Abstract
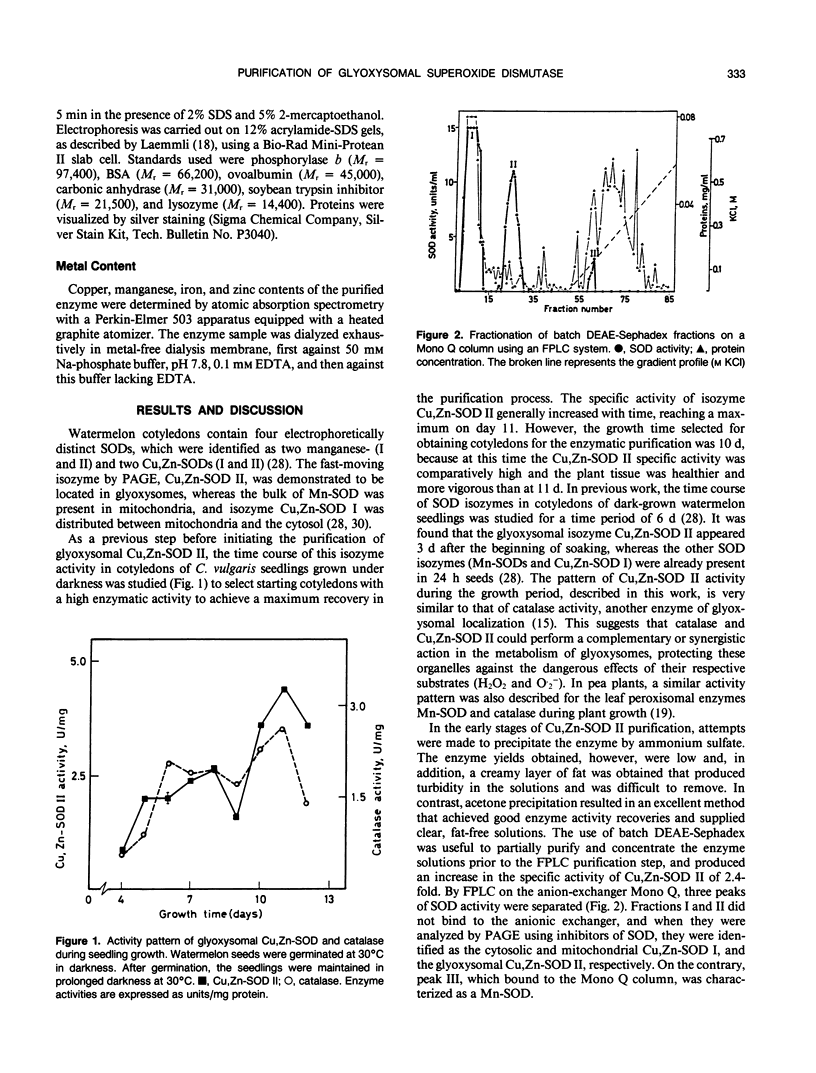
A glyoxysomal copper,zinc-containing superoxide dismutase (EC 1.15.1.1) was purified to homogeneity, for the first time, from watermelon cotyledons (Citrullus vulgaris Schrad.). The stepwise purification procedure consisted of acetone precipitation, batch anion-exchange chromatography, anion-exchange Fast Protein Liquid Chromatography and gel-filtration column chromatography. Pure copper,zinc-superoxide dismutase (Cu,Zn-SOD II) had a specific activity of 1211 units per milligram protein and was purified 400-fold, with a yield of 8 micrograms enzyme per gram cotyledon. The glyoxysomal Cu,Zn-SOD had a relative molecular weight of about 33,000 and was composed of two equal subunits of 16,500 Daltons. Metal analysis showed that the enzyme, unlike other Cu,Zn-SODs, contained 1 gram-atom Cu and 1 gram-atom Zn per mole dimer. No iron and manganese were detected. Ultraviolet and visible absorption spectra were reminiscent of other copper,zinc-superoxide dismutases.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aebi H. Catalase in vitro. Methods Enzymol. 1984;105:121–126. doi: 10.1016/s0076-6879(84)05016-3. [DOI] [PubMed] [Google Scholar]
- Barkley K. B., Gregory E. M. Tetrameric manganese superoxide dismutases from anaerobic Actinomyces. Arch Biochem Biophys. 1990 Jul;280(1):192–200. doi: 10.1016/0003-9861(90)90535-7. [DOI] [PubMed] [Google Scholar]
- Baum J. A., Chandlee J. M., Scandalios J. G. Purification and Partial Characterization of a Genetically-Defined Superoxide Dismutase (SOD-1) Associated with Maize Chloroplasts. Plant Physiol. 1983 Sep;73(1):31–35. doi: 10.1104/pp.73.1.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baum J. A., Scandalios J. G. Isolation and characterization of the cytosolic and mitochondrial superoxide dismutases of maize. Arch Biochem Biophys. 1981 Feb;206(2):249–264. doi: 10.1016/0003-9861(81)90089-8. [DOI] [PubMed] [Google Scholar]
- Beauchamp C., Fridovich I. Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem. 1971 Nov;44(1):276–287. doi: 10.1016/0003-2697(71)90370-8. [DOI] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Droillard M. J., Paulin A. Isozymes of Superoxide Dismutase in Mitochondria and Peroxisomes Isolated from Petals of Carnation (Dianthus caryophyllus) during Senescence. Plant Physiol. 1990 Nov;94(3):1187–1192. doi: 10.1104/pp.94.3.1187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duke M. V., Salin M. L. Purification and characterization of an iron-containing superoxide dismutase from a eucaryote, Ginkgo biloba. Arch Biochem Biophys. 1985 Nov 15;243(1):305–314. doi: 10.1016/0003-9861(85)90800-8. [DOI] [PubMed] [Google Scholar]
- Federico R., Medda R., Floris G. Superoxide Dismutase from Lens esculenta: Purification and Properties. Plant Physiol. 1985 Jun;78(2):357–358. doi: 10.1104/pp.78.2.357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giannopolitis C. N., Ries S. K. Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol. 1977 Feb;59(2):309–314. doi: 10.1104/pp.59.2.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- McCord J. M., Fridovich I. Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J Biol Chem. 1969 Nov 25;244(22):6049–6055. [PubMed] [Google Scholar]
- Puget K., Michelson A. M. Isolation of a new copper-containing superoxide dismutase bacteriocuprein. Biochem Biophys Res Commun. 1974 Jun 4;58(3):830–838. doi: 10.1016/s0006-291x(74)80492-4. [DOI] [PubMed] [Google Scholar]
- Reisner A. H. Gel protein stains: a rapid procedure. Methods Enzymol. 1984;104:439–441. doi: 10.1016/s0076-6879(84)04110-0. [DOI] [PubMed] [Google Scholar]
- Sandalio L. M., Del Río L. A. Intraorganellar distribution of superoxide dismutase in plant peroxisomes (glyoxysomes and leaf peroxisomes). Plant Physiol. 1988 Dec;88(4):1215–1218. doi: 10.1104/pp.88.4.1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- del Río L. A., Sandalio L. M., Palma J. M. A new cellular function for peroxisomes related to oxygen free radicals? Experientia. 1990 Oct 15;46(10):989–992. doi: 10.1007/BF01940651. [DOI] [PubMed] [Google Scholar]



