restricted access to treatment facilities, and lack of breast cancer screening programmes all likely contribute to the late presentation of women with breast cancer living in these countries. Women are diagnosed with advanced disease and frequently do not complete their care due to a number of factors, including financial reasons as health expenditure is largely out of pocket resulting in financial toxicity; health system failures, such as missing services or health worker lack of awareness on common signs and symptoms of cancer; and sociocultural barriers, such as stigma and use of alternative therapies. Clinical breast examination (CBE) is an inexpensive early detection technique for breast cancer in women with palpable breast masses. Training health workers from LMICs to conduct CBE has the potential to improve the quality of the technique and the ability of health workers to detect breast cancers early.
Objectives
To assess whether training in CBE affects the ability of health workers in LMICs to detect early breast cancer.
Search methods
We searched the Cochrane Breast Cancer Specialised Registry, CENTRAL, MEDLINE, Embase, the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) search portal, and ClinicalTrials.gov up to 17 July 2021.
Selection criteria
We included randomised controlled trials (RCTs) (including individual and cluster‐RCTs), quasi‐experimental studies and controlled before‐and‐after studies if they fulfilled the eligibility criteria.
Data collection and analysis
Two review authors independently screened studies for inclusion, and extracted data, assessed risk of bias, and assessed the certainty of the evidence using the GRADE approach. We performed statistical analysis using Review Manager software and presented the main findings of the review in a summary of findings table.
Main results
We included four RCTs that screened a total population of 947,190 women for breast cancer, out of which 593 breast cancers were diagnosed. All included studies were cluster‐RCTs; two were conducted in India, one in the Philippines, and one in Rwanda. Health workers trained to perform CBE in the included studies were primary health workers, nurses, midwives, and community health workers. Three of the four included studies reported on the primary outcome (breast cancer stage at the time of presentation). Amongst secondary outcomes, included studies reported CBE coverage, follow‐up, accuracy of health worker‐performed CBE, and breast cancer mortality. None of the included studies reported knowledge attitude practice (KAP) outcomes and cost‐effectiveness.
Three studies reported diagnosis of breast cancer at early stage (at stage 0+I+II), suggesting that training health workers in CBE may increase the number of women detected with breast cancer at an early stage compared to the non‐training group (45% detected versus 31% detected; risk ratio (RR) 1.44, 95% confidence interval (CI) 1.01 to 2.06; three studies; 593 participants; I2 = 0%; low‐certainty evidence).
Three studies reported diagnosis at late stage (III+IV) suggesting that training health workers in CBE may slightly reduce the number of women detected with breast cancer at late stage compared to the non‐training group (13% detected versus 42%, RR 0.58, 95% CI 0.36 to 0.94; three studies; 593 participants; I2 = 52%; low‐certainty evidence).
Regarding secondary outcomes, two studies reported breast cancer mortality, implying that the evidence is uncertain for the impact on breast cancer mortality (RR 0.88, 95% CI 0.24 to 3.26; two studies; 355 participants; I2 = 68%; very low‐certainty evidence). Due to the study heterogeneity, we could not conduct meta‐analysis for accuracy of health worker‐performed CBE, CBE coverage, and completion of follow‐up, and therefore reported narratively using the 'Synthesis without meta‐analysis' (SWiM) guideline. Sensitivity of health worker‐performed CBE was reported to be 53.2% and 51.7%; while specificity was reported to be 100% and 94.3% respectively in two included studies (very low‐certainty evidence). One trial reported CBE coverage with a mean adherence of 67.07% for the first four screening rounds (low‐certainty evidence). One trial reported follow‐up suggesting that compliance rates for diagnostic confirmation following a positive CBE were 68.29%, 71.20%, 78.84% and 79.98% during the respective first four rounds of screening in the intervention group compared to 90.88%, 82.96%, 79.56% and 80.39% during the respective four rounds of screening in the control group.
Authors' conclusions
Our review findings suggest some benefit of training health workers from LMICs in CBE on early detection of breast cancer. However, the evidence regarding mortality, accuracy of health worker‐performed CBE, and completion of follow up is uncertain and requires further evaluation.
Keywords: Female, Humans, Breast Neoplasms, Breast Neoplasms/diagnosis, Developing Countries, Early Detection of Cancer, Health Facilities, Health Personnel, Health Personnel/education
Plain language summary
Training health workers in breast examination for early detection of breast cancer in low‐ and middle‐income countries
What is the issue?
Limited screening services and inadequate health systems in low‐ and middle‐income countries (LMICs) leads to late diagnosis of breast cancer among women living in LMICs. Advanced screening for breast cancer (using mammography) is mostly unavailable in many health facilities and, if available, is too expensive for most women. This Cochrane Review examines whether training health workers based in LMICs in clinical breast examination (CBE) would have any effect on early detection of breast cancer in these settings.
Why does it matter?
Higher number of women die of breast cancer in LMICs compared to high‐income countries (HIC) despite the lower occurrence rate of breast cancer in HICs compared to LMICs. CBE is an inexpensive early detection technique for breast cancer and training health workers from LMICs to conduct CBE has the potential to improve early detection of breast cancers.
We asked?
Whether training health workers in CBE compared to no training has any effect on improving the detection of breast cancer at an early stage of the disease. We also assessed whether training of health workers in CBE has any effect on the accuracy in detecting breast cancer, impact of CBE on deaths due to breast cancer, and knowledge and uptake of CBE amongst women. We included studies published by 17 July 2021.
We found four studies that answered our research question. The CBE training was provided to health workers, nurses, midwives, and community health workers working in LMICs. A total population of 947,190 women were screened for breast cancer. Of the total population screened, 593 breast cancers were diagnosed, with more cancers diagnosed at an early stage by trained health workers than by health workers who were not trained. The results from these studies suggest that training health workers in CBE may increase breast cancer diagnosis at an early stage, but the existing evidence is of low quality. More research is needed to assess its impact on other outcomes, including how accurately CBE is performed, knowledge about CBE, uptake of CBE, and if CBE has any impact on deaths due to breast cancer.
This means that there is a potential to detect breast cancer at an early stage if health workers in LMICs are trained to perform CBE; however high‐quality studies are needed to answer this research question.
Summary of findings
Summary of findings 1. Summary of findings table 1.
| Clinical breast examination (CBE) training for health workers compared with no training for early detection of breast cancer in LMICs | ||||||
|
Patient or population: health workers Settings: community settings in LMICs Intervention: CBE training for health workers Comparison: standard of care (no CBE training for health works) | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| No training | CBE training | |||||
| Breast cancer stage at the time of presentation: defined as the proportion of women with breast cancer diagnosed at early stage (stage 0, I, II) Time of outcome assessment: 3 years to 20 years Method of measurement: CBE |
212 per 1000 |
132 per 1000 |
RR 1.44 (1.01 to 2.06) |
593 participants (3 studies) out of a total of 947,190 women screened in 4 studies | ⊕⊕⊝⊝ Lowa,b | |
| Breast cancer stage at the time of presentation: defined as the proportion of women with breast cancer diagnosed at late stage (stage III, IV) Time of outcome assessment: 3 years to 20 years Method of measurement: CBE |
418 per 1000 | 128 per 1000 | RR 0.58 (0.36 to 0.94) | 593 participants (3 studies) out of a total of 947,190 women screened in 4 studies | ⊕⊕⊝⊝ Lowa,b | |
| Accuracy of health worker‐performed CBE Time of outcome assessment: 2 years to 20 years Method of measurement: Sensitivity, specificity and positive predictive value |
‐ | ‐ | ‐ | 2 studies | ⊕⊝⊝⊝ Very lowa,c,d | The outcome could not be pooled. The sensitivity ranged between 51.7% (Sankaranarayanan 2011) to 53.2% (Pisani 2006); the specificity ranged between 94.3% (Sankaranarayanan 2011) to 100% (Pisani 2006) ; and the positive predictive value ranged between 1% (Sankaranarayanan 2011) to 1.2% (Pisani 2006). |
| CBE coverage Time of outcome assessment: 20 years Method of measurement: active surveillance |
‐ | ‐ | ‐ | 1 study | ⊕⊕⊕⊝ Moderatea | One study (Mittra 2021) reported mean adherence to the 4 screening rounds to be 67.07%. |
| Completion of follow‐up: defined as the proportion of women who completed follow‐up after screening and CBE and advice to complete follow‐up Time of outcome assessment: 20 years Method of measurement: active surveillance |
‐ | ‐ | ‐ | 1 study | ⊕⊕⊕⊝ Moderatea | One study (Mittra 2021) reported follow‐up defined as the proportion of women who completed follow‐up after screening and CBE and suggested that compliance rates for diagnostic confirmation following a positive CBE were 68.29%, 71.20%, 78.84%, and 79.98% during the 1st, 2nd, 3rd and 4th rounds of screening in the intervention group compared to 90.88%, 82.96%, 79.56% and 80.39% during the respective 4 rounds of screening in the control group. |
| Breast cancer mortality: defined as deaths among women who received a diagnosis of breast cancer Time of outcome assessment: 3 years to 20 years Method of measurement: death registration offices, hospital records, and house visits |
107 per 1000 | 122 per 1000 | RR 0.88 (0.24 to 3.26) | 355 participants (2 studies) | ⊕⊝⊝⊝ Very lowa,e, f |
|
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% CI) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CBE: clinical breast examination; CI: confidence interval; LMIC: low‐ and middle‐income country; RR: risk ratio. | ||||||
| GRADE Working Group grades of evidence High certainty: further research is very unlikely to change our confidence in the estimate of effect. Moderate certainty: further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low certainty: further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low certainty: we are very uncertain about the estimate. | ||||||
aDowngraded by one level due to study limitations. bDowngraded by one level due to small number of events. cDowngraded by one level due to heterogeneity in the intervention between the two studies. dDowngraded by one level due to indirectness. Neither of the two studies were designed to capture diagnostic accuracy. eDowngraded by one level due to inconsistent estimates from the two studies included in meta‐analysis. fDowngraded by one level due to high heterogeneity (I2 = 68%).
Background
Description of the condition
The worldwide incidence of breast cancer represents 11.7% of all known cancer cases, with a global incidence of 2.3 million female cases in 2020 (Sung 2021). According to the International Agency for Research on Cancer (IARC) GLOBOCAN 2020 database, incidence rates of breast cancer are lower in low‐ and middle‐income countries (LMICs) than in high‐income countries (HICs). However, both overall incidence (number of cases) and age‐specific incidence rates appear to be rising in LMICs. A modelling study obtained registry data on the incidence of breast and cervical cancer spanning a period of 30 years (1980 to 2010) and covering 187 countries, and reported that twice as many cases of breast cancer occurred in individuals aged 15 to 49 years in LMICs compared to the same population in HICs (Forouzanfar 2011). This may be attributed to population growth, increasing life expectancy, and a relative decrease in the burden of infectious diseases, or it may be the result of changes in lifestyle‐related risk factors due to urbanisation and economic development (Torre 2015). Deaths attributed to breast cancer were reported to be higher in LMICs than in HICs, with a 17% higher mortality rate (Sung 2021). Women in Northern Africa have a lower incidence of breast cancer than women in Northern America (age‐standardised rate (ASR) of 50.4 versus 89.4) but a higher mortality rate (ASR of 18.8 versus 12.5). One reason for this could be the advanced stage of disease at presentation. A summarised report across 17 sub‐Saharan African countries presented that 77% of staged cases were stage III/IV at diagnosis (Jedy‐Agba 2016). A study from Kenya reported that more than 60% of women with breast cancer received the diagnosis at disease stage III or IV (Sayed 2014). Similarly, 65% of breast cancer patients in a regional cancer centre in India presented with stage III and IV disease (Kumar 2019), as was the case in a Malaysian study where 59.6% of the cases presented with stages III and IV (17.3% for stage III and 42.3% for stage IV) (Li Cheng 2015). Most patients in the Indian Regional Cancer Centre also presented with stage III (54.6%) and stage IV (10.4% disease, and very few women (approximately 5.2%) presented with stage 1 disease (Kumar 2019). This late presentation of breast cancer may reflect a lack of awareness about breast cancer among women and, in particular, little or no knowledge about early symptoms and signs; and a lack of screening and treatment options.
Various barriers at individual and community levels, coupled with poorly functioning health systems, are likely contributing factors. Limited access to cancer screening and treatment facilities in remote areas, where most of the population in LMICs resides, may also contribute to late presentation of breast cancer. There are no national or population‐based screening programmes for breast cancer available in most LMICs, where mammography is too costly to implement and sustain (Dey 2014). In addition, limited evidence is available on the role of mammography in reducing breast cancer mortality among women between 40 and 50 years of age (Lauby‐Secretan 2015) ‐ the age groups within which most cancers in LMICs are diagnosed (Knaul 2012). Even among health workers, knowledge and clinical skills related to screening, diagnosis, and treatment of breast cancer are limited. For example, study results from a train‐the‐trainer model in Ras Al Khaimah, United Arab Emirates reported that of recruited female nurses from various health centres, only 12.5% had received proper training associated with breast cancer throughout their career. Furthermore, there were concerns from the nurses regarding general breast cancer screening (Awwad 2020).
Clinical breast examination (CBE) is an inexpensive early detection technique for breast cancer for which sufficient evidence shows downstaging of disease at presentation (Lauby‐Secretan 2015). However, health workers in LMICs may be inadequately trained to perform good‐quality CBEs and therefore are likely to dismiss breast complaints and to miss the signs of early disease, compounding the problem of late presentation.
Cancer screening targets a healthy population for the identification of pre‐cancerous lesions, whereas early diagnosis/early detection strategies seeks to identify symptomatic individuals with cancer symptoms and signs who can be referred to treatment facilities in a timely manner (World Health Organization 2017).
Mammography or other technologies will hopefully be more readily available in LMICs in the near future. Early detection with CBE could be viewed as a key interim strategy given currently constrained resources.
Description of the intervention
Screening and early detection of breast cancer can provide maximum beneficial impact in lowering morbidity and mortality due to breast cancer (Dey 2014). Screening in women aged 50 years or older can result in reduction of mortality. A 10‐year follow‐up of a randomised controlled trial (RCT) for annual mammography screening for those under 50 years showed a 25% reduction in mortality (Duffy 2020). The most widely adopted method of screening for breast cancer remains mammography. However, in most LMICs, constrained health infrastructure combined with limited trained personnel makes high‐quality population‐based mammographic screening programmes too costly to implement and sustain (Dey 2014). In LMICs, many breast cancers are diagnosed in women younger than 54 years of age (Knaul 2012), whose breasts are denser (Duffy 2020), thus limiting the accuracy of mammographic diagnosis. The level of awareness among women of the utility of mammography as a screening tool for breast cancer is limited (Alharbi 2012; Obajimi 2013), and those who have heard of the tool often cite financial challenges as a limiting factor in accessing the service. For example, in Ghana, a Knowledge Attitude and Practice (KAP) study suggested that 33.5% of respondents did not know of the existence of mammography services, and of those who were aware of such services, 27% cited financial reasons for not accessing mammography (Opoku 2012). American Cancer Society (ACS) guidelines state that CBE is currently not recommended in for average risk women in the USA as opposed to mammography (Zhang 2021), but it is likely that CBE provides significantly greater benefit in settings where mammography is unavailable. Notably, most women in LMICs have neither regular general health exams nor access to good quality mammography; thus their only contact for a breast complaint might be a health worker within the community. Although the utility of CBE as a breast cancer screening and early detection tool has been mired in controversy (Menes 2021), it is possible that CBE is being performed by health workers who have not been adequately trained in the correct technique. Training health workers in CBE and using these trained personnel to implement early detection programmes for breast cancer may provide the only opportunity for women in LMICs to undergo regular breast examination for early detection of breast cancer.
Given the long delays in diagnosis of breast cancer in LMICs and the related prevalence of late stage disease, earlier detection of palpable masses could improve survival.
CBE is a systematic method that involves physical examination of the breast by a health worker (NCI 2016). Its main purpose is to detect breast abnormalities for onward referral to the next level of care. Various techniques can be used to train diverse groups of health workers in CBE. The main considerations for a standard CBE include clinical history, visual inspection, palpation, and interpretation and reporting of findings (Saslow 2004). Some techniques, such as using silicone breast models and adopting objective structured clinical examination training tools, have been reported to increase the sensitivity of the tool (Saslow 2004). Task shifting involves training community health workers (CHWs) in CBE by using role modelling to empower them to detect breast cancer at an earlier disease stage (Wadler 2011). A competency‐based training programme for primary care physicians and nurses in breast health care that combines technical knowledge, skills, and behaviours has been proposed to circumvent the shortage of specialist personnel in LMICs (Harford 2008). Training community nurses to implement a combination of breast self‐examination (BSE) and CBE was shown to downstage breast cancer presentation in the Cairo Breast Screening Trial (Miller 2008). Interim results from a cluster‐RCT showed that training primary health workers for a specified duration of time in CBE, on the basis of the modified Canadian Breast Screening protocol, and implementing the CBE programme as provided by trained healthcare personnel resulted in downstaging of breast cancer presentation (Mittra 2021). Preliminary results of another RCT from India show that upon completion of a structured three‐week training programme in CBE that targeted female health workers with a tertiary level of education, a significant number of early breast cancers were detected in the intervention arm compared with the control arm (Sankaranarayanan 2011). This contrasts with results from four Canadian organised breast cancer screening programmes, which showed that use of trained health workers in CBE contributed little to early detection of breast cancer (Bancej 2003). A trial in the Philippines also failed to demonstrate any benefit on downstaging of breast cancer after nurses were trained in CBE (Pisani 2006). Furthermore, a lack of evidence was reported in 11 systematic reviews that did not show any impact of CBE on reducing mortality (Ngan 2020). This has been some controversy surrounding CBE in LMICs; in particular, the sensitivity of CBEs performed by nurses in LMICs is low, supporting the premise that the quality of CBE performed may be an issue.
How the intervention might work
The Breast Health Global Initiative (BHGI), established in 2002, is an international health alliance that develops evidence‐based and resource‐sensitive guidelines for screening, early detection, diagnosis, and treatment of breast cancer in LMICs. According to its recommended guidelines for breast cancer, screening should be adopted within the local context and should take into account available resources, with a view towards encouraging incremental improvements ultimately leading to an ideal healthcare delivery system (Duggan 2020). This would offer a more sustainable approach for early detection of breast cancer in LMICs.
Breast self‐awareness (BSA) education and CBE may be important techniques for early detection of breast cancer in the LMIC setting. Given that a large proportion of disease is diagnosed at late stages (Dey 2014), care providers have a great opportunity to make a difference in breast cancer mortality through earlier detection of palpable masses, which can be attained by training a broad group of health workers working within communities, including nurses and CHWs.
CBE remains a viable option for detection of breast cancer at an early stage, when the disease is potentially curable. CBE is a simple procedure that can be performed by a health worker at any level of a healthcare facility and, if appropriately performed, it can detect lumps when they are still small and can contribute to early detection and probably reduced morbidity and mortality from breast cancer. In addition, health workers can be trained in screening CBE or evaluation of breast masses. Screening or evaluation could take place in facilities or in patients’ homes, as was demonstrated in a pilot study from Sudan (Abuidris 2013). Performance of a CBE also provides an opportunity for the health worker to educate women on BSA and breast health care (Farid 2014). The CBE programme can be integrated into existing training programmes for CHWs, such as Visual Inspection by Acetic Acid (VIA) for cervical cancer screening, as has been described in a Malaysian trial for CHWs (Devi 2007). In addition, CBE was also successfully established in four cervical cancer prevention programme clinics in Zambia by training health workers for the early detection and surgical conduction of breast cancer (Pinder 2018). The CBE method can be used as an opportunistic early detection tool by a trained health worker when women present at a health facility for other problems, or it can be included as a component of population‐based screening and early detection programmes through integration into existing women's reproductive health programmes (Farid 2014). Through refresher training of CHWs in CBE, the programme can become a sustainable low‐cost model for embedding early detection of breast cancer into LMICs (Wadler 2011).
Why it is important to do this review
Training health workers in CBE for early detection of breast cancer is an important concept for promoting breast health. It has the potential to promote early detection of breast cancer in LMICs, where mammography is limited and inaccessible for most women. Although controversy surrounds the utility of CBE as a mass breast screening tool (Menes 2021), with large RCTs in China and Russia showing no effect on breast cancer mortality when BSE or CBE is performed in isolation (Kösters 2003; Thomas 1997), evidence showing the efficacy of training health workers in CBE as an early detection tool in the LMICs is limited. An RCT from the Philippines that studied clinical outcomes of CBE performed by trained nurses had to be abandoned owing to participant non‐compliance with follow‐up and treatment guidelines (Pisani 2006). This RCT reported that despite training of nurses, both the inexperience of the health worker and breast cancer beliefs and behaviours at the individual level contributed to the low sensitivity (53.2%) of the programme, and that these concerns must be addressed before future screening programmes are implemented.
Within the context of knowledge, experience, and recommendations from global practitioners, it is recognised that community engagement is key to strengthening interventions that improve health outcomes. In particular, community‐based interventions have been recognised as playing an important role in improving maternal, newborn, and child health (Rifkin 2014). CHWs are “in a good position to promote health in ways that reflect the political, environmental, social, and cultural realities of the community, partly through facilitation of broader community participation” (LeBan 2021). Training CHWs and other health workers in CBE for early detection of breast cancer can therefore provide a sustainable model in the LMIC setting, where existing health infrastructure and resources are overwhelmed by management of diseases of infective origin. A participatory approach in a southern Brazilian city displayed how on the basis of minimal skills training and limited education, CBE can be implemented in an LMIC setting by training CHWs and integrating breast healthcare into existing healthcare programmes (Bittencourt 2017). However, the trial authors admit that the pragmatic success of implementing trained CHW breast screening activities requires high‐certainty evidence derived from RCTs. This Cochrane Review aimed to explore different CBE training programmes for various groups of health workers, and determine whether training in CBE improves the quality of the technique and the ability of health workers to detect breast cancers early.
Objectives
To assess whether training in CBE affects the ability of health workers in LMICs to detect early breast cancer.
Methods
Criteria for considering studies for this review
Types of studies
RCTs (including individual and cluster‐RCTs), quasi‐experimental studies and controlled before‐and‐after (CBA) studies.
Types of participants
We included training of any group of health workers (including lay and professional health workers) to perform CBE in community settings for women aged 18 years and older. Community settings included home visitations and visits to primary healthcare centres.
Types of interventions
Intervention
We considered any form of training provided to any group of health workers (lay or professional) to conduct CBE as an opportunistic or population‐based early detection tool for breast cancer in community settings in LMICs (as defined by World Bank 2021 classification).
Comparator
No training of health workers in CBE or use of any other mass screening or early detection tools for breast cancer (BSE, ultrasonography, mammography, or magnetic resonance imaging).
Types of outcome measures
Primary outcomes
Breast cancer stage at the time of presentation: defined as the proportion of women with breast cancer diagnosed at each stage. We dichotomised breast cancer into early and late stages. We defined early stage as breast cancer that had not spread beyond the breast or the axillary lymph nodes, including ductal carcinoma in situ and stages I, IIA, IIB, and IIIA breast cancers; and defined late stage as breast cancer at stages IIIB and IV (NCI 2016). The numerator represented the proportion of women with a diagnosis of early or late breast cancer among the total number of women given a cancer diagnosis.
Secondary outcomes
KAP outcomes: defined as the KAP of breast cancer screening ‐ early detection and diagnosis among health workers and women within communities. We reported KAP as described by trial authors as ordinal or continuous outcomes on a scale, depending on what has been reported in the primary study (e.g. the number of correct answers on a questionnaire on knowledge related to CBE, the extent of current knowledge and practice in performing the CBE).
Accuracy of health worker‐performed CBE (e.g. sensitivity and specificity, positive predictive value, performance on checklists, performance as defined by individual study authors).
CBE coverage: defined as the proportion of the population covered by CBE interventions (intervention group) of the total eligible study population, as defined by respective study authors. The denominator for CBE coverage represented women at risk (the population of women of reproductive age in the study area), and the numerator represented those who actually experienced CBE.
Completion of follow‐up: defined as the proportion of women who completed follow‐up after screening and CBE and advised to complete follow‐up.
Breast cancer mortality: defined as deaths among women who received a diagnosis of breast cancer.
Cost‐effectiveness: defined as relative costs and outcomes of training health workers in CBE. This information was reported as described by study authors.
Search methods for identification of studies
Electronic searches
The following databases were searched up to 17 July 2021.
The Cochrane Breast Cancer Group (CBCG) Specialised Register. Details of search strategies used by the CBCG to identify studies and of the procedure used to code references are provided in the Group module (www.mrw.interscience.wiley.com/cochrane/clabout/articles/BREASTCA/frame.html). We extracted the keywords "breast cancer screening, early detection and diagnosis, low and middle income countries, resource challenged/constrained settings, community screening, clinical breast exam, self breast exam, breast self‐examination, ultrasound exam, mammography, mass screening, population based screening, opportunistic screening, training in clinical breast exam, community health workers training" and considered identified studies for inclusion in the review.
Cochrane Central Register of Controlled Trials (CENTRAL; latest issue) in the Cochrane Library (Appendix 1).
MEDLINE (via OvidSP; from 1946 to 17 July 2021; Appendix 2).
Embase (via OvidSP; from 1974 to 17 July 2021; Appendix 3).
World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) search portal (http://apps.who.int/trialsearch/Default.aspx) for all prospectively registered and ongoing trials (Appendix 4).
ClinicalTrials.gov (http://clinicaltrials.gov/;Appendix 5).
Searching other resources
Bibliographic searching
We identified additional studies from reference lists of identified relevant studies or reviews. We obtained a copy of the full article for each reference reporting a potentially eligible trial. When this was not possible, we attempted to contact study authors to obtain required information.
Data collection and analysis
Selection of studies
Two review authors (SS and PO) independently assessed all studies identified for possible inclusion as a result of the search. We resolved disagreements through discussion and, whenever required, through consulting a third review author (AN). We obtained a copy of the full article for each reference reporting a potentially eligible trial. When this was not possible, we attempted to contact study authors to request additional information. When data were limited or when information on study methods was not provided, we requested further information from the study authors. We included relevant articles published in all languages. We attempted to obtain translated versions of the included articles written in languages other than English. We recorded excluded studies in the 'Characteristics of excluded studies' table along with reasons for their exclusion.
Data extraction and management
Two review authors (SS and AN) extracted data independently, using a pre‐designed data extraction form. We resolved discrepancies through discussion, and whenever required, we consulted a third review author (RAS). We requested further information from study authors where extracted data were limited. For studies with more than one publication, we extracted data from all publications but considered the final or updated version of each study as the primary reference.
We extracted the following information from the included studies.
Publication details (i.e. year, country, study authors).
Methods (i.e. study design, location/setting, follow‐up period).
Participants (i.e. age).
Interventions (i.e. CBE training duration, type, frequency, trainers).
Outcomes reported by study authors.
We entered information related to the included studies into Review Manager 2023 software. When information regarding any of the above was unclear, we attempted to contact authors of the original reports to obtain further details.
Assessment of risk of bias in included studies
Two review authors (SS and AN) independently assessed the risk of bias for each included RCT using the Cochrane 'Risk of bias' tool described in Higgins 2011. We resolved disagreements by discussion or by consulting a third review author (RAS). We assessed the included studies’ risk of bias according to the following domains.
Random sequence generation.
Allocation concealment.
Blinding of participants and personnel.
Blinding of outcome assessment.
Incomplete outcome data.
Selective outcome reporting.
Validity of outcome measure.
Other bias.
We assessed the risk of bias from each potential source for each study as either high, low, or unclear and provided supporting evidence for our decision in the 'Risk of bias' tables.
A study was judged to be at low risk of bias overall when all the domains had a low risk of bias i.e. the study included a sufficiently detailed description of its random sequence generation, allocation concealment, blinding of outcome assessment, complete outcome data, non‐selective outcome reporting, and valid outcome measures. We judged selective outcome reporting on the extent to which all the outcomes mentioned in their methods section were reported in the results. We judged a study to have a high risk of bias if some of the reported features predisposed it to have a high risk of bias in any one of the eight domains, and unclear risk category was allocated when there was no information (or insufficient information) on which to assess a particular domain.
Overall risk of bias
We made explicit judgements about whether studies were at high risk of bias, according to the criteria given in Higgins 2011. We assessed the likely magnitude and direction of bias, and whether we considered bias was likely to have had an impact on study findings.
Measures of treatment effect
Dichotomous data
We reported dichotomous outcomes (e.g. breast cancer mortality; stage at presentation; knowledge attitude and practice outcomes; accuracy of performance; CBE coverage; follow‐up) as risk ratios (RRs) and risk differences (RDs) with 95% confidence intervals (CIs). We pooled data for meta‐analysis using the pooled logRR, when appropriate.
Continuous data
For continuous data, we used the mean difference (MD) if outcomes were measured in the same way between studies. We used the standardised mean difference (SMD) to combine studies that measure the same outcome with 95% CIs while using different scales.
Time‐to‐event data
We did not find time‐to‐event data for reporting in this review. For future review updates, we intend to present breast cancer mortality as a time‐to‐event outcome and perform a generic inverse variance meta‐analysis where estimates of hazard ratios and their uncertainties are reported by the study authors.
Unit of analysis issues
Cluster‐RCTs
Both cluster‐RCTs and individual RCTs were eligible for inclusion. All included studies in the present review are cluster‐RCTs. Where relevant, we adjusted standard errors according to the methods described in Higgins 2011 by using an estimate of the intra‐cluster correlation coefficient (ICC) derived from the study (if possible), from a similar study or from a study of a similar population. For current analysis, we did not use ICCs from other sources; however for future updates, if we use ICCs from other sources, we will report this and conduct sensitivity analyses to investigate the effect of variation in the ICC. We did not identify any individual RCTs but for future updates, if we identify both cluster‐RCTs and individual RCTs, we will synthesise relevant information. We will consider it reasonable to combine results from both if there would be little heterogeneity between study designs, and if the interaction between effects of the intervention and choice of randomisation unit is considered unlikely. We would acknowledge heterogeneity in the randomisation unit and perform a sensitivity analysis to investigate its effects.
Studies with more than two intervention groups
For future updates, if we identify studies with more than two intervention groups (multi‐arm studies), we will combine groups when possible to create a single pair‐wise comparison or use the methods set out in Higgins 2011 to avoid double‐counting of study participants. For subgroup analyses, if the control group is shared by two or more study arms, we will divide the control group (events and total population) over the number of relevant subgroups to avoid double‐counting of participants.
Dealing with missing data
We described missing data, including the number of participants lost to follow‐up. Differential dropout rates can lead to biased estimates of effect size, and bias may arise if the reasons for dropping out differ across groups. We reported the reasons for loss to follow‐up. For included studies, we noted levels of attrition. We intended to explore the impact of including studies with high levels of missing data in the overall assessment of treatment effect by performing sensitivity analysis; however due to limited number of studies included in the review we could not conduct a sensitivity analysis. As far as possible, we performed analyses on an intention‐to‐treat basis for all outcomes; that is, we attempted to include in the analysis all participants randomised to each group, and we analysed all participants in the group to which they were allocated, regardless of whether they received the allocated intervention. The denominator for each outcome in each study is the number randomised minus the number of participants whose outcomes known to be missing.
Assessment of heterogeneity
We examined included studies for clinical, methodological, and statistical heterogeneity. We assessed clinical heterogeneity by comparing the distribution of important factors, such as study participants, study setting, dose, assessment tools, and duration of the intervention and of co‐interventions. We evaluated methodological heterogeneity on the basis of factors such as method of sequence generation, allocation concealment, blinding of outcome assessment, and losses to follow‐up. We assessed statistical heterogeneity in each meta‐analysis using Tau2, I2, and Chi2 statistics. We regarded heterogeneity as substantial if I2 was greater than 50%, and if Tau2 was greater than zero or the P value was low (< 0.10) in the Chi2 test for heterogeneity. The importance of the observed value of I2 depends on (1) magnitude and direction of effects and (2) strength of evidence for heterogeneity. A guide for the I2 statistic follows.
0% to 40%: might not be important.
30% to 60%: may represent moderate heterogeneity.
50% to 90%: may represent substantial heterogeneity.
75% to 100%: shows considerable heterogeneity.
Assessment of reporting biases
None of the outcomes were included in 10 or more studies in the meta‐analysis, hence we could not investigate reporting bias through funnel plots. For future review updates, we will investigate reporting biases (such as publication bias) by using funnel plots. For continuous outcomes, we will use the test proposed by Egger 1997. For dichotomous outcomes, we will use the test proposed by Harbord 2006. If asymmetry were detected in any of these tests or suggested by a visual assessment, we will attempt to explore contextual factors, including the plausibility of publication bias as an explanation for the asymmetry, and compare fixed‐effect and random‐effects estimates of the intervention effect when we observe heterogeneity between studies (Higgins 2011; Sterne 2011).
Data synthesis
We performed statistical analysis using Review Manager 2023. We used fixed‐effect meta‐analysis in combining data when it was reasonable to assume that studies were estimating the same underlying treatment effect; that is, when studies were examining the same intervention, and that study populations and methods were judged to be sufficiently similar. If clinical heterogeneity was sufficient to suggest that underlying treatment effects differ between studies, and if we had detected substantial statistical heterogeneity, we used random‐effects meta‐analysis to produce an overall summary.
For continuous outcomes, we combined data using the inverse variance method on the MD scale. For the random‐effects model, which incorporates an assumption that different studies are estimating different, yet related, intervention effects, we used the DerSimonian and Laird method (DerSimonian 1986). For dichotomous outcomes, we combined data on the log‐RR scale using the Mantel‐Haenszel method.
Data could not be meta‐analysed for the outcome 'Accuracy of health worker‐performed CBE' and therefore we synthesised the results for this outcome using results guideline from 'Synthesis without meta‐analysis (SWiM) in systematic reviews: reporting guideline' (Campbell 2020). We reported the ranges for sensitivity, specificity, and positive predictive value from both the studies reporting this outcome.
We set out the main findings of the review in summary of findings tables prepared via the GRADE approach (Guyatt 2008), using GRADEpro GDT software. We listed the primary outcome for each comparison with estimates of relative effects along with the numbers of participants and studies contributing data for those outcomes. For each individual outcome, we assessed the certainty of the evidence using the GRADE approach, which involves consideration of within‐study risk of bias (methodological quality), directness of evidence, heterogeneity, precision of effect estimates, and risk of publication bias. We rated the certainty of the body of evidence for each key outcome as either high, moderate, low, or very low.
Subgroup analysis and investigation of heterogeneity
We aimed to perform subgroup analysis according to the following.
Types of health workers (nurses, physicians, lay health workers, etc).
Duration of training (as reported by study authors).
However, due to limited number of studies included in the review, we could not perform any subgroup analysis.
Sensitivity analysis
We aimed to perform sensitivity analyses to examine the effect of removing studies at high risk of bias (those with high or unclear risk of bias according to method and adequacy of allocation concealment; blinding status of participants; percentage lost to follow‐up or attrition of 20%; and a random‐effects model of the primary analysis). We also aimed to perform sensitivity analyses to examine different types of study designs and different ICC values. However, due to limited number of included studies, we could not perform sensitivity analysis.
Summary of findings and assessment of the certainty of the evidence
We assessed the certainty of evidence for inclusion in the summary of findings table for the following outcomes.
Breast cancer stage at the time of presentation.
Accuracy of health worker‐performed CBE.
CBE coverage.
Completion of follow‐up.
Breast cancer mortality.
Results
Description of studies
Results of the search
We screened a total of 2099 records and assessed 27 full texts for eligibility. Four studies covering a population of 947,190 women for breast cancer screening, met the inclusion criteria of the Cochrane Review (Mittra 2021; Pace 2019; Pisani 2006; Sankaranarayanan 2011). The study selection process is outlined in Figure 1.
1.
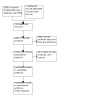
Study flow diagram.
Included studies
The four included studies were cluster‐RCTs. All were conducted in LMICs; two were conducted in India (Mittra 2021; Sankaranarayanan 2011), one in Rwanda (Pace 2019), and one in the Philippines (Pisani 2006). The participants in all four trials were women of reproductive age between 30 and 69 years of age who were being screened for breast cancer. The sample size in the included studies were 151,538 in Mittra 2021; 151,168 in Pisani 2006; 340,000 in Pace 2019, and 115,652 in Sankaranarayanan 2011.
Mittra 2021 compared screening through CBE conducted by trained primary health workers with controls. The intervention clusters underwent nine rounds of biennial monitoring for breast cancer occurrence and mortality, four rounds of screening by CBE and cancer awareness education, and five rounds of active surveillance. The control clusters underwent nine rounds of biennial monitoring for breast cancer occurrence and mortality, one round of cancer awareness education, and eight rounds of active surveillance. Pace 2019 trained nurses and CHWs in performing CBE along with instruction for CHWs in symptoms of breast cancer and messaging that community members should come to their local health centre for any breast symptoms. Nurses were taught about signs and symptoms of benign breast disease and cancer and how to perform a CBE and were provided with simple clinical algorithms for managing breast symptoms and examination findings, with an emphasis on findings that need urgent hospital‐level evaluation. No training was provided to the nurses and CHWs in the intervention clusters. Pisani 2006 compared CBEs conducted by trained nurses and midwives with control. Five annual CBEs were performed in the intervention clusters along with instructions on BSE technique and were given a leaflet in the local language explaining the importance of BSE. The control clusters did not receive any active intervention but were exposed to general health education campaigns carried out by municipal authorities and voluntary bodies. Sankaranarayanan 2011 compared CBE conducted by trained female health workers with controls. Three rounds of triennial CBE were performed in the intervention clusters, while the control clusters received education on cervical cancer prevention and information on how to access cervical treatment and screening.
Three included studies reported on the primary outcome of the review (breast cancer stage at the time of presentation) (Mittra 2021; Pace 2019; Sankaranarayanan 2011). Regarding secondary outcomes, Mittra 2021 reported CBE coverage, accuracy of health worker‐performed CBE, and breast cancer mortality. Pisani 2006 and Sankaranarayanan 2011 reported accuracy of health worker‐performed CBE. Pisani 2006 presented outcome data for the intervention arm and the intervention was discontinued after the first round of screening due to non‐compliance of follow‐up by the participating women. None of the included studies reported on KAP outcomes and cost‐effectiveness.
Refer to the Characteristics of included studies tables for further details.
Excluded studies
We excluded 23 studies after full‐text screening. The reasons for exclusion of most excluded studies were: the intervention did not involve CBE training, the studies were not conducted in LMICs settings, and the studies did not fulfil the study design criteria.
Please refer to the Characteristics of excluded studies for further details.
Risk of bias in included studies
The risk of bias of the included studies are summarised in Figure 2 and Figure 3.
2.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
3.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
Random sequence generation
Three studies were judged to have an unclear risk of bias for this domain since they provided only partial information on the method used to assign participants to groups. Mittra 2021 had 20 clusters that were numbered from 1 to 20 and randomly assigned to either control or screening clusters. Ten groups were allocated to either screening or controlled clusters. Pisani 2006 indicated that block randomisation was used, but it was unclear what method was used to allocate blocks to either arm while Sankaranarayanan 2011 used random cluster allocation, but the method used to generate the random sequences was not mentioned. We judged Pace 2019 to be at low risk for random sequence generation; however randomisation in this study was done in two phases. In the first phase, 18 health centres were randomised into intervention and control clusters (7 intervention clusters and 11 control clusters). In the second phase, an additional five health centres from the control clusters were randomised to receive intervention while six health centres served as controls throughout the study period.
Allocation concealment
We judged all four studies to be at unclear risk of bias for allocation concealment, since they did not provide any information on concealing the allocation.
Blinding
All studies were at high risk of performance and detection bias since the participants, the health workers, and outcome assessors were aware of the arms to which participants had been allocated due to the nature of the intervention.
Incomplete outcome data
We judged Mittra 2021 and Pace 2019 to be at low risk of attrition bias, and Pisani 2006 and Sankaranarayanan 2011 to be at high risk of attrition bias due to high loss to follow‐up.
Selective reporting
We judged two studies as at high risk of reporting bias (Mittra 2021; Pisani 2006). Mittra 2021 reported interim analyses as the study was still ongoing, and did not present cost‐effectiveness analyses of CBE. Pisani 2006 did not report some of the outcomes (including mortality and downstaging of breast cancer at presentation) since the trial was aborted. Pisani 2006 was judged to be at high risk of bias due to very low compliance with clinical follow‐up (65% refused follow‐up or incomplete follow‐up or lost to follow‐up); the intervention ceased after completion of the first round of examinations hence data was not included in the review. We judged Pace 2019 to be at low risk of bias for selective reporting. Sankaranarayanan 2011 was classified as at unclear risk of bias since we did not find any trial registration details or protocol.
Other potential sources of bias
We judged Pisani 2006 to be at high risk of bias since due to very low compliance with clinical follow‐up; the intervention ceased after completion of the first round of examinations. We judged the other three included studies to be at low risk for any other biases (Mittra 2021; Pace 2019; Sankaranarayanan 2011).
Overall risk of bias
Overall, we judged the four included studies to have high risk of bias based on the foregoing.
Effects of interventions
See: Table 1
Primary outcomes
Breast cancer stage at the time of presentation: pooled results
Of the four included studies, with a total population of 947,190 women screened for breast cancer, three studies reported 593 participants diagnosed with breast cancer at an early stage (at stage 0+I+II) (Mittra 2021; Pace 2019; Sankaranarayanan 2011), suggesting that training health workers for CBE may increase the number of women detected with breast cancer at an early stage of breast cancer compared to the non‐training group (RR 1.44, 95% CI 1.01 to 2.06; three studies; 593 participants; I2 = 0%; low‐certainty evidence; Analysis 1.1).
1.1. Analysis.

Comparison 1: Training for CBE versus no training, Outcome 1: Early stage (0+I+II) diagnosis
Three studies reported 593 participants diagnosed with breast cancer at late stages (III+IV) (Mittra 2021; Pace 2019; Sankaranarayanan 2011), suggesting that training health workers for CBE may slightly reduce the number of women detected with breast cancer at late stages compared to the non‐training group (RR 0.58, 95% CI 0.36 to 0.94; three studies; 593 participants; I2 = 52%; low‐certainty evidence; Analysis 1.2).
1.2. Analysis.

Comparison 1: Training for CBE versus no training, Outcome 2: Late stage (III+IV) diagnosis
Secondary outcomes
Accuracy of health worker‐performed CBE: non‐pooled results
Two studies reported this outcome (Pisani 2006; Sankaranarayanan 2011). We were unable to meta‐analyse data, and therefore we used the reporting results guideline from 'Synthesis without meta‐analysis' (SWiM) in systematic reviews: reporting guideline' to synthesise the results (Campbell 2020). We report the ranges for sensitivity, specificity, and positive predictive value from estimates reported in both studies.
The sensitivity ranged between 51.7% (Sankaranarayanan 2011) to 53.2% (Pisani 2006) (very low‐certainty evidence); while the specificity ranged between 94.3% (Sankaranarayanan 2011) to 100% (Pisani 2006) (very low‐certainty evidence). The positive predictive value ranged between 1% (Sankaranarayanan 2011) to 1.2% (Pisani 2006) (very low‐certainty evidence).
CBE coverage: single study results
One study reported CBE coverage (Mittra 2021). Results suggested that out of 75,360 eligible women, the mean adherence for the overall four screening rounds was 67.07% (moderate‐certainty evidence); with 75.62% for round one, 65.42% for round two, 63.51% for round three, and 63.52% for round four.
Follow‐up: single study results
One study reported follow‐up defined as the proportion of women who completed follow‐up after screening and CBE (Mittra 2021). Results suggested that compliance rates for diagnostic confirmation following a positive CBE were 68.29%, 71.20%, 78.84%, and 79.98% during the first, second, third, and fourth rounds of screening in the intervention group compared to 90.88%, 82.96%, 79.56%, and 80.39% during the respective four rounds of screening in the control group. The mean adherence to rounds 5 to 9 of active surveillance after the CBE screening was conducted was 77.57% in the intervention arm, which was similar to the control arm (76.22%).
Breast cancer mortality: pooled results
Two studies including 355 participants with diagnosed breast cancer reported breast cancer mortality (Mittra 2021; Sankaranarayanan 2011). Results suggested that the evidence is uncertain for the impact on breast cancer mortality (RR 0.88, 95% CI 0.24 to 3.26; two studies; 335 participants; I2 = 68%; very low‐certainty evidence; Analysis 1.3).
1.3. Analysis.

Comparison 1: Training for CBE versus no training, Outcome 3: Breast cancer mortality
Secondary outcomes
None of the included studies reported any other secondary outcomes, including KAP outcomes and cost‐effectiveness.
Discussion
Summary of main results
This Cochrane Review summarises findings from four studies (covering a population of 940,190 for screening). Our findings suggest that training health workers in CBE for early detection of breast cancer in LMICs may increase early detection of breast cancer when compared to no training (low‐certainty evidence). Among secondary outcomes, accuracy of health worker‐performed CBE, CBE coverage, follow‐up, and breast cancer mortality were reported. Meta‐analysis could only be conducted for breast cancer mortality, suggesting that the evidence is uncertain for the effect of training for CBE on breast cancer mortality (very low‐certainty evidence). Sensitivity of health worker‐performed CBE was reported to be 53.2% and 51.7%; while specificity was reported to be 100% and 94.3% respectively in two included studies. One study reported CBE coverage with a mean adherence of 67.07% for the first four screening rounds. One study reported follow‐up suggesting that compliance rates for diagnostic confirmation following a positive CBE were 68.29%, 71.20%, 78.84%, and 79.98% during respective first four rounds of screening in the intervention group compared to 90.88%, 82.96%, 79.56%, and 80.39% during the respective four rounds of screening in the control group. None of the included studies reported any other secondary outcomes, such as KAP outcomes and cost‐effectiveness. See Table 1.
Overall completeness and applicability of evidence
All included studies were conducted in LMICs (India, Rwanda, and The Philippines). Various cadres of health workers were trained in the included studies. Mittra 2021 provided training to primary health workers that were women with 10th grade education; Pisani 2006 provided training to paramedics including nurses and midwives; Sankaranarayanan 2011 trained female health workers in the community with a bachelor’s degree; while Pace 2019 provided training to the nurses working at the health centre who in turn provided trainings to the CHWs in the community. The content and duration of training also varied among the included studies. Mittra 2021 provided training for four weeks in CBE using a modified version of the 'Canadian National Breast Screening Study Protocol'; Pisani 2006 provided CBE training using silicone breast models; Pace 2019 provided 1‐day training of trainers to the clinicians who conducted a 3‐4 day training for the nurses working at the health centres, who trained the CHWs in a 1‐day training; while Sankaranarayanan 2011 trained health workers through a three‐week structured course using silicone breast models, followed by visual inspection and palpation of women with normal breasts, fibroadenosis, benign tumours and cancers, followed by periodic reorientation courses.
Due to the limited number of included studies, we could not conduct the planned subgroup and sensitivity analysis. Meta‐analysis could not be performed for any of the secondary outcomes except breast cancer mortality.
Certainty of the evidence
We judged most included studies to be at high risk of bias for blinding and attrition. All the studies were judged to be at unclear risk of bias for sequence generation and allocation concealment, since sufficient information was not provided regarding the randomisation process to permit judgement.
Evidence certainty for the outcomes reported in our review ranged between moderate‐ to very low‐certainty evidence. We judged the evidence certainty for our primary outcome 'Breast cancer stage' to be low‐certainty, due to study limitations based on the risk of bias assessment of the included studies and small number of events. We rated CBE coverage and completion of follow‐up as moderate‐certainty evidence, and downgraded the certainty of the evidence by one level due to study limitations. Accuracy of health worker‐performed CBE and breast cancer mortality were further downgraded by two levels, and we rated these as very low‐certainty evidence due to heterogeneity in intervention, indirectness for measuring diagnostic accuracy, and inconsistent estimates reported in the studies meta‐analysed.
Potential biases in the review process
We were aware of the possibility of introducing bias at every stage of the reviewing process. In this Cochrane Review, we tried to minimise bias in a number of ways; two review authors assessed study eligibility for inclusion, carried out data extraction, and assessed risk of bias and the certainty of the evidence. While we attempted to be as inclusive as possible in the search strategy, the literature identified was predominantly written in English. Although we did attempt to assess reporting bias, this assessment largely relied on information available in the published studies and thus, reporting bias was not usually apparent.
Agreements and disagreements with other studies or reviews
To our knowledge, this is the first systematic review assessing the impact of health worker training in CBE on early breast cancer detection in LMICs.
A recent overview of systematic reviews assessed the effectiveness of CBE as a 'stand‐alone' screening tool for breast cancer compared to no screening with a focus on LMICs (Ngan 2020). The findings from 11 systematic reviews suggested no direct evidence that CBE reduced breast cancer mortality. However, there was indirect evidence that a well‐performed CBE had the same effect as mammography on mortality. The review authors concluded that there is merit in CBE from a LMIC health system perspective as it could be a cheaper alternative to mammography. However, the review did not assess the effectiveness of training health workers in CBE.
Another systematic review analysed the results of implementation trials and screening interventions in sub‐Saharan Africa (Martei 2022). The outcomes from the examined 15 studies showed that CBE screening displayed extreme heterogeneity in the reported results, the personnel managing of the examinations, as well as in the settings CBE was conducted. Results also displayed that there is no set age criteria for when CBE should be implemented in sub‐Saharan Africa. In addition, the review showed that CBE screening connected to comprehensive cancer care is the most cost‐effective, which we were unable to analyse in this Cochrane Review.
One study examined the cost‐effectiveness of the yearly national screening mammography programme for Ethiopian women aged 40 years and older (Kejela 2021). A model programme was used to assess the health benefits and cost of interventions concerning screening mammography. Analysis of cost‐effectiveness for four policy groups constructed upon mammography screening all failed to have a proper incremental cost‐effectiveness ratio. Overall, screening mammography in Ethiopia through this study displayed to not be cost‐effective. Acknowledging that CBE is cheaper and more accessible compared to screening mammography, the study indicated that yearly CBE should be assessed for its cost‐effectiveness using a similar model.
Authors' conclusions
Implications for practice.
The findings of our Cochrane Review suggest there may be some benefit of training health workers from LMICs in CBE on early detection of breast cancer. However, the certainty of the evidence is very low regarding mortality, accuracy of health working performed CBE, and completion of follow‐up.
Implications for research.
There is a need for rigorous, good‐quality studies in LMICs settings to ascertain the impact of this low‐cost intervention on early detection of breast cancer in women living in LMICs. This may involve standardising training modules for training health workers in CBE, and utilising various cadres of health workers in LMICs to evaluate what works well in different settings. Furthermore, monitoring of the performance and quality of CBE by health workers is also important to ensure adequate screening. Future studies should focus on monitoring and reporting participants' compliance and follow‐up at each stage of screening, as low rates of compliance and high loss to follow‐up in these large scale screening programmes can compromise programme effectiveness. Moreover, efforts may be needed to ensure compliance and follow‐up to ascertain the efficacy of the intervention.
History
Protocol first published: Issue 1, 2017
Acknowledgements
We acknowledge assistance received from Cochrane South Africa in refining study objectives.
We thank the following people for their helpful peer review during protocol and review stages:
Cecilia Fabrizio, DrPH (consumer editor);
Nuala Livingstone, Cochrane Evidence Production and Methods Directorate, UK (methods reviewer);
Lydia Pace MD, MPH, Brigham and Women's Hospital, USA (clinical reviewer).
We thank Peta Skeers and Ava Tan for developing the search strategies.
Appendices
Appendix 1. CENTRAL
#1 MeSH descriptor: [Community Health Workers] explode all trees #2 MeSH descriptor: [Health Personnel] explode all trees #3 (health worker* or health care worker* or health professional or health personnel or doctor* or nurse* or physician*) #4 #1 or #2 or #3 #5 MeSH descriptor: [Health Education] explode all trees #6 MeSH descriptor: [Education, Continuing] explode all trees #7 MeSH descriptor: [Education, Medical] explode all trees #8 MeSH descriptor: [Education, Nursing] explode all trees #9 (train* or training or education or curriculum or teaching or learning or staff development or medicine) #10 #5 or #6 or #7 or #8 or #9 #11 #4 and #10 #12 MeSH descriptor: [Health Knowledge, Attitudes, Practice] explode all trees #13 #11 or #12 #14 MeSH descriptor: [Physical Examination] explode all trees #15 ((physical or clinical breast or clinical or breast) adj1 exam*) #16 clinical breast examination #17 MeSH descriptor: [Mass Screening] explode all trees #18 #14 or #15 or #16 or #17 #19 MeSH descriptor: [Breast Neoplasms] explode all trees #20 breast near neoplasm* #21 breast near carcinom* #22 breast near cancer* #23 breast near tumour* #24 breast near tumor* #25 breast near malignan* #26 #19 or #20 or #21 or #22 or #23 or #24 or #25 #27 #13 and #18 and #26
Appendix 2. MEDLINE via OvidSP
| 1 | exp Community Health Workers/ |
| 2 | exp Health Personnel/ |
| 3 | community health worker*.tw. |
| 4 | (health worker* or health care worker* or health professional or health personnel or doctor* or nurse* or physician*).tw. |
| 5 | 1 or 2 or 3 or 4 |
| 6 | exp Health Education/ |
| 7 | exp Education, Continuing/ |
| 8 | exp Education, Medical/ |
| 9 | exp Education, Nursing/ |
| 10 | (train* or training or education or curriculum or teaching or learning or staff development or medicine).tw. |
| 11 | 6 or 7 or 8 or 9 or 10 |
| 12 | 5 and 11 |
| 13 | exp Community Health Workers/ed [Education] |
| 14 | exp Health Knowledge, Attitudes, Practice/ |
| 15 | exp Health Personnel/ed [Education] |
| 16 | 12 or 13 or 14 or 15 |
| 17 | exp Physical Examination/mt [Methods] |
| 18 | ((physical or clinical breast or clinical or breast) adj1 exam*).tw. |
| 19 | clinical breast examination.tw. |
| 20 | exp Mass Screening/mt [Methods] |
| 21 | 17 or 18 or 19 or 20 |
| 22 | exp Breast Neoplasms/ |
| 23 | (breast adj6 cancer$).tw. |
| 24 | (breast adj6 neoplasm$).tw. |
| 25 | (breast adj6 carcinoma$).tw. |
| 26 | (breast adj6 tumo?r$).tw. |
| 27 | or/22‐26 |
| 28 | 16 and 21 and 27 |
| 29 | Animals/ not Humans/ |
| 30 | 28 not 29 |
Appendix 3. Embase via OvidSP
| 1 | exp health auxiliary/ |
| 2 | exp health care personnel/ |
| 3 | community health worker*.tw. |
| 4 | (health worker* or health care worker* or health professional or health personnel or doctor* or nurse* or physician*).tw. |
| 5 | 1 or 2 or 3 or 4 |
| 6 | exp medical education/ |
| 7 | exp continuing education/ |
| 8 | exp nursing education/ |
| 9 | (train* or training or education or curriculum or teaching or learning or staff development or medicine).tw. |
| 10 | 6 or 7 or 8 or 9 |
| 11 | 5 and 10 |
| 12 | exp physical examination/ |
| 13 | ((physical or clinical breast or clinical or breast) adj1 exam*).tw. |
| 14 | clinical breast examination*.tw. |
| 15 | exp mass screening/ |
| 16 | 12 or 13 or 14 or 15 |
| 17 | exp breast/ |
| 18 | exp breast disease/ |
| 19 | (17 or 18) and exp neoplasm/ |
| 20 | exp breast tumor/ |
| 21 | exp breast cancer/ |
| 22 | exp breast carcinoma/ |
| 23 | (breast$ adj5 (neoplas$ or cancer$ or carcin$ or tumo$ or metasta$ or malig$)).ti,ab. |
| 24 | 19 or 20 or 21 or 22 or 23 |
| 25 | 11 and 16 and 24 |
| 26 | limit 25 to (human and embase) |
Appendix 4. WHO ICTRP search portal
Basic search:
Breast exam AND education OR breast exam AND training OR breast exam AND learning OR breast exam AND knowledge OR breast exam AND health worker OR breast exam AND nurse OR breast exam AND community OR breast exam AND lay
Appendix 5. ClinicalTrials.gov
Basic search:"breast exam" AND (education OR training OR learning OR knowledge OR health worker OR nurse OR community OR lay)
Data and analyses
Comparison 1. Training for CBE versus no training.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Early stage (0+I+II) diagnosis | 3 | 593 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.44 [1.01, 2.06] |
| 1.2 Late stage (III+IV) diagnosis | 3 | 593 | Risk Ratio (M‐H, Random, 95% CI) | 0.58 [0.36, 0.94] |
| 1.3 Breast cancer mortality | 2 | 355 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.24, 3.26] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Mittra 2021.
| Study characteristics | ||
| Methods | Design: RCT Unit: 20 clusters (slums) |
|
| Participants | Location/setting: study was carried out in Mumbai, India. Health workers who had passed 10th grade education were trained to conduct CBE within 4 weeks to perform on participants in the study. Sample size: 151,538 women aged 35 to 64 Sex: only females were included. Mean age: mean age for enrolment for all trial participants in the screening and control arm was 44.84 and 44.92 years respectively. Inclusion criteria: women aged 35 to 64 years who did not have a history of breast cancer. Exclusion criteria: women outside of ages 35 to 64 years, and all women with a history of breast cancer. |
|
| Interventions | Intervention (screening) group: (n = 75,360) of 10 clusters must undergo 9 rounds of biennial monitoring for breast cancer occurrence and mortality, 4 rounds of screening by CBE and cancer awareness education and 5 rounds of active surveillance. Control group: (n = 76,178) of 10 clusters must undergo 9 rounds of biennial monitoring for breast cancer occurrence and mortality, 1 round cancer awareness education, 8 rounds of active surveillance. Trial duration was 20 years. |
|
| Outcomes | Primary outcomes: the down‐staging of breast cancer at diagnosis and reduction in mortality from breast cancer. Secondary outcomes: CBE coverage, sensitivity, and specificity. Timing of outcome assessments: total trial duration was 20 years, but database was locked in March 2019 for analysis. |
|
| Notes | Study start date: May 1998 Study end date: March 2019 Funding source: US National Institutes of Health (grant number: RO1CA074801) Conflicts of interest: author conflicts not declared |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "These 20 clusters were then randomly assigned to the screening and control (health education) arms." Comment: insufficient information pertaining to the method of sequence generation to permit judgement. |
| Allocation concealment (selection bias) | Unclear risk | Comment: insufficient information to permit judgement. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: probably not done. Each participant and health worker were aware of the arm to which the participant were allocated. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Comment: probably not done. Each participant and health worker were aware of the arm to which the participant were allocated. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: out of 75,360 eligible women listed in the screening arm 69,227 (90.88%), 62,755 (86.98%) and 59,543 (88.00%) women participated in the first, second, and third screening rounds for breast cancer. |
| Selective reporting (reporting bias) | High risk | Comment: these were interim analysis results as the trial is ongoing and cost‐effectiveness of CBE has not been reported |
| Other bias | Low risk | Comment: no other biases detected. |
Pace 2019.
| Study characteristics | ||
| Methods | Design: cluster RCT Cluster: health centres |
|
| Participants | Location/setting: 18 health centres in Burera District in Rwanda. Sample size: the health centre covers a population of 340,000. 1486 patients seen in intervention health centres with breast complaints; 315 patients seen in intervention health centres with breast complaints. Sex: females Mean age: 30.5 years in intervention clusters; 29.6 years in control clusters Inclusion criteria: not specified. Exclusion criteria: not specified. |
|
| Interventions | Intervention: the intervention consisted of instruction for community health workers (CHWs) in symptoms of breast cancer and messaging that community members should come to their local health centre for any breast symptoms. Health centre nurses were taught about signs and symptoms of benign breast disease and cancer and how to perform a CBE and were provided with simple clinical algorithms for managing breast symptoms and examination findings, with an emphasis on findings that need urgent hospital‐level evaluation. Control: no training was provided to the nurses and CHWs in the control health centres. Duration: 2 years. |
|
| Outcomes | Outcomes: health centre visit volume, visit characteristics, number of patients with breast concerns, health services received, cancer incidence, breast cancer stage. Timing of outcome assessment: 2 years. |
|
| Notes | Study start date: 18 April 2015. Study end date: 17 April 2017. Funding source: grants from the Breast Cancer Research Foundation (BCRF‐18‐149). Conflicts of Interest: Lydia E Pace reported stock and other ownership interests: in Firefly Health, Becton Dickinson; Lawrence N Shulman Research Funding came from Celgene. No other potential conflicts of interest were reported. Randomisation was done in two phases. In the first phase, 18 health centres were randomised into intervention and control clusters (7 intervention clusters and 11 control clusters). In the second phase, an additional 5 health centres from the control clusters were randomised to receive intervention while 6 health centres served as controls throughout the study period. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "To randomize HCs, we used random number generation with Excel (Microsoft Corporation, Redmond, WA) in 2 phases." Comment: randomisation was done in two phases. In the first phase, 18 health centres were randomised into intervention and control clusters (7 intervention clusters and 11 control clusters). In the second phase, an additional five health centres from the control clusters were randomised to receive intervention while six health centres served as controls throughout the study period. |
| Allocation concealment (selection bias) | Unclear risk | Comment: Insufficient information to permit any judgement. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: probably not done.Health workers were aware of the arm to which they were allocated. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Comment: probably not done.Health workers were aware of the arm to which they were allocated. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: all health centres allocated to the study were followed up. |
| Selective reporting (reporting bias) | Low risk | Trial registration number: NCT03919682. |
| Other bias | Low risk | No other biases detected. |
Pisani 2006.
| Study characteristics | ||
| Methods | Design: RCT Unit: cluster (202 health centres) |
|
| Participants | Location/setting: National Capital Region of Manila, Philippines Sample size: 151,168 women Dropouts/withdrawals: N/A Sex: females were included. Mean age: mean age of the compliers was 44.8 years and mean age of those who refused to be examined was 44.7 years. Inclusion criteria: women resident in 12 municipalities of the National Capital Region of Manila, Philippines and were included in the electoral rolls. Exclusion criteria: women who didn't live in the study area. Recruited nurses and midwives were trained in CBE using silicone breast models. Training was repeated for those who missed or over‐reported by 20% the lumps in the silicone models. |
|
| Interventions | Intervention group: (n = 151,168) 5 annual CBEs were performed in the National Capital Region of Manila, Philippines by the trained nurses/midwives with block randomisation amongst 202 clinics based on size of catchment population. First round of examinations took place in 24 months between January 1996 and December 1997, women were interviewed and CBE was implemented on 92% of participants. They were also instructed with BSE technique and given a leaflet in the local language explaining the importance of BSE. Registered cases of breast cancer were followed up until 2001 to assess their vital status. Control group: received no active intervention but were exposed to general health education campaigns carried out by municipal authorities and voluntary bodies. 138,392 women (92%), consented to receive the intervention over a period of 24 months between January 1996 and December 1997 with a 3‐year follow‐up of confirmed breast cancer cases. |
|
| Outcomes | Primary outcomes: number and cumulative incidence of breast cancers in the cohort of interviewed women. Secondary outcomes: diagnostic follow‐up for those with breast cancer. Timing of outcome assessment: outcomes were assessed after intervention was implemented. Because only one screening round was carried out, sensitivity, specificity and predictive value were calculated using as gold standard the incident cases identified by the registries in 2 years from screening examination, including those diagnosed at the time of testing. |
|
| Notes | Study start date: 1995 Study end date: 2001 Conflicts of interest: author conflicts not declared. Outcome data are only presented by arm of intervention and data for the control arm is not provided. The intervention was discontinued after the first round of screening due to non‐compliance of follow up by the participating women. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "These were randomly assigned to intervention or control arm by block randomization." Comment: insufficient information pertaining to the method of sequence generation to permit judgement. |
| Allocation concealment (selection bias) | Unclear risk | Comment: insufficient information to permit judgement. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: probably not done. Each participant and health worker were aware of the arm to which the participant were allocated. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Comment: probably not done. Each participant and health worker were aware of the arm to which the participant were allocated. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: amongst the women detected positive for lump after first round, 35% completed diagnostic follow‐up, whereas 42.4% actively refused further investigation even with home visits, and 22.5% were not traced. |
| Selective reporting (reporting bias) | High risk | Comment: some outcomes like mortality and down staging were not reported as study was aborted. |
| Other bias | High risk | Comment: due to a very low compliance with clinical follow‐up, the intervention ceased after completion of the first round of examinations. |
Sankaranarayanan 2011.
| Study characteristics | ||
| Methods | Design: RCT Unit: 275 clusters |
|
| Participants | Location/setting: Trivandrum district (Kerala, India) Sample size: 115,652 healthy women between 30 and 69 years. Dropouts/withdrawals: not provided. Sex: females were included. Mean age: not provided. Inclusion criteria: healthy women between the ages of 30 to 69 with intact breasts and no history of breast cancer. Exclusion criteria: unhealthy women, women with history of breast cancer, women who did not fit the age bracket necessary. Female health workers with a bachelor’s degree were trained in creating breast awareness and CBE during a 3‐week structured course at the RCC using silicone breast models followed by visual inspection and palpation of women with normal breasts, fibroadenosis, benign tumours and cancers, followed by periodic reorientation courses. The training included communication skills; visual inspection skills to assess breasts for asymmetry, visible lumps, skin changes, oedema, nipple retraction, discharge. |
|
| Interventions | Intervention group: 3 rounds of triennial CBE were performed to evaluate if this reduces the incidence rate of advanced disease and breast cancer mortality (133 clusters; n = 55,844). Control group: received education on cervical cancer prevention and information on how to access cervical treatment and screening (142 clusters; n = 59,808). Performance characteristics (sensitivity, specificity, false‐positive rate, and positive predictive value) of CBE were evaluated. An intention‐to‐treat analysis was performed for the comparison of incidence rates between the intervention and control groups. |
|
| Outcomes | Primary outcome: reduction of incidence rate of advanced disease incidence and breast cancer mortality. Secondary outcome: sensitivity, specificity, false‐positive rate, and positive predictive value of CBE and age standardised incidence rates for early‐stage (stage IIA or lower) breast cancer Timing of outcome assessment: registry staff, who did not know of study group assignments, collected data on the date of diagnosis, stage, treatment, and vital status of breast cancer patients by actively visiting hospitals and laboratories where breast cancers are diagnosed and treated and examined the cause of death using information obtained from the municipal death registration offices, hospital records, and house visits. The screening project staff then matched the breast cancer cases and deaths with the study database. |
|
| Notes | Study start date: January 2006 Study end date: May 2009 Funding Source: International Agency for Research on Cancer (Lyon, France) (grant number SCR/05/04 to KR and ST) Conflicts of interest: author conflicts not declared. Mortality data will only be available after completion of 3 rounds of screening. Further follow‐up will clarify whether earlier detection in the intervention group represents over‐diagnosis or a lead time bias |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "Clusters were randomly assigned to two groups: 133 clusters (55844 women) in the intervention group and 142 clusters (59808 women) in the control group." Comment: insufficient information pertaining to the method of sequence generation to permit judgement. |
| Allocation concealment (selection bias) | Unclear risk | Comment: insufficient information to permit judgement. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Comment: probably not done. Each participant and health worker were aware of the arm to which the participant were allocated. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Comment: probably not done. Each participant and health worker were aware of the arm to which the participant were allocated. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: of the CBE‐positive women, 49.1% attended the clinic. |
| Selective reporting (reporting bias) | Unclear risk | Comment: trial registration not found. |
| Other bias | Low risk | This is an ongoing trial. |
Abbreviations: CBE: clinical breast examination; CHW: community health workers; RCT: randomised controlled trial
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Abu Awwad 2021 | Did not meet the criteria on study design: excluded as it is a cross‐sectional design and not a RCT |
| Al‐Mulhim 2003 | Study was excluded for not meeting the study design criteria, and lacked the type of participants of interest to our review (i.e. trained versus non‐trained health workers) |
| Alameer 2019 | The study did not provide training to health workers, and was conducted in a high‐income country (HIC). |
| Alipour 2014 | The study did not have a control group. Both the study groups received breast screening information: one group via class, and the other group via short message service. |
| Bain 2018 | Study did not meet the study design criteria. It was a descriptive study of a breast cancer early detection model |
| Bawazir 2019 | Study did not meet the study design criteria. It was a cross‐sectional study |
| Bird 1998 | Study did not meet the criteria for location of study: it was conducted in the USA (not a LMIC). Also, it investigated recognition, receipt, and screening‐interval maintenance of CBE (not down staging and other secondary outcomes of interest) |
| Bittencourt 2019 | Study did not meet the criteria on study design. It is a pre‐post evaluation. |
| Buribekova 2018 | It did not meet the study design criteria for inclusion. It is a descriptive study of a breast cancer early detection model that includes training of family medicine nurses through a trainer of trainers programme. It only had early anecdotal evidence of downstaging, which was not adequate for our analyses. In the absence of this information, it wasn’t possible to calculate the sensitivity of the CBE programme in detection of breast cancer (one of the secondary outcomes) |
| Ceber 2010 | Study did not meet the inclusion criteria on objectives. The study assessed the effectiveness of an educational programme for nurses and midwives regarding their own knowledge, practices and health beliefs. |
| Devi 2007 | It did not meet the study design criteria for inclusion. This was a before‐after study with no control group described. |
| Dinshaw 2007 | This is a duplicate report of one of the included studies (Mittra 2021) |
| El‐Mhamdi 2013 | The study did not meet the criteria for outcomes of interest. It was a study on knowledge, attitude and practice of breast cancer screening but no specific item on knowledge of CBE. |
| Heena 2019 | This study did not meet the study design criteria for inclusion. It is a cross‐sectional design and not a RCT or quasi‐experimental. |
| Jeyapaul 2020 | The study did not meet the criteria for study design. It was a community‐based programme evaluation without a comparison group. |
| Kardinah 2014 | The study did not meet the criteria for study design. It is neither a RCT, quasi RCT nor controlled before‐after design. |
| Kociyigit 2011 | The study did not meet the criteria for study design. It was a cross‐sectional survey to evaluate the level of knowledge about breast cancer and diagnostic methods in women who applied to a training and research hospital in a city centre for an ordinary health problem. |
| Kulkarni 2019 | The study did not meet the criteria for study design. It is a community‐based one‐time cancer screening programme. |
| Kurtz 1994 | The study did not meet the criteria for location of study. It was conducted in high‐income country and not an LMIC. It was also a pre‐post design without a control group. |
| Matthieu 2021 | The study did not meet the criteria for study design. It was an evaluation of an implementation programme without a control group. |
| Miller 2011 | The study did not meet the study design criteria. It was a narrative review of CBE. |
| Pace 2018 | This study did not meet the criteria for the study design. This was an intermittent time series without a control group. |
| Zotov 2003 | The study did not meet the study design criteria. |
Abbreviations: CBE: clinical breast examination; high‐income country; LMIC: low‐ and middle‐income country; RCT: randomised controlled trial
Differences between protocol and review
The published Cochrane Protocol did not specify the secondary outcomes (including accuracy of health worker‐performed CBE, CBE coverage, follow‐up, and breast cancer mortality) to be included in the summary of findings table (Sayed 2017). However, we included these outcomes in the summary of findings table at review stage, in consideration of the clinical importance of these outcomes to decision‐makers.
We added two new authors at review stage: Nwosu N and Mutebi MC.
Contributions of authors
Drafted the protocol: RAS, SS, AN, PO, ASM.
Selected studies: SS, PO, RAS, ASM, MCM.
Extracted data from studies: PO, ASM, SS, MCM, NN
Entered data into Review Manager 2023: ASM, PO, SS, AN, NN.
Carried out the analysis: RAS, AN.
Interpreted the analysis: RAS, AN, SS, ASM.
Drafted the final review: RAS, SS, AN, NN.
Resolved disagreements: RAS, SS, AN.
Updated the search: RAS, SS, AN, NN.
All authors read and approval the final review version prior to publication.
Sources of support
Internal sources
No sources of support provided
External sources
No sources of support provided
Declarations of interest
SS: none known. SS works as a pathologist at Aga Khan University Hospital. AN: none known. NN: none known. MCM: none known. MCM works as a breast surgical oncologist at Aga Khan University Hospital and has published an opinion piece on the topic of 'Effectiveness of an abbreviated training program for health workers in breast cancer awareness' in 2013. PO: none known. ASM: none known. RAS: none known.
New
References
References to studies included in this review
Mittra 2021 {published data only}
- Mittra I, Mishra GA, Dikshit RP, Gupta S, Kulkarni VY, Shaikh HK, et al. Effect of screening by clinical breast examination on breast cancer incidence and mortality after 20 years: prospective, cluster randomised controlled trial in Mumbai. BMJ 2021;372:n256. [DOI: 10.1136/bmj.n256] [DOI] [PMC free article] [PubMed] [Google Scholar]
Pace 2019 {published data only}
- Pace LE, Dusengimana JM, Shulman LN, Schleimer LE, Shyirambere C, Rusangwa C, et al. Cluster randomized trial to facilitate breast cancer early diagnosis in a rural district of Rwanda. Journal of Global Oncology 2019;5:1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
Pisani 2006 {published data only}
- Pisani P, Parkin DM, Ngelangel C, Esteban D, Gibson L, Munson M, et al. Outcome of screening by clinical examination of the breast in a trial in the Philippines. International Journal of Cancer 2006;118(1):149-54. [DOI] [PubMed] [Google Scholar]
Sankaranarayanan 2011 {published data only}
- Sankaranarayanan R, Ramadas K, Thara S, Muwonge R, Prabhakar J, Augustine P, et al. Clinical breast examination: preliminary results from a cluster randomised controlled trial in India. Journal of the National Cancer Institute 2011;103(19):1476-80. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Abu Awwad 2021 {published data only}
- Abu Awwad D, Hossain SZ, Mackey M, Brennan P, Adam S. Optimising awareness and knowledge of breast cancer screening for nurses through train-the-trainer workshops. Journal of Cancer Education 2022;37(4):1209-19. [DOI] [PubMed] [Google Scholar]
Alameer 2019 {published data only}
- Alameer A, Mahfouz MS, Alamir Y, Ali N, Darraj A. Effect of health education on female teachers’ knowledge and practices regarding early breast cancer detection and screening in the Jazan Area: a quasi-experimental study. Journal of Cancer Education 2019;34(5):865-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
Alipour 2014 {published data only}
- Alipour S, Jannat F, Hosseini L. Teaching breast cancer screening via text messages as part of continuing education for working nurses: a case-control study. Asian Pacific Journal of Cancer Prevention 2014;15(14):5607-9. [DOI] [PubMed] [Google Scholar]
Al‐Mulhim 2003 {published data only}
- Al-Mulhim AS, Sultan M, Al-Mulhim FM, Al-Wehedy A, Ali AM, Al-Suwaigh A, et al. Accuracy of the “triple test” in the diagnosis of palpable breast masses in Saudi females. Annals of Saudi Medicine 2003;23(3-4):158-61. [DOI] [PubMed] [Google Scholar]
Bain 2018 {published data only}
- Bain C, Constant TH, Contreras I, Vega AMB, Jeronimo J, Tsu V. Model for early detection of breast cancer in low-resource areas: the experience in Peru. Journal of Global Oncology 2018;4:1-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Bawazir 2019 {published data only}
- Bawazir A, Bashateh N Jradi H, Breik AB. Breast cancer screening awareness and practices among women attending primary health care centers in the Ghail Bawazir District of Yemen. Clinical Breast Cancer 2019;19(1):e20-9. [DOI] [PubMed] [Google Scholar]
Bird 1998 {published data only}
- Bird JA, McPhee SJ, Ha NT, Le B, Davis T, Jenkins CN. Opening pathways to cancer screening for Vietnamese-American women: lay health workers hold a key. Preventive Medicine 1998;27(6):821-9. [DOI] [PubMed] [Google Scholar]
Bittencourt 2019 {published data only}
- Bittencourt L, Scarinci IC. Training community health workers to promote breast cancer screening in Brazil. Health Promotion International 2019;34(1):95-101. [DOI] [PubMed] [Google Scholar]
Buribekova 2018 {published data only}
- Buribekova R, Shukurbekova I, Ilnazarova S, Jamshevov N, Guldarbogh S, Sayani S, et al. Promoting clinical breast evaluations in a lower middle-income country setting: an approach toward achieving a sustainable breast health program. Journal of Global Oncology 2018;4:1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Ceber 2010 {published data only}
- Ceber E, Turk M, Ciceklioglu M. The effects of an educational program on knowledge of breast cancer, early detection practices and health beliefs of nurses and midwives. Journal of Clinical Nursing 2010;19(15-16):2363-71. [DOI] [PubMed] [Google Scholar]
Devi 2007 {published data only}
- Devi BCR, Tang TS, Corbex M. Reducing by half the percentage of late-stage presentation for breast and cervix cancer over 4 years: a pilot study of clinical downstaging in Sarawak, Malaysia. Annals of Oncology 2007;18:1172-6. [DOI] [PubMed] [Google Scholar]
Dinshaw 2007 {published data only}
- Dinshaw K, Mishra G, Shastri S, Badwe R, Kerkar R, Ramani S, et al. Determinants of compliance in a cluster randomised controlled trial on screening of breast and cervix cancer in Mumbai, India. 1. Compliance to screening. Oncology 2007;73(3-4):145-53. [DOI] [PubMed] [Google Scholar]
El‐Mhamdi 2013 {published data only}
- El-Mhamdi S, Bouanene I, Mhirsi A, Sriha A, Salem BK, Soltani MS. Women’s knowledge, attitudes and practice about breast cancer screening in the region of Monastir (Tunisia). Australian Journal of Primary Health 2013;19:68-73. [DOI] [PubMed] [Google Scholar]
Heena 2019 {published data only}
- Heena H, Durrani S, Riaz M, AlFayyad I, Tabasim R, Parvez G, et al. Knowledge, attitudes, and practices related to breast cancer screening among female health care professionals: a cross sectional study. BMC Women’s Health 2019;19(1):122. [DOI] [PMC free article] [PubMed] [Google Scholar]
Jeyapaul 2020 {published data only}
- Jeyapaul S, Oommen AM, Cherian AG, Marcus TA, Malini T, Prasad JH, et al. Feasibility, uptake and real-life challenges of a rural cervical and breast cancer screening program in Vellore, Tamil Nadu, South India. Indian Journal of Cancer 2021;58(3):417-24. [DOI] [PubMed] [Google Scholar]
Kardinah 2014 {published data only}
- Kardinah D, Anderson BO, Duggan C, Ali IA, Thomas DB. Short report: limited effectiveness of screening mammography in addition to clinical breast examination by trained nurse midwives in rural Jakarta, Indonesia. International Journal of Cancer 2014;134(5):1250-5. [DOI] [PubMed] [Google Scholar]
Kociyigit 2011 {published data only}
- Kocyigit O, Erel S, Kismet K, Kilicoglu B, Sabuncuoglu MZ, Akkus MA. Knowledge about breast cancer, mammography and breast self examination in women who applied to outpatient clinics: a study conducted in the city center. Nobel Medicus 2011;7(2):19-25. [Google Scholar]
Kulkarni 2019 {published data only}
- Kulkarni SV, Mishra GA, Dusane RR. Determinants of compliance to breast cancer screening and referral in low socio-economic regions of urban India. International Journal of Preventive Medicine 2019;10(1):84. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kurtz 1994 {published data only}
- Kurtz ME, Kurtz JC, Given B, Given CC. Promotion of breast cancer screening in a work site population. Health Care for Women International 1994;15(1):31-42. [DOI] [PubMed] [Google Scholar]
Matthieu 2021 {published data only}
- Mathieu KM, YouYou TG, Hicks ML, Mutombo A, Anaclet MM, Sylvain MK, et al. Building a breast cancer detection and treatment platform in the Democratic Republic of the Congo by integrating training, service and infrastructure development. Ecancermedicalscience 2021;15:1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
Miller 2011 {published data only}
- Miller AB, Baines CJ. The role of clinical breast examination and breast self-examination. Preventive Medicine 2011;53(3):118-20. [DOI] [PubMed] [Google Scholar]
Pace 2018 {published data only}
- Pace LE, Dusengimana JV, Keating NL, Hategekimana V, Rugema V, Bigirimana JB, et al. Impact of breast cancer early detection training on Rwandan health workers’ knowledge and skills. Journal of Global Oncology 2018;4:1-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Zotov 2003 {published data only}
- Zotov V, Shyyan R, PATH Breast Cancer Assistance Program. Introduction of breast cancer screening in Chernihiv Oblast in the Ukraine: report of a PATH Breast Cancer Assistance Program experience. Breast Journal 2003;9(Suppl 2):S75-80. [DOI] [PubMed] [Google Scholar]
Additional references
Abuidris 2013
- Abuidris DO, Elsheikh A, Ali M, Musa H, Elgaili E, Ahmed AO, et al. Breast-cancer screening with trained volunteers in a rural area of Sudan: a pilot study. Lancet Oncology 2013;14(4):363-70. [DOI] [PubMed] [Google Scholar]
Alharbi 2012
- Alharbi NA, Alshammari MS, Almutairi BM, Makboul G, El-Shazly MK. Knowledge, awareness, and practices concerning breast cancer among Kuwaiti female school teachers. Alexandria Journal of Medicine 2012;48(1):75-82. [Google Scholar]
Awwad 2020
- Awwad DA, Hossain SZ, Mackey M, Brennan P, Adam S. Optimising awareness and knowledge of breast cancer screening for nurses through train-the-trainer workshops. Journal of Cancer Education 2022;37(4):1209-19. [DOI] [PubMed] [Google Scholar]
Bancej 2003
- Bancej C, Decker K, Chiarelli A, Harrison M, Turner D, Brisson J. Contribution of clinical breast examination to mammography screening in the early detection of breast cancer. Journal of Medical Screening 2003;10(1):16-21. [DOI] [PubMed] [Google Scholar]
Bittencourt 2017
- Bittencourt L, Scarinci IC. Training community health workers to promote breast cancer screening in Brazil. Health Promotion International 2017;34(1):95-101. [DOI] [PubMed] [Google Scholar]
Campbell 2020
- Campbell M, McKenzie JE, Sowden A, Katikireddi SV, Brennan SE, Ellis S, et al. Synthesis without meta-analysis (SWiM) in systematic reviews: reporting guideline. BMJ 2020;368:l6890. [DOI] [PMC free article] [PubMed] [Google Scholar]
DerSimonian 1986
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled Clinical Trials 1986;7:177-88. [DOI] [PubMed] [Google Scholar]
Devi 2007
- Devi BC, Tang TS, Corbex M. Reducing by half the percentage of late-stage presentation for breast and cervix cancer over 4 years: a pilot study of clinical downstaging in Sarawak, Malaysia. Annals of Oncology 2007;18(7):1172-6. [DOI] [PubMed] [Google Scholar]
Dey 2014
- Dey S. Preventing breast cancer in LMICs via screening and/or early detection: the real and the surreal. World Journal of Clinical Oncology 2014;5(3):509-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
Duffy 2020
- Duffy S, Vulkan D, Cuckle H, Parmar D, Sheikh S, Smith R, et al. Annual mammographic screening toreduce breast cancer mortality in womenfrom age 40 years: long-term follow-upof the UK Age RCT. Health Technology Assessment 2020;24(55):1-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
Duggan 2020
- Duggan C, Dvaladze A, Rositch AF, Ginsburg O, Yip CH, Horton S, et al. The Breast Health Global Initiative 2018 Global Summit on improving breast healthcare through resource-stratified phased implementation: methods and overview. Cancer 2020;126(Suppl 10):2339-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
Egger 1997
- Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315(7109):629-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
Farid 2014
- Farid ND, Aziz NA, Al-Sadat N, Jamaludin M, Dahlui M. Clinical breast examination as the recommended breast cancer screening modality in a rural community in Malaysia: what are the factors that could enhance its uptake? PLOS ONE 2014;9(9):e106469. [DOI] [PMC free article] [PubMed] [Google Scholar]
Forouzanfar 2011
- Forouzanfar MH, Foreman KJ, Delossantos AM, Lozano R, Lopez AD, Murray CJ, et al. Breast and cervical cancer in 187 countries between 1980 and 2010: a systematic analysis. Lancet 2011;378(9801):1461-84. [DOI] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- GRADEpro GDT. Version accessed 13 March 2023. Hamilton (ON): McMaster University (developed by Evidence Prime). Available at gradepro.org.
Guyatt 2008
- Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008;336(7650):924-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Harbord 2006
- Harbord RM, Egger M, Sterne JA. A modified test for small study effects in meta-analyses of controlled trials with binary endpoints. Statistics in Medicine 2006;25(20):3443-57. [DOI] [PubMed] [Google Scholar]
Harford 2008
- Harford J, Azavedo E, Fischietto M, Breast Health Global Initiative Healthcare Systems Panel. Guideline implementation for breast healthcare in low- and middle-income countries: breast healthcare program resource allocation. Cancer 2008;113(8 Suppl):2282-96. [DOI] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from training.cochrane.org/handbook/archive/v5.1/.
Jedy‐Agba 2016
- Jedy-Agba E, McCormack V, Adebamowo C, Dos-Santos-Silva I. Stage at diagnosis of breast cancer in sub-Saharan Africa: a systematic review and meta-analysis. Lancet Global Health 2016;4(12):e923-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kejela 2021
- Kejela S. Cost-effectiveness of screening mammography in a low income country: a Markov simulation analysis. BMC Medical Imaging 2021;21(1):162. [DOI] [PMC free article] [PubMed] [Google Scholar]
Knaul 2012
- Knaul FM, Bhadelia A, Gralow J, Arreola-Ornelas H, Langer A, Frenk J. Meeting the emerging challenge of breast and cervical cancer in low- and middle-income countries. International Journal of Gynecologists and Obstetrics 2012;119(Suppl 1):S85-8. [DOI] [PubMed] [Google Scholar]
Kumar 2019
- Kumar A, Bhagabaty SM, Tripathy JP, Selvaraj K, Purkayastha J, Singh R. Delays in diagnosis and treatment of breast cancer and the pathways of care: a mixed methods study from a tertiary cancer centre in North East India. Asian Pacific Journal of Cancer Prevention 2019;20(12):3711-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kösters 2003
- Kösters JP, Gøtzsche PC. Regular self-examination or clinical examination for early detection of breast cancer. Cochrane Database of Systematic Reviews 2003, Issue 2. Art. No: CD003373. [DOI: 10.1002/14651858.CD003373] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lauby‐Secretan 2015
- Lauby-Secretan B, Scoccianti C, Loomis D, Benbrahim-Tallaa L, Bouvard V, Bianchini F, et al, International Agency for Research on Cancer Handbook Working Group. Breast-cancer screening: viewpoint of the IARC Working Group. New England Journal of Medicine 2015;372(24):2353-8. [DOI] [PubMed] [Google Scholar]
LeBan 2021
- LeBan K, Kok M, Perry HB. Community health workers at the dawn of a new era: 9. CHWs’ relationships with the health system and communities. Health Research Policy and Systems 2021;19(Suppl 3):116. [DOI] [PMC free article] [PubMed] [Google Scholar]
Li Cheng 2015
- Cheng ML, Ling DY, Nanu P KP, Nording H, Lim CH. Factors influencing late stage of breast cancer at presentation in a district Hospital - Segamat Hospital, Johor. Medical Journal of Malaysia 2015;70(3):148-52. [PubMed] [Google Scholar]
Martei 2022
- Martei YM, Dauda B, Vanderpuye V. Breast cancer screening in sub-Saharan Africa: a systematic review and ethical appraisal. BMC Cancer 2022;22(1):203. [DOI] [PMC free article] [PubMed] [Google Scholar]
Menes 2021
- Menes TS, Coster D, Coster D, Shenhar-Tsarfaty S. Contribution of clinical breast exam to cancer detection in women participating in a modern screening program. BMC Women's Health 2021;21(1):368. [DOI] [PMC free article] [PubMed] [Google Scholar]
Miller 2008
- Miller AB. Practical applications for clinical breast examination (CBE) and breast self-examination (BSE) in screening and early detection of breast cancer. Breast Care 2008;3(1):17-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
NCI 2016
- National Cancer Institute (NCI). NCI Dictionary of Cancer Terms. cancer.gov/publications/dictionaries/cancer-terms (accessed 23 November 2016).
Ngan 2020
- Ngan TT, Nguyen NT, Van Minh H, Donnelly M, O'Neill C. Effectiveness of clinical breast examination as a 'stand-alone' screening modality: an overview of systematic reviews. BMC Cancer 2020;20:1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
Obajimi 2013
- Obajimi MO, Ajayi IO, Oluwasola AO, Adedokun BO, Adeniji-Sofoluwe AT, Mosuro OA, et al. Level of awareness of mammography among women attending outpatient clinics in a teaching hospital in Ibadan, South-West Nigeria. BMC Public Health 2013;13:40. [DOI] [PMC free article] [PubMed] [Google Scholar]
Opoku 2012
- Opoku SY, Benwell M, Yarney J. Knowledge, attitudes, beliefs, behaviour and breast cancer screening practices in Ghana, West Africa. Pan African Medical Journal 2012;11:28. [PMC free article] [PubMed] [Google Scholar]
Pinder 2018
- Pinder LF, Henry-Tillman R, Linyama D, Kusweje V, Nzayisenga JB, Shibemba A, et al. Leverage of an existing cervical cancer prevention service platform to initiate breast cancer control services in Zambia: experiences and early outcomes. Journal of Global Oncology 2018;4:1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Pisani 2006
- Pisani P, Parkin DM, Ngelangel C, Esteban D, Gibson L, Munson M, et al. Outcome of screening by clinical examination of the breast in a trial in the Philippines. International Journal of Cancer 2006;118(1):149-54. [DOI] [PubMed] [Google Scholar]
Review Manager 2023 [Computer program]
- Review Manager Web (RevMan Web). Version 4.26.0. The Cochrane Collaboration, 2023. Available at revman.cochrane.org.
Rifkin 2014
- Rifkin SB. Examining the links between community participation and health outcomes: a review of the literature. Health Policy and Planning 2014;29(Suppl 2):ii98-106. [DOI] [PMC free article] [PubMed] [Google Scholar]
Sankaranarayanan 2011
- Sankaranarayanan R, Ramadas K, Thara S, Muwonge R, Prabhakar J, Augustine P, et al. Clinical breast examination: preliminary results from a cluster randomized controlled trial in India. Journal of the National Cancer Institute 2011;103(19):1476-80. [DOI] [PubMed] [Google Scholar]
Saslow 2004
- Saslow D, Hannan J, Osuch J, Alciati MH, Baines C, Barton M, et al. Clinical breast examination: practical recommendations for optimizing performance and reporting. CA: a Cancer Journal for Clinicians 2004;54(6):327-54. [DOI] [PubMed] [Google Scholar]
Sayed 2014
- Sayed S, Moloo Z, Wasike R, Bird P, Oigara R, Govender D, et al. Is breast cancer from Sub Saharan Africa truly receptor poor? Prevalence of ER/PR/HER2 in breast cancer from Kenya. Breast 2014;23(5):591-6. [DOI] [PubMed] [Google Scholar]
Sterne 2011
- Sterne JA, Sutton AJ, Ioannidis JP, Terrin N, Jones DR, Lau J, et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011;343:d4002. [DOI] [PubMed] [Google Scholar]
Sung 2021
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a Cancer Journal for Clinicians 2021;71(3):209-49. [DOI: 10.3322/caac.21660] [PMID: PMID: 33538338] [DOI] [PubMed] [Google Scholar]
Thomas 1997
- Thomas DB, Gao DL, Self SG, Allison CJ, Tao Y, Mahloch J, et al. Randomized trial of breast self-examination in Shanghai: methodology and preliminary results. Journal of the National Cancer Institute 1997;89(5):355-65. [DOI] [PubMed] [Google Scholar]
Torre 2015
- Torre AL, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent, Jemal A. Global cancer statistics, 2012. CA: a Cancer Journal for Clinicians 2015;65(2):87-108. [DOI] [PubMed] [Google Scholar]
Wadler 2011
- Wadler BM, Judge CM, Prout M, Allen JD, Geller AC. Improving breast cancer control via the use of community health workers in South Africa: a critical review. Journal of Oncology 2011;2011:150423. [DOI] [PMC free article] [PubMed] [Google Scholar]
World Bank 2021
- World Bank. The world by income and region. 2021. https://datatopics.worldbank.org/world-development-indicators/the-world-by-income-and-region.html.
World Health Organization 2017
- World Health Organization (WHO). Guide to cancer early diagnosis. 2017. Available at apps.who.int/iris/handle/10665/254500.
Zhang 2021
- Zhang Z, Rao R, Mango VL, Wilson-Gardner P, Vempalle S, Ojutiku O. Presentation of self-detected breast mass in minority women with limited access to care: can self-examination assist in early cancer detection? Clinical Imaging 2021;70:89-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to other published versions of this review
Sayed 2017
- Sayed S, Ngugi A, Ochieng P, Mwenda AS, Salam RA. Training health workers in clinical breast examination for early detection of breast cancer in low‐ and middle‐income countries. Cochrane Database of Systematic Reviews 2017, Issue 1. Art. No: CD012515. [DOI: 10.1002/14651858.CD012515] [DOI] [PMC free article] [PubMed] [Google Scholar]


