Abstract
Contextual and spatial systems facilitate changes in emotional memory regulation brought on by traumatic stress. Cholinergic basal forebrain (chBF) neurons provide input to contextual/spatial systems and although chBF neurons are important for emotional memory, it is unknown how they contribute to the traumatic stress effects on emotional memory. Clusters of chBF neurons that project to the prefrontal cortex (PFC) modulate fear conditioned suppression and passive avoidance, while clusters of chBF neurons that project to the hippocampus (Hipp) and PFC (i.e. cholinergic medial septum and diagonal bands of Broca (chMS/DBB neurons) are critical for fear extinction. Interestingly, neither Hipp nor PFC projecting chMS/DBB neurons are critical for fear extinction. The retrosplenial cortex (RSC) is a contextual/spatial memory system that receives input from chMS/DBB neurons, but whether this chMS/DBB-RSC circuit facilitates traumatic stress effects on emotional memory remain unexplored. Traumatic stress leads to neuroinflammation and the buildup of reactive oxygen species. These two molecular processes may converge to disrupt chBF circuits enhancing the impact of traumatic stress on emotional memory.
Keywords: medial septum, inflammation, oxidative species, fear conditioning, contextual conditioning, occasion-setting, extinction, cholinergic
Introduction
Mammalian organisms use contextual and spatial features to navigate their environment, learn associative rules, and regulate emotional expression 1–6. Cholinergic basal forebrain (chBF) neurons project to neural substrates that represent contexts and space (i.e. contextual/spatial systems) 7–10 and modulate physiological and behavioral processes that are dependent on these systems. Examples include cortical sensory plasticity 11–13, place field re-mapping in the hippocampus (Hipp) 14, and spatial learning and memory 15,16. Contextual/spatial systems are sensitive to traumatic stress and have been implicated in traumatic stress-induced psychiatric disorders such as post traumatic stress disorder (PTSD), especially with regard to traumatic stress effects on emotional memory regulation 2,17. This would suggest that chBF neurons could have a role in facilitating the impact traumatic stress has on emotional memory regulation. This is especially likely, because chBF neurons are sensitive to neuroinflammation and the buildup of reactive species; two molecular processes that emerge with traumatic stress. Also, chBF neurons are critical for emotional regulation as well (see below). Unfortunately, the potential role of chBF neurons in traumatic stress-induced disruptions in emotional memory regulation remains largely unexplored.
In this article, we review studies that point to a role for chBF neurons in traumatic stress effects on emotional memory regulation. First, we describe the impact traumatic stress has on contextual/spatial memory systems and how these traumatic stress effects can lead to changes in hormonal systems that regulate emotional memory and behavioral and physiological measures of emotional memory regulation. Next, we describe what is known about the role of chBF neurons in emotional memory regulation and how chBF neurons may form circuits with contextual/spatial memory systems to regulate emotional memory. A significant gap in the literature that emerges from a review of the literature is the lack of studies examining the role of the retrosplenial cortex (RSC, a contextual/spatial memory system) in traumatic stress effects on emotional memory regulation and chBF neuronal input to the RSC in regulating emotional memory.
In the last sections of the article, we review studies that demonstrate chBF neurons are sensitive to neuroinflammation and the buildup of reactive species, and that disruptions in the integrity of chBF neurons can lead to neuroinflammation. We discuss how traumatic stress could compromise the integrity of chBF neurons, which may lead to increased inflammation in efferent targets of these neurons. Via these mechanisms (i.e. neuroinflammation, oxidative stress) traumatic stress may act via chBF neurons to produce changes in contextual/spatial systems that lead to disruptions in emotional regulation.
2. Contextual/spatial memory systems in PTSD
Two contextual/spatial memory systems that have been consistently implicated in PTSD are the Hipp and prefrontal cortex (PFC) 1–3,5,18–20. The Hipp of PTSD patients is smaller than matched controls and this may emerge from smaller hippocampi as a result of trauma exposure 21–25. Alternatively, smaller hippocampi prior to trauma exposure may represent increased susceptibility to developing PTSD 26,27. Studies have consistently reported smaller PFC volumes in PTSD patients 17,28,29 and a recent consortium study has shown that several PFC regions (e.g. lateral orbitofrontal gyri, left superior temporal gyrus) are smaller in PTSD patients 30.
The inability to regulate fear and anxiety that stem from traumatic events is a core feature of PTSD 31. This deficit in emotional regulation may not be restricted to fear generated from traumatic memory (i.e. re-experiencing), but could also represent a broader deficit in the ability to regulate fear memory 32,33. To examine fear memory formation, expression, and regulation, Pavlovian fear conditioning experiments are often utilized. Pavlovian fear conditioning is a relatively simple behavioral paradigm where a neutral stimulus such as a tone or a light is paired with an aversive event, such as electrical shock 19,34–37. The neutral stimulus, referred to as the cued conditioned stimulus (CS), is paired once or several times with the aversive event, referred to as the unconditioned aversive stimulus (UCS). When a specific cued CS is paired with a UCS (specifically co-terminates with the UCS), the behavioral protocol is referred to as cued (or delayed) fear conditioning 35,36,38. The CS can also be a distinct space or arrangement of objects (e.g. type of background noise, odor, or texture places into a distinct space, virtual reality experience) that form a context 1,2,39. When the CS is a context, fear conditioning is referred to as contextual fear conditioning 1,2,19,36,39. Repeated exposure of a feared CS in the absence of UCS presentation is referred to as fear extinction, which results in the formation of a separate memory that inhibits fear memory expression 1,33,40–45. After the formation of extinction memory, fear and extinction memory compete for expression (i.e. emotional memory regulation).
Below, we review clinical and preclinical studies that have examined the roles of the Hipp and PFC in traumatic stress effects on emotional memory regulation. Adrenal hormones are critical for memory consolidation 46–49, PTSD is characterized by changes in the hypothalamic-pituitary-adrenal (HPA) axis, and contextual/spatial systems regulate HPA axis activity (see below). For these reasons, we review the potential role of traumatic stress-induced changes in HPA axis function that may lead to changes in emotional memory regulation.
2.1. Traumatic stress, HPA axis, and emotional memory
Glucocorticoid receptors (GRs) are densely expressed in the Hipp and PFC. Increased glucocorticoid (e.g. corticosterone, cortisol) release from the adrenal glands into the circulation and binding of glucocorticoids to GRs in the Hipp and PFC influence the HPA axis to subsequently lower the overall levels of circulating glucocorticoids (i.e. negative feedback of the HPA axis) 50–55. Enhanced negative feedback of the HPA axis is often observed in PTSD patients 56–62 (though for contradictory findings see 63,64). Findings of enhanced negative feedback of the HPA axis in PTSD suggest enhanced GR sensitivity in the HPA axis and/or substrates that regulate the HPA axis 26,65. While tangential, studies examining GR dynamics on lymphocytes support this assertion. GR binding on lymphocytes can be enhanced in PTSD patients 66,67, GR levels on lymphocytes can be elevated in PTSD patients 68,69, and enhanced lymphocyte GR levels and binding represent risk factors for developing PTSD after trauma exposure 66,70. Together these findings suggest that enhanced negative feedback of the HPA axis is a feature of PTSD and is driven by enhanced GR expression.
Preclinical experiments are useful in examining neurobiological mechanisms of psychiatric disorders and can be used to further explore how increased GR sensitivity within the brain leads to specific physiological and behavioral effects 71–73. The single prolonged stress (SPS) rodent model of traumatic stress refers to the serial application of multiple stressors, followed by a quiescent period (stress incubation period). The stressors most commonly used for SPS are restraint, forced swim, and ether exposure with stress incubation periods that are at least seven days, but can be as long as 30 days 71–75. Studies have consistently reported that SPS enhances negative feedback of the HPA axis 76–81 and GR sensitivity (most often by enhancing GR levels) in the dorsal Hipp (dHipp, which is believed to correspond to the region of the Hipp in humans that is critical for representation of contexts and space 2,82–85) 78,81,86–95. Furthermore, antagonizing GRs during SPS blocks both upregulation of dHipp GRs and enhancements in negative feedback of the HPA axis 94,96. These findings directly implicate SPS-enhanced dHipp GR expression with SPS-enhanced negative feedback of the HPA axis. GR enhancements in the PFC 90 and ventral Hipp (vHipp) 97 have been reported, but these effects are not as consistently observed 88,90. It is still not clear if SPS differentially enhances dHipp GR levels in the cytoplasm or nucleus of cells as reports are inconsistent 88,94. It should be noted that SPS also affects GR function in cells within brain regions outside of the Hipp and PFC 88 97.
Enhanced negative feedback of the HPA axis may be linked to enhancements in dHipp GR levels within PTSD and within the SPS model. Furthermore, enhanced dHipp GR levels are also associated with broader deficits in hippocampal function within the SPS model, with some studies implicating enhanced dHipp GRs with emotional memory dysregulation in the SPS model (see below).
2.2. Emotional memory regulation
When PTSD patients are subjected to fear conditioning and extinction procedures, these patients consistently exhibit enhanced fear memory 98–100 and/or an inability to acquire or maintain extinction memory 32,101–105. After acquisition of fear extinction, a CS (which at this time is typically referred to as an extinguished CS) can signal either threat or safety. To disambiguate the meaning of an extinguished CS, organisms can use contextual information to determine when a CS potentially signals threat or safety 1,3. In this way, contextual features can act as an occasion-setter and prime the retrieval of extinction memory (i.e. contextual occasion-setting 1,3). This suggests that disruptions in contextual occasion-setting could lead to deficits in emotional regulation (e.g. increased re-experiencing symptoms) in PTSD patients. While less studied, two reports suggest that PTSD may be associated with a disruption in contextual occasion-setting 103,105. In Steiger et al., (2015) valence ratings in response to a feared CS presented in a context that was also paired with electrical shock was resistant to extinction in PTSD patients when compared to non-trauma controls. In Garfinkel et al., (2014) PTSD patients exhibited deficits in the maintenance of extinction in a safe context. In a threat context, PTSD patients showed lower levels of conditioned responding to a feared CS. These results could be interpreted to mean that PTSD patients were unable to use contextual information to regulate fear responses 103.
Both the PFC 18,106–108 and Hipp 35,36,84 are critical for representing contextual information and the rodent dHipp is also critical for contextual occasion-setting 1,2,109–111. Regions of the PFC also have direct roles in regulating expression of fear by inhibiting neural activity within amygdala nuclei 17,40,43,112. From these observations it follows that the PFC and Hipp would have roles in facilitating changes in emotional memory regulation that characterize PTSD. Results from clinical studies that utilize fMRI techniques during fear conditioning and extinction experiments support this hypothesis. PTSD is associated with decreased BOLD signal in regions of the PFC (e.g. ventromedial PFC (vmPFC)) and Hipp during acquisition and/or recall of extinction 103,113,114. During extinction testing, exaggerated amygdala BOLD signal has been observed 115–117. A similar pattern of neural activity (i.e. decreased BOLD signal in the PFC and/or increased signal in amygdala nuclei) are also observed when protocols that require inhibition of behavioral or emotional output 118–120. Because PFC inhibition of amygdala nuclei are critical for regulation of fear 42,43,121–123, a deficit in PFC-mediated inhibition of amygdala nuclei is believed to represent a critical circuit through which dysregulation of fear memory manifests in PTSD 113,114,124,125
In clinical experiments decreased PFC and Hipp BOLD signals have been observed when an extinguished CS is presented in a threat context (i.e. fear renewal). During fear renewal, contextual information can be used to determine if a CS signals threat and fear responses to an extinguished CS typically re-emerge. Diminished BOLD activity in the PFC and Hipp during fear renewal could suggest deficits in contextual occasion-setting 103. However, this effect has also been observed when an extinguished CS is presented in a safe context 113,114 and it should be noted that at least one study has reported enhanced Hipp BOLD activity during contextual fear conditioning in PTSD patients 105. While it is possible that deficits in emotional memory regulation within PTSD emerges because of deficits in contextual processing brought on by changes in neural function within the PFC and Hipp of PTSD patients, further research is needed to better define how (or if) deficits in PFC and Hipp function within PTSD patients lead to deficits in emotional memory regulation via deficits in contextual processing.
The preclinical literature also supports a role of the medial PFC (mPFC) and dHipp in deficits in emotional memory regulation brought on by traumatic stress. This has been explored extensively using the SPS model of traumatic stress. SPS leads to decreased excitatory tone within the mPFC 126,127 and LTP in the CA1 region of the dHipp 91. Enhancements in dHipp GR expression are associated with enhanced contextual fear memory and deficits in maintenance of extinguished fear 87,90,94,128,129. During acquisition of extinction memory neural activity is enhanced in the mPFC 122,130 and SPS disrupts this enhancement specifically within the infralimbic cortex (IL) of the mPFC 130,131; a PFC region that is critical for extinction memory 40,43. Both optogenetic 132 and chemogenetic 131 stimulation of the IL after SPS attenuates SPS effects on extinction memory. These findings suggest that the IL is a locus through which traumatic stress-induced deficits in the maintenance of extinction manifests. In most experiments where the effects of SPS on extinction memory are examined, fear conditioning occurs in one context (e.g. threat context) and extinction training and testing occurs in another context (e.g. safe context) 74,87,90,92,129,133. Because organisms use contextual features to determine if a CS represents threat or safety (see above), the experimental design adopted in many SPS conditioning experiments raise the possibility that deficits in the maintenance of extinction are driven by deficits in the ability to use contextual features to assign meaning to a cued CS (i.e. contextual occasion-setting). One study has observed the IL is important for hierarchal occasion-setting 134, but more research is needed to determine the role of the IL (if any) in contextual occasion-setting and if traumatic stress effects on extinction maintenance are linked to traumatic stress effects on IL modulation of contextual occasion-setting. This is particularly so, because alternative explanations of the role of the IL in facilitating SPS deficits in the maintenance of extinction exists. The effects of SPS on IL neural activity could be construed as a failure to inhibit neural activity in subcortical substrates such as the lateral amygdala and/or central nucleus of the amygdala. This would represent a deficit in extinction memory formation and/or retrieval. In support of this interpretation, SPS disrupts functional connectivity between the mPFC and amygdala 127 and specifically does this during acquisition of extinction memory 135. Still other possibilities exist. The results of other studies point to an enhancement in the resistance of fear memory to extinction as the cause of extinction memory deficits within the SPS model 129,136. It should be noted that these mechanisms are not mutually exclusive. It is possible that traumatic stress, acting via multiple behavioral mechanisms facilitated by overlapping, but distinct, circuits lead to fear memory that is difficult to regulate.
While prior research has examined the contribution of the PFC and Hipp to traumatic stress effects on emotional memory regulation, a contextual/spatial system that has received very little attention is the RSC. The RSC is critical for contextual fear conditioning 137–139, binding elements within an environment into a single context (i.e. configural processing) 140, and association of elements in the absence of reinforcement (i.e. sensory preconditioning) 141. The RSC is connected to brain regions critical for spatial navigation 137,142,143 and neural activity within the RSC may serve as a, ‘homing signal’, during spatial navigation 144. As such, it is a contextual/spatial system, though the impact of traumatic stress on the RSC remains under explored.
To address this, we conducted an experiment where SPS and unstressed (i.e. controls) rats were subjected to fear conditioning, extinction training, and extinction testing (CS-fear). Fear conditioning comprised of five auditory CSs (10s, 2kHz) and footshocks (1s, 1mA) presentations where CSs and footshocks co-terminated together. Extinction training was 30 CS presentations and extinction testing was 10 CS presentations, both in the absence of footshocks. We also subjected another set of SPS and control rats to similar procedures, except during fear conditioning footshock presentation was omitted (CS-only controls). All stimuli presentations were separated by an interval of 60s and behavioral sessions (e.g. fear conditioning, extinction training) were separated by a 1-day interval. We then measured c-Fos levels in the RSC (granular and agranular regions) in subsets of rats that were euthanized after fear conditioning (CS-fear: SPS = 5, Control = 8; CS-only: SPS = 5, Control = 8), extinction training (CS-fear: SPS = 13, Control = 12; CS-only: SPS = 12, Control = 12), extinction testing (CS-fear: SPS = 8, Control = 10; CS-only: SPS = 12, Control = 10) and immediate removal from the housing colony to establish baseline levels of c-Fos (SPS = 17, Control = 8). These data are novel, but are from a larger dataset whose protocols, regulatory approval, and behavioral results have been previously published 130,135. SPS may have decreased c-Fos levels in the RSC (granular and agranular regions) at baseline as this effect approached statistical significance [SPS vs. control: t(23) = 1.951, p = .063; Figure 1]. C-fos levels during all behavioral sessions were subjected to separate stress (SPS vs. control) x session (fear conditioning, extinction training, extinction testing) factor designs for the CS-fear and CS-only treatments. For the CS-fear treatment, SPS may have decreased c-Fos levels during fear conditioning, as a stress x session interaction approached significance [F(2,56) = 2.949, p = .063]. For the CS-only treatment there was a main effect of session [F(2,59) = 8.219, p < .001], which reflected increased c-Fos RSC levels during fear conditioning in comparison to extinction training and testing. Importantly, there were no stress effects (ps > .05).
Figure 1.
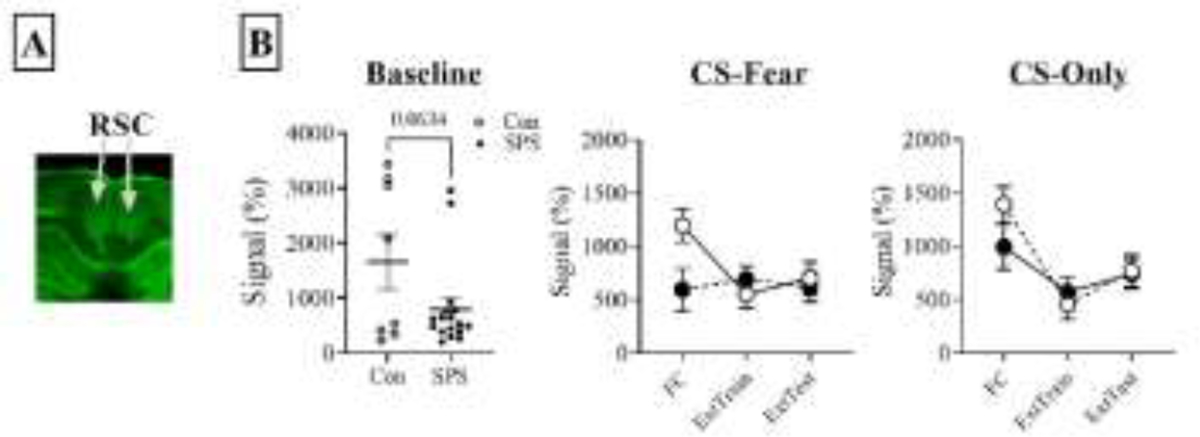
Effects of SPS on c-Fos levels in the RSC (granular and agranular regions) at baseline, fear conditioning, extinction training, and extinction testing. A) representative near-infrared immunohistochemical image staining for c-Fos in the RSC. B) Effects of SPS on c-Fos levels during baseline and behavioral tasks. Baseline c-Fos levels in the RSC was lower in SPS rats when compared to controls and this effect approached significance. For the CS-Fear treatment c-Fos levels were lower during fear conditioning alone and this potential interaction (stress x treatment) approached significance. For the CS-Only treatment there were no stress effects.
Together, these results raise the possibility that SPS decreases activation of the RSC during fear memory formation and this could contribute to extinction retention deficits (i.e. inability to maintain extinction) within the SPS model, though further research is needed to identify mechanisms via which SPS changes in RSC function lead to SPS extinction retention deficits.
3. Role of chBF neuronal input to spatial/contextual memory systems in emotional memory regulation
3.1. chBF neurons
ChBF projection neurons are located in clusters in the base of the forebrain and can be found from the olfactory tubercle to the rostral end of the lateral geniculate bodies. There are excellent reviews and empirical studies about chBF neuron clusters and properties 7–10,145–149. This section provides a brief summary of chBF neurons and their properties, and is not meant to be a comprehensive description of chBF neurons or cholinergic receptor pharmacology.
The activation of chBF and consequent membrane depolarization and Ca2+ influx of presynaptic terminals results into the release of acetylcholine (ACh) into the target terminal fields 150,151. Termination of ACh actions on receptors is accomplished by metabolism of ACh by acetylcholinesterase (AChE) 152 and axonal choline reuptake 153–155. This choline is then used to synthesize ACh at the axon terminal 153,154; a reaction catalyzed by choline acetyltransferase (ChAT) 156. ACh is then packaged into synaptic vesicles via the vesicular acetylcholine transporter (VAChT) for synaptic release 157.
Cholinergic neurons signal through metabotropic muscarinic receptors (mAChRs) and ionotropic nicotinic receptors (nAChRs) 155. There are five broad types of mAChRs with m1AChR and m3–5AChRs being post synaptic and m2AChR being presynaptic 155,158,159. Activation of mAChRs lead to stimulation of phosphoinositol synthesis and or inhibition of cAMP synthesis 160 and changes in the permeability of the neuronal membrane to K+, Ca2+, and Cl− channels 161. NAChRs are pentameric ion channels comprised of α and β subunits in different configurations 155. There are at least 11 variants of the α subunit and eight variants of the β subunit 162. The two most common types of nAChRs in the brain comprise of α4β2 subunits and α7 subunits exclusively 162,163. Furthermore, even though nAChRs form a non-specific cation pore, receptors with different subunits can have selectivity for different types of cation species 164. ChBF neuronal clusters in the anterior region of the BF project to midline cortical regions, Hipp, and medial habenula, while more posterior BF regions project to the dorsolateral neocortex, PFC, and amygdala. Input to chBF neurons originate from multiple systems in the brain with chBFs receiving neocortical, amygdala, and subcortical input 10,145,165–167.
3.2. Role of chNBM neurons in emotional memory regulation
Given the role of the PFC in facilitating traumatic stress effects on emotional memory regulation, chBF input to the PFC could form circuits through which traumatic stress alters these emotional processes. This is especially so, because clusters of cholinergic neurons that project to the PFC have been implicated in emotional memory (see below). ChBF input to the PFC originates from two heterogeneous clusters of cholinergic neurons. Nucleus basalis of Meynert (chNBM), which also has cholinergic neurons that project to the amygdala, and medial septum and diagonal band of Broca (chMS/DBB), which also has cholinergic neurons that project to the Hipp and medial habenula 8–10,168. Other chBF corticopetal neurons that project to the mPFC can be found in the medial and lateral preoptic areas 8,9.
Many studies investigating the role of chNBM corticopetal neurons in mediating emotional memory have utilized the selective cholinergic immunotoxin 192 IgG saporin to induce permanent chNBM corticopetal lesions. ChBF neurons express the p75 receptor 169 and are they are the only known neuronal group in the BF that express this receptor 169,170. The 192 IgG saporin toxin does not affect chNBM amygdalopetal neurons and has minimal effects on non-cholinergic neurons 11,169,170. Thus, when this toxin is infused into the NBM (or the axonal fields of chNBM neurons), selective cholinergic corticopetal neuronal loss can be induced. However, with this treatment residual cholinergic input to the mPFC remains 171–173 and most studies that have used this toxin did not specifically implicate chNBM neuronal input to the PFC as being critical for emotional memory regulation.
chNBM corticopetal lesions attenuate cued and contextual fear conditioned suppression (i.e. cessation of bar pressing for food induced by presentation of a feared CS) 171,172,174, without impacting operant performance per se, and animals with chNBM cholinergic lesions can still display operant suppression to footshocks 171,172,175. Surprisingly, complete chBF or chNBM corticopetal lesions have no effect on cued or contextual fear conditioned freezing 11,168,170,174,176 and non-specific excitatory lesions in the NBM have no effects on fear potentiated startle 177. Two previous studies have reported that contextual fear learning within the inhibitory avoidance paradigm is sensitive to chNBM corticopetal lesions. Within the inhibitory avoidance paradigm, an animal is placed in a light chamber of a passive avoidance box that is connected to a dark chamber by a doorway. Upon entering the dark chamber, the door shuts and animals receive a footshock. One day later, animals are returned to the light chamber of the passive avoidance box and can enter the dark chamber. With conditioning, passive/inhibitory avoidance is observed. In the inhibitory avoidance paradigm, chNBM corticopetal lesions enhance contextual avoidance 173,178. It should be noted that another study has reported that chNBM corticopetal lesions have no effects on inhibitory avoidance, but instead is critical for the memory enhancing effects of norepinephrine release within the basolateral amygdala 179.
Results obtained with permanent chBF neural lesions (selective or nonspecific) can be different to results obtained with temporary manipulation of chBF neurons 180. One study has observed that enhancing cholinergic input to the mPFC enhances contextual fear memory 181 and another study has reported that chNBM input to the auditory cortex is critical for auditory Pavlovian fear conditioning 182. These studies used either pharmacological or optogenetic methods to manipulate chNBM neurons. With regard to extinction memory, one study has observed that chNBM corticopetal lesions have no impact on extinction memory 168, but another study has observed that muscarinic antagonism within the IL disrupts extinction memory 183. However, it is not clear if this finding specifically implicates chNBM input to the IL, because chBF input to the IL may originate from chMS/DBB neurons 168.
Taken together studies examining the role of chNBM corticopetal cholinergic neurons on emotional memory are mixed and difficult to interpret in a straightforward manner. Results to date have observed that chNBM corticopetal lesions can disrupt contextual fear memory in one behavioral paradigm (i.e. conditioned suppression), have no effect in other behavioral paradigms (i.e. conditioned freezing and startle), and inhibit avoidance in yet another paradigm (i.e. inhibitory avoidance). Furthermore, inferences made about the role of chNBM corticopetal neurons in emotional memory can vary depending on the methods used to manipulate these neurons (e.g. optogenetic, pharmacological, permanent lesions). ChNBM neurons project to the PFC, which has been implicated in mediating traumatic stress effects on emotional memory and regulation 17,33,114,119,124. ChNBM corticopetal neurons are also intermingled with chNBM amygdalopetal neurons, which are critical for different aspects of fear memory 184,185. Neurons in the central nucleus of the amygdala project to the NBM 186–189 and central amygdala neurons are critical for conditioned fear memory 190,191. Given these findings, one would expect chNBM corticopetal neurons to be critical for fear memory, but unresolved issues remain. These issues may be resolved with further research that utilizes techniques that can specifically target selective groups of chNBM neurons. The existence of rodents that express cre recombinase in cholinergic neurons should facilitate this endeavor, and a number of studies have used this technology already to explore the role of chNBM neurons in emotional memory 184,185,192. Lastly, selective manipulation of chMS/DBB and preoptic input to the PFC on emotional memory regulation are largely unexplored, but a direction for future research effort.
3.3. Role of chMS/DBB hippocampal projecting neurons in emotional memory
The MS/DBB contains cholinergic neurons that project to the Hipp, but there are neurons in the MS/DBB that also project to the mPFC, midline cortical regions and the medial habenula as well 147,168. Many studies have used pharmacological approaches to specifically target chMS/DBB neurons that project to the dHipp and other studies have selectively targeted the axons of chMS/DBB neurons in their terminal fields, while others have manipulated chMS/DBB neurons without specifically manipulating a select population of chMS/DBB neurons.
3.3.1. Fear memory
ChMS/DBB input to the Hipp is virtually the exclusive source of ACh in the Hipp 7–9,147. As a result, pharmacological manipulation of cholinergic receptors in the Hipp directly manipulates chMS/DBB neuronal input to the Hipp. Research measuring ACh levels in the dHipp (e.g. using microdialysis), pharmacologically manipulating cholinergic receptors in the dHipp, or optogenetic activation of chMS/DBB neurons suggest that chMS/DBB hippocampal neurons are critical for mediating contextual fear conditioned freezing. Hipp ACh levels are enhanced during Pavlovian contextual fear conditioning 193,194 and blockade of mAChRs in the dHipp disrupts acquisition 195,196 and consolidation 196,197 of Pavlovian contextual fear conditioning. Enhancing ACh levels 198 and nAChR activation in the dHipp and vHipp enhances contextual fear memory retrieval 195,199,200, although nAChR antagonism in the dHipp has no effect on Pavlovian contextual fear conditioning 201. Finally, optogenetic stimulation of chMS/DBB neurons enhances contextual fear conditioning 202. Similar results are obtained when contextual fear memory is measured in the inhibitory avoidance paradigm 193,203–211. Together, these findings suggest that chMS/DBB input to the Hipp is critical for contextual fear memory.
The results of studies employing selective chMS/DBB lesions are not consistent with results from pharmacological or optogenetic studies. ChMS/DBB lesions have no effects on contextual Pavlovian fear conditioning 170,176,178,212 or inhibitory avoidance 173,178. However, chMS/DBB lesions disrupt contextual fear memory discrimination 168. If animals are fear conditioned in one context, then tested for fear memory in a novel context, contextual fear expression is observed in this novel context in rats with chMS/DBB lesions, even though this context was never paired with a UCS 168. The pool of chMS/DBB neurons that are critical for this phenomena are unclear as neither loss of chMS/DBB input to the mPFC, dHipp, and mPFC and dHipp replicates this effect 213. ChMS/DBB neurons also project to the RSC 8,9, which is critical for contextual learning and memory 138,140. While chMS/DBB input to the RSC could be critical for contextual fear memory discrimination, this hypothesis remains untested. Disrupting chMS/DBB input to the vHipp disrupts auditory fear conditioning 214, which suggests chMS/DBB input to the vHipp is critical for fear memory.
The reasons for the discrepancies observed when using temporary manipulation in the Hipp vs. chMS/DBB lesions are unclear, but pharmacological, optogenetic, and selective permanent lesion point to a role of chMS/DBB neurons being critical for aspects of contextual fear memory (e.g. acquisition, consolidation, discrimination). Furthermore, chMS/DBB input to the vHipp is critical for cued fear memory
3.3.2. Extinction memory
ChMS/DBB neuronal lesions disrupt acquisition of contextual extinction memory in the fear conditioned freezing paradigm 176 and induce deficits in acquisition and maintenance of cued extinction memory in the fear conditioned freezing paradigm 168. The results from these two studies suggests that chMS/DBB neurons have significant roles to play in facilitating acquisition and maintenance of extinction memory. Through what circuits might chMS/DBB neurons facilitate extinction memory? ChMS/DBB neurons project to the IL, which has a significant role in extinction memory 43,122,215,216. Muscarinic antagonism in the IL disrupts extinction memory 183, which raises the possibility that chMS/DBB input to the IL is critical for extinction memory. This assertion is supported by the observation that chMS/DBB neurons may be the predominant source of ACh input to the IL 168. However, removal of cholinergic input to the mPFC has no effect on extinction memory 214, which raises questions about the role of chMS/DBB input to the IL in associative extinction memory (i.e. CS-no UCS memory).
Contextual processing has a significant role to play in acquisition and maintenance of fear extinction, where contexts can help determine the meaning of a CS that is initially paired with a UCS (i.e. fear conditioning) and then paired with safety (i.e. fear extinction) 1–3. In many fear conditioning paradigms, animals are first conditioned in a one context (e.g. fear context), then subjected to extinction training in a distinct context (e.g. extinction context). A number of studies have shown that animals use contextual information to determine when the CS signals fear vs. extinction (i.e. contextual occasion-setting) 109,110,217 and that the Hipp has a significant role to play in contextual occasion-setting 109,110,217. ChMS/DBB input to the Hipp could facilitate extinction memory by facilitating contextual occasion-setting, but selective removal of chMS/DBB input to the dHipp or vHipp has no effect on extinction memory 214. The RSC is a contextual/spatial system that receives chMS/DBB input, but the role of the RSC in contextual occasion-setting has not been sufficiently explored 137. To determine how chMS/DBB neurons facilitate extinction memory, future studies examining the role of chMS/DBB input to the RSC in fear extinction (e.g. acquisition, maintenance, contextual occasion-setting) is needed.
4. Involvement of cholinergic systems in neuroinflammation and PTSD
4.1. Neuroinflammation and PTSD
Inflammation is a normal physiological response against disrupted homeostasis due to infection, injury, or trauma. This response consists of a complex series of immune reactions and is initiated to neutralize invading pathogens, repair injured tissues, and promote wound healing. The onset of inflammation is characterized by activation of immune cells that release pro-/anti-inflammatory mediators, and other key signaling molecules involved in the inflammatory process including tumor necrosis factor (TNF), interleukin (IL)-1, IL-4, IL-6, IL-10, interferon γ (IFN-γ), transforming growth factor β (TGF-β), adhesion molecules, vasoactive mediators, and reactive oxygen species 218–220. Neuroinflammation refers to inflammatory changes that occur within the central nervous system (CNS) that primarily involves activation of resident brain immune cells such as microglia and astrocytes 221–223. However, peripheral pro-inflammatory cytokines released through macrophages or other peripheral immune cells can also trigger an inflammatory response in brain tissues by crossing the blood-brain barrier 224,225.
Prior research suggests a possible link between neuroinflammation and PTSD 226–228. Clinical studies have shown that PTSD is associated with a dysregulated immune response resulting in neuroinflammation and oxidative stress. In a prospective study involving war zone-deployed marines, pre-existing C-Reactive Protein (CRP, a biomarker of acute inflammatory response) concentrations in the plasma were directly correlated with the occurrence and severity of PTSD three months after deployment 229. Another study that recruited patients with orthopedic injuries found elevated serum levels of IL-6, IL-8, and TGF-β as compared to health control subjects 230. Post-traumatic stress symptoms (PTSS) in injured subjects assessed one month later were predicted by higher serum levels of IL-6 and IL-8 and lower levels of TGF-β. Likewise, multiple clinical studies have reported higher blood levels of pro-inflammatory biomarkers and reduced anti-inflammatory cytokines in patients with PTSD as compared to healthy controls 226,231,235.
Recent neuroimaging studies in PTSD patients demonstrated structural and functional alterations in brain circuits implicated in stress and emotion regulation. A positron emission tomography (PET) study utilizing [11C]PBR28, a PET ligand that binds to 18-kDa translocator protein (TSPO, a microglia biomarker) reported a negative association between PTSD symptoms and reduced TSPO availability in the corticolimbic circuits in patients with PTSD 236. Moreover, reduced TSPO availability and PTSD severity also correlated with higher CRP levels in the plasma of these patients suggesting that peripheral immune activation in PTSD is associated with microglia dysfunction. Another investigation employed 18F-fluorodeoxyglucose (FDG)-PET to determine the relationship between peripheral inflammatory markers and amygdala activity 237. Although the magnitude of the FDG signal in the amygdala was not associated with blood levels of pro-inflammatory cytokines, it did correlate with signal in the spleen and bone marrow. Together, these studies support a role for inflammation in driving PTSD.
An increasing body of evidence points towards a modulatory relationship between the buildup of reactive oxidative species (i.e. ROS, oxidative stress) and neuroinflammation in PTSD. Markers of oxidative stress are shown to be upregulated in PTSD patients 226,228,238–241. Additionally, genetic investigations identified single nucleotide polymorphisms of several oxidative stress-linked genes that either moderate the association between PTSD and cortical thickness (ALOX 12 and ALOX 15) or regulate neuroinflammatory and psychpathological manifestations of PTSD (BDNFVal66Met and RORA) 242,243. Stress-induced neuroinflammatory changes are known to trigger microglia activation which may further facilitate oxidative stress in the brain 244–246. Thus, neuroinflammation and oxidative stress may act in concert to contribute to PTSD symptomatology 247,248.
Consistent with clinical studies, preclinical investigations utilizing animal models of traumatic stress show that inflammatory responses may have a causative role to play in mediating traumatic stress effects in neural circuits. Rats exposed to a predator (an animal model of traumatic stress 71–73) exhibited higher expression of pro-inflammatory cytokines and elevated ROS in the Hipp and the PFC 249. Predator odor stress in mice also increased serum IL-1β levels and anxiety-like behavior via NF-κB signaling, while blockade of this pathway normalized these changes 250. Mice subjected to chronic social defeat (an animal model of traumatic stress 27,36,251), have transcripts associated with innate immunity and inflammatory signaling upregulated in both the blood and brain and defeated mice exhibited impaired hippocampal neurogenesis 252. Exposure to chronic social defeat stress leads to increased reactive species and activation of microglia, and both processes are implicated in behavioral effects induced by chronic social defeat stress 253,254. Intracerebroventricular infusions of an IL-1β antagonist prevented stress-enhanced fear learning (an animal model of traumatic stress 71–73) in rats 255. Using the SPS model of traumatic stress a previous study observed that SPS leads to activation of microglia in the vHipp and this may contribute to extinction retention deficits within the SPS model 256.
It is worthwhile noting two additional mechanisms via which neuroinflammation and the build-up of ROS may alter circuits after traumatic stress exposure. As discussed above, a dysregulated HPA axis and reduced GR sensitivity has been observed with traumatic stress exposure. A reduction in GR activity (or enhanced GR resistance) is suggested to increase the expression of proinflammatory proteins which may cross the blood brain barrier and contribute to neuroinflammation 257. The buildup of reactive species can lead to the induction of apoptosis and neuronal loss 258–260. SPS is associated with increased activity within apoptotic pathways 94,261,262 and misfolding of proteins within the endoplasmic reticulum 262, which leads to apoptosis 251,262. Furthermore, increases in apoptosis has been implicated in extinction retention deficits within the SPS model 94.
Although the existing evidence support a close relationship between neuroinflammation, reactive species buildup, and PTSD, more research is needed to identify specific circuits and mechanisms via which traumatic stress-induced changes in neuroinflammation and the buildup of reactive species lead to emotional memory dysregulation.
4.2. Cholinergic regulation of immune response and neuroinflammation: implications for PTSD
As discussed above, chBF systems are critical for emotional memory regulation. Central and peripheral cholinergic systems are also implicated in the modulation of inflammation and neuroimune communication. Expression of nAChRs and mAChRs on immune cells, including T cells, B cells, macrophages, microglia, and astrocytes, and cholinergic stimulation of these receptors has been shown to evoke various downstream functional effects on the immune system 263–267. In particular, the role of α7 nAChRs in orchestrating the cholinergic anti-inflammatory pathway is well studied. For example, vagus nerve stimulation that increases parasympathetic cholinergic tone attenuated the release of proinflammatory cytokines in a model of septic shock and this effect was mediated via activation of α7 nAChRs on macrophages 263,268. Likewise, the release of TNFα and IL-1β in response to toll-like receptor-mediated activation of human white blood cells was inhibited by selective stimulation of α7 nAChRs 269. Chronic administration of GAT-107, an allosteric agonist and positive allosteric modulator of α7 nAChR, suppressed T-cell proliferation and the production of pro-inflammatory cytokines while increased the secretion of IL-10 (an anti-inflammatory cytokine) in a mouse model of encephalopathy 270. Furthermore, transgenic α7 nAChR knockout mice displayed higher production and secretion of pro-inflammatory cytokines from antigen-stimulated spleen cells 271, and enhanced proliferative response in anti-CD40 antibody-stimulated B-Cells 267. Although α7 nAChRs are traditionally characterized as ionotropic receptors, their anti-inflammatory responses in non-neuronal cells occur in metabotropic ways with downstream signaling involving JAK/STAT and PI3K/Akt pathways that exert negative modulatory effects via nuclear factor NFκB on the expression of pro-inflammatory cytokines 272,273.
Similar to peripheral immune cells, α7 nAChRs regulate neuroinflammatory changes in the central nervous system. For instance, the activation of α7 nAChRs in cultured rat microglia cells, either via exposure to ACh or nicotine, inhibited the LPS-induced release of the proinflammatory cytokine TNF-α and this effect was driven by the suppression of JNK and p38 MAP Kinase pathway 274,275. Moreover, in organotypic hippocampal cultures subjected to oxygen and glucose deprivation, exposure to PNU282987 (an α7 nAChR agonist) reduced TNF-α, ROS production, and cell death, and this effect was abolished in microglia depleted cultures 276. The same study also reported that the treatment with PNU282987 reduced infarct size and improved motor skills in a mouse model of stroke demonstrating neuroprotective effects of cholinergic stimulation. Likewise, stimulation of astrocytic α7 nAChRs has been shown to suppress proinflammatory cytokines following immunogenic stimuli such as LPS and IL-1β 277,278. Furthermore, several studies demonstrated that Aβ-induced and MPTP-induced neuroinflammation and cognitive impairments is ameliorated by stimulation of glial α7 nAChRs implicating central cholinergic system as a key component involved in the regulation of neuroinflammatory changes in age-related neurodegenerative pathologies 279–284. In addition to the α7 nAChRs, muscarinic receptors and other subtypes of nicotinic receptors have also been suggested to suppress neuroinflammation and oxidative stress by regulating the activity of glial cells 285,287.
ChBF neurons are sensitive to increased inflammation and oxidative stress. Chronic LPS infusion into the BF of young rats reduces ChAT activity and increases the number of activated microglia and this effect was prevented by systemic administration of anti-inflammatory drugs 288. Given that activated microglia can generate ROS in the local environment 289, the detrimental effects of neuroinflammation on cholinergic neurons could be driven by oxidative stress. Indeed, a recent study that utilized an olfactory bulbectomy model of cholinergic degeneration, found the loss of chMS/DBB neurons and reduced hippocampal ChAT activity was preceded by accumulation of protein-bound carbonyls suggesting an involvement of oxidative stress in cholinergic disruption 290. Additionally, oxidative stress and elevated cytosolic ROS has been shown to disrupt muscarinic and nicotinic cholinergic signaling and downstream pathways 291,292.
Nerve growth factor is a primary neutrotrophic factor that supports the growth, survival, and maturation of chBF neurons by binding to the high-affinity tropomysin-related kinase A (trkA) receptor 293. Extensive evidence from in vitro and in vivo studies indicate that oxidative stress or disruption of anti-oxidant defense systems reduces trkA expression resulting in cholinergic dysfunction in Alzheimer’s Disease 292,294,295. Additionally, our previous research suggested that disruption of NGF-trkA signaling increases the vulnerability of chBF neurons and cognitive capacities in aging model systems 296,297.
Collectively, the current evidence supports a scenario where neuroinflammation and buildup of reactive species associated with traumatic stress can disrupt chBF circuits leading to emotional memory dysregulation. Further research is warranted to understand the complex relationship between chBF systems, neuroinflammation, reactive species buildup, and traumatic stress.
5. Conclusion
A relatively large body of research has shown that chBF neurons may represent aspects of contextual/spatial systems, these neurons are critical for emotional regulation (e.g. maintenance of fear extinction), and may be sensitive to traumatic stress. Indeed, traumatic stress may disrupt emotional memory regulation by having direct effects on chBF neurons (e.g. neuroinflammation, reactive species buildup, apoptosis). Understanding the cellular and molecular underpinnings of how traumatic stress may compromise chBF neurons may provide insights into the underlying mechanism of PTSD and potentially inform the development of therapeutic approaches. Equally important is the identification of circuits through which chBF neurons facilitate emotional memory regulation. Thus far, a critically unexplored circuit involves chMS/DBB input to the RSC, because the RSC is a contextual/spatial system that may be sensitive to traumatic stress. Future studies examining the potential importance of chMS/DBB input to the RSC in regulating fear and extinction memory and the sensitivity of this circuit to traumatic stress is needed.
Highlights.
chBF systems project to contextual/spatial systems
Contextual/spatial systems are sensitive to traumatic stress
chBF systems are critical for emotional memory regulation
chBF systems may be sensitive to traumatic stress
Traumatic stress may disrupt emotional memory regulation via chBF systems
Acknowledgement
I would like to thank Michele Barth and Jiana Stammas for their help with generating data presented in Figure 1. Scholarship activity in this grant was supported by grants from the National Institute on Aging (AG029592 and AG046580) and from the American Federation for Aging Research awarded to VP and NIGMS grant 1P20GM103653.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Bouton ME, Westbrook RF, Corcoran KA & Maren S Contextual and temporal modulation of extinction: behavioral and biological mechanisms. Biol Psychiatry 60, 352–360, doi:S0006–3223(06)00099–0 [pii] 10.1016/j.biopsych.2005.12.015 (2006). [DOI] [PubMed] [Google Scholar]
- 2.Maren S, Phan KL & Liberzon I The contextual brain: implications for fear conditioning, extinction and psychopathology. Nat Rev Neurosci 14, 417–428, doi: 10.1038/nrn3492 nrn3492 [pii] (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Trask S, Thrailkill EA & Bouton ME Occasion setting, inhibition, and the contextual control of extinction in Pavlovian and instrumental (operant) learning. Behav Processes 137, 64–72, doi: 10.1016/j.beproc.2016.10.003 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lisman J et al. Viewpoints: how the hippocampus contributes to memory, navigation and cognition. Nat Neurosci 20, 1434–1447, doi: 10.1038/nn.4661 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Byrne P, Becker S & Burgess N Remembering the past and imagining the future: a neural model of spatial memory and imagery. Psychol Rev 114, 340–375, doi: 10.1037/0033-295X.114.2.340 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mittelstaedt H & Mittelstaedt M-L in Avian navigation 290–297 (Springer, 1982). [Google Scholar]
- 7.Mesulam MM, Mufson EJ, Levey AI & Wainer BH Cholinergic innervation of cortex by the basal forebrain: cytochemistry and cortical connections of the septal area, diagonal band nuclei, nucleus basalis (substantia innominata), and hypothalamus in the rhesus monkey. J Comp Neurol 214, 170–197 (1983). [DOI] [PubMed] [Google Scholar]
- 8.Mesulam MM, Mufson EJ, Wainer BH & Levey AI Central cholinergic pathways in the rat: an overview based on an alternative nomenclature (Ch1-Ch6). Neuroscience 10, 1185–1201 (1983). [DOI] [PubMed] [Google Scholar]
- 9.Woolf NJ, Eckenstein F & Butcher LL Cholinergic systems in the rat brain: I. projections to the limbic telencephalon. Brain Research Bulletin 13, 751–784 (1984). [DOI] [PubMed] [Google Scholar]
- 10.Zaborszky L et al. Specific Basal Forebrain-Cortical Cholinergic Circuits Coordinate Cognitive Operations. J Neurosci 38, 9446–9458, doi: 10.1523/JNEUROSCI.1676-18.2018 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Conner JM, Culberson A, Packowski C, Chiba AA & Tuszynski MH Lesions of the Basal forebrain cholinergic system impair task acquisition and abolish cortical plasticity associated with motor skill learning. Neuron 38, 819–829 (2003). [DOI] [PubMed] [Google Scholar]
- 12.Ashe JH, McKenna TM & Weinberger NM Cholinergic modulation of frequency receptive fields in auditory cortex: II. Frequency-specific effects of anticholinesterases provide evidence for a modulatory action of endogenous ACh. Synapse 4, 44–54 (1989). [DOI] [PubMed] [Google Scholar]
- 13.Ramanathan D, Tuszynski MH & Conner JM The basal forebrain cholinergic system is required specifically for behaviorally mediated cortical map plasticity. J Neurosci 29, 5992–6000, doi: 10.1523/JNEUROSCI.0230-09.2009 29/18/5992 [pii] (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ikonen S, McMahan R, Gallagher M, Eichenbaum H & Tanila H Cholinergic system regulation of spatial representation by the hippocampus. Hippocampus 12, 386–397, doi: 10.1002/hipo.1109 (2002). [DOI] [PubMed] [Google Scholar]
- 15.McGaughy J, Koene RA, Eichenbaum H & Hasselmo ME Cholinergic deafferentation of the entorhinal cortex in rats impairs encoding of novel but not familiar stimuli in a delayed nonmatch-to-sample task. J Neurosci 25, 10273–10281, doi:25/44/10273 [pii] 10.1523/JNEUROSCI.2386-05.2005 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Winters BD & Bussey TJ Removal of cholinergic input to perirhinal cortex disrupts object recognition but not spatial working memory in the rat. Eur J Neurosci 21, 2263–2270, doi:EJN4055 [pii] 10.1111/j.1460-9568.2005.04055.x (2005). [DOI] [PubMed] [Google Scholar]
- 17.Alexandra Kredlow M, Fenster RJ, Laurent ES, Ressler KJ & Phelps EA Prefrontal cortex, amygdala, and threat processing: implications for PTSD. Neuropsychopharmacology 47, 247–259, doi: 10.1038/s41386-021-01155-7 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Heroux NA, Robinson-Drummer PA, Sanders HR, Rosen JB & Stanton ME Differential involvement of the medial prefrontal cortex across variants of contextual fear conditioning. Learning & memory (Cold Spring Harbor, N.Y 24, 322–330, doi: 10.1101/lm.045286.117 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fanselow MS Contextual fear, gestalt memories, and the hippocampus. Behav Brain Res 110, 73–81, doi:S0166432899001862 [pii] (2000). [DOI] [PubMed] [Google Scholar]
- 20.Zelikowsky M et al. Prefrontal microcircuit underlies contextual learning after hippocampal loss. Proc Natl Acad Sci U S A 110, 9938–9943, doi: 10.1073/pnas.1301691110 1301691110 [pii] (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bremner JD Traumatic stress: effects on the brain. Dialogues Clin Neurosci 8, 445–461 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bremner JD et al. MRI-based measurement of hippocampal volume in patients with combat-related posttraumatic stress disorder. Am J Psychiatry 152, 973–981, doi: 10.1176/ajp.152.7.973 (1995). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chao LL, Yaffe K, Samuelson K & Neylan TC Hippocampal volume is inversely related to PTSD duration. Psychiatry Res 222, 119–123, doi: 10.1016/j.pscychresns.2014.03.005 (2014). [DOI] [PubMed] [Google Scholar]
- 24.Woon FL, Sood S & Hedges DW Hippocampal volume deficits associated with exposure to psychological trauma and posttraumatic stress disorder in adults: a meta-analysis. Prog Neuropsychopharmacol Biol Psychiatry 34, 1181–1188, doi: 10.1016/j.pnpbp.2010.06.016 (2010). [DOI] [PubMed] [Google Scholar]
- 25.Bremner JD Alterations in brain structure and function associated with post-traumatic stress disorder. Semin Clin Neuropsychiatry 4, 249–255, doi: 10.153/SCNP00400249 (1999). [DOI] [PubMed] [Google Scholar]
- 26.Szeszko PR, Lehrner A & Yehuda R Glucocorticoids and Hippocampal Structure and Function in PTSD. Harv Rev Psychiatry 26, 142–157, doi: 10.1097/HRP.0000000000000188 (2018). [DOI] [PubMed] [Google Scholar]
- 27.Gilbertson MW et al. Smaller hippocampal volume predicts pathologic vulnerability to psychological trauma. Nat Neurosci 5, 1242–1247, doi: 10.1038/nn958 nn958 [pii] (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rocha-Rego V et al. Decreased premotor cortex volume in victims of urban violence with posttraumatic stress disorder. PLoS One 7, e42560, doi: 10.1371/journal.pone.0042560 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Clausen AN et al. Combat exposure, posttraumatic stress disorder, and head injuries differentially relate to alterations in cortical thickness in military Veterans. Neuropsychopharmacology 45, 491–498, doi: 10.1038/s41386-019-0539-9 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang X et al. Cortical volume abnormalities in posttraumatic stress disorder: an ENIGMA-psychiatric genomics consortium PTSD workgroup mega-analysis. Mol Psychiatry 26, 4331–4343, doi: 10.1038/s41380-020-00967-1 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.APA. Diagnostic and Statistical Manual of Mental Disorders. 5th edn, (American Psychiatric Association, 2016). [Google Scholar]
- 32.Milad MR et al. Presence and acquired origin of reduced recall for fear extinction in PTSD: results of a twin study. J Psychiatr Res 42, 515–520, doi:S0022–3956(08)00026–5 [pii] 10.1016/j.jpsychires.2008.01.017 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Milad MR, Rauch SL, Pitman RK & Quirk GJ Fear extinction in rats: implications for human brain imaging and anxiety disorders. Biol Psychol 73, 61–71, doi:S0301–0511(06)00026–3 [pii] 10.1016/j.biopsycho.2006.01.008 (2006). [DOI] [PubMed] [Google Scholar]
- 34.Davis M & Whalen PJ The amygdala: vigilance and emotion. Mol Psychiatry 6, 13–34, doi: 10.1038/sj.mp.4000812 (2001). [DOI] [PubMed] [Google Scholar]
- 35.LeDoux JE Emotion circuits in the brain. Annual review of neuroscience 23, 155–184 (2000). [DOI] [PubMed] [Google Scholar]
- 36.Maren S Neurobiology of Pavlovian fear conditioning. Annu Rev Neurosci 24, 897–931, doi: 10.1146/annurev.neuro.24.1.897 24/1/897 [pii] (2001). [DOI] [PubMed] [Google Scholar]
- 37.Lang PJ, Davis M & Ohman A Fear and anxiety: animal models and human cognitive psychophysiology. J Affect Disord 61, 137–159 (2000). [DOI] [PubMed] [Google Scholar]
- 38.LeDoux JE Brain mechanisms of emotion and emotional learning. Curr Opin Neurobiol 2, 191–197, doi:0959-4388(92)90011-9 [pii] (1992). [DOI] [PubMed] [Google Scholar]
- 39.Rudy JW, Huff NC & Matus-Amat P Understanding contextual fear conditioning: insights from a two-process model. Neurosci Biobehav Rev 28, 675–685, doi:S0149–7634(04)00093–4 [pii] 10.1016/j.neubiorev.2004.09.004 (2004). [DOI] [PubMed] [Google Scholar]
- 40.Orsini CA & Maren S Neural and cellular mechanisms of fear and extinction memory formation. Neurosci Biobehav Rev 36, 1773–1802, doi:S0149–7634(11)00221–1 [pii] 10.1016/j.neubiorev.2011.12.014 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pavlov I Conditioned refelexes. (Oxford University Press, 1927). [Google Scholar]
- 42.Quirk GJ Memory for extinction of conditioned fear is long-lasting and persists following spontaneous recovery. Learning & memory (Cold Spring Harbor, N.Y 9, 402–407, doi: 10.1101/lm.49602 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Quirk GJ, Garcia R & Gonzalez-Lima F Prefrontal mechanisms in extinction of conditioned fear. Biol Psychiatry 60, 337–343 (2006). [DOI] [PubMed] [Google Scholar]
- 44.Rescorla RA Spontaneous recovery. Learning & memory (Cold Spring Harbor, N.Y 11, 501–509, doi: 10.1101/lm.77504 (2004). [DOI] [PubMed] [Google Scholar]
- 45.Rothbaum BO & Davis M Applying learning principles to the treatment of post-trauma reactions. Ann N Y Acad Sci 1008, 112–121 (2003). [DOI] [PubMed] [Google Scholar]
- 46.McGaugh JL & Roozendaal B Role of adrenal stress hormones in forming lasting memories in the brain. Curr Opin Neurobiol 12, 205–210 (2002). [DOI] [PubMed] [Google Scholar]
- 47.Roesler R, Roozendaal B & McGaugh JL Basolateral amygdala lesions block the memory-enhancing effect of 8-Br-cAMP infused into the entorhinal cortex of rats after training. Eur J Neurosci 15, 905–910 (2002). [DOI] [PubMed] [Google Scholar]
- 48.Roozendaal B, Brunson KL, Holloway BL, McGaugh JL & Baram TZ Involvement of stress-released corticotropin-releasing hormone in the basolateral amygdala in regulating memory consolidation. Proc Natl Acad Sci U S A 99, 13908–13913, doi: 10.1073/pnas.212504599 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Roozendaal B, Quirarte GL & McGaugh JL Glucocorticoids interact with the basolateral amygdala beta-adrenoceptor--cAMP/cAMP/PKA system in influencing memory consolidation. Eur J Neurosci 15, 553–560 (2002). [DOI] [PubMed] [Google Scholar]
- 50.Zschucke E, Renneberg B, Dimeo F, Wustenberg T & Strohle A The stress-buffering effect of acute exercise: Evidence for HPA axis negative feedback. Psychoneuroendocrinology 51, 414–425, doi: 10.1016/j.psyneuen.2014.10.019 (2015). [DOI] [PubMed] [Google Scholar]
- 51.Diorio D, Viau V & Meaney MJ The role of the medial prefrontal cortex (cingulate gyrus) in the regulation of hypothalamic-pituitary-adrenal responses to stress. J Neurosci 13, 3839–3847 (1993). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Laryea G, Muglia L, Arnett M & Muglia LJ Dissection of glucocorticoid receptor-mediated inhibition of the hypothalamic-pituitary-adrenal axis by gene targeting in mice. Front Neuroendocrinol 36, 150–164, doi: 10.1016/j.yfrne.2014.09.002 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Feldman S & Conforti N Modifications of adrenocortical responses following frontal cortex simulation in rats with hypothalamic deafferentations and medial forebrain bundle lesions. Neuroscience 15, 1045–1047, doi: 10.1016/0306-4522(85)90253-2 (1985). [DOI] [PubMed] [Google Scholar]
- 54.Jacobson L & Sapolsky R The role of the hippocampus in feedback regulation of the hypothalamic-pituitary-adrenocortical axis. Endocr Rev 12, 118–134, doi: 10.1210/edrv-12-2-118 (1991). [DOI] [PubMed] [Google Scholar]
- 55.van Haarst AD, Oitzl MS & de Kloet ER Facilitation of feedback inhibition through blockade of glucocorticoid receptors in the hippocampus. Neurochem Res 22, 1323–1328, doi: 10.1023/a:1022010904600 (1997). [DOI] [PubMed] [Google Scholar]
- 56.Yehuda R et al. Enhanced suppression of cortisol following dexamethasone administration in posttraumatic stress disorder. Am J Psychiatry 150, 83–86 (1993). [DOI] [PubMed] [Google Scholar]
- 57.Stein MB, Yehuda R, Koverola C & Hanna C Enhanced dexamethasone suppression of plasma cortisol in adult women traumatized by childhood sexual abuse. Biol Psychiatry 42, 680–686, doi: 10.1016/s0006-3223(96)00489-1 (1997). [DOI] [PubMed] [Google Scholar]
- 58.Yehuda R, Boisoneau D, Lowy MT & Giller EL Jr. Dose-response changes in plasma cortisol and lymphocyte glucocorticoid receptors following dexamethasone administration in combat veterans with and without posttraumatic stress disorder. Arch Gen Psychiatry 52, 583–593, doi: 10.1001/archpsyc.1995.03950190065010 (1995). [DOI] [PubMed] [Google Scholar]
- 59.de Kloet CS et al. Enhanced cortisol suppression in response to dexamethasone administration in traumatized veterans with and without posttraumatic stress disorder. Psychoneuroendocrinology 32, 215–226, doi: 10.1016/j.psyneuen.2006.12.009 (2007). [DOI] [PubMed] [Google Scholar]
- 60.Yehuda R, Blair W, Labinsky E & Bierer LM Effects of parental PTSD on the cortisol response to dexamethasone administration in their adult offspring. Am J Psychiatry 164, 163–166, doi: 10.1176/ajp.2007.164.1.163 (2007). [DOI] [PubMed] [Google Scholar]
- 61.Golier JA, Schmeidler J, Legge J & Yehuda R Enhanced cortisol suppression to dexamethasone associated with Gulf War deployment. Psychoneuroendocrinology 31, 1181–1189, doi: 10.1016/j.psyneuen.2006.08.005 (2006). [DOI] [PubMed] [Google Scholar]
- 62.Griffin MG, Resick PA & Yehuda R Enhanced cortisol suppression following dexamethasone administration in domestic violence survivors. Am J Psychiatry 162, 1192–1199, doi: 10.1176/appi.ajp.162.6.1192 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Schumacher S et al. HPA axis regulation in posttraumatic stress disorder: A meta-analysis focusing on potential moderators. Neurosci Biobehav Rev 100, 35–57, doi: 10.1016/j.neubiorev.2019.02.005 (2019). [DOI] [PubMed] [Google Scholar]
- 64.Klaassens ER, Giltay EJ, Cuijpers P, van Veen T & Zitman FG Adulthood trauma and HPA-axis functioning in healthy subjects and PTSD patients: a meta-analysis. Psychoneuroendocrinology 37, 317–331, doi: 10.1016/j.psyneuen.2011.07.003 (2012). [DOI] [PubMed] [Google Scholar]
- 65.Yehuda R Biology of posttraumatic stress disorder. The Journal of clinical psychiatry 62 Suppl 17, 41–46 (2001). [PubMed] [Google Scholar]
- 66.van Zuiden M et al. Pre-deployment differences in glucocorticoid sensitivity of leukocytes in soldiers developing symptoms of PTSD, depression or fatigue persist after return from military deployment. Psychoneuroendocrinology 51, 513–524, doi: 10.1016/j.psyneuen.2014.09.014 (2015). [DOI] [PubMed] [Google Scholar]
- 67.de Kloet CS et al. Leukocyte glucocorticoid receptor expression and immunoregulation in veterans with and without post-traumatic stress disorder. Mol Psychiatry 12, 443–453, doi: 10.1038/sj.mp.4001934 (2007). [DOI] [PubMed] [Google Scholar]
- 68.van Zuiden M et al. Glucocorticoid sensitivity of leukocytes predicts PTSD, depressive and fatigue symptoms after military deployment: A prospective study. Psychoneuroendocrinology 37, 1822–1836, doi: 10.1016/j.psyneuen.2012.03.018 (2012). [DOI] [PubMed] [Google Scholar]
- 69.Yehuda R, Lowy MT, Southwick SM, Shaffer D & Giller EL Jr. Lymphocyte glucocorticoid receptor number in posttraumatic stress disorder. Am J Psychiatry 148, 499–504, doi: 10.1176/ajp.148.4.499 (1991). [DOI] [PubMed] [Google Scholar]
- 70.van Zuiden M et al. Pre-existing high glucocorticoid receptor number predicting development of posttraumatic stress symptoms after military deployment. Am J Psychiatry 168, 89–96, doi: 10.1176/appi.ajp.2010.10050706 (2011). [DOI] [PubMed] [Google Scholar]
- 71.Armario A, Escorihuela RM & Nadal R Long-term neuroendocrine and behavioural effects of a single exposure to stress in adult animals. Neurosci Biobehav Rev 32, 1121–1135, doi:S0149–7634(08)00053–5 [pii] 10.1016/j.neubiorev.2008.04.003 (2008). [DOI] [PubMed] [Google Scholar]
- 72.Bowers ME & Ressler KJ An Overview of Translationally Informed Treatments for Posttraumatic Stress Disorder: Animal Models of Pavlovian Fear Conditioning to Human Clinical Trials. Biol Psychiatry 78, E15–27, doi: 10.1016/j.biopsych.2015.06.008 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Deslauriers J, Toth M, Der-Avakian A & Risbrough VB Current Status of Animal Models of Posttraumatic Stress Disorder: Behavioral and Biological Phenotypes, and Future Challenges in Improving Translation. Biol Psychiatry 83, 895–907, doi: 10.1016/j.biopsych.2017.11.019 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ferland-Beckham C et al. Systematic Review and Methodological Considerations for the Use of Single Prolonged Stress and Fear Extinction Retention in Rodents. Front Behav Neurosci 15, 652636, doi: 10.3389/fnbeh.2021.652636 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mancini GF et al. Sex-divergent long-term effects of single prolonged stress in adult rats. Behav Brain Res 401, 113096, doi: 10.1016/j.bbr.2020.113096 (2021). [DOI] [PubMed] [Google Scholar]
- 76.Liberzon I, Abelson JL, Flagel SB, Raz J & Young EA Neuroendocrine and psychophysiologic responses in PTSD: a symptom provocation study. Neuropsychopharmacology 21, 40–50, doi: 10.1016/S0893-133X(98)00128-6 (1999). [DOI] [PubMed] [Google Scholar]
- 77.Liberzon I, Krstov M & Young EA Stress-restress: effects on ACTH and fast feedback. Psychoneuroendocrinology 22, 443–453 (1997). [DOI] [PubMed] [Google Scholar]
- 78.Liberzon I, Lopez JF, Flagel SB, Vazquez DM & Young EA Differential regulation of hippocampal glucocorticoid receptors mRNA and fast feedback: relevance to post-traumatic stress disorder. J Neuroendocrinol 11, 11–17 (1999). [DOI] [PubMed] [Google Scholar]
- 79.Chen J et al. Paeoniflorin regulates the hypothalamic-pituitary-adrenal axis negative feedback in a rat model of post-traumatic stress disorder. Iran J Basic Med Sci 23, 439–448, doi: 10.22038/ijbms.2020.41214.9738 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pooley AE et al. Sex differences in the traumatic stress response: PTSD symptoms in women recapitulated in female rats. Biol Sex Differ 9, 31, doi: 10.1186/s13293-018-0191-9 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Perrine SA et al. Severe, multimodal stress exposure induces PTSD-like characteristics in a mouse model of single prolonged stress. Behav Brain Res 303, 228–237, doi: 10.1016/j.bbr.2016.01.056 (2016). [DOI] [PubMed] [Google Scholar]
- 82.Sasaki M et al. MRI identification of dorsal hippocampus homologue in human brain. Neuroreport 15, 2173–2176, doi: 10.1097/00001756-200410050-00005 (2004). [DOI] [PubMed] [Google Scholar]
- 83.Moser E, Moser MB & Andersen P Spatial learning impairment parallels the magnitude of dorsal hippocampal lesions, but is hardly present following ventral lesions. J Neurosci 13, 3916–3925 (1993). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Fanselow MS & Dong HW Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 65, 7–19, doi: 10.1016/j.neuron.2009.11.031 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Barbas H & Blatt GJ Topographically specific hippocampal projections target functionally distinct prefrontal areas in the rhesus monkey. Hippocampus 5, 511–533, doi: 10.1002/hipo.450050604 (1995). [DOI] [PubMed] [Google Scholar]
- 86.Eagle AL et al. Single prolonged stress enhances hippocampal glucocorticoid receptor and phosphorylated protein kinase B levels. Neurosci Res 75, 130–137, doi: 10.1016/j.neures.2012.11.001 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Knox D, Nault T, Henderson C & Liberzon I Glucocorticoid Receptors And Extinction Retention Deficits In The Single Prolonged Stress Model. Neuroscience 223, 163–173 (2012). [DOI] [PubMed] [Google Scholar]
- 88.Moulton E, Chamness M & Knox D Characterizing changes in glucocorticoid receptor internalization in the fear circuit in an animal model of post traumatic stress disorder. PLoS One 13, e0205144, doi: 10.1371/journal.pone.0205144 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ding J, Chen X, Han F & Meijer OC An Advanced Transcriptional Response to Corticosterone After Single Prolonged Stress in Male Rats. Front Behav Neurosci 15, 756903, doi: 10.3389/fnbeh.2021.756903 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.George SA, Rodriguez-Santiago M, Riley J, Rodriguez E & Liberzon I The effect of chronic phenytoin administration on single prolonged stress induced extinction retention deficits and glucocorticoid upregulation in the rat medial prefrontal cortex. Psychopharmacology (Berl) 232, 47–56, doi: 10.1007/s00213-014-3635-x (2015). [DOI] [PubMed] [Google Scholar]
- 91.Kohda K et al. Glucocorticoid receptor activation is involved in producing abnormal phenotypes of single-prolonged stress rats: a putative post-traumatic stress disorder model. Neuroscience 148, 22–33, doi: 10.1016/j.neuroscience.2007.05.041 (2007). [DOI] [PubMed] [Google Scholar]
- 92.Keller SM, Schreiber WB, Staib JM & Knox D Sex differences in the single prolonged stress model. Behav Brain Res 286, 29–32, doi: 10.1016/j.bbr.2015.02.034 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Wang HT, Han F & Shi YX Activity of the 5-HT1A receptor is involved in the alteration of glucocorticoid receptor in hippocampus and corticotropin-releasing factor in hypothalamus in SPS rats. Int J Mol Med 24, 227–231, doi: 10.3892/ijmm_00000225 (2009). [DOI] [PubMed] [Google Scholar]
- 94.Araki M et al. The role of glucocorticoid receptors in the induction and prevention of hippocampal abnormalities in an animal model of posttraumatic stress disorder. Psychopharmacology (Berl) 237, 2125–2137, doi: 10.1007/s00213-020-05523-x (2020). [DOI] [PubMed] [Google Scholar]
- 95.George SA, Stout SA, Tan M, Knox D & Liberzon I Early handling attenuates enhancement of glucocorticoid receptors in the prefrontal cortex in an animal model of post-traumatic stress disorder. Biol Mood Anxiety Disord 3, 22, doi: 10.1186/2045-5380-3-22 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lin CC, Cheng PY, Hsiao M & Liu YP Effects of RU486 in Treatment of Traumatic Stress-Induced Glucocorticoid Dysregulation and Fear-Related Abnormalities: Early versus Late Intervention. Int J Mol Sci 23, doi: 10.3390/ijms23105494 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Laukova M, Alaluf LG, Serova LI, Arango V & Sabban EL Early intervention with intranasal NPY prevents single prolonged stress-triggered impairments in hypothalamus and ventral hippocampus in male rats. Endocrinology 155, 3920–3933, doi: 10.1210/en.2014-1192 (2014). [DOI] [PubMed] [Google Scholar]
- 98.Bremner JD et al. Positron emission tomographic imaging of neural correlates of a fear acquisition and extinction paradigm in women with childhood sexual-abuse-related post-traumatic stress disorder. Psychol Med 35, 791–806, doi: 10.1017/s0033291704003290 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Grillon C & Morgan CA 3rd. Fear-potentiated startle conditioning to explicit and contextual cues in Gulf War veterans with posttraumatic stress disorder. J Abnorm Psychol 108, 134–142, doi: 10.1037//0021-843x.108.1.134 (1999). [DOI] [PubMed] [Google Scholar]
- 100.Norrholm SD et al. Fear extinction in traumatized civilians with posttraumatic stress disorder: relation to symptom severity. Biol Psychiatry 69, 556–563, doi: 10.1016/j.biopsych.2010.09.013 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Blechert J, Michael T, Vriends N, Margraf J & Wilhelm FH Fear conditioning in posttraumatic stress disorder: evidence for delayed extinction of autonomic, experiential, and behavioural responses. Behav Res Ther 45, 2019–2033, doi: 10.1016/j.brat.2007.02.012 (2007). [DOI] [PubMed] [Google Scholar]
- 102.Glover EM et al. Estrogen levels are associated with extinction deficits in women with posttraumatic stress disorder. Biol Psychiatry 72, 19–24, doi: 10.1016/j.biopsych.2012.02.031 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Garfinkel SN et al. Impaired contextual modulation of memories in PTSD: an fMRI and psychophysiological study of extinction retention and fear renewal. J Neurosci 34, 13435–13443, doi: 10.1523/JNEUROSCI.4287-13.2014 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Wicking M et al. Deficient fear extinction memory in posttraumatic stress disorder. Neurobiol Learn Mem 136, 116–126, doi: 10.1016/j.nlm.2016.09.016 (2016). [DOI] [PubMed] [Google Scholar]
- 105.Steiger F, Nees F, Wicking M, Lang S & Flor H Behavioral and central correlates of contextual fear learning and contextual modulation of cued fear in posttraumatic stress disorder. Int J Psychophysiol 98, 584–593, doi: 10.1016/j.ijpsycho.2015.06.009 (2015). [DOI] [PubMed] [Google Scholar]
- 106.Stubbendorff C, Hale E, Cassaday HJ, Bast T & Stevenson CW Dopamine D1-like receptors in the dorsomedial prefrontal cortex regulate contextual fear conditioning. Psychopharmacology (Berl) 236, 1771–1782, doi: 10.1007/s00213-018-5162-7 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Rozeske RR, Valerio S, Chaudun F & Herry C Prefrontal neuronal circuits of contextual fear conditioning. Genes Brain Behav 14, 22–36, doi: 10.1111/gbb.12181 (2015). [DOI] [PubMed] [Google Scholar]
- 108.Santos TB, Kramer-Soares JC, Favaro VM & Oliveira MGM Involvement of the prelimbic cortex in contextual fear conditioning with temporal and spatial discontinuity. Neurobiol Learn Mem 144, 1–10, doi: 10.1016/j.nlm.2017.05.003 (2017). [DOI] [PubMed] [Google Scholar]
- 109.Corcoran KA & Maren S Hippocampal inactivation disrupts contextual retrieval of fear memory after extinction. J Neurosci 21, 1720–1726, doi:21/5/1720 [pii] (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Corcoran KA & Maren S Factors regulating the effects of hippocampal inactivation on renewal of conditional fear after extinction. Learn Mem 11, 598–603, doi:11/5/598 [pii] 10.1101/lm.78704 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ji J & Maren S Electrolytic lesions of the dorsal hippocampus disrupt renewal of conditional fear after extinction. Learn Mem 12, 270–276, doi:12/3/270 [pii] 10.1101/lm.91705 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Pare D, Quirk GJ & Ledoux JE New vistas on amygdala networks in conditioned fear. J Neurophysiol 92, 1–9, doi: 10.1152/jn.00153.2004 92/1/1 [pii] (2004). [DOI] [PubMed] [Google Scholar]
- 113.Rougemont-Bucking A et al. Altered processing of contextual information during fear extinction in PTSD: an fMRI study. CNS Neurosci Ther 17, 227–236, doi: 10.1111/j.1755-5949.2010.00152.x (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Milad MR et al. Neurobiological basis of failure to recall extinction memory in posttraumatic stress disorder. Biol Psychiatry 66, 1075–1082, doi: 10.1016/j.biopsych.2009.06.026 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Morris JS, Ohman A & Dolan RJ Conscious and unconscious emotional learning in the human amygdala. Nature 393, 467–470, doi: 10.1038/30976 (1998). [DOI] [PubMed] [Google Scholar]
- 116.Buchel C, Dolan RJ, Armony JL & Friston KJ Amygdala-hippocampal involvement in human aversive trace conditioning revealed through event-related functional magnetic resonance imaging. J Neurosci 19, 10869–10876, doi: 10.1523/JNEUROSCI.19-24-10869.1999 (1999). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.LaBar KS, Gatenby JC, Gore JC, LeDoux JE & Phelps EA Human amygdala activation during conditioned fear acquisition and extinction: a mixed-trial fMRI study. Neuron 20, 937–945, doi: 10.1016/s0896-6273(00)80475-4 (1998). [DOI] [PubMed] [Google Scholar]
- 118.Britton JC, Phan KL, Taylor SF, Fig LM & Liberzon I Corticolimbic blood flow in posttraumatic stress disorder during script-driven imagery. Biol Psychiatry 57, 832–840, doi: 10.1016/j.biopsych.2004.12.025 (2005). [DOI] [PubMed] [Google Scholar]
- 119.Jovanovic T et al. Reduced neural activation during an inhibition task is associated with impaired fear inhibition in a traumatized civilian sample. Cortex 49, 1884–1891, doi: 10.1016/j.cortex.2012.08.011 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Lindauer RJ et al. Cerebral blood flow changes during script-driven imagery in police officers with posttraumatic stress disorder. Biol Psychiatry 56, 853–861, doi: 10.1016/j.biopsych.2004.08.003 (2004). [DOI] [PubMed] [Google Scholar]
- 121.Lebron K, Milad MR & Quirk GJ Delayed recall of fear extinction in rats with lesions of ventral medial prefrontal cortex. Learning & memory (Cold Spring Harbor, N.Y 11, 544–548, doi:11/5/544 [pii] 10.1101/lm.78604 (2004). [DOI] [PubMed] [Google Scholar]
- 122.Milad MR & Quirk GJ Neurons in medial prefrontal cortex signal memory for fear extinction. Nature 420, 70–74, doi: 10.1038/nature01138 nature01138 [pii] (2002). [DOI] [PubMed] [Google Scholar]
- 123.Sierra-Mercado D, Padilla-Coreano N & Quirk GJ Dissociable roles of prelimbic and infralimbic cortices, ventral hippocampus, and basolateral amygdala in the expression and extinction of conditioned fear. Neuropsychopharmacology 36, 529–538, doi:npp2010184 [pii] 10.1038/npp.2010.184 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Shin LM, Rauch SL & Pitman RK Amygdala, medial prefrontal cortex, and hippocampal function in PTSD. Ann N Y Acad Sci 1071, 67–79, doi: 10.1196/annals.1364.007 (2006). [DOI] [PubMed] [Google Scholar]
- 125.Liberzon I & Sripada CS The functional neuroanatomy of PTSD: a critical review. Prog Brain Res 167, 151–169, doi: 10.1016/S0079-6123(07)67011-3 (2008). [DOI] [PubMed] [Google Scholar]
- 126.Knox D, Perrine SA, George SA, Galloway MP & Liberzon I Single prolonged stress decreases glutamate, glutamine, and creatine concentrations in the rat medial prefrontal cortex. Neurosci Lett 480, 16–20, doi:S0304–3940(10)00647–6 [pii] 10.1016/j.neulet.2010.05.052 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Piggott VM et al. Single-Prolonged Stress Impairs Prefrontal Cortex Control of Amygdala and Striatum in Rats. Front Behav Neurosci 13, 18, doi: 10.3389/fnbeh.2019.00018 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ganon-Elazar E & Akirav I Cannabinoids and traumatic stress modulation of contextual fear extinction and GR expression in the amygdala-hippocampal-prefrontal circuit. Psychoneuroendocrinology 38, 1675–1687, doi: 10.1016/j.psyneuen.2013.01.014 (2013). [DOI] [PubMed] [Google Scholar]
- 129.Keller SM, Schreiber WB, Stanfield BR & Knox D Inhibiting corticosterone synthesis during fear memory formation exacerbates cued fear extinction memory deficits within the single prolonged stress model. Behav Brain Res 287, 182–186, doi: 10.1016/j.bbr.2015.03.043 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Knox D et al. Neural circuits via which single prolonged stress exposure leads to fear extinction retention deficits. Learning & memory (Cold Spring Harbor, N.Y 23, 689–698, doi: 10.1101/lm.043141.116 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Omura J et al. Chemogenetic activation of the mPFC alleviates impaired fear memory extinction in an animal model of PTSD. Prog Neuropsychopharmacol Biol Psychiatry 108, 110090, doi: 10.1016/j.pnpbp.2020.110090 (2021). [DOI] [PubMed] [Google Scholar]
- 132.Canto-de-Souza L et al. Daily Optogenetic Stimulation of the Left Infralimbic Cortex Reverses Extinction Impairments in Male Rats Exposed to Single Prolonged Stress. Front Behav Neurosci 15, 780326, doi: 10.3389/fnbeh.2021.780326 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Knox D et al. Single prolonged stress disrupts retention of extinguished fear in rats. Learning & memory (Cold Spring Harbor, N.Y 19, 43–49, doi: 10.1101/lm.024356.111 19/2/43 [pii] (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Roughley S & Killcross S Loss of Hierarchical Control by Occasion Setters Following Lesions of the Prelimbic and Infralimbic Medial Prefrontal Cortex in Rats. Brain Sci 9, doi: 10.3390/brainsci9030048 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Knox D et al. Using c-Jun to identify fear extinction learning-specific patterns of neural activity that are affected by single prolonged stress. Behav Brain Res 341, 189–197, doi: 10.1016/j.bbr.2017.12.037 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Knox D et al. PI3K-Akt Signaling in the Basolateral Amygdala Facilitates Traumatic Stress Enhancements in Fear Memory. Int J Neuropsychopharmacol 24, 229–238, doi: 10.1093/ijnp/pyaa083 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Todd TP, Fournier DI & Bucci DJ Retrosplenial cortex and its role in cue-specific learning and memory. Neurosci Biobehav Rev 107, 713–728, doi: 10.1016/j.neubiorev.2019.04.016 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Keene CS & Bucci DJ Neurotoxic lesions of retrosplenial cortex disrupt signaled and unsignaled contextual fear conditioning. Behav Neurosci 122, 1070–1077, doi: 10.1037/a0012895 (2008). [DOI] [PubMed] [Google Scholar]
- 139.Keene CS & Bucci DJ Contributions of the retrosplenial and posterior parietal cortices to cue-specific and contextual fear conditioning. Behav Neurosci 135, 693–701, doi: 10.1037/bne0000435 (2021). [DOI] [PubMed] [Google Scholar]
- 140.Todd TP, DeAngeli NE, Jiang MY & Bucci DJ Retrograde amnesia of contextual fear conditioning: Evidence for retrosplenial cortex involvement in configural processing. Behav Neurosci 131, 46–54, doi: 10.1037/bne0000183 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Robinson S et al. Chemogenetic silencing of neurons in retrosplenial cortex disrupts sensory preconditioning. J Neurosci 34, 10982–10988, doi: 10.1523/JNEUROSCI.1349-14.2014 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Van Groen T & Wyss JM Connections of the retrosplenial granular b cortex in the rat. J Comp Neurol 463, 249–263, doi: 10.1002/cne.10757 (2003). [DOI] [PubMed] [Google Scholar]
- 143.van Groen T & Wyss JM Connections of the retrosplenial granular a cortex in the rat. J Comp Neurol 300, 593–606, doi: 10.1002/cne.903000412 (1990). [DOI] [PubMed] [Google Scholar]
- 144.Chrastil ER, Sherrill KR, Hasselmo ME & Stern CE There and Back Again: Hippocampus and Retrosplenial Cortex Track Homing Distance during Human Path Integration. J Neurosci 35, 15442–15452, doi: 10.1523/JNEUROSCI.1209-15.2015 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Zaborszky L, Pang K, Somogyi J, Nadasdy Z & Kallo I The basal forebrain corticopetal system revisited. Ann N Y Acad Sci 877, 339–367 (1999). [DOI] [PubMed] [Google Scholar]
- 146.Woolf NJ & Butcher LL Cholinergic projections to the basolateral amygdala: a combined Evans Blue and acetylcholinesterase analysis. Brain Res Bull 8, 751–763 (1982). [DOI] [PubMed] [Google Scholar]
- 147.Woolf NJ, Eckenstein F & Butcher LL Cholinergic projections from the basal forebrain to the frontal cortex: a combined fluorescent tracer and immunohistochemical analysis in the rat. Neurosci Lett 40, 93–98 (1983). [DOI] [PubMed] [Google Scholar]
- 148.Semba K The cholinergic basal forebrain: a critical role in cortical arousal. Adv Exp Med Biol 295, 197–218 (1991). [DOI] [PubMed] [Google Scholar]
- 149.Jones BE Activity, modulation and role of basal forebrain cholinergic neurons innervating the cerebral cortex. Prog Brain Res 145, 157–169 (2004). [DOI] [PubMed] [Google Scholar]
- 150.Collier B, Tandon A, Prado MA & Bachoo M Storage and release of acetylcholine in a sympathetic ganglion. Prog Brain Res 98, 183–189 (1993). [DOI] [PubMed] [Google Scholar]
- 151.Birks RI & Fitch JG Storage and release of acetylcholine in a sympathetic ganglion. J Physiol 240, 125–134 (1974). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Chatonnet A & Lockridge O Comparison of butyrylcholinesterase and acetylcholinesterase. Biochem J 260, 625–634 (1989). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Ferguson SM et al. Vesicular localization and activity-dependent trafficking of presynaptic choline transporters. J Neurosci 23, 9697–9709 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Bazalakova MH & Blakely RD The high-affinity choline transporter: a critical protein for sustaining cholinergic signaling as revealed in studies of genetically altered mice. Handb Exp Pharmacol, 525–544 (2006). [DOI] [PubMed] [Google Scholar]
- 155.Cooper J, Bloom F & Roth RH Biochemical Basis of Neuropharmacology 7th edn, 528 (Oxford University Press, USA, 1996). [Google Scholar]
- 156.Benishin CG & Carroll PT Multiple forms of choline-O-acetyltransferase in mouse and rat brain: solubilization and characterization. J Neurochem 41, 1030–1039 (1983). [DOI] [PubMed] [Google Scholar]
- 157.Prado VF, Roy A, Kolisnyk B, Gros R & Prado MA Regulation of cholinergic activity by the vesicular acetylcholine transporter. Biochem J 450, 265–274, doi: 10.1042/BJ20121662 (2013). [DOI] [PubMed] [Google Scholar]
- 158.Quirion R Cholinergic markers in Alzheimer disease and the autoregulation of acetylcholine release. J Psychiatry Neurosci 18, 226–234 (1993). [PMC free article] [PubMed] [Google Scholar]
- 159.Levey AI Immunological localization of m1-m5 muscarinic acetylcholine receptors in peripheral tissues and brain. Life Sci 52, 441–448 (1993). [DOI] [PubMed] [Google Scholar]
- 160.Bonner TI The molecular basis of muscarinic receptor diversity. Trends Neurosci 12, 148–151, doi: 10.1016/0166-2236(89)90054-4 (1989). [DOI] [PubMed] [Google Scholar]
- 161.Schimerlik MI Structure and regulation of muscarinic receptors. Annu Rev Physiol 51, 217–227, doi: 10.1146/annurev.ph.51.030189.001245 (1989). [DOI] [PubMed] [Google Scholar]
- 162.Decker MW, Brioni JD, Bannon AW & Arneric SP Diversity of neuronal nicotinic acetylcholine receptors: lessons from behavior and implications for CNS therapeutics. Life Sci 56, 545–570 (1995). [DOI] [PubMed] [Google Scholar]
- 163.Davis JA & Gould TJ beta2 subunit-containing nicotinic receptors mediate the enhancing effect of nicotine on trace cued fear conditioning in C57BL/6 mice. Psychopharmacology (Berl) 190, 343–352, doi: 10.1007/s00213-006-0624-8 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.McGehee DS Molecular diversity of neuronal nicotinic acetylcholine receptors. Ann N Y Acad Sci 868, 565–577 (1999). [DOI] [PubMed] [Google Scholar]
- 165.Mesulam MM & Geula C Chemoarchitectonics of axonal and perikaryal acetylcholinesterase along information processing systems of the human cerebral cortex. Brain Res Bull 33, 137–153, doi:0361–9230(94)90244–5 [pii] (1994). [DOI] [PubMed] [Google Scholar]
- 166.Zaborszky L & Cullinan WE Direct catecholaminergic-cholinergic interactions in the basal forebrain. I. Dopamine-beta-hydroxylase- and tyrosine hydroxylase input to cholinergic neurons. J Comp Neurol 374, 535–554 (1996). [DOI] [PubMed] [Google Scholar]
- 167.Zaborszky L, Gaykema RP, Swanson DJ & Cullinan WE Cortical input to the basal forebrain. Neuroscience 79, 1051–1078, doi:S0306–4522(97)00049–3 [pii] (1997). [DOI] [PubMed] [Google Scholar]
- 168.Knox D & Keller SM Cholinergic neuronal lesions in the medial septum and vertical limb of the Diagonal Bands of Broca induce contextual fear memory generalization and impair acquisition of fear extinction. Hippocampus 26, 718–726 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Heckers S et al. Complete and selective cholinergic denervation of rat neocortex and hippocampus but not amygdala by an immunotoxin against the p75 NGF receptor. J Neurosci 14, 1271–1289 (1994). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Frick KM, Kim JJ & Baxter MG Effects of complete immunotoxin lesions of the cholinergic basal forebrain on fear conditioning and spatial learning. Hippocampus 14, 244–254 (2004). [DOI] [PubMed] [Google Scholar]
- 171.Knox D & Berntson GG Cortical modulation by nucleus basalis magnocellularis corticopetal cholinergic neurons during anxiety-like states is reflected by decreases in delta. Brain Res 1227, 142–152, doi:S0006–8993(08)01519–9 [pii] 10.1016/j.brainres.2008.06.060 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Stowell JR, Berntson GG & Sarter M Attenuation of the bidirectional effects of chlordiazepoxide and FG 7142 on conditioned response suppression and associated cardiovascular reactivity by loss of cortical cholinergic inputs. Psychopharmacology (Berl) 150, 141–149 (2000). [DOI] [PubMed] [Google Scholar]
- 173.Torres EM et al. Behavioural, histochemical and biochemical consequences of selective immunolesions in discrete regions of the basal forebrain cholinergic system. Neuroscience 63, 95–122 (1994). [DOI] [PubMed] [Google Scholar]
- 174.Knox D & Berntson GG Effect of nucleus basalis magnocellularis cholinergic lesions on fear-like and anxiety-like behavior. Behavioral Neuroscience 120, 307–312 (2006). [DOI] [PubMed] [Google Scholar]
- 175.McGaughy J, Kaiser T & Sarter M Behavioral vigilance following infusions of 192 IgG-saporin into the basal forebrain: selectivity of the behavioral impairment and relation to cortical AChE-positive fiber density. Behav Neurosci 110, 247–265 (1996). [DOI] [PubMed] [Google Scholar]
- 176.Tronson NC et al. Segregated populations of hippocampal principal CA1 neurons mediating conditioning and extinction of contextual fear. J Neurosci 29, 3387–3394, doi:29/11/3387 [pii] 10.1523/JNEUROSCI.5619-08.2009 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Schauz C & Koch M Lesions of the nucleus basalis magnocellularis do not impair prepulse inhibition and latent inhibition of fear-potentiated startle in the rat. Brain Res 815, 98–105, doi:S0006–8993(98)01134–2 [pii] (1999). [DOI] [PubMed] [Google Scholar]
- 178.Pizzo DP, Thal LJ & Winkler J Mnemonic deficits in animals depend upon the degree of cholinergic deficit and task complexity. Exp Neurol 177, 292–305 (2002). [DOI] [PubMed] [Google Scholar]
- 179.Power AE, Thal LJ & McGaugh JL Lesions of the nucleus basalis magnocellularis induced by 192 IgG-saporin block memory enhancement with posttraining norepinephrine in the basolateral amygdala. Proc Natl Acad Sci U S A 99, 2315–2319 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Knox D The role of basal forebrain cholinergic neurons in fear and extinction memory. Neurobiol Learn Mem 133, 39–52, doi: 10.1016/j.nlm.2016.06.001 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Uliana DL et al. Contextual fear expression engages a complex set of interactions between ventromedial prefrontal cortex cholinergic, glutamatergic, nitrergic and cannabinergic signaling. Neuropharmacology 232, 109538, doi: 10.1016/j.neuropharm.2023.109538 (2023). [DOI] [PubMed] [Google Scholar]
- 182.Letzkus JJ et al. A disinhibitory microcircuit for associative fear learning in the auditory cortex. Nature 480, 331–335, doi: 10.1038/nature10674 (2011). [DOI] [PubMed] [Google Scholar]
- 183.Santini E, Sepulveda-Orengo M & Porter JT Muscarinic receptors modulate the intrinsic excitability of infralimbic neurons and consolidation of fear extinction. Neuropsychopharmacology 37, 2047–2056, doi: 10.1038/npp.2012.52 npp201252 [pii] (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Crimmins BE et al. Basal forebrain cholinergic signaling in the basolateral amygdala promotes strength and durability of fear memories. Neuropsychopharmacology 48, 605–614, doi: 10.1038/s41386-022-01427-w (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Jiang L et al. Cholinergic Signaling Controls Conditioned Fear Behaviors and Enhances Plasticity of Cortical-Amygdala Circuits. Neuron, doi: 10.1016/j.neuron.2016.04.028 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Price JL & Amaral DG An autoradiographic study of the projections of the central nucleus of the monkey amygdala. J Neurosci 1, 1242–1259 (1981). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Jolkkonen E, Miettinen R, Pikkarainen M & Pitkanen A Projections from the amygdaloid complex to the magnocellular cholinergic basal forebrain in rat. Neuroscience 111, 133–149, doi:S0306452201005784 [pii] (2002). [DOI] [PubMed] [Google Scholar]
- 188.McDonald AJ Topographical organization of amygdaloid projections to the caudatoputamen, nucleus accumbens, and related striatal-like areas of the rat brain. Neuroscience 44, 15–33, doi:0306–4522(91)90248-M [pii] (1991). [DOI] [PubMed] [Google Scholar]
- 189.Petrovich GD & Swanson LW Projections from the lateral part of the central amygdalar nucleus to the postulated fear conditioning circuit. Brain Res 763, 247–254, doi:S0006–8993(96)01361–3 [pii] (1997). [DOI] [PubMed] [Google Scholar]
- 190.Wilensky AE, Schafe GE, Kristensen MP & LeDoux JE Rethinking the fear circuit: the central nucleus of the amygdala is required for the acquisition, consolidation, and expression of Pavlovian fear conditioning. J Neurosci 26, 12387–12396, doi: 10.1523/JNEUROSCI.4316-06.2006 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Goosens KA & Maren S Contextual and auditory fear conditioning are mediated by the lateral, basal, and central amygdaloid nuclei in rats. Learning & memory (Cold Spring Harbor, N.Y 8, 148–155, doi: 10.1101/lm.37601 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Shultz B, Mohammadmirzaei N & Knox D Role of nucleus basalis magnocellularis cholinergic neurons in contextual fear memory. (Addgene Reports, 2019). [Google Scholar]
- 193.Nail-Boucherie K, Dourmap N, Jaffard R & Costentin J Contextual fear conditioning is associated with an increase of acetylcholine release in the hippocampus of rat. Brain Res Cogn Brain Res 9, 193–197, doi:S0926–6410(99)00058–0 [pii] (2000). [DOI] [PubMed] [Google Scholar]
- 194.Calandreau L et al. Extracellular hippocampal acetylcholine level controls amygdala function and promotes adaptive conditioned emotional response. J Neurosci 26, 13556–13566, doi: 10.1523/JNEUROSCI.3713-06.2006 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Rogers JL & Kesner RP Cholinergic modulation of the hippocampus during encoding and retrieval of tone/shock-induced fear conditioning. Learning & memory (Cold Spring Harbor, N.Y 11, 102–107, doi: 10.1101/lm.64604 11/1/102 [pii] (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Wallenstein GV & Vago DR Intrahippocampal scopolamine impairs both acquisition and consolidation of contextual fear conditioning. Neurobiol Learn Mem 75, 245–252, doi: 10.1006/nlme.2001.4005 S1074–7427(01)94005–5 [pii] (2001). [DOI] [PubMed] [Google Scholar]
- 197.Izquierdo I et al. Neurotransmitter receptors involved in post-training memory processing by the amygdala, medial septum, and hippocampus of the rat. Behav Neural Biol 58, 16–26 (1992). [DOI] [PubMed] [Google Scholar]
- 198.Messamore E, Warpman U, Williams E & Giacobini E Muscarinic receptors mediate attenuation of extracellular acetylcholine levels in rat cerebral cortex after cholinesterase inhibition. Neurosci Lett 158, 205–208 (1993). [DOI] [PubMed] [Google Scholar]
- 199.Davis JA, Kenney JW & Gould TJ Hippocampal alpha4beta2 nicotinic acetylcholine receptor involvement in the enhancing effect of acute nicotine on contextual fear conditioning. J Neurosci 27, 10870–10877, doi:27/40/10870 [pii] 10.1523/JNEUROSCI.3242-07.2007 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Kenney JW, Raybuck JD & Gould TJ Nicotinic receptors in the dorsal and ventral hippocampus differentially modulate contextual fear conditioning. Hippocampus 22, 1681–1690, doi: 10.1002/hipo.22003 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Raybuck JD & Gould TJ The role of nicotinic acetylcholine receptors in the medial prefrontal cortex and hippocampus in trace fear conditioning. Neurobiol Learn Mem 94, 353–363, doi: 10.1016/j.nlm.2010.08.001 S1074–7427(10)00127–9 [pii] (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Hersman S & Fanselow MS in Society for Neuroscience. [Google Scholar]
- 203.Kart E et al. Neurokinin-1 receptor antagonism by SR140333: enhanced in vivo ACh in the hippocampus and promnestic post-trial effects. Peptides 25, 1959–1969, doi:S0196–9781(04)00306–7 [pii] 10.1016/j.peptides.2004.07.003 (2004). [DOI] [PubMed] [Google Scholar]
- 204.Qi Z & Gold PE Intrahippocampal infusions of anisomycin produce amnesia: contribution of increased release of norepinephrine, dopamine, and acetylcholine. Learning & memory (Cold Spring Harbor, N.Y 16, 308–314, doi: 10.1101/lm.1333409 16/5/308 [pii] (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Degroot A & Parent MB Infusions of physostigmine into the hippocampus or the entorhinal cortex attenuate avoidance retention deficits produced by intra-septal infusions of the GABA agonist muscimol. Brain Res 920, 10–18, doi:S0006–8993(01)02798–6 [pii] (2001). [DOI] [PubMed] [Google Scholar]
- 206.Khakpai F, Nasehi M, Haeri-Rohani A, Eidi A & Zarrindast MR Scopolamine induced memory impairment; possible involvement of NMDA receptor mechanisms of dorsal hippocampus and/or septum. Behav Brain Res 231, 1–10, doi: 10.1016/j.bbr.2012.02.049 S0166–4328(12)00178–7 [pii] (2012). [DOI] [PubMed] [Google Scholar]
- 207.Nail-Boucherie K, Dourmap N, Jaffard R & Costentin J The specific dopamine uptake inhibitor GBR 12783 improves learning of inhibitory avoidance and increases hippocampal acetylcholine release. Brain Res Cogn Brain Res 7, 203–205, doi:S0926–6410(98)00023–8 [pii] (1998). [DOI] [PubMed] [Google Scholar]
- 208.Parfitt GM, Campos RC, Barbosa AK, Koth AP & Barros DM Participation of hippocampal cholinergic system in memory persistence for inhibitory avoidance in rats. Neurobiol Learn Mem 97, 183–188, doi: 10.1016/j.nlm.2011.12.001 S1074–7427(11)00200–0 [pii] (2012). [DOI] [PubMed] [Google Scholar]
- 209.Zarrindast MR, Ardjmand A, Ahmadi S & Rezayof A Activation of dopamine D1 receptors in the medial septum improves scopolamine-induced amnesia in the dorsal hippocampus. Behav Brain Res 229, 68–73, doi: 10.1016/j.bbr.2011.12.033 S0166–4328(11)00907–7 [pii] (2012). [DOI] [PubMed] [Google Scholar]
- 210.Boccia MM, Blake MG, Krawczyk MC & Baratti CM Hippocampal alpha7 nicotinic receptors modulate memory reconsolidation of an inhibitory avoidance task in mice. Neuroscience 171, 531–543, doi: 10.1016/j.neuroscience.2010.08.027 S0306–4522(10)01135–8 [pii] (2010). [DOI] [PubMed] [Google Scholar]
- 211.Marti Barros D, Ramirez MR, Dos Reis EA & Izquierdo I Participation of hippocampal nicotinic receptors in acquisition, consolidation and retrieval of memory for one trial inhibitory avoidance in rats. Neuroscience 126, 651–656, doi: 10.1016/j.neuroscience.2004.03.010 S0306452204001927 [pii] (2004). [DOI] [PubMed] [Google Scholar]
- 212.Craig LA, Hong NS, Kopp J & McDonald RJ Cholinergic depletion of the medial septum followed by phase shifting does not impair memory or rest-activity rhythms measured under standard light/dark conditions in rats. Brain Res Bull 79, 53–62, doi: 10.1016/j.brainresbull.2008.10.013 S0361–9230(08)00366–3 [pii] (2009). [DOI] [PubMed] [Google Scholar]
- 213.Staib JM, Della Valle R & Knox D Medial septum and diagonal band of Broca cholinergic projections to the ventral hippocampus facilitate fear memory. Neurobiol Learn Mem ePub, doi: 10.1016/j.nlm (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Staib JM, Della Valle R & Knox DK Disruption of medial septum and diagonal bands of Broca cholinergic projections to the ventral hippocampus disrupt auditory fear memory. Neurobiol Learn Mem 152, 71–79, doi: 10.1016/j.nlm.2018.05.009 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Quirk GJ, Russo GK, Barron JL & Lebron K The role of ventromedial prefrontal cortex in the recovery of extinguished fear. J Neurosci 20, 6225–6231 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Sierra-Mercado D Jr., Corcoran KA, Lebron-Milad K & Quirk GJ Inactivation of the ventromedial prefrontal cortex reduces expression of conditioned fear and impairs subsequent recall of extinction. Eur J Neurosci 24, 1751–1758 (2006). [DOI] [PubMed] [Google Scholar]
- 217.Corcoran KA, Desmond TJ, Frey KA & Maren S Hippocampal inactivation disrupts the acquisition and contextual encoding of fear extinction. J Neurosci 25, 8978–8987, doi:25/39/8978 [pii] 10.1523/JNEUROSCI.2246-05.2005 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Abdulkhaleq LA et al. The crucial roles of inflammatory mediators in inflammation: A review. Vet World 11, 627–635, doi: 10.14202/vetworld.2018.627-635 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Koj A Initiation of acute phase response and synthesis of cytokines. Biochim Biophys Acta 1317, 84–94, doi: 10.1016/s0925-4439(96)00048-8 (1996). [DOI] [PubMed] [Google Scholar]
- 220.Mittal M, Siddiqui MR, Tran K, Reddy SP & Malik AB Reactive oxygen species in inflammation and tissue injury. Antioxid Redox Signal 20, 1126–1167, doi: 10.1089/ars.2012.5149 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Li S et al. Microglial deletion and inhibition alleviate behavior of post-traumatic stress disorder in mice. J Neuroinflammation 18, 7, doi: 10.1186/s12974-020-02069-9 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Li B, Zhang D & Verkhratsky A Astrocytes in Post-traumatic Stress Disorder. Neurosci Bull 38, 953–965, doi: 10.1007/s12264-022-00845-6 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Singh D Astrocytic and microglial cells as the modulators of neuroinflammation in Alzheimer’s disease. J Neuroinflammation 19, 206, doi: 10.1186/s12974-022-02565-0 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Dantzer R, O’Connor JC, Freund GG, Johnson RW & Kelley KW From inflammation to sickness and depression: when the immune system subjugates the brain. Nat Rev Neurosci 9, 46–56, doi: 10.1038/nrn2297 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Erickson MA, Dohi K & Banks WA Neuroinflammation: a common pathway in CNS diseases as mediated at the blood-brain barrier. Neuroimmunomodulation 19, 121–130, doi: 10.1159/000330247 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Hori H & Kim Y Inflammation and post-traumatic stress disorder. Psychiatry Clin Neurosci 73, 143–153, doi: 10.1111/pcn.12820 (2019). [DOI] [PubMed] [Google Scholar]
- 227.Katrinli S, Oliveira NCS, Felger JC, Michopoulos V & Smith AK The role of the immune system in posttraumatic stress disorder. Transl Psychiatry 12, 313, doi: 10.1038/s41398-022-02094-7 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Passos IC et al. Inflammatory markers in post-traumatic stress disorder: a systematic review, meta-analysis, and meta-regression. Lancet Psychiatry 2, 1002–1012, doi: 10.1016/S2215-0366(15)00309-0 (2015). [DOI] [PubMed] [Google Scholar]
- 229.Eraly SA et al. Assessment of plasma C-reactive protein as a biomarker of posttraumatic stress disorder risk. JAMA Psychiatry 71, 423–431, doi: 10.1001/jamapsychiatry.2013.4374 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Cohen M et al. Cytokine levels as potential biomarkers for predicting the development of posttraumatic stress symptoms in casualties of accidents. Int J Psychiatry Med 42, 117–131, doi: 10.2190/PM.42.2.b (2011). [DOI] [PubMed] [Google Scholar]
- 231.de Oliveira JF et al. Serum levels of interleukins IL-6 and IL-10 in individuals with posttraumatic stress disorder in a population-based sample. Psychiatry Res 260, 111–115, doi: 10.1016/j.psychres.2017.11.061 (2018). [DOI] [PubMed] [Google Scholar]
- 232.Koirala R et al. Association of pro-inflammatory cytokines with trauma and post-traumatic stress disorder visiting a tertiary care hospital in Kathmandu. PLoS One 18, e0281125, doi: 10.1371/journal.pone.0281125 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Lindqvist D et al. Increased pro-inflammatory milieu in combat related PTSD - A new cohort replication study. Brain Behav Immun 59, 260–264, doi: 10.1016/j.bbi.2016.09.012 (2017). [DOI] [PubMed] [Google Scholar]
- 234.Wang Z, Mandel H, Levingston CA & Young MRI An exploratory approach demonstrating immune skewing and a loss of coordination among cytokines in plasma and saliva of Veterans with combat-related PTSD. Hum Immunol 77, 652–657, doi: 10.1016/j.humimm.2016.05.018 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Yang JJ & Jiang W Immune biomarkers alterations in post-traumatic stress disorder: A systematic review and meta-analysis. J Affect Disord 268, 39–46, doi: 10.1016/j.jad.2020.02.044 (2020). [DOI] [PubMed] [Google Scholar]
- 236.Bhatt S et al. PTSD is associated with neuroimmune suppression: evidence from PET imaging and postmortem transcriptomic studies. Nat Commun 11, 2360, doi: 10.1038/s41467-020-15930-5 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Toczek J et al. FDG PET imaging of vascular inflammation in post-traumatic stress disorder: A pilot case-control study. J Nucl Cardiol 28, 688–694, doi: 10.1007/s12350-019-01724-w (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Karanikas E, Daskalakis NP & Agorastos A Oxidative Dysregulation in Early Life Stress and Posttraumatic Stress Disorder: A Comprehensive Review. Brain Sci 11, doi: 10.3390/brainsci11060723 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Atli A et al. Altered lipid peroxidation markers are related to post-traumatic stress disorder (PTSD) and not trauma itself in earthquake survivors. Eur Arch Psychiatry Clin Neurosci 266, 329–336, doi: 10.1007/s00406-015-0638-5 (2016). [DOI] [PubMed] [Google Scholar]
- 240.Glatt SJ et al. Blood-based gene-expression predictors of PTSD risk and resilience among deployed marines: a pilot study. Am J Med Genet B Neuropsychiatr Genet 162B, 313–326, doi: 10.1002/ajmg.b.32167 (2013). [DOI] [PubMed] [Google Scholar]
- 241.Zieker J et al. Differential gene expression in peripheral blood of patients suffering from post-traumatic stress disorder. Mol Psychiatry 12, 116–118, doi: 10.1038/sj.mp.4001905 (2007). [DOI] [PubMed] [Google Scholar]
- 242.Logue MW et al. A genome-wide association study of post-traumatic stress disorder identifies the retinoid-related orphan receptor alpha (RORA) gene as a significant risk locus. Mol Psychiatry 18, 937–942, doi: 10.1038/mp.2012.113 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Miller MW, Wolf EJ, Logue MW & Baldwin CT The retinoid-related orphan receptor alpha (RORA) gene and fear-related psychopathology. J Affect Disord 151, 702–708, doi: 10.1016/j.jad.2013.07.022 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Roy A et al. Reactive oxygen species up-regulate CD11b in microglia via nitric oxide: Implications for neurodegenerative diseases. Free Radic Biol Med 45, 686–699, doi: 10.1016/j.freeradbiomed.2008.05.026 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Salminen LE & Paul RH Oxidative stress and genetic markers of suboptimal antioxidant defense in the aging brain: a theoretical review. Rev Neurosci 25, 805–819, doi: 10.1515/revneuro-2014-0046 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Woodburn SC, Bollinger JL & Wohleb ES The semantics of microglia activation: neuroinflammation, homeostasis, and stress. J Neuroinflammation 18, 258, doi: 10.1186/s12974-021-02309-6 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Miller MW, Lin AP, Wolf EJ & Miller DR Oxidative Stress, Inflammation, and Neuroprogression in Chronic PTSD. Harv Rev Psychiatry 26, 57–69, doi: 10.1097/HRP.0000000000000167 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.de Munter J et al. Increased Oxidative Stress in the Prefrontal Cortex as a Shared Feature of Depressive- and PTSD-Like Syndromes: Effects of a Standardized Herbal Antioxidant. Front Nutr 8, 661455, doi: 10.3389/fnut.2021.661455 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Wilson CB et al. Inflammation and oxidative stress are elevated in the brain, blood, and adrenal glands during the progression of post-traumatic stress disorder in a predator exposure animal model. PLoS One 8, e76146, doi: 10.1371/journal.pone.0076146 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Zimmerman G et al. Post-traumatic anxiety associates with failure of the innate immune receptor TLR9 to evade the pro-inflammatory NFkappaB pathway. Transl Psychiatry 2, e78, doi: 10.1038/tp.2012.4 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Jia Y, Han Y, Wang X & Han F Role of apoptosis in the Post-traumatic stress disorder model-single prolonged stressed rats. Psychoneuroendocrinology 95, 97–105, doi: 10.1016/j.psyneuen.2018.05.015 (2018). [DOI] [PubMed] [Google Scholar]
- 252.Muhie S et al. Molecular indicators of stress-induced neuroinflammation in a mouse model simulating features of post-traumatic stress disorder. Transl Psychiatry 7, e1135, doi: 10.1038/tp.2017.91 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Guevara CA, Del Valle P & Mercedes CR Microglia and Reactive Oxygen Species Are Required for Behavioral Susceptibility to Chronic Social Defeat Stress. J Neurosci 40, 1370–1372, doi: 10.1523/JNEUROSCI.2175-19.2019 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Lehmann ML, Weigel TK, Poffenberger CN & Herkenham M The Behavioral Sequelae of Social Defeat Require Microglia and Are Driven by Oxidative Stress in Mice. J Neurosci 39, 5594–5605, doi: 10.1523/JNEUROSCI.0184-19.2019 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Jones ME, Lebonville CL, Barrus D & Lysle DT The role of brain interleukin-1 in stress-enhanced fear learning. Neuropsychopharmacology 40, 1289–1296, doi: 10.1038/npp.2014.317 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Torres-Rodriguez O et al. Sex-dependent effects of microglial reduction on impaired fear extinction induced by single prolonged stress. Front Behav Neurosci 16, 1014767, doi: 10.3389/fnbeh.2022.1014767 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Lee DH et al. Neuroinflammation in Post-Traumatic Stress Disorder. Biomedicines 10, doi: 10.3390/biomedicines10050953 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Khatri N et al. Oxidative Stress: Major Threat in Traumatic Brain Injury. CNS Neurol Disord Drug Targets 17, 689–695, doi: 10.2174/1871527317666180627120501 (2018). [DOI] [PubMed] [Google Scholar]
- 259.Radi E, Formichi P, Battisti C & Federico A Apoptosis and oxidative stress in neurodegenerative diseases. J Alzheimers Dis 42 Suppl 3, S125–152, doi: 10.3233/JAD-132738 (2014). [DOI] [PubMed] [Google Scholar]
- 260.Raghupathi R, Graham DI & McIntosh TK Apoptosis after traumatic brain injury. J Neurotrauma 17, 927–938, doi: 10.1089/neu.2000.17.927 (2000). [DOI] [PubMed] [Google Scholar]
- 261.Li XM, Han F, Liu DJ & Shi YX Single-prolonged stress induced mitochondrial-dependent apoptosis in hippocampus in the rat model of post-traumatic stress disorder. J Chem Neuroanat 40, 248–255, doi: 10.1016/j.jchemneu.2010.07.001 (2010). [DOI] [PubMed] [Google Scholar]
- 262.Han F, Yan S & Shi Y Single-prolonged stress induces endoplasmic reticulum-dependent apoptosis in the hippocampus in a rat model of post-traumatic stress disorder. PLoS One 8, e69340, doi: 10.1371/journal.pone.0069340 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Wang H et al. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature 421, 384–388, doi: 10.1038/nature01339 (2003). [DOI] [PubMed] [Google Scholar]
- 264.Gamage R et al. Cholinergic Modulation of Glial Function During Aging and Chronic Neuroinflammation. Front Cell Neurosci 14, 577912, doi: 10.3389/fncel.2020.577912 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Halder N & Lal G Cholinergic System and Its Therapeutic Importance in Inflammation and Autoimmunity. Front Immunol 12, 660342, doi: 10.3389/fimmu.2021.660342 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Pavlov VA, Wang H, Czura CJ, Friedman SG & Tracey KJ The cholinergic anti-inflammatory pathway: a missing link in neuroimmunomodulation. Mol Med 9, 125–134 (2003). [PMC free article] [PubMed] [Google Scholar]
- 267.Skok M, Grailhe R & Changeux JP Nicotinic receptors regulate B lymphocyte activation and immune response. Eur J Pharmacol 517, 246–251, doi: 10.1016/j.ejphar.2005.05.011 (2005). [DOI] [PubMed] [Google Scholar]
- 268.Borovikova LV et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405, 458–462, doi: 10.1038/35013070 (2000). [DOI] [PubMed] [Google Scholar]
- 269.Rosas-Ballina M et al. The selective alpha7 agonist GTS-21 attenuates cytokine production in human whole blood and human monocytes activated by ligands for TLR2, TLR3, TLR4, TLR9, and RAGE. Mol Med 15, 195–202, doi: 10.2119/molmed.2009.00039 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Mizrachi T et al. Suppression of neuroinflammation by an allosteric agonist and positive allosteric modulator of the alpha7 nicotinic acetylcholine receptor GAT107. J Neuroinflammation 18, 99, doi: 10.1186/s12974-021-02149-4 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Fujii YX et al. Enhanced serum antigen-specific IgG1 and proinflammatory cytokine production in nicotinic acetylcholine receptor alpha7 subunit gene knockout mice. J Neuroimmunol 189, 69–74, doi: 10.1016/j.jneuroim.2007.07.003 (2007). [DOI] [PubMed] [Google Scholar]
- 272.de Jonge WJ et al. Stimulation of the vagus nerve attenuates macrophage activation by activating the Jak2-STAT3 signaling pathway. Nat Immunol 6, 844–851, doi: 10.1038/ni1229 (2005). [DOI] [PubMed] [Google Scholar]
- 273.Piovesana R, Salazar Intriago MS, Dini L & Tata AM Cholinergic Modulation of Neuroinflammation: Focus on alpha7 Nicotinic Receptor. Int J Mol Sci 22, doi: 10.3390/ijms22094912 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Shytle RD et al. Cholinergic modulation of microglial activation by alpha 7 nicotinic receptors. J Neurochem 89, 337–343, doi: 10.1046/j.1471-4159.2004.02347.x (2004). [DOI] [PubMed] [Google Scholar]
- 275.Suzuki T et al. Microglial alpha7 nicotinic acetylcholine receptors drive a phospholipase C/IP3 pathway and modulate the cell activation toward a neuroprotective role. J Neurosci Res 83, 1461–1470, doi: 10.1002/jnr.20850 (2006). [DOI] [PubMed] [Google Scholar]
- 276.Parada E et al. The microglial alpha7-acetylcholine nicotinic receptor is a key element in promoting neuroprotection by inducing heme oxygenase-1 via nuclear factor erythroid-2-related factor 2. Antioxid Redox Signal 19, 1135–1148, doi: 10.1089/ars.2012.4671 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Patel H, McIntire J, Ryan S, Dunah A & Loring R Anti-inflammatory effects of astroglial alpha7 nicotinic acetylcholine receptors are mediated by inhibition of the NF-kappaB pathway and activation of the Nrf2 pathway. J Neuroinflammation 14, 192, doi: 10.1186/s12974-017-0967-6 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Revathikumar P et al. Immunomodulatory effects of nicotine on interleukin 1beta activated human astrocytes and the role of cyclooxygenase 2 in the underlying mechanism. J Neuroinflammation 13, 256, doi: 10.1186/s12974-016-0725-1 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Liu Y et al. alpha7 nicotinic acetylcholine receptor-mediated neuroprotection against dopaminergic neuron loss in an MPTP mouse model via inhibition of astrocyte activation. J Neuroinflammation 9, 98, doi: 10.1186/1742-2094-9-98 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Hua Y et al. Activation of alpha7 Nicotinic Acetylcholine Receptor Protects Against 1-Methyl-4-Phenylpyridinium-Induced Astroglial Apoptosis. Front Cell Neurosci 13, 507, doi: 10.3389/fncel.2019.00507 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Mizrachi T, Vaknin-Dembinsky A, Brenner T & Treinin M Neuroinflammation Modulation via alpha7 Nicotinic Acetylcholine Receptor and Its Chaperone, RIC-3. Molecules 26, doi: 10.3390/molecules26206139 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Medeiros R et al. alpha7 Nicotinic receptor agonist enhances cognition in aged 3xTg-AD mice with robust plaques and tangles. Am J Pathol 184, 520–529, doi: 10.1016/j.ajpath.2013.10.010 (2014). [DOI] [PubMed] [Google Scholar]
- 283.Takata K et al. Alpha7 nicotinic acetylcholine receptor-specific agonist DMXBA (GTS-21) attenuates Abeta accumulation through suppression of neuronal gamma-secretase activity and promotion of microglial amyloid-beta phagocytosis and ameliorates cognitive impairment in a mouse model of Alzheimer’s disease. Neurobiol Aging 62, 197–209, doi: 10.1016/j.neurobiolaging.2017.10.021 (2018). [DOI] [PubMed] [Google Scholar]
- 284.Takata K et al. Nicotinic Acetylcholine Receptors and Microglia as Therapeutic and Imaging Targets in Alzheimer’s Disease. Molecules 27, doi: 10.3390/molecules27092780 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Binning W et al. Chronic hM3Dq signaling in microglia ameliorates neuroinflammation in male mice. Brain Behav Immun 88, 791–801, doi: 10.1016/j.bbi.2020.05.041 (2020). [DOI] [PubMed] [Google Scholar]
- 286.Frinchi M et al. Anti-inflammatory and antioxidant effects of muscarinic acetylcholine receptor (mAChR) activation in the rat hippocampus. Sci Rep 9, 14233, doi: 10.1038/s41598-019-50708-w (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Martin A et al. In vivo PET imaging of the alpha4beta2 nicotinic acetylcholine receptor as a marker for brain inflammation after cerebral ischemia. J Neurosci 35, 5998–6009, doi: 10.1523/JNEUROSCI.3670-14.2015 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Wenk GL et al. Mechanisms to prevent the toxicity of chronic neuroinflammation on forebrain cholinergic neurons. Eur J Pharmacol 402, 77–85, doi: 10.1016/s0014-2999(00)00523-9 (2000). [DOI] [PubMed] [Google Scholar]
- 289.von Bernhardi R, Eugenin-von Bernhardi L & Eugenin J Microglial cell dysregulation in brain aging and neurodegeneration. Front Aging Neurosci 7, 124, doi: 10.3389/fnagi.2015.00124 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Nedogreeva OA et al. Oxidative Damage of Proteins Precedes Loss of Cholinergic Phenotype in the Septal Neurons of Olfactory Bulbectomized Mice. Curr Alzheimer Res 18, 1140–1151, doi: 10.2174/1567205019666211223094051 (2021). [DOI] [PubMed] [Google Scholar]
- 291.Campanucci VA, Krishnaswamy A & Cooper E Mitochondrial reactive oxygen species inactivate neuronal nicotinic acetylcholine receptors and induce long-term depression of fast nicotinic synaptic transmission. J Neurosci 28, 1733–1744, doi: 10.1523/JNEUROSCI.5130-07.2008 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Mattson MP & Pedersen WA Effects of amyloid precursor protein derivatives and oxidative stress on basal forebrain cholinergic systems in Alzheimer’s disease. Int J Dev Neurosci 16, 737–753, doi: 10.1016/s0736-5748(98)00082-3 (1998). [DOI] [PubMed] [Google Scholar]
- 293.Fagan AM, Garber M, Barbacid M, Silos-Santiago I & Holtzman DM A role for TrkA during maturation of striatal and basal forebrain cholinergic neurons in vivo. J Neurosci 17, 7644–7654, doi: 10.1523/JNEUROSCI.17-20-07644.1997 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Kropf E & Fahnestock M Effects of Reactive Oxygen and Nitrogen Species on TrkA Expression and Signalling: Implications for proNGF in Aging and Alzheimer’s Disease. Cells 10, doi: 10.3390/cells10081983 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Lan Z et al. Paeoniflorin attenuates amyloid-beta peptide-induced neurotoxicity by ameliorating oxidative stress and regulating the NGF-mediated signaling in rats. Brain Res 1498, 9–19, doi: 10.1016/j.brainres.2012.12.040 (2013). [DOI] [PubMed] [Google Scholar]
- 296.Parikh V et al. Diminished trkA receptor signaling reveals cholinergic-attentional vulnerability of aging. Eur J Neurosci 37, 278–293, doi: 10.1111/ejn.12090 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Yegla B & Parikh V Developmental suppression of forebrain trkA receptors and attentional capacities in aging rats: A longitudinal study. Behav Brain Res 335, 111–121, doi: 10.1016/j.bbr.2017.08.017 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]


